User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
COVID-19 vaccines do not trigger sudden hearing loss: Study
Anecdotal reports have linked the vaccines against COVID-19 to the sudden loss of hearing in some people. But a new study has found no evidence for such a connection with any of the three approved shots.
The analysis of data from the Centers for Disease Control and Prevention’s Vaccine Adverse Event Reporting System (VAERS) found that
“We’re not finding a signal,” said Eric J. Formeister, MD, a neurotology fellow at the Johns Hopkins University, Baltimore, and the first author of the U.S. study, which appeared Feb. 24 in JAMA Otolaryngology – Head and Neck Surgery.
Dr. Formeister and colleagues undertook the study in response to reports of hearing problems, including hearing loss and tinnitus, that occurred soon after COVID-19 vaccination.
They analyzed reports of sudden hearing loss, experienced within 21 days of vaccination, logged in VAERS. Anyone can report a potential event to the database, which does not require medical documentation in support of the adverse event. To minimize potential misdiagnoses, Dr. Formeister and colleagues reviewed only those reports that indicated that a doctor had diagnosed sudden hearing loss, leaving 555 cases (305 in women; mean age 54 years) between December 2020 and July 2021.
Dividing these reports by the total doses of vaccines administered in the United States during that period yielded an incidence rate of 0.6 cases of sudden hearing loss for every 100,000 people, Dr. Formeister and colleagues reported.
When the researchers divided all cases of hearing loss in the VAERS database (2,170) by the number of people who had received two doses of vaccine, the incidence rate increased to 28 per 100,000 people. For comparison, the authors reported, the incidence of sudden hearing loss within the United States population is between 11 and 77 per 100,000 people, depending on age.
“There was not an increase in cases of sudden [sensorineural] hearing loss associated with COVID-19 vaccination compared to previously published reports before the COVID-19 vaccination era,” study coauthor Elliott D. Kozin, MD, assistant professor of otolaryngology–head and neck surgery at Harvard Medical School, Boston, said in an interview.
Another reassuring sign: If hearing loss were linked to the vaccines, the researchers said, they would expect to see an increase in the number of complaints in lockstep with an increase in the number of doses administered. However, the opposite was true. “[T]he rate of reports per 100,000 doses decreased across the vaccination period, despite large concomitant increases in the absolute number of vaccine doses administered per week,” the researchers reported.
They also looked at case reports of 21 men and women who had experienced sudden hearing loss after having received COVID-19 vaccines, to see if they could discern any clinically relevant signs of people most likely to experience the adverse event. However, the group had a range of preexisting conditions and varying times after receiving a vaccine when their hearing loss occurred, leading Dr. Formeister’s team to conclude that they could find no clear markers of risk.
“When we examined patients across several institutions, there was no obvious pattern. The patient demographics and clinical findings were variable,” Dr. Kozin said. A provisional interpretation of this data, he added, is that no link exists between COVID-19 vaccination and predictable hearing deficits, although the analysis covered a small number of patients.
“Association does not necessarily imply a causal relationship,” said Michael Brenner, MD, FACS, associate professor of otolaryngology–head and neck surgery at the University of Michigan, Ann Arbor. Dr. Brenner, who was not involved in the study, said any hearing loss attributed to the COVID-19 vaccines could have had other causes besides the injections.
But a second study, also published in JAMA Otolaryngology – Head and Neck Surgery on Feb. 24, leaves open the possibility of a link. Researchers in Israel looked for increases in steroid prescriptions used to treat sudden hearing loss as vaccination with the Pfizer version of the shot became widespread in that country. Their conclusion: The vaccine might be associated with a slightly increased risk of sudden hearing loss, although if so, that risk is likely “very small” and the benefits of vaccination “outweigh its potential association” with the side effect.
Dr. Brenner agreed. “The evidence supports [the] clear public health benefit of COVID-19 vaccination, and the scale of those benefits dwarfs associations with hearing, which are of uncertain significance,” he said.
A version of this article first appeared on Medscape.com.
Anecdotal reports have linked the vaccines against COVID-19 to the sudden loss of hearing in some people. But a new study has found no evidence for such a connection with any of the three approved shots.
The analysis of data from the Centers for Disease Control and Prevention’s Vaccine Adverse Event Reporting System (VAERS) found that
“We’re not finding a signal,” said Eric J. Formeister, MD, a neurotology fellow at the Johns Hopkins University, Baltimore, and the first author of the U.S. study, which appeared Feb. 24 in JAMA Otolaryngology – Head and Neck Surgery.
Dr. Formeister and colleagues undertook the study in response to reports of hearing problems, including hearing loss and tinnitus, that occurred soon after COVID-19 vaccination.
They analyzed reports of sudden hearing loss, experienced within 21 days of vaccination, logged in VAERS. Anyone can report a potential event to the database, which does not require medical documentation in support of the adverse event. To minimize potential misdiagnoses, Dr. Formeister and colleagues reviewed only those reports that indicated that a doctor had diagnosed sudden hearing loss, leaving 555 cases (305 in women; mean age 54 years) between December 2020 and July 2021.
Dividing these reports by the total doses of vaccines administered in the United States during that period yielded an incidence rate of 0.6 cases of sudden hearing loss for every 100,000 people, Dr. Formeister and colleagues reported.
When the researchers divided all cases of hearing loss in the VAERS database (2,170) by the number of people who had received two doses of vaccine, the incidence rate increased to 28 per 100,000 people. For comparison, the authors reported, the incidence of sudden hearing loss within the United States population is between 11 and 77 per 100,000 people, depending on age.
“There was not an increase in cases of sudden [sensorineural] hearing loss associated with COVID-19 vaccination compared to previously published reports before the COVID-19 vaccination era,” study coauthor Elliott D. Kozin, MD, assistant professor of otolaryngology–head and neck surgery at Harvard Medical School, Boston, said in an interview.
Another reassuring sign: If hearing loss were linked to the vaccines, the researchers said, they would expect to see an increase in the number of complaints in lockstep with an increase in the number of doses administered. However, the opposite was true. “[T]he rate of reports per 100,000 doses decreased across the vaccination period, despite large concomitant increases in the absolute number of vaccine doses administered per week,” the researchers reported.
They also looked at case reports of 21 men and women who had experienced sudden hearing loss after having received COVID-19 vaccines, to see if they could discern any clinically relevant signs of people most likely to experience the adverse event. However, the group had a range of preexisting conditions and varying times after receiving a vaccine when their hearing loss occurred, leading Dr. Formeister’s team to conclude that they could find no clear markers of risk.
“When we examined patients across several institutions, there was no obvious pattern. The patient demographics and clinical findings were variable,” Dr. Kozin said. A provisional interpretation of this data, he added, is that no link exists between COVID-19 vaccination and predictable hearing deficits, although the analysis covered a small number of patients.
“Association does not necessarily imply a causal relationship,” said Michael Brenner, MD, FACS, associate professor of otolaryngology–head and neck surgery at the University of Michigan, Ann Arbor. Dr. Brenner, who was not involved in the study, said any hearing loss attributed to the COVID-19 vaccines could have had other causes besides the injections.
But a second study, also published in JAMA Otolaryngology – Head and Neck Surgery on Feb. 24, leaves open the possibility of a link. Researchers in Israel looked for increases in steroid prescriptions used to treat sudden hearing loss as vaccination with the Pfizer version of the shot became widespread in that country. Their conclusion: The vaccine might be associated with a slightly increased risk of sudden hearing loss, although if so, that risk is likely “very small” and the benefits of vaccination “outweigh its potential association” with the side effect.
Dr. Brenner agreed. “The evidence supports [the] clear public health benefit of COVID-19 vaccination, and the scale of those benefits dwarfs associations with hearing, which are of uncertain significance,” he said.
A version of this article first appeared on Medscape.com.
Anecdotal reports have linked the vaccines against COVID-19 to the sudden loss of hearing in some people. But a new study has found no evidence for such a connection with any of the three approved shots.
The analysis of data from the Centers for Disease Control and Prevention’s Vaccine Adverse Event Reporting System (VAERS) found that
“We’re not finding a signal,” said Eric J. Formeister, MD, a neurotology fellow at the Johns Hopkins University, Baltimore, and the first author of the U.S. study, which appeared Feb. 24 in JAMA Otolaryngology – Head and Neck Surgery.
Dr. Formeister and colleagues undertook the study in response to reports of hearing problems, including hearing loss and tinnitus, that occurred soon after COVID-19 vaccination.
They analyzed reports of sudden hearing loss, experienced within 21 days of vaccination, logged in VAERS. Anyone can report a potential event to the database, which does not require medical documentation in support of the adverse event. To minimize potential misdiagnoses, Dr. Formeister and colleagues reviewed only those reports that indicated that a doctor had diagnosed sudden hearing loss, leaving 555 cases (305 in women; mean age 54 years) between December 2020 and July 2021.
Dividing these reports by the total doses of vaccines administered in the United States during that period yielded an incidence rate of 0.6 cases of sudden hearing loss for every 100,000 people, Dr. Formeister and colleagues reported.
When the researchers divided all cases of hearing loss in the VAERS database (2,170) by the number of people who had received two doses of vaccine, the incidence rate increased to 28 per 100,000 people. For comparison, the authors reported, the incidence of sudden hearing loss within the United States population is between 11 and 77 per 100,000 people, depending on age.
“There was not an increase in cases of sudden [sensorineural] hearing loss associated with COVID-19 vaccination compared to previously published reports before the COVID-19 vaccination era,” study coauthor Elliott D. Kozin, MD, assistant professor of otolaryngology–head and neck surgery at Harvard Medical School, Boston, said in an interview.
Another reassuring sign: If hearing loss were linked to the vaccines, the researchers said, they would expect to see an increase in the number of complaints in lockstep with an increase in the number of doses administered. However, the opposite was true. “[T]he rate of reports per 100,000 doses decreased across the vaccination period, despite large concomitant increases in the absolute number of vaccine doses administered per week,” the researchers reported.
They also looked at case reports of 21 men and women who had experienced sudden hearing loss after having received COVID-19 vaccines, to see if they could discern any clinically relevant signs of people most likely to experience the adverse event. However, the group had a range of preexisting conditions and varying times after receiving a vaccine when their hearing loss occurred, leading Dr. Formeister’s team to conclude that they could find no clear markers of risk.
“When we examined patients across several institutions, there was no obvious pattern. The patient demographics and clinical findings were variable,” Dr. Kozin said. A provisional interpretation of this data, he added, is that no link exists between COVID-19 vaccination and predictable hearing deficits, although the analysis covered a small number of patients.
“Association does not necessarily imply a causal relationship,” said Michael Brenner, MD, FACS, associate professor of otolaryngology–head and neck surgery at the University of Michigan, Ann Arbor. Dr. Brenner, who was not involved in the study, said any hearing loss attributed to the COVID-19 vaccines could have had other causes besides the injections.
But a second study, also published in JAMA Otolaryngology – Head and Neck Surgery on Feb. 24, leaves open the possibility of a link. Researchers in Israel looked for increases in steroid prescriptions used to treat sudden hearing loss as vaccination with the Pfizer version of the shot became widespread in that country. Their conclusion: The vaccine might be associated with a slightly increased risk of sudden hearing loss, although if so, that risk is likely “very small” and the benefits of vaccination “outweigh its potential association” with the side effect.
Dr. Brenner agreed. “The evidence supports [the] clear public health benefit of COVID-19 vaccination, and the scale of those benefits dwarfs associations with hearing, which are of uncertain significance,” he said.
A version of this article first appeared on Medscape.com.
FROM JAMA OTOLARYNGOLOGY – HEAD AND NECK SURGERY
Why challenging patients can trigger resentment
I have a secret. It’s one I think many physicians and nurses share. Sometimes, when I’m stretched too thin — overbooked, hungry, tired, fielding yet another appeal to an insurance company in the middle of a clinic day —
As soon as this happens, I feel immediate guilt. These are the worst moments of my day. Why the heck would I resent my patients? They’re the entire reason I’m there. I wouldn’t be a physician without patients to care for. I became a physician, and completed subspecialty training, to help patients. People.
Recently, I started thinking more about this emotion of resentment. What exactly is it, and where does it come from? Is what I’m feeling actually resentment? Or is it something else?
Two books I’ve recently read have helped me explore the complicated emotion of resentment and how it might play a role in burnout for both physicians and nurses.
First, Brené Brown’s most recent book, Atlas of the Heart: Mapping Meaningful Connection and the Language of Human Experience, provides a roadmap for 87 of our human emotions. (That’s right — 87!)
One emotion of the 87 that she shares has been a particular struggle for her has been our good old friend, resentment.
In her book, Dr Brown shares that she initially considered resentment to belong to the anger family of emotion. As I read this, I agreed. When I feel resentful, I associate that with feeling angry.
But she then writes about her discovery that resentment, in fact, belongs to the envy family. She explains how this discovery shook her world. I had to close the book for a moment at this point.
Wait a minute, I thought. If resentment is in the envy family, why do we (physicians) often find ourselves resenting patients who take up our time? What are we envious of?
I took some time to think about how this might be true. Could it be that I’m envious they have the time I don’t have? I want to have all the time in the world to answer their questions, but the reality is I don’t.
Or maybe it’s because sometimes I feel the patient is expecting me to offer them something more than is available. A cure when there might be none.
But is this actually true? Or is this my unrealistic expectation of myself?
Here’s how Brené Brown defines resentment in her book: “Resentment is the feeling of frustration, judgment, anger, ‘better than,’ and/or hidden envy related to perceived unfairness or injustice. It’s an emotion that we often experience when we fail to set boundaries or ask for what we need, or when expectations let us down because they were based on things we can’t control, like what other people think, what they feel, or how they’re going to react.”
Wow, I thought, Healthcare checks all of these boxes.
- Perceived unfairness of work schedules? Check.
- Perceived injustice? Of course — we see that in our dealings with insurance company denials every day.
But those are both extrinsic. What about the intrinsic factors she’s calling us out on here?
- Do we, as physicians, fail to set boundaries?
- Do we fail to ask for what we need?
Hard yes and yes. (Do we even know, as physicians, what our own boundaries are?)
And the last one:
- Do our expectations of how our clinic day will go let us down every day because they’re based on things we can’t control?
My brain had to repeat the critical parts of that: Expectations let us down when they’re based on things we can’t control.
But wait, my brain argued back; I’m the physician, I thought I was supposed to get to control things.
Next, the revelation: Could it be that a key to experiencing less resentment is accepting how much control we don’t have in a typical day?
And a corollary: How much does resentment factor into burnout? (To read more on my personal journey with burnout, see this piece).
It so happens that around this same time, I was reading another excellent book, Changing How We Think About Difficult Patients: A Guide for Physicians and Healthcare Professionals, by Joan Naidorf, DO.
Dr Naidorf is an emergency medicine physician of 30 years who wrote the book to “provid[e] insight and tools to manage our negative thoughts about difficult patients” and help “beleaguered colleagues…return to their benevolent guiding principles and find more enjoyment in their vitally important careers.”
As I read Dr Naidorf’s book, I thus did so with the mindset of wanting to further understand for myself where this specific emotion of resentment toward our “difficult” patients could come from and how to best understand it in order to get past it.
Dr. Naidorf writes, “Challenging patients will never stop appearing… You cannot change them or control them—the only person you can control is you.”
I wondered how much the resentment we might involuntarily feel at being asked to see a “difficult” patient has nothing to do with the patient but everything to do with it making us feel not in control of the situation.
Dr. Naidorf also writes, “Negative thoughts about challenging patients can cause, in otherwise capable clinicians, a sense of inadequacy and incompetence.”
Do we perhaps resent our challenging patients because of the negative thoughts they sometimes trigger in us? If so, how does this relate to envy, as Dr. Brown asserts resentment is tied to? Is it triggering us to feel inadequate?
“[Difficult patients] often make us question ourselves,” Dr. Naidorf writes, “and we need to feel comfortable with the answers.”
Again, the discrepancy between expectations and reality creates the negative emotion.
Or, as Dr. Naidorf writes, “What if you could stop judging others so harshly and accept them exactly as they are?”
Hmmm, I thought, then the cessation of harsh judgment and implementation of acceptance would have to apply to us too. The elusive concept of self-compassion.
Maybe the resentment/envy comes from us not allowing ourselves to behave in this way because to do so would allow too much vulnerability. Something most of us were conditioned to avoid to survive medical training.
Dr. Brown also writes about an “aha” moment she had in her struggle to understand resentment. “I’m not mad because you’re resting. I’m mad because I’m so bone tired and I want to rest. But, unlike you, I’m going to pretend that I don’t need to.”
I felt all too seen in that passage. Could it be my old nemesis, perfectionism, creeping its way back in? Is resentment the ugly stepsister to perfectionism?
Perhaps challenging patients can engender resentment because they make us feel like we’re not living up to our own unrealistic expectations. And in that case, we need to change our unrealistic expectations for ourselves.
Dr Naidorf’s book explores much more on the complex matter of what makes a “difficult” patient, but I chose to focus here only on the resentment piece as a tie-in to Dr. Brown’s book. I highly recommend both books for further reading to help physicians and nurses navigate the complex emotions our jobs can trigger.
Most importantly, recognizing that we have these transient negative emotions does not make us bad people or healthcare professionals. It only makes us human.
Dr. Lycette is medical director, Providence Oncology and Hematology Care Clinic, Seaside, Ore. She has disclosed having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
I have a secret. It’s one I think many physicians and nurses share. Sometimes, when I’m stretched too thin — overbooked, hungry, tired, fielding yet another appeal to an insurance company in the middle of a clinic day —
As soon as this happens, I feel immediate guilt. These are the worst moments of my day. Why the heck would I resent my patients? They’re the entire reason I’m there. I wouldn’t be a physician without patients to care for. I became a physician, and completed subspecialty training, to help patients. People.
Recently, I started thinking more about this emotion of resentment. What exactly is it, and where does it come from? Is what I’m feeling actually resentment? Or is it something else?
Two books I’ve recently read have helped me explore the complicated emotion of resentment and how it might play a role in burnout for both physicians and nurses.
First, Brené Brown’s most recent book, Atlas of the Heart: Mapping Meaningful Connection and the Language of Human Experience, provides a roadmap for 87 of our human emotions. (That’s right — 87!)
One emotion of the 87 that she shares has been a particular struggle for her has been our good old friend, resentment.
In her book, Dr Brown shares that she initially considered resentment to belong to the anger family of emotion. As I read this, I agreed. When I feel resentful, I associate that with feeling angry.
But she then writes about her discovery that resentment, in fact, belongs to the envy family. She explains how this discovery shook her world. I had to close the book for a moment at this point.
Wait a minute, I thought. If resentment is in the envy family, why do we (physicians) often find ourselves resenting patients who take up our time? What are we envious of?
I took some time to think about how this might be true. Could it be that I’m envious they have the time I don’t have? I want to have all the time in the world to answer their questions, but the reality is I don’t.
Or maybe it’s because sometimes I feel the patient is expecting me to offer them something more than is available. A cure when there might be none.
But is this actually true? Or is this my unrealistic expectation of myself?
Here’s how Brené Brown defines resentment in her book: “Resentment is the feeling of frustration, judgment, anger, ‘better than,’ and/or hidden envy related to perceived unfairness or injustice. It’s an emotion that we often experience when we fail to set boundaries or ask for what we need, or when expectations let us down because they were based on things we can’t control, like what other people think, what they feel, or how they’re going to react.”
Wow, I thought, Healthcare checks all of these boxes.
- Perceived unfairness of work schedules? Check.
- Perceived injustice? Of course — we see that in our dealings with insurance company denials every day.
But those are both extrinsic. What about the intrinsic factors she’s calling us out on here?
- Do we, as physicians, fail to set boundaries?
- Do we fail to ask for what we need?
Hard yes and yes. (Do we even know, as physicians, what our own boundaries are?)
And the last one:
- Do our expectations of how our clinic day will go let us down every day because they’re based on things we can’t control?
My brain had to repeat the critical parts of that: Expectations let us down when they’re based on things we can’t control.
But wait, my brain argued back; I’m the physician, I thought I was supposed to get to control things.
Next, the revelation: Could it be that a key to experiencing less resentment is accepting how much control we don’t have in a typical day?
And a corollary: How much does resentment factor into burnout? (To read more on my personal journey with burnout, see this piece).
It so happens that around this same time, I was reading another excellent book, Changing How We Think About Difficult Patients: A Guide for Physicians and Healthcare Professionals, by Joan Naidorf, DO.
Dr Naidorf is an emergency medicine physician of 30 years who wrote the book to “provid[e] insight and tools to manage our negative thoughts about difficult patients” and help “beleaguered colleagues…return to their benevolent guiding principles and find more enjoyment in their vitally important careers.”
As I read Dr Naidorf’s book, I thus did so with the mindset of wanting to further understand for myself where this specific emotion of resentment toward our “difficult” patients could come from and how to best understand it in order to get past it.
Dr. Naidorf writes, “Challenging patients will never stop appearing… You cannot change them or control them—the only person you can control is you.”
I wondered how much the resentment we might involuntarily feel at being asked to see a “difficult” patient has nothing to do with the patient but everything to do with it making us feel not in control of the situation.
Dr. Naidorf also writes, “Negative thoughts about challenging patients can cause, in otherwise capable clinicians, a sense of inadequacy and incompetence.”
Do we perhaps resent our challenging patients because of the negative thoughts they sometimes trigger in us? If so, how does this relate to envy, as Dr. Brown asserts resentment is tied to? Is it triggering us to feel inadequate?
“[Difficult patients] often make us question ourselves,” Dr. Naidorf writes, “and we need to feel comfortable with the answers.”
Again, the discrepancy between expectations and reality creates the negative emotion.
Or, as Dr. Naidorf writes, “What if you could stop judging others so harshly and accept them exactly as they are?”
Hmmm, I thought, then the cessation of harsh judgment and implementation of acceptance would have to apply to us too. The elusive concept of self-compassion.
Maybe the resentment/envy comes from us not allowing ourselves to behave in this way because to do so would allow too much vulnerability. Something most of us were conditioned to avoid to survive medical training.
Dr. Brown also writes about an “aha” moment she had in her struggle to understand resentment. “I’m not mad because you’re resting. I’m mad because I’m so bone tired and I want to rest. But, unlike you, I’m going to pretend that I don’t need to.”
I felt all too seen in that passage. Could it be my old nemesis, perfectionism, creeping its way back in? Is resentment the ugly stepsister to perfectionism?
Perhaps challenging patients can engender resentment because they make us feel like we’re not living up to our own unrealistic expectations. And in that case, we need to change our unrealistic expectations for ourselves.
Dr Naidorf’s book explores much more on the complex matter of what makes a “difficult” patient, but I chose to focus here only on the resentment piece as a tie-in to Dr. Brown’s book. I highly recommend both books for further reading to help physicians and nurses navigate the complex emotions our jobs can trigger.
Most importantly, recognizing that we have these transient negative emotions does not make us bad people or healthcare professionals. It only makes us human.
Dr. Lycette is medical director, Providence Oncology and Hematology Care Clinic, Seaside, Ore. She has disclosed having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
I have a secret. It’s one I think many physicians and nurses share. Sometimes, when I’m stretched too thin — overbooked, hungry, tired, fielding yet another appeal to an insurance company in the middle of a clinic day —
As soon as this happens, I feel immediate guilt. These are the worst moments of my day. Why the heck would I resent my patients? They’re the entire reason I’m there. I wouldn’t be a physician without patients to care for. I became a physician, and completed subspecialty training, to help patients. People.
Recently, I started thinking more about this emotion of resentment. What exactly is it, and where does it come from? Is what I’m feeling actually resentment? Or is it something else?
Two books I’ve recently read have helped me explore the complicated emotion of resentment and how it might play a role in burnout for both physicians and nurses.
First, Brené Brown’s most recent book, Atlas of the Heart: Mapping Meaningful Connection and the Language of Human Experience, provides a roadmap for 87 of our human emotions. (That’s right — 87!)
One emotion of the 87 that she shares has been a particular struggle for her has been our good old friend, resentment.
In her book, Dr Brown shares that she initially considered resentment to belong to the anger family of emotion. As I read this, I agreed. When I feel resentful, I associate that with feeling angry.
But she then writes about her discovery that resentment, in fact, belongs to the envy family. She explains how this discovery shook her world. I had to close the book for a moment at this point.
Wait a minute, I thought. If resentment is in the envy family, why do we (physicians) often find ourselves resenting patients who take up our time? What are we envious of?
I took some time to think about how this might be true. Could it be that I’m envious they have the time I don’t have? I want to have all the time in the world to answer their questions, but the reality is I don’t.
Or maybe it’s because sometimes I feel the patient is expecting me to offer them something more than is available. A cure when there might be none.
But is this actually true? Or is this my unrealistic expectation of myself?
Here’s how Brené Brown defines resentment in her book: “Resentment is the feeling of frustration, judgment, anger, ‘better than,’ and/or hidden envy related to perceived unfairness or injustice. It’s an emotion that we often experience when we fail to set boundaries or ask for what we need, or when expectations let us down because they were based on things we can’t control, like what other people think, what they feel, or how they’re going to react.”
Wow, I thought, Healthcare checks all of these boxes.
- Perceived unfairness of work schedules? Check.
- Perceived injustice? Of course — we see that in our dealings with insurance company denials every day.
But those are both extrinsic. What about the intrinsic factors she’s calling us out on here?
- Do we, as physicians, fail to set boundaries?
- Do we fail to ask for what we need?
Hard yes and yes. (Do we even know, as physicians, what our own boundaries are?)
And the last one:
- Do our expectations of how our clinic day will go let us down every day because they’re based on things we can’t control?
My brain had to repeat the critical parts of that: Expectations let us down when they’re based on things we can’t control.
But wait, my brain argued back; I’m the physician, I thought I was supposed to get to control things.
Next, the revelation: Could it be that a key to experiencing less resentment is accepting how much control we don’t have in a typical day?
And a corollary: How much does resentment factor into burnout? (To read more on my personal journey with burnout, see this piece).
It so happens that around this same time, I was reading another excellent book, Changing How We Think About Difficult Patients: A Guide for Physicians and Healthcare Professionals, by Joan Naidorf, DO.
Dr Naidorf is an emergency medicine physician of 30 years who wrote the book to “provid[e] insight and tools to manage our negative thoughts about difficult patients” and help “beleaguered colleagues…return to their benevolent guiding principles and find more enjoyment in their vitally important careers.”
As I read Dr Naidorf’s book, I thus did so with the mindset of wanting to further understand for myself where this specific emotion of resentment toward our “difficult” patients could come from and how to best understand it in order to get past it.
Dr. Naidorf writes, “Challenging patients will never stop appearing… You cannot change them or control them—the only person you can control is you.”
I wondered how much the resentment we might involuntarily feel at being asked to see a “difficult” patient has nothing to do with the patient but everything to do with it making us feel not in control of the situation.
Dr. Naidorf also writes, “Negative thoughts about challenging patients can cause, in otherwise capable clinicians, a sense of inadequacy and incompetence.”
Do we perhaps resent our challenging patients because of the negative thoughts they sometimes trigger in us? If so, how does this relate to envy, as Dr. Brown asserts resentment is tied to? Is it triggering us to feel inadequate?
“[Difficult patients] often make us question ourselves,” Dr. Naidorf writes, “and we need to feel comfortable with the answers.”
Again, the discrepancy between expectations and reality creates the negative emotion.
Or, as Dr. Naidorf writes, “What if you could stop judging others so harshly and accept them exactly as they are?”
Hmmm, I thought, then the cessation of harsh judgment and implementation of acceptance would have to apply to us too. The elusive concept of self-compassion.
Maybe the resentment/envy comes from us not allowing ourselves to behave in this way because to do so would allow too much vulnerability. Something most of us were conditioned to avoid to survive medical training.
Dr. Brown also writes about an “aha” moment she had in her struggle to understand resentment. “I’m not mad because you’re resting. I’m mad because I’m so bone tired and I want to rest. But, unlike you, I’m going to pretend that I don’t need to.”
I felt all too seen in that passage. Could it be my old nemesis, perfectionism, creeping its way back in? Is resentment the ugly stepsister to perfectionism?
Perhaps challenging patients can engender resentment because they make us feel like we’re not living up to our own unrealistic expectations. And in that case, we need to change our unrealistic expectations for ourselves.
Dr Naidorf’s book explores much more on the complex matter of what makes a “difficult” patient, but I chose to focus here only on the resentment piece as a tie-in to Dr. Brown’s book. I highly recommend both books for further reading to help physicians and nurses navigate the complex emotions our jobs can trigger.
Most importantly, recognizing that we have these transient negative emotions does not make us bad people or healthcare professionals. It only makes us human.
Dr. Lycette is medical director, Providence Oncology and Hematology Care Clinic, Seaside, Ore. She has disclosed having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Ukrainian physicians ‘ready to die for their freedom’
Nasogastric tubes. Foley catheter kits. Hydrogel anti-burn bandages and transfusion bags. Heparin, atropine, tramadol.
These items are just a few of some two dozen critical medical supplies that physicians in Ukraine desperately need, according to Leo Wolansky, MD, a Ukrainian-American radiologist and president of the Ukrainian Medical Association of North America (UMANA).
Dr. Wolansky founded a teaching program with an organization called Friends of Radiology in Ukraine in 1996 and has been running courses for specialists there ever since. He last visited the country in 2019, before the COVID-19 pandemic, but has remained in contact with his medical colleagues by phone and email. Over the weekend of Feb. 26-27, UMANA held a fundraiser for Ukraine, raising more than $17,000.
Question: Where is your family from, and do you have relatives in the country now?
Dr. Wolansky: My family is from two different parts of Ukraine. My mother was from central Ukraine. Her father, Ivan Sharyj, was part of the students’ militia that fought at the famous battle of Kruty in 1918. Four hundred Ukrainian militia fought against 5,000 professional Russian soldiers and were massacred. He later wrote the first eye-witness account. Afterwards, he had the opportunity to flee Ukraine but chose to stay under a pseudonym. Eventually, during Stalin’s purges [1929-1933], the regime found him, arrested him, tortured him, and executed him. My mother was seven when she saw her father arrested, never to return home. My father was from Western Ukraine, which did not have a long history of Russian occupation. His mother’s family was very patriotic; her first cousin, Stepan Vytvytskyi, eventually became the president of Ukraine in exile from 1955-1964.
I have second and more distant cousins in Kyiv. My wife has first cousins in Western Ukraine. They and my doctor colleagues are suffering greatly but are ready to die for their freedom.
Question: The Russian invasion of Ukraine has put tremendous stress on the Ukrainian people, including the country’s medical professionals. How do doctors in these kinds of situations handle casualties they can’t prevent? How do they work around that sense that everything is out of their control?
Dr. Wolansky: A lot of infrastructural things are being disrupted; there are limitations that you wouldn’t normally encounter. Ukraine has been developing a lot of sophisticated medical technology, but it still has room to grow. Under these circumstances, when there are bombs going off and transportation is being disrupted, it creates very new and significant obstacles to surmount. It still has not risen to massive casualties, and we can just pray that it does not, but in times of war, a very different kind of medicine is practiced.
But remember, Ukraine has been at war since 2014, when Russia took Crimea and invaded the Eastern provinces. The doctors there are not unfamiliar with war injuries. At our conferences in Ukraine, I have seen radiological presentations of injuries sustained in war – gunshots, fractures, and amputations – as well as other kinds of traumatic injuries. You’re going for a kind of more emergent treatment: to transfuse, to maintain peoples’ blood pressure, put bandages on, sterilize and sanitize wounds to prevent infections. I imagine there will be many field hospitals set up between now and the next few weeks to deal with the acute injuries.
Question: Ukraine has struggled with high rates of HIV and multidrug-resistant tuberculosis, as well as a lack of resources for treating patients with mental illness. Meanwhile, the country has had more that 5 million cases of COVID-19 and an estimated 112,000 deaths from the disease. Are you concerned about an exacerbation of infection rates, including of COVID, particularly among refugees and those who become homeless?
Dr. Wolansky: Because COVID ran pretty rampant in Ukraine, I think that – at a high cost – there is a level of natural immunity in the population. And the weather is going to be getting warmer soon, and respiratory viruses are cyclic in nature, so I don’t know if that’s going to be a big complicating factor. However, people get sick all the time, and the prognosis for them is going to be much worse than it otherwise might be. If you have a heart attack, your chances were way better when the roads were clear and people weren’t shooting at you.
Right now, it’s very regional where the infrastructure is being destroyed. The West, where I used to go, is in much better shape than the East because it has not been the focus of Russian attacks. But Kyiv could turn into a very big humanitarian crisis very quickly if there’s no electricity, no water. All sorts of medical conditions could be greatly exacerbated, and some new health crises could arise from water contamination, bombs causing buildings to collapse, and other problems. Whatever the illness is, it’s going to be harder to take care of it.
Questions: Doctors Without Borders announced that it was suspending its operations in Ukraine because of the invasion – missions that included HIV care in Severodonetsk, tuberculosis care in Zhytomyr, and improving health care access in Donetsk in eastern Ukraine, according to the aid group. What do doctors in Ukraine need most acutely now, other than peace?
Dr. Wolansky: Obviously, money is valuable, and military protection, which would prevent additional damage to their infrastructure. One thing that bears mentioning. There’s been a fair amount of coverage of this, but I’ve witnessed it first-hand: The Ukrainian people are fiercely patriotic, and there’s really no way their spirit can be conquered. The USSR invaded Afghanistan, and after years of thinking they were in command, they left because they could no longer take the guerilla warfare and the constant sniper attacks. Ukraine’s population is many times larger than Afghanistan’s; there’s no way they can be subdued. And remember, the Ukrainian people have been free for 30 years – generations of young people have known no other way of life. They are not going to give that up.
A version of this article first appeared on Medscape.com.
Nasogastric tubes. Foley catheter kits. Hydrogel anti-burn bandages and transfusion bags. Heparin, atropine, tramadol.
These items are just a few of some two dozen critical medical supplies that physicians in Ukraine desperately need, according to Leo Wolansky, MD, a Ukrainian-American radiologist and president of the Ukrainian Medical Association of North America (UMANA).
Dr. Wolansky founded a teaching program with an organization called Friends of Radiology in Ukraine in 1996 and has been running courses for specialists there ever since. He last visited the country in 2019, before the COVID-19 pandemic, but has remained in contact with his medical colleagues by phone and email. Over the weekend of Feb. 26-27, UMANA held a fundraiser for Ukraine, raising more than $17,000.
Question: Where is your family from, and do you have relatives in the country now?
Dr. Wolansky: My family is from two different parts of Ukraine. My mother was from central Ukraine. Her father, Ivan Sharyj, was part of the students’ militia that fought at the famous battle of Kruty in 1918. Four hundred Ukrainian militia fought against 5,000 professional Russian soldiers and were massacred. He later wrote the first eye-witness account. Afterwards, he had the opportunity to flee Ukraine but chose to stay under a pseudonym. Eventually, during Stalin’s purges [1929-1933], the regime found him, arrested him, tortured him, and executed him. My mother was seven when she saw her father arrested, never to return home. My father was from Western Ukraine, which did not have a long history of Russian occupation. His mother’s family was very patriotic; her first cousin, Stepan Vytvytskyi, eventually became the president of Ukraine in exile from 1955-1964.
I have second and more distant cousins in Kyiv. My wife has first cousins in Western Ukraine. They and my doctor colleagues are suffering greatly but are ready to die for their freedom.
Question: The Russian invasion of Ukraine has put tremendous stress on the Ukrainian people, including the country’s medical professionals. How do doctors in these kinds of situations handle casualties they can’t prevent? How do they work around that sense that everything is out of their control?
Dr. Wolansky: A lot of infrastructural things are being disrupted; there are limitations that you wouldn’t normally encounter. Ukraine has been developing a lot of sophisticated medical technology, but it still has room to grow. Under these circumstances, when there are bombs going off and transportation is being disrupted, it creates very new and significant obstacles to surmount. It still has not risen to massive casualties, and we can just pray that it does not, but in times of war, a very different kind of medicine is practiced.
But remember, Ukraine has been at war since 2014, when Russia took Crimea and invaded the Eastern provinces. The doctors there are not unfamiliar with war injuries. At our conferences in Ukraine, I have seen radiological presentations of injuries sustained in war – gunshots, fractures, and amputations – as well as other kinds of traumatic injuries. You’re going for a kind of more emergent treatment: to transfuse, to maintain peoples’ blood pressure, put bandages on, sterilize and sanitize wounds to prevent infections. I imagine there will be many field hospitals set up between now and the next few weeks to deal with the acute injuries.
Question: Ukraine has struggled with high rates of HIV and multidrug-resistant tuberculosis, as well as a lack of resources for treating patients with mental illness. Meanwhile, the country has had more that 5 million cases of COVID-19 and an estimated 112,000 deaths from the disease. Are you concerned about an exacerbation of infection rates, including of COVID, particularly among refugees and those who become homeless?
Dr. Wolansky: Because COVID ran pretty rampant in Ukraine, I think that – at a high cost – there is a level of natural immunity in the population. And the weather is going to be getting warmer soon, and respiratory viruses are cyclic in nature, so I don’t know if that’s going to be a big complicating factor. However, people get sick all the time, and the prognosis for them is going to be much worse than it otherwise might be. If you have a heart attack, your chances were way better when the roads were clear and people weren’t shooting at you.
Right now, it’s very regional where the infrastructure is being destroyed. The West, where I used to go, is in much better shape than the East because it has not been the focus of Russian attacks. But Kyiv could turn into a very big humanitarian crisis very quickly if there’s no electricity, no water. All sorts of medical conditions could be greatly exacerbated, and some new health crises could arise from water contamination, bombs causing buildings to collapse, and other problems. Whatever the illness is, it’s going to be harder to take care of it.
Questions: Doctors Without Borders announced that it was suspending its operations in Ukraine because of the invasion – missions that included HIV care in Severodonetsk, tuberculosis care in Zhytomyr, and improving health care access in Donetsk in eastern Ukraine, according to the aid group. What do doctors in Ukraine need most acutely now, other than peace?
Dr. Wolansky: Obviously, money is valuable, and military protection, which would prevent additional damage to their infrastructure. One thing that bears mentioning. There’s been a fair amount of coverage of this, but I’ve witnessed it first-hand: The Ukrainian people are fiercely patriotic, and there’s really no way their spirit can be conquered. The USSR invaded Afghanistan, and after years of thinking they were in command, they left because they could no longer take the guerilla warfare and the constant sniper attacks. Ukraine’s population is many times larger than Afghanistan’s; there’s no way they can be subdued. And remember, the Ukrainian people have been free for 30 years – generations of young people have known no other way of life. They are not going to give that up.
A version of this article first appeared on Medscape.com.
Nasogastric tubes. Foley catheter kits. Hydrogel anti-burn bandages and transfusion bags. Heparin, atropine, tramadol.
These items are just a few of some two dozen critical medical supplies that physicians in Ukraine desperately need, according to Leo Wolansky, MD, a Ukrainian-American radiologist and president of the Ukrainian Medical Association of North America (UMANA).
Dr. Wolansky founded a teaching program with an organization called Friends of Radiology in Ukraine in 1996 and has been running courses for specialists there ever since. He last visited the country in 2019, before the COVID-19 pandemic, but has remained in contact with his medical colleagues by phone and email. Over the weekend of Feb. 26-27, UMANA held a fundraiser for Ukraine, raising more than $17,000.
Question: Where is your family from, and do you have relatives in the country now?
Dr. Wolansky: My family is from two different parts of Ukraine. My mother was from central Ukraine. Her father, Ivan Sharyj, was part of the students’ militia that fought at the famous battle of Kruty in 1918. Four hundred Ukrainian militia fought against 5,000 professional Russian soldiers and were massacred. He later wrote the first eye-witness account. Afterwards, he had the opportunity to flee Ukraine but chose to stay under a pseudonym. Eventually, during Stalin’s purges [1929-1933], the regime found him, arrested him, tortured him, and executed him. My mother was seven when she saw her father arrested, never to return home. My father was from Western Ukraine, which did not have a long history of Russian occupation. His mother’s family was very patriotic; her first cousin, Stepan Vytvytskyi, eventually became the president of Ukraine in exile from 1955-1964.
I have second and more distant cousins in Kyiv. My wife has first cousins in Western Ukraine. They and my doctor colleagues are suffering greatly but are ready to die for their freedom.
Question: The Russian invasion of Ukraine has put tremendous stress on the Ukrainian people, including the country’s medical professionals. How do doctors in these kinds of situations handle casualties they can’t prevent? How do they work around that sense that everything is out of their control?
Dr. Wolansky: A lot of infrastructural things are being disrupted; there are limitations that you wouldn’t normally encounter. Ukraine has been developing a lot of sophisticated medical technology, but it still has room to grow. Under these circumstances, when there are bombs going off and transportation is being disrupted, it creates very new and significant obstacles to surmount. It still has not risen to massive casualties, and we can just pray that it does not, but in times of war, a very different kind of medicine is practiced.
But remember, Ukraine has been at war since 2014, when Russia took Crimea and invaded the Eastern provinces. The doctors there are not unfamiliar with war injuries. At our conferences in Ukraine, I have seen radiological presentations of injuries sustained in war – gunshots, fractures, and amputations – as well as other kinds of traumatic injuries. You’re going for a kind of more emergent treatment: to transfuse, to maintain peoples’ blood pressure, put bandages on, sterilize and sanitize wounds to prevent infections. I imagine there will be many field hospitals set up between now and the next few weeks to deal with the acute injuries.
Question: Ukraine has struggled with high rates of HIV and multidrug-resistant tuberculosis, as well as a lack of resources for treating patients with mental illness. Meanwhile, the country has had more that 5 million cases of COVID-19 and an estimated 112,000 deaths from the disease. Are you concerned about an exacerbation of infection rates, including of COVID, particularly among refugees and those who become homeless?
Dr. Wolansky: Because COVID ran pretty rampant in Ukraine, I think that – at a high cost – there is a level of natural immunity in the population. And the weather is going to be getting warmer soon, and respiratory viruses are cyclic in nature, so I don’t know if that’s going to be a big complicating factor. However, people get sick all the time, and the prognosis for them is going to be much worse than it otherwise might be. If you have a heart attack, your chances were way better when the roads were clear and people weren’t shooting at you.
Right now, it’s very regional where the infrastructure is being destroyed. The West, where I used to go, is in much better shape than the East because it has not been the focus of Russian attacks. But Kyiv could turn into a very big humanitarian crisis very quickly if there’s no electricity, no water. All sorts of medical conditions could be greatly exacerbated, and some new health crises could arise from water contamination, bombs causing buildings to collapse, and other problems. Whatever the illness is, it’s going to be harder to take care of it.
Questions: Doctors Without Borders announced that it was suspending its operations in Ukraine because of the invasion – missions that included HIV care in Severodonetsk, tuberculosis care in Zhytomyr, and improving health care access in Donetsk in eastern Ukraine, according to the aid group. What do doctors in Ukraine need most acutely now, other than peace?
Dr. Wolansky: Obviously, money is valuable, and military protection, which would prevent additional damage to their infrastructure. One thing that bears mentioning. There’s been a fair amount of coverage of this, but I’ve witnessed it first-hand: The Ukrainian people are fiercely patriotic, and there’s really no way their spirit can be conquered. The USSR invaded Afghanistan, and after years of thinking they were in command, they left because they could no longer take the guerilla warfare and the constant sniper attacks. Ukraine’s population is many times larger than Afghanistan’s; there’s no way they can be subdued. And remember, the Ukrainian people have been free for 30 years – generations of young people have known no other way of life. They are not going to give that up.
A version of this article first appeared on Medscape.com.
Older age for menopause raises risk for lung cancer
This study was published on Medrxiv.org as a preprint and has not yet been peer reviewed.
Key takeaways
- in analyses of more than 100,000 women that used Mendelian randomization (MR) as a tool to reduce residual confounding.
- The MR analyses showed no significant association between ANM and breast cancer, endometrial cancer, ovarian cancer, coronary heart disease, ischemic stroke, and Alzheimer’s disease.
- The clear lack of a causal effect of ANM on the outcomes of coronary heart disease and ischemic stroke in the MR analyses despite a strong inverse association seen in the observational data of this study (without MR) suggests residual confounding plays a substantial role in driving the observed outcomes.
Why this matters
- The authors said that, to their knowledge, this is the first study that has shown a causal association between older ANM and higher risk of postmenopausal lung cancer.
- This finding was directionally opposite to the significant protective effect of increased ANM documented in an observational analysis of roughly the same data as well as prior reports that did not use MR. This “notable inconsistency” suggests very substantial residual confounding without MR that could be driven by factors such as smoking, diet, and exercise.
- If these results are replicated in additional datasets, it would highlight a need for randomized, controlled trials of antiestrogen therapies in postmenopausal women for the prevention or treatment of lung cancer.
Study design
- The study included data from 106,853 postmenopausal women enrolled in the Women’s Health Initiative (WHI) and 95,464 women who were 37-73 years old included in the UK Biobank (UKB). Analyses for each outcome also included data from smaller numbers of women obtained from several additional datasets.
- The MR analysis used up to 55 single-nucleotide polymorphisms previously discovered through a genome-wide association study of about 70,000 women of European ancestry and independent of all datasets analyzed in the current study. The authors included all single-nucleotide polymorphisms with a consistent direction of effect on ANM.
- The MR analysis for lung cancer included 113,371 women from the two primary datasets and an additional 3012 women from six additional datasets.
- The MR analysis for bone fracture involved 113,239 women from the WHI and UKB only. The MR analysis for osteoporosis involved 137,080 women from the WHI, UKB, and one additional external dataset.
Key results
- Results from a meta-analysis of the MR results using data from the WHI, UKB, and the additional datasets showed ANM was causally associated with an increased risk of lung cancer by an odds ratio of 1.35 for each 5-year increase in ANM. In contrast, the adjusted observational analysis of data just from the WHI and UKB showed a significant 11% relative risk reduction in the incidence of lung cancer for each 5-year increase in ANM.
- The MR results also showed causally protective effects for fracture, with a 24% relative risk reduction, and for osteoporosis, with a 19% relative risk reduction for each 5-year increase in ANM.
- The MR analyses showed no significant association between AMN and outcome for breast cancer, endometrial cancer, ovarian cancer, coronary heart disease, ischemic stroke, and Alzheimer’s disease.
Limitations
The main limitation of the MR study was the potential for inadequate power for assessing some outcomes despite the large overall size of the study cohort. Lack of adequate power may be responsible for some of the nonsignificant associations seen in the study, such as for breast and endometrial cancers, where substantial prior evidence has implicated increased risk through the effects of prolonged exposure to endogenous or exogenous estrogens.
The healthy cohort effect in the UKB is a known weakness of this dataset that may have limited the number of cases and generalizability of findings.
Osteoporosis and Alzheimer’s disease were self-reported.
The study only included participants of European ancestry because most subjects in most of the cohorts examined were White women and the applied MR instruments were found by genome-wide association studies run predominantly in White women. The authors said the causal effects of ANM need study in more diverse populations.
Disclosures
- The study received no commercial funding.
- None of the authors had disclosures.
This is a summary of a preprint research study, “Genetic evidence for causal relationships between age at natural menopause and the risk of aging-associated adverse health outcomes,” written by authors primarily based at Stanford University School of Medicine i
A version of this article first appeared on Medscape.com.
This study was published on Medrxiv.org as a preprint and has not yet been peer reviewed.
Key takeaways
- in analyses of more than 100,000 women that used Mendelian randomization (MR) as a tool to reduce residual confounding.
- The MR analyses showed no significant association between ANM and breast cancer, endometrial cancer, ovarian cancer, coronary heart disease, ischemic stroke, and Alzheimer’s disease.
- The clear lack of a causal effect of ANM on the outcomes of coronary heart disease and ischemic stroke in the MR analyses despite a strong inverse association seen in the observational data of this study (without MR) suggests residual confounding plays a substantial role in driving the observed outcomes.
Why this matters
- The authors said that, to their knowledge, this is the first study that has shown a causal association between older ANM and higher risk of postmenopausal lung cancer.
- This finding was directionally opposite to the significant protective effect of increased ANM documented in an observational analysis of roughly the same data as well as prior reports that did not use MR. This “notable inconsistency” suggests very substantial residual confounding without MR that could be driven by factors such as smoking, diet, and exercise.
- If these results are replicated in additional datasets, it would highlight a need for randomized, controlled trials of antiestrogen therapies in postmenopausal women for the prevention or treatment of lung cancer.
Study design
- The study included data from 106,853 postmenopausal women enrolled in the Women’s Health Initiative (WHI) and 95,464 women who were 37-73 years old included in the UK Biobank (UKB). Analyses for each outcome also included data from smaller numbers of women obtained from several additional datasets.
- The MR analysis used up to 55 single-nucleotide polymorphisms previously discovered through a genome-wide association study of about 70,000 women of European ancestry and independent of all datasets analyzed in the current study. The authors included all single-nucleotide polymorphisms with a consistent direction of effect on ANM.
- The MR analysis for lung cancer included 113,371 women from the two primary datasets and an additional 3012 women from six additional datasets.
- The MR analysis for bone fracture involved 113,239 women from the WHI and UKB only. The MR analysis for osteoporosis involved 137,080 women from the WHI, UKB, and one additional external dataset.
Key results
- Results from a meta-analysis of the MR results using data from the WHI, UKB, and the additional datasets showed ANM was causally associated with an increased risk of lung cancer by an odds ratio of 1.35 for each 5-year increase in ANM. In contrast, the adjusted observational analysis of data just from the WHI and UKB showed a significant 11% relative risk reduction in the incidence of lung cancer for each 5-year increase in ANM.
- The MR results also showed causally protective effects for fracture, with a 24% relative risk reduction, and for osteoporosis, with a 19% relative risk reduction for each 5-year increase in ANM.
- The MR analyses showed no significant association between AMN and outcome for breast cancer, endometrial cancer, ovarian cancer, coronary heart disease, ischemic stroke, and Alzheimer’s disease.
Limitations
The main limitation of the MR study was the potential for inadequate power for assessing some outcomes despite the large overall size of the study cohort. Lack of adequate power may be responsible for some of the nonsignificant associations seen in the study, such as for breast and endometrial cancers, where substantial prior evidence has implicated increased risk through the effects of prolonged exposure to endogenous or exogenous estrogens.
The healthy cohort effect in the UKB is a known weakness of this dataset that may have limited the number of cases and generalizability of findings.
Osteoporosis and Alzheimer’s disease were self-reported.
The study only included participants of European ancestry because most subjects in most of the cohorts examined were White women and the applied MR instruments were found by genome-wide association studies run predominantly in White women. The authors said the causal effects of ANM need study in more diverse populations.
Disclosures
- The study received no commercial funding.
- None of the authors had disclosures.
This is a summary of a preprint research study, “Genetic evidence for causal relationships between age at natural menopause and the risk of aging-associated adverse health outcomes,” written by authors primarily based at Stanford University School of Medicine i
A version of this article first appeared on Medscape.com.
This study was published on Medrxiv.org as a preprint and has not yet been peer reviewed.
Key takeaways
- in analyses of more than 100,000 women that used Mendelian randomization (MR) as a tool to reduce residual confounding.
- The MR analyses showed no significant association between ANM and breast cancer, endometrial cancer, ovarian cancer, coronary heart disease, ischemic stroke, and Alzheimer’s disease.
- The clear lack of a causal effect of ANM on the outcomes of coronary heart disease and ischemic stroke in the MR analyses despite a strong inverse association seen in the observational data of this study (without MR) suggests residual confounding plays a substantial role in driving the observed outcomes.
Why this matters
- The authors said that, to their knowledge, this is the first study that has shown a causal association between older ANM and higher risk of postmenopausal lung cancer.
- This finding was directionally opposite to the significant protective effect of increased ANM documented in an observational analysis of roughly the same data as well as prior reports that did not use MR. This “notable inconsistency” suggests very substantial residual confounding without MR that could be driven by factors such as smoking, diet, and exercise.
- If these results are replicated in additional datasets, it would highlight a need for randomized, controlled trials of antiestrogen therapies in postmenopausal women for the prevention or treatment of lung cancer.
Study design
- The study included data from 106,853 postmenopausal women enrolled in the Women’s Health Initiative (WHI) and 95,464 women who were 37-73 years old included in the UK Biobank (UKB). Analyses for each outcome also included data from smaller numbers of women obtained from several additional datasets.
- The MR analysis used up to 55 single-nucleotide polymorphisms previously discovered through a genome-wide association study of about 70,000 women of European ancestry and independent of all datasets analyzed in the current study. The authors included all single-nucleotide polymorphisms with a consistent direction of effect on ANM.
- The MR analysis for lung cancer included 113,371 women from the two primary datasets and an additional 3012 women from six additional datasets.
- The MR analysis for bone fracture involved 113,239 women from the WHI and UKB only. The MR analysis for osteoporosis involved 137,080 women from the WHI, UKB, and one additional external dataset.
Key results
- Results from a meta-analysis of the MR results using data from the WHI, UKB, and the additional datasets showed ANM was causally associated with an increased risk of lung cancer by an odds ratio of 1.35 for each 5-year increase in ANM. In contrast, the adjusted observational analysis of data just from the WHI and UKB showed a significant 11% relative risk reduction in the incidence of lung cancer for each 5-year increase in ANM.
- The MR results also showed causally protective effects for fracture, with a 24% relative risk reduction, and for osteoporosis, with a 19% relative risk reduction for each 5-year increase in ANM.
- The MR analyses showed no significant association between AMN and outcome for breast cancer, endometrial cancer, ovarian cancer, coronary heart disease, ischemic stroke, and Alzheimer’s disease.
Limitations
The main limitation of the MR study was the potential for inadequate power for assessing some outcomes despite the large overall size of the study cohort. Lack of adequate power may be responsible for some of the nonsignificant associations seen in the study, such as for breast and endometrial cancers, where substantial prior evidence has implicated increased risk through the effects of prolonged exposure to endogenous or exogenous estrogens.
The healthy cohort effect in the UKB is a known weakness of this dataset that may have limited the number of cases and generalizability of findings.
Osteoporosis and Alzheimer’s disease were self-reported.
The study only included participants of European ancestry because most subjects in most of the cohorts examined were White women and the applied MR instruments were found by genome-wide association studies run predominantly in White women. The authors said the causal effects of ANM need study in more diverse populations.
Disclosures
- The study received no commercial funding.
- None of the authors had disclosures.
This is a summary of a preprint research study, “Genetic evidence for causal relationships between age at natural menopause and the risk of aging-associated adverse health outcomes,” written by authors primarily based at Stanford University School of Medicine i
A version of this article first appeared on Medscape.com.
Stress and infertility – is it a proven cause and effect?
“Just relax, stop thinking about it and, more than likely, it will happen.” If ever there was a controversial subject in medicine, especially in reproduction, the relationship between stress and infertility would be high on the list. Who among us has not overheard or even personally shared with an infertility patient that they should try and reduce their stress to improve fertility? The theory is certainly not new. Hippocrates, back in the 5th century B.C., was one of the first to associate a woman’s psychological state with her reproductive potential. His contention was that a physical sign of psychological stress in women (which scholars later dubbed “hysteria”) could result in sterility. In medieval times, a German abbess and mystic named Hildegard of Bingen posited women suffering from melancholy – a condition that we today might call depression – were infertile as a result.
The deeper meaning behind the flippant advice to relax is implicit blame; that is, a woman interprets the link of stress and infertility as a declaration that she is sabotaging reproduction. Not only is this assumption flawed, but it does further damage to a woman’s emotional fragility. To provide the presumption of stress affecting reproduction, a recent survey of over 5,000 infertility patients found, remarkably, 98% considered emotional stress as either a cause or a contributor to infertility, and 31% believed stress was a cause of miscarriage, although racial differences existed (J Assist Reprod Genet. 2021 Apr;38[4]:877-87). This relationship was mostly seen in women who used complementary and alternative medicine, Black women, and those who frequented Internet search engines. Whereas women who had a professional degree, had more infertility insurance coverage, and were nonreligious were less likely to attribute stress to infertility. Intriguingly, the more engaged the physicians, the less patients linked stress with infertility, while the contrary also applied.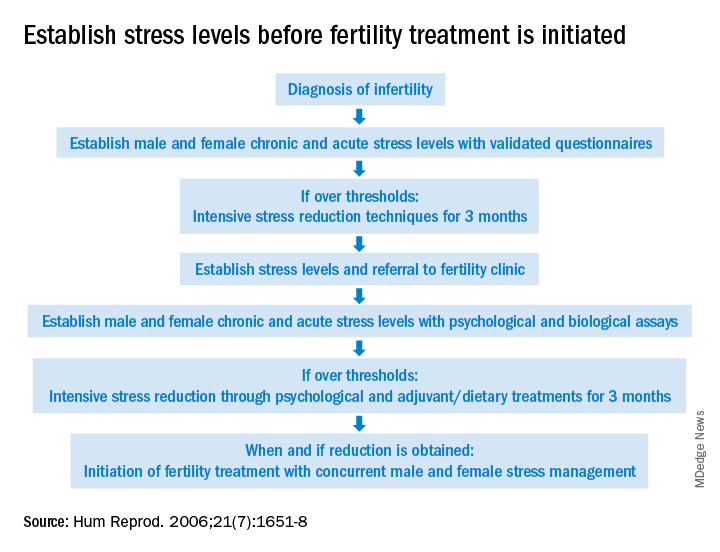
The power of stress can be exemplified by the pathophysiology of amenorrhea. Functional hypothalamic amenorrhea is the most common cause of the female athlete triad of secondary amenorrhea in women of childbearing age. It is a reversible disorder caused by stress related to weight loss, excessive exercise and/or traumatic mental experiences (Endocrines. 2021;2:203-11). Stress of infertility has also been demonstrated to be equivalent to a diagnosis of cancer and other major medical morbidities (J Psychosom Obstet Gynaecol. 1993;14[Suppl]:45-52).
A definitive link between stress and infertility is evasive because of the lack of controlled, prospective longitudinal studies and the challenge of reducing variables in the analysis. The question remains which developed initially – the stress or the infertility? Infertility treatment is a physical, emotional, and financial investment. Stress and the duration of infertility are correlative. The additive factor is that poor insurance coverage for costly fertility treatment can not only heighten stress but, concurrently, subject the patient to the risk of exploitation driven by desperation whereby they accept unproven “add-ons” offered with assisted reproductive technologies (ART).
Both acute and chronic stress affect the number of oocytes retrieved and fertilized with ART as well as live birth delivery and birth weights (Fertil Steril. 2001;76:675-87). Men are also affected by stress, which is manifested by decreased libido and impaired semen, further compromised as the duration of infertility continues. The gut-derived hormone ghrelin appears to play a role with stress and reproduction (Endocr Rev. 2017;38:432-67).
As the relationship between stress and infertility is far from proven, there are conflicting study results. Two meta-analyses failed to show any association between stress and the outcomes of ART cycles (Hum Reprod. 2011;26:2763-76; BMJ. 2011;342:d223). In contrast, a recent study suggested stress during infertility treatment was contributed by the variables of low spousal support, financial constraints, and social coercion in the early years of marriage (J Hum Reprod Sci. 2018;11:172-9). Emotional distress was found to be three times greater in women whose families had unrealistic expectations from treatments.
Fortunately, psychotherapy during the ART cycle has demonstrated a benefit in outcomes. Domar revealed psychological support and cognitive behavior therapy resulted in higher pregnancy rates than in the control group (Fertil Steril. 2000;73:805-12). Another recent study appears to support stress reduction improving reproductive potential (Dialogues Clin Neurosci. 2018;20[1]:41-7).
Given the evidence provided in this article, it behooves infertility clinics to address baseline (chronic) stress and acute stress (because of infertility) prior to initiating treatment (see Figure). While the definitive answer addressing the impact of stress on reproduction remains unknown, we may share with our patients a definition in which they may find enlightenment, “Stress is trying to control an event in which one is incapable.”
Dr. Mark P Trolice is director of Fertility CARE: The IVF Center in Winter Park, Fla., and associate professor of obstetrics and gynecology at the University of Central Florida, Orlando.
“Just relax, stop thinking about it and, more than likely, it will happen.” If ever there was a controversial subject in medicine, especially in reproduction, the relationship between stress and infertility would be high on the list. Who among us has not overheard or even personally shared with an infertility patient that they should try and reduce their stress to improve fertility? The theory is certainly not new. Hippocrates, back in the 5th century B.C., was one of the first to associate a woman’s psychological state with her reproductive potential. His contention was that a physical sign of psychological stress in women (which scholars later dubbed “hysteria”) could result in sterility. In medieval times, a German abbess and mystic named Hildegard of Bingen posited women suffering from melancholy – a condition that we today might call depression – were infertile as a result.
The deeper meaning behind the flippant advice to relax is implicit blame; that is, a woman interprets the link of stress and infertility as a declaration that she is sabotaging reproduction. Not only is this assumption flawed, but it does further damage to a woman’s emotional fragility. To provide the presumption of stress affecting reproduction, a recent survey of over 5,000 infertility patients found, remarkably, 98% considered emotional stress as either a cause or a contributor to infertility, and 31% believed stress was a cause of miscarriage, although racial differences existed (J Assist Reprod Genet. 2021 Apr;38[4]:877-87). This relationship was mostly seen in women who used complementary and alternative medicine, Black women, and those who frequented Internet search engines. Whereas women who had a professional degree, had more infertility insurance coverage, and were nonreligious were less likely to attribute stress to infertility. Intriguingly, the more engaged the physicians, the less patients linked stress with infertility, while the contrary also applied.
The power of stress can be exemplified by the pathophysiology of amenorrhea. Functional hypothalamic amenorrhea is the most common cause of the female athlete triad of secondary amenorrhea in women of childbearing age. It is a reversible disorder caused by stress related to weight loss, excessive exercise and/or traumatic mental experiences (Endocrines. 2021;2:203-11). Stress of infertility has also been demonstrated to be equivalent to a diagnosis of cancer and other major medical morbidities (J Psychosom Obstet Gynaecol. 1993;14[Suppl]:45-52).
A definitive link between stress and infertility is evasive because of the lack of controlled, prospective longitudinal studies and the challenge of reducing variables in the analysis. The question remains which developed initially – the stress or the infertility? Infertility treatment is a physical, emotional, and financial investment. Stress and the duration of infertility are correlative. The additive factor is that poor insurance coverage for costly fertility treatment can not only heighten stress but, concurrently, subject the patient to the risk of exploitation driven by desperation whereby they accept unproven “add-ons” offered with assisted reproductive technologies (ART).
Both acute and chronic stress affect the number of oocytes retrieved and fertilized with ART as well as live birth delivery and birth weights (Fertil Steril. 2001;76:675-87). Men are also affected by stress, which is manifested by decreased libido and impaired semen, further compromised as the duration of infertility continues. The gut-derived hormone ghrelin appears to play a role with stress and reproduction (Endocr Rev. 2017;38:432-67).
As the relationship between stress and infertility is far from proven, there are conflicting study results. Two meta-analyses failed to show any association between stress and the outcomes of ART cycles (Hum Reprod. 2011;26:2763-76; BMJ. 2011;342:d223). In contrast, a recent study suggested stress during infertility treatment was contributed by the variables of low spousal support, financial constraints, and social coercion in the early years of marriage (J Hum Reprod Sci. 2018;11:172-9). Emotional distress was found to be three times greater in women whose families had unrealistic expectations from treatments.
Fortunately, psychotherapy during the ART cycle has demonstrated a benefit in outcomes. Domar revealed psychological support and cognitive behavior therapy resulted in higher pregnancy rates than in the control group (Fertil Steril. 2000;73:805-12). Another recent study appears to support stress reduction improving reproductive potential (Dialogues Clin Neurosci. 2018;20[1]:41-7).
Given the evidence provided in this article, it behooves infertility clinics to address baseline (chronic) stress and acute stress (because of infertility) prior to initiating treatment (see Figure). While the definitive answer addressing the impact of stress on reproduction remains unknown, we may share with our patients a definition in which they may find enlightenment, “Stress is trying to control an event in which one is incapable.”
Dr. Mark P Trolice is director of Fertility CARE: The IVF Center in Winter Park, Fla., and associate professor of obstetrics and gynecology at the University of Central Florida, Orlando.
“Just relax, stop thinking about it and, more than likely, it will happen.” If ever there was a controversial subject in medicine, especially in reproduction, the relationship between stress and infertility would be high on the list. Who among us has not overheard or even personally shared with an infertility patient that they should try and reduce their stress to improve fertility? The theory is certainly not new. Hippocrates, back in the 5th century B.C., was one of the first to associate a woman’s psychological state with her reproductive potential. His contention was that a physical sign of psychological stress in women (which scholars later dubbed “hysteria”) could result in sterility. In medieval times, a German abbess and mystic named Hildegard of Bingen posited women suffering from melancholy – a condition that we today might call depression – were infertile as a result.
The deeper meaning behind the flippant advice to relax is implicit blame; that is, a woman interprets the link of stress and infertility as a declaration that she is sabotaging reproduction. Not only is this assumption flawed, but it does further damage to a woman’s emotional fragility. To provide the presumption of stress affecting reproduction, a recent survey of over 5,000 infertility patients found, remarkably, 98% considered emotional stress as either a cause or a contributor to infertility, and 31% believed stress was a cause of miscarriage, although racial differences existed (J Assist Reprod Genet. 2021 Apr;38[4]:877-87). This relationship was mostly seen in women who used complementary and alternative medicine, Black women, and those who frequented Internet search engines. Whereas women who had a professional degree, had more infertility insurance coverage, and were nonreligious were less likely to attribute stress to infertility. Intriguingly, the more engaged the physicians, the less patients linked stress with infertility, while the contrary also applied.
The power of stress can be exemplified by the pathophysiology of amenorrhea. Functional hypothalamic amenorrhea is the most common cause of the female athlete triad of secondary amenorrhea in women of childbearing age. It is a reversible disorder caused by stress related to weight loss, excessive exercise and/or traumatic mental experiences (Endocrines. 2021;2:203-11). Stress of infertility has also been demonstrated to be equivalent to a diagnosis of cancer and other major medical morbidities (J Psychosom Obstet Gynaecol. 1993;14[Suppl]:45-52).
A definitive link between stress and infertility is evasive because of the lack of controlled, prospective longitudinal studies and the challenge of reducing variables in the analysis. The question remains which developed initially – the stress or the infertility? Infertility treatment is a physical, emotional, and financial investment. Stress and the duration of infertility are correlative. The additive factor is that poor insurance coverage for costly fertility treatment can not only heighten stress but, concurrently, subject the patient to the risk of exploitation driven by desperation whereby they accept unproven “add-ons” offered with assisted reproductive technologies (ART).
Both acute and chronic stress affect the number of oocytes retrieved and fertilized with ART as well as live birth delivery and birth weights (Fertil Steril. 2001;76:675-87). Men are also affected by stress, which is manifested by decreased libido and impaired semen, further compromised as the duration of infertility continues. The gut-derived hormone ghrelin appears to play a role with stress and reproduction (Endocr Rev. 2017;38:432-67).
As the relationship between stress and infertility is far from proven, there are conflicting study results. Two meta-analyses failed to show any association between stress and the outcomes of ART cycles (Hum Reprod. 2011;26:2763-76; BMJ. 2011;342:d223). In contrast, a recent study suggested stress during infertility treatment was contributed by the variables of low spousal support, financial constraints, and social coercion in the early years of marriage (J Hum Reprod Sci. 2018;11:172-9). Emotional distress was found to be three times greater in women whose families had unrealistic expectations from treatments.
Fortunately, psychotherapy during the ART cycle has demonstrated a benefit in outcomes. Domar revealed psychological support and cognitive behavior therapy resulted in higher pregnancy rates than in the control group (Fertil Steril. 2000;73:805-12). Another recent study appears to support stress reduction improving reproductive potential (Dialogues Clin Neurosci. 2018;20[1]:41-7).
Given the evidence provided in this article, it behooves infertility clinics to address baseline (chronic) stress and acute stress (because of infertility) prior to initiating treatment (see Figure). While the definitive answer addressing the impact of stress on reproduction remains unknown, we may share with our patients a definition in which they may find enlightenment, “Stress is trying to control an event in which one is incapable.”
Dr. Mark P Trolice is director of Fertility CARE: The IVF Center in Winter Park, Fla., and associate professor of obstetrics and gynecology at the University of Central Florida, Orlando.
More than half of U.S. women enter pregnancy at higher CVD risk
Less than half of women in the United States enter pregnancy in favorable cardiovascular health, new research suggests.
In 2019, among women aged 20 to 44 years with live births in the United States, only 40.2% were in favorable cardiovascular health prior to pregnancy, defined as normal weight, no diabetes, and no hypertension.
Although all regions and states showed a decline in prepregnancy favorable cardiometabolic health, there were significant differences among geographic regions in the country, the authors report. “These data reveal critical deficiencies and geographic disparities in prepregnancy cardiometabolic health,” they conclude.
“One of the things that we know in the U.S. is that the maternal mortality rate has been increasing, and there are significant differences at the state level in both adverse maternal outcomes, such as maternal mortality, as well as adverse pregnancy outcomes,” corresponding author Sadiya S. Khan, MD, MS, FACC, Northwestern University Feinberg School of Medicine, Chicago, told this news organization.
“These outcomes are often related to health factors that predate pregnancy,” Dr. Khan explained, “and the processes that begin at the very, very beginning of conception are informed by health factors prior to pregnancy, in particular cardiometabolic factors like body mass index or obesity, high blood pressure, and diabetes.”
The results were published online on Feb. 14 in a special “Go Red for Women” spotlight issue of Circulation.
Cardiometabolic health factors
Using maternal birth records from live births in the Centers for Disease Control and Prevention Natality Database between 2016 and 2019, the authors analyzed data on 14,174,625 women with live births aged 20 to 44 years. The majority (81.4%) were 20 to 34 years of age, 22.7% were Hispanic or Latina, and 52.7% were non-Hispanic White.
Favorable cardiometabolic health was defined as a BMI of 18 to 24.9 kg/m2, absence of diabetes, and absence of hypertension.
Although all regions and states experienced a decline in favorable cardiometabolic health during the study period of 2016 to 2019, with a drop overall of 3.2% – from 43.5 to 40.2 per 100 live births – it was especially true of the South and Midwest regions.
In 2019, favorable prepregnancy cardiometabolic health was lowest in the South (38.1%) and Midwest (38.8%) and highest in the West (42.2%) and Northeast (43.6%).
State by state, the lowest prevalence of favorable cardiometabolic health was found in Mississippi, at 31.2%, and highest in Utah, at 47.2%.
They also found a correlation between favorable cardiometabolic health and state-level percentages of high-school education or less and enrollment in Medicaid in 2019.
Similar to what has been seen with cardiovascular disease, “we observe that the states with the lowest prevalence of favorable cardiometabolic health were in the Southeast United States,” said Dr. Khan, “and similar geographic variation was observed with some more patterns in education and Medicaid coverage for birth, and these were used as proxies for socioeconomic status in those areas.”
Although Dr. Khan notes that the relationships cannot be determined to be causal from this analysis, she said that “it does suggest that upstream social determinants of health are important determinants of cardiometabolic health.”
Socioeconomic intervention
Dr. Khan noted that policies at the federal and state level can identify ways to “ensure that individuals who are thinking about pregnancy have access to health care and have access to resources, too, from a broad range of health determinants, including housing stability, food security, as well as access to health care be optimized prior to pregnancy.”
The authors note that this analysis may actually overestimate the prevalence of favorable cardiometabolic health, and data on cholesterol, diet, a distinction between type 1 and type 2 diabetes, and physical activity were not available.
Only individuals with live births were included, which could result in the elimination of a potentially high-risk group; however, late pregnancy losses represent less than 0.3% of all pregnancies, they say.
The authors conclude that “future research is needed to equitably improve health prior to pregnancy and quantify the potential benefits in cardiovascular disease outcomes for birthing individuals and their offspring.”
This work was supported by grants from the National Heart, Lung, and Blood Institute and American Heart Association Transformational Project Award awarded to Sadiya S. Khan.
A version of this article first appeared on Medscape.com.
Less than half of women in the United States enter pregnancy in favorable cardiovascular health, new research suggests.
In 2019, among women aged 20 to 44 years with live births in the United States, only 40.2% were in favorable cardiovascular health prior to pregnancy, defined as normal weight, no diabetes, and no hypertension.
Although all regions and states showed a decline in prepregnancy favorable cardiometabolic health, there were significant differences among geographic regions in the country, the authors report. “These data reveal critical deficiencies and geographic disparities in prepregnancy cardiometabolic health,” they conclude.
“One of the things that we know in the U.S. is that the maternal mortality rate has been increasing, and there are significant differences at the state level in both adverse maternal outcomes, such as maternal mortality, as well as adverse pregnancy outcomes,” corresponding author Sadiya S. Khan, MD, MS, FACC, Northwestern University Feinberg School of Medicine, Chicago, told this news organization.
“These outcomes are often related to health factors that predate pregnancy,” Dr. Khan explained, “and the processes that begin at the very, very beginning of conception are informed by health factors prior to pregnancy, in particular cardiometabolic factors like body mass index or obesity, high blood pressure, and diabetes.”
The results were published online on Feb. 14 in a special “Go Red for Women” spotlight issue of Circulation.
Cardiometabolic health factors
Using maternal birth records from live births in the Centers for Disease Control and Prevention Natality Database between 2016 and 2019, the authors analyzed data on 14,174,625 women with live births aged 20 to 44 years. The majority (81.4%) were 20 to 34 years of age, 22.7% were Hispanic or Latina, and 52.7% were non-Hispanic White.
Favorable cardiometabolic health was defined as a BMI of 18 to 24.9 kg/m2, absence of diabetes, and absence of hypertension.
Although all regions and states experienced a decline in favorable cardiometabolic health during the study period of 2016 to 2019, with a drop overall of 3.2% – from 43.5 to 40.2 per 100 live births – it was especially true of the South and Midwest regions.
In 2019, favorable prepregnancy cardiometabolic health was lowest in the South (38.1%) and Midwest (38.8%) and highest in the West (42.2%) and Northeast (43.6%).
State by state, the lowest prevalence of favorable cardiometabolic health was found in Mississippi, at 31.2%, and highest in Utah, at 47.2%.
They also found a correlation between favorable cardiometabolic health and state-level percentages of high-school education or less and enrollment in Medicaid in 2019.
Similar to what has been seen with cardiovascular disease, “we observe that the states with the lowest prevalence of favorable cardiometabolic health were in the Southeast United States,” said Dr. Khan, “and similar geographic variation was observed with some more patterns in education and Medicaid coverage for birth, and these were used as proxies for socioeconomic status in those areas.”
Although Dr. Khan notes that the relationships cannot be determined to be causal from this analysis, she said that “it does suggest that upstream social determinants of health are important determinants of cardiometabolic health.”
Socioeconomic intervention
Dr. Khan noted that policies at the federal and state level can identify ways to “ensure that individuals who are thinking about pregnancy have access to health care and have access to resources, too, from a broad range of health determinants, including housing stability, food security, as well as access to health care be optimized prior to pregnancy.”
The authors note that this analysis may actually overestimate the prevalence of favorable cardiometabolic health, and data on cholesterol, diet, a distinction between type 1 and type 2 diabetes, and physical activity were not available.
Only individuals with live births were included, which could result in the elimination of a potentially high-risk group; however, late pregnancy losses represent less than 0.3% of all pregnancies, they say.
The authors conclude that “future research is needed to equitably improve health prior to pregnancy and quantify the potential benefits in cardiovascular disease outcomes for birthing individuals and their offspring.”
This work was supported by grants from the National Heart, Lung, and Blood Institute and American Heart Association Transformational Project Award awarded to Sadiya S. Khan.
A version of this article first appeared on Medscape.com.
Less than half of women in the United States enter pregnancy in favorable cardiovascular health, new research suggests.
In 2019, among women aged 20 to 44 years with live births in the United States, only 40.2% were in favorable cardiovascular health prior to pregnancy, defined as normal weight, no diabetes, and no hypertension.
Although all regions and states showed a decline in prepregnancy favorable cardiometabolic health, there were significant differences among geographic regions in the country, the authors report. “These data reveal critical deficiencies and geographic disparities in prepregnancy cardiometabolic health,” they conclude.
“One of the things that we know in the U.S. is that the maternal mortality rate has been increasing, and there are significant differences at the state level in both adverse maternal outcomes, such as maternal mortality, as well as adverse pregnancy outcomes,” corresponding author Sadiya S. Khan, MD, MS, FACC, Northwestern University Feinberg School of Medicine, Chicago, told this news organization.
“These outcomes are often related to health factors that predate pregnancy,” Dr. Khan explained, “and the processes that begin at the very, very beginning of conception are informed by health factors prior to pregnancy, in particular cardiometabolic factors like body mass index or obesity, high blood pressure, and diabetes.”
The results were published online on Feb. 14 in a special “Go Red for Women” spotlight issue of Circulation.
Cardiometabolic health factors
Using maternal birth records from live births in the Centers for Disease Control and Prevention Natality Database between 2016 and 2019, the authors analyzed data on 14,174,625 women with live births aged 20 to 44 years. The majority (81.4%) were 20 to 34 years of age, 22.7% were Hispanic or Latina, and 52.7% were non-Hispanic White.
Favorable cardiometabolic health was defined as a BMI of 18 to 24.9 kg/m2, absence of diabetes, and absence of hypertension.
Although all regions and states experienced a decline in favorable cardiometabolic health during the study period of 2016 to 2019, with a drop overall of 3.2% – from 43.5 to 40.2 per 100 live births – it was especially true of the South and Midwest regions.
In 2019, favorable prepregnancy cardiometabolic health was lowest in the South (38.1%) and Midwest (38.8%) and highest in the West (42.2%) and Northeast (43.6%).
State by state, the lowest prevalence of favorable cardiometabolic health was found in Mississippi, at 31.2%, and highest in Utah, at 47.2%.
They also found a correlation between favorable cardiometabolic health and state-level percentages of high-school education or less and enrollment in Medicaid in 2019.
Similar to what has been seen with cardiovascular disease, “we observe that the states with the lowest prevalence of favorable cardiometabolic health were in the Southeast United States,” said Dr. Khan, “and similar geographic variation was observed with some more patterns in education and Medicaid coverage for birth, and these were used as proxies for socioeconomic status in those areas.”
Although Dr. Khan notes that the relationships cannot be determined to be causal from this analysis, she said that “it does suggest that upstream social determinants of health are important determinants of cardiometabolic health.”
Socioeconomic intervention
Dr. Khan noted that policies at the federal and state level can identify ways to “ensure that individuals who are thinking about pregnancy have access to health care and have access to resources, too, from a broad range of health determinants, including housing stability, food security, as well as access to health care be optimized prior to pregnancy.”
The authors note that this analysis may actually overestimate the prevalence of favorable cardiometabolic health, and data on cholesterol, diet, a distinction between type 1 and type 2 diabetes, and physical activity were not available.
Only individuals with live births were included, which could result in the elimination of a potentially high-risk group; however, late pregnancy losses represent less than 0.3% of all pregnancies, they say.
The authors conclude that “future research is needed to equitably improve health prior to pregnancy and quantify the potential benefits in cardiovascular disease outcomes for birthing individuals and their offspring.”
This work was supported by grants from the National Heart, Lung, and Blood Institute and American Heart Association Transformational Project Award awarded to Sadiya S. Khan.
A version of this article first appeared on Medscape.com.
When physicians are the plaintiffs
Have you experienced malpractice?
No, I’m not asking whether you have experienced litigation. I’m asking whether you, as a physician, have actually experienced substandard care from a colleague. I have heard many such experiences over the years, and mistreatment doesn’t seem to be getting any less frequent.
The first is that, unlike the Pope, who has a dedicated confessor trained to minister to his spiritual needs, no one formally trains physicians to treat physicians. As a result, most of us feel slightly uneasy at treating other physicians. We naturally wish to keep our colleagues well, but at the same time realize that our clinical skills are being very closely scrutinized. What if they are found to be wanting? This discomfiture can make a physician treating a physician overly compulsive, or worse, overtly dismissive.
Second, we physicians are famously poor patients. We pretend we don’t need the advice we give others, to monitor our health and promptly seek care when something feels amiss. And, for the period during which we delay a medical encounter, we often attempt to diagnose and treat ourselves.
Sometimes we are successful, which reinforces this approach. Other times, we fail at being our own caregiver and present to someone else either too late, or with avoidable complications. In the former instance, we congratulate ourselves and learn nothing from the experience. In the latter, we may heap shame upon ourselves for our folly, and we may learn; but it could be a lethal lesson. In the worst scenario, our colleague gives in to frustration (or angst), and heaps even more shame onto their late-presenting physician patient.
Third, when we do submit to being a patient, we often demand VIP treatment. This is probably in response to our anxiety that some of the worst things we have seen happen to patients might happen to us if we are not vigilant to ensure we receive a higher level of care. But of course, such hypervigilance can lead to excessive care and testing, with all the attendant hazards, or alternatively to dilution of care if our caregivers decide we are just too much trouble.
Fourth, as a fifth-generation physician myself, I am convinced that physicians and physician family members are either prone to unusual manifestations of common diseases or unusual diseases, or that rare disease entities and complications are actually more common than literature suggests, and they simply aren’t pursued or diagnosed in nonphysician families.
No matter how we may have arrived in a position to need medical care, how often is such care substandard? And how do we respond when we suspect, or know, this to be the case? Are physicians more, or less, likely to take legal action in the face of it?
I certainly don’t know any statistics. Physicians are in an excellent position to take such action, because judges and juries will likely believe that a doctor can recognize negligence when we fall victim to it. But we may also be reluctant to publicly admit the way (or ways) in which we may have contributed to substandard care or outcome.
Based on decades of working with physician clients who have been sued, and having been sued myself (thus witnessing and also experiencing the effects of litigation), I am probably more reluctant than normal patients or physicians to consider taking legal action. This, despite the fact that I am also a lawyer and (through organized medicine) know many colleagues in all specialties who might serve as expert witnesses.
I have experienced serial substandard care, which has left me highly conflicted about the efficacy of my chosen profession. As a resident, I had my first odd pain condition and consulted an “elder statesperson” from my institution, whom I assumed to be a “doctor’s doctor” because he was a superb teacher (wrong!)
He completely missed the diagnosis and further belittled (indeed, libeled) me in the medical record. (Some years later, I learned that, during that period, he was increasingly demented and tended to view all female patients as having “wandering uterus” equivalents.) Fortunately, I found a better diagnostician, or at least one more willing to lend credence to my complaints, who successfully removed the first of several “zebra” lesions I have experienced.
As a young faculty member, I had an odd presentation of a recurring gynecologic condition, which was treated surgically, successfully, except that my fertility was cut in half – a possibility about which I had not been informed when giving operative consent. Would I have sued this fellow faculty member for that? Never, because she invariably treated me with respect as a colleague.
Later in my career after leaving academia, the same condition recurred in a new location. My old-school gynecologist desired to do an extensive procedure, to which I demurred unless specific pathology was found intraoperatively. Affronted, he subjected me to laparoscopy, did nothing but look, and then left the hospital leaving me and the PACU nurse to try to decipher his instructions (which said, basically, “I didn’t find anything; don’t bother me again.”). Several years of pain later, a younger gynecologist performed the correct procedure to address my problem, which has never recurred. Would I have sued him? No, because I believe he had a disability.
At age 59, I developed a new mole. My beloved general practitioner, in the waning years of his practice, forgot to consult a colleague to remove it for several months. When I forced the issue, the mole was removed and turned out to be a rare pediatric condition considered a precursor to melanoma. The same general practitioner had told me I needn’t worry about my “mild hypercalcemia.”
Ten years later I diagnosed my own parathyroid adenoma, in the interim losing 10% of my bone density. Would I have sued him? No, for he always showed he cared. (Though maybe, if I had fractured my spine or hip.)
If you have been the victim of physician malpractice, how did you respond?
Do we serve our profession well by how we handle substandard care – upon ourselves (or our loved ones)?
Dr. Andrew is a former assistant professor in the department of emergency medicine, Johns Hopkins University, Baltimore, and founder and principal of MDMentor, Victoria, B.C.
A version of this article first appeared on Medscape.com.
Have you experienced malpractice?
No, I’m not asking whether you have experienced litigation. I’m asking whether you, as a physician, have actually experienced substandard care from a colleague. I have heard many such experiences over the years, and mistreatment doesn’t seem to be getting any less frequent.
The first is that, unlike the Pope, who has a dedicated confessor trained to minister to his spiritual needs, no one formally trains physicians to treat physicians. As a result, most of us feel slightly uneasy at treating other physicians. We naturally wish to keep our colleagues well, but at the same time realize that our clinical skills are being very closely scrutinized. What if they are found to be wanting? This discomfiture can make a physician treating a physician overly compulsive, or worse, overtly dismissive.
Second, we physicians are famously poor patients. We pretend we don’t need the advice we give others, to monitor our health and promptly seek care when something feels amiss. And, for the period during which we delay a medical encounter, we often attempt to diagnose and treat ourselves.
Sometimes we are successful, which reinforces this approach. Other times, we fail at being our own caregiver and present to someone else either too late, or with avoidable complications. In the former instance, we congratulate ourselves and learn nothing from the experience. In the latter, we may heap shame upon ourselves for our folly, and we may learn; but it could be a lethal lesson. In the worst scenario, our colleague gives in to frustration (or angst), and heaps even more shame onto their late-presenting physician patient.
Third, when we do submit to being a patient, we often demand VIP treatment. This is probably in response to our anxiety that some of the worst things we have seen happen to patients might happen to us if we are not vigilant to ensure we receive a higher level of care. But of course, such hypervigilance can lead to excessive care and testing, with all the attendant hazards, or alternatively to dilution of care if our caregivers decide we are just too much trouble.
Fourth, as a fifth-generation physician myself, I am convinced that physicians and physician family members are either prone to unusual manifestations of common diseases or unusual diseases, or that rare disease entities and complications are actually more common than literature suggests, and they simply aren’t pursued or diagnosed in nonphysician families.
No matter how we may have arrived in a position to need medical care, how often is such care substandard? And how do we respond when we suspect, or know, this to be the case? Are physicians more, or less, likely to take legal action in the face of it?
I certainly don’t know any statistics. Physicians are in an excellent position to take such action, because judges and juries will likely believe that a doctor can recognize negligence when we fall victim to it. But we may also be reluctant to publicly admit the way (or ways) in which we may have contributed to substandard care or outcome.
Based on decades of working with physician clients who have been sued, and having been sued myself (thus witnessing and also experiencing the effects of litigation), I am probably more reluctant than normal patients or physicians to consider taking legal action. This, despite the fact that I am also a lawyer and (through organized medicine) know many colleagues in all specialties who might serve as expert witnesses.
I have experienced serial substandard care, which has left me highly conflicted about the efficacy of my chosen profession. As a resident, I had my first odd pain condition and consulted an “elder statesperson” from my institution, whom I assumed to be a “doctor’s doctor” because he was a superb teacher (wrong!)
He completely missed the diagnosis and further belittled (indeed, libeled) me in the medical record. (Some years later, I learned that, during that period, he was increasingly demented and tended to view all female patients as having “wandering uterus” equivalents.) Fortunately, I found a better diagnostician, or at least one more willing to lend credence to my complaints, who successfully removed the first of several “zebra” lesions I have experienced.
As a young faculty member, I had an odd presentation of a recurring gynecologic condition, which was treated surgically, successfully, except that my fertility was cut in half – a possibility about which I had not been informed when giving operative consent. Would I have sued this fellow faculty member for that? Never, because she invariably treated me with respect as a colleague.
Later in my career after leaving academia, the same condition recurred in a new location. My old-school gynecologist desired to do an extensive procedure, to which I demurred unless specific pathology was found intraoperatively. Affronted, he subjected me to laparoscopy, did nothing but look, and then left the hospital leaving me and the PACU nurse to try to decipher his instructions (which said, basically, “I didn’t find anything; don’t bother me again.”). Several years of pain later, a younger gynecologist performed the correct procedure to address my problem, which has never recurred. Would I have sued him? No, because I believe he had a disability.
At age 59, I developed a new mole. My beloved general practitioner, in the waning years of his practice, forgot to consult a colleague to remove it for several months. When I forced the issue, the mole was removed and turned out to be a rare pediatric condition considered a precursor to melanoma. The same general practitioner had told me I needn’t worry about my “mild hypercalcemia.”
Ten years later I diagnosed my own parathyroid adenoma, in the interim losing 10% of my bone density. Would I have sued him? No, for he always showed he cared. (Though maybe, if I had fractured my spine or hip.)
If you have been the victim of physician malpractice, how did you respond?
Do we serve our profession well by how we handle substandard care – upon ourselves (or our loved ones)?
Dr. Andrew is a former assistant professor in the department of emergency medicine, Johns Hopkins University, Baltimore, and founder and principal of MDMentor, Victoria, B.C.
A version of this article first appeared on Medscape.com.
Have you experienced malpractice?
No, I’m not asking whether you have experienced litigation. I’m asking whether you, as a physician, have actually experienced substandard care from a colleague. I have heard many such experiences over the years, and mistreatment doesn’t seem to be getting any less frequent.
The first is that, unlike the Pope, who has a dedicated confessor trained to minister to his spiritual needs, no one formally trains physicians to treat physicians. As a result, most of us feel slightly uneasy at treating other physicians. We naturally wish to keep our colleagues well, but at the same time realize that our clinical skills are being very closely scrutinized. What if they are found to be wanting? This discomfiture can make a physician treating a physician overly compulsive, or worse, overtly dismissive.
Second, we physicians are famously poor patients. We pretend we don’t need the advice we give others, to monitor our health and promptly seek care when something feels amiss. And, for the period during which we delay a medical encounter, we often attempt to diagnose and treat ourselves.
Sometimes we are successful, which reinforces this approach. Other times, we fail at being our own caregiver and present to someone else either too late, or with avoidable complications. In the former instance, we congratulate ourselves and learn nothing from the experience. In the latter, we may heap shame upon ourselves for our folly, and we may learn; but it could be a lethal lesson. In the worst scenario, our colleague gives in to frustration (or angst), and heaps even more shame onto their late-presenting physician patient.
Third, when we do submit to being a patient, we often demand VIP treatment. This is probably in response to our anxiety that some of the worst things we have seen happen to patients might happen to us if we are not vigilant to ensure we receive a higher level of care. But of course, such hypervigilance can lead to excessive care and testing, with all the attendant hazards, or alternatively to dilution of care if our caregivers decide we are just too much trouble.
Fourth, as a fifth-generation physician myself, I am convinced that physicians and physician family members are either prone to unusual manifestations of common diseases or unusual diseases, or that rare disease entities and complications are actually more common than literature suggests, and they simply aren’t pursued or diagnosed in nonphysician families.
No matter how we may have arrived in a position to need medical care, how often is such care substandard? And how do we respond when we suspect, or know, this to be the case? Are physicians more, or less, likely to take legal action in the face of it?
I certainly don’t know any statistics. Physicians are in an excellent position to take such action, because judges and juries will likely believe that a doctor can recognize negligence when we fall victim to it. But we may also be reluctant to publicly admit the way (or ways) in which we may have contributed to substandard care or outcome.
Based on decades of working with physician clients who have been sued, and having been sued myself (thus witnessing and also experiencing the effects of litigation), I am probably more reluctant than normal patients or physicians to consider taking legal action. This, despite the fact that I am also a lawyer and (through organized medicine) know many colleagues in all specialties who might serve as expert witnesses.
I have experienced serial substandard care, which has left me highly conflicted about the efficacy of my chosen profession. As a resident, I had my first odd pain condition and consulted an “elder statesperson” from my institution, whom I assumed to be a “doctor’s doctor” because he was a superb teacher (wrong!)
He completely missed the diagnosis and further belittled (indeed, libeled) me in the medical record. (Some years later, I learned that, during that period, he was increasingly demented and tended to view all female patients as having “wandering uterus” equivalents.) Fortunately, I found a better diagnostician, or at least one more willing to lend credence to my complaints, who successfully removed the first of several “zebra” lesions I have experienced.
As a young faculty member, I had an odd presentation of a recurring gynecologic condition, which was treated surgically, successfully, except that my fertility was cut in half – a possibility about which I had not been informed when giving operative consent. Would I have sued this fellow faculty member for that? Never, because she invariably treated me with respect as a colleague.
Later in my career after leaving academia, the same condition recurred in a new location. My old-school gynecologist desired to do an extensive procedure, to which I demurred unless specific pathology was found intraoperatively. Affronted, he subjected me to laparoscopy, did nothing but look, and then left the hospital leaving me and the PACU nurse to try to decipher his instructions (which said, basically, “I didn’t find anything; don’t bother me again.”). Several years of pain later, a younger gynecologist performed the correct procedure to address my problem, which has never recurred. Would I have sued him? No, because I believe he had a disability.
At age 59, I developed a new mole. My beloved general practitioner, in the waning years of his practice, forgot to consult a colleague to remove it for several months. When I forced the issue, the mole was removed and turned out to be a rare pediatric condition considered a precursor to melanoma. The same general practitioner had told me I needn’t worry about my “mild hypercalcemia.”
Ten years later I diagnosed my own parathyroid adenoma, in the interim losing 10% of my bone density. Would I have sued him? No, for he always showed he cared. (Though maybe, if I had fractured my spine or hip.)
If you have been the victim of physician malpractice, how did you respond?
Do we serve our profession well by how we handle substandard care – upon ourselves (or our loved ones)?
Dr. Andrew is a former assistant professor in the department of emergency medicine, Johns Hopkins University, Baltimore, and founder and principal of MDMentor, Victoria, B.C.
A version of this article first appeared on Medscape.com.
AHA targets ‘low-value’ heart care in new scientific statement
Low-value health care services that provide little or no benefit to patients are “common, potentially harmful, and costly,” and there is a critical need to reduce this kind of care, the American Heart Association said in a newly released scientific statement.
Each year, nearly half of patients in the United States will receive at least one low-value test or procedure, with the attendant risk of avoidable complications from cascades of care and excess costs to individuals and society, the authors noted. Reducing low-value care is particularly important in cardiology, given the high prevalence and costs of cardiovascular disease in the United States.
The statement was published online Feb. 22, 2022, in Circulation: Cardiovascular Quality and Outcomes.
High burden with uncertain benefit
“Cardiovascular disease is common and can present suddenly, such as a heart attack or abnormal heart rhythm,” Vinay Kini, MD, chair of the statement writing group and assistant professor of medicine at Weill Cornell Medicine, New York, said in a news release.
“Our desire to be vigilant about treating and preventing cardiovascular disease may sometimes lead to use of tests and procedures where the benefits to patients may be uncertain,” Dr. Kini said. “This may impose burdens on patients, in the form of increased risk of physical harm from the low-value procedure or potential complications, as well as follow-up care and out-of-pocket financial costs.”
For example, studies have shown that up to one in five echocardiograms and up to half of all stress tests performed in the United States may be rated as rarely appropriate, based on established guidelines for their use.
In addition, up to 15% of percutaneous coronary interventions (PCIs) are classified as rarely appropriate, the writing group said.
Annually, among Medicare fee-for-service beneficiaries, low-value stress testing in patients with stable coronary artery disease is estimated to cost between $212 million and $2.1 billion, while costs of PCI for stable CAD range from $212 million to $2.8 billion, the writing group noted.
“At best, spending on low-value care potentially diverts resources from higher-value services that would benefit patients more effectively at the same or reduced cost. At worst, low-value care results in physical harm in the form of preventable morbidity and mortality,” they said.
“Thus, reducing low-value care is one of the few patient-centered solutions that directly address both the need to control health care spending and the societal imperative to devote its limited resources to beneficial health care services that improve health,” they added.
The group outlines several ways to reduce low-value cardiovascular care targeting patients, providers, and payers/policymakers.
For patients, education and shared decision-making may help reduce low-value care and dispel misconceptions about the intended purpose of test or treatment, they suggested.
For clinicians, a “layered” approach to reducing low-value care may be most effective, such as through education, audit and feedback, and behavioral science tools (“nudges”) to shift behaviors and practices, they said.
For payers and policy leaders, interventions to reduce low-value care include national insurance coverage determinations; prior authorization; alternative payment models that reward lower costs and higher-quality health care; value-based insurance designs that financially penalize low-value care; and medical liability reform to reduce defensive medical practices.
Low-value cardiovascular care is a complex problem, the writing group acknowledged, and achieving meaningful reductions in low-value cardiovascular care will require a multidisciplinary approach that includes continuous research, implementation, evaluation, and adjustment while ensuring equitable access to care.
“Each approach has benefits and drawbacks,” Dr. Kini said. “For example, prior authorization imposes a large burden on health care professionals to obtain insurance approval for tests and treatments. Prior authorization and some value-based payment models may unintentionally worsen existing racial and ethnic health care disparities.
“A one-size-fits-all approach to reducing low-value care is unlikely to succeed; rather, acting through multiple perspectives and frequently measuring impacts and potential unintended consequences is critical,” he concluded.
The scientific statement was prepared by the volunteer writing group on behalf of the AHA’s Council on Quality of Care and Outcomes Research.
The research had no commercial funding. Dr. Kini disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Low-value health care services that provide little or no benefit to patients are “common, potentially harmful, and costly,” and there is a critical need to reduce this kind of care, the American Heart Association said in a newly released scientific statement.
Each year, nearly half of patients in the United States will receive at least one low-value test or procedure, with the attendant risk of avoidable complications from cascades of care and excess costs to individuals and society, the authors noted. Reducing low-value care is particularly important in cardiology, given the high prevalence and costs of cardiovascular disease in the United States.
The statement was published online Feb. 22, 2022, in Circulation: Cardiovascular Quality and Outcomes.
High burden with uncertain benefit
“Cardiovascular disease is common and can present suddenly, such as a heart attack or abnormal heart rhythm,” Vinay Kini, MD, chair of the statement writing group and assistant professor of medicine at Weill Cornell Medicine, New York, said in a news release.
“Our desire to be vigilant about treating and preventing cardiovascular disease may sometimes lead to use of tests and procedures where the benefits to patients may be uncertain,” Dr. Kini said. “This may impose burdens on patients, in the form of increased risk of physical harm from the low-value procedure or potential complications, as well as follow-up care and out-of-pocket financial costs.”
For example, studies have shown that up to one in five echocardiograms and up to half of all stress tests performed in the United States may be rated as rarely appropriate, based on established guidelines for their use.
In addition, up to 15% of percutaneous coronary interventions (PCIs) are classified as rarely appropriate, the writing group said.
Annually, among Medicare fee-for-service beneficiaries, low-value stress testing in patients with stable coronary artery disease is estimated to cost between $212 million and $2.1 billion, while costs of PCI for stable CAD range from $212 million to $2.8 billion, the writing group noted.
“At best, spending on low-value care potentially diverts resources from higher-value services that would benefit patients more effectively at the same or reduced cost. At worst, low-value care results in physical harm in the form of preventable morbidity and mortality,” they said.
“Thus, reducing low-value care is one of the few patient-centered solutions that directly address both the need to control health care spending and the societal imperative to devote its limited resources to beneficial health care services that improve health,” they added.
The group outlines several ways to reduce low-value cardiovascular care targeting patients, providers, and payers/policymakers.
For patients, education and shared decision-making may help reduce low-value care and dispel misconceptions about the intended purpose of test or treatment, they suggested.
For clinicians, a “layered” approach to reducing low-value care may be most effective, such as through education, audit and feedback, and behavioral science tools (“nudges”) to shift behaviors and practices, they said.
For payers and policy leaders, interventions to reduce low-value care include national insurance coverage determinations; prior authorization; alternative payment models that reward lower costs and higher-quality health care; value-based insurance designs that financially penalize low-value care; and medical liability reform to reduce defensive medical practices.
Low-value cardiovascular care is a complex problem, the writing group acknowledged, and achieving meaningful reductions in low-value cardiovascular care will require a multidisciplinary approach that includes continuous research, implementation, evaluation, and adjustment while ensuring equitable access to care.
“Each approach has benefits and drawbacks,” Dr. Kini said. “For example, prior authorization imposes a large burden on health care professionals to obtain insurance approval for tests and treatments. Prior authorization and some value-based payment models may unintentionally worsen existing racial and ethnic health care disparities.
“A one-size-fits-all approach to reducing low-value care is unlikely to succeed; rather, acting through multiple perspectives and frequently measuring impacts and potential unintended consequences is critical,” he concluded.
The scientific statement was prepared by the volunteer writing group on behalf of the AHA’s Council on Quality of Care and Outcomes Research.
The research had no commercial funding. Dr. Kini disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Low-value health care services that provide little or no benefit to patients are “common, potentially harmful, and costly,” and there is a critical need to reduce this kind of care, the American Heart Association said in a newly released scientific statement.
Each year, nearly half of patients in the United States will receive at least one low-value test or procedure, with the attendant risk of avoidable complications from cascades of care and excess costs to individuals and society, the authors noted. Reducing low-value care is particularly important in cardiology, given the high prevalence and costs of cardiovascular disease in the United States.
The statement was published online Feb. 22, 2022, in Circulation: Cardiovascular Quality and Outcomes.
High burden with uncertain benefit
“Cardiovascular disease is common and can present suddenly, such as a heart attack or abnormal heart rhythm,” Vinay Kini, MD, chair of the statement writing group and assistant professor of medicine at Weill Cornell Medicine, New York, said in a news release.
“Our desire to be vigilant about treating and preventing cardiovascular disease may sometimes lead to use of tests and procedures where the benefits to patients may be uncertain,” Dr. Kini said. “This may impose burdens on patients, in the form of increased risk of physical harm from the low-value procedure or potential complications, as well as follow-up care and out-of-pocket financial costs.”
For example, studies have shown that up to one in five echocardiograms and up to half of all stress tests performed in the United States may be rated as rarely appropriate, based on established guidelines for their use.
In addition, up to 15% of percutaneous coronary interventions (PCIs) are classified as rarely appropriate, the writing group said.
Annually, among Medicare fee-for-service beneficiaries, low-value stress testing in patients with stable coronary artery disease is estimated to cost between $212 million and $2.1 billion, while costs of PCI for stable CAD range from $212 million to $2.8 billion, the writing group noted.
“At best, spending on low-value care potentially diverts resources from higher-value services that would benefit patients more effectively at the same or reduced cost. At worst, low-value care results in physical harm in the form of preventable morbidity and mortality,” they said.
“Thus, reducing low-value care is one of the few patient-centered solutions that directly address both the need to control health care spending and the societal imperative to devote its limited resources to beneficial health care services that improve health,” they added.
The group outlines several ways to reduce low-value cardiovascular care targeting patients, providers, and payers/policymakers.
For patients, education and shared decision-making may help reduce low-value care and dispel misconceptions about the intended purpose of test or treatment, they suggested.
For clinicians, a “layered” approach to reducing low-value care may be most effective, such as through education, audit and feedback, and behavioral science tools (“nudges”) to shift behaviors and practices, they said.
For payers and policy leaders, interventions to reduce low-value care include national insurance coverage determinations; prior authorization; alternative payment models that reward lower costs and higher-quality health care; value-based insurance designs that financially penalize low-value care; and medical liability reform to reduce defensive medical practices.
Low-value cardiovascular care is a complex problem, the writing group acknowledged, and achieving meaningful reductions in low-value cardiovascular care will require a multidisciplinary approach that includes continuous research, implementation, evaluation, and adjustment while ensuring equitable access to care.
“Each approach has benefits and drawbacks,” Dr. Kini said. “For example, prior authorization imposes a large burden on health care professionals to obtain insurance approval for tests and treatments. Prior authorization and some value-based payment models may unintentionally worsen existing racial and ethnic health care disparities.
“A one-size-fits-all approach to reducing low-value care is unlikely to succeed; rather, acting through multiple perspectives and frequently measuring impacts and potential unintended consequences is critical,” he concluded.
The scientific statement was prepared by the volunteer writing group on behalf of the AHA’s Council on Quality of Care and Outcomes Research.
The research had no commercial funding. Dr. Kini disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CIRCULATION: CARDIOVASCULAR QUALITY AND OUTCOMES
FDA okays empagliflozin for HF regardless of ejection fraction
The Food and Drug Administration has approved an expanded heart failure indication for the sodium-glucose transporter 2 inhibitor empagliflozin (Jardiance) that now includes HF with mid-range or preserved left ventricular ejection fraction (LVEF), the agency announced on Feb. 24.
That means the SGLT2 inhibitor, once considered primarily an antidiabetic agent, is approved for use in patients with HF per se without regard to ventricular function. The drug received approval for HF with reduced LVEF in August 2021.
The expanded indication, specifically for reducing the risk of cardiovascular death and HF hospitalization in adults, was widely anticipated based on the landmark results from the EMPEROR-Preserved trial. The study saw a significant 21% relative reduction in that composite endpoint over about 2 years in patients with New York Heart Association class II-IV heart failure and an LVEF greater than 40% who received empagliflozin along with other standard care.
Interestingly, the drug’s expanded indication in HF resembles that approved for sacubitril/valsartan (Entresto) in February 2021 based mostly on the PARAGON-HF trial, which entered patients with HF and an LVEF at least 45%. The trial was “negative” in that it saw no significant advantage to the drug for its primary clinical outcome but did suggest benefit for some secondary endpoints.
The FDA had used more cautionary language in its expanded indication for sacubitril/valsartan, “to reduce the risk of cardiovascular death and hospitalization for heart failure in adult patients with chronic heart failure. Benefits are most clearly evident in patients with left ventricular ejection fraction below normal.”
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved an expanded heart failure indication for the sodium-glucose transporter 2 inhibitor empagliflozin (Jardiance) that now includes HF with mid-range or preserved left ventricular ejection fraction (LVEF), the agency announced on Feb. 24.
That means the SGLT2 inhibitor, once considered primarily an antidiabetic agent, is approved for use in patients with HF per se without regard to ventricular function. The drug received approval for HF with reduced LVEF in August 2021.
The expanded indication, specifically for reducing the risk of cardiovascular death and HF hospitalization in adults, was widely anticipated based on the landmark results from the EMPEROR-Preserved trial. The study saw a significant 21% relative reduction in that composite endpoint over about 2 years in patients with New York Heart Association class II-IV heart failure and an LVEF greater than 40% who received empagliflozin along with other standard care.
Interestingly, the drug’s expanded indication in HF resembles that approved for sacubitril/valsartan (Entresto) in February 2021 based mostly on the PARAGON-HF trial, which entered patients with HF and an LVEF at least 45%. The trial was “negative” in that it saw no significant advantage to the drug for its primary clinical outcome but did suggest benefit for some secondary endpoints.
The FDA had used more cautionary language in its expanded indication for sacubitril/valsartan, “to reduce the risk of cardiovascular death and hospitalization for heart failure in adult patients with chronic heart failure. Benefits are most clearly evident in patients with left ventricular ejection fraction below normal.”
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved an expanded heart failure indication for the sodium-glucose transporter 2 inhibitor empagliflozin (Jardiance) that now includes HF with mid-range or preserved left ventricular ejection fraction (LVEF), the agency announced on Feb. 24.
That means the SGLT2 inhibitor, once considered primarily an antidiabetic agent, is approved for use in patients with HF per se without regard to ventricular function. The drug received approval for HF with reduced LVEF in August 2021.
The expanded indication, specifically for reducing the risk of cardiovascular death and HF hospitalization in adults, was widely anticipated based on the landmark results from the EMPEROR-Preserved trial. The study saw a significant 21% relative reduction in that composite endpoint over about 2 years in patients with New York Heart Association class II-IV heart failure and an LVEF greater than 40% who received empagliflozin along with other standard care.
Interestingly, the drug’s expanded indication in HF resembles that approved for sacubitril/valsartan (Entresto) in February 2021 based mostly on the PARAGON-HF trial, which entered patients with HF and an LVEF at least 45%. The trial was “negative” in that it saw no significant advantage to the drug for its primary clinical outcome but did suggest benefit for some secondary endpoints.
The FDA had used more cautionary language in its expanded indication for sacubitril/valsartan, “to reduce the risk of cardiovascular death and hospitalization for heart failure in adult patients with chronic heart failure. Benefits are most clearly evident in patients with left ventricular ejection fraction below normal.”
A version of this article first appeared on Medscape.com.
Next-generation Dexcom CGM, G7, accurate and easier to use
The Dexcom G7 continuous glucose monitor (CGM) is as accurate or better than other currently marketed CGM devices for measuring glucose in those with diabetes, new data from a pivotal study suggest.
Currently under review by the U.S. Food and Drug Administration, the G7 is expected to be an improvement over the Dexcom G6 version in several ways.
The on-body size will be 60% smaller, “roughly the size of three stacked quarters,” according to the authors, and will incorporate the sensor with a single-use transmitter, as opposed to the current separate 3-month transmitter used with the G6 sensor. This will eliminate the need for using a transmitter across multiple sensor sessions (as is the case for G6).
The warm-up period after insertion is reduced from 2 hours to 27 minutes, and users are given an extra 12-hour “grace period” after the 10-day wear period to change the device before it stops displaying glucose data. Up to 24 hours of missed data can also be recaptured.
“The enhanced features of G7 may increase clinical adoption, encourage sustained use, and reduce the burden of diabetes management,” write Satish K. Garg, MD, of the University of Colorado, Aurora, and colleagues, in their article, published online Feb. 14 in Diabetes Technology & Therapeutics.
Several features of the G6 remain unchanged, including factory calibration, but also the capacity for optional user calibrations, use of Bluetooth to transmit data up to 20 feet (approximately 6 meters), and data displays on either a dedicated receiver or a variety of iOS and Android smart devices.
It will also allow for user-customized settings and alerts, as well as the option for real-time remote “share” monitoring with caregivers or providers. The G7 will also not be susceptible to interference by acetaminophen (paracetamol) or ascorbic acid.
And, like the G6, the G7 was submitted for approval to the FDA as an “integrated CGM,” meaning that it will be interoperable with other compatible devices, including insulin pumps, glucose meters, or other electronic devices used for diabetes management.
Accuracy shown on abdomen, arm
The prospective, multicenter, single-arm study reported by Dr. Garg and colleagues was conducted at 12 U.S. sites between February and June 2021.
In-clinic visits were conducted on days 1 or 2, 4 or 7, and one additional day for comparisons with a reference glucose measure (YSI 2300 Stat Plus glucose analyzer). Participants wore blinded G7 sensors concurrently on the upper arm and abdomen while continuing to use their own personal glucose monitoring method (CGM or fingerstick) at home.
A total of 316 insulin-using adults with type 1 or type 2 diabetes contributed data from 308 arm- and 311 abdomen-placed blinded devices, which generated 77,774 matched pairs of data within the blood glucose reportable range of 40-400 mg/dL.
The overall mean absolute relative difference (MARD) of each CGM-YSI pair (a standard metric for CGM analysis) was 8.2%, with 9.1% for the abdomen and 8.2% for the arm.
Accuracy remained high in both arm- and abdomen-placed sensors across the 10-day wear period through the 12-hour grace period and across glucose ranges. There were no significant differences between G7 accuracy by diabetes type, insulin regimen, or body mass index.
The highest agreement rates and lowest MARDs occurred when CGM readings were increasing or decreasing by no more than 1 mg/dL per minute. However, even at the highest rates of glucose concentration change, MARD values below 10% were found for arm-placed sensors and below 10.5% for abdomen-placed sensors, Dr. Garg and colleagues report.
When the hypoglycemia threshold alert was set to 55 mg/dL, true alert rates for detection of hypoglycemia below 70 mg/dL by sensors worn on the arm and abdomen were 91.3% and 85.2%, respectively. With hyperglycemia threshold alerts set to 300 mg/dL, the true alert rates for detection of hyperglycemia greater than 250 mg/dL by sensors worn on the arm and abdomen were 99.9% and 99.8% respectively.
The overall mean time lag for the sensors was 3.5 minutes, 3.6 minutes for the arm, and 3.4 minutes for the abdomen. There were no serious adverse events during the study.
The study excluded children and adolescents; data from these populations will be reported separately, the authors note.
Accuracy at least as good as prior Dexcom versions, competitors
The MARD values of 8.2% on the arm and 9.1% on the abdomen were similar to or better than accuracy measurements of other commercially available CGM systems, note Dr. Garg and colleagues, although they acknowledge that few head-to-head studies at different anatomic locations have been conducted.
A study of an older Dexcom version (G4 Platinum) found MARD estimates of 12.0% on the arm and 12.3% on the abdomen, the authors note.
The newly FDA-approved implantable Eversense E3 (Senseonics) CGM, worn on the upper arm, has a MARD of 9.1%, while the arm-placed Abbott FreeStyle Libre 2, approved in the U.S. in June 2020, has an overall MARD of 9.3%.
Lag-time differences between the reference and G7 were also similar to or better than time delays in prior-generation Dexcom CGMs, Dr. Garg and colleagues say.
Participants also completed a survey. “The redesigned applicator allows for sensor deployment with one hand, and most participants found G7 easier to insert than their prior CGM system,” the researchers say.
Finally, “unlike G6, G7 allows for temporary silencing of all audible alerts, including Urgent Low. Taken together, these attributes are anticipated to provide for a better end-user experience with G7 and help reduce diabetes burden,” they conclude.
The study was supported by a grant from Dexcom. Dr. Garg has reported receiving consultant fees from Medtronic, Novo Nordisk, Zealand, LifeScan, Roche, and Lilly, as well as research grants through the University of Colorado from Lilly, Novo Nordisk, Medtronic, Dexcom, T1D Exchange, Helmsley Trust, NIDDK, and JDRF.
A version of this article first appeared on Medscape.com.
The Dexcom G7 continuous glucose monitor (CGM) is as accurate or better than other currently marketed CGM devices for measuring glucose in those with diabetes, new data from a pivotal study suggest.
Currently under review by the U.S. Food and Drug Administration, the G7 is expected to be an improvement over the Dexcom G6 version in several ways.
The on-body size will be 60% smaller, “roughly the size of three stacked quarters,” according to the authors, and will incorporate the sensor with a single-use transmitter, as opposed to the current separate 3-month transmitter used with the G6 sensor. This will eliminate the need for using a transmitter across multiple sensor sessions (as is the case for G6).
The warm-up period after insertion is reduced from 2 hours to 27 minutes, and users are given an extra 12-hour “grace period” after the 10-day wear period to change the device before it stops displaying glucose data. Up to 24 hours of missed data can also be recaptured.
“The enhanced features of G7 may increase clinical adoption, encourage sustained use, and reduce the burden of diabetes management,” write Satish K. Garg, MD, of the University of Colorado, Aurora, and colleagues, in their article, published online Feb. 14 in Diabetes Technology & Therapeutics.
Several features of the G6 remain unchanged, including factory calibration, but also the capacity for optional user calibrations, use of Bluetooth to transmit data up to 20 feet (approximately 6 meters), and data displays on either a dedicated receiver or a variety of iOS and Android smart devices.
It will also allow for user-customized settings and alerts, as well as the option for real-time remote “share” monitoring with caregivers or providers. The G7 will also not be susceptible to interference by acetaminophen (paracetamol) or ascorbic acid.
And, like the G6, the G7 was submitted for approval to the FDA as an “integrated CGM,” meaning that it will be interoperable with other compatible devices, including insulin pumps, glucose meters, or other electronic devices used for diabetes management.
Accuracy shown on abdomen, arm
The prospective, multicenter, single-arm study reported by Dr. Garg and colleagues was conducted at 12 U.S. sites between February and June 2021.
In-clinic visits were conducted on days 1 or 2, 4 or 7, and one additional day for comparisons with a reference glucose measure (YSI 2300 Stat Plus glucose analyzer). Participants wore blinded G7 sensors concurrently on the upper arm and abdomen while continuing to use their own personal glucose monitoring method (CGM or fingerstick) at home.
A total of 316 insulin-using adults with type 1 or type 2 diabetes contributed data from 308 arm- and 311 abdomen-placed blinded devices, which generated 77,774 matched pairs of data within the blood glucose reportable range of 40-400 mg/dL.
The overall mean absolute relative difference (MARD) of each CGM-YSI pair (a standard metric for CGM analysis) was 8.2%, with 9.1% for the abdomen and 8.2% for the arm.
Accuracy remained high in both arm- and abdomen-placed sensors across the 10-day wear period through the 12-hour grace period and across glucose ranges. There were no significant differences between G7 accuracy by diabetes type, insulin regimen, or body mass index.
The highest agreement rates and lowest MARDs occurred when CGM readings were increasing or decreasing by no more than 1 mg/dL per minute. However, even at the highest rates of glucose concentration change, MARD values below 10% were found for arm-placed sensors and below 10.5% for abdomen-placed sensors, Dr. Garg and colleagues report.
When the hypoglycemia threshold alert was set to 55 mg/dL, true alert rates for detection of hypoglycemia below 70 mg/dL by sensors worn on the arm and abdomen were 91.3% and 85.2%, respectively. With hyperglycemia threshold alerts set to 300 mg/dL, the true alert rates for detection of hyperglycemia greater than 250 mg/dL by sensors worn on the arm and abdomen were 99.9% and 99.8% respectively.
The overall mean time lag for the sensors was 3.5 minutes, 3.6 minutes for the arm, and 3.4 minutes for the abdomen. There were no serious adverse events during the study.
The study excluded children and adolescents; data from these populations will be reported separately, the authors note.
Accuracy at least as good as prior Dexcom versions, competitors
The MARD values of 8.2% on the arm and 9.1% on the abdomen were similar to or better than accuracy measurements of other commercially available CGM systems, note Dr. Garg and colleagues, although they acknowledge that few head-to-head studies at different anatomic locations have been conducted.
A study of an older Dexcom version (G4 Platinum) found MARD estimates of 12.0% on the arm and 12.3% on the abdomen, the authors note.
The newly FDA-approved implantable Eversense E3 (Senseonics) CGM, worn on the upper arm, has a MARD of 9.1%, while the arm-placed Abbott FreeStyle Libre 2, approved in the U.S. in June 2020, has an overall MARD of 9.3%.
Lag-time differences between the reference and G7 were also similar to or better than time delays in prior-generation Dexcom CGMs, Dr. Garg and colleagues say.
Participants also completed a survey. “The redesigned applicator allows for sensor deployment with one hand, and most participants found G7 easier to insert than their prior CGM system,” the researchers say.
Finally, “unlike G6, G7 allows for temporary silencing of all audible alerts, including Urgent Low. Taken together, these attributes are anticipated to provide for a better end-user experience with G7 and help reduce diabetes burden,” they conclude.
The study was supported by a grant from Dexcom. Dr. Garg has reported receiving consultant fees from Medtronic, Novo Nordisk, Zealand, LifeScan, Roche, and Lilly, as well as research grants through the University of Colorado from Lilly, Novo Nordisk, Medtronic, Dexcom, T1D Exchange, Helmsley Trust, NIDDK, and JDRF.
A version of this article first appeared on Medscape.com.
The Dexcom G7 continuous glucose monitor (CGM) is as accurate or better than other currently marketed CGM devices for measuring glucose in those with diabetes, new data from a pivotal study suggest.
Currently under review by the U.S. Food and Drug Administration, the G7 is expected to be an improvement over the Dexcom G6 version in several ways.
The on-body size will be 60% smaller, “roughly the size of three stacked quarters,” according to the authors, and will incorporate the sensor with a single-use transmitter, as opposed to the current separate 3-month transmitter used with the G6 sensor. This will eliminate the need for using a transmitter across multiple sensor sessions (as is the case for G6).
The warm-up period after insertion is reduced from 2 hours to 27 minutes, and users are given an extra 12-hour “grace period” after the 10-day wear period to change the device before it stops displaying glucose data. Up to 24 hours of missed data can also be recaptured.
“The enhanced features of G7 may increase clinical adoption, encourage sustained use, and reduce the burden of diabetes management,” write Satish K. Garg, MD, of the University of Colorado, Aurora, and colleagues, in their article, published online Feb. 14 in Diabetes Technology & Therapeutics.
Several features of the G6 remain unchanged, including factory calibration, but also the capacity for optional user calibrations, use of Bluetooth to transmit data up to 20 feet (approximately 6 meters), and data displays on either a dedicated receiver or a variety of iOS and Android smart devices.
It will also allow for user-customized settings and alerts, as well as the option for real-time remote “share” monitoring with caregivers or providers. The G7 will also not be susceptible to interference by acetaminophen (paracetamol) or ascorbic acid.
And, like the G6, the G7 was submitted for approval to the FDA as an “integrated CGM,” meaning that it will be interoperable with other compatible devices, including insulin pumps, glucose meters, or other electronic devices used for diabetes management.
Accuracy shown on abdomen, arm
The prospective, multicenter, single-arm study reported by Dr. Garg and colleagues was conducted at 12 U.S. sites between February and June 2021.
In-clinic visits were conducted on days 1 or 2, 4 or 7, and one additional day for comparisons with a reference glucose measure (YSI 2300 Stat Plus glucose analyzer). Participants wore blinded G7 sensors concurrently on the upper arm and abdomen while continuing to use their own personal glucose monitoring method (CGM or fingerstick) at home.
A total of 316 insulin-using adults with type 1 or type 2 diabetes contributed data from 308 arm- and 311 abdomen-placed blinded devices, which generated 77,774 matched pairs of data within the blood glucose reportable range of 40-400 mg/dL.
The overall mean absolute relative difference (MARD) of each CGM-YSI pair (a standard metric for CGM analysis) was 8.2%, with 9.1% for the abdomen and 8.2% for the arm.
Accuracy remained high in both arm- and abdomen-placed sensors across the 10-day wear period through the 12-hour grace period and across glucose ranges. There were no significant differences between G7 accuracy by diabetes type, insulin regimen, or body mass index.
The highest agreement rates and lowest MARDs occurred when CGM readings were increasing or decreasing by no more than 1 mg/dL per minute. However, even at the highest rates of glucose concentration change, MARD values below 10% were found for arm-placed sensors and below 10.5% for abdomen-placed sensors, Dr. Garg and colleagues report.
When the hypoglycemia threshold alert was set to 55 mg/dL, true alert rates for detection of hypoglycemia below 70 mg/dL by sensors worn on the arm and abdomen were 91.3% and 85.2%, respectively. With hyperglycemia threshold alerts set to 300 mg/dL, the true alert rates for detection of hyperglycemia greater than 250 mg/dL by sensors worn on the arm and abdomen were 99.9% and 99.8% respectively.
The overall mean time lag for the sensors was 3.5 minutes, 3.6 minutes for the arm, and 3.4 minutes for the abdomen. There were no serious adverse events during the study.
The study excluded children and adolescents; data from these populations will be reported separately, the authors note.
Accuracy at least as good as prior Dexcom versions, competitors
The MARD values of 8.2% on the arm and 9.1% on the abdomen were similar to or better than accuracy measurements of other commercially available CGM systems, note Dr. Garg and colleagues, although they acknowledge that few head-to-head studies at different anatomic locations have been conducted.
A study of an older Dexcom version (G4 Platinum) found MARD estimates of 12.0% on the arm and 12.3% on the abdomen, the authors note.
The newly FDA-approved implantable Eversense E3 (Senseonics) CGM, worn on the upper arm, has a MARD of 9.1%, while the arm-placed Abbott FreeStyle Libre 2, approved in the U.S. in June 2020, has an overall MARD of 9.3%.
Lag-time differences between the reference and G7 were also similar to or better than time delays in prior-generation Dexcom CGMs, Dr. Garg and colleagues say.
Participants also completed a survey. “The redesigned applicator allows for sensor deployment with one hand, and most participants found G7 easier to insert than their prior CGM system,” the researchers say.
Finally, “unlike G6, G7 allows for temporary silencing of all audible alerts, including Urgent Low. Taken together, these attributes are anticipated to provide for a better end-user experience with G7 and help reduce diabetes burden,” they conclude.
The study was supported by a grant from Dexcom. Dr. Garg has reported receiving consultant fees from Medtronic, Novo Nordisk, Zealand, LifeScan, Roche, and Lilly, as well as research grants through the University of Colorado from Lilly, Novo Nordisk, Medtronic, Dexcom, T1D Exchange, Helmsley Trust, NIDDK, and JDRF.
A version of this article first appeared on Medscape.com.





