User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
COVID-19 vaccines equally effective in patients on dialysis
Two doses of either the Pfizer-BioNTech COVID-19 vaccine or the Oxford AstraZeneca alternative provide equal and significant protection against severe disease in patients on hemodialysis who have contracted SARS-CoV-2 infection, results of a multicenter observational study indicate.
Following two doses of either vaccine, the risk of hospital admission was 75% lower among vaccinated patients while the risk of death was 88% lower, compared with those who remained unvaccinated.
No difference was seen between the two vaccine types in terms of outcome severity, and there was no loss of protection in patients over the age of 65 or with increasing time since vaccination, the authors add. The need for oxygen and ventilation was also halved among those who had received two shots, compared with those who had not.
“The coronavirus disease 2019 (COVID-19) pandemic has had a devastating effect on the CKD (chronic kidney disease) community, particularly for individuals receiving maintenance dialysis,” Matthew Oliver, MD, University of Toronto, and Peter Blake, MD, Western University, London, Ont., write in an editorial published with the study.
“Overall, [this and other studies] show that COVID-19 vaccination in the maintenance dialysis population provides moderate protection against acquiring SARS-CoV-2 infection but is highly protective against severe outcomes,” they conclude.
The study was published in the June issue of the Clinical Journal of the American Society of Nephrology.
Severe outcomes observed less in patients who tested positive
The cohort included 1,323 patients on hemodialysis who tested positive on PCR testing to SARS-CoV-2 during a surveillance interval between December 2020 and September 2021, report, Damien Ashby, MD, Hammersmith Hospital, London, and colleagues report.
Among those who tested positive, 79% had not been vaccinated, 7% tested positive after their first dose of either vaccine, and 14% tested positive at least 10 days beyond their second dose.
The course of illness was mild in 61% of patients in that they did not require hospital admission, investigators note. Oxygen support was required by 29% of those who tested positive, and 13% died before 28 days, they added. Among those who died within 28 days of testing positive, 90% of the deaths were deemed to be caused by the virus itself.
“Compared with unvaccinated patients, severe COVID-19 outcomes were observed less than half as often in patients testing positive for SARS-Co-V-2 at least 10 days after the second dose,” Dr. Ashby and colleagues emphasize.
“And the protection from severe illness associated with vaccination was most obvious in patients over 65 years, in whom severe COVID-19 outcomes were reduced at least as much after vaccination as in their younger peers,” they add. Following vaccination with the Pfizer-BioNTech vaccine, antibody levels in patients on dialysis were comparable with those of healthy controls.
In contrast, this was not the case for the Oxford AstraZeneca vaccine where neutralizing titers in patients who received the vaccine were less effective against most variants. Despite its ability to produce comparable immunogenicity, the Oxford AstraZeneca vaccine was clearly associated with clinical protection against severe illness, the authors stress.
They also note that their results are relevant to vaccine uptake in the dialysis population where vaccine hesitancy remains a problem. “This study may, therefore, be useful in reducing vaccine hesitancy, which has resulted in low uptake in some countries (for example, Australia, where almost a quarter of patients on dialysis declined),” Dr. Ashby and colleagues point out.
Although significant vulnerability in the dialysis population remains, “this population has much to gain from vaccination, regardless of age or vaccine type,” the authors underscore.
CKD community quick to prioritize vaccine
As the editorialists point out, leaders in the CKD community were quick – and successful – in prioritizing vaccination in the dialysis population right from the beginning of the pandemic. For example, in Ontario, 90% of the maintenance dialysis population had received two doses of a COVID-19 vaccine by September 2021 and 78% had received three doses by January 2022.
Moreover, in Ontario, “our group found that two doses of mRNA vaccine reduced the risk of infection by 69%,” Dr. Oliver and Dr. Blake point out. U.S. researchers also found that the Pfizer mRNA vaccine reduced infection risk from COVID-19 by 79% while the Moderna mRNA vaccine reduced that risk by 73%. Vaccine effectiveness (VE) in the real-world setting indicates that COVID-19 vaccines provide moderate protection against being infected with the SARS-Co-V-2 virus, as the editorialists note.
However, “the VE for preventing severe outcomes is clinically more important for patients on dialysis because their risk of [morbid] events is high,” Dr. Oliver and Dr. Blake write. Indeed, their own study estimated that two doses of an mRNA vaccine reduced severe outcomes by 83%, “a greater benefit than for infection prevention,” they stress.
The editorialists caution that the SARS-CoV-2 virus continues to mutate and serology studies do show that vaccine-induced immunity does wane over time. Thus, while the COVID-19 pandemic is ever-changing, “we should conduct [VE] studies rigorously and expeditiously to bolster the case for prioritizing vaccination in the dialysis population,” Dr. Oliver and Dr. Blake recommend.
Need to increase vaccine acceptance
Commenting on the study, Uwe K.H. Korst from Bensheim, Germany, notes that COVID-19 is a daily reminder of how fragile life is for people with CKD. “Daily, the virus continues its horrific and unprecedented course through immunocompromised and immunosuppressed patients with kidney disease,” he writes.
Thus, Mr. Korst continues to call for additional education for health care professionals, patients, and the public to increase vaccine acceptance as well as more research to better understand the virus and its long-term consequences.
“Finally, patients need to express their needs, and physicians need to listen to patients’ voices,” Mr. Korst advises.
Dr. Oliver is a contracted medical lead of Ontario Renal Network and owner of Oliver Medical Management for which he holds patents and has received royalties. He has also reported receiving honoraria for speaking from Baxter Healthcare and participating in advisory boards for Amgen and Janssen. Dr. Blake has reported receiving honoraria from Baxter Global for speaking engagements and serves on the editorial board for the American Journal of Nephrology. Dr. Ashby and Dr. Korst have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Two doses of either the Pfizer-BioNTech COVID-19 vaccine or the Oxford AstraZeneca alternative provide equal and significant protection against severe disease in patients on hemodialysis who have contracted SARS-CoV-2 infection, results of a multicenter observational study indicate.
Following two doses of either vaccine, the risk of hospital admission was 75% lower among vaccinated patients while the risk of death was 88% lower, compared with those who remained unvaccinated.
No difference was seen between the two vaccine types in terms of outcome severity, and there was no loss of protection in patients over the age of 65 or with increasing time since vaccination, the authors add. The need for oxygen and ventilation was also halved among those who had received two shots, compared with those who had not.
“The coronavirus disease 2019 (COVID-19) pandemic has had a devastating effect on the CKD (chronic kidney disease) community, particularly for individuals receiving maintenance dialysis,” Matthew Oliver, MD, University of Toronto, and Peter Blake, MD, Western University, London, Ont., write in an editorial published with the study.
“Overall, [this and other studies] show that COVID-19 vaccination in the maintenance dialysis population provides moderate protection against acquiring SARS-CoV-2 infection but is highly protective against severe outcomes,” they conclude.
The study was published in the June issue of the Clinical Journal of the American Society of Nephrology.
Severe outcomes observed less in patients who tested positive
The cohort included 1,323 patients on hemodialysis who tested positive on PCR testing to SARS-CoV-2 during a surveillance interval between December 2020 and September 2021, report, Damien Ashby, MD, Hammersmith Hospital, London, and colleagues report.
Among those who tested positive, 79% had not been vaccinated, 7% tested positive after their first dose of either vaccine, and 14% tested positive at least 10 days beyond their second dose.
The course of illness was mild in 61% of patients in that they did not require hospital admission, investigators note. Oxygen support was required by 29% of those who tested positive, and 13% died before 28 days, they added. Among those who died within 28 days of testing positive, 90% of the deaths were deemed to be caused by the virus itself.
“Compared with unvaccinated patients, severe COVID-19 outcomes were observed less than half as often in patients testing positive for SARS-Co-V-2 at least 10 days after the second dose,” Dr. Ashby and colleagues emphasize.
“And the protection from severe illness associated with vaccination was most obvious in patients over 65 years, in whom severe COVID-19 outcomes were reduced at least as much after vaccination as in their younger peers,” they add. Following vaccination with the Pfizer-BioNTech vaccine, antibody levels in patients on dialysis were comparable with those of healthy controls.
In contrast, this was not the case for the Oxford AstraZeneca vaccine where neutralizing titers in patients who received the vaccine were less effective against most variants. Despite its ability to produce comparable immunogenicity, the Oxford AstraZeneca vaccine was clearly associated with clinical protection against severe illness, the authors stress.
They also note that their results are relevant to vaccine uptake in the dialysis population where vaccine hesitancy remains a problem. “This study may, therefore, be useful in reducing vaccine hesitancy, which has resulted in low uptake in some countries (for example, Australia, where almost a quarter of patients on dialysis declined),” Dr. Ashby and colleagues point out.
Although significant vulnerability in the dialysis population remains, “this population has much to gain from vaccination, regardless of age or vaccine type,” the authors underscore.
CKD community quick to prioritize vaccine
As the editorialists point out, leaders in the CKD community were quick – and successful – in prioritizing vaccination in the dialysis population right from the beginning of the pandemic. For example, in Ontario, 90% of the maintenance dialysis population had received two doses of a COVID-19 vaccine by September 2021 and 78% had received three doses by January 2022.
Moreover, in Ontario, “our group found that two doses of mRNA vaccine reduced the risk of infection by 69%,” Dr. Oliver and Dr. Blake point out. U.S. researchers also found that the Pfizer mRNA vaccine reduced infection risk from COVID-19 by 79% while the Moderna mRNA vaccine reduced that risk by 73%. Vaccine effectiveness (VE) in the real-world setting indicates that COVID-19 vaccines provide moderate protection against being infected with the SARS-Co-V-2 virus, as the editorialists note.
However, “the VE for preventing severe outcomes is clinically more important for patients on dialysis because their risk of [morbid] events is high,” Dr. Oliver and Dr. Blake write. Indeed, their own study estimated that two doses of an mRNA vaccine reduced severe outcomes by 83%, “a greater benefit than for infection prevention,” they stress.
The editorialists caution that the SARS-CoV-2 virus continues to mutate and serology studies do show that vaccine-induced immunity does wane over time. Thus, while the COVID-19 pandemic is ever-changing, “we should conduct [VE] studies rigorously and expeditiously to bolster the case for prioritizing vaccination in the dialysis population,” Dr. Oliver and Dr. Blake recommend.
Need to increase vaccine acceptance
Commenting on the study, Uwe K.H. Korst from Bensheim, Germany, notes that COVID-19 is a daily reminder of how fragile life is for people with CKD. “Daily, the virus continues its horrific and unprecedented course through immunocompromised and immunosuppressed patients with kidney disease,” he writes.
Thus, Mr. Korst continues to call for additional education for health care professionals, patients, and the public to increase vaccine acceptance as well as more research to better understand the virus and its long-term consequences.
“Finally, patients need to express their needs, and physicians need to listen to patients’ voices,” Mr. Korst advises.
Dr. Oliver is a contracted medical lead of Ontario Renal Network and owner of Oliver Medical Management for which he holds patents and has received royalties. He has also reported receiving honoraria for speaking from Baxter Healthcare and participating in advisory boards for Amgen and Janssen. Dr. Blake has reported receiving honoraria from Baxter Global for speaking engagements and serves on the editorial board for the American Journal of Nephrology. Dr. Ashby and Dr. Korst have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Two doses of either the Pfizer-BioNTech COVID-19 vaccine or the Oxford AstraZeneca alternative provide equal and significant protection against severe disease in patients on hemodialysis who have contracted SARS-CoV-2 infection, results of a multicenter observational study indicate.
Following two doses of either vaccine, the risk of hospital admission was 75% lower among vaccinated patients while the risk of death was 88% lower, compared with those who remained unvaccinated.
No difference was seen between the two vaccine types in terms of outcome severity, and there was no loss of protection in patients over the age of 65 or with increasing time since vaccination, the authors add. The need for oxygen and ventilation was also halved among those who had received two shots, compared with those who had not.
“The coronavirus disease 2019 (COVID-19) pandemic has had a devastating effect on the CKD (chronic kidney disease) community, particularly for individuals receiving maintenance dialysis,” Matthew Oliver, MD, University of Toronto, and Peter Blake, MD, Western University, London, Ont., write in an editorial published with the study.
“Overall, [this and other studies] show that COVID-19 vaccination in the maintenance dialysis population provides moderate protection against acquiring SARS-CoV-2 infection but is highly protective against severe outcomes,” they conclude.
The study was published in the June issue of the Clinical Journal of the American Society of Nephrology.
Severe outcomes observed less in patients who tested positive
The cohort included 1,323 patients on hemodialysis who tested positive on PCR testing to SARS-CoV-2 during a surveillance interval between December 2020 and September 2021, report, Damien Ashby, MD, Hammersmith Hospital, London, and colleagues report.
Among those who tested positive, 79% had not been vaccinated, 7% tested positive after their first dose of either vaccine, and 14% tested positive at least 10 days beyond their second dose.
The course of illness was mild in 61% of patients in that they did not require hospital admission, investigators note. Oxygen support was required by 29% of those who tested positive, and 13% died before 28 days, they added. Among those who died within 28 days of testing positive, 90% of the deaths were deemed to be caused by the virus itself.
“Compared with unvaccinated patients, severe COVID-19 outcomes were observed less than half as often in patients testing positive for SARS-Co-V-2 at least 10 days after the second dose,” Dr. Ashby and colleagues emphasize.
“And the protection from severe illness associated with vaccination was most obvious in patients over 65 years, in whom severe COVID-19 outcomes were reduced at least as much after vaccination as in their younger peers,” they add. Following vaccination with the Pfizer-BioNTech vaccine, antibody levels in patients on dialysis were comparable with those of healthy controls.
In contrast, this was not the case for the Oxford AstraZeneca vaccine where neutralizing titers in patients who received the vaccine were less effective against most variants. Despite its ability to produce comparable immunogenicity, the Oxford AstraZeneca vaccine was clearly associated with clinical protection against severe illness, the authors stress.
They also note that their results are relevant to vaccine uptake in the dialysis population where vaccine hesitancy remains a problem. “This study may, therefore, be useful in reducing vaccine hesitancy, which has resulted in low uptake in some countries (for example, Australia, where almost a quarter of patients on dialysis declined),” Dr. Ashby and colleagues point out.
Although significant vulnerability in the dialysis population remains, “this population has much to gain from vaccination, regardless of age or vaccine type,” the authors underscore.
CKD community quick to prioritize vaccine
As the editorialists point out, leaders in the CKD community were quick – and successful – in prioritizing vaccination in the dialysis population right from the beginning of the pandemic. For example, in Ontario, 90% of the maintenance dialysis population had received two doses of a COVID-19 vaccine by September 2021 and 78% had received three doses by January 2022.
Moreover, in Ontario, “our group found that two doses of mRNA vaccine reduced the risk of infection by 69%,” Dr. Oliver and Dr. Blake point out. U.S. researchers also found that the Pfizer mRNA vaccine reduced infection risk from COVID-19 by 79% while the Moderna mRNA vaccine reduced that risk by 73%. Vaccine effectiveness (VE) in the real-world setting indicates that COVID-19 vaccines provide moderate protection against being infected with the SARS-Co-V-2 virus, as the editorialists note.
However, “the VE for preventing severe outcomes is clinically more important for patients on dialysis because their risk of [morbid] events is high,” Dr. Oliver and Dr. Blake write. Indeed, their own study estimated that two doses of an mRNA vaccine reduced severe outcomes by 83%, “a greater benefit than for infection prevention,” they stress.
The editorialists caution that the SARS-CoV-2 virus continues to mutate and serology studies do show that vaccine-induced immunity does wane over time. Thus, while the COVID-19 pandemic is ever-changing, “we should conduct [VE] studies rigorously and expeditiously to bolster the case for prioritizing vaccination in the dialysis population,” Dr. Oliver and Dr. Blake recommend.
Need to increase vaccine acceptance
Commenting on the study, Uwe K.H. Korst from Bensheim, Germany, notes that COVID-19 is a daily reminder of how fragile life is for people with CKD. “Daily, the virus continues its horrific and unprecedented course through immunocompromised and immunosuppressed patients with kidney disease,” he writes.
Thus, Mr. Korst continues to call for additional education for health care professionals, patients, and the public to increase vaccine acceptance as well as more research to better understand the virus and its long-term consequences.
“Finally, patients need to express their needs, and physicians need to listen to patients’ voices,” Mr. Korst advises.
Dr. Oliver is a contracted medical lead of Ontario Renal Network and owner of Oliver Medical Management for which he holds patents and has received royalties. He has also reported receiving honoraria for speaking from Baxter Healthcare and participating in advisory boards for Amgen and Janssen. Dr. Blake has reported receiving honoraria from Baxter Global for speaking engagements and serves on the editorial board for the American Journal of Nephrology. Dr. Ashby and Dr. Korst have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
CDC says about 20% get long COVID. New models try to define it
As the number of people reporting persistent, and sometimes debilitating, symptoms from COVID-19 increases, researchers have struggled to pinpoint exactly how common so-called “long COVID” is, as well as how to clearly define exactly who has it or who is likely to get it.
Now, Centers for Disease Control and Prevention researchers have concluded that one in five adults aged 18 and older have at least one health condition that might be related to their previous COVID-19 illness; that number goes up to one in four among those 65 and older. Their data was published in the CDC’s Morbidity and Mortality Weekly Report.
The conditions associated with what’s been officially termed postacute sequelae of COVID-19, or PASC, include kidney failure, blood clots, other vascular issues, respiratory issues, heart problems, mental health or neurologic problems, and musculoskeletal conditions. But none of those conditions is unique to long COVID.
Another new study, published in The Lancet Digital Health, is trying to help better characterize what long COVID is, and what it isn’t.
that could help identify those likely to develop it.
CDC data
The CDC team came to its conclusions by evaluating the EHRs of more than 353,000 adults who were diagnosed with COVID-19 or got a positive test result, then comparing those records with 1.6 million patients who had a medical visit in the same month without a positive test result or a COVID-19 diagnosis.
They looked at data from March 2020 to November 2021, tagging 26 conditions often linked to post-COVID issues.
Overall, more than 38% of the COVID patients and 16% of those without COVID had at least one of these 26 conditions. They assessed the absolute risk difference between the patients and the non-COVID patients who developed one of the conditions, finding a 20.8–percentage point difference for those 18-64, yielding the one in five figure, and a 26.9–percentage point difference for those 65 and above, translating to about one in four.
“These findings suggest the need for increased awareness for post-COVID conditions so that improved post-COVID care and management of patients who survived COVID-19 can be developed and implemented,” said study author Lara Bull-Otterson, PhD, MPH, colead of data analytics at the Healthcare Data Advisory Unit of the CDC.
Pinpointing long COVID characteristics
Long COVID is difficult to identify, because many of its symptoms are similar to those of other conditions, so researchers are looking for better ways to characterize it to help improve both diagnosis and treatment.
Researchers on the Lancet study evaluated data from the National COVID Cohort Collaborative, N3C, a national NIH database that includes information from more than 8 million people. The team looked at the health records of 98,000 adult COVID patients and used that information, along with data from about nearly 600 long-COVID patients treated at three long-COVID clinics, to create three machine learning models for identifying long-COVID patients.
The models aimed to identify long-COVID patients in three groups: all patients, those hospitalized with COVID, and those with COVID but not hospitalized. The models were judged by the researchers to be accurate because those identified at risk for long COVID from the database were similar to those actually treated for long COVID at the clinics.
“Our algorithm is not intended to diagnose long COVID,” said lead author Emily Pfaff, PhD, research assistant professor of medicine at the University of North Carolina at Chapel Hill. “Rather, it is intended to identify patients in EHR data who ‘look like’ patients seen by physicians for long COVID.’’
Next, the researchers say, they will incorporate the new patterns they found with a diagnosis code for COVID and include it in the models to further test their accuracy. The models could also be used to help recruit patients for clinical trials, the researchers say.
Perspective and caveats
The figures of one in five and one in four found by the CDC researchers don’t surprise David Putrino, PT, PhD, director of rehabilitation innovation for Mount Sinai Health System in New York and director of its Abilities Research Center, which cares for long-COVID patients.
“Those numbers are high and it’s alarming,” he said. “But we’ve been sounding the alarm for quite some time, and we’ve been assuming that about one in five end up with long COVID.”
He does see a limitation to the CDC research – that some symptoms could have emerged later, and some in the control group could have had an undiagnosed COVID infection and gone on to develop long COVID.
As for machine learning, “this is something we need to approach with caution,” Dr. Putrino said. “There are a lot of variables we don’t understand about long COVID,’’ and that could result in spurious conclusions.
“Although I am supportive of this work going on, I am saying, ‘Scrutinize the tools with a grain of salt.’ Electronic records, Dr. Putrino points out, include information that the doctors enter, not what the patient says.
Dr. Pfaff responds: “It is entirely appropriate to approach both machine learning and EHR data with relevant caveats in mind. There are many clinical factors that are not recorded in the EHR, and the EHR is not representative of all persons with long COVID.” Those data can only reflect those who seek care for a condition, a natural limitation.
When it comes to algorithms, they are limited by data they have access to, such as the electronic health records in this research. However, the immense size and diversity in the data used “does allow us to make some assertations with much more confidence than if we were using data from a single or small number of health care systems,” she said.
A version of this article first appeared on Medscape.com.
As the number of people reporting persistent, and sometimes debilitating, symptoms from COVID-19 increases, researchers have struggled to pinpoint exactly how common so-called “long COVID” is, as well as how to clearly define exactly who has it or who is likely to get it.
Now, Centers for Disease Control and Prevention researchers have concluded that one in five adults aged 18 and older have at least one health condition that might be related to their previous COVID-19 illness; that number goes up to one in four among those 65 and older. Their data was published in the CDC’s Morbidity and Mortality Weekly Report.
The conditions associated with what’s been officially termed postacute sequelae of COVID-19, or PASC, include kidney failure, blood clots, other vascular issues, respiratory issues, heart problems, mental health or neurologic problems, and musculoskeletal conditions. But none of those conditions is unique to long COVID.
Another new study, published in The Lancet Digital Health, is trying to help better characterize what long COVID is, and what it isn’t.
that could help identify those likely to develop it.
CDC data
The CDC team came to its conclusions by evaluating the EHRs of more than 353,000 adults who were diagnosed with COVID-19 or got a positive test result, then comparing those records with 1.6 million patients who had a medical visit in the same month without a positive test result or a COVID-19 diagnosis.
They looked at data from March 2020 to November 2021, tagging 26 conditions often linked to post-COVID issues.
Overall, more than 38% of the COVID patients and 16% of those without COVID had at least one of these 26 conditions. They assessed the absolute risk difference between the patients and the non-COVID patients who developed one of the conditions, finding a 20.8–percentage point difference for those 18-64, yielding the one in five figure, and a 26.9–percentage point difference for those 65 and above, translating to about one in four.
“These findings suggest the need for increased awareness for post-COVID conditions so that improved post-COVID care and management of patients who survived COVID-19 can be developed and implemented,” said study author Lara Bull-Otterson, PhD, MPH, colead of data analytics at the Healthcare Data Advisory Unit of the CDC.
Pinpointing long COVID characteristics
Long COVID is difficult to identify, because many of its symptoms are similar to those of other conditions, so researchers are looking for better ways to characterize it to help improve both diagnosis and treatment.
Researchers on the Lancet study evaluated data from the National COVID Cohort Collaborative, N3C, a national NIH database that includes information from more than 8 million people. The team looked at the health records of 98,000 adult COVID patients and used that information, along with data from about nearly 600 long-COVID patients treated at three long-COVID clinics, to create three machine learning models for identifying long-COVID patients.
The models aimed to identify long-COVID patients in three groups: all patients, those hospitalized with COVID, and those with COVID but not hospitalized. The models were judged by the researchers to be accurate because those identified at risk for long COVID from the database were similar to those actually treated for long COVID at the clinics.
“Our algorithm is not intended to diagnose long COVID,” said lead author Emily Pfaff, PhD, research assistant professor of medicine at the University of North Carolina at Chapel Hill. “Rather, it is intended to identify patients in EHR data who ‘look like’ patients seen by physicians for long COVID.’’
Next, the researchers say, they will incorporate the new patterns they found with a diagnosis code for COVID and include it in the models to further test their accuracy. The models could also be used to help recruit patients for clinical trials, the researchers say.
Perspective and caveats
The figures of one in five and one in four found by the CDC researchers don’t surprise David Putrino, PT, PhD, director of rehabilitation innovation for Mount Sinai Health System in New York and director of its Abilities Research Center, which cares for long-COVID patients.
“Those numbers are high and it’s alarming,” he said. “But we’ve been sounding the alarm for quite some time, and we’ve been assuming that about one in five end up with long COVID.”
He does see a limitation to the CDC research – that some symptoms could have emerged later, and some in the control group could have had an undiagnosed COVID infection and gone on to develop long COVID.
As for machine learning, “this is something we need to approach with caution,” Dr. Putrino said. “There are a lot of variables we don’t understand about long COVID,’’ and that could result in spurious conclusions.
“Although I am supportive of this work going on, I am saying, ‘Scrutinize the tools with a grain of salt.’ Electronic records, Dr. Putrino points out, include information that the doctors enter, not what the patient says.
Dr. Pfaff responds: “It is entirely appropriate to approach both machine learning and EHR data with relevant caveats in mind. There are many clinical factors that are not recorded in the EHR, and the EHR is not representative of all persons with long COVID.” Those data can only reflect those who seek care for a condition, a natural limitation.
When it comes to algorithms, they are limited by data they have access to, such as the electronic health records in this research. However, the immense size and diversity in the data used “does allow us to make some assertations with much more confidence than if we were using data from a single or small number of health care systems,” she said.
A version of this article first appeared on Medscape.com.
As the number of people reporting persistent, and sometimes debilitating, symptoms from COVID-19 increases, researchers have struggled to pinpoint exactly how common so-called “long COVID” is, as well as how to clearly define exactly who has it or who is likely to get it.
Now, Centers for Disease Control and Prevention researchers have concluded that one in five adults aged 18 and older have at least one health condition that might be related to their previous COVID-19 illness; that number goes up to one in four among those 65 and older. Their data was published in the CDC’s Morbidity and Mortality Weekly Report.
The conditions associated with what’s been officially termed postacute sequelae of COVID-19, or PASC, include kidney failure, blood clots, other vascular issues, respiratory issues, heart problems, mental health or neurologic problems, and musculoskeletal conditions. But none of those conditions is unique to long COVID.
Another new study, published in The Lancet Digital Health, is trying to help better characterize what long COVID is, and what it isn’t.
that could help identify those likely to develop it.
CDC data
The CDC team came to its conclusions by evaluating the EHRs of more than 353,000 adults who were diagnosed with COVID-19 or got a positive test result, then comparing those records with 1.6 million patients who had a medical visit in the same month without a positive test result or a COVID-19 diagnosis.
They looked at data from March 2020 to November 2021, tagging 26 conditions often linked to post-COVID issues.
Overall, more than 38% of the COVID patients and 16% of those without COVID had at least one of these 26 conditions. They assessed the absolute risk difference between the patients and the non-COVID patients who developed one of the conditions, finding a 20.8–percentage point difference for those 18-64, yielding the one in five figure, and a 26.9–percentage point difference for those 65 and above, translating to about one in four.
“These findings suggest the need for increased awareness for post-COVID conditions so that improved post-COVID care and management of patients who survived COVID-19 can be developed and implemented,” said study author Lara Bull-Otterson, PhD, MPH, colead of data analytics at the Healthcare Data Advisory Unit of the CDC.
Pinpointing long COVID characteristics
Long COVID is difficult to identify, because many of its symptoms are similar to those of other conditions, so researchers are looking for better ways to characterize it to help improve both diagnosis and treatment.
Researchers on the Lancet study evaluated data from the National COVID Cohort Collaborative, N3C, a national NIH database that includes information from more than 8 million people. The team looked at the health records of 98,000 adult COVID patients and used that information, along with data from about nearly 600 long-COVID patients treated at three long-COVID clinics, to create three machine learning models for identifying long-COVID patients.
The models aimed to identify long-COVID patients in three groups: all patients, those hospitalized with COVID, and those with COVID but not hospitalized. The models were judged by the researchers to be accurate because those identified at risk for long COVID from the database were similar to those actually treated for long COVID at the clinics.
“Our algorithm is not intended to diagnose long COVID,” said lead author Emily Pfaff, PhD, research assistant professor of medicine at the University of North Carolina at Chapel Hill. “Rather, it is intended to identify patients in EHR data who ‘look like’ patients seen by physicians for long COVID.’’
Next, the researchers say, they will incorporate the new patterns they found with a diagnosis code for COVID and include it in the models to further test their accuracy. The models could also be used to help recruit patients for clinical trials, the researchers say.
Perspective and caveats
The figures of one in five and one in four found by the CDC researchers don’t surprise David Putrino, PT, PhD, director of rehabilitation innovation for Mount Sinai Health System in New York and director of its Abilities Research Center, which cares for long-COVID patients.
“Those numbers are high and it’s alarming,” he said. “But we’ve been sounding the alarm for quite some time, and we’ve been assuming that about one in five end up with long COVID.”
He does see a limitation to the CDC research – that some symptoms could have emerged later, and some in the control group could have had an undiagnosed COVID infection and gone on to develop long COVID.
As for machine learning, “this is something we need to approach with caution,” Dr. Putrino said. “There are a lot of variables we don’t understand about long COVID,’’ and that could result in spurious conclusions.
“Although I am supportive of this work going on, I am saying, ‘Scrutinize the tools with a grain of salt.’ Electronic records, Dr. Putrino points out, include information that the doctors enter, not what the patient says.
Dr. Pfaff responds: “It is entirely appropriate to approach both machine learning and EHR data with relevant caveats in mind. There are many clinical factors that are not recorded in the EHR, and the EHR is not representative of all persons with long COVID.” Those data can only reflect those who seek care for a condition, a natural limitation.
When it comes to algorithms, they are limited by data they have access to, such as the electronic health records in this research. However, the immense size and diversity in the data used “does allow us to make some assertations with much more confidence than if we were using data from a single or small number of health care systems,” she said.
A version of this article first appeared on Medscape.com.
Hospital medicine gains popularity among newly minted physicians
In a new study, published in Annals of Internal Medicine, researchers from ABIM reviewed certification data from 67,902 general internists, accounting for 80% of all general internists certified in the United States from 1990 to 2017.
The researchers also used data from Medicare fee-for-service claims from 2008-2018 to measure and categorize practice setting types. The claims were from patients aged 65 years or older with at least 20 evaluation and management visits each year. Practice settings were categorized as hospitalist, outpatient, or mixed.
“ABIM is always working to understand the real-life experience of physicians, and this project grew out of that sort of analysis,” lead author Bradley M. Gray, PhD, a health services researcher at ABIM in Philadelphia, said in an interview. “We wanted to better understand practice setting, because that relates to the kinds of questions that we ask on our certifying exams. When we did this, we noticed a trend toward hospital medicine.”
Overall, the percentages of general internists in hospitalist practice and outpatient-only practice increased during the study period, from 25% to 40% and from 23% to 38%, respectively. By contrast, the percentage of general internists in a mixed-practice setting decreased from 52% to 23%, a 56% decline. Most of the physicians who left the mixed practice setting switched to outpatient-only practices.
Among the internists certified in 2017, 71% practiced as hospitalists, compared with 8% practicing as outpatient-only physicians. Most physicians remained in their original choice of practice setting. For physicians certified in 1999 and 2012, 86% and 85%, respectively, of those who chose hospitalist medicine remained in the hospital setting 5 years later, as did 95% of outpatient physicians, but only 57% of mixed-practice physicians.
The shift to outpatient practice among senior physicians offset the potential decline in outpatient primary care resulting from the increased choice of hospitalist medicine by new internists, the researchers noted.
The study findings were limited by several factors, including the reliance on Medicare fee-for-service claims, the researchers noted.
“We were surprised by both the dramatic shift toward hospital medicine by new physicians and the shift to outpatient only (an extreme category) for more senior physicians,” Dr. Gray said in an interview.
The shift toward outpatient practice among older physicians may be driven by convenience, said Dr. Gray. “I suspect that it is more efficient to specialize in terms of practice setting. Only seeing patients in the outpatient setting means that you don’t have to travel to the hospital, which can be time consuming.
“Also, with fewer new physicians going into primary care, older physicians need to focus on outpatient visits. This could be problematic in the future as more senior physicians retire and are replaced by new physicians who focus on hospital care,” which could lead to more shortages in primary care physicians, he explained.
The trend toward hospital medicine as a career has been going on since before the pandemic, said Dr. Gray. “I don’t think the pandemic will ultimately impact this trend. That said, at least in the short run, there may have been a decreased demand for primary care, but that is just my speculation. As more data flow in we will be able to answer this question more directly.”
Next steps for research included digging deeper into the data to understand the nature of conditions facing hospitalists, Dr. Gray said.
Implications for primary care
“This study provides an updated snapshot of the popularity of hospital medicine,” said Bradley A. Sharpe, MD, of the division of hospital medicine at the University of California, San Francisco. “It is also important to conduct this study now as health systems think about the challenge of providing high-quality primary care with a rapidly decreasing number of internists choosing to practice outpatient medicine.” Dr. Sharpe was not involved in the study.
“The most surprising finding to me was not the increase in general internists focusing on hospital medicine, but the amount of the increase; it is remarkable that nearly three quarters of general internists are choosing to practice as hospitalists,” Dr. Sharpe noted.
“I think there are a number of key factors at play,” he said. “First, as hospital medicine as a field is now more than 25 years old, hospitals and health systems have evolved to create hospital medicine jobs that are interesting, engaging, rewarding (financially and otherwise), doable, and sustainable. Second, being an outpatient internist is incredibly challenging; multiple studies have shown that it is essentially impossible to complete the evidence-based preventive care for a panel of patients on top of everything else. We know burnout rates are often higher among primary care and family medicine providers. On top of that, the expansion of electronic health records and patient access has led to a massive increase in messages to providers; this has been shown to be associated with burnout.”
The potential impact of the pandemic on physicians’ choices and the trend toward hospital medicine is an interested question, Dr. Sharpe said. The current study showed only trends through 2017.
“To be honest, I think it is difficult to predict,” he said. “Hospitalists shouldered much of the burden of COVID care nationally and burnout rates are high. One could imagine the extra work (as well as concern for personal safety) could lead to fewer providers choosing hospital medicine.
“At the same time, the pandemic has driven many of us to reflect on life and our values and what is important and, through that lens, providers might choose hospital medicine as a more sustainable, do-able, rewarding, and enjoyable career choice,” Dr. Sharpe emphasized.
“Additional research could explore the drivers of this clear trend toward hospital medicine. Determining what is motivating this trend could help hospitals and health systems ensure they have the right workforce for the future and, in particular, how to create outpatient positions that are attractive and rewarding,” he said.
The study received no outside funding. The researchers and Dr. Sharpe disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a new study, published in Annals of Internal Medicine, researchers from ABIM reviewed certification data from 67,902 general internists, accounting for 80% of all general internists certified in the United States from 1990 to 2017.
The researchers also used data from Medicare fee-for-service claims from 2008-2018 to measure and categorize practice setting types. The claims were from patients aged 65 years or older with at least 20 evaluation and management visits each year. Practice settings were categorized as hospitalist, outpatient, or mixed.
“ABIM is always working to understand the real-life experience of physicians, and this project grew out of that sort of analysis,” lead author Bradley M. Gray, PhD, a health services researcher at ABIM in Philadelphia, said in an interview. “We wanted to better understand practice setting, because that relates to the kinds of questions that we ask on our certifying exams. When we did this, we noticed a trend toward hospital medicine.”
Overall, the percentages of general internists in hospitalist practice and outpatient-only practice increased during the study period, from 25% to 40% and from 23% to 38%, respectively. By contrast, the percentage of general internists in a mixed-practice setting decreased from 52% to 23%, a 56% decline. Most of the physicians who left the mixed practice setting switched to outpatient-only practices.
Among the internists certified in 2017, 71% practiced as hospitalists, compared with 8% practicing as outpatient-only physicians. Most physicians remained in their original choice of practice setting. For physicians certified in 1999 and 2012, 86% and 85%, respectively, of those who chose hospitalist medicine remained in the hospital setting 5 years later, as did 95% of outpatient physicians, but only 57% of mixed-practice physicians.
The shift to outpatient practice among senior physicians offset the potential decline in outpatient primary care resulting from the increased choice of hospitalist medicine by new internists, the researchers noted.
The study findings were limited by several factors, including the reliance on Medicare fee-for-service claims, the researchers noted.
“We were surprised by both the dramatic shift toward hospital medicine by new physicians and the shift to outpatient only (an extreme category) for more senior physicians,” Dr. Gray said in an interview.
The shift toward outpatient practice among older physicians may be driven by convenience, said Dr. Gray. “I suspect that it is more efficient to specialize in terms of practice setting. Only seeing patients in the outpatient setting means that you don’t have to travel to the hospital, which can be time consuming.
“Also, with fewer new physicians going into primary care, older physicians need to focus on outpatient visits. This could be problematic in the future as more senior physicians retire and are replaced by new physicians who focus on hospital care,” which could lead to more shortages in primary care physicians, he explained.
The trend toward hospital medicine as a career has been going on since before the pandemic, said Dr. Gray. “I don’t think the pandemic will ultimately impact this trend. That said, at least in the short run, there may have been a decreased demand for primary care, but that is just my speculation. As more data flow in we will be able to answer this question more directly.”
Next steps for research included digging deeper into the data to understand the nature of conditions facing hospitalists, Dr. Gray said.
Implications for primary care
“This study provides an updated snapshot of the popularity of hospital medicine,” said Bradley A. Sharpe, MD, of the division of hospital medicine at the University of California, San Francisco. “It is also important to conduct this study now as health systems think about the challenge of providing high-quality primary care with a rapidly decreasing number of internists choosing to practice outpatient medicine.” Dr. Sharpe was not involved in the study.
“The most surprising finding to me was not the increase in general internists focusing on hospital medicine, but the amount of the increase; it is remarkable that nearly three quarters of general internists are choosing to practice as hospitalists,” Dr. Sharpe noted.
“I think there are a number of key factors at play,” he said. “First, as hospital medicine as a field is now more than 25 years old, hospitals and health systems have evolved to create hospital medicine jobs that are interesting, engaging, rewarding (financially and otherwise), doable, and sustainable. Second, being an outpatient internist is incredibly challenging; multiple studies have shown that it is essentially impossible to complete the evidence-based preventive care for a panel of patients on top of everything else. We know burnout rates are often higher among primary care and family medicine providers. On top of that, the expansion of electronic health records and patient access has led to a massive increase in messages to providers; this has been shown to be associated with burnout.”
The potential impact of the pandemic on physicians’ choices and the trend toward hospital medicine is an interested question, Dr. Sharpe said. The current study showed only trends through 2017.
“To be honest, I think it is difficult to predict,” he said. “Hospitalists shouldered much of the burden of COVID care nationally and burnout rates are high. One could imagine the extra work (as well as concern for personal safety) could lead to fewer providers choosing hospital medicine.
“At the same time, the pandemic has driven many of us to reflect on life and our values and what is important and, through that lens, providers might choose hospital medicine as a more sustainable, do-able, rewarding, and enjoyable career choice,” Dr. Sharpe emphasized.
“Additional research could explore the drivers of this clear trend toward hospital medicine. Determining what is motivating this trend could help hospitals and health systems ensure they have the right workforce for the future and, in particular, how to create outpatient positions that are attractive and rewarding,” he said.
The study received no outside funding. The researchers and Dr. Sharpe disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a new study, published in Annals of Internal Medicine, researchers from ABIM reviewed certification data from 67,902 general internists, accounting for 80% of all general internists certified in the United States from 1990 to 2017.
The researchers also used data from Medicare fee-for-service claims from 2008-2018 to measure and categorize practice setting types. The claims were from patients aged 65 years or older with at least 20 evaluation and management visits each year. Practice settings were categorized as hospitalist, outpatient, or mixed.
“ABIM is always working to understand the real-life experience of physicians, and this project grew out of that sort of analysis,” lead author Bradley M. Gray, PhD, a health services researcher at ABIM in Philadelphia, said in an interview. “We wanted to better understand practice setting, because that relates to the kinds of questions that we ask on our certifying exams. When we did this, we noticed a trend toward hospital medicine.”
Overall, the percentages of general internists in hospitalist practice and outpatient-only practice increased during the study period, from 25% to 40% and from 23% to 38%, respectively. By contrast, the percentage of general internists in a mixed-practice setting decreased from 52% to 23%, a 56% decline. Most of the physicians who left the mixed practice setting switched to outpatient-only practices.
Among the internists certified in 2017, 71% practiced as hospitalists, compared with 8% practicing as outpatient-only physicians. Most physicians remained in their original choice of practice setting. For physicians certified in 1999 and 2012, 86% and 85%, respectively, of those who chose hospitalist medicine remained in the hospital setting 5 years later, as did 95% of outpatient physicians, but only 57% of mixed-practice physicians.
The shift to outpatient practice among senior physicians offset the potential decline in outpatient primary care resulting from the increased choice of hospitalist medicine by new internists, the researchers noted.
The study findings were limited by several factors, including the reliance on Medicare fee-for-service claims, the researchers noted.
“We were surprised by both the dramatic shift toward hospital medicine by new physicians and the shift to outpatient only (an extreme category) for more senior physicians,” Dr. Gray said in an interview.
The shift toward outpatient practice among older physicians may be driven by convenience, said Dr. Gray. “I suspect that it is more efficient to specialize in terms of practice setting. Only seeing patients in the outpatient setting means that you don’t have to travel to the hospital, which can be time consuming.
“Also, with fewer new physicians going into primary care, older physicians need to focus on outpatient visits. This could be problematic in the future as more senior physicians retire and are replaced by new physicians who focus on hospital care,” which could lead to more shortages in primary care physicians, he explained.
The trend toward hospital medicine as a career has been going on since before the pandemic, said Dr. Gray. “I don’t think the pandemic will ultimately impact this trend. That said, at least in the short run, there may have been a decreased demand for primary care, but that is just my speculation. As more data flow in we will be able to answer this question more directly.”
Next steps for research included digging deeper into the data to understand the nature of conditions facing hospitalists, Dr. Gray said.
Implications for primary care
“This study provides an updated snapshot of the popularity of hospital medicine,” said Bradley A. Sharpe, MD, of the division of hospital medicine at the University of California, San Francisco. “It is also important to conduct this study now as health systems think about the challenge of providing high-quality primary care with a rapidly decreasing number of internists choosing to practice outpatient medicine.” Dr. Sharpe was not involved in the study.
“The most surprising finding to me was not the increase in general internists focusing on hospital medicine, but the amount of the increase; it is remarkable that nearly three quarters of general internists are choosing to practice as hospitalists,” Dr. Sharpe noted.
“I think there are a number of key factors at play,” he said. “First, as hospital medicine as a field is now more than 25 years old, hospitals and health systems have evolved to create hospital medicine jobs that are interesting, engaging, rewarding (financially and otherwise), doable, and sustainable. Second, being an outpatient internist is incredibly challenging; multiple studies have shown that it is essentially impossible to complete the evidence-based preventive care for a panel of patients on top of everything else. We know burnout rates are often higher among primary care and family medicine providers. On top of that, the expansion of electronic health records and patient access has led to a massive increase in messages to providers; this has been shown to be associated with burnout.”
The potential impact of the pandemic on physicians’ choices and the trend toward hospital medicine is an interested question, Dr. Sharpe said. The current study showed only trends through 2017.
“To be honest, I think it is difficult to predict,” he said. “Hospitalists shouldered much of the burden of COVID care nationally and burnout rates are high. One could imagine the extra work (as well as concern for personal safety) could lead to fewer providers choosing hospital medicine.
“At the same time, the pandemic has driven many of us to reflect on life and our values and what is important and, through that lens, providers might choose hospital medicine as a more sustainable, do-able, rewarding, and enjoyable career choice,” Dr. Sharpe emphasized.
“Additional research could explore the drivers of this clear trend toward hospital medicine. Determining what is motivating this trend could help hospitals and health systems ensure they have the right workforce for the future and, in particular, how to create outpatient positions that are attractive and rewarding,” he said.
The study received no outside funding. The researchers and Dr. Sharpe disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
ADA prioritizes heart failure in patients with diabetes
All U.S. patients with diabetes should undergo annual biomarker testing to allow for early diagnosis of progressive but presymptomatic heart failure, and treatment with an agent from the sodium-glucose cotransporter 2 (SGLT2) inhibitor class should expand among such patients to include everyone with stage B heart failure (“pre–heart failure”) or more advanced stages.
That’s a recommendation from an American Diabetes Association consensus report published June 1 in Diabetes Care.
The report notes that until now, “implementation of available strategies to detect asymptomatic heart failure [in patients with diabetes] has been suboptimal.” The remedy for this is that, “among individuals with diabetes, measurement of a natriuretic peptide or high-sensitivity cardiac troponin is recommended on at least a yearly basis to identify the earliest heart failure stages and to implement strategies to prevent transition to symptomatic heart failure.”
Written by a 10-member panel, chaired by Rodica Pop-Busui, MD, PhD, and endorsed by the American College of Cardiology, the document also set threshold for levels of these biomarkers that are diagnostic for a more advanced stage (stage B) of heart failure in patients with diabetes but without heart failure symptoms:
- A B-type natriuretic peptide (BNP) level of ≥50 pg/mL;
- An N-terminal pro-BNP level of ≥125 pg/mL; or
- Any high sensitivity cardiac troponin value that’s above the usual upper reference limit set at >99th percentile.
‘Inexpensive’ biomarker testing
“Addition of relatively inexpensive biomarker testing as part of the standard of care may help to refine heart failure risk prediction in individuals with diabetes,” the report says.
“Substantial data indicate the ability of these biomarkers to identify those in stage A or B [heart failure] at highest risk of progressing to symptomatic heart failure or death,” and this identification is useful because “the risk in such individuals may be lowered through targeted intervention or multidisciplinary care.”
It is “impossible to understate the importance of early recognition of heart failure” in patients with heart failure, the authors declare. However, the report also cautions that, “using biomarkers to identify and in turn reduce risk for heart failure should always be done within the context of a thoughtful clinical evaluation, supported by all information available.”
The report, written during March 2021 – March 2022, cites the high prevalence and increasing incidence of heart failure in patients with diabetes as the rationale for the new recommendations.
For a person with diabetes who receives a heart failure diagnosis, the report details several management steps, starting with an evaluation for obstructive coronary artery disease, given the strong link between diabetes and atherosclerotic cardiovascular disease.
It highlights the importance of interventions that involve nutrition, smoking avoidance, minimized alcohol intake, exercise, weight loss, and relevant social determinants of health, but focuses in greater detail on a range of pharmacologic interventions. These include treatment of hypertension for people with early-stage heart failure with an ACE inhibitor or an angiotensin receptor blocker, a thiazide-type diuretic, and a mineralocorticoid receptor antagonist, such as spironolactone or the newer, nonsteroidal agent finerenone for patients with diabetic kidney disease.
Dr. Busui of the division of metabolism, endocrinology, and diabetes at the University of Michigan, Ann Arbor, and colleagues cite recent recommendations for using guidelines-directed medical therapy to treat patients with more advanced, symptomatic stages of heart failure, including heart failure with reduced or with preserved ejection fraction.
‘Prioritize’ the SGLT2-inhibitor class
The consensus report also summarizes the roles for agents in the various classes of antidiabetes drugs now available, with particular emphasis on the role for the SGLT2-inhibitor class.
SGLT2 inhibitors “are recommended for all individuals with [diabetes and] heart failure,” it says. “This consensus recommends prioritizing the use of SGLT2 inhibitors in individuals with stage B heart failure, and that SGLT2 inhibitors be an expected element of care in all individuals with diabetes and symptomatic heart failure.”
Other agents for glycemic control that receive endorsement from the report are those in the glucagonlike peptide 1 receptor agonist class. “Despite the lack of conclusive evidence of direct heart failure risk reduction” with this class, it gets a “should be considered” designation, based on its positive effects on weight loss, blood pressure, and atherothrombotic disease.
Similar acknowledgment of potential benefit in a “should be considered” role goes to metformin. But the report turned a thumb down for both the class of dipeptidyl peptidase 4 inhibitors and the thiazolidinedione class, and said that agents from the insulin and sulfonylurea classes should be used “judiciously.”
The report did not identify any commercial funding. Several of the writing committee members listed personal commercial disclosures.
All U.S. patients with diabetes should undergo annual biomarker testing to allow for early diagnosis of progressive but presymptomatic heart failure, and treatment with an agent from the sodium-glucose cotransporter 2 (SGLT2) inhibitor class should expand among such patients to include everyone with stage B heart failure (“pre–heart failure”) or more advanced stages.
That’s a recommendation from an American Diabetes Association consensus report published June 1 in Diabetes Care.
The report notes that until now, “implementation of available strategies to detect asymptomatic heart failure [in patients with diabetes] has been suboptimal.” The remedy for this is that, “among individuals with diabetes, measurement of a natriuretic peptide or high-sensitivity cardiac troponin is recommended on at least a yearly basis to identify the earliest heart failure stages and to implement strategies to prevent transition to symptomatic heart failure.”
Written by a 10-member panel, chaired by Rodica Pop-Busui, MD, PhD, and endorsed by the American College of Cardiology, the document also set threshold for levels of these biomarkers that are diagnostic for a more advanced stage (stage B) of heart failure in patients with diabetes but without heart failure symptoms:
- A B-type natriuretic peptide (BNP) level of ≥50 pg/mL;
- An N-terminal pro-BNP level of ≥125 pg/mL; or
- Any high sensitivity cardiac troponin value that’s above the usual upper reference limit set at >99th percentile.
‘Inexpensive’ biomarker testing
“Addition of relatively inexpensive biomarker testing as part of the standard of care may help to refine heart failure risk prediction in individuals with diabetes,” the report says.
“Substantial data indicate the ability of these biomarkers to identify those in stage A or B [heart failure] at highest risk of progressing to symptomatic heart failure or death,” and this identification is useful because “the risk in such individuals may be lowered through targeted intervention or multidisciplinary care.”
It is “impossible to understate the importance of early recognition of heart failure” in patients with heart failure, the authors declare. However, the report also cautions that, “using biomarkers to identify and in turn reduce risk for heart failure should always be done within the context of a thoughtful clinical evaluation, supported by all information available.”
The report, written during March 2021 – March 2022, cites the high prevalence and increasing incidence of heart failure in patients with diabetes as the rationale for the new recommendations.
For a person with diabetes who receives a heart failure diagnosis, the report details several management steps, starting with an evaluation for obstructive coronary artery disease, given the strong link between diabetes and atherosclerotic cardiovascular disease.
It highlights the importance of interventions that involve nutrition, smoking avoidance, minimized alcohol intake, exercise, weight loss, and relevant social determinants of health, but focuses in greater detail on a range of pharmacologic interventions. These include treatment of hypertension for people with early-stage heart failure with an ACE inhibitor or an angiotensin receptor blocker, a thiazide-type diuretic, and a mineralocorticoid receptor antagonist, such as spironolactone or the newer, nonsteroidal agent finerenone for patients with diabetic kidney disease.
Dr. Busui of the division of metabolism, endocrinology, and diabetes at the University of Michigan, Ann Arbor, and colleagues cite recent recommendations for using guidelines-directed medical therapy to treat patients with more advanced, symptomatic stages of heart failure, including heart failure with reduced or with preserved ejection fraction.
‘Prioritize’ the SGLT2-inhibitor class
The consensus report also summarizes the roles for agents in the various classes of antidiabetes drugs now available, with particular emphasis on the role for the SGLT2-inhibitor class.
SGLT2 inhibitors “are recommended for all individuals with [diabetes and] heart failure,” it says. “This consensus recommends prioritizing the use of SGLT2 inhibitors in individuals with stage B heart failure, and that SGLT2 inhibitors be an expected element of care in all individuals with diabetes and symptomatic heart failure.”
Other agents for glycemic control that receive endorsement from the report are those in the glucagonlike peptide 1 receptor agonist class. “Despite the lack of conclusive evidence of direct heart failure risk reduction” with this class, it gets a “should be considered” designation, based on its positive effects on weight loss, blood pressure, and atherothrombotic disease.
Similar acknowledgment of potential benefit in a “should be considered” role goes to metformin. But the report turned a thumb down for both the class of dipeptidyl peptidase 4 inhibitors and the thiazolidinedione class, and said that agents from the insulin and sulfonylurea classes should be used “judiciously.”
The report did not identify any commercial funding. Several of the writing committee members listed personal commercial disclosures.
All U.S. patients with diabetes should undergo annual biomarker testing to allow for early diagnosis of progressive but presymptomatic heart failure, and treatment with an agent from the sodium-glucose cotransporter 2 (SGLT2) inhibitor class should expand among such patients to include everyone with stage B heart failure (“pre–heart failure”) or more advanced stages.
That’s a recommendation from an American Diabetes Association consensus report published June 1 in Diabetes Care.
The report notes that until now, “implementation of available strategies to detect asymptomatic heart failure [in patients with diabetes] has been suboptimal.” The remedy for this is that, “among individuals with diabetes, measurement of a natriuretic peptide or high-sensitivity cardiac troponin is recommended on at least a yearly basis to identify the earliest heart failure stages and to implement strategies to prevent transition to symptomatic heart failure.”
Written by a 10-member panel, chaired by Rodica Pop-Busui, MD, PhD, and endorsed by the American College of Cardiology, the document also set threshold for levels of these biomarkers that are diagnostic for a more advanced stage (stage B) of heart failure in patients with diabetes but without heart failure symptoms:
- A B-type natriuretic peptide (BNP) level of ≥50 pg/mL;
- An N-terminal pro-BNP level of ≥125 pg/mL; or
- Any high sensitivity cardiac troponin value that’s above the usual upper reference limit set at >99th percentile.
‘Inexpensive’ biomarker testing
“Addition of relatively inexpensive biomarker testing as part of the standard of care may help to refine heart failure risk prediction in individuals with diabetes,” the report says.
“Substantial data indicate the ability of these biomarkers to identify those in stage A or B [heart failure] at highest risk of progressing to symptomatic heart failure or death,” and this identification is useful because “the risk in such individuals may be lowered through targeted intervention or multidisciplinary care.”
It is “impossible to understate the importance of early recognition of heart failure” in patients with heart failure, the authors declare. However, the report also cautions that, “using biomarkers to identify and in turn reduce risk for heart failure should always be done within the context of a thoughtful clinical evaluation, supported by all information available.”
The report, written during March 2021 – March 2022, cites the high prevalence and increasing incidence of heart failure in patients with diabetes as the rationale for the new recommendations.
For a person with diabetes who receives a heart failure diagnosis, the report details several management steps, starting with an evaluation for obstructive coronary artery disease, given the strong link between diabetes and atherosclerotic cardiovascular disease.
It highlights the importance of interventions that involve nutrition, smoking avoidance, minimized alcohol intake, exercise, weight loss, and relevant social determinants of health, but focuses in greater detail on a range of pharmacologic interventions. These include treatment of hypertension for people with early-stage heart failure with an ACE inhibitor or an angiotensin receptor blocker, a thiazide-type diuretic, and a mineralocorticoid receptor antagonist, such as spironolactone or the newer, nonsteroidal agent finerenone for patients with diabetic kidney disease.
Dr. Busui of the division of metabolism, endocrinology, and diabetes at the University of Michigan, Ann Arbor, and colleagues cite recent recommendations for using guidelines-directed medical therapy to treat patients with more advanced, symptomatic stages of heart failure, including heart failure with reduced or with preserved ejection fraction.
‘Prioritize’ the SGLT2-inhibitor class
The consensus report also summarizes the roles for agents in the various classes of antidiabetes drugs now available, with particular emphasis on the role for the SGLT2-inhibitor class.
SGLT2 inhibitors “are recommended for all individuals with [diabetes and] heart failure,” it says. “This consensus recommends prioritizing the use of SGLT2 inhibitors in individuals with stage B heart failure, and that SGLT2 inhibitors be an expected element of care in all individuals with diabetes and symptomatic heart failure.”
Other agents for glycemic control that receive endorsement from the report are those in the glucagonlike peptide 1 receptor agonist class. “Despite the lack of conclusive evidence of direct heart failure risk reduction” with this class, it gets a “should be considered” designation, based on its positive effects on weight loss, blood pressure, and atherothrombotic disease.
Similar acknowledgment of potential benefit in a “should be considered” role goes to metformin. But the report turned a thumb down for both the class of dipeptidyl peptidase 4 inhibitors and the thiazolidinedione class, and said that agents from the insulin and sulfonylurea classes should be used “judiciously.”
The report did not identify any commercial funding. Several of the writing committee members listed personal commercial disclosures.
FROM DIABETES CARE
The latest on COVID-19 and the heart in children
The 2022 Pediatric Academic Societies meeting included an excellent session on the acute and delayed effects of COVID-19 on children’s hearts. Data on the risk for cardiac injury during acute COVID-19, return-to-play guidelines after COVID-19–related heart injury, and post–vaccine-associated myocarditis were reviewed.
COVID-induced cardiac injury
The risk for COVID-induced cardiac injury is directly associated with age. Recent Centers for Disease Control and Prevention data revealed a “myocarditis or pericarditis” rate in the range of 12-17 cases per 100,000 SARS-CoV-2 infections among male children aged 5-11 years (lower rates for females); the rate jumps to 50-65 cases per 100,000 infections among male children aged 12-17 years. So cardiac injury caused by acute COVID-19 appears rare, but the risk is clearly associated with male sex and adolescent age.
Return to play after COVID-19
Clinicians may be pressed by patients and parents for advice on return to play after illness with COVID-19. In July 2020, the American College of Cardiology published an algorithm that has been adjusted over time, most recently in 2022 by the American Academy of Pediatrics. These algorithms stratify recommendations by degree of illness. One rule of thumb: Patients with severe COVID-19 (ICU care or multisystem inflammatory syndrome in children [MIS-C]) have only one box on the algorithm, and that is to rest for 3-6 months and only return to usual activity after cardiac clearance. Moderate disease (defined as ≥ 4 days of fever > 100.4 °F; ≥ 1 week of myalgia, chills, lethargy, or any non-ICU hospital stay; and no evidence of MIS-C) require undergoing an ECG to look for cardiac dysfunction, followed by at least 10 days of rest if the ECG is negative or referral for cardiac evaluation if either ECG or exam by a pediatric cardiologist is abnormal.
Clinicians can perhaps be more permissible with patients who are younger or who have had less severe disease. For example, if a patient aged younger than 12 years is asymptomatic with routine activity at the time of evaluation, an ECG is not indicated. For patients aged 12-15 years who are asymptomatic at the time of evaluation but participate in a high-intensity sport, clinicians might consider obtaining an ECG. As few as 3 days of rest might be enough for select patients who are asymptomatic at presentation. For other patients, clinicians should work with parents to introduce activity gradually and make it clear to parents that any activity intolerance requires quick reevaluation. On existing athlete registries, no deaths that are attributable to post–COVID-19 cardiac effects have been confirmed in children; however, all data presented during the session were from prior to the Omicron variant surge in early 2022, so more information may be forthcoming.
Considerations for MIS-C
Among children experiencing MIS-C, 35% had ECG changes, 40% exhibited left ventricular systolic or diastolic dysfunction, and 30% had mitral regurgitation, meaning that a large percentage of patients with MIS-C show some degree of cardiac dysfunction. Unfortunately, we are still in the data-gathering phase for long-term outcomes. Functional parameters tend to improve within a week, and most patients will return to normal cardiac function by 3-4 months.
Return to play after MIS-C is quite different from that for acute COVID-19. Patients with MIS-C should be treated much like other patients with myocarditis with an expected return to play in 3-6 months and only after cardiac follow-up. Another good-to-remember recommendation is to delay COVID-19 vaccination for at least 90 days after an episode of MIS-C.
Vaccine-related myocarditis
Once again, older age appears to be a risk factor because most patients with postvaccine myocarditis have been in their mid-teens to early 20s, with events more likely after the second vaccine dose and also more likely in male children (4:1 ratio to female children). No deaths have occurred from postvaccination myocarditis in patients younger than 30 years. Still, many individuals have exhibited residual MRI enhancement in the cardiac tissue for some time after experiencing postvaccination myocarditis; it’s currently unclear whether that has clinical implications. By comparison, CDC data demonstrates convincingly that the risk for cardiac effects is much greater after acute COVID-19 than after COVID-19 vaccination, with risk ratios often higher than 20, depending on age and condition (for example, myocarditis vs. pericarditis). Data are still insufficient to determine whether clinicians should recommend or avoid COVID-19 vaccination in children with congenital heart disease.
In summary, administering COVID-19 vaccines requires a great deal of shared decision-making with parents, and the clinician’s role is to educate parents about all potential risks related to both the vaccine and COVID-19 illness. Research has consistently shown that acute COVID-19 myocarditis and myocarditis associated with MIS-C are much more likely to occur in unvaccinated youth and more likely than postvaccination myocarditis, regardless of age.
William T. Basco, Jr., MD, MS, is a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics. He is an active health services researcher and has published more than 60 manuscripts in the peer-reviewed literature.
A version of this article first appeared on Medscape.com.
The 2022 Pediatric Academic Societies meeting included an excellent session on the acute and delayed effects of COVID-19 on children’s hearts. Data on the risk for cardiac injury during acute COVID-19, return-to-play guidelines after COVID-19–related heart injury, and post–vaccine-associated myocarditis were reviewed.
COVID-induced cardiac injury
The risk for COVID-induced cardiac injury is directly associated with age. Recent Centers for Disease Control and Prevention data revealed a “myocarditis or pericarditis” rate in the range of 12-17 cases per 100,000 SARS-CoV-2 infections among male children aged 5-11 years (lower rates for females); the rate jumps to 50-65 cases per 100,000 infections among male children aged 12-17 years. So cardiac injury caused by acute COVID-19 appears rare, but the risk is clearly associated with male sex and adolescent age.
Return to play after COVID-19
Clinicians may be pressed by patients and parents for advice on return to play after illness with COVID-19. In July 2020, the American College of Cardiology published an algorithm that has been adjusted over time, most recently in 2022 by the American Academy of Pediatrics. These algorithms stratify recommendations by degree of illness. One rule of thumb: Patients with severe COVID-19 (ICU care or multisystem inflammatory syndrome in children [MIS-C]) have only one box on the algorithm, and that is to rest for 3-6 months and only return to usual activity after cardiac clearance. Moderate disease (defined as ≥ 4 days of fever > 100.4 °F; ≥ 1 week of myalgia, chills, lethargy, or any non-ICU hospital stay; and no evidence of MIS-C) require undergoing an ECG to look for cardiac dysfunction, followed by at least 10 days of rest if the ECG is negative or referral for cardiac evaluation if either ECG or exam by a pediatric cardiologist is abnormal.
Clinicians can perhaps be more permissible with patients who are younger or who have had less severe disease. For example, if a patient aged younger than 12 years is asymptomatic with routine activity at the time of evaluation, an ECG is not indicated. For patients aged 12-15 years who are asymptomatic at the time of evaluation but participate in a high-intensity sport, clinicians might consider obtaining an ECG. As few as 3 days of rest might be enough for select patients who are asymptomatic at presentation. For other patients, clinicians should work with parents to introduce activity gradually and make it clear to parents that any activity intolerance requires quick reevaluation. On existing athlete registries, no deaths that are attributable to post–COVID-19 cardiac effects have been confirmed in children; however, all data presented during the session were from prior to the Omicron variant surge in early 2022, so more information may be forthcoming.
Considerations for MIS-C
Among children experiencing MIS-C, 35% had ECG changes, 40% exhibited left ventricular systolic or diastolic dysfunction, and 30% had mitral regurgitation, meaning that a large percentage of patients with MIS-C show some degree of cardiac dysfunction. Unfortunately, we are still in the data-gathering phase for long-term outcomes. Functional parameters tend to improve within a week, and most patients will return to normal cardiac function by 3-4 months.
Return to play after MIS-C is quite different from that for acute COVID-19. Patients with MIS-C should be treated much like other patients with myocarditis with an expected return to play in 3-6 months and only after cardiac follow-up. Another good-to-remember recommendation is to delay COVID-19 vaccination for at least 90 days after an episode of MIS-C.
Vaccine-related myocarditis
Once again, older age appears to be a risk factor because most patients with postvaccine myocarditis have been in their mid-teens to early 20s, with events more likely after the second vaccine dose and also more likely in male children (4:1 ratio to female children). No deaths have occurred from postvaccination myocarditis in patients younger than 30 years. Still, many individuals have exhibited residual MRI enhancement in the cardiac tissue for some time after experiencing postvaccination myocarditis; it’s currently unclear whether that has clinical implications. By comparison, CDC data demonstrates convincingly that the risk for cardiac effects is much greater after acute COVID-19 than after COVID-19 vaccination, with risk ratios often higher than 20, depending on age and condition (for example, myocarditis vs. pericarditis). Data are still insufficient to determine whether clinicians should recommend or avoid COVID-19 vaccination in children with congenital heart disease.
In summary, administering COVID-19 vaccines requires a great deal of shared decision-making with parents, and the clinician’s role is to educate parents about all potential risks related to both the vaccine and COVID-19 illness. Research has consistently shown that acute COVID-19 myocarditis and myocarditis associated with MIS-C are much more likely to occur in unvaccinated youth and more likely than postvaccination myocarditis, regardless of age.
William T. Basco, Jr., MD, MS, is a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics. He is an active health services researcher and has published more than 60 manuscripts in the peer-reviewed literature.
A version of this article first appeared on Medscape.com.
The 2022 Pediatric Academic Societies meeting included an excellent session on the acute and delayed effects of COVID-19 on children’s hearts. Data on the risk for cardiac injury during acute COVID-19, return-to-play guidelines after COVID-19–related heart injury, and post–vaccine-associated myocarditis were reviewed.
COVID-induced cardiac injury
The risk for COVID-induced cardiac injury is directly associated with age. Recent Centers for Disease Control and Prevention data revealed a “myocarditis or pericarditis” rate in the range of 12-17 cases per 100,000 SARS-CoV-2 infections among male children aged 5-11 years (lower rates for females); the rate jumps to 50-65 cases per 100,000 infections among male children aged 12-17 years. So cardiac injury caused by acute COVID-19 appears rare, but the risk is clearly associated with male sex and adolescent age.
Return to play after COVID-19
Clinicians may be pressed by patients and parents for advice on return to play after illness with COVID-19. In July 2020, the American College of Cardiology published an algorithm that has been adjusted over time, most recently in 2022 by the American Academy of Pediatrics. These algorithms stratify recommendations by degree of illness. One rule of thumb: Patients with severe COVID-19 (ICU care or multisystem inflammatory syndrome in children [MIS-C]) have only one box on the algorithm, and that is to rest for 3-6 months and only return to usual activity after cardiac clearance. Moderate disease (defined as ≥ 4 days of fever > 100.4 °F; ≥ 1 week of myalgia, chills, lethargy, or any non-ICU hospital stay; and no evidence of MIS-C) require undergoing an ECG to look for cardiac dysfunction, followed by at least 10 days of rest if the ECG is negative or referral for cardiac evaluation if either ECG or exam by a pediatric cardiologist is abnormal.
Clinicians can perhaps be more permissible with patients who are younger or who have had less severe disease. For example, if a patient aged younger than 12 years is asymptomatic with routine activity at the time of evaluation, an ECG is not indicated. For patients aged 12-15 years who are asymptomatic at the time of evaluation but participate in a high-intensity sport, clinicians might consider obtaining an ECG. As few as 3 days of rest might be enough for select patients who are asymptomatic at presentation. For other patients, clinicians should work with parents to introduce activity gradually and make it clear to parents that any activity intolerance requires quick reevaluation. On existing athlete registries, no deaths that are attributable to post–COVID-19 cardiac effects have been confirmed in children; however, all data presented during the session were from prior to the Omicron variant surge in early 2022, so more information may be forthcoming.
Considerations for MIS-C
Among children experiencing MIS-C, 35% had ECG changes, 40% exhibited left ventricular systolic or diastolic dysfunction, and 30% had mitral regurgitation, meaning that a large percentage of patients with MIS-C show some degree of cardiac dysfunction. Unfortunately, we are still in the data-gathering phase for long-term outcomes. Functional parameters tend to improve within a week, and most patients will return to normal cardiac function by 3-4 months.
Return to play after MIS-C is quite different from that for acute COVID-19. Patients with MIS-C should be treated much like other patients with myocarditis with an expected return to play in 3-6 months and only after cardiac follow-up. Another good-to-remember recommendation is to delay COVID-19 vaccination for at least 90 days after an episode of MIS-C.
Vaccine-related myocarditis
Once again, older age appears to be a risk factor because most patients with postvaccine myocarditis have been in their mid-teens to early 20s, with events more likely after the second vaccine dose and also more likely in male children (4:1 ratio to female children). No deaths have occurred from postvaccination myocarditis in patients younger than 30 years. Still, many individuals have exhibited residual MRI enhancement in the cardiac tissue for some time after experiencing postvaccination myocarditis; it’s currently unclear whether that has clinical implications. By comparison, CDC data demonstrates convincingly that the risk for cardiac effects is much greater after acute COVID-19 than after COVID-19 vaccination, with risk ratios often higher than 20, depending on age and condition (for example, myocarditis vs. pericarditis). Data are still insufficient to determine whether clinicians should recommend or avoid COVID-19 vaccination in children with congenital heart disease.
In summary, administering COVID-19 vaccines requires a great deal of shared decision-making with parents, and the clinician’s role is to educate parents about all potential risks related to both the vaccine and COVID-19 illness. Research has consistently shown that acute COVID-19 myocarditis and myocarditis associated with MIS-C are much more likely to occur in unvaccinated youth and more likely than postvaccination myocarditis, regardless of age.
William T. Basco, Jr., MD, MS, is a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics. He is an active health services researcher and has published more than 60 manuscripts in the peer-reviewed literature.
A version of this article first appeared on Medscape.com.
LDL lowering to specific targets may offset risk from high Lp(a)
MILAN – The increased risk for atherosclerotic cardiovascular disease events caused by elevated lipoprotein(a) levels can potentially be precisely offset by lowering LDL cholesterol to specific levels, suggests a novel study that underscores the importance or early intervention.
The results, derived from an analysis of data on Lp(a) and LDL cholesterol levels and associated genetic risk scores in almost 500,000 individuals from the United Kingdom, have been used to develop a series of age-related targets for lowering LDL cholesterol levels to counter the risk associated with lifetime Lp(a) exposure.
Measuring Lp(a) levels can “substantially refine individual estimates of absolute risk of atherosclerotic cardiovascular disease,” said study presenter Brian A. Ference, MD, Centre for Naturally Randomized Trials, University of Cambridge (England).
This can “directly inform treatment decisions about the intensity of LDL lowering or other risk-factor modification needed to overcome the increased risk caused by Lp(a).”
Dr. Ference said this will allow clinicians to personalize the prevention of atherosclerotic cardiovascular disease and identify people “who may benefit from potent Lp(a)-lowering therapies when they become available.”
The research was presented at the European Atherosclerosis Society (EAS) 2022 congress on May 24.
In addition to producing a tabular version of the intensification of LDL-cholesterol reduction needed to overcome the increased cardiovascular risk at different levels of Lp(a), stratified by age, Dr. Ference is working with the EAS to develop an app to further deliver on that personalized prevention.
It will display an individual’s lifetime risk for myocardial infarction or stroke, with and without the inclusion of Lp(a) levels, and determine not only the percentage of increased risk caused by Lp(a), but also the amount by which LDL cholesterol needs to be lowered to overcome that risk.
“The whole rationale for this study was to say, how can we give practical advice on how to use Lp(a) to inform clinical decisions about how to individualize personal risk reduction,” Dr. Ference told this news organization.
“What the app will do is make it very easy for clinicians to, first, understand how much Lp(a) increases risk, but specifically how they can use that information to directly inform their treatment decisions.”
In addition, Dr. Ference said that it will “show patients why it’s important for them” to intensify LDL lowering to overcome their particular level of Lp(a).
Other key takeaways from the results is the importance of intervention as early as possible to minimize the impact of lifetime exposure to increased Lp(a), and that the reduction in LDL cholesterol required to achieve that remains relatively modest.
For Dr. Ference, this means ideally beginning comprehensive health checks at 30 years of age and starting lipid-lowering interventions immediately for those at risk.
“The good thing about LDL and other causes of atherosclerotic cardiovascular disease is it doesn’t really matter how you lower it,” he said, noting that it could be with diet, lifestyle interventions, or medication.
Handy tool
The new app could be a “handy tool to counsel patients,” Florian Kronenberg, MD, Institute of Genetic Epidemiology, Medical University of Innsbruck, Austria, told this news organization.
“We can say, look, you have high Lp(a),” he said. “This is coming from nature, from your genetics, but here we have a point where we can act on your high risk by lowering LDL further. This is important to explain to the patient,” said Dr. Kronenberg, who was not involved in the study.
He emphasized that it is crucial to get across the idea of an individual’s global risk, with not just Lp(a) or cholesterol levels influencing their likelihood of cardiovascular events, but also their age, blood pressure, smoking status, and underlying genetic risk.
Dr. Kronenberg said the current data will be helpful in explaining to clinicians why they should lower LDL-cholesterol levels when a patients had high Lp(a), again centered on the idea of lowering their global risk.
During his presentation, Dr. Ference noted that an increase in Lp(a) levels is associated with a log-linear increase in atherosclerotic cardiovascular disease that is proportional to the absolute, rather than relative, magnitude of Lp(a) increase.
“Unfortunately, unlike other proteins,” he continued, diet and exercise do not affect levels, and there are currently no effective therapies to lower the risks associated with increased Lp(a) concentrations.
“For that reason,” he said, the 2019 ESC/EAS guidelines for the management of dyslipidemias, on which Dr. Ference was a coauthor, “recommend that we intensify life risk-factor modification in persons with elevated risks.”
However, he added, “this guidance is not specific enough to be useful, and that has created a great deal of inertia among clinicians,” with some concluding that they don’t need to measure Lp(a) “because there’s nothing they can do for it.”
Until the development of novel therapies that directly target Lp(a), the authors sought to quantify the amount of LDL lowering needed to “overcome the increased risk caused by Lp(a),” he said.
They studied data on 455,765 individuals from the UK Biobank who did not have a history of cardiovascular events, diabetes, or any cancer before the age of 30. They also had LDL cholesterol levels below 5 mmol/L at the time of enrollment to exclude people with presumed familial hypercholesterolemia.
The researchers used an Lp(a) genetic risk score based on the variants rs10455872 and rs3798220 and an LDL instrumental variable genetic score comprised of 100 variants to randomly categorize individuals with average Lp(a) levels, higher Lp(a) levels, or higher Lp(a) and lower LDL-cholesterol levels.
The data showed that, with elevated absolute levels of measured Lp(a) and with elevated genetic risk scores, there was a progressive increase in the lifetime risk for major coronary events.
When looking at the combination of both increased Lp(a) levels and lower LDL-cholesterol levels, they found that the increase in risk for major coronary events at Lp(a) of 123 nmol/L could be offset by a reduction in LDL-cholesterol levels of 19.5 mg/dL.
For people with an Lp(a) level of 251 nmol/L, the increase in risk for major coronary events was offset by a reduction in LDL-cholesterol levels of 36.1 mg/dL.
Furthermore, the researchers found that the magnitude of intensification of LDL-cholesterol lowering needed to overcome the risk caused by elevated Lp(a) levels varied by age.
For example, in individuals with an Lp(a) level of 220 nmol/L, the reduction in LDL-cholesterol levels needed to offset the risk for major coronary events was calculated to be 0.8 mmol/L if lipid-lowering was started at 30 years of age, rising to 0.9 mmol/L if started at 40 years, 1.2 mmol/L if started at 50 years, and 1.5 mmol/L if started at 60 years.
This, Dr. Ference said, suggests that “diet and lifestyle modification is unlikely to be an effective strategy if started later.”
No funding was declared. Dr. Ference declared relationships with Amgen, Novartis, Merck, Esperion Therapeutics, Pfizer, Regeneron, Sanofi, AstraZeneca, Eli Lilly, Novo Nordisk, The Medicines Company, Mylan, Daiichi Sankyo, Viatris, Ionis Pharmaceuticals, dalCOR, CiVi Pharma, and KrKa Pharmaceuticals. Dr. Kronenberg declared relationships with Amgen, Novartis, and Kaneka.
A version of this article first appeared on Medscape.com.
MILAN – The increased risk for atherosclerotic cardiovascular disease events caused by elevated lipoprotein(a) levels can potentially be precisely offset by lowering LDL cholesterol to specific levels, suggests a novel study that underscores the importance or early intervention.
The results, derived from an analysis of data on Lp(a) and LDL cholesterol levels and associated genetic risk scores in almost 500,000 individuals from the United Kingdom, have been used to develop a series of age-related targets for lowering LDL cholesterol levels to counter the risk associated with lifetime Lp(a) exposure.
Measuring Lp(a) levels can “substantially refine individual estimates of absolute risk of atherosclerotic cardiovascular disease,” said study presenter Brian A. Ference, MD, Centre for Naturally Randomized Trials, University of Cambridge (England).
This can “directly inform treatment decisions about the intensity of LDL lowering or other risk-factor modification needed to overcome the increased risk caused by Lp(a).”
Dr. Ference said this will allow clinicians to personalize the prevention of atherosclerotic cardiovascular disease and identify people “who may benefit from potent Lp(a)-lowering therapies when they become available.”
The research was presented at the European Atherosclerosis Society (EAS) 2022 congress on May 24.
In addition to producing a tabular version of the intensification of LDL-cholesterol reduction needed to overcome the increased cardiovascular risk at different levels of Lp(a), stratified by age, Dr. Ference is working with the EAS to develop an app to further deliver on that personalized prevention.
It will display an individual’s lifetime risk for myocardial infarction or stroke, with and without the inclusion of Lp(a) levels, and determine not only the percentage of increased risk caused by Lp(a), but also the amount by which LDL cholesterol needs to be lowered to overcome that risk.
“The whole rationale for this study was to say, how can we give practical advice on how to use Lp(a) to inform clinical decisions about how to individualize personal risk reduction,” Dr. Ference told this news organization.
“What the app will do is make it very easy for clinicians to, first, understand how much Lp(a) increases risk, but specifically how they can use that information to directly inform their treatment decisions.”
In addition, Dr. Ference said that it will “show patients why it’s important for them” to intensify LDL lowering to overcome their particular level of Lp(a).
Other key takeaways from the results is the importance of intervention as early as possible to minimize the impact of lifetime exposure to increased Lp(a), and that the reduction in LDL cholesterol required to achieve that remains relatively modest.
For Dr. Ference, this means ideally beginning comprehensive health checks at 30 years of age and starting lipid-lowering interventions immediately for those at risk.
“The good thing about LDL and other causes of atherosclerotic cardiovascular disease is it doesn’t really matter how you lower it,” he said, noting that it could be with diet, lifestyle interventions, or medication.
Handy tool
The new app could be a “handy tool to counsel patients,” Florian Kronenberg, MD, Institute of Genetic Epidemiology, Medical University of Innsbruck, Austria, told this news organization.
“We can say, look, you have high Lp(a),” he said. “This is coming from nature, from your genetics, but here we have a point where we can act on your high risk by lowering LDL further. This is important to explain to the patient,” said Dr. Kronenberg, who was not involved in the study.
He emphasized that it is crucial to get across the idea of an individual’s global risk, with not just Lp(a) or cholesterol levels influencing their likelihood of cardiovascular events, but also their age, blood pressure, smoking status, and underlying genetic risk.
Dr. Kronenberg said the current data will be helpful in explaining to clinicians why they should lower LDL-cholesterol levels when a patients had high Lp(a), again centered on the idea of lowering their global risk.
During his presentation, Dr. Ference noted that an increase in Lp(a) levels is associated with a log-linear increase in atherosclerotic cardiovascular disease that is proportional to the absolute, rather than relative, magnitude of Lp(a) increase.
“Unfortunately, unlike other proteins,” he continued, diet and exercise do not affect levels, and there are currently no effective therapies to lower the risks associated with increased Lp(a) concentrations.
“For that reason,” he said, the 2019 ESC/EAS guidelines for the management of dyslipidemias, on which Dr. Ference was a coauthor, “recommend that we intensify life risk-factor modification in persons with elevated risks.”
However, he added, “this guidance is not specific enough to be useful, and that has created a great deal of inertia among clinicians,” with some concluding that they don’t need to measure Lp(a) “because there’s nothing they can do for it.”
Until the development of novel therapies that directly target Lp(a), the authors sought to quantify the amount of LDL lowering needed to “overcome the increased risk caused by Lp(a),” he said.
They studied data on 455,765 individuals from the UK Biobank who did not have a history of cardiovascular events, diabetes, or any cancer before the age of 30. They also had LDL cholesterol levels below 5 mmol/L at the time of enrollment to exclude people with presumed familial hypercholesterolemia.
The researchers used an Lp(a) genetic risk score based on the variants rs10455872 and rs3798220 and an LDL instrumental variable genetic score comprised of 100 variants to randomly categorize individuals with average Lp(a) levels, higher Lp(a) levels, or higher Lp(a) and lower LDL-cholesterol levels.
The data showed that, with elevated absolute levels of measured Lp(a) and with elevated genetic risk scores, there was a progressive increase in the lifetime risk for major coronary events.
When looking at the combination of both increased Lp(a) levels and lower LDL-cholesterol levels, they found that the increase in risk for major coronary events at Lp(a) of 123 nmol/L could be offset by a reduction in LDL-cholesterol levels of 19.5 mg/dL.
For people with an Lp(a) level of 251 nmol/L, the increase in risk for major coronary events was offset by a reduction in LDL-cholesterol levels of 36.1 mg/dL.
Furthermore, the researchers found that the magnitude of intensification of LDL-cholesterol lowering needed to overcome the risk caused by elevated Lp(a) levels varied by age.
For example, in individuals with an Lp(a) level of 220 nmol/L, the reduction in LDL-cholesterol levels needed to offset the risk for major coronary events was calculated to be 0.8 mmol/L if lipid-lowering was started at 30 years of age, rising to 0.9 mmol/L if started at 40 years, 1.2 mmol/L if started at 50 years, and 1.5 mmol/L if started at 60 years.
This, Dr. Ference said, suggests that “diet and lifestyle modification is unlikely to be an effective strategy if started later.”
No funding was declared. Dr. Ference declared relationships with Amgen, Novartis, Merck, Esperion Therapeutics, Pfizer, Regeneron, Sanofi, AstraZeneca, Eli Lilly, Novo Nordisk, The Medicines Company, Mylan, Daiichi Sankyo, Viatris, Ionis Pharmaceuticals, dalCOR, CiVi Pharma, and KrKa Pharmaceuticals. Dr. Kronenberg declared relationships with Amgen, Novartis, and Kaneka.
A version of this article first appeared on Medscape.com.
MILAN – The increased risk for atherosclerotic cardiovascular disease events caused by elevated lipoprotein(a) levels can potentially be precisely offset by lowering LDL cholesterol to specific levels, suggests a novel study that underscores the importance or early intervention.
The results, derived from an analysis of data on Lp(a) and LDL cholesterol levels and associated genetic risk scores in almost 500,000 individuals from the United Kingdom, have been used to develop a series of age-related targets for lowering LDL cholesterol levels to counter the risk associated with lifetime Lp(a) exposure.
Measuring Lp(a) levels can “substantially refine individual estimates of absolute risk of atherosclerotic cardiovascular disease,” said study presenter Brian A. Ference, MD, Centre for Naturally Randomized Trials, University of Cambridge (England).
This can “directly inform treatment decisions about the intensity of LDL lowering or other risk-factor modification needed to overcome the increased risk caused by Lp(a).”
Dr. Ference said this will allow clinicians to personalize the prevention of atherosclerotic cardiovascular disease and identify people “who may benefit from potent Lp(a)-lowering therapies when they become available.”
The research was presented at the European Atherosclerosis Society (EAS) 2022 congress on May 24.
In addition to producing a tabular version of the intensification of LDL-cholesterol reduction needed to overcome the increased cardiovascular risk at different levels of Lp(a), stratified by age, Dr. Ference is working with the EAS to develop an app to further deliver on that personalized prevention.
It will display an individual’s lifetime risk for myocardial infarction or stroke, with and without the inclusion of Lp(a) levels, and determine not only the percentage of increased risk caused by Lp(a), but also the amount by which LDL cholesterol needs to be lowered to overcome that risk.
“The whole rationale for this study was to say, how can we give practical advice on how to use Lp(a) to inform clinical decisions about how to individualize personal risk reduction,” Dr. Ference told this news organization.
“What the app will do is make it very easy for clinicians to, first, understand how much Lp(a) increases risk, but specifically how they can use that information to directly inform their treatment decisions.”
In addition, Dr. Ference said that it will “show patients why it’s important for them” to intensify LDL lowering to overcome their particular level of Lp(a).
Other key takeaways from the results is the importance of intervention as early as possible to minimize the impact of lifetime exposure to increased Lp(a), and that the reduction in LDL cholesterol required to achieve that remains relatively modest.
For Dr. Ference, this means ideally beginning comprehensive health checks at 30 years of age and starting lipid-lowering interventions immediately for those at risk.
“The good thing about LDL and other causes of atherosclerotic cardiovascular disease is it doesn’t really matter how you lower it,” he said, noting that it could be with diet, lifestyle interventions, or medication.
Handy tool
The new app could be a “handy tool to counsel patients,” Florian Kronenberg, MD, Institute of Genetic Epidemiology, Medical University of Innsbruck, Austria, told this news organization.
“We can say, look, you have high Lp(a),” he said. “This is coming from nature, from your genetics, but here we have a point where we can act on your high risk by lowering LDL further. This is important to explain to the patient,” said Dr. Kronenberg, who was not involved in the study.
He emphasized that it is crucial to get across the idea of an individual’s global risk, with not just Lp(a) or cholesterol levels influencing their likelihood of cardiovascular events, but also their age, blood pressure, smoking status, and underlying genetic risk.
Dr. Kronenberg said the current data will be helpful in explaining to clinicians why they should lower LDL-cholesterol levels when a patients had high Lp(a), again centered on the idea of lowering their global risk.
During his presentation, Dr. Ference noted that an increase in Lp(a) levels is associated with a log-linear increase in atherosclerotic cardiovascular disease that is proportional to the absolute, rather than relative, magnitude of Lp(a) increase.
“Unfortunately, unlike other proteins,” he continued, diet and exercise do not affect levels, and there are currently no effective therapies to lower the risks associated with increased Lp(a) concentrations.
“For that reason,” he said, the 2019 ESC/EAS guidelines for the management of dyslipidemias, on which Dr. Ference was a coauthor, “recommend that we intensify life risk-factor modification in persons with elevated risks.”
However, he added, “this guidance is not specific enough to be useful, and that has created a great deal of inertia among clinicians,” with some concluding that they don’t need to measure Lp(a) “because there’s nothing they can do for it.”
Until the development of novel therapies that directly target Lp(a), the authors sought to quantify the amount of LDL lowering needed to “overcome the increased risk caused by Lp(a),” he said.
They studied data on 455,765 individuals from the UK Biobank who did not have a history of cardiovascular events, diabetes, or any cancer before the age of 30. They also had LDL cholesterol levels below 5 mmol/L at the time of enrollment to exclude people with presumed familial hypercholesterolemia.
The researchers used an Lp(a) genetic risk score based on the variants rs10455872 and rs3798220 and an LDL instrumental variable genetic score comprised of 100 variants to randomly categorize individuals with average Lp(a) levels, higher Lp(a) levels, or higher Lp(a) and lower LDL-cholesterol levels.
The data showed that, with elevated absolute levels of measured Lp(a) and with elevated genetic risk scores, there was a progressive increase in the lifetime risk for major coronary events.
When looking at the combination of both increased Lp(a) levels and lower LDL-cholesterol levels, they found that the increase in risk for major coronary events at Lp(a) of 123 nmol/L could be offset by a reduction in LDL-cholesterol levels of 19.5 mg/dL.
For people with an Lp(a) level of 251 nmol/L, the increase in risk for major coronary events was offset by a reduction in LDL-cholesterol levels of 36.1 mg/dL.
Furthermore, the researchers found that the magnitude of intensification of LDL-cholesterol lowering needed to overcome the risk caused by elevated Lp(a) levels varied by age.
For example, in individuals with an Lp(a) level of 220 nmol/L, the reduction in LDL-cholesterol levels needed to offset the risk for major coronary events was calculated to be 0.8 mmol/L if lipid-lowering was started at 30 years of age, rising to 0.9 mmol/L if started at 40 years, 1.2 mmol/L if started at 50 years, and 1.5 mmol/L if started at 60 years.
This, Dr. Ference said, suggests that “diet and lifestyle modification is unlikely to be an effective strategy if started later.”
No funding was declared. Dr. Ference declared relationships with Amgen, Novartis, Merck, Esperion Therapeutics, Pfizer, Regeneron, Sanofi, AstraZeneca, Eli Lilly, Novo Nordisk, The Medicines Company, Mylan, Daiichi Sankyo, Viatris, Ionis Pharmaceuticals, dalCOR, CiVi Pharma, and KrKa Pharmaceuticals. Dr. Kronenberg declared relationships with Amgen, Novartis, and Kaneka.
A version of this article first appeared on Medscape.com.
AT EAS 2022
Meet the JCOM Author with Dr. Barkoudah: IVIG in Treating Nonventilated COVID-19 Patients With Moderate-to-Severe Hypoxia
Children & COVID: Rise in new cases slows
New cases of COVID-19 in children climbed for the seventh consecutive week, but the latest increase was the smallest of the seven, according to the American Academy of Pediatrics and the Children’s Hospital Association.
Since the weekly total bottomed out at just under 26,000 in early April, the new-case count has risen by 28.0%, 11.8%, 43.5%, 17.4%, 50%, 14.6%, and 5.0%, based on data from the AAP/CHA weekly COVID-19 report.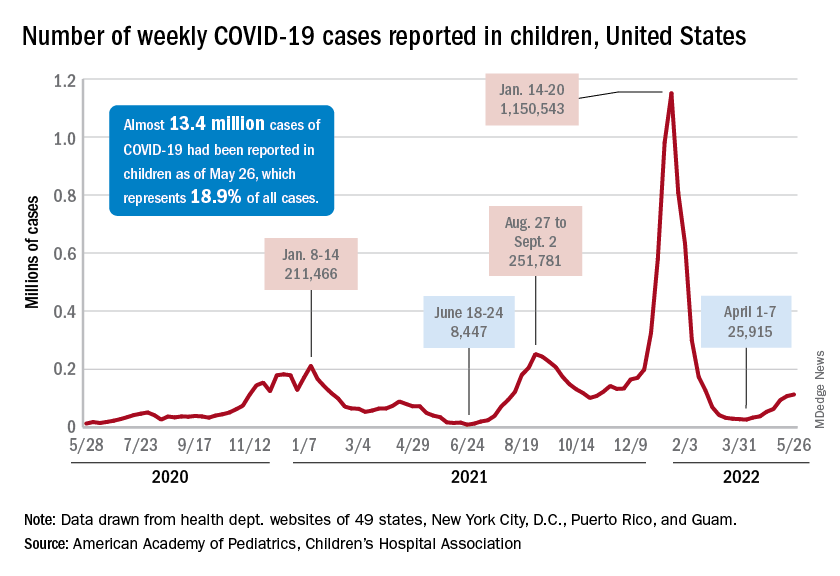
The cumulative number of pediatric cases is almost 13.4 million since the pandemic began, and those infected children represent 18.9% of all cases, the AAP and CHA said based on data from 49 states, New York City, the District of Columbia, Puerto Rico, and Guam.
That 18.9% is noteworthy because it marks the first decline in that particular measure since the AAP and CHA started keeping track in April of 2020. Children’s share of the overall COVID burden had been holding at 19.0% for 14 straight weeks, the AAP/CHA data show.
Regionally, new cases were up in the South and the West, where recent rising trends continued, and down in the Midwest and Northeast, where the recent rising trends were reversed for the first time. At the state/territory level, Puerto Rico had the largest percent increase over the last 2 weeks, followed by Maryland and Delaware, the organizations noted in their joint report.
Hospital admissions in children aged 0-17 have changed little in the last week, with the Centers for Disease Control and Prevention reporting rates of 0.25 per 100,000 population on May 23 and 0.25 per 100,000 on May 29, the latest date available. There was, however, a move up to 0.26 per 100,000 from May 24 to May 28, and the CDC acknowledges a possible reporting delay over the most recent 7-day period.
Emergency department visits have dipped slightly in recent days, with children aged 0-11 years at a 7-day average of 2.0% of ED visits with diagnosed COVID on May 28, down from a 5-day stretch at 2.2% from May 19 to May 23. Children aged 12-15 years were at 1.8% on May 28, compared with 2.0% on May 23-24, and 15- to 17-year-olds were at 2.0% on May 28, down from the 2.1% reached over the previous 2 days, the CDC reported on its COVID Data Tracker.
New cases of COVID-19 in children climbed for the seventh consecutive week, but the latest increase was the smallest of the seven, according to the American Academy of Pediatrics and the Children’s Hospital Association.
Since the weekly total bottomed out at just under 26,000 in early April, the new-case count has risen by 28.0%, 11.8%, 43.5%, 17.4%, 50%, 14.6%, and 5.0%, based on data from the AAP/CHA weekly COVID-19 report.
The cumulative number of pediatric cases is almost 13.4 million since the pandemic began, and those infected children represent 18.9% of all cases, the AAP and CHA said based on data from 49 states, New York City, the District of Columbia, Puerto Rico, and Guam.
That 18.9% is noteworthy because it marks the first decline in that particular measure since the AAP and CHA started keeping track in April of 2020. Children’s share of the overall COVID burden had been holding at 19.0% for 14 straight weeks, the AAP/CHA data show.
Regionally, new cases were up in the South and the West, where recent rising trends continued, and down in the Midwest and Northeast, where the recent rising trends were reversed for the first time. At the state/territory level, Puerto Rico had the largest percent increase over the last 2 weeks, followed by Maryland and Delaware, the organizations noted in their joint report.
Hospital admissions in children aged 0-17 have changed little in the last week, with the Centers for Disease Control and Prevention reporting rates of 0.25 per 100,000 population on May 23 and 0.25 per 100,000 on May 29, the latest date available. There was, however, a move up to 0.26 per 100,000 from May 24 to May 28, and the CDC acknowledges a possible reporting delay over the most recent 7-day period.
Emergency department visits have dipped slightly in recent days, with children aged 0-11 years at a 7-day average of 2.0% of ED visits with diagnosed COVID on May 28, down from a 5-day stretch at 2.2% from May 19 to May 23. Children aged 12-15 years were at 1.8% on May 28, compared with 2.0% on May 23-24, and 15- to 17-year-olds were at 2.0% on May 28, down from the 2.1% reached over the previous 2 days, the CDC reported on its COVID Data Tracker.
New cases of COVID-19 in children climbed for the seventh consecutive week, but the latest increase was the smallest of the seven, according to the American Academy of Pediatrics and the Children’s Hospital Association.
Since the weekly total bottomed out at just under 26,000 in early April, the new-case count has risen by 28.0%, 11.8%, 43.5%, 17.4%, 50%, 14.6%, and 5.0%, based on data from the AAP/CHA weekly COVID-19 report.
The cumulative number of pediatric cases is almost 13.4 million since the pandemic began, and those infected children represent 18.9% of all cases, the AAP and CHA said based on data from 49 states, New York City, the District of Columbia, Puerto Rico, and Guam.
That 18.9% is noteworthy because it marks the first decline in that particular measure since the AAP and CHA started keeping track in April of 2020. Children’s share of the overall COVID burden had been holding at 19.0% for 14 straight weeks, the AAP/CHA data show.
Regionally, new cases were up in the South and the West, where recent rising trends continued, and down in the Midwest and Northeast, where the recent rising trends were reversed for the first time. At the state/territory level, Puerto Rico had the largest percent increase over the last 2 weeks, followed by Maryland and Delaware, the organizations noted in their joint report.
Hospital admissions in children aged 0-17 have changed little in the last week, with the Centers for Disease Control and Prevention reporting rates of 0.25 per 100,000 population on May 23 and 0.25 per 100,000 on May 29, the latest date available. There was, however, a move up to 0.26 per 100,000 from May 24 to May 28, and the CDC acknowledges a possible reporting delay over the most recent 7-day period.
Emergency department visits have dipped slightly in recent days, with children aged 0-11 years at a 7-day average of 2.0% of ED visits with diagnosed COVID on May 28, down from a 5-day stretch at 2.2% from May 19 to May 23. Children aged 12-15 years were at 1.8% on May 28, compared with 2.0% on May 23-24, and 15- to 17-year-olds were at 2.0% on May 28, down from the 2.1% reached over the previous 2 days, the CDC reported on its COVID Data Tracker.
FDA withdraws lymphoma drug approval after investigation
Umbralisib had received accelerated approval in February 2021 to treat adults with relapsed or refractory marginal zone lymphoma following at least one prior therapy and those with relapsed or refractory follicular lymphoma who had received at least three prior therapies.
But safety concerns began to emerge in the phase 3 UNITY-CLL trial, which evaluated the drug in a related cancer type: chronic lymphocytic leukemia.
Last February, the FDA said it was investigating a possible increased risk of death associated with umbralisib.
Five months later, the results are in.
“Updated findings from the UNITY-CLL clinical trial continued to show a possible increased risk of death in patients receiving Ukoniq. As a result, we determined the risks of treatment with Ukoniq outweigh its benefits,” the FDA wrote in a drug safety communication published June 1.
In April, the drug manufacturer, TG Therapeutics, announced it was voluntarily withdrawing umbralisib from the market for its approved uses in marginal zone lymphoma and follicular lymphoma.
The FDA’s safety notice includes instructions for physicians and patients. The FDA urges health care professionals to “stop prescribing Ukoniq and switch patients to alternative treatments” and to “inform patients currently taking Ukoniq of the increased risk of death seen in the clinical trial and advise them to stop taking the medicine.”
In special instances in which a patient may be benefiting from the drug, the company plans to make umbralisib available under expanded access.
The FDA also recommends that patients who discontinue taking the drug dispose of unused umbralisib using a drug take-back location, such as a pharmacy, or throwing it away in the household trash after placing it in a sealed bag mixed with dirt or cat litter and removing personal identification information.
A version of this article first appeared on Medscape.com.
Umbralisib had received accelerated approval in February 2021 to treat adults with relapsed or refractory marginal zone lymphoma following at least one prior therapy and those with relapsed or refractory follicular lymphoma who had received at least three prior therapies.
But safety concerns began to emerge in the phase 3 UNITY-CLL trial, which evaluated the drug in a related cancer type: chronic lymphocytic leukemia.
Last February, the FDA said it was investigating a possible increased risk of death associated with umbralisib.
Five months later, the results are in.
“Updated findings from the UNITY-CLL clinical trial continued to show a possible increased risk of death in patients receiving Ukoniq. As a result, we determined the risks of treatment with Ukoniq outweigh its benefits,” the FDA wrote in a drug safety communication published June 1.
In April, the drug manufacturer, TG Therapeutics, announced it was voluntarily withdrawing umbralisib from the market for its approved uses in marginal zone lymphoma and follicular lymphoma.
The FDA’s safety notice includes instructions for physicians and patients. The FDA urges health care professionals to “stop prescribing Ukoniq and switch patients to alternative treatments” and to “inform patients currently taking Ukoniq of the increased risk of death seen in the clinical trial and advise them to stop taking the medicine.”
In special instances in which a patient may be benefiting from the drug, the company plans to make umbralisib available under expanded access.
The FDA also recommends that patients who discontinue taking the drug dispose of unused umbralisib using a drug take-back location, such as a pharmacy, or throwing it away in the household trash after placing it in a sealed bag mixed with dirt or cat litter and removing personal identification information.
A version of this article first appeared on Medscape.com.
Umbralisib had received accelerated approval in February 2021 to treat adults with relapsed or refractory marginal zone lymphoma following at least one prior therapy and those with relapsed or refractory follicular lymphoma who had received at least three prior therapies.
But safety concerns began to emerge in the phase 3 UNITY-CLL trial, which evaluated the drug in a related cancer type: chronic lymphocytic leukemia.
Last February, the FDA said it was investigating a possible increased risk of death associated with umbralisib.
Five months later, the results are in.
“Updated findings from the UNITY-CLL clinical trial continued to show a possible increased risk of death in patients receiving Ukoniq. As a result, we determined the risks of treatment with Ukoniq outweigh its benefits,” the FDA wrote in a drug safety communication published June 1.
In April, the drug manufacturer, TG Therapeutics, announced it was voluntarily withdrawing umbralisib from the market for its approved uses in marginal zone lymphoma and follicular lymphoma.
The FDA’s safety notice includes instructions for physicians and patients. The FDA urges health care professionals to “stop prescribing Ukoniq and switch patients to alternative treatments” and to “inform patients currently taking Ukoniq of the increased risk of death seen in the clinical trial and advise them to stop taking the medicine.”
In special instances in which a patient may be benefiting from the drug, the company plans to make umbralisib available under expanded access.
The FDA also recommends that patients who discontinue taking the drug dispose of unused umbralisib using a drug take-back location, such as a pharmacy, or throwing it away in the household trash after placing it in a sealed bag mixed with dirt or cat litter and removing personal identification information.
A version of this article first appeared on Medscape.com.
FDA clears Abbott Freestyle Libre 3 glucose sensor
The Food and Drug Administration has cleared Abbot’s Freestyle Libre 3 system for use by people aged 4 years and older with diabetes.
The new system was cleared for use for both iOS- and Android-compatible mobile apps, enabling real-time glucose readings in contrast to the “intermittently scanned” capability of prior Libre versions. The Libre 3 allows for optional alarms and notifications of urgent low or high glucose levels, as well as remote monitoring by health care professionals or the patient’s family members and/or friends.
The FreeStyle Libre 3 was granted a CE Mark in Europe in October 2020.
Smaller, thinner, and better integration
According to Abbott, the Libre 3 is the first continuous glucose monitoring (CGM) system to show a mean absolute relative difference (MARD) of less than 8% compared with a gold-standard glucose measure. The average Libre 3 MARD is 7.9%, compared with 9.3% for the Libre 2. The Libre 3 is also the “smallest and thinnest” CGM, roughly the size of two stacked U.S. pennies, worn on the upper arm.
And, the company said, the Libre 3 has a Bluetooth integration of up to 33 feet, a range 50% further than other CGMs.
This version follows the FreeStyle Libre 2, approved in June 2020, and its compatible iPhone app, approved in August 2021.
The Libre 3 will be priced the same as the Libre 2, at about one-third the cost of other CGM systems. However, it is not currently eligible for Medicare reimbursement. Medicaid eligibility may vary by state.
“I applaud Abbott for making their CGM system the most affordable and addressing disparities in care so patients living with diabetes can avoid complications and optimize their quality of life,” Eugene E. Wright Jr., MD, of Duke University, Durham, N.C., said in an Abbott statement.
“I have seen real-world evidence that diabetes technologies like CGMs have helped my patients safely achieve improved glycemic control,” he said.
The FreeStyle Libre 3 sensor will be available at participating pharmacies later this year.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has cleared Abbot’s Freestyle Libre 3 system for use by people aged 4 years and older with diabetes.
The new system was cleared for use for both iOS- and Android-compatible mobile apps, enabling real-time glucose readings in contrast to the “intermittently scanned” capability of prior Libre versions. The Libre 3 allows for optional alarms and notifications of urgent low or high glucose levels, as well as remote monitoring by health care professionals or the patient’s family members and/or friends.
The FreeStyle Libre 3 was granted a CE Mark in Europe in October 2020.
Smaller, thinner, and better integration
According to Abbott, the Libre 3 is the first continuous glucose monitoring (CGM) system to show a mean absolute relative difference (MARD) of less than 8% compared with a gold-standard glucose measure. The average Libre 3 MARD is 7.9%, compared with 9.3% for the Libre 2. The Libre 3 is also the “smallest and thinnest” CGM, roughly the size of two stacked U.S. pennies, worn on the upper arm.
And, the company said, the Libre 3 has a Bluetooth integration of up to 33 feet, a range 50% further than other CGMs.
This version follows the FreeStyle Libre 2, approved in June 2020, and its compatible iPhone app, approved in August 2021.
The Libre 3 will be priced the same as the Libre 2, at about one-third the cost of other CGM systems. However, it is not currently eligible for Medicare reimbursement. Medicaid eligibility may vary by state.
“I applaud Abbott for making their CGM system the most affordable and addressing disparities in care so patients living with diabetes can avoid complications and optimize their quality of life,” Eugene E. Wright Jr., MD, of Duke University, Durham, N.C., said in an Abbott statement.
“I have seen real-world evidence that diabetes technologies like CGMs have helped my patients safely achieve improved glycemic control,” he said.
The FreeStyle Libre 3 sensor will be available at participating pharmacies later this year.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has cleared Abbot’s Freestyle Libre 3 system for use by people aged 4 years and older with diabetes.
The new system was cleared for use for both iOS- and Android-compatible mobile apps, enabling real-time glucose readings in contrast to the “intermittently scanned” capability of prior Libre versions. The Libre 3 allows for optional alarms and notifications of urgent low or high glucose levels, as well as remote monitoring by health care professionals or the patient’s family members and/or friends.
The FreeStyle Libre 3 was granted a CE Mark in Europe in October 2020.
Smaller, thinner, and better integration
According to Abbott, the Libre 3 is the first continuous glucose monitoring (CGM) system to show a mean absolute relative difference (MARD) of less than 8% compared with a gold-standard glucose measure. The average Libre 3 MARD is 7.9%, compared with 9.3% for the Libre 2. The Libre 3 is also the “smallest and thinnest” CGM, roughly the size of two stacked U.S. pennies, worn on the upper arm.
And, the company said, the Libre 3 has a Bluetooth integration of up to 33 feet, a range 50% further than other CGMs.
This version follows the FreeStyle Libre 2, approved in June 2020, and its compatible iPhone app, approved in August 2021.
The Libre 3 will be priced the same as the Libre 2, at about one-third the cost of other CGM systems. However, it is not currently eligible for Medicare reimbursement. Medicaid eligibility may vary by state.
“I applaud Abbott for making their CGM system the most affordable and addressing disparities in care so patients living with diabetes can avoid complications and optimize their quality of life,” Eugene E. Wright Jr., MD, of Duke University, Durham, N.C., said in an Abbott statement.
“I have seen real-world evidence that diabetes technologies like CGMs have helped my patients safely achieve improved glycemic control,” he said.
The FreeStyle Libre 3 sensor will be available at participating pharmacies later this year.
A version of this article first appeared on Medscape.com.

