User login
Formerly Skin & Allergy News
ass lick
assault rifle
balls
ballsac
black jack
bleach
Boko Haram
bondage
causas
cheap
child abuse
cocaine
compulsive behaviors
cost of miracles
cunt
Daech
display network stats
drug paraphernalia
explosion
fart
fda and death
fda AND warn
fda AND warning
fda AND warns
feom
fuck
gambling
gfc
gun
human trafficking
humira AND expensive
illegal
ISIL
ISIS
Islamic caliphate
Islamic state
madvocate
masturbation
mixed martial arts
MMA
molestation
national rifle association
NRA
nsfw
nuccitelli
pedophile
pedophilia
poker
porn
porn
pornography
psychedelic drug
recreational drug
sex slave rings
shit
slot machine
snort
substance abuse
terrorism
terrorist
texarkana
Texas hold 'em
UFC
section[contains(@class, 'nav-hidden')]
section[contains(@class, 'nav-hidden active')]
The leading independent newspaper covering dermatology news and commentary.
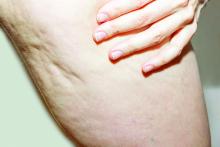
A glimpse at devices designed to tackle cellulite
SAN DIEGO –
A noninvasive treatment, rapid acoustic pulse (RAP) technology (RESONIC), was cleared by the Food and Drug Administration in 2021 for short-term improvement in the appearance of cellulite. The device emits rapid acoustic pulses (shock waves) that are transmitted through the skin to rupture or shear the fibrotic septa; release of the septa results in the smoothing of skin dimples, Arisa E. Ortiz, MD, director of laser and cosmetic dermatology at the University of California, San Diego, said at the annual Masters of Aesthetics Symposium. RAP, however, was “taken off the market temporarily to refine the design.”
According to Dr. Ortiz, RAP’s repetition rate and short rise time provides microscopic mechanical disruption to the targeted cellular level structures and vacuoles, while high peak pressure and the fast repetition rate exploit the viscoelastic nature of tissue. Compressed pulses from electronic filtering and the reflector shape eliminate cavitation, heat, and pain. Researchers have postulated that the procedure stimulates collagenesis and angiogenesis.
“There’s no heat to this; it just uses sound,” Dr. Ortiz explained. “It’s also time dependent. The longer you do the treatment, the more disruption of the septa you see. The procedure takes 20-30 minutes. Unlike other treatments, it’s not just for discreet dimples. You can treat entire areas like the buttock or posterior leg,” said Dr. Ortiz, who did not use the RAP device in her practice.
Another device, targeted verifiable subcision (TVS, marketed as Avéli), is FDA cleared for temporary reduction in the appearance of cellulite in the buttock and thigh areas of adult women. “But studies show lasting results, at least through a year,” Dr. Ortiz said. The device features a light-guided probe and a hook. The light enables clinicians to navigate under the skin, while the hook releases a tiny blade that severs the septa. “Once you find the septa, then you activate the blade and release the septa. You go right to left because the direction of the blade is on the left of the probe. Then you go back to verify that you got everything that was creating that dimple.”
Previous devices, she said, would “blindly shear the area, so you would find the dimple and blindly cut, so there was no way to verify that you got the target dimple. The results were sometimes mediocre because you didn’t really know if you effectively treated the area.”
She emphasized that TVS is only useful for discreet dimples. “Many patients who come in asking for cellulite treatment have a lot of laxity and rippled texture,” said Dr. Ortiz, who is also president-elect of the American Society for Laser Medicine and Surgery. “This is not going to be appropriate for those cases. Setting expectations is important. If patients have laxity and discreet dimples and it’s just the dimples that bother them, that’s fine. They just need to understand the difference,” she said, noting that this is “safe for all skin types.”
Tumescent anesthesia is used to control pain during the procedure. The most common adverse events are bruising and soreness. Results from a pivotal trial showed that clinically significant improvements in the primary endpoint, Cellulite Severity Scale scores, were sustained 1 year after treatment. “Hemosiderin staining can occur, but it eventually dissipates on its own,” Dr. Ortiz added. “You can use laser to speed up healing but sometimes that can make it worse, so you want to be careful with that. Most of the time I have patients wait it out; it does go away on its own.”
She noted that the development of RAP and TVS have helped clinicians better understand the makeup of septa. “We used to think of septa as singular bands that are vertically oriented in cellulite, but what we’ve realized is that it’s more like a network of septa,” she said.
Another noninvasive technology, synchronous parallel ultrasound beam technology from Sofwave (marketed as SUPERB), was FDA cleared in December 2022 for the short-term improvement in the appearance of cellulite. The device has seven parallel beam transducers that increase tissue temperatures of the treatment area to 60-70° C, inducing collagen remodeling and collagen denaturation, she said.
In the pivotal study of 68 women, two blinded reviewers reported an 89% improvement rate for both cellulite and skin laxity, after two treatments 2-4 weeks apart, according to data she presented at the meeting. The mean pain score during treatment was 4.55 on a scale of 1-10. No safety issues were observed and immediate responses were limited to erythema and edema.
Dr. Ortiz disclosed having financial relationships with several pharmaceutical and device companies, including receipt of speaker fees and honoraria from Sofwave. She is also cochair of MOAS.
SAN DIEGO –
A noninvasive treatment, rapid acoustic pulse (RAP) technology (RESONIC), was cleared by the Food and Drug Administration in 2021 for short-term improvement in the appearance of cellulite. The device emits rapid acoustic pulses (shock waves) that are transmitted through the skin to rupture or shear the fibrotic septa; release of the septa results in the smoothing of skin dimples, Arisa E. Ortiz, MD, director of laser and cosmetic dermatology at the University of California, San Diego, said at the annual Masters of Aesthetics Symposium. RAP, however, was “taken off the market temporarily to refine the design.”
According to Dr. Ortiz, RAP’s repetition rate and short rise time provides microscopic mechanical disruption to the targeted cellular level structures and vacuoles, while high peak pressure and the fast repetition rate exploit the viscoelastic nature of tissue. Compressed pulses from electronic filtering and the reflector shape eliminate cavitation, heat, and pain. Researchers have postulated that the procedure stimulates collagenesis and angiogenesis.
“There’s no heat to this; it just uses sound,” Dr. Ortiz explained. “It’s also time dependent. The longer you do the treatment, the more disruption of the septa you see. The procedure takes 20-30 minutes. Unlike other treatments, it’s not just for discreet dimples. You can treat entire areas like the buttock or posterior leg,” said Dr. Ortiz, who did not use the RAP device in her practice.
Another device, targeted verifiable subcision (TVS, marketed as Avéli), is FDA cleared for temporary reduction in the appearance of cellulite in the buttock and thigh areas of adult women. “But studies show lasting results, at least through a year,” Dr. Ortiz said. The device features a light-guided probe and a hook. The light enables clinicians to navigate under the skin, while the hook releases a tiny blade that severs the septa. “Once you find the septa, then you activate the blade and release the septa. You go right to left because the direction of the blade is on the left of the probe. Then you go back to verify that you got everything that was creating that dimple.”
Previous devices, she said, would “blindly shear the area, so you would find the dimple and blindly cut, so there was no way to verify that you got the target dimple. The results were sometimes mediocre because you didn’t really know if you effectively treated the area.”
She emphasized that TVS is only useful for discreet dimples. “Many patients who come in asking for cellulite treatment have a lot of laxity and rippled texture,” said Dr. Ortiz, who is also president-elect of the American Society for Laser Medicine and Surgery. “This is not going to be appropriate for those cases. Setting expectations is important. If patients have laxity and discreet dimples and it’s just the dimples that bother them, that’s fine. They just need to understand the difference,” she said, noting that this is “safe for all skin types.”
Tumescent anesthesia is used to control pain during the procedure. The most common adverse events are bruising and soreness. Results from a pivotal trial showed that clinically significant improvements in the primary endpoint, Cellulite Severity Scale scores, were sustained 1 year after treatment. “Hemosiderin staining can occur, but it eventually dissipates on its own,” Dr. Ortiz added. “You can use laser to speed up healing but sometimes that can make it worse, so you want to be careful with that. Most of the time I have patients wait it out; it does go away on its own.”
She noted that the development of RAP and TVS have helped clinicians better understand the makeup of septa. “We used to think of septa as singular bands that are vertically oriented in cellulite, but what we’ve realized is that it’s more like a network of septa,” she said.
Another noninvasive technology, synchronous parallel ultrasound beam technology from Sofwave (marketed as SUPERB), was FDA cleared in December 2022 for the short-term improvement in the appearance of cellulite. The device has seven parallel beam transducers that increase tissue temperatures of the treatment area to 60-70° C, inducing collagen remodeling and collagen denaturation, she said.
In the pivotal study of 68 women, two blinded reviewers reported an 89% improvement rate for both cellulite and skin laxity, after two treatments 2-4 weeks apart, according to data she presented at the meeting. The mean pain score during treatment was 4.55 on a scale of 1-10. No safety issues were observed and immediate responses were limited to erythema and edema.
Dr. Ortiz disclosed having financial relationships with several pharmaceutical and device companies, including receipt of speaker fees and honoraria from Sofwave. She is also cochair of MOAS.
SAN DIEGO –
A noninvasive treatment, rapid acoustic pulse (RAP) technology (RESONIC), was cleared by the Food and Drug Administration in 2021 for short-term improvement in the appearance of cellulite. The device emits rapid acoustic pulses (shock waves) that are transmitted through the skin to rupture or shear the fibrotic septa; release of the septa results in the smoothing of skin dimples, Arisa E. Ortiz, MD, director of laser and cosmetic dermatology at the University of California, San Diego, said at the annual Masters of Aesthetics Symposium. RAP, however, was “taken off the market temporarily to refine the design.”
According to Dr. Ortiz, RAP’s repetition rate and short rise time provides microscopic mechanical disruption to the targeted cellular level structures and vacuoles, while high peak pressure and the fast repetition rate exploit the viscoelastic nature of tissue. Compressed pulses from electronic filtering and the reflector shape eliminate cavitation, heat, and pain. Researchers have postulated that the procedure stimulates collagenesis and angiogenesis.
“There’s no heat to this; it just uses sound,” Dr. Ortiz explained. “It’s also time dependent. The longer you do the treatment, the more disruption of the septa you see. The procedure takes 20-30 minutes. Unlike other treatments, it’s not just for discreet dimples. You can treat entire areas like the buttock or posterior leg,” said Dr. Ortiz, who did not use the RAP device in her practice.
Another device, targeted verifiable subcision (TVS, marketed as Avéli), is FDA cleared for temporary reduction in the appearance of cellulite in the buttock and thigh areas of adult women. “But studies show lasting results, at least through a year,” Dr. Ortiz said. The device features a light-guided probe and a hook. The light enables clinicians to navigate under the skin, while the hook releases a tiny blade that severs the septa. “Once you find the septa, then you activate the blade and release the septa. You go right to left because the direction of the blade is on the left of the probe. Then you go back to verify that you got everything that was creating that dimple.”
Previous devices, she said, would “blindly shear the area, so you would find the dimple and blindly cut, so there was no way to verify that you got the target dimple. The results were sometimes mediocre because you didn’t really know if you effectively treated the area.”
She emphasized that TVS is only useful for discreet dimples. “Many patients who come in asking for cellulite treatment have a lot of laxity and rippled texture,” said Dr. Ortiz, who is also president-elect of the American Society for Laser Medicine and Surgery. “This is not going to be appropriate for those cases. Setting expectations is important. If patients have laxity and discreet dimples and it’s just the dimples that bother them, that’s fine. They just need to understand the difference,” she said, noting that this is “safe for all skin types.”
Tumescent anesthesia is used to control pain during the procedure. The most common adverse events are bruising and soreness. Results from a pivotal trial showed that clinically significant improvements in the primary endpoint, Cellulite Severity Scale scores, were sustained 1 year after treatment. “Hemosiderin staining can occur, but it eventually dissipates on its own,” Dr. Ortiz added. “You can use laser to speed up healing but sometimes that can make it worse, so you want to be careful with that. Most of the time I have patients wait it out; it does go away on its own.”
She noted that the development of RAP and TVS have helped clinicians better understand the makeup of septa. “We used to think of septa as singular bands that are vertically oriented in cellulite, but what we’ve realized is that it’s more like a network of septa,” she said.
Another noninvasive technology, synchronous parallel ultrasound beam technology from Sofwave (marketed as SUPERB), was FDA cleared in December 2022 for the short-term improvement in the appearance of cellulite. The device has seven parallel beam transducers that increase tissue temperatures of the treatment area to 60-70° C, inducing collagen remodeling and collagen denaturation, she said.
In the pivotal study of 68 women, two blinded reviewers reported an 89% improvement rate for both cellulite and skin laxity, after two treatments 2-4 weeks apart, according to data she presented at the meeting. The mean pain score during treatment was 4.55 on a scale of 1-10. No safety issues were observed and immediate responses were limited to erythema and edema.
Dr. Ortiz disclosed having financial relationships with several pharmaceutical and device companies, including receipt of speaker fees and honoraria from Sofwave. She is also cochair of MOAS.
AT MOAS 2023
Hidradenitis suppurativa experts reach consensus on treatment outcome measures
TOPLINE:
Hidradenitis suppurativa (HS) experts collaborated to reach consensus on a core set of outcome measures, with the intent of improving the management of HS in clinical practice.
METHODOLOGY:
- Participants in the study were 55 HS experts from the HiSTORIC group (dermatologists, internists, surgeons, and nurses) and 24 patient research partners.
- The group identified clinician- and patient-reported HS outcome measures in the literature, then participated in an online item reduction survey, followed by an electronic Delphi survey to reach consensus on which measures should be used in clinical practice. Consensus was defined as at least 67% of participants agreeing/strongly agreeing or disagreeing/strongly disagreeing about the use of a measure in clinical practice.
- The initial literature search yielded 11 HS studies with clinician-reported outcome measures and 12 with patient-reported outcomes; of these, eight and five, respectively, were included in the final reduction survey.
TAKEAWAY:
- The group reached consensus on two HS outcome measures for use in clinical practice: the HS Investigator Global Assessment (HS-IGA) score, a clinician-reported outcome measure selected by the HS experts, and the HS Quality of Life (HiSQOL) score, a patient-reported outcome measure selected by the patient research partners.
- The HS-IGA score uses a number between 0 and 5 based on the sum of abscesses, inflammatory and noninflammatory nodules, and tunnels in regions of the upper or lower body.
- The HiSQOL, a disease-specific quality-of-life measure for adults with HS, is designed to capture unique features of HS, including symptoms (such as pain, itch, odor, and drainage) and psychosocial outcomes and activities that may be affected by the disease.
IN PRACTICE:
“The intent of these recommendations is to provide an objective framework with both clinician and patient input that can facilitate bidirectional discussion, trust building, and decision making on the current treatment strategy and the need to adjust or escalate treatment in an appropriate time frame,” the authors wrote.
SOURCE:
The study was published online in JAMA Dermatology. The lead author was Nicole Mastacouris, MS, and the corresponding author was Amit Garg, MD, both of Northwell Health, New Hyde Park, N.Y.
LIMITATIONS:
The consensus results may have been affected by variations in HS management by region. Neither measure has been studied in clinical practice, and practice variability may limit their implementation.
DISCLOSURES:
The study was supported by grants from UCB and AbbVie. Ms. Mastacouris had no financial disclosures. Dr. Garg disclosed grant support from AbbVie and UCB during the conduct of the study, as well as personal fees from AbbVie, UCB, Aclaris Therapeutics, Anaptys Bio, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Incyte, Insmed, Janssen, Novartis, Pfizer, Sonoma Biotherapeutics, Union Therapeutics, Ventyx Biosciences, and Viela Biosciences during the conduct of the study; Dr. Garg also holds patents for HS-IGA and HiSQOL. Many other coauthors disclosed relationships with multiple companies, including AbbVie and UCB, and some also disclosed patents, including patents for HiSQOL and HS Area and Severity Index.
A version of this article first appeared on Medscape.com.
TOPLINE:
Hidradenitis suppurativa (HS) experts collaborated to reach consensus on a core set of outcome measures, with the intent of improving the management of HS in clinical practice.
METHODOLOGY:
- Participants in the study were 55 HS experts from the HiSTORIC group (dermatologists, internists, surgeons, and nurses) and 24 patient research partners.
- The group identified clinician- and patient-reported HS outcome measures in the literature, then participated in an online item reduction survey, followed by an electronic Delphi survey to reach consensus on which measures should be used in clinical practice. Consensus was defined as at least 67% of participants agreeing/strongly agreeing or disagreeing/strongly disagreeing about the use of a measure in clinical practice.
- The initial literature search yielded 11 HS studies with clinician-reported outcome measures and 12 with patient-reported outcomes; of these, eight and five, respectively, were included in the final reduction survey.
TAKEAWAY:
- The group reached consensus on two HS outcome measures for use in clinical practice: the HS Investigator Global Assessment (HS-IGA) score, a clinician-reported outcome measure selected by the HS experts, and the HS Quality of Life (HiSQOL) score, a patient-reported outcome measure selected by the patient research partners.
- The HS-IGA score uses a number between 0 and 5 based on the sum of abscesses, inflammatory and noninflammatory nodules, and tunnels in regions of the upper or lower body.
- The HiSQOL, a disease-specific quality-of-life measure for adults with HS, is designed to capture unique features of HS, including symptoms (such as pain, itch, odor, and drainage) and psychosocial outcomes and activities that may be affected by the disease.
IN PRACTICE:
“The intent of these recommendations is to provide an objective framework with both clinician and patient input that can facilitate bidirectional discussion, trust building, and decision making on the current treatment strategy and the need to adjust or escalate treatment in an appropriate time frame,” the authors wrote.
SOURCE:
The study was published online in JAMA Dermatology. The lead author was Nicole Mastacouris, MS, and the corresponding author was Amit Garg, MD, both of Northwell Health, New Hyde Park, N.Y.
LIMITATIONS:
The consensus results may have been affected by variations in HS management by region. Neither measure has been studied in clinical practice, and practice variability may limit their implementation.
DISCLOSURES:
The study was supported by grants from UCB and AbbVie. Ms. Mastacouris had no financial disclosures. Dr. Garg disclosed grant support from AbbVie and UCB during the conduct of the study, as well as personal fees from AbbVie, UCB, Aclaris Therapeutics, Anaptys Bio, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Incyte, Insmed, Janssen, Novartis, Pfizer, Sonoma Biotherapeutics, Union Therapeutics, Ventyx Biosciences, and Viela Biosciences during the conduct of the study; Dr. Garg also holds patents for HS-IGA and HiSQOL. Many other coauthors disclosed relationships with multiple companies, including AbbVie and UCB, and some also disclosed patents, including patents for HiSQOL and HS Area and Severity Index.
A version of this article first appeared on Medscape.com.
TOPLINE:
Hidradenitis suppurativa (HS) experts collaborated to reach consensus on a core set of outcome measures, with the intent of improving the management of HS in clinical practice.
METHODOLOGY:
- Participants in the study were 55 HS experts from the HiSTORIC group (dermatologists, internists, surgeons, and nurses) and 24 patient research partners.
- The group identified clinician- and patient-reported HS outcome measures in the literature, then participated in an online item reduction survey, followed by an electronic Delphi survey to reach consensus on which measures should be used in clinical practice. Consensus was defined as at least 67% of participants agreeing/strongly agreeing or disagreeing/strongly disagreeing about the use of a measure in clinical practice.
- The initial literature search yielded 11 HS studies with clinician-reported outcome measures and 12 with patient-reported outcomes; of these, eight and five, respectively, were included in the final reduction survey.
TAKEAWAY:
- The group reached consensus on two HS outcome measures for use in clinical practice: the HS Investigator Global Assessment (HS-IGA) score, a clinician-reported outcome measure selected by the HS experts, and the HS Quality of Life (HiSQOL) score, a patient-reported outcome measure selected by the patient research partners.
- The HS-IGA score uses a number between 0 and 5 based on the sum of abscesses, inflammatory and noninflammatory nodules, and tunnels in regions of the upper or lower body.
- The HiSQOL, a disease-specific quality-of-life measure for adults with HS, is designed to capture unique features of HS, including symptoms (such as pain, itch, odor, and drainage) and psychosocial outcomes and activities that may be affected by the disease.
IN PRACTICE:
“The intent of these recommendations is to provide an objective framework with both clinician and patient input that can facilitate bidirectional discussion, trust building, and decision making on the current treatment strategy and the need to adjust or escalate treatment in an appropriate time frame,” the authors wrote.
SOURCE:
The study was published online in JAMA Dermatology. The lead author was Nicole Mastacouris, MS, and the corresponding author was Amit Garg, MD, both of Northwell Health, New Hyde Park, N.Y.
LIMITATIONS:
The consensus results may have been affected by variations in HS management by region. Neither measure has been studied in clinical practice, and practice variability may limit their implementation.
DISCLOSURES:
The study was supported by grants from UCB and AbbVie. Ms. Mastacouris had no financial disclosures. Dr. Garg disclosed grant support from AbbVie and UCB during the conduct of the study, as well as personal fees from AbbVie, UCB, Aclaris Therapeutics, Anaptys Bio, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Incyte, Insmed, Janssen, Novartis, Pfizer, Sonoma Biotherapeutics, Union Therapeutics, Ventyx Biosciences, and Viela Biosciences during the conduct of the study; Dr. Garg also holds patents for HS-IGA and HiSQOL. Many other coauthors disclosed relationships with multiple companies, including AbbVie and UCB, and some also disclosed patents, including patents for HiSQOL and HS Area and Severity Index.
A version of this article first appeared on Medscape.com.
FROM JAMA DERMATOLOGY
Hidradenitis suppurativa experts reach consensus on treatment outcome measures
TOPLINE:
METHODOLOGY:
- Participants in the study were 55 HS experts from the HiSTORIC group (dermatologists, internists, surgeons, and nurses) and 24 patient research partners.
- The group identified clinician- and patient-reported HS outcome measures in the literature, then participated in an online item reduction survey, followed by an electronic Delphi survey to reach consensus on which measures should be used in clinical practice. Consensus was defined as at least 67% of participants agreeing/strongly agreeing or disagreeing/strongly disagreeing about the use of a measure in clinical practice.
- The initial literature search yielded 11 HS studies with clinician-reported outcome measures and 12 with patient-reported outcomes; of these, eight and five, respectively, were included in the final reduction survey.
TAKEAWAY:
- The group reached consensus on two HS outcome measures for use in clinical practice: the HS Investigator Global Assessment (HS-IGA) score, a clinician-reported outcome measure selected by the HS experts, and the HS Quality of Life (HiSQOL) score, a patient-reported outcome measure selected by the patient research partners.
- The HS-IGA score uses a number between 0 and 5 based on the sum of abscesses, inflammatory and noninflammatory nodules, and tunnels in regions of the upper or lower body.
- The HiSQOL, a disease-specific quality-of-life measure for adults with HS, is designed to capture unique features of HS, including symptoms (such as pain, itch, odor, and drainage) and psychosocial outcomes and activities that may be affected by the disease.
IN PRACTICE:
“The intent of these recommendations is to provide an objective framework with both clinician and patient input that can facilitate bidirectional discussion, trust building, and decision making on the current treatment strategy and the need to adjust or escalate treatment in an appropriate time frame,” the authors wrote.
SOURCE:
The study was published online in JAMA Dermatology. The lead author was Nicole Mastacouris, MS, and the corresponding author was Amit Garg, MD, both of Northwell Health, New Hyde Park, N.Y.
LIMITATIONS:
The consensus results may have been affected by variations in HS management by region. Neither measure has been studied in clinical practice, and practice variability may limit their implementation.
DISCLOSURES:
The study was supported by grants from UCB and AbbVie. Ms. Mastacouris had no financial disclosures. Dr. Garg disclosed grant support from AbbVie and UCB during the conduct of the study, as well as personal fees from AbbVie, UCB, Aclaris Therapeutics, Anaptys Bio, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Incyte, Insmed, Janssen, Novartis, Pfizer, Sonoma Biotherapeutics, Union Therapeutics, Ventyx Biosciences, and Viela Biosciences during the conduct of the study; Dr. Garg also holds patents for HS-IGA and HiSQOL. Many other coauthors disclosed relationships with multiple companies, including AbbVie and UCB, and some also disclosed patents, including patents for HiSQOL and HS Area and Severity Index.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Participants in the study were 55 HS experts from the HiSTORIC group (dermatologists, internists, surgeons, and nurses) and 24 patient research partners.
- The group identified clinician- and patient-reported HS outcome measures in the literature, then participated in an online item reduction survey, followed by an electronic Delphi survey to reach consensus on which measures should be used in clinical practice. Consensus was defined as at least 67% of participants agreeing/strongly agreeing or disagreeing/strongly disagreeing about the use of a measure in clinical practice.
- The initial literature search yielded 11 HS studies with clinician-reported outcome measures and 12 with patient-reported outcomes; of these, eight and five, respectively, were included in the final reduction survey.
TAKEAWAY:
- The group reached consensus on two HS outcome measures for use in clinical practice: the HS Investigator Global Assessment (HS-IGA) score, a clinician-reported outcome measure selected by the HS experts, and the HS Quality of Life (HiSQOL) score, a patient-reported outcome measure selected by the patient research partners.
- The HS-IGA score uses a number between 0 and 5 based on the sum of abscesses, inflammatory and noninflammatory nodules, and tunnels in regions of the upper or lower body.
- The HiSQOL, a disease-specific quality-of-life measure for adults with HS, is designed to capture unique features of HS, including symptoms (such as pain, itch, odor, and drainage) and psychosocial outcomes and activities that may be affected by the disease.
IN PRACTICE:
“The intent of these recommendations is to provide an objective framework with both clinician and patient input that can facilitate bidirectional discussion, trust building, and decision making on the current treatment strategy and the need to adjust or escalate treatment in an appropriate time frame,” the authors wrote.
SOURCE:
The study was published online in JAMA Dermatology. The lead author was Nicole Mastacouris, MS, and the corresponding author was Amit Garg, MD, both of Northwell Health, New Hyde Park, N.Y.
LIMITATIONS:
The consensus results may have been affected by variations in HS management by region. Neither measure has been studied in clinical practice, and practice variability may limit their implementation.
DISCLOSURES:
The study was supported by grants from UCB and AbbVie. Ms. Mastacouris had no financial disclosures. Dr. Garg disclosed grant support from AbbVie and UCB during the conduct of the study, as well as personal fees from AbbVie, UCB, Aclaris Therapeutics, Anaptys Bio, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Incyte, Insmed, Janssen, Novartis, Pfizer, Sonoma Biotherapeutics, Union Therapeutics, Ventyx Biosciences, and Viela Biosciences during the conduct of the study; Dr. Garg also holds patents for HS-IGA and HiSQOL. Many other coauthors disclosed relationships with multiple companies, including AbbVie and UCB, and some also disclosed patents, including patents for HiSQOL and HS Area and Severity Index.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Participants in the study were 55 HS experts from the HiSTORIC group (dermatologists, internists, surgeons, and nurses) and 24 patient research partners.
- The group identified clinician- and patient-reported HS outcome measures in the literature, then participated in an online item reduction survey, followed by an electronic Delphi survey to reach consensus on which measures should be used in clinical practice. Consensus was defined as at least 67% of participants agreeing/strongly agreeing or disagreeing/strongly disagreeing about the use of a measure in clinical practice.
- The initial literature search yielded 11 HS studies with clinician-reported outcome measures and 12 with patient-reported outcomes; of these, eight and five, respectively, were included in the final reduction survey.
TAKEAWAY:
- The group reached consensus on two HS outcome measures for use in clinical practice: the HS Investigator Global Assessment (HS-IGA) score, a clinician-reported outcome measure selected by the HS experts, and the HS Quality of Life (HiSQOL) score, a patient-reported outcome measure selected by the patient research partners.
- The HS-IGA score uses a number between 0 and 5 based on the sum of abscesses, inflammatory and noninflammatory nodules, and tunnels in regions of the upper or lower body.
- The HiSQOL, a disease-specific quality-of-life measure for adults with HS, is designed to capture unique features of HS, including symptoms (such as pain, itch, odor, and drainage) and psychosocial outcomes and activities that may be affected by the disease.
IN PRACTICE:
“The intent of these recommendations is to provide an objective framework with both clinician and patient input that can facilitate bidirectional discussion, trust building, and decision making on the current treatment strategy and the need to adjust or escalate treatment in an appropriate time frame,” the authors wrote.
SOURCE:
The study was published online in JAMA Dermatology. The lead author was Nicole Mastacouris, MS, and the corresponding author was Amit Garg, MD, both of Northwell Health, New Hyde Park, N.Y.
LIMITATIONS:
The consensus results may have been affected by variations in HS management by region. Neither measure has been studied in clinical practice, and practice variability may limit their implementation.
DISCLOSURES:
The study was supported by grants from UCB and AbbVie. Ms. Mastacouris had no financial disclosures. Dr. Garg disclosed grant support from AbbVie and UCB during the conduct of the study, as well as personal fees from AbbVie, UCB, Aclaris Therapeutics, Anaptys Bio, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Incyte, Insmed, Janssen, Novartis, Pfizer, Sonoma Biotherapeutics, Union Therapeutics, Ventyx Biosciences, and Viela Biosciences during the conduct of the study; Dr. Garg also holds patents for HS-IGA and HiSQOL. Many other coauthors disclosed relationships with multiple companies, including AbbVie and UCB, and some also disclosed patents, including patents for HiSQOL and HS Area and Severity Index.
A version of this article first appeared on Medscape.com.
FROM JAMA DERMATOLOGY
Hidradenitis suppurativa experts reach consensus on treatment outcome measures
TOPLINE:
METHODOLOGY:
- Participants in the study were 55 HS experts from the HiSTORIC group (dermatologists, internists, surgeons, and nurses) and 24 patient research partners.
- The group identified clinician- and patient-reported HS outcome measures in the literature, then participated in an online item reduction survey, followed by an electronic Delphi survey to reach consensus on which measures should be used in clinical practice. Consensus was defined as at least 67% of participants agreeing/strongly agreeing or disagreeing/strongly disagreeing about the use of a measure in clinical practice.
- The initial literature search yielded 11 HS studies with clinician-reported outcome measures and 12 with patient-reported outcomes; of these, eight and five, respectively, were included in the final reduction survey.
TAKEAWAY:
- The group reached consensus on two HS outcome measures for use in clinical practice: the HS Investigator Global Assessment (HS-IGA) score, a clinician-reported outcome measure selected by the HS experts, and the HS Quality of Life (HiSQOL) score, a patient-reported outcome measure selected by the patient research partners.
- The HS-IGA score uses a number between 0 and 5 based on the sum of abscesses, inflammatory and noninflammatory nodules, and tunnels in regions of the upper or lower body.
- The HiSQOL, a disease-specific quality-of-life measure for adults with HS, is designed to capture unique features of HS, including symptoms (such as pain, itch, odor, and drainage) and psychosocial outcomes and activities that may be affected by the disease.
IN PRACTICE:
“The intent of these recommendations is to provide an objective framework with both clinician and patient input that can facilitate bidirectional discussion, trust building, and decision making on the current treatment strategy and the need to adjust or escalate treatment in an appropriate time frame,” the authors wrote.
SOURCE:
The study was published online in JAMA Dermatology. The lead author was Nicole Mastacouris, MS, and the corresponding author was Amit Garg, MD, both of Northwell Health, New Hyde Park, N.Y.
LIMITATIONS:
The consensus results may have been affected by variations in HS management by region. Neither measure has been studied in clinical practice, and practice variability may limit their implementation.
DISCLOSURES:
The study was supported by grants from UCB and AbbVie. Ms. Mastacouris had no financial disclosures. Dr. Garg disclosed grant support from AbbVie and UCB during the conduct of the study, as well as personal fees from AbbVie, UCB, Aclaris Therapeutics, Anaptys Bio, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Incyte, Insmed, Janssen, Novartis, Pfizer, Sonoma Biotherapeutics, Union Therapeutics, Ventyx Biosciences, and Viela Biosciences during the conduct of the study; Dr. Garg also holds patents for HS-IGA and HiSQOL. Many other coauthors disclosed relationships with multiple companies, including AbbVie and UCB, and some also disclosed patents, including patents for HiSQOL and HS Area and Severity Index.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Participants in the study were 55 HS experts from the HiSTORIC group (dermatologists, internists, surgeons, and nurses) and 24 patient research partners.
- The group identified clinician- and patient-reported HS outcome measures in the literature, then participated in an online item reduction survey, followed by an electronic Delphi survey to reach consensus on which measures should be used in clinical practice. Consensus was defined as at least 67% of participants agreeing/strongly agreeing or disagreeing/strongly disagreeing about the use of a measure in clinical practice.
- The initial literature search yielded 11 HS studies with clinician-reported outcome measures and 12 with patient-reported outcomes; of these, eight and five, respectively, were included in the final reduction survey.
TAKEAWAY:
- The group reached consensus on two HS outcome measures for use in clinical practice: the HS Investigator Global Assessment (HS-IGA) score, a clinician-reported outcome measure selected by the HS experts, and the HS Quality of Life (HiSQOL) score, a patient-reported outcome measure selected by the patient research partners.
- The HS-IGA score uses a number between 0 and 5 based on the sum of abscesses, inflammatory and noninflammatory nodules, and tunnels in regions of the upper or lower body.
- The HiSQOL, a disease-specific quality-of-life measure for adults with HS, is designed to capture unique features of HS, including symptoms (such as pain, itch, odor, and drainage) and psychosocial outcomes and activities that may be affected by the disease.
IN PRACTICE:
“The intent of these recommendations is to provide an objective framework with both clinician and patient input that can facilitate bidirectional discussion, trust building, and decision making on the current treatment strategy and the need to adjust or escalate treatment in an appropriate time frame,” the authors wrote.
SOURCE:
The study was published online in JAMA Dermatology. The lead author was Nicole Mastacouris, MS, and the corresponding author was Amit Garg, MD, both of Northwell Health, New Hyde Park, N.Y.
LIMITATIONS:
The consensus results may have been affected by variations in HS management by region. Neither measure has been studied in clinical practice, and practice variability may limit their implementation.
DISCLOSURES:
The study was supported by grants from UCB and AbbVie. Ms. Mastacouris had no financial disclosures. Dr. Garg disclosed grant support from AbbVie and UCB during the conduct of the study, as well as personal fees from AbbVie, UCB, Aclaris Therapeutics, Anaptys Bio, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Incyte, Insmed, Janssen, Novartis, Pfizer, Sonoma Biotherapeutics, Union Therapeutics, Ventyx Biosciences, and Viela Biosciences during the conduct of the study; Dr. Garg also holds patents for HS-IGA and HiSQOL. Many other coauthors disclosed relationships with multiple companies, including AbbVie and UCB, and some also disclosed patents, including patents for HiSQOL and HS Area and Severity Index.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Participants in the study were 55 HS experts from the HiSTORIC group (dermatologists, internists, surgeons, and nurses) and 24 patient research partners.
- The group identified clinician- and patient-reported HS outcome measures in the literature, then participated in an online item reduction survey, followed by an electronic Delphi survey to reach consensus on which measures should be used in clinical practice. Consensus was defined as at least 67% of participants agreeing/strongly agreeing or disagreeing/strongly disagreeing about the use of a measure in clinical practice.
- The initial literature search yielded 11 HS studies with clinician-reported outcome measures and 12 with patient-reported outcomes; of these, eight and five, respectively, were included in the final reduction survey.
TAKEAWAY:
- The group reached consensus on two HS outcome measures for use in clinical practice: the HS Investigator Global Assessment (HS-IGA) score, a clinician-reported outcome measure selected by the HS experts, and the HS Quality of Life (HiSQOL) score, a patient-reported outcome measure selected by the patient research partners.
- The HS-IGA score uses a number between 0 and 5 based on the sum of abscesses, inflammatory and noninflammatory nodules, and tunnels in regions of the upper or lower body.
- The HiSQOL, a disease-specific quality-of-life measure for adults with HS, is designed to capture unique features of HS, including symptoms (such as pain, itch, odor, and drainage) and psychosocial outcomes and activities that may be affected by the disease.
IN PRACTICE:
“The intent of these recommendations is to provide an objective framework with both clinician and patient input that can facilitate bidirectional discussion, trust building, and decision making on the current treatment strategy and the need to adjust or escalate treatment in an appropriate time frame,” the authors wrote.
SOURCE:
The study was published online in JAMA Dermatology. The lead author was Nicole Mastacouris, MS, and the corresponding author was Amit Garg, MD, both of Northwell Health, New Hyde Park, N.Y.
LIMITATIONS:
The consensus results may have been affected by variations in HS management by region. Neither measure has been studied in clinical practice, and practice variability may limit their implementation.
DISCLOSURES:
The study was supported by grants from UCB and AbbVie. Ms. Mastacouris had no financial disclosures. Dr. Garg disclosed grant support from AbbVie and UCB during the conduct of the study, as well as personal fees from AbbVie, UCB, Aclaris Therapeutics, Anaptys Bio, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Incyte, Insmed, Janssen, Novartis, Pfizer, Sonoma Biotherapeutics, Union Therapeutics, Ventyx Biosciences, and Viela Biosciences during the conduct of the study; Dr. Garg also holds patents for HS-IGA and HiSQOL. Many other coauthors disclosed relationships with multiple companies, including AbbVie and UCB, and some also disclosed patents, including patents for HiSQOL and HS Area and Severity Index.
A version of this article first appeared on Medscape.com.
FROM JAMA DERMATOLOGY
ACP addresses ethical issues for ‘grateful patients’ physician fundraising
Patients sometimes want to give back to their physician or hospital. In recent years, the practice of soliciting donations from these patients has grown into structured fundraising initiatives at some health care organizations. Some employers mandate clinicians solicit donations, while other doctors participate voluntarily.
“In recent decades, more physician practices have become part of large health systems: these arrangements can offer benefits to care but can also lead to interference in the patient-physician relationship and challenges to the physician’s ethical responsibilities to patients,” said Omar T. Atiq, MD, president of the American College of Physicians.
Grateful patient fundraising (GPF) is largely based on models of charitable giving outside of health care and is relatively new to the industry. Simply defined, it is the solicitation of donations by doctors from current and former patients. Funds may be used for operating costs, clinical research, equipment upgrades, or facility improvements.
In a newly published position paper, the ACP, which represents roughly 161,000 physicians, is clear that clinicians should not try to convert their patients into donors.
“Physicians who directly solicit funds from their own patients do risk interfering with the physician-patient relationship, which is supposed to be based on the patient’s best interests, not the physicians’ interests,” said Stacey A. Tovino, JD, PhD, director of health care law programs at the University of Oklahoma, Norman.
Once involved in fundraising, patients may also develop an unrealistic expectation of what kind of care they should receive, according to the ACP.
Another pitfall clinicians may fall into is the HIPAA Privacy Rule. In 2013, HIPAA was expanded to allow hospital fundraisers to access privileged health information, including demographic, health insurance, treating clinician, and data on outcomes. Dr. Atiq said that, since then, electronic health records have been used as tools to aide fundraising efforts. For instance, some health care organizations have embedded a feature inside EHRs to allow physicians to flag development officers when a patient or family member might be a potential donor.
Patients may be unaware that hospital fundraising departments have access to their electronic health records, or that they have the right to opt out of fundraising solicitations.
“Physicians should not use or reveal patient information for fundraising,” Dr. Atiq said. “Even acknowledging that a person is under one’s care can make it possible for protected health information to be revealed.”
Data-mining EHRs may be legal, Ms. Tovino said, but it hugs a fine ethical line.
“A patient may not expect that their information will be used for these purposes and may not know how to opt out of having their information used in these ways,” Ms. Tovino said.
A clinician’s employment contract, whether it be a full-time position or for specific admitting privileges, may make it hard for them to push back against expectations to ask patients for money or screen for donors. Metrics or expectations to approach potential donors create ethical snares for clinicians – and it pits them between their patient and place of employment.
“GPF does raise ethical concerns, including those surrounding confidentiality and privacy, and whether physicians are being remunerated or evaluated based on their participation,” Ms. Tovino said.
Asked how doctors can avoid being involved in GPF, Dr. Atiq referred to the ACP ethics manual, which separates clinicians from fundraising.
“Redirecting the patient to discuss donations with institutional administrators provides the appropriate venue and firewall,” he said.
An author of the ACP paper reported a paid position on the board of the Government Employees Health Association.
A version of this article first appeared on Medscape.com.
Patients sometimes want to give back to their physician or hospital. In recent years, the practice of soliciting donations from these patients has grown into structured fundraising initiatives at some health care organizations. Some employers mandate clinicians solicit donations, while other doctors participate voluntarily.
“In recent decades, more physician practices have become part of large health systems: these arrangements can offer benefits to care but can also lead to interference in the patient-physician relationship and challenges to the physician’s ethical responsibilities to patients,” said Omar T. Atiq, MD, president of the American College of Physicians.
Grateful patient fundraising (GPF) is largely based on models of charitable giving outside of health care and is relatively new to the industry. Simply defined, it is the solicitation of donations by doctors from current and former patients. Funds may be used for operating costs, clinical research, equipment upgrades, or facility improvements.
In a newly published position paper, the ACP, which represents roughly 161,000 physicians, is clear that clinicians should not try to convert their patients into donors.
“Physicians who directly solicit funds from their own patients do risk interfering with the physician-patient relationship, which is supposed to be based on the patient’s best interests, not the physicians’ interests,” said Stacey A. Tovino, JD, PhD, director of health care law programs at the University of Oklahoma, Norman.
Once involved in fundraising, patients may also develop an unrealistic expectation of what kind of care they should receive, according to the ACP.
Another pitfall clinicians may fall into is the HIPAA Privacy Rule. In 2013, HIPAA was expanded to allow hospital fundraisers to access privileged health information, including demographic, health insurance, treating clinician, and data on outcomes. Dr. Atiq said that, since then, electronic health records have been used as tools to aide fundraising efforts. For instance, some health care organizations have embedded a feature inside EHRs to allow physicians to flag development officers when a patient or family member might be a potential donor.
Patients may be unaware that hospital fundraising departments have access to their electronic health records, or that they have the right to opt out of fundraising solicitations.
“Physicians should not use or reveal patient information for fundraising,” Dr. Atiq said. “Even acknowledging that a person is under one’s care can make it possible for protected health information to be revealed.”
Data-mining EHRs may be legal, Ms. Tovino said, but it hugs a fine ethical line.
“A patient may not expect that their information will be used for these purposes and may not know how to opt out of having their information used in these ways,” Ms. Tovino said.
A clinician’s employment contract, whether it be a full-time position or for specific admitting privileges, may make it hard for them to push back against expectations to ask patients for money or screen for donors. Metrics or expectations to approach potential donors create ethical snares for clinicians – and it pits them between their patient and place of employment.
“GPF does raise ethical concerns, including those surrounding confidentiality and privacy, and whether physicians are being remunerated or evaluated based on their participation,” Ms. Tovino said.
Asked how doctors can avoid being involved in GPF, Dr. Atiq referred to the ACP ethics manual, which separates clinicians from fundraising.
“Redirecting the patient to discuss donations with institutional administrators provides the appropriate venue and firewall,” he said.
An author of the ACP paper reported a paid position on the board of the Government Employees Health Association.
A version of this article first appeared on Medscape.com.
Patients sometimes want to give back to their physician or hospital. In recent years, the practice of soliciting donations from these patients has grown into structured fundraising initiatives at some health care organizations. Some employers mandate clinicians solicit donations, while other doctors participate voluntarily.
“In recent decades, more physician practices have become part of large health systems: these arrangements can offer benefits to care but can also lead to interference in the patient-physician relationship and challenges to the physician’s ethical responsibilities to patients,” said Omar T. Atiq, MD, president of the American College of Physicians.
Grateful patient fundraising (GPF) is largely based on models of charitable giving outside of health care and is relatively new to the industry. Simply defined, it is the solicitation of donations by doctors from current and former patients. Funds may be used for operating costs, clinical research, equipment upgrades, or facility improvements.
In a newly published position paper, the ACP, which represents roughly 161,000 physicians, is clear that clinicians should not try to convert their patients into donors.
“Physicians who directly solicit funds from their own patients do risk interfering with the physician-patient relationship, which is supposed to be based on the patient’s best interests, not the physicians’ interests,” said Stacey A. Tovino, JD, PhD, director of health care law programs at the University of Oklahoma, Norman.
Once involved in fundraising, patients may also develop an unrealistic expectation of what kind of care they should receive, according to the ACP.
Another pitfall clinicians may fall into is the HIPAA Privacy Rule. In 2013, HIPAA was expanded to allow hospital fundraisers to access privileged health information, including demographic, health insurance, treating clinician, and data on outcomes. Dr. Atiq said that, since then, electronic health records have been used as tools to aide fundraising efforts. For instance, some health care organizations have embedded a feature inside EHRs to allow physicians to flag development officers when a patient or family member might be a potential donor.
Patients may be unaware that hospital fundraising departments have access to their electronic health records, or that they have the right to opt out of fundraising solicitations.
“Physicians should not use or reveal patient information for fundraising,” Dr. Atiq said. “Even acknowledging that a person is under one’s care can make it possible for protected health information to be revealed.”
Data-mining EHRs may be legal, Ms. Tovino said, but it hugs a fine ethical line.
“A patient may not expect that their information will be used for these purposes and may not know how to opt out of having their information used in these ways,” Ms. Tovino said.
A clinician’s employment contract, whether it be a full-time position or for specific admitting privileges, may make it hard for them to push back against expectations to ask patients for money or screen for donors. Metrics or expectations to approach potential donors create ethical snares for clinicians – and it pits them between their patient and place of employment.
“GPF does raise ethical concerns, including those surrounding confidentiality and privacy, and whether physicians are being remunerated or evaluated based on their participation,” Ms. Tovino said.
Asked how doctors can avoid being involved in GPF, Dr. Atiq referred to the ACP ethics manual, which separates clinicians from fundraising.
“Redirecting the patient to discuss donations with institutional administrators provides the appropriate venue and firewall,” he said.
An author of the ACP paper reported a paid position on the board of the Government Employees Health Association.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF INTERNAL MEDICINE
Study spotlights paucity of black dermatologists in academia
TOPLINE:
METHODOLOGY:
- To assess the prevalence of Black dermatologists in academic dermatology programs, researchers obtained an inventory of all 142 U.S.-accredited dermatology residency programs from the Accreditation Council for Graduate Medical Education.
- The researchers drew from institutional websites, the Black Derm Directory (an online repository of Black dermatologists), and other sources to identify full- and part-time Black faculty.
- Variables of interest for each Black dermatologist included gender, institution, department title, academic and nonacademic leadership roles, publication number, National Institutes of Health grant funding, degrees, subspecialties, medical school attended, place of residency, and fellowship training.
- The researchers used Pearson’s chi-squared testing to calculate associations.
TAKEAWAY:
- Of the 86 Black faculty identified, 81.4% were female; most (42.4%) were in the southern United States, followed by the Midwest (23.5%); and 83% held full-time positions.
- Slightly more than one-quarter (26.7%) of the Black faculty attended a top 10 medical school, 16.3% graduated from a historically Black college and university medical school, and 43.5% of those with 25 or more research publications had attended a top 10 medical school.
- Only three dermatology department chairs were Black, and all were female. In addition, more than half of Black faculty (59.2%) were assistant professors, 37.7% held leadership positions at their institutions, and 32.6% held outside leadership roles in dermatology (such as leadership titles at professional dermatology organizations or editorial positions at a journal).
IN PRACTICE:
“Greater efforts are needed to recruit Black dermatology graduates into academic faculty positions,” and “faculty development programs offered by academic institutions and dermatologic associations ... should continue to be expanded,” the authors conclude.
SOURCE:
Corresponding author Nada Elbuluk, MD, MSc, director of the skin of color and pigmentary disorders program and the diversity and inclusion program in the department of dermatology at the University of Southern California, Los Angeles, led the research. The study was published in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The process for identifying Black faculty and insufficient or outdated information on department websites were limitations.
DISCLOSURES:
Dr. Elbuluk disclosed that she has served as a consultant for Avita, Scientis, Incyte, VisualDx, La Roche Posay, Beiersdorf, and Unilever. She has served on advisory boards for Allergan, Eli Lilly, Galderma, Incyte, Pfizer, Janssen, La Roche Posay, L’Oreal, McGraw Hill, and Dior. She has been a speaker for La Roche Posay, Scientis, Medscape, Beiersdorf, and Dior, and has served as investigator for Avita. Another author is an investigator and speaker for Castle Biosciences.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- To assess the prevalence of Black dermatologists in academic dermatology programs, researchers obtained an inventory of all 142 U.S.-accredited dermatology residency programs from the Accreditation Council for Graduate Medical Education.
- The researchers drew from institutional websites, the Black Derm Directory (an online repository of Black dermatologists), and other sources to identify full- and part-time Black faculty.
- Variables of interest for each Black dermatologist included gender, institution, department title, academic and nonacademic leadership roles, publication number, National Institutes of Health grant funding, degrees, subspecialties, medical school attended, place of residency, and fellowship training.
- The researchers used Pearson’s chi-squared testing to calculate associations.
TAKEAWAY:
- Of the 86 Black faculty identified, 81.4% were female; most (42.4%) were in the southern United States, followed by the Midwest (23.5%); and 83% held full-time positions.
- Slightly more than one-quarter (26.7%) of the Black faculty attended a top 10 medical school, 16.3% graduated from a historically Black college and university medical school, and 43.5% of those with 25 or more research publications had attended a top 10 medical school.
- Only three dermatology department chairs were Black, and all were female. In addition, more than half of Black faculty (59.2%) were assistant professors, 37.7% held leadership positions at their institutions, and 32.6% held outside leadership roles in dermatology (such as leadership titles at professional dermatology organizations or editorial positions at a journal).
IN PRACTICE:
“Greater efforts are needed to recruit Black dermatology graduates into academic faculty positions,” and “faculty development programs offered by academic institutions and dermatologic associations ... should continue to be expanded,” the authors conclude.
SOURCE:
Corresponding author Nada Elbuluk, MD, MSc, director of the skin of color and pigmentary disorders program and the diversity and inclusion program in the department of dermatology at the University of Southern California, Los Angeles, led the research. The study was published in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The process for identifying Black faculty and insufficient or outdated information on department websites were limitations.
DISCLOSURES:
Dr. Elbuluk disclosed that she has served as a consultant for Avita, Scientis, Incyte, VisualDx, La Roche Posay, Beiersdorf, and Unilever. She has served on advisory boards for Allergan, Eli Lilly, Galderma, Incyte, Pfizer, Janssen, La Roche Posay, L’Oreal, McGraw Hill, and Dior. She has been a speaker for La Roche Posay, Scientis, Medscape, Beiersdorf, and Dior, and has served as investigator for Avita. Another author is an investigator and speaker for Castle Biosciences.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- To assess the prevalence of Black dermatologists in academic dermatology programs, researchers obtained an inventory of all 142 U.S.-accredited dermatology residency programs from the Accreditation Council for Graduate Medical Education.
- The researchers drew from institutional websites, the Black Derm Directory (an online repository of Black dermatologists), and other sources to identify full- and part-time Black faculty.
- Variables of interest for each Black dermatologist included gender, institution, department title, academic and nonacademic leadership roles, publication number, National Institutes of Health grant funding, degrees, subspecialties, medical school attended, place of residency, and fellowship training.
- The researchers used Pearson’s chi-squared testing to calculate associations.
TAKEAWAY:
- Of the 86 Black faculty identified, 81.4% were female; most (42.4%) were in the southern United States, followed by the Midwest (23.5%); and 83% held full-time positions.
- Slightly more than one-quarter (26.7%) of the Black faculty attended a top 10 medical school, 16.3% graduated from a historically Black college and university medical school, and 43.5% of those with 25 or more research publications had attended a top 10 medical school.
- Only three dermatology department chairs were Black, and all were female. In addition, more than half of Black faculty (59.2%) were assistant professors, 37.7% held leadership positions at their institutions, and 32.6% held outside leadership roles in dermatology (such as leadership titles at professional dermatology organizations or editorial positions at a journal).
IN PRACTICE:
“Greater efforts are needed to recruit Black dermatology graduates into academic faculty positions,” and “faculty development programs offered by academic institutions and dermatologic associations ... should continue to be expanded,” the authors conclude.
SOURCE:
Corresponding author Nada Elbuluk, MD, MSc, director of the skin of color and pigmentary disorders program and the diversity and inclusion program in the department of dermatology at the University of Southern California, Los Angeles, led the research. The study was published in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The process for identifying Black faculty and insufficient or outdated information on department websites were limitations.
DISCLOSURES:
Dr. Elbuluk disclosed that she has served as a consultant for Avita, Scientis, Incyte, VisualDx, La Roche Posay, Beiersdorf, and Unilever. She has served on advisory boards for Allergan, Eli Lilly, Galderma, Incyte, Pfizer, Janssen, La Roche Posay, L’Oreal, McGraw Hill, and Dior. She has been a speaker for La Roche Posay, Scientis, Medscape, Beiersdorf, and Dior, and has served as investigator for Avita. Another author is an investigator and speaker for Castle Biosciences.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Most adults with alopecia areata untreated 1 year after diagnosis
TOPLINE:
using data from more than 45,000 individuals.
METHODOLOGY:
- The study population included 45,483 adults aged 18 years and older with new diagnoses of AA between Oct. 15, 2015, and Feb. 28, 2020. Data were from a large U.S. health care database that included medical and pharmacy claims.
- The mean age of the participants was 43.8 years, and 65.7% were female.
- The researchers measured variables that might relate to AA and its treatment patterns within 1 year of starting the study and during the first year of the study, with data collected at 1, 42, 84, and 365 days after study entry.
TAKEAWAYS:
- During the first year after diagnosis, 66.4% of patients received at least one treatment for AA at one or more time points.
- At 1 year, 71.8% of patients were not receiving any active treatment for AA.
- Among those who received treatment, intralesional injections were the most often prescribed therapy (41.8% of patients), followed by topical corticosteroids (40.9%), intramuscular corticosteroids (38.1%), and oral corticosteroids (20.6%).
- Patients diagnosed with either alopecia totalis or alopecia universalis were significantly less likely to receive intralesional steroids and significantly more likely to receive topical corticosteroids than those without these diagnoses (11.1% vs. 44.1% and 25.4% vs. 42.1, respectively).
IN PRACTICE:
The results highlight the need to determine why so many alopecia patients with AA were no longer on treatment after 1 year, although treatment trends may change with the emergence of new therapies, such as JAK inhibitors and others, according to the authors.
SOURCE:
The lead author of the study was Hemin Lee, MD, MPH, Brigham and Women’s Hospital, Boston. The study was published online in JAMA Dermatology.
LIMITATIONS:
The use of insurance claims data did not allow analysis of over-the-counter medications and treatments, and the lack of a single ICD-10 code for defining AA could have resulted in misclassification of outcomes.
DISCLOSURES:
The study received no outside funding, and Dr. Lee had no disclosures. One author had disclosures that included receiving personal fees from Pfizer and Concert outside of the submitted study and participating in alopecia-related trials with Lilly, Concert, Aclaris, and Incyte. Another author’s disclosures included receiving personal fees from companies that included Pfizer, Concert, Lilly, and AbbVie. No other disclosures were reported.
A version of this article first appeared on Medscape.com.
TOPLINE:
using data from more than 45,000 individuals.
METHODOLOGY:
- The study population included 45,483 adults aged 18 years and older with new diagnoses of AA between Oct. 15, 2015, and Feb. 28, 2020. Data were from a large U.S. health care database that included medical and pharmacy claims.
- The mean age of the participants was 43.8 years, and 65.7% were female.
- The researchers measured variables that might relate to AA and its treatment patterns within 1 year of starting the study and during the first year of the study, with data collected at 1, 42, 84, and 365 days after study entry.
TAKEAWAYS:
- During the first year after diagnosis, 66.4% of patients received at least one treatment for AA at one or more time points.
- At 1 year, 71.8% of patients were not receiving any active treatment for AA.
- Among those who received treatment, intralesional injections were the most often prescribed therapy (41.8% of patients), followed by topical corticosteroids (40.9%), intramuscular corticosteroids (38.1%), and oral corticosteroids (20.6%).
- Patients diagnosed with either alopecia totalis or alopecia universalis were significantly less likely to receive intralesional steroids and significantly more likely to receive topical corticosteroids than those without these diagnoses (11.1% vs. 44.1% and 25.4% vs. 42.1, respectively).
IN PRACTICE:
The results highlight the need to determine why so many alopecia patients with AA were no longer on treatment after 1 year, although treatment trends may change with the emergence of new therapies, such as JAK inhibitors and others, according to the authors.
SOURCE:
The lead author of the study was Hemin Lee, MD, MPH, Brigham and Women’s Hospital, Boston. The study was published online in JAMA Dermatology.
LIMITATIONS:
The use of insurance claims data did not allow analysis of over-the-counter medications and treatments, and the lack of a single ICD-10 code for defining AA could have resulted in misclassification of outcomes.
DISCLOSURES:
The study received no outside funding, and Dr. Lee had no disclosures. One author had disclosures that included receiving personal fees from Pfizer and Concert outside of the submitted study and participating in alopecia-related trials with Lilly, Concert, Aclaris, and Incyte. Another author’s disclosures included receiving personal fees from companies that included Pfizer, Concert, Lilly, and AbbVie. No other disclosures were reported.
A version of this article first appeared on Medscape.com.
TOPLINE:
using data from more than 45,000 individuals.
METHODOLOGY:
- The study population included 45,483 adults aged 18 years and older with new diagnoses of AA between Oct. 15, 2015, and Feb. 28, 2020. Data were from a large U.S. health care database that included medical and pharmacy claims.
- The mean age of the participants was 43.8 years, and 65.7% were female.
- The researchers measured variables that might relate to AA and its treatment patterns within 1 year of starting the study and during the first year of the study, with data collected at 1, 42, 84, and 365 days after study entry.
TAKEAWAYS:
- During the first year after diagnosis, 66.4% of patients received at least one treatment for AA at one or more time points.
- At 1 year, 71.8% of patients were not receiving any active treatment for AA.
- Among those who received treatment, intralesional injections were the most often prescribed therapy (41.8% of patients), followed by topical corticosteroids (40.9%), intramuscular corticosteroids (38.1%), and oral corticosteroids (20.6%).
- Patients diagnosed with either alopecia totalis or alopecia universalis were significantly less likely to receive intralesional steroids and significantly more likely to receive topical corticosteroids than those without these diagnoses (11.1% vs. 44.1% and 25.4% vs. 42.1, respectively).
IN PRACTICE:
The results highlight the need to determine why so many alopecia patients with AA were no longer on treatment after 1 year, although treatment trends may change with the emergence of new therapies, such as JAK inhibitors and others, according to the authors.
SOURCE:
The lead author of the study was Hemin Lee, MD, MPH, Brigham and Women’s Hospital, Boston. The study was published online in JAMA Dermatology.
LIMITATIONS:
The use of insurance claims data did not allow analysis of over-the-counter medications and treatments, and the lack of a single ICD-10 code for defining AA could have resulted in misclassification of outcomes.
DISCLOSURES:
The study received no outside funding, and Dr. Lee had no disclosures. One author had disclosures that included receiving personal fees from Pfizer and Concert outside of the submitted study and participating in alopecia-related trials with Lilly, Concert, Aclaris, and Incyte. Another author’s disclosures included receiving personal fees from companies that included Pfizer, Concert, Lilly, and AbbVie. No other disclosures were reported.
A version of this article first appeared on Medscape.com.
FROM JAMA DERMATOLOGY
Overburdened: Health care workers more likely to die by suicide
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study.
If you run into a health care provider these days and ask, “How are you doing?” you’re likely to get a response like this one: “You know, hanging in there.” You smile and move on. But it may be time to go a step further. If you ask that next question – “No, really, how are you doing?” Well, you might need to carve out some time.
It’s been a rough few years for those of us in the health care professions. Our lives, dominated by COVID-related concerns at home, were equally dominated by COVID concerns at work. On the job, there were fewer and fewer of us around as exploitation and COVID-related stressors led doctors, nurses, and others to leave the profession entirely or take early retirement. Even now, I’m not sure we’ve recovered. Staffing in the hospitals is still a huge problem, and the persistence of impersonal meetings via teleconference – which not only prevent any sort of human connection but, audaciously, run from one into another without a break – robs us of even the subtle joy of walking from one hallway to another for 5 minutes of reflection before sitting down to view the next hastily cobbled together PowerPoint.
I’m speaking in generalities, of course.
I’m talking about how bad things are now because, in truth, they’ve never been great. And that may be why health care workers – people with jobs focused on serving others – are nevertheless at substantially increased risk for suicide.
Analyses through the years have shown that physicians tend to have higher rates of death from suicide than the general population. There are reasons for this that may not entirely be because of work-related stress. Doctors’ suicide attempts are more often lethal – we know what is likely to work, after all.
And, according to this paper in JAMA, it is those people who may be suffering most of all.
The study is a nationally representative sample based on the 2008 American Community Survey. Records were linked to the National Death Index through 2019.
Survey respondents were classified into five categories of health care worker, as you can see here. And 1,666,000 non–health care workers served as the control group.
Let’s take a look at the numbers.
I’m showing you age- and sex-standardized rates of death from suicide, starting with non–health care workers. In this study, physicians have similar rates of death from suicide to the general population. Nurses have higher rates, but health care support workers – nurses’ aides, home health aides – have rates nearly twice that of the general population.
Only social and behavioral health workers had rates lower than those in the general population, perhaps because they know how to access life-saving resources.
Of course, these groups differ in a lot of ways – education and income, for example. But even after adjustment for these factors as well as for sex, race, and marital status, the results persist. The only group with even a trend toward lower suicide rates are social and behavioral health workers.
There has been much hand-wringing about rates of physician suicide in the past. It is still a very real problem. But this paper finally highlights that there is a lot more to the health care profession than physicians. It’s time we acknowledge and support the people in our profession who seem to be suffering more than any of us: the aides, the techs, the support staff – the overworked and underpaid who have to deal with all the stresses that physicians like me face and then some.
There’s more to suicide risk than just your job; I know that. Family matters. Relationships matter. Medical and psychiatric illnesses matter. But to ignore this problem when it is right here, in our own house so to speak, can’t continue.
Might I suggest we start by asking someone in our profession – whether doctor, nurse, aide, or tech – how they are doing. How they are really doing. And when we are done listening, we use what we hear to advocate for real change.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study.
If you run into a health care provider these days and ask, “How are you doing?” you’re likely to get a response like this one: “You know, hanging in there.” You smile and move on. But it may be time to go a step further. If you ask that next question – “No, really, how are you doing?” Well, you might need to carve out some time.
It’s been a rough few years for those of us in the health care professions. Our lives, dominated by COVID-related concerns at home, were equally dominated by COVID concerns at work. On the job, there were fewer and fewer of us around as exploitation and COVID-related stressors led doctors, nurses, and others to leave the profession entirely or take early retirement. Even now, I’m not sure we’ve recovered. Staffing in the hospitals is still a huge problem, and the persistence of impersonal meetings via teleconference – which not only prevent any sort of human connection but, audaciously, run from one into another without a break – robs us of even the subtle joy of walking from one hallway to another for 5 minutes of reflection before sitting down to view the next hastily cobbled together PowerPoint.
I’m speaking in generalities, of course.
I’m talking about how bad things are now because, in truth, they’ve never been great. And that may be why health care workers – people with jobs focused on serving others – are nevertheless at substantially increased risk for suicide.
Analyses through the years have shown that physicians tend to have higher rates of death from suicide than the general population. There are reasons for this that may not entirely be because of work-related stress. Doctors’ suicide attempts are more often lethal – we know what is likely to work, after all.
And, according to this paper in JAMA, it is those people who may be suffering most of all.
The study is a nationally representative sample based on the 2008 American Community Survey. Records were linked to the National Death Index through 2019.
Survey respondents were classified into five categories of health care worker, as you can see here. And 1,666,000 non–health care workers served as the control group.
Let’s take a look at the numbers.
I’m showing you age- and sex-standardized rates of death from suicide, starting with non–health care workers. In this study, physicians have similar rates of death from suicide to the general population. Nurses have higher rates, but health care support workers – nurses’ aides, home health aides – have rates nearly twice that of the general population.
Only social and behavioral health workers had rates lower than those in the general population, perhaps because they know how to access life-saving resources.
Of course, these groups differ in a lot of ways – education and income, for example. But even after adjustment for these factors as well as for sex, race, and marital status, the results persist. The only group with even a trend toward lower suicide rates are social and behavioral health workers.
There has been much hand-wringing about rates of physician suicide in the past. It is still a very real problem. But this paper finally highlights that there is a lot more to the health care profession than physicians. It’s time we acknowledge and support the people in our profession who seem to be suffering more than any of us: the aides, the techs, the support staff – the overworked and underpaid who have to deal with all the stresses that physicians like me face and then some.
There’s more to suicide risk than just your job; I know that. Family matters. Relationships matter. Medical and psychiatric illnesses matter. But to ignore this problem when it is right here, in our own house so to speak, can’t continue.
Might I suggest we start by asking someone in our profession – whether doctor, nurse, aide, or tech – how they are doing. How they are really doing. And when we are done listening, we use what we hear to advocate for real change.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study.
If you run into a health care provider these days and ask, “How are you doing?” you’re likely to get a response like this one: “You know, hanging in there.” You smile and move on. But it may be time to go a step further. If you ask that next question – “No, really, how are you doing?” Well, you might need to carve out some time.
It’s been a rough few years for those of us in the health care professions. Our lives, dominated by COVID-related concerns at home, were equally dominated by COVID concerns at work. On the job, there were fewer and fewer of us around as exploitation and COVID-related stressors led doctors, nurses, and others to leave the profession entirely or take early retirement. Even now, I’m not sure we’ve recovered. Staffing in the hospitals is still a huge problem, and the persistence of impersonal meetings via teleconference – which not only prevent any sort of human connection but, audaciously, run from one into another without a break – robs us of even the subtle joy of walking from one hallway to another for 5 minutes of reflection before sitting down to view the next hastily cobbled together PowerPoint.
I’m speaking in generalities, of course.
I’m talking about how bad things are now because, in truth, they’ve never been great. And that may be why health care workers – people with jobs focused on serving others – are nevertheless at substantially increased risk for suicide.
Analyses through the years have shown that physicians tend to have higher rates of death from suicide than the general population. There are reasons for this that may not entirely be because of work-related stress. Doctors’ suicide attempts are more often lethal – we know what is likely to work, after all.
And, according to this paper in JAMA, it is those people who may be suffering most of all.
The study is a nationally representative sample based on the 2008 American Community Survey. Records were linked to the National Death Index through 2019.
Survey respondents were classified into five categories of health care worker, as you can see here. And 1,666,000 non–health care workers served as the control group.
Let’s take a look at the numbers.
I’m showing you age- and sex-standardized rates of death from suicide, starting with non–health care workers. In this study, physicians have similar rates of death from suicide to the general population. Nurses have higher rates, but health care support workers – nurses’ aides, home health aides – have rates nearly twice that of the general population.
Only social and behavioral health workers had rates lower than those in the general population, perhaps because they know how to access life-saving resources.
Of course, these groups differ in a lot of ways – education and income, for example. But even after adjustment for these factors as well as for sex, race, and marital status, the results persist. The only group with even a trend toward lower suicide rates are social and behavioral health workers.
There has been much hand-wringing about rates of physician suicide in the past. It is still a very real problem. But this paper finally highlights that there is a lot more to the health care profession than physicians. It’s time we acknowledge and support the people in our profession who seem to be suffering more than any of us: the aides, the techs, the support staff – the overworked and underpaid who have to deal with all the stresses that physicians like me face and then some.
There’s more to suicide risk than just your job; I know that. Family matters. Relationships matter. Medical and psychiatric illnesses matter. But to ignore this problem when it is right here, in our own house so to speak, can’t continue.
Might I suggest we start by asking someone in our profession – whether doctor, nurse, aide, or tech – how they are doing. How they are really doing. And when we are done listening, we use what we hear to advocate for real change.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
How to get paid if your patient passes on
The death of a patient comes with many challenges for physicians, including a range of emotional and professional issues. Beyond those concerns,
“When a patient passes away, obviously there is, unfortunately, a lot of paperwork and stress for families, and it’s a very difficult situation,” says Shikha Jain, MD, an oncologist and associate professor of medicine at the University of Illinois at Chicago. “Talking about finances in that moment can be difficult and uncomfortable, and one thing I’d recommend is that the physicians themselves not get involved.”
Instead, Dr. Jain said, someone in the billing department in the practice or the hospital should take a lead on dealing with any outstanding debts.
“That doctor-patient relationship is a very precious relationship, so you don’t want to mix that financial aspect of providing care with the doctor-patient relationship,” Dr. Jain said. “That’s one thing that’s really important.”
The best approach in such situations is for practices to have a standing policy in place that dictates how to handle bills once a patient has died.
In most cases, the executor of the patient’s will must inform all creditors, including doctors, that the decedent has died, but sometimes there’s a delay.
Hoping the doctor’s office writes it off
“Even though the person in charge of the estate is supposed to contact the doctor’s office and let them know when a patient has passed, that doesn’t always happen,” says Hope Wen, head of billing at practice management platform Soundry Health. “It can be very challenging to track down that information, and sometimes they’re just crossing their fingers hoping that the doctor’s office will just write off the balance, which they often do.”
Some offices use a service that compares accounts receivable lists to Social Security death files and state records to identify deaths more quickly. Some physicians might also use a debt collection agency or an attorney who has experience collecting decedent debts and dealing with executors and probate courts.
Once the practice becomes aware that a patient has died, it can no longer send communications to the name and address on file, although it can continue to go through the billing process with the insurer for any bills incurred up to the date of the death.
At that point, the estate becomes responsible for the debt, and all communication must go to the executor of the estate (in some states, this might be called a personal representative). The office can reach out to any contacts on file to see if they are able to identify the executor.
“You want to do that in a compassionate way,” says Jack Brown III, JD, MBA, president of Gulf Coast Collection Bureau. “You’ll tell them you’re sorry for their loss, but you’re wondering who is responsible for the estate. Once you’ve identified that person and gotten their letter of administration from the probate court or a power of attorney, then you can speak with that person as if they were the patient.”
The names of executors are also public record and are available through the probate court (sometimes called the surrogate court) in the county where the decedent lived.
“Even if there’s no will or no executive named, the court will appoint an administrator for the estate, which is usually a family member,” said Robert Bernstein, an estate lawyer in Parsippany, N.J. “Their information will be on file in the court.”
Insurance coverage
Typically, insurance will pay for treatment (after deductibles and copays) up until the date of the patient’s death. But, of course, it can take months for some insurance companies to make their final payments, allowing physicians to know exactly how much they’re owed by that estate. In such cases, it’s important for physicians to know the rules in the decedent’s state for how long they have to file a claim.
Most states require that claims occur within 6-9 months of the person’s death. However, in some states, claimants can continue to file for much longer if the estate has not yet paid out all of its assets.
“Sometimes there is real estate to sell or a business to wind down, and it can take years for the estate to distribute all of the assets,” Mr. Bernstein says. “If it’s a year later and they still haven’t distributed the assets, the physician can still file the claim and should be paid.”
In some cases, especially if the decedent received compassionate, quality care, their family will want to make good on any outstanding debts to the health care providers who took care of their loved ones in their final days. In other cases, especially when a family member has had a long illness, their assets have been depleted over time or were transferred to other family members so that there is little left in the estate itself when the patient dies.
Regardless of other circumstances, the estate alone is responsible for such payments, and family members, including spouses and children, typically have no liability. (Though rarely enforced, some states do have filial responsibility laws that could hold children responsible for their parents’ debts, including unpaid medical bills. In addition, states with community property laws might require a surviving spouse to cover their partner’s debt, even after death.)
The probate process varies from state to state, but in general, the probate system and the executor will gather all existing assets and then notify all creditors about how to submit a claim. Typically, the claim will need to include information about how much is owed and documentation, such as bills and an explanation of benefits to back up the claim. It should be borne in mind that even those who’ve passed away have privacy protections under the Health Insurance Portability and Accountability Act, so practices must be careful as to how much information they’re sharing through their claim.
Once the estate has received all the claims, the executor will follow a priority of claims, starting with secured creditors. Typically, medical bills, especially those incurred in the last 90 days of the decedent’s life, have priority in the probate process, Mr. Brown says.
How to minimize losses
In that case, the practice would write off the unpaid debt as a business loss. If there are not enough assets in the estate to pay all claims, the executor will follow a state schedule that apportions those assets that are available.
There are some steps that practices can take to protect themselves from incurring such losses. For example, before beginning treatment, practices might consider asking patients to name a guarantor, who will essentially promise to cover any outstanding debts that the patient incurs.
To be binding, the office will need a signature from both the patient and the guarantor. Some offices may also keep a patient credit card number on file with written authorization that they can use to pay bills that are past due, although this payment method would no longer be valid after a patient dies.
While it’s important for all physicians to document and verify the financial information for their patients, oncologists often must consider an additional layer of fiduciary responsibility when it comes to their patients. Ms. Wen suggests that oncology offices check in with insurance companies to determine whether a patient has exhausted their benefits.
“That can happen with cancer patients, depending on how long they’ve been receiving treatment and what type of treatment they’ve been getting,” she said. “Some of the clinical trials, insurance will pay for them, but they’re really expensive and can get toward that max. So knowing where they are with their insurance coverage is big.”
When time is of the essence, some patients will choose to go forward with a treatment before receiving insurance approval. In those cases, the office must have an additional conversation in which the costs of the treatment are discussed. The office should obtain written confirmation of who will pay if the insurer does not, Ms. Wen said. While it’s the patient’s responsibility to keep track of their insurance benefits, oncology practices and hospitals must also exercise due diligence in monitoring the benefits that are available.
“That’s part of their contract with insurance companies if they’re in network, helping patients understand their benefits,” Ms. Wen saids.
It’s also important for practices to keep clear, consistent records to make it easier to identify outstanding bills and the correct contact information for them. If bills had gone unpaid prior to a patient’s death and the office started legal action and received a judgment, that claim would typically go ahead of other creditors’ claims.
Dr. Jain says that some practices might also consider keeping a financial adviser or social worker on staff who can assist patients and their families with understanding their out-of-pocket costs for treatment.
“Financial toxicity in oncology and medical care is a very real problem,” she says. “At the beginning of the relationship, I recommend that my patients get set up with a financial specialist that can help them navigate that aspect, not only when a patient passes away but during the process of receiving treatment, so they’re not shocked by the bills.”
A version of this article first appeared on Medscape.com.
The death of a patient comes with many challenges for physicians, including a range of emotional and professional issues. Beyond those concerns,
“When a patient passes away, obviously there is, unfortunately, a lot of paperwork and stress for families, and it’s a very difficult situation,” says Shikha Jain, MD, an oncologist and associate professor of medicine at the University of Illinois at Chicago. “Talking about finances in that moment can be difficult and uncomfortable, and one thing I’d recommend is that the physicians themselves not get involved.”
Instead, Dr. Jain said, someone in the billing department in the practice or the hospital should take a lead on dealing with any outstanding debts.
“That doctor-patient relationship is a very precious relationship, so you don’t want to mix that financial aspect of providing care with the doctor-patient relationship,” Dr. Jain said. “That’s one thing that’s really important.”
The best approach in such situations is for practices to have a standing policy in place that dictates how to handle bills once a patient has died.
In most cases, the executor of the patient’s will must inform all creditors, including doctors, that the decedent has died, but sometimes there’s a delay.
Hoping the doctor’s office writes it off
“Even though the person in charge of the estate is supposed to contact the doctor’s office and let them know when a patient has passed, that doesn’t always happen,” says Hope Wen, head of billing at practice management platform Soundry Health. “It can be very challenging to track down that information, and sometimes they’re just crossing their fingers hoping that the doctor’s office will just write off the balance, which they often do.”
Some offices use a service that compares accounts receivable lists to Social Security death files and state records to identify deaths more quickly. Some physicians might also use a debt collection agency or an attorney who has experience collecting decedent debts and dealing with executors and probate courts.
Once the practice becomes aware that a patient has died, it can no longer send communications to the name and address on file, although it can continue to go through the billing process with the insurer for any bills incurred up to the date of the death.
At that point, the estate becomes responsible for the debt, and all communication must go to the executor of the estate (in some states, this might be called a personal representative). The office can reach out to any contacts on file to see if they are able to identify the executor.
“You want to do that in a compassionate way,” says Jack Brown III, JD, MBA, president of Gulf Coast Collection Bureau. “You’ll tell them you’re sorry for their loss, but you’re wondering who is responsible for the estate. Once you’ve identified that person and gotten their letter of administration from the probate court or a power of attorney, then you can speak with that person as if they were the patient.”
The names of executors are also public record and are available through the probate court (sometimes called the surrogate court) in the county where the decedent lived.
“Even if there’s no will or no executive named, the court will appoint an administrator for the estate, which is usually a family member,” said Robert Bernstein, an estate lawyer in Parsippany, N.J. “Their information will be on file in the court.”
Insurance coverage
Typically, insurance will pay for treatment (after deductibles and copays) up until the date of the patient’s death. But, of course, it can take months for some insurance companies to make their final payments, allowing physicians to know exactly how much they’re owed by that estate. In such cases, it’s important for physicians to know the rules in the decedent’s state for how long they have to file a claim.
Most states require that claims occur within 6-9 months of the person’s death. However, in some states, claimants can continue to file for much longer if the estate has not yet paid out all of its assets.
“Sometimes there is real estate to sell or a business to wind down, and it can take years for the estate to distribute all of the assets,” Mr. Bernstein says. “If it’s a year later and they still haven’t distributed the assets, the physician can still file the claim and should be paid.”
In some cases, especially if the decedent received compassionate, quality care, their family will want to make good on any outstanding debts to the health care providers who took care of their loved ones in their final days. In other cases, especially when a family member has had a long illness, their assets have been depleted over time or were transferred to other family members so that there is little left in the estate itself when the patient dies.
Regardless of other circumstances, the estate alone is responsible for such payments, and family members, including spouses and children, typically have no liability. (Though rarely enforced, some states do have filial responsibility laws that could hold children responsible for their parents’ debts, including unpaid medical bills. In addition, states with community property laws might require a surviving spouse to cover their partner’s debt, even after death.)
The probate process varies from state to state, but in general, the probate system and the executor will gather all existing assets and then notify all creditors about how to submit a claim. Typically, the claim will need to include information about how much is owed and documentation, such as bills and an explanation of benefits to back up the claim. It should be borne in mind that even those who’ve passed away have privacy protections under the Health Insurance Portability and Accountability Act, so practices must be careful as to how much information they’re sharing through their claim.
Once the estate has received all the claims, the executor will follow a priority of claims, starting with secured creditors. Typically, medical bills, especially those incurred in the last 90 days of the decedent’s life, have priority in the probate process, Mr. Brown says.
How to minimize losses
In that case, the practice would write off the unpaid debt as a business loss. If there are not enough assets in the estate to pay all claims, the executor will follow a state schedule that apportions those assets that are available.
There are some steps that practices can take to protect themselves from incurring such losses. For example, before beginning treatment, practices might consider asking patients to name a guarantor, who will essentially promise to cover any outstanding debts that the patient incurs.
To be binding, the office will need a signature from both the patient and the guarantor. Some offices may also keep a patient credit card number on file with written authorization that they can use to pay bills that are past due, although this payment method would no longer be valid after a patient dies.
While it’s important for all physicians to document and verify the financial information for their patients, oncologists often must consider an additional layer of fiduciary responsibility when it comes to their patients. Ms. Wen suggests that oncology offices check in with insurance companies to determine whether a patient has exhausted their benefits.
“That can happen with cancer patients, depending on how long they’ve been receiving treatment and what type of treatment they’ve been getting,” she said. “Some of the clinical trials, insurance will pay for them, but they’re really expensive and can get toward that max. So knowing where they are with their insurance coverage is big.”
When time is of the essence, some patients will choose to go forward with a treatment before receiving insurance approval. In those cases, the office must have an additional conversation in which the costs of the treatment are discussed. The office should obtain written confirmation of who will pay if the insurer does not, Ms. Wen said. While it’s the patient’s responsibility to keep track of their insurance benefits, oncology practices and hospitals must also exercise due diligence in monitoring the benefits that are available.
“That’s part of their contract with insurance companies if they’re in network, helping patients understand their benefits,” Ms. Wen saids.
It’s also important for practices to keep clear, consistent records to make it easier to identify outstanding bills and the correct contact information for them. If bills had gone unpaid prior to a patient’s death and the office started legal action and received a judgment, that claim would typically go ahead of other creditors’ claims.
Dr. Jain says that some practices might also consider keeping a financial adviser or social worker on staff who can assist patients and their families with understanding their out-of-pocket costs for treatment.
“Financial toxicity in oncology and medical care is a very real problem,” she says. “At the beginning of the relationship, I recommend that my patients get set up with a financial specialist that can help them navigate that aspect, not only when a patient passes away but during the process of receiving treatment, so they’re not shocked by the bills.”
A version of this article first appeared on Medscape.com.
The death of a patient comes with many challenges for physicians, including a range of emotional and professional issues. Beyond those concerns,
“When a patient passes away, obviously there is, unfortunately, a lot of paperwork and stress for families, and it’s a very difficult situation,” says Shikha Jain, MD, an oncologist and associate professor of medicine at the University of Illinois at Chicago. “Talking about finances in that moment can be difficult and uncomfortable, and one thing I’d recommend is that the physicians themselves not get involved.”
Instead, Dr. Jain said, someone in the billing department in the practice or the hospital should take a lead on dealing with any outstanding debts.
“That doctor-patient relationship is a very precious relationship, so you don’t want to mix that financial aspect of providing care with the doctor-patient relationship,” Dr. Jain said. “That’s one thing that’s really important.”
The best approach in such situations is for practices to have a standing policy in place that dictates how to handle bills once a patient has died.
In most cases, the executor of the patient’s will must inform all creditors, including doctors, that the decedent has died, but sometimes there’s a delay.
Hoping the doctor’s office writes it off
“Even though the person in charge of the estate is supposed to contact the doctor’s office and let them know when a patient has passed, that doesn’t always happen,” says Hope Wen, head of billing at practice management platform Soundry Health. “It can be very challenging to track down that information, and sometimes they’re just crossing their fingers hoping that the doctor’s office will just write off the balance, which they often do.”
Some offices use a service that compares accounts receivable lists to Social Security death files and state records to identify deaths more quickly. Some physicians might also use a debt collection agency or an attorney who has experience collecting decedent debts and dealing with executors and probate courts.
Once the practice becomes aware that a patient has died, it can no longer send communications to the name and address on file, although it can continue to go through the billing process with the insurer for any bills incurred up to the date of the death.
At that point, the estate becomes responsible for the debt, and all communication must go to the executor of the estate (in some states, this might be called a personal representative). The office can reach out to any contacts on file to see if they are able to identify the executor.
“You want to do that in a compassionate way,” says Jack Brown III, JD, MBA, president of Gulf Coast Collection Bureau. “You’ll tell them you’re sorry for their loss, but you’re wondering who is responsible for the estate. Once you’ve identified that person and gotten their letter of administration from the probate court or a power of attorney, then you can speak with that person as if they were the patient.”
The names of executors are also public record and are available through the probate court (sometimes called the surrogate court) in the county where the decedent lived.
“Even if there’s no will or no executive named, the court will appoint an administrator for the estate, which is usually a family member,” said Robert Bernstein, an estate lawyer in Parsippany, N.J. “Their information will be on file in the court.”
Insurance coverage
Typically, insurance will pay for treatment (after deductibles and copays) up until the date of the patient’s death. But, of course, it can take months for some insurance companies to make their final payments, allowing physicians to know exactly how much they’re owed by that estate. In such cases, it’s important for physicians to know the rules in the decedent’s state for how long they have to file a claim.
Most states require that claims occur within 6-9 months of the person’s death. However, in some states, claimants can continue to file for much longer if the estate has not yet paid out all of its assets.
“Sometimes there is real estate to sell or a business to wind down, and it can take years for the estate to distribute all of the assets,” Mr. Bernstein says. “If it’s a year later and they still haven’t distributed the assets, the physician can still file the claim and should be paid.”
In some cases, especially if the decedent received compassionate, quality care, their family will want to make good on any outstanding debts to the health care providers who took care of their loved ones in their final days. In other cases, especially when a family member has had a long illness, their assets have been depleted over time or were transferred to other family members so that there is little left in the estate itself when the patient dies.
Regardless of other circumstances, the estate alone is responsible for such payments, and family members, including spouses and children, typically have no liability. (Though rarely enforced, some states do have filial responsibility laws that could hold children responsible for their parents’ debts, including unpaid medical bills. In addition, states with community property laws might require a surviving spouse to cover their partner’s debt, even after death.)
The probate process varies from state to state, but in general, the probate system and the executor will gather all existing assets and then notify all creditors about how to submit a claim. Typically, the claim will need to include information about how much is owed and documentation, such as bills and an explanation of benefits to back up the claim. It should be borne in mind that even those who’ve passed away have privacy protections under the Health Insurance Portability and Accountability Act, so practices must be careful as to how much information they’re sharing through their claim.
Once the estate has received all the claims, the executor will follow a priority of claims, starting with secured creditors. Typically, medical bills, especially those incurred in the last 90 days of the decedent’s life, have priority in the probate process, Mr. Brown says.
How to minimize losses
In that case, the practice would write off the unpaid debt as a business loss. If there are not enough assets in the estate to pay all claims, the executor will follow a state schedule that apportions those assets that are available.
There are some steps that practices can take to protect themselves from incurring such losses. For example, before beginning treatment, practices might consider asking patients to name a guarantor, who will essentially promise to cover any outstanding debts that the patient incurs.
To be binding, the office will need a signature from both the patient and the guarantor. Some offices may also keep a patient credit card number on file with written authorization that they can use to pay bills that are past due, although this payment method would no longer be valid after a patient dies.
While it’s important for all physicians to document and verify the financial information for their patients, oncologists often must consider an additional layer of fiduciary responsibility when it comes to their patients. Ms. Wen suggests that oncology offices check in with insurance companies to determine whether a patient has exhausted their benefits.
“That can happen with cancer patients, depending on how long they’ve been receiving treatment and what type of treatment they’ve been getting,” she said. “Some of the clinical trials, insurance will pay for them, but they’re really expensive and can get toward that max. So knowing where they are with their insurance coverage is big.”
When time is of the essence, some patients will choose to go forward with a treatment before receiving insurance approval. In those cases, the office must have an additional conversation in which the costs of the treatment are discussed. The office should obtain written confirmation of who will pay if the insurer does not, Ms. Wen said. While it’s the patient’s responsibility to keep track of their insurance benefits, oncology practices and hospitals must also exercise due diligence in monitoring the benefits that are available.
“That’s part of their contract with insurance companies if they’re in network, helping patients understand their benefits,” Ms. Wen saids.
It’s also important for practices to keep clear, consistent records to make it easier to identify outstanding bills and the correct contact information for them. If bills had gone unpaid prior to a patient’s death and the office started legal action and received a judgment, that claim would typically go ahead of other creditors’ claims.
Dr. Jain says that some practices might also consider keeping a financial adviser or social worker on staff who can assist patients and their families with understanding their out-of-pocket costs for treatment.
“Financial toxicity in oncology and medical care is a very real problem,” she says. “At the beginning of the relationship, I recommend that my patients get set up with a financial specialist that can help them navigate that aspect, not only when a patient passes away but during the process of receiving treatment, so they’re not shocked by the bills.”
A version of this article first appeared on Medscape.com.
‘Old school’ laser resurfacing remains an effective option for rejuvenation
SAN DIEGO – , according to Arisa E. Ortiz, MD.
“Fractional resurfacing is great because there is less downtime, but the results are not as dramatic as with fully ablative resurfacing,” Dr. Ortiz, director of laser and cosmetic dermatology at the University of California, San Diego, said at the annual Masters of Aesthetics Symposium. In her practice, she said, “we do a combination,” which can include “fully ablative around the mouth and eyes and fractional everywhere else.”
Key drawbacks to fully ablative laser resurfacing include significant downtime and extensive wound care, “so it’s not for everybody,” she said. Prolonged erythema following treatment is expected, “so patients need to plan for this. It can last 3-4 months, and it will continue to fade and can be covered up with makeup, but it does last a while,” she noted. “One of the things that made ablative resurfacing fall out of favor was the delayed and permanent hypopigmentation where there’s a stark line of demarcation because you can’t treat the neck [with this modality], so patients have this pearly white looking face that appears 6 months after the treatment,” she added.
Preoperatively, Dr. Ortiz asks patients what other cosmetic procedures they have had in the past. For example, if they have had a facelift, they might have neck skin on their jawline, which will react differently to fully ablative resurfacing than facial skin. “I don’t perform fully ablative resurfacing on the neck or body, or in patients with darker skin types,” she said.
To optimize results, she recommends pretreatment of the area with a neuromodulator a week or 2 before the procedure, “so that they’re not actively contracting and recreating creases,” she said. Studies, she noted, have shown that this approach results in better outcomes. She also asks patients to apply a tripeptide serum daily a week or 2 prior to their procedure to stimulate wound healing and collagen remodeling.
For antibiotic and antiviral prophylaxis, Dr. Ortiz typically prescribes doxycycline 100 mg b.i.d. for 7 days and valacyclovir 500 mg b.i.d. for 7 days and asks patients to start the course the night before the procedure. “If they break through the antiviral, I increase to zoster dosing,” she said. “I make sure they have my cell phone number and call me right away if that happens. I don’t routinely prescribe an antifungal, but you can if you want to.”
For anesthesia, Dr. Ortiz applies lidocaine 23%/tetracaine 7% an hour before the procedure and performs nerve blocks at the mentalis, infraorbital, supraorbital, and nasalis muscles. “I also do local infiltration with a three-pronged Mesoram adapter,” she said. “That has changed the comfort level for these patients. I don’t offer any sedation in my practice but that is an option if you have it available. If you’re going to be resurfacing within the orbital rim you need to know how to place corneal shields. Only use injectable lidocaine in this area because if topical lidocaine gets into the eye, it can cause a chemical corneal abrasion. Nothing happens to their vision permanently, but it’s extremely painful for 24-48 hours.”
Dr. Ortiz described postoperative wound care as “the hardest part” of fully ablative laser resurfacing treatments. The treated area will look “bloody and crusty” for 1-2 weeks. She instructs patients to do vinegar soaks four times per day for 2-3 weeks, “depending on how quickly they heal,” she said. She also counsels patients to apply petrolatum ointment to the area and provides them with a bottle of hypochlorous acid spray, an antiseptic – which also helps with the itching they may experience. “They need to avoid the sun, so I recommend full face visors,” she added.
In her clinical experience, postoperative pain medications are not required. “If the patient calls you on day 3 with increased pain, that’s usually a sign of infection; don’t ignore that,” said Dr. Ortiz, who is also president-elect of the American Society for Laser Medicine and Surgery. In a case of suspected infection, she asks the patient to come in right away, and obtains a bacterial culture. “If they break through the doxycycline, it’s usually a gram-negative infection, so I’ll treat them prophylactically for that,” she said.
“Significant itching may be a sign of Candida infection,” she noted. “Because the epidermis has been disrupted, if they have systemic symptoms then you want to consider IV antibiotics because the infection can spread rapidly.”
Dr. Ortiz disclosed having financial relationships with several pharmaceutical and device companies. She is also cochair of the MOAS.
SAN DIEGO – , according to Arisa E. Ortiz, MD.
“Fractional resurfacing is great because there is less downtime, but the results are not as dramatic as with fully ablative resurfacing,” Dr. Ortiz, director of laser and cosmetic dermatology at the University of California, San Diego, said at the annual Masters of Aesthetics Symposium. In her practice, she said, “we do a combination,” which can include “fully ablative around the mouth and eyes and fractional everywhere else.”
Key drawbacks to fully ablative laser resurfacing include significant downtime and extensive wound care, “so it’s not for everybody,” she said. Prolonged erythema following treatment is expected, “so patients need to plan for this. It can last 3-4 months, and it will continue to fade and can be covered up with makeup, but it does last a while,” she noted. “One of the things that made ablative resurfacing fall out of favor was the delayed and permanent hypopigmentation where there’s a stark line of demarcation because you can’t treat the neck [with this modality], so patients have this pearly white looking face that appears 6 months after the treatment,” she added.
Preoperatively, Dr. Ortiz asks patients what other cosmetic procedures they have had in the past. For example, if they have had a facelift, they might have neck skin on their jawline, which will react differently to fully ablative resurfacing than facial skin. “I don’t perform fully ablative resurfacing on the neck or body, or in patients with darker skin types,” she said.
To optimize results, she recommends pretreatment of the area with a neuromodulator a week or 2 before the procedure, “so that they’re not actively contracting and recreating creases,” she said. Studies, she noted, have shown that this approach results in better outcomes. She also asks patients to apply a tripeptide serum daily a week or 2 prior to their procedure to stimulate wound healing and collagen remodeling.
For antibiotic and antiviral prophylaxis, Dr. Ortiz typically prescribes doxycycline 100 mg b.i.d. for 7 days and valacyclovir 500 mg b.i.d. for 7 days and asks patients to start the course the night before the procedure. “If they break through the antiviral, I increase to zoster dosing,” she said. “I make sure they have my cell phone number and call me right away if that happens. I don’t routinely prescribe an antifungal, but you can if you want to.”
For anesthesia, Dr. Ortiz applies lidocaine 23%/tetracaine 7% an hour before the procedure and performs nerve blocks at the mentalis, infraorbital, supraorbital, and nasalis muscles. “I also do local infiltration with a three-pronged Mesoram adapter,” she said. “That has changed the comfort level for these patients. I don’t offer any sedation in my practice but that is an option if you have it available. If you’re going to be resurfacing within the orbital rim you need to know how to place corneal shields. Only use injectable lidocaine in this area because if topical lidocaine gets into the eye, it can cause a chemical corneal abrasion. Nothing happens to their vision permanently, but it’s extremely painful for 24-48 hours.”
Dr. Ortiz described postoperative wound care as “the hardest part” of fully ablative laser resurfacing treatments. The treated area will look “bloody and crusty” for 1-2 weeks. She instructs patients to do vinegar soaks four times per day for 2-3 weeks, “depending on how quickly they heal,” she said. She also counsels patients to apply petrolatum ointment to the area and provides them with a bottle of hypochlorous acid spray, an antiseptic – which also helps with the itching they may experience. “They need to avoid the sun, so I recommend full face visors,” she added.
In her clinical experience, postoperative pain medications are not required. “If the patient calls you on day 3 with increased pain, that’s usually a sign of infection; don’t ignore that,” said Dr. Ortiz, who is also president-elect of the American Society for Laser Medicine and Surgery. In a case of suspected infection, she asks the patient to come in right away, and obtains a bacterial culture. “If they break through the doxycycline, it’s usually a gram-negative infection, so I’ll treat them prophylactically for that,” she said.
“Significant itching may be a sign of Candida infection,” she noted. “Because the epidermis has been disrupted, if they have systemic symptoms then you want to consider IV antibiotics because the infection can spread rapidly.”
Dr. Ortiz disclosed having financial relationships with several pharmaceutical and device companies. She is also cochair of the MOAS.
SAN DIEGO – , according to Arisa E. Ortiz, MD.
“Fractional resurfacing is great because there is less downtime, but the results are not as dramatic as with fully ablative resurfacing,” Dr. Ortiz, director of laser and cosmetic dermatology at the University of California, San Diego, said at the annual Masters of Aesthetics Symposium. In her practice, she said, “we do a combination,” which can include “fully ablative around the mouth and eyes and fractional everywhere else.”
Key drawbacks to fully ablative laser resurfacing include significant downtime and extensive wound care, “so it’s not for everybody,” she said. Prolonged erythema following treatment is expected, “so patients need to plan for this. It can last 3-4 months, and it will continue to fade and can be covered up with makeup, but it does last a while,” she noted. “One of the things that made ablative resurfacing fall out of favor was the delayed and permanent hypopigmentation where there’s a stark line of demarcation because you can’t treat the neck [with this modality], so patients have this pearly white looking face that appears 6 months after the treatment,” she added.
Preoperatively, Dr. Ortiz asks patients what other cosmetic procedures they have had in the past. For example, if they have had a facelift, they might have neck skin on their jawline, which will react differently to fully ablative resurfacing than facial skin. “I don’t perform fully ablative resurfacing on the neck or body, or in patients with darker skin types,” she said.
To optimize results, she recommends pretreatment of the area with a neuromodulator a week or 2 before the procedure, “so that they’re not actively contracting and recreating creases,” she said. Studies, she noted, have shown that this approach results in better outcomes. She also asks patients to apply a tripeptide serum daily a week or 2 prior to their procedure to stimulate wound healing and collagen remodeling.
For antibiotic and antiviral prophylaxis, Dr. Ortiz typically prescribes doxycycline 100 mg b.i.d. for 7 days and valacyclovir 500 mg b.i.d. for 7 days and asks patients to start the course the night before the procedure. “If they break through the antiviral, I increase to zoster dosing,” she said. “I make sure they have my cell phone number and call me right away if that happens. I don’t routinely prescribe an antifungal, but you can if you want to.”
For anesthesia, Dr. Ortiz applies lidocaine 23%/tetracaine 7% an hour before the procedure and performs nerve blocks at the mentalis, infraorbital, supraorbital, and nasalis muscles. “I also do local infiltration with a three-pronged Mesoram adapter,” she said. “That has changed the comfort level for these patients. I don’t offer any sedation in my practice but that is an option if you have it available. If you’re going to be resurfacing within the orbital rim you need to know how to place corneal shields. Only use injectable lidocaine in this area because if topical lidocaine gets into the eye, it can cause a chemical corneal abrasion. Nothing happens to their vision permanently, but it’s extremely painful for 24-48 hours.”
Dr. Ortiz described postoperative wound care as “the hardest part” of fully ablative laser resurfacing treatments. The treated area will look “bloody and crusty” for 1-2 weeks. She instructs patients to do vinegar soaks four times per day for 2-3 weeks, “depending on how quickly they heal,” she said. She also counsels patients to apply petrolatum ointment to the area and provides them with a bottle of hypochlorous acid spray, an antiseptic – which also helps with the itching they may experience. “They need to avoid the sun, so I recommend full face visors,” she added.
In her clinical experience, postoperative pain medications are not required. “If the patient calls you on day 3 with increased pain, that’s usually a sign of infection; don’t ignore that,” said Dr. Ortiz, who is also president-elect of the American Society for Laser Medicine and Surgery. In a case of suspected infection, she asks the patient to come in right away, and obtains a bacterial culture. “If they break through the doxycycline, it’s usually a gram-negative infection, so I’ll treat them prophylactically for that,” she said.
“Significant itching may be a sign of Candida infection,” she noted. “Because the epidermis has been disrupted, if they have systemic symptoms then you want to consider IV antibiotics because the infection can spread rapidly.”
Dr. Ortiz disclosed having financial relationships with several pharmaceutical and device companies. She is also cochair of the MOAS.
AT MOAS 2023