User login
Cardiology News is an independent news source that provides cardiologists with timely and relevant news and commentary about clinical developments and the impact of health care policy on cardiology and the cardiologist's practice. Cardiology News Digital Network is the online destination and multimedia properties of Cardiology News, the independent news publication for cardiologists. Cardiology news is the leading source of news and commentary about clinical developments in cardiology as well as health care policy and regulations that affect the cardiologist's practice. Cardiology News Digital Network is owned by Frontline Medical Communications.
FDA expands list of Getinge IABP system and component shortages
The U.S. Food and Drug Administration issued a letter to health care providers describing a current shortage of Getinge intra-aortic balloon pump (IABP) catheters and other components.
Earlier, the agency announced shortages of the company’s Maquet/Datascope IAB catheters, new Cardiosave IABP devices, and Cardiosave IABP parts. The new notification adds Getinge Maquet/Datascope IABP systems to the list.
The company’s letter explains that “ongoing supply chain issues have significantly impacted our ability to build intra-aortic balloon pumps, intra-aortic balloon catheters, and spare parts due to raw material shortages.”
It also offers guidance on maintaining Cardiosave Safety Disks and lithium-ion batteries in the face of the shortages. “In the event that you need a replacement pump while your IABP is undergoing service, please contact your local sales representative who may be able to assist with a temporary IABP.”
Providers are instructed to inform the company through its sales representatives “if you have any underutilized Maquet/Datascope IAB catheters or IABPs and are willing to share them with hospitals in need.”
The shortages are expected to continue into 2023, the FDA states in its letter.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration issued a letter to health care providers describing a current shortage of Getinge intra-aortic balloon pump (IABP) catheters and other components.
Earlier, the agency announced shortages of the company’s Maquet/Datascope IAB catheters, new Cardiosave IABP devices, and Cardiosave IABP parts. The new notification adds Getinge Maquet/Datascope IABP systems to the list.
The company’s letter explains that “ongoing supply chain issues have significantly impacted our ability to build intra-aortic balloon pumps, intra-aortic balloon catheters, and spare parts due to raw material shortages.”
It also offers guidance on maintaining Cardiosave Safety Disks and lithium-ion batteries in the face of the shortages. “In the event that you need a replacement pump while your IABP is undergoing service, please contact your local sales representative who may be able to assist with a temporary IABP.”
Providers are instructed to inform the company through its sales representatives “if you have any underutilized Maquet/Datascope IAB catheters or IABPs and are willing to share them with hospitals in need.”
The shortages are expected to continue into 2023, the FDA states in its letter.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration issued a letter to health care providers describing a current shortage of Getinge intra-aortic balloon pump (IABP) catheters and other components.
Earlier, the agency announced shortages of the company’s Maquet/Datascope IAB catheters, new Cardiosave IABP devices, and Cardiosave IABP parts. The new notification adds Getinge Maquet/Datascope IABP systems to the list.
The company’s letter explains that “ongoing supply chain issues have significantly impacted our ability to build intra-aortic balloon pumps, intra-aortic balloon catheters, and spare parts due to raw material shortages.”
It also offers guidance on maintaining Cardiosave Safety Disks and lithium-ion batteries in the face of the shortages. “In the event that you need a replacement pump while your IABP is undergoing service, please contact your local sales representative who may be able to assist with a temporary IABP.”
Providers are instructed to inform the company through its sales representatives “if you have any underutilized Maquet/Datascope IAB catheters or IABPs and are willing to share them with hospitals in need.”
The shortages are expected to continue into 2023, the FDA states in its letter.
A version of this article first appeared on Medscape.com.
Employers use patient assistance programs to offset their own costs
Anna Sutton was shocked when she received a letter from her husband’s job-based health plan stating that Humira, an expensive drug used to treat her daughter’s juvenile arthritis, was now on a long list of medications considered “nonessential benefits.”
The July 2021 letter said the family could either participate in a new effort overseen by a company called SaveOnSP and get the drug free of charge or be saddled with a monthly copayment that could top $1,000.
“It really gave us no choice,” said Mrs. Sutton, of Woodinville, Wash. She added that “every single [Food and Drug Administration]–approved medication for juvenile arthritis” was on the list of nonessential benefits.
Mrs. Sutton had unwittingly become part of a strategy that employers are using to deal with the high cost of drugs prescribed to treat conditions such as arthritis, psoriasis, cancer, and hemophilia.
Those employers are tapping into dollars provided through programs they have previously criticized: patient financial assistance initiatives set up by drugmakers, which some benefit managers have complained encourage patients to stay on expensive brand-name drugs when less expensive options might be available.
Now, though, employers, or the vendors and insurers they hire specifically to oversee such efforts, are seeking that money to offset their own costs. Drugmakers object, saying the money was intended primarily for patients. But some benefit brokers and companies like SaveOnSP say they can help trim employers’ spending on insurance – which, they say, could be the difference between an employer offering coverage to workers or not.
It’s the latest twist in a long-running dispute between the drug industry and insurers over which group is more to blame for rising costs to patients. And patients are, again, caught in the middle.
Patient advocates say the term “nonessential” stresses patients out even though it doesn’t mean the drugs – often called “specialty” drugs because of their high prices or the way they are made – are unnecessary.
Some advocates fear the new strategies could be “a way to weed out those with costly health care needs,” said Rachel Klein, deputy executive director of the AIDS Institute, a nonprofit advocacy group. Workers who rely on the drugs may feel pressured to change insurers or jobs.
Two versions of the new strategy are in play. Both are used mainly by self-insured employers that hire vendors, like SaveOnSP, which then work with the employers’ pharmacy benefit managers, such as Express Scripts/Cigna, to implement the strategy. There are also smaller vendors, like SHARx and Payer Matrix, some of which work directly with employers.
In one approach, insurers or employers continue to cover the drugs but designate them as “nonessential,” which allows the health plans to bypass annual limits set by the Affordable Care Act on how much patients can pay in out-of-pocket costs for drugs. The employer or hired vendor then raises the copay required of the worker, often sharply, but offers to substantially cut or eliminate that copay if the patient participates in the new effort. Workers who agree enroll in drugmaker financial assistance programs meant to cover the drug copays, and the vendor monitoring the effort aims to capture the maximum amount the drugmaker provides annually, according to a lawsuit filed in May by drugmaker Johnson & Johnson against SaveOnSP, which is based in Elma, N.Y.
The employer must still cover part of the cost of the drug, but the amount is reduced by the amount of copay assistance that is accessed. That assistance can vary widely and be as much as $20,000 a year for some drugs.
In the other approach, employers don’t bother naming drugs nonessential; they simply drop coverage for specific drugs or classes of drugs. Then, the outside vendor helps patients provide the financial and other information needed to apply for free medication from drugmakers through charity programs intended for uninsured patients.
“We’re seeing it in every state at this point,” said Becky Burns, chief operating officer and chief financial officer at the Bleeding and Clotting Disorders Institute in Peoria, Ill., a federally funded hemophilia treatment center.
The strategies are mostly being used in self-insured employer health plans, which are governed by federal laws that give broad flexibility to employers in designing health benefits.
Still, some patient advocates say these programs can lead to delays for patients in accessing medications while applications are processed – and sometimes unexpected bills for consumers.
“We have patients get billed after they max out their assistance,” said Kollet Koulianos, vice president of payer relations at the National Hemophilia Foundation. Once she gets involved, vendors often claim the bills were sent in error.
Even though only about 2% of the workforce needs the drugs, which can cost thousands of dollars a dose, they can lead to a hefty financial liability for self-insured employers, said Drew Mann, a benefits consultant in Knoxville, Tenn., whose clientele includes employers that use variations of these programs.
Before employer health plans took advantage of such assistance, patients often signed up for these programs on their own, receiving coupons that covered their share of the drug’s cost. In that circumstance, drugmakers often paid less than they do under the new employer schemes because a patient’s out-of-pocket costs were capped at lower amounts.
Brokers and the CEOs of firms offering the new programs say that in most cases patients continue to get their drugs, often with little or no out-of-pocket costs.
If workers do not qualify for charity because their income is too high, or for another reason, the employer might make an exception and pay the claim or look for an alternative solution, Mr. Mann said. Patient groups noted that some specialty drugs may not have any alternatives.
How this practice will play out in the long run remains uncertain. Drugmakers offer both copay assistance and charity care in part because they know many patients, even those with insurance, cannot afford their products. The programs are also good public relations and a tax write-off. But the new emphasis by some employers on maximizing the amount they or their insurers can collect from the programs could cause some drugmakers to take issue with the new strategies or even reconsider their programs.
“Even though our client, like most manufacturers, provides billions in discounts and rebates to health insurers as part of their negotiations, the insurers also want this additional pool of funds, which is meant to help people who can’t meet the copay,” said Harry Sandick, a lawyer representing J&J.
J&J’s lawsuit, filed in U.S. District Court in New Jersey, alleges that patients are “coerced” into participating in copay assistance programs after their drugs are deemed “nonessential” and therefore are “no longer subject to the ACA’s annual out-of-pocket maximum.”
Once patients enroll, the money from the drugmaker goes to the insurer or employer plan, with SaveOnSP retaining 25%, according to the lawsuit. It claims J&J has lost $100 million to these efforts.
None of that money counts toward patients’ deductibles or out-of-pocket maximums for the year.
In addition to the lawsuit over the copay assistance program efforts, there has been other reaction to the new employer strategies. In an October letter to physicians, the Johnson & Johnson Patient Assistance Foundation, a separate entity, said it will no longer offer free medications to patients with insurance starting in January, citing the rise of such “alternative funding programs.”
Still, J&J spokesperson L.D. Platt said the drugmaker has plans, also in January, to roll out other assistance to patients who may be “underinsured” so they won’t be affected by the foundation’s decision.
In a statement, SaveOnSP said that employers object to drug companies’ “using their employees’ ongoing need for these drugs as an excuse to keep hiking the drugs’ prices” and that the firm simply “advises these employers on how to fight back against rising prices while getting employees the drugs they need at no cost to the employees.”
In a court filing, SaveOnSP said drugmakers have another option if they don’t like efforts by insurers and employers to max out what they can get from the programs: reduce the amount of assistance available. J&J, the filing said, did just that when it recently cut its allotted amount of copay assistance for psoriasis drugs Stelara and Tremfya from $20,000 to $6,000 per participant annually. The filing noted that SaveOnSP participants would still have no copay for those drugs.
For Mrs. Sutton’s part, her family did participate in the program offered through her husband’s work-based insurance plan, agreeing to have SaveOnSP monitor their enrollment and payments from the drugmaker.
So far, her 15-year-old daughter has continued to get Humira, and she has not been billed a copay.
Even so, “the whole process seems kind of slimy to me,” she said. “The patients are caught in the middle between the drug industry and the insurance industry, each trying to get as much money as possible out of the other.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Anna Sutton was shocked when she received a letter from her husband’s job-based health plan stating that Humira, an expensive drug used to treat her daughter’s juvenile arthritis, was now on a long list of medications considered “nonessential benefits.”
The July 2021 letter said the family could either participate in a new effort overseen by a company called SaveOnSP and get the drug free of charge or be saddled with a monthly copayment that could top $1,000.
“It really gave us no choice,” said Mrs. Sutton, of Woodinville, Wash. She added that “every single [Food and Drug Administration]–approved medication for juvenile arthritis” was on the list of nonessential benefits.
Mrs. Sutton had unwittingly become part of a strategy that employers are using to deal with the high cost of drugs prescribed to treat conditions such as arthritis, psoriasis, cancer, and hemophilia.
Those employers are tapping into dollars provided through programs they have previously criticized: patient financial assistance initiatives set up by drugmakers, which some benefit managers have complained encourage patients to stay on expensive brand-name drugs when less expensive options might be available.
Now, though, employers, or the vendors and insurers they hire specifically to oversee such efforts, are seeking that money to offset their own costs. Drugmakers object, saying the money was intended primarily for patients. But some benefit brokers and companies like SaveOnSP say they can help trim employers’ spending on insurance – which, they say, could be the difference between an employer offering coverage to workers or not.
It’s the latest twist in a long-running dispute between the drug industry and insurers over which group is more to blame for rising costs to patients. And patients are, again, caught in the middle.
Patient advocates say the term “nonessential” stresses patients out even though it doesn’t mean the drugs – often called “specialty” drugs because of their high prices or the way they are made – are unnecessary.
Some advocates fear the new strategies could be “a way to weed out those with costly health care needs,” said Rachel Klein, deputy executive director of the AIDS Institute, a nonprofit advocacy group. Workers who rely on the drugs may feel pressured to change insurers or jobs.
Two versions of the new strategy are in play. Both are used mainly by self-insured employers that hire vendors, like SaveOnSP, which then work with the employers’ pharmacy benefit managers, such as Express Scripts/Cigna, to implement the strategy. There are also smaller vendors, like SHARx and Payer Matrix, some of which work directly with employers.
In one approach, insurers or employers continue to cover the drugs but designate them as “nonessential,” which allows the health plans to bypass annual limits set by the Affordable Care Act on how much patients can pay in out-of-pocket costs for drugs. The employer or hired vendor then raises the copay required of the worker, often sharply, but offers to substantially cut or eliminate that copay if the patient participates in the new effort. Workers who agree enroll in drugmaker financial assistance programs meant to cover the drug copays, and the vendor monitoring the effort aims to capture the maximum amount the drugmaker provides annually, according to a lawsuit filed in May by drugmaker Johnson & Johnson against SaveOnSP, which is based in Elma, N.Y.
The employer must still cover part of the cost of the drug, but the amount is reduced by the amount of copay assistance that is accessed. That assistance can vary widely and be as much as $20,000 a year for some drugs.
In the other approach, employers don’t bother naming drugs nonessential; they simply drop coverage for specific drugs or classes of drugs. Then, the outside vendor helps patients provide the financial and other information needed to apply for free medication from drugmakers through charity programs intended for uninsured patients.
“We’re seeing it in every state at this point,” said Becky Burns, chief operating officer and chief financial officer at the Bleeding and Clotting Disorders Institute in Peoria, Ill., a federally funded hemophilia treatment center.
The strategies are mostly being used in self-insured employer health plans, which are governed by federal laws that give broad flexibility to employers in designing health benefits.
Still, some patient advocates say these programs can lead to delays for patients in accessing medications while applications are processed – and sometimes unexpected bills for consumers.
“We have patients get billed after they max out their assistance,” said Kollet Koulianos, vice president of payer relations at the National Hemophilia Foundation. Once she gets involved, vendors often claim the bills were sent in error.
Even though only about 2% of the workforce needs the drugs, which can cost thousands of dollars a dose, they can lead to a hefty financial liability for self-insured employers, said Drew Mann, a benefits consultant in Knoxville, Tenn., whose clientele includes employers that use variations of these programs.
Before employer health plans took advantage of such assistance, patients often signed up for these programs on their own, receiving coupons that covered their share of the drug’s cost. In that circumstance, drugmakers often paid less than they do under the new employer schemes because a patient’s out-of-pocket costs were capped at lower amounts.
Brokers and the CEOs of firms offering the new programs say that in most cases patients continue to get their drugs, often with little or no out-of-pocket costs.
If workers do not qualify for charity because their income is too high, or for another reason, the employer might make an exception and pay the claim or look for an alternative solution, Mr. Mann said. Patient groups noted that some specialty drugs may not have any alternatives.
How this practice will play out in the long run remains uncertain. Drugmakers offer both copay assistance and charity care in part because they know many patients, even those with insurance, cannot afford their products. The programs are also good public relations and a tax write-off. But the new emphasis by some employers on maximizing the amount they or their insurers can collect from the programs could cause some drugmakers to take issue with the new strategies or even reconsider their programs.
“Even though our client, like most manufacturers, provides billions in discounts and rebates to health insurers as part of their negotiations, the insurers also want this additional pool of funds, which is meant to help people who can’t meet the copay,” said Harry Sandick, a lawyer representing J&J.
J&J’s lawsuit, filed in U.S. District Court in New Jersey, alleges that patients are “coerced” into participating in copay assistance programs after their drugs are deemed “nonessential” and therefore are “no longer subject to the ACA’s annual out-of-pocket maximum.”
Once patients enroll, the money from the drugmaker goes to the insurer or employer plan, with SaveOnSP retaining 25%, according to the lawsuit. It claims J&J has lost $100 million to these efforts.
None of that money counts toward patients’ deductibles or out-of-pocket maximums for the year.
In addition to the lawsuit over the copay assistance program efforts, there has been other reaction to the new employer strategies. In an October letter to physicians, the Johnson & Johnson Patient Assistance Foundation, a separate entity, said it will no longer offer free medications to patients with insurance starting in January, citing the rise of such “alternative funding programs.”
Still, J&J spokesperson L.D. Platt said the drugmaker has plans, also in January, to roll out other assistance to patients who may be “underinsured” so they won’t be affected by the foundation’s decision.
In a statement, SaveOnSP said that employers object to drug companies’ “using their employees’ ongoing need for these drugs as an excuse to keep hiking the drugs’ prices” and that the firm simply “advises these employers on how to fight back against rising prices while getting employees the drugs they need at no cost to the employees.”
In a court filing, SaveOnSP said drugmakers have another option if they don’t like efforts by insurers and employers to max out what they can get from the programs: reduce the amount of assistance available. J&J, the filing said, did just that when it recently cut its allotted amount of copay assistance for psoriasis drugs Stelara and Tremfya from $20,000 to $6,000 per participant annually. The filing noted that SaveOnSP participants would still have no copay for those drugs.
For Mrs. Sutton’s part, her family did participate in the program offered through her husband’s work-based insurance plan, agreeing to have SaveOnSP monitor their enrollment and payments from the drugmaker.
So far, her 15-year-old daughter has continued to get Humira, and she has not been billed a copay.
Even so, “the whole process seems kind of slimy to me,” she said. “The patients are caught in the middle between the drug industry and the insurance industry, each trying to get as much money as possible out of the other.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Anna Sutton was shocked when she received a letter from her husband’s job-based health plan stating that Humira, an expensive drug used to treat her daughter’s juvenile arthritis, was now on a long list of medications considered “nonessential benefits.”
The July 2021 letter said the family could either participate in a new effort overseen by a company called SaveOnSP and get the drug free of charge or be saddled with a monthly copayment that could top $1,000.
“It really gave us no choice,” said Mrs. Sutton, of Woodinville, Wash. She added that “every single [Food and Drug Administration]–approved medication for juvenile arthritis” was on the list of nonessential benefits.
Mrs. Sutton had unwittingly become part of a strategy that employers are using to deal with the high cost of drugs prescribed to treat conditions such as arthritis, psoriasis, cancer, and hemophilia.
Those employers are tapping into dollars provided through programs they have previously criticized: patient financial assistance initiatives set up by drugmakers, which some benefit managers have complained encourage patients to stay on expensive brand-name drugs when less expensive options might be available.
Now, though, employers, or the vendors and insurers they hire specifically to oversee such efforts, are seeking that money to offset their own costs. Drugmakers object, saying the money was intended primarily for patients. But some benefit brokers and companies like SaveOnSP say they can help trim employers’ spending on insurance – which, they say, could be the difference between an employer offering coverage to workers or not.
It’s the latest twist in a long-running dispute between the drug industry and insurers over which group is more to blame for rising costs to patients. And patients are, again, caught in the middle.
Patient advocates say the term “nonessential” stresses patients out even though it doesn’t mean the drugs – often called “specialty” drugs because of their high prices or the way they are made – are unnecessary.
Some advocates fear the new strategies could be “a way to weed out those with costly health care needs,” said Rachel Klein, deputy executive director of the AIDS Institute, a nonprofit advocacy group. Workers who rely on the drugs may feel pressured to change insurers or jobs.
Two versions of the new strategy are in play. Both are used mainly by self-insured employers that hire vendors, like SaveOnSP, which then work with the employers’ pharmacy benefit managers, such as Express Scripts/Cigna, to implement the strategy. There are also smaller vendors, like SHARx and Payer Matrix, some of which work directly with employers.
In one approach, insurers or employers continue to cover the drugs but designate them as “nonessential,” which allows the health plans to bypass annual limits set by the Affordable Care Act on how much patients can pay in out-of-pocket costs for drugs. The employer or hired vendor then raises the copay required of the worker, often sharply, but offers to substantially cut or eliminate that copay if the patient participates in the new effort. Workers who agree enroll in drugmaker financial assistance programs meant to cover the drug copays, and the vendor monitoring the effort aims to capture the maximum amount the drugmaker provides annually, according to a lawsuit filed in May by drugmaker Johnson & Johnson against SaveOnSP, which is based in Elma, N.Y.
The employer must still cover part of the cost of the drug, but the amount is reduced by the amount of copay assistance that is accessed. That assistance can vary widely and be as much as $20,000 a year for some drugs.
In the other approach, employers don’t bother naming drugs nonessential; they simply drop coverage for specific drugs or classes of drugs. Then, the outside vendor helps patients provide the financial and other information needed to apply for free medication from drugmakers through charity programs intended for uninsured patients.
“We’re seeing it in every state at this point,” said Becky Burns, chief operating officer and chief financial officer at the Bleeding and Clotting Disorders Institute in Peoria, Ill., a federally funded hemophilia treatment center.
The strategies are mostly being used in self-insured employer health plans, which are governed by federal laws that give broad flexibility to employers in designing health benefits.
Still, some patient advocates say these programs can lead to delays for patients in accessing medications while applications are processed – and sometimes unexpected bills for consumers.
“We have patients get billed after they max out their assistance,” said Kollet Koulianos, vice president of payer relations at the National Hemophilia Foundation. Once she gets involved, vendors often claim the bills were sent in error.
Even though only about 2% of the workforce needs the drugs, which can cost thousands of dollars a dose, they can lead to a hefty financial liability for self-insured employers, said Drew Mann, a benefits consultant in Knoxville, Tenn., whose clientele includes employers that use variations of these programs.
Before employer health plans took advantage of such assistance, patients often signed up for these programs on their own, receiving coupons that covered their share of the drug’s cost. In that circumstance, drugmakers often paid less than they do under the new employer schemes because a patient’s out-of-pocket costs were capped at lower amounts.
Brokers and the CEOs of firms offering the new programs say that in most cases patients continue to get their drugs, often with little or no out-of-pocket costs.
If workers do not qualify for charity because their income is too high, or for another reason, the employer might make an exception and pay the claim or look for an alternative solution, Mr. Mann said. Patient groups noted that some specialty drugs may not have any alternatives.
How this practice will play out in the long run remains uncertain. Drugmakers offer both copay assistance and charity care in part because they know many patients, even those with insurance, cannot afford their products. The programs are also good public relations and a tax write-off. But the new emphasis by some employers on maximizing the amount they or their insurers can collect from the programs could cause some drugmakers to take issue with the new strategies or even reconsider their programs.
“Even though our client, like most manufacturers, provides billions in discounts and rebates to health insurers as part of their negotiations, the insurers also want this additional pool of funds, which is meant to help people who can’t meet the copay,” said Harry Sandick, a lawyer representing J&J.
J&J’s lawsuit, filed in U.S. District Court in New Jersey, alleges that patients are “coerced” into participating in copay assistance programs after their drugs are deemed “nonessential” and therefore are “no longer subject to the ACA’s annual out-of-pocket maximum.”
Once patients enroll, the money from the drugmaker goes to the insurer or employer plan, with SaveOnSP retaining 25%, according to the lawsuit. It claims J&J has lost $100 million to these efforts.
None of that money counts toward patients’ deductibles or out-of-pocket maximums for the year.
In addition to the lawsuit over the copay assistance program efforts, there has been other reaction to the new employer strategies. In an October letter to physicians, the Johnson & Johnson Patient Assistance Foundation, a separate entity, said it will no longer offer free medications to patients with insurance starting in January, citing the rise of such “alternative funding programs.”
Still, J&J spokesperson L.D. Platt said the drugmaker has plans, also in January, to roll out other assistance to patients who may be “underinsured” so they won’t be affected by the foundation’s decision.
In a statement, SaveOnSP said that employers object to drug companies’ “using their employees’ ongoing need for these drugs as an excuse to keep hiking the drugs’ prices” and that the firm simply “advises these employers on how to fight back against rising prices while getting employees the drugs they need at no cost to the employees.”
In a court filing, SaveOnSP said drugmakers have another option if they don’t like efforts by insurers and employers to max out what they can get from the programs: reduce the amount of assistance available. J&J, the filing said, did just that when it recently cut its allotted amount of copay assistance for psoriasis drugs Stelara and Tremfya from $20,000 to $6,000 per participant annually. The filing noted that SaveOnSP participants would still have no copay for those drugs.
For Mrs. Sutton’s part, her family did participate in the program offered through her husband’s work-based insurance plan, agreeing to have SaveOnSP monitor their enrollment and payments from the drugmaker.
So far, her 15-year-old daughter has continued to get Humira, and she has not been billed a copay.
Even so, “the whole process seems kind of slimy to me,” she said. “The patients are caught in the middle between the drug industry and the insurance industry, each trying to get as much money as possible out of the other.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Diabetes decision tool yields ‘modest’ benefit in low-resource clinics
a randomized trial in China showed.
The tool required clinicians to enter patient data into a computer in order to generate individualized treatment recommendations, adding to their administrative burdens. It also couldn’t tackle patients’ problems with access and affordability of medications.
Nevertheless, the model could curtail physician burnout and improve the quality of care in primary care clinics with limited resources, the researchers said in a paper published in the Annals of Internal Medicine.
They concluded that the findings support “widespread adoption” of the model in China and other low- or middle-income countries where diabetes is on the rise.
Co–principal investigator Jiang He, MD, PhD, chair of epidemiology at Tulane University, New Orleans, said the findings could apply to federally qualified health care (FQHC) clinics that treat underserved patients in the United States.
“At many FQHC clinics, nurse practitioners have to take care of patients with multiple chronic disease conditions. Team-based care with a computerized clinical decision support system will help them and improve patient care,” Dr. He said.
Small improvements
To conduct the trial, called Diabetes Complication Control in Community Clinics (D4C), Dr. He and colleagues randomly assigned 19 out of the 38 community health centers in Xiamen, China, to have a clinical decision support tool installed on the computers of primary care physicians and health coaches.
Starting in October 2016 the researchers recruited 11,132 patients aged 50 and older with uncontrolled diabetes and at least one comorbid condition, with 5,475 patients receiving team-based care with the CDSS and the remainder receiving team-based care alone.
The CDSS generated individualized risk factor summaries and treatment recommendations, including prescriptions based on Chinese and U.S. clinical guidelines. It incorporated data on patients’ insurance plans and local availability of drugs.
At all centers, primary care physicians received training in managing glycemia, blood pressure, and lipids. Nurses were certified as health coaches after receiving training on nutrition, lifestyle changes, and medication adherence. Patients met with their coaches for half an hour every 3 months, and diabetes specialists visited each clinic monthly for team meetings and consultations.
After 18 months, patients undergoing team-based care alone lowered their hemoglobin A1c by 0.6 percentage points (95% confidence interval, –0.7 to –0.5 percentage points), LDL cholesterol by 12.5 mg/dL (95% CI, –13.6 to –11.3 mg/dL), and systolic blood pressure by 7.5 mm Hg (95% CI, –8.4 to –6.6 mm Hg).
The group whose care teams used the CDSS further reduced A1c by 0.2 percentage points (95% CI, –0.3 to –0.1 percentage points), LDL cholesterol by 6.5 mg/dL (95% CI, –8.3 to -4.6 mg/dL), and blood pressure by 1.5 mm Hg (95% CI, –2.8 to –0.3 mm Hg).
All-cause mortality did not differ between the groups. Serious adverse events occurred in 9.1% of the CDSS group, compared with 10.9% of the group whose care team did not use the CDSS.
Addressing social needs
Experts who were not involved in the trial said the marginal impact of the CDSS was no surprise given the mixed results of such tools in previous studies.
However, the lackluster result “might be a shock to people investing a lot in clinical decision support,” said Elbert Huang, MD, MPH, director of the Center for Chronic Disease Research and Policy at the University of Chicago.
Anne Peters, MD, a professor of medicine at the University of Southern California, Los Angeles, said the administrative burden of entering each patient’s data into the system would slow down care and frustrate clinicians. “The system has to be smarter than this.”
On the other hand, the findings of the D4C trial align with other research showing that team-based care strategies are effective for diabetes management.
Dr. Huang noted that there is a “well-established history” of diabetes quality improvement programs, health coaches, buddy programs, and community health worker programs. He added that the new findings “might help to remind everyone of the importance of these programs, which are not always well supported.”
“The bottom line of the paper might be that investing in patient engagement programs might get us 90% of the way to our goal of improving diabetes care,” Dr. Huang said.
Still, Dr. Peters said the portion of patients in the trial who benefited from team-based care seemed “disturbingly low.” Just 16.9% of patients who received team-based care and CDSS and 13% of those who received team-based care alone improved in all three measures. “This system doesn’t get you to where you want to be by a long shot.”
She added that a team-based approach, particularly the use of health coaches, would be a “huge improvement” over fragmented care provided in much of the U.S. safety-net system.
Another team approach
Many systems are striving to improve diabetes management in response to payment incentives, Dr. Huang said.
In a separate retrospective analysis, published in Annals of Family Medicine, researchers at the Mayo Clinic, Rochester, Minn., reported quality improvement gains among primary care practices that adopted a team-based model called Enhanced Primary Care Diabetes (EPCD). The model deployed a range of strategies, such as empowering nurses to engage with patients outside of scheduled office visits and including pharmacists on care teams.
Mayo’s approach did not specifically target underserved populations. Rather, researchers evaluated the model’s impact on about 17,000 patients treated at 32 Mayo internal medicine and family medicine practices of varying sizes, resources, and community settings.
Among staff clinician practices using the EPCD model improved patients’ scores on a composite quality measure called D5, which incorporates glycemic control, blood pressure control, low-density lipoprotein control, tobacco abstinence, and aspirin use.
Following implementation, the portion of patients in those practices meeting the D5 indicator increased from 42.9% to 45.0% (incident rate ratio, 1.005; P = .001).
Meanwhile, the portion of patients meeting the indicator increased from 38.9% to 42.0% (IRR, 1.011; P = .003) at resident physician practices that used the EPCD model and decreased from 36.2% to 35.5% (IRR, 0.994; P < .001) at staff clinician practices that did not use the model.
In contrast to the team-based approach used in China, the EPCD protocol “is very complex, and it will be difficult to implement in low-resource settings,” Dr. He said.
The D4C trial was funded by the Xiamen Municipal Health Commission. The Mayo study was funded by a National Institutes of Diabetes and Digestive and Kidney Diseases grant. Dr. He, Dr. Peters, and Dr. Huang reported no relevant financial interests.
a randomized trial in China showed.
The tool required clinicians to enter patient data into a computer in order to generate individualized treatment recommendations, adding to their administrative burdens. It also couldn’t tackle patients’ problems with access and affordability of medications.
Nevertheless, the model could curtail physician burnout and improve the quality of care in primary care clinics with limited resources, the researchers said in a paper published in the Annals of Internal Medicine.
They concluded that the findings support “widespread adoption” of the model in China and other low- or middle-income countries where diabetes is on the rise.
Co–principal investigator Jiang He, MD, PhD, chair of epidemiology at Tulane University, New Orleans, said the findings could apply to federally qualified health care (FQHC) clinics that treat underserved patients in the United States.
“At many FQHC clinics, nurse practitioners have to take care of patients with multiple chronic disease conditions. Team-based care with a computerized clinical decision support system will help them and improve patient care,” Dr. He said.
Small improvements
To conduct the trial, called Diabetes Complication Control in Community Clinics (D4C), Dr. He and colleagues randomly assigned 19 out of the 38 community health centers in Xiamen, China, to have a clinical decision support tool installed on the computers of primary care physicians and health coaches.
Starting in October 2016 the researchers recruited 11,132 patients aged 50 and older with uncontrolled diabetes and at least one comorbid condition, with 5,475 patients receiving team-based care with the CDSS and the remainder receiving team-based care alone.
The CDSS generated individualized risk factor summaries and treatment recommendations, including prescriptions based on Chinese and U.S. clinical guidelines. It incorporated data on patients’ insurance plans and local availability of drugs.
At all centers, primary care physicians received training in managing glycemia, blood pressure, and lipids. Nurses were certified as health coaches after receiving training on nutrition, lifestyle changes, and medication adherence. Patients met with their coaches for half an hour every 3 months, and diabetes specialists visited each clinic monthly for team meetings and consultations.
After 18 months, patients undergoing team-based care alone lowered their hemoglobin A1c by 0.6 percentage points (95% confidence interval, –0.7 to –0.5 percentage points), LDL cholesterol by 12.5 mg/dL (95% CI, –13.6 to –11.3 mg/dL), and systolic blood pressure by 7.5 mm Hg (95% CI, –8.4 to –6.6 mm Hg).
The group whose care teams used the CDSS further reduced A1c by 0.2 percentage points (95% CI, –0.3 to –0.1 percentage points), LDL cholesterol by 6.5 mg/dL (95% CI, –8.3 to -4.6 mg/dL), and blood pressure by 1.5 mm Hg (95% CI, –2.8 to –0.3 mm Hg).
All-cause mortality did not differ between the groups. Serious adverse events occurred in 9.1% of the CDSS group, compared with 10.9% of the group whose care team did not use the CDSS.
Addressing social needs
Experts who were not involved in the trial said the marginal impact of the CDSS was no surprise given the mixed results of such tools in previous studies.
However, the lackluster result “might be a shock to people investing a lot in clinical decision support,” said Elbert Huang, MD, MPH, director of the Center for Chronic Disease Research and Policy at the University of Chicago.
Anne Peters, MD, a professor of medicine at the University of Southern California, Los Angeles, said the administrative burden of entering each patient’s data into the system would slow down care and frustrate clinicians. “The system has to be smarter than this.”
On the other hand, the findings of the D4C trial align with other research showing that team-based care strategies are effective for diabetes management.
Dr. Huang noted that there is a “well-established history” of diabetes quality improvement programs, health coaches, buddy programs, and community health worker programs. He added that the new findings “might help to remind everyone of the importance of these programs, which are not always well supported.”
“The bottom line of the paper might be that investing in patient engagement programs might get us 90% of the way to our goal of improving diabetes care,” Dr. Huang said.
Still, Dr. Peters said the portion of patients in the trial who benefited from team-based care seemed “disturbingly low.” Just 16.9% of patients who received team-based care and CDSS and 13% of those who received team-based care alone improved in all three measures. “This system doesn’t get you to where you want to be by a long shot.”
She added that a team-based approach, particularly the use of health coaches, would be a “huge improvement” over fragmented care provided in much of the U.S. safety-net system.
Another team approach
Many systems are striving to improve diabetes management in response to payment incentives, Dr. Huang said.
In a separate retrospective analysis, published in Annals of Family Medicine, researchers at the Mayo Clinic, Rochester, Minn., reported quality improvement gains among primary care practices that adopted a team-based model called Enhanced Primary Care Diabetes (EPCD). The model deployed a range of strategies, such as empowering nurses to engage with patients outside of scheduled office visits and including pharmacists on care teams.
Mayo’s approach did not specifically target underserved populations. Rather, researchers evaluated the model’s impact on about 17,000 patients treated at 32 Mayo internal medicine and family medicine practices of varying sizes, resources, and community settings.
Among staff clinician practices using the EPCD model improved patients’ scores on a composite quality measure called D5, which incorporates glycemic control, blood pressure control, low-density lipoprotein control, tobacco abstinence, and aspirin use.
Following implementation, the portion of patients in those practices meeting the D5 indicator increased from 42.9% to 45.0% (incident rate ratio, 1.005; P = .001).
Meanwhile, the portion of patients meeting the indicator increased from 38.9% to 42.0% (IRR, 1.011; P = .003) at resident physician practices that used the EPCD model and decreased from 36.2% to 35.5% (IRR, 0.994; P < .001) at staff clinician practices that did not use the model.
In contrast to the team-based approach used in China, the EPCD protocol “is very complex, and it will be difficult to implement in low-resource settings,” Dr. He said.
The D4C trial was funded by the Xiamen Municipal Health Commission. The Mayo study was funded by a National Institutes of Diabetes and Digestive and Kidney Diseases grant. Dr. He, Dr. Peters, and Dr. Huang reported no relevant financial interests.
a randomized trial in China showed.
The tool required clinicians to enter patient data into a computer in order to generate individualized treatment recommendations, adding to their administrative burdens. It also couldn’t tackle patients’ problems with access and affordability of medications.
Nevertheless, the model could curtail physician burnout and improve the quality of care in primary care clinics with limited resources, the researchers said in a paper published in the Annals of Internal Medicine.
They concluded that the findings support “widespread adoption” of the model in China and other low- or middle-income countries where diabetes is on the rise.
Co–principal investigator Jiang He, MD, PhD, chair of epidemiology at Tulane University, New Orleans, said the findings could apply to federally qualified health care (FQHC) clinics that treat underserved patients in the United States.
“At many FQHC clinics, nurse practitioners have to take care of patients with multiple chronic disease conditions. Team-based care with a computerized clinical decision support system will help them and improve patient care,” Dr. He said.
Small improvements
To conduct the trial, called Diabetes Complication Control in Community Clinics (D4C), Dr. He and colleagues randomly assigned 19 out of the 38 community health centers in Xiamen, China, to have a clinical decision support tool installed on the computers of primary care physicians and health coaches.
Starting in October 2016 the researchers recruited 11,132 patients aged 50 and older with uncontrolled diabetes and at least one comorbid condition, with 5,475 patients receiving team-based care with the CDSS and the remainder receiving team-based care alone.
The CDSS generated individualized risk factor summaries and treatment recommendations, including prescriptions based on Chinese and U.S. clinical guidelines. It incorporated data on patients’ insurance plans and local availability of drugs.
At all centers, primary care physicians received training in managing glycemia, blood pressure, and lipids. Nurses were certified as health coaches after receiving training on nutrition, lifestyle changes, and medication adherence. Patients met with their coaches for half an hour every 3 months, and diabetes specialists visited each clinic monthly for team meetings and consultations.
After 18 months, patients undergoing team-based care alone lowered their hemoglobin A1c by 0.6 percentage points (95% confidence interval, –0.7 to –0.5 percentage points), LDL cholesterol by 12.5 mg/dL (95% CI, –13.6 to –11.3 mg/dL), and systolic blood pressure by 7.5 mm Hg (95% CI, –8.4 to –6.6 mm Hg).
The group whose care teams used the CDSS further reduced A1c by 0.2 percentage points (95% CI, –0.3 to –0.1 percentage points), LDL cholesterol by 6.5 mg/dL (95% CI, –8.3 to -4.6 mg/dL), and blood pressure by 1.5 mm Hg (95% CI, –2.8 to –0.3 mm Hg).
All-cause mortality did not differ between the groups. Serious adverse events occurred in 9.1% of the CDSS group, compared with 10.9% of the group whose care team did not use the CDSS.
Addressing social needs
Experts who were not involved in the trial said the marginal impact of the CDSS was no surprise given the mixed results of such tools in previous studies.
However, the lackluster result “might be a shock to people investing a lot in clinical decision support,” said Elbert Huang, MD, MPH, director of the Center for Chronic Disease Research and Policy at the University of Chicago.
Anne Peters, MD, a professor of medicine at the University of Southern California, Los Angeles, said the administrative burden of entering each patient’s data into the system would slow down care and frustrate clinicians. “The system has to be smarter than this.”
On the other hand, the findings of the D4C trial align with other research showing that team-based care strategies are effective for diabetes management.
Dr. Huang noted that there is a “well-established history” of diabetes quality improvement programs, health coaches, buddy programs, and community health worker programs. He added that the new findings “might help to remind everyone of the importance of these programs, which are not always well supported.”
“The bottom line of the paper might be that investing in patient engagement programs might get us 90% of the way to our goal of improving diabetes care,” Dr. Huang said.
Still, Dr. Peters said the portion of patients in the trial who benefited from team-based care seemed “disturbingly low.” Just 16.9% of patients who received team-based care and CDSS and 13% of those who received team-based care alone improved in all three measures. “This system doesn’t get you to where you want to be by a long shot.”
She added that a team-based approach, particularly the use of health coaches, would be a “huge improvement” over fragmented care provided in much of the U.S. safety-net system.
Another team approach
Many systems are striving to improve diabetes management in response to payment incentives, Dr. Huang said.
In a separate retrospective analysis, published in Annals of Family Medicine, researchers at the Mayo Clinic, Rochester, Minn., reported quality improvement gains among primary care practices that adopted a team-based model called Enhanced Primary Care Diabetes (EPCD). The model deployed a range of strategies, such as empowering nurses to engage with patients outside of scheduled office visits and including pharmacists on care teams.
Mayo’s approach did not specifically target underserved populations. Rather, researchers evaluated the model’s impact on about 17,000 patients treated at 32 Mayo internal medicine and family medicine practices of varying sizes, resources, and community settings.
Among staff clinician practices using the EPCD model improved patients’ scores on a composite quality measure called D5, which incorporates glycemic control, blood pressure control, low-density lipoprotein control, tobacco abstinence, and aspirin use.
Following implementation, the portion of patients in those practices meeting the D5 indicator increased from 42.9% to 45.0% (incident rate ratio, 1.005; P = .001).
Meanwhile, the portion of patients meeting the indicator increased from 38.9% to 42.0% (IRR, 1.011; P = .003) at resident physician practices that used the EPCD model and decreased from 36.2% to 35.5% (IRR, 0.994; P < .001) at staff clinician practices that did not use the model.
In contrast to the team-based approach used in China, the EPCD protocol “is very complex, and it will be difficult to implement in low-resource settings,” Dr. He said.
The D4C trial was funded by the Xiamen Municipal Health Commission. The Mayo study was funded by a National Institutes of Diabetes and Digestive and Kidney Diseases grant. Dr. He, Dr. Peters, and Dr. Huang reported no relevant financial interests.
FROM ANNALS OF INTERNAL MEDICINE
Florida doc dies by suicide after allegedly drugging and raping patients
according to a police statement.
A week later, a Collier County Sheriff’s deputy found Dr. Salata’s body near his Naples home with a gunshot wound to the head, according to police. The medical examiner later ruled it a suicide.
Dr. Salata co-owned Pura Vida Medical Spa in Naples with his wife Jill Salata, a certified family nurse practitioner. They specialized in cosmetic treatment and surgery.
Naples police said that they arrested Dr. Salata after two female patients accused the doctor of allegedly drugging and raping them while they were still unconscious.
Both victims described being given nitrous oxide, also called laughing gas, for sedation and pain from the cosmetic procedure. The first victim, age 51, said Dr. Salata prescribed alprazolam (Xanax) to take before the procedure and then also gave her nitrous oxide and tequila, causing her to black out, according to NBC2 News.
The second victim, age 72, told police that as the nitrous oxide was wearing off, she found Dr. Salata performing sexual intercourse. The victim felt shocked after the sedation subsided about what had taken place, contacted police, and submitted to a sexual assault examination, according to the police statement.
At Dr. Salata’s November 22 hearing before Judge Michael Provost, a prosecutor asked the judge whether Dr. Salata should surrender his firearms; Provost reportedly dismissed the idea.
“It is disappointing and frustrating that Dr. Salata has escaped justice,” said one victim’s attorney, Adam Horowitz, in a blog post. “Yet, we are relieved that no other women will be assaulted by Dr. Salata again. It took tremendous courage for my client to tell her truth. She was ready to hold him accountable in court.”
Horowitz says he plans to file a civil lawsuit on behalf of his client against Dr. Salata’s estate. The Naples police are continuing their investigation into the victims’ cases, which now includes a third woman, said spokesman Lt. Bryan McGinn.
Meanwhile, the Pura Vida Medical Spa has closed permanently and its website has been deleted. One reviewer named Soul F. wrote on the spa’s Yelp page: “And now may God have mercy on this rapist’s soul. Amen.”
A version of this article first appeared on Medscape.com.
according to a police statement.
A week later, a Collier County Sheriff’s deputy found Dr. Salata’s body near his Naples home with a gunshot wound to the head, according to police. The medical examiner later ruled it a suicide.
Dr. Salata co-owned Pura Vida Medical Spa in Naples with his wife Jill Salata, a certified family nurse practitioner. They specialized in cosmetic treatment and surgery.
Naples police said that they arrested Dr. Salata after two female patients accused the doctor of allegedly drugging and raping them while they were still unconscious.
Both victims described being given nitrous oxide, also called laughing gas, for sedation and pain from the cosmetic procedure. The first victim, age 51, said Dr. Salata prescribed alprazolam (Xanax) to take before the procedure and then also gave her nitrous oxide and tequila, causing her to black out, according to NBC2 News.
The second victim, age 72, told police that as the nitrous oxide was wearing off, she found Dr. Salata performing sexual intercourse. The victim felt shocked after the sedation subsided about what had taken place, contacted police, and submitted to a sexual assault examination, according to the police statement.
At Dr. Salata’s November 22 hearing before Judge Michael Provost, a prosecutor asked the judge whether Dr. Salata should surrender his firearms; Provost reportedly dismissed the idea.
“It is disappointing and frustrating that Dr. Salata has escaped justice,” said one victim’s attorney, Adam Horowitz, in a blog post. “Yet, we are relieved that no other women will be assaulted by Dr. Salata again. It took tremendous courage for my client to tell her truth. She was ready to hold him accountable in court.”
Horowitz says he plans to file a civil lawsuit on behalf of his client against Dr. Salata’s estate. The Naples police are continuing their investigation into the victims’ cases, which now includes a third woman, said spokesman Lt. Bryan McGinn.
Meanwhile, the Pura Vida Medical Spa has closed permanently and its website has been deleted. One reviewer named Soul F. wrote on the spa’s Yelp page: “And now may God have mercy on this rapist’s soul. Amen.”
A version of this article first appeared on Medscape.com.
according to a police statement.
A week later, a Collier County Sheriff’s deputy found Dr. Salata’s body near his Naples home with a gunshot wound to the head, according to police. The medical examiner later ruled it a suicide.
Dr. Salata co-owned Pura Vida Medical Spa in Naples with his wife Jill Salata, a certified family nurse practitioner. They specialized in cosmetic treatment and surgery.
Naples police said that they arrested Dr. Salata after two female patients accused the doctor of allegedly drugging and raping them while they were still unconscious.
Both victims described being given nitrous oxide, also called laughing gas, for sedation and pain from the cosmetic procedure. The first victim, age 51, said Dr. Salata prescribed alprazolam (Xanax) to take before the procedure and then also gave her nitrous oxide and tequila, causing her to black out, according to NBC2 News.
The second victim, age 72, told police that as the nitrous oxide was wearing off, she found Dr. Salata performing sexual intercourse. The victim felt shocked after the sedation subsided about what had taken place, contacted police, and submitted to a sexual assault examination, according to the police statement.
At Dr. Salata’s November 22 hearing before Judge Michael Provost, a prosecutor asked the judge whether Dr. Salata should surrender his firearms; Provost reportedly dismissed the idea.
“It is disappointing and frustrating that Dr. Salata has escaped justice,” said one victim’s attorney, Adam Horowitz, in a blog post. “Yet, we are relieved that no other women will be assaulted by Dr. Salata again. It took tremendous courage for my client to tell her truth. She was ready to hold him accountable in court.”
Horowitz says he plans to file a civil lawsuit on behalf of his client against Dr. Salata’s estate. The Naples police are continuing their investigation into the victims’ cases, which now includes a third woman, said spokesman Lt. Bryan McGinn.
Meanwhile, the Pura Vida Medical Spa has closed permanently and its website has been deleted. One reviewer named Soul F. wrote on the spa’s Yelp page: “And now may God have mercy on this rapist’s soul. Amen.”
A version of this article first appeared on Medscape.com.
Dapagliflozin reduces hospitalizations in patients with CKD
These findings add to a growing body of evidence supporting a range of positive benefits from dapagliflozin, including reduced risks of mortality, cardiovascular events, and kidney events, lead author Meir Schechter, MD, PhD, of the Hebrew University of Jerusalem and colleagues wrote in Annals of Internal Medicine.“Although cardiovascular and kidney outcomes with SGLT2 inhibitors have been studied extensively, there is a paucity of data evaluating the effects of SGLT2 inhibitors on hospitalizations for any cause.”
The findings are based on a post hoc analysis of the DAPA-CKD trial, which involved 4,304 patients with CKD in 21 countries. Patients were randomized in a 1:1 ratio to receive dapagliflozin 10 mg orally once a day or matching placebo. The present analysis quantified first hospitalizations for any cause, all hospitalizations, cause-specific hospitalizations, and several related outcomes.
After a median follow-up of 2.4 years, 28% of the population had been hospitalized a total of 2,072 times.
Compared with placebo, dapagliflozin significantly reduced risk of first hospitalization by 16% (hazard ratio, 0.84; 95% confidence interval, 0.75-0.94) and rate of all hospitalizations by 21% (rate ratio, 0.79; 95% CI, 0.70-0.89). These findings remained significant regardless of type 2 diabetes status, with significant benefits seen across reasons for admission, including renal/urinary disorders, cardiac disorders, neoplasms, and metabolism/nutrition disorders. In addition, dapagliflozin was associated with shorter mean time in hospital (2.3 vs. 2.8 days; P = .027) and longer time alive and out of hospital (354.9 vs. 351.7; P = .023).
“These findings highlight additional benefits of dapagliflozin beyond those seen for cardiovascular and kidney events, all-cause and cause-specific mortality, eGFR [estimated glomerular filtration rate] slope, and albuminuria and should be considered when evaluating the totality of evidence favoring provision of dapagliflozin to patients with CKD,” the investigators concluded.
Positive data, positive experiences
Shree Mulay, MD, a nephrologist in private practice in western Tennessee, said this study is “one of several other articles that already exist” demonstrating the broad benefits of SGLT2 inhibitors.
“The evidence is pretty substantial,” Dr. Mulay said in an interview. “I think SGLT2 inhibitors are the new statin of this era. ... I won’t be surprised if in the next year or 2 or 3 they truly become the standard of care.”
Dr. Mulay also speaks from experience working in both the chronic and acute setting, where he’s observed “some magical stuff happening” in patients started on SGLT2 inhibitors, especially those in heart failure who are fluid overloaded.
“It’s phenomenal stuff,” Dr. Mulay said. “You can really stabilize patients’ hemodynamics.”
In the private health care setting, he described widespread enthusiasm among nephrologists, although others still appear skeptical.
“It’s really our cardiology colleagues that I feel are underprescribing it,” Dr. Mulay said. “So, I’m kind of taking it on myself, when I see a heart failure patient, to go ahead and put them on this.”
It’s unclear why some cardiologists seem apprehensive, Dr. Mulay continued, although he suggested that unclear guidelines and a lack of first-hand experience may be to blame.
Nephrologists and cardiologists sometimes agree
In the academic arena, Leslie Gewin, MD, associate professor at Washington University in St. Louis and the John Cochran VA Hospital, also in St. Louis, has seen similar support for SGLT2 inhibitors among both nephrologists and cardiologists.
“We had a joint nephrology-cardiology medicine grand rounds at Wash U in St. Louis maybe 2 weeks ago,” Dr. Gewin said in an interview. “The cardiologists and nephrologists tag-teamed to present data about SGLT2 inhibitors, and we kind of joked that this was the one thing we both could get behind and support.”
Still, she has seen some reluctance among non-nephrology clinicians lacking SGLT2 experience, specifically when managing patients who have poor kidney function.
“There can be some hesitancy among physicians if the GFR is low,” Dr. Gewin said. “That’s where I’ve had to sort of push the envelope with non-nephrologists, saying: ‘Look, we feel pretty comfortable starting down to a GFR of about 20.’ ”
Early rises in creatinine may also spook providers, she noted.
“Sometimes, when we start SGLT2 inhibitors, the creatinine increases slightly, and the [primary care provider] gets concerned,” Dr. Gewin said. “We say: ‘No, this is expected. Don’t worry, hold the course, this is a good drug.’ ”
Like Dr. Mulay, Dr. Gewin said the present study offers further encouragement for the efficacy of this drug class. She also said sufficient data have been published to allay earlier concerns about potential safety signals, such as bone fractures and amputations.
“SGLT2 inhibitors seem to be a lot safer than what we initially had thought,” Dr. Gewin said. “That’s very encouraging.”
The study was funded by AstraZeneca. The investigators disclosed additional relationships with Bayer, Janssen, Gilead, and others. Dr. Gewin and Dr. Mulay disclosed no relevant conflicts of interest.
These findings add to a growing body of evidence supporting a range of positive benefits from dapagliflozin, including reduced risks of mortality, cardiovascular events, and kidney events, lead author Meir Schechter, MD, PhD, of the Hebrew University of Jerusalem and colleagues wrote in Annals of Internal Medicine.“Although cardiovascular and kidney outcomes with SGLT2 inhibitors have been studied extensively, there is a paucity of data evaluating the effects of SGLT2 inhibitors on hospitalizations for any cause.”
The findings are based on a post hoc analysis of the DAPA-CKD trial, which involved 4,304 patients with CKD in 21 countries. Patients were randomized in a 1:1 ratio to receive dapagliflozin 10 mg orally once a day or matching placebo. The present analysis quantified first hospitalizations for any cause, all hospitalizations, cause-specific hospitalizations, and several related outcomes.
After a median follow-up of 2.4 years, 28% of the population had been hospitalized a total of 2,072 times.
Compared with placebo, dapagliflozin significantly reduced risk of first hospitalization by 16% (hazard ratio, 0.84; 95% confidence interval, 0.75-0.94) and rate of all hospitalizations by 21% (rate ratio, 0.79; 95% CI, 0.70-0.89). These findings remained significant regardless of type 2 diabetes status, with significant benefits seen across reasons for admission, including renal/urinary disorders, cardiac disorders, neoplasms, and metabolism/nutrition disorders. In addition, dapagliflozin was associated with shorter mean time in hospital (2.3 vs. 2.8 days; P = .027) and longer time alive and out of hospital (354.9 vs. 351.7; P = .023).
“These findings highlight additional benefits of dapagliflozin beyond those seen for cardiovascular and kidney events, all-cause and cause-specific mortality, eGFR [estimated glomerular filtration rate] slope, and albuminuria and should be considered when evaluating the totality of evidence favoring provision of dapagliflozin to patients with CKD,” the investigators concluded.
Positive data, positive experiences
Shree Mulay, MD, a nephrologist in private practice in western Tennessee, said this study is “one of several other articles that already exist” demonstrating the broad benefits of SGLT2 inhibitors.
“The evidence is pretty substantial,” Dr. Mulay said in an interview. “I think SGLT2 inhibitors are the new statin of this era. ... I won’t be surprised if in the next year or 2 or 3 they truly become the standard of care.”
Dr. Mulay also speaks from experience working in both the chronic and acute setting, where he’s observed “some magical stuff happening” in patients started on SGLT2 inhibitors, especially those in heart failure who are fluid overloaded.
“It’s phenomenal stuff,” Dr. Mulay said. “You can really stabilize patients’ hemodynamics.”
In the private health care setting, he described widespread enthusiasm among nephrologists, although others still appear skeptical.
“It’s really our cardiology colleagues that I feel are underprescribing it,” Dr. Mulay said. “So, I’m kind of taking it on myself, when I see a heart failure patient, to go ahead and put them on this.”
It’s unclear why some cardiologists seem apprehensive, Dr. Mulay continued, although he suggested that unclear guidelines and a lack of first-hand experience may be to blame.
Nephrologists and cardiologists sometimes agree
In the academic arena, Leslie Gewin, MD, associate professor at Washington University in St. Louis and the John Cochran VA Hospital, also in St. Louis, has seen similar support for SGLT2 inhibitors among both nephrologists and cardiologists.
“We had a joint nephrology-cardiology medicine grand rounds at Wash U in St. Louis maybe 2 weeks ago,” Dr. Gewin said in an interview. “The cardiologists and nephrologists tag-teamed to present data about SGLT2 inhibitors, and we kind of joked that this was the one thing we both could get behind and support.”
Still, she has seen some reluctance among non-nephrology clinicians lacking SGLT2 experience, specifically when managing patients who have poor kidney function.
“There can be some hesitancy among physicians if the GFR is low,” Dr. Gewin said. “That’s where I’ve had to sort of push the envelope with non-nephrologists, saying: ‘Look, we feel pretty comfortable starting down to a GFR of about 20.’ ”
Early rises in creatinine may also spook providers, she noted.
“Sometimes, when we start SGLT2 inhibitors, the creatinine increases slightly, and the [primary care provider] gets concerned,” Dr. Gewin said. “We say: ‘No, this is expected. Don’t worry, hold the course, this is a good drug.’ ”
Like Dr. Mulay, Dr. Gewin said the present study offers further encouragement for the efficacy of this drug class. She also said sufficient data have been published to allay earlier concerns about potential safety signals, such as bone fractures and amputations.
“SGLT2 inhibitors seem to be a lot safer than what we initially had thought,” Dr. Gewin said. “That’s very encouraging.”
The study was funded by AstraZeneca. The investigators disclosed additional relationships with Bayer, Janssen, Gilead, and others. Dr. Gewin and Dr. Mulay disclosed no relevant conflicts of interest.
These findings add to a growing body of evidence supporting a range of positive benefits from dapagliflozin, including reduced risks of mortality, cardiovascular events, and kidney events, lead author Meir Schechter, MD, PhD, of the Hebrew University of Jerusalem and colleagues wrote in Annals of Internal Medicine.“Although cardiovascular and kidney outcomes with SGLT2 inhibitors have been studied extensively, there is a paucity of data evaluating the effects of SGLT2 inhibitors on hospitalizations for any cause.”
The findings are based on a post hoc analysis of the DAPA-CKD trial, which involved 4,304 patients with CKD in 21 countries. Patients were randomized in a 1:1 ratio to receive dapagliflozin 10 mg orally once a day or matching placebo. The present analysis quantified first hospitalizations for any cause, all hospitalizations, cause-specific hospitalizations, and several related outcomes.
After a median follow-up of 2.4 years, 28% of the population had been hospitalized a total of 2,072 times.
Compared with placebo, dapagliflozin significantly reduced risk of first hospitalization by 16% (hazard ratio, 0.84; 95% confidence interval, 0.75-0.94) and rate of all hospitalizations by 21% (rate ratio, 0.79; 95% CI, 0.70-0.89). These findings remained significant regardless of type 2 diabetes status, with significant benefits seen across reasons for admission, including renal/urinary disorders, cardiac disorders, neoplasms, and metabolism/nutrition disorders. In addition, dapagliflozin was associated with shorter mean time in hospital (2.3 vs. 2.8 days; P = .027) and longer time alive and out of hospital (354.9 vs. 351.7; P = .023).
“These findings highlight additional benefits of dapagliflozin beyond those seen for cardiovascular and kidney events, all-cause and cause-specific mortality, eGFR [estimated glomerular filtration rate] slope, and albuminuria and should be considered when evaluating the totality of evidence favoring provision of dapagliflozin to patients with CKD,” the investigators concluded.
Positive data, positive experiences
Shree Mulay, MD, a nephrologist in private practice in western Tennessee, said this study is “one of several other articles that already exist” demonstrating the broad benefits of SGLT2 inhibitors.
“The evidence is pretty substantial,” Dr. Mulay said in an interview. “I think SGLT2 inhibitors are the new statin of this era. ... I won’t be surprised if in the next year or 2 or 3 they truly become the standard of care.”
Dr. Mulay also speaks from experience working in both the chronic and acute setting, where he’s observed “some magical stuff happening” in patients started on SGLT2 inhibitors, especially those in heart failure who are fluid overloaded.
“It’s phenomenal stuff,” Dr. Mulay said. “You can really stabilize patients’ hemodynamics.”
In the private health care setting, he described widespread enthusiasm among nephrologists, although others still appear skeptical.
“It’s really our cardiology colleagues that I feel are underprescribing it,” Dr. Mulay said. “So, I’m kind of taking it on myself, when I see a heart failure patient, to go ahead and put them on this.”
It’s unclear why some cardiologists seem apprehensive, Dr. Mulay continued, although he suggested that unclear guidelines and a lack of first-hand experience may be to blame.
Nephrologists and cardiologists sometimes agree
In the academic arena, Leslie Gewin, MD, associate professor at Washington University in St. Louis and the John Cochran VA Hospital, also in St. Louis, has seen similar support for SGLT2 inhibitors among both nephrologists and cardiologists.
“We had a joint nephrology-cardiology medicine grand rounds at Wash U in St. Louis maybe 2 weeks ago,” Dr. Gewin said in an interview. “The cardiologists and nephrologists tag-teamed to present data about SGLT2 inhibitors, and we kind of joked that this was the one thing we both could get behind and support.”
Still, she has seen some reluctance among non-nephrology clinicians lacking SGLT2 experience, specifically when managing patients who have poor kidney function.
“There can be some hesitancy among physicians if the GFR is low,” Dr. Gewin said. “That’s where I’ve had to sort of push the envelope with non-nephrologists, saying: ‘Look, we feel pretty comfortable starting down to a GFR of about 20.’ ”
Early rises in creatinine may also spook providers, she noted.
“Sometimes, when we start SGLT2 inhibitors, the creatinine increases slightly, and the [primary care provider] gets concerned,” Dr. Gewin said. “We say: ‘No, this is expected. Don’t worry, hold the course, this is a good drug.’ ”
Like Dr. Mulay, Dr. Gewin said the present study offers further encouragement for the efficacy of this drug class. She also said sufficient data have been published to allay earlier concerns about potential safety signals, such as bone fractures and amputations.
“SGLT2 inhibitors seem to be a lot safer than what we initially had thought,” Dr. Gewin said. “That’s very encouraging.”
The study was funded by AstraZeneca. The investigators disclosed additional relationships with Bayer, Janssen, Gilead, and others. Dr. Gewin and Dr. Mulay disclosed no relevant conflicts of interest.
FROM ANNALS OF INTERNAL MEDICINE
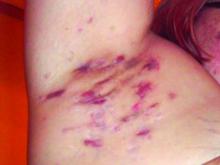
Consider quality of life, comorbidities in hidradenitis suppurativa
LAS VEGAS – , Robert G. Micheletti, MD, said in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
For patients with HS, “the quality-of-life impact is profound, greater than any other systematically studied dermatologic condition,” said Dr. Micheletti, associate professor of dermatology at the Hospital of the University of Pennsylavnia, and chief of hospital dermatology, and chief of dermatology at Pennsylvania Hospital, Philadelphia.
Two key aspects of quality of life that affect HS patients are sexual health and overall pain, he said. The female-to-male ratio of HS is approximately 3:1, and data show that approximately 40% of female HS patients experience fertility issues and have unaddressed questions about HS and pregnancy, said Dr. Micheletti. Additionally, data from a systematic review showed that 50%-60% of patients with HS reported sexual dysfunction. Impaired sexual function is also associated with both overall impaired quality of life ratings and the presence of mood disorders, he noted.
Pain also has a significant impact on quality of life for HS patients. When these patients present in an emergency department, 70% report severe pain, and approximately 60% receive opioids, said Dr. Micheletti.
Data from a 2021 study showed that HS patients are significantly more likely to receive opioids compared with controls, and also more likely to be diagnosed with opioid use disorder than controls, especially if they are seen by nondermatologists, he noted.
For acute pain, Dr. Micheletti recommended starting with acetaminophen 500 mg every 4 to 6 hours as needed, and topical nonsteroidal anti-inflammatory drugs (NSAIDs). “It still makes sense to do topical care,” said Dr. Micheletti, but he added that he also prescribes medications for anxiety for these patients.
Patients with increased pain severity or refractory disease may benefit from systemic NSAIDs, or intralesional triamcinolone, he noted. Incision and draining of abscesses may provide temporary symptomatic relief, but keep in mind that lesions will recur, he noted.
For the most severe cases, Dr. Micheletti advised adding tramadol as a first-line opioid, or another short-acting opioid for breakthrough pain.
To manage patients with HS who have chronic pain, Dr. Micheletti recommended starting with HS disease–directed therapy, but also screening for pain severity and psychological comorbidities.
His strategies in these cases include nonpharmacological pain management in the form of physical therapy, wound care, and behavioral health. His algorithm for nociceptive pain is NSAIDs with or without acetaminophen; duloxetine or nortriptyline are other options. For neuropathic pain, gabapentin and/or duloxetine are top choices, but pregabalin, venlafaxine, and nortriptyline are on the list as well.
Topical NSAIDs or topical lidocaine may serve as add-ons to systemic therapy in more severe cases, or as first-line therapy for milder chronic pain, Dr. Micheletti noted. Patients who have failed treatment with at least two pharmacologic agents, suffer medically refractory HS with debilitating pain, or use opioids on an ongoing basis should be referred to a pain management specialist, he said.
Don’t forget lifestyle
Although data on the impact of diet on patients with HS are limited, “we know anecdotally that dairy and refined carbohydrates are associated with exacerbations,” said Dr. Micheletti.
In addition, many patients use complementary medicine “and they aren’t always telling us,” he emphasized. Smoking is prevalent among patients with HS, and is a risk factor for the disease in general, and for more severe and refractory disease, he added. Consequently, screening for tobacco smoking is recommended for patients with HS not only because of the impact on disease, but because it is a potentially modifiable cardiovascular risk factor, he explained.
Consider comorbidities
Cardiovascular disease is among several comorbidities associated with HS, said Dr. Micheletti. HS foundations in the United States and Canada recently published evidence-based recommendations for comorbidity screening. The recommendations included screening for 19 specific comorbidities: acne, dissecting cellulitis, pilonidal disease, pyoderma gangrenosum, depression, anxiety, suicide, smoking, substance abuse, polycystic ovary syndrome, obesity, dyslipidemia, diabetes mellitus, metabolic syndrome, hypertension, cardiovascular disease, inflammatory bowel disease, spondyloarthritis, and sexual dysfunction.
Dr. Micheletti highlighted cardiovascular comorbidities, and noted the association between HS and modifiable cardiovascular risk factors: smoking, obesity, diabetes mellitus, and dyslipidemia. “HS is also independently associated with cardiovascular disease leading to myocardial infarction, stroke, cardiovascular-associated death, and all-cause mortality compared to controls,” he said. Studies show an incidence rate ratio of 1.53 for major adverse cardiovascular events in patients with HS compared with controls, with the highest relative risk among those aged 18-29 years, he added.
Medical management
Depending on the patient, medical management of HS may involve antibiotics, hormonal agents, and biologics, said Dr. Micheletti. Some of the most commonly used antibiotic regimens for HS are those recommended in treatment guidelines, including doxycycline and a clindamycin/rifampin combination, he said. However, the use of trimethoprim-sulfamethoxazole or ciprofloxacin has been associated with increased antibiotic resistance and is not supported by available evidence, he noted.
Hormonal therapies may help some women with HS, said Dr. Micheletti. Options include spironolactone, metformin, or estrogen-containing hormonal contraceptives, he said.
When it comes to biologics, only 33% of HS patients meet criteria for their use (Hurley stage II or III, moderate or severe HS), he noted. However, research suggests “a huge gap” in the use of anti-TNF therapy even among patients for whom it is recommended, he said.
Of the TNF-alpha inhibitors, data on adalimumab, which is FDA-approved for HS, are the most recent. Adalimumab “is our gold standard biologic and our gateway biologic, for HS at this time,” Dr. Micheletti said.
However, those who respond to adalimumab “can continue to do better, but they can wax and wane and flare,” he cautioned. Infliximab, while not approved for HS, has been studied in patients with HS and is prescribed by some providers. Although no comparative studies have been done for infliximab versus adalimumab, “anecdotally, response to infliximab tends to be better, and it is the most effective biologic in common use for severe HS,” he noted.
Dr. Micheletti’s top treatment recommendations for using biologics start with considering biosimilars. Most patients on biosimilars do fine, but some patients who previously responded to infliximab will unpredictably lose efficacy or have reactions when switched to a biosimilar, he said.
Patients on biologics also may experience waning efficacy in the wake of an immune response stimulated by foreign antibodies, said Dr. Micheletti. “Anti-drug antibody formation is more likely to occur when treatment is interrupted,” he noted. Minimize the risk of antibody formation by paying attention to adherence issues and dosing frequency, he advised.
If patients fail both adalimumab and infliximab, Dr. Micheletti tells them not to lose hope, and that treatment is a trial-and-error process that may involve more than one therapy. Other biologics in active use for HS include ustekinumab, anakinra, secukinumab, brodalumab, golimumab, and JAK inhibitors, any of which might be effective in any given patient, he said.
Surgical solutions
For HS patients with chronic, recurring inflammation and drainage associated with a sinus tract, surgical deroofing may the best treatment option, Dr. Micheletti said. “Deroofing involves the use of a probe to trace the extent of the subcutaneous tract, followed by incision and removal of the tract ‘roof,’ ’’ he explained. The deroofing procedure involves local anesthesia and has a low morbidity rate, as well as a low recurrence rate and high levels of patient satisfaction, he said.
“The acute role for surgery is to remove active foci of inflammation and relieve pain,” which is achieved more effectively with deroofing, said Dr. Micheletti. By contrast, incision and drainage is associated with an almost 100% recurrence rate, he added.
When planning elective surgery for HS, Dr. Micheletti noted that holding infliximab for less than 4 weeks does not affect postoperative infection rates in patients with rheumatoid arthritis, and a recent randomized, controlled trial showed that adalimumab can be continued safely through HS surgeries.
In fact, “continuing TNF inhibitors through elective surgery does not increase infection risk and results in better disease control,” and dermatologists should work with surgery to balance infection and disease flare concerns in HS patients, he said.
Dr. Micheletti disclosed serving as a consultant or advisor for Adaptimmune and Vertex, and research funding from Amgen and Cabaletta Bio. MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – , Robert G. Micheletti, MD, said in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
For patients with HS, “the quality-of-life impact is profound, greater than any other systematically studied dermatologic condition,” said Dr. Micheletti, associate professor of dermatology at the Hospital of the University of Pennsylavnia, and chief of hospital dermatology, and chief of dermatology at Pennsylvania Hospital, Philadelphia.
Two key aspects of quality of life that affect HS patients are sexual health and overall pain, he said. The female-to-male ratio of HS is approximately 3:1, and data show that approximately 40% of female HS patients experience fertility issues and have unaddressed questions about HS and pregnancy, said Dr. Micheletti. Additionally, data from a systematic review showed that 50%-60% of patients with HS reported sexual dysfunction. Impaired sexual function is also associated with both overall impaired quality of life ratings and the presence of mood disorders, he noted.
Pain also has a significant impact on quality of life for HS patients. When these patients present in an emergency department, 70% report severe pain, and approximately 60% receive opioids, said Dr. Micheletti.
Data from a 2021 study showed that HS patients are significantly more likely to receive opioids compared with controls, and also more likely to be diagnosed with opioid use disorder than controls, especially if they are seen by nondermatologists, he noted.
For acute pain, Dr. Micheletti recommended starting with acetaminophen 500 mg every 4 to 6 hours as needed, and topical nonsteroidal anti-inflammatory drugs (NSAIDs). “It still makes sense to do topical care,” said Dr. Micheletti, but he added that he also prescribes medications for anxiety for these patients.
Patients with increased pain severity or refractory disease may benefit from systemic NSAIDs, or intralesional triamcinolone, he noted. Incision and draining of abscesses may provide temporary symptomatic relief, but keep in mind that lesions will recur, he noted.
For the most severe cases, Dr. Micheletti advised adding tramadol as a first-line opioid, or another short-acting opioid for breakthrough pain.
To manage patients with HS who have chronic pain, Dr. Micheletti recommended starting with HS disease–directed therapy, but also screening for pain severity and psychological comorbidities.
His strategies in these cases include nonpharmacological pain management in the form of physical therapy, wound care, and behavioral health. His algorithm for nociceptive pain is NSAIDs with or without acetaminophen; duloxetine or nortriptyline are other options. For neuropathic pain, gabapentin and/or duloxetine are top choices, but pregabalin, venlafaxine, and nortriptyline are on the list as well.
Topical NSAIDs or topical lidocaine may serve as add-ons to systemic therapy in more severe cases, or as first-line therapy for milder chronic pain, Dr. Micheletti noted. Patients who have failed treatment with at least two pharmacologic agents, suffer medically refractory HS with debilitating pain, or use opioids on an ongoing basis should be referred to a pain management specialist, he said.
Don’t forget lifestyle
Although data on the impact of diet on patients with HS are limited, “we know anecdotally that dairy and refined carbohydrates are associated with exacerbations,” said Dr. Micheletti.
In addition, many patients use complementary medicine “and they aren’t always telling us,” he emphasized. Smoking is prevalent among patients with HS, and is a risk factor for the disease in general, and for more severe and refractory disease, he added. Consequently, screening for tobacco smoking is recommended for patients with HS not only because of the impact on disease, but because it is a potentially modifiable cardiovascular risk factor, he explained.
Consider comorbidities
Cardiovascular disease is among several comorbidities associated with HS, said Dr. Micheletti. HS foundations in the United States and Canada recently published evidence-based recommendations for comorbidity screening. The recommendations included screening for 19 specific comorbidities: acne, dissecting cellulitis, pilonidal disease, pyoderma gangrenosum, depression, anxiety, suicide, smoking, substance abuse, polycystic ovary syndrome, obesity, dyslipidemia, diabetes mellitus, metabolic syndrome, hypertension, cardiovascular disease, inflammatory bowel disease, spondyloarthritis, and sexual dysfunction.
Dr. Micheletti highlighted cardiovascular comorbidities, and noted the association between HS and modifiable cardiovascular risk factors: smoking, obesity, diabetes mellitus, and dyslipidemia. “HS is also independently associated with cardiovascular disease leading to myocardial infarction, stroke, cardiovascular-associated death, and all-cause mortality compared to controls,” he said. Studies show an incidence rate ratio of 1.53 for major adverse cardiovascular events in patients with HS compared with controls, with the highest relative risk among those aged 18-29 years, he added.
Medical management
Depending on the patient, medical management of HS may involve antibiotics, hormonal agents, and biologics, said Dr. Micheletti. Some of the most commonly used antibiotic regimens for HS are those recommended in treatment guidelines, including doxycycline and a clindamycin/rifampin combination, he said. However, the use of trimethoprim-sulfamethoxazole or ciprofloxacin has been associated with increased antibiotic resistance and is not supported by available evidence, he noted.
Hormonal therapies may help some women with HS, said Dr. Micheletti. Options include spironolactone, metformin, or estrogen-containing hormonal contraceptives, he said.
When it comes to biologics, only 33% of HS patients meet criteria for their use (Hurley stage II or III, moderate or severe HS), he noted. However, research suggests “a huge gap” in the use of anti-TNF therapy even among patients for whom it is recommended, he said.
Of the TNF-alpha inhibitors, data on adalimumab, which is FDA-approved for HS, are the most recent. Adalimumab “is our gold standard biologic and our gateway biologic, for HS at this time,” Dr. Micheletti said.
However, those who respond to adalimumab “can continue to do better, but they can wax and wane and flare,” he cautioned. Infliximab, while not approved for HS, has been studied in patients with HS and is prescribed by some providers. Although no comparative studies have been done for infliximab versus adalimumab, “anecdotally, response to infliximab tends to be better, and it is the most effective biologic in common use for severe HS,” he noted.
Dr. Micheletti’s top treatment recommendations for using biologics start with considering biosimilars. Most patients on biosimilars do fine, but some patients who previously responded to infliximab will unpredictably lose efficacy or have reactions when switched to a biosimilar, he said.
Patients on biologics also may experience waning efficacy in the wake of an immune response stimulated by foreign antibodies, said Dr. Micheletti. “Anti-drug antibody formation is more likely to occur when treatment is interrupted,” he noted. Minimize the risk of antibody formation by paying attention to adherence issues and dosing frequency, he advised.
If patients fail both adalimumab and infliximab, Dr. Micheletti tells them not to lose hope, and that treatment is a trial-and-error process that may involve more than one therapy. Other biologics in active use for HS include ustekinumab, anakinra, secukinumab, brodalumab, golimumab, and JAK inhibitors, any of which might be effective in any given patient, he said.
Surgical solutions
For HS patients with chronic, recurring inflammation and drainage associated with a sinus tract, surgical deroofing may the best treatment option, Dr. Micheletti said. “Deroofing involves the use of a probe to trace the extent of the subcutaneous tract, followed by incision and removal of the tract ‘roof,’ ’’ he explained. The deroofing procedure involves local anesthesia and has a low morbidity rate, as well as a low recurrence rate and high levels of patient satisfaction, he said.
“The acute role for surgery is to remove active foci of inflammation and relieve pain,” which is achieved more effectively with deroofing, said Dr. Micheletti. By contrast, incision and drainage is associated with an almost 100% recurrence rate, he added.
When planning elective surgery for HS, Dr. Micheletti noted that holding infliximab for less than 4 weeks does not affect postoperative infection rates in patients with rheumatoid arthritis, and a recent randomized, controlled trial showed that adalimumab can be continued safely through HS surgeries.
In fact, “continuing TNF inhibitors through elective surgery does not increase infection risk and results in better disease control,” and dermatologists should work with surgery to balance infection and disease flare concerns in HS patients, he said.
Dr. Micheletti disclosed serving as a consultant or advisor for Adaptimmune and Vertex, and research funding from Amgen and Cabaletta Bio. MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – , Robert G. Micheletti, MD, said in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
For patients with HS, “the quality-of-life impact is profound, greater than any other systematically studied dermatologic condition,” said Dr. Micheletti, associate professor of dermatology at the Hospital of the University of Pennsylavnia, and chief of hospital dermatology, and chief of dermatology at Pennsylvania Hospital, Philadelphia.
Two key aspects of quality of life that affect HS patients are sexual health and overall pain, he said. The female-to-male ratio of HS is approximately 3:1, and data show that approximately 40% of female HS patients experience fertility issues and have unaddressed questions about HS and pregnancy, said Dr. Micheletti. Additionally, data from a systematic review showed that 50%-60% of patients with HS reported sexual dysfunction. Impaired sexual function is also associated with both overall impaired quality of life ratings and the presence of mood disorders, he noted.
Pain also has a significant impact on quality of life for HS patients. When these patients present in an emergency department, 70% report severe pain, and approximately 60% receive opioids, said Dr. Micheletti.
Data from a 2021 study showed that HS patients are significantly more likely to receive opioids compared with controls, and also more likely to be diagnosed with opioid use disorder than controls, especially if they are seen by nondermatologists, he noted.
For acute pain, Dr. Micheletti recommended starting with acetaminophen 500 mg every 4 to 6 hours as needed, and topical nonsteroidal anti-inflammatory drugs (NSAIDs). “It still makes sense to do topical care,” said Dr. Micheletti, but he added that he also prescribes medications for anxiety for these patients.
Patients with increased pain severity or refractory disease may benefit from systemic NSAIDs, or intralesional triamcinolone, he noted. Incision and draining of abscesses may provide temporary symptomatic relief, but keep in mind that lesions will recur, he noted.
For the most severe cases, Dr. Micheletti advised adding tramadol as a first-line opioid, or another short-acting opioid for breakthrough pain.
To manage patients with HS who have chronic pain, Dr. Micheletti recommended starting with HS disease–directed therapy, but also screening for pain severity and psychological comorbidities.
His strategies in these cases include nonpharmacological pain management in the form of physical therapy, wound care, and behavioral health. His algorithm for nociceptive pain is NSAIDs with or without acetaminophen; duloxetine or nortriptyline are other options. For neuropathic pain, gabapentin and/or duloxetine are top choices, but pregabalin, venlafaxine, and nortriptyline are on the list as well.
Topical NSAIDs or topical lidocaine may serve as add-ons to systemic therapy in more severe cases, or as first-line therapy for milder chronic pain, Dr. Micheletti noted. Patients who have failed treatment with at least two pharmacologic agents, suffer medically refractory HS with debilitating pain, or use opioids on an ongoing basis should be referred to a pain management specialist, he said.
Don’t forget lifestyle
Although data on the impact of diet on patients with HS are limited, “we know anecdotally that dairy and refined carbohydrates are associated with exacerbations,” said Dr. Micheletti.
In addition, many patients use complementary medicine “and they aren’t always telling us,” he emphasized. Smoking is prevalent among patients with HS, and is a risk factor for the disease in general, and for more severe and refractory disease, he added. Consequently, screening for tobacco smoking is recommended for patients with HS not only because of the impact on disease, but because it is a potentially modifiable cardiovascular risk factor, he explained.
Consider comorbidities
Cardiovascular disease is among several comorbidities associated with HS, said Dr. Micheletti. HS foundations in the United States and Canada recently published evidence-based recommendations for comorbidity screening. The recommendations included screening for 19 specific comorbidities: acne, dissecting cellulitis, pilonidal disease, pyoderma gangrenosum, depression, anxiety, suicide, smoking, substance abuse, polycystic ovary syndrome, obesity, dyslipidemia, diabetes mellitus, metabolic syndrome, hypertension, cardiovascular disease, inflammatory bowel disease, spondyloarthritis, and sexual dysfunction.
Dr. Micheletti highlighted cardiovascular comorbidities, and noted the association between HS and modifiable cardiovascular risk factors: smoking, obesity, diabetes mellitus, and dyslipidemia. “HS is also independently associated with cardiovascular disease leading to myocardial infarction, stroke, cardiovascular-associated death, and all-cause mortality compared to controls,” he said. Studies show an incidence rate ratio of 1.53 for major adverse cardiovascular events in patients with HS compared with controls, with the highest relative risk among those aged 18-29 years, he added.
Medical management
Depending on the patient, medical management of HS may involve antibiotics, hormonal agents, and biologics, said Dr. Micheletti. Some of the most commonly used antibiotic regimens for HS are those recommended in treatment guidelines, including doxycycline and a clindamycin/rifampin combination, he said. However, the use of trimethoprim-sulfamethoxazole or ciprofloxacin has been associated with increased antibiotic resistance and is not supported by available evidence, he noted.
Hormonal therapies may help some women with HS, said Dr. Micheletti. Options include spironolactone, metformin, or estrogen-containing hormonal contraceptives, he said.
When it comes to biologics, only 33% of HS patients meet criteria for their use (Hurley stage II or III, moderate or severe HS), he noted. However, research suggests “a huge gap” in the use of anti-TNF therapy even among patients for whom it is recommended, he said.
Of the TNF-alpha inhibitors, data on adalimumab, which is FDA-approved for HS, are the most recent. Adalimumab “is our gold standard biologic and our gateway biologic, for HS at this time,” Dr. Micheletti said.
However, those who respond to adalimumab “can continue to do better, but they can wax and wane and flare,” he cautioned. Infliximab, while not approved for HS, has been studied in patients with HS and is prescribed by some providers. Although no comparative studies have been done for infliximab versus adalimumab, “anecdotally, response to infliximab tends to be better, and it is the most effective biologic in common use for severe HS,” he noted.
Dr. Micheletti’s top treatment recommendations for using biologics start with considering biosimilars. Most patients on biosimilars do fine, but some patients who previously responded to infliximab will unpredictably lose efficacy or have reactions when switched to a biosimilar, he said.
Patients on biologics also may experience waning efficacy in the wake of an immune response stimulated by foreign antibodies, said Dr. Micheletti. “Anti-drug antibody formation is more likely to occur when treatment is interrupted,” he noted. Minimize the risk of antibody formation by paying attention to adherence issues and dosing frequency, he advised.
If patients fail both adalimumab and infliximab, Dr. Micheletti tells them not to lose hope, and that treatment is a trial-and-error process that may involve more than one therapy. Other biologics in active use for HS include ustekinumab, anakinra, secukinumab, brodalumab, golimumab, and JAK inhibitors, any of which might be effective in any given patient, he said.
Surgical solutions
For HS patients with chronic, recurring inflammation and drainage associated with a sinus tract, surgical deroofing may the best treatment option, Dr. Micheletti said. “Deroofing involves the use of a probe to trace the extent of the subcutaneous tract, followed by incision and removal of the tract ‘roof,’ ’’ he explained. The deroofing procedure involves local anesthesia and has a low morbidity rate, as well as a low recurrence rate and high levels of patient satisfaction, he said.
“The acute role for surgery is to remove active foci of inflammation and relieve pain,” which is achieved more effectively with deroofing, said Dr. Micheletti. By contrast, incision and drainage is associated with an almost 100% recurrence rate, he added.
When planning elective surgery for HS, Dr. Micheletti noted that holding infliximab for less than 4 weeks does not affect postoperative infection rates in patients with rheumatoid arthritis, and a recent randomized, controlled trial showed that adalimumab can be continued safely through HS surgeries.
In fact, “continuing TNF inhibitors through elective surgery does not increase infection risk and results in better disease control,” and dermatologists should work with surgery to balance infection and disease flare concerns in HS patients, he said.
Dr. Micheletti disclosed serving as a consultant or advisor for Adaptimmune and Vertex, and research funding from Amgen and Cabaletta Bio. MedscapeLive and this news organization are owned by the same parent company.
AT INNOVATIONS IN DERMATOLOGY
Women docs: How your next job contract can reflect your real goals
Rebecca Chester, MD, an Arizona-based interventional cardiologist, recently left her position in a private practice and started employment at a hospital system.
“When I was negotiating my previous contract with the private practice, I found that navigating contracts from the standpoint of a woman still in childbearing years was a little disappointing and challenging,” Dr. Chester told this news organization.
“I wanted to have more children and hired a lawyer recommended by a male colleague to help me not only understand the contract but also negotiate time off and maternity leave, but the lawyer discouraged me from advocating for maternity leave, feeling that it might stigmatize me and prevent me from getting a job,” she says.
He also didn’t explain very much. “He just said it falls under ‘disability leave’ and left it at that.”
Fortunately, Dr. Chester had a good experience with the group. “As things turned out, I did have a child later that year, and they treated me well – I actually got time off – and they didn’t make me take extra call. But it might have turned out very differently because I didn’t know what I was getting into. If I hadn’t worked for such a conscientious group, I might have been in a much tougher situation.”
Since then, Dr. Chester has spoken to female colleagues who received “more support from their legal advisors regarding maternity leave.” She suggests turning to female physicians for recommendations to a lawyer.
Although the central components of a contract (for example, noncompete covenants, malpractice “tail” coverage, bonus structure, vacation time, disability, and call) are relevant to physicians of all genders, the needs of women and men are often different.
Dennis Hursh, managing partner of Physician Agreements Health Law, a Pennsylvania-based law firm that represents physicians, told this news organization that women physicians have “several issues that need special attention when negotiating their physician employment agreements.”
It starts with the interview
“Women have to be sensitive to the interviewer’s casual ‘let’s-get-to-know-each-other’ types of questions that may seem natural but really are unlawful to bring into an employment interview,” said Mr. Hursh.
He warned women to beware of questions such as “Are you married? Do you have kids? Are you planning to start a family?” These may be friendly chit-chat for male interviewees but there may be other agendas when asked to a prospective female employee.
Many of Mr. Hursh’s female clients have been asked this type of question, which “should be regarded as a ‘red flag.’ Yes, it may be an innocent, well-intentioned ice-breaker, but it’s actually unlawful to bring that up in an employment setting and, according to the Equal Opportunity Commission, can be seen as a form of discrimination.” He advises female physicians not to engage with the question and simply to refocus the discussion.
Know your worth and go for it
Medscape’s Physician Compensation surveys have consistently found discrepancies in earnings between male and female physicians, both in primary care and in specialties. In 2022, male primary care physicians earned 23% more than their female counterparts, whereas male specialists earned 31% more.
One reason may be that women tend to be more timid about negotiating for better compensation packages. Amanda Hill of Hill Health Law, a health care practice based in Austin, Tex., told this news organization that in her experience one of the most “overarching” features of female physicians is “that they either don’t know what they’re worth or they undersell themselves.”
In contrast to men, “many women are afraid of coming across as greedy or crass, or even demanding or bossy. But it’s a misperception that if you ask for more money, your future employer will hate you or won’t hire you,” said Ms. Hill.
Ms. Hill and Mr. Hursh encourage physicians to find out what they’re worth, which varies by region and specialty, by consulting benchmarks provided by companies such as Medical Group Management Association.
Jon Appino, MBA, principal and founder of Contract Diagnostics, a Kansas City–based consulting company that specializes in physician employment contract reviews, told this news organization that it’s important to look beyond the salary at other aspects of the position. For example, some figures “don’t take into account how much call a physician is taking. You may know what the average ob.gyn. is making, but an ob.gyn. may be working 3 days a week, while another one is working 6 days a week, one may be on call 15 times per month and another may be on call 15 times a quarter.” Other components of compensation include relative value unit (RVU) thresholds and bonuses.
Once you have that information, “don’t be intimidated, even if you’re sitting in front of several executives who are savvy about negotiations, and don’t worry about coming across as ‘high-maintenance’ or ‘all about money,’ ” Mr. Appino says. Proceed with confidence, knowing your worth and pursuing it.
Part-time vs. full-time
Mr. Appino has seen “more female than male physicians who want to work less than full-time. So it’s important to clarify whether that’s a possibility now or in the future and to understand the implications of working part-time.”
He explained that a full-time employee typically puts in a 40-hour work week, which translates into 1.0 Full-Time Equivalent (FTE), or one unit of work. “For example, if a person wants to work 0.8 FTEs – 4 days a week – is vacation time pro-rated? At what point is there a medical insurance fall-off or a higher monthly premium?”
In medical settings, FTEs may be tied to different metrics rather than the number of weekly hours – for example, for a hospitalist, it might be a certain number of shifts and might also vary by specialty. And it affects the call schedule too. “Call is hard to pro-rate. Many hospital bylaws mandate that call be divided equally, but if one surgeon is working 1.0 FTEs and another is working 0.8 FTEs, how does that call schedule get divided?”
Maternity leave: A tricky question
Many attorneys counsel against raising the question out of fear of scaring away potential employers.
“On the one hand, it is and should be absolutely reasonable to ask about the maternity leave policy or even negotiate for paid leave or additional leave, but it also highlights that you’re planning to have a baby and be out for months,” said Ms. Hill.
“And as much as we want people to be fair and reasonable, on the side of the employer, bias still very much exists, especially in a situation where revenue is based on group numbers. So suddenly, the employer thinks up some ‘nondiscriminatory’ reason why that person isn’t a great fit for the organization.”
Andrew Knoll, MD, JD, a former hospitalist who is now a partner with Cohen Compagni Beckman Appler & Knoll PLLC in Syracuse, N.Y., said that maternity leave is “rare” in an employment agreement, except sometimes in small private practice groups, because it often falls under the purview of “disability leave,” and “from a legal perspective, it’s no different than any other type of disability leave.”
The Family and Medical Leave Act (FMLA), which applies if a group is large enough, allows employees 12 weeks of unpaid leave, during which time their job is protected and their benefits maintained. And some states require employers to offer paid family leave.
“During this time, the woman can take time off – albeit without pay sometimes – to bond with the baby,” Dr. Knoll says. “Since there are statutory laws that protect the employee’s job, offering specific paid maternity leave is very unlikely.”
Ms. Hill advises carefully examining the employer’s comprehensive benefits plan to ascertain if paid maternity leave is included in the benefits. “But unless you’re currently pregnant and want to start off the relationship with true transparency – ‘I’m due in April and curious how we can handle that if you hire me now’ – I would keep the family planning questions to yourself before you get the job.”
Mr. Hursh, author of “The Final Hurdle: A Physician’s Guide to Negotiating a Fair Employment Agreement” (Charleston, S.C.: Advantage, 2012), has a different perspective. “I think all women, no matter how old they are, should ask about maternity leave, whether or not they’re planning a family,” he said.
“The employer may say, ‘We treat maternity leave like any other disability; our policy is such-and-such.’ If they cite an unacceptable policy, it’s a red flag about how they treat women, and should give a woman pause before accepting a position at that organization. Even if your rights are protected under the law and the organization’s policy is violating the law, no one wants to go to battle with HR or to have to go to court.”
Do you want partnership?
Not all female physicians entering a private practice want to advance to becoming partners. Many who are balancing family and work commitments “would prefer to just go into the office, perform their clinical responsibilities, go home, and be done” without the extra headaches, tasks, and time involved with business leadership, says Mr. Appino.
Some private practices have different contracts for those on partnership vs. nonpartnership tracks, so “you should ascertain this information and make sure it’s not automatically assumed that you would like to be on a partnership track,” says Mr. Appino.
On the other hand, you may want to become a partner. “I always suggest asking about the possibility of obtaining a leadership position in an organization or becoming an owner in a private practice,” says Mr. Hursh. “You may be told, ‘We’ve never had a woman in leadership before.’ This might be innocent if you’re the first woman hired, but it might be a red flag as to how women are regarded.” Either way, it’s important to have the information and know what your options are.
The impact of shift schedule
Mr. Hursh advises drilling down into the specifics of the schedule if you’re considering becoming a hospitalist. “Does a ‘week-on/week-off’ shift schedule assume you’ll be taking your vacation time or completing your CME requirements during the week off? This is important for all physicians, but especially for women who might want to use their weeks off to attend to children and family.”
Moreover, “there should be limits on shifts. You shouldn’t have a full day shift followed by a night shift. And there should be a limit on the number of shifts you work without time off. Twelve would be a brutal schedule. Seven is a reasonable amount. Make sure this is in writing and that the contract protects you. Don’t allow the employer to say, ‘We expect you to do the work we assign’ and leave it vague.”
Often, hospitalists will receive an annual salary under the assumption that a certain minimum number of shifts will be completed, but there is no maximum. “It’s important that the salary includes the minimum number of shifts, but that a compensation structure is created so that additional shifts receive additional compensation,” Mr. Hursh said.
Removing the ‘golden handcuffs’
Ms. Hill observes that there are some “really terrible contracts out there, which physicians – especially women – often feel pressured into signing.” They’re told, “This is our standard contract. You won’t find anything better.” Or, “Don’t worry about the small print and legalese.” The physician “gets scared or is artificially reassured, signs an overwhelmingly unfair contract, and then feels stuck.”
Being stuck in a bad contract “is debilitating and adds to burnout, feeling of depression, and the sense that there is no recourse and nowhere to go, especially if your family depends on you,” Ms. Hill said.
“More women than men feel hamstrung or are resigned to being harassed – which is not uncommon in the medical setting, especially in surgical specialties – or just accept poor treatment,” Ms. Hill added. Yes, you can “fight the system and go to HR, but fighting the system is very hard.”
She urges women “not to feel stuck or imprisoned by the ‘golden handcuffs’ but to consult a good lawyer, even if you have to break the contract.” Be aware of the reasons for your unhappiness and bring them to your lawyer – perhaps the system has engaged in fraud, perhaps there has been sexual or racial harassment, perhaps the organization hasn’t followed its own compliance policies.
Dr. Chester consulted Ms. Hill before signing the contract for her current position. “I wanted someone who could give me personalized advice, not only generic advice, and who understood my needs as a woman.”
Ms. Hill helped her to understand “what was and wasn’t fair and reasonable, what changes I could request based on my goals and whether they were realistic, and how to pick my battles. For example, I tried to negotiate tail coverage up front in my previous job but was unsuccessful. The new employer paid tail for me, both from my previous employment and for my current employment.”
Dr. Chester advises other female physicians “never to sign anything without having a lawyer review it and to make sure that the lawyer is sensitive to their specific needs.”
It can be hard to be a female physician. Having the right knowledge and ammunition and knowing how to negotiate well paves the way for success and thriving in an often male-dominated market.
A version of this article first appeared on Medscape.com.
Rebecca Chester, MD, an Arizona-based interventional cardiologist, recently left her position in a private practice and started employment at a hospital system.
“When I was negotiating my previous contract with the private practice, I found that navigating contracts from the standpoint of a woman still in childbearing years was a little disappointing and challenging,” Dr. Chester told this news organization.
“I wanted to have more children and hired a lawyer recommended by a male colleague to help me not only understand the contract but also negotiate time off and maternity leave, but the lawyer discouraged me from advocating for maternity leave, feeling that it might stigmatize me and prevent me from getting a job,” she says.
He also didn’t explain very much. “He just said it falls under ‘disability leave’ and left it at that.”
Fortunately, Dr. Chester had a good experience with the group. “As things turned out, I did have a child later that year, and they treated me well – I actually got time off – and they didn’t make me take extra call. But it might have turned out very differently because I didn’t know what I was getting into. If I hadn’t worked for such a conscientious group, I might have been in a much tougher situation.”
Since then, Dr. Chester has spoken to female colleagues who received “more support from their legal advisors regarding maternity leave.” She suggests turning to female physicians for recommendations to a lawyer.
Although the central components of a contract (for example, noncompete covenants, malpractice “tail” coverage, bonus structure, vacation time, disability, and call) are relevant to physicians of all genders, the needs of women and men are often different.
Dennis Hursh, managing partner of Physician Agreements Health Law, a Pennsylvania-based law firm that represents physicians, told this news organization that women physicians have “several issues that need special attention when negotiating their physician employment agreements.”
It starts with the interview
“Women have to be sensitive to the interviewer’s casual ‘let’s-get-to-know-each-other’ types of questions that may seem natural but really are unlawful to bring into an employment interview,” said Mr. Hursh.
He warned women to beware of questions such as “Are you married? Do you have kids? Are you planning to start a family?” These may be friendly chit-chat for male interviewees but there may be other agendas when asked to a prospective female employee.
Many of Mr. Hursh’s female clients have been asked this type of question, which “should be regarded as a ‘red flag.’ Yes, it may be an innocent, well-intentioned ice-breaker, but it’s actually unlawful to bring that up in an employment setting and, according to the Equal Opportunity Commission, can be seen as a form of discrimination.” He advises female physicians not to engage with the question and simply to refocus the discussion.
Know your worth and go for it
Medscape’s Physician Compensation surveys have consistently found discrepancies in earnings between male and female physicians, both in primary care and in specialties. In 2022, male primary care physicians earned 23% more than their female counterparts, whereas male specialists earned 31% more.
One reason may be that women tend to be more timid about negotiating for better compensation packages. Amanda Hill of Hill Health Law, a health care practice based in Austin, Tex., told this news organization that in her experience one of the most “overarching” features of female physicians is “that they either don’t know what they’re worth or they undersell themselves.”
In contrast to men, “many women are afraid of coming across as greedy or crass, or even demanding or bossy. But it’s a misperception that if you ask for more money, your future employer will hate you or won’t hire you,” said Ms. Hill.
Ms. Hill and Mr. Hursh encourage physicians to find out what they’re worth, which varies by region and specialty, by consulting benchmarks provided by companies such as Medical Group Management Association.
Jon Appino, MBA, principal and founder of Contract Diagnostics, a Kansas City–based consulting company that specializes in physician employment contract reviews, told this news organization that it’s important to look beyond the salary at other aspects of the position. For example, some figures “don’t take into account how much call a physician is taking. You may know what the average ob.gyn. is making, but an ob.gyn. may be working 3 days a week, while another one is working 6 days a week, one may be on call 15 times per month and another may be on call 15 times a quarter.” Other components of compensation include relative value unit (RVU) thresholds and bonuses.
Once you have that information, “don’t be intimidated, even if you’re sitting in front of several executives who are savvy about negotiations, and don’t worry about coming across as ‘high-maintenance’ or ‘all about money,’ ” Mr. Appino says. Proceed with confidence, knowing your worth and pursuing it.
Part-time vs. full-time
Mr. Appino has seen “more female than male physicians who want to work less than full-time. So it’s important to clarify whether that’s a possibility now or in the future and to understand the implications of working part-time.”
He explained that a full-time employee typically puts in a 40-hour work week, which translates into 1.0 Full-Time Equivalent (FTE), or one unit of work. “For example, if a person wants to work 0.8 FTEs – 4 days a week – is vacation time pro-rated? At what point is there a medical insurance fall-off or a higher monthly premium?”
In medical settings, FTEs may be tied to different metrics rather than the number of weekly hours – for example, for a hospitalist, it might be a certain number of shifts and might also vary by specialty. And it affects the call schedule too. “Call is hard to pro-rate. Many hospital bylaws mandate that call be divided equally, but if one surgeon is working 1.0 FTEs and another is working 0.8 FTEs, how does that call schedule get divided?”
Maternity leave: A tricky question
Many attorneys counsel against raising the question out of fear of scaring away potential employers.
“On the one hand, it is and should be absolutely reasonable to ask about the maternity leave policy or even negotiate for paid leave or additional leave, but it also highlights that you’re planning to have a baby and be out for months,” said Ms. Hill.
“And as much as we want people to be fair and reasonable, on the side of the employer, bias still very much exists, especially in a situation where revenue is based on group numbers. So suddenly, the employer thinks up some ‘nondiscriminatory’ reason why that person isn’t a great fit for the organization.”
Andrew Knoll, MD, JD, a former hospitalist who is now a partner with Cohen Compagni Beckman Appler & Knoll PLLC in Syracuse, N.Y., said that maternity leave is “rare” in an employment agreement, except sometimes in small private practice groups, because it often falls under the purview of “disability leave,” and “from a legal perspective, it’s no different than any other type of disability leave.”
The Family and Medical Leave Act (FMLA), which applies if a group is large enough, allows employees 12 weeks of unpaid leave, during which time their job is protected and their benefits maintained. And some states require employers to offer paid family leave.
“During this time, the woman can take time off – albeit without pay sometimes – to bond with the baby,” Dr. Knoll says. “Since there are statutory laws that protect the employee’s job, offering specific paid maternity leave is very unlikely.”
Ms. Hill advises carefully examining the employer’s comprehensive benefits plan to ascertain if paid maternity leave is included in the benefits. “But unless you’re currently pregnant and want to start off the relationship with true transparency – ‘I’m due in April and curious how we can handle that if you hire me now’ – I would keep the family planning questions to yourself before you get the job.”
Mr. Hursh, author of “The Final Hurdle: A Physician’s Guide to Negotiating a Fair Employment Agreement” (Charleston, S.C.: Advantage, 2012), has a different perspective. “I think all women, no matter how old they are, should ask about maternity leave, whether or not they’re planning a family,” he said.
“The employer may say, ‘We treat maternity leave like any other disability; our policy is such-and-such.’ If they cite an unacceptable policy, it’s a red flag about how they treat women, and should give a woman pause before accepting a position at that organization. Even if your rights are protected under the law and the organization’s policy is violating the law, no one wants to go to battle with HR or to have to go to court.”
Do you want partnership?
Not all female physicians entering a private practice want to advance to becoming partners. Many who are balancing family and work commitments “would prefer to just go into the office, perform their clinical responsibilities, go home, and be done” without the extra headaches, tasks, and time involved with business leadership, says Mr. Appino.
Some private practices have different contracts for those on partnership vs. nonpartnership tracks, so “you should ascertain this information and make sure it’s not automatically assumed that you would like to be on a partnership track,” says Mr. Appino.
On the other hand, you may want to become a partner. “I always suggest asking about the possibility of obtaining a leadership position in an organization or becoming an owner in a private practice,” says Mr. Hursh. “You may be told, ‘We’ve never had a woman in leadership before.’ This might be innocent if you’re the first woman hired, but it might be a red flag as to how women are regarded.” Either way, it’s important to have the information and know what your options are.
The impact of shift schedule
Mr. Hursh advises drilling down into the specifics of the schedule if you’re considering becoming a hospitalist. “Does a ‘week-on/week-off’ shift schedule assume you’ll be taking your vacation time or completing your CME requirements during the week off? This is important for all physicians, but especially for women who might want to use their weeks off to attend to children and family.”
Moreover, “there should be limits on shifts. You shouldn’t have a full day shift followed by a night shift. And there should be a limit on the number of shifts you work without time off. Twelve would be a brutal schedule. Seven is a reasonable amount. Make sure this is in writing and that the contract protects you. Don’t allow the employer to say, ‘We expect you to do the work we assign’ and leave it vague.”
Often, hospitalists will receive an annual salary under the assumption that a certain minimum number of shifts will be completed, but there is no maximum. “It’s important that the salary includes the minimum number of shifts, but that a compensation structure is created so that additional shifts receive additional compensation,” Mr. Hursh said.
Removing the ‘golden handcuffs’
Ms. Hill observes that there are some “really terrible contracts out there, which physicians – especially women – often feel pressured into signing.” They’re told, “This is our standard contract. You won’t find anything better.” Or, “Don’t worry about the small print and legalese.” The physician “gets scared or is artificially reassured, signs an overwhelmingly unfair contract, and then feels stuck.”
Being stuck in a bad contract “is debilitating and adds to burnout, feeling of depression, and the sense that there is no recourse and nowhere to go, especially if your family depends on you,” Ms. Hill said.
“More women than men feel hamstrung or are resigned to being harassed – which is not uncommon in the medical setting, especially in surgical specialties – or just accept poor treatment,” Ms. Hill added. Yes, you can “fight the system and go to HR, but fighting the system is very hard.”
She urges women “not to feel stuck or imprisoned by the ‘golden handcuffs’ but to consult a good lawyer, even if you have to break the contract.” Be aware of the reasons for your unhappiness and bring them to your lawyer – perhaps the system has engaged in fraud, perhaps there has been sexual or racial harassment, perhaps the organization hasn’t followed its own compliance policies.
Dr. Chester consulted Ms. Hill before signing the contract for her current position. “I wanted someone who could give me personalized advice, not only generic advice, and who understood my needs as a woman.”
Ms. Hill helped her to understand “what was and wasn’t fair and reasonable, what changes I could request based on my goals and whether they were realistic, and how to pick my battles. For example, I tried to negotiate tail coverage up front in my previous job but was unsuccessful. The new employer paid tail for me, both from my previous employment and for my current employment.”
Dr. Chester advises other female physicians “never to sign anything without having a lawyer review it and to make sure that the lawyer is sensitive to their specific needs.”
It can be hard to be a female physician. Having the right knowledge and ammunition and knowing how to negotiate well paves the way for success and thriving in an often male-dominated market.
A version of this article first appeared on Medscape.com.
Rebecca Chester, MD, an Arizona-based interventional cardiologist, recently left her position in a private practice and started employment at a hospital system.
“When I was negotiating my previous contract with the private practice, I found that navigating contracts from the standpoint of a woman still in childbearing years was a little disappointing and challenging,” Dr. Chester told this news organization.
“I wanted to have more children and hired a lawyer recommended by a male colleague to help me not only understand the contract but also negotiate time off and maternity leave, but the lawyer discouraged me from advocating for maternity leave, feeling that it might stigmatize me and prevent me from getting a job,” she says.
He also didn’t explain very much. “He just said it falls under ‘disability leave’ and left it at that.”
Fortunately, Dr. Chester had a good experience with the group. “As things turned out, I did have a child later that year, and they treated me well – I actually got time off – and they didn’t make me take extra call. But it might have turned out very differently because I didn’t know what I was getting into. If I hadn’t worked for such a conscientious group, I might have been in a much tougher situation.”
Since then, Dr. Chester has spoken to female colleagues who received “more support from their legal advisors regarding maternity leave.” She suggests turning to female physicians for recommendations to a lawyer.
Although the central components of a contract (for example, noncompete covenants, malpractice “tail” coverage, bonus structure, vacation time, disability, and call) are relevant to physicians of all genders, the needs of women and men are often different.
Dennis Hursh, managing partner of Physician Agreements Health Law, a Pennsylvania-based law firm that represents physicians, told this news organization that women physicians have “several issues that need special attention when negotiating their physician employment agreements.”
It starts with the interview
“Women have to be sensitive to the interviewer’s casual ‘let’s-get-to-know-each-other’ types of questions that may seem natural but really are unlawful to bring into an employment interview,” said Mr. Hursh.
He warned women to beware of questions such as “Are you married? Do you have kids? Are you planning to start a family?” These may be friendly chit-chat for male interviewees but there may be other agendas when asked to a prospective female employee.
Many of Mr. Hursh’s female clients have been asked this type of question, which “should be regarded as a ‘red flag.’ Yes, it may be an innocent, well-intentioned ice-breaker, but it’s actually unlawful to bring that up in an employment setting and, according to the Equal Opportunity Commission, can be seen as a form of discrimination.” He advises female physicians not to engage with the question and simply to refocus the discussion.
Know your worth and go for it
Medscape’s Physician Compensation surveys have consistently found discrepancies in earnings between male and female physicians, both in primary care and in specialties. In 2022, male primary care physicians earned 23% more than their female counterparts, whereas male specialists earned 31% more.
One reason may be that women tend to be more timid about negotiating for better compensation packages. Amanda Hill of Hill Health Law, a health care practice based in Austin, Tex., told this news organization that in her experience one of the most “overarching” features of female physicians is “that they either don’t know what they’re worth or they undersell themselves.”
In contrast to men, “many women are afraid of coming across as greedy or crass, or even demanding or bossy. But it’s a misperception that if you ask for more money, your future employer will hate you or won’t hire you,” said Ms. Hill.
Ms. Hill and Mr. Hursh encourage physicians to find out what they’re worth, which varies by region and specialty, by consulting benchmarks provided by companies such as Medical Group Management Association.
Jon Appino, MBA, principal and founder of Contract Diagnostics, a Kansas City–based consulting company that specializes in physician employment contract reviews, told this news organization that it’s important to look beyond the salary at other aspects of the position. For example, some figures “don’t take into account how much call a physician is taking. You may know what the average ob.gyn. is making, but an ob.gyn. may be working 3 days a week, while another one is working 6 days a week, one may be on call 15 times per month and another may be on call 15 times a quarter.” Other components of compensation include relative value unit (RVU) thresholds and bonuses.
Once you have that information, “don’t be intimidated, even if you’re sitting in front of several executives who are savvy about negotiations, and don’t worry about coming across as ‘high-maintenance’ or ‘all about money,’ ” Mr. Appino says. Proceed with confidence, knowing your worth and pursuing it.
Part-time vs. full-time
Mr. Appino has seen “more female than male physicians who want to work less than full-time. So it’s important to clarify whether that’s a possibility now or in the future and to understand the implications of working part-time.”
He explained that a full-time employee typically puts in a 40-hour work week, which translates into 1.0 Full-Time Equivalent (FTE), or one unit of work. “For example, if a person wants to work 0.8 FTEs – 4 days a week – is vacation time pro-rated? At what point is there a medical insurance fall-off or a higher monthly premium?”
In medical settings, FTEs may be tied to different metrics rather than the number of weekly hours – for example, for a hospitalist, it might be a certain number of shifts and might also vary by specialty. And it affects the call schedule too. “Call is hard to pro-rate. Many hospital bylaws mandate that call be divided equally, but if one surgeon is working 1.0 FTEs and another is working 0.8 FTEs, how does that call schedule get divided?”
Maternity leave: A tricky question
Many attorneys counsel against raising the question out of fear of scaring away potential employers.
“On the one hand, it is and should be absolutely reasonable to ask about the maternity leave policy or even negotiate for paid leave or additional leave, but it also highlights that you’re planning to have a baby and be out for months,” said Ms. Hill.
“And as much as we want people to be fair and reasonable, on the side of the employer, bias still very much exists, especially in a situation where revenue is based on group numbers. So suddenly, the employer thinks up some ‘nondiscriminatory’ reason why that person isn’t a great fit for the organization.”
Andrew Knoll, MD, JD, a former hospitalist who is now a partner with Cohen Compagni Beckman Appler & Knoll PLLC in Syracuse, N.Y., said that maternity leave is “rare” in an employment agreement, except sometimes in small private practice groups, because it often falls under the purview of “disability leave,” and “from a legal perspective, it’s no different than any other type of disability leave.”
The Family and Medical Leave Act (FMLA), which applies if a group is large enough, allows employees 12 weeks of unpaid leave, during which time their job is protected and their benefits maintained. And some states require employers to offer paid family leave.
“During this time, the woman can take time off – albeit without pay sometimes – to bond with the baby,” Dr. Knoll says. “Since there are statutory laws that protect the employee’s job, offering specific paid maternity leave is very unlikely.”
Ms. Hill advises carefully examining the employer’s comprehensive benefits plan to ascertain if paid maternity leave is included in the benefits. “But unless you’re currently pregnant and want to start off the relationship with true transparency – ‘I’m due in April and curious how we can handle that if you hire me now’ – I would keep the family planning questions to yourself before you get the job.”
Mr. Hursh, author of “The Final Hurdle: A Physician’s Guide to Negotiating a Fair Employment Agreement” (Charleston, S.C.: Advantage, 2012), has a different perspective. “I think all women, no matter how old they are, should ask about maternity leave, whether or not they’re planning a family,” he said.
“The employer may say, ‘We treat maternity leave like any other disability; our policy is such-and-such.’ If they cite an unacceptable policy, it’s a red flag about how they treat women, and should give a woman pause before accepting a position at that organization. Even if your rights are protected under the law and the organization’s policy is violating the law, no one wants to go to battle with HR or to have to go to court.”
Do you want partnership?
Not all female physicians entering a private practice want to advance to becoming partners. Many who are balancing family and work commitments “would prefer to just go into the office, perform their clinical responsibilities, go home, and be done” without the extra headaches, tasks, and time involved with business leadership, says Mr. Appino.
Some private practices have different contracts for those on partnership vs. nonpartnership tracks, so “you should ascertain this information and make sure it’s not automatically assumed that you would like to be on a partnership track,” says Mr. Appino.
On the other hand, you may want to become a partner. “I always suggest asking about the possibility of obtaining a leadership position in an organization or becoming an owner in a private practice,” says Mr. Hursh. “You may be told, ‘We’ve never had a woman in leadership before.’ This might be innocent if you’re the first woman hired, but it might be a red flag as to how women are regarded.” Either way, it’s important to have the information and know what your options are.
The impact of shift schedule
Mr. Hursh advises drilling down into the specifics of the schedule if you’re considering becoming a hospitalist. “Does a ‘week-on/week-off’ shift schedule assume you’ll be taking your vacation time or completing your CME requirements during the week off? This is important for all physicians, but especially for women who might want to use their weeks off to attend to children and family.”
Moreover, “there should be limits on shifts. You shouldn’t have a full day shift followed by a night shift. And there should be a limit on the number of shifts you work without time off. Twelve would be a brutal schedule. Seven is a reasonable amount. Make sure this is in writing and that the contract protects you. Don’t allow the employer to say, ‘We expect you to do the work we assign’ and leave it vague.”
Often, hospitalists will receive an annual salary under the assumption that a certain minimum number of shifts will be completed, but there is no maximum. “It’s important that the salary includes the minimum number of shifts, but that a compensation structure is created so that additional shifts receive additional compensation,” Mr. Hursh said.
Removing the ‘golden handcuffs’
Ms. Hill observes that there are some “really terrible contracts out there, which physicians – especially women – often feel pressured into signing.” They’re told, “This is our standard contract. You won’t find anything better.” Or, “Don’t worry about the small print and legalese.” The physician “gets scared or is artificially reassured, signs an overwhelmingly unfair contract, and then feels stuck.”
Being stuck in a bad contract “is debilitating and adds to burnout, feeling of depression, and the sense that there is no recourse and nowhere to go, especially if your family depends on you,” Ms. Hill said.
“More women than men feel hamstrung or are resigned to being harassed – which is not uncommon in the medical setting, especially in surgical specialties – or just accept poor treatment,” Ms. Hill added. Yes, you can “fight the system and go to HR, but fighting the system is very hard.”
She urges women “not to feel stuck or imprisoned by the ‘golden handcuffs’ but to consult a good lawyer, even if you have to break the contract.” Be aware of the reasons for your unhappiness and bring them to your lawyer – perhaps the system has engaged in fraud, perhaps there has been sexual or racial harassment, perhaps the organization hasn’t followed its own compliance policies.
Dr. Chester consulted Ms. Hill before signing the contract for her current position. “I wanted someone who could give me personalized advice, not only generic advice, and who understood my needs as a woman.”
Ms. Hill helped her to understand “what was and wasn’t fair and reasonable, what changes I could request based on my goals and whether they were realistic, and how to pick my battles. For example, I tried to negotiate tail coverage up front in my previous job but was unsuccessful. The new employer paid tail for me, both from my previous employment and for my current employment.”
Dr. Chester advises other female physicians “never to sign anything without having a lawyer review it and to make sure that the lawyer is sensitive to their specific needs.”
It can be hard to be a female physician. Having the right knowledge and ammunition and knowing how to negotiate well paves the way for success and thriving in an often male-dominated market.
A version of this article first appeared on Medscape.com.
Shorter fever prevention duration effective after cardiac arrest
a randomized trial shows.
“Since 2005, active fever prevention in comatose patients has been advocated by the guidelines for 72 hours after an out-of-hospital cardiac arrest,” Christian Hassager, MD, of the University of Copenhagen, told this news organization. “Our study is the first randomized trial ever on this subject – and it challenges the guidelines.”
At 90 days, a primary endpoint – a composite of death from any cause or hospital discharge with a high Cerebral Performance Category score – occurred in 32.4% of those in the 36-hour group and 33.6% of those in the 72-hour group; mortality was 29.5% versus 30.3%, respectively.
The study was published online in The New England Journal of Medicine. The results were also presented at the Resuscitation Science Symposium during the American Heart Association scientific sessions.
No significant differences
Assessment of the two device-based fever-prevention strategies for the duration was a predefined, additional randomly assigned open-label intervention in the Blood Pressure and Oxygenation Targets in Post Resuscitation Care (BOX) trial, which involved comatose adult patients who had been resuscitated after out-of-hospital cardiac arrest at two Danish cardiac arrest centers.
The main BOX analysis compared different primary strategies in these patients in a two-by-two factorial design: higher versus lower blood pressure targets and higher versus lower oxygenation targets. They found no difference between the various strategies in terms of death and discharge from hospital in a poor neurologic state. Those results were presented at the European Society of Cardiology Congress on Aug. 27, and simultaneously published in separate articles in The New England Journal of Medicine.
For this current analysis, a total of 789 comatose patients (mean age, 62; 80% men) received device-based temperature control targeting 36° C for 24 hours followed by 37° C for either 12 or 48 hours (total intervention times, 36 and 72 hours, respectively) or until the patient regained consciousness.
Patients were kept sedated and were receiving mechanical ventilation during the temperature control at 36° C, the authors note. Target core body temperature was controlled using commercially available surface cooling at one of the sites in 286 patients (Criticool and Allon, Belmont Medical Technologies) and using intravenous cooling in 503 patients at the other site (Thermogard XP, and Cool Line Catheter, Zoll).
Body temperature was maintained at 37° C with the same type of device that had been used for 36° C during the initial 24 hours. If the patient awakened, cooling was terminated.
Physicians in both groups were permitted to use non–device-based fever treatment (that is, for a body temperature > 37.5° C) with drugs such as paracetamol, by uncovering the patient’s body, or both, at the discretion of the treating physician. Ice packs or pads were not used.
The primary outcome was a composite of death from any cause or hospital discharge with a Cerebral Performance Category of 3 or 4 (range, 1 to 5, with higher scores indicating more severe disability) within 90 days after randomization.
Secondary outcomes at 90 days included death from any cause and the Montreal Cognitive Assessment score (range, 0 to 30, with higher scores indicating better cognitive ability).
A primary endpoint event occurred in 32.3% of patients in the 36-hour group and in 33.6% of those in the 72-hour group (hazard ratio, 0.99). Mortality was 29.5% in the 36-hour group and 30.3% in the 72-hour group.
The median Montreal Cognitive Assessment scores were 26 and 27, respectively. No significant between-group differences in the incidence of adverse events were observed.
The authors concluded that “active device-based fever prevention for 36 or 72 hours after cardiac arrest did not result in significantly different percentages of patients dying or having severe disability or coma.”
Dr. Hassager added, “We will continue with a new trial where we will randomize to treatment as usual or immediate wakeup call and no temperature intervention at all.”
Findings ‘very persuasive’
Intensivist Ken Parhar, MD, clinical associate professor, Critical Care Medicine at the University of Calgary (Alta.) and Alberta Health Services, Edmonton, and medical director, Cardiovascular Intensive Care Unit, commented on the study.
“The findings are very clear and very persuasive,” he said. “I think this should be incorporated into future guidelines, though it would be nice to see the trial repeated in another center.”
Dr. Parhar has kept comatose patients under temperature control for less than 72 hours, but mainly because those patients started to wake up. “This study provides clarity on the safety of that process – that we don’t have to unnecessarily keep somebody sedated just for an arbitrary timeline,” he said. “Beyond 36 hours, we need to continue to use our judgment.”
The study was supported by a grant from the Novo Nordisk Foundation, as was the work of one of the coauthors. Dr. Hassager’s work was funded by a grant from the Lundbeck Foundation; he also received an individual research grant from the Novo Nordisk Foundation, as well as honoraria from ABIOMED. No other disclosures were declared.
A version of this article first appeared on Medscape.com.
a randomized trial shows.
“Since 2005, active fever prevention in comatose patients has been advocated by the guidelines for 72 hours after an out-of-hospital cardiac arrest,” Christian Hassager, MD, of the University of Copenhagen, told this news organization. “Our study is the first randomized trial ever on this subject – and it challenges the guidelines.”
At 90 days, a primary endpoint – a composite of death from any cause or hospital discharge with a high Cerebral Performance Category score – occurred in 32.4% of those in the 36-hour group and 33.6% of those in the 72-hour group; mortality was 29.5% versus 30.3%, respectively.
The study was published online in The New England Journal of Medicine. The results were also presented at the Resuscitation Science Symposium during the American Heart Association scientific sessions.
No significant differences
Assessment of the two device-based fever-prevention strategies for the duration was a predefined, additional randomly assigned open-label intervention in the Blood Pressure and Oxygenation Targets in Post Resuscitation Care (BOX) trial, which involved comatose adult patients who had been resuscitated after out-of-hospital cardiac arrest at two Danish cardiac arrest centers.
The main BOX analysis compared different primary strategies in these patients in a two-by-two factorial design: higher versus lower blood pressure targets and higher versus lower oxygenation targets. They found no difference between the various strategies in terms of death and discharge from hospital in a poor neurologic state. Those results were presented at the European Society of Cardiology Congress on Aug. 27, and simultaneously published in separate articles in The New England Journal of Medicine.
For this current analysis, a total of 789 comatose patients (mean age, 62; 80% men) received device-based temperature control targeting 36° C for 24 hours followed by 37° C for either 12 or 48 hours (total intervention times, 36 and 72 hours, respectively) or until the patient regained consciousness.
Patients were kept sedated and were receiving mechanical ventilation during the temperature control at 36° C, the authors note. Target core body temperature was controlled using commercially available surface cooling at one of the sites in 286 patients (Criticool and Allon, Belmont Medical Technologies) and using intravenous cooling in 503 patients at the other site (Thermogard XP, and Cool Line Catheter, Zoll).
Body temperature was maintained at 37° C with the same type of device that had been used for 36° C during the initial 24 hours. If the patient awakened, cooling was terminated.
Physicians in both groups were permitted to use non–device-based fever treatment (that is, for a body temperature > 37.5° C) with drugs such as paracetamol, by uncovering the patient’s body, or both, at the discretion of the treating physician. Ice packs or pads were not used.
The primary outcome was a composite of death from any cause or hospital discharge with a Cerebral Performance Category of 3 or 4 (range, 1 to 5, with higher scores indicating more severe disability) within 90 days after randomization.
Secondary outcomes at 90 days included death from any cause and the Montreal Cognitive Assessment score (range, 0 to 30, with higher scores indicating better cognitive ability).
A primary endpoint event occurred in 32.3% of patients in the 36-hour group and in 33.6% of those in the 72-hour group (hazard ratio, 0.99). Mortality was 29.5% in the 36-hour group and 30.3% in the 72-hour group.
The median Montreal Cognitive Assessment scores were 26 and 27, respectively. No significant between-group differences in the incidence of adverse events were observed.
The authors concluded that “active device-based fever prevention for 36 or 72 hours after cardiac arrest did not result in significantly different percentages of patients dying or having severe disability or coma.”
Dr. Hassager added, “We will continue with a new trial where we will randomize to treatment as usual or immediate wakeup call and no temperature intervention at all.”
Findings ‘very persuasive’
Intensivist Ken Parhar, MD, clinical associate professor, Critical Care Medicine at the University of Calgary (Alta.) and Alberta Health Services, Edmonton, and medical director, Cardiovascular Intensive Care Unit, commented on the study.
“The findings are very clear and very persuasive,” he said. “I think this should be incorporated into future guidelines, though it would be nice to see the trial repeated in another center.”
Dr. Parhar has kept comatose patients under temperature control for less than 72 hours, but mainly because those patients started to wake up. “This study provides clarity on the safety of that process – that we don’t have to unnecessarily keep somebody sedated just for an arbitrary timeline,” he said. “Beyond 36 hours, we need to continue to use our judgment.”
The study was supported by a grant from the Novo Nordisk Foundation, as was the work of one of the coauthors. Dr. Hassager’s work was funded by a grant from the Lundbeck Foundation; he also received an individual research grant from the Novo Nordisk Foundation, as well as honoraria from ABIOMED. No other disclosures were declared.
A version of this article first appeared on Medscape.com.
a randomized trial shows.
“Since 2005, active fever prevention in comatose patients has been advocated by the guidelines for 72 hours after an out-of-hospital cardiac arrest,” Christian Hassager, MD, of the University of Copenhagen, told this news organization. “Our study is the first randomized trial ever on this subject – and it challenges the guidelines.”
At 90 days, a primary endpoint – a composite of death from any cause or hospital discharge with a high Cerebral Performance Category score – occurred in 32.4% of those in the 36-hour group and 33.6% of those in the 72-hour group; mortality was 29.5% versus 30.3%, respectively.
The study was published online in The New England Journal of Medicine. The results were also presented at the Resuscitation Science Symposium during the American Heart Association scientific sessions.
No significant differences
Assessment of the two device-based fever-prevention strategies for the duration was a predefined, additional randomly assigned open-label intervention in the Blood Pressure and Oxygenation Targets in Post Resuscitation Care (BOX) trial, which involved comatose adult patients who had been resuscitated after out-of-hospital cardiac arrest at two Danish cardiac arrest centers.
The main BOX analysis compared different primary strategies in these patients in a two-by-two factorial design: higher versus lower blood pressure targets and higher versus lower oxygenation targets. They found no difference between the various strategies in terms of death and discharge from hospital in a poor neurologic state. Those results were presented at the European Society of Cardiology Congress on Aug. 27, and simultaneously published in separate articles in The New England Journal of Medicine.
For this current analysis, a total of 789 comatose patients (mean age, 62; 80% men) received device-based temperature control targeting 36° C for 24 hours followed by 37° C for either 12 or 48 hours (total intervention times, 36 and 72 hours, respectively) or until the patient regained consciousness.
Patients were kept sedated and were receiving mechanical ventilation during the temperature control at 36° C, the authors note. Target core body temperature was controlled using commercially available surface cooling at one of the sites in 286 patients (Criticool and Allon, Belmont Medical Technologies) and using intravenous cooling in 503 patients at the other site (Thermogard XP, and Cool Line Catheter, Zoll).
Body temperature was maintained at 37° C with the same type of device that had been used for 36° C during the initial 24 hours. If the patient awakened, cooling was terminated.
Physicians in both groups were permitted to use non–device-based fever treatment (that is, for a body temperature > 37.5° C) with drugs such as paracetamol, by uncovering the patient’s body, or both, at the discretion of the treating physician. Ice packs or pads were not used.
The primary outcome was a composite of death from any cause or hospital discharge with a Cerebral Performance Category of 3 or 4 (range, 1 to 5, with higher scores indicating more severe disability) within 90 days after randomization.
Secondary outcomes at 90 days included death from any cause and the Montreal Cognitive Assessment score (range, 0 to 30, with higher scores indicating better cognitive ability).
A primary endpoint event occurred in 32.3% of patients in the 36-hour group and in 33.6% of those in the 72-hour group (hazard ratio, 0.99). Mortality was 29.5% in the 36-hour group and 30.3% in the 72-hour group.
The median Montreal Cognitive Assessment scores were 26 and 27, respectively. No significant between-group differences in the incidence of adverse events were observed.
The authors concluded that “active device-based fever prevention for 36 or 72 hours after cardiac arrest did not result in significantly different percentages of patients dying or having severe disability or coma.”
Dr. Hassager added, “We will continue with a new trial where we will randomize to treatment as usual or immediate wakeup call and no temperature intervention at all.”
Findings ‘very persuasive’
Intensivist Ken Parhar, MD, clinical associate professor, Critical Care Medicine at the University of Calgary (Alta.) and Alberta Health Services, Edmonton, and medical director, Cardiovascular Intensive Care Unit, commented on the study.
“The findings are very clear and very persuasive,” he said. “I think this should be incorporated into future guidelines, though it would be nice to see the trial repeated in another center.”
Dr. Parhar has kept comatose patients under temperature control for less than 72 hours, but mainly because those patients started to wake up. “This study provides clarity on the safety of that process – that we don’t have to unnecessarily keep somebody sedated just for an arbitrary timeline,” he said. “Beyond 36 hours, we need to continue to use our judgment.”
The study was supported by a grant from the Novo Nordisk Foundation, as was the work of one of the coauthors. Dr. Hassager’s work was funded by a grant from the Lundbeck Foundation; he also received an individual research grant from the Novo Nordisk Foundation, as well as honoraria from ABIOMED. No other disclosures were declared.
A version of this article first appeared on Medscape.com.
FROM NEJM
How blunt is too blunt for informed consent?
Sitting across from a patient explaining a complicated treatment proposal, protocol, or medication may be one of the most complex yet crucial tasks you have as a physician. Although informed consent is at the forefront of shared decisions between you and your patient, there’s a fine line between providing enough information on the risks and benefits of a particular treatment and knowing you’ve explained it well enough to fully educate your patient about their choices.
“It is a bit of a fine line because unless your patient happens to be a health care provider, medicine is complicated for patients to understand,” said David L. Feldman, MD, chief medical officer at The Doctors Company, the nation’s largest medical malpractice insurer in New York.
In addition, documenting the interaction is critical, said James Giordano, PhD, MPhil, professor in the departments of neurology and biochemistry and chief of the neuroethics studies program at the Pellegrino Center for Clinical Bioethics at Georgetown University Medical Center, Washington.
“As with anything in medicine, the key rule is that if it’s not documented, it’s not done,” he said. “This also means diligent documentation in all aspects of the medical record, including the electronic medical record and the written one.”
That said, it’s important to know what’s enough and what’s too granular when you discuss a procedure with your patients, said Erum N. Ilyas, MD, a board-certified dermatologist at Schweiger Dermatology and a bioethicist near Philadelphia.
“One of the most challenging aspects of informed consent, especially for young physicians, is how to discuss a procedure or a medication in a manner that is both relevant and concise,” Dr. llyas said. “I’ve had residents about to perform a skin biopsy spend several minutes covering every aspect of every potential outcome of a routine skin biopsy. The patient is left traumatized and confused as to whether they should proceed with the small procedure.”
Instead, the goal of informed consent is to ensure that the patient has a general overview of the procedure and is empowered, knowing that the decision to proceed is, indeed, part of their decision-making process.
How long an informed consent discussion takes depends on the procedure.
“When I was in practice as a plastic surgeon, the conversations varied from the straightforward ‘I’m taking this mole off your cheek, and there’s a risk of scarring and bleeding’ to talking about a mastectomy and breast reconstruction, which could take an hour or more to discuss,” Dr. Feldman said.
Ultimately, it’s as essential for doctors to explain the risks associated with a procedure as it is for patients to understand precisely what’s involved, Dr. Ilyas added.
She also recommends creating a flow to the conversation that places the discussion of risks within the context of why the procedure is being performed. This way, clarity about both the risks and the need for the treatment or procedure can be achieved.
When doing so, it’s critical to make sure you’re speaking your patient’s language – literally.
“Have a translator in the room if needed,” Dr. Feldman added. “If your patient is hearing or sight impaired, you need to have every contingency ready to ensure that everyone is in complete communication.”
Document, document, document!
To best protect yourself, the patient must consent to each procedure and intervention via active, informed consent, said Dr. Giordano.
“It’s not enough to hand a patient a piece of paper and say sign it,” he said. “There should be some documented evidence that the patient has not only read the document but that the key parts of the document have been explained and that the patient’s level of comprehension has been assessed and verified.”
It is vital if the patient has a disability, a neurological impairment, or a neurocognitive or psychiatric condition that might impede his or her ability to understand the consent that’s being sought.
In addition, it’s best if a ‘clinical proxy’ handles the consent (for example, a nurse, office worker, or case manager).
“This can be very helpful because it means you’ve had third-party documentation of informed consent,” Dr. Giordano said. “It should then be re-documented with you as the clinician and stated that the patient has affirmatively and actively agreed to treatment.”
What happens when things go wrong?
If you’re sued over informed consent, with the patient claiming that you didn’t fully explain the potential risks, the first thing to consider is why this happened.
“Very often, these situations occur if there was some difficulty or competency of communication,” Dr. Giordano said. “You may have done everything right, but somehow the patient hasn’t gained an understanding of the procedure required.”
Physicians must take a hard look at how they’re explaining risks and possible side effects. For doctors who perform these procedures regularly, the risks may seem small, and they may unconsciously minimize them to the patient. But when something goes wrong, the patient may then feel that they didn’t fully understand the frequency of poor outcomes, or the potential severity.
Next, it’s important to perform a ‘gap analysis’ to assess why something went awry. That means, look at all the potential factors involved to identify which one was the weak link.
“It might be that the patient was on a signing frenzy and signed away but didn’t receive active and informed content,” Dr. Giordano said. “The goal is to learn how to close the gap for this case and for future cases.”
To protect yourself, consider using technology to your advantage, especially since lawsuits over informed consent usually happen several years after the procedure. This is when a patient might argue that you didn’t tell them about possible complications and that they might have opted out of the procedure if they had known about those issues ahead of time.
“Even before the statute of limitations is up for a lawsuit, it could be five years from the time the procedure occurred due to the length of time a lawsuit can take,” Dr. Feldman said. “That’s why it’s important to take a video of your conversation or make a recording of the informed consent conversation. This way if there’s a question of what you said, there’s a video of it.”
For many physicians, this would be a big change – to video record and then store all their informed consent conversations. It could most likely help you if a lawsuit occurs, but some physicians may feel that process to be cumbersome and time-consuming, and they’d rather find another way to ensure that patients understand the risks.
Ultimately, however, if there’s a legal question involved with informed consent, the general thinking is that the effect on the patient must be harmful for it to stand up.
“The question becomes whether the outcome rendered that gap in the consenting process forgivable,” Dr. Giordano said. “The hope is that there was nothing harmful to the patient and that the benefit of the procedure was demonstrable despite any gaps in the informed consent process.”
In the end, informed consent should be a matter of good communication before, during, and after any treatment or procedure.
“When you form a relationship with a patient who needs any procedure, small or large, you’re going to be guiding them through a very scary thing,” Dr. Feldman said. “You want to make patients feel like you care about them and that, while neither you nor the system is perfect, you’ll take care of them. That’s the bottom line.”
A version of this article first appeared on Medscape.com.
Sitting across from a patient explaining a complicated treatment proposal, protocol, or medication may be one of the most complex yet crucial tasks you have as a physician. Although informed consent is at the forefront of shared decisions between you and your patient, there’s a fine line between providing enough information on the risks and benefits of a particular treatment and knowing you’ve explained it well enough to fully educate your patient about their choices.
“It is a bit of a fine line because unless your patient happens to be a health care provider, medicine is complicated for patients to understand,” said David L. Feldman, MD, chief medical officer at The Doctors Company, the nation’s largest medical malpractice insurer in New York.
In addition, documenting the interaction is critical, said James Giordano, PhD, MPhil, professor in the departments of neurology and biochemistry and chief of the neuroethics studies program at the Pellegrino Center for Clinical Bioethics at Georgetown University Medical Center, Washington.
“As with anything in medicine, the key rule is that if it’s not documented, it’s not done,” he said. “This also means diligent documentation in all aspects of the medical record, including the electronic medical record and the written one.”
That said, it’s important to know what’s enough and what’s too granular when you discuss a procedure with your patients, said Erum N. Ilyas, MD, a board-certified dermatologist at Schweiger Dermatology and a bioethicist near Philadelphia.
“One of the most challenging aspects of informed consent, especially for young physicians, is how to discuss a procedure or a medication in a manner that is both relevant and concise,” Dr. llyas said. “I’ve had residents about to perform a skin biopsy spend several minutes covering every aspect of every potential outcome of a routine skin biopsy. The patient is left traumatized and confused as to whether they should proceed with the small procedure.”
Instead, the goal of informed consent is to ensure that the patient has a general overview of the procedure and is empowered, knowing that the decision to proceed is, indeed, part of their decision-making process.
How long an informed consent discussion takes depends on the procedure.
“When I was in practice as a plastic surgeon, the conversations varied from the straightforward ‘I’m taking this mole off your cheek, and there’s a risk of scarring and bleeding’ to talking about a mastectomy and breast reconstruction, which could take an hour or more to discuss,” Dr. Feldman said.
Ultimately, it’s as essential for doctors to explain the risks associated with a procedure as it is for patients to understand precisely what’s involved, Dr. Ilyas added.
She also recommends creating a flow to the conversation that places the discussion of risks within the context of why the procedure is being performed. This way, clarity about both the risks and the need for the treatment or procedure can be achieved.
When doing so, it’s critical to make sure you’re speaking your patient’s language – literally.
“Have a translator in the room if needed,” Dr. Feldman added. “If your patient is hearing or sight impaired, you need to have every contingency ready to ensure that everyone is in complete communication.”
Document, document, document!
To best protect yourself, the patient must consent to each procedure and intervention via active, informed consent, said Dr. Giordano.
“It’s not enough to hand a patient a piece of paper and say sign it,” he said. “There should be some documented evidence that the patient has not only read the document but that the key parts of the document have been explained and that the patient’s level of comprehension has been assessed and verified.”
It is vital if the patient has a disability, a neurological impairment, or a neurocognitive or psychiatric condition that might impede his or her ability to understand the consent that’s being sought.
In addition, it’s best if a ‘clinical proxy’ handles the consent (for example, a nurse, office worker, or case manager).
“This can be very helpful because it means you’ve had third-party documentation of informed consent,” Dr. Giordano said. “It should then be re-documented with you as the clinician and stated that the patient has affirmatively and actively agreed to treatment.”
What happens when things go wrong?
If you’re sued over informed consent, with the patient claiming that you didn’t fully explain the potential risks, the first thing to consider is why this happened.
“Very often, these situations occur if there was some difficulty or competency of communication,” Dr. Giordano said. “You may have done everything right, but somehow the patient hasn’t gained an understanding of the procedure required.”
Physicians must take a hard look at how they’re explaining risks and possible side effects. For doctors who perform these procedures regularly, the risks may seem small, and they may unconsciously minimize them to the patient. But when something goes wrong, the patient may then feel that they didn’t fully understand the frequency of poor outcomes, or the potential severity.
Next, it’s important to perform a ‘gap analysis’ to assess why something went awry. That means, look at all the potential factors involved to identify which one was the weak link.
“It might be that the patient was on a signing frenzy and signed away but didn’t receive active and informed content,” Dr. Giordano said. “The goal is to learn how to close the gap for this case and for future cases.”
To protect yourself, consider using technology to your advantage, especially since lawsuits over informed consent usually happen several years after the procedure. This is when a patient might argue that you didn’t tell them about possible complications and that they might have opted out of the procedure if they had known about those issues ahead of time.
“Even before the statute of limitations is up for a lawsuit, it could be five years from the time the procedure occurred due to the length of time a lawsuit can take,” Dr. Feldman said. “That’s why it’s important to take a video of your conversation or make a recording of the informed consent conversation. This way if there’s a question of what you said, there’s a video of it.”
For many physicians, this would be a big change – to video record and then store all their informed consent conversations. It could most likely help you if a lawsuit occurs, but some physicians may feel that process to be cumbersome and time-consuming, and they’d rather find another way to ensure that patients understand the risks.
Ultimately, however, if there’s a legal question involved with informed consent, the general thinking is that the effect on the patient must be harmful for it to stand up.
“The question becomes whether the outcome rendered that gap in the consenting process forgivable,” Dr. Giordano said. “The hope is that there was nothing harmful to the patient and that the benefit of the procedure was demonstrable despite any gaps in the informed consent process.”
In the end, informed consent should be a matter of good communication before, during, and after any treatment or procedure.
“When you form a relationship with a patient who needs any procedure, small or large, you’re going to be guiding them through a very scary thing,” Dr. Feldman said. “You want to make patients feel like you care about them and that, while neither you nor the system is perfect, you’ll take care of them. That’s the bottom line.”
A version of this article first appeared on Medscape.com.
Sitting across from a patient explaining a complicated treatment proposal, protocol, or medication may be one of the most complex yet crucial tasks you have as a physician. Although informed consent is at the forefront of shared decisions between you and your patient, there’s a fine line between providing enough information on the risks and benefits of a particular treatment and knowing you’ve explained it well enough to fully educate your patient about their choices.
“It is a bit of a fine line because unless your patient happens to be a health care provider, medicine is complicated for patients to understand,” said David L. Feldman, MD, chief medical officer at The Doctors Company, the nation’s largest medical malpractice insurer in New York.
In addition, documenting the interaction is critical, said James Giordano, PhD, MPhil, professor in the departments of neurology and biochemistry and chief of the neuroethics studies program at the Pellegrino Center for Clinical Bioethics at Georgetown University Medical Center, Washington.
“As with anything in medicine, the key rule is that if it’s not documented, it’s not done,” he said. “This also means diligent documentation in all aspects of the medical record, including the electronic medical record and the written one.”
That said, it’s important to know what’s enough and what’s too granular when you discuss a procedure with your patients, said Erum N. Ilyas, MD, a board-certified dermatologist at Schweiger Dermatology and a bioethicist near Philadelphia.
“One of the most challenging aspects of informed consent, especially for young physicians, is how to discuss a procedure or a medication in a manner that is both relevant and concise,” Dr. llyas said. “I’ve had residents about to perform a skin biopsy spend several minutes covering every aspect of every potential outcome of a routine skin biopsy. The patient is left traumatized and confused as to whether they should proceed with the small procedure.”
Instead, the goal of informed consent is to ensure that the patient has a general overview of the procedure and is empowered, knowing that the decision to proceed is, indeed, part of their decision-making process.
How long an informed consent discussion takes depends on the procedure.
“When I was in practice as a plastic surgeon, the conversations varied from the straightforward ‘I’m taking this mole off your cheek, and there’s a risk of scarring and bleeding’ to talking about a mastectomy and breast reconstruction, which could take an hour or more to discuss,” Dr. Feldman said.
Ultimately, it’s as essential for doctors to explain the risks associated with a procedure as it is for patients to understand precisely what’s involved, Dr. Ilyas added.
She also recommends creating a flow to the conversation that places the discussion of risks within the context of why the procedure is being performed. This way, clarity about both the risks and the need for the treatment or procedure can be achieved.
When doing so, it’s critical to make sure you’re speaking your patient’s language – literally.
“Have a translator in the room if needed,” Dr. Feldman added. “If your patient is hearing or sight impaired, you need to have every contingency ready to ensure that everyone is in complete communication.”
Document, document, document!
To best protect yourself, the patient must consent to each procedure and intervention via active, informed consent, said Dr. Giordano.
“It’s not enough to hand a patient a piece of paper and say sign it,” he said. “There should be some documented evidence that the patient has not only read the document but that the key parts of the document have been explained and that the patient’s level of comprehension has been assessed and verified.”
It is vital if the patient has a disability, a neurological impairment, or a neurocognitive or psychiatric condition that might impede his or her ability to understand the consent that’s being sought.
In addition, it’s best if a ‘clinical proxy’ handles the consent (for example, a nurse, office worker, or case manager).
“This can be very helpful because it means you’ve had third-party documentation of informed consent,” Dr. Giordano said. “It should then be re-documented with you as the clinician and stated that the patient has affirmatively and actively agreed to treatment.”
What happens when things go wrong?
If you’re sued over informed consent, with the patient claiming that you didn’t fully explain the potential risks, the first thing to consider is why this happened.
“Very often, these situations occur if there was some difficulty or competency of communication,” Dr. Giordano said. “You may have done everything right, but somehow the patient hasn’t gained an understanding of the procedure required.”
Physicians must take a hard look at how they’re explaining risks and possible side effects. For doctors who perform these procedures regularly, the risks may seem small, and they may unconsciously minimize them to the patient. But when something goes wrong, the patient may then feel that they didn’t fully understand the frequency of poor outcomes, or the potential severity.
Next, it’s important to perform a ‘gap analysis’ to assess why something went awry. That means, look at all the potential factors involved to identify which one was the weak link.
“It might be that the patient was on a signing frenzy and signed away but didn’t receive active and informed content,” Dr. Giordano said. “The goal is to learn how to close the gap for this case and for future cases.”
To protect yourself, consider using technology to your advantage, especially since lawsuits over informed consent usually happen several years after the procedure. This is when a patient might argue that you didn’t tell them about possible complications and that they might have opted out of the procedure if they had known about those issues ahead of time.
“Even before the statute of limitations is up for a lawsuit, it could be five years from the time the procedure occurred due to the length of time a lawsuit can take,” Dr. Feldman said. “That’s why it’s important to take a video of your conversation or make a recording of the informed consent conversation. This way if there’s a question of what you said, there’s a video of it.”
For many physicians, this would be a big change – to video record and then store all their informed consent conversations. It could most likely help you if a lawsuit occurs, but some physicians may feel that process to be cumbersome and time-consuming, and they’d rather find another way to ensure that patients understand the risks.
Ultimately, however, if there’s a legal question involved with informed consent, the general thinking is that the effect on the patient must be harmful for it to stand up.
“The question becomes whether the outcome rendered that gap in the consenting process forgivable,” Dr. Giordano said. “The hope is that there was nothing harmful to the patient and that the benefit of the procedure was demonstrable despite any gaps in the informed consent process.”
In the end, informed consent should be a matter of good communication before, during, and after any treatment or procedure.
“When you form a relationship with a patient who needs any procedure, small or large, you’re going to be guiding them through a very scary thing,” Dr. Feldman said. “You want to make patients feel like you care about them and that, while neither you nor the system is perfect, you’ll take care of them. That’s the bottom line.”
A version of this article first appeared on Medscape.com.
Analysis of doctors’ EHR email finds infrequent but notable hostility
Among the emails, 43% were from patients; the remainder were mostly from other physicians or clinicians, or automated. The content of the messages wasn’t associated with doctor burnout, as the researchers had hypothesized. And only about 5% of the messages had negative sentiment.
But the researchers were struck by the hostility of that sentiment, displayed in messages like these that surely would be distressing for physicians to read:
“I hope and expect that you will spend eternity in he**. You are an abusive, nasty, cheap person.”
“Your office is full of liars, hypocrites and I will do everything in my power to prevent anyone from going to your bullsh** office again.”
About 5% of emails had an overall negative sentiment, with high-frequency words like “cancel,” “pain,” or “problem.” Among patient messages, 3% were negative and contained words and expletives suggesting hatred, hostility, or violence.
“F***” was the most common expletive used by patients.
Researchers provided examples of profanity-laced messages, including one patient who said, “I am so upset that I was told the blood work would include the gender of the baby. I have been waiting 5 [days] to find it, and it wasn’t even fu**ing tested!!!! What a disappointment in your office and the bullsh** I was told. I will be switching plans because this is sh**!”
Researchers also noted some high-frequency words associated with violence, such as “shoot,” “fight,” and “kill.”
“This is concerning, especially given documentation of patient-inflicted violence against physicians. Health systems should be proactive in ensuring that the in-basket does not become a venue for physician abuse and cyberbullying,” the researchers wrote in JAMA Network Open.
“Posting reminders in EHR patient portals to use kind language when sending messages, applying filters for expletives or threatening words, and creating frameworks for identifying patients who frequently send negative messages are potential strategies for mitigating this risk.”
Using a form of artificial intelligence technology called natural language processing (NLP), researchers at the University of California, San Diego, analyzed the characteristics of more than 1.4 million emails received by the university’s physicians, 43% of them from patients. They specifically looked at the volume of messages, word count, and overall sentiment.
Whereas other studies have examined the growing burden of EHR messaging for doctors, this type of email sentiment analysis could help in creating solutions. Researchers say that one such solution could involve applying filters for expletives or threatening words. It also could help identify fixable health system issues that make patients so angry, the researchers say.
Among the emails from physicians to physicians, just over half reported burnout, which correlated to the following phrases: “I am beginning to burn out and have one or more symptoms of burnout” and “I feel completely burned out [and] am at the point where I may need to seek help.”
On average, physicians who reported burnout received a greater volume of patient messages. The odds of burnout were significantly higher among Hispanic/Latinx physicians and females. Physicians with more than 15 years of clinical practice had markedly lower burnout.
Despite physicians now spending more time on EHR in-basket tasks than they did before the pandemic, the study found no significant associations between message characteristics and burnout.
Data for the cross-sectional study were collected from multiple specialties from April to September 2020. Physicians then completed a survey and assessed their burnout on a 5-point scale. Of the 609 physician responses, approximately 49% of participants were women, 56% were White, and 64% worked in outpatient settings. About 70% of the doctors had been in practice for 15 years or less.
The sentiment score was based on word content as well as the use of negation, punctuation, degree modifiers, all caps, emoticons, emojis, and acronyms. Positive patient messages were more likely to convey gratitude and thanks, along with casual expressions, such as “fyi” and “lol.”
A version of this article first appeared on Medscape.com.
Among the emails, 43% were from patients; the remainder were mostly from other physicians or clinicians, or automated. The content of the messages wasn’t associated with doctor burnout, as the researchers had hypothesized. And only about 5% of the messages had negative sentiment.
But the researchers were struck by the hostility of that sentiment, displayed in messages like these that surely would be distressing for physicians to read:
“I hope and expect that you will spend eternity in he**. You are an abusive, nasty, cheap person.”
“Your office is full of liars, hypocrites and I will do everything in my power to prevent anyone from going to your bullsh** office again.”
About 5% of emails had an overall negative sentiment, with high-frequency words like “cancel,” “pain,” or “problem.” Among patient messages, 3% were negative and contained words and expletives suggesting hatred, hostility, or violence.
“F***” was the most common expletive used by patients.
Researchers provided examples of profanity-laced messages, including one patient who said, “I am so upset that I was told the blood work would include the gender of the baby. I have been waiting 5 [days] to find it, and it wasn’t even fu**ing tested!!!! What a disappointment in your office and the bullsh** I was told. I will be switching plans because this is sh**!”
Researchers also noted some high-frequency words associated with violence, such as “shoot,” “fight,” and “kill.”
“This is concerning, especially given documentation of patient-inflicted violence against physicians. Health systems should be proactive in ensuring that the in-basket does not become a venue for physician abuse and cyberbullying,” the researchers wrote in JAMA Network Open.
“Posting reminders in EHR patient portals to use kind language when sending messages, applying filters for expletives or threatening words, and creating frameworks for identifying patients who frequently send negative messages are potential strategies for mitigating this risk.”
Using a form of artificial intelligence technology called natural language processing (NLP), researchers at the University of California, San Diego, analyzed the characteristics of more than 1.4 million emails received by the university’s physicians, 43% of them from patients. They specifically looked at the volume of messages, word count, and overall sentiment.
Whereas other studies have examined the growing burden of EHR messaging for doctors, this type of email sentiment analysis could help in creating solutions. Researchers say that one such solution could involve applying filters for expletives or threatening words. It also could help identify fixable health system issues that make patients so angry, the researchers say.
Among the emails from physicians to physicians, just over half reported burnout, which correlated to the following phrases: “I am beginning to burn out and have one or more symptoms of burnout” and “I feel completely burned out [and] am at the point where I may need to seek help.”
On average, physicians who reported burnout received a greater volume of patient messages. The odds of burnout were significantly higher among Hispanic/Latinx physicians and females. Physicians with more than 15 years of clinical practice had markedly lower burnout.
Despite physicians now spending more time on EHR in-basket tasks than they did before the pandemic, the study found no significant associations between message characteristics and burnout.
Data for the cross-sectional study were collected from multiple specialties from April to September 2020. Physicians then completed a survey and assessed their burnout on a 5-point scale. Of the 609 physician responses, approximately 49% of participants were women, 56% were White, and 64% worked in outpatient settings. About 70% of the doctors had been in practice for 15 years or less.
The sentiment score was based on word content as well as the use of negation, punctuation, degree modifiers, all caps, emoticons, emojis, and acronyms. Positive patient messages were more likely to convey gratitude and thanks, along with casual expressions, such as “fyi” and “lol.”
A version of this article first appeared on Medscape.com.
Among the emails, 43% were from patients; the remainder were mostly from other physicians or clinicians, or automated. The content of the messages wasn’t associated with doctor burnout, as the researchers had hypothesized. And only about 5% of the messages had negative sentiment.
But the researchers were struck by the hostility of that sentiment, displayed in messages like these that surely would be distressing for physicians to read:
“I hope and expect that you will spend eternity in he**. You are an abusive, nasty, cheap person.”
“Your office is full of liars, hypocrites and I will do everything in my power to prevent anyone from going to your bullsh** office again.”
About 5% of emails had an overall negative sentiment, with high-frequency words like “cancel,” “pain,” or “problem.” Among patient messages, 3% were negative and contained words and expletives suggesting hatred, hostility, or violence.
“F***” was the most common expletive used by patients.
Researchers provided examples of profanity-laced messages, including one patient who said, “I am so upset that I was told the blood work would include the gender of the baby. I have been waiting 5 [days] to find it, and it wasn’t even fu**ing tested!!!! What a disappointment in your office and the bullsh** I was told. I will be switching plans because this is sh**!”
Researchers also noted some high-frequency words associated with violence, such as “shoot,” “fight,” and “kill.”
“This is concerning, especially given documentation of patient-inflicted violence against physicians. Health systems should be proactive in ensuring that the in-basket does not become a venue for physician abuse and cyberbullying,” the researchers wrote in JAMA Network Open.
“Posting reminders in EHR patient portals to use kind language when sending messages, applying filters for expletives or threatening words, and creating frameworks for identifying patients who frequently send negative messages are potential strategies for mitigating this risk.”
Using a form of artificial intelligence technology called natural language processing (NLP), researchers at the University of California, San Diego, analyzed the characteristics of more than 1.4 million emails received by the university’s physicians, 43% of them from patients. They specifically looked at the volume of messages, word count, and overall sentiment.
Whereas other studies have examined the growing burden of EHR messaging for doctors, this type of email sentiment analysis could help in creating solutions. Researchers say that one such solution could involve applying filters for expletives or threatening words. It also could help identify fixable health system issues that make patients so angry, the researchers say.
Among the emails from physicians to physicians, just over half reported burnout, which correlated to the following phrases: “I am beginning to burn out and have one or more symptoms of burnout” and “I feel completely burned out [and] am at the point where I may need to seek help.”
On average, physicians who reported burnout received a greater volume of patient messages. The odds of burnout were significantly higher among Hispanic/Latinx physicians and females. Physicians with more than 15 years of clinical practice had markedly lower burnout.
Despite physicians now spending more time on EHR in-basket tasks than they did before the pandemic, the study found no significant associations between message characteristics and burnout.
Data for the cross-sectional study were collected from multiple specialties from April to September 2020. Physicians then completed a survey and assessed their burnout on a 5-point scale. Of the 609 physician responses, approximately 49% of participants were women, 56% were White, and 64% worked in outpatient settings. About 70% of the doctors had been in practice for 15 years or less.
The sentiment score was based on word content as well as the use of negation, punctuation, degree modifiers, all caps, emoticons, emojis, and acronyms. Positive patient messages were more likely to convey gratitude and thanks, along with casual expressions, such as “fyi” and “lol.”
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN