User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
FDA expands rimegepant indication to include migraine prevention
Last year, rimegepant became the first calcitonin gene-related peptide (CGRP) receptor antagonist, available in a fast-acting orally disintegrating tablet, to be approved for the acute treatment of migraine with or without aura in adults.
Rimegepant is currently the only migraine medication approved to both treat acute migraine attacks and help prevent future migraine attacks.
The new indication allows for use of rimegepant for preventive treatment in adults with episodic migraine (more than 15 migraine days per month). Rimegepant may be used for up to 18 doses per month, which includes both acute and preventive therapy.
In a phase 2/3 study, oral rimegepant was superior to placebo in reducing monthly migraine days. About half of adults who took rimegepant experienced a 50% or greater reduction in the number of days of moderate to severe migraines per month.
The most common adverse effects of rimegepant therapy were nausea (2.7%) and stomach pain or indigestion (2.4%).
The FDA approval of rimegepant for the preventive treatment of migraine, along with its acute treatment indication, is “one of the most ground-breaking things to happen to migraine treatment in my 40 years of practicing headache medicine,” Peter J. Goadsby, MD, PhD, an investigator in the prevention study, said in a company news release.
“To have one medication patients can use to treat and prevent migraine will likely change the treatment paradigm for many of the millions of people who live with migraine,” said Dr. Goadsby, professor of neurology, University of California, Los Angeles and King’s College, London.
A version of this article first appeared on Medscape.com.
Last year, rimegepant became the first calcitonin gene-related peptide (CGRP) receptor antagonist, available in a fast-acting orally disintegrating tablet, to be approved for the acute treatment of migraine with or without aura in adults.
Rimegepant is currently the only migraine medication approved to both treat acute migraine attacks and help prevent future migraine attacks.
The new indication allows for use of rimegepant for preventive treatment in adults with episodic migraine (more than 15 migraine days per month). Rimegepant may be used for up to 18 doses per month, which includes both acute and preventive therapy.
In a phase 2/3 study, oral rimegepant was superior to placebo in reducing monthly migraine days. About half of adults who took rimegepant experienced a 50% or greater reduction in the number of days of moderate to severe migraines per month.
The most common adverse effects of rimegepant therapy were nausea (2.7%) and stomach pain or indigestion (2.4%).
The FDA approval of rimegepant for the preventive treatment of migraine, along with its acute treatment indication, is “one of the most ground-breaking things to happen to migraine treatment in my 40 years of practicing headache medicine,” Peter J. Goadsby, MD, PhD, an investigator in the prevention study, said in a company news release.
“To have one medication patients can use to treat and prevent migraine will likely change the treatment paradigm for many of the millions of people who live with migraine,” said Dr. Goadsby, professor of neurology, University of California, Los Angeles and King’s College, London.
A version of this article first appeared on Medscape.com.
Last year, rimegepant became the first calcitonin gene-related peptide (CGRP) receptor antagonist, available in a fast-acting orally disintegrating tablet, to be approved for the acute treatment of migraine with or without aura in adults.
Rimegepant is currently the only migraine medication approved to both treat acute migraine attacks and help prevent future migraine attacks.
The new indication allows for use of rimegepant for preventive treatment in adults with episodic migraine (more than 15 migraine days per month). Rimegepant may be used for up to 18 doses per month, which includes both acute and preventive therapy.
In a phase 2/3 study, oral rimegepant was superior to placebo in reducing monthly migraine days. About half of adults who took rimegepant experienced a 50% or greater reduction in the number of days of moderate to severe migraines per month.
The most common adverse effects of rimegepant therapy were nausea (2.7%) and stomach pain or indigestion (2.4%).
The FDA approval of rimegepant for the preventive treatment of migraine, along with its acute treatment indication, is “one of the most ground-breaking things to happen to migraine treatment in my 40 years of practicing headache medicine,” Peter J. Goadsby, MD, PhD, an investigator in the prevention study, said in a company news release.
“To have one medication patients can use to treat and prevent migraine will likely change the treatment paradigm for many of the millions of people who live with migraine,” said Dr. Goadsby, professor of neurology, University of California, Los Angeles and King’s College, London.
A version of this article first appeared on Medscape.com.
Subclinical myocarditis found in some athletes post COVID
Myocarditis is present in a small percentage of competitive athletes after COVID-19 infection, even in those without symptoms, new research suggests.
In a cohort study of 1,597 competitive collegiate athletes undergoing comprehensive cardiovascular testing in the United States, the prevalence of clinical myocarditis based on a symptom-based screening strategy was only 0.31%.
But screening with cardiac MRI increased the prevalence of clinical and subclinical myocarditis by a factor of 7.4, to 2.3%, the authors reported.
The findings are published online May 27, 2021, in JAMA Cardiology.
“It was the largest study to evaluate college athletes who have had COVID with extensive cardiac testing, including MRI, and this gave us a very objective look at the cardiac findings, as they were not purely based upon a subjective evaluation of symptoms,” lead investigator Curt J. Daniels, MD, professor at Ohio State University Wexner Medical Center, Columbus, said in an interview.
“Unfortunately, our study showed that athletes can be asymptomatic, or at least not report symptoms. This is a very subjective feature, and we don’t know if they don’t report symptoms because they didn’t want to get tested. That is why we took a very objective approach,” Dr. Daniels said.
The finding that more than half of the asymptomatic athletes had myocarditis, or as the investigators called it, “subclinical myocarditis,” was a surprise, he acknowledged.
“More than half of the athletes found to have myocarditis reported no symptoms, and yes, that was a surprise, because prior to this study, the protocols that had been published stated that you had to have symptoms to even enter into the protocol for cardiac MRI. But, as our ... paper shows, if we had followed that protocol, we only would have found about 5 cases of myocarditis, as opposed to the total of 37 we found with cardiac MRI,” Dr. Daniels said.
In October 2020, the American College of Cardiology’s Sports and Exercise Council recommended that cardiac MRI be limited to athletes who exhibited symptoms as part of their guide to ensuring a safe return to play.
As reported by this news organization the council recommended a tiered approach to screening based on the presence of symptoms, followed by electrocardiography, injury biomarkers, and echocardiography. Any abnormalities detected were to be further characterized by the selective use of cardiac MRI.
At the time, there were relatively few data to support the recommendations, and all stakeholders called for larger datasets to better drive informed recommendations in the future.
In the current study, Dr. Daniels and associates conducted comprehensive cardiac screening – including ECG, troponin testing, echocardiography, and cardiac MRI – of 1,597 college athlete survivors of COVID-19.
The athletes were part of the Big Ten athletic conference, which consists of 13 major American universities.
Cardiac MRI revealed that 37 (2.3%) of these athletes demonstrated diagnostic criteria for COVID-19 myocarditis; of these, 20 had no cardiovascular symptoms and had normal ECGs, echocardiography, and troponin test results.
“These patients would not have been identified without CMR imaging. If we were going according to the older protocol, we would not have made this discovery. Cardiac MRI is the most sensitive and specific test for myocardial inflammation, there is no argument about that,” Dr. Daniels said.
The catch is, cardiac MRI is expensive and often difficult to access, especially in remote, rural, or other underserviced areas.
“You can’t get an MRI for every person who has had COVID, it’s just not feasible,” Dr. Daniels said. “We are not advocating that everybody get an MRI. But we do hope that our study creates awareness among clinicians and athletes themselves that if you’ve had COVID, even if you’re asymptomatic, there may be some heart changes. So be aware when you start to exercise again, if you have any symptoms, pause and seek medical care.”
Kudos to the sports cardiology community
In an accompanying editorial, James E. Udelson, MD, Ethan J. Rowin, MD, and Barry J. Maron, MD, from the CardioVascular Center at Tufts Medical Center, Boston, applauded the sports cardiology community for its diligence in acquiring and publishing data about the post–COVID-19 prevalence of cardiac abnormalities in competitive athletes.
“It is a real tribute to the sports cardiology community. There has been an amazing growth of information, and they not only gathered this information, they analyzed and published it, starting out with a study of 29 or 30 athletes, and now thousands,” Dr. Udelson said in an interview.
At the start of the pandemic, it appeared that 15%-20% of athletes had myocarditis, and athletic conferences were discussing canceling sports events.
However, with greater numbers comes a more accurate picture of the extent of the problem.
“Once you get thousands of subjects in these studies, you can hone in on what the real number is, so now we understand that if you screen everybody with a cardiac MRI, 1%, 2%, or 3% will have some evidence of what looks like myocarditis,” he said.
Dr. Udelson agreed that doing cardiac imaging in everyone is not feasible.
“This study looked at a very large number of people who all had an MRI, but that doesn’t mean everyone should have them. If you just do an echo, an EKG, and a troponin test, and if everything is normal, which is kind of what current recommendations are, this paper tells us that we are going to miss one or two people out of a hundred, and that might be okay,” he said. “So, if you are at a huge university that has a large medical center and you want to screen all your athletes with MRI, great. But if you’re at a high school in a remote area, you know that the alternative, not having an MRI, isn’t so bad, either.”
A version of this article first appeared on Medscape.com.
Myocarditis is present in a small percentage of competitive athletes after COVID-19 infection, even in those without symptoms, new research suggests.
In a cohort study of 1,597 competitive collegiate athletes undergoing comprehensive cardiovascular testing in the United States, the prevalence of clinical myocarditis based on a symptom-based screening strategy was only 0.31%.
But screening with cardiac MRI increased the prevalence of clinical and subclinical myocarditis by a factor of 7.4, to 2.3%, the authors reported.
The findings are published online May 27, 2021, in JAMA Cardiology.
“It was the largest study to evaluate college athletes who have had COVID with extensive cardiac testing, including MRI, and this gave us a very objective look at the cardiac findings, as they were not purely based upon a subjective evaluation of symptoms,” lead investigator Curt J. Daniels, MD, professor at Ohio State University Wexner Medical Center, Columbus, said in an interview.
“Unfortunately, our study showed that athletes can be asymptomatic, or at least not report symptoms. This is a very subjective feature, and we don’t know if they don’t report symptoms because they didn’t want to get tested. That is why we took a very objective approach,” Dr. Daniels said.
The finding that more than half of the asymptomatic athletes had myocarditis, or as the investigators called it, “subclinical myocarditis,” was a surprise, he acknowledged.
“More than half of the athletes found to have myocarditis reported no symptoms, and yes, that was a surprise, because prior to this study, the protocols that had been published stated that you had to have symptoms to even enter into the protocol for cardiac MRI. But, as our ... paper shows, if we had followed that protocol, we only would have found about 5 cases of myocarditis, as opposed to the total of 37 we found with cardiac MRI,” Dr. Daniels said.
In October 2020, the American College of Cardiology’s Sports and Exercise Council recommended that cardiac MRI be limited to athletes who exhibited symptoms as part of their guide to ensuring a safe return to play.
As reported by this news organization the council recommended a tiered approach to screening based on the presence of symptoms, followed by electrocardiography, injury biomarkers, and echocardiography. Any abnormalities detected were to be further characterized by the selective use of cardiac MRI.
At the time, there were relatively few data to support the recommendations, and all stakeholders called for larger datasets to better drive informed recommendations in the future.
In the current study, Dr. Daniels and associates conducted comprehensive cardiac screening – including ECG, troponin testing, echocardiography, and cardiac MRI – of 1,597 college athlete survivors of COVID-19.
The athletes were part of the Big Ten athletic conference, which consists of 13 major American universities.
Cardiac MRI revealed that 37 (2.3%) of these athletes demonstrated diagnostic criteria for COVID-19 myocarditis; of these, 20 had no cardiovascular symptoms and had normal ECGs, echocardiography, and troponin test results.
“These patients would not have been identified without CMR imaging. If we were going according to the older protocol, we would not have made this discovery. Cardiac MRI is the most sensitive and specific test for myocardial inflammation, there is no argument about that,” Dr. Daniels said.
The catch is, cardiac MRI is expensive and often difficult to access, especially in remote, rural, or other underserviced areas.
“You can’t get an MRI for every person who has had COVID, it’s just not feasible,” Dr. Daniels said. “We are not advocating that everybody get an MRI. But we do hope that our study creates awareness among clinicians and athletes themselves that if you’ve had COVID, even if you’re asymptomatic, there may be some heart changes. So be aware when you start to exercise again, if you have any symptoms, pause and seek medical care.”
Kudos to the sports cardiology community
In an accompanying editorial, James E. Udelson, MD, Ethan J. Rowin, MD, and Barry J. Maron, MD, from the CardioVascular Center at Tufts Medical Center, Boston, applauded the sports cardiology community for its diligence in acquiring and publishing data about the post–COVID-19 prevalence of cardiac abnormalities in competitive athletes.
“It is a real tribute to the sports cardiology community. There has been an amazing growth of information, and they not only gathered this information, they analyzed and published it, starting out with a study of 29 or 30 athletes, and now thousands,” Dr. Udelson said in an interview.
At the start of the pandemic, it appeared that 15%-20% of athletes had myocarditis, and athletic conferences were discussing canceling sports events.
However, with greater numbers comes a more accurate picture of the extent of the problem.
“Once you get thousands of subjects in these studies, you can hone in on what the real number is, so now we understand that if you screen everybody with a cardiac MRI, 1%, 2%, or 3% will have some evidence of what looks like myocarditis,” he said.
Dr. Udelson agreed that doing cardiac imaging in everyone is not feasible.
“This study looked at a very large number of people who all had an MRI, but that doesn’t mean everyone should have them. If you just do an echo, an EKG, and a troponin test, and if everything is normal, which is kind of what current recommendations are, this paper tells us that we are going to miss one or two people out of a hundred, and that might be okay,” he said. “So, if you are at a huge university that has a large medical center and you want to screen all your athletes with MRI, great. But if you’re at a high school in a remote area, you know that the alternative, not having an MRI, isn’t so bad, either.”
A version of this article first appeared on Medscape.com.
Myocarditis is present in a small percentage of competitive athletes after COVID-19 infection, even in those without symptoms, new research suggests.
In a cohort study of 1,597 competitive collegiate athletes undergoing comprehensive cardiovascular testing in the United States, the prevalence of clinical myocarditis based on a symptom-based screening strategy was only 0.31%.
But screening with cardiac MRI increased the prevalence of clinical and subclinical myocarditis by a factor of 7.4, to 2.3%, the authors reported.
The findings are published online May 27, 2021, in JAMA Cardiology.
“It was the largest study to evaluate college athletes who have had COVID with extensive cardiac testing, including MRI, and this gave us a very objective look at the cardiac findings, as they were not purely based upon a subjective evaluation of symptoms,” lead investigator Curt J. Daniels, MD, professor at Ohio State University Wexner Medical Center, Columbus, said in an interview.
“Unfortunately, our study showed that athletes can be asymptomatic, or at least not report symptoms. This is a very subjective feature, and we don’t know if they don’t report symptoms because they didn’t want to get tested. That is why we took a very objective approach,” Dr. Daniels said.
The finding that more than half of the asymptomatic athletes had myocarditis, or as the investigators called it, “subclinical myocarditis,” was a surprise, he acknowledged.
“More than half of the athletes found to have myocarditis reported no symptoms, and yes, that was a surprise, because prior to this study, the protocols that had been published stated that you had to have symptoms to even enter into the protocol for cardiac MRI. But, as our ... paper shows, if we had followed that protocol, we only would have found about 5 cases of myocarditis, as opposed to the total of 37 we found with cardiac MRI,” Dr. Daniels said.
In October 2020, the American College of Cardiology’s Sports and Exercise Council recommended that cardiac MRI be limited to athletes who exhibited symptoms as part of their guide to ensuring a safe return to play.
As reported by this news organization the council recommended a tiered approach to screening based on the presence of symptoms, followed by electrocardiography, injury biomarkers, and echocardiography. Any abnormalities detected were to be further characterized by the selective use of cardiac MRI.
At the time, there were relatively few data to support the recommendations, and all stakeholders called for larger datasets to better drive informed recommendations in the future.
In the current study, Dr. Daniels and associates conducted comprehensive cardiac screening – including ECG, troponin testing, echocardiography, and cardiac MRI – of 1,597 college athlete survivors of COVID-19.
The athletes were part of the Big Ten athletic conference, which consists of 13 major American universities.
Cardiac MRI revealed that 37 (2.3%) of these athletes demonstrated diagnostic criteria for COVID-19 myocarditis; of these, 20 had no cardiovascular symptoms and had normal ECGs, echocardiography, and troponin test results.
“These patients would not have been identified without CMR imaging. If we were going according to the older protocol, we would not have made this discovery. Cardiac MRI is the most sensitive and specific test for myocardial inflammation, there is no argument about that,” Dr. Daniels said.
The catch is, cardiac MRI is expensive and often difficult to access, especially in remote, rural, or other underserviced areas.
“You can’t get an MRI for every person who has had COVID, it’s just not feasible,” Dr. Daniels said. “We are not advocating that everybody get an MRI. But we do hope that our study creates awareness among clinicians and athletes themselves that if you’ve had COVID, even if you’re asymptomatic, there may be some heart changes. So be aware when you start to exercise again, if you have any symptoms, pause and seek medical care.”
Kudos to the sports cardiology community
In an accompanying editorial, James E. Udelson, MD, Ethan J. Rowin, MD, and Barry J. Maron, MD, from the CardioVascular Center at Tufts Medical Center, Boston, applauded the sports cardiology community for its diligence in acquiring and publishing data about the post–COVID-19 prevalence of cardiac abnormalities in competitive athletes.
“It is a real tribute to the sports cardiology community. There has been an amazing growth of information, and they not only gathered this information, they analyzed and published it, starting out with a study of 29 or 30 athletes, and now thousands,” Dr. Udelson said in an interview.
At the start of the pandemic, it appeared that 15%-20% of athletes had myocarditis, and athletic conferences were discussing canceling sports events.
However, with greater numbers comes a more accurate picture of the extent of the problem.
“Once you get thousands of subjects in these studies, you can hone in on what the real number is, so now we understand that if you screen everybody with a cardiac MRI, 1%, 2%, or 3% will have some evidence of what looks like myocarditis,” he said.
Dr. Udelson agreed that doing cardiac imaging in everyone is not feasible.
“This study looked at a very large number of people who all had an MRI, but that doesn’t mean everyone should have them. If you just do an echo, an EKG, and a troponin test, and if everything is normal, which is kind of what current recommendations are, this paper tells us that we are going to miss one or two people out of a hundred, and that might be okay,” he said. “So, if you are at a huge university that has a large medical center and you want to screen all your athletes with MRI, great. But if you’re at a high school in a remote area, you know that the alternative, not having an MRI, isn’t so bad, either.”
A version of this article first appeared on Medscape.com.
Pericardial fat an independent risk factor for heart failure
Pericardial fat is associated with a heightened risk for heart failure, particularly in women, new research suggests.
In a prospective cohort study of nearly 7,000 individuals, excess pericardial fat was linked to a higher risk for heart failure, even after adjustment for established risk factors for heart failure.
Women with high pericardial fat volume (PFV), defined as more than 70 cm3 or 2.4 fluid ounces, had double the risk of developing heart failure. For men, high PFV, defined as more than 120 cm3 or 4.0 fluid ounces, was associated with a 50% increase in the risk for heart failure.
The findings were published in the Journal of the American College of Cardiology.
“People will ask why should they measure fat around the heart. Why can’t they just take the waist circumference or body mass index as a measure for increased risk?” lead author Satish Kenchaiah, MD, MPH, Icahn School of Medicine at Mount Sinai, New York, said in an interview.
“Yet, when we adjusted for waist circumference, hip circumference, waist to hip ratio, and other known variables, pericardial fat was still associated with an increased risk of heart failure. This tells me that it is not just overall fat in the body but something about its location around the heart that is playing a role,” Dr. Kenchaiah said.
“Now that we have found an association between any amount of fat around the pericardium and heart failure, it gives us an impetus to build future research on identifying how exactly these fat deposits influence the development of cardiomyopathy,” he said.
Dr. Kenchaiah and colleagues investigated the association of pericardial fat with incident heart failure by examining chest CT scans from 6,785 participants (3,584 women and 3,201 men aged 45-84 years) in the Multi-Ethnic Study of Atherosclerosis.
The participants were from four different ethnic groups: 38% were White; 28% were Black, 22% were Hispanic, and 12% were Chinese American. They were recruited between July 17, 2000, and Aug. 31, 2002, from six communities in the United States: Baltimore and Baltimore County; Chicago; Forsyth County, N.C.; Los Angeles County northern Manhattan and the Bronx, New York; and St. Paul, Minn.
All participants were free of cardiovascular disease at baseline.
The researchers followed participants for more than 17 years. During this time, 385 (5.7%; 164 women and 221 men) developed newly diagnosed heart failure.
In women, the hazard ratio for every 42 cm3 increase in PFV was 1.44 (95% confidence interval, 1.21-1.71; P < .001). In men, the HR was 1.13 (95% CI, 1.01-1.27; P = .03).
High PVF conferred a twofold greater risk for heart failure in women (HR, 2.06; 95% CI, 1.48-2.87; P < .001) and a 53% higher risk in men (HR, 1.53; 95% CI, 1.13-2.07; P = .006).
These associations remained significant after further adjustment for circulating markers of systemic inflammation (that is, C-reactive protein and interleukin-6), and abdominal subcutaneous or visceral fat.
They also found that the heightened risk persisted, even after adjustment for established risk factors for heart failure, such as age, cigarette smoking, alcohol consumption, sedentary lifestyle, high blood pressure, high blood sugar, high cholesterol, and myocardial infarction.
Results were similar among all of the ethnic groups studied.
A surprise finding
“The most surprising part of this study was that the risk for heart failure with increased pericardial fat does not seem to be explained by obesity and systemic inflammation alone,” Andreas P. Kalogeropoulos, MD, MPH, PhD, Stony Brook (N.Y.) University, said in an interview.
“If pericardial fat was merely a proxy for increased visceral fat, one would expect the association of pericardial fat with heart failure risk to go away after factoring in abdominal CT findings, which was not the case here. Also, accounting for inflammatory markers did not change things dramatically. However, we need to be careful here, as abdominal CT scans have not been done simultaneously with the pericardial fat scans in the study,” said Dr. Kalogeropoulos, who coauthored an accompanying editorial with Michael E. Hall, MD, University of Mississippi Medical Center, Jackson.
The other striking finding, although not entirely surprising, was the stronger association of pericardial fat with heart failure risk in women, he noted.
“Although several clues have been reported pointing to women being more sensitive to the adverse cardiac effects of pericardial fat, this is the first large prospective study to connect the dots and show much higher risk in women in a convincing way. For the record, this is the first prospective study to show the connection between pericardial fat and heart failure risk altogether,” Dr. Kalogeropoulos said.
“Obviously, we need to do more work to see how we can use the important findings of Kenchaiah and colleagues to reduce risk for heart failure among patients with increased pericardial fat, especially women. For starters, we would need a way to identify these patients,” he said. “In this aspect, it is encouraging that pericardial fat can be measured in low-radiation CT scans, similar to those used for coronary calcium, and that automation technology to speed up pericardial fat measurements is already in the pipeline.
“The next step would be to see what kind of interventions would reduce risk for heart failure in these patients,” he added. “Weight loss would be an obvious thing, but novel agents with favorable cardiometabolic effects, like newer antidiabetic medications, are intriguing options, too.”
The study was supported by the National Heart, Lung, and Blood Institute and the National Institutes of Health. Dr. Kenchaiah and Dr. Kalogeropoulos reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pericardial fat is associated with a heightened risk for heart failure, particularly in women, new research suggests.
In a prospective cohort study of nearly 7,000 individuals, excess pericardial fat was linked to a higher risk for heart failure, even after adjustment for established risk factors for heart failure.
Women with high pericardial fat volume (PFV), defined as more than 70 cm3 or 2.4 fluid ounces, had double the risk of developing heart failure. For men, high PFV, defined as more than 120 cm3 or 4.0 fluid ounces, was associated with a 50% increase in the risk for heart failure.
The findings were published in the Journal of the American College of Cardiology.
“People will ask why should they measure fat around the heart. Why can’t they just take the waist circumference or body mass index as a measure for increased risk?” lead author Satish Kenchaiah, MD, MPH, Icahn School of Medicine at Mount Sinai, New York, said in an interview.
“Yet, when we adjusted for waist circumference, hip circumference, waist to hip ratio, and other known variables, pericardial fat was still associated with an increased risk of heart failure. This tells me that it is not just overall fat in the body but something about its location around the heart that is playing a role,” Dr. Kenchaiah said.
“Now that we have found an association between any amount of fat around the pericardium and heart failure, it gives us an impetus to build future research on identifying how exactly these fat deposits influence the development of cardiomyopathy,” he said.
Dr. Kenchaiah and colleagues investigated the association of pericardial fat with incident heart failure by examining chest CT scans from 6,785 participants (3,584 women and 3,201 men aged 45-84 years) in the Multi-Ethnic Study of Atherosclerosis.
The participants were from four different ethnic groups: 38% were White; 28% were Black, 22% were Hispanic, and 12% were Chinese American. They were recruited between July 17, 2000, and Aug. 31, 2002, from six communities in the United States: Baltimore and Baltimore County; Chicago; Forsyth County, N.C.; Los Angeles County northern Manhattan and the Bronx, New York; and St. Paul, Minn.
All participants were free of cardiovascular disease at baseline.
The researchers followed participants for more than 17 years. During this time, 385 (5.7%; 164 women and 221 men) developed newly diagnosed heart failure.
In women, the hazard ratio for every 42 cm3 increase in PFV was 1.44 (95% confidence interval, 1.21-1.71; P < .001). In men, the HR was 1.13 (95% CI, 1.01-1.27; P = .03).
High PVF conferred a twofold greater risk for heart failure in women (HR, 2.06; 95% CI, 1.48-2.87; P < .001) and a 53% higher risk in men (HR, 1.53; 95% CI, 1.13-2.07; P = .006).
These associations remained significant after further adjustment for circulating markers of systemic inflammation (that is, C-reactive protein and interleukin-6), and abdominal subcutaneous or visceral fat.
They also found that the heightened risk persisted, even after adjustment for established risk factors for heart failure, such as age, cigarette smoking, alcohol consumption, sedentary lifestyle, high blood pressure, high blood sugar, high cholesterol, and myocardial infarction.
Results were similar among all of the ethnic groups studied.
A surprise finding
“The most surprising part of this study was that the risk for heart failure with increased pericardial fat does not seem to be explained by obesity and systemic inflammation alone,” Andreas P. Kalogeropoulos, MD, MPH, PhD, Stony Brook (N.Y.) University, said in an interview.
“If pericardial fat was merely a proxy for increased visceral fat, one would expect the association of pericardial fat with heart failure risk to go away after factoring in abdominal CT findings, which was not the case here. Also, accounting for inflammatory markers did not change things dramatically. However, we need to be careful here, as abdominal CT scans have not been done simultaneously with the pericardial fat scans in the study,” said Dr. Kalogeropoulos, who coauthored an accompanying editorial with Michael E. Hall, MD, University of Mississippi Medical Center, Jackson.
The other striking finding, although not entirely surprising, was the stronger association of pericardial fat with heart failure risk in women, he noted.
“Although several clues have been reported pointing to women being more sensitive to the adverse cardiac effects of pericardial fat, this is the first large prospective study to connect the dots and show much higher risk in women in a convincing way. For the record, this is the first prospective study to show the connection between pericardial fat and heart failure risk altogether,” Dr. Kalogeropoulos said.
“Obviously, we need to do more work to see how we can use the important findings of Kenchaiah and colleagues to reduce risk for heart failure among patients with increased pericardial fat, especially women. For starters, we would need a way to identify these patients,” he said. “In this aspect, it is encouraging that pericardial fat can be measured in low-radiation CT scans, similar to those used for coronary calcium, and that automation technology to speed up pericardial fat measurements is already in the pipeline.
“The next step would be to see what kind of interventions would reduce risk for heart failure in these patients,” he added. “Weight loss would be an obvious thing, but novel agents with favorable cardiometabolic effects, like newer antidiabetic medications, are intriguing options, too.”
The study was supported by the National Heart, Lung, and Blood Institute and the National Institutes of Health. Dr. Kenchaiah and Dr. Kalogeropoulos reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pericardial fat is associated with a heightened risk for heart failure, particularly in women, new research suggests.
In a prospective cohort study of nearly 7,000 individuals, excess pericardial fat was linked to a higher risk for heart failure, even after adjustment for established risk factors for heart failure.
Women with high pericardial fat volume (PFV), defined as more than 70 cm3 or 2.4 fluid ounces, had double the risk of developing heart failure. For men, high PFV, defined as more than 120 cm3 or 4.0 fluid ounces, was associated with a 50% increase in the risk for heart failure.
The findings were published in the Journal of the American College of Cardiology.
“People will ask why should they measure fat around the heart. Why can’t they just take the waist circumference or body mass index as a measure for increased risk?” lead author Satish Kenchaiah, MD, MPH, Icahn School of Medicine at Mount Sinai, New York, said in an interview.
“Yet, when we adjusted for waist circumference, hip circumference, waist to hip ratio, and other known variables, pericardial fat was still associated with an increased risk of heart failure. This tells me that it is not just overall fat in the body but something about its location around the heart that is playing a role,” Dr. Kenchaiah said.
“Now that we have found an association between any amount of fat around the pericardium and heart failure, it gives us an impetus to build future research on identifying how exactly these fat deposits influence the development of cardiomyopathy,” he said.
Dr. Kenchaiah and colleagues investigated the association of pericardial fat with incident heart failure by examining chest CT scans from 6,785 participants (3,584 women and 3,201 men aged 45-84 years) in the Multi-Ethnic Study of Atherosclerosis.
The participants were from four different ethnic groups: 38% were White; 28% were Black, 22% were Hispanic, and 12% were Chinese American. They were recruited between July 17, 2000, and Aug. 31, 2002, from six communities in the United States: Baltimore and Baltimore County; Chicago; Forsyth County, N.C.; Los Angeles County northern Manhattan and the Bronx, New York; and St. Paul, Minn.
All participants were free of cardiovascular disease at baseline.
The researchers followed participants for more than 17 years. During this time, 385 (5.7%; 164 women and 221 men) developed newly diagnosed heart failure.
In women, the hazard ratio for every 42 cm3 increase in PFV was 1.44 (95% confidence interval, 1.21-1.71; P < .001). In men, the HR was 1.13 (95% CI, 1.01-1.27; P = .03).
High PVF conferred a twofold greater risk for heart failure in women (HR, 2.06; 95% CI, 1.48-2.87; P < .001) and a 53% higher risk in men (HR, 1.53; 95% CI, 1.13-2.07; P = .006).
These associations remained significant after further adjustment for circulating markers of systemic inflammation (that is, C-reactive protein and interleukin-6), and abdominal subcutaneous or visceral fat.
They also found that the heightened risk persisted, even after adjustment for established risk factors for heart failure, such as age, cigarette smoking, alcohol consumption, sedentary lifestyle, high blood pressure, high blood sugar, high cholesterol, and myocardial infarction.
Results were similar among all of the ethnic groups studied.
A surprise finding
“The most surprising part of this study was that the risk for heart failure with increased pericardial fat does not seem to be explained by obesity and systemic inflammation alone,” Andreas P. Kalogeropoulos, MD, MPH, PhD, Stony Brook (N.Y.) University, said in an interview.
“If pericardial fat was merely a proxy for increased visceral fat, one would expect the association of pericardial fat with heart failure risk to go away after factoring in abdominal CT findings, which was not the case here. Also, accounting for inflammatory markers did not change things dramatically. However, we need to be careful here, as abdominal CT scans have not been done simultaneously with the pericardial fat scans in the study,” said Dr. Kalogeropoulos, who coauthored an accompanying editorial with Michael E. Hall, MD, University of Mississippi Medical Center, Jackson.
The other striking finding, although not entirely surprising, was the stronger association of pericardial fat with heart failure risk in women, he noted.
“Although several clues have been reported pointing to women being more sensitive to the adverse cardiac effects of pericardial fat, this is the first large prospective study to connect the dots and show much higher risk in women in a convincing way. For the record, this is the first prospective study to show the connection between pericardial fat and heart failure risk altogether,” Dr. Kalogeropoulos said.
“Obviously, we need to do more work to see how we can use the important findings of Kenchaiah and colleagues to reduce risk for heart failure among patients with increased pericardial fat, especially women. For starters, we would need a way to identify these patients,” he said. “In this aspect, it is encouraging that pericardial fat can be measured in low-radiation CT scans, similar to those used for coronary calcium, and that automation technology to speed up pericardial fat measurements is already in the pipeline.
“The next step would be to see what kind of interventions would reduce risk for heart failure in these patients,” he added. “Weight loss would be an obvious thing, but novel agents with favorable cardiometabolic effects, like newer antidiabetic medications, are intriguing options, too.”
The study was supported by the National Heart, Lung, and Blood Institute and the National Institutes of Health. Dr. Kenchaiah and Dr. Kalogeropoulos reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
DOJ charges 14 with COVID-19–related fraud nearing $150M
The U.S. Department of Justice (DOJ) on May 26 announced charges against 14 defendants across the country who allegedly engaged in health care fraud schemes that exploited the COVID-19 pandemic and resulted in over $143 million in false billings to Medicare.
Among the defendants, a DOJ news release said, were a telemedicine company executive, a physician, marketers, and medical business owners.
In addition, the Centers for Medicare and Medicaid Services separately announced that it had taken “adverse administrative actions” against more than 50 providers for their involvement in fraud schemes related to COVID-19 or the abuse of CMS programs that were designed to encourage access to medical care during the pandemic.
Several of the defendants allegedly offered COVID-19 tests to Medicare beneficiaries in senior living facilities, drive-through COVID-19 testing sites, and medical offices to induce the beneficiaries to provide their personal identifying information and a saliva or a blood sample.
The DOJ charges claim the defendants then misused the information and the samples to submit claims to Medicare for unrelated, medically unnecessary, and far more expensive lab tests, including cancer genetic testing, allergy testing, and respiratory pathogen panel tests.
In some cases, it’s alleged, the lab results were not provided to the individuals in a timely fashion or were not reliable.
Other defendants are charged with exploiting temporary changes in CMS telehealth regulations that were designed to increase access to health care during the pandemic. In these cases, which the DOJ said were the first charges related to the expansion of telehealth under the COVID-19 emergency declaration, the defendants allegedly submitted false and fraudulent claims to Medicare for sham telemedicine encounters that did not occur.
“As part of these cases, medical professionals are alleged to have [been] offered and paid bribes in exchange for the medical professionals’ referral of unnecessary testing,” the DOJ news release said. However, no physicians were identified by the department.
Commenting on this aspect of the law enforcement action, FBI Director Christopher Wray said in the release: “Medical providers have been the unsung heroes for the American public throughout the pandemic. It’s disheartening that some have abused their authorities and committed COVID-19–related fraud against trusting citizens. The FBI, along with our federal law enforcement and private sector partners, are committed to continuing to combat health care fraud and protect the American people.”
The law enforcement action includes the third set of criminal charges related to the misuse of Provider Relief Fund monies, according to the release.
More than 340 individuals were charged in September 2020 with submitting $6 billion in fraudulent claims to federal health care programs and private insurers for telehealth consultations and substance abuse treatment. About $4.5 billion of that was related to telehealth, as reported by this news organization.
The new criminal charges were brought in federal district courts in Arkansas, California, Louisiana, Florida, New Jersey, and New York.
Case summaries
The DOJ provided several case summaries. One defendant, lab owner Billy Joe Taylor of Lavaca, Ark., was charged with participating in a scheme to defraud the government of over $42 million by filing false claims that were billed in combination with COVID-19 testing claims. He also allegedly billed for tests that were not performed.
Petros Hannesyan of Burbank, Calif., the owner of a home health agency, was charged with obtaining over $229,000 from COVID-19 relief programs under false pretenses. His firm allegedly misappropriated funds from the CARES Act Provider Relief Fund and submitted false loan applications and a false loan agreement to the Economic Injury Disaster Loan Program.
Michael Stein and Leonel Palatnik of Palm Beach County, Fla., were charged in a connection with an alleged $73 million conspiracy to defraud the government and to pay and receive health care kickbacks during the pandemic.
Mr. Stein, who owned a “purported” consulting company, and Mr. Palatnik, who owned testing labs in Texas, allegedly exploited Medicare’s waiver of telehealth restrictions “by offering telehealth providers access to Medicare beneficiaries for whom they could bill consultations. In exchange, these providers agreed to refer beneficiaries to [Mr. Palatnik’s] laboratories for expensive and medically unnecessary cancer and cardiovascular genetic testing.”
A version of this article first appeared on Medscape.com.
The U.S. Department of Justice (DOJ) on May 26 announced charges against 14 defendants across the country who allegedly engaged in health care fraud schemes that exploited the COVID-19 pandemic and resulted in over $143 million in false billings to Medicare.
Among the defendants, a DOJ news release said, were a telemedicine company executive, a physician, marketers, and medical business owners.
In addition, the Centers for Medicare and Medicaid Services separately announced that it had taken “adverse administrative actions” against more than 50 providers for their involvement in fraud schemes related to COVID-19 or the abuse of CMS programs that were designed to encourage access to medical care during the pandemic.
Several of the defendants allegedly offered COVID-19 tests to Medicare beneficiaries in senior living facilities, drive-through COVID-19 testing sites, and medical offices to induce the beneficiaries to provide their personal identifying information and a saliva or a blood sample.
The DOJ charges claim the defendants then misused the information and the samples to submit claims to Medicare for unrelated, medically unnecessary, and far more expensive lab tests, including cancer genetic testing, allergy testing, and respiratory pathogen panel tests.
In some cases, it’s alleged, the lab results were not provided to the individuals in a timely fashion or were not reliable.
Other defendants are charged with exploiting temporary changes in CMS telehealth regulations that were designed to increase access to health care during the pandemic. In these cases, which the DOJ said were the first charges related to the expansion of telehealth under the COVID-19 emergency declaration, the defendants allegedly submitted false and fraudulent claims to Medicare for sham telemedicine encounters that did not occur.
“As part of these cases, medical professionals are alleged to have [been] offered and paid bribes in exchange for the medical professionals’ referral of unnecessary testing,” the DOJ news release said. However, no physicians were identified by the department.
Commenting on this aspect of the law enforcement action, FBI Director Christopher Wray said in the release: “Medical providers have been the unsung heroes for the American public throughout the pandemic. It’s disheartening that some have abused their authorities and committed COVID-19–related fraud against trusting citizens. The FBI, along with our federal law enforcement and private sector partners, are committed to continuing to combat health care fraud and protect the American people.”
The law enforcement action includes the third set of criminal charges related to the misuse of Provider Relief Fund monies, according to the release.
More than 340 individuals were charged in September 2020 with submitting $6 billion in fraudulent claims to federal health care programs and private insurers for telehealth consultations and substance abuse treatment. About $4.5 billion of that was related to telehealth, as reported by this news organization.
The new criminal charges were brought in federal district courts in Arkansas, California, Louisiana, Florida, New Jersey, and New York.
Case summaries
The DOJ provided several case summaries. One defendant, lab owner Billy Joe Taylor of Lavaca, Ark., was charged with participating in a scheme to defraud the government of over $42 million by filing false claims that were billed in combination with COVID-19 testing claims. He also allegedly billed for tests that were not performed.
Petros Hannesyan of Burbank, Calif., the owner of a home health agency, was charged with obtaining over $229,000 from COVID-19 relief programs under false pretenses. His firm allegedly misappropriated funds from the CARES Act Provider Relief Fund and submitted false loan applications and a false loan agreement to the Economic Injury Disaster Loan Program.
Michael Stein and Leonel Palatnik of Palm Beach County, Fla., were charged in a connection with an alleged $73 million conspiracy to defraud the government and to pay and receive health care kickbacks during the pandemic.
Mr. Stein, who owned a “purported” consulting company, and Mr. Palatnik, who owned testing labs in Texas, allegedly exploited Medicare’s waiver of telehealth restrictions “by offering telehealth providers access to Medicare beneficiaries for whom they could bill consultations. In exchange, these providers agreed to refer beneficiaries to [Mr. Palatnik’s] laboratories for expensive and medically unnecessary cancer and cardiovascular genetic testing.”
A version of this article first appeared on Medscape.com.
The U.S. Department of Justice (DOJ) on May 26 announced charges against 14 defendants across the country who allegedly engaged in health care fraud schemes that exploited the COVID-19 pandemic and resulted in over $143 million in false billings to Medicare.
Among the defendants, a DOJ news release said, were a telemedicine company executive, a physician, marketers, and medical business owners.
In addition, the Centers for Medicare and Medicaid Services separately announced that it had taken “adverse administrative actions” against more than 50 providers for their involvement in fraud schemes related to COVID-19 or the abuse of CMS programs that were designed to encourage access to medical care during the pandemic.
Several of the defendants allegedly offered COVID-19 tests to Medicare beneficiaries in senior living facilities, drive-through COVID-19 testing sites, and medical offices to induce the beneficiaries to provide their personal identifying information and a saliva or a blood sample.
The DOJ charges claim the defendants then misused the information and the samples to submit claims to Medicare for unrelated, medically unnecessary, and far more expensive lab tests, including cancer genetic testing, allergy testing, and respiratory pathogen panel tests.
In some cases, it’s alleged, the lab results were not provided to the individuals in a timely fashion or were not reliable.
Other defendants are charged with exploiting temporary changes in CMS telehealth regulations that were designed to increase access to health care during the pandemic. In these cases, which the DOJ said were the first charges related to the expansion of telehealth under the COVID-19 emergency declaration, the defendants allegedly submitted false and fraudulent claims to Medicare for sham telemedicine encounters that did not occur.
“As part of these cases, medical professionals are alleged to have [been] offered and paid bribes in exchange for the medical professionals’ referral of unnecessary testing,” the DOJ news release said. However, no physicians were identified by the department.
Commenting on this aspect of the law enforcement action, FBI Director Christopher Wray said in the release: “Medical providers have been the unsung heroes for the American public throughout the pandemic. It’s disheartening that some have abused their authorities and committed COVID-19–related fraud against trusting citizens. The FBI, along with our federal law enforcement and private sector partners, are committed to continuing to combat health care fraud and protect the American people.”
The law enforcement action includes the third set of criminal charges related to the misuse of Provider Relief Fund monies, according to the release.
More than 340 individuals were charged in September 2020 with submitting $6 billion in fraudulent claims to federal health care programs and private insurers for telehealth consultations and substance abuse treatment. About $4.5 billion of that was related to telehealth, as reported by this news organization.
The new criminal charges were brought in federal district courts in Arkansas, California, Louisiana, Florida, New Jersey, and New York.
Case summaries
The DOJ provided several case summaries. One defendant, lab owner Billy Joe Taylor of Lavaca, Ark., was charged with participating in a scheme to defraud the government of over $42 million by filing false claims that were billed in combination with COVID-19 testing claims. He also allegedly billed for tests that were not performed.
Petros Hannesyan of Burbank, Calif., the owner of a home health agency, was charged with obtaining over $229,000 from COVID-19 relief programs under false pretenses. His firm allegedly misappropriated funds from the CARES Act Provider Relief Fund and submitted false loan applications and a false loan agreement to the Economic Injury Disaster Loan Program.
Michael Stein and Leonel Palatnik of Palm Beach County, Fla., were charged in a connection with an alleged $73 million conspiracy to defraud the government and to pay and receive health care kickbacks during the pandemic.
Mr. Stein, who owned a “purported” consulting company, and Mr. Palatnik, who owned testing labs in Texas, allegedly exploited Medicare’s waiver of telehealth restrictions “by offering telehealth providers access to Medicare beneficiaries for whom they could bill consultations. In exchange, these providers agreed to refer beneficiaries to [Mr. Palatnik’s] laboratories for expensive and medically unnecessary cancer and cardiovascular genetic testing.”
A version of this article first appeared on Medscape.com.
Elevated factor VIII troughs can lead to a higher proportion of zero bleeds in hemophilia
Rurioctocog alfa pegol prophylaxis was linked to fewer bleeding episodes in people with hemophilia A when it targeted higher levels of factor VIII (FVIII) troughs, according to a report published in Blood (2021;137[13]:1818-27).
Earlier studies demonstrated that the treatment effectively prevented bleeds with an acceptable safety profile in people with hemophilia A. The current prospective, randomized, open label PROPEL trial compared safety and efficacy of two target FVIII troughs in this population. Targeting 1%-3% and 8%-12% FVIII troughs was efficacious, with fewer bleeds in the latter arm and acceptable safety across both, according to Robert Klamroth, MD, of Vivantes Klinikum Friedrichshain, Berlin, and colleagues.
The PROPEL trial (NCT02585960) population comprised 155 patients with hepatitis A, aged 12-65 years, with severe disease and an annualized bleeding rate of at least 2 during the 12 months before enrollment in the study. All had previous FVIII treatment. Patients were randomized to 12 months’ pharmacokinetic rurioctocog alfa pegol prophylaxis targeting FVIII troughs of 1%-3% (reference arm) or 8%-12%.
The primary endpoint was absence of bleeds during the second 6-month period. A total of 95 patients completed the protocol.
Promising results
In the 1%-3% and 8%-12% arms, the proportions of patients who completed the protocol and had no bleeds were 40% and 67% respectively (P = .015). Serious adverse events occurred in 7 of 115 (6%) patients, including one treatment-related event in the 8%-12% arm. There were no deaths, serious thrombotic events, or adverse event-related discontinuations.
“Targeting 8% to 12% FVIII troughs resulted in a higher proportion of [patients] with no bleeds than prophylaxis that targeted 1% to 3% FVIII troughs. These results support the hypothesis that an elevated FVIII trough can benefit [patients]without changing the safety profile,” the researchers reported. Personalized treatment in this patient population should be considered, they added.
Problems remain
In an invited commentary, Christine L. Kempton, MD, of Emory University, Atlanta, pointed out that the study did not answer the question of what trough level is best, and that the target trough level may be up to a patient’s individual clinician to decide. “Many participants (42%) treated with the target trough level of 1% to 3% had no bleeding events during the study period, but some (38%) continued to have bleeding events despite higher target trough levels,” Dr. Kempton wrote. She added that, beyond this concern, the presence of subclinical bleeding is difficult to study and quantify, but its presence is supported in the literature by magnetic resonance imaging that demonstrated joint damage despite a lack of clinically evident bleeding.
“Thus, targeting zero clinical bleeding events does not mean that all joint disease, dysfunction, and pain will be eliminated. This reality underscores the need for better, not just more convenient, therapies,” she concluded.
The authors reported numerous relationships with a variety of pharmaceutical companies including grants, honoraria, and participation in speakers bureaus. Dr. Kempton reported honoraria from Takeda, Spark, Octapharma, and Pfizer, and research grants from Novo Nordisk.
Rurioctocog alfa pegol prophylaxis was linked to fewer bleeding episodes in people with hemophilia A when it targeted higher levels of factor VIII (FVIII) troughs, according to a report published in Blood (2021;137[13]:1818-27).
Earlier studies demonstrated that the treatment effectively prevented bleeds with an acceptable safety profile in people with hemophilia A. The current prospective, randomized, open label PROPEL trial compared safety and efficacy of two target FVIII troughs in this population. Targeting 1%-3% and 8%-12% FVIII troughs was efficacious, with fewer bleeds in the latter arm and acceptable safety across both, according to Robert Klamroth, MD, of Vivantes Klinikum Friedrichshain, Berlin, and colleagues.
The PROPEL trial (NCT02585960) population comprised 155 patients with hepatitis A, aged 12-65 years, with severe disease and an annualized bleeding rate of at least 2 during the 12 months before enrollment in the study. All had previous FVIII treatment. Patients were randomized to 12 months’ pharmacokinetic rurioctocog alfa pegol prophylaxis targeting FVIII troughs of 1%-3% (reference arm) or 8%-12%.
The primary endpoint was absence of bleeds during the second 6-month period. A total of 95 patients completed the protocol.
Promising results
In the 1%-3% and 8%-12% arms, the proportions of patients who completed the protocol and had no bleeds were 40% and 67% respectively (P = .015). Serious adverse events occurred in 7 of 115 (6%) patients, including one treatment-related event in the 8%-12% arm. There were no deaths, serious thrombotic events, or adverse event-related discontinuations.
“Targeting 8% to 12% FVIII troughs resulted in a higher proportion of [patients] with no bleeds than prophylaxis that targeted 1% to 3% FVIII troughs. These results support the hypothesis that an elevated FVIII trough can benefit [patients]without changing the safety profile,” the researchers reported. Personalized treatment in this patient population should be considered, they added.
Problems remain
In an invited commentary, Christine L. Kempton, MD, of Emory University, Atlanta, pointed out that the study did not answer the question of what trough level is best, and that the target trough level may be up to a patient’s individual clinician to decide. “Many participants (42%) treated with the target trough level of 1% to 3% had no bleeding events during the study period, but some (38%) continued to have bleeding events despite higher target trough levels,” Dr. Kempton wrote. She added that, beyond this concern, the presence of subclinical bleeding is difficult to study and quantify, but its presence is supported in the literature by magnetic resonance imaging that demonstrated joint damage despite a lack of clinically evident bleeding.
“Thus, targeting zero clinical bleeding events does not mean that all joint disease, dysfunction, and pain will be eliminated. This reality underscores the need for better, not just more convenient, therapies,” she concluded.
The authors reported numerous relationships with a variety of pharmaceutical companies including grants, honoraria, and participation in speakers bureaus. Dr. Kempton reported honoraria from Takeda, Spark, Octapharma, and Pfizer, and research grants from Novo Nordisk.
Rurioctocog alfa pegol prophylaxis was linked to fewer bleeding episodes in people with hemophilia A when it targeted higher levels of factor VIII (FVIII) troughs, according to a report published in Blood (2021;137[13]:1818-27).
Earlier studies demonstrated that the treatment effectively prevented bleeds with an acceptable safety profile in people with hemophilia A. The current prospective, randomized, open label PROPEL trial compared safety and efficacy of two target FVIII troughs in this population. Targeting 1%-3% and 8%-12% FVIII troughs was efficacious, with fewer bleeds in the latter arm and acceptable safety across both, according to Robert Klamroth, MD, of Vivantes Klinikum Friedrichshain, Berlin, and colleagues.
The PROPEL trial (NCT02585960) population comprised 155 patients with hepatitis A, aged 12-65 years, with severe disease and an annualized bleeding rate of at least 2 during the 12 months before enrollment in the study. All had previous FVIII treatment. Patients were randomized to 12 months’ pharmacokinetic rurioctocog alfa pegol prophylaxis targeting FVIII troughs of 1%-3% (reference arm) or 8%-12%.
The primary endpoint was absence of bleeds during the second 6-month period. A total of 95 patients completed the protocol.
Promising results
In the 1%-3% and 8%-12% arms, the proportions of patients who completed the protocol and had no bleeds were 40% and 67% respectively (P = .015). Serious adverse events occurred in 7 of 115 (6%) patients, including one treatment-related event in the 8%-12% arm. There were no deaths, serious thrombotic events, or adverse event-related discontinuations.
“Targeting 8% to 12% FVIII troughs resulted in a higher proportion of [patients] with no bleeds than prophylaxis that targeted 1% to 3% FVIII troughs. These results support the hypothesis that an elevated FVIII trough can benefit [patients]without changing the safety profile,” the researchers reported. Personalized treatment in this patient population should be considered, they added.
Problems remain
In an invited commentary, Christine L. Kempton, MD, of Emory University, Atlanta, pointed out that the study did not answer the question of what trough level is best, and that the target trough level may be up to a patient’s individual clinician to decide. “Many participants (42%) treated with the target trough level of 1% to 3% had no bleeding events during the study period, but some (38%) continued to have bleeding events despite higher target trough levels,” Dr. Kempton wrote. She added that, beyond this concern, the presence of subclinical bleeding is difficult to study and quantify, but its presence is supported in the literature by magnetic resonance imaging that demonstrated joint damage despite a lack of clinically evident bleeding.
“Thus, targeting zero clinical bleeding events does not mean that all joint disease, dysfunction, and pain will be eliminated. This reality underscores the need for better, not just more convenient, therapies,” she concluded.
The authors reported numerous relationships with a variety of pharmaceutical companies including grants, honoraria, and participation in speakers bureaus. Dr. Kempton reported honoraria from Takeda, Spark, Octapharma, and Pfizer, and research grants from Novo Nordisk.
FROM BLOOD
‘A better picture’: First AACE guidelines on diabetes technology
The American Association of Clinical Endocrinology (AACE) has issued its first-ever official guidelines addressing the use of advanced technologies in the management of people with diabetes.
The guidelines cover use of continuous glucose monitoring (CGM), insulin pumps, connected pens, automated insulin delivery systems, telemedicine technologies, and smartphone apps. They also address safety considerations, special situations such as hospitalization, and implementation in clinical practice.
They were presented on May 28 at the annual scientific & clinical congress of the American Association of Clinical Endocrinologists and simultaneously published in Endocrine Practice.
Previous AACE guidance on the clinical use of insulin pumps and CGM over the past decade has been published in the form of consensus or position statements rather than official evidence-based guidelines, task force cochair George Grunberger, MD, of the Grunberger Diabetes Institute, Bloomfield Hills, Mich., explained.
“There’s never really been, until now, hardcore evidence, [with] peer-reviewed, quality trials published in the literature to go after the evidence that is required for guidelines. ... This is not an opinion piece or position statement.”
The problem with that strict approach to “guidelines” is how quickly the diabetes technology field is evolving, he acknowledged. “It’s frustrating because we know what’s [coming up], but we can’t put it in a guideline because it hasn’t been published yet.”
In an AACE podcast, Dr. Grunberger said the guidelines will likely become a “living” document, along the lines of the American Diabetes Association’s annual Standards of Care, as “any cutoff date is arbitrary. More and more papers will be published on these technologies. ... This is certainly not a static field.”
In the meantime, task force cochair and author Jennifer Sherr, MD, PhD, a pediatric endocrinologist, said she hopes the guidelines will help to reduce insurance company barriers to use of the currently available technologies.
“I am very hopeful that these guidelines will also encourage payers to change their stance. And I think that we as a community can continue to advocate and inform them of these guidelines so they can appropriately change their coverage practices,” added Dr. Sherr, of Yale University, New Haven, Conn.
Recommendations address CGM, pumps, and connected systems
In the guidelines, CGM is “strongly recommended for all persons with diabetes treated with intensive insulin therapy, defined as three or more injections of insulin per day or the use of an insulin pump.” For those with diabetes who use CGM, “priority metrics” include a “time in range” of greater than 70% from 14 days of active use. Targets for mean glucose should be individualized, with glycemic variability 36% or lower.
Further specific CGM target metrics are given for people with type 1 diabetes, older/high risk individuals, and for pregnant women. The recommendations align with those issued in a 2019 joint consensus statement on CGM time-in-range endorsed by several organizations, including AACE.
In response to an audience question about whether AACE is advising that time-in-range replace A1c for glycemia assessment, Dr. Sherr responded: “I think currently we’re not in a position where we can completely replace A1c with time in range. However, I’m hopeful that in future years we’ll see further data gathered ... to allow for that recommendation to occur.”
For now, she said, “What we really want to hone in on in the guidelines is that time-in-range and use of CGM truly allow clinicians to better understand how to optimize care for their persons with diabetes. It gives us a better picture. It’s not just a number of whether we’re hitting target. It tells us whether we need to attack time above range or time below range. So we really think it’s critical for clinical care.”
The document also provides specifics about real-time versus intermittently scanned CGM and use of diagnostic/professional CGM.
The “insulin delivery technologies” section covers use of connected pens, insulin pumps without CGM, insulin pumps with separate CGM, and the more advanced combined insulin pump-CGM systems including those with low-glucose suspend, predictive low-glucose suspend, and hybrid closed-loops (sometimes called the artificial pancreas).
In general, these automated insulin delivery systems (artificial pancreas), “are strongly recommended for all persons with [type 1 diabetes], since their use has been shown to increase time in range, especially in the overnight period, without causing an increased risk of hypoglycemia,” Dr. Sherr observed.
Other tech topics: Apps, telemedicine, and safety
The new guidelines say that “clinically validated” smartphone apps should be recommended to help teach or reinforce diabetes self-management skills and provide support and encouragement for healthy behaviors around food and exercise.
Dr. Grunberger pointed out: “As we know, there are tons of apps out there, and patients are using them. The problem is that very few of them have actually been validated in clinical trials in published peer-reviewed [journals].”
He recommended a joint statement on diabetes apps from the American Diabetes Association and the European Association for the Study of Diabetes that was initially discussed at the 2019 EASD meeting, as reported by this news organization, and subsequently published in January 2020 in Diabetes Care and Diabetologia.
“Telemedicine, including periodic phone calls, smartphone-web interactions ... by health care professionals ... is strongly recommended to treat persons with diabetes, provide diabetes education, remotely monitor glucose and/or insulin data to indicate the need for therapy adjustments, and improve diabetes-related outcomes/control with better engagement,” the document says.
Safety concerns addressed include the issue of certain medications interfering with CGM [readings] ... including acetaminophen, high-dose vitamin C, and hydroxyurea, as well as cautions about what to do in the event of device malfunction and assessing that the patient is sufficiently trained in proper device use. Criteria for insulin pump discontinuation are also given.
Implementation: Who will be prescribing? ‘This is not for amateurs’
A final section on implementation recommends that “initiation and use of diabetes technology should be implemented by health care professionals who are trained, committed, and experienced to prescribe and direct the use of these tools. Clinicians should have the infrastructure to support the needs of persons with diabetes using the technology.”
Dr. Grunberger commented: “I think the key is going to be who should be doing this? What is the role of a clinical endocrinologist in the future? What is our responsibility, [since] we don’t have the manpower and womanpower to take care of all these people as these technologies advance? It’s our responsibility to provide these hopefully valued recommendations as a resource for those who want to know more about it.”
However, he noted, “This is not for amateurs. If you want to actually use this in your practice, you need the infrastructure, the expertise, the training, the dedication, and the energy to be there for the patients all the time ... This clinical practice guideline is a foundation.”
Dr. Sherr added: “To me, it’s really thinking about ... changing our mindset from who is an appropriate candidate to who can benefit and how vast a group that entails ... I’m hopeful that we will see more technology use through continued conversations with our patients with diabetes, and hopefully through more clinicians being excited to be part of this revolution.”
Dr. Grunberger has reported being on speakers bureaus for Eli Lilly, Novo Nordisk, and Abbott. Dr. Sherr has reported being a consultant and speaker for Lilly and Medtronic Diabetes, a consultant for Insulet and Sanofi, and on advisory boards for Bigfoot Biomedical, Cecelia Health, Insulet, JDRF T1D fund, and Medtronic.
A version of this article first appeared on Medscape.com.
The American Association of Clinical Endocrinology (AACE) has issued its first-ever official guidelines addressing the use of advanced technologies in the management of people with diabetes.
The guidelines cover use of continuous glucose monitoring (CGM), insulin pumps, connected pens, automated insulin delivery systems, telemedicine technologies, and smartphone apps. They also address safety considerations, special situations such as hospitalization, and implementation in clinical practice.
They were presented on May 28 at the annual scientific & clinical congress of the American Association of Clinical Endocrinologists and simultaneously published in Endocrine Practice.
Previous AACE guidance on the clinical use of insulin pumps and CGM over the past decade has been published in the form of consensus or position statements rather than official evidence-based guidelines, task force cochair George Grunberger, MD, of the Grunberger Diabetes Institute, Bloomfield Hills, Mich., explained.
“There’s never really been, until now, hardcore evidence, [with] peer-reviewed, quality trials published in the literature to go after the evidence that is required for guidelines. ... This is not an opinion piece or position statement.”
The problem with that strict approach to “guidelines” is how quickly the diabetes technology field is evolving, he acknowledged. “It’s frustrating because we know what’s [coming up], but we can’t put it in a guideline because it hasn’t been published yet.”
In an AACE podcast, Dr. Grunberger said the guidelines will likely become a “living” document, along the lines of the American Diabetes Association’s annual Standards of Care, as “any cutoff date is arbitrary. More and more papers will be published on these technologies. ... This is certainly not a static field.”
In the meantime, task force cochair and author Jennifer Sherr, MD, PhD, a pediatric endocrinologist, said she hopes the guidelines will help to reduce insurance company barriers to use of the currently available technologies.
“I am very hopeful that these guidelines will also encourage payers to change their stance. And I think that we as a community can continue to advocate and inform them of these guidelines so they can appropriately change their coverage practices,” added Dr. Sherr, of Yale University, New Haven, Conn.
Recommendations address CGM, pumps, and connected systems
In the guidelines, CGM is “strongly recommended for all persons with diabetes treated with intensive insulin therapy, defined as three or more injections of insulin per day or the use of an insulin pump.” For those with diabetes who use CGM, “priority metrics” include a “time in range” of greater than 70% from 14 days of active use. Targets for mean glucose should be individualized, with glycemic variability 36% or lower.
Further specific CGM target metrics are given for people with type 1 diabetes, older/high risk individuals, and for pregnant women. The recommendations align with those issued in a 2019 joint consensus statement on CGM time-in-range endorsed by several organizations, including AACE.
In response to an audience question about whether AACE is advising that time-in-range replace A1c for glycemia assessment, Dr. Sherr responded: “I think currently we’re not in a position where we can completely replace A1c with time in range. However, I’m hopeful that in future years we’ll see further data gathered ... to allow for that recommendation to occur.”
For now, she said, “What we really want to hone in on in the guidelines is that time-in-range and use of CGM truly allow clinicians to better understand how to optimize care for their persons with diabetes. It gives us a better picture. It’s not just a number of whether we’re hitting target. It tells us whether we need to attack time above range or time below range. So we really think it’s critical for clinical care.”
The document also provides specifics about real-time versus intermittently scanned CGM and use of diagnostic/professional CGM.
The “insulin delivery technologies” section covers use of connected pens, insulin pumps without CGM, insulin pumps with separate CGM, and the more advanced combined insulin pump-CGM systems including those with low-glucose suspend, predictive low-glucose suspend, and hybrid closed-loops (sometimes called the artificial pancreas).
In general, these automated insulin delivery systems (artificial pancreas), “are strongly recommended for all persons with [type 1 diabetes], since their use has been shown to increase time in range, especially in the overnight period, without causing an increased risk of hypoglycemia,” Dr. Sherr observed.
Other tech topics: Apps, telemedicine, and safety
The new guidelines say that “clinically validated” smartphone apps should be recommended to help teach or reinforce diabetes self-management skills and provide support and encouragement for healthy behaviors around food and exercise.
Dr. Grunberger pointed out: “As we know, there are tons of apps out there, and patients are using them. The problem is that very few of them have actually been validated in clinical trials in published peer-reviewed [journals].”
He recommended a joint statement on diabetes apps from the American Diabetes Association and the European Association for the Study of Diabetes that was initially discussed at the 2019 EASD meeting, as reported by this news organization, and subsequently published in January 2020 in Diabetes Care and Diabetologia.
“Telemedicine, including periodic phone calls, smartphone-web interactions ... by health care professionals ... is strongly recommended to treat persons with diabetes, provide diabetes education, remotely monitor glucose and/or insulin data to indicate the need for therapy adjustments, and improve diabetes-related outcomes/control with better engagement,” the document says.
Safety concerns addressed include the issue of certain medications interfering with CGM [readings] ... including acetaminophen, high-dose vitamin C, and hydroxyurea, as well as cautions about what to do in the event of device malfunction and assessing that the patient is sufficiently trained in proper device use. Criteria for insulin pump discontinuation are also given.
Implementation: Who will be prescribing? ‘This is not for amateurs’
A final section on implementation recommends that “initiation and use of diabetes technology should be implemented by health care professionals who are trained, committed, and experienced to prescribe and direct the use of these tools. Clinicians should have the infrastructure to support the needs of persons with diabetes using the technology.”
Dr. Grunberger commented: “I think the key is going to be who should be doing this? What is the role of a clinical endocrinologist in the future? What is our responsibility, [since] we don’t have the manpower and womanpower to take care of all these people as these technologies advance? It’s our responsibility to provide these hopefully valued recommendations as a resource for those who want to know more about it.”
However, he noted, “This is not for amateurs. If you want to actually use this in your practice, you need the infrastructure, the expertise, the training, the dedication, and the energy to be there for the patients all the time ... This clinical practice guideline is a foundation.”
Dr. Sherr added: “To me, it’s really thinking about ... changing our mindset from who is an appropriate candidate to who can benefit and how vast a group that entails ... I’m hopeful that we will see more technology use through continued conversations with our patients with diabetes, and hopefully through more clinicians being excited to be part of this revolution.”
Dr. Grunberger has reported being on speakers bureaus for Eli Lilly, Novo Nordisk, and Abbott. Dr. Sherr has reported being a consultant and speaker for Lilly and Medtronic Diabetes, a consultant for Insulet and Sanofi, and on advisory boards for Bigfoot Biomedical, Cecelia Health, Insulet, JDRF T1D fund, and Medtronic.
A version of this article first appeared on Medscape.com.
The American Association of Clinical Endocrinology (AACE) has issued its first-ever official guidelines addressing the use of advanced technologies in the management of people with diabetes.
The guidelines cover use of continuous glucose monitoring (CGM), insulin pumps, connected pens, automated insulin delivery systems, telemedicine technologies, and smartphone apps. They also address safety considerations, special situations such as hospitalization, and implementation in clinical practice.
They were presented on May 28 at the annual scientific & clinical congress of the American Association of Clinical Endocrinologists and simultaneously published in Endocrine Practice.
Previous AACE guidance on the clinical use of insulin pumps and CGM over the past decade has been published in the form of consensus or position statements rather than official evidence-based guidelines, task force cochair George Grunberger, MD, of the Grunberger Diabetes Institute, Bloomfield Hills, Mich., explained.
“There’s never really been, until now, hardcore evidence, [with] peer-reviewed, quality trials published in the literature to go after the evidence that is required for guidelines. ... This is not an opinion piece or position statement.”
The problem with that strict approach to “guidelines” is how quickly the diabetes technology field is evolving, he acknowledged. “It’s frustrating because we know what’s [coming up], but we can’t put it in a guideline because it hasn’t been published yet.”
In an AACE podcast, Dr. Grunberger said the guidelines will likely become a “living” document, along the lines of the American Diabetes Association’s annual Standards of Care, as “any cutoff date is arbitrary. More and more papers will be published on these technologies. ... This is certainly not a static field.”
In the meantime, task force cochair and author Jennifer Sherr, MD, PhD, a pediatric endocrinologist, said she hopes the guidelines will help to reduce insurance company barriers to use of the currently available technologies.
“I am very hopeful that these guidelines will also encourage payers to change their stance. And I think that we as a community can continue to advocate and inform them of these guidelines so they can appropriately change their coverage practices,” added Dr. Sherr, of Yale University, New Haven, Conn.
Recommendations address CGM, pumps, and connected systems
In the guidelines, CGM is “strongly recommended for all persons with diabetes treated with intensive insulin therapy, defined as three or more injections of insulin per day or the use of an insulin pump.” For those with diabetes who use CGM, “priority metrics” include a “time in range” of greater than 70% from 14 days of active use. Targets for mean glucose should be individualized, with glycemic variability 36% or lower.
Further specific CGM target metrics are given for people with type 1 diabetes, older/high risk individuals, and for pregnant women. The recommendations align with those issued in a 2019 joint consensus statement on CGM time-in-range endorsed by several organizations, including AACE.
In response to an audience question about whether AACE is advising that time-in-range replace A1c for glycemia assessment, Dr. Sherr responded: “I think currently we’re not in a position where we can completely replace A1c with time in range. However, I’m hopeful that in future years we’ll see further data gathered ... to allow for that recommendation to occur.”
For now, she said, “What we really want to hone in on in the guidelines is that time-in-range and use of CGM truly allow clinicians to better understand how to optimize care for their persons with diabetes. It gives us a better picture. It’s not just a number of whether we’re hitting target. It tells us whether we need to attack time above range or time below range. So we really think it’s critical for clinical care.”
The document also provides specifics about real-time versus intermittently scanned CGM and use of diagnostic/professional CGM.
The “insulin delivery technologies” section covers use of connected pens, insulin pumps without CGM, insulin pumps with separate CGM, and the more advanced combined insulin pump-CGM systems including those with low-glucose suspend, predictive low-glucose suspend, and hybrid closed-loops (sometimes called the artificial pancreas).
In general, these automated insulin delivery systems (artificial pancreas), “are strongly recommended for all persons with [type 1 diabetes], since their use has been shown to increase time in range, especially in the overnight period, without causing an increased risk of hypoglycemia,” Dr. Sherr observed.
Other tech topics: Apps, telemedicine, and safety
The new guidelines say that “clinically validated” smartphone apps should be recommended to help teach or reinforce diabetes self-management skills and provide support and encouragement for healthy behaviors around food and exercise.
Dr. Grunberger pointed out: “As we know, there are tons of apps out there, and patients are using them. The problem is that very few of them have actually been validated in clinical trials in published peer-reviewed [journals].”
He recommended a joint statement on diabetes apps from the American Diabetes Association and the European Association for the Study of Diabetes that was initially discussed at the 2019 EASD meeting, as reported by this news organization, and subsequently published in January 2020 in Diabetes Care and Diabetologia.
“Telemedicine, including periodic phone calls, smartphone-web interactions ... by health care professionals ... is strongly recommended to treat persons with diabetes, provide diabetes education, remotely monitor glucose and/or insulin data to indicate the need for therapy adjustments, and improve diabetes-related outcomes/control with better engagement,” the document says.
Safety concerns addressed include the issue of certain medications interfering with CGM [readings] ... including acetaminophen, high-dose vitamin C, and hydroxyurea, as well as cautions about what to do in the event of device malfunction and assessing that the patient is sufficiently trained in proper device use. Criteria for insulin pump discontinuation are also given.
Implementation: Who will be prescribing? ‘This is not for amateurs’
A final section on implementation recommends that “initiation and use of diabetes technology should be implemented by health care professionals who are trained, committed, and experienced to prescribe and direct the use of these tools. Clinicians should have the infrastructure to support the needs of persons with diabetes using the technology.”
Dr. Grunberger commented: “I think the key is going to be who should be doing this? What is the role of a clinical endocrinologist in the future? What is our responsibility, [since] we don’t have the manpower and womanpower to take care of all these people as these technologies advance? It’s our responsibility to provide these hopefully valued recommendations as a resource for those who want to know more about it.”
However, he noted, “This is not for amateurs. If you want to actually use this in your practice, you need the infrastructure, the expertise, the training, the dedication, and the energy to be there for the patients all the time ... This clinical practice guideline is a foundation.”
Dr. Sherr added: “To me, it’s really thinking about ... changing our mindset from who is an appropriate candidate to who can benefit and how vast a group that entails ... I’m hopeful that we will see more technology use through continued conversations with our patients with diabetes, and hopefully through more clinicians being excited to be part of this revolution.”
Dr. Grunberger has reported being on speakers bureaus for Eli Lilly, Novo Nordisk, and Abbott. Dr. Sherr has reported being a consultant and speaker for Lilly and Medtronic Diabetes, a consultant for Insulet and Sanofi, and on advisory boards for Bigfoot Biomedical, Cecelia Health, Insulet, JDRF T1D fund, and Medtronic.
A version of this article first appeared on Medscape.com.
Single subcutaneous shot offers fast, potent platelet inhibition in STEMI
A subcutaneous dose of the second-generation glycoprotein IIb/IIIa inhibitor RUC-4 achieved rapid dose-dependent platelet inhibition in patients with ST-segment elevation MI (STEMI) undergoing stenting in the CEL-02 study.
Platelet inhibition occurred within 15 minutes among the 27 patients, and wore off rapidly, with almost 50% of platelet function recovered within 122 minutes.
The drug was well tolerated, with no thrombocytopenia in the first 72 hours after administration, one injection-site reaction, and two major bleeds likely caused by catheter-based trauma to the proximal radial artery, reported Jurrien ten Berg, MD, PhD, St. Antonius Hospital, Nieuwegein, the Netherlands.
The results were reported during the annual meeting of the European Association of Percutaneous Cardiovascular Interventions (EuroPCR 2021) and published simultaneously in EuroIntervention.
Dr. ten Berg noted that there is a need for drugs like RUC-4 in the early treatment of STEMI because oral P2Y12 inhibitors have a “seriously delayed” onset by about 2-4 hours. Prehospital use of the glycoprotein inhibitor (GPI) tirofiban was shown to improve reperfusion and late outcomes in the ON-TIME 2 trial, but GPIs require continuous intravenous administration and are associated with thrombocytopenia.
“Since RUC-4 is unique among small-molecule GPI in not inducing the receptor to undergo a major conformational change that has been implicated in the development of thrombocytopenia, it is possible that RUC-4 may be associated with fewer episodes of thrombocytopenia than current GPI,” the authors wrote.
RUC-4, also called zalunfiban, can be delivered with a single subcutaneous dose and, in a phase 1 study, demonstrated platelet inhibition within 15 minutes and was well tolerated up to a dose of 0.075 mg/kg among healthy volunteers and patients with stable coronary artery disease on aspirin.
In the CEL-02 study, 27 STEMI patients received a weight-adjusted subcutaneous injection of RUC-4 before primary percutaneous coronary intervention (PCI) in escalating doses of 0.075 mg/kg, 0.090 mg/kg, and 0.110 mg/kg. Patients were given standard treatment in the ambulance, which included aspirin (93%), ticagrelor (93%), and unfractionated heparin (96%). The activated clotting time was less than 200 seconds in 92% of patients who received additional heparin during cardiac catheterization.
The patients’ mean age was 62 years, 26% were women, and 96% were White. Pharmacodynamic data were available for 24 patients.
The average platelet inhibition 15 minutes after the injection was 77.5%, 87.5%, and 91.7%, respectively, for the three escalating doses (P = .002 for trend).
The primary endpoint of at least 77% inhibition of the iso-TRAP channel – which corresponds to 80% inhibition of light transmission aggregometry stimulated by 20 mcM adenosine diphosphate within 15 minutes – was achieved in three of eight patients at the lowest dose and in seven of eight patients at the middle and highest doses.
“Single-dose subcutaneous RUC-4 induces a fast, potent dose-dependent response of platelet inhibition in patients with STEMI presenting for primary PCI,” Dr. ten Berg concluded. “It is therefore promising for prehospital platelet inhibition in STEMI patients, and the results support further research on clinical benefit.”
The double-blind, randomized phase 2b CELEBRATE trial is underway, evaluating 1,668 STEMI patients treated with a 0.110 mg/kg or 0.130 mg/kg dose of RUC-4 or placebo in the ambulance. The coprimary outcomes are restoration of coronary artery blood flow and resolution of ST-segment deviation post-PCI/angiography. Primary completion is set for March 2023.
Marco Valgimigli, MD, who was not involved in the study, said in an interview that RUC-4 has “some theoretical advantages, compared with conventional IIb/IIIa inhibitors, namely the absence of thrombocytopenia which is, however, relatively rare, especially with tirofiban or eptifibatide.”
The subcutaneous approach may also offer an advantage. Yet, if the administration of RUC-4 is “to happen in the ambulance – a setting where an IV line is usually established – whether the subcutaneous versus IV administration of the treatment proves to be advantageous remains to be seen,” said Dr. Valgimigli, from Cardiocentro Ticino Institute, Ente Ospedaliero Cantonale, Lugano, Switzerland.
“We would need to see the results of large randomized trials embracing this treatment option before a clinical decision can be made, especially considering that IIb/IIa inhibitors in the ambulance have been tested in the past but ultimately abandoned,” he said.
Limitations of the study are its open-label design, the fact that iso-TRAP channel assay data were not reported by the VeryifyNow instrument and had to be calculated from the raw data, and the fact that the timing of the RUC-4 injection immediately before PCI does not fully resemble the expected use of RUC-4 in clinical practice, where RUC-4 would be administered at the same time as the aspirin, ticagrelor, and heparin, and about an hour before PCI, ten Berg and colleagues wrote.
CeleCor Therapeutics sponsored the study and provided study materials. Dr. ten Berg reported receiving lecture or consultancy fees from AstraZeneca, Eli Lilly, Daiichi Sankyo, The Medicines Company, AccuMetrics, Boehringer Ingelheim, Bristol-Myers Squibb, Pfizer, Bayer, Ferrer, and Idorsia, and institutional research grants from ZonMw and AstraZeneca. Coauthor Barry S. Coller is an inventor of RUC-4 and a founder, equity holder, and consultant to CeleCor. He also receives royalties from Centocor/Janssen and the VerifyNow assays. Dr. Valgimigli has received grants from Abbott, Terumo, Medicure, and AstraZeneca, and personal fees from Abbott, Chiesi, Bayer, Daiichi Sankyo, Amgen, Terumo, Alvimedica, AstraZeneca, Biosensors, and Idorsia.
A version of this article first appeared on Medscape.com.
A subcutaneous dose of the second-generation glycoprotein IIb/IIIa inhibitor RUC-4 achieved rapid dose-dependent platelet inhibition in patients with ST-segment elevation MI (STEMI) undergoing stenting in the CEL-02 study.
Platelet inhibition occurred within 15 minutes among the 27 patients, and wore off rapidly, with almost 50% of platelet function recovered within 122 minutes.
The drug was well tolerated, with no thrombocytopenia in the first 72 hours after administration, one injection-site reaction, and two major bleeds likely caused by catheter-based trauma to the proximal radial artery, reported Jurrien ten Berg, MD, PhD, St. Antonius Hospital, Nieuwegein, the Netherlands.
The results were reported during the annual meeting of the European Association of Percutaneous Cardiovascular Interventions (EuroPCR 2021) and published simultaneously in EuroIntervention.
Dr. ten Berg noted that there is a need for drugs like RUC-4 in the early treatment of STEMI because oral P2Y12 inhibitors have a “seriously delayed” onset by about 2-4 hours. Prehospital use of the glycoprotein inhibitor (GPI) tirofiban was shown to improve reperfusion and late outcomes in the ON-TIME 2 trial, but GPIs require continuous intravenous administration and are associated with thrombocytopenia.
“Since RUC-4 is unique among small-molecule GPI in not inducing the receptor to undergo a major conformational change that has been implicated in the development of thrombocytopenia, it is possible that RUC-4 may be associated with fewer episodes of thrombocytopenia than current GPI,” the authors wrote.
RUC-4, also called zalunfiban, can be delivered with a single subcutaneous dose and, in a phase 1 study, demonstrated platelet inhibition within 15 minutes and was well tolerated up to a dose of 0.075 mg/kg among healthy volunteers and patients with stable coronary artery disease on aspirin.
In the CEL-02 study, 27 STEMI patients received a weight-adjusted subcutaneous injection of RUC-4 before primary percutaneous coronary intervention (PCI) in escalating doses of 0.075 mg/kg, 0.090 mg/kg, and 0.110 mg/kg. Patients were given standard treatment in the ambulance, which included aspirin (93%), ticagrelor (93%), and unfractionated heparin (96%). The activated clotting time was less than 200 seconds in 92% of patients who received additional heparin during cardiac catheterization.
The patients’ mean age was 62 years, 26% were women, and 96% were White. Pharmacodynamic data were available for 24 patients.
The average platelet inhibition 15 minutes after the injection was 77.5%, 87.5%, and 91.7%, respectively, for the three escalating doses (P = .002 for trend).
The primary endpoint of at least 77% inhibition of the iso-TRAP channel – which corresponds to 80% inhibition of light transmission aggregometry stimulated by 20 mcM adenosine diphosphate within 15 minutes – was achieved in three of eight patients at the lowest dose and in seven of eight patients at the middle and highest doses.
“Single-dose subcutaneous RUC-4 induces a fast, potent dose-dependent response of platelet inhibition in patients with STEMI presenting for primary PCI,” Dr. ten Berg concluded. “It is therefore promising for prehospital platelet inhibition in STEMI patients, and the results support further research on clinical benefit.”
The double-blind, randomized phase 2b CELEBRATE trial is underway, evaluating 1,668 STEMI patients treated with a 0.110 mg/kg or 0.130 mg/kg dose of RUC-4 or placebo in the ambulance. The coprimary outcomes are restoration of coronary artery blood flow and resolution of ST-segment deviation post-PCI/angiography. Primary completion is set for March 2023.
Marco Valgimigli, MD, who was not involved in the study, said in an interview that RUC-4 has “some theoretical advantages, compared with conventional IIb/IIIa inhibitors, namely the absence of thrombocytopenia which is, however, relatively rare, especially with tirofiban or eptifibatide.”
The subcutaneous approach may also offer an advantage. Yet, if the administration of RUC-4 is “to happen in the ambulance – a setting where an IV line is usually established – whether the subcutaneous versus IV administration of the treatment proves to be advantageous remains to be seen,” said Dr. Valgimigli, from Cardiocentro Ticino Institute, Ente Ospedaliero Cantonale, Lugano, Switzerland.
“We would need to see the results of large randomized trials embracing this treatment option before a clinical decision can be made, especially considering that IIb/IIa inhibitors in the ambulance have been tested in the past but ultimately abandoned,” he said.
Limitations of the study are its open-label design, the fact that iso-TRAP channel assay data were not reported by the VeryifyNow instrument and had to be calculated from the raw data, and the fact that the timing of the RUC-4 injection immediately before PCI does not fully resemble the expected use of RUC-4 in clinical practice, where RUC-4 would be administered at the same time as the aspirin, ticagrelor, and heparin, and about an hour before PCI, ten Berg and colleagues wrote.
CeleCor Therapeutics sponsored the study and provided study materials. Dr. ten Berg reported receiving lecture or consultancy fees from AstraZeneca, Eli Lilly, Daiichi Sankyo, The Medicines Company, AccuMetrics, Boehringer Ingelheim, Bristol-Myers Squibb, Pfizer, Bayer, Ferrer, and Idorsia, and institutional research grants from ZonMw and AstraZeneca. Coauthor Barry S. Coller is an inventor of RUC-4 and a founder, equity holder, and consultant to CeleCor. He also receives royalties from Centocor/Janssen and the VerifyNow assays. Dr. Valgimigli has received grants from Abbott, Terumo, Medicure, and AstraZeneca, and personal fees from Abbott, Chiesi, Bayer, Daiichi Sankyo, Amgen, Terumo, Alvimedica, AstraZeneca, Biosensors, and Idorsia.
A version of this article first appeared on Medscape.com.
A subcutaneous dose of the second-generation glycoprotein IIb/IIIa inhibitor RUC-4 achieved rapid dose-dependent platelet inhibition in patients with ST-segment elevation MI (STEMI) undergoing stenting in the CEL-02 study.
Platelet inhibition occurred within 15 minutes among the 27 patients, and wore off rapidly, with almost 50% of platelet function recovered within 122 minutes.
The drug was well tolerated, with no thrombocytopenia in the first 72 hours after administration, one injection-site reaction, and two major bleeds likely caused by catheter-based trauma to the proximal radial artery, reported Jurrien ten Berg, MD, PhD, St. Antonius Hospital, Nieuwegein, the Netherlands.
The results were reported during the annual meeting of the European Association of Percutaneous Cardiovascular Interventions (EuroPCR 2021) and published simultaneously in EuroIntervention.
Dr. ten Berg noted that there is a need for drugs like RUC-4 in the early treatment of STEMI because oral P2Y12 inhibitors have a “seriously delayed” onset by about 2-4 hours. Prehospital use of the glycoprotein inhibitor (GPI) tirofiban was shown to improve reperfusion and late outcomes in the ON-TIME 2 trial, but GPIs require continuous intravenous administration and are associated with thrombocytopenia.
“Since RUC-4 is unique among small-molecule GPI in not inducing the receptor to undergo a major conformational change that has been implicated in the development of thrombocytopenia, it is possible that RUC-4 may be associated with fewer episodes of thrombocytopenia than current GPI,” the authors wrote.
RUC-4, also called zalunfiban, can be delivered with a single subcutaneous dose and, in a phase 1 study, demonstrated platelet inhibition within 15 minutes and was well tolerated up to a dose of 0.075 mg/kg among healthy volunteers and patients with stable coronary artery disease on aspirin.
In the CEL-02 study, 27 STEMI patients received a weight-adjusted subcutaneous injection of RUC-4 before primary percutaneous coronary intervention (PCI) in escalating doses of 0.075 mg/kg, 0.090 mg/kg, and 0.110 mg/kg. Patients were given standard treatment in the ambulance, which included aspirin (93%), ticagrelor (93%), and unfractionated heparin (96%). The activated clotting time was less than 200 seconds in 92% of patients who received additional heparin during cardiac catheterization.
The patients’ mean age was 62 years, 26% were women, and 96% were White. Pharmacodynamic data were available for 24 patients.
The average platelet inhibition 15 minutes after the injection was 77.5%, 87.5%, and 91.7%, respectively, for the three escalating doses (P = .002 for trend).
The primary endpoint of at least 77% inhibition of the iso-TRAP channel – which corresponds to 80% inhibition of light transmission aggregometry stimulated by 20 mcM adenosine diphosphate within 15 minutes – was achieved in three of eight patients at the lowest dose and in seven of eight patients at the middle and highest doses.
“Single-dose subcutaneous RUC-4 induces a fast, potent dose-dependent response of platelet inhibition in patients with STEMI presenting for primary PCI,” Dr. ten Berg concluded. “It is therefore promising for prehospital platelet inhibition in STEMI patients, and the results support further research on clinical benefit.”
The double-blind, randomized phase 2b CELEBRATE trial is underway, evaluating 1,668 STEMI patients treated with a 0.110 mg/kg or 0.130 mg/kg dose of RUC-4 or placebo in the ambulance. The coprimary outcomes are restoration of coronary artery blood flow and resolution of ST-segment deviation post-PCI/angiography. Primary completion is set for March 2023.
Marco Valgimigli, MD, who was not involved in the study, said in an interview that RUC-4 has “some theoretical advantages, compared with conventional IIb/IIIa inhibitors, namely the absence of thrombocytopenia which is, however, relatively rare, especially with tirofiban or eptifibatide.”
The subcutaneous approach may also offer an advantage. Yet, if the administration of RUC-4 is “to happen in the ambulance – a setting where an IV line is usually established – whether the subcutaneous versus IV administration of the treatment proves to be advantageous remains to be seen,” said Dr. Valgimigli, from Cardiocentro Ticino Institute, Ente Ospedaliero Cantonale, Lugano, Switzerland.
“We would need to see the results of large randomized trials embracing this treatment option before a clinical decision can be made, especially considering that IIb/IIa inhibitors in the ambulance have been tested in the past but ultimately abandoned,” he said.
Limitations of the study are its open-label design, the fact that iso-TRAP channel assay data were not reported by the VeryifyNow instrument and had to be calculated from the raw data, and the fact that the timing of the RUC-4 injection immediately before PCI does not fully resemble the expected use of RUC-4 in clinical practice, where RUC-4 would be administered at the same time as the aspirin, ticagrelor, and heparin, and about an hour before PCI, ten Berg and colleagues wrote.
CeleCor Therapeutics sponsored the study and provided study materials. Dr. ten Berg reported receiving lecture or consultancy fees from AstraZeneca, Eli Lilly, Daiichi Sankyo, The Medicines Company, AccuMetrics, Boehringer Ingelheim, Bristol-Myers Squibb, Pfizer, Bayer, Ferrer, and Idorsia, and institutional research grants from ZonMw and AstraZeneca. Coauthor Barry S. Coller is an inventor of RUC-4 and a founder, equity holder, and consultant to CeleCor. He also receives royalties from Centocor/Janssen and the VerifyNow assays. Dr. Valgimigli has received grants from Abbott, Terumo, Medicure, and AstraZeneca, and personal fees from Abbott, Chiesi, Bayer, Daiichi Sankyo, Amgen, Terumo, Alvimedica, AstraZeneca, Biosensors, and Idorsia.
A version of this article first appeared on Medscape.com.
FDA okays new drug option for schizophrenia, bipolar I disorder
The U.S. Food and Drug Administration has approved a once-daily oral medication, which is a combination of olanzapine and samidorphan (Lybalvi, Alkermes), for the treatment of schizophrenia and bipolar I disorder.
The drug is approved for the treatment of adults with schizophrenia and for adults with bipolar I disorder as a maintenance monotherapy or to treat acute manic or mixed episodes, as either monotherapy or an adjunct to lithium or valproate.
An atypical antipsychotic, the drug is a combination of olanzapine, an established antipsychotic medication, and samidorphan, a new chemical entity.
“Schizophrenia and bipolar I disorder are complex, chronic diseases, and there remains a persistent need for new medications with proven efficacy and safety. Olanzapine, a highly efficacious atypical antipsychotic, is associated with significant side effects, including weight gain that may impact patients’ treatment experiences and limit its use. With the efficacy of olanzapine and evidence of less weight gain in patients with schizophrenia, Lybalvi brings a welcome new addition to our medication arsenal,” René S. Kahn, MD, PhD, Esther and Joseph Klingenstein professor & chair, department of psychiatry and Behavioral Health System at the Icahn School of Medicine at Mount Sinai, New York, said in a company press release.
In a clinical development program, the drug demonstrated antipsychotic efficacy, safety, and tolerability, including significantly less weight gain than olanzapine in patients with schizophrenia in the ENLIGHTEN-2 study.
The FDA approved Lybalvi under the 505(b)(2) regulatory pathway based on data from 27 clinical studies, including 18 studies evaluating Lybalvi and nine studies evaluating samidorphan alone and the FDA’s findings of the safety and effectiveness of olanzapine in the treatment of bipolar I disorder and schizophrenia. Data suggest that olanzapine-associated weight gain is disease independent, the company reports.
“People living with schizophrenia or bipolar I disorder must evaluate both efficacy and tolerability when making treatment decisions,” Paul Gionfriddo, president and CEO of Mental Health America, said in the same company press release. “We are grateful that companies like Alkermes are driven to continue developing new treatment options in psychiatry that seek to address unmet needs of our community, and we applaud the FDA for considering the experiences of individuals living with these conditions.”
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has approved a once-daily oral medication, which is a combination of olanzapine and samidorphan (Lybalvi, Alkermes), for the treatment of schizophrenia and bipolar I disorder.
The drug is approved for the treatment of adults with schizophrenia and for adults with bipolar I disorder as a maintenance monotherapy or to treat acute manic or mixed episodes, as either monotherapy or an adjunct to lithium or valproate.
An atypical antipsychotic, the drug is a combination of olanzapine, an established antipsychotic medication, and samidorphan, a new chemical entity.
“Schizophrenia and bipolar I disorder are complex, chronic diseases, and there remains a persistent need for new medications with proven efficacy and safety. Olanzapine, a highly efficacious atypical antipsychotic, is associated with significant side effects, including weight gain that may impact patients’ treatment experiences and limit its use. With the efficacy of olanzapine and evidence of less weight gain in patients with schizophrenia, Lybalvi brings a welcome new addition to our medication arsenal,” René S. Kahn, MD, PhD, Esther and Joseph Klingenstein professor & chair, department of psychiatry and Behavioral Health System at the Icahn School of Medicine at Mount Sinai, New York, said in a company press release.
In a clinical development program, the drug demonstrated antipsychotic efficacy, safety, and tolerability, including significantly less weight gain than olanzapine in patients with schizophrenia in the ENLIGHTEN-2 study.
The FDA approved Lybalvi under the 505(b)(2) regulatory pathway based on data from 27 clinical studies, including 18 studies evaluating Lybalvi and nine studies evaluating samidorphan alone and the FDA’s findings of the safety and effectiveness of olanzapine in the treatment of bipolar I disorder and schizophrenia. Data suggest that olanzapine-associated weight gain is disease independent, the company reports.
“People living with schizophrenia or bipolar I disorder must evaluate both efficacy and tolerability when making treatment decisions,” Paul Gionfriddo, president and CEO of Mental Health America, said in the same company press release. “We are grateful that companies like Alkermes are driven to continue developing new treatment options in psychiatry that seek to address unmet needs of our community, and we applaud the FDA for considering the experiences of individuals living with these conditions.”
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has approved a once-daily oral medication, which is a combination of olanzapine and samidorphan (Lybalvi, Alkermes), for the treatment of schizophrenia and bipolar I disorder.
The drug is approved for the treatment of adults with schizophrenia and for adults with bipolar I disorder as a maintenance monotherapy or to treat acute manic or mixed episodes, as either monotherapy or an adjunct to lithium or valproate.
An atypical antipsychotic, the drug is a combination of olanzapine, an established antipsychotic medication, and samidorphan, a new chemical entity.
“Schizophrenia and bipolar I disorder are complex, chronic diseases, and there remains a persistent need for new medications with proven efficacy and safety. Olanzapine, a highly efficacious atypical antipsychotic, is associated with significant side effects, including weight gain that may impact patients’ treatment experiences and limit its use. With the efficacy of olanzapine and evidence of less weight gain in patients with schizophrenia, Lybalvi brings a welcome new addition to our medication arsenal,” René S. Kahn, MD, PhD, Esther and Joseph Klingenstein professor & chair, department of psychiatry and Behavioral Health System at the Icahn School of Medicine at Mount Sinai, New York, said in a company press release.
In a clinical development program, the drug demonstrated antipsychotic efficacy, safety, and tolerability, including significantly less weight gain than olanzapine in patients with schizophrenia in the ENLIGHTEN-2 study.
The FDA approved Lybalvi under the 505(b)(2) regulatory pathway based on data from 27 clinical studies, including 18 studies evaluating Lybalvi and nine studies evaluating samidorphan alone and the FDA’s findings of the safety and effectiveness of olanzapine in the treatment of bipolar I disorder and schizophrenia. Data suggest that olanzapine-associated weight gain is disease independent, the company reports.
“People living with schizophrenia or bipolar I disorder must evaluate both efficacy and tolerability when making treatment decisions,” Paul Gionfriddo, president and CEO of Mental Health America, said in the same company press release. “We are grateful that companies like Alkermes are driven to continue developing new treatment options in psychiatry that seek to address unmet needs of our community, and we applaud the FDA for considering the experiences of individuals living with these conditions.”
A version of this article first appeared on Medscape.com.
Children aged 12-15 years continue to close COVID-19 vaccination gap
More children aged 12-15 years already have received at least one dose of a COVID-19 vaccine than have 16- and 17-year-olds, based on data from the Centers for Disease Control and Prevention.
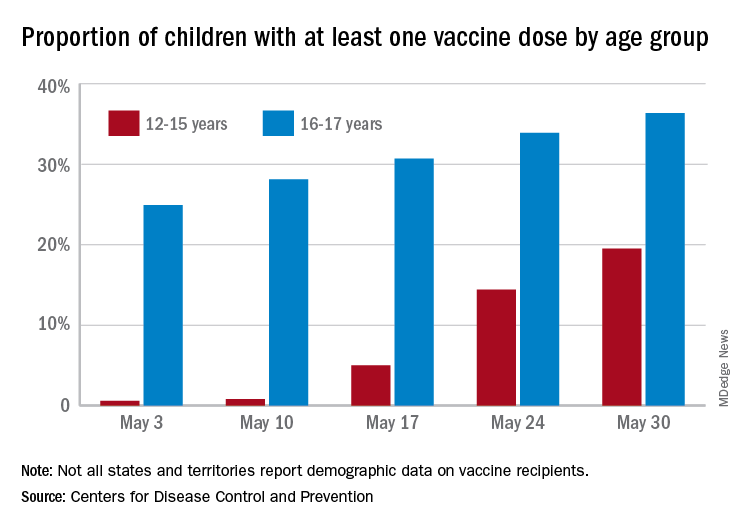
with those figures representing increases of 31.6% and 6.6% in the past week, respectively. Since the overall size of the 12-15 population is much larger, however, the proportion vaccinated is still smaller: 19.5% to 36.4%, according to the CDC’s COVID Data Tracker.
A look at full vaccination status shows that only 0.7% of those aged 12-15 years have received both doses of a two-dose vaccine or one dose of the single-shot variety, compared with 24% of those aged 16-17. For the country as a whole, 50.5% of all ages have received at least one dose and 40.7% are fully vaccinated, the CDC said.
Children aged 12-15 represent the largest share of the U.S. population (23.4%) initiating vaccination in the 14 days ending May 30, while children aged 16-17 made up just 4.5% of those getting their first dose. The younger group’s later entry into the vaccination pool shows up again when looking at completion rates, though, representing just 0.4% of all Americans who reached full vaccination during that same 14-day period, compared with 4.6% of the older children, the CDC data show.
Not all states are reporting data such as age for vaccine recipients, the CDC noted, and there are other variables that affect data collection. “Demographic data ... might differ by populations prioritized within each state or jurisdiction’s vaccination phase. Every geographic area has a different racial and ethnic composition, and not all are in the same vaccination phase,” the CDC said.
More children aged 12-15 years already have received at least one dose of a COVID-19 vaccine than have 16- and 17-year-olds, based on data from the Centers for Disease Control and Prevention.

with those figures representing increases of 31.6% and 6.6% in the past week, respectively. Since the overall size of the 12-15 population is much larger, however, the proportion vaccinated is still smaller: 19.5% to 36.4%, according to the CDC’s COVID Data Tracker.
A look at full vaccination status shows that only 0.7% of those aged 12-15 years have received both doses of a two-dose vaccine or one dose of the single-shot variety, compared with 24% of those aged 16-17. For the country as a whole, 50.5% of all ages have received at least one dose and 40.7% are fully vaccinated, the CDC said.
Children aged 12-15 represent the largest share of the U.S. population (23.4%) initiating vaccination in the 14 days ending May 30, while children aged 16-17 made up just 4.5% of those getting their first dose. The younger group’s later entry into the vaccination pool shows up again when looking at completion rates, though, representing just 0.4% of all Americans who reached full vaccination during that same 14-day period, compared with 4.6% of the older children, the CDC data show.
Not all states are reporting data such as age for vaccine recipients, the CDC noted, and there are other variables that affect data collection. “Demographic data ... might differ by populations prioritized within each state or jurisdiction’s vaccination phase. Every geographic area has a different racial and ethnic composition, and not all are in the same vaccination phase,” the CDC said.
More children aged 12-15 years already have received at least one dose of a COVID-19 vaccine than have 16- and 17-year-olds, based on data from the Centers for Disease Control and Prevention.

with those figures representing increases of 31.6% and 6.6% in the past week, respectively. Since the overall size of the 12-15 population is much larger, however, the proportion vaccinated is still smaller: 19.5% to 36.4%, according to the CDC’s COVID Data Tracker.
A look at full vaccination status shows that only 0.7% of those aged 12-15 years have received both doses of a two-dose vaccine or one dose of the single-shot variety, compared with 24% of those aged 16-17. For the country as a whole, 50.5% of all ages have received at least one dose and 40.7% are fully vaccinated, the CDC said.
Children aged 12-15 represent the largest share of the U.S. population (23.4%) initiating vaccination in the 14 days ending May 30, while children aged 16-17 made up just 4.5% of those getting their first dose. The younger group’s later entry into the vaccination pool shows up again when looking at completion rates, though, representing just 0.4% of all Americans who reached full vaccination during that same 14-day period, compared with 4.6% of the older children, the CDC data show.
Not all states are reporting data such as age for vaccine recipients, the CDC noted, and there are other variables that affect data collection. “Demographic data ... might differ by populations prioritized within each state or jurisdiction’s vaccination phase. Every geographic area has a different racial and ethnic composition, and not all are in the same vaccination phase,” the CDC said.
First drug for lung cancer with KRAS mutation gains FDA approval
The first drug to target KRAS mutations in non–small cell lung cancer (NSCLC) has been approved by the Food and Drug Administration.
KRAS mutations are the most common mutations to occur in NSCLC tumors, accounting for about 25% of them, but for a long time they appeared to be resistant to drug therapy.
The new drug, sotorasib (Lumakras), specifically targets the KRAS G12C mutation, which accounts for about 13% of NSCLC mutations.
It is considered to be something of a breakthrough in cancer research. When clinical data on the new drug (from 126 patients) were presented last year at the World Conference on Lung Cancer, lung cancer experts greeted the results enthusiastically, as reported by Medscape Medical News at the time.
“This is a historic milestone in lung cancer therapy. After four decades of scientific efforts in targeting KRAS, sotorasib has potential to be the first targeted treatment option for this patient population with a high unmet need,” Bob T. Li, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York, said at the time.
Now, in a press release from the manufacturer, Amgen, he said: “Sotorasib represents a major advancement in oncology and changes the treatment paradigm for patients with KRAS G12C-mutated non–small cell lung cancer.
“Patients with non–small cell lung cancer who have progressed beyond first-line treatment face a poor prognosis and have limited treatment options available to them. Sotorasib delivers a new option for these patients, and it is the first KRAS-targeted therapy to be approved after nearly four decades of research,” he added.
Details of clinical data
This is an accelerated approval based on response rate data.
The FDA notes that the clinical data come from a study of 124 patients with locally advanced or metastatic KRAS G12C-mutated NSCLC with disease progression after receiving an immune checkpoint inhibitor and/or platinum-based chemotherapy.
The major outcome measured was overall response rate (ORR), which was 36%. Of the patients who responded, 58% had a duration of response of 6 months or longer.
Sotorasib was approved at a dose of 960 mg, and this dose was based on available clinical data as well as pharmacokinetic and pharmacodynamic modeling, the FDA noted. As part of the evaluation for this accelerated approval, the agency is requiring a postmarketing trial to investigate whether a lower dose will have a similar clinical effect.
The most common side effects include diarrhea, musculoskeletal pain, nausea, fatigue, liver damage, and cough. Sotorasib should not be used if patients develop symptoms of interstitial lung disease, and should be permanently discontinued if interstitial lung disease is confirmed.
Patients on sotorasib should have liver function tests prior to starting and while taking the drug; if liver damage develops, the drug should be stopped or the dose reduced. Patients should avoid taking acid-reducing agents, drugs that induce or are substrates for certain enzymes in the liver, and drugs that are substrates of P-glycoprotein (P-gp).
Companion diagnostic tests also approved
Along with the new drug, the FDA approved two companion diagnostic tests – the QIAGEN therascreen KRAS RGQ PCR kit (approval granted to QIAGEN GmbH) for analyzing tumor tissue and the Guardant360 CDx (approval granted to Guardant Health) for analyzing plasma specimens to determine if the KRAS G12C mutation is present. The agency notes that if the plasma test comes back negative, the patient’s tumor tissue should be tested.
A version of this article first appeared on Medscape.com.
The first drug to target KRAS mutations in non–small cell lung cancer (NSCLC) has been approved by the Food and Drug Administration.
KRAS mutations are the most common mutations to occur in NSCLC tumors, accounting for about 25% of them, but for a long time they appeared to be resistant to drug therapy.
The new drug, sotorasib (Lumakras), specifically targets the KRAS G12C mutation, which accounts for about 13% of NSCLC mutations.
It is considered to be something of a breakthrough in cancer research. When clinical data on the new drug (from 126 patients) were presented last year at the World Conference on Lung Cancer, lung cancer experts greeted the results enthusiastically, as reported by Medscape Medical News at the time.
“This is a historic milestone in lung cancer therapy. After four decades of scientific efforts in targeting KRAS, sotorasib has potential to be the first targeted treatment option for this patient population with a high unmet need,” Bob T. Li, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York, said at the time.
Now, in a press release from the manufacturer, Amgen, he said: “Sotorasib represents a major advancement in oncology and changes the treatment paradigm for patients with KRAS G12C-mutated non–small cell lung cancer.
“Patients with non–small cell lung cancer who have progressed beyond first-line treatment face a poor prognosis and have limited treatment options available to them. Sotorasib delivers a new option for these patients, and it is the first KRAS-targeted therapy to be approved after nearly four decades of research,” he added.
Details of clinical data
This is an accelerated approval based on response rate data.
The FDA notes that the clinical data come from a study of 124 patients with locally advanced or metastatic KRAS G12C-mutated NSCLC with disease progression after receiving an immune checkpoint inhibitor and/or platinum-based chemotherapy.
The major outcome measured was overall response rate (ORR), which was 36%. Of the patients who responded, 58% had a duration of response of 6 months or longer.
Sotorasib was approved at a dose of 960 mg, and this dose was based on available clinical data as well as pharmacokinetic and pharmacodynamic modeling, the FDA noted. As part of the evaluation for this accelerated approval, the agency is requiring a postmarketing trial to investigate whether a lower dose will have a similar clinical effect.
The most common side effects include diarrhea, musculoskeletal pain, nausea, fatigue, liver damage, and cough. Sotorasib should not be used if patients develop symptoms of interstitial lung disease, and should be permanently discontinued if interstitial lung disease is confirmed.
Patients on sotorasib should have liver function tests prior to starting and while taking the drug; if liver damage develops, the drug should be stopped or the dose reduced. Patients should avoid taking acid-reducing agents, drugs that induce or are substrates for certain enzymes in the liver, and drugs that are substrates of P-glycoprotein (P-gp).
Companion diagnostic tests also approved
Along with the new drug, the FDA approved two companion diagnostic tests – the QIAGEN therascreen KRAS RGQ PCR kit (approval granted to QIAGEN GmbH) for analyzing tumor tissue and the Guardant360 CDx (approval granted to Guardant Health) for analyzing plasma specimens to determine if the KRAS G12C mutation is present. The agency notes that if the plasma test comes back negative, the patient’s tumor tissue should be tested.
A version of this article first appeared on Medscape.com.
The first drug to target KRAS mutations in non–small cell lung cancer (NSCLC) has been approved by the Food and Drug Administration.
KRAS mutations are the most common mutations to occur in NSCLC tumors, accounting for about 25% of them, but for a long time they appeared to be resistant to drug therapy.
The new drug, sotorasib (Lumakras), specifically targets the KRAS G12C mutation, which accounts for about 13% of NSCLC mutations.
It is considered to be something of a breakthrough in cancer research. When clinical data on the new drug (from 126 patients) were presented last year at the World Conference on Lung Cancer, lung cancer experts greeted the results enthusiastically, as reported by Medscape Medical News at the time.
“This is a historic milestone in lung cancer therapy. After four decades of scientific efforts in targeting KRAS, sotorasib has potential to be the first targeted treatment option for this patient population with a high unmet need,” Bob T. Li, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York, said at the time.
Now, in a press release from the manufacturer, Amgen, he said: “Sotorasib represents a major advancement in oncology and changes the treatment paradigm for patients with KRAS G12C-mutated non–small cell lung cancer.
“Patients with non–small cell lung cancer who have progressed beyond first-line treatment face a poor prognosis and have limited treatment options available to them. Sotorasib delivers a new option for these patients, and it is the first KRAS-targeted therapy to be approved after nearly four decades of research,” he added.
Details of clinical data
This is an accelerated approval based on response rate data.
The FDA notes that the clinical data come from a study of 124 patients with locally advanced or metastatic KRAS G12C-mutated NSCLC with disease progression after receiving an immune checkpoint inhibitor and/or platinum-based chemotherapy.
The major outcome measured was overall response rate (ORR), which was 36%. Of the patients who responded, 58% had a duration of response of 6 months or longer.
Sotorasib was approved at a dose of 960 mg, and this dose was based on available clinical data as well as pharmacokinetic and pharmacodynamic modeling, the FDA noted. As part of the evaluation for this accelerated approval, the agency is requiring a postmarketing trial to investigate whether a lower dose will have a similar clinical effect.
The most common side effects include diarrhea, musculoskeletal pain, nausea, fatigue, liver damage, and cough. Sotorasib should not be used if patients develop symptoms of interstitial lung disease, and should be permanently discontinued if interstitial lung disease is confirmed.
Patients on sotorasib should have liver function tests prior to starting and while taking the drug; if liver damage develops, the drug should be stopped or the dose reduced. Patients should avoid taking acid-reducing agents, drugs that induce or are substrates for certain enzymes in the liver, and drugs that are substrates of P-glycoprotein (P-gp).
Companion diagnostic tests also approved
Along with the new drug, the FDA approved two companion diagnostic tests – the QIAGEN therascreen KRAS RGQ PCR kit (approval granted to QIAGEN GmbH) for analyzing tumor tissue and the Guardant360 CDx (approval granted to Guardant Health) for analyzing plasma specimens to determine if the KRAS G12C mutation is present. The agency notes that if the plasma test comes back negative, the patient’s tumor tissue should be tested.
A version of this article first appeared on Medscape.com.