User login
Clinical Psychiatry News is the online destination and multimedia properties of Clinica Psychiatry News, the independent news publication for psychiatrists. Since 1971, Clinical Psychiatry News has been the leading source of news and commentary about clinical developments in psychiatry as well as health care policy and regulations that affect the physician's practice.
Dear Drupal User: You're seeing this because you're logged in to Drupal, and not redirected to MDedge.com/psychiatry.
Depression
adolescent depression
adolescent major depressive disorder
adolescent schizophrenia
adolescent with major depressive disorder
animals
autism
baby
brexpiprazole
child
child bipolar
child depression
child schizophrenia
children with bipolar disorder
children with depression
children with major depressive disorder
compulsive behaviors
cure
elderly bipolar
elderly depression
elderly major depressive disorder
elderly schizophrenia
elderly with dementia
first break
first episode
gambling
gaming
geriatric depression
geriatric major depressive disorder
geriatric schizophrenia
infant
ketamine
kid
major depressive disorder
major depressive disorder in adolescents
major depressive disorder in children
parenting
pediatric
pediatric bipolar
pediatric depression
pediatric major depressive disorder
pediatric schizophrenia
pregnancy
pregnant
rexulti
skin care
suicide
teen
wine
section[contains(@class, 'nav-hidden')]
footer[@id='footer']
div[contains(@class, 'pane-pub-article-cpn')]
div[contains(@class, 'pane-pub-home-cpn')]
div[contains(@class, 'pane-pub-topic-cpn')]
div[contains(@class, 'panel-panel-inner')]
div[contains(@class, 'pane-node-field-article-topics')]
section[contains(@class, 'footer-nav-section-wrapper')]
Hydroxychloroquine ineffective for COVID-19, VA study suggests
Hydroxychloroquine (HCQ) with or without azithromycin (AZ) is not associated with a lower risk of requiring mechanical ventilation, according to a retrospective study of Veterans Affairs patients hospitalized with COVID-19.
The study, which was posted on a preprint server April 21 and has not been peer reviewed, also showed an increased risk of death associated with COVID-19 patients treated with HCQ alone.
“These findings highlight the importance of awaiting the results of ongoing prospective, randomized controlled studies before widespread adoption of these drugs,” write Joseph Magagnoli with Dorn Research Institute at the Columbia (S.C.) VA Health Care System and the department of clinical pharmacy & outcomes sciences, University of South Carolina, and colleagues.
A spokesperson with the University of Virginia, Charlottesville, where several of coauthors practice, said that the authors declined to comment for this article before peer review is completed.
The new data are not the first to suggest no benefit with HCQ among patients with COVID-19. A randomized trial showed no benefit and more side effects among 75 patients in China treated with HCQ, compared with 75 who received standard of care alone, according to a preprint posted online April 14.
No benefit in ventilation, death rates
The current analysis included data from all 368 male patients hospitalized with confirmed COVID-19 and treated at Veterans Health Administration medical centers in the United States through April 11.
Patients were categorized into three groups: those treated with HCQ in addition to standard of care (n = 97); those treated with HCQ and the antibiotic azithromycin plus standard of care (n = 113); and those who received standard supportive care only (n = 158).
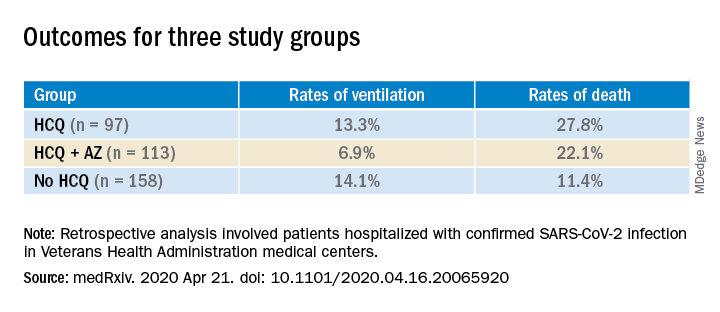
Compared with the no HCQ group, the risk of death from any cause was higher in the HCQ group (adjusted hazard ratio, 2.61; 95% confidence interval, 1.10-6.17; P = .03) but not in the HCQ+AZ group (aHR, 1.14; 95% CI, 0.56-2.32; P = .72).
The risk of ventilation was similar in the HCQ group (aHR, 1.43; 95% CI, 0.53-3.79; P = .48) and in the HCQ+AZ group (aHR, 0.43; 95% CI, 0.16-1.12; P = .09), compared with the no-HCQ group.
This study provides another counterbalance to claims of HCQ efficacy, David R. Wessner, PhD, professor of biology and chair of the department of health and human values at Davidson (N.C.) College, said in an interview.
Interest in HCQ spiked after an open-label, nonrandomized, single-center study of COVID-19 patients in France suggested that hydroxychloroquine helped clear the virus and had a potential enhanced effect when combined with azithromycin.
But the 36-patient trial has since been called into question.
Wait for convincing data
Dr. Wessner, whose research focuses on viral pathogenesis, says that, although the current data don’t definitively answer the question of whether HCQ is effective in treating COVID-19, taking a “let’s try it and see” approach is not reasonable.
“Until we have good, prospective randomized trials, it’s hard to know what to make of this. But this is more evidence that there’s not a good reason to use [HCQ],” Dr. Wessner said. He points out that the small randomized trial from China shows that HCQ comes with potential harms.
Anecdotal evidence is often cited by those who promote HCQ as a potential treatment, but “those are one-off examples,” Wessner continued. “That doesn’t really tell us anything.”
Some HCQ proponents have said that trials finding no benefit are flawed in that the drug is given too late. However, Dr. Wessner says, there’s no way to prove or disprove that claim without randomized controlled trials.
Conflicting messages
Despite lack of clear evidence of benefit for patients with COVID-19, HCQ is recommended off-label by the Chinese National guideline, and the U.S. Food and Drug Administration has issued an emergency-use authorization for the treatment of adult patients with COVID-19.
Conversely, the Infectious Diseases Society of America and a guideline panel convened by the National Institutes of Health each concluded recently that because of insufficient data, they could not recommend any specific treatments for patients with COVID-19.
The VA data for the current study came from the Veterans Affairs Informatics and Computing Infrastructure, which includes inpatient, outpatient and laboratory data and pharmacy claims.
The authors acknowledge some limitations, “including those inherent to all retrospective analyses such as nonrandomization of treatments.”
However, they note that they did adjust for potential confounders, including comorbidities, medications, and clinical and laboratory factors.
A coauthor, Jayakrishna Ambati, MD, is a cofounder of iVeena Holdings, iVeena Delivery Systems and Inflammasome Therapeutics, and has received consultancy fees from Allergan, Biogen, Boehringer Ingelheim, Immunovant, Janssen, Olix Pharmaceuticals, Retinal Solutions, and Saksin LifeSciences, all unrelated to this work. Dr. Ambati is named as an inventor on a patent application filed by the University of Virginia relating to COVID-19 but unrelated to this work. Another coauthor has received research grants from Boehringer Ingelheim, Gilead Sciences, Portola Pharmaceuticals, and United Therapeutics, all unrelated to this work. The other authors and Dr. Wessner have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Hydroxychloroquine (HCQ) with or without azithromycin (AZ) is not associated with a lower risk of requiring mechanical ventilation, according to a retrospective study of Veterans Affairs patients hospitalized with COVID-19.
The study, which was posted on a preprint server April 21 and has not been peer reviewed, also showed an increased risk of death associated with COVID-19 patients treated with HCQ alone.
“These findings highlight the importance of awaiting the results of ongoing prospective, randomized controlled studies before widespread adoption of these drugs,” write Joseph Magagnoli with Dorn Research Institute at the Columbia (S.C.) VA Health Care System and the department of clinical pharmacy & outcomes sciences, University of South Carolina, and colleagues.
A spokesperson with the University of Virginia, Charlottesville, where several of coauthors practice, said that the authors declined to comment for this article before peer review is completed.
The new data are not the first to suggest no benefit with HCQ among patients with COVID-19. A randomized trial showed no benefit and more side effects among 75 patients in China treated with HCQ, compared with 75 who received standard of care alone, according to a preprint posted online April 14.
No benefit in ventilation, death rates
The current analysis included data from all 368 male patients hospitalized with confirmed COVID-19 and treated at Veterans Health Administration medical centers in the United States through April 11.
Patients were categorized into three groups: those treated with HCQ in addition to standard of care (n = 97); those treated with HCQ and the antibiotic azithromycin plus standard of care (n = 113); and those who received standard supportive care only (n = 158).

Compared with the no HCQ group, the risk of death from any cause was higher in the HCQ group (adjusted hazard ratio, 2.61; 95% confidence interval, 1.10-6.17; P = .03) but not in the HCQ+AZ group (aHR, 1.14; 95% CI, 0.56-2.32; P = .72).
The risk of ventilation was similar in the HCQ group (aHR, 1.43; 95% CI, 0.53-3.79; P = .48) and in the HCQ+AZ group (aHR, 0.43; 95% CI, 0.16-1.12; P = .09), compared with the no-HCQ group.
This study provides another counterbalance to claims of HCQ efficacy, David R. Wessner, PhD, professor of biology and chair of the department of health and human values at Davidson (N.C.) College, said in an interview.
Interest in HCQ spiked after an open-label, nonrandomized, single-center study of COVID-19 patients in France suggested that hydroxychloroquine helped clear the virus and had a potential enhanced effect when combined with azithromycin.
But the 36-patient trial has since been called into question.
Wait for convincing data
Dr. Wessner, whose research focuses on viral pathogenesis, says that, although the current data don’t definitively answer the question of whether HCQ is effective in treating COVID-19, taking a “let’s try it and see” approach is not reasonable.
“Until we have good, prospective randomized trials, it’s hard to know what to make of this. But this is more evidence that there’s not a good reason to use [HCQ],” Dr. Wessner said. He points out that the small randomized trial from China shows that HCQ comes with potential harms.
Anecdotal evidence is often cited by those who promote HCQ as a potential treatment, but “those are one-off examples,” Wessner continued. “That doesn’t really tell us anything.”
Some HCQ proponents have said that trials finding no benefit are flawed in that the drug is given too late. However, Dr. Wessner says, there’s no way to prove or disprove that claim without randomized controlled trials.
Conflicting messages
Despite lack of clear evidence of benefit for patients with COVID-19, HCQ is recommended off-label by the Chinese National guideline, and the U.S. Food and Drug Administration has issued an emergency-use authorization for the treatment of adult patients with COVID-19.
Conversely, the Infectious Diseases Society of America and a guideline panel convened by the National Institutes of Health each concluded recently that because of insufficient data, they could not recommend any specific treatments for patients with COVID-19.
The VA data for the current study came from the Veterans Affairs Informatics and Computing Infrastructure, which includes inpatient, outpatient and laboratory data and pharmacy claims.
The authors acknowledge some limitations, “including those inherent to all retrospective analyses such as nonrandomization of treatments.”
However, they note that they did adjust for potential confounders, including comorbidities, medications, and clinical and laboratory factors.
A coauthor, Jayakrishna Ambati, MD, is a cofounder of iVeena Holdings, iVeena Delivery Systems and Inflammasome Therapeutics, and has received consultancy fees from Allergan, Biogen, Boehringer Ingelheim, Immunovant, Janssen, Olix Pharmaceuticals, Retinal Solutions, and Saksin LifeSciences, all unrelated to this work. Dr. Ambati is named as an inventor on a patent application filed by the University of Virginia relating to COVID-19 but unrelated to this work. Another coauthor has received research grants from Boehringer Ingelheim, Gilead Sciences, Portola Pharmaceuticals, and United Therapeutics, all unrelated to this work. The other authors and Dr. Wessner have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Hydroxychloroquine (HCQ) with or without azithromycin (AZ) is not associated with a lower risk of requiring mechanical ventilation, according to a retrospective study of Veterans Affairs patients hospitalized with COVID-19.
The study, which was posted on a preprint server April 21 and has not been peer reviewed, also showed an increased risk of death associated with COVID-19 patients treated with HCQ alone.
“These findings highlight the importance of awaiting the results of ongoing prospective, randomized controlled studies before widespread adoption of these drugs,” write Joseph Magagnoli with Dorn Research Institute at the Columbia (S.C.) VA Health Care System and the department of clinical pharmacy & outcomes sciences, University of South Carolina, and colleagues.
A spokesperson with the University of Virginia, Charlottesville, where several of coauthors practice, said that the authors declined to comment for this article before peer review is completed.
The new data are not the first to suggest no benefit with HCQ among patients with COVID-19. A randomized trial showed no benefit and more side effects among 75 patients in China treated with HCQ, compared with 75 who received standard of care alone, according to a preprint posted online April 14.
No benefit in ventilation, death rates
The current analysis included data from all 368 male patients hospitalized with confirmed COVID-19 and treated at Veterans Health Administration medical centers in the United States through April 11.
Patients were categorized into three groups: those treated with HCQ in addition to standard of care (n = 97); those treated with HCQ and the antibiotic azithromycin plus standard of care (n = 113); and those who received standard supportive care only (n = 158).

Compared with the no HCQ group, the risk of death from any cause was higher in the HCQ group (adjusted hazard ratio, 2.61; 95% confidence interval, 1.10-6.17; P = .03) but not in the HCQ+AZ group (aHR, 1.14; 95% CI, 0.56-2.32; P = .72).
The risk of ventilation was similar in the HCQ group (aHR, 1.43; 95% CI, 0.53-3.79; P = .48) and in the HCQ+AZ group (aHR, 0.43; 95% CI, 0.16-1.12; P = .09), compared with the no-HCQ group.
This study provides another counterbalance to claims of HCQ efficacy, David R. Wessner, PhD, professor of biology and chair of the department of health and human values at Davidson (N.C.) College, said in an interview.
Interest in HCQ spiked after an open-label, nonrandomized, single-center study of COVID-19 patients in France suggested that hydroxychloroquine helped clear the virus and had a potential enhanced effect when combined with azithromycin.
But the 36-patient trial has since been called into question.
Wait for convincing data
Dr. Wessner, whose research focuses on viral pathogenesis, says that, although the current data don’t definitively answer the question of whether HCQ is effective in treating COVID-19, taking a “let’s try it and see” approach is not reasonable.
“Until we have good, prospective randomized trials, it’s hard to know what to make of this. But this is more evidence that there’s not a good reason to use [HCQ],” Dr. Wessner said. He points out that the small randomized trial from China shows that HCQ comes with potential harms.
Anecdotal evidence is often cited by those who promote HCQ as a potential treatment, but “those are one-off examples,” Wessner continued. “That doesn’t really tell us anything.”
Some HCQ proponents have said that trials finding no benefit are flawed in that the drug is given too late. However, Dr. Wessner says, there’s no way to prove or disprove that claim without randomized controlled trials.
Conflicting messages
Despite lack of clear evidence of benefit for patients with COVID-19, HCQ is recommended off-label by the Chinese National guideline, and the U.S. Food and Drug Administration has issued an emergency-use authorization for the treatment of adult patients with COVID-19.
Conversely, the Infectious Diseases Society of America and a guideline panel convened by the National Institutes of Health each concluded recently that because of insufficient data, they could not recommend any specific treatments for patients with COVID-19.
The VA data for the current study came from the Veterans Affairs Informatics and Computing Infrastructure, which includes inpatient, outpatient and laboratory data and pharmacy claims.
The authors acknowledge some limitations, “including those inherent to all retrospective analyses such as nonrandomization of treatments.”
However, they note that they did adjust for potential confounders, including comorbidities, medications, and clinical and laboratory factors.
A coauthor, Jayakrishna Ambati, MD, is a cofounder of iVeena Holdings, iVeena Delivery Systems and Inflammasome Therapeutics, and has received consultancy fees from Allergan, Biogen, Boehringer Ingelheim, Immunovant, Janssen, Olix Pharmaceuticals, Retinal Solutions, and Saksin LifeSciences, all unrelated to this work. Dr. Ambati is named as an inventor on a patent application filed by the University of Virginia relating to COVID-19 but unrelated to this work. Another coauthor has received research grants from Boehringer Ingelheim, Gilead Sciences, Portola Pharmaceuticals, and United Therapeutics, all unrelated to this work. The other authors and Dr. Wessner have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Prioritizing ambulatory gynecology care during COVID-19: The latest guidance
What exactly constitutes appropriate ambulatory gynecology during this time of social distancing?
On March 30, 2020, the American College of Obstetricians and Gynecologists (ACOG) weighed in, releasing COVID-19 FAQs for Obstetrician-Gynecologists. These recommendations, which include information about obstetric and gynecologic surgery, are available to everyone, including the general public. They are intended to supplement guidance from the Centers for Disease Control and Prevention, as well as previously released ACOG guidance.
The recommendations include examples of patients needing in-person appointments, telehealth visits, or visits that should be deferred.
In-person appointments. Examples of patients for whom in-person appointments are appropriate include those with suspected ectopic pregnancy or profuse vaginal bleeding. With respect to contraceptive services, ACOG suggests that placement of IUDs and implants should continue whenever possible. If placement of the contraceptive device is deferred, use of self-administered hormonal contraceptives (including subcutaneous injections, oral, transdermal patch, and vaginal ring) should be encouraged as a bridge to later initiation of long-acting methods.
Telehealth visits. Video or telephone visits are advised for women desiring counseling and prescribing for contraception or menopausal symptoms.
Deferred. Deferral of office visits until after COVID-19 lockdowns is advised for average-risk women wishing routine well-woman visits. Other situations in which deferral should be considered include the following:
- For patients with abnormal cervical cancer screening results, ACOG suggests that colposcopy with cervical biopsies could be deferred for 6-12 months for patients with low-grade test results. In contrast, for patients with high-grade results, ACOG recommends that evaluation be performed within 3 months.
- For women who wish to discontinue their contraceptive, ACOG advises that removal of IUDs and implants be postponed when possible. These women should be counseled regarding extended use of these devices.
ACOG emphasizes that decisions regarding ambulatory gynecology should be individualized and take into consideration such issues as availability of local and regional resources, staffing, personal protective equipment, and the local prevalence of COVID-19.
As a gynecologist focused on ambulatory care, I believe that many clinicians will welcome this guidance from ACOG, which helps us provide optimal care during these challenging times.
Dr. Kaunitz is professor and associate chairman in the department of obstetrics and gynecology at the University of Florida, Jacksonville. He has disclosed receiving royalties from UpToDate, serving on the safety monitoring board for Femasys, and serving as a consultant for AMAG Pharmaceuticals, Merck & Co, Mithra, and Pfizer. His institution has received funding from pharmaceutical companies and nonprofits.
A version of this article originally appeared on Medscape.com.
What exactly constitutes appropriate ambulatory gynecology during this time of social distancing?
On March 30, 2020, the American College of Obstetricians and Gynecologists (ACOG) weighed in, releasing COVID-19 FAQs for Obstetrician-Gynecologists. These recommendations, which include information about obstetric and gynecologic surgery, are available to everyone, including the general public. They are intended to supplement guidance from the Centers for Disease Control and Prevention, as well as previously released ACOG guidance.
The recommendations include examples of patients needing in-person appointments, telehealth visits, or visits that should be deferred.
In-person appointments. Examples of patients for whom in-person appointments are appropriate include those with suspected ectopic pregnancy or profuse vaginal bleeding. With respect to contraceptive services, ACOG suggests that placement of IUDs and implants should continue whenever possible. If placement of the contraceptive device is deferred, use of self-administered hormonal contraceptives (including subcutaneous injections, oral, transdermal patch, and vaginal ring) should be encouraged as a bridge to later initiation of long-acting methods.
Telehealth visits. Video or telephone visits are advised for women desiring counseling and prescribing for contraception or menopausal symptoms.
Deferred. Deferral of office visits until after COVID-19 lockdowns is advised for average-risk women wishing routine well-woman visits. Other situations in which deferral should be considered include the following:
- For patients with abnormal cervical cancer screening results, ACOG suggests that colposcopy with cervical biopsies could be deferred for 6-12 months for patients with low-grade test results. In contrast, for patients with high-grade results, ACOG recommends that evaluation be performed within 3 months.
- For women who wish to discontinue their contraceptive, ACOG advises that removal of IUDs and implants be postponed when possible. These women should be counseled regarding extended use of these devices.
ACOG emphasizes that decisions regarding ambulatory gynecology should be individualized and take into consideration such issues as availability of local and regional resources, staffing, personal protective equipment, and the local prevalence of COVID-19.
As a gynecologist focused on ambulatory care, I believe that many clinicians will welcome this guidance from ACOG, which helps us provide optimal care during these challenging times.
Dr. Kaunitz is professor and associate chairman in the department of obstetrics and gynecology at the University of Florida, Jacksonville. He has disclosed receiving royalties from UpToDate, serving on the safety monitoring board for Femasys, and serving as a consultant for AMAG Pharmaceuticals, Merck & Co, Mithra, and Pfizer. His institution has received funding from pharmaceutical companies and nonprofits.
A version of this article originally appeared on Medscape.com.
What exactly constitutes appropriate ambulatory gynecology during this time of social distancing?
On March 30, 2020, the American College of Obstetricians and Gynecologists (ACOG) weighed in, releasing COVID-19 FAQs for Obstetrician-Gynecologists. These recommendations, which include information about obstetric and gynecologic surgery, are available to everyone, including the general public. They are intended to supplement guidance from the Centers for Disease Control and Prevention, as well as previously released ACOG guidance.
The recommendations include examples of patients needing in-person appointments, telehealth visits, or visits that should be deferred.
In-person appointments. Examples of patients for whom in-person appointments are appropriate include those with suspected ectopic pregnancy or profuse vaginal bleeding. With respect to contraceptive services, ACOG suggests that placement of IUDs and implants should continue whenever possible. If placement of the contraceptive device is deferred, use of self-administered hormonal contraceptives (including subcutaneous injections, oral, transdermal patch, and vaginal ring) should be encouraged as a bridge to later initiation of long-acting methods.
Telehealth visits. Video or telephone visits are advised for women desiring counseling and prescribing for contraception or menopausal symptoms.
Deferred. Deferral of office visits until after COVID-19 lockdowns is advised for average-risk women wishing routine well-woman visits. Other situations in which deferral should be considered include the following:
- For patients with abnormal cervical cancer screening results, ACOG suggests that colposcopy with cervical biopsies could be deferred for 6-12 months for patients with low-grade test results. In contrast, for patients with high-grade results, ACOG recommends that evaluation be performed within 3 months.
- For women who wish to discontinue their contraceptive, ACOG advises that removal of IUDs and implants be postponed when possible. These women should be counseled regarding extended use of these devices.
ACOG emphasizes that decisions regarding ambulatory gynecology should be individualized and take into consideration such issues as availability of local and regional resources, staffing, personal protective equipment, and the local prevalence of COVID-19.
As a gynecologist focused on ambulatory care, I believe that many clinicians will welcome this guidance from ACOG, which helps us provide optimal care during these challenging times.
Dr. Kaunitz is professor and associate chairman in the department of obstetrics and gynecology at the University of Florida, Jacksonville. He has disclosed receiving royalties from UpToDate, serving on the safety monitoring board for Femasys, and serving as a consultant for AMAG Pharmaceuticals, Merck & Co, Mithra, and Pfizer. His institution has received funding from pharmaceutical companies and nonprofits.
A version of this article originally appeared on Medscape.com.
European COVID-19 insights: Try helmet CPAP
Noninvasive ventilation with helmet continuous positive air pressure (CPAP) deserves to be embraced as an effective strategy in preventing self-induced lung injury, often a key factor in progression from the early milder expression of COVID-19 disease to classic severe acute respiratory distress syndrome, according to European physicians who have been through what they hope are the worst days of the pandemic in the Lombardy region of Northern Italy.
Helmet CPAP is a relatively inexpensive, convenient, well-tolerated intervention. It allows patients to remain conscious and responsive to commands such as “Time to roll over,” which in turn frees up nursing staff. The purpose of helmet CPAP is to curb the huge inspiratory drive that’s a defining feature of this disease and which, unchecked, can lead to self-induced lung injury (SILI), Luciano Gattinoni, MD, explained at a webinar hosted by the European Society of Anaesthesiology.
“Paranoid attention to inspiratory effort – checking it and correcting it – is something where we can make the difference between death and life. It’s extremely important,” said Dr. Gattinoni, guest professor of anesthesiology and intensive care at the University of Gottingen (Germany).
He and his fellow panelists were in accord regarding the merits of helmet CPAP as the premier method of noninvasive ventilatory assistance. They also addressed the importance of monitoring for hypercoagulation, as well as what they’ve come to see as the essential role of pronation in what they define as Type H disease, and the need to have detailed respiratory physiotherapy protocols in place.
“COVID-19 doesn’t like physiotherapy,” explained Paolo Pelosi, MD, professor of anesthesiology and intensive care medicine at the University of Genoa (Italy).
Dr. Gattinoni is credited for identification of two polar phenotypes of what he considers to be a single COVID-19 disease. Early on, many patients present with an atypical form of acute respiratory distress syndrome (ARDS), distinguished by an often-unexpected degree of hypoxia accompanied by high pulmonary compliance and surprisingly little shortness of breath. Dr. Gattinoni and colleagues call this Type L disease, which stands for low elastane, low ventilation to perfusion ratio, low lung weight on CT, and low lung recruitability, which means the patient has a high proportion of aerated lung tissue. Over time, because of either the natural history of the disease or SILI, this may shift to Type H disease, marked by high elastane, high right-to-left shunt, high lung weight, and high recruitability.
“If the pulmonary compliance is above 60 [mL/cm H2O], I’m pretty sure it’s Type L. If it’s 30 [mL/cm H2O] or less, I’m pretty sure it’s Type H. Don’t ask me about 45-55 [mL/cm H2O]; it’s a grey zone,” Dr. Gattinoni said.
Giuseppe Foti, MD, said helmet CPAP in patients with COVID-19 should be free flow, not attached to a ventilator, and the gas flow should be set high – at least 50 L/min – in order to prevent CO2 rebreathing. Although noninvasive ventilation is well accepted for patients with chronic obstructive pulmonary disease or acute cardiogenic pulmonary edema, it hasn’t been extensively studied in the setting of ARDS. A notable exception is a single-center randomized trial in which 83 patients with ARDS at the University of Chicago were assigned to noninvasive ventilation delivered by helmet or face mask (JAMA. 2016 Jun 14;315[22]:2435-41). The endotracheal intubation rate was just 18% in the helmet group, compared with 62% in the face mask group. The 90-day mortality rate was significantly lower in the helmet group as well, noted Dr. Foti, director of the department of anesthesia and intensive care at Monza University Hospital in Milan.
Christian Putensen, MD, said he views intubation for mechanical ventilation as wise in moderate or severe ARDS with an arterial oxygen partial pressure/fraction of inspired oxygen (PaO2/FiO2) ratio below 150. But in milder, Type L COVID-19 disease, he also likes helmet CPAP. It spares the patient from the traumatic compressive stress to the lung induced by mechanical ventilation, which may cause alveolar edema and SILI.
There is, however, a caveat: “Watch carefully and do not delay intubation if you see helmet CPAP is not working; that is, if the blood gas analysis doesn’t improve, the respiratory rate increases, tidal volume increases, and there is still increased respiratory drive,” advised Dr. Putensen, an anesthesiologist at the University of Bonn (Germany).
There is no agreed-upon practical quantitative measure of respiratory drive. A clinical evaluation of the patient’s depth of inspiration is the best guide, he added.
Dr. Gattinoni said that, when helmet CPAP can’t control respiratory drive in a patient with early-stage disease, he feels the only way to interrupt this destructive process is through early intubation and what he termed “gentle mechanical ventilation,” not with a positive end expiratory pressure of 20 cm H2O, but more like 4-5.
Watch for hypercoagulation
Thromboembolic complications are a common feature in COVID-19 disease.
“I’ve had occasion to see the autopsy results in more than 100 patients. It’s devastating to see the number of thromboses and microthromboses in the lungs, the liver, the kidney, and in the brain,” Dr. Gattinoni said.
“COVID-19 is a serial killer, no doubt,” Dr. Pelosi agreed. “He has no mercy for anyone. And he has two bullets: The first one is for the lung, the second is on the vascular side.”
Dr. Putensen is aggressive in utilizing prophylactic high-dose anticoagulation with heparin. He carefully monitors levels of fibrinogen, Factors V and VIII, and d-dimers. In the setting of COVID-19, he has found thromboelastography to be more reliable than partial thromboplastin time in guiding heparin titration.
Pronation
Panelists agreed that pronation is an especially valuable means of enhancing oxygenation in patients with Type H disease. Dr. Putensen tries for more than 16 hours per day. Dr. Foti is preparing a study of the impact of pronation in 50 awake, nonintubated patients, most of whom were on helmet CPAP. Seven of them couldn’t tolerate pronation for even an hour at a time; for the others, the median duration was 3.5 hours at a time.
“We saw a dramatic improvement, a nearly doubling in the PaO2/FiO2 ratio,” Dr. Foti said.
The helmet CPAP study was done outside of the ICU because, in March 2020, the Milan hospital was utterly overwhelmed by COVID-19. The university hospital ordinarily has 25 ICU beds. This was expanded to 100 ICU beds in an effort to meet the emergency, but that still wasn’t sufficient. Indeed, COVID-19 patients occupied 600 of the hospital’s 650 beds. Physicians were forced to do something formerly unthinkable: triage patients for intubation and mechanical ventilation based upon age, comorbidities, and survival prospects.
“We felt schizophrenic. I completely agree with Luciano’s idea to intubate early when we cannot control the respiratory drive that’s due to the disease. But we couldn’t do it because we had too many patients. So we had to triage,” Dr. Foti recalled, breaking off with a sob as other panelists wiped away their own tears during the webcast.
Respiratory physical therapy
Dr. Pelosi said he believes that optimal care of patients with COVID-19 disease requires a major commitment to physical therapy. He strongly recommends having thoughtfully designed separate written protocols in place for respiratory physiotherapy during mechanical ventilation, weaning, and postextubation. COVID-19 patients typically require 7-10 days of assisted ventilation before weaning, and weaning is a protracted process as well.
“I like to say COVID-19 always requires patience. You have to be very, very patient with this disease,” he emphasized. “These patients have a long and difficult weaning. If the patient isn’t improving during weaning, look at two issues: superinfection and thrombembolism, macro and micro.” The physical therapy measures routinely utilized at his hospital during mechanical ventilation include elevation of the bed head greater than 30 degrees, neuromuscular electrical stimulation, subglottic secretion suctioning, tracheal and oral aspiration, and cough assistance. Separate physical therapy menus are used during before and after extubation.
Dr. Gattinoni offered a final word: “We can do almost nothing with this disease. We try our best to keep the patient alive. What we can do is avoid excessive ventilation of the patient. Applying the typical treatment of ARDS in atypical [Type L] ARDS does not make sense and may be extremely harmful.”
Noninvasive ventilation with helmet continuous positive air pressure (CPAP) deserves to be embraced as an effective strategy in preventing self-induced lung injury, often a key factor in progression from the early milder expression of COVID-19 disease to classic severe acute respiratory distress syndrome, according to European physicians who have been through what they hope are the worst days of the pandemic in the Lombardy region of Northern Italy.
Helmet CPAP is a relatively inexpensive, convenient, well-tolerated intervention. It allows patients to remain conscious and responsive to commands such as “Time to roll over,” which in turn frees up nursing staff. The purpose of helmet CPAP is to curb the huge inspiratory drive that’s a defining feature of this disease and which, unchecked, can lead to self-induced lung injury (SILI), Luciano Gattinoni, MD, explained at a webinar hosted by the European Society of Anaesthesiology.
“Paranoid attention to inspiratory effort – checking it and correcting it – is something where we can make the difference between death and life. It’s extremely important,” said Dr. Gattinoni, guest professor of anesthesiology and intensive care at the University of Gottingen (Germany).
He and his fellow panelists were in accord regarding the merits of helmet CPAP as the premier method of noninvasive ventilatory assistance. They also addressed the importance of monitoring for hypercoagulation, as well as what they’ve come to see as the essential role of pronation in what they define as Type H disease, and the need to have detailed respiratory physiotherapy protocols in place.
“COVID-19 doesn’t like physiotherapy,” explained Paolo Pelosi, MD, professor of anesthesiology and intensive care medicine at the University of Genoa (Italy).
Dr. Gattinoni is credited for identification of two polar phenotypes of what he considers to be a single COVID-19 disease. Early on, many patients present with an atypical form of acute respiratory distress syndrome (ARDS), distinguished by an often-unexpected degree of hypoxia accompanied by high pulmonary compliance and surprisingly little shortness of breath. Dr. Gattinoni and colleagues call this Type L disease, which stands for low elastane, low ventilation to perfusion ratio, low lung weight on CT, and low lung recruitability, which means the patient has a high proportion of aerated lung tissue. Over time, because of either the natural history of the disease or SILI, this may shift to Type H disease, marked by high elastane, high right-to-left shunt, high lung weight, and high recruitability.
“If the pulmonary compliance is above 60 [mL/cm H2O], I’m pretty sure it’s Type L. If it’s 30 [mL/cm H2O] or less, I’m pretty sure it’s Type H. Don’t ask me about 45-55 [mL/cm H2O]; it’s a grey zone,” Dr. Gattinoni said.
Giuseppe Foti, MD, said helmet CPAP in patients with COVID-19 should be free flow, not attached to a ventilator, and the gas flow should be set high – at least 50 L/min – in order to prevent CO2 rebreathing. Although noninvasive ventilation is well accepted for patients with chronic obstructive pulmonary disease or acute cardiogenic pulmonary edema, it hasn’t been extensively studied in the setting of ARDS. A notable exception is a single-center randomized trial in which 83 patients with ARDS at the University of Chicago were assigned to noninvasive ventilation delivered by helmet or face mask (JAMA. 2016 Jun 14;315[22]:2435-41). The endotracheal intubation rate was just 18% in the helmet group, compared with 62% in the face mask group. The 90-day mortality rate was significantly lower in the helmet group as well, noted Dr. Foti, director of the department of anesthesia and intensive care at Monza University Hospital in Milan.
Christian Putensen, MD, said he views intubation for mechanical ventilation as wise in moderate or severe ARDS with an arterial oxygen partial pressure/fraction of inspired oxygen (PaO2/FiO2) ratio below 150. But in milder, Type L COVID-19 disease, he also likes helmet CPAP. It spares the patient from the traumatic compressive stress to the lung induced by mechanical ventilation, which may cause alveolar edema and SILI.
There is, however, a caveat: “Watch carefully and do not delay intubation if you see helmet CPAP is not working; that is, if the blood gas analysis doesn’t improve, the respiratory rate increases, tidal volume increases, and there is still increased respiratory drive,” advised Dr. Putensen, an anesthesiologist at the University of Bonn (Germany).
There is no agreed-upon practical quantitative measure of respiratory drive. A clinical evaluation of the patient’s depth of inspiration is the best guide, he added.
Dr. Gattinoni said that, when helmet CPAP can’t control respiratory drive in a patient with early-stage disease, he feels the only way to interrupt this destructive process is through early intubation and what he termed “gentle mechanical ventilation,” not with a positive end expiratory pressure of 20 cm H2O, but more like 4-5.
Watch for hypercoagulation
Thromboembolic complications are a common feature in COVID-19 disease.
“I’ve had occasion to see the autopsy results in more than 100 patients. It’s devastating to see the number of thromboses and microthromboses in the lungs, the liver, the kidney, and in the brain,” Dr. Gattinoni said.
“COVID-19 is a serial killer, no doubt,” Dr. Pelosi agreed. “He has no mercy for anyone. And he has two bullets: The first one is for the lung, the second is on the vascular side.”
Dr. Putensen is aggressive in utilizing prophylactic high-dose anticoagulation with heparin. He carefully monitors levels of fibrinogen, Factors V and VIII, and d-dimers. In the setting of COVID-19, he has found thromboelastography to be more reliable than partial thromboplastin time in guiding heparin titration.
Pronation
Panelists agreed that pronation is an especially valuable means of enhancing oxygenation in patients with Type H disease. Dr. Putensen tries for more than 16 hours per day. Dr. Foti is preparing a study of the impact of pronation in 50 awake, nonintubated patients, most of whom were on helmet CPAP. Seven of them couldn’t tolerate pronation for even an hour at a time; for the others, the median duration was 3.5 hours at a time.
“We saw a dramatic improvement, a nearly doubling in the PaO2/FiO2 ratio,” Dr. Foti said.
The helmet CPAP study was done outside of the ICU because, in March 2020, the Milan hospital was utterly overwhelmed by COVID-19. The university hospital ordinarily has 25 ICU beds. This was expanded to 100 ICU beds in an effort to meet the emergency, but that still wasn’t sufficient. Indeed, COVID-19 patients occupied 600 of the hospital’s 650 beds. Physicians were forced to do something formerly unthinkable: triage patients for intubation and mechanical ventilation based upon age, comorbidities, and survival prospects.
“We felt schizophrenic. I completely agree with Luciano’s idea to intubate early when we cannot control the respiratory drive that’s due to the disease. But we couldn’t do it because we had too many patients. So we had to triage,” Dr. Foti recalled, breaking off with a sob as other panelists wiped away their own tears during the webcast.
Respiratory physical therapy
Dr. Pelosi said he believes that optimal care of patients with COVID-19 disease requires a major commitment to physical therapy. He strongly recommends having thoughtfully designed separate written protocols in place for respiratory physiotherapy during mechanical ventilation, weaning, and postextubation. COVID-19 patients typically require 7-10 days of assisted ventilation before weaning, and weaning is a protracted process as well.
“I like to say COVID-19 always requires patience. You have to be very, very patient with this disease,” he emphasized. “These patients have a long and difficult weaning. If the patient isn’t improving during weaning, look at two issues: superinfection and thrombembolism, macro and micro.” The physical therapy measures routinely utilized at his hospital during mechanical ventilation include elevation of the bed head greater than 30 degrees, neuromuscular electrical stimulation, subglottic secretion suctioning, tracheal and oral aspiration, and cough assistance. Separate physical therapy menus are used during before and after extubation.
Dr. Gattinoni offered a final word: “We can do almost nothing with this disease. We try our best to keep the patient alive. What we can do is avoid excessive ventilation of the patient. Applying the typical treatment of ARDS in atypical [Type L] ARDS does not make sense and may be extremely harmful.”
Noninvasive ventilation with helmet continuous positive air pressure (CPAP) deserves to be embraced as an effective strategy in preventing self-induced lung injury, often a key factor in progression from the early milder expression of COVID-19 disease to classic severe acute respiratory distress syndrome, according to European physicians who have been through what they hope are the worst days of the pandemic in the Lombardy region of Northern Italy.
Helmet CPAP is a relatively inexpensive, convenient, well-tolerated intervention. It allows patients to remain conscious and responsive to commands such as “Time to roll over,” which in turn frees up nursing staff. The purpose of helmet CPAP is to curb the huge inspiratory drive that’s a defining feature of this disease and which, unchecked, can lead to self-induced lung injury (SILI), Luciano Gattinoni, MD, explained at a webinar hosted by the European Society of Anaesthesiology.
“Paranoid attention to inspiratory effort – checking it and correcting it – is something where we can make the difference between death and life. It’s extremely important,” said Dr. Gattinoni, guest professor of anesthesiology and intensive care at the University of Gottingen (Germany).
He and his fellow panelists were in accord regarding the merits of helmet CPAP as the premier method of noninvasive ventilatory assistance. They also addressed the importance of monitoring for hypercoagulation, as well as what they’ve come to see as the essential role of pronation in what they define as Type H disease, and the need to have detailed respiratory physiotherapy protocols in place.
“COVID-19 doesn’t like physiotherapy,” explained Paolo Pelosi, MD, professor of anesthesiology and intensive care medicine at the University of Genoa (Italy).
Dr. Gattinoni is credited for identification of two polar phenotypes of what he considers to be a single COVID-19 disease. Early on, many patients present with an atypical form of acute respiratory distress syndrome (ARDS), distinguished by an often-unexpected degree of hypoxia accompanied by high pulmonary compliance and surprisingly little shortness of breath. Dr. Gattinoni and colleagues call this Type L disease, which stands for low elastane, low ventilation to perfusion ratio, low lung weight on CT, and low lung recruitability, which means the patient has a high proportion of aerated lung tissue. Over time, because of either the natural history of the disease or SILI, this may shift to Type H disease, marked by high elastane, high right-to-left shunt, high lung weight, and high recruitability.
“If the pulmonary compliance is above 60 [mL/cm H2O], I’m pretty sure it’s Type L. If it’s 30 [mL/cm H2O] or less, I’m pretty sure it’s Type H. Don’t ask me about 45-55 [mL/cm H2O]; it’s a grey zone,” Dr. Gattinoni said.
Giuseppe Foti, MD, said helmet CPAP in patients with COVID-19 should be free flow, not attached to a ventilator, and the gas flow should be set high – at least 50 L/min – in order to prevent CO2 rebreathing. Although noninvasive ventilation is well accepted for patients with chronic obstructive pulmonary disease or acute cardiogenic pulmonary edema, it hasn’t been extensively studied in the setting of ARDS. A notable exception is a single-center randomized trial in which 83 patients with ARDS at the University of Chicago were assigned to noninvasive ventilation delivered by helmet or face mask (JAMA. 2016 Jun 14;315[22]:2435-41). The endotracheal intubation rate was just 18% in the helmet group, compared with 62% in the face mask group. The 90-day mortality rate was significantly lower in the helmet group as well, noted Dr. Foti, director of the department of anesthesia and intensive care at Monza University Hospital in Milan.
Christian Putensen, MD, said he views intubation for mechanical ventilation as wise in moderate or severe ARDS with an arterial oxygen partial pressure/fraction of inspired oxygen (PaO2/FiO2) ratio below 150. But in milder, Type L COVID-19 disease, he also likes helmet CPAP. It spares the patient from the traumatic compressive stress to the lung induced by mechanical ventilation, which may cause alveolar edema and SILI.
There is, however, a caveat: “Watch carefully and do not delay intubation if you see helmet CPAP is not working; that is, if the blood gas analysis doesn’t improve, the respiratory rate increases, tidal volume increases, and there is still increased respiratory drive,” advised Dr. Putensen, an anesthesiologist at the University of Bonn (Germany).
There is no agreed-upon practical quantitative measure of respiratory drive. A clinical evaluation of the patient’s depth of inspiration is the best guide, he added.
Dr. Gattinoni said that, when helmet CPAP can’t control respiratory drive in a patient with early-stage disease, he feels the only way to interrupt this destructive process is through early intubation and what he termed “gentle mechanical ventilation,” not with a positive end expiratory pressure of 20 cm H2O, but more like 4-5.
Watch for hypercoagulation
Thromboembolic complications are a common feature in COVID-19 disease.
“I’ve had occasion to see the autopsy results in more than 100 patients. It’s devastating to see the number of thromboses and microthromboses in the lungs, the liver, the kidney, and in the brain,” Dr. Gattinoni said.
“COVID-19 is a serial killer, no doubt,” Dr. Pelosi agreed. “He has no mercy for anyone. And he has two bullets: The first one is for the lung, the second is on the vascular side.”
Dr. Putensen is aggressive in utilizing prophylactic high-dose anticoagulation with heparin. He carefully monitors levels of fibrinogen, Factors V and VIII, and d-dimers. In the setting of COVID-19, he has found thromboelastography to be more reliable than partial thromboplastin time in guiding heparin titration.
Pronation
Panelists agreed that pronation is an especially valuable means of enhancing oxygenation in patients with Type H disease. Dr. Putensen tries for more than 16 hours per day. Dr. Foti is preparing a study of the impact of pronation in 50 awake, nonintubated patients, most of whom were on helmet CPAP. Seven of them couldn’t tolerate pronation for even an hour at a time; for the others, the median duration was 3.5 hours at a time.
“We saw a dramatic improvement, a nearly doubling in the PaO2/FiO2 ratio,” Dr. Foti said.
The helmet CPAP study was done outside of the ICU because, in March 2020, the Milan hospital was utterly overwhelmed by COVID-19. The university hospital ordinarily has 25 ICU beds. This was expanded to 100 ICU beds in an effort to meet the emergency, but that still wasn’t sufficient. Indeed, COVID-19 patients occupied 600 of the hospital’s 650 beds. Physicians were forced to do something formerly unthinkable: triage patients for intubation and mechanical ventilation based upon age, comorbidities, and survival prospects.
“We felt schizophrenic. I completely agree with Luciano’s idea to intubate early when we cannot control the respiratory drive that’s due to the disease. But we couldn’t do it because we had too many patients. So we had to triage,” Dr. Foti recalled, breaking off with a sob as other panelists wiped away their own tears during the webcast.
Respiratory physical therapy
Dr. Pelosi said he believes that optimal care of patients with COVID-19 disease requires a major commitment to physical therapy. He strongly recommends having thoughtfully designed separate written protocols in place for respiratory physiotherapy during mechanical ventilation, weaning, and postextubation. COVID-19 patients typically require 7-10 days of assisted ventilation before weaning, and weaning is a protracted process as well.
“I like to say COVID-19 always requires patience. You have to be very, very patient with this disease,” he emphasized. “These patients have a long and difficult weaning. If the patient isn’t improving during weaning, look at two issues: superinfection and thrombembolism, macro and micro.” The physical therapy measures routinely utilized at his hospital during mechanical ventilation include elevation of the bed head greater than 30 degrees, neuromuscular electrical stimulation, subglottic secretion suctioning, tracheal and oral aspiration, and cough assistance. Separate physical therapy menus are used during before and after extubation.
Dr. Gattinoni offered a final word: “We can do almost nothing with this disease. We try our best to keep the patient alive. What we can do is avoid excessive ventilation of the patient. Applying the typical treatment of ARDS in atypical [Type L] ARDS does not make sense and may be extremely harmful.”
Are patients with epilepsy at increased risk of COVID-19 infection?
Chronic conditions such as lung disease, diabetes, and heart disease frequently receive attention for increasing the risk of complications for people who contract the coronavirus. Meanwhile, many members of the epilepsy community continue to wonder how the virus affects them. To address these concerns, the Epilepsy Foundation has released information that answers many common questions that people with epilepsy have about how COVID-19 can impact their health.
Perhaps the most pressing of these questions is: Does epilepsy increase the risk or severity of the coronavirus?
“The most common thing we’re hearing from patients in my practice is their proactive concern for being at increased risk for getting the coronavirus,” confirmed Selim Benbadis, MD, division director, epilepsy, EEG, and sleep medicine at the University of South Florida in Tampa. “Epilepsy patients are not at increased risk for complications from the coronavirus because epilepsy does not affect the immune system.”
In other words, people who have epilepsy face the same health challenges as people who do not have the condition and are otherwise healthy. For this reason, people who have epilepsy should exercise the same habits and preventative measures that healthy people would typically take, such as social distancing; avoiding contact with sick people; washing hands regularly; disinfecting surfaces regularly; and avoiding touching hands, eyes, nose and mouth.
However, as Dr. Benbadis explained, the high fever associated with coronavirus can trigger seizures. The increased risk is another reason people who have epilepsy should do their best to avoid getting sick.
Seizure medications do not increase COVID-19 risk but other conditions can
Similarly, epilepsy medications do not increase the risk of contracting the disease.
“The medications patients take to treat their epilepsy do not affect their immune system,” said Andrew Wilner, MD, associate professor of neurology at the University of Tennessee Health Science Center, Memphis. There are a few exceptions – such as adrenocorticotropic hormone and everolimus – but doctors rarely use these drugs to treat epilepsy.
However, there are some situations and conditions that may pose a risk for people who contact the coronavirus. For instance, people who have problems swallowing their food and tend to suck food down their windpipes are more likely to develop pneumonia. Also, much like the general population, having diabetes, heart disease, or lung problems increase the chances of developing complications from the virus.
The best ways to avoid additional risks in epilepsy
Because of the pandemic, people who have epilepsy may have found that many of their doctors’ appointments have been canceled. Many clinics and medical practices have done this in order minimize exposing people who have acute illnesses to the virus. By focusing more on patients with acute conditions, doctors and nurses can better tend to patients with acute problems. As a result, practices have shifted to providing patient care using telemedicine as much as possible.
“Telemedicine services have surged, and I’ve been saying for years that telemedicine was going to grow,” Dr. Benbadis said. “It’s more convenient, and I believe that we’re going to see increased use of telemedicine long after the coronavirus pandemic is over.”
Aside from communicating with their doctors, the Epilepsy Foundation and Dr. Wilner stress that the best way for people who have epilepsy to stay healthy is by taking their medications on a regular basis exactly as prescribed.
“Taking mediation correctly and regularly is the best strategy for epilepsy patients to avoid unnecessary hospitalizations,” Dr. Wilner said. “If they have breakthrough seizures and get sent to the emergency room, then they risk being exposed to the virus in the ER.”
Also, because ERs are more crowded than usual, the Epilepsy Foundation encourages people who suspect they have the coronavirus to call their doctor’s office first. The goal is to try to make sure that people who have severe or life-threatening symptoms have access to treatment in the ER.
As with the general population, the first thing that epilepsy patients who suspect they have the coronavirus should do is call his or her doctor’s office. The health care professional taking the call will ask the patient a series of questions to determine whether the patient has COVID-19 or another condition or needs to seek emergency medical attention.
Fever, cough, and trouble breathing fall among the most commonly reported symptoms of the coronavirus. In many cases, health care providers recommend that people with mild versions of these symptoms stay at home.
Helpful tips
The Epilepsy Foundation offers tips on signs to look for when trying to figure out when a seizure requires an ER visit. These are:
- Seizures in which awareness is lost for more than 5 minutes and no reversal medications are available.
- Seizures with an unusual pattern or duration.
- Seizures that cannot be treated safely at home or are not responding to rescue medication even after the medication has had enough time to work.
- Seizures that occur after a severe blow to the head.
Additionally, while COVID-19 can cause death and sudden death in patients, the virus does not cause sudden unexpected death in epilepsy (SUDEP). Because SUDEP is extremely rare, Dr. Benbadis said that there is no information to suggest that contracting the coronavirus will increase the risk,
Finally, no shortages of seizures medications have been reported as a result of COVID-19. However, there were shortages of generic levetiracetam immediate-release and levetiracetam extended-release medications prior to and during COVID-19. Experts expect the shortage to continue.
Overall, people who have epilepsy should be able to stay healthy – provided they exercise healthy and preventative habits.
“The majority of epilepsy patients should be reassured that if they continue their usual care, take their meds as directed, get adequate sleep, nutritious diet, they’re not at any increased risk compared to the general population,” said Dr. Wilner.
Dr. Benbadis reported the following disclosures: consultant for Bioserenity (DigiTrace), Brain Sentinel, Cavion, Ceribell, Eisai, Greenwich, LivaNova, Neuropace, SK biopharmaceuticals, Sunovion; speakers bureau for Eisai, Greenwich, LivaNova, Sunovion; Florida Medical Director of Stratus/Alliance; Member: Epilepsy Study Consortium; grant support from Cavion, LivaNova, Greenwich, SK biopharmaceuticals, Sunovion, Takeda, UCB, Xenon; royalties as an author or editor for Emedicine-Medscape-WebMD, UpToDate; editorial board for the Epilepsy.com (Epilepsy Foundation) controversy section, Emedicine-Medscape-WebMD, Epileptic Disorders, Epilepsy and Behavior, and Expert Review of Neurotherapeutics. Dr. Wilner reports Medical Advisory Board of Accordant Health Services, Greensboro, S.C., and book royalties: “The Locum Life: A Physician’s Guide to Locum Tenens,” Lulu Press.
Chronic conditions such as lung disease, diabetes, and heart disease frequently receive attention for increasing the risk of complications for people who contract the coronavirus. Meanwhile, many members of the epilepsy community continue to wonder how the virus affects them. To address these concerns, the Epilepsy Foundation has released information that answers many common questions that people with epilepsy have about how COVID-19 can impact their health.
Perhaps the most pressing of these questions is: Does epilepsy increase the risk or severity of the coronavirus?
“The most common thing we’re hearing from patients in my practice is their proactive concern for being at increased risk for getting the coronavirus,” confirmed Selim Benbadis, MD, division director, epilepsy, EEG, and sleep medicine at the University of South Florida in Tampa. “Epilepsy patients are not at increased risk for complications from the coronavirus because epilepsy does not affect the immune system.”
In other words, people who have epilepsy face the same health challenges as people who do not have the condition and are otherwise healthy. For this reason, people who have epilepsy should exercise the same habits and preventative measures that healthy people would typically take, such as social distancing; avoiding contact with sick people; washing hands regularly; disinfecting surfaces regularly; and avoiding touching hands, eyes, nose and mouth.
However, as Dr. Benbadis explained, the high fever associated with coronavirus can trigger seizures. The increased risk is another reason people who have epilepsy should do their best to avoid getting sick.
Seizure medications do not increase COVID-19 risk but other conditions can
Similarly, epilepsy medications do not increase the risk of contracting the disease.
“The medications patients take to treat their epilepsy do not affect their immune system,” said Andrew Wilner, MD, associate professor of neurology at the University of Tennessee Health Science Center, Memphis. There are a few exceptions – such as adrenocorticotropic hormone and everolimus – but doctors rarely use these drugs to treat epilepsy.
However, there are some situations and conditions that may pose a risk for people who contact the coronavirus. For instance, people who have problems swallowing their food and tend to suck food down their windpipes are more likely to develop pneumonia. Also, much like the general population, having diabetes, heart disease, or lung problems increase the chances of developing complications from the virus.
The best ways to avoid additional risks in epilepsy
Because of the pandemic, people who have epilepsy may have found that many of their doctors’ appointments have been canceled. Many clinics and medical practices have done this in order minimize exposing people who have acute illnesses to the virus. By focusing more on patients with acute conditions, doctors and nurses can better tend to patients with acute problems. As a result, practices have shifted to providing patient care using telemedicine as much as possible.
“Telemedicine services have surged, and I’ve been saying for years that telemedicine was going to grow,” Dr. Benbadis said. “It’s more convenient, and I believe that we’re going to see increased use of telemedicine long after the coronavirus pandemic is over.”
Aside from communicating with their doctors, the Epilepsy Foundation and Dr. Wilner stress that the best way for people who have epilepsy to stay healthy is by taking their medications on a regular basis exactly as prescribed.
“Taking mediation correctly and regularly is the best strategy for epilepsy patients to avoid unnecessary hospitalizations,” Dr. Wilner said. “If they have breakthrough seizures and get sent to the emergency room, then they risk being exposed to the virus in the ER.”
Also, because ERs are more crowded than usual, the Epilepsy Foundation encourages people who suspect they have the coronavirus to call their doctor’s office first. The goal is to try to make sure that people who have severe or life-threatening symptoms have access to treatment in the ER.
As with the general population, the first thing that epilepsy patients who suspect they have the coronavirus should do is call his or her doctor’s office. The health care professional taking the call will ask the patient a series of questions to determine whether the patient has COVID-19 or another condition or needs to seek emergency medical attention.
Fever, cough, and trouble breathing fall among the most commonly reported symptoms of the coronavirus. In many cases, health care providers recommend that people with mild versions of these symptoms stay at home.
Helpful tips
The Epilepsy Foundation offers tips on signs to look for when trying to figure out when a seizure requires an ER visit. These are:
- Seizures in which awareness is lost for more than 5 minutes and no reversal medications are available.
- Seizures with an unusual pattern or duration.
- Seizures that cannot be treated safely at home or are not responding to rescue medication even after the medication has had enough time to work.
- Seizures that occur after a severe blow to the head.
Additionally, while COVID-19 can cause death and sudden death in patients, the virus does not cause sudden unexpected death in epilepsy (SUDEP). Because SUDEP is extremely rare, Dr. Benbadis said that there is no information to suggest that contracting the coronavirus will increase the risk,
Finally, no shortages of seizures medications have been reported as a result of COVID-19. However, there were shortages of generic levetiracetam immediate-release and levetiracetam extended-release medications prior to and during COVID-19. Experts expect the shortage to continue.
Overall, people who have epilepsy should be able to stay healthy – provided they exercise healthy and preventative habits.
“The majority of epilepsy patients should be reassured that if they continue their usual care, take their meds as directed, get adequate sleep, nutritious diet, they’re not at any increased risk compared to the general population,” said Dr. Wilner.
Dr. Benbadis reported the following disclosures: consultant for Bioserenity (DigiTrace), Brain Sentinel, Cavion, Ceribell, Eisai, Greenwich, LivaNova, Neuropace, SK biopharmaceuticals, Sunovion; speakers bureau for Eisai, Greenwich, LivaNova, Sunovion; Florida Medical Director of Stratus/Alliance; Member: Epilepsy Study Consortium; grant support from Cavion, LivaNova, Greenwich, SK biopharmaceuticals, Sunovion, Takeda, UCB, Xenon; royalties as an author or editor for Emedicine-Medscape-WebMD, UpToDate; editorial board for the Epilepsy.com (Epilepsy Foundation) controversy section, Emedicine-Medscape-WebMD, Epileptic Disorders, Epilepsy and Behavior, and Expert Review of Neurotherapeutics. Dr. Wilner reports Medical Advisory Board of Accordant Health Services, Greensboro, S.C., and book royalties: “The Locum Life: A Physician’s Guide to Locum Tenens,” Lulu Press.
Chronic conditions such as lung disease, diabetes, and heart disease frequently receive attention for increasing the risk of complications for people who contract the coronavirus. Meanwhile, many members of the epilepsy community continue to wonder how the virus affects them. To address these concerns, the Epilepsy Foundation has released information that answers many common questions that people with epilepsy have about how COVID-19 can impact their health.
Perhaps the most pressing of these questions is: Does epilepsy increase the risk or severity of the coronavirus?
“The most common thing we’re hearing from patients in my practice is their proactive concern for being at increased risk for getting the coronavirus,” confirmed Selim Benbadis, MD, division director, epilepsy, EEG, and sleep medicine at the University of South Florida in Tampa. “Epilepsy patients are not at increased risk for complications from the coronavirus because epilepsy does not affect the immune system.”
In other words, people who have epilepsy face the same health challenges as people who do not have the condition and are otherwise healthy. For this reason, people who have epilepsy should exercise the same habits and preventative measures that healthy people would typically take, such as social distancing; avoiding contact with sick people; washing hands regularly; disinfecting surfaces regularly; and avoiding touching hands, eyes, nose and mouth.
However, as Dr. Benbadis explained, the high fever associated with coronavirus can trigger seizures. The increased risk is another reason people who have epilepsy should do their best to avoid getting sick.
Seizure medications do not increase COVID-19 risk but other conditions can
Similarly, epilepsy medications do not increase the risk of contracting the disease.
“The medications patients take to treat their epilepsy do not affect their immune system,” said Andrew Wilner, MD, associate professor of neurology at the University of Tennessee Health Science Center, Memphis. There are a few exceptions – such as adrenocorticotropic hormone and everolimus – but doctors rarely use these drugs to treat epilepsy.
However, there are some situations and conditions that may pose a risk for people who contact the coronavirus. For instance, people who have problems swallowing their food and tend to suck food down their windpipes are more likely to develop pneumonia. Also, much like the general population, having diabetes, heart disease, or lung problems increase the chances of developing complications from the virus.
The best ways to avoid additional risks in epilepsy
Because of the pandemic, people who have epilepsy may have found that many of their doctors’ appointments have been canceled. Many clinics and medical practices have done this in order minimize exposing people who have acute illnesses to the virus. By focusing more on patients with acute conditions, doctors and nurses can better tend to patients with acute problems. As a result, practices have shifted to providing patient care using telemedicine as much as possible.
“Telemedicine services have surged, and I’ve been saying for years that telemedicine was going to grow,” Dr. Benbadis said. “It’s more convenient, and I believe that we’re going to see increased use of telemedicine long after the coronavirus pandemic is over.”
Aside from communicating with their doctors, the Epilepsy Foundation and Dr. Wilner stress that the best way for people who have epilepsy to stay healthy is by taking their medications on a regular basis exactly as prescribed.
“Taking mediation correctly and regularly is the best strategy for epilepsy patients to avoid unnecessary hospitalizations,” Dr. Wilner said. “If they have breakthrough seizures and get sent to the emergency room, then they risk being exposed to the virus in the ER.”
Also, because ERs are more crowded than usual, the Epilepsy Foundation encourages people who suspect they have the coronavirus to call their doctor’s office first. The goal is to try to make sure that people who have severe or life-threatening symptoms have access to treatment in the ER.
As with the general population, the first thing that epilepsy patients who suspect they have the coronavirus should do is call his or her doctor’s office. The health care professional taking the call will ask the patient a series of questions to determine whether the patient has COVID-19 or another condition or needs to seek emergency medical attention.
Fever, cough, and trouble breathing fall among the most commonly reported symptoms of the coronavirus. In many cases, health care providers recommend that people with mild versions of these symptoms stay at home.
Helpful tips
The Epilepsy Foundation offers tips on signs to look for when trying to figure out when a seizure requires an ER visit. These are:
- Seizures in which awareness is lost for more than 5 minutes and no reversal medications are available.
- Seizures with an unusual pattern or duration.
- Seizures that cannot be treated safely at home or are not responding to rescue medication even after the medication has had enough time to work.
- Seizures that occur after a severe blow to the head.
Additionally, while COVID-19 can cause death and sudden death in patients, the virus does not cause sudden unexpected death in epilepsy (SUDEP). Because SUDEP is extremely rare, Dr. Benbadis said that there is no information to suggest that contracting the coronavirus will increase the risk,
Finally, no shortages of seizures medications have been reported as a result of COVID-19. However, there were shortages of generic levetiracetam immediate-release and levetiracetam extended-release medications prior to and during COVID-19. Experts expect the shortage to continue.
Overall, people who have epilepsy should be able to stay healthy – provided they exercise healthy and preventative habits.
“The majority of epilepsy patients should be reassured that if they continue their usual care, take their meds as directed, get adequate sleep, nutritious diet, they’re not at any increased risk compared to the general population,” said Dr. Wilner.
Dr. Benbadis reported the following disclosures: consultant for Bioserenity (DigiTrace), Brain Sentinel, Cavion, Ceribell, Eisai, Greenwich, LivaNova, Neuropace, SK biopharmaceuticals, Sunovion; speakers bureau for Eisai, Greenwich, LivaNova, Sunovion; Florida Medical Director of Stratus/Alliance; Member: Epilepsy Study Consortium; grant support from Cavion, LivaNova, Greenwich, SK biopharmaceuticals, Sunovion, Takeda, UCB, Xenon; royalties as an author or editor for Emedicine-Medscape-WebMD, UpToDate; editorial board for the Epilepsy.com (Epilepsy Foundation) controversy section, Emedicine-Medscape-WebMD, Epileptic Disorders, Epilepsy and Behavior, and Expert Review of Neurotherapeutics. Dr. Wilner reports Medical Advisory Board of Accordant Health Services, Greensboro, S.C., and book royalties: “The Locum Life: A Physician’s Guide to Locum Tenens,” Lulu Press.
COVID-19 antibody tests proliferate, but what do they show?
Noopur Raje, MD, has been sitting at home for 5 weeks waiting for her COVID-19 test to turn negative so she can get back to work. She’s a cancer specialist – head of the Massachusetts General Hospital’s Center for Multiple Myeloma – but Raje says as soon as she’s allowed back to the hospital, she’ll head straight to the front line of COVID-19 caregivers.
“It’s people like us who have to get back in the trenches and do the work now,” she told Medscape Medical News.
“I still will be at risk,” she said. But, having nursed her physician husband through COVID-19 at home until he was admitted to an intensive care unit, she is determined to help in the COVID-19 wards.
“I will be the first one to volunteer to take care of these patients,” she said. “I can’t wait, as I want to give these folks hope. They are so scared.”
Around the world, it’s assumed that she and others like her who’ve recovered from COVID-19 will be immune to the infection.
Some have suggested that with antibodies to the virus coursing through their veins, these survivors might be given immunity passports. They could be the ones to jump-start people’s lives again ― the first to be let out from lockdown, and in healthcare, the ones to head the ongoing battle against this pandemic.
So, there has been a race to develop COVID-19 antibody tests to identify these people.
Circumventing the Usual Clearance Process
To speed up the process, the US Food and Drug Administration (FDA) made a much-criticized move to allow a free-for-all for developers to begin marketing antibody tests that had not gone through the agency’s usual evaluation process. The result was a flood of more than 90 unapproved tests “that have, frankly, dubious quality,” said Scott Becker, CEO of the Association of Public Health Laboratories (APHL), which represents local and state public laboratories.
The APHL spoke out in dismay – its chief program officer, Eric Blank, decried the “Wild West” of tests unleashed on the public.
“These tests create more uncertainty than before,” said Kelly Wroblewski, APHL’s director of infectious diseases, in a news conference on April 14. “Having many inaccurate tests is worse than having no tests at all.”
The APHL and the FDA, working with the Centers for Disease Control and Prevention and the National Institutes of Health (NIH), have moved quickly into damage control, conducting evaluations of the tests in an effort to distinguish the potentially useful from the useless.
So far, they have succeeded in issuing emergency use authorizations (EUAs) to only four tests, those marketed by Cellex, Ortho Clinical Diagnostics, Chembio Diagnostic Systems, and the Mount Sinai Laboratory.
For all the other antibody tests on the market that do not have an EUA, “They’re trusting that the test developer has done a good job in validation,” Becker said. But there are worrying anecdotes. “Our members have reported that they’ve seen fraudulent marketing.... We’ve seen the FDA clamp down on some companies... [and] a number of cities and health departments have issued warnings because of what they’ve seen,” he added.
In particular, Wroblewski said, some companies are marketing tests for use in physicians’ offices or pharmacies. “Today, there are no serology tests approved for point-of-care settings,” she warned. “We don’t know how to interpret the test results, if the presence of antibodies indicates immunity, how long it will last, or what titer might be sufficient.”
Uncertainty Emphasized
The FDA emphasized the uncertainty about antibody tests in a statement released on April 18.
Although the tests can identify people who have been exposed and who developed an immune response to the virus, the agency noted, “we don’t yet know that just because someone has developed antibodies, that they are fully protected from reinfection, or how long any immunity lasts.”
The FDA says that the role of these antibody tests, at present, lies in providing information to “help us track the spread of the virus nationwide and assess the impact of our public health efforts now, while also informing our COVID-19 response as we continue to move forward.”
The World Health Organization (WHO) also emphasized the current uncertainty over antibody tests at a press briefing on April 17. “Nobody is sure about the length of protection that antibodies may give and whether they fully protect against ... the disease,” said Mike Ryan, MD, executive director of the WHO’s emergencies program. There is also a concern that such tests may give false assurance or be misused. “There is still a lot of work that needs to be done to validate these antibody tests,” he added.
“The WHO are right to highlight that any antibody test, if we get one, won’t be able to definitely say whether someone is immune to the infection, because we just don’t know enough yet about how immunity works with COVID-19,” commented Prof. Chris Dye, Oxford Martin School, University of Oxford, in reaction on the UK Science Media Center.
Expanding on this point on the same site, Andrew Easton PhD, professor of virology at the University of Warwick, noted that “a serology test does not discriminate between neutralising and non-neutralising antibodies; a discriminatory test is much more complex and slow.”
Only the neutralizing antibodies have the ability to inactivate the invading virus, he noted.
“When people are infected, the proportions of neutralising and non-neutralising antibodies can differ. It is not always understood what makes an antibody neutralising and another non-neutralising, or why an infection leads to production of more of one of these types of antibodies,” he explained. “The initial immune response immediately following infection sets the memory of the immune system, so if the person had generated mostly non-neutralising antibodies, the next time that person encounters the same virus, they may not be able to prevent an infection.”
So at present, the information from antibody testing is largely unhelpful to individuals, but it could be valuable to epidemiologists and policy makers.
“States are looking at ways they can integrate reliable serologic tests for surveillance,” explained APHL’s Blank.
Knowing how widespread the infection has been within a community could guide research and possibly public health decisions, Wroblewski said at the APHL press conference. But she’s hesitant here, too. “I know there has been a lot of talk about using this testing to ease restrictions, but I do think we need to be cautious on how quickly we move in that direction.” If people don’t have antibodies, it means they haven’t been exposed and that they’re still vulnerable, she noted. “If nothing else, that still informs policy decisions, even if they’re not the policy decisions we want.”
Trials Recruiting, Medical Centers Develop Own Tests
Despite the uncertainties over antibody testing, many efforts are still being guided by this strategy.
The NIH is recruiting volunteers to its antibody testing study and suggests that immunity is “likely” for those who test positive.
In addition, several large medical centers have developed their own antibody tests, including Stanford, the Yale New Haven Hospital, and the Mayo Clinic.
The Stanford test detects two types of antibodies: IgM, which is made early in an immune response and usually wanes quickly, and IgG, which rises more slowly after infection but usually persists longer.
“There’s limited data out of China and Europe showing that this appears to be the response pattern followed with this virus,” commented Thomas Montine, MD, PhD, professor and chair of pathology at Stanford University. “But no one has had this long enough to know how long after infection the antibodies persist,” he added.
“There is enormous demand for serologic testing,” said William Morice, MD, PhD, president of Mayo Clinic Laboratories. “At this time, serology testing needs to be prioritized for efforts to identify individuals in areas where potential immunity is key ― supporting healthcare workers, screening for potential plasma donors, and helping advance the most promising vaccine candidates.”
During a recent webinar with the Association for Value-Based Cancer Care, the largest physician-owned oncology-hematology practice in the country, the president, Lucio Gordan, MD, said his organization was looking into antibody testing for staff. “They wanted to see how many have been exposed,” he said, although “what it means is uncertain.”
When Medscape Medical News checked back with him a few weeks later, Gordan, president of Florida Cancer Specialists and Research Institute, reported that no progress had been made.
“We unfortunately have not been able to test yet, due to concerns with reliability of kits. We are waiting for a better solution so we can reassess our strategy,” he said.
This article first appeared on Medscape.com.
Noopur Raje, MD, has been sitting at home for 5 weeks waiting for her COVID-19 test to turn negative so she can get back to work. She’s a cancer specialist – head of the Massachusetts General Hospital’s Center for Multiple Myeloma – but Raje says as soon as she’s allowed back to the hospital, she’ll head straight to the front line of COVID-19 caregivers.
“It’s people like us who have to get back in the trenches and do the work now,” she told Medscape Medical News.
“I still will be at risk,” she said. But, having nursed her physician husband through COVID-19 at home until he was admitted to an intensive care unit, she is determined to help in the COVID-19 wards.
“I will be the first one to volunteer to take care of these patients,” she said. “I can’t wait, as I want to give these folks hope. They are so scared.”
Around the world, it’s assumed that she and others like her who’ve recovered from COVID-19 will be immune to the infection.
Some have suggested that with antibodies to the virus coursing through their veins, these survivors might be given immunity passports. They could be the ones to jump-start people’s lives again ― the first to be let out from lockdown, and in healthcare, the ones to head the ongoing battle against this pandemic.
So, there has been a race to develop COVID-19 antibody tests to identify these people.
Circumventing the Usual Clearance Process
To speed up the process, the US Food and Drug Administration (FDA) made a much-criticized move to allow a free-for-all for developers to begin marketing antibody tests that had not gone through the agency’s usual evaluation process. The result was a flood of more than 90 unapproved tests “that have, frankly, dubious quality,” said Scott Becker, CEO of the Association of Public Health Laboratories (APHL), which represents local and state public laboratories.
The APHL spoke out in dismay – its chief program officer, Eric Blank, decried the “Wild West” of tests unleashed on the public.
“These tests create more uncertainty than before,” said Kelly Wroblewski, APHL’s director of infectious diseases, in a news conference on April 14. “Having many inaccurate tests is worse than having no tests at all.”
The APHL and the FDA, working with the Centers for Disease Control and Prevention and the National Institutes of Health (NIH), have moved quickly into damage control, conducting evaluations of the tests in an effort to distinguish the potentially useful from the useless.
So far, they have succeeded in issuing emergency use authorizations (EUAs) to only four tests, those marketed by Cellex, Ortho Clinical Diagnostics, Chembio Diagnostic Systems, and the Mount Sinai Laboratory.
For all the other antibody tests on the market that do not have an EUA, “They’re trusting that the test developer has done a good job in validation,” Becker said. But there are worrying anecdotes. “Our members have reported that they’ve seen fraudulent marketing.... We’ve seen the FDA clamp down on some companies... [and] a number of cities and health departments have issued warnings because of what they’ve seen,” he added.
In particular, Wroblewski said, some companies are marketing tests for use in physicians’ offices or pharmacies. “Today, there are no serology tests approved for point-of-care settings,” she warned. “We don’t know how to interpret the test results, if the presence of antibodies indicates immunity, how long it will last, or what titer might be sufficient.”
Uncertainty Emphasized
The FDA emphasized the uncertainty about antibody tests in a statement released on April 18.
Although the tests can identify people who have been exposed and who developed an immune response to the virus, the agency noted, “we don’t yet know that just because someone has developed antibodies, that they are fully protected from reinfection, or how long any immunity lasts.”
The FDA says that the role of these antibody tests, at present, lies in providing information to “help us track the spread of the virus nationwide and assess the impact of our public health efforts now, while also informing our COVID-19 response as we continue to move forward.”
The World Health Organization (WHO) also emphasized the current uncertainty over antibody tests at a press briefing on April 17. “Nobody is sure about the length of protection that antibodies may give and whether they fully protect against ... the disease,” said Mike Ryan, MD, executive director of the WHO’s emergencies program. There is also a concern that such tests may give false assurance or be misused. “There is still a lot of work that needs to be done to validate these antibody tests,” he added.
“The WHO are right to highlight that any antibody test, if we get one, won’t be able to definitely say whether someone is immune to the infection, because we just don’t know enough yet about how immunity works with COVID-19,” commented Prof. Chris Dye, Oxford Martin School, University of Oxford, in reaction on the UK Science Media Center.
Expanding on this point on the same site, Andrew Easton PhD, professor of virology at the University of Warwick, noted that “a serology test does not discriminate between neutralising and non-neutralising antibodies; a discriminatory test is much more complex and slow.”
Only the neutralizing antibodies have the ability to inactivate the invading virus, he noted.
“When people are infected, the proportions of neutralising and non-neutralising antibodies can differ. It is not always understood what makes an antibody neutralising and another non-neutralising, or why an infection leads to production of more of one of these types of antibodies,” he explained. “The initial immune response immediately following infection sets the memory of the immune system, so if the person had generated mostly non-neutralising antibodies, the next time that person encounters the same virus, they may not be able to prevent an infection.”
So at present, the information from antibody testing is largely unhelpful to individuals, but it could be valuable to epidemiologists and policy makers.
“States are looking at ways they can integrate reliable serologic tests for surveillance,” explained APHL’s Blank.
Knowing how widespread the infection has been within a community could guide research and possibly public health decisions, Wroblewski said at the APHL press conference. But she’s hesitant here, too. “I know there has been a lot of talk about using this testing to ease restrictions, but I do think we need to be cautious on how quickly we move in that direction.” If people don’t have antibodies, it means they haven’t been exposed and that they’re still vulnerable, she noted. “If nothing else, that still informs policy decisions, even if they’re not the policy decisions we want.”
Trials Recruiting, Medical Centers Develop Own Tests
Despite the uncertainties over antibody testing, many efforts are still being guided by this strategy.
The NIH is recruiting volunteers to its antibody testing study and suggests that immunity is “likely” for those who test positive.
In addition, several large medical centers have developed their own antibody tests, including Stanford, the Yale New Haven Hospital, and the Mayo Clinic.
The Stanford test detects two types of antibodies: IgM, which is made early in an immune response and usually wanes quickly, and IgG, which rises more slowly after infection but usually persists longer.
“There’s limited data out of China and Europe showing that this appears to be the response pattern followed with this virus,” commented Thomas Montine, MD, PhD, professor and chair of pathology at Stanford University. “But no one has had this long enough to know how long after infection the antibodies persist,” he added.
“There is enormous demand for serologic testing,” said William Morice, MD, PhD, president of Mayo Clinic Laboratories. “At this time, serology testing needs to be prioritized for efforts to identify individuals in areas where potential immunity is key ― supporting healthcare workers, screening for potential plasma donors, and helping advance the most promising vaccine candidates.”
During a recent webinar with the Association for Value-Based Cancer Care, the largest physician-owned oncology-hematology practice in the country, the president, Lucio Gordan, MD, said his organization was looking into antibody testing for staff. “They wanted to see how many have been exposed,” he said, although “what it means is uncertain.”
When Medscape Medical News checked back with him a few weeks later, Gordan, president of Florida Cancer Specialists and Research Institute, reported that no progress had been made.
“We unfortunately have not been able to test yet, due to concerns with reliability of kits. We are waiting for a better solution so we can reassess our strategy,” he said.
This article first appeared on Medscape.com.
Noopur Raje, MD, has been sitting at home for 5 weeks waiting for her COVID-19 test to turn negative so she can get back to work. She’s a cancer specialist – head of the Massachusetts General Hospital’s Center for Multiple Myeloma – but Raje says as soon as she’s allowed back to the hospital, she’ll head straight to the front line of COVID-19 caregivers.
“It’s people like us who have to get back in the trenches and do the work now,” she told Medscape Medical News.
“I still will be at risk,” she said. But, having nursed her physician husband through COVID-19 at home until he was admitted to an intensive care unit, she is determined to help in the COVID-19 wards.
“I will be the first one to volunteer to take care of these patients,” she said. “I can’t wait, as I want to give these folks hope. They are so scared.”
Around the world, it’s assumed that she and others like her who’ve recovered from COVID-19 will be immune to the infection.
Some have suggested that with antibodies to the virus coursing through their veins, these survivors might be given immunity passports. They could be the ones to jump-start people’s lives again ― the first to be let out from lockdown, and in healthcare, the ones to head the ongoing battle against this pandemic.
So, there has been a race to develop COVID-19 antibody tests to identify these people.
Circumventing the Usual Clearance Process
To speed up the process, the US Food and Drug Administration (FDA) made a much-criticized move to allow a free-for-all for developers to begin marketing antibody tests that had not gone through the agency’s usual evaluation process. The result was a flood of more than 90 unapproved tests “that have, frankly, dubious quality,” said Scott Becker, CEO of the Association of Public Health Laboratories (APHL), which represents local and state public laboratories.
The APHL spoke out in dismay – its chief program officer, Eric Blank, decried the “Wild West” of tests unleashed on the public.
“These tests create more uncertainty than before,” said Kelly Wroblewski, APHL’s director of infectious diseases, in a news conference on April 14. “Having many inaccurate tests is worse than having no tests at all.”
The APHL and the FDA, working with the Centers for Disease Control and Prevention and the National Institutes of Health (NIH), have moved quickly into damage control, conducting evaluations of the tests in an effort to distinguish the potentially useful from the useless.
So far, they have succeeded in issuing emergency use authorizations (EUAs) to only four tests, those marketed by Cellex, Ortho Clinical Diagnostics, Chembio Diagnostic Systems, and the Mount Sinai Laboratory.
For all the other antibody tests on the market that do not have an EUA, “They’re trusting that the test developer has done a good job in validation,” Becker said. But there are worrying anecdotes. “Our members have reported that they’ve seen fraudulent marketing.... We’ve seen the FDA clamp down on some companies... [and] a number of cities and health departments have issued warnings because of what they’ve seen,” he added.
In particular, Wroblewski said, some companies are marketing tests for use in physicians’ offices or pharmacies. “Today, there are no serology tests approved for point-of-care settings,” she warned. “We don’t know how to interpret the test results, if the presence of antibodies indicates immunity, how long it will last, or what titer might be sufficient.”
Uncertainty Emphasized
The FDA emphasized the uncertainty about antibody tests in a statement released on April 18.
Although the tests can identify people who have been exposed and who developed an immune response to the virus, the agency noted, “we don’t yet know that just because someone has developed antibodies, that they are fully protected from reinfection, or how long any immunity lasts.”
The FDA says that the role of these antibody tests, at present, lies in providing information to “help us track the spread of the virus nationwide and assess the impact of our public health efforts now, while also informing our COVID-19 response as we continue to move forward.”
The World Health Organization (WHO) also emphasized the current uncertainty over antibody tests at a press briefing on April 17. “Nobody is sure about the length of protection that antibodies may give and whether they fully protect against ... the disease,” said Mike Ryan, MD, executive director of the WHO’s emergencies program. There is also a concern that such tests may give false assurance or be misused. “There is still a lot of work that needs to be done to validate these antibody tests,” he added.
“The WHO are right to highlight that any antibody test, if we get one, won’t be able to definitely say whether someone is immune to the infection, because we just don’t know enough yet about how immunity works with COVID-19,” commented Prof. Chris Dye, Oxford Martin School, University of Oxford, in reaction on the UK Science Media Center.
Expanding on this point on the same site, Andrew Easton PhD, professor of virology at the University of Warwick, noted that “a serology test does not discriminate between neutralising and non-neutralising antibodies; a discriminatory test is much more complex and slow.”
Only the neutralizing antibodies have the ability to inactivate the invading virus, he noted.
“When people are infected, the proportions of neutralising and non-neutralising antibodies can differ. It is not always understood what makes an antibody neutralising and another non-neutralising, or why an infection leads to production of more of one of these types of antibodies,” he explained. “The initial immune response immediately following infection sets the memory of the immune system, so if the person had generated mostly non-neutralising antibodies, the next time that person encounters the same virus, they may not be able to prevent an infection.”
So at present, the information from antibody testing is largely unhelpful to individuals, but it could be valuable to epidemiologists and policy makers.
“States are looking at ways they can integrate reliable serologic tests for surveillance,” explained APHL’s Blank.
Knowing how widespread the infection has been within a community could guide research and possibly public health decisions, Wroblewski said at the APHL press conference. But she’s hesitant here, too. “I know there has been a lot of talk about using this testing to ease restrictions, but I do think we need to be cautious on how quickly we move in that direction.” If people don’t have antibodies, it means they haven’t been exposed and that they’re still vulnerable, she noted. “If nothing else, that still informs policy decisions, even if they’re not the policy decisions we want.”
Trials Recruiting, Medical Centers Develop Own Tests
Despite the uncertainties over antibody testing, many efforts are still being guided by this strategy.
The NIH is recruiting volunteers to its antibody testing study and suggests that immunity is “likely” for those who test positive.
In addition, several large medical centers have developed their own antibody tests, including Stanford, the Yale New Haven Hospital, and the Mayo Clinic.
The Stanford test detects two types of antibodies: IgM, which is made early in an immune response and usually wanes quickly, and IgG, which rises more slowly after infection but usually persists longer.
“There’s limited data out of China and Europe showing that this appears to be the response pattern followed with this virus,” commented Thomas Montine, MD, PhD, professor and chair of pathology at Stanford University. “But no one has had this long enough to know how long after infection the antibodies persist,” he added.
“There is enormous demand for serologic testing,” said William Morice, MD, PhD, president of Mayo Clinic Laboratories. “At this time, serology testing needs to be prioritized for efforts to identify individuals in areas where potential immunity is key ― supporting healthcare workers, screening for potential plasma donors, and helping advance the most promising vaccine candidates.”
During a recent webinar with the Association for Value-Based Cancer Care, the largest physician-owned oncology-hematology practice in the country, the president, Lucio Gordan, MD, said his organization was looking into antibody testing for staff. “They wanted to see how many have been exposed,” he said, although “what it means is uncertain.”
When Medscape Medical News checked back with him a few weeks later, Gordan, president of Florida Cancer Specialists and Research Institute, reported that no progress had been made.
“We unfortunately have not been able to test yet, due to concerns with reliability of kits. We are waiting for a better solution so we can reassess our strategy,” he said.
This article first appeared on Medscape.com.
Sudden loss of taste and smell should be part of COVID-19 screen
A number of new publications show that a high proportion of people infected with COVID-19 report loss of smell and/or taste, with their authors adding to the clamor to recognize these symptoms as potentially indicative of the infection.
In particular, there is a belief that these signs may be present in many with asymptomatic COVID-19, and therefore asking about them could be a way to prioritize people for initial testing for the SARS-CoV-2 virus in the absence of other symptoms.
Anyone testing positive could then quarantine, and their contacts could be traced.
Despite this, the World Health Organization (WHO) has not listed loss of smell or taste as potential symptoms of SARS-CoV-2 infection.
But the US Centers for Disease Control and Prevention (CDC) has now added “new loss of taste or smell” as a symptom on its COVID-19 information page.
American Academy of Otolaryngology—Head and Neck Surgery (AAO-HNS) executive vice president and CEO James C. Denneny III, MD, believes the symptoms may be an early warning signal.
And there’s no downside to checking for these, Denneny told Medscape Medical News.
“Given the fact that this doesn’t require any surgical procedure, biopsy, or specific treatment, I think the upside of getting it early is great,” he said. “The downside of using it as a symptom, and if someone doesn’t turn out to have it, is virtually zero.”
Claire Hopkins, MD, president of the British Rhinological Society, and colleagues, writing in Lancet Infectious Diseases, agree.
“Physicians evaluating patients with acute-onset loss of smell or taste, particularly in the context of a patent nasal airway, should have a high index of suspicion for concomitant SARS-CoV-2 infection.”
They also observe that this appears to occur, in contrast to other respiratory infections, “in the absence of nasal congestion or rhinorrhea.”
Newest Publications Find Smell and Taste Loss Is Common
Author of one of the newly published studies, Carol H. Yan, MD, an otolaryngologist and head and neck surgeon at the University of California, San Diego, also thinks that sudden smell and taste loss seem to be fairly specific markers of COVID-19.
In her survey of patients who presented to UC San Diego Health for SARS-CoV-2 testing, Yan and colleagues reported that 68% (40 of 59) of COVID-19–positive patients reported olfactory impairment and 71% (42 of 59) reported taste impairment.
Among the 203 people in the “control” group who were polymerase chain reaction–negative (PCR–) for SARS-CoV-2, just 16% had smell loss and 17% had taste loss, according to their results published in the International Forum of Allergy & Rhinology.
“Based on our study, if you have smell and taste loss, you are more than 10 times more likely to have COVID-19 infection than other causes of infection. The most common first sign of a COVID-19 infection remains fever, but fatigue and loss of smell and taste follow as other very common initial symptoms,” said Yan.
“We know COVID-19 is an extremely contagious virus. This study supports the need to be aware of smell and taste loss as early signs of COVID-19.”
Yan told Medscape Medical News that another not-yet-published analysis indicates that sudden loss of smell or taste “may be more representative of a mild form of disease.”
Getting these people tested and isolated could therefore help prevent spread of COVID-19, she urged.
Based on Yan’s report and other case reports, the UC San Diego Health system is now asking all callers to its COVID-19 hotlines, and all visitors and staff, if they’ve had a sudden loss of taste or smell in the last few weeks, she explained.
And Ahmad R. Sedaghat, MD, PhD, at the University of Cincinnati, Ohio, takes a similar view.
In a new systematic review of the topic published April 14 in Laryngoscope Investigative Otolaryngology, Sedaghat and colleagues write: “Anosmia (total loss of smell) without nasal obstruction, in particular, appears to be a highly specific indicator of COVID-19.”
Sedaghat said a sudden loss of sense of smell wouldn’t necessarily lead people to think they have COVID-19, particularly if they remain asymptomatic, so “these individuals could continue business as usual and spread the disease as a carrier.”
“If someone experiences anosmia without nasal obstruction, aside from quarantining, it would not be unreasonable to reach out to one’s primary care physician about getting tested,” he said in a statement from his institution.
Symptom Checkers Add Weight
Several organizations around the world have begun collecting symptom reports from patients and clinicians, which has shone more light on the sudden loss of taste and smell as potential flags for COVID-19.
In an April 14 Morbidity and Mortality Weekly report from the CDC on COVID-19 infections in healthcare workers, of the 5000 who reported symptoms, 750 (16%) wrote “loss of smell or taste” as an “other” symptom.
Meanwhile, the COVID Symptom Tracker smartphone app, a joint effort by Massachusetts General Hospital, Boston, Stanford (Calif.) University, and King’s College, London, which as of press time, was monitoring some 2.5 million people, has had similar findings.
In a preprint publication on 400,000 people reporting one or more symptoms between March 24 and 29 on the tracker, 18% had lost their sense of smell or taste — more than the 10% who reported fever, but far less than the 53% who reported fatigue.
Only 1702 of the 400,000 had received a COVID-19 test.
Of those, 579 had tested positive and 1123 were negative.
The organizers estimated that of those who were positive, 59% reported losing smell or taste, compared with just 18% who tested negative.
“When combined with other symptoms, people with loss of smell and taste appear to be three times more likely to have contracted COVID-19 according to our data,” said Tim Spector, MD, a genetic epidemiologist at King’s College and the app’s lead researcher, on the symptom tracker’s website.
These people “should therefore self-isolate for 7 days to reduce the spread of the disease,” he urged.
Anosmia Is the Initial Symptom in Many Patients With COVID-19
The AAO-HNS also began collecting data from physicians and patients on March 25 through its Web-based 16-question symptom tracking tool.
It has received more than 500 reports of sudden taste or smell loss, said Denneny.
In a report on the first 237 responses, published in Otolaryngology-Head and Neck Surgery, anosmia (profound loss of smell) was found in 73% of subjects before a COVID-19 diagnosis and was the initial symptom in 27% of those subjects.
That latter determination “was the single most important finding,” said Denneny, noting it shows that smell and taste loss are “a sentinel symptom.”
Anosmia led to testing in only 40% of the cases.
Half of the reports came from otolaryngologists, but a large number came from other medical specialties, especially from family medicine.
Just 2% of reports came from patients in that first group, which was based on responses through April 3.
Denneny said that more reports are now coming in from patients, which he attributes to widespread media coverage about the loss of taste and smell.
It’s still not entirely clear why SARS-CoV-2 might inhibit taste or smell. More common viruses like influenza and other coronaviruses can also cause smell and taste loss.
So far, it seems like the sensory recovery is faster for SARS-CoV-2 than the other viruses, which suggests a potentially different mechanism of action, said Yan. Patients she surveyed at UC San Diego recovered the senses within a few weeks to a month, compared to months or a year with the more common viruses.
Yan’s study was partially supported by the National Institutes of Health. Sedaghat has reported no relevant financial relationships. The COVID Symptom Tracker is supported by Zoe Global Limited and has received grants from the Wellcome Trust, Medical Research Council/British Heart Foundation, and Biological Informative Markers for Stratification of Hypertension.
This article first appeared on Medscape.com.
A number of new publications show that a high proportion of people infected with COVID-19 report loss of smell and/or taste, with their authors adding to the clamor to recognize these symptoms as potentially indicative of the infection.
In particular, there is a belief that these signs may be present in many with asymptomatic COVID-19, and therefore asking about them could be a way to prioritize people for initial testing for the SARS-CoV-2 virus in the absence of other symptoms.
Anyone testing positive could then quarantine, and their contacts could be traced.
Despite this, the World Health Organization (WHO) has not listed loss of smell or taste as potential symptoms of SARS-CoV-2 infection.
But the US Centers for Disease Control and Prevention (CDC) has now added “new loss of taste or smell” as a symptom on its COVID-19 information page.
American Academy of Otolaryngology—Head and Neck Surgery (AAO-HNS) executive vice president and CEO James C. Denneny III, MD, believes the symptoms may be an early warning signal.
And there’s no downside to checking for these, Denneny told Medscape Medical News.
“Given the fact that this doesn’t require any surgical procedure, biopsy, or specific treatment, I think the upside of getting it early is great,” he said. “The downside of using it as a symptom, and if someone doesn’t turn out to have it, is virtually zero.”
Claire Hopkins, MD, president of the British Rhinological Society, and colleagues, writing in Lancet Infectious Diseases, agree.
“Physicians evaluating patients with acute-onset loss of smell or taste, particularly in the context of a patent nasal airway, should have a high index of suspicion for concomitant SARS-CoV-2 infection.”
They also observe that this appears to occur, in contrast to other respiratory infections, “in the absence of nasal congestion or rhinorrhea.”
Newest Publications Find Smell and Taste Loss Is Common
Author of one of the newly published studies, Carol H. Yan, MD, an otolaryngologist and head and neck surgeon at the University of California, San Diego, also thinks that sudden smell and taste loss seem to be fairly specific markers of COVID-19.
In her survey of patients who presented to UC San Diego Health for SARS-CoV-2 testing, Yan and colleagues reported that 68% (40 of 59) of COVID-19–positive patients reported olfactory impairment and 71% (42 of 59) reported taste impairment.
Among the 203 people in the “control” group who were polymerase chain reaction–negative (PCR–) for SARS-CoV-2, just 16% had smell loss and 17% had taste loss, according to their results published in the International Forum of Allergy & Rhinology.
“Based on our study, if you have smell and taste loss, you are more than 10 times more likely to have COVID-19 infection than other causes of infection. The most common first sign of a COVID-19 infection remains fever, but fatigue and loss of smell and taste follow as other very common initial symptoms,” said Yan.
“We know COVID-19 is an extremely contagious virus. This study supports the need to be aware of smell and taste loss as early signs of COVID-19.”
Yan told Medscape Medical News that another not-yet-published analysis indicates that sudden loss of smell or taste “may be more representative of a mild form of disease.”
Getting these people tested and isolated could therefore help prevent spread of COVID-19, she urged.
Based on Yan’s report and other case reports, the UC San Diego Health system is now asking all callers to its COVID-19 hotlines, and all visitors and staff, if they’ve had a sudden loss of taste or smell in the last few weeks, she explained.
And Ahmad R. Sedaghat, MD, PhD, at the University of Cincinnati, Ohio, takes a similar view.
In a new systematic review of the topic published April 14 in Laryngoscope Investigative Otolaryngology, Sedaghat and colleagues write: “Anosmia (total loss of smell) without nasal obstruction, in particular, appears to be a highly specific indicator of COVID-19.”
Sedaghat said a sudden loss of sense of smell wouldn’t necessarily lead people to think they have COVID-19, particularly if they remain asymptomatic, so “these individuals could continue business as usual and spread the disease as a carrier.”
“If someone experiences anosmia without nasal obstruction, aside from quarantining, it would not be unreasonable to reach out to one’s primary care physician about getting tested,” he said in a statement from his institution.
Symptom Checkers Add Weight
Several organizations around the world have begun collecting symptom reports from patients and clinicians, which has shone more light on the sudden loss of taste and smell as potential flags for COVID-19.
In an April 14 Morbidity and Mortality Weekly report from the CDC on COVID-19 infections in healthcare workers, of the 5000 who reported symptoms, 750 (16%) wrote “loss of smell or taste” as an “other” symptom.
Meanwhile, the COVID Symptom Tracker smartphone app, a joint effort by Massachusetts General Hospital, Boston, Stanford (Calif.) University, and King’s College, London, which as of press time, was monitoring some 2.5 million people, has had similar findings.
In a preprint publication on 400,000 people reporting one or more symptoms between March 24 and 29 on the tracker, 18% had lost their sense of smell or taste — more than the 10% who reported fever, but far less than the 53% who reported fatigue.
Only 1702 of the 400,000 had received a COVID-19 test.
Of those, 579 had tested positive and 1123 were negative.
The organizers estimated that of those who were positive, 59% reported losing smell or taste, compared with just 18% who tested negative.
“When combined with other symptoms, people with loss of smell and taste appear to be three times more likely to have contracted COVID-19 according to our data,” said Tim Spector, MD, a genetic epidemiologist at King’s College and the app’s lead researcher, on the symptom tracker’s website.
These people “should therefore self-isolate for 7 days to reduce the spread of the disease,” he urged.
Anosmia Is the Initial Symptom in Many Patients With COVID-19
The AAO-HNS also began collecting data from physicians and patients on March 25 through its Web-based 16-question symptom tracking tool.
It has received more than 500 reports of sudden taste or smell loss, said Denneny.
In a report on the first 237 responses, published in Otolaryngology-Head and Neck Surgery, anosmia (profound loss of smell) was found in 73% of subjects before a COVID-19 diagnosis and was the initial symptom in 27% of those subjects.
That latter determination “was the single most important finding,” said Denneny, noting it shows that smell and taste loss are “a sentinel symptom.”
Anosmia led to testing in only 40% of the cases.
Half of the reports came from otolaryngologists, but a large number came from other medical specialties, especially from family medicine.
Just 2% of reports came from patients in that first group, which was based on responses through April 3.
Denneny said that more reports are now coming in from patients, which he attributes to widespread media coverage about the loss of taste and smell.
It’s still not entirely clear why SARS-CoV-2 might inhibit taste or smell. More common viruses like influenza and other coronaviruses can also cause smell and taste loss.
So far, it seems like the sensory recovery is faster for SARS-CoV-2 than the other viruses, which suggests a potentially different mechanism of action, said Yan. Patients she surveyed at UC San Diego recovered the senses within a few weeks to a month, compared to months or a year with the more common viruses.
Yan’s study was partially supported by the National Institutes of Health. Sedaghat has reported no relevant financial relationships. The COVID Symptom Tracker is supported by Zoe Global Limited and has received grants from the Wellcome Trust, Medical Research Council/British Heart Foundation, and Biological Informative Markers for Stratification of Hypertension.
This article first appeared on Medscape.com.
A number of new publications show that a high proportion of people infected with COVID-19 report loss of smell and/or taste, with their authors adding to the clamor to recognize these symptoms as potentially indicative of the infection.
In particular, there is a belief that these signs may be present in many with asymptomatic COVID-19, and therefore asking about them could be a way to prioritize people for initial testing for the SARS-CoV-2 virus in the absence of other symptoms.
Anyone testing positive could then quarantine, and their contacts could be traced.
Despite this, the World Health Organization (WHO) has not listed loss of smell or taste as potential symptoms of SARS-CoV-2 infection.
But the US Centers for Disease Control and Prevention (CDC) has now added “new loss of taste or smell” as a symptom on its COVID-19 information page.
American Academy of Otolaryngology—Head and Neck Surgery (AAO-HNS) executive vice president and CEO James C. Denneny III, MD, believes the symptoms may be an early warning signal.
And there’s no downside to checking for these, Denneny told Medscape Medical News.
“Given the fact that this doesn’t require any surgical procedure, biopsy, or specific treatment, I think the upside of getting it early is great,” he said. “The downside of using it as a symptom, and if someone doesn’t turn out to have it, is virtually zero.”
Claire Hopkins, MD, president of the British Rhinological Society, and colleagues, writing in Lancet Infectious Diseases, agree.
“Physicians evaluating patients with acute-onset loss of smell or taste, particularly in the context of a patent nasal airway, should have a high index of suspicion for concomitant SARS-CoV-2 infection.”
They also observe that this appears to occur, in contrast to other respiratory infections, “in the absence of nasal congestion or rhinorrhea.”
Newest Publications Find Smell and Taste Loss Is Common
Author of one of the newly published studies, Carol H. Yan, MD, an otolaryngologist and head and neck surgeon at the University of California, San Diego, also thinks that sudden smell and taste loss seem to be fairly specific markers of COVID-19.
In her survey of patients who presented to UC San Diego Health for SARS-CoV-2 testing, Yan and colleagues reported that 68% (40 of 59) of COVID-19–positive patients reported olfactory impairment and 71% (42 of 59) reported taste impairment.
Among the 203 people in the “control” group who were polymerase chain reaction–negative (PCR–) for SARS-CoV-2, just 16% had smell loss and 17% had taste loss, according to their results published in the International Forum of Allergy & Rhinology.
“Based on our study, if you have smell and taste loss, you are more than 10 times more likely to have COVID-19 infection than other causes of infection. The most common first sign of a COVID-19 infection remains fever, but fatigue and loss of smell and taste follow as other very common initial symptoms,” said Yan.
“We know COVID-19 is an extremely contagious virus. This study supports the need to be aware of smell and taste loss as early signs of COVID-19.”
Yan told Medscape Medical News that another not-yet-published analysis indicates that sudden loss of smell or taste “may be more representative of a mild form of disease.”
Getting these people tested and isolated could therefore help prevent spread of COVID-19, she urged.
Based on Yan’s report and other case reports, the UC San Diego Health system is now asking all callers to its COVID-19 hotlines, and all visitors and staff, if they’ve had a sudden loss of taste or smell in the last few weeks, she explained.
And Ahmad R. Sedaghat, MD, PhD, at the University of Cincinnati, Ohio, takes a similar view.
In a new systematic review of the topic published April 14 in Laryngoscope Investigative Otolaryngology, Sedaghat and colleagues write: “Anosmia (total loss of smell) without nasal obstruction, in particular, appears to be a highly specific indicator of COVID-19.”
Sedaghat said a sudden loss of sense of smell wouldn’t necessarily lead people to think they have COVID-19, particularly if they remain asymptomatic, so “these individuals could continue business as usual and spread the disease as a carrier.”
“If someone experiences anosmia without nasal obstruction, aside from quarantining, it would not be unreasonable to reach out to one’s primary care physician about getting tested,” he said in a statement from his institution.
Symptom Checkers Add Weight
Several organizations around the world have begun collecting symptom reports from patients and clinicians, which has shone more light on the sudden loss of taste and smell as potential flags for COVID-19.
In an April 14 Morbidity and Mortality Weekly report from the CDC on COVID-19 infections in healthcare workers, of the 5000 who reported symptoms, 750 (16%) wrote “loss of smell or taste” as an “other” symptom.
Meanwhile, the COVID Symptom Tracker smartphone app, a joint effort by Massachusetts General Hospital, Boston, Stanford (Calif.) University, and King’s College, London, which as of press time, was monitoring some 2.5 million people, has had similar findings.
In a preprint publication on 400,000 people reporting one or more symptoms between March 24 and 29 on the tracker, 18% had lost their sense of smell or taste — more than the 10% who reported fever, but far less than the 53% who reported fatigue.
Only 1702 of the 400,000 had received a COVID-19 test.
Of those, 579 had tested positive and 1123 were negative.
The organizers estimated that of those who were positive, 59% reported losing smell or taste, compared with just 18% who tested negative.
“When combined with other symptoms, people with loss of smell and taste appear to be three times more likely to have contracted COVID-19 according to our data,” said Tim Spector, MD, a genetic epidemiologist at King’s College and the app’s lead researcher, on the symptom tracker’s website.
These people “should therefore self-isolate for 7 days to reduce the spread of the disease,” he urged.
Anosmia Is the Initial Symptom in Many Patients With COVID-19
The AAO-HNS also began collecting data from physicians and patients on March 25 through its Web-based 16-question symptom tracking tool.
It has received more than 500 reports of sudden taste or smell loss, said Denneny.
In a report on the first 237 responses, published in Otolaryngology-Head and Neck Surgery, anosmia (profound loss of smell) was found in 73% of subjects before a COVID-19 diagnosis and was the initial symptom in 27% of those subjects.
That latter determination “was the single most important finding,” said Denneny, noting it shows that smell and taste loss are “a sentinel symptom.”
Anosmia led to testing in only 40% of the cases.
Half of the reports came from otolaryngologists, but a large number came from other medical specialties, especially from family medicine.
Just 2% of reports came from patients in that first group, which was based on responses through April 3.
Denneny said that more reports are now coming in from patients, which he attributes to widespread media coverage about the loss of taste and smell.
It’s still not entirely clear why SARS-CoV-2 might inhibit taste or smell. More common viruses like influenza and other coronaviruses can also cause smell and taste loss.
So far, it seems like the sensory recovery is faster for SARS-CoV-2 than the other viruses, which suggests a potentially different mechanism of action, said Yan. Patients she surveyed at UC San Diego recovered the senses within a few weeks to a month, compared to months or a year with the more common viruses.
Yan’s study was partially supported by the National Institutes of Health. Sedaghat has reported no relevant financial relationships. The COVID Symptom Tracker is supported by Zoe Global Limited and has received grants from the Wellcome Trust, Medical Research Council/British Heart Foundation, and Biological Informative Markers for Stratification of Hypertension.
This article first appeared on Medscape.com.
COVID-19: Experts hasten to head off mental health crisis
The COVID-19 pandemic is already affecting mental health at a population level, with increased anxiety, feelings of isolation, and concerns about access to mental health care.
Two U.K. surveys were conducted to inform research priorities for mental health research and in an effort to head off a mental health crisis. The U.K. charity MQ conducted a “stakeholder” survey of 2,198 individuals who had a lived experience of mental illness, while Ipsos MORI conducted a poll of 1,099 members of the public.
The online surveys were conducted in late March, the same week the U.K.’s nationwide lockdown measures were announced. Respondents were asked about their biggest mental health and well-being concerns and coping strategies as they relate to the COVID-19 pandemic.
Results showed that across the two surveys, respondents’ primary concern was anxiety, which was cited in 750 responses.
In addition, respondents were worried about being social isolated, becoming mentally unwell, and having a lack of access to mental health services, as well as the impact of the pandemic on personal relationships.
The findings were used by a panel of experts to inform a position paper published in the Lancet Psychiatry. The paper outlines a proposed government response to curb the long-term “profound” and “pervasive” impact of the pandemic on mental health.
‘Unprecedented response’ needed
“Governments must find evidence-based ways to boost the resilience of our societies and ... to treat those with mental ill health remotely to come out of this pandemic in good mental health,” coauthor of the paper Emily A. Holmes, PhD, of the department of psychology at Uppsala (Sweden) University, said in a press release.
“Frontline medical staff and vulnerable groups such as the elderly and those with serious mental health conditions must be prioritized for rapid mental health support,” she added.
The position paper authors call for “moment-to-moment” monitoring of anxiety, depression, self-harm, and suicide, as well as using digital technology and rapid deployment of evidence-based programs and treatments.
Patients will need to be accessible via computer, cell phone, and other remote technologies in order to receive treatment during physical isolation. However, they noted that there is no “one-size-fits-all” approach, and novel approaches custom tailored to particular populations, including frontline health care workers, are necessary.
“To make a real difference we will need to harness the tools of our digital age, finding smart new ways to measure the mental health of individuals remotely, finding creative ways to boost resilience, and finding ways to treat people in their homes. This effort must be considered central to our global response to the pandemic,” coauthor Ed Bullmore, PhD, of the department of psychiatry at the University of Cambridge (England), said in a statement.
Dr. Bullmore added that it will take “unprecedented research response if we are to limit the negative consequences of this pandemic on the mental health of our society now and in the future.”
Most vulnerable will bear the brunt
During a webinar held to discuss the paper, Matthew Hotopf, PhD, of the Institute of Psychiatry, Psychology, and Neuroscience at King’s College London, cautioned that society’s most vulnerable citizens will bear the brunt of the pandemic’s mental health consequences.
“These individuals often have unstable housing, unstable work, and are disadvantaged in terms of their physical health and their mental health,” with a “very significant gap” in life expectancy versus the rest of the population, he said. The COVID-19 pandemic will widen the gap between “the haves and the have nots.”
“People with established and significant mental disorders are one version of the ‘have nots’ but actually it applies to a lot of people,” said Dr. Hotopf, noting that his experience of lockdown is “very different” from that of someone “living in overcrowded, unstable accommodation, with kids running around and maybe a partner who has problems with anger control.”
The authors of the position paper noted that the COVID-19 pandemic highlights several important research priorities that need to be addressed in the coming weeks and months. These include:
- Understanding the effect of COVID-19 on risk of anxiety, depression, and other outcomes, such as self-harm and suicide
- Understanding how to create physical and social supports to ensure mental health in a climate of physical distancing
- Determining the mental health consequences of social isolation for vulnerable groups, and how can these be mitigated under pandemic conditions
- Understanding the mental health impact of media reporting of COVID-19 in traditional and social media
- Determining the best methods for promoting successful adherence to behavioral advice about COVID-19 while enabling mental well-being and minimizing distress
Another area highlighted by the experts is the potential for neuropsychiatric sequelae in individuals infected with COVID-19. They called for “experimental medicine studies to validate clinical biomarkers and repurpose new treatments for the potentially neurotoxic effects of the virus.”
The authors/investigators disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The COVID-19 pandemic is already affecting mental health at a population level, with increased anxiety, feelings of isolation, and concerns about access to mental health care.
Two U.K. surveys were conducted to inform research priorities for mental health research and in an effort to head off a mental health crisis. The U.K. charity MQ conducted a “stakeholder” survey of 2,198 individuals who had a lived experience of mental illness, while Ipsos MORI conducted a poll of 1,099 members of the public.
The online surveys were conducted in late March, the same week the U.K.’s nationwide lockdown measures were announced. Respondents were asked about their biggest mental health and well-being concerns and coping strategies as they relate to the COVID-19 pandemic.
Results showed that across the two surveys, respondents’ primary concern was anxiety, which was cited in 750 responses.
In addition, respondents were worried about being social isolated, becoming mentally unwell, and having a lack of access to mental health services, as well as the impact of the pandemic on personal relationships.
The findings were used by a panel of experts to inform a position paper published in the Lancet Psychiatry. The paper outlines a proposed government response to curb the long-term “profound” and “pervasive” impact of the pandemic on mental health.
‘Unprecedented response’ needed
“Governments must find evidence-based ways to boost the resilience of our societies and ... to treat those with mental ill health remotely to come out of this pandemic in good mental health,” coauthor of the paper Emily A. Holmes, PhD, of the department of psychology at Uppsala (Sweden) University, said in a press release.
“Frontline medical staff and vulnerable groups such as the elderly and those with serious mental health conditions must be prioritized for rapid mental health support,” she added.
The position paper authors call for “moment-to-moment” monitoring of anxiety, depression, self-harm, and suicide, as well as using digital technology and rapid deployment of evidence-based programs and treatments.
Patients will need to be accessible via computer, cell phone, and other remote technologies in order to receive treatment during physical isolation. However, they noted that there is no “one-size-fits-all” approach, and novel approaches custom tailored to particular populations, including frontline health care workers, are necessary.
“To make a real difference we will need to harness the tools of our digital age, finding smart new ways to measure the mental health of individuals remotely, finding creative ways to boost resilience, and finding ways to treat people in their homes. This effort must be considered central to our global response to the pandemic,” coauthor Ed Bullmore, PhD, of the department of psychiatry at the University of Cambridge (England), said in a statement.
Dr. Bullmore added that it will take “unprecedented research response if we are to limit the negative consequences of this pandemic on the mental health of our society now and in the future.”
Most vulnerable will bear the brunt
During a webinar held to discuss the paper, Matthew Hotopf, PhD, of the Institute of Psychiatry, Psychology, and Neuroscience at King’s College London, cautioned that society’s most vulnerable citizens will bear the brunt of the pandemic’s mental health consequences.
“These individuals often have unstable housing, unstable work, and are disadvantaged in terms of their physical health and their mental health,” with a “very significant gap” in life expectancy versus the rest of the population, he said. The COVID-19 pandemic will widen the gap between “the haves and the have nots.”
“People with established and significant mental disorders are one version of the ‘have nots’ but actually it applies to a lot of people,” said Dr. Hotopf, noting that his experience of lockdown is “very different” from that of someone “living in overcrowded, unstable accommodation, with kids running around and maybe a partner who has problems with anger control.”
The authors of the position paper noted that the COVID-19 pandemic highlights several important research priorities that need to be addressed in the coming weeks and months. These include:
- Understanding the effect of COVID-19 on risk of anxiety, depression, and other outcomes, such as self-harm and suicide
- Understanding how to create physical and social supports to ensure mental health in a climate of physical distancing
- Determining the mental health consequences of social isolation for vulnerable groups, and how can these be mitigated under pandemic conditions
- Understanding the mental health impact of media reporting of COVID-19 in traditional and social media
- Determining the best methods for promoting successful adherence to behavioral advice about COVID-19 while enabling mental well-being and minimizing distress
Another area highlighted by the experts is the potential for neuropsychiatric sequelae in individuals infected with COVID-19. They called for “experimental medicine studies to validate clinical biomarkers and repurpose new treatments for the potentially neurotoxic effects of the virus.”
The authors/investigators disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The COVID-19 pandemic is already affecting mental health at a population level, with increased anxiety, feelings of isolation, and concerns about access to mental health care.
Two U.K. surveys were conducted to inform research priorities for mental health research and in an effort to head off a mental health crisis. The U.K. charity MQ conducted a “stakeholder” survey of 2,198 individuals who had a lived experience of mental illness, while Ipsos MORI conducted a poll of 1,099 members of the public.
The online surveys were conducted in late March, the same week the U.K.’s nationwide lockdown measures were announced. Respondents were asked about their biggest mental health and well-being concerns and coping strategies as they relate to the COVID-19 pandemic.
Results showed that across the two surveys, respondents’ primary concern was anxiety, which was cited in 750 responses.
In addition, respondents were worried about being social isolated, becoming mentally unwell, and having a lack of access to mental health services, as well as the impact of the pandemic on personal relationships.
The findings were used by a panel of experts to inform a position paper published in the Lancet Psychiatry. The paper outlines a proposed government response to curb the long-term “profound” and “pervasive” impact of the pandemic on mental health.
‘Unprecedented response’ needed
“Governments must find evidence-based ways to boost the resilience of our societies and ... to treat those with mental ill health remotely to come out of this pandemic in good mental health,” coauthor of the paper Emily A. Holmes, PhD, of the department of psychology at Uppsala (Sweden) University, said in a press release.
“Frontline medical staff and vulnerable groups such as the elderly and those with serious mental health conditions must be prioritized for rapid mental health support,” she added.
The position paper authors call for “moment-to-moment” monitoring of anxiety, depression, self-harm, and suicide, as well as using digital technology and rapid deployment of evidence-based programs and treatments.
Patients will need to be accessible via computer, cell phone, and other remote technologies in order to receive treatment during physical isolation. However, they noted that there is no “one-size-fits-all” approach, and novel approaches custom tailored to particular populations, including frontline health care workers, are necessary.
“To make a real difference we will need to harness the tools of our digital age, finding smart new ways to measure the mental health of individuals remotely, finding creative ways to boost resilience, and finding ways to treat people in their homes. This effort must be considered central to our global response to the pandemic,” coauthor Ed Bullmore, PhD, of the department of psychiatry at the University of Cambridge (England), said in a statement.
Dr. Bullmore added that it will take “unprecedented research response if we are to limit the negative consequences of this pandemic on the mental health of our society now and in the future.”
Most vulnerable will bear the brunt
During a webinar held to discuss the paper, Matthew Hotopf, PhD, of the Institute of Psychiatry, Psychology, and Neuroscience at King’s College London, cautioned that society’s most vulnerable citizens will bear the brunt of the pandemic’s mental health consequences.
“These individuals often have unstable housing, unstable work, and are disadvantaged in terms of their physical health and their mental health,” with a “very significant gap” in life expectancy versus the rest of the population, he said. The COVID-19 pandemic will widen the gap between “the haves and the have nots.”
“People with established and significant mental disorders are one version of the ‘have nots’ but actually it applies to a lot of people,” said Dr. Hotopf, noting that his experience of lockdown is “very different” from that of someone “living in overcrowded, unstable accommodation, with kids running around and maybe a partner who has problems with anger control.”
The authors of the position paper noted that the COVID-19 pandemic highlights several important research priorities that need to be addressed in the coming weeks and months. These include:
- Understanding the effect of COVID-19 on risk of anxiety, depression, and other outcomes, such as self-harm and suicide
- Understanding how to create physical and social supports to ensure mental health in a climate of physical distancing
- Determining the mental health consequences of social isolation for vulnerable groups, and how can these be mitigated under pandemic conditions
- Understanding the mental health impact of media reporting of COVID-19 in traditional and social media
- Determining the best methods for promoting successful adherence to behavioral advice about COVID-19 while enabling mental well-being and minimizing distress
Another area highlighted by the experts is the potential for neuropsychiatric sequelae in individuals infected with COVID-19. They called for “experimental medicine studies to validate clinical biomarkers and repurpose new treatments for the potentially neurotoxic effects of the virus.”
The authors/investigators disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
During a pandemic, infusion center nursing team pitches in to keep patients on track
How do you run a chemotherapy infusion center during a pandemic?
Quick action, innovative staffing solutions, and nimble leadership are allowing one cancer center to continue providing care for the most vulnerable patients, while keeping patients and staff safe.
When nursing leaders at Atrium Health’s Levine Cancer Institute in Charlotte, N.C., realized that business was not going to continue as usual for American health care during the COVID-19 pandemic, they knew they had to act quickly to keep the institute’s 82-chair infusion center up and running.
North Carolina had already imposed restrictions on mass gatherings and closed educational facilities and some businesses by mid-March. Stay-at-home orders were being issued in surrounding states (North Carolina came under a statewide order on March 30). Physical distancing and a healthy, resilient team were prerequisites to an effective COVID-19 solution for the infusion clinic, said Angela Hosking, MBA, MSN, RN, director of nursing for Levine Cancer Institute. In an interview, she said that, at meetings on Monday, March 23, “we divided the team exactly in half.”
Infusion center staff members were broken into an “A” and a “B” rotation, with each team either on site or remotely for a 14-day stretch, and then switching at the 2-week mark. The 14-day rotation, she said, was chosen so that each cohort would have a full 2 weeks away after having been in clinic to ensure they were symptom free before returning. The cohorting scheme also serves to minimize between-staff exposure and risk of transmission.
These changes were implemented immediately, said Ms. Hosking, and included all but the most senior leadership – Ms. Hosking alternates days on site with another senior colleague to help with continuity.
Infusion center patients were triaged to determine “who absolutely needed to be seen,” and clinic staff started making phone calls and reshuffling the schedule so the clinic could continue at half-strength staffing.
The clinic was rearranged to ensure each infusion chair had appropriate space but the nursing work flow was still safe with reduced staff, said Jessica Stewart, MSN, RN, Levine Cancer Institute’s hematology–sickle cell nurse manager.
Patients were receptive, said Ms. Stewart. The team that was working remotely made sure all patients were called the day before their appointments, so they could understand what to expect when they arrived. Any needed updates to the medical history and patient teaching can also be done over the phone the day before the visit, she said, noting that patients are also queried about any concerning symptoms such as fever or cough.
In the spirit of providing information and managing expectations, patients are also informed that they will not be able to bring a visitor along and are advised to expect additional screening when they arrive. In addition to a repeat of symptom screening, patients are checked for fever with a temporal thermometer.
Any patient who arrives reporting symptoms or who has a fever is then subject to additional screening. Physician phone consultation is available, if needed, and patients may be routed to a drive-through screening and testing setup, or to the ED if there are concerns the patient may be seriously ill.
Several weeks into the new operations, Ms. Stewart said, “we’ve fine-tuned the processes we currently have in place. There’s new practices with virtual visits to make reaching our patients easier. Our senior leadership is communicating in a weekly video sent to all [Levine Cancer Institute] teammates for updates; it’s very transparent and the team is appreciative of being kept in the loop.”
Thus far, said Ms. Hosking, “it’s gone well – we’ve successfully operationalized this plan. … I think it shows that people that care about each other and their mission can collaborate with each other” to make change happen in a hurry.
Though it’s too soon to know exactly what the future holds once the pandemic has passed, some aspects of the new way of doing things may carry forward, said Ms. Stewart. “Communication has been massively streamlined,” and staff has found the previsit phone calls an efficient and effective way to gather and impart information.
A staff nurse at the infusion center, Whitney Hollifield, RN, added that patients have seen – and appreciate – the added precautions taken by all. “I feel that we have done well with protecting our patients from unneeded exposure and patients have expressed this to me,” said Ms. Hollifield. “They have said: ‘Thank you for doing this because I am scared to come in right now so I appreciate that your office is thinking of protecting us.’ ”
Ms. Hollifield added that “patients have been very responsive to our strategy for their care because we are truly concerned for them and I think that this shows. I believe that we are doing everything we can to keep them safe during a tumultuous time, and they feel genuine care for them during a frightening time is reassuring.”
On the practical side of things, Ms. Stewart noted, patients and families have provided infusion center staff with a seemingly endless supply of food: “We have never been more well fed!”
Rhonda Davis, RN, is a nurse at the Levine Cancer Institute. Speaking of the changes that have been made in recent weeks, she said, “Some of the changes that I think have been meaningful these last 3 weeks are making sure that the patients are the No. 1 priority. We are doing this by allowing patients options such as phone and virtual visits. This helps patients have some control over their health during this scary time for all.”
Ms. Davis acknowledged her own feelings about the uncertain times ahead. “As an individual with good health, I am scared, so to imagine the fear that these patients are facing must be overwhelming to them. Along that line, one of the most meaningful things that has happened for me is calling patients and having them concerned about my health and telling me to be safe.”
Despite her trepidation, she said, it’s meaningful for her to hear from patients who are in the clinic that they appreciate her presence. She found it heartening “that they are also considering our safety as well as their own.”
The two-cohort scheme has been well received by nursing staff, both administrators and clinic staff agreed. “I think that allowing staff to work 2 weeks on and 2 weeks at home helps keep patients and teammates safe,” Ms. Davis said.
Another infusion nurse, Ursel Wallace, RN, said that she appreciated the speed and efficiency with which the pandemic adaptations were made, including the nuts and bolts of reshuffling a complicated infusion schedule. “I know there were many different moving parts and it took a village” to move with such alacrity without dropping balls, she said.
The infusion nursing team’s spirit was summed up by Patricia Ashworth, RN: “Together, we will prevail!”
How do you run a chemotherapy infusion center during a pandemic?
Quick action, innovative staffing solutions, and nimble leadership are allowing one cancer center to continue providing care for the most vulnerable patients, while keeping patients and staff safe.
When nursing leaders at Atrium Health’s Levine Cancer Institute in Charlotte, N.C., realized that business was not going to continue as usual for American health care during the COVID-19 pandemic, they knew they had to act quickly to keep the institute’s 82-chair infusion center up and running.
North Carolina had already imposed restrictions on mass gatherings and closed educational facilities and some businesses by mid-March. Stay-at-home orders were being issued in surrounding states (North Carolina came under a statewide order on March 30). Physical distancing and a healthy, resilient team were prerequisites to an effective COVID-19 solution for the infusion clinic, said Angela Hosking, MBA, MSN, RN, director of nursing for Levine Cancer Institute. In an interview, she said that, at meetings on Monday, March 23, “we divided the team exactly in half.”
Infusion center staff members were broken into an “A” and a “B” rotation, with each team either on site or remotely for a 14-day stretch, and then switching at the 2-week mark. The 14-day rotation, she said, was chosen so that each cohort would have a full 2 weeks away after having been in clinic to ensure they were symptom free before returning. The cohorting scheme also serves to minimize between-staff exposure and risk of transmission.
These changes were implemented immediately, said Ms. Hosking, and included all but the most senior leadership – Ms. Hosking alternates days on site with another senior colleague to help with continuity.
Infusion center patients were triaged to determine “who absolutely needed to be seen,” and clinic staff started making phone calls and reshuffling the schedule so the clinic could continue at half-strength staffing.
The clinic was rearranged to ensure each infusion chair had appropriate space but the nursing work flow was still safe with reduced staff, said Jessica Stewart, MSN, RN, Levine Cancer Institute’s hematology–sickle cell nurse manager.
Patients were receptive, said Ms. Stewart. The team that was working remotely made sure all patients were called the day before their appointments, so they could understand what to expect when they arrived. Any needed updates to the medical history and patient teaching can also be done over the phone the day before the visit, she said, noting that patients are also queried about any concerning symptoms such as fever or cough.
In the spirit of providing information and managing expectations, patients are also informed that they will not be able to bring a visitor along and are advised to expect additional screening when they arrive. In addition to a repeat of symptom screening, patients are checked for fever with a temporal thermometer.
Any patient who arrives reporting symptoms or who has a fever is then subject to additional screening. Physician phone consultation is available, if needed, and patients may be routed to a drive-through screening and testing setup, or to the ED if there are concerns the patient may be seriously ill.
Several weeks into the new operations, Ms. Stewart said, “we’ve fine-tuned the processes we currently have in place. There’s new practices with virtual visits to make reaching our patients easier. Our senior leadership is communicating in a weekly video sent to all [Levine Cancer Institute] teammates for updates; it’s very transparent and the team is appreciative of being kept in the loop.”
Thus far, said Ms. Hosking, “it’s gone well – we’ve successfully operationalized this plan. … I think it shows that people that care about each other and their mission can collaborate with each other” to make change happen in a hurry.
Though it’s too soon to know exactly what the future holds once the pandemic has passed, some aspects of the new way of doing things may carry forward, said Ms. Stewart. “Communication has been massively streamlined,” and staff has found the previsit phone calls an efficient and effective way to gather and impart information.
A staff nurse at the infusion center, Whitney Hollifield, RN, added that patients have seen – and appreciate – the added precautions taken by all. “I feel that we have done well with protecting our patients from unneeded exposure and patients have expressed this to me,” said Ms. Hollifield. “They have said: ‘Thank you for doing this because I am scared to come in right now so I appreciate that your office is thinking of protecting us.’ ”
Ms. Hollifield added that “patients have been very responsive to our strategy for their care because we are truly concerned for them and I think that this shows. I believe that we are doing everything we can to keep them safe during a tumultuous time, and they feel genuine care for them during a frightening time is reassuring.”
On the practical side of things, Ms. Stewart noted, patients and families have provided infusion center staff with a seemingly endless supply of food: “We have never been more well fed!”
Rhonda Davis, RN, is a nurse at the Levine Cancer Institute. Speaking of the changes that have been made in recent weeks, she said, “Some of the changes that I think have been meaningful these last 3 weeks are making sure that the patients are the No. 1 priority. We are doing this by allowing patients options such as phone and virtual visits. This helps patients have some control over their health during this scary time for all.”
Ms. Davis acknowledged her own feelings about the uncertain times ahead. “As an individual with good health, I am scared, so to imagine the fear that these patients are facing must be overwhelming to them. Along that line, one of the most meaningful things that has happened for me is calling patients and having them concerned about my health and telling me to be safe.”
Despite her trepidation, she said, it’s meaningful for her to hear from patients who are in the clinic that they appreciate her presence. She found it heartening “that they are also considering our safety as well as their own.”
The two-cohort scheme has been well received by nursing staff, both administrators and clinic staff agreed. “I think that allowing staff to work 2 weeks on and 2 weeks at home helps keep patients and teammates safe,” Ms. Davis said.
Another infusion nurse, Ursel Wallace, RN, said that she appreciated the speed and efficiency with which the pandemic adaptations were made, including the nuts and bolts of reshuffling a complicated infusion schedule. “I know there were many different moving parts and it took a village” to move with such alacrity without dropping balls, she said.
The infusion nursing team’s spirit was summed up by Patricia Ashworth, RN: “Together, we will prevail!”
How do you run a chemotherapy infusion center during a pandemic?
Quick action, innovative staffing solutions, and nimble leadership are allowing one cancer center to continue providing care for the most vulnerable patients, while keeping patients and staff safe.
When nursing leaders at Atrium Health’s Levine Cancer Institute in Charlotte, N.C., realized that business was not going to continue as usual for American health care during the COVID-19 pandemic, they knew they had to act quickly to keep the institute’s 82-chair infusion center up and running.
North Carolina had already imposed restrictions on mass gatherings and closed educational facilities and some businesses by mid-March. Stay-at-home orders were being issued in surrounding states (North Carolina came under a statewide order on March 30). Physical distancing and a healthy, resilient team were prerequisites to an effective COVID-19 solution for the infusion clinic, said Angela Hosking, MBA, MSN, RN, director of nursing for Levine Cancer Institute. In an interview, she said that, at meetings on Monday, March 23, “we divided the team exactly in half.”
Infusion center staff members were broken into an “A” and a “B” rotation, with each team either on site or remotely for a 14-day stretch, and then switching at the 2-week mark. The 14-day rotation, she said, was chosen so that each cohort would have a full 2 weeks away after having been in clinic to ensure they were symptom free before returning. The cohorting scheme also serves to minimize between-staff exposure and risk of transmission.
These changes were implemented immediately, said Ms. Hosking, and included all but the most senior leadership – Ms. Hosking alternates days on site with another senior colleague to help with continuity.
Infusion center patients were triaged to determine “who absolutely needed to be seen,” and clinic staff started making phone calls and reshuffling the schedule so the clinic could continue at half-strength staffing.
The clinic was rearranged to ensure each infusion chair had appropriate space but the nursing work flow was still safe with reduced staff, said Jessica Stewart, MSN, RN, Levine Cancer Institute’s hematology–sickle cell nurse manager.
Patients were receptive, said Ms. Stewart. The team that was working remotely made sure all patients were called the day before their appointments, so they could understand what to expect when they arrived. Any needed updates to the medical history and patient teaching can also be done over the phone the day before the visit, she said, noting that patients are also queried about any concerning symptoms such as fever or cough.
In the spirit of providing information and managing expectations, patients are also informed that they will not be able to bring a visitor along and are advised to expect additional screening when they arrive. In addition to a repeat of symptom screening, patients are checked for fever with a temporal thermometer.
Any patient who arrives reporting symptoms or who has a fever is then subject to additional screening. Physician phone consultation is available, if needed, and patients may be routed to a drive-through screening and testing setup, or to the ED if there are concerns the patient may be seriously ill.
Several weeks into the new operations, Ms. Stewart said, “we’ve fine-tuned the processes we currently have in place. There’s new practices with virtual visits to make reaching our patients easier. Our senior leadership is communicating in a weekly video sent to all [Levine Cancer Institute] teammates for updates; it’s very transparent and the team is appreciative of being kept in the loop.”
Thus far, said Ms. Hosking, “it’s gone well – we’ve successfully operationalized this plan. … I think it shows that people that care about each other and their mission can collaborate with each other” to make change happen in a hurry.
Though it’s too soon to know exactly what the future holds once the pandemic has passed, some aspects of the new way of doing things may carry forward, said Ms. Stewart. “Communication has been massively streamlined,” and staff has found the previsit phone calls an efficient and effective way to gather and impart information.
A staff nurse at the infusion center, Whitney Hollifield, RN, added that patients have seen – and appreciate – the added precautions taken by all. “I feel that we have done well with protecting our patients from unneeded exposure and patients have expressed this to me,” said Ms. Hollifield. “They have said: ‘Thank you for doing this because I am scared to come in right now so I appreciate that your office is thinking of protecting us.’ ”
Ms. Hollifield added that “patients have been very responsive to our strategy for their care because we are truly concerned for them and I think that this shows. I believe that we are doing everything we can to keep them safe during a tumultuous time, and they feel genuine care for them during a frightening time is reassuring.”
On the practical side of things, Ms. Stewart noted, patients and families have provided infusion center staff with a seemingly endless supply of food: “We have never been more well fed!”
Rhonda Davis, RN, is a nurse at the Levine Cancer Institute. Speaking of the changes that have been made in recent weeks, she said, “Some of the changes that I think have been meaningful these last 3 weeks are making sure that the patients are the No. 1 priority. We are doing this by allowing patients options such as phone and virtual visits. This helps patients have some control over their health during this scary time for all.”
Ms. Davis acknowledged her own feelings about the uncertain times ahead. “As an individual with good health, I am scared, so to imagine the fear that these patients are facing must be overwhelming to them. Along that line, one of the most meaningful things that has happened for me is calling patients and having them concerned about my health and telling me to be safe.”
Despite her trepidation, she said, it’s meaningful for her to hear from patients who are in the clinic that they appreciate her presence. She found it heartening “that they are also considering our safety as well as their own.”
The two-cohort scheme has been well received by nursing staff, both administrators and clinic staff agreed. “I think that allowing staff to work 2 weeks on and 2 weeks at home helps keep patients and teammates safe,” Ms. Davis said.
Another infusion nurse, Ursel Wallace, RN, said that she appreciated the speed and efficiency with which the pandemic adaptations were made, including the nuts and bolts of reshuffling a complicated infusion schedule. “I know there were many different moving parts and it took a village” to move with such alacrity without dropping balls, she said.
The infusion nursing team’s spirit was summed up by Patricia Ashworth, RN: “Together, we will prevail!”
Latest data on COVID-19 patients with rheumatic diseases revealed in registry
An international registry of adult and pediatric rheumatology patients is beginning to identify trends in the types of patients with COVID-19 and who is recovering.
The COVID-19 Global Rheumatology Alliance (GRA) has created pediatric and adult registries for health care providers to enter information on their rheumatology patients with COVID-19. The adult registry is hosted by the University of California, San Francisco, Research Electronic Data Capture system, while the Childhood Arthritis and Rheumatology Research Alliance is supporting the pediatric registry. A separate path for data entry of both adult and pediatric cases has been established through the European League Against Rheumatism for European countries and countries with EULAR member organizations.
Prior to the creation of the registries, there were no data available to guide rheumatologists in clinical decision making for their patients, noted Jinoos Yazdany, MD, MPH, COVID-19 GRA steering committee member and chief of the division of rheumatology at Zuckerberg San Francisco General Hospital. “COVID-19 has potential to severely affect those with rheumatologic diseases or those taking immunosuppressive drugs,” she said in an interview. “The GRA registries were designed to answer critical questions that will inform the medical care of this population.”
The GRA began on Twitter, with conversations between Leonard H. Calabrese, DO, of the Cleveland Clinic; Paul Sufka, MD, of HealthPartners in St. Paul, Minn.; Philip Robinson, MBChB, PhD, of the Royal Brisbane (Australia) Hospital; and herself, Dr. Yazdany said. Dr. Robinson started work on the governance of the GRA, Dr. Yazdany designed the data infrastructure, and Dr. Sufka approached his professional networks and social media followings to promote the effort and ask for support. The COVID-19 GRA steering committee representatives include patients, private practice rheumatologists, and international investigators. Listed among official supporters of the alliance are the American College of Rheumatology and EULAR along with more than 290 medical societies, institutions, journals, and other organizations in rheumatology.
The goal of the registries is to examine the health outcomes of patients with rheumatic diseases and COVID-19 based on sociodemographic factors, comorbidities, and clinical presentations of COVID-19 as well as what role taking immunosuppressive drugs prior to a COVID-19 infection play in helping or hindering outcomes. Hydroxychloroquine, used to treat lupus and arthritis, is a potential treatment candidate for COVID-19. Biologics such as tocilizumab (Actemra) and sarilumab (Kevzara), which target interleukin-6, and anakinra (Kineret), which targets IL-1, are treatment candidates for patients who have experienced COVID-related cytokine storm syndrome, which researchers believe may contribute to worsening or fatal cases.
Dr. Yazdany, who is also vice chair of real-world data infrastructure, registry, and institutional review board/ethics for the GRA, said that there are some important high-level trends in the data thus far. “People with lupus and those taking hydroxychloroquine are becoming infected with SARS-CoV-2, which is counter to misinformation on social media. Most people with rheumatic diseases on immunosuppression are recovering, which is great news for our patients.”
One of the major strengths of the registries is that each case is entered by the rheumatologist treating the patient and contains detailed clinical information, Dr. Yazdany said. However, the registry has no control group, it is not a population surveillance study, and it may contain selection bias through rheumatologists omitting milder, undiagnosed cases.
“The Global Alliance case reporting registry represents the collective effort of hundreds of rheumatologists across the world. I have never been more inspired by the strength and collaboration of the rheumatology community,” Dr. Yazdany said.
According to a paper published in the Lancet Rheumatology, which references data on 110 cases from the combined databases up to April 1, about three-fourths of cases presented with fever (79%) and cough (77%), and about half presented with shortness of breath (50%) and myalgia (45%).
Results from the global and UCSF registries
As of April 18, 334 cases were in the global and UCSF registries, with 121 patients (36%) in the database having both COVID-19 and RA, 33 patients (10%) with psoriatic arthritis, 58 patients (17%) with systemic lupus erythematosus, 28 patients (8%) with axial spondyloarthritis, 27 patients (8%) with vasculitis, and 19 patients (6%) with Sjögren’s syndrome. There were less than five cases reported for patients with the following rheumatic diseases: inflammatory myopathy, ocular inflammation, other inflammatory arthritis, polymyalgia rheumatica, sarcoidosis, systemic sclerosis, osteoporosis, psoriasis, isolated pulmonary capillaritis, gout, and autoinflammatory disease. A majority of the patients in the registries are women (74%) aged younger than 65 years (78%) and are white (52%).
The most common comorbid conditions among patients in the registry are hypertension (33%), lung disease (18%), diabetes (11%), cardiovascular disease (10%), chronic renal insufficiency or end-stage renal disease (7%), morbid obesity (7%), and cancer (4%). Before being diagnosed with COVID-19, 219 patients (66%) in the registry were taking conventional synthetic disease-modifying antirheumatic drugs (csDMARDs), which included antimalarials, azathioprine, cyclophosphamide, cyclosporine, leflunomide, methotrexate, mycophenolate mofetil/mycophenolic acid, sulfasalazine, and tacrolimus. A total of 122 patients (37%) were taking biologic DMARDs, 101 patients were taking glucocorticoids (30%), 86 patients (26%) were taking hydroxychloroquine, 41 patients (12%) were taking NSAIDs, and 18 patients (5%) were taking a Janus kinase inhibitor.
The most recent data from the registry show that 128 patients (38%) have been hospitalized for COVID-19, and 19 patients (6%) have died. Although 104 patients (31%) resolved their infections, 177 patients (53%) have a COVID-19 infection status of “unresolved,” and 53 patients (16%) have an unknown infection status.
EULAR registry results
As of April 21, 249 cases were in the EULAR registry, including 110 hospitalizations (44%) and 37 deaths (15%). Overall, 64% of these patients were women, and they had a median age of 60 years.
The top five diagnoses of these patients were RA (39%), psoriatic arthritis (15%), spondyloarthritis (9%), systemic lupus erythematosus (9%), and gout (5%). A total of 27% had no reported comorbidities, while lung disease occurred in 26%, hypertension in 34%, diabetes in 11%, and cardiovascular disease on 11%. The registry also reported use of any DMARD in 80%, including 62% on csDMARDs, 31% on biologics, and 2% on targeted synthetic DMARDs.
Ten authors in the Lancet Rheumatology paper reported personal and institutional relationships in the form of grants, corporate sponsorships, advisory board memberships, investigator appointments, speaker’s bureau positions, personal fees, and consultancies for a variety of pharmaceutical companies, agencies, societies, and other organizations. The other authors reported no relevant conflicts of interest.
An international registry of adult and pediatric rheumatology patients is beginning to identify trends in the types of patients with COVID-19 and who is recovering.
The COVID-19 Global Rheumatology Alliance (GRA) has created pediatric and adult registries for health care providers to enter information on their rheumatology patients with COVID-19. The adult registry is hosted by the University of California, San Francisco, Research Electronic Data Capture system, while the Childhood Arthritis and Rheumatology Research Alliance is supporting the pediatric registry. A separate path for data entry of both adult and pediatric cases has been established through the European League Against Rheumatism for European countries and countries with EULAR member organizations.
Prior to the creation of the registries, there were no data available to guide rheumatologists in clinical decision making for their patients, noted Jinoos Yazdany, MD, MPH, COVID-19 GRA steering committee member and chief of the division of rheumatology at Zuckerberg San Francisco General Hospital. “COVID-19 has potential to severely affect those with rheumatologic diseases or those taking immunosuppressive drugs,” she said in an interview. “The GRA registries were designed to answer critical questions that will inform the medical care of this population.”
The GRA began on Twitter, with conversations between Leonard H. Calabrese, DO, of the Cleveland Clinic; Paul Sufka, MD, of HealthPartners in St. Paul, Minn.; Philip Robinson, MBChB, PhD, of the Royal Brisbane (Australia) Hospital; and herself, Dr. Yazdany said. Dr. Robinson started work on the governance of the GRA, Dr. Yazdany designed the data infrastructure, and Dr. Sufka approached his professional networks and social media followings to promote the effort and ask for support. The COVID-19 GRA steering committee representatives include patients, private practice rheumatologists, and international investigators. Listed among official supporters of the alliance are the American College of Rheumatology and EULAR along with more than 290 medical societies, institutions, journals, and other organizations in rheumatology.
The goal of the registries is to examine the health outcomes of patients with rheumatic diseases and COVID-19 based on sociodemographic factors, comorbidities, and clinical presentations of COVID-19 as well as what role taking immunosuppressive drugs prior to a COVID-19 infection play in helping or hindering outcomes. Hydroxychloroquine, used to treat lupus and arthritis, is a potential treatment candidate for COVID-19. Biologics such as tocilizumab (Actemra) and sarilumab (Kevzara), which target interleukin-6, and anakinra (Kineret), which targets IL-1, are treatment candidates for patients who have experienced COVID-related cytokine storm syndrome, which researchers believe may contribute to worsening or fatal cases.
Dr. Yazdany, who is also vice chair of real-world data infrastructure, registry, and institutional review board/ethics for the GRA, said that there are some important high-level trends in the data thus far. “People with lupus and those taking hydroxychloroquine are becoming infected with SARS-CoV-2, which is counter to misinformation on social media. Most people with rheumatic diseases on immunosuppression are recovering, which is great news for our patients.”
One of the major strengths of the registries is that each case is entered by the rheumatologist treating the patient and contains detailed clinical information, Dr. Yazdany said. However, the registry has no control group, it is not a population surveillance study, and it may contain selection bias through rheumatologists omitting milder, undiagnosed cases.
“The Global Alliance case reporting registry represents the collective effort of hundreds of rheumatologists across the world. I have never been more inspired by the strength and collaboration of the rheumatology community,” Dr. Yazdany said.
According to a paper published in the Lancet Rheumatology, which references data on 110 cases from the combined databases up to April 1, about three-fourths of cases presented with fever (79%) and cough (77%), and about half presented with shortness of breath (50%) and myalgia (45%).
Results from the global and UCSF registries
As of April 18, 334 cases were in the global and UCSF registries, with 121 patients (36%) in the database having both COVID-19 and RA, 33 patients (10%) with psoriatic arthritis, 58 patients (17%) with systemic lupus erythematosus, 28 patients (8%) with axial spondyloarthritis, 27 patients (8%) with vasculitis, and 19 patients (6%) with Sjögren’s syndrome. There were less than five cases reported for patients with the following rheumatic diseases: inflammatory myopathy, ocular inflammation, other inflammatory arthritis, polymyalgia rheumatica, sarcoidosis, systemic sclerosis, osteoporosis, psoriasis, isolated pulmonary capillaritis, gout, and autoinflammatory disease. A majority of the patients in the registries are women (74%) aged younger than 65 years (78%) and are white (52%).
The most common comorbid conditions among patients in the registry are hypertension (33%), lung disease (18%), diabetes (11%), cardiovascular disease (10%), chronic renal insufficiency or end-stage renal disease (7%), morbid obesity (7%), and cancer (4%). Before being diagnosed with COVID-19, 219 patients (66%) in the registry were taking conventional synthetic disease-modifying antirheumatic drugs (csDMARDs), which included antimalarials, azathioprine, cyclophosphamide, cyclosporine, leflunomide, methotrexate, mycophenolate mofetil/mycophenolic acid, sulfasalazine, and tacrolimus. A total of 122 patients (37%) were taking biologic DMARDs, 101 patients were taking glucocorticoids (30%), 86 patients (26%) were taking hydroxychloroquine, 41 patients (12%) were taking NSAIDs, and 18 patients (5%) were taking a Janus kinase inhibitor.
The most recent data from the registry show that 128 patients (38%) have been hospitalized for COVID-19, and 19 patients (6%) have died. Although 104 patients (31%) resolved their infections, 177 patients (53%) have a COVID-19 infection status of “unresolved,” and 53 patients (16%) have an unknown infection status.
EULAR registry results
As of April 21, 249 cases were in the EULAR registry, including 110 hospitalizations (44%) and 37 deaths (15%). Overall, 64% of these patients were women, and they had a median age of 60 years.
The top five diagnoses of these patients were RA (39%), psoriatic arthritis (15%), spondyloarthritis (9%), systemic lupus erythematosus (9%), and gout (5%). A total of 27% had no reported comorbidities, while lung disease occurred in 26%, hypertension in 34%, diabetes in 11%, and cardiovascular disease on 11%. The registry also reported use of any DMARD in 80%, including 62% on csDMARDs, 31% on biologics, and 2% on targeted synthetic DMARDs.
Ten authors in the Lancet Rheumatology paper reported personal and institutional relationships in the form of grants, corporate sponsorships, advisory board memberships, investigator appointments, speaker’s bureau positions, personal fees, and consultancies for a variety of pharmaceutical companies, agencies, societies, and other organizations. The other authors reported no relevant conflicts of interest.
An international registry of adult and pediatric rheumatology patients is beginning to identify trends in the types of patients with COVID-19 and who is recovering.
The COVID-19 Global Rheumatology Alliance (GRA) has created pediatric and adult registries for health care providers to enter information on their rheumatology patients with COVID-19. The adult registry is hosted by the University of California, San Francisco, Research Electronic Data Capture system, while the Childhood Arthritis and Rheumatology Research Alliance is supporting the pediatric registry. A separate path for data entry of both adult and pediatric cases has been established through the European League Against Rheumatism for European countries and countries with EULAR member organizations.
Prior to the creation of the registries, there were no data available to guide rheumatologists in clinical decision making for their patients, noted Jinoos Yazdany, MD, MPH, COVID-19 GRA steering committee member and chief of the division of rheumatology at Zuckerberg San Francisco General Hospital. “COVID-19 has potential to severely affect those with rheumatologic diseases or those taking immunosuppressive drugs,” she said in an interview. “The GRA registries were designed to answer critical questions that will inform the medical care of this population.”
The GRA began on Twitter, with conversations between Leonard H. Calabrese, DO, of the Cleveland Clinic; Paul Sufka, MD, of HealthPartners in St. Paul, Minn.; Philip Robinson, MBChB, PhD, of the Royal Brisbane (Australia) Hospital; and herself, Dr. Yazdany said. Dr. Robinson started work on the governance of the GRA, Dr. Yazdany designed the data infrastructure, and Dr. Sufka approached his professional networks and social media followings to promote the effort and ask for support. The COVID-19 GRA steering committee representatives include patients, private practice rheumatologists, and international investigators. Listed among official supporters of the alliance are the American College of Rheumatology and EULAR along with more than 290 medical societies, institutions, journals, and other organizations in rheumatology.
The goal of the registries is to examine the health outcomes of patients with rheumatic diseases and COVID-19 based on sociodemographic factors, comorbidities, and clinical presentations of COVID-19 as well as what role taking immunosuppressive drugs prior to a COVID-19 infection play in helping or hindering outcomes. Hydroxychloroquine, used to treat lupus and arthritis, is a potential treatment candidate for COVID-19. Biologics such as tocilizumab (Actemra) and sarilumab (Kevzara), which target interleukin-6, and anakinra (Kineret), which targets IL-1, are treatment candidates for patients who have experienced COVID-related cytokine storm syndrome, which researchers believe may contribute to worsening or fatal cases.
Dr. Yazdany, who is also vice chair of real-world data infrastructure, registry, and institutional review board/ethics for the GRA, said that there are some important high-level trends in the data thus far. “People with lupus and those taking hydroxychloroquine are becoming infected with SARS-CoV-2, which is counter to misinformation on social media. Most people with rheumatic diseases on immunosuppression are recovering, which is great news for our patients.”
One of the major strengths of the registries is that each case is entered by the rheumatologist treating the patient and contains detailed clinical information, Dr. Yazdany said. However, the registry has no control group, it is not a population surveillance study, and it may contain selection bias through rheumatologists omitting milder, undiagnosed cases.
“The Global Alliance case reporting registry represents the collective effort of hundreds of rheumatologists across the world. I have never been more inspired by the strength and collaboration of the rheumatology community,” Dr. Yazdany said.
According to a paper published in the Lancet Rheumatology, which references data on 110 cases from the combined databases up to April 1, about three-fourths of cases presented with fever (79%) and cough (77%), and about half presented with shortness of breath (50%) and myalgia (45%).
Results from the global and UCSF registries
As of April 18, 334 cases were in the global and UCSF registries, with 121 patients (36%) in the database having both COVID-19 and RA, 33 patients (10%) with psoriatic arthritis, 58 patients (17%) with systemic lupus erythematosus, 28 patients (8%) with axial spondyloarthritis, 27 patients (8%) with vasculitis, and 19 patients (6%) with Sjögren’s syndrome. There were less than five cases reported for patients with the following rheumatic diseases: inflammatory myopathy, ocular inflammation, other inflammatory arthritis, polymyalgia rheumatica, sarcoidosis, systemic sclerosis, osteoporosis, psoriasis, isolated pulmonary capillaritis, gout, and autoinflammatory disease. A majority of the patients in the registries are women (74%) aged younger than 65 years (78%) and are white (52%).
The most common comorbid conditions among patients in the registry are hypertension (33%), lung disease (18%), diabetes (11%), cardiovascular disease (10%), chronic renal insufficiency or end-stage renal disease (7%), morbid obesity (7%), and cancer (4%). Before being diagnosed with COVID-19, 219 patients (66%) in the registry were taking conventional synthetic disease-modifying antirheumatic drugs (csDMARDs), which included antimalarials, azathioprine, cyclophosphamide, cyclosporine, leflunomide, methotrexate, mycophenolate mofetil/mycophenolic acid, sulfasalazine, and tacrolimus. A total of 122 patients (37%) were taking biologic DMARDs, 101 patients were taking glucocorticoids (30%), 86 patients (26%) were taking hydroxychloroquine, 41 patients (12%) were taking NSAIDs, and 18 patients (5%) were taking a Janus kinase inhibitor.
The most recent data from the registry show that 128 patients (38%) have been hospitalized for COVID-19, and 19 patients (6%) have died. Although 104 patients (31%) resolved their infections, 177 patients (53%) have a COVID-19 infection status of “unresolved,” and 53 patients (16%) have an unknown infection status.
EULAR registry results
As of April 21, 249 cases were in the EULAR registry, including 110 hospitalizations (44%) and 37 deaths (15%). Overall, 64% of these patients were women, and they had a median age of 60 years.
The top five diagnoses of these patients were RA (39%), psoriatic arthritis (15%), spondyloarthritis (9%), systemic lupus erythematosus (9%), and gout (5%). A total of 27% had no reported comorbidities, while lung disease occurred in 26%, hypertension in 34%, diabetes in 11%, and cardiovascular disease on 11%. The registry also reported use of any DMARD in 80%, including 62% on csDMARDs, 31% on biologics, and 2% on targeted synthetic DMARDs.
Ten authors in the Lancet Rheumatology paper reported personal and institutional relationships in the form of grants, corporate sponsorships, advisory board memberships, investigator appointments, speaker’s bureau positions, personal fees, and consultancies for a variety of pharmaceutical companies, agencies, societies, and other organizations. The other authors reported no relevant conflicts of interest.
COVID-19: Helping health care workers on front lines
Psychiatrists are intervening with less conventional strategies
Across the country, psychiatrists are stepping up to provide urgent care to fellow health care workers in need amid the coronavirus pandemic. They’re offering stress management strategies, spearheading unusual partnerships, and discovering that psychotherapy and medication might not be their most helpful tools to help their colleagues at this time.
“This is completely the opposite of the way we practice psychiatry,” said Allison Cotton, MD, of the University of Nevada, Reno. “Our interventions are quite different from a psychotherapeutic standpoint.”
In March, she worked with four colleagues, Suzan Song, MD, MPH, PhD; Ben Cheng, MD; Smita Gautam, MD; and Mona S. Masood, DO, to create the Physician Support Line, a confidential and free hotline that links physicians to volunteer psychiatrists who are available to listen and offer advice on coping. The hotline (888-409-0141) is available every day from 8 a.m. to midnight Eastern time. Calls typically take 15-45 minutes; no appointment is needed, and conversations are not reportable to state medical boards.

“The calls can be very intense,” Dr. Cotton said, and they’re unusual for several reasons. The hotline is not like a suicide or crisis hotline, when “a person calls because they need help, and then they can go get that help – they go to the hospital and get admitted to a psychiatric unit. Our callers don’t have that luxury.”
It’s also impossible to take an extensive history and create a sophisticated, long-term treatment plan as psychiatrists would during normal office visits. At the hotline, Dr. Cotton said, “we’re really focusing on the caller’s strengths and helping them come up with a plan for today to get through whatever they’re facing,” she said.
Stress management is critical
Psychiatrists at the University of Colorado Anschutz Medical Campus are embracing a similar approach to help health care workers cope, said Steven Berkowitz, MD. “We focus on stress management, and the notion that they are generally healthy and understandably struggling with extraordinary circumstances,” he said. “We are conservative in our use of medications and really only prescribe medications, such as trazodone, to help with sleep. We do not use benzodiazepines unless there is a history of more severe psychiatric problems.”
The pressure on health care workers during the pandemic is intense. A survey of 1,257 workers in 34 Chinese hospitals found high levels of symptoms of depression (50%), anxiety (45%), insomnia (35%), and distress (72%). Several groups appeared to be more vulnerable: women, nurses, front-line health care workers, and those in the coronavirus-stricken city of Wuhan (JAMA Netw Open. 2020;3[3]:e203976).
In Colorado, “providers are depleted,” Dr. Berkowitz said. “We are hearing about sleep disturbances and even some traumatic nightmares from ICU staff. During our support sessions, tears come most frequently when they talk about the struggle to care for their families and how they’re putting them at risk.”
Also, he said, “one of the most upsetting issues has been around language and cultural issues. Because of the language barriers, providers cannot explain why families can’t be with their sick members, which has led to acrimony.”
Guilt is a prevailing theme
Guilt also is a common emotion among health care workers, said psychiatrist Tia Konzer, DO, of Charlotte, N.C. “The ones on the front line question whether they were able to do enough to save someone or if they could have done more. Those of us not on the front lines feel guilty that we’re not there with our colleagues, that we don’t face the same fears and are in the safety of our outpatient clinics.”
The focus on social distancing is creating its own strains, she said. “A lot of people are recognizing the power of human touch and how comforting that is,” she said. “The healers aren’t able to comfort the loved ones of the deceased, and we’re not able to comfort each other. And people are having a hard time not being able to hug their kids and their spouses, having to ward off their kids when they come home or avoid them until they’ve showered.”
How can mental health professionals be most helpful to health care workers in need? The simple act of listening is crucial, several such professionals said in interviews.
“Your main job is to bear witness to their experiences and to hear their story, then secondarily to make sure they have a basic self-care plan to recover from what they’re doing each day,” said psychologist Leah Welch, PhD, of the Scripps Health network in San Diego. “Don’t talk too much or try to give advice too quickly before you’ve listened to what the caregiver has shared. They’re accumulating small traumas and need time and space to sort them out, and that takes patience and a listening ear on the part of the provider. Rushing in too quickly with advice deprives them of making sense of their own experience.”
She added that “they should also be thanked for what they’re doing, because it requires skill, empathy, and courage. They are being heroic, and they need to know they’re appreciated by those of us not on the front lines for what they’re putting themselves through.”
Partnerships are forming
At Zuckerberg San Francisco General Hospital and Trauma Center, psychiatry chief Lisa Fortuna, MD, MPH, MDiv, said her team has had success by working closely with the hospital’s chaplains. “A lot of the staff are not saying: ‘We’re stressing out; help us.’ The chaplains had starting rounding, asking how they’re doing, and they’d open up because there was already a relationship. The chaplains are very well trained in dealing with being support for people under situations of death, loss, and immediate stress.”
The chaplains themselves became overwhelmed, and the hospital responded by reaching out to bring in more chaplains. The psychiatry team, meanwhile, worked to partner with the chaplains to provide a continuum of support for staff. “We have an opportunity to build on the trust that they have,” said Dr. Fortuna, who is an ordained Episcopal minister. “They’re the perfect partners.”
What happens now? Dr. Fortuna has seen the long-term aftermath of a crisis. She previously worked in Massachusetts and helped to support health care workers in that state after the Boston Marathon bombing.
She cautioned that health care workers may first run on adrenaline in a crisis, spurred by “heroic high energy.” But then, the full extent of the tragedy begins to set in, and they start to process their feelings. “You have to keep people going through those phases,” she said.
Going forward, she said, “there will be a prolonged tail of stress,” especially if virus outbreaks recur. “We’ll have a long time enduring this.”
Don’t forget the self-care
There was a time during the pandemic when Dr. Cotton had become so overwhelmed by anxiety that she called the Physician Support Line to get some support from fellow psychiatrists.
“I thought, ‘Why not?’” she recalled. “I helped create the hotline. Why wouldn’t I call it?”
The calls took only a few minutes but they made a difference to Dr. Cotton, who had been severely ill with what she believed was an unconfirmed case of the novel coronavirus. “I immediately felt more like I improved my outlook by focusing on what I could control,” she said, “and accepting the things I could not control.”
Many psychiatrists are finding themselves in similar situations. Fortunately, colleagues are highlighting ways for psychiatrists to care for themselves just as they care for patients.
“One of the challenges clinicians are facing is that they are living through a shared experience in this global pandemic with their patients right now,” said psychologist Randi Pochtar, PhD, who is managing support groups for front-line workers at NYU Langone Health in New York City. “Some might find the work to be overwhelming and anxiety-inducing, and others might find their work to be helpful in managing their own anxiety and stress about the pandemic and its impact.”
Dr. Cotton said her breaking points came when she felt panic amid the pandemic. “I had watched too much news, and I’d seen protesters not taking it seriously, and I was scared for my family and myself. I just needed to feel like someone heard me feeling that way.”
The calls to the hotline were helpful, she said, and so was sharing news about her illness with friends. “So many people reached out to me and checked in on me, people I haven’t seen in years, and that was immensely helpful,” she said.
This sort of personal exposure may not come naturally to physicians and nurses, she said. “We don’t seek that kind of attention when we’re ill. Instead, we say: ‘I’m fine; how are you doing?’ That’s what we do every day of our lives at work.”
How can clinicians help themselves and one another? “Clinicians in our practice have been coping and supporting each other through peer supervision, connecting with colleagues in team meetings, and simply checking in on one another,” said Dr. Pochtar. “In addition, we can adopt many of the strategies that we are likely recommending to our patients, such as maintaining routines as much as possible, engaging in regular exercise, eating well and consistently, and connecting with friends and family.”
Managers can play important roles, said Dr. Fortuna. “I’ve been checking in with my faculty, being as supportive as I can be and highlighting the extraordinary things that people are doing, like going from zero to 100 percent in setting up telehealth.”
Dr. Konzer offered another perspective on recognizing the value of the work that psychiatrists are doing. “We’re on the front line of helping heal the front line, and in that responsibility comes an additional stress,” she said. “But there’s an additional gift of being able to contribute where we are most beneficial. We can try to be present now, versus worrying about what may happen or what lies ahead, and appreciate the beauty in the helpers and the small joys of life.”
Dr. Cotton, Dr. Berkowitz, Dr. Konzer, Dr. Welch, Dr. Fortuna, and Dr. Pochtar reported no relevant disclosures.
Psychiatrists are intervening with less conventional strategies
Psychiatrists are intervening with less conventional strategies
Across the country, psychiatrists are stepping up to provide urgent care to fellow health care workers in need amid the coronavirus pandemic. They’re offering stress management strategies, spearheading unusual partnerships, and discovering that psychotherapy and medication might not be their most helpful tools to help their colleagues at this time.
“This is completely the opposite of the way we practice psychiatry,” said Allison Cotton, MD, of the University of Nevada, Reno. “Our interventions are quite different from a psychotherapeutic standpoint.”
In March, she worked with four colleagues, Suzan Song, MD, MPH, PhD; Ben Cheng, MD; Smita Gautam, MD; and Mona S. Masood, DO, to create the Physician Support Line, a confidential and free hotline that links physicians to volunteer psychiatrists who are available to listen and offer advice on coping. The hotline (888-409-0141) is available every day from 8 a.m. to midnight Eastern time. Calls typically take 15-45 minutes; no appointment is needed, and conversations are not reportable to state medical boards.

“The calls can be very intense,” Dr. Cotton said, and they’re unusual for several reasons. The hotline is not like a suicide or crisis hotline, when “a person calls because they need help, and then they can go get that help – they go to the hospital and get admitted to a psychiatric unit. Our callers don’t have that luxury.”
It’s also impossible to take an extensive history and create a sophisticated, long-term treatment plan as psychiatrists would during normal office visits. At the hotline, Dr. Cotton said, “we’re really focusing on the caller’s strengths and helping them come up with a plan for today to get through whatever they’re facing,” she said.
Stress management is critical
Psychiatrists at the University of Colorado Anschutz Medical Campus are embracing a similar approach to help health care workers cope, said Steven Berkowitz, MD. “We focus on stress management, and the notion that they are generally healthy and understandably struggling with extraordinary circumstances,” he said. “We are conservative in our use of medications and really only prescribe medications, such as trazodone, to help with sleep. We do not use benzodiazepines unless there is a history of more severe psychiatric problems.”
The pressure on health care workers during the pandemic is intense. A survey of 1,257 workers in 34 Chinese hospitals found high levels of symptoms of depression (50%), anxiety (45%), insomnia (35%), and distress (72%). Several groups appeared to be more vulnerable: women, nurses, front-line health care workers, and those in the coronavirus-stricken city of Wuhan (JAMA Netw Open. 2020;3[3]:e203976).
In Colorado, “providers are depleted,” Dr. Berkowitz said. “We are hearing about sleep disturbances and even some traumatic nightmares from ICU staff. During our support sessions, tears come most frequently when they talk about the struggle to care for their families and how they’re putting them at risk.”
Also, he said, “one of the most upsetting issues has been around language and cultural issues. Because of the language barriers, providers cannot explain why families can’t be with their sick members, which has led to acrimony.”
Guilt is a prevailing theme
Guilt also is a common emotion among health care workers, said psychiatrist Tia Konzer, DO, of Charlotte, N.C. “The ones on the front line question whether they were able to do enough to save someone or if they could have done more. Those of us not on the front lines feel guilty that we’re not there with our colleagues, that we don’t face the same fears and are in the safety of our outpatient clinics.”
The focus on social distancing is creating its own strains, she said. “A lot of people are recognizing the power of human touch and how comforting that is,” she said. “The healers aren’t able to comfort the loved ones of the deceased, and we’re not able to comfort each other. And people are having a hard time not being able to hug their kids and their spouses, having to ward off their kids when they come home or avoid them until they’ve showered.”
How can mental health professionals be most helpful to health care workers in need? The simple act of listening is crucial, several such professionals said in interviews.
“Your main job is to bear witness to their experiences and to hear their story, then secondarily to make sure they have a basic self-care plan to recover from what they’re doing each day,” said psychologist Leah Welch, PhD, of the Scripps Health network in San Diego. “Don’t talk too much or try to give advice too quickly before you’ve listened to what the caregiver has shared. They’re accumulating small traumas and need time and space to sort them out, and that takes patience and a listening ear on the part of the provider. Rushing in too quickly with advice deprives them of making sense of their own experience.”
She added that “they should also be thanked for what they’re doing, because it requires skill, empathy, and courage. They are being heroic, and they need to know they’re appreciated by those of us not on the front lines for what they’re putting themselves through.”
Partnerships are forming
At Zuckerberg San Francisco General Hospital and Trauma Center, psychiatry chief Lisa Fortuna, MD, MPH, MDiv, said her team has had success by working closely with the hospital’s chaplains. “A lot of the staff are not saying: ‘We’re stressing out; help us.’ The chaplains had starting rounding, asking how they’re doing, and they’d open up because there was already a relationship. The chaplains are very well trained in dealing with being support for people under situations of death, loss, and immediate stress.”
The chaplains themselves became overwhelmed, and the hospital responded by reaching out to bring in more chaplains. The psychiatry team, meanwhile, worked to partner with the chaplains to provide a continuum of support for staff. “We have an opportunity to build on the trust that they have,” said Dr. Fortuna, who is an ordained Episcopal minister. “They’re the perfect partners.”
What happens now? Dr. Fortuna has seen the long-term aftermath of a crisis. She previously worked in Massachusetts and helped to support health care workers in that state after the Boston Marathon bombing.
She cautioned that health care workers may first run on adrenaline in a crisis, spurred by “heroic high energy.” But then, the full extent of the tragedy begins to set in, and they start to process their feelings. “You have to keep people going through those phases,” she said.
Going forward, she said, “there will be a prolonged tail of stress,” especially if virus outbreaks recur. “We’ll have a long time enduring this.”
Don’t forget the self-care
There was a time during the pandemic when Dr. Cotton had become so overwhelmed by anxiety that she called the Physician Support Line to get some support from fellow psychiatrists.
“I thought, ‘Why not?’” she recalled. “I helped create the hotline. Why wouldn’t I call it?”
The calls took only a few minutes but they made a difference to Dr. Cotton, who had been severely ill with what she believed was an unconfirmed case of the novel coronavirus. “I immediately felt more like I improved my outlook by focusing on what I could control,” she said, “and accepting the things I could not control.”
Many psychiatrists are finding themselves in similar situations. Fortunately, colleagues are highlighting ways for psychiatrists to care for themselves just as they care for patients.
“One of the challenges clinicians are facing is that they are living through a shared experience in this global pandemic with their patients right now,” said psychologist Randi Pochtar, PhD, who is managing support groups for front-line workers at NYU Langone Health in New York City. “Some might find the work to be overwhelming and anxiety-inducing, and others might find their work to be helpful in managing their own anxiety and stress about the pandemic and its impact.”
Dr. Cotton said her breaking points came when she felt panic amid the pandemic. “I had watched too much news, and I’d seen protesters not taking it seriously, and I was scared for my family and myself. I just needed to feel like someone heard me feeling that way.”
The calls to the hotline were helpful, she said, and so was sharing news about her illness with friends. “So many people reached out to me and checked in on me, people I haven’t seen in years, and that was immensely helpful,” she said.
This sort of personal exposure may not come naturally to physicians and nurses, she said. “We don’t seek that kind of attention when we’re ill. Instead, we say: ‘I’m fine; how are you doing?’ That’s what we do every day of our lives at work.”
How can clinicians help themselves and one another? “Clinicians in our practice have been coping and supporting each other through peer supervision, connecting with colleagues in team meetings, and simply checking in on one another,” said Dr. Pochtar. “In addition, we can adopt many of the strategies that we are likely recommending to our patients, such as maintaining routines as much as possible, engaging in regular exercise, eating well and consistently, and connecting with friends and family.”
Managers can play important roles, said Dr. Fortuna. “I’ve been checking in with my faculty, being as supportive as I can be and highlighting the extraordinary things that people are doing, like going from zero to 100 percent in setting up telehealth.”
Dr. Konzer offered another perspective on recognizing the value of the work that psychiatrists are doing. “We’re on the front line of helping heal the front line, and in that responsibility comes an additional stress,” she said. “But there’s an additional gift of being able to contribute where we are most beneficial. We can try to be present now, versus worrying about what may happen or what lies ahead, and appreciate the beauty in the helpers and the small joys of life.”
Dr. Cotton, Dr. Berkowitz, Dr. Konzer, Dr. Welch, Dr. Fortuna, and Dr. Pochtar reported no relevant disclosures.
Across the country, psychiatrists are stepping up to provide urgent care to fellow health care workers in need amid the coronavirus pandemic. They’re offering stress management strategies, spearheading unusual partnerships, and discovering that psychotherapy and medication might not be their most helpful tools to help their colleagues at this time.
“This is completely the opposite of the way we practice psychiatry,” said Allison Cotton, MD, of the University of Nevada, Reno. “Our interventions are quite different from a psychotherapeutic standpoint.”
In March, she worked with four colleagues, Suzan Song, MD, MPH, PhD; Ben Cheng, MD; Smita Gautam, MD; and Mona S. Masood, DO, to create the Physician Support Line, a confidential and free hotline that links physicians to volunteer psychiatrists who are available to listen and offer advice on coping. The hotline (888-409-0141) is available every day from 8 a.m. to midnight Eastern time. Calls typically take 15-45 minutes; no appointment is needed, and conversations are not reportable to state medical boards.

“The calls can be very intense,” Dr. Cotton said, and they’re unusual for several reasons. The hotline is not like a suicide or crisis hotline, when “a person calls because they need help, and then they can go get that help – they go to the hospital and get admitted to a psychiatric unit. Our callers don’t have that luxury.”
It’s also impossible to take an extensive history and create a sophisticated, long-term treatment plan as psychiatrists would during normal office visits. At the hotline, Dr. Cotton said, “we’re really focusing on the caller’s strengths and helping them come up with a plan for today to get through whatever they’re facing,” she said.
Stress management is critical
Psychiatrists at the University of Colorado Anschutz Medical Campus are embracing a similar approach to help health care workers cope, said Steven Berkowitz, MD. “We focus on stress management, and the notion that they are generally healthy and understandably struggling with extraordinary circumstances,” he said. “We are conservative in our use of medications and really only prescribe medications, such as trazodone, to help with sleep. We do not use benzodiazepines unless there is a history of more severe psychiatric problems.”
The pressure on health care workers during the pandemic is intense. A survey of 1,257 workers in 34 Chinese hospitals found high levels of symptoms of depression (50%), anxiety (45%), insomnia (35%), and distress (72%). Several groups appeared to be more vulnerable: women, nurses, front-line health care workers, and those in the coronavirus-stricken city of Wuhan (JAMA Netw Open. 2020;3[3]:e203976).
In Colorado, “providers are depleted,” Dr. Berkowitz said. “We are hearing about sleep disturbances and even some traumatic nightmares from ICU staff. During our support sessions, tears come most frequently when they talk about the struggle to care for their families and how they’re putting them at risk.”
Also, he said, “one of the most upsetting issues has been around language and cultural issues. Because of the language barriers, providers cannot explain why families can’t be with their sick members, which has led to acrimony.”
Guilt is a prevailing theme
Guilt also is a common emotion among health care workers, said psychiatrist Tia Konzer, DO, of Charlotte, N.C. “The ones on the front line question whether they were able to do enough to save someone or if they could have done more. Those of us not on the front lines feel guilty that we’re not there with our colleagues, that we don’t face the same fears and are in the safety of our outpatient clinics.”
The focus on social distancing is creating its own strains, she said. “A lot of people are recognizing the power of human touch and how comforting that is,” she said. “The healers aren’t able to comfort the loved ones of the deceased, and we’re not able to comfort each other. And people are having a hard time not being able to hug their kids and their spouses, having to ward off their kids when they come home or avoid them until they’ve showered.”
How can mental health professionals be most helpful to health care workers in need? The simple act of listening is crucial, several such professionals said in interviews.
“Your main job is to bear witness to their experiences and to hear their story, then secondarily to make sure they have a basic self-care plan to recover from what they’re doing each day,” said psychologist Leah Welch, PhD, of the Scripps Health network in San Diego. “Don’t talk too much or try to give advice too quickly before you’ve listened to what the caregiver has shared. They’re accumulating small traumas and need time and space to sort them out, and that takes patience and a listening ear on the part of the provider. Rushing in too quickly with advice deprives them of making sense of their own experience.”
She added that “they should also be thanked for what they’re doing, because it requires skill, empathy, and courage. They are being heroic, and they need to know they’re appreciated by those of us not on the front lines for what they’re putting themselves through.”
Partnerships are forming
At Zuckerberg San Francisco General Hospital and Trauma Center, psychiatry chief Lisa Fortuna, MD, MPH, MDiv, said her team has had success by working closely with the hospital’s chaplains. “A lot of the staff are not saying: ‘We’re stressing out; help us.’ The chaplains had starting rounding, asking how they’re doing, and they’d open up because there was already a relationship. The chaplains are very well trained in dealing with being support for people under situations of death, loss, and immediate stress.”
The chaplains themselves became overwhelmed, and the hospital responded by reaching out to bring in more chaplains. The psychiatry team, meanwhile, worked to partner with the chaplains to provide a continuum of support for staff. “We have an opportunity to build on the trust that they have,” said Dr. Fortuna, who is an ordained Episcopal minister. “They’re the perfect partners.”
What happens now? Dr. Fortuna has seen the long-term aftermath of a crisis. She previously worked in Massachusetts and helped to support health care workers in that state after the Boston Marathon bombing.
She cautioned that health care workers may first run on adrenaline in a crisis, spurred by “heroic high energy.” But then, the full extent of the tragedy begins to set in, and they start to process their feelings. “You have to keep people going through those phases,” she said.
Going forward, she said, “there will be a prolonged tail of stress,” especially if virus outbreaks recur. “We’ll have a long time enduring this.”
Don’t forget the self-care
There was a time during the pandemic when Dr. Cotton had become so overwhelmed by anxiety that she called the Physician Support Line to get some support from fellow psychiatrists.
“I thought, ‘Why not?’” she recalled. “I helped create the hotline. Why wouldn’t I call it?”
The calls took only a few minutes but they made a difference to Dr. Cotton, who had been severely ill with what she believed was an unconfirmed case of the novel coronavirus. “I immediately felt more like I improved my outlook by focusing on what I could control,” she said, “and accepting the things I could not control.”
Many psychiatrists are finding themselves in similar situations. Fortunately, colleagues are highlighting ways for psychiatrists to care for themselves just as they care for patients.
“One of the challenges clinicians are facing is that they are living through a shared experience in this global pandemic with their patients right now,” said psychologist Randi Pochtar, PhD, who is managing support groups for front-line workers at NYU Langone Health in New York City. “Some might find the work to be overwhelming and anxiety-inducing, and others might find their work to be helpful in managing their own anxiety and stress about the pandemic and its impact.”
Dr. Cotton said her breaking points came when she felt panic amid the pandemic. “I had watched too much news, and I’d seen protesters not taking it seriously, and I was scared for my family and myself. I just needed to feel like someone heard me feeling that way.”
The calls to the hotline were helpful, she said, and so was sharing news about her illness with friends. “So many people reached out to me and checked in on me, people I haven’t seen in years, and that was immensely helpful,” she said.
This sort of personal exposure may not come naturally to physicians and nurses, she said. “We don’t seek that kind of attention when we’re ill. Instead, we say: ‘I’m fine; how are you doing?’ That’s what we do every day of our lives at work.”
How can clinicians help themselves and one another? “Clinicians in our practice have been coping and supporting each other through peer supervision, connecting with colleagues in team meetings, and simply checking in on one another,” said Dr. Pochtar. “In addition, we can adopt many of the strategies that we are likely recommending to our patients, such as maintaining routines as much as possible, engaging in regular exercise, eating well and consistently, and connecting with friends and family.”
Managers can play important roles, said Dr. Fortuna. “I’ve been checking in with my faculty, being as supportive as I can be and highlighting the extraordinary things that people are doing, like going from zero to 100 percent in setting up telehealth.”
Dr. Konzer offered another perspective on recognizing the value of the work that psychiatrists are doing. “We’re on the front line of helping heal the front line, and in that responsibility comes an additional stress,” she said. “But there’s an additional gift of being able to contribute where we are most beneficial. We can try to be present now, versus worrying about what may happen or what lies ahead, and appreciate the beauty in the helpers and the small joys of life.”
Dr. Cotton, Dr. Berkowitz, Dr. Konzer, Dr. Welch, Dr. Fortuna, and Dr. Pochtar reported no relevant disclosures.










