User login
AVAHO
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]


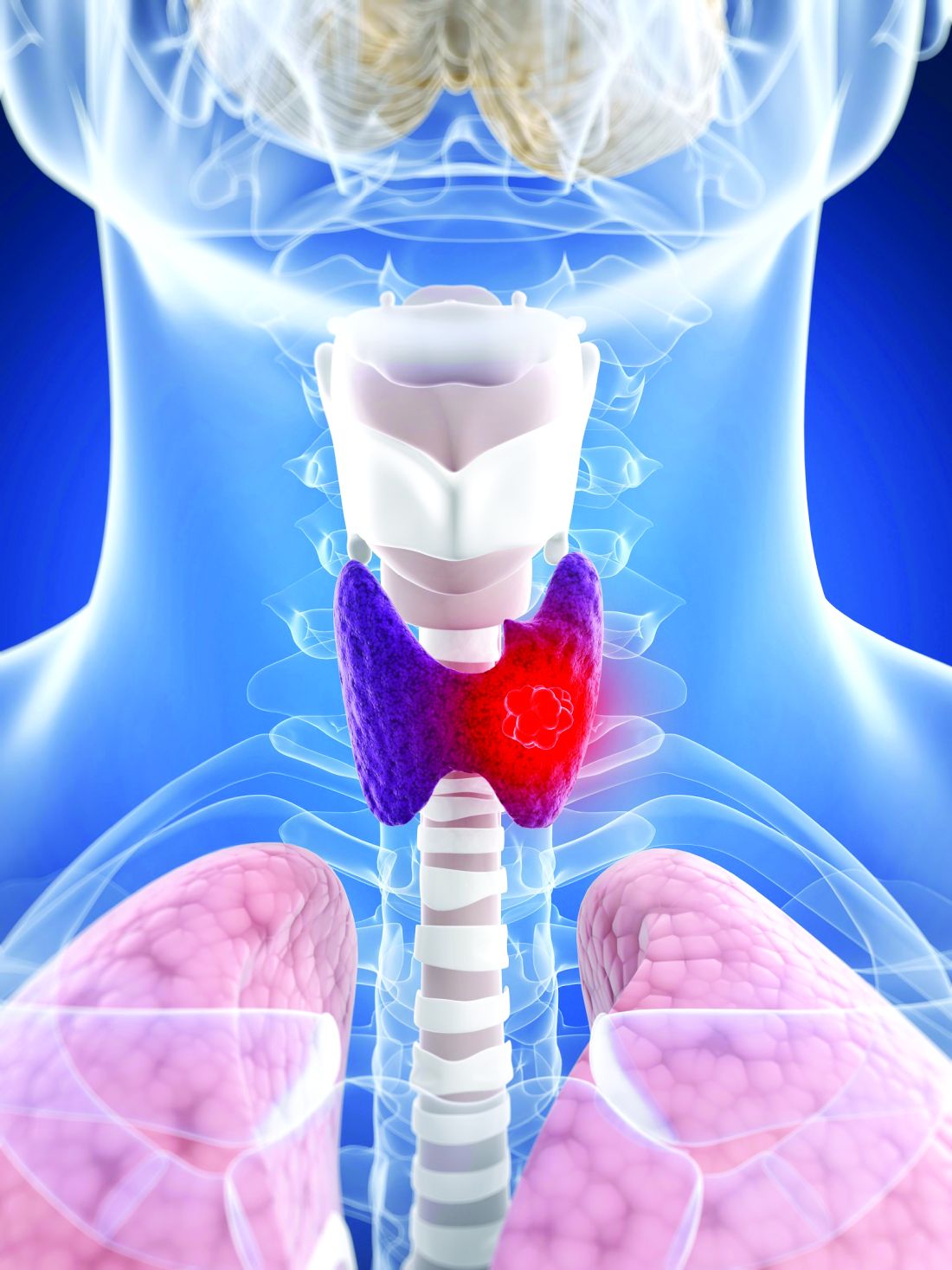
Cervical cancer rise in White women: A ‘canary in the coal mine’
Cervical cancer appears to be rising more rapidly in White women than in Black women in the United States, according to two independent studies. Researchers puzzling over this counterintuitive finding say that, if true, the findings may be a “canary in the coal mine,” signaling problems with U.S. health care that go way beyond women’s health.
Cervical cancer incidence in the United States has plateaued since 2010 and now stands at 7.5 per 100,000 people. Well-known disparities exist: Compared with White women, Black women are more likely to have distant-stage disease at diagnosis and more commonly die of their cancer.
However, two unconnected studies published in the past 5 months suggest that White women are catching up fast.
Cervical cancer rates in White women aged 30-34 are rising 2.8% a year, but holding steady for Black women, according to a recent study led by Ashish A. Deshmukh, PhD, of the Medical University of South Carolina, Charleston. His team analyzed the 2001-2019 National Program of Cancer Registries (NPCR) and Surveillance, Epidemiology, and End Results (SEER) dataset, which covers 98% of the U.S. population and 227,062 cervical cancer cases.
These findings were echoed by an analysis of the same database for 2001-2018 by the University of California, Los Angeles (UCLA), suggesting that distant-stage cervical cancer (defined as disease that has spread to the bladder and/or rectum at diagnosis) is climbing 1.69% a year in White people versus 0.67% in Black individuals.
The UCLA researchers, headed by Alex Francoeur, MD, a resident in the department of obstetrics and gynecology, found that disparities were most stark in adenocarcinoma, with an annual increase of 3.40% a year among White women and 1.71% in Black women.
Such findings have equity researchers scratching their heads. In cancer, it’s rare to see evidence that Black patients are doing better than their White counterparts.
One theoretical explanation is that the data are flawed, Ahmedin Jemal, DVM, PhD, senior vice president of surveillance and health equity science at the American Cancer Society, told this news organization. For example, the UCLA analysis may have been fogged by changes in staging definitions over time, Dr. Jemal said, although this would not explain the racial disparities per se.
Dr. Deshmukh stands by his data and said that, for him, the message is clear: “If rising incidence is not for localized-stage disease, but for advanced stages, that means it’s attributable to lack of screening,” he told this news organization.
However, this ‘simple’ explanation generates even more questions, Dr. Deshmukh said: “Screening is not a one-time procedure [but] a spectrum of timely cervical precancer treatment if [required]. We don’t know when exactly non-Hispanic White women are falling behind on that spectrum.”
The UCLA study supports Dr. Deshmukh’s conclusions. Using data from the Behavioral Risk Factor Surveillance System to calculate trends in “nonguideline screening,” the researchers found that White women were almost twice as likely to report that they were not following screening guidelines compared with Black women over the period of 2001-2016 (26.6% vs. 13.8%; P < .001).
“It’s not an artifact, it’s real,” said Timothy Rebbeck, PhD, the Vincent L. Gregory Jr. Professor of Cancer Prevention at Harvard TH Chan School of Public Health, Boston, who was not an author of either study and was approached for comment.
“The data are correct but there are so many things going on that might explain these patterns,” he told this news organization.
“This is a great example of complex changes in our social system, political system, health care system that are having really clear, measurable effects,” Dr. Rebbeck said. “Cervical cancer is almost a canary in the coal mine for some of this because it’s so preventable.”
(The saying “canary in a coal mine” is a warning of danger or trouble ahead. It comes from the time when coal miners would carry a caged canary down into the tunnels to warn them of noxious gases, which would kill the bird but give men time to escape.)
For example, Dr. Rebbeck said, recent turmoil in U.S. health care has left many people distrustful of the system. Although he acknowledged this was “high speculation,” he suggested that some women may have become less willing to participate in any mass health care intervention because of their political beliefs.
The UCLA study found that distant cervical cancer was rising fastest in middle-aged White women in the U.S. South, at a rate of 4.5% per year (P < .001).
Dr. Rebbeck also suggested that Medicaid expansion – the broadening of health insurance coverage in some states since the Affordable Care Act in 2014 – could be implicated. Of the 11 states that have not yet expanded Medicaid, eight are in the South.
“White populations who are in states that didn’t expand Medicaid are not getting a lot of the standard treatment and care that you would expect ...” Dr. Rebbeck said. “You could very well imagine that Medicaid nonexpanding states would have all kinds of patterns that would lead to more aggressive disease.”
In fact, there is already evidence that Medicaid expansion has affected racial disparities, disproportionately benefiting Black and Hispanic families, as for example, from an analysis of 65 studies by the Kaiser Family Foundation in 2020.
Commenting on these data, Dr. Rebbeck said, “Does that mean that the patterns of advanced cervical cancer had a smaller effect on Black women in this period because there was a greater shift in access to care? This is again a speculation, but it does fit with the ‘canary in a coal mine’ concept that advanced cervical cancer may be more rapidly influenced by health care access than other health conditions.”
The authors of the UCLA study suggested another explanation for their results: Differing enthusiasm for human papillomavirus (HPV) vaccination among White and Black families. The team also analyzed data on HPV vaccination, which offers protection against cervical cancer. The researchers found that vaccination rates were lowest, at 66.1%, among White teenagers aged 13-17 years, compared with Hispanics at 75.3%, Black teenagers at 74.6%, and Asians at 68.1%.
However, this theory was dismissed by both Dr. Jemal and Dr. Rebbeck due to timing. HPV vaccines have been around for approximately 15 years, so women who benefited (or didn’t benefit) from vaccination would be only in their late 20s today, they pointed out.
“Ninety-five percent of the cervical cancer cases we see now are in women who have not been vaccinated,” said Dr. Jemal, “So that’s out of the equation.”
Dr. Rebbeck agreed: “HPV may or may not be a thing here because it’s [got] such a latency.”
HPV vaccination may be out of the picture, but what about the epidemiology of HPV itself? Could the virus directly or indirectly be boosting advanced cervical cancer in White women?
Dr. Deshmukh thinks that it might be doing so.
He published an analysis of 2000-2018 SEER data showing that U.S. counties with the highest incidences of HPV-associated cancers also had the highest levels of smoking.
Other recent data suggest that middle-aged White women in the United States are more likely to reach for a smoke than are Black women.
Dr. Deshmukh acknowledges that the link is speculative but reasonable: “We don’t know exactly what the impact of smoking would be in terms of ... the ability to clear HPV infection. It may inhibit apoptosis, promoting tumor growth. There’s no causal association. It’s a cofactor risk.”
Dr. Rebbeck is also suspicious that smoking patterns might be a factor, pointing out that “smoking is certainly associated with both health behaviors and advanced cervical cancer.”
Both Dr. Rebbeck and Dr. Deshmukh concluded that, at this point, we can only speculate on what’s driving the puzzling acceleration of cervical cancer in White women in the United States.
However, whether it’s political aversion to screening, smoking-boosted HPV infection, Medicaid expansion or lack of it, or something else, they all agree that this canary in the coal mine clearly needs urgent medical attention.
Dr. Rebbeck and Dr. Jemal have declared no conflicts of interest. Dr. Deshmukh has declared consultant or advisory roles for Merck and Value Analytics Labs. None of the authors of the UCLA study have declared competing interests.
A version of this article first appeared on Medscape.com.
Cervical cancer appears to be rising more rapidly in White women than in Black women in the United States, according to two independent studies. Researchers puzzling over this counterintuitive finding say that, if true, the findings may be a “canary in the coal mine,” signaling problems with U.S. health care that go way beyond women’s health.
Cervical cancer incidence in the United States has plateaued since 2010 and now stands at 7.5 per 100,000 people. Well-known disparities exist: Compared with White women, Black women are more likely to have distant-stage disease at diagnosis and more commonly die of their cancer.
However, two unconnected studies published in the past 5 months suggest that White women are catching up fast.
Cervical cancer rates in White women aged 30-34 are rising 2.8% a year, but holding steady for Black women, according to a recent study led by Ashish A. Deshmukh, PhD, of the Medical University of South Carolina, Charleston. His team analyzed the 2001-2019 National Program of Cancer Registries (NPCR) and Surveillance, Epidemiology, and End Results (SEER) dataset, which covers 98% of the U.S. population and 227,062 cervical cancer cases.
These findings were echoed by an analysis of the same database for 2001-2018 by the University of California, Los Angeles (UCLA), suggesting that distant-stage cervical cancer (defined as disease that has spread to the bladder and/or rectum at diagnosis) is climbing 1.69% a year in White people versus 0.67% in Black individuals.
The UCLA researchers, headed by Alex Francoeur, MD, a resident in the department of obstetrics and gynecology, found that disparities were most stark in adenocarcinoma, with an annual increase of 3.40% a year among White women and 1.71% in Black women.
Such findings have equity researchers scratching their heads. In cancer, it’s rare to see evidence that Black patients are doing better than their White counterparts.
One theoretical explanation is that the data are flawed, Ahmedin Jemal, DVM, PhD, senior vice president of surveillance and health equity science at the American Cancer Society, told this news organization. For example, the UCLA analysis may have been fogged by changes in staging definitions over time, Dr. Jemal said, although this would not explain the racial disparities per se.
Dr. Deshmukh stands by his data and said that, for him, the message is clear: “If rising incidence is not for localized-stage disease, but for advanced stages, that means it’s attributable to lack of screening,” he told this news organization.
However, this ‘simple’ explanation generates even more questions, Dr. Deshmukh said: “Screening is not a one-time procedure [but] a spectrum of timely cervical precancer treatment if [required]. We don’t know when exactly non-Hispanic White women are falling behind on that spectrum.”
The UCLA study supports Dr. Deshmukh’s conclusions. Using data from the Behavioral Risk Factor Surveillance System to calculate trends in “nonguideline screening,” the researchers found that White women were almost twice as likely to report that they were not following screening guidelines compared with Black women over the period of 2001-2016 (26.6% vs. 13.8%; P < .001).
“It’s not an artifact, it’s real,” said Timothy Rebbeck, PhD, the Vincent L. Gregory Jr. Professor of Cancer Prevention at Harvard TH Chan School of Public Health, Boston, who was not an author of either study and was approached for comment.
“The data are correct but there are so many things going on that might explain these patterns,” he told this news organization.
“This is a great example of complex changes in our social system, political system, health care system that are having really clear, measurable effects,” Dr. Rebbeck said. “Cervical cancer is almost a canary in the coal mine for some of this because it’s so preventable.”
(The saying “canary in a coal mine” is a warning of danger or trouble ahead. It comes from the time when coal miners would carry a caged canary down into the tunnels to warn them of noxious gases, which would kill the bird but give men time to escape.)
For example, Dr. Rebbeck said, recent turmoil in U.S. health care has left many people distrustful of the system. Although he acknowledged this was “high speculation,” he suggested that some women may have become less willing to participate in any mass health care intervention because of their political beliefs.
The UCLA study found that distant cervical cancer was rising fastest in middle-aged White women in the U.S. South, at a rate of 4.5% per year (P < .001).
Dr. Rebbeck also suggested that Medicaid expansion – the broadening of health insurance coverage in some states since the Affordable Care Act in 2014 – could be implicated. Of the 11 states that have not yet expanded Medicaid, eight are in the South.
“White populations who are in states that didn’t expand Medicaid are not getting a lot of the standard treatment and care that you would expect ...” Dr. Rebbeck said. “You could very well imagine that Medicaid nonexpanding states would have all kinds of patterns that would lead to more aggressive disease.”
In fact, there is already evidence that Medicaid expansion has affected racial disparities, disproportionately benefiting Black and Hispanic families, as for example, from an analysis of 65 studies by the Kaiser Family Foundation in 2020.
Commenting on these data, Dr. Rebbeck said, “Does that mean that the patterns of advanced cervical cancer had a smaller effect on Black women in this period because there was a greater shift in access to care? This is again a speculation, but it does fit with the ‘canary in a coal mine’ concept that advanced cervical cancer may be more rapidly influenced by health care access than other health conditions.”
The authors of the UCLA study suggested another explanation for their results: Differing enthusiasm for human papillomavirus (HPV) vaccination among White and Black families. The team also analyzed data on HPV vaccination, which offers protection against cervical cancer. The researchers found that vaccination rates were lowest, at 66.1%, among White teenagers aged 13-17 years, compared with Hispanics at 75.3%, Black teenagers at 74.6%, and Asians at 68.1%.
However, this theory was dismissed by both Dr. Jemal and Dr. Rebbeck due to timing. HPV vaccines have been around for approximately 15 years, so women who benefited (or didn’t benefit) from vaccination would be only in their late 20s today, they pointed out.
“Ninety-five percent of the cervical cancer cases we see now are in women who have not been vaccinated,” said Dr. Jemal, “So that’s out of the equation.”
Dr. Rebbeck agreed: “HPV may or may not be a thing here because it’s [got] such a latency.”
HPV vaccination may be out of the picture, but what about the epidemiology of HPV itself? Could the virus directly or indirectly be boosting advanced cervical cancer in White women?
Dr. Deshmukh thinks that it might be doing so.
He published an analysis of 2000-2018 SEER data showing that U.S. counties with the highest incidences of HPV-associated cancers also had the highest levels of smoking.
Other recent data suggest that middle-aged White women in the United States are more likely to reach for a smoke than are Black women.
Dr. Deshmukh acknowledges that the link is speculative but reasonable: “We don’t know exactly what the impact of smoking would be in terms of ... the ability to clear HPV infection. It may inhibit apoptosis, promoting tumor growth. There’s no causal association. It’s a cofactor risk.”
Dr. Rebbeck is also suspicious that smoking patterns might be a factor, pointing out that “smoking is certainly associated with both health behaviors and advanced cervical cancer.”
Both Dr. Rebbeck and Dr. Deshmukh concluded that, at this point, we can only speculate on what’s driving the puzzling acceleration of cervical cancer in White women in the United States.
However, whether it’s political aversion to screening, smoking-boosted HPV infection, Medicaid expansion or lack of it, or something else, they all agree that this canary in the coal mine clearly needs urgent medical attention.
Dr. Rebbeck and Dr. Jemal have declared no conflicts of interest. Dr. Deshmukh has declared consultant or advisory roles for Merck and Value Analytics Labs. None of the authors of the UCLA study have declared competing interests.
A version of this article first appeared on Medscape.com.
Cervical cancer appears to be rising more rapidly in White women than in Black women in the United States, according to two independent studies. Researchers puzzling over this counterintuitive finding say that, if true, the findings may be a “canary in the coal mine,” signaling problems with U.S. health care that go way beyond women’s health.
Cervical cancer incidence in the United States has plateaued since 2010 and now stands at 7.5 per 100,000 people. Well-known disparities exist: Compared with White women, Black women are more likely to have distant-stage disease at diagnosis and more commonly die of their cancer.
However, two unconnected studies published in the past 5 months suggest that White women are catching up fast.
Cervical cancer rates in White women aged 30-34 are rising 2.8% a year, but holding steady for Black women, according to a recent study led by Ashish A. Deshmukh, PhD, of the Medical University of South Carolina, Charleston. His team analyzed the 2001-2019 National Program of Cancer Registries (NPCR) and Surveillance, Epidemiology, and End Results (SEER) dataset, which covers 98% of the U.S. population and 227,062 cervical cancer cases.
These findings were echoed by an analysis of the same database for 2001-2018 by the University of California, Los Angeles (UCLA), suggesting that distant-stage cervical cancer (defined as disease that has spread to the bladder and/or rectum at diagnosis) is climbing 1.69% a year in White people versus 0.67% in Black individuals.
The UCLA researchers, headed by Alex Francoeur, MD, a resident in the department of obstetrics and gynecology, found that disparities were most stark in adenocarcinoma, with an annual increase of 3.40% a year among White women and 1.71% in Black women.
Such findings have equity researchers scratching their heads. In cancer, it’s rare to see evidence that Black patients are doing better than their White counterparts.
One theoretical explanation is that the data are flawed, Ahmedin Jemal, DVM, PhD, senior vice president of surveillance and health equity science at the American Cancer Society, told this news organization. For example, the UCLA analysis may have been fogged by changes in staging definitions over time, Dr. Jemal said, although this would not explain the racial disparities per se.
Dr. Deshmukh stands by his data and said that, for him, the message is clear: “If rising incidence is not for localized-stage disease, but for advanced stages, that means it’s attributable to lack of screening,” he told this news organization.
However, this ‘simple’ explanation generates even more questions, Dr. Deshmukh said: “Screening is not a one-time procedure [but] a spectrum of timely cervical precancer treatment if [required]. We don’t know when exactly non-Hispanic White women are falling behind on that spectrum.”
The UCLA study supports Dr. Deshmukh’s conclusions. Using data from the Behavioral Risk Factor Surveillance System to calculate trends in “nonguideline screening,” the researchers found that White women were almost twice as likely to report that they were not following screening guidelines compared with Black women over the period of 2001-2016 (26.6% vs. 13.8%; P < .001).
“It’s not an artifact, it’s real,” said Timothy Rebbeck, PhD, the Vincent L. Gregory Jr. Professor of Cancer Prevention at Harvard TH Chan School of Public Health, Boston, who was not an author of either study and was approached for comment.
“The data are correct but there are so many things going on that might explain these patterns,” he told this news organization.
“This is a great example of complex changes in our social system, political system, health care system that are having really clear, measurable effects,” Dr. Rebbeck said. “Cervical cancer is almost a canary in the coal mine for some of this because it’s so preventable.”
(The saying “canary in a coal mine” is a warning of danger or trouble ahead. It comes from the time when coal miners would carry a caged canary down into the tunnels to warn them of noxious gases, which would kill the bird but give men time to escape.)
For example, Dr. Rebbeck said, recent turmoil in U.S. health care has left many people distrustful of the system. Although he acknowledged this was “high speculation,” he suggested that some women may have become less willing to participate in any mass health care intervention because of their political beliefs.
The UCLA study found that distant cervical cancer was rising fastest in middle-aged White women in the U.S. South, at a rate of 4.5% per year (P < .001).
Dr. Rebbeck also suggested that Medicaid expansion – the broadening of health insurance coverage in some states since the Affordable Care Act in 2014 – could be implicated. Of the 11 states that have not yet expanded Medicaid, eight are in the South.
“White populations who are in states that didn’t expand Medicaid are not getting a lot of the standard treatment and care that you would expect ...” Dr. Rebbeck said. “You could very well imagine that Medicaid nonexpanding states would have all kinds of patterns that would lead to more aggressive disease.”
In fact, there is already evidence that Medicaid expansion has affected racial disparities, disproportionately benefiting Black and Hispanic families, as for example, from an analysis of 65 studies by the Kaiser Family Foundation in 2020.
Commenting on these data, Dr. Rebbeck said, “Does that mean that the patterns of advanced cervical cancer had a smaller effect on Black women in this period because there was a greater shift in access to care? This is again a speculation, but it does fit with the ‘canary in a coal mine’ concept that advanced cervical cancer may be more rapidly influenced by health care access than other health conditions.”
The authors of the UCLA study suggested another explanation for their results: Differing enthusiasm for human papillomavirus (HPV) vaccination among White and Black families. The team also analyzed data on HPV vaccination, which offers protection against cervical cancer. The researchers found that vaccination rates were lowest, at 66.1%, among White teenagers aged 13-17 years, compared with Hispanics at 75.3%, Black teenagers at 74.6%, and Asians at 68.1%.
However, this theory was dismissed by both Dr. Jemal and Dr. Rebbeck due to timing. HPV vaccines have been around for approximately 15 years, so women who benefited (or didn’t benefit) from vaccination would be only in their late 20s today, they pointed out.
“Ninety-five percent of the cervical cancer cases we see now are in women who have not been vaccinated,” said Dr. Jemal, “So that’s out of the equation.”
Dr. Rebbeck agreed: “HPV may or may not be a thing here because it’s [got] such a latency.”
HPV vaccination may be out of the picture, but what about the epidemiology of HPV itself? Could the virus directly or indirectly be boosting advanced cervical cancer in White women?
Dr. Deshmukh thinks that it might be doing so.
He published an analysis of 2000-2018 SEER data showing that U.S. counties with the highest incidences of HPV-associated cancers also had the highest levels of smoking.
Other recent data suggest that middle-aged White women in the United States are more likely to reach for a smoke than are Black women.
Dr. Deshmukh acknowledges that the link is speculative but reasonable: “We don’t know exactly what the impact of smoking would be in terms of ... the ability to clear HPV infection. It may inhibit apoptosis, promoting tumor growth. There’s no causal association. It’s a cofactor risk.”
Dr. Rebbeck is also suspicious that smoking patterns might be a factor, pointing out that “smoking is certainly associated with both health behaviors and advanced cervical cancer.”
Both Dr. Rebbeck and Dr. Deshmukh concluded that, at this point, we can only speculate on what’s driving the puzzling acceleration of cervical cancer in White women in the United States.
However, whether it’s political aversion to screening, smoking-boosted HPV infection, Medicaid expansion or lack of it, or something else, they all agree that this canary in the coal mine clearly needs urgent medical attention.
Dr. Rebbeck and Dr. Jemal have declared no conflicts of interest. Dr. Deshmukh has declared consultant or advisory roles for Merck and Value Analytics Labs. None of the authors of the UCLA study have declared competing interests.
A version of this article first appeared on Medscape.com.
Study of beliefs about what causes cancer sparks debate
The study, entitled, “Everything Causes Cancer? Beliefs and Attitudes Towards Cancer Prevention Among Anti-Vaxxers, Flat Earthers, and Reptilian Conspiracists: Online Cross Sectional Survey,” was published in the Christmas 2022 issue of The British Medical Journal (BMJ).
The authors explain that they set out to evaluate “the patterns of beliefs about cancer among people who believed in conspiracies, rejected the COVID-19 vaccine, or preferred alternative medicine.”
They sought such people on social media and online chat platforms and asked them questions about real and mythical causes of cancer.
Almost half of survey participants agreed with the statement, “It seems like everything causes cancer.”
Overall, among all participants, awareness of the actual causes of cancer was greater than awareness of the mythical causes of cancer, the authors report. However, awareness of the actual causes of cancer was lower among the unvaccinated and members of conspiracy groups than among their counterparts.
The authors are concerned that their findings suggest “a direct connection between digital misinformation and consequent potential erroneous health decisions, which may represent a further preventable fraction of cancer.”
Backlash and criticism
The study “highlights the difficulty society encounters in distinguishing the actual causes of cancer from mythical causes,” The BMJ commented on Twitter.
However, both the study and the journal received some backlash.
This is a “horrible article seeking to smear people with concerns about COVID vaccines,” commented Clare Craig, a British consultant pathologist who specializes in cancer diagnostics.
The study and its methodology were also harshly criticized on Twitter by Normal Fenton, professor of risk information management at the Queen Mary University of London.
The senior author of the study, Laura Costas, a medical epidemiologist with the Catalan Institute of Oncology, Barcelona, told this news organization that the naysayers on social media, many of whom focused their comments on the COVID-19 vaccine, prove the purpose of the study – that misinformation spreads widely on the internet.
“Most comments focused on spreading COVID-19 myths, which were not the direct subject of the study, and questioned the motivations of BMJ authors and the scientific community, assuming they had a common malevolent hidden agenda,” Ms. Costas said.
“They stated the need of having critical thinking, a trait in common with the scientific method, but dogmatically dismissed any information that comes from official sources,” she added.
Ms. Costas commented that “society encounters difficulty in differentiating actual from mythical causes of cancer owing to mass information. We therefore planned this study with a certain satire, which is in line with the essence of The BMJ Christmas issue.”
The BMJ has a long history of publishing a lighthearted Christmas edition full of original, satirical, and nontraditional studies. Previous years have seen studies that explored potential harms from holly and ivy, survival time of chocolates on hospital wards, and the question, “Were James Bond’s drinks shaken because of alcohol induced tremor?”
Study details
Ms. Costas and colleagues sought participants for their survey from online forums that included 4chan and Reddit, which are known for their controversial content posted by anonymous users. Data were also collected from ForoCoches and HispaChan, well-known Spanish online forums. These online sites were intentionally chosen because researchers thought “conspiracy beliefs would be more prevalent,” according to Ms. Costas.
Across the multiple forums, there were 1,494 participants. Of these, 209 participants were unvaccinated against COVID-19, 112 preferred alternatives rather than conventional medicine, and 62 reported that they believed the earth was flat or believed that humanoids take reptilian forms to manipulate human societies.
The team then sought to assess beliefs about actual and mythical (nonestablished) causes of cancer by presenting the participants with the closed risk factor questions on two validated scales – the Cancer Awareness Measure (CAM) and CAM–Mythical Causes Scale (CAM-MYCS).
Responses to both were recorded on a five-point scale; answers ranged from “strongly disagree” to “strongly agree.”
The CAM assesses cancer risk perceptions of 11 established risk factors for cancer: smoking actively or passively, consuming alcohol, low levels of physical activity, consuming red or processed meat, getting sunburnt as a child, family history of cancer, human papillomavirus infection, being overweight, age greater than or equal to 70 years, and low vegetable and fruit consumption.
The CAM-MYCS measure includes 12 questions on risk perceptions of mythical causes of cancer – nonestablished causes that are commonly believed to cause cancer but for which there is no supporting scientific evidence, the authors explain. These items include drinking from plastic bottles; eating food containing artificial sweeteners or additives and genetically modified food; using microwave ovens, aerosol containers, mobile phones, and cleaning products; living near power lines; feeling stressed; experiencing physical trauma; and being exposed to electromagnetic frequencies/non-ionizing radiation, such as wi-fi networks, radio, and television.
The most endorsed mythical causes of cancer were eating food containing additives (63.9%) or sweeteners (50.7%), feeling stressed (59.7%), and eating genetically modified foods (38.4%).
A version of this article first appeared on Medscape.com.
The study, entitled, “Everything Causes Cancer? Beliefs and Attitudes Towards Cancer Prevention Among Anti-Vaxxers, Flat Earthers, and Reptilian Conspiracists: Online Cross Sectional Survey,” was published in the Christmas 2022 issue of The British Medical Journal (BMJ).
The authors explain that they set out to evaluate “the patterns of beliefs about cancer among people who believed in conspiracies, rejected the COVID-19 vaccine, or preferred alternative medicine.”
They sought such people on social media and online chat platforms and asked them questions about real and mythical causes of cancer.
Almost half of survey participants agreed with the statement, “It seems like everything causes cancer.”
Overall, among all participants, awareness of the actual causes of cancer was greater than awareness of the mythical causes of cancer, the authors report. However, awareness of the actual causes of cancer was lower among the unvaccinated and members of conspiracy groups than among their counterparts.
The authors are concerned that their findings suggest “a direct connection between digital misinformation and consequent potential erroneous health decisions, which may represent a further preventable fraction of cancer.”
Backlash and criticism
The study “highlights the difficulty society encounters in distinguishing the actual causes of cancer from mythical causes,” The BMJ commented on Twitter.
However, both the study and the journal received some backlash.
This is a “horrible article seeking to smear people with concerns about COVID vaccines,” commented Clare Craig, a British consultant pathologist who specializes in cancer diagnostics.
The study and its methodology were also harshly criticized on Twitter by Normal Fenton, professor of risk information management at the Queen Mary University of London.
The senior author of the study, Laura Costas, a medical epidemiologist with the Catalan Institute of Oncology, Barcelona, told this news organization that the naysayers on social media, many of whom focused their comments on the COVID-19 vaccine, prove the purpose of the study – that misinformation spreads widely on the internet.
“Most comments focused on spreading COVID-19 myths, which were not the direct subject of the study, and questioned the motivations of BMJ authors and the scientific community, assuming they had a common malevolent hidden agenda,” Ms. Costas said.
“They stated the need of having critical thinking, a trait in common with the scientific method, but dogmatically dismissed any information that comes from official sources,” she added.
Ms. Costas commented that “society encounters difficulty in differentiating actual from mythical causes of cancer owing to mass information. We therefore planned this study with a certain satire, which is in line with the essence of The BMJ Christmas issue.”
The BMJ has a long history of publishing a lighthearted Christmas edition full of original, satirical, and nontraditional studies. Previous years have seen studies that explored potential harms from holly and ivy, survival time of chocolates on hospital wards, and the question, “Were James Bond’s drinks shaken because of alcohol induced tremor?”
Study details
Ms. Costas and colleagues sought participants for their survey from online forums that included 4chan and Reddit, which are known for their controversial content posted by anonymous users. Data were also collected from ForoCoches and HispaChan, well-known Spanish online forums. These online sites were intentionally chosen because researchers thought “conspiracy beliefs would be more prevalent,” according to Ms. Costas.
Across the multiple forums, there were 1,494 participants. Of these, 209 participants were unvaccinated against COVID-19, 112 preferred alternatives rather than conventional medicine, and 62 reported that they believed the earth was flat or believed that humanoids take reptilian forms to manipulate human societies.
The team then sought to assess beliefs about actual and mythical (nonestablished) causes of cancer by presenting the participants with the closed risk factor questions on two validated scales – the Cancer Awareness Measure (CAM) and CAM–Mythical Causes Scale (CAM-MYCS).
Responses to both were recorded on a five-point scale; answers ranged from “strongly disagree” to “strongly agree.”
The CAM assesses cancer risk perceptions of 11 established risk factors for cancer: smoking actively or passively, consuming alcohol, low levels of physical activity, consuming red or processed meat, getting sunburnt as a child, family history of cancer, human papillomavirus infection, being overweight, age greater than or equal to 70 years, and low vegetable and fruit consumption.
The CAM-MYCS measure includes 12 questions on risk perceptions of mythical causes of cancer – nonestablished causes that are commonly believed to cause cancer but for which there is no supporting scientific evidence, the authors explain. These items include drinking from plastic bottles; eating food containing artificial sweeteners or additives and genetically modified food; using microwave ovens, aerosol containers, mobile phones, and cleaning products; living near power lines; feeling stressed; experiencing physical trauma; and being exposed to electromagnetic frequencies/non-ionizing radiation, such as wi-fi networks, radio, and television.
The most endorsed mythical causes of cancer were eating food containing additives (63.9%) or sweeteners (50.7%), feeling stressed (59.7%), and eating genetically modified foods (38.4%).
A version of this article first appeared on Medscape.com.
The study, entitled, “Everything Causes Cancer? Beliefs and Attitudes Towards Cancer Prevention Among Anti-Vaxxers, Flat Earthers, and Reptilian Conspiracists: Online Cross Sectional Survey,” was published in the Christmas 2022 issue of The British Medical Journal (BMJ).
The authors explain that they set out to evaluate “the patterns of beliefs about cancer among people who believed in conspiracies, rejected the COVID-19 vaccine, or preferred alternative medicine.”
They sought such people on social media and online chat platforms and asked them questions about real and mythical causes of cancer.
Almost half of survey participants agreed with the statement, “It seems like everything causes cancer.”
Overall, among all participants, awareness of the actual causes of cancer was greater than awareness of the mythical causes of cancer, the authors report. However, awareness of the actual causes of cancer was lower among the unvaccinated and members of conspiracy groups than among their counterparts.
The authors are concerned that their findings suggest “a direct connection between digital misinformation and consequent potential erroneous health decisions, which may represent a further preventable fraction of cancer.”
Backlash and criticism
The study “highlights the difficulty society encounters in distinguishing the actual causes of cancer from mythical causes,” The BMJ commented on Twitter.
However, both the study and the journal received some backlash.
This is a “horrible article seeking to smear people with concerns about COVID vaccines,” commented Clare Craig, a British consultant pathologist who specializes in cancer diagnostics.
The study and its methodology were also harshly criticized on Twitter by Normal Fenton, professor of risk information management at the Queen Mary University of London.
The senior author of the study, Laura Costas, a medical epidemiologist with the Catalan Institute of Oncology, Barcelona, told this news organization that the naysayers on social media, many of whom focused their comments on the COVID-19 vaccine, prove the purpose of the study – that misinformation spreads widely on the internet.
“Most comments focused on spreading COVID-19 myths, which were not the direct subject of the study, and questioned the motivations of BMJ authors and the scientific community, assuming they had a common malevolent hidden agenda,” Ms. Costas said.
“They stated the need of having critical thinking, a trait in common with the scientific method, but dogmatically dismissed any information that comes from official sources,” she added.
Ms. Costas commented that “society encounters difficulty in differentiating actual from mythical causes of cancer owing to mass information. We therefore planned this study with a certain satire, which is in line with the essence of The BMJ Christmas issue.”
The BMJ has a long history of publishing a lighthearted Christmas edition full of original, satirical, and nontraditional studies. Previous years have seen studies that explored potential harms from holly and ivy, survival time of chocolates on hospital wards, and the question, “Were James Bond’s drinks shaken because of alcohol induced tremor?”
Study details
Ms. Costas and colleagues sought participants for their survey from online forums that included 4chan and Reddit, which are known for their controversial content posted by anonymous users. Data were also collected from ForoCoches and HispaChan, well-known Spanish online forums. These online sites were intentionally chosen because researchers thought “conspiracy beliefs would be more prevalent,” according to Ms. Costas.
Across the multiple forums, there were 1,494 participants. Of these, 209 participants were unvaccinated against COVID-19, 112 preferred alternatives rather than conventional medicine, and 62 reported that they believed the earth was flat or believed that humanoids take reptilian forms to manipulate human societies.
The team then sought to assess beliefs about actual and mythical (nonestablished) causes of cancer by presenting the participants with the closed risk factor questions on two validated scales – the Cancer Awareness Measure (CAM) and CAM–Mythical Causes Scale (CAM-MYCS).
Responses to both were recorded on a five-point scale; answers ranged from “strongly disagree” to “strongly agree.”
The CAM assesses cancer risk perceptions of 11 established risk factors for cancer: smoking actively or passively, consuming alcohol, low levels of physical activity, consuming red or processed meat, getting sunburnt as a child, family history of cancer, human papillomavirus infection, being overweight, age greater than or equal to 70 years, and low vegetable and fruit consumption.
The CAM-MYCS measure includes 12 questions on risk perceptions of mythical causes of cancer – nonestablished causes that are commonly believed to cause cancer but for which there is no supporting scientific evidence, the authors explain. These items include drinking from plastic bottles; eating food containing artificial sweeteners or additives and genetically modified food; using microwave ovens, aerosol containers, mobile phones, and cleaning products; living near power lines; feeling stressed; experiencing physical trauma; and being exposed to electromagnetic frequencies/non-ionizing radiation, such as wi-fi networks, radio, and television.
The most endorsed mythical causes of cancer were eating food containing additives (63.9%) or sweeteners (50.7%), feeling stressed (59.7%), and eating genetically modified foods (38.4%).
A version of this article first appeared on Medscape.com.
Genetic test identifies parental source of cancer variant
A new hereditary test can determine whether a cancer-disposing gene was inherited from a patient’s father or mother without the need for parental DNA, potentially improving disease screening and management.
“The presence of parental imprints in regions of the genome has been known for a long time,” study author Peter Lansdorp, MD, PhD, of the BC Cancer Research Centre in Vancouver, said in an interview. In addition, the ability of a specific sequencing technology (Strand-seq) to generate a set of DNA variants that tend to be inherited together from a single parent has been documented in several studies.
“That these two pieces can be put together to assign alleles in a patient to one of the parents without studying the DNA of the parents is a major advance,” said Dr. Lansdorp.
Principal author Steven J.M. Jones, PhD, associate director of bioinformatics at BC Cancer Research Centre, explained, “for directing cascade genetic testing, the test could be used almost immediately, even as a research test. It just guides which side of the family to focus familial genetic testing efforts on and is internally validated by the patient’s variant and later confirmed by clinical testing in the family.”
Dr. Jones added, especially when parents are deceased or unavailable.
The study was published online in Cell Genomics.
Low error rate
Determining a parent of origin for hereditary variants “is essential to evaluate disease risk when a pathogenic variant has PofO effects, that is, when a patient’s risk of disease depends on from which parent it is inherited,” the authors wrote. An example is hereditary paraganglioma-pheochromocytoma syndrome as a result of pathogenic variants in SDHD or SDHAF2 genes. Individuals with the variants are at high risk of developing certain cancers, but only if a defective gene is inherited from their father. If inherited from their mother, there is no increased risk.
The new method relies on a technique called “phased DNA methylation” at maternally and paternally imprinted gene loci, as well as chromosome length phasing of DNA sequences.
The team used five human genome “trios” – two parents and the proband (the first person in a family to receive genetic testing or counseling for a suspected hereditary risk) – to pilot the approach. They showed that the method can correctly identify the PofO with an average mismatch error rate of 0.31% for single nucleotide variants and 1.89% for insertions or deletions (indels).
“We will need to validate this technology for different genes in real-world samples from individuals of diverse backgrounds,” said Dr. Jones. The first step is to validate the technology in scenarios with immediate clinical utility, like with SDHD, where lifelong medical management is affected by knowledge of whether the variant was inherited from the mother or father.
“We would also like to quickly validate this for common hereditary cancer genes, like BRCA1, BRCA2, and Lynch syndrome–associated genes, where prediction of PofO may improve low rates of genetic testing in family members by providing more accurate estimates of their risk to carry the familial variant.”
Challenges to moving the test to the clinic, Dr. Jones said, include scaling up the technology, demonstrating clinical and economic utility, compared with existing testing approaches, “and familiarizing clinicians with a new type of test that will routinely give this added dimension of information.”
‘Tremendously promising technology’
Pathologist Stephen Yip, MD, PhD, of the Vancouver Coastal Health Research Institute, who was not involved in the study but disclosed that he collaborates with the authors on other grant-funded projects, said in a comment that “this is a tremendously promising technology that has immediate practical implications in the investigation of PofO of a pathogenic locus, particularly when genetic material is available only from the proband.”
However, “rigorous validation against the current gold standard of short-reading, next-generation sequencing of trios is needed prior to clinical deployment,” he said. “This will take time and effort. However, the promise of this technology is worth the effort.
“Also, there is the possibility of uncovering novel genetics during testing, which could present an ethical dilemma,” he noted. “A robust consenting and ethical framework and early involvement of an ethicist would be helpful.”
Research in Dr. Lansdorp’s laboratory is funded by the Terry Fox Research Institute, the Canadian Institutes of Health Research, the Canadian Foundation for Innovation, and the government of British Columbia. Dr. Lansdorp, Dr. Jones, and Dr. Yip reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new hereditary test can determine whether a cancer-disposing gene was inherited from a patient’s father or mother without the need for parental DNA, potentially improving disease screening and management.
“The presence of parental imprints in regions of the genome has been known for a long time,” study author Peter Lansdorp, MD, PhD, of the BC Cancer Research Centre in Vancouver, said in an interview. In addition, the ability of a specific sequencing technology (Strand-seq) to generate a set of DNA variants that tend to be inherited together from a single parent has been documented in several studies.
“That these two pieces can be put together to assign alleles in a patient to one of the parents without studying the DNA of the parents is a major advance,” said Dr. Lansdorp.
Principal author Steven J.M. Jones, PhD, associate director of bioinformatics at BC Cancer Research Centre, explained, “for directing cascade genetic testing, the test could be used almost immediately, even as a research test. It just guides which side of the family to focus familial genetic testing efforts on and is internally validated by the patient’s variant and later confirmed by clinical testing in the family.”
Dr. Jones added, especially when parents are deceased or unavailable.
The study was published online in Cell Genomics.
Low error rate
Determining a parent of origin for hereditary variants “is essential to evaluate disease risk when a pathogenic variant has PofO effects, that is, when a patient’s risk of disease depends on from which parent it is inherited,” the authors wrote. An example is hereditary paraganglioma-pheochromocytoma syndrome as a result of pathogenic variants in SDHD or SDHAF2 genes. Individuals with the variants are at high risk of developing certain cancers, but only if a defective gene is inherited from their father. If inherited from their mother, there is no increased risk.
The new method relies on a technique called “phased DNA methylation” at maternally and paternally imprinted gene loci, as well as chromosome length phasing of DNA sequences.
The team used five human genome “trios” – two parents and the proband (the first person in a family to receive genetic testing or counseling for a suspected hereditary risk) – to pilot the approach. They showed that the method can correctly identify the PofO with an average mismatch error rate of 0.31% for single nucleotide variants and 1.89% for insertions or deletions (indels).
“We will need to validate this technology for different genes in real-world samples from individuals of diverse backgrounds,” said Dr. Jones. The first step is to validate the technology in scenarios with immediate clinical utility, like with SDHD, where lifelong medical management is affected by knowledge of whether the variant was inherited from the mother or father.
“We would also like to quickly validate this for common hereditary cancer genes, like BRCA1, BRCA2, and Lynch syndrome–associated genes, where prediction of PofO may improve low rates of genetic testing in family members by providing more accurate estimates of their risk to carry the familial variant.”
Challenges to moving the test to the clinic, Dr. Jones said, include scaling up the technology, demonstrating clinical and economic utility, compared with existing testing approaches, “and familiarizing clinicians with a new type of test that will routinely give this added dimension of information.”
‘Tremendously promising technology’
Pathologist Stephen Yip, MD, PhD, of the Vancouver Coastal Health Research Institute, who was not involved in the study but disclosed that he collaborates with the authors on other grant-funded projects, said in a comment that “this is a tremendously promising technology that has immediate practical implications in the investigation of PofO of a pathogenic locus, particularly when genetic material is available only from the proband.”
However, “rigorous validation against the current gold standard of short-reading, next-generation sequencing of trios is needed prior to clinical deployment,” he said. “This will take time and effort. However, the promise of this technology is worth the effort.
“Also, there is the possibility of uncovering novel genetics during testing, which could present an ethical dilemma,” he noted. “A robust consenting and ethical framework and early involvement of an ethicist would be helpful.”
Research in Dr. Lansdorp’s laboratory is funded by the Terry Fox Research Institute, the Canadian Institutes of Health Research, the Canadian Foundation for Innovation, and the government of British Columbia. Dr. Lansdorp, Dr. Jones, and Dr. Yip reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new hereditary test can determine whether a cancer-disposing gene was inherited from a patient’s father or mother without the need for parental DNA, potentially improving disease screening and management.
“The presence of parental imprints in regions of the genome has been known for a long time,” study author Peter Lansdorp, MD, PhD, of the BC Cancer Research Centre in Vancouver, said in an interview. In addition, the ability of a specific sequencing technology (Strand-seq) to generate a set of DNA variants that tend to be inherited together from a single parent has been documented in several studies.
“That these two pieces can be put together to assign alleles in a patient to one of the parents without studying the DNA of the parents is a major advance,” said Dr. Lansdorp.
Principal author Steven J.M. Jones, PhD, associate director of bioinformatics at BC Cancer Research Centre, explained, “for directing cascade genetic testing, the test could be used almost immediately, even as a research test. It just guides which side of the family to focus familial genetic testing efforts on and is internally validated by the patient’s variant and later confirmed by clinical testing in the family.”
Dr. Jones added, especially when parents are deceased or unavailable.
The study was published online in Cell Genomics.
Low error rate
Determining a parent of origin for hereditary variants “is essential to evaluate disease risk when a pathogenic variant has PofO effects, that is, when a patient’s risk of disease depends on from which parent it is inherited,” the authors wrote. An example is hereditary paraganglioma-pheochromocytoma syndrome as a result of pathogenic variants in SDHD or SDHAF2 genes. Individuals with the variants are at high risk of developing certain cancers, but only if a defective gene is inherited from their father. If inherited from their mother, there is no increased risk.
The new method relies on a technique called “phased DNA methylation” at maternally and paternally imprinted gene loci, as well as chromosome length phasing of DNA sequences.
The team used five human genome “trios” – two parents and the proband (the first person in a family to receive genetic testing or counseling for a suspected hereditary risk) – to pilot the approach. They showed that the method can correctly identify the PofO with an average mismatch error rate of 0.31% for single nucleotide variants and 1.89% for insertions or deletions (indels).
“We will need to validate this technology for different genes in real-world samples from individuals of diverse backgrounds,” said Dr. Jones. The first step is to validate the technology in scenarios with immediate clinical utility, like with SDHD, where lifelong medical management is affected by knowledge of whether the variant was inherited from the mother or father.
“We would also like to quickly validate this for common hereditary cancer genes, like BRCA1, BRCA2, and Lynch syndrome–associated genes, where prediction of PofO may improve low rates of genetic testing in family members by providing more accurate estimates of their risk to carry the familial variant.”
Challenges to moving the test to the clinic, Dr. Jones said, include scaling up the technology, demonstrating clinical and economic utility, compared with existing testing approaches, “and familiarizing clinicians with a new type of test that will routinely give this added dimension of information.”
‘Tremendously promising technology’
Pathologist Stephen Yip, MD, PhD, of the Vancouver Coastal Health Research Institute, who was not involved in the study but disclosed that he collaborates with the authors on other grant-funded projects, said in a comment that “this is a tremendously promising technology that has immediate practical implications in the investigation of PofO of a pathogenic locus, particularly when genetic material is available only from the proband.”
However, “rigorous validation against the current gold standard of short-reading, next-generation sequencing of trios is needed prior to clinical deployment,” he said. “This will take time and effort. However, the promise of this technology is worth the effort.
“Also, there is the possibility of uncovering novel genetics during testing, which could present an ethical dilemma,” he noted. “A robust consenting and ethical framework and early involvement of an ethicist would be helpful.”
Research in Dr. Lansdorp’s laboratory is funded by the Terry Fox Research Institute, the Canadian Institutes of Health Research, the Canadian Foundation for Innovation, and the government of British Columbia. Dr. Lansdorp, Dr. Jones, and Dr. Yip reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CELL GENOMICS
Most women with breast cancer elude serious COVID-19 vaccine side effects
Findings from the LymphVAX study recently presented at the San Antonio Breast Cancer Symposium show that relatively
Lymph node swelling can be a particularly troubling side effect, since it could be mistaken for breast cancer progression. In this study, of 621 women who received the first dose of an mRNA COVID-19 vaccine, 9.8% developed lymph node swelling as compared with 12.9% of 621 women who received the second dose, and 11.3% of 469 women who received the third dose. The findings were comparable to those of studies conducted of the general population, said study author Brooke C. Juhel, BS, a clinical research coordinator in the lymphedema research program at Massachusetts General Hospital and a student at Harvard Medical School, both in Boston. In the general population, 10.2% experienced lymph node swelling after the first dose and 14% after the second dose, according to the Centers for Disease Control and studies of the Pfizer and Moderna vaccines.
“This is consistent with the hypothesis that, after repeated vaccine doses, the immune system already has the antigens ready to fight the virus, thus the side effects may worsen as the immune response has increased,” she said. “Having screened over 6,500 women for breast cancer–related lymphedema, and with our patients reaching out with concerns about vaccine side effects, we were in a unique position to conduct this study.”
The study also confirmed that the most common side effects of receiving mRNA COVID-19 vaccines for women treated for breast cancer included injection site soreness, fatigue, muscle soreness, headache and chills lasting an average of 48 hours, which are symptoms comparable with those experienced by the general population.
“The side-effect profiles reported in this study for a cohort of women treated for breast cancer can be used to provide evidence-based patient education regarding future COVID-19 vaccine administration. The effect of the COVID-19 vaccines on breast cancer–related lymphedema risk is currently unknown and more research is required. In the interim, we would recommend vaccination away from the side of lymph node removal, either in the contralateral arm or in the thigh,” Ms. Juhel said.
The median duration of lymph node swelling was less than 1 week. In cases where lymph node swelling occurred after the first dose, 54.1% had swelling in ipsilateral axillary lymph nodes, and 45.9% in contralateral axillary lymph nodes. About 29.5% experienced swelling in ipsilateral supraclavicular lymph nodes, and 18.0% in contralateral supraclavicular lymph nodes.
Injection-site soreness, fatigue, GMS, headache, and chills occurred less often among older individuals (P < .001), and fatigue, muscle soreness, headache, and chills occurred more frequently after the second dose than the first (P < .001). The median duration of all side effects was 48 hours or less.
“The informed education that can be produced based on these results will hopefully ease the fears of women treated for breast cancer and empower them to make informed decisions regarding future vaccine doses,” Ms. Juhel said.
Ms. Juhel has no relevant financial disclosures.
Findings from the LymphVAX study recently presented at the San Antonio Breast Cancer Symposium show that relatively
Lymph node swelling can be a particularly troubling side effect, since it could be mistaken for breast cancer progression. In this study, of 621 women who received the first dose of an mRNA COVID-19 vaccine, 9.8% developed lymph node swelling as compared with 12.9% of 621 women who received the second dose, and 11.3% of 469 women who received the third dose. The findings were comparable to those of studies conducted of the general population, said study author Brooke C. Juhel, BS, a clinical research coordinator in the lymphedema research program at Massachusetts General Hospital and a student at Harvard Medical School, both in Boston. In the general population, 10.2% experienced lymph node swelling after the first dose and 14% after the second dose, according to the Centers for Disease Control and studies of the Pfizer and Moderna vaccines.
“This is consistent with the hypothesis that, after repeated vaccine doses, the immune system already has the antigens ready to fight the virus, thus the side effects may worsen as the immune response has increased,” she said. “Having screened over 6,500 women for breast cancer–related lymphedema, and with our patients reaching out with concerns about vaccine side effects, we were in a unique position to conduct this study.”
The study also confirmed that the most common side effects of receiving mRNA COVID-19 vaccines for women treated for breast cancer included injection site soreness, fatigue, muscle soreness, headache and chills lasting an average of 48 hours, which are symptoms comparable with those experienced by the general population.
“The side-effect profiles reported in this study for a cohort of women treated for breast cancer can be used to provide evidence-based patient education regarding future COVID-19 vaccine administration. The effect of the COVID-19 vaccines on breast cancer–related lymphedema risk is currently unknown and more research is required. In the interim, we would recommend vaccination away from the side of lymph node removal, either in the contralateral arm or in the thigh,” Ms. Juhel said.
The median duration of lymph node swelling was less than 1 week. In cases where lymph node swelling occurred after the first dose, 54.1% had swelling in ipsilateral axillary lymph nodes, and 45.9% in contralateral axillary lymph nodes. About 29.5% experienced swelling in ipsilateral supraclavicular lymph nodes, and 18.0% in contralateral supraclavicular lymph nodes.
Injection-site soreness, fatigue, GMS, headache, and chills occurred less often among older individuals (P < .001), and fatigue, muscle soreness, headache, and chills occurred more frequently after the second dose than the first (P < .001). The median duration of all side effects was 48 hours or less.
“The informed education that can be produced based on these results will hopefully ease the fears of women treated for breast cancer and empower them to make informed decisions regarding future vaccine doses,” Ms. Juhel said.
Ms. Juhel has no relevant financial disclosures.
Findings from the LymphVAX study recently presented at the San Antonio Breast Cancer Symposium show that relatively
Lymph node swelling can be a particularly troubling side effect, since it could be mistaken for breast cancer progression. In this study, of 621 women who received the first dose of an mRNA COVID-19 vaccine, 9.8% developed lymph node swelling as compared with 12.9% of 621 women who received the second dose, and 11.3% of 469 women who received the third dose. The findings were comparable to those of studies conducted of the general population, said study author Brooke C. Juhel, BS, a clinical research coordinator in the lymphedema research program at Massachusetts General Hospital and a student at Harvard Medical School, both in Boston. In the general population, 10.2% experienced lymph node swelling after the first dose and 14% after the second dose, according to the Centers for Disease Control and studies of the Pfizer and Moderna vaccines.
“This is consistent with the hypothesis that, after repeated vaccine doses, the immune system already has the antigens ready to fight the virus, thus the side effects may worsen as the immune response has increased,” she said. “Having screened over 6,500 women for breast cancer–related lymphedema, and with our patients reaching out with concerns about vaccine side effects, we were in a unique position to conduct this study.”
The study also confirmed that the most common side effects of receiving mRNA COVID-19 vaccines for women treated for breast cancer included injection site soreness, fatigue, muscle soreness, headache and chills lasting an average of 48 hours, which are symptoms comparable with those experienced by the general population.
“The side-effect profiles reported in this study for a cohort of women treated for breast cancer can be used to provide evidence-based patient education regarding future COVID-19 vaccine administration. The effect of the COVID-19 vaccines on breast cancer–related lymphedema risk is currently unknown and more research is required. In the interim, we would recommend vaccination away from the side of lymph node removal, either in the contralateral arm or in the thigh,” Ms. Juhel said.
The median duration of lymph node swelling was less than 1 week. In cases where lymph node swelling occurred after the first dose, 54.1% had swelling in ipsilateral axillary lymph nodes, and 45.9% in contralateral axillary lymph nodes. About 29.5% experienced swelling in ipsilateral supraclavicular lymph nodes, and 18.0% in contralateral supraclavicular lymph nodes.
Injection-site soreness, fatigue, GMS, headache, and chills occurred less often among older individuals (P < .001), and fatigue, muscle soreness, headache, and chills occurred more frequently after the second dose than the first (P < .001). The median duration of all side effects was 48 hours or less.
“The informed education that can be produced based on these results will hopefully ease the fears of women treated for breast cancer and empower them to make informed decisions regarding future vaccine doses,” Ms. Juhel said.
Ms. Juhel has no relevant financial disclosures.
FROM SABCS 2022
New test that detects 14 cancers focuses on sugars, not DNA
The leader in this field is the Galleri test (from GRAIL) which is already in clinical use in some health care networks across the United States. That test uses next-generation sequencing to analyze the arrangement of methyl groups on circulating tumor (or cell-free) DNA (cfDNA) in a blood sample.
The new test, under development by Swedish biotechnology company Elypta AB, has a different premise. It can detect 14 cancer types based on the analysis of glycosaminoglycans, which are a diverse group of polysaccharides that are altered by the presence of tumors. Using plasma and urine samples, the method had a 41.6%-62.3% sensitivity for detecting stage I cancer at 95% specificity.
In comparison, say the authors, other assays have reported 39%-73% sensitivity to stage I cancers, but these estimates are usually limited to 12 cancer types that are considered “high-signal,” and the assays perform poorly in cancers that emit little cfDNA, such as genitourinary and brain malignancies.
“The main advantage of glycosaminoglycans appears to be that they change in the blood and urine at the earliest stages of cancer,” said study author Francesco Gatto, PhD, founder and chief scientific officer at Elypta. “Consequently, this method showed an impressive detection rate in stage I compared to other emerging methods.”
The study was published online in Proceedings of the National Academy of Sciences.
Combine tests?
Dr. Gatto commented that he “could envision that one day we may be able to combine these methods.”
“The same blood specimen could be used to test both glycosaminoglycans and genomic biomarkers,” said Dr. Gatto. “This strategy could hopefully detect even more cancers than with either method alone, and the resulting performance may well be sufficient as a one-stop-shop screening program.”
So how does the new test from Elypta compare with the Galleri test?
“Galleri and similar methods mostly focused on information coming from molecules of DNA naturally floating in the blood,” explained Dr. Gatto. “It makes sense to conduct research there because cancers typically start with events in the DNA.”
He noted that the current study explored a new layer of information, molecules called glycosaminoglycans, that participate in the metabolism of cancer.
“This method detected many cancers that the previous methods missed, and a substantial proportion of these were at stage I,” said Dr. Gatto. “Cancer is a complex disease, so the most layers of information we can probe noninvasively, say with a blood test, the more likely we can catch more cancers at its earliest stage.”
Other platforms typically rely on sequencing and detecting cancer-derived fractions of cfDNA, but these methods have challenges that can interfere with their usage. For example, some cancer types do not shed sufficient cfDNA and it cannot be accurately measured.
“An advantage on focusing on glycosaminoglycans is that the method does not require next-generation sequencing or similarly complex assays because glycosaminoglycans are informative with less than 10 simultaneous measurements as opposed to Galleri that looks at over 1 million DNA methylation sites,” he said.
“This makes the assay behind the test much cheaper and robust – we estimated a 5-10 times lower cost difference,” Dr. Gatto said.
Prospective and comparative data needed
In a comment, Eric Klein, MD, emeritus chair of the Glickman Urological and Kidney Institute at the Cleveland Clinic explained that the “only accurate way to know how a test will perform in an intended-use population is to actually test it in that population. It’s not possible to extrapolate results directly from a case-control study.”
Cancers shed many different biologic markers into body fluids, but which of these signals will be best to serve as the basis of an MCED (multi-cancer detection test) that has clinical utility in a screening population has yet to be determined, he noted. “And it’s possible that no single test will be optimum for every clinical situation.”
“The results of this study appear promising, but it is not possible to claim superiority of one test over another based on individual case-control studies because of uncontrolled differences in the selected populations,” Dr. Klein continued. “The only scientifically accurate way to do this is to perform different tests on the same patient samples in a head-to-head comparison.”
There is only one study that he is aware of that has done this recently, in which multiple different assays looking at various signals in cell-free DNA were directly compared on the same samples (Cancer cell. 2022;40:1537-49.e12). “A targeted methylation assay that is the basis for Galleri was best for the lowest limit of detection and for predicting cancer site of origin,” said Dr. Klein.
Another expert agreed that a direct head-to-head study is needed to compare assays. “Based on this data, you cannot say that this method is better than the other one because that requires a comparative study,” said Fred Hirsch, MD, PhD, executive director of the Center for Thoracic Oncology, Tisch Cancer Institute at Mount Sinai, New York.
Metabolomics is interesting, and the data are encouraging, he continued. “But this is a multicancer early detection test and metabolism changes may vary from cancer type to cancer type. I’m not sure that the metabolism of lung cancer is the same as that of a gynecologic cancer.”
Dr. Hirsch also pointed out that there could also be confounding factors. “They have excluded inflammatory disease, but there can be other variables such as smoking,” he said. “Overall it gives some interesting perspectives but I would like to see more prospective validation and studies in specific disease groups, and eventually comparative studies with other methodologies.”
Study details
The authors evaluated if plasma and urine free GAGomes (free glycosaminoglycan profiles) deviated from baseline physiological levels in 14 cancer types and could serve as metabolic cancer biomarkers. They also then validated using free GAGomes for MCED in an external population with 2,064 samples obtained from 1,260 patients with cancer and healthy individuals.
In an in vivo cancer progression model, they observed widespread cancer-specific changes in biofluidic free GAGomes and then developed three machine-learning models based on urine (nurine = 220 cancer vs. 360 healthy) and plasma (nplasma = 517 cancer vs. 425 healthy) free GAGomes that were able to detect any cancer with an area under the receiver operating characteristic curve of 0.83-0.93 (with up to 62% sensitivity to stage I disease at 95% specificity).
To assess if altered GAGome features associated with cancer suggested more aggressive tumor biology, they correlated each score with overall survival. The median follow-up time was 17 months in the plasma cohort (n = 370 across 13 cancer types), 15 months in the urine cohort (n = 162 across 4 cancer types), and 15 months in the combined cohort (n = 152 across 4 cancer types).
They found that all three scores independently predicted overall survival in a multivariable analysis (hazard ratio, 1.29; P = .0009 for plasma; HR, 1.79; P = .0009 for urine; HR, 1.91; P = .0004 for combined) after adjusting for cancer type, age, sex, and stage IV or high-grade disease.
These findings showed an association of free GAGome alterations with aggressive cancer phenotypes and suggested that scores below the 95% specificity cutoff might have a better prognosis, the authors comment.
In addition, other analyses showed that free GAGomes predicted the putative cancer location with 89% accuracy. And finally, to confirm whether the free GAGome MCED scores could be used for screening, a validation analysis was conducted using a typical “screening population,” which requires at least 99% specificity. The combined free GAGomes were able to predict a poor prognosis of any cancer type within 18 months and with 43% sensitivity (21% in stage I; n = 121 and 49 cases).
Dr. Gatto believes that these results, as well as those from other studies looking at glycosaminoglycans as cancer biomarkers, will lead to the next steps of development. “But I speculate that this test could be most useful to assess in a cheap, practical, and noninvasive manner if a person at increased risk of cancer should be selected for cancer screening as part of established or emerging screening programs.”
The study was sponsored by Elypta. Dr. Gatto is listed as an inventor in patent applications related to the biomarkers described in this study and later assigned to Elypta, and is a shareholder and employed at Elypta. Dr. Hirsch reports no relevant financial relationships. Dr. Klein is a consultant for GRAIL and an investigator for CCGA and Pathfinder.
A version of this article first appeared on Medscape.com.
The leader in this field is the Galleri test (from GRAIL) which is already in clinical use in some health care networks across the United States. That test uses next-generation sequencing to analyze the arrangement of methyl groups on circulating tumor (or cell-free) DNA (cfDNA) in a blood sample.
The new test, under development by Swedish biotechnology company Elypta AB, has a different premise. It can detect 14 cancer types based on the analysis of glycosaminoglycans, which are a diverse group of polysaccharides that are altered by the presence of tumors. Using plasma and urine samples, the method had a 41.6%-62.3% sensitivity for detecting stage I cancer at 95% specificity.
In comparison, say the authors, other assays have reported 39%-73% sensitivity to stage I cancers, but these estimates are usually limited to 12 cancer types that are considered “high-signal,” and the assays perform poorly in cancers that emit little cfDNA, such as genitourinary and brain malignancies.
“The main advantage of glycosaminoglycans appears to be that they change in the blood and urine at the earliest stages of cancer,” said study author Francesco Gatto, PhD, founder and chief scientific officer at Elypta. “Consequently, this method showed an impressive detection rate in stage I compared to other emerging methods.”
The study was published online in Proceedings of the National Academy of Sciences.
Combine tests?
Dr. Gatto commented that he “could envision that one day we may be able to combine these methods.”
“The same blood specimen could be used to test both glycosaminoglycans and genomic biomarkers,” said Dr. Gatto. “This strategy could hopefully detect even more cancers than with either method alone, and the resulting performance may well be sufficient as a one-stop-shop screening program.”
So how does the new test from Elypta compare with the Galleri test?
“Galleri and similar methods mostly focused on information coming from molecules of DNA naturally floating in the blood,” explained Dr. Gatto. “It makes sense to conduct research there because cancers typically start with events in the DNA.”
He noted that the current study explored a new layer of information, molecules called glycosaminoglycans, that participate in the metabolism of cancer.
“This method detected many cancers that the previous methods missed, and a substantial proportion of these were at stage I,” said Dr. Gatto. “Cancer is a complex disease, so the most layers of information we can probe noninvasively, say with a blood test, the more likely we can catch more cancers at its earliest stage.”
Other platforms typically rely on sequencing and detecting cancer-derived fractions of cfDNA, but these methods have challenges that can interfere with their usage. For example, some cancer types do not shed sufficient cfDNA and it cannot be accurately measured.
“An advantage on focusing on glycosaminoglycans is that the method does not require next-generation sequencing or similarly complex assays because glycosaminoglycans are informative with less than 10 simultaneous measurements as opposed to Galleri that looks at over 1 million DNA methylation sites,” he said.
“This makes the assay behind the test much cheaper and robust – we estimated a 5-10 times lower cost difference,” Dr. Gatto said.
Prospective and comparative data needed
In a comment, Eric Klein, MD, emeritus chair of the Glickman Urological and Kidney Institute at the Cleveland Clinic explained that the “only accurate way to know how a test will perform in an intended-use population is to actually test it in that population. It’s not possible to extrapolate results directly from a case-control study.”
Cancers shed many different biologic markers into body fluids, but which of these signals will be best to serve as the basis of an MCED (multi-cancer detection test) that has clinical utility in a screening population has yet to be determined, he noted. “And it’s possible that no single test will be optimum for every clinical situation.”
“The results of this study appear promising, but it is not possible to claim superiority of one test over another based on individual case-control studies because of uncontrolled differences in the selected populations,” Dr. Klein continued. “The only scientifically accurate way to do this is to perform different tests on the same patient samples in a head-to-head comparison.”
There is only one study that he is aware of that has done this recently, in which multiple different assays looking at various signals in cell-free DNA were directly compared on the same samples (Cancer cell. 2022;40:1537-49.e12). “A targeted methylation assay that is the basis for Galleri was best for the lowest limit of detection and for predicting cancer site of origin,” said Dr. Klein.
Another expert agreed that a direct head-to-head study is needed to compare assays. “Based on this data, you cannot say that this method is better than the other one because that requires a comparative study,” said Fred Hirsch, MD, PhD, executive director of the Center for Thoracic Oncology, Tisch Cancer Institute at Mount Sinai, New York.
Metabolomics is interesting, and the data are encouraging, he continued. “But this is a multicancer early detection test and metabolism changes may vary from cancer type to cancer type. I’m not sure that the metabolism of lung cancer is the same as that of a gynecologic cancer.”
Dr. Hirsch also pointed out that there could also be confounding factors. “They have excluded inflammatory disease, but there can be other variables such as smoking,” he said. “Overall it gives some interesting perspectives but I would like to see more prospective validation and studies in specific disease groups, and eventually comparative studies with other methodologies.”
Study details
The authors evaluated if plasma and urine free GAGomes (free glycosaminoglycan profiles) deviated from baseline physiological levels in 14 cancer types and could serve as metabolic cancer biomarkers. They also then validated using free GAGomes for MCED in an external population with 2,064 samples obtained from 1,260 patients with cancer and healthy individuals.
In an in vivo cancer progression model, they observed widespread cancer-specific changes in biofluidic free GAGomes and then developed three machine-learning models based on urine (nurine = 220 cancer vs. 360 healthy) and plasma (nplasma = 517 cancer vs. 425 healthy) free GAGomes that were able to detect any cancer with an area under the receiver operating characteristic curve of 0.83-0.93 (with up to 62% sensitivity to stage I disease at 95% specificity).
To assess if altered GAGome features associated with cancer suggested more aggressive tumor biology, they correlated each score with overall survival. The median follow-up time was 17 months in the plasma cohort (n = 370 across 13 cancer types), 15 months in the urine cohort (n = 162 across 4 cancer types), and 15 months in the combined cohort (n = 152 across 4 cancer types).
They found that all three scores independently predicted overall survival in a multivariable analysis (hazard ratio, 1.29; P = .0009 for plasma; HR, 1.79; P = .0009 for urine; HR, 1.91; P = .0004 for combined) after adjusting for cancer type, age, sex, and stage IV or high-grade disease.
These findings showed an association of free GAGome alterations with aggressive cancer phenotypes and suggested that scores below the 95% specificity cutoff might have a better prognosis, the authors comment.
In addition, other analyses showed that free GAGomes predicted the putative cancer location with 89% accuracy. And finally, to confirm whether the free GAGome MCED scores could be used for screening, a validation analysis was conducted using a typical “screening population,” which requires at least 99% specificity. The combined free GAGomes were able to predict a poor prognosis of any cancer type within 18 months and with 43% sensitivity (21% in stage I; n = 121 and 49 cases).
Dr. Gatto believes that these results, as well as those from other studies looking at glycosaminoglycans as cancer biomarkers, will lead to the next steps of development. “But I speculate that this test could be most useful to assess in a cheap, practical, and noninvasive manner if a person at increased risk of cancer should be selected for cancer screening as part of established or emerging screening programs.”
The study was sponsored by Elypta. Dr. Gatto is listed as an inventor in patent applications related to the biomarkers described in this study and later assigned to Elypta, and is a shareholder and employed at Elypta. Dr. Hirsch reports no relevant financial relationships. Dr. Klein is a consultant for GRAIL and an investigator for CCGA and Pathfinder.
A version of this article first appeared on Medscape.com.
The leader in this field is the Galleri test (from GRAIL) which is already in clinical use in some health care networks across the United States. That test uses next-generation sequencing to analyze the arrangement of methyl groups on circulating tumor (or cell-free) DNA (cfDNA) in a blood sample.
The new test, under development by Swedish biotechnology company Elypta AB, has a different premise. It can detect 14 cancer types based on the analysis of glycosaminoglycans, which are a diverse group of polysaccharides that are altered by the presence of tumors. Using plasma and urine samples, the method had a 41.6%-62.3% sensitivity for detecting stage I cancer at 95% specificity.
In comparison, say the authors, other assays have reported 39%-73% sensitivity to stage I cancers, but these estimates are usually limited to 12 cancer types that are considered “high-signal,” and the assays perform poorly in cancers that emit little cfDNA, such as genitourinary and brain malignancies.
“The main advantage of glycosaminoglycans appears to be that they change in the blood and urine at the earliest stages of cancer,” said study author Francesco Gatto, PhD, founder and chief scientific officer at Elypta. “Consequently, this method showed an impressive detection rate in stage I compared to other emerging methods.”
The study was published online in Proceedings of the National Academy of Sciences.
Combine tests?
Dr. Gatto commented that he “could envision that one day we may be able to combine these methods.”
“The same blood specimen could be used to test both glycosaminoglycans and genomic biomarkers,” said Dr. Gatto. “This strategy could hopefully detect even more cancers than with either method alone, and the resulting performance may well be sufficient as a one-stop-shop screening program.”
So how does the new test from Elypta compare with the Galleri test?
“Galleri and similar methods mostly focused on information coming from molecules of DNA naturally floating in the blood,” explained Dr. Gatto. “It makes sense to conduct research there because cancers typically start with events in the DNA.”
He noted that the current study explored a new layer of information, molecules called glycosaminoglycans, that participate in the metabolism of cancer.
“This method detected many cancers that the previous methods missed, and a substantial proportion of these were at stage I,” said Dr. Gatto. “Cancer is a complex disease, so the most layers of information we can probe noninvasively, say with a blood test, the more likely we can catch more cancers at its earliest stage.”
Other platforms typically rely on sequencing and detecting cancer-derived fractions of cfDNA, but these methods have challenges that can interfere with their usage. For example, some cancer types do not shed sufficient cfDNA and it cannot be accurately measured.
“An advantage on focusing on glycosaminoglycans is that the method does not require next-generation sequencing or similarly complex assays because glycosaminoglycans are informative with less than 10 simultaneous measurements as opposed to Galleri that looks at over 1 million DNA methylation sites,” he said.
“This makes the assay behind the test much cheaper and robust – we estimated a 5-10 times lower cost difference,” Dr. Gatto said.
Prospective and comparative data needed
In a comment, Eric Klein, MD, emeritus chair of the Glickman Urological and Kidney Institute at the Cleveland Clinic explained that the “only accurate way to know how a test will perform in an intended-use population is to actually test it in that population. It’s not possible to extrapolate results directly from a case-control study.”
Cancers shed many different biologic markers into body fluids, but which of these signals will be best to serve as the basis of an MCED (multi-cancer detection test) that has clinical utility in a screening population has yet to be determined, he noted. “And it’s possible that no single test will be optimum for every clinical situation.”
“The results of this study appear promising, but it is not possible to claim superiority of one test over another based on individual case-control studies because of uncontrolled differences in the selected populations,” Dr. Klein continued. “The only scientifically accurate way to do this is to perform different tests on the same patient samples in a head-to-head comparison.”
There is only one study that he is aware of that has done this recently, in which multiple different assays looking at various signals in cell-free DNA were directly compared on the same samples (Cancer cell. 2022;40:1537-49.e12). “A targeted methylation assay that is the basis for Galleri was best for the lowest limit of detection and for predicting cancer site of origin,” said Dr. Klein.
Another expert agreed that a direct head-to-head study is needed to compare assays. “Based on this data, you cannot say that this method is better than the other one because that requires a comparative study,” said Fred Hirsch, MD, PhD, executive director of the Center for Thoracic Oncology, Tisch Cancer Institute at Mount Sinai, New York.
Metabolomics is interesting, and the data are encouraging, he continued. “But this is a multicancer early detection test and metabolism changes may vary from cancer type to cancer type. I’m not sure that the metabolism of lung cancer is the same as that of a gynecologic cancer.”
Dr. Hirsch also pointed out that there could also be confounding factors. “They have excluded inflammatory disease, but there can be other variables such as smoking,” he said. “Overall it gives some interesting perspectives but I would like to see more prospective validation and studies in specific disease groups, and eventually comparative studies with other methodologies.”
Study details
The authors evaluated if plasma and urine free GAGomes (free glycosaminoglycan profiles) deviated from baseline physiological levels in 14 cancer types and could serve as metabolic cancer biomarkers. They also then validated using free GAGomes for MCED in an external population with 2,064 samples obtained from 1,260 patients with cancer and healthy individuals.
In an in vivo cancer progression model, they observed widespread cancer-specific changes in biofluidic free GAGomes and then developed three machine-learning models based on urine (nurine = 220 cancer vs. 360 healthy) and plasma (nplasma = 517 cancer vs. 425 healthy) free GAGomes that were able to detect any cancer with an area under the receiver operating characteristic curve of 0.83-0.93 (with up to 62% sensitivity to stage I disease at 95% specificity).
To assess if altered GAGome features associated with cancer suggested more aggressive tumor biology, they correlated each score with overall survival. The median follow-up time was 17 months in the plasma cohort (n = 370 across 13 cancer types), 15 months in the urine cohort (n = 162 across 4 cancer types), and 15 months in the combined cohort (n = 152 across 4 cancer types).
They found that all three scores independently predicted overall survival in a multivariable analysis (hazard ratio, 1.29; P = .0009 for plasma; HR, 1.79; P = .0009 for urine; HR, 1.91; P = .0004 for combined) after adjusting for cancer type, age, sex, and stage IV or high-grade disease.
These findings showed an association of free GAGome alterations with aggressive cancer phenotypes and suggested that scores below the 95% specificity cutoff might have a better prognosis, the authors comment.
In addition, other analyses showed that free GAGomes predicted the putative cancer location with 89% accuracy. And finally, to confirm whether the free GAGome MCED scores could be used for screening, a validation analysis was conducted using a typical “screening population,” which requires at least 99% specificity. The combined free GAGomes were able to predict a poor prognosis of any cancer type within 18 months and with 43% sensitivity (21% in stage I; n = 121 and 49 cases).
Dr. Gatto believes that these results, as well as those from other studies looking at glycosaminoglycans as cancer biomarkers, will lead to the next steps of development. “But I speculate that this test could be most useful to assess in a cheap, practical, and noninvasive manner if a person at increased risk of cancer should be selected for cancer screening as part of established or emerging screening programs.”
The study was sponsored by Elypta. Dr. Gatto is listed as an inventor in patent applications related to the biomarkers described in this study and later assigned to Elypta, and is a shareholder and employed at Elypta. Dr. Hirsch reports no relevant financial relationships. Dr. Klein is a consultant for GRAIL and an investigator for CCGA and Pathfinder.
A version of this article first appeared on Medscape.com.
FROM PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES
Exciting advances in HR-positive breast cancer: Top five picks from SABCS
SAN ANTONIO –
This news organization spoke with SABCS program director Virginia Kaklamani, MD, leader of the Breast Cancer Program at UT Health, San Antonio, and Jason A. Mouabbi, MD, of the University of Texas MD Anderson Cancer Center, Houston, about their top five picks on HR-positive disease – the research they were most excited about and what the findings could mean for clinical practice and patient outcomes.
1. Addressing an unmet need
Data from the phase 3 CAPItello-291 clinical trial showed that the addition of the investigational AKT inhibitor capivasertib to fulvestrant resulted in statistically significant and clinically meaningful improvement in progression-free survival (PFS) among 708 patients with HR-positive, HER2-negative advanced breast cancer, compared with those who received placebo plus fulvestrant (GS3-04).
For patients treated with capivasertib plus fulvestrant, median PFS was 7.2 months, compared with 3.6 months for those who received placebo plus fulvestrant (hazard ratio, 0.60). Among patients assigned to the capivasertib group, 41% had tumors with AKT pathway mutations. In this group, the median PFS was 7.3 months vs. 3.1 months in the placebo cohort. The objective response rate among patients with measurable disease was 23% overall in the capivasertib group, compared with 12.2% in the placebo arm; it was 28.8% vs. 9.7% among the patients with AKT alterations.
Dr. Mouabbi noted that the study “met its primary endpoint” and that, importantly, it “addresses an area of unmet need.”
“The study’s treatment targets the PI3K/AKT/mTOR pathway, which is a very active pathway in hormone-positive metastatic breast cancer,” Dr. Mouabbi explained. He noted, “We’ve always wanted to tackle that pathway effectively, and it looks like this drug can do that.”
2. Next-generation SERD
Data from the phase 2 SERENA-2 trial offers evidence that camizestrant, a next-generation selective estrogen-receptor degrader (SERD), improved PFS compared with fulvestrant for patients with HR-positive, HER2-negative breast cancer (GS3-02).
Overall, 240 patients were randomly assigned to receive camizestrant monotherapy at various doses or fulvestrant at 500 mg. Among patients who received camizestrant 75 mg, median PFS was 7.2 months; among those who received camizestrant 150 mg, PFS was slightly longer, at 7.7 months vs. 3.7 months for patients treated with fulvestrant. Compared with fulvestrant, camizestrant reduced the risk of disease progression by 42% at 75 mg (HR, 0.58) and by 33% at 150 mg (HR, 0.67). In a subgroup of patients with ESR1 mutations, camizestrant reduced the risk of disease progression by 67% in the group that received 75 mg and by 45% in the group that received 150 mg, compared with fulvestrant (median PFS, 6.3, 9.2, and 2.2 months, respectively).
“In this trial, camizestrant looks like a more beneficial treatment in the target group,” said Dr. Kaklamani. “This is significant because it means that camizestrant could be used in the future in HR-positive metastatic breast cancer instead of fulvestrant.” In addition, “camizestrant is taken orally and is much more convenient for patients, unlike fulvestrant, which is taken intramuscularly.”
3. Pregnancy risks
Can endocrine therapy be safely interrupted for women with breast cancer who wish to become pregnant? That’s what researchers tried to glean in a recent prospective trial presented at the meeting (GS4-09).
The study enrolled over 500 women for whom endocrine therapy had been stopped in the hopes of their becoming pregnant. Almost all (93.4%) had stage I/II HR-positive breast cancer. The primary objective was to determine the risk of breast cancer relapse associated with interrupting therapy for about 2 years. The authors defined no more than 46 breast cancer–free interval (BCFI) events as the safety threshold. A BCFI event was defined as local, regional, or distant recurrence or a new invasive contralateral breast cancer.
Among 497 women, 368 (74%) had at least one pregnancy and 317 (64%) had at least one live birth, for a total of 365 babies born. At a median follow-up of 41 months, 44 participants experienced a BCFI event, in line with the safety threshold. The 3-year BCFI failure rate was 8.9%, similar to the 9.2% rate in an external control cohort from the SOFT/TEXT trials. In addition, 76.3% of patients resumed endocrine therapy; 15.4% had not yet resumed therapy.
“This trial is more confirmatory but an extremely important step for young women who want to get pregnant after diagnosis and recovery from HR-positive breast cancer,” Dr. Kaklamani said. “It seems that stopping endocrine therapy to become pregnant did not cause any adverse outcomes or increase the risk of reoccurrence of cancer in the women in the study.”
Dr. Mouabbi agreed, noting, “Many of our patients are afraid that they will miss the window to get pregnant because they have to be on treatment for so long. This is the first study that let us know pregnancy and safety outcomes in patients who took a break from endocrine therapy to get pregnant. The results are promising and will be exciting for many of our patients.”
4. Assay identifies OFS benefit
A genomic assay was able to distinguish premenopausal patients with early-stage HR-positive breast cancer who benefited from the addition of ovarian function suppression (OFS) to adjuvant endocrine therapy, according to new data presented at the meeting (GS1-06).
In the study, investigators analyzed 1,717 patient tumor samples from the landmark Suppression of Ovarian Function Trial (SOFT) trial. The Breast Cancer Index identified 58% of women who benefited from the addition of ovarian function suppression to tamoxifen or exemestane therapy. They experienced an absolute benefit of 11.6% (42% did not benefit), compared with those with received tamoxifen alone. The predictive benefit was observed regardless of age, lymph node involvement, and receipt of chemotherapy.
Dr. Kaklamani highlighted this study’s importance, saying, “Ovarian suppression is associated with severe adverse events for patients. Obviously, the women who will get a benefit should continue, but this research is important because it will hopefully show us who to recommend ovarian suppression to while not exposing patients who are likely to get little benefit to unneeded toxicity.”
5. Optimizing elacestrant PFS
Last year, data from the Emerald trial showed that elacestrant is superior to standard-of-care therapy for HR-positive metastatic breast cancer. An update that Dr. Kaklamani presented at SABCS (GS3-01) explored whether the duration of a prior CDK4/6 inhibitor affects PFS.
The study was a randomized, open-label, phase 3 trial in which 478 patients with ER-positive/HER2-negative metastatic breast cancer received either elacestrant or standard of care. These patients had previously received one or two lines of endocrine therapy, a CDK4/6 inhibitor, and ≤ 1 line of chemotherapy.
Overall, the duration of prior CDK4/6 inhibitor in the metastatic setting was positively associated with PFS – the longer the duration of prior CDK4/6 inhibitor therapy, the longer the PFS with elacestrant. PFS outcomes were even stronger among patients with ESR1 mutations.
“What we found was that the women who benefit most from elacestrant had previously received a CDK4/6 inhibitor for at least 6 months,” Dr. Kaklamani said. These data can help us determine who may do best on the drug, she added.
A version of this article first appeared on Medscape.com.
SAN ANTONIO –
This news organization spoke with SABCS program director Virginia Kaklamani, MD, leader of the Breast Cancer Program at UT Health, San Antonio, and Jason A. Mouabbi, MD, of the University of Texas MD Anderson Cancer Center, Houston, about their top five picks on HR-positive disease – the research they were most excited about and what the findings could mean for clinical practice and patient outcomes.
1. Addressing an unmet need
Data from the phase 3 CAPItello-291 clinical trial showed that the addition of the investigational AKT inhibitor capivasertib to fulvestrant resulted in statistically significant and clinically meaningful improvement in progression-free survival (PFS) among 708 patients with HR-positive, HER2-negative advanced breast cancer, compared with those who received placebo plus fulvestrant (GS3-04).
For patients treated with capivasertib plus fulvestrant, median PFS was 7.2 months, compared with 3.6 months for those who received placebo plus fulvestrant (hazard ratio, 0.60). Among patients assigned to the capivasertib group, 41% had tumors with AKT pathway mutations. In this group, the median PFS was 7.3 months vs. 3.1 months in the placebo cohort. The objective response rate among patients with measurable disease was 23% overall in the capivasertib group, compared with 12.2% in the placebo arm; it was 28.8% vs. 9.7% among the patients with AKT alterations.
Dr. Mouabbi noted that the study “met its primary endpoint” and that, importantly, it “addresses an area of unmet need.”
“The study’s treatment targets the PI3K/AKT/mTOR pathway, which is a very active pathway in hormone-positive metastatic breast cancer,” Dr. Mouabbi explained. He noted, “We’ve always wanted to tackle that pathway effectively, and it looks like this drug can do that.”
2. Next-generation SERD
Data from the phase 2 SERENA-2 trial offers evidence that camizestrant, a next-generation selective estrogen-receptor degrader (SERD), improved PFS compared with fulvestrant for patients with HR-positive, HER2-negative breast cancer (GS3-02).
Overall, 240 patients were randomly assigned to receive camizestrant monotherapy at various doses or fulvestrant at 500 mg. Among patients who received camizestrant 75 mg, median PFS was 7.2 months; among those who received camizestrant 150 mg, PFS was slightly longer, at 7.7 months vs. 3.7 months for patients treated with fulvestrant. Compared with fulvestrant, camizestrant reduced the risk of disease progression by 42% at 75 mg (HR, 0.58) and by 33% at 150 mg (HR, 0.67). In a subgroup of patients with ESR1 mutations, camizestrant reduced the risk of disease progression by 67% in the group that received 75 mg and by 45% in the group that received 150 mg, compared with fulvestrant (median PFS, 6.3, 9.2, and 2.2 months, respectively).
“In this trial, camizestrant looks like a more beneficial treatment in the target group,” said Dr. Kaklamani. “This is significant because it means that camizestrant could be used in the future in HR-positive metastatic breast cancer instead of fulvestrant.” In addition, “camizestrant is taken orally and is much more convenient for patients, unlike fulvestrant, which is taken intramuscularly.”
3. Pregnancy risks
Can endocrine therapy be safely interrupted for women with breast cancer who wish to become pregnant? That’s what researchers tried to glean in a recent prospective trial presented at the meeting (GS4-09).
The study enrolled over 500 women for whom endocrine therapy had been stopped in the hopes of their becoming pregnant. Almost all (93.4%) had stage I/II HR-positive breast cancer. The primary objective was to determine the risk of breast cancer relapse associated with interrupting therapy for about 2 years. The authors defined no more than 46 breast cancer–free interval (BCFI) events as the safety threshold. A BCFI event was defined as local, regional, or distant recurrence or a new invasive contralateral breast cancer.
Among 497 women, 368 (74%) had at least one pregnancy and 317 (64%) had at least one live birth, for a total of 365 babies born. At a median follow-up of 41 months, 44 participants experienced a BCFI event, in line with the safety threshold. The 3-year BCFI failure rate was 8.9%, similar to the 9.2% rate in an external control cohort from the SOFT/TEXT trials. In addition, 76.3% of patients resumed endocrine therapy; 15.4% had not yet resumed therapy.
“This trial is more confirmatory but an extremely important step for young women who want to get pregnant after diagnosis and recovery from HR-positive breast cancer,” Dr. Kaklamani said. “It seems that stopping endocrine therapy to become pregnant did not cause any adverse outcomes or increase the risk of reoccurrence of cancer in the women in the study.”
Dr. Mouabbi agreed, noting, “Many of our patients are afraid that they will miss the window to get pregnant because they have to be on treatment for so long. This is the first study that let us know pregnancy and safety outcomes in patients who took a break from endocrine therapy to get pregnant. The results are promising and will be exciting for many of our patients.”
4. Assay identifies OFS benefit
A genomic assay was able to distinguish premenopausal patients with early-stage HR-positive breast cancer who benefited from the addition of ovarian function suppression (OFS) to adjuvant endocrine therapy, according to new data presented at the meeting (GS1-06).
In the study, investigators analyzed 1,717 patient tumor samples from the landmark Suppression of Ovarian Function Trial (SOFT) trial. The Breast Cancer Index identified 58% of women who benefited from the addition of ovarian function suppression to tamoxifen or exemestane therapy. They experienced an absolute benefit of 11.6% (42% did not benefit), compared with those with received tamoxifen alone. The predictive benefit was observed regardless of age, lymph node involvement, and receipt of chemotherapy.
Dr. Kaklamani highlighted this study’s importance, saying, “Ovarian suppression is associated with severe adverse events for patients. Obviously, the women who will get a benefit should continue, but this research is important because it will hopefully show us who to recommend ovarian suppression to while not exposing patients who are likely to get little benefit to unneeded toxicity.”
5. Optimizing elacestrant PFS
Last year, data from the Emerald trial showed that elacestrant is superior to standard-of-care therapy for HR-positive metastatic breast cancer. An update that Dr. Kaklamani presented at SABCS (GS3-01) explored whether the duration of a prior CDK4/6 inhibitor affects PFS.
The study was a randomized, open-label, phase 3 trial in which 478 patients with ER-positive/HER2-negative metastatic breast cancer received either elacestrant or standard of care. These patients had previously received one or two lines of endocrine therapy, a CDK4/6 inhibitor, and ≤ 1 line of chemotherapy.
Overall, the duration of prior CDK4/6 inhibitor in the metastatic setting was positively associated with PFS – the longer the duration of prior CDK4/6 inhibitor therapy, the longer the PFS with elacestrant. PFS outcomes were even stronger among patients with ESR1 mutations.
“What we found was that the women who benefit most from elacestrant had previously received a CDK4/6 inhibitor for at least 6 months,” Dr. Kaklamani said. These data can help us determine who may do best on the drug, she added.
A version of this article first appeared on Medscape.com.
SAN ANTONIO –
This news organization spoke with SABCS program director Virginia Kaklamani, MD, leader of the Breast Cancer Program at UT Health, San Antonio, and Jason A. Mouabbi, MD, of the University of Texas MD Anderson Cancer Center, Houston, about their top five picks on HR-positive disease – the research they were most excited about and what the findings could mean for clinical practice and patient outcomes.
1. Addressing an unmet need
Data from the phase 3 CAPItello-291 clinical trial showed that the addition of the investigational AKT inhibitor capivasertib to fulvestrant resulted in statistically significant and clinically meaningful improvement in progression-free survival (PFS) among 708 patients with HR-positive, HER2-negative advanced breast cancer, compared with those who received placebo plus fulvestrant (GS3-04).
For patients treated with capivasertib plus fulvestrant, median PFS was 7.2 months, compared with 3.6 months for those who received placebo plus fulvestrant (hazard ratio, 0.60). Among patients assigned to the capivasertib group, 41% had tumors with AKT pathway mutations. In this group, the median PFS was 7.3 months vs. 3.1 months in the placebo cohort. The objective response rate among patients with measurable disease was 23% overall in the capivasertib group, compared with 12.2% in the placebo arm; it was 28.8% vs. 9.7% among the patients with AKT alterations.
Dr. Mouabbi noted that the study “met its primary endpoint” and that, importantly, it “addresses an area of unmet need.”
“The study’s treatment targets the PI3K/AKT/mTOR pathway, which is a very active pathway in hormone-positive metastatic breast cancer,” Dr. Mouabbi explained. He noted, “We’ve always wanted to tackle that pathway effectively, and it looks like this drug can do that.”
2. Next-generation SERD
Data from the phase 2 SERENA-2 trial offers evidence that camizestrant, a next-generation selective estrogen-receptor degrader (SERD), improved PFS compared with fulvestrant for patients with HR-positive, HER2-negative breast cancer (GS3-02).
Overall, 240 patients were randomly assigned to receive camizestrant monotherapy at various doses or fulvestrant at 500 mg. Among patients who received camizestrant 75 mg, median PFS was 7.2 months; among those who received camizestrant 150 mg, PFS was slightly longer, at 7.7 months vs. 3.7 months for patients treated with fulvestrant. Compared with fulvestrant, camizestrant reduced the risk of disease progression by 42% at 75 mg (HR, 0.58) and by 33% at 150 mg (HR, 0.67). In a subgroup of patients with ESR1 mutations, camizestrant reduced the risk of disease progression by 67% in the group that received 75 mg and by 45% in the group that received 150 mg, compared with fulvestrant (median PFS, 6.3, 9.2, and 2.2 months, respectively).
“In this trial, camizestrant looks like a more beneficial treatment in the target group,” said Dr. Kaklamani. “This is significant because it means that camizestrant could be used in the future in HR-positive metastatic breast cancer instead of fulvestrant.” In addition, “camizestrant is taken orally and is much more convenient for patients, unlike fulvestrant, which is taken intramuscularly.”
3. Pregnancy risks
Can endocrine therapy be safely interrupted for women with breast cancer who wish to become pregnant? That’s what researchers tried to glean in a recent prospective trial presented at the meeting (GS4-09).
The study enrolled over 500 women for whom endocrine therapy had been stopped in the hopes of their becoming pregnant. Almost all (93.4%) had stage I/II HR-positive breast cancer. The primary objective was to determine the risk of breast cancer relapse associated with interrupting therapy for about 2 years. The authors defined no more than 46 breast cancer–free interval (BCFI) events as the safety threshold. A BCFI event was defined as local, regional, or distant recurrence or a new invasive contralateral breast cancer.
Among 497 women, 368 (74%) had at least one pregnancy and 317 (64%) had at least one live birth, for a total of 365 babies born. At a median follow-up of 41 months, 44 participants experienced a BCFI event, in line with the safety threshold. The 3-year BCFI failure rate was 8.9%, similar to the 9.2% rate in an external control cohort from the SOFT/TEXT trials. In addition, 76.3% of patients resumed endocrine therapy; 15.4% had not yet resumed therapy.
“This trial is more confirmatory but an extremely important step for young women who want to get pregnant after diagnosis and recovery from HR-positive breast cancer,” Dr. Kaklamani said. “It seems that stopping endocrine therapy to become pregnant did not cause any adverse outcomes or increase the risk of reoccurrence of cancer in the women in the study.”
Dr. Mouabbi agreed, noting, “Many of our patients are afraid that they will miss the window to get pregnant because they have to be on treatment for so long. This is the first study that let us know pregnancy and safety outcomes in patients who took a break from endocrine therapy to get pregnant. The results are promising and will be exciting for many of our patients.”
4. Assay identifies OFS benefit
A genomic assay was able to distinguish premenopausal patients with early-stage HR-positive breast cancer who benefited from the addition of ovarian function suppression (OFS) to adjuvant endocrine therapy, according to new data presented at the meeting (GS1-06).
In the study, investigators analyzed 1,717 patient tumor samples from the landmark Suppression of Ovarian Function Trial (SOFT) trial. The Breast Cancer Index identified 58% of women who benefited from the addition of ovarian function suppression to tamoxifen or exemestane therapy. They experienced an absolute benefit of 11.6% (42% did not benefit), compared with those with received tamoxifen alone. The predictive benefit was observed regardless of age, lymph node involvement, and receipt of chemotherapy.
Dr. Kaklamani highlighted this study’s importance, saying, “Ovarian suppression is associated with severe adverse events for patients. Obviously, the women who will get a benefit should continue, but this research is important because it will hopefully show us who to recommend ovarian suppression to while not exposing patients who are likely to get little benefit to unneeded toxicity.”
5. Optimizing elacestrant PFS
Last year, data from the Emerald trial showed that elacestrant is superior to standard-of-care therapy for HR-positive metastatic breast cancer. An update that Dr. Kaklamani presented at SABCS (GS3-01) explored whether the duration of a prior CDK4/6 inhibitor affects PFS.
The study was a randomized, open-label, phase 3 trial in which 478 patients with ER-positive/HER2-negative metastatic breast cancer received either elacestrant or standard of care. These patients had previously received one or two lines of endocrine therapy, a CDK4/6 inhibitor, and ≤ 1 line of chemotherapy.
Overall, the duration of prior CDK4/6 inhibitor in the metastatic setting was positively associated with PFS – the longer the duration of prior CDK4/6 inhibitor therapy, the longer the PFS with elacestrant. PFS outcomes were even stronger among patients with ESR1 mutations.
“What we found was that the women who benefit most from elacestrant had previously received a CDK4/6 inhibitor for at least 6 months,” Dr. Kaklamani said. These data can help us determine who may do best on the drug, she added.
A version of this article first appeared on Medscape.com.
AT SABCS 2022
Liver cancer exacts high financial toll on older adults
In the first year after a diagnosis of HCC, median Medicare payments exceed $65,000 and out-of-pocket costs top $10,000.
Even after adjustment for the presence of cirrhosis and its related costs, patients with HCC still have Medicare payments exceeding $50,000 and out-of-pocket costs topping $7000.
Amit Singal, MD, of UT Southwestern Medical Center in Dallas, and colleagues reported their findings in Clinical Gastroenterology and Hepatology.
Common and costly
HCC, the most common type of primary liver cancer, is a leading cause of death in patients with cirrhosis and is projected to become the third leading cause of cancer-related death in the United States by 2040, the researchers wrote.
The treatment landscape for HCC has changed over the past decade, with expanded surgical options, introduction of radiation-based therapies, and approval of immunotherapies – all of which are costly.
Yet the magnitude of financial burden of HCC therapy has been understudied, the researchers noted.
To investigate, Dr. Singal and colleagues evaluated Surveillance, Epidemiology, and End Results (SEER)–Medicare data for 4,525 adults with traditional Medicare coverage who were diagnosed with HCC between 2011 and 2015 and a propensity-matched cohort of 4,525 adults with cirrhosis but no HCC as a comparator group to tease out HCC-specific costs beyond those related to cirrhosis. Patients in Medicare managed care were excluded because their cost information is not available in the database.
In the first year after a diagnosis of HCC, the median total Medicare payments were $66,338 (interquartile range [IQR], $30,931-$158,740) and patient liabilities (a proxy for out-of-pocket costs) were $10,008 (IQR, $5,427-$19,669).
First-year costs were higher for patients with HCC than matched patients without HCC; the former group incurred median incremental Medicare payments of $50,110 (IQR, $14,242-$136,239) and patient liabilities of $7,166 (IQR, $2,401-$16,099), the investigators found.
Patients with early-stage HCC had lower incremental patient liabilities (median, $4,195 vs. $8,238) and Medicare payments (median, $28,207 vs. $59,509) than did their peers with larger tumor burden.
NAFLD notably tied to higher costs
Factors associated with higher HCC-related costs were nonalcoholic fatty liver disease (NAFLD) etiology, higher comorbidities, presence of ascites and hepatic encephalopathy, and larger tumor burden.
The researchers said that the link between NAFLD and higher costs is notable, given that NAFLD is an increasingly common underlying cause of HCC.
The link between larger tumor burden and higher costs underscores “another benefit of HCC surveillance and early detection,” they added.
“By separating the financial liabilities borne by patients and Medicare, we provide a clearer outlook of how cancer-related costs are distributed between patients and public payers,” Dr. Singal and colleagues said.
“Our findings will inform policy interventions and will help formulate better financial supports targeting the most vulnerable HCC patients,” they concluded.
The study had no commercial funding. Dr. Singal has been on advisory boards and served as a consultant for Wako Diagnostics, Glycotest, Exact Sciences, Roche, Genentech, Bayer, Eisai, BMS, Exelixis, AstraZeneca, and TARGET RWE.
A version of this article first appeared on Medscape.com.
In the first year after a diagnosis of HCC, median Medicare payments exceed $65,000 and out-of-pocket costs top $10,000.
Even after adjustment for the presence of cirrhosis and its related costs, patients with HCC still have Medicare payments exceeding $50,000 and out-of-pocket costs topping $7000.
Amit Singal, MD, of UT Southwestern Medical Center in Dallas, and colleagues reported their findings in Clinical Gastroenterology and Hepatology.
Common and costly
HCC, the most common type of primary liver cancer, is a leading cause of death in patients with cirrhosis and is projected to become the third leading cause of cancer-related death in the United States by 2040, the researchers wrote.
The treatment landscape for HCC has changed over the past decade, with expanded surgical options, introduction of radiation-based therapies, and approval of immunotherapies – all of which are costly.
Yet the magnitude of financial burden of HCC therapy has been understudied, the researchers noted.
To investigate, Dr. Singal and colleagues evaluated Surveillance, Epidemiology, and End Results (SEER)–Medicare data for 4,525 adults with traditional Medicare coverage who were diagnosed with HCC between 2011 and 2015 and a propensity-matched cohort of 4,525 adults with cirrhosis but no HCC as a comparator group to tease out HCC-specific costs beyond those related to cirrhosis. Patients in Medicare managed care were excluded because their cost information is not available in the database.
In the first year after a diagnosis of HCC, the median total Medicare payments were $66,338 (interquartile range [IQR], $30,931-$158,740) and patient liabilities (a proxy for out-of-pocket costs) were $10,008 (IQR, $5,427-$19,669).
First-year costs were higher for patients with HCC than matched patients without HCC; the former group incurred median incremental Medicare payments of $50,110 (IQR, $14,242-$136,239) and patient liabilities of $7,166 (IQR, $2,401-$16,099), the investigators found.
Patients with early-stage HCC had lower incremental patient liabilities (median, $4,195 vs. $8,238) and Medicare payments (median, $28,207 vs. $59,509) than did their peers with larger tumor burden.
NAFLD notably tied to higher costs
Factors associated with higher HCC-related costs were nonalcoholic fatty liver disease (NAFLD) etiology, higher comorbidities, presence of ascites and hepatic encephalopathy, and larger tumor burden.
The researchers said that the link between NAFLD and higher costs is notable, given that NAFLD is an increasingly common underlying cause of HCC.
The link between larger tumor burden and higher costs underscores “another benefit of HCC surveillance and early detection,” they added.
“By separating the financial liabilities borne by patients and Medicare, we provide a clearer outlook of how cancer-related costs are distributed between patients and public payers,” Dr. Singal and colleagues said.
“Our findings will inform policy interventions and will help formulate better financial supports targeting the most vulnerable HCC patients,” they concluded.
The study had no commercial funding. Dr. Singal has been on advisory boards and served as a consultant for Wako Diagnostics, Glycotest, Exact Sciences, Roche, Genentech, Bayer, Eisai, BMS, Exelixis, AstraZeneca, and TARGET RWE.
A version of this article first appeared on Medscape.com.
In the first year after a diagnosis of HCC, median Medicare payments exceed $65,000 and out-of-pocket costs top $10,000.
Even after adjustment for the presence of cirrhosis and its related costs, patients with HCC still have Medicare payments exceeding $50,000 and out-of-pocket costs topping $7000.
Amit Singal, MD, of UT Southwestern Medical Center in Dallas, and colleagues reported their findings in Clinical Gastroenterology and Hepatology.
Common and costly
HCC, the most common type of primary liver cancer, is a leading cause of death in patients with cirrhosis and is projected to become the third leading cause of cancer-related death in the United States by 2040, the researchers wrote.
The treatment landscape for HCC has changed over the past decade, with expanded surgical options, introduction of radiation-based therapies, and approval of immunotherapies – all of which are costly.
Yet the magnitude of financial burden of HCC therapy has been understudied, the researchers noted.
To investigate, Dr. Singal and colleagues evaluated Surveillance, Epidemiology, and End Results (SEER)–Medicare data for 4,525 adults with traditional Medicare coverage who were diagnosed with HCC between 2011 and 2015 and a propensity-matched cohort of 4,525 adults with cirrhosis but no HCC as a comparator group to tease out HCC-specific costs beyond those related to cirrhosis. Patients in Medicare managed care were excluded because their cost information is not available in the database.
In the first year after a diagnosis of HCC, the median total Medicare payments were $66,338 (interquartile range [IQR], $30,931-$158,740) and patient liabilities (a proxy for out-of-pocket costs) were $10,008 (IQR, $5,427-$19,669).
First-year costs were higher for patients with HCC than matched patients without HCC; the former group incurred median incremental Medicare payments of $50,110 (IQR, $14,242-$136,239) and patient liabilities of $7,166 (IQR, $2,401-$16,099), the investigators found.
Patients with early-stage HCC had lower incremental patient liabilities (median, $4,195 vs. $8,238) and Medicare payments (median, $28,207 vs. $59,509) than did their peers with larger tumor burden.
NAFLD notably tied to higher costs
Factors associated with higher HCC-related costs were nonalcoholic fatty liver disease (NAFLD) etiology, higher comorbidities, presence of ascites and hepatic encephalopathy, and larger tumor burden.
The researchers said that the link between NAFLD and higher costs is notable, given that NAFLD is an increasingly common underlying cause of HCC.
The link between larger tumor burden and higher costs underscores “another benefit of HCC surveillance and early detection,” they added.
“By separating the financial liabilities borne by patients and Medicare, we provide a clearer outlook of how cancer-related costs are distributed between patients and public payers,” Dr. Singal and colleagues said.
“Our findings will inform policy interventions and will help formulate better financial supports targeting the most vulnerable HCC patients,” they concluded.
The study had no commercial funding. Dr. Singal has been on advisory boards and served as a consultant for Wako Diagnostics, Glycotest, Exact Sciences, Roche, Genentech, Bayer, Eisai, BMS, Exelixis, AstraZeneca, and TARGET RWE.
A version of this article first appeared on Medscape.com.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Cancer researcher banned from federal funding for faking data in nearly 400 images in 16 grant applications
according to a U.S. government research watchdog.
Alice C. Chang, PhD, whose publications and grants listed her name as Chun-Ju Chang, received nearly $700,000 in funding from the National Institutes of Health through grant applications that the U.S. Office of Research Integrity said contained fake data. She will be banned from receiving federal grants for a decade – a more severe sanction than ORI has typically imposed in recent years.
In its findings, ORI said Dr. Chang, who was an associate professor of basic medical sciences at Purdue’s College of Veterinary Medicine, West Lafayette, Ind., “knowingly, intentionally, or recklessly falsified and/or fabricated data from the same mouse models or cell lines by reusing the data, with or without manipulation, to represent unrelated experiments from different mouse models or cell lines with different treatments in three hundred eighty-four (384) figure panels in sixteen (16) grant applications.”
Two of the grant applications were funded. Dr. Chang received $688,196 from the National Cancer Institute, a division of NIH, from 2018 to 2019 for “Targeting metformin-directed stem cell fate in triple negative breast cancer.” The other grant ORI says was submitted in 2014 and funded, “Targeting cell polarity machinery to exhaust breast cancer stem cell pool,” does not show up in NIH RePorter. The rest of the grants were not approved.
We found a Chun-Ju Chang who is dean of the College of Life Sciences at China Medical University in Taiwan and has published papers with a group that Chun-Ju Chang at Purdue also published with. She did not immediately respond to our request for comment.
ORI’s finding also stated Dr. Chang faked data in two papers supported by government funding by reusing figures reporting gene expression in mice and cells after drug treatments, relabeling them to say they showed the results of different experiments. According to the agency, she has agreed to request corrections for the papers:
“Leptin–STAT3–G9a Signaling Promotes Obesity-Mediated Breast Cancer Progression,” published in May 2015 in Cancer Research and cited 83 times, according to Clarivate’s Web of Science.
“Retinoic acid directs breast cancer cell state changes through regulation of TET2-PKC-zeta pathway,” published in February 2017 in Oncogene and cited 26 times.
Between the two papers and 15 of the grant applications, ORI said that Dr. Chang reused gene expression data, sometimes with manipulation, in 119 figure panels. She reused other types of data and images in hundreds of figures across multiple grant applications, ORI found.
As well as correcting the two papers, Dr. Chang agreed to a 10-year ban from all federal contracting, including grant funding. She also agreed not to serve in any advisory or consulting role with the U.S. Public Health Service, which includes the NIH, for that time period.
A version of this article first appeared on Retraction Watch.
according to a U.S. government research watchdog.
Alice C. Chang, PhD, whose publications and grants listed her name as Chun-Ju Chang, received nearly $700,000 in funding from the National Institutes of Health through grant applications that the U.S. Office of Research Integrity said contained fake data. She will be banned from receiving federal grants for a decade – a more severe sanction than ORI has typically imposed in recent years.
In its findings, ORI said Dr. Chang, who was an associate professor of basic medical sciences at Purdue’s College of Veterinary Medicine, West Lafayette, Ind., “knowingly, intentionally, or recklessly falsified and/or fabricated data from the same mouse models or cell lines by reusing the data, with or without manipulation, to represent unrelated experiments from different mouse models or cell lines with different treatments in three hundred eighty-four (384) figure panels in sixteen (16) grant applications.”
Two of the grant applications were funded. Dr. Chang received $688,196 from the National Cancer Institute, a division of NIH, from 2018 to 2019 for “Targeting metformin-directed stem cell fate in triple negative breast cancer.” The other grant ORI says was submitted in 2014 and funded, “Targeting cell polarity machinery to exhaust breast cancer stem cell pool,” does not show up in NIH RePorter. The rest of the grants were not approved.
We found a Chun-Ju Chang who is dean of the College of Life Sciences at China Medical University in Taiwan and has published papers with a group that Chun-Ju Chang at Purdue also published with. She did not immediately respond to our request for comment.
ORI’s finding also stated Dr. Chang faked data in two papers supported by government funding by reusing figures reporting gene expression in mice and cells after drug treatments, relabeling them to say they showed the results of different experiments. According to the agency, she has agreed to request corrections for the papers:
“Leptin–STAT3–G9a Signaling Promotes Obesity-Mediated Breast Cancer Progression,” published in May 2015 in Cancer Research and cited 83 times, according to Clarivate’s Web of Science.
“Retinoic acid directs breast cancer cell state changes through regulation of TET2-PKC-zeta pathway,” published in February 2017 in Oncogene and cited 26 times.
Between the two papers and 15 of the grant applications, ORI said that Dr. Chang reused gene expression data, sometimes with manipulation, in 119 figure panels. She reused other types of data and images in hundreds of figures across multiple grant applications, ORI found.
As well as correcting the two papers, Dr. Chang agreed to a 10-year ban from all federal contracting, including grant funding. She also agreed not to serve in any advisory or consulting role with the U.S. Public Health Service, which includes the NIH, for that time period.
A version of this article first appeared on Retraction Watch.
according to a U.S. government research watchdog.
Alice C. Chang, PhD, whose publications and grants listed her name as Chun-Ju Chang, received nearly $700,000 in funding from the National Institutes of Health through grant applications that the U.S. Office of Research Integrity said contained fake data. She will be banned from receiving federal grants for a decade – a more severe sanction than ORI has typically imposed in recent years.
In its findings, ORI said Dr. Chang, who was an associate professor of basic medical sciences at Purdue’s College of Veterinary Medicine, West Lafayette, Ind., “knowingly, intentionally, or recklessly falsified and/or fabricated data from the same mouse models or cell lines by reusing the data, with or without manipulation, to represent unrelated experiments from different mouse models or cell lines with different treatments in three hundred eighty-four (384) figure panels in sixteen (16) grant applications.”
Two of the grant applications were funded. Dr. Chang received $688,196 from the National Cancer Institute, a division of NIH, from 2018 to 2019 for “Targeting metformin-directed stem cell fate in triple negative breast cancer.” The other grant ORI says was submitted in 2014 and funded, “Targeting cell polarity machinery to exhaust breast cancer stem cell pool,” does not show up in NIH RePorter. The rest of the grants were not approved.
We found a Chun-Ju Chang who is dean of the College of Life Sciences at China Medical University in Taiwan and has published papers with a group that Chun-Ju Chang at Purdue also published with. She did not immediately respond to our request for comment.
ORI’s finding also stated Dr. Chang faked data in two papers supported by government funding by reusing figures reporting gene expression in mice and cells after drug treatments, relabeling them to say they showed the results of different experiments. According to the agency, she has agreed to request corrections for the papers:
“Leptin–STAT3–G9a Signaling Promotes Obesity-Mediated Breast Cancer Progression,” published in May 2015 in Cancer Research and cited 83 times, according to Clarivate’s Web of Science.
“Retinoic acid directs breast cancer cell state changes through regulation of TET2-PKC-zeta pathway,” published in February 2017 in Oncogene and cited 26 times.
Between the two papers and 15 of the grant applications, ORI said that Dr. Chang reused gene expression data, sometimes with manipulation, in 119 figure panels. She reused other types of data and images in hundreds of figures across multiple grant applications, ORI found.
As well as correcting the two papers, Dr. Chang agreed to a 10-year ban from all federal contracting, including grant funding. She also agreed not to serve in any advisory or consulting role with the U.S. Public Health Service, which includes the NIH, for that time period.
A version of this article first appeared on Retraction Watch.
Contralateral Constrictor Dose Predicts Swallowing Function After Radiation for Head and Neck Cancer
Radiation therapy can cause long-term dysphagia that seriously affects quality of life for survivors of head and neck (H&N) cancer.1-3 Numerous studies have linked pharyngeal constrictor dose to long-term dysphagia, but conclusions about the dose distribution that can be safely tolerated have been inconsistent. For example, a group from the Netherlands found that the mean dose to the superior pharyngeal constrictor muscle and the supraglottic larynx were each predictive of dysphagia.4 A subsequent Vanderbilt study refuted these findings, reporting that these structures were not predictive but that dose to the inferior pharyngeal constrictor muscle was.5 Other studies have connected late dysphagia with dose to the middle pharyngeal constrictor muscle, total larynx, oral cavity, contralateral submandibular gland, contralateral parotid gland, or a combination of these structures.6-14 NRG Oncology trials commonly evaluate dose to the “uninvolved pharynx,” which is the total pharyngeal constrictor muscle volume minus the planning target volume for the lowest dose target volume. NRG H&N trials 3, 4, 5, 6, 8, and 9 all use uninvolved pharynx mean dose ≤ 45 Gy as a constraint to judge radiation plan quality.
Differences in methodology or patient population may explain the inconsistency of prior studies on dosimetric predictors of dysphagia, but it is possible that these studies did not evaluate the optimal metric for dysphagia. This study evaluates a novel organ at risk, the contralateral pharyngeal constrictor muscle, to determine whether dose to this structure is predictive of late swallowing function. The study also compares a constraint based on this structure to the NRG uninvolved pharynx constraint mentioned earlier.
Methods
This study is a retrospective review of patients treated at the Richard L. Roudebush Veterans Affairs (VA) Medical Center in Indianapolis, Indiana. Patients were identified by searching the VA Cancer Registry for patients treated for H&N squamous cell carcinoma between September 1, 2016, and August 30, 2019. Eligible sites included cancers of the nasopharynx, oropharynx, hypopharynx, larynx and oral cavity, as well as H&N cancer of an unknown primary site. Only patients treated with primary radiation with concurrent systemic therapy were included. Patients were excluded if they had prior surgery or radiation to the H&N.
The pharyngeal constrictor muscles were contoured per the techniques described by Bhide and colleagues.11 The contralateral constrictor was defined as the half of the constrictor volume contralateral to the primary site. For midline tumors, the side of the neck with a lower volume of lymph node metastases was judged to be the contralateral side.
One-year dysphagia was defined as having a gastronomy tube (G-tube) in place or an abnormal modified barium swallow (MBS) ≥ 12 months after the completion of radiation. At the study institution, MBS is not routinely done after therapy but is ordered if a patient or clinician has concerns about swallowing function. MBS was considered abnormal if there was laryngeal penetration that reached the level of the glottis or was not ejected from the larynx.
Results
The VA Cancer Registry identified 113 patients treated for H&N cancer during the study period. Of these, 55 patients met the inclusion criteria. No patients were lost to follow-up. The median follow-up was 29 months. The median age was 67 years (range, 41-83) (Table 1).
All patients were treated with intensity-modulated radiotherapy (IMRT). Patients treated with a sequential boost had an initial dose of 54 Gy and/or 50 Gy, followed by a boost to a total of 70 Gy at 2 Gy per fraction. Patients treated with a simultaneous integrated boost (SIB) technique received 69.96 Gy in 33 fractions, with elective volumes treated to 54.45 Gy in 33 fractions. Both patients with nasopharyngeal cancer were treated with SIB plans and had an intermediate dose volume of 59.4 Gy.
Systemic therapy was weekly cisplatin in 41 patients (75%) and cetuximab in 14 (25%). Twenty percent of patients receiving cisplatin switched to an alternative agent during treatment, most commonly carboplatin.
Forty-nine patients (89%) had a G-tube placed before starting radiation. G-tubes were in place for an interval of 0 to 47 months (mean, 8.6); 12 (22%) had a G-tube > 12 months. After completion of radiation, 18 patients (33%) had an abnormal MBS. These were done 1 to 50 months (mean, 14.8) after completion of radiation. Abnormal MBS occurred ≥ 12 months after radiation in 9 patients, 5 of whom had their G-tube in place for less than a year.
Forty-six patients (84%) survived more than 1 year and could be evaluated for late swallowing function. One-year dysphagia was seen in 17 (37%) of these patients. Recurrence was seen in 20 patients (36%), with locoregional recurrence in 12 (60%) of these cases. Recurrence occurred at a range of 0 to 15 months (mean, 5.6). Neither recurrence (P = .69) nor locoregional recurrence (P = .11) was associated with increased 1-year dysphagia.
In patients who could be evaluated for long-term swallowing function, contralateral constrictor V60 ranged from 0% to 100% (median, 51%). V60 was < 40% in 18 patients (39%). With V60 < 40%, there was a 6% rate of 1-year dysphagia compared with 57% for V60 ≥ 40% (P < .001).
Patients with contralateral constrictor V60 < 40 and V60 ≥ 40 both had a mean age of 65 years. χ2 analysis did not show a difference in T stage or systemic treatment but did show that patients with V60 < 40% were more likely to have N1 disease (P = .01), and less likely to have N2 disease (P = .01) compared with patients with V60 ≥ 40%. The difference in 1-year dysphagia between N0 to N1 patients (27%) and N2 to N3 patients (46%) was not statistically significant (P = .19).
In patients who could be evaluated for long-term swallowing function, the uninvolved pharynx volume median of the total constrictor volume was 32% (range, < 1%-62%). The uninvolved pharynx mean dose ranged from 28 to 68 Gy (median, 45). When the uninvolved pharynx mean dose was < 45 Gy, 1-year dysphagia was 22% compared with 52% with a dose ≥ 45 Gy (P = .03).
Air cavity editing was performed in 27 patients (49%). One-year survival was 93% with air cavity editing, and 75% without, which was not statistically significant. Locoregional recurrence occurred in 3 patients (11%) with air cavity editing, and 9 (32%) without, which was not statistically significant. In patients surviving at least 1 year, contralateral constrictor V60 averaged 33% with editing and 62% without editing (P < .001). One-year dysphagia was 12% with air cavity editing and 67% without editing (P < .001).
An SIB technique was done in 26 patients (47%). One-year survival was 85% (n = 22) with SIB and 83% (n = 24) with sequential boost, which was not statistically significant. Locoregional recurrence occurred in 19% with SIB, and 32% with sequential boost, which was not statistically significant. For SIB patients alive at 1 year, the median contralateral V60 was 28%, compared with 66% for patients treated with sequential technique. Seventeen patients (77%) with SIB had V60 < 40%. Nineteen (86%) of SIB plans also had air cavity editing. One patient (5%) with SIB had dysphagia at 1 year, compared with 16 (67%) sequential patients (P < .001).
Discussion
This is the first study to link contralateral constrictor dose to long-term dysphagia in patients treated with radiation for H&N cancer. Editing the boost volume off air cavities was associated with lower contralateral constrictor V60 and with less long-term dysphagia. This may indicate that optimizing plans to meet a contralateral constrictor constraint can reduce rates of long-term dysphagia.
The most useful clinical predictors are those that identify a patient at low risk for toxicity. These constraints are useful because they reassure physicians that treatments will have a favorable risk/benefit ratio while identifying plans that may need modification before starting treatment.
The contralateral constrictor outperformed the uninvolved pharynx in identifying patients at low risk for long-term dysphagia. This difference could not be overcome by decreasing the threshold of the pharynx constraint, as 17% of patients with dysphagia had a mean dose of < 40 Gy to the uninvolved pharynx, which was not statistically significant.
An advantage of contralateral constrictor is that it is independent of planning target volume (PTV) size. The uninvolved pharynx structure depends on the PTV contour, so it may obscure a connection between PTV size and dysphagia.
In the context of a clinical trial, only measuring dose to the uninvolved pharynx may allow more plans to meet constraints, but even in NRG trials, physicians have some control over target volumes. For example, NRG HN009, a national trial for patients with H&N cancer, recommends editing the CTV_7000 (clinical target volume treated to 70 Gy) off air cavities but does not define how much the volume should be cropped or specify protocol violations if the volume is not cropped.15 Furthermore, constraints used in clinical trials are often adopted for use outside the trial, where physicians have extensive control over target volumes.
The broad range of uninvolved pharynx volume relative to total constrictor volume confounds predictions using this variable. For example, according to the NRG constraint, a patient with an uninvolved pharynx mean dose of 44 Gy will have a low risk of dysphagia even if this structure is only 1% of the total constrictor. The contralateral constrictor is always about 50% of the total constrictor volume, which means that predictions using this structure will not be confounded by the same variation in volume size.
Figure 2 shows a representative patient who met the NRG uninvolved pharynx constraint but developed long-term dysphagia.
Pharyngoesophageal stricture is a common cause of dysphagia after IMRT for H&N cancer.16 Radiation has been shown to decrease pharyngeal function in patients with H&N cancer.17 Sparing one side of the pharynx may allow for better pharyngeal compliance throughout the length of the pharynx, possibly decreasing the rate of pharyngoesophageal stricture. Additionally, constraining the contralateral constrictor may preserve strength on this side, allowing it to compensate for weakness on the side of the primary cancer. An exercise sometimes used for dysphagia involves head rotation toward the affected side during swallowing. This technique has been shown to cause food to move to the unaffected side.18 Sparing the contralateral constrictor may help such techniques work better in patients with H&N cancer.
Few studies have commented specifically on dose to swallowing structures contralateral to the primary tumor. Two studies have proposed contralateral submandibular gland constraints for dysphagia (not xerostomia), but neither measured the dose to the contralateral constrictor muscle.9,10 Although the contralateral submandibular dose may correlate with dose to the constrictor on that side, the submandibular gland may have a less direct impact on swallowing than the constrictor muscle, and its limited dimensions may make constraints based on the gland less robust for cancers outside the oropharynx.
Another study reported improved quality of life in patients who were not treated with elective contralateral retropharyngeal radiation.19 Although it is likely that doses to the contralateral constrictor were lower in patients who did not receive elective radiation to this area, this study did not measure or constrain doses to the contralateral constrictors.
Limitations
This study is limited by its single institution, retrospective design, small sample size, and by all patients being male. The high correlation between air cavity editing and the use of SIB makes it impossible to assess the impact of each technique individually. Patients with contralateral constrictor V60 < 40% were less likely to have N2 disease, but N2 to N3 disease did not predict higher 1-year dysphagia, so the difference in N-category cannot fully explain the difference in 1-year dysphagia. It is possible that unreported factors, such as CTV, may contribute significantly to swallowing function. Nevertheless, within the study population, contralateral constrictor dose was able to identify a group with a low rate of long-term dysphagia.
Conclusions
Contralateral constrictor dose is a promising predictor of late dysphagia for patients with H&N cancer treated with radiation with concurrent systemic therapy. Contralateral constrictor V60 < 40% was able to identify a group of patients with a low rate of 1-year dysphagia in this single-center retrospective study. The correlation between air cavity editing and contralateral constrictor V60 suggests that contralateral constrictor dose may depend partly on technique. Further studies are needed to see if the contralateral constrictor dose can be used to predict long-term dysphagia prospectively and in other patient populations.
1. Langendijk JA, Doornaert P, Verdonck-de Leeuw IM, et al. Impact of late treatment-related toxicity on quality of life among patients with head and neck cancer treated with radiotherapy. J Clin Oncol. 2008;26(22):3770-3776. doi:10.1200/JCO.2007.14.6647
2. Nguyen NP, Frank C, Moltz CC, et al. Impact of dysphagia on quality of life after treatment of head-and-neck cancer. Int J Radiat Oncol Biol Phys. 2005;61(3):772-778. doi:10.1016/j.ijrobp.2004.06.017
3. Ramaekers BLT, Joore MA, Grutters JPC, et al. The impact of late treatment-toxicity on generic health-related quality of life in head and neck cancer patients after radiotherapy. Oral Oncol. 2011;47(8):768-774. doi:10.1016/j.oraloncology.2011.05.012
4. Christianen MEMC, Schilstra C, Beetz I, et al. Predictive modelling for swallowing dysfunction after primary (chemo)radiation: results of a prospective observational study. Radiother Oncol. 2012;105(1):107-114. doi:10.1016/j.radonc.2011.08.009
5. Vlachich G, Spratt DE, Diaz R, et al. Dose to inferior pharyngeal conctrictor predicts prolonged gastrostomy tube dependence with concurrent intensity-modulated radiation therapy and chemotherapy for locally-advanced head and neck cancer. Radiother Oncol. 2014;110(3):435-440. doi:10.1016/j.radonc.2013.12.007
6. Mogadas S, Busch CJ, Pflug Cet al. Influence of radiation dose to pharyngeal constrictor muscles on late dysphagia and quality of life in patients with locally advanced oropharyngeal carcinoma. Strahlenther Onkol. 2020;196(6):522-529. doi:10.1007/s00066-019-01572-0
7. Caglar HB, Tishler RB, Othus M, et al. Dose to larynx predicts of swallowing complications after intensity-modulated radiotherapy. Int J Radiat Oncol Biol Phys. 2008;72(4):1110-1118. doi:10.1016/j.ijrobp.2008.02.048
8. Schwartz DL, Hutcheson K, Barringer D, et al. Candidate dosimetric predictors of long-term swallowing dysfunction after oropharyngeal intensity-modulated radiotherapy. Int J Radiat Oncol Biol Phys. 2010;78(5):1356-1365. doi:10.1016/j.ijrobp.2009.10.002
9. Gensheimer MF, Nyflot M, Laramore GE, Laio JL, Parvathaneni U. Contribution of submandibular gland and swallowing structure sparing to post-radiation therapy peg dependence in oropharynx cancer patients treated with split-neck IMRT technique. Radiat Oncol. 2015;11(1):1-7. doi:10.1186/s13014-016-0726-3
10. Hedström J, Tuomi L, Finizia C, Olsson C. Identifying organs at risk for radiation-induced late dysphagia in head and neck cancer patients. Clin Transl Radiat Oncol. 2019;19:87-95. doi:10.1016/j.ctro.2019.08.005
11. Bhide SA, Gulliford S, Kazi R, et al. Correlation between dose to the pharyngeal constrictors and patient quality of life and late dysphagia following chemo-IMRT for head and neck cancer. Radiother Oncol. 2009;93(3):539-544. doi:10.1016/j.radonc.2009.09.017
12. Caudell JJ, Schaner PE, Desmond RA, Meredith RF, Spencer SA, Bonner JA. Dosimetric factors associated with long-term dysphagia after definitive radiotherapy for squamous cell carcinoma of the head and neck. Int J Radiat Oncol Biol Phys. 2010;76(2):403-409. doi:10.1016/j.ijrobp.2009.02.017
13. Levendag PC, Teguh DN, Voet P, et al. Dysphagia disorders in patients with cancer of the oropharynx are significantly affected by the radiation therapy dose to the superior and middle constrictor muscle: a dose-effect relationship. Radiother Oncol. 2007;85(1):64-73. doi:10.1016/j.radonc.2007.07.009
14. Eisbruch A, Schwartz M, Rasch C, et al. Dysphagia and aspiration after chemoradiotherapy for head-and-neck cancer: which anatomic structures are affected and can they be spared by IMRT? Int J Radiat Oncol Biol Phys. 2004;60(5):1425-1439. doi:10.1016/j.ijrobp.2004.05.050
15. Harari PM; NRG Oncology. Comparing high-dose cisplatin every three weeks to low-dose cisplatin weekly when combined with radiation for patients with advanced head and neck cancer. ClinicalTrials.gov identifier: NCT05050162. Updated November 25, 2022. Accessed December 7, 2022. https://clinicaltrials.gov/ct2/show/NCT05050162
16. Wang JJ, Goldsmith TA, Holman AS, Cianchetti M, Chan AW. Pharyngoesophageal stricture after treatment for head and neck cancer. Head Neck. 2011;34(7):967-973. doi:10.1002/hed.21842
17. Kendall KA, McKenzie SW, Leonard RJ, Jones CU. Timing of swallowing events after single-modality treatment of head and neck carcinoma with radiotherapy. Ann Otol Rhinol Laryngol. 2000;109(8, pt 1):767-775. doi:10.1177/000348940010900812
18. Ohmae Y, Ogura M, Kitahara S. Effects of head rotation on pharyngeal function during normal swallow. Ann Otol Rhinol Laryngol. 1998;107(4):344-348. doi:10.1177/000348949810700414
19. Spencer CR, Gay HA, Haughey BH, et al. Eliminating radiotherapy to the contralateral retropharyngeal and high level II lymph nodes in head and neck squamous cell carcinoma is safe and improves quality of life. Cancer. 2014;120(24):3994-4002. doi:10.1002/cncr.28938
Radiation therapy can cause long-term dysphagia that seriously affects quality of life for survivors of head and neck (H&N) cancer.1-3 Numerous studies have linked pharyngeal constrictor dose to long-term dysphagia, but conclusions about the dose distribution that can be safely tolerated have been inconsistent. For example, a group from the Netherlands found that the mean dose to the superior pharyngeal constrictor muscle and the supraglottic larynx were each predictive of dysphagia.4 A subsequent Vanderbilt study refuted these findings, reporting that these structures were not predictive but that dose to the inferior pharyngeal constrictor muscle was.5 Other studies have connected late dysphagia with dose to the middle pharyngeal constrictor muscle, total larynx, oral cavity, contralateral submandibular gland, contralateral parotid gland, or a combination of these structures.6-14 NRG Oncology trials commonly evaluate dose to the “uninvolved pharynx,” which is the total pharyngeal constrictor muscle volume minus the planning target volume for the lowest dose target volume. NRG H&N trials 3, 4, 5, 6, 8, and 9 all use uninvolved pharynx mean dose ≤ 45 Gy as a constraint to judge radiation plan quality.
Differences in methodology or patient population may explain the inconsistency of prior studies on dosimetric predictors of dysphagia, but it is possible that these studies did not evaluate the optimal metric for dysphagia. This study evaluates a novel organ at risk, the contralateral pharyngeal constrictor muscle, to determine whether dose to this structure is predictive of late swallowing function. The study also compares a constraint based on this structure to the NRG uninvolved pharynx constraint mentioned earlier.
Methods
This study is a retrospective review of patients treated at the Richard L. Roudebush Veterans Affairs (VA) Medical Center in Indianapolis, Indiana. Patients were identified by searching the VA Cancer Registry for patients treated for H&N squamous cell carcinoma between September 1, 2016, and August 30, 2019. Eligible sites included cancers of the nasopharynx, oropharynx, hypopharynx, larynx and oral cavity, as well as H&N cancer of an unknown primary site. Only patients treated with primary radiation with concurrent systemic therapy were included. Patients were excluded if they had prior surgery or radiation to the H&N.
The pharyngeal constrictor muscles were contoured per the techniques described by Bhide and colleagues.11 The contralateral constrictor was defined as the half of the constrictor volume contralateral to the primary site. For midline tumors, the side of the neck with a lower volume of lymph node metastases was judged to be the contralateral side.
One-year dysphagia was defined as having a gastronomy tube (G-tube) in place or an abnormal modified barium swallow (MBS) ≥ 12 months after the completion of radiation. At the study institution, MBS is not routinely done after therapy but is ordered if a patient or clinician has concerns about swallowing function. MBS was considered abnormal if there was laryngeal penetration that reached the level of the glottis or was not ejected from the larynx.
Results
The VA Cancer Registry identified 113 patients treated for H&N cancer during the study period. Of these, 55 patients met the inclusion criteria. No patients were lost to follow-up. The median follow-up was 29 months. The median age was 67 years (range, 41-83) (Table 1).
All patients were treated with intensity-modulated radiotherapy (IMRT). Patients treated with a sequential boost had an initial dose of 54 Gy and/or 50 Gy, followed by a boost to a total of 70 Gy at 2 Gy per fraction. Patients treated with a simultaneous integrated boost (SIB) technique received 69.96 Gy in 33 fractions, with elective volumes treated to 54.45 Gy in 33 fractions. Both patients with nasopharyngeal cancer were treated with SIB plans and had an intermediate dose volume of 59.4 Gy.
Systemic therapy was weekly cisplatin in 41 patients (75%) and cetuximab in 14 (25%). Twenty percent of patients receiving cisplatin switched to an alternative agent during treatment, most commonly carboplatin.
Forty-nine patients (89%) had a G-tube placed before starting radiation. G-tubes were in place for an interval of 0 to 47 months (mean, 8.6); 12 (22%) had a G-tube > 12 months. After completion of radiation, 18 patients (33%) had an abnormal MBS. These were done 1 to 50 months (mean, 14.8) after completion of radiation. Abnormal MBS occurred ≥ 12 months after radiation in 9 patients, 5 of whom had their G-tube in place for less than a year.
Forty-six patients (84%) survived more than 1 year and could be evaluated for late swallowing function. One-year dysphagia was seen in 17 (37%) of these patients. Recurrence was seen in 20 patients (36%), with locoregional recurrence in 12 (60%) of these cases. Recurrence occurred at a range of 0 to 15 months (mean, 5.6). Neither recurrence (P = .69) nor locoregional recurrence (P = .11) was associated with increased 1-year dysphagia.
In patients who could be evaluated for long-term swallowing function, contralateral constrictor V60 ranged from 0% to 100% (median, 51%). V60 was < 40% in 18 patients (39%). With V60 < 40%, there was a 6% rate of 1-year dysphagia compared with 57% for V60 ≥ 40% (P < .001).
Patients with contralateral constrictor V60 < 40 and V60 ≥ 40 both had a mean age of 65 years. χ2 analysis did not show a difference in T stage or systemic treatment but did show that patients with V60 < 40% were more likely to have N1 disease (P = .01), and less likely to have N2 disease (P = .01) compared with patients with V60 ≥ 40%. The difference in 1-year dysphagia between N0 to N1 patients (27%) and N2 to N3 patients (46%) was not statistically significant (P = .19).
In patients who could be evaluated for long-term swallowing function, the uninvolved pharynx volume median of the total constrictor volume was 32% (range, < 1%-62%). The uninvolved pharynx mean dose ranged from 28 to 68 Gy (median, 45). When the uninvolved pharynx mean dose was < 45 Gy, 1-year dysphagia was 22% compared with 52% with a dose ≥ 45 Gy (P = .03).
Air cavity editing was performed in 27 patients (49%). One-year survival was 93% with air cavity editing, and 75% without, which was not statistically significant. Locoregional recurrence occurred in 3 patients (11%) with air cavity editing, and 9 (32%) without, which was not statistically significant. In patients surviving at least 1 year, contralateral constrictor V60 averaged 33% with editing and 62% without editing (P < .001). One-year dysphagia was 12% with air cavity editing and 67% without editing (P < .001).
An SIB technique was done in 26 patients (47%). One-year survival was 85% (n = 22) with SIB and 83% (n = 24) with sequential boost, which was not statistically significant. Locoregional recurrence occurred in 19% with SIB, and 32% with sequential boost, which was not statistically significant. For SIB patients alive at 1 year, the median contralateral V60 was 28%, compared with 66% for patients treated with sequential technique. Seventeen patients (77%) with SIB had V60 < 40%. Nineteen (86%) of SIB plans also had air cavity editing. One patient (5%) with SIB had dysphagia at 1 year, compared with 16 (67%) sequential patients (P < .001).
Discussion
This is the first study to link contralateral constrictor dose to long-term dysphagia in patients treated with radiation for H&N cancer. Editing the boost volume off air cavities was associated with lower contralateral constrictor V60 and with less long-term dysphagia. This may indicate that optimizing plans to meet a contralateral constrictor constraint can reduce rates of long-term dysphagia.
The most useful clinical predictors are those that identify a patient at low risk for toxicity. These constraints are useful because they reassure physicians that treatments will have a favorable risk/benefit ratio while identifying plans that may need modification before starting treatment.
The contralateral constrictor outperformed the uninvolved pharynx in identifying patients at low risk for long-term dysphagia. This difference could not be overcome by decreasing the threshold of the pharynx constraint, as 17% of patients with dysphagia had a mean dose of < 40 Gy to the uninvolved pharynx, which was not statistically significant.
An advantage of contralateral constrictor is that it is independent of planning target volume (PTV) size. The uninvolved pharynx structure depends on the PTV contour, so it may obscure a connection between PTV size and dysphagia.
In the context of a clinical trial, only measuring dose to the uninvolved pharynx may allow more plans to meet constraints, but even in NRG trials, physicians have some control over target volumes. For example, NRG HN009, a national trial for patients with H&N cancer, recommends editing the CTV_7000 (clinical target volume treated to 70 Gy) off air cavities but does not define how much the volume should be cropped or specify protocol violations if the volume is not cropped.15 Furthermore, constraints used in clinical trials are often adopted for use outside the trial, where physicians have extensive control over target volumes.
The broad range of uninvolved pharynx volume relative to total constrictor volume confounds predictions using this variable. For example, according to the NRG constraint, a patient with an uninvolved pharynx mean dose of 44 Gy will have a low risk of dysphagia even if this structure is only 1% of the total constrictor. The contralateral constrictor is always about 50% of the total constrictor volume, which means that predictions using this structure will not be confounded by the same variation in volume size.
Figure 2 shows a representative patient who met the NRG uninvolved pharynx constraint but developed long-term dysphagia.
Pharyngoesophageal stricture is a common cause of dysphagia after IMRT for H&N cancer.16 Radiation has been shown to decrease pharyngeal function in patients with H&N cancer.17 Sparing one side of the pharynx may allow for better pharyngeal compliance throughout the length of the pharynx, possibly decreasing the rate of pharyngoesophageal stricture. Additionally, constraining the contralateral constrictor may preserve strength on this side, allowing it to compensate for weakness on the side of the primary cancer. An exercise sometimes used for dysphagia involves head rotation toward the affected side during swallowing. This technique has been shown to cause food to move to the unaffected side.18 Sparing the contralateral constrictor may help such techniques work better in patients with H&N cancer.
Few studies have commented specifically on dose to swallowing structures contralateral to the primary tumor. Two studies have proposed contralateral submandibular gland constraints for dysphagia (not xerostomia), but neither measured the dose to the contralateral constrictor muscle.9,10 Although the contralateral submandibular dose may correlate with dose to the constrictor on that side, the submandibular gland may have a less direct impact on swallowing than the constrictor muscle, and its limited dimensions may make constraints based on the gland less robust for cancers outside the oropharynx.
Another study reported improved quality of life in patients who were not treated with elective contralateral retropharyngeal radiation.19 Although it is likely that doses to the contralateral constrictor were lower in patients who did not receive elective radiation to this area, this study did not measure or constrain doses to the contralateral constrictors.
Limitations
This study is limited by its single institution, retrospective design, small sample size, and by all patients being male. The high correlation between air cavity editing and the use of SIB makes it impossible to assess the impact of each technique individually. Patients with contralateral constrictor V60 < 40% were less likely to have N2 disease, but N2 to N3 disease did not predict higher 1-year dysphagia, so the difference in N-category cannot fully explain the difference in 1-year dysphagia. It is possible that unreported factors, such as CTV, may contribute significantly to swallowing function. Nevertheless, within the study population, contralateral constrictor dose was able to identify a group with a low rate of long-term dysphagia.
Conclusions
Contralateral constrictor dose is a promising predictor of late dysphagia for patients with H&N cancer treated with radiation with concurrent systemic therapy. Contralateral constrictor V60 < 40% was able to identify a group of patients with a low rate of 1-year dysphagia in this single-center retrospective study. The correlation between air cavity editing and contralateral constrictor V60 suggests that contralateral constrictor dose may depend partly on technique. Further studies are needed to see if the contralateral constrictor dose can be used to predict long-term dysphagia prospectively and in other patient populations.
Radiation therapy can cause long-term dysphagia that seriously affects quality of life for survivors of head and neck (H&N) cancer.1-3 Numerous studies have linked pharyngeal constrictor dose to long-term dysphagia, but conclusions about the dose distribution that can be safely tolerated have been inconsistent. For example, a group from the Netherlands found that the mean dose to the superior pharyngeal constrictor muscle and the supraglottic larynx were each predictive of dysphagia.4 A subsequent Vanderbilt study refuted these findings, reporting that these structures were not predictive but that dose to the inferior pharyngeal constrictor muscle was.5 Other studies have connected late dysphagia with dose to the middle pharyngeal constrictor muscle, total larynx, oral cavity, contralateral submandibular gland, contralateral parotid gland, or a combination of these structures.6-14 NRG Oncology trials commonly evaluate dose to the “uninvolved pharynx,” which is the total pharyngeal constrictor muscle volume minus the planning target volume for the lowest dose target volume. NRG H&N trials 3, 4, 5, 6, 8, and 9 all use uninvolved pharynx mean dose ≤ 45 Gy as a constraint to judge radiation plan quality.
Differences in methodology or patient population may explain the inconsistency of prior studies on dosimetric predictors of dysphagia, but it is possible that these studies did not evaluate the optimal metric for dysphagia. This study evaluates a novel organ at risk, the contralateral pharyngeal constrictor muscle, to determine whether dose to this structure is predictive of late swallowing function. The study also compares a constraint based on this structure to the NRG uninvolved pharynx constraint mentioned earlier.
Methods
This study is a retrospective review of patients treated at the Richard L. Roudebush Veterans Affairs (VA) Medical Center in Indianapolis, Indiana. Patients were identified by searching the VA Cancer Registry for patients treated for H&N squamous cell carcinoma between September 1, 2016, and August 30, 2019. Eligible sites included cancers of the nasopharynx, oropharynx, hypopharynx, larynx and oral cavity, as well as H&N cancer of an unknown primary site. Only patients treated with primary radiation with concurrent systemic therapy were included. Patients were excluded if they had prior surgery or radiation to the H&N.
The pharyngeal constrictor muscles were contoured per the techniques described by Bhide and colleagues.11 The contralateral constrictor was defined as the half of the constrictor volume contralateral to the primary site. For midline tumors, the side of the neck with a lower volume of lymph node metastases was judged to be the contralateral side.
One-year dysphagia was defined as having a gastronomy tube (G-tube) in place or an abnormal modified barium swallow (MBS) ≥ 12 months after the completion of radiation. At the study institution, MBS is not routinely done after therapy but is ordered if a patient or clinician has concerns about swallowing function. MBS was considered abnormal if there was laryngeal penetration that reached the level of the glottis or was not ejected from the larynx.
Results
The VA Cancer Registry identified 113 patients treated for H&N cancer during the study period. Of these, 55 patients met the inclusion criteria. No patients were lost to follow-up. The median follow-up was 29 months. The median age was 67 years (range, 41-83) (Table 1).
All patients were treated with intensity-modulated radiotherapy (IMRT). Patients treated with a sequential boost had an initial dose of 54 Gy and/or 50 Gy, followed by a boost to a total of 70 Gy at 2 Gy per fraction. Patients treated with a simultaneous integrated boost (SIB) technique received 69.96 Gy in 33 fractions, with elective volumes treated to 54.45 Gy in 33 fractions. Both patients with nasopharyngeal cancer were treated with SIB plans and had an intermediate dose volume of 59.4 Gy.
Systemic therapy was weekly cisplatin in 41 patients (75%) and cetuximab in 14 (25%). Twenty percent of patients receiving cisplatin switched to an alternative agent during treatment, most commonly carboplatin.
Forty-nine patients (89%) had a G-tube placed before starting radiation. G-tubes were in place for an interval of 0 to 47 months (mean, 8.6); 12 (22%) had a G-tube > 12 months. After completion of radiation, 18 patients (33%) had an abnormal MBS. These were done 1 to 50 months (mean, 14.8) after completion of radiation. Abnormal MBS occurred ≥ 12 months after radiation in 9 patients, 5 of whom had their G-tube in place for less than a year.
Forty-six patients (84%) survived more than 1 year and could be evaluated for late swallowing function. One-year dysphagia was seen in 17 (37%) of these patients. Recurrence was seen in 20 patients (36%), with locoregional recurrence in 12 (60%) of these cases. Recurrence occurred at a range of 0 to 15 months (mean, 5.6). Neither recurrence (P = .69) nor locoregional recurrence (P = .11) was associated with increased 1-year dysphagia.
In patients who could be evaluated for long-term swallowing function, contralateral constrictor V60 ranged from 0% to 100% (median, 51%). V60 was < 40% in 18 patients (39%). With V60 < 40%, there was a 6% rate of 1-year dysphagia compared with 57% for V60 ≥ 40% (P < .001).
Patients with contralateral constrictor V60 < 40 and V60 ≥ 40 both had a mean age of 65 years. χ2 analysis did not show a difference in T stage or systemic treatment but did show that patients with V60 < 40% were more likely to have N1 disease (P = .01), and less likely to have N2 disease (P = .01) compared with patients with V60 ≥ 40%. The difference in 1-year dysphagia between N0 to N1 patients (27%) and N2 to N3 patients (46%) was not statistically significant (P = .19).
In patients who could be evaluated for long-term swallowing function, the uninvolved pharynx volume median of the total constrictor volume was 32% (range, < 1%-62%). The uninvolved pharynx mean dose ranged from 28 to 68 Gy (median, 45). When the uninvolved pharynx mean dose was < 45 Gy, 1-year dysphagia was 22% compared with 52% with a dose ≥ 45 Gy (P = .03).
Air cavity editing was performed in 27 patients (49%). One-year survival was 93% with air cavity editing, and 75% without, which was not statistically significant. Locoregional recurrence occurred in 3 patients (11%) with air cavity editing, and 9 (32%) without, which was not statistically significant. In patients surviving at least 1 year, contralateral constrictor V60 averaged 33% with editing and 62% without editing (P < .001). One-year dysphagia was 12% with air cavity editing and 67% without editing (P < .001).
An SIB technique was done in 26 patients (47%). One-year survival was 85% (n = 22) with SIB and 83% (n = 24) with sequential boost, which was not statistically significant. Locoregional recurrence occurred in 19% with SIB, and 32% with sequential boost, which was not statistically significant. For SIB patients alive at 1 year, the median contralateral V60 was 28%, compared with 66% for patients treated with sequential technique. Seventeen patients (77%) with SIB had V60 < 40%. Nineteen (86%) of SIB plans also had air cavity editing. One patient (5%) with SIB had dysphagia at 1 year, compared with 16 (67%) sequential patients (P < .001).
Discussion
This is the first study to link contralateral constrictor dose to long-term dysphagia in patients treated with radiation for H&N cancer. Editing the boost volume off air cavities was associated with lower contralateral constrictor V60 and with less long-term dysphagia. This may indicate that optimizing plans to meet a contralateral constrictor constraint can reduce rates of long-term dysphagia.
The most useful clinical predictors are those that identify a patient at low risk for toxicity. These constraints are useful because they reassure physicians that treatments will have a favorable risk/benefit ratio while identifying plans that may need modification before starting treatment.
The contralateral constrictor outperformed the uninvolved pharynx in identifying patients at low risk for long-term dysphagia. This difference could not be overcome by decreasing the threshold of the pharynx constraint, as 17% of patients with dysphagia had a mean dose of < 40 Gy to the uninvolved pharynx, which was not statistically significant.
An advantage of contralateral constrictor is that it is independent of planning target volume (PTV) size. The uninvolved pharynx structure depends on the PTV contour, so it may obscure a connection between PTV size and dysphagia.
In the context of a clinical trial, only measuring dose to the uninvolved pharynx may allow more plans to meet constraints, but even in NRG trials, physicians have some control over target volumes. For example, NRG HN009, a national trial for patients with H&N cancer, recommends editing the CTV_7000 (clinical target volume treated to 70 Gy) off air cavities but does not define how much the volume should be cropped or specify protocol violations if the volume is not cropped.15 Furthermore, constraints used in clinical trials are often adopted for use outside the trial, where physicians have extensive control over target volumes.
The broad range of uninvolved pharynx volume relative to total constrictor volume confounds predictions using this variable. For example, according to the NRG constraint, a patient with an uninvolved pharynx mean dose of 44 Gy will have a low risk of dysphagia even if this structure is only 1% of the total constrictor. The contralateral constrictor is always about 50% of the total constrictor volume, which means that predictions using this structure will not be confounded by the same variation in volume size.
Figure 2 shows a representative patient who met the NRG uninvolved pharynx constraint but developed long-term dysphagia.
Pharyngoesophageal stricture is a common cause of dysphagia after IMRT for H&N cancer.16 Radiation has been shown to decrease pharyngeal function in patients with H&N cancer.17 Sparing one side of the pharynx may allow for better pharyngeal compliance throughout the length of the pharynx, possibly decreasing the rate of pharyngoesophageal stricture. Additionally, constraining the contralateral constrictor may preserve strength on this side, allowing it to compensate for weakness on the side of the primary cancer. An exercise sometimes used for dysphagia involves head rotation toward the affected side during swallowing. This technique has been shown to cause food to move to the unaffected side.18 Sparing the contralateral constrictor may help such techniques work better in patients with H&N cancer.
Few studies have commented specifically on dose to swallowing structures contralateral to the primary tumor. Two studies have proposed contralateral submandibular gland constraints for dysphagia (not xerostomia), but neither measured the dose to the contralateral constrictor muscle.9,10 Although the contralateral submandibular dose may correlate with dose to the constrictor on that side, the submandibular gland may have a less direct impact on swallowing than the constrictor muscle, and its limited dimensions may make constraints based on the gland less robust for cancers outside the oropharynx.
Another study reported improved quality of life in patients who were not treated with elective contralateral retropharyngeal radiation.19 Although it is likely that doses to the contralateral constrictor were lower in patients who did not receive elective radiation to this area, this study did not measure or constrain doses to the contralateral constrictors.
Limitations
This study is limited by its single institution, retrospective design, small sample size, and by all patients being male. The high correlation between air cavity editing and the use of SIB makes it impossible to assess the impact of each technique individually. Patients with contralateral constrictor V60 < 40% were less likely to have N2 disease, but N2 to N3 disease did not predict higher 1-year dysphagia, so the difference in N-category cannot fully explain the difference in 1-year dysphagia. It is possible that unreported factors, such as CTV, may contribute significantly to swallowing function. Nevertheless, within the study population, contralateral constrictor dose was able to identify a group with a low rate of long-term dysphagia.
Conclusions
Contralateral constrictor dose is a promising predictor of late dysphagia for patients with H&N cancer treated with radiation with concurrent systemic therapy. Contralateral constrictor V60 < 40% was able to identify a group of patients with a low rate of 1-year dysphagia in this single-center retrospective study. The correlation between air cavity editing and contralateral constrictor V60 suggests that contralateral constrictor dose may depend partly on technique. Further studies are needed to see if the contralateral constrictor dose can be used to predict long-term dysphagia prospectively and in other patient populations.
1. Langendijk JA, Doornaert P, Verdonck-de Leeuw IM, et al. Impact of late treatment-related toxicity on quality of life among patients with head and neck cancer treated with radiotherapy. J Clin Oncol. 2008;26(22):3770-3776. doi:10.1200/JCO.2007.14.6647
2. Nguyen NP, Frank C, Moltz CC, et al. Impact of dysphagia on quality of life after treatment of head-and-neck cancer. Int J Radiat Oncol Biol Phys. 2005;61(3):772-778. doi:10.1016/j.ijrobp.2004.06.017
3. Ramaekers BLT, Joore MA, Grutters JPC, et al. The impact of late treatment-toxicity on generic health-related quality of life in head and neck cancer patients after radiotherapy. Oral Oncol. 2011;47(8):768-774. doi:10.1016/j.oraloncology.2011.05.012
4. Christianen MEMC, Schilstra C, Beetz I, et al. Predictive modelling for swallowing dysfunction after primary (chemo)radiation: results of a prospective observational study. Radiother Oncol. 2012;105(1):107-114. doi:10.1016/j.radonc.2011.08.009
5. Vlachich G, Spratt DE, Diaz R, et al. Dose to inferior pharyngeal conctrictor predicts prolonged gastrostomy tube dependence with concurrent intensity-modulated radiation therapy and chemotherapy for locally-advanced head and neck cancer. Radiother Oncol. 2014;110(3):435-440. doi:10.1016/j.radonc.2013.12.007
6. Mogadas S, Busch CJ, Pflug Cet al. Influence of radiation dose to pharyngeal constrictor muscles on late dysphagia and quality of life in patients with locally advanced oropharyngeal carcinoma. Strahlenther Onkol. 2020;196(6):522-529. doi:10.1007/s00066-019-01572-0
7. Caglar HB, Tishler RB, Othus M, et al. Dose to larynx predicts of swallowing complications after intensity-modulated radiotherapy. Int J Radiat Oncol Biol Phys. 2008;72(4):1110-1118. doi:10.1016/j.ijrobp.2008.02.048
8. Schwartz DL, Hutcheson K, Barringer D, et al. Candidate dosimetric predictors of long-term swallowing dysfunction after oropharyngeal intensity-modulated radiotherapy. Int J Radiat Oncol Biol Phys. 2010;78(5):1356-1365. doi:10.1016/j.ijrobp.2009.10.002
9. Gensheimer MF, Nyflot M, Laramore GE, Laio JL, Parvathaneni U. Contribution of submandibular gland and swallowing structure sparing to post-radiation therapy peg dependence in oropharynx cancer patients treated with split-neck IMRT technique. Radiat Oncol. 2015;11(1):1-7. doi:10.1186/s13014-016-0726-3
10. Hedström J, Tuomi L, Finizia C, Olsson C. Identifying organs at risk for radiation-induced late dysphagia in head and neck cancer patients. Clin Transl Radiat Oncol. 2019;19:87-95. doi:10.1016/j.ctro.2019.08.005
11. Bhide SA, Gulliford S, Kazi R, et al. Correlation between dose to the pharyngeal constrictors and patient quality of life and late dysphagia following chemo-IMRT for head and neck cancer. Radiother Oncol. 2009;93(3):539-544. doi:10.1016/j.radonc.2009.09.017
12. Caudell JJ, Schaner PE, Desmond RA, Meredith RF, Spencer SA, Bonner JA. Dosimetric factors associated with long-term dysphagia after definitive radiotherapy for squamous cell carcinoma of the head and neck. Int J Radiat Oncol Biol Phys. 2010;76(2):403-409. doi:10.1016/j.ijrobp.2009.02.017
13. Levendag PC, Teguh DN, Voet P, et al. Dysphagia disorders in patients with cancer of the oropharynx are significantly affected by the radiation therapy dose to the superior and middle constrictor muscle: a dose-effect relationship. Radiother Oncol. 2007;85(1):64-73. doi:10.1016/j.radonc.2007.07.009
14. Eisbruch A, Schwartz M, Rasch C, et al. Dysphagia and aspiration after chemoradiotherapy for head-and-neck cancer: which anatomic structures are affected and can they be spared by IMRT? Int J Radiat Oncol Biol Phys. 2004;60(5):1425-1439. doi:10.1016/j.ijrobp.2004.05.050
15. Harari PM; NRG Oncology. Comparing high-dose cisplatin every three weeks to low-dose cisplatin weekly when combined with radiation for patients with advanced head and neck cancer. ClinicalTrials.gov identifier: NCT05050162. Updated November 25, 2022. Accessed December 7, 2022. https://clinicaltrials.gov/ct2/show/NCT05050162
16. Wang JJ, Goldsmith TA, Holman AS, Cianchetti M, Chan AW. Pharyngoesophageal stricture after treatment for head and neck cancer. Head Neck. 2011;34(7):967-973. doi:10.1002/hed.21842
17. Kendall KA, McKenzie SW, Leonard RJ, Jones CU. Timing of swallowing events after single-modality treatment of head and neck carcinoma with radiotherapy. Ann Otol Rhinol Laryngol. 2000;109(8, pt 1):767-775. doi:10.1177/000348940010900812
18. Ohmae Y, Ogura M, Kitahara S. Effects of head rotation on pharyngeal function during normal swallow. Ann Otol Rhinol Laryngol. 1998;107(4):344-348. doi:10.1177/000348949810700414
19. Spencer CR, Gay HA, Haughey BH, et al. Eliminating radiotherapy to the contralateral retropharyngeal and high level II lymph nodes in head and neck squamous cell carcinoma is safe and improves quality of life. Cancer. 2014;120(24):3994-4002. doi:10.1002/cncr.28938
1. Langendijk JA, Doornaert P, Verdonck-de Leeuw IM, et al. Impact of late treatment-related toxicity on quality of life among patients with head and neck cancer treated with radiotherapy. J Clin Oncol. 2008;26(22):3770-3776. doi:10.1200/JCO.2007.14.6647
2. Nguyen NP, Frank C, Moltz CC, et al. Impact of dysphagia on quality of life after treatment of head-and-neck cancer. Int J Radiat Oncol Biol Phys. 2005;61(3):772-778. doi:10.1016/j.ijrobp.2004.06.017
3. Ramaekers BLT, Joore MA, Grutters JPC, et al. The impact of late treatment-toxicity on generic health-related quality of life in head and neck cancer patients after radiotherapy. Oral Oncol. 2011;47(8):768-774. doi:10.1016/j.oraloncology.2011.05.012
4. Christianen MEMC, Schilstra C, Beetz I, et al. Predictive modelling for swallowing dysfunction after primary (chemo)radiation: results of a prospective observational study. Radiother Oncol. 2012;105(1):107-114. doi:10.1016/j.radonc.2011.08.009
5. Vlachich G, Spratt DE, Diaz R, et al. Dose to inferior pharyngeal conctrictor predicts prolonged gastrostomy tube dependence with concurrent intensity-modulated radiation therapy and chemotherapy for locally-advanced head and neck cancer. Radiother Oncol. 2014;110(3):435-440. doi:10.1016/j.radonc.2013.12.007
6. Mogadas S, Busch CJ, Pflug Cet al. Influence of radiation dose to pharyngeal constrictor muscles on late dysphagia and quality of life in patients with locally advanced oropharyngeal carcinoma. Strahlenther Onkol. 2020;196(6):522-529. doi:10.1007/s00066-019-01572-0
7. Caglar HB, Tishler RB, Othus M, et al. Dose to larynx predicts of swallowing complications after intensity-modulated radiotherapy. Int J Radiat Oncol Biol Phys. 2008;72(4):1110-1118. doi:10.1016/j.ijrobp.2008.02.048
8. Schwartz DL, Hutcheson K, Barringer D, et al. Candidate dosimetric predictors of long-term swallowing dysfunction after oropharyngeal intensity-modulated radiotherapy. Int J Radiat Oncol Biol Phys. 2010;78(5):1356-1365. doi:10.1016/j.ijrobp.2009.10.002
9. Gensheimer MF, Nyflot M, Laramore GE, Laio JL, Parvathaneni U. Contribution of submandibular gland and swallowing structure sparing to post-radiation therapy peg dependence in oropharynx cancer patients treated with split-neck IMRT technique. Radiat Oncol. 2015;11(1):1-7. doi:10.1186/s13014-016-0726-3
10. Hedström J, Tuomi L, Finizia C, Olsson C. Identifying organs at risk for radiation-induced late dysphagia in head and neck cancer patients. Clin Transl Radiat Oncol. 2019;19:87-95. doi:10.1016/j.ctro.2019.08.005
11. Bhide SA, Gulliford S, Kazi R, et al. Correlation between dose to the pharyngeal constrictors and patient quality of life and late dysphagia following chemo-IMRT for head and neck cancer. Radiother Oncol. 2009;93(3):539-544. doi:10.1016/j.radonc.2009.09.017
12. Caudell JJ, Schaner PE, Desmond RA, Meredith RF, Spencer SA, Bonner JA. Dosimetric factors associated with long-term dysphagia after definitive radiotherapy for squamous cell carcinoma of the head and neck. Int J Radiat Oncol Biol Phys. 2010;76(2):403-409. doi:10.1016/j.ijrobp.2009.02.017
13. Levendag PC, Teguh DN, Voet P, et al. Dysphagia disorders in patients with cancer of the oropharynx are significantly affected by the radiation therapy dose to the superior and middle constrictor muscle: a dose-effect relationship. Radiother Oncol. 2007;85(1):64-73. doi:10.1016/j.radonc.2007.07.009
14. Eisbruch A, Schwartz M, Rasch C, et al. Dysphagia and aspiration after chemoradiotherapy for head-and-neck cancer: which anatomic structures are affected and can they be spared by IMRT? Int J Radiat Oncol Biol Phys. 2004;60(5):1425-1439. doi:10.1016/j.ijrobp.2004.05.050
15. Harari PM; NRG Oncology. Comparing high-dose cisplatin every three weeks to low-dose cisplatin weekly when combined with radiation for patients with advanced head and neck cancer. ClinicalTrials.gov identifier: NCT05050162. Updated November 25, 2022. Accessed December 7, 2022. https://clinicaltrials.gov/ct2/show/NCT05050162
16. Wang JJ, Goldsmith TA, Holman AS, Cianchetti M, Chan AW. Pharyngoesophageal stricture after treatment for head and neck cancer. Head Neck. 2011;34(7):967-973. doi:10.1002/hed.21842
17. Kendall KA, McKenzie SW, Leonard RJ, Jones CU. Timing of swallowing events after single-modality treatment of head and neck carcinoma with radiotherapy. Ann Otol Rhinol Laryngol. 2000;109(8, pt 1):767-775. doi:10.1177/000348940010900812
18. Ohmae Y, Ogura M, Kitahara S. Effects of head rotation on pharyngeal function during normal swallow. Ann Otol Rhinol Laryngol. 1998;107(4):344-348. doi:10.1177/000348949810700414
19. Spencer CR, Gay HA, Haughey BH, et al. Eliminating radiotherapy to the contralateral retropharyngeal and high level II lymph nodes in head and neck squamous cell carcinoma is safe and improves quality of life. Cancer. 2014;120(24):3994-4002. doi:10.1002/cncr.28938
Lifestyle choices could curb genetic risk for thyroid cancer
A healthier lifestyle mitigated the impact of genetic factors on the risk of thyroid cancer, in a study based on data from more than 260,000 individuals.
Thyroid cancer has increased globally in recent years and ranks 9th among 36 cancers worldwide, at a considerable cost to health care systems, wrote Xiuming Feng of Guangxi Medical University, Nanning, Guangxi, China, and colleagues.
Both genetic and lifestyle factors are related to thyroid cancer; previous research suggests a heritability of about 50%, but data on the impact of modifiable lifestyle factors on thyroid cancer are limited, the researchers said.
In a prospective cohort study published in JAMA Network Open, the researchers used data from the UK Biobank and recruited adults aged 40-69 years during March 2006–October 2010. The final study population included 264,956 individuals of European descent. The median age of the participants was 57 years, and 52% were women.
Data on lifestyle behaviors were collected using interviews and questionnaires. The researchers constructed a total lifestyle score based on five variables: diet, physical activity, weight, smoking, and alcohol consumption. Each variable was assigned a score of 0 or 1, with 1 being favorable lifestyle behavior. Lifestyle was divided into three categories: unfavorable (scores 0-1), intermediate (score 2), and favorable (scores 3-5).
Each individual’s polygenic risk score (PRS) was categorized as low, intermediate, or high based on a meta–genome-wide association study of three cohorts.
The main outcome was the development of thyroid cancer.
The researchers identified 423 incident thyroid cancer cases over a median follow-up of 11.1 years.
Overall, higher PRSs were significantly associated with thyroid cancer (hazard ratio, 2.25; 95% confidence interval [CI], 1.91-2.64; P < .00001) as was an unfavorable lifestyle score (HR, 1.93; 95% CI, 1.50-2.49; P < .001 for trend).
An unfavorable lifestyle was significantly associated with thyroid cancer in the highest PRS group, and individuals with high PRS and unfavorable lifestyle had a nearly fivefold increased risk of thyroid cancer (HR, 4.89; 95% CI, 3.03-7.91; P < .001). By extension, “Adherence to a healthier lifestyle could decrease the incidence of thyroid cancer in individuals with a higher PRS,” the researchers wrote in their discussion.
The findings were limited by several factors, including the availability of only baseline lifestyle data, and lack of data on iodine intake, radiation exposure, experience, and family history, the researchers noted. Other limitations include the potential lack of generalizability to populations other than the individuals of European descent in the current study, they said.
However, the study is the first known to address the association among lifestyle, genetic factors, and risk of thyroid cancer, and was strengthened by the large study population, and the results suggest that lifestyle interventions may help reduce the risk of thyroid cancer in those with a genetic predisposition, they concluded.
Healthy living can make a difference
The incidence of thyroid cancer has increased annually, and exploring the possible risk factors could prevent the occurrence of thyroid cancer, corresponding author Xiaobo Yang, PhD, said in an interview.
Previous studies have reported that thyroid cancer is related to genetics and lifestyle, said Dr. Yang. “However, whether healthy lifestyle was associated with thyroid cancer risk and could attenuate the impact of genetic variants on thyroid cancer remains equivocal; therefore, it is crucial to determine the associations between genetic and lifestyle with thyroid cancer,” he said.
“To our surprise, we found that adherence to healthier lifestyle also could reduce the risk of thyroid cancer in those with high genetic predispositions,” said Dr. Yang. “The findings highlight the potential role of lifestyle interventions on thyroid cancer, especially in those with high genetic risk, because the heritability of thyroid cancer was very high, approximately 50%,” he said. “More attention should be paid to the role of healthier lifestyle in the prevention of cancer,” he added.
“Adherence to a healthier lifestyle could decrease the risk of thyroid cancer, which is the important message for clinicians,” said Dr. Yang. “It is not too soon to comment on implications for clinical practice, because many studies have maintained the consistent comment that healthier lifestyle could prevent the occurrence of cancer,” he said.
The relationship between sex-specific lifestyle factors such as smoking and alcohol use and thyroid cancer remains uncertain, and more research is needed to validate these associations, Dr. Yang said. More research also is needed to confirm the complex mechanism between lifestyle and genetics in thyroid cancer, he added.
The study was supported by the National Key R&D Program of China and the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
A healthier lifestyle mitigated the impact of genetic factors on the risk of thyroid cancer, in a study based on data from more than 260,000 individuals.
Thyroid cancer has increased globally in recent years and ranks 9th among 36 cancers worldwide, at a considerable cost to health care systems, wrote Xiuming Feng of Guangxi Medical University, Nanning, Guangxi, China, and colleagues.
Both genetic and lifestyle factors are related to thyroid cancer; previous research suggests a heritability of about 50%, but data on the impact of modifiable lifestyle factors on thyroid cancer are limited, the researchers said.
In a prospective cohort study published in JAMA Network Open, the researchers used data from the UK Biobank and recruited adults aged 40-69 years during March 2006–October 2010. The final study population included 264,956 individuals of European descent. The median age of the participants was 57 years, and 52% were women.
Data on lifestyle behaviors were collected using interviews and questionnaires. The researchers constructed a total lifestyle score based on five variables: diet, physical activity, weight, smoking, and alcohol consumption. Each variable was assigned a score of 0 or 1, with 1 being favorable lifestyle behavior. Lifestyle was divided into three categories: unfavorable (scores 0-1), intermediate (score 2), and favorable (scores 3-5).
Each individual’s polygenic risk score (PRS) was categorized as low, intermediate, or high based on a meta–genome-wide association study of three cohorts.
The main outcome was the development of thyroid cancer.
The researchers identified 423 incident thyroid cancer cases over a median follow-up of 11.1 years.
Overall, higher PRSs were significantly associated with thyroid cancer (hazard ratio, 2.25; 95% confidence interval [CI], 1.91-2.64; P < .00001) as was an unfavorable lifestyle score (HR, 1.93; 95% CI, 1.50-2.49; P < .001 for trend).
An unfavorable lifestyle was significantly associated with thyroid cancer in the highest PRS group, and individuals with high PRS and unfavorable lifestyle had a nearly fivefold increased risk of thyroid cancer (HR, 4.89; 95% CI, 3.03-7.91; P < .001). By extension, “Adherence to a healthier lifestyle could decrease the incidence of thyroid cancer in individuals with a higher PRS,” the researchers wrote in their discussion.
The findings were limited by several factors, including the availability of only baseline lifestyle data, and lack of data on iodine intake, radiation exposure, experience, and family history, the researchers noted. Other limitations include the potential lack of generalizability to populations other than the individuals of European descent in the current study, they said.
However, the study is the first known to address the association among lifestyle, genetic factors, and risk of thyroid cancer, and was strengthened by the large study population, and the results suggest that lifestyle interventions may help reduce the risk of thyroid cancer in those with a genetic predisposition, they concluded.
Healthy living can make a difference
The incidence of thyroid cancer has increased annually, and exploring the possible risk factors could prevent the occurrence of thyroid cancer, corresponding author Xiaobo Yang, PhD, said in an interview.
Previous studies have reported that thyroid cancer is related to genetics and lifestyle, said Dr. Yang. “However, whether healthy lifestyle was associated with thyroid cancer risk and could attenuate the impact of genetic variants on thyroid cancer remains equivocal; therefore, it is crucial to determine the associations between genetic and lifestyle with thyroid cancer,” he said.
“To our surprise, we found that adherence to healthier lifestyle also could reduce the risk of thyroid cancer in those with high genetic predispositions,” said Dr. Yang. “The findings highlight the potential role of lifestyle interventions on thyroid cancer, especially in those with high genetic risk, because the heritability of thyroid cancer was very high, approximately 50%,” he said. “More attention should be paid to the role of healthier lifestyle in the prevention of cancer,” he added.
“Adherence to a healthier lifestyle could decrease the risk of thyroid cancer, which is the important message for clinicians,” said Dr. Yang. “It is not too soon to comment on implications for clinical practice, because many studies have maintained the consistent comment that healthier lifestyle could prevent the occurrence of cancer,” he said.
The relationship between sex-specific lifestyle factors such as smoking and alcohol use and thyroid cancer remains uncertain, and more research is needed to validate these associations, Dr. Yang said. More research also is needed to confirm the complex mechanism between lifestyle and genetics in thyroid cancer, he added.
The study was supported by the National Key R&D Program of China and the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
A healthier lifestyle mitigated the impact of genetic factors on the risk of thyroid cancer, in a study based on data from more than 260,000 individuals.
Thyroid cancer has increased globally in recent years and ranks 9th among 36 cancers worldwide, at a considerable cost to health care systems, wrote Xiuming Feng of Guangxi Medical University, Nanning, Guangxi, China, and colleagues.
Both genetic and lifestyle factors are related to thyroid cancer; previous research suggests a heritability of about 50%, but data on the impact of modifiable lifestyle factors on thyroid cancer are limited, the researchers said.
In a prospective cohort study published in JAMA Network Open, the researchers used data from the UK Biobank and recruited adults aged 40-69 years during March 2006–October 2010. The final study population included 264,956 individuals of European descent. The median age of the participants was 57 years, and 52% were women.
Data on lifestyle behaviors were collected using interviews and questionnaires. The researchers constructed a total lifestyle score based on five variables: diet, physical activity, weight, smoking, and alcohol consumption. Each variable was assigned a score of 0 or 1, with 1 being favorable lifestyle behavior. Lifestyle was divided into three categories: unfavorable (scores 0-1), intermediate (score 2), and favorable (scores 3-5).
Each individual’s polygenic risk score (PRS) was categorized as low, intermediate, or high based on a meta–genome-wide association study of three cohorts.
The main outcome was the development of thyroid cancer.
The researchers identified 423 incident thyroid cancer cases over a median follow-up of 11.1 years.
Overall, higher PRSs were significantly associated with thyroid cancer (hazard ratio, 2.25; 95% confidence interval [CI], 1.91-2.64; P < .00001) as was an unfavorable lifestyle score (HR, 1.93; 95% CI, 1.50-2.49; P < .001 for trend).
An unfavorable lifestyle was significantly associated with thyroid cancer in the highest PRS group, and individuals with high PRS and unfavorable lifestyle had a nearly fivefold increased risk of thyroid cancer (HR, 4.89; 95% CI, 3.03-7.91; P < .001). By extension, “Adherence to a healthier lifestyle could decrease the incidence of thyroid cancer in individuals with a higher PRS,” the researchers wrote in their discussion.
The findings were limited by several factors, including the availability of only baseline lifestyle data, and lack of data on iodine intake, radiation exposure, experience, and family history, the researchers noted. Other limitations include the potential lack of generalizability to populations other than the individuals of European descent in the current study, they said.
However, the study is the first known to address the association among lifestyle, genetic factors, and risk of thyroid cancer, and was strengthened by the large study population, and the results suggest that lifestyle interventions may help reduce the risk of thyroid cancer in those with a genetic predisposition, they concluded.
Healthy living can make a difference
The incidence of thyroid cancer has increased annually, and exploring the possible risk factors could prevent the occurrence of thyroid cancer, corresponding author Xiaobo Yang, PhD, said in an interview.
Previous studies have reported that thyroid cancer is related to genetics and lifestyle, said Dr. Yang. “However, whether healthy lifestyle was associated with thyroid cancer risk and could attenuate the impact of genetic variants on thyroid cancer remains equivocal; therefore, it is crucial to determine the associations between genetic and lifestyle with thyroid cancer,” he said.
“To our surprise, we found that adherence to healthier lifestyle also could reduce the risk of thyroid cancer in those with high genetic predispositions,” said Dr. Yang. “The findings highlight the potential role of lifestyle interventions on thyroid cancer, especially in those with high genetic risk, because the heritability of thyroid cancer was very high, approximately 50%,” he said. “More attention should be paid to the role of healthier lifestyle in the prevention of cancer,” he added.
“Adherence to a healthier lifestyle could decrease the risk of thyroid cancer, which is the important message for clinicians,” said Dr. Yang. “It is not too soon to comment on implications for clinical practice, because many studies have maintained the consistent comment that healthier lifestyle could prevent the occurrence of cancer,” he said.
The relationship between sex-specific lifestyle factors such as smoking and alcohol use and thyroid cancer remains uncertain, and more research is needed to validate these associations, Dr. Yang said. More research also is needed to confirm the complex mechanism between lifestyle and genetics in thyroid cancer, he added.
The study was supported by the National Key R&D Program of China and the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
FROM JAMA NETWORK OPEN