User login
-
Biden’s COVID-19 challenge: 100 million vaccinations in the first 100 days. It won’t be easy.
It’s in the nature of presidential candidates and new presidents to promise big things. Just months after his 1961 inauguration, President John F. Kennedy vowed to send a man to the moon by the end of the decade. That pledge was kept, but many others haven’t been, such as candidate Bill Clinton’s promise to provide universal health care and presidential hopeful George H.W. Bush’s guarantee of no new taxes.
Now, during a once-in-a-century pandemic, incoming President Joe Biden has promised to provide 100 million COVID-19 vaccinations in his first 100 days in office.
“This team will help get … at least 100 million covid vaccine shots into the arms of the American people in the first 100 days,” Biden said during a Dec. 8 news conference introducing key members of his health team.
When first asked about his pledge, the Biden team said the president-elect meant 50 million people would get their two-dose regimen. The incoming administration has since updated this plan, saying it will release vaccine doses as soon as they’re available instead of holding back some of that supply for second doses.
Either way, Biden may run into difficulty meeting that 100 million mark.
“I think it’s an attainable goal. I think it’s going to be extremely challenging,” said Claire Hannan, executive director of the Association of Immunization Managers.
While a pace of 1 million doses a day is “somewhat of an increase over what we’re already doing,” a much higher rate of vaccinations will be necessary to stem the pandemic, said Larry Levitt, executive vice president for health policy at Kaiser Family Foundation. (KHN is an editorially independent program of KFF.) “The Biden administration has plans to rationalize vaccine distribution, but increasing the supply quickly” could be a difficult task.
Under the Trump administration, vaccine deployment has been much slower than Biden’s plan. The rollout began on Dec. 14. Since then, 12 million shots have been given and 31 million doses have been shipped out, according to the Centers for Disease Control and Prevention’s vaccine tracker.
This sluggishness has been attributed to a lack of communication between the federal government and state and local health departments, not enough funding for large-scale vaccination efforts, and confusing federal guidance on distribution of the vaccines.
The same problems could plague the Biden administration, said experts.
States still aren’t sure how much vaccine they’ll get and whether there will be a sufficient supply, said Dr. Marcus Plescia, chief medical officer for the Association of State and Territorial Health Officials, which represents state public health agencies.
“We have been given little information about the amount of vaccine the states will receive in the near future and are of the impression that there may not be 1 million doses available per day in the first 100 days of the Biden administration,” said Dr. Plescia. “Or at least not in the early stages of the 100 days.”
Another challenge has been a lack of funding. Public health departments have had to start vaccination campaigns while also operating testing centers and conducting contact tracing efforts with budgets that have been critically underfunded for years.
“States have to pay for creating the systems, identifying the personnel, training, staffing, tracking people, information campaigns – all the things that go into getting a shot in someone’s arm,” said Jennifer Kates, director of global health & HIV policy at KFF. “They’re having to create an unprecedented mass vaccination program on a shaky foundation.”
The latest covid stimulus bill, signed into law in December, allocates almost $9 billion in funding to the CDC for vaccination efforts. About $4.5 billion is supposed to go to states, territories and tribal organizations, and $3 billion of that is slated to arrive soon.
But it’s not clear that level of funding can sustain mass vaccination campaigns as more groups become eligible for the vaccine.
Biden released a $1.9 trillion plan last week to address covid and the struggling economy. It includes $160 billion to create national vaccination and testing programs, but also earmarks funds for $1,400 stimulus payments to individuals, state and local government aid, extension of unemployment insurance, and financial assistance for schools to reopen safely.
Though it took Congress almost eight months to pass the last covid relief bill after Republican objections to the cost, Biden seems optimistic he’ll get some Republicans on board for his plan. But it’s not yet clear that will work.
There’s also the question of whether outgoing President Donald Trump’s impeachment trial will get in the way of Biden’s legislative priorities.
In addition, states have complained about a lack of guidance and confusing instructions on which groups should be given priority status for vaccination, an issue the Biden administration will need to address.
On Dec. 3, the CDC recommended health care personnel, residents of long-term care facilities, those 75 and older, and front-line essential workers should be immunized first. But on Jan. 12, the CDC shifted course and recommended that everyone over age 65 should be immunized. In a speech Biden gave on Jan. 15 detailing his vaccination plan, he said he would stick to the CDC’s recommendation to prioritize those over 65.
Outgoing Health and Human Services Secretary Alex Azar also said on Jan. 12 that states that moved their vaccine supply fastest would be prioritized in getting more shipments. It’s not known yet whether the Biden administration’s CDC will stick to this guidance. Critics have said it could make vaccine distribution less equitable.
In general, taking over with a strong vision and clear communication will be key to ramping up vaccine distribution, said Ms. Hannan.
“Everyone needs to understand what the goal is and how it’s going to work,” she said.
A challenge for Biden will be tamping expectations that the vaccine is all that is needed to end the pandemic. Across the country, covid cases are higher than ever, and in many locations officials cannot control the spread.
Public health experts said Biden must amp up efforts to increase testing across the country, as he has suggested he will do by promising to establish a national pandemic testing board.
With so much focus on vaccine distribution, it’s important that this part of the equation not be lost. Right now, “it’s completely all over the map,” said KFF’s Ms. Kates, adding that the federal government will need a “good sense” of who is and is not being tested in different areas in order to “fix” public health capacity.
Jan. 20, 2021, marks the launch of The Biden Promise Tracker, which monitors the 100 most important campaign promises of President Joseph R. Biden. Biden listed the coronavirus and a variety of other health-related issues among his top priorities. You can see the entire list – including improving the economy, responding to calls for racial justice and combating climate change – here. As part of KHN’s partnership with PolitiFact, we will follow the health-related issues and then rate them on whether the promise was achieved: Promise Kept, Promise Broken, Compromise, Stalled, In the Works or Not Yet Rated. We rate the promise not on the president’s intentions or effort, but on verifiable outcomes. PolitiFact previously tracked the promises of President Donald Trump and President Barack Obama.
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF, which is not affiliated with Kaiser Permanente.
It’s in the nature of presidential candidates and new presidents to promise big things. Just months after his 1961 inauguration, President John F. Kennedy vowed to send a man to the moon by the end of the decade. That pledge was kept, but many others haven’t been, such as candidate Bill Clinton’s promise to provide universal health care and presidential hopeful George H.W. Bush’s guarantee of no new taxes.
Now, during a once-in-a-century pandemic, incoming President Joe Biden has promised to provide 100 million COVID-19 vaccinations in his first 100 days in office.
“This team will help get … at least 100 million covid vaccine shots into the arms of the American people in the first 100 days,” Biden said during a Dec. 8 news conference introducing key members of his health team.
When first asked about his pledge, the Biden team said the president-elect meant 50 million people would get their two-dose regimen. The incoming administration has since updated this plan, saying it will release vaccine doses as soon as they’re available instead of holding back some of that supply for second doses.
Either way, Biden may run into difficulty meeting that 100 million mark.
“I think it’s an attainable goal. I think it’s going to be extremely challenging,” said Claire Hannan, executive director of the Association of Immunization Managers.
While a pace of 1 million doses a day is “somewhat of an increase over what we’re already doing,” a much higher rate of vaccinations will be necessary to stem the pandemic, said Larry Levitt, executive vice president for health policy at Kaiser Family Foundation. (KHN is an editorially independent program of KFF.) “The Biden administration has plans to rationalize vaccine distribution, but increasing the supply quickly” could be a difficult task.
Under the Trump administration, vaccine deployment has been much slower than Biden’s plan. The rollout began on Dec. 14. Since then, 12 million shots have been given and 31 million doses have been shipped out, according to the Centers for Disease Control and Prevention’s vaccine tracker.
This sluggishness has been attributed to a lack of communication between the federal government and state and local health departments, not enough funding for large-scale vaccination efforts, and confusing federal guidance on distribution of the vaccines.
The same problems could plague the Biden administration, said experts.
States still aren’t sure how much vaccine they’ll get and whether there will be a sufficient supply, said Dr. Marcus Plescia, chief medical officer for the Association of State and Territorial Health Officials, which represents state public health agencies.
“We have been given little information about the amount of vaccine the states will receive in the near future and are of the impression that there may not be 1 million doses available per day in the first 100 days of the Biden administration,” said Dr. Plescia. “Or at least not in the early stages of the 100 days.”
Another challenge has been a lack of funding. Public health departments have had to start vaccination campaigns while also operating testing centers and conducting contact tracing efforts with budgets that have been critically underfunded for years.
“States have to pay for creating the systems, identifying the personnel, training, staffing, tracking people, information campaigns – all the things that go into getting a shot in someone’s arm,” said Jennifer Kates, director of global health & HIV policy at KFF. “They’re having to create an unprecedented mass vaccination program on a shaky foundation.”
The latest covid stimulus bill, signed into law in December, allocates almost $9 billion in funding to the CDC for vaccination efforts. About $4.5 billion is supposed to go to states, territories and tribal organizations, and $3 billion of that is slated to arrive soon.
But it’s not clear that level of funding can sustain mass vaccination campaigns as more groups become eligible for the vaccine.
Biden released a $1.9 trillion plan last week to address covid and the struggling economy. It includes $160 billion to create national vaccination and testing programs, but also earmarks funds for $1,400 stimulus payments to individuals, state and local government aid, extension of unemployment insurance, and financial assistance for schools to reopen safely.
Though it took Congress almost eight months to pass the last covid relief bill after Republican objections to the cost, Biden seems optimistic he’ll get some Republicans on board for his plan. But it’s not yet clear that will work.
There’s also the question of whether outgoing President Donald Trump’s impeachment trial will get in the way of Biden’s legislative priorities.
In addition, states have complained about a lack of guidance and confusing instructions on which groups should be given priority status for vaccination, an issue the Biden administration will need to address.
On Dec. 3, the CDC recommended health care personnel, residents of long-term care facilities, those 75 and older, and front-line essential workers should be immunized first. But on Jan. 12, the CDC shifted course and recommended that everyone over age 65 should be immunized. In a speech Biden gave on Jan. 15 detailing his vaccination plan, he said he would stick to the CDC’s recommendation to prioritize those over 65.
Outgoing Health and Human Services Secretary Alex Azar also said on Jan. 12 that states that moved their vaccine supply fastest would be prioritized in getting more shipments. It’s not known yet whether the Biden administration’s CDC will stick to this guidance. Critics have said it could make vaccine distribution less equitable.
In general, taking over with a strong vision and clear communication will be key to ramping up vaccine distribution, said Ms. Hannan.
“Everyone needs to understand what the goal is and how it’s going to work,” she said.
A challenge for Biden will be tamping expectations that the vaccine is all that is needed to end the pandemic. Across the country, covid cases are higher than ever, and in many locations officials cannot control the spread.
Public health experts said Biden must amp up efforts to increase testing across the country, as he has suggested he will do by promising to establish a national pandemic testing board.
With so much focus on vaccine distribution, it’s important that this part of the equation not be lost. Right now, “it’s completely all over the map,” said KFF’s Ms. Kates, adding that the federal government will need a “good sense” of who is and is not being tested in different areas in order to “fix” public health capacity.
Jan. 20, 2021, marks the launch of The Biden Promise Tracker, which monitors the 100 most important campaign promises of President Joseph R. Biden. Biden listed the coronavirus and a variety of other health-related issues among his top priorities. You can see the entire list – including improving the economy, responding to calls for racial justice and combating climate change – here. As part of KHN’s partnership with PolitiFact, we will follow the health-related issues and then rate them on whether the promise was achieved: Promise Kept, Promise Broken, Compromise, Stalled, In the Works or Not Yet Rated. We rate the promise not on the president’s intentions or effort, but on verifiable outcomes. PolitiFact previously tracked the promises of President Donald Trump and President Barack Obama.
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF, which is not affiliated with Kaiser Permanente.
It’s in the nature of presidential candidates and new presidents to promise big things. Just months after his 1961 inauguration, President John F. Kennedy vowed to send a man to the moon by the end of the decade. That pledge was kept, but many others haven’t been, such as candidate Bill Clinton’s promise to provide universal health care and presidential hopeful George H.W. Bush’s guarantee of no new taxes.
Now, during a once-in-a-century pandemic, incoming President Joe Biden has promised to provide 100 million COVID-19 vaccinations in his first 100 days in office.
“This team will help get … at least 100 million covid vaccine shots into the arms of the American people in the first 100 days,” Biden said during a Dec. 8 news conference introducing key members of his health team.
When first asked about his pledge, the Biden team said the president-elect meant 50 million people would get their two-dose regimen. The incoming administration has since updated this plan, saying it will release vaccine doses as soon as they’re available instead of holding back some of that supply for second doses.
Either way, Biden may run into difficulty meeting that 100 million mark.
“I think it’s an attainable goal. I think it’s going to be extremely challenging,” said Claire Hannan, executive director of the Association of Immunization Managers.
While a pace of 1 million doses a day is “somewhat of an increase over what we’re already doing,” a much higher rate of vaccinations will be necessary to stem the pandemic, said Larry Levitt, executive vice president for health policy at Kaiser Family Foundation. (KHN is an editorially independent program of KFF.) “The Biden administration has plans to rationalize vaccine distribution, but increasing the supply quickly” could be a difficult task.
Under the Trump administration, vaccine deployment has been much slower than Biden’s plan. The rollout began on Dec. 14. Since then, 12 million shots have been given and 31 million doses have been shipped out, according to the Centers for Disease Control and Prevention’s vaccine tracker.
This sluggishness has been attributed to a lack of communication between the federal government and state and local health departments, not enough funding for large-scale vaccination efforts, and confusing federal guidance on distribution of the vaccines.
The same problems could plague the Biden administration, said experts.
States still aren’t sure how much vaccine they’ll get and whether there will be a sufficient supply, said Dr. Marcus Plescia, chief medical officer for the Association of State and Territorial Health Officials, which represents state public health agencies.
“We have been given little information about the amount of vaccine the states will receive in the near future and are of the impression that there may not be 1 million doses available per day in the first 100 days of the Biden administration,” said Dr. Plescia. “Or at least not in the early stages of the 100 days.”
Another challenge has been a lack of funding. Public health departments have had to start vaccination campaigns while also operating testing centers and conducting contact tracing efforts with budgets that have been critically underfunded for years.
“States have to pay for creating the systems, identifying the personnel, training, staffing, tracking people, information campaigns – all the things that go into getting a shot in someone’s arm,” said Jennifer Kates, director of global health & HIV policy at KFF. “They’re having to create an unprecedented mass vaccination program on a shaky foundation.”
The latest covid stimulus bill, signed into law in December, allocates almost $9 billion in funding to the CDC for vaccination efforts. About $4.5 billion is supposed to go to states, territories and tribal organizations, and $3 billion of that is slated to arrive soon.
But it’s not clear that level of funding can sustain mass vaccination campaigns as more groups become eligible for the vaccine.
Biden released a $1.9 trillion plan last week to address covid and the struggling economy. It includes $160 billion to create national vaccination and testing programs, but also earmarks funds for $1,400 stimulus payments to individuals, state and local government aid, extension of unemployment insurance, and financial assistance for schools to reopen safely.
Though it took Congress almost eight months to pass the last covid relief bill after Republican objections to the cost, Biden seems optimistic he’ll get some Republicans on board for his plan. But it’s not yet clear that will work.
There’s also the question of whether outgoing President Donald Trump’s impeachment trial will get in the way of Biden’s legislative priorities.
In addition, states have complained about a lack of guidance and confusing instructions on which groups should be given priority status for vaccination, an issue the Biden administration will need to address.
On Dec. 3, the CDC recommended health care personnel, residents of long-term care facilities, those 75 and older, and front-line essential workers should be immunized first. But on Jan. 12, the CDC shifted course and recommended that everyone over age 65 should be immunized. In a speech Biden gave on Jan. 15 detailing his vaccination plan, he said he would stick to the CDC’s recommendation to prioritize those over 65.
Outgoing Health and Human Services Secretary Alex Azar also said on Jan. 12 that states that moved their vaccine supply fastest would be prioritized in getting more shipments. It’s not known yet whether the Biden administration’s CDC will stick to this guidance. Critics have said it could make vaccine distribution less equitable.
In general, taking over with a strong vision and clear communication will be key to ramping up vaccine distribution, said Ms. Hannan.
“Everyone needs to understand what the goal is and how it’s going to work,” she said.
A challenge for Biden will be tamping expectations that the vaccine is all that is needed to end the pandemic. Across the country, covid cases are higher than ever, and in many locations officials cannot control the spread.
Public health experts said Biden must amp up efforts to increase testing across the country, as he has suggested he will do by promising to establish a national pandemic testing board.
With so much focus on vaccine distribution, it’s important that this part of the equation not be lost. Right now, “it’s completely all over the map,” said KFF’s Ms. Kates, adding that the federal government will need a “good sense” of who is and is not being tested in different areas in order to “fix” public health capacity.
Jan. 20, 2021, marks the launch of The Biden Promise Tracker, which monitors the 100 most important campaign promises of President Joseph R. Biden. Biden listed the coronavirus and a variety of other health-related issues among his top priorities. You can see the entire list – including improving the economy, responding to calls for racial justice and combating climate change – here. As part of KHN’s partnership with PolitiFact, we will follow the health-related issues and then rate them on whether the promise was achieved: Promise Kept, Promise Broken, Compromise, Stalled, In the Works or Not Yet Rated. We rate the promise not on the president’s intentions or effort, but on verifiable outcomes. PolitiFact previously tracked the promises of President Donald Trump and President Barack Obama.
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF, which is not affiliated with Kaiser Permanente.
Von Willebrand disease guidelines address women’s bleeding concerns
New guidelines issued jointly by four major international hematology groups focus on the management of patients with von Willebrand disease (VWD), the most common bleeding disorder in the world.
The evidence-based guidelines, published in Blood Advances, were developed in collaboration by the American Society of Hematology (ASH), the International Society on Thrombosis and Haemostasis, the National Hemophilia Foundation, and the World Federation of Hemophilia. They outline key recommendations spanning the care of patients with a broad range of therapeutic needs.
“We addressed some of the questions that were most important to the community, but certainly there are a lot of areas that we couldn’t cover” said coauthor Veronica H. Flood, MD, of the Medical College of Wisconsin in Milwaukee.
The guidelines process began with a survey sent to the von Willebrand disease community, including patients, caregivers, nurses, physicians, and scientists. The respondents were asked to prioritize issues that they felt should be addressed in the guidelines.
“Interestingly, some of the issues were the same between patients and caregivers and physicians, and some were different, but there were obviously some areas that we just couldn’t cover,” she said in an interview.
One of the areas of greatest concern for respondents was bleeding in women, and many of the recommendations include specific considerations for management of gynecologic and obstetric patients, Dr. Flood said.
“We also tried to make the questions applicable to as many patients with von Willebrand disease as possible,” she added.
Some of the questions, such as recommendation 1, regarding prophylaxis, are geared toward management of patients with severe disease, while others, such as recommendations for treatment of menstrual bleeding, are more suited for patients with milder VWD.
All of the recommendations in the guidelines are “conditional” (suggested), due to very low certainty in the evidence of effects, the authors noted.
Prophylaxis
The guidelines suggest long-term prophylaxis for patients with a history of severe and frequent bleeds, with periodic assessment of the need for prophylaxis.
Desmopressin
For those patients who may benefit from the use of desmopressin, primarily those with type 1 VWD, and who have a baseline von Willebrand factor (VWF) level below 0.30 IU/mL, the panel issued a conditional recommendation for a desmopressin trial with treatment based on the patient’s results compared with not performing a trial and treating with tranexamic acid or factor concentrate. The guidelines also advise against treating with desmopressin in the absence of a trial. In a section of “good practice statements,” the guidelines indicate that using desmopressin in patients with type 2B VWD is generally contraindicated, because of the risk of thrombocytopenia as a result of increased platelet binding. In addition, desmopressin is generally contraindicated in patients with active cardiovascular disease, patients with seizure disorders, patients less than 2 years old, and patients with type 1C VWD in the setting of surgery.
Antithrombotic therapy
The guideline panelists conditionally recommend antithrombotic therapy with either antiplatelet agents or anticoagulants, with an emphasis on reassessing bleeding risk throughout the course of treatment.
An accompanying good practice statement calls for individualized assessments of risks and benefits of specific antithrombotic therapies by a multidisciplinary team including hematologists, cardiovascular specialists, and the patient.
Major surgery
This section includes a recommendation for targeting both factor VIII and VWF activity levels to a minimum of 50 IU/mL for at least 3 days after surgery, and a suggestion against using factor VIII target levels alone.
Minor surgery/invasive procedures
The panelists suggest increasing VWF activity levels to a minimum of 0.50 IU/mL with desmopressin or factor concentrate with the addition of tranexamic acid over raising VWF levels to at least 0.50 IU/mL with desmopressin or factor concentrate alone.
In addition, the panelists suggest “giving tranexamic acid alone over increasing VWF activity levels to a minimum threshold of 0.50 IU/mL with any intervention in patients with type 1 VWD with baseline VWF activity levels of 0.30 IU/mL and a mild bleeding phenotype undergoing minor mucosal procedures.”
Heavy menstrual bleeding
In women with heavy menstrual bleeding who do not plan to conceive, the panel suggests either combined hormonal therapy or levonorgestrel-releasing intrauterine system, or tranexamic acid over desmopressin.
In women who wish to conceive, the panel suggests using tranexamic acid over desmopressin.
Neuraxial anesthesia during labor
For women in labor for whom neuraxial anesthesia is considered, the guidelines suggest targeting a VWF activity level from 0.50 to 1.50 IU/mL over targeting a level above 1.50 IU/mL.
Postpartum management
“The guideline panel suggests the use of tranexamic acid over not using it in women with type 1 VWD or low VWF levels (and this may also apply to types 2 and 3 VWD) during the postpartum period,” the guidelines say.
An accompanying good practice statement says that tranexamic acid can be provided orally or intravenously. The oral dose is 25 mg/kg three times daily for 10-14 days, or longer if blood loss remains heavy.
Dr. Flood said that the guidelines were developed under the assumption that they would apply to care of patients in regions with a high or moderately high degree of clinical resources.
“We recognize that this eliminates a great deal of the globe, and our hope is that ASH and the other sponsoring organizations are going to let us revise this and do a version for lower-resourced settings,” she said.
New guidelines issued jointly by four major international hematology groups focus on the management of patients with von Willebrand disease (VWD), the most common bleeding disorder in the world.
The evidence-based guidelines, published in Blood Advances, were developed in collaboration by the American Society of Hematology (ASH), the International Society on Thrombosis and Haemostasis, the National Hemophilia Foundation, and the World Federation of Hemophilia. They outline key recommendations spanning the care of patients with a broad range of therapeutic needs.
“We addressed some of the questions that were most important to the community, but certainly there are a lot of areas that we couldn’t cover” said coauthor Veronica H. Flood, MD, of the Medical College of Wisconsin in Milwaukee.
The guidelines process began with a survey sent to the von Willebrand disease community, including patients, caregivers, nurses, physicians, and scientists. The respondents were asked to prioritize issues that they felt should be addressed in the guidelines.
“Interestingly, some of the issues were the same between patients and caregivers and physicians, and some were different, but there were obviously some areas that we just couldn’t cover,” she said in an interview.
One of the areas of greatest concern for respondents was bleeding in women, and many of the recommendations include specific considerations for management of gynecologic and obstetric patients, Dr. Flood said.
“We also tried to make the questions applicable to as many patients with von Willebrand disease as possible,” she added.
Some of the questions, such as recommendation 1, regarding prophylaxis, are geared toward management of patients with severe disease, while others, such as recommendations for treatment of menstrual bleeding, are more suited for patients with milder VWD.
All of the recommendations in the guidelines are “conditional” (suggested), due to very low certainty in the evidence of effects, the authors noted.
Prophylaxis
The guidelines suggest long-term prophylaxis for patients with a history of severe and frequent bleeds, with periodic assessment of the need for prophylaxis.
Desmopressin
For those patients who may benefit from the use of desmopressin, primarily those with type 1 VWD, and who have a baseline von Willebrand factor (VWF) level below 0.30 IU/mL, the panel issued a conditional recommendation for a desmopressin trial with treatment based on the patient’s results compared with not performing a trial and treating with tranexamic acid or factor concentrate. The guidelines also advise against treating with desmopressin in the absence of a trial. In a section of “good practice statements,” the guidelines indicate that using desmopressin in patients with type 2B VWD is generally contraindicated, because of the risk of thrombocytopenia as a result of increased platelet binding. In addition, desmopressin is generally contraindicated in patients with active cardiovascular disease, patients with seizure disorders, patients less than 2 years old, and patients with type 1C VWD in the setting of surgery.
Antithrombotic therapy
The guideline panelists conditionally recommend antithrombotic therapy with either antiplatelet agents or anticoagulants, with an emphasis on reassessing bleeding risk throughout the course of treatment.
An accompanying good practice statement calls for individualized assessments of risks and benefits of specific antithrombotic therapies by a multidisciplinary team including hematologists, cardiovascular specialists, and the patient.
Major surgery
This section includes a recommendation for targeting both factor VIII and VWF activity levels to a minimum of 50 IU/mL for at least 3 days after surgery, and a suggestion against using factor VIII target levels alone.
Minor surgery/invasive procedures
The panelists suggest increasing VWF activity levels to a minimum of 0.50 IU/mL with desmopressin or factor concentrate with the addition of tranexamic acid over raising VWF levels to at least 0.50 IU/mL with desmopressin or factor concentrate alone.
In addition, the panelists suggest “giving tranexamic acid alone over increasing VWF activity levels to a minimum threshold of 0.50 IU/mL with any intervention in patients with type 1 VWD with baseline VWF activity levels of 0.30 IU/mL and a mild bleeding phenotype undergoing minor mucosal procedures.”
Heavy menstrual bleeding
In women with heavy menstrual bleeding who do not plan to conceive, the panel suggests either combined hormonal therapy or levonorgestrel-releasing intrauterine system, or tranexamic acid over desmopressin.
In women who wish to conceive, the panel suggests using tranexamic acid over desmopressin.
Neuraxial anesthesia during labor
For women in labor for whom neuraxial anesthesia is considered, the guidelines suggest targeting a VWF activity level from 0.50 to 1.50 IU/mL over targeting a level above 1.50 IU/mL.
Postpartum management
“The guideline panel suggests the use of tranexamic acid over not using it in women with type 1 VWD or low VWF levels (and this may also apply to types 2 and 3 VWD) during the postpartum period,” the guidelines say.
An accompanying good practice statement says that tranexamic acid can be provided orally or intravenously. The oral dose is 25 mg/kg three times daily for 10-14 days, or longer if blood loss remains heavy.
Dr. Flood said that the guidelines were developed under the assumption that they would apply to care of patients in regions with a high or moderately high degree of clinical resources.
“We recognize that this eliminates a great deal of the globe, and our hope is that ASH and the other sponsoring organizations are going to let us revise this and do a version for lower-resourced settings,” she said.
New guidelines issued jointly by four major international hematology groups focus on the management of patients with von Willebrand disease (VWD), the most common bleeding disorder in the world.
The evidence-based guidelines, published in Blood Advances, were developed in collaboration by the American Society of Hematology (ASH), the International Society on Thrombosis and Haemostasis, the National Hemophilia Foundation, and the World Federation of Hemophilia. They outline key recommendations spanning the care of patients with a broad range of therapeutic needs.
“We addressed some of the questions that were most important to the community, but certainly there are a lot of areas that we couldn’t cover” said coauthor Veronica H. Flood, MD, of the Medical College of Wisconsin in Milwaukee.
The guidelines process began with a survey sent to the von Willebrand disease community, including patients, caregivers, nurses, physicians, and scientists. The respondents were asked to prioritize issues that they felt should be addressed in the guidelines.
“Interestingly, some of the issues were the same between patients and caregivers and physicians, and some were different, but there were obviously some areas that we just couldn’t cover,” she said in an interview.
One of the areas of greatest concern for respondents was bleeding in women, and many of the recommendations include specific considerations for management of gynecologic and obstetric patients, Dr. Flood said.
“We also tried to make the questions applicable to as many patients with von Willebrand disease as possible,” she added.
Some of the questions, such as recommendation 1, regarding prophylaxis, are geared toward management of patients with severe disease, while others, such as recommendations for treatment of menstrual bleeding, are more suited for patients with milder VWD.
All of the recommendations in the guidelines are “conditional” (suggested), due to very low certainty in the evidence of effects, the authors noted.
Prophylaxis
The guidelines suggest long-term prophylaxis for patients with a history of severe and frequent bleeds, with periodic assessment of the need for prophylaxis.
Desmopressin
For those patients who may benefit from the use of desmopressin, primarily those with type 1 VWD, and who have a baseline von Willebrand factor (VWF) level below 0.30 IU/mL, the panel issued a conditional recommendation for a desmopressin trial with treatment based on the patient’s results compared with not performing a trial and treating with tranexamic acid or factor concentrate. The guidelines also advise against treating with desmopressin in the absence of a trial. In a section of “good practice statements,” the guidelines indicate that using desmopressin in patients with type 2B VWD is generally contraindicated, because of the risk of thrombocytopenia as a result of increased platelet binding. In addition, desmopressin is generally contraindicated in patients with active cardiovascular disease, patients with seizure disorders, patients less than 2 years old, and patients with type 1C VWD in the setting of surgery.
Antithrombotic therapy
The guideline panelists conditionally recommend antithrombotic therapy with either antiplatelet agents or anticoagulants, with an emphasis on reassessing bleeding risk throughout the course of treatment.
An accompanying good practice statement calls for individualized assessments of risks and benefits of specific antithrombotic therapies by a multidisciplinary team including hematologists, cardiovascular specialists, and the patient.
Major surgery
This section includes a recommendation for targeting both factor VIII and VWF activity levels to a minimum of 50 IU/mL for at least 3 days after surgery, and a suggestion against using factor VIII target levels alone.
Minor surgery/invasive procedures
The panelists suggest increasing VWF activity levels to a minimum of 0.50 IU/mL with desmopressin or factor concentrate with the addition of tranexamic acid over raising VWF levels to at least 0.50 IU/mL with desmopressin or factor concentrate alone.
In addition, the panelists suggest “giving tranexamic acid alone over increasing VWF activity levels to a minimum threshold of 0.50 IU/mL with any intervention in patients with type 1 VWD with baseline VWF activity levels of 0.30 IU/mL and a mild bleeding phenotype undergoing minor mucosal procedures.”
Heavy menstrual bleeding
In women with heavy menstrual bleeding who do not plan to conceive, the panel suggests either combined hormonal therapy or levonorgestrel-releasing intrauterine system, or tranexamic acid over desmopressin.
In women who wish to conceive, the panel suggests using tranexamic acid over desmopressin.
Neuraxial anesthesia during labor
For women in labor for whom neuraxial anesthesia is considered, the guidelines suggest targeting a VWF activity level from 0.50 to 1.50 IU/mL over targeting a level above 1.50 IU/mL.
Postpartum management
“The guideline panel suggests the use of tranexamic acid over not using it in women with type 1 VWD or low VWF levels (and this may also apply to types 2 and 3 VWD) during the postpartum period,” the guidelines say.
An accompanying good practice statement says that tranexamic acid can be provided orally or intravenously. The oral dose is 25 mg/kg three times daily for 10-14 days, or longer if blood loss remains heavy.
Dr. Flood said that the guidelines were developed under the assumption that they would apply to care of patients in regions with a high or moderately high degree of clinical resources.
“We recognize that this eliminates a great deal of the globe, and our hope is that ASH and the other sponsoring organizations are going to let us revise this and do a version for lower-resourced settings,” she said.
FROM BLOOD ADVANCES
How do you answer patients’ emails?
The pandemic has isolated our patients to an unprecedented degree, forcing them to find other ways to communicate with us, including email.
Responses varied all over the map. Some refuse the medium entirely. “I politely say that I don’t practice dermatology via email,” said one. “Please schedule a teledermatology appointment and I’d be happy to help.”
Others are ambivalent: “I do email with some patients who have complex situations or quick questions, but if it gets out of hand then I let them know someone will call to make an appointment.” Another office treats them as a one-way street: “We set up one account to receive patients’ emails, but we tell them clearly that we don’t respond ... my staff or I call them back.”
Still others have assimilated it completely. “Patients email through the portal and my MA routes [them] to me. I answer questions and the MA responds ... staff loves it because it’s so much faster than the phone.”
A 1998 study in JAMA was more scientifically designed, but basically reached the same conclusion. The authors found “a striking lack of consensus” on how to deal with patient emails: 50% responded to them, but 31% of responders refused to give advice without seeing the patient, while 59% offered a diagnosis, and a third of that group went on to provide specific advice about therapy. In response to a follow-up questionnaire, 28% said that they tended not to answer any patient emails, 24% said they usually replied with a standard message, and 24% said they answer each request individually. The authors concluded that “standards for physician response to unsolicited patient emails are needed.”
Indeed, my own unscientific survey suggests that, more than 20 years later, there is still nothing resembling a consensus on this issue. In the interim, several groups, including the American Medical Informatics Association and the American Medical Association have proposed standards, but none have been generally accepted. Until that happens, it seems prudent for each individual practice to adopt its own guidelines. For ideas, take a look at the proposals from the groups I mentioned, plus any others you can find. When you’re done, consider running your list past your attorney to make sure you haven’t forgotten anything, and that there are no unique requirements in your state.
Your guidelines may be very simple (if you decide never to answer any queries) or very complex, depending on your situation and personal philosophy. But all guidelines should cover such issues as authentication of correspondents, informed consent, licensing jurisdiction (if you receive emails from states in which you are not licensed), and of course, confidentiality.
Contrary to popular belief, HIPAA does not prohibit email communication with patients, nor require that it be encrypted. The HIPAA website specifically says: “Patients may initiate communications with a provider using email. If this situation occurs, the health care provider can assume (unless the patient has explicitly stated otherwise) that e-mail communications are acceptable to the individual.”
Still, if you are not comfortable with unencrypted communication, encryption software can be added to your practice’s email system. Proofpoint, Tumbleweed, Zix, and many other vendors sell encryption packages. (As always, I have no financial interest in any product or enterprise mentioned in this column.)
Another option is web-based messaging: Patients enter your website and send a message using an electronic template that you design. A designated staffer will be notified by regular email when messages are received, and can post a reply on a page that can only be accessed by the patient. Besides enhancing privacy and security, you can state your guidelines in plain English to preclude any misunderstanding of what you will and will not address online.
Web-based messaging services can be freestanding or incorporated into existing secure websites. Medfusion and klara are among the leading vendors of secure messaging services.
The important thing is to make a firm decision on how you want to deal with emails, and stick with that method. And follow your guidelines.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
The pandemic has isolated our patients to an unprecedented degree, forcing them to find other ways to communicate with us, including email.
Responses varied all over the map. Some refuse the medium entirely. “I politely say that I don’t practice dermatology via email,” said one. “Please schedule a teledermatology appointment and I’d be happy to help.”
Others are ambivalent: “I do email with some patients who have complex situations or quick questions, but if it gets out of hand then I let them know someone will call to make an appointment.” Another office treats them as a one-way street: “We set up one account to receive patients’ emails, but we tell them clearly that we don’t respond ... my staff or I call them back.”
Still others have assimilated it completely. “Patients email through the portal and my MA routes [them] to me. I answer questions and the MA responds ... staff loves it because it’s so much faster than the phone.”
A 1998 study in JAMA was more scientifically designed, but basically reached the same conclusion. The authors found “a striking lack of consensus” on how to deal with patient emails: 50% responded to them, but 31% of responders refused to give advice without seeing the patient, while 59% offered a diagnosis, and a third of that group went on to provide specific advice about therapy. In response to a follow-up questionnaire, 28% said that they tended not to answer any patient emails, 24% said they usually replied with a standard message, and 24% said they answer each request individually. The authors concluded that “standards for physician response to unsolicited patient emails are needed.”
Indeed, my own unscientific survey suggests that, more than 20 years later, there is still nothing resembling a consensus on this issue. In the interim, several groups, including the American Medical Informatics Association and the American Medical Association have proposed standards, but none have been generally accepted. Until that happens, it seems prudent for each individual practice to adopt its own guidelines. For ideas, take a look at the proposals from the groups I mentioned, plus any others you can find. When you’re done, consider running your list past your attorney to make sure you haven’t forgotten anything, and that there are no unique requirements in your state.
Your guidelines may be very simple (if you decide never to answer any queries) or very complex, depending on your situation and personal philosophy. But all guidelines should cover such issues as authentication of correspondents, informed consent, licensing jurisdiction (if you receive emails from states in which you are not licensed), and of course, confidentiality.
Contrary to popular belief, HIPAA does not prohibit email communication with patients, nor require that it be encrypted. The HIPAA website specifically says: “Patients may initiate communications with a provider using email. If this situation occurs, the health care provider can assume (unless the patient has explicitly stated otherwise) that e-mail communications are acceptable to the individual.”
Still, if you are not comfortable with unencrypted communication, encryption software can be added to your practice’s email system. Proofpoint, Tumbleweed, Zix, and many other vendors sell encryption packages. (As always, I have no financial interest in any product or enterprise mentioned in this column.)
Another option is web-based messaging: Patients enter your website and send a message using an electronic template that you design. A designated staffer will be notified by regular email when messages are received, and can post a reply on a page that can only be accessed by the patient. Besides enhancing privacy and security, you can state your guidelines in plain English to preclude any misunderstanding of what you will and will not address online.
Web-based messaging services can be freestanding or incorporated into existing secure websites. Medfusion and klara are among the leading vendors of secure messaging services.
The important thing is to make a firm decision on how you want to deal with emails, and stick with that method. And follow your guidelines.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
The pandemic has isolated our patients to an unprecedented degree, forcing them to find other ways to communicate with us, including email.
Responses varied all over the map. Some refuse the medium entirely. “I politely say that I don’t practice dermatology via email,” said one. “Please schedule a teledermatology appointment and I’d be happy to help.”
Others are ambivalent: “I do email with some patients who have complex situations or quick questions, but if it gets out of hand then I let them know someone will call to make an appointment.” Another office treats them as a one-way street: “We set up one account to receive patients’ emails, but we tell them clearly that we don’t respond ... my staff or I call them back.”
Still others have assimilated it completely. “Patients email through the portal and my MA routes [them] to me. I answer questions and the MA responds ... staff loves it because it’s so much faster than the phone.”
A 1998 study in JAMA was more scientifically designed, but basically reached the same conclusion. The authors found “a striking lack of consensus” on how to deal with patient emails: 50% responded to them, but 31% of responders refused to give advice without seeing the patient, while 59% offered a diagnosis, and a third of that group went on to provide specific advice about therapy. In response to a follow-up questionnaire, 28% said that they tended not to answer any patient emails, 24% said they usually replied with a standard message, and 24% said they answer each request individually. The authors concluded that “standards for physician response to unsolicited patient emails are needed.”
Indeed, my own unscientific survey suggests that, more than 20 years later, there is still nothing resembling a consensus on this issue. In the interim, several groups, including the American Medical Informatics Association and the American Medical Association have proposed standards, but none have been generally accepted. Until that happens, it seems prudent for each individual practice to adopt its own guidelines. For ideas, take a look at the proposals from the groups I mentioned, plus any others you can find. When you’re done, consider running your list past your attorney to make sure you haven’t forgotten anything, and that there are no unique requirements in your state.
Your guidelines may be very simple (if you decide never to answer any queries) or very complex, depending on your situation and personal philosophy. But all guidelines should cover such issues as authentication of correspondents, informed consent, licensing jurisdiction (if you receive emails from states in which you are not licensed), and of course, confidentiality.
Contrary to popular belief, HIPAA does not prohibit email communication with patients, nor require that it be encrypted. The HIPAA website specifically says: “Patients may initiate communications with a provider using email. If this situation occurs, the health care provider can assume (unless the patient has explicitly stated otherwise) that e-mail communications are acceptable to the individual.”
Still, if you are not comfortable with unencrypted communication, encryption software can be added to your practice’s email system. Proofpoint, Tumbleweed, Zix, and many other vendors sell encryption packages. (As always, I have no financial interest in any product or enterprise mentioned in this column.)
Another option is web-based messaging: Patients enter your website and send a message using an electronic template that you design. A designated staffer will be notified by regular email when messages are received, and can post a reply on a page that can only be accessed by the patient. Besides enhancing privacy and security, you can state your guidelines in plain English to preclude any misunderstanding of what you will and will not address online.
Web-based messaging services can be freestanding or incorporated into existing secure websites. Medfusion and klara are among the leading vendors of secure messaging services.
The important thing is to make a firm decision on how you want to deal with emails, and stick with that method. And follow your guidelines.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
An introduction to Naikan
The list of things to be ungrateful for last year is long. You’re not supposed to make this list, though. The best practice is to list what you’re grateful for, even when living in trying times. That’s a long list too, but I find making it similarly unfruitful.
Of course, I’m grateful I don’t have COVID-19, thankful my practice hasn’t been significantly impacted, grateful I got the vaccine. But simply repeating these gratitudes daily seems ineffective. I’ve learned a different “gratefulness practice” that perhaps works better.
It’s a Japanese method called Naikan (pronounced “nye-kan”). The word means introspection and the practice is one of self-reflection. But . Yoshimoto Ishin developed Naikan in the 1940s. He was a Japanese businessman and devout Buddhist who wanted to make a difficult form of meditation more accessible. He removed the ascetic bits like sleep deprivation and refined the exercises such that they better see how others see us. The result is a way to reframe your life experiences and help you understand how much others do for us and how our actions and attitudes impact others. It can be done alone or with a partner. You can do it at the beginning or end of your day.
The method is simple. You ask three questions:
What have I received today from ___________?
What have I given today to ___________?
What difficulty or trouble have I caused to ___________?
The first question is similar to most gratitude practices. For example, you might ask, “What have I received from (my husband or nurse or friend, etc.)? Today, I received a beautifully tidied-up office from my wife who spent time last night sorting things. This made it easy for me to sit down and start writing this piece.
The second question is better. What have I given today to (my wife, or patient, or mom, etc.)? It can be simple as: Today, I slowed down to let everyone who was in the closed highway lane back into traffic (even though some were clearly undeserving of my generosity). Or last night, I worked to coordinate with anesthesia and scheduling to help a little girl who would benefit from conscious sedation for her procedure.
Combined, these two questions pull you 180 degrees from our default mode, which is complaining. We are wired to find, and talk about, all the inconveniences in our lives: Roadway construction caused a traffic backup that led to running late for clinic. First patient was peeved and had a list of complaints, the last of which was hair loss. Isn’t it much better to rave about how our dermatology nurse volunteered to work the hospital COVID-19 unit to give her colleagues a break? Or how my 10:15 patient came early to be sure she was on time? (It happens.)
The last question is the best. We all spend time thinking about what others think of us. We should spend time thinking about what impact we’ve had on them. Like a cold shower, it’s both briskly awakening and easy to do. Go back through your day and reflect on what you did that made things difficult for others. It can be as simple as I started whining about how a patient waylaid me with her silly complaints. That led to my colleague’s joining in about difficult patients. Or I was late turning in my article, which made my editor have to work harder to get it completed in time.
There’s plenty of things we should be grateful for. In doing these exercises you’ll learn just how much others have cared for you and, I hope, how you might do things to make them grateful for you.
If you’re interested in learning more about Naikan, I discovered this from Brett McKay’s The Art of Manliness podcast and the teaching of Gregg Krech, summarized in his book, “Naikan: Gratitude, Grace, and the Japanese Art of Self-Reflection.”
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected] .
The list of things to be ungrateful for last year is long. You’re not supposed to make this list, though. The best practice is to list what you’re grateful for, even when living in trying times. That’s a long list too, but I find making it similarly unfruitful.
Of course, I’m grateful I don’t have COVID-19, thankful my practice hasn’t been significantly impacted, grateful I got the vaccine. But simply repeating these gratitudes daily seems ineffective. I’ve learned a different “gratefulness practice” that perhaps works better.
It’s a Japanese method called Naikan (pronounced “nye-kan”). The word means introspection and the practice is one of self-reflection. But . Yoshimoto Ishin developed Naikan in the 1940s. He was a Japanese businessman and devout Buddhist who wanted to make a difficult form of meditation more accessible. He removed the ascetic bits like sleep deprivation and refined the exercises such that they better see how others see us. The result is a way to reframe your life experiences and help you understand how much others do for us and how our actions and attitudes impact others. It can be done alone or with a partner. You can do it at the beginning or end of your day.
The method is simple. You ask three questions:
What have I received today from ___________?
What have I given today to ___________?
What difficulty or trouble have I caused to ___________?
The first question is similar to most gratitude practices. For example, you might ask, “What have I received from (my husband or nurse or friend, etc.)? Today, I received a beautifully tidied-up office from my wife who spent time last night sorting things. This made it easy for me to sit down and start writing this piece.
The second question is better. What have I given today to (my wife, or patient, or mom, etc.)? It can be simple as: Today, I slowed down to let everyone who was in the closed highway lane back into traffic (even though some were clearly undeserving of my generosity). Or last night, I worked to coordinate with anesthesia and scheduling to help a little girl who would benefit from conscious sedation for her procedure.
Combined, these two questions pull you 180 degrees from our default mode, which is complaining. We are wired to find, and talk about, all the inconveniences in our lives: Roadway construction caused a traffic backup that led to running late for clinic. First patient was peeved and had a list of complaints, the last of which was hair loss. Isn’t it much better to rave about how our dermatology nurse volunteered to work the hospital COVID-19 unit to give her colleagues a break? Or how my 10:15 patient came early to be sure she was on time? (It happens.)
The last question is the best. We all spend time thinking about what others think of us. We should spend time thinking about what impact we’ve had on them. Like a cold shower, it’s both briskly awakening and easy to do. Go back through your day and reflect on what you did that made things difficult for others. It can be as simple as I started whining about how a patient waylaid me with her silly complaints. That led to my colleague’s joining in about difficult patients. Or I was late turning in my article, which made my editor have to work harder to get it completed in time.
There’s plenty of things we should be grateful for. In doing these exercises you’ll learn just how much others have cared for you and, I hope, how you might do things to make them grateful for you.
If you’re interested in learning more about Naikan, I discovered this from Brett McKay’s The Art of Manliness podcast and the teaching of Gregg Krech, summarized in his book, “Naikan: Gratitude, Grace, and the Japanese Art of Self-Reflection.”
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected] .
The list of things to be ungrateful for last year is long. You’re not supposed to make this list, though. The best practice is to list what you’re grateful for, even when living in trying times. That’s a long list too, but I find making it similarly unfruitful.
Of course, I’m grateful I don’t have COVID-19, thankful my practice hasn’t been significantly impacted, grateful I got the vaccine. But simply repeating these gratitudes daily seems ineffective. I’ve learned a different “gratefulness practice” that perhaps works better.
It’s a Japanese method called Naikan (pronounced “nye-kan”). The word means introspection and the practice is one of self-reflection. But . Yoshimoto Ishin developed Naikan in the 1940s. He was a Japanese businessman and devout Buddhist who wanted to make a difficult form of meditation more accessible. He removed the ascetic bits like sleep deprivation and refined the exercises such that they better see how others see us. The result is a way to reframe your life experiences and help you understand how much others do for us and how our actions and attitudes impact others. It can be done alone or with a partner. You can do it at the beginning or end of your day.
The method is simple. You ask three questions:
What have I received today from ___________?
What have I given today to ___________?
What difficulty or trouble have I caused to ___________?
The first question is similar to most gratitude practices. For example, you might ask, “What have I received from (my husband or nurse or friend, etc.)? Today, I received a beautifully tidied-up office from my wife who spent time last night sorting things. This made it easy for me to sit down and start writing this piece.
The second question is better. What have I given today to (my wife, or patient, or mom, etc.)? It can be simple as: Today, I slowed down to let everyone who was in the closed highway lane back into traffic (even though some were clearly undeserving of my generosity). Or last night, I worked to coordinate with anesthesia and scheduling to help a little girl who would benefit from conscious sedation for her procedure.
Combined, these two questions pull you 180 degrees from our default mode, which is complaining. We are wired to find, and talk about, all the inconveniences in our lives: Roadway construction caused a traffic backup that led to running late for clinic. First patient was peeved and had a list of complaints, the last of which was hair loss. Isn’t it much better to rave about how our dermatology nurse volunteered to work the hospital COVID-19 unit to give her colleagues a break? Or how my 10:15 patient came early to be sure she was on time? (It happens.)
The last question is the best. We all spend time thinking about what others think of us. We should spend time thinking about what impact we’ve had on them. Like a cold shower, it’s both briskly awakening and easy to do. Go back through your day and reflect on what you did that made things difficult for others. It can be as simple as I started whining about how a patient waylaid me with her silly complaints. That led to my colleague’s joining in about difficult patients. Or I was late turning in my article, which made my editor have to work harder to get it completed in time.
There’s plenty of things we should be grateful for. In doing these exercises you’ll learn just how much others have cared for you and, I hope, how you might do things to make them grateful for you.
If you’re interested in learning more about Naikan, I discovered this from Brett McKay’s The Art of Manliness podcast and the teaching of Gregg Krech, summarized in his book, “Naikan: Gratitude, Grace, and the Japanese Art of Self-Reflection.”
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected] .
Long-haul COVID-19 cases rise as stigma of chronic fatigue taunts
When Margot Gage-Witvliet began feeling run down after her family returned from a trip to the Netherlands in late February 2020, she initially chalked up her symptoms to jet lag. Three days later, however, her situation went from concerning to alarming as she struggled to breathe. “It felt like there was an elephant sitting on my chest,” she said.
Her husband and daughters also became ill with COVID-19, but Ms. Gage-Witvliet was the only one in her family who didn’t get better. After an early improvement, a rare coronavirus-induced tonic-clonic seizure in early April sent her spiraling back down. Ms. Gage-Witvliet spent the next several weeks in bed with the curtains drawn, unable to tolerate light or sound.
Today, Ms. Gage-Witvliet’s life looks nothing like it did 6 months ago when she first got sick. As one of COVID-19’s so called long-haulers, she continues to struggle with crushing fatigue, brain fog, and headaches – symptoms that worsen when she pushes herself to do more. Across the country, as many as 1 in 10 COVID-19 patients are reporting illnesses that continue for weeks and months after their initial diagnosis. Nearly all report neurologic issues like Ms. Gage-Witvliet, as well as shortness of breath and psychiatric concerns.
For Avindra Nath, MD, a neurologist at the National Institutes of Health, the experience of these long-haul COVID-19 patients feels familiar and reminds him of myalgic encephalomyelitis, also known as chronic fatigue syndrome.
Dr. Nath has long been interested in the lingering neurologic issues connected to chronic fatigue. An estimated three-quarters of all patients with chronic fatigue syndrome report that their symptoms started after a viral infection, and they suffer unrelenting exhaustion, difficulties regulating pulse and blood pressure, aches and pains, and brain fog. When Dr. Nath first read about the novel coronavirus, he began to worry that the virus would trigger symptoms in a subset of those infected. Hearing about the experiences of long-haulers like Ms. Gage-Witvliet raised his suspicions even more.
Unlike COVID-19 long-haulers, however, many patients with chronic fatigue syndrome go at least a year with these symptoms before receiving a diagnosis, according to a British survey. That means researchers have had few opportunities to study the early stages of the syndrome. “When we see patients with myalgic encephalomyelitis, whatever infection they might have had occurred in the remote past, so there’s no way for us to know how they got infected with it, what the infection was, or what the effects of it were in that early phase. We’re seeing them 2 years afterward,” Dr. Nath said.
Dr. Nath quickly realized that studying patients like Ms. Gage-Witvliet would give physicians and scientists a unique opportunity to understand not only long-term outcomes of COVID-19 infections, but also other postviral syndromes, including chronic fatigue syndrome at their earliest stages. It’s why Dr. Nath has spent the past several months scrambling to launch two NIH studies to examine the phenomenon.
Although Dr. Nath said that the parallels between COVID-19 long-haulers and those with chronic fatigue syndrome are obvious, he cautions against assuming that they are the same phenomenon. Some long-haulers might simply be taking a much slower path to recovery, or they might have a condition that looks similar on the surface but differs from chronic fatigue syndrome on a molecular level. But even if Dr. Nath fails to see links to chronic fatigue syndrome, with more than 92.5 million documented cases of COVID-19 around the world, the work will be relevant to the substantial number of infected individuals who don’t recover quickly.
“With so many people having exposure to the same virus over a similar time period, we really have the opportunity to look at these manifestations and at the very least to understand postviral syndromes,” said Mady Hornig, MD, a psychiatrist at Columbia University, New York.
The origins of chronic fatigue syndrome date back to 1985, when the Centers for Disease Control and Prevention received a request from two physicians – Paul Cheney, MD, and Daniel Peterson, MD – to investigate a mysterious disease outbreak in Nevada. In November 1984, residents in and around the idyllic vacation spot of Incline Village, a small town tucked into the north shore of Lake Tahoe, had begun reporting flu-like symptoms that persisted for weeks, even months. The doctors had searched high and low for a cause, but they couldn’t figure out what was making their patients sick.
They reported a range of symptoms – including muscle aches and pains, low-grade fevers, sore throats, and headaches – but everyone said that crippling fatigue was the most debilitating issue. This wasn’t the kind of fatigue that could be cured by a nap or even a long holiday. No matter how much their patients slept – and some were almost completely bedbound – their fatigue didn’t abate. What’s more, the fatigue got worse whenever they tried to push themselves to do more. Puzzled, the CDC sent two epidemic intelligence service (EIS) officers to try to get to the bottom of what might be happening.
Muscle aches and pains with crippling fatigue
After their visit to Incline Village, however, the CDC was just as perplexed as Dr. Cheney and Dr. Peterson. Many of the people with the condition reported flu-like symptoms right around the time they first got sick, and the physicians’ leading hypothesis was that the outbreak and its lasting symptoms were caused by chronic Epstein-Barr virus infection. But neither the CDC nor anyone else could identify the infection or any other microbial cause. The two EIS officers duly wrote up a report for the CDC’s flagship publication, Morbidity and Mortality Weekly ReportI, titled “Chronic Fatigue Possibly Related to Epstein-Barr Virus – Nevada”.
That investigators focused on the fatigue aspect made sense, says Leonard A. Jason, PhD, professor of psychology at DePaul University and director of the Center for Community Research, both in Chicago, because it was one of the few symptoms shared by all the individuals studied and it was also the most debilitating. But that focus – and the name “chronic fatigue syndrome” – led to broad public dismissal of the condition’s severity, as did an editorial note in MMWR urging physicians to look for “more definable, and possibly treatable, conditions.” Subsequent research failed to confirm a specific link to the Epstein-Barr virus, which only added to the condition’s phony reputation. Rather than being considered a potentially disabling illness, it was disregarded as a “yuppie flu” or a fancy name for malingering.
“It’s not a surprise that patients are being dismissed because there’s already this sort of grandfathered-in sense that fatigue is not real,” said Jennifer Frankovich, MD, a pediatric rheumatologist at Stanford (Calif.) University’s Lucile Packard Children’s Hospital in Palo Alto. “I’m sure that’s frustrating for them to be tired and then to have the clinician not believe them or dismiss them or think they’re making it up. It would be more helpful to the families to say: ‘You know what, we don’t know, we do not have the answer, and we believe you.’ ”
A syndrome’s shame
As time passed, patient advocacy groups began pushing back against the negative way the condition was being perceived. This criticism came as organizations like the CDC worked to develop a set of diagnostic criteria that researchers and clinicians dealing with chronic fatigue syndrome could use. With such a heterogeneous group of patients and symptoms, the task was no small challenge. The discussions, which took place over nearly 2 decades, played a key role in helping scientists home in on the single factor that was central to chronic fatigue: postexertional malaise.
“This is quite unique for chronic fatigue syndrome. With other diseases, yes, you may have fatigue as one of the components of the disease, but postexertional fatigue is very specific,” said Alain Moreau, PhD, a molecular biologist at the University of Montreal.
Of course, plenty of people have pushed themselves too hard physically and paid the price the next day. But those with chronic fatigue syndrome weren’t running marathons. To them, exertion could be anything from getting the mail to reading a book. Nor could the resulting exhaustion be resolved by an afternoon on the couch or a long vacation.
“If they do these activities, they can crash for weeks, even months,” Dr. Moreau said. It was deep, persistent, and – for 40% of those with chronic fatigue syndrome – disabling. In 2015, a study group from the Institute of Medicine proposed renaming chronic fatigue to “systemic exercise intolerance disease” because of the centrality of this symptom. Although that effort mostly stalled, their report did bring the condition out of its historic place as a scientific backwater. What resulted was an uptick in research on chronic fatigue syndrome, which helped define some of the physiological issues that either contribute to or result from the condition.
Researchers had long known about the link between infection and fatigue, said Dr. Frankovich. Work included mysterious outbreaks like the one in Lake Tahoe and well-documented issues like the wave of encephalitis lethargica (a condition that leaves patients in an almost vegetative state) that followed the 1918 H1N1 influenza pandemic.
“As a clinician, when you see someone who comes in with a chronic infection, they’re tired. I think that’s why, in the chronic-fatigue world, people are desperately looking for the infection so we can treat it, and maybe these poor suffering people will feel better,” Dr. Frankovich added. Then the pandemic struck, giving him yet another opportunity to study postviral syndromes.
Immunologic symptoms
Given the close link between a nonspecific viral illness and the onset of symptoms in chronic fatigue syndrome, scientists like Dr. Hornig opted to focus on immunologic symptoms. In a 2015 analysis published in Science, Dr. Hornig and colleagues showed that immune problems can be found in the earliest stages of chronic fatigue syndrome, and that they change as the illness progresses. Patients who had been sick for less than 3 years showed significant increases in levels of both pro- and anti-inflammatory cytokines, and the factor most strongly correlated to this inability to regulate cytokine levels was the duration of symptoms, not their severity. A series of other studies also revealed problems with regulation of the immune system, although no one could show what might have set these problems in motion.
Other researchers found signs of mitochondrial dysfunction in those with chronic fatigue syndrome. Because mitochondria make energy for cells, it wasn’t an intellectual stretch to believe that glitches in this process could contribute to fatigue. As early as 1991, scientists had discovered signs of mitochondrial degeneration in muscle biopsies from people with chronic fatigue syndrome. Subsequent studies showed that those affected by chronic fatigue were missing segments of mitochondrial DNA and had significantly reduced levels of mitochondrial activity. Although exercise normally improves mitochondrial functioning, the opposite appears to happen in chronic fatigue.
To Dr. Nath, these dual hypotheses aren’t necessarily mutually exclusive. Some studies have hinted that infection with the common human herpesvirus–6 (HHV-6) can lead to an autoimmune condition in which the body makes antibodies against the mitochondria. Mitochondria also play a key role in the ability of the innate immune system to produce interferon and other proinflammatory cytokines. It might also be that the link between immune and mitochondrial problems is more convoluted than originally thought, or that the two systems are affected independent of one another, Dr. Nath said.
Finding answers, especially those that could lead to potential treatments, wouldn’t be easy, however. In 2016, the NIH launched an in-depth study of a small number of individuals with chronic fatigue, hoping to find clues about what the condition was and how it might be treated.
For scientists like Dr. Nath, the NIH study provided a way to get at the underlying biology of chronic fatigue syndrome. Then the pandemic struck, giving him yet another opportunity to study postviral syndromes.
Chronic post-SARS syndrome
In March 2020, retired physician Harvey Moldofsky, MD, began receiving inquiries about a 2011 study he and his colleague, John Patcai, MD, had published in BMC Neurology about something they dubbed “chronic post-SARS syndrome.” The small case-control study, which involved mainly health care workers in Toronto, received little attention when it was first published, but with COVID-19, it was suddenly relevant.
Early clusters of similar cases in Miami made local physicians desperate for Dr. Moldofsky’s expertise. Luckily, he was nearby; he had fled the frigid Canadian winter for the warmth of Sarasota, Fla.
“I had people from various countries around the world writing to me and asking what they should do. And of course I don’t have any answers,” he said. But the study contained one of the world’s only references to the syndrome.
In 2003, a woman arrived in Toronto from Hong Kong. She didn’t know it at the time, but her preairport stay at the Hotel Metropole had infected her with the first SARS (severe acute respiratory syndrome) coronavirus. Her subsequent hospitalization in Toronto sparked a city-wide outbreak of SARS in which 273 people became ill and 44 died. Many of those affected were health care workers, including nurses and respiratory therapists. Although most eventually returned to work, a subset couldn’t. They complained of energy-sapping fatigue, poor sleep, brain fog, and assorted body aches and pains that persisted for more than 18 months. The aches and pains brought them to the attention of Dr. Moldofsky, then director of the Centre for the Study of Pain at the University of Toronto.
His primary interest at the time was fibromyalgia, which caused symptoms similar to those reported by the original SARS long-haulers. Intrigued, Dr. Moldofsky agreed to take a look. Their chest x-rays were clear and the nurses showed no signs of lingering viral infection. Dr. Moldofsky could see that the nurses were ill and suffering, but no lab tests or anything else could identify what was causing their symptoms.
In 2011, Dr. Moldofsky and Dr. Patcai found a strong overlap between chronic SARS, fibromyalgia, and chronic fatigue syndrome when they compared 22 patients with long-term SARS issues with 21 who had fibromyalgia. “Their problems are exactly the same. They have strange symptoms and nobody can figure out what they’re about. And these symptoms are aches and pains, and they have trouble thinking and concentrating,” Dr. Moldofsky said. Reports of COVID-19 long-haulers didn’t surprise Dr. Moldofsky, and he immediately recognized that Nath’s intention to follow these patients could provide insights into both fibromyalgia and chronic fatigue syndrome.
That’s exactly what Dr. Nath is proposing with the two NIH studies. One will focus solely on the neurologic impacts of COVID-19, including stroke, loss of taste and smell, and brain fog. The other will bring patients who have had COVID-19 symptoms for at least 6 months to the NIH Clinical Center for an inpatient stay during which they will undergo detailed physiologic tests.
Scientists around the world are launching their own post–COVID-19 studies. Dr. Moreau’s group in Montreal has laid the groundwork for such an endeavor, and the CoroNerve group in the United Kingdom is monitoring neurologic complications from the coronavirus. Many of them have the same goals as the NIH studies: Leverage the large number of COVID-19 long-haulers to better understand the earliest stages of postviral syndrome.
“At this juncture, after all the reports that we’ve seen so far, I think it’s very unlikely that there will be no relationship whatsoever between COVID-19 and chronic fatigue syndrome,” Dr. Hornig said. “I think there certainly will be some, but again, what’s the scope, what’s the size? And then, of course, even more importantly, if it is happening, what is the mechanism and how is it happening?”
For people like Ms. Gage-Witvliet, the answers can’t come soon enough. For the first time in more than a decade, the full-time professor of epidemiology didn’t prepare to teach this year because she simply can’t. It’s too taxing for her brain to deal with impromptu student questions. Ms. Gage-Witvliet hopes that, by sharing her own experiences with post COVID-19, she can help others.
“In my work, I use data to give a voice to people who don’t have a voice,” she said. “Now, I am one of those people.”
A version of this article first appeared on Medscape.com.
When Margot Gage-Witvliet began feeling run down after her family returned from a trip to the Netherlands in late February 2020, she initially chalked up her symptoms to jet lag. Three days later, however, her situation went from concerning to alarming as she struggled to breathe. “It felt like there was an elephant sitting on my chest,” she said.
Her husband and daughters also became ill with COVID-19, but Ms. Gage-Witvliet was the only one in her family who didn’t get better. After an early improvement, a rare coronavirus-induced tonic-clonic seizure in early April sent her spiraling back down. Ms. Gage-Witvliet spent the next several weeks in bed with the curtains drawn, unable to tolerate light or sound.
Today, Ms. Gage-Witvliet’s life looks nothing like it did 6 months ago when she first got sick. As one of COVID-19’s so called long-haulers, she continues to struggle with crushing fatigue, brain fog, and headaches – symptoms that worsen when she pushes herself to do more. Across the country, as many as 1 in 10 COVID-19 patients are reporting illnesses that continue for weeks and months after their initial diagnosis. Nearly all report neurologic issues like Ms. Gage-Witvliet, as well as shortness of breath and psychiatric concerns.
For Avindra Nath, MD, a neurologist at the National Institutes of Health, the experience of these long-haul COVID-19 patients feels familiar and reminds him of myalgic encephalomyelitis, also known as chronic fatigue syndrome.
Dr. Nath has long been interested in the lingering neurologic issues connected to chronic fatigue. An estimated three-quarters of all patients with chronic fatigue syndrome report that their symptoms started after a viral infection, and they suffer unrelenting exhaustion, difficulties regulating pulse and blood pressure, aches and pains, and brain fog. When Dr. Nath first read about the novel coronavirus, he began to worry that the virus would trigger symptoms in a subset of those infected. Hearing about the experiences of long-haulers like Ms. Gage-Witvliet raised his suspicions even more.
Unlike COVID-19 long-haulers, however, many patients with chronic fatigue syndrome go at least a year with these symptoms before receiving a diagnosis, according to a British survey. That means researchers have had few opportunities to study the early stages of the syndrome. “When we see patients with myalgic encephalomyelitis, whatever infection they might have had occurred in the remote past, so there’s no way for us to know how they got infected with it, what the infection was, or what the effects of it were in that early phase. We’re seeing them 2 years afterward,” Dr. Nath said.
Dr. Nath quickly realized that studying patients like Ms. Gage-Witvliet would give physicians and scientists a unique opportunity to understand not only long-term outcomes of COVID-19 infections, but also other postviral syndromes, including chronic fatigue syndrome at their earliest stages. It’s why Dr. Nath has spent the past several months scrambling to launch two NIH studies to examine the phenomenon.
Although Dr. Nath said that the parallels between COVID-19 long-haulers and those with chronic fatigue syndrome are obvious, he cautions against assuming that they are the same phenomenon. Some long-haulers might simply be taking a much slower path to recovery, or they might have a condition that looks similar on the surface but differs from chronic fatigue syndrome on a molecular level. But even if Dr. Nath fails to see links to chronic fatigue syndrome, with more than 92.5 million documented cases of COVID-19 around the world, the work will be relevant to the substantial number of infected individuals who don’t recover quickly.
“With so many people having exposure to the same virus over a similar time period, we really have the opportunity to look at these manifestations and at the very least to understand postviral syndromes,” said Mady Hornig, MD, a psychiatrist at Columbia University, New York.
The origins of chronic fatigue syndrome date back to 1985, when the Centers for Disease Control and Prevention received a request from two physicians – Paul Cheney, MD, and Daniel Peterson, MD – to investigate a mysterious disease outbreak in Nevada. In November 1984, residents in and around the idyllic vacation spot of Incline Village, a small town tucked into the north shore of Lake Tahoe, had begun reporting flu-like symptoms that persisted for weeks, even months. The doctors had searched high and low for a cause, but they couldn’t figure out what was making their patients sick.
They reported a range of symptoms – including muscle aches and pains, low-grade fevers, sore throats, and headaches – but everyone said that crippling fatigue was the most debilitating issue. This wasn’t the kind of fatigue that could be cured by a nap or even a long holiday. No matter how much their patients slept – and some were almost completely bedbound – their fatigue didn’t abate. What’s more, the fatigue got worse whenever they tried to push themselves to do more. Puzzled, the CDC sent two epidemic intelligence service (EIS) officers to try to get to the bottom of what might be happening.
Muscle aches and pains with crippling fatigue
After their visit to Incline Village, however, the CDC was just as perplexed as Dr. Cheney and Dr. Peterson. Many of the people with the condition reported flu-like symptoms right around the time they first got sick, and the physicians’ leading hypothesis was that the outbreak and its lasting symptoms were caused by chronic Epstein-Barr virus infection. But neither the CDC nor anyone else could identify the infection or any other microbial cause. The two EIS officers duly wrote up a report for the CDC’s flagship publication, Morbidity and Mortality Weekly ReportI, titled “Chronic Fatigue Possibly Related to Epstein-Barr Virus – Nevada”.
That investigators focused on the fatigue aspect made sense, says Leonard A. Jason, PhD, professor of psychology at DePaul University and director of the Center for Community Research, both in Chicago, because it was one of the few symptoms shared by all the individuals studied and it was also the most debilitating. But that focus – and the name “chronic fatigue syndrome” – led to broad public dismissal of the condition’s severity, as did an editorial note in MMWR urging physicians to look for “more definable, and possibly treatable, conditions.” Subsequent research failed to confirm a specific link to the Epstein-Barr virus, which only added to the condition’s phony reputation. Rather than being considered a potentially disabling illness, it was disregarded as a “yuppie flu” or a fancy name for malingering.
“It’s not a surprise that patients are being dismissed because there’s already this sort of grandfathered-in sense that fatigue is not real,” said Jennifer Frankovich, MD, a pediatric rheumatologist at Stanford (Calif.) University’s Lucile Packard Children’s Hospital in Palo Alto. “I’m sure that’s frustrating for them to be tired and then to have the clinician not believe them or dismiss them or think they’re making it up. It would be more helpful to the families to say: ‘You know what, we don’t know, we do not have the answer, and we believe you.’ ”
A syndrome’s shame
As time passed, patient advocacy groups began pushing back against the negative way the condition was being perceived. This criticism came as organizations like the CDC worked to develop a set of diagnostic criteria that researchers and clinicians dealing with chronic fatigue syndrome could use. With such a heterogeneous group of patients and symptoms, the task was no small challenge. The discussions, which took place over nearly 2 decades, played a key role in helping scientists home in on the single factor that was central to chronic fatigue: postexertional malaise.
“This is quite unique for chronic fatigue syndrome. With other diseases, yes, you may have fatigue as one of the components of the disease, but postexertional fatigue is very specific,” said Alain Moreau, PhD, a molecular biologist at the University of Montreal.
Of course, plenty of people have pushed themselves too hard physically and paid the price the next day. But those with chronic fatigue syndrome weren’t running marathons. To them, exertion could be anything from getting the mail to reading a book. Nor could the resulting exhaustion be resolved by an afternoon on the couch or a long vacation.
“If they do these activities, they can crash for weeks, even months,” Dr. Moreau said. It was deep, persistent, and – for 40% of those with chronic fatigue syndrome – disabling. In 2015, a study group from the Institute of Medicine proposed renaming chronic fatigue to “systemic exercise intolerance disease” because of the centrality of this symptom. Although that effort mostly stalled, their report did bring the condition out of its historic place as a scientific backwater. What resulted was an uptick in research on chronic fatigue syndrome, which helped define some of the physiological issues that either contribute to or result from the condition.
Researchers had long known about the link between infection and fatigue, said Dr. Frankovich. Work included mysterious outbreaks like the one in Lake Tahoe and well-documented issues like the wave of encephalitis lethargica (a condition that leaves patients in an almost vegetative state) that followed the 1918 H1N1 influenza pandemic.
“As a clinician, when you see someone who comes in with a chronic infection, they’re tired. I think that’s why, in the chronic-fatigue world, people are desperately looking for the infection so we can treat it, and maybe these poor suffering people will feel better,” Dr. Frankovich added. Then the pandemic struck, giving him yet another opportunity to study postviral syndromes.
Immunologic symptoms
Given the close link between a nonspecific viral illness and the onset of symptoms in chronic fatigue syndrome, scientists like Dr. Hornig opted to focus on immunologic symptoms. In a 2015 analysis published in Science, Dr. Hornig and colleagues showed that immune problems can be found in the earliest stages of chronic fatigue syndrome, and that they change as the illness progresses. Patients who had been sick for less than 3 years showed significant increases in levels of both pro- and anti-inflammatory cytokines, and the factor most strongly correlated to this inability to regulate cytokine levels was the duration of symptoms, not their severity. A series of other studies also revealed problems with regulation of the immune system, although no one could show what might have set these problems in motion.
Other researchers found signs of mitochondrial dysfunction in those with chronic fatigue syndrome. Because mitochondria make energy for cells, it wasn’t an intellectual stretch to believe that glitches in this process could contribute to fatigue. As early as 1991, scientists had discovered signs of mitochondrial degeneration in muscle biopsies from people with chronic fatigue syndrome. Subsequent studies showed that those affected by chronic fatigue were missing segments of mitochondrial DNA and had significantly reduced levels of mitochondrial activity. Although exercise normally improves mitochondrial functioning, the opposite appears to happen in chronic fatigue.
To Dr. Nath, these dual hypotheses aren’t necessarily mutually exclusive. Some studies have hinted that infection with the common human herpesvirus–6 (HHV-6) can lead to an autoimmune condition in which the body makes antibodies against the mitochondria. Mitochondria also play a key role in the ability of the innate immune system to produce interferon and other proinflammatory cytokines. It might also be that the link between immune and mitochondrial problems is more convoluted than originally thought, or that the two systems are affected independent of one another, Dr. Nath said.
Finding answers, especially those that could lead to potential treatments, wouldn’t be easy, however. In 2016, the NIH launched an in-depth study of a small number of individuals with chronic fatigue, hoping to find clues about what the condition was and how it might be treated.
For scientists like Dr. Nath, the NIH study provided a way to get at the underlying biology of chronic fatigue syndrome. Then the pandemic struck, giving him yet another opportunity to study postviral syndromes.
Chronic post-SARS syndrome
In March 2020, retired physician Harvey Moldofsky, MD, began receiving inquiries about a 2011 study he and his colleague, John Patcai, MD, had published in BMC Neurology about something they dubbed “chronic post-SARS syndrome.” The small case-control study, which involved mainly health care workers in Toronto, received little attention when it was first published, but with COVID-19, it was suddenly relevant.
Early clusters of similar cases in Miami made local physicians desperate for Dr. Moldofsky’s expertise. Luckily, he was nearby; he had fled the frigid Canadian winter for the warmth of Sarasota, Fla.
“I had people from various countries around the world writing to me and asking what they should do. And of course I don’t have any answers,” he said. But the study contained one of the world’s only references to the syndrome.
In 2003, a woman arrived in Toronto from Hong Kong. She didn’t know it at the time, but her preairport stay at the Hotel Metropole had infected her with the first SARS (severe acute respiratory syndrome) coronavirus. Her subsequent hospitalization in Toronto sparked a city-wide outbreak of SARS in which 273 people became ill and 44 died. Many of those affected were health care workers, including nurses and respiratory therapists. Although most eventually returned to work, a subset couldn’t. They complained of energy-sapping fatigue, poor sleep, brain fog, and assorted body aches and pains that persisted for more than 18 months. The aches and pains brought them to the attention of Dr. Moldofsky, then director of the Centre for the Study of Pain at the University of Toronto.
His primary interest at the time was fibromyalgia, which caused symptoms similar to those reported by the original SARS long-haulers. Intrigued, Dr. Moldofsky agreed to take a look. Their chest x-rays were clear and the nurses showed no signs of lingering viral infection. Dr. Moldofsky could see that the nurses were ill and suffering, but no lab tests or anything else could identify what was causing their symptoms.
In 2011, Dr. Moldofsky and Dr. Patcai found a strong overlap between chronic SARS, fibromyalgia, and chronic fatigue syndrome when they compared 22 patients with long-term SARS issues with 21 who had fibromyalgia. “Their problems are exactly the same. They have strange symptoms and nobody can figure out what they’re about. And these symptoms are aches and pains, and they have trouble thinking and concentrating,” Dr. Moldofsky said. Reports of COVID-19 long-haulers didn’t surprise Dr. Moldofsky, and he immediately recognized that Nath’s intention to follow these patients could provide insights into both fibromyalgia and chronic fatigue syndrome.
That’s exactly what Dr. Nath is proposing with the two NIH studies. One will focus solely on the neurologic impacts of COVID-19, including stroke, loss of taste and smell, and brain fog. The other will bring patients who have had COVID-19 symptoms for at least 6 months to the NIH Clinical Center for an inpatient stay during which they will undergo detailed physiologic tests.
Scientists around the world are launching their own post–COVID-19 studies. Dr. Moreau’s group in Montreal has laid the groundwork for such an endeavor, and the CoroNerve group in the United Kingdom is monitoring neurologic complications from the coronavirus. Many of them have the same goals as the NIH studies: Leverage the large number of COVID-19 long-haulers to better understand the earliest stages of postviral syndrome.
“At this juncture, after all the reports that we’ve seen so far, I think it’s very unlikely that there will be no relationship whatsoever between COVID-19 and chronic fatigue syndrome,” Dr. Hornig said. “I think there certainly will be some, but again, what’s the scope, what’s the size? And then, of course, even more importantly, if it is happening, what is the mechanism and how is it happening?”
For people like Ms. Gage-Witvliet, the answers can’t come soon enough. For the first time in more than a decade, the full-time professor of epidemiology didn’t prepare to teach this year because she simply can’t. It’s too taxing for her brain to deal with impromptu student questions. Ms. Gage-Witvliet hopes that, by sharing her own experiences with post COVID-19, she can help others.
“In my work, I use data to give a voice to people who don’t have a voice,” she said. “Now, I am one of those people.”
A version of this article first appeared on Medscape.com.
When Margot Gage-Witvliet began feeling run down after her family returned from a trip to the Netherlands in late February 2020, she initially chalked up her symptoms to jet lag. Three days later, however, her situation went from concerning to alarming as she struggled to breathe. “It felt like there was an elephant sitting on my chest,” she said.
Her husband and daughters also became ill with COVID-19, but Ms. Gage-Witvliet was the only one in her family who didn’t get better. After an early improvement, a rare coronavirus-induced tonic-clonic seizure in early April sent her spiraling back down. Ms. Gage-Witvliet spent the next several weeks in bed with the curtains drawn, unable to tolerate light or sound.
Today, Ms. Gage-Witvliet’s life looks nothing like it did 6 months ago when she first got sick. As one of COVID-19’s so called long-haulers, she continues to struggle with crushing fatigue, brain fog, and headaches – symptoms that worsen when she pushes herself to do more. Across the country, as many as 1 in 10 COVID-19 patients are reporting illnesses that continue for weeks and months after their initial diagnosis. Nearly all report neurologic issues like Ms. Gage-Witvliet, as well as shortness of breath and psychiatric concerns.
For Avindra Nath, MD, a neurologist at the National Institutes of Health, the experience of these long-haul COVID-19 patients feels familiar and reminds him of myalgic encephalomyelitis, also known as chronic fatigue syndrome.
Dr. Nath has long been interested in the lingering neurologic issues connected to chronic fatigue. An estimated three-quarters of all patients with chronic fatigue syndrome report that their symptoms started after a viral infection, and they suffer unrelenting exhaustion, difficulties regulating pulse and blood pressure, aches and pains, and brain fog. When Dr. Nath first read about the novel coronavirus, he began to worry that the virus would trigger symptoms in a subset of those infected. Hearing about the experiences of long-haulers like Ms. Gage-Witvliet raised his suspicions even more.
Unlike COVID-19 long-haulers, however, many patients with chronic fatigue syndrome go at least a year with these symptoms before receiving a diagnosis, according to a British survey. That means researchers have had few opportunities to study the early stages of the syndrome. “When we see patients with myalgic encephalomyelitis, whatever infection they might have had occurred in the remote past, so there’s no way for us to know how they got infected with it, what the infection was, or what the effects of it were in that early phase. We’re seeing them 2 years afterward,” Dr. Nath said.
Dr. Nath quickly realized that studying patients like Ms. Gage-Witvliet would give physicians and scientists a unique opportunity to understand not only long-term outcomes of COVID-19 infections, but also other postviral syndromes, including chronic fatigue syndrome at their earliest stages. It’s why Dr. Nath has spent the past several months scrambling to launch two NIH studies to examine the phenomenon.
Although Dr. Nath said that the parallels between COVID-19 long-haulers and those with chronic fatigue syndrome are obvious, he cautions against assuming that they are the same phenomenon. Some long-haulers might simply be taking a much slower path to recovery, or they might have a condition that looks similar on the surface but differs from chronic fatigue syndrome on a molecular level. But even if Dr. Nath fails to see links to chronic fatigue syndrome, with more than 92.5 million documented cases of COVID-19 around the world, the work will be relevant to the substantial number of infected individuals who don’t recover quickly.
“With so many people having exposure to the same virus over a similar time period, we really have the opportunity to look at these manifestations and at the very least to understand postviral syndromes,” said Mady Hornig, MD, a psychiatrist at Columbia University, New York.
The origins of chronic fatigue syndrome date back to 1985, when the Centers for Disease Control and Prevention received a request from two physicians – Paul Cheney, MD, and Daniel Peterson, MD – to investigate a mysterious disease outbreak in Nevada. In November 1984, residents in and around the idyllic vacation spot of Incline Village, a small town tucked into the north shore of Lake Tahoe, had begun reporting flu-like symptoms that persisted for weeks, even months. The doctors had searched high and low for a cause, but they couldn’t figure out what was making their patients sick.
They reported a range of symptoms – including muscle aches and pains, low-grade fevers, sore throats, and headaches – but everyone said that crippling fatigue was the most debilitating issue. This wasn’t the kind of fatigue that could be cured by a nap or even a long holiday. No matter how much their patients slept – and some were almost completely bedbound – their fatigue didn’t abate. What’s more, the fatigue got worse whenever they tried to push themselves to do more. Puzzled, the CDC sent two epidemic intelligence service (EIS) officers to try to get to the bottom of what might be happening.
Muscle aches and pains with crippling fatigue
After their visit to Incline Village, however, the CDC was just as perplexed as Dr. Cheney and Dr. Peterson. Many of the people with the condition reported flu-like symptoms right around the time they first got sick, and the physicians’ leading hypothesis was that the outbreak and its lasting symptoms were caused by chronic Epstein-Barr virus infection. But neither the CDC nor anyone else could identify the infection or any other microbial cause. The two EIS officers duly wrote up a report for the CDC’s flagship publication, Morbidity and Mortality Weekly ReportI, titled “Chronic Fatigue Possibly Related to Epstein-Barr Virus – Nevada”.
That investigators focused on the fatigue aspect made sense, says Leonard A. Jason, PhD, professor of psychology at DePaul University and director of the Center for Community Research, both in Chicago, because it was one of the few symptoms shared by all the individuals studied and it was also the most debilitating. But that focus – and the name “chronic fatigue syndrome” – led to broad public dismissal of the condition’s severity, as did an editorial note in MMWR urging physicians to look for “more definable, and possibly treatable, conditions.” Subsequent research failed to confirm a specific link to the Epstein-Barr virus, which only added to the condition’s phony reputation. Rather than being considered a potentially disabling illness, it was disregarded as a “yuppie flu” or a fancy name for malingering.
“It’s not a surprise that patients are being dismissed because there’s already this sort of grandfathered-in sense that fatigue is not real,” said Jennifer Frankovich, MD, a pediatric rheumatologist at Stanford (Calif.) University’s Lucile Packard Children’s Hospital in Palo Alto. “I’m sure that’s frustrating for them to be tired and then to have the clinician not believe them or dismiss them or think they’re making it up. It would be more helpful to the families to say: ‘You know what, we don’t know, we do not have the answer, and we believe you.’ ”
A syndrome’s shame
As time passed, patient advocacy groups began pushing back against the negative way the condition was being perceived. This criticism came as organizations like the CDC worked to develop a set of diagnostic criteria that researchers and clinicians dealing with chronic fatigue syndrome could use. With such a heterogeneous group of patients and symptoms, the task was no small challenge. The discussions, which took place over nearly 2 decades, played a key role in helping scientists home in on the single factor that was central to chronic fatigue: postexertional malaise.
“This is quite unique for chronic fatigue syndrome. With other diseases, yes, you may have fatigue as one of the components of the disease, but postexertional fatigue is very specific,” said Alain Moreau, PhD, a molecular biologist at the University of Montreal.
Of course, plenty of people have pushed themselves too hard physically and paid the price the next day. But those with chronic fatigue syndrome weren’t running marathons. To them, exertion could be anything from getting the mail to reading a book. Nor could the resulting exhaustion be resolved by an afternoon on the couch or a long vacation.
“If they do these activities, they can crash for weeks, even months,” Dr. Moreau said. It was deep, persistent, and – for 40% of those with chronic fatigue syndrome – disabling. In 2015, a study group from the Institute of Medicine proposed renaming chronic fatigue to “systemic exercise intolerance disease” because of the centrality of this symptom. Although that effort mostly stalled, their report did bring the condition out of its historic place as a scientific backwater. What resulted was an uptick in research on chronic fatigue syndrome, which helped define some of the physiological issues that either contribute to or result from the condition.
Researchers had long known about the link between infection and fatigue, said Dr. Frankovich. Work included mysterious outbreaks like the one in Lake Tahoe and well-documented issues like the wave of encephalitis lethargica (a condition that leaves patients in an almost vegetative state) that followed the 1918 H1N1 influenza pandemic.
“As a clinician, when you see someone who comes in with a chronic infection, they’re tired. I think that’s why, in the chronic-fatigue world, people are desperately looking for the infection so we can treat it, and maybe these poor suffering people will feel better,” Dr. Frankovich added. Then the pandemic struck, giving him yet another opportunity to study postviral syndromes.
Immunologic symptoms
Given the close link between a nonspecific viral illness and the onset of symptoms in chronic fatigue syndrome, scientists like Dr. Hornig opted to focus on immunologic symptoms. In a 2015 analysis published in Science, Dr. Hornig and colleagues showed that immune problems can be found in the earliest stages of chronic fatigue syndrome, and that they change as the illness progresses. Patients who had been sick for less than 3 years showed significant increases in levels of both pro- and anti-inflammatory cytokines, and the factor most strongly correlated to this inability to regulate cytokine levels was the duration of symptoms, not their severity. A series of other studies also revealed problems with regulation of the immune system, although no one could show what might have set these problems in motion.
Other researchers found signs of mitochondrial dysfunction in those with chronic fatigue syndrome. Because mitochondria make energy for cells, it wasn’t an intellectual stretch to believe that glitches in this process could contribute to fatigue. As early as 1991, scientists had discovered signs of mitochondrial degeneration in muscle biopsies from people with chronic fatigue syndrome. Subsequent studies showed that those affected by chronic fatigue were missing segments of mitochondrial DNA and had significantly reduced levels of mitochondrial activity. Although exercise normally improves mitochondrial functioning, the opposite appears to happen in chronic fatigue.
To Dr. Nath, these dual hypotheses aren’t necessarily mutually exclusive. Some studies have hinted that infection with the common human herpesvirus–6 (HHV-6) can lead to an autoimmune condition in which the body makes antibodies against the mitochondria. Mitochondria also play a key role in the ability of the innate immune system to produce interferon and other proinflammatory cytokines. It might also be that the link between immune and mitochondrial problems is more convoluted than originally thought, or that the two systems are affected independent of one another, Dr. Nath said.
Finding answers, especially those that could lead to potential treatments, wouldn’t be easy, however. In 2016, the NIH launched an in-depth study of a small number of individuals with chronic fatigue, hoping to find clues about what the condition was and how it might be treated.
For scientists like Dr. Nath, the NIH study provided a way to get at the underlying biology of chronic fatigue syndrome. Then the pandemic struck, giving him yet another opportunity to study postviral syndromes.
Chronic post-SARS syndrome
In March 2020, retired physician Harvey Moldofsky, MD, began receiving inquiries about a 2011 study he and his colleague, John Patcai, MD, had published in BMC Neurology about something they dubbed “chronic post-SARS syndrome.” The small case-control study, which involved mainly health care workers in Toronto, received little attention when it was first published, but with COVID-19, it was suddenly relevant.
Early clusters of similar cases in Miami made local physicians desperate for Dr. Moldofsky’s expertise. Luckily, he was nearby; he had fled the frigid Canadian winter for the warmth of Sarasota, Fla.
“I had people from various countries around the world writing to me and asking what they should do. And of course I don’t have any answers,” he said. But the study contained one of the world’s only references to the syndrome.
In 2003, a woman arrived in Toronto from Hong Kong. She didn’t know it at the time, but her preairport stay at the Hotel Metropole had infected her with the first SARS (severe acute respiratory syndrome) coronavirus. Her subsequent hospitalization in Toronto sparked a city-wide outbreak of SARS in which 273 people became ill and 44 died. Many of those affected were health care workers, including nurses and respiratory therapists. Although most eventually returned to work, a subset couldn’t. They complained of energy-sapping fatigue, poor sleep, brain fog, and assorted body aches and pains that persisted for more than 18 months. The aches and pains brought them to the attention of Dr. Moldofsky, then director of the Centre for the Study of Pain at the University of Toronto.
His primary interest at the time was fibromyalgia, which caused symptoms similar to those reported by the original SARS long-haulers. Intrigued, Dr. Moldofsky agreed to take a look. Their chest x-rays were clear and the nurses showed no signs of lingering viral infection. Dr. Moldofsky could see that the nurses were ill and suffering, but no lab tests or anything else could identify what was causing their symptoms.
In 2011, Dr. Moldofsky and Dr. Patcai found a strong overlap between chronic SARS, fibromyalgia, and chronic fatigue syndrome when they compared 22 patients with long-term SARS issues with 21 who had fibromyalgia. “Their problems are exactly the same. They have strange symptoms and nobody can figure out what they’re about. And these symptoms are aches and pains, and they have trouble thinking and concentrating,” Dr. Moldofsky said. Reports of COVID-19 long-haulers didn’t surprise Dr. Moldofsky, and he immediately recognized that Nath’s intention to follow these patients could provide insights into both fibromyalgia and chronic fatigue syndrome.
That’s exactly what Dr. Nath is proposing with the two NIH studies. One will focus solely on the neurologic impacts of COVID-19, including stroke, loss of taste and smell, and brain fog. The other will bring patients who have had COVID-19 symptoms for at least 6 months to the NIH Clinical Center for an inpatient stay during which they will undergo detailed physiologic tests.
Scientists around the world are launching their own post–COVID-19 studies. Dr. Moreau’s group in Montreal has laid the groundwork for such an endeavor, and the CoroNerve group in the United Kingdom is monitoring neurologic complications from the coronavirus. Many of them have the same goals as the NIH studies: Leverage the large number of COVID-19 long-haulers to better understand the earliest stages of postviral syndrome.
“At this juncture, after all the reports that we’ve seen so far, I think it’s very unlikely that there will be no relationship whatsoever between COVID-19 and chronic fatigue syndrome,” Dr. Hornig said. “I think there certainly will be some, but again, what’s the scope, what’s the size? And then, of course, even more importantly, if it is happening, what is the mechanism and how is it happening?”
For people like Ms. Gage-Witvliet, the answers can’t come soon enough. For the first time in more than a decade, the full-time professor of epidemiology didn’t prepare to teach this year because she simply can’t. It’s too taxing for her brain to deal with impromptu student questions. Ms. Gage-Witvliet hopes that, by sharing her own experiences with post COVID-19, she can help others.
“In my work, I use data to give a voice to people who don’t have a voice,” she said. “Now, I am one of those people.”
A version of this article first appeared on Medscape.com.
The next likely COVID-19 vaccine has its advantages
Among the multiple vaccine candidates around the globe, next up in the arsenal against COVID-19 is likely the single-dose Ad26.COV2.S vaccine in development from Johnson & Johnson/Janssen, infectious disease experts predict.
And it got closer with promising interim phase 1/2a trial results, published online Jan. 13 in The New England Journal of Medicine.
A single Ad26.COV2.S dose was associated with S-binding and neutralizing antibodies in more than 90% of the participants. The finding was observed in both adults aged 18-55 years and participants 65 and older, as well as for participants given low-dose or high-dose vaccinations.
The results also suggest a durable vaccine response. “The take-home message [includes] a high neutralizing antibody responder rate to a single dose of our Ad26.COV2.S COVID-19 vaccine candidate. In addition, we see that these responses and antibody titers are stable for at least 71 days,” senior study author Hanneke Schuitemaker, PhD, global head of viral vaccine discovery and translational medicine at Johnson & Johnson in Leiden, the Netherlands, said in an interview.
If the single-dose Johnson & Johnson product gains Food and Drug Administration emergency use authorization (EUA), it could significantly boost the number of overall immunizations available. Less stringent storage requirements – only regular refrigeration vs. a need to freeze the Pfizer/BioNTech and Moderna COVID-19 vaccines – is another potential advantage. The Ad26.COV2.S vaccine can be refrigerated for up to 3 months at 36°-46 °F (2°-8 °C).
“Phase 1-2 trial data on the J&J vaccine: If it works as well as the mRNA options, it will have substantial advantages,” Jeremy Faust, MD, an emergency room physician affiliated with Brigham & Women’s Hospital and Harvard Medical School, Boston, tweeted on Jan. 13.
Unlike the Pfizer/BioNTech and Moderna messenger RNA vaccines, the Johnson & Johnson product is a recombinant, replication-incompetent adenovirus serotype 26 (Ad26) vector encoding a full-length and stabilized SARS-CoV-2 spike (S) protein.
Phase 3 efficacy/safety results pending
Under normal circumstances, phase 3 trial results would not be anticipated within weeks of phase 1/2a trial findings. However, the urgency of the COVID-19 pandemic accelerated the vaccine development process, so preclinical trials were conducted simultaneously and not sequentially. For this reason, phase 3 interim results for the Johnson & Johnson vaccine are expected within weeks, and a company executive told Reuters that the rollout is on track for March.
“We hope to report data from our first phase 3 study, ENSEMBLE, in which we are testing the protective efficacy of a single dose of Ad26.COV2.S, by the end of this month or early February,” Dr. Schuitemaker said.
In the meantime, the phase 1/2a ongoing, multicenter, randomized, double-blind, and placebo-controlled trial interim results have drawn positive reactions.
“Data is highly encouraging and supports the single inoculation approach that makes this vaccine unique,” Carlos del Rio, executive associate dean for Emory University at Grady in Atlanta, wrote in a tweet on Jan. 13.
“Encouraging COVID vaccine data from J&J published [Jan. 13]. Solid antibody, CD4 T cell, and CD8 T cell responses – a nice trifecta of vaccine immune responses to see! And safe!” tweeted Shane Crotty, PhD, vaccine scientist and professor at the La Jolla (Calif.) Institute for Immunology.
First results in 800+ participants
At baseline for the phase 1/2a trial, 2% of the younger group and 1% of the 65+ group were seropositive for SARS-CoV-2 S-specific antibodies.
A total of 402 people in the younger age cohort and 403 in the 65 and older group received a first dose of the Johnson & Johnson vaccine. Many participants also received a second dose 56 days later for a separate trial, ENSEMBLE2, designed to compare safety and efficacy between single- and double-dose regimens. Results of that trial are still pending.
Safety profile
A single dose was associated with a higher incidence of solicited systemic adverse events in the higher vaccine dose group. They also found that grade 3 adverse events decreased with increasing age.
Injection site pain on the day of immunization or the next day was the most common local reaction. The pain generally resolved within 24 hours. Fever was reported by 15% of the low-dose vaccine group and 39% of the high-dose cohort. Fatigue, headache, and myalgia were the most common grade 1 or 2 solicited systemic adverse events reported.
Five serious adverse events were reported, including four that investigators deemed unrelated to vaccination: hypotension, bilateral nephrolithiasis, legionella pneumonia, and one case of worsening of multiple sclerosis. The vaccine-related serious adverse event was a fever that resulted in hospitalization because of suspicion of COVID-19. The patient recovered within 12 hours.
“These data confirm our previous experience with vaccine candidates based on our Ad26 viral vector platform in the younger age group. The almost similar performance in older adults is promising,” Dr. Schuitemaker said.
A potential limitation of the phase 1/2a trial is “the lack of representation of minority groups,” the researchers noted. Johnson & Johnson is working on improving the diversity of study participants “with respect to groups that seem to be affected most by the COVID-19 pandemic.”
AstraZeneca/Oxford vaccine status
The AstraZeneca/Oxford AZD1222 vaccine in development received approval for use in the United Kingdom on Dec. 30. The approval came after Public Health England said the country was facing “unprecedented” levels of infections, the BBC reported. AstraZeneca applied for European Medical Agency approval earlier in the week of Jan. 10, which could lead to more widespread use across Europe.
The status of the vaccine remains uncertain in the United States. A phase 3 trial that started in August was paused for about 6 weeks in September and October after an adverse event in a British volunteer halted studies worldwide. On Oct. 23, the FDA permitted researchers to continue the trial with approximately 40,000 participants.
There was some suggestion in the clinical trials that a half dose of the AstraZeneca vaccine was more effective than a full dose, 90% vs. 62%, but some irregularities in the research require further investigation.
Although the AstraZeneca vaccine is delivered to cells by an adenovirus – as with the Johnson & Johnson product – it is designed to be delivered in two doses 28 days apart, like the administration schedule of the Moderna mRNA vaccine.
A need for speed, and more doses
Regardless of which vaccine product is next to gain an EUA in the United States, many experts agree the COVID-19 vaccine rollouts so far have been problematic, at a time when cases are climbing to record-breaking levels, and likely more related to logistics over administration of the vaccine than production of the doses.
“Lots of doses being manufactured. In December 20 million, January 40 million, February 80 million and J&J hopefully soon to add to the count. The shortage is the number arms not getting vaccinated. Freezers do not get COVID. They do not need all those vaccines,” Daniel Griffin, MD, PhD, an infectious disease expert in Port Washington, N.Y., tweeted on Jan. 12.
“Unfortunately, the rollout has not gone smoothly, partly due to a lack of resources for this distribution phase we’re in,” Andrew T. Pavia, MD, chief of the division of pediatric infectious diseases at the University of Utah, Salt Lake City, said during a media briefing Jan. 14 sponsored by the Infectious Diseases Society of America (IDSA).
“We’re concerned about the mismatch between the number of people who are being told they are eligible and the amount of vaccine that is being distributed,” he said.
Complicating the rollout is a directive from U.S. Health and Human Services Secretary Alex Azar that states should start vaccinating everyone 65 and older as well as those with underlying conditions.
Expanding distribution to the 15% of Americans in just this age group is a big challenge, Dr. Pavia said. “We have enough vaccine maybe to vaccinate 40 million by the end of this month. There is a huge disconnect, and that creates a lot of problems.”
“One of the biggest problems is we are trying to do this mass vaccination program in the middle of the biggest surge we’ve ever seen,” Julie Vaishampayan, MD, MPH, chair of the IDSA Public Health Committee, said during the briefing. Without sufficient time for public health officials to plan for vaccinating a larger population, “people will come and stand in extremely long lines.”
Trying to expand immunization access without a proportionate increase in available doses prompted Dr. Vaishampayan to share an analogy from a colleague: “We are trying to fill a lake with a garden hose. Rather than making the lake bigger, what we really need is more water.”
Dr. Pavia emphasized that infectious disease experts “know the measures that work.” Not using masks, physical distancing, and hand hygiene, he said, “is a bit like knowing that really good shark repellents will be available in summer, so I’m going to jump into the ocean covered in blood while the great whites are swimming around.”
An official at the World Health Organization agreed. “Vaccines are coming online and I do believe vaccines will make a huge difference. But they are not here yet in enough quantities and in enough people to make that difference,” Michael Ryan, MB, WHO executive director of health emergencies, said during an online media briefing Jan. 13, held in conjunction with Emory University.
Dr. Ryan predicted that “we’ve got weeks if not months ahead of us in which our weapon is our knowledge ... what we know about this virus, its transmission, and stopping that transmission.
“And as the vaccines roll in, we can hopefully end this horrific pandemic.”
Dr. Schuitemaker reports grants from BARDA during the conduct of the study; personal fees and other from Janssen Vaccines and Prevention, a J&J company, outside the submitted work. Johnson & Johnson and the Biomedical Advanced Research and Development Authority of the Department of Health and Human Services funded the phase 1/2a study.
A version of this article first appeared on Medscape.com.
Among the multiple vaccine candidates around the globe, next up in the arsenal against COVID-19 is likely the single-dose Ad26.COV2.S vaccine in development from Johnson & Johnson/Janssen, infectious disease experts predict.
And it got closer with promising interim phase 1/2a trial results, published online Jan. 13 in The New England Journal of Medicine.
A single Ad26.COV2.S dose was associated with S-binding and neutralizing antibodies in more than 90% of the participants. The finding was observed in both adults aged 18-55 years and participants 65 and older, as well as for participants given low-dose or high-dose vaccinations.
The results also suggest a durable vaccine response. “The take-home message [includes] a high neutralizing antibody responder rate to a single dose of our Ad26.COV2.S COVID-19 vaccine candidate. In addition, we see that these responses and antibody titers are stable for at least 71 days,” senior study author Hanneke Schuitemaker, PhD, global head of viral vaccine discovery and translational medicine at Johnson & Johnson in Leiden, the Netherlands, said in an interview.
If the single-dose Johnson & Johnson product gains Food and Drug Administration emergency use authorization (EUA), it could significantly boost the number of overall immunizations available. Less stringent storage requirements – only regular refrigeration vs. a need to freeze the Pfizer/BioNTech and Moderna COVID-19 vaccines – is another potential advantage. The Ad26.COV2.S vaccine can be refrigerated for up to 3 months at 36°-46 °F (2°-8 °C).
“Phase 1-2 trial data on the J&J vaccine: If it works as well as the mRNA options, it will have substantial advantages,” Jeremy Faust, MD, an emergency room physician affiliated with Brigham & Women’s Hospital and Harvard Medical School, Boston, tweeted on Jan. 13.
Unlike the Pfizer/BioNTech and Moderna messenger RNA vaccines, the Johnson & Johnson product is a recombinant, replication-incompetent adenovirus serotype 26 (Ad26) vector encoding a full-length and stabilized SARS-CoV-2 spike (S) protein.
Phase 3 efficacy/safety results pending
Under normal circumstances, phase 3 trial results would not be anticipated within weeks of phase 1/2a trial findings. However, the urgency of the COVID-19 pandemic accelerated the vaccine development process, so preclinical trials were conducted simultaneously and not sequentially. For this reason, phase 3 interim results for the Johnson & Johnson vaccine are expected within weeks, and a company executive told Reuters that the rollout is on track for March.
“We hope to report data from our first phase 3 study, ENSEMBLE, in which we are testing the protective efficacy of a single dose of Ad26.COV2.S, by the end of this month or early February,” Dr. Schuitemaker said.
In the meantime, the phase 1/2a ongoing, multicenter, randomized, double-blind, and placebo-controlled trial interim results have drawn positive reactions.
“Data is highly encouraging and supports the single inoculation approach that makes this vaccine unique,” Carlos del Rio, executive associate dean for Emory University at Grady in Atlanta, wrote in a tweet on Jan. 13.
“Encouraging COVID vaccine data from J&J published [Jan. 13]. Solid antibody, CD4 T cell, and CD8 T cell responses – a nice trifecta of vaccine immune responses to see! And safe!” tweeted Shane Crotty, PhD, vaccine scientist and professor at the La Jolla (Calif.) Institute for Immunology.
First results in 800+ participants
At baseline for the phase 1/2a trial, 2% of the younger group and 1% of the 65+ group were seropositive for SARS-CoV-2 S-specific antibodies.
A total of 402 people in the younger age cohort and 403 in the 65 and older group received a first dose of the Johnson & Johnson vaccine. Many participants also received a second dose 56 days later for a separate trial, ENSEMBLE2, designed to compare safety and efficacy between single- and double-dose regimens. Results of that trial are still pending.
Safety profile
A single dose was associated with a higher incidence of solicited systemic adverse events in the higher vaccine dose group. They also found that grade 3 adverse events decreased with increasing age.
Injection site pain on the day of immunization or the next day was the most common local reaction. The pain generally resolved within 24 hours. Fever was reported by 15% of the low-dose vaccine group and 39% of the high-dose cohort. Fatigue, headache, and myalgia were the most common grade 1 or 2 solicited systemic adverse events reported.
Five serious adverse events were reported, including four that investigators deemed unrelated to vaccination: hypotension, bilateral nephrolithiasis, legionella pneumonia, and one case of worsening of multiple sclerosis. The vaccine-related serious adverse event was a fever that resulted in hospitalization because of suspicion of COVID-19. The patient recovered within 12 hours.
“These data confirm our previous experience with vaccine candidates based on our Ad26 viral vector platform in the younger age group. The almost similar performance in older adults is promising,” Dr. Schuitemaker said.
A potential limitation of the phase 1/2a trial is “the lack of representation of minority groups,” the researchers noted. Johnson & Johnson is working on improving the diversity of study participants “with respect to groups that seem to be affected most by the COVID-19 pandemic.”
AstraZeneca/Oxford vaccine status
The AstraZeneca/Oxford AZD1222 vaccine in development received approval for use in the United Kingdom on Dec. 30. The approval came after Public Health England said the country was facing “unprecedented” levels of infections, the BBC reported. AstraZeneca applied for European Medical Agency approval earlier in the week of Jan. 10, which could lead to more widespread use across Europe.
The status of the vaccine remains uncertain in the United States. A phase 3 trial that started in August was paused for about 6 weeks in September and October after an adverse event in a British volunteer halted studies worldwide. On Oct. 23, the FDA permitted researchers to continue the trial with approximately 40,000 participants.
There was some suggestion in the clinical trials that a half dose of the AstraZeneca vaccine was more effective than a full dose, 90% vs. 62%, but some irregularities in the research require further investigation.
Although the AstraZeneca vaccine is delivered to cells by an adenovirus – as with the Johnson & Johnson product – it is designed to be delivered in two doses 28 days apart, like the administration schedule of the Moderna mRNA vaccine.
A need for speed, and more doses
Regardless of which vaccine product is next to gain an EUA in the United States, many experts agree the COVID-19 vaccine rollouts so far have been problematic, at a time when cases are climbing to record-breaking levels, and likely more related to logistics over administration of the vaccine than production of the doses.
“Lots of doses being manufactured. In December 20 million, January 40 million, February 80 million and J&J hopefully soon to add to the count. The shortage is the number arms not getting vaccinated. Freezers do not get COVID. They do not need all those vaccines,” Daniel Griffin, MD, PhD, an infectious disease expert in Port Washington, N.Y., tweeted on Jan. 12.
“Unfortunately, the rollout has not gone smoothly, partly due to a lack of resources for this distribution phase we’re in,” Andrew T. Pavia, MD, chief of the division of pediatric infectious diseases at the University of Utah, Salt Lake City, said during a media briefing Jan. 14 sponsored by the Infectious Diseases Society of America (IDSA).
“We’re concerned about the mismatch between the number of people who are being told they are eligible and the amount of vaccine that is being distributed,” he said.
Complicating the rollout is a directive from U.S. Health and Human Services Secretary Alex Azar that states should start vaccinating everyone 65 and older as well as those with underlying conditions.
Expanding distribution to the 15% of Americans in just this age group is a big challenge, Dr. Pavia said. “We have enough vaccine maybe to vaccinate 40 million by the end of this month. There is a huge disconnect, and that creates a lot of problems.”
“One of the biggest problems is we are trying to do this mass vaccination program in the middle of the biggest surge we’ve ever seen,” Julie Vaishampayan, MD, MPH, chair of the IDSA Public Health Committee, said during the briefing. Without sufficient time for public health officials to plan for vaccinating a larger population, “people will come and stand in extremely long lines.”
Trying to expand immunization access without a proportionate increase in available doses prompted Dr. Vaishampayan to share an analogy from a colleague: “We are trying to fill a lake with a garden hose. Rather than making the lake bigger, what we really need is more water.”
Dr. Pavia emphasized that infectious disease experts “know the measures that work.” Not using masks, physical distancing, and hand hygiene, he said, “is a bit like knowing that really good shark repellents will be available in summer, so I’m going to jump into the ocean covered in blood while the great whites are swimming around.”
An official at the World Health Organization agreed. “Vaccines are coming online and I do believe vaccines will make a huge difference. But they are not here yet in enough quantities and in enough people to make that difference,” Michael Ryan, MB, WHO executive director of health emergencies, said during an online media briefing Jan. 13, held in conjunction with Emory University.
Dr. Ryan predicted that “we’ve got weeks if not months ahead of us in which our weapon is our knowledge ... what we know about this virus, its transmission, and stopping that transmission.
“And as the vaccines roll in, we can hopefully end this horrific pandemic.”
Dr. Schuitemaker reports grants from BARDA during the conduct of the study; personal fees and other from Janssen Vaccines and Prevention, a J&J company, outside the submitted work. Johnson & Johnson and the Biomedical Advanced Research and Development Authority of the Department of Health and Human Services funded the phase 1/2a study.
A version of this article first appeared on Medscape.com.
Among the multiple vaccine candidates around the globe, next up in the arsenal against COVID-19 is likely the single-dose Ad26.COV2.S vaccine in development from Johnson & Johnson/Janssen, infectious disease experts predict.
And it got closer with promising interim phase 1/2a trial results, published online Jan. 13 in The New England Journal of Medicine.
A single Ad26.COV2.S dose was associated with S-binding and neutralizing antibodies in more than 90% of the participants. The finding was observed in both adults aged 18-55 years and participants 65 and older, as well as for participants given low-dose or high-dose vaccinations.
The results also suggest a durable vaccine response. “The take-home message [includes] a high neutralizing antibody responder rate to a single dose of our Ad26.COV2.S COVID-19 vaccine candidate. In addition, we see that these responses and antibody titers are stable for at least 71 days,” senior study author Hanneke Schuitemaker, PhD, global head of viral vaccine discovery and translational medicine at Johnson & Johnson in Leiden, the Netherlands, said in an interview.
If the single-dose Johnson & Johnson product gains Food and Drug Administration emergency use authorization (EUA), it could significantly boost the number of overall immunizations available. Less stringent storage requirements – only regular refrigeration vs. a need to freeze the Pfizer/BioNTech and Moderna COVID-19 vaccines – is another potential advantage. The Ad26.COV2.S vaccine can be refrigerated for up to 3 months at 36°-46 °F (2°-8 °C).
“Phase 1-2 trial data on the J&J vaccine: If it works as well as the mRNA options, it will have substantial advantages,” Jeremy Faust, MD, an emergency room physician affiliated with Brigham & Women’s Hospital and Harvard Medical School, Boston, tweeted on Jan. 13.
Unlike the Pfizer/BioNTech and Moderna messenger RNA vaccines, the Johnson & Johnson product is a recombinant, replication-incompetent adenovirus serotype 26 (Ad26) vector encoding a full-length and stabilized SARS-CoV-2 spike (S) protein.
Phase 3 efficacy/safety results pending
Under normal circumstances, phase 3 trial results would not be anticipated within weeks of phase 1/2a trial findings. However, the urgency of the COVID-19 pandemic accelerated the vaccine development process, so preclinical trials were conducted simultaneously and not sequentially. For this reason, phase 3 interim results for the Johnson & Johnson vaccine are expected within weeks, and a company executive told Reuters that the rollout is on track for March.
“We hope to report data from our first phase 3 study, ENSEMBLE, in which we are testing the protective efficacy of a single dose of Ad26.COV2.S, by the end of this month or early February,” Dr. Schuitemaker said.
In the meantime, the phase 1/2a ongoing, multicenter, randomized, double-blind, and placebo-controlled trial interim results have drawn positive reactions.
“Data is highly encouraging and supports the single inoculation approach that makes this vaccine unique,” Carlos del Rio, executive associate dean for Emory University at Grady in Atlanta, wrote in a tweet on Jan. 13.
“Encouraging COVID vaccine data from J&J published [Jan. 13]. Solid antibody, CD4 T cell, and CD8 T cell responses – a nice trifecta of vaccine immune responses to see! And safe!” tweeted Shane Crotty, PhD, vaccine scientist and professor at the La Jolla (Calif.) Institute for Immunology.
First results in 800+ participants
At baseline for the phase 1/2a trial, 2% of the younger group and 1% of the 65+ group were seropositive for SARS-CoV-2 S-specific antibodies.
A total of 402 people in the younger age cohort and 403 in the 65 and older group received a first dose of the Johnson & Johnson vaccine. Many participants also received a second dose 56 days later for a separate trial, ENSEMBLE2, designed to compare safety and efficacy between single- and double-dose regimens. Results of that trial are still pending.
Safety profile
A single dose was associated with a higher incidence of solicited systemic adverse events in the higher vaccine dose group. They also found that grade 3 adverse events decreased with increasing age.
Injection site pain on the day of immunization or the next day was the most common local reaction. The pain generally resolved within 24 hours. Fever was reported by 15% of the low-dose vaccine group and 39% of the high-dose cohort. Fatigue, headache, and myalgia were the most common grade 1 or 2 solicited systemic adverse events reported.
Five serious adverse events were reported, including four that investigators deemed unrelated to vaccination: hypotension, bilateral nephrolithiasis, legionella pneumonia, and one case of worsening of multiple sclerosis. The vaccine-related serious adverse event was a fever that resulted in hospitalization because of suspicion of COVID-19. The patient recovered within 12 hours.
“These data confirm our previous experience with vaccine candidates based on our Ad26 viral vector platform in the younger age group. The almost similar performance in older adults is promising,” Dr. Schuitemaker said.
A potential limitation of the phase 1/2a trial is “the lack of representation of minority groups,” the researchers noted. Johnson & Johnson is working on improving the diversity of study participants “with respect to groups that seem to be affected most by the COVID-19 pandemic.”
AstraZeneca/Oxford vaccine status
The AstraZeneca/Oxford AZD1222 vaccine in development received approval for use in the United Kingdom on Dec. 30. The approval came after Public Health England said the country was facing “unprecedented” levels of infections, the BBC reported. AstraZeneca applied for European Medical Agency approval earlier in the week of Jan. 10, which could lead to more widespread use across Europe.
The status of the vaccine remains uncertain in the United States. A phase 3 trial that started in August was paused for about 6 weeks in September and October after an adverse event in a British volunteer halted studies worldwide. On Oct. 23, the FDA permitted researchers to continue the trial with approximately 40,000 participants.
There was some suggestion in the clinical trials that a half dose of the AstraZeneca vaccine was more effective than a full dose, 90% vs. 62%, but some irregularities in the research require further investigation.
Although the AstraZeneca vaccine is delivered to cells by an adenovirus – as with the Johnson & Johnson product – it is designed to be delivered in two doses 28 days apart, like the administration schedule of the Moderna mRNA vaccine.
A need for speed, and more doses
Regardless of which vaccine product is next to gain an EUA in the United States, many experts agree the COVID-19 vaccine rollouts so far have been problematic, at a time when cases are climbing to record-breaking levels, and likely more related to logistics over administration of the vaccine than production of the doses.
“Lots of doses being manufactured. In December 20 million, January 40 million, February 80 million and J&J hopefully soon to add to the count. The shortage is the number arms not getting vaccinated. Freezers do not get COVID. They do not need all those vaccines,” Daniel Griffin, MD, PhD, an infectious disease expert in Port Washington, N.Y., tweeted on Jan. 12.
“Unfortunately, the rollout has not gone smoothly, partly due to a lack of resources for this distribution phase we’re in,” Andrew T. Pavia, MD, chief of the division of pediatric infectious diseases at the University of Utah, Salt Lake City, said during a media briefing Jan. 14 sponsored by the Infectious Diseases Society of America (IDSA).
“We’re concerned about the mismatch between the number of people who are being told they are eligible and the amount of vaccine that is being distributed,” he said.
Complicating the rollout is a directive from U.S. Health and Human Services Secretary Alex Azar that states should start vaccinating everyone 65 and older as well as those with underlying conditions.
Expanding distribution to the 15% of Americans in just this age group is a big challenge, Dr. Pavia said. “We have enough vaccine maybe to vaccinate 40 million by the end of this month. There is a huge disconnect, and that creates a lot of problems.”
“One of the biggest problems is we are trying to do this mass vaccination program in the middle of the biggest surge we’ve ever seen,” Julie Vaishampayan, MD, MPH, chair of the IDSA Public Health Committee, said during the briefing. Without sufficient time for public health officials to plan for vaccinating a larger population, “people will come and stand in extremely long lines.”
Trying to expand immunization access without a proportionate increase in available doses prompted Dr. Vaishampayan to share an analogy from a colleague: “We are trying to fill a lake with a garden hose. Rather than making the lake bigger, what we really need is more water.”
Dr. Pavia emphasized that infectious disease experts “know the measures that work.” Not using masks, physical distancing, and hand hygiene, he said, “is a bit like knowing that really good shark repellents will be available in summer, so I’m going to jump into the ocean covered in blood while the great whites are swimming around.”
An official at the World Health Organization agreed. “Vaccines are coming online and I do believe vaccines will make a huge difference. But they are not here yet in enough quantities and in enough people to make that difference,” Michael Ryan, MB, WHO executive director of health emergencies, said during an online media briefing Jan. 13, held in conjunction with Emory University.
Dr. Ryan predicted that “we’ve got weeks if not months ahead of us in which our weapon is our knowledge ... what we know about this virus, its transmission, and stopping that transmission.
“And as the vaccines roll in, we can hopefully end this horrific pandemic.”
Dr. Schuitemaker reports grants from BARDA during the conduct of the study; personal fees and other from Janssen Vaccines and Prevention, a J&J company, outside the submitted work. Johnson & Johnson and the Biomedical Advanced Research and Development Authority of the Department of Health and Human Services funded the phase 1/2a study.
A version of this article first appeared on Medscape.com.
COVID-19 symptoms persist months after acute infection
, according to a follow-up study involving 1,733 patients.
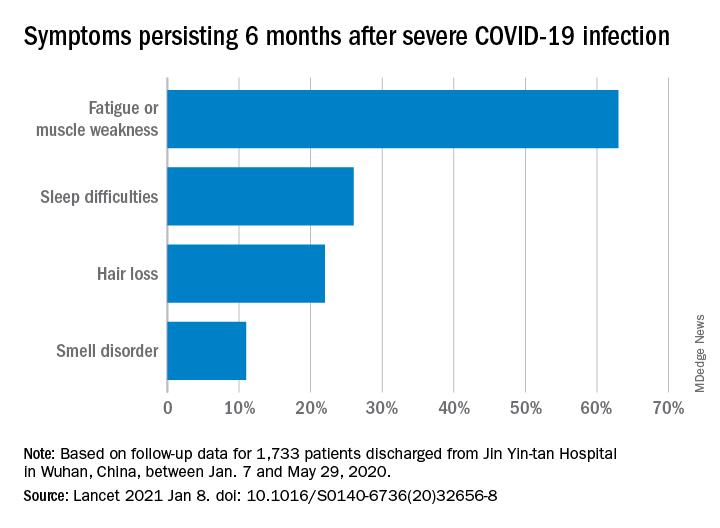
“Patients with COVID-19 had symptoms of fatigue or muscle weakness, sleep difficulties, and anxiety or depression,” and those with “more severe illness during their hospital stay had increasingly impaired pulmonary diffusion capacities and abnormal chest imaging manifestations,” Chaolin Huang, MD, of Jin Yin-tan Hospital in Wuhan, China, and associates wrote in the Lancet.
Fatigue or muscle weakness, reported by 63% of patients, was the most common symptom, followed by sleep difficulties, hair loss, and smell disorder. Altogether, 76% of those examined 6 months after discharge from Jin Yin-tan hospital – the first designated for patients with COVID-19 in Wuhan – reported at least one symptom, they said.
Symptoms were more common in women than men: 81% vs. 73% had at least one symptom, and 66% vs. 59% had fatigue or muscle weakness. Women were also more likely than men to report anxiety or depression at follow-up: 28% vs. 18% (23% overall), the investigators said.
Patients with the most severe COVID-19 were 2.4 times as likely to report any symptom later, compared with those who had the least severe levels of infection. Among the 349 participants who completed a lung function test at follow-up, lung diffusion impairment was seen in 56% of those with the most severe illness and 22% of those with the lowest level, Dr. Huang and associates reported.
In a different subset of 94 patients from whom plasma samples were collected, the “seropositivity and median titres of the neutralising antibodies were significantly lower than at the acute phase,” raising concern for reinfection, they said.
The results of the study, the investigators noted, “support that those with severe disease need post-discharge care. Longer follow-up studies in a larger population are necessary to understand the full spectrum of health consequences from COVID-19.”
, according to a follow-up study involving 1,733 patients.

“Patients with COVID-19 had symptoms of fatigue or muscle weakness, sleep difficulties, and anxiety or depression,” and those with “more severe illness during their hospital stay had increasingly impaired pulmonary diffusion capacities and abnormal chest imaging manifestations,” Chaolin Huang, MD, of Jin Yin-tan Hospital in Wuhan, China, and associates wrote in the Lancet.
Fatigue or muscle weakness, reported by 63% of patients, was the most common symptom, followed by sleep difficulties, hair loss, and smell disorder. Altogether, 76% of those examined 6 months after discharge from Jin Yin-tan hospital – the first designated for patients with COVID-19 in Wuhan – reported at least one symptom, they said.
Symptoms were more common in women than men: 81% vs. 73% had at least one symptom, and 66% vs. 59% had fatigue or muscle weakness. Women were also more likely than men to report anxiety or depression at follow-up: 28% vs. 18% (23% overall), the investigators said.
Patients with the most severe COVID-19 were 2.4 times as likely to report any symptom later, compared with those who had the least severe levels of infection. Among the 349 participants who completed a lung function test at follow-up, lung diffusion impairment was seen in 56% of those with the most severe illness and 22% of those with the lowest level, Dr. Huang and associates reported.
In a different subset of 94 patients from whom plasma samples were collected, the “seropositivity and median titres of the neutralising antibodies were significantly lower than at the acute phase,” raising concern for reinfection, they said.
The results of the study, the investigators noted, “support that those with severe disease need post-discharge care. Longer follow-up studies in a larger population are necessary to understand the full spectrum of health consequences from COVID-19.”
, according to a follow-up study involving 1,733 patients.

“Patients with COVID-19 had symptoms of fatigue or muscle weakness, sleep difficulties, and anxiety or depression,” and those with “more severe illness during their hospital stay had increasingly impaired pulmonary diffusion capacities and abnormal chest imaging manifestations,” Chaolin Huang, MD, of Jin Yin-tan Hospital in Wuhan, China, and associates wrote in the Lancet.
Fatigue or muscle weakness, reported by 63% of patients, was the most common symptom, followed by sleep difficulties, hair loss, and smell disorder. Altogether, 76% of those examined 6 months after discharge from Jin Yin-tan hospital – the first designated for patients with COVID-19 in Wuhan – reported at least one symptom, they said.
Symptoms were more common in women than men: 81% vs. 73% had at least one symptom, and 66% vs. 59% had fatigue or muscle weakness. Women were also more likely than men to report anxiety or depression at follow-up: 28% vs. 18% (23% overall), the investigators said.
Patients with the most severe COVID-19 were 2.4 times as likely to report any symptom later, compared with those who had the least severe levels of infection. Among the 349 participants who completed a lung function test at follow-up, lung diffusion impairment was seen in 56% of those with the most severe illness and 22% of those with the lowest level, Dr. Huang and associates reported.
In a different subset of 94 patients from whom plasma samples were collected, the “seropositivity and median titres of the neutralising antibodies were significantly lower than at the acute phase,” raising concern for reinfection, they said.
The results of the study, the investigators noted, “support that those with severe disease need post-discharge care. Longer follow-up studies in a larger population are necessary to understand the full spectrum of health consequences from COVID-19.”
FROM THE LANCET
CVD deaths rose, imaging declined during pandemic
While the direct toll of the COVID-19 pandemic is being tallied and shared on the nightly news, the indirect effects will undoubtedly take years to fully measure.
In two papers published online Jan. 11 in the Journal of the American College of Cardiology, researchers have started the process of quantifying the impact of the pandemic on the care of patients with cardiovascular disease (CVD).
In the first study, Rishi Wadhera, MD, MPP, MPhil, and colleagues from the Beth Israel Deaconess Medical Center and Harvard Medical School in Boston examined population-level data to determine how deaths from cardiovascular causes changed in the United States in the early months of the pandemic relative to the same periods in 2019.
In a second paper, Andrew J. Einstein, MD, PhD, from Columbia University Irving Medical Center/New York–Presbyterian Hospital and colleagues looked at the pandemic’s international impact on the diagnosis of heart disease.
Using data from the National Center for Health Statistics, Dr. Wadhera and colleagues compared death rates from cardiovascular causes in the United States from March 18, 2020, to June 2, 2020, (the first wave of the pandemic) and from Jan. 1, 2020, to March 17, 2020, (the period just before the pandemic started) and compared them to the same periods in 2019. ICD codes were used to identify underlying causes of death.
Relative to 2019, they found a significant increase in deaths from ischemic heart disease nationally (1.11; 95% confidence interval, 1.04-1.18), as well as an increase in deaths caused by hypertensive disease (1.17; 95% CI, 1.09-1.26). There was no apparent increase in deaths from heart failure, cerebrovascular disease, or other diseases of the circulatory system.
When they looked just at New York City, the area hit hardest during the early part of the pandemic, the relative increases in deaths from ischemic heart disease were more pronounced.
Deaths from ischemic heart disease or hypertensive diseases jumped 139% and 164%, respectively, between March 18, 2020, and June 2, 2020.
More modest increases in deaths were seen in the remainder of New York state, New Jersey, Michigan and Illinois, while Massachusetts and Louisiana did not see a change in cardiovascular deaths.
Several studies from different parts of the world have indicated a 40%-50% drop in hospitalization for myocardial infarction in the initial months of the pandemic, said Dr. Wadhera in an interview.
“We wanted to understand where did all the heart attacks go? And we worried that patients with urgent heart conditions were not seeking the medical care they needed. I think our data suggest that this may have been the case,” reported Dr. Wadhera.
“This very much reflects the reality of what we’re seeing on the ground,” he told this news organization. “After the initial surge ended, when hospital volumes began to return to normal, we saw patients come into the hospital who clearly had a heart attack during the surge months – and were now experiencing complications of that event – because they had initially not come into the hospital due to concerns about exposure to the virus.”
A limitation of their data, he stressed, is whether some deaths coded as CVD deaths were really deaths from undiagnosed COVID-19. “It’s possible that some portion of the increased deaths we observed really reflect the cardiovascular complications of undiagnosed COVID-19, because we know that testing was quite limited during the early first surge of cases.”
“I think that basically three factors – patients avoiding the health care system because of fear of getting COVID, health care systems being strained and overwhelmed leading to the deferral of cardiovascular care and semi-elective procedures, and the cardiovascular complications of COVID-19 itself – all probably collectively contributed to the rise in cardiovascular deaths that we observed,” said Dr. Wadhera.
In an accompanying editorial, Michael N. Young, MD, Geisel School of Medicine at Dartmouth, Lebanon, N.H., and colleagues write that these data, taken together with an earlier study showing an increase in out-of-hospital cardiac arrests at the pandemic peak in New York City, “support the notion of excess fatalities due to unattended comorbid illnesses.” That said, attribution of death in the COVID era “remains problematic.”
In the second article, Andrew Einstein, MD, PhD, and the INCAPS COVID Investigators Group took a broader approach and looked at the impact of COVID-19 on cardiac diagnostic procedures in over 100 countries.
The INCAPS (International Atomic Energy Agency Noninvasive Cardiology Protocols Study) group has for the past decade conducted numerous studies addressing the use of best practices and worldwide practice variation in CVD diagnosis.
For this effort, they sent a survey link to INCAPS participants worldwide, ultimately including 909 survey responses from 108 countries in the final analysis.
Compared with March 2019, overall procedure volume decreased 42% in March 2020 and 64% in April 2020.
The greatest decreases were seen in stress testing (78%) and transesophageal echocardiography (76%), both procedures, noted Dr. Einstein, associated with a greater risk of aerosolization.
“Whether as we reset after COVID we return to the same place in terms of the use of cardiovascular diagnostic testing remains to be seen, but it certainly poses an opportunity to improve our utilization of various modes of testing,” said Dr. Einstein.
Using regression analysis, Dr. Einstein and colleagues were able to see that sites located in low-income and lower-middle-income countries saw an additional 22% reduction in cardiac procedures and less availability of personal protective equipment (PPE) and telehealth.
Fifty-two percent of survey respondents reported significant shortages of N95 masks early in the pandemic, with fewer issues in supplies of gloves, gowns, and face shields. Lower-income countries were more likely to face significant PPE shortages and less likely to be able to implement telehealth strategies to make up for reduced in-person care. PPE shortage itself, however, was not related to lower procedural volume on multivariable regression.
“It all really begs the question of whether there is more that the world can do to help out the developing world in terms of managing the pandemic in all its facets,” said Dr. Einstein in an interview, adding he was “shocked” to learn how difficult it was for some lower-income countries to get sufficient PPE.
Did shutdowns go too far?
Calling this a “remarkable study,” an editorial written by Darryl P. Leong, MBBS, PhD, John W. Eikelboom, MBBS, and Salim Yusuf, MBBS, DPhil, all from McMaster University, Hamilton, Ont., suggests that perhaps health systems in some places went too far in closing down during the first wave of the pandemic, naming specifically Canada, Eastern Europe, and Saudi Arabia as examples.
“Although these measures were taken to prepare for the worst, overwhelming numbers of patients with COVID-19 did not materialize during the first wave of the pandemic in these countries. It is possible that delaying so-called nonessential services may have been unnecessary and potentially harmful, because it likely led to delays in providing care for the treatment of serious non–COVID-19 illnesses.”
Since then, more experience and more data have largely allowed hospital systems to “tackle the ebb and flow” of COVID-19 cases in ways that limit shutdowns of important health services, they said.
Given the more pronounced effect in low- and middle-income countries, they stressed the need to focus resources on ways to promote prevention and treatment that do not rely on diagnostic procedures.
“This calls for more emphasis on developing efficient systems of telehealth, especially in poorer countries or in remote settings in all countries,” Dr. Leong and colleagues conclude.
Dr. Wadhera has reported research support from the National Heart, Lung, and Blood Institute, along with fellow senior author Robert W. Yeh, MD, MBA, who has also received personal fees and grants from several companies not related to the submitted work. Dr. Einstein, Dr. Leong, Dr. Eikelboom, and Dr. Yusuf have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
While the direct toll of the COVID-19 pandemic is being tallied and shared on the nightly news, the indirect effects will undoubtedly take years to fully measure.
In two papers published online Jan. 11 in the Journal of the American College of Cardiology, researchers have started the process of quantifying the impact of the pandemic on the care of patients with cardiovascular disease (CVD).
In the first study, Rishi Wadhera, MD, MPP, MPhil, and colleagues from the Beth Israel Deaconess Medical Center and Harvard Medical School in Boston examined population-level data to determine how deaths from cardiovascular causes changed in the United States in the early months of the pandemic relative to the same periods in 2019.
In a second paper, Andrew J. Einstein, MD, PhD, from Columbia University Irving Medical Center/New York–Presbyterian Hospital and colleagues looked at the pandemic’s international impact on the diagnosis of heart disease.
Using data from the National Center for Health Statistics, Dr. Wadhera and colleagues compared death rates from cardiovascular causes in the United States from March 18, 2020, to June 2, 2020, (the first wave of the pandemic) and from Jan. 1, 2020, to March 17, 2020, (the period just before the pandemic started) and compared them to the same periods in 2019. ICD codes were used to identify underlying causes of death.
Relative to 2019, they found a significant increase in deaths from ischemic heart disease nationally (1.11; 95% confidence interval, 1.04-1.18), as well as an increase in deaths caused by hypertensive disease (1.17; 95% CI, 1.09-1.26). There was no apparent increase in deaths from heart failure, cerebrovascular disease, or other diseases of the circulatory system.
When they looked just at New York City, the area hit hardest during the early part of the pandemic, the relative increases in deaths from ischemic heart disease were more pronounced.
Deaths from ischemic heart disease or hypertensive diseases jumped 139% and 164%, respectively, between March 18, 2020, and June 2, 2020.
More modest increases in deaths were seen in the remainder of New York state, New Jersey, Michigan and Illinois, while Massachusetts and Louisiana did not see a change in cardiovascular deaths.
Several studies from different parts of the world have indicated a 40%-50% drop in hospitalization for myocardial infarction in the initial months of the pandemic, said Dr. Wadhera in an interview.
“We wanted to understand where did all the heart attacks go? And we worried that patients with urgent heart conditions were not seeking the medical care they needed. I think our data suggest that this may have been the case,” reported Dr. Wadhera.
“This very much reflects the reality of what we’re seeing on the ground,” he told this news organization. “After the initial surge ended, when hospital volumes began to return to normal, we saw patients come into the hospital who clearly had a heart attack during the surge months – and were now experiencing complications of that event – because they had initially not come into the hospital due to concerns about exposure to the virus.”
A limitation of their data, he stressed, is whether some deaths coded as CVD deaths were really deaths from undiagnosed COVID-19. “It’s possible that some portion of the increased deaths we observed really reflect the cardiovascular complications of undiagnosed COVID-19, because we know that testing was quite limited during the early first surge of cases.”
“I think that basically three factors – patients avoiding the health care system because of fear of getting COVID, health care systems being strained and overwhelmed leading to the deferral of cardiovascular care and semi-elective procedures, and the cardiovascular complications of COVID-19 itself – all probably collectively contributed to the rise in cardiovascular deaths that we observed,” said Dr. Wadhera.
In an accompanying editorial, Michael N. Young, MD, Geisel School of Medicine at Dartmouth, Lebanon, N.H., and colleagues write that these data, taken together with an earlier study showing an increase in out-of-hospital cardiac arrests at the pandemic peak in New York City, “support the notion of excess fatalities due to unattended comorbid illnesses.” That said, attribution of death in the COVID era “remains problematic.”
In the second article, Andrew Einstein, MD, PhD, and the INCAPS COVID Investigators Group took a broader approach and looked at the impact of COVID-19 on cardiac diagnostic procedures in over 100 countries.
The INCAPS (International Atomic Energy Agency Noninvasive Cardiology Protocols Study) group has for the past decade conducted numerous studies addressing the use of best practices and worldwide practice variation in CVD diagnosis.
For this effort, they sent a survey link to INCAPS participants worldwide, ultimately including 909 survey responses from 108 countries in the final analysis.
Compared with March 2019, overall procedure volume decreased 42% in March 2020 and 64% in April 2020.
The greatest decreases were seen in stress testing (78%) and transesophageal echocardiography (76%), both procedures, noted Dr. Einstein, associated with a greater risk of aerosolization.
“Whether as we reset after COVID we return to the same place in terms of the use of cardiovascular diagnostic testing remains to be seen, but it certainly poses an opportunity to improve our utilization of various modes of testing,” said Dr. Einstein.
Using regression analysis, Dr. Einstein and colleagues were able to see that sites located in low-income and lower-middle-income countries saw an additional 22% reduction in cardiac procedures and less availability of personal protective equipment (PPE) and telehealth.
Fifty-two percent of survey respondents reported significant shortages of N95 masks early in the pandemic, with fewer issues in supplies of gloves, gowns, and face shields. Lower-income countries were more likely to face significant PPE shortages and less likely to be able to implement telehealth strategies to make up for reduced in-person care. PPE shortage itself, however, was not related to lower procedural volume on multivariable regression.
“It all really begs the question of whether there is more that the world can do to help out the developing world in terms of managing the pandemic in all its facets,” said Dr. Einstein in an interview, adding he was “shocked” to learn how difficult it was for some lower-income countries to get sufficient PPE.
Did shutdowns go too far?
Calling this a “remarkable study,” an editorial written by Darryl P. Leong, MBBS, PhD, John W. Eikelboom, MBBS, and Salim Yusuf, MBBS, DPhil, all from McMaster University, Hamilton, Ont., suggests that perhaps health systems in some places went too far in closing down during the first wave of the pandemic, naming specifically Canada, Eastern Europe, and Saudi Arabia as examples.
“Although these measures were taken to prepare for the worst, overwhelming numbers of patients with COVID-19 did not materialize during the first wave of the pandemic in these countries. It is possible that delaying so-called nonessential services may have been unnecessary and potentially harmful, because it likely led to delays in providing care for the treatment of serious non–COVID-19 illnesses.”
Since then, more experience and more data have largely allowed hospital systems to “tackle the ebb and flow” of COVID-19 cases in ways that limit shutdowns of important health services, they said.
Given the more pronounced effect in low- and middle-income countries, they stressed the need to focus resources on ways to promote prevention and treatment that do not rely on diagnostic procedures.
“This calls for more emphasis on developing efficient systems of telehealth, especially in poorer countries or in remote settings in all countries,” Dr. Leong and colleagues conclude.
Dr. Wadhera has reported research support from the National Heart, Lung, and Blood Institute, along with fellow senior author Robert W. Yeh, MD, MBA, who has also received personal fees and grants from several companies not related to the submitted work. Dr. Einstein, Dr. Leong, Dr. Eikelboom, and Dr. Yusuf have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
While the direct toll of the COVID-19 pandemic is being tallied and shared on the nightly news, the indirect effects will undoubtedly take years to fully measure.
In two papers published online Jan. 11 in the Journal of the American College of Cardiology, researchers have started the process of quantifying the impact of the pandemic on the care of patients with cardiovascular disease (CVD).
In the first study, Rishi Wadhera, MD, MPP, MPhil, and colleagues from the Beth Israel Deaconess Medical Center and Harvard Medical School in Boston examined population-level data to determine how deaths from cardiovascular causes changed in the United States in the early months of the pandemic relative to the same periods in 2019.
In a second paper, Andrew J. Einstein, MD, PhD, from Columbia University Irving Medical Center/New York–Presbyterian Hospital and colleagues looked at the pandemic’s international impact on the diagnosis of heart disease.
Using data from the National Center for Health Statistics, Dr. Wadhera and colleagues compared death rates from cardiovascular causes in the United States from March 18, 2020, to June 2, 2020, (the first wave of the pandemic) and from Jan. 1, 2020, to March 17, 2020, (the period just before the pandemic started) and compared them to the same periods in 2019. ICD codes were used to identify underlying causes of death.
Relative to 2019, they found a significant increase in deaths from ischemic heart disease nationally (1.11; 95% confidence interval, 1.04-1.18), as well as an increase in deaths caused by hypertensive disease (1.17; 95% CI, 1.09-1.26). There was no apparent increase in deaths from heart failure, cerebrovascular disease, or other diseases of the circulatory system.
When they looked just at New York City, the area hit hardest during the early part of the pandemic, the relative increases in deaths from ischemic heart disease were more pronounced.
Deaths from ischemic heart disease or hypertensive diseases jumped 139% and 164%, respectively, between March 18, 2020, and June 2, 2020.
More modest increases in deaths were seen in the remainder of New York state, New Jersey, Michigan and Illinois, while Massachusetts and Louisiana did not see a change in cardiovascular deaths.
Several studies from different parts of the world have indicated a 40%-50% drop in hospitalization for myocardial infarction in the initial months of the pandemic, said Dr. Wadhera in an interview.
“We wanted to understand where did all the heart attacks go? And we worried that patients with urgent heart conditions were not seeking the medical care they needed. I think our data suggest that this may have been the case,” reported Dr. Wadhera.
“This very much reflects the reality of what we’re seeing on the ground,” he told this news organization. “After the initial surge ended, when hospital volumes began to return to normal, we saw patients come into the hospital who clearly had a heart attack during the surge months – and were now experiencing complications of that event – because they had initially not come into the hospital due to concerns about exposure to the virus.”
A limitation of their data, he stressed, is whether some deaths coded as CVD deaths were really deaths from undiagnosed COVID-19. “It’s possible that some portion of the increased deaths we observed really reflect the cardiovascular complications of undiagnosed COVID-19, because we know that testing was quite limited during the early first surge of cases.”
“I think that basically three factors – patients avoiding the health care system because of fear of getting COVID, health care systems being strained and overwhelmed leading to the deferral of cardiovascular care and semi-elective procedures, and the cardiovascular complications of COVID-19 itself – all probably collectively contributed to the rise in cardiovascular deaths that we observed,” said Dr. Wadhera.
In an accompanying editorial, Michael N. Young, MD, Geisel School of Medicine at Dartmouth, Lebanon, N.H., and colleagues write that these data, taken together with an earlier study showing an increase in out-of-hospital cardiac arrests at the pandemic peak in New York City, “support the notion of excess fatalities due to unattended comorbid illnesses.” That said, attribution of death in the COVID era “remains problematic.”
In the second article, Andrew Einstein, MD, PhD, and the INCAPS COVID Investigators Group took a broader approach and looked at the impact of COVID-19 on cardiac diagnostic procedures in over 100 countries.
The INCAPS (International Atomic Energy Agency Noninvasive Cardiology Protocols Study) group has for the past decade conducted numerous studies addressing the use of best practices and worldwide practice variation in CVD diagnosis.
For this effort, they sent a survey link to INCAPS participants worldwide, ultimately including 909 survey responses from 108 countries in the final analysis.
Compared with March 2019, overall procedure volume decreased 42% in March 2020 and 64% in April 2020.
The greatest decreases were seen in stress testing (78%) and transesophageal echocardiography (76%), both procedures, noted Dr. Einstein, associated with a greater risk of aerosolization.
“Whether as we reset after COVID we return to the same place in terms of the use of cardiovascular diagnostic testing remains to be seen, but it certainly poses an opportunity to improve our utilization of various modes of testing,” said Dr. Einstein.
Using regression analysis, Dr. Einstein and colleagues were able to see that sites located in low-income and lower-middle-income countries saw an additional 22% reduction in cardiac procedures and less availability of personal protective equipment (PPE) and telehealth.
Fifty-two percent of survey respondents reported significant shortages of N95 masks early in the pandemic, with fewer issues in supplies of gloves, gowns, and face shields. Lower-income countries were more likely to face significant PPE shortages and less likely to be able to implement telehealth strategies to make up for reduced in-person care. PPE shortage itself, however, was not related to lower procedural volume on multivariable regression.
“It all really begs the question of whether there is more that the world can do to help out the developing world in terms of managing the pandemic in all its facets,” said Dr. Einstein in an interview, adding he was “shocked” to learn how difficult it was for some lower-income countries to get sufficient PPE.
Did shutdowns go too far?
Calling this a “remarkable study,” an editorial written by Darryl P. Leong, MBBS, PhD, John W. Eikelboom, MBBS, and Salim Yusuf, MBBS, DPhil, all from McMaster University, Hamilton, Ont., suggests that perhaps health systems in some places went too far in closing down during the first wave of the pandemic, naming specifically Canada, Eastern Europe, and Saudi Arabia as examples.
“Although these measures were taken to prepare for the worst, overwhelming numbers of patients with COVID-19 did not materialize during the first wave of the pandemic in these countries. It is possible that delaying so-called nonessential services may have been unnecessary and potentially harmful, because it likely led to delays in providing care for the treatment of serious non–COVID-19 illnesses.”
Since then, more experience and more data have largely allowed hospital systems to “tackle the ebb and flow” of COVID-19 cases in ways that limit shutdowns of important health services, they said.
Given the more pronounced effect in low- and middle-income countries, they stressed the need to focus resources on ways to promote prevention and treatment that do not rely on diagnostic procedures.
“This calls for more emphasis on developing efficient systems of telehealth, especially in poorer countries or in remote settings in all countries,” Dr. Leong and colleagues conclude.
Dr. Wadhera has reported research support from the National Heart, Lung, and Blood Institute, along with fellow senior author Robert W. Yeh, MD, MBA, who has also received personal fees and grants from several companies not related to the submitted work. Dr. Einstein, Dr. Leong, Dr. Eikelboom, and Dr. Yusuf have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Natural immunity from COVID-19 ‘may last months’
Infection with the SARS-CoV-2 virus may provide some immunity for at least 5 months, interim results from a study has found.
The first report from the Sarscov2 Immunity & Reinfection Evaluation (SIREN) study suggested that antibodies from people who had recovered from COVID-19 gave at least 83% protection against reinfection compared with people who had not had the disease before.
However, Public Health England (PHE) researchers said some people with antibodies may still be able to carry and transmit the SARS-CoV-2 virus.
‘Strongly encouraged’
Susan Hopkins, PhD, senior medical advisor at PHE, who is leading the study, said the overall findings were good news. She told a briefing hosted by the Science Media Centre: “I am strongly encouraged that people have immunity that is lasting much more than the few months that was speculated before the summer.”
She added: “It allows people to feel that their prior infection will protect them from future infections but at the same time it is not complete protection, and therefore they still need to be careful when they are out and about.”
PHE scientists said they would continue to assess whether protection might last longer than 5 months.
Eleanor Riley, PhD, professor of immunology and infectious disease at the University of Edinburgh, said the report suggested that “natural infection provides short-term protection against COVID-19 that is very similar to that conferred by vaccination.”
Simon Clarke, PhD, associate professor in cellular microbiology at the University of Reading, said: “The concerning finding is that some people who have COVID antibodies appear to still be able to carry the coronavirus and could spread it to others. This means that the vast majority of the population will either need to have natural immunity or have been immunised for us to fully lift restrictions on our lives.”
The analysis took place before the new variant of SARS-CoV-2 became widespread in the UK. The PHE scientists said that further work was underway to establish whether and to what extent antibodies also provide protection from the VOC202012/01 variant.
Healthcare Workers
The SIREN preprint analysed data from 20,787 health care workers from 102 NHS trusts who had undergone antibody and PCR testing from June 18 to November 9, 2020.
Of those, 6614 tested positive for COVID-19 antibodies.
Of the 44 potential reinfections identified, two were designated ‘probable’ and 42 ‘possible’, based on available evidence.
Both of the two individuals classified as probable reinfections reported having experienced COVID-19 symptoms during the first wave of the pandemic but were not tested at the time. Both reported that their symptoms were less severe the second time.
None of the 44 potential reinfection cases were PCR tested during the first wave, but all tested positive for COVID-19 antibodies at the time they were recruited to the study.
Tom Wingfield, PhD, senior clinical lecturer at the Liverpool School of Tropical Medicine, said that given the high risk of SARS-CoV-2 infection for frontline NHS staff, it was “vital that we do all that we can to understand, predict, and prevent risk of SARS-CoV-2 amongst healthcare workers”.
The study will continue to follow participants for 12 months to explore how long any immunity may last, the effectiveness of vaccines, and to what extent people with immunity are able to carry and transmit the virus.
A version of this article first appeared on Medscape.com.
Infection with the SARS-CoV-2 virus may provide some immunity for at least 5 months, interim results from a study has found.
The first report from the Sarscov2 Immunity & Reinfection Evaluation (SIREN) study suggested that antibodies from people who had recovered from COVID-19 gave at least 83% protection against reinfection compared with people who had not had the disease before.
However, Public Health England (PHE) researchers said some people with antibodies may still be able to carry and transmit the SARS-CoV-2 virus.
‘Strongly encouraged’
Susan Hopkins, PhD, senior medical advisor at PHE, who is leading the study, said the overall findings were good news. She told a briefing hosted by the Science Media Centre: “I am strongly encouraged that people have immunity that is lasting much more than the few months that was speculated before the summer.”
She added: “It allows people to feel that their prior infection will protect them from future infections but at the same time it is not complete protection, and therefore they still need to be careful when they are out and about.”
PHE scientists said they would continue to assess whether protection might last longer than 5 months.
Eleanor Riley, PhD, professor of immunology and infectious disease at the University of Edinburgh, said the report suggested that “natural infection provides short-term protection against COVID-19 that is very similar to that conferred by vaccination.”
Simon Clarke, PhD, associate professor in cellular microbiology at the University of Reading, said: “The concerning finding is that some people who have COVID antibodies appear to still be able to carry the coronavirus and could spread it to others. This means that the vast majority of the population will either need to have natural immunity or have been immunised for us to fully lift restrictions on our lives.”
The analysis took place before the new variant of SARS-CoV-2 became widespread in the UK. The PHE scientists said that further work was underway to establish whether and to what extent antibodies also provide protection from the VOC202012/01 variant.
Healthcare Workers
The SIREN preprint analysed data from 20,787 health care workers from 102 NHS trusts who had undergone antibody and PCR testing from June 18 to November 9, 2020.
Of those, 6614 tested positive for COVID-19 antibodies.
Of the 44 potential reinfections identified, two were designated ‘probable’ and 42 ‘possible’, based on available evidence.
Both of the two individuals classified as probable reinfections reported having experienced COVID-19 symptoms during the first wave of the pandemic but were not tested at the time. Both reported that their symptoms were less severe the second time.
None of the 44 potential reinfection cases were PCR tested during the first wave, but all tested positive for COVID-19 antibodies at the time they were recruited to the study.
Tom Wingfield, PhD, senior clinical lecturer at the Liverpool School of Tropical Medicine, said that given the high risk of SARS-CoV-2 infection for frontline NHS staff, it was “vital that we do all that we can to understand, predict, and prevent risk of SARS-CoV-2 amongst healthcare workers”.
The study will continue to follow participants for 12 months to explore how long any immunity may last, the effectiveness of vaccines, and to what extent people with immunity are able to carry and transmit the virus.
A version of this article first appeared on Medscape.com.
Infection with the SARS-CoV-2 virus may provide some immunity for at least 5 months, interim results from a study has found.
The first report from the Sarscov2 Immunity & Reinfection Evaluation (SIREN) study suggested that antibodies from people who had recovered from COVID-19 gave at least 83% protection against reinfection compared with people who had not had the disease before.
However, Public Health England (PHE) researchers said some people with antibodies may still be able to carry and transmit the SARS-CoV-2 virus.
‘Strongly encouraged’
Susan Hopkins, PhD, senior medical advisor at PHE, who is leading the study, said the overall findings were good news. She told a briefing hosted by the Science Media Centre: “I am strongly encouraged that people have immunity that is lasting much more than the few months that was speculated before the summer.”
She added: “It allows people to feel that their prior infection will protect them from future infections but at the same time it is not complete protection, and therefore they still need to be careful when they are out and about.”
PHE scientists said they would continue to assess whether protection might last longer than 5 months.
Eleanor Riley, PhD, professor of immunology and infectious disease at the University of Edinburgh, said the report suggested that “natural infection provides short-term protection against COVID-19 that is very similar to that conferred by vaccination.”
Simon Clarke, PhD, associate professor in cellular microbiology at the University of Reading, said: “The concerning finding is that some people who have COVID antibodies appear to still be able to carry the coronavirus and could spread it to others. This means that the vast majority of the population will either need to have natural immunity or have been immunised for us to fully lift restrictions on our lives.”
The analysis took place before the new variant of SARS-CoV-2 became widespread in the UK. The PHE scientists said that further work was underway to establish whether and to what extent antibodies also provide protection from the VOC202012/01 variant.
Healthcare Workers
The SIREN preprint analysed data from 20,787 health care workers from 102 NHS trusts who had undergone antibody and PCR testing from June 18 to November 9, 2020.
Of those, 6614 tested positive for COVID-19 antibodies.
Of the 44 potential reinfections identified, two were designated ‘probable’ and 42 ‘possible’, based on available evidence.
Both of the two individuals classified as probable reinfections reported having experienced COVID-19 symptoms during the first wave of the pandemic but were not tested at the time. Both reported that their symptoms were less severe the second time.
None of the 44 potential reinfection cases were PCR tested during the first wave, but all tested positive for COVID-19 antibodies at the time they were recruited to the study.
Tom Wingfield, PhD, senior clinical lecturer at the Liverpool School of Tropical Medicine, said that given the high risk of SARS-CoV-2 infection for frontline NHS staff, it was “vital that we do all that we can to understand, predict, and prevent risk of SARS-CoV-2 amongst healthcare workers”.
The study will continue to follow participants for 12 months to explore how long any immunity may last, the effectiveness of vaccines, and to what extent people with immunity are able to carry and transmit the virus.
A version of this article first appeared on Medscape.com.
COVID protections suppressed flu season in U.S.
Last fall, health experts said it was possible the United States could experience an easy 2020-21 flu season because health measures to fight COVID-19 would also thwart the spread of influenza.
It looks like that happened – and then some. Numbers are strikingly low for cases of the flu and other common respiratory and gastrointestinal viruses, health experts told the Washington Post.
“It’s crazy,” Lynnette Brammer, MPH, who leads the domestic influenza surveillance team at the Centers for Disease Control and Prevention, told the Washington Post. “This is my 30th flu season. I never would have expected to see flu activity this low.”
Influenza A, influenza B, parainfluenza, norovirus, respiratory syncytial virus, human metapneumovirus, and the bacteria that cause whooping cough and pneumonia are circulating at near-record-low levels.
As an example, the Washington Post said in the third week of December 2019, the CDC’s network of clinical labs reported 16.2% of almost 30,000 samples tested positive for influenza A. During the same period in 2020, only 0.3% tested positive.
But there’s a possible downside to this suppression of viruses, because flu and other viruses may rebound once the coronavirus is brought under control.
“The best analogy is to a forest fire,” Bryan Grenfell, PhD, an epidemiologist and population biologist at Princeton (N.J.) University, told the Washington Post. “For the fire to spread, it needs to have unburned wood. For epidemics to spread, they require people who haven’t previously been infected. So if people don’t get infected this year by these viruses, they likely will at some point later on.”
American health experts like Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Disease, said last fall that they noticed Australia and other nations in the southern hemisphere had easy flu seasons, apparently because of COVID protection measures. The flu season there runs March through August.
COVID-19 now has a very low presence in Australia, but in recent months the flu has been making a comeback. Flu cases among children aged 5 and younger rose sixfold by December, when such cases are usually at their lowest, the Washington Post said.
“That’s an important cautionary tale for us,” said Kevin Messacar, MD, an infectious disease doctor at Children’s Hospital Colorado, Aurora. “Just because we get through the winter and don’t see much RSV or influenza doesn’t mean we’ll be out of the woods.”
A version of this article first appeared on WebMD.com.
Last fall, health experts said it was possible the United States could experience an easy 2020-21 flu season because health measures to fight COVID-19 would also thwart the spread of influenza.
It looks like that happened – and then some. Numbers are strikingly low for cases of the flu and other common respiratory and gastrointestinal viruses, health experts told the Washington Post.
“It’s crazy,” Lynnette Brammer, MPH, who leads the domestic influenza surveillance team at the Centers for Disease Control and Prevention, told the Washington Post. “This is my 30th flu season. I never would have expected to see flu activity this low.”
Influenza A, influenza B, parainfluenza, norovirus, respiratory syncytial virus, human metapneumovirus, and the bacteria that cause whooping cough and pneumonia are circulating at near-record-low levels.
As an example, the Washington Post said in the third week of December 2019, the CDC’s network of clinical labs reported 16.2% of almost 30,000 samples tested positive for influenza A. During the same period in 2020, only 0.3% tested positive.
But there’s a possible downside to this suppression of viruses, because flu and other viruses may rebound once the coronavirus is brought under control.
“The best analogy is to a forest fire,” Bryan Grenfell, PhD, an epidemiologist and population biologist at Princeton (N.J.) University, told the Washington Post. “For the fire to spread, it needs to have unburned wood. For epidemics to spread, they require people who haven’t previously been infected. So if people don’t get infected this year by these viruses, they likely will at some point later on.”
American health experts like Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Disease, said last fall that they noticed Australia and other nations in the southern hemisphere had easy flu seasons, apparently because of COVID protection measures. The flu season there runs March through August.
COVID-19 now has a very low presence in Australia, but in recent months the flu has been making a comeback. Flu cases among children aged 5 and younger rose sixfold by December, when such cases are usually at their lowest, the Washington Post said.
“That’s an important cautionary tale for us,” said Kevin Messacar, MD, an infectious disease doctor at Children’s Hospital Colorado, Aurora. “Just because we get through the winter and don’t see much RSV or influenza doesn’t mean we’ll be out of the woods.”
A version of this article first appeared on WebMD.com.
Last fall, health experts said it was possible the United States could experience an easy 2020-21 flu season because health measures to fight COVID-19 would also thwart the spread of influenza.
It looks like that happened – and then some. Numbers are strikingly low for cases of the flu and other common respiratory and gastrointestinal viruses, health experts told the Washington Post.
“It’s crazy,” Lynnette Brammer, MPH, who leads the domestic influenza surveillance team at the Centers for Disease Control and Prevention, told the Washington Post. “This is my 30th flu season. I never would have expected to see flu activity this low.”
Influenza A, influenza B, parainfluenza, norovirus, respiratory syncytial virus, human metapneumovirus, and the bacteria that cause whooping cough and pneumonia are circulating at near-record-low levels.
As an example, the Washington Post said in the third week of December 2019, the CDC’s network of clinical labs reported 16.2% of almost 30,000 samples tested positive for influenza A. During the same period in 2020, only 0.3% tested positive.
But there’s a possible downside to this suppression of viruses, because flu and other viruses may rebound once the coronavirus is brought under control.
“The best analogy is to a forest fire,” Bryan Grenfell, PhD, an epidemiologist and population biologist at Princeton (N.J.) University, told the Washington Post. “For the fire to spread, it needs to have unburned wood. For epidemics to spread, they require people who haven’t previously been infected. So if people don’t get infected this year by these viruses, they likely will at some point later on.”
American health experts like Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Disease, said last fall that they noticed Australia and other nations in the southern hemisphere had easy flu seasons, apparently because of COVID protection measures. The flu season there runs March through August.
COVID-19 now has a very low presence in Australia, but in recent months the flu has been making a comeback. Flu cases among children aged 5 and younger rose sixfold by December, when such cases are usually at their lowest, the Washington Post said.
“That’s an important cautionary tale for us,” said Kevin Messacar, MD, an infectious disease doctor at Children’s Hospital Colorado, Aurora. “Just because we get through the winter and don’t see much RSV or influenza doesn’t mean we’ll be out of the woods.”
A version of this article first appeared on WebMD.com.