User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
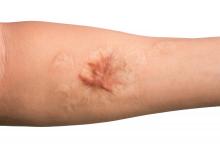
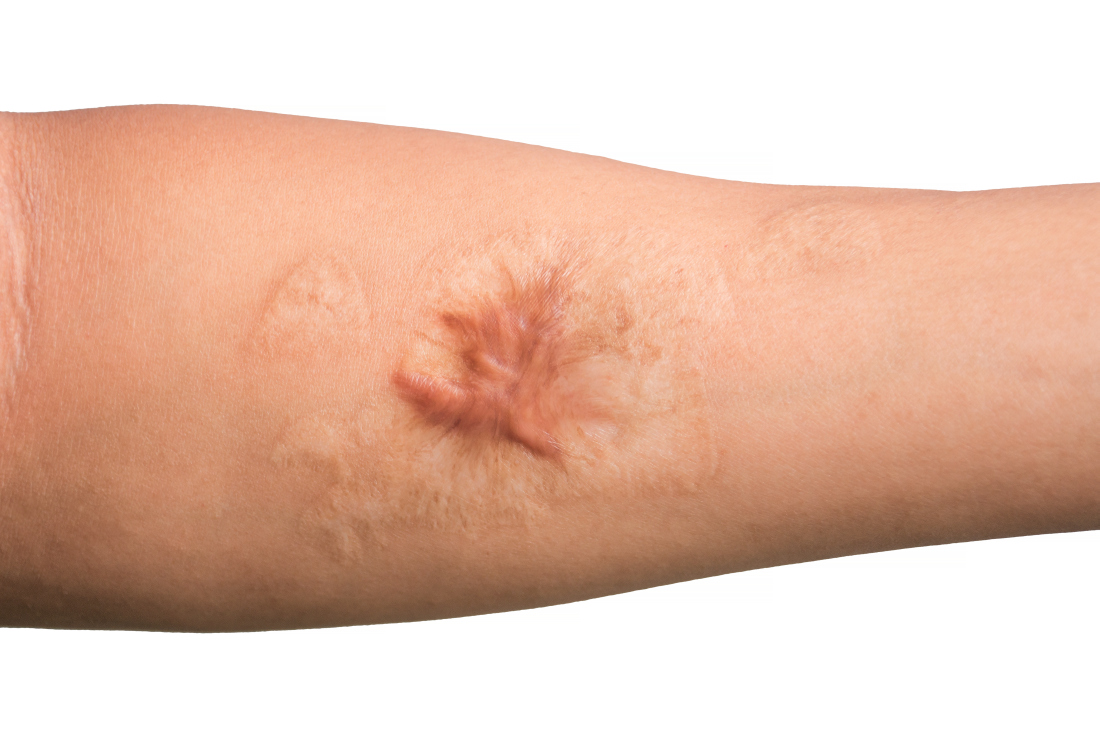
Combo of excision, cryosurgery found to benefit keloid scar outcomes
Treating keloid scars by combining excision and contact cryosurgery is a plausible way to decrease the volume of scars, results from a single-center observational study suggest.
“There is currently no consensus regarding the best treatment of keloid scars,” corresponding author Manon Artz, of the department of plastic, reconstructive, and aesthetic surgery at University Hospital of Brest (France), and colleagues wrote in a research letter published online in JAMA Dermatology.
“Earlier studies report a decreased scar volume and a substantial reduction of recurrence in keloid scars treated by cryosurgery,” they wrote. “In this study, our objective was to assess whether intramarginal excision (shaving) of the keloid scar followed by an immediate single session of contact cryosurgery is associated with decreased scar volume.”
Between March 2014 and May 2020, the researchers evaluated the approach in 31 patients with 40 keloid scars who were treated at University Hospital of Brest. Of these study participants, four were lost to follow-up, leaving 27 patients with 35 keloid scars in the final analysis. Their mean age was 24 years, 60% were female, and there was fairly even distribution of Fitzpatrick skin types II-VI.
Most of the keloid scars were located on the ear (69%) and the chest (23%), while the rest were on the head and neck. The primary outcome was reduction of keloid scar volume after 12 months, which was measured with the Vancouver scar scale. The researchers defined 80%-100% reduction in scar volume as “major,” a 50%-80% reduction as “substantial,” and a 0%-50% reduction or recurrence as “moderate.”
After 12 months, 19 scars (54%) showed a major reduction in volume, while 6 (17%) had a substantial reduction, and seven (20%) experienced no reduction. Across all keloid scars, the median scar volume decreased significantly by 81.9%.
Scar volume reduction differed by anatomical location. Specifically, 84% of ear scars showed major or substantial reduction, while 60% of scars on the chest showed a moderate reduction in scar volume or recurrence. In another key finding, the Vancouver scar scale score was reduced overall in 25 scars by 71.4%, from 7 before treatment to 5 after treatment.
“There remains no silver bullet for the treatment of keloids, but this study adds invaluable evidence that tangential excision followed by contact cryosurgery can be a viable treatment regimen with low recurrence rates,” said Marcus G. Tan, MD, who recently completed his dermatology residency at the University of Ottawa and who was asked to comment on the study. “Clinicians should exercise caution especially when treating individuals with darker skin phototypes due to their increased risk of scarring and dyspigmentation.”
Limitations of this study, he said, include a smaller study population with some patient dropouts and a lack of adverse effects reported.
The researchers and Dr. Tan reported having no financial conflicts.
Treating keloid scars by combining excision and contact cryosurgery is a plausible way to decrease the volume of scars, results from a single-center observational study suggest.
“There is currently no consensus regarding the best treatment of keloid scars,” corresponding author Manon Artz, of the department of plastic, reconstructive, and aesthetic surgery at University Hospital of Brest (France), and colleagues wrote in a research letter published online in JAMA Dermatology.
“Earlier studies report a decreased scar volume and a substantial reduction of recurrence in keloid scars treated by cryosurgery,” they wrote. “In this study, our objective was to assess whether intramarginal excision (shaving) of the keloid scar followed by an immediate single session of contact cryosurgery is associated with decreased scar volume.”
Between March 2014 and May 2020, the researchers evaluated the approach in 31 patients with 40 keloid scars who were treated at University Hospital of Brest. Of these study participants, four were lost to follow-up, leaving 27 patients with 35 keloid scars in the final analysis. Their mean age was 24 years, 60% were female, and there was fairly even distribution of Fitzpatrick skin types II-VI.
Most of the keloid scars were located on the ear (69%) and the chest (23%), while the rest were on the head and neck. The primary outcome was reduction of keloid scar volume after 12 months, which was measured with the Vancouver scar scale. The researchers defined 80%-100% reduction in scar volume as “major,” a 50%-80% reduction as “substantial,” and a 0%-50% reduction or recurrence as “moderate.”
After 12 months, 19 scars (54%) showed a major reduction in volume, while 6 (17%) had a substantial reduction, and seven (20%) experienced no reduction. Across all keloid scars, the median scar volume decreased significantly by 81.9%.
Scar volume reduction differed by anatomical location. Specifically, 84% of ear scars showed major or substantial reduction, while 60% of scars on the chest showed a moderate reduction in scar volume or recurrence. In another key finding, the Vancouver scar scale score was reduced overall in 25 scars by 71.4%, from 7 before treatment to 5 after treatment.
“There remains no silver bullet for the treatment of keloids, but this study adds invaluable evidence that tangential excision followed by contact cryosurgery can be a viable treatment regimen with low recurrence rates,” said Marcus G. Tan, MD, who recently completed his dermatology residency at the University of Ottawa and who was asked to comment on the study. “Clinicians should exercise caution especially when treating individuals with darker skin phototypes due to their increased risk of scarring and dyspigmentation.”
Limitations of this study, he said, include a smaller study population with some patient dropouts and a lack of adverse effects reported.
The researchers and Dr. Tan reported having no financial conflicts.
Treating keloid scars by combining excision and contact cryosurgery is a plausible way to decrease the volume of scars, results from a single-center observational study suggest.
“There is currently no consensus regarding the best treatment of keloid scars,” corresponding author Manon Artz, of the department of plastic, reconstructive, and aesthetic surgery at University Hospital of Brest (France), and colleagues wrote in a research letter published online in JAMA Dermatology.
“Earlier studies report a decreased scar volume and a substantial reduction of recurrence in keloid scars treated by cryosurgery,” they wrote. “In this study, our objective was to assess whether intramarginal excision (shaving) of the keloid scar followed by an immediate single session of contact cryosurgery is associated with decreased scar volume.”
Between March 2014 and May 2020, the researchers evaluated the approach in 31 patients with 40 keloid scars who were treated at University Hospital of Brest. Of these study participants, four were lost to follow-up, leaving 27 patients with 35 keloid scars in the final analysis. Their mean age was 24 years, 60% were female, and there was fairly even distribution of Fitzpatrick skin types II-VI.
Most of the keloid scars were located on the ear (69%) and the chest (23%), while the rest were on the head and neck. The primary outcome was reduction of keloid scar volume after 12 months, which was measured with the Vancouver scar scale. The researchers defined 80%-100% reduction in scar volume as “major,” a 50%-80% reduction as “substantial,” and a 0%-50% reduction or recurrence as “moderate.”
After 12 months, 19 scars (54%) showed a major reduction in volume, while 6 (17%) had a substantial reduction, and seven (20%) experienced no reduction. Across all keloid scars, the median scar volume decreased significantly by 81.9%.
Scar volume reduction differed by anatomical location. Specifically, 84% of ear scars showed major or substantial reduction, while 60% of scars on the chest showed a moderate reduction in scar volume or recurrence. In another key finding, the Vancouver scar scale score was reduced overall in 25 scars by 71.4%, from 7 before treatment to 5 after treatment.
“There remains no silver bullet for the treatment of keloids, but this study adds invaluable evidence that tangential excision followed by contact cryosurgery can be a viable treatment regimen with low recurrence rates,” said Marcus G. Tan, MD, who recently completed his dermatology residency at the University of Ottawa and who was asked to comment on the study. “Clinicians should exercise caution especially when treating individuals with darker skin phototypes due to their increased risk of scarring and dyspigmentation.”
Limitations of this study, he said, include a smaller study population with some patient dropouts and a lack of adverse effects reported.
The researchers and Dr. Tan reported having no financial conflicts.
FROM JAMA DERMATOLOGY
Why it’s so hard to prevent physician suicide
Kip Wenger, DO, an emergency physician and systems medical director of Team Health, Knoxville, Tenn., was asked to see a patient in the emergency department. He was shocked when he realized who the patient was – a 33-year-old female physician friend and colleague.
She was bleeding from multiple self-inflicted injuries and ultimately died. “I was devastated and couldn’t wrap my head around what had just happened,” Dr. Wenger told this news organization.
It’s important for physicians to be aware of warning signs in their colleagues, such as showing up late, being irritable and short-tempered with staff, missing shifts, making mistakes, or receiving an increasing number of patient complaints, Dr. Wenger says.
Dr. Wenger had had dinner with her several weeks earlier and saw some subtle changes. He had known her as a “positive, upbeat person,” but her demeanor was different during dinner.
“There were no typical telltale signs – she was talking about her plans for the future, including buying a new bicycle – but she wasn’t herself and seemed to become tearful when I hugged her at the end of the evening,” he said. He later heard from another colleague that she had shared feeling “hopeless.”
The scope of the problem
According to the American Society for Suicide Prevention, roughly 300-400 physicians die by suicide annually. Although one study suggests a lower number, official reports likely underestimate suicides, study author Katherine Gold, MD, MSW, associate professor of family medicine, obstetrics, and gynecology, Michigan Medicine, University of Michigan, Ann Arbor, said in an interview.
Peter Yellowlees, MD, MBBS, professor of psychiatry, University of California, Davis, concurs, suggesting that some single-car accidents involving physicians might be suicides. Perry Lin, MD, assistant clinical professor, Heritage College of Osteopathic Medicine, Ohio University, Athens, and national co-chair of the Physician Suicide Awareness Committee of the American Association of Suicidology, says that some death certificates state that the deceased died of “accidental causes” because the physician who completes the certificate, possibly a colleague, is reluctant to list the actual cause of death to protect his colleague’s memory or the family’s feelings.
In general, and among physicians, White men older than 65 “represent the largest percentage of people who die from suicide nationwide,” says Dr. Lin.
But younger people are also susceptible, Dr. Lin adds. One of the most vulnerable periods for potential suicide is during the first few months of residency. This dovetails with the findings of Medscape’s 2022 report Suicide: A Tragedy of the Profession. In that report, a difference was found between frequency of suicidal thoughts in younger physicians, compared with older physicians (14% in those < 35 years vs. 8% for those ≥ 45 years).
Hurdles to preventing physician suicide
“The best thing that can happen in our profession is upstream intervention – if people seek help before they get to the point of suicidality, recognizing they’re under stress and duress and that they might be going down a bad pathway,” says Dr. Lin. But research suggests that many physicians don’t do so.
Gary Price, MD, attending surgeon and clinical assistant professor of surgery, Yale–New Haven Hospital, Connecticut, and president of the Physicians Foundation, says his organization has identified barriers that prevent physicians from seeking help.
Physicians feel they may put their licensure at risk if they admit to receiving help for mental issues. These concerns were expressed by respondents in Medscape’s above mentioned 2022 report, many of whom didn’t seek treatment for depression, burnout, or suicidal thoughts lest it affect their professional standing when renewing their license or seeking credentialing.
Although organizations and societies are advocating against these questions, a recent study found that almost 70% of U.S. states and territories continue to ask physicians about their mental health, and 28% ask for diagnoses (beyond current impairments) – a violation of the Americans With Disabilities Act.
“Mental health illness is different from mental health impairment,” Ryan Mire, MD, a Nashville, Tenn.–based internist, said in an interview. “As physicians, we’re comfortable with licensing boards asking whether the physician has any condition that might impair their care for patients, but not about a history of mental illness.”
The second barrier, says Dr. Price, is that hospital credentialing committees sometimes ask similar questions, as do commercial and malpractice insurers.
Another roadblock is that in some states, undergoing treatment for a mental health problem could be subject to discovery by a plaintiff’s attorney in a malpractice case, even if the physician’s mental health history had no effect on patient care. But that’s uncommon, says Daniel Shapiro, PhD, author of “Delivering Doctor Amelia,” a book about his treatment of a suicidal physician who underwent a malpractice lawsuit. “I’ve never seen that happen.”
A final barrier is that many employers require employees to receive treatment within their own institution or health system. “Physicians may be reluctant to get help where they work, with colleagues and friends knowing about their illness or being involved with their care,” says Dr. Price.
In 2022, the American College of Physicians (ACP) issued a toolkit to help members encourage licensing and credentialing boards to remove questions about mental health on applications and include language that supports receiving treatment, Dr. Mire says.
Layers of vulnerability
There are few data regarding relative risk among particular races or ethnicities, “but we know racism is a social stressor,” says Dr. Mire. “Obviously, people from historically disadvantaged populations tend to have societal stressors like discrimination and racism that add an extra layer of burden.”
Intersectionality – having multiple intersecting risk factors – may confer even higher risk. “For example, if you’re a female physician from a historically marginalized race and a resident dealing with the ‘hidden curriculum’ of trying to be resilient, you have multiple layers of vulnerability.”
There are also limited data regarding which specialties or work environments are associated with highest risk. “Obviously, challenges exist in every segment of medicine and at different ages, stages, and work environments, and they intersect with each individual physician’s personal risk factors,” says Dr. Mire, president of the ACP and assistant clinical professor of clinical medical education, University of Tennessee Health Science Center, Memphis.
Pamela Wible, MD, is an Oregon-based retired physician who herself went through a suicidal period about 11 years into her career that motivated her to embrace a new vision of clinical practice and change her practice model. After a series of physician suicides in her area, she began to speak and write openly about physician suicide, and since her retirement from clinical practice, she makes herself available on a full-time basis to distressed physicians. “When I address a conference of a particular medical specialty or a group in a particular geographical region, I focus on the specific vulnerabilities in that specialty or region,” she says.
What increases the chances of suicide?
“Many factors, both within and outside the professional setting, affect someone’s decision to die by suicide – after all, physicians have the same stressors as other people, like family, finances, and their own health,” Dr. Mire says. When it comes to non–work-related factors, marital stressors and comorbid psychiatric illness particularly raise the risk, says Dr. Lin.
But certain drivers are specific to the practice of medicine, with burnout and depression first in line.
Dr. Shapiro, who is vice dean for faculty and administrative affairs, Penn State University, Hershey, and the Garner James Cline Professor of Medical Humanism, conducts burnout evaluations throughout the country. “Simple depression screeners prior to the pandemic showed about a 10% major depression rate in physicians,” he told this news organization. “Now, we’re seeing a 30%-33% depression rate, even in those who weren’t frontline providers during the pandemic.”
Dr. Price agrees, noting that burnout in physicians has gone from 40% to 60% since the pandemic. But burnout doesn’t always lead to suicide. It’s when burnout progresses to depression, becomes more severe, and is untreated that the suicidal risk arises, he emphasizes.
Additionally, being a doctor isn’t “just a profession” but a “calling and identity,” says Dr. Gold. Job-related problems (for example, a malpractice suit, complaints to the medical board, loss of autonomy, changing work demands) can raise suicidal risk.
And job-related problems can inform the location of suicide, says Dr. Wible, who is the author of “Physicians Suicide Letters – Answered.”
“A work-related catalyst makes it more likely that the person will attempt or complete suicide in the work setting. Physicians have stepped off hospital rooftops, shot or stabbed themselves in hospital parking lots, or [hanged] themselves in hospital chapels. Perhaps it’s because they’re choosing to die in the place where they’ve been most wounded.”
You are not at fault
“If you’re feeling suicidal, you might feel utterly alone, but if there’s one message I can give you, it’s that you’re not alone, and there are many things you can do to mitigate your pain and despair,” Dr. Wible says. “And you’re not defective. It’s the health care system that’s defective. You have nothing to be ashamed of.”
Some institutions have a “buddy system” that pairs clinicians to provide mutual peer support. A partner who notices concerning signs can refer the other partner for help. Physicians can also be paired with a “buddy,” even without a formal institutional structure.
A “buddy” is a step in the right direction, but Dr. Shapiro cautions it might be necessary to consult a trained professional for serious depression or suicidality. Several states provide connection to local resources. Employee assistance programs (EAPs) might be helpful, although many physicians don’t trust their institution’s EAP. Or physicians can ask colleagues to recommend a “doctor’s doctor” who specializes in treating physicians, suggests Dr. Yellowlees, author of “Physician Suicide: Cases and Commentaries.”
In Medscape’s 2022 report, almost all respondents who reported having suicidal colleagues said they offered help, including emotional support, practical assistance, referrals, speaking to family members, or even personally taking the colleague to the ED or to a therapist.
To enhance physicians’ ability to help each other, Dr. Lin recommends “gatekeeper training,” which has been shown to reduce suicide. “This strategy utilizes a peer-to-peer model, but, rather than a single ‘peer buddy,’ everyone is a ‘gatekeeper’ trained in approaches, such as QPR – Question, Persuade, Refer. ‘Gatekeepers’ are taught how to recognize warning signs of suicide, question the potentially suicidal individual, persuade him/her to get help, and provide referrals.”
Other ways to prevent suicide
Dr. Lin advises physicians to “create a personalized safety plan and write down signs and clues that they may be going down the wrong path and what they can do – like breathing exercises, relaxation – and identifying people to talk to, places to go, or phone numbers to call, if those initial measures aren’t enough.” The plan is private and allows the physician to determine at what point help is needed and who should be consulted. “Sometimes, when a person is in acute stress, even looking up a phone number can seem insurmountable. But having it on paper lowers the barrier, making it more achievable.”
Resources should be posted in places where physicians gather so that those who don’t already have a safety plan have easy access to that information, he suggests.
In addition, consideration may be given to reaching out for support if a colleague has died by suicide, experts suggest. Whether offered by one’s institution, a peer arrangement, spiritual counseling, or psychotherapy, one may need help dealing with the trauma, guilt, and grief that often accompany this type of loss.
A version of this article first appeared on Medscape.com.
Kip Wenger, DO, an emergency physician and systems medical director of Team Health, Knoxville, Tenn., was asked to see a patient in the emergency department. He was shocked when he realized who the patient was – a 33-year-old female physician friend and colleague.
She was bleeding from multiple self-inflicted injuries and ultimately died. “I was devastated and couldn’t wrap my head around what had just happened,” Dr. Wenger told this news organization.
It’s important for physicians to be aware of warning signs in their colleagues, such as showing up late, being irritable and short-tempered with staff, missing shifts, making mistakes, or receiving an increasing number of patient complaints, Dr. Wenger says.
Dr. Wenger had had dinner with her several weeks earlier and saw some subtle changes. He had known her as a “positive, upbeat person,” but her demeanor was different during dinner.
“There were no typical telltale signs – she was talking about her plans for the future, including buying a new bicycle – but she wasn’t herself and seemed to become tearful when I hugged her at the end of the evening,” he said. He later heard from another colleague that she had shared feeling “hopeless.”
The scope of the problem
According to the American Society for Suicide Prevention, roughly 300-400 physicians die by suicide annually. Although one study suggests a lower number, official reports likely underestimate suicides, study author Katherine Gold, MD, MSW, associate professor of family medicine, obstetrics, and gynecology, Michigan Medicine, University of Michigan, Ann Arbor, said in an interview.
Peter Yellowlees, MD, MBBS, professor of psychiatry, University of California, Davis, concurs, suggesting that some single-car accidents involving physicians might be suicides. Perry Lin, MD, assistant clinical professor, Heritage College of Osteopathic Medicine, Ohio University, Athens, and national co-chair of the Physician Suicide Awareness Committee of the American Association of Suicidology, says that some death certificates state that the deceased died of “accidental causes” because the physician who completes the certificate, possibly a colleague, is reluctant to list the actual cause of death to protect his colleague’s memory or the family’s feelings.
In general, and among physicians, White men older than 65 “represent the largest percentage of people who die from suicide nationwide,” says Dr. Lin.
But younger people are also susceptible, Dr. Lin adds. One of the most vulnerable periods for potential suicide is during the first few months of residency. This dovetails with the findings of Medscape’s 2022 report Suicide: A Tragedy of the Profession. In that report, a difference was found between frequency of suicidal thoughts in younger physicians, compared with older physicians (14% in those < 35 years vs. 8% for those ≥ 45 years).
Hurdles to preventing physician suicide
“The best thing that can happen in our profession is upstream intervention – if people seek help before they get to the point of suicidality, recognizing they’re under stress and duress and that they might be going down a bad pathway,” says Dr. Lin. But research suggests that many physicians don’t do so.
Gary Price, MD, attending surgeon and clinical assistant professor of surgery, Yale–New Haven Hospital, Connecticut, and president of the Physicians Foundation, says his organization has identified barriers that prevent physicians from seeking help.
Physicians feel they may put their licensure at risk if they admit to receiving help for mental issues. These concerns were expressed by respondents in Medscape’s above mentioned 2022 report, many of whom didn’t seek treatment for depression, burnout, or suicidal thoughts lest it affect their professional standing when renewing their license or seeking credentialing.
Although organizations and societies are advocating against these questions, a recent study found that almost 70% of U.S. states and territories continue to ask physicians about their mental health, and 28% ask for diagnoses (beyond current impairments) – a violation of the Americans With Disabilities Act.
“Mental health illness is different from mental health impairment,” Ryan Mire, MD, a Nashville, Tenn.–based internist, said in an interview. “As physicians, we’re comfortable with licensing boards asking whether the physician has any condition that might impair their care for patients, but not about a history of mental illness.”
The second barrier, says Dr. Price, is that hospital credentialing committees sometimes ask similar questions, as do commercial and malpractice insurers.
Another roadblock is that in some states, undergoing treatment for a mental health problem could be subject to discovery by a plaintiff’s attorney in a malpractice case, even if the physician’s mental health history had no effect on patient care. But that’s uncommon, says Daniel Shapiro, PhD, author of “Delivering Doctor Amelia,” a book about his treatment of a suicidal physician who underwent a malpractice lawsuit. “I’ve never seen that happen.”
A final barrier is that many employers require employees to receive treatment within their own institution or health system. “Physicians may be reluctant to get help where they work, with colleagues and friends knowing about their illness or being involved with their care,” says Dr. Price.
In 2022, the American College of Physicians (ACP) issued a toolkit to help members encourage licensing and credentialing boards to remove questions about mental health on applications and include language that supports receiving treatment, Dr. Mire says.
Layers of vulnerability
There are few data regarding relative risk among particular races or ethnicities, “but we know racism is a social stressor,” says Dr. Mire. “Obviously, people from historically disadvantaged populations tend to have societal stressors like discrimination and racism that add an extra layer of burden.”
Intersectionality – having multiple intersecting risk factors – may confer even higher risk. “For example, if you’re a female physician from a historically marginalized race and a resident dealing with the ‘hidden curriculum’ of trying to be resilient, you have multiple layers of vulnerability.”
There are also limited data regarding which specialties or work environments are associated with highest risk. “Obviously, challenges exist in every segment of medicine and at different ages, stages, and work environments, and they intersect with each individual physician’s personal risk factors,” says Dr. Mire, president of the ACP and assistant clinical professor of clinical medical education, University of Tennessee Health Science Center, Memphis.
Pamela Wible, MD, is an Oregon-based retired physician who herself went through a suicidal period about 11 years into her career that motivated her to embrace a new vision of clinical practice and change her practice model. After a series of physician suicides in her area, she began to speak and write openly about physician suicide, and since her retirement from clinical practice, she makes herself available on a full-time basis to distressed physicians. “When I address a conference of a particular medical specialty or a group in a particular geographical region, I focus on the specific vulnerabilities in that specialty or region,” she says.
What increases the chances of suicide?
“Many factors, both within and outside the professional setting, affect someone’s decision to die by suicide – after all, physicians have the same stressors as other people, like family, finances, and their own health,” Dr. Mire says. When it comes to non–work-related factors, marital stressors and comorbid psychiatric illness particularly raise the risk, says Dr. Lin.
But certain drivers are specific to the practice of medicine, with burnout and depression first in line.
Dr. Shapiro, who is vice dean for faculty and administrative affairs, Penn State University, Hershey, and the Garner James Cline Professor of Medical Humanism, conducts burnout evaluations throughout the country. “Simple depression screeners prior to the pandemic showed about a 10% major depression rate in physicians,” he told this news organization. “Now, we’re seeing a 30%-33% depression rate, even in those who weren’t frontline providers during the pandemic.”
Dr. Price agrees, noting that burnout in physicians has gone from 40% to 60% since the pandemic. But burnout doesn’t always lead to suicide. It’s when burnout progresses to depression, becomes more severe, and is untreated that the suicidal risk arises, he emphasizes.
Additionally, being a doctor isn’t “just a profession” but a “calling and identity,” says Dr. Gold. Job-related problems (for example, a malpractice suit, complaints to the medical board, loss of autonomy, changing work demands) can raise suicidal risk.
And job-related problems can inform the location of suicide, says Dr. Wible, who is the author of “Physicians Suicide Letters – Answered.”
“A work-related catalyst makes it more likely that the person will attempt or complete suicide in the work setting. Physicians have stepped off hospital rooftops, shot or stabbed themselves in hospital parking lots, or [hanged] themselves in hospital chapels. Perhaps it’s because they’re choosing to die in the place where they’ve been most wounded.”
You are not at fault
“If you’re feeling suicidal, you might feel utterly alone, but if there’s one message I can give you, it’s that you’re not alone, and there are many things you can do to mitigate your pain and despair,” Dr. Wible says. “And you’re not defective. It’s the health care system that’s defective. You have nothing to be ashamed of.”
Some institutions have a “buddy system” that pairs clinicians to provide mutual peer support. A partner who notices concerning signs can refer the other partner for help. Physicians can also be paired with a “buddy,” even without a formal institutional structure.
A “buddy” is a step in the right direction, but Dr. Shapiro cautions it might be necessary to consult a trained professional for serious depression or suicidality. Several states provide connection to local resources. Employee assistance programs (EAPs) might be helpful, although many physicians don’t trust their institution’s EAP. Or physicians can ask colleagues to recommend a “doctor’s doctor” who specializes in treating physicians, suggests Dr. Yellowlees, author of “Physician Suicide: Cases and Commentaries.”
In Medscape’s 2022 report, almost all respondents who reported having suicidal colleagues said they offered help, including emotional support, practical assistance, referrals, speaking to family members, or even personally taking the colleague to the ED or to a therapist.
To enhance physicians’ ability to help each other, Dr. Lin recommends “gatekeeper training,” which has been shown to reduce suicide. “This strategy utilizes a peer-to-peer model, but, rather than a single ‘peer buddy,’ everyone is a ‘gatekeeper’ trained in approaches, such as QPR – Question, Persuade, Refer. ‘Gatekeepers’ are taught how to recognize warning signs of suicide, question the potentially suicidal individual, persuade him/her to get help, and provide referrals.”
Other ways to prevent suicide
Dr. Lin advises physicians to “create a personalized safety plan and write down signs and clues that they may be going down the wrong path and what they can do – like breathing exercises, relaxation – and identifying people to talk to, places to go, or phone numbers to call, if those initial measures aren’t enough.” The plan is private and allows the physician to determine at what point help is needed and who should be consulted. “Sometimes, when a person is in acute stress, even looking up a phone number can seem insurmountable. But having it on paper lowers the barrier, making it more achievable.”
Resources should be posted in places where physicians gather so that those who don’t already have a safety plan have easy access to that information, he suggests.
In addition, consideration may be given to reaching out for support if a colleague has died by suicide, experts suggest. Whether offered by one’s institution, a peer arrangement, spiritual counseling, or psychotherapy, one may need help dealing with the trauma, guilt, and grief that often accompany this type of loss.
A version of this article first appeared on Medscape.com.
Kip Wenger, DO, an emergency physician and systems medical director of Team Health, Knoxville, Tenn., was asked to see a patient in the emergency department. He was shocked when he realized who the patient was – a 33-year-old female physician friend and colleague.
She was bleeding from multiple self-inflicted injuries and ultimately died. “I was devastated and couldn’t wrap my head around what had just happened,” Dr. Wenger told this news organization.
It’s important for physicians to be aware of warning signs in their colleagues, such as showing up late, being irritable and short-tempered with staff, missing shifts, making mistakes, or receiving an increasing number of patient complaints, Dr. Wenger says.
Dr. Wenger had had dinner with her several weeks earlier and saw some subtle changes. He had known her as a “positive, upbeat person,” but her demeanor was different during dinner.
“There were no typical telltale signs – she was talking about her plans for the future, including buying a new bicycle – but she wasn’t herself and seemed to become tearful when I hugged her at the end of the evening,” he said. He later heard from another colleague that she had shared feeling “hopeless.”
The scope of the problem
According to the American Society for Suicide Prevention, roughly 300-400 physicians die by suicide annually. Although one study suggests a lower number, official reports likely underestimate suicides, study author Katherine Gold, MD, MSW, associate professor of family medicine, obstetrics, and gynecology, Michigan Medicine, University of Michigan, Ann Arbor, said in an interview.
Peter Yellowlees, MD, MBBS, professor of psychiatry, University of California, Davis, concurs, suggesting that some single-car accidents involving physicians might be suicides. Perry Lin, MD, assistant clinical professor, Heritage College of Osteopathic Medicine, Ohio University, Athens, and national co-chair of the Physician Suicide Awareness Committee of the American Association of Suicidology, says that some death certificates state that the deceased died of “accidental causes” because the physician who completes the certificate, possibly a colleague, is reluctant to list the actual cause of death to protect his colleague’s memory or the family’s feelings.
In general, and among physicians, White men older than 65 “represent the largest percentage of people who die from suicide nationwide,” says Dr. Lin.
But younger people are also susceptible, Dr. Lin adds. One of the most vulnerable periods for potential suicide is during the first few months of residency. This dovetails with the findings of Medscape’s 2022 report Suicide: A Tragedy of the Profession. In that report, a difference was found between frequency of suicidal thoughts in younger physicians, compared with older physicians (14% in those < 35 years vs. 8% for those ≥ 45 years).
Hurdles to preventing physician suicide
“The best thing that can happen in our profession is upstream intervention – if people seek help before they get to the point of suicidality, recognizing they’re under stress and duress and that they might be going down a bad pathway,” says Dr. Lin. But research suggests that many physicians don’t do so.
Gary Price, MD, attending surgeon and clinical assistant professor of surgery, Yale–New Haven Hospital, Connecticut, and president of the Physicians Foundation, says his organization has identified barriers that prevent physicians from seeking help.
Physicians feel they may put their licensure at risk if they admit to receiving help for mental issues. These concerns were expressed by respondents in Medscape’s above mentioned 2022 report, many of whom didn’t seek treatment for depression, burnout, or suicidal thoughts lest it affect their professional standing when renewing their license or seeking credentialing.
Although organizations and societies are advocating against these questions, a recent study found that almost 70% of U.S. states and territories continue to ask physicians about their mental health, and 28% ask for diagnoses (beyond current impairments) – a violation of the Americans With Disabilities Act.
“Mental health illness is different from mental health impairment,” Ryan Mire, MD, a Nashville, Tenn.–based internist, said in an interview. “As physicians, we’re comfortable with licensing boards asking whether the physician has any condition that might impair their care for patients, but not about a history of mental illness.”
The second barrier, says Dr. Price, is that hospital credentialing committees sometimes ask similar questions, as do commercial and malpractice insurers.
Another roadblock is that in some states, undergoing treatment for a mental health problem could be subject to discovery by a plaintiff’s attorney in a malpractice case, even if the physician’s mental health history had no effect on patient care. But that’s uncommon, says Daniel Shapiro, PhD, author of “Delivering Doctor Amelia,” a book about his treatment of a suicidal physician who underwent a malpractice lawsuit. “I’ve never seen that happen.”
A final barrier is that many employers require employees to receive treatment within their own institution or health system. “Physicians may be reluctant to get help where they work, with colleagues and friends knowing about their illness or being involved with their care,” says Dr. Price.
In 2022, the American College of Physicians (ACP) issued a toolkit to help members encourage licensing and credentialing boards to remove questions about mental health on applications and include language that supports receiving treatment, Dr. Mire says.
Layers of vulnerability
There are few data regarding relative risk among particular races or ethnicities, “but we know racism is a social stressor,” says Dr. Mire. “Obviously, people from historically disadvantaged populations tend to have societal stressors like discrimination and racism that add an extra layer of burden.”
Intersectionality – having multiple intersecting risk factors – may confer even higher risk. “For example, if you’re a female physician from a historically marginalized race and a resident dealing with the ‘hidden curriculum’ of trying to be resilient, you have multiple layers of vulnerability.”
There are also limited data regarding which specialties or work environments are associated with highest risk. “Obviously, challenges exist in every segment of medicine and at different ages, stages, and work environments, and they intersect with each individual physician’s personal risk factors,” says Dr. Mire, president of the ACP and assistant clinical professor of clinical medical education, University of Tennessee Health Science Center, Memphis.
Pamela Wible, MD, is an Oregon-based retired physician who herself went through a suicidal period about 11 years into her career that motivated her to embrace a new vision of clinical practice and change her practice model. After a series of physician suicides in her area, she began to speak and write openly about physician suicide, and since her retirement from clinical practice, she makes herself available on a full-time basis to distressed physicians. “When I address a conference of a particular medical specialty or a group in a particular geographical region, I focus on the specific vulnerabilities in that specialty or region,” she says.
What increases the chances of suicide?
“Many factors, both within and outside the professional setting, affect someone’s decision to die by suicide – after all, physicians have the same stressors as other people, like family, finances, and their own health,” Dr. Mire says. When it comes to non–work-related factors, marital stressors and comorbid psychiatric illness particularly raise the risk, says Dr. Lin.
But certain drivers are specific to the practice of medicine, with burnout and depression first in line.
Dr. Shapiro, who is vice dean for faculty and administrative affairs, Penn State University, Hershey, and the Garner James Cline Professor of Medical Humanism, conducts burnout evaluations throughout the country. “Simple depression screeners prior to the pandemic showed about a 10% major depression rate in physicians,” he told this news organization. “Now, we’re seeing a 30%-33% depression rate, even in those who weren’t frontline providers during the pandemic.”
Dr. Price agrees, noting that burnout in physicians has gone from 40% to 60% since the pandemic. But burnout doesn’t always lead to suicide. It’s when burnout progresses to depression, becomes more severe, and is untreated that the suicidal risk arises, he emphasizes.
Additionally, being a doctor isn’t “just a profession” but a “calling and identity,” says Dr. Gold. Job-related problems (for example, a malpractice suit, complaints to the medical board, loss of autonomy, changing work demands) can raise suicidal risk.
And job-related problems can inform the location of suicide, says Dr. Wible, who is the author of “Physicians Suicide Letters – Answered.”
“A work-related catalyst makes it more likely that the person will attempt or complete suicide in the work setting. Physicians have stepped off hospital rooftops, shot or stabbed themselves in hospital parking lots, or [hanged] themselves in hospital chapels. Perhaps it’s because they’re choosing to die in the place where they’ve been most wounded.”
You are not at fault
“If you’re feeling suicidal, you might feel utterly alone, but if there’s one message I can give you, it’s that you’re not alone, and there are many things you can do to mitigate your pain and despair,” Dr. Wible says. “And you’re not defective. It’s the health care system that’s defective. You have nothing to be ashamed of.”
Some institutions have a “buddy system” that pairs clinicians to provide mutual peer support. A partner who notices concerning signs can refer the other partner for help. Physicians can also be paired with a “buddy,” even without a formal institutional structure.
A “buddy” is a step in the right direction, but Dr. Shapiro cautions it might be necessary to consult a trained professional for serious depression or suicidality. Several states provide connection to local resources. Employee assistance programs (EAPs) might be helpful, although many physicians don’t trust their institution’s EAP. Or physicians can ask colleagues to recommend a “doctor’s doctor” who specializes in treating physicians, suggests Dr. Yellowlees, author of “Physician Suicide: Cases and Commentaries.”
In Medscape’s 2022 report, almost all respondents who reported having suicidal colleagues said they offered help, including emotional support, practical assistance, referrals, speaking to family members, or even personally taking the colleague to the ED or to a therapist.
To enhance physicians’ ability to help each other, Dr. Lin recommends “gatekeeper training,” which has been shown to reduce suicide. “This strategy utilizes a peer-to-peer model, but, rather than a single ‘peer buddy,’ everyone is a ‘gatekeeper’ trained in approaches, such as QPR – Question, Persuade, Refer. ‘Gatekeepers’ are taught how to recognize warning signs of suicide, question the potentially suicidal individual, persuade him/her to get help, and provide referrals.”
Other ways to prevent suicide
Dr. Lin advises physicians to “create a personalized safety plan and write down signs and clues that they may be going down the wrong path and what they can do – like breathing exercises, relaxation – and identifying people to talk to, places to go, or phone numbers to call, if those initial measures aren’t enough.” The plan is private and allows the physician to determine at what point help is needed and who should be consulted. “Sometimes, when a person is in acute stress, even looking up a phone number can seem insurmountable. But having it on paper lowers the barrier, making it more achievable.”
Resources should be posted in places where physicians gather so that those who don’t already have a safety plan have easy access to that information, he suggests.
In addition, consideration may be given to reaching out for support if a colleague has died by suicide, experts suggest. Whether offered by one’s institution, a peer arrangement, spiritual counseling, or psychotherapy, one may need help dealing with the trauma, guilt, and grief that often accompany this type of loss.
A version of this article first appeared on Medscape.com.
Class I recall for Medtronic’s HeartWare HVAD batteries
Medtronic is recalling a single lot of HeartWare Ventricular Assist Device (HVAD) System batteries because of welding defects that may cause separation of the two cell battery packs used to power the system, according to an alert on the Food and Drug Administration website.
“The welding defect may cause the battery to malfunction and no longer provide power or prevent the battery from holding a full charge or properly recharging,” the FDA said.
The agency has identified this as a class I recall, the most serious type because of the potential for serious injury or death.
Medtronic reports one death associated with this recall and two complaints in the affected lot.
Back in April, as reported by this news organization, Medtronic alerted providers that patients implanted with the Medtronic HVAD System who develop pump thrombosis could have a welding defect in the internal pump that causes the pump to malfunction.
The batteries from the recalled lot have a model number of 1650DE, were manufactured from April 13 to 19, 2021 and distributed from April 20 to July 19, 2021. The recall affects a total of 429 devices.
On May 5, 2022, Medtronic sent an urgent medical device correction notice to customers asking them to identify and quarantine all affected batteries and notify affected patients. The notice includes a patient template to help communicate directly with patients.
It also includes a customer confirmation form to initiate an exchange. The completed form should be returned to [email protected].
Medtronic is replacing the affected batteries with new product and has implemented actions to improve control of the welding process.
The Medtronic HVAD System was approved as a bridge to heart transplantation in 2012. Since then, it’s been fraught with problems.
Earlier in June, the company announced it was stopping all sales of the device and advised physicians to stop implanting it, as reported by this news organization.
Problems related to the Medtronic HVAD System should be reported to the FDA’s MedWatch program.
A version of this article first appeared on Medscape.com.
Medtronic is recalling a single lot of HeartWare Ventricular Assist Device (HVAD) System batteries because of welding defects that may cause separation of the two cell battery packs used to power the system, according to an alert on the Food and Drug Administration website.
“The welding defect may cause the battery to malfunction and no longer provide power or prevent the battery from holding a full charge or properly recharging,” the FDA said.
The agency has identified this as a class I recall, the most serious type because of the potential for serious injury or death.
Medtronic reports one death associated with this recall and two complaints in the affected lot.
Back in April, as reported by this news organization, Medtronic alerted providers that patients implanted with the Medtronic HVAD System who develop pump thrombosis could have a welding defect in the internal pump that causes the pump to malfunction.
The batteries from the recalled lot have a model number of 1650DE, were manufactured from April 13 to 19, 2021 and distributed from April 20 to July 19, 2021. The recall affects a total of 429 devices.
On May 5, 2022, Medtronic sent an urgent medical device correction notice to customers asking them to identify and quarantine all affected batteries and notify affected patients. The notice includes a patient template to help communicate directly with patients.
It also includes a customer confirmation form to initiate an exchange. The completed form should be returned to [email protected].
Medtronic is replacing the affected batteries with new product and has implemented actions to improve control of the welding process.
The Medtronic HVAD System was approved as a bridge to heart transplantation in 2012. Since then, it’s been fraught with problems.
Earlier in June, the company announced it was stopping all sales of the device and advised physicians to stop implanting it, as reported by this news organization.
Problems related to the Medtronic HVAD System should be reported to the FDA’s MedWatch program.
A version of this article first appeared on Medscape.com.
Medtronic is recalling a single lot of HeartWare Ventricular Assist Device (HVAD) System batteries because of welding defects that may cause separation of the two cell battery packs used to power the system, according to an alert on the Food and Drug Administration website.
“The welding defect may cause the battery to malfunction and no longer provide power or prevent the battery from holding a full charge or properly recharging,” the FDA said.
The agency has identified this as a class I recall, the most serious type because of the potential for serious injury or death.
Medtronic reports one death associated with this recall and two complaints in the affected lot.
Back in April, as reported by this news organization, Medtronic alerted providers that patients implanted with the Medtronic HVAD System who develop pump thrombosis could have a welding defect in the internal pump that causes the pump to malfunction.
The batteries from the recalled lot have a model number of 1650DE, were manufactured from April 13 to 19, 2021 and distributed from April 20 to July 19, 2021. The recall affects a total of 429 devices.
On May 5, 2022, Medtronic sent an urgent medical device correction notice to customers asking them to identify and quarantine all affected batteries and notify affected patients. The notice includes a patient template to help communicate directly with patients.
It also includes a customer confirmation form to initiate an exchange. The completed form should be returned to [email protected].
Medtronic is replacing the affected batteries with new product and has implemented actions to improve control of the welding process.
The Medtronic HVAD System was approved as a bridge to heart transplantation in 2012. Since then, it’s been fraught with problems.
Earlier in June, the company announced it was stopping all sales of the device and advised physicians to stop implanting it, as reported by this news organization.
Problems related to the Medtronic HVAD System should be reported to the FDA’s MedWatch program.
A version of this article first appeared on Medscape.com.
Add AFib to noncardiac surgery risk evaluation: New support
Practice has gone back and forth on whether atrial fibrillation (AFib) should be considered in the preoperative cardiovascular risk (CV) evaluation of patients slated for noncardiac surgery, and the Revised Cardiac Risk Index (RCRI), currently widely used as an assessment tool, doesn’t include the arrhythmia.
But consideration of preexisting AFib along with the RCRI predicted 30-day mortality more sharply than the RCRI alone in an analysis of data covering several million patients slated for such procedures.
Indeed, AFib emerged as a significant, independent risk factor for a number of bad postoperative outcomes. Mortality within a month of the procedure climbed about 30% for patients with AFib before the noncardiac surgery. Their 30-day risks for stroke and for heart failure hospitalization went up similarly.
The addition of AFib to the RCRI significantly improved its ability to discriminate 30-day postoperative risk levels regardless of age, sex, and type of noncardiac surgery, Amgad Mentias, MD, Cleveland Clinic, told this news organization. And “it was able to correctly up-classify patients to high risk, if AFib was there, and it was able to down-classify some patients to lower risk if it wasn’t there.”
“I think [the findings] are convincing evidence that atrial fib should at least be part of the thought process for the surgical team and the medical team taking care of the patient,” said Dr. Mentias, who is senior author on the study published in the Journal of the American College of Cardiology, with lead author Sameer Prasada, MD, also of the Cleveland Clinic.
The results “call for incorporating AFib as a risk factor in perioperative risk scores for cardiovascular morbidity and mortality,” the published report states.
Supraventricular arrhythmias had been part of the Goldman Risk Index once widely used preoperatively to assess cardiac risk before practice adopted the RCRI in the past decade, observe Anne B. Curtis, MD, and Sai Krishna C. Korada, MD, University at Buffalo, New York, in an accompanying editorial.
The current findings “demonstrate improved prediction of adverse postsurgical outcomes” from supplementing the RCRI with AFib, they write. Given associations between preexisting AFib and serious cardiac events, “it is time to ‘re-revise’ the RCRI and acknowledge the importance of AFib in predicting adverse outcomes” after noncardiac surgery.
The new findings, however, aren’t all straightforward. In one result that remains a bit of a head-scratcher, postoperative risk of myocardial infarction (MI) in patients with preexisting AFib went in the opposite direction of risk for death and other CV outcomes, falling by almost 20%.
That is “hard to explain with the available data,” the report states, but “the use of anticoagulation, whether oral or parenteral (as a bridge therapy in the perioperative period), is a plausible explanation” given the frequent role of thrombosis in triggering MIs.
Consistent with such a mechanism, the group argues, the MI risk reduction was seen primarily among patients with AFib and a CHA2DS2-VASc score of 2 or higher – that is, those at highest risk for stroke and therefore most likely to be on oral anticoagulation. The MI risk reduction wasn’t seen in such patients with a CHA2DS2-VASc score of 0 or 1.
“I think that’s part of the explanation, that anticoagulation can reduce risk of MI. But it’s not the whole explanation,” Dr. Mentias said in an interview. If it were the sole mechanism, he said, then the same oral anticoagulation that protected against MI should have also cut the postoperative stroke risk. Yet that risk climbed 40% among patients with preexisting AFib.
The analysis started with 8.6 million Medicare patients with planned noncardiac surgery, seen from 2015 to 2019, of whom 16.4% had preexisting AFib. Propensity matching for demographics, urgency and type of surgery, CHA2DS2-VASc score, and RCRI index created two cohorts for comparison: 1.13 million patients with and 1.92 million without preexisting AFib.
Preexisting AFib was associated with a higher 30-day risk for death from any cause, the primary endpoint being 8.3% versus 5.8% for those without such AFib (P < .001), for an odds ratio of 1.31 (95% confidence interval, 1.30-1.32).
Corresponding 30-day ORs for other events, all significant at P < .001, were:
- 1.31 (95% CI, 1.30-1.33) for heart failure
- 1.40 (95% CI, 1.37-1.43) for stroke
- 1.59 (95% CI, 1.43-1.75) for systemic embolism
- 1.14 (95% CI, 1.13-1.16) for major bleeding
- 0.81 (95% CI, 0.79-0.82) for MI
Those with preexisting AFib also had longer hospitalizations at a median 5 days, compared with 4 days for those without such AFib (P < .001).
The study has the limitations of most any retrospective cohort analysis. Other limitations, the report notes, include lack of information on any antiarrhythmic meds given during hospitalization or type of AFib.
For example, AFib that is permanent – compared with paroxysmal or persistent – may be associated with more atrial fibrosis, greater atrial dilatation, “and probably higher pressures inside the heart,” Dr. Mentias observed.
“That’s not always the case, but that’s the notion. So presumably people with persistent or permanent atrial fib would have more advanced heart disease, and that could imply more risk. But we did not have that kind of data.”
Dr. Mentias and Dr. Prasada report no relevant financial relationships; disclosures for the other authors are in the report. Dr. Curtis discloses serving on advisory boards for Abbott, Janssen Pharmaceuticals, Sanofi, and Milestone Pharmaceuticals; receiving honoraria for speaking from Medtronic and Zoll; and serving on a data-monitoring board for Medtronic. Dr. Korada reports he has no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Practice has gone back and forth on whether atrial fibrillation (AFib) should be considered in the preoperative cardiovascular risk (CV) evaluation of patients slated for noncardiac surgery, and the Revised Cardiac Risk Index (RCRI), currently widely used as an assessment tool, doesn’t include the arrhythmia.
But consideration of preexisting AFib along with the RCRI predicted 30-day mortality more sharply than the RCRI alone in an analysis of data covering several million patients slated for such procedures.
Indeed, AFib emerged as a significant, independent risk factor for a number of bad postoperative outcomes. Mortality within a month of the procedure climbed about 30% for patients with AFib before the noncardiac surgery. Their 30-day risks for stroke and for heart failure hospitalization went up similarly.
The addition of AFib to the RCRI significantly improved its ability to discriminate 30-day postoperative risk levels regardless of age, sex, and type of noncardiac surgery, Amgad Mentias, MD, Cleveland Clinic, told this news organization. And “it was able to correctly up-classify patients to high risk, if AFib was there, and it was able to down-classify some patients to lower risk if it wasn’t there.”
“I think [the findings] are convincing evidence that atrial fib should at least be part of the thought process for the surgical team and the medical team taking care of the patient,” said Dr. Mentias, who is senior author on the study published in the Journal of the American College of Cardiology, with lead author Sameer Prasada, MD, also of the Cleveland Clinic.
The results “call for incorporating AFib as a risk factor in perioperative risk scores for cardiovascular morbidity and mortality,” the published report states.
Supraventricular arrhythmias had been part of the Goldman Risk Index once widely used preoperatively to assess cardiac risk before practice adopted the RCRI in the past decade, observe Anne B. Curtis, MD, and Sai Krishna C. Korada, MD, University at Buffalo, New York, in an accompanying editorial.
The current findings “demonstrate improved prediction of adverse postsurgical outcomes” from supplementing the RCRI with AFib, they write. Given associations between preexisting AFib and serious cardiac events, “it is time to ‘re-revise’ the RCRI and acknowledge the importance of AFib in predicting adverse outcomes” after noncardiac surgery.
The new findings, however, aren’t all straightforward. In one result that remains a bit of a head-scratcher, postoperative risk of myocardial infarction (MI) in patients with preexisting AFib went in the opposite direction of risk for death and other CV outcomes, falling by almost 20%.
That is “hard to explain with the available data,” the report states, but “the use of anticoagulation, whether oral or parenteral (as a bridge therapy in the perioperative period), is a plausible explanation” given the frequent role of thrombosis in triggering MIs.
Consistent with such a mechanism, the group argues, the MI risk reduction was seen primarily among patients with AFib and a CHA2DS2-VASc score of 2 or higher – that is, those at highest risk for stroke and therefore most likely to be on oral anticoagulation. The MI risk reduction wasn’t seen in such patients with a CHA2DS2-VASc score of 0 or 1.
“I think that’s part of the explanation, that anticoagulation can reduce risk of MI. But it’s not the whole explanation,” Dr. Mentias said in an interview. If it were the sole mechanism, he said, then the same oral anticoagulation that protected against MI should have also cut the postoperative stroke risk. Yet that risk climbed 40% among patients with preexisting AFib.
The analysis started with 8.6 million Medicare patients with planned noncardiac surgery, seen from 2015 to 2019, of whom 16.4% had preexisting AFib. Propensity matching for demographics, urgency and type of surgery, CHA2DS2-VASc score, and RCRI index created two cohorts for comparison: 1.13 million patients with and 1.92 million without preexisting AFib.
Preexisting AFib was associated with a higher 30-day risk for death from any cause, the primary endpoint being 8.3% versus 5.8% for those without such AFib (P < .001), for an odds ratio of 1.31 (95% confidence interval, 1.30-1.32).
Corresponding 30-day ORs for other events, all significant at P < .001, were:
- 1.31 (95% CI, 1.30-1.33) for heart failure
- 1.40 (95% CI, 1.37-1.43) for stroke
- 1.59 (95% CI, 1.43-1.75) for systemic embolism
- 1.14 (95% CI, 1.13-1.16) for major bleeding
- 0.81 (95% CI, 0.79-0.82) for MI
Those with preexisting AFib also had longer hospitalizations at a median 5 days, compared with 4 days for those without such AFib (P < .001).
The study has the limitations of most any retrospective cohort analysis. Other limitations, the report notes, include lack of information on any antiarrhythmic meds given during hospitalization or type of AFib.
For example, AFib that is permanent – compared with paroxysmal or persistent – may be associated with more atrial fibrosis, greater atrial dilatation, “and probably higher pressures inside the heart,” Dr. Mentias observed.
“That’s not always the case, but that’s the notion. So presumably people with persistent or permanent atrial fib would have more advanced heart disease, and that could imply more risk. But we did not have that kind of data.”
Dr. Mentias and Dr. Prasada report no relevant financial relationships; disclosures for the other authors are in the report. Dr. Curtis discloses serving on advisory boards for Abbott, Janssen Pharmaceuticals, Sanofi, and Milestone Pharmaceuticals; receiving honoraria for speaking from Medtronic and Zoll; and serving on a data-monitoring board for Medtronic. Dr. Korada reports he has no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Practice has gone back and forth on whether atrial fibrillation (AFib) should be considered in the preoperative cardiovascular risk (CV) evaluation of patients slated for noncardiac surgery, and the Revised Cardiac Risk Index (RCRI), currently widely used as an assessment tool, doesn’t include the arrhythmia.
But consideration of preexisting AFib along with the RCRI predicted 30-day mortality more sharply than the RCRI alone in an analysis of data covering several million patients slated for such procedures.
Indeed, AFib emerged as a significant, independent risk factor for a number of bad postoperative outcomes. Mortality within a month of the procedure climbed about 30% for patients with AFib before the noncardiac surgery. Their 30-day risks for stroke and for heart failure hospitalization went up similarly.
The addition of AFib to the RCRI significantly improved its ability to discriminate 30-day postoperative risk levels regardless of age, sex, and type of noncardiac surgery, Amgad Mentias, MD, Cleveland Clinic, told this news organization. And “it was able to correctly up-classify patients to high risk, if AFib was there, and it was able to down-classify some patients to lower risk if it wasn’t there.”
“I think [the findings] are convincing evidence that atrial fib should at least be part of the thought process for the surgical team and the medical team taking care of the patient,” said Dr. Mentias, who is senior author on the study published in the Journal of the American College of Cardiology, with lead author Sameer Prasada, MD, also of the Cleveland Clinic.
The results “call for incorporating AFib as a risk factor in perioperative risk scores for cardiovascular morbidity and mortality,” the published report states.
Supraventricular arrhythmias had been part of the Goldman Risk Index once widely used preoperatively to assess cardiac risk before practice adopted the RCRI in the past decade, observe Anne B. Curtis, MD, and Sai Krishna C. Korada, MD, University at Buffalo, New York, in an accompanying editorial.
The current findings “demonstrate improved prediction of adverse postsurgical outcomes” from supplementing the RCRI with AFib, they write. Given associations between preexisting AFib and serious cardiac events, “it is time to ‘re-revise’ the RCRI and acknowledge the importance of AFib in predicting adverse outcomes” after noncardiac surgery.
The new findings, however, aren’t all straightforward. In one result that remains a bit of a head-scratcher, postoperative risk of myocardial infarction (MI) in patients with preexisting AFib went in the opposite direction of risk for death and other CV outcomes, falling by almost 20%.
That is “hard to explain with the available data,” the report states, but “the use of anticoagulation, whether oral or parenteral (as a bridge therapy in the perioperative period), is a plausible explanation” given the frequent role of thrombosis in triggering MIs.
Consistent with such a mechanism, the group argues, the MI risk reduction was seen primarily among patients with AFib and a CHA2DS2-VASc score of 2 or higher – that is, those at highest risk for stroke and therefore most likely to be on oral anticoagulation. The MI risk reduction wasn’t seen in such patients with a CHA2DS2-VASc score of 0 or 1.
“I think that’s part of the explanation, that anticoagulation can reduce risk of MI. But it’s not the whole explanation,” Dr. Mentias said in an interview. If it were the sole mechanism, he said, then the same oral anticoagulation that protected against MI should have also cut the postoperative stroke risk. Yet that risk climbed 40% among patients with preexisting AFib.
The analysis started with 8.6 million Medicare patients with planned noncardiac surgery, seen from 2015 to 2019, of whom 16.4% had preexisting AFib. Propensity matching for demographics, urgency and type of surgery, CHA2DS2-VASc score, and RCRI index created two cohorts for comparison: 1.13 million patients with and 1.92 million without preexisting AFib.
Preexisting AFib was associated with a higher 30-day risk for death from any cause, the primary endpoint being 8.3% versus 5.8% for those without such AFib (P < .001), for an odds ratio of 1.31 (95% confidence interval, 1.30-1.32).
Corresponding 30-day ORs for other events, all significant at P < .001, were:
- 1.31 (95% CI, 1.30-1.33) for heart failure
- 1.40 (95% CI, 1.37-1.43) for stroke
- 1.59 (95% CI, 1.43-1.75) for systemic embolism
- 1.14 (95% CI, 1.13-1.16) for major bleeding
- 0.81 (95% CI, 0.79-0.82) for MI
Those with preexisting AFib also had longer hospitalizations at a median 5 days, compared with 4 days for those without such AFib (P < .001).
The study has the limitations of most any retrospective cohort analysis. Other limitations, the report notes, include lack of information on any antiarrhythmic meds given during hospitalization or type of AFib.
For example, AFib that is permanent – compared with paroxysmal or persistent – may be associated with more atrial fibrosis, greater atrial dilatation, “and probably higher pressures inside the heart,” Dr. Mentias observed.
“That’s not always the case, but that’s the notion. So presumably people with persistent or permanent atrial fib would have more advanced heart disease, and that could imply more risk. But we did not have that kind of data.”
Dr. Mentias and Dr. Prasada report no relevant financial relationships; disclosures for the other authors are in the report. Dr. Curtis discloses serving on advisory boards for Abbott, Janssen Pharmaceuticals, Sanofi, and Milestone Pharmaceuticals; receiving honoraria for speaking from Medtronic and Zoll; and serving on a data-monitoring board for Medtronic. Dr. Korada reports he has no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Roe v. Wade overturned, ending 50 years of abortion protections
According to some estimates, about 25 million women of reproductive age will now live in states that ban or severely restrict abortion. Twenty-six states are “certain or likely” to ban abortion, according to the Guttmacher Institute, which supports abortion rights.
Thirteen states have so-called trigger laws that will ban abortion almost immediately, while nine other states are now likely to try to enforce near-total bans or severe restrictions that have been blocked by courts pending the outcome of the just-issued decision in Dobbs v. Jackson Women’s Health Organization. Four states also have a history or have shown a recent desire to prohibit abortion, according to the Guttmacher Institute.
Doctors and others who provide abortion services, or in some states “aid or abet” an abortion, could be fined thousands of dollars or sent to prison.
The court voted in favor of Mississippi and its 2018 law that outlawed abortion after 15 weeks. Jackson Women’s Health, the state’s sole remaining abortion provider, sued to block the law soon after it passed.
The Supreme Court decision is not a surprise, as the justices indicated they were leaning that way during oral arguments in December. The majority’s thoughts were further revealed when a draft of the opinion was leaked to the news outlet Politico on May 2.
In the final opinion, Justice Samuel Alito, writing for the majority, “It is time to heed the Constitution and return the issue of abortion to the people’s elected representatives.”
The decision strikes down both precedent-setting rulings that established a right to abortion until the point of viability, long considered to be 24 weeks: Roe v. Wade (1973) and Planned Parenthood v. Casey (1992).
Twenty-five medical professional societies – representing OB/GYNs, family medicine doctors, fertility specialists, geneticists, hospitalists, internists, pediatricians, psychiatrists, nurses, nurse practitioners, and midwives – had urged the court to throw out the Mississippi law. And more than 2,500 medical professionals signed on to a petition in June, urging the court to uphold the right to abortion.
The number of abortions has recently increased from what had been a long decline. The Guttmacher Institute estimates there were there were 930,160 abortion procedures in 2020 (compared to 3.6 million births), an 8% increase from 2017. The number does not include self-managed abortions. The organization said the increase was potentially due to expanded Medicaid coverage and reduced access to contraception due to Trump administration policies.
Trigger laws and bans
When trigger laws and new restrictions go into effect, women in the South, Midwest, and Inter-Mountain West will likely have to drive hundreds of miles for an abortion, according to Guttmacher. Women in Louisiana, for instance, would have to drive 660 miles to get to the nearest provider in Illinois.
University of Utah researchers estimated that almost half of women will see a big increase in the distance to abortion care, from a median distance of 39 miles to 113 miles. State bans will disproportionately impact women of color, those living in poverty, and people with less education, they said.
The CDC has reported that Black women are three times more likely to die from a pregnancy-related cause than white women.
Doctors and other abortion providers could face serious penalties. The maximum penalty in Texas is life in prison, and the sentence could be 10 to 15 years in 11 other states, according to an article in the medical journal JAMA by attorneys Rebecca B. Reingold and Lawrence O. Gostin.
“Threats of prosecution undermine clinicians’ ability to provide safe, evidence-based care and to counsel patients honestly, impeding the patient-physician relationship,” they wrote. “Given harsh penalties, physicians may cease treating pregnancy loss, with no clear line between treating miscarriages and abortions.”
In preparing for these attacks on patients and doctors, New York Gov. Kathy Hochul on June 13 signed a bill that immediately protects anyone who has an abortion and medical professionals in the state who provide them from legal retaliation by states that restrict or prohibit abortion.
Even while Roe was still the law, Mississippi had banned most abortions after 20 weeks, and 16 states prohibited abortion after 22 weeks. A Texas ban on abortion after 6 weeks – which also allows private citizens to sue abortion providers – was allowed to stay in place while it was being challenged.
On May 26, Oklahoma Gov. Kevin Stitt signed a bill banning abortion from the moment of conception. Just as in Texas, the Oklahoma law allows what critics have called “bounty hunting” of abortion providers.
Four states have a constitutional amendment declaring that the state constitution does not secure or protect the right to abortion or allow the use of public funds for abortion: Alabama, Louisiana, Tennessee, and West Virginia.
Some states protecting rights
At least 16 states have proactively protected a right to an abortion, according to Guttmacher, while The New York Times reports that Washington, DC, has laws that protect abortion, along with 20 states: Alaska, Colorado, Illinois, Maine, Massachusetts, Minnesota, Nevada, New Hampshire, New Mexico, Rhode Island, California, Connecticut, Delaware, Hawaii, Maryland, New Jersey, New York, Oregon, Vermont, and Washington.
Some of these states are gearing up for a potential influx of patients. Washington Gov. Jay Inslee signed a law that authorizes physician assistants, advanced registered nurse practitioners, and other providers acting within their scope of practice to perform abortions. And the Maryland Legislature overrode a veto by Gov. Larry Hogan of a law that expands who can perform abortions.
Wisconsin Gov. Tony Evers in early June called a special legislative session to repeal the state’s 173-year-old dormant ban on abortion. But the majority Republican legislature vowed to take no action.
B. Jessie Hill, JD, associate dean for academic affairs and a professor at the Case Western Reserve University School of Law, says she expects anti-abortion groups to challenge these protective laws, “by saying that fetuses are persons under the Constitution with a right to life and therefore that the state has to protect them.”
But, she says, “there’s going to be big, big challenges with those lawsuits,” and they will not be “winners off the bat.”
Medication abortions, travel next battle
Some states are also trying to outlaw or severely restrict the use of RU-486, the abortion pill. A Tennessee law that goes into effect in 2023 would ban delivery of pills by mail and require a patient to have two doctor visits – one consultation and one to pick up the pills.
Mississippi has also enacted restrictions including the requirement that women meet with a doctor first – and is being sued by pill maker GenBioPro.
Guttmacher estimates that medication abortion accounted for 39% of all abortions in the U.S. in 2017 and 60% of all abortions that occurred before 10 weeks’ gestation.
Some states have floated the idea of prohibiting anyone from traveling to another state for an abortion.
George Mason University law professor Ilya Somin, JD, has written that such a law would likely violate the Dormant Commerce Clause, “which forbids state regulations that specifically restrict interstate commerce or discriminate against it.”
He also wrote that states lack the authority to regulate activity that takes place beyond their borders and that such bans “are open to challenge because they violate the constitutional right to travel.”
Hill also said a travel ban would be problematic, noting that it might be difficult to prosecute someone for “something you did completely in another state.”
A version of this article first appeared on Medscape.com.
According to some estimates, about 25 million women of reproductive age will now live in states that ban or severely restrict abortion. Twenty-six states are “certain or likely” to ban abortion, according to the Guttmacher Institute, which supports abortion rights.
Thirteen states have so-called trigger laws that will ban abortion almost immediately, while nine other states are now likely to try to enforce near-total bans or severe restrictions that have been blocked by courts pending the outcome of the just-issued decision in Dobbs v. Jackson Women’s Health Organization. Four states also have a history or have shown a recent desire to prohibit abortion, according to the Guttmacher Institute.
Doctors and others who provide abortion services, or in some states “aid or abet” an abortion, could be fined thousands of dollars or sent to prison.
The court voted in favor of Mississippi and its 2018 law that outlawed abortion after 15 weeks. Jackson Women’s Health, the state’s sole remaining abortion provider, sued to block the law soon after it passed.
The Supreme Court decision is not a surprise, as the justices indicated they were leaning that way during oral arguments in December. The majority’s thoughts were further revealed when a draft of the opinion was leaked to the news outlet Politico on May 2.
In the final opinion, Justice Samuel Alito, writing for the majority, “It is time to heed the Constitution and return the issue of abortion to the people’s elected representatives.”
The decision strikes down both precedent-setting rulings that established a right to abortion until the point of viability, long considered to be 24 weeks: Roe v. Wade (1973) and Planned Parenthood v. Casey (1992).
Twenty-five medical professional societies – representing OB/GYNs, family medicine doctors, fertility specialists, geneticists, hospitalists, internists, pediatricians, psychiatrists, nurses, nurse practitioners, and midwives – had urged the court to throw out the Mississippi law. And more than 2,500 medical professionals signed on to a petition in June, urging the court to uphold the right to abortion.
The number of abortions has recently increased from what had been a long decline. The Guttmacher Institute estimates there were there were 930,160 abortion procedures in 2020 (compared to 3.6 million births), an 8% increase from 2017. The number does not include self-managed abortions. The organization said the increase was potentially due to expanded Medicaid coverage and reduced access to contraception due to Trump administration policies.
Trigger laws and bans
When trigger laws and new restrictions go into effect, women in the South, Midwest, and Inter-Mountain West will likely have to drive hundreds of miles for an abortion, according to Guttmacher. Women in Louisiana, for instance, would have to drive 660 miles to get to the nearest provider in Illinois.
University of Utah researchers estimated that almost half of women will see a big increase in the distance to abortion care, from a median distance of 39 miles to 113 miles. State bans will disproportionately impact women of color, those living in poverty, and people with less education, they said.
The CDC has reported that Black women are three times more likely to die from a pregnancy-related cause than white women.
Doctors and other abortion providers could face serious penalties. The maximum penalty in Texas is life in prison, and the sentence could be 10 to 15 years in 11 other states, according to an article in the medical journal JAMA by attorneys Rebecca B. Reingold and Lawrence O. Gostin.
“Threats of prosecution undermine clinicians’ ability to provide safe, evidence-based care and to counsel patients honestly, impeding the patient-physician relationship,” they wrote. “Given harsh penalties, physicians may cease treating pregnancy loss, with no clear line between treating miscarriages and abortions.”
In preparing for these attacks on patients and doctors, New York Gov. Kathy Hochul on June 13 signed a bill that immediately protects anyone who has an abortion and medical professionals in the state who provide them from legal retaliation by states that restrict or prohibit abortion.
Even while Roe was still the law, Mississippi had banned most abortions after 20 weeks, and 16 states prohibited abortion after 22 weeks. A Texas ban on abortion after 6 weeks – which also allows private citizens to sue abortion providers – was allowed to stay in place while it was being challenged.
On May 26, Oklahoma Gov. Kevin Stitt signed a bill banning abortion from the moment of conception. Just as in Texas, the Oklahoma law allows what critics have called “bounty hunting” of abortion providers.
Four states have a constitutional amendment declaring that the state constitution does not secure or protect the right to abortion or allow the use of public funds for abortion: Alabama, Louisiana, Tennessee, and West Virginia.
Some states protecting rights
At least 16 states have proactively protected a right to an abortion, according to Guttmacher, while The New York Times reports that Washington, DC, has laws that protect abortion, along with 20 states: Alaska, Colorado, Illinois, Maine, Massachusetts, Minnesota, Nevada, New Hampshire, New Mexico, Rhode Island, California, Connecticut, Delaware, Hawaii, Maryland, New Jersey, New York, Oregon, Vermont, and Washington.
Some of these states are gearing up for a potential influx of patients. Washington Gov. Jay Inslee signed a law that authorizes physician assistants, advanced registered nurse practitioners, and other providers acting within their scope of practice to perform abortions. And the Maryland Legislature overrode a veto by Gov. Larry Hogan of a law that expands who can perform abortions.
Wisconsin Gov. Tony Evers in early June called a special legislative session to repeal the state’s 173-year-old dormant ban on abortion. But the majority Republican legislature vowed to take no action.
B. Jessie Hill, JD, associate dean for academic affairs and a professor at the Case Western Reserve University School of Law, says she expects anti-abortion groups to challenge these protective laws, “by saying that fetuses are persons under the Constitution with a right to life and therefore that the state has to protect them.”
But, she says, “there’s going to be big, big challenges with those lawsuits,” and they will not be “winners off the bat.”
Medication abortions, travel next battle
Some states are also trying to outlaw or severely restrict the use of RU-486, the abortion pill. A Tennessee law that goes into effect in 2023 would ban delivery of pills by mail and require a patient to have two doctor visits – one consultation and one to pick up the pills.
Mississippi has also enacted restrictions including the requirement that women meet with a doctor first – and is being sued by pill maker GenBioPro.
Guttmacher estimates that medication abortion accounted for 39% of all abortions in the U.S. in 2017 and 60% of all abortions that occurred before 10 weeks’ gestation.
Some states have floated the idea of prohibiting anyone from traveling to another state for an abortion.
George Mason University law professor Ilya Somin, JD, has written that such a law would likely violate the Dormant Commerce Clause, “which forbids state regulations that specifically restrict interstate commerce or discriminate against it.”
He also wrote that states lack the authority to regulate activity that takes place beyond their borders and that such bans “are open to challenge because they violate the constitutional right to travel.”
Hill also said a travel ban would be problematic, noting that it might be difficult to prosecute someone for “something you did completely in another state.”
A version of this article first appeared on Medscape.com.
According to some estimates, about 25 million women of reproductive age will now live in states that ban or severely restrict abortion. Twenty-six states are “certain or likely” to ban abortion, according to the Guttmacher Institute, which supports abortion rights.
Thirteen states have so-called trigger laws that will ban abortion almost immediately, while nine other states are now likely to try to enforce near-total bans or severe restrictions that have been blocked by courts pending the outcome of the just-issued decision in Dobbs v. Jackson Women’s Health Organization. Four states also have a history or have shown a recent desire to prohibit abortion, according to the Guttmacher Institute.
Doctors and others who provide abortion services, or in some states “aid or abet” an abortion, could be fined thousands of dollars or sent to prison.
The court voted in favor of Mississippi and its 2018 law that outlawed abortion after 15 weeks. Jackson Women’s Health, the state’s sole remaining abortion provider, sued to block the law soon after it passed.
The Supreme Court decision is not a surprise, as the justices indicated they were leaning that way during oral arguments in December. The majority’s thoughts were further revealed when a draft of the opinion was leaked to the news outlet Politico on May 2.
In the final opinion, Justice Samuel Alito, writing for the majority, “It is time to heed the Constitution and return the issue of abortion to the people’s elected representatives.”
The decision strikes down both precedent-setting rulings that established a right to abortion until the point of viability, long considered to be 24 weeks: Roe v. Wade (1973) and Planned Parenthood v. Casey (1992).
Twenty-five medical professional societies – representing OB/GYNs, family medicine doctors, fertility specialists, geneticists, hospitalists, internists, pediatricians, psychiatrists, nurses, nurse practitioners, and midwives – had urged the court to throw out the Mississippi law. And more than 2,500 medical professionals signed on to a petition in June, urging the court to uphold the right to abortion.
The number of abortions has recently increased from what had been a long decline. The Guttmacher Institute estimates there were there were 930,160 abortion procedures in 2020 (compared to 3.6 million births), an 8% increase from 2017. The number does not include self-managed abortions. The organization said the increase was potentially due to expanded Medicaid coverage and reduced access to contraception due to Trump administration policies.
Trigger laws and bans
When trigger laws and new restrictions go into effect, women in the South, Midwest, and Inter-Mountain West will likely have to drive hundreds of miles for an abortion, according to Guttmacher. Women in Louisiana, for instance, would have to drive 660 miles to get to the nearest provider in Illinois.
University of Utah researchers estimated that almost half of women will see a big increase in the distance to abortion care, from a median distance of 39 miles to 113 miles. State bans will disproportionately impact women of color, those living in poverty, and people with less education, they said.
The CDC has reported that Black women are three times more likely to die from a pregnancy-related cause than white women.
Doctors and other abortion providers could face serious penalties. The maximum penalty in Texas is life in prison, and the sentence could be 10 to 15 years in 11 other states, according to an article in the medical journal JAMA by attorneys Rebecca B. Reingold and Lawrence O. Gostin.
“Threats of prosecution undermine clinicians’ ability to provide safe, evidence-based care and to counsel patients honestly, impeding the patient-physician relationship,” they wrote. “Given harsh penalties, physicians may cease treating pregnancy loss, with no clear line between treating miscarriages and abortions.”
In preparing for these attacks on patients and doctors, New York Gov. Kathy Hochul on June 13 signed a bill that immediately protects anyone who has an abortion and medical professionals in the state who provide them from legal retaliation by states that restrict or prohibit abortion.
Even while Roe was still the law, Mississippi had banned most abortions after 20 weeks, and 16 states prohibited abortion after 22 weeks. A Texas ban on abortion after 6 weeks – which also allows private citizens to sue abortion providers – was allowed to stay in place while it was being challenged.
On May 26, Oklahoma Gov. Kevin Stitt signed a bill banning abortion from the moment of conception. Just as in Texas, the Oklahoma law allows what critics have called “bounty hunting” of abortion providers.
Four states have a constitutional amendment declaring that the state constitution does not secure or protect the right to abortion or allow the use of public funds for abortion: Alabama, Louisiana, Tennessee, and West Virginia.
Some states protecting rights
At least 16 states have proactively protected a right to an abortion, according to Guttmacher, while The New York Times reports that Washington, DC, has laws that protect abortion, along with 20 states: Alaska, Colorado, Illinois, Maine, Massachusetts, Minnesota, Nevada, New Hampshire, New Mexico, Rhode Island, California, Connecticut, Delaware, Hawaii, Maryland, New Jersey, New York, Oregon, Vermont, and Washington.
Some of these states are gearing up for a potential influx of patients. Washington Gov. Jay Inslee signed a law that authorizes physician assistants, advanced registered nurse practitioners, and other providers acting within their scope of practice to perform abortions. And the Maryland Legislature overrode a veto by Gov. Larry Hogan of a law that expands who can perform abortions.
Wisconsin Gov. Tony Evers in early June called a special legislative session to repeal the state’s 173-year-old dormant ban on abortion. But the majority Republican legislature vowed to take no action.
B. Jessie Hill, JD, associate dean for academic affairs and a professor at the Case Western Reserve University School of Law, says she expects anti-abortion groups to challenge these protective laws, “by saying that fetuses are persons under the Constitution with a right to life and therefore that the state has to protect them.”
But, she says, “there’s going to be big, big challenges with those lawsuits,” and they will not be “winners off the bat.”
Medication abortions, travel next battle
Some states are also trying to outlaw or severely restrict the use of RU-486, the abortion pill. A Tennessee law that goes into effect in 2023 would ban delivery of pills by mail and require a patient to have two doctor visits – one consultation and one to pick up the pills.
Mississippi has also enacted restrictions including the requirement that women meet with a doctor first – and is being sued by pill maker GenBioPro.
Guttmacher estimates that medication abortion accounted for 39% of all abortions in the U.S. in 2017 and 60% of all abortions that occurred before 10 weeks’ gestation.
Some states have floated the idea of prohibiting anyone from traveling to another state for an abortion.
George Mason University law professor Ilya Somin, JD, has written that such a law would likely violate the Dormant Commerce Clause, “which forbids state regulations that specifically restrict interstate commerce or discriminate against it.”
He also wrote that states lack the authority to regulate activity that takes place beyond their borders and that such bans “are open to challenge because they violate the constitutional right to travel.”
Hill also said a travel ban would be problematic, noting that it might be difficult to prosecute someone for “something you did completely in another state.”
A version of this article first appeared on Medscape.com.
Deaths rare in tonsillectomy, but some children at more risk
It’s rare for a child to die after a tonsillectomy, but children who die are more likely to have a complex chronic condition such as cerebral palsy or Down syndrome, according to a retrospective cohort study published in JAMA.
“Among children undergoing tonsillectomy, the rate of postoperative death was 7 per 100,000 operations overall, [but] among children with complex chronic conditions, the rate of postoperative death was 117 per 100,000 operations, representing 44% of overall deaths,” write researchers at the University of Wisconsin–Madison. “These findings may inform decisionmaking for pediatric tonsillectomy.”
The rate of death in children after tonsillectomy has been uncertain, the authors write. Specific mortality rates for children at increased risk for complications, including those under 3 years old and those with sleep-disordered breathing or complex chronic conditions, have not been available.
To learn how likely children undergoing tonsillectomy are to die after their surgery, as well as which children are most at risk, lead study author M. Bruce Edmonson, MD, MPH, department of pediatrics, University of Wisconsin School of Medicine and Public Health, Madison, and his colleagues drew data from five states, including ambulatory surgery, inpatient, and emergency department discharge data sets provided by the Healthcare Cost and Utilization Project, Agency for Healthcare Research and Quality for California, Florida, Maryland, New York, and Wisconsin.
Participants included 504,262 patients under 21 years of age whose discharge records linked their inpatient or outpatient tonsillectomy, with or without adenoidectomy, with at least 90 days of follow-up.
In a longitudinal analysis, the research team investigated postoperative death within 30 days or during a surgical stay lasting over 30 days. They calculated postoperative mortality per 100,000 operations, both overall and classified by age group, sleep-disordered breathing, and complex chronic conditions.
The 504,262 children ranged in age from 0 to 20 years and underwent a total of 505,182 tonsillectomies. Of these, 10.1% were performed in children aged under 3 years, 28.9% in children with sleep-disordered breathing, and 2.8% in those with complex chronic conditions.
The 36 linked postoperative deaths occurred between 2 and 20.5 days after surgical admission, and 19 (53%) of the deaths occurred after surgical discharge.
The unadjusted mortality rate was 7.04 (95% confidence interval, 4.97-9.98) deaths per 100,000 procedures. In multivariable models, children younger than 3 years and children with sleep-disordered breathing were not significantly more likely to die.
But children with complex chronic conditions were significantly more likely to die than were children without those conditions (117.22 vs. 3.87 deaths per 100,000 procedures, respectively).
Children with complex chronic conditions underwent only 2.8% of all tonsillectomies, but they accounted for 44% of postoperative deaths. Most deaths linked with complex chronic conditions occurred among children with neurologic, neuromuscular, congenital, or genetic disorders.
Findings can help providers advise patients and their families about tonsillectomy risks
Kavita Dedhia, MD, MSHP, attending otolaryngologist, Division of Otolaryngology, Children’s Hospital of Philadelphia, Pennsylvania, told this news organization that she was not surprised by the findings.
“This study suggests that mortality is an extremely rare complication of tonsillectomy, and that children with complex medical conditions are at highest risk,” Dr. Dedhia, who was not involved in the study, said in an email.
“Due to their underlying comorbidities, medically fragile children are considered to be at higher risk while undergoing anesthesia and surgical procedures,” she added.
Dr. Dedhia noted that nonpatient factors the study did not explore may have affected the mortality rates, including each hospital’s experience with managing children with complex medical conditions, as well as whether the hospitals were tertiary care facilities, and pediatric or adult hospitals.
She would like to know what hospital or practice characteristics may have contributed to the mortality risk and whether increased mortality in these patients is limited to tonsillectomy or is also found with other surgical procedures.
“The strength of this study is that it is large and multi-regional and that it informs providers about patient factors impacting mortality in pediatric tonsillectomy,” Dr. Dedhia said. “This study arms surgeons with data to discuss mortality risk with the families of medically complex children undergoing tonsillectomy.”
The study authors and Dr. Dedhia report no relevant financial relationships. Funding information was not provided.
A version of this article first appeared on Medscape.com.
It’s rare for a child to die after a tonsillectomy, but children who die are more likely to have a complex chronic condition such as cerebral palsy or Down syndrome, according to a retrospective cohort study published in JAMA.
“Among children undergoing tonsillectomy, the rate of postoperative death was 7 per 100,000 operations overall, [but] among children with complex chronic conditions, the rate of postoperative death was 117 per 100,000 operations, representing 44% of overall deaths,” write researchers at the University of Wisconsin–Madison. “These findings may inform decisionmaking for pediatric tonsillectomy.”
The rate of death in children after tonsillectomy has been uncertain, the authors write. Specific mortality rates for children at increased risk for complications, including those under 3 years old and those with sleep-disordered breathing or complex chronic conditions, have not been available.
To learn how likely children undergoing tonsillectomy are to die after their surgery, as well as which children are most at risk, lead study author M. Bruce Edmonson, MD, MPH, department of pediatrics, University of Wisconsin School of Medicine and Public Health, Madison, and his colleagues drew data from five states, including ambulatory surgery, inpatient, and emergency department discharge data sets provided by the Healthcare Cost and Utilization Project, Agency for Healthcare Research and Quality for California, Florida, Maryland, New York, and Wisconsin.
Participants included 504,262 patients under 21 years of age whose discharge records linked their inpatient or outpatient tonsillectomy, with or without adenoidectomy, with at least 90 days of follow-up.
In a longitudinal analysis, the research team investigated postoperative death within 30 days or during a surgical stay lasting over 30 days. They calculated postoperative mortality per 100,000 operations, both overall and classified by age group, sleep-disordered breathing, and complex chronic conditions.
The 504,262 children ranged in age from 0 to 20 years and underwent a total of 505,182 tonsillectomies. Of these, 10.1% were performed in children aged under 3 years, 28.9% in children with sleep-disordered breathing, and 2.8% in those with complex chronic conditions.
The 36 linked postoperative deaths occurred between 2 and 20.5 days after surgical admission, and 19 (53%) of the deaths occurred after surgical discharge.
The unadjusted mortality rate was 7.04 (95% confidence interval, 4.97-9.98) deaths per 100,000 procedures. In multivariable models, children younger than 3 years and children with sleep-disordered breathing were not significantly more likely to die.
But children with complex chronic conditions were significantly more likely to die than were children without those conditions (117.22 vs. 3.87 deaths per 100,000 procedures, respectively).
Children with complex chronic conditions underwent only 2.8% of all tonsillectomies, but they accounted for 44% of postoperative deaths. Most deaths linked with complex chronic conditions occurred among children with neurologic, neuromuscular, congenital, or genetic disorders.
Findings can help providers advise patients and their families about tonsillectomy risks
Kavita Dedhia, MD, MSHP, attending otolaryngologist, Division of Otolaryngology, Children’s Hospital of Philadelphia, Pennsylvania, told this news organization that she was not surprised by the findings.
“This study suggests that mortality is an extremely rare complication of tonsillectomy, and that children with complex medical conditions are at highest risk,” Dr. Dedhia, who was not involved in the study, said in an email.
“Due to their underlying comorbidities, medically fragile children are considered to be at higher risk while undergoing anesthesia and surgical procedures,” she added.
Dr. Dedhia noted that nonpatient factors the study did not explore may have affected the mortality rates, including each hospital’s experience with managing children with complex medical conditions, as well as whether the hospitals were tertiary care facilities, and pediatric or adult hospitals.
She would like to know what hospital or practice characteristics may have contributed to the mortality risk and whether increased mortality in these patients is limited to tonsillectomy or is also found with other surgical procedures.
“The strength of this study is that it is large and multi-regional and that it informs providers about patient factors impacting mortality in pediatric tonsillectomy,” Dr. Dedhia said. “This study arms surgeons with data to discuss mortality risk with the families of medically complex children undergoing tonsillectomy.”
The study authors and Dr. Dedhia report no relevant financial relationships. Funding information was not provided.
A version of this article first appeared on Medscape.com.
It’s rare for a child to die after a tonsillectomy, but children who die are more likely to have a complex chronic condition such as cerebral palsy or Down syndrome, according to a retrospective cohort study published in JAMA.
“Among children undergoing tonsillectomy, the rate of postoperative death was 7 per 100,000 operations overall, [but] among children with complex chronic conditions, the rate of postoperative death was 117 per 100,000 operations, representing 44% of overall deaths,” write researchers at the University of Wisconsin–Madison. “These findings may inform decisionmaking for pediatric tonsillectomy.”
The rate of death in children after tonsillectomy has been uncertain, the authors write. Specific mortality rates for children at increased risk for complications, including those under 3 years old and those with sleep-disordered breathing or complex chronic conditions, have not been available.
To learn how likely children undergoing tonsillectomy are to die after their surgery, as well as which children are most at risk, lead study author M. Bruce Edmonson, MD, MPH, department of pediatrics, University of Wisconsin School of Medicine and Public Health, Madison, and his colleagues drew data from five states, including ambulatory surgery, inpatient, and emergency department discharge data sets provided by the Healthcare Cost and Utilization Project, Agency for Healthcare Research and Quality for California, Florida, Maryland, New York, and Wisconsin.
Participants included 504,262 patients under 21 years of age whose discharge records linked their inpatient or outpatient tonsillectomy, with or without adenoidectomy, with at least 90 days of follow-up.
In a longitudinal analysis, the research team investigated postoperative death within 30 days or during a surgical stay lasting over 30 days. They calculated postoperative mortality per 100,000 operations, both overall and classified by age group, sleep-disordered breathing, and complex chronic conditions.
The 504,262 children ranged in age from 0 to 20 years and underwent a total of 505,182 tonsillectomies. Of these, 10.1% were performed in children aged under 3 years, 28.9% in children with sleep-disordered breathing, and 2.8% in those with complex chronic conditions.
The 36 linked postoperative deaths occurred between 2 and 20.5 days after surgical admission, and 19 (53%) of the deaths occurred after surgical discharge.
The unadjusted mortality rate was 7.04 (95% confidence interval, 4.97-9.98) deaths per 100,000 procedures. In multivariable models, children younger than 3 years and children with sleep-disordered breathing were not significantly more likely to die.
But children with complex chronic conditions were significantly more likely to die than were children without those conditions (117.22 vs. 3.87 deaths per 100,000 procedures, respectively).
Children with complex chronic conditions underwent only 2.8% of all tonsillectomies, but they accounted for 44% of postoperative deaths. Most deaths linked with complex chronic conditions occurred among children with neurologic, neuromuscular, congenital, or genetic disorders.
Findings can help providers advise patients and their families about tonsillectomy risks
Kavita Dedhia, MD, MSHP, attending otolaryngologist, Division of Otolaryngology, Children’s Hospital of Philadelphia, Pennsylvania, told this news organization that she was not surprised by the findings.
“This study suggests that mortality is an extremely rare complication of tonsillectomy, and that children with complex medical conditions are at highest risk,” Dr. Dedhia, who was not involved in the study, said in an email.
“Due to their underlying comorbidities, medically fragile children are considered to be at higher risk while undergoing anesthesia and surgical procedures,” she added.
Dr. Dedhia noted that nonpatient factors the study did not explore may have affected the mortality rates, including each hospital’s experience with managing children with complex medical conditions, as well as whether the hospitals were tertiary care facilities, and pediatric or adult hospitals.
She would like to know what hospital or practice characteristics may have contributed to the mortality risk and whether increased mortality in these patients is limited to tonsillectomy or is also found with other surgical procedures.
“The strength of this study is that it is large and multi-regional and that it informs providers about patient factors impacting mortality in pediatric tonsillectomy,” Dr. Dedhia said. “This study arms surgeons with data to discuss mortality risk with the families of medically complex children undergoing tonsillectomy.”
The study authors and Dr. Dedhia report no relevant financial relationships. Funding information was not provided.
A version of this article first appeared on Medscape.com.
FROM JAMA
Artificial intelligence: The Netflix of cancer treatment
Chemotherapy, now streaming at just $15.99 a month!
It’s a lazy Sunday and you flip on Netflix, looking for something new to watch. There’s an almost-overwhelming number of shows out there, but right at the top of the recommended list is something that strikes your fancy right away. The algorithm behind the scenes is doing its job well, winnowing the universe of content right down to the few things you’ll find relevant, based on what you’ve watched and liked in the past.
Now, the almighty content algorithm is coming for something a little more useful than binge watching obscure 80s sitcoms: cancer treatment.
By plugging the fully sequenced genomes of nearly 10,000 patients with 33 different types of cancer into an algorithm powered by the same sort of artificial intelligence used by Netflix, researchers from London and San Diego found 21 common faults in the chromosomes of tumors, which they called copy number signatures. While cancer is a complex disease, when faults occur in those copy number signatures, the results were similar across the board. If X genetic defect occurs within a tumor, Y result will happen, even across cancer types. For example, tumors whose chromosomes had shattered and reformed had by far the worst disease outcomes.
The eventual hope is that, just as Netflix can predict what you’ll want to watch based on what you’ve already seen, oncologists will be able to predict the course of a cancer, based on the tumor’s early genetic traits, and get ahead of future genetic degradation to prevent the worst outcomes. A sort of “Oh, your tumor has enjoyed The Office. Might we suggest a treatment of 30 Rock” situation. Further research will be required to determine whether or not the cancer algorithm can get us part 2 of “Stranger Things 4” a week early.
Pay criminals, cut crime?
What is the best method for punishing those who commit wrongdoing? Fines? Jail time? Actually, no. A recent study says that financial compensation works best.
In other words, pay them for their actions. Really.
Psychologist Tage S. Rai, PhD, of the University of California, San Diego, Rady School of Management, found that people who hurt others or commit crimes are actually doing it because they think it’s the right thing to do. The results of this study say play at the angle of their morality. When that’s compromised, the offender is less likely to do it again.
Four different experiments were conducted using an online economics game with nearly 1,500 participants. Dr. Rai found that providing a monetary bonus for inflicting a punishment on a third party within the game cut the participants’ willingness to do it again by 50%.
“People punish others to signal their own goodness and receiving compensation might make it seem as though they’re driven by greed rather than justice,” he said.
The big deterrent, though, was negative judgment from peers. People in the study were even more hesitant to inflict harm and gain a profit if they thought they were going to be judged for it.
So maybe the answer to cutting crime isn’t as simple as slapping on a fine. It’s slapping on shame and paying them for it.
A conspiracy of chronobiologic proportions
The Golden State Warriors just won the NBA championship – that much is true – but we’ve got some news that you didn’t get from ESPN. The kind of news that their “partners” from the NBA didn’t want them to report. Unlike most conspiracy theories, however, this one has some science behind it.
In this case, science in the form of a study published in Frontiers in Physiology says that jet lag had a greater effect on the Boston Celtics than it did on the Warriors.
“Eastward travel – where the destination time is later than the origin time – requires the athlete to shorten their day (known as a phase advance). During phase advance, athletes often struggle to fall asleep at an earlier bedtime, leading to sleep loss and, consequently, potential impaired physiological performance and motivation the next day,” senior author Elise Facer-Childs, PhD, of Monash University, Melbourne, said in written statement.
Dr. Facer-Childs and associates took a very close look at 10 seasons’ worth of NBA games – 11,481 games, to be exact – and found “that eastward (but not westward) jet lag was associated with impaired performance for home (but not away) teams.” The existence of a pro-Western bias against teams that traveled eastward for their home games was clear:
- The chance of winning for eastern teams was reduced by 6.0%.
- They grabbed 1.3 fewer rebounds per game.
- Their field goal percentage was 1.2% lower.
And here’s the final nail in the conspiracy coffin: The NBA knew about the jet lag effect and changed the schedule of the finals in 2014 in a way that makes it worse. Before that, the higher-seeded team got two home games, then the lower-seeded team had three at home, followed by two more at the home of the higher seed. Now it’s a 2-2-1-1-1 arrangement that leads to more travel and, of course, more jet lag.
The study was published during the championship series, so the investigators suggested that the Celtics “might benefit from chronobiology-informed strategies designed to mitigate eastward jet lag symptomatology.”
So there you have it, sports fans/conspiracy theorists: You can’t chase Steph Curry around the court for 48 minutes without the right chronobiology-informed strategy. Everyone knows that.
Being hungry can alter your ‘type’
Fasting and being hungry can be a dangerous mix for becoming “hangry” and irritable, but did you know being hungry can also affect your attraction to other people?
Evidence has shown that being hungry can affect important things such as decision-making, memory, cognition, and function. It might affect decision-making in the sense that those six tacos at Taco Bell might win out over grilled chicken breast and veggies at home, but can hunger make you think that the person you just swiped right on isn’t really your type after all?
We’ll leave that up to Valentina Cazzato of Liverpool (England) John Moores University and associates, whose study involved 44 people, of whom 21 were women in their early 20s. The participants were shown computer-generated images of men and women of different sizes. The same background was used for each picture and all the expressions of the models were neutral. Participants were asked to rate each image on how much they liked it. One study was done on participants who had been fasting for 12 hours, and the second was done on those who had just eaten something.
The subjects generally preferred slim models over more rounded ones, but not after fasting. When they were hungry, they found the round human bodies and faces more attractive. So, yes, it’s definitely possible that hunger can alter your attraction to others.
“Future work might seek to elucidate the relationship between physiological states of hunger and shifts in appreciation of the human bodies and whether this relationship might be mediated by individual traits associated with to beholder’s body adiposity,” said researchers.
Chemotherapy, now streaming at just $15.99 a month!
It’s a lazy Sunday and you flip on Netflix, looking for something new to watch. There’s an almost-overwhelming number of shows out there, but right at the top of the recommended list is something that strikes your fancy right away. The algorithm behind the scenes is doing its job well, winnowing the universe of content right down to the few things you’ll find relevant, based on what you’ve watched and liked in the past.
Now, the almighty content algorithm is coming for something a little more useful than binge watching obscure 80s sitcoms: cancer treatment.
By plugging the fully sequenced genomes of nearly 10,000 patients with 33 different types of cancer into an algorithm powered by the same sort of artificial intelligence used by Netflix, researchers from London and San Diego found 21 common faults in the chromosomes of tumors, which they called copy number signatures. While cancer is a complex disease, when faults occur in those copy number signatures, the results were similar across the board. If X genetic defect occurs within a tumor, Y result will happen, even across cancer types. For example, tumors whose chromosomes had shattered and reformed had by far the worst disease outcomes.
The eventual hope is that, just as Netflix can predict what you’ll want to watch based on what you’ve already seen, oncologists will be able to predict the course of a cancer, based on the tumor’s early genetic traits, and get ahead of future genetic degradation to prevent the worst outcomes. A sort of “Oh, your tumor has enjoyed The Office. Might we suggest a treatment of 30 Rock” situation. Further research will be required to determine whether or not the cancer algorithm can get us part 2 of “Stranger Things 4” a week early.
Pay criminals, cut crime?
What is the best method for punishing those who commit wrongdoing? Fines? Jail time? Actually, no. A recent study says that financial compensation works best.
In other words, pay them for their actions. Really.
Psychologist Tage S. Rai, PhD, of the University of California, San Diego, Rady School of Management, found that people who hurt others or commit crimes are actually doing it because they think it’s the right thing to do. The results of this study say play at the angle of their morality. When that’s compromised, the offender is less likely to do it again.
Four different experiments were conducted using an online economics game with nearly 1,500 participants. Dr. Rai found that providing a monetary bonus for inflicting a punishment on a third party within the game cut the participants’ willingness to do it again by 50%.
“People punish others to signal their own goodness and receiving compensation might make it seem as though they’re driven by greed rather than justice,” he said.
The big deterrent, though, was negative judgment from peers. People in the study were even more hesitant to inflict harm and gain a profit if they thought they were going to be judged for it.
So maybe the answer to cutting crime isn’t as simple as slapping on a fine. It’s slapping on shame and paying them for it.
A conspiracy of chronobiologic proportions
The Golden State Warriors just won the NBA championship – that much is true – but we’ve got some news that you didn’t get from ESPN. The kind of news that their “partners” from the NBA didn’t want them to report. Unlike most conspiracy theories, however, this one has some science behind it.
In this case, science in the form of a study published in Frontiers in Physiology says that jet lag had a greater effect on the Boston Celtics than it did on the Warriors.
“Eastward travel – where the destination time is later than the origin time – requires the athlete to shorten their day (known as a phase advance). During phase advance, athletes often struggle to fall asleep at an earlier bedtime, leading to sleep loss and, consequently, potential impaired physiological performance and motivation the next day,” senior author Elise Facer-Childs, PhD, of Monash University, Melbourne, said in written statement.
Dr. Facer-Childs and associates took a very close look at 10 seasons’ worth of NBA games – 11,481 games, to be exact – and found “that eastward (but not westward) jet lag was associated with impaired performance for home (but not away) teams.” The existence of a pro-Western bias against teams that traveled eastward for their home games was clear:
- The chance of winning for eastern teams was reduced by 6.0%.
- They grabbed 1.3 fewer rebounds per game.
- Their field goal percentage was 1.2% lower.
And here’s the final nail in the conspiracy coffin: The NBA knew about the jet lag effect and changed the schedule of the finals in 2014 in a way that makes it worse. Before that, the higher-seeded team got two home games, then the lower-seeded team had three at home, followed by two more at the home of the higher seed. Now it’s a 2-2-1-1-1 arrangement that leads to more travel and, of course, more jet lag.
The study was published during the championship series, so the investigators suggested that the Celtics “might benefit from chronobiology-informed strategies designed to mitigate eastward jet lag symptomatology.”
So there you have it, sports fans/conspiracy theorists: You can’t chase Steph Curry around the court for 48 minutes without the right chronobiology-informed strategy. Everyone knows that.
Being hungry can alter your ‘type’
Fasting and being hungry can be a dangerous mix for becoming “hangry” and irritable, but did you know being hungry can also affect your attraction to other people?
Evidence has shown that being hungry can affect important things such as decision-making, memory, cognition, and function. It might affect decision-making in the sense that those six tacos at Taco Bell might win out over grilled chicken breast and veggies at home, but can hunger make you think that the person you just swiped right on isn’t really your type after all?
We’ll leave that up to Valentina Cazzato of Liverpool (England) John Moores University and associates, whose study involved 44 people, of whom 21 were women in their early 20s. The participants were shown computer-generated images of men and women of different sizes. The same background was used for each picture and all the expressions of the models were neutral. Participants were asked to rate each image on how much they liked it. One study was done on participants who had been fasting for 12 hours, and the second was done on those who had just eaten something.
The subjects generally preferred slim models over more rounded ones, but not after fasting. When they were hungry, they found the round human bodies and faces more attractive. So, yes, it’s definitely possible that hunger can alter your attraction to others.
“Future work might seek to elucidate the relationship between physiological states of hunger and shifts in appreciation of the human bodies and whether this relationship might be mediated by individual traits associated with to beholder’s body adiposity,” said researchers.
Chemotherapy, now streaming at just $15.99 a month!
It’s a lazy Sunday and you flip on Netflix, looking for something new to watch. There’s an almost-overwhelming number of shows out there, but right at the top of the recommended list is something that strikes your fancy right away. The algorithm behind the scenes is doing its job well, winnowing the universe of content right down to the few things you’ll find relevant, based on what you’ve watched and liked in the past.
Now, the almighty content algorithm is coming for something a little more useful than binge watching obscure 80s sitcoms: cancer treatment.
By plugging the fully sequenced genomes of nearly 10,000 patients with 33 different types of cancer into an algorithm powered by the same sort of artificial intelligence used by Netflix, researchers from London and San Diego found 21 common faults in the chromosomes of tumors, which they called copy number signatures. While cancer is a complex disease, when faults occur in those copy number signatures, the results were similar across the board. If X genetic defect occurs within a tumor, Y result will happen, even across cancer types. For example, tumors whose chromosomes had shattered and reformed had by far the worst disease outcomes.
The eventual hope is that, just as Netflix can predict what you’ll want to watch based on what you’ve already seen, oncologists will be able to predict the course of a cancer, based on the tumor’s early genetic traits, and get ahead of future genetic degradation to prevent the worst outcomes. A sort of “Oh, your tumor has enjoyed The Office. Might we suggest a treatment of 30 Rock” situation. Further research will be required to determine whether or not the cancer algorithm can get us part 2 of “Stranger Things 4” a week early.
Pay criminals, cut crime?
What is the best method for punishing those who commit wrongdoing? Fines? Jail time? Actually, no. A recent study says that financial compensation works best.
In other words, pay them for their actions. Really.
Psychologist Tage S. Rai, PhD, of the University of California, San Diego, Rady School of Management, found that people who hurt others or commit crimes are actually doing it because they think it’s the right thing to do. The results of this study say play at the angle of their morality. When that’s compromised, the offender is less likely to do it again.
Four different experiments were conducted using an online economics game with nearly 1,500 participants. Dr. Rai found that providing a monetary bonus for inflicting a punishment on a third party within the game cut the participants’ willingness to do it again by 50%.
“People punish others to signal their own goodness and receiving compensation might make it seem as though they’re driven by greed rather than justice,” he said.
The big deterrent, though, was negative judgment from peers. People in the study were even more hesitant to inflict harm and gain a profit if they thought they were going to be judged for it.
So maybe the answer to cutting crime isn’t as simple as slapping on a fine. It’s slapping on shame and paying them for it.
A conspiracy of chronobiologic proportions
The Golden State Warriors just won the NBA championship – that much is true – but we’ve got some news that you didn’t get from ESPN. The kind of news that their “partners” from the NBA didn’t want them to report. Unlike most conspiracy theories, however, this one has some science behind it.
In this case, science in the form of a study published in Frontiers in Physiology says that jet lag had a greater effect on the Boston Celtics than it did on the Warriors.
“Eastward travel – where the destination time is later than the origin time – requires the athlete to shorten their day (known as a phase advance). During phase advance, athletes often struggle to fall asleep at an earlier bedtime, leading to sleep loss and, consequently, potential impaired physiological performance and motivation the next day,” senior author Elise Facer-Childs, PhD, of Monash University, Melbourne, said in written statement.
Dr. Facer-Childs and associates took a very close look at 10 seasons’ worth of NBA games – 11,481 games, to be exact – and found “that eastward (but not westward) jet lag was associated with impaired performance for home (but not away) teams.” The existence of a pro-Western bias against teams that traveled eastward for their home games was clear:
- The chance of winning for eastern teams was reduced by 6.0%.
- They grabbed 1.3 fewer rebounds per game.
- Their field goal percentage was 1.2% lower.
And here’s the final nail in the conspiracy coffin: The NBA knew about the jet lag effect and changed the schedule of the finals in 2014 in a way that makes it worse. Before that, the higher-seeded team got two home games, then the lower-seeded team had three at home, followed by two more at the home of the higher seed. Now it’s a 2-2-1-1-1 arrangement that leads to more travel and, of course, more jet lag.
The study was published during the championship series, so the investigators suggested that the Celtics “might benefit from chronobiology-informed strategies designed to mitigate eastward jet lag symptomatology.”
So there you have it, sports fans/conspiracy theorists: You can’t chase Steph Curry around the court for 48 minutes without the right chronobiology-informed strategy. Everyone knows that.
Being hungry can alter your ‘type’
Fasting and being hungry can be a dangerous mix for becoming “hangry” and irritable, but did you know being hungry can also affect your attraction to other people?
Evidence has shown that being hungry can affect important things such as decision-making, memory, cognition, and function. It might affect decision-making in the sense that those six tacos at Taco Bell might win out over grilled chicken breast and veggies at home, but can hunger make you think that the person you just swiped right on isn’t really your type after all?
We’ll leave that up to Valentina Cazzato of Liverpool (England) John Moores University and associates, whose study involved 44 people, of whom 21 were women in their early 20s. The participants were shown computer-generated images of men and women of different sizes. The same background was used for each picture and all the expressions of the models were neutral. Participants were asked to rate each image on how much they liked it. One study was done on participants who had been fasting for 12 hours, and the second was done on those who had just eaten something.
The subjects generally preferred slim models over more rounded ones, but not after fasting. When they were hungry, they found the round human bodies and faces more attractive. So, yes, it’s definitely possible that hunger can alter your attraction to others.
“Future work might seek to elucidate the relationship between physiological states of hunger and shifts in appreciation of the human bodies and whether this relationship might be mediated by individual traits associated with to beholder’s body adiposity,” said researchers.
A doctor’s missed diagnosis results in mega award
, according to a story from WCCO CBS Minnesota, among other news outlets. The award has been called the largest judgment of its kind in Minnesota history.
In January 2017, Nepalese immigrant Anuj Thapa was playing in an indoor soccer game at St. Cloud State University when another player tackled him. His left leg badly injured, Mr. Thapa was taken by ambulance to CentraCare’s St. Cloud Hospital. The orthopedic surgeon on call that day was Chad Holien, MD, who is affiliated with St. Cloud Orthopedics, a private clinic in nearby Sartell, Minn. Following preparations, and with the help of a physician assistant, Dr. Holien operated on the patient’s broken leg.
But Mr. Thapa experienced post-surgical complications – severe pain, numbness, burning, and muscle issues. Despite the complications, he was discharged from the hospital that afternoon and sent home.
Six days later, Mr. Thapa returned to St. Cloud Hospital, still complaining of severe pain. A second orthopedic surgeon operated and found that Mr. Thapa had “acute compartment syndrome,” the result of internal pressure that had built up in his leg muscles.
Over time, Mr. Thapa underwent more than 20 surgeries on his leg to deal with the ongoing pain and other complications, according to WCCO.
In 2019, he filed a medical malpractice suit in U.S. district court against St. Cloud Orthopedics, the private practice that employed the surgeon and the PA. (Under Minnesota law, an employer is responsible for the actions of its employees.)
In his complaint, Mr. Thapa alleged that in treating him, “the defendants departed from accepted standards of medical practice.” Among other things, he claimed that Dr. Holien and the PA had not properly evaluated his postoperative symptoms, failed to diagnose and treat his compartment syndrome, and improperly discharged him from the hospital. These lapses, Mr. Thapa said, led to his “severe, permanent, and disabling injuries.”
The federal jury agreed. After a weeklong trial, it awarded the plaintiff $100 million for future “pain, disability, disfigurement, embarrassment, and emotional distress.” It also gave him $10 million for past suffering and a little more than $1 million for past and future medical bills.
In a postverdict statement, Mr. Thapa’s attorney said that, while the surgeon and PA are undoubtedly good providers, they made mistakes in this case.
A defense attorney for St. Cloud Orthopedics disputes this: “We maintain the care provided in this case was in accordance with accepted standards of care.”
At press time, the defense had not determined whether to appeal the jury’s $111 million verdict. “St. Cloud continues to support its providers,” said the clinic’s defense attorney. “We are evaluating our options regarding this verdict.”
The content contained in this article is for informational purposes only and does not constitute legal advice. Reliance on any information provided in this article is solely at your own risk.
A version of this article first appeared on Medscape.com.
, according to a story from WCCO CBS Minnesota, among other news outlets. The award has been called the largest judgment of its kind in Minnesota history.
In January 2017, Nepalese immigrant Anuj Thapa was playing in an indoor soccer game at St. Cloud State University when another player tackled him. His left leg badly injured, Mr. Thapa was taken by ambulance to CentraCare’s St. Cloud Hospital. The orthopedic surgeon on call that day was Chad Holien, MD, who is affiliated with St. Cloud Orthopedics, a private clinic in nearby Sartell, Minn. Following preparations, and with the help of a physician assistant, Dr. Holien operated on the patient’s broken leg.
But Mr. Thapa experienced post-surgical complications – severe pain, numbness, burning, and muscle issues. Despite the complications, he was discharged from the hospital that afternoon and sent home.
Six days later, Mr. Thapa returned to St. Cloud Hospital, still complaining of severe pain. A second orthopedic surgeon operated and found that Mr. Thapa had “acute compartment syndrome,” the result of internal pressure that had built up in his leg muscles.
Over time, Mr. Thapa underwent more than 20 surgeries on his leg to deal with the ongoing pain and other complications, according to WCCO.
In 2019, he filed a medical malpractice suit in U.S. district court against St. Cloud Orthopedics, the private practice that employed the surgeon and the PA. (Under Minnesota law, an employer is responsible for the actions of its employees.)
In his complaint, Mr. Thapa alleged that in treating him, “the defendants departed from accepted standards of medical practice.” Among other things, he claimed that Dr. Holien and the PA had not properly evaluated his postoperative symptoms, failed to diagnose and treat his compartment syndrome, and improperly discharged him from the hospital. These lapses, Mr. Thapa said, led to his “severe, permanent, and disabling injuries.”
The federal jury agreed. After a weeklong trial, it awarded the plaintiff $100 million for future “pain, disability, disfigurement, embarrassment, and emotional distress.” It also gave him $10 million for past suffering and a little more than $1 million for past and future medical bills.
In a postverdict statement, Mr. Thapa’s attorney said that, while the surgeon and PA are undoubtedly good providers, they made mistakes in this case.
A defense attorney for St. Cloud Orthopedics disputes this: “We maintain the care provided in this case was in accordance with accepted standards of care.”
At press time, the defense had not determined whether to appeal the jury’s $111 million verdict. “St. Cloud continues to support its providers,” said the clinic’s defense attorney. “We are evaluating our options regarding this verdict.”
The content contained in this article is for informational purposes only and does not constitute legal advice. Reliance on any information provided in this article is solely at your own risk.
A version of this article first appeared on Medscape.com.
, according to a story from WCCO CBS Minnesota, among other news outlets. The award has been called the largest judgment of its kind in Minnesota history.
In January 2017, Nepalese immigrant Anuj Thapa was playing in an indoor soccer game at St. Cloud State University when another player tackled him. His left leg badly injured, Mr. Thapa was taken by ambulance to CentraCare’s St. Cloud Hospital. The orthopedic surgeon on call that day was Chad Holien, MD, who is affiliated with St. Cloud Orthopedics, a private clinic in nearby Sartell, Minn. Following preparations, and with the help of a physician assistant, Dr. Holien operated on the patient’s broken leg.
But Mr. Thapa experienced post-surgical complications – severe pain, numbness, burning, and muscle issues. Despite the complications, he was discharged from the hospital that afternoon and sent home.
Six days later, Mr. Thapa returned to St. Cloud Hospital, still complaining of severe pain. A second orthopedic surgeon operated and found that Mr. Thapa had “acute compartment syndrome,” the result of internal pressure that had built up in his leg muscles.
Over time, Mr. Thapa underwent more than 20 surgeries on his leg to deal with the ongoing pain and other complications, according to WCCO.
In 2019, he filed a medical malpractice suit in U.S. district court against St. Cloud Orthopedics, the private practice that employed the surgeon and the PA. (Under Minnesota law, an employer is responsible for the actions of its employees.)
In his complaint, Mr. Thapa alleged that in treating him, “the defendants departed from accepted standards of medical practice.” Among other things, he claimed that Dr. Holien and the PA had not properly evaluated his postoperative symptoms, failed to diagnose and treat his compartment syndrome, and improperly discharged him from the hospital. These lapses, Mr. Thapa said, led to his “severe, permanent, and disabling injuries.”
The federal jury agreed. After a weeklong trial, it awarded the plaintiff $100 million for future “pain, disability, disfigurement, embarrassment, and emotional distress.” It also gave him $10 million for past suffering and a little more than $1 million for past and future medical bills.
In a postverdict statement, Mr. Thapa’s attorney said that, while the surgeon and PA are undoubtedly good providers, they made mistakes in this case.
A defense attorney for St. Cloud Orthopedics disputes this: “We maintain the care provided in this case was in accordance with accepted standards of care.”
At press time, the defense had not determined whether to appeal the jury’s $111 million verdict. “St. Cloud continues to support its providers,” said the clinic’s defense attorney. “We are evaluating our options regarding this verdict.”
The content contained in this article is for informational purposes only and does not constitute legal advice. Reliance on any information provided in this article is solely at your own risk.
A version of this article first appeared on Medscape.com.
Care gaps common after anal sphincter injuries from childbirth
Postpartum complications may go unrecognized in women who incur anal sphincter injuries during childbirth, a review of electronic medical records at one academic health system suggests.
In the first 3 months after delivery, few patients with an obstetric anal sphincter injury (OASI) had documented pelvic floor problems, compared with higher rates documented in medical literature, the researchers found.
“Lack of identified pelvic floor dysfunction in this population differs from the incidence in previously published data and may reflect lack of identification by obstetric providers,” the researchers reported. The findings “highlight a gap in health care that, when addressed, could significantly improve postpartum quality of life.”
The findings are scheduled to be presented at the annual scientific meeting of the American Urogynecologic Society and International Urogynecological Association.
Anal sphincter injuries occur in about 4.4% of vaginal deliveries and are the most common cause of anal incontinence in women of reproductive age.
For the new study, researchers reviewed records of 287 women who underwent a vaginal birth that resulted in an anal sphincter injury at five Ohio hospitals affiliated with Cleveland Clinic from 2013 to 2015.
Of those who met eligibility criteria, 209 (72.8%) were White, 262 (91.3%) were non-Hispanic, and 249 (86.8%) were aged 20-34 years. Most had an epidural (92%), did not require a blood transfusion (97.9%), did not develop a vaginal hematoma (98.9%), and did not have their injury repaired in an operating room (97.2%), the researchers reported.
Among pelvic floor disorders, urinary incontinence was not reported in 96% of patients, fecal incontinence was not reported in 97.1%, and pelvic organ prolapse was not reported in 99.3%. Most had no recorded complications from their lacerations (87.8%) or postpartum depression (92%), the researchers found.
However, a 2015 study found that, 12 weeks after delivery, women with OASIs commonly reported symptoms of incontinence, with 26% reporting urinary stress incontinence, 21.4% urinary urgency incontinence, 59% anal incontinence, and 15% fecal incontinence.
Depression was also seldom identified despite higher risk of mood disorders among women with OASI, the researchers found.
The team also examined interpregnancy intervals, defined as the time between a woman’s first vaginal delivery and conception of a subsequent pregnancy. Of 178 women for whom data were available, the median interval was 26.4 months (95% confidence interval, 23.7-29.9), similar to the median for births nationally.
Lead researcher Alexandra Nutaitis, DO, a resident in obstetrics and gynecology at Cleveland Clinic Akron General, said in an interview that it’s unclear whether physicians did not inquire about symptoms or didn’t record them. She noted that anal sphincter injuries are a “stigmatized topic.”
Not asked, not told
Carolyn Swenson, MD, an associate professor in urogynecology at the University of Utah, Salt Lake City, said physicians in the study may have relied on patients to bring up their symptoms rather than using questionnaires to screen for problems.
“What we know is that if you don’t ask women about pelvic floor disorders, they often don’t tell you that they are experiencing symptoms,” said Dr. Swenson, who was not involved in the new research.
Dr. Swenson called for validated questionnaires to assess pelvic floor symptoms in postpartum patients.
Regarding interpregnancy intervals, Dr. Nutaitis said she would be surprised if women who experienced an OASI didn’t delay having another child longer than women who did not undergo that physical and psychological trauma – but other factors such as societal pressures may override any reluctance to proceed with another pregnancy.
Dr. Swenson said it’s possible that a subgroup of women who have severe complications, such as those with a fourth-degree tear, might put off having another child. However, more research is needed to find out, she said.
Dr. Nutaitis and Dr. Swenson disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Postpartum complications may go unrecognized in women who incur anal sphincter injuries during childbirth, a review of electronic medical records at one academic health system suggests.
In the first 3 months after delivery, few patients with an obstetric anal sphincter injury (OASI) had documented pelvic floor problems, compared with higher rates documented in medical literature, the researchers found.
“Lack of identified pelvic floor dysfunction in this population differs from the incidence in previously published data and may reflect lack of identification by obstetric providers,” the researchers reported. The findings “highlight a gap in health care that, when addressed, could significantly improve postpartum quality of life.”
The findings are scheduled to be presented at the annual scientific meeting of the American Urogynecologic Society and International Urogynecological Association.
Anal sphincter injuries occur in about 4.4% of vaginal deliveries and are the most common cause of anal incontinence in women of reproductive age.
For the new study, researchers reviewed records of 287 women who underwent a vaginal birth that resulted in an anal sphincter injury at five Ohio hospitals affiliated with Cleveland Clinic from 2013 to 2015.
Of those who met eligibility criteria, 209 (72.8%) were White, 262 (91.3%) were non-Hispanic, and 249 (86.8%) were aged 20-34 years. Most had an epidural (92%), did not require a blood transfusion (97.9%), did not develop a vaginal hematoma (98.9%), and did not have their injury repaired in an operating room (97.2%), the researchers reported.
Among pelvic floor disorders, urinary incontinence was not reported in 96% of patients, fecal incontinence was not reported in 97.1%, and pelvic organ prolapse was not reported in 99.3%. Most had no recorded complications from their lacerations (87.8%) or postpartum depression (92%), the researchers found.
However, a 2015 study found that, 12 weeks after delivery, women with OASIs commonly reported symptoms of incontinence, with 26% reporting urinary stress incontinence, 21.4% urinary urgency incontinence, 59% anal incontinence, and 15% fecal incontinence.
Depression was also seldom identified despite higher risk of mood disorders among women with OASI, the researchers found.
The team also examined interpregnancy intervals, defined as the time between a woman’s first vaginal delivery and conception of a subsequent pregnancy. Of 178 women for whom data were available, the median interval was 26.4 months (95% confidence interval, 23.7-29.9), similar to the median for births nationally.
Lead researcher Alexandra Nutaitis, DO, a resident in obstetrics and gynecology at Cleveland Clinic Akron General, said in an interview that it’s unclear whether physicians did not inquire about symptoms or didn’t record them. She noted that anal sphincter injuries are a “stigmatized topic.”
Not asked, not told
Carolyn Swenson, MD, an associate professor in urogynecology at the University of Utah, Salt Lake City, said physicians in the study may have relied on patients to bring up their symptoms rather than using questionnaires to screen for problems.
“What we know is that if you don’t ask women about pelvic floor disorders, they often don’t tell you that they are experiencing symptoms,” said Dr. Swenson, who was not involved in the new research.
Dr. Swenson called for validated questionnaires to assess pelvic floor symptoms in postpartum patients.
Regarding interpregnancy intervals, Dr. Nutaitis said she would be surprised if women who experienced an OASI didn’t delay having another child longer than women who did not undergo that physical and psychological trauma – but other factors such as societal pressures may override any reluctance to proceed with another pregnancy.
Dr. Swenson said it’s possible that a subgroup of women who have severe complications, such as those with a fourth-degree tear, might put off having another child. However, more research is needed to find out, she said.
Dr. Nutaitis and Dr. Swenson disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Postpartum complications may go unrecognized in women who incur anal sphincter injuries during childbirth, a review of electronic medical records at one academic health system suggests.
In the first 3 months after delivery, few patients with an obstetric anal sphincter injury (OASI) had documented pelvic floor problems, compared with higher rates documented in medical literature, the researchers found.
“Lack of identified pelvic floor dysfunction in this population differs from the incidence in previously published data and may reflect lack of identification by obstetric providers,” the researchers reported. The findings “highlight a gap in health care that, when addressed, could significantly improve postpartum quality of life.”
The findings are scheduled to be presented at the annual scientific meeting of the American Urogynecologic Society and International Urogynecological Association.
Anal sphincter injuries occur in about 4.4% of vaginal deliveries and are the most common cause of anal incontinence in women of reproductive age.
For the new study, researchers reviewed records of 287 women who underwent a vaginal birth that resulted in an anal sphincter injury at five Ohio hospitals affiliated with Cleveland Clinic from 2013 to 2015.
Of those who met eligibility criteria, 209 (72.8%) were White, 262 (91.3%) were non-Hispanic, and 249 (86.8%) were aged 20-34 years. Most had an epidural (92%), did not require a blood transfusion (97.9%), did not develop a vaginal hematoma (98.9%), and did not have their injury repaired in an operating room (97.2%), the researchers reported.
Among pelvic floor disorders, urinary incontinence was not reported in 96% of patients, fecal incontinence was not reported in 97.1%, and pelvic organ prolapse was not reported in 99.3%. Most had no recorded complications from their lacerations (87.8%) or postpartum depression (92%), the researchers found.
However, a 2015 study found that, 12 weeks after delivery, women with OASIs commonly reported symptoms of incontinence, with 26% reporting urinary stress incontinence, 21.4% urinary urgency incontinence, 59% anal incontinence, and 15% fecal incontinence.
Depression was also seldom identified despite higher risk of mood disorders among women with OASI, the researchers found.
The team also examined interpregnancy intervals, defined as the time between a woman’s first vaginal delivery and conception of a subsequent pregnancy. Of 178 women for whom data were available, the median interval was 26.4 months (95% confidence interval, 23.7-29.9), similar to the median for births nationally.
Lead researcher Alexandra Nutaitis, DO, a resident in obstetrics and gynecology at Cleveland Clinic Akron General, said in an interview that it’s unclear whether physicians did not inquire about symptoms or didn’t record them. She noted that anal sphincter injuries are a “stigmatized topic.”
Not asked, not told
Carolyn Swenson, MD, an associate professor in urogynecology at the University of Utah, Salt Lake City, said physicians in the study may have relied on patients to bring up their symptoms rather than using questionnaires to screen for problems.
“What we know is that if you don’t ask women about pelvic floor disorders, they often don’t tell you that they are experiencing symptoms,” said Dr. Swenson, who was not involved in the new research.
Dr. Swenson called for validated questionnaires to assess pelvic floor symptoms in postpartum patients.
Regarding interpregnancy intervals, Dr. Nutaitis said she would be surprised if women who experienced an OASI didn’t delay having another child longer than women who did not undergo that physical and psychological trauma – but other factors such as societal pressures may override any reluctance to proceed with another pregnancy.
Dr. Swenson said it’s possible that a subgroup of women who have severe complications, such as those with a fourth-degree tear, might put off having another child. However, more research is needed to find out, she said.
Dr. Nutaitis and Dr. Swenson disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AUGS 2022
Microbiome’s new happy place: The beer gut
Your gut microbiome will thank you later
A healthy gut seems like the new catch-all to better overall health these days. Nutrition and diet culture has us drinking kombucha and ginger tea and coffee, but what if we told you that going to happy hour might also help?
In a recent double-blind study published in the Journal of Agricultural and Food Chemistry, 19 men were divided into two groups and asked to drink 11 ounces of alcoholic lager (5.2% by volume) or nonalcoholic lager with dinner for 4 weeks.
Beer? Yes. Beer.
We humans have trillions of microorganisms running rampant through our digestive tracts. When they’re happy, we have a lower chance of developing heart disease and diabetes. You know what else has millions of happy microorganisms from fermentation? Beer. It also has polyphenols that can help the body’s tissues fight cancers, as well as heart disease and inflammation. So beer is looking a little more healthy now, isn’t it?
In the study, the researchers found that both the alcoholic- and nonalcoholic-lager groups had a boost in bacterial diversity in the gut and higher fecal alkaline phosphatase levels, which showed improved intestinal health. They acknowledged, however, that the nonalcoholic route would be safer and healthier for overall health.
So add a lager to the list of gut-healthy foods that you should be consuming. It may give the phrase “beer gut” a whole new meaning.
We’ve lost our minds, but at least we know how fast they’re going
The phrase “quantum consciousness” sounds like something out of a particularly cheesy episode of Star Trek: “Oh no, Captain, the quantum consciousness has invaded our computer, and the only way to drive it out is to reverse the polarity of a focused tachyon beam.”
When it comes to understanding such basic existential issues as the origin of consciousness, however, quantum mechanics wasn’t off the table. The theory of the quantum origin of consciousness dates back to the 1990s (thanks in part to noted physician Roger Penrose), and goes something like this: There are microtubules within neurons in the brain that are small enough and isolated enough from the warm, wet, and chaotic brain environment where quantum effects can briefly come into play. We’re talking miniscule fractions of a second here, but still, long enough for quantum calculations to take place in the form of system wavefunction collapse, courtesy of gravity.
To plunge even deeper into the rabbit hole of quantum mechanics, the reason Schrödinger’s cat doesn’t occur in real life is wavefunction collapse; the more massive a quantum system is, the more likely it is to collapse into one state or another (alive or dead, in the cat’s case). The quantum origin of consciousness, or Orch OR theory, holds that human consciousness arises from electrical oscillations within the neuronal microtubules caused by the computations stemming from the collapse of small quantum systems.
That is an awful lot of overly simplified explanation, especially considering the study that just came out essentially disproved it. Oops. The research, published in Physics of Life Reviews, is pretty simple. The researchers went to a lab deep underground to avoid interference from cosmic rays, and sat around for months, observing a chunk of germanium for signs of spontaneous radiation, attributable to the same sort of wavefunction collapse that is supposedly occurring in our brains. They found nothing out of the ordinary, pretty definitively disproving most of Orch OR theory.
The researchers were unwilling to completely dismiss the idea (this is quantum mechanics, after all, uncertainty kind of goes with the territory), but it does seem like we’ll have to search elsewhere for sources of human consciousness. Personally, we’re big fans of the cymbal-playing monkey.
Missing links: A real fish story
Dear LOTME:
Ear’s a question that’s been keeping me up at night. Is the human middle ear the result of top-secret government experiments involving alien technology, Abraham Lincoln, and the Illuminati?
Restless in Roswell
Dear Restless:
The paleoanthropologic community has been sorting through this mystery for decades, and fossils discovered in China over the past 20 years finally provide a much less conspiratorially satisfying answer.
For some time now, experts in the field have believed that the bones of the human middle ear evolved from the spiracular gill of a fish. The spiracle is a small hole behind each eye that opens to the mouth in some fishes and was used to breathe air in the earliest, most primitive species. But how did we get from spiracle to ear?
The missing links come in the form of the cranial anatomy of Shuyu, a 438-million-year-old, fingernail-sized skull of a jawless fish, and the 419-million-year-old fossil of a completely preserved fish with gill filaments in the first branchial chamber.
“These fossils provided the first anatomical and fossil evidence for a vertebrate spiracle originating from fish gills,” senior author Gai Zhikun, PhD, of the Institute of Vertebrate Paleontology and Paleoanthropology, Beijing, said in a written statement.
In many ways, it seems, we are fish: “Many important structures of human beings can be traced back to our fish ancestors, such as our teeth, jaws, middle ears, etc,” added Zhu Min, PhD, also of the institute.
So, Restless, the next time you hear the soothing sounds of an angry mob storming the Capitol or you chew on a slab, slice, or chunk of mutant, laboratory-produced chicken in your favorite fast-food restaurant, be sure to thank Shuyu.
Can you lend me an ear?
If you thought locusts were only a nuisance, think again. They have their uses. If you take a locust’s ear and put it inside a robot, the robot will be able to hear and receive signals. Who knew?
Researchers from Tel Aviv University in Israel showed the robot’s hearing abilities by giving clap signals that told the robot what to do: One clap means go forward, two claps mean move back. What do you think the robot would do if it heard the clap break from Cha Cha Slide?
“Our task was to replace the robot’s electronic microphone with a dead insect’s ear, use the ear’s ability to detect the electrical signals from the environment, in this case vibrations in the air, and, using a special chip, convert the insect input to that of the robot,” Ben M. Maoz, PhD, said in a statement from the university.
And how does a dead locust ear work in a robot? Well, Dr. Maoz explained: “My laboratory has developed a special device – Ear-on-a-Chip – that allows the ear to be kept alive throughout the experiment by supplying oxygen and food to the organ while allowing the electrical signals to be taken out of the locust’s ear and amplified and transmitted to the robot.”
The research won’t stop at hearing, he said, as the other four senses also will be taken into consideration. This could help us sense dangers in the future, such as earthquakes or diseases. We said it before and we’ll say it again: We’re rooting for you, science!
Your gut microbiome will thank you later
A healthy gut seems like the new catch-all to better overall health these days. Nutrition and diet culture has us drinking kombucha and ginger tea and coffee, but what if we told you that going to happy hour might also help?
In a recent double-blind study published in the Journal of Agricultural and Food Chemistry, 19 men were divided into two groups and asked to drink 11 ounces of alcoholic lager (5.2% by volume) or nonalcoholic lager with dinner for 4 weeks.
Beer? Yes. Beer.
We humans have trillions of microorganisms running rampant through our digestive tracts. When they’re happy, we have a lower chance of developing heart disease and diabetes. You know what else has millions of happy microorganisms from fermentation? Beer. It also has polyphenols that can help the body’s tissues fight cancers, as well as heart disease and inflammation. So beer is looking a little more healthy now, isn’t it?
In the study, the researchers found that both the alcoholic- and nonalcoholic-lager groups had a boost in bacterial diversity in the gut and higher fecal alkaline phosphatase levels, which showed improved intestinal health. They acknowledged, however, that the nonalcoholic route would be safer and healthier for overall health.
So add a lager to the list of gut-healthy foods that you should be consuming. It may give the phrase “beer gut” a whole new meaning.
We’ve lost our minds, but at least we know how fast they’re going
The phrase “quantum consciousness” sounds like something out of a particularly cheesy episode of Star Trek: “Oh no, Captain, the quantum consciousness has invaded our computer, and the only way to drive it out is to reverse the polarity of a focused tachyon beam.”
When it comes to understanding such basic existential issues as the origin of consciousness, however, quantum mechanics wasn’t off the table. The theory of the quantum origin of consciousness dates back to the 1990s (thanks in part to noted physician Roger Penrose), and goes something like this: There are microtubules within neurons in the brain that are small enough and isolated enough from the warm, wet, and chaotic brain environment where quantum effects can briefly come into play. We’re talking miniscule fractions of a second here, but still, long enough for quantum calculations to take place in the form of system wavefunction collapse, courtesy of gravity.
To plunge even deeper into the rabbit hole of quantum mechanics, the reason Schrödinger’s cat doesn’t occur in real life is wavefunction collapse; the more massive a quantum system is, the more likely it is to collapse into one state or another (alive or dead, in the cat’s case). The quantum origin of consciousness, or Orch OR theory, holds that human consciousness arises from electrical oscillations within the neuronal microtubules caused by the computations stemming from the collapse of small quantum systems.
That is an awful lot of overly simplified explanation, especially considering the study that just came out essentially disproved it. Oops. The research, published in Physics of Life Reviews, is pretty simple. The researchers went to a lab deep underground to avoid interference from cosmic rays, and sat around for months, observing a chunk of germanium for signs of spontaneous radiation, attributable to the same sort of wavefunction collapse that is supposedly occurring in our brains. They found nothing out of the ordinary, pretty definitively disproving most of Orch OR theory.
The researchers were unwilling to completely dismiss the idea (this is quantum mechanics, after all, uncertainty kind of goes with the territory), but it does seem like we’ll have to search elsewhere for sources of human consciousness. Personally, we’re big fans of the cymbal-playing monkey.
Missing links: A real fish story
Dear LOTME:
Ear’s a question that’s been keeping me up at night. Is the human middle ear the result of top-secret government experiments involving alien technology, Abraham Lincoln, and the Illuminati?
Restless in Roswell
Dear Restless:
The paleoanthropologic community has been sorting through this mystery for decades, and fossils discovered in China over the past 20 years finally provide a much less conspiratorially satisfying answer.
For some time now, experts in the field have believed that the bones of the human middle ear evolved from the spiracular gill of a fish. The spiracle is a small hole behind each eye that opens to the mouth in some fishes and was used to breathe air in the earliest, most primitive species. But how did we get from spiracle to ear?
The missing links come in the form of the cranial anatomy of Shuyu, a 438-million-year-old, fingernail-sized skull of a jawless fish, and the 419-million-year-old fossil of a completely preserved fish with gill filaments in the first branchial chamber.
“These fossils provided the first anatomical and fossil evidence for a vertebrate spiracle originating from fish gills,” senior author Gai Zhikun, PhD, of the Institute of Vertebrate Paleontology and Paleoanthropology, Beijing, said in a written statement.
In many ways, it seems, we are fish: “Many important structures of human beings can be traced back to our fish ancestors, such as our teeth, jaws, middle ears, etc,” added Zhu Min, PhD, also of the institute.
So, Restless, the next time you hear the soothing sounds of an angry mob storming the Capitol or you chew on a slab, slice, or chunk of mutant, laboratory-produced chicken in your favorite fast-food restaurant, be sure to thank Shuyu.
Can you lend me an ear?
If you thought locusts were only a nuisance, think again. They have their uses. If you take a locust’s ear and put it inside a robot, the robot will be able to hear and receive signals. Who knew?
Researchers from Tel Aviv University in Israel showed the robot’s hearing abilities by giving clap signals that told the robot what to do: One clap means go forward, two claps mean move back. What do you think the robot would do if it heard the clap break from Cha Cha Slide?
“Our task was to replace the robot’s electronic microphone with a dead insect’s ear, use the ear’s ability to detect the electrical signals from the environment, in this case vibrations in the air, and, using a special chip, convert the insect input to that of the robot,” Ben M. Maoz, PhD, said in a statement from the university.
And how does a dead locust ear work in a robot? Well, Dr. Maoz explained: “My laboratory has developed a special device – Ear-on-a-Chip – that allows the ear to be kept alive throughout the experiment by supplying oxygen and food to the organ while allowing the electrical signals to be taken out of the locust’s ear and amplified and transmitted to the robot.”
The research won’t stop at hearing, he said, as the other four senses also will be taken into consideration. This could help us sense dangers in the future, such as earthquakes or diseases. We said it before and we’ll say it again: We’re rooting for you, science!
Your gut microbiome will thank you later
A healthy gut seems like the new catch-all to better overall health these days. Nutrition and diet culture has us drinking kombucha and ginger tea and coffee, but what if we told you that going to happy hour might also help?
In a recent double-blind study published in the Journal of Agricultural and Food Chemistry, 19 men were divided into two groups and asked to drink 11 ounces of alcoholic lager (5.2% by volume) or nonalcoholic lager with dinner for 4 weeks.
Beer? Yes. Beer.
We humans have trillions of microorganisms running rampant through our digestive tracts. When they’re happy, we have a lower chance of developing heart disease and diabetes. You know what else has millions of happy microorganisms from fermentation? Beer. It also has polyphenols that can help the body’s tissues fight cancers, as well as heart disease and inflammation. So beer is looking a little more healthy now, isn’t it?
In the study, the researchers found that both the alcoholic- and nonalcoholic-lager groups had a boost in bacterial diversity in the gut and higher fecal alkaline phosphatase levels, which showed improved intestinal health. They acknowledged, however, that the nonalcoholic route would be safer and healthier for overall health.
So add a lager to the list of gut-healthy foods that you should be consuming. It may give the phrase “beer gut” a whole new meaning.
We’ve lost our minds, but at least we know how fast they’re going
The phrase “quantum consciousness” sounds like something out of a particularly cheesy episode of Star Trek: “Oh no, Captain, the quantum consciousness has invaded our computer, and the only way to drive it out is to reverse the polarity of a focused tachyon beam.”
When it comes to understanding such basic existential issues as the origin of consciousness, however, quantum mechanics wasn’t off the table. The theory of the quantum origin of consciousness dates back to the 1990s (thanks in part to noted physician Roger Penrose), and goes something like this: There are microtubules within neurons in the brain that are small enough and isolated enough from the warm, wet, and chaotic brain environment where quantum effects can briefly come into play. We’re talking miniscule fractions of a second here, but still, long enough for quantum calculations to take place in the form of system wavefunction collapse, courtesy of gravity.
To plunge even deeper into the rabbit hole of quantum mechanics, the reason Schrödinger’s cat doesn’t occur in real life is wavefunction collapse; the more massive a quantum system is, the more likely it is to collapse into one state or another (alive or dead, in the cat’s case). The quantum origin of consciousness, or Orch OR theory, holds that human consciousness arises from electrical oscillations within the neuronal microtubules caused by the computations stemming from the collapse of small quantum systems.
That is an awful lot of overly simplified explanation, especially considering the study that just came out essentially disproved it. Oops. The research, published in Physics of Life Reviews, is pretty simple. The researchers went to a lab deep underground to avoid interference from cosmic rays, and sat around for months, observing a chunk of germanium for signs of spontaneous radiation, attributable to the same sort of wavefunction collapse that is supposedly occurring in our brains. They found nothing out of the ordinary, pretty definitively disproving most of Orch OR theory.
The researchers were unwilling to completely dismiss the idea (this is quantum mechanics, after all, uncertainty kind of goes with the territory), but it does seem like we’ll have to search elsewhere for sources of human consciousness. Personally, we’re big fans of the cymbal-playing monkey.
Missing links: A real fish story
Dear LOTME:
Ear’s a question that’s been keeping me up at night. Is the human middle ear the result of top-secret government experiments involving alien technology, Abraham Lincoln, and the Illuminati?
Restless in Roswell
Dear Restless:
The paleoanthropologic community has been sorting through this mystery for decades, and fossils discovered in China over the past 20 years finally provide a much less conspiratorially satisfying answer.
For some time now, experts in the field have believed that the bones of the human middle ear evolved from the spiracular gill of a fish. The spiracle is a small hole behind each eye that opens to the mouth in some fishes and was used to breathe air in the earliest, most primitive species. But how did we get from spiracle to ear?
The missing links come in the form of the cranial anatomy of Shuyu, a 438-million-year-old, fingernail-sized skull of a jawless fish, and the 419-million-year-old fossil of a completely preserved fish with gill filaments in the first branchial chamber.
“These fossils provided the first anatomical and fossil evidence for a vertebrate spiracle originating from fish gills,” senior author Gai Zhikun, PhD, of the Institute of Vertebrate Paleontology and Paleoanthropology, Beijing, said in a written statement.
In many ways, it seems, we are fish: “Many important structures of human beings can be traced back to our fish ancestors, such as our teeth, jaws, middle ears, etc,” added Zhu Min, PhD, also of the institute.
So, Restless, the next time you hear the soothing sounds of an angry mob storming the Capitol or you chew on a slab, slice, or chunk of mutant, laboratory-produced chicken in your favorite fast-food restaurant, be sure to thank Shuyu.
Can you lend me an ear?
If you thought locusts were only a nuisance, think again. They have their uses. If you take a locust’s ear and put it inside a robot, the robot will be able to hear and receive signals. Who knew?
Researchers from Tel Aviv University in Israel showed the robot’s hearing abilities by giving clap signals that told the robot what to do: One clap means go forward, two claps mean move back. What do you think the robot would do if it heard the clap break from Cha Cha Slide?
“Our task was to replace the robot’s electronic microphone with a dead insect’s ear, use the ear’s ability to detect the electrical signals from the environment, in this case vibrations in the air, and, using a special chip, convert the insect input to that of the robot,” Ben M. Maoz, PhD, said in a statement from the university.
And how does a dead locust ear work in a robot? Well, Dr. Maoz explained: “My laboratory has developed a special device – Ear-on-a-Chip – that allows the ear to be kept alive throughout the experiment by supplying oxygen and food to the organ while allowing the electrical signals to be taken out of the locust’s ear and amplified and transmitted to the robot.”
The research won’t stop at hearing, he said, as the other four senses also will be taken into consideration. This could help us sense dangers in the future, such as earthquakes or diseases. We said it before and we’ll say it again: We’re rooting for you, science!