User login
AVAHO
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]


Few of those eligible get lung cancer screening, despite USPSTF recommendations
Only 12.8% of eligible adults get CT screening for lung cancer, despite recommendations from the U.S. Preventive Services Task Force.
Kristin G. Maki, PhD, with Karmanos Cancer Institute, Wayne State University, Detroit, led a team that estimated lung cancer screening (LCS) from the 2021 Behavioral Risk Factor Surveillance System in four states (Maine, Michigan, New Jersey, and Rhode Island).
“Increasing LCS among eligible adults is a national priority,” the authors wrote in the study, published online in JAMA Network Open. Lung cancer remains the top cause of cancer in the United States and smoking accounts for approximately 90% of cases.
Screening much higher for other cancers
The authors pointed out that screening rates for eligible people are much higher for other cancers. Melzer and colleagues wrote in a 2021 editorial that breast and colon cancer screening rates are near 70% “despite combined annual death rates less than two-thirds that of lung cancer.”
The USPSTF updated its recommendations for lung cancer screening in March 2021.
Eligibility now includes anyone aged between 50 and 80 years who has smoked at least 20 pack-years and either still smokes or quit within the last 15 years.
The researchers found that, when comparing screening by health status, the highest odds for screening were seen in those who reported they were in poor health, which is concerning, the authors note, because those patients may not be healthy enough to benefit from treatment for their lung cancer.
The odds ratio for getting screening was 2.88 (95% confidence interval, 0.85-9.77) times higher than that of the reference group, which reported excellent health.
Rates differ by state
Consistent with previous studies, this analysis found that screening rates differed by state. Their analysis, for example, showed a higher likelihood of screening for respondents in Rhode Island, compared with Maine (OR, 1.96; 95% CI, 1.05-3.67; P = .03).
Patients who reported having a primary health professional were more than five times more likely to undergo screening, compared with those without one (OR, 5.62; 95% CI, 1.19-26.49).
The authors said their results also highlight the need for Medicare coverage for screening as those with public insurance had lower odds of screening than those with private insurance (OR, 0.81; 95% CI, 0.42-1.56).
Neelima Navuluri, MD, assistant professor in the division of pulmonary, allergy, and critical care at Duke University and the Duke Global Health Institute, both in Durham, N.C., pointed out that the study highlights age, smoking status, and health care access as key factors associated with lack of uptake.
Work needed on all levels
Dr. Navuluri said in an interview that multifaceted patient-, provider- and system-level interventions are needed to improve screening rates.
“For example, we need more community engagement to increase knowledge and awareness of eligibility for lung cancer screening,” she said.
She highlighted the need for interventions around improving and streamlining shared decision-making conversations about screening (a CMS requirement that does not exist for other cancer screening).
Emphasis is needed on younger age groups, people who currently smoke, and communities of color as well as policy to improve insurance coverage of screening, she said.
Dr. Navuluri, who also works with the Durham Veterans Affairs Medical Center, was lead author on a study published in JAMA Network Open on racial disparities in screening among veterans.
“We demonstrate similar findings related to age, smoking status, and poor health status,” she said. “We discuss the need for more qualitative studies to better understand the role of these factors as well as implementation studies to assess effectiveness of various interventions to improve disparities in lung cancer screening rates.”
“Research to identify facilitators for LCS among persons who currently smoke is needed, including a focus on the role of stigma as a barrier to screening,” they wrote.
One coauthor is supported by the cancer prevention and research training program at the University of Texas MD Anderson Cancer Center and the Cancer Prevention and Research Institute of Texas. No other disclosures were reported. Dr. Navuluri receives funding from the National Comprehensive Cancer Network for work on lung cancer screening.
Only 12.8% of eligible adults get CT screening for lung cancer, despite recommendations from the U.S. Preventive Services Task Force.
Kristin G. Maki, PhD, with Karmanos Cancer Institute, Wayne State University, Detroit, led a team that estimated lung cancer screening (LCS) from the 2021 Behavioral Risk Factor Surveillance System in four states (Maine, Michigan, New Jersey, and Rhode Island).
“Increasing LCS among eligible adults is a national priority,” the authors wrote in the study, published online in JAMA Network Open. Lung cancer remains the top cause of cancer in the United States and smoking accounts for approximately 90% of cases.
Screening much higher for other cancers
The authors pointed out that screening rates for eligible people are much higher for other cancers. Melzer and colleagues wrote in a 2021 editorial that breast and colon cancer screening rates are near 70% “despite combined annual death rates less than two-thirds that of lung cancer.”
The USPSTF updated its recommendations for lung cancer screening in March 2021.
Eligibility now includes anyone aged between 50 and 80 years who has smoked at least 20 pack-years and either still smokes or quit within the last 15 years.
The researchers found that, when comparing screening by health status, the highest odds for screening were seen in those who reported they were in poor health, which is concerning, the authors note, because those patients may not be healthy enough to benefit from treatment for their lung cancer.
The odds ratio for getting screening was 2.88 (95% confidence interval, 0.85-9.77) times higher than that of the reference group, which reported excellent health.
Rates differ by state
Consistent with previous studies, this analysis found that screening rates differed by state. Their analysis, for example, showed a higher likelihood of screening for respondents in Rhode Island, compared with Maine (OR, 1.96; 95% CI, 1.05-3.67; P = .03).
Patients who reported having a primary health professional were more than five times more likely to undergo screening, compared with those without one (OR, 5.62; 95% CI, 1.19-26.49).
The authors said their results also highlight the need for Medicare coverage for screening as those with public insurance had lower odds of screening than those with private insurance (OR, 0.81; 95% CI, 0.42-1.56).
Neelima Navuluri, MD, assistant professor in the division of pulmonary, allergy, and critical care at Duke University and the Duke Global Health Institute, both in Durham, N.C., pointed out that the study highlights age, smoking status, and health care access as key factors associated with lack of uptake.
Work needed on all levels
Dr. Navuluri said in an interview that multifaceted patient-, provider- and system-level interventions are needed to improve screening rates.
“For example, we need more community engagement to increase knowledge and awareness of eligibility for lung cancer screening,” she said.
She highlighted the need for interventions around improving and streamlining shared decision-making conversations about screening (a CMS requirement that does not exist for other cancer screening).
Emphasis is needed on younger age groups, people who currently smoke, and communities of color as well as policy to improve insurance coverage of screening, she said.
Dr. Navuluri, who also works with the Durham Veterans Affairs Medical Center, was lead author on a study published in JAMA Network Open on racial disparities in screening among veterans.
“We demonstrate similar findings related to age, smoking status, and poor health status,” she said. “We discuss the need for more qualitative studies to better understand the role of these factors as well as implementation studies to assess effectiveness of various interventions to improve disparities in lung cancer screening rates.”
“Research to identify facilitators for LCS among persons who currently smoke is needed, including a focus on the role of stigma as a barrier to screening,” they wrote.
One coauthor is supported by the cancer prevention and research training program at the University of Texas MD Anderson Cancer Center and the Cancer Prevention and Research Institute of Texas. No other disclosures were reported. Dr. Navuluri receives funding from the National Comprehensive Cancer Network for work on lung cancer screening.
Only 12.8% of eligible adults get CT screening for lung cancer, despite recommendations from the U.S. Preventive Services Task Force.
Kristin G. Maki, PhD, with Karmanos Cancer Institute, Wayne State University, Detroit, led a team that estimated lung cancer screening (LCS) from the 2021 Behavioral Risk Factor Surveillance System in four states (Maine, Michigan, New Jersey, and Rhode Island).
“Increasing LCS among eligible adults is a national priority,” the authors wrote in the study, published online in JAMA Network Open. Lung cancer remains the top cause of cancer in the United States and smoking accounts for approximately 90% of cases.
Screening much higher for other cancers
The authors pointed out that screening rates for eligible people are much higher for other cancers. Melzer and colleagues wrote in a 2021 editorial that breast and colon cancer screening rates are near 70% “despite combined annual death rates less than two-thirds that of lung cancer.”
The USPSTF updated its recommendations for lung cancer screening in March 2021.
Eligibility now includes anyone aged between 50 and 80 years who has smoked at least 20 pack-years and either still smokes or quit within the last 15 years.
The researchers found that, when comparing screening by health status, the highest odds for screening were seen in those who reported they were in poor health, which is concerning, the authors note, because those patients may not be healthy enough to benefit from treatment for their lung cancer.
The odds ratio for getting screening was 2.88 (95% confidence interval, 0.85-9.77) times higher than that of the reference group, which reported excellent health.
Rates differ by state
Consistent with previous studies, this analysis found that screening rates differed by state. Their analysis, for example, showed a higher likelihood of screening for respondents in Rhode Island, compared with Maine (OR, 1.96; 95% CI, 1.05-3.67; P = .03).
Patients who reported having a primary health professional were more than five times more likely to undergo screening, compared with those without one (OR, 5.62; 95% CI, 1.19-26.49).
The authors said their results also highlight the need for Medicare coverage for screening as those with public insurance had lower odds of screening than those with private insurance (OR, 0.81; 95% CI, 0.42-1.56).
Neelima Navuluri, MD, assistant professor in the division of pulmonary, allergy, and critical care at Duke University and the Duke Global Health Institute, both in Durham, N.C., pointed out that the study highlights age, smoking status, and health care access as key factors associated with lack of uptake.
Work needed on all levels
Dr. Navuluri said in an interview that multifaceted patient-, provider- and system-level interventions are needed to improve screening rates.
“For example, we need more community engagement to increase knowledge and awareness of eligibility for lung cancer screening,” she said.
She highlighted the need for interventions around improving and streamlining shared decision-making conversations about screening (a CMS requirement that does not exist for other cancer screening).
Emphasis is needed on younger age groups, people who currently smoke, and communities of color as well as policy to improve insurance coverage of screening, she said.
Dr. Navuluri, who also works with the Durham Veterans Affairs Medical Center, was lead author on a study published in JAMA Network Open on racial disparities in screening among veterans.
“We demonstrate similar findings related to age, smoking status, and poor health status,” she said. “We discuss the need for more qualitative studies to better understand the role of these factors as well as implementation studies to assess effectiveness of various interventions to improve disparities in lung cancer screening rates.”
“Research to identify facilitators for LCS among persons who currently smoke is needed, including a focus on the role of stigma as a barrier to screening,” they wrote.
One coauthor is supported by the cancer prevention and research training program at the University of Texas MD Anderson Cancer Center and the Cancer Prevention and Research Institute of Texas. No other disclosures were reported. Dr. Navuluri receives funding from the National Comprehensive Cancer Network for work on lung cancer screening.
FROM JAMA NETWORK OPEN
Gilteritinib maintenance reduces relapse in MRD+ AML
The research was presented at the European Hematology Association Hybrid Congress 2023.
For the study, AML patients with the most common form of mutation in the proto-oncogene fms-like tyrosine kinase 3 (FLT3), known as the internal tandem duplication (ITD), were randomized to 24 months of maintenance therapy with either the FLT3 inhibitor gilteritinib or placebo.
The trial did not meet its primary endpoint, as there was no significant difference in relapse-free survival (RFS) between those assigned to the active drug and those given placebo, and there was no difference in overall survival rates.
However, subgroup analysis revealed that FLT3/ITD AML patients who were MRD+ after transplant, which represented approximately half of the participants, experienced a significant 48% improvement in RFS with gilteritinib versus placebo, while no benefit was seen in MRD– patients.
While acknowledging that the trial did not meet its primary endpoint, presenter Mark J. Levis, MD, PhD, program leader, hematologic malignancies and bone marrow transplant program, Johns Hopkins Sidney Kimmel Comprehensive Cancer Center, Baltimore, said it was nevertheless “a successful study.”
“We learned how to use these drugs and in whom,” he continued, adding: “No, not everybody needs and should get a FLT3 inhibitor post-transplant, but we can use this [MRD] assay to identify who.”
Consequently, Dr. Levis believes that gilteritinib “should be a standard of care for those who are MRD positive,” although the decision to use it “should be balanced against the potential for toxicity,” compared with not adding an additional treatment after HCT.
He told a press conference that “we’re going to certainly make sure that patients who are MRD positive get [gilteritinib],” although the MRD negative patients “are going to be more questionable,” especially because the assay that they used in the study is not “perfect.”
Dr. Levis also suggested that the trial did not meet its endpoint because of regional differences in the clinical practice, such as in the number of treatment cycles prior to HCT, the time to transplant, and the previous use of a FLT3 inhibitor, all of which may have skewed the findings.
“Everybody in the world is convinced that they’re the best transplanter,” he said, and yet “they all do it differently, and the heterogeneity is astounding.”
He added: “If we’d restricted everybody [to a] pretransplant regimen, I suspect we would have had a different result than what we’re getting here, but this is releasing the drug into the world and saying: ‘Here, transplant however you want, however it’s practiced in the real world. Tell us how this works.’ ”
Approached for comment, Claudio Brunstein, MD, PhD, vice-chair of the department of hematology and oncology in the Cleveland Clinic Taussig Cancer Institute, said that while there was “some disappointment” with the results, he was “not surprised” that the trial did not meet its primary endpoint.
He said in an interview that the patient population was not of “high enough risk” to demonstrate an overall difference between gilteritinib and placebo, although he conceded that it is “hard to get to high-risk patients in a timely way” and so conduct a trial with them.
As to the notion that variations in clinical practice could have been responsible, Dr. Brunstein pointed out that it was a randomized trial, so the issue would have applied equally to both sides.
He nevertheless believes that it is “a very important study,” and “just the fact that it was done in the context of a number of drugs coming and being approved by the [U.S. Food and Drug Administration] in AML is quite remarkable.”
This is especially the case given that “many centers are already using [gilteritinib] as off-label maintenance therapy.”
Dr. Brunstein added that it is “good news” that the drug was effective in MRD+ patients, as it shows “you can overcome that with maintenance therapy rather than keeping giving more and more chemotherapy, especially as there are patients you’re worried about giving more intensive chemotherapy to make them MRD negative.”
He pointed out, however, that the assay used in the trial was “research grade” and very sensitive to MRD and “is not available everywhere, so there is an adjustment that the community will have to do to in order to apply this data.”
“But for those who are more obviously MRD positive with less sensitive assays, gilteritinib is already something that can be used,” Dr. Brunstein said.
Presenting the findings, Dr. Levis stated: “We all know that patients with FLT3/ITD AML have a high risk of relapse and are routinely referred for transplant. And we know that the detection of measurable residual disease pretransplant is highly predictive of outcome post-transplant.”
He continued that FLT3 inhibitors are “routinely given as post-transplant maintenance ... based on some prior trials, mostly with sorafenib.”
“But uncertainty exists as to the broad applicability of these trials,” Dr. Levis said. Moreover, the use of sorafenib in this context is “off label and can be difficult to tolerate,” and “we know that most patients are cured with allogeneic transplant alone.”
Gilteritinib is already known to be well tolerated as a monotherapy, and was approved by the FDA for the treatment of adult patients with FLT3 mutation–positive relapsed or refractory AML in 2018.
The investigators therefore examined whether it would be beneficial as a post-HCT maintenance therapy in FLT3-ITD AML. Patients were required to be in morphologic remission after one or two courses of induction therapy, with Dr. Levis underlining: “We did not allow patients who had been salvaged onto the study.”
They subsequently had a marrow aspirate sample taken for MRD analysis before undergoing allogeneic transplant, with any conditioning regimen, donor, or graft-versus-host disease (GVHD) prophylaxis allowed.
Between 30 and 90 days later, patients with successful engraftment who were able to take oral medication were then randomized to 24 months of maintenance therapy with either gilteritinib or placebo.
Dr. Levis showed that, among 620 patients screened at 110 centers in 16 countries, 356 were randomized between Aug. 15, 2017, and July 8, 2020. The median age was 53 years, and 49% of gilteritinib patients and 48% of those given placebo were female.
He noted that there was a “fairly even global distribution” of patients from North America, Europe, and the Asia/Pacific region, and that 60% of patients underwent a myeloablative conditioning regimen. Approximately the same proportion had received an FLT3 inhibitor prior to HCT.
MRD positivity, assessed at a cell count of ≥ 10-6, was observed pre-HCT in 47% of patients in both treatment groups, and in 50% of gilteritinib patients and 51% of placebo patients at both pre- and post-transplant assessments.
The treatment regimen was completed by 52.8% of patients assigned to gilteritinib and 53.9% in the placebo arm. Dr. Levis said that 18.5% and 20.3% of patients, respectively, experienced a grade 3/4 treatment emergent acute GVHD event, while 32.6% and 21.5%, respectively, had a grade ≥ 3 treatment emergent infection.
He noted that “adverse events were clearly more common in the gilteritinib arm and often led to either dose reduction or interruption, or withdrawal of treatment.”
The most common grade ≥ 3 treatment emergent adverse event was a decrease in neutrophil count, seen in 24.7% of gilteritinib patients and 7.9% of those given placebo, followed by reduced platelet count, in 15.2% and 5.6%, respectively, and anemia, in 6.2% and 1.7%, respectively.
Turning to the efficacy outcomes, Dr. Levis reported that the trial did not meet its primary endpoint, with no significant difference in RFS between the gilteritinib and placebo arms, at a hazard ratio of 0.679 (P = .0518). There was also no significant difference in the key secondary objective of overall survival, at a hazard ratio of 0.846 (P = .4394).
However, Dr. Levis noted that there was a “clear difference in the benefit of gilteritinib by region,” and, “at every level,” MRD predicted a benefit from gilteritinib, which he said was a “big surprise” and “really leapt out in the subgroup analysis.”
He explained that the researchers used a modified version of a two-step assay that has been used in previous studies, and was able to detect MRD at a sensitivity of approximately 1x10-6. “In our study, 98% of participants had samples pre- and post-[transplant].”
Regardless of treatment arm, MRD positivity measured at that sensitivity was associated with a significant reduction in overall survival, at a hazard ratio versus MRD– status of 0.514 (P = .0025).
When stratifying the patients by MRD status, the researchers found that, among MRD+ participants, gilteritinib was associated with a significant improvement in RFS, at a hazard ratio versus placebo of 0.515 (P = .0065), while there was no significant difference in MRD– patients.
Stratifying the patients by their conditioning regimen prior to HCT also revealed differences, with those undergoing myeloablative conditioning having significantly greater overall survival than those who underwent reduced-intensity conditioning, at a hazard ratio for death of 0.529 (P = .0027).
Dr. Levis said there is “no surprise there,” and the result could reflect the selection of fitter, younger patients to undergo the more intensive regimen.
He then showed that MRD+ patients who had undergone myeloablative conditioning had better overall survival with gilteritinib than placebo, at a hazard ratio for death of 0.418 (P = .0087). Again, the difference disappeared when looking at MRD– patients.
“So conditioning doesn’t help you in the setting of MRD,” Dr. Levis said.
Finally, he took a deeper dive into the regional differences in outcomes, noting that patients in the Asia/Pacific region, where gilteritinib showed no benefit over placebo, “were 10 years younger” than those in other regions, “tended to get myeloablative conditioning, and hardly ever used FLT3 inhibitors.”
In contrast, North American patients, who experienced a significant gilteritinib benefit in terms of RFS, underwent HCT an average of 26 days earlier than those elsewhere, and received fewer courses of chemotherapy pre-HCT. Moreover, 93.5% received an FLT3 inhibitor pretransplant.
The study was funded by Astellas Pharma Global Development. Dr. Levis declares relationships with Abbvie, Amgen, Astellas, Bristol-Myers-Squibb, Daiichi-Sankyo, GlaxoSmithKline, Jazz, Menarini, Pfizer, Sumitomo-Dainippon, Syndax, Takeda. Dr. Brunstein declares no relevant relationships.
The research was presented at the European Hematology Association Hybrid Congress 2023.
For the study, AML patients with the most common form of mutation in the proto-oncogene fms-like tyrosine kinase 3 (FLT3), known as the internal tandem duplication (ITD), were randomized to 24 months of maintenance therapy with either the FLT3 inhibitor gilteritinib or placebo.
The trial did not meet its primary endpoint, as there was no significant difference in relapse-free survival (RFS) between those assigned to the active drug and those given placebo, and there was no difference in overall survival rates.
However, subgroup analysis revealed that FLT3/ITD AML patients who were MRD+ after transplant, which represented approximately half of the participants, experienced a significant 48% improvement in RFS with gilteritinib versus placebo, while no benefit was seen in MRD– patients.
While acknowledging that the trial did not meet its primary endpoint, presenter Mark J. Levis, MD, PhD, program leader, hematologic malignancies and bone marrow transplant program, Johns Hopkins Sidney Kimmel Comprehensive Cancer Center, Baltimore, said it was nevertheless “a successful study.”
“We learned how to use these drugs and in whom,” he continued, adding: “No, not everybody needs and should get a FLT3 inhibitor post-transplant, but we can use this [MRD] assay to identify who.”
Consequently, Dr. Levis believes that gilteritinib “should be a standard of care for those who are MRD positive,” although the decision to use it “should be balanced against the potential for toxicity,” compared with not adding an additional treatment after HCT.
He told a press conference that “we’re going to certainly make sure that patients who are MRD positive get [gilteritinib],” although the MRD negative patients “are going to be more questionable,” especially because the assay that they used in the study is not “perfect.”
Dr. Levis also suggested that the trial did not meet its endpoint because of regional differences in the clinical practice, such as in the number of treatment cycles prior to HCT, the time to transplant, and the previous use of a FLT3 inhibitor, all of which may have skewed the findings.
“Everybody in the world is convinced that they’re the best transplanter,” he said, and yet “they all do it differently, and the heterogeneity is astounding.”
He added: “If we’d restricted everybody [to a] pretransplant regimen, I suspect we would have had a different result than what we’re getting here, but this is releasing the drug into the world and saying: ‘Here, transplant however you want, however it’s practiced in the real world. Tell us how this works.’ ”
Approached for comment, Claudio Brunstein, MD, PhD, vice-chair of the department of hematology and oncology in the Cleveland Clinic Taussig Cancer Institute, said that while there was “some disappointment” with the results, he was “not surprised” that the trial did not meet its primary endpoint.
He said in an interview that the patient population was not of “high enough risk” to demonstrate an overall difference between gilteritinib and placebo, although he conceded that it is “hard to get to high-risk patients in a timely way” and so conduct a trial with them.
As to the notion that variations in clinical practice could have been responsible, Dr. Brunstein pointed out that it was a randomized trial, so the issue would have applied equally to both sides.
He nevertheless believes that it is “a very important study,” and “just the fact that it was done in the context of a number of drugs coming and being approved by the [U.S. Food and Drug Administration] in AML is quite remarkable.”
This is especially the case given that “many centers are already using [gilteritinib] as off-label maintenance therapy.”
Dr. Brunstein added that it is “good news” that the drug was effective in MRD+ patients, as it shows “you can overcome that with maintenance therapy rather than keeping giving more and more chemotherapy, especially as there are patients you’re worried about giving more intensive chemotherapy to make them MRD negative.”
He pointed out, however, that the assay used in the trial was “research grade” and very sensitive to MRD and “is not available everywhere, so there is an adjustment that the community will have to do to in order to apply this data.”
“But for those who are more obviously MRD positive with less sensitive assays, gilteritinib is already something that can be used,” Dr. Brunstein said.
Presenting the findings, Dr. Levis stated: “We all know that patients with FLT3/ITD AML have a high risk of relapse and are routinely referred for transplant. And we know that the detection of measurable residual disease pretransplant is highly predictive of outcome post-transplant.”
He continued that FLT3 inhibitors are “routinely given as post-transplant maintenance ... based on some prior trials, mostly with sorafenib.”
“But uncertainty exists as to the broad applicability of these trials,” Dr. Levis said. Moreover, the use of sorafenib in this context is “off label and can be difficult to tolerate,” and “we know that most patients are cured with allogeneic transplant alone.”
Gilteritinib is already known to be well tolerated as a monotherapy, and was approved by the FDA for the treatment of adult patients with FLT3 mutation–positive relapsed or refractory AML in 2018.
The investigators therefore examined whether it would be beneficial as a post-HCT maintenance therapy in FLT3-ITD AML. Patients were required to be in morphologic remission after one or two courses of induction therapy, with Dr. Levis underlining: “We did not allow patients who had been salvaged onto the study.”
They subsequently had a marrow aspirate sample taken for MRD analysis before undergoing allogeneic transplant, with any conditioning regimen, donor, or graft-versus-host disease (GVHD) prophylaxis allowed.
Between 30 and 90 days later, patients with successful engraftment who were able to take oral medication were then randomized to 24 months of maintenance therapy with either gilteritinib or placebo.
Dr. Levis showed that, among 620 patients screened at 110 centers in 16 countries, 356 were randomized between Aug. 15, 2017, and July 8, 2020. The median age was 53 years, and 49% of gilteritinib patients and 48% of those given placebo were female.
He noted that there was a “fairly even global distribution” of patients from North America, Europe, and the Asia/Pacific region, and that 60% of patients underwent a myeloablative conditioning regimen. Approximately the same proportion had received an FLT3 inhibitor prior to HCT.
MRD positivity, assessed at a cell count of ≥ 10-6, was observed pre-HCT in 47% of patients in both treatment groups, and in 50% of gilteritinib patients and 51% of placebo patients at both pre- and post-transplant assessments.
The treatment regimen was completed by 52.8% of patients assigned to gilteritinib and 53.9% in the placebo arm. Dr. Levis said that 18.5% and 20.3% of patients, respectively, experienced a grade 3/4 treatment emergent acute GVHD event, while 32.6% and 21.5%, respectively, had a grade ≥ 3 treatment emergent infection.
He noted that “adverse events were clearly more common in the gilteritinib arm and often led to either dose reduction or interruption, or withdrawal of treatment.”
The most common grade ≥ 3 treatment emergent adverse event was a decrease in neutrophil count, seen in 24.7% of gilteritinib patients and 7.9% of those given placebo, followed by reduced platelet count, in 15.2% and 5.6%, respectively, and anemia, in 6.2% and 1.7%, respectively.
Turning to the efficacy outcomes, Dr. Levis reported that the trial did not meet its primary endpoint, with no significant difference in RFS between the gilteritinib and placebo arms, at a hazard ratio of 0.679 (P = .0518). There was also no significant difference in the key secondary objective of overall survival, at a hazard ratio of 0.846 (P = .4394).
However, Dr. Levis noted that there was a “clear difference in the benefit of gilteritinib by region,” and, “at every level,” MRD predicted a benefit from gilteritinib, which he said was a “big surprise” and “really leapt out in the subgroup analysis.”
He explained that the researchers used a modified version of a two-step assay that has been used in previous studies, and was able to detect MRD at a sensitivity of approximately 1x10-6. “In our study, 98% of participants had samples pre- and post-[transplant].”
Regardless of treatment arm, MRD positivity measured at that sensitivity was associated with a significant reduction in overall survival, at a hazard ratio versus MRD– status of 0.514 (P = .0025).
When stratifying the patients by MRD status, the researchers found that, among MRD+ participants, gilteritinib was associated with a significant improvement in RFS, at a hazard ratio versus placebo of 0.515 (P = .0065), while there was no significant difference in MRD– patients.
Stratifying the patients by their conditioning regimen prior to HCT also revealed differences, with those undergoing myeloablative conditioning having significantly greater overall survival than those who underwent reduced-intensity conditioning, at a hazard ratio for death of 0.529 (P = .0027).
Dr. Levis said there is “no surprise there,” and the result could reflect the selection of fitter, younger patients to undergo the more intensive regimen.
He then showed that MRD+ patients who had undergone myeloablative conditioning had better overall survival with gilteritinib than placebo, at a hazard ratio for death of 0.418 (P = .0087). Again, the difference disappeared when looking at MRD– patients.
“So conditioning doesn’t help you in the setting of MRD,” Dr. Levis said.
Finally, he took a deeper dive into the regional differences in outcomes, noting that patients in the Asia/Pacific region, where gilteritinib showed no benefit over placebo, “were 10 years younger” than those in other regions, “tended to get myeloablative conditioning, and hardly ever used FLT3 inhibitors.”
In contrast, North American patients, who experienced a significant gilteritinib benefit in terms of RFS, underwent HCT an average of 26 days earlier than those elsewhere, and received fewer courses of chemotherapy pre-HCT. Moreover, 93.5% received an FLT3 inhibitor pretransplant.
The study was funded by Astellas Pharma Global Development. Dr. Levis declares relationships with Abbvie, Amgen, Astellas, Bristol-Myers-Squibb, Daiichi-Sankyo, GlaxoSmithKline, Jazz, Menarini, Pfizer, Sumitomo-Dainippon, Syndax, Takeda. Dr. Brunstein declares no relevant relationships.
The research was presented at the European Hematology Association Hybrid Congress 2023.
For the study, AML patients with the most common form of mutation in the proto-oncogene fms-like tyrosine kinase 3 (FLT3), known as the internal tandem duplication (ITD), were randomized to 24 months of maintenance therapy with either the FLT3 inhibitor gilteritinib or placebo.
The trial did not meet its primary endpoint, as there was no significant difference in relapse-free survival (RFS) between those assigned to the active drug and those given placebo, and there was no difference in overall survival rates.
However, subgroup analysis revealed that FLT3/ITD AML patients who were MRD+ after transplant, which represented approximately half of the participants, experienced a significant 48% improvement in RFS with gilteritinib versus placebo, while no benefit was seen in MRD– patients.
While acknowledging that the trial did not meet its primary endpoint, presenter Mark J. Levis, MD, PhD, program leader, hematologic malignancies and bone marrow transplant program, Johns Hopkins Sidney Kimmel Comprehensive Cancer Center, Baltimore, said it was nevertheless “a successful study.”
“We learned how to use these drugs and in whom,” he continued, adding: “No, not everybody needs and should get a FLT3 inhibitor post-transplant, but we can use this [MRD] assay to identify who.”
Consequently, Dr. Levis believes that gilteritinib “should be a standard of care for those who are MRD positive,” although the decision to use it “should be balanced against the potential for toxicity,” compared with not adding an additional treatment after HCT.
He told a press conference that “we’re going to certainly make sure that patients who are MRD positive get [gilteritinib],” although the MRD negative patients “are going to be more questionable,” especially because the assay that they used in the study is not “perfect.”
Dr. Levis also suggested that the trial did not meet its endpoint because of regional differences in the clinical practice, such as in the number of treatment cycles prior to HCT, the time to transplant, and the previous use of a FLT3 inhibitor, all of which may have skewed the findings.
“Everybody in the world is convinced that they’re the best transplanter,” he said, and yet “they all do it differently, and the heterogeneity is astounding.”
He added: “If we’d restricted everybody [to a] pretransplant regimen, I suspect we would have had a different result than what we’re getting here, but this is releasing the drug into the world and saying: ‘Here, transplant however you want, however it’s practiced in the real world. Tell us how this works.’ ”
Approached for comment, Claudio Brunstein, MD, PhD, vice-chair of the department of hematology and oncology in the Cleveland Clinic Taussig Cancer Institute, said that while there was “some disappointment” with the results, he was “not surprised” that the trial did not meet its primary endpoint.
He said in an interview that the patient population was not of “high enough risk” to demonstrate an overall difference between gilteritinib and placebo, although he conceded that it is “hard to get to high-risk patients in a timely way” and so conduct a trial with them.
As to the notion that variations in clinical practice could have been responsible, Dr. Brunstein pointed out that it was a randomized trial, so the issue would have applied equally to both sides.
He nevertheless believes that it is “a very important study,” and “just the fact that it was done in the context of a number of drugs coming and being approved by the [U.S. Food and Drug Administration] in AML is quite remarkable.”
This is especially the case given that “many centers are already using [gilteritinib] as off-label maintenance therapy.”
Dr. Brunstein added that it is “good news” that the drug was effective in MRD+ patients, as it shows “you can overcome that with maintenance therapy rather than keeping giving more and more chemotherapy, especially as there are patients you’re worried about giving more intensive chemotherapy to make them MRD negative.”
He pointed out, however, that the assay used in the trial was “research grade” and very sensitive to MRD and “is not available everywhere, so there is an adjustment that the community will have to do to in order to apply this data.”
“But for those who are more obviously MRD positive with less sensitive assays, gilteritinib is already something that can be used,” Dr. Brunstein said.
Presenting the findings, Dr. Levis stated: “We all know that patients with FLT3/ITD AML have a high risk of relapse and are routinely referred for transplant. And we know that the detection of measurable residual disease pretransplant is highly predictive of outcome post-transplant.”
He continued that FLT3 inhibitors are “routinely given as post-transplant maintenance ... based on some prior trials, mostly with sorafenib.”
“But uncertainty exists as to the broad applicability of these trials,” Dr. Levis said. Moreover, the use of sorafenib in this context is “off label and can be difficult to tolerate,” and “we know that most patients are cured with allogeneic transplant alone.”
Gilteritinib is already known to be well tolerated as a monotherapy, and was approved by the FDA for the treatment of adult patients with FLT3 mutation–positive relapsed or refractory AML in 2018.
The investigators therefore examined whether it would be beneficial as a post-HCT maintenance therapy in FLT3-ITD AML. Patients were required to be in morphologic remission after one or two courses of induction therapy, with Dr. Levis underlining: “We did not allow patients who had been salvaged onto the study.”
They subsequently had a marrow aspirate sample taken for MRD analysis before undergoing allogeneic transplant, with any conditioning regimen, donor, or graft-versus-host disease (GVHD) prophylaxis allowed.
Between 30 and 90 days later, patients with successful engraftment who were able to take oral medication were then randomized to 24 months of maintenance therapy with either gilteritinib or placebo.
Dr. Levis showed that, among 620 patients screened at 110 centers in 16 countries, 356 were randomized between Aug. 15, 2017, and July 8, 2020. The median age was 53 years, and 49% of gilteritinib patients and 48% of those given placebo were female.
He noted that there was a “fairly even global distribution” of patients from North America, Europe, and the Asia/Pacific region, and that 60% of patients underwent a myeloablative conditioning regimen. Approximately the same proportion had received an FLT3 inhibitor prior to HCT.
MRD positivity, assessed at a cell count of ≥ 10-6, was observed pre-HCT in 47% of patients in both treatment groups, and in 50% of gilteritinib patients and 51% of placebo patients at both pre- and post-transplant assessments.
The treatment regimen was completed by 52.8% of patients assigned to gilteritinib and 53.9% in the placebo arm. Dr. Levis said that 18.5% and 20.3% of patients, respectively, experienced a grade 3/4 treatment emergent acute GVHD event, while 32.6% and 21.5%, respectively, had a grade ≥ 3 treatment emergent infection.
He noted that “adverse events were clearly more common in the gilteritinib arm and often led to either dose reduction or interruption, or withdrawal of treatment.”
The most common grade ≥ 3 treatment emergent adverse event was a decrease in neutrophil count, seen in 24.7% of gilteritinib patients and 7.9% of those given placebo, followed by reduced platelet count, in 15.2% and 5.6%, respectively, and anemia, in 6.2% and 1.7%, respectively.
Turning to the efficacy outcomes, Dr. Levis reported that the trial did not meet its primary endpoint, with no significant difference in RFS between the gilteritinib and placebo arms, at a hazard ratio of 0.679 (P = .0518). There was also no significant difference in the key secondary objective of overall survival, at a hazard ratio of 0.846 (P = .4394).
However, Dr. Levis noted that there was a “clear difference in the benefit of gilteritinib by region,” and, “at every level,” MRD predicted a benefit from gilteritinib, which he said was a “big surprise” and “really leapt out in the subgroup analysis.”
He explained that the researchers used a modified version of a two-step assay that has been used in previous studies, and was able to detect MRD at a sensitivity of approximately 1x10-6. “In our study, 98% of participants had samples pre- and post-[transplant].”
Regardless of treatment arm, MRD positivity measured at that sensitivity was associated with a significant reduction in overall survival, at a hazard ratio versus MRD– status of 0.514 (P = .0025).
When stratifying the patients by MRD status, the researchers found that, among MRD+ participants, gilteritinib was associated with a significant improvement in RFS, at a hazard ratio versus placebo of 0.515 (P = .0065), while there was no significant difference in MRD– patients.
Stratifying the patients by their conditioning regimen prior to HCT also revealed differences, with those undergoing myeloablative conditioning having significantly greater overall survival than those who underwent reduced-intensity conditioning, at a hazard ratio for death of 0.529 (P = .0027).
Dr. Levis said there is “no surprise there,” and the result could reflect the selection of fitter, younger patients to undergo the more intensive regimen.
He then showed that MRD+ patients who had undergone myeloablative conditioning had better overall survival with gilteritinib than placebo, at a hazard ratio for death of 0.418 (P = .0087). Again, the difference disappeared when looking at MRD– patients.
“So conditioning doesn’t help you in the setting of MRD,” Dr. Levis said.
Finally, he took a deeper dive into the regional differences in outcomes, noting that patients in the Asia/Pacific region, where gilteritinib showed no benefit over placebo, “were 10 years younger” than those in other regions, “tended to get myeloablative conditioning, and hardly ever used FLT3 inhibitors.”
In contrast, North American patients, who experienced a significant gilteritinib benefit in terms of RFS, underwent HCT an average of 26 days earlier than those elsewhere, and received fewer courses of chemotherapy pre-HCT. Moreover, 93.5% received an FLT3 inhibitor pretransplant.
The study was funded by Astellas Pharma Global Development. Dr. Levis declares relationships with Abbvie, Amgen, Astellas, Bristol-Myers-Squibb, Daiichi-Sankyo, GlaxoSmithKline, Jazz, Menarini, Pfizer, Sumitomo-Dainippon, Syndax, Takeda. Dr. Brunstein declares no relevant relationships.
AT EHA 2023
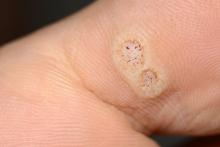
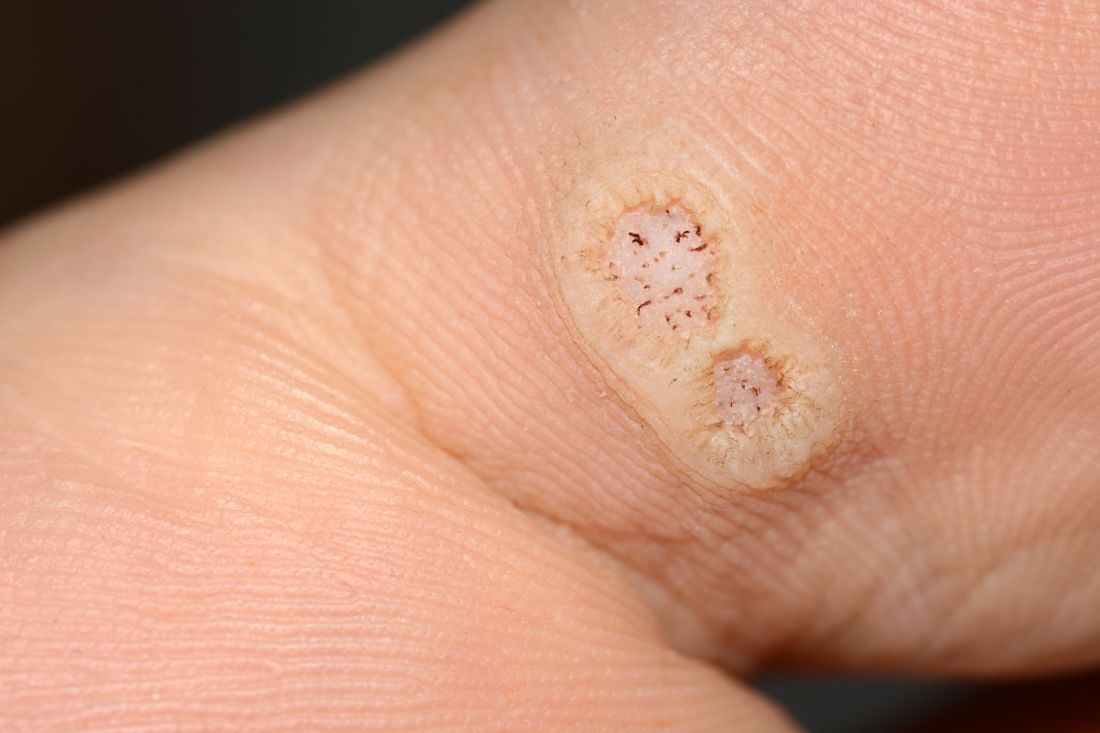
Warts difficult to eradicate in immunocompromised children
.
Only a quarter of patients (24%) who were undergoing active cancer treatment experienced complete resolution of their warts, compared with 63.3% of patients who were not on active treatment.
In addition, warts persisted or worsened in 56.0% of patients receiving active treatment compared with 13.4% of those who were not receiving it.
“These data enable providers treating warts in children with cancer to have an educated discussion regarding the expected clinical progression of warts and the likelihood of response to wart therapy while on and off anti-cancer treatment,” the authors wrote in the study, published in Pediatric Dermatology.
In immunocompromised children, warts are more common than in the general pediatric population, and more resistant to treatment. But as the authors noted, data on the course and prognosis of warts in pediatric patients who are actively receiving anti-cancer therapy compared with patients who have completed treatment are limited.
Tina Ho, MD, PhD, of the department of dermatology, and colleagues from Boston Children’s Hospital, sought to analyze the clinical course of warts treated in this patient population at their institution over a 10-year period. They conducted a retrospective study of 72 children who were treated for cancer between 2011 and 2021, and who had also been treated for warts.
The median age of the cohort was 12 years, and they were followed for a median of 2 years following their diagnosis of warts. Within this group, more than half (55%) had hematologic malignancies, while 27% had a history of bone marrow transplantation.
Of note, the authors pointed out, 54% of the patients had plantar warts, and 60% of patients (38 of 63) with a documented number of warts had more than five at the time of presentation.
The treatment regimens that the children had received varied, with 81% of patients receiving cytotoxic chemotherapy and 23% of patients on targeted therapies that included immunotherapy.
The warts were most commonly treated with cryotherapy and topical salicylic acid; this was the case for those actively receiving oncology treatment or those who had completed their treatment regimens.
Outcomes of wart treatments were available in 25 of the patients undergoing active cancer treatment and in 30 of those who had completed treatment. For children on active oncology treatment, 5 (20%) achieved partial resolution, 6 (24%) achieved complete resolution, and 14 (56%) experienced persistence or worsening of their warts following therapy. Those who had completed treatment had better outcomes: Seven (23.3%) had a partial response, 19 (63.3%) had complete resolution, and 4 (13.4%) had persistence or worsening of warts after treatment of warts.
The authors also pointed out the treatment of warts can be painful, expensive, and time-consuming. “It is thus imperative that the risks and benefits of these treatments are carefully considered before proceeding with treatment,” wrote Dr. Ho and colleagues. “This is especially true in medically complex children with cancer who may be fearful of procedures and spend significant portions of their young lives within the medical system.”
Limitations to the study include its retrospective design and small sample size. Clinical data were not uniformly complete, and follow-up intervals varied among the participants. Also, it was conducted at a single-institution and at a large tertiary center, so the results may not be fully generalizable.
The authors declared no conflict of interest. No outside funding source was listed.
.
Only a quarter of patients (24%) who were undergoing active cancer treatment experienced complete resolution of their warts, compared with 63.3% of patients who were not on active treatment.
In addition, warts persisted or worsened in 56.0% of patients receiving active treatment compared with 13.4% of those who were not receiving it.
“These data enable providers treating warts in children with cancer to have an educated discussion regarding the expected clinical progression of warts and the likelihood of response to wart therapy while on and off anti-cancer treatment,” the authors wrote in the study, published in Pediatric Dermatology.
In immunocompromised children, warts are more common than in the general pediatric population, and more resistant to treatment. But as the authors noted, data on the course and prognosis of warts in pediatric patients who are actively receiving anti-cancer therapy compared with patients who have completed treatment are limited.
Tina Ho, MD, PhD, of the department of dermatology, and colleagues from Boston Children’s Hospital, sought to analyze the clinical course of warts treated in this patient population at their institution over a 10-year period. They conducted a retrospective study of 72 children who were treated for cancer between 2011 and 2021, and who had also been treated for warts.
The median age of the cohort was 12 years, and they were followed for a median of 2 years following their diagnosis of warts. Within this group, more than half (55%) had hematologic malignancies, while 27% had a history of bone marrow transplantation.
Of note, the authors pointed out, 54% of the patients had plantar warts, and 60% of patients (38 of 63) with a documented number of warts had more than five at the time of presentation.
The treatment regimens that the children had received varied, with 81% of patients receiving cytotoxic chemotherapy and 23% of patients on targeted therapies that included immunotherapy.
The warts were most commonly treated with cryotherapy and topical salicylic acid; this was the case for those actively receiving oncology treatment or those who had completed their treatment regimens.
Outcomes of wart treatments were available in 25 of the patients undergoing active cancer treatment and in 30 of those who had completed treatment. For children on active oncology treatment, 5 (20%) achieved partial resolution, 6 (24%) achieved complete resolution, and 14 (56%) experienced persistence or worsening of their warts following therapy. Those who had completed treatment had better outcomes: Seven (23.3%) had a partial response, 19 (63.3%) had complete resolution, and 4 (13.4%) had persistence or worsening of warts after treatment of warts.
The authors also pointed out the treatment of warts can be painful, expensive, and time-consuming. “It is thus imperative that the risks and benefits of these treatments are carefully considered before proceeding with treatment,” wrote Dr. Ho and colleagues. “This is especially true in medically complex children with cancer who may be fearful of procedures and spend significant portions of their young lives within the medical system.”
Limitations to the study include its retrospective design and small sample size. Clinical data were not uniformly complete, and follow-up intervals varied among the participants. Also, it was conducted at a single-institution and at a large tertiary center, so the results may not be fully generalizable.
The authors declared no conflict of interest. No outside funding source was listed.
.
Only a quarter of patients (24%) who were undergoing active cancer treatment experienced complete resolution of their warts, compared with 63.3% of patients who were not on active treatment.
In addition, warts persisted or worsened in 56.0% of patients receiving active treatment compared with 13.4% of those who were not receiving it.
“These data enable providers treating warts in children with cancer to have an educated discussion regarding the expected clinical progression of warts and the likelihood of response to wart therapy while on and off anti-cancer treatment,” the authors wrote in the study, published in Pediatric Dermatology.
In immunocompromised children, warts are more common than in the general pediatric population, and more resistant to treatment. But as the authors noted, data on the course and prognosis of warts in pediatric patients who are actively receiving anti-cancer therapy compared with patients who have completed treatment are limited.
Tina Ho, MD, PhD, of the department of dermatology, and colleagues from Boston Children’s Hospital, sought to analyze the clinical course of warts treated in this patient population at their institution over a 10-year period. They conducted a retrospective study of 72 children who were treated for cancer between 2011 and 2021, and who had also been treated for warts.
The median age of the cohort was 12 years, and they were followed for a median of 2 years following their diagnosis of warts. Within this group, more than half (55%) had hematologic malignancies, while 27% had a history of bone marrow transplantation.
Of note, the authors pointed out, 54% of the patients had plantar warts, and 60% of patients (38 of 63) with a documented number of warts had more than five at the time of presentation.
The treatment regimens that the children had received varied, with 81% of patients receiving cytotoxic chemotherapy and 23% of patients on targeted therapies that included immunotherapy.
The warts were most commonly treated with cryotherapy and topical salicylic acid; this was the case for those actively receiving oncology treatment or those who had completed their treatment regimens.
Outcomes of wart treatments were available in 25 of the patients undergoing active cancer treatment and in 30 of those who had completed treatment. For children on active oncology treatment, 5 (20%) achieved partial resolution, 6 (24%) achieved complete resolution, and 14 (56%) experienced persistence or worsening of their warts following therapy. Those who had completed treatment had better outcomes: Seven (23.3%) had a partial response, 19 (63.3%) had complete resolution, and 4 (13.4%) had persistence or worsening of warts after treatment of warts.
The authors also pointed out the treatment of warts can be painful, expensive, and time-consuming. “It is thus imperative that the risks and benefits of these treatments are carefully considered before proceeding with treatment,” wrote Dr. Ho and colleagues. “This is especially true in medically complex children with cancer who may be fearful of procedures and spend significant portions of their young lives within the medical system.”
Limitations to the study include its retrospective design and small sample size. Clinical data were not uniformly complete, and follow-up intervals varied among the participants. Also, it was conducted at a single-institution and at a large tertiary center, so the results may not be fully generalizable.
The authors declared no conflict of interest. No outside funding source was listed.
FROM PEDIATRIC DERMATOLOGY
Prognostic factors of SCCs in organ transplant recipients worse compared with general population
, results from a dual cohort study demonstrated.
The findings build on previous research and underscore the need for early diagnosis and aggressive surveillance in this patient population, corresponding author Adele C. Green, MBBS, PhD, professor and senior scientist at the QIMR Berghofer Medical Research Institute, Brisbane, Queensland, Australia, and colleagues wrote in the study, which was published online in JAMA Dermatology. “Squamous cell carcinomas (SCCs) of the skin develop up to 77 times more frequently in immunosuppressed organ transplant recipients (OTRs) than the general population,” they wrote. “Because SCCs cause substantially more morbidity and death in the former, they are postulated to be innately more aggressive than in immunocompetent patients, but OTRs’ higher SCC mortality may simply reflect greater SCC tumor burdens per patient.”
In what is believed to be the first study of its kind, Dr. Green and colleagues drew data from two cohort studies to evaluate five key clinicopathologic indicators of poor SCC outcomes in organ transplant recipients, and in those from the general population in Queensland, Australia: cephalic location, perineural invasion, invasion to/beyond subcutaneous fat, poor differentiation, and tumor size greater than 20 mm. The study population included organ transplant recipients at high risk of skin cancer, who were enrolled in the Skin Tumours in Allograft Recipients (STAR) study, and those from a population-based cohort, the QSkin Sun and Health Study. STAR consisted of lung transplant recipients and kidney and liver transplant recipients at high risk of skin cancer who were recruited from tertiary centers and diagnosed with histopathologically confirmed SCC from 2012 to 2015. QSkin consisted of individuals from Queensland’s general adult population diagnosed with SCCs from 2012 to 2015.
SCC cases in QSkin were ascertained through Australia’s universal health insurance agency and linked with histopathology records. Next, the researchers performed data analysis from both cohort studies to determine the prevalence ratio (PR) of head/neck location, perineural invasion, tumor invasion to/beyond subcutaneous fat, poor cellular differentiation, and tumor diameter greater than 20 mm among SCCs among organ transplant recipients compared with the general population.
After combining the two studies, the researchers compared 741 SCCs excised from 191 organ transplant recipients and 2,558 SCCs excised from 1,507 individuals in the general population. Their median ages were similar (62.7 and 63.7 years, respectively) and most were male (78% and 63.4%, respectively).
As for site of involvement, SCCs developed most often on the head and neck in the transplant recipients (38.6%) and on the arms and hands in the general population (35.2%). After adjustment for age and sex, perineural invasion of SCCs was more than twice as common in transplant recipients than among cases in the general population, as was invasion to/beyond subcutaneous fat (PR of 2.37 for both associations).
In other findings, compared with SCCs in the general population, poorly vs. well-differentiated SCCs were more than threefold more common in transplant recipients (PR, 3.45), while the prevalence of tumors greater than 20 mm vs. 20 mm or smaller was moderately higher in transplant recipients (PR, 1.52).
“These findings are considered generalizable, confirming that OTRs’ poorer SCC outcomes are associated with not only their sheer numbers of SCC tumors, but also with a strong shift toward more invasive, less differentiated, and larger SCC tumors, in agreement with previous findings,” the researchers wrote. “This shift is likely associated with decreased immunosurveillance resulting from immunosuppressive therapy (since carcinogenesis decelerates with therapy cessation) interacting with effects of high UV radiation exposure.”
They acknowledged certain limitations of their analysis, chiefly the lack of central review of SCCs to ensure standard assessment of histopathologic features “including caliber of nerves with perineural invasion and cell differentiation; such a review would not have been feasible logistically.”
The study was supported by grants from the National Health and Medical Research Council of Australia. The researchers reported having no disclosures related to the submitted work.
, results from a dual cohort study demonstrated.
The findings build on previous research and underscore the need for early diagnosis and aggressive surveillance in this patient population, corresponding author Adele C. Green, MBBS, PhD, professor and senior scientist at the QIMR Berghofer Medical Research Institute, Brisbane, Queensland, Australia, and colleagues wrote in the study, which was published online in JAMA Dermatology. “Squamous cell carcinomas (SCCs) of the skin develop up to 77 times more frequently in immunosuppressed organ transplant recipients (OTRs) than the general population,” they wrote. “Because SCCs cause substantially more morbidity and death in the former, they are postulated to be innately more aggressive than in immunocompetent patients, but OTRs’ higher SCC mortality may simply reflect greater SCC tumor burdens per patient.”
In what is believed to be the first study of its kind, Dr. Green and colleagues drew data from two cohort studies to evaluate five key clinicopathologic indicators of poor SCC outcomes in organ transplant recipients, and in those from the general population in Queensland, Australia: cephalic location, perineural invasion, invasion to/beyond subcutaneous fat, poor differentiation, and tumor size greater than 20 mm. The study population included organ transplant recipients at high risk of skin cancer, who were enrolled in the Skin Tumours in Allograft Recipients (STAR) study, and those from a population-based cohort, the QSkin Sun and Health Study. STAR consisted of lung transplant recipients and kidney and liver transplant recipients at high risk of skin cancer who were recruited from tertiary centers and diagnosed with histopathologically confirmed SCC from 2012 to 2015. QSkin consisted of individuals from Queensland’s general adult population diagnosed with SCCs from 2012 to 2015.
SCC cases in QSkin were ascertained through Australia’s universal health insurance agency and linked with histopathology records. Next, the researchers performed data analysis from both cohort studies to determine the prevalence ratio (PR) of head/neck location, perineural invasion, tumor invasion to/beyond subcutaneous fat, poor cellular differentiation, and tumor diameter greater than 20 mm among SCCs among organ transplant recipients compared with the general population.
After combining the two studies, the researchers compared 741 SCCs excised from 191 organ transplant recipients and 2,558 SCCs excised from 1,507 individuals in the general population. Their median ages were similar (62.7 and 63.7 years, respectively) and most were male (78% and 63.4%, respectively).
As for site of involvement, SCCs developed most often on the head and neck in the transplant recipients (38.6%) and on the arms and hands in the general population (35.2%). After adjustment for age and sex, perineural invasion of SCCs was more than twice as common in transplant recipients than among cases in the general population, as was invasion to/beyond subcutaneous fat (PR of 2.37 for both associations).
In other findings, compared with SCCs in the general population, poorly vs. well-differentiated SCCs were more than threefold more common in transplant recipients (PR, 3.45), while the prevalence of tumors greater than 20 mm vs. 20 mm or smaller was moderately higher in transplant recipients (PR, 1.52).
“These findings are considered generalizable, confirming that OTRs’ poorer SCC outcomes are associated with not only their sheer numbers of SCC tumors, but also with a strong shift toward more invasive, less differentiated, and larger SCC tumors, in agreement with previous findings,” the researchers wrote. “This shift is likely associated with decreased immunosurveillance resulting from immunosuppressive therapy (since carcinogenesis decelerates with therapy cessation) interacting with effects of high UV radiation exposure.”
They acknowledged certain limitations of their analysis, chiefly the lack of central review of SCCs to ensure standard assessment of histopathologic features “including caliber of nerves with perineural invasion and cell differentiation; such a review would not have been feasible logistically.”
The study was supported by grants from the National Health and Medical Research Council of Australia. The researchers reported having no disclosures related to the submitted work.
, results from a dual cohort study demonstrated.
The findings build on previous research and underscore the need for early diagnosis and aggressive surveillance in this patient population, corresponding author Adele C. Green, MBBS, PhD, professor and senior scientist at the QIMR Berghofer Medical Research Institute, Brisbane, Queensland, Australia, and colleagues wrote in the study, which was published online in JAMA Dermatology. “Squamous cell carcinomas (SCCs) of the skin develop up to 77 times more frequently in immunosuppressed organ transplant recipients (OTRs) than the general population,” they wrote. “Because SCCs cause substantially more morbidity and death in the former, they are postulated to be innately more aggressive than in immunocompetent patients, but OTRs’ higher SCC mortality may simply reflect greater SCC tumor burdens per patient.”
In what is believed to be the first study of its kind, Dr. Green and colleagues drew data from two cohort studies to evaluate five key clinicopathologic indicators of poor SCC outcomes in organ transplant recipients, and in those from the general population in Queensland, Australia: cephalic location, perineural invasion, invasion to/beyond subcutaneous fat, poor differentiation, and tumor size greater than 20 mm. The study population included organ transplant recipients at high risk of skin cancer, who were enrolled in the Skin Tumours in Allograft Recipients (STAR) study, and those from a population-based cohort, the QSkin Sun and Health Study. STAR consisted of lung transplant recipients and kidney and liver transplant recipients at high risk of skin cancer who were recruited from tertiary centers and diagnosed with histopathologically confirmed SCC from 2012 to 2015. QSkin consisted of individuals from Queensland’s general adult population diagnosed with SCCs from 2012 to 2015.
SCC cases in QSkin were ascertained through Australia’s universal health insurance agency and linked with histopathology records. Next, the researchers performed data analysis from both cohort studies to determine the prevalence ratio (PR) of head/neck location, perineural invasion, tumor invasion to/beyond subcutaneous fat, poor cellular differentiation, and tumor diameter greater than 20 mm among SCCs among organ transplant recipients compared with the general population.
After combining the two studies, the researchers compared 741 SCCs excised from 191 organ transplant recipients and 2,558 SCCs excised from 1,507 individuals in the general population. Their median ages were similar (62.7 and 63.7 years, respectively) and most were male (78% and 63.4%, respectively).
As for site of involvement, SCCs developed most often on the head and neck in the transplant recipients (38.6%) and on the arms and hands in the general population (35.2%). After adjustment for age and sex, perineural invasion of SCCs was more than twice as common in transplant recipients than among cases in the general population, as was invasion to/beyond subcutaneous fat (PR of 2.37 for both associations).
In other findings, compared with SCCs in the general population, poorly vs. well-differentiated SCCs were more than threefold more common in transplant recipients (PR, 3.45), while the prevalence of tumors greater than 20 mm vs. 20 mm or smaller was moderately higher in transplant recipients (PR, 1.52).
“These findings are considered generalizable, confirming that OTRs’ poorer SCC outcomes are associated with not only their sheer numbers of SCC tumors, but also with a strong shift toward more invasive, less differentiated, and larger SCC tumors, in agreement with previous findings,” the researchers wrote. “This shift is likely associated with decreased immunosurveillance resulting from immunosuppressive therapy (since carcinogenesis decelerates with therapy cessation) interacting with effects of high UV radiation exposure.”
They acknowledged certain limitations of their analysis, chiefly the lack of central review of SCCs to ensure standard assessment of histopathologic features “including caliber of nerves with perineural invasion and cell differentiation; such a review would not have been feasible logistically.”
The study was supported by grants from the National Health and Medical Research Council of Australia. The researchers reported having no disclosures related to the submitted work.
FROM JAMA DERMATOLOGY
CMML: GM-CSF inhibitor lenzilumab shows early promise
There is currently no international standard of care for patients with CMML, but given its overlap with other myelodysplastic and myeloproliferative syndromes, CMML is usually treated with the hypomethylating agent azacitidine (Vidaza, Onureg), which is associated with objective response rates of 40%-50% and a complete response rate of less than 20%. Alternatively, some patients are treated with the antimetabolite hydroxurea in the palliative setting.
CMML is “insidious, it’s rare, but we think the incidence is increasing because more patients are now getting sequencing done by their doctors, and therapy [related] cases, patients that have survived chemo in the last 10 years, can also develop this disease,” said Daniel Thomas, MD, PhD, from the South Australian Health and Medical Research Institute, Adelaide, in an interview.
Dr. Thomas is a co-investigator of the ongoing phase 2/3 PREACH-M trial, which is testing a novel strategy of treating CMML with mutations in the RAS pathway with a combination of azacitidine and the investigational antibody lenzilumab, which is a targeted inhibitor of granulocyte-macrophage colony-stimulating factor (GM-CSF).
Preliminary results from the trial, reported at the European Hematology Association (EHA) annual meeting, showed that among 10 patients with CMML bearing mutations in the RAS pathway, the combination was associated with durable decreases in monocyte counts, increases in platelet counts and hemoglobin levels, and reductions in both spleen size and C-reactive protein level.
Targeting GM-CSF
More than 90% of cases of CMML carry somatic mutations that are thought to be leukemogenic, with an estimated 46%-60% of cases having mutations in TET2, a tumor suppressor, and an estimated 40% having mutations in KRAS, NRAS, or CBL, all of which are involved in cellular proliferation, and which, research suggests, are sensitive to GM-CSF inhibition.
“I was very surprised that the RAS-mutant arm – so, patients that have KRAS, NRAS, or CBL mutations – are just responding beautifully to [lenzilumab], ” Dr. Thomas said.
“It’s [in the] early days, but if what we’re seeing is durable across the next 10 patients, then I think we’re looking at a game changer,” he added.
Cameron Durrant, MD, DRCOG, MRCGP, chairman and CEO of lenzilumab’s maker Humanigen, said in an interview that the development of the antibody for CMML was spurred in part by research from investigators at the Mayo Clinic, showing that patients with mutations that increased sensitivity to GM-CSF seemed to have better clinical outcomes when the growth factor was blocked.
In addition, Dr. Durrant said, preclinical research from investigators at the Moffitt Cancer Center, Tampa, found that myeloid and monocytic progenitors “fed” on GM-CSF and were sensitive to GM-CSF signal inhibition.
“The biological idea that’s being explored here in the clinic in this study is that by blocking, or starving, if you will, those cells of that food, then you can prevent this overgrowth of certain blood cells that lead to chronic myelomonocytic leukemia,” he said.
PREACH-M details
Lenzilumab is an engineered human immunoglobulin G1-kappa monoclonal antibody with high affinity for human GM-CSF.
In the open label, nonrandomized PREACH-M trial, 72 patients with CMML were enrolled and were assigned to receive 24 monthly cycles of therapy depending on mutational status.
Patients with RAS pathway mutations were assigned to receive azacitidine delivered subcutaneously 75 mg/m2 for 7 days, plus intravenous lenzilumab 552 mg on days 1 and 15 of cycle 1 and on day 1 only of all subsequent cycles.
Patients with TET2 mutations only were assigned to receive azacitidine on the same schedule, plus IV sodium ascorbate 30 g for 7 days, with the first dose 15 g, and subsequent doses 30 g if there is no evidence of tumor lysis syndrome. Following IV administration, patients continue on oral sodium ascorbate 1.1 g on all other days.
The primary endpoint of complete and partial responses any time during the first 12 cycles is planned for reporting at the annual meeting of the American Society of Hematology in December, Dr. Thomas said.
At EHA 2023, the investigators reported available data on 10 patients enrolled in the lenzilumab arm and one enrolled in the azacitidine-sodium ascorbate arm.
Among patients in the lenzilumab arm there was a 5.1-fold decrease in monocyte counts (P = .03) and 2.4-fold decrease in blast counts (P = .04) at 12 months of follow-up.
In addition there was a trend toward increased platelet counts over baseline at 12 months, a significant increase in blood hemoglobin concentration (P = .024), a significant reduction in spleen size (P = .03) and a trend toward lower levels of the inflammatory marker C-reactive protein.
There were 21 grade 3 or 4 adverse events reported, of which 5 were deemed to be possibly related to lenzilumab.
Dr. Thomas told this news organization that the investigators have been “pleasantly surprised” at how well patients tolerated the monoclonal antibody.
“We haven’t had any infusion reactions, we haven’t had any pulmonary alveolar proteinosis, [and] we haven’t had any fevers from the infusion, from the antibody,” he said.
There were some instances of neutropenia and thrombocytopenia that the investigators think may have been related to azacitidine, he noted.
The study is sponsored by the National Health and Medical Research Council of Australia. Dr. Thomas reported no relevant financial relationships. Dr. Durrant is an employee and director of Humanigen.
A version of this article first appeared on Medscape.com.
There is currently no international standard of care for patients with CMML, but given its overlap with other myelodysplastic and myeloproliferative syndromes, CMML is usually treated with the hypomethylating agent azacitidine (Vidaza, Onureg), which is associated with objective response rates of 40%-50% and a complete response rate of less than 20%. Alternatively, some patients are treated with the antimetabolite hydroxurea in the palliative setting.
CMML is “insidious, it’s rare, but we think the incidence is increasing because more patients are now getting sequencing done by their doctors, and therapy [related] cases, patients that have survived chemo in the last 10 years, can also develop this disease,” said Daniel Thomas, MD, PhD, from the South Australian Health and Medical Research Institute, Adelaide, in an interview.
Dr. Thomas is a co-investigator of the ongoing phase 2/3 PREACH-M trial, which is testing a novel strategy of treating CMML with mutations in the RAS pathway with a combination of azacitidine and the investigational antibody lenzilumab, which is a targeted inhibitor of granulocyte-macrophage colony-stimulating factor (GM-CSF).
Preliminary results from the trial, reported at the European Hematology Association (EHA) annual meeting, showed that among 10 patients with CMML bearing mutations in the RAS pathway, the combination was associated with durable decreases in monocyte counts, increases in platelet counts and hemoglobin levels, and reductions in both spleen size and C-reactive protein level.
Targeting GM-CSF
More than 90% of cases of CMML carry somatic mutations that are thought to be leukemogenic, with an estimated 46%-60% of cases having mutations in TET2, a tumor suppressor, and an estimated 40% having mutations in KRAS, NRAS, or CBL, all of which are involved in cellular proliferation, and which, research suggests, are sensitive to GM-CSF inhibition.
“I was very surprised that the RAS-mutant arm – so, patients that have KRAS, NRAS, or CBL mutations – are just responding beautifully to [lenzilumab], ” Dr. Thomas said.
“It’s [in the] early days, but if what we’re seeing is durable across the next 10 patients, then I think we’re looking at a game changer,” he added.
Cameron Durrant, MD, DRCOG, MRCGP, chairman and CEO of lenzilumab’s maker Humanigen, said in an interview that the development of the antibody for CMML was spurred in part by research from investigators at the Mayo Clinic, showing that patients with mutations that increased sensitivity to GM-CSF seemed to have better clinical outcomes when the growth factor was blocked.
In addition, Dr. Durrant said, preclinical research from investigators at the Moffitt Cancer Center, Tampa, found that myeloid and monocytic progenitors “fed” on GM-CSF and were sensitive to GM-CSF signal inhibition.
“The biological idea that’s being explored here in the clinic in this study is that by blocking, or starving, if you will, those cells of that food, then you can prevent this overgrowth of certain blood cells that lead to chronic myelomonocytic leukemia,” he said.
PREACH-M details
Lenzilumab is an engineered human immunoglobulin G1-kappa monoclonal antibody with high affinity for human GM-CSF.
In the open label, nonrandomized PREACH-M trial, 72 patients with CMML were enrolled and were assigned to receive 24 monthly cycles of therapy depending on mutational status.
Patients with RAS pathway mutations were assigned to receive azacitidine delivered subcutaneously 75 mg/m2 for 7 days, plus intravenous lenzilumab 552 mg on days 1 and 15 of cycle 1 and on day 1 only of all subsequent cycles.
Patients with TET2 mutations only were assigned to receive azacitidine on the same schedule, plus IV sodium ascorbate 30 g for 7 days, with the first dose 15 g, and subsequent doses 30 g if there is no evidence of tumor lysis syndrome. Following IV administration, patients continue on oral sodium ascorbate 1.1 g on all other days.
The primary endpoint of complete and partial responses any time during the first 12 cycles is planned for reporting at the annual meeting of the American Society of Hematology in December, Dr. Thomas said.
At EHA 2023, the investigators reported available data on 10 patients enrolled in the lenzilumab arm and one enrolled in the azacitidine-sodium ascorbate arm.
Among patients in the lenzilumab arm there was a 5.1-fold decrease in monocyte counts (P = .03) and 2.4-fold decrease in blast counts (P = .04) at 12 months of follow-up.
In addition there was a trend toward increased platelet counts over baseline at 12 months, a significant increase in blood hemoglobin concentration (P = .024), a significant reduction in spleen size (P = .03) and a trend toward lower levels of the inflammatory marker C-reactive protein.
There were 21 grade 3 or 4 adverse events reported, of which 5 were deemed to be possibly related to lenzilumab.
Dr. Thomas told this news organization that the investigators have been “pleasantly surprised” at how well patients tolerated the monoclonal antibody.
“We haven’t had any infusion reactions, we haven’t had any pulmonary alveolar proteinosis, [and] we haven’t had any fevers from the infusion, from the antibody,” he said.
There were some instances of neutropenia and thrombocytopenia that the investigators think may have been related to azacitidine, he noted.
The study is sponsored by the National Health and Medical Research Council of Australia. Dr. Thomas reported no relevant financial relationships. Dr. Durrant is an employee and director of Humanigen.
A version of this article first appeared on Medscape.com.
There is currently no international standard of care for patients with CMML, but given its overlap with other myelodysplastic and myeloproliferative syndromes, CMML is usually treated with the hypomethylating agent azacitidine (Vidaza, Onureg), which is associated with objective response rates of 40%-50% and a complete response rate of less than 20%. Alternatively, some patients are treated with the antimetabolite hydroxurea in the palliative setting.
CMML is “insidious, it’s rare, but we think the incidence is increasing because more patients are now getting sequencing done by their doctors, and therapy [related] cases, patients that have survived chemo in the last 10 years, can also develop this disease,” said Daniel Thomas, MD, PhD, from the South Australian Health and Medical Research Institute, Adelaide, in an interview.
Dr. Thomas is a co-investigator of the ongoing phase 2/3 PREACH-M trial, which is testing a novel strategy of treating CMML with mutations in the RAS pathway with a combination of azacitidine and the investigational antibody lenzilumab, which is a targeted inhibitor of granulocyte-macrophage colony-stimulating factor (GM-CSF).
Preliminary results from the trial, reported at the European Hematology Association (EHA) annual meeting, showed that among 10 patients with CMML bearing mutations in the RAS pathway, the combination was associated with durable decreases in monocyte counts, increases in platelet counts and hemoglobin levels, and reductions in both spleen size and C-reactive protein level.
Targeting GM-CSF
More than 90% of cases of CMML carry somatic mutations that are thought to be leukemogenic, with an estimated 46%-60% of cases having mutations in TET2, a tumor suppressor, and an estimated 40% having mutations in KRAS, NRAS, or CBL, all of which are involved in cellular proliferation, and which, research suggests, are sensitive to GM-CSF inhibition.
“I was very surprised that the RAS-mutant arm – so, patients that have KRAS, NRAS, or CBL mutations – are just responding beautifully to [lenzilumab], ” Dr. Thomas said.
“It’s [in the] early days, but if what we’re seeing is durable across the next 10 patients, then I think we’re looking at a game changer,” he added.
Cameron Durrant, MD, DRCOG, MRCGP, chairman and CEO of lenzilumab’s maker Humanigen, said in an interview that the development of the antibody for CMML was spurred in part by research from investigators at the Mayo Clinic, showing that patients with mutations that increased sensitivity to GM-CSF seemed to have better clinical outcomes when the growth factor was blocked.
In addition, Dr. Durrant said, preclinical research from investigators at the Moffitt Cancer Center, Tampa, found that myeloid and monocytic progenitors “fed” on GM-CSF and were sensitive to GM-CSF signal inhibition.
“The biological idea that’s being explored here in the clinic in this study is that by blocking, or starving, if you will, those cells of that food, then you can prevent this overgrowth of certain blood cells that lead to chronic myelomonocytic leukemia,” he said.
PREACH-M details
Lenzilumab is an engineered human immunoglobulin G1-kappa monoclonal antibody with high affinity for human GM-CSF.
In the open label, nonrandomized PREACH-M trial, 72 patients with CMML were enrolled and were assigned to receive 24 monthly cycles of therapy depending on mutational status.
Patients with RAS pathway mutations were assigned to receive azacitidine delivered subcutaneously 75 mg/m2 for 7 days, plus intravenous lenzilumab 552 mg on days 1 and 15 of cycle 1 and on day 1 only of all subsequent cycles.
Patients with TET2 mutations only were assigned to receive azacitidine on the same schedule, plus IV sodium ascorbate 30 g for 7 days, with the first dose 15 g, and subsequent doses 30 g if there is no evidence of tumor lysis syndrome. Following IV administration, patients continue on oral sodium ascorbate 1.1 g on all other days.
The primary endpoint of complete and partial responses any time during the first 12 cycles is planned for reporting at the annual meeting of the American Society of Hematology in December, Dr. Thomas said.
At EHA 2023, the investigators reported available data on 10 patients enrolled in the lenzilumab arm and one enrolled in the azacitidine-sodium ascorbate arm.
Among patients in the lenzilumab arm there was a 5.1-fold decrease in monocyte counts (P = .03) and 2.4-fold decrease in blast counts (P = .04) at 12 months of follow-up.
In addition there was a trend toward increased platelet counts over baseline at 12 months, a significant increase in blood hemoglobin concentration (P = .024), a significant reduction in spleen size (P = .03) and a trend toward lower levels of the inflammatory marker C-reactive protein.
There were 21 grade 3 or 4 adverse events reported, of which 5 were deemed to be possibly related to lenzilumab.
Dr. Thomas told this news organization that the investigators have been “pleasantly surprised” at how well patients tolerated the monoclonal antibody.
“We haven’t had any infusion reactions, we haven’t had any pulmonary alveolar proteinosis, [and] we haven’t had any fevers from the infusion, from the antibody,” he said.
There were some instances of neutropenia and thrombocytopenia that the investigators think may have been related to azacitidine, he noted.
The study is sponsored by the National Health and Medical Research Council of Australia. Dr. Thomas reported no relevant financial relationships. Dr. Durrant is an employee and director of Humanigen.
A version of this article first appeared on Medscape.com.
FROM EHA 2023
PV: Novel rusfertide shows ‘impressive’ efficacy
“The results are surprisingly positive,” said senior author Ronald Hoffman, MD, of the Icahn School of Medicine at Mount Sinai, New York, in discussing the late-breaking research at a press briefing during the European Hematology Association Hybrid Congress 2023.
“Importantly, the study met all of its efficacy endpoints, including the proportion of responders, absence of phlebotomy eligibility, and hematocrit control,” Dr. Hoffman said.
PV, a relatively common clonal myeloproliferative neoplasm, is characterized by uncontrolled erythrocytosis, or excessive production of red blood cells, increasing the risk for serious complications such as thromboembolic and cardiovascular events – the most common causes of morbidity and mortality in this blood cancer.
To treat PV, the maintenance of hematocrit levels at below 45% is critical. However, the current standard of care, therapeutic phlebotomy, with or without cytoreductive agents, falls short in maintaining those lower levels in the majority of patients, Dr. Hoffman explained.
To improve responses, rusfertide was developed as a novel, synthetic form of hepcidin, a peptide hormone that is produced by the liver and functions to maintain iron homeostasis and control the formation of red blood cells.
“This is somewhat of a paradigm shift,” said Dr. Hoffman in the press briefing. “We’re trying to use a hormone made by the liver to control excessive red blood cell production from polycythemia vera.”
For the phase 2 REVIVE study evaluating rusfertide in PV, the authors enrolled 53 patients with PV who had a high phlebotomy burden while receiving the current standard of care. The study’s criteria called for patients to have received at least three therapeutic phlebotomies in the 28 weeks prior to enrollment, with or without concurrent cytoreductive agents.
During a first part of the study, patients received subcutaneous rusfertide once weekly over 28 weeks, during which period the dose was adjusted individually to achieve control of HCT levels below 45%.
The second part was a withdrawal phase extending from weeks 29 to 41, in which patients were randomized in a blinded fashion to either continue on rusfertide (n = 26) or receive a placebo (n = 27).
The patients had a median age of 58; they were 71.7% male, and 54.7% had previously been treated with therapeutic phlebotomy alone while 45.3% received therapeutic phlebotomy plus cytoreductive agents.
Patients were considered to be responders if they met three criteria, including having HCT control without phlebotomy eligibility, no therapeutic phlebotomy, and having completed 12 weeks of treatment.
At the end of the second phase, 69.2% of patients receiving rusfertide were responders versus just 18.5% in the placebo group (P = .0003).
Notably, the improvement with rusfertide was observed among those receiving therapeutic phlebotomy alone, as well as with cytoreductive agents (both P = .02).
Compared with placebo, rusfertide provided significant improvement in measures including the maintenance of response, the absence of the need for therapeutic phlebotomy, and persistent HCT control (P < .0001 for all).
Whereas the phlebotomy-free rate with rusfertide during the dose-finding weeks of 1-17 was 76.9% and in weeks 17-29, 87.3%, the rate increased in part 2 of the study to 92.3%.
Additional symptom benefits reported with rusfertide at week 29 versus baseline in part 1 of the study included significant improvements in concentration (P = .0018), itching (P = .0054), fatigue (P = .0074), and inactivity (P = .0005).
In terms of safety, rusfertide was generally well tolerated, with 83% of treatment-emergent adverse events (TEAEs) being grade 1-2, while 17% were grade 3, and none were grade 4 or 5.
The most common TEAEs consisted of injection-site reactions, which were localized, and grade 1-2 in severity. The incidence of reactions decreased with ongoing treatment. There were only two discontinuations resulting from TEAEs.
Among a total of 70 patients who were enrolled, 52 (74.3%) have continued to receive rusfertide for at least 1 year, 32 (45.7%) for at least 1.5 years, and 10 (14.3%) for at least 2 years, indicating the long-term tolerability of rusfertide.
Further commenting, first author Marina Kremyanskaya, MD, PhD, an assistant professor of medicine, hematology, and medical oncology at the Icahn School of Medicine at Mount Sinai, added that a key benefit is rusfertide’s tolerability with combination therapies, which is important in enabling the avoidance of phlebotomies.
“Many patients on cytoreductive therapies still require phlebotomies, and they can’t tolerate a dose increase, either due to cytopenias or other adverse reactions,” she said in an interview. “So adding rusfertide allows for better control of their hematocrits on a lower dose of their respective cytoreductive drug.”
“The combination treatment thus allows for elimination of phlebotomy requirements and potentially improves their symptoms,” Dr. Kremyanskaya said, adding that “using a lower dose of cytoreductive drug such as interferon or hydroxyurea could offer a symptomatic relief to patients as well.”
Overall, she agreed that the responses are remarkably positive.
“I think this is what is so impressive about this agent – basically everybody responds,” Dr. Kremyanskaya said. “When we first started treating patients, we were so impressed, as none of the other drugs we use to treat PV, or any other hematologic malignancy, come anywhere close to this response rate.”
In commenting on the study, Claire Harrison, MD, a professor of myeloproliferative neoplasms and deputy medical director of research at Guy’s and St Thomas’ NHS Foundation Trust in London, agreed that “these data show a strong signal for effectiveness of this therapy in controlling red cell proliferation in PV without inducing iron deficiency and adding to the symptom burden of patients.”
The alternative of phlebotomy “is painful and consumes patient time and hospital resources,” she said in an interview.
Dr. Harrison noted that an earlier signal suggested squamous cell cancer might be of potential concern, but the signal “has not re-emerged [suggesting] this does indeed seem to be a safe and extremely effective therapy.”
Further commenting on the study during the press briefing, Konstanze Döhner, MD, of the University of Ulm (Germany) added that “this is exciting data.”
“For a long time, we had no therapeutic options for PV, and now the field is rapidly developing,” she said.
In ongoing research, rusfertide is currently being studied in the phase 3, placebo-controlled VERIFY randomized trial.
The study was sponsored by Protagonist Therapeutics. Dr. Hoffman reports being on the advisory board for Protagonist Therapeutics, and Dr. Kremyanskaya is a consultant for Protagonist Therapeutics. Dr. Harrison had no disclosures to report.
“The results are surprisingly positive,” said senior author Ronald Hoffman, MD, of the Icahn School of Medicine at Mount Sinai, New York, in discussing the late-breaking research at a press briefing during the European Hematology Association Hybrid Congress 2023.
“Importantly, the study met all of its efficacy endpoints, including the proportion of responders, absence of phlebotomy eligibility, and hematocrit control,” Dr. Hoffman said.
PV, a relatively common clonal myeloproliferative neoplasm, is characterized by uncontrolled erythrocytosis, or excessive production of red blood cells, increasing the risk for serious complications such as thromboembolic and cardiovascular events – the most common causes of morbidity and mortality in this blood cancer.
To treat PV, the maintenance of hematocrit levels at below 45% is critical. However, the current standard of care, therapeutic phlebotomy, with or without cytoreductive agents, falls short in maintaining those lower levels in the majority of patients, Dr. Hoffman explained.
To improve responses, rusfertide was developed as a novel, synthetic form of hepcidin, a peptide hormone that is produced by the liver and functions to maintain iron homeostasis and control the formation of red blood cells.
“This is somewhat of a paradigm shift,” said Dr. Hoffman in the press briefing. “We’re trying to use a hormone made by the liver to control excessive red blood cell production from polycythemia vera.”
For the phase 2 REVIVE study evaluating rusfertide in PV, the authors enrolled 53 patients with PV who had a high phlebotomy burden while receiving the current standard of care. The study’s criteria called for patients to have received at least three therapeutic phlebotomies in the 28 weeks prior to enrollment, with or without concurrent cytoreductive agents.
During a first part of the study, patients received subcutaneous rusfertide once weekly over 28 weeks, during which period the dose was adjusted individually to achieve control of HCT levels below 45%.
The second part was a withdrawal phase extending from weeks 29 to 41, in which patients were randomized in a blinded fashion to either continue on rusfertide (n = 26) or receive a placebo (n = 27).
The patients had a median age of 58; they were 71.7% male, and 54.7% had previously been treated with therapeutic phlebotomy alone while 45.3% received therapeutic phlebotomy plus cytoreductive agents.
Patients were considered to be responders if they met three criteria, including having HCT control without phlebotomy eligibility, no therapeutic phlebotomy, and having completed 12 weeks of treatment.
At the end of the second phase, 69.2% of patients receiving rusfertide were responders versus just 18.5% in the placebo group (P = .0003).
Notably, the improvement with rusfertide was observed among those receiving therapeutic phlebotomy alone, as well as with cytoreductive agents (both P = .02).
Compared with placebo, rusfertide provided significant improvement in measures including the maintenance of response, the absence of the need for therapeutic phlebotomy, and persistent HCT control (P < .0001 for all).
Whereas the phlebotomy-free rate with rusfertide during the dose-finding weeks of 1-17 was 76.9% and in weeks 17-29, 87.3%, the rate increased in part 2 of the study to 92.3%.
Additional symptom benefits reported with rusfertide at week 29 versus baseline in part 1 of the study included significant improvements in concentration (P = .0018), itching (P = .0054), fatigue (P = .0074), and inactivity (P = .0005).
In terms of safety, rusfertide was generally well tolerated, with 83% of treatment-emergent adverse events (TEAEs) being grade 1-2, while 17% were grade 3, and none were grade 4 or 5.
The most common TEAEs consisted of injection-site reactions, which were localized, and grade 1-2 in severity. The incidence of reactions decreased with ongoing treatment. There were only two discontinuations resulting from TEAEs.
Among a total of 70 patients who were enrolled, 52 (74.3%) have continued to receive rusfertide for at least 1 year, 32 (45.7%) for at least 1.5 years, and 10 (14.3%) for at least 2 years, indicating the long-term tolerability of rusfertide.
Further commenting, first author Marina Kremyanskaya, MD, PhD, an assistant professor of medicine, hematology, and medical oncology at the Icahn School of Medicine at Mount Sinai, added that a key benefit is rusfertide’s tolerability with combination therapies, which is important in enabling the avoidance of phlebotomies.
“Many patients on cytoreductive therapies still require phlebotomies, and they can’t tolerate a dose increase, either due to cytopenias or other adverse reactions,” she said in an interview. “So adding rusfertide allows for better control of their hematocrits on a lower dose of their respective cytoreductive drug.”
“The combination treatment thus allows for elimination of phlebotomy requirements and potentially improves their symptoms,” Dr. Kremyanskaya said, adding that “using a lower dose of cytoreductive drug such as interferon or hydroxyurea could offer a symptomatic relief to patients as well.”
Overall, she agreed that the responses are remarkably positive.
“I think this is what is so impressive about this agent – basically everybody responds,” Dr. Kremyanskaya said. “When we first started treating patients, we were so impressed, as none of the other drugs we use to treat PV, or any other hematologic malignancy, come anywhere close to this response rate.”
In commenting on the study, Claire Harrison, MD, a professor of myeloproliferative neoplasms and deputy medical director of research at Guy’s and St Thomas’ NHS Foundation Trust in London, agreed that “these data show a strong signal for effectiveness of this therapy in controlling red cell proliferation in PV without inducing iron deficiency and adding to the symptom burden of patients.”
The alternative of phlebotomy “is painful and consumes patient time and hospital resources,” she said in an interview.
Dr. Harrison noted that an earlier signal suggested squamous cell cancer might be of potential concern, but the signal “has not re-emerged [suggesting] this does indeed seem to be a safe and extremely effective therapy.”
Further commenting on the study during the press briefing, Konstanze Döhner, MD, of the University of Ulm (Germany) added that “this is exciting data.”
“For a long time, we had no therapeutic options for PV, and now the field is rapidly developing,” she said.
In ongoing research, rusfertide is currently being studied in the phase 3, placebo-controlled VERIFY randomized trial.
The study was sponsored by Protagonist Therapeutics. Dr. Hoffman reports being on the advisory board for Protagonist Therapeutics, and Dr. Kremyanskaya is a consultant for Protagonist Therapeutics. Dr. Harrison had no disclosures to report.
“The results are surprisingly positive,” said senior author Ronald Hoffman, MD, of the Icahn School of Medicine at Mount Sinai, New York, in discussing the late-breaking research at a press briefing during the European Hematology Association Hybrid Congress 2023.
“Importantly, the study met all of its efficacy endpoints, including the proportion of responders, absence of phlebotomy eligibility, and hematocrit control,” Dr. Hoffman said.
PV, a relatively common clonal myeloproliferative neoplasm, is characterized by uncontrolled erythrocytosis, or excessive production of red blood cells, increasing the risk for serious complications such as thromboembolic and cardiovascular events – the most common causes of morbidity and mortality in this blood cancer.
To treat PV, the maintenance of hematocrit levels at below 45% is critical. However, the current standard of care, therapeutic phlebotomy, with or without cytoreductive agents, falls short in maintaining those lower levels in the majority of patients, Dr. Hoffman explained.
To improve responses, rusfertide was developed as a novel, synthetic form of hepcidin, a peptide hormone that is produced by the liver and functions to maintain iron homeostasis and control the formation of red blood cells.
“This is somewhat of a paradigm shift,” said Dr. Hoffman in the press briefing. “We’re trying to use a hormone made by the liver to control excessive red blood cell production from polycythemia vera.”
For the phase 2 REVIVE study evaluating rusfertide in PV, the authors enrolled 53 patients with PV who had a high phlebotomy burden while receiving the current standard of care. The study’s criteria called for patients to have received at least three therapeutic phlebotomies in the 28 weeks prior to enrollment, with or without concurrent cytoreductive agents.
During a first part of the study, patients received subcutaneous rusfertide once weekly over 28 weeks, during which period the dose was adjusted individually to achieve control of HCT levels below 45%.
The second part was a withdrawal phase extending from weeks 29 to 41, in which patients were randomized in a blinded fashion to either continue on rusfertide (n = 26) or receive a placebo (n = 27).
The patients had a median age of 58; they were 71.7% male, and 54.7% had previously been treated with therapeutic phlebotomy alone while 45.3% received therapeutic phlebotomy plus cytoreductive agents.
Patients were considered to be responders if they met three criteria, including having HCT control without phlebotomy eligibility, no therapeutic phlebotomy, and having completed 12 weeks of treatment.
At the end of the second phase, 69.2% of patients receiving rusfertide were responders versus just 18.5% in the placebo group (P = .0003).
Notably, the improvement with rusfertide was observed among those receiving therapeutic phlebotomy alone, as well as with cytoreductive agents (both P = .02).
Compared with placebo, rusfertide provided significant improvement in measures including the maintenance of response, the absence of the need for therapeutic phlebotomy, and persistent HCT control (P < .0001 for all).
Whereas the phlebotomy-free rate with rusfertide during the dose-finding weeks of 1-17 was 76.9% and in weeks 17-29, 87.3%, the rate increased in part 2 of the study to 92.3%.
Additional symptom benefits reported with rusfertide at week 29 versus baseline in part 1 of the study included significant improvements in concentration (P = .0018), itching (P = .0054), fatigue (P = .0074), and inactivity (P = .0005).
In terms of safety, rusfertide was generally well tolerated, with 83% of treatment-emergent adverse events (TEAEs) being grade 1-2, while 17% were grade 3, and none were grade 4 or 5.
The most common TEAEs consisted of injection-site reactions, which were localized, and grade 1-2 in severity. The incidence of reactions decreased with ongoing treatment. There were only two discontinuations resulting from TEAEs.
Among a total of 70 patients who were enrolled, 52 (74.3%) have continued to receive rusfertide for at least 1 year, 32 (45.7%) for at least 1.5 years, and 10 (14.3%) for at least 2 years, indicating the long-term tolerability of rusfertide.
Further commenting, first author Marina Kremyanskaya, MD, PhD, an assistant professor of medicine, hematology, and medical oncology at the Icahn School of Medicine at Mount Sinai, added that a key benefit is rusfertide’s tolerability with combination therapies, which is important in enabling the avoidance of phlebotomies.
“Many patients on cytoreductive therapies still require phlebotomies, and they can’t tolerate a dose increase, either due to cytopenias or other adverse reactions,” she said in an interview. “So adding rusfertide allows for better control of their hematocrits on a lower dose of their respective cytoreductive drug.”
“The combination treatment thus allows for elimination of phlebotomy requirements and potentially improves their symptoms,” Dr. Kremyanskaya said, adding that “using a lower dose of cytoreductive drug such as interferon or hydroxyurea could offer a symptomatic relief to patients as well.”
Overall, she agreed that the responses are remarkably positive.
“I think this is what is so impressive about this agent – basically everybody responds,” Dr. Kremyanskaya said. “When we first started treating patients, we were so impressed, as none of the other drugs we use to treat PV, or any other hematologic malignancy, come anywhere close to this response rate.”
In commenting on the study, Claire Harrison, MD, a professor of myeloproliferative neoplasms and deputy medical director of research at Guy’s and St Thomas’ NHS Foundation Trust in London, agreed that “these data show a strong signal for effectiveness of this therapy in controlling red cell proliferation in PV without inducing iron deficiency and adding to the symptom burden of patients.”
The alternative of phlebotomy “is painful and consumes patient time and hospital resources,” she said in an interview.
Dr. Harrison noted that an earlier signal suggested squamous cell cancer might be of potential concern, but the signal “has not re-emerged [suggesting] this does indeed seem to be a safe and extremely effective therapy.”
Further commenting on the study during the press briefing, Konstanze Döhner, MD, of the University of Ulm (Germany) added that “this is exciting data.”
“For a long time, we had no therapeutic options for PV, and now the field is rapidly developing,” she said.
In ongoing research, rusfertide is currently being studied in the phase 3, placebo-controlled VERIFY randomized trial.
The study was sponsored by Protagonist Therapeutics. Dr. Hoffman reports being on the advisory board for Protagonist Therapeutics, and Dr. Kremyanskaya is a consultant for Protagonist Therapeutics. Dr. Harrison had no disclosures to report.
FROM EHA 2023
CLL: Venetoclax-obinutuzumab combo effective long term
Initial results from the trial were shown at the EHA 2019 annual meeting and reported at the time by this news organization.
They revealed that, among more than 430 CLL patients with a median age of over 70 years and multiple comorbidities, the combination of venetoclax, a B-cell lymphoma 2 protein blocker, plus obinutuzumab, an anti-CD20 monoclonal antibody, was associated with a 65% improvement in PFS, compared with chlorambucil, a chemotherapy agent, plus obinutuzumab.
On the strength of these findings, the venetoclax-obinutuzumab combination received Food and Drug Administration approval for previously untreated CLL and small lymphocytic lymphoma in March 2019.
The latest analysis, presented by Othman Al-Sawaf, MD, University Hospital of Cologne (Germany), showed that despite having just 12 cycles of treatment, patients treated with venetoclax-obinutuzumab continued to experience a significant PFS benefit over those given the chemotherapy-based regimen, including in high-risk patients, after more than 6 years of follow-up.
Dr. Al-Sawaf noted that more than 50% of patients given the experimental combination remained without a PFS event at the latest follow-up, and that over 60% had not required a second treatment, equating to a 66% reduction in the likelihood of needing a second treatment versus chlorambucil-obinutuzumab.
Dr. Al-Sawaf said at a press conference that, “clinically, the standard of care for any CLL if it is asymptomatic” is watch and wait, which is “true in the frontline setting, but also in the relapse setting.”
Therefore, these patients “do not need to initiate the next line of treatment, and that’s why time to next treatment is so interesting.”
He added that there also were no new safety signals, with adverse event rates dropping markedly once treatment was over, although there was a suggestion of an increase in second malignancies with venetoclax-obinutuzumab.
“We’ve seen, in many studies now that use fixed-duration approaches, that there is virtually no posttreatment toxicity once patients are able to get off treatment,” Dr. Al-Sawaf said, adding: “This really highlights the benefit” of stopping treatment, “which is a clear advantage compared to having any kind of continuous treatment.”
Approached for comment, William G. Wierda, MD, PhD, professor, department of leukemia, division of cancer medicine, University of Texas MD Anderson Cancer Center, Houston, emphasized the value of the 6-year follow-up of the study, adding that these are “very impressive data.”
He told this news organization that, in terms of the ongoing PFS improvement, “we wouldn’t expect anything otherwise” with venetoclax-obinutuzumab when compared with the chemotherapy-based regimen, but that the trend for an improvement in overall survival is of particular interest.
This “is a notable feature of the update,” Dr. Wierda said, and “we will continue to watch the long-term overall survival curves with a longer follow-up,” especially as the separation of the curves between the two regimens is “more prominent” than in previous analyses of CLL14.
He also pointed to the low incidence of grade ≥ 3 adverse events in patients who are in remission, which “support the use of fixed-duration chemo-free” treatments, and the longer follow-up now allowing the contribution of high-risk features to outcomes to be teased out in multivariate analysis.
“The data that we’re looking for in the next update of this is some indication about improved outcomes between patients with a mutated and unmutated immunoglobulin heavy chain gene [IgHV], in regard to undetectable MRD [minimal residual disease] status,” Dr. Wierda said.
“We know that mutational status correlates with progression free survival,” he explained. “What we would like to see moving forward is how that is associated with undetectable MRD status at the end of treatment.”
Dr. Wierda said that the next hotly anticipated trial in the field is CLL17, which is comparing ibrutinib monotherapy to fixed-duration venetoclax-obinutuzumab to fixed-duration ibrutinib-venetoclax in patients with previously untreated CLL.
“That’s the next question: Is there any advantage of a BTK [Bruton’s tyrosine kinase] inhibitor with venetoclax over venetoclax plus the CD20 antibody?”
Dr. Al-Sawaf, in presenting the latest analysis, reminded the audience that CLL14 was a randomized phase 3 study focusing on patients with previously untreated CLL and coexisting conditions who were randomized to either venetoclax-obinutuzumab for six cycles, followed by six cycles of venetoclax, or chlorambucil-obinutuzumab for six cycles, followed by chlorambucil for six cycles.
The patients, who were enrolled between 2015 and 2016, were required to have a Cumulative Illness Rating Scale (CIRS) score > 6 and/or creatinine clearance < 70 mL/min, which Dr. Al-Sawaf explained serves as “indicator of the unfitness of the patients.”
A total of 432 patients took part in the study. The median age across the two treatment groups was 71-72 years, and the median total CIRS score was 8-9. The majority of patients (79%-80%) had Binet stage B or C CLL. An intermediate tumor lysis syndrome risk was identified in 64%-68%.
“We also had a fair share of patients with high-risk disease,” Dr. Al-Sawaf noted, with approximately 60% having an unmutated IGHV status, and 12% having a TP53 mutation, both of which are associated with a poorer prognosis.
He added that the “aim of these long-term observations that we try to do every year is not so much to do the comparisons to chlorambucil-obinutuzumab, which we appreciate is not necessarily a standard of care anymore,” but rather to understand the safety and effectiveness of venetoclax-obinutuzumab “in the long run, given that all patients are off treatment.”
Beginning with the safety data, Dr. Al-Sawaf showed that rates of grade ≥ 3 adverse events plummeted after the treatment period, with rates of neutropenia falling from 51.9% with venetoclax-obinutuzumab and 47.2% with chlorambucil-obinutuzumab during treatment to 3.8% and 1.9%, respectively, post treatment.
Similarly, rates of thrombocytopenia decreased from 14.2% on treatment to 0.5% off treatment in patients given venetoclax-obinutuzumab, and from 15.0% to 0.0% in the chlorambucil-obinutuzumab group.
One note of caution was sounded over the proportion of patients with at least one second primary malignancy following treatment, which was numerically higher with venetoclax-obinutuzumab, at 14.2% versus 8.4% with the chemotherapy-based regimen.
“But this is a rather a heterogeneous pattern of solid organ tumors and melanoma,” Dr. Al-Sawaf said, referring to the additional malignancies in the venetoclax-obinutuzumab arm. These included lung cancer, prostate cancer and breast cancer.
He said, however, there was no “specific pattern that we can really pinpoint ... and, importantly, the difference is not statistically significant.”
Turning to the efficacy outcomes, Dr. Al-Sawaf showed that, after median follow-up of 76.4 months, the separation in PFS between the two treatment arms continued, with the median PFS 76.2 months with venetoclax-obinutuzumab versus 36.4 months with chlorambucil-obinutuzumab, at a hazard ratio 0.40 (P < .0001).
The 6-year PFS rate in patients treated with venetoclax-obinutuzumab was 53.1% versus 21.7% with the chemotherapy-based regimen. Looking at the high-risk groups, Dr. Al-Sawaf reported that there was a similar pattern of benefit with venetoclax-obinutuzumab.
Among patients with a TP53 mutation, the median PFS was 51.9 months with the combination versus 20.8 months in those given chlorambucil-obinutuzumab, while the corresponding durations in patients with unmutated IGHV were 64.8 months and 26.9 months, respectively.
Multivariate analysis demonstrated that IGHV status was an independent predictor of PFS in patients treated with venetoclax-obinutuzumab, as was the presence of a TP53 mutation, and lymph node size ≥ 5 cm.
There was no significant difference in overall survival between the two treatment groups, although there was a numerical difference in 6-year overall survival rates, at 78.7% with the experimental combination versus 69.2% with chlorambucil-obinutuzumab.
Patients with a minimal residual disease (MRD) count ≥ 10-4 had a shorter overall survival than did those with MRD < 10-4.
“We are currently working up to understand which group of patients experiences these tremendous long term remissions,” Dr. Al-Sawaf said, “and we will keep you posted on this.”
He also showed that the time to next treatment (TTNT), defined as time to death or next anti-leukemic treatment, was significantly longer with venetoclax-obinutuzumab, with the median not reached before the current data lock versus 52.9 months with the chemotherapy-based regimen.
This equated to a hazard ratio in favor of the experimental combination of 0.44 (P < .0001), and a 6-year TTNT rate of 65.2% versus 37.1% for chlorambucil-obinutuzumab.
That second treatment was a Bruton’s tyrosine kinase inhibitor in 59.0% of cases in the venetoclax-obinutuzumab arm and 53.4% in the chlorambucil-obinutuzumab group.
Dr. Al-Sawaf noted, however, that 23.1% and 30.1%, respectively, of patients were given a chemotherapy or chemo-immunotherapy regimen, “which we nowadays would not necessarily consider a standard of care.”
“This ultimately reflects, as in many global clinical studies, the disparities that we still have across the world in terms of access to state-of-the-art therapies.”
The study was sponsored by Hoffmann–La Roche, and conducted in collaboration with AbbVie, and the German CLL Study Group. Dr. Al-Sawaf disclosed relationships with AbbVie, Adaptive, Ascentage, AstraZeneca, BeiGene, Gilead, Janssen, Lilly, and Roche.
Initial results from the trial were shown at the EHA 2019 annual meeting and reported at the time by this news organization.
They revealed that, among more than 430 CLL patients with a median age of over 70 years and multiple comorbidities, the combination of venetoclax, a B-cell lymphoma 2 protein blocker, plus obinutuzumab, an anti-CD20 monoclonal antibody, was associated with a 65% improvement in PFS, compared with chlorambucil, a chemotherapy agent, plus obinutuzumab.
On the strength of these findings, the venetoclax-obinutuzumab combination received Food and Drug Administration approval for previously untreated CLL and small lymphocytic lymphoma in March 2019.
The latest analysis, presented by Othman Al-Sawaf, MD, University Hospital of Cologne (Germany), showed that despite having just 12 cycles of treatment, patients treated with venetoclax-obinutuzumab continued to experience a significant PFS benefit over those given the chemotherapy-based regimen, including in high-risk patients, after more than 6 years of follow-up.
Dr. Al-Sawaf noted that more than 50% of patients given the experimental combination remained without a PFS event at the latest follow-up, and that over 60% had not required a second treatment, equating to a 66% reduction in the likelihood of needing a second treatment versus chlorambucil-obinutuzumab.
Dr. Al-Sawaf said at a press conference that, “clinically, the standard of care for any CLL if it is asymptomatic” is watch and wait, which is “true in the frontline setting, but also in the relapse setting.”
Therefore, these patients “do not need to initiate the next line of treatment, and that’s why time to next treatment is so interesting.”
He added that there also were no new safety signals, with adverse event rates dropping markedly once treatment was over, although there was a suggestion of an increase in second malignancies with venetoclax-obinutuzumab.
“We’ve seen, in many studies now that use fixed-duration approaches, that there is virtually no posttreatment toxicity once patients are able to get off treatment,” Dr. Al-Sawaf said, adding: “This really highlights the benefit” of stopping treatment, “which is a clear advantage compared to having any kind of continuous treatment.”
Approached for comment, William G. Wierda, MD, PhD, professor, department of leukemia, division of cancer medicine, University of Texas MD Anderson Cancer Center, Houston, emphasized the value of the 6-year follow-up of the study, adding that these are “very impressive data.”
He told this news organization that, in terms of the ongoing PFS improvement, “we wouldn’t expect anything otherwise” with venetoclax-obinutuzumab when compared with the chemotherapy-based regimen, but that the trend for an improvement in overall survival is of particular interest.
This “is a notable feature of the update,” Dr. Wierda said, and “we will continue to watch the long-term overall survival curves with a longer follow-up,” especially as the separation of the curves between the two regimens is “more prominent” than in previous analyses of CLL14.
He also pointed to the low incidence of grade ≥ 3 adverse events in patients who are in remission, which “support the use of fixed-duration chemo-free” treatments, and the longer follow-up now allowing the contribution of high-risk features to outcomes to be teased out in multivariate analysis.
“The data that we’re looking for in the next update of this is some indication about improved outcomes between patients with a mutated and unmutated immunoglobulin heavy chain gene [IgHV], in regard to undetectable MRD [minimal residual disease] status,” Dr. Wierda said.
“We know that mutational status correlates with progression free survival,” he explained. “What we would like to see moving forward is how that is associated with undetectable MRD status at the end of treatment.”
Dr. Wierda said that the next hotly anticipated trial in the field is CLL17, which is comparing ibrutinib monotherapy to fixed-duration venetoclax-obinutuzumab to fixed-duration ibrutinib-venetoclax in patients with previously untreated CLL.
“That’s the next question: Is there any advantage of a BTK [Bruton’s tyrosine kinase] inhibitor with venetoclax over venetoclax plus the CD20 antibody?”
Dr. Al-Sawaf, in presenting the latest analysis, reminded the audience that CLL14 was a randomized phase 3 study focusing on patients with previously untreated CLL and coexisting conditions who were randomized to either venetoclax-obinutuzumab for six cycles, followed by six cycles of venetoclax, or chlorambucil-obinutuzumab for six cycles, followed by chlorambucil for six cycles.
The patients, who were enrolled between 2015 and 2016, were required to have a Cumulative Illness Rating Scale (CIRS) score > 6 and/or creatinine clearance < 70 mL/min, which Dr. Al-Sawaf explained serves as “indicator of the unfitness of the patients.”
A total of 432 patients took part in the study. The median age across the two treatment groups was 71-72 years, and the median total CIRS score was 8-9. The majority of patients (79%-80%) had Binet stage B or C CLL. An intermediate tumor lysis syndrome risk was identified in 64%-68%.
“We also had a fair share of patients with high-risk disease,” Dr. Al-Sawaf noted, with approximately 60% having an unmutated IGHV status, and 12% having a TP53 mutation, both of which are associated with a poorer prognosis.
He added that the “aim of these long-term observations that we try to do every year is not so much to do the comparisons to chlorambucil-obinutuzumab, which we appreciate is not necessarily a standard of care anymore,” but rather to understand the safety and effectiveness of venetoclax-obinutuzumab “in the long run, given that all patients are off treatment.”
Beginning with the safety data, Dr. Al-Sawaf showed that rates of grade ≥ 3 adverse events plummeted after the treatment period, with rates of neutropenia falling from 51.9% with venetoclax-obinutuzumab and 47.2% with chlorambucil-obinutuzumab during treatment to 3.8% and 1.9%, respectively, post treatment.
Similarly, rates of thrombocytopenia decreased from 14.2% on treatment to 0.5% off treatment in patients given venetoclax-obinutuzumab, and from 15.0% to 0.0% in the chlorambucil-obinutuzumab group.
One note of caution was sounded over the proportion of patients with at least one second primary malignancy following treatment, which was numerically higher with venetoclax-obinutuzumab, at 14.2% versus 8.4% with the chemotherapy-based regimen.
“But this is a rather a heterogeneous pattern of solid organ tumors and melanoma,” Dr. Al-Sawaf said, referring to the additional malignancies in the venetoclax-obinutuzumab arm. These included lung cancer, prostate cancer and breast cancer.
He said, however, there was no “specific pattern that we can really pinpoint ... and, importantly, the difference is not statistically significant.”
Turning to the efficacy outcomes, Dr. Al-Sawaf showed that, after median follow-up of 76.4 months, the separation in PFS between the two treatment arms continued, with the median PFS 76.2 months with venetoclax-obinutuzumab versus 36.4 months with chlorambucil-obinutuzumab, at a hazard ratio 0.40 (P < .0001).
The 6-year PFS rate in patients treated with venetoclax-obinutuzumab was 53.1% versus 21.7% with the chemotherapy-based regimen. Looking at the high-risk groups, Dr. Al-Sawaf reported that there was a similar pattern of benefit with venetoclax-obinutuzumab.
Among patients with a TP53 mutation, the median PFS was 51.9 months with the combination versus 20.8 months in those given chlorambucil-obinutuzumab, while the corresponding durations in patients with unmutated IGHV were 64.8 months and 26.9 months, respectively.
Multivariate analysis demonstrated that IGHV status was an independent predictor of PFS in patients treated with venetoclax-obinutuzumab, as was the presence of a TP53 mutation, and lymph node size ≥ 5 cm.
There was no significant difference in overall survival between the two treatment groups, although there was a numerical difference in 6-year overall survival rates, at 78.7% with the experimental combination versus 69.2% with chlorambucil-obinutuzumab.
Patients with a minimal residual disease (MRD) count ≥ 10-4 had a shorter overall survival than did those with MRD < 10-4.
“We are currently working up to understand which group of patients experiences these tremendous long term remissions,” Dr. Al-Sawaf said, “and we will keep you posted on this.”
He also showed that the time to next treatment (TTNT), defined as time to death or next anti-leukemic treatment, was significantly longer with venetoclax-obinutuzumab, with the median not reached before the current data lock versus 52.9 months with the chemotherapy-based regimen.
This equated to a hazard ratio in favor of the experimental combination of 0.44 (P < .0001), and a 6-year TTNT rate of 65.2% versus 37.1% for chlorambucil-obinutuzumab.
That second treatment was a Bruton’s tyrosine kinase inhibitor in 59.0% of cases in the venetoclax-obinutuzumab arm and 53.4% in the chlorambucil-obinutuzumab group.
Dr. Al-Sawaf noted, however, that 23.1% and 30.1%, respectively, of patients were given a chemotherapy or chemo-immunotherapy regimen, “which we nowadays would not necessarily consider a standard of care.”
“This ultimately reflects, as in many global clinical studies, the disparities that we still have across the world in terms of access to state-of-the-art therapies.”
The study was sponsored by Hoffmann–La Roche, and conducted in collaboration with AbbVie, and the German CLL Study Group. Dr. Al-Sawaf disclosed relationships with AbbVie, Adaptive, Ascentage, AstraZeneca, BeiGene, Gilead, Janssen, Lilly, and Roche.
Initial results from the trial were shown at the EHA 2019 annual meeting and reported at the time by this news organization.
They revealed that, among more than 430 CLL patients with a median age of over 70 years and multiple comorbidities, the combination of venetoclax, a B-cell lymphoma 2 protein blocker, plus obinutuzumab, an anti-CD20 monoclonal antibody, was associated with a 65% improvement in PFS, compared with chlorambucil, a chemotherapy agent, plus obinutuzumab.
On the strength of these findings, the venetoclax-obinutuzumab combination received Food and Drug Administration approval for previously untreated CLL and small lymphocytic lymphoma in March 2019.
The latest analysis, presented by Othman Al-Sawaf, MD, University Hospital of Cologne (Germany), showed that despite having just 12 cycles of treatment, patients treated with venetoclax-obinutuzumab continued to experience a significant PFS benefit over those given the chemotherapy-based regimen, including in high-risk patients, after more than 6 years of follow-up.
Dr. Al-Sawaf noted that more than 50% of patients given the experimental combination remained without a PFS event at the latest follow-up, and that over 60% had not required a second treatment, equating to a 66% reduction in the likelihood of needing a second treatment versus chlorambucil-obinutuzumab.
Dr. Al-Sawaf said at a press conference that, “clinically, the standard of care for any CLL if it is asymptomatic” is watch and wait, which is “true in the frontline setting, but also in the relapse setting.”
Therefore, these patients “do not need to initiate the next line of treatment, and that’s why time to next treatment is so interesting.”
He added that there also were no new safety signals, with adverse event rates dropping markedly once treatment was over, although there was a suggestion of an increase in second malignancies with venetoclax-obinutuzumab.
“We’ve seen, in many studies now that use fixed-duration approaches, that there is virtually no posttreatment toxicity once patients are able to get off treatment,” Dr. Al-Sawaf said, adding: “This really highlights the benefit” of stopping treatment, “which is a clear advantage compared to having any kind of continuous treatment.”
Approached for comment, William G. Wierda, MD, PhD, professor, department of leukemia, division of cancer medicine, University of Texas MD Anderson Cancer Center, Houston, emphasized the value of the 6-year follow-up of the study, adding that these are “very impressive data.”
He told this news organization that, in terms of the ongoing PFS improvement, “we wouldn’t expect anything otherwise” with venetoclax-obinutuzumab when compared with the chemotherapy-based regimen, but that the trend for an improvement in overall survival is of particular interest.
This “is a notable feature of the update,” Dr. Wierda said, and “we will continue to watch the long-term overall survival curves with a longer follow-up,” especially as the separation of the curves between the two regimens is “more prominent” than in previous analyses of CLL14.
He also pointed to the low incidence of grade ≥ 3 adverse events in patients who are in remission, which “support the use of fixed-duration chemo-free” treatments, and the longer follow-up now allowing the contribution of high-risk features to outcomes to be teased out in multivariate analysis.
“The data that we’re looking for in the next update of this is some indication about improved outcomes between patients with a mutated and unmutated immunoglobulin heavy chain gene [IgHV], in regard to undetectable MRD [minimal residual disease] status,” Dr. Wierda said.
“We know that mutational status correlates with progression free survival,” he explained. “What we would like to see moving forward is how that is associated with undetectable MRD status at the end of treatment.”
Dr. Wierda said that the next hotly anticipated trial in the field is CLL17, which is comparing ibrutinib monotherapy to fixed-duration venetoclax-obinutuzumab to fixed-duration ibrutinib-venetoclax in patients with previously untreated CLL.
“That’s the next question: Is there any advantage of a BTK [Bruton’s tyrosine kinase] inhibitor with venetoclax over venetoclax plus the CD20 antibody?”
Dr. Al-Sawaf, in presenting the latest analysis, reminded the audience that CLL14 was a randomized phase 3 study focusing on patients with previously untreated CLL and coexisting conditions who were randomized to either venetoclax-obinutuzumab for six cycles, followed by six cycles of venetoclax, or chlorambucil-obinutuzumab for six cycles, followed by chlorambucil for six cycles.
The patients, who were enrolled between 2015 and 2016, were required to have a Cumulative Illness Rating Scale (CIRS) score > 6 and/or creatinine clearance < 70 mL/min, which Dr. Al-Sawaf explained serves as “indicator of the unfitness of the patients.”
A total of 432 patients took part in the study. The median age across the two treatment groups was 71-72 years, and the median total CIRS score was 8-9. The majority of patients (79%-80%) had Binet stage B or C CLL. An intermediate tumor lysis syndrome risk was identified in 64%-68%.
“We also had a fair share of patients with high-risk disease,” Dr. Al-Sawaf noted, with approximately 60% having an unmutated IGHV status, and 12% having a TP53 mutation, both of which are associated with a poorer prognosis.
He added that the “aim of these long-term observations that we try to do every year is not so much to do the comparisons to chlorambucil-obinutuzumab, which we appreciate is not necessarily a standard of care anymore,” but rather to understand the safety and effectiveness of venetoclax-obinutuzumab “in the long run, given that all patients are off treatment.”
Beginning with the safety data, Dr. Al-Sawaf showed that rates of grade ≥ 3 adverse events plummeted after the treatment period, with rates of neutropenia falling from 51.9% with venetoclax-obinutuzumab and 47.2% with chlorambucil-obinutuzumab during treatment to 3.8% and 1.9%, respectively, post treatment.
Similarly, rates of thrombocytopenia decreased from 14.2% on treatment to 0.5% off treatment in patients given venetoclax-obinutuzumab, and from 15.0% to 0.0% in the chlorambucil-obinutuzumab group.
One note of caution was sounded over the proportion of patients with at least one second primary malignancy following treatment, which was numerically higher with venetoclax-obinutuzumab, at 14.2% versus 8.4% with the chemotherapy-based regimen.
“But this is a rather a heterogeneous pattern of solid organ tumors and melanoma,” Dr. Al-Sawaf said, referring to the additional malignancies in the venetoclax-obinutuzumab arm. These included lung cancer, prostate cancer and breast cancer.
He said, however, there was no “specific pattern that we can really pinpoint ... and, importantly, the difference is not statistically significant.”
Turning to the efficacy outcomes, Dr. Al-Sawaf showed that, after median follow-up of 76.4 months, the separation in PFS between the two treatment arms continued, with the median PFS 76.2 months with venetoclax-obinutuzumab versus 36.4 months with chlorambucil-obinutuzumab, at a hazard ratio 0.40 (P < .0001).
The 6-year PFS rate in patients treated with venetoclax-obinutuzumab was 53.1% versus 21.7% with the chemotherapy-based regimen. Looking at the high-risk groups, Dr. Al-Sawaf reported that there was a similar pattern of benefit with venetoclax-obinutuzumab.
Among patients with a TP53 mutation, the median PFS was 51.9 months with the combination versus 20.8 months in those given chlorambucil-obinutuzumab, while the corresponding durations in patients with unmutated IGHV were 64.8 months and 26.9 months, respectively.
Multivariate analysis demonstrated that IGHV status was an independent predictor of PFS in patients treated with venetoclax-obinutuzumab, as was the presence of a TP53 mutation, and lymph node size ≥ 5 cm.
There was no significant difference in overall survival between the two treatment groups, although there was a numerical difference in 6-year overall survival rates, at 78.7% with the experimental combination versus 69.2% with chlorambucil-obinutuzumab.
Patients with a minimal residual disease (MRD) count ≥ 10-4 had a shorter overall survival than did those with MRD < 10-4.
“We are currently working up to understand which group of patients experiences these tremendous long term remissions,” Dr. Al-Sawaf said, “and we will keep you posted on this.”
He also showed that the time to next treatment (TTNT), defined as time to death or next anti-leukemic treatment, was significantly longer with venetoclax-obinutuzumab, with the median not reached before the current data lock versus 52.9 months with the chemotherapy-based regimen.
This equated to a hazard ratio in favor of the experimental combination of 0.44 (P < .0001), and a 6-year TTNT rate of 65.2% versus 37.1% for chlorambucil-obinutuzumab.
That second treatment was a Bruton’s tyrosine kinase inhibitor in 59.0% of cases in the venetoclax-obinutuzumab arm and 53.4% in the chlorambucil-obinutuzumab group.
Dr. Al-Sawaf noted, however, that 23.1% and 30.1%, respectively, of patients were given a chemotherapy or chemo-immunotherapy regimen, “which we nowadays would not necessarily consider a standard of care.”
“This ultimately reflects, as in many global clinical studies, the disparities that we still have across the world in terms of access to state-of-the-art therapies.”
The study was sponsored by Hoffmann–La Roche, and conducted in collaboration with AbbVie, and the German CLL Study Group. Dr. Al-Sawaf disclosed relationships with AbbVie, Adaptive, Ascentage, AstraZeneca, BeiGene, Gilead, Janssen, Lilly, and Roche.
FROM EHA 2023
FDA approves glofitamab for DLBCL
The indication is for use in adult patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL) not otherwise specified or with LBCL arising from follicular lymphoma who have received two or more lines of systemic therapy.
The product is a T cell–engaging bispecific antibody developed by Genentech, which has a similar product, mosunetuzumab-axgb (Lunsumio), for the treatment of follicular lymphoma. Lunsumio was approved in December 2022.
These drugs could be considered a first choice in the setting of third-line therapy, suggests an expert writing recently in The New England Journal of Medicine.
Nancy Bartlett, MD, from the Siteman Cancer Center, Washington University in St. Louis, is the author of an editorial that accompanied the publication of results with glofitamab in the pivotal trial that led to its approval.
“Bispecific agents will be an excellent option for the 60% of patients in whom second-line CAR [chimeric antigen receptor] T-cell therapy fails,” she wrote in her editorial.
Dr. Bartlett suggests that these agents may be preferred over CAR T cells. “If longer follow-up confirms that the majority of complete remissions with bispecific agents are durable, on the basis of the advantages of availability (including in the community setting) and more favorable immediate and late toxic-effect profiles, bispecific agents could be considered as the initial choice. ... CAR T-cell therapy could be held in reserve for patients who do not have a complete response or who have a relapse after a complete response.”
Most common form of non-Hodgkin’s lymphoma
DLBCL is the most common form of non-Hodgkin’s lymphoma in the United States, the company noted in a press release. While many people with DLBCL are responsive to treatment, the majority of those who experience relapse or whose condition is refractory to subsequent treatments have poor outcomes.
“Patients with relapsed or refractory diffuse large B-cell lymphoma may experience rapid progression of their cancer and often urgently need an effective treatment option that can be administered without delay,” commented Krish Patel, MD, director of the lymphoma program at the Swedish Cancer Institute in Seattle, who is an investigator on the clinical trial that led to the product’s approval. He said that the results from trials suggest that glofitamab gives patients “a chance for complete remission with a fixed-duration immunotherapy and that such remissions can potentially be sustained after the end of their treatment.”
The accelerated approval is based on response rate and durability of response results from the phase 1/2 NP30179 study.
Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
This trial involved 132 patients with DLBCL who experienced relapse or whose condition was refractory to prior therapies. About one-third of patients (30%) had received prior CAR T-cell therapy. Additionally, for 83% of patients, the condition was refractory to their most recent therapy.
Glofitamab was given to all patients as a fixed course for 8.5 months.
More than half (56%) achieved an overall response, and 43% achieved a complete response. Over two-thirds (68.5%) of those who responded continued to respond for at least 9 months The median duration of response was 1.5 years.
The most common adverse events were cytokine release syndrome (CRS; 70%), which may be serious or life-threatening; musculoskeletal pain (21%); fatigue (20%); and rash (20%). CRS was generally of low grade (52% of patients experienced grade 1 CRS, and 14% experienced grade 2).
Results from the NP30179 trial were published in December 2022.
The complete response rates seen with glofitamab rivals the durable complete response that has been observed with CAR T-cell therapy, Dr. Bartlett noted in the accompanying editorial. “Although these results are promising, it is still too early to estimate the curative potential of glofitamab.”
The indication is for use in adult patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL) not otherwise specified or with LBCL arising from follicular lymphoma who have received two or more lines of systemic therapy.
The product is a T cell–engaging bispecific antibody developed by Genentech, which has a similar product, mosunetuzumab-axgb (Lunsumio), for the treatment of follicular lymphoma. Lunsumio was approved in December 2022.
These drugs could be considered a first choice in the setting of third-line therapy, suggests an expert writing recently in The New England Journal of Medicine.
Nancy Bartlett, MD, from the Siteman Cancer Center, Washington University in St. Louis, is the author of an editorial that accompanied the publication of results with glofitamab in the pivotal trial that led to its approval.
“Bispecific agents will be an excellent option for the 60% of patients in whom second-line CAR [chimeric antigen receptor] T-cell therapy fails,” she wrote in her editorial.
Dr. Bartlett suggests that these agents may be preferred over CAR T cells. “If longer follow-up confirms that the majority of complete remissions with bispecific agents are durable, on the basis of the advantages of availability (including in the community setting) and more favorable immediate and late toxic-effect profiles, bispecific agents could be considered as the initial choice. ... CAR T-cell therapy could be held in reserve for patients who do not have a complete response or who have a relapse after a complete response.”
Most common form of non-Hodgkin’s lymphoma
DLBCL is the most common form of non-Hodgkin’s lymphoma in the United States, the company noted in a press release. While many people with DLBCL are responsive to treatment, the majority of those who experience relapse or whose condition is refractory to subsequent treatments have poor outcomes.
“Patients with relapsed or refractory diffuse large B-cell lymphoma may experience rapid progression of their cancer and often urgently need an effective treatment option that can be administered without delay,” commented Krish Patel, MD, director of the lymphoma program at the Swedish Cancer Institute in Seattle, who is an investigator on the clinical trial that led to the product’s approval. He said that the results from trials suggest that glofitamab gives patients “a chance for complete remission with a fixed-duration immunotherapy and that such remissions can potentially be sustained after the end of their treatment.”
The accelerated approval is based on response rate and durability of response results from the phase 1/2 NP30179 study.
Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
This trial involved 132 patients with DLBCL who experienced relapse or whose condition was refractory to prior therapies. About one-third of patients (30%) had received prior CAR T-cell therapy. Additionally, for 83% of patients, the condition was refractory to their most recent therapy.
Glofitamab was given to all patients as a fixed course for 8.5 months.
More than half (56%) achieved an overall response, and 43% achieved a complete response. Over two-thirds (68.5%) of those who responded continued to respond for at least 9 months The median duration of response was 1.5 years.
The most common adverse events were cytokine release syndrome (CRS; 70%), which may be serious or life-threatening; musculoskeletal pain (21%); fatigue (20%); and rash (20%). CRS was generally of low grade (52% of patients experienced grade 1 CRS, and 14% experienced grade 2).
Results from the NP30179 trial were published in December 2022.
The complete response rates seen with glofitamab rivals the durable complete response that has been observed with CAR T-cell therapy, Dr. Bartlett noted in the accompanying editorial. “Although these results are promising, it is still too early to estimate the curative potential of glofitamab.”
The indication is for use in adult patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL) not otherwise specified or with LBCL arising from follicular lymphoma who have received two or more lines of systemic therapy.
The product is a T cell–engaging bispecific antibody developed by Genentech, which has a similar product, mosunetuzumab-axgb (Lunsumio), for the treatment of follicular lymphoma. Lunsumio was approved in December 2022.
These drugs could be considered a first choice in the setting of third-line therapy, suggests an expert writing recently in The New England Journal of Medicine.
Nancy Bartlett, MD, from the Siteman Cancer Center, Washington University in St. Louis, is the author of an editorial that accompanied the publication of results with glofitamab in the pivotal trial that led to its approval.
“Bispecific agents will be an excellent option for the 60% of patients in whom second-line CAR [chimeric antigen receptor] T-cell therapy fails,” she wrote in her editorial.
Dr. Bartlett suggests that these agents may be preferred over CAR T cells. “If longer follow-up confirms that the majority of complete remissions with bispecific agents are durable, on the basis of the advantages of availability (including in the community setting) and more favorable immediate and late toxic-effect profiles, bispecific agents could be considered as the initial choice. ... CAR T-cell therapy could be held in reserve for patients who do not have a complete response or who have a relapse after a complete response.”
Most common form of non-Hodgkin’s lymphoma
DLBCL is the most common form of non-Hodgkin’s lymphoma in the United States, the company noted in a press release. While many people with DLBCL are responsive to treatment, the majority of those who experience relapse or whose condition is refractory to subsequent treatments have poor outcomes.
“Patients with relapsed or refractory diffuse large B-cell lymphoma may experience rapid progression of their cancer and often urgently need an effective treatment option that can be administered without delay,” commented Krish Patel, MD, director of the lymphoma program at the Swedish Cancer Institute in Seattle, who is an investigator on the clinical trial that led to the product’s approval. He said that the results from trials suggest that glofitamab gives patients “a chance for complete remission with a fixed-duration immunotherapy and that such remissions can potentially be sustained after the end of their treatment.”
The accelerated approval is based on response rate and durability of response results from the phase 1/2 NP30179 study.
Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
This trial involved 132 patients with DLBCL who experienced relapse or whose condition was refractory to prior therapies. About one-third of patients (30%) had received prior CAR T-cell therapy. Additionally, for 83% of patients, the condition was refractory to their most recent therapy.
Glofitamab was given to all patients as a fixed course for 8.5 months.
More than half (56%) achieved an overall response, and 43% achieved a complete response. Over two-thirds (68.5%) of those who responded continued to respond for at least 9 months The median duration of response was 1.5 years.
The most common adverse events were cytokine release syndrome (CRS; 70%), which may be serious or life-threatening; musculoskeletal pain (21%); fatigue (20%); and rash (20%). CRS was generally of low grade (52% of patients experienced grade 1 CRS, and 14% experienced grade 2).
Results from the NP30179 trial were published in December 2022.
The complete response rates seen with glofitamab rivals the durable complete response that has been observed with CAR T-cell therapy, Dr. Bartlett noted in the accompanying editorial. “Although these results are promising, it is still too early to estimate the curative potential of glofitamab.”
‘Professional grief’ is a daily reality for oncologists
– but when it is also accompanied by a sense of emotional isolation, it can lead to reduced well-being and burnout.
The issue was discussed at a special session at the annual meeting of the American Society of Clinical Oncology, and several speakers offered solutions.
Laurie Jean Lyckholm, MD, professor, Hematology/Oncology, West Virginia University School of Medicine, Morgantown, polled the audience to ask how they deal with patient-related loss and grief.
The responses showed that 44.4% said they talk with their colleagues, 16.7% said they talk about it with family and friends, but 22.2% said that they simply move on to the next patient.
Dr. Lyckholm noted that there are positive and negative ways of dealing with grief.
One example of a positive way comes from an oncologist who attended one of her talks and shared with her how his practice deals with the issue.
“At the end of every fourth Friday, he closes his community practice office early and all the oncologists, everyone, stays for a while, and they have a list of the people who have died,” Dr. Lyckholm explained. As a group, they go through the list and reminisce about the patients who died, recalling funny incidents or things that person had said.
“I love this idea,” she said. “The most important thing is to commemorate that person.”
Amplified during pandemic
Like many other issues, the problem of how to deal with “professional grief” was amplified during the COVID-19 pandemic. Many people were unable to see their dying relatives because of the restricted access to sealed-off, dedicated COVID-19 units. One oncologist who had developed a friendly relationship with a patient while treating them for cancer over several years was unable to visit the patient once they were ill with the disease and was left to communicate via an iPad. “It was the only way I could say ‘goodbye’ before she died. ... It still haunts me today, 2 years later,” the clinician recalled.
This anecdote illustrates “disenfranchised grief,” which occurs when an individual experiences a “significant loss and the resultant grief is not openly acknowledged, socially validated, or publicly mourned,” Dr. Lyckholm explained.
If this goes unrecognized, it can lead to shame, guilt, and organizational mistrust, resulting in reduced well-being and clinician burnout, she warned.
The pandemic also had an impact on clinicians directly. Dr. Lyckholm quoted one nurse practitioner who talked about coming back to a new “lonely normal” when returning to a Veterans Affairs hospital.
“I am still getting used to calling colleagues, and paging colleagues, and realizing that they just aren’t there,” the nurse practitioner said. “They aren’t there because they either left or died. I just didn’t expect that.”
Dr. Lyckholm said, “I don’t think we can ever stop acknowledging COVID, because it just had such a terrible impact on all of us.”
Teamwork intervention
The next speaker also polled the audience. Christopher Ryan Friese, PhD, RN, AOCN, Elizabeth Tone Hosmer Professor of Nursing, University of Michigan, Ann Arbor, asked the audience what strategy they would prioritize to reduce burnout, from the perspective of the entire cancer care team.
The response indicated that many (43.6%) would like to see team-based grief and bereavement sessions, while 31.1% thought it best to tackle low-value administrative work.
Dr. Friese drew on a teamwork intervention that researchers at the Dana-Farber Cancer Institute, Boston, with support from the National Cancer Institute, implemented to help identify opportunities to improve cancer care delivery services.
It began with a focus group of nurses who were invited to identify practice pain points, then six 2-hour sessions with all members of the clinical team to identify and develop service expectations and commitments across the various roles.
After these sessions, the researchers saw a decrease in missing orders from 30% to 2%, while patient satisfaction increased from 93% to 97% as a result. Interestingly, there was also a reported rise in efficiency, practice quality and safety, and respectful professional behaviors.
The pilot was then rolled out across the whole institution, and Dr. Friese and colleagues also implemented a version of the program at their community medical oncology practice.
They had a huge response from patients and clinicians alike (with participation rates of 90% and 78%, respectively), and the survey results led to changes in workflow and the standardization of communications.
Importantly for Dr. Friese, the clinicians who took part wanted to repeat the survey to evaluate any practice changes, which was not part of the study protocol and had not been envisaged by the researchers.
So they developed a survey for clinicians, using as an inspiration the Choosing Wisely campaign by the American Board of Internal Medicine Foundation to identify the best treatments to improve patient outcomes and those to deprioritize.
They used the survey on 373 clinicians at the University of Michigan Health System and found that “the number one thing was getting rid of the administrative work” – that it doesn’t have to be done specifically by physicians or other providers and that other people can do it.
The second was time-consuming electronic health record tasks.
Both of these have since been the focus of an elimination and reduction process to give clinicians more time to do what matters most to them and their patients.
“We have the opportunity to do this in a different way,” Dr. Friese said, “and I think it’s a really powerful opportunity.”
“We can retrofit the solution, which is the pizza parties, and the yoga apps, and the T-shirts ... [or] we could actually redesign the work that we’re asking clinicians to do on a daily basis,” he commented.
“We could make the work easier to do so that you have more time with patients and less time with administrative work and have more time to process grief or to celebrate successes,” he concluded.
Tackling burnout
The final speaker, Vicki A. Jackson, MD, MPH, chief of palliative care, Massachusetts General Hospital, emphasized that the recognition of grief by a cancer care provider is “imperative” for physician well-being and pointed out that that interventions to help “do exist,” including ASCO’s SafeHaven collection of physician well-being resources.
Oncology inherently carries with it “threats” to well-being, including uncertainty and doubt, isolation, fears over one’s usefulness, exhaustion, the witnessing of suffering, and moral distress, she noted.
Things that are necessary for well-being, in contrast, include a sense of connection and community, having boundaries between work and personal life, self-awareness, compassion, and empowerment, among others.
Dr. Jackson believes that in the current era community building within oncology must be “intentional” and not just based around “water cooler moments,” as the sense of isolation experienced by clinicians is “not fluff; this is critical.”
Initiatives such as virtual happy hours and game nights may be helpful, she suggested.
A colleague of hers likes to send out the dad joke of the day, “which made everybody groan, but let me tell you, it changed the affective tone before they started seeing all these really hard, sad patients.”
Setting boundaries, which was the topic of another session at ASCO 2023, is also an important way to address the “emotionally powerful” work of oncology, Dr. Jackson commented.
She underlined the need to channel or be “fully present when you are in the room” but emphasized the need to detach at the end of the day, commenting that “when you leave, you leave.”
No funding was declared. Dr. Friese reported relationships with Merck, NCCN/Pfizer, National Cancer Institute, Patient-Centered Outcomes Research Institute, and the Simms/Mann Foundation. No other speakers reported relevant financial relationships.
A version of this article first appeared on Medscape.com.
– but when it is also accompanied by a sense of emotional isolation, it can lead to reduced well-being and burnout.
The issue was discussed at a special session at the annual meeting of the American Society of Clinical Oncology, and several speakers offered solutions.
Laurie Jean Lyckholm, MD, professor, Hematology/Oncology, West Virginia University School of Medicine, Morgantown, polled the audience to ask how they deal with patient-related loss and grief.
The responses showed that 44.4% said they talk with their colleagues, 16.7% said they talk about it with family and friends, but 22.2% said that they simply move on to the next patient.
Dr. Lyckholm noted that there are positive and negative ways of dealing with grief.
One example of a positive way comes from an oncologist who attended one of her talks and shared with her how his practice deals with the issue.
“At the end of every fourth Friday, he closes his community practice office early and all the oncologists, everyone, stays for a while, and they have a list of the people who have died,” Dr. Lyckholm explained. As a group, they go through the list and reminisce about the patients who died, recalling funny incidents or things that person had said.
“I love this idea,” she said. “The most important thing is to commemorate that person.”
Amplified during pandemic
Like many other issues, the problem of how to deal with “professional grief” was amplified during the COVID-19 pandemic. Many people were unable to see their dying relatives because of the restricted access to sealed-off, dedicated COVID-19 units. One oncologist who had developed a friendly relationship with a patient while treating them for cancer over several years was unable to visit the patient once they were ill with the disease and was left to communicate via an iPad. “It was the only way I could say ‘goodbye’ before she died. ... It still haunts me today, 2 years later,” the clinician recalled.
This anecdote illustrates “disenfranchised grief,” which occurs when an individual experiences a “significant loss and the resultant grief is not openly acknowledged, socially validated, or publicly mourned,” Dr. Lyckholm explained.
If this goes unrecognized, it can lead to shame, guilt, and organizational mistrust, resulting in reduced well-being and clinician burnout, she warned.
The pandemic also had an impact on clinicians directly. Dr. Lyckholm quoted one nurse practitioner who talked about coming back to a new “lonely normal” when returning to a Veterans Affairs hospital.
“I am still getting used to calling colleagues, and paging colleagues, and realizing that they just aren’t there,” the nurse practitioner said. “They aren’t there because they either left or died. I just didn’t expect that.”
Dr. Lyckholm said, “I don’t think we can ever stop acknowledging COVID, because it just had such a terrible impact on all of us.”
Teamwork intervention
The next speaker also polled the audience. Christopher Ryan Friese, PhD, RN, AOCN, Elizabeth Tone Hosmer Professor of Nursing, University of Michigan, Ann Arbor, asked the audience what strategy they would prioritize to reduce burnout, from the perspective of the entire cancer care team.
The response indicated that many (43.6%) would like to see team-based grief and bereavement sessions, while 31.1% thought it best to tackle low-value administrative work.
Dr. Friese drew on a teamwork intervention that researchers at the Dana-Farber Cancer Institute, Boston, with support from the National Cancer Institute, implemented to help identify opportunities to improve cancer care delivery services.
It began with a focus group of nurses who were invited to identify practice pain points, then six 2-hour sessions with all members of the clinical team to identify and develop service expectations and commitments across the various roles.
After these sessions, the researchers saw a decrease in missing orders from 30% to 2%, while patient satisfaction increased from 93% to 97% as a result. Interestingly, there was also a reported rise in efficiency, practice quality and safety, and respectful professional behaviors.
The pilot was then rolled out across the whole institution, and Dr. Friese and colleagues also implemented a version of the program at their community medical oncology practice.
They had a huge response from patients and clinicians alike (with participation rates of 90% and 78%, respectively), and the survey results led to changes in workflow and the standardization of communications.
Importantly for Dr. Friese, the clinicians who took part wanted to repeat the survey to evaluate any practice changes, which was not part of the study protocol and had not been envisaged by the researchers.
So they developed a survey for clinicians, using as an inspiration the Choosing Wisely campaign by the American Board of Internal Medicine Foundation to identify the best treatments to improve patient outcomes and those to deprioritize.
They used the survey on 373 clinicians at the University of Michigan Health System and found that “the number one thing was getting rid of the administrative work” – that it doesn’t have to be done specifically by physicians or other providers and that other people can do it.
The second was time-consuming electronic health record tasks.
Both of these have since been the focus of an elimination and reduction process to give clinicians more time to do what matters most to them and their patients.
“We have the opportunity to do this in a different way,” Dr. Friese said, “and I think it’s a really powerful opportunity.”
“We can retrofit the solution, which is the pizza parties, and the yoga apps, and the T-shirts ... [or] we could actually redesign the work that we’re asking clinicians to do on a daily basis,” he commented.
“We could make the work easier to do so that you have more time with patients and less time with administrative work and have more time to process grief or to celebrate successes,” he concluded.
Tackling burnout
The final speaker, Vicki A. Jackson, MD, MPH, chief of palliative care, Massachusetts General Hospital, emphasized that the recognition of grief by a cancer care provider is “imperative” for physician well-being and pointed out that that interventions to help “do exist,” including ASCO’s SafeHaven collection of physician well-being resources.
Oncology inherently carries with it “threats” to well-being, including uncertainty and doubt, isolation, fears over one’s usefulness, exhaustion, the witnessing of suffering, and moral distress, she noted.
Things that are necessary for well-being, in contrast, include a sense of connection and community, having boundaries between work and personal life, self-awareness, compassion, and empowerment, among others.
Dr. Jackson believes that in the current era community building within oncology must be “intentional” and not just based around “water cooler moments,” as the sense of isolation experienced by clinicians is “not fluff; this is critical.”
Initiatives such as virtual happy hours and game nights may be helpful, she suggested.
A colleague of hers likes to send out the dad joke of the day, “which made everybody groan, but let me tell you, it changed the affective tone before they started seeing all these really hard, sad patients.”
Setting boundaries, which was the topic of another session at ASCO 2023, is also an important way to address the “emotionally powerful” work of oncology, Dr. Jackson commented.
She underlined the need to channel or be “fully present when you are in the room” but emphasized the need to detach at the end of the day, commenting that “when you leave, you leave.”
No funding was declared. Dr. Friese reported relationships with Merck, NCCN/Pfizer, National Cancer Institute, Patient-Centered Outcomes Research Institute, and the Simms/Mann Foundation. No other speakers reported relevant financial relationships.
A version of this article first appeared on Medscape.com.
– but when it is also accompanied by a sense of emotional isolation, it can lead to reduced well-being and burnout.
The issue was discussed at a special session at the annual meeting of the American Society of Clinical Oncology, and several speakers offered solutions.
Laurie Jean Lyckholm, MD, professor, Hematology/Oncology, West Virginia University School of Medicine, Morgantown, polled the audience to ask how they deal with patient-related loss and grief.
The responses showed that 44.4% said they talk with their colleagues, 16.7% said they talk about it with family and friends, but 22.2% said that they simply move on to the next patient.
Dr. Lyckholm noted that there are positive and negative ways of dealing with grief.
One example of a positive way comes from an oncologist who attended one of her talks and shared with her how his practice deals with the issue.
“At the end of every fourth Friday, he closes his community practice office early and all the oncologists, everyone, stays for a while, and they have a list of the people who have died,” Dr. Lyckholm explained. As a group, they go through the list and reminisce about the patients who died, recalling funny incidents or things that person had said.
“I love this idea,” she said. “The most important thing is to commemorate that person.”
Amplified during pandemic
Like many other issues, the problem of how to deal with “professional grief” was amplified during the COVID-19 pandemic. Many people were unable to see their dying relatives because of the restricted access to sealed-off, dedicated COVID-19 units. One oncologist who had developed a friendly relationship with a patient while treating them for cancer over several years was unable to visit the patient once they were ill with the disease and was left to communicate via an iPad. “It was the only way I could say ‘goodbye’ before she died. ... It still haunts me today, 2 years later,” the clinician recalled.
This anecdote illustrates “disenfranchised grief,” which occurs when an individual experiences a “significant loss and the resultant grief is not openly acknowledged, socially validated, or publicly mourned,” Dr. Lyckholm explained.
If this goes unrecognized, it can lead to shame, guilt, and organizational mistrust, resulting in reduced well-being and clinician burnout, she warned.
The pandemic also had an impact on clinicians directly. Dr. Lyckholm quoted one nurse practitioner who talked about coming back to a new “lonely normal” when returning to a Veterans Affairs hospital.
“I am still getting used to calling colleagues, and paging colleagues, and realizing that they just aren’t there,” the nurse practitioner said. “They aren’t there because they either left or died. I just didn’t expect that.”
Dr. Lyckholm said, “I don’t think we can ever stop acknowledging COVID, because it just had such a terrible impact on all of us.”
Teamwork intervention
The next speaker also polled the audience. Christopher Ryan Friese, PhD, RN, AOCN, Elizabeth Tone Hosmer Professor of Nursing, University of Michigan, Ann Arbor, asked the audience what strategy they would prioritize to reduce burnout, from the perspective of the entire cancer care team.
The response indicated that many (43.6%) would like to see team-based grief and bereavement sessions, while 31.1% thought it best to tackle low-value administrative work.
Dr. Friese drew on a teamwork intervention that researchers at the Dana-Farber Cancer Institute, Boston, with support from the National Cancer Institute, implemented to help identify opportunities to improve cancer care delivery services.
It began with a focus group of nurses who were invited to identify practice pain points, then six 2-hour sessions with all members of the clinical team to identify and develop service expectations and commitments across the various roles.
After these sessions, the researchers saw a decrease in missing orders from 30% to 2%, while patient satisfaction increased from 93% to 97% as a result. Interestingly, there was also a reported rise in efficiency, practice quality and safety, and respectful professional behaviors.
The pilot was then rolled out across the whole institution, and Dr. Friese and colleagues also implemented a version of the program at their community medical oncology practice.
They had a huge response from patients and clinicians alike (with participation rates of 90% and 78%, respectively), and the survey results led to changes in workflow and the standardization of communications.
Importantly for Dr. Friese, the clinicians who took part wanted to repeat the survey to evaluate any practice changes, which was not part of the study protocol and had not been envisaged by the researchers.
So they developed a survey for clinicians, using as an inspiration the Choosing Wisely campaign by the American Board of Internal Medicine Foundation to identify the best treatments to improve patient outcomes and those to deprioritize.
They used the survey on 373 clinicians at the University of Michigan Health System and found that “the number one thing was getting rid of the administrative work” – that it doesn’t have to be done specifically by physicians or other providers and that other people can do it.
The second was time-consuming electronic health record tasks.
Both of these have since been the focus of an elimination and reduction process to give clinicians more time to do what matters most to them and their patients.
“We have the opportunity to do this in a different way,” Dr. Friese said, “and I think it’s a really powerful opportunity.”
“We can retrofit the solution, which is the pizza parties, and the yoga apps, and the T-shirts ... [or] we could actually redesign the work that we’re asking clinicians to do on a daily basis,” he commented.
“We could make the work easier to do so that you have more time with patients and less time with administrative work and have more time to process grief or to celebrate successes,” he concluded.
Tackling burnout
The final speaker, Vicki A. Jackson, MD, MPH, chief of palliative care, Massachusetts General Hospital, emphasized that the recognition of grief by a cancer care provider is “imperative” for physician well-being and pointed out that that interventions to help “do exist,” including ASCO’s SafeHaven collection of physician well-being resources.
Oncology inherently carries with it “threats” to well-being, including uncertainty and doubt, isolation, fears over one’s usefulness, exhaustion, the witnessing of suffering, and moral distress, she noted.
Things that are necessary for well-being, in contrast, include a sense of connection and community, having boundaries between work and personal life, self-awareness, compassion, and empowerment, among others.
Dr. Jackson believes that in the current era community building within oncology must be “intentional” and not just based around “water cooler moments,” as the sense of isolation experienced by clinicians is “not fluff; this is critical.”
Initiatives such as virtual happy hours and game nights may be helpful, she suggested.
A colleague of hers likes to send out the dad joke of the day, “which made everybody groan, but let me tell you, it changed the affective tone before they started seeing all these really hard, sad patients.”
Setting boundaries, which was the topic of another session at ASCO 2023, is also an important way to address the “emotionally powerful” work of oncology, Dr. Jackson commented.
She underlined the need to channel or be “fully present when you are in the room” but emphasized the need to detach at the end of the day, commenting that “when you leave, you leave.”
No funding was declared. Dr. Friese reported relationships with Merck, NCCN/Pfizer, National Cancer Institute, Patient-Centered Outcomes Research Institute, and the Simms/Mann Foundation. No other speakers reported relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ASCO 2023
ASCO 2023: Promising results in breast cancer from NATALEE and PHERGain
This transcript has been edited for clarity.
Hi. It’s Dr. Kathy Miller from Indiana University, coming to you today from the 2023 ASCO annual meeting in Chicago.
It’s been an exciting year for breast cancer news. I want to make sure that you hear about the two studies that I find the most impactful. One is the NATALEE study looking at ribociclib in adjuvant ER-positive patients at high risk for recurrence. You saw the press release a few weeks ago, and we now have the data. There is no doubt that this is a positive trial.
The details here are important. These were pre- or postmenopausal women, and men as well. Premenopausal women and men also had an LHRH agonist in addition to an aromatase inhibitor – that could have been either letrozole or anastrozole – then randomized to ribociclib or placebo.
The dose of ribociclib that you’re used to thinking about is 600 mg daily for 3 weeks and 7 days off. That’s the approved dose in the metastatic setting. In the adjuvant trial, they used 400 mg, and that was intentional to try to reduce some of the toxicity because the plan was for 3 years of therapy. Managing toxicity and really making this tolerable for patients was crucial.
We’ve now seen the efficacy results, with a roughly 3% reduction in the risk for recurrence; 90% disease-free survival in the ribociclib arm, 87% in the control arm, some patients still having prolongation of QTc but no serious arrhythmias; some patients still with myelosuppression, but risk for serious infections was really very low.
We have no head-to-head trials in any setting, and I doubt that our industry colleagues are going to be interested in a head-to-head setting.
We’re going to need to pay particular attention to long-term follow-up and to quality of life and toxicity data as to which our patients prefer. We may need to think about other ways of doing those direct comparisons with public funding, where we can get the answers our patients deserve.
I also want to think about the other end of the spectrum, those patients with HER2-positive disease. We saw fantastic results from the PHERGain study from our colleagues in Spain. This was a trial that took patients with predominantly stage II and III HER2-positive breast cancer. These are patients that we would treat with neoadjuvant chemotherapy, with dual HER2-targeted therapy.
Years ago, we saw results of some small, single-arm, phase 2 studies, suggesting that some of those patients may be so sensitive to biologic therapy that they have a pathologic complete response with HER2-targeted therapy – HER2-targeted therapy with endocrine therapy if they are positive – with no chemotherapy at all. Our question has always been how to identify those patients. Can we identify them well enough that we would be comfortable not treating them with chemotherapy? Importantly, If they didn’t get chemotherapy, what’s their long-term outcome?
The PHERGain trial lets us look at all those things. The PHERGain trial gave patients two cycles of dual HER2-targeted therapy, pertuzumab and trastuzumab, hormone therapy if also ER positive, and they got an FDG-PET scan after two cycles of therapy.
If they had a significant PET response, those patients were then randomized to switch to chemotherapy, standard TCHP, or continue biologic therapy alone for a total of six cycles. They then went to surgery. If they had a pathologic complete response, whether they had gotten chemotherapy or no chemotherapy, they completed the HER2-targeted therapy. If they still had residual disease, they got chemotherapy if chemotherapy had not been administered before, and they may have gotten other HER2-targeted therapies if they had already received chemotherapy.
There were over 300 patients in this trial, and my memory is that roughly two thirds of them had a PET response. About 86 patients randomized to continue biologic therapy had a pathologic complete response, so about one-third of those for whom the PET imaging said they were responding with biologic therapy only had a pathologic complete response.
They have now been followed for 3 years. The 3-year disease-free survival results look very reassuring. Of those 86 patients, one patient had a local recurrence and no patient had a distant recurrence.
This is what we’ve been waiting for. Can we identify those patients who have an excellent prognosis with biologic therapy alone so that we can avoid the toxicities? This is really where you’ll see the research over the coming years in breast cancer, looking at additional therapies in high-risk patients who don’t do so well with our standard therapies, and better stratification of patients who do so well with our standard therapies that we may be able to do less.
This is one of the ways that we’ll be able to do that. I look forward to sharing those results with you over coming years.
Kathy D. Miller, MD, is associate director of clinical research and codirector of the breast cancer program at the Melvin and Bren Simon Cancer Center at Indiana University, Indianapolis. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. It’s Dr. Kathy Miller from Indiana University, coming to you today from the 2023 ASCO annual meeting in Chicago.
It’s been an exciting year for breast cancer news. I want to make sure that you hear about the two studies that I find the most impactful. One is the NATALEE study looking at ribociclib in adjuvant ER-positive patients at high risk for recurrence. You saw the press release a few weeks ago, and we now have the data. There is no doubt that this is a positive trial.
The details here are important. These were pre- or postmenopausal women, and men as well. Premenopausal women and men also had an LHRH agonist in addition to an aromatase inhibitor – that could have been either letrozole or anastrozole – then randomized to ribociclib or placebo.
The dose of ribociclib that you’re used to thinking about is 600 mg daily for 3 weeks and 7 days off. That’s the approved dose in the metastatic setting. In the adjuvant trial, they used 400 mg, and that was intentional to try to reduce some of the toxicity because the plan was for 3 years of therapy. Managing toxicity and really making this tolerable for patients was crucial.
We’ve now seen the efficacy results, with a roughly 3% reduction in the risk for recurrence; 90% disease-free survival in the ribociclib arm, 87% in the control arm, some patients still having prolongation of QTc but no serious arrhythmias; some patients still with myelosuppression, but risk for serious infections was really very low.
We have no head-to-head trials in any setting, and I doubt that our industry colleagues are going to be interested in a head-to-head setting.
We’re going to need to pay particular attention to long-term follow-up and to quality of life and toxicity data as to which our patients prefer. We may need to think about other ways of doing those direct comparisons with public funding, where we can get the answers our patients deserve.
I also want to think about the other end of the spectrum, those patients with HER2-positive disease. We saw fantastic results from the PHERGain study from our colleagues in Spain. This was a trial that took patients with predominantly stage II and III HER2-positive breast cancer. These are patients that we would treat with neoadjuvant chemotherapy, with dual HER2-targeted therapy.
Years ago, we saw results of some small, single-arm, phase 2 studies, suggesting that some of those patients may be so sensitive to biologic therapy that they have a pathologic complete response with HER2-targeted therapy – HER2-targeted therapy with endocrine therapy if they are positive – with no chemotherapy at all. Our question has always been how to identify those patients. Can we identify them well enough that we would be comfortable not treating them with chemotherapy? Importantly, If they didn’t get chemotherapy, what’s their long-term outcome?
The PHERGain trial lets us look at all those things. The PHERGain trial gave patients two cycles of dual HER2-targeted therapy, pertuzumab and trastuzumab, hormone therapy if also ER positive, and they got an FDG-PET scan after two cycles of therapy.
If they had a significant PET response, those patients were then randomized to switch to chemotherapy, standard TCHP, or continue biologic therapy alone for a total of six cycles. They then went to surgery. If they had a pathologic complete response, whether they had gotten chemotherapy or no chemotherapy, they completed the HER2-targeted therapy. If they still had residual disease, they got chemotherapy if chemotherapy had not been administered before, and they may have gotten other HER2-targeted therapies if they had already received chemotherapy.
There were over 300 patients in this trial, and my memory is that roughly two thirds of them had a PET response. About 86 patients randomized to continue biologic therapy had a pathologic complete response, so about one-third of those for whom the PET imaging said they were responding with biologic therapy only had a pathologic complete response.
They have now been followed for 3 years. The 3-year disease-free survival results look very reassuring. Of those 86 patients, one patient had a local recurrence and no patient had a distant recurrence.
This is what we’ve been waiting for. Can we identify those patients who have an excellent prognosis with biologic therapy alone so that we can avoid the toxicities? This is really where you’ll see the research over the coming years in breast cancer, looking at additional therapies in high-risk patients who don’t do so well with our standard therapies, and better stratification of patients who do so well with our standard therapies that we may be able to do less.
This is one of the ways that we’ll be able to do that. I look forward to sharing those results with you over coming years.
Kathy D. Miller, MD, is associate director of clinical research and codirector of the breast cancer program at the Melvin and Bren Simon Cancer Center at Indiana University, Indianapolis. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. It’s Dr. Kathy Miller from Indiana University, coming to you today from the 2023 ASCO annual meeting in Chicago.
It’s been an exciting year for breast cancer news. I want to make sure that you hear about the two studies that I find the most impactful. One is the NATALEE study looking at ribociclib in adjuvant ER-positive patients at high risk for recurrence. You saw the press release a few weeks ago, and we now have the data. There is no doubt that this is a positive trial.
The details here are important. These were pre- or postmenopausal women, and men as well. Premenopausal women and men also had an LHRH agonist in addition to an aromatase inhibitor – that could have been either letrozole or anastrozole – then randomized to ribociclib or placebo.
The dose of ribociclib that you’re used to thinking about is 600 mg daily for 3 weeks and 7 days off. That’s the approved dose in the metastatic setting. In the adjuvant trial, they used 400 mg, and that was intentional to try to reduce some of the toxicity because the plan was for 3 years of therapy. Managing toxicity and really making this tolerable for patients was crucial.
We’ve now seen the efficacy results, with a roughly 3% reduction in the risk for recurrence; 90% disease-free survival in the ribociclib arm, 87% in the control arm, some patients still having prolongation of QTc but no serious arrhythmias; some patients still with myelosuppression, but risk for serious infections was really very low.
We have no head-to-head trials in any setting, and I doubt that our industry colleagues are going to be interested in a head-to-head setting.
We’re going to need to pay particular attention to long-term follow-up and to quality of life and toxicity data as to which our patients prefer. We may need to think about other ways of doing those direct comparisons with public funding, where we can get the answers our patients deserve.
I also want to think about the other end of the spectrum, those patients with HER2-positive disease. We saw fantastic results from the PHERGain study from our colleagues in Spain. This was a trial that took patients with predominantly stage II and III HER2-positive breast cancer. These are patients that we would treat with neoadjuvant chemotherapy, with dual HER2-targeted therapy.
Years ago, we saw results of some small, single-arm, phase 2 studies, suggesting that some of those patients may be so sensitive to biologic therapy that they have a pathologic complete response with HER2-targeted therapy – HER2-targeted therapy with endocrine therapy if they are positive – with no chemotherapy at all. Our question has always been how to identify those patients. Can we identify them well enough that we would be comfortable not treating them with chemotherapy? Importantly, If they didn’t get chemotherapy, what’s their long-term outcome?
The PHERGain trial lets us look at all those things. The PHERGain trial gave patients two cycles of dual HER2-targeted therapy, pertuzumab and trastuzumab, hormone therapy if also ER positive, and they got an FDG-PET scan after two cycles of therapy.
If they had a significant PET response, those patients were then randomized to switch to chemotherapy, standard TCHP, or continue biologic therapy alone for a total of six cycles. They then went to surgery. If they had a pathologic complete response, whether they had gotten chemotherapy or no chemotherapy, they completed the HER2-targeted therapy. If they still had residual disease, they got chemotherapy if chemotherapy had not been administered before, and they may have gotten other HER2-targeted therapies if they had already received chemotherapy.
There were over 300 patients in this trial, and my memory is that roughly two thirds of them had a PET response. About 86 patients randomized to continue biologic therapy had a pathologic complete response, so about one-third of those for whom the PET imaging said they were responding with biologic therapy only had a pathologic complete response.
They have now been followed for 3 years. The 3-year disease-free survival results look very reassuring. Of those 86 patients, one patient had a local recurrence and no patient had a distant recurrence.
This is what we’ve been waiting for. Can we identify those patients who have an excellent prognosis with biologic therapy alone so that we can avoid the toxicities? This is really where you’ll see the research over the coming years in breast cancer, looking at additional therapies in high-risk patients who don’t do so well with our standard therapies, and better stratification of patients who do so well with our standard therapies that we may be able to do less.
This is one of the ways that we’ll be able to do that. I look forward to sharing those results with you over coming years.
Kathy D. Miller, MD, is associate director of clinical research and codirector of the breast cancer program at the Melvin and Bren Simon Cancer Center at Indiana University, Indianapolis. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.