User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
More evidence that COVID-19 started in Wuhan marketplace
The original spread of the virus was a one-two punch, the studies found. Twice, the virus jumped from animals to humans. Virus genetics and outbreak modeling in one study revealed two strains released a few weeks apart in November and December 2019.
“Now I realize it sounds like I just said that a once-in-a-generation event happened twice in short succession, and pandemics are indeed rare,” Joel O. Wertheim, PhD, said at a briefing sponsored by the American Association for the Advancement of Science.
A unique storm of factors had to be present for the outbreak to blow up into a pandemic: Animals carrying a virus that could spread to humans, close human contact with these animals, and a city large enough for the infection to take off before it could be contained are examples.
Unluckily for us humans, this coronavirus – SARS-CoV-2 – is a “generalist virus” capable of infecting many animals, including humans.
“Once all the conditions are in place ... the barriers to spillover have been lowered,” said Dr. Wertheim, a researcher in genetic and molecular networks at the University of California, San Diego. In fact, beyond the two strains of the virus that took hold, there were likely up to two dozen more times where people got the virus but did not spread it far and wide, and it died out.
Overall, the odds were against the virus – 78% of the time, the “introduction” to humans was likely to go extinct, the study showed.
The research revealed the COVID-19 pandemic started small.
“Our model shows that there were likely only a few dozen infections, and only several hospitalizations due to COVID-19, by early December,” said Jonathan Pekar, a graduate student working with Dr. Wertheim.
In Wuhan in late 2019, Mr. Pekar said, there was not a single positive coronavirus sample from thousands of samples from healthy blood donors tested between September and December. Likewise, not one blood sample from patients hospitalized with flu-like illness from October to December 2019 tested positive for SARS-CoV-2.
Mapping the outbreak
A second study published in the journal Science mapped out the earliest COVID-19 cases. This effort showed a tight cluster around the wholesale seafood market inside Wuhan, a city of 11 million residents.
When researchers tried other scenarios – modeling outbreaks in other parts of the city – the pattern did not hold. Again, the Wuhan market appeared to be ground zero for the start of the pandemic.
Michael Worobey, PhD, and colleagues used data from Chinese scientists and the World Health Organization for the study.
“There was this extraordinary pattern where the highest density of cases was both extremely near to and very centered on this market,” said Dr. Worobey, head of ecology and evolutionary biology at the University of Arizona, Tucson.
The highest density of cases, in a city of 8,000 square kilometers, was a “very, very small area of about a third of a kilometer square,” he said.
The outbreak pattern showed the Wuhan market “smack dab in the middle.”
So if it started with infected workers at the market, how did it spread from there? It’s likely the virus got into the community as the vendors at the market went to local shops, infecting people in those stores. Then local community members not linked to the market started getting the virus, Dr. Worobey said.
The investigators also identified which stalls in the market were most likely involved, a sort of internal clustering. “That clustering is very, very specifically in the parts of the market where ... they were selling wildlife, including, for example, raccoon dogs and other animals that we know are susceptible to infection with SARS-CoV-2,” said Kristian Andersen, PhD, director of infectious disease genomics at the Scripps Research Institute in La Jolla, Calif.
What remains unknown is which animal or animals carried the virus, although the raccoon dog – an animal similar to a fox that is native to parts of Asia – remains central to most theories. In addition, many of the farms supplying animals to the market have since been closed, making it challenging for researchers to figure out exactly where infected animals came from.
“We don’t know necessarily, but raccoon dogs were sold at this market all the way up to the beginning of the pandemic,” Dr. Andersen said.
Not ruling out other theories
People who believe SARS-CoV-2 was released from a laboratory in China at first included Dr. Worobey himself. “I’ve in the past been much more open to the lab leak idea,” he said. “And published that in a letter in Science” in November 2021.
The letter was “much more influential than I thought it would be in ways that I think it turned out to be quite damaging,” he said. As more evidence emerged since then, Dr. Worobey said he came around to the Wuhan market source theory.
Dr. Andersen agreed he was more open to the lab-leak theory at first. “I was quite convinced of the lab leak myself until we dove into this very carefully and looked at it much closer,” he said. Newer evidence convinced him “that actually, the data points to this particular market.”
“Have we disproved the lab-leak theory? No,” Dr. Anderson said. “Will we ever be able to? No.” But the Wuhan market origin scenario is more plausible. “I would say these two papers combined present the strongest evidence of that to date.”
Identifying the source of the outbreak that led to the COVID-19 pandemic is based in science, Dr. Andersen said. “What we’re trying to understand is the origin of the pandemic. We’re not trying to place blame.”
Future directions
“With pandemics being pandemics, they affect all of us,” Dr. Andersen said. “We can’t prevent these kinds of events that led to the COVID-19 pandemic. But what we can hope to do is to prevent outbreaks from becoming pandemics.”
Rapid reporting of data and cooperation are needed going forward, Dr. Andersen said. Very strong surveillance systems, including wastewater surveillance, could help monitor for SARS-CoV-2, and other pathogens of potential concern in the future as well.
It should be standard practice for medical professionals to be on alert for unusual respiratory infections too, the researchers said.
“It’s a bloody lucky thing that the doctors at the Shinwa hospital were so on the ball, that they noticed that these cases were something unusual at the end of December,” Dr. Worobey said. “It didn’t have to work out that way.”
A version of this article first appeared on WebMD.com.
The original spread of the virus was a one-two punch, the studies found. Twice, the virus jumped from animals to humans. Virus genetics and outbreak modeling in one study revealed two strains released a few weeks apart in November and December 2019.
“Now I realize it sounds like I just said that a once-in-a-generation event happened twice in short succession, and pandemics are indeed rare,” Joel O. Wertheim, PhD, said at a briefing sponsored by the American Association for the Advancement of Science.
A unique storm of factors had to be present for the outbreak to blow up into a pandemic: Animals carrying a virus that could spread to humans, close human contact with these animals, and a city large enough for the infection to take off before it could be contained are examples.
Unluckily for us humans, this coronavirus – SARS-CoV-2 – is a “generalist virus” capable of infecting many animals, including humans.
“Once all the conditions are in place ... the barriers to spillover have been lowered,” said Dr. Wertheim, a researcher in genetic and molecular networks at the University of California, San Diego. In fact, beyond the two strains of the virus that took hold, there were likely up to two dozen more times where people got the virus but did not spread it far and wide, and it died out.
Overall, the odds were against the virus – 78% of the time, the “introduction” to humans was likely to go extinct, the study showed.
The research revealed the COVID-19 pandemic started small.
“Our model shows that there were likely only a few dozen infections, and only several hospitalizations due to COVID-19, by early December,” said Jonathan Pekar, a graduate student working with Dr. Wertheim.
In Wuhan in late 2019, Mr. Pekar said, there was not a single positive coronavirus sample from thousands of samples from healthy blood donors tested between September and December. Likewise, not one blood sample from patients hospitalized with flu-like illness from October to December 2019 tested positive for SARS-CoV-2.
Mapping the outbreak
A second study published in the journal Science mapped out the earliest COVID-19 cases. This effort showed a tight cluster around the wholesale seafood market inside Wuhan, a city of 11 million residents.
When researchers tried other scenarios – modeling outbreaks in other parts of the city – the pattern did not hold. Again, the Wuhan market appeared to be ground zero for the start of the pandemic.
Michael Worobey, PhD, and colleagues used data from Chinese scientists and the World Health Organization for the study.
“There was this extraordinary pattern where the highest density of cases was both extremely near to and very centered on this market,” said Dr. Worobey, head of ecology and evolutionary biology at the University of Arizona, Tucson.
The highest density of cases, in a city of 8,000 square kilometers, was a “very, very small area of about a third of a kilometer square,” he said.
The outbreak pattern showed the Wuhan market “smack dab in the middle.”
So if it started with infected workers at the market, how did it spread from there? It’s likely the virus got into the community as the vendors at the market went to local shops, infecting people in those stores. Then local community members not linked to the market started getting the virus, Dr. Worobey said.
The investigators also identified which stalls in the market were most likely involved, a sort of internal clustering. “That clustering is very, very specifically in the parts of the market where ... they were selling wildlife, including, for example, raccoon dogs and other animals that we know are susceptible to infection with SARS-CoV-2,” said Kristian Andersen, PhD, director of infectious disease genomics at the Scripps Research Institute in La Jolla, Calif.
What remains unknown is which animal or animals carried the virus, although the raccoon dog – an animal similar to a fox that is native to parts of Asia – remains central to most theories. In addition, many of the farms supplying animals to the market have since been closed, making it challenging for researchers to figure out exactly where infected animals came from.
“We don’t know necessarily, but raccoon dogs were sold at this market all the way up to the beginning of the pandemic,” Dr. Andersen said.
Not ruling out other theories
People who believe SARS-CoV-2 was released from a laboratory in China at first included Dr. Worobey himself. “I’ve in the past been much more open to the lab leak idea,” he said. “And published that in a letter in Science” in November 2021.
The letter was “much more influential than I thought it would be in ways that I think it turned out to be quite damaging,” he said. As more evidence emerged since then, Dr. Worobey said he came around to the Wuhan market source theory.
Dr. Andersen agreed he was more open to the lab-leak theory at first. “I was quite convinced of the lab leak myself until we dove into this very carefully and looked at it much closer,” he said. Newer evidence convinced him “that actually, the data points to this particular market.”
“Have we disproved the lab-leak theory? No,” Dr. Anderson said. “Will we ever be able to? No.” But the Wuhan market origin scenario is more plausible. “I would say these two papers combined present the strongest evidence of that to date.”
Identifying the source of the outbreak that led to the COVID-19 pandemic is based in science, Dr. Andersen said. “What we’re trying to understand is the origin of the pandemic. We’re not trying to place blame.”
Future directions
“With pandemics being pandemics, they affect all of us,” Dr. Andersen said. “We can’t prevent these kinds of events that led to the COVID-19 pandemic. But what we can hope to do is to prevent outbreaks from becoming pandemics.”
Rapid reporting of data and cooperation are needed going forward, Dr. Andersen said. Very strong surveillance systems, including wastewater surveillance, could help monitor for SARS-CoV-2, and other pathogens of potential concern in the future as well.
It should be standard practice for medical professionals to be on alert for unusual respiratory infections too, the researchers said.
“It’s a bloody lucky thing that the doctors at the Shinwa hospital were so on the ball, that they noticed that these cases were something unusual at the end of December,” Dr. Worobey said. “It didn’t have to work out that way.”
A version of this article first appeared on WebMD.com.
The original spread of the virus was a one-two punch, the studies found. Twice, the virus jumped from animals to humans. Virus genetics and outbreak modeling in one study revealed two strains released a few weeks apart in November and December 2019.
“Now I realize it sounds like I just said that a once-in-a-generation event happened twice in short succession, and pandemics are indeed rare,” Joel O. Wertheim, PhD, said at a briefing sponsored by the American Association for the Advancement of Science.
A unique storm of factors had to be present for the outbreak to blow up into a pandemic: Animals carrying a virus that could spread to humans, close human contact with these animals, and a city large enough for the infection to take off before it could be contained are examples.
Unluckily for us humans, this coronavirus – SARS-CoV-2 – is a “generalist virus” capable of infecting many animals, including humans.
“Once all the conditions are in place ... the barriers to spillover have been lowered,” said Dr. Wertheim, a researcher in genetic and molecular networks at the University of California, San Diego. In fact, beyond the two strains of the virus that took hold, there were likely up to two dozen more times where people got the virus but did not spread it far and wide, and it died out.
Overall, the odds were against the virus – 78% of the time, the “introduction” to humans was likely to go extinct, the study showed.
The research revealed the COVID-19 pandemic started small.
“Our model shows that there were likely only a few dozen infections, and only several hospitalizations due to COVID-19, by early December,” said Jonathan Pekar, a graduate student working with Dr. Wertheim.
In Wuhan in late 2019, Mr. Pekar said, there was not a single positive coronavirus sample from thousands of samples from healthy blood donors tested between September and December. Likewise, not one blood sample from patients hospitalized with flu-like illness from October to December 2019 tested positive for SARS-CoV-2.
Mapping the outbreak
A second study published in the journal Science mapped out the earliest COVID-19 cases. This effort showed a tight cluster around the wholesale seafood market inside Wuhan, a city of 11 million residents.
When researchers tried other scenarios – modeling outbreaks in other parts of the city – the pattern did not hold. Again, the Wuhan market appeared to be ground zero for the start of the pandemic.
Michael Worobey, PhD, and colleagues used data from Chinese scientists and the World Health Organization for the study.
“There was this extraordinary pattern where the highest density of cases was both extremely near to and very centered on this market,” said Dr. Worobey, head of ecology and evolutionary biology at the University of Arizona, Tucson.
The highest density of cases, in a city of 8,000 square kilometers, was a “very, very small area of about a third of a kilometer square,” he said.
The outbreak pattern showed the Wuhan market “smack dab in the middle.”
So if it started with infected workers at the market, how did it spread from there? It’s likely the virus got into the community as the vendors at the market went to local shops, infecting people in those stores. Then local community members not linked to the market started getting the virus, Dr. Worobey said.
The investigators also identified which stalls in the market were most likely involved, a sort of internal clustering. “That clustering is very, very specifically in the parts of the market where ... they were selling wildlife, including, for example, raccoon dogs and other animals that we know are susceptible to infection with SARS-CoV-2,” said Kristian Andersen, PhD, director of infectious disease genomics at the Scripps Research Institute in La Jolla, Calif.
What remains unknown is which animal or animals carried the virus, although the raccoon dog – an animal similar to a fox that is native to parts of Asia – remains central to most theories. In addition, many of the farms supplying animals to the market have since been closed, making it challenging for researchers to figure out exactly where infected animals came from.
“We don’t know necessarily, but raccoon dogs were sold at this market all the way up to the beginning of the pandemic,” Dr. Andersen said.
Not ruling out other theories
People who believe SARS-CoV-2 was released from a laboratory in China at first included Dr. Worobey himself. “I’ve in the past been much more open to the lab leak idea,” he said. “And published that in a letter in Science” in November 2021.
The letter was “much more influential than I thought it would be in ways that I think it turned out to be quite damaging,” he said. As more evidence emerged since then, Dr. Worobey said he came around to the Wuhan market source theory.
Dr. Andersen agreed he was more open to the lab-leak theory at first. “I was quite convinced of the lab leak myself until we dove into this very carefully and looked at it much closer,” he said. Newer evidence convinced him “that actually, the data points to this particular market.”
“Have we disproved the lab-leak theory? No,” Dr. Anderson said. “Will we ever be able to? No.” But the Wuhan market origin scenario is more plausible. “I would say these two papers combined present the strongest evidence of that to date.”
Identifying the source of the outbreak that led to the COVID-19 pandemic is based in science, Dr. Andersen said. “What we’re trying to understand is the origin of the pandemic. We’re not trying to place blame.”
Future directions
“With pandemics being pandemics, they affect all of us,” Dr. Andersen said. “We can’t prevent these kinds of events that led to the COVID-19 pandemic. But what we can hope to do is to prevent outbreaks from becoming pandemics.”
Rapid reporting of data and cooperation are needed going forward, Dr. Andersen said. Very strong surveillance systems, including wastewater surveillance, could help monitor for SARS-CoV-2, and other pathogens of potential concern in the future as well.
It should be standard practice for medical professionals to be on alert for unusual respiratory infections too, the researchers said.
“It’s a bloody lucky thing that the doctors at the Shinwa hospital were so on the ball, that they noticed that these cases were something unusual at the end of December,” Dr. Worobey said. “It didn’t have to work out that way.”
A version of this article first appeared on WebMD.com.
Pandemic tied to misdiagnosis of rare pneumonia
Psittacosis, a rare disease, has been underdiagnosed or misdiagnosed during the COVID-19 pandemic, likely because the symptoms of the disease are similar to COVID-19 symptoms, researchers suggest on the basis of data from 32 individuals.
Diagnosis of and screening for COVID-19 continues to increase; however, cases of atypical pneumonia caused by uncommon pathogens, which presents with similar symptoms, may be missed, wrote Qiaoqiao Yin, MS, of Zhejiang Provincial People’s Hospital, China, and colleagues.
“The clinical manifestations of human psittacosis can present as rapidly progressing severe pneumonia, acute respiratory distress syndrome, sepsis, and multiple organ failure,” but human cases have not been well studied, they say.
In a study published in the International Journal of Infectious Diseases, the researchers reviewed data from 32 adults diagnosed with Chlamydia psittaci pneumonia during the COVID-19 pandemic between April 2020 and June 2021 in China. The median age of the patients was 63 years, 20 were men, and 20 had underlying diseases.
A total of 17 patients presented with fever, cough, and expectoration of yellow-white sputum. At the time of hospital admission, three patients had myalgia, two had headache, and two had hypertension. The patients were originally suspected of having COVID-19.
all of which could be observed in COVID-19 patients as well, the researchers wrote.
Reverse transcription-polymerase chain reaction (RT-PCR) and enzyme-linked immunosorbent assay (ELISA) testing were used to rule out COVID-19. The researchers then used metagenomic next-generation sequencing (mNGS) to identify the disease-causing pathogens. They collected 18 bronchoalveolar lavage fluid (BALF) samples, 9 peripheral blood samples, and 5 sputum samples. The mNGS identified C. psittaci as the suspected pathogen within 48 hours. Suspected C. psittaci infections were confirmed by endpoint PCR for the BALF and sputum samples and six of nine blood samples, “indicating a lower sensitivity of PCR compared to mNGS for blood samples,” the researchers say. No other potential pathogens were identified.
Psittacosis is common in birds but is rare in humans. C. psittaci is responsible for 1%-8% of cases involving community-acquired pneumonia in China, the researchers note. Although poultry is a source of infection, 25 of the patients in the study did not report a history of exposure to poultry or pigeons at the time of their initial hospital admission. Many patients may be unaware of exposures to poultry, which further complicates the C. psittaci diagnosis, they note.
All patients were treated with doxycycline-based regimens and showed improvement.
The findings were limited by several factors, including the lack of a definitive diagnostic tool for C. psittaci and the lack of convalescent serum samples to confirm cases, the researchers note. In addition, molecular detections for PCR are unavailable in most hospitals in China, they say. The results represent the largest known collection of suspected C. psittaci pneumonia cases and highlight the need for clinician vigilance and awareness of this rare condition, especially in light of the potential for misdiagnosis during the ongoing COVID-19 pandemic, they conclude.
The study received no outside funding. The researchers have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Psittacosis, a rare disease, has been underdiagnosed or misdiagnosed during the COVID-19 pandemic, likely because the symptoms of the disease are similar to COVID-19 symptoms, researchers suggest on the basis of data from 32 individuals.
Diagnosis of and screening for COVID-19 continues to increase; however, cases of atypical pneumonia caused by uncommon pathogens, which presents with similar symptoms, may be missed, wrote Qiaoqiao Yin, MS, of Zhejiang Provincial People’s Hospital, China, and colleagues.
“The clinical manifestations of human psittacosis can present as rapidly progressing severe pneumonia, acute respiratory distress syndrome, sepsis, and multiple organ failure,” but human cases have not been well studied, they say.
In a study published in the International Journal of Infectious Diseases, the researchers reviewed data from 32 adults diagnosed with Chlamydia psittaci pneumonia during the COVID-19 pandemic between April 2020 and June 2021 in China. The median age of the patients was 63 years, 20 were men, and 20 had underlying diseases.
A total of 17 patients presented with fever, cough, and expectoration of yellow-white sputum. At the time of hospital admission, three patients had myalgia, two had headache, and two had hypertension. The patients were originally suspected of having COVID-19.
all of which could be observed in COVID-19 patients as well, the researchers wrote.
Reverse transcription-polymerase chain reaction (RT-PCR) and enzyme-linked immunosorbent assay (ELISA) testing were used to rule out COVID-19. The researchers then used metagenomic next-generation sequencing (mNGS) to identify the disease-causing pathogens. They collected 18 bronchoalveolar lavage fluid (BALF) samples, 9 peripheral blood samples, and 5 sputum samples. The mNGS identified C. psittaci as the suspected pathogen within 48 hours. Suspected C. psittaci infections were confirmed by endpoint PCR for the BALF and sputum samples and six of nine blood samples, “indicating a lower sensitivity of PCR compared to mNGS for blood samples,” the researchers say. No other potential pathogens were identified.
Psittacosis is common in birds but is rare in humans. C. psittaci is responsible for 1%-8% of cases involving community-acquired pneumonia in China, the researchers note. Although poultry is a source of infection, 25 of the patients in the study did not report a history of exposure to poultry or pigeons at the time of their initial hospital admission. Many patients may be unaware of exposures to poultry, which further complicates the C. psittaci diagnosis, they note.
All patients were treated with doxycycline-based regimens and showed improvement.
The findings were limited by several factors, including the lack of a definitive diagnostic tool for C. psittaci and the lack of convalescent serum samples to confirm cases, the researchers note. In addition, molecular detections for PCR are unavailable in most hospitals in China, they say. The results represent the largest known collection of suspected C. psittaci pneumonia cases and highlight the need for clinician vigilance and awareness of this rare condition, especially in light of the potential for misdiagnosis during the ongoing COVID-19 pandemic, they conclude.
The study received no outside funding. The researchers have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Psittacosis, a rare disease, has been underdiagnosed or misdiagnosed during the COVID-19 pandemic, likely because the symptoms of the disease are similar to COVID-19 symptoms, researchers suggest on the basis of data from 32 individuals.
Diagnosis of and screening for COVID-19 continues to increase; however, cases of atypical pneumonia caused by uncommon pathogens, which presents with similar symptoms, may be missed, wrote Qiaoqiao Yin, MS, of Zhejiang Provincial People’s Hospital, China, and colleagues.
“The clinical manifestations of human psittacosis can present as rapidly progressing severe pneumonia, acute respiratory distress syndrome, sepsis, and multiple organ failure,” but human cases have not been well studied, they say.
In a study published in the International Journal of Infectious Diseases, the researchers reviewed data from 32 adults diagnosed with Chlamydia psittaci pneumonia during the COVID-19 pandemic between April 2020 and June 2021 in China. The median age of the patients was 63 years, 20 were men, and 20 had underlying diseases.
A total of 17 patients presented with fever, cough, and expectoration of yellow-white sputum. At the time of hospital admission, three patients had myalgia, two had headache, and two had hypertension. The patients were originally suspected of having COVID-19.
all of which could be observed in COVID-19 patients as well, the researchers wrote.
Reverse transcription-polymerase chain reaction (RT-PCR) and enzyme-linked immunosorbent assay (ELISA) testing were used to rule out COVID-19. The researchers then used metagenomic next-generation sequencing (mNGS) to identify the disease-causing pathogens. They collected 18 bronchoalveolar lavage fluid (BALF) samples, 9 peripheral blood samples, and 5 sputum samples. The mNGS identified C. psittaci as the suspected pathogen within 48 hours. Suspected C. psittaci infections were confirmed by endpoint PCR for the BALF and sputum samples and six of nine blood samples, “indicating a lower sensitivity of PCR compared to mNGS for blood samples,” the researchers say. No other potential pathogens were identified.
Psittacosis is common in birds but is rare in humans. C. psittaci is responsible for 1%-8% of cases involving community-acquired pneumonia in China, the researchers note. Although poultry is a source of infection, 25 of the patients in the study did not report a history of exposure to poultry or pigeons at the time of their initial hospital admission. Many patients may be unaware of exposures to poultry, which further complicates the C. psittaci diagnosis, they note.
All patients were treated with doxycycline-based regimens and showed improvement.
The findings were limited by several factors, including the lack of a definitive diagnostic tool for C. psittaci and the lack of convalescent serum samples to confirm cases, the researchers note. In addition, molecular detections for PCR are unavailable in most hospitals in China, they say. The results represent the largest known collection of suspected C. psittaci pneumonia cases and highlight the need for clinician vigilance and awareness of this rare condition, especially in light of the potential for misdiagnosis during the ongoing COVID-19 pandemic, they conclude.
The study received no outside funding. The researchers have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM INTERNATIONAL JOURNAL OF INFECTIOUS DISEASES
Linezolid succeeds against gram-positive bacterial infections in ICU
Approximately 80% of patients in intensive care showed clinical improvement in gram-positive bacterial infections after treatment with linezolid, based on data from more than 300 individuals.
Bacterial infections remain a challenge in the management of critically ill patients, as many gram-positive pathogens have become resistant to multiple drug options, Aijia Ma, MD, of West China Hospital of Sichuan University, and colleagues wrote.
Linezolid has demonstrated effectiveness against MRSA and skin and soft-tissue infections (SSTIs), but its use in critically ill patients with gram-positive infections in the ICU has not been characterized, they said.
In a multicenter, real-world study published in the Journal of Intensive Medicine, the researchers reviewed data from 52 hospitals between June 2018 and December 2019. The study population included 366 patients admitted to the ICU with a clinical or laboratory diagnosis of a gram-positive bacterial infection. Patients were treated with linezolid injections (200 mg/100 mL) and followed up once a day until 48 hours after discontinuing therapy, transferring out of the ICU, or death. Most of the patients (243) were older than 65 years; 90 were aged 18-65 years, and 30 were younger than 18 years. Approximately two-thirds (67%) were men. The primary outcome of clinical efficacy was success (cured or improved).
Linezolid was used as second-line and first-line treatment in 232 (63.4%) and 134 (36.6%) patients, respectively. The most common isolated strain was Staphylococcus aureus (31% MRSA; 12.6% methicillin-susceptible S. aureus [MSSA]) followed by Enterococci (6.7% vancomycin resistant, 9.2% vancomycin susceptible) and Streptococcus pneumoniae (3.4% multidrug resistant, 1.7% non–multidrug resistant).
Overall, 82.2% of patients met the criteria for clinical success; 34 (9.3%) were cured and 267 (73%) improved. Clinical success rates for first-line and second-line linezolid therapy were 79.9% and 83.6%, respectively. Failure rates for linezolid were higher for second-line versus first-line treatment (9.5% vs. 5.2%).
The clinical success rate was highest against MSSA (93.3%), followed by MRSA (83.8%). The average daily linezolid dose was 1,109 mg, and the mean treatment time was 5.1 days.
A total of eight patients (2.2%) reported linezolid-related adverse events, and four patients discontinued the medication because of them; none reported treatment-related serious adverse events. The low incidence of thrombocytopenia in the current study (two patients), compared with previous studies may have been related to avoidance of linezolid for at-risk patients as determined by clinicians, and the relatively short duration of linezolid use, the researchers wrote.
The study findings were limited by several factors, including the observational design and inability to compare the efficacy of different drugs; the small sample size; and the lack of data on drugs used prior to ICU admission, the researchers noted. Other limitations included the low detection rate of gram-positive bacteria and potential underreporting of adverse events.
However, the although clinicians will need to pay close attention to possible side effects and evaluate patient conditions on an individual basis before using linezolid in the clinic, they concluded.
The study was supported by grants from West China Hospital of Sichuan University. The researchers reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Approximately 80% of patients in intensive care showed clinical improvement in gram-positive bacterial infections after treatment with linezolid, based on data from more than 300 individuals.
Bacterial infections remain a challenge in the management of critically ill patients, as many gram-positive pathogens have become resistant to multiple drug options, Aijia Ma, MD, of West China Hospital of Sichuan University, and colleagues wrote.
Linezolid has demonstrated effectiveness against MRSA and skin and soft-tissue infections (SSTIs), but its use in critically ill patients with gram-positive infections in the ICU has not been characterized, they said.
In a multicenter, real-world study published in the Journal of Intensive Medicine, the researchers reviewed data from 52 hospitals between June 2018 and December 2019. The study population included 366 patients admitted to the ICU with a clinical or laboratory diagnosis of a gram-positive bacterial infection. Patients were treated with linezolid injections (200 mg/100 mL) and followed up once a day until 48 hours after discontinuing therapy, transferring out of the ICU, or death. Most of the patients (243) were older than 65 years; 90 were aged 18-65 years, and 30 were younger than 18 years. Approximately two-thirds (67%) were men. The primary outcome of clinical efficacy was success (cured or improved).
Linezolid was used as second-line and first-line treatment in 232 (63.4%) and 134 (36.6%) patients, respectively. The most common isolated strain was Staphylococcus aureus (31% MRSA; 12.6% methicillin-susceptible S. aureus [MSSA]) followed by Enterococci (6.7% vancomycin resistant, 9.2% vancomycin susceptible) and Streptococcus pneumoniae (3.4% multidrug resistant, 1.7% non–multidrug resistant).
Overall, 82.2% of patients met the criteria for clinical success; 34 (9.3%) were cured and 267 (73%) improved. Clinical success rates for first-line and second-line linezolid therapy were 79.9% and 83.6%, respectively. Failure rates for linezolid were higher for second-line versus first-line treatment (9.5% vs. 5.2%).
The clinical success rate was highest against MSSA (93.3%), followed by MRSA (83.8%). The average daily linezolid dose was 1,109 mg, and the mean treatment time was 5.1 days.
A total of eight patients (2.2%) reported linezolid-related adverse events, and four patients discontinued the medication because of them; none reported treatment-related serious adverse events. The low incidence of thrombocytopenia in the current study (two patients), compared with previous studies may have been related to avoidance of linezolid for at-risk patients as determined by clinicians, and the relatively short duration of linezolid use, the researchers wrote.
The study findings were limited by several factors, including the observational design and inability to compare the efficacy of different drugs; the small sample size; and the lack of data on drugs used prior to ICU admission, the researchers noted. Other limitations included the low detection rate of gram-positive bacteria and potential underreporting of adverse events.
However, the although clinicians will need to pay close attention to possible side effects and evaluate patient conditions on an individual basis before using linezolid in the clinic, they concluded.
The study was supported by grants from West China Hospital of Sichuan University. The researchers reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Approximately 80% of patients in intensive care showed clinical improvement in gram-positive bacterial infections after treatment with linezolid, based on data from more than 300 individuals.
Bacterial infections remain a challenge in the management of critically ill patients, as many gram-positive pathogens have become resistant to multiple drug options, Aijia Ma, MD, of West China Hospital of Sichuan University, and colleagues wrote.
Linezolid has demonstrated effectiveness against MRSA and skin and soft-tissue infections (SSTIs), but its use in critically ill patients with gram-positive infections in the ICU has not been characterized, they said.
In a multicenter, real-world study published in the Journal of Intensive Medicine, the researchers reviewed data from 52 hospitals between June 2018 and December 2019. The study population included 366 patients admitted to the ICU with a clinical or laboratory diagnosis of a gram-positive bacterial infection. Patients were treated with linezolid injections (200 mg/100 mL) and followed up once a day until 48 hours after discontinuing therapy, transferring out of the ICU, or death. Most of the patients (243) were older than 65 years; 90 were aged 18-65 years, and 30 were younger than 18 years. Approximately two-thirds (67%) were men. The primary outcome of clinical efficacy was success (cured or improved).
Linezolid was used as second-line and first-line treatment in 232 (63.4%) and 134 (36.6%) patients, respectively. The most common isolated strain was Staphylococcus aureus (31% MRSA; 12.6% methicillin-susceptible S. aureus [MSSA]) followed by Enterococci (6.7% vancomycin resistant, 9.2% vancomycin susceptible) and Streptococcus pneumoniae (3.4% multidrug resistant, 1.7% non–multidrug resistant).
Overall, 82.2% of patients met the criteria for clinical success; 34 (9.3%) were cured and 267 (73%) improved. Clinical success rates for first-line and second-line linezolid therapy were 79.9% and 83.6%, respectively. Failure rates for linezolid were higher for second-line versus first-line treatment (9.5% vs. 5.2%).
The clinical success rate was highest against MSSA (93.3%), followed by MRSA (83.8%). The average daily linezolid dose was 1,109 mg, and the mean treatment time was 5.1 days.
A total of eight patients (2.2%) reported linezolid-related adverse events, and four patients discontinued the medication because of them; none reported treatment-related serious adverse events. The low incidence of thrombocytopenia in the current study (two patients), compared with previous studies may have been related to avoidance of linezolid for at-risk patients as determined by clinicians, and the relatively short duration of linezolid use, the researchers wrote.
The study findings were limited by several factors, including the observational design and inability to compare the efficacy of different drugs; the small sample size; and the lack of data on drugs used prior to ICU admission, the researchers noted. Other limitations included the low detection rate of gram-positive bacteria and potential underreporting of adverse events.
However, the although clinicians will need to pay close attention to possible side effects and evaluate patient conditions on an individual basis before using linezolid in the clinic, they concluded.
The study was supported by grants from West China Hospital of Sichuan University. The researchers reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF INTENSIVE MEDICINE
Single dose of HPV vaccine is ‘game changer,’ says WHO
The World Health Organization’s Strategic Advisory Group of Experts on Immunization (SAGE) has changed the recommendation for vaccines against human papillomavirus (HPV).
From the available evidence, SAGE has concluded that a single dose of vaccine offers solid protection against HPV, comparable to that achieved with two-dose schedules.
This could be a “game-changer for the prevention of the disease,” as it would allow “more doses of the life-saving jab reach more girls,” the WHO declared in a press release.
SAGE recommends updating HPV dose schedules as follows:
- One- or two-dose schedule for the primary target of girls aged 9-14 years.
- One- or two-dose schedule for young women aged 15-20.
- Two doses with a 6-month interval for women older than 21.
The HPV vaccine is highly effective for the prevention of HPV serotypes 16 and 18, which cause 70% of cases of cervical cancer, said Alejandro Cravioto, MD, PhD, SAGE chair, in a statement.
“SAGE urges all countries to introduce HPV vaccines and prioritize multi-age cohort catch up of missed and older cohorts of girls. These recommendations will enable more girls and women to be vaccinated and thus preventing them from having cervical cancer and all its consequences over the course of their lifetimes,” he added.
For individuals who are immunocompromised, including those with HIV, three doses of the vaccine should be given if feasible, and if not, then at least two doses. There is limited evidence regarding the efficacy of a single dose in this group, the advisory group noted.
Policy makers need to make changes
Now that the WHO has deemed that one dose of HPV vaccine is sufficient, policy makers should make changes, say experts in a recent editorial comment published in The Lancet Oncology.
“Policy makers should consider modifying their HPV immunization schedules for girls aged 9-14 years from a two-dose regimen to a one-dose regimen,” wrote Jeff D’Souza, PhD, Institute for Better Health, Trillium Health Partners, Mississauga, Ont., and David Nderitu, PhD, Egerton University, Nakuru County, Kenya.
Policy makers also need to consider reorienting their efforts on cervical cancer screening and treatment, and they should ensure that all girls globally have access to an effective HPV vaccination schedule, they add.
The editorialists also make a radical proposal.
Existing supply constraints of the HPV vaccine at the country level are expected to continue for the next 3 years, and the vast majority of new cervical cancer cases and related deaths occur in low- and middle-income countries (LMICs).
To overcome these problems, they suggest that “high-income countries that currently offer two-dose regimens to girls aged 9-14 years should consider opting for a one-dose vaccination schedule, and give any excess of vaccines to countries in greater need of them.”
Two doses in high-income countries
But it is unclear whether high-income countries are ready to move to a one-dose schedule.
Approached for comment, Maurie Markman, MD, president of medicine and science at Cancer Treatment Centers of America, Philadelphia, told this news organization that while he can’t say for certain, he suspects that the United States will be slower to accept this recommendation for a single dose of HPV vaccine “as a component of a ‘standard-of-care’ approach.”
However, it “might formally acknowledge that if an individual/parent will only accept a single vaccine dose (or ultimately refuses to return for a recommended second dose), this will be considered a favorable outcome, both for the individual and society.
“I do not know if regulatory bodies in the United States will accept the existing studies performed to address the one-dose vaccination strategy to rather dramatically change the approach in our country,” he said. “The issue would be that if a single dose was stated to be a clinically acceptable option in the United States, it would rapidly become the standard approach, and the regulators would want to be as certain as possible that this would not have a negative effect on what is now recognized to be a remarkably safe and effective cancer prevention effort.”
Another expert who was approached for comment, Stephanie V. Blank, MD, professor of gynecologic oncology at the Icahn School of Medicine at Mount Sinai, New York, said: “In higher-resourced countries, two doses are still preferred, as they are more effective than one.
“The modeling on which the SAGE recommendation is based is all from studies in LMICs and other modeling studies,” she added.
At present, the Centers for Disease Control and Prevention recommends a two-dose schedule of HPV vaccines for individuals who receive the first dose before their 15th birthday. The three-dose schedule is recommended for those who receive the first dose on or after their 15th birthday and for people with certain immunocompromising conditions.
Studies have shown that two doses of HPV vaccine given to children aged 9-14 years provide as good or better protection than three doses given to older adolescents or young adults.
But even with a two-dose schedule, the WHO reports that uptake of the vaccine has been slow, and coverage is much lower than their 90% target. In 2020, global coverage with two doses was only 13%.
Factors that have influenced the slow uptake and low coverage of HPV vaccines include supply challenges, programmatic challenges, and costs related to delivering a two-dose regimen to older girls who are not typically included in childhood vaccination programs. The relatively high cost of HPV vaccines has also been problematic, particularly for middle-income countries.
Trials of one-dose schedules
The one-dose vaccine schedule has garnered a lot of interest, with several studies showing efficacy.
The KEN SHE trial, based in Kenya, showed that a single dose of the HPV vaccine was highly effective at preventing oncogenic infection, rivaling the protection offered by multidose regimens. Vaccine efficacy was 97.5% (P < .001) against HPV 16/18 for both the bivalent and monovalent vaccines, which is “comparable to that seen in multidose vaccine trials,” the researchers noted.
A study in India found that efficacy against persistent HPV 16 and 18 infection among participants evaluable for the endpoint was 95.4% for the single dose, 93.1% for the two-dose schedule, and 93.3% for the three-dose series.
Commenting on this trial in India in a recent interview with this news organization, Geoffroy Canlorbe, MD, PhD, of the department of gynecologic and breast surgery and oncology, Pitié-Salpêtrière Hospital, Paris, said the findings from India would need “to be confirmed by other studies.” The results were nonetheless “excellent news for developing countries where there are challenges when it comes to access to vaccination.”
Speaking at the 45th Congress of the French Society for Colposcopy and Cervical and Vaginal Diseases, he emphasized that at this stage, the findings “cannot be extrapolated” to France. HPV vaccination coverage is low in France (it is estimated that the rate is 23.7%, placing the country 28th of 31 countries in Europe), and he recommended continuing with the two- or three-dose schedule for the time being.
“This poor coverage has nothing to do with health care–related logistical or organizational issues; instead, it has to do with people’s mistrust when it comes to vaccination. Here, people who get the first dose get the subsequent ones,” said Dr. Canlorbe. “The very fact of getting two to three doses allows the person’s body to increase the production of antibodies and get a longer-lasting response to the vaccine.”
Ethics of the vaccine
In their editorial, Dr. D’Souza and Dr. Nderitu note that there are ethical considerations with the HPV vaccine that can “help guide deliberations, covering nonmaleficence, beneficence, health equity, stewardship, and solidarity.”
It would be inequitable and unjustifiable, they write, to offer a two-dose regimen to girls aged 9-14 years without also introducing multi-age cohort catch-up campaigns or programs for women who do not have access. “When it comes to an effective HPV vaccination schedule, no woman or girl should be left behind,” they say.
To achieve the goal of eliminating cervical cancer, “countries must ensure that 90% of girls are vaccinated, 70% of women are screened, and 90% of women with precancerous lesions receive treatment and care,” they write. “Given resource constraints, particularly in low-middle income countries, policy makers have a responsibility to ensure that resources are used in an optimal manner that promotes the right to health of all individuals.”
Thus, countries that are lagging far behind in cervical cancer education, screening, and treatment should consider opting for a one-dose regimen for girls aged 9-14 years, as well as using additional resources to close the gap in these other areas.
Dr. Markman has relationships with Genentech, AstraZeneca, Celgene, Clovis, and Amgen; he is also a regular contributor to Medscape Oncology with the Markamn on Oncology video column. Dr. D’Souza and Dr. Nderitu have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The World Health Organization’s Strategic Advisory Group of Experts on Immunization (SAGE) has changed the recommendation for vaccines against human papillomavirus (HPV).
From the available evidence, SAGE has concluded that a single dose of vaccine offers solid protection against HPV, comparable to that achieved with two-dose schedules.
This could be a “game-changer for the prevention of the disease,” as it would allow “more doses of the life-saving jab reach more girls,” the WHO declared in a press release.
SAGE recommends updating HPV dose schedules as follows:
- One- or two-dose schedule for the primary target of girls aged 9-14 years.
- One- or two-dose schedule for young women aged 15-20.
- Two doses with a 6-month interval for women older than 21.
The HPV vaccine is highly effective for the prevention of HPV serotypes 16 and 18, which cause 70% of cases of cervical cancer, said Alejandro Cravioto, MD, PhD, SAGE chair, in a statement.
“SAGE urges all countries to introduce HPV vaccines and prioritize multi-age cohort catch up of missed and older cohorts of girls. These recommendations will enable more girls and women to be vaccinated and thus preventing them from having cervical cancer and all its consequences over the course of their lifetimes,” he added.
For individuals who are immunocompromised, including those with HIV, three doses of the vaccine should be given if feasible, and if not, then at least two doses. There is limited evidence regarding the efficacy of a single dose in this group, the advisory group noted.
Policy makers need to make changes
Now that the WHO has deemed that one dose of HPV vaccine is sufficient, policy makers should make changes, say experts in a recent editorial comment published in The Lancet Oncology.
“Policy makers should consider modifying their HPV immunization schedules for girls aged 9-14 years from a two-dose regimen to a one-dose regimen,” wrote Jeff D’Souza, PhD, Institute for Better Health, Trillium Health Partners, Mississauga, Ont., and David Nderitu, PhD, Egerton University, Nakuru County, Kenya.
Policy makers also need to consider reorienting their efforts on cervical cancer screening and treatment, and they should ensure that all girls globally have access to an effective HPV vaccination schedule, they add.
The editorialists also make a radical proposal.
Existing supply constraints of the HPV vaccine at the country level are expected to continue for the next 3 years, and the vast majority of new cervical cancer cases and related deaths occur in low- and middle-income countries (LMICs).
To overcome these problems, they suggest that “high-income countries that currently offer two-dose regimens to girls aged 9-14 years should consider opting for a one-dose vaccination schedule, and give any excess of vaccines to countries in greater need of them.”
Two doses in high-income countries
But it is unclear whether high-income countries are ready to move to a one-dose schedule.
Approached for comment, Maurie Markman, MD, president of medicine and science at Cancer Treatment Centers of America, Philadelphia, told this news organization that while he can’t say for certain, he suspects that the United States will be slower to accept this recommendation for a single dose of HPV vaccine “as a component of a ‘standard-of-care’ approach.”
However, it “might formally acknowledge that if an individual/parent will only accept a single vaccine dose (or ultimately refuses to return for a recommended second dose), this will be considered a favorable outcome, both for the individual and society.
“I do not know if regulatory bodies in the United States will accept the existing studies performed to address the one-dose vaccination strategy to rather dramatically change the approach in our country,” he said. “The issue would be that if a single dose was stated to be a clinically acceptable option in the United States, it would rapidly become the standard approach, and the regulators would want to be as certain as possible that this would not have a negative effect on what is now recognized to be a remarkably safe and effective cancer prevention effort.”
Another expert who was approached for comment, Stephanie V. Blank, MD, professor of gynecologic oncology at the Icahn School of Medicine at Mount Sinai, New York, said: “In higher-resourced countries, two doses are still preferred, as they are more effective than one.
“The modeling on which the SAGE recommendation is based is all from studies in LMICs and other modeling studies,” she added.
At present, the Centers for Disease Control and Prevention recommends a two-dose schedule of HPV vaccines for individuals who receive the first dose before their 15th birthday. The three-dose schedule is recommended for those who receive the first dose on or after their 15th birthday and for people with certain immunocompromising conditions.
Studies have shown that two doses of HPV vaccine given to children aged 9-14 years provide as good or better protection than three doses given to older adolescents or young adults.
But even with a two-dose schedule, the WHO reports that uptake of the vaccine has been slow, and coverage is much lower than their 90% target. In 2020, global coverage with two doses was only 13%.
Factors that have influenced the slow uptake and low coverage of HPV vaccines include supply challenges, programmatic challenges, and costs related to delivering a two-dose regimen to older girls who are not typically included in childhood vaccination programs. The relatively high cost of HPV vaccines has also been problematic, particularly for middle-income countries.
Trials of one-dose schedules
The one-dose vaccine schedule has garnered a lot of interest, with several studies showing efficacy.
The KEN SHE trial, based in Kenya, showed that a single dose of the HPV vaccine was highly effective at preventing oncogenic infection, rivaling the protection offered by multidose regimens. Vaccine efficacy was 97.5% (P < .001) against HPV 16/18 for both the bivalent and monovalent vaccines, which is “comparable to that seen in multidose vaccine trials,” the researchers noted.
A study in India found that efficacy against persistent HPV 16 and 18 infection among participants evaluable for the endpoint was 95.4% for the single dose, 93.1% for the two-dose schedule, and 93.3% for the three-dose series.
Commenting on this trial in India in a recent interview with this news organization, Geoffroy Canlorbe, MD, PhD, of the department of gynecologic and breast surgery and oncology, Pitié-Salpêtrière Hospital, Paris, said the findings from India would need “to be confirmed by other studies.” The results were nonetheless “excellent news for developing countries where there are challenges when it comes to access to vaccination.”
Speaking at the 45th Congress of the French Society for Colposcopy and Cervical and Vaginal Diseases, he emphasized that at this stage, the findings “cannot be extrapolated” to France. HPV vaccination coverage is low in France (it is estimated that the rate is 23.7%, placing the country 28th of 31 countries in Europe), and he recommended continuing with the two- or three-dose schedule for the time being.
“This poor coverage has nothing to do with health care–related logistical or organizational issues; instead, it has to do with people’s mistrust when it comes to vaccination. Here, people who get the first dose get the subsequent ones,” said Dr. Canlorbe. “The very fact of getting two to three doses allows the person’s body to increase the production of antibodies and get a longer-lasting response to the vaccine.”
Ethics of the vaccine
In their editorial, Dr. D’Souza and Dr. Nderitu note that there are ethical considerations with the HPV vaccine that can “help guide deliberations, covering nonmaleficence, beneficence, health equity, stewardship, and solidarity.”
It would be inequitable and unjustifiable, they write, to offer a two-dose regimen to girls aged 9-14 years without also introducing multi-age cohort catch-up campaigns or programs for women who do not have access. “When it comes to an effective HPV vaccination schedule, no woman or girl should be left behind,” they say.
To achieve the goal of eliminating cervical cancer, “countries must ensure that 90% of girls are vaccinated, 70% of women are screened, and 90% of women with precancerous lesions receive treatment and care,” they write. “Given resource constraints, particularly in low-middle income countries, policy makers have a responsibility to ensure that resources are used in an optimal manner that promotes the right to health of all individuals.”
Thus, countries that are lagging far behind in cervical cancer education, screening, and treatment should consider opting for a one-dose regimen for girls aged 9-14 years, as well as using additional resources to close the gap in these other areas.
Dr. Markman has relationships with Genentech, AstraZeneca, Celgene, Clovis, and Amgen; he is also a regular contributor to Medscape Oncology with the Markamn on Oncology video column. Dr. D’Souza and Dr. Nderitu have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The World Health Organization’s Strategic Advisory Group of Experts on Immunization (SAGE) has changed the recommendation for vaccines against human papillomavirus (HPV).
From the available evidence, SAGE has concluded that a single dose of vaccine offers solid protection against HPV, comparable to that achieved with two-dose schedules.
This could be a “game-changer for the prevention of the disease,” as it would allow “more doses of the life-saving jab reach more girls,” the WHO declared in a press release.
SAGE recommends updating HPV dose schedules as follows:
- One- or two-dose schedule for the primary target of girls aged 9-14 years.
- One- or two-dose schedule for young women aged 15-20.
- Two doses with a 6-month interval for women older than 21.
The HPV vaccine is highly effective for the prevention of HPV serotypes 16 and 18, which cause 70% of cases of cervical cancer, said Alejandro Cravioto, MD, PhD, SAGE chair, in a statement.
“SAGE urges all countries to introduce HPV vaccines and prioritize multi-age cohort catch up of missed and older cohorts of girls. These recommendations will enable more girls and women to be vaccinated and thus preventing them from having cervical cancer and all its consequences over the course of their lifetimes,” he added.
For individuals who are immunocompromised, including those with HIV, three doses of the vaccine should be given if feasible, and if not, then at least two doses. There is limited evidence regarding the efficacy of a single dose in this group, the advisory group noted.
Policy makers need to make changes
Now that the WHO has deemed that one dose of HPV vaccine is sufficient, policy makers should make changes, say experts in a recent editorial comment published in The Lancet Oncology.
“Policy makers should consider modifying their HPV immunization schedules for girls aged 9-14 years from a two-dose regimen to a one-dose regimen,” wrote Jeff D’Souza, PhD, Institute for Better Health, Trillium Health Partners, Mississauga, Ont., and David Nderitu, PhD, Egerton University, Nakuru County, Kenya.
Policy makers also need to consider reorienting their efforts on cervical cancer screening and treatment, and they should ensure that all girls globally have access to an effective HPV vaccination schedule, they add.
The editorialists also make a radical proposal.
Existing supply constraints of the HPV vaccine at the country level are expected to continue for the next 3 years, and the vast majority of new cervical cancer cases and related deaths occur in low- and middle-income countries (LMICs).
To overcome these problems, they suggest that “high-income countries that currently offer two-dose regimens to girls aged 9-14 years should consider opting for a one-dose vaccination schedule, and give any excess of vaccines to countries in greater need of them.”
Two doses in high-income countries
But it is unclear whether high-income countries are ready to move to a one-dose schedule.
Approached for comment, Maurie Markman, MD, president of medicine and science at Cancer Treatment Centers of America, Philadelphia, told this news organization that while he can’t say for certain, he suspects that the United States will be slower to accept this recommendation for a single dose of HPV vaccine “as a component of a ‘standard-of-care’ approach.”
However, it “might formally acknowledge that if an individual/parent will only accept a single vaccine dose (or ultimately refuses to return for a recommended second dose), this will be considered a favorable outcome, both for the individual and society.
“I do not know if regulatory bodies in the United States will accept the existing studies performed to address the one-dose vaccination strategy to rather dramatically change the approach in our country,” he said. “The issue would be that if a single dose was stated to be a clinically acceptable option in the United States, it would rapidly become the standard approach, and the regulators would want to be as certain as possible that this would not have a negative effect on what is now recognized to be a remarkably safe and effective cancer prevention effort.”
Another expert who was approached for comment, Stephanie V. Blank, MD, professor of gynecologic oncology at the Icahn School of Medicine at Mount Sinai, New York, said: “In higher-resourced countries, two doses are still preferred, as they are more effective than one.
“The modeling on which the SAGE recommendation is based is all from studies in LMICs and other modeling studies,” she added.
At present, the Centers for Disease Control and Prevention recommends a two-dose schedule of HPV vaccines for individuals who receive the first dose before their 15th birthday. The three-dose schedule is recommended for those who receive the first dose on or after their 15th birthday and for people with certain immunocompromising conditions.
Studies have shown that two doses of HPV vaccine given to children aged 9-14 years provide as good or better protection than three doses given to older adolescents or young adults.
But even with a two-dose schedule, the WHO reports that uptake of the vaccine has been slow, and coverage is much lower than their 90% target. In 2020, global coverage with two doses was only 13%.
Factors that have influenced the slow uptake and low coverage of HPV vaccines include supply challenges, programmatic challenges, and costs related to delivering a two-dose regimen to older girls who are not typically included in childhood vaccination programs. The relatively high cost of HPV vaccines has also been problematic, particularly for middle-income countries.
Trials of one-dose schedules
The one-dose vaccine schedule has garnered a lot of interest, with several studies showing efficacy.
The KEN SHE trial, based in Kenya, showed that a single dose of the HPV vaccine was highly effective at preventing oncogenic infection, rivaling the protection offered by multidose regimens. Vaccine efficacy was 97.5% (P < .001) against HPV 16/18 for both the bivalent and monovalent vaccines, which is “comparable to that seen in multidose vaccine trials,” the researchers noted.
A study in India found that efficacy against persistent HPV 16 and 18 infection among participants evaluable for the endpoint was 95.4% for the single dose, 93.1% for the two-dose schedule, and 93.3% for the three-dose series.
Commenting on this trial in India in a recent interview with this news organization, Geoffroy Canlorbe, MD, PhD, of the department of gynecologic and breast surgery and oncology, Pitié-Salpêtrière Hospital, Paris, said the findings from India would need “to be confirmed by other studies.” The results were nonetheless “excellent news for developing countries where there are challenges when it comes to access to vaccination.”
Speaking at the 45th Congress of the French Society for Colposcopy and Cervical and Vaginal Diseases, he emphasized that at this stage, the findings “cannot be extrapolated” to France. HPV vaccination coverage is low in France (it is estimated that the rate is 23.7%, placing the country 28th of 31 countries in Europe), and he recommended continuing with the two- or three-dose schedule for the time being.
“This poor coverage has nothing to do with health care–related logistical or organizational issues; instead, it has to do with people’s mistrust when it comes to vaccination. Here, people who get the first dose get the subsequent ones,” said Dr. Canlorbe. “The very fact of getting two to three doses allows the person’s body to increase the production of antibodies and get a longer-lasting response to the vaccine.”
Ethics of the vaccine
In their editorial, Dr. D’Souza and Dr. Nderitu note that there are ethical considerations with the HPV vaccine that can “help guide deliberations, covering nonmaleficence, beneficence, health equity, stewardship, and solidarity.”
It would be inequitable and unjustifiable, they write, to offer a two-dose regimen to girls aged 9-14 years without also introducing multi-age cohort catch-up campaigns or programs for women who do not have access. “When it comes to an effective HPV vaccination schedule, no woman or girl should be left behind,” they say.
To achieve the goal of eliminating cervical cancer, “countries must ensure that 90% of girls are vaccinated, 70% of women are screened, and 90% of women with precancerous lesions receive treatment and care,” they write. “Given resource constraints, particularly in low-middle income countries, policy makers have a responsibility to ensure that resources are used in an optimal manner that promotes the right to health of all individuals.”
Thus, countries that are lagging far behind in cervical cancer education, screening, and treatment should consider opting for a one-dose regimen for girls aged 9-14 years, as well as using additional resources to close the gap in these other areas.
Dr. Markman has relationships with Genentech, AstraZeneca, Celgene, Clovis, and Amgen; he is also a regular contributor to Medscape Oncology with the Markamn on Oncology video column. Dr. D’Souza and Dr. Nderitu have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
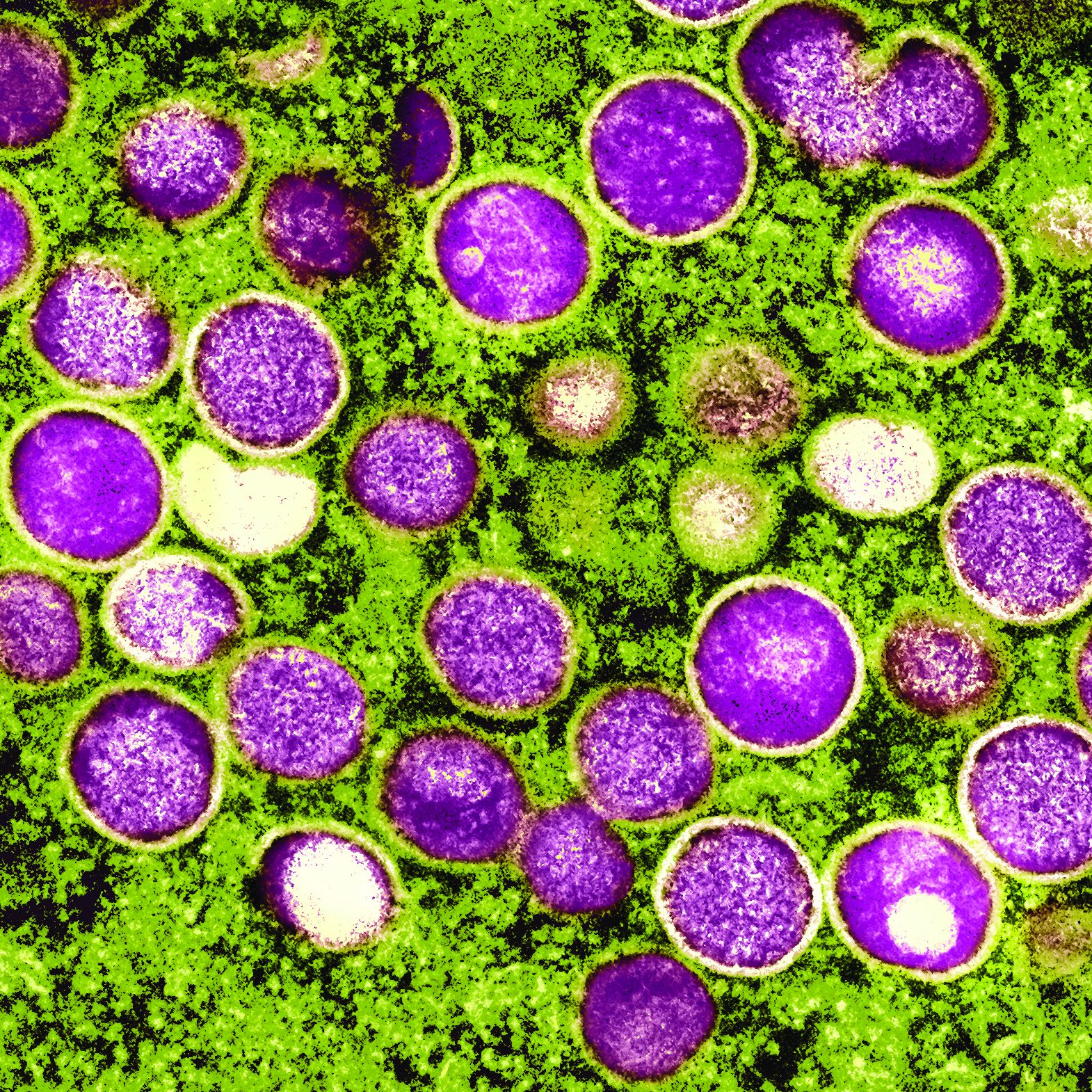
To gauge monkeypox spread, researchers eye cases in women
As cases of monkeypox continue to mount in the United States and abroad, infectious disease experts are closely monitoring one group of people in particular: women.
So far, the overwhelming majority of cases of the viral disease have been reported in men who have sex with men. But in recent days, officials have learned of a handful of cases in women – possibly indicating that the outbreak may be widening.
Researchers are keeping close tabs on the proportion of cases in women to “assess whether the outbreak is moving away” from networks of men who have sex with men, where most of the initial cases have been identified, according to a briefing from the UK Health Security Agency (UKHSA).
“There is insufficient evidence to support a change in the transmission dynamics,” the agency said. “However, over the last few weeks the proportion of female cases has been increasing, so this trend needs to be monitored closely.”
A global collaboration of researchers and clinicians recently described 528 cases of monkeypox in 16 countries – but none were in women.
Since data collection for that study ended in June, the research group has confirmed cases in women, said study coauthor John P. Thornhill, MD, PhD, consultant physician in sexual health and HIV and clinical senior lecturer at Barts Health NHS Trust and Queen Mary University of London.
“Cases in women have certainly been reported but are currently far less common,” Dr. Thornhill told this news organization.
Although infections in women have been outliers during the current outbreak, they can be severe when they do occur. Several women in England have been hospitalized with severe symptoms.
A similar pattern has been seen in New York City, where just one woman is among the 639 total cases, according to a July 21 report from the city’s health agency.
Researchers have recently published guidance on monkeypox for ob.gyns., maternal-fetal medicine subspecialists, and people who are pregnant or breastfeeding in anticipation of the possibility of more cases in women.
The Centers for Disease Control and Prevention advises that “pregnant, recently pregnant, and breastfeeding people should be prioritized for medical treatment” of monkeypox if needed.
One monkeypox vaccine, Jynneos, can be offered to people who are pregnant or breastfeeding and are otherwise eligible for vaccination on the basis of confirmed or likely contact with cases, ideally within 4 days of exposure. Some people at high risk for exposure, such as laboratory workers, may receive the vaccine preemptively.
Another vaccine, ACAM2000, is contraindicated in people who are pregnant or breastfeeding, according to the CDC.
Transmission dynamics
Investigators have not yet identified substantial spread of monkeypox beyond men who have sex with men, although transmission among household contacts, including women and children, has been reported.
Most initial infections during the current outbreak occurred during sexual activity. But monkeypox can spread through any close contact with skin lesions or body fluids and possibly through touching contaminated items like clothing or linens, according to the CDC. It also may spread from mother to child in utero.
Infected pets have been known to spread the disease as well. A multistate monkeypox outbreak in the United States in 2003 was linked to pet prairie dogs, including in childcare and school settings. That year, 55% of the 71 cases occurred in female patients.
More testing, higher positivity rates in men
Since May, more men than women in the United Kingdom have undergone testing for monkeypox, with 3,467 tests in men versus 447 tests in women. Among those tested, the positivity rate has been far higher in men than in women, 54% versus 2.2%, respectively.
As of July 20, about 0.65% of U.K. cases with known gender were in women. Two weeks prior, about 0.4% were in women.
In all, 13 monkeypox cases in England have been in women, and four had severe manifestations that required hospitalization, according to the UKHSA.
Globally, more than 16,000 monkeypox cases have been reported, according to the World Health Organization. The agency said that it plans to rename the disease to reduce stigma.
Monkeypox and pregnancy
Ob.gyns. are often on the “front line in terms of identifying people with infectious diseases,” said Denise J. Jamieson, MD, MPH, Emory University, Atlanta. Dr. Jamieson coauthored “A Primer on Monkeypox Virus for Obstetrician-Gynecologists,” published in Obstetrics & Gynecology.
“Obstetricians need to be aware of what infectious diseases are circulating and be aware of what is going on in the community,” she said.
With monkeypox, “it is anybody’s guess as to how widespread this is going to be,” Dr. Jamieson said.
“The initial monkeypox cases in the current outbreak have been predominately but not exclusively among men who have sex with men; enhanced transmission in this group may be facilitated by sexual activity and spread through complex sexual networks,” Dr. Thornhill said. “As the outbreak continues, we will likely see more monkeypox infections” outside that group.
“Those working in sexual health should have a high index of suspicion in all individuals presenting with genital and oral ulcers and those with proctitis,” he added.
During previous monkeypox outbreaks, the chain of household transmissions has been short, typically two or three people, said Chloe M. Orkin, MD, professor of HIV medicine at Queen Mary University of London. Dr. Orkin directs the Sexual Health and HIV All East Research (SHARE) Collaborative, which has worked to compile the international case series.
Though monkeypox has mainly been transmitted among men who have sex with men, not all identify as gay and some may also have female and nonbinary partners, Dr. Orkin said.
“Clinicians should bear this in mind when examining any person,” she said.
A version of this article first appeared on Medscape.com.
As cases of monkeypox continue to mount in the United States and abroad, infectious disease experts are closely monitoring one group of people in particular: women.
So far, the overwhelming majority of cases of the viral disease have been reported in men who have sex with men. But in recent days, officials have learned of a handful of cases in women – possibly indicating that the outbreak may be widening.
Researchers are keeping close tabs on the proportion of cases in women to “assess whether the outbreak is moving away” from networks of men who have sex with men, where most of the initial cases have been identified, according to a briefing from the UK Health Security Agency (UKHSA).
“There is insufficient evidence to support a change in the transmission dynamics,” the agency said. “However, over the last few weeks the proportion of female cases has been increasing, so this trend needs to be monitored closely.”
A global collaboration of researchers and clinicians recently described 528 cases of monkeypox in 16 countries – but none were in women.
Since data collection for that study ended in June, the research group has confirmed cases in women, said study coauthor John P. Thornhill, MD, PhD, consultant physician in sexual health and HIV and clinical senior lecturer at Barts Health NHS Trust and Queen Mary University of London.
“Cases in women have certainly been reported but are currently far less common,” Dr. Thornhill told this news organization.
Although infections in women have been outliers during the current outbreak, they can be severe when they do occur. Several women in England have been hospitalized with severe symptoms.
A similar pattern has been seen in New York City, where just one woman is among the 639 total cases, according to a July 21 report from the city’s health agency.
Researchers have recently published guidance on monkeypox for ob.gyns., maternal-fetal medicine subspecialists, and people who are pregnant or breastfeeding in anticipation of the possibility of more cases in women.
The Centers for Disease Control and Prevention advises that “pregnant, recently pregnant, and breastfeeding people should be prioritized for medical treatment” of monkeypox if needed.
One monkeypox vaccine, Jynneos, can be offered to people who are pregnant or breastfeeding and are otherwise eligible for vaccination on the basis of confirmed or likely contact with cases, ideally within 4 days of exposure. Some people at high risk for exposure, such as laboratory workers, may receive the vaccine preemptively.
Another vaccine, ACAM2000, is contraindicated in people who are pregnant or breastfeeding, according to the CDC.
Transmission dynamics
Investigators have not yet identified substantial spread of monkeypox beyond men who have sex with men, although transmission among household contacts, including women and children, has been reported.
Most initial infections during the current outbreak occurred during sexual activity. But monkeypox can spread through any close contact with skin lesions or body fluids and possibly through touching contaminated items like clothing or linens, according to the CDC. It also may spread from mother to child in utero.
Infected pets have been known to spread the disease as well. A multistate monkeypox outbreak in the United States in 2003 was linked to pet prairie dogs, including in childcare and school settings. That year, 55% of the 71 cases occurred in female patients.
More testing, higher positivity rates in men
Since May, more men than women in the United Kingdom have undergone testing for monkeypox, with 3,467 tests in men versus 447 tests in women. Among those tested, the positivity rate has been far higher in men than in women, 54% versus 2.2%, respectively.
As of July 20, about 0.65% of U.K. cases with known gender were in women. Two weeks prior, about 0.4% were in women.
In all, 13 monkeypox cases in England have been in women, and four had severe manifestations that required hospitalization, according to the UKHSA.
Globally, more than 16,000 monkeypox cases have been reported, according to the World Health Organization. The agency said that it plans to rename the disease to reduce stigma.
Monkeypox and pregnancy
Ob.gyns. are often on the “front line in terms of identifying people with infectious diseases,” said Denise J. Jamieson, MD, MPH, Emory University, Atlanta. Dr. Jamieson coauthored “A Primer on Monkeypox Virus for Obstetrician-Gynecologists,” published in Obstetrics & Gynecology.
“Obstetricians need to be aware of what infectious diseases are circulating and be aware of what is going on in the community,” she said.
With monkeypox, “it is anybody’s guess as to how widespread this is going to be,” Dr. Jamieson said.
“The initial monkeypox cases in the current outbreak have been predominately but not exclusively among men who have sex with men; enhanced transmission in this group may be facilitated by sexual activity and spread through complex sexual networks,” Dr. Thornhill said. “As the outbreak continues, we will likely see more monkeypox infections” outside that group.
“Those working in sexual health should have a high index of suspicion in all individuals presenting with genital and oral ulcers and those with proctitis,” he added.
During previous monkeypox outbreaks, the chain of household transmissions has been short, typically two or three people, said Chloe M. Orkin, MD, professor of HIV medicine at Queen Mary University of London. Dr. Orkin directs the Sexual Health and HIV All East Research (SHARE) Collaborative, which has worked to compile the international case series.
Though monkeypox has mainly been transmitted among men who have sex with men, not all identify as gay and some may also have female and nonbinary partners, Dr. Orkin said.
“Clinicians should bear this in mind when examining any person,” she said.
A version of this article first appeared on Medscape.com.
As cases of monkeypox continue to mount in the United States and abroad, infectious disease experts are closely monitoring one group of people in particular: women.
So far, the overwhelming majority of cases of the viral disease have been reported in men who have sex with men. But in recent days, officials have learned of a handful of cases in women – possibly indicating that the outbreak may be widening.
Researchers are keeping close tabs on the proportion of cases in women to “assess whether the outbreak is moving away” from networks of men who have sex with men, where most of the initial cases have been identified, according to a briefing from the UK Health Security Agency (UKHSA).
“There is insufficient evidence to support a change in the transmission dynamics,” the agency said. “However, over the last few weeks the proportion of female cases has been increasing, so this trend needs to be monitored closely.”
A global collaboration of researchers and clinicians recently described 528 cases of monkeypox in 16 countries – but none were in women.
Since data collection for that study ended in June, the research group has confirmed cases in women, said study coauthor John P. Thornhill, MD, PhD, consultant physician in sexual health and HIV and clinical senior lecturer at Barts Health NHS Trust and Queen Mary University of London.
“Cases in women have certainly been reported but are currently far less common,” Dr. Thornhill told this news organization.
Although infections in women have been outliers during the current outbreak, they can be severe when they do occur. Several women in England have been hospitalized with severe symptoms.
A similar pattern has been seen in New York City, where just one woman is among the 639 total cases, according to a July 21 report from the city’s health agency.
Researchers have recently published guidance on monkeypox for ob.gyns., maternal-fetal medicine subspecialists, and people who are pregnant or breastfeeding in anticipation of the possibility of more cases in women.
The Centers for Disease Control and Prevention advises that “pregnant, recently pregnant, and breastfeeding people should be prioritized for medical treatment” of monkeypox if needed.
One monkeypox vaccine, Jynneos, can be offered to people who are pregnant or breastfeeding and are otherwise eligible for vaccination on the basis of confirmed or likely contact with cases, ideally within 4 days of exposure. Some people at high risk for exposure, such as laboratory workers, may receive the vaccine preemptively.
Another vaccine, ACAM2000, is contraindicated in people who are pregnant or breastfeeding, according to the CDC.
Transmission dynamics
Investigators have not yet identified substantial spread of monkeypox beyond men who have sex with men, although transmission among household contacts, including women and children, has been reported.
Most initial infections during the current outbreak occurred during sexual activity. But monkeypox can spread through any close contact with skin lesions or body fluids and possibly through touching contaminated items like clothing or linens, according to the CDC. It also may spread from mother to child in utero.
Infected pets have been known to spread the disease as well. A multistate monkeypox outbreak in the United States in 2003 was linked to pet prairie dogs, including in childcare and school settings. That year, 55% of the 71 cases occurred in female patients.
More testing, higher positivity rates in men
Since May, more men than women in the United Kingdom have undergone testing for monkeypox, with 3,467 tests in men versus 447 tests in women. Among those tested, the positivity rate has been far higher in men than in women, 54% versus 2.2%, respectively.
As of July 20, about 0.65% of U.K. cases with known gender were in women. Two weeks prior, about 0.4% were in women.
In all, 13 monkeypox cases in England have been in women, and four had severe manifestations that required hospitalization, according to the UKHSA.
Globally, more than 16,000 monkeypox cases have been reported, according to the World Health Organization. The agency said that it plans to rename the disease to reduce stigma.
Monkeypox and pregnancy
Ob.gyns. are often on the “front line in terms of identifying people with infectious diseases,” said Denise J. Jamieson, MD, MPH, Emory University, Atlanta. Dr. Jamieson coauthored “A Primer on Monkeypox Virus for Obstetrician-Gynecologists,” published in Obstetrics & Gynecology.
“Obstetricians need to be aware of what infectious diseases are circulating and be aware of what is going on in the community,” she said.
With monkeypox, “it is anybody’s guess as to how widespread this is going to be,” Dr. Jamieson said.
“The initial monkeypox cases in the current outbreak have been predominately but not exclusively among men who have sex with men; enhanced transmission in this group may be facilitated by sexual activity and spread through complex sexual networks,” Dr. Thornhill said. “As the outbreak continues, we will likely see more monkeypox infections” outside that group.
“Those working in sexual health should have a high index of suspicion in all individuals presenting with genital and oral ulcers and those with proctitis,” he added.
During previous monkeypox outbreaks, the chain of household transmissions has been short, typically two or three people, said Chloe M. Orkin, MD, professor of HIV medicine at Queen Mary University of London. Dr. Orkin directs the Sexual Health and HIV All East Research (SHARE) Collaborative, which has worked to compile the international case series.
Though monkeypox has mainly been transmitted among men who have sex with men, not all identify as gay and some may also have female and nonbinary partners, Dr. Orkin said.
“Clinicians should bear this in mind when examining any person,” she said.
A version of this article first appeared on Medscape.com.
Fourth patient cleared of HIV after stem cell transplant for blood cancer
MONTREAL – from a naturally HIV-resistant donor, U.S. researchers announced at a meeting of the International AIDS Society.
The man received the transplant nearly 3.5 years ago. Since discontinuation of antiretroviral therapy (ART) more than 17 months ago, he has shown no evidence of HIV-1 RNA rebound and no detectable HIV-1 DNA, reported lead investigator Jana K. Dickter, MD, associate clinical professor in the division of infectious diseases at City of Hope, a Duarte, Calif.–based stem cell transplantation center for patients with blood cancers and patients with HIV/blood cancer.
Known as the City of Hope (COH) patient, he is different from the three previously reported patients in that “he was the oldest person to successfully undergo a stem cell transplant with HIV and leukemia and then achieve remission from both conditions,” Dr. Dickter said during a press briefing for the meeting. “He has been living with HIV the longest of any of the patients to date – more than 31 years prior to transplant – and he had also received the least immunosuppressive preparative regimen prior to transplant,” she added.
She said that, like the three previous patients, known as the Berlin, London, and New York patients, the COH patient received a transplant from a donor with natural resistance to HIV because of a rare CCR5-delta 32 mutation.
Dr. Dickter and her coinvestigators used the term “remission” but went further, suggesting that an “HIV cure is feasible” after transplant, given this and the previous cases.
“It’s a bit early to say the patient is cured, but they are clearly in remission,” said Sharon Lewin, MD, president-elect of the International AIDS Society, which runs the meeting. Nevertheless, Dr. Lewin, professor of medicine at the University of Melbourne and director of the Peter Doherty Institute for Infection and Immunity, in Melbourne, acknowledged that cure is “very likely.”
“Two of the previously reported patients have been off ART for long periods of time – Berlin, 12 years (until Timothy’s death in 2020); London, 4 years – and both had far more extensive investigations to try and find intact virus, including very large blood draws, tissue biopsies, etc. For the New York and now this COH patient, the duration off ART has been much shorter. ... But given the prior cases, it is very likely that the New York and COH patients are indeed cured. But I think it’s too early to make that call, hence my preference to use the word, ‘remission,’ “ she told this news organization.
“Although a transplant is not an option for most people with HIV, these cases are still interesting, still inspiring, and help illuminate the search for a cure,” she added.
Dr. Dickter acknowledged that the complexity of stem cell transplant procedures and their potential for significant side effects make them unsuitable as treatment options for most people with HIV, although she said the COH case is evidence that some HIV patients with blood cancers may not need such intensive pretransplant conditioning regimens.
The COH patient received a reduced-intensity fludarabine and melphalan regimen that had been designed at Dr. Dickter’s center “for older and less fit patients to make transplantation more tolerable,” she said. In addition, the graft-vs.-host disease prophylaxis that the COH patient received included only tacrolimus and sirolimus, whereas the previous patients received additional immunosuppressive therapies, and some also had undergone total body irradiation.
Dr. Dickter has disclosed no relevant financial relationships. Dr. Lewin has relationships with AbbVie, BMS, Esfam, Genentech, Gilead, Immunocore, Merck, Vaxxinity, and Viiv.
A version of this article first appeared on Medscape.com.
MONTREAL – from a naturally HIV-resistant donor, U.S. researchers announced at a meeting of the International AIDS Society.
The man received the transplant nearly 3.5 years ago. Since discontinuation of antiretroviral therapy (ART) more than 17 months ago, he has shown no evidence of HIV-1 RNA rebound and no detectable HIV-1 DNA, reported lead investigator Jana K. Dickter, MD, associate clinical professor in the division of infectious diseases at City of Hope, a Duarte, Calif.–based stem cell transplantation center for patients with blood cancers and patients with HIV/blood cancer.
Known as the City of Hope (COH) patient, he is different from the three previously reported patients in that “he was the oldest person to successfully undergo a stem cell transplant with HIV and leukemia and then achieve remission from both conditions,” Dr. Dickter said during a press briefing for the meeting. “He has been living with HIV the longest of any of the patients to date – more than 31 years prior to transplant – and he had also received the least immunosuppressive preparative regimen prior to transplant,” she added.
She said that, like the three previous patients, known as the Berlin, London, and New York patients, the COH patient received a transplant from a donor with natural resistance to HIV because of a rare CCR5-delta 32 mutation.
Dr. Dickter and her coinvestigators used the term “remission” but went further, suggesting that an “HIV cure is feasible” after transplant, given this and the previous cases.
“It’s a bit early to say the patient is cured, but they are clearly in remission,” said Sharon Lewin, MD, president-elect of the International AIDS Society, which runs the meeting. Nevertheless, Dr. Lewin, professor of medicine at the University of Melbourne and director of the Peter Doherty Institute for Infection and Immunity, in Melbourne, acknowledged that cure is “very likely.”
“Two of the previously reported patients have been off ART for long periods of time – Berlin, 12 years (until Timothy’s death in 2020); London, 4 years – and both had far more extensive investigations to try and find intact virus, including very large blood draws, tissue biopsies, etc. For the New York and now this COH patient, the duration off ART has been much shorter. ... But given the prior cases, it is very likely that the New York and COH patients are indeed cured. But I think it’s too early to make that call, hence my preference to use the word, ‘remission,’ “ she told this news organization.
“Although a transplant is not an option for most people with HIV, these cases are still interesting, still inspiring, and help illuminate the search for a cure,” she added.
Dr. Dickter acknowledged that the complexity of stem cell transplant procedures and their potential for significant side effects make them unsuitable as treatment options for most people with HIV, although she said the COH case is evidence that some HIV patients with blood cancers may not need such intensive pretransplant conditioning regimens.
The COH patient received a reduced-intensity fludarabine and melphalan regimen that had been designed at Dr. Dickter’s center “for older and less fit patients to make transplantation more tolerable,” she said. In addition, the graft-vs.-host disease prophylaxis that the COH patient received included only tacrolimus and sirolimus, whereas the previous patients received additional immunosuppressive therapies, and some also had undergone total body irradiation.
Dr. Dickter has disclosed no relevant financial relationships. Dr. Lewin has relationships with AbbVie, BMS, Esfam, Genentech, Gilead, Immunocore, Merck, Vaxxinity, and Viiv.
A version of this article first appeared on Medscape.com.
MONTREAL – from a naturally HIV-resistant donor, U.S. researchers announced at a meeting of the International AIDS Society.
The man received the transplant nearly 3.5 years ago. Since discontinuation of antiretroviral therapy (ART) more than 17 months ago, he has shown no evidence of HIV-1 RNA rebound and no detectable HIV-1 DNA, reported lead investigator Jana K. Dickter, MD, associate clinical professor in the division of infectious diseases at City of Hope, a Duarte, Calif.–based stem cell transplantation center for patients with blood cancers and patients with HIV/blood cancer.
Known as the City of Hope (COH) patient, he is different from the three previously reported patients in that “he was the oldest person to successfully undergo a stem cell transplant with HIV and leukemia and then achieve remission from both conditions,” Dr. Dickter said during a press briefing for the meeting. “He has been living with HIV the longest of any of the patients to date – more than 31 years prior to transplant – and he had also received the least immunosuppressive preparative regimen prior to transplant,” she added.
She said that, like the three previous patients, known as the Berlin, London, and New York patients, the COH patient received a transplant from a donor with natural resistance to HIV because of a rare CCR5-delta 32 mutation.
Dr. Dickter and her coinvestigators used the term “remission” but went further, suggesting that an “HIV cure is feasible” after transplant, given this and the previous cases.
“It’s a bit early to say the patient is cured, but they are clearly in remission,” said Sharon Lewin, MD, president-elect of the International AIDS Society, which runs the meeting. Nevertheless, Dr. Lewin, professor of medicine at the University of Melbourne and director of the Peter Doherty Institute for Infection and Immunity, in Melbourne, acknowledged that cure is “very likely.”
“Two of the previously reported patients have been off ART for long periods of time – Berlin, 12 years (until Timothy’s death in 2020); London, 4 years – and both had far more extensive investigations to try and find intact virus, including very large blood draws, tissue biopsies, etc. For the New York and now this COH patient, the duration off ART has been much shorter. ... But given the prior cases, it is very likely that the New York and COH patients are indeed cured. But I think it’s too early to make that call, hence my preference to use the word, ‘remission,’ “ she told this news organization.
“Although a transplant is not an option for most people with HIV, these cases are still interesting, still inspiring, and help illuminate the search for a cure,” she added.
Dr. Dickter acknowledged that the complexity of stem cell transplant procedures and their potential for significant side effects make them unsuitable as treatment options for most people with HIV, although she said the COH case is evidence that some HIV patients with blood cancers may not need such intensive pretransplant conditioning regimens.
The COH patient received a reduced-intensity fludarabine and melphalan regimen that had been designed at Dr. Dickter’s center “for older and less fit patients to make transplantation more tolerable,” she said. In addition, the graft-vs.-host disease prophylaxis that the COH patient received included only tacrolimus and sirolimus, whereas the previous patients received additional immunosuppressive therapies, and some also had undergone total body irradiation.
Dr. Dickter has disclosed no relevant financial relationships. Dr. Lewin has relationships with AbbVie, BMS, Esfam, Genentech, Gilead, Immunocore, Merck, Vaxxinity, and Viiv.
A version of this article first appeared on Medscape.com.
AT AIDS 2022
Coming to a pill near you: The exercise molecule
Exercise in a pill? Sign us up
You just got home from a long shift and you know you should go to the gym, but the bed is calling and you just answered. We know sometimes we have to make sacrifices in the name of fitness, but there just aren’t enough hours in the day. Unless our prayers have been answered. There could be a pill that has the benefits of working out without having to work out.
In a study published in Nature, investigators reported that they have identified a molecule made during exercise and used it on mice, which took in less food after being given the pill, which may open doors to understanding how exercise affects hunger.
In the first part of the study, the researchers found the molecule, known as Lac-Phe – which is synthesized from lactate and phenylalanine – in the blood plasma of mice after they had run on a treadmill.
The investigators then gave a Lac-Phe supplement to mice on high-fat diets and found that their food intake was about 50% of what other mice were eating. The supplement also improved their glucose tolerance.
Because the research also found Lac-Phe in humans who exercised, they hope that this pill will be in our future. “Our next steps include finding more details about how Lac-Phe mediates its effects in the body, including the brain,” Yong Xu, MD, of Baylor College of Medicine, Houston, said in a written statement. “Our goal is to learn to modulate this exercise pathway for therapeutic interventions.”
As always, we are rooting for you, science!
Gonorrhea and grandparents: A match made in prehistoric heaven
*Editorial note: LOTME takes no responsibility for any unfortunate imagery the reader may have experienced from the above headline.
Old people are the greatest. Back pains, cognitive decline, aches in all the diodes down your left side, there’s nothing quite like your golden years. Notably, however, humans are one of the few animals who experience true old age, as most creatures are adapted to maximize reproductive potential. As such, living past menopause is rare in the animal kingdom.
This is where the “grandmother hypothesis” comes in: Back in Ye Olde Stone Age, women who lived into old age could provide child care for younger women, because human babies require a lot more time and attention than other animal offspring. But how did humans end up living so long? Enter a group of Californian researchers, who believe they have an answer. It was gonorrhea.
When compared with the chimpanzee genome (as well as with Neanderthals and Denisovans, our closest ancestors), humans have a unique mutated version of the CD33 gene that lacks a sugar-binding site; the standard version uses the sugar-binding site to protect against autoimmune response in the body, but that same site actually suppresses the brain’s ability to clear away damaged brain cells and amyloid, which eventually leads to diseases like dementia. The mutated version allows microglia (brain immune cells) to attack and clear out this unwanted material. People with higher levels of this mutated CD33 variant actually have higher protection against Alzheimer’s.
Interestingly, gonorrhea bacteria are coated in the same sugar that standard CD33 receptors bind to, thus allowing them to bypass the body’s immune system. According to the researchers, the mutated CD33 version likely emerged as a protection against gonorrhea, depriving the bacteria of their “molecular mimicry” abilities. In one of life’s happy accidents, it turned out this mutation also protects against age-related diseases, thus allowing humans with the mutation to live longer. Obviously, this was a good thing, and we ran with it until the modern day. Now we have senior citizens climbing Everest, and all our politicians keep on politicking into their 70s and 80s ... well, everything has its drawbacks.
Parents raise a glass to children’s food addiction
There can be something pretty addicting about processed foods. Have you ever eaten just one french fry? Or taken just one cookie? If so, your willpower is incredible. For many of us, it can be a struggle to stop.
A recent study from the University of Michigan, which considered the existence of an eating phenotype, suggests our parents’ habits could be to blame.
By administering a series of questionnaires that inquired about food addiction, alcohol use disorders, cannabis use disorder, nicotine/e-cigarette dependence, and their family tree, investigators found that participants with a “paternal history of problematic alcohol use” had higher risk of food addiction but not obesity.
Apparently about one in five people display a clinically significant addiction to highly processed foods. It was noted that foods like ice cream, pizza, and french fries have high amounts of refined carbs and fats, which could trigger an addictive response.
Lindzey Hoover, a graduate student at the university who was the study’s lead author, noted that living in an environment where these foods are cheap and accessible can be really challenging for those with a family history of addiction. The investigators suggested that public health approaches, like restriction of other substances and marketing to kids, should be put in place for highly processed foods.
Maybe french fries should come with a warning label.
A prescription for America’s traffic problems
Nostalgia is a funny thing. Do you ever feel nostalgic about things that really weren’t very pleasant in the first place? Take, for instance, the morning commute. Here in the Washington area, more than 2 years into the COVID era, the traffic is still not what it used to be … and we kind of miss it.
Nah, not really. That was just a way to get everyone thinking about driving, because AAA has something of an explanation for the situation out there on the highways and byways of America. It’s drugs. No, not those kinds of drugs. This time it’s prescription drugs that are the problem. Well, part of the problem, anyway.
AAA did a survey last summer and found that nearly 50% of drivers “used one or more potentially impairing medications in the past 30 days. … The proportion of those choosing to drive is higher among those taking multiple medications.” How much higher? More than 63% of those with two or more prescriptions were driving within 2 hours of taking at least one of those meds, as were 71% of those taking three or more.
The 2,657 respondents also were asked about the types of potentially impairing drugs they were taking: 61% of those using antidepressants had been on the road within 2 hours of use at least once in the past 30 days, as had 73% of those taking an amphetamine, AAA said.
So there you have it. That guy in the BMW who’s been tailgating you for the last 3 miles? He may be a jerk, but there’s a good chance he’s a jerk with a prescription … or two … or three.
Exercise in a pill? Sign us up
You just got home from a long shift and you know you should go to the gym, but the bed is calling and you just answered. We know sometimes we have to make sacrifices in the name of fitness, but there just aren’t enough hours in the day. Unless our prayers have been answered. There could be a pill that has the benefits of working out without having to work out.
In a study published in Nature, investigators reported that they have identified a molecule made during exercise and used it on mice, which took in less food after being given the pill, which may open doors to understanding how exercise affects hunger.
In the first part of the study, the researchers found the molecule, known as Lac-Phe – which is synthesized from lactate and phenylalanine – in the blood plasma of mice after they had run on a treadmill.
The investigators then gave a Lac-Phe supplement to mice on high-fat diets and found that their food intake was about 50% of what other mice were eating. The supplement also improved their glucose tolerance.
Because the research also found Lac-Phe in humans who exercised, they hope that this pill will be in our future. “Our next steps include finding more details about how Lac-Phe mediates its effects in the body, including the brain,” Yong Xu, MD, of Baylor College of Medicine, Houston, said in a written statement. “Our goal is to learn to modulate this exercise pathway for therapeutic interventions.”
As always, we are rooting for you, science!
Gonorrhea and grandparents: A match made in prehistoric heaven
*Editorial note: LOTME takes no responsibility for any unfortunate imagery the reader may have experienced from the above headline.
Old people are the greatest. Back pains, cognitive decline, aches in all the diodes down your left side, there’s nothing quite like your golden years. Notably, however, humans are one of the few animals who experience true old age, as most creatures are adapted to maximize reproductive potential. As such, living past menopause is rare in the animal kingdom.
This is where the “grandmother hypothesis” comes in: Back in Ye Olde Stone Age, women who lived into old age could provide child care for younger women, because human babies require a lot more time and attention than other animal offspring. But how did humans end up living so long? Enter a group of Californian researchers, who believe they have an answer. It was gonorrhea.
When compared with the chimpanzee genome (as well as with Neanderthals and Denisovans, our closest ancestors), humans have a unique mutated version of the CD33 gene that lacks a sugar-binding site; the standard version uses the sugar-binding site to protect against autoimmune response in the body, but that same site actually suppresses the brain’s ability to clear away damaged brain cells and amyloid, which eventually leads to diseases like dementia. The mutated version allows microglia (brain immune cells) to attack and clear out this unwanted material. People with higher levels of this mutated CD33 variant actually have higher protection against Alzheimer’s.
Interestingly, gonorrhea bacteria are coated in the same sugar that standard CD33 receptors bind to, thus allowing them to bypass the body’s immune system. According to the researchers, the mutated CD33 version likely emerged as a protection against gonorrhea, depriving the bacteria of their “molecular mimicry” abilities. In one of life’s happy accidents, it turned out this mutation also protects against age-related diseases, thus allowing humans with the mutation to live longer. Obviously, this was a good thing, and we ran with it until the modern day. Now we have senior citizens climbing Everest, and all our politicians keep on politicking into their 70s and 80s ... well, everything has its drawbacks.
Parents raise a glass to children’s food addiction
There can be something pretty addicting about processed foods. Have you ever eaten just one french fry? Or taken just one cookie? If so, your willpower is incredible. For many of us, it can be a struggle to stop.
A recent study from the University of Michigan, which considered the existence of an eating phenotype, suggests our parents’ habits could be to blame.
By administering a series of questionnaires that inquired about food addiction, alcohol use disorders, cannabis use disorder, nicotine/e-cigarette dependence, and their family tree, investigators found that participants with a “paternal history of problematic alcohol use” had higher risk of food addiction but not obesity.
Apparently about one in five people display a clinically significant addiction to highly processed foods. It was noted that foods like ice cream, pizza, and french fries have high amounts of refined carbs and fats, which could trigger an addictive response.
Lindzey Hoover, a graduate student at the university who was the study’s lead author, noted that living in an environment where these foods are cheap and accessible can be really challenging for those with a family history of addiction. The investigators suggested that public health approaches, like restriction of other substances and marketing to kids, should be put in place for highly processed foods.
Maybe french fries should come with a warning label.
A prescription for America’s traffic problems
Nostalgia is a funny thing. Do you ever feel nostalgic about things that really weren’t very pleasant in the first place? Take, for instance, the morning commute. Here in the Washington area, more than 2 years into the COVID era, the traffic is still not what it used to be … and we kind of miss it.
Nah, not really. That was just a way to get everyone thinking about driving, because AAA has something of an explanation for the situation out there on the highways and byways of America. It’s drugs. No, not those kinds of drugs. This time it’s prescription drugs that are the problem. Well, part of the problem, anyway.
AAA did a survey last summer and found that nearly 50% of drivers “used one or more potentially impairing medications in the past 30 days. … The proportion of those choosing to drive is higher among those taking multiple medications.” How much higher? More than 63% of those with two or more prescriptions were driving within 2 hours of taking at least one of those meds, as were 71% of those taking three or more.
The 2,657 respondents also were asked about the types of potentially impairing drugs they were taking: 61% of those using antidepressants had been on the road within 2 hours of use at least once in the past 30 days, as had 73% of those taking an amphetamine, AAA said.
So there you have it. That guy in the BMW who’s been tailgating you for the last 3 miles? He may be a jerk, but there’s a good chance he’s a jerk with a prescription … or two … or three.
Exercise in a pill? Sign us up
You just got home from a long shift and you know you should go to the gym, but the bed is calling and you just answered. We know sometimes we have to make sacrifices in the name of fitness, but there just aren’t enough hours in the day. Unless our prayers have been answered. There could be a pill that has the benefits of working out without having to work out.
In a study published in Nature, investigators reported that they have identified a molecule made during exercise and used it on mice, which took in less food after being given the pill, which may open doors to understanding how exercise affects hunger.
In the first part of the study, the researchers found the molecule, known as Lac-Phe – which is synthesized from lactate and phenylalanine – in the blood plasma of mice after they had run on a treadmill.
The investigators then gave a Lac-Phe supplement to mice on high-fat diets and found that their food intake was about 50% of what other mice were eating. The supplement also improved their glucose tolerance.
Because the research also found Lac-Phe in humans who exercised, they hope that this pill will be in our future. “Our next steps include finding more details about how Lac-Phe mediates its effects in the body, including the brain,” Yong Xu, MD, of Baylor College of Medicine, Houston, said in a written statement. “Our goal is to learn to modulate this exercise pathway for therapeutic interventions.”
As always, we are rooting for you, science!
Gonorrhea and grandparents: A match made in prehistoric heaven
*Editorial note: LOTME takes no responsibility for any unfortunate imagery the reader may have experienced from the above headline.
Old people are the greatest. Back pains, cognitive decline, aches in all the diodes down your left side, there’s nothing quite like your golden years. Notably, however, humans are one of the few animals who experience true old age, as most creatures are adapted to maximize reproductive potential. As such, living past menopause is rare in the animal kingdom.
This is where the “grandmother hypothesis” comes in: Back in Ye Olde Stone Age, women who lived into old age could provide child care for younger women, because human babies require a lot more time and attention than other animal offspring. But how did humans end up living so long? Enter a group of Californian researchers, who believe they have an answer. It was gonorrhea.
When compared with the chimpanzee genome (as well as with Neanderthals and Denisovans, our closest ancestors), humans have a unique mutated version of the CD33 gene that lacks a sugar-binding site; the standard version uses the sugar-binding site to protect against autoimmune response in the body, but that same site actually suppresses the brain’s ability to clear away damaged brain cells and amyloid, which eventually leads to diseases like dementia. The mutated version allows microglia (brain immune cells) to attack and clear out this unwanted material. People with higher levels of this mutated CD33 variant actually have higher protection against Alzheimer’s.
Interestingly, gonorrhea bacteria are coated in the same sugar that standard CD33 receptors bind to, thus allowing them to bypass the body’s immune system. According to the researchers, the mutated CD33 version likely emerged as a protection against gonorrhea, depriving the bacteria of their “molecular mimicry” abilities. In one of life’s happy accidents, it turned out this mutation also protects against age-related diseases, thus allowing humans with the mutation to live longer. Obviously, this was a good thing, and we ran with it until the modern day. Now we have senior citizens climbing Everest, and all our politicians keep on politicking into their 70s and 80s ... well, everything has its drawbacks.
Parents raise a glass to children’s food addiction
There can be something pretty addicting about processed foods. Have you ever eaten just one french fry? Or taken just one cookie? If so, your willpower is incredible. For many of us, it can be a struggle to stop.
A recent study from the University of Michigan, which considered the existence of an eating phenotype, suggests our parents’ habits could be to blame.
By administering a series of questionnaires that inquired about food addiction, alcohol use disorders, cannabis use disorder, nicotine/e-cigarette dependence, and their family tree, investigators found that participants with a “paternal history of problematic alcohol use” had higher risk of food addiction but not obesity.
Apparently about one in five people display a clinically significant addiction to highly processed foods. It was noted that foods like ice cream, pizza, and french fries have high amounts of refined carbs and fats, which could trigger an addictive response.
Lindzey Hoover, a graduate student at the university who was the study’s lead author, noted that living in an environment where these foods are cheap and accessible can be really challenging for those with a family history of addiction. The investigators suggested that public health approaches, like restriction of other substances and marketing to kids, should be put in place for highly processed foods.
Maybe french fries should come with a warning label.
A prescription for America’s traffic problems
Nostalgia is a funny thing. Do you ever feel nostalgic about things that really weren’t very pleasant in the first place? Take, for instance, the morning commute. Here in the Washington area, more than 2 years into the COVID era, the traffic is still not what it used to be … and we kind of miss it.
Nah, not really. That was just a way to get everyone thinking about driving, because AAA has something of an explanation for the situation out there on the highways and byways of America. It’s drugs. No, not those kinds of drugs. This time it’s prescription drugs that are the problem. Well, part of the problem, anyway.
AAA did a survey last summer and found that nearly 50% of drivers “used one or more potentially impairing medications in the past 30 days. … The proportion of those choosing to drive is higher among those taking multiple medications.” How much higher? More than 63% of those with two or more prescriptions were driving within 2 hours of taking at least one of those meds, as were 71% of those taking three or more.
The 2,657 respondents also were asked about the types of potentially impairing drugs they were taking: 61% of those using antidepressants had been on the road within 2 hours of use at least once in the past 30 days, as had 73% of those taking an amphetamine, AAA said.
So there you have it. That guy in the BMW who’s been tailgating you for the last 3 miles? He may be a jerk, but there’s a good chance he’s a jerk with a prescription … or two … or three.
Scientists aim to combat COVID with a shot in the nose
Scientists seeking to stay ahead of an evolving SARS-Cov-2 virus are looking at new strategies, including developing intranasal vaccines, according to speakers at a conference on July 26.
inviting researchers to provide a public update on efforts to try to keep ahead of SARS-CoV-2.
Scientists and federal officials are looking to build on the successes seen in developing the original crop of COVID vaccines, which were authorized for use in the United States less than a year after the pandemic took hold.
But emerging variants are eroding these gains. For months now, officials at the Centers for Disease Control and Prevention and Food and Drug Administration have been keeping an eye on how the level of effectiveness of COVID vaccines has waned during the rise of the Omicron strain. And there’s continual concern about how SARS-CoV-2 might evolve over time.
“Our vaccines are terrific,” Ashish K. Jha, MD, the White House’s COVID-19 response coordinator, said at the summit. “[But] we have to do better.”
Among the approaches being considered are vaccines that would be applied intranasally, with the idea that this might be able to boost the immune response to SARS-CoV-2.
At the summit, Akiko Iwasaki, PhD, of Yale University, New Haven, Conn., said the intranasal approach might be helpful in preventing transmission as well as reducing the burden of illness for those who are infected with SARS-CoV-2.
“We’re stopping the virus from spreading right at the border,” Dr. Iwasaki said at the summit. “This is akin to putting a guard outside of the house in order to patrol for invaders compared to putting the guards in the hallway of the building in the hope that they capture the invader.”
Dr. Iwasaki is one of the founders of Xanadu Bio, a private company created last year to focus on ways to kill SARS-CoV-2 in the nasosinus before it spreads deeper into the respiratory tract. In an editorial in Science Immunology, Dr. Iwasaki and Eric J. Topol, MD, director of the Scripps Research Translational Institute, urged greater federal investment in this approach to fighting SARS-CoV-2. (Dr. Topol is editor-in-chief of Medscape.)
Titled “Operation Nasal Vaccine – Lightning speed to counter COVID-19,” their editorial noted the “unprecedented success” seen in the rapid development of the first two mRNA shots. Dr. Iwasaki and Dr. Topol noted that these victories had been “fueled by the $10 billion governmental investment in Operation Warp Speed.
“During the first year of the pandemic, meaningful evolution of the virus was slow-paced, without any functional consequences, but since that time we have seen a succession of important variants of concern, with increasing transmissibility and immune evasion, culminating in the Omicron lineages,” wrote Dr. Iwasaki and Dr. Topol.
Recent developments have “spotlighted the possibility of nasal vaccines, with their allure for achieving mucosal immunity, complementing, and likely bolstering the circulating immunity achieved via intramuscular shots,” they added.
An early setback
Scientists at the National Institutes of Health and the Biomedical Advanced Research and Development Authority (BARDA) have for some time been looking to vet an array of next-generation vaccine concepts, including ones that trigger mucosal immunity, the Washington Post reported in April.
At the summit on July 26, several participants, including Dr. Jha, stressed the role that public-private partnerships were key to the rapid development of the initial COVID vaccines. They said continued U.S. government support will be needed to make advances in this field.
One of the presenters, Biao He, PhD, founder and president of CyanVac and Blue Lake Biotechnology, spoke of the federal support that his efforts have received over the years to develop intranasal vaccines. His Georgia-based firm already has an experimental intranasal vaccine candidate, CVXGA1-001, in phase 1 testing (NCT04954287).
The CVXGA-001 builds on technology already used in a veterinary product, an intranasal vaccine long used to prevent kennel cough in dogs, he said at the summit.
The emerging field of experimental intranasal COVID vaccines already has had at least one setback.
The biotech firm Altimmune in June 2021 announced that it would discontinue development of its experimental intranasal AdCOVID vaccine following disappointing phase 1 results. The vaccine appeared to be well tolerated in the test, but the immunogenicity data demonstrated lower than expected results in healthy volunteers, especially in light of the responses seen to already cleared vaccines, Altimmune said in a release.
In the statement, Scot Roberts, PhD, chief scientific officer at Altimmune, noted that the study participants lacked immunity from prior infection or vaccination. “We believe that prior immunity in humans may be important for a robust immune response to intranasal dosing with AdCOVID,” he said.
At the summit, Marty Moore, PhD, cofounder and chief scientific officer for Redwood City, Calif.–based Meissa Vaccines, noted the challenges that remain ahead for intranasal COVID vaccines, while also highlighting what he sees as the potential of this approach.
Meissa also has advanced an experimental intranasal COVID vaccine as far as phase 1 testing (NCT04798001).
“No one here today can tell you that mucosal COVID vaccines work. We’re not there yet. We need clinical efficacy data to answer that question,” Dr. Moore said.
But there’s a potential for a “knockout blow to COVID, a transmission-blocking vaccine” from the intranasal approach, he said.
“The virus is mutating faster than our ability to manage vaccines and not enough people are getting boosters. These injectable vaccines do a great job of preventing severe disease, but they do little to prevent infection” from spreading, Dr. Moore said.
A version of this article first appeared on Medscape.com.
Scientists seeking to stay ahead of an evolving SARS-Cov-2 virus are looking at new strategies, including developing intranasal vaccines, according to speakers at a conference on July 26.
inviting researchers to provide a public update on efforts to try to keep ahead of SARS-CoV-2.
Scientists and federal officials are looking to build on the successes seen in developing the original crop of COVID vaccines, which were authorized for use in the United States less than a year after the pandemic took hold.
But emerging variants are eroding these gains. For months now, officials at the Centers for Disease Control and Prevention and Food and Drug Administration have been keeping an eye on how the level of effectiveness of COVID vaccines has waned during the rise of the Omicron strain. And there’s continual concern about how SARS-CoV-2 might evolve over time.
“Our vaccines are terrific,” Ashish K. Jha, MD, the White House’s COVID-19 response coordinator, said at the summit. “[But] we have to do better.”
Among the approaches being considered are vaccines that would be applied intranasally, with the idea that this might be able to boost the immune response to SARS-CoV-2.
At the summit, Akiko Iwasaki, PhD, of Yale University, New Haven, Conn., said the intranasal approach might be helpful in preventing transmission as well as reducing the burden of illness for those who are infected with SARS-CoV-2.
“We’re stopping the virus from spreading right at the border,” Dr. Iwasaki said at the summit. “This is akin to putting a guard outside of the house in order to patrol for invaders compared to putting the guards in the hallway of the building in the hope that they capture the invader.”
Dr. Iwasaki is one of the founders of Xanadu Bio, a private company created last year to focus on ways to kill SARS-CoV-2 in the nasosinus before it spreads deeper into the respiratory tract. In an editorial in Science Immunology, Dr. Iwasaki and Eric J. Topol, MD, director of the Scripps Research Translational Institute, urged greater federal investment in this approach to fighting SARS-CoV-2. (Dr. Topol is editor-in-chief of Medscape.)
Titled “Operation Nasal Vaccine – Lightning speed to counter COVID-19,” their editorial noted the “unprecedented success” seen in the rapid development of the first two mRNA shots. Dr. Iwasaki and Dr. Topol noted that these victories had been “fueled by the $10 billion governmental investment in Operation Warp Speed.
“During the first year of the pandemic, meaningful evolution of the virus was slow-paced, without any functional consequences, but since that time we have seen a succession of important variants of concern, with increasing transmissibility and immune evasion, culminating in the Omicron lineages,” wrote Dr. Iwasaki and Dr. Topol.
Recent developments have “spotlighted the possibility of nasal vaccines, with their allure for achieving mucosal immunity, complementing, and likely bolstering the circulating immunity achieved via intramuscular shots,” they added.
An early setback
Scientists at the National Institutes of Health and the Biomedical Advanced Research and Development Authority (BARDA) have for some time been looking to vet an array of next-generation vaccine concepts, including ones that trigger mucosal immunity, the Washington Post reported in April.
At the summit on July 26, several participants, including Dr. Jha, stressed the role that public-private partnerships were key to the rapid development of the initial COVID vaccines. They said continued U.S. government support will be needed to make advances in this field.
One of the presenters, Biao He, PhD, founder and president of CyanVac and Blue Lake Biotechnology, spoke of the federal support that his efforts have received over the years to develop intranasal vaccines. His Georgia-based firm already has an experimental intranasal vaccine candidate, CVXGA1-001, in phase 1 testing (NCT04954287).
The CVXGA-001 builds on technology already used in a veterinary product, an intranasal vaccine long used to prevent kennel cough in dogs, he said at the summit.
The emerging field of experimental intranasal COVID vaccines already has had at least one setback.
The biotech firm Altimmune in June 2021 announced that it would discontinue development of its experimental intranasal AdCOVID vaccine following disappointing phase 1 results. The vaccine appeared to be well tolerated in the test, but the immunogenicity data demonstrated lower than expected results in healthy volunteers, especially in light of the responses seen to already cleared vaccines, Altimmune said in a release.
In the statement, Scot Roberts, PhD, chief scientific officer at Altimmune, noted that the study participants lacked immunity from prior infection or vaccination. “We believe that prior immunity in humans may be important for a robust immune response to intranasal dosing with AdCOVID,” he said.
At the summit, Marty Moore, PhD, cofounder and chief scientific officer for Redwood City, Calif.–based Meissa Vaccines, noted the challenges that remain ahead for intranasal COVID vaccines, while also highlighting what he sees as the potential of this approach.
Meissa also has advanced an experimental intranasal COVID vaccine as far as phase 1 testing (NCT04798001).
“No one here today can tell you that mucosal COVID vaccines work. We’re not there yet. We need clinical efficacy data to answer that question,” Dr. Moore said.
But there’s a potential for a “knockout blow to COVID, a transmission-blocking vaccine” from the intranasal approach, he said.
“The virus is mutating faster than our ability to manage vaccines and not enough people are getting boosters. These injectable vaccines do a great job of preventing severe disease, but they do little to prevent infection” from spreading, Dr. Moore said.
A version of this article first appeared on Medscape.com.
Scientists seeking to stay ahead of an evolving SARS-Cov-2 virus are looking at new strategies, including developing intranasal vaccines, according to speakers at a conference on July 26.
inviting researchers to provide a public update on efforts to try to keep ahead of SARS-CoV-2.
Scientists and federal officials are looking to build on the successes seen in developing the original crop of COVID vaccines, which were authorized for use in the United States less than a year after the pandemic took hold.
But emerging variants are eroding these gains. For months now, officials at the Centers for Disease Control and Prevention and Food and Drug Administration have been keeping an eye on how the level of effectiveness of COVID vaccines has waned during the rise of the Omicron strain. And there’s continual concern about how SARS-CoV-2 might evolve over time.
“Our vaccines are terrific,” Ashish K. Jha, MD, the White House’s COVID-19 response coordinator, said at the summit. “[But] we have to do better.”
Among the approaches being considered are vaccines that would be applied intranasally, with the idea that this might be able to boost the immune response to SARS-CoV-2.
At the summit, Akiko Iwasaki, PhD, of Yale University, New Haven, Conn., said the intranasal approach might be helpful in preventing transmission as well as reducing the burden of illness for those who are infected with SARS-CoV-2.
“We’re stopping the virus from spreading right at the border,” Dr. Iwasaki said at the summit. “This is akin to putting a guard outside of the house in order to patrol for invaders compared to putting the guards in the hallway of the building in the hope that they capture the invader.”
Dr. Iwasaki is one of the founders of Xanadu Bio, a private company created last year to focus on ways to kill SARS-CoV-2 in the nasosinus before it spreads deeper into the respiratory tract. In an editorial in Science Immunology, Dr. Iwasaki and Eric J. Topol, MD, director of the Scripps Research Translational Institute, urged greater federal investment in this approach to fighting SARS-CoV-2. (Dr. Topol is editor-in-chief of Medscape.)
Titled “Operation Nasal Vaccine – Lightning speed to counter COVID-19,” their editorial noted the “unprecedented success” seen in the rapid development of the first two mRNA shots. Dr. Iwasaki and Dr. Topol noted that these victories had been “fueled by the $10 billion governmental investment in Operation Warp Speed.
“During the first year of the pandemic, meaningful evolution of the virus was slow-paced, without any functional consequences, but since that time we have seen a succession of important variants of concern, with increasing transmissibility and immune evasion, culminating in the Omicron lineages,” wrote Dr. Iwasaki and Dr. Topol.
Recent developments have “spotlighted the possibility of nasal vaccines, with their allure for achieving mucosal immunity, complementing, and likely bolstering the circulating immunity achieved via intramuscular shots,” they added.
An early setback
Scientists at the National Institutes of Health and the Biomedical Advanced Research and Development Authority (BARDA) have for some time been looking to vet an array of next-generation vaccine concepts, including ones that trigger mucosal immunity, the Washington Post reported in April.
At the summit on July 26, several participants, including Dr. Jha, stressed the role that public-private partnerships were key to the rapid development of the initial COVID vaccines. They said continued U.S. government support will be needed to make advances in this field.
One of the presenters, Biao He, PhD, founder and president of CyanVac and Blue Lake Biotechnology, spoke of the federal support that his efforts have received over the years to develop intranasal vaccines. His Georgia-based firm already has an experimental intranasal vaccine candidate, CVXGA1-001, in phase 1 testing (NCT04954287).
The CVXGA-001 builds on technology already used in a veterinary product, an intranasal vaccine long used to prevent kennel cough in dogs, he said at the summit.
The emerging field of experimental intranasal COVID vaccines already has had at least one setback.
The biotech firm Altimmune in June 2021 announced that it would discontinue development of its experimental intranasal AdCOVID vaccine following disappointing phase 1 results. The vaccine appeared to be well tolerated in the test, but the immunogenicity data demonstrated lower than expected results in healthy volunteers, especially in light of the responses seen to already cleared vaccines, Altimmune said in a release.
In the statement, Scot Roberts, PhD, chief scientific officer at Altimmune, noted that the study participants lacked immunity from prior infection or vaccination. “We believe that prior immunity in humans may be important for a robust immune response to intranasal dosing with AdCOVID,” he said.
At the summit, Marty Moore, PhD, cofounder and chief scientific officer for Redwood City, Calif.–based Meissa Vaccines, noted the challenges that remain ahead for intranasal COVID vaccines, while also highlighting what he sees as the potential of this approach.
Meissa also has advanced an experimental intranasal COVID vaccine as far as phase 1 testing (NCT04798001).
“No one here today can tell you that mucosal COVID vaccines work. We’re not there yet. We need clinical efficacy data to answer that question,” Dr. Moore said.
But there’s a potential for a “knockout blow to COVID, a transmission-blocking vaccine” from the intranasal approach, he said.
“The virus is mutating faster than our ability to manage vaccines and not enough people are getting boosters. These injectable vaccines do a great job of preventing severe disease, but they do little to prevent infection” from spreading, Dr. Moore said.
A version of this article first appeared on Medscape.com.
VA foster program helps older vets manage COVID challenges
Susan Snead used to live in an apartment complex for older adults. The complex had a nice dayroom, and neighbors would knock on her door every now and then to check in.
But despite not being lonely, Ms. Snead, 89, did live alone in downtown Charleston, S.C. Eventually, that became dangerous.
“I fell a few times,” she says. “I had to call somebody to come and get me up.”
Sometimes help would come from the apartment complex’s office. Sometimes it came with a police escort.
Over time, needing to make those calls became a burden. Making and keeping appointments with her doctor, something she had to do regularly, as she has diabetes, got harder, too.
“It kind of wore me out,” she says. “Like you’re going up a hill.”
As she was beginning to accept she could no longer live alone, Ms. Snead, an Air Force veteran, learned about a program run by the Department of Veterans Affairs called Medical Foster Home.
Caregivers help aging veterans with activities of daily living like bathing, cooking, making and getting to appointments, getting dressed, and taking daily medication.
Caregivers can take care of up to three residents in their home at a time. While most residents are veterans, caregivers sometimes care for non-veteran residents, such as a veteran’s spouse or a caregiver’s family member.
Veterans typically pay about $1,500 to $3,000 out-of-pocket per month for the service, depending on location.
According to the VA, the concept of medical foster homes has been around since 1999, when VA hospitals across the country began reaching out to people willing to provide live-in care for veterans. The option is led by local VA hospitals, which approve caregivers and provide administrative services. There are now 517 medical foster homes, the VA says.
Much like other residential care facilities, medical foster homes get regular inspections for safety, nutrition, and more.
In 2019, Ms. Snead signed up for the program. She expected to be cared for, but she found a sense of family with her caregiver, Wilhelmina Brown, and another veteran in the home.
Ms. Brown started taking care of people – but not necessarily veterans – in 1997 when her grandmother was unable to care for herself, she says.
“My grandmama carried me to church every Sunday, she carried me to the beach – everywhere she went, she took me with her,” Ms. Brown says. As her grandmother got older, “I said, ‘I’m going to take care of her in my home.’ ”
Caring for others must come from the heart, Ms. Brown says.
She cooks her residents’ meals three times a day with dietary restrictions in mind, washes their dishes, does their laundry, remembers birthdays, and plans little parties.
“That’s my family,” Ms. Brown says.
In 2020, the COVID-19 pandemic upended the world – but at the same time, it highlighted the advantages of the medical foster home model.
Home-based primary care keeps veterans out of nursing homes – something that became particularly important as COVID-19 hit nursing homes and long-term care facilities.
Caregivers in the system were also able to help veterans, often living in rural areas, pivot and adapt to telehealth during a time of crisis.
One study, published in the journal Geriatrics, set out to identify how medical foster homes were able to deliver safe, effective health care during the early stages of the pandemic.
Researchers interviewed 37 VA care providers at 16 rural medical foster home programs across the country. The interviews took place between December 2020 and February 2021. They found medical foster home caregivers, coordinators, and health care providers communicated to move office visits to the home, helped veterans navigate telehealth, advocated to get veterans vaccinated in-home, and relied on each other to fight social isolation.
Caregivers also adapted quickly to telehealth, according to Leah Haverhals, PhD, a health research scientist and communications director for the Seattle-Denver Center of Innovation for Veteran Centered and Value Driven Care, who led the study.
Most veterans in the foster home program are older and find new technology difficult to use.
Caregivers, coordinators, and health care providers were largely new to the technology, too.
While the study found that most veterans and caregivers preferred in-person care, they were able to work together to make the best of telehealth.
“That speaks to the nature of the care being given, being able to pivot in a crisis like that,” Dr. Haverhals says.
If caregivers didn’t already have computers or telehealth-compatible devices, the VA provided iPads that would connect to the internet using cellular signals. According to the study, this helped to overcome connectivity issues that may have caused problems in rural areas.
Ms. Snead says Ms. Brown helped a lot with her telehealth calls.
“If we had to do things over the phone or with video, she was able to set that up to work with the person on the other end. She knows a lot about that stuff – about computers and things like that,” Ms. Snead says, adding that she hadn’t worked with computers since retirement in 1998.
Telehealth helped health care providers identify infections and quickly prescribe antibiotics to veterans in rural areas and provide other care that was more safely delivered in private homes.
“The findings from our study highlighted that when working together for the common goal of keeping vulnerable populations like veterans in MFHs [medical foster homes] safe during times of crisis, adaptation and collaboration facilitated the ongoing provision of high-quality care,” Dr. Haverhals’s group wrote. “Such collaboration has been shown to be critical in recent research in the United States on supporting older adults during the pandemic.”
Cari Levy, MD, PhD, a professor at the University of Colorado at Denver, Aurora, and a co-author of the study, specializes in palliative and telenursing home care for the VA.
Dr. Levy, who has worked for the VA for about 20 years, says how medical foster homes provided care during the pandemic carries lessons for civilian clinics. One of the most important lessons, she says, is that medical professionals will need to provide more care where people are, especially in populations that are too sick to get to the clinic.
“For years, there was all this hope that telehealth would expand,” but it took a pandemic to authorize approval from federal agencies to explode, she says. “I shudder to think what would have happened if we didn’t have telehealth. Fortunately, it was the right time to be able to flip a switch.”
Crisis aside, Dr. Levy says her dream would be for health care providers to do more home-based care. The model allows people to preserve the relational aspects of medicine, which can counteract a lot of the moral injury and burnout in the field, she says, adding:
“I see this as the kind of medicine many people intended to do when they got into medicine.”
A version of this article first appeared on WebMD.com.
Susan Snead used to live in an apartment complex for older adults. The complex had a nice dayroom, and neighbors would knock on her door every now and then to check in.
But despite not being lonely, Ms. Snead, 89, did live alone in downtown Charleston, S.C. Eventually, that became dangerous.
“I fell a few times,” she says. “I had to call somebody to come and get me up.”
Sometimes help would come from the apartment complex’s office. Sometimes it came with a police escort.
Over time, needing to make those calls became a burden. Making and keeping appointments with her doctor, something she had to do regularly, as she has diabetes, got harder, too.
“It kind of wore me out,” she says. “Like you’re going up a hill.”
As she was beginning to accept she could no longer live alone, Ms. Snead, an Air Force veteran, learned about a program run by the Department of Veterans Affairs called Medical Foster Home.
Caregivers help aging veterans with activities of daily living like bathing, cooking, making and getting to appointments, getting dressed, and taking daily medication.
Caregivers can take care of up to three residents in their home at a time. While most residents are veterans, caregivers sometimes care for non-veteran residents, such as a veteran’s spouse or a caregiver’s family member.
Veterans typically pay about $1,500 to $3,000 out-of-pocket per month for the service, depending on location.
According to the VA, the concept of medical foster homes has been around since 1999, when VA hospitals across the country began reaching out to people willing to provide live-in care for veterans. The option is led by local VA hospitals, which approve caregivers and provide administrative services. There are now 517 medical foster homes, the VA says.
Much like other residential care facilities, medical foster homes get regular inspections for safety, nutrition, and more.
In 2019, Ms. Snead signed up for the program. She expected to be cared for, but she found a sense of family with her caregiver, Wilhelmina Brown, and another veteran in the home.
Ms. Brown started taking care of people – but not necessarily veterans – in 1997 when her grandmother was unable to care for herself, she says.
“My grandmama carried me to church every Sunday, she carried me to the beach – everywhere she went, she took me with her,” Ms. Brown says. As her grandmother got older, “I said, ‘I’m going to take care of her in my home.’ ”
Caring for others must come from the heart, Ms. Brown says.
She cooks her residents’ meals three times a day with dietary restrictions in mind, washes their dishes, does their laundry, remembers birthdays, and plans little parties.
“That’s my family,” Ms. Brown says.
In 2020, the COVID-19 pandemic upended the world – but at the same time, it highlighted the advantages of the medical foster home model.
Home-based primary care keeps veterans out of nursing homes – something that became particularly important as COVID-19 hit nursing homes and long-term care facilities.
Caregivers in the system were also able to help veterans, often living in rural areas, pivot and adapt to telehealth during a time of crisis.
One study, published in the journal Geriatrics, set out to identify how medical foster homes were able to deliver safe, effective health care during the early stages of the pandemic.
Researchers interviewed 37 VA care providers at 16 rural medical foster home programs across the country. The interviews took place between December 2020 and February 2021. They found medical foster home caregivers, coordinators, and health care providers communicated to move office visits to the home, helped veterans navigate telehealth, advocated to get veterans vaccinated in-home, and relied on each other to fight social isolation.
Caregivers also adapted quickly to telehealth, according to Leah Haverhals, PhD, a health research scientist and communications director for the Seattle-Denver Center of Innovation for Veteran Centered and Value Driven Care, who led the study.
Most veterans in the foster home program are older and find new technology difficult to use.
Caregivers, coordinators, and health care providers were largely new to the technology, too.
While the study found that most veterans and caregivers preferred in-person care, they were able to work together to make the best of telehealth.
“That speaks to the nature of the care being given, being able to pivot in a crisis like that,” Dr. Haverhals says.
If caregivers didn’t already have computers or telehealth-compatible devices, the VA provided iPads that would connect to the internet using cellular signals. According to the study, this helped to overcome connectivity issues that may have caused problems in rural areas.
Ms. Snead says Ms. Brown helped a lot with her telehealth calls.
“If we had to do things over the phone or with video, she was able to set that up to work with the person on the other end. She knows a lot about that stuff – about computers and things like that,” Ms. Snead says, adding that she hadn’t worked with computers since retirement in 1998.
Telehealth helped health care providers identify infections and quickly prescribe antibiotics to veterans in rural areas and provide other care that was more safely delivered in private homes.
“The findings from our study highlighted that when working together for the common goal of keeping vulnerable populations like veterans in MFHs [medical foster homes] safe during times of crisis, adaptation and collaboration facilitated the ongoing provision of high-quality care,” Dr. Haverhals’s group wrote. “Such collaboration has been shown to be critical in recent research in the United States on supporting older adults during the pandemic.”
Cari Levy, MD, PhD, a professor at the University of Colorado at Denver, Aurora, and a co-author of the study, specializes in palliative and telenursing home care for the VA.
Dr. Levy, who has worked for the VA for about 20 years, says how medical foster homes provided care during the pandemic carries lessons for civilian clinics. One of the most important lessons, she says, is that medical professionals will need to provide more care where people are, especially in populations that are too sick to get to the clinic.
“For years, there was all this hope that telehealth would expand,” but it took a pandemic to authorize approval from federal agencies to explode, she says. “I shudder to think what would have happened if we didn’t have telehealth. Fortunately, it was the right time to be able to flip a switch.”
Crisis aside, Dr. Levy says her dream would be for health care providers to do more home-based care. The model allows people to preserve the relational aspects of medicine, which can counteract a lot of the moral injury and burnout in the field, she says, adding:
“I see this as the kind of medicine many people intended to do when they got into medicine.”
A version of this article first appeared on WebMD.com.
Susan Snead used to live in an apartment complex for older adults. The complex had a nice dayroom, and neighbors would knock on her door every now and then to check in.
But despite not being lonely, Ms. Snead, 89, did live alone in downtown Charleston, S.C. Eventually, that became dangerous.
“I fell a few times,” she says. “I had to call somebody to come and get me up.”
Sometimes help would come from the apartment complex’s office. Sometimes it came with a police escort.
Over time, needing to make those calls became a burden. Making and keeping appointments with her doctor, something she had to do regularly, as she has diabetes, got harder, too.
“It kind of wore me out,” she says. “Like you’re going up a hill.”
As she was beginning to accept she could no longer live alone, Ms. Snead, an Air Force veteran, learned about a program run by the Department of Veterans Affairs called Medical Foster Home.
Caregivers help aging veterans with activities of daily living like bathing, cooking, making and getting to appointments, getting dressed, and taking daily medication.
Caregivers can take care of up to three residents in their home at a time. While most residents are veterans, caregivers sometimes care for non-veteran residents, such as a veteran’s spouse or a caregiver’s family member.
Veterans typically pay about $1,500 to $3,000 out-of-pocket per month for the service, depending on location.
According to the VA, the concept of medical foster homes has been around since 1999, when VA hospitals across the country began reaching out to people willing to provide live-in care for veterans. The option is led by local VA hospitals, which approve caregivers and provide administrative services. There are now 517 medical foster homes, the VA says.
Much like other residential care facilities, medical foster homes get regular inspections for safety, nutrition, and more.
In 2019, Ms. Snead signed up for the program. She expected to be cared for, but she found a sense of family with her caregiver, Wilhelmina Brown, and another veteran in the home.
Ms. Brown started taking care of people – but not necessarily veterans – in 1997 when her grandmother was unable to care for herself, she says.
“My grandmama carried me to church every Sunday, she carried me to the beach – everywhere she went, she took me with her,” Ms. Brown says. As her grandmother got older, “I said, ‘I’m going to take care of her in my home.’ ”
Caring for others must come from the heart, Ms. Brown says.
She cooks her residents’ meals three times a day with dietary restrictions in mind, washes their dishes, does their laundry, remembers birthdays, and plans little parties.
“That’s my family,” Ms. Brown says.
In 2020, the COVID-19 pandemic upended the world – but at the same time, it highlighted the advantages of the medical foster home model.
Home-based primary care keeps veterans out of nursing homes – something that became particularly important as COVID-19 hit nursing homes and long-term care facilities.
Caregivers in the system were also able to help veterans, often living in rural areas, pivot and adapt to telehealth during a time of crisis.
One study, published in the journal Geriatrics, set out to identify how medical foster homes were able to deliver safe, effective health care during the early stages of the pandemic.
Researchers interviewed 37 VA care providers at 16 rural medical foster home programs across the country. The interviews took place between December 2020 and February 2021. They found medical foster home caregivers, coordinators, and health care providers communicated to move office visits to the home, helped veterans navigate telehealth, advocated to get veterans vaccinated in-home, and relied on each other to fight social isolation.
Caregivers also adapted quickly to telehealth, according to Leah Haverhals, PhD, a health research scientist and communications director for the Seattle-Denver Center of Innovation for Veteran Centered and Value Driven Care, who led the study.
Most veterans in the foster home program are older and find new technology difficult to use.
Caregivers, coordinators, and health care providers were largely new to the technology, too.
While the study found that most veterans and caregivers preferred in-person care, they were able to work together to make the best of telehealth.
“That speaks to the nature of the care being given, being able to pivot in a crisis like that,” Dr. Haverhals says.
If caregivers didn’t already have computers or telehealth-compatible devices, the VA provided iPads that would connect to the internet using cellular signals. According to the study, this helped to overcome connectivity issues that may have caused problems in rural areas.
Ms. Snead says Ms. Brown helped a lot with her telehealth calls.
“If we had to do things over the phone or with video, she was able to set that up to work with the person on the other end. She knows a lot about that stuff – about computers and things like that,” Ms. Snead says, adding that she hadn’t worked with computers since retirement in 1998.
Telehealth helped health care providers identify infections and quickly prescribe antibiotics to veterans in rural areas and provide other care that was more safely delivered in private homes.
“The findings from our study highlighted that when working together for the common goal of keeping vulnerable populations like veterans in MFHs [medical foster homes] safe during times of crisis, adaptation and collaboration facilitated the ongoing provision of high-quality care,” Dr. Haverhals’s group wrote. “Such collaboration has been shown to be critical in recent research in the United States on supporting older adults during the pandemic.”
Cari Levy, MD, PhD, a professor at the University of Colorado at Denver, Aurora, and a co-author of the study, specializes in palliative and telenursing home care for the VA.
Dr. Levy, who has worked for the VA for about 20 years, says how medical foster homes provided care during the pandemic carries lessons for civilian clinics. One of the most important lessons, she says, is that medical professionals will need to provide more care where people are, especially in populations that are too sick to get to the clinic.
“For years, there was all this hope that telehealth would expand,” but it took a pandemic to authorize approval from federal agencies to explode, she says. “I shudder to think what would have happened if we didn’t have telehealth. Fortunately, it was the right time to be able to flip a switch.”
Crisis aside, Dr. Levy says her dream would be for health care providers to do more home-based care. The model allows people to preserve the relational aspects of medicine, which can counteract a lot of the moral injury and burnout in the field, she says, adding:
“I see this as the kind of medicine many people intended to do when they got into medicine.”
A version of this article first appeared on WebMD.com.
Children and COVID: Many parents see vaccine as the greater risk
New COVID-19 cases rose for the second week in a row as cumulative cases among U.S. children passed the 14-million mark, but a recent survey shows that more than half of parents believe that the vaccine is a greater risk to children under age 5 years than the virus.
In a Kaiser Family Foundation survey conducted July 7-17, 53% of parents with children aged 6 months to 5 years said that the vaccine is “a bigger risk to their child’s health than getting infected with COVID-19, compared to 44% who say getting infected is the bigger risk,” KFF reported July 26.
More than 4 out of 10 of respondents (43%) said that they will “definitely not” get their eligible children vaccinated, while only 7% said that their children had already received it and 10% said their children would get it as soon as possible, according to the KFF survey, which had an overall sample size of 1,847 adults, including an oversample of 471 parents of children under age 5.
Vaccine initiation has been slow in the first month since it was approved for the youngest children. Just 2.8% of all eligible children under age 5 had received an initial dose as of July 19, compared with first-month uptake figures of more than 18% for the 5- to 11-year-olds and 27% for those aged 12-15, based on data from the Centers for Disease Control and Prevention.
The current rates for vaccination in those aged 5 and older look like this: 70.2% of 12- to 17-year-olds have received at least one dose, versus 37.1% of those aged 5-11. Just over 60% of the older children were fully vaccinated as of July 19, as were 30.2% of the 5- to 11-year-olds, the CDC reported on its COVID Data Tracker.
Number of new cases hits 2-month high
Despite the vaccine, SARS-CoV-2 and its various mutations have continued with their summer travels. With 92,000 newly infected children added for the week of July 15-21, there have now been a total of 14,003,497 pediatric cases reported since the start of the pandemic, which works out to 18.6% of cases in all ages, the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID-19 report.
The 92,000 new cases represent an increase of almost 22% over the previous week and mark the highest 1-week count since May, when the total passed 100,000 for 2 consecutive weeks. More recently the trend had seemed more stable as weekly cases dropped twice and rose twice as the total hovered around 70,000, based on the data collected by the AAP and CHA from state and territorial health departments.
A different scenario has played out for emergency department visits and hospital admissions, which have risen steadily since the beginning of April. The admission rate for children aged 0-17, which was just 0.13 new patients per 100,000 population on April 11, was up to 0.44 per 100,000 on July 21. By comparison, the highest rate reached last year during the Delta surge was 0.47 per 100,000, based on CDC data.
The 7-day average of emergency dept. visits among the youngest age group, 0-11 years, shows the same general increase as hospital admissions, but the older children have diverged form that path (see graph). For those aged 12-15 and 16-17, hospitalizations started dropping in late May and into mid-June before climbing again, although more slowly than for the youngest group, the CDC data show.
The ED visit rate with diagnosed COVID among those aged 0-11, measured at 6.1% of all visits on July 19, is, in fact, considerably higher than at any time during the Delta surge last year, when it never passed 4.0%, although much lower than peak Omicron (14.1%). That 6.1% was also higher than any other age group on that day, adults included, the CDC said.
New COVID-19 cases rose for the second week in a row as cumulative cases among U.S. children passed the 14-million mark, but a recent survey shows that more than half of parents believe that the vaccine is a greater risk to children under age 5 years than the virus.
In a Kaiser Family Foundation survey conducted July 7-17, 53% of parents with children aged 6 months to 5 years said that the vaccine is “a bigger risk to their child’s health than getting infected with COVID-19, compared to 44% who say getting infected is the bigger risk,” KFF reported July 26.
More than 4 out of 10 of respondents (43%) said that they will “definitely not” get their eligible children vaccinated, while only 7% said that their children had already received it and 10% said their children would get it as soon as possible, according to the KFF survey, which had an overall sample size of 1,847 adults, including an oversample of 471 parents of children under age 5.
Vaccine initiation has been slow in the first month since it was approved for the youngest children. Just 2.8% of all eligible children under age 5 had received an initial dose as of July 19, compared with first-month uptake figures of more than 18% for the 5- to 11-year-olds and 27% for those aged 12-15, based on data from the Centers for Disease Control and Prevention.
The current rates for vaccination in those aged 5 and older look like this: 70.2% of 12- to 17-year-olds have received at least one dose, versus 37.1% of those aged 5-11. Just over 60% of the older children were fully vaccinated as of July 19, as were 30.2% of the 5- to 11-year-olds, the CDC reported on its COVID Data Tracker.
Number of new cases hits 2-month high
Despite the vaccine, SARS-CoV-2 and its various mutations have continued with their summer travels. With 92,000 newly infected children added for the week of July 15-21, there have now been a total of 14,003,497 pediatric cases reported since the start of the pandemic, which works out to 18.6% of cases in all ages, the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID-19 report.
The 92,000 new cases represent an increase of almost 22% over the previous week and mark the highest 1-week count since May, when the total passed 100,000 for 2 consecutive weeks. More recently the trend had seemed more stable as weekly cases dropped twice and rose twice as the total hovered around 70,000, based on the data collected by the AAP and CHA from state and territorial health departments.
A different scenario has played out for emergency department visits and hospital admissions, which have risen steadily since the beginning of April. The admission rate for children aged 0-17, which was just 0.13 new patients per 100,000 population on April 11, was up to 0.44 per 100,000 on July 21. By comparison, the highest rate reached last year during the Delta surge was 0.47 per 100,000, based on CDC data.
The 7-day average of emergency dept. visits among the youngest age group, 0-11 years, shows the same general increase as hospital admissions, but the older children have diverged form that path (see graph). For those aged 12-15 and 16-17, hospitalizations started dropping in late May and into mid-June before climbing again, although more slowly than for the youngest group, the CDC data show.
The ED visit rate with diagnosed COVID among those aged 0-11, measured at 6.1% of all visits on July 19, is, in fact, considerably higher than at any time during the Delta surge last year, when it never passed 4.0%, although much lower than peak Omicron (14.1%). That 6.1% was also higher than any other age group on that day, adults included, the CDC said.
New COVID-19 cases rose for the second week in a row as cumulative cases among U.S. children passed the 14-million mark, but a recent survey shows that more than half of parents believe that the vaccine is a greater risk to children under age 5 years than the virus.
In a Kaiser Family Foundation survey conducted July 7-17, 53% of parents with children aged 6 months to 5 years said that the vaccine is “a bigger risk to their child’s health than getting infected with COVID-19, compared to 44% who say getting infected is the bigger risk,” KFF reported July 26.
More than 4 out of 10 of respondents (43%) said that they will “definitely not” get their eligible children vaccinated, while only 7% said that their children had already received it and 10% said their children would get it as soon as possible, according to the KFF survey, which had an overall sample size of 1,847 adults, including an oversample of 471 parents of children under age 5.
Vaccine initiation has been slow in the first month since it was approved for the youngest children. Just 2.8% of all eligible children under age 5 had received an initial dose as of July 19, compared with first-month uptake figures of more than 18% for the 5- to 11-year-olds and 27% for those aged 12-15, based on data from the Centers for Disease Control and Prevention.
The current rates for vaccination in those aged 5 and older look like this: 70.2% of 12- to 17-year-olds have received at least one dose, versus 37.1% of those aged 5-11. Just over 60% of the older children were fully vaccinated as of July 19, as were 30.2% of the 5- to 11-year-olds, the CDC reported on its COVID Data Tracker.
Number of new cases hits 2-month high
Despite the vaccine, SARS-CoV-2 and its various mutations have continued with their summer travels. With 92,000 newly infected children added for the week of July 15-21, there have now been a total of 14,003,497 pediatric cases reported since the start of the pandemic, which works out to 18.6% of cases in all ages, the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID-19 report.
The 92,000 new cases represent an increase of almost 22% over the previous week and mark the highest 1-week count since May, when the total passed 100,000 for 2 consecutive weeks. More recently the trend had seemed more stable as weekly cases dropped twice and rose twice as the total hovered around 70,000, based on the data collected by the AAP and CHA from state and territorial health departments.
A different scenario has played out for emergency department visits and hospital admissions, which have risen steadily since the beginning of April. The admission rate for children aged 0-17, which was just 0.13 new patients per 100,000 population on April 11, was up to 0.44 per 100,000 on July 21. By comparison, the highest rate reached last year during the Delta surge was 0.47 per 100,000, based on CDC data.
The 7-day average of emergency dept. visits among the youngest age group, 0-11 years, shows the same general increase as hospital admissions, but the older children have diverged form that path (see graph). For those aged 12-15 and 16-17, hospitalizations started dropping in late May and into mid-June before climbing again, although more slowly than for the youngest group, the CDC data show.
The ED visit rate with diagnosed COVID among those aged 0-11, measured at 6.1% of all visits on July 19, is, in fact, considerably higher than at any time during the Delta surge last year, when it never passed 4.0%, although much lower than peak Omicron (14.1%). That 6.1% was also higher than any other age group on that day, adults included, the CDC said.