User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
FDA to recommend limits on heavy metals in baby foods
The Food and Drug Administration has responded to congressional criticism and launched a multiyear plan to reduce the amount of heavy metals such as mercury and arsenic found in baby food.
Called “Closer to Zero,” the FDA plan calls for continued scientific investigation, establishes acceptable levels of heavy metals, sets up a way to monitor manufacturers’ compliance, and sets “action levels.”
“Although the FDA’s testing shows that children are not at an immediate health risk from exposure to toxic elements at the levels found in foods, we are starting the plan’s work immediately, with both short- and long-term goals for achieving continued improvements in reducing levels of toxic elements in these foods over time,” the FDA said.
However, Closer to Zero will only make recommendations on heavy metal levels.
“Although action levels are not binding, we have seen that, over the years, our guidance on action levels and other actions have contributed to significant reductions of toxic elements in food,” an FDA spokeswoman wrote in a statement, according to the Washington Post.
A congressional panel said in February 2021 that major brands of commercial baby food routinely have high levels of toxic heavy metals. The House Oversight Committee said this leaves babies at risk for serious developmental and neurologic problems.
The committee sharply criticized the FDA for not taking action.
“Despite the well-known risks of harm to babies from toxic heavy metals, FDA has not taken adequate steps to decrease their presence in baby foods,” the committee said. “FDA has not issued thresholds for the vast majority of toxic heavy metals in baby foods and does not require warning labels on any baby food products.”
A version of this article first appeared on WebMD.com.
The Food and Drug Administration has responded to congressional criticism and launched a multiyear plan to reduce the amount of heavy metals such as mercury and arsenic found in baby food.
Called “Closer to Zero,” the FDA plan calls for continued scientific investigation, establishes acceptable levels of heavy metals, sets up a way to monitor manufacturers’ compliance, and sets “action levels.”
“Although the FDA’s testing shows that children are not at an immediate health risk from exposure to toxic elements at the levels found in foods, we are starting the plan’s work immediately, with both short- and long-term goals for achieving continued improvements in reducing levels of toxic elements in these foods over time,” the FDA said.
However, Closer to Zero will only make recommendations on heavy metal levels.
“Although action levels are not binding, we have seen that, over the years, our guidance on action levels and other actions have contributed to significant reductions of toxic elements in food,” an FDA spokeswoman wrote in a statement, according to the Washington Post.
A congressional panel said in February 2021 that major brands of commercial baby food routinely have high levels of toxic heavy metals. The House Oversight Committee said this leaves babies at risk for serious developmental and neurologic problems.
The committee sharply criticized the FDA for not taking action.
“Despite the well-known risks of harm to babies from toxic heavy metals, FDA has not taken adequate steps to decrease their presence in baby foods,” the committee said. “FDA has not issued thresholds for the vast majority of toxic heavy metals in baby foods and does not require warning labels on any baby food products.”
A version of this article first appeared on WebMD.com.
The Food and Drug Administration has responded to congressional criticism and launched a multiyear plan to reduce the amount of heavy metals such as mercury and arsenic found in baby food.
Called “Closer to Zero,” the FDA plan calls for continued scientific investigation, establishes acceptable levels of heavy metals, sets up a way to monitor manufacturers’ compliance, and sets “action levels.”
“Although the FDA’s testing shows that children are not at an immediate health risk from exposure to toxic elements at the levels found in foods, we are starting the plan’s work immediately, with both short- and long-term goals for achieving continued improvements in reducing levels of toxic elements in these foods over time,” the FDA said.
However, Closer to Zero will only make recommendations on heavy metal levels.
“Although action levels are not binding, we have seen that, over the years, our guidance on action levels and other actions have contributed to significant reductions of toxic elements in food,” an FDA spokeswoman wrote in a statement, according to the Washington Post.
A congressional panel said in February 2021 that major brands of commercial baby food routinely have high levels of toxic heavy metals. The House Oversight Committee said this leaves babies at risk for serious developmental and neurologic problems.
The committee sharply criticized the FDA for not taking action.
“Despite the well-known risks of harm to babies from toxic heavy metals, FDA has not taken adequate steps to decrease their presence in baby foods,” the committee said. “FDA has not issued thresholds for the vast majority of toxic heavy metals in baby foods and does not require warning labels on any baby food products.”
A version of this article first appeared on WebMD.com.
COVID-19 in children: New cases on the rise again
The number of new COVID-19 cases in children rose for the third time in the last 4 weeks, reaching the highest point since mid-February, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
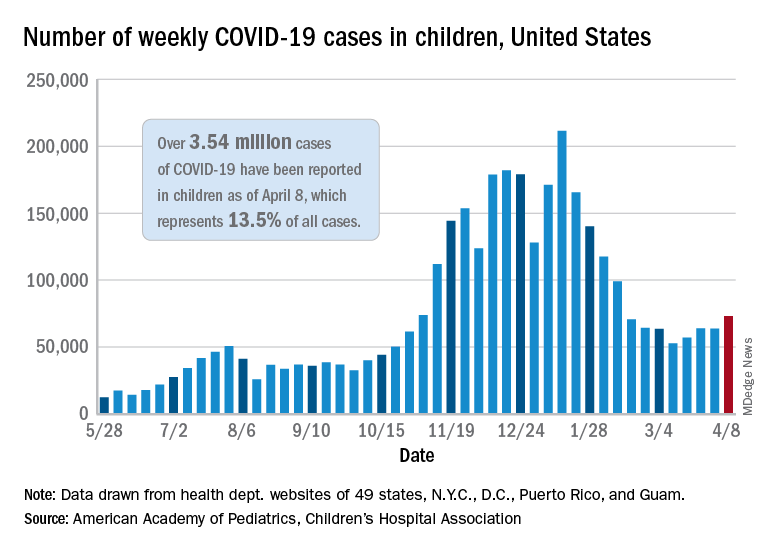
Just over 73,000 cases were reported during the week of April 2-8, up by 14.6% over the previous week. For the latest week, children represented 18.8% of all COVID-19 cases in the United States – also up from the week before and the second-highest proportion seen during the entire pandemic, based on data in the weekly AAP/CHA report.
The 3.54 million children who have been infected with SARS-CoV-2 make up 13.5% of all cases reported in the United States during the pandemic, a figure that climbed again after 2 weeks at 13.4%. The overall rate of infection was just over 4,700 cases per 100,000 children as of April 8, the AAP and CHA said.
State-level data show that Vermont, Michigan, and Maine have been the COVID-19 hotspots over the past 2 weeks. The total number of cases has jumped by almost 19% in Vermont since the week of March 19-25, by 18% in Michigan, and by 12% in Maine, according to the report.
Cumulative data also indicate that the children of Vermont are bearing a greater share of the COVID-19 burden – 21.5% of all cases – than in any other state. North Dakota, meanwhile, has the highest cumulative rate of infection at 9,057 cases per 100,000 children, based on data from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
The number of COVID-19–related deaths in children increased by 8 during the week of April 2-8 and now stands at 292, just 0.06% of all deaths reported in the 43 states (along with New York City, Puerto Rico, and Guam) that provide age distributions for mortality data, the AAP and CHA said.
The number of new COVID-19 cases in children rose for the third time in the last 4 weeks, reaching the highest point since mid-February, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

Just over 73,000 cases were reported during the week of April 2-8, up by 14.6% over the previous week. For the latest week, children represented 18.8% of all COVID-19 cases in the United States – also up from the week before and the second-highest proportion seen during the entire pandemic, based on data in the weekly AAP/CHA report.
The 3.54 million children who have been infected with SARS-CoV-2 make up 13.5% of all cases reported in the United States during the pandemic, a figure that climbed again after 2 weeks at 13.4%. The overall rate of infection was just over 4,700 cases per 100,000 children as of April 8, the AAP and CHA said.
State-level data show that Vermont, Michigan, and Maine have been the COVID-19 hotspots over the past 2 weeks. The total number of cases has jumped by almost 19% in Vermont since the week of March 19-25, by 18% in Michigan, and by 12% in Maine, according to the report.
Cumulative data also indicate that the children of Vermont are bearing a greater share of the COVID-19 burden – 21.5% of all cases – than in any other state. North Dakota, meanwhile, has the highest cumulative rate of infection at 9,057 cases per 100,000 children, based on data from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
The number of COVID-19–related deaths in children increased by 8 during the week of April 2-8 and now stands at 292, just 0.06% of all deaths reported in the 43 states (along with New York City, Puerto Rico, and Guam) that provide age distributions for mortality data, the AAP and CHA said.
The number of new COVID-19 cases in children rose for the third time in the last 4 weeks, reaching the highest point since mid-February, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

Just over 73,000 cases were reported during the week of April 2-8, up by 14.6% over the previous week. For the latest week, children represented 18.8% of all COVID-19 cases in the United States – also up from the week before and the second-highest proportion seen during the entire pandemic, based on data in the weekly AAP/CHA report.
The 3.54 million children who have been infected with SARS-CoV-2 make up 13.5% of all cases reported in the United States during the pandemic, a figure that climbed again after 2 weeks at 13.4%. The overall rate of infection was just over 4,700 cases per 100,000 children as of April 8, the AAP and CHA said.
State-level data show that Vermont, Michigan, and Maine have been the COVID-19 hotspots over the past 2 weeks. The total number of cases has jumped by almost 19% in Vermont since the week of March 19-25, by 18% in Michigan, and by 12% in Maine, according to the report.
Cumulative data also indicate that the children of Vermont are bearing a greater share of the COVID-19 burden – 21.5% of all cases – than in any other state. North Dakota, meanwhile, has the highest cumulative rate of infection at 9,057 cases per 100,000 children, based on data from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
The number of COVID-19–related deaths in children increased by 8 during the week of April 2-8 and now stands at 292, just 0.06% of all deaths reported in the 43 states (along with New York City, Puerto Rico, and Guam) that provide age distributions for mortality data, the AAP and CHA said.
Arthritis drug may curb myocardial damage in acute STEMI
Early use of tocilizumab (Actemra) does not reduce myocardial infarct size but modestly increases myocardial salvage in patients with acute ST-segment elevation MI (STEMI), results of the ASSAIL-MI trial suggest.
“We’re among the first to show that you can actually affect the reperfusion injury through anti-inflammatory treatment – it’s sort of a new attack point for treatments in STEMI,” lead author Kaspar Broch, MD, PhD, Oslo University Hospital Rikshospitalet, said in an interview. “What we do now is reperfuse as soon as we can and then add drugs in order to prevent new events, but we don’t really attack the reperfusion injury that occurs when you perform PCI [percutaneous coronary intervention], which has been shown to actually account for some 50% of the final injury.”
The phase 2, proof-of-concept study was prompted by the team’s earlier work in non-STEMI patients, in which a single dose of the interleukin-6 receptor antagonist cut C-reactive protein (CRP) levels by more than 50% during hospitalization and reduced troponin T release after PCI.
For ASSAIL-MI, Dr. Broch and colleagues randomly assigned 199 patients presenting with acute STEMI within 6 hours of symptom onset to a single intravenous injection of 280 mg tocilizumab or placebo during PCI. Patients, study personnel, and caretakers were blinded to treatment. Data were available for 195 patients for the primary endpoint of myocardial salvage index.
As reported in the Journal of the American College of Cardiology, tocilizumab was associated with a higher adjusted myocardial salvage index on cardiac MRI 3-7 days after PCI than placebo (69.3% vs. 63.6%; P = .04).
The extent of microvascular obstruction was less with tocilizumab (0% vs. 4%; P = .03), as was the area under the curve of CRP during hospitalization (1.9 vs. 8.6 mg/L per hour; P < .001).
The final infarct size at 6 months was 21% lower in the tocilizumab group but the difference did not reach statistical significance (7.2% vs. 9.1% of left ventricular mass; P = .08).
There were no between-group differences in troponin T area under the curve during hospitalization (1,614 vs. 2,357 ng/L per hour; P = .13), N-terminal of the prohormone brain natriuretic peptide concentrations at 6 months (79 vs. 63 ng/L; P = .25), or baseline-adjusted left ventricular end-diastolic volume at 6 months (157 vs. 160 mL; P = .54).
Subgroup analyses suggested the positive effect of tocilizumab on myocardial salvage index is limited to patients presenting at least 3 hours after symptom onset versus 3 hours or less (P = .034), with a trend for greater benefit among men versus women (P = .053).
Dr. Broch noted that the absolute effect of tocilizumab on myocardial necrosis was smaller than anticipated when the trial was designed, which may explain the lack of significant reduction in infarct size.
“We were aiming for patients with larger infarctions than we actually ended up with, which is partly due to the strict inclusion criteria and the fact that, with modern treatments, patients don’t end up with large myocardial infarctions,” he said. “But if they had been larger, I think that 20% absolute reduction would have meant a lot in terms of clinical events.”
The study also used a very modest dose of tocilizumab, compared with that used for inflammatory diseases, to minimize a potential negative effect on myocardial healing, for instance, myocardial ruptures, Dr. Broch said. “I’m not sure whether you gain anything by giving a larger dose.”
Serious adverse events were similar in the tocilizumab and placebo groups (19 vs. 15; P = .57). There were no myocardial ruptures, and no patient died or developed heart failure. LDL cholesterol, triglycerides, and liver enzymes increased in the tocilizumab group but were similar at 3 and 6 months.
“IL-6 is a central cytokine involved in all stages of plaque growth, progression, and rupture,” Paul Ridker, MD, MPH, of the Brigham and Women’s Hospital in Boston, and a long-standing investigator in inflammation and atherothrombosis, said in an interview. “These preliminary data in STEMI, like the authors’ prior data in non-STEMI, are consistent with the idea that inhibiting IL-6 could have clinical benefit, a concept that will be taken into a major cardiovascular outcomes trial later this year.”
The cardiovascular outcomes trial, known as ZEUS, will test the novel IL-6 inhibitor ziltivekimab among more than 6,000 very-high-risk atherosclerosis patients who have moderate to severe chronic kidney disease and high sensitivity CRP greater than 2 mg/L, he noted.
Moving beyond IL-1b blockade as done in CANTOS to direct downstream inhibition of IL-6 represents a “logical next scientific step” in the development of anti-inflammatory therapies for acute ischemia and chronic atherosclerosis, Dr. Ridker, who led the CANTOS trial, noted in an accompanying editorial.
“Preventive cardiologists, however, need not wait until outcome trials are complete to use this evolving biological knowledge to their patient’s advantage,” he wrote. “As recently confirmed in the pages of the Journal, exercise, smoking cessation, and a healthy diet reduce both C-reactive protein and IL-6, and clearly have lifelong benefits. Our immediate task is thus to incorporate inflammation inhibition through lifestyle management into our daily practice.”
The study was supported by the South-Eastern Norway Regional Health Authority, Central Norway Regional Health Authority, and Roche, which provided the medicinal products and an unrestricted grant. Dr. Broch has disclosed no relevant financial relationships. Dr. Ridker has received investigator-initiated research grant support from Kowa, Novartis, Amarin, Pfizer, and the National Heart, Lung, and Blood Institute; and has served as a consultant to Novartis, Janssen, Agepha, Flame, Civi Biopharma, Inflazome, Corvidia, Novo Nordisk, SOCAR, IQVIA, and AstraZeneca.
A version of this article first appeared on Medscape.com.
Early use of tocilizumab (Actemra) does not reduce myocardial infarct size but modestly increases myocardial salvage in patients with acute ST-segment elevation MI (STEMI), results of the ASSAIL-MI trial suggest.
“We’re among the first to show that you can actually affect the reperfusion injury through anti-inflammatory treatment – it’s sort of a new attack point for treatments in STEMI,” lead author Kaspar Broch, MD, PhD, Oslo University Hospital Rikshospitalet, said in an interview. “What we do now is reperfuse as soon as we can and then add drugs in order to prevent new events, but we don’t really attack the reperfusion injury that occurs when you perform PCI [percutaneous coronary intervention], which has been shown to actually account for some 50% of the final injury.”
The phase 2, proof-of-concept study was prompted by the team’s earlier work in non-STEMI patients, in which a single dose of the interleukin-6 receptor antagonist cut C-reactive protein (CRP) levels by more than 50% during hospitalization and reduced troponin T release after PCI.
For ASSAIL-MI, Dr. Broch and colleagues randomly assigned 199 patients presenting with acute STEMI within 6 hours of symptom onset to a single intravenous injection of 280 mg tocilizumab or placebo during PCI. Patients, study personnel, and caretakers were blinded to treatment. Data were available for 195 patients for the primary endpoint of myocardial salvage index.
As reported in the Journal of the American College of Cardiology, tocilizumab was associated with a higher adjusted myocardial salvage index on cardiac MRI 3-7 days after PCI than placebo (69.3% vs. 63.6%; P = .04).
The extent of microvascular obstruction was less with tocilizumab (0% vs. 4%; P = .03), as was the area under the curve of CRP during hospitalization (1.9 vs. 8.6 mg/L per hour; P < .001).
The final infarct size at 6 months was 21% lower in the tocilizumab group but the difference did not reach statistical significance (7.2% vs. 9.1% of left ventricular mass; P = .08).
There were no between-group differences in troponin T area under the curve during hospitalization (1,614 vs. 2,357 ng/L per hour; P = .13), N-terminal of the prohormone brain natriuretic peptide concentrations at 6 months (79 vs. 63 ng/L; P = .25), or baseline-adjusted left ventricular end-diastolic volume at 6 months (157 vs. 160 mL; P = .54).
Subgroup analyses suggested the positive effect of tocilizumab on myocardial salvage index is limited to patients presenting at least 3 hours after symptom onset versus 3 hours or less (P = .034), with a trend for greater benefit among men versus women (P = .053).
Dr. Broch noted that the absolute effect of tocilizumab on myocardial necrosis was smaller than anticipated when the trial was designed, which may explain the lack of significant reduction in infarct size.
“We were aiming for patients with larger infarctions than we actually ended up with, which is partly due to the strict inclusion criteria and the fact that, with modern treatments, patients don’t end up with large myocardial infarctions,” he said. “But if they had been larger, I think that 20% absolute reduction would have meant a lot in terms of clinical events.”
The study also used a very modest dose of tocilizumab, compared with that used for inflammatory diseases, to minimize a potential negative effect on myocardial healing, for instance, myocardial ruptures, Dr. Broch said. “I’m not sure whether you gain anything by giving a larger dose.”
Serious adverse events were similar in the tocilizumab and placebo groups (19 vs. 15; P = .57). There were no myocardial ruptures, and no patient died or developed heart failure. LDL cholesterol, triglycerides, and liver enzymes increased in the tocilizumab group but were similar at 3 and 6 months.
“IL-6 is a central cytokine involved in all stages of plaque growth, progression, and rupture,” Paul Ridker, MD, MPH, of the Brigham and Women’s Hospital in Boston, and a long-standing investigator in inflammation and atherothrombosis, said in an interview. “These preliminary data in STEMI, like the authors’ prior data in non-STEMI, are consistent with the idea that inhibiting IL-6 could have clinical benefit, a concept that will be taken into a major cardiovascular outcomes trial later this year.”
The cardiovascular outcomes trial, known as ZEUS, will test the novel IL-6 inhibitor ziltivekimab among more than 6,000 very-high-risk atherosclerosis patients who have moderate to severe chronic kidney disease and high sensitivity CRP greater than 2 mg/L, he noted.
Moving beyond IL-1b blockade as done in CANTOS to direct downstream inhibition of IL-6 represents a “logical next scientific step” in the development of anti-inflammatory therapies for acute ischemia and chronic atherosclerosis, Dr. Ridker, who led the CANTOS trial, noted in an accompanying editorial.
“Preventive cardiologists, however, need not wait until outcome trials are complete to use this evolving biological knowledge to their patient’s advantage,” he wrote. “As recently confirmed in the pages of the Journal, exercise, smoking cessation, and a healthy diet reduce both C-reactive protein and IL-6, and clearly have lifelong benefits. Our immediate task is thus to incorporate inflammation inhibition through lifestyle management into our daily practice.”
The study was supported by the South-Eastern Norway Regional Health Authority, Central Norway Regional Health Authority, and Roche, which provided the medicinal products and an unrestricted grant. Dr. Broch has disclosed no relevant financial relationships. Dr. Ridker has received investigator-initiated research grant support from Kowa, Novartis, Amarin, Pfizer, and the National Heart, Lung, and Blood Institute; and has served as a consultant to Novartis, Janssen, Agepha, Flame, Civi Biopharma, Inflazome, Corvidia, Novo Nordisk, SOCAR, IQVIA, and AstraZeneca.
A version of this article first appeared on Medscape.com.
Early use of tocilizumab (Actemra) does not reduce myocardial infarct size but modestly increases myocardial salvage in patients with acute ST-segment elevation MI (STEMI), results of the ASSAIL-MI trial suggest.
“We’re among the first to show that you can actually affect the reperfusion injury through anti-inflammatory treatment – it’s sort of a new attack point for treatments in STEMI,” lead author Kaspar Broch, MD, PhD, Oslo University Hospital Rikshospitalet, said in an interview. “What we do now is reperfuse as soon as we can and then add drugs in order to prevent new events, but we don’t really attack the reperfusion injury that occurs when you perform PCI [percutaneous coronary intervention], which has been shown to actually account for some 50% of the final injury.”
The phase 2, proof-of-concept study was prompted by the team’s earlier work in non-STEMI patients, in which a single dose of the interleukin-6 receptor antagonist cut C-reactive protein (CRP) levels by more than 50% during hospitalization and reduced troponin T release after PCI.
For ASSAIL-MI, Dr. Broch and colleagues randomly assigned 199 patients presenting with acute STEMI within 6 hours of symptom onset to a single intravenous injection of 280 mg tocilizumab or placebo during PCI. Patients, study personnel, and caretakers were blinded to treatment. Data were available for 195 patients for the primary endpoint of myocardial salvage index.
As reported in the Journal of the American College of Cardiology, tocilizumab was associated with a higher adjusted myocardial salvage index on cardiac MRI 3-7 days after PCI than placebo (69.3% vs. 63.6%; P = .04).
The extent of microvascular obstruction was less with tocilizumab (0% vs. 4%; P = .03), as was the area under the curve of CRP during hospitalization (1.9 vs. 8.6 mg/L per hour; P < .001).
The final infarct size at 6 months was 21% lower in the tocilizumab group but the difference did not reach statistical significance (7.2% vs. 9.1% of left ventricular mass; P = .08).
There were no between-group differences in troponin T area under the curve during hospitalization (1,614 vs. 2,357 ng/L per hour; P = .13), N-terminal of the prohormone brain natriuretic peptide concentrations at 6 months (79 vs. 63 ng/L; P = .25), or baseline-adjusted left ventricular end-diastolic volume at 6 months (157 vs. 160 mL; P = .54).
Subgroup analyses suggested the positive effect of tocilizumab on myocardial salvage index is limited to patients presenting at least 3 hours after symptom onset versus 3 hours or less (P = .034), with a trend for greater benefit among men versus women (P = .053).
Dr. Broch noted that the absolute effect of tocilizumab on myocardial necrosis was smaller than anticipated when the trial was designed, which may explain the lack of significant reduction in infarct size.
“We were aiming for patients with larger infarctions than we actually ended up with, which is partly due to the strict inclusion criteria and the fact that, with modern treatments, patients don’t end up with large myocardial infarctions,” he said. “But if they had been larger, I think that 20% absolute reduction would have meant a lot in terms of clinical events.”
The study also used a very modest dose of tocilizumab, compared with that used for inflammatory diseases, to minimize a potential negative effect on myocardial healing, for instance, myocardial ruptures, Dr. Broch said. “I’m not sure whether you gain anything by giving a larger dose.”
Serious adverse events were similar in the tocilizumab and placebo groups (19 vs. 15; P = .57). There were no myocardial ruptures, and no patient died or developed heart failure. LDL cholesterol, triglycerides, and liver enzymes increased in the tocilizumab group but were similar at 3 and 6 months.
“IL-6 is a central cytokine involved in all stages of plaque growth, progression, and rupture,” Paul Ridker, MD, MPH, of the Brigham and Women’s Hospital in Boston, and a long-standing investigator in inflammation and atherothrombosis, said in an interview. “These preliminary data in STEMI, like the authors’ prior data in non-STEMI, are consistent with the idea that inhibiting IL-6 could have clinical benefit, a concept that will be taken into a major cardiovascular outcomes trial later this year.”
The cardiovascular outcomes trial, known as ZEUS, will test the novel IL-6 inhibitor ziltivekimab among more than 6,000 very-high-risk atherosclerosis patients who have moderate to severe chronic kidney disease and high sensitivity CRP greater than 2 mg/L, he noted.
Moving beyond IL-1b blockade as done in CANTOS to direct downstream inhibition of IL-6 represents a “logical next scientific step” in the development of anti-inflammatory therapies for acute ischemia and chronic atherosclerosis, Dr. Ridker, who led the CANTOS trial, noted in an accompanying editorial.
“Preventive cardiologists, however, need not wait until outcome trials are complete to use this evolving biological knowledge to their patient’s advantage,” he wrote. “As recently confirmed in the pages of the Journal, exercise, smoking cessation, and a healthy diet reduce both C-reactive protein and IL-6, and clearly have lifelong benefits. Our immediate task is thus to incorporate inflammation inhibition through lifestyle management into our daily practice.”
The study was supported by the South-Eastern Norway Regional Health Authority, Central Norway Regional Health Authority, and Roche, which provided the medicinal products and an unrestricted grant. Dr. Broch has disclosed no relevant financial relationships. Dr. Ridker has received investigator-initiated research grant support from Kowa, Novartis, Amarin, Pfizer, and the National Heart, Lung, and Blood Institute; and has served as a consultant to Novartis, Janssen, Agepha, Flame, Civi Biopharma, Inflazome, Corvidia, Novo Nordisk, SOCAR, IQVIA, and AstraZeneca.
A version of this article first appeared on Medscape.com.
The obesity risk everyone forgets
Clinicians in pediatrics have noticed a troubling pattern emerge during the pandemic, something that is darkly referred to as “the COVID 19,” or the 19 or more pounds that many of our patients have gained in the past year. This phenomenon has underscored many maxims in pediatric weight management: Mainly that frequent snacking, decreased physical activity, and less parental supervision lead to increased weight gain. But could we be missing another lesson this trend is teaching us? What about the relationship between catastrophe and childhood obesity?
Beyond the increased weight gain with lockdowns, I have observed other evidence in my own practice that childhood trauma or adverse experiences increase obesity. Our electronic medical record system gives an alert when a chart with sensitive information is accessed. One example might be if the patient had been seen at a clinic for children who have been abused. I am heartbroken at how often this happens. Academically, I understand the dire statistics about the incidence of child abuse, but the frequency at which I see this pattern is jarring.
Over the years, one striking correlation became clear among my patient population: Children with obesity were more likely to have been seen in the child abuse clinic than normal-weight peers.
I am far from the only one to have observed this relationship. Television shows focusing on severe obesity, such as “My 600-Pound Life,” often show trauma as both a cause and effect of severe obesity. This theme also became apparent on the show “The Biggest Loser,” which highlighted the difficulty of achieving and maintaining substantial weight loss. If even Hollywood has noticed this association, shouldn’t we be much farther ahead?
Pathways to obesity
Adverse childhood experiences (ACE) encompass various causes of child trauma, including abuse or neglect; poverty; household or neighborhood violence; and death, illness, or incarceration of a parent. A pivotal report in 1998 formalized the suspicion that many of us could plainly see: People who suffered ACE have higher incidence of heart disease, COPD, liver disease, incarceration, and drug abuse. For those with six or more ACE, life expectancy averaged 20 years less than those who had none. More recently, a meta-analysis found an odds ratio of 1.46 for adult obesity with known history of childhood trauma.
As a pediatric endocrinologist living in the poorest state of the country, I have clearly observed the correlation between childhood obesity and poverty. While prior generations may have associated child poverty with malnutrition and starvation, we are seeing in modern times that obesity has become a disease of lack. Calorie-dense and processed foods tend to be less expensive, more shelf-stable, and more accessible to people living in both urban and rural food deserts.
I am also a foster mother and have received extensive training in parenting children who have lived through trauma and neglect. For children who have endured food scarcity and deprivation, hoarding food and overeating are expected responses.
But the pathways to abnormal weight gain are myriad and expand beyond binge eating or numbing with food. ACE are particularly troubling because they affect developing brains and the neuroendocrine system; they alter epigenetics and cause heritable changes. Structural brain differences have been evident in the frontopolar cortex, which is linked to centers in the hypothalamus that control appetite. And increased stress raises cortisol release, increases insulin resistance, and alters satiety.
Shifting our approach to treatment
The significant cost of ACE is enormous and affects us all. Health professionals in pediatrics must understand these connections to effectively counsel children and their families dealing with obesity. Handing someone a diet plan and lecturing them about weight loss is never effective, but this common tactic is especially cruel if we do not assess for and address underlying pain. Obviously, blame and shame are ineffective motivators for lifestyle change in any circumstance, but these tactics may be especially harmful in the light of childhood trauma.
Screening for ACE is important in every aspect of pediatric care. The presence of obesity, however, should remind us to be more sensitive to the possibility of causative trauma. Clinicians for adults are not off the hook either. Fully 60% of adults suffered ACE and are dealing with the aftermath.
To improve health outcomes across the board, we must screen for trauma and become educated on trauma-informed care. Perhaps the most important first referral for a child suffering ACE and obesity is to a trained counselor or a social worker. Shepherding children through trauma will be more effective for attaining healthy weight than any remedy I can prescribe as an endocrinologist. Furthermore, this is our necessary role as healers. More than ever, we need to approach chronic diseases, including obesity, with the utmost compassion.
A version of this article first appeared on Medscape.com.
Clinicians in pediatrics have noticed a troubling pattern emerge during the pandemic, something that is darkly referred to as “the COVID 19,” or the 19 or more pounds that many of our patients have gained in the past year. This phenomenon has underscored many maxims in pediatric weight management: Mainly that frequent snacking, decreased physical activity, and less parental supervision lead to increased weight gain. But could we be missing another lesson this trend is teaching us? What about the relationship between catastrophe and childhood obesity?
Beyond the increased weight gain with lockdowns, I have observed other evidence in my own practice that childhood trauma or adverse experiences increase obesity. Our electronic medical record system gives an alert when a chart with sensitive information is accessed. One example might be if the patient had been seen at a clinic for children who have been abused. I am heartbroken at how often this happens. Academically, I understand the dire statistics about the incidence of child abuse, but the frequency at which I see this pattern is jarring.
Over the years, one striking correlation became clear among my patient population: Children with obesity were more likely to have been seen in the child abuse clinic than normal-weight peers.
I am far from the only one to have observed this relationship. Television shows focusing on severe obesity, such as “My 600-Pound Life,” often show trauma as both a cause and effect of severe obesity. This theme also became apparent on the show “The Biggest Loser,” which highlighted the difficulty of achieving and maintaining substantial weight loss. If even Hollywood has noticed this association, shouldn’t we be much farther ahead?
Pathways to obesity
Adverse childhood experiences (ACE) encompass various causes of child trauma, including abuse or neglect; poverty; household or neighborhood violence; and death, illness, or incarceration of a parent. A pivotal report in 1998 formalized the suspicion that many of us could plainly see: People who suffered ACE have higher incidence of heart disease, COPD, liver disease, incarceration, and drug abuse. For those with six or more ACE, life expectancy averaged 20 years less than those who had none. More recently, a meta-analysis found an odds ratio of 1.46 for adult obesity with known history of childhood trauma.
As a pediatric endocrinologist living in the poorest state of the country, I have clearly observed the correlation between childhood obesity and poverty. While prior generations may have associated child poverty with malnutrition and starvation, we are seeing in modern times that obesity has become a disease of lack. Calorie-dense and processed foods tend to be less expensive, more shelf-stable, and more accessible to people living in both urban and rural food deserts.
I am also a foster mother and have received extensive training in parenting children who have lived through trauma and neglect. For children who have endured food scarcity and deprivation, hoarding food and overeating are expected responses.
But the pathways to abnormal weight gain are myriad and expand beyond binge eating or numbing with food. ACE are particularly troubling because they affect developing brains and the neuroendocrine system; they alter epigenetics and cause heritable changes. Structural brain differences have been evident in the frontopolar cortex, which is linked to centers in the hypothalamus that control appetite. And increased stress raises cortisol release, increases insulin resistance, and alters satiety.
Shifting our approach to treatment
The significant cost of ACE is enormous and affects us all. Health professionals in pediatrics must understand these connections to effectively counsel children and their families dealing with obesity. Handing someone a diet plan and lecturing them about weight loss is never effective, but this common tactic is especially cruel if we do not assess for and address underlying pain. Obviously, blame and shame are ineffective motivators for lifestyle change in any circumstance, but these tactics may be especially harmful in the light of childhood trauma.
Screening for ACE is important in every aspect of pediatric care. The presence of obesity, however, should remind us to be more sensitive to the possibility of causative trauma. Clinicians for adults are not off the hook either. Fully 60% of adults suffered ACE and are dealing with the aftermath.
To improve health outcomes across the board, we must screen for trauma and become educated on trauma-informed care. Perhaps the most important first referral for a child suffering ACE and obesity is to a trained counselor or a social worker. Shepherding children through trauma will be more effective for attaining healthy weight than any remedy I can prescribe as an endocrinologist. Furthermore, this is our necessary role as healers. More than ever, we need to approach chronic diseases, including obesity, with the utmost compassion.
A version of this article first appeared on Medscape.com.
Clinicians in pediatrics have noticed a troubling pattern emerge during the pandemic, something that is darkly referred to as “the COVID 19,” or the 19 or more pounds that many of our patients have gained in the past year. This phenomenon has underscored many maxims in pediatric weight management: Mainly that frequent snacking, decreased physical activity, and less parental supervision lead to increased weight gain. But could we be missing another lesson this trend is teaching us? What about the relationship between catastrophe and childhood obesity?
Beyond the increased weight gain with lockdowns, I have observed other evidence in my own practice that childhood trauma or adverse experiences increase obesity. Our electronic medical record system gives an alert when a chart with sensitive information is accessed. One example might be if the patient had been seen at a clinic for children who have been abused. I am heartbroken at how often this happens. Academically, I understand the dire statistics about the incidence of child abuse, but the frequency at which I see this pattern is jarring.
Over the years, one striking correlation became clear among my patient population: Children with obesity were more likely to have been seen in the child abuse clinic than normal-weight peers.
I am far from the only one to have observed this relationship. Television shows focusing on severe obesity, such as “My 600-Pound Life,” often show trauma as both a cause and effect of severe obesity. This theme also became apparent on the show “The Biggest Loser,” which highlighted the difficulty of achieving and maintaining substantial weight loss. If even Hollywood has noticed this association, shouldn’t we be much farther ahead?
Pathways to obesity
Adverse childhood experiences (ACE) encompass various causes of child trauma, including abuse or neglect; poverty; household or neighborhood violence; and death, illness, or incarceration of a parent. A pivotal report in 1998 formalized the suspicion that many of us could plainly see: People who suffered ACE have higher incidence of heart disease, COPD, liver disease, incarceration, and drug abuse. For those with six or more ACE, life expectancy averaged 20 years less than those who had none. More recently, a meta-analysis found an odds ratio of 1.46 for adult obesity with known history of childhood trauma.
As a pediatric endocrinologist living in the poorest state of the country, I have clearly observed the correlation between childhood obesity and poverty. While prior generations may have associated child poverty with malnutrition and starvation, we are seeing in modern times that obesity has become a disease of lack. Calorie-dense and processed foods tend to be less expensive, more shelf-stable, and more accessible to people living in both urban and rural food deserts.
I am also a foster mother and have received extensive training in parenting children who have lived through trauma and neglect. For children who have endured food scarcity and deprivation, hoarding food and overeating are expected responses.
But the pathways to abnormal weight gain are myriad and expand beyond binge eating or numbing with food. ACE are particularly troubling because they affect developing brains and the neuroendocrine system; they alter epigenetics and cause heritable changes. Structural brain differences have been evident in the frontopolar cortex, which is linked to centers in the hypothalamus that control appetite. And increased stress raises cortisol release, increases insulin resistance, and alters satiety.
Shifting our approach to treatment
The significant cost of ACE is enormous and affects us all. Health professionals in pediatrics must understand these connections to effectively counsel children and their families dealing with obesity. Handing someone a diet plan and lecturing them about weight loss is never effective, but this common tactic is especially cruel if we do not assess for and address underlying pain. Obviously, blame and shame are ineffective motivators for lifestyle change in any circumstance, but these tactics may be especially harmful in the light of childhood trauma.
Screening for ACE is important in every aspect of pediatric care. The presence of obesity, however, should remind us to be more sensitive to the possibility of causative trauma. Clinicians for adults are not off the hook either. Fully 60% of adults suffered ACE and are dealing with the aftermath.
To improve health outcomes across the board, we must screen for trauma and become educated on trauma-informed care. Perhaps the most important first referral for a child suffering ACE and obesity is to a trained counselor or a social worker. Shepherding children through trauma will be more effective for attaining healthy weight than any remedy I can prescribe as an endocrinologist. Furthermore, this is our necessary role as healers. More than ever, we need to approach chronic diseases, including obesity, with the utmost compassion.
A version of this article first appeared on Medscape.com.
FDA lifts in-person dispensing requirement for mifepristone
The Food and Drug Administration has lifted in-person dispensing requirements for mifepristone when used for medical termination of early pregnancy.
In an April 12, 2021, letter to the American College of Obstetricians and Gynecologists and the Society of Maternal-Fetal Medicine, acting commissioner of food and drugs Janet Woodcock stated that the FDA would exercise discretion to permit the dispensing of mifepristone through the mail when done by or under the supervision of a certified prescriber; or through a mail-order pharmacy under the supervision of a certified prescriber.
The decision follows a trial period of suspension of the in-person dispensing requirement in response to safety concerns for patients as well as providers associated with in-person clinic visits during the COVID-19 pandemic. The Center for Drug Evaluation and Research reviewed safety and clinical outcomes data on mifepristone use when prescriptions were handled by mail or mail-order pharmacy and found that "the small number of adverse events reported to FDA during the COVID-19 public health emergency [PHE] provide no indication that any program deviation or noncompliance with the mifepristone [Risk Evaluation and Mitigation Strategy] program contributed to the reported adverse events," according to the letter. The analysis covers Mifeprex and the approved generic, mifepristone tablets, both 200-mg doses.
As long as other mifepristone REMS criteria are met, the FDA will continue to permit mail and mail-order prescriptions, according to the letter.
"By halting enforcement of the in-person dispensing requirement during the COVID-19 pandemic, the FDA is recognizing and responding to the available evidence - which has clearly and definitively demonstrated that the in-person dispensing requirement for mifepristone is unnecessary and restrictive," Maureen G. Phipps, MD, MPH, CEO of ACOG, said in a statement in response to the FDA decision.
ACOG petitioned the FDA to suspend the in-person requirement to reduce the risk of transmission in the wake of the COVID-19 pandemic, given safety concerns and the potential impact on hard-hit communities, particularly communities of color, Dr. Phipps emphasized. Data from a review period with a suspension of the in-person requirement yielded no additional safety concerns with mifepristone use, and contributed to the FDA decision to lift the requirement.
"Thanks to the FDA's intent to exercise discretion in enforcing the in-person dispensing requirement, those in need of an abortion or miscarriage management will be able to do so safety and effectively by acquiring mifepristone though the mail - just as they would any other medication with a similarly strong safety profile," said Dr. Phipps. "We are pleased to see mifepristone regulated on the basis of the scientific evidence during the pandemic, rather than political bias against comprehensive reproductive health care, and we look forward to working with policy makers to ensure this principle governs postpandemic care."
CDER is communicating the decision to all approved application holders subject to the mifepristone REMS program, according to the letter.
[email protected]
The Food and Drug Administration has lifted in-person dispensing requirements for mifepristone when used for medical termination of early pregnancy.
In an April 12, 2021, letter to the American College of Obstetricians and Gynecologists and the Society of Maternal-Fetal Medicine, acting commissioner of food and drugs Janet Woodcock stated that the FDA would exercise discretion to permit the dispensing of mifepristone through the mail when done by or under the supervision of a certified prescriber; or through a mail-order pharmacy under the supervision of a certified prescriber.
The decision follows a trial period of suspension of the in-person dispensing requirement in response to safety concerns for patients as well as providers associated with in-person clinic visits during the COVID-19 pandemic. The Center for Drug Evaluation and Research reviewed safety and clinical outcomes data on mifepristone use when prescriptions were handled by mail or mail-order pharmacy and found that "the small number of adverse events reported to FDA during the COVID-19 public health emergency [PHE] provide no indication that any program deviation or noncompliance with the mifepristone [Risk Evaluation and Mitigation Strategy] program contributed to the reported adverse events," according to the letter. The analysis covers Mifeprex and the approved generic, mifepristone tablets, both 200-mg doses.
As long as other mifepristone REMS criteria are met, the FDA will continue to permit mail and mail-order prescriptions, according to the letter.
"By halting enforcement of the in-person dispensing requirement during the COVID-19 pandemic, the FDA is recognizing and responding to the available evidence - which has clearly and definitively demonstrated that the in-person dispensing requirement for mifepristone is unnecessary and restrictive," Maureen G. Phipps, MD, MPH, CEO of ACOG, said in a statement in response to the FDA decision.
ACOG petitioned the FDA to suspend the in-person requirement to reduce the risk of transmission in the wake of the COVID-19 pandemic, given safety concerns and the potential impact on hard-hit communities, particularly communities of color, Dr. Phipps emphasized. Data from a review period with a suspension of the in-person requirement yielded no additional safety concerns with mifepristone use, and contributed to the FDA decision to lift the requirement.
"Thanks to the FDA's intent to exercise discretion in enforcing the in-person dispensing requirement, those in need of an abortion or miscarriage management will be able to do so safety and effectively by acquiring mifepristone though the mail - just as they would any other medication with a similarly strong safety profile," said Dr. Phipps. "We are pleased to see mifepristone regulated on the basis of the scientific evidence during the pandemic, rather than political bias against comprehensive reproductive health care, and we look forward to working with policy makers to ensure this principle governs postpandemic care."
CDER is communicating the decision to all approved application holders subject to the mifepristone REMS program, according to the letter.
[email protected]
The Food and Drug Administration has lifted in-person dispensing requirements for mifepristone when used for medical termination of early pregnancy.
In an April 12, 2021, letter to the American College of Obstetricians and Gynecologists and the Society of Maternal-Fetal Medicine, acting commissioner of food and drugs Janet Woodcock stated that the FDA would exercise discretion to permit the dispensing of mifepristone through the mail when done by or under the supervision of a certified prescriber; or through a mail-order pharmacy under the supervision of a certified prescriber.
The decision follows a trial period of suspension of the in-person dispensing requirement in response to safety concerns for patients as well as providers associated with in-person clinic visits during the COVID-19 pandemic. The Center for Drug Evaluation and Research reviewed safety and clinical outcomes data on mifepristone use when prescriptions were handled by mail or mail-order pharmacy and found that "the small number of adverse events reported to FDA during the COVID-19 public health emergency [PHE] provide no indication that any program deviation or noncompliance with the mifepristone [Risk Evaluation and Mitigation Strategy] program contributed to the reported adverse events," according to the letter. The analysis covers Mifeprex and the approved generic, mifepristone tablets, both 200-mg doses.
As long as other mifepristone REMS criteria are met, the FDA will continue to permit mail and mail-order prescriptions, according to the letter.
"By halting enforcement of the in-person dispensing requirement during the COVID-19 pandemic, the FDA is recognizing and responding to the available evidence - which has clearly and definitively demonstrated that the in-person dispensing requirement for mifepristone is unnecessary and restrictive," Maureen G. Phipps, MD, MPH, CEO of ACOG, said in a statement in response to the FDA decision.
ACOG petitioned the FDA to suspend the in-person requirement to reduce the risk of transmission in the wake of the COVID-19 pandemic, given safety concerns and the potential impact on hard-hit communities, particularly communities of color, Dr. Phipps emphasized. Data from a review period with a suspension of the in-person requirement yielded no additional safety concerns with mifepristone use, and contributed to the FDA decision to lift the requirement.
"Thanks to the FDA's intent to exercise discretion in enforcing the in-person dispensing requirement, those in need of an abortion or miscarriage management will be able to do so safety and effectively by acquiring mifepristone though the mail - just as they would any other medication with a similarly strong safety profile," said Dr. Phipps. "We are pleased to see mifepristone regulated on the basis of the scientific evidence during the pandemic, rather than political bias against comprehensive reproductive health care, and we look forward to working with policy makers to ensure this principle governs postpandemic care."
CDER is communicating the decision to all approved application holders subject to the mifepristone REMS program, according to the letter.
[email protected]
Medtronic recall of almost 240,000 ICDs is class I, FDA says
The Food and Drug Administration has declared Medtronic’s recall of seven models of defibrillating cardiac rhythm devices, caused by a risk for premature battery depletion, as class I, which implies a potential risk for serious injury or death. A total of 444 complaints, but no deaths, have been reported in association with the 239,171 affected devices, the agency said in a statement on April 12, 2021.
Physicians were notified of the company’s recall in early February. It covered implantable cardioverter defibrillator (ICD) and cardiac resynchronization therapy–defibrillator (CRT-D) models Evera, Viva, Brava, Claria, Amplia, Compia, and Visia distributed from Aug. 31, 2012 to May 9, 2018.
The devices could be subject to “an unexpected and rapid decrease in battery life” because of a possible short circuit that could lead to a device-replacement alert “earlier than expected.” Some devices may experience full battery depletion “within as little as 1 day” after such an alert.
“If the user does not respond to the first warning, the device may stop functioning. The likelihood that this issue will occur is constant after approximately 3 years after device use,” the announcement said.
Medtronic recommends device replacement no more than 1 week after such an early warning for patients who are not pacing dependent or who have them for primary prevention, but right away for pacing-dependent patients.
A version of this article first appeared on Medscape.com
The Food and Drug Administration has declared Medtronic’s recall of seven models of defibrillating cardiac rhythm devices, caused by a risk for premature battery depletion, as class I, which implies a potential risk for serious injury or death. A total of 444 complaints, but no deaths, have been reported in association with the 239,171 affected devices, the agency said in a statement on April 12, 2021.
Physicians were notified of the company’s recall in early February. It covered implantable cardioverter defibrillator (ICD) and cardiac resynchronization therapy–defibrillator (CRT-D) models Evera, Viva, Brava, Claria, Amplia, Compia, and Visia distributed from Aug. 31, 2012 to May 9, 2018.
The devices could be subject to “an unexpected and rapid decrease in battery life” because of a possible short circuit that could lead to a device-replacement alert “earlier than expected.” Some devices may experience full battery depletion “within as little as 1 day” after such an alert.
“If the user does not respond to the first warning, the device may stop functioning. The likelihood that this issue will occur is constant after approximately 3 years after device use,” the announcement said.
Medtronic recommends device replacement no more than 1 week after such an early warning for patients who are not pacing dependent or who have them for primary prevention, but right away for pacing-dependent patients.
A version of this article first appeared on Medscape.com
The Food and Drug Administration has declared Medtronic’s recall of seven models of defibrillating cardiac rhythm devices, caused by a risk for premature battery depletion, as class I, which implies a potential risk for serious injury or death. A total of 444 complaints, but no deaths, have been reported in association with the 239,171 affected devices, the agency said in a statement on April 12, 2021.
Physicians were notified of the company’s recall in early February. It covered implantable cardioverter defibrillator (ICD) and cardiac resynchronization therapy–defibrillator (CRT-D) models Evera, Viva, Brava, Claria, Amplia, Compia, and Visia distributed from Aug. 31, 2012 to May 9, 2018.
The devices could be subject to “an unexpected and rapid decrease in battery life” because of a possible short circuit that could lead to a device-replacement alert “earlier than expected.” Some devices may experience full battery depletion “within as little as 1 day” after such an alert.
“If the user does not respond to the first warning, the device may stop functioning. The likelihood that this issue will occur is constant after approximately 3 years after device use,” the announcement said.
Medtronic recommends device replacement no more than 1 week after such an early warning for patients who are not pacing dependent or who have them for primary prevention, but right away for pacing-dependent patients.
A version of this article first appeared on Medscape.com
Next winter may be rough: Models predict ‘considerable surge’ of COVID
It’s likely the United States will see another surge of COVID-19 this winter, warned Christopher Murray, MD, director of the Institute for Health Metrics and Evaluation (IHME) at the University of Washington in Seattle.
Speaking at the national conference of State of Reform on April 8, Dr. Murray cited the seasonality of the SARS-CoV-2 virus, which wanes in the summer and waxes in the winter. The “optimistic forecast” of IHME, which has modeled the course of the pandemic for the past 13 months, is that daily deaths will rise a bit in the next month, then decline from May through August, he said.
“Summer should be fairly quiet in terms of COVID, if vaccinations rise and people don’t stop wearing masks,” Dr. Murray said.
But he added that “a considerable surge will occur over next winter,” because the new variants are more transmissible, and people will likely relax social distancing and mask wearing. The IHME predicts that the percentage of Americans who usually don masks will decline from 73% today to 21% by Aug. 1.
With a rapid decline in mask use and a rise in mobility, there will still be more than 1,000 deaths each day by July 1, Dr. Murray said. In a forecast released the day after Dr. Murray spoke, the IHME predicted that by Aug. 1, there will be a total of 618,523 U.S. deaths from COVID-19. Deaths could be as high as 696,651 if mobility among the vaccinated returns to prepandemic levels, the institute forecasts.
Based on cell phone data, Dr. Murray said, the amount of mobility in the United States has already risen to the level of March 2020, when the pandemic was just getting underway.
Decreased infections
If there’s one piece of good news in the latest IHME report, it’s that the estimated number of people infected (including those not tested) will drop from 111,581 today to a projected 17,502 on Aug. 1. But in a worst-case scenario, with sharply higher mobility among vaccinated people, the case count on that date would only fall to 73,842.
The SARS-CoV-2 variants are another factor of concern. Dr. Murray distinguished between variants like the one first identified in the U.K. (B.1.1.7) and other “escape variants.”
B.1.1.7, which is now the dominant strain in the United States, increases transmission but doesn’t necessarily escape the immune system or vaccines, he explained.
In contrast, if someone is infected with a variant such as the South African or the Brazilian mutations, he said, a previous COVID-19 infection might not protect the person, and vaccines are less effective against those variants.
Cross-variant immunity may range from 0% to 60% for escape variants, based on the slim amount of data now available, Dr. Murray said. In his view, these variants will be the long-term driver of the pandemic in the United States, while the United Kingdom variant is the short-term driver.
The latest data, he said, show that the Pfizer/BioNTech and Moderna vaccines are 75% effective against the escape variants, with lower efficacy for other vaccines. But booster shots may still be required to protect people against some variants.
Human factors
Human behavior will also help determine the course of the pandemic, he noted. Vaccine hesitancy, for example, is still high in the United States.
By the end of May, he predicted, about 180 million people will have received about two doses of vaccine. After that, he said, “vaccination will flatline due to lack of demand.” The two unknowns are how much campaigns to promote vaccination will increase vaccine confidence, and when children will be vaccinated.
In the United States, he said, 69% of adults have been vaccinated or want to get a shot. But that percentage has dropped 5 points since February, and vaccine confidence varies by state.
Dr. Murray emphasized that the winter surge he predicts can be blocked if people change their behaviors. These include a rise in vaccine confidence to 80% and continued mask wearing by most people.
However, if vaccine confidence and mask wearing decline, state governments continue to drop social distancing rules, and the uptake of boosters is low, the winter surge could be more serious, he said.
Double surge
Murray also raised the possibility of a double surge of COVID-19 and influenza this winter. Widely expected last winter, this double surge never materialized here or elsewhere, partly because of mask wearing. But Dr. Murray said it could happen this year: History shows that the flu tends to be stronger in years after weak outbreaks.
He advised hospitals to prepare now for whatever might come later this year. Public health authorities, he said, should speed up vaccination, monitor variants closely with additional sequencing, and try to modify behavior in high-risk groups.
Asked to explain the recent surge of COVID-19 cases in Michigan, Dr. Murray attributed it partly to the spread of the B.1.1.7 (U.K.) variant. But he noted that the U.K. variant has expanded even more widely in some other states that haven’t had an explosive surge like Michigan’s.
Moreover, he noted, Michigan doesn’t have low mask use or high mobility. So the upward spiral of COVID-19 infections there is very concerning, he said.
In regard to the role of children as reservoirs of the virus, Dr. Murray pointed out that views on this have changed around the world. For a while, people thought kids didn’t spread COVID-19 very much. That view shifted when U.K. data showed that child transmission of the B.1.1.7 variant increased by half to 9% of contacts in comparison with the original virus strain.
Dutch data, similarly, showed schools contributing to the latest outbreaks, and some European nations have closed schools. In the United States, the trend is to open them.
A version of this article first appeared on Medscape.com.
It’s likely the United States will see another surge of COVID-19 this winter, warned Christopher Murray, MD, director of the Institute for Health Metrics and Evaluation (IHME) at the University of Washington in Seattle.
Speaking at the national conference of State of Reform on April 8, Dr. Murray cited the seasonality of the SARS-CoV-2 virus, which wanes in the summer and waxes in the winter. The “optimistic forecast” of IHME, which has modeled the course of the pandemic for the past 13 months, is that daily deaths will rise a bit in the next month, then decline from May through August, he said.
“Summer should be fairly quiet in terms of COVID, if vaccinations rise and people don’t stop wearing masks,” Dr. Murray said.
But he added that “a considerable surge will occur over next winter,” because the new variants are more transmissible, and people will likely relax social distancing and mask wearing. The IHME predicts that the percentage of Americans who usually don masks will decline from 73% today to 21% by Aug. 1.
With a rapid decline in mask use and a rise in mobility, there will still be more than 1,000 deaths each day by July 1, Dr. Murray said. In a forecast released the day after Dr. Murray spoke, the IHME predicted that by Aug. 1, there will be a total of 618,523 U.S. deaths from COVID-19. Deaths could be as high as 696,651 if mobility among the vaccinated returns to prepandemic levels, the institute forecasts.
Based on cell phone data, Dr. Murray said, the amount of mobility in the United States has already risen to the level of March 2020, when the pandemic was just getting underway.
Decreased infections
If there’s one piece of good news in the latest IHME report, it’s that the estimated number of people infected (including those not tested) will drop from 111,581 today to a projected 17,502 on Aug. 1. But in a worst-case scenario, with sharply higher mobility among vaccinated people, the case count on that date would only fall to 73,842.
The SARS-CoV-2 variants are another factor of concern. Dr. Murray distinguished between variants like the one first identified in the U.K. (B.1.1.7) and other “escape variants.”
B.1.1.7, which is now the dominant strain in the United States, increases transmission but doesn’t necessarily escape the immune system or vaccines, he explained.
In contrast, if someone is infected with a variant such as the South African or the Brazilian mutations, he said, a previous COVID-19 infection might not protect the person, and vaccines are less effective against those variants.
Cross-variant immunity may range from 0% to 60% for escape variants, based on the slim amount of data now available, Dr. Murray said. In his view, these variants will be the long-term driver of the pandemic in the United States, while the United Kingdom variant is the short-term driver.
The latest data, he said, show that the Pfizer/BioNTech and Moderna vaccines are 75% effective against the escape variants, with lower efficacy for other vaccines. But booster shots may still be required to protect people against some variants.
Human factors
Human behavior will also help determine the course of the pandemic, he noted. Vaccine hesitancy, for example, is still high in the United States.
By the end of May, he predicted, about 180 million people will have received about two doses of vaccine. After that, he said, “vaccination will flatline due to lack of demand.” The two unknowns are how much campaigns to promote vaccination will increase vaccine confidence, and when children will be vaccinated.
In the United States, he said, 69% of adults have been vaccinated or want to get a shot. But that percentage has dropped 5 points since February, and vaccine confidence varies by state.
Dr. Murray emphasized that the winter surge he predicts can be blocked if people change their behaviors. These include a rise in vaccine confidence to 80% and continued mask wearing by most people.
However, if vaccine confidence and mask wearing decline, state governments continue to drop social distancing rules, and the uptake of boosters is low, the winter surge could be more serious, he said.
Double surge
Murray also raised the possibility of a double surge of COVID-19 and influenza this winter. Widely expected last winter, this double surge never materialized here or elsewhere, partly because of mask wearing. But Dr. Murray said it could happen this year: History shows that the flu tends to be stronger in years after weak outbreaks.
He advised hospitals to prepare now for whatever might come later this year. Public health authorities, he said, should speed up vaccination, monitor variants closely with additional sequencing, and try to modify behavior in high-risk groups.
Asked to explain the recent surge of COVID-19 cases in Michigan, Dr. Murray attributed it partly to the spread of the B.1.1.7 (U.K.) variant. But he noted that the U.K. variant has expanded even more widely in some other states that haven’t had an explosive surge like Michigan’s.
Moreover, he noted, Michigan doesn’t have low mask use or high mobility. So the upward spiral of COVID-19 infections there is very concerning, he said.
In regard to the role of children as reservoirs of the virus, Dr. Murray pointed out that views on this have changed around the world. For a while, people thought kids didn’t spread COVID-19 very much. That view shifted when U.K. data showed that child transmission of the B.1.1.7 variant increased by half to 9% of contacts in comparison with the original virus strain.
Dutch data, similarly, showed schools contributing to the latest outbreaks, and some European nations have closed schools. In the United States, the trend is to open them.
A version of this article first appeared on Medscape.com.
It’s likely the United States will see another surge of COVID-19 this winter, warned Christopher Murray, MD, director of the Institute for Health Metrics and Evaluation (IHME) at the University of Washington in Seattle.
Speaking at the national conference of State of Reform on April 8, Dr. Murray cited the seasonality of the SARS-CoV-2 virus, which wanes in the summer and waxes in the winter. The “optimistic forecast” of IHME, which has modeled the course of the pandemic for the past 13 months, is that daily deaths will rise a bit in the next month, then decline from May through August, he said.
“Summer should be fairly quiet in terms of COVID, if vaccinations rise and people don’t stop wearing masks,” Dr. Murray said.
But he added that “a considerable surge will occur over next winter,” because the new variants are more transmissible, and people will likely relax social distancing and mask wearing. The IHME predicts that the percentage of Americans who usually don masks will decline from 73% today to 21% by Aug. 1.
With a rapid decline in mask use and a rise in mobility, there will still be more than 1,000 deaths each day by July 1, Dr. Murray said. In a forecast released the day after Dr. Murray spoke, the IHME predicted that by Aug. 1, there will be a total of 618,523 U.S. deaths from COVID-19. Deaths could be as high as 696,651 if mobility among the vaccinated returns to prepandemic levels, the institute forecasts.
Based on cell phone data, Dr. Murray said, the amount of mobility in the United States has already risen to the level of March 2020, when the pandemic was just getting underway.
Decreased infections
If there’s one piece of good news in the latest IHME report, it’s that the estimated number of people infected (including those not tested) will drop from 111,581 today to a projected 17,502 on Aug. 1. But in a worst-case scenario, with sharply higher mobility among vaccinated people, the case count on that date would only fall to 73,842.
The SARS-CoV-2 variants are another factor of concern. Dr. Murray distinguished between variants like the one first identified in the U.K. (B.1.1.7) and other “escape variants.”
B.1.1.7, which is now the dominant strain in the United States, increases transmission but doesn’t necessarily escape the immune system or vaccines, he explained.
In contrast, if someone is infected with a variant such as the South African or the Brazilian mutations, he said, a previous COVID-19 infection might not protect the person, and vaccines are less effective against those variants.
Cross-variant immunity may range from 0% to 60% for escape variants, based on the slim amount of data now available, Dr. Murray said. In his view, these variants will be the long-term driver of the pandemic in the United States, while the United Kingdom variant is the short-term driver.
The latest data, he said, show that the Pfizer/BioNTech and Moderna vaccines are 75% effective against the escape variants, with lower efficacy for other vaccines. But booster shots may still be required to protect people against some variants.
Human factors
Human behavior will also help determine the course of the pandemic, he noted. Vaccine hesitancy, for example, is still high in the United States.
By the end of May, he predicted, about 180 million people will have received about two doses of vaccine. After that, he said, “vaccination will flatline due to lack of demand.” The two unknowns are how much campaigns to promote vaccination will increase vaccine confidence, and when children will be vaccinated.
In the United States, he said, 69% of adults have been vaccinated or want to get a shot. But that percentage has dropped 5 points since February, and vaccine confidence varies by state.
Dr. Murray emphasized that the winter surge he predicts can be blocked if people change their behaviors. These include a rise in vaccine confidence to 80% and continued mask wearing by most people.
However, if vaccine confidence and mask wearing decline, state governments continue to drop social distancing rules, and the uptake of boosters is low, the winter surge could be more serious, he said.
Double surge
Murray also raised the possibility of a double surge of COVID-19 and influenza this winter. Widely expected last winter, this double surge never materialized here or elsewhere, partly because of mask wearing. But Dr. Murray said it could happen this year: History shows that the flu tends to be stronger in years after weak outbreaks.
He advised hospitals to prepare now for whatever might come later this year. Public health authorities, he said, should speed up vaccination, monitor variants closely with additional sequencing, and try to modify behavior in high-risk groups.
Asked to explain the recent surge of COVID-19 cases in Michigan, Dr. Murray attributed it partly to the spread of the B.1.1.7 (U.K.) variant. But he noted that the U.K. variant has expanded even more widely in some other states that haven’t had an explosive surge like Michigan’s.
Moreover, he noted, Michigan doesn’t have low mask use or high mobility. So the upward spiral of COVID-19 infections there is very concerning, he said.
In regard to the role of children as reservoirs of the virus, Dr. Murray pointed out that views on this have changed around the world. For a while, people thought kids didn’t spread COVID-19 very much. That view shifted when U.K. data showed that child transmission of the B.1.1.7 variant increased by half to 9% of contacts in comparison with the original virus strain.
Dutch data, similarly, showed schools contributing to the latest outbreaks, and some European nations have closed schools. In the United States, the trend is to open them.
A version of this article first appeared on Medscape.com.
Remote cardio visits expand access for underserved during COVID
Remote cardiology clinic visits during COVID-19 were used more often by certain traditionally underserved patient groups, but were also associated with less frequent testing and prescribing, new research shows.
“The COVID-19 pandemic has led to an unprecedented shift in ambulatory cardiovascular care from in-person to remote visits,” lead author Neal Yuan, MD, a cardiology fellow at the Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles, said in an interview.
Their findings were published online April 5 in JAMA Network Open.
“We wanted to explore whether the transition to remote visits was associated with disparities in how patients accessed care, and also how this transition affected diagnostic test ordering and medication prescribing,” Dr. Yuan said.
The researchers used electronic health records data for all ambulatory cardiology visits at an urban, multisite health system in Los Angeles County during two periods: April 1 to Dec. 31, 2019, the pre-COVID era; and April 1 to Dec. 31, 2020, the COVID era.
The investigators compared patient characteristics and frequencies of medication ordering and cardiology-specific testing across four visit types: pre-COVID in person, used as reference; COVID-era in person; COVID-era video; and COVID-era telephone.
The study looked at 176,781 ambulatory cardiology visits. Of these visits, 87,182 were conducted in person in the pre-COVID period; 74,498 were conducted in person in the COVID era; 4,720 were COVID-era video visits; and 10,381 were COVID-era telephone visits.
In the study cohort, 79,572 patients (45.0%) were female, 127,080 patients (71.9%) were non-Hispanic White, and the mean age was 68.1 years (standard deviation, 17.0).
Patients accessing COVID-era remote visits were more likely to be Asian, Black, or Hispanic, to have private insurance, and to have cardiovascular comorbidities, such as hypertension and heart failure.
Also, patients whose visits were conducted by video were significantly younger than patients whose visits were conducted in person or by telephone (P < .001).
In addition, the study found that clinicians ordered fewer diagnostic tests, such as electrocardiograms and echocardiograms, and were less likely to order any medication, in the pre-COVID era than during the COVID era.
“If you don’t have a patient in front of you, it’s much more difficult to get a physical exam or obtain reliable vital signs,” said Dr. Yuan. Communication can sometimes be difficult, often because of technical issues, like a bad connection. “You might be more reticent to get testing or to prescribe medications if you don’t feel confident knowing what the patient’s vital signs are.”
In addition, he added, “a lot of medications used in the cardiology setting require monitoring patients’ kidney function and electrolytes, and if you can’t do that reliably, you might be more cautious about prescribing those types of medications.”
An eye-opening study
Cardiologist Nieca Goldberg, MD, medical director of the New York University Langone womens’ heart program and spokesperson for the American Heart Association, recounted her experience with telemedicine at the height of the pandemic in New York, when everything, including medical outpatient offices, had to close.
“We were experienced with telemedicine because we had started a virtual urgent care program well ahead of the pandemic,” she said. “We started using that to screen people with potential COVID symptoms so that they wouldn’t have to come into the hospital, the medical center, or to the offices and expose people. We learned that it was great to have the telemedicine option from the infectious disease standpoint, and I did visits like that for my own patient population.”
An equally if not more important finding from the study is the fact that telemedicine increased access to care among traditionally underserved demographics, she said.
“This is eye-opening, that you can actually improve access to care by doing telemedicine visits. It was really important to see that telemedicine has added benefit to the way we can see people in the health care system.”
Telemedicine visits had a positive impact at a time when people were isolated at home, Dr. Goldberg said.
“It was a way for them to connect with their doctor and in some ways it was more personal,” she added. “I actually got to meet some of my patients’ family members. It was like making a remote house call.”
Stable cardiology patients can take their blood pressure at home, weigh themselves, and take their own pulse to give an excellent set of vital signs that will indicate how they are doing, said Dr. Goldberg.
“During a remote visit, we can talk to the patient and notice whether or not they are short of breath or coughing, but we can’t listen to their heart or do an EKG or any of the traditional cardiac testing. Still, for someone who is not having symptoms and is able to reliably monitor their blood pressure and weight, a remote visit is sufficient to give you a good sense of how that patient is doing,” she said. “We can talk to them about their medications, any potential side effects, and we can use their blood pressure information to adjust their medications.”
Many patients are becoming more savvy about using tech gadgets and devices to monitor their health.
“Some of my patients were using Apple watches and the Kardia app to address their heart rate. Many had purchased inexpensive pulse oximeters to check their oxygen during the pandemic, and that also reads the pulse,” Dr. Goldberg said.
In-person visits were reserved for symptomatic cardiac patients, she explained.
“Initially during the pandemic, we did mostly telemedicine visits and we organized the office so that each cardiologist would come in 1 day a week to take care of symptomatic cardiac patients. In that way, we were able to socially distance – they provided us with [personal protective equipment]; at NYU there was no problem with that – and nobody waited in the waiting room. To this day, office issues are more efficient and people are not waiting in the waiting room,” she added. “Telemedicine improves access to health care in populations where such access is limited.”
Dr. Yuan’s research is supported by a grant from the National Institutes of Health. Dr. Goldberg reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Remote cardiology clinic visits during COVID-19 were used more often by certain traditionally underserved patient groups, but were also associated with less frequent testing and prescribing, new research shows.
“The COVID-19 pandemic has led to an unprecedented shift in ambulatory cardiovascular care from in-person to remote visits,” lead author Neal Yuan, MD, a cardiology fellow at the Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles, said in an interview.
Their findings were published online April 5 in JAMA Network Open.
“We wanted to explore whether the transition to remote visits was associated with disparities in how patients accessed care, and also how this transition affected diagnostic test ordering and medication prescribing,” Dr. Yuan said.
The researchers used electronic health records data for all ambulatory cardiology visits at an urban, multisite health system in Los Angeles County during two periods: April 1 to Dec. 31, 2019, the pre-COVID era; and April 1 to Dec. 31, 2020, the COVID era.
The investigators compared patient characteristics and frequencies of medication ordering and cardiology-specific testing across four visit types: pre-COVID in person, used as reference; COVID-era in person; COVID-era video; and COVID-era telephone.
The study looked at 176,781 ambulatory cardiology visits. Of these visits, 87,182 were conducted in person in the pre-COVID period; 74,498 were conducted in person in the COVID era; 4,720 were COVID-era video visits; and 10,381 were COVID-era telephone visits.
In the study cohort, 79,572 patients (45.0%) were female, 127,080 patients (71.9%) were non-Hispanic White, and the mean age was 68.1 years (standard deviation, 17.0).
Patients accessing COVID-era remote visits were more likely to be Asian, Black, or Hispanic, to have private insurance, and to have cardiovascular comorbidities, such as hypertension and heart failure.
Also, patients whose visits were conducted by video were significantly younger than patients whose visits were conducted in person or by telephone (P < .001).
In addition, the study found that clinicians ordered fewer diagnostic tests, such as electrocardiograms and echocardiograms, and were less likely to order any medication, in the pre-COVID era than during the COVID era.
“If you don’t have a patient in front of you, it’s much more difficult to get a physical exam or obtain reliable vital signs,” said Dr. Yuan. Communication can sometimes be difficult, often because of technical issues, like a bad connection. “You might be more reticent to get testing or to prescribe medications if you don’t feel confident knowing what the patient’s vital signs are.”
In addition, he added, “a lot of medications used in the cardiology setting require monitoring patients’ kidney function and electrolytes, and if you can’t do that reliably, you might be more cautious about prescribing those types of medications.”
An eye-opening study
Cardiologist Nieca Goldberg, MD, medical director of the New York University Langone womens’ heart program and spokesperson for the American Heart Association, recounted her experience with telemedicine at the height of the pandemic in New York, when everything, including medical outpatient offices, had to close.
“We were experienced with telemedicine because we had started a virtual urgent care program well ahead of the pandemic,” she said. “We started using that to screen people with potential COVID symptoms so that they wouldn’t have to come into the hospital, the medical center, or to the offices and expose people. We learned that it was great to have the telemedicine option from the infectious disease standpoint, and I did visits like that for my own patient population.”
An equally if not more important finding from the study is the fact that telemedicine increased access to care among traditionally underserved demographics, she said.
“This is eye-opening, that you can actually improve access to care by doing telemedicine visits. It was really important to see that telemedicine has added benefit to the way we can see people in the health care system.”
Telemedicine visits had a positive impact at a time when people were isolated at home, Dr. Goldberg said.
“It was a way for them to connect with their doctor and in some ways it was more personal,” she added. “I actually got to meet some of my patients’ family members. It was like making a remote house call.”
Stable cardiology patients can take their blood pressure at home, weigh themselves, and take their own pulse to give an excellent set of vital signs that will indicate how they are doing, said Dr. Goldberg.
“During a remote visit, we can talk to the patient and notice whether or not they are short of breath or coughing, but we can’t listen to their heart or do an EKG or any of the traditional cardiac testing. Still, for someone who is not having symptoms and is able to reliably monitor their blood pressure and weight, a remote visit is sufficient to give you a good sense of how that patient is doing,” she said. “We can talk to them about their medications, any potential side effects, and we can use their blood pressure information to adjust their medications.”
Many patients are becoming more savvy about using tech gadgets and devices to monitor their health.
“Some of my patients were using Apple watches and the Kardia app to address their heart rate. Many had purchased inexpensive pulse oximeters to check their oxygen during the pandemic, and that also reads the pulse,” Dr. Goldberg said.
In-person visits were reserved for symptomatic cardiac patients, she explained.
“Initially during the pandemic, we did mostly telemedicine visits and we organized the office so that each cardiologist would come in 1 day a week to take care of symptomatic cardiac patients. In that way, we were able to socially distance – they provided us with [personal protective equipment]; at NYU there was no problem with that – and nobody waited in the waiting room. To this day, office issues are more efficient and people are not waiting in the waiting room,” she added. “Telemedicine improves access to health care in populations where such access is limited.”
Dr. Yuan’s research is supported by a grant from the National Institutes of Health. Dr. Goldberg reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Remote cardiology clinic visits during COVID-19 were used more often by certain traditionally underserved patient groups, but were also associated with less frequent testing and prescribing, new research shows.
“The COVID-19 pandemic has led to an unprecedented shift in ambulatory cardiovascular care from in-person to remote visits,” lead author Neal Yuan, MD, a cardiology fellow at the Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles, said in an interview.
Their findings were published online April 5 in JAMA Network Open.
“We wanted to explore whether the transition to remote visits was associated with disparities in how patients accessed care, and also how this transition affected diagnostic test ordering and medication prescribing,” Dr. Yuan said.
The researchers used electronic health records data for all ambulatory cardiology visits at an urban, multisite health system in Los Angeles County during two periods: April 1 to Dec. 31, 2019, the pre-COVID era; and April 1 to Dec. 31, 2020, the COVID era.
The investigators compared patient characteristics and frequencies of medication ordering and cardiology-specific testing across four visit types: pre-COVID in person, used as reference; COVID-era in person; COVID-era video; and COVID-era telephone.
The study looked at 176,781 ambulatory cardiology visits. Of these visits, 87,182 were conducted in person in the pre-COVID period; 74,498 were conducted in person in the COVID era; 4,720 were COVID-era video visits; and 10,381 were COVID-era telephone visits.
In the study cohort, 79,572 patients (45.0%) were female, 127,080 patients (71.9%) were non-Hispanic White, and the mean age was 68.1 years (standard deviation, 17.0).
Patients accessing COVID-era remote visits were more likely to be Asian, Black, or Hispanic, to have private insurance, and to have cardiovascular comorbidities, such as hypertension and heart failure.
Also, patients whose visits were conducted by video were significantly younger than patients whose visits were conducted in person or by telephone (P < .001).
In addition, the study found that clinicians ordered fewer diagnostic tests, such as electrocardiograms and echocardiograms, and were less likely to order any medication, in the pre-COVID era than during the COVID era.
“If you don’t have a patient in front of you, it’s much more difficult to get a physical exam or obtain reliable vital signs,” said Dr. Yuan. Communication can sometimes be difficult, often because of technical issues, like a bad connection. “You might be more reticent to get testing or to prescribe medications if you don’t feel confident knowing what the patient’s vital signs are.”
In addition, he added, “a lot of medications used in the cardiology setting require monitoring patients’ kidney function and electrolytes, and if you can’t do that reliably, you might be more cautious about prescribing those types of medications.”
An eye-opening study
Cardiologist Nieca Goldberg, MD, medical director of the New York University Langone womens’ heart program and spokesperson for the American Heart Association, recounted her experience with telemedicine at the height of the pandemic in New York, when everything, including medical outpatient offices, had to close.
“We were experienced with telemedicine because we had started a virtual urgent care program well ahead of the pandemic,” she said. “We started using that to screen people with potential COVID symptoms so that they wouldn’t have to come into the hospital, the medical center, or to the offices and expose people. We learned that it was great to have the telemedicine option from the infectious disease standpoint, and I did visits like that for my own patient population.”
An equally if not more important finding from the study is the fact that telemedicine increased access to care among traditionally underserved demographics, she said.
“This is eye-opening, that you can actually improve access to care by doing telemedicine visits. It was really important to see that telemedicine has added benefit to the way we can see people in the health care system.”
Telemedicine visits had a positive impact at a time when people were isolated at home, Dr. Goldberg said.
“It was a way for them to connect with their doctor and in some ways it was more personal,” she added. “I actually got to meet some of my patients’ family members. It was like making a remote house call.”
Stable cardiology patients can take their blood pressure at home, weigh themselves, and take their own pulse to give an excellent set of vital signs that will indicate how they are doing, said Dr. Goldberg.
“During a remote visit, we can talk to the patient and notice whether or not they are short of breath or coughing, but we can’t listen to their heart or do an EKG or any of the traditional cardiac testing. Still, for someone who is not having symptoms and is able to reliably monitor their blood pressure and weight, a remote visit is sufficient to give you a good sense of how that patient is doing,” she said. “We can talk to them about their medications, any potential side effects, and we can use their blood pressure information to adjust their medications.”
Many patients are becoming more savvy about using tech gadgets and devices to monitor their health.
“Some of my patients were using Apple watches and the Kardia app to address their heart rate. Many had purchased inexpensive pulse oximeters to check their oxygen during the pandemic, and that also reads the pulse,” Dr. Goldberg said.
In-person visits were reserved for symptomatic cardiac patients, she explained.
“Initially during the pandemic, we did mostly telemedicine visits and we organized the office so that each cardiologist would come in 1 day a week to take care of symptomatic cardiac patients. In that way, we were able to socially distance – they provided us with [personal protective equipment]; at NYU there was no problem with that – and nobody waited in the waiting room. To this day, office issues are more efficient and people are not waiting in the waiting room,” she added. “Telemedicine improves access to health care in populations where such access is limited.”
Dr. Yuan’s research is supported by a grant from the National Institutes of Health. Dr. Goldberg reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FDA, CDC urge pause of J&J COVID vaccine
The Food and Drug Administration and Centers for Disease Control and Prevention on April 13 recommended that use of the Johnson & Johnson COVID-19 vaccine be paused after reports of blood clots in patients receiving the shot, the agencies have announced.
In a statement, FDA said 6.8 million doses of the J&J vaccine have been administered and the agency is investigating six reported cases of a rare and severe blood clot occurring in patients who received the vaccine.
The pause is intended to give time to alert the public to this "very rare" condition, experts said during a joint CDC-FDA media briefing April 13.
"It was clear to us that we needed to alert the public," Janet Woodcock, MD, acting FDA commissioner, said. The move also will allow "time for the healthcare community to learn what they need to know about how to diagnose, treat and report" any additional cases.
The CDC will convene a meeting of the Advisory Committee on Immunization Practices on April 14 to review the cases.
"I know the information today will be very concerning to Americans who have already received the Johnson & Johnson vaccine," said Anne Schuchat, MD, principal deputy director at the CDC.
"For people who got the vaccine more than one month ago, the risk is very low at this time," she added. "For people who recently got the vaccine, in the last couple of weeks, look for symptoms."
Headache, leg pain, abdominal pain, and shortness of breath were among the reported symptoms. All six cases arose within 6 to 13 days of receipt of the Johnson & Johnson vaccine.
Traditional treatment dangerous
Importantly, treatment for traditional blood clots, such as the drug heparin, should not be used for these clots. "The issue here with these types of blood clots is that if one administers the standard treatment we give for blood clots, one can cause tremendous harm or it can be fatal," said Peter Marks, MD, director of the FDA Center for Biologics Evaluation and Research.
If health care providers see people with these symptoms along with a low platelet count or blood clots, they should ask about any recent vaccinations, Dr. Marks added.
Headache is a common side effect of COVID-19 vaccination, Dr. Marks said, but it typically happens within a day or two. In contrast, the headaches associated with these blood clots come 1 to 2 weeks later and were very severe.
Not all of the six women involved in the events had a pre-existing condition or risk factor, Dr. Schuchat said.
Severe but 'extremely rare'
To put the numbers in context, the six reported events occurred among millions of people who received the Johnson & Johnson vaccine to date.
"There have been six reports of a severe stroke-like illness due to low platelet count and more than six million doses of the Johnson & Johnson vaccine have been administered so far," Dr. Schuchat said.
"I would like to stress these events are extremely rare," Dr. Woodcock said, "but we take all reports of adverse events after vaccination very seriously."
The company response
Johnson & Johnson in a statement said, "We are aware of an extremely rare disorder involving people with blood clots in combination with low platelets in a small number of individuals who have received our COVID-19 vaccine. The United States Centers for Disease Control (CDC) and Food and Drug Administration (FDA) are reviewing data involving six reported U.S. cases out of more than 6.8 million doses administered. Out of an abundance of caution, the CDC and FDA have recommended a pause in the use of our vaccine."
The company said they are also reviewing these cases with European regulators and "we have made the decision to proactively delay the rollout of our vaccine in Europe."
Overall vaccinations continuing apace
"This announcement will not have a significant impact on our vaccination plan. Johnson & Johnson vaccine makes up less than 5% of the recorded shots in arms in the United States to date," Jeff Zients, White House COVID-19 Response Coordinator, said in a statement.
"Based on actions taken by the president earlier this year, the United States has secured enough Pfizer and Moderna doses for 300 million Americans. We are working now with our state and federal partners to get anyone scheduled for a J&J vaccine quickly rescheduled for a Pfizer or Moderna vaccine," he added.
The likely duration of the pause remains unclear.
"I know this has been a long and difficult pandemic, and people are tired of the steps they have to take," Dr. Schuchat said. "Steps taken today make sure the health care system is ready to diagnose, treat and report [any additional cases] and the public has the information necessary to stay safe."
A version of this article first appeared on WebMD.com.
This article was updated 4/13/21.
The Food and Drug Administration and Centers for Disease Control and Prevention on April 13 recommended that use of the Johnson & Johnson COVID-19 vaccine be paused after reports of blood clots in patients receiving the shot, the agencies have announced.
In a statement, FDA said 6.8 million doses of the J&J vaccine have been administered and the agency is investigating six reported cases of a rare and severe blood clot occurring in patients who received the vaccine.
The pause is intended to give time to alert the public to this "very rare" condition, experts said during a joint CDC-FDA media briefing April 13.
"It was clear to us that we needed to alert the public," Janet Woodcock, MD, acting FDA commissioner, said. The move also will allow "time for the healthcare community to learn what they need to know about how to diagnose, treat and report" any additional cases.
The CDC will convene a meeting of the Advisory Committee on Immunization Practices on April 14 to review the cases.
"I know the information today will be very concerning to Americans who have already received the Johnson & Johnson vaccine," said Anne Schuchat, MD, principal deputy director at the CDC.
"For people who got the vaccine more than one month ago, the risk is very low at this time," she added. "For people who recently got the vaccine, in the last couple of weeks, look for symptoms."
Headache, leg pain, abdominal pain, and shortness of breath were among the reported symptoms. All six cases arose within 6 to 13 days of receipt of the Johnson & Johnson vaccine.
Traditional treatment dangerous
Importantly, treatment for traditional blood clots, such as the drug heparin, should not be used for these clots. "The issue here with these types of blood clots is that if one administers the standard treatment we give for blood clots, one can cause tremendous harm or it can be fatal," said Peter Marks, MD, director of the FDA Center for Biologics Evaluation and Research.
If health care providers see people with these symptoms along with a low platelet count or blood clots, they should ask about any recent vaccinations, Dr. Marks added.
Headache is a common side effect of COVID-19 vaccination, Dr. Marks said, but it typically happens within a day or two. In contrast, the headaches associated with these blood clots come 1 to 2 weeks later and were very severe.
Not all of the six women involved in the events had a pre-existing condition or risk factor, Dr. Schuchat said.
Severe but 'extremely rare'
To put the numbers in context, the six reported events occurred among millions of people who received the Johnson & Johnson vaccine to date.
"There have been six reports of a severe stroke-like illness due to low platelet count and more than six million doses of the Johnson & Johnson vaccine have been administered so far," Dr. Schuchat said.
"I would like to stress these events are extremely rare," Dr. Woodcock said, "but we take all reports of adverse events after vaccination very seriously."
The company response
Johnson & Johnson in a statement said, "We are aware of an extremely rare disorder involving people with blood clots in combination with low platelets in a small number of individuals who have received our COVID-19 vaccine. The United States Centers for Disease Control (CDC) and Food and Drug Administration (FDA) are reviewing data involving six reported U.S. cases out of more than 6.8 million doses administered. Out of an abundance of caution, the CDC and FDA have recommended a pause in the use of our vaccine."
The company said they are also reviewing these cases with European regulators and "we have made the decision to proactively delay the rollout of our vaccine in Europe."
Overall vaccinations continuing apace
"This announcement will not have a significant impact on our vaccination plan. Johnson & Johnson vaccine makes up less than 5% of the recorded shots in arms in the United States to date," Jeff Zients, White House COVID-19 Response Coordinator, said in a statement.
"Based on actions taken by the president earlier this year, the United States has secured enough Pfizer and Moderna doses for 300 million Americans. We are working now with our state and federal partners to get anyone scheduled for a J&J vaccine quickly rescheduled for a Pfizer or Moderna vaccine," he added.
The likely duration of the pause remains unclear.
"I know this has been a long and difficult pandemic, and people are tired of the steps they have to take," Dr. Schuchat said. "Steps taken today make sure the health care system is ready to diagnose, treat and report [any additional cases] and the public has the information necessary to stay safe."
A version of this article first appeared on WebMD.com.
This article was updated 4/13/21.
The Food and Drug Administration and Centers for Disease Control and Prevention on April 13 recommended that use of the Johnson & Johnson COVID-19 vaccine be paused after reports of blood clots in patients receiving the shot, the agencies have announced.
In a statement, FDA said 6.8 million doses of the J&J vaccine have been administered and the agency is investigating six reported cases of a rare and severe blood clot occurring in patients who received the vaccine.
The pause is intended to give time to alert the public to this "very rare" condition, experts said during a joint CDC-FDA media briefing April 13.
"It was clear to us that we needed to alert the public," Janet Woodcock, MD, acting FDA commissioner, said. The move also will allow "time for the healthcare community to learn what they need to know about how to diagnose, treat and report" any additional cases.
The CDC will convene a meeting of the Advisory Committee on Immunization Practices on April 14 to review the cases.
"I know the information today will be very concerning to Americans who have already received the Johnson & Johnson vaccine," said Anne Schuchat, MD, principal deputy director at the CDC.
"For people who got the vaccine more than one month ago, the risk is very low at this time," she added. "For people who recently got the vaccine, in the last couple of weeks, look for symptoms."
Headache, leg pain, abdominal pain, and shortness of breath were among the reported symptoms. All six cases arose within 6 to 13 days of receipt of the Johnson & Johnson vaccine.
Traditional treatment dangerous
Importantly, treatment for traditional blood clots, such as the drug heparin, should not be used for these clots. "The issue here with these types of blood clots is that if one administers the standard treatment we give for blood clots, one can cause tremendous harm or it can be fatal," said Peter Marks, MD, director of the FDA Center for Biologics Evaluation and Research.
If health care providers see people with these symptoms along with a low platelet count or blood clots, they should ask about any recent vaccinations, Dr. Marks added.
Headache is a common side effect of COVID-19 vaccination, Dr. Marks said, but it typically happens within a day or two. In contrast, the headaches associated with these blood clots come 1 to 2 weeks later and were very severe.
Not all of the six women involved in the events had a pre-existing condition or risk factor, Dr. Schuchat said.
Severe but 'extremely rare'
To put the numbers in context, the six reported events occurred among millions of people who received the Johnson & Johnson vaccine to date.
"There have been six reports of a severe stroke-like illness due to low platelet count and more than six million doses of the Johnson & Johnson vaccine have been administered so far," Dr. Schuchat said.
"I would like to stress these events are extremely rare," Dr. Woodcock said, "but we take all reports of adverse events after vaccination very seriously."
The company response
Johnson & Johnson in a statement said, "We are aware of an extremely rare disorder involving people with blood clots in combination with low platelets in a small number of individuals who have received our COVID-19 vaccine. The United States Centers for Disease Control (CDC) and Food and Drug Administration (FDA) are reviewing data involving six reported U.S. cases out of more than 6.8 million doses administered. Out of an abundance of caution, the CDC and FDA have recommended a pause in the use of our vaccine."
The company said they are also reviewing these cases with European regulators and "we have made the decision to proactively delay the rollout of our vaccine in Europe."
Overall vaccinations continuing apace
"This announcement will not have a significant impact on our vaccination plan. Johnson & Johnson vaccine makes up less than 5% of the recorded shots in arms in the United States to date," Jeff Zients, White House COVID-19 Response Coordinator, said in a statement.
"Based on actions taken by the president earlier this year, the United States has secured enough Pfizer and Moderna doses for 300 million Americans. We are working now with our state and federal partners to get anyone scheduled for a J&J vaccine quickly rescheduled for a Pfizer or Moderna vaccine," he added.
The likely duration of the pause remains unclear.
"I know this has been a long and difficult pandemic, and people are tired of the steps they have to take," Dr. Schuchat said. "Steps taken today make sure the health care system is ready to diagnose, treat and report [any additional cases] and the public has the information necessary to stay safe."
A version of this article first appeared on WebMD.com.
This article was updated 4/13/21.
OCS heart system earns hard-won backing of FDA panel
After more than 10 hours of intense debate, a Food and Drug Administration advisory panel gave its support to a premarket approval application (PMA) for the TransMedics Organ Care System (OCS) Heart system.
The OCS Heart is a portable extracorporeal perfusion and monitoring system designed to keep a donor heart in a normothermic, beating state. The “heart in a box” technology allows donor hearts to be transported across longer distances than is possible with standard cold storage, which can safely preserve donor hearts for about 4 hours.
The Circulatory System Devices Panel of the Medical Devices Advisory Committee voted 12 to 5, with 1 abstention, that the benefits of the OCS Heart System outweigh its risks.
The panel voted in favor of the OCS Heart being effective (10 yes, 6 no, and 2 abstaining) and safe (9 yes, 7 no, 2 abstaining) but not without mixed feelings.
James Blankenship, MD, a cardiologist at the University of New Mexico, Albuquerque, voted yes to all three questions but said: “If it had been compared to standard of care, I would have voted no to all three. But if it’s compared to getting an [left ventricular assist device] LVAD or not getting a heart at all, I would say the benefits outweigh the risks.”
Marc R. Katz, MD, chief of cardiothoracic surgery, Medical University of South Carolina, Charleston, also gave universal support, noting that the rate of heart transplantations has been flat for years. “This is a big step forward toward being able to expand that number. Now all that said, it obviously was a less-than-perfect study and I do think there needs to be some constraints put on the utilization.”
The panel reviewed data from the single-arm OCS Heart EXPAND trial and associated EXPAND Continued Access Protocol (CAP), as well the sponsor’s first OCS Heart trial, PROCEED II.
EXPAND met its effectiveness endpoint, with 88% of donor hearts successfully transplanted, an 8% incidence of severe primary graft dysfunction (PGD) 24 hours after transplantation, and 94.6% survival at 30 days.
Data from 41 patients with 30-day follow-up in the ongoing EXPAND CAP show 91% of donor hearts were utilized, a 2.4% incidence of severe PGD, and 100% 30-day survival.
The sponsor and the FDA clashed over changes made to the trial after the PMA was submitted, the appropriateness of the effectiveness outcome, and claims by the FDA that there was substantial overlap in demographic characteristics between the extended criteria donor hearts in the EXPAND trials and the standard criteria donor hearts in PROCEED II.
TransMedics previously submitted a PMA based on PROCEED II but it noted in submitted documents that it was withdrawn because of “fundamental disagreements with FDA” on the interpretation of a post hoc analysis with United Network for Organ Sharing registry data that identified increased all-cause mortality risk but comparable cardiac-related mortality in patients with OCS hearts.
During the marathon hearing, FDA officials presented several post hoc analyses, including one stratified by donor inclusion criteria, in which 30-day survival estimates were worse in recipients of single-criterion organs than for those receiving donor organs with multiple inclusion criteria (85% vs. 91.4%). In a second analysis, 2-year point estimates of survival also trended lower with donor organs having only one extended criterion.
Reported EXPAND CAP 6- and 12-month survival estimates were 100% and 93%, respectively, which was higher than EXPAND (93% and 84%), but there was substantial censoring (>50%) at 6 months and beyond, FDA officials said.
When EXPAND and CAP data were pooled, modeled survival curves shifted upward but there was a substantial site effect, with a single site contributing 46% of data, which may affect generalizability of the results, they noted.
“I voted yes for safety, no for efficacy, and no for approval and I’d just like to say I found this to be the most difficult vote in my experience on this panel,” John Hirshfeld, MD, University of Pennsylvania, Philadelphia, said. “I was very concerned that the PROCEED data suggests a possible harm, and in the absence of an interpretable comparator for the EXPAND trial, it’s really not possible to decide if there’s efficacy.”
Keith B. Allen, MD, director of surgical research at Saint Luke’s Hospital of Kansas City (Mo.), said, “I voted no on safety; I’m not going to give the company a pass. I think their animal data was sorely lacking and a lot of issues over the last 10 years could have been addressed with some key animal studies.
“For efficacy and risk/benefit, I voted yes for both,” he said. “Had this been standard of care and only PROCEED II, I would have voted no, but I do think there are a lot of hearts that go in the bucket and this is a challenging population.”
More than a dozen physicians and patients spoke at the open public hearing about the potential for the device to expand donor heart utilization, including a recipient whose own father died while waiting on the transplant list. Only about 3 out of every 10 donated hearts are used for transplant. To ensure fair access, particularly for patients in rural areas, federal changes in 2020 mandate that organs be allocated to the sickest patients first.
Data showed that the OCS Heart System was associated with shorter waiting list times, compared with U.S. averages but longer preservation times than cold static preservation.
In all, 13% of accepted donor organs were subsequently turned down after OCS heart preservation. Lactate levels were cited as the principal reason for turn-down but, FDA officials said, the validity of using lactate as a marker for transplantability is unclear.
Pathologic analysis of OCS Heart turned-down donor hearts with stable antemortem hemodynamics, normal or near-normal anatomy and normal ventricular function by echocardiography, and autopsy findings of acute diffuse or multifocal myocardial damage “suggest that in an important proportion of cases the OCS Heart system did not provide effective organ preservation or its use caused severe myocardial damage to what might have been an acceptable graft for transplant,” said Andrew Farb, MD, chief medical officer of the FDA’s Office of Cardiovascular Devices.
Proposed indication
In the present PMA, the OCS Heart System is indicated for donor hearts with one or more of the following characteristics: an expected cross-clamp or ischemic time of at least 4 hours because of donor or recipient characteristics; or an expected total cross-clamp time of at least 2 hours plus one of the following risk factors: donor age 55 or older, history of cardiac arrest and downtime of at least 20 minutes, history of alcoholism, history of diabetes, donor ejection fraction of 40%-50%,history of left ventricular hypertrophy, and donor angiogram with luminal irregularities but no significant coronary artery disease
Several members voiced concern about “indication creep” should the device be approved by the FDA, and highlighted the 2-hour cross-clamp time plus wide-ranging risk factors.
“I’m a surgeon and I voted no on all three counts,” said Murray H. Kwon, MD, Ronald Reagan University of California, Los Angeles Medical Center. “As far as risk/benefit, if it was just limited to one group – the 4-hour plus – I would say yes, but if you’re going to tell me that there’s a risk/benefit for the 2-hour with the alcoholic, I don’t know how that was proved in anything.”
Dr. Kwon was also troubled by lack of proper controls and by the one quarter of patients who ended up on mechanical circulatory support in the first 30 days after transplant. “I find that highly aberrant.”
Joaquin E. Cigarroa, MD, head of cardiovascular medicine, Oregon Health & Science University, Portland, said the unmet need for patients with refractory, end-stage heart failure is challenging and quite emotional, but also voted no across the board, citing concerns about a lack of comparator in the EXPAND trials and overall out-of-body ischemic time.
“As it relates to risk/benefit, I thought long and hard about voting yes despite all the unknowns because of this emotion, but ultimately I voted no because of the secondary 2-hours plus alcoholism, diabetes, or minor coronary disease, in which the ischemic burden and ongoing lactate production concern me,” he said.
Although the panel decision is nonbinding, there was strong support from the committee members for a randomized, postapproval trial and more complete animal studies.
A version of this article first appeared on Medscape.com.
After more than 10 hours of intense debate, a Food and Drug Administration advisory panel gave its support to a premarket approval application (PMA) for the TransMedics Organ Care System (OCS) Heart system.
The OCS Heart is a portable extracorporeal perfusion and monitoring system designed to keep a donor heart in a normothermic, beating state. The “heart in a box” technology allows donor hearts to be transported across longer distances than is possible with standard cold storage, which can safely preserve donor hearts for about 4 hours.
The Circulatory System Devices Panel of the Medical Devices Advisory Committee voted 12 to 5, with 1 abstention, that the benefits of the OCS Heart System outweigh its risks.
The panel voted in favor of the OCS Heart being effective (10 yes, 6 no, and 2 abstaining) and safe (9 yes, 7 no, 2 abstaining) but not without mixed feelings.
James Blankenship, MD, a cardiologist at the University of New Mexico, Albuquerque, voted yes to all three questions but said: “If it had been compared to standard of care, I would have voted no to all three. But if it’s compared to getting an [left ventricular assist device] LVAD or not getting a heart at all, I would say the benefits outweigh the risks.”
Marc R. Katz, MD, chief of cardiothoracic surgery, Medical University of South Carolina, Charleston, also gave universal support, noting that the rate of heart transplantations has been flat for years. “This is a big step forward toward being able to expand that number. Now all that said, it obviously was a less-than-perfect study and I do think there needs to be some constraints put on the utilization.”
The panel reviewed data from the single-arm OCS Heart EXPAND trial and associated EXPAND Continued Access Protocol (CAP), as well the sponsor’s first OCS Heart trial, PROCEED II.
EXPAND met its effectiveness endpoint, with 88% of donor hearts successfully transplanted, an 8% incidence of severe primary graft dysfunction (PGD) 24 hours after transplantation, and 94.6% survival at 30 days.
Data from 41 patients with 30-day follow-up in the ongoing EXPAND CAP show 91% of donor hearts were utilized, a 2.4% incidence of severe PGD, and 100% 30-day survival.
The sponsor and the FDA clashed over changes made to the trial after the PMA was submitted, the appropriateness of the effectiveness outcome, and claims by the FDA that there was substantial overlap in demographic characteristics between the extended criteria donor hearts in the EXPAND trials and the standard criteria donor hearts in PROCEED II.
TransMedics previously submitted a PMA based on PROCEED II but it noted in submitted documents that it was withdrawn because of “fundamental disagreements with FDA” on the interpretation of a post hoc analysis with United Network for Organ Sharing registry data that identified increased all-cause mortality risk but comparable cardiac-related mortality in patients with OCS hearts.
During the marathon hearing, FDA officials presented several post hoc analyses, including one stratified by donor inclusion criteria, in which 30-day survival estimates were worse in recipients of single-criterion organs than for those receiving donor organs with multiple inclusion criteria (85% vs. 91.4%). In a second analysis, 2-year point estimates of survival also trended lower with donor organs having only one extended criterion.
Reported EXPAND CAP 6- and 12-month survival estimates were 100% and 93%, respectively, which was higher than EXPAND (93% and 84%), but there was substantial censoring (>50%) at 6 months and beyond, FDA officials said.
When EXPAND and CAP data were pooled, modeled survival curves shifted upward but there was a substantial site effect, with a single site contributing 46% of data, which may affect generalizability of the results, they noted.
“I voted yes for safety, no for efficacy, and no for approval and I’d just like to say I found this to be the most difficult vote in my experience on this panel,” John Hirshfeld, MD, University of Pennsylvania, Philadelphia, said. “I was very concerned that the PROCEED data suggests a possible harm, and in the absence of an interpretable comparator for the EXPAND trial, it’s really not possible to decide if there’s efficacy.”
Keith B. Allen, MD, director of surgical research at Saint Luke’s Hospital of Kansas City (Mo.), said, “I voted no on safety; I’m not going to give the company a pass. I think their animal data was sorely lacking and a lot of issues over the last 10 years could have been addressed with some key animal studies.
“For efficacy and risk/benefit, I voted yes for both,” he said. “Had this been standard of care and only PROCEED II, I would have voted no, but I do think there are a lot of hearts that go in the bucket and this is a challenging population.”
More than a dozen physicians and patients spoke at the open public hearing about the potential for the device to expand donor heart utilization, including a recipient whose own father died while waiting on the transplant list. Only about 3 out of every 10 donated hearts are used for transplant. To ensure fair access, particularly for patients in rural areas, federal changes in 2020 mandate that organs be allocated to the sickest patients first.
Data showed that the OCS Heart System was associated with shorter waiting list times, compared with U.S. averages but longer preservation times than cold static preservation.
In all, 13% of accepted donor organs were subsequently turned down after OCS heart preservation. Lactate levels were cited as the principal reason for turn-down but, FDA officials said, the validity of using lactate as a marker for transplantability is unclear.
Pathologic analysis of OCS Heart turned-down donor hearts with stable antemortem hemodynamics, normal or near-normal anatomy and normal ventricular function by echocardiography, and autopsy findings of acute diffuse or multifocal myocardial damage “suggest that in an important proportion of cases the OCS Heart system did not provide effective organ preservation or its use caused severe myocardial damage to what might have been an acceptable graft for transplant,” said Andrew Farb, MD, chief medical officer of the FDA’s Office of Cardiovascular Devices.
Proposed indication
In the present PMA, the OCS Heart System is indicated for donor hearts with one or more of the following characteristics: an expected cross-clamp or ischemic time of at least 4 hours because of donor or recipient characteristics; or an expected total cross-clamp time of at least 2 hours plus one of the following risk factors: donor age 55 or older, history of cardiac arrest and downtime of at least 20 minutes, history of alcoholism, history of diabetes, donor ejection fraction of 40%-50%,history of left ventricular hypertrophy, and donor angiogram with luminal irregularities but no significant coronary artery disease
Several members voiced concern about “indication creep” should the device be approved by the FDA, and highlighted the 2-hour cross-clamp time plus wide-ranging risk factors.
“I’m a surgeon and I voted no on all three counts,” said Murray H. Kwon, MD, Ronald Reagan University of California, Los Angeles Medical Center. “As far as risk/benefit, if it was just limited to one group – the 4-hour plus – I would say yes, but if you’re going to tell me that there’s a risk/benefit for the 2-hour with the alcoholic, I don’t know how that was proved in anything.”
Dr. Kwon was also troubled by lack of proper controls and by the one quarter of patients who ended up on mechanical circulatory support in the first 30 days after transplant. “I find that highly aberrant.”
Joaquin E. Cigarroa, MD, head of cardiovascular medicine, Oregon Health & Science University, Portland, said the unmet need for patients with refractory, end-stage heart failure is challenging and quite emotional, but also voted no across the board, citing concerns about a lack of comparator in the EXPAND trials and overall out-of-body ischemic time.
“As it relates to risk/benefit, I thought long and hard about voting yes despite all the unknowns because of this emotion, but ultimately I voted no because of the secondary 2-hours plus alcoholism, diabetes, or minor coronary disease, in which the ischemic burden and ongoing lactate production concern me,” he said.
Although the panel decision is nonbinding, there was strong support from the committee members for a randomized, postapproval trial and more complete animal studies.
A version of this article first appeared on Medscape.com.
After more than 10 hours of intense debate, a Food and Drug Administration advisory panel gave its support to a premarket approval application (PMA) for the TransMedics Organ Care System (OCS) Heart system.
The OCS Heart is a portable extracorporeal perfusion and monitoring system designed to keep a donor heart in a normothermic, beating state. The “heart in a box” technology allows donor hearts to be transported across longer distances than is possible with standard cold storage, which can safely preserve donor hearts for about 4 hours.
The Circulatory System Devices Panel of the Medical Devices Advisory Committee voted 12 to 5, with 1 abstention, that the benefits of the OCS Heart System outweigh its risks.
The panel voted in favor of the OCS Heart being effective (10 yes, 6 no, and 2 abstaining) and safe (9 yes, 7 no, 2 abstaining) but not without mixed feelings.
James Blankenship, MD, a cardiologist at the University of New Mexico, Albuquerque, voted yes to all three questions but said: “If it had been compared to standard of care, I would have voted no to all three. But if it’s compared to getting an [left ventricular assist device] LVAD or not getting a heart at all, I would say the benefits outweigh the risks.”
Marc R. Katz, MD, chief of cardiothoracic surgery, Medical University of South Carolina, Charleston, also gave universal support, noting that the rate of heart transplantations has been flat for years. “This is a big step forward toward being able to expand that number. Now all that said, it obviously was a less-than-perfect study and I do think there needs to be some constraints put on the utilization.”
The panel reviewed data from the single-arm OCS Heart EXPAND trial and associated EXPAND Continued Access Protocol (CAP), as well the sponsor’s first OCS Heart trial, PROCEED II.
EXPAND met its effectiveness endpoint, with 88% of donor hearts successfully transplanted, an 8% incidence of severe primary graft dysfunction (PGD) 24 hours after transplantation, and 94.6% survival at 30 days.
Data from 41 patients with 30-day follow-up in the ongoing EXPAND CAP show 91% of donor hearts were utilized, a 2.4% incidence of severe PGD, and 100% 30-day survival.
The sponsor and the FDA clashed over changes made to the trial after the PMA was submitted, the appropriateness of the effectiveness outcome, and claims by the FDA that there was substantial overlap in demographic characteristics between the extended criteria donor hearts in the EXPAND trials and the standard criteria donor hearts in PROCEED II.
TransMedics previously submitted a PMA based on PROCEED II but it noted in submitted documents that it was withdrawn because of “fundamental disagreements with FDA” on the interpretation of a post hoc analysis with United Network for Organ Sharing registry data that identified increased all-cause mortality risk but comparable cardiac-related mortality in patients with OCS hearts.
During the marathon hearing, FDA officials presented several post hoc analyses, including one stratified by donor inclusion criteria, in which 30-day survival estimates were worse in recipients of single-criterion organs than for those receiving donor organs with multiple inclusion criteria (85% vs. 91.4%). In a second analysis, 2-year point estimates of survival also trended lower with donor organs having only one extended criterion.
Reported EXPAND CAP 6- and 12-month survival estimates were 100% and 93%, respectively, which was higher than EXPAND (93% and 84%), but there was substantial censoring (>50%) at 6 months and beyond, FDA officials said.
When EXPAND and CAP data were pooled, modeled survival curves shifted upward but there was a substantial site effect, with a single site contributing 46% of data, which may affect generalizability of the results, they noted.
“I voted yes for safety, no for efficacy, and no for approval and I’d just like to say I found this to be the most difficult vote in my experience on this panel,” John Hirshfeld, MD, University of Pennsylvania, Philadelphia, said. “I was very concerned that the PROCEED data suggests a possible harm, and in the absence of an interpretable comparator for the EXPAND trial, it’s really not possible to decide if there’s efficacy.”
Keith B. Allen, MD, director of surgical research at Saint Luke’s Hospital of Kansas City (Mo.), said, “I voted no on safety; I’m not going to give the company a pass. I think their animal data was sorely lacking and a lot of issues over the last 10 years could have been addressed with some key animal studies.
“For efficacy and risk/benefit, I voted yes for both,” he said. “Had this been standard of care and only PROCEED II, I would have voted no, but I do think there are a lot of hearts that go in the bucket and this is a challenging population.”
More than a dozen physicians and patients spoke at the open public hearing about the potential for the device to expand donor heart utilization, including a recipient whose own father died while waiting on the transplant list. Only about 3 out of every 10 donated hearts are used for transplant. To ensure fair access, particularly for patients in rural areas, federal changes in 2020 mandate that organs be allocated to the sickest patients first.
Data showed that the OCS Heart System was associated with shorter waiting list times, compared with U.S. averages but longer preservation times than cold static preservation.
In all, 13% of accepted donor organs were subsequently turned down after OCS heart preservation. Lactate levels were cited as the principal reason for turn-down but, FDA officials said, the validity of using lactate as a marker for transplantability is unclear.
Pathologic analysis of OCS Heart turned-down donor hearts with stable antemortem hemodynamics, normal or near-normal anatomy and normal ventricular function by echocardiography, and autopsy findings of acute diffuse or multifocal myocardial damage “suggest that in an important proportion of cases the OCS Heart system did not provide effective organ preservation or its use caused severe myocardial damage to what might have been an acceptable graft for transplant,” said Andrew Farb, MD, chief medical officer of the FDA’s Office of Cardiovascular Devices.
Proposed indication
In the present PMA, the OCS Heart System is indicated for donor hearts with one or more of the following characteristics: an expected cross-clamp or ischemic time of at least 4 hours because of donor or recipient characteristics; or an expected total cross-clamp time of at least 2 hours plus one of the following risk factors: donor age 55 or older, history of cardiac arrest and downtime of at least 20 minutes, history of alcoholism, history of diabetes, donor ejection fraction of 40%-50%,history of left ventricular hypertrophy, and donor angiogram with luminal irregularities but no significant coronary artery disease
Several members voiced concern about “indication creep” should the device be approved by the FDA, and highlighted the 2-hour cross-clamp time plus wide-ranging risk factors.
“I’m a surgeon and I voted no on all three counts,” said Murray H. Kwon, MD, Ronald Reagan University of California, Los Angeles Medical Center. “As far as risk/benefit, if it was just limited to one group – the 4-hour plus – I would say yes, but if you’re going to tell me that there’s a risk/benefit for the 2-hour with the alcoholic, I don’t know how that was proved in anything.”
Dr. Kwon was also troubled by lack of proper controls and by the one quarter of patients who ended up on mechanical circulatory support in the first 30 days after transplant. “I find that highly aberrant.”
Joaquin E. Cigarroa, MD, head of cardiovascular medicine, Oregon Health & Science University, Portland, said the unmet need for patients with refractory, end-stage heart failure is challenging and quite emotional, but also voted no across the board, citing concerns about a lack of comparator in the EXPAND trials and overall out-of-body ischemic time.
“As it relates to risk/benefit, I thought long and hard about voting yes despite all the unknowns because of this emotion, but ultimately I voted no because of the secondary 2-hours plus alcoholism, diabetes, or minor coronary disease, in which the ischemic burden and ongoing lactate production concern me,” he said.
Although the panel decision is nonbinding, there was strong support from the committee members for a randomized, postapproval trial and more complete animal studies.
A version of this article first appeared on Medscape.com.









