User login
AVAHO
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]


Off their pricey CML meds, many thrive
When imatinib (Gleevec) appeared on the market just over 2 decades ago, it revolutionized the treatment of chronic myelogenous leukemia (CML) and transformed it from a grim diagnosis into a largely treatable form of blood cancer. New generations of tyrosine kinase inhibitors (TKIs) have continued to expand options for patients, and many can look forward to normal lifespans.
But these medications cause side effects and can be expensive. Long-term data doesn’t exist for the newer therapies, so no one knows whether they can harm patients over time. None of this is particularly unusual for medications to treat chronic illness, but now there’s a twist:
“Our focus has changed because the results of treatment are so good,” hematologist-oncologist Ehab L. Atallah, MD, of the Medical College of Wisconsin, Milwaukee, said in an interview. “We’re trying to get people off their medication.”
Still, research estimates that only 20% of patients with CML will be eligible for treatment discontinuation and benefit from it in the long term. As a result, the wide majority of patients will need to be on drugs indefinitely.
Gleevec: A new age dawns
In the early 1990s, before the era of TKIs, the 5-year relative survival rate from CML was just 27%, and the 10-year rate was only 9.5%, according to a 2008 report. “If someone showed up with CML, their only option was to go to a bone marrow transplant. About half survived the transplant, and half of those had significant complications from it,” Dr. Atallah said. According to him, just about everyone who didn’t get transplantation would go on to die.
Then came Gleevec, which received Food and Drug Administration approval in 2001. It ushered in the era of “targeted” cancer treatment by specifically killing CML cells, instead of relying on traditional chemotherapy’s carpet-bombing approach.
“Gleevec and other TKIs have revolutionized how CML is treated, and patients are now living normal lives,” hematologist-oncologist Catherine Lai, MD, MPH, of the University of Pennsylvania, Philadelphia, said in an interview.
Alan Fahnestock, a 68-year-old retired telecommunications specialist in north-central Washington state, is one of the fortunate patients.
He was diagnosed with CML in 2004 after he underwent a thoracic CT scan in light of his tobacco use. “My GP found something odd in my lungs and referred me to a pulmonologist, who couldn’t figure it out either. He transmitted blood samples to my eventual hematologist/oncologist,” Mr. Fahnestock said in an interview. “It’s not clear to me that anybody ever figured out what the ‘oddity’ was. It has since apparently gone away. But the oncologist ran all the tests and came up with CML.”
Mr. Fahnestock hadn’t noticed any symptoms, although “this is, perhaps, because I tend not to pay a lot of attention to such things, having abused my body fairly severely over the years and having been borderline anemic since I was a kid. I don’t really expect to feel great and am a bit of a fatalist: I just get on with things until I no longer can.”
His physician prescribed Gleevec. “I had no particularly notable side effects, and carried on with my life pretty much as if nothing had happened,” Mr. Fahnestock said. He stayed on the drug for almost 20 years.
CML rooted in chromosome swap
It’s not clear exactly what causes CML, although the Mayo Clinic says most cases are linked to an abnormal, extra-short “Philadelphia chromosome,” created when two chromosomes swap material. This happens after birth.
Mr. Fahnestock thinks he happened to develop a random mutation. He also wonders if his work stints in the former Soviet Union in Vladivostok, “where the Soviet nuclear submarine fleet was decomposing,” and in Kiev, Ukraine, “which is not all that far from Chernobyl,” may be responsible.
Most patients, like Mr. Fahnestock, are men. Males will account for about 5,190 of the cases diagnosed in 2023, according to the American Cancer Society, compared to 3,740 in females.
Mr. Fahnestock’s CML diagnosis came at a fairly young age, when he was in his 40s. The average patient is diagnosed at 64. But it’s not unusual that he experienced no apparent symptoms when the cancer was found. In fact, that’s the norm.
Most patients with the disease – which is diagnosed in about 8,900 patients in the United States each year – are asymptomatic or have mild symptoms, Dr. Lai said. Their disease is discovered when “an elevated white count is found on routine blood work,” she said.
“The other group of patients typically present with very elevated white blood cell counts and splenomegaly with symptoms of fatigue and other constitutional symptoms. When the WBC count is very high, it is important to rule out transformation to accelerated or blast phase and also rule out an acute leukemia.”
Polymerase chain reaction is an especially important test during diagnosis, Dr. Atallah said, since it provides baseline data about the cancer that can be tracked.
TKIs: Mainstay of treatment
Four drugs are FDA approved for initial treatment of CML: imatinib (Gleevec), the second-generation TKIs dasatinib (Sprycel) and the third-generation TKI nilotinib (Tasigna). The third-generation TKIs bosutinib (Bosulif) and ponatinib (Iclusig) are approved for use as first-line treatments for patients who cannot tolerate the other drugs or are resistant to them.
The first-in-class drug asciminib (Scemblix), approved by the FDA in 2021, is a third-line drug for patients who failed treatment with two other TKIs and certain patients with the T315I mutation.
Dr. Lai said that it’s crucial to avoid side effects as much as possible “since the goal is for patients to be compliant and take the pill every day and not miss doses.” In younger patients, “I typically choose a second-generation TKI as my first choice, since there is a higher likelihood of getting into a deep molecular remission more quickly. If treatment-free remission is something a patient is interested in, a second-generation TKI is more likely to make this happen.”
According to Dr. Atallah, about half of patients end up using more than one drug because their initial choices either don’t work or cause intolerable side effects. Nevertheless, Dr. Lai noted: “Overall, patients do extremely well if compliant with their medication.”
Exceptions include the noncompliant and patients with more aggressive disease, like an accelerated or a blast phase, she said. For the latter patients, “allogenic bone marrow transplant should be considered once the patient is in remission.”
In remission, consider drug omission
How should patients be monitored if they are doing well?
“In general, I tend to follow patients monthly for the first six months after starting therapy, to make sure they are tolerating it well and to help manage side effects,” Dr. Lai said. “After that, I follow once every three months, and then often space out visits depending on whether they hit their molecular milestones and how long they’re in remission.”
In certain cases, patients may be taken off medication. The most recent National Comprehensive Cancer Network guidelines for treatment of CML, published in 2021, say that “discontinuation of TKI therapy (with close monitoring) is feasible in carefully selected, consenting patients” with early stage CML who’ve reached remission, defined as deep molecular response (DMR) of at least MR 4.0 for at least 2 years.
The guidelines caution that disease recurrence appears in “approximately 40%-60% of patients who discontinue TKI therapy after achieving DMR experience recurrence within 12 months of treatment cessation, in some cases as early as one month after discontinuation of TKI therapy.”
Still, the guidelines add that “resumption of TKI therapy immediately after recurrence results in the achievement of DMR in almost all patients.”
Dr. Atallah said stopping medication can be especially helpful for patients who grapple with side effects such as fatigue, diarrhea, and muscle aches. Some patients who take the drugs fear losing their health insurance and facing sky-high drug expenses. In 2018, average daily TKI costs for patients with CML were over $350, a 2020 report found.
Many patients were prescribed hugely expensive second-line treatments rather than inexpensive generic imatinib, the report said, despite “no evidence that later-generation TKIs provide superior progression free or overall survival.”
Many patients, however, refuse to consider stopping their medication, Dr. Atallah said. More data about treatment-free remission is needed, and the 21 U.S. academic medical centers in the H. Jean Khoury Cure CML Consortium are gathering information about patient outcomes.
Mr. Fahnestock is a fan of treatment-free remission. He stopped taking Gleevec about 2 years ago on the advice of his physician after he reached undetectable levels of disease.
“It was sort of a nonevent, really, with no discernible physical effects beyond exacerbation of the osteoarthritis in my hands,” he said. According to him, it’s not clear if this effect is linked to his eliminating the medication.
“I also vaguely hoped I’d feel better, even though I’d never been able to nail down any deleterious side effects,” he said. “No such luck, as it happens.”
Blood work has indicated no resurgence of the disease, and Mr. Fahnestock continues to volunteer as a rural firefighter.
“In general, I’m apparently reasonably healthy for my age, despite my folly [in younger years], and firefighting requires me to stay in reasonable shape,” he said. “I’ve recently been made aware of minor kidney issues and prediabetes. But, hell, I’m genetically scheduled to croak within 5 years or so, so why worry?”
National survival statistics in CML vary by factors such as gender and age, as a 2021 study revealed, and men have worse outcomes. Still, there’s a good chance Mr. Fahnestock won’t need to worry about CML ever again.
Dr. Atallah disclosed research support from Novartis and Takeda and has served both of those firms and Bristol-Myers Squibb as a consultant advisor. Dr. Lai discloses tied with Bristol-Myers Squibb, Jazz, Genentech, Novartis, Abbvie, Daiichi Sankyo, Astellas, MacroGenics, Servier, and Taiho. Mr. Fahnestock has no disclosures.
When imatinib (Gleevec) appeared on the market just over 2 decades ago, it revolutionized the treatment of chronic myelogenous leukemia (CML) and transformed it from a grim diagnosis into a largely treatable form of blood cancer. New generations of tyrosine kinase inhibitors (TKIs) have continued to expand options for patients, and many can look forward to normal lifespans.
But these medications cause side effects and can be expensive. Long-term data doesn’t exist for the newer therapies, so no one knows whether they can harm patients over time. None of this is particularly unusual for medications to treat chronic illness, but now there’s a twist:
“Our focus has changed because the results of treatment are so good,” hematologist-oncologist Ehab L. Atallah, MD, of the Medical College of Wisconsin, Milwaukee, said in an interview. “We’re trying to get people off their medication.”
Still, research estimates that only 20% of patients with CML will be eligible for treatment discontinuation and benefit from it in the long term. As a result, the wide majority of patients will need to be on drugs indefinitely.
Gleevec: A new age dawns
In the early 1990s, before the era of TKIs, the 5-year relative survival rate from CML was just 27%, and the 10-year rate was only 9.5%, according to a 2008 report. “If someone showed up with CML, their only option was to go to a bone marrow transplant. About half survived the transplant, and half of those had significant complications from it,” Dr. Atallah said. According to him, just about everyone who didn’t get transplantation would go on to die.
Then came Gleevec, which received Food and Drug Administration approval in 2001. It ushered in the era of “targeted” cancer treatment by specifically killing CML cells, instead of relying on traditional chemotherapy’s carpet-bombing approach.
“Gleevec and other TKIs have revolutionized how CML is treated, and patients are now living normal lives,” hematologist-oncologist Catherine Lai, MD, MPH, of the University of Pennsylvania, Philadelphia, said in an interview.
Alan Fahnestock, a 68-year-old retired telecommunications specialist in north-central Washington state, is one of the fortunate patients.
He was diagnosed with CML in 2004 after he underwent a thoracic CT scan in light of his tobacco use. “My GP found something odd in my lungs and referred me to a pulmonologist, who couldn’t figure it out either. He transmitted blood samples to my eventual hematologist/oncologist,” Mr. Fahnestock said in an interview. “It’s not clear to me that anybody ever figured out what the ‘oddity’ was. It has since apparently gone away. But the oncologist ran all the tests and came up with CML.”
Mr. Fahnestock hadn’t noticed any symptoms, although “this is, perhaps, because I tend not to pay a lot of attention to such things, having abused my body fairly severely over the years and having been borderline anemic since I was a kid. I don’t really expect to feel great and am a bit of a fatalist: I just get on with things until I no longer can.”
His physician prescribed Gleevec. “I had no particularly notable side effects, and carried on with my life pretty much as if nothing had happened,” Mr. Fahnestock said. He stayed on the drug for almost 20 years.
CML rooted in chromosome swap
It’s not clear exactly what causes CML, although the Mayo Clinic says most cases are linked to an abnormal, extra-short “Philadelphia chromosome,” created when two chromosomes swap material. This happens after birth.
Mr. Fahnestock thinks he happened to develop a random mutation. He also wonders if his work stints in the former Soviet Union in Vladivostok, “where the Soviet nuclear submarine fleet was decomposing,” and in Kiev, Ukraine, “which is not all that far from Chernobyl,” may be responsible.
Most patients, like Mr. Fahnestock, are men. Males will account for about 5,190 of the cases diagnosed in 2023, according to the American Cancer Society, compared to 3,740 in females.
Mr. Fahnestock’s CML diagnosis came at a fairly young age, when he was in his 40s. The average patient is diagnosed at 64. But it’s not unusual that he experienced no apparent symptoms when the cancer was found. In fact, that’s the norm.
Most patients with the disease – which is diagnosed in about 8,900 patients in the United States each year – are asymptomatic or have mild symptoms, Dr. Lai said. Their disease is discovered when “an elevated white count is found on routine blood work,” she said.
“The other group of patients typically present with very elevated white blood cell counts and splenomegaly with symptoms of fatigue and other constitutional symptoms. When the WBC count is very high, it is important to rule out transformation to accelerated or blast phase and also rule out an acute leukemia.”
Polymerase chain reaction is an especially important test during diagnosis, Dr. Atallah said, since it provides baseline data about the cancer that can be tracked.
TKIs: Mainstay of treatment
Four drugs are FDA approved for initial treatment of CML: imatinib (Gleevec), the second-generation TKIs dasatinib (Sprycel) and the third-generation TKI nilotinib (Tasigna). The third-generation TKIs bosutinib (Bosulif) and ponatinib (Iclusig) are approved for use as first-line treatments for patients who cannot tolerate the other drugs or are resistant to them.
The first-in-class drug asciminib (Scemblix), approved by the FDA in 2021, is a third-line drug for patients who failed treatment with two other TKIs and certain patients with the T315I mutation.
Dr. Lai said that it’s crucial to avoid side effects as much as possible “since the goal is for patients to be compliant and take the pill every day and not miss doses.” In younger patients, “I typically choose a second-generation TKI as my first choice, since there is a higher likelihood of getting into a deep molecular remission more quickly. If treatment-free remission is something a patient is interested in, a second-generation TKI is more likely to make this happen.”
According to Dr. Atallah, about half of patients end up using more than one drug because their initial choices either don’t work or cause intolerable side effects. Nevertheless, Dr. Lai noted: “Overall, patients do extremely well if compliant with their medication.”
Exceptions include the noncompliant and patients with more aggressive disease, like an accelerated or a blast phase, she said. For the latter patients, “allogenic bone marrow transplant should be considered once the patient is in remission.”
In remission, consider drug omission
How should patients be monitored if they are doing well?
“In general, I tend to follow patients monthly for the first six months after starting therapy, to make sure they are tolerating it well and to help manage side effects,” Dr. Lai said. “After that, I follow once every three months, and then often space out visits depending on whether they hit their molecular milestones and how long they’re in remission.”
In certain cases, patients may be taken off medication. The most recent National Comprehensive Cancer Network guidelines for treatment of CML, published in 2021, say that “discontinuation of TKI therapy (with close monitoring) is feasible in carefully selected, consenting patients” with early stage CML who’ve reached remission, defined as deep molecular response (DMR) of at least MR 4.0 for at least 2 years.
The guidelines caution that disease recurrence appears in “approximately 40%-60% of patients who discontinue TKI therapy after achieving DMR experience recurrence within 12 months of treatment cessation, in some cases as early as one month after discontinuation of TKI therapy.”
Still, the guidelines add that “resumption of TKI therapy immediately after recurrence results in the achievement of DMR in almost all patients.”
Dr. Atallah said stopping medication can be especially helpful for patients who grapple with side effects such as fatigue, diarrhea, and muscle aches. Some patients who take the drugs fear losing their health insurance and facing sky-high drug expenses. In 2018, average daily TKI costs for patients with CML were over $350, a 2020 report found.
Many patients were prescribed hugely expensive second-line treatments rather than inexpensive generic imatinib, the report said, despite “no evidence that later-generation TKIs provide superior progression free or overall survival.”
Many patients, however, refuse to consider stopping their medication, Dr. Atallah said. More data about treatment-free remission is needed, and the 21 U.S. academic medical centers in the H. Jean Khoury Cure CML Consortium are gathering information about patient outcomes.
Mr. Fahnestock is a fan of treatment-free remission. He stopped taking Gleevec about 2 years ago on the advice of his physician after he reached undetectable levels of disease.
“It was sort of a nonevent, really, with no discernible physical effects beyond exacerbation of the osteoarthritis in my hands,” he said. According to him, it’s not clear if this effect is linked to his eliminating the medication.
“I also vaguely hoped I’d feel better, even though I’d never been able to nail down any deleterious side effects,” he said. “No such luck, as it happens.”
Blood work has indicated no resurgence of the disease, and Mr. Fahnestock continues to volunteer as a rural firefighter.
“In general, I’m apparently reasonably healthy for my age, despite my folly [in younger years], and firefighting requires me to stay in reasonable shape,” he said. “I’ve recently been made aware of minor kidney issues and prediabetes. But, hell, I’m genetically scheduled to croak within 5 years or so, so why worry?”
National survival statistics in CML vary by factors such as gender and age, as a 2021 study revealed, and men have worse outcomes. Still, there’s a good chance Mr. Fahnestock won’t need to worry about CML ever again.
Dr. Atallah disclosed research support from Novartis and Takeda and has served both of those firms and Bristol-Myers Squibb as a consultant advisor. Dr. Lai discloses tied with Bristol-Myers Squibb, Jazz, Genentech, Novartis, Abbvie, Daiichi Sankyo, Astellas, MacroGenics, Servier, and Taiho. Mr. Fahnestock has no disclosures.
When imatinib (Gleevec) appeared on the market just over 2 decades ago, it revolutionized the treatment of chronic myelogenous leukemia (CML) and transformed it from a grim diagnosis into a largely treatable form of blood cancer. New generations of tyrosine kinase inhibitors (TKIs) have continued to expand options for patients, and many can look forward to normal lifespans.
But these medications cause side effects and can be expensive. Long-term data doesn’t exist for the newer therapies, so no one knows whether they can harm patients over time. None of this is particularly unusual for medications to treat chronic illness, but now there’s a twist:
“Our focus has changed because the results of treatment are so good,” hematologist-oncologist Ehab L. Atallah, MD, of the Medical College of Wisconsin, Milwaukee, said in an interview. “We’re trying to get people off their medication.”
Still, research estimates that only 20% of patients with CML will be eligible for treatment discontinuation and benefit from it in the long term. As a result, the wide majority of patients will need to be on drugs indefinitely.
Gleevec: A new age dawns
In the early 1990s, before the era of TKIs, the 5-year relative survival rate from CML was just 27%, and the 10-year rate was only 9.5%, according to a 2008 report. “If someone showed up with CML, their only option was to go to a bone marrow transplant. About half survived the transplant, and half of those had significant complications from it,” Dr. Atallah said. According to him, just about everyone who didn’t get transplantation would go on to die.
Then came Gleevec, which received Food and Drug Administration approval in 2001. It ushered in the era of “targeted” cancer treatment by specifically killing CML cells, instead of relying on traditional chemotherapy’s carpet-bombing approach.
“Gleevec and other TKIs have revolutionized how CML is treated, and patients are now living normal lives,” hematologist-oncologist Catherine Lai, MD, MPH, of the University of Pennsylvania, Philadelphia, said in an interview.
Alan Fahnestock, a 68-year-old retired telecommunications specialist in north-central Washington state, is one of the fortunate patients.
He was diagnosed with CML in 2004 after he underwent a thoracic CT scan in light of his tobacco use. “My GP found something odd in my lungs and referred me to a pulmonologist, who couldn’t figure it out either. He transmitted blood samples to my eventual hematologist/oncologist,” Mr. Fahnestock said in an interview. “It’s not clear to me that anybody ever figured out what the ‘oddity’ was. It has since apparently gone away. But the oncologist ran all the tests and came up with CML.”
Mr. Fahnestock hadn’t noticed any symptoms, although “this is, perhaps, because I tend not to pay a lot of attention to such things, having abused my body fairly severely over the years and having been borderline anemic since I was a kid. I don’t really expect to feel great and am a bit of a fatalist: I just get on with things until I no longer can.”
His physician prescribed Gleevec. “I had no particularly notable side effects, and carried on with my life pretty much as if nothing had happened,” Mr. Fahnestock said. He stayed on the drug for almost 20 years.
CML rooted in chromosome swap
It’s not clear exactly what causes CML, although the Mayo Clinic says most cases are linked to an abnormal, extra-short “Philadelphia chromosome,” created when two chromosomes swap material. This happens after birth.
Mr. Fahnestock thinks he happened to develop a random mutation. He also wonders if his work stints in the former Soviet Union in Vladivostok, “where the Soviet nuclear submarine fleet was decomposing,” and in Kiev, Ukraine, “which is not all that far from Chernobyl,” may be responsible.
Most patients, like Mr. Fahnestock, are men. Males will account for about 5,190 of the cases diagnosed in 2023, according to the American Cancer Society, compared to 3,740 in females.
Mr. Fahnestock’s CML diagnosis came at a fairly young age, when he was in his 40s. The average patient is diagnosed at 64. But it’s not unusual that he experienced no apparent symptoms when the cancer was found. In fact, that’s the norm.
Most patients with the disease – which is diagnosed in about 8,900 patients in the United States each year – are asymptomatic or have mild symptoms, Dr. Lai said. Their disease is discovered when “an elevated white count is found on routine blood work,” she said.
“The other group of patients typically present with very elevated white blood cell counts and splenomegaly with symptoms of fatigue and other constitutional symptoms. When the WBC count is very high, it is important to rule out transformation to accelerated or blast phase and also rule out an acute leukemia.”
Polymerase chain reaction is an especially important test during diagnosis, Dr. Atallah said, since it provides baseline data about the cancer that can be tracked.
TKIs: Mainstay of treatment
Four drugs are FDA approved for initial treatment of CML: imatinib (Gleevec), the second-generation TKIs dasatinib (Sprycel) and the third-generation TKI nilotinib (Tasigna). The third-generation TKIs bosutinib (Bosulif) and ponatinib (Iclusig) are approved for use as first-line treatments for patients who cannot tolerate the other drugs or are resistant to them.
The first-in-class drug asciminib (Scemblix), approved by the FDA in 2021, is a third-line drug for patients who failed treatment with two other TKIs and certain patients with the T315I mutation.
Dr. Lai said that it’s crucial to avoid side effects as much as possible “since the goal is for patients to be compliant and take the pill every day and not miss doses.” In younger patients, “I typically choose a second-generation TKI as my first choice, since there is a higher likelihood of getting into a deep molecular remission more quickly. If treatment-free remission is something a patient is interested in, a second-generation TKI is more likely to make this happen.”
According to Dr. Atallah, about half of patients end up using more than one drug because their initial choices either don’t work or cause intolerable side effects. Nevertheless, Dr. Lai noted: “Overall, patients do extremely well if compliant with their medication.”
Exceptions include the noncompliant and patients with more aggressive disease, like an accelerated or a blast phase, she said. For the latter patients, “allogenic bone marrow transplant should be considered once the patient is in remission.”
In remission, consider drug omission
How should patients be monitored if they are doing well?
“In general, I tend to follow patients monthly for the first six months after starting therapy, to make sure they are tolerating it well and to help manage side effects,” Dr. Lai said. “After that, I follow once every three months, and then often space out visits depending on whether they hit their molecular milestones and how long they’re in remission.”
In certain cases, patients may be taken off medication. The most recent National Comprehensive Cancer Network guidelines for treatment of CML, published in 2021, say that “discontinuation of TKI therapy (with close monitoring) is feasible in carefully selected, consenting patients” with early stage CML who’ve reached remission, defined as deep molecular response (DMR) of at least MR 4.0 for at least 2 years.
The guidelines caution that disease recurrence appears in “approximately 40%-60% of patients who discontinue TKI therapy after achieving DMR experience recurrence within 12 months of treatment cessation, in some cases as early as one month after discontinuation of TKI therapy.”
Still, the guidelines add that “resumption of TKI therapy immediately after recurrence results in the achievement of DMR in almost all patients.”
Dr. Atallah said stopping medication can be especially helpful for patients who grapple with side effects such as fatigue, diarrhea, and muscle aches. Some patients who take the drugs fear losing their health insurance and facing sky-high drug expenses. In 2018, average daily TKI costs for patients with CML were over $350, a 2020 report found.
Many patients were prescribed hugely expensive second-line treatments rather than inexpensive generic imatinib, the report said, despite “no evidence that later-generation TKIs provide superior progression free or overall survival.”
Many patients, however, refuse to consider stopping their medication, Dr. Atallah said. More data about treatment-free remission is needed, and the 21 U.S. academic medical centers in the H. Jean Khoury Cure CML Consortium are gathering information about patient outcomes.
Mr. Fahnestock is a fan of treatment-free remission. He stopped taking Gleevec about 2 years ago on the advice of his physician after he reached undetectable levels of disease.
“It was sort of a nonevent, really, with no discernible physical effects beyond exacerbation of the osteoarthritis in my hands,” he said. According to him, it’s not clear if this effect is linked to his eliminating the medication.
“I also vaguely hoped I’d feel better, even though I’d never been able to nail down any deleterious side effects,” he said. “No such luck, as it happens.”
Blood work has indicated no resurgence of the disease, and Mr. Fahnestock continues to volunteer as a rural firefighter.
“In general, I’m apparently reasonably healthy for my age, despite my folly [in younger years], and firefighting requires me to stay in reasonable shape,” he said. “I’ve recently been made aware of minor kidney issues and prediabetes. But, hell, I’m genetically scheduled to croak within 5 years or so, so why worry?”
National survival statistics in CML vary by factors such as gender and age, as a 2021 study revealed, and men have worse outcomes. Still, there’s a good chance Mr. Fahnestock won’t need to worry about CML ever again.
Dr. Atallah disclosed research support from Novartis and Takeda and has served both of those firms and Bristol-Myers Squibb as a consultant advisor. Dr. Lai discloses tied with Bristol-Myers Squibb, Jazz, Genentech, Novartis, Abbvie, Daiichi Sankyo, Astellas, MacroGenics, Servier, and Taiho. Mr. Fahnestock has no disclosures.
Interval FITs could cut colonoscopies in those at above-average risk
In a new retrospective analysis of patients with above-average risk of colorectal cancer, multiple negative fecal immunohistochemical tests (FITs) were associated with a lower risk of advanced neoplasia. The findings suggest that multiple negative FITs could potentially identify individuals in high-risk surveillance who aren’t truly high risk, which could in turn ease the logjam of colonoscopies and free resources for truly high-risk individuals.
The study, conducted in Australia, was published online in Clinical Gastroenterology and Hepatology. It included patients who completed at least two FIT exams between surveillance colonoscopies and had no neoplasia or nonadvanced adenoma at prior colonoscopy. Above-average risk was defined as a family history or by findings at surveillance colonoscopy.
The study has some limitations. It is a retrospective analysis between the years 2008 and 2019, and colonoscopy guidelines in the United States have since changed, with a recommendation of surveillance colonoscopy at 7-10 years following 1-2 adenomas discovered at surveillance colonoscopy, and the current study includes follow-up colonoscopy at 5 years. “These data are informative for patients up to 5 years, but they’re not really informative afterwards. They just don’t have those data yet,” said Reed Ness, MD, who was asked to comment on the study.
The authors also don’t describe what they mean by a family history of colorectal cancer risk. “My take was that it’s an interesting result which would seem to support the possibility of returning some patients with a family history or adenoma history to a noncolonoscopy screening regimen after a negative surveillance colonoscopy. We’ll need to see where the data lead us in the future,” said Dr. Ness, who is an associate professor of medicine at Vanderbilt University Medical Center, Nashville, Tenn.
“We’re letting people go 10 years now, and some people are uncomfortable with allowing patients to go 10 years. So you could think of a scenario where you use FIT to try to find people that might have higher-risk lesions that need to come back for colonoscopy within that 10 years,” said Dr. Ness. That issue is particularly relevant given the wide range of adenoma detection rates among gastroenterologists, because FIT could detect a polyp that was missed during a colonoscopy.
The study included two groups with increased risk – those with a family history of colon cancer, and those with previously detected adenomas. The family history cohort may be useful for clinical practice, according to Priyanka Kanth, MD, who was also asked to comment on the study. “Some people may not need [a colonoscopy] at 5 years if they have no polyps found and negative FIT,” said Dr. Kanth, who is an associate professor of gastroenterology at Georgetown University, Washington.
She feels less certain about the group with previously detected adenomas, given the change in U.S. guidelines. “We have already changed that, so I don’t think we need to really do FIT intervals for that cohort,” said Dr. Kanth. “I think this is a good study that has a lot of information and also reassures us that we don’t need such frequent colonoscopy surveillance,” she added.
Steve Serrao, MD, PhD, who was also asked for comment, emphasized the importance of high-quality colonoscopies that reach the cecum 95% of the time, and achieving high adenoma-detection rates. The system can get overwhelmed conducting colonoscopies on patients with good insurance coverage who have already undergone high-quality colonoscopies. “That pushes out patients that haven’t necessarily had a colonoscopy or a FIT. People who don’t have access are kind of crowded out by these false-positive tests. The best modality is actually to do a high-quality colonoscopy and then to have a really well-directed strategy following that colonoscopy,” said Dr. Serrao, who is division chief of gastroenterology and hepatology at Riverside University Health System, Moreno Valley, Calif.
The researchers analyzed data from 4,021 surveillance intervals and 3,369 participants. A total of 1,436 had no neoplasia at the prior colonoscopy, 1,704 had nonadvanced adenoma, and 880 had advanced adenoma. Participants completed no or one to four FIT tests between colonoscopies, with the final colonoscopy performed within 2 years of FIT tests. The median age was 63.9 years; 53.6% were female; 71.1% had a prior adenoma; and 28.9% had a family history of colorectal cancer. A total of 29.4% of participants had one negative FIT; 6.9% had four negative FITs during the interval period; and 31.0% did not complete any FIT tests.
Of follow-up colonoscopies, 9.9% revealed advanced adenomas. Among the patients with no prior neoplasia, those with one negative FIT had a cumulative index function for advanced neoplasia at 5 years of 8.5% (95% confidence interval, 4.9%-13.3%). This was higher than for those with three negative FITs (4.5%; 95% CI, 2.0%-8.6%) or four negative FITs (1.9%; 95% CI, 0.5%-5.0%). The association held for individuals with prior nonadvanced adenoma but not those with advanced adenoma.
Over the 5-year interval, three or more negative FIT tests were associated with a 50%-70% reduction in advanced neoplasia risk at follow-up colonoscopy (P < .001). There was no significant association over a 3-year interval. Dr. Kanth, Dr. Serrao, and Dr. Ness have no relevant financial disclosures.
In a new retrospective analysis of patients with above-average risk of colorectal cancer, multiple negative fecal immunohistochemical tests (FITs) were associated with a lower risk of advanced neoplasia. The findings suggest that multiple negative FITs could potentially identify individuals in high-risk surveillance who aren’t truly high risk, which could in turn ease the logjam of colonoscopies and free resources for truly high-risk individuals.
The study, conducted in Australia, was published online in Clinical Gastroenterology and Hepatology. It included patients who completed at least two FIT exams between surveillance colonoscopies and had no neoplasia or nonadvanced adenoma at prior colonoscopy. Above-average risk was defined as a family history or by findings at surveillance colonoscopy.
The study has some limitations. It is a retrospective analysis between the years 2008 and 2019, and colonoscopy guidelines in the United States have since changed, with a recommendation of surveillance colonoscopy at 7-10 years following 1-2 adenomas discovered at surveillance colonoscopy, and the current study includes follow-up colonoscopy at 5 years. “These data are informative for patients up to 5 years, but they’re not really informative afterwards. They just don’t have those data yet,” said Reed Ness, MD, who was asked to comment on the study.
The authors also don’t describe what they mean by a family history of colorectal cancer risk. “My take was that it’s an interesting result which would seem to support the possibility of returning some patients with a family history or adenoma history to a noncolonoscopy screening regimen after a negative surveillance colonoscopy. We’ll need to see where the data lead us in the future,” said Dr. Ness, who is an associate professor of medicine at Vanderbilt University Medical Center, Nashville, Tenn.
“We’re letting people go 10 years now, and some people are uncomfortable with allowing patients to go 10 years. So you could think of a scenario where you use FIT to try to find people that might have higher-risk lesions that need to come back for colonoscopy within that 10 years,” said Dr. Ness. That issue is particularly relevant given the wide range of adenoma detection rates among gastroenterologists, because FIT could detect a polyp that was missed during a colonoscopy.
The study included two groups with increased risk – those with a family history of colon cancer, and those with previously detected adenomas. The family history cohort may be useful for clinical practice, according to Priyanka Kanth, MD, who was also asked to comment on the study. “Some people may not need [a colonoscopy] at 5 years if they have no polyps found and negative FIT,” said Dr. Kanth, who is an associate professor of gastroenterology at Georgetown University, Washington.
She feels less certain about the group with previously detected adenomas, given the change in U.S. guidelines. “We have already changed that, so I don’t think we need to really do FIT intervals for that cohort,” said Dr. Kanth. “I think this is a good study that has a lot of information and also reassures us that we don’t need such frequent colonoscopy surveillance,” she added.
Steve Serrao, MD, PhD, who was also asked for comment, emphasized the importance of high-quality colonoscopies that reach the cecum 95% of the time, and achieving high adenoma-detection rates. The system can get overwhelmed conducting colonoscopies on patients with good insurance coverage who have already undergone high-quality colonoscopies. “That pushes out patients that haven’t necessarily had a colonoscopy or a FIT. People who don’t have access are kind of crowded out by these false-positive tests. The best modality is actually to do a high-quality colonoscopy and then to have a really well-directed strategy following that colonoscopy,” said Dr. Serrao, who is division chief of gastroenterology and hepatology at Riverside University Health System, Moreno Valley, Calif.
The researchers analyzed data from 4,021 surveillance intervals and 3,369 participants. A total of 1,436 had no neoplasia at the prior colonoscopy, 1,704 had nonadvanced adenoma, and 880 had advanced adenoma. Participants completed no or one to four FIT tests between colonoscopies, with the final colonoscopy performed within 2 years of FIT tests. The median age was 63.9 years; 53.6% were female; 71.1% had a prior adenoma; and 28.9% had a family history of colorectal cancer. A total of 29.4% of participants had one negative FIT; 6.9% had four negative FITs during the interval period; and 31.0% did not complete any FIT tests.
Of follow-up colonoscopies, 9.9% revealed advanced adenomas. Among the patients with no prior neoplasia, those with one negative FIT had a cumulative index function for advanced neoplasia at 5 years of 8.5% (95% confidence interval, 4.9%-13.3%). This was higher than for those with three negative FITs (4.5%; 95% CI, 2.0%-8.6%) or four negative FITs (1.9%; 95% CI, 0.5%-5.0%). The association held for individuals with prior nonadvanced adenoma but not those with advanced adenoma.
Over the 5-year interval, three or more negative FIT tests were associated with a 50%-70% reduction in advanced neoplasia risk at follow-up colonoscopy (P < .001). There was no significant association over a 3-year interval. Dr. Kanth, Dr. Serrao, and Dr. Ness have no relevant financial disclosures.
In a new retrospective analysis of patients with above-average risk of colorectal cancer, multiple negative fecal immunohistochemical tests (FITs) were associated with a lower risk of advanced neoplasia. The findings suggest that multiple negative FITs could potentially identify individuals in high-risk surveillance who aren’t truly high risk, which could in turn ease the logjam of colonoscopies and free resources for truly high-risk individuals.
The study, conducted in Australia, was published online in Clinical Gastroenterology and Hepatology. It included patients who completed at least two FIT exams between surveillance colonoscopies and had no neoplasia or nonadvanced adenoma at prior colonoscopy. Above-average risk was defined as a family history or by findings at surveillance colonoscopy.
The study has some limitations. It is a retrospective analysis between the years 2008 and 2019, and colonoscopy guidelines in the United States have since changed, with a recommendation of surveillance colonoscopy at 7-10 years following 1-2 adenomas discovered at surveillance colonoscopy, and the current study includes follow-up colonoscopy at 5 years. “These data are informative for patients up to 5 years, but they’re not really informative afterwards. They just don’t have those data yet,” said Reed Ness, MD, who was asked to comment on the study.
The authors also don’t describe what they mean by a family history of colorectal cancer risk. “My take was that it’s an interesting result which would seem to support the possibility of returning some patients with a family history or adenoma history to a noncolonoscopy screening regimen after a negative surveillance colonoscopy. We’ll need to see where the data lead us in the future,” said Dr. Ness, who is an associate professor of medicine at Vanderbilt University Medical Center, Nashville, Tenn.
“We’re letting people go 10 years now, and some people are uncomfortable with allowing patients to go 10 years. So you could think of a scenario where you use FIT to try to find people that might have higher-risk lesions that need to come back for colonoscopy within that 10 years,” said Dr. Ness. That issue is particularly relevant given the wide range of adenoma detection rates among gastroenterologists, because FIT could detect a polyp that was missed during a colonoscopy.
The study included two groups with increased risk – those with a family history of colon cancer, and those with previously detected adenomas. The family history cohort may be useful for clinical practice, according to Priyanka Kanth, MD, who was also asked to comment on the study. “Some people may not need [a colonoscopy] at 5 years if they have no polyps found and negative FIT,” said Dr. Kanth, who is an associate professor of gastroenterology at Georgetown University, Washington.
She feels less certain about the group with previously detected adenomas, given the change in U.S. guidelines. “We have already changed that, so I don’t think we need to really do FIT intervals for that cohort,” said Dr. Kanth. “I think this is a good study that has a lot of information and also reassures us that we don’t need such frequent colonoscopy surveillance,” she added.
Steve Serrao, MD, PhD, who was also asked for comment, emphasized the importance of high-quality colonoscopies that reach the cecum 95% of the time, and achieving high adenoma-detection rates. The system can get overwhelmed conducting colonoscopies on patients with good insurance coverage who have already undergone high-quality colonoscopies. “That pushes out patients that haven’t necessarily had a colonoscopy or a FIT. People who don’t have access are kind of crowded out by these false-positive tests. The best modality is actually to do a high-quality colonoscopy and then to have a really well-directed strategy following that colonoscopy,” said Dr. Serrao, who is division chief of gastroenterology and hepatology at Riverside University Health System, Moreno Valley, Calif.
The researchers analyzed data from 4,021 surveillance intervals and 3,369 participants. A total of 1,436 had no neoplasia at the prior colonoscopy, 1,704 had nonadvanced adenoma, and 880 had advanced adenoma. Participants completed no or one to four FIT tests between colonoscopies, with the final colonoscopy performed within 2 years of FIT tests. The median age was 63.9 years; 53.6% were female; 71.1% had a prior adenoma; and 28.9% had a family history of colorectal cancer. A total of 29.4% of participants had one negative FIT; 6.9% had four negative FITs during the interval period; and 31.0% did not complete any FIT tests.
Of follow-up colonoscopies, 9.9% revealed advanced adenomas. Among the patients with no prior neoplasia, those with one negative FIT had a cumulative index function for advanced neoplasia at 5 years of 8.5% (95% confidence interval, 4.9%-13.3%). This was higher than for those with three negative FITs (4.5%; 95% CI, 2.0%-8.6%) or four negative FITs (1.9%; 95% CI, 0.5%-5.0%). The association held for individuals with prior nonadvanced adenoma but not those with advanced adenoma.
Over the 5-year interval, three or more negative FIT tests were associated with a 50%-70% reduction in advanced neoplasia risk at follow-up colonoscopy (P < .001). There was no significant association over a 3-year interval. Dr. Kanth, Dr. Serrao, and Dr. Ness have no relevant financial disclosures.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Oncologists may be too quick to refer patients to palliative care
I recently met Jane, a 53-year-old woman with metastatic breast cancer. She was referred to me by the breast oncology team, which routinely refers all metastatic patients to our palliative care clinic.
Clocking in at under 20 minutes, my consultation with Jane might have been one of my shortest on record. Not only had the breast oncology team already addressed Jane’s symptoms, which mainly consisted of hot flashes and joint pain attributable to treatment with an aromatase inhibitor, but they had already started planning ahead for the future of her illness. Jane had completed an advance directive and had a realistic and hopeful perspective on how her illness would progress. She understood the goal of her treatment was to “keep the cancer asleep,” as she put it, and she was very clear about her own goals: to live long enough to see her granddaughter graduate from high school in 2 years and to take a long-awaited trip to Australia later in 2023.
There wasn’t much for me to do. In fact, I daresay that Jane really did not need to see a palliative care specialist because the primary palliative care she was receiving from the breast oncology team was superb. Jane was receiving excellent symptom management from a nurse practitioner and oncologist, plus a social worker provided her with coping strategies. She was already having conversations with her primary medical team and her family about what to expect in the future and how to plan ahead for all possible outcomes.
When should a patient be referred to palliative care?
Integrating palliative care into routine oncologic care need not always require the time and skill of a palliative care team for every patient. Oncology providers can provide basic palliative care services without consulting a palliative care specialist.
For example, if a primary care doctor tried to refer every patient with hypertension to cardiology, the cardiologist would probably say that primary care should be able to handle basic hypertension management. In my experience from working in an oncology clinic for the past 9 years, I’ve found that oncology providers don’t need to refer every advanced cancer patient to our palliative care program. Most oncologists have good communication skills and are more than capable of managing symptoms for patients.
But don’t get me wrong.
Palliative care for all?
In 2010, Jennifer S. Temel MD, published a landmark study in the New England Journal of Medicine that demonstrated significant improvements in quality of life and mood in patients with metastatic lung cancer who received concurrent palliative care. After the study was published many voices inside oncology and palliative care began to advocate for a “palliative care for all” approach to patients with metastatic disease. But this is often interpreted as “specialty palliative care for all,” rather than its original intended meaning that all patients with metastatic disease receive the essential elements of palliative care (biopsychosocial symptom support and conversations about goals of care) either through their primary oncology teams or, if needed, specialty palliative care teams.
The fact is that most specialty palliative care clinics do not have the manpower to meet the needs of all patients with advanced cancers, much less all patients living with serious illness. A main goal of integrating palliative care into routine outpatient health care has always been (and in my opinion, should continue to be) to enhance the primary palliative care skills of specialists, such as oncologists and cardiologists, who care for some of our sickest patients.
This could take many forms. For one, it can be helpful to screen patients for palliative care needs. The American College of Surgeons Commission on Cancer mandates distress screening for all patients as a condition of accreditation. Distress screening using a validated tool such as the National Comprehensive Cancer Network Distress Thermometer can differentiate patients who have minimal distress and may not need much additional support beyond what is provided by their oncology team from those whose distress feels unmanageable and overwhelming.
In terms of primary palliative care symptom management, most oncology teams I work with are comfortable prescribing basic medications for pain, nausea, constipation, and anxiety. They’re also comfortable referring oncology patients for nutrition needs while undergoing chemotherapy as well as to social work and spiritual care for emotional support and counseling.
Oncology teams should continually work on communications skills. They should use “Ask, Tell, Ask” to elicit prognostic awareness, convey critical information, and assess for recall and understanding at pivotal points in the cancer journey, such as when the disease progresses or the patient’s clinical condition changes. They should practice a normalizing script they can use to introduce advance care planning to their patients in the first few visits. When I meet with a patient for the first time, I usually begin by asking if they have prepared an advanced directive. If not, I ask if they’ve thought about who will make medical decisions for them should the need arise. If the patient has documented in writing their preference for care in an emergency situation, I ask for a copy for their chart.
When should patients be referred to a specialty palliative care program?
I tell our oncology teams to involve me after they have tried to intervene, but unsuccessfully because of the patient having intractable symptoms, such as pain, or the disease is not responding to treatments. Or, because there are significant communication or health literacy barriers. Or, because there are challenging family dynamics that are impeding progress in establishing goals of care.
A physician should refer to specialty palliative care when there are multiple comorbid conditions that impact a patient’s prognosis and ability to tolerate treatments. These patients will need detailed symptom management and nuanced conversations about the delicate balance of maintaining quality of life and trying to address their malignancy while also avoiding treatments that may do more harm than good.
At the end of the day, all patients with serious illnesses deserve a palliative care approach to their care from all of their clinicians, not just from the palliative care team. By continuously honing and implementing primary palliative care skills, oncology teams can feel empowered to meet the needs of their patients themselves, strengthening their bond with their patients making truly patient-centered care much more likely.
Ms. D’Ambruoso is a hospice and palliative care nurse practitioner for UCLA Health Cancer Care, Santa Monica, Calif.
I recently met Jane, a 53-year-old woman with metastatic breast cancer. She was referred to me by the breast oncology team, which routinely refers all metastatic patients to our palliative care clinic.
Clocking in at under 20 minutes, my consultation with Jane might have been one of my shortest on record. Not only had the breast oncology team already addressed Jane’s symptoms, which mainly consisted of hot flashes and joint pain attributable to treatment with an aromatase inhibitor, but they had already started planning ahead for the future of her illness. Jane had completed an advance directive and had a realistic and hopeful perspective on how her illness would progress. She understood the goal of her treatment was to “keep the cancer asleep,” as she put it, and she was very clear about her own goals: to live long enough to see her granddaughter graduate from high school in 2 years and to take a long-awaited trip to Australia later in 2023.
There wasn’t much for me to do. In fact, I daresay that Jane really did not need to see a palliative care specialist because the primary palliative care she was receiving from the breast oncology team was superb. Jane was receiving excellent symptom management from a nurse practitioner and oncologist, plus a social worker provided her with coping strategies. She was already having conversations with her primary medical team and her family about what to expect in the future and how to plan ahead for all possible outcomes.
When should a patient be referred to palliative care?
Integrating palliative care into routine oncologic care need not always require the time and skill of a palliative care team for every patient. Oncology providers can provide basic palliative care services without consulting a palliative care specialist.
For example, if a primary care doctor tried to refer every patient with hypertension to cardiology, the cardiologist would probably say that primary care should be able to handle basic hypertension management. In my experience from working in an oncology clinic for the past 9 years, I’ve found that oncology providers don’t need to refer every advanced cancer patient to our palliative care program. Most oncologists have good communication skills and are more than capable of managing symptoms for patients.
But don’t get me wrong.
Palliative care for all?
In 2010, Jennifer S. Temel MD, published a landmark study in the New England Journal of Medicine that demonstrated significant improvements in quality of life and mood in patients with metastatic lung cancer who received concurrent palliative care. After the study was published many voices inside oncology and palliative care began to advocate for a “palliative care for all” approach to patients with metastatic disease. But this is often interpreted as “specialty palliative care for all,” rather than its original intended meaning that all patients with metastatic disease receive the essential elements of palliative care (biopsychosocial symptom support and conversations about goals of care) either through their primary oncology teams or, if needed, specialty palliative care teams.
The fact is that most specialty palliative care clinics do not have the manpower to meet the needs of all patients with advanced cancers, much less all patients living with serious illness. A main goal of integrating palliative care into routine outpatient health care has always been (and in my opinion, should continue to be) to enhance the primary palliative care skills of specialists, such as oncologists and cardiologists, who care for some of our sickest patients.
This could take many forms. For one, it can be helpful to screen patients for palliative care needs. The American College of Surgeons Commission on Cancer mandates distress screening for all patients as a condition of accreditation. Distress screening using a validated tool such as the National Comprehensive Cancer Network Distress Thermometer can differentiate patients who have minimal distress and may not need much additional support beyond what is provided by their oncology team from those whose distress feels unmanageable and overwhelming.
In terms of primary palliative care symptom management, most oncology teams I work with are comfortable prescribing basic medications for pain, nausea, constipation, and anxiety. They’re also comfortable referring oncology patients for nutrition needs while undergoing chemotherapy as well as to social work and spiritual care for emotional support and counseling.
Oncology teams should continually work on communications skills. They should use “Ask, Tell, Ask” to elicit prognostic awareness, convey critical information, and assess for recall and understanding at pivotal points in the cancer journey, such as when the disease progresses or the patient’s clinical condition changes. They should practice a normalizing script they can use to introduce advance care planning to their patients in the first few visits. When I meet with a patient for the first time, I usually begin by asking if they have prepared an advanced directive. If not, I ask if they’ve thought about who will make medical decisions for them should the need arise. If the patient has documented in writing their preference for care in an emergency situation, I ask for a copy for their chart.
When should patients be referred to a specialty palliative care program?
I tell our oncology teams to involve me after they have tried to intervene, but unsuccessfully because of the patient having intractable symptoms, such as pain, or the disease is not responding to treatments. Or, because there are significant communication or health literacy barriers. Or, because there are challenging family dynamics that are impeding progress in establishing goals of care.
A physician should refer to specialty palliative care when there are multiple comorbid conditions that impact a patient’s prognosis and ability to tolerate treatments. These patients will need detailed symptom management and nuanced conversations about the delicate balance of maintaining quality of life and trying to address their malignancy while also avoiding treatments that may do more harm than good.
At the end of the day, all patients with serious illnesses deserve a palliative care approach to their care from all of their clinicians, not just from the palliative care team. By continuously honing and implementing primary palliative care skills, oncology teams can feel empowered to meet the needs of their patients themselves, strengthening their bond with their patients making truly patient-centered care much more likely.
Ms. D’Ambruoso is a hospice and palliative care nurse practitioner for UCLA Health Cancer Care, Santa Monica, Calif.
I recently met Jane, a 53-year-old woman with metastatic breast cancer. She was referred to me by the breast oncology team, which routinely refers all metastatic patients to our palliative care clinic.
Clocking in at under 20 minutes, my consultation with Jane might have been one of my shortest on record. Not only had the breast oncology team already addressed Jane’s symptoms, which mainly consisted of hot flashes and joint pain attributable to treatment with an aromatase inhibitor, but they had already started planning ahead for the future of her illness. Jane had completed an advance directive and had a realistic and hopeful perspective on how her illness would progress. She understood the goal of her treatment was to “keep the cancer asleep,” as she put it, and she was very clear about her own goals: to live long enough to see her granddaughter graduate from high school in 2 years and to take a long-awaited trip to Australia later in 2023.
There wasn’t much for me to do. In fact, I daresay that Jane really did not need to see a palliative care specialist because the primary palliative care she was receiving from the breast oncology team was superb. Jane was receiving excellent symptom management from a nurse practitioner and oncologist, plus a social worker provided her with coping strategies. She was already having conversations with her primary medical team and her family about what to expect in the future and how to plan ahead for all possible outcomes.
When should a patient be referred to palliative care?
Integrating palliative care into routine oncologic care need not always require the time and skill of a palliative care team for every patient. Oncology providers can provide basic palliative care services without consulting a palliative care specialist.
For example, if a primary care doctor tried to refer every patient with hypertension to cardiology, the cardiologist would probably say that primary care should be able to handle basic hypertension management. In my experience from working in an oncology clinic for the past 9 years, I’ve found that oncology providers don’t need to refer every advanced cancer patient to our palliative care program. Most oncologists have good communication skills and are more than capable of managing symptoms for patients.
But don’t get me wrong.
Palliative care for all?
In 2010, Jennifer S. Temel MD, published a landmark study in the New England Journal of Medicine that demonstrated significant improvements in quality of life and mood in patients with metastatic lung cancer who received concurrent palliative care. After the study was published many voices inside oncology and palliative care began to advocate for a “palliative care for all” approach to patients with metastatic disease. But this is often interpreted as “specialty palliative care for all,” rather than its original intended meaning that all patients with metastatic disease receive the essential elements of palliative care (biopsychosocial symptom support and conversations about goals of care) either through their primary oncology teams or, if needed, specialty palliative care teams.
The fact is that most specialty palliative care clinics do not have the manpower to meet the needs of all patients with advanced cancers, much less all patients living with serious illness. A main goal of integrating palliative care into routine outpatient health care has always been (and in my opinion, should continue to be) to enhance the primary palliative care skills of specialists, such as oncologists and cardiologists, who care for some of our sickest patients.
This could take many forms. For one, it can be helpful to screen patients for palliative care needs. The American College of Surgeons Commission on Cancer mandates distress screening for all patients as a condition of accreditation. Distress screening using a validated tool such as the National Comprehensive Cancer Network Distress Thermometer can differentiate patients who have minimal distress and may not need much additional support beyond what is provided by their oncology team from those whose distress feels unmanageable and overwhelming.
In terms of primary palliative care symptom management, most oncology teams I work with are comfortable prescribing basic medications for pain, nausea, constipation, and anxiety. They’re also comfortable referring oncology patients for nutrition needs while undergoing chemotherapy as well as to social work and spiritual care for emotional support and counseling.
Oncology teams should continually work on communications skills. They should use “Ask, Tell, Ask” to elicit prognostic awareness, convey critical information, and assess for recall and understanding at pivotal points in the cancer journey, such as when the disease progresses or the patient’s clinical condition changes. They should practice a normalizing script they can use to introduce advance care planning to their patients in the first few visits. When I meet with a patient for the first time, I usually begin by asking if they have prepared an advanced directive. If not, I ask if they’ve thought about who will make medical decisions for them should the need arise. If the patient has documented in writing their preference for care in an emergency situation, I ask for a copy for their chart.
When should patients be referred to a specialty palliative care program?
I tell our oncology teams to involve me after they have tried to intervene, but unsuccessfully because of the patient having intractable symptoms, such as pain, or the disease is not responding to treatments. Or, because there are significant communication or health literacy barriers. Or, because there are challenging family dynamics that are impeding progress in establishing goals of care.
A physician should refer to specialty palliative care when there are multiple comorbid conditions that impact a patient’s prognosis and ability to tolerate treatments. These patients will need detailed symptom management and nuanced conversations about the delicate balance of maintaining quality of life and trying to address their malignancy while also avoiding treatments that may do more harm than good.
At the end of the day, all patients with serious illnesses deserve a palliative care approach to their care from all of their clinicians, not just from the palliative care team. By continuously honing and implementing primary palliative care skills, oncology teams can feel empowered to meet the needs of their patients themselves, strengthening their bond with their patients making truly patient-centered care much more likely.
Ms. D’Ambruoso is a hospice and palliative care nurse practitioner for UCLA Health Cancer Care, Santa Monica, Calif.
Manicure gone wrong leads to cancer diagnosis
. Now, she and her doctor are spreading the word about her ordeal as a lesson that speed and persistence in seeking treatment are the keys that make her type of cancer – squamous cell carcinoma – completely curable.
“She cut me, and the cut wasn’t just a regular cuticle cut. She cut me deep, and that was one of the first times that happened to me,” Grace Garcia, 50, told TODAY.com, recalling the November 2021 incident.
Ms. Garcia had been getting her nails done regularly for 20 years, she said, but happened to go to a different salon than her usual spot because she couldn’t get an appointment during the busy pre-Thanksgiving season. She doesn’t recall whether the technician opened packaging that signals unused tools.
She put antibiotic ointment on the cut, but it didn’t heal after a few days. Eventually, the skin closed and a darkened bump formed. It was painful. She went to her doctor, who said it was a “callus from writing,” she told TODAY.com. But it was on her ring finger, which didn’t seem connected to writing. Her doctor said to keep an eye on it.
Five months after the cut occurred, she mentioned it during a gynecology appointment and was referred to a dermatologist, who also advised keeping an eye on it. A wart developed. She went back to her primary care physician and then to another dermatologist. The spot was biopsied.
Squamous cell carcinoma is a common type of skin cancer, according to the American Academy of Dermatology. It can have many causes, but the cause in Ms. Garcia’s case was both very common and very rare: human papillomavirus, or HPV. HPV is a virus that infects millions of people every year, but it’s not a typical cause of skin cancer.
“It’s pretty rare for several reasons. Generally speaking, the strains that cause cancer from an HPV standpoint tend to be more sexually transmitted,” dermatologist Teo Soleymani told TODAY.com. “In Grace’s case, she had an injury, which became the portal of entry. So that thick skin that we have on our hands and feet that acts as a natural barrier against infections and things like that was no longer the case, and the virus was able to infect her skin.”
Dr. Soleymani said Ms. Garcia’s persistence to get answers likely saved her from losing a finger.
“Your outcomes are entirely dictated by how early you catch them, and very often they’re completely curable,” he said. “Her persistence – not only was she able to have a great outcome, she probably saved herself from having her finger amputated.”
. Now, she and her doctor are spreading the word about her ordeal as a lesson that speed and persistence in seeking treatment are the keys that make her type of cancer – squamous cell carcinoma – completely curable.
“She cut me, and the cut wasn’t just a regular cuticle cut. She cut me deep, and that was one of the first times that happened to me,” Grace Garcia, 50, told TODAY.com, recalling the November 2021 incident.
Ms. Garcia had been getting her nails done regularly for 20 years, she said, but happened to go to a different salon than her usual spot because she couldn’t get an appointment during the busy pre-Thanksgiving season. She doesn’t recall whether the technician opened packaging that signals unused tools.
She put antibiotic ointment on the cut, but it didn’t heal after a few days. Eventually, the skin closed and a darkened bump formed. It was painful. She went to her doctor, who said it was a “callus from writing,” she told TODAY.com. But it was on her ring finger, which didn’t seem connected to writing. Her doctor said to keep an eye on it.
Five months after the cut occurred, she mentioned it during a gynecology appointment and was referred to a dermatologist, who also advised keeping an eye on it. A wart developed. She went back to her primary care physician and then to another dermatologist. The spot was biopsied.
Squamous cell carcinoma is a common type of skin cancer, according to the American Academy of Dermatology. It can have many causes, but the cause in Ms. Garcia’s case was both very common and very rare: human papillomavirus, or HPV. HPV is a virus that infects millions of people every year, but it’s not a typical cause of skin cancer.
“It’s pretty rare for several reasons. Generally speaking, the strains that cause cancer from an HPV standpoint tend to be more sexually transmitted,” dermatologist Teo Soleymani told TODAY.com. “In Grace’s case, she had an injury, which became the portal of entry. So that thick skin that we have on our hands and feet that acts as a natural barrier against infections and things like that was no longer the case, and the virus was able to infect her skin.”
Dr. Soleymani said Ms. Garcia’s persistence to get answers likely saved her from losing a finger.
“Your outcomes are entirely dictated by how early you catch them, and very often they’re completely curable,” he said. “Her persistence – not only was she able to have a great outcome, she probably saved herself from having her finger amputated.”
. Now, she and her doctor are spreading the word about her ordeal as a lesson that speed and persistence in seeking treatment are the keys that make her type of cancer – squamous cell carcinoma – completely curable.
“She cut me, and the cut wasn’t just a regular cuticle cut. She cut me deep, and that was one of the first times that happened to me,” Grace Garcia, 50, told TODAY.com, recalling the November 2021 incident.
Ms. Garcia had been getting her nails done regularly for 20 years, she said, but happened to go to a different salon than her usual spot because she couldn’t get an appointment during the busy pre-Thanksgiving season. She doesn’t recall whether the technician opened packaging that signals unused tools.
She put antibiotic ointment on the cut, but it didn’t heal after a few days. Eventually, the skin closed and a darkened bump formed. It was painful. She went to her doctor, who said it was a “callus from writing,” she told TODAY.com. But it was on her ring finger, which didn’t seem connected to writing. Her doctor said to keep an eye on it.
Five months after the cut occurred, she mentioned it during a gynecology appointment and was referred to a dermatologist, who also advised keeping an eye on it. A wart developed. She went back to her primary care physician and then to another dermatologist. The spot was biopsied.
Squamous cell carcinoma is a common type of skin cancer, according to the American Academy of Dermatology. It can have many causes, but the cause in Ms. Garcia’s case was both very common and very rare: human papillomavirus, or HPV. HPV is a virus that infects millions of people every year, but it’s not a typical cause of skin cancer.
“It’s pretty rare for several reasons. Generally speaking, the strains that cause cancer from an HPV standpoint tend to be more sexually transmitted,” dermatologist Teo Soleymani told TODAY.com. “In Grace’s case, she had an injury, which became the portal of entry. So that thick skin that we have on our hands and feet that acts as a natural barrier against infections and things like that was no longer the case, and the virus was able to infect her skin.”
Dr. Soleymani said Ms. Garcia’s persistence to get answers likely saved her from losing a finger.
“Your outcomes are entirely dictated by how early you catch them, and very often they’re completely curable,” he said. “Her persistence – not only was she able to have a great outcome, she probably saved herself from having her finger amputated.”
Follicular Lymphoma Highlights From ASH 2022
Highlights in follicular lymphoma from the 2022 American Society of Hematology (ASH) Annual Meeting are discussed by Dr Thomas Rodgers of the Durham VA Medical Center.
Dr Rodgers begins with a prognostic model designed to evaluate the risk for disease progression in high-risk patients within 24 months of starting first-line treatment with the intention of better individualizing management in this group.
Next, he presents long-term phase 3 data comparing first-line rituximab with a watch-and-wait approach. After 12 years of follow-up, results showed no significant difference in overall survival between watch and wait, rituximab induction, and rituximab induction plus maintenance, suggesting to Dr Rodgers that individualized upfront management can lead to similarly excellent outcomes in patients with low tumor burden.
Turning to relapsed/refractory disease, Dr Rodgers cites a study comparing rituximab plus lenalidomide with rituximab plus placebo. The combination yielded superior results and more durable efficacy than did the control group.
He also discusses studies on the use of novel agent tazemetostat in combination with lenalidomide, and the bispecific monoclonal antibody mosunetuzumab as monotherapy. The US Food and Drug Administration approved mosunetuzumab in December, expanding the armamentarium for patients with follicular lymphoma who have undergone multiple lines of therapy.
--
Thomas Rodgers, MD, Assistant Professor, Department of Hematologic Malignancies and Cellular Therapy, Duke University; Staff Physician, Department of Hematology/Oncology, Durham VA Medical Center, Durham, North Carolina
Thomas Rodgers, MD, has disclosed no relevant financial relationships.
Highlights in follicular lymphoma from the 2022 American Society of Hematology (ASH) Annual Meeting are discussed by Dr Thomas Rodgers of the Durham VA Medical Center.
Dr Rodgers begins with a prognostic model designed to evaluate the risk for disease progression in high-risk patients within 24 months of starting first-line treatment with the intention of better individualizing management in this group.
Next, he presents long-term phase 3 data comparing first-line rituximab with a watch-and-wait approach. After 12 years of follow-up, results showed no significant difference in overall survival between watch and wait, rituximab induction, and rituximab induction plus maintenance, suggesting to Dr Rodgers that individualized upfront management can lead to similarly excellent outcomes in patients with low tumor burden.
Turning to relapsed/refractory disease, Dr Rodgers cites a study comparing rituximab plus lenalidomide with rituximab plus placebo. The combination yielded superior results and more durable efficacy than did the control group.
He also discusses studies on the use of novel agent tazemetostat in combination with lenalidomide, and the bispecific monoclonal antibody mosunetuzumab as monotherapy. The US Food and Drug Administration approved mosunetuzumab in December, expanding the armamentarium for patients with follicular lymphoma who have undergone multiple lines of therapy.
--
Thomas Rodgers, MD, Assistant Professor, Department of Hematologic Malignancies and Cellular Therapy, Duke University; Staff Physician, Department of Hematology/Oncology, Durham VA Medical Center, Durham, North Carolina
Thomas Rodgers, MD, has disclosed no relevant financial relationships.
Highlights in follicular lymphoma from the 2022 American Society of Hematology (ASH) Annual Meeting are discussed by Dr Thomas Rodgers of the Durham VA Medical Center.
Dr Rodgers begins with a prognostic model designed to evaluate the risk for disease progression in high-risk patients within 24 months of starting first-line treatment with the intention of better individualizing management in this group.
Next, he presents long-term phase 3 data comparing first-line rituximab with a watch-and-wait approach. After 12 years of follow-up, results showed no significant difference in overall survival between watch and wait, rituximab induction, and rituximab induction plus maintenance, suggesting to Dr Rodgers that individualized upfront management can lead to similarly excellent outcomes in patients with low tumor burden.
Turning to relapsed/refractory disease, Dr Rodgers cites a study comparing rituximab plus lenalidomide with rituximab plus placebo. The combination yielded superior results and more durable efficacy than did the control group.
He also discusses studies on the use of novel agent tazemetostat in combination with lenalidomide, and the bispecific monoclonal antibody mosunetuzumab as monotherapy. The US Food and Drug Administration approved mosunetuzumab in December, expanding the armamentarium for patients with follicular lymphoma who have undergone multiple lines of therapy.
--
Thomas Rodgers, MD, Assistant Professor, Department of Hematologic Malignancies and Cellular Therapy, Duke University; Staff Physician, Department of Hematology/Oncology, Durham VA Medical Center, Durham, North Carolina
Thomas Rodgers, MD, has disclosed no relevant financial relationships.

Chronic Lymphocytic Leukemia Highlights From ASH 2022
Reporting on chronic lymphocytic leukemia (CLL) highlights from the American Society of Hematology meetings, Dr Nicholas Burwick of the Puget Sound Veterans Administration Health Care System discusses studies ranging from first-line treatment to options for relapsed/refractory disease.
Dr Burwick cites a Veterans Health Administration study documenting the increasing movement toward novel agents as frontline treatment in CLL since 2018, and the emerging trend toward use of second-generation Bruton tyrosine kinase (BTK) inhibitors.
Next, he discusses a study examining the frontline combination of ibrutinib and venetoclax, which may help overcome poor prognosis associated with unmutated IGHV CLL.
In another study, the combination of venetoclax and ibrutinib was examined in high-risk patients treated with ibrutinib for 1 year. The results showed the combination deepens responses and even offers the potential for discontinuation of therapy.
Turning to relapsed/refractory disease, Dr Burwick highlights the latest results of pirtobrutinib, which continues to show efficacy among heavily pretreated patients regardless of prior therapy, reason for discontinuation, or mutation status.
Finally, he points to zanubrutinib, which demonstrated superiority to ibrutinib in a long-range study of progression-free survival.
--
Nicholas R. Burwick, MD, Associate Professor, Division of Hematology, Department of Medicine, University of Washington; Attending Physician, Division of Hematology, Department of Medicine, Puget Sound VA Health Care System, Seattle, Washington
Nicholas R. Burwick, MD, has disclosed no relevant financial relationships.
Reporting on chronic lymphocytic leukemia (CLL) highlights from the American Society of Hematology meetings, Dr Nicholas Burwick of the Puget Sound Veterans Administration Health Care System discusses studies ranging from first-line treatment to options for relapsed/refractory disease.
Dr Burwick cites a Veterans Health Administration study documenting the increasing movement toward novel agents as frontline treatment in CLL since 2018, and the emerging trend toward use of second-generation Bruton tyrosine kinase (BTK) inhibitors.
Next, he discusses a study examining the frontline combination of ibrutinib and venetoclax, which may help overcome poor prognosis associated with unmutated IGHV CLL.
In another study, the combination of venetoclax and ibrutinib was examined in high-risk patients treated with ibrutinib for 1 year. The results showed the combination deepens responses and even offers the potential for discontinuation of therapy.
Turning to relapsed/refractory disease, Dr Burwick highlights the latest results of pirtobrutinib, which continues to show efficacy among heavily pretreated patients regardless of prior therapy, reason for discontinuation, or mutation status.
Finally, he points to zanubrutinib, which demonstrated superiority to ibrutinib in a long-range study of progression-free survival.
--
Nicholas R. Burwick, MD, Associate Professor, Division of Hematology, Department of Medicine, University of Washington; Attending Physician, Division of Hematology, Department of Medicine, Puget Sound VA Health Care System, Seattle, Washington
Nicholas R. Burwick, MD, has disclosed no relevant financial relationships.
Reporting on chronic lymphocytic leukemia (CLL) highlights from the American Society of Hematology meetings, Dr Nicholas Burwick of the Puget Sound Veterans Administration Health Care System discusses studies ranging from first-line treatment to options for relapsed/refractory disease.
Dr Burwick cites a Veterans Health Administration study documenting the increasing movement toward novel agents as frontline treatment in CLL since 2018, and the emerging trend toward use of second-generation Bruton tyrosine kinase (BTK) inhibitors.
Next, he discusses a study examining the frontline combination of ibrutinib and venetoclax, which may help overcome poor prognosis associated with unmutated IGHV CLL.
In another study, the combination of venetoclax and ibrutinib was examined in high-risk patients treated with ibrutinib for 1 year. The results showed the combination deepens responses and even offers the potential for discontinuation of therapy.
Turning to relapsed/refractory disease, Dr Burwick highlights the latest results of pirtobrutinib, which continues to show efficacy among heavily pretreated patients regardless of prior therapy, reason for discontinuation, or mutation status.
Finally, he points to zanubrutinib, which demonstrated superiority to ibrutinib in a long-range study of progression-free survival.
--
Nicholas R. Burwick, MD, Associate Professor, Division of Hematology, Department of Medicine, University of Washington; Attending Physician, Division of Hematology, Department of Medicine, Puget Sound VA Health Care System, Seattle, Washington
Nicholas R. Burwick, MD, has disclosed no relevant financial relationships.

Kaposi’s sarcoma: Antiretroviral-related improvements in survival measured
than their uninfected counterparts, based on the first such analysis of the American College of Surgeons’ National Cancer Database.
One-year overall survival for all patients with Kaposi’s sarcoma (KS), 74.9% in 2004-2007, rose by 6.4 percentage points to 81.3% in 2016-2018, with the use of ART for HIV starting in 2008. Two-year survival was up by an even larger 8.3 percentage points: 68.0% to 76.3%, said Amar D. Desai of New Jersey Medical School, Newark, and Shari R. Lipner, MD, of Weill Cornell Medicine, New York.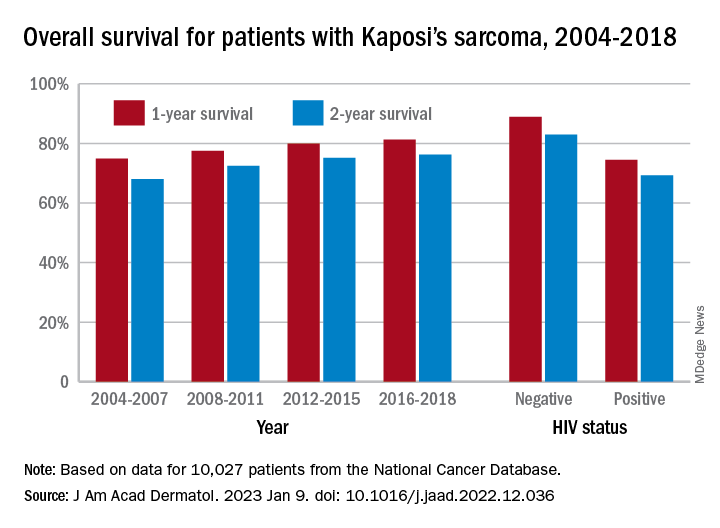
Since HIV-infected patients represented a much lower 46.7% of the Kaposi’s population in 2016-2018 than in 2004-2007 (70.5%), “better outcomes for all KS patients likely reflects advancements in ART, preventing many HIV+ patients from progressing to AIDS, changes in clinical practice with earlier treatment start, and more off-label treatments,” they wrote in the Journal of the American Academy of Dermatology.
Overall survival rates for the 10,027 patients with KS with data available in the National Cancer Database were 77.9% at 1 year and 72.4% at 2 years. HIV status had a significant (P < .0074) effect over the entire study period: One-year survival rates were 88.9% for HIV-negative and 74.5% for HIV-positive patients, and 2-year rates were 83.0% (HIV-negative) and 69.3% (HIV-positive), the investigators reported in what they called “the largest analysis since the advent of antiretroviral therapy for HIV in 2008.”
The improvement in overall survival, along with the continued differences in survival between HIV infected and noninfected patients, indicate that “dermatologists, as part of a multidisciplinary team including oncologists and infectious disease physicians, can play significant roles in early KS diagnosis,” Mr. Desai and Dr. Lipner said.
Mr. Desai had no conflicts of interest to report. Dr. Lipner has served as a consultant for Ortho-Dermatologics, Hoth Therapeutics, and BelleTorus Corporation.
than their uninfected counterparts, based on the first such analysis of the American College of Surgeons’ National Cancer Database.
One-year overall survival for all patients with Kaposi’s sarcoma (KS), 74.9% in 2004-2007, rose by 6.4 percentage points to 81.3% in 2016-2018, with the use of ART for HIV starting in 2008. Two-year survival was up by an even larger 8.3 percentage points: 68.0% to 76.3%, said Amar D. Desai of New Jersey Medical School, Newark, and Shari R. Lipner, MD, of Weill Cornell Medicine, New York.
Since HIV-infected patients represented a much lower 46.7% of the Kaposi’s population in 2016-2018 than in 2004-2007 (70.5%), “better outcomes for all KS patients likely reflects advancements in ART, preventing many HIV+ patients from progressing to AIDS, changes in clinical practice with earlier treatment start, and more off-label treatments,” they wrote in the Journal of the American Academy of Dermatology.
Overall survival rates for the 10,027 patients with KS with data available in the National Cancer Database were 77.9% at 1 year and 72.4% at 2 years. HIV status had a significant (P < .0074) effect over the entire study period: One-year survival rates were 88.9% for HIV-negative and 74.5% for HIV-positive patients, and 2-year rates were 83.0% (HIV-negative) and 69.3% (HIV-positive), the investigators reported in what they called “the largest analysis since the advent of antiretroviral therapy for HIV in 2008.”
The improvement in overall survival, along with the continued differences in survival between HIV infected and noninfected patients, indicate that “dermatologists, as part of a multidisciplinary team including oncologists and infectious disease physicians, can play significant roles in early KS diagnosis,” Mr. Desai and Dr. Lipner said.
Mr. Desai had no conflicts of interest to report. Dr. Lipner has served as a consultant for Ortho-Dermatologics, Hoth Therapeutics, and BelleTorus Corporation.
than their uninfected counterparts, based on the first such analysis of the American College of Surgeons’ National Cancer Database.
One-year overall survival for all patients with Kaposi’s sarcoma (KS), 74.9% in 2004-2007, rose by 6.4 percentage points to 81.3% in 2016-2018, with the use of ART for HIV starting in 2008. Two-year survival was up by an even larger 8.3 percentage points: 68.0% to 76.3%, said Amar D. Desai of New Jersey Medical School, Newark, and Shari R. Lipner, MD, of Weill Cornell Medicine, New York.
Since HIV-infected patients represented a much lower 46.7% of the Kaposi’s population in 2016-2018 than in 2004-2007 (70.5%), “better outcomes for all KS patients likely reflects advancements in ART, preventing many HIV+ patients from progressing to AIDS, changes in clinical practice with earlier treatment start, and more off-label treatments,” they wrote in the Journal of the American Academy of Dermatology.
Overall survival rates for the 10,027 patients with KS with data available in the National Cancer Database were 77.9% at 1 year and 72.4% at 2 years. HIV status had a significant (P < .0074) effect over the entire study period: One-year survival rates were 88.9% for HIV-negative and 74.5% for HIV-positive patients, and 2-year rates were 83.0% (HIV-negative) and 69.3% (HIV-positive), the investigators reported in what they called “the largest analysis since the advent of antiretroviral therapy for HIV in 2008.”
The improvement in overall survival, along with the continued differences in survival between HIV infected and noninfected patients, indicate that “dermatologists, as part of a multidisciplinary team including oncologists and infectious disease physicians, can play significant roles in early KS diagnosis,” Mr. Desai and Dr. Lipner said.
Mr. Desai had no conflicts of interest to report. Dr. Lipner has served as a consultant for Ortho-Dermatologics, Hoth Therapeutics, and BelleTorus Corporation.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
‘Low-value’ prostate cancer screening prevalent in primary care
Yet a new study shows that testing for prostate-specific antigen (PSA) and also digital rectal examinations (DRE) are both carried out frequently in older men, even when there is no indication for such testing.
“As a man ages, the risk for a false-positive result increases,” said lead author Chris Gillette, PhD, associate professor of physician assistant studies at Wake Forest University, Winston-Salem, N.C., in a statement
The study authors looked at primary care visits for men who were age 70 or older, and found that, per 100 visits, there were 6.7 PSA tests and 1.6 DRE performed.
Dr. Gillette and colleagues emphasized the importance of their findings. Whereas prior studies have relied on commercially insured men or patient-reported rates of PSA testing, they used a nationally representative clinical dataset that is much more inclusive, as it includes men who are also uninsured or insured through traditional Medicare.
The study was published online in the Journal of the American Board of Family Medicine.
Screening for prostate cancer has been much debated, and the guidelines have changed in recent years. In the period 2012-2018, the U.S. Preventive Services Task Force recommended against PSA-based screening in all men, but then the guidelines changed, and the USPSTF subsequently endorsed individualized screening in those aged 55-69 years after a shared decision-making discussion. That same 2018 update also recommends against PSA screening in men over the age of 70.
In addition, the American Urological Association has recommended against PSA-based prostate cancer screening for men over the age of 70 since 2013.
Previous studies have shown that clinicians are not adhering to the guidelines. An analysis conducted in March 2022 found that about one in four accredited U.S. cancer centers fails to follow national guidelines for PSA testing to screen for prostate cancer. Contrary to national guidelines, which advocate shared decision-making, 22% of centers recommend all men universally initiate PSA screening at either age 50 or 55 and another 4% of centers recommend this before age 50, earlier than the guidelines advise.
In the current study, Dr. Gillette and colleagues conducted a secondary analysis of the National Ambulatory Medical Care Survey datasets from 2013 to 2016 and 2018. The dataset is a nationally representative sample of visits to nonfederal, office-based physician clinics. This analysis was restricted to male patients aged 70 years and older who visited a primary care clinic.
The team found that health care professionals who order a lot of tests are more likely to order low-value screening such as PSA and DRE.
The data also showed that when there were a higher number of services ordered/provided, the patients were significantly more likely to receive a low-value PSA (odds ratio, 1.49) and a low-value DRE (OR, 1.37). In contrast, patients who had more previous visits to the clinician were less likely to receive a low-value DRE (OR, 0.92).
Overall, there a decline in low-value PSA screening after 2014, but this trend was not seen for DRE during primary care visits.
Speculating as to why these low-value tests are being carried out, Dr. Gillette suggested that health care professionals might be responding to patient requests when ordering these screening tests, or they may be using what’s known as a “shotgun” approach to medical testing where all possible tests are ordered during a medical visit.
“However, as health care systems move toward a more value-based care system – where the benefit of services provided outweighs any risks – clinicians need to engage patients in these discussions on the complexity of this testing,” he commented. “Ultimately, when and if to screen is a decision best left between a provider and the patient.”
There was no outside funding and the authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Yet a new study shows that testing for prostate-specific antigen (PSA) and also digital rectal examinations (DRE) are both carried out frequently in older men, even when there is no indication for such testing.
“As a man ages, the risk for a false-positive result increases,” said lead author Chris Gillette, PhD, associate professor of physician assistant studies at Wake Forest University, Winston-Salem, N.C., in a statement
The study authors looked at primary care visits for men who were age 70 or older, and found that, per 100 visits, there were 6.7 PSA tests and 1.6 DRE performed.
Dr. Gillette and colleagues emphasized the importance of their findings. Whereas prior studies have relied on commercially insured men or patient-reported rates of PSA testing, they used a nationally representative clinical dataset that is much more inclusive, as it includes men who are also uninsured or insured through traditional Medicare.
The study was published online in the Journal of the American Board of Family Medicine.
Screening for prostate cancer has been much debated, and the guidelines have changed in recent years. In the period 2012-2018, the U.S. Preventive Services Task Force recommended against PSA-based screening in all men, but then the guidelines changed, and the USPSTF subsequently endorsed individualized screening in those aged 55-69 years after a shared decision-making discussion. That same 2018 update also recommends against PSA screening in men over the age of 70.
In addition, the American Urological Association has recommended against PSA-based prostate cancer screening for men over the age of 70 since 2013.
Previous studies have shown that clinicians are not adhering to the guidelines. An analysis conducted in March 2022 found that about one in four accredited U.S. cancer centers fails to follow national guidelines for PSA testing to screen for prostate cancer. Contrary to national guidelines, which advocate shared decision-making, 22% of centers recommend all men universally initiate PSA screening at either age 50 or 55 and another 4% of centers recommend this before age 50, earlier than the guidelines advise.
In the current study, Dr. Gillette and colleagues conducted a secondary analysis of the National Ambulatory Medical Care Survey datasets from 2013 to 2016 and 2018. The dataset is a nationally representative sample of visits to nonfederal, office-based physician clinics. This analysis was restricted to male patients aged 70 years and older who visited a primary care clinic.
The team found that health care professionals who order a lot of tests are more likely to order low-value screening such as PSA and DRE.
The data also showed that when there were a higher number of services ordered/provided, the patients were significantly more likely to receive a low-value PSA (odds ratio, 1.49) and a low-value DRE (OR, 1.37). In contrast, patients who had more previous visits to the clinician were less likely to receive a low-value DRE (OR, 0.92).
Overall, there a decline in low-value PSA screening after 2014, but this trend was not seen for DRE during primary care visits.
Speculating as to why these low-value tests are being carried out, Dr. Gillette suggested that health care professionals might be responding to patient requests when ordering these screening tests, or they may be using what’s known as a “shotgun” approach to medical testing where all possible tests are ordered during a medical visit.
“However, as health care systems move toward a more value-based care system – where the benefit of services provided outweighs any risks – clinicians need to engage patients in these discussions on the complexity of this testing,” he commented. “Ultimately, when and if to screen is a decision best left between a provider and the patient.”
There was no outside funding and the authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Yet a new study shows that testing for prostate-specific antigen (PSA) and also digital rectal examinations (DRE) are both carried out frequently in older men, even when there is no indication for such testing.
“As a man ages, the risk for a false-positive result increases,” said lead author Chris Gillette, PhD, associate professor of physician assistant studies at Wake Forest University, Winston-Salem, N.C., in a statement
The study authors looked at primary care visits for men who were age 70 or older, and found that, per 100 visits, there were 6.7 PSA tests and 1.6 DRE performed.
Dr. Gillette and colleagues emphasized the importance of their findings. Whereas prior studies have relied on commercially insured men or patient-reported rates of PSA testing, they used a nationally representative clinical dataset that is much more inclusive, as it includes men who are also uninsured or insured through traditional Medicare.
The study was published online in the Journal of the American Board of Family Medicine.
Screening for prostate cancer has been much debated, and the guidelines have changed in recent years. In the period 2012-2018, the U.S. Preventive Services Task Force recommended against PSA-based screening in all men, but then the guidelines changed, and the USPSTF subsequently endorsed individualized screening in those aged 55-69 years after a shared decision-making discussion. That same 2018 update also recommends against PSA screening in men over the age of 70.
In addition, the American Urological Association has recommended against PSA-based prostate cancer screening for men over the age of 70 since 2013.
Previous studies have shown that clinicians are not adhering to the guidelines. An analysis conducted in March 2022 found that about one in four accredited U.S. cancer centers fails to follow national guidelines for PSA testing to screen for prostate cancer. Contrary to national guidelines, which advocate shared decision-making, 22% of centers recommend all men universally initiate PSA screening at either age 50 or 55 and another 4% of centers recommend this before age 50, earlier than the guidelines advise.
In the current study, Dr. Gillette and colleagues conducted a secondary analysis of the National Ambulatory Medical Care Survey datasets from 2013 to 2016 and 2018. The dataset is a nationally representative sample of visits to nonfederal, office-based physician clinics. This analysis was restricted to male patients aged 70 years and older who visited a primary care clinic.
The team found that health care professionals who order a lot of tests are more likely to order low-value screening such as PSA and DRE.
The data also showed that when there were a higher number of services ordered/provided, the patients were significantly more likely to receive a low-value PSA (odds ratio, 1.49) and a low-value DRE (OR, 1.37). In contrast, patients who had more previous visits to the clinician were less likely to receive a low-value DRE (OR, 0.92).
Overall, there a decline in low-value PSA screening after 2014, but this trend was not seen for DRE during primary care visits.
Speculating as to why these low-value tests are being carried out, Dr. Gillette suggested that health care professionals might be responding to patient requests when ordering these screening tests, or they may be using what’s known as a “shotgun” approach to medical testing where all possible tests are ordered during a medical visit.
“However, as health care systems move toward a more value-based care system – where the benefit of services provided outweighs any risks – clinicians need to engage patients in these discussions on the complexity of this testing,” he commented. “Ultimately, when and if to screen is a decision best left between a provider and the patient.”
There was no outside funding and the authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN BOARD OF FAMILY MEDICINE
Small study finds high dose vitamin D relieved toxic erythema of chemotherapy
seen on an inpatient dermatology consultative service.
Currently, chemotherapy cessation, delay, or dose modification are the “only reliable methods of resolving TEC,” and supportive agents such as topical corticosteroids, topical keratolytics, and pain control are associated with variable and “relatively slow improvement involving 2 to 4 weeks of recovery after chemotherapy interruption,” Cuong V. Nguyen, MD, of the department of dermatology at Northwestern University, Chicago, and colleagues, wrote in a research letter.
Onset of TEC in the six patients occurred a mean of 8.5 days after chemotherapy. Vitamin D – 50,000 IU for one patient and 100,000 IU for the others – was administered a mean of 4.3 days from rash onset and again in 7 days. Triamcinolone, 0.1%, or clobetasol, 0.05%, ointments were also prescribed.
All patients experienced symptomatic improvement in pain, pruritus, or swelling within a day of the first vitamin D treatment, and improvement in redness within 1 to 4 days, the authors said. The second treatment was administered for residual symptoms.
Adam Friedman, MD, professor and chair of dermatology and director of the supportive oncodermatology clinic at George Washington University, Washington, said that supporting patients through the “expected, disabling and often treatment-limiting side effects of oncologic therapies” is an area that is “in its infancy” and is characterized by limited evidence-based approaches.
“Creativity is therefore a must,” he said, commenting on the research letter. “Practice starts with anecdote, and this is certainly an exciting finding ... I look forward to trialing this with our patients at GW.”
Five of the six patients had a hematologic condition that required induction chemotherapy before hematopoietic stem cell transplant, and one was receiving regorafenib for treatment of glioblastoma multiforme. Diagnosis of TEC was established by clinical presentation, and five of the six patients underwent a biopsy. Biopsy findings were consistent with a TEC diagnosis in three patients, and showed nonspecific perivascular dermatitis in two, the investigators reported.
Further research is needed to determine optimal dosing, “delineate safety concerns and potential role in cancer treatment, and establish whether a durable response in patients with continuous chemotherapy, such as in an outpatient setting, is possible,” they said.
Dr. Nguyen and his coauthors reported no conflict of interest disclosures.
seen on an inpatient dermatology consultative service.
Currently, chemotherapy cessation, delay, or dose modification are the “only reliable methods of resolving TEC,” and supportive agents such as topical corticosteroids, topical keratolytics, and pain control are associated with variable and “relatively slow improvement involving 2 to 4 weeks of recovery after chemotherapy interruption,” Cuong V. Nguyen, MD, of the department of dermatology at Northwestern University, Chicago, and colleagues, wrote in a research letter.
Onset of TEC in the six patients occurred a mean of 8.5 days after chemotherapy. Vitamin D – 50,000 IU for one patient and 100,000 IU for the others – was administered a mean of 4.3 days from rash onset and again in 7 days. Triamcinolone, 0.1%, or clobetasol, 0.05%, ointments were also prescribed.
All patients experienced symptomatic improvement in pain, pruritus, or swelling within a day of the first vitamin D treatment, and improvement in redness within 1 to 4 days, the authors said. The second treatment was administered for residual symptoms.
Adam Friedman, MD, professor and chair of dermatology and director of the supportive oncodermatology clinic at George Washington University, Washington, said that supporting patients through the “expected, disabling and often treatment-limiting side effects of oncologic therapies” is an area that is “in its infancy” and is characterized by limited evidence-based approaches.
“Creativity is therefore a must,” he said, commenting on the research letter. “Practice starts with anecdote, and this is certainly an exciting finding ... I look forward to trialing this with our patients at GW.”
Five of the six patients had a hematologic condition that required induction chemotherapy before hematopoietic stem cell transplant, and one was receiving regorafenib for treatment of glioblastoma multiforme. Diagnosis of TEC was established by clinical presentation, and five of the six patients underwent a biopsy. Biopsy findings were consistent with a TEC diagnosis in three patients, and showed nonspecific perivascular dermatitis in two, the investigators reported.
Further research is needed to determine optimal dosing, “delineate safety concerns and potential role in cancer treatment, and establish whether a durable response in patients with continuous chemotherapy, such as in an outpatient setting, is possible,” they said.
Dr. Nguyen and his coauthors reported no conflict of interest disclosures.
seen on an inpatient dermatology consultative service.
Currently, chemotherapy cessation, delay, or dose modification are the “only reliable methods of resolving TEC,” and supportive agents such as topical corticosteroids, topical keratolytics, and pain control are associated with variable and “relatively slow improvement involving 2 to 4 weeks of recovery after chemotherapy interruption,” Cuong V. Nguyen, MD, of the department of dermatology at Northwestern University, Chicago, and colleagues, wrote in a research letter.
Onset of TEC in the six patients occurred a mean of 8.5 days after chemotherapy. Vitamin D – 50,000 IU for one patient and 100,000 IU for the others – was administered a mean of 4.3 days from rash onset and again in 7 days. Triamcinolone, 0.1%, or clobetasol, 0.05%, ointments were also prescribed.
All patients experienced symptomatic improvement in pain, pruritus, or swelling within a day of the first vitamin D treatment, and improvement in redness within 1 to 4 days, the authors said. The second treatment was administered for residual symptoms.
Adam Friedman, MD, professor and chair of dermatology and director of the supportive oncodermatology clinic at George Washington University, Washington, said that supporting patients through the “expected, disabling and often treatment-limiting side effects of oncologic therapies” is an area that is “in its infancy” and is characterized by limited evidence-based approaches.
“Creativity is therefore a must,” he said, commenting on the research letter. “Practice starts with anecdote, and this is certainly an exciting finding ... I look forward to trialing this with our patients at GW.”
Five of the six patients had a hematologic condition that required induction chemotherapy before hematopoietic stem cell transplant, and one was receiving regorafenib for treatment of glioblastoma multiforme. Diagnosis of TEC was established by clinical presentation, and five of the six patients underwent a biopsy. Biopsy findings were consistent with a TEC diagnosis in three patients, and showed nonspecific perivascular dermatitis in two, the investigators reported.
Further research is needed to determine optimal dosing, “delineate safety concerns and potential role in cancer treatment, and establish whether a durable response in patients with continuous chemotherapy, such as in an outpatient setting, is possible,” they said.
Dr. Nguyen and his coauthors reported no conflict of interest disclosures.
FROM JAMA DERMATOLOGY
Lifestyle changes may reduce colorectal cancer risk
Changes regarding smoking, drinking, body weight, and physical activity may alter the risk for colorectal cancer (CRC), the results of a study on a large European cohort suggest.
“This is a clear message that practicing clinicians and gastroenterologists could give to their patients and to CRC screening participants to improve CRC prevention,” write Edoardo Botteri, PhD, Cancer Registry of Norway, Oslo, and colleagues in an article published in the American Journal of Gastroenterology.
Previous studies have shown a correlation between cancer in general and unhealthy lifestyle factors. They have also shown an association between weight gain and an increased risk for CRC and a reduced risk with smoking cessation. But Dr. Botteri and colleagues could not find any published research on the association of other lifestyle factors and the risk for CRC specifically, they write.
To help fill this gap, they followed 295,865 people who participated in the European Prospective Investigation into Cancer (EPIC) for a median of 7.8 years. The participants were mostly aged from 35 to 70 years and lived in Denmark, France, Germany, Greece, Italy, the Netherlands, Norway, Spain, Sweden, and the United Kingdom.
The researchers calculated a healthy lifestyle index (HLI) score on the basis of smoking status, alcohol consumption, body mass index, and physical activity. The median time between baseline and the follow-up questionnaire was 5.7 years.
They awarded points as indicated in the following table.
Participants’ scores ranged from 0 to 16. At baseline, the mean HLI score was 10.04. It dipped slightly to 9.95 at follow-up.
Men had more favorable changes than women, and the associations between the HLI score and CRC risk were only statistically significant among men.
Overall, a 1-unit increase in the HLI score was associated with a 3% lower risk for CRC.
When the HLI scores were grouped into tertiles, improvements from an “unfavorable lifestyle” (0-9) to a “favorable lifestyle” (12-16) were associated with a 23% lower risk for CRC (compared with no change). Likewise, a decline from a “favorable lifestyle” to an “unfavorable lifestyle” was associated with a 34% higher risk.
Changes in the BMI score from baseline showed a trend toward an association with CRC risk.
Decreases in alcohol consumption were significantly associated with a reduction in CRC risk among participants aged 55 years or younger at baseline.
Increases in physical activity were significantly associated with a lower risk for proximal colon cancer, especially in younger participants.
On the other hand, reductions in smoking were associated with an increase in CRC risk. This correlation might be the result of “inverse causation,” the researchers note; that is, people may have quit smoking because they experienced early symptoms of CRC. Smoking had only a marginal influence on the HLI calculations in this study because only a small proportion of participants changed their smoking rates.
Information on diet was collected only at baseline, so changes in this factor could not be measured. The researchers adjusted their analysis for diet at baseline, but they acknowledge that their inability to incorporate diet into the HLI score was a limitation of the study.
Similarly, they used education as a marker of socioeconomic status but acknowledge that this is only a proxy.
“The HLI score may therefore not accurately capture the complex relationship between lifestyle habits and risk for CRC,” they write.
Still, if the results of this observational study are confirmed by other research, the findings could provide evidence to design intervention studies to prevent CRC, they conclude.
The study was supported by the grant LIBERTY from the French Institut National du Cancer. Financial supporters of the national cohorts and the coordination of EPIC are listed in the published study. The researchers reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Changes regarding smoking, drinking, body weight, and physical activity may alter the risk for colorectal cancer (CRC), the results of a study on a large European cohort suggest.
“This is a clear message that practicing clinicians and gastroenterologists could give to their patients and to CRC screening participants to improve CRC prevention,” write Edoardo Botteri, PhD, Cancer Registry of Norway, Oslo, and colleagues in an article published in the American Journal of Gastroenterology.
Previous studies have shown a correlation between cancer in general and unhealthy lifestyle factors. They have also shown an association between weight gain and an increased risk for CRC and a reduced risk with smoking cessation. But Dr. Botteri and colleagues could not find any published research on the association of other lifestyle factors and the risk for CRC specifically, they write.
To help fill this gap, they followed 295,865 people who participated in the European Prospective Investigation into Cancer (EPIC) for a median of 7.8 years. The participants were mostly aged from 35 to 70 years and lived in Denmark, France, Germany, Greece, Italy, the Netherlands, Norway, Spain, Sweden, and the United Kingdom.
The researchers calculated a healthy lifestyle index (HLI) score on the basis of smoking status, alcohol consumption, body mass index, and physical activity. The median time between baseline and the follow-up questionnaire was 5.7 years.
They awarded points as indicated in the following table.
Participants’ scores ranged from 0 to 16. At baseline, the mean HLI score was 10.04. It dipped slightly to 9.95 at follow-up.
Men had more favorable changes than women, and the associations between the HLI score and CRC risk were only statistically significant among men.
Overall, a 1-unit increase in the HLI score was associated with a 3% lower risk for CRC.
When the HLI scores were grouped into tertiles, improvements from an “unfavorable lifestyle” (0-9) to a “favorable lifestyle” (12-16) were associated with a 23% lower risk for CRC (compared with no change). Likewise, a decline from a “favorable lifestyle” to an “unfavorable lifestyle” was associated with a 34% higher risk.
Changes in the BMI score from baseline showed a trend toward an association with CRC risk.
Decreases in alcohol consumption were significantly associated with a reduction in CRC risk among participants aged 55 years or younger at baseline.
Increases in physical activity were significantly associated with a lower risk for proximal colon cancer, especially in younger participants.
On the other hand, reductions in smoking were associated with an increase in CRC risk. This correlation might be the result of “inverse causation,” the researchers note; that is, people may have quit smoking because they experienced early symptoms of CRC. Smoking had only a marginal influence on the HLI calculations in this study because only a small proportion of participants changed their smoking rates.
Information on diet was collected only at baseline, so changes in this factor could not be measured. The researchers adjusted their analysis for diet at baseline, but they acknowledge that their inability to incorporate diet into the HLI score was a limitation of the study.
Similarly, they used education as a marker of socioeconomic status but acknowledge that this is only a proxy.
“The HLI score may therefore not accurately capture the complex relationship between lifestyle habits and risk for CRC,” they write.
Still, if the results of this observational study are confirmed by other research, the findings could provide evidence to design intervention studies to prevent CRC, they conclude.
The study was supported by the grant LIBERTY from the French Institut National du Cancer. Financial supporters of the national cohorts and the coordination of EPIC are listed in the published study. The researchers reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Changes regarding smoking, drinking, body weight, and physical activity may alter the risk for colorectal cancer (CRC), the results of a study on a large European cohort suggest.
“This is a clear message that practicing clinicians and gastroenterologists could give to their patients and to CRC screening participants to improve CRC prevention,” write Edoardo Botteri, PhD, Cancer Registry of Norway, Oslo, and colleagues in an article published in the American Journal of Gastroenterology.
Previous studies have shown a correlation between cancer in general and unhealthy lifestyle factors. They have also shown an association between weight gain and an increased risk for CRC and a reduced risk with smoking cessation. But Dr. Botteri and colleagues could not find any published research on the association of other lifestyle factors and the risk for CRC specifically, they write.
To help fill this gap, they followed 295,865 people who participated in the European Prospective Investigation into Cancer (EPIC) for a median of 7.8 years. The participants were mostly aged from 35 to 70 years and lived in Denmark, France, Germany, Greece, Italy, the Netherlands, Norway, Spain, Sweden, and the United Kingdom.
The researchers calculated a healthy lifestyle index (HLI) score on the basis of smoking status, alcohol consumption, body mass index, and physical activity. The median time between baseline and the follow-up questionnaire was 5.7 years.
They awarded points as indicated in the following table.
Participants’ scores ranged from 0 to 16. At baseline, the mean HLI score was 10.04. It dipped slightly to 9.95 at follow-up.
Men had more favorable changes than women, and the associations between the HLI score and CRC risk were only statistically significant among men.
Overall, a 1-unit increase in the HLI score was associated with a 3% lower risk for CRC.
When the HLI scores were grouped into tertiles, improvements from an “unfavorable lifestyle” (0-9) to a “favorable lifestyle” (12-16) were associated with a 23% lower risk for CRC (compared with no change). Likewise, a decline from a “favorable lifestyle” to an “unfavorable lifestyle” was associated with a 34% higher risk.
Changes in the BMI score from baseline showed a trend toward an association with CRC risk.
Decreases in alcohol consumption were significantly associated with a reduction in CRC risk among participants aged 55 years or younger at baseline.
Increases in physical activity were significantly associated with a lower risk for proximal colon cancer, especially in younger participants.
On the other hand, reductions in smoking were associated with an increase in CRC risk. This correlation might be the result of “inverse causation,” the researchers note; that is, people may have quit smoking because they experienced early symptoms of CRC. Smoking had only a marginal influence on the HLI calculations in this study because only a small proportion of participants changed their smoking rates.
Information on diet was collected only at baseline, so changes in this factor could not be measured. The researchers adjusted their analysis for diet at baseline, but they acknowledge that their inability to incorporate diet into the HLI score was a limitation of the study.
Similarly, they used education as a marker of socioeconomic status but acknowledge that this is only a proxy.
“The HLI score may therefore not accurately capture the complex relationship between lifestyle habits and risk for CRC,” they write.
Still, if the results of this observational study are confirmed by other research, the findings could provide evidence to design intervention studies to prevent CRC, they conclude.
The study was supported by the grant LIBERTY from the French Institut National du Cancer. Financial supporters of the national cohorts and the coordination of EPIC are listed in the published study. The researchers reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF GASTROENTEROLOGY



