User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Infusion centers may best EDs for treating sickle cell crises
At infusion centers, patients received pain medication an average of 70 minutes faster compared with patients treated in EDs (62 vs. 132 minutes), according to a study published online in the Annals of Internal Medicine. In addition, patients at infusion centers were 3.8 times more likely to have their pain reassessed within 30 minutes of the first dose. And they were 4 times more likely to be discharged home, the researchers found.
“It’s not that the emergency room doctors don’t want to do the right thing,” study author Sophie Lanzkron, MD, said in an interview. “They do, but they aren’t experts in sickle cell disease. They work in an emergency room, which is an incredibly busy, stressful place where they see trauma and heart attacks and strokes and all of these things that need emergency care. And so it just is not the right setting to treat people with sickle cell disease.”
To assess whether care at specialty infusion centers or EDs leads to better outcomes for patients with sickle cell disease with uncomplicated vaso-occlusive crises, Dr. Lanzkron, director of the Sickle Cell Center for Adults at the Johns Hopkins Hospital, Baltimore, and colleagues conducted the ESCAPED (Examining Sickle Cell Acute Pain in the Emergency vs. Day Hospital) study.
The trial included 483 adults with sickle cell disease who lived within 60 miles of an infusion center in four U.S. cities: Baltimore, Maryland; Cleveland, Ohio; Milwaukee, Wisconsin; and Baton Rouge, Louisiana. Investigators recruited patients between April 2015 and December 2016 and followed them for 18 months after enrollment.
The present analysis focused on data from 269 participants who had infusion center visits or ED visits that occurred during weekdays when infusion centers were open. Two sites had infusion centers solely for adults with sickle cell disease (Baltimore and Milwaukee), and two infusion centers shared infusion space with other hematology-oncology patients. All four sites were in hospitals that also had EDs.
Although participants may have received comprehensive care at one of the sites with an infusion center, those who lived farther from an infusion center were likely to receive care for acute pain at an ED closer to home, the authors explain in the article.
The investigators used propensity score methodology to balance patient characteristics in the study groups.
Quick, effective pain reduction is beneficial
The results suggest that infusion centers “are more likely to provide guideline-based care than EDs,” and this care “can improve overall outcomes,” the authors write.
Although the specialty infusion centers the researchers studied used various models, similar outcomes were seen at all of them.
The study did not include patients who had complications of sickle cell disease in addition to vaso-occlusive crisis, the researchers note.
“[Because] the magnitude of the treatment effects estimated in our study is large and we have captured most of the important potential confounders, an unmeasured confounder that can nullify the treatment effect is unlikely to exist,” the authors write.
“Sickle cell disease is a complicated condition that affects multiple organs. Patients who present with acute pain will have better outcomes being treated under providers who know and understand the disease,” commented Julie Kanter, MD, director of the adult sickle cell disease program and codirector of the Comprehensive Sickle Cell Center at the University of Alabama at Birmingham. “Specialized infusion centers offer the opportunity to both improve outcomes and decrease the cost of care. Most importantly, it is better for the individual with sickle cell disease,” she said.
Dr. Kanter wrote an accompanying editorial about the ESCAPED findings. The editorialist notes that “opioid medications are the only option to reduce the pain caused by microvascular injury” in patients with sickle cell crisis, although these treatments do not reduce the underlying damage and have substantial side effects and risks. Nevertheless, “quick and effective reduction of pain can allow patients to more easily move, stretch, and breathe ... important to increase oxygenation and restore blood flow, which will eventually abate the crisis,” Dr. Kanter wrote.
The study shows that the infusion center treatment approach can benefit patients across different settings, commented John J. Strouse, MD, PhD, medical director of the adult sickle cell program at Duke University Sickle Cell Center, Durham, N.C., who was not involved in the study.
“They show that they can definitely get closer to the recommendations of guidelines for acute pain management and sickle cell disease” in a setting that is focused on one problem, he said. “The other piece that is really important is that people are much more likely to go home if you follow the guideline.”
Infusion centers are scarce
“These systems need to be built,” Dr. Lanzkron said. “In most places, patients don’t have access to the infusion center model for their care. And in some places, it is not going to be practical.” Still, there may be ways to establish infusion locations, such as at oncology centers. And while there are challenges to delivering sickle cell disease care in EDs, “emergency rooms need to try to meet the needs of this patient population as best as they can,” Dr. Lanzkron said.
“Structural racism has played a role in the quality of care delivered” to patients with sickle cell disease, Dr. Lanzkron said. “The big message is [that] there is a better way to do this.”
The study was funded by the Patient-Centered Outcomes Research Institute. Dr. Lanzkron’s disclosures included grants or contracts with government agencies and companies that are paid to her institution, as well as consulting fees from Bluebird Bio, Novo Nordisk, and Pfizer. Coauthors have disclosed working with sickle cell organizations and various medical companies. Dr. Kanter and Dr. Strouse have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
At infusion centers, patients received pain medication an average of 70 minutes faster compared with patients treated in EDs (62 vs. 132 minutes), according to a study published online in the Annals of Internal Medicine. In addition, patients at infusion centers were 3.8 times more likely to have their pain reassessed within 30 minutes of the first dose. And they were 4 times more likely to be discharged home, the researchers found.
“It’s not that the emergency room doctors don’t want to do the right thing,” study author Sophie Lanzkron, MD, said in an interview. “They do, but they aren’t experts in sickle cell disease. They work in an emergency room, which is an incredibly busy, stressful place where they see trauma and heart attacks and strokes and all of these things that need emergency care. And so it just is not the right setting to treat people with sickle cell disease.”
To assess whether care at specialty infusion centers or EDs leads to better outcomes for patients with sickle cell disease with uncomplicated vaso-occlusive crises, Dr. Lanzkron, director of the Sickle Cell Center for Adults at the Johns Hopkins Hospital, Baltimore, and colleagues conducted the ESCAPED (Examining Sickle Cell Acute Pain in the Emergency vs. Day Hospital) study.
The trial included 483 adults with sickle cell disease who lived within 60 miles of an infusion center in four U.S. cities: Baltimore, Maryland; Cleveland, Ohio; Milwaukee, Wisconsin; and Baton Rouge, Louisiana. Investigators recruited patients between April 2015 and December 2016 and followed them for 18 months after enrollment.
The present analysis focused on data from 269 participants who had infusion center visits or ED visits that occurred during weekdays when infusion centers were open. Two sites had infusion centers solely for adults with sickle cell disease (Baltimore and Milwaukee), and two infusion centers shared infusion space with other hematology-oncology patients. All four sites were in hospitals that also had EDs.
Although participants may have received comprehensive care at one of the sites with an infusion center, those who lived farther from an infusion center were likely to receive care for acute pain at an ED closer to home, the authors explain in the article.
The investigators used propensity score methodology to balance patient characteristics in the study groups.
Quick, effective pain reduction is beneficial
The results suggest that infusion centers “are more likely to provide guideline-based care than EDs,” and this care “can improve overall outcomes,” the authors write.
Although the specialty infusion centers the researchers studied used various models, similar outcomes were seen at all of them.
The study did not include patients who had complications of sickle cell disease in addition to vaso-occlusive crisis, the researchers note.
“[Because] the magnitude of the treatment effects estimated in our study is large and we have captured most of the important potential confounders, an unmeasured confounder that can nullify the treatment effect is unlikely to exist,” the authors write.
“Sickle cell disease is a complicated condition that affects multiple organs. Patients who present with acute pain will have better outcomes being treated under providers who know and understand the disease,” commented Julie Kanter, MD, director of the adult sickle cell disease program and codirector of the Comprehensive Sickle Cell Center at the University of Alabama at Birmingham. “Specialized infusion centers offer the opportunity to both improve outcomes and decrease the cost of care. Most importantly, it is better for the individual with sickle cell disease,” she said.
Dr. Kanter wrote an accompanying editorial about the ESCAPED findings. The editorialist notes that “opioid medications are the only option to reduce the pain caused by microvascular injury” in patients with sickle cell crisis, although these treatments do not reduce the underlying damage and have substantial side effects and risks. Nevertheless, “quick and effective reduction of pain can allow patients to more easily move, stretch, and breathe ... important to increase oxygenation and restore blood flow, which will eventually abate the crisis,” Dr. Kanter wrote.
The study shows that the infusion center treatment approach can benefit patients across different settings, commented John J. Strouse, MD, PhD, medical director of the adult sickle cell program at Duke University Sickle Cell Center, Durham, N.C., who was not involved in the study.
“They show that they can definitely get closer to the recommendations of guidelines for acute pain management and sickle cell disease” in a setting that is focused on one problem, he said. “The other piece that is really important is that people are much more likely to go home if you follow the guideline.”
Infusion centers are scarce
“These systems need to be built,” Dr. Lanzkron said. “In most places, patients don’t have access to the infusion center model for their care. And in some places, it is not going to be practical.” Still, there may be ways to establish infusion locations, such as at oncology centers. And while there are challenges to delivering sickle cell disease care in EDs, “emergency rooms need to try to meet the needs of this patient population as best as they can,” Dr. Lanzkron said.
“Structural racism has played a role in the quality of care delivered” to patients with sickle cell disease, Dr. Lanzkron said. “The big message is [that] there is a better way to do this.”
The study was funded by the Patient-Centered Outcomes Research Institute. Dr. Lanzkron’s disclosures included grants or contracts with government agencies and companies that are paid to her institution, as well as consulting fees from Bluebird Bio, Novo Nordisk, and Pfizer. Coauthors have disclosed working with sickle cell organizations and various medical companies. Dr. Kanter and Dr. Strouse have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
At infusion centers, patients received pain medication an average of 70 minutes faster compared with patients treated in EDs (62 vs. 132 minutes), according to a study published online in the Annals of Internal Medicine. In addition, patients at infusion centers were 3.8 times more likely to have their pain reassessed within 30 minutes of the first dose. And they were 4 times more likely to be discharged home, the researchers found.
“It’s not that the emergency room doctors don’t want to do the right thing,” study author Sophie Lanzkron, MD, said in an interview. “They do, but they aren’t experts in sickle cell disease. They work in an emergency room, which is an incredibly busy, stressful place where they see trauma and heart attacks and strokes and all of these things that need emergency care. And so it just is not the right setting to treat people with sickle cell disease.”
To assess whether care at specialty infusion centers or EDs leads to better outcomes for patients with sickle cell disease with uncomplicated vaso-occlusive crises, Dr. Lanzkron, director of the Sickle Cell Center for Adults at the Johns Hopkins Hospital, Baltimore, and colleagues conducted the ESCAPED (Examining Sickle Cell Acute Pain in the Emergency vs. Day Hospital) study.
The trial included 483 adults with sickle cell disease who lived within 60 miles of an infusion center in four U.S. cities: Baltimore, Maryland; Cleveland, Ohio; Milwaukee, Wisconsin; and Baton Rouge, Louisiana. Investigators recruited patients between April 2015 and December 2016 and followed them for 18 months after enrollment.
The present analysis focused on data from 269 participants who had infusion center visits or ED visits that occurred during weekdays when infusion centers were open. Two sites had infusion centers solely for adults with sickle cell disease (Baltimore and Milwaukee), and two infusion centers shared infusion space with other hematology-oncology patients. All four sites were in hospitals that also had EDs.
Although participants may have received comprehensive care at one of the sites with an infusion center, those who lived farther from an infusion center were likely to receive care for acute pain at an ED closer to home, the authors explain in the article.
The investigators used propensity score methodology to balance patient characteristics in the study groups.
Quick, effective pain reduction is beneficial
The results suggest that infusion centers “are more likely to provide guideline-based care than EDs,” and this care “can improve overall outcomes,” the authors write.
Although the specialty infusion centers the researchers studied used various models, similar outcomes were seen at all of them.
The study did not include patients who had complications of sickle cell disease in addition to vaso-occlusive crisis, the researchers note.
“[Because] the magnitude of the treatment effects estimated in our study is large and we have captured most of the important potential confounders, an unmeasured confounder that can nullify the treatment effect is unlikely to exist,” the authors write.
“Sickle cell disease is a complicated condition that affects multiple organs. Patients who present with acute pain will have better outcomes being treated under providers who know and understand the disease,” commented Julie Kanter, MD, director of the adult sickle cell disease program and codirector of the Comprehensive Sickle Cell Center at the University of Alabama at Birmingham. “Specialized infusion centers offer the opportunity to both improve outcomes and decrease the cost of care. Most importantly, it is better for the individual with sickle cell disease,” she said.
Dr. Kanter wrote an accompanying editorial about the ESCAPED findings. The editorialist notes that “opioid medications are the only option to reduce the pain caused by microvascular injury” in patients with sickle cell crisis, although these treatments do not reduce the underlying damage and have substantial side effects and risks. Nevertheless, “quick and effective reduction of pain can allow patients to more easily move, stretch, and breathe ... important to increase oxygenation and restore blood flow, which will eventually abate the crisis,” Dr. Kanter wrote.
The study shows that the infusion center treatment approach can benefit patients across different settings, commented John J. Strouse, MD, PhD, medical director of the adult sickle cell program at Duke University Sickle Cell Center, Durham, N.C., who was not involved in the study.
“They show that they can definitely get closer to the recommendations of guidelines for acute pain management and sickle cell disease” in a setting that is focused on one problem, he said. “The other piece that is really important is that people are much more likely to go home if you follow the guideline.”
Infusion centers are scarce
“These systems need to be built,” Dr. Lanzkron said. “In most places, patients don’t have access to the infusion center model for their care. And in some places, it is not going to be practical.” Still, there may be ways to establish infusion locations, such as at oncology centers. And while there are challenges to delivering sickle cell disease care in EDs, “emergency rooms need to try to meet the needs of this patient population as best as they can,” Dr. Lanzkron said.
“Structural racism has played a role in the quality of care delivered” to patients with sickle cell disease, Dr. Lanzkron said. “The big message is [that] there is a better way to do this.”
The study was funded by the Patient-Centered Outcomes Research Institute. Dr. Lanzkron’s disclosures included grants or contracts with government agencies and companies that are paid to her institution, as well as consulting fees from Bluebird Bio, Novo Nordisk, and Pfizer. Coauthors have disclosed working with sickle cell organizations and various medical companies. Dr. Kanter and Dr. Strouse have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Female doctors of color say they feel pressure to change their look
It started when a Latina doctor tweeted that she lost points on a practical exam in medical school because of her hoop earrings, with the evaluator writing “earrings, unprofessional.”
That led other female doctors to cite their own experiences, reported The Lily, a Washington Post publication aimed at millennial women. Many women posted photos of themselves wearing hoops, which have long been associated with Latina and African American women, the outlet said.
“There’s a big movement to police women of color and how they present themselves in medical spaces,” said Briana Christophers, an MD-PhD student at the Tri-Institutional MD-PhD Program in New York. “I think in part it’s a way of trying to make people who don’t usually fit the mold, fit the mold.”
Ms. Christophers, who identifies as Latina, said she was urged to wear a black or navy suit when interviewing for doctorate programs. She wore a black suit with a lavender blouse and received comments about that – some positive, some not, she said.
“Sometimes you don’t know how to interpret those sorts of comments,” Ms. Christophers said. “Do you remember because you like the shirt, or because you don’t think I should have done that?”
Doctors of color still stand out in American medicine. The Lily cited the Association of American Medical Colleges as saying that in 2018, Hispanics made up 5.8% of active American doctors and African Americans made up 5%.
Studies show that medical professionals of color often don’t receive the same respect as their White counterparts, with some people questioning whether they’re actually doctors.
“At work, wearing my white coat that has my name pretty big on it with a badge that says doctor on it, I still get asked if I’m the environmental services staff,” Alexandra Sims, MD, a pediatrician in Cincinnati, told The Lily. “I think it just demonstrates how deeply ingrained bias, racism, and sexism are in society and that we have a lot of work to do to disrupt that.”
Dr. Sims said the tweet about hoop earrings led her to wonder about daily decisions she makes about dress.
“Am I too much? Is this too much? Is this earring too big? Is this nail polish color too loud? And how will that be received at work?” she said, noting that she may opt not to wear hoops in certain situations, such as when she’s dealing with a grabby baby.
Monica Verduzco-Gutierrez, MD, professor and chair of the department of rehabilitation medicine at University of Texas Health, San Antonio, said doctors should be judged on the care they provide, not their appearance.
“Judging someone based on their earrings or their jumpsuit or whatever else that they’re noticing about the student is not an appropriate way to judge the student’s ability to take care of a patient,” Dr. Verduzco-Gutierrez said, noting that she was not speaking on behalf of the school.
A version of this article was first published on WebMD.com .
It started when a Latina doctor tweeted that she lost points on a practical exam in medical school because of her hoop earrings, with the evaluator writing “earrings, unprofessional.”
That led other female doctors to cite their own experiences, reported The Lily, a Washington Post publication aimed at millennial women. Many women posted photos of themselves wearing hoops, which have long been associated with Latina and African American women, the outlet said.
“There’s a big movement to police women of color and how they present themselves in medical spaces,” said Briana Christophers, an MD-PhD student at the Tri-Institutional MD-PhD Program in New York. “I think in part it’s a way of trying to make people who don’t usually fit the mold, fit the mold.”
Ms. Christophers, who identifies as Latina, said she was urged to wear a black or navy suit when interviewing for doctorate programs. She wore a black suit with a lavender blouse and received comments about that – some positive, some not, she said.
“Sometimes you don’t know how to interpret those sorts of comments,” Ms. Christophers said. “Do you remember because you like the shirt, or because you don’t think I should have done that?”
Doctors of color still stand out in American medicine. The Lily cited the Association of American Medical Colleges as saying that in 2018, Hispanics made up 5.8% of active American doctors and African Americans made up 5%.
Studies show that medical professionals of color often don’t receive the same respect as their White counterparts, with some people questioning whether they’re actually doctors.
“At work, wearing my white coat that has my name pretty big on it with a badge that says doctor on it, I still get asked if I’m the environmental services staff,” Alexandra Sims, MD, a pediatrician in Cincinnati, told The Lily. “I think it just demonstrates how deeply ingrained bias, racism, and sexism are in society and that we have a lot of work to do to disrupt that.”
Dr. Sims said the tweet about hoop earrings led her to wonder about daily decisions she makes about dress.
“Am I too much? Is this too much? Is this earring too big? Is this nail polish color too loud? And how will that be received at work?” she said, noting that she may opt not to wear hoops in certain situations, such as when she’s dealing with a grabby baby.
Monica Verduzco-Gutierrez, MD, professor and chair of the department of rehabilitation medicine at University of Texas Health, San Antonio, said doctors should be judged on the care they provide, not their appearance.
“Judging someone based on their earrings or their jumpsuit or whatever else that they’re noticing about the student is not an appropriate way to judge the student’s ability to take care of a patient,” Dr. Verduzco-Gutierrez said, noting that she was not speaking on behalf of the school.
A version of this article was first published on WebMD.com .
It started when a Latina doctor tweeted that she lost points on a practical exam in medical school because of her hoop earrings, with the evaluator writing “earrings, unprofessional.”
That led other female doctors to cite their own experiences, reported The Lily, a Washington Post publication aimed at millennial women. Many women posted photos of themselves wearing hoops, which have long been associated with Latina and African American women, the outlet said.
“There’s a big movement to police women of color and how they present themselves in medical spaces,” said Briana Christophers, an MD-PhD student at the Tri-Institutional MD-PhD Program in New York. “I think in part it’s a way of trying to make people who don’t usually fit the mold, fit the mold.”
Ms. Christophers, who identifies as Latina, said she was urged to wear a black or navy suit when interviewing for doctorate programs. She wore a black suit with a lavender blouse and received comments about that – some positive, some not, she said.
“Sometimes you don’t know how to interpret those sorts of comments,” Ms. Christophers said. “Do you remember because you like the shirt, or because you don’t think I should have done that?”
Doctors of color still stand out in American medicine. The Lily cited the Association of American Medical Colleges as saying that in 2018, Hispanics made up 5.8% of active American doctors and African Americans made up 5%.
Studies show that medical professionals of color often don’t receive the same respect as their White counterparts, with some people questioning whether they’re actually doctors.
“At work, wearing my white coat that has my name pretty big on it with a badge that says doctor on it, I still get asked if I’m the environmental services staff,” Alexandra Sims, MD, a pediatrician in Cincinnati, told The Lily. “I think it just demonstrates how deeply ingrained bias, racism, and sexism are in society and that we have a lot of work to do to disrupt that.”
Dr. Sims said the tweet about hoop earrings led her to wonder about daily decisions she makes about dress.
“Am I too much? Is this too much? Is this earring too big? Is this nail polish color too loud? And how will that be received at work?” she said, noting that she may opt not to wear hoops in certain situations, such as when she’s dealing with a grabby baby.
Monica Verduzco-Gutierrez, MD, professor and chair of the department of rehabilitation medicine at University of Texas Health, San Antonio, said doctors should be judged on the care they provide, not their appearance.
“Judging someone based on their earrings or their jumpsuit or whatever else that they’re noticing about the student is not an appropriate way to judge the student’s ability to take care of a patient,” Dr. Verduzco-Gutierrez said, noting that she was not speaking on behalf of the school.
A version of this article was first published on WebMD.com .
A pacemaker that 'just disappears' and a magnetic diet device
Ignore this pacemaker and it will go away
At some point – and now seems to be that point – we have to say enough is enough. The throwaway culture that produces phones, TVs, and computers that get tossed in the trash because they can’t be repaired has gone too far. That’s right, we’re looking at you, medical science!
This time, it’s a pacemaker that just disappears when it’s no longer needed. Some lazy heart surgeon decided that it was way too much trouble to do another surgery to remove the leads when a temporary pacemaker was no longer needed. You know the type: “It sure would be nice if the pacemaker components were biocompatible and were naturally absorbed by the body over the course of a few weeks and wouldn’t need to be surgically extracted.” Slacker.
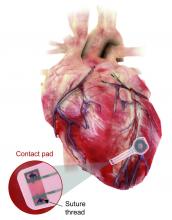
Well, get a load of this. Researchers at Northwestern and George Washington universities say that they have come up with a transient pacemaker that “harvests energy from an external, remote antenna using near-field communication protocols – the same technology used in smartphones for electronic payments and in RFID tags.”
That means no batteries and no wires that have to be removed and can cause infections. Because the infectious disease docs also are too lazy to do their jobs, apparently.
The lack of onboard infrastructure means that the device can be very small – it weighs less than half a gram and is only 250 microns thick. And yes, it is bioresorbable and completely harmless. It fully degrades and disappears in 5-7 weeks through the body’s natural biologic processes, “thereby avoiding the need for physical removal of the pacemaker electrodes. This is potentially a major victory for postoperative patients,” said Dr. Rishi Arora, one of the investigators.
A victory for patients, he says. Not a word about the time and effort saved by the surgeons. Typical.
It’s a mask! No, it’s a COVID-19 test!
Mask wearing has gotten more lax as people get vaccinated for COVID-19, but as wearing masks for virus prevention is becoming more normalized in western society, some saw an opportunity to make them work for diagnosis.
Researchers from the Massachusetts Institute of Technology and the Wyss Institute for Biologically Inspired Engineering at Harvard University have found a way to do just that with their wearable freeze-dried cell-free (wFDCF) technology. A single push of a button releases water from a reservoir in the mask that sequentially activates three different freeze-dried biological reactions, which detect the SARS-CoV-2 virus in the wearer’s breath.
Initially meant as a tool for the Zika outbreak in 2015, the team made a quick pivot in May 2020. But this isn’t just some run-of-the-mill, at-home test. The data prove that the wFDCF mask is comparable to polymerase chain reactions tests, the standard in COVID-19 detection. Plus there aren’t any extra factors to deal with, like room or instrument temperature to ensure accuracy. In just 90 minutes, the mask gives results on a readout in a way similar to that of a pregnancy test. Voilà! To have COVID-19 or not to have COVID-19 is an easily answered question.
At LOTME, we think this is a big improvement from having dogs, or even three-foot rats, sniffing out coronavirus.
But wait, there’s more. “In addition to face masks, our programmable biosensors can be integrated into other garments to provide on-the-go detection of dangerous substances including viruses, bacteria, toxins, and chemical agents,” said Peter Nguyen, PhD, study coauthor and research scientist at the Wyss Institute. The technology can be used on lab coats, scrubs, military uniforms, and uniforms of first responders who may come in contact with hazardous pathogens and toxins. Think of all the lives saved and possible avoidances.
If only it could diagnose bad breath.
Finally, an excuse for the all-beer diet
Weight loss is hard work. Extremely hard work, and, as evidenced by the constant inundation and advertisement of quick fixes, crash diets, and expensive gym memberships, there’s not really a solid, 100% solution to the issue. Until now, thanks to a team of doctors from New Zealand, who’ve decided that the best way to combat obesity is to leave you in constant agony.
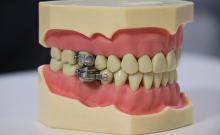
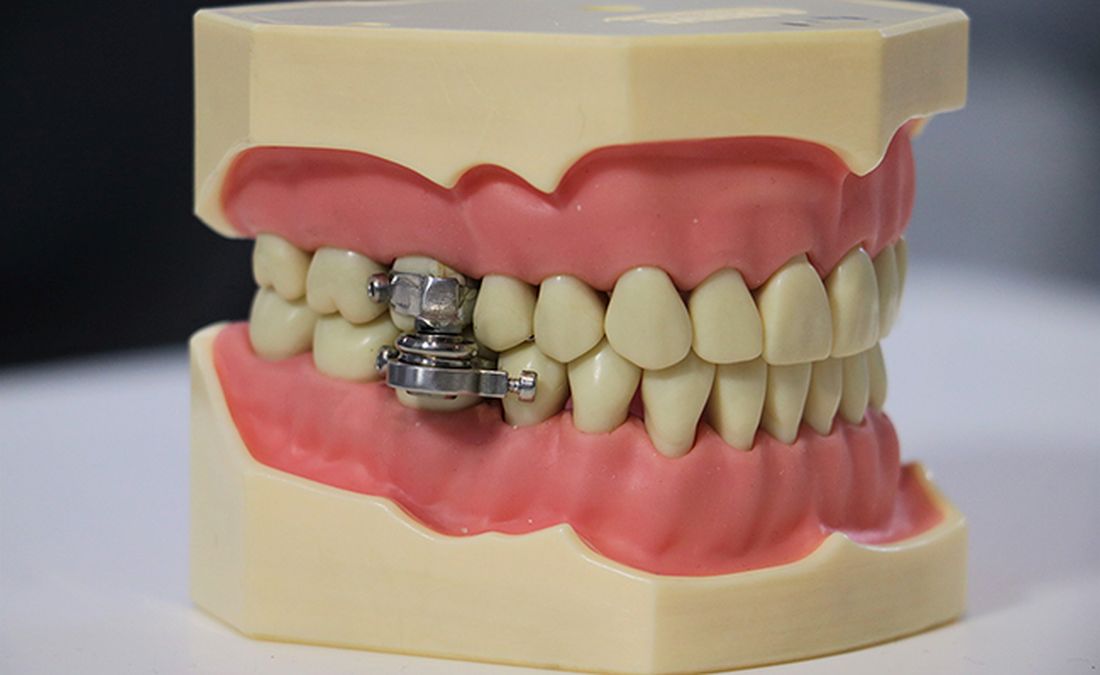
The DentalSlim Diet Control device is certainly a radical yet comically logical attempt to combat obesity. The creators say that the biggest problem with dieting is compliance, and, well, it’s difficult to eat too much if you can’t actually open your mouth. The metal contraption is mounted onto your teeth and uses magnetic locks to prevent the user from opening their mouths more than 2 mm. That’s less than a tenth of an inch. Which is not a lot. So not a lot that essentially all you can consume is liquid.
Oh, and they’ve got results to back up their madness. In a small study, seven otherwise healthy obese women lost an average of 5.1% of their body weight after using the DentalSlim for 2 weeks, though they did complain that the device was difficult to use, caused discomfort and difficulty speaking, made them more tense, and in general made life “less satisfying.” And one participant was able to cheat the system and consume nonhealthy food like chocolate by melting it.
So, there you are, if you want a weight-loss solution that tortures you and has far bigger holes than the one it leaves for your mouth, try the DentalSlim. Or, you know, don’t eat that eighth slice of pizza and maybe go for a walk later. Your choice.
Ignore this pacemaker and it will go away
At some point – and now seems to be that point – we have to say enough is enough. The throwaway culture that produces phones, TVs, and computers that get tossed in the trash because they can’t be repaired has gone too far. That’s right, we’re looking at you, medical science!
This time, it’s a pacemaker that just disappears when it’s no longer needed. Some lazy heart surgeon decided that it was way too much trouble to do another surgery to remove the leads when a temporary pacemaker was no longer needed. You know the type: “It sure would be nice if the pacemaker components were biocompatible and were naturally absorbed by the body over the course of a few weeks and wouldn’t need to be surgically extracted.” Slacker.
Well, get a load of this. Researchers at Northwestern and George Washington universities say that they have come up with a transient pacemaker that “harvests energy from an external, remote antenna using near-field communication protocols – the same technology used in smartphones for electronic payments and in RFID tags.”
That means no batteries and no wires that have to be removed and can cause infections. Because the infectious disease docs also are too lazy to do their jobs, apparently.
The lack of onboard infrastructure means that the device can be very small – it weighs less than half a gram and is only 250 microns thick. And yes, it is bioresorbable and completely harmless. It fully degrades and disappears in 5-7 weeks through the body’s natural biologic processes, “thereby avoiding the need for physical removal of the pacemaker electrodes. This is potentially a major victory for postoperative patients,” said Dr. Rishi Arora, one of the investigators.
A victory for patients, he says. Not a word about the time and effort saved by the surgeons. Typical.
It’s a mask! No, it’s a COVID-19 test!
Mask wearing has gotten more lax as people get vaccinated for COVID-19, but as wearing masks for virus prevention is becoming more normalized in western society, some saw an opportunity to make them work for diagnosis.
Researchers from the Massachusetts Institute of Technology and the Wyss Institute for Biologically Inspired Engineering at Harvard University have found a way to do just that with their wearable freeze-dried cell-free (wFDCF) technology. A single push of a button releases water from a reservoir in the mask that sequentially activates three different freeze-dried biological reactions, which detect the SARS-CoV-2 virus in the wearer’s breath.
Initially meant as a tool for the Zika outbreak in 2015, the team made a quick pivot in May 2020. But this isn’t just some run-of-the-mill, at-home test. The data prove that the wFDCF mask is comparable to polymerase chain reactions tests, the standard in COVID-19 detection. Plus there aren’t any extra factors to deal with, like room or instrument temperature to ensure accuracy. In just 90 minutes, the mask gives results on a readout in a way similar to that of a pregnancy test. Voilà! To have COVID-19 or not to have COVID-19 is an easily answered question.
At LOTME, we think this is a big improvement from having dogs, or even three-foot rats, sniffing out coronavirus.
But wait, there’s more. “In addition to face masks, our programmable biosensors can be integrated into other garments to provide on-the-go detection of dangerous substances including viruses, bacteria, toxins, and chemical agents,” said Peter Nguyen, PhD, study coauthor and research scientist at the Wyss Institute. The technology can be used on lab coats, scrubs, military uniforms, and uniforms of first responders who may come in contact with hazardous pathogens and toxins. Think of all the lives saved and possible avoidances.
If only it could diagnose bad breath.
Finally, an excuse for the all-beer diet
Weight loss is hard work. Extremely hard work, and, as evidenced by the constant inundation and advertisement of quick fixes, crash diets, and expensive gym memberships, there’s not really a solid, 100% solution to the issue. Until now, thanks to a team of doctors from New Zealand, who’ve decided that the best way to combat obesity is to leave you in constant agony.
The DentalSlim Diet Control device is certainly a radical yet comically logical attempt to combat obesity. The creators say that the biggest problem with dieting is compliance, and, well, it’s difficult to eat too much if you can’t actually open your mouth. The metal contraption is mounted onto your teeth and uses magnetic locks to prevent the user from opening their mouths more than 2 mm. That’s less than a tenth of an inch. Which is not a lot. So not a lot that essentially all you can consume is liquid.
Oh, and they’ve got results to back up their madness. In a small study, seven otherwise healthy obese women lost an average of 5.1% of their body weight after using the DentalSlim for 2 weeks, though they did complain that the device was difficult to use, caused discomfort and difficulty speaking, made them more tense, and in general made life “less satisfying.” And one participant was able to cheat the system and consume nonhealthy food like chocolate by melting it.
So, there you are, if you want a weight-loss solution that tortures you and has far bigger holes than the one it leaves for your mouth, try the DentalSlim. Or, you know, don’t eat that eighth slice of pizza and maybe go for a walk later. Your choice.
Ignore this pacemaker and it will go away
At some point – and now seems to be that point – we have to say enough is enough. The throwaway culture that produces phones, TVs, and computers that get tossed in the trash because they can’t be repaired has gone too far. That’s right, we’re looking at you, medical science!
This time, it’s a pacemaker that just disappears when it’s no longer needed. Some lazy heart surgeon decided that it was way too much trouble to do another surgery to remove the leads when a temporary pacemaker was no longer needed. You know the type: “It sure would be nice if the pacemaker components were biocompatible and were naturally absorbed by the body over the course of a few weeks and wouldn’t need to be surgically extracted.” Slacker.
Well, get a load of this. Researchers at Northwestern and George Washington universities say that they have come up with a transient pacemaker that “harvests energy from an external, remote antenna using near-field communication protocols – the same technology used in smartphones for electronic payments and in RFID tags.”
That means no batteries and no wires that have to be removed and can cause infections. Because the infectious disease docs also are too lazy to do their jobs, apparently.
The lack of onboard infrastructure means that the device can be very small – it weighs less than half a gram and is only 250 microns thick. And yes, it is bioresorbable and completely harmless. It fully degrades and disappears in 5-7 weeks through the body’s natural biologic processes, “thereby avoiding the need for physical removal of the pacemaker electrodes. This is potentially a major victory for postoperative patients,” said Dr. Rishi Arora, one of the investigators.
A victory for patients, he says. Not a word about the time and effort saved by the surgeons. Typical.
It’s a mask! No, it’s a COVID-19 test!
Mask wearing has gotten more lax as people get vaccinated for COVID-19, but as wearing masks for virus prevention is becoming more normalized in western society, some saw an opportunity to make them work for diagnosis.
Researchers from the Massachusetts Institute of Technology and the Wyss Institute for Biologically Inspired Engineering at Harvard University have found a way to do just that with their wearable freeze-dried cell-free (wFDCF) technology. A single push of a button releases water from a reservoir in the mask that sequentially activates three different freeze-dried biological reactions, which detect the SARS-CoV-2 virus in the wearer’s breath.
Initially meant as a tool for the Zika outbreak in 2015, the team made a quick pivot in May 2020. But this isn’t just some run-of-the-mill, at-home test. The data prove that the wFDCF mask is comparable to polymerase chain reactions tests, the standard in COVID-19 detection. Plus there aren’t any extra factors to deal with, like room or instrument temperature to ensure accuracy. In just 90 minutes, the mask gives results on a readout in a way similar to that of a pregnancy test. Voilà! To have COVID-19 or not to have COVID-19 is an easily answered question.
At LOTME, we think this is a big improvement from having dogs, or even three-foot rats, sniffing out coronavirus.
But wait, there’s more. “In addition to face masks, our programmable biosensors can be integrated into other garments to provide on-the-go detection of dangerous substances including viruses, bacteria, toxins, and chemical agents,” said Peter Nguyen, PhD, study coauthor and research scientist at the Wyss Institute. The technology can be used on lab coats, scrubs, military uniforms, and uniforms of first responders who may come in contact with hazardous pathogens and toxins. Think of all the lives saved and possible avoidances.
If only it could diagnose bad breath.
Finally, an excuse for the all-beer diet
Weight loss is hard work. Extremely hard work, and, as evidenced by the constant inundation and advertisement of quick fixes, crash diets, and expensive gym memberships, there’s not really a solid, 100% solution to the issue. Until now, thanks to a team of doctors from New Zealand, who’ve decided that the best way to combat obesity is to leave you in constant agony.
The DentalSlim Diet Control device is certainly a radical yet comically logical attempt to combat obesity. The creators say that the biggest problem with dieting is compliance, and, well, it’s difficult to eat too much if you can’t actually open your mouth. The metal contraption is mounted onto your teeth and uses magnetic locks to prevent the user from opening their mouths more than 2 mm. That’s less than a tenth of an inch. Which is not a lot. So not a lot that essentially all you can consume is liquid.
Oh, and they’ve got results to back up their madness. In a small study, seven otherwise healthy obese women lost an average of 5.1% of their body weight after using the DentalSlim for 2 weeks, though they did complain that the device was difficult to use, caused discomfort and difficulty speaking, made them more tense, and in general made life “less satisfying.” And one participant was able to cheat the system and consume nonhealthy food like chocolate by melting it.
So, there you are, if you want a weight-loss solution that tortures you and has far bigger holes than the one it leaves for your mouth, try the DentalSlim. Or, you know, don’t eat that eighth slice of pizza and maybe go for a walk later. Your choice.
Almost all U.S. COVID-19 deaths now in the unvaccinated
If you, a friend, or a loved one remain unvaccinated against COVID-19 at this point – for whatever reason – you are at higher risk of dying if you become infected.
That’s the conclusion of a new report released by the Associated Press looking at COVID-19 deaths during May 2021.
Of more than 18,000 people who died from COVID-19, for example, only about 150 were fully vaccinated. That’s less than 1%.
“Recently, I was working in the emergency room [and] I saw a 21-year-old African American who came in with shortness of breath,” said Vino K. Palli, MD, MPH, a physician specializing in emergency medicine, internal medicine, and urgent care.
The patient rapidly deteriorated and required intubation and ventilation. She was transferred to a specialized hospital for possible extracorporeal membrane oxygenation (ECMO) treatment.
“This patient was unvaccinated, along with her entire family. This would have been easily preventable,” added Dr. Palli, who is also founder and CEO of MiDoctor Urgent Care in New York City.
“Vaccine misinformation, compounded with vaccine inertia and vaccine access, have contributed to this,” he added. “Even though we have a surplus amount of vaccines at this time, we are only seeing 50% to 55% of completely vaccinated patients.”
Authors of the Associated Press report also acknowledge that some people who are fully vaccinated can get a breakthrough infection. These occurred in fewer than 1,200 of more than 853,000 people hospitalized for COVID-19 in May, or about 0.1%.
The Associated Press came up with these numbers using data from the Centers for Disease Control and Prevention. The CDC tracks the numbers of cases, hospitalizations, and deaths but does not breakdown rates by vaccination status.
Stronger argument for vaccination?
“The fact that only 0.8% of COVID-19 deaths are in the fully vaccinated should persuade those people still hesitant about vaccination,” said Hugh Cassiere, MD, medical director of Respiratory Therapy Services at North Shore University Hospital in Manhasset, New York.
Stuart C. Ray, MD, professor of medicine and oncology in the Division of Infectious Diseases at Johns Hopkins University, Baltimore, agreed. “It seems compelling, even for skeptics, that unvaccinated people represent 99% of those now dying from COVID-19 when they represent less than 50% of the adult population in the United States.”
The findings from the study could be more persuasive than previous arguments made in favor of immunization, Dr. Ray said. “These recent findings of striking reductions in risk of death in the vaccinated are more directly attributable and harder to ignore or dismiss.”
Brian Labus, PhD, MPH, of the University of Nevada Las Vegas (UNLV) is less convinced. “While this might change some peoples’ minds, it probably won’t make a major difference. People have many different reasons for not getting vaccinated, and this is only one of the things they consider.”
The study adds information that was not available before, said Dr. Labus, assistant professor in the Department of Epidemiology and Biostatistics at the UNLV School of Public Health. “We study the vaccine under tightly controlled, ideal conditions. This is the evidence that it works as well in the real world as it did in the trials, and that is what is most important in implementing a vaccination program,” added Dr. Labus.
“The scientific data has honed in on one thing: Vaccines are effective in preventing hospitalizations, ICU admissions, ventilations, and deaths,” agreed Dr. Palli.
“We now know that almost all deaths occurred in patients who were not vaccinated. We also know that all vaccines are effective against various strains that are in circulation right now, including the Delta variant, which is rapidly spreading,” Dr. Palli said.
Dr. Cassiere pointed out that the unvaccinated are not only at higher risk of developing COVID-19 but also of spreading, being hospitalized for, and dying from the infection. Avoiding “long hauler” symptoms is another argument in favor of immunization, he added.
As of June 28, the CDC reports that 63% of Americans 12 years and older have received at least one dose of a COVID-19 vaccine, and 54% are fully vaccinated.
Worldwide worry?
Although overall rates of U.S. COVID-19 hospitalizations and deaths are down, the outlook may not remain as encouraging. “I hope I’m wrong about this, but I anticipate that the coming fall and winter will bring increasingly localized versions of similar findings – severe disease and death due to SARS-CoV-2 infection in regions or groups with lower vaccination rates,” Dr. Ray said.
There could be a silver lining, he added: “If this unfortunate surge occurs, the health and economic consequences seem likely to erode much of the remaining hesitancy regarding vaccination.”
The rise of more infectious SARS-CoV-2 variants, such as the Delta variant, could also throw a wrench in controlling COVID-19. “This isn’t just a domestic issue,” Dr. Ray said. “We have learned that the world is a small place in pandemic times.”
The Associated Press investigators state that their findings support the high efficacy of the vaccine. Also, given the current widespread availability of COVID-19 vaccines in the United States, they believe many of the COVID-19 deaths now occurring are preventable.
Public health measures should have continued longer to protect unvaccinated individuals, especially Black Americans, Hispanic Americans, and other minorities, Dr. Palli said. “Only time will tell if re-opening and abandoning all public health measures by the CDC was premature.”
A version of this article first appeared on Medscape.com.
If you, a friend, or a loved one remain unvaccinated against COVID-19 at this point – for whatever reason – you are at higher risk of dying if you become infected.
That’s the conclusion of a new report released by the Associated Press looking at COVID-19 deaths during May 2021.
Of more than 18,000 people who died from COVID-19, for example, only about 150 were fully vaccinated. That’s less than 1%.
“Recently, I was working in the emergency room [and] I saw a 21-year-old African American who came in with shortness of breath,” said Vino K. Palli, MD, MPH, a physician specializing in emergency medicine, internal medicine, and urgent care.
The patient rapidly deteriorated and required intubation and ventilation. She was transferred to a specialized hospital for possible extracorporeal membrane oxygenation (ECMO) treatment.
“This patient was unvaccinated, along with her entire family. This would have been easily preventable,” added Dr. Palli, who is also founder and CEO of MiDoctor Urgent Care in New York City.
“Vaccine misinformation, compounded with vaccine inertia and vaccine access, have contributed to this,” he added. “Even though we have a surplus amount of vaccines at this time, we are only seeing 50% to 55% of completely vaccinated patients.”
Authors of the Associated Press report also acknowledge that some people who are fully vaccinated can get a breakthrough infection. These occurred in fewer than 1,200 of more than 853,000 people hospitalized for COVID-19 in May, or about 0.1%.
The Associated Press came up with these numbers using data from the Centers for Disease Control and Prevention. The CDC tracks the numbers of cases, hospitalizations, and deaths but does not breakdown rates by vaccination status.
Stronger argument for vaccination?
“The fact that only 0.8% of COVID-19 deaths are in the fully vaccinated should persuade those people still hesitant about vaccination,” said Hugh Cassiere, MD, medical director of Respiratory Therapy Services at North Shore University Hospital in Manhasset, New York.
Stuart C. Ray, MD, professor of medicine and oncology in the Division of Infectious Diseases at Johns Hopkins University, Baltimore, agreed. “It seems compelling, even for skeptics, that unvaccinated people represent 99% of those now dying from COVID-19 when they represent less than 50% of the adult population in the United States.”
The findings from the study could be more persuasive than previous arguments made in favor of immunization, Dr. Ray said. “These recent findings of striking reductions in risk of death in the vaccinated are more directly attributable and harder to ignore or dismiss.”
Brian Labus, PhD, MPH, of the University of Nevada Las Vegas (UNLV) is less convinced. “While this might change some peoples’ minds, it probably won’t make a major difference. People have many different reasons for not getting vaccinated, and this is only one of the things they consider.”
The study adds information that was not available before, said Dr. Labus, assistant professor in the Department of Epidemiology and Biostatistics at the UNLV School of Public Health. “We study the vaccine under tightly controlled, ideal conditions. This is the evidence that it works as well in the real world as it did in the trials, and that is what is most important in implementing a vaccination program,” added Dr. Labus.
“The scientific data has honed in on one thing: Vaccines are effective in preventing hospitalizations, ICU admissions, ventilations, and deaths,” agreed Dr. Palli.
“We now know that almost all deaths occurred in patients who were not vaccinated. We also know that all vaccines are effective against various strains that are in circulation right now, including the Delta variant, which is rapidly spreading,” Dr. Palli said.
Dr. Cassiere pointed out that the unvaccinated are not only at higher risk of developing COVID-19 but also of spreading, being hospitalized for, and dying from the infection. Avoiding “long hauler” symptoms is another argument in favor of immunization, he added.
As of June 28, the CDC reports that 63% of Americans 12 years and older have received at least one dose of a COVID-19 vaccine, and 54% are fully vaccinated.
Worldwide worry?
Although overall rates of U.S. COVID-19 hospitalizations and deaths are down, the outlook may not remain as encouraging. “I hope I’m wrong about this, but I anticipate that the coming fall and winter will bring increasingly localized versions of similar findings – severe disease and death due to SARS-CoV-2 infection in regions or groups with lower vaccination rates,” Dr. Ray said.
There could be a silver lining, he added: “If this unfortunate surge occurs, the health and economic consequences seem likely to erode much of the remaining hesitancy regarding vaccination.”
The rise of more infectious SARS-CoV-2 variants, such as the Delta variant, could also throw a wrench in controlling COVID-19. “This isn’t just a domestic issue,” Dr. Ray said. “We have learned that the world is a small place in pandemic times.”
The Associated Press investigators state that their findings support the high efficacy of the vaccine. Also, given the current widespread availability of COVID-19 vaccines in the United States, they believe many of the COVID-19 deaths now occurring are preventable.
Public health measures should have continued longer to protect unvaccinated individuals, especially Black Americans, Hispanic Americans, and other minorities, Dr. Palli said. “Only time will tell if re-opening and abandoning all public health measures by the CDC was premature.”
A version of this article first appeared on Medscape.com.
If you, a friend, or a loved one remain unvaccinated against COVID-19 at this point – for whatever reason – you are at higher risk of dying if you become infected.
That’s the conclusion of a new report released by the Associated Press looking at COVID-19 deaths during May 2021.
Of more than 18,000 people who died from COVID-19, for example, only about 150 were fully vaccinated. That’s less than 1%.
“Recently, I was working in the emergency room [and] I saw a 21-year-old African American who came in with shortness of breath,” said Vino K. Palli, MD, MPH, a physician specializing in emergency medicine, internal medicine, and urgent care.
The patient rapidly deteriorated and required intubation and ventilation. She was transferred to a specialized hospital for possible extracorporeal membrane oxygenation (ECMO) treatment.
“This patient was unvaccinated, along with her entire family. This would have been easily preventable,” added Dr. Palli, who is also founder and CEO of MiDoctor Urgent Care in New York City.
“Vaccine misinformation, compounded with vaccine inertia and vaccine access, have contributed to this,” he added. “Even though we have a surplus amount of vaccines at this time, we are only seeing 50% to 55% of completely vaccinated patients.”
Authors of the Associated Press report also acknowledge that some people who are fully vaccinated can get a breakthrough infection. These occurred in fewer than 1,200 of more than 853,000 people hospitalized for COVID-19 in May, or about 0.1%.
The Associated Press came up with these numbers using data from the Centers for Disease Control and Prevention. The CDC tracks the numbers of cases, hospitalizations, and deaths but does not breakdown rates by vaccination status.
Stronger argument for vaccination?
“The fact that only 0.8% of COVID-19 deaths are in the fully vaccinated should persuade those people still hesitant about vaccination,” said Hugh Cassiere, MD, medical director of Respiratory Therapy Services at North Shore University Hospital in Manhasset, New York.
Stuart C. Ray, MD, professor of medicine and oncology in the Division of Infectious Diseases at Johns Hopkins University, Baltimore, agreed. “It seems compelling, even for skeptics, that unvaccinated people represent 99% of those now dying from COVID-19 when they represent less than 50% of the adult population in the United States.”
The findings from the study could be more persuasive than previous arguments made in favor of immunization, Dr. Ray said. “These recent findings of striking reductions in risk of death in the vaccinated are more directly attributable and harder to ignore or dismiss.”
Brian Labus, PhD, MPH, of the University of Nevada Las Vegas (UNLV) is less convinced. “While this might change some peoples’ minds, it probably won’t make a major difference. People have many different reasons for not getting vaccinated, and this is only one of the things they consider.”
The study adds information that was not available before, said Dr. Labus, assistant professor in the Department of Epidemiology and Biostatistics at the UNLV School of Public Health. “We study the vaccine under tightly controlled, ideal conditions. This is the evidence that it works as well in the real world as it did in the trials, and that is what is most important in implementing a vaccination program,” added Dr. Labus.
“The scientific data has honed in on one thing: Vaccines are effective in preventing hospitalizations, ICU admissions, ventilations, and deaths,” agreed Dr. Palli.
“We now know that almost all deaths occurred in patients who were not vaccinated. We also know that all vaccines are effective against various strains that are in circulation right now, including the Delta variant, which is rapidly spreading,” Dr. Palli said.
Dr. Cassiere pointed out that the unvaccinated are not only at higher risk of developing COVID-19 but also of spreading, being hospitalized for, and dying from the infection. Avoiding “long hauler” symptoms is another argument in favor of immunization, he added.
As of June 28, the CDC reports that 63% of Americans 12 years and older have received at least one dose of a COVID-19 vaccine, and 54% are fully vaccinated.
Worldwide worry?
Although overall rates of U.S. COVID-19 hospitalizations and deaths are down, the outlook may not remain as encouraging. “I hope I’m wrong about this, but I anticipate that the coming fall and winter will bring increasingly localized versions of similar findings – severe disease and death due to SARS-CoV-2 infection in regions or groups with lower vaccination rates,” Dr. Ray said.
There could be a silver lining, he added: “If this unfortunate surge occurs, the health and economic consequences seem likely to erode much of the remaining hesitancy regarding vaccination.”
The rise of more infectious SARS-CoV-2 variants, such as the Delta variant, could also throw a wrench in controlling COVID-19. “This isn’t just a domestic issue,” Dr. Ray said. “We have learned that the world is a small place in pandemic times.”
The Associated Press investigators state that their findings support the high efficacy of the vaccine. Also, given the current widespread availability of COVID-19 vaccines in the United States, they believe many of the COVID-19 deaths now occurring are preventable.
Public health measures should have continued longer to protect unvaccinated individuals, especially Black Americans, Hispanic Americans, and other minorities, Dr. Palli said. “Only time will tell if re-opening and abandoning all public health measures by the CDC was premature.”
A version of this article first appeared on Medscape.com.
Physician fired after slurs, including ‘cannibalism,’ against Israel
Fidaa Wishah, MD, a pediatric radiologist at Phoenix Children’s Hospital in Arizona, has been fired after the hospital reviewed evidence that included her anti-Israel comments on social media, according to the hospital’s statement.
On May 26, Dr. Wishah posted, “We will uncover your thirst to kill our Palestinian children. … We sense your fear. The fear of your collapse. A state based on atrocity, inhumanity, racism and cannibalism never last long! Hey #israel … your end is coming sooner than you think.”
Phoenix Children’s Hospital did not respond to this news organization’s request for comment but said in a statement to the Jewish News Syndicate : “After a thorough review of the facts related to this matter, this individual is no longer providing care at Phoenix Children’s. All children in the care of Phoenix Children’s receive hope, healing and the best possible health care, regardless of race, color, disability, religion, gender, gender identity, sexual orientation or national origin.”
Dr. Wishah’s profile has been removed from the hospital website. Her LinkedIn profile indicates she had been a pediatric radiology fellow at Stanford (Calif.) University, specializing in advanced magnetic resonance imaging and fetal imaging and had been a senior staff pediatric radiologist at Henry Ford Health System in Detroit.
It wasn’t the first time antisemitic comments have led to the firing of a physician. Last year, this news organization wrote about Lara Kollab, DO, a first-year resident fired for her antisemitic tweets. She was subsequently barred from medicine.
In the same post from May 26, Dr. Wishah also wrote: “We will not be #censored anymore! Bomb our media buildings and we have the phones[.] Bribe the mainstream media and we have our small #socialmedia platforms[.] From our windows ... from our streets ... next the rubble we will expose you to the world[.] We will expose the #massacre and #genocide you #zionists are proud of[.]”
Today, CAIR-AZ, a group whose mission is to “enhance understanding of Islam, protect civil rights, promote justice, and empower American Muslims,” according to its website, announced that it, along with three private law firms, will represent Dr. Wishah in what they referred to as “her wrongful termination case against Phoenix Children’s Hospital.”
The announcement, which mentions that Dr. Wishah was born and raised in Gaza, said, “Dr. Wishah has been a medical doctor since 2010 and has spent the vast majority of her career as a pediatric physician. Despite caring for thousands of children, many of whom are Jewish, she has never been accused of discriminating against any of her patients or colleagues.”
The statement added, “PCH’s decision to terminate Dr. Wishah is shameful and an attack on freedom of speech.”
A version of this article first appeared on Medscape.com.
Fidaa Wishah, MD, a pediatric radiologist at Phoenix Children’s Hospital in Arizona, has been fired after the hospital reviewed evidence that included her anti-Israel comments on social media, according to the hospital’s statement.
On May 26, Dr. Wishah posted, “We will uncover your thirst to kill our Palestinian children. … We sense your fear. The fear of your collapse. A state based on atrocity, inhumanity, racism and cannibalism never last long! Hey #israel … your end is coming sooner than you think.”
Phoenix Children’s Hospital did not respond to this news organization’s request for comment but said in a statement to the Jewish News Syndicate : “After a thorough review of the facts related to this matter, this individual is no longer providing care at Phoenix Children’s. All children in the care of Phoenix Children’s receive hope, healing and the best possible health care, regardless of race, color, disability, religion, gender, gender identity, sexual orientation or national origin.”
Dr. Wishah’s profile has been removed from the hospital website. Her LinkedIn profile indicates she had been a pediatric radiology fellow at Stanford (Calif.) University, specializing in advanced magnetic resonance imaging and fetal imaging and had been a senior staff pediatric radiologist at Henry Ford Health System in Detroit.
It wasn’t the first time antisemitic comments have led to the firing of a physician. Last year, this news organization wrote about Lara Kollab, DO, a first-year resident fired for her antisemitic tweets. She was subsequently barred from medicine.
In the same post from May 26, Dr. Wishah also wrote: “We will not be #censored anymore! Bomb our media buildings and we have the phones[.] Bribe the mainstream media and we have our small #socialmedia platforms[.] From our windows ... from our streets ... next the rubble we will expose you to the world[.] We will expose the #massacre and #genocide you #zionists are proud of[.]”
Today, CAIR-AZ, a group whose mission is to “enhance understanding of Islam, protect civil rights, promote justice, and empower American Muslims,” according to its website, announced that it, along with three private law firms, will represent Dr. Wishah in what they referred to as “her wrongful termination case against Phoenix Children’s Hospital.”
The announcement, which mentions that Dr. Wishah was born and raised in Gaza, said, “Dr. Wishah has been a medical doctor since 2010 and has spent the vast majority of her career as a pediatric physician. Despite caring for thousands of children, many of whom are Jewish, she has never been accused of discriminating against any of her patients or colleagues.”
The statement added, “PCH’s decision to terminate Dr. Wishah is shameful and an attack on freedom of speech.”
A version of this article first appeared on Medscape.com.
Fidaa Wishah, MD, a pediatric radiologist at Phoenix Children’s Hospital in Arizona, has been fired after the hospital reviewed evidence that included her anti-Israel comments on social media, according to the hospital’s statement.
On May 26, Dr. Wishah posted, “We will uncover your thirst to kill our Palestinian children. … We sense your fear. The fear of your collapse. A state based on atrocity, inhumanity, racism and cannibalism never last long! Hey #israel … your end is coming sooner than you think.”
Phoenix Children’s Hospital did not respond to this news organization’s request for comment but said in a statement to the Jewish News Syndicate : “After a thorough review of the facts related to this matter, this individual is no longer providing care at Phoenix Children’s. All children in the care of Phoenix Children’s receive hope, healing and the best possible health care, regardless of race, color, disability, religion, gender, gender identity, sexual orientation or national origin.”
Dr. Wishah’s profile has been removed from the hospital website. Her LinkedIn profile indicates she had been a pediatric radiology fellow at Stanford (Calif.) University, specializing in advanced magnetic resonance imaging and fetal imaging and had been a senior staff pediatric radiologist at Henry Ford Health System in Detroit.
It wasn’t the first time antisemitic comments have led to the firing of a physician. Last year, this news organization wrote about Lara Kollab, DO, a first-year resident fired for her antisemitic tweets. She was subsequently barred from medicine.
In the same post from May 26, Dr. Wishah also wrote: “We will not be #censored anymore! Bomb our media buildings and we have the phones[.] Bribe the mainstream media and we have our small #socialmedia platforms[.] From our windows ... from our streets ... next the rubble we will expose you to the world[.] We will expose the #massacre and #genocide you #zionists are proud of[.]”
Today, CAIR-AZ, a group whose mission is to “enhance understanding of Islam, protect civil rights, promote justice, and empower American Muslims,” according to its website, announced that it, along with three private law firms, will represent Dr. Wishah in what they referred to as “her wrongful termination case against Phoenix Children’s Hospital.”
The announcement, which mentions that Dr. Wishah was born and raised in Gaza, said, “Dr. Wishah has been a medical doctor since 2010 and has spent the vast majority of her career as a pediatric physician. Despite caring for thousands of children, many of whom are Jewish, she has never been accused of discriminating against any of her patients or colleagues.”
The statement added, “PCH’s decision to terminate Dr. Wishah is shameful and an attack on freedom of speech.”
A version of this article first appeared on Medscape.com.
Wrong-site surgery doc says he can’t be sued
A neurosurgeon who operated on the wrong side of his patient’s spine claims he can’t be sued because of a federal law that protects health care professionals during a public health emergency, according to a report by KSDK, an NBC-affiliated television station in St. Louis.
Natalie Avilez, who lives in Missouri with her husband and five children, had been suffering from intense back pain. At some point in the recent past (the story doesn’t identify precisely when), she was referred to Fangxiang Chen, MD, a neurosurgeon affiliated with Mercy Hospital and Mercy Hospital South, in St. Louis. Ms. Avilez reportedly claims that Dr. Chen told her that an “easy” surgery – a hemilaminectomy – could relieve her back pain.
Something went wrong during the procedure, however. Dr. Chen ended up operating on the left side of Avilez’s spine instead of the right side, where he had initially diagnosed disk-related pressure. Dr. Chen realized his mistake while his patient was under anesthesia but couldn’t remedy it.
As the patient awakened, Dr. Chen asked her to authorize an immediate right-side surgery, but, as Ms. Avilez told the TV station, her “charge nurse would not let him get authorization because I wasn’t fully awake.” In the recovery room afterward, Dr. Chen explained what had happened to his patient, who permitted him to redo the surgery the following day.
But the redo didn’t remedy Ms. Avilez’s pain; in fact, the second surgery made things worse. “I’m always in constant pain,” she said. “I kind of feel like I would have been better off not even doing it at all.”
In January of this year, Ms. Avilez filed a medical malpractice suit against Dr. Chen and Mercy. But the neurosurgeon made a surprising claim:
Initially passed in 2005, PREP was intended to shield doctors and other licensed health care professionals from liability during a public health emergency except in cases of willful misconduct. On March 17, 2020, then–Health and Human Services Secretary Alex Azar invoked the PREP Act “for activities related to medical countermeasures against COVID-19.”
But could this declaration – which has since been amended multiple times – shield a physician from a claim of wrong-site surgery?
Ms. Avilez’s attorney, Morgan Murphy, doesn’t think so. “Obviously, we are not claiming that COVID had anything to do with the fact that Dr. Chen operated on the incorrect side of Natalie’s spine. It is a fairly straightforward situation. A doctor should never perform the incorrect surgery, period.”
Other observers are less certain that the Chen defense won’t hold. It’s true the PREP Act doesn’t protect doctors against claims of willful or intentional misconduct, says Deidre Gilbert, who leads a national medical malpractice patient-advocacy group. But such claims are, she quickly adds, very difficult to prove, never more so than during a pandemic.
Several states, including Missouri, have passed or are considering additional measures to protect health care professionals against the expected wave of COVID-related claims. (One estimate places the number of those claims at almost 6,000 as of February 2021.) “We want to make sure that there is a heightened standard for holding somebody liable in ... COVID transmission cases,” said the sponsor of the proposed Show-Me State legislation.
As for Ms. Avilez, she feels lucky that she’s not even worse off than she is now. She worries, though, about other patients who are less fortunate and who are told that the pandemic protects their health care professionals from liability. “That’s just not fair,” she says.
Hidden beliefs about people of color raise liability risks
Clinicians’ “implicit bias” can exacerbate medical disparities and also malpractice claims, a story in the Dayton Daily News reports.
The story’s authors cite La Fleur Small, PhD, a medical sociologist at Wayne State University, in Detroit, who sees “implicit bias” as a set of “unconscious associations and judgments” that affect social behavior, causing people to act in ways that are often contrary to their perceived value system. In the medical profession, such thinking can have unintended consequences, especially for people of color.
Implicit bias can erode the physician-patient relationship, which in turn can make a malpractice suit more likely should an adverse event occur. Studies reported in recent years in the AMA Journal of Ethics, for instance, found that poor communication was a factor in almost three-quarters of closed claims. Other studies have revealed that, of patients seeking legal advice following a medical mishap, more than half cited a poor doctor-patient relationship as a contributing factor in their decision.
To remedy things, it would be helpful to boost the number of doctors of color, at least to the point that it more closely reflects the percentage in the general population, say experts. Currently, although Black and Hispanic persons constitute 13.4% and 18.5%, respectively, of the overall U.S. population, they make up only 5.0% and 5.8% of active physicians. (As of 2018, 56.2% of all physicians were White and 17.2% were Asian, according to data from the Association of American Medical Colleges.)
Father of impaired baby seeks mega damages
An Oregon man whose son sustained permanent neurologic injuries during childbirth has sued the hospital where the 2017 delivery took place, as reported in The Astorian.
In the suit on behalf of his son, Wesley Humphries claims that Columbia Memorial Hospital in Astoria, Oregon, failed to monitor the baby’s heart rate and other aspects of the labor and delivery. As a consequence, the baby needed to be transferred to Oregon Health and Science University Hospital in Portland, approximately 100 miles away, for emergency treatment. Doctors there diagnosed the child as having hypoxic ischemic encephalopathy, which his lawyers say resulted in cerebral palsy, among other neurologic conditions.
Because of his son’s permanent impairment, Mr. Humphries is seeking significant damages: more than $45 million in medical, custodial, and life-care expenses and $65 million in noneconomic damages. Should his claim prove successful, the payout would mark one of the largest awards – if not the largest award – in Oregon State history. The hospital has declined to comment.
At press time, a trial date hadn’t been set.
A version of this article first appeared on Medscape.com.
A neurosurgeon who operated on the wrong side of his patient’s spine claims he can’t be sued because of a federal law that protects health care professionals during a public health emergency, according to a report by KSDK, an NBC-affiliated television station in St. Louis.
Natalie Avilez, who lives in Missouri with her husband and five children, had been suffering from intense back pain. At some point in the recent past (the story doesn’t identify precisely when), she was referred to Fangxiang Chen, MD, a neurosurgeon affiliated with Mercy Hospital and Mercy Hospital South, in St. Louis. Ms. Avilez reportedly claims that Dr. Chen told her that an “easy” surgery – a hemilaminectomy – could relieve her back pain.
Something went wrong during the procedure, however. Dr. Chen ended up operating on the left side of Avilez’s spine instead of the right side, where he had initially diagnosed disk-related pressure. Dr. Chen realized his mistake while his patient was under anesthesia but couldn’t remedy it.
As the patient awakened, Dr. Chen asked her to authorize an immediate right-side surgery, but, as Ms. Avilez told the TV station, her “charge nurse would not let him get authorization because I wasn’t fully awake.” In the recovery room afterward, Dr. Chen explained what had happened to his patient, who permitted him to redo the surgery the following day.
But the redo didn’t remedy Ms. Avilez’s pain; in fact, the second surgery made things worse. “I’m always in constant pain,” she said. “I kind of feel like I would have been better off not even doing it at all.”
In January of this year, Ms. Avilez filed a medical malpractice suit against Dr. Chen and Mercy. But the neurosurgeon made a surprising claim:
Initially passed in 2005, PREP was intended to shield doctors and other licensed health care professionals from liability during a public health emergency except in cases of willful misconduct. On March 17, 2020, then–Health and Human Services Secretary Alex Azar invoked the PREP Act “for activities related to medical countermeasures against COVID-19.”
But could this declaration – which has since been amended multiple times – shield a physician from a claim of wrong-site surgery?
Ms. Avilez’s attorney, Morgan Murphy, doesn’t think so. “Obviously, we are not claiming that COVID had anything to do with the fact that Dr. Chen operated on the incorrect side of Natalie’s spine. It is a fairly straightforward situation. A doctor should never perform the incorrect surgery, period.”
Other observers are less certain that the Chen defense won’t hold. It’s true the PREP Act doesn’t protect doctors against claims of willful or intentional misconduct, says Deidre Gilbert, who leads a national medical malpractice patient-advocacy group. But such claims are, she quickly adds, very difficult to prove, never more so than during a pandemic.
Several states, including Missouri, have passed or are considering additional measures to protect health care professionals against the expected wave of COVID-related claims. (One estimate places the number of those claims at almost 6,000 as of February 2021.) “We want to make sure that there is a heightened standard for holding somebody liable in ... COVID transmission cases,” said the sponsor of the proposed Show-Me State legislation.
As for Ms. Avilez, she feels lucky that she’s not even worse off than she is now. She worries, though, about other patients who are less fortunate and who are told that the pandemic protects their health care professionals from liability. “That’s just not fair,” she says.
Hidden beliefs about people of color raise liability risks
Clinicians’ “implicit bias” can exacerbate medical disparities and also malpractice claims, a story in the Dayton Daily News reports.
The story’s authors cite La Fleur Small, PhD, a medical sociologist at Wayne State University, in Detroit, who sees “implicit bias” as a set of “unconscious associations and judgments” that affect social behavior, causing people to act in ways that are often contrary to their perceived value system. In the medical profession, such thinking can have unintended consequences, especially for people of color.
Implicit bias can erode the physician-patient relationship, which in turn can make a malpractice suit more likely should an adverse event occur. Studies reported in recent years in the AMA Journal of Ethics, for instance, found that poor communication was a factor in almost three-quarters of closed claims. Other studies have revealed that, of patients seeking legal advice following a medical mishap, more than half cited a poor doctor-patient relationship as a contributing factor in their decision.
To remedy things, it would be helpful to boost the number of doctors of color, at least to the point that it more closely reflects the percentage in the general population, say experts. Currently, although Black and Hispanic persons constitute 13.4% and 18.5%, respectively, of the overall U.S. population, they make up only 5.0% and 5.8% of active physicians. (As of 2018, 56.2% of all physicians were White and 17.2% were Asian, according to data from the Association of American Medical Colleges.)
Father of impaired baby seeks mega damages
An Oregon man whose son sustained permanent neurologic injuries during childbirth has sued the hospital where the 2017 delivery took place, as reported in The Astorian.
In the suit on behalf of his son, Wesley Humphries claims that Columbia Memorial Hospital in Astoria, Oregon, failed to monitor the baby’s heart rate and other aspects of the labor and delivery. As a consequence, the baby needed to be transferred to Oregon Health and Science University Hospital in Portland, approximately 100 miles away, for emergency treatment. Doctors there diagnosed the child as having hypoxic ischemic encephalopathy, which his lawyers say resulted in cerebral palsy, among other neurologic conditions.
Because of his son’s permanent impairment, Mr. Humphries is seeking significant damages: more than $45 million in medical, custodial, and life-care expenses and $65 million in noneconomic damages. Should his claim prove successful, the payout would mark one of the largest awards – if not the largest award – in Oregon State history. The hospital has declined to comment.
At press time, a trial date hadn’t been set.
A version of this article first appeared on Medscape.com.
A neurosurgeon who operated on the wrong side of his patient’s spine claims he can’t be sued because of a federal law that protects health care professionals during a public health emergency, according to a report by KSDK, an NBC-affiliated television station in St. Louis.
Natalie Avilez, who lives in Missouri with her husband and five children, had been suffering from intense back pain. At some point in the recent past (the story doesn’t identify precisely when), she was referred to Fangxiang Chen, MD, a neurosurgeon affiliated with Mercy Hospital and Mercy Hospital South, in St. Louis. Ms. Avilez reportedly claims that Dr. Chen told her that an “easy” surgery – a hemilaminectomy – could relieve her back pain.
Something went wrong during the procedure, however. Dr. Chen ended up operating on the left side of Avilez’s spine instead of the right side, where he had initially diagnosed disk-related pressure. Dr. Chen realized his mistake while his patient was under anesthesia but couldn’t remedy it.
As the patient awakened, Dr. Chen asked her to authorize an immediate right-side surgery, but, as Ms. Avilez told the TV station, her “charge nurse would not let him get authorization because I wasn’t fully awake.” In the recovery room afterward, Dr. Chen explained what had happened to his patient, who permitted him to redo the surgery the following day.
But the redo didn’t remedy Ms. Avilez’s pain; in fact, the second surgery made things worse. “I’m always in constant pain,” she said. “I kind of feel like I would have been better off not even doing it at all.”
In January of this year, Ms. Avilez filed a medical malpractice suit against Dr. Chen and Mercy. But the neurosurgeon made a surprising claim:
Initially passed in 2005, PREP was intended to shield doctors and other licensed health care professionals from liability during a public health emergency except in cases of willful misconduct. On March 17, 2020, then–Health and Human Services Secretary Alex Azar invoked the PREP Act “for activities related to medical countermeasures against COVID-19.”
But could this declaration – which has since been amended multiple times – shield a physician from a claim of wrong-site surgery?
Ms. Avilez’s attorney, Morgan Murphy, doesn’t think so. “Obviously, we are not claiming that COVID had anything to do with the fact that Dr. Chen operated on the incorrect side of Natalie’s spine. It is a fairly straightforward situation. A doctor should never perform the incorrect surgery, period.”
Other observers are less certain that the Chen defense won’t hold. It’s true the PREP Act doesn’t protect doctors against claims of willful or intentional misconduct, says Deidre Gilbert, who leads a national medical malpractice patient-advocacy group. But such claims are, she quickly adds, very difficult to prove, never more so than during a pandemic.
Several states, including Missouri, have passed or are considering additional measures to protect health care professionals against the expected wave of COVID-related claims. (One estimate places the number of those claims at almost 6,000 as of February 2021.) “We want to make sure that there is a heightened standard for holding somebody liable in ... COVID transmission cases,” said the sponsor of the proposed Show-Me State legislation.
As for Ms. Avilez, she feels lucky that she’s not even worse off than she is now. She worries, though, about other patients who are less fortunate and who are told that the pandemic protects their health care professionals from liability. “That’s just not fair,” she says.
Hidden beliefs about people of color raise liability risks
Clinicians’ “implicit bias” can exacerbate medical disparities and also malpractice claims, a story in the Dayton Daily News reports.
The story’s authors cite La Fleur Small, PhD, a medical sociologist at Wayne State University, in Detroit, who sees “implicit bias” as a set of “unconscious associations and judgments” that affect social behavior, causing people to act in ways that are often contrary to their perceived value system. In the medical profession, such thinking can have unintended consequences, especially for people of color.
Implicit bias can erode the physician-patient relationship, which in turn can make a malpractice suit more likely should an adverse event occur. Studies reported in recent years in the AMA Journal of Ethics, for instance, found that poor communication was a factor in almost three-quarters of closed claims. Other studies have revealed that, of patients seeking legal advice following a medical mishap, more than half cited a poor doctor-patient relationship as a contributing factor in their decision.
To remedy things, it would be helpful to boost the number of doctors of color, at least to the point that it more closely reflects the percentage in the general population, say experts. Currently, although Black and Hispanic persons constitute 13.4% and 18.5%, respectively, of the overall U.S. population, they make up only 5.0% and 5.8% of active physicians. (As of 2018, 56.2% of all physicians were White and 17.2% were Asian, according to data from the Association of American Medical Colleges.)
Father of impaired baby seeks mega damages
An Oregon man whose son sustained permanent neurologic injuries during childbirth has sued the hospital where the 2017 delivery took place, as reported in The Astorian.
In the suit on behalf of his son, Wesley Humphries claims that Columbia Memorial Hospital in Astoria, Oregon, failed to monitor the baby’s heart rate and other aspects of the labor and delivery. As a consequence, the baby needed to be transferred to Oregon Health and Science University Hospital in Portland, approximately 100 miles away, for emergency treatment. Doctors there diagnosed the child as having hypoxic ischemic encephalopathy, which his lawyers say resulted in cerebral palsy, among other neurologic conditions.
Because of his son’s permanent impairment, Mr. Humphries is seeking significant damages: more than $45 million in medical, custodial, and life-care expenses and $65 million in noneconomic damages. Should his claim prove successful, the payout would mark one of the largest awards – if not the largest award – in Oregon State history. The hospital has declined to comment.
At press time, a trial date hadn’t been set.
A version of this article first appeared on Medscape.com.
EAS lipid guidance: Start high-risk patients on combo drug
Very-high-risk dyslipidemia patients unlikely to reach goal with a statin should be given combination statin–ezetimibe (Nustendi) therapy upfront, rather than wasting time and resources on trialing a statin alone, suggests a practical guidance document.
The document points out that, even with high-intensity statin therapy, patients achieve a reduction in low-density-lipoprotein (LDL) cholesterol levels of around 50%, which for many is not enough for them to achieve the stringent new guideline targets deemed necessary for risk reduction.
Instead, clinicians should determine at the first visit whether their patient, if they are not already on a statin, is likely to reach their goal with that drug alone, and if not, should immediately start them on the combination.
The guidance, which aims to offer a practical way to implement the 2019 European Society of Cardiology/EAS guidelines for the management of dyslipidemias, was published April 12 in Atherosclerosis .
Lead author Alberico L. Catapano, MD, PhD, discussed the new practical guidance at the recent European Atherosclerosis Society (EAS) 2021 Virtual Congress.
He explained that the motivation for creating the practical guidance was “very simple” and concerns something already embedded in the ESC/EAS guidelines; it’s just that “people didn’t notice” it.
Dr. Catapano, professor of pharmacology at the University of Milan and past president of the EAS, said the guidelines set out the average reduction in LDL-cholesterol levels “you can get by starting with high-intensity therapy and/or starting with a combination therapy.”
The guidelines, he said, suggest steps for achieving lipid control: Begin with a statin, add ezetimibe if the patient is still not at goal, and proceed to a proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor if the patient is still not at target levels.
Dr. Catapano added that, “having said that, at the beginning, you can guess by knowing how far you are from the goal as to whether a statin by itself with help you get [there].”
If clinicians follow the new practical guidance of giving upfront combination statin–ezetimibe therapy in very-high-risk patients with high LDL-cholesterol levels, it will “save a lot of time, a lot of clinic visits, and will you get you to goal earlier.”
He gave the example of a patient who has an LDL-cholesterol level of 190 mg/dL, who would be classified as being at very high risk. With the target goal of 55 mg/dL, “you would never be able to get them to goal [with only] a high-intensity statin.”
The addition of ezetimibe to the regimen of this patient would have two advantages, Dr. Catapano said. The first is that “you get to goal more easily,” and the second is that, with the drugs available as a single-pill combination, it “makes it easier for the patient to be compliant.”
Consequently, there will be no “unnecessary back and forth,” he said. “Some of these are young people. They go to work; one less visit is less time lost at work.
“This is a practical issue,” he added. “It doesn’t contradict the guidelines,” it’s about “everyday clinical practice.”
Useful between updates
Responding to the guidance, Donald Lloyd-Jones, MD, president-elect of the American Heart Association (AHA), told this news organization that “this kind of document can be useful in periods between updates of the formal guidelines.”
New evidence comes out in between guidelines, and they “don’t often provide us with all of the practical solutions needed for everyday guidance when we’re dealing with individual patients with real-world problems.”
Dr. Lloyd-Jones, who is chair of the Department of Preventive Medicine at Northwestern University, Chicago, said the 2019 ESC/EAS guidelines set “quite aggressive targets, particularly for LDL cholesterol … but didn’t really provide much practical advice on how clinicians could get there for their patients.”
“While this document doesn’t completely address all patient groups, it does provide some good practical advice,” recognizing that “if you need to get to a certain LDL target, it’s unlikely you’re going to get there with just a statin in certain types of patients,” and “if you need a certain amount of LDL lowering, it’s certainly reasonable to start upfront with a statin and ezetimibe and see how you do.”
Crucially, Dr. Lloyd-Jones believes that the practical guidance does “flesh out some of the details the guidelines didn’t address.”
In terms of the aggressive LDL-cholesterol targets set out in the original guidelines, he said that “everyone agrees that lower is always better … and we’ve not get yet found a level that is too low.”
Further, “we’re certainly pushing patients lower and lower, especially with the use of PCSK9 inhibitors, so I think the general philosophy is consistent and correct,” although “it’s difficult to point to great evidence from clinical trials that specially says that 55 mg/dL or 40 mg/dL is the right target for a given group of patients.”
“There’s really very limited evidence for those specific numbers,” Dr. Lloyd-Jones added, “but I think everyone agrees, especially for patients at higher risk, the lower we can get them the better. What really matters, and what this document starts to address, is how we achieve as low as possible, and I think there are some important considerations that they take into account.”
Aside from how far patients need their LDL cholesterol lowered from baseline, there are issues like cost and patient preference for different types of medication, and these “weren’t particularly well addressed in the guidelines,” he added.
Scott D. Isaacs, MD, Secretary of the American Association of Clinical Endocrinologists (AACE), commented that the ESC/EAS recommendations echo the 2017 AACE/American College of Endocrinology guidelines for management of dyslipidemia and prevention of cardiovascular disease.
He said that both guidelines “call for the need to lower LDL cholesterol as much as possible to prevent cardiovascular disease.”
He agreed, however, that high- or very-high-risk patients “have aggressive LDL targets that are often lower than what can be accomplished with high-dose, high-intensity statin monotherapy. Therefore, starting with combination therapy … will get more patients to goal more quickly and will prevent more cardiovascular events.”
Isaacs added: “It just makes sense that if you know a drug will not be strong enough, then you should start with two drugs.”
He noted that this approach is commonly used for conditions such as diabetes and hypertension, “when monotherapy is not expected to achieve the desired results.”
Dr. Isaacs also underlined that the combination of a statin plus ezetimibe “is appealing because of the price and ease of use.”
Although PCSK9 inhibitors are more potent and achieve even lower LDL levels, “the higher price and need to take an injection has limited their use,” he noted.
“One would expect that as the prices of PCSK9 inhibitors come down, their place in care pathways will move up since they are more effective and have proven cardiovascular benefit, but for now, statin plus ezetimibe is a potent and cost-effective way to achieve LDL targets in high- and very-high-risk patients,” Dr. Isaacs concluded.
One issue Dr. Lloyd-Jones raised with the ESC/EAS guidelines is that they seem to have put a lot of weight Mendelian randomization analysis.
“Those are useful in understanding whether having low LDL-cholesterol levels or triglycerides naturally are better for you – of course they are – but they actually provide no evidence about treatment effects, so I think what we need from that is actual data from the clinical trials to understand the treatment effects, both positive and negative.”
He added that that “really then helps us to drive to how and in whom we want to achieve the lowest levels possible.”
Dr. Lloyd-Jones said that Mendelian randomization analyses “continue to crop in a lot of these ESC and EAS documents,” and although they are “elegant and interesting,” they “don’t really inform treatment at all.”
No funding declared. Catapano declares relationships with Pfizer, Sanofi, Regeneron, Merck, Mediolanum, SigmaTau, Menarini, Kowa, Recordati, Eli Lilly, AstraZeneca, Merck, Aegerion and Amgen.
A version of this article first appeared on Medscape.com.
Very-high-risk dyslipidemia patients unlikely to reach goal with a statin should be given combination statin–ezetimibe (Nustendi) therapy upfront, rather than wasting time and resources on trialing a statin alone, suggests a practical guidance document.
The document points out that, even with high-intensity statin therapy, patients achieve a reduction in low-density-lipoprotein (LDL) cholesterol levels of around 50%, which for many is not enough for them to achieve the stringent new guideline targets deemed necessary for risk reduction.
Instead, clinicians should determine at the first visit whether their patient, if they are not already on a statin, is likely to reach their goal with that drug alone, and if not, should immediately start them on the combination.
The guidance, which aims to offer a practical way to implement the 2019 European Society of Cardiology/EAS guidelines for the management of dyslipidemias, was published April 12 in Atherosclerosis .
Lead author Alberico L. Catapano, MD, PhD, discussed the new practical guidance at the recent European Atherosclerosis Society (EAS) 2021 Virtual Congress.
He explained that the motivation for creating the practical guidance was “very simple” and concerns something already embedded in the ESC/EAS guidelines; it’s just that “people didn’t notice” it.
Dr. Catapano, professor of pharmacology at the University of Milan and past president of the EAS, said the guidelines set out the average reduction in LDL-cholesterol levels “you can get by starting with high-intensity therapy and/or starting with a combination therapy.”
The guidelines, he said, suggest steps for achieving lipid control: Begin with a statin, add ezetimibe if the patient is still not at goal, and proceed to a proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor if the patient is still not at target levels.
Dr. Catapano added that, “having said that, at the beginning, you can guess by knowing how far you are from the goal as to whether a statin by itself with help you get [there].”
If clinicians follow the new practical guidance of giving upfront combination statin–ezetimibe therapy in very-high-risk patients with high LDL-cholesterol levels, it will “save a lot of time, a lot of clinic visits, and will you get you to goal earlier.”
He gave the example of a patient who has an LDL-cholesterol level of 190 mg/dL, who would be classified as being at very high risk. With the target goal of 55 mg/dL, “you would never be able to get them to goal [with only] a high-intensity statin.”
The addition of ezetimibe to the regimen of this patient would have two advantages, Dr. Catapano said. The first is that “you get to goal more easily,” and the second is that, with the drugs available as a single-pill combination, it “makes it easier for the patient to be compliant.”
Consequently, there will be no “unnecessary back and forth,” he said. “Some of these are young people. They go to work; one less visit is less time lost at work.
“This is a practical issue,” he added. “It doesn’t contradict the guidelines,” it’s about “everyday clinical practice.”
Useful between updates
Responding to the guidance, Donald Lloyd-Jones, MD, president-elect of the American Heart Association (AHA), told this news organization that “this kind of document can be useful in periods between updates of the formal guidelines.”
New evidence comes out in between guidelines, and they “don’t often provide us with all of the practical solutions needed for everyday guidance when we’re dealing with individual patients with real-world problems.”
Dr. Lloyd-Jones, who is chair of the Department of Preventive Medicine at Northwestern University, Chicago, said the 2019 ESC/EAS guidelines set “quite aggressive targets, particularly for LDL cholesterol … but didn’t really provide much practical advice on how clinicians could get there for their patients.”
“While this document doesn’t completely address all patient groups, it does provide some good practical advice,” recognizing that “if you need to get to a certain LDL target, it’s unlikely you’re going to get there with just a statin in certain types of patients,” and “if you need a certain amount of LDL lowering, it’s certainly reasonable to start upfront with a statin and ezetimibe and see how you do.”
Crucially, Dr. Lloyd-Jones believes that the practical guidance does “flesh out some of the details the guidelines didn’t address.”
In terms of the aggressive LDL-cholesterol targets set out in the original guidelines, he said that “everyone agrees that lower is always better … and we’ve not get yet found a level that is too low.”
Further, “we’re certainly pushing patients lower and lower, especially with the use of PCSK9 inhibitors, so I think the general philosophy is consistent and correct,” although “it’s difficult to point to great evidence from clinical trials that specially says that 55 mg/dL or 40 mg/dL is the right target for a given group of patients.”
“There’s really very limited evidence for those specific numbers,” Dr. Lloyd-Jones added, “but I think everyone agrees, especially for patients at higher risk, the lower we can get them the better. What really matters, and what this document starts to address, is how we achieve as low as possible, and I think there are some important considerations that they take into account.”
Aside from how far patients need their LDL cholesterol lowered from baseline, there are issues like cost and patient preference for different types of medication, and these “weren’t particularly well addressed in the guidelines,” he added.
Scott D. Isaacs, MD, Secretary of the American Association of Clinical Endocrinologists (AACE), commented that the ESC/EAS recommendations echo the 2017 AACE/American College of Endocrinology guidelines for management of dyslipidemia and prevention of cardiovascular disease.
He said that both guidelines “call for the need to lower LDL cholesterol as much as possible to prevent cardiovascular disease.”
He agreed, however, that high- or very-high-risk patients “have aggressive LDL targets that are often lower than what can be accomplished with high-dose, high-intensity statin monotherapy. Therefore, starting with combination therapy … will get more patients to goal more quickly and will prevent more cardiovascular events.”
Isaacs added: “It just makes sense that if you know a drug will not be strong enough, then you should start with two drugs.”
He noted that this approach is commonly used for conditions such as diabetes and hypertension, “when monotherapy is not expected to achieve the desired results.”
Dr. Isaacs also underlined that the combination of a statin plus ezetimibe “is appealing because of the price and ease of use.”
Although PCSK9 inhibitors are more potent and achieve even lower LDL levels, “the higher price and need to take an injection has limited their use,” he noted.
“One would expect that as the prices of PCSK9 inhibitors come down, their place in care pathways will move up since they are more effective and have proven cardiovascular benefit, but for now, statin plus ezetimibe is a potent and cost-effective way to achieve LDL targets in high- and very-high-risk patients,” Dr. Isaacs concluded.
One issue Dr. Lloyd-Jones raised with the ESC/EAS guidelines is that they seem to have put a lot of weight Mendelian randomization analysis.
“Those are useful in understanding whether having low LDL-cholesterol levels or triglycerides naturally are better for you – of course they are – but they actually provide no evidence about treatment effects, so I think what we need from that is actual data from the clinical trials to understand the treatment effects, both positive and negative.”
He added that that “really then helps us to drive to how and in whom we want to achieve the lowest levels possible.”
Dr. Lloyd-Jones said that Mendelian randomization analyses “continue to crop in a lot of these ESC and EAS documents,” and although they are “elegant and interesting,” they “don’t really inform treatment at all.”
No funding declared. Catapano declares relationships with Pfizer, Sanofi, Regeneron, Merck, Mediolanum, SigmaTau, Menarini, Kowa, Recordati, Eli Lilly, AstraZeneca, Merck, Aegerion and Amgen.
A version of this article first appeared on Medscape.com.
Very-high-risk dyslipidemia patients unlikely to reach goal with a statin should be given combination statin–ezetimibe (Nustendi) therapy upfront, rather than wasting time and resources on trialing a statin alone, suggests a practical guidance document.
The document points out that, even with high-intensity statin therapy, patients achieve a reduction in low-density-lipoprotein (LDL) cholesterol levels of around 50%, which for many is not enough for them to achieve the stringent new guideline targets deemed necessary for risk reduction.
Instead, clinicians should determine at the first visit whether their patient, if they are not already on a statin, is likely to reach their goal with that drug alone, and if not, should immediately start them on the combination.
The guidance, which aims to offer a practical way to implement the 2019 European Society of Cardiology/EAS guidelines for the management of dyslipidemias, was published April 12 in Atherosclerosis .
Lead author Alberico L. Catapano, MD, PhD, discussed the new practical guidance at the recent European Atherosclerosis Society (EAS) 2021 Virtual Congress.
He explained that the motivation for creating the practical guidance was “very simple” and concerns something already embedded in the ESC/EAS guidelines; it’s just that “people didn’t notice” it.
Dr. Catapano, professor of pharmacology at the University of Milan and past president of the EAS, said the guidelines set out the average reduction in LDL-cholesterol levels “you can get by starting with high-intensity therapy and/or starting with a combination therapy.”
The guidelines, he said, suggest steps for achieving lipid control: Begin with a statin, add ezetimibe if the patient is still not at goal, and proceed to a proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor if the patient is still not at target levels.
Dr. Catapano added that, “having said that, at the beginning, you can guess by knowing how far you are from the goal as to whether a statin by itself with help you get [there].”
If clinicians follow the new practical guidance of giving upfront combination statin–ezetimibe therapy in very-high-risk patients with high LDL-cholesterol levels, it will “save a lot of time, a lot of clinic visits, and will you get you to goal earlier.”
He gave the example of a patient who has an LDL-cholesterol level of 190 mg/dL, who would be classified as being at very high risk. With the target goal of 55 mg/dL, “you would never be able to get them to goal [with only] a high-intensity statin.”
The addition of ezetimibe to the regimen of this patient would have two advantages, Dr. Catapano said. The first is that “you get to goal more easily,” and the second is that, with the drugs available as a single-pill combination, it “makes it easier for the patient to be compliant.”
Consequently, there will be no “unnecessary back and forth,” he said. “Some of these are young people. They go to work; one less visit is less time lost at work.
“This is a practical issue,” he added. “It doesn’t contradict the guidelines,” it’s about “everyday clinical practice.”
Useful between updates
Responding to the guidance, Donald Lloyd-Jones, MD, president-elect of the American Heart Association (AHA), told this news organization that “this kind of document can be useful in periods between updates of the formal guidelines.”
New evidence comes out in between guidelines, and they “don’t often provide us with all of the practical solutions needed for everyday guidance when we’re dealing with individual patients with real-world problems.”
Dr. Lloyd-Jones, who is chair of the Department of Preventive Medicine at Northwestern University, Chicago, said the 2019 ESC/EAS guidelines set “quite aggressive targets, particularly for LDL cholesterol … but didn’t really provide much practical advice on how clinicians could get there for their patients.”
“While this document doesn’t completely address all patient groups, it does provide some good practical advice,” recognizing that “if you need to get to a certain LDL target, it’s unlikely you’re going to get there with just a statin in certain types of patients,” and “if you need a certain amount of LDL lowering, it’s certainly reasonable to start upfront with a statin and ezetimibe and see how you do.”
Crucially, Dr. Lloyd-Jones believes that the practical guidance does “flesh out some of the details the guidelines didn’t address.”
In terms of the aggressive LDL-cholesterol targets set out in the original guidelines, he said that “everyone agrees that lower is always better … and we’ve not get yet found a level that is too low.”
Further, “we’re certainly pushing patients lower and lower, especially with the use of PCSK9 inhibitors, so I think the general philosophy is consistent and correct,” although “it’s difficult to point to great evidence from clinical trials that specially says that 55 mg/dL or 40 mg/dL is the right target for a given group of patients.”
“There’s really very limited evidence for those specific numbers,” Dr. Lloyd-Jones added, “but I think everyone agrees, especially for patients at higher risk, the lower we can get them the better. What really matters, and what this document starts to address, is how we achieve as low as possible, and I think there are some important considerations that they take into account.”
Aside from how far patients need their LDL cholesterol lowered from baseline, there are issues like cost and patient preference for different types of medication, and these “weren’t particularly well addressed in the guidelines,” he added.
Scott D. Isaacs, MD, Secretary of the American Association of Clinical Endocrinologists (AACE), commented that the ESC/EAS recommendations echo the 2017 AACE/American College of Endocrinology guidelines for management of dyslipidemia and prevention of cardiovascular disease.
He said that both guidelines “call for the need to lower LDL cholesterol as much as possible to prevent cardiovascular disease.”
He agreed, however, that high- or very-high-risk patients “have aggressive LDL targets that are often lower than what can be accomplished with high-dose, high-intensity statin monotherapy. Therefore, starting with combination therapy … will get more patients to goal more quickly and will prevent more cardiovascular events.”
Isaacs added: “It just makes sense that if you know a drug will not be strong enough, then you should start with two drugs.”
He noted that this approach is commonly used for conditions such as diabetes and hypertension, “when monotherapy is not expected to achieve the desired results.”
Dr. Isaacs also underlined that the combination of a statin plus ezetimibe “is appealing because of the price and ease of use.”
Although PCSK9 inhibitors are more potent and achieve even lower LDL levels, “the higher price and need to take an injection has limited their use,” he noted.
“One would expect that as the prices of PCSK9 inhibitors come down, their place in care pathways will move up since they are more effective and have proven cardiovascular benefit, but for now, statin plus ezetimibe is a potent and cost-effective way to achieve LDL targets in high- and very-high-risk patients,” Dr. Isaacs concluded.
One issue Dr. Lloyd-Jones raised with the ESC/EAS guidelines is that they seem to have put a lot of weight Mendelian randomization analysis.
“Those are useful in understanding whether having low LDL-cholesterol levels or triglycerides naturally are better for you – of course they are – but they actually provide no evidence about treatment effects, so I think what we need from that is actual data from the clinical trials to understand the treatment effects, both positive and negative.”
He added that that “really then helps us to drive to how and in whom we want to achieve the lowest levels possible.”
Dr. Lloyd-Jones said that Mendelian randomization analyses “continue to crop in a lot of these ESC and EAS documents,” and although they are “elegant and interesting,” they “don’t really inform treatment at all.”
No funding declared. Catapano declares relationships with Pfizer, Sanofi, Regeneron, Merck, Mediolanum, SigmaTau, Menarini, Kowa, Recordati, Eli Lilly, AstraZeneca, Merck, Aegerion and Amgen.
A version of this article first appeared on Medscape.com.
Maintain OMT for 5 years after revascularization, boost survival at 10 years: SYNTAXES
When it comes to medical therapy after a coronary revascularization procedure, more is better. Patients started and then maintained indefinitely on more rather than fewer of the drugs identified as optimal medical therapy (OMT) achieve a major survival benefit 10 years later, according to long-term follow-up from an extended analysis of the SYNTAX trial.
For the survival benefit at 10 years, “the present study suggests that at least three types of optimal medical therapy should be maintained for at least 5 years after revascularization,” reported a multinational team of cardiovascular specialists led by Hideyuki Kawashima, MD and Patrick Serruys, MD, who both have affiliations with the department of cardiology of the National University of Ireland, Galway.
The SYNTAX trial was conducted to compare percutaneous coronary intervention (PCI) with coronary artery bypass grafting (CABG) for patients with previously untreated three-vessel and/or left main disease (N Engl J Med 2009;360:961-72). The conclusion from that study, published in 2009 and subsequently reinforced by a 5-year follow-up, was that CABG should remain the standard of care for complex lesions.
Optimal medical therapy defined
In the course of SYNTAX, the impact of OMT on outcome was also evaluated in a subanalysis. At 5 years, there was a mortality advantage for those receiving an antiplatelet drug, a statin, a renin-angiotensin system inhibitor (ACE inhibitor or angiotensin receptor blocker), and a beta-blocker when compared with fewer of these agents.
When an investigator-initiated extension of SYNTAX, called SYNTAXES, was conducted to compare the outcomes of PCI and CABG at 10 years, it also permitted an extended analysis of OMT. Although the primary comparison of SYNTAXES, reported 2 years ago, did not show a significant difference between PCI and CABG for mortality at 10 years, there was a difference for OMT.
When investigators compared treatment with three or more OMT agents with that with two or fewer OMT drugs at 5 years, the result for all-cause death at 10 years translated into a more than 50% relative reduction (hazard ratio, 0.47; P = .002). The absolute difference in mortality was a more than 6% reduction (13.1% vs. 19.9%).
OMT data offer major message
The current study is considered to have a major message for patients as well as physicians.
“OMT even outweighs the survival benefit from revascularization alone, so our patients should convince themselves of the value of rigorous adherence and compliance,” Dr. Serruys said in an interview. According to him, these are compelling data for telling patients that OMT “is the best insurance for extended survival.” We now know from these data “the longer, the better.”
The same message from these data extends to physicians.
“I wish I could understand the apparent blind spot physicians have with respect to prescribing OMT despite the overwhelming benefit from multiple clinical trials,” said William E. Boden, MD, professor of medicine, Boston University.
Dr. Boden was a coauthor of an editorial accompanying the newly published SYNTAXES subanalysis. In the editorial, he noted that OMT following revascularization and in other high-risk patients “has been unacceptably low,” but he was asked to expand on the lessons from the newly released SYNTAXES subanalysis in an interview.
“There has often been a belief that revascularization negates the need for OMT and that’s why the SYNTAXES trial 10-year mortality reduction – which builds upon an earlier 5-year mortality reduction analysis – is so important,” he said.
Patients should take OMT long term
These data “should be both a motivator for physicians to prescribe OMT and for patients to remain adherent to OMT,” he said. “It is the best warranty to blunt the progression of atherosclerosis and to reduce subsequent cardiac events.”
For the 10-year subanalysis of OMT in SYNTAXES, the patients were stratified by the number of OMTs they were taking at 5 years after revascularization and then evaluated for survival at 10 years. Of the 1,472 patients available for analysis at 5 years, only 678 (46%) were on OMT. The other 794 patients were not.
Graphically, the Kaplan-Meier survival curve for those on three types of OMT was consistently beneath that of those on four OMTs, but the gap narrowed over time. At the end of 10 years, the advantage of the four-drug OMT was not statistically significant relative to three or fewer (13.1% vs. 12.7%).
Statins and antiplatelets show largest effect
When analyzed individually and in different combinations, the agents with OMT did not appear to be equal. For example, the biggest survival gap at 10 years was for those who were on an antiplatelet therapy and a statin at 5 years relative to those who were not on either (13.2% vs. 22.6%; P = .006). Even after adjustment, there was nearly 45% survival benefit for these two agents (HR, 0.556; P = .02).
Conversely, the 10-year survival advantage for being on a renin-angiotensin system inhibitor at 5 years versus not being exposed to this therapy was small and nonsignificant (14.7% vs. 13.7%; P = .651).
The precise proportion of patients who were prescribed and adhered to OMT between 5 years and 10 years is unknown, acknowledged the authors, so conclusions are limited about the added benefit of 10- versus 5-year OMT, although the authors presume that a substantial proportion of those adherent for 5 years would likely continue on these therapies.
It can be said with confidence that those adherent for at least 5 years are more likely to be alive at 10 years than those who are not, according to Dr. Boden. He considers these data a call for physicians and all high-risk patients, not just those who have undergone revascularization, to take these standard therapies.
There are plenty of data to “show how poorly we treat patients with OMT,” said Dr. Boden, citing several studies. In one, which looked at OMT in a nationally representative sample in the United States, only a third of patients with angina were taking an antiplatelet, a statin, and a beta-blocker, all of which are indicated.
“Hospitalization for revascularization provides an opportune time to capture the attention of patients and their physicians,” he wrote in his editorial. He called OMT “an imperative to optimize clinical outcomes.”
Many of the investigators involved in the SYNTAXES subanalysis, including Dr. Serruys, have financial relationships with multiple pharmaceutical companies, including Boston Scientific, which provided the initial funding for the SYNTAX trial. Dr. Boden reports no potential conflicts of interest.
When it comes to medical therapy after a coronary revascularization procedure, more is better. Patients started and then maintained indefinitely on more rather than fewer of the drugs identified as optimal medical therapy (OMT) achieve a major survival benefit 10 years later, according to long-term follow-up from an extended analysis of the SYNTAX trial.
For the survival benefit at 10 years, “the present study suggests that at least three types of optimal medical therapy should be maintained for at least 5 years after revascularization,” reported a multinational team of cardiovascular specialists led by Hideyuki Kawashima, MD and Patrick Serruys, MD, who both have affiliations with the department of cardiology of the National University of Ireland, Galway.
The SYNTAX trial was conducted to compare percutaneous coronary intervention (PCI) with coronary artery bypass grafting (CABG) for patients with previously untreated three-vessel and/or left main disease (N Engl J Med 2009;360:961-72). The conclusion from that study, published in 2009 and subsequently reinforced by a 5-year follow-up, was that CABG should remain the standard of care for complex lesions.
Optimal medical therapy defined
In the course of SYNTAX, the impact of OMT on outcome was also evaluated in a subanalysis. At 5 years, there was a mortality advantage for those receiving an antiplatelet drug, a statin, a renin-angiotensin system inhibitor (ACE inhibitor or angiotensin receptor blocker), and a beta-blocker when compared with fewer of these agents.
When an investigator-initiated extension of SYNTAX, called SYNTAXES, was conducted to compare the outcomes of PCI and CABG at 10 years, it also permitted an extended analysis of OMT. Although the primary comparison of SYNTAXES, reported 2 years ago, did not show a significant difference between PCI and CABG for mortality at 10 years, there was a difference for OMT.
When investigators compared treatment with three or more OMT agents with that with two or fewer OMT drugs at 5 years, the result for all-cause death at 10 years translated into a more than 50% relative reduction (hazard ratio, 0.47; P = .002). The absolute difference in mortality was a more than 6% reduction (13.1% vs. 19.9%).
OMT data offer major message
The current study is considered to have a major message for patients as well as physicians.
“OMT even outweighs the survival benefit from revascularization alone, so our patients should convince themselves of the value of rigorous adherence and compliance,” Dr. Serruys said in an interview. According to him, these are compelling data for telling patients that OMT “is the best insurance for extended survival.” We now know from these data “the longer, the better.”
The same message from these data extends to physicians.
“I wish I could understand the apparent blind spot physicians have with respect to prescribing OMT despite the overwhelming benefit from multiple clinical trials,” said William E. Boden, MD, professor of medicine, Boston University.
Dr. Boden was a coauthor of an editorial accompanying the newly published SYNTAXES subanalysis. In the editorial, he noted that OMT following revascularization and in other high-risk patients “has been unacceptably low,” but he was asked to expand on the lessons from the newly released SYNTAXES subanalysis in an interview.
“There has often been a belief that revascularization negates the need for OMT and that’s why the SYNTAXES trial 10-year mortality reduction – which builds upon an earlier 5-year mortality reduction analysis – is so important,” he said.
Patients should take OMT long term
These data “should be both a motivator for physicians to prescribe OMT and for patients to remain adherent to OMT,” he said. “It is the best warranty to blunt the progression of atherosclerosis and to reduce subsequent cardiac events.”
For the 10-year subanalysis of OMT in SYNTAXES, the patients were stratified by the number of OMTs they were taking at 5 years after revascularization and then evaluated for survival at 10 years. Of the 1,472 patients available for analysis at 5 years, only 678 (46%) were on OMT. The other 794 patients were not.
Graphically, the Kaplan-Meier survival curve for those on three types of OMT was consistently beneath that of those on four OMTs, but the gap narrowed over time. At the end of 10 years, the advantage of the four-drug OMT was not statistically significant relative to three or fewer (13.1% vs. 12.7%).
Statins and antiplatelets show largest effect
When analyzed individually and in different combinations, the agents with OMT did not appear to be equal. For example, the biggest survival gap at 10 years was for those who were on an antiplatelet therapy and a statin at 5 years relative to those who were not on either (13.2% vs. 22.6%; P = .006). Even after adjustment, there was nearly 45% survival benefit for these two agents (HR, 0.556; P = .02).
Conversely, the 10-year survival advantage for being on a renin-angiotensin system inhibitor at 5 years versus not being exposed to this therapy was small and nonsignificant (14.7% vs. 13.7%; P = .651).
The precise proportion of patients who were prescribed and adhered to OMT between 5 years and 10 years is unknown, acknowledged the authors, so conclusions are limited about the added benefit of 10- versus 5-year OMT, although the authors presume that a substantial proportion of those adherent for 5 years would likely continue on these therapies.
It can be said with confidence that those adherent for at least 5 years are more likely to be alive at 10 years than those who are not, according to Dr. Boden. He considers these data a call for physicians and all high-risk patients, not just those who have undergone revascularization, to take these standard therapies.
There are plenty of data to “show how poorly we treat patients with OMT,” said Dr. Boden, citing several studies. In one, which looked at OMT in a nationally representative sample in the United States, only a third of patients with angina were taking an antiplatelet, a statin, and a beta-blocker, all of which are indicated.
“Hospitalization for revascularization provides an opportune time to capture the attention of patients and their physicians,” he wrote in his editorial. He called OMT “an imperative to optimize clinical outcomes.”
Many of the investigators involved in the SYNTAXES subanalysis, including Dr. Serruys, have financial relationships with multiple pharmaceutical companies, including Boston Scientific, which provided the initial funding for the SYNTAX trial. Dr. Boden reports no potential conflicts of interest.
When it comes to medical therapy after a coronary revascularization procedure, more is better. Patients started and then maintained indefinitely on more rather than fewer of the drugs identified as optimal medical therapy (OMT) achieve a major survival benefit 10 years later, according to long-term follow-up from an extended analysis of the SYNTAX trial.
For the survival benefit at 10 years, “the present study suggests that at least three types of optimal medical therapy should be maintained for at least 5 years after revascularization,” reported a multinational team of cardiovascular specialists led by Hideyuki Kawashima, MD and Patrick Serruys, MD, who both have affiliations with the department of cardiology of the National University of Ireland, Galway.
The SYNTAX trial was conducted to compare percutaneous coronary intervention (PCI) with coronary artery bypass grafting (CABG) for patients with previously untreated three-vessel and/or left main disease (N Engl J Med 2009;360:961-72). The conclusion from that study, published in 2009 and subsequently reinforced by a 5-year follow-up, was that CABG should remain the standard of care for complex lesions.
Optimal medical therapy defined
In the course of SYNTAX, the impact of OMT on outcome was also evaluated in a subanalysis. At 5 years, there was a mortality advantage for those receiving an antiplatelet drug, a statin, a renin-angiotensin system inhibitor (ACE inhibitor or angiotensin receptor blocker), and a beta-blocker when compared with fewer of these agents.
When an investigator-initiated extension of SYNTAX, called SYNTAXES, was conducted to compare the outcomes of PCI and CABG at 10 years, it also permitted an extended analysis of OMT. Although the primary comparison of SYNTAXES, reported 2 years ago, did not show a significant difference between PCI and CABG for mortality at 10 years, there was a difference for OMT.
When investigators compared treatment with three or more OMT agents with that with two or fewer OMT drugs at 5 years, the result for all-cause death at 10 years translated into a more than 50% relative reduction (hazard ratio, 0.47; P = .002). The absolute difference in mortality was a more than 6% reduction (13.1% vs. 19.9%).
OMT data offer major message
The current study is considered to have a major message for patients as well as physicians.
“OMT even outweighs the survival benefit from revascularization alone, so our patients should convince themselves of the value of rigorous adherence and compliance,” Dr. Serruys said in an interview. According to him, these are compelling data for telling patients that OMT “is the best insurance for extended survival.” We now know from these data “the longer, the better.”
The same message from these data extends to physicians.
“I wish I could understand the apparent blind spot physicians have with respect to prescribing OMT despite the overwhelming benefit from multiple clinical trials,” said William E. Boden, MD, professor of medicine, Boston University.
Dr. Boden was a coauthor of an editorial accompanying the newly published SYNTAXES subanalysis. In the editorial, he noted that OMT following revascularization and in other high-risk patients “has been unacceptably low,” but he was asked to expand on the lessons from the newly released SYNTAXES subanalysis in an interview.
“There has often been a belief that revascularization negates the need for OMT and that’s why the SYNTAXES trial 10-year mortality reduction – which builds upon an earlier 5-year mortality reduction analysis – is so important,” he said.
Patients should take OMT long term
These data “should be both a motivator for physicians to prescribe OMT and for patients to remain adherent to OMT,” he said. “It is the best warranty to blunt the progression of atherosclerosis and to reduce subsequent cardiac events.”
For the 10-year subanalysis of OMT in SYNTAXES, the patients were stratified by the number of OMTs they were taking at 5 years after revascularization and then evaluated for survival at 10 years. Of the 1,472 patients available for analysis at 5 years, only 678 (46%) were on OMT. The other 794 patients were not.
Graphically, the Kaplan-Meier survival curve for those on three types of OMT was consistently beneath that of those on four OMTs, but the gap narrowed over time. At the end of 10 years, the advantage of the four-drug OMT was not statistically significant relative to three or fewer (13.1% vs. 12.7%).
Statins and antiplatelets show largest effect
When analyzed individually and in different combinations, the agents with OMT did not appear to be equal. For example, the biggest survival gap at 10 years was for those who were on an antiplatelet therapy and a statin at 5 years relative to those who were not on either (13.2% vs. 22.6%; P = .006). Even after adjustment, there was nearly 45% survival benefit for these two agents (HR, 0.556; P = .02).
Conversely, the 10-year survival advantage for being on a renin-angiotensin system inhibitor at 5 years versus not being exposed to this therapy was small and nonsignificant (14.7% vs. 13.7%; P = .651).
The precise proportion of patients who were prescribed and adhered to OMT between 5 years and 10 years is unknown, acknowledged the authors, so conclusions are limited about the added benefit of 10- versus 5-year OMT, although the authors presume that a substantial proportion of those adherent for 5 years would likely continue on these therapies.
It can be said with confidence that those adherent for at least 5 years are more likely to be alive at 10 years than those who are not, according to Dr. Boden. He considers these data a call for physicians and all high-risk patients, not just those who have undergone revascularization, to take these standard therapies.
There are plenty of data to “show how poorly we treat patients with OMT,” said Dr. Boden, citing several studies. In one, which looked at OMT in a nationally representative sample in the United States, only a third of patients with angina were taking an antiplatelet, a statin, and a beta-blocker, all of which are indicated.
“Hospitalization for revascularization provides an opportune time to capture the attention of patients and their physicians,” he wrote in his editorial. He called OMT “an imperative to optimize clinical outcomes.”
Many of the investigators involved in the SYNTAXES subanalysis, including Dr. Serruys, have financial relationships with multiple pharmaceutical companies, including Boston Scientific, which provided the initial funding for the SYNTAX trial. Dr. Boden reports no potential conflicts of interest.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Nocturnal hypoglycemia halved with insulin degludec vs. glargine
Patients with type 1 diabetes who used insulin degludec as their basal insulin had fewer than half the number of nocturnal hypoglycemia events, compared with patients who used insulin glargine U100, in a head-to-head crossover study with 51 patients who had a history of nighttime hypoglycemia episodes.
Patients with type 1 diabetes who are “struggling with nocturnal hypoglycemia would benefit from insulin degludec treatment,” said Julie M. Brøsen, MD, at the annual scientific sessions of the American Diabetes Association.
Accumulating evidence for less hypoglycemia with insulin degludec
Results from several studies comparing insulin degludec (Tresiba), a second-generation, longer-acting insulin with more stable steady-state performance, with the first-generation basal insulin analogue glargine (Lantus), have built the case that degludec produces fewer hypoglycemia events.
The landmark SWITCH 1 crossover study published in 2017 showed in about 500 patients with type 1 diabetes and a risk factor for hypoglycemia that treatment with insulin degludec led to significantly few total hypoglycemia episodes and significantly fewer nocturnal episodes, compared with insulin glargine.
Next came similar findings from ReFLeCT, a multicenter observational study that followed 556 unselected patients with type 1 diabetes in routine practice settings who switched to insulin degludec following treatment with a different basal insulin. The results again showed a significant drop-off in total, nonsevere, severe, and nocturnal hypoglycemia events.
Homing in on higher-risk patients
The current study, HypoDeg (Insulin Degludec and Symptomatic Nocturnal Hypoglycaemia), ran at 10 Danish centers and enrolled 149 adults with type 1 diabetes who had at least one episode of severe nocturnal hypoglycemia within the prior 2 years, focusing on patients most at risk for future nocturnal hypoglycemia events. In an unusual study design, researchers identified nocturnal hypoglycemic episodes with hourly venous blood samples drawn from a subcutaneous line.
They randomized the patients to basal insulin treatment with either insulin degludec or to insulin glargine U100, allowed their treatment to stabilize for 3 months, and then tallied nocturnal hypoglycemia events for 9 months. They then crossed patients to the alternative basal insulin and repeated the process.
Results from the full study have not yet appeared in published form but were in a pair of reports at the 2020 scientific sessions of the ADA.
One report included findings based on 136 episodes of severe hypoglycemia identified clinically and showed these events occurred 35% less often during treatment with insulin degludec, a significant difference. The overall finding was primarily driven by 48% fewer episodes of severe nocturnal hypoglycemia, but this difference was not significant.
The second report identified hypoglycemia events with continuous glucose monitoring in 74 of the study participants, which identified 193 episodes of nonsevere nocturnal hypoglycemia and found that treatment with insulin degludec cut the rate by 47%, primarily by reducing asymptomatic episodes.
Hourly blood draws track overnight hypoglycemia
The current study included 51 of the 149 HypoDeg patients who agreed to undergo overnight blood sampling and had this done at least once while treated with each of the two study insulins. (The study design called for two blood sampling nights for each willing patient during each of the two treatment periods.) The 51 patients had type 1 diabetes for an average of 28 years and an average age of 58 years. Two-thirds were men, their baseline A1c was 7.8%, and on average had 2.6 episodes of severe nocturnal hypoglycemia during the prior 2 years.
The researchers drew hourly blood specimens on a total of 196 nights from the 51 participating patients and identified 57 nights when blood glucose levels reached hypoglycemia thresholds in 33 patients. One-third of the events occurred when patients were on insulin degludec treatment, and two-thirds when they were on insulin glargine, reported Dr. Brøsen.
She presented three separate analyses of the data. One analysis focused on level 1 hypoglycemia events, when blood glucose dips to 70 mg/dL or less, which occurred 54% less often when patients were on insulin degludec. A second analysis looked at level 2 events, when blood glucose falls below 54 mg/dL, and treatment with insulin degludec cut this by 64% compared with insulin glargine. The third analysis focused on symptomatic events when blood glucose was 70 mg/dL or less, and treatment with insulin degludec linked with a 62% cut in this metric. All three between-group differences were significant.
Evidence supports already-changed practice
This new evidence “supports recommending” insulin degludec over insulin glargine, commented Bastiaan E. de Galan, MD, PhD, an endocrinologist and professor at Maastrict (the Netherlands) University Medical Center. The new results “extend those from previous trials in populations with type 1 diabetes that were unselected for the risk of hypoglycemia. In clinical practice, insulin degludec is already considered for patients who reported nocturnal hypoglycemia while on insulin glargine U100, but it’s great this study provides the scientific evidence,” said Dr. de Galan in an interview.
“The lower rate of nocturnal hypoglycemia with degludec, compared with glargine U100 is well established. Inpatient assessment of hypoglycemia with measurement of hourly plasma glucose allowed HypoDeg to provide stronger evidence than prior studies. The benefit of delgudec versus glargine U100 was significant and clinically meaningful, in hypo-prone patients who would benefit the most” by using insulin degludec, commented Gian Paolo Fadini, MD, an endocrinologist at the University of Padova (Italy), and a lead investigator on the ReFLeCT study.
But insulin degludec is not a completely silver bullet. Its prolonged duration of action and stability that may in part explain why it limits hypoglycemia events can also be a drawback: “It probably offers fewer options for flexibility. Any change in dose takes at least a day or 2 to settle, which may be unfavorable in certain circumstances,” noted Dr. de Galan.
“I wouldn’t recommend insulin degludec for all patients with type 1 diabetes. It’s an individual evaluation in each patient,” said Dr. Brøsen. “We will be looking into whether some patients are better off on insulin glargine.”
Cost makes a difference
Another, potentially more consequential flaw is insulin degludec’s relative expense.
“To date, use of degludec in routine practice has been limited by its cost, compared with older basal insulins,” observed Dr. Fadini in an interview. “In several countries, including the United States, degludec is substantially more expensive than glargine.”
The ADA’s Standards of Medical Care in Diabetes–2021 includes table 9.3 that lists the costs of various insulins and shows the median average wholesale price of insulin glargine U100 follow-on products as $190/vial, compared with a $407 price for a similar vial of insulin degludec.
Insulin degludec “is clearly superior from a hypoglycemia standpoint. Patients with type 1 diabetes like the reduction because hypoglycemia is scary, and dangerous. The main issue is cost, and the extent to which it may be covered by insurance,” commented Lisa Chow, MD, an endocrinologist at the University of Minnesota, Minneapolis. “We generally won’t prescribe degludec unless it is at a price affordable to the patient. We try to use patient assistance programs sponsored by the company [that markets insulin degludec: Novo Nordisk] to try to make it more affordable.”
Dr. Chow also highlighted that a new wrinkle has been introduction of a more concentrated formulation of insulin glargine, U300, which appears to cause less hypoglycemia than insulin glargine U100. Recent study results indicated that no significant difference exists in the incidence of hypoglycemia among patients treated with insulin glargine U300 and those treated with insulin degludec, such as findings from the BRIGHT trial, which included just over 900 patients, and in the CONCLUDE trial, which randomized more than 1,600 patients.
The HypoDeg study was sponsored by Novo Nordisk, the company that markets insulin degludec. Dr. Brøsen had no personal disclosures, but several of her coauthors were either Novo Nordisk employees or had financial relationships with the company. Dr. de Galan has received research funding from Novo Nordisk. Dr. Fadini has received lecture fees and research funding from Novo Nordisk, from Sanofi, the company that markets insulin glargine, and from several other companies. Dr. Chow has received research funding from Dexcom.
Patients with type 1 diabetes who used insulin degludec as their basal insulin had fewer than half the number of nocturnal hypoglycemia events, compared with patients who used insulin glargine U100, in a head-to-head crossover study with 51 patients who had a history of nighttime hypoglycemia episodes.
Patients with type 1 diabetes who are “struggling with nocturnal hypoglycemia would benefit from insulin degludec treatment,” said Julie M. Brøsen, MD, at the annual scientific sessions of the American Diabetes Association.
Accumulating evidence for less hypoglycemia with insulin degludec
Results from several studies comparing insulin degludec (Tresiba), a second-generation, longer-acting insulin with more stable steady-state performance, with the first-generation basal insulin analogue glargine (Lantus), have built the case that degludec produces fewer hypoglycemia events.
The landmark SWITCH 1 crossover study published in 2017 showed in about 500 patients with type 1 diabetes and a risk factor for hypoglycemia that treatment with insulin degludec led to significantly few total hypoglycemia episodes and significantly fewer nocturnal episodes, compared with insulin glargine.
Next came similar findings from ReFLeCT, a multicenter observational study that followed 556 unselected patients with type 1 diabetes in routine practice settings who switched to insulin degludec following treatment with a different basal insulin. The results again showed a significant drop-off in total, nonsevere, severe, and nocturnal hypoglycemia events.
Homing in on higher-risk patients
The current study, HypoDeg (Insulin Degludec and Symptomatic Nocturnal Hypoglycaemia), ran at 10 Danish centers and enrolled 149 adults with type 1 diabetes who had at least one episode of severe nocturnal hypoglycemia within the prior 2 years, focusing on patients most at risk for future nocturnal hypoglycemia events. In an unusual study design, researchers identified nocturnal hypoglycemic episodes with hourly venous blood samples drawn from a subcutaneous line.
They randomized the patients to basal insulin treatment with either insulin degludec or to insulin glargine U100, allowed their treatment to stabilize for 3 months, and then tallied nocturnal hypoglycemia events for 9 months. They then crossed patients to the alternative basal insulin and repeated the process.
Results from the full study have not yet appeared in published form but were in a pair of reports at the 2020 scientific sessions of the ADA.
One report included findings based on 136 episodes of severe hypoglycemia identified clinically and showed these events occurred 35% less often during treatment with insulin degludec, a significant difference. The overall finding was primarily driven by 48% fewer episodes of severe nocturnal hypoglycemia, but this difference was not significant.
The second report identified hypoglycemia events with continuous glucose monitoring in 74 of the study participants, which identified 193 episodes of nonsevere nocturnal hypoglycemia and found that treatment with insulin degludec cut the rate by 47%, primarily by reducing asymptomatic episodes.
Hourly blood draws track overnight hypoglycemia
The current study included 51 of the 149 HypoDeg patients who agreed to undergo overnight blood sampling and had this done at least once while treated with each of the two study insulins. (The study design called for two blood sampling nights for each willing patient during each of the two treatment periods.) The 51 patients had type 1 diabetes for an average of 28 years and an average age of 58 years. Two-thirds were men, their baseline A1c was 7.8%, and on average had 2.6 episodes of severe nocturnal hypoglycemia during the prior 2 years.
The researchers drew hourly blood specimens on a total of 196 nights from the 51 participating patients and identified 57 nights when blood glucose levels reached hypoglycemia thresholds in 33 patients. One-third of the events occurred when patients were on insulin degludec treatment, and two-thirds when they were on insulin glargine, reported Dr. Brøsen.
She presented three separate analyses of the data. One analysis focused on level 1 hypoglycemia events, when blood glucose dips to 70 mg/dL or less, which occurred 54% less often when patients were on insulin degludec. A second analysis looked at level 2 events, when blood glucose falls below 54 mg/dL, and treatment with insulin degludec cut this by 64% compared with insulin glargine. The third analysis focused on symptomatic events when blood glucose was 70 mg/dL or less, and treatment with insulin degludec linked with a 62% cut in this metric. All three between-group differences were significant.
Evidence supports already-changed practice
This new evidence “supports recommending” insulin degludec over insulin glargine, commented Bastiaan E. de Galan, MD, PhD, an endocrinologist and professor at Maastrict (the Netherlands) University Medical Center. The new results “extend those from previous trials in populations with type 1 diabetes that were unselected for the risk of hypoglycemia. In clinical practice, insulin degludec is already considered for patients who reported nocturnal hypoglycemia while on insulin glargine U100, but it’s great this study provides the scientific evidence,” said Dr. de Galan in an interview.
“The lower rate of nocturnal hypoglycemia with degludec, compared with glargine U100 is well established. Inpatient assessment of hypoglycemia with measurement of hourly plasma glucose allowed HypoDeg to provide stronger evidence than prior studies. The benefit of delgudec versus glargine U100 was significant and clinically meaningful, in hypo-prone patients who would benefit the most” by using insulin degludec, commented Gian Paolo Fadini, MD, an endocrinologist at the University of Padova (Italy), and a lead investigator on the ReFLeCT study.
But insulin degludec is not a completely silver bullet. Its prolonged duration of action and stability that may in part explain why it limits hypoglycemia events can also be a drawback: “It probably offers fewer options for flexibility. Any change in dose takes at least a day or 2 to settle, which may be unfavorable in certain circumstances,” noted Dr. de Galan.
“I wouldn’t recommend insulin degludec for all patients with type 1 diabetes. It’s an individual evaluation in each patient,” said Dr. Brøsen. “We will be looking into whether some patients are better off on insulin glargine.”
Cost makes a difference
Another, potentially more consequential flaw is insulin degludec’s relative expense.
“To date, use of degludec in routine practice has been limited by its cost, compared with older basal insulins,” observed Dr. Fadini in an interview. “In several countries, including the United States, degludec is substantially more expensive than glargine.”
The ADA’s Standards of Medical Care in Diabetes–2021 includes table 9.3 that lists the costs of various insulins and shows the median average wholesale price of insulin glargine U100 follow-on products as $190/vial, compared with a $407 price for a similar vial of insulin degludec.
Insulin degludec “is clearly superior from a hypoglycemia standpoint. Patients with type 1 diabetes like the reduction because hypoglycemia is scary, and dangerous. The main issue is cost, and the extent to which it may be covered by insurance,” commented Lisa Chow, MD, an endocrinologist at the University of Minnesota, Minneapolis. “We generally won’t prescribe degludec unless it is at a price affordable to the patient. We try to use patient assistance programs sponsored by the company [that markets insulin degludec: Novo Nordisk] to try to make it more affordable.”
Dr. Chow also highlighted that a new wrinkle has been introduction of a more concentrated formulation of insulin glargine, U300, which appears to cause less hypoglycemia than insulin glargine U100. Recent study results indicated that no significant difference exists in the incidence of hypoglycemia among patients treated with insulin glargine U300 and those treated with insulin degludec, such as findings from the BRIGHT trial, which included just over 900 patients, and in the CONCLUDE trial, which randomized more than 1,600 patients.
The HypoDeg study was sponsored by Novo Nordisk, the company that markets insulin degludec. Dr. Brøsen had no personal disclosures, but several of her coauthors were either Novo Nordisk employees or had financial relationships with the company. Dr. de Galan has received research funding from Novo Nordisk. Dr. Fadini has received lecture fees and research funding from Novo Nordisk, from Sanofi, the company that markets insulin glargine, and from several other companies. Dr. Chow has received research funding from Dexcom.
Patients with type 1 diabetes who used insulin degludec as their basal insulin had fewer than half the number of nocturnal hypoglycemia events, compared with patients who used insulin glargine U100, in a head-to-head crossover study with 51 patients who had a history of nighttime hypoglycemia episodes.
Patients with type 1 diabetes who are “struggling with nocturnal hypoglycemia would benefit from insulin degludec treatment,” said Julie M. Brøsen, MD, at the annual scientific sessions of the American Diabetes Association.
Accumulating evidence for less hypoglycemia with insulin degludec
Results from several studies comparing insulin degludec (Tresiba), a second-generation, longer-acting insulin with more stable steady-state performance, with the first-generation basal insulin analogue glargine (Lantus), have built the case that degludec produces fewer hypoglycemia events.
The landmark SWITCH 1 crossover study published in 2017 showed in about 500 patients with type 1 diabetes and a risk factor for hypoglycemia that treatment with insulin degludec led to significantly few total hypoglycemia episodes and significantly fewer nocturnal episodes, compared with insulin glargine.
Next came similar findings from ReFLeCT, a multicenter observational study that followed 556 unselected patients with type 1 diabetes in routine practice settings who switched to insulin degludec following treatment with a different basal insulin. The results again showed a significant drop-off in total, nonsevere, severe, and nocturnal hypoglycemia events.
Homing in on higher-risk patients
The current study, HypoDeg (Insulin Degludec and Symptomatic Nocturnal Hypoglycaemia), ran at 10 Danish centers and enrolled 149 adults with type 1 diabetes who had at least one episode of severe nocturnal hypoglycemia within the prior 2 years, focusing on patients most at risk for future nocturnal hypoglycemia events. In an unusual study design, researchers identified nocturnal hypoglycemic episodes with hourly venous blood samples drawn from a subcutaneous line.
They randomized the patients to basal insulin treatment with either insulin degludec or to insulin glargine U100, allowed their treatment to stabilize for 3 months, and then tallied nocturnal hypoglycemia events for 9 months. They then crossed patients to the alternative basal insulin and repeated the process.
Results from the full study have not yet appeared in published form but were in a pair of reports at the 2020 scientific sessions of the ADA.
One report included findings based on 136 episodes of severe hypoglycemia identified clinically and showed these events occurred 35% less often during treatment with insulin degludec, a significant difference. The overall finding was primarily driven by 48% fewer episodes of severe nocturnal hypoglycemia, but this difference was not significant.
The second report identified hypoglycemia events with continuous glucose monitoring in 74 of the study participants, which identified 193 episodes of nonsevere nocturnal hypoglycemia and found that treatment with insulin degludec cut the rate by 47%, primarily by reducing asymptomatic episodes.
Hourly blood draws track overnight hypoglycemia
The current study included 51 of the 149 HypoDeg patients who agreed to undergo overnight blood sampling and had this done at least once while treated with each of the two study insulins. (The study design called for two blood sampling nights for each willing patient during each of the two treatment periods.) The 51 patients had type 1 diabetes for an average of 28 years and an average age of 58 years. Two-thirds were men, their baseline A1c was 7.8%, and on average had 2.6 episodes of severe nocturnal hypoglycemia during the prior 2 years.
The researchers drew hourly blood specimens on a total of 196 nights from the 51 participating patients and identified 57 nights when blood glucose levels reached hypoglycemia thresholds in 33 patients. One-third of the events occurred when patients were on insulin degludec treatment, and two-thirds when they were on insulin glargine, reported Dr. Brøsen.
She presented three separate analyses of the data. One analysis focused on level 1 hypoglycemia events, when blood glucose dips to 70 mg/dL or less, which occurred 54% less often when patients were on insulin degludec. A second analysis looked at level 2 events, when blood glucose falls below 54 mg/dL, and treatment with insulin degludec cut this by 64% compared with insulin glargine. The third analysis focused on symptomatic events when blood glucose was 70 mg/dL or less, and treatment with insulin degludec linked with a 62% cut in this metric. All three between-group differences were significant.
Evidence supports already-changed practice
This new evidence “supports recommending” insulin degludec over insulin glargine, commented Bastiaan E. de Galan, MD, PhD, an endocrinologist and professor at Maastrict (the Netherlands) University Medical Center. The new results “extend those from previous trials in populations with type 1 diabetes that were unselected for the risk of hypoglycemia. In clinical practice, insulin degludec is already considered for patients who reported nocturnal hypoglycemia while on insulin glargine U100, but it’s great this study provides the scientific evidence,” said Dr. de Galan in an interview.
“The lower rate of nocturnal hypoglycemia with degludec, compared with glargine U100 is well established. Inpatient assessment of hypoglycemia with measurement of hourly plasma glucose allowed HypoDeg to provide stronger evidence than prior studies. The benefit of delgudec versus glargine U100 was significant and clinically meaningful, in hypo-prone patients who would benefit the most” by using insulin degludec, commented Gian Paolo Fadini, MD, an endocrinologist at the University of Padova (Italy), and a lead investigator on the ReFLeCT study.
But insulin degludec is not a completely silver bullet. Its prolonged duration of action and stability that may in part explain why it limits hypoglycemia events can also be a drawback: “It probably offers fewer options for flexibility. Any change in dose takes at least a day or 2 to settle, which may be unfavorable in certain circumstances,” noted Dr. de Galan.
“I wouldn’t recommend insulin degludec for all patients with type 1 diabetes. It’s an individual evaluation in each patient,” said Dr. Brøsen. “We will be looking into whether some patients are better off on insulin glargine.”
Cost makes a difference
Another, potentially more consequential flaw is insulin degludec’s relative expense.
“To date, use of degludec in routine practice has been limited by its cost, compared with older basal insulins,” observed Dr. Fadini in an interview. “In several countries, including the United States, degludec is substantially more expensive than glargine.”
The ADA’s Standards of Medical Care in Diabetes–2021 includes table 9.3 that lists the costs of various insulins and shows the median average wholesale price of insulin glargine U100 follow-on products as $190/vial, compared with a $407 price for a similar vial of insulin degludec.
Insulin degludec “is clearly superior from a hypoglycemia standpoint. Patients with type 1 diabetes like the reduction because hypoglycemia is scary, and dangerous. The main issue is cost, and the extent to which it may be covered by insurance,” commented Lisa Chow, MD, an endocrinologist at the University of Minnesota, Minneapolis. “We generally won’t prescribe degludec unless it is at a price affordable to the patient. We try to use patient assistance programs sponsored by the company [that markets insulin degludec: Novo Nordisk] to try to make it more affordable.”
Dr. Chow also highlighted that a new wrinkle has been introduction of a more concentrated formulation of insulin glargine, U300, which appears to cause less hypoglycemia than insulin glargine U100. Recent study results indicated that no significant difference exists in the incidence of hypoglycemia among patients treated with insulin glargine U300 and those treated with insulin degludec, such as findings from the BRIGHT trial, which included just over 900 patients, and in the CONCLUDE trial, which randomized more than 1,600 patients.
The HypoDeg study was sponsored by Novo Nordisk, the company that markets insulin degludec. Dr. Brøsen had no personal disclosures, but several of her coauthors were either Novo Nordisk employees or had financial relationships with the company. Dr. de Galan has received research funding from Novo Nordisk. Dr. Fadini has received lecture fees and research funding from Novo Nordisk, from Sanofi, the company that markets insulin glargine, and from several other companies. Dr. Chow has received research funding from Dexcom.
FROM ADA 2021
Cannabis use tied to increased risk for suicidal thoughts, actions
Young adults who use cannabis – either sporadically, daily, or those who have cannabis use disorder – have a significantly increased risk for suicidal thoughts and actions, according to U.S. national drug survey data.
The risks appear greater for women than men and remained regardless of whether the individual was depressed.
“We cannot establish that cannabis use caused increased suicidality,” Nora Volkow, MD, director, National Institute on Drug Abuse (NIDA), told this news organization.
“However, it is likely that these two factors influence one another bidirectionally, meaning people with suicidal thinking might be more vulnerable to cannabis use to self-medicate their distress, and cannabis use may trigger negative moods and suicidal thinking in some people,” said Dr. Volkow.
“It is also possible that these factors are not causally linked to one another at all but rather reflect the common and related risk factors underlying both suicidality and substance use. For instance, one’s genetics may put them at a higher risk for both suicide and for using marijuana,” she added.
The study was published online June 22 in JAMA Network Open.
Marked increase in use
Cannabis use among U.S. adults has increased markedly over the past 10 years, with a parallel increase in suicidality. However, the links between cannabis use and suicidality among young adults are poorly understood.
NIDA researchers sought to fill this gap. They examined data on 281,650 young men and women aged 18 to 34 years who participated in National Surveys on Drug Use and Health from 2008 to 2019.
Status regarding past-year cannabis use was categorized as past-year daily or near-daily use (greater than or equal to 300 days), non-daily use, and no cannabis use.
Although suicidality was associated with cannabis use, even young adults who did not use cannabis on a daily basis were more likely to have suicidal thoughts or actions than those who did not use the drug at all, the researchers found.
Among young adults without a major depressive episode, about 3% of those who did not use cannabis had suicidal ideation, compared with about 7% of non-daily cannabis users, about 9% of daily cannabis users, and 14% of those with a cannabis use disorder.
Among young adults with depression, the corresponding percentages were 35%, 44%, 53%, and 50%.
Similar trends existed for the associations between the different levels of cannabis use and suicide plan or attempt.
Women at greatest risk
Gender differences also emerged. than men with the same levels of cannabis use.
Among those without a major depressive episode, the prevalence of suicidal ideation for those with versus without a cannabis use disorder was around 14% versus 4.0% among women and 10% versus 3.0% among men.
Among young adults with both cannabis use disorder and major depressive episode, the prevalence of past-year suicide plan was 52% higher for women (24%) than for men (16%).
“Suicide is a leading cause of death among young adults in the United States, and the findings of this study offer important information that may help us reduce this risk,” lead author and NIDA researcher Beth Han, MD, PhD, MPH, said in a news release.
“Depression and cannabis use disorder are treatable conditions, and cannabis use can be modified. Through better understanding the associations of different risk factors for suicidality, we hope to offer new targets for prevention and intervention in individuals that we know may be at high risk. These findings also underscore the importance of tailoring interventions in a way that takes sex and gender into account,” said Dr. Han.
“Additional research is needed to better understand these complex associations, especially given the great burden of suicide on young adults,” said Dr. Volkow.
Gender difference ‘striking’
Commenting on the findings for this news organization, Charles B. Nemeroff, MD, PhD, professor and chair, department of psychiatry and behavioral sciences, Dell Medical School, University of Texas at Austin, said this study is “clearly of great interest; of course correlation and causality are completely distinct entities, and this study is all about correlation.
“This does not, of course, mean that cannabis use causes suicide but suggests that in individuals who use cannabis, suicidality in the broadest sense is increased in prevalence rate,” said Dr. Nemeroff, who serves as principal investigator of the Texas Child Trauma Network.
Dr. Nemeroff said “the most striking finding” was the larger effect in women than men – “striking because suicide is, in almost all cultures, higher in prevalence in men versus women.”
Dr. Nemeroff said he’d like to know more about other potential contributing factors, “which would include a history of child abuse and neglect, a major vulnerability factor for suicidality, comorbid alcohol and other substance abuse, [and] comorbid psychiatric diagnosis such as posttraumatic stress disorder.”
The study was sponsored by NIDA, of the National Institutes of Health. Dr. Volkow, Dr. Han, and Dr. Nemeroff have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Young adults who use cannabis – either sporadically, daily, or those who have cannabis use disorder – have a significantly increased risk for suicidal thoughts and actions, according to U.S. national drug survey data.
The risks appear greater for women than men and remained regardless of whether the individual was depressed.
“We cannot establish that cannabis use caused increased suicidality,” Nora Volkow, MD, director, National Institute on Drug Abuse (NIDA), told this news organization.
“However, it is likely that these two factors influence one another bidirectionally, meaning people with suicidal thinking might be more vulnerable to cannabis use to self-medicate their distress, and cannabis use may trigger negative moods and suicidal thinking in some people,” said Dr. Volkow.
“It is also possible that these factors are not causally linked to one another at all but rather reflect the common and related risk factors underlying both suicidality and substance use. For instance, one’s genetics may put them at a higher risk for both suicide and for using marijuana,” she added.
The study was published online June 22 in JAMA Network Open.
Marked increase in use
Cannabis use among U.S. adults has increased markedly over the past 10 years, with a parallel increase in suicidality. However, the links between cannabis use and suicidality among young adults are poorly understood.
NIDA researchers sought to fill this gap. They examined data on 281,650 young men and women aged 18 to 34 years who participated in National Surveys on Drug Use and Health from 2008 to 2019.
Status regarding past-year cannabis use was categorized as past-year daily or near-daily use (greater than or equal to 300 days), non-daily use, and no cannabis use.
Although suicidality was associated with cannabis use, even young adults who did not use cannabis on a daily basis were more likely to have suicidal thoughts or actions than those who did not use the drug at all, the researchers found.
Among young adults without a major depressive episode, about 3% of those who did not use cannabis had suicidal ideation, compared with about 7% of non-daily cannabis users, about 9% of daily cannabis users, and 14% of those with a cannabis use disorder.
Among young adults with depression, the corresponding percentages were 35%, 44%, 53%, and 50%.
Similar trends existed for the associations between the different levels of cannabis use and suicide plan or attempt.
Women at greatest risk
Gender differences also emerged. than men with the same levels of cannabis use.
Among those without a major depressive episode, the prevalence of suicidal ideation for those with versus without a cannabis use disorder was around 14% versus 4.0% among women and 10% versus 3.0% among men.
Among young adults with both cannabis use disorder and major depressive episode, the prevalence of past-year suicide plan was 52% higher for women (24%) than for men (16%).
“Suicide is a leading cause of death among young adults in the United States, and the findings of this study offer important information that may help us reduce this risk,” lead author and NIDA researcher Beth Han, MD, PhD, MPH, said in a news release.
“Depression and cannabis use disorder are treatable conditions, and cannabis use can be modified. Through better understanding the associations of different risk factors for suicidality, we hope to offer new targets for prevention and intervention in individuals that we know may be at high risk. These findings also underscore the importance of tailoring interventions in a way that takes sex and gender into account,” said Dr. Han.
“Additional research is needed to better understand these complex associations, especially given the great burden of suicide on young adults,” said Dr. Volkow.
Gender difference ‘striking’
Commenting on the findings for this news organization, Charles B. Nemeroff, MD, PhD, professor and chair, department of psychiatry and behavioral sciences, Dell Medical School, University of Texas at Austin, said this study is “clearly of great interest; of course correlation and causality are completely distinct entities, and this study is all about correlation.
“This does not, of course, mean that cannabis use causes suicide but suggests that in individuals who use cannabis, suicidality in the broadest sense is increased in prevalence rate,” said Dr. Nemeroff, who serves as principal investigator of the Texas Child Trauma Network.
Dr. Nemeroff said “the most striking finding” was the larger effect in women than men – “striking because suicide is, in almost all cultures, higher in prevalence in men versus women.”
Dr. Nemeroff said he’d like to know more about other potential contributing factors, “which would include a history of child abuse and neglect, a major vulnerability factor for suicidality, comorbid alcohol and other substance abuse, [and] comorbid psychiatric diagnosis such as posttraumatic stress disorder.”
The study was sponsored by NIDA, of the National Institutes of Health. Dr. Volkow, Dr. Han, and Dr. Nemeroff have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Young adults who use cannabis – either sporadically, daily, or those who have cannabis use disorder – have a significantly increased risk for suicidal thoughts and actions, according to U.S. national drug survey data.
The risks appear greater for women than men and remained regardless of whether the individual was depressed.
“We cannot establish that cannabis use caused increased suicidality,” Nora Volkow, MD, director, National Institute on Drug Abuse (NIDA), told this news organization.
“However, it is likely that these two factors influence one another bidirectionally, meaning people with suicidal thinking might be more vulnerable to cannabis use to self-medicate their distress, and cannabis use may trigger negative moods and suicidal thinking in some people,” said Dr. Volkow.
“It is also possible that these factors are not causally linked to one another at all but rather reflect the common and related risk factors underlying both suicidality and substance use. For instance, one’s genetics may put them at a higher risk for both suicide and for using marijuana,” she added.
The study was published online June 22 in JAMA Network Open.
Marked increase in use
Cannabis use among U.S. adults has increased markedly over the past 10 years, with a parallel increase in suicidality. However, the links between cannabis use and suicidality among young adults are poorly understood.
NIDA researchers sought to fill this gap. They examined data on 281,650 young men and women aged 18 to 34 years who participated in National Surveys on Drug Use and Health from 2008 to 2019.
Status regarding past-year cannabis use was categorized as past-year daily or near-daily use (greater than or equal to 300 days), non-daily use, and no cannabis use.
Although suicidality was associated with cannabis use, even young adults who did not use cannabis on a daily basis were more likely to have suicidal thoughts or actions than those who did not use the drug at all, the researchers found.
Among young adults without a major depressive episode, about 3% of those who did not use cannabis had suicidal ideation, compared with about 7% of non-daily cannabis users, about 9% of daily cannabis users, and 14% of those with a cannabis use disorder.
Among young adults with depression, the corresponding percentages were 35%, 44%, 53%, and 50%.
Similar trends existed for the associations between the different levels of cannabis use and suicide plan or attempt.
Women at greatest risk
Gender differences also emerged. than men with the same levels of cannabis use.
Among those without a major depressive episode, the prevalence of suicidal ideation for those with versus without a cannabis use disorder was around 14% versus 4.0% among women and 10% versus 3.0% among men.
Among young adults with both cannabis use disorder and major depressive episode, the prevalence of past-year suicide plan was 52% higher for women (24%) than for men (16%).
“Suicide is a leading cause of death among young adults in the United States, and the findings of this study offer important information that may help us reduce this risk,” lead author and NIDA researcher Beth Han, MD, PhD, MPH, said in a news release.
“Depression and cannabis use disorder are treatable conditions, and cannabis use can be modified. Through better understanding the associations of different risk factors for suicidality, we hope to offer new targets for prevention and intervention in individuals that we know may be at high risk. These findings also underscore the importance of tailoring interventions in a way that takes sex and gender into account,” said Dr. Han.
“Additional research is needed to better understand these complex associations, especially given the great burden of suicide on young adults,” said Dr. Volkow.
Gender difference ‘striking’
Commenting on the findings for this news organization, Charles B. Nemeroff, MD, PhD, professor and chair, department of psychiatry and behavioral sciences, Dell Medical School, University of Texas at Austin, said this study is “clearly of great interest; of course correlation and causality are completely distinct entities, and this study is all about correlation.
“This does not, of course, mean that cannabis use causes suicide but suggests that in individuals who use cannabis, suicidality in the broadest sense is increased in prevalence rate,” said Dr. Nemeroff, who serves as principal investigator of the Texas Child Trauma Network.
Dr. Nemeroff said “the most striking finding” was the larger effect in women than men – “striking because suicide is, in almost all cultures, higher in prevalence in men versus women.”
Dr. Nemeroff said he’d like to know more about other potential contributing factors, “which would include a history of child abuse and neglect, a major vulnerability factor for suicidality, comorbid alcohol and other substance abuse, [and] comorbid psychiatric diagnosis such as posttraumatic stress disorder.”
The study was sponsored by NIDA, of the National Institutes of Health. Dr. Volkow, Dr. Han, and Dr. Nemeroff have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.