User login
-
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]


Meet the FISH Bowl finalists
CHEST 2019 marked the inaugural FISH Bowl competition for attendees. Inspired by Shark Tank, our kinder, gentler, yet still competitive and cutting-edge FISH Bowl (Furthering Innovation and Science for Health) featured CHEST members disrupting our beliefs about how clinical care and education are performed. As health-care providers, they presented innovative ideas pertaining to education and clinical disease for pulmonary, critical care, and sleep medicine. Six finalists were chosen from dozens of submissions, and three emerged winners! In this new Meet the FISH Bowl Finalists series, CHEST introduces you to many of them – including winner Dr. Rachel Quaney.
Name: Rachel Quaney, MD
Institutional Affiliation: The Ohio State University
Position: Pulmonary and Critical Care Medicine Fellow
Title: Teaching Assessment Committee (TAC)
Brief Summary of Submission: Teaching Assessment Committee (TAC) is a novel approach to faculty feedback. We are modeling it after the success of the Clinical Competency Committees, but, in reverse, as fellows will give group-consensus-based feedback to faculty members.
Fellows will meet twice yearly with trained facilitators who help elicit constructive, nuanced feedback. The group setting ensures personal anonymity, which will serve to encourage more honest feedback. Then delivering this consensus-based information to program leadership and faculty members will hopefully provide helpful feedback regarding what is going well and what could be improved.
This pilot feasibility project is being employed at three fellowship programs this academic year. The goal will be to improve the feedback that faculty receive, while simultaneously increasing both faculty and fellow satisfaction with the process and the learning environment.
1. What inspired your innovation? More like who – and that would be the esteemed Dr. Gabe Bosslet of Indiana University. He brought the faculty perspective that attendings want better feedback. And, I supplied the fellow perspective—that even those of us who prioritize all things medical education often do a subpar job at providing effective feedback.
2. Who do you think can benefit most from it, and why? With some variation, almost all graduate medical education programs could benefit from the TAC method of faculty feedback. However, the most benefit would likely be seen in small programs or those that struggle with anonymity using current feedback methods.
3. What do you see as challenges to your innovation gaining widespread acceptance? How can they be overcome? I foresee two main challenges to implementation: time and buy-in. Fellows and residents are busy individuals with plenty on their plates, and this would require asking them for more time. This barrier could be solved by program and leadership buy-in or be exacerbated if it is lacking. If the process is endorsed by departmental and program leadership, this will provide credibility and ensure the necessary time is allotted.
4. What impact has winning FISH Bowl 2019 had on your vision for the innovation? The big picture vision I have for my innovation has not changed, but I am more acutely aware of the challenges and opportunities I will have to navigate, thanks to Drs. Morris, Niven, and Schulman. I am simultaneously more excited about this project but also feel the pressure to not disappoint!
5. How do you think your success at FISH Bowl 2019 will continue to impact your career overall in the months and years to come? It’s hard to imagine in what exact ways my career will be impacted, but I feel strongly that it will be positively influenced by this experience. I had the privilege of meeting a lot of individuals who feel passionate about medical education, both those established in our field and those at the beginning of their careers. These connections will likely lead to future collaborations and innovations.
CHEST 2019 marked the inaugural FISH Bowl competition for attendees. Inspired by Shark Tank, our kinder, gentler, yet still competitive and cutting-edge FISH Bowl (Furthering Innovation and Science for Health) featured CHEST members disrupting our beliefs about how clinical care and education are performed. As health-care providers, they presented innovative ideas pertaining to education and clinical disease for pulmonary, critical care, and sleep medicine. Six finalists were chosen from dozens of submissions, and three emerged winners! In this new Meet the FISH Bowl Finalists series, CHEST introduces you to many of them – including winner Dr. Rachel Quaney.
Name: Rachel Quaney, MD
Institutional Affiliation: The Ohio State University
Position: Pulmonary and Critical Care Medicine Fellow
Title: Teaching Assessment Committee (TAC)
Brief Summary of Submission: Teaching Assessment Committee (TAC) is a novel approach to faculty feedback. We are modeling it after the success of the Clinical Competency Committees, but, in reverse, as fellows will give group-consensus-based feedback to faculty members.
Fellows will meet twice yearly with trained facilitators who help elicit constructive, nuanced feedback. The group setting ensures personal anonymity, which will serve to encourage more honest feedback. Then delivering this consensus-based information to program leadership and faculty members will hopefully provide helpful feedback regarding what is going well and what could be improved.
This pilot feasibility project is being employed at three fellowship programs this academic year. The goal will be to improve the feedback that faculty receive, while simultaneously increasing both faculty and fellow satisfaction with the process and the learning environment.
1. What inspired your innovation? More like who – and that would be the esteemed Dr. Gabe Bosslet of Indiana University. He brought the faculty perspective that attendings want better feedback. And, I supplied the fellow perspective—that even those of us who prioritize all things medical education often do a subpar job at providing effective feedback.
2. Who do you think can benefit most from it, and why? With some variation, almost all graduate medical education programs could benefit from the TAC method of faculty feedback. However, the most benefit would likely be seen in small programs or those that struggle with anonymity using current feedback methods.
3. What do you see as challenges to your innovation gaining widespread acceptance? How can they be overcome? I foresee two main challenges to implementation: time and buy-in. Fellows and residents are busy individuals with plenty on their plates, and this would require asking them for more time. This barrier could be solved by program and leadership buy-in or be exacerbated if it is lacking. If the process is endorsed by departmental and program leadership, this will provide credibility and ensure the necessary time is allotted.
4. What impact has winning FISH Bowl 2019 had on your vision for the innovation? The big picture vision I have for my innovation has not changed, but I am more acutely aware of the challenges and opportunities I will have to navigate, thanks to Drs. Morris, Niven, and Schulman. I am simultaneously more excited about this project but also feel the pressure to not disappoint!
5. How do you think your success at FISH Bowl 2019 will continue to impact your career overall in the months and years to come? It’s hard to imagine in what exact ways my career will be impacted, but I feel strongly that it will be positively influenced by this experience. I had the privilege of meeting a lot of individuals who feel passionate about medical education, both those established in our field and those at the beginning of their careers. These connections will likely lead to future collaborations and innovations.
CHEST 2019 marked the inaugural FISH Bowl competition for attendees. Inspired by Shark Tank, our kinder, gentler, yet still competitive and cutting-edge FISH Bowl (Furthering Innovation and Science for Health) featured CHEST members disrupting our beliefs about how clinical care and education are performed. As health-care providers, they presented innovative ideas pertaining to education and clinical disease for pulmonary, critical care, and sleep medicine. Six finalists were chosen from dozens of submissions, and three emerged winners! In this new Meet the FISH Bowl Finalists series, CHEST introduces you to many of them – including winner Dr. Rachel Quaney.
Name: Rachel Quaney, MD
Institutional Affiliation: The Ohio State University
Position: Pulmonary and Critical Care Medicine Fellow
Title: Teaching Assessment Committee (TAC)
Brief Summary of Submission: Teaching Assessment Committee (TAC) is a novel approach to faculty feedback. We are modeling it after the success of the Clinical Competency Committees, but, in reverse, as fellows will give group-consensus-based feedback to faculty members.
Fellows will meet twice yearly with trained facilitators who help elicit constructive, nuanced feedback. The group setting ensures personal anonymity, which will serve to encourage more honest feedback. Then delivering this consensus-based information to program leadership and faculty members will hopefully provide helpful feedback regarding what is going well and what could be improved.
This pilot feasibility project is being employed at three fellowship programs this academic year. The goal will be to improve the feedback that faculty receive, while simultaneously increasing both faculty and fellow satisfaction with the process and the learning environment.
1. What inspired your innovation? More like who – and that would be the esteemed Dr. Gabe Bosslet of Indiana University. He brought the faculty perspective that attendings want better feedback. And, I supplied the fellow perspective—that even those of us who prioritize all things medical education often do a subpar job at providing effective feedback.
2. Who do you think can benefit most from it, and why? With some variation, almost all graduate medical education programs could benefit from the TAC method of faculty feedback. However, the most benefit would likely be seen in small programs or those that struggle with anonymity using current feedback methods.
3. What do you see as challenges to your innovation gaining widespread acceptance? How can they be overcome? I foresee two main challenges to implementation: time and buy-in. Fellows and residents are busy individuals with plenty on their plates, and this would require asking them for more time. This barrier could be solved by program and leadership buy-in or be exacerbated if it is lacking. If the process is endorsed by departmental and program leadership, this will provide credibility and ensure the necessary time is allotted.
4. What impact has winning FISH Bowl 2019 had on your vision for the innovation? The big picture vision I have for my innovation has not changed, but I am more acutely aware of the challenges and opportunities I will have to navigate, thanks to Drs. Morris, Niven, and Schulman. I am simultaneously more excited about this project but also feel the pressure to not disappoint!
5. How do you think your success at FISH Bowl 2019 will continue to impact your career overall in the months and years to come? It’s hard to imagine in what exact ways my career will be impacted, but I feel strongly that it will be positively influenced by this experience. I had the privilege of meeting a lot of individuals who feel passionate about medical education, both those established in our field and those at the beginning of their careers. These connections will likely lead to future collaborations and innovations.
COVID-19: Just a virus, right?
My first exposure to the notion of scarce resources was in medical school. I had to discuss the ethical principles behind the allocation of organs for transplantation, specifically livers and the required abstinence from alcohol ... but this was just an exercise, right?
A few years later, during residency, I heard the anecdotes from one of my internal medicine attendings about the time he spent in Europe as a visiting geriatrics fellow in the 1970s. The health-care districts in the region would be allotted an annual budget, and it was up to those districts how to best allocate those resources to meet, to the best of their abilities, the health-care needs of their population. He vividly recalled that a patient he cared for, an individual over 65 in need of renal replacement therapy for a reversible condition, who was not offered such therapy despite the clear benefit. There was a finite amount of resources, and those resources were thought to be better spent on public health measures like vaccination ... but that was on another continent and in another era, right?
I remember when I first heard of an outbreak of viral pneumonia in China in January of this year. As someone prone to anxiety, my first strategy was to put my head in the sand and wait it out. This strategy didn’t last very long – within a couple of weeks, there were confirmed cases in the United States. It was now apparent that this virus was not going to be contained. In an impressively short amount of time, SARS-CoV 2 has infected over 3.5 million individuals and killed almost a quarter million people worldwide. In the United States, we have seen almost 1.2 million cases and lost over 68 thousand lives. This pandemic has managed to devastate multiple countries, health care systems, and economies. It has also challenged every physician’s ideas of beneficence and justice ... but it’s just a virus, right?
Beneficence, the principle of medical ethics regarding acting in the patient’s best interest, had always seemed to me to be a no-brainer. Not like autonomy, which can get sticky, or justice, which I really had not had to consider much prior to 2020. Of course, I would always do what was best for my patient, I thought, why wouldn’t I?
Justice, the principle that deals with the distribution of scarce health-care resources, is the wrench that has been thrown into the beneficence works in the age of COVID-19. In a country and an era in which I had not dreamed we would ever have to think about how to support multiple people with one ventilator, we have had to do just that (“Joint Statement on Multiple Patients per Ventilator,” CHEST News, Mar 27, 2020). Things that I have taken for granted through all of my training are now worth their weight in gold—from sedative drips and inhalers down to videolaryngoscopy blades and face masks. I can’t just do what is best for my patient because sometimes what is best for my patient is not what is best for my next patient, what is best for my team, or even what is best for me and for my family. COVID-19 has reminded us of the uncomfortable truth that when contemplating justice, the patient in front of us is not the only person we have to consider.
Early on, before things in the United States had surged, I asked the twitter community what I thought would be a hypothetical question: “An employee needs to urgently help a COVID-19 patient. There is no appropriate PPE available due to shortage. What should happen?”
Like the idea of splitting ventilators, it was a thought I had never considered pre-COVID-19. Our instinct as physicians, especially as critical care physicians, is to intervene in emergency situations as quickly as possible. The extensive PPE required to manage COVID-19 patients has slowed that process, but, as many institutions are reaching the ends of their PPE stores, our safety is now placed at odds with that of our patient’s. To stay back violates what we feel is our duty to our patients, to go in violates our duty to ourselves, to our families, and to the rest of our patients. To care adequately for your patient is to put yourself at risk (and vice-versa), and this is a problem that I don’t think we have an answer for.
COVID-19 threatens many good and noble things, and what is worse, it directly puts them at odds with one another. They are paired sliding scales, where more of one means less of the other. If I have enough masks, it means my colleague probably doesn’t. If we have enough ventilators, it means another city doesn’t. If I get a break to be with my family, it means someone else is having to leave theirs to tend to patients who are sicker, lonelier, and more numerous than in any other time in recent memory.
And if these situations and resource limitations don’t provide enough moral injury for health-care workers, there are some specifics of humanity’s response to the pandemic that are exceptionally hurtful.
We as a country had notice, which was squandered. Instead of caution and preparation, we saw the powers that be make light of the serious situation most scientists and clinicians warned was coming. Instead of efforts to find or create PPE, we saw accusations against us of misuse and waste (“Trump comments about hospital mask thefts spark backlash from doctors,” Newsweek, March 30, 2020). Instead of support, we saw our altruism taken advantage of and used against us in unsafe and unfair situations. We have seen physicians in training and full-fledged attendings alike treated unfairly by their supervisors, instead of protected. Every instance of anti-science opinion or action from our friends and families that we once tolerated now feels like a personal affront, as these directly increase our risk and our immediate family’s risk of contracting the illness. We are being touted as heroes and angels, but really, we’re afraid—afraid of our patients, afraid of illness, afraid for our families, and afraid of jobs that we used to love. We don’t want to be praised; we just want to work our regular jobs safely and with adequate support.
I don’t know what health care looks like at the end of all of this. Relationships between physicians and health-care administrations were strained before the pandemic, to say the least. How can health-care workers just go back to business as usual, working for entities that were so ill-prepared, and, in many cases, calloused toward the concerns of their employees?
COVID-19 has revealed the fragility of our health-care system, our public health capabilities, and our economy. The pandemic has forced us to finally acknowledge something that has been true all along—our resources are finite, and tension exists between what is right and what is profitable, and between what is just and what is easy.
But it’s just a virus, right?
Dr. Fridenmaker is a Pulmonary and Critical Care Fellow at the University of Kentucky, Lexington.
My first exposure to the notion of scarce resources was in medical school. I had to discuss the ethical principles behind the allocation of organs for transplantation, specifically livers and the required abstinence from alcohol ... but this was just an exercise, right?
A few years later, during residency, I heard the anecdotes from one of my internal medicine attendings about the time he spent in Europe as a visiting geriatrics fellow in the 1970s. The health-care districts in the region would be allotted an annual budget, and it was up to those districts how to best allocate those resources to meet, to the best of their abilities, the health-care needs of their population. He vividly recalled that a patient he cared for, an individual over 65 in need of renal replacement therapy for a reversible condition, who was not offered such therapy despite the clear benefit. There was a finite amount of resources, and those resources were thought to be better spent on public health measures like vaccination ... but that was on another continent and in another era, right?
I remember when I first heard of an outbreak of viral pneumonia in China in January of this year. As someone prone to anxiety, my first strategy was to put my head in the sand and wait it out. This strategy didn’t last very long – within a couple of weeks, there were confirmed cases in the United States. It was now apparent that this virus was not going to be contained. In an impressively short amount of time, SARS-CoV 2 has infected over 3.5 million individuals and killed almost a quarter million people worldwide. In the United States, we have seen almost 1.2 million cases and lost over 68 thousand lives. This pandemic has managed to devastate multiple countries, health care systems, and economies. It has also challenged every physician’s ideas of beneficence and justice ... but it’s just a virus, right?
Beneficence, the principle of medical ethics regarding acting in the patient’s best interest, had always seemed to me to be a no-brainer. Not like autonomy, which can get sticky, or justice, which I really had not had to consider much prior to 2020. Of course, I would always do what was best for my patient, I thought, why wouldn’t I?
Justice, the principle that deals with the distribution of scarce health-care resources, is the wrench that has been thrown into the beneficence works in the age of COVID-19. In a country and an era in which I had not dreamed we would ever have to think about how to support multiple people with one ventilator, we have had to do just that (“Joint Statement on Multiple Patients per Ventilator,” CHEST News, Mar 27, 2020). Things that I have taken for granted through all of my training are now worth their weight in gold—from sedative drips and inhalers down to videolaryngoscopy blades and face masks. I can’t just do what is best for my patient because sometimes what is best for my patient is not what is best for my next patient, what is best for my team, or even what is best for me and for my family. COVID-19 has reminded us of the uncomfortable truth that when contemplating justice, the patient in front of us is not the only person we have to consider.
Early on, before things in the United States had surged, I asked the twitter community what I thought would be a hypothetical question: “An employee needs to urgently help a COVID-19 patient. There is no appropriate PPE available due to shortage. What should happen?”
Like the idea of splitting ventilators, it was a thought I had never considered pre-COVID-19. Our instinct as physicians, especially as critical care physicians, is to intervene in emergency situations as quickly as possible. The extensive PPE required to manage COVID-19 patients has slowed that process, but, as many institutions are reaching the ends of their PPE stores, our safety is now placed at odds with that of our patient’s. To stay back violates what we feel is our duty to our patients, to go in violates our duty to ourselves, to our families, and to the rest of our patients. To care adequately for your patient is to put yourself at risk (and vice-versa), and this is a problem that I don’t think we have an answer for.
COVID-19 threatens many good and noble things, and what is worse, it directly puts them at odds with one another. They are paired sliding scales, where more of one means less of the other. If I have enough masks, it means my colleague probably doesn’t. If we have enough ventilators, it means another city doesn’t. If I get a break to be with my family, it means someone else is having to leave theirs to tend to patients who are sicker, lonelier, and more numerous than in any other time in recent memory.
And if these situations and resource limitations don’t provide enough moral injury for health-care workers, there are some specifics of humanity’s response to the pandemic that are exceptionally hurtful.
We as a country had notice, which was squandered. Instead of caution and preparation, we saw the powers that be make light of the serious situation most scientists and clinicians warned was coming. Instead of efforts to find or create PPE, we saw accusations against us of misuse and waste (“Trump comments about hospital mask thefts spark backlash from doctors,” Newsweek, March 30, 2020). Instead of support, we saw our altruism taken advantage of and used against us in unsafe and unfair situations. We have seen physicians in training and full-fledged attendings alike treated unfairly by their supervisors, instead of protected. Every instance of anti-science opinion or action from our friends and families that we once tolerated now feels like a personal affront, as these directly increase our risk and our immediate family’s risk of contracting the illness. We are being touted as heroes and angels, but really, we’re afraid—afraid of our patients, afraid of illness, afraid for our families, and afraid of jobs that we used to love. We don’t want to be praised; we just want to work our regular jobs safely and with adequate support.
I don’t know what health care looks like at the end of all of this. Relationships between physicians and health-care administrations were strained before the pandemic, to say the least. How can health-care workers just go back to business as usual, working for entities that were so ill-prepared, and, in many cases, calloused toward the concerns of their employees?
COVID-19 has revealed the fragility of our health-care system, our public health capabilities, and our economy. The pandemic has forced us to finally acknowledge something that has been true all along—our resources are finite, and tension exists between what is right and what is profitable, and between what is just and what is easy.
But it’s just a virus, right?
Dr. Fridenmaker is a Pulmonary and Critical Care Fellow at the University of Kentucky, Lexington.
My first exposure to the notion of scarce resources was in medical school. I had to discuss the ethical principles behind the allocation of organs for transplantation, specifically livers and the required abstinence from alcohol ... but this was just an exercise, right?
A few years later, during residency, I heard the anecdotes from one of my internal medicine attendings about the time he spent in Europe as a visiting geriatrics fellow in the 1970s. The health-care districts in the region would be allotted an annual budget, and it was up to those districts how to best allocate those resources to meet, to the best of their abilities, the health-care needs of their population. He vividly recalled that a patient he cared for, an individual over 65 in need of renal replacement therapy for a reversible condition, who was not offered such therapy despite the clear benefit. There was a finite amount of resources, and those resources were thought to be better spent on public health measures like vaccination ... but that was on another continent and in another era, right?
I remember when I first heard of an outbreak of viral pneumonia in China in January of this year. As someone prone to anxiety, my first strategy was to put my head in the sand and wait it out. This strategy didn’t last very long – within a couple of weeks, there were confirmed cases in the United States. It was now apparent that this virus was not going to be contained. In an impressively short amount of time, SARS-CoV 2 has infected over 3.5 million individuals and killed almost a quarter million people worldwide. In the United States, we have seen almost 1.2 million cases and lost over 68 thousand lives. This pandemic has managed to devastate multiple countries, health care systems, and economies. It has also challenged every physician’s ideas of beneficence and justice ... but it’s just a virus, right?
Beneficence, the principle of medical ethics regarding acting in the patient’s best interest, had always seemed to me to be a no-brainer. Not like autonomy, which can get sticky, or justice, which I really had not had to consider much prior to 2020. Of course, I would always do what was best for my patient, I thought, why wouldn’t I?
Justice, the principle that deals with the distribution of scarce health-care resources, is the wrench that has been thrown into the beneficence works in the age of COVID-19. In a country and an era in which I had not dreamed we would ever have to think about how to support multiple people with one ventilator, we have had to do just that (“Joint Statement on Multiple Patients per Ventilator,” CHEST News, Mar 27, 2020). Things that I have taken for granted through all of my training are now worth their weight in gold—from sedative drips and inhalers down to videolaryngoscopy blades and face masks. I can’t just do what is best for my patient because sometimes what is best for my patient is not what is best for my next patient, what is best for my team, or even what is best for me and for my family. COVID-19 has reminded us of the uncomfortable truth that when contemplating justice, the patient in front of us is not the only person we have to consider.
Early on, before things in the United States had surged, I asked the twitter community what I thought would be a hypothetical question: “An employee needs to urgently help a COVID-19 patient. There is no appropriate PPE available due to shortage. What should happen?”
Like the idea of splitting ventilators, it was a thought I had never considered pre-COVID-19. Our instinct as physicians, especially as critical care physicians, is to intervene in emergency situations as quickly as possible. The extensive PPE required to manage COVID-19 patients has slowed that process, but, as many institutions are reaching the ends of their PPE stores, our safety is now placed at odds with that of our patient’s. To stay back violates what we feel is our duty to our patients, to go in violates our duty to ourselves, to our families, and to the rest of our patients. To care adequately for your patient is to put yourself at risk (and vice-versa), and this is a problem that I don’t think we have an answer for.
COVID-19 threatens many good and noble things, and what is worse, it directly puts them at odds with one another. They are paired sliding scales, where more of one means less of the other. If I have enough masks, it means my colleague probably doesn’t. If we have enough ventilators, it means another city doesn’t. If I get a break to be with my family, it means someone else is having to leave theirs to tend to patients who are sicker, lonelier, and more numerous than in any other time in recent memory.
And if these situations and resource limitations don’t provide enough moral injury for health-care workers, there are some specifics of humanity’s response to the pandemic that are exceptionally hurtful.
We as a country had notice, which was squandered. Instead of caution and preparation, we saw the powers that be make light of the serious situation most scientists and clinicians warned was coming. Instead of efforts to find or create PPE, we saw accusations against us of misuse and waste (“Trump comments about hospital mask thefts spark backlash from doctors,” Newsweek, March 30, 2020). Instead of support, we saw our altruism taken advantage of and used against us in unsafe and unfair situations. We have seen physicians in training and full-fledged attendings alike treated unfairly by their supervisors, instead of protected. Every instance of anti-science opinion or action from our friends and families that we once tolerated now feels like a personal affront, as these directly increase our risk and our immediate family’s risk of contracting the illness. We are being touted as heroes and angels, but really, we’re afraid—afraid of our patients, afraid of illness, afraid for our families, and afraid of jobs that we used to love. We don’t want to be praised; we just want to work our regular jobs safely and with adequate support.
I don’t know what health care looks like at the end of all of this. Relationships between physicians and health-care administrations were strained before the pandemic, to say the least. How can health-care workers just go back to business as usual, working for entities that were so ill-prepared, and, in many cases, calloused toward the concerns of their employees?
COVID-19 has revealed the fragility of our health-care system, our public health capabilities, and our economy. The pandemic has forced us to finally acknowledge something that has been true all along—our resources are finite, and tension exists between what is right and what is profitable, and between what is just and what is easy.
But it’s just a virus, right?
Dr. Fridenmaker is a Pulmonary and Critical Care Fellow at the University of Kentucky, Lexington.
COVID-19 and the cardiovascular system. Thrombotic events in COVID-19. Interprofessional collaboration.
Cardiovascular medicine and surgery
COVID-19 and the cardiovascular system
With the global outbreak of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) ongoing, there is increased awareness of the cardiovascular manifestations and implications of COVID-19. Approximately 20% of inpatients with COVID-19 have acute cardiac injury (defined as cardiac troponin elevation) (Shi S, et al. JAMA Cardiol. 2020 Mar 25. doi: 10.1001/jamacardio.2020.0950). Moreover, in one cohort, both acute cardiac injury and preexisting cardiovascular disease (CVD) were associated with COVID-19 hospital mortality: 69% with elevated troponin levels and underlying CVD vs 7.6% with neither (Guo T, et al. JAMA Cardiol. 2020 Mar 27. doi: 10.1001/jamacardio.2020.1017). Moreover, case reports suggest COVID-19 may present as myopericarditis, cardiomyopathy, acute on chronic decompensated heart failure, and acute coronary syndrome (Fried JA, et al. Circulation. 2020 Apr 3. doi: 10.1161/circulationaha.120.047164). Adding to this clinical variability, one case series suggests that electrocardiographic ST-segment elevation may not reliably identify obstructive coronary disease (Bangalore S, et al. N Engl J Med. 2020 Apr 17. doi: 10.1056/NEJMc2009020). Intriguingly, the angiotensin-converting enzyme 2 (ACE2) protein is the functional receptor for SARS-CoV-2 cell entry, and ACE2 is highly expressed in pulmonary and cardiac cells (Driggin E, et al. J Am Coll Cardiol. 2020;75[18]:2352). Given the central role of ACE2 and the renin-angiotensin-aldosterone (RAAS) system in cardiovascular pathophysiology and pharmacotherapy, RAAS modulation could have beneficial and/or detrimental effects with COVID-19 (Vaduganathan M, et al. N Engl J Med. 2020;382:1653). Available evidence and societal guidelines support continuing RAAS antagonists in patients per established clinical practice (Mancia G, et al. N Engl J Med. 2020 May 1. doi: 10.1056/NEJMoa2006923); (Mehra MR, et al. N Engl J Med. 2020 May 1. doi: 10.1056/NEJMoa2007621). A better understanding of the direct and indirect effect of SARS-CoV-2 on the cardiovascular system will require additional evidence.
Benjamin B. Kenigsberg, MD
Fellow-in-Training Steering Committee Member
Thrombotic events in COVID-19: Implications and evolving practice recommendations
A startling potential complication of infection with SARS-CoV2 has been the reported predisposition to thrombotic events. Mortality in COVID-19 patients is associated with notable increases in hemostatic parameters such as levels of
Vascular societies led by International Society on Thrombosis and Haemostasis (ISTH) have published consensus recommendations for guidance. If no contraindications exist, pharmacologic venous thromboembolism (VTE) prophylaxis with unfractionated heparin (UFH) or low-molecular-weight heparin (LMWH) is recommended for hospitalized patients with moderate or severe COVID-19 without disseminated intravascular coagulation (DIC). VTE prophylaxis should also be considered for patients with moderate or severe COVID-19 and in DIC but without overt bleeding. There is insufficient evidence to consider routine therapeutic or intermediate-dose parenteral anticoagulation with UFH or LMWH. Many institutions have developed protocols advising therapeutic-intensity anticoagulation when certain thresholds of
Saiprakash B. Venkateshiah, MD, FCCP,
Chair
Gabriela Magda, MD
Fellow-in-Training Steering Committee Member
Interprofessional Team
Quality of interprofessional collaboration in the medical intensive care unit: perceptions by caregivers
A recent study examining caregivers’ perceptions of team interactions and interprofessional collaborative practice (IPCP) behaviors offers new, exciting insights on the importance of interprofessional team functioning in the medical intensive care unit (MICU) (Chen DW, et al. J Gen Intern Med. 2018;33[10]:1708).
The Support Person Jefferson Teamwork Observation Guide (JTOG)TM survey was administered to 161 random caregivers of patients hospitalized in a single large urban academic medical center MICU between May 2016 and December 2016. The survey tool was designed to elicit the perceptions of caregivers regarding team functioning. Survey questions were directly mapped to the 2011 Interprofessional Education Collaborative (IPEC) Expert Panel core competencies for IPCP and divided into four domains (values/ethics; interprofessional communication; roles/responsibilities; teams/teamwork).
Also appended to the surveys were additional follow-up questions that addressed the overall satisfaction with the team and general attitudes regarding the importance of interprofessional team-based care. Caregivers agreed on the importance of health-care professionals working together as a team to provide patient care (3.97/4.00 Likert scale 4 = extremely important). Caregivers expressed satisfaction with the MICU team (3.74/4.00). Furthermore, caregivers agreed that the MICU team demonstrated competencies in all four domains of IPCP: values/ethics (3.55/4.00), interprofessional communication (3.58/4.00), roles/responsibilities (3.61/4.00), and teams/teamwork (3.64/4.00). Caregivers felt the MICU team provided patient/family-centered care (sub-competency 3.58/4.00). Notably, the overall caregiver survey scores detailing how well each MICU team functioned were positively correlated to overall satisfaction with the MICU team (r = 0.596 P < .01).
Limitations of the study included:
1. The sample is from a single institution, and perceptions of caregivers cannot be applied to all populations.
2. No information regarding patient, such as diagnosis, was obtained.
3. Caregivers satisfied with care might be more likely to participate.
4. No distinction was made between data collected from caregivers surveyed in the resident-fellow staffed MICU vs NP-staffed MICU.
It has been described that ineffective teamwork and team communication in health care settings are associated with increased patient harm and sentinel events (Kohn LT et al. Washington (DC): National Academies Press(US); 2000); (Page A, Washington (DC): National Academies Press (US); (The Joint Commission. Sentinel Event Alert 2008, 40); (Brennan TA, et al. N Engl J Med. 1991;324:370). Cultural differences between members of the health-care team and established hierarchies of control have been identified as barriers to communication and teamwork in ICUs (Alexanian JA, et al. J Crit Care Med. 2015;43[9]:1880); (Manias E, Street A. Int J Nurs Stud. 2001;38[2]:129).
Overall, the findings from this study emphasize the importance of interprofessional communication and teamwork in the MICU and delivery of patient/family-centered care from the caregivers’ perspective. The unique insight into caregivers’ perspectives on specific team behaviors may be the basis for future quality improvement initiatives.
Justin K. Lui, MD,
Mary Jo Farmer, MD, PhD, FCCP
Kristina E. Ramirez, RRT, MPH, FCCP
Cardiovascular medicine and surgery
COVID-19 and the cardiovascular system
With the global outbreak of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) ongoing, there is increased awareness of the cardiovascular manifestations and implications of COVID-19. Approximately 20% of inpatients with COVID-19 have acute cardiac injury (defined as cardiac troponin elevation) (Shi S, et al. JAMA Cardiol. 2020 Mar 25. doi: 10.1001/jamacardio.2020.0950). Moreover, in one cohort, both acute cardiac injury and preexisting cardiovascular disease (CVD) were associated with COVID-19 hospital mortality: 69% with elevated troponin levels and underlying CVD vs 7.6% with neither (Guo T, et al. JAMA Cardiol. 2020 Mar 27. doi: 10.1001/jamacardio.2020.1017). Moreover, case reports suggest COVID-19 may present as myopericarditis, cardiomyopathy, acute on chronic decompensated heart failure, and acute coronary syndrome (Fried JA, et al. Circulation. 2020 Apr 3. doi: 10.1161/circulationaha.120.047164). Adding to this clinical variability, one case series suggests that electrocardiographic ST-segment elevation may not reliably identify obstructive coronary disease (Bangalore S, et al. N Engl J Med. 2020 Apr 17. doi: 10.1056/NEJMc2009020). Intriguingly, the angiotensin-converting enzyme 2 (ACE2) protein is the functional receptor for SARS-CoV-2 cell entry, and ACE2 is highly expressed in pulmonary and cardiac cells (Driggin E, et al. J Am Coll Cardiol. 2020;75[18]:2352). Given the central role of ACE2 and the renin-angiotensin-aldosterone (RAAS) system in cardiovascular pathophysiology and pharmacotherapy, RAAS modulation could have beneficial and/or detrimental effects with COVID-19 (Vaduganathan M, et al. N Engl J Med. 2020;382:1653). Available evidence and societal guidelines support continuing RAAS antagonists in patients per established clinical practice (Mancia G, et al. N Engl J Med. 2020 May 1. doi: 10.1056/NEJMoa2006923); (Mehra MR, et al. N Engl J Med. 2020 May 1. doi: 10.1056/NEJMoa2007621). A better understanding of the direct and indirect effect of SARS-CoV-2 on the cardiovascular system will require additional evidence.
Benjamin B. Kenigsberg, MD
Fellow-in-Training Steering Committee Member
Thrombotic events in COVID-19: Implications and evolving practice recommendations
A startling potential complication of infection with SARS-CoV2 has been the reported predisposition to thrombotic events. Mortality in COVID-19 patients is associated with notable increases in hemostatic parameters such as levels of
Vascular societies led by International Society on Thrombosis and Haemostasis (ISTH) have published consensus recommendations for guidance. If no contraindications exist, pharmacologic venous thromboembolism (VTE) prophylaxis with unfractionated heparin (UFH) or low-molecular-weight heparin (LMWH) is recommended for hospitalized patients with moderate or severe COVID-19 without disseminated intravascular coagulation (DIC). VTE prophylaxis should also be considered for patients with moderate or severe COVID-19 and in DIC but without overt bleeding. There is insufficient evidence to consider routine therapeutic or intermediate-dose parenteral anticoagulation with UFH or LMWH. Many institutions have developed protocols advising therapeutic-intensity anticoagulation when certain thresholds of
Saiprakash B. Venkateshiah, MD, FCCP,
Chair
Gabriela Magda, MD
Fellow-in-Training Steering Committee Member
Interprofessional Team
Quality of interprofessional collaboration in the medical intensive care unit: perceptions by caregivers
A recent study examining caregivers’ perceptions of team interactions and interprofessional collaborative practice (IPCP) behaviors offers new, exciting insights on the importance of interprofessional team functioning in the medical intensive care unit (MICU) (Chen DW, et al. J Gen Intern Med. 2018;33[10]:1708).
The Support Person Jefferson Teamwork Observation Guide (JTOG)TM survey was administered to 161 random caregivers of patients hospitalized in a single large urban academic medical center MICU between May 2016 and December 2016. The survey tool was designed to elicit the perceptions of caregivers regarding team functioning. Survey questions were directly mapped to the 2011 Interprofessional Education Collaborative (IPEC) Expert Panel core competencies for IPCP and divided into four domains (values/ethics; interprofessional communication; roles/responsibilities; teams/teamwork).
Also appended to the surveys were additional follow-up questions that addressed the overall satisfaction with the team and general attitudes regarding the importance of interprofessional team-based care. Caregivers agreed on the importance of health-care professionals working together as a team to provide patient care (3.97/4.00 Likert scale 4 = extremely important). Caregivers expressed satisfaction with the MICU team (3.74/4.00). Furthermore, caregivers agreed that the MICU team demonstrated competencies in all four domains of IPCP: values/ethics (3.55/4.00), interprofessional communication (3.58/4.00), roles/responsibilities (3.61/4.00), and teams/teamwork (3.64/4.00). Caregivers felt the MICU team provided patient/family-centered care (sub-competency 3.58/4.00). Notably, the overall caregiver survey scores detailing how well each MICU team functioned were positively correlated to overall satisfaction with the MICU team (r = 0.596 P < .01).
Limitations of the study included:
1. The sample is from a single institution, and perceptions of caregivers cannot be applied to all populations.
2. No information regarding patient, such as diagnosis, was obtained.
3. Caregivers satisfied with care might be more likely to participate.
4. No distinction was made between data collected from caregivers surveyed in the resident-fellow staffed MICU vs NP-staffed MICU.
It has been described that ineffective teamwork and team communication in health care settings are associated with increased patient harm and sentinel events (Kohn LT et al. Washington (DC): National Academies Press(US); 2000); (Page A, Washington (DC): National Academies Press (US); (The Joint Commission. Sentinel Event Alert 2008, 40); (Brennan TA, et al. N Engl J Med. 1991;324:370). Cultural differences between members of the health-care team and established hierarchies of control have been identified as barriers to communication and teamwork in ICUs (Alexanian JA, et al. J Crit Care Med. 2015;43[9]:1880); (Manias E, Street A. Int J Nurs Stud. 2001;38[2]:129).
Overall, the findings from this study emphasize the importance of interprofessional communication and teamwork in the MICU and delivery of patient/family-centered care from the caregivers’ perspective. The unique insight into caregivers’ perspectives on specific team behaviors may be the basis for future quality improvement initiatives.
Justin K. Lui, MD,
Mary Jo Farmer, MD, PhD, FCCP
Kristina E. Ramirez, RRT, MPH, FCCP
Cardiovascular medicine and surgery
COVID-19 and the cardiovascular system
With the global outbreak of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) ongoing, there is increased awareness of the cardiovascular manifestations and implications of COVID-19. Approximately 20% of inpatients with COVID-19 have acute cardiac injury (defined as cardiac troponin elevation) (Shi S, et al. JAMA Cardiol. 2020 Mar 25. doi: 10.1001/jamacardio.2020.0950). Moreover, in one cohort, both acute cardiac injury and preexisting cardiovascular disease (CVD) were associated with COVID-19 hospital mortality: 69% with elevated troponin levels and underlying CVD vs 7.6% with neither (Guo T, et al. JAMA Cardiol. 2020 Mar 27. doi: 10.1001/jamacardio.2020.1017). Moreover, case reports suggest COVID-19 may present as myopericarditis, cardiomyopathy, acute on chronic decompensated heart failure, and acute coronary syndrome (Fried JA, et al. Circulation. 2020 Apr 3. doi: 10.1161/circulationaha.120.047164). Adding to this clinical variability, one case series suggests that electrocardiographic ST-segment elevation may not reliably identify obstructive coronary disease (Bangalore S, et al. N Engl J Med. 2020 Apr 17. doi: 10.1056/NEJMc2009020). Intriguingly, the angiotensin-converting enzyme 2 (ACE2) protein is the functional receptor for SARS-CoV-2 cell entry, and ACE2 is highly expressed in pulmonary and cardiac cells (Driggin E, et al. J Am Coll Cardiol. 2020;75[18]:2352). Given the central role of ACE2 and the renin-angiotensin-aldosterone (RAAS) system in cardiovascular pathophysiology and pharmacotherapy, RAAS modulation could have beneficial and/or detrimental effects with COVID-19 (Vaduganathan M, et al. N Engl J Med. 2020;382:1653). Available evidence and societal guidelines support continuing RAAS antagonists in patients per established clinical practice (Mancia G, et al. N Engl J Med. 2020 May 1. doi: 10.1056/NEJMoa2006923); (Mehra MR, et al. N Engl J Med. 2020 May 1. doi: 10.1056/NEJMoa2007621). A better understanding of the direct and indirect effect of SARS-CoV-2 on the cardiovascular system will require additional evidence.
Benjamin B. Kenigsberg, MD
Fellow-in-Training Steering Committee Member
Thrombotic events in COVID-19: Implications and evolving practice recommendations
A startling potential complication of infection with SARS-CoV2 has been the reported predisposition to thrombotic events. Mortality in COVID-19 patients is associated with notable increases in hemostatic parameters such as levels of
Vascular societies led by International Society on Thrombosis and Haemostasis (ISTH) have published consensus recommendations for guidance. If no contraindications exist, pharmacologic venous thromboembolism (VTE) prophylaxis with unfractionated heparin (UFH) or low-molecular-weight heparin (LMWH) is recommended for hospitalized patients with moderate or severe COVID-19 without disseminated intravascular coagulation (DIC). VTE prophylaxis should also be considered for patients with moderate or severe COVID-19 and in DIC but without overt bleeding. There is insufficient evidence to consider routine therapeutic or intermediate-dose parenteral anticoagulation with UFH or LMWH. Many institutions have developed protocols advising therapeutic-intensity anticoagulation when certain thresholds of
Saiprakash B. Venkateshiah, MD, FCCP,
Chair
Gabriela Magda, MD
Fellow-in-Training Steering Committee Member
Interprofessional Team
Quality of interprofessional collaboration in the medical intensive care unit: perceptions by caregivers
A recent study examining caregivers’ perceptions of team interactions and interprofessional collaborative practice (IPCP) behaviors offers new, exciting insights on the importance of interprofessional team functioning in the medical intensive care unit (MICU) (Chen DW, et al. J Gen Intern Med. 2018;33[10]:1708).
The Support Person Jefferson Teamwork Observation Guide (JTOG)TM survey was administered to 161 random caregivers of patients hospitalized in a single large urban academic medical center MICU between May 2016 and December 2016. The survey tool was designed to elicit the perceptions of caregivers regarding team functioning. Survey questions were directly mapped to the 2011 Interprofessional Education Collaborative (IPEC) Expert Panel core competencies for IPCP and divided into four domains (values/ethics; interprofessional communication; roles/responsibilities; teams/teamwork).
Also appended to the surveys were additional follow-up questions that addressed the overall satisfaction with the team and general attitudes regarding the importance of interprofessional team-based care. Caregivers agreed on the importance of health-care professionals working together as a team to provide patient care (3.97/4.00 Likert scale 4 = extremely important). Caregivers expressed satisfaction with the MICU team (3.74/4.00). Furthermore, caregivers agreed that the MICU team demonstrated competencies in all four domains of IPCP: values/ethics (3.55/4.00), interprofessional communication (3.58/4.00), roles/responsibilities (3.61/4.00), and teams/teamwork (3.64/4.00). Caregivers felt the MICU team provided patient/family-centered care (sub-competency 3.58/4.00). Notably, the overall caregiver survey scores detailing how well each MICU team functioned were positively correlated to overall satisfaction with the MICU team (r = 0.596 P < .01).
Limitations of the study included:
1. The sample is from a single institution, and perceptions of caregivers cannot be applied to all populations.
2. No information regarding patient, such as diagnosis, was obtained.
3. Caregivers satisfied with care might be more likely to participate.
4. No distinction was made between data collected from caregivers surveyed in the resident-fellow staffed MICU vs NP-staffed MICU.
It has been described that ineffective teamwork and team communication in health care settings are associated with increased patient harm and sentinel events (Kohn LT et al. Washington (DC): National Academies Press(US); 2000); (Page A, Washington (DC): National Academies Press (US); (The Joint Commission. Sentinel Event Alert 2008, 40); (Brennan TA, et al. N Engl J Med. 1991;324:370). Cultural differences between members of the health-care team and established hierarchies of control have been identified as barriers to communication and teamwork in ICUs (Alexanian JA, et al. J Crit Care Med. 2015;43[9]:1880); (Manias E, Street A. Int J Nurs Stud. 2001;38[2]:129).
Overall, the findings from this study emphasize the importance of interprofessional communication and teamwork in the MICU and delivery of patient/family-centered care from the caregivers’ perspective. The unique insight into caregivers’ perspectives on specific team behaviors may be the basis for future quality improvement initiatives.
Justin K. Lui, MD,
Mary Jo Farmer, MD, PhD, FCCP
Kristina E. Ramirez, RRT, MPH, FCCP
This month in the journal CHEST®
Editor’s picks
Preparing for the COVID-19 Pandemic: Our Experience in New York.By Dr. H. Zubair, et al.
The Utility of Electronic Inhaler Monitoring in COPD Management: Promises and Challenges.By Dr. A. H. Attaway, et al.
Patterns of Use of Adjunctive Therapies in Patients With Early Moderate-Severe Acute Respiratory Distress Syndrome: Insights From the LUNG SAFE Study.By Dr. A. Duggal, et al.
Clinical Evaluation of Deployed Military Personnel with Chronic Respiratory Symptoms: STAMPEDE III (Study of Active Duty Military for Pulmonary Disease Related to Environmental Deployment Exposures). By Dr. M. J. Morris, et al.
Editor’s picks
Editor’s picks
Preparing for the COVID-19 Pandemic: Our Experience in New York.By Dr. H. Zubair, et al.
The Utility of Electronic Inhaler Monitoring in COPD Management: Promises and Challenges.By Dr. A. H. Attaway, et al.
Patterns of Use of Adjunctive Therapies in Patients With Early Moderate-Severe Acute Respiratory Distress Syndrome: Insights From the LUNG SAFE Study.By Dr. A. Duggal, et al.
Clinical Evaluation of Deployed Military Personnel with Chronic Respiratory Symptoms: STAMPEDE III (Study of Active Duty Military for Pulmonary Disease Related to Environmental Deployment Exposures). By Dr. M. J. Morris, et al.
Preparing for the COVID-19 Pandemic: Our Experience in New York.By Dr. H. Zubair, et al.
The Utility of Electronic Inhaler Monitoring in COPD Management: Promises and Challenges.By Dr. A. H. Attaway, et al.
Patterns of Use of Adjunctive Therapies in Patients With Early Moderate-Severe Acute Respiratory Distress Syndrome: Insights From the LUNG SAFE Study.By Dr. A. Duggal, et al.
Clinical Evaluation of Deployed Military Personnel with Chronic Respiratory Symptoms: STAMPEDE III (Study of Active Duty Military for Pulmonary Disease Related to Environmental Deployment Exposures). By Dr. M. J. Morris, et al.
Evolving treatment of cystic fibrosis: Path toward a normal lifespan
Cystic fibrosis (CF) is an autosomal recessive disorder affecting thousands of people worldwide. When this genetic disease was first discovered in the first half of the 20th century, the median survival was approximately 5 years of age. Since then, median survival for patients with CF has steadily improved. Today, it is 47.4 years based on Cystic Fibrosis Foundation registry data from 2018. Patients with CF are living longer and staying healthier; the discussion to follow is how patients, researchers, and the CF Foundation reached this point.
In 1938, pediatrician and pathologist Dorothy Anderson observed on the autopsies of children thought to have celiac disease pancreatic lesions she termed “cystic fibrosis of the pancreas.” In addition to the abnormal pancreas, she noted abnormal lungs filled with mucus that obstructed the airways.
Paul Di Sant’Agnese recognized during a heatwave in late 1948 that children with CF were routinely being diagnosed with heatstroke and dehydration. This helped lead to the discovery that these children had elevated salt content in their sweat, paving the way for the development of the sweat chloride test in 1959 by Gibson and Cooke. Not only did Dr. Di Sant’Agnese recognize excess salt in the sweat of patients with CF, but with the help of several concerned parents of children with CF, he established the Cystic Fibrosis Foundation in 1955. The Foundation helped organize the care center model over the next decades, increasing from 30 care centers in 1962 to over 100 in 1978. The care center model also developed a patient registry to track patient care longitudinally.
In June 1989, Francis Collins and Lap-Chee Tsui discovered the location of the CF transmembrane conductance regulator (CFTR) protein using a novel technique called chromosome jumping (Rommens JM, et al. Science. 1989;245[4922]1059). The discovery was a breakthrough in basic science research, but it would take 3 more decades before this discovery could be translated into a medication that could be used by most patients for everyday care.
In the early 1990s, when median survival for patients with CF was 29 years of age, the CF Foundation and Genentech, Inc., coordinated a 24-week multicenter double-blind randomized control trial (RCT) for a new inhaled medication that digested the extracellular DNA from the neutrophils that accumulated in the airways of patients with CF. Inhaled recombinant human DNase in these patients reduced the risk of pulmonary exacerbations and also had a small improvement in pulmonary function in the group compared with the placebo group (Fuchs H, et al. N Engl J Med. 1994;331:637). Five years later, another double-blind RCT demonstrated that inhaled tobramycin in patients with CF whose disease was colonized with Pseudomonas aeruginosa improved pulmonary function and reduced the risk of hospitalizations (Ramsey B, et al. N Engl J Med. 1999;340:23). In 2006, the use of hypertonic saline solution in patients with CF decreased the overall pulmonary exacerbation rate (Elkins MR, et al. N Engl J Med. 2006;354:229). The combination of these inhaled medications, along with inhaled aztreonam, formed the backbone of inhalation therapy for CF care today.
In 1998, even with the ongoing development and approval of new CF medications by the pharmaceutical industry, Robert Beall, CEO of the CF Foundation, realized that he needed to challenge the current drug development paradigm. Instead of trying to convince companies to develop CF medications, he started a concept called venture philanthropy. This concept entailed the CF Foundation financially investing in pharmaceutical companies’ development of new medications. The Foundation first invested in a small company named Aurora Biosciences (known today as Vertex Pharmaceuticals) in 2000. Aurora Biosciences specialized in high throughput screening. This process uses a unique technology allowing one to test the therapeutic reaction of airway cells to thousands of chemical compounds in a single day, instead of using the traditional process of tediously pipetting compounds one by one. Today, the CF Foundation has invested millions of dollars into bioscience research to advance CF care.
In 2011, the results of a study were published in which a small molecule altered defective CFTR protein in patients with CF with the CFTR mutation G551D, thus improving chloride transport at the airway surface. In the original study, after 24 weeks of therapy receiving the medication known as ivacaftor, predicted FEV1 in patients with CF improved 10.6%, and the patients were 55% less likely to have a pulmonary exacerbation compared with those receiving a placebo. This breakthrough provided patients with CF the first medication that could correct the CFTR at the source of the problem (Ramsey BW, et al. N Engl J Med. 2011;365:1663). Ivacaftor was approved by the US FDA in 2012.
Ivacaftor provided proof of concept that using small molecules could improve CFTR function. Ivacaftor was only beneficial to a small percentage of patients and was not effective in patients with CF who had either 1 or 2 F508del CFTR mutations. In 2015, patients with CF with F508del homozygous treated with a combination therapy of lumacaftor/ivacaftor had predicted FEV1% improved 2.6% to 4.0%. More importantly, there was a significant reduction in the number of pulmonary exacerbations per year compared with placebo. Unexpectedly, some of the patients experienced bronchoconstriction while receiving lumacaftor/ivacaftor (Wainwright CE, et al. N Engl J Med. 2015; 373:220). The problem was recognized, and a new small molecule to improve the processing and trafficking of CFTR called tezacaftor was developed. The combination of tezacaftor/ivacaftor in patients with CF who were F508del homozygous demonstrated a similar reduction in pulmonary exacerbations, an absolute improvement of predicted FEV1 of 4%, and no increased respiratory symptoms compared with the placebo arm (Taylor-Cousar JL, et al. N Engl J Med. 2017;377[21]2013).
CFTR modulators were a major breakthrough for patients with CF, but the efficacy of these therapies was dependent on the patients’ genotype and ranged from mildly to moderately effective. Unfortunately, these therapies were ineffective for the patients who were delta 508 heterozygotes. Starting in the summer of 2018, VX 445-tezacaftor-ivacaftor (ETI) was compared with placebo in patients with CF who were 1 copy of F508del and a second CFTR mutation that has minimal function. The study found an absolute improvement in predicted FEV1 of 14.3% and a 63% reduction in exacerbations at 24 weeks compared with placebo (Middleton PG, et al. N Engl J Med. 2019;381:1809). In late 2019, based on these data, ETI was approved by the FDA for all patients with CF who were F508del heterozygous. This innovation provided effective therapy to 90% of the CF population.
With the discovery of many highly effective therapies beneficial in most patients, the CF Foundation started a program called Path to a Cure to find therapies for the 10% of patients with CF who were not candidates for ETI or other CFTR modulators. This program looks to develop novel methods to restore CFTR protein function and repair or replace the CFTR protein via gene editing or gene transfer. This process creates many challenges that are quite complex, but patients, researchers, physicians, and CF Foundation will not stop working until CF stands for CURE FOUND.
Today, patients with CF are living longer, and many are eligible or have already started ETI therapy. This medication and the many others being developed will hopefully lead to patients with CF living a normal lifespan in the near future.
Dr. Finklea is Assistant Professor of Medicine, Division of Pulmonary and Critical Care, University of Texas Southwestern, Dallas, Texas. Dr. Finklea receives grant support from the Cystic Fibrosis Foundation.
Cystic fibrosis (CF) is an autosomal recessive disorder affecting thousands of people worldwide. When this genetic disease was first discovered in the first half of the 20th century, the median survival was approximately 5 years of age. Since then, median survival for patients with CF has steadily improved. Today, it is 47.4 years based on Cystic Fibrosis Foundation registry data from 2018. Patients with CF are living longer and staying healthier; the discussion to follow is how patients, researchers, and the CF Foundation reached this point.
In 1938, pediatrician and pathologist Dorothy Anderson observed on the autopsies of children thought to have celiac disease pancreatic lesions she termed “cystic fibrosis of the pancreas.” In addition to the abnormal pancreas, she noted abnormal lungs filled with mucus that obstructed the airways.
Paul Di Sant’Agnese recognized during a heatwave in late 1948 that children with CF were routinely being diagnosed with heatstroke and dehydration. This helped lead to the discovery that these children had elevated salt content in their sweat, paving the way for the development of the sweat chloride test in 1959 by Gibson and Cooke. Not only did Dr. Di Sant’Agnese recognize excess salt in the sweat of patients with CF, but with the help of several concerned parents of children with CF, he established the Cystic Fibrosis Foundation in 1955. The Foundation helped organize the care center model over the next decades, increasing from 30 care centers in 1962 to over 100 in 1978. The care center model also developed a patient registry to track patient care longitudinally.
In June 1989, Francis Collins and Lap-Chee Tsui discovered the location of the CF transmembrane conductance regulator (CFTR) protein using a novel technique called chromosome jumping (Rommens JM, et al. Science. 1989;245[4922]1059). The discovery was a breakthrough in basic science research, but it would take 3 more decades before this discovery could be translated into a medication that could be used by most patients for everyday care.
In the early 1990s, when median survival for patients with CF was 29 years of age, the CF Foundation and Genentech, Inc., coordinated a 24-week multicenter double-blind randomized control trial (RCT) for a new inhaled medication that digested the extracellular DNA from the neutrophils that accumulated in the airways of patients with CF. Inhaled recombinant human DNase in these patients reduced the risk of pulmonary exacerbations and also had a small improvement in pulmonary function in the group compared with the placebo group (Fuchs H, et al. N Engl J Med. 1994;331:637). Five years later, another double-blind RCT demonstrated that inhaled tobramycin in patients with CF whose disease was colonized with Pseudomonas aeruginosa improved pulmonary function and reduced the risk of hospitalizations (Ramsey B, et al. N Engl J Med. 1999;340:23). In 2006, the use of hypertonic saline solution in patients with CF decreased the overall pulmonary exacerbation rate (Elkins MR, et al. N Engl J Med. 2006;354:229). The combination of these inhaled medications, along with inhaled aztreonam, formed the backbone of inhalation therapy for CF care today.
In 1998, even with the ongoing development and approval of new CF medications by the pharmaceutical industry, Robert Beall, CEO of the CF Foundation, realized that he needed to challenge the current drug development paradigm. Instead of trying to convince companies to develop CF medications, he started a concept called venture philanthropy. This concept entailed the CF Foundation financially investing in pharmaceutical companies’ development of new medications. The Foundation first invested in a small company named Aurora Biosciences (known today as Vertex Pharmaceuticals) in 2000. Aurora Biosciences specialized in high throughput screening. This process uses a unique technology allowing one to test the therapeutic reaction of airway cells to thousands of chemical compounds in a single day, instead of using the traditional process of tediously pipetting compounds one by one. Today, the CF Foundation has invested millions of dollars into bioscience research to advance CF care.
In 2011, the results of a study were published in which a small molecule altered defective CFTR protein in patients with CF with the CFTR mutation G551D, thus improving chloride transport at the airway surface. In the original study, after 24 weeks of therapy receiving the medication known as ivacaftor, predicted FEV1 in patients with CF improved 10.6%, and the patients were 55% less likely to have a pulmonary exacerbation compared with those receiving a placebo. This breakthrough provided patients with CF the first medication that could correct the CFTR at the source of the problem (Ramsey BW, et al. N Engl J Med. 2011;365:1663). Ivacaftor was approved by the US FDA in 2012.
Ivacaftor provided proof of concept that using small molecules could improve CFTR function. Ivacaftor was only beneficial to a small percentage of patients and was not effective in patients with CF who had either 1 or 2 F508del CFTR mutations. In 2015, patients with CF with F508del homozygous treated with a combination therapy of lumacaftor/ivacaftor had predicted FEV1% improved 2.6% to 4.0%. More importantly, there was a significant reduction in the number of pulmonary exacerbations per year compared with placebo. Unexpectedly, some of the patients experienced bronchoconstriction while receiving lumacaftor/ivacaftor (Wainwright CE, et al. N Engl J Med. 2015; 373:220). The problem was recognized, and a new small molecule to improve the processing and trafficking of CFTR called tezacaftor was developed. The combination of tezacaftor/ivacaftor in patients with CF who were F508del homozygous demonstrated a similar reduction in pulmonary exacerbations, an absolute improvement of predicted FEV1 of 4%, and no increased respiratory symptoms compared with the placebo arm (Taylor-Cousar JL, et al. N Engl J Med. 2017;377[21]2013).
CFTR modulators were a major breakthrough for patients with CF, but the efficacy of these therapies was dependent on the patients’ genotype and ranged from mildly to moderately effective. Unfortunately, these therapies were ineffective for the patients who were delta 508 heterozygotes. Starting in the summer of 2018, VX 445-tezacaftor-ivacaftor (ETI) was compared with placebo in patients with CF who were 1 copy of F508del and a second CFTR mutation that has minimal function. The study found an absolute improvement in predicted FEV1 of 14.3% and a 63% reduction in exacerbations at 24 weeks compared with placebo (Middleton PG, et al. N Engl J Med. 2019;381:1809). In late 2019, based on these data, ETI was approved by the FDA for all patients with CF who were F508del heterozygous. This innovation provided effective therapy to 90% of the CF population.
With the discovery of many highly effective therapies beneficial in most patients, the CF Foundation started a program called Path to a Cure to find therapies for the 10% of patients with CF who were not candidates for ETI or other CFTR modulators. This program looks to develop novel methods to restore CFTR protein function and repair or replace the CFTR protein via gene editing or gene transfer. This process creates many challenges that are quite complex, but patients, researchers, physicians, and CF Foundation will not stop working until CF stands for CURE FOUND.
Today, patients with CF are living longer, and many are eligible or have already started ETI therapy. This medication and the many others being developed will hopefully lead to patients with CF living a normal lifespan in the near future.
Dr. Finklea is Assistant Professor of Medicine, Division of Pulmonary and Critical Care, University of Texas Southwestern, Dallas, Texas. Dr. Finklea receives grant support from the Cystic Fibrosis Foundation.
Cystic fibrosis (CF) is an autosomal recessive disorder affecting thousands of people worldwide. When this genetic disease was first discovered in the first half of the 20th century, the median survival was approximately 5 years of age. Since then, median survival for patients with CF has steadily improved. Today, it is 47.4 years based on Cystic Fibrosis Foundation registry data from 2018. Patients with CF are living longer and staying healthier; the discussion to follow is how patients, researchers, and the CF Foundation reached this point.
In 1938, pediatrician and pathologist Dorothy Anderson observed on the autopsies of children thought to have celiac disease pancreatic lesions she termed “cystic fibrosis of the pancreas.” In addition to the abnormal pancreas, she noted abnormal lungs filled with mucus that obstructed the airways.
Paul Di Sant’Agnese recognized during a heatwave in late 1948 that children with CF were routinely being diagnosed with heatstroke and dehydration. This helped lead to the discovery that these children had elevated salt content in their sweat, paving the way for the development of the sweat chloride test in 1959 by Gibson and Cooke. Not only did Dr. Di Sant’Agnese recognize excess salt in the sweat of patients with CF, but with the help of several concerned parents of children with CF, he established the Cystic Fibrosis Foundation in 1955. The Foundation helped organize the care center model over the next decades, increasing from 30 care centers in 1962 to over 100 in 1978. The care center model also developed a patient registry to track patient care longitudinally.
In June 1989, Francis Collins and Lap-Chee Tsui discovered the location of the CF transmembrane conductance regulator (CFTR) protein using a novel technique called chromosome jumping (Rommens JM, et al. Science. 1989;245[4922]1059). The discovery was a breakthrough in basic science research, but it would take 3 more decades before this discovery could be translated into a medication that could be used by most patients for everyday care.
In the early 1990s, when median survival for patients with CF was 29 years of age, the CF Foundation and Genentech, Inc., coordinated a 24-week multicenter double-blind randomized control trial (RCT) for a new inhaled medication that digested the extracellular DNA from the neutrophils that accumulated in the airways of patients with CF. Inhaled recombinant human DNase in these patients reduced the risk of pulmonary exacerbations and also had a small improvement in pulmonary function in the group compared with the placebo group (Fuchs H, et al. N Engl J Med. 1994;331:637). Five years later, another double-blind RCT demonstrated that inhaled tobramycin in patients with CF whose disease was colonized with Pseudomonas aeruginosa improved pulmonary function and reduced the risk of hospitalizations (Ramsey B, et al. N Engl J Med. 1999;340:23). In 2006, the use of hypertonic saline solution in patients with CF decreased the overall pulmonary exacerbation rate (Elkins MR, et al. N Engl J Med. 2006;354:229). The combination of these inhaled medications, along with inhaled aztreonam, formed the backbone of inhalation therapy for CF care today.
In 1998, even with the ongoing development and approval of new CF medications by the pharmaceutical industry, Robert Beall, CEO of the CF Foundation, realized that he needed to challenge the current drug development paradigm. Instead of trying to convince companies to develop CF medications, he started a concept called venture philanthropy. This concept entailed the CF Foundation financially investing in pharmaceutical companies’ development of new medications. The Foundation first invested in a small company named Aurora Biosciences (known today as Vertex Pharmaceuticals) in 2000. Aurora Biosciences specialized in high throughput screening. This process uses a unique technology allowing one to test the therapeutic reaction of airway cells to thousands of chemical compounds in a single day, instead of using the traditional process of tediously pipetting compounds one by one. Today, the CF Foundation has invested millions of dollars into bioscience research to advance CF care.
In 2011, the results of a study were published in which a small molecule altered defective CFTR protein in patients with CF with the CFTR mutation G551D, thus improving chloride transport at the airway surface. In the original study, after 24 weeks of therapy receiving the medication known as ivacaftor, predicted FEV1 in patients with CF improved 10.6%, and the patients were 55% less likely to have a pulmonary exacerbation compared with those receiving a placebo. This breakthrough provided patients with CF the first medication that could correct the CFTR at the source of the problem (Ramsey BW, et al. N Engl J Med. 2011;365:1663). Ivacaftor was approved by the US FDA in 2012.
Ivacaftor provided proof of concept that using small molecules could improve CFTR function. Ivacaftor was only beneficial to a small percentage of patients and was not effective in patients with CF who had either 1 or 2 F508del CFTR mutations. In 2015, patients with CF with F508del homozygous treated with a combination therapy of lumacaftor/ivacaftor had predicted FEV1% improved 2.6% to 4.0%. More importantly, there was a significant reduction in the number of pulmonary exacerbations per year compared with placebo. Unexpectedly, some of the patients experienced bronchoconstriction while receiving lumacaftor/ivacaftor (Wainwright CE, et al. N Engl J Med. 2015; 373:220). The problem was recognized, and a new small molecule to improve the processing and trafficking of CFTR called tezacaftor was developed. The combination of tezacaftor/ivacaftor in patients with CF who were F508del homozygous demonstrated a similar reduction in pulmonary exacerbations, an absolute improvement of predicted FEV1 of 4%, and no increased respiratory symptoms compared with the placebo arm (Taylor-Cousar JL, et al. N Engl J Med. 2017;377[21]2013).
CFTR modulators were a major breakthrough for patients with CF, but the efficacy of these therapies was dependent on the patients’ genotype and ranged from mildly to moderately effective. Unfortunately, these therapies were ineffective for the patients who were delta 508 heterozygotes. Starting in the summer of 2018, VX 445-tezacaftor-ivacaftor (ETI) was compared with placebo in patients with CF who were 1 copy of F508del and a second CFTR mutation that has minimal function. The study found an absolute improvement in predicted FEV1 of 14.3% and a 63% reduction in exacerbations at 24 weeks compared with placebo (Middleton PG, et al. N Engl J Med. 2019;381:1809). In late 2019, based on these data, ETI was approved by the FDA for all patients with CF who were F508del heterozygous. This innovation provided effective therapy to 90% of the CF population.
With the discovery of many highly effective therapies beneficial in most patients, the CF Foundation started a program called Path to a Cure to find therapies for the 10% of patients with CF who were not candidates for ETI or other CFTR modulators. This program looks to develop novel methods to restore CFTR protein function and repair or replace the CFTR protein via gene editing or gene transfer. This process creates many challenges that are quite complex, but patients, researchers, physicians, and CF Foundation will not stop working until CF stands for CURE FOUND.
Today, patients with CF are living longer, and many are eligible or have already started ETI therapy. This medication and the many others being developed will hopefully lead to patients with CF living a normal lifespan in the near future.
Dr. Finklea is Assistant Professor of Medicine, Division of Pulmonary and Critical Care, University of Texas Southwestern, Dallas, Texas. Dr. Finklea receives grant support from the Cystic Fibrosis Foundation.
Daily Recap: Avoid alcohol to reduce cancer risk, COVID’s lasting health system impact
Here are the stories our MDedge editors across specialties think you need to know about today:
ACS Update: ‘It is best not to drink alcohol’
The American Cancer Society (ACS) is taking its strongest stance yet against drinking. In its updated cancer prevention guidelines, the ACS now recommends that “it is best not to drink alcohol.” Previously, the organizations had suggested that, for those who consume alcoholic beverages, intake should be no more than one drink per day for women or two per day for men. That recommendation is still in place, but is now accompanied by this new, stronger directive. The guidelines also place more emphasis on reducing the consumption of processed and red meat and highly processed foods, and on increasing physical activity. “Individual choice is an important part of a healthy lifestyle, but having the right policies and environmental factors to break down these barriers is also important, and that is something that clinicians can support,” said Laura Makaroff, DO, American Cancer Society senior vice president. The guidelines were published in CA: A Cancer Journal for Clinicians. Read more.
COVID health system changes may be here to stay
The COVID-19 pandemic has forced sudden major changes to the nation’s health care system that are unlikely to be reversed. While there’s some good news, there are also some alarming trends. Experts said there are three trends that are likely to stick around: telehealth for all, an exodus of primary care physicians, and less emphasis on hospital care. “I’ve been trying to raise the alarm about the kind of perilous future of primary care,” said Farzad Mostashari, MD, a top Department of Health & Human Services official in the Obama administration. Dr. Mostashari runs Aledade, a company that helps primary care doctors make the transition from fee-for-service medicine to new payment models. The American Academy of Family Physicians reports that 70% of primary care physicians are reporting declines in patient volume of 50% or more since March, and 40% have laid off or furloughed staff. The AAFP has joined other primary care and insurance groups in asking HHS for an infusion of cash. “This is absolutely essential to effectively treat patients today and to maintain their ongoing operations until we overcome this public health emergency,” the groups wrote. Read more.
Asthma-COPD overlap deaths
Death rates for combined asthma and chronic obstructive pulmonary disease declined during 1999-2016, but the risk remains higher among women, compared with men, and in certain occupations, according to a recent report from the Centers for Disease Control and Prevention. There is also an association between mortality and nonworking status among adults aged 25-64 years, which “suggests that asthma-COPD overlap might be associated with substantial morbidity,” Katelynn E. Dodd, MPH, and associates at the CDC’s National Institute for Occupational Safety and Health said in the Morbidity and Mortality Weekly Report. “These patients have been reported to have worse health outcomes than do those with asthma or COPD alone.” Read more.
Cancer triage in a pandemic: There’s an app for that
Deciding which cancer patients need immediate treatment and who can safely wait is an uncomfortable assessment for cancer clinicians during the COVID-19 pandemic. Now, a new tool, which appears to be the first of its kind, quantifies that risk-benefit analysis. But its presence immediately raises the question: can it help? OncCOVID is a free tool that was launched in May by the University of Michigan. It allows physicians to individualize risk estimates for delaying treatment of up to 25 early- to late-stage cancers. It includes more than 45 patient characteristics, such as age, location, cancer type, cancer stage, treatment plan, underlying medical conditions, and proposed length of delay in care. “We thought, isn’t it better to at least provide some evidence-based quantification, rather than a back-of-the-envelope three-tier system that is just sort of ‘made up’?“ explained one of the developers, Daniel Spratt, MD, associate professor of radiation oncology at Michigan Medicine. Read more.
For more on COVID-19, visit our Resource Center . All of our latest news is available on MDedge.com .
Here are the stories our MDedge editors across specialties think you need to know about today:
ACS Update: ‘It is best not to drink alcohol’
The American Cancer Society (ACS) is taking its strongest stance yet against drinking. In its updated cancer prevention guidelines, the ACS now recommends that “it is best not to drink alcohol.” Previously, the organizations had suggested that, for those who consume alcoholic beverages, intake should be no more than one drink per day for women or two per day for men. That recommendation is still in place, but is now accompanied by this new, stronger directive. The guidelines also place more emphasis on reducing the consumption of processed and red meat and highly processed foods, and on increasing physical activity. “Individual choice is an important part of a healthy lifestyle, but having the right policies and environmental factors to break down these barriers is also important, and that is something that clinicians can support,” said Laura Makaroff, DO, American Cancer Society senior vice president. The guidelines were published in CA: A Cancer Journal for Clinicians. Read more.
COVID health system changes may be here to stay
The COVID-19 pandemic has forced sudden major changes to the nation’s health care system that are unlikely to be reversed. While there’s some good news, there are also some alarming trends. Experts said there are three trends that are likely to stick around: telehealth for all, an exodus of primary care physicians, and less emphasis on hospital care. “I’ve been trying to raise the alarm about the kind of perilous future of primary care,” said Farzad Mostashari, MD, a top Department of Health & Human Services official in the Obama administration. Dr. Mostashari runs Aledade, a company that helps primary care doctors make the transition from fee-for-service medicine to new payment models. The American Academy of Family Physicians reports that 70% of primary care physicians are reporting declines in patient volume of 50% or more since March, and 40% have laid off or furloughed staff. The AAFP has joined other primary care and insurance groups in asking HHS for an infusion of cash. “This is absolutely essential to effectively treat patients today and to maintain their ongoing operations until we overcome this public health emergency,” the groups wrote. Read more.
Asthma-COPD overlap deaths
Death rates for combined asthma and chronic obstructive pulmonary disease declined during 1999-2016, but the risk remains higher among women, compared with men, and in certain occupations, according to a recent report from the Centers for Disease Control and Prevention. There is also an association between mortality and nonworking status among adults aged 25-64 years, which “suggests that asthma-COPD overlap might be associated with substantial morbidity,” Katelynn E. Dodd, MPH, and associates at the CDC’s National Institute for Occupational Safety and Health said in the Morbidity and Mortality Weekly Report. “These patients have been reported to have worse health outcomes than do those with asthma or COPD alone.” Read more.
Cancer triage in a pandemic: There’s an app for that
Deciding which cancer patients need immediate treatment and who can safely wait is an uncomfortable assessment for cancer clinicians during the COVID-19 pandemic. Now, a new tool, which appears to be the first of its kind, quantifies that risk-benefit analysis. But its presence immediately raises the question: can it help? OncCOVID is a free tool that was launched in May by the University of Michigan. It allows physicians to individualize risk estimates for delaying treatment of up to 25 early- to late-stage cancers. It includes more than 45 patient characteristics, such as age, location, cancer type, cancer stage, treatment plan, underlying medical conditions, and proposed length of delay in care. “We thought, isn’t it better to at least provide some evidence-based quantification, rather than a back-of-the-envelope three-tier system that is just sort of ‘made up’?“ explained one of the developers, Daniel Spratt, MD, associate professor of radiation oncology at Michigan Medicine. Read more.
For more on COVID-19, visit our Resource Center . All of our latest news is available on MDedge.com .
Here are the stories our MDedge editors across specialties think you need to know about today:
ACS Update: ‘It is best not to drink alcohol’
The American Cancer Society (ACS) is taking its strongest stance yet against drinking. In its updated cancer prevention guidelines, the ACS now recommends that “it is best not to drink alcohol.” Previously, the organizations had suggested that, for those who consume alcoholic beverages, intake should be no more than one drink per day for women or two per day for men. That recommendation is still in place, but is now accompanied by this new, stronger directive. The guidelines also place more emphasis on reducing the consumption of processed and red meat and highly processed foods, and on increasing physical activity. “Individual choice is an important part of a healthy lifestyle, but having the right policies and environmental factors to break down these barriers is also important, and that is something that clinicians can support,” said Laura Makaroff, DO, American Cancer Society senior vice president. The guidelines were published in CA: A Cancer Journal for Clinicians. Read more.
COVID health system changes may be here to stay
The COVID-19 pandemic has forced sudden major changes to the nation’s health care system that are unlikely to be reversed. While there’s some good news, there are also some alarming trends. Experts said there are three trends that are likely to stick around: telehealth for all, an exodus of primary care physicians, and less emphasis on hospital care. “I’ve been trying to raise the alarm about the kind of perilous future of primary care,” said Farzad Mostashari, MD, a top Department of Health & Human Services official in the Obama administration. Dr. Mostashari runs Aledade, a company that helps primary care doctors make the transition from fee-for-service medicine to new payment models. The American Academy of Family Physicians reports that 70% of primary care physicians are reporting declines in patient volume of 50% or more since March, and 40% have laid off or furloughed staff. The AAFP has joined other primary care and insurance groups in asking HHS for an infusion of cash. “This is absolutely essential to effectively treat patients today and to maintain their ongoing operations until we overcome this public health emergency,” the groups wrote. Read more.
Asthma-COPD overlap deaths
Death rates for combined asthma and chronic obstructive pulmonary disease declined during 1999-2016, but the risk remains higher among women, compared with men, and in certain occupations, according to a recent report from the Centers for Disease Control and Prevention. There is also an association between mortality and nonworking status among adults aged 25-64 years, which “suggests that asthma-COPD overlap might be associated with substantial morbidity,” Katelynn E. Dodd, MPH, and associates at the CDC’s National Institute for Occupational Safety and Health said in the Morbidity and Mortality Weekly Report. “These patients have been reported to have worse health outcomes than do those with asthma or COPD alone.” Read more.
Cancer triage in a pandemic: There’s an app for that
Deciding which cancer patients need immediate treatment and who can safely wait is an uncomfortable assessment for cancer clinicians during the COVID-19 pandemic. Now, a new tool, which appears to be the first of its kind, quantifies that risk-benefit analysis. But its presence immediately raises the question: can it help? OncCOVID is a free tool that was launched in May by the University of Michigan. It allows physicians to individualize risk estimates for delaying treatment of up to 25 early- to late-stage cancers. It includes more than 45 patient characteristics, such as age, location, cancer type, cancer stage, treatment plan, underlying medical conditions, and proposed length of delay in care. “We thought, isn’t it better to at least provide some evidence-based quantification, rather than a back-of-the-envelope three-tier system that is just sort of ‘made up’?“ explained one of the developers, Daniel Spratt, MD, associate professor of radiation oncology at Michigan Medicine. Read more.
For more on COVID-19, visit our Resource Center . All of our latest news is available on MDedge.com .
Human sitters in the COVID era
Data collection needed for care of suicidal hospitalized patients
I am writing this commentary to bring to readers’ attention a medical and ethical complexity related to human sitters for presumably suicidal, COVID-19–positive hospitalized patients.
To shape and bundle the ethics issues addressed here into a single question, I offer the following: Should policies and practices requiring that patients in presumed need of a sitter because of assessed suicidality change when the patient is COVID-19–positive? Although the analysis might be similar when a sitter is monitoring a Patient Under Investigation (PUI), here I focus only on COVID-19–positive patients. Similarly, there are other reasons for sitters, of course, such as to prevent elopement, or, if a patient is in restraints, to prevent the patient from pulling out lines or tubes. Again, discussion of some of these ethical complications is beyond the scope of this piece. Just considering the matter of potential suicidality and sitters is complex enough. And so, to start, I sought out existing sources for guidance.
In looking for such sources, I first turned to the Centers for Medicare and Medicaid Services before COVID-19. CMS has required that there be a sitter for a patient who is suicidal and that the sitter remain in the room so that the sitter can intervene expeditiously if the patient tries to hurt himself or herself. There has been no change in this guidance since the COVID-19 pandemic in the United States. To the best of my knowledge, there is no substantive guidance for protecting sitters from contagion other than PPE. Given this, it begs the question:
In my hospital, I already have begun discussing the potential risks of harm and potential benefits to our suicidal patients of having a sitter directly outside the patient’s room. I also have considered whether to have one sitter watching several room cameras at once, commonly referred to as “telehealth strategies.”
To be sure, sitting for hours in the room of a COVID-19–positive patient is onerous. The sitter is required to be in full PPE (N-95 mask, gown, and gloves), which is hot and uncomfortable. Current practice is resource intensive in other ways. It requires changing out the sitter every 2 hours, which uses substantial amounts of PPE and multiple sitters.
Regardless, however, there are really no data upon which to base any sound ethics judgment about what should or should not be tried. We just have no information on how to attempt to balance potential risks and prospects for the benefit of whom and when. And, given that good clinical ethics always begin with the facts, I write this piece to see whether readers have thought about these issues before – and whether any of clinicians have started collecting the valuable data needed to begin making sound ethical judgments about how to care for our presumably suicidal COVID-19–positive patients and the sitters who watch over them.
Dr. Ritchie is chair of psychiatry at Medstar Washington Hospital Center and professor of psychiatry at Georgetown University, Washington. She has no disclosures and can be reached at [email protected].
This column is an outcome of a discussion that occurred during Psych/Ethics rounds on June 5, and does not represent any official statements of Medstar Washington Hospital Center or any entity of the MedStar Corp. Dr. Ritchie would like to thank Evan G. DeRenzo, PhD, of the John J. Lynch Center for Ethics, for her thoughtful review of a previous draft of this commentary.
Data collection needed for care of suicidal hospitalized patients
Data collection needed for care of suicidal hospitalized patients
I am writing this commentary to bring to readers’ attention a medical and ethical complexity related to human sitters for presumably suicidal, COVID-19–positive hospitalized patients.
To shape and bundle the ethics issues addressed here into a single question, I offer the following: Should policies and practices requiring that patients in presumed need of a sitter because of assessed suicidality change when the patient is COVID-19–positive? Although the analysis might be similar when a sitter is monitoring a Patient Under Investigation (PUI), here I focus only on COVID-19–positive patients. Similarly, there are other reasons for sitters, of course, such as to prevent elopement, or, if a patient is in restraints, to prevent the patient from pulling out lines or tubes. Again, discussion of some of these ethical complications is beyond the scope of this piece. Just considering the matter of potential suicidality and sitters is complex enough. And so, to start, I sought out existing sources for guidance.
In looking for such sources, I first turned to the Centers for Medicare and Medicaid Services before COVID-19. CMS has required that there be a sitter for a patient who is suicidal and that the sitter remain in the room so that the sitter can intervene expeditiously if the patient tries to hurt himself or herself. There has been no change in this guidance since the COVID-19 pandemic in the United States. To the best of my knowledge, there is no substantive guidance for protecting sitters from contagion other than PPE. Given this, it begs the question:
In my hospital, I already have begun discussing the potential risks of harm and potential benefits to our suicidal patients of having a sitter directly outside the patient’s room. I also have considered whether to have one sitter watching several room cameras at once, commonly referred to as “telehealth strategies.”
To be sure, sitting for hours in the room of a COVID-19–positive patient is onerous. The sitter is required to be in full PPE (N-95 mask, gown, and gloves), which is hot and uncomfortable. Current practice is resource intensive in other ways. It requires changing out the sitter every 2 hours, which uses substantial amounts of PPE and multiple sitters.
Regardless, however, there are really no data upon which to base any sound ethics judgment about what should or should not be tried. We just have no information on how to attempt to balance potential risks and prospects for the benefit of whom and when. And, given that good clinical ethics always begin with the facts, I write this piece to see whether readers have thought about these issues before – and whether any of clinicians have started collecting the valuable data needed to begin making sound ethical judgments about how to care for our presumably suicidal COVID-19–positive patients and the sitters who watch over them.
Dr. Ritchie is chair of psychiatry at Medstar Washington Hospital Center and professor of psychiatry at Georgetown University, Washington. She has no disclosures and can be reached at [email protected].
This column is an outcome of a discussion that occurred during Psych/Ethics rounds on June 5, and does not represent any official statements of Medstar Washington Hospital Center or any entity of the MedStar Corp. Dr. Ritchie would like to thank Evan G. DeRenzo, PhD, of the John J. Lynch Center for Ethics, for her thoughtful review of a previous draft of this commentary.
I am writing this commentary to bring to readers’ attention a medical and ethical complexity related to human sitters for presumably suicidal, COVID-19–positive hospitalized patients.
To shape and bundle the ethics issues addressed here into a single question, I offer the following: Should policies and practices requiring that patients in presumed need of a sitter because of assessed suicidality change when the patient is COVID-19–positive? Although the analysis might be similar when a sitter is monitoring a Patient Under Investigation (PUI), here I focus only on COVID-19–positive patients. Similarly, there are other reasons for sitters, of course, such as to prevent elopement, or, if a patient is in restraints, to prevent the patient from pulling out lines or tubes. Again, discussion of some of these ethical complications is beyond the scope of this piece. Just considering the matter of potential suicidality and sitters is complex enough. And so, to start, I sought out existing sources for guidance.
In looking for such sources, I first turned to the Centers for Medicare and Medicaid Services before COVID-19. CMS has required that there be a sitter for a patient who is suicidal and that the sitter remain in the room so that the sitter can intervene expeditiously if the patient tries to hurt himself or herself. There has been no change in this guidance since the COVID-19 pandemic in the United States. To the best of my knowledge, there is no substantive guidance for protecting sitters from contagion other than PPE. Given this, it begs the question:
In my hospital, I already have begun discussing the potential risks of harm and potential benefits to our suicidal patients of having a sitter directly outside the patient’s room. I also have considered whether to have one sitter watching several room cameras at once, commonly referred to as “telehealth strategies.”
To be sure, sitting for hours in the room of a COVID-19–positive patient is onerous. The sitter is required to be in full PPE (N-95 mask, gown, and gloves), which is hot and uncomfortable. Current practice is resource intensive in other ways. It requires changing out the sitter every 2 hours, which uses substantial amounts of PPE and multiple sitters.
Regardless, however, there are really no data upon which to base any sound ethics judgment about what should or should not be tried. We just have no information on how to attempt to balance potential risks and prospects for the benefit of whom and when. And, given that good clinical ethics always begin with the facts, I write this piece to see whether readers have thought about these issues before – and whether any of clinicians have started collecting the valuable data needed to begin making sound ethical judgments about how to care for our presumably suicidal COVID-19–positive patients and the sitters who watch over them.
Dr. Ritchie is chair of psychiatry at Medstar Washington Hospital Center and professor of psychiatry at Georgetown University, Washington. She has no disclosures and can be reached at [email protected].
This column is an outcome of a discussion that occurred during Psych/Ethics rounds on June 5, and does not represent any official statements of Medstar Washington Hospital Center or any entity of the MedStar Corp. Dr. Ritchie would like to thank Evan G. DeRenzo, PhD, of the John J. Lynch Center for Ethics, for her thoughtful review of a previous draft of this commentary.
Elevated inflammation common in children’s severe COVID-19 disease
according to data from 50 patients at a single tertiary care center.
“Risk factors for severe disease in pediatric populations have not been clearly identified and the high prevalence of SARS-CoV-2 in NYC offers an opportunity to describe severe pediatric disease in more detail,” wrote Philip Zachariah, MD, of New York–Presbyterian Hospital, New York, and colleagues.
In a retrospective case series published in JAMA Pediatrics, the researchers reviewed data from 50 patients: 41 classified as severe and 9 classified as nonsevere. Among the patients, 27 were male and 25 were Hispanic. The patient population had a median of 2 days from symptom onset to hospital admission. The most common symptoms were fever (80%) and respiratory symptoms (64%). Seventy-six percent of patients had a median length of stay of 3 days (range 1-30 days).
At hospital admission, children with severe disease had significantly higher levels of several inflammatory markers compared with those without severe disease, notably C-reactive protein (median 8.978 mg/dL vs. 0.64 mg/dL) and procalcitonin (median 0.31 ng/mL vs. 0.17 ng/mL, (P < .001 for both). High mean peak levels of C-reactive protein, procalcitonin, interleukin 6, ferritin, and D-dimer were seen among the nine children (16%) who required mechanical ventilation, Dr. Zachariah and associates said.
None of the 14 infants and 1 of the 8 immunocompromised children in the study had severe disease, the researchers wrote.
Bacterial coinfections detected while patients were hospitalized were bacteremia in 6%, suspected bacterial pneumonia in 18%, urinary tract infections in 10%, skin and soft tissue infections in 6%, and streptococcus pharyngitis in 2%, Dr. Zachariah and associates reported.
Overall, 61% of the children had comorbidities identified in previous COVID-19 studies, of which obesity was the most common (22%); other comorbidities included asthma, sickle cell disease, cardiac disease, and diabetes. Obesity also was significantly associated with the need for mechanical ventilation in children aged 2 years and older (67%). A total of 16 patients required respiratory support, 9 of these were placed on mechanical ventilation; 6 of these 9 children were obese.
Fifteen patients (30%) who met criteria for increased oxygen requirements and respiratory distress received hydroxychloroquine, but the small sample size did not allow for assessment of treatment efficacy, the researchers said.
“Expanding our knowledge of COVID-19 [disease] in children will potentially permit early recognition of SARS-CoV-2 infection, understanding of the natural history of disease, and potential complications, said Stephen I. Pelton, MD, professor of pediatrics and epidemiology at Boston University and senior attending physician at Boston Medical Center. This review of 50 SARS-CoV-2 infected children (less than 21 years of age) “provides insight into the short period of symptoms prior to hospitalization, challenges the concept that infants less than 1 year are at greatest risk of severe disease (as from the experience in China), and suggests rapid recovery in many children, as median length of stay was 3 days.
“The review revealed two findings that were surprising to me. First, the median length of stay of 3 days. As nearly 20% of the children required mechanical ventilation, it suggests many of the children were discharged quickly after evaluation, suggesting that we need to identify markers of severity to predict those children likely to have progressive disease and require respiratory support,” Dr. Pelton noted.
“The second observation suggests high rates of bacterial infection (bacteremia, pneumonia, UTI, and skin and soft tissue infection). I do not think this has been widely reported in adults, and may represent a difference between child and adult disease. More studies such as this will be required to identify how common coinfection with bacteria is,” he said.
“The take-home message is that although most children with COVID-19 have a mild or even asymptomatic course, some become severely ill requiring ventilator support and potentially ECMO [extracorporeal membrane oxygenation]. Potential predictors of severity include high C-reactive protein, obesity, and older age [adolescence], said Dr. Pelton, who was not involved in the study.
What additional research is needed? Dr. Pelton said that better markers of severe disease are needed, as well as an understanding of why obesity is a risk factor for severe disease in both children and adults. Are these prediabetic patients? he asked.
The study findings were limited by the small sample size and high proportion of Hispanic patients, which may limit generalizability, and some symptoms and comorbidities may have been missed because of the retrospective nature of the study, the researchers noted. However, the results support the need for hospitals to remain vigilant to the variable presentations of COVID-19 infections in children.
“Therapeutic considerations need to [include] the risk of toxicity, control of antiviral replication, and early recognition and management of immune dysregulation,” they concluded.
The study received no outside funding. Dr. Zachariah had no financial conflicts to disclose. Two coauthors reported ties with various pharmaceutical companies and organizations. Dr. Pelton said he had no relevant financial disclosures.
SOURCE: Zachariah P et al. JAMA Pediatr. 2020 June 3. doi:10.1001/jamapediatrics.2020.2430.
according to data from 50 patients at a single tertiary care center.
“Risk factors for severe disease in pediatric populations have not been clearly identified and the high prevalence of SARS-CoV-2 in NYC offers an opportunity to describe severe pediatric disease in more detail,” wrote Philip Zachariah, MD, of New York–Presbyterian Hospital, New York, and colleagues.
In a retrospective case series published in JAMA Pediatrics, the researchers reviewed data from 50 patients: 41 classified as severe and 9 classified as nonsevere. Among the patients, 27 were male and 25 were Hispanic. The patient population had a median of 2 days from symptom onset to hospital admission. The most common symptoms were fever (80%) and respiratory symptoms (64%). Seventy-six percent of patients had a median length of stay of 3 days (range 1-30 days).
At hospital admission, children with severe disease had significantly higher levels of several inflammatory markers compared with those without severe disease, notably C-reactive protein (median 8.978 mg/dL vs. 0.64 mg/dL) and procalcitonin (median 0.31 ng/mL vs. 0.17 ng/mL, (P < .001 for both). High mean peak levels of C-reactive protein, procalcitonin, interleukin 6, ferritin, and D-dimer were seen among the nine children (16%) who required mechanical ventilation, Dr. Zachariah and associates said.
None of the 14 infants and 1 of the 8 immunocompromised children in the study had severe disease, the researchers wrote.
Bacterial coinfections detected while patients were hospitalized were bacteremia in 6%, suspected bacterial pneumonia in 18%, urinary tract infections in 10%, skin and soft tissue infections in 6%, and streptococcus pharyngitis in 2%, Dr. Zachariah and associates reported.
Overall, 61% of the children had comorbidities identified in previous COVID-19 studies, of which obesity was the most common (22%); other comorbidities included asthma, sickle cell disease, cardiac disease, and diabetes. Obesity also was significantly associated with the need for mechanical ventilation in children aged 2 years and older (67%). A total of 16 patients required respiratory support, 9 of these were placed on mechanical ventilation; 6 of these 9 children were obese.
Fifteen patients (30%) who met criteria for increased oxygen requirements and respiratory distress received hydroxychloroquine, but the small sample size did not allow for assessment of treatment efficacy, the researchers said.
“Expanding our knowledge of COVID-19 [disease] in children will potentially permit early recognition of SARS-CoV-2 infection, understanding of the natural history of disease, and potential complications, said Stephen I. Pelton, MD, professor of pediatrics and epidemiology at Boston University and senior attending physician at Boston Medical Center. This review of 50 SARS-CoV-2 infected children (less than 21 years of age) “provides insight into the short period of symptoms prior to hospitalization, challenges the concept that infants less than 1 year are at greatest risk of severe disease (as from the experience in China), and suggests rapid recovery in many children, as median length of stay was 3 days.
“The review revealed two findings that were surprising to me. First, the median length of stay of 3 days. As nearly 20% of the children required mechanical ventilation, it suggests many of the children were discharged quickly after evaluation, suggesting that we need to identify markers of severity to predict those children likely to have progressive disease and require respiratory support,” Dr. Pelton noted.
“The second observation suggests high rates of bacterial infection (bacteremia, pneumonia, UTI, and skin and soft tissue infection). I do not think this has been widely reported in adults, and may represent a difference between child and adult disease. More studies such as this will be required to identify how common coinfection with bacteria is,” he said.
“The take-home message is that although most children with COVID-19 have a mild or even asymptomatic course, some become severely ill requiring ventilator support and potentially ECMO [extracorporeal membrane oxygenation]. Potential predictors of severity include high C-reactive protein, obesity, and older age [adolescence], said Dr. Pelton, who was not involved in the study.
What additional research is needed? Dr. Pelton said that better markers of severe disease are needed, as well as an understanding of why obesity is a risk factor for severe disease in both children and adults. Are these prediabetic patients? he asked.
The study findings were limited by the small sample size and high proportion of Hispanic patients, which may limit generalizability, and some symptoms and comorbidities may have been missed because of the retrospective nature of the study, the researchers noted. However, the results support the need for hospitals to remain vigilant to the variable presentations of COVID-19 infections in children.
“Therapeutic considerations need to [include] the risk of toxicity, control of antiviral replication, and early recognition and management of immune dysregulation,” they concluded.
The study received no outside funding. Dr. Zachariah had no financial conflicts to disclose. Two coauthors reported ties with various pharmaceutical companies and organizations. Dr. Pelton said he had no relevant financial disclosures.
SOURCE: Zachariah P et al. JAMA Pediatr. 2020 June 3. doi:10.1001/jamapediatrics.2020.2430.
according to data from 50 patients at a single tertiary care center.
“Risk factors for severe disease in pediatric populations have not been clearly identified and the high prevalence of SARS-CoV-2 in NYC offers an opportunity to describe severe pediatric disease in more detail,” wrote Philip Zachariah, MD, of New York–Presbyterian Hospital, New York, and colleagues.
In a retrospective case series published in JAMA Pediatrics, the researchers reviewed data from 50 patients: 41 classified as severe and 9 classified as nonsevere. Among the patients, 27 were male and 25 were Hispanic. The patient population had a median of 2 days from symptom onset to hospital admission. The most common symptoms were fever (80%) and respiratory symptoms (64%). Seventy-six percent of patients had a median length of stay of 3 days (range 1-30 days).
At hospital admission, children with severe disease had significantly higher levels of several inflammatory markers compared with those without severe disease, notably C-reactive protein (median 8.978 mg/dL vs. 0.64 mg/dL) and procalcitonin (median 0.31 ng/mL vs. 0.17 ng/mL, (P < .001 for both). High mean peak levels of C-reactive protein, procalcitonin, interleukin 6, ferritin, and D-dimer were seen among the nine children (16%) who required mechanical ventilation, Dr. Zachariah and associates said.
None of the 14 infants and 1 of the 8 immunocompromised children in the study had severe disease, the researchers wrote.
Bacterial coinfections detected while patients were hospitalized were bacteremia in 6%, suspected bacterial pneumonia in 18%, urinary tract infections in 10%, skin and soft tissue infections in 6%, and streptococcus pharyngitis in 2%, Dr. Zachariah and associates reported.
Overall, 61% of the children had comorbidities identified in previous COVID-19 studies, of which obesity was the most common (22%); other comorbidities included asthma, sickle cell disease, cardiac disease, and diabetes. Obesity also was significantly associated with the need for mechanical ventilation in children aged 2 years and older (67%). A total of 16 patients required respiratory support, 9 of these were placed on mechanical ventilation; 6 of these 9 children were obese.
Fifteen patients (30%) who met criteria for increased oxygen requirements and respiratory distress received hydroxychloroquine, but the small sample size did not allow for assessment of treatment efficacy, the researchers said.
“Expanding our knowledge of COVID-19 [disease] in children will potentially permit early recognition of SARS-CoV-2 infection, understanding of the natural history of disease, and potential complications, said Stephen I. Pelton, MD, professor of pediatrics and epidemiology at Boston University and senior attending physician at Boston Medical Center. This review of 50 SARS-CoV-2 infected children (less than 21 years of age) “provides insight into the short period of symptoms prior to hospitalization, challenges the concept that infants less than 1 year are at greatest risk of severe disease (as from the experience in China), and suggests rapid recovery in many children, as median length of stay was 3 days.
“The review revealed two findings that were surprising to me. First, the median length of stay of 3 days. As nearly 20% of the children required mechanical ventilation, it suggests many of the children were discharged quickly after evaluation, suggesting that we need to identify markers of severity to predict those children likely to have progressive disease and require respiratory support,” Dr. Pelton noted.
“The second observation suggests high rates of bacterial infection (bacteremia, pneumonia, UTI, and skin and soft tissue infection). I do not think this has been widely reported in adults, and may represent a difference between child and adult disease. More studies such as this will be required to identify how common coinfection with bacteria is,” he said.
“The take-home message is that although most children with COVID-19 have a mild or even asymptomatic course, some become severely ill requiring ventilator support and potentially ECMO [extracorporeal membrane oxygenation]. Potential predictors of severity include high C-reactive protein, obesity, and older age [adolescence], said Dr. Pelton, who was not involved in the study.
What additional research is needed? Dr. Pelton said that better markers of severe disease are needed, as well as an understanding of why obesity is a risk factor for severe disease in both children and adults. Are these prediabetic patients? he asked.
The study findings were limited by the small sample size and high proportion of Hispanic patients, which may limit generalizability, and some symptoms and comorbidities may have been missed because of the retrospective nature of the study, the researchers noted. However, the results support the need for hospitals to remain vigilant to the variable presentations of COVID-19 infections in children.
“Therapeutic considerations need to [include] the risk of toxicity, control of antiviral replication, and early recognition and management of immune dysregulation,” they concluded.
The study received no outside funding. Dr. Zachariah had no financial conflicts to disclose. Two coauthors reported ties with various pharmaceutical companies and organizations. Dr. Pelton said he had no relevant financial disclosures.
SOURCE: Zachariah P et al. JAMA Pediatr. 2020 June 3. doi:10.1001/jamapediatrics.2020.2430.
FROM JAMA PEDIATRICS
By the numbers: Asthma-COPD overlap deaths
Death rates for combined asthma and chronic obstructive pulmonary disease declined during 1999-2016, but the risk remains higher among women, compared with men, and in certain occupations, according to a recent report from the Centers for Disease Control and Prevention.
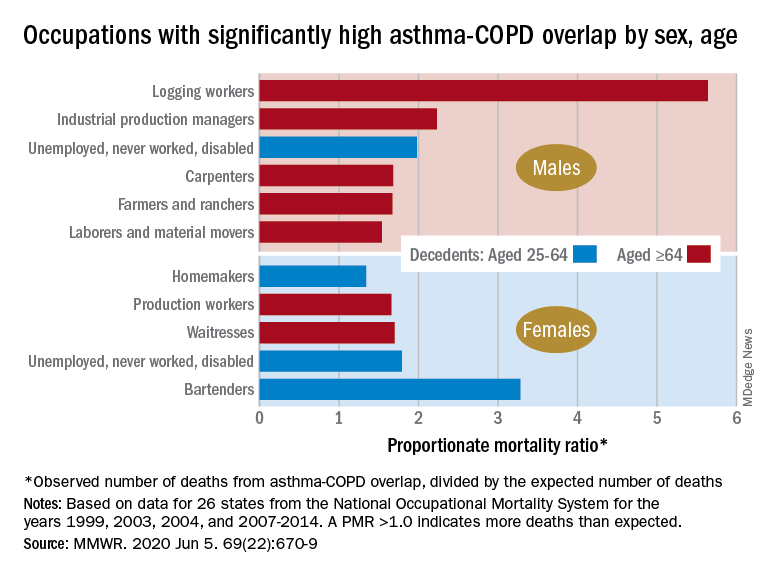
There is also an association between mortality and nonworking status among adults aged 25-64 years, which “suggests that asthma-COPD overlap might be associated with substantial morbidity,” Katelynn E. Dodd, MPH, and associates at the CDC’s National Institute for Occupational Safety and Health said in the Morbidity and Mortality Weekly Report. “These patients have been reported to have worse health outcomes than do those with asthma or COPD alone.”
For females with asthma-COPD overlap, the age-adjusted death rate among adults aged 25 years and older dropped from 7.71 per million in 1999 to 4.01 in 2016, with corresponding rates of 6.70 and 3.01 per million for males, they reported.
In 1999-2016, a total of 18,766 U.S. decedents aged ≥25 years had both asthma and COPD assigned as the underlying or contributing cause of death (12,028 women and 6,738 men), for an overall death rate of 5.03 per million persons (women, 5.59; men, 4.30), data from the National Vital Statistics System show.
Additional analysis, based on the calculation of proportionate mortality ratios (PMRs), also showed that mortality varied by occupational status and age for both males and females, the investigators said, noting that workplace exposures, such as dusts and secondhand smoke, are known to cause both asthma and COPD.
The PMR represents the observed number of deaths from asthma-COPD overlap in a specified industry or occupation, divided by the expected number of deaths, so a value over 1.0 indicates that there were more deaths associated with the condition than expected, Ms. Dodd and her associates explained.
Among female decedents, the occupation with the highest PMR that was statistically significant was bartending at 3.28. For men, the highest significant PMR, 5.64, occurred in logging workers. Those rates, however, only applied to one of the two age groups: 25-64 years in women and ≥65 in men, based on data from the National Occupational Mortality Surveillance, which included information from 26 states for the years 1999, 2003, 2004, and 2007-2014.
Occupationally speaking, the one area of common ground between males and females was lack of occupation. PMRs for those aged 25-64 years “were significantly elevated among men (1.98) and women (1.79) who were unemployed, never worked, or were disabled workers,” they said. PMRs were elevated for nonworking older males and females but were not significant.
The elevated PMRs suggest “that asthma-COPD overlap might be associated with substantial morbidity resulting in loss of employment [because] retired and unemployed persons might have left the workforce because of severe asthma or COPD,” the investigators wrote.
SOURCE: Dodd KE et al. MMWR. 2020 Jun 5. 69(22):670-9.
Death rates for combined asthma and chronic obstructive pulmonary disease declined during 1999-2016, but the risk remains higher among women, compared with men, and in certain occupations, according to a recent report from the Centers for Disease Control and Prevention.

There is also an association between mortality and nonworking status among adults aged 25-64 years, which “suggests that asthma-COPD overlap might be associated with substantial morbidity,” Katelynn E. Dodd, MPH, and associates at the CDC’s National Institute for Occupational Safety and Health said in the Morbidity and Mortality Weekly Report. “These patients have been reported to have worse health outcomes than do those with asthma or COPD alone.”
For females with asthma-COPD overlap, the age-adjusted death rate among adults aged 25 years and older dropped from 7.71 per million in 1999 to 4.01 in 2016, with corresponding rates of 6.70 and 3.01 per million for males, they reported.
In 1999-2016, a total of 18,766 U.S. decedents aged ≥25 years had both asthma and COPD assigned as the underlying or contributing cause of death (12,028 women and 6,738 men), for an overall death rate of 5.03 per million persons (women, 5.59; men, 4.30), data from the National Vital Statistics System show.
Additional analysis, based on the calculation of proportionate mortality ratios (PMRs), also showed that mortality varied by occupational status and age for both males and females, the investigators said, noting that workplace exposures, such as dusts and secondhand smoke, are known to cause both asthma and COPD.
The PMR represents the observed number of deaths from asthma-COPD overlap in a specified industry or occupation, divided by the expected number of deaths, so a value over 1.0 indicates that there were more deaths associated with the condition than expected, Ms. Dodd and her associates explained.
Among female decedents, the occupation with the highest PMR that was statistically significant was bartending at 3.28. For men, the highest significant PMR, 5.64, occurred in logging workers. Those rates, however, only applied to one of the two age groups: 25-64 years in women and ≥65 in men, based on data from the National Occupational Mortality Surveillance, which included information from 26 states for the years 1999, 2003, 2004, and 2007-2014.
Occupationally speaking, the one area of common ground between males and females was lack of occupation. PMRs for those aged 25-64 years “were significantly elevated among men (1.98) and women (1.79) who were unemployed, never worked, or were disabled workers,” they said. PMRs were elevated for nonworking older males and females but were not significant.
The elevated PMRs suggest “that asthma-COPD overlap might be associated with substantial morbidity resulting in loss of employment [because] retired and unemployed persons might have left the workforce because of severe asthma or COPD,” the investigators wrote.
SOURCE: Dodd KE et al. MMWR. 2020 Jun 5. 69(22):670-9.
Death rates for combined asthma and chronic obstructive pulmonary disease declined during 1999-2016, but the risk remains higher among women, compared with men, and in certain occupations, according to a recent report from the Centers for Disease Control and Prevention.

There is also an association between mortality and nonworking status among adults aged 25-64 years, which “suggests that asthma-COPD overlap might be associated with substantial morbidity,” Katelynn E. Dodd, MPH, and associates at the CDC’s National Institute for Occupational Safety and Health said in the Morbidity and Mortality Weekly Report. “These patients have been reported to have worse health outcomes than do those with asthma or COPD alone.”
For females with asthma-COPD overlap, the age-adjusted death rate among adults aged 25 years and older dropped from 7.71 per million in 1999 to 4.01 in 2016, with corresponding rates of 6.70 and 3.01 per million for males, they reported.
In 1999-2016, a total of 18,766 U.S. decedents aged ≥25 years had both asthma and COPD assigned as the underlying or contributing cause of death (12,028 women and 6,738 men), for an overall death rate of 5.03 per million persons (women, 5.59; men, 4.30), data from the National Vital Statistics System show.
Additional analysis, based on the calculation of proportionate mortality ratios (PMRs), also showed that mortality varied by occupational status and age for both males and females, the investigators said, noting that workplace exposures, such as dusts and secondhand smoke, are known to cause both asthma and COPD.
The PMR represents the observed number of deaths from asthma-COPD overlap in a specified industry or occupation, divided by the expected number of deaths, so a value over 1.0 indicates that there were more deaths associated with the condition than expected, Ms. Dodd and her associates explained.
Among female decedents, the occupation with the highest PMR that was statistically significant was bartending at 3.28. For men, the highest significant PMR, 5.64, occurred in logging workers. Those rates, however, only applied to one of the two age groups: 25-64 years in women and ≥65 in men, based on data from the National Occupational Mortality Surveillance, which included information from 26 states for the years 1999, 2003, 2004, and 2007-2014.
Occupationally speaking, the one area of common ground between males and females was lack of occupation. PMRs for those aged 25-64 years “were significantly elevated among men (1.98) and women (1.79) who were unemployed, never worked, or were disabled workers,” they said. PMRs were elevated for nonworking older males and females but were not significant.
The elevated PMRs suggest “that asthma-COPD overlap might be associated with substantial morbidity resulting in loss of employment [because] retired and unemployed persons might have left the workforce because of severe asthma or COPD,” the investigators wrote.
SOURCE: Dodd KE et al. MMWR. 2020 Jun 5. 69(22):670-9.
FROM MMWR
Rapid changes to health system spurred by COVID might be here to stay
The U.S. health care system is famously resistant to government-imposed change. It took decades to create Medicare and Medicaid, mostly because of opposition from the medical-industrial complex. Then it was nearly another half-century before the passage of the Affordable Care Act.
“Health care is never going back to the way it was before,” said Gail Wilensky, a health economist who ran the Medicare and Medicaid programs for President George H.W. Bush in the early 1990s.
Ms. Wilensky is far from the only longtime observer of the American health care system to marvel at the speed of some long-sought changes. But experts warn that the breakthroughs may not all make the health system work better or make it less expensive.
That said, here are three trends that seem likely to continue.
Telehealth for all
Telehealth is not new; medical professionals have used it to reach patients in rural or remote settings since the late 1980s.
But even while technology has made video visits easier, it has failed to reach critical mass, largely because of political fights. Licensing has been one main obstacle – determining how a doctor in one state can legally treat a patient in a state where the doctor is not licensed.
The other obstacle, not surprisingly, is payment. Should a video visit be reimbursed at the same rate as an in-person visit? Will making it easier for doctors and other medical professionals to use telehealth encourage unnecessary care, thus driving up the nation’s $3.6 trillion health tab even more? Or could it replace care once provided free by phone?
Still, the pandemic has pushed aside those sticking points. Almost overnight, by necessity, every health care provider who can is delivering telemedicine. A new survey from Gallup found the number of patients reporting “virtual” medical visits more than doubled, from 12% to 27%, from late March to mid-May. That is attributable, at least in part, to Medicare having made it easier for doctors to bill for virtual visits.
It’s easy to see why many patients like video visits – there’s no parking to find and pay for, and it takes far less time out of a workday than going to an office.
Doctors and other practitioners seem more ambivalent. On one hand, it can be harder to examine a patient over video and some services just can’t be done via a digital connection. On the other hand, they can see more patients in the same amount of time and may need less support staff and possibly smaller offices if more visits are conducted virtually.
Of course, telemedicine doesn’t work for everyone. Many areas and patients don’t have reliable or robust broadband connections that make video visits work. And some patients, particularly the oldest seniors, lack the technological skills needed to connect.
Primary care doctors in peril
Another trend that has suddenly accelerated is worry over the nation’s dwindling supply of primary care doctors. The exodus of practitioners performing primary care has been a concern over the past several years, as baby boomer doctors retire and others have grown weary of more and more bureaucracy from government and private payers. Having faced a difficult financial crisis during the pandemic, more family physicians may move into retirement or seek other professional options.
At the same time, fewer current medical students are choosing specialties in primary care.
“I’ve been trying to raise the alarm about the kind of perilous future of primary care,” said Farzad Mostashari, MD, a top Department of Health & Human Services official in the Obama administration. Dr. Mostashari runs Aledade, a company that helps primary care doctors make the transition from fee-for-service medicine to new payment models.
The American Academy of Family Physicians reports that 70% of primary care physicians are reporting declines in patient volume of 50% or more since March, and 40% have laid off or furloughed staff. The AAFP has joined other primary care and insurance groups in asking HHS for an infusion of cash.
“This is absolutely essential to effectively treat patients today and to maintain their ongoing operations until we overcome this public health emergency,” the groups wrote.
One easy way to help keep primary care doctors afloat would be to pay them not according to what they do, but in a lump sum to keep patients healthy. This move from fee-for-service to what’s known as capitation or value-based care has unfolded gradually and was championed in the Affordable Care Act.
But some experts argue it needs to happen more quickly and they predict that the coronavirus pandemic could finally mark the beginning of the end for doctors who still charge for each service individually. Dr. Mostashari, who spends his time helping doctors make the transition, said in times like these, it would make more sense for primary care doctors to have “a steady monthly revenue stream, and [the doctor] can decide the best way to deliver that care: unlimited texts, phone calls, video calls. The goal is to give you satisfactory outcomes and a great patient experience.”
Still, many physicians, particularly those in solo or small practices, worry about the potential financial risk – especially the possibility of getting paid less if they don’t meet certain benchmarks that the doctors may not be able to directly control.
But with many practices now ground to a halt, or just starting to reopen, those physicians who get paid per patient rather than per service are in a much better position to stay afloat. That model may gain traction as doctors ponder the next pandemic, or the next wave of this one.
Hospitals on the decline?
The pandemic also might lead to less emphasis on hospital-based care. While hospitals in many parts of the country have obviously been full of very sick COVID patients, they have closed down other nonemergency services to preserve supplies and resources to fight the pandemic. People with other ailments have stayed away in droves even when services were available, for fear of catching something worse than what they already have.
Many experts predict that care won’t just snap back when the current emergency wanes. Mark Smith, MD, former president of the California Health Care Foundation, said among consumers, a switch has been flipped. “Overnight it seems we’ve gone from high-touch to no-touch.”
Which is not great for hospitals that have spent millions trying to attract patients to their labor-and-delivery units, orthopedic centers, and other parts of the facility that once generated lots of income.
Even more concerning is that hospitals’ ability to weather the current financial shock varies widely. Those most in danger of closing are in rural and underserved areas, where patients could wind up with even less access to care that is scarce already.
All of which underscores the point that not all these changes will necessarily be good for the health system or society. Financial pressures could end up driving more consolidation, which could push up prices as large groups of hospitals and doctors gain more bargaining clout.
But the changes are definitely happening at a pace few have ever seen, said Ms. Wilensky, “When you’re forced to find different ways of doing things, and you find out they are easier and more efficient, it’s going to be hard to go back to the old way.”
A version of this article originally appeared on Kaiser Health News, which is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
The U.S. health care system is famously resistant to government-imposed change. It took decades to create Medicare and Medicaid, mostly because of opposition from the medical-industrial complex. Then it was nearly another half-century before the passage of the Affordable Care Act.
“Health care is never going back to the way it was before,” said Gail Wilensky, a health economist who ran the Medicare and Medicaid programs for President George H.W. Bush in the early 1990s.
Ms. Wilensky is far from the only longtime observer of the American health care system to marvel at the speed of some long-sought changes. But experts warn that the breakthroughs may not all make the health system work better or make it less expensive.
That said, here are three trends that seem likely to continue.
Telehealth for all
Telehealth is not new; medical professionals have used it to reach patients in rural or remote settings since the late 1980s.
But even while technology has made video visits easier, it has failed to reach critical mass, largely because of political fights. Licensing has been one main obstacle – determining how a doctor in one state can legally treat a patient in a state where the doctor is not licensed.
The other obstacle, not surprisingly, is payment. Should a video visit be reimbursed at the same rate as an in-person visit? Will making it easier for doctors and other medical professionals to use telehealth encourage unnecessary care, thus driving up the nation’s $3.6 trillion health tab even more? Or could it replace care once provided free by phone?
Still, the pandemic has pushed aside those sticking points. Almost overnight, by necessity, every health care provider who can is delivering telemedicine. A new survey from Gallup found the number of patients reporting “virtual” medical visits more than doubled, from 12% to 27%, from late March to mid-May. That is attributable, at least in part, to Medicare having made it easier for doctors to bill for virtual visits.
It’s easy to see why many patients like video visits – there’s no parking to find and pay for, and it takes far less time out of a workday than going to an office.
Doctors and other practitioners seem more ambivalent. On one hand, it can be harder to examine a patient over video and some services just can’t be done via a digital connection. On the other hand, they can see more patients in the same amount of time and may need less support staff and possibly smaller offices if more visits are conducted virtually.
Of course, telemedicine doesn’t work for everyone. Many areas and patients don’t have reliable or robust broadband connections that make video visits work. And some patients, particularly the oldest seniors, lack the technological skills needed to connect.
Primary care doctors in peril
Another trend that has suddenly accelerated is worry over the nation’s dwindling supply of primary care doctors. The exodus of practitioners performing primary care has been a concern over the past several years, as baby boomer doctors retire and others have grown weary of more and more bureaucracy from government and private payers. Having faced a difficult financial crisis during the pandemic, more family physicians may move into retirement or seek other professional options.
At the same time, fewer current medical students are choosing specialties in primary care.
“I’ve been trying to raise the alarm about the kind of perilous future of primary care,” said Farzad Mostashari, MD, a top Department of Health & Human Services official in the Obama administration. Dr. Mostashari runs Aledade, a company that helps primary care doctors make the transition from fee-for-service medicine to new payment models.
The American Academy of Family Physicians reports that 70% of primary care physicians are reporting declines in patient volume of 50% or more since March, and 40% have laid off or furloughed staff. The AAFP has joined other primary care and insurance groups in asking HHS for an infusion of cash.
“This is absolutely essential to effectively treat patients today and to maintain their ongoing operations until we overcome this public health emergency,” the groups wrote.
One easy way to help keep primary care doctors afloat would be to pay them not according to what they do, but in a lump sum to keep patients healthy. This move from fee-for-service to what’s known as capitation or value-based care has unfolded gradually and was championed in the Affordable Care Act.
But some experts argue it needs to happen more quickly and they predict that the coronavirus pandemic could finally mark the beginning of the end for doctors who still charge for each service individually. Dr. Mostashari, who spends his time helping doctors make the transition, said in times like these, it would make more sense for primary care doctors to have “a steady monthly revenue stream, and [the doctor] can decide the best way to deliver that care: unlimited texts, phone calls, video calls. The goal is to give you satisfactory outcomes and a great patient experience.”
Still, many physicians, particularly those in solo or small practices, worry about the potential financial risk – especially the possibility of getting paid less if they don’t meet certain benchmarks that the doctors may not be able to directly control.
But with many practices now ground to a halt, or just starting to reopen, those physicians who get paid per patient rather than per service are in a much better position to stay afloat. That model may gain traction as doctors ponder the next pandemic, or the next wave of this one.
Hospitals on the decline?
The pandemic also might lead to less emphasis on hospital-based care. While hospitals in many parts of the country have obviously been full of very sick COVID patients, they have closed down other nonemergency services to preserve supplies and resources to fight the pandemic. People with other ailments have stayed away in droves even when services were available, for fear of catching something worse than what they already have.
Many experts predict that care won’t just snap back when the current emergency wanes. Mark Smith, MD, former president of the California Health Care Foundation, said among consumers, a switch has been flipped. “Overnight it seems we’ve gone from high-touch to no-touch.”
Which is not great for hospitals that have spent millions trying to attract patients to their labor-and-delivery units, orthopedic centers, and other parts of the facility that once generated lots of income.
Even more concerning is that hospitals’ ability to weather the current financial shock varies widely. Those most in danger of closing are in rural and underserved areas, where patients could wind up with even less access to care that is scarce already.
All of which underscores the point that not all these changes will necessarily be good for the health system or society. Financial pressures could end up driving more consolidation, which could push up prices as large groups of hospitals and doctors gain more bargaining clout.
But the changes are definitely happening at a pace few have ever seen, said Ms. Wilensky, “When you’re forced to find different ways of doing things, and you find out they are easier and more efficient, it’s going to be hard to go back to the old way.”
A version of this article originally appeared on Kaiser Health News, which is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
The U.S. health care system is famously resistant to government-imposed change. It took decades to create Medicare and Medicaid, mostly because of opposition from the medical-industrial complex. Then it was nearly another half-century before the passage of the Affordable Care Act.
“Health care is never going back to the way it was before,” said Gail Wilensky, a health economist who ran the Medicare and Medicaid programs for President George H.W. Bush in the early 1990s.
Ms. Wilensky is far from the only longtime observer of the American health care system to marvel at the speed of some long-sought changes. But experts warn that the breakthroughs may not all make the health system work better or make it less expensive.
That said, here are three trends that seem likely to continue.
Telehealth for all
Telehealth is not new; medical professionals have used it to reach patients in rural or remote settings since the late 1980s.
But even while technology has made video visits easier, it has failed to reach critical mass, largely because of political fights. Licensing has been one main obstacle – determining how a doctor in one state can legally treat a patient in a state where the doctor is not licensed.
The other obstacle, not surprisingly, is payment. Should a video visit be reimbursed at the same rate as an in-person visit? Will making it easier for doctors and other medical professionals to use telehealth encourage unnecessary care, thus driving up the nation’s $3.6 trillion health tab even more? Or could it replace care once provided free by phone?
Still, the pandemic has pushed aside those sticking points. Almost overnight, by necessity, every health care provider who can is delivering telemedicine. A new survey from Gallup found the number of patients reporting “virtual” medical visits more than doubled, from 12% to 27%, from late March to mid-May. That is attributable, at least in part, to Medicare having made it easier for doctors to bill for virtual visits.
It’s easy to see why many patients like video visits – there’s no parking to find and pay for, and it takes far less time out of a workday than going to an office.
Doctors and other practitioners seem more ambivalent. On one hand, it can be harder to examine a patient over video and some services just can’t be done via a digital connection. On the other hand, they can see more patients in the same amount of time and may need less support staff and possibly smaller offices if more visits are conducted virtually.
Of course, telemedicine doesn’t work for everyone. Many areas and patients don’t have reliable or robust broadband connections that make video visits work. And some patients, particularly the oldest seniors, lack the technological skills needed to connect.
Primary care doctors in peril
Another trend that has suddenly accelerated is worry over the nation’s dwindling supply of primary care doctors. The exodus of practitioners performing primary care has been a concern over the past several years, as baby boomer doctors retire and others have grown weary of more and more bureaucracy from government and private payers. Having faced a difficult financial crisis during the pandemic, more family physicians may move into retirement or seek other professional options.
At the same time, fewer current medical students are choosing specialties in primary care.
“I’ve been trying to raise the alarm about the kind of perilous future of primary care,” said Farzad Mostashari, MD, a top Department of Health & Human Services official in the Obama administration. Dr. Mostashari runs Aledade, a company that helps primary care doctors make the transition from fee-for-service medicine to new payment models.
The American Academy of Family Physicians reports that 70% of primary care physicians are reporting declines in patient volume of 50% or more since March, and 40% have laid off or furloughed staff. The AAFP has joined other primary care and insurance groups in asking HHS for an infusion of cash.
“This is absolutely essential to effectively treat patients today and to maintain their ongoing operations until we overcome this public health emergency,” the groups wrote.
One easy way to help keep primary care doctors afloat would be to pay them not according to what they do, but in a lump sum to keep patients healthy. This move from fee-for-service to what’s known as capitation or value-based care has unfolded gradually and was championed in the Affordable Care Act.
But some experts argue it needs to happen more quickly and they predict that the coronavirus pandemic could finally mark the beginning of the end for doctors who still charge for each service individually. Dr. Mostashari, who spends his time helping doctors make the transition, said in times like these, it would make more sense for primary care doctors to have “a steady monthly revenue stream, and [the doctor] can decide the best way to deliver that care: unlimited texts, phone calls, video calls. The goal is to give you satisfactory outcomes and a great patient experience.”
Still, many physicians, particularly those in solo or small practices, worry about the potential financial risk – especially the possibility of getting paid less if they don’t meet certain benchmarks that the doctors may not be able to directly control.
But with many practices now ground to a halt, or just starting to reopen, those physicians who get paid per patient rather than per service are in a much better position to stay afloat. That model may gain traction as doctors ponder the next pandemic, or the next wave of this one.
Hospitals on the decline?
The pandemic also might lead to less emphasis on hospital-based care. While hospitals in many parts of the country have obviously been full of very sick COVID patients, they have closed down other nonemergency services to preserve supplies and resources to fight the pandemic. People with other ailments have stayed away in droves even when services were available, for fear of catching something worse than what they already have.
Many experts predict that care won’t just snap back when the current emergency wanes. Mark Smith, MD, former president of the California Health Care Foundation, said among consumers, a switch has been flipped. “Overnight it seems we’ve gone from high-touch to no-touch.”
Which is not great for hospitals that have spent millions trying to attract patients to their labor-and-delivery units, orthopedic centers, and other parts of the facility that once generated lots of income.
Even more concerning is that hospitals’ ability to weather the current financial shock varies widely. Those most in danger of closing are in rural and underserved areas, where patients could wind up with even less access to care that is scarce already.
All of which underscores the point that not all these changes will necessarily be good for the health system or society. Financial pressures could end up driving more consolidation, which could push up prices as large groups of hospitals and doctors gain more bargaining clout.
But the changes are definitely happening at a pace few have ever seen, said Ms. Wilensky, “When you’re forced to find different ways of doing things, and you find out they are easier and more efficient, it’s going to be hard to go back to the old way.”
A version of this article originally appeared on Kaiser Health News, which is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.