User login
-
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]


Guidance defines vaping-related respiratory syndrome
ORLANDO – Knowledge of vaping devices, familiarity with terminology, and the ability to quickly pinpoint individuals at risk of lung injury are just a few skills that can help critical care professionals confronted with patients who may have vaping-associated lung disease, according to a new guidance document.
The guidance offers a risk-stratification system that classifies patients into groups based on exposure, symptoms, and imaging results, and provides specific evaluation needs and management strategies for each. The guidance is designed to help critical care professionals efficiently identify those at high risk of respiratory failure.
Physicians also need to communicate with patients to identify what substances are being vaped and develop effective methods to encourage abstinence, according to the authors, led by Craig M. Lilly, MD, FCCP, professor of medicine, anesthesiology, and surgery at the University of Massachusetts, Worcester.
“I would encourage every intensivist, when they leave their intensive care unit at night, [to ask], ‘have I advised against vaping today?’ ” Dr. Lilly said at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
The guidelines, concurrently published as a review article in Critical Care Explorations, propose the term vaping-associated respiratory distress syndrome (VARDS), which the authors say constitutes an acute and progressive respiratory syndrome marked by pathologic changes of lung injury and potentially life-threatening hypoxemic respiratory failure.
They also introduce the three-group Worcester classification system, which is intended to triage vaping-exposed individuals for risk of VARDS based on the presence or absence of vaping-related symptoms and infiltrates, and normal or abnormal oxygen saturation.
“It’s very simple,” said Dr. Lilly, who added that the risk stratification model was developed at the request of Massachusetts public health officials.
Patients with vaping exposure but no symptoms attributable to vaping, such as cough, chest pain, or weight loss, are classified as Worcester Low Risk and testing is not recommended, he said.
By contrast, individuals are considered Worcester Medium Risk if they have vaping exposure, symptoms, and a vaping-associated abnormal pattern on imaging, but no hypoxemia; the presence of hypoxemia would tip the scale toward Worcester High Risk.
“Most patients that have died from vaping have been sent out of emergency rooms when they were noted to be hypoxic,” Dr. Lilly told meeting attendees.
Louella B. Amos, MD, a pediatric pulmonologist at Children’s Hospital of Wisconsin in Milwaukee, said she expects the guidance and risk stratification system will be useful not only for critical care specialists, but for other health care providers as well.
“It’s important to make decisions relatively quickly, depending on the severity of symptoms, and I think this is nice and simple,” Dr. Amos said in an interview.
“We always triage when we see patients, either at the door or in our clinic, or behind that, even in the hospital,” she said. “So I think this can be a great tool for everybody, not only the intensivist, but people who are triaging at the front.”
Management of individuals at low risk of VARDS begins with encouragement of abstinence. “We think that every vaping patient should be advised to quit vaping,” Dr. Lilly said. Patients who are interested in quitting who have not yet worked with someone in their health care team whom they trust can be referred to their primary care physicians for counseling, he added, while those struggling with addiction, unable to quit, and unable to partner with a primary care physician can be referred to an addiction medicine specialist.
For moderate-risk patients, vaping cessation is “absolutely mandatory,” said Dr. Lilly, who recommended monitoring of vaping abstinence, outpatient evaluation based on imaging studies, and adequate follow-up to ensure symptoms resolve, tests normalize, and daily activities bounce back to baseline levels.
The guidance offers more extensive recommendations for the VARDS high-risk group, including supervised vaping abstinence, continuous pulse oximetry, and early intervention with noninvasive ventilation, and mechanical ventilation if required, Dr. Lilly said.
Judging vaping exposure is challenging, requiring clinicians to have a familiarity with the many different devices that are available.
Beyond device type, he added, it’s important to know the various terms for devices and lingo that patients may use to describe them, what solutions are vaped, whether those solutions are commercially prepared or off the street, the dose the device delivers, and a number of other factors, he said.
Clinical evaluation typically comes down to unexplained cough, chest pain, weight loss, fatigue, or dyspnea, though one other clue is whether there are gastrointestinal symptoms: “The same way that aerosols can go down to the lungs, they also go into the GI tract, and when nausea, vomiting, or cramping abdominal pain is tightly associated with vaping exposure, one should assume that the patient has been toxin exposed,” he explained.
Dr. Lilly said he had no financial relationships to disclose.
ORLANDO – Knowledge of vaping devices, familiarity with terminology, and the ability to quickly pinpoint individuals at risk of lung injury are just a few skills that can help critical care professionals confronted with patients who may have vaping-associated lung disease, according to a new guidance document.
The guidance offers a risk-stratification system that classifies patients into groups based on exposure, symptoms, and imaging results, and provides specific evaluation needs and management strategies for each. The guidance is designed to help critical care professionals efficiently identify those at high risk of respiratory failure.
Physicians also need to communicate with patients to identify what substances are being vaped and develop effective methods to encourage abstinence, according to the authors, led by Craig M. Lilly, MD, FCCP, professor of medicine, anesthesiology, and surgery at the University of Massachusetts, Worcester.
“I would encourage every intensivist, when they leave their intensive care unit at night, [to ask], ‘have I advised against vaping today?’ ” Dr. Lilly said at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
The guidelines, concurrently published as a review article in Critical Care Explorations, propose the term vaping-associated respiratory distress syndrome (VARDS), which the authors say constitutes an acute and progressive respiratory syndrome marked by pathologic changes of lung injury and potentially life-threatening hypoxemic respiratory failure.
They also introduce the three-group Worcester classification system, which is intended to triage vaping-exposed individuals for risk of VARDS based on the presence or absence of vaping-related symptoms and infiltrates, and normal or abnormal oxygen saturation.
“It’s very simple,” said Dr. Lilly, who added that the risk stratification model was developed at the request of Massachusetts public health officials.
Patients with vaping exposure but no symptoms attributable to vaping, such as cough, chest pain, or weight loss, are classified as Worcester Low Risk and testing is not recommended, he said.
By contrast, individuals are considered Worcester Medium Risk if they have vaping exposure, symptoms, and a vaping-associated abnormal pattern on imaging, but no hypoxemia; the presence of hypoxemia would tip the scale toward Worcester High Risk.
“Most patients that have died from vaping have been sent out of emergency rooms when they were noted to be hypoxic,” Dr. Lilly told meeting attendees.
Louella B. Amos, MD, a pediatric pulmonologist at Children’s Hospital of Wisconsin in Milwaukee, said she expects the guidance and risk stratification system will be useful not only for critical care specialists, but for other health care providers as well.
“It’s important to make decisions relatively quickly, depending on the severity of symptoms, and I think this is nice and simple,” Dr. Amos said in an interview.
“We always triage when we see patients, either at the door or in our clinic, or behind that, even in the hospital,” she said. “So I think this can be a great tool for everybody, not only the intensivist, but people who are triaging at the front.”
Management of individuals at low risk of VARDS begins with encouragement of abstinence. “We think that every vaping patient should be advised to quit vaping,” Dr. Lilly said. Patients who are interested in quitting who have not yet worked with someone in their health care team whom they trust can be referred to their primary care physicians for counseling, he added, while those struggling with addiction, unable to quit, and unable to partner with a primary care physician can be referred to an addiction medicine specialist.
For moderate-risk patients, vaping cessation is “absolutely mandatory,” said Dr. Lilly, who recommended monitoring of vaping abstinence, outpatient evaluation based on imaging studies, and adequate follow-up to ensure symptoms resolve, tests normalize, and daily activities bounce back to baseline levels.
The guidance offers more extensive recommendations for the VARDS high-risk group, including supervised vaping abstinence, continuous pulse oximetry, and early intervention with noninvasive ventilation, and mechanical ventilation if required, Dr. Lilly said.
Judging vaping exposure is challenging, requiring clinicians to have a familiarity with the many different devices that are available.
Beyond device type, he added, it’s important to know the various terms for devices and lingo that patients may use to describe them, what solutions are vaped, whether those solutions are commercially prepared or off the street, the dose the device delivers, and a number of other factors, he said.
Clinical evaluation typically comes down to unexplained cough, chest pain, weight loss, fatigue, or dyspnea, though one other clue is whether there are gastrointestinal symptoms: “The same way that aerosols can go down to the lungs, they also go into the GI tract, and when nausea, vomiting, or cramping abdominal pain is tightly associated with vaping exposure, one should assume that the patient has been toxin exposed,” he explained.
Dr. Lilly said he had no financial relationships to disclose.
ORLANDO – Knowledge of vaping devices, familiarity with terminology, and the ability to quickly pinpoint individuals at risk of lung injury are just a few skills that can help critical care professionals confronted with patients who may have vaping-associated lung disease, according to a new guidance document.
The guidance offers a risk-stratification system that classifies patients into groups based on exposure, symptoms, and imaging results, and provides specific evaluation needs and management strategies for each. The guidance is designed to help critical care professionals efficiently identify those at high risk of respiratory failure.
Physicians also need to communicate with patients to identify what substances are being vaped and develop effective methods to encourage abstinence, according to the authors, led by Craig M. Lilly, MD, FCCP, professor of medicine, anesthesiology, and surgery at the University of Massachusetts, Worcester.
“I would encourage every intensivist, when they leave their intensive care unit at night, [to ask], ‘have I advised against vaping today?’ ” Dr. Lilly said at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
The guidelines, concurrently published as a review article in Critical Care Explorations, propose the term vaping-associated respiratory distress syndrome (VARDS), which the authors say constitutes an acute and progressive respiratory syndrome marked by pathologic changes of lung injury and potentially life-threatening hypoxemic respiratory failure.
They also introduce the three-group Worcester classification system, which is intended to triage vaping-exposed individuals for risk of VARDS based on the presence or absence of vaping-related symptoms and infiltrates, and normal or abnormal oxygen saturation.
“It’s very simple,” said Dr. Lilly, who added that the risk stratification model was developed at the request of Massachusetts public health officials.
Patients with vaping exposure but no symptoms attributable to vaping, such as cough, chest pain, or weight loss, are classified as Worcester Low Risk and testing is not recommended, he said.
By contrast, individuals are considered Worcester Medium Risk if they have vaping exposure, symptoms, and a vaping-associated abnormal pattern on imaging, but no hypoxemia; the presence of hypoxemia would tip the scale toward Worcester High Risk.
“Most patients that have died from vaping have been sent out of emergency rooms when they were noted to be hypoxic,” Dr. Lilly told meeting attendees.
Louella B. Amos, MD, a pediatric pulmonologist at Children’s Hospital of Wisconsin in Milwaukee, said she expects the guidance and risk stratification system will be useful not only for critical care specialists, but for other health care providers as well.
“It’s important to make decisions relatively quickly, depending on the severity of symptoms, and I think this is nice and simple,” Dr. Amos said in an interview.
“We always triage when we see patients, either at the door or in our clinic, or behind that, even in the hospital,” she said. “So I think this can be a great tool for everybody, not only the intensivist, but people who are triaging at the front.”
Management of individuals at low risk of VARDS begins with encouragement of abstinence. “We think that every vaping patient should be advised to quit vaping,” Dr. Lilly said. Patients who are interested in quitting who have not yet worked with someone in their health care team whom they trust can be referred to their primary care physicians for counseling, he added, while those struggling with addiction, unable to quit, and unable to partner with a primary care physician can be referred to an addiction medicine specialist.
For moderate-risk patients, vaping cessation is “absolutely mandatory,” said Dr. Lilly, who recommended monitoring of vaping abstinence, outpatient evaluation based on imaging studies, and adequate follow-up to ensure symptoms resolve, tests normalize, and daily activities bounce back to baseline levels.
The guidance offers more extensive recommendations for the VARDS high-risk group, including supervised vaping abstinence, continuous pulse oximetry, and early intervention with noninvasive ventilation, and mechanical ventilation if required, Dr. Lilly said.
Judging vaping exposure is challenging, requiring clinicians to have a familiarity with the many different devices that are available.
Beyond device type, he added, it’s important to know the various terms for devices and lingo that patients may use to describe them, what solutions are vaped, whether those solutions are commercially prepared or off the street, the dose the device delivers, and a number of other factors, he said.
Clinical evaluation typically comes down to unexplained cough, chest pain, weight loss, fatigue, or dyspnea, though one other clue is whether there are gastrointestinal symptoms: “The same way that aerosols can go down to the lungs, they also go into the GI tract, and when nausea, vomiting, or cramping abdominal pain is tightly associated with vaping exposure, one should assume that the patient has been toxin exposed,” he explained.
Dr. Lilly said he had no financial relationships to disclose.
REPORTING FROM CCC49
Medicare beneficiaries get few home health visits after ICU stay
ORLANDO – , an analysis of hospital and home health claims data suggests.
The beneficiaries, all discharged directly to home health after an intensive care unit stay, received an average of less than one visit per week in the ensuing month, while a full third received no visits at all, according to authors of the analysis, presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
Living alone and living in a rural area were associated with significantly fewer home health rehabilitation visits, according to investigator Jason Raymond Falvey, PT, DPT, PhD, of Yale University, New Haven, Conn.
“We identified that these patients are receiving low doses of rehabilitation in home health care settings, and perhaps doses low enough to not be physiologically adequate to overcome the deconditioning and aerobic capacity concerns that these patients have,” Dr. Falvey said.
These findings reflect an “underrecognition” of the importance of rehabilitation both outside and inside the hospital setting, according to Patricia J. Posa, RN, of Saint Joseph Mercy Hospital, Northville, Mich.
“We even struggle to provide sufficient rehabilitation while they’re in the hospital,” Ms. Posa said in an interview. “So I think that we still have a major gap in providing rehab services across the continuum, and part of that is recognizing the deficits that patients, especially our elderly patients, might be leaving the hospital with.”
Medicare beneficiaries who survive a critical illness are often discharged with referrals for physical, occupational, or speech therapy, yet there are not much data on the delivery of that care or how many visits actually take place, according to Dr. Falvey.
He and coinvestigators analyzed data on 3,176 Medicare beneficiaries discharged to home health right after an acute hospitalization with an ICU stay of at least 24 hours. To do this, they linked 2012 Medicare hospital and home health claims data with Medicare demographic and patient assessment data.
They found that the beneficiaries received just 3.5 home rehabilitation visits in 30 days, while 33% had no visits on record.
The factors most strongly associated with receiving fewer rehabilitation visits, in adjusted models, included living in a rural setting, with a rate ratio (RR) of 0.87 and living alone, with an RR of 0.88.
Higher comorbidity count also was associated with fewer visits (RR, 0.98), according to the investigators.
On the other hand, Medicare beneficiaries who received more visits were more likely to be older (RR, 1.03; 1.01-1.04; for every 5 years), more likely to have higher disability scores (RR, 1.03; 1.02-1.04; per point on the Elixhauser Comorbidity Index), and more likely to have reported severe dyspnea (RR, 1.12; 1.04-1.21), according to the report.
More research will be needed to determine the appropriate number of home health rehabilitation visits for older hospitalized patients, according to Ms. Pena, a member of the Society of Critical Care Medicine’s ICU Liberation initiative, which aims to free patients from the harmful effects of pain, agitation/sedation, delirium, immobility, and sleep disruption in the ICU, as well as improve patient outcomes after an ICU stay.
The literature is already fairly robust, she said, on how frequently visits are warranted following specific scenarios such as postsurgical hip or knee replacement or stroke.
“For the general hospitalized patients that are just losing function because they were sick and didn’t get out of bed enough, we don’t really have good data to say, ‘you know, they need three visits a week, or they need two visits a week for an hour in order to improve,’ ” she said, “so the science is still not caught up with the frequency.”
In the absence of data, the number of visits may be left up to an individual clinician’s knowledge and past experience as well as what insurance will pay for, Ms. Pena said.
Dr. Falvey reported royalties related to an online continuing education course on hospital readmissions. No other disclosures were reported.
SOURCE: Falvey J et al. Crit Care Med. 2020 Jan;48(1):28.
ORLANDO – , an analysis of hospital and home health claims data suggests.
The beneficiaries, all discharged directly to home health after an intensive care unit stay, received an average of less than one visit per week in the ensuing month, while a full third received no visits at all, according to authors of the analysis, presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
Living alone and living in a rural area were associated with significantly fewer home health rehabilitation visits, according to investigator Jason Raymond Falvey, PT, DPT, PhD, of Yale University, New Haven, Conn.
“We identified that these patients are receiving low doses of rehabilitation in home health care settings, and perhaps doses low enough to not be physiologically adequate to overcome the deconditioning and aerobic capacity concerns that these patients have,” Dr. Falvey said.
These findings reflect an “underrecognition” of the importance of rehabilitation both outside and inside the hospital setting, according to Patricia J. Posa, RN, of Saint Joseph Mercy Hospital, Northville, Mich.
“We even struggle to provide sufficient rehabilitation while they’re in the hospital,” Ms. Posa said in an interview. “So I think that we still have a major gap in providing rehab services across the continuum, and part of that is recognizing the deficits that patients, especially our elderly patients, might be leaving the hospital with.”
Medicare beneficiaries who survive a critical illness are often discharged with referrals for physical, occupational, or speech therapy, yet there are not much data on the delivery of that care or how many visits actually take place, according to Dr. Falvey.
He and coinvestigators analyzed data on 3,176 Medicare beneficiaries discharged to home health right after an acute hospitalization with an ICU stay of at least 24 hours. To do this, they linked 2012 Medicare hospital and home health claims data with Medicare demographic and patient assessment data.
They found that the beneficiaries received just 3.5 home rehabilitation visits in 30 days, while 33% had no visits on record.
The factors most strongly associated with receiving fewer rehabilitation visits, in adjusted models, included living in a rural setting, with a rate ratio (RR) of 0.87 and living alone, with an RR of 0.88.
Higher comorbidity count also was associated with fewer visits (RR, 0.98), according to the investigators.
On the other hand, Medicare beneficiaries who received more visits were more likely to be older (RR, 1.03; 1.01-1.04; for every 5 years), more likely to have higher disability scores (RR, 1.03; 1.02-1.04; per point on the Elixhauser Comorbidity Index), and more likely to have reported severe dyspnea (RR, 1.12; 1.04-1.21), according to the report.
More research will be needed to determine the appropriate number of home health rehabilitation visits for older hospitalized patients, according to Ms. Pena, a member of the Society of Critical Care Medicine’s ICU Liberation initiative, which aims to free patients from the harmful effects of pain, agitation/sedation, delirium, immobility, and sleep disruption in the ICU, as well as improve patient outcomes after an ICU stay.
The literature is already fairly robust, she said, on how frequently visits are warranted following specific scenarios such as postsurgical hip or knee replacement or stroke.
“For the general hospitalized patients that are just losing function because they were sick and didn’t get out of bed enough, we don’t really have good data to say, ‘you know, they need three visits a week, or they need two visits a week for an hour in order to improve,’ ” she said, “so the science is still not caught up with the frequency.”
In the absence of data, the number of visits may be left up to an individual clinician’s knowledge and past experience as well as what insurance will pay for, Ms. Pena said.
Dr. Falvey reported royalties related to an online continuing education course on hospital readmissions. No other disclosures were reported.
SOURCE: Falvey J et al. Crit Care Med. 2020 Jan;48(1):28.
ORLANDO – , an analysis of hospital and home health claims data suggests.
The beneficiaries, all discharged directly to home health after an intensive care unit stay, received an average of less than one visit per week in the ensuing month, while a full third received no visits at all, according to authors of the analysis, presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
Living alone and living in a rural area were associated with significantly fewer home health rehabilitation visits, according to investigator Jason Raymond Falvey, PT, DPT, PhD, of Yale University, New Haven, Conn.
“We identified that these patients are receiving low doses of rehabilitation in home health care settings, and perhaps doses low enough to not be physiologically adequate to overcome the deconditioning and aerobic capacity concerns that these patients have,” Dr. Falvey said.
These findings reflect an “underrecognition” of the importance of rehabilitation both outside and inside the hospital setting, according to Patricia J. Posa, RN, of Saint Joseph Mercy Hospital, Northville, Mich.
“We even struggle to provide sufficient rehabilitation while they’re in the hospital,” Ms. Posa said in an interview. “So I think that we still have a major gap in providing rehab services across the continuum, and part of that is recognizing the deficits that patients, especially our elderly patients, might be leaving the hospital with.”
Medicare beneficiaries who survive a critical illness are often discharged with referrals for physical, occupational, or speech therapy, yet there are not much data on the delivery of that care or how many visits actually take place, according to Dr. Falvey.
He and coinvestigators analyzed data on 3,176 Medicare beneficiaries discharged to home health right after an acute hospitalization with an ICU stay of at least 24 hours. To do this, they linked 2012 Medicare hospital and home health claims data with Medicare demographic and patient assessment data.
They found that the beneficiaries received just 3.5 home rehabilitation visits in 30 days, while 33% had no visits on record.
The factors most strongly associated with receiving fewer rehabilitation visits, in adjusted models, included living in a rural setting, with a rate ratio (RR) of 0.87 and living alone, with an RR of 0.88.
Higher comorbidity count also was associated with fewer visits (RR, 0.98), according to the investigators.
On the other hand, Medicare beneficiaries who received more visits were more likely to be older (RR, 1.03; 1.01-1.04; for every 5 years), more likely to have higher disability scores (RR, 1.03; 1.02-1.04; per point on the Elixhauser Comorbidity Index), and more likely to have reported severe dyspnea (RR, 1.12; 1.04-1.21), according to the report.
More research will be needed to determine the appropriate number of home health rehabilitation visits for older hospitalized patients, according to Ms. Pena, a member of the Society of Critical Care Medicine’s ICU Liberation initiative, which aims to free patients from the harmful effects of pain, agitation/sedation, delirium, immobility, and sleep disruption in the ICU, as well as improve patient outcomes after an ICU stay.
The literature is already fairly robust, she said, on how frequently visits are warranted following specific scenarios such as postsurgical hip or knee replacement or stroke.
“For the general hospitalized patients that are just losing function because they were sick and didn’t get out of bed enough, we don’t really have good data to say, ‘you know, they need three visits a week, or they need two visits a week for an hour in order to improve,’ ” she said, “so the science is still not caught up with the frequency.”
In the absence of data, the number of visits may be left up to an individual clinician’s knowledge and past experience as well as what insurance will pay for, Ms. Pena said.
Dr. Falvey reported royalties related to an online continuing education course on hospital readmissions. No other disclosures were reported.
SOURCE: Falvey J et al. Crit Care Med. 2020 Jan;48(1):28.
REPORTING FROM CCC49
Opioid use disorder up in sepsis hospitalizations
ORLANDO –
The prevalence of opioid use disorder (OUD) has significantly increased over the past 15 years, the analysis further shows.
Results of the study, presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine, further suggested that OUD disproportionately contributes to sepsis deaths in younger, healthier patients.
Together, these findings underscore the importance of ongoing efforts to address the opioid epidemic in the United States, according to researcher Mohammad Alrawashdeh, PhD, MSN, a postdoctoral research fellow with Harvard Medical School and Harvard Pilgrim Health Care Institute, Boston.
“In addition to ongoing efforts to combat the opioid crisis, future public health interventions should focus on increasing awareness, recognition, and aggressive treatment of sepsis in this population,” Dr. Alrawashdeh said in an oral presentation of the study.
This study fills an important knowledge gap regarding the connection between OUD and sepsis, according to Greg S. Martin, MD, MS, FCCM, professor of medicine in pulmonary critical care at Emory University, Atlanta, and secretary for the Society of Critical Care Medicine.
“We’ve not really ever been able to piece together the relationship between opioid use disorders and sepsis,” Dr. Martin said in an interview. “It’s not that people wouldn’t suspect that there’s a connection – it’s more that we have simply not been able to get the kind of data that you can use, like they’ve done here, that really helps you to answer that question.”
The study suggests not only that OUD and sepsis are linked, Dr. Martin added, but that health care providers need to be prepared to potentially see further increases in the number of patients with OUD seen in the intensive care unit.
“Both of those are things that we certainly need to be aware of, both from the individual practitioner perspective and also the public health planning perspective,” he said.
The retrospective study by Dr. Alrawashdeh and coinvestigators focused on electronic health record data for adults admitted to 373 hospitals in the United States between 2009 and 2015, including 375,479 who had sepsis.
Over time, there was a significant increase in the prevalence of OUD among those hospitalized for sepsis, from less than 2.0% in 2009 to more than 3% in 2015, representing a significant 77.3% increase. In general, the prevalence of sepsis was significantly higher among hospitalized patients with OUD compared with patients without the disorder, at 7.2% and 5.6%, respectively.
The sepsis patients with OUD tended to be younger, healthier, and more likely to be white compared with patients without OUD, according to the report. Moreover, the sepsis patients with OUD more often had endocarditis and gram-positive and fungal bloodstream infections. They also required more mechanical ventilation and had more ICU admissions, with longer stays in both the ICU and hospital.
The OUD patients accounted for 2.1% of sepsis-associated deaths overall, but 3.3% of those deaths in healthy patients, and 7.1% of deaths among younger patients, according to the report.
Those findings provide some clues that could help guide clinical practice, according to Dr. Martin. For example, the data show a nearly fivefold increased risk of endocarditis with OUD (3.9% versus 0.7%), which may inform screening practices.
“While we don’t necessarily screen every sepsis patient for endocarditis, if it’s an opioid use disorder patient – particularly one with a bloodstream infection – then that’s almost certainly something you should be doing,” Dr. Martin said.
The data suggest gram-positive bacterial and fungal infections will more likely be encountered among these patients, which could guide empiric treatment, he said.
Providers specializing in OUD should have a heightened awareness of the potential for infection and sepsis among those patients, and perhaps be more attuned to fever and other signs of infection that might warrant a referral or additional care, Dr. Martin added.
Dr. Alrawashdeh reported no disclosures related to the study.
SOURCE: Alrawashdeh M et al. Crit Care Med. 2020 Jan;48(1):28. Abstract 56.
ORLANDO –
The prevalence of opioid use disorder (OUD) has significantly increased over the past 15 years, the analysis further shows.
Results of the study, presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine, further suggested that OUD disproportionately contributes to sepsis deaths in younger, healthier patients.
Together, these findings underscore the importance of ongoing efforts to address the opioid epidemic in the United States, according to researcher Mohammad Alrawashdeh, PhD, MSN, a postdoctoral research fellow with Harvard Medical School and Harvard Pilgrim Health Care Institute, Boston.
“In addition to ongoing efforts to combat the opioid crisis, future public health interventions should focus on increasing awareness, recognition, and aggressive treatment of sepsis in this population,” Dr. Alrawashdeh said in an oral presentation of the study.
This study fills an important knowledge gap regarding the connection between OUD and sepsis, according to Greg S. Martin, MD, MS, FCCM, professor of medicine in pulmonary critical care at Emory University, Atlanta, and secretary for the Society of Critical Care Medicine.
“We’ve not really ever been able to piece together the relationship between opioid use disorders and sepsis,” Dr. Martin said in an interview. “It’s not that people wouldn’t suspect that there’s a connection – it’s more that we have simply not been able to get the kind of data that you can use, like they’ve done here, that really helps you to answer that question.”
The study suggests not only that OUD and sepsis are linked, Dr. Martin added, but that health care providers need to be prepared to potentially see further increases in the number of patients with OUD seen in the intensive care unit.
“Both of those are things that we certainly need to be aware of, both from the individual practitioner perspective and also the public health planning perspective,” he said.
The retrospective study by Dr. Alrawashdeh and coinvestigators focused on electronic health record data for adults admitted to 373 hospitals in the United States between 2009 and 2015, including 375,479 who had sepsis.
Over time, there was a significant increase in the prevalence of OUD among those hospitalized for sepsis, from less than 2.0% in 2009 to more than 3% in 2015, representing a significant 77.3% increase. In general, the prevalence of sepsis was significantly higher among hospitalized patients with OUD compared with patients without the disorder, at 7.2% and 5.6%, respectively.
The sepsis patients with OUD tended to be younger, healthier, and more likely to be white compared with patients without OUD, according to the report. Moreover, the sepsis patients with OUD more often had endocarditis and gram-positive and fungal bloodstream infections. They also required more mechanical ventilation and had more ICU admissions, with longer stays in both the ICU and hospital.
The OUD patients accounted for 2.1% of sepsis-associated deaths overall, but 3.3% of those deaths in healthy patients, and 7.1% of deaths among younger patients, according to the report.
Those findings provide some clues that could help guide clinical practice, according to Dr. Martin. For example, the data show a nearly fivefold increased risk of endocarditis with OUD (3.9% versus 0.7%), which may inform screening practices.
“While we don’t necessarily screen every sepsis patient for endocarditis, if it’s an opioid use disorder patient – particularly one with a bloodstream infection – then that’s almost certainly something you should be doing,” Dr. Martin said.
The data suggest gram-positive bacterial and fungal infections will more likely be encountered among these patients, which could guide empiric treatment, he said.
Providers specializing in OUD should have a heightened awareness of the potential for infection and sepsis among those patients, and perhaps be more attuned to fever and other signs of infection that might warrant a referral or additional care, Dr. Martin added.
Dr. Alrawashdeh reported no disclosures related to the study.
SOURCE: Alrawashdeh M et al. Crit Care Med. 2020 Jan;48(1):28. Abstract 56.
ORLANDO –
The prevalence of opioid use disorder (OUD) has significantly increased over the past 15 years, the analysis further shows.
Results of the study, presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine, further suggested that OUD disproportionately contributes to sepsis deaths in younger, healthier patients.
Together, these findings underscore the importance of ongoing efforts to address the opioid epidemic in the United States, according to researcher Mohammad Alrawashdeh, PhD, MSN, a postdoctoral research fellow with Harvard Medical School and Harvard Pilgrim Health Care Institute, Boston.
“In addition to ongoing efforts to combat the opioid crisis, future public health interventions should focus on increasing awareness, recognition, and aggressive treatment of sepsis in this population,” Dr. Alrawashdeh said in an oral presentation of the study.
This study fills an important knowledge gap regarding the connection between OUD and sepsis, according to Greg S. Martin, MD, MS, FCCM, professor of medicine in pulmonary critical care at Emory University, Atlanta, and secretary for the Society of Critical Care Medicine.
“We’ve not really ever been able to piece together the relationship between opioid use disorders and sepsis,” Dr. Martin said in an interview. “It’s not that people wouldn’t suspect that there’s a connection – it’s more that we have simply not been able to get the kind of data that you can use, like they’ve done here, that really helps you to answer that question.”
The study suggests not only that OUD and sepsis are linked, Dr. Martin added, but that health care providers need to be prepared to potentially see further increases in the number of patients with OUD seen in the intensive care unit.
“Both of those are things that we certainly need to be aware of, both from the individual practitioner perspective and also the public health planning perspective,” he said.
The retrospective study by Dr. Alrawashdeh and coinvestigators focused on electronic health record data for adults admitted to 373 hospitals in the United States between 2009 and 2015, including 375,479 who had sepsis.
Over time, there was a significant increase in the prevalence of OUD among those hospitalized for sepsis, from less than 2.0% in 2009 to more than 3% in 2015, representing a significant 77.3% increase. In general, the prevalence of sepsis was significantly higher among hospitalized patients with OUD compared with patients without the disorder, at 7.2% and 5.6%, respectively.
The sepsis patients with OUD tended to be younger, healthier, and more likely to be white compared with patients without OUD, according to the report. Moreover, the sepsis patients with OUD more often had endocarditis and gram-positive and fungal bloodstream infections. They also required more mechanical ventilation and had more ICU admissions, with longer stays in both the ICU and hospital.
The OUD patients accounted for 2.1% of sepsis-associated deaths overall, but 3.3% of those deaths in healthy patients, and 7.1% of deaths among younger patients, according to the report.
Those findings provide some clues that could help guide clinical practice, according to Dr. Martin. For example, the data show a nearly fivefold increased risk of endocarditis with OUD (3.9% versus 0.7%), which may inform screening practices.
“While we don’t necessarily screen every sepsis patient for endocarditis, if it’s an opioid use disorder patient – particularly one with a bloodstream infection – then that’s almost certainly something you should be doing,” Dr. Martin said.
The data suggest gram-positive bacterial and fungal infections will more likely be encountered among these patients, which could guide empiric treatment, he said.
Providers specializing in OUD should have a heightened awareness of the potential for infection and sepsis among those patients, and perhaps be more attuned to fever and other signs of infection that might warrant a referral or additional care, Dr. Martin added.
Dr. Alrawashdeh reported no disclosures related to the study.
SOURCE: Alrawashdeh M et al. Crit Care Med. 2020 Jan;48(1):28. Abstract 56.
REPORTING FROM CCC49
Vitamin E acetate found in more vapers’ lung fluid
Analysis of additional lung fluid samples confirms the presence of vitamin E acetate in patients with electronic-cigarette, or vaping, product use–associated lung injury, according to a report on 51 patients in 16 states.
The average age of the patients was 23 years; 69% were male.
The report extends previous work by the Centers for Disease Control and Prevention to test for harmful substances in bronchoalveolar-lavage (BAL) fluid obtained from patients with electronic-cigarette, or vaping, product use–associated lung injury (EVALI) as part of a strategy to understand and manage the recent outbreak of EVALI cases in the United States, wrote Benjamin C. Blount, PhD, of the Division of Laboratory Sciences at the CDC’s National Center for Environmental Health, and colleagues.
“CDC was addressing a serious outbreak of lung injury that was sometimes lethal; but after the first 10 weeks of the outbreak investigation, the cause was still unknown,” Dr. Blount said in an interview. “Possible theories could not be evaluated unless the laboratory could develop tests that could confidently connect exposure to lung injury. Detection of toxicants in bronchoalveolar-lavage fluid from patients with EVALI can provide direct information on exposure within the lung.”
In a study published in the New England Journal of Medicine, the researchers examined the BAL of 51 cases of EVALI from 16 states. They analyzed the samples for multiple toxicants, including vitamin E acetate, plant oils, medium-chain triglyceride oil, coconut oil, petroleum distillates, and diluent terpenes.
Overall, 77% of the patients reported using products containing THC, 67% reported using products containing nicotine, and 51% reported using both types.
Researchers found vitamin E acetate in 48 of the 51 patients (94%); no vitamin E acetate was found in the BAL of healthy controls. Coconut oil and limonene were found in one patient each, but none of the other toxicants was found in the samples from the patients or controls.
In addition, 47 of the 50 patients for whom data were available either had detectable tetrahydrocannabinol (THC) or its metabolites in their BAL fluid samples, or they reported vaping THC products within 90 days before they became ill. Nicotine or its metabolites were found in 30 of 47 patients (64%).
The study findings were limited by several factors, including the potential role of vitamin E acetate as a marker for exposure to other toxicants, the uncertainty of the role of aerosolized constituents formed when vitamin E acetate is heated, and the lack of data on the timing and burden of toxicant exposure, the investigators noted.
As for the next steps in research, “additional studies are needed to examine the respiratory effects of inhaling aerosolized vitamin E acetate and provide information on whether vitamin E acetate in isolation causes lung injury,” Dr. Blount explained. Analysis of the aerosol and gases generated by case-associated product fluids is ongoing.
“When CDC developed the BAL study for this response, we considered several possible toxicants in this investigation to find a possible cause of the outbreak,” Dr. Blount noted. “To accomplish the study, CDC’s Environmental Health Laboratory developed 12 analytical methods and validated them in less than 3 weeks because of the urgent nature of the emergency.”
Dr. Blount said he would advise clinicians to “continue to reference CDC guidance on treating suspected or EVALI patients.” In December, the CDC published updated guidance for clinicians on hospitalized EVALI patients. “Following this guidance and other recommendations could reduce EVALI-associated morbidity and mortality,” Dr. Blount said.
The study was supported in part by the National Cancer Institute, the FDA Center for Tobacco Products, and Ohio State University Pelotonia Intramural Research. The researchers had no financial conflicts to disclose.
SOURCE: Blount BC et al. N Engl J Med. 2020 Feb 20. doi: 10.1056/NEJMoa1916433.
Analysis of additional lung fluid samples confirms the presence of vitamin E acetate in patients with electronic-cigarette, or vaping, product use–associated lung injury, according to a report on 51 patients in 16 states.
The average age of the patients was 23 years; 69% were male.
The report extends previous work by the Centers for Disease Control and Prevention to test for harmful substances in bronchoalveolar-lavage (BAL) fluid obtained from patients with electronic-cigarette, or vaping, product use–associated lung injury (EVALI) as part of a strategy to understand and manage the recent outbreak of EVALI cases in the United States, wrote Benjamin C. Blount, PhD, of the Division of Laboratory Sciences at the CDC’s National Center for Environmental Health, and colleagues.
“CDC was addressing a serious outbreak of lung injury that was sometimes lethal; but after the first 10 weeks of the outbreak investigation, the cause was still unknown,” Dr. Blount said in an interview. “Possible theories could not be evaluated unless the laboratory could develop tests that could confidently connect exposure to lung injury. Detection of toxicants in bronchoalveolar-lavage fluid from patients with EVALI can provide direct information on exposure within the lung.”
In a study published in the New England Journal of Medicine, the researchers examined the BAL of 51 cases of EVALI from 16 states. They analyzed the samples for multiple toxicants, including vitamin E acetate, plant oils, medium-chain triglyceride oil, coconut oil, petroleum distillates, and diluent terpenes.
Overall, 77% of the patients reported using products containing THC, 67% reported using products containing nicotine, and 51% reported using both types.
Researchers found vitamin E acetate in 48 of the 51 patients (94%); no vitamin E acetate was found in the BAL of healthy controls. Coconut oil and limonene were found in one patient each, but none of the other toxicants was found in the samples from the patients or controls.
In addition, 47 of the 50 patients for whom data were available either had detectable tetrahydrocannabinol (THC) or its metabolites in their BAL fluid samples, or they reported vaping THC products within 90 days before they became ill. Nicotine or its metabolites were found in 30 of 47 patients (64%).
The study findings were limited by several factors, including the potential role of vitamin E acetate as a marker for exposure to other toxicants, the uncertainty of the role of aerosolized constituents formed when vitamin E acetate is heated, and the lack of data on the timing and burden of toxicant exposure, the investigators noted.
As for the next steps in research, “additional studies are needed to examine the respiratory effects of inhaling aerosolized vitamin E acetate and provide information on whether vitamin E acetate in isolation causes lung injury,” Dr. Blount explained. Analysis of the aerosol and gases generated by case-associated product fluids is ongoing.
“When CDC developed the BAL study for this response, we considered several possible toxicants in this investigation to find a possible cause of the outbreak,” Dr. Blount noted. “To accomplish the study, CDC’s Environmental Health Laboratory developed 12 analytical methods and validated them in less than 3 weeks because of the urgent nature of the emergency.”
Dr. Blount said he would advise clinicians to “continue to reference CDC guidance on treating suspected or EVALI patients.” In December, the CDC published updated guidance for clinicians on hospitalized EVALI patients. “Following this guidance and other recommendations could reduce EVALI-associated morbidity and mortality,” Dr. Blount said.
The study was supported in part by the National Cancer Institute, the FDA Center for Tobacco Products, and Ohio State University Pelotonia Intramural Research. The researchers had no financial conflicts to disclose.
SOURCE: Blount BC et al. N Engl J Med. 2020 Feb 20. doi: 10.1056/NEJMoa1916433.
Analysis of additional lung fluid samples confirms the presence of vitamin E acetate in patients with electronic-cigarette, or vaping, product use–associated lung injury, according to a report on 51 patients in 16 states.
The average age of the patients was 23 years; 69% were male.
The report extends previous work by the Centers for Disease Control and Prevention to test for harmful substances in bronchoalveolar-lavage (BAL) fluid obtained from patients with electronic-cigarette, or vaping, product use–associated lung injury (EVALI) as part of a strategy to understand and manage the recent outbreak of EVALI cases in the United States, wrote Benjamin C. Blount, PhD, of the Division of Laboratory Sciences at the CDC’s National Center for Environmental Health, and colleagues.
“CDC was addressing a serious outbreak of lung injury that was sometimes lethal; but after the first 10 weeks of the outbreak investigation, the cause was still unknown,” Dr. Blount said in an interview. “Possible theories could not be evaluated unless the laboratory could develop tests that could confidently connect exposure to lung injury. Detection of toxicants in bronchoalveolar-lavage fluid from patients with EVALI can provide direct information on exposure within the lung.”
In a study published in the New England Journal of Medicine, the researchers examined the BAL of 51 cases of EVALI from 16 states. They analyzed the samples for multiple toxicants, including vitamin E acetate, plant oils, medium-chain triglyceride oil, coconut oil, petroleum distillates, and diluent terpenes.
Overall, 77% of the patients reported using products containing THC, 67% reported using products containing nicotine, and 51% reported using both types.
Researchers found vitamin E acetate in 48 of the 51 patients (94%); no vitamin E acetate was found in the BAL of healthy controls. Coconut oil and limonene were found in one patient each, but none of the other toxicants was found in the samples from the patients or controls.
In addition, 47 of the 50 patients for whom data were available either had detectable tetrahydrocannabinol (THC) or its metabolites in their BAL fluid samples, or they reported vaping THC products within 90 days before they became ill. Nicotine or its metabolites were found in 30 of 47 patients (64%).
The study findings were limited by several factors, including the potential role of vitamin E acetate as a marker for exposure to other toxicants, the uncertainty of the role of aerosolized constituents formed when vitamin E acetate is heated, and the lack of data on the timing and burden of toxicant exposure, the investigators noted.
As for the next steps in research, “additional studies are needed to examine the respiratory effects of inhaling aerosolized vitamin E acetate and provide information on whether vitamin E acetate in isolation causes lung injury,” Dr. Blount explained. Analysis of the aerosol and gases generated by case-associated product fluids is ongoing.
“When CDC developed the BAL study for this response, we considered several possible toxicants in this investigation to find a possible cause of the outbreak,” Dr. Blount noted. “To accomplish the study, CDC’s Environmental Health Laboratory developed 12 analytical methods and validated them in less than 3 weeks because of the urgent nature of the emergency.”
Dr. Blount said he would advise clinicians to “continue to reference CDC guidance on treating suspected or EVALI patients.” In December, the CDC published updated guidance for clinicians on hospitalized EVALI patients. “Following this guidance and other recommendations could reduce EVALI-associated morbidity and mortality,” Dr. Blount said.
The study was supported in part by the National Cancer Institute, the FDA Center for Tobacco Products, and Ohio State University Pelotonia Intramural Research. The researchers had no financial conflicts to disclose.
SOURCE: Blount BC et al. N Engl J Med. 2020 Feb 20. doi: 10.1056/NEJMoa1916433.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
New lipid-lowering drug earns FDA approval
The Food and Drug Administration has approved bempedoic acid (Nexletol) for the treatment of adults with heterozygous familial hypercholesterolemia (HeFH) or established atherosclerotic cardiovascular disease (ASCVD) who require additional LDL cholesterol lowering.
The oral adenosine triphosphate–citrate lyase (ACL) inhibitor is indicated as an adjunct to diet and maximally tolerated statin therapy in these patients, and approved at the 180 mg once daily dose, the agency announced today.
The safety and efficacy of bempedoic acid were demonstrated over 52 weeks in two multicenter randomized, clinical trials involving 3,009 adults with HeFH or established ASCVD on maximally tolerated statin therapy.
The difference between bempedoic acid and placebo for the primary outcome of change in LDL cholesterol from baseline to week 12 was –18% in the first trial, CLEAR Harmony (95% confidence interval, –20% to –16%; P less than .001), and –17% in the second trial, CLEAR Wisdom (95% CI, –21% to –14%; P less than .001).
The label notes that the effect on cardiovascular morbidity and mortality has not been determined. The label also includes warnings stating that bempedoic acid may increase blood uric acid levels and is associated with an increased risk of tendon rupture or injury.
In clinical trials, 26% of bempedoic acid–treated patients with normal baseline uric acid values versus 9.5% of placebo-treated patients experienced hyperuricemia one or more times, and 3.5% of patients experienced clinically significant hyperuricemia reported as an adverse reaction versus 1.1% with placebo, according to the label. Gout was reported in 1.5% of patients treated with bempedoic acid and 0.4% of those treated with placebo.
Also in clinical trials, the risk of tendon rupture was 0.5% with bempedoic acid and 0% with placebo. Tendon rupture involved the rotator cuff, biceps tendon, or Achilles tendon, and occurred within weeks to months of starting the drug. Rupture may “occur more frequently in patients over 60 years of age, in those taking corticosteroid or fluoroquinolone drugs, in patients with renal failure, and in patients with previous tendon disorders,” the label states.
The label also advises that patients avoid concomitant use of bempedoic acid with simvastatin greater than 20 mg or pravastatin greater than 40 mg because it causes an increase in statin concentrations and may increase the risk of related myopathy.
A decision is expected shortly on a new drug application submitted by Esperion for an LDL cholesterol–lowering indication for bempedoic acid 180 mg/ezetimibe 10 mg combination tablet.
Full prescribing information is available online.
This article first appeared on Medscape.com.
The Food and Drug Administration has approved bempedoic acid (Nexletol) for the treatment of adults with heterozygous familial hypercholesterolemia (HeFH) or established atherosclerotic cardiovascular disease (ASCVD) who require additional LDL cholesterol lowering.
The oral adenosine triphosphate–citrate lyase (ACL) inhibitor is indicated as an adjunct to diet and maximally tolerated statin therapy in these patients, and approved at the 180 mg once daily dose, the agency announced today.
The safety and efficacy of bempedoic acid were demonstrated over 52 weeks in two multicenter randomized, clinical trials involving 3,009 adults with HeFH or established ASCVD on maximally tolerated statin therapy.
The difference between bempedoic acid and placebo for the primary outcome of change in LDL cholesterol from baseline to week 12 was –18% in the first trial, CLEAR Harmony (95% confidence interval, –20% to –16%; P less than .001), and –17% in the second trial, CLEAR Wisdom (95% CI, –21% to –14%; P less than .001).
The label notes that the effect on cardiovascular morbidity and mortality has not been determined. The label also includes warnings stating that bempedoic acid may increase blood uric acid levels and is associated with an increased risk of tendon rupture or injury.
In clinical trials, 26% of bempedoic acid–treated patients with normal baseline uric acid values versus 9.5% of placebo-treated patients experienced hyperuricemia one or more times, and 3.5% of patients experienced clinically significant hyperuricemia reported as an adverse reaction versus 1.1% with placebo, according to the label. Gout was reported in 1.5% of patients treated with bempedoic acid and 0.4% of those treated with placebo.
Also in clinical trials, the risk of tendon rupture was 0.5% with bempedoic acid and 0% with placebo. Tendon rupture involved the rotator cuff, biceps tendon, or Achilles tendon, and occurred within weeks to months of starting the drug. Rupture may “occur more frequently in patients over 60 years of age, in those taking corticosteroid or fluoroquinolone drugs, in patients with renal failure, and in patients with previous tendon disorders,” the label states.
The label also advises that patients avoid concomitant use of bempedoic acid with simvastatin greater than 20 mg or pravastatin greater than 40 mg because it causes an increase in statin concentrations and may increase the risk of related myopathy.
A decision is expected shortly on a new drug application submitted by Esperion for an LDL cholesterol–lowering indication for bempedoic acid 180 mg/ezetimibe 10 mg combination tablet.
Full prescribing information is available online.
This article first appeared on Medscape.com.
The Food and Drug Administration has approved bempedoic acid (Nexletol) for the treatment of adults with heterozygous familial hypercholesterolemia (HeFH) or established atherosclerotic cardiovascular disease (ASCVD) who require additional LDL cholesterol lowering.
The oral adenosine triphosphate–citrate lyase (ACL) inhibitor is indicated as an adjunct to diet and maximally tolerated statin therapy in these patients, and approved at the 180 mg once daily dose, the agency announced today.
The safety and efficacy of bempedoic acid were demonstrated over 52 weeks in two multicenter randomized, clinical trials involving 3,009 adults with HeFH or established ASCVD on maximally tolerated statin therapy.
The difference between bempedoic acid and placebo for the primary outcome of change in LDL cholesterol from baseline to week 12 was –18% in the first trial, CLEAR Harmony (95% confidence interval, –20% to –16%; P less than .001), and –17% in the second trial, CLEAR Wisdom (95% CI, –21% to –14%; P less than .001).
The label notes that the effect on cardiovascular morbidity and mortality has not been determined. The label also includes warnings stating that bempedoic acid may increase blood uric acid levels and is associated with an increased risk of tendon rupture or injury.
In clinical trials, 26% of bempedoic acid–treated patients with normal baseline uric acid values versus 9.5% of placebo-treated patients experienced hyperuricemia one or more times, and 3.5% of patients experienced clinically significant hyperuricemia reported as an adverse reaction versus 1.1% with placebo, according to the label. Gout was reported in 1.5% of patients treated with bempedoic acid and 0.4% of those treated with placebo.
Also in clinical trials, the risk of tendon rupture was 0.5% with bempedoic acid and 0% with placebo. Tendon rupture involved the rotator cuff, biceps tendon, or Achilles tendon, and occurred within weeks to months of starting the drug. Rupture may “occur more frequently in patients over 60 years of age, in those taking corticosteroid or fluoroquinolone drugs, in patients with renal failure, and in patients with previous tendon disorders,” the label states.
The label also advises that patients avoid concomitant use of bempedoic acid with simvastatin greater than 20 mg or pravastatin greater than 40 mg because it causes an increase in statin concentrations and may increase the risk of related myopathy.
A decision is expected shortly on a new drug application submitted by Esperion for an LDL cholesterol–lowering indication for bempedoic acid 180 mg/ezetimibe 10 mg combination tablet.
Full prescribing information is available online.
This article first appeared on Medscape.com.
Drop in flu activity suggests season may have peaked
Influenza activity dropped during the week ending Feb. 15, according to the Centers for Disease Control and Prevention. That decline, along with revised data from the 2 previous weeks, suggests that the 2019-2020 season has peaked for the second time. The rate of outpatient visits for influenza-like illness (ILI) came in at 6.1% for the week ending Feb. 15, after two straight weeks at 6.7%, the CDC’s influenza division reported Feb. 21.
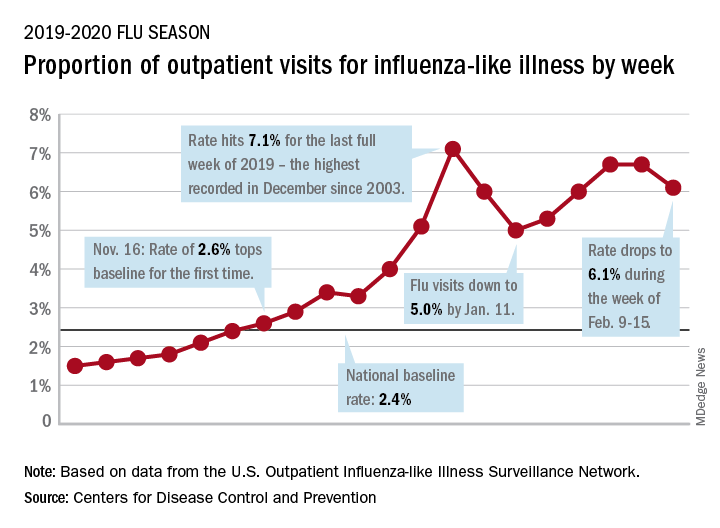
The rates for those 2 earlier weeks had previously been reported at 6.8% (Feb. 8) and 6.6% (Feb. 1), which means that there have now been 2 consecutive weeks without an increase in national ILI activity.
State-level activity was down slightly as well. For the week ending Feb. 15, there were 39 states and Puerto Rico at the highest level of activity on the CDC’s 1-10 scale, compared with 41 states and Puerto Rico the week before. The number of states in the “high” range, which includes levels 8 and 9, went from 44 to 45, however, CDC data show.
Laboratory measures also dropped a bit. For the week, 29.6% of respiratory specimens tested positive for influenza, compared with 30.3% the previous week. The predominance of influenza A continued to increase, as type A went from 59.4% to 63.5% of positive specimens and type B dropped from 40.6% to 36.5%, the influenza division said.
In a separate report, the CDC announced interim flu vaccine effectiveness estimates.For the 2019-2020 season so far, “flu vaccines are reducing doctor’s visits for flu illness by almost half (45%). This is consistent with estimates of flu vaccine effectiveness (VE) from previous flu seasons that ranged from 40% to 60% when flu vaccine viruses were similar to circulating influenza viruses,” the CDC said.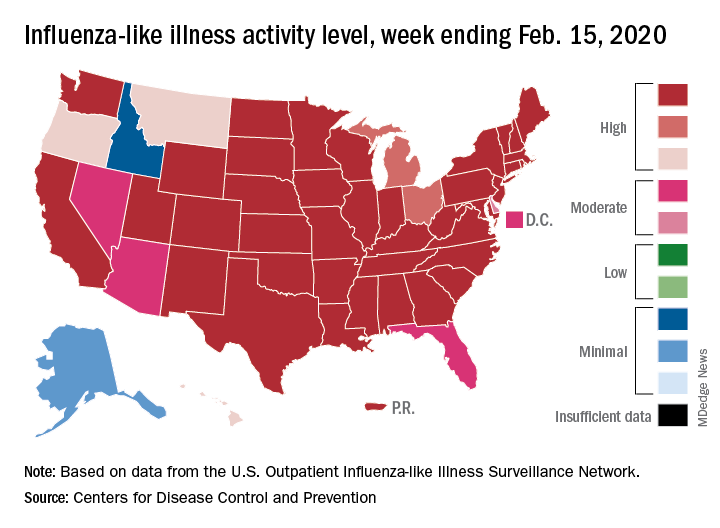
Although VE among children aged 6 months to 17 years is even higher, at 55%, this season “has been especially bad for children. Flu hospitalization rates among children are higher than at this time in other recent seasons, including the 2017-18 season,” the CDC noted.
The number of pediatric flu deaths for 2019-2020 – now up to 105 – is “higher for the same time period than in every season since reporting began in 2004-05, with the exception of the 2009 pandemic,” the CDC added.
Interim VE estimates for other age groups are 25% for adults aged 18-49 and 43% for those 50 years and older. “The lower VE point estimates observed among adults 18-49 years appear to be associated with a trend suggesting lower VE in this age group against A(H1N1)pdm09 viruses,” the CDC said.
Influenza activity dropped during the week ending Feb. 15, according to the Centers for Disease Control and Prevention. That decline, along with revised data from the 2 previous weeks, suggests that the 2019-2020 season has peaked for the second time. The rate of outpatient visits for influenza-like illness (ILI) came in at 6.1% for the week ending Feb. 15, after two straight weeks at 6.7%, the CDC’s influenza division reported Feb. 21.

The rates for those 2 earlier weeks had previously been reported at 6.8% (Feb. 8) and 6.6% (Feb. 1), which means that there have now been 2 consecutive weeks without an increase in national ILI activity.
State-level activity was down slightly as well. For the week ending Feb. 15, there were 39 states and Puerto Rico at the highest level of activity on the CDC’s 1-10 scale, compared with 41 states and Puerto Rico the week before. The number of states in the “high” range, which includes levels 8 and 9, went from 44 to 45, however, CDC data show.
Laboratory measures also dropped a bit. For the week, 29.6% of respiratory specimens tested positive for influenza, compared with 30.3% the previous week. The predominance of influenza A continued to increase, as type A went from 59.4% to 63.5% of positive specimens and type B dropped from 40.6% to 36.5%, the influenza division said.
In a separate report, the CDC announced interim flu vaccine effectiveness estimates.For the 2019-2020 season so far, “flu vaccines are reducing doctor’s visits for flu illness by almost half (45%). This is consistent with estimates of flu vaccine effectiveness (VE) from previous flu seasons that ranged from 40% to 60% when flu vaccine viruses were similar to circulating influenza viruses,” the CDC said.
Although VE among children aged 6 months to 17 years is even higher, at 55%, this season “has been especially bad for children. Flu hospitalization rates among children are higher than at this time in other recent seasons, including the 2017-18 season,” the CDC noted.
The number of pediatric flu deaths for 2019-2020 – now up to 105 – is “higher for the same time period than in every season since reporting began in 2004-05, with the exception of the 2009 pandemic,” the CDC added.
Interim VE estimates for other age groups are 25% for adults aged 18-49 and 43% for those 50 years and older. “The lower VE point estimates observed among adults 18-49 years appear to be associated with a trend suggesting lower VE in this age group against A(H1N1)pdm09 viruses,” the CDC said.
Influenza activity dropped during the week ending Feb. 15, according to the Centers for Disease Control and Prevention. That decline, along with revised data from the 2 previous weeks, suggests that the 2019-2020 season has peaked for the second time. The rate of outpatient visits for influenza-like illness (ILI) came in at 6.1% for the week ending Feb. 15, after two straight weeks at 6.7%, the CDC’s influenza division reported Feb. 21.

The rates for those 2 earlier weeks had previously been reported at 6.8% (Feb. 8) and 6.6% (Feb. 1), which means that there have now been 2 consecutive weeks without an increase in national ILI activity.
State-level activity was down slightly as well. For the week ending Feb. 15, there were 39 states and Puerto Rico at the highest level of activity on the CDC’s 1-10 scale, compared with 41 states and Puerto Rico the week before. The number of states in the “high” range, which includes levels 8 and 9, went from 44 to 45, however, CDC data show.
Laboratory measures also dropped a bit. For the week, 29.6% of respiratory specimens tested positive for influenza, compared with 30.3% the previous week. The predominance of influenza A continued to increase, as type A went from 59.4% to 63.5% of positive specimens and type B dropped from 40.6% to 36.5%, the influenza division said.
In a separate report, the CDC announced interim flu vaccine effectiveness estimates.For the 2019-2020 season so far, “flu vaccines are reducing doctor’s visits for flu illness by almost half (45%). This is consistent with estimates of flu vaccine effectiveness (VE) from previous flu seasons that ranged from 40% to 60% when flu vaccine viruses were similar to circulating influenza viruses,” the CDC said.
Although VE among children aged 6 months to 17 years is even higher, at 55%, this season “has been especially bad for children. Flu hospitalization rates among children are higher than at this time in other recent seasons, including the 2017-18 season,” the CDC noted.
The number of pediatric flu deaths for 2019-2020 – now up to 105 – is “higher for the same time period than in every season since reporting began in 2004-05, with the exception of the 2009 pandemic,” the CDC added.
Interim VE estimates for other age groups are 25% for adults aged 18-49 and 43% for those 50 years and older. “The lower VE point estimates observed among adults 18-49 years appear to be associated with a trend suggesting lower VE in this age group against A(H1N1)pdm09 viruses,” the CDC said.
FROM THE CDC
WHO urges that ‘window of opportunity’ on containing novel coronavirus not be missed
As of 6 a.m. Geneva time, Feb. 21, China reported 75,567 cases of COVID-19 and 2,239 deaths, including a total of 892 new confirmed cases that were reported in China in the past 24 hours, with 118 deaths, stated Tedros Adhanom Ghebreyesus, MD, World Health Organization Director-General, in a Feb. 21 news conference on the COVID-19 outbreak.
What he described as “the significant decline in newly reported cases” is partly because of a change in reporting in which China switched from including “clinically diagnosed” cases to reporting only “suspected” and “laboratory confirmed cases.” The reporting procedure changed because the medical facilities in Wuhan regained the capability of checking all suspected cases with laboratory tests. As a result, some cases that were clinically confirmed were subtracted from the total because they tested negative, said Dr. Ghebreyesus.
Although the number of cases in Hubuei province continues to decline, the WHO is concerned about an increase seen in Shandong province and they are seeking more information. Outside China, there are now 1,152 cases in 26 countries and eight deaths. “Although the number of cases outside of China remains small, we are concerned about the cases with no clear epidemiological link, such as travel history to China, or contact with a confirmed case,” said Dr. Ghebreyesus. “Apart from the Diamond Princess cruise ship, the Republic of Korea now has the most cases outside China, and we are working closely with that government to understand the transmission dynamics that led to this increase.”
“We are also concerned about the increase of cases in the Islamic Republic of Iran, where there are now 18 cases and four deaths in just the past 2 days.”
“Our particular concern is for COVID-19 to spread in countries with weaker health systems,” he said, adding that tomorrow, he will address an emergency meeting of African health ministers held jointly by the African Union and the African Centres for Disease Control and Prevention on dealing with COVID-19.
Dr. Ghebreyesus also announced that today the WHO has designated six special envoys on COVID-19 to provide strategic advice and high-level political advocacy and engagement in different parts of the world.
In his remarks, Dr. Ghebreyesus particularly stressed that: “The measures that China and other countries have taken have given us a fighting chance of containing the spread of the virus. We call on all countries to continue their commitment for containment measures, while preparing for community transmission if it occurs. We must not look back and regret that we failed to take advantage of the window of opportunity that we have now.”
In the question and answer period, Dr. Ghebreyesus specifically addressed the issue of misinformation and conspiracy theories being promulgated by certain individuals and on social media about the source of the virus, especially those people who believe that it was designed in a Chinese virus laboratory. Scientists play an important role in refuting such particular misinformation, he said, and research must continue to track down the actual source in nature.
To that regard, a paper published online in the Lancet on Feb. 19, provided a consensus statement by more than 25 health scientists outside of China stating: “We stand together to strongly condemn conspiracy theories suggesting that COVID-19 does not have a natural origin. Scientists from multiple countries have published and analyzed genomes of the causative agent, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), and they overwhelmingly conclude that this coronavirus originated in wildlife, as have so many other emerging pathogens.”
The WHO issues daily coronavirus disease (COVID-2019) situation reports on its website and provides these telebriefing updates daily.
As of 6 a.m. Geneva time, Feb. 21, China reported 75,567 cases of COVID-19 and 2,239 deaths, including a total of 892 new confirmed cases that were reported in China in the past 24 hours, with 118 deaths, stated Tedros Adhanom Ghebreyesus, MD, World Health Organization Director-General, in a Feb. 21 news conference on the COVID-19 outbreak.
What he described as “the significant decline in newly reported cases” is partly because of a change in reporting in which China switched from including “clinically diagnosed” cases to reporting only “suspected” and “laboratory confirmed cases.” The reporting procedure changed because the medical facilities in Wuhan regained the capability of checking all suspected cases with laboratory tests. As a result, some cases that were clinically confirmed were subtracted from the total because they tested negative, said Dr. Ghebreyesus.
Although the number of cases in Hubuei province continues to decline, the WHO is concerned about an increase seen in Shandong province and they are seeking more information. Outside China, there are now 1,152 cases in 26 countries and eight deaths. “Although the number of cases outside of China remains small, we are concerned about the cases with no clear epidemiological link, such as travel history to China, or contact with a confirmed case,” said Dr. Ghebreyesus. “Apart from the Diamond Princess cruise ship, the Republic of Korea now has the most cases outside China, and we are working closely with that government to understand the transmission dynamics that led to this increase.”
“We are also concerned about the increase of cases in the Islamic Republic of Iran, where there are now 18 cases and four deaths in just the past 2 days.”
“Our particular concern is for COVID-19 to spread in countries with weaker health systems,” he said, adding that tomorrow, he will address an emergency meeting of African health ministers held jointly by the African Union and the African Centres for Disease Control and Prevention on dealing with COVID-19.
Dr. Ghebreyesus also announced that today the WHO has designated six special envoys on COVID-19 to provide strategic advice and high-level political advocacy and engagement in different parts of the world.
In his remarks, Dr. Ghebreyesus particularly stressed that: “The measures that China and other countries have taken have given us a fighting chance of containing the spread of the virus. We call on all countries to continue their commitment for containment measures, while preparing for community transmission if it occurs. We must not look back and regret that we failed to take advantage of the window of opportunity that we have now.”
In the question and answer period, Dr. Ghebreyesus specifically addressed the issue of misinformation and conspiracy theories being promulgated by certain individuals and on social media about the source of the virus, especially those people who believe that it was designed in a Chinese virus laboratory. Scientists play an important role in refuting such particular misinformation, he said, and research must continue to track down the actual source in nature.
To that regard, a paper published online in the Lancet on Feb. 19, provided a consensus statement by more than 25 health scientists outside of China stating: “We stand together to strongly condemn conspiracy theories suggesting that COVID-19 does not have a natural origin. Scientists from multiple countries have published and analyzed genomes of the causative agent, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), and they overwhelmingly conclude that this coronavirus originated in wildlife, as have so many other emerging pathogens.”
The WHO issues daily coronavirus disease (COVID-2019) situation reports on its website and provides these telebriefing updates daily.
As of 6 a.m. Geneva time, Feb. 21, China reported 75,567 cases of COVID-19 and 2,239 deaths, including a total of 892 new confirmed cases that were reported in China in the past 24 hours, with 118 deaths, stated Tedros Adhanom Ghebreyesus, MD, World Health Organization Director-General, in a Feb. 21 news conference on the COVID-19 outbreak.
What he described as “the significant decline in newly reported cases” is partly because of a change in reporting in which China switched from including “clinically diagnosed” cases to reporting only “suspected” and “laboratory confirmed cases.” The reporting procedure changed because the medical facilities in Wuhan regained the capability of checking all suspected cases with laboratory tests. As a result, some cases that were clinically confirmed were subtracted from the total because they tested negative, said Dr. Ghebreyesus.
Although the number of cases in Hubuei province continues to decline, the WHO is concerned about an increase seen in Shandong province and they are seeking more information. Outside China, there are now 1,152 cases in 26 countries and eight deaths. “Although the number of cases outside of China remains small, we are concerned about the cases with no clear epidemiological link, such as travel history to China, or contact with a confirmed case,” said Dr. Ghebreyesus. “Apart from the Diamond Princess cruise ship, the Republic of Korea now has the most cases outside China, and we are working closely with that government to understand the transmission dynamics that led to this increase.”
“We are also concerned about the increase of cases in the Islamic Republic of Iran, where there are now 18 cases and four deaths in just the past 2 days.”
“Our particular concern is for COVID-19 to spread in countries with weaker health systems,” he said, adding that tomorrow, he will address an emergency meeting of African health ministers held jointly by the African Union and the African Centres for Disease Control and Prevention on dealing with COVID-19.
Dr. Ghebreyesus also announced that today the WHO has designated six special envoys on COVID-19 to provide strategic advice and high-level political advocacy and engagement in different parts of the world.
In his remarks, Dr. Ghebreyesus particularly stressed that: “The measures that China and other countries have taken have given us a fighting chance of containing the spread of the virus. We call on all countries to continue their commitment for containment measures, while preparing for community transmission if it occurs. We must not look back and regret that we failed to take advantage of the window of opportunity that we have now.”
In the question and answer period, Dr. Ghebreyesus specifically addressed the issue of misinformation and conspiracy theories being promulgated by certain individuals and on social media about the source of the virus, especially those people who believe that it was designed in a Chinese virus laboratory. Scientists play an important role in refuting such particular misinformation, he said, and research must continue to track down the actual source in nature.
To that regard, a paper published online in the Lancet on Feb. 19, provided a consensus statement by more than 25 health scientists outside of China stating: “We stand together to strongly condemn conspiracy theories suggesting that COVID-19 does not have a natural origin. Scientists from multiple countries have published and analyzed genomes of the causative agent, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), and they overwhelmingly conclude that this coronavirus originated in wildlife, as have so many other emerging pathogens.”
The WHO issues daily coronavirus disease (COVID-2019) situation reports on its website and provides these telebriefing updates daily.
REPORTING FROM A WHO TELEBRIEFING
2019-nCoV: Structure, characteristics of key potential therapy target determined
Researchers have identified the structure of a protein that could turn out to be a potential vaccine target for the 2019-nCoV.
As is typical of other coronaviruses, 2019-nCoV makes use of a densely glycosylated spike protein to gain entry into host cells. The spike protein is a trimeric class I fusion protein that exists in a metastable prefusion conformation that undergoes a dramatic structural rearrangement to fuse the viral membrane with the host-cell membrane, according to Daniel Wrapp of the University of Texas at Austin and colleagues.
The researchers performed a study to synthesize and determine the 3-D structure of the spike protein because it is a logical target for vaccine development and for the development of targeted therapeutics for COVID-19, the disease caused by the virus.
“As soon as we knew this was a coronavirus, we felt we had to jump at it,” senior author Jason S. McLellan, PhD, associate professor of molecular science, said in a press release from the University, “because we could be one of the first ones to get this structure. We knew exactly what mutations to put into this because we’ve already shown these mutations work for a bunch of other coronaviruses.”
Because recent reports by other researchers demonstrated that 2019-nCoV and SARS-CoV spike proteins share the same functional host-cell receptor–angiotensin-converting enzyme 2 (ACE2), Dr. McLellan and his colleagues examined the relation between the two viruses. They found biophysical and structural evidence that the 2019-nCoV spike protein binds ACE2 with higher affinity than the closely related SARS-CoV spike protein. “The high affinity of 2019-nCoV S for human ACE2 may contribute to the apparent ease with which 2019-nCoV can spread from human-to-human; however, additional studies are needed to investigate this possibility,” the researchers wrote.
Focusing their attention on the receptor-binding domain (RBD) of the 2019-nCoV spike protein, they tested several published SARS-CoV RBD-specific monoclonal antibodies against it and found that these antibodies showed no appreciable binding to 2019-nCoV spike protein, which suggests limited antibody cross-reactivity. For this reason, they suggested that future antibody isolation and therapeutic design efforts will benefit from specifically using 2019-nCoV spike proteins as probes.
“This information will support precision vaccine design and discovery of anti-viral therapeutics, accelerating medical countermeasure development,” they concluded.
The research was supported in part by an National Institutes of Health/National Institute of Allergy and Infectious Diseases grant and by intramural funding from the National Institute of Allergy and Infectious Diseases. Four authors are inventors on US patent application No. 62/412,703 (Prefusion Coronavirus Spike Proteins and Their Use) and all are inventors on US patent application No. 62/972,886 (2019-nCoV Vaccine).
SOURCE: Wrapp D et al. Science. 2020 Feb 19. doi: 10.1126/science.abb2507.
Researchers have identified the structure of a protein that could turn out to be a potential vaccine target for the 2019-nCoV.
As is typical of other coronaviruses, 2019-nCoV makes use of a densely glycosylated spike protein to gain entry into host cells. The spike protein is a trimeric class I fusion protein that exists in a metastable prefusion conformation that undergoes a dramatic structural rearrangement to fuse the viral membrane with the host-cell membrane, according to Daniel Wrapp of the University of Texas at Austin and colleagues.
The researchers performed a study to synthesize and determine the 3-D structure of the spike protein because it is a logical target for vaccine development and for the development of targeted therapeutics for COVID-19, the disease caused by the virus.
“As soon as we knew this was a coronavirus, we felt we had to jump at it,” senior author Jason S. McLellan, PhD, associate professor of molecular science, said in a press release from the University, “because we could be one of the first ones to get this structure. We knew exactly what mutations to put into this because we’ve already shown these mutations work for a bunch of other coronaviruses.”
Because recent reports by other researchers demonstrated that 2019-nCoV and SARS-CoV spike proteins share the same functional host-cell receptor–angiotensin-converting enzyme 2 (ACE2), Dr. McLellan and his colleagues examined the relation between the two viruses. They found biophysical and structural evidence that the 2019-nCoV spike protein binds ACE2 with higher affinity than the closely related SARS-CoV spike protein. “The high affinity of 2019-nCoV S for human ACE2 may contribute to the apparent ease with which 2019-nCoV can spread from human-to-human; however, additional studies are needed to investigate this possibility,” the researchers wrote.
Focusing their attention on the receptor-binding domain (RBD) of the 2019-nCoV spike protein, they tested several published SARS-CoV RBD-specific monoclonal antibodies against it and found that these antibodies showed no appreciable binding to 2019-nCoV spike protein, which suggests limited antibody cross-reactivity. For this reason, they suggested that future antibody isolation and therapeutic design efforts will benefit from specifically using 2019-nCoV spike proteins as probes.
“This information will support precision vaccine design and discovery of anti-viral therapeutics, accelerating medical countermeasure development,” they concluded.
The research was supported in part by an National Institutes of Health/National Institute of Allergy and Infectious Diseases grant and by intramural funding from the National Institute of Allergy and Infectious Diseases. Four authors are inventors on US patent application No. 62/412,703 (Prefusion Coronavirus Spike Proteins and Their Use) and all are inventors on US patent application No. 62/972,886 (2019-nCoV Vaccine).
SOURCE: Wrapp D et al. Science. 2020 Feb 19. doi: 10.1126/science.abb2507.
Researchers have identified the structure of a protein that could turn out to be a potential vaccine target for the 2019-nCoV.
As is typical of other coronaviruses, 2019-nCoV makes use of a densely glycosylated spike protein to gain entry into host cells. The spike protein is a trimeric class I fusion protein that exists in a metastable prefusion conformation that undergoes a dramatic structural rearrangement to fuse the viral membrane with the host-cell membrane, according to Daniel Wrapp of the University of Texas at Austin and colleagues.
The researchers performed a study to synthesize and determine the 3-D structure of the spike protein because it is a logical target for vaccine development and for the development of targeted therapeutics for COVID-19, the disease caused by the virus.
“As soon as we knew this was a coronavirus, we felt we had to jump at it,” senior author Jason S. McLellan, PhD, associate professor of molecular science, said in a press release from the University, “because we could be one of the first ones to get this structure. We knew exactly what mutations to put into this because we’ve already shown these mutations work for a bunch of other coronaviruses.”
Because recent reports by other researchers demonstrated that 2019-nCoV and SARS-CoV spike proteins share the same functional host-cell receptor–angiotensin-converting enzyme 2 (ACE2), Dr. McLellan and his colleagues examined the relation between the two viruses. They found biophysical and structural evidence that the 2019-nCoV spike protein binds ACE2 with higher affinity than the closely related SARS-CoV spike protein. “The high affinity of 2019-nCoV S for human ACE2 may contribute to the apparent ease with which 2019-nCoV can spread from human-to-human; however, additional studies are needed to investigate this possibility,” the researchers wrote.
Focusing their attention on the receptor-binding domain (RBD) of the 2019-nCoV spike protein, they tested several published SARS-CoV RBD-specific monoclonal antibodies against it and found that these antibodies showed no appreciable binding to 2019-nCoV spike protein, which suggests limited antibody cross-reactivity. For this reason, they suggested that future antibody isolation and therapeutic design efforts will benefit from specifically using 2019-nCoV spike proteins as probes.
“This information will support precision vaccine design and discovery of anti-viral therapeutics, accelerating medical countermeasure development,” they concluded.
The research was supported in part by an National Institutes of Health/National Institute of Allergy and Infectious Diseases grant and by intramural funding from the National Institute of Allergy and Infectious Diseases. Four authors are inventors on US patent application No. 62/412,703 (Prefusion Coronavirus Spike Proteins and Their Use) and all are inventors on US patent application No. 62/972,886 (2019-nCoV Vaccine).
SOURCE: Wrapp D et al. Science. 2020 Feb 19. doi: 10.1126/science.abb2507.
FROM SCIENCE
Pulsed field catheter ablation shows huge clinical promise for AFib
NATIONAL HARBOR, MD. – Cardiac electrophysiologists have reported using pulsed field ablation, a new power source for catheter ablation of atrial fibrillation, on fewer than 150 patients worldwide in initial clinical studies, but its performance so far and the promise it carries for substantially improving the safety and efficacy of catheter ablation has convinced many experts that it represents the future for this intervention.
“I’m very excited about PFA [pulsed field ablation]. It may make everything else obsolete,” Andrea Natale, MD, said at the annual International AF Symposium. “We need to see more efficacy data, but just for safety alone there is no reason to use anything else,” commented Dr. Natale, executive medical director of the Texas Cardiac Arrhythmia Institute at St. David’s Medical Center in Austin,Tex.
“The main issue is safety, and if PFA lives up to its promise, then [using it preferentially] is not a difficult decision,” commented Francis E. Marchlinski, MD, professor of medicine and director of electrophysiology at the University of Pennsylvania.
“The only question is whether it has good long-term efficacy” because so far no patients have been followed for longer than about a year after PFA treatment, noted Moussa Mansour, MD, director of the cardiac electrophysiology laboratory at Massachusetts General Hospital in Boston. “If that piece turns out to be true, then I think it will be a winner.”
Vivek Y. Reddy, MD, one of the few investigators to have already collaborated on clinical studies that used PFA to catheter ablate both in patients with paroxysmal and, more recently, persistent atrial fibrillation (AFib), put it this way: “I’m 99% sure” PFA will be the energy of choice in the near future for AFib catheter ablation. The 1% of uncertainty “is only because of what might be unknown, something we’re not expecting,” said Dr. Reddy, professor of medicine and director of the cardiac arrhythmia service at Mount Sinai Medical Center in New York.
He and his associates at a center in Prague and at a second site in Bordeaux, France, reported their collective experience in 2019 regarding use of PFA on 81 patients with symptomatic, paroxysmal AFib who had not responded to at least one antiarrhythmic drug (J Am Coll Cardiol. 2019 Jul;74[3]:315-26). During a session on PFA at the symposium, Pierre Jaïs, MD, a cardiac electrophysiologist and professor of cardiology at the University of Bordeaux, updated this experience to now include 113 patients treated by the end of 2019 at the same two centers plus now an added third site, an experience accumulated by a total of five operators. Fifty-one patients have now been followed for at least a year, with no “unexpected” safety events, said Dr. Jaïs, The most recent 88 patients underwent PFA without general anesthesia. The ablation technique has undergone several refinements during this experience, and with use of the most recent, biphasic protocol that’s so far treated 26 patients, 24 (92%) of the treated patients had no reconnected AFib circuits in their atrial tissue when they underwent remapping 3 months after their procedure.
Magnetic resonance imaging of the left atria of these patients after pulmonary vein isolation with PFA showed a uniquely homogeneous and continuous lesion that functionally isolated each vein from surrounding atrial tissue and denoted a more uniform and complete ablation, Dr. Jaïs noted. “I have never seen [an ablation] as homogeneous.” The Magnetic resonance pictures also showed that the esophagus in each treated patient remained completely undamaged. “Esophageal sparing is systematically observed,” along with phrenic nerve sparing that’s in notable contrast with what’s seen with conventional energy sources, he said. The images also indicated that edema was substantially reduced compared with both radiofrequency and cryoablation, while mechanical function of treated left atria has consistently been “well preserved.”
“For the first time, we can use extra power to ensure durable lesions without compromising safety,” Dr. Jaïs concluded. PFA appears to put AFib ablation “on the verge of a totally new era.”
The less extensive and briefer experience in patients with persistent AFib has been completely consistent. This included 25 patients who had not responded to at least one antiarrhythmic drug treated by either of two operators, one in Prague and the other in Split, Croatia. All 25 patients who underwent pulmonary vein isolation had the procedure successfully completed as assessed with acute mapping of arrhythmia circuits after ablation, and the 24 of these patients who also underwent posterior wall ablation with the PFA device all had a successful acute result according to mapping, Dr. Reddy reported. No patient had an adverse event. PFA treatments were relatively fast, with an average procedure time in this series of 132 minutes. Repeat mapping 3 months after treatment is still pending.
At the heart of PFA’s safety is its “myocardial selectivity” which has so far kept PFA from causing any esophageal or phrenic nerve injuries, two potential complications of conventional AFib catheter ablation with use of either radiofrequency or cryo energy. Dr. Reddy was quick to highlight that there is no absolute selectivity for myocardium. “If you create a big enough field, it will electroporate everything, but the margin [between safety and damage] seems wide enough to take advantage” of focally damaging myocardial tissue in the left atrium to disrupt arrhythmia circuits while sparing adjacent tissue. Irreversible electroporation is the means by which PFA destroys targets cells while leaving other tissue unscathed, and a precisely adjusted PFA signal can focus its lethal effect exclusively on myocardial cells, a feature of PFA that Dr. Reddy called “lucky.”
The pulsed field ablation studies have been sponsored by Farapulse, the company developing this device, which in May 2019 received breakthrough designation for priority review from the Food and Drug Administration.
Dr. Reddy and Dr. Jaïs are both consultants to and shareholders in Farapulse. Dr. Natale has received honoraria from or has been a consultant to Biotronik, Janssen, Medtronic, and St. Jude. Dr. Marchlinski has been a consultant to or has received honoraria from Abbott EP/St. Jude, Biotronik, and Medtronic. Dr. Mansour has been a consultant for Abbott and Medtronic, has an equity interest or stock options in NewPace and EPD Solutions, and has received research grants from Abbott, Boehringer Ingelheim, Pfizer, and Sentre Heart. In addition, all sources have received consulting fees, honoraria, and/or research grants from Biosense Webster and Boston Scientific.
NATIONAL HARBOR, MD. – Cardiac electrophysiologists have reported using pulsed field ablation, a new power source for catheter ablation of atrial fibrillation, on fewer than 150 patients worldwide in initial clinical studies, but its performance so far and the promise it carries for substantially improving the safety and efficacy of catheter ablation has convinced many experts that it represents the future for this intervention.
“I’m very excited about PFA [pulsed field ablation]. It may make everything else obsolete,” Andrea Natale, MD, said at the annual International AF Symposium. “We need to see more efficacy data, but just for safety alone there is no reason to use anything else,” commented Dr. Natale, executive medical director of the Texas Cardiac Arrhythmia Institute at St. David’s Medical Center in Austin,Tex.
“The main issue is safety, and if PFA lives up to its promise, then [using it preferentially] is not a difficult decision,” commented Francis E. Marchlinski, MD, professor of medicine and director of electrophysiology at the University of Pennsylvania.
“The only question is whether it has good long-term efficacy” because so far no patients have been followed for longer than about a year after PFA treatment, noted Moussa Mansour, MD, director of the cardiac electrophysiology laboratory at Massachusetts General Hospital in Boston. “If that piece turns out to be true, then I think it will be a winner.”
Vivek Y. Reddy, MD, one of the few investigators to have already collaborated on clinical studies that used PFA to catheter ablate both in patients with paroxysmal and, more recently, persistent atrial fibrillation (AFib), put it this way: “I’m 99% sure” PFA will be the energy of choice in the near future for AFib catheter ablation. The 1% of uncertainty “is only because of what might be unknown, something we’re not expecting,” said Dr. Reddy, professor of medicine and director of the cardiac arrhythmia service at Mount Sinai Medical Center in New York.
He and his associates at a center in Prague and at a second site in Bordeaux, France, reported their collective experience in 2019 regarding use of PFA on 81 patients with symptomatic, paroxysmal AFib who had not responded to at least one antiarrhythmic drug (J Am Coll Cardiol. 2019 Jul;74[3]:315-26). During a session on PFA at the symposium, Pierre Jaïs, MD, a cardiac electrophysiologist and professor of cardiology at the University of Bordeaux, updated this experience to now include 113 patients treated by the end of 2019 at the same two centers plus now an added third site, an experience accumulated by a total of five operators. Fifty-one patients have now been followed for at least a year, with no “unexpected” safety events, said Dr. Jaïs, The most recent 88 patients underwent PFA without general anesthesia. The ablation technique has undergone several refinements during this experience, and with use of the most recent, biphasic protocol that’s so far treated 26 patients, 24 (92%) of the treated patients had no reconnected AFib circuits in their atrial tissue when they underwent remapping 3 months after their procedure.
Magnetic resonance imaging of the left atria of these patients after pulmonary vein isolation with PFA showed a uniquely homogeneous and continuous lesion that functionally isolated each vein from surrounding atrial tissue and denoted a more uniform and complete ablation, Dr. Jaïs noted. “I have never seen [an ablation] as homogeneous.” The Magnetic resonance pictures also showed that the esophagus in each treated patient remained completely undamaged. “Esophageal sparing is systematically observed,” along with phrenic nerve sparing that’s in notable contrast with what’s seen with conventional energy sources, he said. The images also indicated that edema was substantially reduced compared with both radiofrequency and cryoablation, while mechanical function of treated left atria has consistently been “well preserved.”
“For the first time, we can use extra power to ensure durable lesions without compromising safety,” Dr. Jaïs concluded. PFA appears to put AFib ablation “on the verge of a totally new era.”
The less extensive and briefer experience in patients with persistent AFib has been completely consistent. This included 25 patients who had not responded to at least one antiarrhythmic drug treated by either of two operators, one in Prague and the other in Split, Croatia. All 25 patients who underwent pulmonary vein isolation had the procedure successfully completed as assessed with acute mapping of arrhythmia circuits after ablation, and the 24 of these patients who also underwent posterior wall ablation with the PFA device all had a successful acute result according to mapping, Dr. Reddy reported. No patient had an adverse event. PFA treatments were relatively fast, with an average procedure time in this series of 132 minutes. Repeat mapping 3 months after treatment is still pending.
At the heart of PFA’s safety is its “myocardial selectivity” which has so far kept PFA from causing any esophageal or phrenic nerve injuries, two potential complications of conventional AFib catheter ablation with use of either radiofrequency or cryo energy. Dr. Reddy was quick to highlight that there is no absolute selectivity for myocardium. “If you create a big enough field, it will electroporate everything, but the margin [between safety and damage] seems wide enough to take advantage” of focally damaging myocardial tissue in the left atrium to disrupt arrhythmia circuits while sparing adjacent tissue. Irreversible electroporation is the means by which PFA destroys targets cells while leaving other tissue unscathed, and a precisely adjusted PFA signal can focus its lethal effect exclusively on myocardial cells, a feature of PFA that Dr. Reddy called “lucky.”
The pulsed field ablation studies have been sponsored by Farapulse, the company developing this device, which in May 2019 received breakthrough designation for priority review from the Food and Drug Administration.
Dr. Reddy and Dr. Jaïs are both consultants to and shareholders in Farapulse. Dr. Natale has received honoraria from or has been a consultant to Biotronik, Janssen, Medtronic, and St. Jude. Dr. Marchlinski has been a consultant to or has received honoraria from Abbott EP/St. Jude, Biotronik, and Medtronic. Dr. Mansour has been a consultant for Abbott and Medtronic, has an equity interest or stock options in NewPace and EPD Solutions, and has received research grants from Abbott, Boehringer Ingelheim, Pfizer, and Sentre Heart. In addition, all sources have received consulting fees, honoraria, and/or research grants from Biosense Webster and Boston Scientific.
NATIONAL HARBOR, MD. – Cardiac electrophysiologists have reported using pulsed field ablation, a new power source for catheter ablation of atrial fibrillation, on fewer than 150 patients worldwide in initial clinical studies, but its performance so far and the promise it carries for substantially improving the safety and efficacy of catheter ablation has convinced many experts that it represents the future for this intervention.
“I’m very excited about PFA [pulsed field ablation]. It may make everything else obsolete,” Andrea Natale, MD, said at the annual International AF Symposium. “We need to see more efficacy data, but just for safety alone there is no reason to use anything else,” commented Dr. Natale, executive medical director of the Texas Cardiac Arrhythmia Institute at St. David’s Medical Center in Austin,Tex.
“The main issue is safety, and if PFA lives up to its promise, then [using it preferentially] is not a difficult decision,” commented Francis E. Marchlinski, MD, professor of medicine and director of electrophysiology at the University of Pennsylvania.
“The only question is whether it has good long-term efficacy” because so far no patients have been followed for longer than about a year after PFA treatment, noted Moussa Mansour, MD, director of the cardiac electrophysiology laboratory at Massachusetts General Hospital in Boston. “If that piece turns out to be true, then I think it will be a winner.”
Vivek Y. Reddy, MD, one of the few investigators to have already collaborated on clinical studies that used PFA to catheter ablate both in patients with paroxysmal and, more recently, persistent atrial fibrillation (AFib), put it this way: “I’m 99% sure” PFA will be the energy of choice in the near future for AFib catheter ablation. The 1% of uncertainty “is only because of what might be unknown, something we’re not expecting,” said Dr. Reddy, professor of medicine and director of the cardiac arrhythmia service at Mount Sinai Medical Center in New York.
He and his associates at a center in Prague and at a second site in Bordeaux, France, reported their collective experience in 2019 regarding use of PFA on 81 patients with symptomatic, paroxysmal AFib who had not responded to at least one antiarrhythmic drug (J Am Coll Cardiol. 2019 Jul;74[3]:315-26). During a session on PFA at the symposium, Pierre Jaïs, MD, a cardiac electrophysiologist and professor of cardiology at the University of Bordeaux, updated this experience to now include 113 patients treated by the end of 2019 at the same two centers plus now an added third site, an experience accumulated by a total of five operators. Fifty-one patients have now been followed for at least a year, with no “unexpected” safety events, said Dr. Jaïs, The most recent 88 patients underwent PFA without general anesthesia. The ablation technique has undergone several refinements during this experience, and with use of the most recent, biphasic protocol that’s so far treated 26 patients, 24 (92%) of the treated patients had no reconnected AFib circuits in their atrial tissue when they underwent remapping 3 months after their procedure.
Magnetic resonance imaging of the left atria of these patients after pulmonary vein isolation with PFA showed a uniquely homogeneous and continuous lesion that functionally isolated each vein from surrounding atrial tissue and denoted a more uniform and complete ablation, Dr. Jaïs noted. “I have never seen [an ablation] as homogeneous.” The Magnetic resonance pictures also showed that the esophagus in each treated patient remained completely undamaged. “Esophageal sparing is systematically observed,” along with phrenic nerve sparing that’s in notable contrast with what’s seen with conventional energy sources, he said. The images also indicated that edema was substantially reduced compared with both radiofrequency and cryoablation, while mechanical function of treated left atria has consistently been “well preserved.”
“For the first time, we can use extra power to ensure durable lesions without compromising safety,” Dr. Jaïs concluded. PFA appears to put AFib ablation “on the verge of a totally new era.”
The less extensive and briefer experience in patients with persistent AFib has been completely consistent. This included 25 patients who had not responded to at least one antiarrhythmic drug treated by either of two operators, one in Prague and the other in Split, Croatia. All 25 patients who underwent pulmonary vein isolation had the procedure successfully completed as assessed with acute mapping of arrhythmia circuits after ablation, and the 24 of these patients who also underwent posterior wall ablation with the PFA device all had a successful acute result according to mapping, Dr. Reddy reported. No patient had an adverse event. PFA treatments were relatively fast, with an average procedure time in this series of 132 minutes. Repeat mapping 3 months after treatment is still pending.
At the heart of PFA’s safety is its “myocardial selectivity” which has so far kept PFA from causing any esophageal or phrenic nerve injuries, two potential complications of conventional AFib catheter ablation with use of either radiofrequency or cryo energy. Dr. Reddy was quick to highlight that there is no absolute selectivity for myocardium. “If you create a big enough field, it will electroporate everything, but the margin [between safety and damage] seems wide enough to take advantage” of focally damaging myocardial tissue in the left atrium to disrupt arrhythmia circuits while sparing adjacent tissue. Irreversible electroporation is the means by which PFA destroys targets cells while leaving other tissue unscathed, and a precisely adjusted PFA signal can focus its lethal effect exclusively on myocardial cells, a feature of PFA that Dr. Reddy called “lucky.”
The pulsed field ablation studies have been sponsored by Farapulse, the company developing this device, which in May 2019 received breakthrough designation for priority review from the Food and Drug Administration.
Dr. Reddy and Dr. Jaïs are both consultants to and shareholders in Farapulse. Dr. Natale has received honoraria from or has been a consultant to Biotronik, Janssen, Medtronic, and St. Jude. Dr. Marchlinski has been a consultant to or has received honoraria from Abbott EP/St. Jude, Biotronik, and Medtronic. Dr. Mansour has been a consultant for Abbott and Medtronic, has an equity interest or stock options in NewPace and EPD Solutions, and has received research grants from Abbott, Boehringer Ingelheim, Pfizer, and Sentre Heart. In addition, all sources have received consulting fees, honoraria, and/or research grants from Biosense Webster and Boston Scientific.
EXPERT ANALYSIS FROM THE AF SYMPOSIUM 2020
Risk factors found for respiratory AEs in children following OSA surgery
Underlying cardiac disease, airway anomalies, and younger age each independently boosted the risk of severe perioperative respiratory adverse events (PRAE) in children undergoing adenotonsillectomy to treat obstructive sleep apnea, in a review of 374 patients treated at a single Canadian tertiary-referral center.
In contrast, the analysis failed to show independent, significant effects from any assessed polysomnography or oximetry parameters on the rate of postoperative respiratory complications. The utility of preoperative polysomnography or oximetry for risk stratification is questionable for pediatric patients scheduled to adenotonsillectomy to treat obstructive sleep apnea, wrote Sherri L. Katz, MD, of the University of Ottawa, and associates in a recent report published in the Journal of Clinical Sleep Medicine, although they also added that making these assessments may be “unavoidable” because of their need for diagnosing obstructive sleep apnea and determining the need for surgery.
Despite this caveat, “overall our study results highlight the need to better define the complex interaction between comorbidities, age, nocturnal respiratory events, and gas exchange abnormalities in predicting risk for PRAE” after adenotonsillectomy, the researchers wrote. These findings “are consistent with existing clinical care guidelines,” and “cardiac and craniofacial conditions have been associated with risk of postoperative complications in other studies.”
The analysis used data collected from all children aged 0-18 years who underwent polysomnography assessment followed by adenotonsillectomy at one Canadian tertiary-referral center, Children’s Hospital of Eastern Ontario in Ottawa, during 2010-2016. Their median age was just over 6 years, and 39 patients (10%) were younger than 3 years at the time of their surgery. More than three-quarters of the patients, 286, had at least one identified comorbidity, and nearly half had at least two comorbidities. Polysomnography identified sleep-disordered breathing in 344 of the children (92%), and diagnosed obstructive sleep apnea in 256 (68%), including 148 (43% of the full cohort) with a severe apnea-hypopnea index.
Sixty-six of the children (18%) had at least one severe PRAE that required intervention. Specifically these were either oxygen desaturations requiring intervention or need for airway or ventilatory support with interventions such as jaw thrust, oral or nasal airway placement, bag and mask ventilation, or endotracheal intubation.
A multivariate regression analysis of the measured comorbidity, polysomnography, and oximetry parameters, as well as age, identified three factors that independently linked with a statistically significant increase in the rate of severe PRAE: airway anomaly, underlying cardiac disease, and young age. Patients with an airway anomaly had a 219% increased rate of PRAE, compared with those with no anomaly; patients with underlying cardiac disease had a 109% increased rate, compared with those without cardiac disease; and patients aged younger than 3 years had a 310% higher rate of PRAE, compared with the children aged 6 years or older, while children aged 3-5 years had a 121% higher rate of PRAE, compared with older children.
The study received no commercial funding. Dr. Katz has received honoraria for speaking from Biogen that had no relevance to the study.
SOURCE: Katz SL et al. J Clin Sleep Med. 2020 Jan 15;16(1):41-8.
This well-conducted, retrospective, chart-review study adds important information to the published literature about risk stratification for children in a tertiary-referral population undergoing adenotonsillectomy. Their findings indicate that younger children remain at higher risk as well as those children with complex comorbid medical disease. They also show that children with severe sleep apnea or significant oxyhemoglobin desaturation are likewise at higher risk of postoperative respiratory compromise – emphasizing the need for preoperative polysomnography – particularly in a tertiary setting where many patients have medical comorbidities.
Despite the strengths of this study in assessing perioperative risk for respiratory compromise in a referral population with highly prevalent medical comorbidities, this study does not provide significant insight into the management of otherwise healthy children in a community setting who are undergoing adenotonsillectomy. This is important because a large number of adenotonsillectomies are performed outside of a tertiary-referral center and many of these children may not have undergone preoperative polysomnography to stratify risk. The utility of preoperative polysomnography in the evaluation of all children undergoing adenotonsillectomy remains controversial, with diverging recommendations from two major U.S. medical groups.
This study does not address the utility of polysomnography in community-based populations of otherwise healthy children. It is imperative to accurately ascertain risk so perioperative planning can ensure the safety of children at higher risk following adenotonsillectomy; however, there remains a paucity of studies assessing the cost-effectiveness as well as the positive and negative predictive value of polysomnographic findings. This study highlights the need for community-based studies of otherwise healthy children undergoing adenotonsillectomy to ensure that children at risk receive appropriate monitoring in an inpatient setting whereas those at lesser risk are not unnecessarily hospitalized postoperatively.
Heidi V. Connolly, MD, and Laura E. Tomaselli, MD, are pediatric sleep medicine physicians, and Margo K. McKenna Benoit, MD, is an otolaryngologist at the University of Rochester (N.Y.). They made these comments in a commentary that accompanied the published report ( J Clin Sleep Med. 2020 Jan 15;16[1]:3-4 ). They had no disclosures.
This well-conducted, retrospective, chart-review study adds important information to the published literature about risk stratification for children in a tertiary-referral population undergoing adenotonsillectomy. Their findings indicate that younger children remain at higher risk as well as those children with complex comorbid medical disease. They also show that children with severe sleep apnea or significant oxyhemoglobin desaturation are likewise at higher risk of postoperative respiratory compromise – emphasizing the need for preoperative polysomnography – particularly in a tertiary setting where many patients have medical comorbidities.
Despite the strengths of this study in assessing perioperative risk for respiratory compromise in a referral population with highly prevalent medical comorbidities, this study does not provide significant insight into the management of otherwise healthy children in a community setting who are undergoing adenotonsillectomy. This is important because a large number of adenotonsillectomies are performed outside of a tertiary-referral center and many of these children may not have undergone preoperative polysomnography to stratify risk. The utility of preoperative polysomnography in the evaluation of all children undergoing adenotonsillectomy remains controversial, with diverging recommendations from two major U.S. medical groups.
This study does not address the utility of polysomnography in community-based populations of otherwise healthy children. It is imperative to accurately ascertain risk so perioperative planning can ensure the safety of children at higher risk following adenotonsillectomy; however, there remains a paucity of studies assessing the cost-effectiveness as well as the positive and negative predictive value of polysomnographic findings. This study highlights the need for community-based studies of otherwise healthy children undergoing adenotonsillectomy to ensure that children at risk receive appropriate monitoring in an inpatient setting whereas those at lesser risk are not unnecessarily hospitalized postoperatively.
Heidi V. Connolly, MD, and Laura E. Tomaselli, MD, are pediatric sleep medicine physicians, and Margo K. McKenna Benoit, MD, is an otolaryngologist at the University of Rochester (N.Y.). They made these comments in a commentary that accompanied the published report ( J Clin Sleep Med. 2020 Jan 15;16[1]:3-4 ). They had no disclosures.
This well-conducted, retrospective, chart-review study adds important information to the published literature about risk stratification for children in a tertiary-referral population undergoing adenotonsillectomy. Their findings indicate that younger children remain at higher risk as well as those children with complex comorbid medical disease. They also show that children with severe sleep apnea or significant oxyhemoglobin desaturation are likewise at higher risk of postoperative respiratory compromise – emphasizing the need for preoperative polysomnography – particularly in a tertiary setting where many patients have medical comorbidities.
Despite the strengths of this study in assessing perioperative risk for respiratory compromise in a referral population with highly prevalent medical comorbidities, this study does not provide significant insight into the management of otherwise healthy children in a community setting who are undergoing adenotonsillectomy. This is important because a large number of adenotonsillectomies are performed outside of a tertiary-referral center and many of these children may not have undergone preoperative polysomnography to stratify risk. The utility of preoperative polysomnography in the evaluation of all children undergoing adenotonsillectomy remains controversial, with diverging recommendations from two major U.S. medical groups.
This study does not address the utility of polysomnography in community-based populations of otherwise healthy children. It is imperative to accurately ascertain risk so perioperative planning can ensure the safety of children at higher risk following adenotonsillectomy; however, there remains a paucity of studies assessing the cost-effectiveness as well as the positive and negative predictive value of polysomnographic findings. This study highlights the need for community-based studies of otherwise healthy children undergoing adenotonsillectomy to ensure that children at risk receive appropriate monitoring in an inpatient setting whereas those at lesser risk are not unnecessarily hospitalized postoperatively.
Heidi V. Connolly, MD, and Laura E. Tomaselli, MD, are pediatric sleep medicine physicians, and Margo K. McKenna Benoit, MD, is an otolaryngologist at the University of Rochester (N.Y.). They made these comments in a commentary that accompanied the published report ( J Clin Sleep Med. 2020 Jan 15;16[1]:3-4 ). They had no disclosures.
Underlying cardiac disease, airway anomalies, and younger age each independently boosted the risk of severe perioperative respiratory adverse events (PRAE) in children undergoing adenotonsillectomy to treat obstructive sleep apnea, in a review of 374 patients treated at a single Canadian tertiary-referral center.
In contrast, the analysis failed to show independent, significant effects from any assessed polysomnography or oximetry parameters on the rate of postoperative respiratory complications. The utility of preoperative polysomnography or oximetry for risk stratification is questionable for pediatric patients scheduled to adenotonsillectomy to treat obstructive sleep apnea, wrote Sherri L. Katz, MD, of the University of Ottawa, and associates in a recent report published in the Journal of Clinical Sleep Medicine, although they also added that making these assessments may be “unavoidable” because of their need for diagnosing obstructive sleep apnea and determining the need for surgery.
Despite this caveat, “overall our study results highlight the need to better define the complex interaction between comorbidities, age, nocturnal respiratory events, and gas exchange abnormalities in predicting risk for PRAE” after adenotonsillectomy, the researchers wrote. These findings “are consistent with existing clinical care guidelines,” and “cardiac and craniofacial conditions have been associated with risk of postoperative complications in other studies.”
The analysis used data collected from all children aged 0-18 years who underwent polysomnography assessment followed by adenotonsillectomy at one Canadian tertiary-referral center, Children’s Hospital of Eastern Ontario in Ottawa, during 2010-2016. Their median age was just over 6 years, and 39 patients (10%) were younger than 3 years at the time of their surgery. More than three-quarters of the patients, 286, had at least one identified comorbidity, and nearly half had at least two comorbidities. Polysomnography identified sleep-disordered breathing in 344 of the children (92%), and diagnosed obstructive sleep apnea in 256 (68%), including 148 (43% of the full cohort) with a severe apnea-hypopnea index.
Sixty-six of the children (18%) had at least one severe PRAE that required intervention. Specifically these were either oxygen desaturations requiring intervention or need for airway or ventilatory support with interventions such as jaw thrust, oral or nasal airway placement, bag and mask ventilation, or endotracheal intubation.
A multivariate regression analysis of the measured comorbidity, polysomnography, and oximetry parameters, as well as age, identified three factors that independently linked with a statistically significant increase in the rate of severe PRAE: airway anomaly, underlying cardiac disease, and young age. Patients with an airway anomaly had a 219% increased rate of PRAE, compared with those with no anomaly; patients with underlying cardiac disease had a 109% increased rate, compared with those without cardiac disease; and patients aged younger than 3 years had a 310% higher rate of PRAE, compared with the children aged 6 years or older, while children aged 3-5 years had a 121% higher rate of PRAE, compared with older children.
The study received no commercial funding. Dr. Katz has received honoraria for speaking from Biogen that had no relevance to the study.
SOURCE: Katz SL et al. J Clin Sleep Med. 2020 Jan 15;16(1):41-8.
Underlying cardiac disease, airway anomalies, and younger age each independently boosted the risk of severe perioperative respiratory adverse events (PRAE) in children undergoing adenotonsillectomy to treat obstructive sleep apnea, in a review of 374 patients treated at a single Canadian tertiary-referral center.
In contrast, the analysis failed to show independent, significant effects from any assessed polysomnography or oximetry parameters on the rate of postoperative respiratory complications. The utility of preoperative polysomnography or oximetry for risk stratification is questionable for pediatric patients scheduled to adenotonsillectomy to treat obstructive sleep apnea, wrote Sherri L. Katz, MD, of the University of Ottawa, and associates in a recent report published in the Journal of Clinical Sleep Medicine, although they also added that making these assessments may be “unavoidable” because of their need for diagnosing obstructive sleep apnea and determining the need for surgery.
Despite this caveat, “overall our study results highlight the need to better define the complex interaction between comorbidities, age, nocturnal respiratory events, and gas exchange abnormalities in predicting risk for PRAE” after adenotonsillectomy, the researchers wrote. These findings “are consistent with existing clinical care guidelines,” and “cardiac and craniofacial conditions have been associated with risk of postoperative complications in other studies.”
The analysis used data collected from all children aged 0-18 years who underwent polysomnography assessment followed by adenotonsillectomy at one Canadian tertiary-referral center, Children’s Hospital of Eastern Ontario in Ottawa, during 2010-2016. Their median age was just over 6 years, and 39 patients (10%) were younger than 3 years at the time of their surgery. More than three-quarters of the patients, 286, had at least one identified comorbidity, and nearly half had at least two comorbidities. Polysomnography identified sleep-disordered breathing in 344 of the children (92%), and diagnosed obstructive sleep apnea in 256 (68%), including 148 (43% of the full cohort) with a severe apnea-hypopnea index.
Sixty-six of the children (18%) had at least one severe PRAE that required intervention. Specifically these were either oxygen desaturations requiring intervention or need for airway or ventilatory support with interventions such as jaw thrust, oral or nasal airway placement, bag and mask ventilation, or endotracheal intubation.
A multivariate regression analysis of the measured comorbidity, polysomnography, and oximetry parameters, as well as age, identified three factors that independently linked with a statistically significant increase in the rate of severe PRAE: airway anomaly, underlying cardiac disease, and young age. Patients with an airway anomaly had a 219% increased rate of PRAE, compared with those with no anomaly; patients with underlying cardiac disease had a 109% increased rate, compared with those without cardiac disease; and patients aged younger than 3 years had a 310% higher rate of PRAE, compared with the children aged 6 years or older, while children aged 3-5 years had a 121% higher rate of PRAE, compared with older children.
The study received no commercial funding. Dr. Katz has received honoraria for speaking from Biogen that had no relevance to the study.
SOURCE: Katz SL et al. J Clin Sleep Med. 2020 Jan 15;16(1):41-8.
FROM THE JOURNAL OF CLINICAL SLEEP MEDICINE