User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Less ambulatory care occurred than expected in pandemic, according to study
according to an analysis of national claims data from Jan. 1, 2019, to Oct. 31, 2020.
“The COVID-19 pandemic has seriously disrupted access to U.S. ambulatory care, endangering population health,” said John N. Mafi, MD, of the University of California, Los Angeles, in a presentation at the annual meeting of the Society of General Internal Medicine.
Dr. Mafi and colleagues conducted the analysis, which included 20 monthly cohorts, and measured outpatient visit rates per 100 members across all 20 study months. The researchers used a “difference-in-differences study design” and compared changes in rates of ambulatory care visits in January-February 2019 through September-October 2019 with the same periods in 2020.
They found that overall utilization fell to 68.9% of expected rates. This number increased to 82.6% of expected rates by May-June 2020 and to 87.7% of expected rates by July-August 2020.
To examine the impact of COVID-19 on U.S. ambulatory care patterns, the researchers identified 10.4 million individuals aged 18 years and older using the MedInsight research claims database. This database included Medicaid, commercial, dual eligible (receiving both Medicare and Medicaid benefits), Medicare Advantage (MA), and Medicare fee-for-service (FFS) patients. The average age of the individuals studied was 52 years, and 55% of the population were women. The researchers measured outpatient visit rates per 100 beneficiaries for several types of ambulatory care visits: emergency, urgent care, office, physical exams, preventive, alcohol/drug, and psychiatric care.
The researchers verified parallel trends in visits between 2018 and 2019 to establish a historical benchmark and divided the patient population into three groups based on insurance enrollment (continuously enrolled, not continuously enrolled, and fully enrolled) to account for new members adding insurance and disrupted coverage caused by job losses or other factors. The trends in ambulatory care utilization were similar between cohorts across the groups.
The rebound seen by the summer of 2020 showed variation when broken out by insurance type: 94.0% for Medicare FFS; 88.9% for commercial insurance; 86.3% for Medicare Advantage; 83.6% for dual eligible; and 78.0% for Medicaid.
“The big picture is that utilization looks similar across the three groups and has not attained prepandemic levels,” Dr. Mafi said.
When the results were divided by service type, utilization rates remained below expected rates while needs remain similar for U.S. Preventive Services Task Force–recommended preventive screening services, Dr. Mafi noted. The demand for psychiatric and substance use services has increased, but use rates are below expected rates. In addition, both avoidable and nonavoidable ED utilization both remained below expected rates.
In-person visits are down across insurance groups, but virtual visits are skyrocketing, across all insurance groups, Dr. Mafi added. However, virtual care visits have not completely compensated for declines in in-person visits, notably among dual-eligible and Medicaid insurance members.
Takeaways for policy makers include the fact that, while some reductions in unnecessary care, such as avoidable ED visits, may be beneficial, the “reduced USPSTF-recommended cancer and other evidence-based disease prevention may worsen health outcomes, particularly for Medicaid beneficiaries,” he said.
Outreach and outcomes
The study is important because “understanding ambulatory care patterns during the pandemic can highlight vulnerabilities and opportunities in our health care system,” Dr. Mafi said in an interview.
“While the COVID-19 pandemic has seriously disrupted access to U.S. ambulatory care, most studies have focused on the early months of the pandemic,” he noted.
Dr. Mafi said he was not surprised that ambulatory care utilization has not rebounded among Medicaid beneficiaries relative to other insurance groups.
“Medicaid beneficiaries are underresourced individuals who are disproportionately racial/ethnic minorities, and they historically have had difficulties accessing care. Our data suggest that the COVID-19 pandemic may be widening these preexisting inequities in access to ambulatory care,” he observed.
The study findings were limited by the use of the MedInsight research dataset, which is a convenience sample; and, therefore, the results might not be generalizable nationally, Dr. Mafi said. “However, it does include beneficiaries from all major insurance types across all 50 U.S. states. Additionally, our analysis was completed at the population level rather than the patient level, and so we were unable to account for patient-level characteristics such as clinical complexity,” he explained.
“The take-home message for clinicians is that our patients with Medicaid insurance may need additional efforts to overcome barriers to accessing ambulatory care, such as creating robust telemedicine outreach programs,” said Dr. Mafi. “Policy makers should also consider providing additional support and resources to safety net health systems who disproportionately care for Medicaid beneficiaries, such as higher reimbursements for both in-person and telemedicine visits.”
More research is needed, he emphasized. “We urgently need further inquiry into the impact of this persistently deferred ambulatory care utilization on important health outcomes such as preventable death/disability and quality of care.”
COVID consequences challenge ambulatory care
“These study findings mirror what we are seeing in primary care settings,” Maureen Lyons, MD, of Washington University. St. Louis, said in an interview. “With the pandemic, there are many additional barriers for patients accessing care, and these barriers have disproportionately impacted those who are already disadvantaged.
“From clinical experience, there are barriers directly related to COVID-19, such as the risk of infection or discomfort being in a clinic setting with other people. However, there also are barriers related to change in financial situation or insurance related to changes or loss of employment,” she said.
“Additionally, many patients have needed to take on increased responsibilities in other areas of their lives, such as caring for an ill family member or being responsible for children’s virtual school,” she said. These new responsibilities can lead people to skip or postpone ambulatory care visits.
“Loss of ambulatory care is likely to lead to increases in preventable illnesses with long-lasting effects,” Dr. Lyons noted. “Studying this in a robust fashion, as Dr. Mafi and colleagues have done, is a critical step in understanding and addressing this urgent need.”
Dr. Mafi noted that the data he presented is preliminary, and that he and his team hope to publish finalized estimates of ambulatory utilization rates in a forthcoming scientific paper.
The study was a collaboration between UCLA and Millman MedInsight, an actuarial health analytics company. Several coauthors are Millman employees. Dr. Mafi and the other researchers had no other relevant financial conflicts to disclose. Dr. Lyons had no financial conflicts to disclose.
according to an analysis of national claims data from Jan. 1, 2019, to Oct. 31, 2020.
“The COVID-19 pandemic has seriously disrupted access to U.S. ambulatory care, endangering population health,” said John N. Mafi, MD, of the University of California, Los Angeles, in a presentation at the annual meeting of the Society of General Internal Medicine.
Dr. Mafi and colleagues conducted the analysis, which included 20 monthly cohorts, and measured outpatient visit rates per 100 members across all 20 study months. The researchers used a “difference-in-differences study design” and compared changes in rates of ambulatory care visits in January-February 2019 through September-October 2019 with the same periods in 2020.
They found that overall utilization fell to 68.9% of expected rates. This number increased to 82.6% of expected rates by May-June 2020 and to 87.7% of expected rates by July-August 2020.
To examine the impact of COVID-19 on U.S. ambulatory care patterns, the researchers identified 10.4 million individuals aged 18 years and older using the MedInsight research claims database. This database included Medicaid, commercial, dual eligible (receiving both Medicare and Medicaid benefits), Medicare Advantage (MA), and Medicare fee-for-service (FFS) patients. The average age of the individuals studied was 52 years, and 55% of the population were women. The researchers measured outpatient visit rates per 100 beneficiaries for several types of ambulatory care visits: emergency, urgent care, office, physical exams, preventive, alcohol/drug, and psychiatric care.
The researchers verified parallel trends in visits between 2018 and 2019 to establish a historical benchmark and divided the patient population into three groups based on insurance enrollment (continuously enrolled, not continuously enrolled, and fully enrolled) to account for new members adding insurance and disrupted coverage caused by job losses or other factors. The trends in ambulatory care utilization were similar between cohorts across the groups.
The rebound seen by the summer of 2020 showed variation when broken out by insurance type: 94.0% for Medicare FFS; 88.9% for commercial insurance; 86.3% for Medicare Advantage; 83.6% for dual eligible; and 78.0% for Medicaid.
“The big picture is that utilization looks similar across the three groups and has not attained prepandemic levels,” Dr. Mafi said.
When the results were divided by service type, utilization rates remained below expected rates while needs remain similar for U.S. Preventive Services Task Force–recommended preventive screening services, Dr. Mafi noted. The demand for psychiatric and substance use services has increased, but use rates are below expected rates. In addition, both avoidable and nonavoidable ED utilization both remained below expected rates.
In-person visits are down across insurance groups, but virtual visits are skyrocketing, across all insurance groups, Dr. Mafi added. However, virtual care visits have not completely compensated for declines in in-person visits, notably among dual-eligible and Medicaid insurance members.
Takeaways for policy makers include the fact that, while some reductions in unnecessary care, such as avoidable ED visits, may be beneficial, the “reduced USPSTF-recommended cancer and other evidence-based disease prevention may worsen health outcomes, particularly for Medicaid beneficiaries,” he said.
Outreach and outcomes
The study is important because “understanding ambulatory care patterns during the pandemic can highlight vulnerabilities and opportunities in our health care system,” Dr. Mafi said in an interview.
“While the COVID-19 pandemic has seriously disrupted access to U.S. ambulatory care, most studies have focused on the early months of the pandemic,” he noted.
Dr. Mafi said he was not surprised that ambulatory care utilization has not rebounded among Medicaid beneficiaries relative to other insurance groups.
“Medicaid beneficiaries are underresourced individuals who are disproportionately racial/ethnic minorities, and they historically have had difficulties accessing care. Our data suggest that the COVID-19 pandemic may be widening these preexisting inequities in access to ambulatory care,” he observed.
The study findings were limited by the use of the MedInsight research dataset, which is a convenience sample; and, therefore, the results might not be generalizable nationally, Dr. Mafi said. “However, it does include beneficiaries from all major insurance types across all 50 U.S. states. Additionally, our analysis was completed at the population level rather than the patient level, and so we were unable to account for patient-level characteristics such as clinical complexity,” he explained.
“The take-home message for clinicians is that our patients with Medicaid insurance may need additional efforts to overcome barriers to accessing ambulatory care, such as creating robust telemedicine outreach programs,” said Dr. Mafi. “Policy makers should also consider providing additional support and resources to safety net health systems who disproportionately care for Medicaid beneficiaries, such as higher reimbursements for both in-person and telemedicine visits.”
More research is needed, he emphasized. “We urgently need further inquiry into the impact of this persistently deferred ambulatory care utilization on important health outcomes such as preventable death/disability and quality of care.”
COVID consequences challenge ambulatory care
“These study findings mirror what we are seeing in primary care settings,” Maureen Lyons, MD, of Washington University. St. Louis, said in an interview. “With the pandemic, there are many additional barriers for patients accessing care, and these barriers have disproportionately impacted those who are already disadvantaged.
“From clinical experience, there are barriers directly related to COVID-19, such as the risk of infection or discomfort being in a clinic setting with other people. However, there also are barriers related to change in financial situation or insurance related to changes or loss of employment,” she said.
“Additionally, many patients have needed to take on increased responsibilities in other areas of their lives, such as caring for an ill family member or being responsible for children’s virtual school,” she said. These new responsibilities can lead people to skip or postpone ambulatory care visits.
“Loss of ambulatory care is likely to lead to increases in preventable illnesses with long-lasting effects,” Dr. Lyons noted. “Studying this in a robust fashion, as Dr. Mafi and colleagues have done, is a critical step in understanding and addressing this urgent need.”
Dr. Mafi noted that the data he presented is preliminary, and that he and his team hope to publish finalized estimates of ambulatory utilization rates in a forthcoming scientific paper.
The study was a collaboration between UCLA and Millman MedInsight, an actuarial health analytics company. Several coauthors are Millman employees. Dr. Mafi and the other researchers had no other relevant financial conflicts to disclose. Dr. Lyons had no financial conflicts to disclose.
according to an analysis of national claims data from Jan. 1, 2019, to Oct. 31, 2020.
“The COVID-19 pandemic has seriously disrupted access to U.S. ambulatory care, endangering population health,” said John N. Mafi, MD, of the University of California, Los Angeles, in a presentation at the annual meeting of the Society of General Internal Medicine.
Dr. Mafi and colleagues conducted the analysis, which included 20 monthly cohorts, and measured outpatient visit rates per 100 members across all 20 study months. The researchers used a “difference-in-differences study design” and compared changes in rates of ambulatory care visits in January-February 2019 through September-October 2019 with the same periods in 2020.
They found that overall utilization fell to 68.9% of expected rates. This number increased to 82.6% of expected rates by May-June 2020 and to 87.7% of expected rates by July-August 2020.
To examine the impact of COVID-19 on U.S. ambulatory care patterns, the researchers identified 10.4 million individuals aged 18 years and older using the MedInsight research claims database. This database included Medicaid, commercial, dual eligible (receiving both Medicare and Medicaid benefits), Medicare Advantage (MA), and Medicare fee-for-service (FFS) patients. The average age of the individuals studied was 52 years, and 55% of the population were women. The researchers measured outpatient visit rates per 100 beneficiaries for several types of ambulatory care visits: emergency, urgent care, office, physical exams, preventive, alcohol/drug, and psychiatric care.
The researchers verified parallel trends in visits between 2018 and 2019 to establish a historical benchmark and divided the patient population into three groups based on insurance enrollment (continuously enrolled, not continuously enrolled, and fully enrolled) to account for new members adding insurance and disrupted coverage caused by job losses or other factors. The trends in ambulatory care utilization were similar between cohorts across the groups.
The rebound seen by the summer of 2020 showed variation when broken out by insurance type: 94.0% for Medicare FFS; 88.9% for commercial insurance; 86.3% for Medicare Advantage; 83.6% for dual eligible; and 78.0% for Medicaid.
“The big picture is that utilization looks similar across the three groups and has not attained prepandemic levels,” Dr. Mafi said.
When the results were divided by service type, utilization rates remained below expected rates while needs remain similar for U.S. Preventive Services Task Force–recommended preventive screening services, Dr. Mafi noted. The demand for psychiatric and substance use services has increased, but use rates are below expected rates. In addition, both avoidable and nonavoidable ED utilization both remained below expected rates.
In-person visits are down across insurance groups, but virtual visits are skyrocketing, across all insurance groups, Dr. Mafi added. However, virtual care visits have not completely compensated for declines in in-person visits, notably among dual-eligible and Medicaid insurance members.
Takeaways for policy makers include the fact that, while some reductions in unnecessary care, such as avoidable ED visits, may be beneficial, the “reduced USPSTF-recommended cancer and other evidence-based disease prevention may worsen health outcomes, particularly for Medicaid beneficiaries,” he said.
Outreach and outcomes
The study is important because “understanding ambulatory care patterns during the pandemic can highlight vulnerabilities and opportunities in our health care system,” Dr. Mafi said in an interview.
“While the COVID-19 pandemic has seriously disrupted access to U.S. ambulatory care, most studies have focused on the early months of the pandemic,” he noted.
Dr. Mafi said he was not surprised that ambulatory care utilization has not rebounded among Medicaid beneficiaries relative to other insurance groups.
“Medicaid beneficiaries are underresourced individuals who are disproportionately racial/ethnic minorities, and they historically have had difficulties accessing care. Our data suggest that the COVID-19 pandemic may be widening these preexisting inequities in access to ambulatory care,” he observed.
The study findings were limited by the use of the MedInsight research dataset, which is a convenience sample; and, therefore, the results might not be generalizable nationally, Dr. Mafi said. “However, it does include beneficiaries from all major insurance types across all 50 U.S. states. Additionally, our analysis was completed at the population level rather than the patient level, and so we were unable to account for patient-level characteristics such as clinical complexity,” he explained.
“The take-home message for clinicians is that our patients with Medicaid insurance may need additional efforts to overcome barriers to accessing ambulatory care, such as creating robust telemedicine outreach programs,” said Dr. Mafi. “Policy makers should also consider providing additional support and resources to safety net health systems who disproportionately care for Medicaid beneficiaries, such as higher reimbursements for both in-person and telemedicine visits.”
More research is needed, he emphasized. “We urgently need further inquiry into the impact of this persistently deferred ambulatory care utilization on important health outcomes such as preventable death/disability and quality of care.”
COVID consequences challenge ambulatory care
“These study findings mirror what we are seeing in primary care settings,” Maureen Lyons, MD, of Washington University. St. Louis, said in an interview. “With the pandemic, there are many additional barriers for patients accessing care, and these barriers have disproportionately impacted those who are already disadvantaged.
“From clinical experience, there are barriers directly related to COVID-19, such as the risk of infection or discomfort being in a clinic setting with other people. However, there also are barriers related to change in financial situation or insurance related to changes or loss of employment,” she said.
“Additionally, many patients have needed to take on increased responsibilities in other areas of their lives, such as caring for an ill family member or being responsible for children’s virtual school,” she said. These new responsibilities can lead people to skip or postpone ambulatory care visits.
“Loss of ambulatory care is likely to lead to increases in preventable illnesses with long-lasting effects,” Dr. Lyons noted. “Studying this in a robust fashion, as Dr. Mafi and colleagues have done, is a critical step in understanding and addressing this urgent need.”
Dr. Mafi noted that the data he presented is preliminary, and that he and his team hope to publish finalized estimates of ambulatory utilization rates in a forthcoming scientific paper.
The study was a collaboration between UCLA and Millman MedInsight, an actuarial health analytics company. Several coauthors are Millman employees. Dr. Mafi and the other researchers had no other relevant financial conflicts to disclose. Dr. Lyons had no financial conflicts to disclose.
FROM SGIM 2021
Novel rehab program fights frailty, boosts capacity in advanced HF
A novel physical rehabilitation program for patients with advanced heart failure that aimed to improve their ability to exercise before focusing on endurance was successful in a randomized trial in ways that seem to have eluded some earlier exercise-training studies in the setting of HF.
The often-frail patients following the training regimen, initiated before discharge from hospitalization for acute decompensation, worked on capabilities such as mobility, balance, and strength deemed necessary if exercises meant to build exercise capacity were to succeed.
A huge percentage stayed with the 12-week program, which featured personalized, one-on-one training from a physical therapist. The patients benefited, with improvements in balance, walking ability, and strength, which were followed by significant gains in 6-minute walk distance (6MWD) and measures of physical functioning, frailty, and quality of life. The patients then continued elements of the program at home out to 6 months.
At that time, death and rehospitalizations did not differ between those assigned to the regimen and similar patients who had not participated in the program, although the trial wasn’t powered for clinical events.
The rehab strategy seemed to work across a wide range of patient subgroups. In particular, there was evidence that the benefits were more pronounced in patients with HF and preserved ejection fraction (HFpEF) than in those with HF and reduced ejection fraction (HFrEF), observed Dalane W. Kitzman, MD, Wake Forest University, Winston-Salem, N.C.
Dr. Kitzman presented results from the REHAB-HF (Rehabilitation Therapy in Older Acute Heart Failure Patients) trial at the annual scientific sessions of the American College of Cardiology and is lead author on its same-day publication in the New England Journal of Medicine.
An earlier pilot program unexpectedly showed that such patients recently hospitalized with HF “have significant impairments in mobility and balance,” he explained. If so, “it would be hazardous to subject them to traditional endurance training, such as walking-based treadmill or even bicycle.”
The unusual program, said Dr. Kitzman, looks to those issues before engaging the patients in endurance exercise by addressing mobility, balance, and basic strength – enough to repeatedly stand up from a sitting position, for example. “If you’re not able to stand with confidence, then you’re not able to walk on a treadmill.”
This model of exercise rehab “is used in geriatrics research, and enables them to safely increase endurance. It’s well known from geriatric studies that if you go directly to endurance in these, frail, older patients, you have little improvement and often have injuries and falls,” he added.
Guidance from telemedicine?
The functional outcomes examined in REHAB-HF “are the ones that matter to patients the most,” observed Eileen M. Handberg, PhD, of Shands Hospital at the University of Florida, Gainesville, at a presentation on the trial for the media.
“This is about being able to get out of a chair without assistance, not falling, walking farther, and feeling better as opposed to the more traditional outcome measure that has been used in cardiac rehab trials, which has been the exercise treadmill test – which most patients don’t have the capacity to do very well anyway,” said Dr. Handberg, who is not a part of REHAB-HF.
“This opens up rehab, potentially, to the more sick, who also need a better quality of life,” she said.
However, many patients invited to participate in the trial could not because they lived too far from the program, Dr. Handberg observed. “It would be nice to see if the lessons from COVID-19 might apply to this population” by making participation possible remotely, “perhaps using family members as rehab assistance,” she said.
“I was really very impressed that you had 83% adherence to a home exercise 6 months down the road, which far eclipses what we had in HF-ACTION,” said Vera Bittner, MD, University of Alabama at Birmingham, as the invited discussant following Dr. Kitzman’s formal presentation of the trial. “And it certainly eclipses what we see in the typical cardiac rehab program.”
Both Dr. Bittner and Dr. Kitzman participated in HF-ACTION, a randomized exercise-training trial for patients with chronic, stable HFrEF who were all-around less sick than those in REHAB-HF.
Four functional domains
Historically, HF exercise or rehab trials have excluded patients hospitalized with acute decompensation, and third-party reimbursement often has not covered such programs because of a lack of supporting evidence and a supposed potential for harm, Dr. Kitzman said.
Entry to REHAB-HF required the patients to be fit enough to walk 4 meters, with or without a walker or other assistant device, and to have been in the hospital for at least 24 hours with a primary diagnosis of acute decompensated HF.
The intervention relied on exercises aimed at improving the four functional domains of strength, balance, mobility, and – when those three were sufficiently developed – endurance, Dr. Kitzman and associates wrote in their published report.
“The intervention was initiated in the hospital when feasible and was subsequently transitioned to an outpatient facility as soon as possible after discharge,” they wrote. Afterward, “a key goal of the intervention during the first 3 months [the outpatient phase] was to prepare the patient to transition to the independent maintenance phase (months 4-6).”
The study’s control patients “received frequent calls from study staff to try to approximate the increased attention received by the intervention group,” Dr. Kitzman said in an interview. “They were allowed to receive all usual care as ordered by their treating physicians. This included, if ordered, standard physical therapy or cardiac rehabilitation” in 43% of the control cohort. Of the trial’s 349 patients, those assigned to the intervention scored significantly higher on the three-component Short Physical Performance Battery (SPPB) at 12 weeks than those assigned to a usual care approach that included, for some, more conventional cardiac rehabilitation (8.3 vs. 6.9; P < .001).
The SPPB, validated in trials as a proxy for clinical outcomes includes tests of balance while standing, gait speed during a 4-minute walk, and strength. The latter is the test that measures time needed to rise from a chair five times.
They also showed consistent gains in other measures of physical functioning and quality of life by 12 weeks months.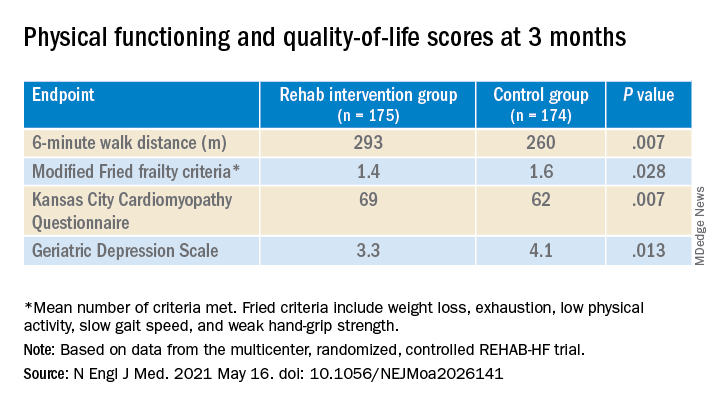
The observed SPPB treatment effect is “impressive” and “compares very favorably with previously reported estimates,” observed an accompanying editorial from Stefan D. Anker, MD, PhD, of the German Center for Cardiovascular Research and Charité Universitätsmedizin, Berlin, and Andrew J.S. Coats, DM, of the University of Warwick, Coventry, England.
“Similarly, the between-group differences seen in 6-minute walk distance (34 m) and gait speed (0.12 m/s) are clinically meaningful and sizable.”
They propose that some of the substantial quality-of-life benefit in the intervention group “may be due to better physical performance, and that part may be due to improvements in psychosocial factors and mood. It appears that exercise also resulted in patients becoming happier, or at least less depressed, as evidenced by the positive results on the Geriatric Depression Scale.”
Similar results across most subgroups
In subgroup analyses, the intervention was successful against the standard-care approach in both men and women at all ages and regardless of ejection fraction; symptom status; and whether the patient had diabetes, ischemic heart disease, or atrial fibrillation, or was obese.
Clinical outcomes were not significantly different at 6 months. The rate of death from any cause was 13% for the intervention group and 10% for the control group. There were 194 and 213 hospitalizations from any cause, respectively.
Not included in the trial’s current publication but soon to be published, Dr. Kitzman said when interviewed, is a comparison of outcomes in patients with HFpEF and HFrEF. “We found at baseline that those with HFpEF had worse impairment in physical function, quality of life, and frailty. After the intervention, there appeared to be consistently larger improvements in all outcomes, including SPPB, 6-minute walk, qualify of life, and frailty, in HFpEF versus HFrEF.”
The signals of potential benefit in HFpEF extended to clinical endpoints, he said. In contrast to similar rates of all-cause rehospitalization in HFrEF, “in patients with HFpEF, rehospitalizations were 17% lower in the intervention group, compared to the control group.” Still, he noted, the interaction P value wasn’t significant.
However, Dr. Kitzman added, mortality in the intervention group, compared with the control group, was reduced by 35% among patients with HFpEF, “but was 250% higher in HFrEF,” with a significant interaction P value.
He was careful to note that, as a phase 2 trial, REHAB-HF was underpowered for clinical events, “and even the results in the HFpEF group should not be seen as adequate evidence to change clinical care.” They were from an exploratory analysis that included relatively few events.
“Because definitive demonstration of improvement in clinical events is critical for altering clinical care guidelines and for third-party payer reimbursement decisions, we believe that a subsequent phase 3 trial is needed and are currently planning toward that,” Dr. Kitzman said.
The study was supported by research grants from the National Institutes of Health, the Kermit Glenn Phillips II Chair in Cardiovascular Medicine, and the Oristano Family Fund at Wake Forest. Dr. Kitzman disclosed receiving consulting fees or honoraria from AbbVie, AstraZeneca, Bayer Healthcare, Boehringer Ingelheim, CinRx, Corviamedical, GlaxoSmithKline, and Merck; and having an unspecified relationship with Gilead. Dr. Handberg disclosed receiving grants from Aastom Biosciences, Abbott Laboratories, Amgen, Amorcyte, AstraZeneca, Biocardia, Boehringer Ingelheim, Capricor, Cytori Therapeutics, Department of Defense, Direct Flow Medical, Everyfit, Gilead, Ionis, Medtronic, Merck, Mesoblast, Relypsa, and Sanofi-Aventis. Dr. Bittner discloses receiving consulting fees or honoraria from Pfizer and Sanofi; receiving research grants from Amgen and The Medicines Company; and having unspecified relationships with AstraZeneca, DalCor, Esperion, and Sanofi-Aventis. Dr. Anker reported receiving grants and personal fees from Abbott Vascular and Vifor; personal fees from Bayer, Boehringer Ingelheim, Novartis, Servier, Cardiac Dimensions, Thermo Fisher Scientific, AstraZeneca, Occlutech, Actimed, and Respicardia. Dr. Coats disclosed receiving personal fees from AstraZeneca, Bayer, Boehringer Ingelheim, Menarini, Novartis, Nutricia, Servier, Vifor, Abbott, Actimed, Arena, Cardiac Dimensions, Corvia, CVRx, Enopace, ESN Cleer, Faraday, WL Gore, Impulse Dynamics, and Respicardia.
A version of this article first appeared on Medscape.com.
A novel physical rehabilitation program for patients with advanced heart failure that aimed to improve their ability to exercise before focusing on endurance was successful in a randomized trial in ways that seem to have eluded some earlier exercise-training studies in the setting of HF.
The often-frail patients following the training regimen, initiated before discharge from hospitalization for acute decompensation, worked on capabilities such as mobility, balance, and strength deemed necessary if exercises meant to build exercise capacity were to succeed.
A huge percentage stayed with the 12-week program, which featured personalized, one-on-one training from a physical therapist. The patients benefited, with improvements in balance, walking ability, and strength, which were followed by significant gains in 6-minute walk distance (6MWD) and measures of physical functioning, frailty, and quality of life. The patients then continued elements of the program at home out to 6 months.
At that time, death and rehospitalizations did not differ between those assigned to the regimen and similar patients who had not participated in the program, although the trial wasn’t powered for clinical events.
The rehab strategy seemed to work across a wide range of patient subgroups. In particular, there was evidence that the benefits were more pronounced in patients with HF and preserved ejection fraction (HFpEF) than in those with HF and reduced ejection fraction (HFrEF), observed Dalane W. Kitzman, MD, Wake Forest University, Winston-Salem, N.C.
Dr. Kitzman presented results from the REHAB-HF (Rehabilitation Therapy in Older Acute Heart Failure Patients) trial at the annual scientific sessions of the American College of Cardiology and is lead author on its same-day publication in the New England Journal of Medicine.
An earlier pilot program unexpectedly showed that such patients recently hospitalized with HF “have significant impairments in mobility and balance,” he explained. If so, “it would be hazardous to subject them to traditional endurance training, such as walking-based treadmill or even bicycle.”
The unusual program, said Dr. Kitzman, looks to those issues before engaging the patients in endurance exercise by addressing mobility, balance, and basic strength – enough to repeatedly stand up from a sitting position, for example. “If you’re not able to stand with confidence, then you’re not able to walk on a treadmill.”
This model of exercise rehab “is used in geriatrics research, and enables them to safely increase endurance. It’s well known from geriatric studies that if you go directly to endurance in these, frail, older patients, you have little improvement and often have injuries and falls,” he added.
Guidance from telemedicine?
The functional outcomes examined in REHAB-HF “are the ones that matter to patients the most,” observed Eileen M. Handberg, PhD, of Shands Hospital at the University of Florida, Gainesville, at a presentation on the trial for the media.
“This is about being able to get out of a chair without assistance, not falling, walking farther, and feeling better as opposed to the more traditional outcome measure that has been used in cardiac rehab trials, which has been the exercise treadmill test – which most patients don’t have the capacity to do very well anyway,” said Dr. Handberg, who is not a part of REHAB-HF.
“This opens up rehab, potentially, to the more sick, who also need a better quality of life,” she said.
However, many patients invited to participate in the trial could not because they lived too far from the program, Dr. Handberg observed. “It would be nice to see if the lessons from COVID-19 might apply to this population” by making participation possible remotely, “perhaps using family members as rehab assistance,” she said.
“I was really very impressed that you had 83% adherence to a home exercise 6 months down the road, which far eclipses what we had in HF-ACTION,” said Vera Bittner, MD, University of Alabama at Birmingham, as the invited discussant following Dr. Kitzman’s formal presentation of the trial. “And it certainly eclipses what we see in the typical cardiac rehab program.”
Both Dr. Bittner and Dr. Kitzman participated in HF-ACTION, a randomized exercise-training trial for patients with chronic, stable HFrEF who were all-around less sick than those in REHAB-HF.
Four functional domains
Historically, HF exercise or rehab trials have excluded patients hospitalized with acute decompensation, and third-party reimbursement often has not covered such programs because of a lack of supporting evidence and a supposed potential for harm, Dr. Kitzman said.
Entry to REHAB-HF required the patients to be fit enough to walk 4 meters, with or without a walker or other assistant device, and to have been in the hospital for at least 24 hours with a primary diagnosis of acute decompensated HF.
The intervention relied on exercises aimed at improving the four functional domains of strength, balance, mobility, and – when those three were sufficiently developed – endurance, Dr. Kitzman and associates wrote in their published report.
“The intervention was initiated in the hospital when feasible and was subsequently transitioned to an outpatient facility as soon as possible after discharge,” they wrote. Afterward, “a key goal of the intervention during the first 3 months [the outpatient phase] was to prepare the patient to transition to the independent maintenance phase (months 4-6).”
The study’s control patients “received frequent calls from study staff to try to approximate the increased attention received by the intervention group,” Dr. Kitzman said in an interview. “They were allowed to receive all usual care as ordered by their treating physicians. This included, if ordered, standard physical therapy or cardiac rehabilitation” in 43% of the control cohort. Of the trial’s 349 patients, those assigned to the intervention scored significantly higher on the three-component Short Physical Performance Battery (SPPB) at 12 weeks than those assigned to a usual care approach that included, for some, more conventional cardiac rehabilitation (8.3 vs. 6.9; P < .001).
The SPPB, validated in trials as a proxy for clinical outcomes includes tests of balance while standing, gait speed during a 4-minute walk, and strength. The latter is the test that measures time needed to rise from a chair five times.
They also showed consistent gains in other measures of physical functioning and quality of life by 12 weeks months.
The observed SPPB treatment effect is “impressive” and “compares very favorably with previously reported estimates,” observed an accompanying editorial from Stefan D. Anker, MD, PhD, of the German Center for Cardiovascular Research and Charité Universitätsmedizin, Berlin, and Andrew J.S. Coats, DM, of the University of Warwick, Coventry, England.
“Similarly, the between-group differences seen in 6-minute walk distance (34 m) and gait speed (0.12 m/s) are clinically meaningful and sizable.”
They propose that some of the substantial quality-of-life benefit in the intervention group “may be due to better physical performance, and that part may be due to improvements in psychosocial factors and mood. It appears that exercise also resulted in patients becoming happier, or at least less depressed, as evidenced by the positive results on the Geriatric Depression Scale.”
Similar results across most subgroups
In subgroup analyses, the intervention was successful against the standard-care approach in both men and women at all ages and regardless of ejection fraction; symptom status; and whether the patient had diabetes, ischemic heart disease, or atrial fibrillation, or was obese.
Clinical outcomes were not significantly different at 6 months. The rate of death from any cause was 13% for the intervention group and 10% for the control group. There were 194 and 213 hospitalizations from any cause, respectively.
Not included in the trial’s current publication but soon to be published, Dr. Kitzman said when interviewed, is a comparison of outcomes in patients with HFpEF and HFrEF. “We found at baseline that those with HFpEF had worse impairment in physical function, quality of life, and frailty. After the intervention, there appeared to be consistently larger improvements in all outcomes, including SPPB, 6-minute walk, qualify of life, and frailty, in HFpEF versus HFrEF.”
The signals of potential benefit in HFpEF extended to clinical endpoints, he said. In contrast to similar rates of all-cause rehospitalization in HFrEF, “in patients with HFpEF, rehospitalizations were 17% lower in the intervention group, compared to the control group.” Still, he noted, the interaction P value wasn’t significant.
However, Dr. Kitzman added, mortality in the intervention group, compared with the control group, was reduced by 35% among patients with HFpEF, “but was 250% higher in HFrEF,” with a significant interaction P value.
He was careful to note that, as a phase 2 trial, REHAB-HF was underpowered for clinical events, “and even the results in the HFpEF group should not be seen as adequate evidence to change clinical care.” They were from an exploratory analysis that included relatively few events.
“Because definitive demonstration of improvement in clinical events is critical for altering clinical care guidelines and for third-party payer reimbursement decisions, we believe that a subsequent phase 3 trial is needed and are currently planning toward that,” Dr. Kitzman said.
The study was supported by research grants from the National Institutes of Health, the Kermit Glenn Phillips II Chair in Cardiovascular Medicine, and the Oristano Family Fund at Wake Forest. Dr. Kitzman disclosed receiving consulting fees or honoraria from AbbVie, AstraZeneca, Bayer Healthcare, Boehringer Ingelheim, CinRx, Corviamedical, GlaxoSmithKline, and Merck; and having an unspecified relationship with Gilead. Dr. Handberg disclosed receiving grants from Aastom Biosciences, Abbott Laboratories, Amgen, Amorcyte, AstraZeneca, Biocardia, Boehringer Ingelheim, Capricor, Cytori Therapeutics, Department of Defense, Direct Flow Medical, Everyfit, Gilead, Ionis, Medtronic, Merck, Mesoblast, Relypsa, and Sanofi-Aventis. Dr. Bittner discloses receiving consulting fees or honoraria from Pfizer and Sanofi; receiving research grants from Amgen and The Medicines Company; and having unspecified relationships with AstraZeneca, DalCor, Esperion, and Sanofi-Aventis. Dr. Anker reported receiving grants and personal fees from Abbott Vascular and Vifor; personal fees from Bayer, Boehringer Ingelheim, Novartis, Servier, Cardiac Dimensions, Thermo Fisher Scientific, AstraZeneca, Occlutech, Actimed, and Respicardia. Dr. Coats disclosed receiving personal fees from AstraZeneca, Bayer, Boehringer Ingelheim, Menarini, Novartis, Nutricia, Servier, Vifor, Abbott, Actimed, Arena, Cardiac Dimensions, Corvia, CVRx, Enopace, ESN Cleer, Faraday, WL Gore, Impulse Dynamics, and Respicardia.
A version of this article first appeared on Medscape.com.
A novel physical rehabilitation program for patients with advanced heart failure that aimed to improve their ability to exercise before focusing on endurance was successful in a randomized trial in ways that seem to have eluded some earlier exercise-training studies in the setting of HF.
The often-frail patients following the training regimen, initiated before discharge from hospitalization for acute decompensation, worked on capabilities such as mobility, balance, and strength deemed necessary if exercises meant to build exercise capacity were to succeed.
A huge percentage stayed with the 12-week program, which featured personalized, one-on-one training from a physical therapist. The patients benefited, with improvements in balance, walking ability, and strength, which were followed by significant gains in 6-minute walk distance (6MWD) and measures of physical functioning, frailty, and quality of life. The patients then continued elements of the program at home out to 6 months.
At that time, death and rehospitalizations did not differ between those assigned to the regimen and similar patients who had not participated in the program, although the trial wasn’t powered for clinical events.
The rehab strategy seemed to work across a wide range of patient subgroups. In particular, there was evidence that the benefits were more pronounced in patients with HF and preserved ejection fraction (HFpEF) than in those with HF and reduced ejection fraction (HFrEF), observed Dalane W. Kitzman, MD, Wake Forest University, Winston-Salem, N.C.
Dr. Kitzman presented results from the REHAB-HF (Rehabilitation Therapy in Older Acute Heart Failure Patients) trial at the annual scientific sessions of the American College of Cardiology and is lead author on its same-day publication in the New England Journal of Medicine.
An earlier pilot program unexpectedly showed that such patients recently hospitalized with HF “have significant impairments in mobility and balance,” he explained. If so, “it would be hazardous to subject them to traditional endurance training, such as walking-based treadmill or even bicycle.”
The unusual program, said Dr. Kitzman, looks to those issues before engaging the patients in endurance exercise by addressing mobility, balance, and basic strength – enough to repeatedly stand up from a sitting position, for example. “If you’re not able to stand with confidence, then you’re not able to walk on a treadmill.”
This model of exercise rehab “is used in geriatrics research, and enables them to safely increase endurance. It’s well known from geriatric studies that if you go directly to endurance in these, frail, older patients, you have little improvement and often have injuries and falls,” he added.
Guidance from telemedicine?
The functional outcomes examined in REHAB-HF “are the ones that matter to patients the most,” observed Eileen M. Handberg, PhD, of Shands Hospital at the University of Florida, Gainesville, at a presentation on the trial for the media.
“This is about being able to get out of a chair without assistance, not falling, walking farther, and feeling better as opposed to the more traditional outcome measure that has been used in cardiac rehab trials, which has been the exercise treadmill test – which most patients don’t have the capacity to do very well anyway,” said Dr. Handberg, who is not a part of REHAB-HF.
“This opens up rehab, potentially, to the more sick, who also need a better quality of life,” she said.
However, many patients invited to participate in the trial could not because they lived too far from the program, Dr. Handberg observed. “It would be nice to see if the lessons from COVID-19 might apply to this population” by making participation possible remotely, “perhaps using family members as rehab assistance,” she said.
“I was really very impressed that you had 83% adherence to a home exercise 6 months down the road, which far eclipses what we had in HF-ACTION,” said Vera Bittner, MD, University of Alabama at Birmingham, as the invited discussant following Dr. Kitzman’s formal presentation of the trial. “And it certainly eclipses what we see in the typical cardiac rehab program.”
Both Dr. Bittner and Dr. Kitzman participated in HF-ACTION, a randomized exercise-training trial for patients with chronic, stable HFrEF who were all-around less sick than those in REHAB-HF.
Four functional domains
Historically, HF exercise or rehab trials have excluded patients hospitalized with acute decompensation, and third-party reimbursement often has not covered such programs because of a lack of supporting evidence and a supposed potential for harm, Dr. Kitzman said.
Entry to REHAB-HF required the patients to be fit enough to walk 4 meters, with or without a walker or other assistant device, and to have been in the hospital for at least 24 hours with a primary diagnosis of acute decompensated HF.
The intervention relied on exercises aimed at improving the four functional domains of strength, balance, mobility, and – when those three were sufficiently developed – endurance, Dr. Kitzman and associates wrote in their published report.
“The intervention was initiated in the hospital when feasible and was subsequently transitioned to an outpatient facility as soon as possible after discharge,” they wrote. Afterward, “a key goal of the intervention during the first 3 months [the outpatient phase] was to prepare the patient to transition to the independent maintenance phase (months 4-6).”
The study’s control patients “received frequent calls from study staff to try to approximate the increased attention received by the intervention group,” Dr. Kitzman said in an interview. “They were allowed to receive all usual care as ordered by their treating physicians. This included, if ordered, standard physical therapy or cardiac rehabilitation” in 43% of the control cohort. Of the trial’s 349 patients, those assigned to the intervention scored significantly higher on the three-component Short Physical Performance Battery (SPPB) at 12 weeks than those assigned to a usual care approach that included, for some, more conventional cardiac rehabilitation (8.3 vs. 6.9; P < .001).
The SPPB, validated in trials as a proxy for clinical outcomes includes tests of balance while standing, gait speed during a 4-minute walk, and strength. The latter is the test that measures time needed to rise from a chair five times.
They also showed consistent gains in other measures of physical functioning and quality of life by 12 weeks months.
The observed SPPB treatment effect is “impressive” and “compares very favorably with previously reported estimates,” observed an accompanying editorial from Stefan D. Anker, MD, PhD, of the German Center for Cardiovascular Research and Charité Universitätsmedizin, Berlin, and Andrew J.S. Coats, DM, of the University of Warwick, Coventry, England.
“Similarly, the between-group differences seen in 6-minute walk distance (34 m) and gait speed (0.12 m/s) are clinically meaningful and sizable.”
They propose that some of the substantial quality-of-life benefit in the intervention group “may be due to better physical performance, and that part may be due to improvements in psychosocial factors and mood. It appears that exercise also resulted in patients becoming happier, or at least less depressed, as evidenced by the positive results on the Geriatric Depression Scale.”
Similar results across most subgroups
In subgroup analyses, the intervention was successful against the standard-care approach in both men and women at all ages and regardless of ejection fraction; symptom status; and whether the patient had diabetes, ischemic heart disease, or atrial fibrillation, or was obese.
Clinical outcomes were not significantly different at 6 months. The rate of death from any cause was 13% for the intervention group and 10% for the control group. There were 194 and 213 hospitalizations from any cause, respectively.
Not included in the trial’s current publication but soon to be published, Dr. Kitzman said when interviewed, is a comparison of outcomes in patients with HFpEF and HFrEF. “We found at baseline that those with HFpEF had worse impairment in physical function, quality of life, and frailty. After the intervention, there appeared to be consistently larger improvements in all outcomes, including SPPB, 6-minute walk, qualify of life, and frailty, in HFpEF versus HFrEF.”
The signals of potential benefit in HFpEF extended to clinical endpoints, he said. In contrast to similar rates of all-cause rehospitalization in HFrEF, “in patients with HFpEF, rehospitalizations were 17% lower in the intervention group, compared to the control group.” Still, he noted, the interaction P value wasn’t significant.
However, Dr. Kitzman added, mortality in the intervention group, compared with the control group, was reduced by 35% among patients with HFpEF, “but was 250% higher in HFrEF,” with a significant interaction P value.
He was careful to note that, as a phase 2 trial, REHAB-HF was underpowered for clinical events, “and even the results in the HFpEF group should not be seen as adequate evidence to change clinical care.” They were from an exploratory analysis that included relatively few events.
“Because definitive demonstration of improvement in clinical events is critical for altering clinical care guidelines and for third-party payer reimbursement decisions, we believe that a subsequent phase 3 trial is needed and are currently planning toward that,” Dr. Kitzman said.
The study was supported by research grants from the National Institutes of Health, the Kermit Glenn Phillips II Chair in Cardiovascular Medicine, and the Oristano Family Fund at Wake Forest. Dr. Kitzman disclosed receiving consulting fees or honoraria from AbbVie, AstraZeneca, Bayer Healthcare, Boehringer Ingelheim, CinRx, Corviamedical, GlaxoSmithKline, and Merck; and having an unspecified relationship with Gilead. Dr. Handberg disclosed receiving grants from Aastom Biosciences, Abbott Laboratories, Amgen, Amorcyte, AstraZeneca, Biocardia, Boehringer Ingelheim, Capricor, Cytori Therapeutics, Department of Defense, Direct Flow Medical, Everyfit, Gilead, Ionis, Medtronic, Merck, Mesoblast, Relypsa, and Sanofi-Aventis. Dr. Bittner discloses receiving consulting fees or honoraria from Pfizer and Sanofi; receiving research grants from Amgen and The Medicines Company; and having unspecified relationships with AstraZeneca, DalCor, Esperion, and Sanofi-Aventis. Dr. Anker reported receiving grants and personal fees from Abbott Vascular and Vifor; personal fees from Bayer, Boehringer Ingelheim, Novartis, Servier, Cardiac Dimensions, Thermo Fisher Scientific, AstraZeneca, Occlutech, Actimed, and Respicardia. Dr. Coats disclosed receiving personal fees from AstraZeneca, Bayer, Boehringer Ingelheim, Menarini, Novartis, Nutricia, Servier, Vifor, Abbott, Actimed, Arena, Cardiac Dimensions, Corvia, CVRx, Enopace, ESN Cleer, Faraday, WL Gore, Impulse Dynamics, and Respicardia.
A version of this article first appeared on Medscape.com.
Omics analysis links blood type to COVID-19
A new analysis of gene expression and protein content in lung and blood tissue suggests that certain variants of the ABO gene, which plays a central role in determining blood type, may also influence susceptibility to COVID-19. Researchers at the University of British Columbia, Vancouver, analyzed data from three studies to link gene and protein expression in lungs and blood with genetic regions associated with COVID-19 susceptibility.
“These genes may also prove to be good markers for disease as well as potential drug targets,” said lead author Ana Hernandez Cordero, PhD, postdoctoral fellow with the Center for Heart Lung Innovation, University of British Columbia, in a statement. Dr. Cordero presented the study at the American Thoracic Society’s virtual international conference.
Dr. Cordero noted that genomewide association studies have been used to identify genetic regions associated with COVID-19 susceptibility, but they cannot be used to identify specific genes. To pinpoint genes, the researchers employed integrated genomics, which combines Bayesian colocalization summary-based Mendelian randomization and Mendelian randomization.
Searching for candidate genes
The researchers combined genetic data and transcriptomics data, which are a measurement of the messenger RNA produced in a cell. Messenger RNA is used as a blueprint for protein production. The genetics data came from the COVID-19 Host Genetics Initiative genomewide association meta-analysis version 4 (patients with COVID-19 vs. patients without COVID-19). Blood transcriptomics data came from the INTERVAL study (n = 3301), and lung transcriptomics data came from the Lung eQTL study (n = 1038). “From the integration of these three datasets we identified the candidate genes that are most likely to influence COVID-19 through gene expression. We further investigated the most consistent candidate genes and tested the causal association between their plasma protein levels and COVID-19 susceptibility using Bayesian colocalization and Mendelian randomization,” said Dr. Cordero during her talk.
Susceptibility drivers
The researchers identified six genes expressed in the lung and five expressed in blood that colocalized with COVID-19 susceptibility loci. They found that an increase in plasma levels of ABO was associated with greater risk for COVID-19 (Mendelian randomization, P = .000025) and that expression of the SLC6A20 gene in the lung was also associated with higher COVID-19 risk. They also found novel associations at genes associated with respiratory diseases, such as asthma, as well as genes associated with the host immune responses, such as neutrophil and eosinophil counts.
Possibly protective?
Within the ABO gene, the research also turned up evidence that blood type O may be protective against COVID-19. “The most significant variant used for the Mendelian randomization test was in complete linkage disagreement with the variant responsible for the blood type O genotype, conferring reduced risk,” said Dr. Cordero.
The study’s method is a powerful technique, said Jeremy Alexander Hirota, PhD, who was asked to comment. “The present study uses integrative omics to determine COVID-19 susceptibility factors which would have been challenging to identify with a single technology,” said Dr. Hirota, who is an assistant professor of medicine at McMaster University, Hamilton, Ont.; an adjunct professor of biology at the University of Waterloo (Ont.); and an affiliate professor of medicine at the University of British Columbia. He trained with the senior author of the study but was not directly involved in the research.
The host response is widely believed to be most responsible for the symptoms of COVID-19, so it isn’t surprising that host genes can be identified, according to Dr. Hirota. The identification of variants in the ABO protein is interesting, though. It suggests ‘that systemic effects beyond respiratory mucosal immunity are a driver for susceptibility.’ To my understanding, ABO protein is not expressed in the respiratory mucosa, which is a common site of first contact for SARS-CoV-2. The links between blood ABO levels and initial infection of the respiratory mucosa by SARS-CoV-2 are unclear,” he said.
Severity link needed
Dr. Hirota also said that although the study points toward associations with susceptibility to COVID-19, it isn’t clear from the available data whether such associations are related to severity of disease. “If the [patients with gene variants] are more susceptible but [the disease is] less severe, then the results need to be interpreted accordingly. If the susceptibility is increased and the severity is also increased, maybe measured by increased risk for ICU admission, ventilator use, or mortality, then the work carries a much more important message. Future studies extending this work and integrating measures of severity are warranted to better understand the clinical utility of these findings for managing COVID-19 patients optimally,” said Dr. Hirota.
It’s also unclear whether the study populations are reflective of the populations that are currently at highest risk for COVID-19, such as residents of India, where the burden of disease is currently severe.
Dr. Cordero and Dr. Hirota disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new analysis of gene expression and protein content in lung and blood tissue suggests that certain variants of the ABO gene, which plays a central role in determining blood type, may also influence susceptibility to COVID-19. Researchers at the University of British Columbia, Vancouver, analyzed data from three studies to link gene and protein expression in lungs and blood with genetic regions associated with COVID-19 susceptibility.
“These genes may also prove to be good markers for disease as well as potential drug targets,” said lead author Ana Hernandez Cordero, PhD, postdoctoral fellow with the Center for Heart Lung Innovation, University of British Columbia, in a statement. Dr. Cordero presented the study at the American Thoracic Society’s virtual international conference.
Dr. Cordero noted that genomewide association studies have been used to identify genetic regions associated with COVID-19 susceptibility, but they cannot be used to identify specific genes. To pinpoint genes, the researchers employed integrated genomics, which combines Bayesian colocalization summary-based Mendelian randomization and Mendelian randomization.
Searching for candidate genes
The researchers combined genetic data and transcriptomics data, which are a measurement of the messenger RNA produced in a cell. Messenger RNA is used as a blueprint for protein production. The genetics data came from the COVID-19 Host Genetics Initiative genomewide association meta-analysis version 4 (patients with COVID-19 vs. patients without COVID-19). Blood transcriptomics data came from the INTERVAL study (n = 3301), and lung transcriptomics data came from the Lung eQTL study (n = 1038). “From the integration of these three datasets we identified the candidate genes that are most likely to influence COVID-19 through gene expression. We further investigated the most consistent candidate genes and tested the causal association between their plasma protein levels and COVID-19 susceptibility using Bayesian colocalization and Mendelian randomization,” said Dr. Cordero during her talk.
Susceptibility drivers
The researchers identified six genes expressed in the lung and five expressed in blood that colocalized with COVID-19 susceptibility loci. They found that an increase in plasma levels of ABO was associated with greater risk for COVID-19 (Mendelian randomization, P = .000025) and that expression of the SLC6A20 gene in the lung was also associated with higher COVID-19 risk. They also found novel associations at genes associated with respiratory diseases, such as asthma, as well as genes associated with the host immune responses, such as neutrophil and eosinophil counts.
Possibly protective?
Within the ABO gene, the research also turned up evidence that blood type O may be protective against COVID-19. “The most significant variant used for the Mendelian randomization test was in complete linkage disagreement with the variant responsible for the blood type O genotype, conferring reduced risk,” said Dr. Cordero.
The study’s method is a powerful technique, said Jeremy Alexander Hirota, PhD, who was asked to comment. “The present study uses integrative omics to determine COVID-19 susceptibility factors which would have been challenging to identify with a single technology,” said Dr. Hirota, who is an assistant professor of medicine at McMaster University, Hamilton, Ont.; an adjunct professor of biology at the University of Waterloo (Ont.); and an affiliate professor of medicine at the University of British Columbia. He trained with the senior author of the study but was not directly involved in the research.
The host response is widely believed to be most responsible for the symptoms of COVID-19, so it isn’t surprising that host genes can be identified, according to Dr. Hirota. The identification of variants in the ABO protein is interesting, though. It suggests ‘that systemic effects beyond respiratory mucosal immunity are a driver for susceptibility.’ To my understanding, ABO protein is not expressed in the respiratory mucosa, which is a common site of first contact for SARS-CoV-2. The links between blood ABO levels and initial infection of the respiratory mucosa by SARS-CoV-2 are unclear,” he said.
Severity link needed
Dr. Hirota also said that although the study points toward associations with susceptibility to COVID-19, it isn’t clear from the available data whether such associations are related to severity of disease. “If the [patients with gene variants] are more susceptible but [the disease is] less severe, then the results need to be interpreted accordingly. If the susceptibility is increased and the severity is also increased, maybe measured by increased risk for ICU admission, ventilator use, or mortality, then the work carries a much more important message. Future studies extending this work and integrating measures of severity are warranted to better understand the clinical utility of these findings for managing COVID-19 patients optimally,” said Dr. Hirota.
It’s also unclear whether the study populations are reflective of the populations that are currently at highest risk for COVID-19, such as residents of India, where the burden of disease is currently severe.
Dr. Cordero and Dr. Hirota disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new analysis of gene expression and protein content in lung and blood tissue suggests that certain variants of the ABO gene, which plays a central role in determining blood type, may also influence susceptibility to COVID-19. Researchers at the University of British Columbia, Vancouver, analyzed data from three studies to link gene and protein expression in lungs and blood with genetic regions associated with COVID-19 susceptibility.
“These genes may also prove to be good markers for disease as well as potential drug targets,” said lead author Ana Hernandez Cordero, PhD, postdoctoral fellow with the Center for Heart Lung Innovation, University of British Columbia, in a statement. Dr. Cordero presented the study at the American Thoracic Society’s virtual international conference.
Dr. Cordero noted that genomewide association studies have been used to identify genetic regions associated with COVID-19 susceptibility, but they cannot be used to identify specific genes. To pinpoint genes, the researchers employed integrated genomics, which combines Bayesian colocalization summary-based Mendelian randomization and Mendelian randomization.
Searching for candidate genes
The researchers combined genetic data and transcriptomics data, which are a measurement of the messenger RNA produced in a cell. Messenger RNA is used as a blueprint for protein production. The genetics data came from the COVID-19 Host Genetics Initiative genomewide association meta-analysis version 4 (patients with COVID-19 vs. patients without COVID-19). Blood transcriptomics data came from the INTERVAL study (n = 3301), and lung transcriptomics data came from the Lung eQTL study (n = 1038). “From the integration of these three datasets we identified the candidate genes that are most likely to influence COVID-19 through gene expression. We further investigated the most consistent candidate genes and tested the causal association between their plasma protein levels and COVID-19 susceptibility using Bayesian colocalization and Mendelian randomization,” said Dr. Cordero during her talk.
Susceptibility drivers
The researchers identified six genes expressed in the lung and five expressed in blood that colocalized with COVID-19 susceptibility loci. They found that an increase in plasma levels of ABO was associated with greater risk for COVID-19 (Mendelian randomization, P = .000025) and that expression of the SLC6A20 gene in the lung was also associated with higher COVID-19 risk. They also found novel associations at genes associated with respiratory diseases, such as asthma, as well as genes associated with the host immune responses, such as neutrophil and eosinophil counts.
Possibly protective?
Within the ABO gene, the research also turned up evidence that blood type O may be protective against COVID-19. “The most significant variant used for the Mendelian randomization test was in complete linkage disagreement with the variant responsible for the blood type O genotype, conferring reduced risk,” said Dr. Cordero.
The study’s method is a powerful technique, said Jeremy Alexander Hirota, PhD, who was asked to comment. “The present study uses integrative omics to determine COVID-19 susceptibility factors which would have been challenging to identify with a single technology,” said Dr. Hirota, who is an assistant professor of medicine at McMaster University, Hamilton, Ont.; an adjunct professor of biology at the University of Waterloo (Ont.); and an affiliate professor of medicine at the University of British Columbia. He trained with the senior author of the study but was not directly involved in the research.
The host response is widely believed to be most responsible for the symptoms of COVID-19, so it isn’t surprising that host genes can be identified, according to Dr. Hirota. The identification of variants in the ABO protein is interesting, though. It suggests ‘that systemic effects beyond respiratory mucosal immunity are a driver for susceptibility.’ To my understanding, ABO protein is not expressed in the respiratory mucosa, which is a common site of first contact for SARS-CoV-2. The links between blood ABO levels and initial infection of the respiratory mucosa by SARS-CoV-2 are unclear,” he said.
Severity link needed
Dr. Hirota also said that although the study points toward associations with susceptibility to COVID-19, it isn’t clear from the available data whether such associations are related to severity of disease. “If the [patients with gene variants] are more susceptible but [the disease is] less severe, then the results need to be interpreted accordingly. If the susceptibility is increased and the severity is also increased, maybe measured by increased risk for ICU admission, ventilator use, or mortality, then the work carries a much more important message. Future studies extending this work and integrating measures of severity are warranted to better understand the clinical utility of these findings for managing COVID-19 patients optimally,” said Dr. Hirota.
It’s also unclear whether the study populations are reflective of the populations that are currently at highest risk for COVID-19, such as residents of India, where the burden of disease is currently severe.
Dr. Cordero and Dr. Hirota disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FDA preparing an environmental impact statement for 2 sunscreen ingredients
The Food and Drug Administration is launching a process to prepare an environmental impact statement (EIS) regarding the use oxybenzone and octinoxate in over-the-counter sunscreen products.
According to the “Intent to Prepare an Environmental Impact Statement for Certain Sunscreen Drug Products for Over-The-Counter Use,” which was published in the Federal Register on May 13, 2021, the FDA will prepare an EIS “when data or information in an environmental assessment or otherwise available to the Agency leads to a finding that the proposed agency action may significantly affect the quality of the human environment.” The first step in this effort involves a “public scoping process” to evaluate any potential environmental impacts associated with the use of oxybenzone and octinoxate in sunscreens so that an EIS, if required, “can be completed prior to issuance of a final sunscreen order addressing sunscreens containing these ingredients.”
The American Academy of Dermatology Association weighed in on the FDA’s announcement, noting that it “appreciates the efforts of the agency to thoroughly examine all relevant science before issuing a final sunscreen order on these ingredients,” according to a statement released by the AADA on May 13, 2021.
The statement added: “Skin cancer is the most common cancer in the U.S., and unprotected exposure to the sun’s harmful ultraviolet rays is a major risk factor. The AADA continues to focus on encouraging members of the public to protect themselves by seeking shade, wearing protective clothing – including a lightweight and long-sleeved shirt, pants, a wide-brimmed hat and sunglasses – and applying a broad-spectrum sunscreen with an SPF of 30 or higher to all exposed skin.”
According to the FDA document, a series of developments regarding oxybenzone and octinoxate prompted the agency to take this step, including comments the agency received in response to the 2019 proposed rule titled “Sunscreen Drug Products for Over-The-Counter Human Use,” which raised concern about the potential effects of the two ingredients on coral and/or coral reefs, as well as research efforts by the National Oceanic and Atmospheric Administration Coral Reef Conservation Programs on the potential impacts of sunscreen products that include oxybenzone and octinoxate on coral reefs and other aquatic systems. Hawaii’s 2018 state law prohibiting the sale, offer of sale, and distribution of sunscreens that contain oxybenzone and/or octinoxate also influenced the agency’s decision to further evaluate the topic.
“The purpose of the public scoping process is to determine relevant issues that will influence the scope of the environmental analysis, including potential alternatives and the extent to which those issues and impacts will be analyzed,” the FDA document states. “At this initial stage of the scoping process, we have identified the following four alternatives: FDA will conclude that the inclusion of oxybenzone and octinoxate in sunscreens marketed without an NDA [new drug application] is impermissible; FDA will conclude that the inclusion of oxybenzone and octinoxate in sunscreens marketed without an NDA is permissible; FDA will conclude that inclusion of oxybenzone in sunscreens marketed without an NDA is permissible but that the inclusion of octinoxate in sunscreens marketed without an NDA is impermissible; or FDA will conclude that inclusion of octinoxate in sunscreens marketed without an NDA is permissible but that the inclusion of oxybenzone in sunscreens marketed without an NDA is impermissible.”
Until June 14, the FDA is accepting comments from the public electronically via the Federal eRulemaking Portal at www.regulations.gov (search for Docket No. FDA-2021-N-0352) or by mail to: Dockets Management Staff (HFA-305), Food and Drug Administration, 5630 Fishers Lane, Rm. 1061, Rockville, Md., 20852. Refer to Docket No. FDA-2021-N-0352.
The Food and Drug Administration is launching a process to prepare an environmental impact statement (EIS) regarding the use oxybenzone and octinoxate in over-the-counter sunscreen products.
According to the “Intent to Prepare an Environmental Impact Statement for Certain Sunscreen Drug Products for Over-The-Counter Use,” which was published in the Federal Register on May 13, 2021, the FDA will prepare an EIS “when data or information in an environmental assessment or otherwise available to the Agency leads to a finding that the proposed agency action may significantly affect the quality of the human environment.” The first step in this effort involves a “public scoping process” to evaluate any potential environmental impacts associated with the use of oxybenzone and octinoxate in sunscreens so that an EIS, if required, “can be completed prior to issuance of a final sunscreen order addressing sunscreens containing these ingredients.”
The American Academy of Dermatology Association weighed in on the FDA’s announcement, noting that it “appreciates the efforts of the agency to thoroughly examine all relevant science before issuing a final sunscreen order on these ingredients,” according to a statement released by the AADA on May 13, 2021.
The statement added: “Skin cancer is the most common cancer in the U.S., and unprotected exposure to the sun’s harmful ultraviolet rays is a major risk factor. The AADA continues to focus on encouraging members of the public to protect themselves by seeking shade, wearing protective clothing – including a lightweight and long-sleeved shirt, pants, a wide-brimmed hat and sunglasses – and applying a broad-spectrum sunscreen with an SPF of 30 or higher to all exposed skin.”
According to the FDA document, a series of developments regarding oxybenzone and octinoxate prompted the agency to take this step, including comments the agency received in response to the 2019 proposed rule titled “Sunscreen Drug Products for Over-The-Counter Human Use,” which raised concern about the potential effects of the two ingredients on coral and/or coral reefs, as well as research efforts by the National Oceanic and Atmospheric Administration Coral Reef Conservation Programs on the potential impacts of sunscreen products that include oxybenzone and octinoxate on coral reefs and other aquatic systems. Hawaii’s 2018 state law prohibiting the sale, offer of sale, and distribution of sunscreens that contain oxybenzone and/or octinoxate also influenced the agency’s decision to further evaluate the topic.
“The purpose of the public scoping process is to determine relevant issues that will influence the scope of the environmental analysis, including potential alternatives and the extent to which those issues and impacts will be analyzed,” the FDA document states. “At this initial stage of the scoping process, we have identified the following four alternatives: FDA will conclude that the inclusion of oxybenzone and octinoxate in sunscreens marketed without an NDA [new drug application] is impermissible; FDA will conclude that the inclusion of oxybenzone and octinoxate in sunscreens marketed without an NDA is permissible; FDA will conclude that inclusion of oxybenzone in sunscreens marketed without an NDA is permissible but that the inclusion of octinoxate in sunscreens marketed without an NDA is impermissible; or FDA will conclude that inclusion of octinoxate in sunscreens marketed without an NDA is permissible but that the inclusion of oxybenzone in sunscreens marketed without an NDA is impermissible.”
Until June 14, the FDA is accepting comments from the public electronically via the Federal eRulemaking Portal at www.regulations.gov (search for Docket No. FDA-2021-N-0352) or by mail to: Dockets Management Staff (HFA-305), Food and Drug Administration, 5630 Fishers Lane, Rm. 1061, Rockville, Md., 20852. Refer to Docket No. FDA-2021-N-0352.
The Food and Drug Administration is launching a process to prepare an environmental impact statement (EIS) regarding the use oxybenzone and octinoxate in over-the-counter sunscreen products.
According to the “Intent to Prepare an Environmental Impact Statement for Certain Sunscreen Drug Products for Over-The-Counter Use,” which was published in the Federal Register on May 13, 2021, the FDA will prepare an EIS “when data or information in an environmental assessment or otherwise available to the Agency leads to a finding that the proposed agency action may significantly affect the quality of the human environment.” The first step in this effort involves a “public scoping process” to evaluate any potential environmental impacts associated with the use of oxybenzone and octinoxate in sunscreens so that an EIS, if required, “can be completed prior to issuance of a final sunscreen order addressing sunscreens containing these ingredients.”
The American Academy of Dermatology Association weighed in on the FDA’s announcement, noting that it “appreciates the efforts of the agency to thoroughly examine all relevant science before issuing a final sunscreen order on these ingredients,” according to a statement released by the AADA on May 13, 2021.
The statement added: “Skin cancer is the most common cancer in the U.S., and unprotected exposure to the sun’s harmful ultraviolet rays is a major risk factor. The AADA continues to focus on encouraging members of the public to protect themselves by seeking shade, wearing protective clothing – including a lightweight and long-sleeved shirt, pants, a wide-brimmed hat and sunglasses – and applying a broad-spectrum sunscreen with an SPF of 30 or higher to all exposed skin.”
According to the FDA document, a series of developments regarding oxybenzone and octinoxate prompted the agency to take this step, including comments the agency received in response to the 2019 proposed rule titled “Sunscreen Drug Products for Over-The-Counter Human Use,” which raised concern about the potential effects of the two ingredients on coral and/or coral reefs, as well as research efforts by the National Oceanic and Atmospheric Administration Coral Reef Conservation Programs on the potential impacts of sunscreen products that include oxybenzone and octinoxate on coral reefs and other aquatic systems. Hawaii’s 2018 state law prohibiting the sale, offer of sale, and distribution of sunscreens that contain oxybenzone and/or octinoxate also influenced the agency’s decision to further evaluate the topic.
“The purpose of the public scoping process is to determine relevant issues that will influence the scope of the environmental analysis, including potential alternatives and the extent to which those issues and impacts will be analyzed,” the FDA document states. “At this initial stage of the scoping process, we have identified the following four alternatives: FDA will conclude that the inclusion of oxybenzone and octinoxate in sunscreens marketed without an NDA [new drug application] is impermissible; FDA will conclude that the inclusion of oxybenzone and octinoxate in sunscreens marketed without an NDA is permissible; FDA will conclude that inclusion of oxybenzone in sunscreens marketed without an NDA is permissible but that the inclusion of octinoxate in sunscreens marketed without an NDA is impermissible; or FDA will conclude that inclusion of octinoxate in sunscreens marketed without an NDA is permissible but that the inclusion of oxybenzone in sunscreens marketed without an NDA is impermissible.”
Until June 14, the FDA is accepting comments from the public electronically via the Federal eRulemaking Portal at www.regulations.gov (search for Docket No. FDA-2021-N-0352) or by mail to: Dockets Management Staff (HFA-305), Food and Drug Administration, 5630 Fishers Lane, Rm. 1061, Rockville, Md., 20852. Refer to Docket No. FDA-2021-N-0352.
Pediatricians see drop in income during the pandemic
The average income for pediatricians declined slightly from 2019 to 2020, according to the Medscape Pediatrician Compensation Report 2021.

The report, which was conducted between October 2020 and February 2021, found that the average pediatrician income was down $11,000 – from $232,000 in 2019 to $221,000 in 2020, with 48% of pediatricians reporting at least some decline in compensation.
The specialty also earned the least amount of money in 2020, compared with all of the other specialties, which isn’t surprising since pediatricians have been among the lowest-paid physician specialties since 2013. The highest-earning specialty was plastic surgery with an average income of $526,000 annually.
Most pediatricians who saw a drop in income cited pandemic-related issues such as job loss, fewer hours, and fewer patients.
Jesse Hackell, MD, vice president and chief operating officer of Ponoma Pediatrics in New York, said in an interview the reduced wages pediatricians saw in 2020 didn’t surprise him because many pediatric offices saw a huge drop in visits that were not urgent.
“[The report] shows that procedural specialties tended to do a lot better than the nonprocedural specialties,” Dr. Hackell said. “That’s because, during the shutdown, if you broke your leg, you still needed the orthopedist. And even though the hospitals weren’t doing elective surgeries, they were certainly doing the emergency stuff.”
Meanwhile, in pediatrician offices, where Dr. Hackell said office visits dropped 70%-80% at the beginning of the pandemic, “parents weren’t going to bring a healthy kid out for routine visits and they weren’t going to bring a kid out for minor illnesses and expose them to possibly communicable diseases in the office.”
About 52% of pediatricians who lost income because of the pandemic believe their income levels will return to normal in 2-3 years. Meanwhile 30% of pediatricians expect their income to return to normal within a year, and 8% believe it will take 4 years for them to bounce back.
Physician work hours generally declined for some time during the pandemic, according to the report. However, most pediatricians are working about the same number of hours as they did before the pandemic, which is 47 hours per week.
Despite working the same number of hours per week that they did prepandemic, they are seeing fewer patients. They are currently seeing on average 64 patients per week, compared with the 78 patients they used to see weekly before the pandemic.
Dr. Hackell said that might be because pediatric offices are trying to make up the loss of revenue during the beginning of the pandemic, from the reduced number of well visits and immunizations, in the second half of the year with outreach.
“Since about June 2020, we’ve been making concerted efforts to remind parents that preventing other infectious diseases is critically important,” Dr. Hackell explained. “And so actually, for the second half of the year, many of us saw more well visits and immunization volume than in 2019 as we sought to make up the gap. It wasn’t that we were seeing more overall, but we’re trying to make up the gap that happened from March, April, May, [and] June.”
Most pediatricians find their work rewarding. One-third say the most rewarding part of their job is gratitude from and relationships with their patients. Meanwhile, 31% of pediatricians said knowing they are making the world a better place was a rewarding part of their job. Only 8% of them said making money was a rewarding part of their job.
Dr. Hackell said he did not go into pediatrics to make money, it was because he found it stimulating and has “no complaints.”
“I’ve been a pediatrician for 40 years and I wouldn’t do anything else,” Dr. Hackell said. “I don’t know that there’s anything that I would find as rewarding as the relationships that I’ve had over 40 years with my patients. You know, getting invited to weddings of kids who I saw when they were newborns is pretty impressive. It’s the gratification of having ongoing relationships with families.”
Furthermore, the report revealed that 77% of pediatricians said they would pick medicine again if they had a choice, and 82% said they would choose the same specialty.
The experts disclosed no relevant financial interests.
*This story was updated on 5/18/2021.
The average income for pediatricians declined slightly from 2019 to 2020, according to the Medscape Pediatrician Compensation Report 2021.

The report, which was conducted between October 2020 and February 2021, found that the average pediatrician income was down $11,000 – from $232,000 in 2019 to $221,000 in 2020, with 48% of pediatricians reporting at least some decline in compensation.
The specialty also earned the least amount of money in 2020, compared with all of the other specialties, which isn’t surprising since pediatricians have been among the lowest-paid physician specialties since 2013. The highest-earning specialty was plastic surgery with an average income of $526,000 annually.
Most pediatricians who saw a drop in income cited pandemic-related issues such as job loss, fewer hours, and fewer patients.
Jesse Hackell, MD, vice president and chief operating officer of Ponoma Pediatrics in New York, said in an interview the reduced wages pediatricians saw in 2020 didn’t surprise him because many pediatric offices saw a huge drop in visits that were not urgent.
“[The report] shows that procedural specialties tended to do a lot better than the nonprocedural specialties,” Dr. Hackell said. “That’s because, during the shutdown, if you broke your leg, you still needed the orthopedist. And even though the hospitals weren’t doing elective surgeries, they were certainly doing the emergency stuff.”
Meanwhile, in pediatrician offices, where Dr. Hackell said office visits dropped 70%-80% at the beginning of the pandemic, “parents weren’t going to bring a healthy kid out for routine visits and they weren’t going to bring a kid out for minor illnesses and expose them to possibly communicable diseases in the office.”
About 52% of pediatricians who lost income because of the pandemic believe their income levels will return to normal in 2-3 years. Meanwhile 30% of pediatricians expect their income to return to normal within a year, and 8% believe it will take 4 years for them to bounce back.
Physician work hours generally declined for some time during the pandemic, according to the report. However, most pediatricians are working about the same number of hours as they did before the pandemic, which is 47 hours per week.
Despite working the same number of hours per week that they did prepandemic, they are seeing fewer patients. They are currently seeing on average 64 patients per week, compared with the 78 patients they used to see weekly before the pandemic.
Dr. Hackell said that might be because pediatric offices are trying to make up the loss of revenue during the beginning of the pandemic, from the reduced number of well visits and immunizations, in the second half of the year with outreach.
“Since about June 2020, we’ve been making concerted efforts to remind parents that preventing other infectious diseases is critically important,” Dr. Hackell explained. “And so actually, for the second half of the year, many of us saw more well visits and immunization volume than in 2019 as we sought to make up the gap. It wasn’t that we were seeing more overall, but we’re trying to make up the gap that happened from March, April, May, [and] June.”
Most pediatricians find their work rewarding. One-third say the most rewarding part of their job is gratitude from and relationships with their patients. Meanwhile, 31% of pediatricians said knowing they are making the world a better place was a rewarding part of their job. Only 8% of them said making money was a rewarding part of their job.
Dr. Hackell said he did not go into pediatrics to make money, it was because he found it stimulating and has “no complaints.”
“I’ve been a pediatrician for 40 years and I wouldn’t do anything else,” Dr. Hackell said. “I don’t know that there’s anything that I would find as rewarding as the relationships that I’ve had over 40 years with my patients. You know, getting invited to weddings of kids who I saw when they were newborns is pretty impressive. It’s the gratification of having ongoing relationships with families.”
Furthermore, the report revealed that 77% of pediatricians said they would pick medicine again if they had a choice, and 82% said they would choose the same specialty.
The experts disclosed no relevant financial interests.
*This story was updated on 5/18/2021.
The average income for pediatricians declined slightly from 2019 to 2020, according to the Medscape Pediatrician Compensation Report 2021.

The report, which was conducted between October 2020 and February 2021, found that the average pediatrician income was down $11,000 – from $232,000 in 2019 to $221,000 in 2020, with 48% of pediatricians reporting at least some decline in compensation.
The specialty also earned the least amount of money in 2020, compared with all of the other specialties, which isn’t surprising since pediatricians have been among the lowest-paid physician specialties since 2013. The highest-earning specialty was plastic surgery with an average income of $526,000 annually.
Most pediatricians who saw a drop in income cited pandemic-related issues such as job loss, fewer hours, and fewer patients.
Jesse Hackell, MD, vice president and chief operating officer of Ponoma Pediatrics in New York, said in an interview the reduced wages pediatricians saw in 2020 didn’t surprise him because many pediatric offices saw a huge drop in visits that were not urgent.
“[The report] shows that procedural specialties tended to do a lot better than the nonprocedural specialties,” Dr. Hackell said. “That’s because, during the shutdown, if you broke your leg, you still needed the orthopedist. And even though the hospitals weren’t doing elective surgeries, they were certainly doing the emergency stuff.”
Meanwhile, in pediatrician offices, where Dr. Hackell said office visits dropped 70%-80% at the beginning of the pandemic, “parents weren’t going to bring a healthy kid out for routine visits and they weren’t going to bring a kid out for minor illnesses and expose them to possibly communicable diseases in the office.”
About 52% of pediatricians who lost income because of the pandemic believe their income levels will return to normal in 2-3 years. Meanwhile 30% of pediatricians expect their income to return to normal within a year, and 8% believe it will take 4 years for them to bounce back.
Physician work hours generally declined for some time during the pandemic, according to the report. However, most pediatricians are working about the same number of hours as they did before the pandemic, which is 47 hours per week.
Despite working the same number of hours per week that they did prepandemic, they are seeing fewer patients. They are currently seeing on average 64 patients per week, compared with the 78 patients they used to see weekly before the pandemic.
Dr. Hackell said that might be because pediatric offices are trying to make up the loss of revenue during the beginning of the pandemic, from the reduced number of well visits and immunizations, in the second half of the year with outreach.
“Since about June 2020, we’ve been making concerted efforts to remind parents that preventing other infectious diseases is critically important,” Dr. Hackell explained. “And so actually, for the second half of the year, many of us saw more well visits and immunization volume than in 2019 as we sought to make up the gap. It wasn’t that we were seeing more overall, but we’re trying to make up the gap that happened from March, April, May, [and] June.”
Most pediatricians find their work rewarding. One-third say the most rewarding part of their job is gratitude from and relationships with their patients. Meanwhile, 31% of pediatricians said knowing they are making the world a better place was a rewarding part of their job. Only 8% of them said making money was a rewarding part of their job.
Dr. Hackell said he did not go into pediatrics to make money, it was because he found it stimulating and has “no complaints.”
“I’ve been a pediatrician for 40 years and I wouldn’t do anything else,” Dr. Hackell said. “I don’t know that there’s anything that I would find as rewarding as the relationships that I’ve had over 40 years with my patients. You know, getting invited to weddings of kids who I saw when they were newborns is pretty impressive. It’s the gratification of having ongoing relationships with families.”
Furthermore, the report revealed that 77% of pediatricians said they would pick medicine again if they had a choice, and 82% said they would choose the same specialty.
The experts disclosed no relevant financial interests.
*This story was updated on 5/18/2021.
Are more naturopaths trying to compete with docs?
Jon Hislop, MD, PhD, hadn’t been in practice very long before patients began coming to him with requests to order tests that their naturopaths had recommended.
The family physician in North Vancouver, British Columbia, knew little about naturopathy but began researching it.
“I was finding that some of what the naturopaths were telling them was a little odd. Some of the tests they were asking for were unnecessary,” Dr. Hislop said.
The more he learned about naturopathy, the more appalled he became. He eventually took to Twitter, where he wages a campaign against naturopathy and alternative medicine.
“There is no alternative medicine,” he said. “There’s medicine and there’s other stuff. We need to stick to medicine and stay away from the other stuff.”
Dr. Hislop is not alone in his criticism of naturopathic medicine. Professional medical societies almost universally oppose naturopathy, but that has not stopped its spread or prevented it from becoming part of some health care systems.
Americans spent $30.2 billion on out-of-pocket complementary health care, according to a 2016 report from the National Institutes of Health. That includes everything from herbal supplements and massage therapy to chiropractic care.
What is naturopathic medicine?
Naturopathy came to the United States from Germany in the 1800s, but some of its practices are thousands of years old. Naturopathic treatments include homeopathy, IV vitamin infusions, acupuncture, Reiki, and herbal supplements.
Naturopathy is based on the belief that the body has an innate ability to heal itself. It discourages drugs and surgery in favor of supplements, herbs, and other so-called natural treatments. Much of it centers around addressing lifestyle issues and counseling patients to improve their diets, quit smoking, exercise more, lose weight, etc., in order to address the root causes of some health problems.
Practitioners are critical of Western medicine for what they regard as an over-reliance on drugs and technology and for treating symptoms rather than the causes of disease.
“We get a lot of people who are at the end of their ropes, people with hard-to-diagnose diseases who know they are sick but whose labs are normal,” said Jaquel Patterson, ND, former president of the American Association of Naturopathic Physicians (AANP) and medical director of a naturopathic practice in Connecticut.
Separate training and licensing
There are major differences among naturopaths.
At one extreme are unlicensed, self-taught “healers,” who can embrace everything from homeopathy to aromatherapy.
At the other end are naturopathic doctors (NDs), who are more likely to become part of health care systems. These caregivers are trained and licensed, though not by the same institutions as traditional physicians.
To be licensed, NDs must graduate from one of seven accredited naturopathic medical schools in the United States and Canada. In addition to a standard medical curriculum, schools require graduates to complete 4 years of training in clinical nutrition, acupuncture, homeopathic medicine, botanical medicine, physical medicine, and counseling. Medical students intern in clinical settings for 2 years.
NDs are eager to distinguish themselves from their uncredentialed counterparts.
“Some people go to a weekend class and call themselves naturopaths. That’s very concerning. I don’t want those people to be licensed either,” said Hallie Armstrong, ND, who practices in Michigan.
In the United States, there are 6,000 practicing NDs and an unknown number of unlicensed naturopathic healers.
Can naturopaths call themselves ‘physicians’?
Twenty-two states, the District of Columbia, Puerto Rico, and the U.S. Virgin Islands have licensing or registration laws for naturopathic doctors. Three states – South Carolina, Tennessee, and Florida – prohibit practicing naturopathic medicine without a license, according to the AANP.
States that license NDs differ in what they permit them to do.
Nine states allow licensed NDs to use the term “physician,” although this is prohibited in seven states. Most licensed states allow naturopathic practitioners some prescribing authority, including the prescribing of many controlled substances, although only a few states permit full prescribing rights. Most states that license NDs allow them to prescribe and administer nonprescription therapeutic substances, drugs, and therapies.
Twelve states and the District of Columbia allow licensed naturopathic doctors to perform some minor procedures, such as stitching up wounds. Additionally, 13 states allow NDs to order diagnostic tests.
Although the AANP lobbies to get licensure in more states and to expand the activities that NDs can perform, the medical establishment in those states nearly always opposes the legislation, as do national organizations, such as the American Academy of Family Physicians and the American College of Physicians.
“They absolutely will not stop until they get licenses. They’ve done a really good job of selling themselves as legitimate health care professionals to state legislatures,” said David Gorski, MD, PhD, FACS, a surgical oncologist and managing editor of Science-Based Medicine, a blog that attacks unproven medical claims and defends traditional medicine. Naturopathy is a favorite target.
Are naturopaths gaining ground anyway?
Despite the opposition of the medical establishment and many individual health care professionals, a growing number of health care systems are adopting alternative medicine.
In 2018, the AANP stated that 28 prominent health systems, hospitals, and cancer treatment centers had one or more licensed NDs on staff. Among them were Cancer Treatment Centers of America, Cedars-Sinai, Columbia University’s Herbert Irving Comprehensive Cancer Center, and the Fred Hutchinson Cancer Research Center.
Other health care systems may not have NDs on staff but provide naturopathic treatments, usually under the heading of “complementary medicine” or “integrative medicine.” For example, the Cleveland Clinic’s Center for Integrative and Lifestyle Medicine offers acupuncture, Chinese herbal medicine, Reiki, yoga, and culinary medicine.
Critics find this appalling.
“I think it’s a mistake to integrate that kind of practice into a science-based health care setting. If we learned anything over the past year, it’s that medicine based on magical thinking is dangerous,” said Timothy Caulfield, LLM, FCAHS, research director at the Health Law Institute of the University of Alberta, Edmonton.
Dr. Gorski added: “I’m not exactly sure why doctors who should know better have become more accepting of practices that aren’t science-based or are outright quackery.”
Becoming part of the system
Beaumont Health, Michigan’s largest health care system, added integrative medicine in 2006 and hired its first naturopathic practitioners a year later.
The integrative practitioners began in oncology, offering such things as massage therapy, acupuncture, guided imagery, and Reiki. “Very quickly, people outside oncology began saying, ‘I’ve got a cardiology patient who would really benefit from this ... I’ve got a GI patient who could benefit from this...,’” said Maureen Anderson, MD, medical director of Beaumont Integrative Medicine.
Beaumont now offers integrative medicine at three locations. They average 20,000 visits a year and work with 50 to 60 practitioners, many of whom work part-time.
Because Michigan does not license NDs, their scope of practice at Beaumont is limited. They take patient histories, provide advice on nutrition, diet, and exercise, and prescribe herbs and supplements. Beaumont operates its own herbal and supplement pharmacy.
NDs work under the medical supervision of Dr. Anderson, an emergency medicine physician who became interested in naturopathy because she thought traditional medicine doesn’t do a good job of providing care for chronic conditions. Any initial skepticism on the part of the medical staff has been overcome by seeing the benefits naturopathy provides, Dr. Anderson said. The claim is echoed by Mr. Armstrong, an ND who works in the system part-time: “As soon as [doctors] understand our schooling and where we’re coming from and understand that we want to do the same things, then they’re very accepting.”
The University of California, Irvine, health care system has one of the largest naturopathic medicine programs in the country, the result of a $200 million donation in 2017 from a couple who champion alternative medicine. The Susan Samueli Integrative Health Institute includes 28 health care professionals, including MDs, NDs, RNs, acupuncturists, dietitians, yoga instructors, and others. It includes a research arm, which is focused primarily on acupuncture.
The alternative medicine offerings benefit the system, said Kim Hecht, DO, medical director of inpatient and ambulatory services at the Samueli Institute.
“I’m not against traditional medicine, because I think everything has a time and a place,” Dr. Hecht said. However, she rejects the idea that MDs can offer the same holistic approach as NDs.
“Medical science likes to say we’re interested in treating the whole person, but if you look at medical school courses, that’s not what’s being taught,” she said.
The chance to work within a traditional health care system was attractive to Arvin Jenab, ND, medical director of naturopathic medicine at the institute.
“It offers the opportunity to refine our medicine and trim the things that aren’t necessary or are controversial and concentrate on the things at the core of what we do,” he said.
UCI Health practices a conservative model of naturopathy that supports traditional practitioners, Mr. Jenab said.
Is there any harm?
Some patients clearly want what naturopathy offers. So what’s the harm?
Health care systems that integrate alternative medicine legitimize it and lower the overall standard of care, Mr. Caulfield said. Most naturopathy claims are not backed by evidence, and making it available to patients amounts to deceiving them, he said.
“If there’s good science behind it, it’s not going to be alternative medicine; it’s going to be medicine,” Mr. Caulfield said.
Family physician Dr. Hislop said that refusing to order naturopath-recommended tests interferes with his relationships with patients and often requires lengthy conversations to explain the problems with naturopathy.
Naturopathic medicine can deter patients from seeking proven conventional treatments, which can put their health at risk, Dr. Gorski said.
Some naturopaths could potentially be harmful.
In 2017, a California woman died after receiving an IV preparation of curcumin, a chemical constituent in the Indian spice turmeric featured in alternative medicine. The U.S. Food and Drug Administration found that the treating ND mixed the curcumin emulsion product with ungraded castor oil that had a warning label stating: “CAUTION: For manufacturing or laboratory use only.”
Because naturopathic care is generally not covered by insurance, it can also be expensive for patients who pay out of pocket.
Ironically, the mainstream health care system helps create the environment in which naturopathic medicine thrives.
It offers patients a more relaxed and personal alternative to rushed visits with harried doctors scrambling to see the required number of patients in a day. By contrast, an initial visit with an ND might last a leisurely 60 minutes, with 30-minute follow-up appointments.
Mr. Caulfield acknowledged that the relaxed naturopathic approach can be more attractive to patients but said the answer is to reform the current system: “You don’t fix a broken arm by acupuncture.”
A version of this article first appeared on Medscape.com.
Jon Hislop, MD, PhD, hadn’t been in practice very long before patients began coming to him with requests to order tests that their naturopaths had recommended.
The family physician in North Vancouver, British Columbia, knew little about naturopathy but began researching it.
“I was finding that some of what the naturopaths were telling them was a little odd. Some of the tests they were asking for were unnecessary,” Dr. Hislop said.
The more he learned about naturopathy, the more appalled he became. He eventually took to Twitter, where he wages a campaign against naturopathy and alternative medicine.
“There is no alternative medicine,” he said. “There’s medicine and there’s other stuff. We need to stick to medicine and stay away from the other stuff.”
Dr. Hislop is not alone in his criticism of naturopathic medicine. Professional medical societies almost universally oppose naturopathy, but that has not stopped its spread or prevented it from becoming part of some health care systems.
Americans spent $30.2 billion on out-of-pocket complementary health care, according to a 2016 report from the National Institutes of Health. That includes everything from herbal supplements and massage therapy to chiropractic care.
What is naturopathic medicine?
Naturopathy came to the United States from Germany in the 1800s, but some of its practices are thousands of years old. Naturopathic treatments include homeopathy, IV vitamin infusions, acupuncture, Reiki, and herbal supplements.
Naturopathy is based on the belief that the body has an innate ability to heal itself. It discourages drugs and surgery in favor of supplements, herbs, and other so-called natural treatments. Much of it centers around addressing lifestyle issues and counseling patients to improve their diets, quit smoking, exercise more, lose weight, etc., in order to address the root causes of some health problems.
Practitioners are critical of Western medicine for what they regard as an over-reliance on drugs and technology and for treating symptoms rather than the causes of disease.
“We get a lot of people who are at the end of their ropes, people with hard-to-diagnose diseases who know they are sick but whose labs are normal,” said Jaquel Patterson, ND, former president of the American Association of Naturopathic Physicians (AANP) and medical director of a naturopathic practice in Connecticut.
Separate training and licensing
There are major differences among naturopaths.
At one extreme are unlicensed, self-taught “healers,” who can embrace everything from homeopathy to aromatherapy.
At the other end are naturopathic doctors (NDs), who are more likely to become part of health care systems. These caregivers are trained and licensed, though not by the same institutions as traditional physicians.
To be licensed, NDs must graduate from one of seven accredited naturopathic medical schools in the United States and Canada. In addition to a standard medical curriculum, schools require graduates to complete 4 years of training in clinical nutrition, acupuncture, homeopathic medicine, botanical medicine, physical medicine, and counseling. Medical students intern in clinical settings for 2 years.
NDs are eager to distinguish themselves from their uncredentialed counterparts.
“Some people go to a weekend class and call themselves naturopaths. That’s very concerning. I don’t want those people to be licensed either,” said Hallie Armstrong, ND, who practices in Michigan.
In the United States, there are 6,000 practicing NDs and an unknown number of unlicensed naturopathic healers.
Can naturopaths call themselves ‘physicians’?
Twenty-two states, the District of Columbia, Puerto Rico, and the U.S. Virgin Islands have licensing or registration laws for naturopathic doctors. Three states – South Carolina, Tennessee, and Florida – prohibit practicing naturopathic medicine without a license, according to the AANP.
States that license NDs differ in what they permit them to do.
Nine states allow licensed NDs to use the term “physician,” although this is prohibited in seven states. Most licensed states allow naturopathic practitioners some prescribing authority, including the prescribing of many controlled substances, although only a few states permit full prescribing rights. Most states that license NDs allow them to prescribe and administer nonprescription therapeutic substances, drugs, and therapies.
Twelve states and the District of Columbia allow licensed naturopathic doctors to perform some minor procedures, such as stitching up wounds. Additionally, 13 states allow NDs to order diagnostic tests.
Although the AANP lobbies to get licensure in more states and to expand the activities that NDs can perform, the medical establishment in those states nearly always opposes the legislation, as do national organizations, such as the American Academy of Family Physicians and the American College of Physicians.
“They absolutely will not stop until they get licenses. They’ve done a really good job of selling themselves as legitimate health care professionals to state legislatures,” said David Gorski, MD, PhD, FACS, a surgical oncologist and managing editor of Science-Based Medicine, a blog that attacks unproven medical claims and defends traditional medicine. Naturopathy is a favorite target.
Are naturopaths gaining ground anyway?
Despite the opposition of the medical establishment and many individual health care professionals, a growing number of health care systems are adopting alternative medicine.
In 2018, the AANP stated that 28 prominent health systems, hospitals, and cancer treatment centers had one or more licensed NDs on staff. Among them were Cancer Treatment Centers of America, Cedars-Sinai, Columbia University’s Herbert Irving Comprehensive Cancer Center, and the Fred Hutchinson Cancer Research Center.
Other health care systems may not have NDs on staff but provide naturopathic treatments, usually under the heading of “complementary medicine” or “integrative medicine.” For example, the Cleveland Clinic’s Center for Integrative and Lifestyle Medicine offers acupuncture, Chinese herbal medicine, Reiki, yoga, and culinary medicine.
Critics find this appalling.
“I think it’s a mistake to integrate that kind of practice into a science-based health care setting. If we learned anything over the past year, it’s that medicine based on magical thinking is dangerous,” said Timothy Caulfield, LLM, FCAHS, research director at the Health Law Institute of the University of Alberta, Edmonton.
Dr. Gorski added: “I’m not exactly sure why doctors who should know better have become more accepting of practices that aren’t science-based or are outright quackery.”
Becoming part of the system
Beaumont Health, Michigan’s largest health care system, added integrative medicine in 2006 and hired its first naturopathic practitioners a year later.
The integrative practitioners began in oncology, offering such things as massage therapy, acupuncture, guided imagery, and Reiki. “Very quickly, people outside oncology began saying, ‘I’ve got a cardiology patient who would really benefit from this ... I’ve got a GI patient who could benefit from this...,’” said Maureen Anderson, MD, medical director of Beaumont Integrative Medicine.
Beaumont now offers integrative medicine at three locations. They average 20,000 visits a year and work with 50 to 60 practitioners, many of whom work part-time.
Because Michigan does not license NDs, their scope of practice at Beaumont is limited. They take patient histories, provide advice on nutrition, diet, and exercise, and prescribe herbs and supplements. Beaumont operates its own herbal and supplement pharmacy.
NDs work under the medical supervision of Dr. Anderson, an emergency medicine physician who became interested in naturopathy because she thought traditional medicine doesn’t do a good job of providing care for chronic conditions. Any initial skepticism on the part of the medical staff has been overcome by seeing the benefits naturopathy provides, Dr. Anderson said. The claim is echoed by Mr. Armstrong, an ND who works in the system part-time: “As soon as [doctors] understand our schooling and where we’re coming from and understand that we want to do the same things, then they’re very accepting.”
The University of California, Irvine, health care system has one of the largest naturopathic medicine programs in the country, the result of a $200 million donation in 2017 from a couple who champion alternative medicine. The Susan Samueli Integrative Health Institute includes 28 health care professionals, including MDs, NDs, RNs, acupuncturists, dietitians, yoga instructors, and others. It includes a research arm, which is focused primarily on acupuncture.
The alternative medicine offerings benefit the system, said Kim Hecht, DO, medical director of inpatient and ambulatory services at the Samueli Institute.
“I’m not against traditional medicine, because I think everything has a time and a place,” Dr. Hecht said. However, she rejects the idea that MDs can offer the same holistic approach as NDs.
“Medical science likes to say we’re interested in treating the whole person, but if you look at medical school courses, that’s not what’s being taught,” she said.
The chance to work within a traditional health care system was attractive to Arvin Jenab, ND, medical director of naturopathic medicine at the institute.
“It offers the opportunity to refine our medicine and trim the things that aren’t necessary or are controversial and concentrate on the things at the core of what we do,” he said.
UCI Health practices a conservative model of naturopathy that supports traditional practitioners, Mr. Jenab said.
Is there any harm?
Some patients clearly want what naturopathy offers. So what’s the harm?
Health care systems that integrate alternative medicine legitimize it and lower the overall standard of care, Mr. Caulfield said. Most naturopathy claims are not backed by evidence, and making it available to patients amounts to deceiving them, he said.
“If there’s good science behind it, it’s not going to be alternative medicine; it’s going to be medicine,” Mr. Caulfield said.
Family physician Dr. Hislop said that refusing to order naturopath-recommended tests interferes with his relationships with patients and often requires lengthy conversations to explain the problems with naturopathy.
Naturopathic medicine can deter patients from seeking proven conventional treatments, which can put their health at risk, Dr. Gorski said.
Some naturopaths could potentially be harmful.
In 2017, a California woman died after receiving an IV preparation of curcumin, a chemical constituent in the Indian spice turmeric featured in alternative medicine. The U.S. Food and Drug Administration found that the treating ND mixed the curcumin emulsion product with ungraded castor oil that had a warning label stating: “CAUTION: For manufacturing or laboratory use only.”
Because naturopathic care is generally not covered by insurance, it can also be expensive for patients who pay out of pocket.
Ironically, the mainstream health care system helps create the environment in which naturopathic medicine thrives.
It offers patients a more relaxed and personal alternative to rushed visits with harried doctors scrambling to see the required number of patients in a day. By contrast, an initial visit with an ND might last a leisurely 60 minutes, with 30-minute follow-up appointments.
Mr. Caulfield acknowledged that the relaxed naturopathic approach can be more attractive to patients but said the answer is to reform the current system: “You don’t fix a broken arm by acupuncture.”
A version of this article first appeared on Medscape.com.
Jon Hislop, MD, PhD, hadn’t been in practice very long before patients began coming to him with requests to order tests that their naturopaths had recommended.
The family physician in North Vancouver, British Columbia, knew little about naturopathy but began researching it.
“I was finding that some of what the naturopaths were telling them was a little odd. Some of the tests they were asking for were unnecessary,” Dr. Hislop said.
The more he learned about naturopathy, the more appalled he became. He eventually took to Twitter, where he wages a campaign against naturopathy and alternative medicine.
“There is no alternative medicine,” he said. “There’s medicine and there’s other stuff. We need to stick to medicine and stay away from the other stuff.”
Dr. Hislop is not alone in his criticism of naturopathic medicine. Professional medical societies almost universally oppose naturopathy, but that has not stopped its spread or prevented it from becoming part of some health care systems.
Americans spent $30.2 billion on out-of-pocket complementary health care, according to a 2016 report from the National Institutes of Health. That includes everything from herbal supplements and massage therapy to chiropractic care.
What is naturopathic medicine?
Naturopathy came to the United States from Germany in the 1800s, but some of its practices are thousands of years old. Naturopathic treatments include homeopathy, IV vitamin infusions, acupuncture, Reiki, and herbal supplements.
Naturopathy is based on the belief that the body has an innate ability to heal itself. It discourages drugs and surgery in favor of supplements, herbs, and other so-called natural treatments. Much of it centers around addressing lifestyle issues and counseling patients to improve their diets, quit smoking, exercise more, lose weight, etc., in order to address the root causes of some health problems.
Practitioners are critical of Western medicine for what they regard as an over-reliance on drugs and technology and for treating symptoms rather than the causes of disease.
“We get a lot of people who are at the end of their ropes, people with hard-to-diagnose diseases who know they are sick but whose labs are normal,” said Jaquel Patterson, ND, former president of the American Association of Naturopathic Physicians (AANP) and medical director of a naturopathic practice in Connecticut.
Separate training and licensing
There are major differences among naturopaths.
At one extreme are unlicensed, self-taught “healers,” who can embrace everything from homeopathy to aromatherapy.
At the other end are naturopathic doctors (NDs), who are more likely to become part of health care systems. These caregivers are trained and licensed, though not by the same institutions as traditional physicians.
To be licensed, NDs must graduate from one of seven accredited naturopathic medical schools in the United States and Canada. In addition to a standard medical curriculum, schools require graduates to complete 4 years of training in clinical nutrition, acupuncture, homeopathic medicine, botanical medicine, physical medicine, and counseling. Medical students intern in clinical settings for 2 years.
NDs are eager to distinguish themselves from their uncredentialed counterparts.
“Some people go to a weekend class and call themselves naturopaths. That’s very concerning. I don’t want those people to be licensed either,” said Hallie Armstrong, ND, who practices in Michigan.
In the United States, there are 6,000 practicing NDs and an unknown number of unlicensed naturopathic healers.
Can naturopaths call themselves ‘physicians’?
Twenty-two states, the District of Columbia, Puerto Rico, and the U.S. Virgin Islands have licensing or registration laws for naturopathic doctors. Three states – South Carolina, Tennessee, and Florida – prohibit practicing naturopathic medicine without a license, according to the AANP.
States that license NDs differ in what they permit them to do.
Nine states allow licensed NDs to use the term “physician,” although this is prohibited in seven states. Most licensed states allow naturopathic practitioners some prescribing authority, including the prescribing of many controlled substances, although only a few states permit full prescribing rights. Most states that license NDs allow them to prescribe and administer nonprescription therapeutic substances, drugs, and therapies.
Twelve states and the District of Columbia allow licensed naturopathic doctors to perform some minor procedures, such as stitching up wounds. Additionally, 13 states allow NDs to order diagnostic tests.
Although the AANP lobbies to get licensure in more states and to expand the activities that NDs can perform, the medical establishment in those states nearly always opposes the legislation, as do national organizations, such as the American Academy of Family Physicians and the American College of Physicians.
“They absolutely will not stop until they get licenses. They’ve done a really good job of selling themselves as legitimate health care professionals to state legislatures,” said David Gorski, MD, PhD, FACS, a surgical oncologist and managing editor of Science-Based Medicine, a blog that attacks unproven medical claims and defends traditional medicine. Naturopathy is a favorite target.
Are naturopaths gaining ground anyway?
Despite the opposition of the medical establishment and many individual health care professionals, a growing number of health care systems are adopting alternative medicine.
In 2018, the AANP stated that 28 prominent health systems, hospitals, and cancer treatment centers had one or more licensed NDs on staff. Among them were Cancer Treatment Centers of America, Cedars-Sinai, Columbia University’s Herbert Irving Comprehensive Cancer Center, and the Fred Hutchinson Cancer Research Center.
Other health care systems may not have NDs on staff but provide naturopathic treatments, usually under the heading of “complementary medicine” or “integrative medicine.” For example, the Cleveland Clinic’s Center for Integrative and Lifestyle Medicine offers acupuncture, Chinese herbal medicine, Reiki, yoga, and culinary medicine.
Critics find this appalling.
“I think it’s a mistake to integrate that kind of practice into a science-based health care setting. If we learned anything over the past year, it’s that medicine based on magical thinking is dangerous,” said Timothy Caulfield, LLM, FCAHS, research director at the Health Law Institute of the University of Alberta, Edmonton.
Dr. Gorski added: “I’m not exactly sure why doctors who should know better have become more accepting of practices that aren’t science-based or are outright quackery.”
Becoming part of the system
Beaumont Health, Michigan’s largest health care system, added integrative medicine in 2006 and hired its first naturopathic practitioners a year later.
The integrative practitioners began in oncology, offering such things as massage therapy, acupuncture, guided imagery, and Reiki. “Very quickly, people outside oncology began saying, ‘I’ve got a cardiology patient who would really benefit from this ... I’ve got a GI patient who could benefit from this...,’” said Maureen Anderson, MD, medical director of Beaumont Integrative Medicine.
Beaumont now offers integrative medicine at three locations. They average 20,000 visits a year and work with 50 to 60 practitioners, many of whom work part-time.
Because Michigan does not license NDs, their scope of practice at Beaumont is limited. They take patient histories, provide advice on nutrition, diet, and exercise, and prescribe herbs and supplements. Beaumont operates its own herbal and supplement pharmacy.
NDs work under the medical supervision of Dr. Anderson, an emergency medicine physician who became interested in naturopathy because she thought traditional medicine doesn’t do a good job of providing care for chronic conditions. Any initial skepticism on the part of the medical staff has been overcome by seeing the benefits naturopathy provides, Dr. Anderson said. The claim is echoed by Mr. Armstrong, an ND who works in the system part-time: “As soon as [doctors] understand our schooling and where we’re coming from and understand that we want to do the same things, then they’re very accepting.”
The University of California, Irvine, health care system has one of the largest naturopathic medicine programs in the country, the result of a $200 million donation in 2017 from a couple who champion alternative medicine. The Susan Samueli Integrative Health Institute includes 28 health care professionals, including MDs, NDs, RNs, acupuncturists, dietitians, yoga instructors, and others. It includes a research arm, which is focused primarily on acupuncture.
The alternative medicine offerings benefit the system, said Kim Hecht, DO, medical director of inpatient and ambulatory services at the Samueli Institute.
“I’m not against traditional medicine, because I think everything has a time and a place,” Dr. Hecht said. However, she rejects the idea that MDs can offer the same holistic approach as NDs.
“Medical science likes to say we’re interested in treating the whole person, but if you look at medical school courses, that’s not what’s being taught,” she said.
The chance to work within a traditional health care system was attractive to Arvin Jenab, ND, medical director of naturopathic medicine at the institute.
“It offers the opportunity to refine our medicine and trim the things that aren’t necessary or are controversial and concentrate on the things at the core of what we do,” he said.
UCI Health practices a conservative model of naturopathy that supports traditional practitioners, Mr. Jenab said.
Is there any harm?
Some patients clearly want what naturopathy offers. So what’s the harm?
Health care systems that integrate alternative medicine legitimize it and lower the overall standard of care, Mr. Caulfield said. Most naturopathy claims are not backed by evidence, and making it available to patients amounts to deceiving them, he said.
“If there’s good science behind it, it’s not going to be alternative medicine; it’s going to be medicine,” Mr. Caulfield said.
Family physician Dr. Hislop said that refusing to order naturopath-recommended tests interferes with his relationships with patients and often requires lengthy conversations to explain the problems with naturopathy.
Naturopathic medicine can deter patients from seeking proven conventional treatments, which can put their health at risk, Dr. Gorski said.
Some naturopaths could potentially be harmful.
In 2017, a California woman died after receiving an IV preparation of curcumin, a chemical constituent in the Indian spice turmeric featured in alternative medicine. The U.S. Food and Drug Administration found that the treating ND mixed the curcumin emulsion product with ungraded castor oil that had a warning label stating: “CAUTION: For manufacturing or laboratory use only.”
Because naturopathic care is generally not covered by insurance, it can also be expensive for patients who pay out of pocket.
Ironically, the mainstream health care system helps create the environment in which naturopathic medicine thrives.
It offers patients a more relaxed and personal alternative to rushed visits with harried doctors scrambling to see the required number of patients in a day. By contrast, an initial visit with an ND might last a leisurely 60 minutes, with 30-minute follow-up appointments.
Mr. Caulfield acknowledged that the relaxed naturopathic approach can be more attractive to patients but said the answer is to reform the current system: “You don’t fix a broken arm by acupuncture.”
A version of this article first appeared on Medscape.com.
New STRENGTH analysis reignites debate on omega-3 CV benefits
Questions over the cardiovascular benefits shown in the REDUCE-IT trial with icosapent ethyl, a high-dose eicosapentaenoic acid (EPA) product, have been reignited with a new analysis from the STRENGTH trial showing no benefit of a high-dose combined omega-3 fatty acid product in patients who achieved the highest EPA levels and no harm in those with the highest levels of docosahexaenoic acid (DHA).
STRENGTH investigator Steven Nissen, MD, said these new results add to concerns about the positive result in the previously reported REDUCE-IT trial and suggest that “there is no strong evidence of a benefit of fish oil in preventing major cardiovascular events.”
But Dr. Nissen, who is chair of the department of cardiovascular medicine at the Cleveland Clinic in Ohio, pointed out evidence of harm, with both REDUCE-IT and STRENGTH showing an increase in atrial fibrillation with the high-dose omega-3 fatty acid products.
“Fish oils increase the risk of atrial fibrillation substantially, and there is no solid evidence that they help the heart in any way,” he stated.
The new STRENGTH analysis was presented at the annual scientific sessions of the American College of Cardiology. and was simultaneously published in JAMA Cardiology.
The REDUCE-IT trial showed a large 25% relative-risk reduction in cardiovascular events in patients taking icosapent ethyl (Vascepa, Amarin), a high-dose purified formulation of EPA, compared with patients taking a mineral oil placebo. But a similar trial, STRENGTH, showed no effect of a similar high dose of the mixed EPA/DHA product (Epanova, AstraZeneca), compared with a corn oil placebo.
The different results from these two studies have led to many questions about how the benefits seen in REDUCE-IT were brought about, and why they weren’t replicated in the STRENGTH study.
Dr. Nissen noted that several hypotheses have been proposed. These include a potential adverse effect of the mineral oil placebo in the REDUCE-IT trial, which may have elevated risk in the placebo treatment group and led to a false-positive result for icosapent ethyl. Another possibility is that the moderately higher plasma levels of EPA achieved in REDUCE-IT were responsible for the observed benefits or that the coadministration of DHA in STRENGTH may have counteracted the potential beneficial effects of EPA.
The current post hoc analysis of STRENGTH was conducted to address these latter two possibilities. It aimed to assess the association between cardiovascular outcomes and achieved levels of EPA, DHA, or changes in levels of these fatty acids.
“In our new analysis, among patients treated with fish oil, we found no evidence that EPA is beneficial or that DHA is harmful,” Dr. Nissen said.
Results of the new analysis showed an absence of a benefit from achieving high levels of EPA or harm from achieving high levels of DHA which, the authors say, “strengthens the concerns that the choice of comparator may have influenced the divergent results observed in the two trials.”
“Unlike corn oil, which is inert, mineral oil has major adverse effects, increasing LDL by 10.9% and CRP [C-reactive protein] by 32% in the REDUCE-IT trial,” Dr. Nissen said. “If you give a toxic placebo, then the active drug may falsely look really good.”
The STRENGTH trial randomly assigned 13,078 individuals at high risk for major cardiovascular events to receive 4 g daily of the EPA/DHA combined product (omega-3 carboxylic acid) or corn oil as the placebo. Main results, reported previously, showed no difference between the two groups in terms of the primary outcome – a composite of cardiovascular death, myocardial infarction, stroke, coronary revascularization, or unstable angina requiring hospitalization.
The current analysis, in 10,382 patients with available omega-3 fatty acid levels, looked at event rates according to tertiles of achieved EPA and DHA levels. The median plasma EPA level for patients taking the omega-3 product was 89 mcg/mL, with the top tertile achieving levels of 151 mcg/mL (a 443% increase). Dr. Nissen pointed out that this was higher than the median level of EPA reported in the REDUCE-IT trial (144 mcg/mL).
The median level of DHA was 91 mcg/mL, rising to 118 mcg/mL (a 68% increase) in the top tertile in the STRENGTH analysis.
Results showed no difference in the occurrence of the prespecified primary outcome among patients treated with omega-3 carboxylic acid who were in the top tertile of achieved EPA levels at 1 year (event rate, 11.3%), compared with patients treated with corn oil (11.0%), a nonsignificant difference (hazard ratio, 0.98; P = .81).
For DHA, patients in the top tertile of achieved DHA levels had an event rate of 11.4% vs. 11.0% in the corn oil group, also a nonsignificant difference (HR, 1.02; P = .85)
Sensitivity analyses based on the highest tertile of change in EPA or DHA levels showed similarly neutral results.
Because plasma levels may not reflect tissue levels of EPA or DHA, additional analyses assessed red blood cell EPA and DHA levels, neither of which showed any evidence of benefit or harm.
“These findings suggest that supplementation of omega-3 fatty acids in high-risk cardiovascular patients is neutral even at the highest achieved levels,” Dr. Nissen said. “And, in the context of increased risk of atrial fibrillation in omega-3 trials, they cast uncertainty over whether there is net benefit or harm with any omega-3 preparation,” he concluded.
He suggested that the choice of placebo comparator may play an important role in determining outcome for trials of omega-3 products, adding that further research is needed with trials specifically designed to compare corn oil with mineral oil and compare purified EPA with other formulations of omega-3 fatty acids.
At an press conference, Dr. Nissen said he could not recommend use of omega-3 fatty acid products for cardiovascular risk reduction given the uncertainty over the benefit in REDUCE-IT.
“We need replication, and the problem is STRENGTH did not replicate REDUCE-IT,” he stated.
REDUCE-IT investigator responds
The discussant of the STRENGTH analysis at the ACC presentation, Deepak L. Bhatt, MD, who was lead investigator of the REDUCE-IT trial, suggested that one conclusion could be that “an absence of a relationship in a negative trial doesn’t tell us that much other than that specific drug doesn’t work.”
Dr. Bhatt, who is executive director of interventional cardiovascular programs at Brigham and Women’s Hospital Heart & Vascular Center, Boston, said in an interview that comparisons should not be made between different trials using different products.
“I commend the STRENGTH investigators on a well-conducted trial that provided a definitive answer about the specific drug they studied, finding no benefit. But in a completely negative trial, I wouldn’t necessarily expect to see a relationship between any biomarker and outcome,” he said.
“With respect to icosapent ethyl (pure EPA), every cardiovascular trial to date has been positive: REDUCE-IT (randomized, placebo-controlled), JELIS (randomized, no placebo), EVAPORATE (randomized, placebo-controlled), CHERRY (randomized, no placebo), and some smaller ones,” Dr. Bhatt added. “Both REDUCE-IT and JELIS found associations between higher levels of EPA and lower rates of cardiovascular events, suggesting that higher EPA levels attained specifically with icosapent ethyl are beneficial.”
Pointing out that all the glucagonlike peptide–1 agonists lower glucose, for example, but not all reduce cardiovascular events, Dr. Bhatt said it was best to focus on clinical trial results and not overly focus on biomarker changes.
“Yes, the drug in STRENGTH raised EPA (and raised DHA, as well as lowering triglycerides), but the drug in REDUCE-IT and JELIS raised EPA much more, without raising DHA – and more importantly, the increase in EPA was via a totally different drug, with many different properties,” he added.
In his discussion of the study at the ACC presentation, Dr. Bhatt pointed out that in the STRENGTH trial overall there was no reduction in major adverse cardiovascular events despite a 19% reduction in triglycerides, which he said was a “very interesting disconnect.” He asked Dr. Nissen what he thought the reason was for the observation in this analysis of no relationship between EPA or DHA level and triglyceride reduction.
Dr. Nissen said that was an interesting point. “When we look at the two trials, they both reduced triglyceride levels by an almost identical amount, 19%, but we don’t see a relationship with that and EPA levels achieved.” He suggested this may be because of different threshold levels.
Dr. Bhatt also noted that high-intensity statin use was lower in the patients with higher EPA levels in the STRENGTH analysis, but Dr. Nissen countered: “I don’t think that was enough of a difference to explain the lack of an effect.”
Invited commentator on the new analysis at an ACC press conference, Eileen Handberg, PhD, said it was important to try to understand the reasons behind the different results of the STRENGTH and REDUCE-IT trials. “These new findings are important because they explain potentially why these outcomes are different,” she stated.
Dr. Handberg, who is professor of medicine at the University of Florida, Gainesville, said she hoped the additional research called for by Dr. Nissen would go ahead as a head-to-head study of the two omega-3 products or of the two different placebo oils.
The STRENGTH trial was sponsored by Astra Zeneca. Dr. Nissen reports research grants from AbbVie, Amgen, Astra Zeneca, Eli Lilly, Esperion Therapeutics, MEDTRONIC, MyoKardia, Novartis, Novo Nordisk, Pfizer, and Silence Therapeutics. Dr. Bhatt reports constant fees/honoraria from CellProthera, Elsevier Practice Update Cardiology, K2P, Level Ex, Medtelligence, MJH Life Sciences, and WebMD; data safety monitoring board activities with Contego; other roles with TobeSoft, Belvoir Publications, Cardax, Cereno Scientific, Clinical Cardiology, Elsevier, HMP Global, Janssen Pharmaceuticals, Journal of Invasive Cardiology, Medscape Cardiology, Merck, MyoKardia, Novo Nordisk, PhaseBio, PLx Pharma, Regado Biosciences, and Slack Publications/Cardiology Research Foundation; and research grants from Abbott, Afimmune, Amarin, Amgen, Astra Zeneca, Bayer Healthcare Pharmaceuticals, Boehringer Ingelheim Pharmaceuticals, Bristol-Myers Squibb, Cardax, Chiesi, Eisai, Eli Lilly, Ethicon, FlowCo, Forest Laboratories, Fractyl, HLS Therapeutics, Idorsia, Ironwood, Ischemix, Lexicon, MEDTRONIC, MyoKardia, Owkin, Pfizer, PhaseBio, PLx Pharma, Regeneron, Roche, Sanofi Aventis, Synaptic, Takeda, and The Medicines Company.
A version of this article first appeared on Medscape.com.
Questions over the cardiovascular benefits shown in the REDUCE-IT trial with icosapent ethyl, a high-dose eicosapentaenoic acid (EPA) product, have been reignited with a new analysis from the STRENGTH trial showing no benefit of a high-dose combined omega-3 fatty acid product in patients who achieved the highest EPA levels and no harm in those with the highest levels of docosahexaenoic acid (DHA).
STRENGTH investigator Steven Nissen, MD, said these new results add to concerns about the positive result in the previously reported REDUCE-IT trial and suggest that “there is no strong evidence of a benefit of fish oil in preventing major cardiovascular events.”
But Dr. Nissen, who is chair of the department of cardiovascular medicine at the Cleveland Clinic in Ohio, pointed out evidence of harm, with both REDUCE-IT and STRENGTH showing an increase in atrial fibrillation with the high-dose omega-3 fatty acid products.
“Fish oils increase the risk of atrial fibrillation substantially, and there is no solid evidence that they help the heart in any way,” he stated.
The new STRENGTH analysis was presented at the annual scientific sessions of the American College of Cardiology. and was simultaneously published in JAMA Cardiology.
The REDUCE-IT trial showed a large 25% relative-risk reduction in cardiovascular events in patients taking icosapent ethyl (Vascepa, Amarin), a high-dose purified formulation of EPA, compared with patients taking a mineral oil placebo. But a similar trial, STRENGTH, showed no effect of a similar high dose of the mixed EPA/DHA product (Epanova, AstraZeneca), compared with a corn oil placebo.
The different results from these two studies have led to many questions about how the benefits seen in REDUCE-IT were brought about, and why they weren’t replicated in the STRENGTH study.
Dr. Nissen noted that several hypotheses have been proposed. These include a potential adverse effect of the mineral oil placebo in the REDUCE-IT trial, which may have elevated risk in the placebo treatment group and led to a false-positive result for icosapent ethyl. Another possibility is that the moderately higher plasma levels of EPA achieved in REDUCE-IT were responsible for the observed benefits or that the coadministration of DHA in STRENGTH may have counteracted the potential beneficial effects of EPA.
The current post hoc analysis of STRENGTH was conducted to address these latter two possibilities. It aimed to assess the association between cardiovascular outcomes and achieved levels of EPA, DHA, or changes in levels of these fatty acids.
“In our new analysis, among patients treated with fish oil, we found no evidence that EPA is beneficial or that DHA is harmful,” Dr. Nissen said.
Results of the new analysis showed an absence of a benefit from achieving high levels of EPA or harm from achieving high levels of DHA which, the authors say, “strengthens the concerns that the choice of comparator may have influenced the divergent results observed in the two trials.”
“Unlike corn oil, which is inert, mineral oil has major adverse effects, increasing LDL by 10.9% and CRP [C-reactive protein] by 32% in the REDUCE-IT trial,” Dr. Nissen said. “If you give a toxic placebo, then the active drug may falsely look really good.”
The STRENGTH trial randomly assigned 13,078 individuals at high risk for major cardiovascular events to receive 4 g daily of the EPA/DHA combined product (omega-3 carboxylic acid) or corn oil as the placebo. Main results, reported previously, showed no difference between the two groups in terms of the primary outcome – a composite of cardiovascular death, myocardial infarction, stroke, coronary revascularization, or unstable angina requiring hospitalization.
The current analysis, in 10,382 patients with available omega-3 fatty acid levels, looked at event rates according to tertiles of achieved EPA and DHA levels. The median plasma EPA level for patients taking the omega-3 product was 89 mcg/mL, with the top tertile achieving levels of 151 mcg/mL (a 443% increase). Dr. Nissen pointed out that this was higher than the median level of EPA reported in the REDUCE-IT trial (144 mcg/mL).
The median level of DHA was 91 mcg/mL, rising to 118 mcg/mL (a 68% increase) in the top tertile in the STRENGTH analysis.
Results showed no difference in the occurrence of the prespecified primary outcome among patients treated with omega-3 carboxylic acid who were in the top tertile of achieved EPA levels at 1 year (event rate, 11.3%), compared with patients treated with corn oil (11.0%), a nonsignificant difference (hazard ratio, 0.98; P = .81).
For DHA, patients in the top tertile of achieved DHA levels had an event rate of 11.4% vs. 11.0% in the corn oil group, also a nonsignificant difference (HR, 1.02; P = .85)
Sensitivity analyses based on the highest tertile of change in EPA or DHA levels showed similarly neutral results.
Because plasma levels may not reflect tissue levels of EPA or DHA, additional analyses assessed red blood cell EPA and DHA levels, neither of which showed any evidence of benefit or harm.
“These findings suggest that supplementation of omega-3 fatty acids in high-risk cardiovascular patients is neutral even at the highest achieved levels,” Dr. Nissen said. “And, in the context of increased risk of atrial fibrillation in omega-3 trials, they cast uncertainty over whether there is net benefit or harm with any omega-3 preparation,” he concluded.
He suggested that the choice of placebo comparator may play an important role in determining outcome for trials of omega-3 products, adding that further research is needed with trials specifically designed to compare corn oil with mineral oil and compare purified EPA with other formulations of omega-3 fatty acids.
At an press conference, Dr. Nissen said he could not recommend use of omega-3 fatty acid products for cardiovascular risk reduction given the uncertainty over the benefit in REDUCE-IT.
“We need replication, and the problem is STRENGTH did not replicate REDUCE-IT,” he stated.
REDUCE-IT investigator responds
The discussant of the STRENGTH analysis at the ACC presentation, Deepak L. Bhatt, MD, who was lead investigator of the REDUCE-IT trial, suggested that one conclusion could be that “an absence of a relationship in a negative trial doesn’t tell us that much other than that specific drug doesn’t work.”
Dr. Bhatt, who is executive director of interventional cardiovascular programs at Brigham and Women’s Hospital Heart & Vascular Center, Boston, said in an interview that comparisons should not be made between different trials using different products.
“I commend the STRENGTH investigators on a well-conducted trial that provided a definitive answer about the specific drug they studied, finding no benefit. But in a completely negative trial, I wouldn’t necessarily expect to see a relationship between any biomarker and outcome,” he said.
“With respect to icosapent ethyl (pure EPA), every cardiovascular trial to date has been positive: REDUCE-IT (randomized, placebo-controlled), JELIS (randomized, no placebo), EVAPORATE (randomized, placebo-controlled), CHERRY (randomized, no placebo), and some smaller ones,” Dr. Bhatt added. “Both REDUCE-IT and JELIS found associations between higher levels of EPA and lower rates of cardiovascular events, suggesting that higher EPA levels attained specifically with icosapent ethyl are beneficial.”
Pointing out that all the glucagonlike peptide–1 agonists lower glucose, for example, but not all reduce cardiovascular events, Dr. Bhatt said it was best to focus on clinical trial results and not overly focus on biomarker changes.
“Yes, the drug in STRENGTH raised EPA (and raised DHA, as well as lowering triglycerides), but the drug in REDUCE-IT and JELIS raised EPA much more, without raising DHA – and more importantly, the increase in EPA was via a totally different drug, with many different properties,” he added.
In his discussion of the study at the ACC presentation, Dr. Bhatt pointed out that in the STRENGTH trial overall there was no reduction in major adverse cardiovascular events despite a 19% reduction in triglycerides, which he said was a “very interesting disconnect.” He asked Dr. Nissen what he thought the reason was for the observation in this analysis of no relationship between EPA or DHA level and triglyceride reduction.
Dr. Nissen said that was an interesting point. “When we look at the two trials, they both reduced triglyceride levels by an almost identical amount, 19%, but we don’t see a relationship with that and EPA levels achieved.” He suggested this may be because of different threshold levels.
Dr. Bhatt also noted that high-intensity statin use was lower in the patients with higher EPA levels in the STRENGTH analysis, but Dr. Nissen countered: “I don’t think that was enough of a difference to explain the lack of an effect.”
Invited commentator on the new analysis at an ACC press conference, Eileen Handberg, PhD, said it was important to try to understand the reasons behind the different results of the STRENGTH and REDUCE-IT trials. “These new findings are important because they explain potentially why these outcomes are different,” she stated.
Dr. Handberg, who is professor of medicine at the University of Florida, Gainesville, said she hoped the additional research called for by Dr. Nissen would go ahead as a head-to-head study of the two omega-3 products or of the two different placebo oils.
The STRENGTH trial was sponsored by Astra Zeneca. Dr. Nissen reports research grants from AbbVie, Amgen, Astra Zeneca, Eli Lilly, Esperion Therapeutics, MEDTRONIC, MyoKardia, Novartis, Novo Nordisk, Pfizer, and Silence Therapeutics. Dr. Bhatt reports constant fees/honoraria from CellProthera, Elsevier Practice Update Cardiology, K2P, Level Ex, Medtelligence, MJH Life Sciences, and WebMD; data safety monitoring board activities with Contego; other roles with TobeSoft, Belvoir Publications, Cardax, Cereno Scientific, Clinical Cardiology, Elsevier, HMP Global, Janssen Pharmaceuticals, Journal of Invasive Cardiology, Medscape Cardiology, Merck, MyoKardia, Novo Nordisk, PhaseBio, PLx Pharma, Regado Biosciences, and Slack Publications/Cardiology Research Foundation; and research grants from Abbott, Afimmune, Amarin, Amgen, Astra Zeneca, Bayer Healthcare Pharmaceuticals, Boehringer Ingelheim Pharmaceuticals, Bristol-Myers Squibb, Cardax, Chiesi, Eisai, Eli Lilly, Ethicon, FlowCo, Forest Laboratories, Fractyl, HLS Therapeutics, Idorsia, Ironwood, Ischemix, Lexicon, MEDTRONIC, MyoKardia, Owkin, Pfizer, PhaseBio, PLx Pharma, Regeneron, Roche, Sanofi Aventis, Synaptic, Takeda, and The Medicines Company.
A version of this article first appeared on Medscape.com.
Questions over the cardiovascular benefits shown in the REDUCE-IT trial with icosapent ethyl, a high-dose eicosapentaenoic acid (EPA) product, have been reignited with a new analysis from the STRENGTH trial showing no benefit of a high-dose combined omega-3 fatty acid product in patients who achieved the highest EPA levels and no harm in those with the highest levels of docosahexaenoic acid (DHA).
STRENGTH investigator Steven Nissen, MD, said these new results add to concerns about the positive result in the previously reported REDUCE-IT trial and suggest that “there is no strong evidence of a benefit of fish oil in preventing major cardiovascular events.”
But Dr. Nissen, who is chair of the department of cardiovascular medicine at the Cleveland Clinic in Ohio, pointed out evidence of harm, with both REDUCE-IT and STRENGTH showing an increase in atrial fibrillation with the high-dose omega-3 fatty acid products.
“Fish oils increase the risk of atrial fibrillation substantially, and there is no solid evidence that they help the heart in any way,” he stated.
The new STRENGTH analysis was presented at the annual scientific sessions of the American College of Cardiology. and was simultaneously published in JAMA Cardiology.
The REDUCE-IT trial showed a large 25% relative-risk reduction in cardiovascular events in patients taking icosapent ethyl (Vascepa, Amarin), a high-dose purified formulation of EPA, compared with patients taking a mineral oil placebo. But a similar trial, STRENGTH, showed no effect of a similar high dose of the mixed EPA/DHA product (Epanova, AstraZeneca), compared with a corn oil placebo.
The different results from these two studies have led to many questions about how the benefits seen in REDUCE-IT were brought about, and why they weren’t replicated in the STRENGTH study.
Dr. Nissen noted that several hypotheses have been proposed. These include a potential adverse effect of the mineral oil placebo in the REDUCE-IT trial, which may have elevated risk in the placebo treatment group and led to a false-positive result for icosapent ethyl. Another possibility is that the moderately higher plasma levels of EPA achieved in REDUCE-IT were responsible for the observed benefits or that the coadministration of DHA in STRENGTH may have counteracted the potential beneficial effects of EPA.
The current post hoc analysis of STRENGTH was conducted to address these latter two possibilities. It aimed to assess the association between cardiovascular outcomes and achieved levels of EPA, DHA, or changes in levels of these fatty acids.
“In our new analysis, among patients treated with fish oil, we found no evidence that EPA is beneficial or that DHA is harmful,” Dr. Nissen said.
Results of the new analysis showed an absence of a benefit from achieving high levels of EPA or harm from achieving high levels of DHA which, the authors say, “strengthens the concerns that the choice of comparator may have influenced the divergent results observed in the two trials.”
“Unlike corn oil, which is inert, mineral oil has major adverse effects, increasing LDL by 10.9% and CRP [C-reactive protein] by 32% in the REDUCE-IT trial,” Dr. Nissen said. “If you give a toxic placebo, then the active drug may falsely look really good.”
The STRENGTH trial randomly assigned 13,078 individuals at high risk for major cardiovascular events to receive 4 g daily of the EPA/DHA combined product (omega-3 carboxylic acid) or corn oil as the placebo. Main results, reported previously, showed no difference between the two groups in terms of the primary outcome – a composite of cardiovascular death, myocardial infarction, stroke, coronary revascularization, or unstable angina requiring hospitalization.
The current analysis, in 10,382 patients with available omega-3 fatty acid levels, looked at event rates according to tertiles of achieved EPA and DHA levels. The median plasma EPA level for patients taking the omega-3 product was 89 mcg/mL, with the top tertile achieving levels of 151 mcg/mL (a 443% increase). Dr. Nissen pointed out that this was higher than the median level of EPA reported in the REDUCE-IT trial (144 mcg/mL).
The median level of DHA was 91 mcg/mL, rising to 118 mcg/mL (a 68% increase) in the top tertile in the STRENGTH analysis.
Results showed no difference in the occurrence of the prespecified primary outcome among patients treated with omega-3 carboxylic acid who were in the top tertile of achieved EPA levels at 1 year (event rate, 11.3%), compared with patients treated with corn oil (11.0%), a nonsignificant difference (hazard ratio, 0.98; P = .81).
For DHA, patients in the top tertile of achieved DHA levels had an event rate of 11.4% vs. 11.0% in the corn oil group, also a nonsignificant difference (HR, 1.02; P = .85)
Sensitivity analyses based on the highest tertile of change in EPA or DHA levels showed similarly neutral results.
Because plasma levels may not reflect tissue levels of EPA or DHA, additional analyses assessed red blood cell EPA and DHA levels, neither of which showed any evidence of benefit or harm.
“These findings suggest that supplementation of omega-3 fatty acids in high-risk cardiovascular patients is neutral even at the highest achieved levels,” Dr. Nissen said. “And, in the context of increased risk of atrial fibrillation in omega-3 trials, they cast uncertainty over whether there is net benefit or harm with any omega-3 preparation,” he concluded.
He suggested that the choice of placebo comparator may play an important role in determining outcome for trials of omega-3 products, adding that further research is needed with trials specifically designed to compare corn oil with mineral oil and compare purified EPA with other formulations of omega-3 fatty acids.
At an press conference, Dr. Nissen said he could not recommend use of omega-3 fatty acid products for cardiovascular risk reduction given the uncertainty over the benefit in REDUCE-IT.
“We need replication, and the problem is STRENGTH did not replicate REDUCE-IT,” he stated.
REDUCE-IT investigator responds
The discussant of the STRENGTH analysis at the ACC presentation, Deepak L. Bhatt, MD, who was lead investigator of the REDUCE-IT trial, suggested that one conclusion could be that “an absence of a relationship in a negative trial doesn’t tell us that much other than that specific drug doesn’t work.”
Dr. Bhatt, who is executive director of interventional cardiovascular programs at Brigham and Women’s Hospital Heart & Vascular Center, Boston, said in an interview that comparisons should not be made between different trials using different products.
“I commend the STRENGTH investigators on a well-conducted trial that provided a definitive answer about the specific drug they studied, finding no benefit. But in a completely negative trial, I wouldn’t necessarily expect to see a relationship between any biomarker and outcome,” he said.
“With respect to icosapent ethyl (pure EPA), every cardiovascular trial to date has been positive: REDUCE-IT (randomized, placebo-controlled), JELIS (randomized, no placebo), EVAPORATE (randomized, placebo-controlled), CHERRY (randomized, no placebo), and some smaller ones,” Dr. Bhatt added. “Both REDUCE-IT and JELIS found associations between higher levels of EPA and lower rates of cardiovascular events, suggesting that higher EPA levels attained specifically with icosapent ethyl are beneficial.”
Pointing out that all the glucagonlike peptide–1 agonists lower glucose, for example, but not all reduce cardiovascular events, Dr. Bhatt said it was best to focus on clinical trial results and not overly focus on biomarker changes.
“Yes, the drug in STRENGTH raised EPA (and raised DHA, as well as lowering triglycerides), but the drug in REDUCE-IT and JELIS raised EPA much more, without raising DHA – and more importantly, the increase in EPA was via a totally different drug, with many different properties,” he added.
In his discussion of the study at the ACC presentation, Dr. Bhatt pointed out that in the STRENGTH trial overall there was no reduction in major adverse cardiovascular events despite a 19% reduction in triglycerides, which he said was a “very interesting disconnect.” He asked Dr. Nissen what he thought the reason was for the observation in this analysis of no relationship between EPA or DHA level and triglyceride reduction.
Dr. Nissen said that was an interesting point. “When we look at the two trials, they both reduced triglyceride levels by an almost identical amount, 19%, but we don’t see a relationship with that and EPA levels achieved.” He suggested this may be because of different threshold levels.
Dr. Bhatt also noted that high-intensity statin use was lower in the patients with higher EPA levels in the STRENGTH analysis, but Dr. Nissen countered: “I don’t think that was enough of a difference to explain the lack of an effect.”
Invited commentator on the new analysis at an ACC press conference, Eileen Handberg, PhD, said it was important to try to understand the reasons behind the different results of the STRENGTH and REDUCE-IT trials. “These new findings are important because they explain potentially why these outcomes are different,” she stated.
Dr. Handberg, who is professor of medicine at the University of Florida, Gainesville, said she hoped the additional research called for by Dr. Nissen would go ahead as a head-to-head study of the two omega-3 products or of the two different placebo oils.
The STRENGTH trial was sponsored by Astra Zeneca. Dr. Nissen reports research grants from AbbVie, Amgen, Astra Zeneca, Eli Lilly, Esperion Therapeutics, MEDTRONIC, MyoKardia, Novartis, Novo Nordisk, Pfizer, and Silence Therapeutics. Dr. Bhatt reports constant fees/honoraria from CellProthera, Elsevier Practice Update Cardiology, K2P, Level Ex, Medtelligence, MJH Life Sciences, and WebMD; data safety monitoring board activities with Contego; other roles with TobeSoft, Belvoir Publications, Cardax, Cereno Scientific, Clinical Cardiology, Elsevier, HMP Global, Janssen Pharmaceuticals, Journal of Invasive Cardiology, Medscape Cardiology, Merck, MyoKardia, Novo Nordisk, PhaseBio, PLx Pharma, Regado Biosciences, and Slack Publications/Cardiology Research Foundation; and research grants from Abbott, Afimmune, Amarin, Amgen, Astra Zeneca, Bayer Healthcare Pharmaceuticals, Boehringer Ingelheim Pharmaceuticals, Bristol-Myers Squibb, Cardax, Chiesi, Eisai, Eli Lilly, Ethicon, FlowCo, Forest Laboratories, Fractyl, HLS Therapeutics, Idorsia, Ironwood, Ischemix, Lexicon, MEDTRONIC, MyoKardia, Owkin, Pfizer, PhaseBio, PLx Pharma, Regeneron, Roche, Sanofi Aventis, Synaptic, Takeda, and The Medicines Company.
A version of this article first appeared on Medscape.com.
FROM ACC 2021
FLOWER-MI: FFR-guided complete revascularization shows no advantage in STEMI
For patients with ST-elevated myocardial infarction (STEMI) undergoing complete revascularization, percutaneous coronary interventions (PCI) guided by fractional flow reserve (FFR) relative to angiography-guided PCI do not result in significantly lower risk of death or events, according to data from the randomized FLOWER-MI trial.
Rather, the events at 1 year were numerically lower among those randomized to the angiography-guided approach, according to the principal investigator of the trial, Etienne Puymirat, MD, PhD.
Prior studies showing an advantage for FFR-guided PCI in patients with coronary syndromes provided the hypothesis that FFR-guided PCI would also be superior for guiding PCI in STEMI patients. In the multicenter FAME trial, for example, FFR-guided PCI for patients with multivessel disease was associated with fewer stent placements (P < .001) and a nearly 30% lower rate of events at 1 year (P = .02).
While the advantage of complete revascularization, meaning PCI treatment of nonculprit as well as culprit lesions, has already been shown to be a better strategy than treatment of culprit lesions alone, FLOWER-MI is the first large study to compare FFR to angiography for guiding this approach to STEMI patients with multivessel disease, said Dr. Puymirat of Hôpital Européen George Pompidou, Paris, at the annual scientific sessions of the American College of Cardiology.
In this trial, involving multiple centers in France, STEMI patients were eligible for randomization if they had successful PCI of a culprit lesion and 50% or greater stenosis in at least one additional nonculprit lesion. The complete revascularization, whether patients were randomized to PCI guided by angiography or FFR, was performed during the index hospital admission. Patient management and follow-up was otherwise the same.
After a small number of exclusions, the intention-to-treat populations were 577 patients in the angiography-guided group and 586 in the FFR-guided group. The characteristics of the groups were well matched with an average age of about 62 years and similar rates of risk factors, such as hypertension and diabetes.
Angiography guidance just as good
The primary outcome was a composite of all-cause mortality, nonfatal MI, and unplanned revascularization. By hazard ratio, the risk of having one of these events within 1 year of PCI was numerically greater, at 32 in the FFR-guided group and 24 in the angiography-guided group, but the difference was not statistically significant (1.32; P = .31).
However, the total rate of events was low (5.5% vs. 4.2% for the angiography-guided and FFR-guided groups, respectively) and the confidence intervals were wide (95% CI, 0.78-2.23). This was also true of the components of the primary outcome.
No signal for a difference between strategies could be derived from these components, which included a higher rate of MI in the FFR-guided group (3.1% vs. 1.7%) but a lower rate of death (1.5% vs. 1.7%).
Unplanned hospitalizations leading to revascularization rates were also low (1.9% and 2.6% for angiography-guided and FFR-guided PCI, respectively), although it was reported that the rate of revascularization for nonculprit lesions was about twice as high in the FFR group (53.3% vs. 27.3%).
At 1 year, there were also low rates and no significant differences in a list of secondary outcomes that included hospitalization for recurrent ischemia or heart failure, stent thrombosis, and revascularization. As within the primary composite outcome, no pattern could be seen in the secondary events, some of which were numerically more common in the FFR-guided group and some numerically lower.
In a cost-efficacy analysis, the median per-patient cost of the FFR-guided strategy was about 500 Euros ($607) greater (8,832 vs. 8,322; P < .01), leading Dr. Puymirat to conclude that “the use of FFR for nonculprit lesions appears to be less effective but more expensive,” at least by costs derived in France.
Lack of statistical power limits interpretation
The conclusion of FLOWER-MI is that FFR-guided PCI in complete revascularization of nonculprit lesions in STEMI patients is not superior to an angiography-guided approach, but Dr. Puymirat cautioned that the low number of events precludes a definitive message.
William Fearon, MD, professor of cardiovascular medicine at Stanford (Calif.) University Medical Center, agreed. Based on his calculations, the trial was substantially underpowered. Evaluating the details of treatment in the FFR group, Dr. Fearon pointed out that a nonculprit lesion with a FFR of 0.80 or less was identified in about 55% of patients. Ultimately, 66% in the FFR group received PCI, eliminating the key distinction between strategies for the majority of patients enrolled.
“Only about one-third of the FFR-guided patients, or about 200 patients, did not receive nonculprit PCI, and therefore only in this small group could we expect a difference in outcomes from the angio-guided group,” Dr. Fearon said.
Fewer stents were placed in the FFR-guided than angiography-guided group (1.01 vs. 1.5), but Dr. Fearon suggested that it would be very difficult to show a difference in risk of events in a study of this size when event rates at 1 year reached only about 5%.
In response, Dr. Puymirat acknowledged that the rate of events for this trial, which was designed in 2015, were lower than expected. In recalculating the power needed based on the rate of events observed in FLOWER-MI, he estimated that about 8,000 patients would have been needed to show a meaningful difference in these PCI strategies.
Dr. Puymirat reports financial relationships with more than a dozen pharmaceutical companies, including Abbott, which provided some of the funding for this trial. Dr. Fearon reports financial relationships with Abbott, CathWorks, HeartFlow, and Medtronic.
For patients with ST-elevated myocardial infarction (STEMI) undergoing complete revascularization, percutaneous coronary interventions (PCI) guided by fractional flow reserve (FFR) relative to angiography-guided PCI do not result in significantly lower risk of death or events, according to data from the randomized FLOWER-MI trial.
Rather, the events at 1 year were numerically lower among those randomized to the angiography-guided approach, according to the principal investigator of the trial, Etienne Puymirat, MD, PhD.
Prior studies showing an advantage for FFR-guided PCI in patients with coronary syndromes provided the hypothesis that FFR-guided PCI would also be superior for guiding PCI in STEMI patients. In the multicenter FAME trial, for example, FFR-guided PCI for patients with multivessel disease was associated with fewer stent placements (P < .001) and a nearly 30% lower rate of events at 1 year (P = .02).
While the advantage of complete revascularization, meaning PCI treatment of nonculprit as well as culprit lesions, has already been shown to be a better strategy than treatment of culprit lesions alone, FLOWER-MI is the first large study to compare FFR to angiography for guiding this approach to STEMI patients with multivessel disease, said Dr. Puymirat of Hôpital Européen George Pompidou, Paris, at the annual scientific sessions of the American College of Cardiology.
In this trial, involving multiple centers in France, STEMI patients were eligible for randomization if they had successful PCI of a culprit lesion and 50% or greater stenosis in at least one additional nonculprit lesion. The complete revascularization, whether patients were randomized to PCI guided by angiography or FFR, was performed during the index hospital admission. Patient management and follow-up was otherwise the same.
After a small number of exclusions, the intention-to-treat populations were 577 patients in the angiography-guided group and 586 in the FFR-guided group. The characteristics of the groups were well matched with an average age of about 62 years and similar rates of risk factors, such as hypertension and diabetes.
Angiography guidance just as good
The primary outcome was a composite of all-cause mortality, nonfatal MI, and unplanned revascularization. By hazard ratio, the risk of having one of these events within 1 year of PCI was numerically greater, at 32 in the FFR-guided group and 24 in the angiography-guided group, but the difference was not statistically significant (1.32; P = .31).
However, the total rate of events was low (5.5% vs. 4.2% for the angiography-guided and FFR-guided groups, respectively) and the confidence intervals were wide (95% CI, 0.78-2.23). This was also true of the components of the primary outcome.
No signal for a difference between strategies could be derived from these components, which included a higher rate of MI in the FFR-guided group (3.1% vs. 1.7%) but a lower rate of death (1.5% vs. 1.7%).
Unplanned hospitalizations leading to revascularization rates were also low (1.9% and 2.6% for angiography-guided and FFR-guided PCI, respectively), although it was reported that the rate of revascularization for nonculprit lesions was about twice as high in the FFR group (53.3% vs. 27.3%).
At 1 year, there were also low rates and no significant differences in a list of secondary outcomes that included hospitalization for recurrent ischemia or heart failure, stent thrombosis, and revascularization. As within the primary composite outcome, no pattern could be seen in the secondary events, some of which were numerically more common in the FFR-guided group and some numerically lower.
In a cost-efficacy analysis, the median per-patient cost of the FFR-guided strategy was about 500 Euros ($607) greater (8,832 vs. 8,322; P < .01), leading Dr. Puymirat to conclude that “the use of FFR for nonculprit lesions appears to be less effective but more expensive,” at least by costs derived in France.
Lack of statistical power limits interpretation
The conclusion of FLOWER-MI is that FFR-guided PCI in complete revascularization of nonculprit lesions in STEMI patients is not superior to an angiography-guided approach, but Dr. Puymirat cautioned that the low number of events precludes a definitive message.
William Fearon, MD, professor of cardiovascular medicine at Stanford (Calif.) University Medical Center, agreed. Based on his calculations, the trial was substantially underpowered. Evaluating the details of treatment in the FFR group, Dr. Fearon pointed out that a nonculprit lesion with a FFR of 0.80 or less was identified in about 55% of patients. Ultimately, 66% in the FFR group received PCI, eliminating the key distinction between strategies for the majority of patients enrolled.
“Only about one-third of the FFR-guided patients, or about 200 patients, did not receive nonculprit PCI, and therefore only in this small group could we expect a difference in outcomes from the angio-guided group,” Dr. Fearon said.
Fewer stents were placed in the FFR-guided than angiography-guided group (1.01 vs. 1.5), but Dr. Fearon suggested that it would be very difficult to show a difference in risk of events in a study of this size when event rates at 1 year reached only about 5%.
In response, Dr. Puymirat acknowledged that the rate of events for this trial, which was designed in 2015, were lower than expected. In recalculating the power needed based on the rate of events observed in FLOWER-MI, he estimated that about 8,000 patients would have been needed to show a meaningful difference in these PCI strategies.
Dr. Puymirat reports financial relationships with more than a dozen pharmaceutical companies, including Abbott, which provided some of the funding for this trial. Dr. Fearon reports financial relationships with Abbott, CathWorks, HeartFlow, and Medtronic.
For patients with ST-elevated myocardial infarction (STEMI) undergoing complete revascularization, percutaneous coronary interventions (PCI) guided by fractional flow reserve (FFR) relative to angiography-guided PCI do not result in significantly lower risk of death or events, according to data from the randomized FLOWER-MI trial.
Rather, the events at 1 year were numerically lower among those randomized to the angiography-guided approach, according to the principal investigator of the trial, Etienne Puymirat, MD, PhD.
Prior studies showing an advantage for FFR-guided PCI in patients with coronary syndromes provided the hypothesis that FFR-guided PCI would also be superior for guiding PCI in STEMI patients. In the multicenter FAME trial, for example, FFR-guided PCI for patients with multivessel disease was associated with fewer stent placements (P < .001) and a nearly 30% lower rate of events at 1 year (P = .02).
While the advantage of complete revascularization, meaning PCI treatment of nonculprit as well as culprit lesions, has already been shown to be a better strategy than treatment of culprit lesions alone, FLOWER-MI is the first large study to compare FFR to angiography for guiding this approach to STEMI patients with multivessel disease, said Dr. Puymirat of Hôpital Européen George Pompidou, Paris, at the annual scientific sessions of the American College of Cardiology.
In this trial, involving multiple centers in France, STEMI patients were eligible for randomization if they had successful PCI of a culprit lesion and 50% or greater stenosis in at least one additional nonculprit lesion. The complete revascularization, whether patients were randomized to PCI guided by angiography or FFR, was performed during the index hospital admission. Patient management and follow-up was otherwise the same.
After a small number of exclusions, the intention-to-treat populations were 577 patients in the angiography-guided group and 586 in the FFR-guided group. The characteristics of the groups were well matched with an average age of about 62 years and similar rates of risk factors, such as hypertension and diabetes.
Angiography guidance just as good
The primary outcome was a composite of all-cause mortality, nonfatal MI, and unplanned revascularization. By hazard ratio, the risk of having one of these events within 1 year of PCI was numerically greater, at 32 in the FFR-guided group and 24 in the angiography-guided group, but the difference was not statistically significant (1.32; P = .31).
However, the total rate of events was low (5.5% vs. 4.2% for the angiography-guided and FFR-guided groups, respectively) and the confidence intervals were wide (95% CI, 0.78-2.23). This was also true of the components of the primary outcome.
No signal for a difference between strategies could be derived from these components, which included a higher rate of MI in the FFR-guided group (3.1% vs. 1.7%) but a lower rate of death (1.5% vs. 1.7%).
Unplanned hospitalizations leading to revascularization rates were also low (1.9% and 2.6% for angiography-guided and FFR-guided PCI, respectively), although it was reported that the rate of revascularization for nonculprit lesions was about twice as high in the FFR group (53.3% vs. 27.3%).
At 1 year, there were also low rates and no significant differences in a list of secondary outcomes that included hospitalization for recurrent ischemia or heart failure, stent thrombosis, and revascularization. As within the primary composite outcome, no pattern could be seen in the secondary events, some of which were numerically more common in the FFR-guided group and some numerically lower.
In a cost-efficacy analysis, the median per-patient cost of the FFR-guided strategy was about 500 Euros ($607) greater (8,832 vs. 8,322; P < .01), leading Dr. Puymirat to conclude that “the use of FFR for nonculprit lesions appears to be less effective but more expensive,” at least by costs derived in France.
Lack of statistical power limits interpretation
The conclusion of FLOWER-MI is that FFR-guided PCI in complete revascularization of nonculprit lesions in STEMI patients is not superior to an angiography-guided approach, but Dr. Puymirat cautioned that the low number of events precludes a definitive message.
William Fearon, MD, professor of cardiovascular medicine at Stanford (Calif.) University Medical Center, agreed. Based on his calculations, the trial was substantially underpowered. Evaluating the details of treatment in the FFR group, Dr. Fearon pointed out that a nonculprit lesion with a FFR of 0.80 or less was identified in about 55% of patients. Ultimately, 66% in the FFR group received PCI, eliminating the key distinction between strategies for the majority of patients enrolled.
“Only about one-third of the FFR-guided patients, or about 200 patients, did not receive nonculprit PCI, and therefore only in this small group could we expect a difference in outcomes from the angio-guided group,” Dr. Fearon said.
Fewer stents were placed in the FFR-guided than angiography-guided group (1.01 vs. 1.5), but Dr. Fearon suggested that it would be very difficult to show a difference in risk of events in a study of this size when event rates at 1 year reached only about 5%.
In response, Dr. Puymirat acknowledged that the rate of events for this trial, which was designed in 2015, were lower than expected. In recalculating the power needed based on the rate of events observed in FLOWER-MI, he estimated that about 8,000 patients would have been needed to show a meaningful difference in these PCI strategies.
Dr. Puymirat reports financial relationships with more than a dozen pharmaceutical companies, including Abbott, which provided some of the funding for this trial. Dr. Fearon reports financial relationships with Abbott, CathWorks, HeartFlow, and Medtronic.
FROM ACC 2021
LAAOS III: Surgical LAA closure cuts AFib stroke risk by one third
Left atrial appendage occlusion performed at the time of other heart surgery reduces the risk for stroke by about one-third in high-risk patients with atrial fibrillation (AFib), according to results of the Left Atrial Appendage Occlusion Study III (LAAOS III).
At 3.8 years’ follow-up, the primary endpoint of ischemic stroke or systemic embolism occurred in 4.8% of patients randomly assigned to left atrial appendage occlusion (LAAO) and 7.0% of those with no occlusion. This translated into a 33% relative risk reduction (hazard ratio, 0.67; 95% confidence interval, 0.53-0.85; P = .001).
In a landmark analysis, the effect was present early on but was more pronounced after the first 30 days, reducing the relative risk by 42% (HR, 0.58; 95% CI, 0.42-0.80), the researchers report.
The reduction in ongoing stroke risk was on top of oral anticoagulation (OAC) and consistent across all subgroups, Richard Whitlock, MD, PhD, professor of surgery, McMaster University, Hamilton, Ont., reported in a late-breaking trial session at the annual scientific sessions of the American College of Cardiology.
The procedure was safe and added, on average, just 6 minutes to cardiopulmonary bypass time, according to the results, simultaneously published in the New England Journal of Medicine.
“Any patient who comes to the operating room who fits the profile of a LAAOS III patient – so has atrial fibrillation and an elevated stroke risk based on their CHA2DS2-VASc score – the appendage should come off,” he said in an interview.
Commenting during the formal discussion, panelist Michael J. Mack, MD, of Baylor Health Care System in Houston, said, “This is potentially a game-changing, practice-changing study” but asked if there are any patients who shouldn’t undergo LAAO, such as those with heart failure (HF).
Dr. Whitlock said about 10%-15% of patients coming for heart surgery have a history of AFib and “as surgeons, you do need to individualize therapy. If you have a very frail patient, have concerns about tissue quality, you really need to think about how you would occlude the left atrial appendage or if you would occlude.”
Reassuringly, he noted, the data show no increase in HF hospitalizations and a beneficial effect on stroke among patients with HF and those with low ejection fractions, below 50%.
Observational data on surgical occlusion have been inconsistent, and current guidelines offer a weak recommendation in patients with AFib who have a contraindication to long-term anticoagulation. This is the first study to definitively prove that ischemic stroke is reduced by managing the left atrial appendage, he said in an interview.
“The previous percutaneous trials failed to demonstrate that; they demonstrated noninferiority but it was driven primarily by the avoidance of hemorrhagic events or strokes through taking patients off oral anticoagulation,” he said.
The results should translate into a class I guideline recommendation, he added. “This opens up a new paradigm of treatment for atrial fibrillation and stroke prevention in that it is really the first study that has looked at the additive effects of managing the left atrial appendage in addition to oral anticoagulation, and it’s protective on top of oral anticoagulation. That is a paradigm shift.”
In an accompanying editorial, Richard L. Page, MD, University of Vermont in Burlington, said the trial provides no insight on the possible benefit of surgical occlusion in patients unable to receive anticoagulation or with a lower CHA2DS2-VASc score, but he agreed a class I recommendation is likely for the population studied.
“I hope and anticipate that the results of this paper will strengthen the guideline indications for surgical left atrial appendage occlusion and will increase the number of cardiac surgeons who routinely perform this add-on procedure,” he said. “While many already perform this procedure, cardiac surgeons should now feel more comfortable that surgical left atrial appendage occlusion is indicated and supported by high-quality randomized data.”
Unfortunately, LAAOS III does not answer the question of whether patients can come off anticoagulation, but it does show surgical occlusion provides added protection from strokes, which can be huge with atrial fibrillation, Dr. Whitlock said.
“I spoke with a patient today who is an active 66-year-old individual on a [direct oral anticoagulant], and his stroke risk has been further reduced by 30%-40%, so he was ecstatic to hear the results,” Dr. Whitlock said. “I think it’s peace of mind.”
Global, nonindustry effort
LAAOS III investigators at 105 centers in 27 countries enrolled 4,811 patients undergoing cardiac surgery (mean age, 71 years; 68% male) who had a CHA2DS2-VASc score of at least 2.
In all, 4,770 were randomly assigned to no LAAO or occlusion via the preferred technique of amputation with suture closure of the stump as well as stapler occlusion, or epicardial device closure with the AtriClip (AtriCure) or TigerPaw (Maquet Medical). The treating team, researchers, and patients were blinded to assignment.
Patients were followed every 6 months with a validated stroke questionnaire. The trial was stopped early by the data safety monitoring board after the second interim analysis.
The mean CHA2DS2-VASc score was 4.2, one-third of patients had permanent AFib, 9% had a history of stroke, and more than two-thirds underwent a valve procedure, which makes LAAOS III unique, as many previous trials excluded valvular AFib, Dr. Whitlock pointed out.
Operative outcomes in the LAAO and no-LAAO groups were as follows:
- Bypass time: mean, 119 minutes vs. 113 minutes.
- Cross-clamp time: mean, 86 minutes vs. 82 minutes.
- Chest tube output: median, 520 mL vs. 500 mL.
- Reoperation for bleeding: both, 4.0%.
- Prolonged hospitalization due to HF: 5 vs. 14 events.
- 30-day mortality: 3.7% vs 4.0%.
The primary safety outcome of HF hospitalization at 3.8 years occurred in 7.7% of patients with LAAO and 6.8% without occlusion (HR, 1.13; 95% CI, 0.92-1.40), despite concerns that taking off the appendage could worsen HF risk by impairing renal clearance of salt and water.
“There’s observational data on either side of the fence, so it was an important endpoint that people were concerned about,” Dr. Whitlock told this news organization. “We had a data collection firm dedicated to admission for heart failure to really tease that out and, in the end, we saw no adverse effect.”
Although rates of ischemic stroke at 3.8 years were lower with LAAO than without (4.2% vs. 6.6%; HR, 0.62; 95% CI, 0.48-0.80), there was no difference in systemic embolism (0.3% for both) or death (22.6% vs. 22.5%).
In LAAOS III, fewer than 2% of the deaths were attributed to stroke, which is consistent with large stroke registries, Dr. Whitlock said. “Stroke is not what causes people with atrial fibrillation to die; it’s actually the progression on to heart failure.”
The positive effect on stroke was consistent across all subgroups, including sex, age, rheumatic heart disease, type of OAC at baseline, CHA2DS2-VASc score (≤4 vs. >4), type of surgery, history of heart failure or hypertension, and prior stroke/transient ischemic attack/systemic embolism.
Panelist Anne B. Curtis, MD, State University of New York at Buffalo, expressed surprise that about half of patients at baseline were not receiving anticoagulation and questioned whether event rates varied among those who did and didn’t stay on OAC.
Dr. Whitlock noted that OAC is often underused in AFib and that analyses showed the effects were consistent whether patients were on or off anticoagulants.
The study was sponsored by the Population Health Research Institute, McMaster University. Dr. Whitlock reported no relevant disclosures. Dr. Curtis reported consultant fees/honoraria from Abbott, Janssen, Medtronic, Milestone Pharmaceuticals, and Sanofi Aventis, and data safety monitoring board participation for Medtronic.
A version of this article first appeared on Medscape.com.
Left atrial appendage occlusion performed at the time of other heart surgery reduces the risk for stroke by about one-third in high-risk patients with atrial fibrillation (AFib), according to results of the Left Atrial Appendage Occlusion Study III (LAAOS III).
At 3.8 years’ follow-up, the primary endpoint of ischemic stroke or systemic embolism occurred in 4.8% of patients randomly assigned to left atrial appendage occlusion (LAAO) and 7.0% of those with no occlusion. This translated into a 33% relative risk reduction (hazard ratio, 0.67; 95% confidence interval, 0.53-0.85; P = .001).
In a landmark analysis, the effect was present early on but was more pronounced after the first 30 days, reducing the relative risk by 42% (HR, 0.58; 95% CI, 0.42-0.80), the researchers report.
The reduction in ongoing stroke risk was on top of oral anticoagulation (OAC) and consistent across all subgroups, Richard Whitlock, MD, PhD, professor of surgery, McMaster University, Hamilton, Ont., reported in a late-breaking trial session at the annual scientific sessions of the American College of Cardiology.
The procedure was safe and added, on average, just 6 minutes to cardiopulmonary bypass time, according to the results, simultaneously published in the New England Journal of Medicine.
“Any patient who comes to the operating room who fits the profile of a LAAOS III patient – so has atrial fibrillation and an elevated stroke risk based on their CHA2DS2-VASc score – the appendage should come off,” he said in an interview.
Commenting during the formal discussion, panelist Michael J. Mack, MD, of Baylor Health Care System in Houston, said, “This is potentially a game-changing, practice-changing study” but asked if there are any patients who shouldn’t undergo LAAO, such as those with heart failure (HF).
Dr. Whitlock said about 10%-15% of patients coming for heart surgery have a history of AFib and “as surgeons, you do need to individualize therapy. If you have a very frail patient, have concerns about tissue quality, you really need to think about how you would occlude the left atrial appendage or if you would occlude.”
Reassuringly, he noted, the data show no increase in HF hospitalizations and a beneficial effect on stroke among patients with HF and those with low ejection fractions, below 50%.
Observational data on surgical occlusion have been inconsistent, and current guidelines offer a weak recommendation in patients with AFib who have a contraindication to long-term anticoagulation. This is the first study to definitively prove that ischemic stroke is reduced by managing the left atrial appendage, he said in an interview.
“The previous percutaneous trials failed to demonstrate that; they demonstrated noninferiority but it was driven primarily by the avoidance of hemorrhagic events or strokes through taking patients off oral anticoagulation,” he said.
The results should translate into a class I guideline recommendation, he added. “This opens up a new paradigm of treatment for atrial fibrillation and stroke prevention in that it is really the first study that has looked at the additive effects of managing the left atrial appendage in addition to oral anticoagulation, and it’s protective on top of oral anticoagulation. That is a paradigm shift.”
In an accompanying editorial, Richard L. Page, MD, University of Vermont in Burlington, said the trial provides no insight on the possible benefit of surgical occlusion in patients unable to receive anticoagulation or with a lower CHA2DS2-VASc score, but he agreed a class I recommendation is likely for the population studied.
“I hope and anticipate that the results of this paper will strengthen the guideline indications for surgical left atrial appendage occlusion and will increase the number of cardiac surgeons who routinely perform this add-on procedure,” he said. “While many already perform this procedure, cardiac surgeons should now feel more comfortable that surgical left atrial appendage occlusion is indicated and supported by high-quality randomized data.”
Unfortunately, LAAOS III does not answer the question of whether patients can come off anticoagulation, but it does show surgical occlusion provides added protection from strokes, which can be huge with atrial fibrillation, Dr. Whitlock said.
“I spoke with a patient today who is an active 66-year-old individual on a [direct oral anticoagulant], and his stroke risk has been further reduced by 30%-40%, so he was ecstatic to hear the results,” Dr. Whitlock said. “I think it’s peace of mind.”
Global, nonindustry effort
LAAOS III investigators at 105 centers in 27 countries enrolled 4,811 patients undergoing cardiac surgery (mean age, 71 years; 68% male) who had a CHA2DS2-VASc score of at least 2.
In all, 4,770 were randomly assigned to no LAAO or occlusion via the preferred technique of amputation with suture closure of the stump as well as stapler occlusion, or epicardial device closure with the AtriClip (AtriCure) or TigerPaw (Maquet Medical). The treating team, researchers, and patients were blinded to assignment.
Patients were followed every 6 months with a validated stroke questionnaire. The trial was stopped early by the data safety monitoring board after the second interim analysis.
The mean CHA2DS2-VASc score was 4.2, one-third of patients had permanent AFib, 9% had a history of stroke, and more than two-thirds underwent a valve procedure, which makes LAAOS III unique, as many previous trials excluded valvular AFib, Dr. Whitlock pointed out.
Operative outcomes in the LAAO and no-LAAO groups were as follows:
- Bypass time: mean, 119 minutes vs. 113 minutes.
- Cross-clamp time: mean, 86 minutes vs. 82 minutes.
- Chest tube output: median, 520 mL vs. 500 mL.
- Reoperation for bleeding: both, 4.0%.
- Prolonged hospitalization due to HF: 5 vs. 14 events.
- 30-day mortality: 3.7% vs 4.0%.
The primary safety outcome of HF hospitalization at 3.8 years occurred in 7.7% of patients with LAAO and 6.8% without occlusion (HR, 1.13; 95% CI, 0.92-1.40), despite concerns that taking off the appendage could worsen HF risk by impairing renal clearance of salt and water.
“There’s observational data on either side of the fence, so it was an important endpoint that people were concerned about,” Dr. Whitlock told this news organization. “We had a data collection firm dedicated to admission for heart failure to really tease that out and, in the end, we saw no adverse effect.”
Although rates of ischemic stroke at 3.8 years were lower with LAAO than without (4.2% vs. 6.6%; HR, 0.62; 95% CI, 0.48-0.80), there was no difference in systemic embolism (0.3% for both) or death (22.6% vs. 22.5%).
In LAAOS III, fewer than 2% of the deaths were attributed to stroke, which is consistent with large stroke registries, Dr. Whitlock said. “Stroke is not what causes people with atrial fibrillation to die; it’s actually the progression on to heart failure.”
The positive effect on stroke was consistent across all subgroups, including sex, age, rheumatic heart disease, type of OAC at baseline, CHA2DS2-VASc score (≤4 vs. >4), type of surgery, history of heart failure or hypertension, and prior stroke/transient ischemic attack/systemic embolism.
Panelist Anne B. Curtis, MD, State University of New York at Buffalo, expressed surprise that about half of patients at baseline were not receiving anticoagulation and questioned whether event rates varied among those who did and didn’t stay on OAC.
Dr. Whitlock noted that OAC is often underused in AFib and that analyses showed the effects were consistent whether patients were on or off anticoagulants.
The study was sponsored by the Population Health Research Institute, McMaster University. Dr. Whitlock reported no relevant disclosures. Dr. Curtis reported consultant fees/honoraria from Abbott, Janssen, Medtronic, Milestone Pharmaceuticals, and Sanofi Aventis, and data safety monitoring board participation for Medtronic.
A version of this article first appeared on Medscape.com.
Left atrial appendage occlusion performed at the time of other heart surgery reduces the risk for stroke by about one-third in high-risk patients with atrial fibrillation (AFib), according to results of the Left Atrial Appendage Occlusion Study III (LAAOS III).
At 3.8 years’ follow-up, the primary endpoint of ischemic stroke or systemic embolism occurred in 4.8% of patients randomly assigned to left atrial appendage occlusion (LAAO) and 7.0% of those with no occlusion. This translated into a 33% relative risk reduction (hazard ratio, 0.67; 95% confidence interval, 0.53-0.85; P = .001).
In a landmark analysis, the effect was present early on but was more pronounced after the first 30 days, reducing the relative risk by 42% (HR, 0.58; 95% CI, 0.42-0.80), the researchers report.
The reduction in ongoing stroke risk was on top of oral anticoagulation (OAC) and consistent across all subgroups, Richard Whitlock, MD, PhD, professor of surgery, McMaster University, Hamilton, Ont., reported in a late-breaking trial session at the annual scientific sessions of the American College of Cardiology.
The procedure was safe and added, on average, just 6 minutes to cardiopulmonary bypass time, according to the results, simultaneously published in the New England Journal of Medicine.
“Any patient who comes to the operating room who fits the profile of a LAAOS III patient – so has atrial fibrillation and an elevated stroke risk based on their CHA2DS2-VASc score – the appendage should come off,” he said in an interview.
Commenting during the formal discussion, panelist Michael J. Mack, MD, of Baylor Health Care System in Houston, said, “This is potentially a game-changing, practice-changing study” but asked if there are any patients who shouldn’t undergo LAAO, such as those with heart failure (HF).
Dr. Whitlock said about 10%-15% of patients coming for heart surgery have a history of AFib and “as surgeons, you do need to individualize therapy. If you have a very frail patient, have concerns about tissue quality, you really need to think about how you would occlude the left atrial appendage or if you would occlude.”
Reassuringly, he noted, the data show no increase in HF hospitalizations and a beneficial effect on stroke among patients with HF and those with low ejection fractions, below 50%.
Observational data on surgical occlusion have been inconsistent, and current guidelines offer a weak recommendation in patients with AFib who have a contraindication to long-term anticoagulation. This is the first study to definitively prove that ischemic stroke is reduced by managing the left atrial appendage, he said in an interview.
“The previous percutaneous trials failed to demonstrate that; they demonstrated noninferiority but it was driven primarily by the avoidance of hemorrhagic events or strokes through taking patients off oral anticoagulation,” he said.
The results should translate into a class I guideline recommendation, he added. “This opens up a new paradigm of treatment for atrial fibrillation and stroke prevention in that it is really the first study that has looked at the additive effects of managing the left atrial appendage in addition to oral anticoagulation, and it’s protective on top of oral anticoagulation. That is a paradigm shift.”
In an accompanying editorial, Richard L. Page, MD, University of Vermont in Burlington, said the trial provides no insight on the possible benefit of surgical occlusion in patients unable to receive anticoagulation or with a lower CHA2DS2-VASc score, but he agreed a class I recommendation is likely for the population studied.
“I hope and anticipate that the results of this paper will strengthen the guideline indications for surgical left atrial appendage occlusion and will increase the number of cardiac surgeons who routinely perform this add-on procedure,” he said. “While many already perform this procedure, cardiac surgeons should now feel more comfortable that surgical left atrial appendage occlusion is indicated and supported by high-quality randomized data.”
Unfortunately, LAAOS III does not answer the question of whether patients can come off anticoagulation, but it does show surgical occlusion provides added protection from strokes, which can be huge with atrial fibrillation, Dr. Whitlock said.
“I spoke with a patient today who is an active 66-year-old individual on a [direct oral anticoagulant], and his stroke risk has been further reduced by 30%-40%, so he was ecstatic to hear the results,” Dr. Whitlock said. “I think it’s peace of mind.”
Global, nonindustry effort
LAAOS III investigators at 105 centers in 27 countries enrolled 4,811 patients undergoing cardiac surgery (mean age, 71 years; 68% male) who had a CHA2DS2-VASc score of at least 2.
In all, 4,770 were randomly assigned to no LAAO or occlusion via the preferred technique of amputation with suture closure of the stump as well as stapler occlusion, or epicardial device closure with the AtriClip (AtriCure) or TigerPaw (Maquet Medical). The treating team, researchers, and patients were blinded to assignment.
Patients were followed every 6 months with a validated stroke questionnaire. The trial was stopped early by the data safety monitoring board after the second interim analysis.
The mean CHA2DS2-VASc score was 4.2, one-third of patients had permanent AFib, 9% had a history of stroke, and more than two-thirds underwent a valve procedure, which makes LAAOS III unique, as many previous trials excluded valvular AFib, Dr. Whitlock pointed out.
Operative outcomes in the LAAO and no-LAAO groups were as follows:
- Bypass time: mean, 119 minutes vs. 113 minutes.
- Cross-clamp time: mean, 86 minutes vs. 82 minutes.
- Chest tube output: median, 520 mL vs. 500 mL.
- Reoperation for bleeding: both, 4.0%.
- Prolonged hospitalization due to HF: 5 vs. 14 events.
- 30-day mortality: 3.7% vs 4.0%.
The primary safety outcome of HF hospitalization at 3.8 years occurred in 7.7% of patients with LAAO and 6.8% without occlusion (HR, 1.13; 95% CI, 0.92-1.40), despite concerns that taking off the appendage could worsen HF risk by impairing renal clearance of salt and water.
“There’s observational data on either side of the fence, so it was an important endpoint that people were concerned about,” Dr. Whitlock told this news organization. “We had a data collection firm dedicated to admission for heart failure to really tease that out and, in the end, we saw no adverse effect.”
Although rates of ischemic stroke at 3.8 years were lower with LAAO than without (4.2% vs. 6.6%; HR, 0.62; 95% CI, 0.48-0.80), there was no difference in systemic embolism (0.3% for both) or death (22.6% vs. 22.5%).
In LAAOS III, fewer than 2% of the deaths were attributed to stroke, which is consistent with large stroke registries, Dr. Whitlock said. “Stroke is not what causes people with atrial fibrillation to die; it’s actually the progression on to heart failure.”
The positive effect on stroke was consistent across all subgroups, including sex, age, rheumatic heart disease, type of OAC at baseline, CHA2DS2-VASc score (≤4 vs. >4), type of surgery, history of heart failure or hypertension, and prior stroke/transient ischemic attack/systemic embolism.
Panelist Anne B. Curtis, MD, State University of New York at Buffalo, expressed surprise that about half of patients at baseline were not receiving anticoagulation and questioned whether event rates varied among those who did and didn’t stay on OAC.
Dr. Whitlock noted that OAC is often underused in AFib and that analyses showed the effects were consistent whether patients were on or off anticoagulants.
The study was sponsored by the Population Health Research Institute, McMaster University. Dr. Whitlock reported no relevant disclosures. Dr. Curtis reported consultant fees/honoraria from Abbott, Janssen, Medtronic, Milestone Pharmaceuticals, and Sanofi Aventis, and data safety monitoring board participation for Medtronic.
A version of this article first appeared on Medscape.com.
FROM ACC 2021
PARADISE-MI: Sacubitril/valsartan can’t beat ramipril in patients with acute MI
Treatment with sacubitril/valsartan, a pillar of therapy for patients with chronic heart failure with below-normal ejection fraction, came suggestively close to showing efficacy for preventing cardiovascular death or heart failure events in patients who have just had an MI but have no history of heart failure in a controlled trial with more than 5,600 patients.
Although sacubitril/valsartan (Entresto) fell short of producing a significant benefit, it did show good safety that was similar to the study’s comparator treatment, ramipril, an agent from the angiotensin-converting enzyme inhibitor class that is a mainstay of treatment in these patients.
“To say that, with no run-in, sacubitril/valsartan is as well tolerated and as safe as one of the best-studied ACE inhibitors – ramipril – in acutely ill MI patients, is a big statement,” said Marc A. Pfeffer, MD, at the annual scientific sessions of the American College of Cardiology. This high level of safety without gradual uptitration of sacubitril/valsartan (Entresto) “should lower barriers” to broader use of the dual-drug formulation for its approved indication in patients with chronic heart failure, especially patients with a left ventricular ejection fraction that is below normal. In addition, results from the PARADISE-MI trial suggested that “patients seemed to benefit before they develop heart failure. We couldn’t prove that, but we should build on this, and make it easier for patients to use this treatment,” Dr. Pfeffer said during a press briefing following his talk at the sessions.
Preventing heart failures to come
Treatment with sacubitril/valsartan in acute MI patients within a few days of their event “is perhaps addressing prevention of the heart failure that’s to come,” commented Lynne W. Stevenson, MD, designated discussant for the report and professor of medicine at Vanderbilt University Medical Center in Nashville. “Patients who are destined to develop heart failure are beginning their treatment early. The subgroup analyses suggest that it’s the sicker patients who benefited the most,” she said.
But Dr. Pfeffer stressed that “I don’t think this is a subgroup discussion. I would like to pursue this, but that’s up to the sponsor,” Novartis, the company that markets sacubitril/valsartan.
‘Exceedingly reassuring’ safety
The safety data that Dr. Pfeffer reported “are exceedingly reassuring. We didn’t see a signal of harm, and in some of the exploratory endpoints there was some evidence of benefit, so we need to encourage you to continue,” commented Mary N. Walsh, MD, medical director of the heart failure and cardiac transplantation program at Ascension St. Vincent Heart Center of Indiana in Indianapolis.
The PARADISE-MI (Prospective ARNI vs. ACE Inhibitor Trial to Determine Superiority in Reducing Heart Failure Events After MI) trial enrolled 5,669 patients with no history of heart failure within an average of 4 days following an acute MI at 495 sites in 41 countries during 2016-2020, with 8% of enrolled patients from the United States. Patients averaged 64 years of age, about three-quarters were men, about 43% had a history of diabetes, and only 1% were Black; Dr. Pfeffer noted that this is because most patients came from countries with low Black populations. The enrollment criteria required a left ventricular ejection fraction no greater than 40%, and among the enrolled patients this averaged about 37%.
A 10% nonsignificant relative risk reduction for the primary endpoint
The study’s primary endpoint was the combined first-event rate of cardiovascular death, hospitalization for heart failure, or an outpatient visit for heart failure. During a median follow-up of 23 months, this occurred at a rate of 7.4/100 patient years in the ramipril arm and 6.7/100 patient years in the sacubitril/valsartan arm, a 10% relative risk reduction with sacubitril/valsartan that was not significant, which meant all other efficacy analyses were exploratory, Dr. Pfeffer stressed.
Several secondary efficacy analyses showed significant benefits from sacubitril/valsartan, compared with ramipril, including the total number of events that comprised the primary endpoint, with a 21% relative risk reduction associated with sacubitril/valsartan, as well as investigator-reported events. The primary-endpoint benefit from sacubitril/valsartan was also significant in two subgroup analyses: patients aged 65 years or older (roughly half the study cohort), who had a 24% relative risk reduction on sacubitril/valsartan, compared with ramipril, and the 88% of patients who received treatment with percutaneous coronary intervention for their acute MI, who had a 19% relative risk reduction on sacubitril/valsartan, compared with patients who received ramipril.
The study’s safety data showed nearly identical rates in the two treatment arms for total adverse events, serious adverse events, adverse events that led to stopping the study drug, as well as in laboratory measures. The biggest between-treatment differences were a modest excess of hypotension on sacubitril valsartan, 28%, compared with 22% on ramipril, and a modest excess rate of cough on ramipril, 13%, compared with 9% on sacubitril/valsartan.
The added insight the results provide about sacubitril/valsartan comes at a time when U.S. patients continue to struggle to get health insurance coverage for an agent that has been approved for U.S. use in treating heart failure since 2015.
“Our patients do not have access to this important treatment,” declared Dr. Walsh during the press briefing. “The prior authorization process is unbelievable, and some patients have no access unless they pay the full cost on their own. This is an important, real-world problem that we face with this drug.”
PARADISE-MI was sponsored by Novartis, the company that markets sacubitril/valsartan (Entresto). Dr. Pfeffer has received research funding from and is a consultant to Novartis. He is also a consultant to AstraZeneca, Boehringer Ingelheim, Corvidia, DalCor, Eli Lilly, GlaxoSmithKline, Novo Nordisk, Peerbridge, and Sanofi, and he holds equity in DalCor and Peerbridge. Dr. Stevenson has received honoraria from LivaNova and has received research support from Abbott. Dr. Walsh had no disclosures.
Treatment with sacubitril/valsartan, a pillar of therapy for patients with chronic heart failure with below-normal ejection fraction, came suggestively close to showing efficacy for preventing cardiovascular death or heart failure events in patients who have just had an MI but have no history of heart failure in a controlled trial with more than 5,600 patients.
Although sacubitril/valsartan (Entresto) fell short of producing a significant benefit, it did show good safety that was similar to the study’s comparator treatment, ramipril, an agent from the angiotensin-converting enzyme inhibitor class that is a mainstay of treatment in these patients.
“To say that, with no run-in, sacubitril/valsartan is as well tolerated and as safe as one of the best-studied ACE inhibitors – ramipril – in acutely ill MI patients, is a big statement,” said Marc A. Pfeffer, MD, at the annual scientific sessions of the American College of Cardiology. This high level of safety without gradual uptitration of sacubitril/valsartan (Entresto) “should lower barriers” to broader use of the dual-drug formulation for its approved indication in patients with chronic heart failure, especially patients with a left ventricular ejection fraction that is below normal. In addition, results from the PARADISE-MI trial suggested that “patients seemed to benefit before they develop heart failure. We couldn’t prove that, but we should build on this, and make it easier for patients to use this treatment,” Dr. Pfeffer said during a press briefing following his talk at the sessions.
Preventing heart failures to come
Treatment with sacubitril/valsartan in acute MI patients within a few days of their event “is perhaps addressing prevention of the heart failure that’s to come,” commented Lynne W. Stevenson, MD, designated discussant for the report and professor of medicine at Vanderbilt University Medical Center in Nashville. “Patients who are destined to develop heart failure are beginning their treatment early. The subgroup analyses suggest that it’s the sicker patients who benefited the most,” she said.
But Dr. Pfeffer stressed that “I don’t think this is a subgroup discussion. I would like to pursue this, but that’s up to the sponsor,” Novartis, the company that markets sacubitril/valsartan.
‘Exceedingly reassuring’ safety
The safety data that Dr. Pfeffer reported “are exceedingly reassuring. We didn’t see a signal of harm, and in some of the exploratory endpoints there was some evidence of benefit, so we need to encourage you to continue,” commented Mary N. Walsh, MD, medical director of the heart failure and cardiac transplantation program at Ascension St. Vincent Heart Center of Indiana in Indianapolis.
The PARADISE-MI (Prospective ARNI vs. ACE Inhibitor Trial to Determine Superiority in Reducing Heart Failure Events After MI) trial enrolled 5,669 patients with no history of heart failure within an average of 4 days following an acute MI at 495 sites in 41 countries during 2016-2020, with 8% of enrolled patients from the United States. Patients averaged 64 years of age, about three-quarters were men, about 43% had a history of diabetes, and only 1% were Black; Dr. Pfeffer noted that this is because most patients came from countries with low Black populations. The enrollment criteria required a left ventricular ejection fraction no greater than 40%, and among the enrolled patients this averaged about 37%.
A 10% nonsignificant relative risk reduction for the primary endpoint
The study’s primary endpoint was the combined first-event rate of cardiovascular death, hospitalization for heart failure, or an outpatient visit for heart failure. During a median follow-up of 23 months, this occurred at a rate of 7.4/100 patient years in the ramipril arm and 6.7/100 patient years in the sacubitril/valsartan arm, a 10% relative risk reduction with sacubitril/valsartan that was not significant, which meant all other efficacy analyses were exploratory, Dr. Pfeffer stressed.
Several secondary efficacy analyses showed significant benefits from sacubitril/valsartan, compared with ramipril, including the total number of events that comprised the primary endpoint, with a 21% relative risk reduction associated with sacubitril/valsartan, as well as investigator-reported events. The primary-endpoint benefit from sacubitril/valsartan was also significant in two subgroup analyses: patients aged 65 years or older (roughly half the study cohort), who had a 24% relative risk reduction on sacubitril/valsartan, compared with ramipril, and the 88% of patients who received treatment with percutaneous coronary intervention for their acute MI, who had a 19% relative risk reduction on sacubitril/valsartan, compared with patients who received ramipril.
The study’s safety data showed nearly identical rates in the two treatment arms for total adverse events, serious adverse events, adverse events that led to stopping the study drug, as well as in laboratory measures. The biggest between-treatment differences were a modest excess of hypotension on sacubitril valsartan, 28%, compared with 22% on ramipril, and a modest excess rate of cough on ramipril, 13%, compared with 9% on sacubitril/valsartan.
The added insight the results provide about sacubitril/valsartan comes at a time when U.S. patients continue to struggle to get health insurance coverage for an agent that has been approved for U.S. use in treating heart failure since 2015.
“Our patients do not have access to this important treatment,” declared Dr. Walsh during the press briefing. “The prior authorization process is unbelievable, and some patients have no access unless they pay the full cost on their own. This is an important, real-world problem that we face with this drug.”
PARADISE-MI was sponsored by Novartis, the company that markets sacubitril/valsartan (Entresto). Dr. Pfeffer has received research funding from and is a consultant to Novartis. He is also a consultant to AstraZeneca, Boehringer Ingelheim, Corvidia, DalCor, Eli Lilly, GlaxoSmithKline, Novo Nordisk, Peerbridge, and Sanofi, and he holds equity in DalCor and Peerbridge. Dr. Stevenson has received honoraria from LivaNova and has received research support from Abbott. Dr. Walsh had no disclosures.
Treatment with sacubitril/valsartan, a pillar of therapy for patients with chronic heart failure with below-normal ejection fraction, came suggestively close to showing efficacy for preventing cardiovascular death or heart failure events in patients who have just had an MI but have no history of heart failure in a controlled trial with more than 5,600 patients.
Although sacubitril/valsartan (Entresto) fell short of producing a significant benefit, it did show good safety that was similar to the study’s comparator treatment, ramipril, an agent from the angiotensin-converting enzyme inhibitor class that is a mainstay of treatment in these patients.
“To say that, with no run-in, sacubitril/valsartan is as well tolerated and as safe as one of the best-studied ACE inhibitors – ramipril – in acutely ill MI patients, is a big statement,” said Marc A. Pfeffer, MD, at the annual scientific sessions of the American College of Cardiology. This high level of safety without gradual uptitration of sacubitril/valsartan (Entresto) “should lower barriers” to broader use of the dual-drug formulation for its approved indication in patients with chronic heart failure, especially patients with a left ventricular ejection fraction that is below normal. In addition, results from the PARADISE-MI trial suggested that “patients seemed to benefit before they develop heart failure. We couldn’t prove that, but we should build on this, and make it easier for patients to use this treatment,” Dr. Pfeffer said during a press briefing following his talk at the sessions.
Preventing heart failures to come
Treatment with sacubitril/valsartan in acute MI patients within a few days of their event “is perhaps addressing prevention of the heart failure that’s to come,” commented Lynne W. Stevenson, MD, designated discussant for the report and professor of medicine at Vanderbilt University Medical Center in Nashville. “Patients who are destined to develop heart failure are beginning their treatment early. The subgroup analyses suggest that it’s the sicker patients who benefited the most,” she said.
But Dr. Pfeffer stressed that “I don’t think this is a subgroup discussion. I would like to pursue this, but that’s up to the sponsor,” Novartis, the company that markets sacubitril/valsartan.
‘Exceedingly reassuring’ safety
The safety data that Dr. Pfeffer reported “are exceedingly reassuring. We didn’t see a signal of harm, and in some of the exploratory endpoints there was some evidence of benefit, so we need to encourage you to continue,” commented Mary N. Walsh, MD, medical director of the heart failure and cardiac transplantation program at Ascension St. Vincent Heart Center of Indiana in Indianapolis.
The PARADISE-MI (Prospective ARNI vs. ACE Inhibitor Trial to Determine Superiority in Reducing Heart Failure Events After MI) trial enrolled 5,669 patients with no history of heart failure within an average of 4 days following an acute MI at 495 sites in 41 countries during 2016-2020, with 8% of enrolled patients from the United States. Patients averaged 64 years of age, about three-quarters were men, about 43% had a history of diabetes, and only 1% were Black; Dr. Pfeffer noted that this is because most patients came from countries with low Black populations. The enrollment criteria required a left ventricular ejection fraction no greater than 40%, and among the enrolled patients this averaged about 37%.
A 10% nonsignificant relative risk reduction for the primary endpoint
The study’s primary endpoint was the combined first-event rate of cardiovascular death, hospitalization for heart failure, or an outpatient visit for heart failure. During a median follow-up of 23 months, this occurred at a rate of 7.4/100 patient years in the ramipril arm and 6.7/100 patient years in the sacubitril/valsartan arm, a 10% relative risk reduction with sacubitril/valsartan that was not significant, which meant all other efficacy analyses were exploratory, Dr. Pfeffer stressed.
Several secondary efficacy analyses showed significant benefits from sacubitril/valsartan, compared with ramipril, including the total number of events that comprised the primary endpoint, with a 21% relative risk reduction associated with sacubitril/valsartan, as well as investigator-reported events. The primary-endpoint benefit from sacubitril/valsartan was also significant in two subgroup analyses: patients aged 65 years or older (roughly half the study cohort), who had a 24% relative risk reduction on sacubitril/valsartan, compared with ramipril, and the 88% of patients who received treatment with percutaneous coronary intervention for their acute MI, who had a 19% relative risk reduction on sacubitril/valsartan, compared with patients who received ramipril.
The study’s safety data showed nearly identical rates in the two treatment arms for total adverse events, serious adverse events, adverse events that led to stopping the study drug, as well as in laboratory measures. The biggest between-treatment differences were a modest excess of hypotension on sacubitril valsartan, 28%, compared with 22% on ramipril, and a modest excess rate of cough on ramipril, 13%, compared with 9% on sacubitril/valsartan.
The added insight the results provide about sacubitril/valsartan comes at a time when U.S. patients continue to struggle to get health insurance coverage for an agent that has been approved for U.S. use in treating heart failure since 2015.
“Our patients do not have access to this important treatment,” declared Dr. Walsh during the press briefing. “The prior authorization process is unbelievable, and some patients have no access unless they pay the full cost on their own. This is an important, real-world problem that we face with this drug.”
PARADISE-MI was sponsored by Novartis, the company that markets sacubitril/valsartan (Entresto). Dr. Pfeffer has received research funding from and is a consultant to Novartis. He is also a consultant to AstraZeneca, Boehringer Ingelheim, Corvidia, DalCor, Eli Lilly, GlaxoSmithKline, Novo Nordisk, Peerbridge, and Sanofi, and he holds equity in DalCor and Peerbridge. Dr. Stevenson has received honoraria from LivaNova and has received research support from Abbott. Dr. Walsh had no disclosures.
FROM ACC 2021




















