User login
Formerly Skin & Allergy News
ass lick
assault rifle
balls
ballsac
black jack
bleach
Boko Haram
bondage
causas
cheap
child abuse
cocaine
compulsive behaviors
cost of miracles
cunt
Daech
display network stats
drug paraphernalia
explosion
fart
fda and death
fda AND warn
fda AND warning
fda AND warns
feom
fuck
gambling
gfc
gun
human trafficking
humira AND expensive
illegal
ISIL
ISIS
Islamic caliphate
Islamic state
madvocate
masturbation
mixed martial arts
MMA
molestation
national rifle association
NRA
nsfw
nuccitelli
pedophile
pedophilia
poker
porn
porn
pornography
psychedelic drug
recreational drug
sex slave rings
shit
slot machine
snort
substance abuse
terrorism
terrorist
texarkana
Texas hold 'em
UFC
section[contains(@class, 'nav-hidden')]
section[contains(@class, 'nav-hidden active')]
The leading independent newspaper covering dermatology news and commentary.
Study of beliefs about what causes cancer sparks debate
The study, entitled, “Everything Causes Cancer? Beliefs and Attitudes Towards Cancer Prevention Among Anti-Vaxxers, Flat Earthers, and Reptilian Conspiracists: Online Cross Sectional Survey,” was published in the Christmas 2022 issue of The British Medical Journal (BMJ).
The authors explain that they set out to evaluate “the patterns of beliefs about cancer among people who believed in conspiracies, rejected the COVID-19 vaccine, or preferred alternative medicine.”
They sought such people on social media and online chat platforms and asked them questions about real and mythical causes of cancer.
Almost half of survey participants agreed with the statement, “It seems like everything causes cancer.”
Overall, among all participants, awareness of the actual causes of cancer was greater than awareness of the mythical causes of cancer, the authors report. However, awareness of the actual causes of cancer was lower among the unvaccinated and members of conspiracy groups than among their counterparts.
The authors are concerned that their findings suggest “a direct connection between digital misinformation and consequent potential erroneous health decisions, which may represent a further preventable fraction of cancer.”
Backlash and criticism
The study “highlights the difficulty society encounters in distinguishing the actual causes of cancer from mythical causes,” The BMJ commented on Twitter.
However, both the study and the journal received some backlash.
This is a “horrible article seeking to smear people with concerns about COVID vaccines,” commented Clare Craig, a British consultant pathologist who specializes in cancer diagnostics.
The study and its methodology were also harshly criticized on Twitter by Normal Fenton, professor of risk information management at the Queen Mary University of London.
The senior author of the study, Laura Costas, a medical epidemiologist with the Catalan Institute of Oncology, Barcelona, told this news organization that the naysayers on social media, many of whom focused their comments on the COVID-19 vaccine, prove the purpose of the study – that misinformation spreads widely on the internet.
“Most comments focused on spreading COVID-19 myths, which were not the direct subject of the study, and questioned the motivations of BMJ authors and the scientific community, assuming they had a common malevolent hidden agenda,” Ms. Costas said.
“They stated the need of having critical thinking, a trait in common with the scientific method, but dogmatically dismissed any information that comes from official sources,” she added.
Ms. Costas commented that “society encounters difficulty in differentiating actual from mythical causes of cancer owing to mass information. We therefore planned this study with a certain satire, which is in line with the essence of The BMJ Christmas issue.”
The BMJ has a long history of publishing a lighthearted Christmas edition full of original, satirical, and nontraditional studies. Previous years have seen studies that explored potential harms from holly and ivy, survival time of chocolates on hospital wards, and the question, “Were James Bond’s drinks shaken because of alcohol induced tremor?”
Study details
Ms. Costas and colleagues sought participants for their survey from online forums that included 4chan and Reddit, which are known for their controversial content posted by anonymous users. Data were also collected from ForoCoches and HispaChan, well-known Spanish online forums. These online sites were intentionally chosen because researchers thought “conspiracy beliefs would be more prevalent,” according to Ms. Costas.
Across the multiple forums, there were 1,494 participants. Of these, 209 participants were unvaccinated against COVID-19, 112 preferred alternatives rather than conventional medicine, and 62 reported that they believed the earth was flat or believed that humanoids take reptilian forms to manipulate human societies.
The team then sought to assess beliefs about actual and mythical (nonestablished) causes of cancer by presenting the participants with the closed risk factor questions on two validated scales – the Cancer Awareness Measure (CAM) and CAM–Mythical Causes Scale (CAM-MYCS).
Responses to both were recorded on a five-point scale; answers ranged from “strongly disagree” to “strongly agree.”
The CAM assesses cancer risk perceptions of 11 established risk factors for cancer: smoking actively or passively, consuming alcohol, low levels of physical activity, consuming red or processed meat, getting sunburnt as a child, family history of cancer, human papillomavirus infection, being overweight, age greater than or equal to 70 years, and low vegetable and fruit consumption.
The CAM-MYCS measure includes 12 questions on risk perceptions of mythical causes of cancer – nonestablished causes that are commonly believed to cause cancer but for which there is no supporting scientific evidence, the authors explain. These items include drinking from plastic bottles; eating food containing artificial sweeteners or additives and genetically modified food; using microwave ovens, aerosol containers, mobile phones, and cleaning products; living near power lines; feeling stressed; experiencing physical trauma; and being exposed to electromagnetic frequencies/non-ionizing radiation, such as wi-fi networks, radio, and television.
The most endorsed mythical causes of cancer were eating food containing additives (63.9%) or sweeteners (50.7%), feeling stressed (59.7%), and eating genetically modified foods (38.4%).
A version of this article first appeared on Medscape.com.
The study, entitled, “Everything Causes Cancer? Beliefs and Attitudes Towards Cancer Prevention Among Anti-Vaxxers, Flat Earthers, and Reptilian Conspiracists: Online Cross Sectional Survey,” was published in the Christmas 2022 issue of The British Medical Journal (BMJ).
The authors explain that they set out to evaluate “the patterns of beliefs about cancer among people who believed in conspiracies, rejected the COVID-19 vaccine, or preferred alternative medicine.”
They sought such people on social media and online chat platforms and asked them questions about real and mythical causes of cancer.
Almost half of survey participants agreed with the statement, “It seems like everything causes cancer.”
Overall, among all participants, awareness of the actual causes of cancer was greater than awareness of the mythical causes of cancer, the authors report. However, awareness of the actual causes of cancer was lower among the unvaccinated and members of conspiracy groups than among their counterparts.
The authors are concerned that their findings suggest “a direct connection between digital misinformation and consequent potential erroneous health decisions, which may represent a further preventable fraction of cancer.”
Backlash and criticism
The study “highlights the difficulty society encounters in distinguishing the actual causes of cancer from mythical causes,” The BMJ commented on Twitter.
However, both the study and the journal received some backlash.
This is a “horrible article seeking to smear people with concerns about COVID vaccines,” commented Clare Craig, a British consultant pathologist who specializes in cancer diagnostics.
The study and its methodology were also harshly criticized on Twitter by Normal Fenton, professor of risk information management at the Queen Mary University of London.
The senior author of the study, Laura Costas, a medical epidemiologist with the Catalan Institute of Oncology, Barcelona, told this news organization that the naysayers on social media, many of whom focused their comments on the COVID-19 vaccine, prove the purpose of the study – that misinformation spreads widely on the internet.
“Most comments focused on spreading COVID-19 myths, which were not the direct subject of the study, and questioned the motivations of BMJ authors and the scientific community, assuming they had a common malevolent hidden agenda,” Ms. Costas said.
“They stated the need of having critical thinking, a trait in common with the scientific method, but dogmatically dismissed any information that comes from official sources,” she added.
Ms. Costas commented that “society encounters difficulty in differentiating actual from mythical causes of cancer owing to mass information. We therefore planned this study with a certain satire, which is in line with the essence of The BMJ Christmas issue.”
The BMJ has a long history of publishing a lighthearted Christmas edition full of original, satirical, and nontraditional studies. Previous years have seen studies that explored potential harms from holly and ivy, survival time of chocolates on hospital wards, and the question, “Were James Bond’s drinks shaken because of alcohol induced tremor?”
Study details
Ms. Costas and colleagues sought participants for their survey from online forums that included 4chan and Reddit, which are known for their controversial content posted by anonymous users. Data were also collected from ForoCoches and HispaChan, well-known Spanish online forums. These online sites were intentionally chosen because researchers thought “conspiracy beliefs would be more prevalent,” according to Ms. Costas.
Across the multiple forums, there were 1,494 participants. Of these, 209 participants were unvaccinated against COVID-19, 112 preferred alternatives rather than conventional medicine, and 62 reported that they believed the earth was flat or believed that humanoids take reptilian forms to manipulate human societies.
The team then sought to assess beliefs about actual and mythical (nonestablished) causes of cancer by presenting the participants with the closed risk factor questions on two validated scales – the Cancer Awareness Measure (CAM) and CAM–Mythical Causes Scale (CAM-MYCS).
Responses to both were recorded on a five-point scale; answers ranged from “strongly disagree” to “strongly agree.”
The CAM assesses cancer risk perceptions of 11 established risk factors for cancer: smoking actively or passively, consuming alcohol, low levels of physical activity, consuming red or processed meat, getting sunburnt as a child, family history of cancer, human papillomavirus infection, being overweight, age greater than or equal to 70 years, and low vegetable and fruit consumption.
The CAM-MYCS measure includes 12 questions on risk perceptions of mythical causes of cancer – nonestablished causes that are commonly believed to cause cancer but for which there is no supporting scientific evidence, the authors explain. These items include drinking from plastic bottles; eating food containing artificial sweeteners or additives and genetically modified food; using microwave ovens, aerosol containers, mobile phones, and cleaning products; living near power lines; feeling stressed; experiencing physical trauma; and being exposed to electromagnetic frequencies/non-ionizing radiation, such as wi-fi networks, radio, and television.
The most endorsed mythical causes of cancer were eating food containing additives (63.9%) or sweeteners (50.7%), feeling stressed (59.7%), and eating genetically modified foods (38.4%).
A version of this article first appeared on Medscape.com.
The study, entitled, “Everything Causes Cancer? Beliefs and Attitudes Towards Cancer Prevention Among Anti-Vaxxers, Flat Earthers, and Reptilian Conspiracists: Online Cross Sectional Survey,” was published in the Christmas 2022 issue of The British Medical Journal (BMJ).
The authors explain that they set out to evaluate “the patterns of beliefs about cancer among people who believed in conspiracies, rejected the COVID-19 vaccine, or preferred alternative medicine.”
They sought such people on social media and online chat platforms and asked them questions about real and mythical causes of cancer.
Almost half of survey participants agreed with the statement, “It seems like everything causes cancer.”
Overall, among all participants, awareness of the actual causes of cancer was greater than awareness of the mythical causes of cancer, the authors report. However, awareness of the actual causes of cancer was lower among the unvaccinated and members of conspiracy groups than among their counterparts.
The authors are concerned that their findings suggest “a direct connection between digital misinformation and consequent potential erroneous health decisions, which may represent a further preventable fraction of cancer.”
Backlash and criticism
The study “highlights the difficulty society encounters in distinguishing the actual causes of cancer from mythical causes,” The BMJ commented on Twitter.
However, both the study and the journal received some backlash.
This is a “horrible article seeking to smear people with concerns about COVID vaccines,” commented Clare Craig, a British consultant pathologist who specializes in cancer diagnostics.
The study and its methodology were also harshly criticized on Twitter by Normal Fenton, professor of risk information management at the Queen Mary University of London.
The senior author of the study, Laura Costas, a medical epidemiologist with the Catalan Institute of Oncology, Barcelona, told this news organization that the naysayers on social media, many of whom focused their comments on the COVID-19 vaccine, prove the purpose of the study – that misinformation spreads widely on the internet.
“Most comments focused on spreading COVID-19 myths, which were not the direct subject of the study, and questioned the motivations of BMJ authors and the scientific community, assuming they had a common malevolent hidden agenda,” Ms. Costas said.
“They stated the need of having critical thinking, a trait in common with the scientific method, but dogmatically dismissed any information that comes from official sources,” she added.
Ms. Costas commented that “society encounters difficulty in differentiating actual from mythical causes of cancer owing to mass information. We therefore planned this study with a certain satire, which is in line with the essence of The BMJ Christmas issue.”
The BMJ has a long history of publishing a lighthearted Christmas edition full of original, satirical, and nontraditional studies. Previous years have seen studies that explored potential harms from holly and ivy, survival time of chocolates on hospital wards, and the question, “Were James Bond’s drinks shaken because of alcohol induced tremor?”
Study details
Ms. Costas and colleagues sought participants for their survey from online forums that included 4chan and Reddit, which are known for their controversial content posted by anonymous users. Data were also collected from ForoCoches and HispaChan, well-known Spanish online forums. These online sites were intentionally chosen because researchers thought “conspiracy beliefs would be more prevalent,” according to Ms. Costas.
Across the multiple forums, there were 1,494 participants. Of these, 209 participants were unvaccinated against COVID-19, 112 preferred alternatives rather than conventional medicine, and 62 reported that they believed the earth was flat or believed that humanoids take reptilian forms to manipulate human societies.
The team then sought to assess beliefs about actual and mythical (nonestablished) causes of cancer by presenting the participants with the closed risk factor questions on two validated scales – the Cancer Awareness Measure (CAM) and CAM–Mythical Causes Scale (CAM-MYCS).
Responses to both were recorded on a five-point scale; answers ranged from “strongly disagree” to “strongly agree.”
The CAM assesses cancer risk perceptions of 11 established risk factors for cancer: smoking actively or passively, consuming alcohol, low levels of physical activity, consuming red or processed meat, getting sunburnt as a child, family history of cancer, human papillomavirus infection, being overweight, age greater than or equal to 70 years, and low vegetable and fruit consumption.
The CAM-MYCS measure includes 12 questions on risk perceptions of mythical causes of cancer – nonestablished causes that are commonly believed to cause cancer but for which there is no supporting scientific evidence, the authors explain. These items include drinking from plastic bottles; eating food containing artificial sweeteners or additives and genetically modified food; using microwave ovens, aerosol containers, mobile phones, and cleaning products; living near power lines; feeling stressed; experiencing physical trauma; and being exposed to electromagnetic frequencies/non-ionizing radiation, such as wi-fi networks, radio, and television.
The most endorsed mythical causes of cancer were eating food containing additives (63.9%) or sweeteners (50.7%), feeling stressed (59.7%), and eating genetically modified foods (38.4%).
A version of this article first appeared on Medscape.com.
Time for a rest
“More than Jews have kept Shabbat, Shabbat has kept the Jews.” – Ahad Ha’am
You should all be well rested by now. After all, we’ve just come through the festive shutdown of the holiday season where all of your pumpkin/peppermint/marshmallow flavored coffees were sipped while walking around in your jimjams at 10 a.m. It was the time of year for you to take time off to get a proper rest and be energized to get back to work. Yet, I’m not feeling it from you.
So let’s talk about burnout – just kidding, that would only make it worse. “Burned-out’’ is a hackneyed and defective phrase to describe what many of us are feeling. We are not “destroyed, gutted by fire or by overheating.” No, we are, as one of our docs put it to me: “Just tired.” Ah, a much better Old English word! “Tired” captures it. It means to feel “in need of rest.” We are not ruined, we are just depleted. We don’t need discarding. We need some rest.
I asked some docs when they thought this feeling of exhaustion first began. We agreed that the pandemic, doubledemic, tripledemic, backlog have taken a toll. But The consumerization of medicine? All factors, but not the beginning. No, the beginning was before paper charts. Well, actually it was before paper. We have to go back to the 5th or 6th century BCE. That is when scholars believe the book of Genesis originated from the Yahwist source. In it, it is written that the 7th day be set aside as a day of rest from labor. It is not written that burnout would ensue if sabbath wasn’t observed; however, if you failed to keep it, then you might have been killed. They took rest seriously back then.
This innovation of setting aside a day each week to rest, reflect, and worship was such a good idea that it was codified as one of the 10 commandments. It spread widely. Early Christians kept the Jewish tradition of observing Shabbat from Friday sundown to Saturday until the ever practical Romans decided that Sunday would be a better day. Sunday was already the day to worship the sun god. The newly-converted Christian Emperor Constantine issued an edict on March 7th, 321 CE that all “city people and craftsmen shall rest from labor upon the venerable day of the sun.” And so Sunday it was.
Protestant Seventh-day denomination churches later shifted sabbath back to Saturday believing that Sunday must have been the Pope’s idea. The best deal seems to have been around 1273 when the Ethiopian Orthodox leader Ewostatewos decreed that both Saturday AND Sunday would be days of rest. (But when would one go to Costco?!) In Islam, there is Jumu’ah on Friday. Buddhists have Uposatha, a day of rest and observance every 7 or 8 days. Bah’ai keep Friday as a day of rest and worship. So vital are days of respite to the health of our communities that the state has made working on certain days a violation of the law, “blue laws” they are called. We’ve had blue laws on the books since the time of the Jamestown Colony in 1619 where the first Virginia Assembly required taking Sunday off for worship. Most of these laws have been repealed, although a few states, such as Rhode Island, still have blue laws prohibiting retail and grocery stores from opening on Thanksgiving or Christmas. So there – enjoy your two days off this year!
Ironically, this column, like most of mine, comes to you after my having written it on a Saturday and Sunday. I also just logged on to my EMR and checked results, renewed a few prescriptions, and answered a couple messages. If I didn’t, my Monday’s work would be crushingly heavy.
Maybe I need to be more efficient and finish my work during the week. Or maybe I need to realize that work has not let up since about 600 BCE and taking one day off each week to rest is an obligation to myself, my family and my community.
I wonder if I can choose Mondays.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
“More than Jews have kept Shabbat, Shabbat has kept the Jews.” – Ahad Ha’am
You should all be well rested by now. After all, we’ve just come through the festive shutdown of the holiday season where all of your pumpkin/peppermint/marshmallow flavored coffees were sipped while walking around in your jimjams at 10 a.m. It was the time of year for you to take time off to get a proper rest and be energized to get back to work. Yet, I’m not feeling it from you.
So let’s talk about burnout – just kidding, that would only make it worse. “Burned-out’’ is a hackneyed and defective phrase to describe what many of us are feeling. We are not “destroyed, gutted by fire or by overheating.” No, we are, as one of our docs put it to me: “Just tired.” Ah, a much better Old English word! “Tired” captures it. It means to feel “in need of rest.” We are not ruined, we are just depleted. We don’t need discarding. We need some rest.
I asked some docs when they thought this feeling of exhaustion first began. We agreed that the pandemic, doubledemic, tripledemic, backlog have taken a toll. But The consumerization of medicine? All factors, but not the beginning. No, the beginning was before paper charts. Well, actually it was before paper. We have to go back to the 5th or 6th century BCE. That is when scholars believe the book of Genesis originated from the Yahwist source. In it, it is written that the 7th day be set aside as a day of rest from labor. It is not written that burnout would ensue if sabbath wasn’t observed; however, if you failed to keep it, then you might have been killed. They took rest seriously back then.
This innovation of setting aside a day each week to rest, reflect, and worship was such a good idea that it was codified as one of the 10 commandments. It spread widely. Early Christians kept the Jewish tradition of observing Shabbat from Friday sundown to Saturday until the ever practical Romans decided that Sunday would be a better day. Sunday was already the day to worship the sun god. The newly-converted Christian Emperor Constantine issued an edict on March 7th, 321 CE that all “city people and craftsmen shall rest from labor upon the venerable day of the sun.” And so Sunday it was.
Protestant Seventh-day denomination churches later shifted sabbath back to Saturday believing that Sunday must have been the Pope’s idea. The best deal seems to have been around 1273 when the Ethiopian Orthodox leader Ewostatewos decreed that both Saturday AND Sunday would be days of rest. (But when would one go to Costco?!) In Islam, there is Jumu’ah on Friday. Buddhists have Uposatha, a day of rest and observance every 7 or 8 days. Bah’ai keep Friday as a day of rest and worship. So vital are days of respite to the health of our communities that the state has made working on certain days a violation of the law, “blue laws” they are called. We’ve had blue laws on the books since the time of the Jamestown Colony in 1619 where the first Virginia Assembly required taking Sunday off for worship. Most of these laws have been repealed, although a few states, such as Rhode Island, still have blue laws prohibiting retail and grocery stores from opening on Thanksgiving or Christmas. So there – enjoy your two days off this year!
Ironically, this column, like most of mine, comes to you after my having written it on a Saturday and Sunday. I also just logged on to my EMR and checked results, renewed a few prescriptions, and answered a couple messages. If I didn’t, my Monday’s work would be crushingly heavy.
Maybe I need to be more efficient and finish my work during the week. Or maybe I need to realize that work has not let up since about 600 BCE and taking one day off each week to rest is an obligation to myself, my family and my community.
I wonder if I can choose Mondays.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
“More than Jews have kept Shabbat, Shabbat has kept the Jews.” – Ahad Ha’am
You should all be well rested by now. After all, we’ve just come through the festive shutdown of the holiday season where all of your pumpkin/peppermint/marshmallow flavored coffees were sipped while walking around in your jimjams at 10 a.m. It was the time of year for you to take time off to get a proper rest and be energized to get back to work. Yet, I’m not feeling it from you.
So let’s talk about burnout – just kidding, that would only make it worse. “Burned-out’’ is a hackneyed and defective phrase to describe what many of us are feeling. We are not “destroyed, gutted by fire or by overheating.” No, we are, as one of our docs put it to me: “Just tired.” Ah, a much better Old English word! “Tired” captures it. It means to feel “in need of rest.” We are not ruined, we are just depleted. We don’t need discarding. We need some rest.
I asked some docs when they thought this feeling of exhaustion first began. We agreed that the pandemic, doubledemic, tripledemic, backlog have taken a toll. But The consumerization of medicine? All factors, but not the beginning. No, the beginning was before paper charts. Well, actually it was before paper. We have to go back to the 5th or 6th century BCE. That is when scholars believe the book of Genesis originated from the Yahwist source. In it, it is written that the 7th day be set aside as a day of rest from labor. It is not written that burnout would ensue if sabbath wasn’t observed; however, if you failed to keep it, then you might have been killed. They took rest seriously back then.
This innovation of setting aside a day each week to rest, reflect, and worship was such a good idea that it was codified as one of the 10 commandments. It spread widely. Early Christians kept the Jewish tradition of observing Shabbat from Friday sundown to Saturday until the ever practical Romans decided that Sunday would be a better day. Sunday was already the day to worship the sun god. The newly-converted Christian Emperor Constantine issued an edict on March 7th, 321 CE that all “city people and craftsmen shall rest from labor upon the venerable day of the sun.” And so Sunday it was.
Protestant Seventh-day denomination churches later shifted sabbath back to Saturday believing that Sunday must have been the Pope’s idea. The best deal seems to have been around 1273 when the Ethiopian Orthodox leader Ewostatewos decreed that both Saturday AND Sunday would be days of rest. (But when would one go to Costco?!) In Islam, there is Jumu’ah on Friday. Buddhists have Uposatha, a day of rest and observance every 7 or 8 days. Bah’ai keep Friday as a day of rest and worship. So vital are days of respite to the health of our communities that the state has made working on certain days a violation of the law, “blue laws” they are called. We’ve had blue laws on the books since the time of the Jamestown Colony in 1619 where the first Virginia Assembly required taking Sunday off for worship. Most of these laws have been repealed, although a few states, such as Rhode Island, still have blue laws prohibiting retail and grocery stores from opening on Thanksgiving or Christmas. So there – enjoy your two days off this year!
Ironically, this column, like most of mine, comes to you after my having written it on a Saturday and Sunday. I also just logged on to my EMR and checked results, renewed a few prescriptions, and answered a couple messages. If I didn’t, my Monday’s work would be crushingly heavy.
Maybe I need to be more efficient and finish my work during the week. Or maybe I need to realize that work has not let up since about 600 BCE and taking one day off each week to rest is an obligation to myself, my family and my community.
I wonder if I can choose Mondays.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
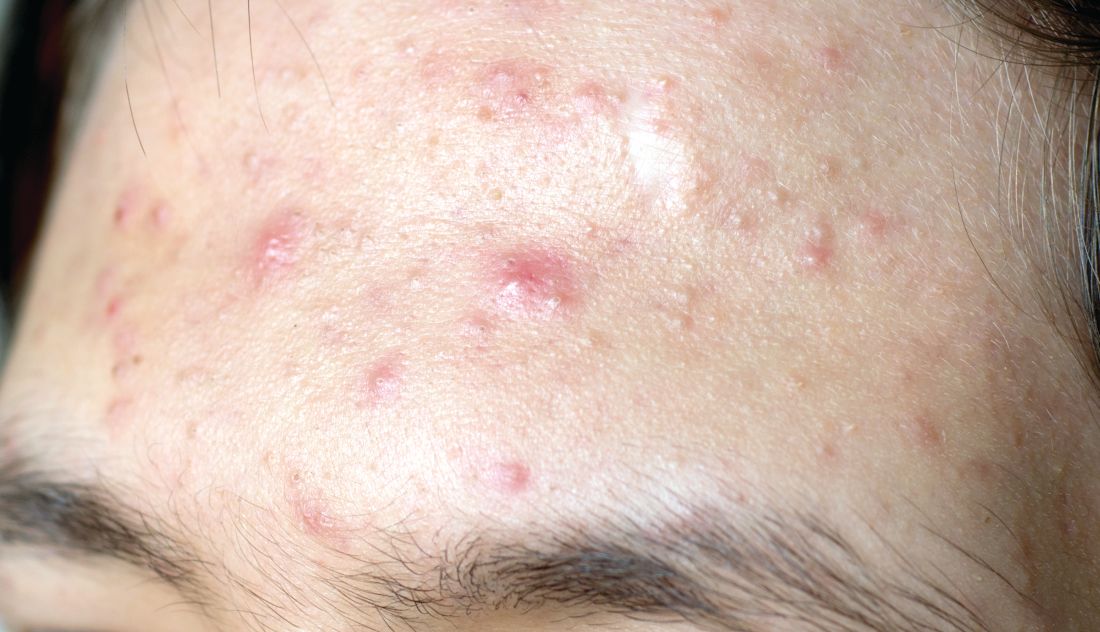
Topical treatment options for acne continue to expand
SAN DIEGO – , according to Lawrence F. Eichenfield, MD.
The product, known as IDP-126 and being developed by Ortho Dermatologics, is a fixed dose triple combination of clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% being evaluated in patients nine years of age and older. According to a 2021 press release from the company, results from a second 12-week pivotal phase 3 trial showed a treatment success of 50.5% and 20.5% for IDP-126 and its vehicle, respectively, along with significant changes from baseline in inflammatory lesion count and non-inflammatory lesion count.
More recently, researchers led by Linda Stein Gold, MD, conducted a 12-week multicenter, randomized, double-blind study of IDP-126 in 741 children, adolescents, and adults with moderate to severe acne. They reported 52.5% of patients treated with IDP-126 gel achieved treatment success by week 12, with over 70% reduction in inflammatory and noninflammatory lesions.
“This will be interesting to follow as it moves along,” Dr. Eichenfield, chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, said at the annual Masters of Aesthetics Symposium in a presentation on the newest topical acne treatments.
“If approved, we probably will be able to decrease our need for systemic therapies in some individuals,” he said. “It’s something that may become important in practices that mix and match between medical and procedural or surgical approaches to acne.”
Dr. Eichenfield highlighted other products for the topical treatment of acne:
- Trifarotene cream 0.005% (Aklief). In 2019, Food and Drug Administration approval made trifarotene cream the first new retinoid indicated for acne in several decades. It is indicated for the topical treatment of acne vulgaris in patients age 9 and older and has been studied in acne of the face, chest, and back.
- Tazarotene lotion 0.045% (Arazlo). The 0.1% formulation of tazarotene is commonly used for acne, but it can cause skin irritation, dryness, and erythema. The new 0.045% formulation was developed in a three-dimensional mesh matrix, with ingredients from an oil-in-water emulsion. “Many of the new acne products come with a background of vehicle delivery systems that minimize the concentration gradient, so it decreases irritation,” said Dr. Eichenfield, one of the authors of a 2021 review article on the management of acne vulgaris in JAMA. “This has very good efficacy without the traditional irritation of other tazarotene products,” Dr. Eichenfield said.
- Minocycline 4% topical foam (Amzeeq). The 2019 U.S. approval marked the first and so far only topical minocycline prescription treatment for acne. “Its hydrophobic composition allows for stable and efficient delivery of inherently unstable pharmaceutical ingredients,” he said. “It’s generally well tolerated.”
- Clascoterone cream 1% (Winlevi). This first-in-class topical androgen receptor inhibitor is approved for the treatment of acne in patients 12 years and older. It competes with dihydrotestosterone and selectively targets androgen receptors in sebocytes and hair papilla cells. “It is safe for use in men, has been studied on the face and trunk, and has been shown to inhibit sebum production, reduce secretion of inflammatory cytokines, and inhibit inflammatory pathways,” Dr. Eichenfield said.
- Micro-encapsulated benzoyl peroxide 3% and tretinoin 0.1% cream (Twyneo). This is a once-daily fixed-dose combination of tretinoin and benzoyl peroxide indicated for the treatment of acne vulgaris in patients age 9 and older. According to a press release from Sol-Gel, the manufacturer, silica (silicon dioxide) core shell structures separate micro-encapsulate tretinoin crystals and benzoyl peroxide crystals, enabling inclusion of the two active ingredients in the cream.
Dr. Eichenfield disclosed that he has been an investigator and/or consultant for Almirall, Cassiopea, Dermata, Galderma, and Ortho Dermatologics.
SAN DIEGO – , according to Lawrence F. Eichenfield, MD.
The product, known as IDP-126 and being developed by Ortho Dermatologics, is a fixed dose triple combination of clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% being evaluated in patients nine years of age and older. According to a 2021 press release from the company, results from a second 12-week pivotal phase 3 trial showed a treatment success of 50.5% and 20.5% for IDP-126 and its vehicle, respectively, along with significant changes from baseline in inflammatory lesion count and non-inflammatory lesion count.
More recently, researchers led by Linda Stein Gold, MD, conducted a 12-week multicenter, randomized, double-blind study of IDP-126 in 741 children, adolescents, and adults with moderate to severe acne. They reported 52.5% of patients treated with IDP-126 gel achieved treatment success by week 12, with over 70% reduction in inflammatory and noninflammatory lesions.
“This will be interesting to follow as it moves along,” Dr. Eichenfield, chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, said at the annual Masters of Aesthetics Symposium in a presentation on the newest topical acne treatments.
“If approved, we probably will be able to decrease our need for systemic therapies in some individuals,” he said. “It’s something that may become important in practices that mix and match between medical and procedural or surgical approaches to acne.”
Dr. Eichenfield highlighted other products for the topical treatment of acne:
- Trifarotene cream 0.005% (Aklief). In 2019, Food and Drug Administration approval made trifarotene cream the first new retinoid indicated for acne in several decades. It is indicated for the topical treatment of acne vulgaris in patients age 9 and older and has been studied in acne of the face, chest, and back.
- Tazarotene lotion 0.045% (Arazlo). The 0.1% formulation of tazarotene is commonly used for acne, but it can cause skin irritation, dryness, and erythema. The new 0.045% formulation was developed in a three-dimensional mesh matrix, with ingredients from an oil-in-water emulsion. “Many of the new acne products come with a background of vehicle delivery systems that minimize the concentration gradient, so it decreases irritation,” said Dr. Eichenfield, one of the authors of a 2021 review article on the management of acne vulgaris in JAMA. “This has very good efficacy without the traditional irritation of other tazarotene products,” Dr. Eichenfield said.
- Minocycline 4% topical foam (Amzeeq). The 2019 U.S. approval marked the first and so far only topical minocycline prescription treatment for acne. “Its hydrophobic composition allows for stable and efficient delivery of inherently unstable pharmaceutical ingredients,” he said. “It’s generally well tolerated.”
- Clascoterone cream 1% (Winlevi). This first-in-class topical androgen receptor inhibitor is approved for the treatment of acne in patients 12 years and older. It competes with dihydrotestosterone and selectively targets androgen receptors in sebocytes and hair papilla cells. “It is safe for use in men, has been studied on the face and trunk, and has been shown to inhibit sebum production, reduce secretion of inflammatory cytokines, and inhibit inflammatory pathways,” Dr. Eichenfield said.
- Micro-encapsulated benzoyl peroxide 3% and tretinoin 0.1% cream (Twyneo). This is a once-daily fixed-dose combination of tretinoin and benzoyl peroxide indicated for the treatment of acne vulgaris in patients age 9 and older. According to a press release from Sol-Gel, the manufacturer, silica (silicon dioxide) core shell structures separate micro-encapsulate tretinoin crystals and benzoyl peroxide crystals, enabling inclusion of the two active ingredients in the cream.
Dr. Eichenfield disclosed that he has been an investigator and/or consultant for Almirall, Cassiopea, Dermata, Galderma, and Ortho Dermatologics.
SAN DIEGO – , according to Lawrence F. Eichenfield, MD.
The product, known as IDP-126 and being developed by Ortho Dermatologics, is a fixed dose triple combination of clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% being evaluated in patients nine years of age and older. According to a 2021 press release from the company, results from a second 12-week pivotal phase 3 trial showed a treatment success of 50.5% and 20.5% for IDP-126 and its vehicle, respectively, along with significant changes from baseline in inflammatory lesion count and non-inflammatory lesion count.
More recently, researchers led by Linda Stein Gold, MD, conducted a 12-week multicenter, randomized, double-blind study of IDP-126 in 741 children, adolescents, and adults with moderate to severe acne. They reported 52.5% of patients treated with IDP-126 gel achieved treatment success by week 12, with over 70% reduction in inflammatory and noninflammatory lesions.
“This will be interesting to follow as it moves along,” Dr. Eichenfield, chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, said at the annual Masters of Aesthetics Symposium in a presentation on the newest topical acne treatments.
“If approved, we probably will be able to decrease our need for systemic therapies in some individuals,” he said. “It’s something that may become important in practices that mix and match between medical and procedural or surgical approaches to acne.”
Dr. Eichenfield highlighted other products for the topical treatment of acne:
- Trifarotene cream 0.005% (Aklief). In 2019, Food and Drug Administration approval made trifarotene cream the first new retinoid indicated for acne in several decades. It is indicated for the topical treatment of acne vulgaris in patients age 9 and older and has been studied in acne of the face, chest, and back.
- Tazarotene lotion 0.045% (Arazlo). The 0.1% formulation of tazarotene is commonly used for acne, but it can cause skin irritation, dryness, and erythema. The new 0.045% formulation was developed in a three-dimensional mesh matrix, with ingredients from an oil-in-water emulsion. “Many of the new acne products come with a background of vehicle delivery systems that minimize the concentration gradient, so it decreases irritation,” said Dr. Eichenfield, one of the authors of a 2021 review article on the management of acne vulgaris in JAMA. “This has very good efficacy without the traditional irritation of other tazarotene products,” Dr. Eichenfield said.
- Minocycline 4% topical foam (Amzeeq). The 2019 U.S. approval marked the first and so far only topical minocycline prescription treatment for acne. “Its hydrophobic composition allows for stable and efficient delivery of inherently unstable pharmaceutical ingredients,” he said. “It’s generally well tolerated.”
- Clascoterone cream 1% (Winlevi). This first-in-class topical androgen receptor inhibitor is approved for the treatment of acne in patients 12 years and older. It competes with dihydrotestosterone and selectively targets androgen receptors in sebocytes and hair papilla cells. “It is safe for use in men, has been studied on the face and trunk, and has been shown to inhibit sebum production, reduce secretion of inflammatory cytokines, and inhibit inflammatory pathways,” Dr. Eichenfield said.
- Micro-encapsulated benzoyl peroxide 3% and tretinoin 0.1% cream (Twyneo). This is a once-daily fixed-dose combination of tretinoin and benzoyl peroxide indicated for the treatment of acne vulgaris in patients age 9 and older. According to a press release from Sol-Gel, the manufacturer, silica (silicon dioxide) core shell structures separate micro-encapsulate tretinoin crystals and benzoyl peroxide crystals, enabling inclusion of the two active ingredients in the cream.
Dr. Eichenfield disclosed that he has been an investigator and/or consultant for Almirall, Cassiopea, Dermata, Galderma, and Ortho Dermatologics.
AT MOAS 2022
New treatments aim to tame vitiligo
LAS VEGAS – in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
Vitiligo, an autoimmune condition that results in patches of skin depigmentation, occurs in 0.5% to 2% of the population. The average age of onset is 20 years, with 25% of cases occurring before age 10, and 70%-80% of cases by age 30 years, which means a long-term effect on quality of life, especially for younger patients, said Dr. Rosmarin, vice chair of education and research and director of the clinical trials unit at Tufts University, Boston.
Studies have shown that 95% of 15- to 17-year-olds with vitiligo are bothered by it, as are approximately 50% of children aged 6-14 years, he said. Although patients with more extensive lesions on the face, arms, legs, and hands report worse quality of life, they report that uncontrolled progression of vitiligo is more concerning than the presence of lesions in exposed areas, he noted.
The current strategy for getting vitiligo under control is a two-step process, said Dr. Rosmarin. First, improve the skin environment by suppressing the overactive immune system, then encourage repigmentation and “nudge the melanocytes to return,” he said.
Topical ruxolitinib, a Janus kinase (JAK) inhibitor, is the latest tool for dermatologists to help give the melanocytes that nudge. In July 2022, the Food and Drug Administration approved ruxolitinib cream for treating nonsegmental vitiligo in patients 12 years of age and older – the first treatment approved to repigment patients with vitiligo.
Vitiligo is driven in part by interferon (IFN)-gamma signaling through JAK 1 and 2, and ruxolitinib acts as an inhibitor, Dr. Rosmarin said.
In the TRuE-V1 and TRuE-V2 studies presented at the 2022 European Academy of Dermatology and Venereology meeting in Milan, adolescents and adults with vitiligo who were randomized to 1.5% ruxolitinib cream twice daily showed significant improvement over those randomized to the vehicle by 24 weeks, at which time all patients could continue with ruxolitinib through 52 weeks, he said.
Dr. Rosmarin presented 52-week data from the TRuE-V1 and TRuE-V2 studies at the 2022 American Academy of Dermatology meeting in Boston. He was the lead author of the studies that were subsequently published in the New England Journal of Medicine.
In the two studies, 52.6% and 48% of the patients in the ruxolitinib groups achieved the primary outcome of at least 75% improvement on the Facial Vitiligo Area Scoring Index (F-VASI75) by 52 weeks, compared with 26.8% and 29.6% of patients on the vehicle, respectively.
In addition, at 52 weeks, 53.2% and 49.2% of patients treated with ruxolitinib in the two studies achieved 50% improvement on the Total Vitiligo Area Scoring Index (T-VASI50), a clinician assessment of affected body surface area and level of depigmentation, compared with 31.7% and 22.2% of those on vehicle, respectively.
Patient satisfaction was high with ruxolitinib, Dr. Rosmarin said. In the TRuE-V1 and TRuE-V2 studies, 39.9% and 32.8% of patients, respectively, achieved a successful treatment response based on the patient-reported Vitiligo Noticeability Scale (VNS) by week 52, versus 19.5% and 13.6% of those on vehicle.
Ruxolitinib cream was well tolerated, with “no clinically significant application site reactions or serious treatment-related adverse events,” he noted. The most common treatment-related adverse events across the TRuE-V1 and TRuE-V2 studies were acne at the application site (affecting about 6% of patients) and pruritus at the application site about (affecting 5%), said Dr. Rosmarin.
JAK inhibitors, including ruxolitinib, baricitinib, and tofacitinib, have shown effectiveness for vitiligo, which supports the potential role of the IFN-gamma-chemokine signaling axis in the pathogenesis of the disease, said Dr. Rosmarin. However, more studies are required to determine the ideal dosage of JAK inhibitors for the treatment of vitiligo, and to identify other inflammatory pathways that may be implicated in the pathogenesis of this condition.
Ruxolitinib’s success has been consistent across subgroups of age, gender, race, geographic region, and Fitzpatrick skin phototype. Notably, ruxolitinib was effective among the adolescent population, with approximately 60% achieving T-VASI50 and success based on VNS in TRuE-V1 and TRuE-V2.
An oral version of ruxolitinib is in clinical trials, which “makes a lot of sense,” Dr. Rosmarin said. “Patients don’t always have localized disease,” and such patients may benefit from an oral therapy. Topicals may have the advantage in terms of safety, but questions of maintenance remain, he said. Oral treatments may be useful for patients with large body surface areas affected, and those with unstable or progressive disease, he added.
Areas for additional research include combination therapy with ruxolitinib and phototherapy, and an anti-IL 15 therapy in the pipeline has the potential to drive vitiligo into remission, Dr. Rosmarin said. In a study known as REVEAL that is still recruiting patients, researchers will test the efficacy of an IL-15 inhibitor known as AMG 714 to induce facial repigmentation in adults with vitiligo.
Dr. Rosmarin disclosed ties with AbbVie, Abcuro, AltruBio, Amgen, Arena Pharmaceuticals, Boehringer Ingelheim, Bristol Myers Squibb Company, Celgene, Concert Pharmaceuticals, CSL Behring, Dermavant, Dermira, Eli Lilly, Galderma, Incyte, Janssen, Kyowa Kirin, Merck, Novartis, Pfizer, Regeneron, Revolo, Sanofi, Sun, UCB, and Viela Bio.
MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
Vitiligo, an autoimmune condition that results in patches of skin depigmentation, occurs in 0.5% to 2% of the population. The average age of onset is 20 years, with 25% of cases occurring before age 10, and 70%-80% of cases by age 30 years, which means a long-term effect on quality of life, especially for younger patients, said Dr. Rosmarin, vice chair of education and research and director of the clinical trials unit at Tufts University, Boston.
Studies have shown that 95% of 15- to 17-year-olds with vitiligo are bothered by it, as are approximately 50% of children aged 6-14 years, he said. Although patients with more extensive lesions on the face, arms, legs, and hands report worse quality of life, they report that uncontrolled progression of vitiligo is more concerning than the presence of lesions in exposed areas, he noted.
The current strategy for getting vitiligo under control is a two-step process, said Dr. Rosmarin. First, improve the skin environment by suppressing the overactive immune system, then encourage repigmentation and “nudge the melanocytes to return,” he said.
Topical ruxolitinib, a Janus kinase (JAK) inhibitor, is the latest tool for dermatologists to help give the melanocytes that nudge. In July 2022, the Food and Drug Administration approved ruxolitinib cream for treating nonsegmental vitiligo in patients 12 years of age and older – the first treatment approved to repigment patients with vitiligo.
Vitiligo is driven in part by interferon (IFN)-gamma signaling through JAK 1 and 2, and ruxolitinib acts as an inhibitor, Dr. Rosmarin said.
In the TRuE-V1 and TRuE-V2 studies presented at the 2022 European Academy of Dermatology and Venereology meeting in Milan, adolescents and adults with vitiligo who were randomized to 1.5% ruxolitinib cream twice daily showed significant improvement over those randomized to the vehicle by 24 weeks, at which time all patients could continue with ruxolitinib through 52 weeks, he said.
Dr. Rosmarin presented 52-week data from the TRuE-V1 and TRuE-V2 studies at the 2022 American Academy of Dermatology meeting in Boston. He was the lead author of the studies that were subsequently published in the New England Journal of Medicine.
In the two studies, 52.6% and 48% of the patients in the ruxolitinib groups achieved the primary outcome of at least 75% improvement on the Facial Vitiligo Area Scoring Index (F-VASI75) by 52 weeks, compared with 26.8% and 29.6% of patients on the vehicle, respectively.
In addition, at 52 weeks, 53.2% and 49.2% of patients treated with ruxolitinib in the two studies achieved 50% improvement on the Total Vitiligo Area Scoring Index (T-VASI50), a clinician assessment of affected body surface area and level of depigmentation, compared with 31.7% and 22.2% of those on vehicle, respectively.
Patient satisfaction was high with ruxolitinib, Dr. Rosmarin said. In the TRuE-V1 and TRuE-V2 studies, 39.9% and 32.8% of patients, respectively, achieved a successful treatment response based on the patient-reported Vitiligo Noticeability Scale (VNS) by week 52, versus 19.5% and 13.6% of those on vehicle.
Ruxolitinib cream was well tolerated, with “no clinically significant application site reactions or serious treatment-related adverse events,” he noted. The most common treatment-related adverse events across the TRuE-V1 and TRuE-V2 studies were acne at the application site (affecting about 6% of patients) and pruritus at the application site about (affecting 5%), said Dr. Rosmarin.
JAK inhibitors, including ruxolitinib, baricitinib, and tofacitinib, have shown effectiveness for vitiligo, which supports the potential role of the IFN-gamma-chemokine signaling axis in the pathogenesis of the disease, said Dr. Rosmarin. However, more studies are required to determine the ideal dosage of JAK inhibitors for the treatment of vitiligo, and to identify other inflammatory pathways that may be implicated in the pathogenesis of this condition.
Ruxolitinib’s success has been consistent across subgroups of age, gender, race, geographic region, and Fitzpatrick skin phototype. Notably, ruxolitinib was effective among the adolescent population, with approximately 60% achieving T-VASI50 and success based on VNS in TRuE-V1 and TRuE-V2.
An oral version of ruxolitinib is in clinical trials, which “makes a lot of sense,” Dr. Rosmarin said. “Patients don’t always have localized disease,” and such patients may benefit from an oral therapy. Topicals may have the advantage in terms of safety, but questions of maintenance remain, he said. Oral treatments may be useful for patients with large body surface areas affected, and those with unstable or progressive disease, he added.
Areas for additional research include combination therapy with ruxolitinib and phototherapy, and an anti-IL 15 therapy in the pipeline has the potential to drive vitiligo into remission, Dr. Rosmarin said. In a study known as REVEAL that is still recruiting patients, researchers will test the efficacy of an IL-15 inhibitor known as AMG 714 to induce facial repigmentation in adults with vitiligo.
Dr. Rosmarin disclosed ties with AbbVie, Abcuro, AltruBio, Amgen, Arena Pharmaceuticals, Boehringer Ingelheim, Bristol Myers Squibb Company, Celgene, Concert Pharmaceuticals, CSL Behring, Dermavant, Dermira, Eli Lilly, Galderma, Incyte, Janssen, Kyowa Kirin, Merck, Novartis, Pfizer, Regeneron, Revolo, Sanofi, Sun, UCB, and Viela Bio.
MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
Vitiligo, an autoimmune condition that results in patches of skin depigmentation, occurs in 0.5% to 2% of the population. The average age of onset is 20 years, with 25% of cases occurring before age 10, and 70%-80% of cases by age 30 years, which means a long-term effect on quality of life, especially for younger patients, said Dr. Rosmarin, vice chair of education and research and director of the clinical trials unit at Tufts University, Boston.
Studies have shown that 95% of 15- to 17-year-olds with vitiligo are bothered by it, as are approximately 50% of children aged 6-14 years, he said. Although patients with more extensive lesions on the face, arms, legs, and hands report worse quality of life, they report that uncontrolled progression of vitiligo is more concerning than the presence of lesions in exposed areas, he noted.
The current strategy for getting vitiligo under control is a two-step process, said Dr. Rosmarin. First, improve the skin environment by suppressing the overactive immune system, then encourage repigmentation and “nudge the melanocytes to return,” he said.
Topical ruxolitinib, a Janus kinase (JAK) inhibitor, is the latest tool for dermatologists to help give the melanocytes that nudge. In July 2022, the Food and Drug Administration approved ruxolitinib cream for treating nonsegmental vitiligo in patients 12 years of age and older – the first treatment approved to repigment patients with vitiligo.
Vitiligo is driven in part by interferon (IFN)-gamma signaling through JAK 1 and 2, and ruxolitinib acts as an inhibitor, Dr. Rosmarin said.
In the TRuE-V1 and TRuE-V2 studies presented at the 2022 European Academy of Dermatology and Venereology meeting in Milan, adolescents and adults with vitiligo who were randomized to 1.5% ruxolitinib cream twice daily showed significant improvement over those randomized to the vehicle by 24 weeks, at which time all patients could continue with ruxolitinib through 52 weeks, he said.
Dr. Rosmarin presented 52-week data from the TRuE-V1 and TRuE-V2 studies at the 2022 American Academy of Dermatology meeting in Boston. He was the lead author of the studies that were subsequently published in the New England Journal of Medicine.
In the two studies, 52.6% and 48% of the patients in the ruxolitinib groups achieved the primary outcome of at least 75% improvement on the Facial Vitiligo Area Scoring Index (F-VASI75) by 52 weeks, compared with 26.8% and 29.6% of patients on the vehicle, respectively.
In addition, at 52 weeks, 53.2% and 49.2% of patients treated with ruxolitinib in the two studies achieved 50% improvement on the Total Vitiligo Area Scoring Index (T-VASI50), a clinician assessment of affected body surface area and level of depigmentation, compared with 31.7% and 22.2% of those on vehicle, respectively.
Patient satisfaction was high with ruxolitinib, Dr. Rosmarin said. In the TRuE-V1 and TRuE-V2 studies, 39.9% and 32.8% of patients, respectively, achieved a successful treatment response based on the patient-reported Vitiligo Noticeability Scale (VNS) by week 52, versus 19.5% and 13.6% of those on vehicle.
Ruxolitinib cream was well tolerated, with “no clinically significant application site reactions or serious treatment-related adverse events,” he noted. The most common treatment-related adverse events across the TRuE-V1 and TRuE-V2 studies were acne at the application site (affecting about 6% of patients) and pruritus at the application site about (affecting 5%), said Dr. Rosmarin.
JAK inhibitors, including ruxolitinib, baricitinib, and tofacitinib, have shown effectiveness for vitiligo, which supports the potential role of the IFN-gamma-chemokine signaling axis in the pathogenesis of the disease, said Dr. Rosmarin. However, more studies are required to determine the ideal dosage of JAK inhibitors for the treatment of vitiligo, and to identify other inflammatory pathways that may be implicated in the pathogenesis of this condition.
Ruxolitinib’s success has been consistent across subgroups of age, gender, race, geographic region, and Fitzpatrick skin phototype. Notably, ruxolitinib was effective among the adolescent population, with approximately 60% achieving T-VASI50 and success based on VNS in TRuE-V1 and TRuE-V2.
An oral version of ruxolitinib is in clinical trials, which “makes a lot of sense,” Dr. Rosmarin said. “Patients don’t always have localized disease,” and such patients may benefit from an oral therapy. Topicals may have the advantage in terms of safety, but questions of maintenance remain, he said. Oral treatments may be useful for patients with large body surface areas affected, and those with unstable or progressive disease, he added.
Areas for additional research include combination therapy with ruxolitinib and phototherapy, and an anti-IL 15 therapy in the pipeline has the potential to drive vitiligo into remission, Dr. Rosmarin said. In a study known as REVEAL that is still recruiting patients, researchers will test the efficacy of an IL-15 inhibitor known as AMG 714 to induce facial repigmentation in adults with vitiligo.
Dr. Rosmarin disclosed ties with AbbVie, Abcuro, AltruBio, Amgen, Arena Pharmaceuticals, Boehringer Ingelheim, Bristol Myers Squibb Company, Celgene, Concert Pharmaceuticals, CSL Behring, Dermavant, Dermira, Eli Lilly, Galderma, Incyte, Janssen, Kyowa Kirin, Merck, Novartis, Pfizer, Regeneron, Revolo, Sanofi, Sun, UCB, and Viela Bio.
MedscapeLive and this news organization are owned by the same parent company.
AT INNOVATIONS IN DERMATOLOGY
Oral minoxidil improves anticancer treatment–induced alopecia in women with breast cancer
Topical minoxidil is widely used to treat hair loss, but new findings suggest that
In a retrospective cohort study of women with breast cancer and anticancer therapy–induced alopecia, researchers found that combining low-dose oral minoxidil (LDOM) and topical minoxidil achieved better results than topical minoxidil alone and that the treatment was well tolerated. A total of 5 of the 37 patients (13.5%) in the combination therapy group achieved a complete response, defined as an improvement of alopecia severity from grade 2 to grade 1, compared with none of the 19 patients in the topical therapy–only group.
In contrast, none of the patients in the combination group experienced worsening of alopecia, compared with two (10.5%) in the topical monotherapy group.
The study was published online in the Journal of the American Academy of Dermatology. Topical minoxidil is approved by the Food and Drug Administration to treat androgenetic alopecia. Oral minoxidil is not approved for treating hair loss but has been receiving increased attention as an adjunctive therapy for hair loss, particularly for women. Oral minoxidil is approved for treating hypertension but at much higher doses.
An increasing number of studies have been conducted on the use of oral minoxidil for the treatment of female pattern hair loss, dating back to a pilot study in 2017, with promising results. The findings suggest that LDOM might be more effective than topical therapy, well tolerated, and more convenient for individuals to take.
Hypothesis generating
In a comment, Kai Johnson, MD, a medical oncologist who specializes in treating patients with breast cancer at the Ohio State University Comprehensive Cancer Center – Arthur G. James Cancer Hospital and Richard J. Solove Research Institute, Columbus, noted that the study, like most small-scale retrospective studies, is hypothesis generating. However, “I’d be hesitant to broadly recommend this practice of dual therapy – oral and topical minoxidil together – until we see a placebo-controlled prospective study performed demonstrating clinically meaningful benefits for patients.”
Another factor is the study endpoints. “While there was a statistically significant benefit documented with dual therapy in this study, it’s important to have study endpoints that are more patient oriented,” Dr. Johnson said. The most important endpoint for patients would be improvements “in the actual alopecia grade, which did occur in 5 of the 37 of dual-therapy patients, versus 0 topical minoxidil patients.”
George Cotsarelis, MD, chair of the department of dermatology and professor of dermatology at the University of Pennsylvania, Philadelphia, also weighed in. He questioned whether adding the topical therapy to oral minoxidil actually improved the results. “What was missing was a study arm that used the oral alone,” he said in an interview. “So we don’t know how effective the oral therapy would be by itself and if combining it with the topical is really adding anything.”
Oral minoxidil as a treatment for hair loss is gaining traction, and it’s clear that it is effective. However, the risk of side effects is higher, he said. “The risk isn’t that high with the low dose, but it can grow hair on places other than the scalp, and that can be disconcerting.” In this study, two women who took the oral drug reported edema, and one reported headache and dizziness. Hypertrichosis was reported by five patients who received the combination.
Study details
In the study, Jeewoo Kang, MD, and colleagues from the Seoul National University evaluated the efficacy of LDOM in 100 patients with breast cancer who had been diagnosed with persistent chemotherapy-induced alopecia (pCIA) and endocrine therapy–induced alopecia (EIA) at a dermatology clinic.
They conducted an analysis of medical records, standardized clinical photographs, and trichoscopic images to evaluate the alopecia pattern, severity, treatment response, and posttreatment changes in vertex hair density and thickness.
Compared with those with EIA alone, patients with pCIA were significantly more likely to have diffuse alopecia (P < .001), and they were more likely to have more severe alopecia, although this difference was not significant (P = .058). Outcomes were evaluated for 56 patients who were treated with minoxidil (19 with topical minoxidil alone and 37 with both LDOM and topical minoxidil) and for whom clinical and trichoscopic photos were available at baseline and at the last follow-up (all patients were scheduled for follow-up at 3-month intervals).
The results showed that those treated with 1.25-5.0 mg/d of oral minoxidil and 5% topical minoxidil solution once a day had better responses (P = .002) and a higher percentage increase in hair density from baseline (P = .003), compared with those who received topical minoxidil monotherapy.
However, changes in hair thickness after treatment were not significantly different between the two groups (P = .540).
In addition to the five (13.5%) cases of hypertrichosis, two cases of edema (5.4%), and one case of headache/dizziness (2.7%) among those who received the combination, there was also one report of palpitations (2.7%). Palpitations were reported in one patient (5%) who received topical monotherapy, the only adverse event reported in this group.
Dr. Johnson noted that, at his institution, a dermatologist is conducting a clinical trial with oncology patients post chemotherapy and endocrine therapy. “She is looking at a similar question, although she is comparing oral minoxidil to topical minoxidil directly rather than in combination.” There is also an active clinical trial at Northwestern University, Chicago, of LDOM alone for patients with chemotherapy-induced alopecia.
“So there is a lot of momentum surrounding this concept, and I feel we will continue to see it come up as a possible treatment option, but more data are needed at this time before it can become standard of care,” Dr. Johnson added.
No funding for the study was reported. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Topical minoxidil is widely used to treat hair loss, but new findings suggest that
In a retrospective cohort study of women with breast cancer and anticancer therapy–induced alopecia, researchers found that combining low-dose oral minoxidil (LDOM) and topical minoxidil achieved better results than topical minoxidil alone and that the treatment was well tolerated. A total of 5 of the 37 patients (13.5%) in the combination therapy group achieved a complete response, defined as an improvement of alopecia severity from grade 2 to grade 1, compared with none of the 19 patients in the topical therapy–only group.
In contrast, none of the patients in the combination group experienced worsening of alopecia, compared with two (10.5%) in the topical monotherapy group.
The study was published online in the Journal of the American Academy of Dermatology. Topical minoxidil is approved by the Food and Drug Administration to treat androgenetic alopecia. Oral minoxidil is not approved for treating hair loss but has been receiving increased attention as an adjunctive therapy for hair loss, particularly for women. Oral minoxidil is approved for treating hypertension but at much higher doses.
An increasing number of studies have been conducted on the use of oral minoxidil for the treatment of female pattern hair loss, dating back to a pilot study in 2017, with promising results. The findings suggest that LDOM might be more effective than topical therapy, well tolerated, and more convenient for individuals to take.
Hypothesis generating
In a comment, Kai Johnson, MD, a medical oncologist who specializes in treating patients with breast cancer at the Ohio State University Comprehensive Cancer Center – Arthur G. James Cancer Hospital and Richard J. Solove Research Institute, Columbus, noted that the study, like most small-scale retrospective studies, is hypothesis generating. However, “I’d be hesitant to broadly recommend this practice of dual therapy – oral and topical minoxidil together – until we see a placebo-controlled prospective study performed demonstrating clinically meaningful benefits for patients.”
Another factor is the study endpoints. “While there was a statistically significant benefit documented with dual therapy in this study, it’s important to have study endpoints that are more patient oriented,” Dr. Johnson said. The most important endpoint for patients would be improvements “in the actual alopecia grade, which did occur in 5 of the 37 of dual-therapy patients, versus 0 topical minoxidil patients.”
George Cotsarelis, MD, chair of the department of dermatology and professor of dermatology at the University of Pennsylvania, Philadelphia, also weighed in. He questioned whether adding the topical therapy to oral minoxidil actually improved the results. “What was missing was a study arm that used the oral alone,” he said in an interview. “So we don’t know how effective the oral therapy would be by itself and if combining it with the topical is really adding anything.”
Oral minoxidil as a treatment for hair loss is gaining traction, and it’s clear that it is effective. However, the risk of side effects is higher, he said. “The risk isn’t that high with the low dose, but it can grow hair on places other than the scalp, and that can be disconcerting.” In this study, two women who took the oral drug reported edema, and one reported headache and dizziness. Hypertrichosis was reported by five patients who received the combination.
Study details
In the study, Jeewoo Kang, MD, and colleagues from the Seoul National University evaluated the efficacy of LDOM in 100 patients with breast cancer who had been diagnosed with persistent chemotherapy-induced alopecia (pCIA) and endocrine therapy–induced alopecia (EIA) at a dermatology clinic.
They conducted an analysis of medical records, standardized clinical photographs, and trichoscopic images to evaluate the alopecia pattern, severity, treatment response, and posttreatment changes in vertex hair density and thickness.
Compared with those with EIA alone, patients with pCIA were significantly more likely to have diffuse alopecia (P < .001), and they were more likely to have more severe alopecia, although this difference was not significant (P = .058). Outcomes were evaluated for 56 patients who were treated with minoxidil (19 with topical minoxidil alone and 37 with both LDOM and topical minoxidil) and for whom clinical and trichoscopic photos were available at baseline and at the last follow-up (all patients were scheduled for follow-up at 3-month intervals).
The results showed that those treated with 1.25-5.0 mg/d of oral minoxidil and 5% topical minoxidil solution once a day had better responses (P = .002) and a higher percentage increase in hair density from baseline (P = .003), compared with those who received topical minoxidil monotherapy.
However, changes in hair thickness after treatment were not significantly different between the two groups (P = .540).
In addition to the five (13.5%) cases of hypertrichosis, two cases of edema (5.4%), and one case of headache/dizziness (2.7%) among those who received the combination, there was also one report of palpitations (2.7%). Palpitations were reported in one patient (5%) who received topical monotherapy, the only adverse event reported in this group.
Dr. Johnson noted that, at his institution, a dermatologist is conducting a clinical trial with oncology patients post chemotherapy and endocrine therapy. “She is looking at a similar question, although she is comparing oral minoxidil to topical minoxidil directly rather than in combination.” There is also an active clinical trial at Northwestern University, Chicago, of LDOM alone for patients with chemotherapy-induced alopecia.
“So there is a lot of momentum surrounding this concept, and I feel we will continue to see it come up as a possible treatment option, but more data are needed at this time before it can become standard of care,” Dr. Johnson added.
No funding for the study was reported. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Topical minoxidil is widely used to treat hair loss, but new findings suggest that
In a retrospective cohort study of women with breast cancer and anticancer therapy–induced alopecia, researchers found that combining low-dose oral minoxidil (LDOM) and topical minoxidil achieved better results than topical minoxidil alone and that the treatment was well tolerated. A total of 5 of the 37 patients (13.5%) in the combination therapy group achieved a complete response, defined as an improvement of alopecia severity from grade 2 to grade 1, compared with none of the 19 patients in the topical therapy–only group.
In contrast, none of the patients in the combination group experienced worsening of alopecia, compared with two (10.5%) in the topical monotherapy group.
The study was published online in the Journal of the American Academy of Dermatology. Topical minoxidil is approved by the Food and Drug Administration to treat androgenetic alopecia. Oral minoxidil is not approved for treating hair loss but has been receiving increased attention as an adjunctive therapy for hair loss, particularly for women. Oral minoxidil is approved for treating hypertension but at much higher doses.
An increasing number of studies have been conducted on the use of oral minoxidil for the treatment of female pattern hair loss, dating back to a pilot study in 2017, with promising results. The findings suggest that LDOM might be more effective than topical therapy, well tolerated, and more convenient for individuals to take.
Hypothesis generating
In a comment, Kai Johnson, MD, a medical oncologist who specializes in treating patients with breast cancer at the Ohio State University Comprehensive Cancer Center – Arthur G. James Cancer Hospital and Richard J. Solove Research Institute, Columbus, noted that the study, like most small-scale retrospective studies, is hypothesis generating. However, “I’d be hesitant to broadly recommend this practice of dual therapy – oral and topical minoxidil together – until we see a placebo-controlled prospective study performed demonstrating clinically meaningful benefits for patients.”
Another factor is the study endpoints. “While there was a statistically significant benefit documented with dual therapy in this study, it’s important to have study endpoints that are more patient oriented,” Dr. Johnson said. The most important endpoint for patients would be improvements “in the actual alopecia grade, which did occur in 5 of the 37 of dual-therapy patients, versus 0 topical minoxidil patients.”
George Cotsarelis, MD, chair of the department of dermatology and professor of dermatology at the University of Pennsylvania, Philadelphia, also weighed in. He questioned whether adding the topical therapy to oral minoxidil actually improved the results. “What was missing was a study arm that used the oral alone,” he said in an interview. “So we don’t know how effective the oral therapy would be by itself and if combining it with the topical is really adding anything.”
Oral minoxidil as a treatment for hair loss is gaining traction, and it’s clear that it is effective. However, the risk of side effects is higher, he said. “The risk isn’t that high with the low dose, but it can grow hair on places other than the scalp, and that can be disconcerting.” In this study, two women who took the oral drug reported edema, and one reported headache and dizziness. Hypertrichosis was reported by five patients who received the combination.
Study details
In the study, Jeewoo Kang, MD, and colleagues from the Seoul National University evaluated the efficacy of LDOM in 100 patients with breast cancer who had been diagnosed with persistent chemotherapy-induced alopecia (pCIA) and endocrine therapy–induced alopecia (EIA) at a dermatology clinic.
They conducted an analysis of medical records, standardized clinical photographs, and trichoscopic images to evaluate the alopecia pattern, severity, treatment response, and posttreatment changes in vertex hair density and thickness.
Compared with those with EIA alone, patients with pCIA were significantly more likely to have diffuse alopecia (P < .001), and they were more likely to have more severe alopecia, although this difference was not significant (P = .058). Outcomes were evaluated for 56 patients who were treated with minoxidil (19 with topical minoxidil alone and 37 with both LDOM and topical minoxidil) and for whom clinical and trichoscopic photos were available at baseline and at the last follow-up (all patients were scheduled for follow-up at 3-month intervals).
The results showed that those treated with 1.25-5.0 mg/d of oral minoxidil and 5% topical minoxidil solution once a day had better responses (P = .002) and a higher percentage increase in hair density from baseline (P = .003), compared with those who received topical minoxidil monotherapy.
However, changes in hair thickness after treatment were not significantly different between the two groups (P = .540).
In addition to the five (13.5%) cases of hypertrichosis, two cases of edema (5.4%), and one case of headache/dizziness (2.7%) among those who received the combination, there was also one report of palpitations (2.7%). Palpitations were reported in one patient (5%) who received topical monotherapy, the only adverse event reported in this group.
Dr. Johnson noted that, at his institution, a dermatologist is conducting a clinical trial with oncology patients post chemotherapy and endocrine therapy. “She is looking at a similar question, although she is comparing oral minoxidil to topical minoxidil directly rather than in combination.” There is also an active clinical trial at Northwestern University, Chicago, of LDOM alone for patients with chemotherapy-induced alopecia.
“So there is a lot of momentum surrounding this concept, and I feel we will continue to see it come up as a possible treatment option, but more data are needed at this time before it can become standard of care,” Dr. Johnson added.
No funding for the study was reported. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Adverse events linked to better survival with ICIs in melanoma
Among Survival is further improved if the immunotherapy is continued after the adverse event develops, a new study confirms.
“In the largest clinical cohort to date, our data support a positive association with overall survival for patients who develop clinically significant immune-related adverse events while receiving combination immune checkpoint blockade, in keeping with other reported series,” the authors wrote.
The study was published online in JAMA Network Open.
Immune-related adverse events are common with these drugs. Severe events of grade 3 or higher occur in 59% of trial patients who receive combination ICI therapy.
The adverse events have increasingly been positively associated with survival. However, the effects for patients with metastatic melanoma, in particular, are less clear. There is little research on the effects in relation to combination therapy with ipilimumab and nivolumab, which is the standard of care for many patients with metastatic melanoma.
To investigate, Alexander S. Watson, MD, and colleagues evaluated data on 492 patients with metastatic melanoma who had been treated with one or more doses of an anti–programmed death 1 agent as single or combination immune checkpoint blockade in the multicenter Alberta Immunotherapy Database from August 2013 to May 2020.
Of these 492 patients, 198 patients (40%) developed immune-related adverse events. The mean age of the patients who developed adverse events was 61.8 years; of those who did not develop adverse events, the mean age was 65.5 years. Men made up 69.2% and 62.2%, respectively.
A total of 288 patients received pembrolizumab as their first ICI therapy, 80 received nivolumab, and 124 received combination blockade with ipilimumab-nivolumab.
Overall, with a median follow-up of 36.6 months, among patients who experienced clinically significant immune-related adverse events, defined as requiring systemic corticosteroids and/or a treatment delay, median overall survival was significantly improved, at 56.3 months, compared with 18.5 months among those who did not experience immune-related adverse events (P < .001).
In addition, among those who received combination ICI treatment, the median overall survival was 56.2 months for those who experienced adverse events versus 19.0 months for those who did not (P < .001).
There were no significant differences in overall survival between those who were and those who were not hospitalized for their immune-related adverse events (P = .53).
For patients who resumed their ICI therapy following the adverse events, overall survival was longer, compared with those who did not resume the therapy (median, 56.3 months vs. 31.5 months; P = .009).
The improvements in overall survival seen with immune-related adverse events remained consistent after adjustment in a multivariable analysis (hazard ratio for death, 0.382; P < .001).
There were no significant differences in the median number of cycles of ICIs between those with and those without the adverse events.
The risk of recurrence of immune-related adverse events following the reintroduction of therapy after initial events was a concern, so the improved overall survival among those patients is encouraging, although further investigation is needed, commented lead author Dr. Watson, from the department of oncology, University of Calgary (Alta.).
“It may be, for certain patients with immune-related adverse events, that continued immune-priming is safe and optimizes anticancer response,” he told this news organization. “However, in a retrospective analysis such as ours, selection bias can have an impact.”
“Confirming this finding and better identifying patients who may benefit from resumption will be an area for future investigation,” he said.
Patients who developed immune-related adverse events were more likely to be younger than 50 years (21.8% vs. 13.9%), have normal albumin levels (86.4% vs. 74.8%), and have a more robust Eastern Cooperative Oncology Group status, which is consistent with other studies that have shown survival benefits among those who experience adverse events.
“We, and others, speculate this could be due to such groups having immune systems more ready to respond strongly to immunotherapy,” Dr. Watson explained.
After controlling for age and performance status in the multivariable analysis, however, “immune-related adverse events remained strongly associated with survival, potentially [indicating] that robust responses to immunotherapy lead to both cancer control and immune-related adverse events,” he said.
Overall, “we feel these findings will help clinicians in discussions with patients and in clinical decision-making after adverse events develop,” Dr. Watson said.
Dr. Watson has received personal fees from Apobiologix Canada.
A version of this article first appeared on Medscape.com.
Among Survival is further improved if the immunotherapy is continued after the adverse event develops, a new study confirms.
“In the largest clinical cohort to date, our data support a positive association with overall survival for patients who develop clinically significant immune-related adverse events while receiving combination immune checkpoint blockade, in keeping with other reported series,” the authors wrote.
The study was published online in JAMA Network Open.
Immune-related adverse events are common with these drugs. Severe events of grade 3 or higher occur in 59% of trial patients who receive combination ICI therapy.
The adverse events have increasingly been positively associated with survival. However, the effects for patients with metastatic melanoma, in particular, are less clear. There is little research on the effects in relation to combination therapy with ipilimumab and nivolumab, which is the standard of care for many patients with metastatic melanoma.
To investigate, Alexander S. Watson, MD, and colleagues evaluated data on 492 patients with metastatic melanoma who had been treated with one or more doses of an anti–programmed death 1 agent as single or combination immune checkpoint blockade in the multicenter Alberta Immunotherapy Database from August 2013 to May 2020.
Of these 492 patients, 198 patients (40%) developed immune-related adverse events. The mean age of the patients who developed adverse events was 61.8 years; of those who did not develop adverse events, the mean age was 65.5 years. Men made up 69.2% and 62.2%, respectively.
A total of 288 patients received pembrolizumab as their first ICI therapy, 80 received nivolumab, and 124 received combination blockade with ipilimumab-nivolumab.
Overall, with a median follow-up of 36.6 months, among patients who experienced clinically significant immune-related adverse events, defined as requiring systemic corticosteroids and/or a treatment delay, median overall survival was significantly improved, at 56.3 months, compared with 18.5 months among those who did not experience immune-related adverse events (P < .001).
In addition, among those who received combination ICI treatment, the median overall survival was 56.2 months for those who experienced adverse events versus 19.0 months for those who did not (P < .001).
There were no significant differences in overall survival between those who were and those who were not hospitalized for their immune-related adverse events (P = .53).
For patients who resumed their ICI therapy following the adverse events, overall survival was longer, compared with those who did not resume the therapy (median, 56.3 months vs. 31.5 months; P = .009).
The improvements in overall survival seen with immune-related adverse events remained consistent after adjustment in a multivariable analysis (hazard ratio for death, 0.382; P < .001).
There were no significant differences in the median number of cycles of ICIs between those with and those without the adverse events.
The risk of recurrence of immune-related adverse events following the reintroduction of therapy after initial events was a concern, so the improved overall survival among those patients is encouraging, although further investigation is needed, commented lead author Dr. Watson, from the department of oncology, University of Calgary (Alta.).
“It may be, for certain patients with immune-related adverse events, that continued immune-priming is safe and optimizes anticancer response,” he told this news organization. “However, in a retrospective analysis such as ours, selection bias can have an impact.”
“Confirming this finding and better identifying patients who may benefit from resumption will be an area for future investigation,” he said.
Patients who developed immune-related adverse events were more likely to be younger than 50 years (21.8% vs. 13.9%), have normal albumin levels (86.4% vs. 74.8%), and have a more robust Eastern Cooperative Oncology Group status, which is consistent with other studies that have shown survival benefits among those who experience adverse events.
“We, and others, speculate this could be due to such groups having immune systems more ready to respond strongly to immunotherapy,” Dr. Watson explained.
After controlling for age and performance status in the multivariable analysis, however, “immune-related adverse events remained strongly associated with survival, potentially [indicating] that robust responses to immunotherapy lead to both cancer control and immune-related adverse events,” he said.
Overall, “we feel these findings will help clinicians in discussions with patients and in clinical decision-making after adverse events develop,” Dr. Watson said.
Dr. Watson has received personal fees from Apobiologix Canada.
A version of this article first appeared on Medscape.com.
Among Survival is further improved if the immunotherapy is continued after the adverse event develops, a new study confirms.
“In the largest clinical cohort to date, our data support a positive association with overall survival for patients who develop clinically significant immune-related adverse events while receiving combination immune checkpoint blockade, in keeping with other reported series,” the authors wrote.
The study was published online in JAMA Network Open.
Immune-related adverse events are common with these drugs. Severe events of grade 3 or higher occur in 59% of trial patients who receive combination ICI therapy.
The adverse events have increasingly been positively associated with survival. However, the effects for patients with metastatic melanoma, in particular, are less clear. There is little research on the effects in relation to combination therapy with ipilimumab and nivolumab, which is the standard of care for many patients with metastatic melanoma.
To investigate, Alexander S. Watson, MD, and colleagues evaluated data on 492 patients with metastatic melanoma who had been treated with one or more doses of an anti–programmed death 1 agent as single or combination immune checkpoint blockade in the multicenter Alberta Immunotherapy Database from August 2013 to May 2020.
Of these 492 patients, 198 patients (40%) developed immune-related adverse events. The mean age of the patients who developed adverse events was 61.8 years; of those who did not develop adverse events, the mean age was 65.5 years. Men made up 69.2% and 62.2%, respectively.
A total of 288 patients received pembrolizumab as their first ICI therapy, 80 received nivolumab, and 124 received combination blockade with ipilimumab-nivolumab.
Overall, with a median follow-up of 36.6 months, among patients who experienced clinically significant immune-related adverse events, defined as requiring systemic corticosteroids and/or a treatment delay, median overall survival was significantly improved, at 56.3 months, compared with 18.5 months among those who did not experience immune-related adverse events (P < .001).
In addition, among those who received combination ICI treatment, the median overall survival was 56.2 months for those who experienced adverse events versus 19.0 months for those who did not (P < .001).
There were no significant differences in overall survival between those who were and those who were not hospitalized for their immune-related adverse events (P = .53).
For patients who resumed their ICI therapy following the adverse events, overall survival was longer, compared with those who did not resume the therapy (median, 56.3 months vs. 31.5 months; P = .009).
The improvements in overall survival seen with immune-related adverse events remained consistent after adjustment in a multivariable analysis (hazard ratio for death, 0.382; P < .001).
There were no significant differences in the median number of cycles of ICIs between those with and those without the adverse events.
The risk of recurrence of immune-related adverse events following the reintroduction of therapy after initial events was a concern, so the improved overall survival among those patients is encouraging, although further investigation is needed, commented lead author Dr. Watson, from the department of oncology, University of Calgary (Alta.).
“It may be, for certain patients with immune-related adverse events, that continued immune-priming is safe and optimizes anticancer response,” he told this news organization. “However, in a retrospective analysis such as ours, selection bias can have an impact.”
“Confirming this finding and better identifying patients who may benefit from resumption will be an area for future investigation,” he said.
Patients who developed immune-related adverse events were more likely to be younger than 50 years (21.8% vs. 13.9%), have normal albumin levels (86.4% vs. 74.8%), and have a more robust Eastern Cooperative Oncology Group status, which is consistent with other studies that have shown survival benefits among those who experience adverse events.
“We, and others, speculate this could be due to such groups having immune systems more ready to respond strongly to immunotherapy,” Dr. Watson explained.
After controlling for age and performance status in the multivariable analysis, however, “immune-related adverse events remained strongly associated with survival, potentially [indicating] that robust responses to immunotherapy lead to both cancer control and immune-related adverse events,” he said.
Overall, “we feel these findings will help clinicians in discussions with patients and in clinical decision-making after adverse events develop,” Dr. Watson said.
Dr. Watson has received personal fees from Apobiologix Canada.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Topical psoriasis treatments
LAS VEGAS – ,” said Linda Stein Gold, MD, in a presentation at Medscape Live’s annual Las Vegas Dermatology Seminar.
However, when using topical treatments, combination therapy is generally more effective than monotherapy for psoriasis, especially for plaque psoriasis, said Dr. Stein Gold, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit.
Two combination products, calcipotriene/betamethasone (CAL/BDP) and tazarotene/halobetasol lotion, each offer a complimentary mechanism of action that minimizes side effects, with decreased irritation and less atrophy, she said. Calcipotriene/betamethasone (CAL/BDP) is available as a cream or foam, Dr. Stein Gold noted. The cream is engineered for rapid onset, as well as enhanced penetration, she said. CAL/BDP foam also is designed for enhanced penetration, and has been shown to have long-term maintenance efficacy, she said.
The currently available CAL/BDP cream is made using a patented technology known as “PAD,” in which the internal oil of the cream vehicle is stabilized by encapsulation in “a robust aqueous film,” Dr. Stein Gold said, noting that the greater solubility enhances skin penetration. The creation of “a robust oil droplet” addresses the problems associated with the surfactants present in many cream vehicles, namely irritation and impedance of skin penetration of the cream, she said.
In an 8-week study published in 2021, researchers compared CAL/BDP cream with PAD technology to CAL/BDP topical suspension in adults with mild to moderate psoriasis.
Patients randomized to treatment with CAL/BDP cream were significantly more likely to achieve the primary endpoint of Physician Global Assessment (PGA) treatment success than those randomized to the topical solution or vehicle (37.4%, 22.8%, and 3.7%, respectively).
Get proactive to maintain results
With topical psoriasis treatment, a proactive strategy helps maintain results over time, Dr. Stein Gold said. As an example, she cited a study published in 2021. In that study, known as PSO-LONG, which evaluated topical CAL/BDP foam, proactive management with the CAL/BDP foam formulation, “reduced the risk of experiencing relapse by 43%,” compared with reactive management (treatment with the vehicle foam), she said. Patients in the proactive-management group experienced an average of 41 more days in remission, compared with those in the reactive management group over a 1-year period.
Dr. Stein Gold also highlighted the value of tazarotene/halobetasol lotion for psoriasis, which she described as having synergistic efficacy,
She shared data presented at the 2021 Maui Dermatology meeting showing treatment success by 8 weeks with halobetasol/tazarotene with significantly reduced mean scores on measures of itching, dryness, and burning/stinging, compared with those on vehicle.
What’s new and approved
Joining the current topical treatment options for psoriasis is tapinarof, a small molecule that works by down-regulating Th17 cytokines, said Dr. Stein Gold. Tapinarof is Food and Drug Administration approved for treating psoriasis and is being studied in clinical trials for atopic dermatitis, she noted.
Dr. Stein Gold reviewed data from the PSOARING program published in the New England Journal of Medicine that served as a foundation for the FDA approval of tapinarof 1% cream. In the PSOARING 1 and 2 studies, patients with PSORIASIS showed significant improvement compared with vehicle over 12 weeks for the primary endpoint of Physicians’ Global Assessment scores of 0 or 1 (clear or almost clear). In the two studies, 60.7% and 56.9% of patients randomized to tapinarof met the patient-reported outcome of a minimum 4-point improvement in peak pruritus on the numerical rating scale (NRS) from baseline vs. 43.2% and 29.7% of placebo patients in the two studies, respectively.
In PSOARING 1 and 2, folliculitis (mostly mild or moderate), contact dermatitis, headache, pruritus, and dermatitis were the most common treatment-emergent adverse events, occurring in 1% or more of patients. Adverse event profiles for tapinarof are similar to those seen in previous studies, and a long-term extension showed a consistent safety profile, Dr. Stein Gold said.
Another recently approved topical treatment for psoriasis, a cream formulation of roflumilast, a phosphodiesterase (PDE)-4 inhibitor, has shown efficacy for treating plaque psoriasis, she said.
Patients with psoriasis in the DERMIS 1 and DERMIS 2 phase 3 studies randomized to 0.3% roflumilast cream showed significant improvement compared with those randomized to vehicle in terms of Investigator Global Assessment scores of clear or almost clear with an improvement of at least 2 grades from baseline.
Roflumilast foam also has shown success in improving scalp and body psoriasis, but this vehicle and indication has not yet been approved, Dr. Stein Gold said.
Dr. Stein Gold disclosed serving as a consultant or adviser for companies including AbbVie, Amgen, Arcutis, Bristol Myers Squibb, Dermavant, EPI Health, Galderma, Janssen, Incyte, Ortho Dermatologics, Pfizer, Regeneron, Sanofi; UCB, and serving as a speaker or member of speakers’ bureau for Amgen, AbbVie, Incyte, Pfizer, Regeneron, Sanofi, and Sun Research. She also disclosed receiving funding from AbbVie Amgen, Arcutis, Dermata, Dermavant, Galderma, Incyte, Ortho Dermatologics, Pfizer, and UCB.
MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – ,” said Linda Stein Gold, MD, in a presentation at Medscape Live’s annual Las Vegas Dermatology Seminar.
However, when using topical treatments, combination therapy is generally more effective than monotherapy for psoriasis, especially for plaque psoriasis, said Dr. Stein Gold, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit.
Two combination products, calcipotriene/betamethasone (CAL/BDP) and tazarotene/halobetasol lotion, each offer a complimentary mechanism of action that minimizes side effects, with decreased irritation and less atrophy, she said. Calcipotriene/betamethasone (CAL/BDP) is available as a cream or foam, Dr. Stein Gold noted. The cream is engineered for rapid onset, as well as enhanced penetration, she said. CAL/BDP foam also is designed for enhanced penetration, and has been shown to have long-term maintenance efficacy, she said.
The currently available CAL/BDP cream is made using a patented technology known as “PAD,” in which the internal oil of the cream vehicle is stabilized by encapsulation in “a robust aqueous film,” Dr. Stein Gold said, noting that the greater solubility enhances skin penetration. The creation of “a robust oil droplet” addresses the problems associated with the surfactants present in many cream vehicles, namely irritation and impedance of skin penetration of the cream, she said.
In an 8-week study published in 2021, researchers compared CAL/BDP cream with PAD technology to CAL/BDP topical suspension in adults with mild to moderate psoriasis.
Patients randomized to treatment with CAL/BDP cream were significantly more likely to achieve the primary endpoint of Physician Global Assessment (PGA) treatment success than those randomized to the topical solution or vehicle (37.4%, 22.8%, and 3.7%, respectively).
Get proactive to maintain results
With topical psoriasis treatment, a proactive strategy helps maintain results over time, Dr. Stein Gold said. As an example, she cited a study published in 2021. In that study, known as PSO-LONG, which evaluated topical CAL/BDP foam, proactive management with the CAL/BDP foam formulation, “reduced the risk of experiencing relapse by 43%,” compared with reactive management (treatment with the vehicle foam), she said. Patients in the proactive-management group experienced an average of 41 more days in remission, compared with those in the reactive management group over a 1-year period.
Dr. Stein Gold also highlighted the value of tazarotene/halobetasol lotion for psoriasis, which she described as having synergistic efficacy,
She shared data presented at the 2021 Maui Dermatology meeting showing treatment success by 8 weeks with halobetasol/tazarotene with significantly reduced mean scores on measures of itching, dryness, and burning/stinging, compared with those on vehicle.
What’s new and approved
Joining the current topical treatment options for psoriasis is tapinarof, a small molecule that works by down-regulating Th17 cytokines, said Dr. Stein Gold. Tapinarof is Food and Drug Administration approved for treating psoriasis and is being studied in clinical trials for atopic dermatitis, she noted.
Dr. Stein Gold reviewed data from the PSOARING program published in the New England Journal of Medicine that served as a foundation for the FDA approval of tapinarof 1% cream. In the PSOARING 1 and 2 studies, patients with PSORIASIS showed significant improvement compared with vehicle over 12 weeks for the primary endpoint of Physicians’ Global Assessment scores of 0 or 1 (clear or almost clear). In the two studies, 60.7% and 56.9% of patients randomized to tapinarof met the patient-reported outcome of a minimum 4-point improvement in peak pruritus on the numerical rating scale (NRS) from baseline vs. 43.2% and 29.7% of placebo patients in the two studies, respectively.
In PSOARING 1 and 2, folliculitis (mostly mild or moderate), contact dermatitis, headache, pruritus, and dermatitis were the most common treatment-emergent adverse events, occurring in 1% or more of patients. Adverse event profiles for tapinarof are similar to those seen in previous studies, and a long-term extension showed a consistent safety profile, Dr. Stein Gold said.
Another recently approved topical treatment for psoriasis, a cream formulation of roflumilast, a phosphodiesterase (PDE)-4 inhibitor, has shown efficacy for treating plaque psoriasis, she said.
Patients with psoriasis in the DERMIS 1 and DERMIS 2 phase 3 studies randomized to 0.3% roflumilast cream showed significant improvement compared with those randomized to vehicle in terms of Investigator Global Assessment scores of clear or almost clear with an improvement of at least 2 grades from baseline.
Roflumilast foam also has shown success in improving scalp and body psoriasis, but this vehicle and indication has not yet been approved, Dr. Stein Gold said.
Dr. Stein Gold disclosed serving as a consultant or adviser for companies including AbbVie, Amgen, Arcutis, Bristol Myers Squibb, Dermavant, EPI Health, Galderma, Janssen, Incyte, Ortho Dermatologics, Pfizer, Regeneron, Sanofi; UCB, and serving as a speaker or member of speakers’ bureau for Amgen, AbbVie, Incyte, Pfizer, Regeneron, Sanofi, and Sun Research. She also disclosed receiving funding from AbbVie Amgen, Arcutis, Dermata, Dermavant, Galderma, Incyte, Ortho Dermatologics, Pfizer, and UCB.
MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – ,” said Linda Stein Gold, MD, in a presentation at Medscape Live’s annual Las Vegas Dermatology Seminar.
However, when using topical treatments, combination therapy is generally more effective than monotherapy for psoriasis, especially for plaque psoriasis, said Dr. Stein Gold, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit.
Two combination products, calcipotriene/betamethasone (CAL/BDP) and tazarotene/halobetasol lotion, each offer a complimentary mechanism of action that minimizes side effects, with decreased irritation and less atrophy, she said. Calcipotriene/betamethasone (CAL/BDP) is available as a cream or foam, Dr. Stein Gold noted. The cream is engineered for rapid onset, as well as enhanced penetration, she said. CAL/BDP foam also is designed for enhanced penetration, and has been shown to have long-term maintenance efficacy, she said.
The currently available CAL/BDP cream is made using a patented technology known as “PAD,” in which the internal oil of the cream vehicle is stabilized by encapsulation in “a robust aqueous film,” Dr. Stein Gold said, noting that the greater solubility enhances skin penetration. The creation of “a robust oil droplet” addresses the problems associated with the surfactants present in many cream vehicles, namely irritation and impedance of skin penetration of the cream, she said.
In an 8-week study published in 2021, researchers compared CAL/BDP cream with PAD technology to CAL/BDP topical suspension in adults with mild to moderate psoriasis.
Patients randomized to treatment with CAL/BDP cream were significantly more likely to achieve the primary endpoint of Physician Global Assessment (PGA) treatment success than those randomized to the topical solution or vehicle (37.4%, 22.8%, and 3.7%, respectively).
Get proactive to maintain results
With topical psoriasis treatment, a proactive strategy helps maintain results over time, Dr. Stein Gold said. As an example, she cited a study published in 2021. In that study, known as PSO-LONG, which evaluated topical CAL/BDP foam, proactive management with the CAL/BDP foam formulation, “reduced the risk of experiencing relapse by 43%,” compared with reactive management (treatment with the vehicle foam), she said. Patients in the proactive-management group experienced an average of 41 more days in remission, compared with those in the reactive management group over a 1-year period.
Dr. Stein Gold also highlighted the value of tazarotene/halobetasol lotion for psoriasis, which she described as having synergistic efficacy,
She shared data presented at the 2021 Maui Dermatology meeting showing treatment success by 8 weeks with halobetasol/tazarotene with significantly reduced mean scores on measures of itching, dryness, and burning/stinging, compared with those on vehicle.
What’s new and approved
Joining the current topical treatment options for psoriasis is tapinarof, a small molecule that works by down-regulating Th17 cytokines, said Dr. Stein Gold. Tapinarof is Food and Drug Administration approved for treating psoriasis and is being studied in clinical trials for atopic dermatitis, she noted.
Dr. Stein Gold reviewed data from the PSOARING program published in the New England Journal of Medicine that served as a foundation for the FDA approval of tapinarof 1% cream. In the PSOARING 1 and 2 studies, patients with PSORIASIS showed significant improvement compared with vehicle over 12 weeks for the primary endpoint of Physicians’ Global Assessment scores of 0 or 1 (clear or almost clear). In the two studies, 60.7% and 56.9% of patients randomized to tapinarof met the patient-reported outcome of a minimum 4-point improvement in peak pruritus on the numerical rating scale (NRS) from baseline vs. 43.2% and 29.7% of placebo patients in the two studies, respectively.
In PSOARING 1 and 2, folliculitis (mostly mild or moderate), contact dermatitis, headache, pruritus, and dermatitis were the most common treatment-emergent adverse events, occurring in 1% or more of patients. Adverse event profiles for tapinarof are similar to those seen in previous studies, and a long-term extension showed a consistent safety profile, Dr. Stein Gold said.
Another recently approved topical treatment for psoriasis, a cream formulation of roflumilast, a phosphodiesterase (PDE)-4 inhibitor, has shown efficacy for treating plaque psoriasis, she said.
Patients with psoriasis in the DERMIS 1 and DERMIS 2 phase 3 studies randomized to 0.3% roflumilast cream showed significant improvement compared with those randomized to vehicle in terms of Investigator Global Assessment scores of clear or almost clear with an improvement of at least 2 grades from baseline.
Roflumilast foam also has shown success in improving scalp and body psoriasis, but this vehicle and indication has not yet been approved, Dr. Stein Gold said.
Dr. Stein Gold disclosed serving as a consultant or adviser for companies including AbbVie, Amgen, Arcutis, Bristol Myers Squibb, Dermavant, EPI Health, Galderma, Janssen, Incyte, Ortho Dermatologics, Pfizer, Regeneron, Sanofi; UCB, and serving as a speaker or member of speakers’ bureau for Amgen, AbbVie, Incyte, Pfizer, Regeneron, Sanofi, and Sun Research. She also disclosed receiving funding from AbbVie Amgen, Arcutis, Dermata, Dermavant, Galderma, Incyte, Ortho Dermatologics, Pfizer, and UCB.
MedscapeLive and this news organization are owned by the same parent company.
AT INNOVATIONS IN DERMATOLOGY
Cosmetic medicine expert shares male facial aesthetics pearls
SAN DIEGO – .
“Men generally have larger facial muscle mass,” Dr. Green, a dermatologist in Coral Gables, Fla., said at the annual Masters of Aesthetics Symposium. “We need a higher dose to treat them, or they will not be happy. In general, I try to increase the dose by about 50% for my male patients.”
Two early trials of dose adjustments support this practice, he said. In one, 80 men were randomized to receive a total dose of either 20, 40, 60, or 80 U of botulinum toxin type A (Botox) in the glabellar area. The researchers found that the 40, 60, and 80 U doses of botulinum toxin type A were consistently more effective in reducing glabellar lines than the 20 U dose.
In a subsequent study, researchers administered botulinum toxin type A (Dysport) 0.5 to 0.7 mL for men (60, 70, or 80 units), based on procerus/corrugator muscle mass. Efficacy was assessed by a blinded evaluator and patient self-evaluation at several time points up to 150 days post treatment. The median duration of effect was 109 days vs. 0 days for placebo in the blinded evaluator evaluation and 107 days vs. 0 for placebo in the patient self-evaluation.
Most injection algorithms for treating the glabella rely on a 5- or 7-point injection technique, but in 2021, researchers led by Sebastian Cotofana, MD, PhD, of the department of clinical anatomy at Mayo Clinic, Rochester, Minn., reported results from a study of the efficacy and safety of a refined 3-point injection technique targeting horizontal and vertical lines to prevent brow ptosis.
“Prior to this study Sebastian asked me, ‘Why do you guys always inject the body of the muscle?’ ” Dr. Green said. “‘If you inject the origin of the muscle on bone, you could more effectively wipe out the entire muscle’s movement. You’re going to get a better result at a lower dose, so let’s study this.’”
The injection technique involves targeting the midline level of the connecting line between left and right medial canthal ligaments with a 90-degree injection angle with bone contact, as well as the medial and inferior margin of eyebrows with a 45-degree injection angle in relation to midline with frontal bone contact. These three points are located inferior to the traditional (on-label) glabellar frown line injections used to treat the frontalis and the brow depressors.
The researchers used the 5-point glabellar line severity scale to evaluate the time of effect onset and the injection-related outcome 120 days after the treatment in 27 men and 78 women. They found that the onset of the neuromodulator effect occurred in an average of 3.5 days, and no adverse events such as eyebrow ptosis, upper eyelid ptosis, medial eyebrow ptosis, and lateral frontalis hyperactivity occurred during the study period.
“If you inject the origin of these muscles, you can get a brow lift with this technique by avoiding frontalis altogether,” Dr. Green said. “The caveat is, it’s so great at lifting the brows that if you treat the forehead, you may create a midline horizontal ‘shelf’ like I’ve never seen before, where the eyebrows elevate into an immobile superior frontalis.”
To avoid this when treating the forehead as well, he’s learned to split the dose of neuromodulator. “If I was injecting 5 units in the procerus before, I’ll do 2.5 units on nasal bone at the insertion of the muscle and then 2.5 units higher up in the traditional midline procerus injection site,” Dr. Green said.
“Same with the corrugators,” he continued. “Then, remember to inject more superficially in the lateral part, the tail of the corrugators, because the tail of the corrugators is inserting into the undersurface of the dermis. That’s why you see that skin puckering in the lateral brows when people frown. You’re pretty safe to chase that laterally if the brow’s already flat as in men, but I caution you [not] to do that in women, because you may flatten the brow.”
Dr. Green said that he is aware of two cases of lid ptosis from the 3-point technique, one of which happened to him.
“When you’re on the bone with your thumb you can feel that liquid traveling along the bone,” he said. “It can travel all the way to the midline pupil where the levator palpebrae superioris muscle is. I now don’t come in contact with bone with my corrugator origin injections, but rather float the needle a couple of millimeters off bone (in muscle) to hopefully prevent that from happening. Alternatively, some people will compress the brow along frontal bone lateral to that corrugator injection site while they’re injecting to prevent backflow of the neuromodulator.”
Dr. Green reported having received research funding and/or consulting fees from many device and pharmaceutical companies.
SAN DIEGO – .
“Men generally have larger facial muscle mass,” Dr. Green, a dermatologist in Coral Gables, Fla., said at the annual Masters of Aesthetics Symposium. “We need a higher dose to treat them, or they will not be happy. In general, I try to increase the dose by about 50% for my male patients.”
Two early trials of dose adjustments support this practice, he said. In one, 80 men were randomized to receive a total dose of either 20, 40, 60, or 80 U of botulinum toxin type A (Botox) in the glabellar area. The researchers found that the 40, 60, and 80 U doses of botulinum toxin type A were consistently more effective in reducing glabellar lines than the 20 U dose.
In a subsequent study, researchers administered botulinum toxin type A (Dysport) 0.5 to 0.7 mL for men (60, 70, or 80 units), based on procerus/corrugator muscle mass. Efficacy was assessed by a blinded evaluator and patient self-evaluation at several time points up to 150 days post treatment. The median duration of effect was 109 days vs. 0 days for placebo in the blinded evaluator evaluation and 107 days vs. 0 for placebo in the patient self-evaluation.
Most injection algorithms for treating the glabella rely on a 5- or 7-point injection technique, but in 2021, researchers led by Sebastian Cotofana, MD, PhD, of the department of clinical anatomy at Mayo Clinic, Rochester, Minn., reported results from a study of the efficacy and safety of a refined 3-point injection technique targeting horizontal and vertical lines to prevent brow ptosis.
“Prior to this study Sebastian asked me, ‘Why do you guys always inject the body of the muscle?’ ” Dr. Green said. “‘If you inject the origin of the muscle on bone, you could more effectively wipe out the entire muscle’s movement. You’re going to get a better result at a lower dose, so let’s study this.’”
The injection technique involves targeting the midline level of the connecting line between left and right medial canthal ligaments with a 90-degree injection angle with bone contact, as well as the medial and inferior margin of eyebrows with a 45-degree injection angle in relation to midline with frontal bone contact. These three points are located inferior to the traditional (on-label) glabellar frown line injections used to treat the frontalis and the brow depressors.
The researchers used the 5-point glabellar line severity scale to evaluate the time of effect onset and the injection-related outcome 120 days after the treatment in 27 men and 78 women. They found that the onset of the neuromodulator effect occurred in an average of 3.5 days, and no adverse events such as eyebrow ptosis, upper eyelid ptosis, medial eyebrow ptosis, and lateral frontalis hyperactivity occurred during the study period.
“If you inject the origin of these muscles, you can get a brow lift with this technique by avoiding frontalis altogether,” Dr. Green said. “The caveat is, it’s so great at lifting the brows that if you treat the forehead, you may create a midline horizontal ‘shelf’ like I’ve never seen before, where the eyebrows elevate into an immobile superior frontalis.”
To avoid this when treating the forehead as well, he’s learned to split the dose of neuromodulator. “If I was injecting 5 units in the procerus before, I’ll do 2.5 units on nasal bone at the insertion of the muscle and then 2.5 units higher up in the traditional midline procerus injection site,” Dr. Green said.
“Same with the corrugators,” he continued. “Then, remember to inject more superficially in the lateral part, the tail of the corrugators, because the tail of the corrugators is inserting into the undersurface of the dermis. That’s why you see that skin puckering in the lateral brows when people frown. You’re pretty safe to chase that laterally if the brow’s already flat as in men, but I caution you [not] to do that in women, because you may flatten the brow.”
Dr. Green said that he is aware of two cases of lid ptosis from the 3-point technique, one of which happened to him.
“When you’re on the bone with your thumb you can feel that liquid traveling along the bone,” he said. “It can travel all the way to the midline pupil where the levator palpebrae superioris muscle is. I now don’t come in contact with bone with my corrugator origin injections, but rather float the needle a couple of millimeters off bone (in muscle) to hopefully prevent that from happening. Alternatively, some people will compress the brow along frontal bone lateral to that corrugator injection site while they’re injecting to prevent backflow of the neuromodulator.”
Dr. Green reported having received research funding and/or consulting fees from many device and pharmaceutical companies.
SAN DIEGO – .
“Men generally have larger facial muscle mass,” Dr. Green, a dermatologist in Coral Gables, Fla., said at the annual Masters of Aesthetics Symposium. “We need a higher dose to treat them, or they will not be happy. In general, I try to increase the dose by about 50% for my male patients.”
Two early trials of dose adjustments support this practice, he said. In one, 80 men were randomized to receive a total dose of either 20, 40, 60, or 80 U of botulinum toxin type A (Botox) in the glabellar area. The researchers found that the 40, 60, and 80 U doses of botulinum toxin type A were consistently more effective in reducing glabellar lines than the 20 U dose.
In a subsequent study, researchers administered botulinum toxin type A (Dysport) 0.5 to 0.7 mL for men (60, 70, or 80 units), based on procerus/corrugator muscle mass. Efficacy was assessed by a blinded evaluator and patient self-evaluation at several time points up to 150 days post treatment. The median duration of effect was 109 days vs. 0 days for placebo in the blinded evaluator evaluation and 107 days vs. 0 for placebo in the patient self-evaluation.
Most injection algorithms for treating the glabella rely on a 5- or 7-point injection technique, but in 2021, researchers led by Sebastian Cotofana, MD, PhD, of the department of clinical anatomy at Mayo Clinic, Rochester, Minn., reported results from a study of the efficacy and safety of a refined 3-point injection technique targeting horizontal and vertical lines to prevent brow ptosis.
“Prior to this study Sebastian asked me, ‘Why do you guys always inject the body of the muscle?’ ” Dr. Green said. “‘If you inject the origin of the muscle on bone, you could more effectively wipe out the entire muscle’s movement. You’re going to get a better result at a lower dose, so let’s study this.’”
The injection technique involves targeting the midline level of the connecting line between left and right medial canthal ligaments with a 90-degree injection angle with bone contact, as well as the medial and inferior margin of eyebrows with a 45-degree injection angle in relation to midline with frontal bone contact. These three points are located inferior to the traditional (on-label) glabellar frown line injections used to treat the frontalis and the brow depressors.
The researchers used the 5-point glabellar line severity scale to evaluate the time of effect onset and the injection-related outcome 120 days after the treatment in 27 men and 78 women. They found that the onset of the neuromodulator effect occurred in an average of 3.5 days, and no adverse events such as eyebrow ptosis, upper eyelid ptosis, medial eyebrow ptosis, and lateral frontalis hyperactivity occurred during the study period.
“If you inject the origin of these muscles, you can get a brow lift with this technique by avoiding frontalis altogether,” Dr. Green said. “The caveat is, it’s so great at lifting the brows that if you treat the forehead, you may create a midline horizontal ‘shelf’ like I’ve never seen before, where the eyebrows elevate into an immobile superior frontalis.”
To avoid this when treating the forehead as well, he’s learned to split the dose of neuromodulator. “If I was injecting 5 units in the procerus before, I’ll do 2.5 units on nasal bone at the insertion of the muscle and then 2.5 units higher up in the traditional midline procerus injection site,” Dr. Green said.
“Same with the corrugators,” he continued. “Then, remember to inject more superficially in the lateral part, the tail of the corrugators, because the tail of the corrugators is inserting into the undersurface of the dermis. That’s why you see that skin puckering in the lateral brows when people frown. You’re pretty safe to chase that laterally if the brow’s already flat as in men, but I caution you [not] to do that in women, because you may flatten the brow.”
Dr. Green said that he is aware of two cases of lid ptosis from the 3-point technique, one of which happened to him.
“When you’re on the bone with your thumb you can feel that liquid traveling along the bone,” he said. “It can travel all the way to the midline pupil where the levator palpebrae superioris muscle is. I now don’t come in contact with bone with my corrugator origin injections, but rather float the needle a couple of millimeters off bone (in muscle) to hopefully prevent that from happening. Alternatively, some people will compress the brow along frontal bone lateral to that corrugator injection site while they’re injecting to prevent backflow of the neuromodulator.”
Dr. Green reported having received research funding and/or consulting fees from many device and pharmaceutical companies.
AT MOAS 2022
Study evaluates features of alopecia areata in Hispanic/Latinx patients
.
Those are among key findings from a retrospective analysis of Hispanic/Latinx patients at the University of California, Irvine (UCI) by Natasha Mesinkovska, MD, PhD, of UCI’s department of dermatology, and her coauthors. The findings were published online in the Journal of the American Academy of Dermatology.
A recent study examined the epidemiology of alopecia areata (AA) in Black patients, wrote Dr. Mesinkovska and coauthors Celine Phong, a UCI medical student, and Amy J. McMichael, MD, professor of dermatology at Wake Forest University, Winston-Salem, N.C. “A similar unmet need exists to describe the characteristics of AA in Hispanic/Latinx (H/L) patients, the prevalent majority in California,” they added.
Drawing from chart reviews, ICD codes, and documented physical exams, they retrospectively identified 197 Hispanic/Latinx patients diagnosed with AA at UCI between 2015 and 2022, including alopecia totalis and alopecia universalis.
Nearly two-thirds of patients with alopecia were female (63%), and their mean age at diagnosis was 33 years. Most patients (79%) presented with patchy pattern AA, 13% had diffuse pattern AA, and only 12% had eyebrow, eyelash, or beard involvement. The most common comorbidity in patients overall was atopy (24%), including allergic rhinitis (12%), asthma (10%), and/or atopic dermatitis (7%).
The authors found that 18% of patients had one or more coexisting autoimmune conditions, most commonly rheumatoid arthritis (9%) and thyroid disease (6%). No patients had celiac disease, myasthenia gravis, or inflammatory bowel disease, but 43% had another dermatologic condition.
In other findings, 22% of patients had vitamin D deficiency, 20% had hyperlipidemia, 18% had obesity, 16% had gastroesophageal reflux disease, and 12% had anemia. At the same time, depression, anxiety, or sleep disorders were identified in 14% of patients.
“Interestingly, the most common autoimmune comorbidity in H/L was rheumatoid arthritis, compared to thyroid disease in Black patients and overall AA patients,” the authors wrote. “This finding may be a reflection of a larger trend, as rheumatoid arthritis in the H/L population has been on the rise.”
The authors acknowledged certain limitations of the study including its small sample size and lack of a control group, and reported having no financial disclosures.
.
Those are among key findings from a retrospective analysis of Hispanic/Latinx patients at the University of California, Irvine (UCI) by Natasha Mesinkovska, MD, PhD, of UCI’s department of dermatology, and her coauthors. The findings were published online in the Journal of the American Academy of Dermatology.
A recent study examined the epidemiology of alopecia areata (AA) in Black patients, wrote Dr. Mesinkovska and coauthors Celine Phong, a UCI medical student, and Amy J. McMichael, MD, professor of dermatology at Wake Forest University, Winston-Salem, N.C. “A similar unmet need exists to describe the characteristics of AA in Hispanic/Latinx (H/L) patients, the prevalent majority in California,” they added.
Drawing from chart reviews, ICD codes, and documented physical exams, they retrospectively identified 197 Hispanic/Latinx patients diagnosed with AA at UCI between 2015 and 2022, including alopecia totalis and alopecia universalis.
Nearly two-thirds of patients with alopecia were female (63%), and their mean age at diagnosis was 33 years. Most patients (79%) presented with patchy pattern AA, 13% had diffuse pattern AA, and only 12% had eyebrow, eyelash, or beard involvement. The most common comorbidity in patients overall was atopy (24%), including allergic rhinitis (12%), asthma (10%), and/or atopic dermatitis (7%).
The authors found that 18% of patients had one or more coexisting autoimmune conditions, most commonly rheumatoid arthritis (9%) and thyroid disease (6%). No patients had celiac disease, myasthenia gravis, or inflammatory bowel disease, but 43% had another dermatologic condition.
In other findings, 22% of patients had vitamin D deficiency, 20% had hyperlipidemia, 18% had obesity, 16% had gastroesophageal reflux disease, and 12% had anemia. At the same time, depression, anxiety, or sleep disorders were identified in 14% of patients.
“Interestingly, the most common autoimmune comorbidity in H/L was rheumatoid arthritis, compared to thyroid disease in Black patients and overall AA patients,” the authors wrote. “This finding may be a reflection of a larger trend, as rheumatoid arthritis in the H/L population has been on the rise.”
The authors acknowledged certain limitations of the study including its small sample size and lack of a control group, and reported having no financial disclosures.
.
Those are among key findings from a retrospective analysis of Hispanic/Latinx patients at the University of California, Irvine (UCI) by Natasha Mesinkovska, MD, PhD, of UCI’s department of dermatology, and her coauthors. The findings were published online in the Journal of the American Academy of Dermatology.
A recent study examined the epidemiology of alopecia areata (AA) in Black patients, wrote Dr. Mesinkovska and coauthors Celine Phong, a UCI medical student, and Amy J. McMichael, MD, professor of dermatology at Wake Forest University, Winston-Salem, N.C. “A similar unmet need exists to describe the characteristics of AA in Hispanic/Latinx (H/L) patients, the prevalent majority in California,” they added.
Drawing from chart reviews, ICD codes, and documented physical exams, they retrospectively identified 197 Hispanic/Latinx patients diagnosed with AA at UCI between 2015 and 2022, including alopecia totalis and alopecia universalis.
Nearly two-thirds of patients with alopecia were female (63%), and their mean age at diagnosis was 33 years. Most patients (79%) presented with patchy pattern AA, 13% had diffuse pattern AA, and only 12% had eyebrow, eyelash, or beard involvement. The most common comorbidity in patients overall was atopy (24%), including allergic rhinitis (12%), asthma (10%), and/or atopic dermatitis (7%).
The authors found that 18% of patients had one or more coexisting autoimmune conditions, most commonly rheumatoid arthritis (9%) and thyroid disease (6%). No patients had celiac disease, myasthenia gravis, or inflammatory bowel disease, but 43% had another dermatologic condition.
In other findings, 22% of patients had vitamin D deficiency, 20% had hyperlipidemia, 18% had obesity, 16% had gastroesophageal reflux disease, and 12% had anemia. At the same time, depression, anxiety, or sleep disorders were identified in 14% of patients.
“Interestingly, the most common autoimmune comorbidity in H/L was rheumatoid arthritis, compared to thyroid disease in Black patients and overall AA patients,” the authors wrote. “This finding may be a reflection of a larger trend, as rheumatoid arthritis in the H/L population has been on the rise.”
The authors acknowledged certain limitations of the study including its small sample size and lack of a control group, and reported having no financial disclosures.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Bad breath? Mouthwash is out. Yogurt is in.
Leave the mouthwash. Take the yogurt
Most of us have experienced some sort of bad breath. It’s common in the morning right after waking up, but it also may be a sign for underlying medical issues like dental problems or acid reflux. Wherever it comes from, we always want to get rid of it. A recent meta-analysis in BMJ Open may have found the answer in some common foods.
For those with halitosis, the basic problem is that the bacteria in their mouths are not happy about where they are. The researchers looked at 130 studies and found seven that suggested fermented food has some effect in combating bad breath.
Now when we say fermented food, we’re not talking about that science project waiting to happen in the back of the refrigerator. Think yogurt, sourdough bread, or miso soup. Anything that contains probiotic bacteria.
Matthew J. Messina, DDS, assistant professor of dentistry at Ohio State University, who was not involved with the study, told Healthline that “the whole idea behind probiotics is [bacteria replacement]. Supplant the ‘bad guys’ with the ‘good guys,’ then we’ll end up with a better result.” Essentially balancing the scales in your mouth.
It may not be a long-term solution, Dr. Messina said, but the short-term data are positive. So if you experience bad breath from time to time, try a little bowl of yogurt instead of chewing gum. If nothing else, the bacteria in your mouth will thank you.
You can talk the silly talk, but can you walk the silly walk?
The Ministry of Silly Walks sketch from Monty Python is an enduring comedy classic, and one of surprising relevance for doctors. After all, this isn’t the first time a study has analyzed the unusual strides of Mr. Putey and Mr. Teabag.
The BMJ Christmas edition truly is the gift that keeps on giving. For this plunge into the Flying Circus, the study authors recruited a small group of fairly average adults and had them walk normally around a track for 5 minutes, monitoring their oxygen intake and energy expenditure. After that, the study participants imitated Mr. Putey’s walk and then Mr. Teabag’s.
In the sketch, Mr. Teabag notes that Mr. Putey’s walk is “not particularly silly,” which is borne out in the research. When imitating Mr. Putey’s walk, oxygen intake and energy expenditure were barely higher than a normal walk, not enough to achieve a meaningful difference. Hopefully he’ll get that government grant to further develop his silly walk, because right now Mr. Putey’s walk simply doesn’t cut it.
Mr. Teabag’s walk is a different story and the very image of inefficiency. Oxygen intake was 2.5 times higher than during the normal walk, and energy expenditure was noticeably higher (8 kcal in men and 5.2 kcal in women). In fact, the walk was so inefficient and its effect so drastic it actually reached the level of vigorous exercise. Thanks to this, the study authors noted that just 11 minutes a day of walking like Mr. Teabag would be enough to reach the general goal of 75 minutes of vigorous exercise per week. Boosting that to 12-19 minutes would increase daily energy expenditure by 100 kcal.
The study authors wrote, “Had an initiative to promote inefficient movement been adopted in the early 1970s, we might now be living among a healthier society. Efforts to promote higher energy – and perhaps more joyful – walking should ensure inclusivity and inefficiency for all.” We think they just advocated for a real-life Ministry of Silly Walks. Well, there have been worse ideas. Just look at Twitter.
When efficient gut microbes go bad
With the latest news from the Ministry of Silly Walks, is it time for humans to embrace all things inefficient? Maybe.
Turns out that individuals with more efficient digestive systems – those that extract more energy from the fuel supplied to them by the busy mouths above – tend to gain more weight than those with less efficient guts, even when they eat the same food, according to a recent study published in Microbiome.
The researchers took a look at the composition of gut microbes in a group of 85 volunteers and found that about 40% had microbiomes dominated by Bacteroides bacteria, which are more effective at extracting nutrients from food. That group also weighed 10% more on average, amounting to an extra 9 kg.
In a rather blatant demonstration of efficiency, the investigators also measured the speed of the participants’ digestion, as they had hypothesized that those with the longest digestive travel times would be the ones who harvested the most nutrition from their food. That was not the case.
The study subjects with the most efficient gut bacteria “also have the fastest passage through the gastrointestinal system, which has given us something to think about,” senior author Henrik Roager of the University of Copenhagen said in a written statement.
You know what gives us something to think about? Stool energy density and intestinal transit time and faecal bacterial cell counts, that’s what. Ick. Sometimes science is gross.
Here’s another thought, though: Seeing faecal instead of fecal is kind of funny to our American eyes, but adding that extra letter is also inefficient, which could mean that it’s good. So, in the spirit of embracing the inefficient as a new year begins, we’re resolving to wrap our editorial arms around faecal and the faeces it represents. Well, not literally, of course. More like we’re embracing the spirit of faeces.
Leave the mouthwash. Take the yogurt
Most of us have experienced some sort of bad breath. It’s common in the morning right after waking up, but it also may be a sign for underlying medical issues like dental problems or acid reflux. Wherever it comes from, we always want to get rid of it. A recent meta-analysis in BMJ Open may have found the answer in some common foods.
For those with halitosis, the basic problem is that the bacteria in their mouths are not happy about where they are. The researchers looked at 130 studies and found seven that suggested fermented food has some effect in combating bad breath.
Now when we say fermented food, we’re not talking about that science project waiting to happen in the back of the refrigerator. Think yogurt, sourdough bread, or miso soup. Anything that contains probiotic bacteria.
Matthew J. Messina, DDS, assistant professor of dentistry at Ohio State University, who was not involved with the study, told Healthline that “the whole idea behind probiotics is [bacteria replacement]. Supplant the ‘bad guys’ with the ‘good guys,’ then we’ll end up with a better result.” Essentially balancing the scales in your mouth.
It may not be a long-term solution, Dr. Messina said, but the short-term data are positive. So if you experience bad breath from time to time, try a little bowl of yogurt instead of chewing gum. If nothing else, the bacteria in your mouth will thank you.
You can talk the silly talk, but can you walk the silly walk?
The Ministry of Silly Walks sketch from Monty Python is an enduring comedy classic, and one of surprising relevance for doctors. After all, this isn’t the first time a study has analyzed the unusual strides of Mr. Putey and Mr. Teabag.
The BMJ Christmas edition truly is the gift that keeps on giving. For this plunge into the Flying Circus, the study authors recruited a small group of fairly average adults and had them walk normally around a track for 5 minutes, monitoring their oxygen intake and energy expenditure. After that, the study participants imitated Mr. Putey’s walk and then Mr. Teabag’s.
In the sketch, Mr. Teabag notes that Mr. Putey’s walk is “not particularly silly,” which is borne out in the research. When imitating Mr. Putey’s walk, oxygen intake and energy expenditure were barely higher than a normal walk, not enough to achieve a meaningful difference. Hopefully he’ll get that government grant to further develop his silly walk, because right now Mr. Putey’s walk simply doesn’t cut it.
Mr. Teabag’s walk is a different story and the very image of inefficiency. Oxygen intake was 2.5 times higher than during the normal walk, and energy expenditure was noticeably higher (8 kcal in men and 5.2 kcal in women). In fact, the walk was so inefficient and its effect so drastic it actually reached the level of vigorous exercise. Thanks to this, the study authors noted that just 11 minutes a day of walking like Mr. Teabag would be enough to reach the general goal of 75 minutes of vigorous exercise per week. Boosting that to 12-19 minutes would increase daily energy expenditure by 100 kcal.
The study authors wrote, “Had an initiative to promote inefficient movement been adopted in the early 1970s, we might now be living among a healthier society. Efforts to promote higher energy – and perhaps more joyful – walking should ensure inclusivity and inefficiency for all.” We think they just advocated for a real-life Ministry of Silly Walks. Well, there have been worse ideas. Just look at Twitter.
When efficient gut microbes go bad
With the latest news from the Ministry of Silly Walks, is it time for humans to embrace all things inefficient? Maybe.
Turns out that individuals with more efficient digestive systems – those that extract more energy from the fuel supplied to them by the busy mouths above – tend to gain more weight than those with less efficient guts, even when they eat the same food, according to a recent study published in Microbiome.
The researchers took a look at the composition of gut microbes in a group of 85 volunteers and found that about 40% had microbiomes dominated by Bacteroides bacteria, which are more effective at extracting nutrients from food. That group also weighed 10% more on average, amounting to an extra 9 kg.
In a rather blatant demonstration of efficiency, the investigators also measured the speed of the participants’ digestion, as they had hypothesized that those with the longest digestive travel times would be the ones who harvested the most nutrition from their food. That was not the case.
The study subjects with the most efficient gut bacteria “also have the fastest passage through the gastrointestinal system, which has given us something to think about,” senior author Henrik Roager of the University of Copenhagen said in a written statement.
You know what gives us something to think about? Stool energy density and intestinal transit time and faecal bacterial cell counts, that’s what. Ick. Sometimes science is gross.
Here’s another thought, though: Seeing faecal instead of fecal is kind of funny to our American eyes, but adding that extra letter is also inefficient, which could mean that it’s good. So, in the spirit of embracing the inefficient as a new year begins, we’re resolving to wrap our editorial arms around faecal and the faeces it represents. Well, not literally, of course. More like we’re embracing the spirit of faeces.
Leave the mouthwash. Take the yogurt
Most of us have experienced some sort of bad breath. It’s common in the morning right after waking up, but it also may be a sign for underlying medical issues like dental problems or acid reflux. Wherever it comes from, we always want to get rid of it. A recent meta-analysis in BMJ Open may have found the answer in some common foods.
For those with halitosis, the basic problem is that the bacteria in their mouths are not happy about where they are. The researchers looked at 130 studies and found seven that suggested fermented food has some effect in combating bad breath.
Now when we say fermented food, we’re not talking about that science project waiting to happen in the back of the refrigerator. Think yogurt, sourdough bread, or miso soup. Anything that contains probiotic bacteria.
Matthew J. Messina, DDS, assistant professor of dentistry at Ohio State University, who was not involved with the study, told Healthline that “the whole idea behind probiotics is [bacteria replacement]. Supplant the ‘bad guys’ with the ‘good guys,’ then we’ll end up with a better result.” Essentially balancing the scales in your mouth.
It may not be a long-term solution, Dr. Messina said, but the short-term data are positive. So if you experience bad breath from time to time, try a little bowl of yogurt instead of chewing gum. If nothing else, the bacteria in your mouth will thank you.
You can talk the silly talk, but can you walk the silly walk?
The Ministry of Silly Walks sketch from Monty Python is an enduring comedy classic, and one of surprising relevance for doctors. After all, this isn’t the first time a study has analyzed the unusual strides of Mr. Putey and Mr. Teabag.
The BMJ Christmas edition truly is the gift that keeps on giving. For this plunge into the Flying Circus, the study authors recruited a small group of fairly average adults and had them walk normally around a track for 5 minutes, monitoring their oxygen intake and energy expenditure. After that, the study participants imitated Mr. Putey’s walk and then Mr. Teabag’s.
In the sketch, Mr. Teabag notes that Mr. Putey’s walk is “not particularly silly,” which is borne out in the research. When imitating Mr. Putey’s walk, oxygen intake and energy expenditure were barely higher than a normal walk, not enough to achieve a meaningful difference. Hopefully he’ll get that government grant to further develop his silly walk, because right now Mr. Putey’s walk simply doesn’t cut it.
Mr. Teabag’s walk is a different story and the very image of inefficiency. Oxygen intake was 2.5 times higher than during the normal walk, and energy expenditure was noticeably higher (8 kcal in men and 5.2 kcal in women). In fact, the walk was so inefficient and its effect so drastic it actually reached the level of vigorous exercise. Thanks to this, the study authors noted that just 11 minutes a day of walking like Mr. Teabag would be enough to reach the general goal of 75 minutes of vigorous exercise per week. Boosting that to 12-19 minutes would increase daily energy expenditure by 100 kcal.
The study authors wrote, “Had an initiative to promote inefficient movement been adopted in the early 1970s, we might now be living among a healthier society. Efforts to promote higher energy – and perhaps more joyful – walking should ensure inclusivity and inefficiency for all.” We think they just advocated for a real-life Ministry of Silly Walks. Well, there have been worse ideas. Just look at Twitter.
When efficient gut microbes go bad
With the latest news from the Ministry of Silly Walks, is it time for humans to embrace all things inefficient? Maybe.
Turns out that individuals with more efficient digestive systems – those that extract more energy from the fuel supplied to them by the busy mouths above – tend to gain more weight than those with less efficient guts, even when they eat the same food, according to a recent study published in Microbiome.
The researchers took a look at the composition of gut microbes in a group of 85 volunteers and found that about 40% had microbiomes dominated by Bacteroides bacteria, which are more effective at extracting nutrients from food. That group also weighed 10% more on average, amounting to an extra 9 kg.
In a rather blatant demonstration of efficiency, the investigators also measured the speed of the participants’ digestion, as they had hypothesized that those with the longest digestive travel times would be the ones who harvested the most nutrition from their food. That was not the case.
The study subjects with the most efficient gut bacteria “also have the fastest passage through the gastrointestinal system, which has given us something to think about,” senior author Henrik Roager of the University of Copenhagen said in a written statement.
You know what gives us something to think about? Stool energy density and intestinal transit time and faecal bacterial cell counts, that’s what. Ick. Sometimes science is gross.
Here’s another thought, though: Seeing faecal instead of fecal is kind of funny to our American eyes, but adding that extra letter is also inefficient, which could mean that it’s good. So, in the spirit of embracing the inefficient as a new year begins, we’re resolving to wrap our editorial arms around faecal and the faeces it represents. Well, not literally, of course. More like we’re embracing the spirit of faeces.