User login
Clinical Endocrinology News is an independent news source that provides endocrinologists with timely and relevant news and commentary about clinical developments and the impact of health care policy on the endocrinologist's practice. Specialty topics include Diabetes, Lipid & Metabolic Disorders Menopause, Obesity, Osteoporosis, Pediatric Endocrinology, Pituitary, Thyroid & Adrenal Disorders, and Reproductive Endocrinology. Featured content includes Commentaries, Implementin Health Reform, Law & Medicine, and In the Loop, the blog of Clinical Endocrinology News. Clinical Endocrinology News is owned by Frontline Medical Communications.
addict
addicted
addicting
addiction
adult sites
alcohol
antibody
ass
attorney
audit
auditor
babies
babpa
baby
ban
banned
banning
best
bisexual
bitch
bleach
blog
blow job
bondage
boobs
booty
buy
cannabis
certificate
certification
certified
cheap
cheapest
class action
cocaine
cock
counterfeit drug
crack
crap
crime
criminal
cunt
curable
cure
dangerous
dangers
dead
deadly
death
defend
defended
depedent
dependence
dependent
detergent
dick
die
dildo
drug abuse
drug recall
dying
fag
fake
fatal
fatalities
fatality
free
fuck
gangs
gingivitis
guns
hardcore
herbal
herbs
heroin
herpes
home remedies
homo
horny
hypersensitivity
hypoglycemia treatment
illegal drug use
illegal use of prescription
incest
infant
infants
job
ketoacidosis
kill
killer
killing
kinky
law suit
lawsuit
lawyer
lesbian
marijuana
medicine for hypoglycemia
murder
naked
natural
newborn
nigger
noise
nude
nudity
orgy
over the counter
overdosage
overdose
overdosed
overdosing
penis
pimp
pistol
porn
porno
pornographic
pornography
prison
profanity
purchase
purchasing
pussy
queer
rape
rapist
recall
recreational drug
rob
robberies
sale
sales
sex
sexual
shit
shoot
slut
slutty
stole
stolen
store
sue
suicidal
suicide
supplements
supply company
theft
thief
thieves
tit
toddler
toddlers
toxic
toxin
tragedy
treating dka
treating hypoglycemia
treatment for hypoglycemia
vagina
violence
whore
withdrawal
without prescription
section[contains(@class, 'nav-hidden')]
footer[@id='footer']
div[contains(@class, 'pane-pub-article-imn')]
div[contains(@class, 'pane-pub-home-imn')]
div[contains(@class, 'pane-pub-topic-imn')]
div[contains(@class, 'panel-panel-inner')]
div[contains(@class, 'pane-node-field-article-topics')]
section[contains(@class, 'footer-nav-section-wrapper')]
Ancient plague, cyclical pandemics … history lesson?
Even the plague wanted to visit Stonehenge
We’re about to blow your mind: The history you learned in school was often inaccurate. Shocking, we know, so we’ll give you a minute to process this incredible news.
Better? Good. Now, let’s look back at high school European history. The Black Death, specifically. The common narrative is that the Mongols, while besieging a Crimean city belonging to the Genoese, catapulted dead bodies infected with some mystery disease that turned out to be the plague. The Genoese then brought the plague back to Italy, and from there, we all know the rest of the story.
The Black Death was certainly extremely important to the development of modern Europe as we know it, but the history books gloss over the much longer history of the plague. Yersinia pestis did not suddenly appear unbidden in a Mongol war camp in 1347. The Black Death wasn’t even the first horrific, continent-wide pandemic caused by the plague; the Plague of Justinian 800 years earlier crippled the Byzantine Empire during an expansionist phase and killed anywhere between 15 million and 100 million.
Today, though, LOTME looks even deeper into history, nearly beyond even history itself, back into the depths of early Bronze Age northern Europe. Specifically, to two ancient burial sites in England, where researchers have identified three 4,000-year-old cases of Y. pestis, the first recorded incidence of the disease in Britain.
Two of the individuals, identified through analysis of dental pulp, were young children buried at a mass grave in Somerset, while the third, a middle-aged woman, was found in a ring cairn in Cumbria. These sites are hundreds of miles apart, yet carbon dating suggests all three people lived and died at roughly the same time. The strain found is very similar to other samples of plague found across central and western Europe starting around 3,000 BCE, suggesting a single, easily spread disease affecting a large area in a relatively small period of time. In other words, a pandemic. Even in these ancient times, the world was connected. Not even the island of Britain could escape.
Beyond that though, the research helps confirm the cyclical nature of the plague; over time, it loses its effectiveness and goes into hiding, only to mutate and come roaring back. This is a story with absolutely no relevance at all to the modern world. Nope, no plagues or pandemics going around right now, no viruses fading into the background in any way. What a ridiculous inference to make.
Uncovering the invisible with artificial intelligence
This week in “What Else Can AI Do?” new research shows that a computer program can reveal brain injury that couldn’t be seen before with typical MRI.
The hot new AI, birthed by researchers at New York University, could potentially be a game changer by linking repeated head impacts with tiny, structural changes in the brains of athletes who have not been diagnosed with a concussion. By using machine learning to train the AI, the researchers were, for the first time, able to distinguish the brain of athletes who played contact sports (football, soccer, lacrosse) from those participating in noncontact sports such as baseball, basketball, and cross-country.
How did they do it? The investigators “designed statistical techniques that gave their computer program the ability to ‘learn’ how to predict exposure to repeated head impacts using mathematical models,” they explained in a written statement. Adding in data from the MRI scans of 81 male athletes with no known concussion diagnosis and the ability to identify unusual brain features between athletes with and without head trauma allowed the AI to predict results with accuracy even Miss Cleo would envy.
“This method may provide an important diagnostic tool not only for concussion, but also for detecting the damage that stems from subtler and more frequent head impacts,” said lead author Junbo Chen, an engineering doctoral candidate at NYU. That could make this new AI a valuable asset to science and medicine.
There are many things the human brain can do that AI can’t, and delegation could be one of them. Examining the data that represent the human brain in minute detail? Maybe we leave that to the machine.
Talk about your field promotions
If you’re a surgeon doing an amputation, the list of possible assistants pretty much starts and ends in only one place: Not the closest available janitor.
That may seem like an oddly obvious thing for us to say, but there’s at least one former Mainz (Germany) University Hospital physician who really needed to get this bit of advice before he attempted an unassisted toe amputation back in October of 2020. Yes, that does seem like kind of a long time ago for us to be reporting it now, but the details of the incident only just came to light a few days ago, thanks to German public broadcaster SWR.
Since it was just a toe, the surgeon thought he could perform the operation without any help. The toe, unfortunately, had other plans. The partially anesthetized patient got restless in the operating room, but with no actual trained nurse in the vicinity, the surgeon asked the closest available person – that would be the janitor – to lend a hand.
The surgical manager heard about these goings-on and got to the operating room too late to stop the procedure but soon enough to see the cleaning staffer “at the operating table with a bloody suction cup and a bloody compress in their hands,” SWR recently reported.
The incident was reported to the hospital’s medical director and the surgeon was fired, but since the patient experienced no complications not much fuss was made about it at the time.
Well, guess what? It’s toe-tally our job to make a fuss about these kinds of things. Or could it be that our job, much like the surgeon’s employment and the patient’s digit, is here toe-day and gone toe-morrow?
Even the plague wanted to visit Stonehenge
We’re about to blow your mind: The history you learned in school was often inaccurate. Shocking, we know, so we’ll give you a minute to process this incredible news.
Better? Good. Now, let’s look back at high school European history. The Black Death, specifically. The common narrative is that the Mongols, while besieging a Crimean city belonging to the Genoese, catapulted dead bodies infected with some mystery disease that turned out to be the plague. The Genoese then brought the plague back to Italy, and from there, we all know the rest of the story.
The Black Death was certainly extremely important to the development of modern Europe as we know it, but the history books gloss over the much longer history of the plague. Yersinia pestis did not suddenly appear unbidden in a Mongol war camp in 1347. The Black Death wasn’t even the first horrific, continent-wide pandemic caused by the plague; the Plague of Justinian 800 years earlier crippled the Byzantine Empire during an expansionist phase and killed anywhere between 15 million and 100 million.
Today, though, LOTME looks even deeper into history, nearly beyond even history itself, back into the depths of early Bronze Age northern Europe. Specifically, to two ancient burial sites in England, where researchers have identified three 4,000-year-old cases of Y. pestis, the first recorded incidence of the disease in Britain.
Two of the individuals, identified through analysis of dental pulp, were young children buried at a mass grave in Somerset, while the third, a middle-aged woman, was found in a ring cairn in Cumbria. These sites are hundreds of miles apart, yet carbon dating suggests all three people lived and died at roughly the same time. The strain found is very similar to other samples of plague found across central and western Europe starting around 3,000 BCE, suggesting a single, easily spread disease affecting a large area in a relatively small period of time. In other words, a pandemic. Even in these ancient times, the world was connected. Not even the island of Britain could escape.
Beyond that though, the research helps confirm the cyclical nature of the plague; over time, it loses its effectiveness and goes into hiding, only to mutate and come roaring back. This is a story with absolutely no relevance at all to the modern world. Nope, no plagues or pandemics going around right now, no viruses fading into the background in any way. What a ridiculous inference to make.
Uncovering the invisible with artificial intelligence
This week in “What Else Can AI Do?” new research shows that a computer program can reveal brain injury that couldn’t be seen before with typical MRI.
The hot new AI, birthed by researchers at New York University, could potentially be a game changer by linking repeated head impacts with tiny, structural changes in the brains of athletes who have not been diagnosed with a concussion. By using machine learning to train the AI, the researchers were, for the first time, able to distinguish the brain of athletes who played contact sports (football, soccer, lacrosse) from those participating in noncontact sports such as baseball, basketball, and cross-country.
How did they do it? The investigators “designed statistical techniques that gave their computer program the ability to ‘learn’ how to predict exposure to repeated head impacts using mathematical models,” they explained in a written statement. Adding in data from the MRI scans of 81 male athletes with no known concussion diagnosis and the ability to identify unusual brain features between athletes with and without head trauma allowed the AI to predict results with accuracy even Miss Cleo would envy.
“This method may provide an important diagnostic tool not only for concussion, but also for detecting the damage that stems from subtler and more frequent head impacts,” said lead author Junbo Chen, an engineering doctoral candidate at NYU. That could make this new AI a valuable asset to science and medicine.
There are many things the human brain can do that AI can’t, and delegation could be one of them. Examining the data that represent the human brain in minute detail? Maybe we leave that to the machine.
Talk about your field promotions
If you’re a surgeon doing an amputation, the list of possible assistants pretty much starts and ends in only one place: Not the closest available janitor.
That may seem like an oddly obvious thing for us to say, but there’s at least one former Mainz (Germany) University Hospital physician who really needed to get this bit of advice before he attempted an unassisted toe amputation back in October of 2020. Yes, that does seem like kind of a long time ago for us to be reporting it now, but the details of the incident only just came to light a few days ago, thanks to German public broadcaster SWR.
Since it was just a toe, the surgeon thought he could perform the operation without any help. The toe, unfortunately, had other plans. The partially anesthetized patient got restless in the operating room, but with no actual trained nurse in the vicinity, the surgeon asked the closest available person – that would be the janitor – to lend a hand.
The surgical manager heard about these goings-on and got to the operating room too late to stop the procedure but soon enough to see the cleaning staffer “at the operating table with a bloody suction cup and a bloody compress in their hands,” SWR recently reported.
The incident was reported to the hospital’s medical director and the surgeon was fired, but since the patient experienced no complications not much fuss was made about it at the time.
Well, guess what? It’s toe-tally our job to make a fuss about these kinds of things. Or could it be that our job, much like the surgeon’s employment and the patient’s digit, is here toe-day and gone toe-morrow?
Even the plague wanted to visit Stonehenge
We’re about to blow your mind: The history you learned in school was often inaccurate. Shocking, we know, so we’ll give you a minute to process this incredible news.
Better? Good. Now, let’s look back at high school European history. The Black Death, specifically. The common narrative is that the Mongols, while besieging a Crimean city belonging to the Genoese, catapulted dead bodies infected with some mystery disease that turned out to be the plague. The Genoese then brought the plague back to Italy, and from there, we all know the rest of the story.
The Black Death was certainly extremely important to the development of modern Europe as we know it, but the history books gloss over the much longer history of the plague. Yersinia pestis did not suddenly appear unbidden in a Mongol war camp in 1347. The Black Death wasn’t even the first horrific, continent-wide pandemic caused by the plague; the Plague of Justinian 800 years earlier crippled the Byzantine Empire during an expansionist phase and killed anywhere between 15 million and 100 million.
Today, though, LOTME looks even deeper into history, nearly beyond even history itself, back into the depths of early Bronze Age northern Europe. Specifically, to two ancient burial sites in England, where researchers have identified three 4,000-year-old cases of Y. pestis, the first recorded incidence of the disease in Britain.
Two of the individuals, identified through analysis of dental pulp, were young children buried at a mass grave in Somerset, while the third, a middle-aged woman, was found in a ring cairn in Cumbria. These sites are hundreds of miles apart, yet carbon dating suggests all three people lived and died at roughly the same time. The strain found is very similar to other samples of plague found across central and western Europe starting around 3,000 BCE, suggesting a single, easily spread disease affecting a large area in a relatively small period of time. In other words, a pandemic. Even in these ancient times, the world was connected. Not even the island of Britain could escape.
Beyond that though, the research helps confirm the cyclical nature of the plague; over time, it loses its effectiveness and goes into hiding, only to mutate and come roaring back. This is a story with absolutely no relevance at all to the modern world. Nope, no plagues or pandemics going around right now, no viruses fading into the background in any way. What a ridiculous inference to make.
Uncovering the invisible with artificial intelligence
This week in “What Else Can AI Do?” new research shows that a computer program can reveal brain injury that couldn’t be seen before with typical MRI.
The hot new AI, birthed by researchers at New York University, could potentially be a game changer by linking repeated head impacts with tiny, structural changes in the brains of athletes who have not been diagnosed with a concussion. By using machine learning to train the AI, the researchers were, for the first time, able to distinguish the brain of athletes who played contact sports (football, soccer, lacrosse) from those participating in noncontact sports such as baseball, basketball, and cross-country.
How did they do it? The investigators “designed statistical techniques that gave their computer program the ability to ‘learn’ how to predict exposure to repeated head impacts using mathematical models,” they explained in a written statement. Adding in data from the MRI scans of 81 male athletes with no known concussion diagnosis and the ability to identify unusual brain features between athletes with and without head trauma allowed the AI to predict results with accuracy even Miss Cleo would envy.
“This method may provide an important diagnostic tool not only for concussion, but also for detecting the damage that stems from subtler and more frequent head impacts,” said lead author Junbo Chen, an engineering doctoral candidate at NYU. That could make this new AI a valuable asset to science and medicine.
There are many things the human brain can do that AI can’t, and delegation could be one of them. Examining the data that represent the human brain in minute detail? Maybe we leave that to the machine.
Talk about your field promotions
If you’re a surgeon doing an amputation, the list of possible assistants pretty much starts and ends in only one place: Not the closest available janitor.
That may seem like an oddly obvious thing for us to say, but there’s at least one former Mainz (Germany) University Hospital physician who really needed to get this bit of advice before he attempted an unassisted toe amputation back in October of 2020. Yes, that does seem like kind of a long time ago for us to be reporting it now, but the details of the incident only just came to light a few days ago, thanks to German public broadcaster SWR.
Since it was just a toe, the surgeon thought he could perform the operation without any help. The toe, unfortunately, had other plans. The partially anesthetized patient got restless in the operating room, but with no actual trained nurse in the vicinity, the surgeon asked the closest available person – that would be the janitor – to lend a hand.
The surgical manager heard about these goings-on and got to the operating room too late to stop the procedure but soon enough to see the cleaning staffer “at the operating table with a bloody suction cup and a bloody compress in their hands,” SWR recently reported.
The incident was reported to the hospital’s medical director and the surgeon was fired, but since the patient experienced no complications not much fuss was made about it at the time.
Well, guess what? It’s toe-tally our job to make a fuss about these kinds of things. Or could it be that our job, much like the surgeon’s employment and the patient’s digit, is here toe-day and gone toe-morrow?
Troponin to ID diabetes patients with silent heart disease?
– based on data from a representative sample of more than 10,000 U.S. adults.
The finding suggests hs-cTnT maybe a useful marker for adults with diabetes who could benefit from more aggressive CVD risk reduction despite having no clinical indications of CVD.
The results “highlight the substantial burden of subclinical CVD in persons with diabetes and emphasize the importance of early detection and treatment of CVD for this high-risk population,” say the authors of the research, published in the Journal of the American Heart Association.
“This is the first study to examine subclinical CVD, defined by elevated cardiac biomarkers, in a nationally representative population of adults with or without diabetes. It provides novel information on the high burden of subclinical CVD [in American adults with diabetes] and the potential utility of hs-cTnT for monitoring this risk in people with diabetes,” said Elizabeth Selvin, PhD, senior author and a professor of epidemiology at Johns Hopkins University, Baltimore.
“What we are seeing is that many people with type 2 diabetes who have not had a heart attack or a history of cardiovascular disease are at high risk for cardiovascular complications,” added Dr. Selvin in an AHA press release. “When we look at the whole population of people diagnosed with type 2 diabetes, about 27 million adults in the U.S., according to the [Centers for Disease Control and Prevention], some are at low risk and some are at high risk for cardiovascular disease, so the open question is: ‘Who is most at risk?’ These cardiac biomarkers give us a window into cardiovascular risk in people who otherwise might not be recognized as highest risk.”
“Our results provide evidence to support use of cardiac biomarkers for routine risk monitoring in high-risk populations such as people with diabetes,” Dr. Selvin noted in an interview.
Need for aggressive CVD risk reduction
The findings also indicate that people with diabetes and an elevated hs-cTnT “should be targeted for aggressive cardiovascular risk reduction, including lifestyle interventions, weight loss, and treatment with statins, blood pressure medications, and cardioprotective therapies such as sodium-glucose cotransporter 2 (SGLT-2) inhibitors and glucagonlike peptide-1 (GLP-1) receptor agonists,” Dr. Selvin added.
“Cholesterol is often the factor that we target to reduce the risk of cardiovascular disease in people with type 2 diabetes,” she observed. “However, type 2 diabetes may have a direct effect on the heart not related to cholesterol levels. If type 2 diabetes is directly causing damage to the small vessels in the heart unrelated to cholesterol plaque buildup, then cholesterol-lowering medications are not going to prevent cardiac damage,” Dr. Selvin explained. “Our research suggests that additional non–statin-related therapies are needed to lower the cardiovascular disease risk in people with type 2 diabetes.”
However, she noted that a necessary step prior to formally recommending such a strategy is to run clinical trials to assess the efficacy of specific treatments, such as SGLT-2 inhibitors and GLP-1 agonists, in people with diabetes and elevated hs-cTnT.
“Randomized controlled trials would be best to test the relevance of measuring these biomarkers to assess risk in asymptomatic people with diabetes,” as well as prospective study of the value of hs-cTnT to guide treatment, commented Robert H. Eckel, MD, an endocrinologist affiliated with the University of Colorado at Denver, Aurora.
“I doubt measurements [of hs-cTnT] would be reimbursed [by third-party payers] if carried out without such outcome data,” he added.
Dr. Eckel also highlights the need to further validate in additional cohorts the link between elevations in hs-cTnT and CVD events in adults with diabetes, and to confirm that elevated levels of another cardiac biomarker – N-terminal of the prohormone brain natriuretic peptide (NT-proBNP) – do not work as well as troponin as a risk marker for people with diabetes, another finding of the study.
ADA report already recommends testing these biomarkers for HF
However, a consensus report published in 2022 by the American Diabetes Association laid out the case for routinely and regularly measuring levels of both high sensitivity cardiac troponin and natriuretic peptides in people with diabetes for early identification of incident heart failure.
“Among individuals with diabetes, measurement of a natriuretic peptide or high-sensitivity cardiac troponin is recommended on at least a yearly basis to identify the earliest heart failure stages and implement strategies to prevent transition to symptomatic heart failure,” noted the ADA consensus report on heart failure.
The new study run by Dr. Selvin and coauthors used data collected by the National Health and Nutrition Examination Survey (NHANES) between 1999 and 2004 from U.S. adults who were at least 20 years old and had no history of CVD: myocardial infarction, stroke, coronary heart disease, or heart failure. This included 9,273 people without diabetes and 1,031 with diabetes, defined as a prior diagnosis or hemoglobin A1c of at least 6.5%.
“Cardiovascular risk varies substantially in adults with type 2 diabetes, highlighting the need for accurate risk stratification,” the authors observed.
All study participants had recorded measures of hs-cTnT and NT-proBNP.
The researchers considered an hs-cTnT level of greater than 14 ng/L and an NT-proBNP level of greater than 125 pg/mL as indicators of subclinical CVD.
The crude prevalence of elevated NT-proBNP was 33.4% among those with diabetes and 16.1% in those without diabetes. Elevated hs-cTnT occurred in 19% of those with diabetes and in 5% of those without diabetes. Elevated levels of both markers existed in 9% of those with diabetes and in 3% of those without diabetes.
“Approximately one in three adults with diabetes had subclinical CVD, with 19% having elevated levels of hs-cTnT, 23% having elevated NT-proBNP, and 9% having elevations in both cardiac biomarkers,” the researchers noted.
Diabetes linked with a doubled prevalence of elevated hs-cTnT
After adjustment for several demographic variables as well as traditional CVD risk factors, people with diabetes had a significant 98% higher rate of elevated hs-cTnT, compared with those without diabetes. But after similar adjustments, the rate of elevated NT-proBNP was significantly lower among people with diabetes, compared with controls, by a relative reduction of 24%.
“Our findings suggest that, in people with diabetes, hs-cTnT may be more useful [than NT-proBNP] for general risk monitoring, as its interpretation is less complicated,” said Dr. Selvin, who explained that “NT-proBNP is affected by overweight and obesity.”
In people with diabetes, the age-adjusted prevalence of elevated hs-cTnT ran higher in those with longer duration diabetes, and in those with less well-controlled diabetes based on a higher level of A1c. Neither of these factors showed any significant relationship with measured levels of NT-proBNP.
Further analysis linked the NHANES findings during 1999-2004 with U.S. national death records through the end of 2019. This showed that elevated levels of both hs-cTnT and NT-proBNP significantly linked with subsequently higher rates of all-cause mortality among people with diabetes. Elevated hs-cTnT linked with a 77% increased mortality and NT-proBNP linked with a 78% increased rate, compared with people with diabetes and no elevations in these markers, after adjustment for demographic variables and CVD risk factors.
However, for the outcome of cardiovascular death, elevated hs-cTnT linked with a nonsignificant 54% relative increase, while elevated NT-proBNP linked with a significant 2.46-fold relative increase.
The study “adds new data on biomarkers that are not routinely measured in asymptomatic people with or without diabetes” and the relationships of these markers to CVD mortality and all-cause mortality, Dr. Eckel concluded.
The study received no commercial funding, but used reagents donated by Abbott Laboratories, Ortho Clinical Diagnostics, Roche Diagnostics, and Siemens Healthcare Diagnostics. Dr. Selvin and Dr. Eckel had no disclosures.
A version of this article first appeared on Medscape.com.
– based on data from a representative sample of more than 10,000 U.S. adults.
The finding suggests hs-cTnT maybe a useful marker for adults with diabetes who could benefit from more aggressive CVD risk reduction despite having no clinical indications of CVD.
The results “highlight the substantial burden of subclinical CVD in persons with diabetes and emphasize the importance of early detection and treatment of CVD for this high-risk population,” say the authors of the research, published in the Journal of the American Heart Association.
“This is the first study to examine subclinical CVD, defined by elevated cardiac biomarkers, in a nationally representative population of adults with or without diabetes. It provides novel information on the high burden of subclinical CVD [in American adults with diabetes] and the potential utility of hs-cTnT for monitoring this risk in people with diabetes,” said Elizabeth Selvin, PhD, senior author and a professor of epidemiology at Johns Hopkins University, Baltimore.
“What we are seeing is that many people with type 2 diabetes who have not had a heart attack or a history of cardiovascular disease are at high risk for cardiovascular complications,” added Dr. Selvin in an AHA press release. “When we look at the whole population of people diagnosed with type 2 diabetes, about 27 million adults in the U.S., according to the [Centers for Disease Control and Prevention], some are at low risk and some are at high risk for cardiovascular disease, so the open question is: ‘Who is most at risk?’ These cardiac biomarkers give us a window into cardiovascular risk in people who otherwise might not be recognized as highest risk.”
“Our results provide evidence to support use of cardiac biomarkers for routine risk monitoring in high-risk populations such as people with diabetes,” Dr. Selvin noted in an interview.
Need for aggressive CVD risk reduction
The findings also indicate that people with diabetes and an elevated hs-cTnT “should be targeted for aggressive cardiovascular risk reduction, including lifestyle interventions, weight loss, and treatment with statins, blood pressure medications, and cardioprotective therapies such as sodium-glucose cotransporter 2 (SGLT-2) inhibitors and glucagonlike peptide-1 (GLP-1) receptor agonists,” Dr. Selvin added.
“Cholesterol is often the factor that we target to reduce the risk of cardiovascular disease in people with type 2 diabetes,” she observed. “However, type 2 diabetes may have a direct effect on the heart not related to cholesterol levels. If type 2 diabetes is directly causing damage to the small vessels in the heart unrelated to cholesterol plaque buildup, then cholesterol-lowering medications are not going to prevent cardiac damage,” Dr. Selvin explained. “Our research suggests that additional non–statin-related therapies are needed to lower the cardiovascular disease risk in people with type 2 diabetes.”
However, she noted that a necessary step prior to formally recommending such a strategy is to run clinical trials to assess the efficacy of specific treatments, such as SGLT-2 inhibitors and GLP-1 agonists, in people with diabetes and elevated hs-cTnT.
“Randomized controlled trials would be best to test the relevance of measuring these biomarkers to assess risk in asymptomatic people with diabetes,” as well as prospective study of the value of hs-cTnT to guide treatment, commented Robert H. Eckel, MD, an endocrinologist affiliated with the University of Colorado at Denver, Aurora.
“I doubt measurements [of hs-cTnT] would be reimbursed [by third-party payers] if carried out without such outcome data,” he added.
Dr. Eckel also highlights the need to further validate in additional cohorts the link between elevations in hs-cTnT and CVD events in adults with diabetes, and to confirm that elevated levels of another cardiac biomarker – N-terminal of the prohormone brain natriuretic peptide (NT-proBNP) – do not work as well as troponin as a risk marker for people with diabetes, another finding of the study.
ADA report already recommends testing these biomarkers for HF
However, a consensus report published in 2022 by the American Diabetes Association laid out the case for routinely and regularly measuring levels of both high sensitivity cardiac troponin and natriuretic peptides in people with diabetes for early identification of incident heart failure.
“Among individuals with diabetes, measurement of a natriuretic peptide or high-sensitivity cardiac troponin is recommended on at least a yearly basis to identify the earliest heart failure stages and implement strategies to prevent transition to symptomatic heart failure,” noted the ADA consensus report on heart failure.
The new study run by Dr. Selvin and coauthors used data collected by the National Health and Nutrition Examination Survey (NHANES) between 1999 and 2004 from U.S. adults who were at least 20 years old and had no history of CVD: myocardial infarction, stroke, coronary heart disease, or heart failure. This included 9,273 people without diabetes and 1,031 with diabetes, defined as a prior diagnosis or hemoglobin A1c of at least 6.5%.
“Cardiovascular risk varies substantially in adults with type 2 diabetes, highlighting the need for accurate risk stratification,” the authors observed.
All study participants had recorded measures of hs-cTnT and NT-proBNP.
The researchers considered an hs-cTnT level of greater than 14 ng/L and an NT-proBNP level of greater than 125 pg/mL as indicators of subclinical CVD.
The crude prevalence of elevated NT-proBNP was 33.4% among those with diabetes and 16.1% in those without diabetes. Elevated hs-cTnT occurred in 19% of those with diabetes and in 5% of those without diabetes. Elevated levels of both markers existed in 9% of those with diabetes and in 3% of those without diabetes.
“Approximately one in three adults with diabetes had subclinical CVD, with 19% having elevated levels of hs-cTnT, 23% having elevated NT-proBNP, and 9% having elevations in both cardiac biomarkers,” the researchers noted.
Diabetes linked with a doubled prevalence of elevated hs-cTnT
After adjustment for several demographic variables as well as traditional CVD risk factors, people with diabetes had a significant 98% higher rate of elevated hs-cTnT, compared with those without diabetes. But after similar adjustments, the rate of elevated NT-proBNP was significantly lower among people with diabetes, compared with controls, by a relative reduction of 24%.
“Our findings suggest that, in people with diabetes, hs-cTnT may be more useful [than NT-proBNP] for general risk monitoring, as its interpretation is less complicated,” said Dr. Selvin, who explained that “NT-proBNP is affected by overweight and obesity.”
In people with diabetes, the age-adjusted prevalence of elevated hs-cTnT ran higher in those with longer duration diabetes, and in those with less well-controlled diabetes based on a higher level of A1c. Neither of these factors showed any significant relationship with measured levels of NT-proBNP.
Further analysis linked the NHANES findings during 1999-2004 with U.S. national death records through the end of 2019. This showed that elevated levels of both hs-cTnT and NT-proBNP significantly linked with subsequently higher rates of all-cause mortality among people with diabetes. Elevated hs-cTnT linked with a 77% increased mortality and NT-proBNP linked with a 78% increased rate, compared with people with diabetes and no elevations in these markers, after adjustment for demographic variables and CVD risk factors.
However, for the outcome of cardiovascular death, elevated hs-cTnT linked with a nonsignificant 54% relative increase, while elevated NT-proBNP linked with a significant 2.46-fold relative increase.
The study “adds new data on biomarkers that are not routinely measured in asymptomatic people with or without diabetes” and the relationships of these markers to CVD mortality and all-cause mortality, Dr. Eckel concluded.
The study received no commercial funding, but used reagents donated by Abbott Laboratories, Ortho Clinical Diagnostics, Roche Diagnostics, and Siemens Healthcare Diagnostics. Dr. Selvin and Dr. Eckel had no disclosures.
A version of this article first appeared on Medscape.com.
– based on data from a representative sample of more than 10,000 U.S. adults.
The finding suggests hs-cTnT maybe a useful marker for adults with diabetes who could benefit from more aggressive CVD risk reduction despite having no clinical indications of CVD.
The results “highlight the substantial burden of subclinical CVD in persons with diabetes and emphasize the importance of early detection and treatment of CVD for this high-risk population,” say the authors of the research, published in the Journal of the American Heart Association.
“This is the first study to examine subclinical CVD, defined by elevated cardiac biomarkers, in a nationally representative population of adults with or without diabetes. It provides novel information on the high burden of subclinical CVD [in American adults with diabetes] and the potential utility of hs-cTnT for monitoring this risk in people with diabetes,” said Elizabeth Selvin, PhD, senior author and a professor of epidemiology at Johns Hopkins University, Baltimore.
“What we are seeing is that many people with type 2 diabetes who have not had a heart attack or a history of cardiovascular disease are at high risk for cardiovascular complications,” added Dr. Selvin in an AHA press release. “When we look at the whole population of people diagnosed with type 2 diabetes, about 27 million adults in the U.S., according to the [Centers for Disease Control and Prevention], some are at low risk and some are at high risk for cardiovascular disease, so the open question is: ‘Who is most at risk?’ These cardiac biomarkers give us a window into cardiovascular risk in people who otherwise might not be recognized as highest risk.”
“Our results provide evidence to support use of cardiac biomarkers for routine risk monitoring in high-risk populations such as people with diabetes,” Dr. Selvin noted in an interview.
Need for aggressive CVD risk reduction
The findings also indicate that people with diabetes and an elevated hs-cTnT “should be targeted for aggressive cardiovascular risk reduction, including lifestyle interventions, weight loss, and treatment with statins, blood pressure medications, and cardioprotective therapies such as sodium-glucose cotransporter 2 (SGLT-2) inhibitors and glucagonlike peptide-1 (GLP-1) receptor agonists,” Dr. Selvin added.
“Cholesterol is often the factor that we target to reduce the risk of cardiovascular disease in people with type 2 diabetes,” she observed. “However, type 2 diabetes may have a direct effect on the heart not related to cholesterol levels. If type 2 diabetes is directly causing damage to the small vessels in the heart unrelated to cholesterol plaque buildup, then cholesterol-lowering medications are not going to prevent cardiac damage,” Dr. Selvin explained. “Our research suggests that additional non–statin-related therapies are needed to lower the cardiovascular disease risk in people with type 2 diabetes.”
However, she noted that a necessary step prior to formally recommending such a strategy is to run clinical trials to assess the efficacy of specific treatments, such as SGLT-2 inhibitors and GLP-1 agonists, in people with diabetes and elevated hs-cTnT.
“Randomized controlled trials would be best to test the relevance of measuring these biomarkers to assess risk in asymptomatic people with diabetes,” as well as prospective study of the value of hs-cTnT to guide treatment, commented Robert H. Eckel, MD, an endocrinologist affiliated with the University of Colorado at Denver, Aurora.
“I doubt measurements [of hs-cTnT] would be reimbursed [by third-party payers] if carried out without such outcome data,” he added.
Dr. Eckel also highlights the need to further validate in additional cohorts the link between elevations in hs-cTnT and CVD events in adults with diabetes, and to confirm that elevated levels of another cardiac biomarker – N-terminal of the prohormone brain natriuretic peptide (NT-proBNP) – do not work as well as troponin as a risk marker for people with diabetes, another finding of the study.
ADA report already recommends testing these biomarkers for HF
However, a consensus report published in 2022 by the American Diabetes Association laid out the case for routinely and regularly measuring levels of both high sensitivity cardiac troponin and natriuretic peptides in people with diabetes for early identification of incident heart failure.
“Among individuals with diabetes, measurement of a natriuretic peptide or high-sensitivity cardiac troponin is recommended on at least a yearly basis to identify the earliest heart failure stages and implement strategies to prevent transition to symptomatic heart failure,” noted the ADA consensus report on heart failure.
The new study run by Dr. Selvin and coauthors used data collected by the National Health and Nutrition Examination Survey (NHANES) between 1999 and 2004 from U.S. adults who were at least 20 years old and had no history of CVD: myocardial infarction, stroke, coronary heart disease, or heart failure. This included 9,273 people without diabetes and 1,031 with diabetes, defined as a prior diagnosis or hemoglobin A1c of at least 6.5%.
“Cardiovascular risk varies substantially in adults with type 2 diabetes, highlighting the need for accurate risk stratification,” the authors observed.
All study participants had recorded measures of hs-cTnT and NT-proBNP.
The researchers considered an hs-cTnT level of greater than 14 ng/L and an NT-proBNP level of greater than 125 pg/mL as indicators of subclinical CVD.
The crude prevalence of elevated NT-proBNP was 33.4% among those with diabetes and 16.1% in those without diabetes. Elevated hs-cTnT occurred in 19% of those with diabetes and in 5% of those without diabetes. Elevated levels of both markers existed in 9% of those with diabetes and in 3% of those without diabetes.
“Approximately one in three adults with diabetes had subclinical CVD, with 19% having elevated levels of hs-cTnT, 23% having elevated NT-proBNP, and 9% having elevations in both cardiac biomarkers,” the researchers noted.
Diabetes linked with a doubled prevalence of elevated hs-cTnT
After adjustment for several demographic variables as well as traditional CVD risk factors, people with diabetes had a significant 98% higher rate of elevated hs-cTnT, compared with those without diabetes. But after similar adjustments, the rate of elevated NT-proBNP was significantly lower among people with diabetes, compared with controls, by a relative reduction of 24%.
“Our findings suggest that, in people with diabetes, hs-cTnT may be more useful [than NT-proBNP] for general risk monitoring, as its interpretation is less complicated,” said Dr. Selvin, who explained that “NT-proBNP is affected by overweight and obesity.”
In people with diabetes, the age-adjusted prevalence of elevated hs-cTnT ran higher in those with longer duration diabetes, and in those with less well-controlled diabetes based on a higher level of A1c. Neither of these factors showed any significant relationship with measured levels of NT-proBNP.
Further analysis linked the NHANES findings during 1999-2004 with U.S. national death records through the end of 2019. This showed that elevated levels of both hs-cTnT and NT-proBNP significantly linked with subsequently higher rates of all-cause mortality among people with diabetes. Elevated hs-cTnT linked with a 77% increased mortality and NT-proBNP linked with a 78% increased rate, compared with people with diabetes and no elevations in these markers, after adjustment for demographic variables and CVD risk factors.
However, for the outcome of cardiovascular death, elevated hs-cTnT linked with a nonsignificant 54% relative increase, while elevated NT-proBNP linked with a significant 2.46-fold relative increase.
The study “adds new data on biomarkers that are not routinely measured in asymptomatic people with or without diabetes” and the relationships of these markers to CVD mortality and all-cause mortality, Dr. Eckel concluded.
The study received no commercial funding, but used reagents donated by Abbott Laboratories, Ortho Clinical Diagnostics, Roche Diagnostics, and Siemens Healthcare Diagnostics. Dr. Selvin and Dr. Eckel had no disclosures.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN HEART ASSOCIATION
Few patients take weight control medications after bariatric surgery
CHICAGO –
Obesity is a chronic, relapsing condition that must be treated as such, said the study’s author Stephen A. Firkins, MD, a fellow of gastroenterology and hepatology at the Cleveland Clinic. “Barriers to antiobesity medications must be identified.”
“If a quarter of all patients experience weight regain and another quarter experience insufficient weight loss – but only 5% are being prescribed an FDA-approved AOM – that means there’s underutilization,” Dr. Firkins said.
Data from the National Health and Nutrition Examination Survey show that 30.7% of all men and women in the United States are overweight and of these, 42.4% are obese. For the severely obese, bariatric surgery, including sleeve gastrectomy, Roux-en-Y gastric bypass, and one anastomosis gastric bypass, are viable options with differing degrees of long-term success.
And while antiobesity medications such as orlistat (Xenical, Alli), phentermine/topiramate (Qsymia), naltrexone/bupropion (Contrave), liraglutide (Saxenda), semaglutide (Wegovy), and setmelanotide (Imcivree), may be considered before bariatric surgery, there are questions about the need for its use after surgery.
“While these are often employed or considered presurgically, there is a paucity of literature describing their utilization postsurgically, particularly in regards to newer antiobesity medications such as the [glucagonlike peptide–1] receptor agonists,” Dr. Firkins said.
The aim of Dr. Firkins’ analysis of the large, publicly available IBM Explorys Electronic Health Record, which included 59,160 adult post–bariatric surgery patients, was to identify postoperative weight control medication use and trends among different populations.
He found rates of postsurgical weight control medication use at 8% for topiramate (off label), 2.9% for liraglutide, 1.03% for phentermine/topiramate, 0.95% for naltrexone/bupropion, 0.52% for semaglutide and 0.1% for orlistat. Rates of topiramate use were higher for patients in the 35- 39-year range, and for orlistat and liraglutide in the 65- to 69-year range.
The differences, Dr. Firkins said, were likely related to side-effect profiles and accumulations of comorbidities with advancing age. Black patients were more likely to be prescribed the medications. Also, further analysis showed a significantly higher use of these medications among individuals with hypertension, diabetes, and hyperlipidemia.
The analyses raised several questions for future study, Dr. Firkins said: “What is the optimal timing of antiobesity medication initiation? Is it at the plateau of peak weight loss typically seen at 1-3 years post surgery? Or, after weight is regained? What is the phenotype of patients who are going to be good responders versus poor responders to a particular medication category?”
“Upon recognition of insufficient weight loss/weight regain, a multidisciplinary strategy towards management is warranted, including behavioral and dietary counseling, and consideration of AOM early use, as well as endoscopic or surgical revision,” he said.
Dr. Firkins had no disclosures.
DDW is sponsored by the American Association for the Study of Liver Diseases, the American Gastroenterological Association, the American Society for Gastrointestinal Endoscopy, and The Society for Surgery of the Alimentary Tract.
CHICAGO –
Obesity is a chronic, relapsing condition that must be treated as such, said the study’s author Stephen A. Firkins, MD, a fellow of gastroenterology and hepatology at the Cleveland Clinic. “Barriers to antiobesity medications must be identified.”
“If a quarter of all patients experience weight regain and another quarter experience insufficient weight loss – but only 5% are being prescribed an FDA-approved AOM – that means there’s underutilization,” Dr. Firkins said.
Data from the National Health and Nutrition Examination Survey show that 30.7% of all men and women in the United States are overweight and of these, 42.4% are obese. For the severely obese, bariatric surgery, including sleeve gastrectomy, Roux-en-Y gastric bypass, and one anastomosis gastric bypass, are viable options with differing degrees of long-term success.
And while antiobesity medications such as orlistat (Xenical, Alli), phentermine/topiramate (Qsymia), naltrexone/bupropion (Contrave), liraglutide (Saxenda), semaglutide (Wegovy), and setmelanotide (Imcivree), may be considered before bariatric surgery, there are questions about the need for its use after surgery.
“While these are often employed or considered presurgically, there is a paucity of literature describing their utilization postsurgically, particularly in regards to newer antiobesity medications such as the [glucagonlike peptide–1] receptor agonists,” Dr. Firkins said.
The aim of Dr. Firkins’ analysis of the large, publicly available IBM Explorys Electronic Health Record, which included 59,160 adult post–bariatric surgery patients, was to identify postoperative weight control medication use and trends among different populations.
He found rates of postsurgical weight control medication use at 8% for topiramate (off label), 2.9% for liraglutide, 1.03% for phentermine/topiramate, 0.95% for naltrexone/bupropion, 0.52% for semaglutide and 0.1% for orlistat. Rates of topiramate use were higher for patients in the 35- 39-year range, and for orlistat and liraglutide in the 65- to 69-year range.
The differences, Dr. Firkins said, were likely related to side-effect profiles and accumulations of comorbidities with advancing age. Black patients were more likely to be prescribed the medications. Also, further analysis showed a significantly higher use of these medications among individuals with hypertension, diabetes, and hyperlipidemia.
The analyses raised several questions for future study, Dr. Firkins said: “What is the optimal timing of antiobesity medication initiation? Is it at the plateau of peak weight loss typically seen at 1-3 years post surgery? Or, after weight is regained? What is the phenotype of patients who are going to be good responders versus poor responders to a particular medication category?”
“Upon recognition of insufficient weight loss/weight regain, a multidisciplinary strategy towards management is warranted, including behavioral and dietary counseling, and consideration of AOM early use, as well as endoscopic or surgical revision,” he said.
Dr. Firkins had no disclosures.
DDW is sponsored by the American Association for the Study of Liver Diseases, the American Gastroenterological Association, the American Society for Gastrointestinal Endoscopy, and The Society for Surgery of the Alimentary Tract.
CHICAGO –
Obesity is a chronic, relapsing condition that must be treated as such, said the study’s author Stephen A. Firkins, MD, a fellow of gastroenterology and hepatology at the Cleveland Clinic. “Barriers to antiobesity medications must be identified.”
“If a quarter of all patients experience weight regain and another quarter experience insufficient weight loss – but only 5% are being prescribed an FDA-approved AOM – that means there’s underutilization,” Dr. Firkins said.
Data from the National Health and Nutrition Examination Survey show that 30.7% of all men and women in the United States are overweight and of these, 42.4% are obese. For the severely obese, bariatric surgery, including sleeve gastrectomy, Roux-en-Y gastric bypass, and one anastomosis gastric bypass, are viable options with differing degrees of long-term success.
And while antiobesity medications such as orlistat (Xenical, Alli), phentermine/topiramate (Qsymia), naltrexone/bupropion (Contrave), liraglutide (Saxenda), semaglutide (Wegovy), and setmelanotide (Imcivree), may be considered before bariatric surgery, there are questions about the need for its use after surgery.
“While these are often employed or considered presurgically, there is a paucity of literature describing their utilization postsurgically, particularly in regards to newer antiobesity medications such as the [glucagonlike peptide–1] receptor agonists,” Dr. Firkins said.
The aim of Dr. Firkins’ analysis of the large, publicly available IBM Explorys Electronic Health Record, which included 59,160 adult post–bariatric surgery patients, was to identify postoperative weight control medication use and trends among different populations.
He found rates of postsurgical weight control medication use at 8% for topiramate (off label), 2.9% for liraglutide, 1.03% for phentermine/topiramate, 0.95% for naltrexone/bupropion, 0.52% for semaglutide and 0.1% for orlistat. Rates of topiramate use were higher for patients in the 35- 39-year range, and for orlistat and liraglutide in the 65- to 69-year range.
The differences, Dr. Firkins said, were likely related to side-effect profiles and accumulations of comorbidities with advancing age. Black patients were more likely to be prescribed the medications. Also, further analysis showed a significantly higher use of these medications among individuals with hypertension, diabetes, and hyperlipidemia.
The analyses raised several questions for future study, Dr. Firkins said: “What is the optimal timing of antiobesity medication initiation? Is it at the plateau of peak weight loss typically seen at 1-3 years post surgery? Or, after weight is regained? What is the phenotype of patients who are going to be good responders versus poor responders to a particular medication category?”
“Upon recognition of insufficient weight loss/weight regain, a multidisciplinary strategy towards management is warranted, including behavioral and dietary counseling, and consideration of AOM early use, as well as endoscopic or surgical revision,” he said.
Dr. Firkins had no disclosures.
DDW is sponsored by the American Association for the Study of Liver Diseases, the American Gastroenterological Association, the American Society for Gastrointestinal Endoscopy, and The Society for Surgery of the Alimentary Tract.
AT DDW 2023
Can a saliva test predict the best way to manage obesity?
It sounds like a simple solution to a complicated problem: Find out what kind of obesity someone has based on a one-time genetic saliva test. Then patients and their doctor can get a better idea if antiobesity drugs or other treatments are more likely to work for them.
It’s what Mayo Clinic researchers had in mind when they created four phenotypes of obesity.
Obesity experts not affiliated with the research have some concerns and say independent studies are needed to verify the potential of this strategy.
This research could help predict who will respond best to popular antiobesity medications, said Andres Acosta, MD, PhD, cofounder of Phenomix Sciences, the company behind the tests. These medications include glucagonlike peptide–1 (GLP-1) receptor agonists like liraglutide (Saxenda, Victoza) and semaglutide (Ozempic, Wegovy).
“We know that not everyone on a GLP-1 will respond. In reality, about a third of the patients don’t do well with GLP-1s,” said Dr. Acosta, an assistant professor of medicine and researcher in the division of gastroenterology and hepatology at the Mayo Clinic in Rochester, Minn.
Furthest along in development is the “My Phenome Hungry Gut” test for predicting GLP-1 response. People in this Hungry Gut group tend to empty their stomach after a meal faster and are more likely to feel hungry again a short time later, as explained on the company’s website.
A pilot study to test how well it works started in April at three primary care practices. Plans are to expand real-world testing for this and other obesity types later in 2023.
The other obesity categories are:
- “Hungry brain,” where the brain does not recognize signals that the stomach is full
- “Emotional hunger,” where cravings to eat are driven by emotions, anxiety, and negative feelings
- “Slow burn,” where people have a slow metabolism and low energy level
People in these categories might be more likely to benefit from other obesity management strategies, like changes to their diet or placement of an intragastric balloon.
Some things to consider
While applauding their efforts to be more precise in treating people with obesity, not all experts are convinced this saliva test will be the answer. The company’s research might look promising, but verification of results is warranted.
“Can we get better outcomes with things like this? Well, that’s the hope,” said Jaime Almandoz, MD, medical director of weight wellness at the University of Texas Southwestern Medical Center, Dallas.
“We still don’t have randomized trials where we’re looking at obesity phenotyping yet,” said Dr. Almandoz, who is also a spokesperson for the Obesity Society, a professional group of clinicians, researchers, educators, and others focused on obesity science, treatment, and prevention.
There is always concern when a diagnostic test is being developed for commercial use, said Daniel Bessesen, MD, a professor of medicine–endocrinology, metabolism, and diabetes at the University of Colorado at Denver, Aurora. “What they’re talking about doing is super important. But this is a company. This is a company that is, I think, selling a product.”
In an online search, Dr. Bessesen did not find any external studies that showed how well the saliva testing worked. But referring to work by Dr. Acosta and Michael Camilleri, MD, the other cofounder of Phenomix, he said, “I found some papers that they did that I hadn’t read before that are good.”
“These guys are smart guys. And they’ve done a lot of work on [the movement of food through the gut] and how that correlates with obesity and response to some therapies,” said Dr. Bessesen, who is also a spokesperson for the Obesity Society. “So their scientific work does line up with this area.”
Validation of any research is important because the obesity industry has been known for a lot of lose-weight-quick strategies, some with little or no science behind them, he said.
It is also essential, he said, because “anytime you do something commercial in the area of obesity, you have to acknowledge that people with obesity are a vulnerable population. These people face stigma and bias all the time.”
Removing the stigma
If knowing your obesity type ends up making a difference, it could change the conversation people have with their medical provider, Dr. Acosta said. It could also help remove some of the stigma around obesity.
“We’re going to change the conversation because now we can say: ‘Hey, you have obesity because you have ‘Hungry Gut’ phenotype. And because of that, you’re going to respond to this medication,” Dr. Acosta said. The phenotyping suggests a strong genetic tendency – a biologic basis for obesity.
“So it’s not only a way of taking the blame out, but it’s also way of explaining that there’s a reason why you have obesity,” Dr. Acosta said. It tells people: “You’re not a failure.”
More cost-effective treatment?
Targeting obesity treatment could also save on overall health care costs, Dr. Almandoz said. He estimated a cost of $1,400 per month “for forever and ever semaglutide” or at least $1,400 a month for a 3-month trial to see if this medication works in a particular person with obesity.
“That’s a lot of money when you extrapolate that out over the number of people who probably meet the criteria for treatment,” he said. A total 42% of Americans meet the Centers for Disease Control and Prevention definition for obesity.
“You can imagine the potential cost if we were to provide antiobesity therapies to everybody and we were to use what is the most effective class of medication, which is more than a thousand dollars per month, indefinitely,” Dr. Almandoz said. “Not that we should not treat everybody. That’s not the message I’m saying. But if we’re looking at yield or value in terms of treating obesity in a setting with limited resources, it may be best to start with who is most likely to benefit.”
How they created four obesity types
Starting in 2015, Dr. Acosta and colleagues started comparing tests in people with normal weight versus obesity. They used artificial intelligence and machine learning to classify obesity into 11 types at first. They realized this many obesity types were not practical for doctors and people with obesity, so they combined them into four phenotypes.
“The AI machine learning was followed by, as I like to call, HI, or human intelligence,” he said.
The saliva test checks for about 6,000 relevant genetic single-nucleotide polymorphisms. Six thousand genetic changes may sound like a large number to check; however, the average individual carries 5 million and 6 million SNPs in their DNA.
The results are translated to a score that yields a low risk or high risk for Hungry Gut or other types of obesity. “You can have all six thousand genetic mutations, or you can have zero,” Dr. Acosta said.
Moving forward
After the soft launch of Hungry Gut testing in April, Phenomix plans to continue studying their saliva test on other obesity types.
Dr. Acosta is not aware of any direct competitors to Phenomix, although that could change. “I think we’re the only diagnostic company in the space right now. But if it’s really a $14.8 billion market, we’re going to see a lot of diagnostic companies trying to do what we’re doing – if we’re successful,” he said.
An October 2022 report from Polaris Market Research estimates that the global market for obesity treatment – medications, surgery, and all others – was about $14 billion in 2021. The same report predicts the market will grow to $32 billion by 2030.
A version of this article first appeared on WebMD.com.
It sounds like a simple solution to a complicated problem: Find out what kind of obesity someone has based on a one-time genetic saliva test. Then patients and their doctor can get a better idea if antiobesity drugs or other treatments are more likely to work for them.
It’s what Mayo Clinic researchers had in mind when they created four phenotypes of obesity.
Obesity experts not affiliated with the research have some concerns and say independent studies are needed to verify the potential of this strategy.
This research could help predict who will respond best to popular antiobesity medications, said Andres Acosta, MD, PhD, cofounder of Phenomix Sciences, the company behind the tests. These medications include glucagonlike peptide–1 (GLP-1) receptor agonists like liraglutide (Saxenda, Victoza) and semaglutide (Ozempic, Wegovy).
“We know that not everyone on a GLP-1 will respond. In reality, about a third of the patients don’t do well with GLP-1s,” said Dr. Acosta, an assistant professor of medicine and researcher in the division of gastroenterology and hepatology at the Mayo Clinic in Rochester, Minn.
Furthest along in development is the “My Phenome Hungry Gut” test for predicting GLP-1 response. People in this Hungry Gut group tend to empty their stomach after a meal faster and are more likely to feel hungry again a short time later, as explained on the company’s website.
A pilot study to test how well it works started in April at three primary care practices. Plans are to expand real-world testing for this and other obesity types later in 2023.
The other obesity categories are:
- “Hungry brain,” where the brain does not recognize signals that the stomach is full
- “Emotional hunger,” where cravings to eat are driven by emotions, anxiety, and negative feelings
- “Slow burn,” where people have a slow metabolism and low energy level
People in these categories might be more likely to benefit from other obesity management strategies, like changes to their diet or placement of an intragastric balloon.
Some things to consider
While applauding their efforts to be more precise in treating people with obesity, not all experts are convinced this saliva test will be the answer. The company’s research might look promising, but verification of results is warranted.
“Can we get better outcomes with things like this? Well, that’s the hope,” said Jaime Almandoz, MD, medical director of weight wellness at the University of Texas Southwestern Medical Center, Dallas.
“We still don’t have randomized trials where we’re looking at obesity phenotyping yet,” said Dr. Almandoz, who is also a spokesperson for the Obesity Society, a professional group of clinicians, researchers, educators, and others focused on obesity science, treatment, and prevention.
There is always concern when a diagnostic test is being developed for commercial use, said Daniel Bessesen, MD, a professor of medicine–endocrinology, metabolism, and diabetes at the University of Colorado at Denver, Aurora. “What they’re talking about doing is super important. But this is a company. This is a company that is, I think, selling a product.”
In an online search, Dr. Bessesen did not find any external studies that showed how well the saliva testing worked. But referring to work by Dr. Acosta and Michael Camilleri, MD, the other cofounder of Phenomix, he said, “I found some papers that they did that I hadn’t read before that are good.”
“These guys are smart guys. And they’ve done a lot of work on [the movement of food through the gut] and how that correlates with obesity and response to some therapies,” said Dr. Bessesen, who is also a spokesperson for the Obesity Society. “So their scientific work does line up with this area.”
Validation of any research is important because the obesity industry has been known for a lot of lose-weight-quick strategies, some with little or no science behind them, he said.
It is also essential, he said, because “anytime you do something commercial in the area of obesity, you have to acknowledge that people with obesity are a vulnerable population. These people face stigma and bias all the time.”
Removing the stigma
If knowing your obesity type ends up making a difference, it could change the conversation people have with their medical provider, Dr. Acosta said. It could also help remove some of the stigma around obesity.
“We’re going to change the conversation because now we can say: ‘Hey, you have obesity because you have ‘Hungry Gut’ phenotype. And because of that, you’re going to respond to this medication,” Dr. Acosta said. The phenotyping suggests a strong genetic tendency – a biologic basis for obesity.
“So it’s not only a way of taking the blame out, but it’s also way of explaining that there’s a reason why you have obesity,” Dr. Acosta said. It tells people: “You’re not a failure.”
More cost-effective treatment?
Targeting obesity treatment could also save on overall health care costs, Dr. Almandoz said. He estimated a cost of $1,400 per month “for forever and ever semaglutide” or at least $1,400 a month for a 3-month trial to see if this medication works in a particular person with obesity.
“That’s a lot of money when you extrapolate that out over the number of people who probably meet the criteria for treatment,” he said. A total 42% of Americans meet the Centers for Disease Control and Prevention definition for obesity.
“You can imagine the potential cost if we were to provide antiobesity therapies to everybody and we were to use what is the most effective class of medication, which is more than a thousand dollars per month, indefinitely,” Dr. Almandoz said. “Not that we should not treat everybody. That’s not the message I’m saying. But if we’re looking at yield or value in terms of treating obesity in a setting with limited resources, it may be best to start with who is most likely to benefit.”
How they created four obesity types
Starting in 2015, Dr. Acosta and colleagues started comparing tests in people with normal weight versus obesity. They used artificial intelligence and machine learning to classify obesity into 11 types at first. They realized this many obesity types were not practical for doctors and people with obesity, so they combined them into four phenotypes.
“The AI machine learning was followed by, as I like to call, HI, or human intelligence,” he said.
The saliva test checks for about 6,000 relevant genetic single-nucleotide polymorphisms. Six thousand genetic changes may sound like a large number to check; however, the average individual carries 5 million and 6 million SNPs in their DNA.
The results are translated to a score that yields a low risk or high risk for Hungry Gut or other types of obesity. “You can have all six thousand genetic mutations, or you can have zero,” Dr. Acosta said.
Moving forward
After the soft launch of Hungry Gut testing in April, Phenomix plans to continue studying their saliva test on other obesity types.
Dr. Acosta is not aware of any direct competitors to Phenomix, although that could change. “I think we’re the only diagnostic company in the space right now. But if it’s really a $14.8 billion market, we’re going to see a lot of diagnostic companies trying to do what we’re doing – if we’re successful,” he said.
An October 2022 report from Polaris Market Research estimates that the global market for obesity treatment – medications, surgery, and all others – was about $14 billion in 2021. The same report predicts the market will grow to $32 billion by 2030.
A version of this article first appeared on WebMD.com.
It sounds like a simple solution to a complicated problem: Find out what kind of obesity someone has based on a one-time genetic saliva test. Then patients and their doctor can get a better idea if antiobesity drugs or other treatments are more likely to work for them.
It’s what Mayo Clinic researchers had in mind when they created four phenotypes of obesity.
Obesity experts not affiliated with the research have some concerns and say independent studies are needed to verify the potential of this strategy.
This research could help predict who will respond best to popular antiobesity medications, said Andres Acosta, MD, PhD, cofounder of Phenomix Sciences, the company behind the tests. These medications include glucagonlike peptide–1 (GLP-1) receptor agonists like liraglutide (Saxenda, Victoza) and semaglutide (Ozempic, Wegovy).
“We know that not everyone on a GLP-1 will respond. In reality, about a third of the patients don’t do well with GLP-1s,” said Dr. Acosta, an assistant professor of medicine and researcher in the division of gastroenterology and hepatology at the Mayo Clinic in Rochester, Minn.
Furthest along in development is the “My Phenome Hungry Gut” test for predicting GLP-1 response. People in this Hungry Gut group tend to empty their stomach after a meal faster and are more likely to feel hungry again a short time later, as explained on the company’s website.
A pilot study to test how well it works started in April at three primary care practices. Plans are to expand real-world testing for this and other obesity types later in 2023.
The other obesity categories are:
- “Hungry brain,” where the brain does not recognize signals that the stomach is full
- “Emotional hunger,” where cravings to eat are driven by emotions, anxiety, and negative feelings
- “Slow burn,” where people have a slow metabolism and low energy level
People in these categories might be more likely to benefit from other obesity management strategies, like changes to their diet or placement of an intragastric balloon.
Some things to consider
While applauding their efforts to be more precise in treating people with obesity, not all experts are convinced this saliva test will be the answer. The company’s research might look promising, but verification of results is warranted.
“Can we get better outcomes with things like this? Well, that’s the hope,” said Jaime Almandoz, MD, medical director of weight wellness at the University of Texas Southwestern Medical Center, Dallas.
“We still don’t have randomized trials where we’re looking at obesity phenotyping yet,” said Dr. Almandoz, who is also a spokesperson for the Obesity Society, a professional group of clinicians, researchers, educators, and others focused on obesity science, treatment, and prevention.
There is always concern when a diagnostic test is being developed for commercial use, said Daniel Bessesen, MD, a professor of medicine–endocrinology, metabolism, and diabetes at the University of Colorado at Denver, Aurora. “What they’re talking about doing is super important. But this is a company. This is a company that is, I think, selling a product.”
In an online search, Dr. Bessesen did not find any external studies that showed how well the saliva testing worked. But referring to work by Dr. Acosta and Michael Camilleri, MD, the other cofounder of Phenomix, he said, “I found some papers that they did that I hadn’t read before that are good.”
“These guys are smart guys. And they’ve done a lot of work on [the movement of food through the gut] and how that correlates with obesity and response to some therapies,” said Dr. Bessesen, who is also a spokesperson for the Obesity Society. “So their scientific work does line up with this area.”
Validation of any research is important because the obesity industry has been known for a lot of lose-weight-quick strategies, some with little or no science behind them, he said.
It is also essential, he said, because “anytime you do something commercial in the area of obesity, you have to acknowledge that people with obesity are a vulnerable population. These people face stigma and bias all the time.”
Removing the stigma
If knowing your obesity type ends up making a difference, it could change the conversation people have with their medical provider, Dr. Acosta said. It could also help remove some of the stigma around obesity.
“We’re going to change the conversation because now we can say: ‘Hey, you have obesity because you have ‘Hungry Gut’ phenotype. And because of that, you’re going to respond to this medication,” Dr. Acosta said. The phenotyping suggests a strong genetic tendency – a biologic basis for obesity.
“So it’s not only a way of taking the blame out, but it’s also way of explaining that there’s a reason why you have obesity,” Dr. Acosta said. It tells people: “You’re not a failure.”
More cost-effective treatment?
Targeting obesity treatment could also save on overall health care costs, Dr. Almandoz said. He estimated a cost of $1,400 per month “for forever and ever semaglutide” or at least $1,400 a month for a 3-month trial to see if this medication works in a particular person with obesity.
“That’s a lot of money when you extrapolate that out over the number of people who probably meet the criteria for treatment,” he said. A total 42% of Americans meet the Centers for Disease Control and Prevention definition for obesity.
“You can imagine the potential cost if we were to provide antiobesity therapies to everybody and we were to use what is the most effective class of medication, which is more than a thousand dollars per month, indefinitely,” Dr. Almandoz said. “Not that we should not treat everybody. That’s not the message I’m saying. But if we’re looking at yield or value in terms of treating obesity in a setting with limited resources, it may be best to start with who is most likely to benefit.”
How they created four obesity types
Starting in 2015, Dr. Acosta and colleagues started comparing tests in people with normal weight versus obesity. They used artificial intelligence and machine learning to classify obesity into 11 types at first. They realized this many obesity types were not practical for doctors and people with obesity, so they combined them into four phenotypes.
“The AI machine learning was followed by, as I like to call, HI, or human intelligence,” he said.
The saliva test checks for about 6,000 relevant genetic single-nucleotide polymorphisms. Six thousand genetic changes may sound like a large number to check; however, the average individual carries 5 million and 6 million SNPs in their DNA.
The results are translated to a score that yields a low risk or high risk for Hungry Gut or other types of obesity. “You can have all six thousand genetic mutations, or you can have zero,” Dr. Acosta said.
Moving forward
After the soft launch of Hungry Gut testing in April, Phenomix plans to continue studying their saliva test on other obesity types.
Dr. Acosta is not aware of any direct competitors to Phenomix, although that could change. “I think we’re the only diagnostic company in the space right now. But if it’s really a $14.8 billion market, we’re going to see a lot of diagnostic companies trying to do what we’re doing – if we’re successful,” he said.
An October 2022 report from Polaris Market Research estimates that the global market for obesity treatment – medications, surgery, and all others – was about $14 billion in 2021. The same report predicts the market will grow to $32 billion by 2030.
A version of this article first appeared on WebMD.com.
FDA approves new drug, sotagliflozin, for heart failure
Sotagliflozin, a novel agent that inhibits sodium-glucose cotransporter 1 as well as SGLT2, has received marketing approval from the Food and Drug Administration for reducing the risk for cardiovascular death, hospitalization for heart failure, and urgent heart failure visits in patients with heart failure, and also for preventing these same events in patients with type 2 diabetes, chronic kidney disease (CKD), and other cardiovascular disease risk factors.
This puts sotagliflozin in direct competition with two SGLT2 inhibitors, dapagliflozin (Farxiga) and empagliflozin (Jardiance), that already have indications for preventing heart failure hospitalizations in patients with heart failure as well as approvals for type 2 diabetes and preservation of renal function.
Officials at Lexicon Pharmaceuticals, the company that developed and will market sotagliflozin under the trade name Inpefa, said in a press release that they expect U.S. sales of the agent to begin before the end of June 2023. The release also highlighted that the approval broadly covered use in patients with heart failure across the full range of both reduced and preserved left ventricular ejection fractions.
They base this niche target for sotagliflozin on results from the SOLOIST-WHF trial, which randomized 1,222 patients with type 2 diabetes recently hospitalized for worsening heart failure and showed a significant 33% reduction in the rate of deaths from cardiovascular causes and hospitalizations and urgent visits for heart failure, compared with control patients during a median 9 months of follow-up. Nearly half of the enrolled patients received their first dose while still hospitalized, while the other half received their first dose a median of 2 days after hospital discharge. The drug appeared safe.
Cutting heart failure rehospitalizations in half
An exploratory post hoc analysis of SOLOIST-WHF showed that treatment with sotagliflozin cut the rate of rehospitalizations roughly in half after both 30 and 90 days compared with control patients, according to an abstract presented at the 2022 annual scientific sessions of the AHA that has not yet been published in a peer-reviewed journal.
The only SGLT2 inhibitor tested so far when initiated in patients during hospitalization for heart failure is empagliflozin, in the EMPULSE trial, which randomized 530 patients. EMPULSE also showed that starting an SGLT2 inhibitor in this setting was safe and resulted in significant clinical benefit, the study’s primary endpoint, defined as a composite of death from any cause, number of heart failure events, and time to first heart failure event, or a 5-point or greater difference in change from baseline in the Kansas City Cardiomyopathy Questionnaire Total Symptom Score at 90 days.
In the DELIVER trial, which tested dapagliflozin in patients with heart failure with preserved ejection fraction, roughly 10% of patients started study treatment during or within 30 days of heart failure hospitalization, and in this subgroup, dapagliflozin appeared as effective as it was in the other 90% of patients who did not start the drug during an acute or subacute phase.
Despite the SOLOIST-WHF evidence for sotagliflozin’s safety and efficacy in this economically important clinical setting, some experts say the drug faces an uphill path as it contends for market share against two solidly established, albeit dramatically underused, SGLT2 inhibitors. (Recent data document that 20% or fewer of U.S. patients eligible for treatment with an SGLT2 inhibitor receive it, such as a review of 49,000 patients hospitalized during 2021-2022 with heart failure with reduced ejection fraction.)
Others foresee a clear role for sotagliflozin, particularly because of additional facets of the drug’s performance in trials that they perceive give it an edge over dapagliflozin and empagliflozin. This includes evidence that sotagliflozin treatment uniquely (within the SGLT2 inhibitor class) cuts the rate of strokes and myocardial infarctions, as well as evidence of its apparent ability to lower hemoglobin A1c levels in patients with type 2 diabetes and with an estimated glomerular filtration rate below 30 mL/min per 1.73 m2, a property likely linked to inhibition of SGLT1 in the gut that dampens intestinal glucose absorption.
Sotagliflozin uptake ‘will be a challenge’
“It will be a challenge” for sotagliflozin uptake, given the head start that both dapagliflozin and empagliflozin have had as well-documented agents for patients with heart failure, commented Javed Butler, MD, a heart failure clinician and trialist who is president of the Baylor Scott & White Research Institute in Dallas.
Given the position dapagliflozin and empagliflozin currently have in U.S. heart failure management – with the SGLT2 inhibitor class called out in guidelines as foundational for treating patients with heart failure with reduced ejection fraction and likely soon for heart failure with preserved ejection fraction as well – “I can’t imagine [sotagliflozin] will be considered a preferred option,” Dr. Butler said in an interview.
Another expert was even more dismissive of sotagliflozin’s role.
“There is no persuasive evidence that sotagliflozin has any advantages, compared with the SGLT2 inhibitors, for the treatment of heart failure,” said Milton Packer, MD, a heart failure specialist and trialist at Baylor University Medical Center, Dallas. “I do not see why U.S. physicians might pivot from established SGLT2 inhibitors to sotagliflozin,” unless it was priced “at a very meaningful discount to available SGLT2 inhibitors.”
At the time it announced the FDA’s approval, Lexicon did not provide details on how it would price sotagliflozin. Existing retail prices for dapagliflozin and empagliflozin run about $550-$600/month, a price point that has contributed to slow U.S. uptake of the drug class. But experts anticipate a dramatic shake-up of the U.S. market for SGLT2 inhibitors with expected introduction of a generic SGLT2 inhibitor formulation by 2025, a development that could further dampen sotagliflozin’s prospects.
Other experts are more optimistic about the new agent’s uptake, perhaps none more than Deepak L. Bhatt, MD, MPH, who led both pivotal trials that provide the bulk of sotagliflozin’s evidence package.
In addition to SOLOIST-WHF, Dr. Bhatt also headed the SCORED trial, with 10,584 patients with type 2 diabetes, CKD, and risks for cardiovascular disease randomized to sotagliflozin or placebo and followed for a median of 16 months. The primary result showed that sotagliflozin treatment cut the combined rate of deaths from cardiovascular causes, hospitalizations for heart failure, and urgent visits for heart failure by a significant 26% relative to control patients.
A clear MACE benefit
“The data from SOLOIST-WHF and SCORED look at least as good as the data for the SGLT2 inhibitors for heart failure, and what appears to be different are the rates for MI and stroke in SCORED,” said Dr. Bhatt, director of Mount Sinai Heart, New York.
“I believe the rate of major adverse cardiovascular events [MACE] were reduced [in SCORED], and this is different from the SGLT2 inhibitors,” he said in an interview.
In 2022, Dr. Bhatt reported results from a prespecified secondary analysis of SCORED that showed that treatment with sotagliflozin cut the rate of MACE by a significant 21%-26%, compared with placebo. This finding was, in part, driven by the first data to show a substantial benefit from an SGLT inhibitor on stroke rates.
And while SCORED did not report a significant benefit for slowing progression of CKD, subsequent post hoc analyses have suggested this advantage also in as-yet-unpublished findings, Dr. Bhatt added.
But he said he doubted nephrologists will see it as a first-line agent for slowing CKD progression – an indication already held by dapagliflozin, pending for empagliflozin, and also in place for a third SGLT2 inhibitor, canagliflozin (Invokana) – because sotagliflozin lacks clear significant and prespecified evidence for this effect.
Dr. Bhatt also acknowledged the limitation of sotagliflozin compared with the SGLT2 inhibitors as an agent for glucose control, again because of no evidence for this effect from a prospective analysis and no pending indication for type 2 diabetes treatment. But the SCORED data showed a clear A1c benefit, even in patients with severely reduced renal function.
Mostly for cardiologists? ‘Compelling’ reductions in MIs and strokes
That may mean sotagliflozin “won’t get much use by endocrinologists nor by primary care physicians,” commented Carol L. Wysham, MD, an endocrinologist with MultiCare in Spokane, Wash.
Sotagliflozin “will be a cardiology drug,” and will “have a hard time” competing with the SGLT2 inhibitors, she predicted.
Dr. Bhatt agreed that sotagliflozin “will be perceived as a drug for cardiologists to prescribe. I don’t see endocrinologists, nephrologists, and primary care physicians reaching for this drug if it has a heart failure label.” But, he added, “my hope is that the company files for additional indications. It deserves an indication for glycemic control.”
The evidence for a heart failure benefit from sotagliflozin is “valid and compelling,” and “having this option is great,” commented Mikhail N. Kosiborod, MD, a cardiologist, vice president of research at Saint Luke’s Health System, and codirector of the Haverty Cardiometabolic Center of Excellence at Saint Luke’s Mid America Heart Institute in Kansas City, Mo. But, he added, “it will be a reasonably tall task for sotagliflozin to come from behind and be disruptive in a space where there are already two well-established SGLT2 inhibitors” approved for preventing heart failure hospitalizations, “with a lot of data to back them up,”
The feature that sets sotagliflozin apart from the approved SGLT2 inhibitors is the “really compelling decrease” it produced in rates of MIs and strokes “that we simply do not see with SGLT2 inhibitors,” Dr. Kosiborod said in an interview.
He also cited results from SCORED that suggest “a meaningful reduction in A1c” when indirectly compared with SGLT2 inhibitors, especially in patients with more severe CKD. The lack of a dedicated A1c-lowering trial or an approved type 2 diabetes indication “will not be a problem for cardiologists,” he predicted, but also agreed that it is less likely to be used by primary care physicians in low-risk patients.
“I can see myself prescribing sotagliflozin,” said Dr. Kosiborod, a SCORED coinvestigator, especially for patients with coexisting type 2 diabetes, heart failure, CKD, and atherosclerotic cardiovascular disease. These patients may get “more bang for the buck” because of a reduced risk for MI and stroke, making sotagliflozin “a solid consideration in these patients if the economic factors align.”
Like others, Dr. Kosiborod cited the big impact pricing will have, especially if, as expected, a generic SGLT2 inhibitor soon comes onto the U.S. market. “Access and affordability are very important.”
SOLOIST-WHF and SCORED were sponsored initially by Sanofi and later by Lexicon after Sanofi pulled out of sotagliflozin development. Dr. Butler has been a consultant for Lexicon as well as for AstraZeneca (which markets Farxiga), Boehringer Ingelheim and Lilly (which jointly market Jardiance), and Janssen (which markets Invokana), as well as for numerous other companies. Dr. Packer has been a consultant for AstraZeneca, Boehringer Ingelheim, Lilly, and numerous other companies. Dr. Bhatt was lead investigator for SOLOIST-WHF and SCORED and has been an adviser for Boehringer Ingelheim and Janssen and numerous other companies. Dr. Wysham has been an adviser, speaker, and consultant for AstraZeneca, Boehringer Ingelheim, Lilly, Janssen, Novo Nordisk, and Sanofi, an adviser for Abbott, and a speaker for Insulet. Dr. Kosiborod was a member of the SCORED Steering Committee and has been a consultant for Lexicon, AstraZeneca, Boehringer Ingelheim, Janssen, Lilly, Novo Nordisk, and numerous other companies.
A version of this article first appeared on Medscape.com.
Sotagliflozin, a novel agent that inhibits sodium-glucose cotransporter 1 as well as SGLT2, has received marketing approval from the Food and Drug Administration for reducing the risk for cardiovascular death, hospitalization for heart failure, and urgent heart failure visits in patients with heart failure, and also for preventing these same events in patients with type 2 diabetes, chronic kidney disease (CKD), and other cardiovascular disease risk factors.
This puts sotagliflozin in direct competition with two SGLT2 inhibitors, dapagliflozin (Farxiga) and empagliflozin (Jardiance), that already have indications for preventing heart failure hospitalizations in patients with heart failure as well as approvals for type 2 diabetes and preservation of renal function.
Officials at Lexicon Pharmaceuticals, the company that developed and will market sotagliflozin under the trade name Inpefa, said in a press release that they expect U.S. sales of the agent to begin before the end of June 2023. The release also highlighted that the approval broadly covered use in patients with heart failure across the full range of both reduced and preserved left ventricular ejection fractions.
They base this niche target for sotagliflozin on results from the SOLOIST-WHF trial, which randomized 1,222 patients with type 2 diabetes recently hospitalized for worsening heart failure and showed a significant 33% reduction in the rate of deaths from cardiovascular causes and hospitalizations and urgent visits for heart failure, compared with control patients during a median 9 months of follow-up. Nearly half of the enrolled patients received their first dose while still hospitalized, while the other half received their first dose a median of 2 days after hospital discharge. The drug appeared safe.
Cutting heart failure rehospitalizations in half
An exploratory post hoc analysis of SOLOIST-WHF showed that treatment with sotagliflozin cut the rate of rehospitalizations roughly in half after both 30 and 90 days compared with control patients, according to an abstract presented at the 2022 annual scientific sessions of the AHA that has not yet been published in a peer-reviewed journal.
The only SGLT2 inhibitor tested so far when initiated in patients during hospitalization for heart failure is empagliflozin, in the EMPULSE trial, which randomized 530 patients. EMPULSE also showed that starting an SGLT2 inhibitor in this setting was safe and resulted in significant clinical benefit, the study’s primary endpoint, defined as a composite of death from any cause, number of heart failure events, and time to first heart failure event, or a 5-point or greater difference in change from baseline in the Kansas City Cardiomyopathy Questionnaire Total Symptom Score at 90 days.
In the DELIVER trial, which tested dapagliflozin in patients with heart failure with preserved ejection fraction, roughly 10% of patients started study treatment during or within 30 days of heart failure hospitalization, and in this subgroup, dapagliflozin appeared as effective as it was in the other 90% of patients who did not start the drug during an acute or subacute phase.
Despite the SOLOIST-WHF evidence for sotagliflozin’s safety and efficacy in this economically important clinical setting, some experts say the drug faces an uphill path as it contends for market share against two solidly established, albeit dramatically underused, SGLT2 inhibitors. (Recent data document that 20% or fewer of U.S. patients eligible for treatment with an SGLT2 inhibitor receive it, such as a review of 49,000 patients hospitalized during 2021-2022 with heart failure with reduced ejection fraction.)
Others foresee a clear role for sotagliflozin, particularly because of additional facets of the drug’s performance in trials that they perceive give it an edge over dapagliflozin and empagliflozin. This includes evidence that sotagliflozin treatment uniquely (within the SGLT2 inhibitor class) cuts the rate of strokes and myocardial infarctions, as well as evidence of its apparent ability to lower hemoglobin A1c levels in patients with type 2 diabetes and with an estimated glomerular filtration rate below 30 mL/min per 1.73 m2, a property likely linked to inhibition of SGLT1 in the gut that dampens intestinal glucose absorption.
Sotagliflozin uptake ‘will be a challenge’
“It will be a challenge” for sotagliflozin uptake, given the head start that both dapagliflozin and empagliflozin have had as well-documented agents for patients with heart failure, commented Javed Butler, MD, a heart failure clinician and trialist who is president of the Baylor Scott & White Research Institute in Dallas.
Given the position dapagliflozin and empagliflozin currently have in U.S. heart failure management – with the SGLT2 inhibitor class called out in guidelines as foundational for treating patients with heart failure with reduced ejection fraction and likely soon for heart failure with preserved ejection fraction as well – “I can’t imagine [sotagliflozin] will be considered a preferred option,” Dr. Butler said in an interview.
Another expert was even more dismissive of sotagliflozin’s role.
“There is no persuasive evidence that sotagliflozin has any advantages, compared with the SGLT2 inhibitors, for the treatment of heart failure,” said Milton Packer, MD, a heart failure specialist and trialist at Baylor University Medical Center, Dallas. “I do not see why U.S. physicians might pivot from established SGLT2 inhibitors to sotagliflozin,” unless it was priced “at a very meaningful discount to available SGLT2 inhibitors.”
At the time it announced the FDA’s approval, Lexicon did not provide details on how it would price sotagliflozin. Existing retail prices for dapagliflozin and empagliflozin run about $550-$600/month, a price point that has contributed to slow U.S. uptake of the drug class. But experts anticipate a dramatic shake-up of the U.S. market for SGLT2 inhibitors with expected introduction of a generic SGLT2 inhibitor formulation by 2025, a development that could further dampen sotagliflozin’s prospects.
Other experts are more optimistic about the new agent’s uptake, perhaps none more than Deepak L. Bhatt, MD, MPH, who led both pivotal trials that provide the bulk of sotagliflozin’s evidence package.
In addition to SOLOIST-WHF, Dr. Bhatt also headed the SCORED trial, with 10,584 patients with type 2 diabetes, CKD, and risks for cardiovascular disease randomized to sotagliflozin or placebo and followed for a median of 16 months. The primary result showed that sotagliflozin treatment cut the combined rate of deaths from cardiovascular causes, hospitalizations for heart failure, and urgent visits for heart failure by a significant 26% relative to control patients.
A clear MACE benefit
“The data from SOLOIST-WHF and SCORED look at least as good as the data for the SGLT2 inhibitors for heart failure, and what appears to be different are the rates for MI and stroke in SCORED,” said Dr. Bhatt, director of Mount Sinai Heart, New York.
“I believe the rate of major adverse cardiovascular events [MACE] were reduced [in SCORED], and this is different from the SGLT2 inhibitors,” he said in an interview.
In 2022, Dr. Bhatt reported results from a prespecified secondary analysis of SCORED that showed that treatment with sotagliflozin cut the rate of MACE by a significant 21%-26%, compared with placebo. This finding was, in part, driven by the first data to show a substantial benefit from an SGLT inhibitor on stroke rates.
And while SCORED did not report a significant benefit for slowing progression of CKD, subsequent post hoc analyses have suggested this advantage also in as-yet-unpublished findings, Dr. Bhatt added.
But he said he doubted nephrologists will see it as a first-line agent for slowing CKD progression – an indication already held by dapagliflozin, pending for empagliflozin, and also in place for a third SGLT2 inhibitor, canagliflozin (Invokana) – because sotagliflozin lacks clear significant and prespecified evidence for this effect.
Dr. Bhatt also acknowledged the limitation of sotagliflozin compared with the SGLT2 inhibitors as an agent for glucose control, again because of no evidence for this effect from a prospective analysis and no pending indication for type 2 diabetes treatment. But the SCORED data showed a clear A1c benefit, even in patients with severely reduced renal function.
Mostly for cardiologists? ‘Compelling’ reductions in MIs and strokes
That may mean sotagliflozin “won’t get much use by endocrinologists nor by primary care physicians,” commented Carol L. Wysham, MD, an endocrinologist with MultiCare in Spokane, Wash.
Sotagliflozin “will be a cardiology drug,” and will “have a hard time” competing with the SGLT2 inhibitors, she predicted.
Dr. Bhatt agreed that sotagliflozin “will be perceived as a drug for cardiologists to prescribe. I don’t see endocrinologists, nephrologists, and primary care physicians reaching for this drug if it has a heart failure label.” But, he added, “my hope is that the company files for additional indications. It deserves an indication for glycemic control.”
The evidence for a heart failure benefit from sotagliflozin is “valid and compelling,” and “having this option is great,” commented Mikhail N. Kosiborod, MD, a cardiologist, vice president of research at Saint Luke’s Health System, and codirector of the Haverty Cardiometabolic Center of Excellence at Saint Luke’s Mid America Heart Institute in Kansas City, Mo. But, he added, “it will be a reasonably tall task for sotagliflozin to come from behind and be disruptive in a space where there are already two well-established SGLT2 inhibitors” approved for preventing heart failure hospitalizations, “with a lot of data to back them up,”
The feature that sets sotagliflozin apart from the approved SGLT2 inhibitors is the “really compelling decrease” it produced in rates of MIs and strokes “that we simply do not see with SGLT2 inhibitors,” Dr. Kosiborod said in an interview.
He also cited results from SCORED that suggest “a meaningful reduction in A1c” when indirectly compared with SGLT2 inhibitors, especially in patients with more severe CKD. The lack of a dedicated A1c-lowering trial or an approved type 2 diabetes indication “will not be a problem for cardiologists,” he predicted, but also agreed that it is less likely to be used by primary care physicians in low-risk patients.
“I can see myself prescribing sotagliflozin,” said Dr. Kosiborod, a SCORED coinvestigator, especially for patients with coexisting type 2 diabetes, heart failure, CKD, and atherosclerotic cardiovascular disease. These patients may get “more bang for the buck” because of a reduced risk for MI and stroke, making sotagliflozin “a solid consideration in these patients if the economic factors align.”
Like others, Dr. Kosiborod cited the big impact pricing will have, especially if, as expected, a generic SGLT2 inhibitor soon comes onto the U.S. market. “Access and affordability are very important.”
SOLOIST-WHF and SCORED were sponsored initially by Sanofi and later by Lexicon after Sanofi pulled out of sotagliflozin development. Dr. Butler has been a consultant for Lexicon as well as for AstraZeneca (which markets Farxiga), Boehringer Ingelheim and Lilly (which jointly market Jardiance), and Janssen (which markets Invokana), as well as for numerous other companies. Dr. Packer has been a consultant for AstraZeneca, Boehringer Ingelheim, Lilly, and numerous other companies. Dr. Bhatt was lead investigator for SOLOIST-WHF and SCORED and has been an adviser for Boehringer Ingelheim and Janssen and numerous other companies. Dr. Wysham has been an adviser, speaker, and consultant for AstraZeneca, Boehringer Ingelheim, Lilly, Janssen, Novo Nordisk, and Sanofi, an adviser for Abbott, and a speaker for Insulet. Dr. Kosiborod was a member of the SCORED Steering Committee and has been a consultant for Lexicon, AstraZeneca, Boehringer Ingelheim, Janssen, Lilly, Novo Nordisk, and numerous other companies.
A version of this article first appeared on Medscape.com.
Sotagliflozin, a novel agent that inhibits sodium-glucose cotransporter 1 as well as SGLT2, has received marketing approval from the Food and Drug Administration for reducing the risk for cardiovascular death, hospitalization for heart failure, and urgent heart failure visits in patients with heart failure, and also for preventing these same events in patients with type 2 diabetes, chronic kidney disease (CKD), and other cardiovascular disease risk factors.
This puts sotagliflozin in direct competition with two SGLT2 inhibitors, dapagliflozin (Farxiga) and empagliflozin (Jardiance), that already have indications for preventing heart failure hospitalizations in patients with heart failure as well as approvals for type 2 diabetes and preservation of renal function.
Officials at Lexicon Pharmaceuticals, the company that developed and will market sotagliflozin under the trade name Inpefa, said in a press release that they expect U.S. sales of the agent to begin before the end of June 2023. The release also highlighted that the approval broadly covered use in patients with heart failure across the full range of both reduced and preserved left ventricular ejection fractions.
They base this niche target for sotagliflozin on results from the SOLOIST-WHF trial, which randomized 1,222 patients with type 2 diabetes recently hospitalized for worsening heart failure and showed a significant 33% reduction in the rate of deaths from cardiovascular causes and hospitalizations and urgent visits for heart failure, compared with control patients during a median 9 months of follow-up. Nearly half of the enrolled patients received their first dose while still hospitalized, while the other half received their first dose a median of 2 days after hospital discharge. The drug appeared safe.
Cutting heart failure rehospitalizations in half
An exploratory post hoc analysis of SOLOIST-WHF showed that treatment with sotagliflozin cut the rate of rehospitalizations roughly in half after both 30 and 90 days compared with control patients, according to an abstract presented at the 2022 annual scientific sessions of the AHA that has not yet been published in a peer-reviewed journal.
The only SGLT2 inhibitor tested so far when initiated in patients during hospitalization for heart failure is empagliflozin, in the EMPULSE trial, which randomized 530 patients. EMPULSE also showed that starting an SGLT2 inhibitor in this setting was safe and resulted in significant clinical benefit, the study’s primary endpoint, defined as a composite of death from any cause, number of heart failure events, and time to first heart failure event, or a 5-point or greater difference in change from baseline in the Kansas City Cardiomyopathy Questionnaire Total Symptom Score at 90 days.
In the DELIVER trial, which tested dapagliflozin in patients with heart failure with preserved ejection fraction, roughly 10% of patients started study treatment during or within 30 days of heart failure hospitalization, and in this subgroup, dapagliflozin appeared as effective as it was in the other 90% of patients who did not start the drug during an acute or subacute phase.
Despite the SOLOIST-WHF evidence for sotagliflozin’s safety and efficacy in this economically important clinical setting, some experts say the drug faces an uphill path as it contends for market share against two solidly established, albeit dramatically underused, SGLT2 inhibitors. (Recent data document that 20% or fewer of U.S. patients eligible for treatment with an SGLT2 inhibitor receive it, such as a review of 49,000 patients hospitalized during 2021-2022 with heart failure with reduced ejection fraction.)
Others foresee a clear role for sotagliflozin, particularly because of additional facets of the drug’s performance in trials that they perceive give it an edge over dapagliflozin and empagliflozin. This includes evidence that sotagliflozin treatment uniquely (within the SGLT2 inhibitor class) cuts the rate of strokes and myocardial infarctions, as well as evidence of its apparent ability to lower hemoglobin A1c levels in patients with type 2 diabetes and with an estimated glomerular filtration rate below 30 mL/min per 1.73 m2, a property likely linked to inhibition of SGLT1 in the gut that dampens intestinal glucose absorption.
Sotagliflozin uptake ‘will be a challenge’
“It will be a challenge” for sotagliflozin uptake, given the head start that both dapagliflozin and empagliflozin have had as well-documented agents for patients with heart failure, commented Javed Butler, MD, a heart failure clinician and trialist who is president of the Baylor Scott & White Research Institute in Dallas.
Given the position dapagliflozin and empagliflozin currently have in U.S. heart failure management – with the SGLT2 inhibitor class called out in guidelines as foundational for treating patients with heart failure with reduced ejection fraction and likely soon for heart failure with preserved ejection fraction as well – “I can’t imagine [sotagliflozin] will be considered a preferred option,” Dr. Butler said in an interview.
Another expert was even more dismissive of sotagliflozin’s role.
“There is no persuasive evidence that sotagliflozin has any advantages, compared with the SGLT2 inhibitors, for the treatment of heart failure,” said Milton Packer, MD, a heart failure specialist and trialist at Baylor University Medical Center, Dallas. “I do not see why U.S. physicians might pivot from established SGLT2 inhibitors to sotagliflozin,” unless it was priced “at a very meaningful discount to available SGLT2 inhibitors.”
At the time it announced the FDA’s approval, Lexicon did not provide details on how it would price sotagliflozin. Existing retail prices for dapagliflozin and empagliflozin run about $550-$600/month, a price point that has contributed to slow U.S. uptake of the drug class. But experts anticipate a dramatic shake-up of the U.S. market for SGLT2 inhibitors with expected introduction of a generic SGLT2 inhibitor formulation by 2025, a development that could further dampen sotagliflozin’s prospects.
Other experts are more optimistic about the new agent’s uptake, perhaps none more than Deepak L. Bhatt, MD, MPH, who led both pivotal trials that provide the bulk of sotagliflozin’s evidence package.
In addition to SOLOIST-WHF, Dr. Bhatt also headed the SCORED trial, with 10,584 patients with type 2 diabetes, CKD, and risks for cardiovascular disease randomized to sotagliflozin or placebo and followed for a median of 16 months. The primary result showed that sotagliflozin treatment cut the combined rate of deaths from cardiovascular causes, hospitalizations for heart failure, and urgent visits for heart failure by a significant 26% relative to control patients.
A clear MACE benefit
“The data from SOLOIST-WHF and SCORED look at least as good as the data for the SGLT2 inhibitors for heart failure, and what appears to be different are the rates for MI and stroke in SCORED,” said Dr. Bhatt, director of Mount Sinai Heart, New York.
“I believe the rate of major adverse cardiovascular events [MACE] were reduced [in SCORED], and this is different from the SGLT2 inhibitors,” he said in an interview.
In 2022, Dr. Bhatt reported results from a prespecified secondary analysis of SCORED that showed that treatment with sotagliflozin cut the rate of MACE by a significant 21%-26%, compared with placebo. This finding was, in part, driven by the first data to show a substantial benefit from an SGLT inhibitor on stroke rates.
And while SCORED did not report a significant benefit for slowing progression of CKD, subsequent post hoc analyses have suggested this advantage also in as-yet-unpublished findings, Dr. Bhatt added.
But he said he doubted nephrologists will see it as a first-line agent for slowing CKD progression – an indication already held by dapagliflozin, pending for empagliflozin, and also in place for a third SGLT2 inhibitor, canagliflozin (Invokana) – because sotagliflozin lacks clear significant and prespecified evidence for this effect.
Dr. Bhatt also acknowledged the limitation of sotagliflozin compared with the SGLT2 inhibitors as an agent for glucose control, again because of no evidence for this effect from a prospective analysis and no pending indication for type 2 diabetes treatment. But the SCORED data showed a clear A1c benefit, even in patients with severely reduced renal function.
Mostly for cardiologists? ‘Compelling’ reductions in MIs and strokes
That may mean sotagliflozin “won’t get much use by endocrinologists nor by primary care physicians,” commented Carol L. Wysham, MD, an endocrinologist with MultiCare in Spokane, Wash.
Sotagliflozin “will be a cardiology drug,” and will “have a hard time” competing with the SGLT2 inhibitors, she predicted.
Dr. Bhatt agreed that sotagliflozin “will be perceived as a drug for cardiologists to prescribe. I don’t see endocrinologists, nephrologists, and primary care physicians reaching for this drug if it has a heart failure label.” But, he added, “my hope is that the company files for additional indications. It deserves an indication for glycemic control.”
The evidence for a heart failure benefit from sotagliflozin is “valid and compelling,” and “having this option is great,” commented Mikhail N. Kosiborod, MD, a cardiologist, vice president of research at Saint Luke’s Health System, and codirector of the Haverty Cardiometabolic Center of Excellence at Saint Luke’s Mid America Heart Institute in Kansas City, Mo. But, he added, “it will be a reasonably tall task for sotagliflozin to come from behind and be disruptive in a space where there are already two well-established SGLT2 inhibitors” approved for preventing heart failure hospitalizations, “with a lot of data to back them up,”
The feature that sets sotagliflozin apart from the approved SGLT2 inhibitors is the “really compelling decrease” it produced in rates of MIs and strokes “that we simply do not see with SGLT2 inhibitors,” Dr. Kosiborod said in an interview.
He also cited results from SCORED that suggest “a meaningful reduction in A1c” when indirectly compared with SGLT2 inhibitors, especially in patients with more severe CKD. The lack of a dedicated A1c-lowering trial or an approved type 2 diabetes indication “will not be a problem for cardiologists,” he predicted, but also agreed that it is less likely to be used by primary care physicians in low-risk patients.
“I can see myself prescribing sotagliflozin,” said Dr. Kosiborod, a SCORED coinvestigator, especially for patients with coexisting type 2 diabetes, heart failure, CKD, and atherosclerotic cardiovascular disease. These patients may get “more bang for the buck” because of a reduced risk for MI and stroke, making sotagliflozin “a solid consideration in these patients if the economic factors align.”
Like others, Dr. Kosiborod cited the big impact pricing will have, especially if, as expected, a generic SGLT2 inhibitor soon comes onto the U.S. market. “Access and affordability are very important.”
SOLOIST-WHF and SCORED were sponsored initially by Sanofi and later by Lexicon after Sanofi pulled out of sotagliflozin development. Dr. Butler has been a consultant for Lexicon as well as for AstraZeneca (which markets Farxiga), Boehringer Ingelheim and Lilly (which jointly market Jardiance), and Janssen (which markets Invokana), as well as for numerous other companies. Dr. Packer has been a consultant for AstraZeneca, Boehringer Ingelheim, Lilly, and numerous other companies. Dr. Bhatt was lead investigator for SOLOIST-WHF and SCORED and has been an adviser for Boehringer Ingelheim and Janssen and numerous other companies. Dr. Wysham has been an adviser, speaker, and consultant for AstraZeneca, Boehringer Ingelheim, Lilly, Janssen, Novo Nordisk, and Sanofi, an adviser for Abbott, and a speaker for Insulet. Dr. Kosiborod was a member of the SCORED Steering Committee and has been a consultant for Lexicon, AstraZeneca, Boehringer Ingelheim, Janssen, Lilly, Novo Nordisk, and numerous other companies.
A version of this article first appeared on Medscape.com.
Plant-based diet tied to healthier blood lipid levels
, in a new meta-analysis of 30 trials.
The findings suggest that “plant-based diets have the potential to lessen the atherosclerotic burden from atherogenic lipoproteins and thereby reduce the risk of cardiovascular disease,” write Caroline Amelie Koch, a medical student at the University of Copenhagen, and colleagues. Their findings were published online in the European Heart Journal (2023 May 24. doi: 10.1093/eurheartj/ehad211).
“Vegetarian and vegan diets were associated with a 14% reduction in all artery-clogging lipoproteins as indicated by apoB,” senior author Ruth Frikke-Schmidt, DMSc, PhD, Rigshospitalet, Copenhagen, and professor, University of Copenhagen, said in a press release from her university.
“This corresponds to a third of the effect of taking cholesterol-lowering medications such as statins,” she added, “and would result in a 7% reduction in the risk of cardiovascular disease in someone who maintained a plant-based diet for 5 years.”
“Importantly, we found similar results, across continents, ages, different ranges of body mass index, and among people in different states of health,” Dr. Frikke-Schmidt stressed.
And combining statins with plant-based diets would likely produce a synergistic effect, she speculated.
“If people start eating vegetarian or vegan diets from an early age,” she said, “the potential for reducing the risk of cardiovascular disease caused by blocked arteries is substantial.”
In addition, the researchers conclude: “Shifting to plant-based diets at a populational level will reduce emissions of greenhouse gases considerably – together making these diets efficient means [moving] towards a more sustainable development, while at the same time reducing the growing burden of atherosclerotic cardiovascular disease.”
More support for vegan, vegetarian diets
These new findings “add to the body of evidence supporting favorable effects of healthy vegan and vegetarian dietary patterns on circulating levels of LDL-C and atherogenic lipoproteins, which would be expected to reduce ASCVD risk,” Kevin C. Maki, PhD, and Carol Kirkpatrick, PhD, MPH, write in an accompanying editorial.
“While it is not necessary to entirely omit foods such as meat, poultry, and fish/seafood to follow a recommended dietary pattern, reducing consumption of such foods is a reasonable option for those who prefer to do so,” note Dr. Maki, of Indiana University School of Public Health, Bloomington, and Kirkpatrick, of Idaho State University, Pocatello.
Plant-based diet needs to be ‘well-planned’
Several experts who were not involved in this meta-analysis shed light on the study and its implications in comments to the U.K. Science Media Center.
“Although a vegetarian and vegan diet can be very healthy and beneficial with respect to cardiovascular risk, it is important that it is well planned so that nutrients it can be low in are included, including iron, iodine, vitamin B12, and vitamin D,” said Duane Mellor, PhD, a registered dietitian and senior lecturer, Aston Medical School, Aston University, Birmingham, England.
Some people “may find it easier to follow a Mediterranean-style diet that features plenty of fruit, vegetables, pulses, wholegrains, fish, eggs and low-fat dairy, with only small amounts of meat,” Tracy Parker, senior dietitian at the British Heart Foundation, London, suggested.
“There is considerable evidence that this type of diet can help lower your risk of developing heart and circulatory diseases by improving cholesterol and blood pressure levels, reducing inflammation, and controlling blood glucose levels,” she added.
And Aedin Cassidy, PhD, chair in nutrition & preventative medicine, Queen’s University Belfast (Ireland), noted that “not all plant-based diets are equal. Healthy plant-based diets, characterized by fruits, vegetables, and whole grains improve health, but other plant diets (for example, those including refined carbohydrates, processed foods high in fat/salt, etc.) do not.”
This new study shows that plant-based diets have the potential to improve health by improving blood lipids, “but this is one of many potential mechanisms, including impact on blood pressure, weight maintenance, and blood sugars,” she added.
“This work represents a well-conducted analysis of 30 clinical trials involving over two thousand participants and highlights the value of a vegetarian diet in reducing the risk of heart attack or stroke through reduction in blood cholesterol levels,” said Robert Storey, BM, DM, professor of cardiology, University of Sheffield, U.K.
However, it also demonstrates that the impact of diet on an individual’s cholesterol level is relatively limited, he added.
“This is because people inherit the tendency for their livers to produce too much cholesterol, meaning that high cholesterol is more strongly influenced by our genes than by our diet,” he explained.
This is “why statins are needed to block cholesterol production in people who are at higher risk of or have already suffered from a heart attack, stroke, or other illness related to cholesterol build-up in blood vessels.”
Beneficial effect on ApoB, LDL-C, and total cholesterol
ApoB is the main apolipoprotein in LDL-C (“bad” cholesterol), the researchers note. Previous studies have shown that LDL-C and apoB-containing particles are associated with increased risk of ASCVD.
They aimed to estimate the effect of vegetarian or vegan diets on blood levels of total cholesterol, LDL-C, triglycerides, and apoB in people randomized to a vegetarian or vegan diet versus an omnivorous diet (that is, including meat and dairy).
They identified 30 studies published between 1982 and 2022 and conducted in the United States (18 studies), Sweden (2), Finland (2), South Korea (2), Australia (1), Brazil (1), Czech Republic (1), Italy (1), Iran (1), and New Zealand (1).
The diet interventions lasted from 10 days to 5 years with a mean of 29 weeks (15 studies ≤ 3 months; 12 studies 3-12 months; and three studies > 1 year). Nine studies used a crossover design, and the rest used a parallel design whereby participants followed only one diet.
The studies had 11 to 291 participants (mean, 79 participants) with a mean BMI of 21.5-35.1 kg/m2 and a mean age of 20-67 years. Thirteen studies included participants treated with lipid-lowering therapy at baseline.
The dietary intervention was vegetarian in 15 trials (three lacto-vegetarian and 12 lacto-ovo-vegetarian) and vegan in the other 15 trials.
On average, compared with people eating an omnivore diet, people eating a plant-based diet had a 7% reduction in total cholesterol from baseline (–0.34 mmol/L), a 10% reduction in LDL-C from baseline (–0.30 mmol/L), and a 14% reduction in apoB from baseline (–12.9 mg/dL) (all P < .01).
The effects were similar across age, continent, study duration, health status, intervention diet, intervention program, and study design subgroups.
There was no significant difference in triglyceride levels in patients in the omnivore versus plant-based diet groups.
Such diets could considerably reduce greenhouse gases
Senior author Dr. Frikke-Schmidt noted: “Recent systematic reviews have shown that if the populations of high-income countries shift to plant-based diets, this can reduce net emissions of greenhouse gases by between 35% to 49%.”
“Plant-based diets are key instruments for changing food production to more environmentally sustainable forms, while at the same time reducing the burden of cardiovascular disease” in an aging population, she said.
“We should be eating a varied, plant-rich diet, not too much, and quenching our thirst with water,” she concluded.
The study was funded by the Lundbeck Foundation, the Danish Heart Foundation, and the Leducq Foundation. The authors, editorialists, Ms. Parker, Dr. Cassidy, and Dr. Storey have reported no relevant financial relationships. Dr. Mellor has disclosed that he is a vegetarian.
A version of this article first appeared on Medscape.com.
, in a new meta-analysis of 30 trials.
The findings suggest that “plant-based diets have the potential to lessen the atherosclerotic burden from atherogenic lipoproteins and thereby reduce the risk of cardiovascular disease,” write Caroline Amelie Koch, a medical student at the University of Copenhagen, and colleagues. Their findings were published online in the European Heart Journal (2023 May 24. doi: 10.1093/eurheartj/ehad211).
“Vegetarian and vegan diets were associated with a 14% reduction in all artery-clogging lipoproteins as indicated by apoB,” senior author Ruth Frikke-Schmidt, DMSc, PhD, Rigshospitalet, Copenhagen, and professor, University of Copenhagen, said in a press release from her university.
“This corresponds to a third of the effect of taking cholesterol-lowering medications such as statins,” she added, “and would result in a 7% reduction in the risk of cardiovascular disease in someone who maintained a plant-based diet for 5 years.”
“Importantly, we found similar results, across continents, ages, different ranges of body mass index, and among people in different states of health,” Dr. Frikke-Schmidt stressed.
And combining statins with plant-based diets would likely produce a synergistic effect, she speculated.
“If people start eating vegetarian or vegan diets from an early age,” she said, “the potential for reducing the risk of cardiovascular disease caused by blocked arteries is substantial.”
In addition, the researchers conclude: “Shifting to plant-based diets at a populational level will reduce emissions of greenhouse gases considerably – together making these diets efficient means [moving] towards a more sustainable development, while at the same time reducing the growing burden of atherosclerotic cardiovascular disease.”
More support for vegan, vegetarian diets
These new findings “add to the body of evidence supporting favorable effects of healthy vegan and vegetarian dietary patterns on circulating levels of LDL-C and atherogenic lipoproteins, which would be expected to reduce ASCVD risk,” Kevin C. Maki, PhD, and Carol Kirkpatrick, PhD, MPH, write in an accompanying editorial.
“While it is not necessary to entirely omit foods such as meat, poultry, and fish/seafood to follow a recommended dietary pattern, reducing consumption of such foods is a reasonable option for those who prefer to do so,” note Dr. Maki, of Indiana University School of Public Health, Bloomington, and Kirkpatrick, of Idaho State University, Pocatello.
Plant-based diet needs to be ‘well-planned’
Several experts who were not involved in this meta-analysis shed light on the study and its implications in comments to the U.K. Science Media Center.
“Although a vegetarian and vegan diet can be very healthy and beneficial with respect to cardiovascular risk, it is important that it is well planned so that nutrients it can be low in are included, including iron, iodine, vitamin B12, and vitamin D,” said Duane Mellor, PhD, a registered dietitian and senior lecturer, Aston Medical School, Aston University, Birmingham, England.
Some people “may find it easier to follow a Mediterranean-style diet that features plenty of fruit, vegetables, pulses, wholegrains, fish, eggs and low-fat dairy, with only small amounts of meat,” Tracy Parker, senior dietitian at the British Heart Foundation, London, suggested.
“There is considerable evidence that this type of diet can help lower your risk of developing heart and circulatory diseases by improving cholesterol and blood pressure levels, reducing inflammation, and controlling blood glucose levels,” she added.
And Aedin Cassidy, PhD, chair in nutrition & preventative medicine, Queen’s University Belfast (Ireland), noted that “not all plant-based diets are equal. Healthy plant-based diets, characterized by fruits, vegetables, and whole grains improve health, but other plant diets (for example, those including refined carbohydrates, processed foods high in fat/salt, etc.) do not.”
This new study shows that plant-based diets have the potential to improve health by improving blood lipids, “but this is one of many potential mechanisms, including impact on blood pressure, weight maintenance, and blood sugars,” she added.
“This work represents a well-conducted analysis of 30 clinical trials involving over two thousand participants and highlights the value of a vegetarian diet in reducing the risk of heart attack or stroke through reduction in blood cholesterol levels,” said Robert Storey, BM, DM, professor of cardiology, University of Sheffield, U.K.
However, it also demonstrates that the impact of diet on an individual’s cholesterol level is relatively limited, he added.
“This is because people inherit the tendency for their livers to produce too much cholesterol, meaning that high cholesterol is more strongly influenced by our genes than by our diet,” he explained.
This is “why statins are needed to block cholesterol production in people who are at higher risk of or have already suffered from a heart attack, stroke, or other illness related to cholesterol build-up in blood vessels.”
Beneficial effect on ApoB, LDL-C, and total cholesterol
ApoB is the main apolipoprotein in LDL-C (“bad” cholesterol), the researchers note. Previous studies have shown that LDL-C and apoB-containing particles are associated with increased risk of ASCVD.
They aimed to estimate the effect of vegetarian or vegan diets on blood levels of total cholesterol, LDL-C, triglycerides, and apoB in people randomized to a vegetarian or vegan diet versus an omnivorous diet (that is, including meat and dairy).
They identified 30 studies published between 1982 and 2022 and conducted in the United States (18 studies), Sweden (2), Finland (2), South Korea (2), Australia (1), Brazil (1), Czech Republic (1), Italy (1), Iran (1), and New Zealand (1).
The diet interventions lasted from 10 days to 5 years with a mean of 29 weeks (15 studies ≤ 3 months; 12 studies 3-12 months; and three studies > 1 year). Nine studies used a crossover design, and the rest used a parallel design whereby participants followed only one diet.
The studies had 11 to 291 participants (mean, 79 participants) with a mean BMI of 21.5-35.1 kg/m2 and a mean age of 20-67 years. Thirteen studies included participants treated with lipid-lowering therapy at baseline.
The dietary intervention was vegetarian in 15 trials (three lacto-vegetarian and 12 lacto-ovo-vegetarian) and vegan in the other 15 trials.
On average, compared with people eating an omnivore diet, people eating a plant-based diet had a 7% reduction in total cholesterol from baseline (–0.34 mmol/L), a 10% reduction in LDL-C from baseline (–0.30 mmol/L), and a 14% reduction in apoB from baseline (–12.9 mg/dL) (all P < .01).
The effects were similar across age, continent, study duration, health status, intervention diet, intervention program, and study design subgroups.
There was no significant difference in triglyceride levels in patients in the omnivore versus plant-based diet groups.
Such diets could considerably reduce greenhouse gases
Senior author Dr. Frikke-Schmidt noted: “Recent systematic reviews have shown that if the populations of high-income countries shift to plant-based diets, this can reduce net emissions of greenhouse gases by between 35% to 49%.”
“Plant-based diets are key instruments for changing food production to more environmentally sustainable forms, while at the same time reducing the burden of cardiovascular disease” in an aging population, she said.
“We should be eating a varied, plant-rich diet, not too much, and quenching our thirst with water,” she concluded.
The study was funded by the Lundbeck Foundation, the Danish Heart Foundation, and the Leducq Foundation. The authors, editorialists, Ms. Parker, Dr. Cassidy, and Dr. Storey have reported no relevant financial relationships. Dr. Mellor has disclosed that he is a vegetarian.
A version of this article first appeared on Medscape.com.
, in a new meta-analysis of 30 trials.
The findings suggest that “plant-based diets have the potential to lessen the atherosclerotic burden from atherogenic lipoproteins and thereby reduce the risk of cardiovascular disease,” write Caroline Amelie Koch, a medical student at the University of Copenhagen, and colleagues. Their findings were published online in the European Heart Journal (2023 May 24. doi: 10.1093/eurheartj/ehad211).
“Vegetarian and vegan diets were associated with a 14% reduction in all artery-clogging lipoproteins as indicated by apoB,” senior author Ruth Frikke-Schmidt, DMSc, PhD, Rigshospitalet, Copenhagen, and professor, University of Copenhagen, said in a press release from her university.
“This corresponds to a third of the effect of taking cholesterol-lowering medications such as statins,” she added, “and would result in a 7% reduction in the risk of cardiovascular disease in someone who maintained a plant-based diet for 5 years.”
“Importantly, we found similar results, across continents, ages, different ranges of body mass index, and among people in different states of health,” Dr. Frikke-Schmidt stressed.
And combining statins with plant-based diets would likely produce a synergistic effect, she speculated.
“If people start eating vegetarian or vegan diets from an early age,” she said, “the potential for reducing the risk of cardiovascular disease caused by blocked arteries is substantial.”
In addition, the researchers conclude: “Shifting to plant-based diets at a populational level will reduce emissions of greenhouse gases considerably – together making these diets efficient means [moving] towards a more sustainable development, while at the same time reducing the growing burden of atherosclerotic cardiovascular disease.”
More support for vegan, vegetarian diets
These new findings “add to the body of evidence supporting favorable effects of healthy vegan and vegetarian dietary patterns on circulating levels of LDL-C and atherogenic lipoproteins, which would be expected to reduce ASCVD risk,” Kevin C. Maki, PhD, and Carol Kirkpatrick, PhD, MPH, write in an accompanying editorial.
“While it is not necessary to entirely omit foods such as meat, poultry, and fish/seafood to follow a recommended dietary pattern, reducing consumption of such foods is a reasonable option for those who prefer to do so,” note Dr. Maki, of Indiana University School of Public Health, Bloomington, and Kirkpatrick, of Idaho State University, Pocatello.
Plant-based diet needs to be ‘well-planned’
Several experts who were not involved in this meta-analysis shed light on the study and its implications in comments to the U.K. Science Media Center.
“Although a vegetarian and vegan diet can be very healthy and beneficial with respect to cardiovascular risk, it is important that it is well planned so that nutrients it can be low in are included, including iron, iodine, vitamin B12, and vitamin D,” said Duane Mellor, PhD, a registered dietitian and senior lecturer, Aston Medical School, Aston University, Birmingham, England.
Some people “may find it easier to follow a Mediterranean-style diet that features plenty of fruit, vegetables, pulses, wholegrains, fish, eggs and low-fat dairy, with only small amounts of meat,” Tracy Parker, senior dietitian at the British Heart Foundation, London, suggested.
“There is considerable evidence that this type of diet can help lower your risk of developing heart and circulatory diseases by improving cholesterol and blood pressure levels, reducing inflammation, and controlling blood glucose levels,” she added.
And Aedin Cassidy, PhD, chair in nutrition & preventative medicine, Queen’s University Belfast (Ireland), noted that “not all plant-based diets are equal. Healthy plant-based diets, characterized by fruits, vegetables, and whole grains improve health, but other plant diets (for example, those including refined carbohydrates, processed foods high in fat/salt, etc.) do not.”
This new study shows that plant-based diets have the potential to improve health by improving blood lipids, “but this is one of many potential mechanisms, including impact on blood pressure, weight maintenance, and blood sugars,” she added.
“This work represents a well-conducted analysis of 30 clinical trials involving over two thousand participants and highlights the value of a vegetarian diet in reducing the risk of heart attack or stroke through reduction in blood cholesterol levels,” said Robert Storey, BM, DM, professor of cardiology, University of Sheffield, U.K.
However, it also demonstrates that the impact of diet on an individual’s cholesterol level is relatively limited, he added.
“This is because people inherit the tendency for their livers to produce too much cholesterol, meaning that high cholesterol is more strongly influenced by our genes than by our diet,” he explained.
This is “why statins are needed to block cholesterol production in people who are at higher risk of or have already suffered from a heart attack, stroke, or other illness related to cholesterol build-up in blood vessels.”
Beneficial effect on ApoB, LDL-C, and total cholesterol
ApoB is the main apolipoprotein in LDL-C (“bad” cholesterol), the researchers note. Previous studies have shown that LDL-C and apoB-containing particles are associated with increased risk of ASCVD.
They aimed to estimate the effect of vegetarian or vegan diets on blood levels of total cholesterol, LDL-C, triglycerides, and apoB in people randomized to a vegetarian or vegan diet versus an omnivorous diet (that is, including meat and dairy).
They identified 30 studies published between 1982 and 2022 and conducted in the United States (18 studies), Sweden (2), Finland (2), South Korea (2), Australia (1), Brazil (1), Czech Republic (1), Italy (1), Iran (1), and New Zealand (1).
The diet interventions lasted from 10 days to 5 years with a mean of 29 weeks (15 studies ≤ 3 months; 12 studies 3-12 months; and three studies > 1 year). Nine studies used a crossover design, and the rest used a parallel design whereby participants followed only one diet.
The studies had 11 to 291 participants (mean, 79 participants) with a mean BMI of 21.5-35.1 kg/m2 and a mean age of 20-67 years. Thirteen studies included participants treated with lipid-lowering therapy at baseline.
The dietary intervention was vegetarian in 15 trials (three lacto-vegetarian and 12 lacto-ovo-vegetarian) and vegan in the other 15 trials.
On average, compared with people eating an omnivore diet, people eating a plant-based diet had a 7% reduction in total cholesterol from baseline (–0.34 mmol/L), a 10% reduction in LDL-C from baseline (–0.30 mmol/L), and a 14% reduction in apoB from baseline (–12.9 mg/dL) (all P < .01).
The effects were similar across age, continent, study duration, health status, intervention diet, intervention program, and study design subgroups.
There was no significant difference in triglyceride levels in patients in the omnivore versus plant-based diet groups.
Such diets could considerably reduce greenhouse gases
Senior author Dr. Frikke-Schmidt noted: “Recent systematic reviews have shown that if the populations of high-income countries shift to plant-based diets, this can reduce net emissions of greenhouse gases by between 35% to 49%.”
“Plant-based diets are key instruments for changing food production to more environmentally sustainable forms, while at the same time reducing the burden of cardiovascular disease” in an aging population, she said.
“We should be eating a varied, plant-rich diet, not too much, and quenching our thirst with water,” she concluded.
The study was funded by the Lundbeck Foundation, the Danish Heart Foundation, and the Leducq Foundation. The authors, editorialists, Ms. Parker, Dr. Cassidy, and Dr. Storey have reported no relevant financial relationships. Dr. Mellor has disclosed that he is a vegetarian.
A version of this article first appeared on Medscape.com.
FROM EUROPEAN HEART JOURNAL
States move to curb insurers’ prior authorization requirements as federal reforms lag
Amid growing criticism of health insurers’ onerous prior authorization practices, lawmakers in 30 states have introduced bills this year that aim to rein in insurer gatekeeping and improve patient care.
“This is something that goes on in every doctor’s office every day; the frustrations, the delays, and the use of office staff time are just unbelievable,” said Steven Orland, MD, a board-certified urologist and president of the Medical Society of New Jersey.
The bills, which cover private health plans and insurers that states regulate, may provide some relief for physicians as federal efforts to streamline prior authorization for some Medicare patients have lagged.
Last year, Congress failed to pass the Improving Seniors’ Timely Access to Care Act of 2021, despite 326 co-sponsors. The bill would have compelled insurers covering Medicare Advantage enrollees to speed up prior authorizations, make the process more transparent, and remove obstacles such as requiring fax machine submissions.
Last month, however, the Centers for Medicare & Medicaid Services issued a final rule that will improve some aspects of prior authorizations in Medicare Advantage insurance plans and ensure that enrollees have the same access to necessary care as traditional Medicare enrollees.
The insurance industry has long defended prior authorization requirements and opposed legislation that would limit them.
America’s Health Insurance Plans (AHIP) and the Blue Cross Blue Shield Association said in a 2019 letter to a congressional committee when the federal legislation was first introduced, “Prior authorizations enforce best practices and guidelines for care management and help physicians identify and avoid care techniques that would harm patient outcomes, such as designating prescriptions that could feed into an opioid addiction.” AHIP didn’t respond to repeated requests for comment.
But some major insurers now appear willing to compromise and voluntarily reduce the volume of prior authorizations they require. Days before the federal final rule was released, three major insurers – United HealthCare, Cigna, and Aetna CVS Health – announced they plan to drop some prior authorization requirements and automate processes.
United HealthCare said it will eliminate almost 20% of its prior authorizations for some nonurgent surgeries and procedures starting this summer. It also will create a national Gold Card program in 2024 for physicians who meet its eligibility requirements, which would eliminate prior authorization requirements for most procedures. Both initiatives will apply to commercial, Medicare Advantage, and Medicaid businesses, said the insurer in a statement.
However, United HealthCare also announced that in June it will start requiring prior authorization for diagnostic (not screening) gastrointestinal endoscopies for its nearly 27 million privately insured patients, citing data it says shows potentially harmful overuse of scopes. Physician groups have publicly criticized the move, saying it could delay lifesaving treatment, and have asked the insurer to reconsider.
Cigna and Aetna also have moved to pare back prior authorization processes. Scott Josephs, national medical officer for Cigna, told Healthcare Dive that Cigna has removed prior authorization reviews from nearly 500 services since 2020.
An Aetna spokesperson told Healthcare Dive that the CVS-owned payer has implemented a gold card program and rolled back prior authorization requirements on cataract surgeries, video EEGs, and home infusion for some drugs, according to Healthcare Dive.
Cigna has faced increased scrutiny from some state regulators since a ProPublica/The Capitol Forum article revealed in March that its doctors were denying claims without opening patients’ files, contrary to what insurance laws and regulations require in many states.
Over a period of 2 months last year, Cigna doctors denied over 300,000 requests for payments using this method, spending an average of 1.2 seconds on each case, the investigation found. In a written response, Cigna said the reporting by ProPublica and The Capitol Forum was “biased and incomplete.”
States aim to reduce prior authorization volume
The American Medical Association said it has been tracking nearly 90 prior authorization reform bills in 30 states. More than a dozen bills are still being considered in this legislative session, including in Arkansas, California, New Jersey, North Carolina, Maryland, and Washington, D.C.
“The groundswell of activity in the states reflects how big a problem this is,” said an AMA legislative expert. “The issue used to be ‘how can we automate and streamline processes’; now the issue is focused on reducing the volume of prior authorizations and the harm that can cause patients.”
The state bills use different strategies to reduce excessive prior authorization requirements. Maryland’s proposed bill, for example, would require just one prior authorization to stay on a prescription drug, if the insurer has previously approved the drug and the patient continues to successfully be treated by the drug.
Washington, D.C. and New Jersey have introduced comprehensive reform bills that include a “grace period” of 60 days, to ensure continuity of care when a patient switches health plans. They also would eliminate repeat authorizations for chronic and long-term conditions, set explicit timelines for insurers to respond to prior authorization requests and appeals, and require that practicing physicians review denials that are appealed.
Many state bills also would require insurers to be more transparent by posting information on their websites about which services and drugs require prior authorization and what their approval rates are for them, said AMA’s legislative expert.
“There’s a black hole of information that insurers have access to. We would really like to know how many prior authorization requests are denied, the time it takes to deny them, and the reasons for denial,” said Josh Bengal, JD, the director of government relations for the Medical Society of New Jersey.
The legislation in New Jersey and other states faces stiff opposition from the insurance lobby, especially state associations of health plans affiliated with AHIP. The California Association of Health Plans, for example, opposes a “gold card” bill (SB 598), introduced in February, that would allow a select group of high-performing doctors to skip prior authorizations for 1 year.
The CAHP states, “Californians deserve safe, high quality, high-value health care. Yet SB 598 will derail the progress we have made in our health care system by lowering the value and safety that Californians should expect from their health care providers,” according to a fact sheet.
The fact-sheet defines “low-value care” as medical services for which there is little to no benefit and poses potential physical or financial harm to patients, such as unnecessary CT scans or MRIs for uncomplicated conditions.
California is one of about a dozen states that have introduced gold card legislation this year. If enacted, they would join five states with gold card laws: West Virginia, Texas, Vermont, Michigan, and Louisiana.
How do gold cards work?
Physicians who achieve a high approval rate of prior authorizations from insurers for 1 year are eligible to be exempted from obtaining prior authorizations the following year.
The approval rate is at least 90% for a certain number of eligible health services, but the number of prior authorizations required to qualify can range from 5 to 30, depending on the state law.
Gold card legislation typically also gives the treating physician the right to have an appeal of a prior authorization denial by a physician peer of the same or similar specialty.
California’s bill would also apply to all covered health services, which is broader than what United HealthCare has proposed for its gold card exemption. The bill would also require a plan or insurer to annually monitor rates of prior authorization approval, modification, appeal, and denial, and to discontinue services, items, and supplies that are approved 95% of the time.
“These are important reforms that will help ensure that patients can receive the care they need, when they need it,” said CMA president Donaldo Hernandez, MD.
However, it’s not clear how many physicians will meet “gold card” status based on Texas’ recent experience with its own “gold card” law.
The Texas Department of Insurance estimated that only 3.3% of licensed physicians in the state have met “gold card” status since the bill became law in 2021, said Zeke Silva, MD, an interventional radiologist who serves on the Council of Legislation for the Texas Medical Association.
He noted that the legislation has had a limited effect for several reasons. Commercial health plans only make up only about 20% of all health plans in Texas. Also, the final regulations didn’t go into effect until last May and physicians are evaluated by health plans for “gold card” status every 6 months, said Dr. Silva.
In addition, physicians must have at least five prior authorizations approved for the same health service, which the law left up to the health plans to define, said Dr. Silva.
Now, the Texas Medical Association is lobbying for legislative improvements. “We want to reduce the number of eligible services that health plans require for prior authorizations and have more oversight of prior authorization denials by the Texas Department of Insurance and the Texas Medical Board,” said Dr. Silva.
He’s optimistic that if the bill becomes law, the number of physicians eligible for gold cards may increase.
Meanwhile, the AMA’s legislative expert, who declined to be identified because of organization policy, acknowledged the possibility that some prior authorization bills will die in state legislatures this year.
“We remain hopeful, but it’s an uphill battle. The state medical associations face a lot of opposition from health plans who don’t want to see these reforms become law.”
A version of this article originally appeared on Medscape.com.
Amid growing criticism of health insurers’ onerous prior authorization practices, lawmakers in 30 states have introduced bills this year that aim to rein in insurer gatekeeping and improve patient care.
“This is something that goes on in every doctor’s office every day; the frustrations, the delays, and the use of office staff time are just unbelievable,” said Steven Orland, MD, a board-certified urologist and president of the Medical Society of New Jersey.
The bills, which cover private health plans and insurers that states regulate, may provide some relief for physicians as federal efforts to streamline prior authorization for some Medicare patients have lagged.
Last year, Congress failed to pass the Improving Seniors’ Timely Access to Care Act of 2021, despite 326 co-sponsors. The bill would have compelled insurers covering Medicare Advantage enrollees to speed up prior authorizations, make the process more transparent, and remove obstacles such as requiring fax machine submissions.
Last month, however, the Centers for Medicare & Medicaid Services issued a final rule that will improve some aspects of prior authorizations in Medicare Advantage insurance plans and ensure that enrollees have the same access to necessary care as traditional Medicare enrollees.
The insurance industry has long defended prior authorization requirements and opposed legislation that would limit them.
America’s Health Insurance Plans (AHIP) and the Blue Cross Blue Shield Association said in a 2019 letter to a congressional committee when the federal legislation was first introduced, “Prior authorizations enforce best practices and guidelines for care management and help physicians identify and avoid care techniques that would harm patient outcomes, such as designating prescriptions that could feed into an opioid addiction.” AHIP didn’t respond to repeated requests for comment.
But some major insurers now appear willing to compromise and voluntarily reduce the volume of prior authorizations they require. Days before the federal final rule was released, three major insurers – United HealthCare, Cigna, and Aetna CVS Health – announced they plan to drop some prior authorization requirements and automate processes.
United HealthCare said it will eliminate almost 20% of its prior authorizations for some nonurgent surgeries and procedures starting this summer. It also will create a national Gold Card program in 2024 for physicians who meet its eligibility requirements, which would eliminate prior authorization requirements for most procedures. Both initiatives will apply to commercial, Medicare Advantage, and Medicaid businesses, said the insurer in a statement.
However, United HealthCare also announced that in June it will start requiring prior authorization for diagnostic (not screening) gastrointestinal endoscopies for its nearly 27 million privately insured patients, citing data it says shows potentially harmful overuse of scopes. Physician groups have publicly criticized the move, saying it could delay lifesaving treatment, and have asked the insurer to reconsider.
Cigna and Aetna also have moved to pare back prior authorization processes. Scott Josephs, national medical officer for Cigna, told Healthcare Dive that Cigna has removed prior authorization reviews from nearly 500 services since 2020.
An Aetna spokesperson told Healthcare Dive that the CVS-owned payer has implemented a gold card program and rolled back prior authorization requirements on cataract surgeries, video EEGs, and home infusion for some drugs, according to Healthcare Dive.
Cigna has faced increased scrutiny from some state regulators since a ProPublica/The Capitol Forum article revealed in March that its doctors were denying claims without opening patients’ files, contrary to what insurance laws and regulations require in many states.
Over a period of 2 months last year, Cigna doctors denied over 300,000 requests for payments using this method, spending an average of 1.2 seconds on each case, the investigation found. In a written response, Cigna said the reporting by ProPublica and The Capitol Forum was “biased and incomplete.”
States aim to reduce prior authorization volume
The American Medical Association said it has been tracking nearly 90 prior authorization reform bills in 30 states. More than a dozen bills are still being considered in this legislative session, including in Arkansas, California, New Jersey, North Carolina, Maryland, and Washington, D.C.
“The groundswell of activity in the states reflects how big a problem this is,” said an AMA legislative expert. “The issue used to be ‘how can we automate and streamline processes’; now the issue is focused on reducing the volume of prior authorizations and the harm that can cause patients.”
The state bills use different strategies to reduce excessive prior authorization requirements. Maryland’s proposed bill, for example, would require just one prior authorization to stay on a prescription drug, if the insurer has previously approved the drug and the patient continues to successfully be treated by the drug.
Washington, D.C. and New Jersey have introduced comprehensive reform bills that include a “grace period” of 60 days, to ensure continuity of care when a patient switches health plans. They also would eliminate repeat authorizations for chronic and long-term conditions, set explicit timelines for insurers to respond to prior authorization requests and appeals, and require that practicing physicians review denials that are appealed.
Many state bills also would require insurers to be more transparent by posting information on their websites about which services and drugs require prior authorization and what their approval rates are for them, said AMA’s legislative expert.
“There’s a black hole of information that insurers have access to. We would really like to know how many prior authorization requests are denied, the time it takes to deny them, and the reasons for denial,” said Josh Bengal, JD, the director of government relations for the Medical Society of New Jersey.
The legislation in New Jersey and other states faces stiff opposition from the insurance lobby, especially state associations of health plans affiliated with AHIP. The California Association of Health Plans, for example, opposes a “gold card” bill (SB 598), introduced in February, that would allow a select group of high-performing doctors to skip prior authorizations for 1 year.
The CAHP states, “Californians deserve safe, high quality, high-value health care. Yet SB 598 will derail the progress we have made in our health care system by lowering the value and safety that Californians should expect from their health care providers,” according to a fact sheet.
The fact-sheet defines “low-value care” as medical services for which there is little to no benefit and poses potential physical or financial harm to patients, such as unnecessary CT scans or MRIs for uncomplicated conditions.
California is one of about a dozen states that have introduced gold card legislation this year. If enacted, they would join five states with gold card laws: West Virginia, Texas, Vermont, Michigan, and Louisiana.
How do gold cards work?
Physicians who achieve a high approval rate of prior authorizations from insurers for 1 year are eligible to be exempted from obtaining prior authorizations the following year.
The approval rate is at least 90% for a certain number of eligible health services, but the number of prior authorizations required to qualify can range from 5 to 30, depending on the state law.
Gold card legislation typically also gives the treating physician the right to have an appeal of a prior authorization denial by a physician peer of the same or similar specialty.
California’s bill would also apply to all covered health services, which is broader than what United HealthCare has proposed for its gold card exemption. The bill would also require a plan or insurer to annually monitor rates of prior authorization approval, modification, appeal, and denial, and to discontinue services, items, and supplies that are approved 95% of the time.
“These are important reforms that will help ensure that patients can receive the care they need, when they need it,” said CMA president Donaldo Hernandez, MD.
However, it’s not clear how many physicians will meet “gold card” status based on Texas’ recent experience with its own “gold card” law.
The Texas Department of Insurance estimated that only 3.3% of licensed physicians in the state have met “gold card” status since the bill became law in 2021, said Zeke Silva, MD, an interventional radiologist who serves on the Council of Legislation for the Texas Medical Association.
He noted that the legislation has had a limited effect for several reasons. Commercial health plans only make up only about 20% of all health plans in Texas. Also, the final regulations didn’t go into effect until last May and physicians are evaluated by health plans for “gold card” status every 6 months, said Dr. Silva.
In addition, physicians must have at least five prior authorizations approved for the same health service, which the law left up to the health plans to define, said Dr. Silva.
Now, the Texas Medical Association is lobbying for legislative improvements. “We want to reduce the number of eligible services that health plans require for prior authorizations and have more oversight of prior authorization denials by the Texas Department of Insurance and the Texas Medical Board,” said Dr. Silva.
He’s optimistic that if the bill becomes law, the number of physicians eligible for gold cards may increase.
Meanwhile, the AMA’s legislative expert, who declined to be identified because of organization policy, acknowledged the possibility that some prior authorization bills will die in state legislatures this year.
“We remain hopeful, but it’s an uphill battle. The state medical associations face a lot of opposition from health plans who don’t want to see these reforms become law.”
A version of this article originally appeared on Medscape.com.
Amid growing criticism of health insurers’ onerous prior authorization practices, lawmakers in 30 states have introduced bills this year that aim to rein in insurer gatekeeping and improve patient care.
“This is something that goes on in every doctor’s office every day; the frustrations, the delays, and the use of office staff time are just unbelievable,” said Steven Orland, MD, a board-certified urologist and president of the Medical Society of New Jersey.
The bills, which cover private health plans and insurers that states regulate, may provide some relief for physicians as federal efforts to streamline prior authorization for some Medicare patients have lagged.
Last year, Congress failed to pass the Improving Seniors’ Timely Access to Care Act of 2021, despite 326 co-sponsors. The bill would have compelled insurers covering Medicare Advantage enrollees to speed up prior authorizations, make the process more transparent, and remove obstacles such as requiring fax machine submissions.
Last month, however, the Centers for Medicare & Medicaid Services issued a final rule that will improve some aspects of prior authorizations in Medicare Advantage insurance plans and ensure that enrollees have the same access to necessary care as traditional Medicare enrollees.
The insurance industry has long defended prior authorization requirements and opposed legislation that would limit them.
America’s Health Insurance Plans (AHIP) and the Blue Cross Blue Shield Association said in a 2019 letter to a congressional committee when the federal legislation was first introduced, “Prior authorizations enforce best practices and guidelines for care management and help physicians identify and avoid care techniques that would harm patient outcomes, such as designating prescriptions that could feed into an opioid addiction.” AHIP didn’t respond to repeated requests for comment.
But some major insurers now appear willing to compromise and voluntarily reduce the volume of prior authorizations they require. Days before the federal final rule was released, three major insurers – United HealthCare, Cigna, and Aetna CVS Health – announced they plan to drop some prior authorization requirements and automate processes.
United HealthCare said it will eliminate almost 20% of its prior authorizations for some nonurgent surgeries and procedures starting this summer. It also will create a national Gold Card program in 2024 for physicians who meet its eligibility requirements, which would eliminate prior authorization requirements for most procedures. Both initiatives will apply to commercial, Medicare Advantage, and Medicaid businesses, said the insurer in a statement.
However, United HealthCare also announced that in June it will start requiring prior authorization for diagnostic (not screening) gastrointestinal endoscopies for its nearly 27 million privately insured patients, citing data it says shows potentially harmful overuse of scopes. Physician groups have publicly criticized the move, saying it could delay lifesaving treatment, and have asked the insurer to reconsider.
Cigna and Aetna also have moved to pare back prior authorization processes. Scott Josephs, national medical officer for Cigna, told Healthcare Dive that Cigna has removed prior authorization reviews from nearly 500 services since 2020.
An Aetna spokesperson told Healthcare Dive that the CVS-owned payer has implemented a gold card program and rolled back prior authorization requirements on cataract surgeries, video EEGs, and home infusion for some drugs, according to Healthcare Dive.
Cigna has faced increased scrutiny from some state regulators since a ProPublica/The Capitol Forum article revealed in March that its doctors were denying claims without opening patients’ files, contrary to what insurance laws and regulations require in many states.
Over a period of 2 months last year, Cigna doctors denied over 300,000 requests for payments using this method, spending an average of 1.2 seconds on each case, the investigation found. In a written response, Cigna said the reporting by ProPublica and The Capitol Forum was “biased and incomplete.”
States aim to reduce prior authorization volume
The American Medical Association said it has been tracking nearly 90 prior authorization reform bills in 30 states. More than a dozen bills are still being considered in this legislative session, including in Arkansas, California, New Jersey, North Carolina, Maryland, and Washington, D.C.
“The groundswell of activity in the states reflects how big a problem this is,” said an AMA legislative expert. “The issue used to be ‘how can we automate and streamline processes’; now the issue is focused on reducing the volume of prior authorizations and the harm that can cause patients.”
The state bills use different strategies to reduce excessive prior authorization requirements. Maryland’s proposed bill, for example, would require just one prior authorization to stay on a prescription drug, if the insurer has previously approved the drug and the patient continues to successfully be treated by the drug.
Washington, D.C. and New Jersey have introduced comprehensive reform bills that include a “grace period” of 60 days, to ensure continuity of care when a patient switches health plans. They also would eliminate repeat authorizations for chronic and long-term conditions, set explicit timelines for insurers to respond to prior authorization requests and appeals, and require that practicing physicians review denials that are appealed.
Many state bills also would require insurers to be more transparent by posting information on their websites about which services and drugs require prior authorization and what their approval rates are for them, said AMA’s legislative expert.
“There’s a black hole of information that insurers have access to. We would really like to know how many prior authorization requests are denied, the time it takes to deny them, and the reasons for denial,” said Josh Bengal, JD, the director of government relations for the Medical Society of New Jersey.
The legislation in New Jersey and other states faces stiff opposition from the insurance lobby, especially state associations of health plans affiliated with AHIP. The California Association of Health Plans, for example, opposes a “gold card” bill (SB 598), introduced in February, that would allow a select group of high-performing doctors to skip prior authorizations for 1 year.
The CAHP states, “Californians deserve safe, high quality, high-value health care. Yet SB 598 will derail the progress we have made in our health care system by lowering the value and safety that Californians should expect from their health care providers,” according to a fact sheet.
The fact-sheet defines “low-value care” as medical services for which there is little to no benefit and poses potential physical or financial harm to patients, such as unnecessary CT scans or MRIs for uncomplicated conditions.
California is one of about a dozen states that have introduced gold card legislation this year. If enacted, they would join five states with gold card laws: West Virginia, Texas, Vermont, Michigan, and Louisiana.
How do gold cards work?
Physicians who achieve a high approval rate of prior authorizations from insurers for 1 year are eligible to be exempted from obtaining prior authorizations the following year.
The approval rate is at least 90% for a certain number of eligible health services, but the number of prior authorizations required to qualify can range from 5 to 30, depending on the state law.
Gold card legislation typically also gives the treating physician the right to have an appeal of a prior authorization denial by a physician peer of the same or similar specialty.
California’s bill would also apply to all covered health services, which is broader than what United HealthCare has proposed for its gold card exemption. The bill would also require a plan or insurer to annually monitor rates of prior authorization approval, modification, appeal, and denial, and to discontinue services, items, and supplies that are approved 95% of the time.
“These are important reforms that will help ensure that patients can receive the care they need, when they need it,” said CMA president Donaldo Hernandez, MD.
However, it’s not clear how many physicians will meet “gold card” status based on Texas’ recent experience with its own “gold card” law.
The Texas Department of Insurance estimated that only 3.3% of licensed physicians in the state have met “gold card” status since the bill became law in 2021, said Zeke Silva, MD, an interventional radiologist who serves on the Council of Legislation for the Texas Medical Association.
He noted that the legislation has had a limited effect for several reasons. Commercial health plans only make up only about 20% of all health plans in Texas. Also, the final regulations didn’t go into effect until last May and physicians are evaluated by health plans for “gold card” status every 6 months, said Dr. Silva.
In addition, physicians must have at least five prior authorizations approved for the same health service, which the law left up to the health plans to define, said Dr. Silva.
Now, the Texas Medical Association is lobbying for legislative improvements. “We want to reduce the number of eligible services that health plans require for prior authorizations and have more oversight of prior authorization denials by the Texas Department of Insurance and the Texas Medical Board,” said Dr. Silva.
He’s optimistic that if the bill becomes law, the number of physicians eligible for gold cards may increase.
Meanwhile, the AMA’s legislative expert, who declined to be identified because of organization policy, acknowledged the possibility that some prior authorization bills will die in state legislatures this year.
“We remain hopeful, but it’s an uphill battle. The state medical associations face a lot of opposition from health plans who don’t want to see these reforms become law.”
A version of this article originally appeared on Medscape.com.
Common fracture risk predictors often fail for women of any race
according to a study published in JAMA Internal Medicine.
One of the screenings, the U.S. Fracture Risk Assessment Tool (FRAX), proved relatively ineffective at identifying women who developed osteoporosis. The other screening, the Osteoporosis Self-Assessment Tool (OST), excelled at identifying osteoporosis for women in every racial and ethnic group, but also failed at identifying who was most likely to experience a fracture. Osteoporosis experts say that primary care physicians should test for the condition in anyone with any risk factor for it, even if a screening tool suggests doing so is unnecessary.
The United States Preventive Services Task Force (USPSTF) recommends routine testing of bone mineral density in women age 65 years and older to detect risk of developing osteoporosis, which in turn leads to an increased risk for fractures of the hip, spine, shoulder, or forearm. For women aged 50-64, whether bone mineral density accurately reflects who will develop osteoporosis is less clear. In this age range, the USPSTF recommends using either FRAX or OST rather than routine bone mineral density tests.
“I have the utmost respect for the United States Preventive Services Task Force, which lists both of these as valid screening tools for younger postmenopausal women. What I hope this study does is to inform the next iteration of the screening guidelines,” by maintaining the recommendation to use the OST while not keeping FRAX, said Carolyn J. Crandall, MD, MS, an internal medicine physician and health services researcher at University of California, Los Angeles, who helped conduct the research.
The U.S. version of FRAX requires identifying someone’s race, height, and weight, then answering whether they have different risk factors for a fracture such as a previous fracture, rheumatoid arthritis, or smoking. The result was thought to indicate a cumulative risk for major fracture over the next 10 years. Patients at significant risk should then undergo a bone density test.
The tool can also incorporate information about bone mineral density, if available, but the FRAX analyses in Dr. Crandall’s study did not include those data because the study aimed to test the measure’s predictive ability in the absence of a bone scan.
The OST includes only two variables – weight and age – to calculate risk for osteoporosis, and generally takes seconds to complete. It does not include race. As with FRAX, anyone deemed at significant risk for developing osteoporosis should undergo a bone density test.
“OST is really simple; that makes it very appealing,” Dr. Crandall said. “OST could probably be automatically calculated in the electronic medical record.”
Using data from the Women’s Health Initiative, Dr. Crandall and colleagues tracked more than 67,000 women aged 50-64 years for 10 years following enrollment in the study to see who experienced a fracture or developed osteoporosis over that decade. The investigators found that neither FRAX nor OST was particularly good at predicting who went on to experience a fracture.
The accuracy of FRAX at fracture prediction peaked at 65% for Asian women (area under the receiver operating curve, 0.65; 95% confidence interval, 0.58-0.71), and was lowest for Black women (AUC 0.55; 95% CI, 0.52-0.59). OST also was most accurate for Asian women, but only up to 62% (AUC 0.62; 95% CI, 0.56-0.69), and was again lowest for Black women (AUC 0.53; 95% CI, 0.50 - 0.57)
“It is just very hard to predict fractures in this age group,” Dr. Crandall said, noting that more evidence exists about risk for fracture in people older than 65.
The story diverges with predicting risk of osteoporosis in the neck. The OST did this roughly 80% of the time, for all racial groups. That figure proved better than FRAX, without including race.
Treatment gap
“This evidence supports using OST instead of FRAX” for selecting younger postmenopausal women who should undergo a bone mineral density exam, said E. Michael Lewiecki, MD, director of the New Mexico Clinical Research & Osteoporosis Center in Albuquerque.

Dr. Lewiecki, who was not involved in the new study, noted that the U.S. version of FRAX specifies race because of some clinical evidence that different races have different rates of fracture. But he and Dr. Crandall said the validity of race-based algorithms to guide clinical care is a controversial and evolving topic in medicine. Dr. Lewiecki said the Canadian version of FRAX, which is similarly applied to a diverse population as in the United States, omits race and works as well as the U.S. version. Future iterations of the instrument in the United States may not include race, Dr. Lewiecki said.
“The study is perfectly valid as far as it goes. But the big gorilla in the room is that most patients who need a bone density test are not getting it,” Dr. Lewiecki added. Sometimes a patient might break a bone in their wrist, for example, and tell their primary care provider that anyone would have broken that bone because the fall was so hard. Even if that’s true, Dr. Lewiecki said, any woman older than 45 who has broken a bone should undergo a bone density test to determine if they have osteoporosis, even if it seems like there are other possible reasons for why the break occurred.
“Most of the clinical practice guidelines that are used by physicians recommend getting a bone density test in postmenopausal women under the age of 65 who have a risk factor for fracture,” Dr. Lewiecki said, with a primary risk factor being a prior fracture. Dr. Lewiecki said he would rather that anyone who could benefit from a bone density test receive it, rather than someone foregoing a scan based on a screening tool that may be flawed.
“Most patients – men and women – who have osteoporosis are currently not being identified. Even when they are being identified, they are commonly not being treated. And when they are started on treatment, many patients discontinue treatment before they’ve taken it long enough to benefit,” Dr. Lewiecki said.
Dr. Crandall and Dr. Lewiecki report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to a study published in JAMA Internal Medicine.
One of the screenings, the U.S. Fracture Risk Assessment Tool (FRAX), proved relatively ineffective at identifying women who developed osteoporosis. The other screening, the Osteoporosis Self-Assessment Tool (OST), excelled at identifying osteoporosis for women in every racial and ethnic group, but also failed at identifying who was most likely to experience a fracture. Osteoporosis experts say that primary care physicians should test for the condition in anyone with any risk factor for it, even if a screening tool suggests doing so is unnecessary.
The United States Preventive Services Task Force (USPSTF) recommends routine testing of bone mineral density in women age 65 years and older to detect risk of developing osteoporosis, which in turn leads to an increased risk for fractures of the hip, spine, shoulder, or forearm. For women aged 50-64, whether bone mineral density accurately reflects who will develop osteoporosis is less clear. In this age range, the USPSTF recommends using either FRAX or OST rather than routine bone mineral density tests.
“I have the utmost respect for the United States Preventive Services Task Force, which lists both of these as valid screening tools for younger postmenopausal women. What I hope this study does is to inform the next iteration of the screening guidelines,” by maintaining the recommendation to use the OST while not keeping FRAX, said Carolyn J. Crandall, MD, MS, an internal medicine physician and health services researcher at University of California, Los Angeles, who helped conduct the research.
The U.S. version of FRAX requires identifying someone’s race, height, and weight, then answering whether they have different risk factors for a fracture such as a previous fracture, rheumatoid arthritis, or smoking. The result was thought to indicate a cumulative risk for major fracture over the next 10 years. Patients at significant risk should then undergo a bone density test.
The tool can also incorporate information about bone mineral density, if available, but the FRAX analyses in Dr. Crandall’s study did not include those data because the study aimed to test the measure’s predictive ability in the absence of a bone scan.
The OST includes only two variables – weight and age – to calculate risk for osteoporosis, and generally takes seconds to complete. It does not include race. As with FRAX, anyone deemed at significant risk for developing osteoporosis should undergo a bone density test.
“OST is really simple; that makes it very appealing,” Dr. Crandall said. “OST could probably be automatically calculated in the electronic medical record.”
Using data from the Women’s Health Initiative, Dr. Crandall and colleagues tracked more than 67,000 women aged 50-64 years for 10 years following enrollment in the study to see who experienced a fracture or developed osteoporosis over that decade. The investigators found that neither FRAX nor OST was particularly good at predicting who went on to experience a fracture.
The accuracy of FRAX at fracture prediction peaked at 65% for Asian women (area under the receiver operating curve, 0.65; 95% confidence interval, 0.58-0.71), and was lowest for Black women (AUC 0.55; 95% CI, 0.52-0.59). OST also was most accurate for Asian women, but only up to 62% (AUC 0.62; 95% CI, 0.56-0.69), and was again lowest for Black women (AUC 0.53; 95% CI, 0.50 - 0.57)
“It is just very hard to predict fractures in this age group,” Dr. Crandall said, noting that more evidence exists about risk for fracture in people older than 65.
The story diverges with predicting risk of osteoporosis in the neck. The OST did this roughly 80% of the time, for all racial groups. That figure proved better than FRAX, without including race.
Treatment gap
“This evidence supports using OST instead of FRAX” for selecting younger postmenopausal women who should undergo a bone mineral density exam, said E. Michael Lewiecki, MD, director of the New Mexico Clinical Research & Osteoporosis Center in Albuquerque.

Dr. Lewiecki, who was not involved in the new study, noted that the U.S. version of FRAX specifies race because of some clinical evidence that different races have different rates of fracture. But he and Dr. Crandall said the validity of race-based algorithms to guide clinical care is a controversial and evolving topic in medicine. Dr. Lewiecki said the Canadian version of FRAX, which is similarly applied to a diverse population as in the United States, omits race and works as well as the U.S. version. Future iterations of the instrument in the United States may not include race, Dr. Lewiecki said.
“The study is perfectly valid as far as it goes. But the big gorilla in the room is that most patients who need a bone density test are not getting it,” Dr. Lewiecki added. Sometimes a patient might break a bone in their wrist, for example, and tell their primary care provider that anyone would have broken that bone because the fall was so hard. Even if that’s true, Dr. Lewiecki said, any woman older than 45 who has broken a bone should undergo a bone density test to determine if they have osteoporosis, even if it seems like there are other possible reasons for why the break occurred.
“Most of the clinical practice guidelines that are used by physicians recommend getting a bone density test in postmenopausal women under the age of 65 who have a risk factor for fracture,” Dr. Lewiecki said, with a primary risk factor being a prior fracture. Dr. Lewiecki said he would rather that anyone who could benefit from a bone density test receive it, rather than someone foregoing a scan based on a screening tool that may be flawed.
“Most patients – men and women – who have osteoporosis are currently not being identified. Even when they are being identified, they are commonly not being treated. And when they are started on treatment, many patients discontinue treatment before they’ve taken it long enough to benefit,” Dr. Lewiecki said.
Dr. Crandall and Dr. Lewiecki report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to a study published in JAMA Internal Medicine.
One of the screenings, the U.S. Fracture Risk Assessment Tool (FRAX), proved relatively ineffective at identifying women who developed osteoporosis. The other screening, the Osteoporosis Self-Assessment Tool (OST), excelled at identifying osteoporosis for women in every racial and ethnic group, but also failed at identifying who was most likely to experience a fracture. Osteoporosis experts say that primary care physicians should test for the condition in anyone with any risk factor for it, even if a screening tool suggests doing so is unnecessary.
The United States Preventive Services Task Force (USPSTF) recommends routine testing of bone mineral density in women age 65 years and older to detect risk of developing osteoporosis, which in turn leads to an increased risk for fractures of the hip, spine, shoulder, or forearm. For women aged 50-64, whether bone mineral density accurately reflects who will develop osteoporosis is less clear. In this age range, the USPSTF recommends using either FRAX or OST rather than routine bone mineral density tests.
“I have the utmost respect for the United States Preventive Services Task Force, which lists both of these as valid screening tools for younger postmenopausal women. What I hope this study does is to inform the next iteration of the screening guidelines,” by maintaining the recommendation to use the OST while not keeping FRAX, said Carolyn J. Crandall, MD, MS, an internal medicine physician and health services researcher at University of California, Los Angeles, who helped conduct the research.
The U.S. version of FRAX requires identifying someone’s race, height, and weight, then answering whether they have different risk factors for a fracture such as a previous fracture, rheumatoid arthritis, or smoking. The result was thought to indicate a cumulative risk for major fracture over the next 10 years. Patients at significant risk should then undergo a bone density test.
The tool can also incorporate information about bone mineral density, if available, but the FRAX analyses in Dr. Crandall’s study did not include those data because the study aimed to test the measure’s predictive ability in the absence of a bone scan.
The OST includes only two variables – weight and age – to calculate risk for osteoporosis, and generally takes seconds to complete. It does not include race. As with FRAX, anyone deemed at significant risk for developing osteoporosis should undergo a bone density test.
“OST is really simple; that makes it very appealing,” Dr. Crandall said. “OST could probably be automatically calculated in the electronic medical record.”
Using data from the Women’s Health Initiative, Dr. Crandall and colleagues tracked more than 67,000 women aged 50-64 years for 10 years following enrollment in the study to see who experienced a fracture or developed osteoporosis over that decade. The investigators found that neither FRAX nor OST was particularly good at predicting who went on to experience a fracture.
The accuracy of FRAX at fracture prediction peaked at 65% for Asian women (area under the receiver operating curve, 0.65; 95% confidence interval, 0.58-0.71), and was lowest for Black women (AUC 0.55; 95% CI, 0.52-0.59). OST also was most accurate for Asian women, but only up to 62% (AUC 0.62; 95% CI, 0.56-0.69), and was again lowest for Black women (AUC 0.53; 95% CI, 0.50 - 0.57)
“It is just very hard to predict fractures in this age group,” Dr. Crandall said, noting that more evidence exists about risk for fracture in people older than 65.
The story diverges with predicting risk of osteoporosis in the neck. The OST did this roughly 80% of the time, for all racial groups. That figure proved better than FRAX, without including race.
Treatment gap
“This evidence supports using OST instead of FRAX” for selecting younger postmenopausal women who should undergo a bone mineral density exam, said E. Michael Lewiecki, MD, director of the New Mexico Clinical Research & Osteoporosis Center in Albuquerque.

Dr. Lewiecki, who was not involved in the new study, noted that the U.S. version of FRAX specifies race because of some clinical evidence that different races have different rates of fracture. But he and Dr. Crandall said the validity of race-based algorithms to guide clinical care is a controversial and evolving topic in medicine. Dr. Lewiecki said the Canadian version of FRAX, which is similarly applied to a diverse population as in the United States, omits race and works as well as the U.S. version. Future iterations of the instrument in the United States may not include race, Dr. Lewiecki said.
“The study is perfectly valid as far as it goes. But the big gorilla in the room is that most patients who need a bone density test are not getting it,” Dr. Lewiecki added. Sometimes a patient might break a bone in their wrist, for example, and tell their primary care provider that anyone would have broken that bone because the fall was so hard. Even if that’s true, Dr. Lewiecki said, any woman older than 45 who has broken a bone should undergo a bone density test to determine if they have osteoporosis, even if it seems like there are other possible reasons for why the break occurred.
“Most of the clinical practice guidelines that are used by physicians recommend getting a bone density test in postmenopausal women under the age of 65 who have a risk factor for fracture,” Dr. Lewiecki said, with a primary risk factor being a prior fracture. Dr. Lewiecki said he would rather that anyone who could benefit from a bone density test receive it, rather than someone foregoing a scan based on a screening tool that may be flawed.
“Most patients – men and women – who have osteoporosis are currently not being identified. Even when they are being identified, they are commonly not being treated. And when they are started on treatment, many patients discontinue treatment before they’ve taken it long enough to benefit,” Dr. Lewiecki said.
Dr. Crandall and Dr. Lewiecki report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA INTERNAL MEDICINE
The weird world of hydrogels: How they’ll change health care
Imagine a day when a simple injection prompts a broken bone to heal. When tiny, ingestible devices linger in the body, unnoticed, tracking our health or delivering life-saving medications. When brain and heart implants mesh with flesh so seamlessly that the body thinks they’ve been there all along.
These are the dreams of materials scientists who have toiled for decades to mimic the complex architecture of the human body in hopes of replacing broken parts or treating disease.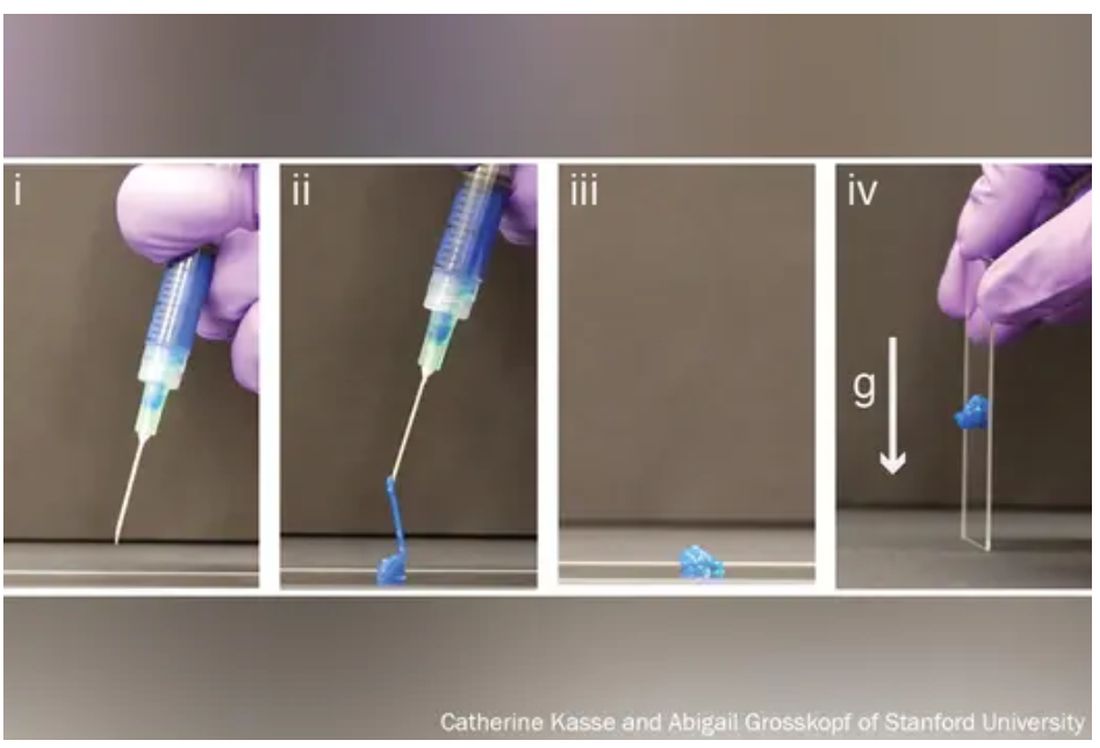
The problem, say bioengineers, is that most replacement and corrective parts – from prosthetics to pacemakers – are made of hard, dry, lifeless materials, like metal or plastic, while biological tissue is soft, wet, and living.
The body knows the difference and tends to reject imitations.
Enter hydrogels, three-dimensional networks of molecules swollen with – by definition – water.
First described in 1960 by creators of soft contact lenses, these weird, shape-shifting substances are able to morph from liquid to solid to a squishy in-between. (Early, simple uses include hair gel or Jell-O.). Slow to gain attention, growing to just 1,000 studies published by 1982, they’ve become the subject of intense study recently, with 100,000 papers published by 2020, and 3,800 already this year alone.
As chemists, biologists, and engineers begin to work more with one another and with medical doctors,
“We are, essentially, hydrogels,” said Benjamin Wiley, PhD, a chemistry professor at Duke University in Durham, N.C. “As people develop new hydrogels that more closely match the tissues in our body, we’ll be able to treat a whole host of ailments we couldn’t treat before.”
From contact lenses to brain implants
Put simply, a hydrogel is like a mesh bag of water.
The mesh is made of polymers, or spaghetti-like strands of molecules, stitched together in a repeating pattern and swollen with H2O, much like the way 3D matrixes in our body surround, support, and give structure to our cells and tissues.
“Imagine a soccer net, with all of these long fibers woven together to create the net,” said Eric Appel, PhD, associate professor of materials science and engineering at Stanford (Calif.) University.
While the broader category of “gels” could be filled with anything, including chemical solvents, water is the key ingredient that sets hydrogels apart, making them ideal for, as some scientists put it, “merging humans and machines.”
Human bones are about 25% water, while muscles hover around 70% and the brain is 85%. The precious liquid plays a host of critical roles, from shuttling nutrients in and waste out to helping cells talk to each other.
Lab-made hydrogels can be loaded with cargo (like a ball in the net), including cells or drugs that help mimic some of those functions.
Hydrogels are soft and pliable like flesh. So, if used in implants, they may be less likely to damage surrounding tissue.
“Think about a metal spoon in your bowl of pudding. As you’re shaking the bowl, the spoon doesn’t stay in place, and you get scarring around the spoon,” said Christina Tringides, PhD, a materials scientist who studies neural engineering. That, she says, is exactly what happens to brain implants when patients breathe or move. “It’s a mechanical mismatch. But with hydrogels, you could get perfect mechanical matching.”
Hydrogels also tend to be nontoxic, so the immune system may be less likely to attack them as foreign bodies.
All this has made hydrogels the new darling of the bioengineering world.
“There has been an absolute explosion of interest in these materials,” Dr. Appel said.
Smarter drug delivery and ingestible electronics
Early versions of hydrogels were thick and gooey, making it hard to get them inside the body.
“Think of a block of Jell-O. You couldn’t inject something like that,” Dr. Appel said.
But Dr. Appel, whose lab develops new drug delivery systems, has been tinkering with gel formulas for years in hopes that these high-tech globs could someday ferry timed-release drugs to just the right spot in the body.
His new hydrogels start as fully formed gels (which help preserve the drug contents) inside a syringe. But once the plunger is pushed, they magically shape-shift to a liquid thin enough to flow easily through a standard needle. Upon exit, they immediately reform into gels, protecting the inherent cargo from degrading.
This could be a game changer at a time when many cutting-edge drugs – think Humira for arthritis or Ozempic for type 2 diabetes – are made of quickly degrading proteins too large and complex to simply jam into a pill. Instead, they must be injected, often frequently.
“Because the gel takes months to dissolve, it slowly delivers the drug over time,” Dr. Appel said. “You could conceivably go from a shot once a week to once every 4 months.”
Such slow-release hydrogels could make vaccines last longer, in turn teaching the body to better resist emerging virus variants, and deliver tumor-busting therapies more precisely, said Dr. Appel, who has formed a startup and hopes to fast-track the first hydrogel drug delivery system to clinical trials within a few years.
Meanwhile, another team at the Massachusetts Institute of Technology has taken a different approach, developing a standard-sized ingestible hydrogel pill that swells up like a puffer fish in the stomach, lasting a month and slowly releasing drugs all the while. To remove the pill, a patient simply drinks a salt-based solution that shrivels the ping-pong ball–sized device so it can be passed out of the body.
In a paper in Nature Communications, the scientists showed the puffer fish pill could also be loaded with tiny cameras or monitors to track conditions like ulcers or cancer.
“The dream is to have a Jell-O-like smart pill that, once swallowed, stays in the stomach and monitors the patient’s health,” said Xuanhe Zhao, PhD, a researcher on the project and associate professor of mechanical engineering at MIT.
Building joints and regrowing bones
Since the 1970s, researchers have mulled using hydrogels to replace human cartilage, a remarkably strong and flexible tissue made of about 90% water but able to withstand the weight of a car on an area about the size of a coin.
Until recently, those efforts have largely failed. Meaning when knee cartilage wears down, things like cartilage transplants, drilling holes to stimulate new growth, or total joint replacements – all of which require lengthy rehab – are the only options.
But that may be about to change.
Dr. Wiley and his colleagues at Duke recently reported that they’d developed the first gel-based cartilage substitute even stronger and more durable than the real thing.
By attaching their hydrogel to a titanium backing to help stick it in place, they hope to repair damaged cartilage “much like a dentist fills a cavity” long before surgery is necessary.
They too have partnered with industry to bring their hydrogel to market – starting with knees.
“Ultimately, the goal is to do any joint – hips, ankles, fingers, and toes,” Dr. Wiley said.
At the University of Toronto, chemist Karina Carneiro, PhD, and dentist Christopher McCulloch, DDS, are also thinking big.
In a recent paper in Proceedings of the National Academy of Sciences, they describe a hydrogel, designed by Dr. Carneiro and made of DNA, that can be injected, migrate to a defect in bone – an irreparable break, hole from surgery, or jawbone withered by age – and fill in the gap like putty. But not only does it patch the hole, it prompts the bone to regenerate.
In rats with holes in their skulls due to surgery, they found that the treatment did not work as well as the existing gold standard for repairing holes in bone – grafting bone from elsewhere in the body. But it did work.
“These are very early days for DNA hydrogels,” cautioned Dr. McCulloch, a study coauthor and professor in the Faculty of Dentistry, noting that it will likely be a decade or more before such technology could be available to patients. “But there is the potential that DNA hydrogel could someday grow bone without having to have highly invasive surgical procedures. That’s a significant advancement.”
A sci-fi future
Perhaps the wildest, and weirdest, potential applications of hydrogels come in the realm of human-machine interaction.
Numerous companies are already dabbling in neural prosthetic or brain computer interfaces that might someday, for instance, let someone who is paralyzed and can’t speak write on a laptop using their thoughts.
The spoon-in-the-Jell-O problem has been a major stumbling block.
But Dr. Tringides, who recently earned her PhD in biophysics from Harvard, is working on it.
She and her team have developed a seaweed-based hydrogel loaded with tiny flecks of nanomaterials that can not only meld nicely into squishy brain tissue but also conduct electricity.
Within a decade, she says, this could replace the clunky platinum metal discs used for electrocorticography – recording electrical activity in the brain to identify where seizures start or doing precise brain surgery.
In 30 to 50 years? Let your imagination run wild.
“I’m a skeptic. I like to take research step by step,” she said. “But things are definitely progressing in an interesting direction.”
A version of this article first appeared on WebMD.com.
Imagine a day when a simple injection prompts a broken bone to heal. When tiny, ingestible devices linger in the body, unnoticed, tracking our health or delivering life-saving medications. When brain and heart implants mesh with flesh so seamlessly that the body thinks they’ve been there all along.
These are the dreams of materials scientists who have toiled for decades to mimic the complex architecture of the human body in hopes of replacing broken parts or treating disease.
The problem, say bioengineers, is that most replacement and corrective parts – from prosthetics to pacemakers – are made of hard, dry, lifeless materials, like metal or plastic, while biological tissue is soft, wet, and living.
The body knows the difference and tends to reject imitations.
Enter hydrogels, three-dimensional networks of molecules swollen with – by definition – water.
First described in 1960 by creators of soft contact lenses, these weird, shape-shifting substances are able to morph from liquid to solid to a squishy in-between. (Early, simple uses include hair gel or Jell-O.). Slow to gain attention, growing to just 1,000 studies published by 1982, they’ve become the subject of intense study recently, with 100,000 papers published by 2020, and 3,800 already this year alone.
As chemists, biologists, and engineers begin to work more with one another and with medical doctors,
“We are, essentially, hydrogels,” said Benjamin Wiley, PhD, a chemistry professor at Duke University in Durham, N.C. “As people develop new hydrogels that more closely match the tissues in our body, we’ll be able to treat a whole host of ailments we couldn’t treat before.”
From contact lenses to brain implants
Put simply, a hydrogel is like a mesh bag of water.
The mesh is made of polymers, or spaghetti-like strands of molecules, stitched together in a repeating pattern and swollen with H2O, much like the way 3D matrixes in our body surround, support, and give structure to our cells and tissues.
“Imagine a soccer net, with all of these long fibers woven together to create the net,” said Eric Appel, PhD, associate professor of materials science and engineering at Stanford (Calif.) University.
While the broader category of “gels” could be filled with anything, including chemical solvents, water is the key ingredient that sets hydrogels apart, making them ideal for, as some scientists put it, “merging humans and machines.”
Human bones are about 25% water, while muscles hover around 70% and the brain is 85%. The precious liquid plays a host of critical roles, from shuttling nutrients in and waste out to helping cells talk to each other.
Lab-made hydrogels can be loaded with cargo (like a ball in the net), including cells or drugs that help mimic some of those functions.
Hydrogels are soft and pliable like flesh. So, if used in implants, they may be less likely to damage surrounding tissue.
“Think about a metal spoon in your bowl of pudding. As you’re shaking the bowl, the spoon doesn’t stay in place, and you get scarring around the spoon,” said Christina Tringides, PhD, a materials scientist who studies neural engineering. That, she says, is exactly what happens to brain implants when patients breathe or move. “It’s a mechanical mismatch. But with hydrogels, you could get perfect mechanical matching.”
Hydrogels also tend to be nontoxic, so the immune system may be less likely to attack them as foreign bodies.
All this has made hydrogels the new darling of the bioengineering world.
“There has been an absolute explosion of interest in these materials,” Dr. Appel said.
Smarter drug delivery and ingestible electronics
Early versions of hydrogels were thick and gooey, making it hard to get them inside the body.
“Think of a block of Jell-O. You couldn’t inject something like that,” Dr. Appel said.
But Dr. Appel, whose lab develops new drug delivery systems, has been tinkering with gel formulas for years in hopes that these high-tech globs could someday ferry timed-release drugs to just the right spot in the body.
His new hydrogels start as fully formed gels (which help preserve the drug contents) inside a syringe. But once the plunger is pushed, they magically shape-shift to a liquid thin enough to flow easily through a standard needle. Upon exit, they immediately reform into gels, protecting the inherent cargo from degrading.
This could be a game changer at a time when many cutting-edge drugs – think Humira for arthritis or Ozempic for type 2 diabetes – are made of quickly degrading proteins too large and complex to simply jam into a pill. Instead, they must be injected, often frequently.
“Because the gel takes months to dissolve, it slowly delivers the drug over time,” Dr. Appel said. “You could conceivably go from a shot once a week to once every 4 months.”
Such slow-release hydrogels could make vaccines last longer, in turn teaching the body to better resist emerging virus variants, and deliver tumor-busting therapies more precisely, said Dr. Appel, who has formed a startup and hopes to fast-track the first hydrogel drug delivery system to clinical trials within a few years.
Meanwhile, another team at the Massachusetts Institute of Technology has taken a different approach, developing a standard-sized ingestible hydrogel pill that swells up like a puffer fish in the stomach, lasting a month and slowly releasing drugs all the while. To remove the pill, a patient simply drinks a salt-based solution that shrivels the ping-pong ball–sized device so it can be passed out of the body.
In a paper in Nature Communications, the scientists showed the puffer fish pill could also be loaded with tiny cameras or monitors to track conditions like ulcers or cancer.
“The dream is to have a Jell-O-like smart pill that, once swallowed, stays in the stomach and monitors the patient’s health,” said Xuanhe Zhao, PhD, a researcher on the project and associate professor of mechanical engineering at MIT.
Building joints and regrowing bones
Since the 1970s, researchers have mulled using hydrogels to replace human cartilage, a remarkably strong and flexible tissue made of about 90% water but able to withstand the weight of a car on an area about the size of a coin.
Until recently, those efforts have largely failed. Meaning when knee cartilage wears down, things like cartilage transplants, drilling holes to stimulate new growth, or total joint replacements – all of which require lengthy rehab – are the only options.
But that may be about to change.
Dr. Wiley and his colleagues at Duke recently reported that they’d developed the first gel-based cartilage substitute even stronger and more durable than the real thing.
By attaching their hydrogel to a titanium backing to help stick it in place, they hope to repair damaged cartilage “much like a dentist fills a cavity” long before surgery is necessary.
They too have partnered with industry to bring their hydrogel to market – starting with knees.
“Ultimately, the goal is to do any joint – hips, ankles, fingers, and toes,” Dr. Wiley said.
At the University of Toronto, chemist Karina Carneiro, PhD, and dentist Christopher McCulloch, DDS, are also thinking big.
In a recent paper in Proceedings of the National Academy of Sciences, they describe a hydrogel, designed by Dr. Carneiro and made of DNA, that can be injected, migrate to a defect in bone – an irreparable break, hole from surgery, or jawbone withered by age – and fill in the gap like putty. But not only does it patch the hole, it prompts the bone to regenerate.
In rats with holes in their skulls due to surgery, they found that the treatment did not work as well as the existing gold standard for repairing holes in bone – grafting bone from elsewhere in the body. But it did work.
“These are very early days for DNA hydrogels,” cautioned Dr. McCulloch, a study coauthor and professor in the Faculty of Dentistry, noting that it will likely be a decade or more before such technology could be available to patients. “But there is the potential that DNA hydrogel could someday grow bone without having to have highly invasive surgical procedures. That’s a significant advancement.”
A sci-fi future
Perhaps the wildest, and weirdest, potential applications of hydrogels come in the realm of human-machine interaction.
Numerous companies are already dabbling in neural prosthetic or brain computer interfaces that might someday, for instance, let someone who is paralyzed and can’t speak write on a laptop using their thoughts.
The spoon-in-the-Jell-O problem has been a major stumbling block.
But Dr. Tringides, who recently earned her PhD in biophysics from Harvard, is working on it.
She and her team have developed a seaweed-based hydrogel loaded with tiny flecks of nanomaterials that can not only meld nicely into squishy brain tissue but also conduct electricity.
Within a decade, she says, this could replace the clunky platinum metal discs used for electrocorticography – recording electrical activity in the brain to identify where seizures start or doing precise brain surgery.
In 30 to 50 years? Let your imagination run wild.
“I’m a skeptic. I like to take research step by step,” she said. “But things are definitely progressing in an interesting direction.”
A version of this article first appeared on WebMD.com.
Imagine a day when a simple injection prompts a broken bone to heal. When tiny, ingestible devices linger in the body, unnoticed, tracking our health or delivering life-saving medications. When brain and heart implants mesh with flesh so seamlessly that the body thinks they’ve been there all along.
These are the dreams of materials scientists who have toiled for decades to mimic the complex architecture of the human body in hopes of replacing broken parts or treating disease.
The problem, say bioengineers, is that most replacement and corrective parts – from prosthetics to pacemakers – are made of hard, dry, lifeless materials, like metal or plastic, while biological tissue is soft, wet, and living.
The body knows the difference and tends to reject imitations.
Enter hydrogels, three-dimensional networks of molecules swollen with – by definition – water.
First described in 1960 by creators of soft contact lenses, these weird, shape-shifting substances are able to morph from liquid to solid to a squishy in-between. (Early, simple uses include hair gel or Jell-O.). Slow to gain attention, growing to just 1,000 studies published by 1982, they’ve become the subject of intense study recently, with 100,000 papers published by 2020, and 3,800 already this year alone.
As chemists, biologists, and engineers begin to work more with one another and with medical doctors,
“We are, essentially, hydrogels,” said Benjamin Wiley, PhD, a chemistry professor at Duke University in Durham, N.C. “As people develop new hydrogels that more closely match the tissues in our body, we’ll be able to treat a whole host of ailments we couldn’t treat before.”
From contact lenses to brain implants
Put simply, a hydrogel is like a mesh bag of water.
The mesh is made of polymers, or spaghetti-like strands of molecules, stitched together in a repeating pattern and swollen with H2O, much like the way 3D matrixes in our body surround, support, and give structure to our cells and tissues.
“Imagine a soccer net, with all of these long fibers woven together to create the net,” said Eric Appel, PhD, associate professor of materials science and engineering at Stanford (Calif.) University.
While the broader category of “gels” could be filled with anything, including chemical solvents, water is the key ingredient that sets hydrogels apart, making them ideal for, as some scientists put it, “merging humans and machines.”
Human bones are about 25% water, while muscles hover around 70% and the brain is 85%. The precious liquid plays a host of critical roles, from shuttling nutrients in and waste out to helping cells talk to each other.
Lab-made hydrogels can be loaded with cargo (like a ball in the net), including cells or drugs that help mimic some of those functions.
Hydrogels are soft and pliable like flesh. So, if used in implants, they may be less likely to damage surrounding tissue.
“Think about a metal spoon in your bowl of pudding. As you’re shaking the bowl, the spoon doesn’t stay in place, and you get scarring around the spoon,” said Christina Tringides, PhD, a materials scientist who studies neural engineering. That, she says, is exactly what happens to brain implants when patients breathe or move. “It’s a mechanical mismatch. But with hydrogels, you could get perfect mechanical matching.”
Hydrogels also tend to be nontoxic, so the immune system may be less likely to attack them as foreign bodies.
All this has made hydrogels the new darling of the bioengineering world.
“There has been an absolute explosion of interest in these materials,” Dr. Appel said.
Smarter drug delivery and ingestible electronics
Early versions of hydrogels were thick and gooey, making it hard to get them inside the body.
“Think of a block of Jell-O. You couldn’t inject something like that,” Dr. Appel said.
But Dr. Appel, whose lab develops new drug delivery systems, has been tinkering with gel formulas for years in hopes that these high-tech globs could someday ferry timed-release drugs to just the right spot in the body.
His new hydrogels start as fully formed gels (which help preserve the drug contents) inside a syringe. But once the plunger is pushed, they magically shape-shift to a liquid thin enough to flow easily through a standard needle. Upon exit, they immediately reform into gels, protecting the inherent cargo from degrading.
This could be a game changer at a time when many cutting-edge drugs – think Humira for arthritis or Ozempic for type 2 diabetes – are made of quickly degrading proteins too large and complex to simply jam into a pill. Instead, they must be injected, often frequently.
“Because the gel takes months to dissolve, it slowly delivers the drug over time,” Dr. Appel said. “You could conceivably go from a shot once a week to once every 4 months.”
Such slow-release hydrogels could make vaccines last longer, in turn teaching the body to better resist emerging virus variants, and deliver tumor-busting therapies more precisely, said Dr. Appel, who has formed a startup and hopes to fast-track the first hydrogel drug delivery system to clinical trials within a few years.
Meanwhile, another team at the Massachusetts Institute of Technology has taken a different approach, developing a standard-sized ingestible hydrogel pill that swells up like a puffer fish in the stomach, lasting a month and slowly releasing drugs all the while. To remove the pill, a patient simply drinks a salt-based solution that shrivels the ping-pong ball–sized device so it can be passed out of the body.
In a paper in Nature Communications, the scientists showed the puffer fish pill could also be loaded with tiny cameras or monitors to track conditions like ulcers or cancer.
“The dream is to have a Jell-O-like smart pill that, once swallowed, stays in the stomach and monitors the patient’s health,” said Xuanhe Zhao, PhD, a researcher on the project and associate professor of mechanical engineering at MIT.
Building joints and regrowing bones
Since the 1970s, researchers have mulled using hydrogels to replace human cartilage, a remarkably strong and flexible tissue made of about 90% water but able to withstand the weight of a car on an area about the size of a coin.
Until recently, those efforts have largely failed. Meaning when knee cartilage wears down, things like cartilage transplants, drilling holes to stimulate new growth, or total joint replacements – all of which require lengthy rehab – are the only options.
But that may be about to change.
Dr. Wiley and his colleagues at Duke recently reported that they’d developed the first gel-based cartilage substitute even stronger and more durable than the real thing.
By attaching their hydrogel to a titanium backing to help stick it in place, they hope to repair damaged cartilage “much like a dentist fills a cavity” long before surgery is necessary.
They too have partnered with industry to bring their hydrogel to market – starting with knees.
“Ultimately, the goal is to do any joint – hips, ankles, fingers, and toes,” Dr. Wiley said.
At the University of Toronto, chemist Karina Carneiro, PhD, and dentist Christopher McCulloch, DDS, are also thinking big.
In a recent paper in Proceedings of the National Academy of Sciences, they describe a hydrogel, designed by Dr. Carneiro and made of DNA, that can be injected, migrate to a defect in bone – an irreparable break, hole from surgery, or jawbone withered by age – and fill in the gap like putty. But not only does it patch the hole, it prompts the bone to regenerate.
In rats with holes in their skulls due to surgery, they found that the treatment did not work as well as the existing gold standard for repairing holes in bone – grafting bone from elsewhere in the body. But it did work.
“These are very early days for DNA hydrogels,” cautioned Dr. McCulloch, a study coauthor and professor in the Faculty of Dentistry, noting that it will likely be a decade or more before such technology could be available to patients. “But there is the potential that DNA hydrogel could someday grow bone without having to have highly invasive surgical procedures. That’s a significant advancement.”
A sci-fi future
Perhaps the wildest, and weirdest, potential applications of hydrogels come in the realm of human-machine interaction.
Numerous companies are already dabbling in neural prosthetic or brain computer interfaces that might someday, for instance, let someone who is paralyzed and can’t speak write on a laptop using their thoughts.
The spoon-in-the-Jell-O problem has been a major stumbling block.
But Dr. Tringides, who recently earned her PhD in biophysics from Harvard, is working on it.
She and her team have developed a seaweed-based hydrogel loaded with tiny flecks of nanomaterials that can not only meld nicely into squishy brain tissue but also conduct electricity.
Within a decade, she says, this could replace the clunky platinum metal discs used for electrocorticography – recording electrical activity in the brain to identify where seizures start or doing precise brain surgery.
In 30 to 50 years? Let your imagination run wild.
“I’m a skeptic. I like to take research step by step,” she said. “But things are definitely progressing in an interesting direction.”
A version of this article first appeared on WebMD.com.
Diabetes, cholesterol meds use drops after bariatric surgery
compared with patients with obesity who did not have such an operation. However, these declines didn’t extend to cardiovascular medication use.
“In this study, undergoing bariatric surgery was associated with a substantial and long-lasting reduction in the use of lipid-lowering and antidiabetic medications, compared with no surgery for obesity, while for cardiovascular medications this reduction was only transient,” the authors report in research published in JAMA Surgery.
“The results can aid in informed decision-making when considering bariatric surgery for patients with morbid obesity and inform patients and professionals about the expected long-term effects of medication use for obesity-related comorbidities,” they write.
The study “highlights the benefits of mandated databases that report metabolic bariatric surgery, obesity-related comorbidities, and medications,” writes Paulina Salminen, MD, in an accompanying editorial.
However, key limitations include a lack of weight data, which is important in light of previous studies showing that suboptimal weight loss after bariatric surgery is linked to a higher incidence of type 2 diabetes, dyslipidemia, and hypertension, note Dr. Salminen, of the department of digestive surgery, University Hospital, Turku, Finland, and colleagues.
Swedish, Finnish obesity data probed
When significant weight loss is achieved, bariatric surgery has been well documented to be associated with improvements in a variety of comorbidities, quality of life, and even life expectancy.
Key comorbidities shown to improve with the surgery include hyperlipidemia, cardiovascular disease, and type 2 diabetes.
However, data are lacking on the association between bariatric surgery and the use of medications for those conditions, particularly compared with people with obesity who don’t have bariatric surgery.
To investigate, first author Joonas H. Kauppila, MD, PhD, of Upper Gastrointestinal Surgery, Karolinska University Hospital, Stockholm, and colleagues conducted a population-based cohort study, evaluating data on 26,396 patients who underwent bariatric surgery with gastric bypass or sleeve gastrectomy in Sweden between 2005 and 2020 or Finland between 1995 and 2018.
Overall, 66.4% of patients were women and their median age was 50.
They were compared with five times as many matched controls with obesity who had not had bariatric surgery from the same population databases, representing a total of 131,980 patients who were matched based on age, country, sex, calendar year, and medication use.
In terms of lipid-lowering medication, rates of use after bariatric surgery decreased from 20.3% at baseline to 12.9% after 2 years and bounced back somewhat to 17.6% after 15 years. Comparatively, in the no surgery group, baseline lipid-lowering medication use of 21.0% increased to 44.6% after 15 years, more than twice the rate of usage in the bariatric surgery group in the same period.
Antidiabetic medications were used by 27.7% of patients in the bariatric surgery group at baseline, with a drop to 10.0% after 2 years, followed by an increase to 23.5% after 15 years. In the no surgery group, the rate of antidiabetic medication use steadily increased from 27.7% at baseline to 54.2% after 15 years, which again was nearly double the rate of antidiabetic medication use in the bariatric surgery group at 15 years.
Meanwhile, cardiovascular medications were used by 60.2% of patients receiving bariatric surgery at baseline, with the rate decreasing to 43.2% after 2 years but increasing to 74.6% after 15 years. Among the nonbariatric surgery patients, use of cardiovascular medications increased from 54.4% at baseline to 83.3% after 15 years.
Causes?
As for the cause of the lack of any decline in use of cardiovascular medications versus other medications in the surgery patients, the authors speculate that the effect “may be related to aging and regain of weight over time after bariatric surgery, a phenomenon caused by hormonal, dietary, physical, and behavioral factors.”
“In contrast, as expected, a gradual increase in the use of all three medication groups was observed over time among the patients treated with no surgery for obesity,” they note.
The lower medication use with bariatric surgery can also translate to economic benefits, the authors add.
“Economically, the long-lasting reductions in medication use for hyperlipidemia, cardiovascular morbidity, and diabetes suggest that surgical treatment of morbid obesity may infer savings in medication expenses for patients, health care, and society,” they report.
“Future research may focus on subgroups that are most likely to benefit from bariatric surgery, including resolution and severity of comorbidities,” they continue.
In their editorial, Dr. Salminen and colleagues note that previous research has shown remission of dyslipidemia in up to 70% of patients after bariatric surgery that was independent of weight loss, which appears to support the sustained reduction in lipid-lowering medications following surgery observed in the current study, suggesting some benefits on lipids beyond weight loss.
Other limitations, however, include that the bariatric surgery group in the study was older and had more comorbidities than those in previous bariatric surgery studies.
“Future studies should assess this in a younger cohort with less disease at baseline and differentiation within cardiovascular disease regarding at least hypertension, ischemic heart disease, and heart failure,” the authors conclude.
The authors have reported no relevant financial relationships. Dr. Salminen has reported receiving grants from the Sigrid Jusélius Foundation, Academy of Finland, Government Research Grant Foundation, and the University of Turku (Finland).
A version of this article first appeared on Medscape.com.
compared with patients with obesity who did not have such an operation. However, these declines didn’t extend to cardiovascular medication use.
“In this study, undergoing bariatric surgery was associated with a substantial and long-lasting reduction in the use of lipid-lowering and antidiabetic medications, compared with no surgery for obesity, while for cardiovascular medications this reduction was only transient,” the authors report in research published in JAMA Surgery.
“The results can aid in informed decision-making when considering bariatric surgery for patients with morbid obesity and inform patients and professionals about the expected long-term effects of medication use for obesity-related comorbidities,” they write.
The study “highlights the benefits of mandated databases that report metabolic bariatric surgery, obesity-related comorbidities, and medications,” writes Paulina Salminen, MD, in an accompanying editorial.
However, key limitations include a lack of weight data, which is important in light of previous studies showing that suboptimal weight loss after bariatric surgery is linked to a higher incidence of type 2 diabetes, dyslipidemia, and hypertension, note Dr. Salminen, of the department of digestive surgery, University Hospital, Turku, Finland, and colleagues.
Swedish, Finnish obesity data probed
When significant weight loss is achieved, bariatric surgery has been well documented to be associated with improvements in a variety of comorbidities, quality of life, and even life expectancy.
Key comorbidities shown to improve with the surgery include hyperlipidemia, cardiovascular disease, and type 2 diabetes.
However, data are lacking on the association between bariatric surgery and the use of medications for those conditions, particularly compared with people with obesity who don’t have bariatric surgery.
To investigate, first author Joonas H. Kauppila, MD, PhD, of Upper Gastrointestinal Surgery, Karolinska University Hospital, Stockholm, and colleagues conducted a population-based cohort study, evaluating data on 26,396 patients who underwent bariatric surgery with gastric bypass or sleeve gastrectomy in Sweden between 2005 and 2020 or Finland between 1995 and 2018.
Overall, 66.4% of patients were women and their median age was 50.
They were compared with five times as many matched controls with obesity who had not had bariatric surgery from the same population databases, representing a total of 131,980 patients who were matched based on age, country, sex, calendar year, and medication use.
In terms of lipid-lowering medication, rates of use after bariatric surgery decreased from 20.3% at baseline to 12.9% after 2 years and bounced back somewhat to 17.6% after 15 years. Comparatively, in the no surgery group, baseline lipid-lowering medication use of 21.0% increased to 44.6% after 15 years, more than twice the rate of usage in the bariatric surgery group in the same period.
Antidiabetic medications were used by 27.7% of patients in the bariatric surgery group at baseline, with a drop to 10.0% after 2 years, followed by an increase to 23.5% after 15 years. In the no surgery group, the rate of antidiabetic medication use steadily increased from 27.7% at baseline to 54.2% after 15 years, which again was nearly double the rate of antidiabetic medication use in the bariatric surgery group at 15 years.
Meanwhile, cardiovascular medications were used by 60.2% of patients receiving bariatric surgery at baseline, with the rate decreasing to 43.2% after 2 years but increasing to 74.6% after 15 years. Among the nonbariatric surgery patients, use of cardiovascular medications increased from 54.4% at baseline to 83.3% after 15 years.
Causes?
As for the cause of the lack of any decline in use of cardiovascular medications versus other medications in the surgery patients, the authors speculate that the effect “may be related to aging and regain of weight over time after bariatric surgery, a phenomenon caused by hormonal, dietary, physical, and behavioral factors.”
“In contrast, as expected, a gradual increase in the use of all three medication groups was observed over time among the patients treated with no surgery for obesity,” they note.
The lower medication use with bariatric surgery can also translate to economic benefits, the authors add.
“Economically, the long-lasting reductions in medication use for hyperlipidemia, cardiovascular morbidity, and diabetes suggest that surgical treatment of morbid obesity may infer savings in medication expenses for patients, health care, and society,” they report.
“Future research may focus on subgroups that are most likely to benefit from bariatric surgery, including resolution and severity of comorbidities,” they continue.
In their editorial, Dr. Salminen and colleagues note that previous research has shown remission of dyslipidemia in up to 70% of patients after bariatric surgery that was independent of weight loss, which appears to support the sustained reduction in lipid-lowering medications following surgery observed in the current study, suggesting some benefits on lipids beyond weight loss.
Other limitations, however, include that the bariatric surgery group in the study was older and had more comorbidities than those in previous bariatric surgery studies.
“Future studies should assess this in a younger cohort with less disease at baseline and differentiation within cardiovascular disease regarding at least hypertension, ischemic heart disease, and heart failure,” the authors conclude.
The authors have reported no relevant financial relationships. Dr. Salminen has reported receiving grants from the Sigrid Jusélius Foundation, Academy of Finland, Government Research Grant Foundation, and the University of Turku (Finland).
A version of this article first appeared on Medscape.com.
compared with patients with obesity who did not have such an operation. However, these declines didn’t extend to cardiovascular medication use.
“In this study, undergoing bariatric surgery was associated with a substantial and long-lasting reduction in the use of lipid-lowering and antidiabetic medications, compared with no surgery for obesity, while for cardiovascular medications this reduction was only transient,” the authors report in research published in JAMA Surgery.
“The results can aid in informed decision-making when considering bariatric surgery for patients with morbid obesity and inform patients and professionals about the expected long-term effects of medication use for obesity-related comorbidities,” they write.
The study “highlights the benefits of mandated databases that report metabolic bariatric surgery, obesity-related comorbidities, and medications,” writes Paulina Salminen, MD, in an accompanying editorial.
However, key limitations include a lack of weight data, which is important in light of previous studies showing that suboptimal weight loss after bariatric surgery is linked to a higher incidence of type 2 diabetes, dyslipidemia, and hypertension, note Dr. Salminen, of the department of digestive surgery, University Hospital, Turku, Finland, and colleagues.
Swedish, Finnish obesity data probed
When significant weight loss is achieved, bariatric surgery has been well documented to be associated with improvements in a variety of comorbidities, quality of life, and even life expectancy.
Key comorbidities shown to improve with the surgery include hyperlipidemia, cardiovascular disease, and type 2 diabetes.
However, data are lacking on the association between bariatric surgery and the use of medications for those conditions, particularly compared with people with obesity who don’t have bariatric surgery.
To investigate, first author Joonas H. Kauppila, MD, PhD, of Upper Gastrointestinal Surgery, Karolinska University Hospital, Stockholm, and colleagues conducted a population-based cohort study, evaluating data on 26,396 patients who underwent bariatric surgery with gastric bypass or sleeve gastrectomy in Sweden between 2005 and 2020 or Finland between 1995 and 2018.
Overall, 66.4% of patients were women and their median age was 50.
They were compared with five times as many matched controls with obesity who had not had bariatric surgery from the same population databases, representing a total of 131,980 patients who were matched based on age, country, sex, calendar year, and medication use.
In terms of lipid-lowering medication, rates of use after bariatric surgery decreased from 20.3% at baseline to 12.9% after 2 years and bounced back somewhat to 17.6% after 15 years. Comparatively, in the no surgery group, baseline lipid-lowering medication use of 21.0% increased to 44.6% after 15 years, more than twice the rate of usage in the bariatric surgery group in the same period.
Antidiabetic medications were used by 27.7% of patients in the bariatric surgery group at baseline, with a drop to 10.0% after 2 years, followed by an increase to 23.5% after 15 years. In the no surgery group, the rate of antidiabetic medication use steadily increased from 27.7% at baseline to 54.2% after 15 years, which again was nearly double the rate of antidiabetic medication use in the bariatric surgery group at 15 years.
Meanwhile, cardiovascular medications were used by 60.2% of patients receiving bariatric surgery at baseline, with the rate decreasing to 43.2% after 2 years but increasing to 74.6% after 15 years. Among the nonbariatric surgery patients, use of cardiovascular medications increased from 54.4% at baseline to 83.3% after 15 years.
Causes?
As for the cause of the lack of any decline in use of cardiovascular medications versus other medications in the surgery patients, the authors speculate that the effect “may be related to aging and regain of weight over time after bariatric surgery, a phenomenon caused by hormonal, dietary, physical, and behavioral factors.”
“In contrast, as expected, a gradual increase in the use of all three medication groups was observed over time among the patients treated with no surgery for obesity,” they note.
The lower medication use with bariatric surgery can also translate to economic benefits, the authors add.
“Economically, the long-lasting reductions in medication use for hyperlipidemia, cardiovascular morbidity, and diabetes suggest that surgical treatment of morbid obesity may infer savings in medication expenses for patients, health care, and society,” they report.
“Future research may focus on subgroups that are most likely to benefit from bariatric surgery, including resolution and severity of comorbidities,” they continue.
In their editorial, Dr. Salminen and colleagues note that previous research has shown remission of dyslipidemia in up to 70% of patients after bariatric surgery that was independent of weight loss, which appears to support the sustained reduction in lipid-lowering medications following surgery observed in the current study, suggesting some benefits on lipids beyond weight loss.
Other limitations, however, include that the bariatric surgery group in the study was older and had more comorbidities than those in previous bariatric surgery studies.
“Future studies should assess this in a younger cohort with less disease at baseline and differentiation within cardiovascular disease regarding at least hypertension, ischemic heart disease, and heart failure,” the authors conclude.
The authors have reported no relevant financial relationships. Dr. Salminen has reported receiving grants from the Sigrid Jusélius Foundation, Academy of Finland, Government Research Grant Foundation, and the University of Turku (Finland).
A version of this article first appeared on Medscape.com.
FROM JAMA SURGERY



















