User login
Applying a Text-Search Algorithm to Radiology Reports Can Find More Patients With Pulmonary Nodules Than Radiology Coding Alone (FULL)
Rapid advances in imaging technology have led to better spatial resolution with lower radiation doses to patients. These advances have helped to increase the use of diagnostic chest imaging, particularly in emergency departments and oncology centers, and in screening for coronary artery disease. As a result, there has been an explosion of incidental findings on chest imaging—including indeterminate lung nodules.1,2
Lung nodules are rounded and well-circumscribed lung opacities (≤ 3 cm in diameter) that may present as solitary or multiple lesions in usually asymptomatic patients. Most lung nodules are benign, the result of an infectious or inflammatory process. Nodules that are ≤ 8 mm in diameter, unless they show increase in size over time, often can be safely followed with imaging surveillance. In contrast, lung nodules > 8 mm could represent an early-stage lung cancer, especially among patients with high-risk for developing lung cancer (ie, those with advanced age, heavy tobacco abuse, or emphysema) and should be further assessed with close imaging surveillance, either chest computed tomography (CT) alone or positron-emission tomography (PET)/CT, or tissue biopsy, based on the underlying likelihood of malignancy.
Patients who receive an early-stage lung cancer diagnosis can be offered curative treatments leading to improved 5-year survival rates.3,4 Consequently, health care systems need to be able to identify these nodules accurately, in order to categorize and manage them accordingly to the Fleischner radiographic and American College of Chest Physicians clinical guidelines.5,6 Unfortunately, many hospitals struggle to identify patients with incidental lung nodules found during diagnostic chest and abdominal imaging, due in part to poor adherence to Fleischner guidelines among radiologists for categorizing pulmonary nodules.7,8
The Veterans Health Administration (VHA) system is interested in effectively detecting patients with incidental lung nodules. Veterans have a higher risk of developing lung cancer when compared with the entire US population, mainly due to a higher incidence of tobacco use.6 The prevalence of lung nodules among veterans with significant risk factors for lung cancer is about 60% nationwide, and up to 85% in the Midwest, due to the high prevalence of histoplasmosis.7 However, only a small percentage of these nodules represent an early stage primary lung cancer.
Several Veterans Integrated Service Networks (VISNs) in the VHA use a radiology diagnostic code to systematically identify imaging studies with presence of lung nodules. In VISN 23, which includes Minnesota, North Dakota, South Dakota, Iowa, and portions of neighboring states, the code used to identify these radiology studies is 44. However, there is high variability in the reporting and coding of imaging studies among radiologists, which could lead to misclassifying patients with lung nodules.8
Some studies suggest that using an automated text search algorithm within radiology reports can be a highly effective strategy to identify patients with lung nodules.9,10 In this study, we compared the diagnostic performance of a newly developed text search algorithm applied to radiology reports with the current standard practice of using a radiology diagnostic code for identifying patients with lung nodules at the Iowa City US Department of Veterans Affairs (VA) Health Care System (ICVAHCS) hospital in Iowa.
Methods
Since 2014, The ICVAHCS has used a radiology diagnostic code to identify any imaging studies with lung nodules. The radiologist enters “44” at the end of the reading process using the Nuance Powerscribe 360 radiation reporting system. The code is uploaded into the VHA Corporate Data Warehouse (CDW), and it is located within the radiology exam domain. This strategy was created and implemented by the Minneapolis VA Health Care System in Minnesota for all the VA hospitals in VISN 23. A lung nodule registry nurse was provided with a list of radiology studies flagged with this radiology diagnostic code every 2 weeks. A chart review was then performed for all these studies to determine the presence of a lung nodule. When detected, the ordering health care provider was alerted and given recommendations for managing the nodule.
We initially searched for the radiology studies with a presumptive lung nodule using the radiology code 44 within the CDW. Separately, we applied the text search strategy only to radiology reports from chest and abdomen studies (ie, X-rays, CT, magnetic resonance imaging [MRI], and PET) that contained any of the keyword phrases. The text search strategy was modeled based on a natural language processing (NLP) algorithm developed by the Puget Sound VA Healthcare System in Seattle, Washington to identify lung nodules on radiology reports.9 Our algorithm included a series of text searches using Microsoft SQL. After several simulations using a random group of radiology reports, we chose the keywords: “lung AND nodul”; “pulm AND nodul”; “pulm AND mass”; “lung AND mass”; and “ground glass”. We selected only chest and abdomen studies because on several simulations using a random group of radiology reports, the vast majority of lung nodules were identified on chest and abdomen imaging studies. Also, it would not have been feasible to chart review the approximately 30,000 total radiology reports that were generated during the study period.
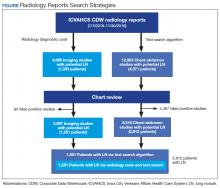
From January 1, 2016 through November 30, 2016, we applied both search strategies independently: radiology diagnostic code for lung nodules to all imaging studies, and text search to all radiology reports of chest and abdomen imaging studies in the CDW (Figure). We also collected demographic (eg, age, sex, race, rurality) and clinical (eg, medical comorbidities, tobacco use) information that were uploaded to the database automatically from CDW using International Statistical Classification of Diseases, Tenth Edition and demographic codes. The VHA uses the Rural-Urban Commuting Areas (RUCA) system to define rurality, which takes into account population density and how closely a community is linked socioeconomically to larger urban centers.11 The protocol was reviewed and approved by the institutional review board of ICVAHCS and the University of Iowa.
The presence of a lung nodule was established by having the lung nodule registry nurse manually review the charts of every patient with a radiology report identified by either code 44 or the text search algorithm. The goal was to ensure that our text search strategy identified all reports with a code 44 to be compliant with VISN expectations. Cases in which a lung nodule was described in the radiology report were considered true positives, and those without a lung nodule description were considered false positives.
We compared the sociodemographic and clinical characteristics of patients with lung nodules between those identified with both code 44 and the text search and those identified with the text search alone. We used χ2 tests for categorical variables (eg, age, gender, RUCA, chronic obstructive pulmonary disease (COPD), smoking status) and t tests for continuous variables (eg, Charlson comorbidity score). A P value ≤ .05 was considered statistically significant. To assess the yield of each search strategy, we determined the number of patients with lung nodules detected by the text search and the radiology diagnostic code. We also calculated the positive predictive value (PPV) and 95% CI of each search strategy.
Results
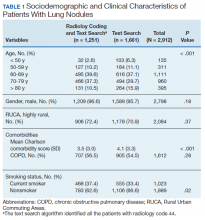
We identified 12,983 radiology studies that required manual review during the study period. We confirmed that 8,516 imaging studies had lung nodules, representing 2,912 patients. Subjects with lung nodules were predominantly male (96%), aged between 60 and 79 years (71%), and lived in a rural area (72%). More than 50% of these patients had COPD and over a third were current smokers (Table 1). The text search algorithm identified all of the patients identified by the radiology diagnostic code (n = 1,251). It also identified an additional 1,661 patients with lung nodules that otherwise would have been missed by the radiology code. Compared with those identified only by the text search, those identified by both the radiology coding and text search were older, had lower Charlson comorbidity scores, and were more likely to be a current smoker.
The text search algorithm identified more than twice as many patients with potential lung nodules compared with the radiology diagnostic code (4,071 vs 1,363) (Table 2). However, the text search algorithm was associated with a much higher number of false positives than was the diagnostic code (1,159 vs 112) and a lower PPV (72% [95% CI, 70.6-73.4] vs 92% [95% CI, 90.6-93.4], respectively). The text search algorithm identified 130 patients with lung nodules of moderate to high risk for malignancy (> 8 mm diameter) that were not identified by the radiology code. When the PPV of each search strategy was calculated based on imaging studies with nodules (most patients had > 1 imaging study), the results remained similar (98% for radiology code and 66% for text search). A larger proportion of the lung nodules detected by code 44 vs the text search algorithm were from CT chest studies.
Discussion
In a population of predominantly older male veterans with significant risk factors for lung cancer and high incidence of incidental lung nodules, applying a text search algorithm on radiology reports identified a substantial number of patients with lung nodules, including some with nodules > 8 mm, that were missed by the radiologist-generated code.9,10 Improving the yield of detection for lung nodules in a population with high risk for lung cancer would increase the likelihood of detecting patients with potentially curable early-stage lung cancers, decreasing lung cancer mortality.
The reasons for the high number of patients with lung nodules missed by the radiology code are unclear. Potential explanations may include the lack of standardization of imaging reports by the radiologists (ie, only 21% of chest CTs used a standardized template describing a lung nodule in our study), a problem well recognized both within and outside VHA.8,12
The text search algorithm identified more patients with lung nodules but had a higher rate of false positives when compared with the diagnostic code. The high rate of false positives resulted in more charts to review and an increased workload for the lung nodule registry team. The challenges presented by an increased workload should be balanced against the potential harms of missing nodules that develop into advanced cancer.
Text Search Adjustments
Refining the text search criteria algorithm and the chart review process may decrease the rate of false positives significantly without affecting detection of lung nodules. In subsequent simulations, we found that by adding an exclusion criteria to text search algorithm to remove reports with specific keywords we could substantially reduce the number of false positive reports without affecting the detection rate of the lung nodules. These exclusion criteria would exclude any reports that: (1) contain “nodul” within the next 8 words after mentioning “no”; (2) contain “clear” within the next 8 words after mentioning “lung” in the text (eg, “lungs appear to be clear”); (3) contain “clear” within the next 4 words after mentioning “otherwise” in the text (eg, “otherwise appear to be clear”). Based on our study results, we further refined the text search strategy by limiting the search to only chest imaging studies. When we applied the revised algorithm to a random sample of imaging reports, we found all the code 44 radiology reports were still captured, but we were able to reduce the number of radiology reports needing review by about 80%.
Although classification approaches are being refined to improve radiology performance in multiple categories of nodules, this study suggests that alternative approaches based on text algorithms can improve the capture of pulmonary nodules that require surveillance. These algorithms also can be used to augment radiologist reporting systems. This represents an investment in resources to build a team that should include a bioinformatics specialist, lung nodule registry personnel (review charts of the detected imaging studies with lung nodules, populating the lung nodule database, and determining and tracking the need of imaging follow up), a lung nodule clinic nurse coordinator, and a dedicated lung nodule clinic pulmonologist.
Radiology departments could employ this text search approach to identify missed nodules and use an audit and feedback system to train radiologists to code lung nodules consistently at the time of the initial reading to avoid delays in identifying patients with nodules. Alternatively, the more widespread use of a standardized CT chest radiology reports using Fleischner or the American College of Radiology Lung Imaging Reporting and Data System (Lung RADS) templates might improve the detection of patients with lung nodules.5,13,14
The VHA system should have an effective strategy for identifying incidental lung nodules during routine radiology examinations. Relying only on radiologists to identify and code pulmonary nodules can lead to missing a significant number of patients with lung nodules and some patients with early stage lung cancer who could receive curative therapy.12,14-16 The use of a standardized algorithm, like a text search strategy, might decrease the risk of variation in the execution and result in a more sensitive detection of patients with lung nodules. The text search strategy might be easily implemented and shared with other hospitals both within and outside the VHA.
Limitations
This study was performed in a single VHA hospital and the findings may not be generalizable to other settings of care. Second, our study design is susceptible to work-up bias because the results of a diagnostic test (eg, chest or abdomen imaging) affected whether the chart review was used to verify the test result. It was not feasible to review the patient records of all radiology studies done at the facility during the study period, consequently complete 2 × 2 tables could not be created to calculate sensitivity, specificity, and negative predictive value.
Conclusion
A text search algorithm of radiology reports increased the detection of patients with lung nodules when compared with radiology diagnostic coding alone. However, the improved detection was associated with a higher rate of false positives, which requires manually reviewing a larger number of patient’s chart reports. Future research and quality improvement should focus on standardizing the radiology reporting process and improving the efficiency and reliability of follow up and tracking of incidental lung nodules.
Acknowledgments
The work reported here was supported by a grant from the Office of Rural Health (N32-FY16Q1-S1-P01577), US Department of Veterans Affairs, Veterans Health Administration. We also had the support from the Veterans Rural Health Resource Center-Iowa City, and the Health Services Research and Development (HSR&D) Service through the Comprehensive Access and Delivery Research and Evaluation (CADRE) Center (REA 09-220).
1. Jacobs PC, Mali WP, Grobbee DE, van der Graaf Y. Prevalence of incidental findings in computed tomographic screening of the chest: a systematic review. Journal of computer assisted tomography. 2008;32(2):214-221.
2. Frank L, Quint LE. Chest CT incidentalomas: thyroid lesions, enlarged mediastinal lymph nodes, and lung nodules. Cancer Imaging. 2012;12(1):41-48.
3. National Institutes of Health, National Cancer Institute, Surveillance, Epidemiology, and End Results Program. Cancer stat facts: lung and bronchus cancer. https://seer.cancer.gov/statfacts/html/lungb.html. Accessed April 8, 2020.
4. Alberg AJ, Brock MV, Ford JG, Samet JM, Spivack SD. Epidemiology of lung cancer: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2013;143(5 Suppl):e1S-e29S.
5. MacMahon H, Naidich DP, Goo JM, et al. Guidelines for Management of Incidental Pulmonary Nodules Detected on CT Images: From the Fleischner Society 2017. Radiology. 2017;284(1):228-243.
6. Zullig LL, Jackson GL, Dorn RA, et al. Cancer incidence among patients of the U.S. Veterans Affairs Health Care System. Mil Med. 2012;177(6):693-701.
7. Kinsinger LS, Anderson C, Kim J, et al. Implementation of lung cancer screening in the Veterans Health Administration. JAMA Intern Med. 2017;177(3):399-406.
8. Iqbal MN, Stott E, Huml AM, et al. What’s in a name? Factors associated with documentation and evaluation of incidental pulmonary nodules. Ann Am Thorac Soc. 2016;13(10):1704-1711.
9. Farjah F, Halgrim S, Buist DS, et al. An automated method for identifying individuals with a lung nodule can be feasibly implemented across health systems. Egems (Wash DC). 2016;4(1):1254.
10. Danforth KN, Early MI, Ngan S, Kosco AE, Zheng C, Gould MK. Automated identification of patients with pulmonary nodules in an integrated health system using administrative health plan data, radiology reports, and natural language processing. J Thorac Oncol. 2012;7(8):1257-1262.
11. US Department of Veterans Affairs, Office of Rural Health. https://www.ruralhealth.va.gov/aboutus/ruralvets.asp. Updated January 28, 2020. Accessed April 8, 2020.
12. Blagev DP, Lloyd JF, Conner K, et al. Follow-up of incidental pulmonary nodules and the radiology report. J Am Coll Radiol. 2016;13(2 suppl):R18-R24.
13. Eisenberg RL, Fleischner S. Ways to improve radiologists’ adherence to Fleischner Society guidelines for management of pulmonary nodules. J Am Coll Radiol. 2013;10(6):439-441.
14. Aberle DR. Implementing lung cancer screening: the US experience. Clin Radiol. 2017;72(5):401-406.
15. Gould MK, Donington J, Lynch WR, et al. Evaluation of individuals with pulmonary nodules: when is it lung cancer? Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2013;143(5 Suppl):e93S-e120S.
16. Callister ME, Baldwin DR. How should pulmonary nodules be optimally investigated and managed? Lung Cancer. 2016;91:48-55.
Rapid advances in imaging technology have led to better spatial resolution with lower radiation doses to patients. These advances have helped to increase the use of diagnostic chest imaging, particularly in emergency departments and oncology centers, and in screening for coronary artery disease. As a result, there has been an explosion of incidental findings on chest imaging—including indeterminate lung nodules.1,2
Lung nodules are rounded and well-circumscribed lung opacities (≤ 3 cm in diameter) that may present as solitary or multiple lesions in usually asymptomatic patients. Most lung nodules are benign, the result of an infectious or inflammatory process. Nodules that are ≤ 8 mm in diameter, unless they show increase in size over time, often can be safely followed with imaging surveillance. In contrast, lung nodules > 8 mm could represent an early-stage lung cancer, especially among patients with high-risk for developing lung cancer (ie, those with advanced age, heavy tobacco abuse, or emphysema) and should be further assessed with close imaging surveillance, either chest computed tomography (CT) alone or positron-emission tomography (PET)/CT, or tissue biopsy, based on the underlying likelihood of malignancy.
Patients who receive an early-stage lung cancer diagnosis can be offered curative treatments leading to improved 5-year survival rates.3,4 Consequently, health care systems need to be able to identify these nodules accurately, in order to categorize and manage them accordingly to the Fleischner radiographic and American College of Chest Physicians clinical guidelines.5,6 Unfortunately, many hospitals struggle to identify patients with incidental lung nodules found during diagnostic chest and abdominal imaging, due in part to poor adherence to Fleischner guidelines among radiologists for categorizing pulmonary nodules.7,8
The Veterans Health Administration (VHA) system is interested in effectively detecting patients with incidental lung nodules. Veterans have a higher risk of developing lung cancer when compared with the entire US population, mainly due to a higher incidence of tobacco use.6 The prevalence of lung nodules among veterans with significant risk factors for lung cancer is about 60% nationwide, and up to 85% in the Midwest, due to the high prevalence of histoplasmosis.7 However, only a small percentage of these nodules represent an early stage primary lung cancer.
Several Veterans Integrated Service Networks (VISNs) in the VHA use a radiology diagnostic code to systematically identify imaging studies with presence of lung nodules. In VISN 23, which includes Minnesota, North Dakota, South Dakota, Iowa, and portions of neighboring states, the code used to identify these radiology studies is 44. However, there is high variability in the reporting and coding of imaging studies among radiologists, which could lead to misclassifying patients with lung nodules.8
Some studies suggest that using an automated text search algorithm within radiology reports can be a highly effective strategy to identify patients with lung nodules.9,10 In this study, we compared the diagnostic performance of a newly developed text search algorithm applied to radiology reports with the current standard practice of using a radiology diagnostic code for identifying patients with lung nodules at the Iowa City US Department of Veterans Affairs (VA) Health Care System (ICVAHCS) hospital in Iowa.
Methods
Since 2014, The ICVAHCS has used a radiology diagnostic code to identify any imaging studies with lung nodules. The radiologist enters “44” at the end of the reading process using the Nuance Powerscribe 360 radiation reporting system. The code is uploaded into the VHA Corporate Data Warehouse (CDW), and it is located within the radiology exam domain. This strategy was created and implemented by the Minneapolis VA Health Care System in Minnesota for all the VA hospitals in VISN 23. A lung nodule registry nurse was provided with a list of radiology studies flagged with this radiology diagnostic code every 2 weeks. A chart review was then performed for all these studies to determine the presence of a lung nodule. When detected, the ordering health care provider was alerted and given recommendations for managing the nodule.
We initially searched for the radiology studies with a presumptive lung nodule using the radiology code 44 within the CDW. Separately, we applied the text search strategy only to radiology reports from chest and abdomen studies (ie, X-rays, CT, magnetic resonance imaging [MRI], and PET) that contained any of the keyword phrases. The text search strategy was modeled based on a natural language processing (NLP) algorithm developed by the Puget Sound VA Healthcare System in Seattle, Washington to identify lung nodules on radiology reports.9 Our algorithm included a series of text searches using Microsoft SQL. After several simulations using a random group of radiology reports, we chose the keywords: “lung AND nodul”; “pulm AND nodul”; “pulm AND mass”; “lung AND mass”; and “ground glass”. We selected only chest and abdomen studies because on several simulations using a random group of radiology reports, the vast majority of lung nodules were identified on chest and abdomen imaging studies. Also, it would not have been feasible to chart review the approximately 30,000 total radiology reports that were generated during the study period.
From January 1, 2016 through November 30, 2016, we applied both search strategies independently: radiology diagnostic code for lung nodules to all imaging studies, and text search to all radiology reports of chest and abdomen imaging studies in the CDW (Figure). We also collected demographic (eg, age, sex, race, rurality) and clinical (eg, medical comorbidities, tobacco use) information that were uploaded to the database automatically from CDW using International Statistical Classification of Diseases, Tenth Edition and demographic codes. The VHA uses the Rural-Urban Commuting Areas (RUCA) system to define rurality, which takes into account population density and how closely a community is linked socioeconomically to larger urban centers.11 The protocol was reviewed and approved by the institutional review board of ICVAHCS and the University of Iowa.
The presence of a lung nodule was established by having the lung nodule registry nurse manually review the charts of every patient with a radiology report identified by either code 44 or the text search algorithm. The goal was to ensure that our text search strategy identified all reports with a code 44 to be compliant with VISN expectations. Cases in which a lung nodule was described in the radiology report were considered true positives, and those without a lung nodule description were considered false positives.
We compared the sociodemographic and clinical characteristics of patients with lung nodules between those identified with both code 44 and the text search and those identified with the text search alone. We used χ2 tests for categorical variables (eg, age, gender, RUCA, chronic obstructive pulmonary disease (COPD), smoking status) and t tests for continuous variables (eg, Charlson comorbidity score). A P value ≤ .05 was considered statistically significant. To assess the yield of each search strategy, we determined the number of patients with lung nodules detected by the text search and the radiology diagnostic code. We also calculated the positive predictive value (PPV) and 95% CI of each search strategy.
Results
We identified 12,983 radiology studies that required manual review during the study period. We confirmed that 8,516 imaging studies had lung nodules, representing 2,912 patients. Subjects with lung nodules were predominantly male (96%), aged between 60 and 79 years (71%), and lived in a rural area (72%). More than 50% of these patients had COPD and over a third were current smokers (Table 1). The text search algorithm identified all of the patients identified by the radiology diagnostic code (n = 1,251). It also identified an additional 1,661 patients with lung nodules that otherwise would have been missed by the radiology code. Compared with those identified only by the text search, those identified by both the radiology coding and text search were older, had lower Charlson comorbidity scores, and were more likely to be a current smoker.
The text search algorithm identified more than twice as many patients with potential lung nodules compared with the radiology diagnostic code (4,071 vs 1,363) (Table 2). However, the text search algorithm was associated with a much higher number of false positives than was the diagnostic code (1,159 vs 112) and a lower PPV (72% [95% CI, 70.6-73.4] vs 92% [95% CI, 90.6-93.4], respectively). The text search algorithm identified 130 patients with lung nodules of moderate to high risk for malignancy (> 8 mm diameter) that were not identified by the radiology code. When the PPV of each search strategy was calculated based on imaging studies with nodules (most patients had > 1 imaging study), the results remained similar (98% for radiology code and 66% for text search). A larger proportion of the lung nodules detected by code 44 vs the text search algorithm were from CT chest studies.
Discussion
In a population of predominantly older male veterans with significant risk factors for lung cancer and high incidence of incidental lung nodules, applying a text search algorithm on radiology reports identified a substantial number of patients with lung nodules, including some with nodules > 8 mm, that were missed by the radiologist-generated code.9,10 Improving the yield of detection for lung nodules in a population with high risk for lung cancer would increase the likelihood of detecting patients with potentially curable early-stage lung cancers, decreasing lung cancer mortality.
The reasons for the high number of patients with lung nodules missed by the radiology code are unclear. Potential explanations may include the lack of standardization of imaging reports by the radiologists (ie, only 21% of chest CTs used a standardized template describing a lung nodule in our study), a problem well recognized both within and outside VHA.8,12
The text search algorithm identified more patients with lung nodules but had a higher rate of false positives when compared with the diagnostic code. The high rate of false positives resulted in more charts to review and an increased workload for the lung nodule registry team. The challenges presented by an increased workload should be balanced against the potential harms of missing nodules that develop into advanced cancer.
Text Search Adjustments
Refining the text search criteria algorithm and the chart review process may decrease the rate of false positives significantly without affecting detection of lung nodules. In subsequent simulations, we found that by adding an exclusion criteria to text search algorithm to remove reports with specific keywords we could substantially reduce the number of false positive reports without affecting the detection rate of the lung nodules. These exclusion criteria would exclude any reports that: (1) contain “nodul” within the next 8 words after mentioning “no”; (2) contain “clear” within the next 8 words after mentioning “lung” in the text (eg, “lungs appear to be clear”); (3) contain “clear” within the next 4 words after mentioning “otherwise” in the text (eg, “otherwise appear to be clear”). Based on our study results, we further refined the text search strategy by limiting the search to only chest imaging studies. When we applied the revised algorithm to a random sample of imaging reports, we found all the code 44 radiology reports were still captured, but we were able to reduce the number of radiology reports needing review by about 80%.
Although classification approaches are being refined to improve radiology performance in multiple categories of nodules, this study suggests that alternative approaches based on text algorithms can improve the capture of pulmonary nodules that require surveillance. These algorithms also can be used to augment radiologist reporting systems. This represents an investment in resources to build a team that should include a bioinformatics specialist, lung nodule registry personnel (review charts of the detected imaging studies with lung nodules, populating the lung nodule database, and determining and tracking the need of imaging follow up), a lung nodule clinic nurse coordinator, and a dedicated lung nodule clinic pulmonologist.
Radiology departments could employ this text search approach to identify missed nodules and use an audit and feedback system to train radiologists to code lung nodules consistently at the time of the initial reading to avoid delays in identifying patients with nodules. Alternatively, the more widespread use of a standardized CT chest radiology reports using Fleischner or the American College of Radiology Lung Imaging Reporting and Data System (Lung RADS) templates might improve the detection of patients with lung nodules.5,13,14
The VHA system should have an effective strategy for identifying incidental lung nodules during routine radiology examinations. Relying only on radiologists to identify and code pulmonary nodules can lead to missing a significant number of patients with lung nodules and some patients with early stage lung cancer who could receive curative therapy.12,14-16 The use of a standardized algorithm, like a text search strategy, might decrease the risk of variation in the execution and result in a more sensitive detection of patients with lung nodules. The text search strategy might be easily implemented and shared with other hospitals both within and outside the VHA.
Limitations
This study was performed in a single VHA hospital and the findings may not be generalizable to other settings of care. Second, our study design is susceptible to work-up bias because the results of a diagnostic test (eg, chest or abdomen imaging) affected whether the chart review was used to verify the test result. It was not feasible to review the patient records of all radiology studies done at the facility during the study period, consequently complete 2 × 2 tables could not be created to calculate sensitivity, specificity, and negative predictive value.
Conclusion
A text search algorithm of radiology reports increased the detection of patients with lung nodules when compared with radiology diagnostic coding alone. However, the improved detection was associated with a higher rate of false positives, which requires manually reviewing a larger number of patient’s chart reports. Future research and quality improvement should focus on standardizing the radiology reporting process and improving the efficiency and reliability of follow up and tracking of incidental lung nodules.
Acknowledgments
The work reported here was supported by a grant from the Office of Rural Health (N32-FY16Q1-S1-P01577), US Department of Veterans Affairs, Veterans Health Administration. We also had the support from the Veterans Rural Health Resource Center-Iowa City, and the Health Services Research and Development (HSR&D) Service through the Comprehensive Access and Delivery Research and Evaluation (CADRE) Center (REA 09-220).
Rapid advances in imaging technology have led to better spatial resolution with lower radiation doses to patients. These advances have helped to increase the use of diagnostic chest imaging, particularly in emergency departments and oncology centers, and in screening for coronary artery disease. As a result, there has been an explosion of incidental findings on chest imaging—including indeterminate lung nodules.1,2
Lung nodules are rounded and well-circumscribed lung opacities (≤ 3 cm in diameter) that may present as solitary or multiple lesions in usually asymptomatic patients. Most lung nodules are benign, the result of an infectious or inflammatory process. Nodules that are ≤ 8 mm in diameter, unless they show increase in size over time, often can be safely followed with imaging surveillance. In contrast, lung nodules > 8 mm could represent an early-stage lung cancer, especially among patients with high-risk for developing lung cancer (ie, those with advanced age, heavy tobacco abuse, or emphysema) and should be further assessed with close imaging surveillance, either chest computed tomography (CT) alone or positron-emission tomography (PET)/CT, or tissue biopsy, based on the underlying likelihood of malignancy.
Patients who receive an early-stage lung cancer diagnosis can be offered curative treatments leading to improved 5-year survival rates.3,4 Consequently, health care systems need to be able to identify these nodules accurately, in order to categorize and manage them accordingly to the Fleischner radiographic and American College of Chest Physicians clinical guidelines.5,6 Unfortunately, many hospitals struggle to identify patients with incidental lung nodules found during diagnostic chest and abdominal imaging, due in part to poor adherence to Fleischner guidelines among radiologists for categorizing pulmonary nodules.7,8
The Veterans Health Administration (VHA) system is interested in effectively detecting patients with incidental lung nodules. Veterans have a higher risk of developing lung cancer when compared with the entire US population, mainly due to a higher incidence of tobacco use.6 The prevalence of lung nodules among veterans with significant risk factors for lung cancer is about 60% nationwide, and up to 85% in the Midwest, due to the high prevalence of histoplasmosis.7 However, only a small percentage of these nodules represent an early stage primary lung cancer.
Several Veterans Integrated Service Networks (VISNs) in the VHA use a radiology diagnostic code to systematically identify imaging studies with presence of lung nodules. In VISN 23, which includes Minnesota, North Dakota, South Dakota, Iowa, and portions of neighboring states, the code used to identify these radiology studies is 44. However, there is high variability in the reporting and coding of imaging studies among radiologists, which could lead to misclassifying patients with lung nodules.8
Some studies suggest that using an automated text search algorithm within radiology reports can be a highly effective strategy to identify patients with lung nodules.9,10 In this study, we compared the diagnostic performance of a newly developed text search algorithm applied to radiology reports with the current standard practice of using a radiology diagnostic code for identifying patients with lung nodules at the Iowa City US Department of Veterans Affairs (VA) Health Care System (ICVAHCS) hospital in Iowa.
Methods
Since 2014, The ICVAHCS has used a radiology diagnostic code to identify any imaging studies with lung nodules. The radiologist enters “44” at the end of the reading process using the Nuance Powerscribe 360 radiation reporting system. The code is uploaded into the VHA Corporate Data Warehouse (CDW), and it is located within the radiology exam domain. This strategy was created and implemented by the Minneapolis VA Health Care System in Minnesota for all the VA hospitals in VISN 23. A lung nodule registry nurse was provided with a list of radiology studies flagged with this radiology diagnostic code every 2 weeks. A chart review was then performed for all these studies to determine the presence of a lung nodule. When detected, the ordering health care provider was alerted and given recommendations for managing the nodule.
We initially searched for the radiology studies with a presumptive lung nodule using the radiology code 44 within the CDW. Separately, we applied the text search strategy only to radiology reports from chest and abdomen studies (ie, X-rays, CT, magnetic resonance imaging [MRI], and PET) that contained any of the keyword phrases. The text search strategy was modeled based on a natural language processing (NLP) algorithm developed by the Puget Sound VA Healthcare System in Seattle, Washington to identify lung nodules on radiology reports.9 Our algorithm included a series of text searches using Microsoft SQL. After several simulations using a random group of radiology reports, we chose the keywords: “lung AND nodul”; “pulm AND nodul”; “pulm AND mass”; “lung AND mass”; and “ground glass”. We selected only chest and abdomen studies because on several simulations using a random group of radiology reports, the vast majority of lung nodules were identified on chest and abdomen imaging studies. Also, it would not have been feasible to chart review the approximately 30,000 total radiology reports that were generated during the study period.
From January 1, 2016 through November 30, 2016, we applied both search strategies independently: radiology diagnostic code for lung nodules to all imaging studies, and text search to all radiology reports of chest and abdomen imaging studies in the CDW (Figure). We also collected demographic (eg, age, sex, race, rurality) and clinical (eg, medical comorbidities, tobacco use) information that were uploaded to the database automatically from CDW using International Statistical Classification of Diseases, Tenth Edition and demographic codes. The VHA uses the Rural-Urban Commuting Areas (RUCA) system to define rurality, which takes into account population density and how closely a community is linked socioeconomically to larger urban centers.11 The protocol was reviewed and approved by the institutional review board of ICVAHCS and the University of Iowa.
The presence of a lung nodule was established by having the lung nodule registry nurse manually review the charts of every patient with a radiology report identified by either code 44 or the text search algorithm. The goal was to ensure that our text search strategy identified all reports with a code 44 to be compliant with VISN expectations. Cases in which a lung nodule was described in the radiology report were considered true positives, and those without a lung nodule description were considered false positives.
We compared the sociodemographic and clinical characteristics of patients with lung nodules between those identified with both code 44 and the text search and those identified with the text search alone. We used χ2 tests for categorical variables (eg, age, gender, RUCA, chronic obstructive pulmonary disease (COPD), smoking status) and t tests for continuous variables (eg, Charlson comorbidity score). A P value ≤ .05 was considered statistically significant. To assess the yield of each search strategy, we determined the number of patients with lung nodules detected by the text search and the radiology diagnostic code. We also calculated the positive predictive value (PPV) and 95% CI of each search strategy.
Results
We identified 12,983 radiology studies that required manual review during the study period. We confirmed that 8,516 imaging studies had lung nodules, representing 2,912 patients. Subjects with lung nodules were predominantly male (96%), aged between 60 and 79 years (71%), and lived in a rural area (72%). More than 50% of these patients had COPD and over a third were current smokers (Table 1). The text search algorithm identified all of the patients identified by the radiology diagnostic code (n = 1,251). It also identified an additional 1,661 patients with lung nodules that otherwise would have been missed by the radiology code. Compared with those identified only by the text search, those identified by both the radiology coding and text search were older, had lower Charlson comorbidity scores, and were more likely to be a current smoker.
The text search algorithm identified more than twice as many patients with potential lung nodules compared with the radiology diagnostic code (4,071 vs 1,363) (Table 2). However, the text search algorithm was associated with a much higher number of false positives than was the diagnostic code (1,159 vs 112) and a lower PPV (72% [95% CI, 70.6-73.4] vs 92% [95% CI, 90.6-93.4], respectively). The text search algorithm identified 130 patients with lung nodules of moderate to high risk for malignancy (> 8 mm diameter) that were not identified by the radiology code. When the PPV of each search strategy was calculated based on imaging studies with nodules (most patients had > 1 imaging study), the results remained similar (98% for radiology code and 66% for text search). A larger proportion of the lung nodules detected by code 44 vs the text search algorithm were from CT chest studies.
Discussion
In a population of predominantly older male veterans with significant risk factors for lung cancer and high incidence of incidental lung nodules, applying a text search algorithm on radiology reports identified a substantial number of patients with lung nodules, including some with nodules > 8 mm, that were missed by the radiologist-generated code.9,10 Improving the yield of detection for lung nodules in a population with high risk for lung cancer would increase the likelihood of detecting patients with potentially curable early-stage lung cancers, decreasing lung cancer mortality.
The reasons for the high number of patients with lung nodules missed by the radiology code are unclear. Potential explanations may include the lack of standardization of imaging reports by the radiologists (ie, only 21% of chest CTs used a standardized template describing a lung nodule in our study), a problem well recognized both within and outside VHA.8,12
The text search algorithm identified more patients with lung nodules but had a higher rate of false positives when compared with the diagnostic code. The high rate of false positives resulted in more charts to review and an increased workload for the lung nodule registry team. The challenges presented by an increased workload should be balanced against the potential harms of missing nodules that develop into advanced cancer.
Text Search Adjustments
Refining the text search criteria algorithm and the chart review process may decrease the rate of false positives significantly without affecting detection of lung nodules. In subsequent simulations, we found that by adding an exclusion criteria to text search algorithm to remove reports with specific keywords we could substantially reduce the number of false positive reports without affecting the detection rate of the lung nodules. These exclusion criteria would exclude any reports that: (1) contain “nodul” within the next 8 words after mentioning “no”; (2) contain “clear” within the next 8 words after mentioning “lung” in the text (eg, “lungs appear to be clear”); (3) contain “clear” within the next 4 words after mentioning “otherwise” in the text (eg, “otherwise appear to be clear”). Based on our study results, we further refined the text search strategy by limiting the search to only chest imaging studies. When we applied the revised algorithm to a random sample of imaging reports, we found all the code 44 radiology reports were still captured, but we were able to reduce the number of radiology reports needing review by about 80%.
Although classification approaches are being refined to improve radiology performance in multiple categories of nodules, this study suggests that alternative approaches based on text algorithms can improve the capture of pulmonary nodules that require surveillance. These algorithms also can be used to augment radiologist reporting systems. This represents an investment in resources to build a team that should include a bioinformatics specialist, lung nodule registry personnel (review charts of the detected imaging studies with lung nodules, populating the lung nodule database, and determining and tracking the need of imaging follow up), a lung nodule clinic nurse coordinator, and a dedicated lung nodule clinic pulmonologist.
Radiology departments could employ this text search approach to identify missed nodules and use an audit and feedback system to train radiologists to code lung nodules consistently at the time of the initial reading to avoid delays in identifying patients with nodules. Alternatively, the more widespread use of a standardized CT chest radiology reports using Fleischner or the American College of Radiology Lung Imaging Reporting and Data System (Lung RADS) templates might improve the detection of patients with lung nodules.5,13,14
The VHA system should have an effective strategy for identifying incidental lung nodules during routine radiology examinations. Relying only on radiologists to identify and code pulmonary nodules can lead to missing a significant number of patients with lung nodules and some patients with early stage lung cancer who could receive curative therapy.12,14-16 The use of a standardized algorithm, like a text search strategy, might decrease the risk of variation in the execution and result in a more sensitive detection of patients with lung nodules. The text search strategy might be easily implemented and shared with other hospitals both within and outside the VHA.
Limitations
This study was performed in a single VHA hospital and the findings may not be generalizable to other settings of care. Second, our study design is susceptible to work-up bias because the results of a diagnostic test (eg, chest or abdomen imaging) affected whether the chart review was used to verify the test result. It was not feasible to review the patient records of all radiology studies done at the facility during the study period, consequently complete 2 × 2 tables could not be created to calculate sensitivity, specificity, and negative predictive value.
Conclusion
A text search algorithm of radiology reports increased the detection of patients with lung nodules when compared with radiology diagnostic coding alone. However, the improved detection was associated with a higher rate of false positives, which requires manually reviewing a larger number of patient’s chart reports. Future research and quality improvement should focus on standardizing the radiology reporting process and improving the efficiency and reliability of follow up and tracking of incidental lung nodules.
Acknowledgments
The work reported here was supported by a grant from the Office of Rural Health (N32-FY16Q1-S1-P01577), US Department of Veterans Affairs, Veterans Health Administration. We also had the support from the Veterans Rural Health Resource Center-Iowa City, and the Health Services Research and Development (HSR&D) Service through the Comprehensive Access and Delivery Research and Evaluation (CADRE) Center (REA 09-220).
1. Jacobs PC, Mali WP, Grobbee DE, van der Graaf Y. Prevalence of incidental findings in computed tomographic screening of the chest: a systematic review. Journal of computer assisted tomography. 2008;32(2):214-221.
2. Frank L, Quint LE. Chest CT incidentalomas: thyroid lesions, enlarged mediastinal lymph nodes, and lung nodules. Cancer Imaging. 2012;12(1):41-48.
3. National Institutes of Health, National Cancer Institute, Surveillance, Epidemiology, and End Results Program. Cancer stat facts: lung and bronchus cancer. https://seer.cancer.gov/statfacts/html/lungb.html. Accessed April 8, 2020.
4. Alberg AJ, Brock MV, Ford JG, Samet JM, Spivack SD. Epidemiology of lung cancer: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2013;143(5 Suppl):e1S-e29S.
5. MacMahon H, Naidich DP, Goo JM, et al. Guidelines for Management of Incidental Pulmonary Nodules Detected on CT Images: From the Fleischner Society 2017. Radiology. 2017;284(1):228-243.
6. Zullig LL, Jackson GL, Dorn RA, et al. Cancer incidence among patients of the U.S. Veterans Affairs Health Care System. Mil Med. 2012;177(6):693-701.
7. Kinsinger LS, Anderson C, Kim J, et al. Implementation of lung cancer screening in the Veterans Health Administration. JAMA Intern Med. 2017;177(3):399-406.
8. Iqbal MN, Stott E, Huml AM, et al. What’s in a name? Factors associated with documentation and evaluation of incidental pulmonary nodules. Ann Am Thorac Soc. 2016;13(10):1704-1711.
9. Farjah F, Halgrim S, Buist DS, et al. An automated method for identifying individuals with a lung nodule can be feasibly implemented across health systems. Egems (Wash DC). 2016;4(1):1254.
10. Danforth KN, Early MI, Ngan S, Kosco AE, Zheng C, Gould MK. Automated identification of patients with pulmonary nodules in an integrated health system using administrative health plan data, radiology reports, and natural language processing. J Thorac Oncol. 2012;7(8):1257-1262.
11. US Department of Veterans Affairs, Office of Rural Health. https://www.ruralhealth.va.gov/aboutus/ruralvets.asp. Updated January 28, 2020. Accessed April 8, 2020.
12. Blagev DP, Lloyd JF, Conner K, et al. Follow-up of incidental pulmonary nodules and the radiology report. J Am Coll Radiol. 2016;13(2 suppl):R18-R24.
13. Eisenberg RL, Fleischner S. Ways to improve radiologists’ adherence to Fleischner Society guidelines for management of pulmonary nodules. J Am Coll Radiol. 2013;10(6):439-441.
14. Aberle DR. Implementing lung cancer screening: the US experience. Clin Radiol. 2017;72(5):401-406.
15. Gould MK, Donington J, Lynch WR, et al. Evaluation of individuals with pulmonary nodules: when is it lung cancer? Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2013;143(5 Suppl):e93S-e120S.
16. Callister ME, Baldwin DR. How should pulmonary nodules be optimally investigated and managed? Lung Cancer. 2016;91:48-55.
1. Jacobs PC, Mali WP, Grobbee DE, van der Graaf Y. Prevalence of incidental findings in computed tomographic screening of the chest: a systematic review. Journal of computer assisted tomography. 2008;32(2):214-221.
2. Frank L, Quint LE. Chest CT incidentalomas: thyroid lesions, enlarged mediastinal lymph nodes, and lung nodules. Cancer Imaging. 2012;12(1):41-48.
3. National Institutes of Health, National Cancer Institute, Surveillance, Epidemiology, and End Results Program. Cancer stat facts: lung and bronchus cancer. https://seer.cancer.gov/statfacts/html/lungb.html. Accessed April 8, 2020.
4. Alberg AJ, Brock MV, Ford JG, Samet JM, Spivack SD. Epidemiology of lung cancer: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2013;143(5 Suppl):e1S-e29S.
5. MacMahon H, Naidich DP, Goo JM, et al. Guidelines for Management of Incidental Pulmonary Nodules Detected on CT Images: From the Fleischner Society 2017. Radiology. 2017;284(1):228-243.
6. Zullig LL, Jackson GL, Dorn RA, et al. Cancer incidence among patients of the U.S. Veterans Affairs Health Care System. Mil Med. 2012;177(6):693-701.
7. Kinsinger LS, Anderson C, Kim J, et al. Implementation of lung cancer screening in the Veterans Health Administration. JAMA Intern Med. 2017;177(3):399-406.
8. Iqbal MN, Stott E, Huml AM, et al. What’s in a name? Factors associated with documentation and evaluation of incidental pulmonary nodules. Ann Am Thorac Soc. 2016;13(10):1704-1711.
9. Farjah F, Halgrim S, Buist DS, et al. An automated method for identifying individuals with a lung nodule can be feasibly implemented across health systems. Egems (Wash DC). 2016;4(1):1254.
10. Danforth KN, Early MI, Ngan S, Kosco AE, Zheng C, Gould MK. Automated identification of patients with pulmonary nodules in an integrated health system using administrative health plan data, radiology reports, and natural language processing. J Thorac Oncol. 2012;7(8):1257-1262.
11. US Department of Veterans Affairs, Office of Rural Health. https://www.ruralhealth.va.gov/aboutus/ruralvets.asp. Updated January 28, 2020. Accessed April 8, 2020.
12. Blagev DP, Lloyd JF, Conner K, et al. Follow-up of incidental pulmonary nodules and the radiology report. J Am Coll Radiol. 2016;13(2 suppl):R18-R24.
13. Eisenberg RL, Fleischner S. Ways to improve radiologists’ adherence to Fleischner Society guidelines for management of pulmonary nodules. J Am Coll Radiol. 2013;10(6):439-441.
14. Aberle DR. Implementing lung cancer screening: the US experience. Clin Radiol. 2017;72(5):401-406.
15. Gould MK, Donington J, Lynch WR, et al. Evaluation of individuals with pulmonary nodules: when is it lung cancer? Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2013;143(5 Suppl):e93S-e120S.
16. Callister ME, Baldwin DR. How should pulmonary nodules be optimally investigated and managed? Lung Cancer. 2016;91:48-55.
Incidental Findings of Pulmonary and Hilar Malignancy by Low-Resolution Computed Tomography Used in Myocardial Perfusion Imaging (FULL)
Single-photon emission computed tomography (SPECT) myocardial perfusion imaging (MPI) is a well-established technique for the evaluation of coronary artery disease (CAD).1 To improve image quality, low-resolution computed tomography (CT) is used commonly for anatomical correct and artifact attenuation during SPECT MPI.2 The low resolution, unenhanced CT images are considered low quality and are, therefore, labeled by the manufacturer as nondiagnostic. The CT portion of the MPI in many centers is used only for image fusion and attenuation correction, and these images are not routinely reviewed or reported by cardiologists.
Incidental findings by these low-resolution CT were frequent. However, clinically significant findings, including lung cancer, although relatively infrequent, were serious enough for major clinical management.3-5 Currently, there are no consensus recommendations for reviewing low-resolution CT images or the interpretation of such incidental findings during cardiac MPI.6 Clinically, low-dose CT were used for early detection and screening of lung cancer and were associated with reduced lung-cancer and any cause mortality in National Lung Screening Trial (NLST).7,8 Therefore, low-dose CT is recommended for lung cancer screening of high-risk patients by the US Preventive Service Task Force (USPSTF).9 In the veteran population, current and past smoking history are more common when compared with the general population; therefore, veterans are potentially at increased risk of lung cancer.10 In this study, we did not intend to use low-resolution CT for lung cancer screening or detection but rather to identify and report incidental findings of pulmonary/hilar malignancy detected during cardiac MPI.
Methods
The Siemens’ (Munich, Germany) Symbia Intevo Excel SPECT/CT MPI cameras with dedicated cardiac collimators were used at both the Dwight D. Eisenhower VA Medical Center (VAMC) in Leavenworth, Kansas and Colmery-O'Neil VAMC in Topeka, Kansas. The integrated CT scanner (x-ray tube current 30 to 240 mA; voltage 110 Kv with a 40 kW power generator) has the capability to image up to a 2-slice/rotation, each of 5.0 mm per slice with a scan time of about 30 seconds. The SPECT/CT gamma camera has a low energy (140 KeV), high resolution, parallel hole collimator with IQ SPECT capabilities.
The radiation dose received by the patients were expressed in dose length product (DLP), which reflects the total energy absorbed by the patient and represents integrated dose in terms of the total scan length. Additionally, each patients received 2 injections of Technetium Tc 99m sestamibi (1-day Protocol: 10 mCi rest injection, 30 mCi stress injection: 2-day Protocol for patients weighing > 350 pounds: 30 mCi at rest injection and 30 mCi at stress injection) for myocardial perfusion imaging.
All CT images and cardiac MPI findings were reviewed and reported contemporaneously by 1 of 2 experienced, board-certified radiologists who were blinded to patients’ clinical information except the indication for the cardiac stress testing. When suspicious pulmonary/hilar nodules or masses were detected, these findings and recommendations for further evaluation were conveyed to primary care provider or ordering physician via the electronic health record system.
All CT images were reviewed with cardiac MPI from September 1, 2017 to August 31, 2018. When pulmonary/hilar malignancies were identified, the health records were reviewed. Patients with known history of prior pulmonary malignancy were excluded from the study.
Results
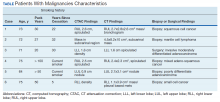
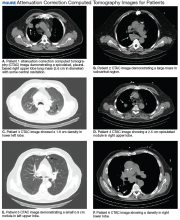
A total of 1,098 patients underwent cardiac MPI during the study period. When the CT imaging and cardiac MPI were reviewed, incidental findings led to the diagnosis of lung cancer in 5 patients and hilar mantle cell lymphoma in 1 patient. Their clinical characteristics, CT findings, and types of malignancies for these 6 patients are summarized in the Table and Figure. Only 0.55% (6 of 1,098) patients were found to have incidental pulmonary/hilar malignancy with the cardiac evaluation low-resolution CT. Four patients with prior, known history of lung cancer were excluded from the study.
For the 6 patients found to have cancer, the average CT radiation dose during the cardiac MPI was 100 mGy-cm (range, 77 -133 mCy-cm). The subsequent chest CT with or without contrast delivered a radiation dose of 726.4 mGy-cm (range, 279.4 - 1,075 mGy-cm).
A total of 79 (7.2%) patients were found to have significant pulmonary nodules that required further evaluation; after CT examination, 32 patients had findings of benign nature and required no further follow-up; the other 47 patients are being followed according to the Fleischner Society 2017 guidelines for pulmonary nodules.11 The follow-up findings on these patients are not within the scope of this report.
Discussion
Although incidental findings on low-resolution CT during cardiac MPI are frequent, clinically significant findings are less common. However, some incidental findings may be of important clinical significance.3-5 A multicenter analysis by Coward and colleagues reported that 2.4% findings on low-resolution CT were significant enough to warrant follow-up tests, but only 0.2% were deemed potentially detrimental to patient outcomes (ie, pathology confirmed malignancies).12 Thus, the authors suggested that routine reporting of incidental findings on low-dose CT images was not beneficial.12,13
Currently, the majority of cardiac MPIs are reviewed and interpreted by nuclear cardiologists, the use of hybrid SPECT/CT for attenuation correction give rise of issue of reviewing and interpreting these CT images during cardiac MPI. Since low-dose, low-resolution CT are considered nondiagnostic, these images are not routinely and readily reviewed by cardiologists who are not trained or skilled in CT interpretations.
Studies of high-resolution cardiac CT (including multidetector CT with contrast) suggest that incidental extracardiac findings should always be reported as there was a 0.7% incidence of previously unknown malignancies, while others have argued against “performing large field reconstructionsfor the explicit purpose of screening as it will lead to additional cost, liability and anxiety without proven benefits.”14-16 A review of incidental findings of cardiac CT by Earls suggested that all cardiac CT should be reconstructed in the maximal field of view available and images should be adequately reviewed to detect pathological findings.17 This led to an interesting discussion by Douglas and colleagues regarding the role of cardiologists and radiologists in this issue.18 Currently there is no uniform or consensus recommendations regarding incidental findings during cardiac CT imaging. Guidances range from no recommendations to optional reporting or mandatory reporting.19-23
Risk Factors for Veterans
Lung cancer is the second most common cancer and the leading cause of cancer-related death in the US.24 Smoking is the most important risk factor for lung cancer and CAD.25 Current or past smoking are more common among the veterans.10 According to a report for the US Centers for Disease Control and Prevention report, about 29.2% US veterans use tobacco products between 2010-2015, which is similar to the rate reported in 1997.26
When low-dose CT was used for lung cancer screening, it was associated with a 20.0% reduction in lung cancer mortality and a 6.7% reduction in any cause mortality.7 Currently, the US Preventive Services Task Force (USPSTF) recommends annual low-dose CT screening for lung cancer in high-risk adults that includes patients aged 55 to 80 years who have a 30-pack-year smoking history and currently smoke or have quit within the past 15 years.8
It is likely that the cardiac patients in this study might have pulmonary malignancy mortality similar to those reported in the NLST. While other studies have shown a low incidence (0.2%) of detection of malignancy by low-resolution CT during cardiac MPI,12,13 in this study we found pulmonary or hilar malignancy in 0.55% of patients.The higher incidence of malignancy in our study might be due in part to differences in the patient population studied (ie, our veterans patients have a higher proportion of current or past smoking history).10
The CT used in this study is part of the cardiac imaging process. Therefore, there was no additional radiation exposure besides that of the cardiac MPI for patients. Despite the limitations of low-resolution CT, which may miss small lesions, this study showed 0.55% incidence of incidental detection of pulmonary/hilar malignancy. This is comparable with 0.65%/year of diagnosing lung cancer using low-dose CT for lung cancer screening in NLST.8
Two of the 5 study patients who were found to have lung cancer, had quit smoking > 15 years previously and thus would not be considered as high-risk for lung cancer screening according to USPSTF guideline. These patients would not have been candidates for annual low-dose CT lung cancer screening. This study suggests that it is appropriate and necessary to review the low-resolution CT images for incidental findings during cardiac MPI.
Limitations
The study was retrospective in nature and limited by its small number of patients. The CT modality used in the study also has limitations, including low resolution, respiratory motion artifacts, and scans that did not include the entire chest area. Therefore, small and apical lesions may have been missed. However, both sets of CT at rest and after stress were reviewed to reduce or minimize the effects of respiratory motion artifacts. The true prevalence or incidence of pulmonary/hilar malignancies may have been higher than reported here. Our study population of veterans may not be representative of the general population with regards to gender (as most of our veteran patient population are of male gender, vs general population), smoking history, or lung cancer risk, thus the results should be interpreted with caution.
Conclusion
Low-resolution CTs used for attenuation correction during cardiac MPI should be routinely reviewed and interpreted by a physician or radiologist skilled in CT interpretation in order to identify incidental findings of pulmonary/hilar malignancy. This would require close collaboration between cardiologists and radiologists in the field to ensure unfragmented and high-quality patient care.
Acknowledgements
We want to thank all the staffs in cardiology and radiology department on both campuses for their dedication for our patients. Special thanks to Laura Knox, Radiation Safety Officer, Nuclear Medicine Supervisor for her technical assistance.
1. Hendel RC, Berman DS, Di Carli MF, et al. ACCF/ASNC/ACR/AHA/ASE/SCCT/SCMR/SNM 2009 appropriate use criteria for cardiac radionuclide imaging: a report of the American College of Cardiology Foundation Appropriate Use Criteria Task Force, the American Society of Nuclear Cardiology, the American College of Radiology, the American Heart Association, the American Society of Echocardiography, the Society of Cardiovascular Computed Tomography, the Society for Cardiovascular Magnetic Resonance, and the Society of Nuclear Medicine. Circulation. 2009;119(22):e561-e587.
2. Hendel RC, Corbett JR, Cullom SJ, DePuey EG, Garcia EV, Bateman TM. The value and practice of attenuation correction for myocardial perfusion SPECT imaging: a joint position statement from the American Society of Nuclear Cardiology and the Society of Nuclear Medicine. J Nucl Cardiol. 2002;9(1):135–143.
3. Coward J, Nightingale J, Hogg P. The clinical dilemma of incidental findings on the low-resolution CT images from SPECT/CT MPI studies. J Nucl Med Technol. 2016;44(3):167-172.
4. Osman MM, Cohade C, Fishman E, Wahl RL. Clinically significant incidental findings on the unenhanced CT portion of PET/CT studies: frequency in 250 patients. J Nucl Med. 2005;46(8):1352-1355.
5. Goetze S, Pannu HK, Wahl RL. Clinically significant abnormal findings on the “nondiagnostic” CT portion of low-amperage-CT attenuation-corrected myocardial perfusion SPECT/CT studies. J Nucl Med. 2006;47(8):1312-1318.
6. American College of Cardiology Foundation Task Force on Expert Consensus Documents, Mark DB, Berman DS, et al. ACCF/ACR/AHA/NASCI/SAIP/SCAI/SCCT 2010 expert consensus document on coronary computed tomographic angiography: a report of the American College of Cardiology Foundation Task Force on Expert Consensus Documents. J Am Coll Cardiol. 2010;55(23):2663-2699.
7. Diederich S, Wormanns D, Semik M, et al. Screening for early lung cancer with low-dose spiral CT: prevalence in 817 asymptomatic smokers. Radiology. 2002;222(3):773-781.
8. National Lung Screening Trial Research Team, Aberle DR, Adams AM, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med. 2011;365(5):395-409.
9. Moyer VA; U.S. Preventive Services Task Force. Screening for lung cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;160(5):330-338.
10. McKinney WP, McIntire DD, Carmody TJ, Joseph A. Comparing the smoking behavior of veterans and nonveterans. Public Health Rep. 1997;112(3):212-218.
11. MacMahon H, Naidich DP, Goo JM, et al. Guidelines for Management of Incidental Pulmonary Nodules Detected on CT Images: From the Fleischner Society 2017. Radiology. 2017;284(1):228-243.
12. Coward J, Lawson R, Kane T, et al. Multi-centre analysis of incidental findings on low-resolution CT attenuation correction images. Br J Radiol. 2014;87(1042):20130701.
13. Coward J, Lawson R, Kane T, et al. Multicentre analysis of incidental findings on low-resolution CT attenuation correction images: an extended study. Br J Radiol. 2015;88(1056):20150555.
14. Haller S, Kaiser C, Buser P, Bongartz G, Bremerich J. Coronary artery imaging with contrast-enhanced MDCT: extracardiac findings. AJR Am J Roentgenol. 2006;187(1):105-110.
15. Flor N, Di Leo G, Squarza SA, et al. Malignant incidental extracardiac findings on cardiac CT: systematic review and meta-analysis. AJR Am J Roentgenol. 2013;201(3):555-564.
16. Budoff MJ, Gopal A. Incidental findings on cardiac computed tomography. Should we look? J Cardiovasc Comput Tomogr. 2007;1(2):97-105.
17. Earls JP. The pros and cons of searching for extracardiac findings at cardiac CT: studies should be reconstructed in the maximum field of view and adequately reviewed to detect pathologic findings. Radiology. 2011;261(2):342-346.
18. Douglas PS, Cerqueria M, Rubin GD, Chin AS. Extracardiac findings: what is a cardiologist to do? JACC Cardiovasc Imaging. 2008;1(5):682-687.
19. Holly TA, Abbott BG, Al-Mallah M, et al. Single photon-emission computed tomography. J Nucl Cardiol. 2010;17(5):941-973.
20. Dorbala S, Ananthasubramaniam K, Armstrong IS, et al. Single photon emission computed tomography (SPECT) myocardial perfusion imaging guidelines: instrumentation, acquisition, processing, and interpretation. J Nucl Cardiol. 2018;25(5):1784-1846.
21. Tilkemeier PL, Bourque J, Doukky R, Sanghani R, Weinberg RL. ASNC imaging guidelines for nuclear cardiology procedures : Standardized reporting of nuclear cardiology procedures. J Nucl Cardiol. 2017;24(6):2064-2128.
22. Dorbala S, Di Carli MF, Delbeke D, et al. SNMMI/ASNC/SCCT guideline for cardiac SPECT/CT and PET/CT 1.0. J Nucl Med. 2013;54(8):1485-1507.
23. Dilsizian V, Bacharach SL, Beanlands RS, et al. ASNC imaging guidelines/SNMMI procedure standard for positron emission tomography (PET) nuclear cardiology procedures. J Nucl Cardiol. 2016;23(5):1187-1226.
24. Jemal A, Ward EM, Johnson CJ, et al. Annual report to the nation on the status of cancer, 1975-2014, Featuring Survival. J Natl Cancer Inst. 2017;109(9):djx030.
25. US Department of Health and Human Services. The Health Consequences of Smoking: 50 Years of Progress. A Report of the Surgeon General. Atlanta, GA: US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2014. Printed with corrections, January 2014.
26. Odani S, Agaku IT, Graffunder CM, Tynan MA, Armour BS. Tobacco Product Use Among Military Veterans - United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2018;67(1):7-12.
Single-photon emission computed tomography (SPECT) myocardial perfusion imaging (MPI) is a well-established technique for the evaluation of coronary artery disease (CAD).1 To improve image quality, low-resolution computed tomography (CT) is used commonly for anatomical correct and artifact attenuation during SPECT MPI.2 The low resolution, unenhanced CT images are considered low quality and are, therefore, labeled by the manufacturer as nondiagnostic. The CT portion of the MPI in many centers is used only for image fusion and attenuation correction, and these images are not routinely reviewed or reported by cardiologists.
Incidental findings by these low-resolution CT were frequent. However, clinically significant findings, including lung cancer, although relatively infrequent, were serious enough for major clinical management.3-5 Currently, there are no consensus recommendations for reviewing low-resolution CT images or the interpretation of such incidental findings during cardiac MPI.6 Clinically, low-dose CT were used for early detection and screening of lung cancer and were associated with reduced lung-cancer and any cause mortality in National Lung Screening Trial (NLST).7,8 Therefore, low-dose CT is recommended for lung cancer screening of high-risk patients by the US Preventive Service Task Force (USPSTF).9 In the veteran population, current and past smoking history are more common when compared with the general population; therefore, veterans are potentially at increased risk of lung cancer.10 In this study, we did not intend to use low-resolution CT for lung cancer screening or detection but rather to identify and report incidental findings of pulmonary/hilar malignancy detected during cardiac MPI.
Methods
The Siemens’ (Munich, Germany) Symbia Intevo Excel SPECT/CT MPI cameras with dedicated cardiac collimators were used at both the Dwight D. Eisenhower VA Medical Center (VAMC) in Leavenworth, Kansas and Colmery-O'Neil VAMC in Topeka, Kansas. The integrated CT scanner (x-ray tube current 30 to 240 mA; voltage 110 Kv with a 40 kW power generator) has the capability to image up to a 2-slice/rotation, each of 5.0 mm per slice with a scan time of about 30 seconds. The SPECT/CT gamma camera has a low energy (140 KeV), high resolution, parallel hole collimator with IQ SPECT capabilities.
The radiation dose received by the patients were expressed in dose length product (DLP), which reflects the total energy absorbed by the patient and represents integrated dose in terms of the total scan length. Additionally, each patients received 2 injections of Technetium Tc 99m sestamibi (1-day Protocol: 10 mCi rest injection, 30 mCi stress injection: 2-day Protocol for patients weighing > 350 pounds: 30 mCi at rest injection and 30 mCi at stress injection) for myocardial perfusion imaging.
All CT images and cardiac MPI findings were reviewed and reported contemporaneously by 1 of 2 experienced, board-certified radiologists who were blinded to patients’ clinical information except the indication for the cardiac stress testing. When suspicious pulmonary/hilar nodules or masses were detected, these findings and recommendations for further evaluation were conveyed to primary care provider or ordering physician via the electronic health record system.
All CT images were reviewed with cardiac MPI from September 1, 2017 to August 31, 2018. When pulmonary/hilar malignancies were identified, the health records were reviewed. Patients with known history of prior pulmonary malignancy were excluded from the study.
Results
A total of 1,098 patients underwent cardiac MPI during the study period. When the CT imaging and cardiac MPI were reviewed, incidental findings led to the diagnosis of lung cancer in 5 patients and hilar mantle cell lymphoma in 1 patient. Their clinical characteristics, CT findings, and types of malignancies for these 6 patients are summarized in the Table and Figure. Only 0.55% (6 of 1,098) patients were found to have incidental pulmonary/hilar malignancy with the cardiac evaluation low-resolution CT. Four patients with prior, known history of lung cancer were excluded from the study.
For the 6 patients found to have cancer, the average CT radiation dose during the cardiac MPI was 100 mGy-cm (range, 77 -133 mCy-cm). The subsequent chest CT with or without contrast delivered a radiation dose of 726.4 mGy-cm (range, 279.4 - 1,075 mGy-cm).
A total of 79 (7.2%) patients were found to have significant pulmonary nodules that required further evaluation; after CT examination, 32 patients had findings of benign nature and required no further follow-up; the other 47 patients are being followed according to the Fleischner Society 2017 guidelines for pulmonary nodules.11 The follow-up findings on these patients are not within the scope of this report.
Discussion
Although incidental findings on low-resolution CT during cardiac MPI are frequent, clinically significant findings are less common. However, some incidental findings may be of important clinical significance.3-5 A multicenter analysis by Coward and colleagues reported that 2.4% findings on low-resolution CT were significant enough to warrant follow-up tests, but only 0.2% were deemed potentially detrimental to patient outcomes (ie, pathology confirmed malignancies).12 Thus, the authors suggested that routine reporting of incidental findings on low-dose CT images was not beneficial.12,13
Currently, the majority of cardiac MPIs are reviewed and interpreted by nuclear cardiologists, the use of hybrid SPECT/CT for attenuation correction give rise of issue of reviewing and interpreting these CT images during cardiac MPI. Since low-dose, low-resolution CT are considered nondiagnostic, these images are not routinely and readily reviewed by cardiologists who are not trained or skilled in CT interpretations.
Studies of high-resolution cardiac CT (including multidetector CT with contrast) suggest that incidental extracardiac findings should always be reported as there was a 0.7% incidence of previously unknown malignancies, while others have argued against “performing large field reconstructionsfor the explicit purpose of screening as it will lead to additional cost, liability and anxiety without proven benefits.”14-16 A review of incidental findings of cardiac CT by Earls suggested that all cardiac CT should be reconstructed in the maximal field of view available and images should be adequately reviewed to detect pathological findings.17 This led to an interesting discussion by Douglas and colleagues regarding the role of cardiologists and radiologists in this issue.18 Currently there is no uniform or consensus recommendations regarding incidental findings during cardiac CT imaging. Guidances range from no recommendations to optional reporting or mandatory reporting.19-23
Risk Factors for Veterans
Lung cancer is the second most common cancer and the leading cause of cancer-related death in the US.24 Smoking is the most important risk factor for lung cancer and CAD.25 Current or past smoking are more common among the veterans.10 According to a report for the US Centers for Disease Control and Prevention report, about 29.2% US veterans use tobacco products between 2010-2015, which is similar to the rate reported in 1997.26
When low-dose CT was used for lung cancer screening, it was associated with a 20.0% reduction in lung cancer mortality and a 6.7% reduction in any cause mortality.7 Currently, the US Preventive Services Task Force (USPSTF) recommends annual low-dose CT screening for lung cancer in high-risk adults that includes patients aged 55 to 80 years who have a 30-pack-year smoking history and currently smoke or have quit within the past 15 years.8
It is likely that the cardiac patients in this study might have pulmonary malignancy mortality similar to those reported in the NLST. While other studies have shown a low incidence (0.2%) of detection of malignancy by low-resolution CT during cardiac MPI,12,13 in this study we found pulmonary or hilar malignancy in 0.55% of patients.The higher incidence of malignancy in our study might be due in part to differences in the patient population studied (ie, our veterans patients have a higher proportion of current or past smoking history).10
The CT used in this study is part of the cardiac imaging process. Therefore, there was no additional radiation exposure besides that of the cardiac MPI for patients. Despite the limitations of low-resolution CT, which may miss small lesions, this study showed 0.55% incidence of incidental detection of pulmonary/hilar malignancy. This is comparable with 0.65%/year of diagnosing lung cancer using low-dose CT for lung cancer screening in NLST.8
Two of the 5 study patients who were found to have lung cancer, had quit smoking > 15 years previously and thus would not be considered as high-risk for lung cancer screening according to USPSTF guideline. These patients would not have been candidates for annual low-dose CT lung cancer screening. This study suggests that it is appropriate and necessary to review the low-resolution CT images for incidental findings during cardiac MPI.
Limitations
The study was retrospective in nature and limited by its small number of patients. The CT modality used in the study also has limitations, including low resolution, respiratory motion artifacts, and scans that did not include the entire chest area. Therefore, small and apical lesions may have been missed. However, both sets of CT at rest and after stress were reviewed to reduce or minimize the effects of respiratory motion artifacts. The true prevalence or incidence of pulmonary/hilar malignancies may have been higher than reported here. Our study population of veterans may not be representative of the general population with regards to gender (as most of our veteran patient population are of male gender, vs general population), smoking history, or lung cancer risk, thus the results should be interpreted with caution.
Conclusion
Low-resolution CTs used for attenuation correction during cardiac MPI should be routinely reviewed and interpreted by a physician or radiologist skilled in CT interpretation in order to identify incidental findings of pulmonary/hilar malignancy. This would require close collaboration between cardiologists and radiologists in the field to ensure unfragmented and high-quality patient care.
Acknowledgements
We want to thank all the staffs in cardiology and radiology department on both campuses for their dedication for our patients. Special thanks to Laura Knox, Radiation Safety Officer, Nuclear Medicine Supervisor for her technical assistance.
Single-photon emission computed tomography (SPECT) myocardial perfusion imaging (MPI) is a well-established technique for the evaluation of coronary artery disease (CAD).1 To improve image quality, low-resolution computed tomography (CT) is used commonly for anatomical correct and artifact attenuation during SPECT MPI.2 The low resolution, unenhanced CT images are considered low quality and are, therefore, labeled by the manufacturer as nondiagnostic. The CT portion of the MPI in many centers is used only for image fusion and attenuation correction, and these images are not routinely reviewed or reported by cardiologists.
Incidental findings by these low-resolution CT were frequent. However, clinically significant findings, including lung cancer, although relatively infrequent, were serious enough for major clinical management.3-5 Currently, there are no consensus recommendations for reviewing low-resolution CT images or the interpretation of such incidental findings during cardiac MPI.6 Clinically, low-dose CT were used for early detection and screening of lung cancer and were associated with reduced lung-cancer and any cause mortality in National Lung Screening Trial (NLST).7,8 Therefore, low-dose CT is recommended for lung cancer screening of high-risk patients by the US Preventive Service Task Force (USPSTF).9 In the veteran population, current and past smoking history are more common when compared with the general population; therefore, veterans are potentially at increased risk of lung cancer.10 In this study, we did not intend to use low-resolution CT for lung cancer screening or detection but rather to identify and report incidental findings of pulmonary/hilar malignancy detected during cardiac MPI.
Methods
The Siemens’ (Munich, Germany) Symbia Intevo Excel SPECT/CT MPI cameras with dedicated cardiac collimators were used at both the Dwight D. Eisenhower VA Medical Center (VAMC) in Leavenworth, Kansas and Colmery-O'Neil VAMC in Topeka, Kansas. The integrated CT scanner (x-ray tube current 30 to 240 mA; voltage 110 Kv with a 40 kW power generator) has the capability to image up to a 2-slice/rotation, each of 5.0 mm per slice with a scan time of about 30 seconds. The SPECT/CT gamma camera has a low energy (140 KeV), high resolution, parallel hole collimator with IQ SPECT capabilities.
The radiation dose received by the patients were expressed in dose length product (DLP), which reflects the total energy absorbed by the patient and represents integrated dose in terms of the total scan length. Additionally, each patients received 2 injections of Technetium Tc 99m sestamibi (1-day Protocol: 10 mCi rest injection, 30 mCi stress injection: 2-day Protocol for patients weighing > 350 pounds: 30 mCi at rest injection and 30 mCi at stress injection) for myocardial perfusion imaging.
All CT images and cardiac MPI findings were reviewed and reported contemporaneously by 1 of 2 experienced, board-certified radiologists who were blinded to patients’ clinical information except the indication for the cardiac stress testing. When suspicious pulmonary/hilar nodules or masses were detected, these findings and recommendations for further evaluation were conveyed to primary care provider or ordering physician via the electronic health record system.
All CT images were reviewed with cardiac MPI from September 1, 2017 to August 31, 2018. When pulmonary/hilar malignancies were identified, the health records were reviewed. Patients with known history of prior pulmonary malignancy were excluded from the study.
Results
A total of 1,098 patients underwent cardiac MPI during the study period. When the CT imaging and cardiac MPI were reviewed, incidental findings led to the diagnosis of lung cancer in 5 patients and hilar mantle cell lymphoma in 1 patient. Their clinical characteristics, CT findings, and types of malignancies for these 6 patients are summarized in the Table and Figure. Only 0.55% (6 of 1,098) patients were found to have incidental pulmonary/hilar malignancy with the cardiac evaluation low-resolution CT. Four patients with prior, known history of lung cancer were excluded from the study.
For the 6 patients found to have cancer, the average CT radiation dose during the cardiac MPI was 100 mGy-cm (range, 77 -133 mCy-cm). The subsequent chest CT with or without contrast delivered a radiation dose of 726.4 mGy-cm (range, 279.4 - 1,075 mGy-cm).
A total of 79 (7.2%) patients were found to have significant pulmonary nodules that required further evaluation; after CT examination, 32 patients had findings of benign nature and required no further follow-up; the other 47 patients are being followed according to the Fleischner Society 2017 guidelines for pulmonary nodules.11 The follow-up findings on these patients are not within the scope of this report.
Discussion
Although incidental findings on low-resolution CT during cardiac MPI are frequent, clinically significant findings are less common. However, some incidental findings may be of important clinical significance.3-5 A multicenter analysis by Coward and colleagues reported that 2.4% findings on low-resolution CT were significant enough to warrant follow-up tests, but only 0.2% were deemed potentially detrimental to patient outcomes (ie, pathology confirmed malignancies).12 Thus, the authors suggested that routine reporting of incidental findings on low-dose CT images was not beneficial.12,13
Currently, the majority of cardiac MPIs are reviewed and interpreted by nuclear cardiologists, the use of hybrid SPECT/CT for attenuation correction give rise of issue of reviewing and interpreting these CT images during cardiac MPI. Since low-dose, low-resolution CT are considered nondiagnostic, these images are not routinely and readily reviewed by cardiologists who are not trained or skilled in CT interpretations.
Studies of high-resolution cardiac CT (including multidetector CT with contrast) suggest that incidental extracardiac findings should always be reported as there was a 0.7% incidence of previously unknown malignancies, while others have argued against “performing large field reconstructionsfor the explicit purpose of screening as it will lead to additional cost, liability and anxiety without proven benefits.”14-16 A review of incidental findings of cardiac CT by Earls suggested that all cardiac CT should be reconstructed in the maximal field of view available and images should be adequately reviewed to detect pathological findings.17 This led to an interesting discussion by Douglas and colleagues regarding the role of cardiologists and radiologists in this issue.18 Currently there is no uniform or consensus recommendations regarding incidental findings during cardiac CT imaging. Guidances range from no recommendations to optional reporting or mandatory reporting.19-23
Risk Factors for Veterans
Lung cancer is the second most common cancer and the leading cause of cancer-related death in the US.24 Smoking is the most important risk factor for lung cancer and CAD.25 Current or past smoking are more common among the veterans.10 According to a report for the US Centers for Disease Control and Prevention report, about 29.2% US veterans use tobacco products between 2010-2015, which is similar to the rate reported in 1997.26
When low-dose CT was used for lung cancer screening, it was associated with a 20.0% reduction in lung cancer mortality and a 6.7% reduction in any cause mortality.7 Currently, the US Preventive Services Task Force (USPSTF) recommends annual low-dose CT screening for lung cancer in high-risk adults that includes patients aged 55 to 80 years who have a 30-pack-year smoking history and currently smoke or have quit within the past 15 years.8
It is likely that the cardiac patients in this study might have pulmonary malignancy mortality similar to those reported in the NLST. While other studies have shown a low incidence (0.2%) of detection of malignancy by low-resolution CT during cardiac MPI,12,13 in this study we found pulmonary or hilar malignancy in 0.55% of patients.The higher incidence of malignancy in our study might be due in part to differences in the patient population studied (ie, our veterans patients have a higher proportion of current or past smoking history).10
The CT used in this study is part of the cardiac imaging process. Therefore, there was no additional radiation exposure besides that of the cardiac MPI for patients. Despite the limitations of low-resolution CT, which may miss small lesions, this study showed 0.55% incidence of incidental detection of pulmonary/hilar malignancy. This is comparable with 0.65%/year of diagnosing lung cancer using low-dose CT for lung cancer screening in NLST.8
Two of the 5 study patients who were found to have lung cancer, had quit smoking > 15 years previously and thus would not be considered as high-risk for lung cancer screening according to USPSTF guideline. These patients would not have been candidates for annual low-dose CT lung cancer screening. This study suggests that it is appropriate and necessary to review the low-resolution CT images for incidental findings during cardiac MPI.
Limitations
The study was retrospective in nature and limited by its small number of patients. The CT modality used in the study also has limitations, including low resolution, respiratory motion artifacts, and scans that did not include the entire chest area. Therefore, small and apical lesions may have been missed. However, both sets of CT at rest and after stress were reviewed to reduce or minimize the effects of respiratory motion artifacts. The true prevalence or incidence of pulmonary/hilar malignancies may have been higher than reported here. Our study population of veterans may not be representative of the general population with regards to gender (as most of our veteran patient population are of male gender, vs general population), smoking history, or lung cancer risk, thus the results should be interpreted with caution.
Conclusion
Low-resolution CTs used for attenuation correction during cardiac MPI should be routinely reviewed and interpreted by a physician or radiologist skilled in CT interpretation in order to identify incidental findings of pulmonary/hilar malignancy. This would require close collaboration between cardiologists and radiologists in the field to ensure unfragmented and high-quality patient care.
Acknowledgements
We want to thank all the staffs in cardiology and radiology department on both campuses for their dedication for our patients. Special thanks to Laura Knox, Radiation Safety Officer, Nuclear Medicine Supervisor for her technical assistance.
1. Hendel RC, Berman DS, Di Carli MF, et al. ACCF/ASNC/ACR/AHA/ASE/SCCT/SCMR/SNM 2009 appropriate use criteria for cardiac radionuclide imaging: a report of the American College of Cardiology Foundation Appropriate Use Criteria Task Force, the American Society of Nuclear Cardiology, the American College of Radiology, the American Heart Association, the American Society of Echocardiography, the Society of Cardiovascular Computed Tomography, the Society for Cardiovascular Magnetic Resonance, and the Society of Nuclear Medicine. Circulation. 2009;119(22):e561-e587.
2. Hendel RC, Corbett JR, Cullom SJ, DePuey EG, Garcia EV, Bateman TM. The value and practice of attenuation correction for myocardial perfusion SPECT imaging: a joint position statement from the American Society of Nuclear Cardiology and the Society of Nuclear Medicine. J Nucl Cardiol. 2002;9(1):135–143.
3. Coward J, Nightingale J, Hogg P. The clinical dilemma of incidental findings on the low-resolution CT images from SPECT/CT MPI studies. J Nucl Med Technol. 2016;44(3):167-172.
4. Osman MM, Cohade C, Fishman E, Wahl RL. Clinically significant incidental findings on the unenhanced CT portion of PET/CT studies: frequency in 250 patients. J Nucl Med. 2005;46(8):1352-1355.
5. Goetze S, Pannu HK, Wahl RL. Clinically significant abnormal findings on the “nondiagnostic” CT portion of low-amperage-CT attenuation-corrected myocardial perfusion SPECT/CT studies. J Nucl Med. 2006;47(8):1312-1318.
6. American College of Cardiology Foundation Task Force on Expert Consensus Documents, Mark DB, Berman DS, et al. ACCF/ACR/AHA/NASCI/SAIP/SCAI/SCCT 2010 expert consensus document on coronary computed tomographic angiography: a report of the American College of Cardiology Foundation Task Force on Expert Consensus Documents. J Am Coll Cardiol. 2010;55(23):2663-2699.
7. Diederich S, Wormanns D, Semik M, et al. Screening for early lung cancer with low-dose spiral CT: prevalence in 817 asymptomatic smokers. Radiology. 2002;222(3):773-781.
8. National Lung Screening Trial Research Team, Aberle DR, Adams AM, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med. 2011;365(5):395-409.
9. Moyer VA; U.S. Preventive Services Task Force. Screening for lung cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;160(5):330-338.
10. McKinney WP, McIntire DD, Carmody TJ, Joseph A. Comparing the smoking behavior of veterans and nonveterans. Public Health Rep. 1997;112(3):212-218.
11. MacMahon H, Naidich DP, Goo JM, et al. Guidelines for Management of Incidental Pulmonary Nodules Detected on CT Images: From the Fleischner Society 2017. Radiology. 2017;284(1):228-243.
12. Coward J, Lawson R, Kane T, et al. Multi-centre analysis of incidental findings on low-resolution CT attenuation correction images. Br J Radiol. 2014;87(1042):20130701.
13. Coward J, Lawson R, Kane T, et al. Multicentre analysis of incidental findings on low-resolution CT attenuation correction images: an extended study. Br J Radiol. 2015;88(1056):20150555.
14. Haller S, Kaiser C, Buser P, Bongartz G, Bremerich J. Coronary artery imaging with contrast-enhanced MDCT: extracardiac findings. AJR Am J Roentgenol. 2006;187(1):105-110.
15. Flor N, Di Leo G, Squarza SA, et al. Malignant incidental extracardiac findings on cardiac CT: systematic review and meta-analysis. AJR Am J Roentgenol. 2013;201(3):555-564.
16. Budoff MJ, Gopal A. Incidental findings on cardiac computed tomography. Should we look? J Cardiovasc Comput Tomogr. 2007;1(2):97-105.
17. Earls JP. The pros and cons of searching for extracardiac findings at cardiac CT: studies should be reconstructed in the maximum field of view and adequately reviewed to detect pathologic findings. Radiology. 2011;261(2):342-346.
18. Douglas PS, Cerqueria M, Rubin GD, Chin AS. Extracardiac findings: what is a cardiologist to do? JACC Cardiovasc Imaging. 2008;1(5):682-687.
19. Holly TA, Abbott BG, Al-Mallah M, et al. Single photon-emission computed tomography. J Nucl Cardiol. 2010;17(5):941-973.
20. Dorbala S, Ananthasubramaniam K, Armstrong IS, et al. Single photon emission computed tomography (SPECT) myocardial perfusion imaging guidelines: instrumentation, acquisition, processing, and interpretation. J Nucl Cardiol. 2018;25(5):1784-1846.
21. Tilkemeier PL, Bourque J, Doukky R, Sanghani R, Weinberg RL. ASNC imaging guidelines for nuclear cardiology procedures : Standardized reporting of nuclear cardiology procedures. J Nucl Cardiol. 2017;24(6):2064-2128.
22. Dorbala S, Di Carli MF, Delbeke D, et al. SNMMI/ASNC/SCCT guideline for cardiac SPECT/CT and PET/CT 1.0. J Nucl Med. 2013;54(8):1485-1507.
23. Dilsizian V, Bacharach SL, Beanlands RS, et al. ASNC imaging guidelines/SNMMI procedure standard for positron emission tomography (PET) nuclear cardiology procedures. J Nucl Cardiol. 2016;23(5):1187-1226.
24. Jemal A, Ward EM, Johnson CJ, et al. Annual report to the nation on the status of cancer, 1975-2014, Featuring Survival. J Natl Cancer Inst. 2017;109(9):djx030.
25. US Department of Health and Human Services. The Health Consequences of Smoking: 50 Years of Progress. A Report of the Surgeon General. Atlanta, GA: US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2014. Printed with corrections, January 2014.
26. Odani S, Agaku IT, Graffunder CM, Tynan MA, Armour BS. Tobacco Product Use Among Military Veterans - United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2018;67(1):7-12.
1. Hendel RC, Berman DS, Di Carli MF, et al. ACCF/ASNC/ACR/AHA/ASE/SCCT/SCMR/SNM 2009 appropriate use criteria for cardiac radionuclide imaging: a report of the American College of Cardiology Foundation Appropriate Use Criteria Task Force, the American Society of Nuclear Cardiology, the American College of Radiology, the American Heart Association, the American Society of Echocardiography, the Society of Cardiovascular Computed Tomography, the Society for Cardiovascular Magnetic Resonance, and the Society of Nuclear Medicine. Circulation. 2009;119(22):e561-e587.
2. Hendel RC, Corbett JR, Cullom SJ, DePuey EG, Garcia EV, Bateman TM. The value and practice of attenuation correction for myocardial perfusion SPECT imaging: a joint position statement from the American Society of Nuclear Cardiology and the Society of Nuclear Medicine. J Nucl Cardiol. 2002;9(1):135–143.
3. Coward J, Nightingale J, Hogg P. The clinical dilemma of incidental findings on the low-resolution CT images from SPECT/CT MPI studies. J Nucl Med Technol. 2016;44(3):167-172.
4. Osman MM, Cohade C, Fishman E, Wahl RL. Clinically significant incidental findings on the unenhanced CT portion of PET/CT studies: frequency in 250 patients. J Nucl Med. 2005;46(8):1352-1355.
5. Goetze S, Pannu HK, Wahl RL. Clinically significant abnormal findings on the “nondiagnostic” CT portion of low-amperage-CT attenuation-corrected myocardial perfusion SPECT/CT studies. J Nucl Med. 2006;47(8):1312-1318.
6. American College of Cardiology Foundation Task Force on Expert Consensus Documents, Mark DB, Berman DS, et al. ACCF/ACR/AHA/NASCI/SAIP/SCAI/SCCT 2010 expert consensus document on coronary computed tomographic angiography: a report of the American College of Cardiology Foundation Task Force on Expert Consensus Documents. J Am Coll Cardiol. 2010;55(23):2663-2699.
7. Diederich S, Wormanns D, Semik M, et al. Screening for early lung cancer with low-dose spiral CT: prevalence in 817 asymptomatic smokers. Radiology. 2002;222(3):773-781.
8. National Lung Screening Trial Research Team, Aberle DR, Adams AM, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med. 2011;365(5):395-409.
9. Moyer VA; U.S. Preventive Services Task Force. Screening for lung cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;160(5):330-338.
10. McKinney WP, McIntire DD, Carmody TJ, Joseph A. Comparing the smoking behavior of veterans and nonveterans. Public Health Rep. 1997;112(3):212-218.
11. MacMahon H, Naidich DP, Goo JM, et al. Guidelines for Management of Incidental Pulmonary Nodules Detected on CT Images: From the Fleischner Society 2017. Radiology. 2017;284(1):228-243.
12. Coward J, Lawson R, Kane T, et al. Multi-centre analysis of incidental findings on low-resolution CT attenuation correction images. Br J Radiol. 2014;87(1042):20130701.
13. Coward J, Lawson R, Kane T, et al. Multicentre analysis of incidental findings on low-resolution CT attenuation correction images: an extended study. Br J Radiol. 2015;88(1056):20150555.
14. Haller S, Kaiser C, Buser P, Bongartz G, Bremerich J. Coronary artery imaging with contrast-enhanced MDCT: extracardiac findings. AJR Am J Roentgenol. 2006;187(1):105-110.
15. Flor N, Di Leo G, Squarza SA, et al. Malignant incidental extracardiac findings on cardiac CT: systematic review and meta-analysis. AJR Am J Roentgenol. 2013;201(3):555-564.
16. Budoff MJ, Gopal A. Incidental findings on cardiac computed tomography. Should we look? J Cardiovasc Comput Tomogr. 2007;1(2):97-105.
17. Earls JP. The pros and cons of searching for extracardiac findings at cardiac CT: studies should be reconstructed in the maximum field of view and adequately reviewed to detect pathologic findings. Radiology. 2011;261(2):342-346.
18. Douglas PS, Cerqueria M, Rubin GD, Chin AS. Extracardiac findings: what is a cardiologist to do? JACC Cardiovasc Imaging. 2008;1(5):682-687.
19. Holly TA, Abbott BG, Al-Mallah M, et al. Single photon-emission computed tomography. J Nucl Cardiol. 2010;17(5):941-973.
20. Dorbala S, Ananthasubramaniam K, Armstrong IS, et al. Single photon emission computed tomography (SPECT) myocardial perfusion imaging guidelines: instrumentation, acquisition, processing, and interpretation. J Nucl Cardiol. 2018;25(5):1784-1846.
21. Tilkemeier PL, Bourque J, Doukky R, Sanghani R, Weinberg RL. ASNC imaging guidelines for nuclear cardiology procedures : Standardized reporting of nuclear cardiology procedures. J Nucl Cardiol. 2017;24(6):2064-2128.
22. Dorbala S, Di Carli MF, Delbeke D, et al. SNMMI/ASNC/SCCT guideline for cardiac SPECT/CT and PET/CT 1.0. J Nucl Med. 2013;54(8):1485-1507.
23. Dilsizian V, Bacharach SL, Beanlands RS, et al. ASNC imaging guidelines/SNMMI procedure standard for positron emission tomography (PET) nuclear cardiology procedures. J Nucl Cardiol. 2016;23(5):1187-1226.
24. Jemal A, Ward EM, Johnson CJ, et al. Annual report to the nation on the status of cancer, 1975-2014, Featuring Survival. J Natl Cancer Inst. 2017;109(9):djx030.
25. US Department of Health and Human Services. The Health Consequences of Smoking: 50 Years of Progress. A Report of the Surgeon General. Atlanta, GA: US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2014. Printed with corrections, January 2014.
26. Odani S, Agaku IT, Graffunder CM, Tynan MA, Armour BS. Tobacco Product Use Among Military Veterans - United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2018;67(1):7-12.
Distress and Factors Associated with Suicidal Ideation in Veterans Living with Cancer (FULL)
It was estimated that physicians would diagnose a form of invasive cancer > 1.7 million times in 2019. As the second most common cause of death in the US, > 600,000 people were projected to die from cancer in 2019.1 Many individuals with cancer endure distress, which the National Comprehensive Cancer Network (NCCN) defines as a “multifactorial unpleasant experience of a psychological (ie, cognitive, behavioral, emotional), social, spiritual, and/or physical nature that may interfere with the ability to cope effectively with cancer, its physical symptoms, and its treatment.”2,3 Distress in people living with cancer has been attributed to various psychosocial concerns, such as family problems, whichinclude dealing with partners and children; emotional problems, such as depression and anxiety; and physical symptoms, such as pain and fatigue.4-9 Certain factors associated with distress may increase a patient’s risk for suicide.4
Veterans are at particularly high risk for suicide.10 In 2014, veterans accounted for 18% of completed suicides in the US but only were 8.5% of the total population that same year.10 Yet, little research has been done on the relationship between distress and suicide in veterans living with cancer. Aboumrad and colleagues found that 45% of veterans with cancer who completed suicide reported family issues and 41% endorsed chronic pain.11 This study recommended continued efforts to assess and treat distress to lessen risk of suicide in veterans living with cancer; however, to date, only 1 study has specifically evaluated distress and problems endorsed among veterans living with cancer.7
Suicide prevention is of the highest priority to the US Department of Veterans Affairs (VA).12 Consistent with the VA mission to end veteran suicide, the current study aimed to better understand the relationship between distress and suicide within a sample of veterans living with cancer. Findings would additionally be used to tailor clinical assessments and interventions for veterans living with cancer.
This study had 3 primary goals. First, we sought to understand demographic and clinical factors associated with low, moderate, and severe levels of distress in veterans living with cancer who were referred for psychology services. Second, the study investigated the most commonly endorsed problems by veterans living with cancer. Finally, we examined which problems were related to suicidal ideation (SI). It was hypothesized that veterans who reported severe distress would be significantly more likely to endorse SI when compared with veterans who reported mild or moderate distress. Based on existing literature, it was further hypothesized that family, emotional, and physical problems would be significantly associated with SI.7,11
Methods
The current study was conducted at James A. Haley Veterans’ Hospital (JAHVH) in Tampa, Florida. Inclusion criteria included veterans who were diagnosed with cancer, attended an outpatient psychology-oncology evaluation, and completed mental health screening measures provided during their evaluation. Exclusion criteria included veterans who: were seen in response to an inpatient consult, were seen solely for a stem cell transplant evaluation, or did not complete the screening measures.
Measures
A veteran’s demographic (eg, age, sex, ethnicity) and clinical (eg, cancer type, stage of disease, recurrence, cancer treatments received) information was abstracted from their VA medical records. Marital status was assessed during a clinical interview and documented as part of the standardized suicide risk assessment.
The Distress Thermometer (DT) is a subjective measure developed by the NCCN.2 The DT provides a visual representation of a thermometer and asks patients to rate their level of distress over the past week with 0 indicating no distress and 10 indicating extreme distress.
The measurement additionally lists 39 problems nested within 5 domains: practical, family, emotional, spiritual/religious, and physical. Patients may endorse listed items under each problem domain by indicating yes or no. Endorsement of various items are intended to provide more detailed information about sources of distress. Due to the predominantly male and mostly older population included in this study the ability to have children measure was removed from the family problem domain.
SI was assessed in 2 ways. First, by patients’ self-report through item-9 of the Patient Health Questionnaire-9 (PHQ-9).14 Item-9 asks “over the last 2 weeks, how often have you been bothered by thoughts that you would be better off dead or of hurting yourself in some way?” Responses range from 0 (not at all) to 3 (nearly every day).14 Responses > 0 were considered a positive screen for SI.
Procedure
Participants were a sample of veterans who were referred for psychology-oncology services. The NCCN DT and Problems List were administered prior to the start of clinical interviews, which followed a checklist and included standardized assessments of SI and history of a suicide attempt(s). A licensed clinical psychologist or a postdoctoral resident conducted these assessments under the supervision of a licensed psychologist. Data gathered during the clinical interview and from the DT and problems list were documented in health records, which were retrospectively reviewed for relevant information (eg, cancer diagnosis, SI). Therefore, informed consent was waived. This study was approved by the JAHVH Institutional Review Board.
Analysis
Data were analyzed using SPSS Version 25. Data analysis proceeded in 3 steps. First, descriptive statistics included the demographic and clinical factors present in the current sample. Difference between those with and without suicidal ideation were compared using F-statistic for continuous variables and χ2 analyses for categorical variables. Second, to examine relationships between each DT problem domain and SI, χ2 analyses were conducted. Third, DT problem domains that had a significant relationship with SI were entered in a logistic regression. This analysis determined which items, if any, from a DT problem domain predicted SI. In the logistic regression model, history of suicide attempts was entered into the first block, as history of suicide attempts is a well-established risk factor for subsequent suicidal ideation. In the second block, other variables that were significantly related to suicidal ideation in the second step of analyses were included. Before interpreting the results of the logistic regression, model fit was tested using the Hosmer-Lemeshow test. Significance of each individual predictor variable in the model is reported using the Wald χ2 statistic; each Wald statistic is compared with a χ2 distribution with 1 degree of freedom (df). Results of logistic regression models also provide information about the effect of each predictor variable in the regression equation (beta weight), odds a veteran who endorsed each predictor variable in the model would also endorse SI (as indicated by the odds ratio), and an estimate of the amount of variance accounted for by each predictor variable (using Nagelkerke’s pseudo R2, which ranges in value from 0 to 1 with higher values indicating more variance explained). For all analyses, P value of .05 (2-tailed) was used for statistical significance.
Results
The sample consisted of 174 veterans (Table 1). The majority (77.6%) were male with a mean age of nearly 62 years (range, 29-87). Most identified as white (74.1%) with half reporting they were either married or living with a partner.
Prostate cancer (19.0%) was the most common type of cancer among study participants followed by head and neck (18.4%), lymphoma/leukemia (11.5%), lung (11.5%), and breast (10.9%); 31.6% had metastatic disease and 14.9% had recurrent disease. Chemotherapy (42.5%) was the most common treatment modality, followed by surgery (38.5%) and radiation (31.6%). The sample was distributed among the 3 distress DT categories: mild (18.4%), moderate (42.5%), and severe (39.1%).
Problems Endorsed
Treatment decisions (44.3%) and insurance/financial concerns (35.1%) were the most frequently endorsed practical problems (Figure 1). Family health issues (33.9%) and dealing with partner (23.0%) were the most frequently endorsed family problems (Figure 2). Worry (73.0%) and depression (69.5%) were the most frequent emotional problems; of note, all emotional problems were endorsed by at least 50% of veterans (Figure 3). Fatigue (71.3%), sleep (70.7%), and pain (69%), were the most frequently endorsed physical problems (Figure 4). Spiritual/religious problems were endorsed by 15% of veterans.
Suicidal Ideation
Overall, 25.3% of veterans endorsed SI. About 20% of veterans reported a history of ≥ 1 suicide attempts in their lifetime. A significant relationship among distress categories and SI was found (χ2 = 18.36, P < .001). Veterans with severe distress were more likely to endorse SI (42.7%) when compared with veterans with mild (9.4%) or moderate (16.2%) distress.
Similarly, a significant relationship among distress categories and a history of a suicide attempt(s) was found (χ2 = 6.08, P = .048). Veterans with severe distress were more likely to have attempted suicide (29.4%) when compared with veterans with mild (12.5%) or moderate (14.9%) distress.
χ2 analyses were conducted to examine the relationships between DT problem domains and SI. A significant relationship was found between family problems and SI (
Logistic regression analyses determined whether items representative of the family problems domain were predictive of SI. Suicide attempt(s) were entered in the first step of the model to evaluate risk factors for SI over this already established risk factor. The assumptions of logistic regression were met.
The Hosmer-Lemeshow test (χ2 = 3.66, df = 5, P = .56) demonstrated that the model fit was good. The group of predictors used in the model differentiate between people who were experiencing SI and those who were not experiencing SI at the time of evaluation. A history of a suicide attempt(s) predicted SI, as expected (Wald = 6.821, df = 1, P = .01). The odds that a veteran with a history of a suicide attempt(s) would endorse SI at the time of the evaluation was nearly 3 times greater than that of veterans without a history of a suicide attempt(s). Over and above suicide attempts, problems dealing with partner (Wald = 15.142; df = 1, P < .001) was a second significant predictor of current SI. The odds that a veteran who endorsed problems dealing with partner would also endorse SI was > 5 times higher than that of veterans who did not endorse problems dealing with partner. This finding represents a significant risk factor for SI, over and above a history of a suicide attempt(s). The other items from the family problems domains were not significant (P > .05) (Table 3).
Discussion
This study aimed to understand factors associated with low, moderate, and severe levels of distress in veterans living with cancer who were referred for psychology services. As hypothesized, veterans who endorsed severe distress were significantly more likely to endorse SI. They also were more likely to have a history of a suicide attempt(s) when compared with those with mild or moderate distress.
A second aim of this study was to understand the most commonly endorsed problems. Consistent with prior literature, treatment decisions were the most commonly endorsed practical problem; worry and depression were the most common emotional problems; and fatigue, sleep, and pain were the most common physical problems.7
A finding unique to the current study is that family health issues and dealing with partner were specified as the most common family problems. However, a study by Smith and colleagues did not provide information about the rank of most frequently reported problems within this domain.7
The third aim was to understand which problems were related to SI. It was hypothesized that family, emotional, and physical problems would be related to SI. However, results indicated that only family problems (specifically, problems dealing with a partner) were significantly associated with SI among veterans living with cancer.
Contrary to expectations, emotional and physical problems were not found to have a significant relationship with SI. This is likely because veterans endorsed items nested within these problem domains with similar frequency. The lack of significant findings does not suggest that emotional and physical problems are not significant predictors of SI for veterans living with cancer, but that no specific emotional or physical symptom stood out as a predictor of suicidal ideation above the others.
The finding of a significant relationship between family problems (specifically, problems dealing with a partner) and SI in this study is consistent with findings of Aboumrad and colleagues in a study that examined root-cause analyses of completed suicides by veterans living with cancer.11 They found that nearly half the sample endorsed family problems prior to their death, and a small but notable percentage of veterans who completed suicide reported divorce as a stressor prior to their death.
This finding may be explained by Thomas Joiner's interpersonal-psychological theory of suicidal behavior (IPT), which suggests that completed suicide may result from a thwarted sense of belonging, perceived burdensomeness, and acquired capability for suicide.16 Problems dealing with a partner may impact a veteran’s sense of belonging or social connectedness. Problems dealing with a partner also may be attributed to perceived burdens due to limitations imposed by living with cancer and/or undergoing treatment. In both circumstances, the veteran’s social support system may be negatively impacted, and perceived social support is a well-established protective factor against suicide.17
Partner distress is a second consideration. It is likely that veterans’ partners experienced their own distress in response to the veteran’s cancer diagnosis and/or treatment. The partner’s cause, severity, and expression of distress may contribute to problems for the couple.
Finally, the latter point of the IPT refers to acquired capability, or the ability to inflict deadly harm to oneself.18 A military sample was found to have more acquired capability for suicide when compared with a college undergraduate sample.19 A history of a suicide attempt(s) and male gender have been found to significantly predict acquired capability to complete suicide.18 Furthermore, because veterans living with cancer often are in pain, fear of pain associated with suicide may be reduced and, therefore, acquired capability increased. This suggests that male veterans living with cancer who are in pain, have a history of a suicide attempt(s), and current problems with their partner may be an extremely vulnerable population at-risk for suicide. Results from the current study emphasize the importance of veterans having access to mental health and crisis resources for problems dealing with their partner. Partner problems may foreshadow a potentially lethal type of distress.
Strengths
This study’s aims are consistent with the VA’s mission to end veteran suicide and contributes to literature in several important ways.12 First, veterans living with cancer are an understudied population. The current study addresses a gap in existing literature by researching veterans living with cancer and aims to better understand the relationship between cancer-related distress and SI. Second, to the best of the authors’ knowledge, this study is the first to find that problems dealing with a partner significantly increases a veteran’s risk for SI above a history of a suicide attempt(s). Risk assessments now may be more comprehensive through inclusion of this distress factor.
It is recommended that future research use IPT to further investigate the relationship between problems dealing with a partner and SI.16 Future research may do so by including specific measures to assess for the tenants of the theory, including measurements of burdensomeness and belongingness. An expanded knowledge base about what makes problems dealing with a partner a significant suicide risk factor (eg, increased conflict, lack of support, etc.) would better enable clinicians to intervene effectively. Effective intervention may lessen suicidal behaviors or deaths from suicides within the Veteran population.
Limitations
One limitation is the focus on patients who accepted a mental health referral. This study design may limit the generalizability of results to veterans who would not accept mental health treatment. The homogenous sample of veterans is a second limitation. Most participants were male, white, and had a mean age of 62 years. These demographics are representative of the veterans that most typically utilize VA services; however, more research is needed on veterans living with cancer who are female and of diverse racial and ethnic backgrounds. There are likely differences in problems endorsed and factors associated with SI based on age, race, sex, and other socioeconomic factors. A third limitation is the cross-sectional, retrospective nature of this study. Future studies are advised to assess for distress at multiple time points. This is consistent with NCCN Standards of Care for Distress Management.2 Longitudinal data would enable more findings about distress and SI throughout the course of cancer diagnosis and treatment, therefore enhancing clinical implications and informing future research.
Conclusion
This is among the first of studies to investigate distress and factors associated with SI in veterans living with cancer who were referred for psychology services. The prevalence of distress caused by psychosocial factors (including treatment decisions, worry, and depression) highlights the importance of including mental health services as part of comprehensive cancer treatment.
Distress due to treatment decisions may be attributed to a litany of factors such as a veteran’s consideration of adverse effects, effectiveness of treatments, changes to quality of life or functioning, and inclusion of alternative or complimentary treatments. These types of decisions often are reported to be difficult conversations to have with family members or loved ones, who are likely experiencing distress of their own. The role of a mental health provider to assist veterans in exploring their treatment decisions and the implications of such decisions appears important to lessening distress.
Early intervention for emotional symptoms would likely benefit veterans’ management of distress and may lessen suicide risk as depression is known to place veterans at-risk for SI.20 This underscores the importance of timely distress assessment to prevent mild emotional distress from progressing to potentially severe or life-threatening emotional distress. For veterans with a psychiatric history, timely assessment and intervention is essential because psychiatric history is an established suicide risk factor that may be exacerbated by cancer-related distress.12
Furthermore, management of intolerable physical symptoms may lessen risk for suicide.4 Under medical guidance, fatigue may be improved using exercise.21 Behavioral intervention is commonly used as first-line treatment for sleep problems.22 While pain may be lessened through medication or nonpharmacological interventions.23
Considering the numerous ways that distress may present itself (eg, practical, emotional, or physical) and increase risk for SI, it is essential that all veterans living with cancer are assessed for distress and SI, regardless of their presentation. Although veterans may not outwardly express distress, this does not indicate the absence of either distress or risk for suicide. For example, a veteran may be distressed due to financial concerns, transportation issues, and the health of his/her partner or spouse. This veteran may not exhibit visible symptoms of distress, as would be expected when the source of distress is emotional (eg, depression, anxiety). However, this veteran is equally vulnerable to impairing distress and SI as someone who exhibits emotional distress. Distress assessments should be further developed to capture both the visible and less apparent sources of distress, while also serving the imperative function of screening for suicide. Other researchers also have noted the necessity of this development.24 Currently, the NCCN DT and Problems List does not include any assessment of SI or behavior.
Finally, this study identified a potentially critical factor to include in distress assessment: problems dealing with a partner. Problems dealing with a partner have been noted as a source of distress in existing literature, but this is the first study to find problems dealing with a partner to be a predictor of SI in veterans living with cancer.4-6
Because partners often attend appointments with veterans, it is not surprising that problems dealing with their partner are not disclosed more readily. It is recommended that clinicians ask veterans about potential problems with their partner when they are alone. Directly gathering information about such problems while assessing for distress may assist health care workers in providing the most effective, accurate type of intervention in a timely manner, and potentially mitigate risk for suicide.
As recommended by the NCCN and numerous researchers, findings from the current study underscore the importance of accurate, timely assessment of distress.2,4,8 This study makes several important recommendations about how distress assessment may be strengthened and further developed, specifically for the veteran population. This study also expands the current knowledge base of what is known about veterans living with cancer, and has begun to fill a gap in the existing literature. Consistent with the VA mission to end veteran suicide, results suggest that veterans living with cancer should be regularly screened for distress, asked about distress related to their partner, and assessed for SI. Continued efforts to enhance assessment of and response to distress may lessen suicide risk in veterans with cancer.11
Acknowledgements
This study is the result of work supported with resources and the use of facilities at the James A. Haley Veterans’ Hospital.
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69(1):7-34.
2. Riba MB, Donovan, KA, Andersen, B. National Comprehensive Cancer Network clinical practice guidelines in oncology. Distress management (Version 3.2019). J Natl Compr Can Net, 2019;17(10):1229-1249.
3. Zabora J, BrintzenhofeSzoc K, Curbow B, Hooker C, Pianta dosi S. The prevalence of psychological distress by cancer site. Psychooncology. 2001;10(1):19–28.
4. Holland JC, Alici Y. Management of distress in cancer patients. J Support Oncol. 2010;8(1):4-12.
5. Bulli F, Miccinesi G, Maruelli A, Katz M, Paci E. The measure of psychological distress in cancer patients: the use of distress thermometer in the oncological rehabilitation center of Florence. Support Care Cancer. 2009;17(7):771–779.
6. Jacobsen PB, Donovan KA, Trask PC, et al. Screening for psychologic distress in ambulatory cancer patients. Cancer. 2005;103(7):1494-1502.
7. Smith J, Berman S, Dimick J, et al. Distress Screening and Management in an Outpatient VA Cancer Clinic: A Pilot Project Involving Ambulatory Patients Across the Disease Trajectory. Fed Pract. 2017;34(Suppl 1):43S–50S.
8. Carlson LE, Waller A, Groff SL, Bultz BD. Screening for distress, the sixth vital sign, in lung cancer patients: effects on pain, fatigue, and common problems--secondary outcomes of a randomized controlled trial. Psychooncology. 2013;22(8):1880-1888.
9. Cooley ME, Short TH, Moriarty HJ. Symptom prevalence, distress, and change over time in adults receiving treatment for lung cancer. Psychooncology. 2003;12(7):694-708.
10. US Department of Veterans Affairs Office of Suicide Prevention. Suicide among veterans and other Americans 2001-2014. https://www.mentalhealth.va.gov/docs/2016suicidedatareport.pdf. Published August 3, 2016. Accessed April 13, 2020.
11. Aboumrad M, Shiner B, Riblet N, Mills, PD, Watts BV. Factors contributing to cancer-related suicide: a study of root-cause-analysis reports. Psychooncology. 2018;27(9):2237-2244.
12. US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention. National Strategy for Preventing Veteran Suicide 2018–2028. https://www.mentalhealth.va.gov/suicide_prevention/docs/Office-of-Mental-Health-and-Suicide-Prevention-National-Strategy-for-Preventing-Veterans-Suicide.pdf Published 2018. Accessed April 13, 2020.
13. Carlson LE, Waller A, Mitchell AJ. Screening for distress and unmet needs in patients with cancer: review and recommendations. J Clin Oncol. 2012;30(11):1160-1177.
14. Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606–613.
15. Martin A, Rief W, Klaiberg A, Braehler E. Validity of the brief patient health questionnaire mood scale (PHQ-9) in the general population. Gen Hosp Psychiatry. 2006;28(1):71-77.
16. Joiner TE. Why People Die by Suicide. Cambridge, MA: Harvard University Press, 2005.
17. Kleiman EM, Riskind JH, Schaefer KE. Social support and positive events as suicide resiliency factors: examination of synergistic buffering effects. Arch Suicide Res. 2014;18(2):144-155.
18. Van Orden KA, Witte TK, Gordon KH, Bender TW, Joiner TE Jr. Suicidal desire and the capability for suicide: tests of the interpersonal-psychological theory of suicidal behavior among adults. J Consult Clin Psychol. 2008;76(1):72–83.
19. Bryan CJ, Morrow CE, Anestis MD, Joiner TE. A preliminary test of the interpersonal -psychological theory of suicidal behavior in a military sample. Personal Individual Differ. 2010;48(3):347-350.
20. Miller SN, Monahan CJ, Phillips KM, Agliata D, Gironda RJ. Mental health utilization among veterans at risk for suicide: Data from a post-deployment clinic [published online ahead of print, 2018 Oct 8]. Psychol Serv. 2018;10.1037/ser0000311.
21. Galvão DA, Newton RU. Review of exercise intervention studies in cancer patients. J Clin Oncol. 2005;23(4):899-909.
22. Qaseem A, Kansagara D, Forciea MA, Cooke M, Denberg TD; Clinical Guidelines Committee of the American College of Physicians. Management of chronic insomnia disorder in adults: A clinical practice guideline from the American College of Physicians. Ann Intern Med. 2016;165(2):125-133.
23. Ngamkham S, Holden JE, Smith EL. A systematic review: Mindfulness intervention for cancer-related pain. Asia Pac J Oncol Nurs. 2019;6(2):161-169.
24. Granek L, Nakash O, Ben-David M, Shapira S, Ariad S. Oncologists’, nurses’, and social workers’ strategies and barriers to identifying suicide risk in cancer patients. Psychooncology. 2018;27(1):148-154.
It was estimated that physicians would diagnose a form of invasive cancer > 1.7 million times in 2019. As the second most common cause of death in the US, > 600,000 people were projected to die from cancer in 2019.1 Many individuals with cancer endure distress, which the National Comprehensive Cancer Network (NCCN) defines as a “multifactorial unpleasant experience of a psychological (ie, cognitive, behavioral, emotional), social, spiritual, and/or physical nature that may interfere with the ability to cope effectively with cancer, its physical symptoms, and its treatment.”2,3 Distress in people living with cancer has been attributed to various psychosocial concerns, such as family problems, whichinclude dealing with partners and children; emotional problems, such as depression and anxiety; and physical symptoms, such as pain and fatigue.4-9 Certain factors associated with distress may increase a patient’s risk for suicide.4
Veterans are at particularly high risk for suicide.10 In 2014, veterans accounted for 18% of completed suicides in the US but only were 8.5% of the total population that same year.10 Yet, little research has been done on the relationship between distress and suicide in veterans living with cancer. Aboumrad and colleagues found that 45% of veterans with cancer who completed suicide reported family issues and 41% endorsed chronic pain.11 This study recommended continued efforts to assess and treat distress to lessen risk of suicide in veterans living with cancer; however, to date, only 1 study has specifically evaluated distress and problems endorsed among veterans living with cancer.7
Suicide prevention is of the highest priority to the US Department of Veterans Affairs (VA).12 Consistent with the VA mission to end veteran suicide, the current study aimed to better understand the relationship between distress and suicide within a sample of veterans living with cancer. Findings would additionally be used to tailor clinical assessments and interventions for veterans living with cancer.
This study had 3 primary goals. First, we sought to understand demographic and clinical factors associated with low, moderate, and severe levels of distress in veterans living with cancer who were referred for psychology services. Second, the study investigated the most commonly endorsed problems by veterans living with cancer. Finally, we examined which problems were related to suicidal ideation (SI). It was hypothesized that veterans who reported severe distress would be significantly more likely to endorse SI when compared with veterans who reported mild or moderate distress. Based on existing literature, it was further hypothesized that family, emotional, and physical problems would be significantly associated with SI.7,11
Methods
The current study was conducted at James A. Haley Veterans’ Hospital (JAHVH) in Tampa, Florida. Inclusion criteria included veterans who were diagnosed with cancer, attended an outpatient psychology-oncology evaluation, and completed mental health screening measures provided during their evaluation. Exclusion criteria included veterans who: were seen in response to an inpatient consult, were seen solely for a stem cell transplant evaluation, or did not complete the screening measures.
Measures
A veteran’s demographic (eg, age, sex, ethnicity) and clinical (eg, cancer type, stage of disease, recurrence, cancer treatments received) information was abstracted from their VA medical records. Marital status was assessed during a clinical interview and documented as part of the standardized suicide risk assessment.
The Distress Thermometer (DT) is a subjective measure developed by the NCCN.2 The DT provides a visual representation of a thermometer and asks patients to rate their level of distress over the past week with 0 indicating no distress and 10 indicating extreme distress.
The measurement additionally lists 39 problems nested within 5 domains: practical, family, emotional, spiritual/religious, and physical. Patients may endorse listed items under each problem domain by indicating yes or no. Endorsement of various items are intended to provide more detailed information about sources of distress. Due to the predominantly male and mostly older population included in this study the ability to have children measure was removed from the family problem domain.
SI was assessed in 2 ways. First, by patients’ self-report through item-9 of the Patient Health Questionnaire-9 (PHQ-9).14 Item-9 asks “over the last 2 weeks, how often have you been bothered by thoughts that you would be better off dead or of hurting yourself in some way?” Responses range from 0 (not at all) to 3 (nearly every day).14 Responses > 0 were considered a positive screen for SI.
Procedure
Participants were a sample of veterans who were referred for psychology-oncology services. The NCCN DT and Problems List were administered prior to the start of clinical interviews, which followed a checklist and included standardized assessments of SI and history of a suicide attempt(s). A licensed clinical psychologist or a postdoctoral resident conducted these assessments under the supervision of a licensed psychologist. Data gathered during the clinical interview and from the DT and problems list were documented in health records, which were retrospectively reviewed for relevant information (eg, cancer diagnosis, SI). Therefore, informed consent was waived. This study was approved by the JAHVH Institutional Review Board.
Analysis
Data were analyzed using SPSS Version 25. Data analysis proceeded in 3 steps. First, descriptive statistics included the demographic and clinical factors present in the current sample. Difference between those with and without suicidal ideation were compared using F-statistic for continuous variables and χ2 analyses for categorical variables. Second, to examine relationships between each DT problem domain and SI, χ2 analyses were conducted. Third, DT problem domains that had a significant relationship with SI were entered in a logistic regression. This analysis determined which items, if any, from a DT problem domain predicted SI. In the logistic regression model, history of suicide attempts was entered into the first block, as history of suicide attempts is a well-established risk factor for subsequent suicidal ideation. In the second block, other variables that were significantly related to suicidal ideation in the second step of analyses were included. Before interpreting the results of the logistic regression, model fit was tested using the Hosmer-Lemeshow test. Significance of each individual predictor variable in the model is reported using the Wald χ2 statistic; each Wald statistic is compared with a χ2 distribution with 1 degree of freedom (df). Results of logistic regression models also provide information about the effect of each predictor variable in the regression equation (beta weight), odds a veteran who endorsed each predictor variable in the model would also endorse SI (as indicated by the odds ratio), and an estimate of the amount of variance accounted for by each predictor variable (using Nagelkerke’s pseudo R2, which ranges in value from 0 to 1 with higher values indicating more variance explained). For all analyses, P value of .05 (2-tailed) was used for statistical significance.
Results
The sample consisted of 174 veterans (Table 1). The majority (77.6%) were male with a mean age of nearly 62 years (range, 29-87). Most identified as white (74.1%) with half reporting they were either married or living with a partner.
Prostate cancer (19.0%) was the most common type of cancer among study participants followed by head and neck (18.4%), lymphoma/leukemia (11.5%), lung (11.5%), and breast (10.9%); 31.6% had metastatic disease and 14.9% had recurrent disease. Chemotherapy (42.5%) was the most common treatment modality, followed by surgery (38.5%) and radiation (31.6%). The sample was distributed among the 3 distress DT categories: mild (18.4%), moderate (42.5%), and severe (39.1%).
Problems Endorsed
Treatment decisions (44.3%) and insurance/financial concerns (35.1%) were the most frequently endorsed practical problems (Figure 1). Family health issues (33.9%) and dealing with partner (23.0%) were the most frequently endorsed family problems (Figure 2). Worry (73.0%) and depression (69.5%) were the most frequent emotional problems; of note, all emotional problems were endorsed by at least 50% of veterans (Figure 3). Fatigue (71.3%), sleep (70.7%), and pain (69%), were the most frequently endorsed physical problems (Figure 4). Spiritual/religious problems were endorsed by 15% of veterans.
Suicidal Ideation
Overall, 25.3% of veterans endorsed SI. About 20% of veterans reported a history of ≥ 1 suicide attempts in their lifetime. A significant relationship among distress categories and SI was found (χ2 = 18.36, P < .001). Veterans with severe distress were more likely to endorse SI (42.7%) when compared with veterans with mild (9.4%) or moderate (16.2%) distress.
Similarly, a significant relationship among distress categories and a history of a suicide attempt(s) was found (χ2 = 6.08, P = .048). Veterans with severe distress were more likely to have attempted suicide (29.4%) when compared with veterans with mild (12.5%) or moderate (14.9%) distress.
χ2 analyses were conducted to examine the relationships between DT problem domains and SI. A significant relationship was found between family problems and SI (
Logistic regression analyses determined whether items representative of the family problems domain were predictive of SI. Suicide attempt(s) were entered in the first step of the model to evaluate risk factors for SI over this already established risk factor. The assumptions of logistic regression were met.
The Hosmer-Lemeshow test (χ2 = 3.66, df = 5, P = .56) demonstrated that the model fit was good. The group of predictors used in the model differentiate between people who were experiencing SI and those who were not experiencing SI at the time of evaluation. A history of a suicide attempt(s) predicted SI, as expected (Wald = 6.821, df = 1, P = .01). The odds that a veteran with a history of a suicide attempt(s) would endorse SI at the time of the evaluation was nearly 3 times greater than that of veterans without a history of a suicide attempt(s). Over and above suicide attempts, problems dealing with partner (Wald = 15.142; df = 1, P < .001) was a second significant predictor of current SI. The odds that a veteran who endorsed problems dealing with partner would also endorse SI was > 5 times higher than that of veterans who did not endorse problems dealing with partner. This finding represents a significant risk factor for SI, over and above a history of a suicide attempt(s). The other items from the family problems domains were not significant (P > .05) (Table 3).
Discussion
This study aimed to understand factors associated with low, moderate, and severe levels of distress in veterans living with cancer who were referred for psychology services. As hypothesized, veterans who endorsed severe distress were significantly more likely to endorse SI. They also were more likely to have a history of a suicide attempt(s) when compared with those with mild or moderate distress.
A second aim of this study was to understand the most commonly endorsed problems. Consistent with prior literature, treatment decisions were the most commonly endorsed practical problem; worry and depression were the most common emotional problems; and fatigue, sleep, and pain were the most common physical problems.7
A finding unique to the current study is that family health issues and dealing with partner were specified as the most common family problems. However, a study by Smith and colleagues did not provide information about the rank of most frequently reported problems within this domain.7
The third aim was to understand which problems were related to SI. It was hypothesized that family, emotional, and physical problems would be related to SI. However, results indicated that only family problems (specifically, problems dealing with a partner) were significantly associated with SI among veterans living with cancer.
Contrary to expectations, emotional and physical problems were not found to have a significant relationship with SI. This is likely because veterans endorsed items nested within these problem domains with similar frequency. The lack of significant findings does not suggest that emotional and physical problems are not significant predictors of SI for veterans living with cancer, but that no specific emotional or physical symptom stood out as a predictor of suicidal ideation above the others.
The finding of a significant relationship between family problems (specifically, problems dealing with a partner) and SI in this study is consistent with findings of Aboumrad and colleagues in a study that examined root-cause analyses of completed suicides by veterans living with cancer.11 They found that nearly half the sample endorsed family problems prior to their death, and a small but notable percentage of veterans who completed suicide reported divorce as a stressor prior to their death.
This finding may be explained by Thomas Joiner's interpersonal-psychological theory of suicidal behavior (IPT), which suggests that completed suicide may result from a thwarted sense of belonging, perceived burdensomeness, and acquired capability for suicide.16 Problems dealing with a partner may impact a veteran’s sense of belonging or social connectedness. Problems dealing with a partner also may be attributed to perceived burdens due to limitations imposed by living with cancer and/or undergoing treatment. In both circumstances, the veteran’s social support system may be negatively impacted, and perceived social support is a well-established protective factor against suicide.17
Partner distress is a second consideration. It is likely that veterans’ partners experienced their own distress in response to the veteran’s cancer diagnosis and/or treatment. The partner’s cause, severity, and expression of distress may contribute to problems for the couple.
Finally, the latter point of the IPT refers to acquired capability, or the ability to inflict deadly harm to oneself.18 A military sample was found to have more acquired capability for suicide when compared with a college undergraduate sample.19 A history of a suicide attempt(s) and male gender have been found to significantly predict acquired capability to complete suicide.18 Furthermore, because veterans living with cancer often are in pain, fear of pain associated with suicide may be reduced and, therefore, acquired capability increased. This suggests that male veterans living with cancer who are in pain, have a history of a suicide attempt(s), and current problems with their partner may be an extremely vulnerable population at-risk for suicide. Results from the current study emphasize the importance of veterans having access to mental health and crisis resources for problems dealing with their partner. Partner problems may foreshadow a potentially lethal type of distress.
Strengths
This study’s aims are consistent with the VA’s mission to end veteran suicide and contributes to literature in several important ways.12 First, veterans living with cancer are an understudied population. The current study addresses a gap in existing literature by researching veterans living with cancer and aims to better understand the relationship between cancer-related distress and SI. Second, to the best of the authors’ knowledge, this study is the first to find that problems dealing with a partner significantly increases a veteran’s risk for SI above a history of a suicide attempt(s). Risk assessments now may be more comprehensive through inclusion of this distress factor.
It is recommended that future research use IPT to further investigate the relationship between problems dealing with a partner and SI.16 Future research may do so by including specific measures to assess for the tenants of the theory, including measurements of burdensomeness and belongingness. An expanded knowledge base about what makes problems dealing with a partner a significant suicide risk factor (eg, increased conflict, lack of support, etc.) would better enable clinicians to intervene effectively. Effective intervention may lessen suicidal behaviors or deaths from suicides within the Veteran population.
Limitations
One limitation is the focus on patients who accepted a mental health referral. This study design may limit the generalizability of results to veterans who would not accept mental health treatment. The homogenous sample of veterans is a second limitation. Most participants were male, white, and had a mean age of 62 years. These demographics are representative of the veterans that most typically utilize VA services; however, more research is needed on veterans living with cancer who are female and of diverse racial and ethnic backgrounds. There are likely differences in problems endorsed and factors associated with SI based on age, race, sex, and other socioeconomic factors. A third limitation is the cross-sectional, retrospective nature of this study. Future studies are advised to assess for distress at multiple time points. This is consistent with NCCN Standards of Care for Distress Management.2 Longitudinal data would enable more findings about distress and SI throughout the course of cancer diagnosis and treatment, therefore enhancing clinical implications and informing future research.
Conclusion
This is among the first of studies to investigate distress and factors associated with SI in veterans living with cancer who were referred for psychology services. The prevalence of distress caused by psychosocial factors (including treatment decisions, worry, and depression) highlights the importance of including mental health services as part of comprehensive cancer treatment.
Distress due to treatment decisions may be attributed to a litany of factors such as a veteran’s consideration of adverse effects, effectiveness of treatments, changes to quality of life or functioning, and inclusion of alternative or complimentary treatments. These types of decisions often are reported to be difficult conversations to have with family members or loved ones, who are likely experiencing distress of their own. The role of a mental health provider to assist veterans in exploring their treatment decisions and the implications of such decisions appears important to lessening distress.
Early intervention for emotional symptoms would likely benefit veterans’ management of distress and may lessen suicide risk as depression is known to place veterans at-risk for SI.20 This underscores the importance of timely distress assessment to prevent mild emotional distress from progressing to potentially severe or life-threatening emotional distress. For veterans with a psychiatric history, timely assessment and intervention is essential because psychiatric history is an established suicide risk factor that may be exacerbated by cancer-related distress.12
Furthermore, management of intolerable physical symptoms may lessen risk for suicide.4 Under medical guidance, fatigue may be improved using exercise.21 Behavioral intervention is commonly used as first-line treatment for sleep problems.22 While pain may be lessened through medication or nonpharmacological interventions.23
Considering the numerous ways that distress may present itself (eg, practical, emotional, or physical) and increase risk for SI, it is essential that all veterans living with cancer are assessed for distress and SI, regardless of their presentation. Although veterans may not outwardly express distress, this does not indicate the absence of either distress or risk for suicide. For example, a veteran may be distressed due to financial concerns, transportation issues, and the health of his/her partner or spouse. This veteran may not exhibit visible symptoms of distress, as would be expected when the source of distress is emotional (eg, depression, anxiety). However, this veteran is equally vulnerable to impairing distress and SI as someone who exhibits emotional distress. Distress assessments should be further developed to capture both the visible and less apparent sources of distress, while also serving the imperative function of screening for suicide. Other researchers also have noted the necessity of this development.24 Currently, the NCCN DT and Problems List does not include any assessment of SI or behavior.
Finally, this study identified a potentially critical factor to include in distress assessment: problems dealing with a partner. Problems dealing with a partner have been noted as a source of distress in existing literature, but this is the first study to find problems dealing with a partner to be a predictor of SI in veterans living with cancer.4-6
Because partners often attend appointments with veterans, it is not surprising that problems dealing with their partner are not disclosed more readily. It is recommended that clinicians ask veterans about potential problems with their partner when they are alone. Directly gathering information about such problems while assessing for distress may assist health care workers in providing the most effective, accurate type of intervention in a timely manner, and potentially mitigate risk for suicide.
As recommended by the NCCN and numerous researchers, findings from the current study underscore the importance of accurate, timely assessment of distress.2,4,8 This study makes several important recommendations about how distress assessment may be strengthened and further developed, specifically for the veteran population. This study also expands the current knowledge base of what is known about veterans living with cancer, and has begun to fill a gap in the existing literature. Consistent with the VA mission to end veteran suicide, results suggest that veterans living with cancer should be regularly screened for distress, asked about distress related to their partner, and assessed for SI. Continued efforts to enhance assessment of and response to distress may lessen suicide risk in veterans with cancer.11
Acknowledgements
This study is the result of work supported with resources and the use of facilities at the James A. Haley Veterans’ Hospital.
It was estimated that physicians would diagnose a form of invasive cancer > 1.7 million times in 2019. As the second most common cause of death in the US, > 600,000 people were projected to die from cancer in 2019.1 Many individuals with cancer endure distress, which the National Comprehensive Cancer Network (NCCN) defines as a “multifactorial unpleasant experience of a psychological (ie, cognitive, behavioral, emotional), social, spiritual, and/or physical nature that may interfere with the ability to cope effectively with cancer, its physical symptoms, and its treatment.”2,3 Distress in people living with cancer has been attributed to various psychosocial concerns, such as family problems, whichinclude dealing with partners and children; emotional problems, such as depression and anxiety; and physical symptoms, such as pain and fatigue.4-9 Certain factors associated with distress may increase a patient’s risk for suicide.4
Veterans are at particularly high risk for suicide.10 In 2014, veterans accounted for 18% of completed suicides in the US but only were 8.5% of the total population that same year.10 Yet, little research has been done on the relationship between distress and suicide in veterans living with cancer. Aboumrad and colleagues found that 45% of veterans with cancer who completed suicide reported family issues and 41% endorsed chronic pain.11 This study recommended continued efforts to assess and treat distress to lessen risk of suicide in veterans living with cancer; however, to date, only 1 study has specifically evaluated distress and problems endorsed among veterans living with cancer.7
Suicide prevention is of the highest priority to the US Department of Veterans Affairs (VA).12 Consistent with the VA mission to end veteran suicide, the current study aimed to better understand the relationship between distress and suicide within a sample of veterans living with cancer. Findings would additionally be used to tailor clinical assessments and interventions for veterans living with cancer.
This study had 3 primary goals. First, we sought to understand demographic and clinical factors associated with low, moderate, and severe levels of distress in veterans living with cancer who were referred for psychology services. Second, the study investigated the most commonly endorsed problems by veterans living with cancer. Finally, we examined which problems were related to suicidal ideation (SI). It was hypothesized that veterans who reported severe distress would be significantly more likely to endorse SI when compared with veterans who reported mild or moderate distress. Based on existing literature, it was further hypothesized that family, emotional, and physical problems would be significantly associated with SI.7,11
Methods
The current study was conducted at James A. Haley Veterans’ Hospital (JAHVH) in Tampa, Florida. Inclusion criteria included veterans who were diagnosed with cancer, attended an outpatient psychology-oncology evaluation, and completed mental health screening measures provided during their evaluation. Exclusion criteria included veterans who: were seen in response to an inpatient consult, were seen solely for a stem cell transplant evaluation, or did not complete the screening measures.
Measures
A veteran’s demographic (eg, age, sex, ethnicity) and clinical (eg, cancer type, stage of disease, recurrence, cancer treatments received) information was abstracted from their VA medical records. Marital status was assessed during a clinical interview and documented as part of the standardized suicide risk assessment.
The Distress Thermometer (DT) is a subjective measure developed by the NCCN.2 The DT provides a visual representation of a thermometer and asks patients to rate their level of distress over the past week with 0 indicating no distress and 10 indicating extreme distress.
The measurement additionally lists 39 problems nested within 5 domains: practical, family, emotional, spiritual/religious, and physical. Patients may endorse listed items under each problem domain by indicating yes or no. Endorsement of various items are intended to provide more detailed information about sources of distress. Due to the predominantly male and mostly older population included in this study the ability to have children measure was removed from the family problem domain.
SI was assessed in 2 ways. First, by patients’ self-report through item-9 of the Patient Health Questionnaire-9 (PHQ-9).14 Item-9 asks “over the last 2 weeks, how often have you been bothered by thoughts that you would be better off dead or of hurting yourself in some way?” Responses range from 0 (not at all) to 3 (nearly every day).14 Responses > 0 were considered a positive screen for SI.
Procedure
Participants were a sample of veterans who were referred for psychology-oncology services. The NCCN DT and Problems List were administered prior to the start of clinical interviews, which followed a checklist and included standardized assessments of SI and history of a suicide attempt(s). A licensed clinical psychologist or a postdoctoral resident conducted these assessments under the supervision of a licensed psychologist. Data gathered during the clinical interview and from the DT and problems list were documented in health records, which were retrospectively reviewed for relevant information (eg, cancer diagnosis, SI). Therefore, informed consent was waived. This study was approved by the JAHVH Institutional Review Board.
Analysis
Data were analyzed using SPSS Version 25. Data analysis proceeded in 3 steps. First, descriptive statistics included the demographic and clinical factors present in the current sample. Difference between those with and without suicidal ideation were compared using F-statistic for continuous variables and χ2 analyses for categorical variables. Second, to examine relationships between each DT problem domain and SI, χ2 analyses were conducted. Third, DT problem domains that had a significant relationship with SI were entered in a logistic regression. This analysis determined which items, if any, from a DT problem domain predicted SI. In the logistic regression model, history of suicide attempts was entered into the first block, as history of suicide attempts is a well-established risk factor for subsequent suicidal ideation. In the second block, other variables that were significantly related to suicidal ideation in the second step of analyses were included. Before interpreting the results of the logistic regression, model fit was tested using the Hosmer-Lemeshow test. Significance of each individual predictor variable in the model is reported using the Wald χ2 statistic; each Wald statistic is compared with a χ2 distribution with 1 degree of freedom (df). Results of logistic regression models also provide information about the effect of each predictor variable in the regression equation (beta weight), odds a veteran who endorsed each predictor variable in the model would also endorse SI (as indicated by the odds ratio), and an estimate of the amount of variance accounted for by each predictor variable (using Nagelkerke’s pseudo R2, which ranges in value from 0 to 1 with higher values indicating more variance explained). For all analyses, P value of .05 (2-tailed) was used for statistical significance.
Results
The sample consisted of 174 veterans (Table 1). The majority (77.6%) were male with a mean age of nearly 62 years (range, 29-87). Most identified as white (74.1%) with half reporting they were either married or living with a partner.
Prostate cancer (19.0%) was the most common type of cancer among study participants followed by head and neck (18.4%), lymphoma/leukemia (11.5%), lung (11.5%), and breast (10.9%); 31.6% had metastatic disease and 14.9% had recurrent disease. Chemotherapy (42.5%) was the most common treatment modality, followed by surgery (38.5%) and radiation (31.6%). The sample was distributed among the 3 distress DT categories: mild (18.4%), moderate (42.5%), and severe (39.1%).
Problems Endorsed
Treatment decisions (44.3%) and insurance/financial concerns (35.1%) were the most frequently endorsed practical problems (Figure 1). Family health issues (33.9%) and dealing with partner (23.0%) were the most frequently endorsed family problems (Figure 2). Worry (73.0%) and depression (69.5%) were the most frequent emotional problems; of note, all emotional problems were endorsed by at least 50% of veterans (Figure 3). Fatigue (71.3%), sleep (70.7%), and pain (69%), were the most frequently endorsed physical problems (Figure 4). Spiritual/religious problems were endorsed by 15% of veterans.
Suicidal Ideation
Overall, 25.3% of veterans endorsed SI. About 20% of veterans reported a history of ≥ 1 suicide attempts in their lifetime. A significant relationship among distress categories and SI was found (χ2 = 18.36, P < .001). Veterans with severe distress were more likely to endorse SI (42.7%) when compared with veterans with mild (9.4%) or moderate (16.2%) distress.
Similarly, a significant relationship among distress categories and a history of a suicide attempt(s) was found (χ2 = 6.08, P = .048). Veterans with severe distress were more likely to have attempted suicide (29.4%) when compared with veterans with mild (12.5%) or moderate (14.9%) distress.
χ2 analyses were conducted to examine the relationships between DT problem domains and SI. A significant relationship was found between family problems and SI (
Logistic regression analyses determined whether items representative of the family problems domain were predictive of SI. Suicide attempt(s) were entered in the first step of the model to evaluate risk factors for SI over this already established risk factor. The assumptions of logistic regression were met.
The Hosmer-Lemeshow test (χ2 = 3.66, df = 5, P = .56) demonstrated that the model fit was good. The group of predictors used in the model differentiate between people who were experiencing SI and those who were not experiencing SI at the time of evaluation. A history of a suicide attempt(s) predicted SI, as expected (Wald = 6.821, df = 1, P = .01). The odds that a veteran with a history of a suicide attempt(s) would endorse SI at the time of the evaluation was nearly 3 times greater than that of veterans without a history of a suicide attempt(s). Over and above suicide attempts, problems dealing with partner (Wald = 15.142; df = 1, P < .001) was a second significant predictor of current SI. The odds that a veteran who endorsed problems dealing with partner would also endorse SI was > 5 times higher than that of veterans who did not endorse problems dealing with partner. This finding represents a significant risk factor for SI, over and above a history of a suicide attempt(s). The other items from the family problems domains were not significant (P > .05) (Table 3).
Discussion
This study aimed to understand factors associated with low, moderate, and severe levels of distress in veterans living with cancer who were referred for psychology services. As hypothesized, veterans who endorsed severe distress were significantly more likely to endorse SI. They also were more likely to have a history of a suicide attempt(s) when compared with those with mild or moderate distress.
A second aim of this study was to understand the most commonly endorsed problems. Consistent with prior literature, treatment decisions were the most commonly endorsed practical problem; worry and depression were the most common emotional problems; and fatigue, sleep, and pain were the most common physical problems.7
A finding unique to the current study is that family health issues and dealing with partner were specified as the most common family problems. However, a study by Smith and colleagues did not provide information about the rank of most frequently reported problems within this domain.7
The third aim was to understand which problems were related to SI. It was hypothesized that family, emotional, and physical problems would be related to SI. However, results indicated that only family problems (specifically, problems dealing with a partner) were significantly associated with SI among veterans living with cancer.
Contrary to expectations, emotional and physical problems were not found to have a significant relationship with SI. This is likely because veterans endorsed items nested within these problem domains with similar frequency. The lack of significant findings does not suggest that emotional and physical problems are not significant predictors of SI for veterans living with cancer, but that no specific emotional or physical symptom stood out as a predictor of suicidal ideation above the others.
The finding of a significant relationship between family problems (specifically, problems dealing with a partner) and SI in this study is consistent with findings of Aboumrad and colleagues in a study that examined root-cause analyses of completed suicides by veterans living with cancer.11 They found that nearly half the sample endorsed family problems prior to their death, and a small but notable percentage of veterans who completed suicide reported divorce as a stressor prior to their death.
This finding may be explained by Thomas Joiner's interpersonal-psychological theory of suicidal behavior (IPT), which suggests that completed suicide may result from a thwarted sense of belonging, perceived burdensomeness, and acquired capability for suicide.16 Problems dealing with a partner may impact a veteran’s sense of belonging or social connectedness. Problems dealing with a partner also may be attributed to perceived burdens due to limitations imposed by living with cancer and/or undergoing treatment. In both circumstances, the veteran’s social support system may be negatively impacted, and perceived social support is a well-established protective factor against suicide.17
Partner distress is a second consideration. It is likely that veterans’ partners experienced their own distress in response to the veteran’s cancer diagnosis and/or treatment. The partner’s cause, severity, and expression of distress may contribute to problems for the couple.
Finally, the latter point of the IPT refers to acquired capability, or the ability to inflict deadly harm to oneself.18 A military sample was found to have more acquired capability for suicide when compared with a college undergraduate sample.19 A history of a suicide attempt(s) and male gender have been found to significantly predict acquired capability to complete suicide.18 Furthermore, because veterans living with cancer often are in pain, fear of pain associated with suicide may be reduced and, therefore, acquired capability increased. This suggests that male veterans living with cancer who are in pain, have a history of a suicide attempt(s), and current problems with their partner may be an extremely vulnerable population at-risk for suicide. Results from the current study emphasize the importance of veterans having access to mental health and crisis resources for problems dealing with their partner. Partner problems may foreshadow a potentially lethal type of distress.
Strengths
This study’s aims are consistent with the VA’s mission to end veteran suicide and contributes to literature in several important ways.12 First, veterans living with cancer are an understudied population. The current study addresses a gap in existing literature by researching veterans living with cancer and aims to better understand the relationship between cancer-related distress and SI. Second, to the best of the authors’ knowledge, this study is the first to find that problems dealing with a partner significantly increases a veteran’s risk for SI above a history of a suicide attempt(s). Risk assessments now may be more comprehensive through inclusion of this distress factor.
It is recommended that future research use IPT to further investigate the relationship between problems dealing with a partner and SI.16 Future research may do so by including specific measures to assess for the tenants of the theory, including measurements of burdensomeness and belongingness. An expanded knowledge base about what makes problems dealing with a partner a significant suicide risk factor (eg, increased conflict, lack of support, etc.) would better enable clinicians to intervene effectively. Effective intervention may lessen suicidal behaviors or deaths from suicides within the Veteran population.
Limitations
One limitation is the focus on patients who accepted a mental health referral. This study design may limit the generalizability of results to veterans who would not accept mental health treatment. The homogenous sample of veterans is a second limitation. Most participants were male, white, and had a mean age of 62 years. These demographics are representative of the veterans that most typically utilize VA services; however, more research is needed on veterans living with cancer who are female and of diverse racial and ethnic backgrounds. There are likely differences in problems endorsed and factors associated with SI based on age, race, sex, and other socioeconomic factors. A third limitation is the cross-sectional, retrospective nature of this study. Future studies are advised to assess for distress at multiple time points. This is consistent with NCCN Standards of Care for Distress Management.2 Longitudinal data would enable more findings about distress and SI throughout the course of cancer diagnosis and treatment, therefore enhancing clinical implications and informing future research.
Conclusion
This is among the first of studies to investigate distress and factors associated with SI in veterans living with cancer who were referred for psychology services. The prevalence of distress caused by psychosocial factors (including treatment decisions, worry, and depression) highlights the importance of including mental health services as part of comprehensive cancer treatment.
Distress due to treatment decisions may be attributed to a litany of factors such as a veteran’s consideration of adverse effects, effectiveness of treatments, changes to quality of life or functioning, and inclusion of alternative or complimentary treatments. These types of decisions often are reported to be difficult conversations to have with family members or loved ones, who are likely experiencing distress of their own. The role of a mental health provider to assist veterans in exploring their treatment decisions and the implications of such decisions appears important to lessening distress.
Early intervention for emotional symptoms would likely benefit veterans’ management of distress and may lessen suicide risk as depression is known to place veterans at-risk for SI.20 This underscores the importance of timely distress assessment to prevent mild emotional distress from progressing to potentially severe or life-threatening emotional distress. For veterans with a psychiatric history, timely assessment and intervention is essential because psychiatric history is an established suicide risk factor that may be exacerbated by cancer-related distress.12
Furthermore, management of intolerable physical symptoms may lessen risk for suicide.4 Under medical guidance, fatigue may be improved using exercise.21 Behavioral intervention is commonly used as first-line treatment for sleep problems.22 While pain may be lessened through medication or nonpharmacological interventions.23
Considering the numerous ways that distress may present itself (eg, practical, emotional, or physical) and increase risk for SI, it is essential that all veterans living with cancer are assessed for distress and SI, regardless of their presentation. Although veterans may not outwardly express distress, this does not indicate the absence of either distress or risk for suicide. For example, a veteran may be distressed due to financial concerns, transportation issues, and the health of his/her partner or spouse. This veteran may not exhibit visible symptoms of distress, as would be expected when the source of distress is emotional (eg, depression, anxiety). However, this veteran is equally vulnerable to impairing distress and SI as someone who exhibits emotional distress. Distress assessments should be further developed to capture both the visible and less apparent sources of distress, while also serving the imperative function of screening for suicide. Other researchers also have noted the necessity of this development.24 Currently, the NCCN DT and Problems List does not include any assessment of SI or behavior.
Finally, this study identified a potentially critical factor to include in distress assessment: problems dealing with a partner. Problems dealing with a partner have been noted as a source of distress in existing literature, but this is the first study to find problems dealing with a partner to be a predictor of SI in veterans living with cancer.4-6
Because partners often attend appointments with veterans, it is not surprising that problems dealing with their partner are not disclosed more readily. It is recommended that clinicians ask veterans about potential problems with their partner when they are alone. Directly gathering information about such problems while assessing for distress may assist health care workers in providing the most effective, accurate type of intervention in a timely manner, and potentially mitigate risk for suicide.
As recommended by the NCCN and numerous researchers, findings from the current study underscore the importance of accurate, timely assessment of distress.2,4,8 This study makes several important recommendations about how distress assessment may be strengthened and further developed, specifically for the veteran population. This study also expands the current knowledge base of what is known about veterans living with cancer, and has begun to fill a gap in the existing literature. Consistent with the VA mission to end veteran suicide, results suggest that veterans living with cancer should be regularly screened for distress, asked about distress related to their partner, and assessed for SI. Continued efforts to enhance assessment of and response to distress may lessen suicide risk in veterans with cancer.11
Acknowledgements
This study is the result of work supported with resources and the use of facilities at the James A. Haley Veterans’ Hospital.
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69(1):7-34.
2. Riba MB, Donovan, KA, Andersen, B. National Comprehensive Cancer Network clinical practice guidelines in oncology. Distress management (Version 3.2019). J Natl Compr Can Net, 2019;17(10):1229-1249.
3. Zabora J, BrintzenhofeSzoc K, Curbow B, Hooker C, Pianta dosi S. The prevalence of psychological distress by cancer site. Psychooncology. 2001;10(1):19–28.
4. Holland JC, Alici Y. Management of distress in cancer patients. J Support Oncol. 2010;8(1):4-12.
5. Bulli F, Miccinesi G, Maruelli A, Katz M, Paci E. The measure of psychological distress in cancer patients: the use of distress thermometer in the oncological rehabilitation center of Florence. Support Care Cancer. 2009;17(7):771–779.
6. Jacobsen PB, Donovan KA, Trask PC, et al. Screening for psychologic distress in ambulatory cancer patients. Cancer. 2005;103(7):1494-1502.
7. Smith J, Berman S, Dimick J, et al. Distress Screening and Management in an Outpatient VA Cancer Clinic: A Pilot Project Involving Ambulatory Patients Across the Disease Trajectory. Fed Pract. 2017;34(Suppl 1):43S–50S.
8. Carlson LE, Waller A, Groff SL, Bultz BD. Screening for distress, the sixth vital sign, in lung cancer patients: effects on pain, fatigue, and common problems--secondary outcomes of a randomized controlled trial. Psychooncology. 2013;22(8):1880-1888.
9. Cooley ME, Short TH, Moriarty HJ. Symptom prevalence, distress, and change over time in adults receiving treatment for lung cancer. Psychooncology. 2003;12(7):694-708.
10. US Department of Veterans Affairs Office of Suicide Prevention. Suicide among veterans and other Americans 2001-2014. https://www.mentalhealth.va.gov/docs/2016suicidedatareport.pdf. Published August 3, 2016. Accessed April 13, 2020.
11. Aboumrad M, Shiner B, Riblet N, Mills, PD, Watts BV. Factors contributing to cancer-related suicide: a study of root-cause-analysis reports. Psychooncology. 2018;27(9):2237-2244.
12. US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention. National Strategy for Preventing Veteran Suicide 2018–2028. https://www.mentalhealth.va.gov/suicide_prevention/docs/Office-of-Mental-Health-and-Suicide-Prevention-National-Strategy-for-Preventing-Veterans-Suicide.pdf Published 2018. Accessed April 13, 2020.
13. Carlson LE, Waller A, Mitchell AJ. Screening for distress and unmet needs in patients with cancer: review and recommendations. J Clin Oncol. 2012;30(11):1160-1177.
14. Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606–613.
15. Martin A, Rief W, Klaiberg A, Braehler E. Validity of the brief patient health questionnaire mood scale (PHQ-9) in the general population. Gen Hosp Psychiatry. 2006;28(1):71-77.
16. Joiner TE. Why People Die by Suicide. Cambridge, MA: Harvard University Press, 2005.
17. Kleiman EM, Riskind JH, Schaefer KE. Social support and positive events as suicide resiliency factors: examination of synergistic buffering effects. Arch Suicide Res. 2014;18(2):144-155.
18. Van Orden KA, Witte TK, Gordon KH, Bender TW, Joiner TE Jr. Suicidal desire and the capability for suicide: tests of the interpersonal-psychological theory of suicidal behavior among adults. J Consult Clin Psychol. 2008;76(1):72–83.
19. Bryan CJ, Morrow CE, Anestis MD, Joiner TE. A preliminary test of the interpersonal -psychological theory of suicidal behavior in a military sample. Personal Individual Differ. 2010;48(3):347-350.
20. Miller SN, Monahan CJ, Phillips KM, Agliata D, Gironda RJ. Mental health utilization among veterans at risk for suicide: Data from a post-deployment clinic [published online ahead of print, 2018 Oct 8]. Psychol Serv. 2018;10.1037/ser0000311.
21. Galvão DA, Newton RU. Review of exercise intervention studies in cancer patients. J Clin Oncol. 2005;23(4):899-909.
22. Qaseem A, Kansagara D, Forciea MA, Cooke M, Denberg TD; Clinical Guidelines Committee of the American College of Physicians. Management of chronic insomnia disorder in adults: A clinical practice guideline from the American College of Physicians. Ann Intern Med. 2016;165(2):125-133.
23. Ngamkham S, Holden JE, Smith EL. A systematic review: Mindfulness intervention for cancer-related pain. Asia Pac J Oncol Nurs. 2019;6(2):161-169.
24. Granek L, Nakash O, Ben-David M, Shapira S, Ariad S. Oncologists’, nurses’, and social workers’ strategies and barriers to identifying suicide risk in cancer patients. Psychooncology. 2018;27(1):148-154.
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69(1):7-34.
2. Riba MB, Donovan, KA, Andersen, B. National Comprehensive Cancer Network clinical practice guidelines in oncology. Distress management (Version 3.2019). J Natl Compr Can Net, 2019;17(10):1229-1249.
3. Zabora J, BrintzenhofeSzoc K, Curbow B, Hooker C, Pianta dosi S. The prevalence of psychological distress by cancer site. Psychooncology. 2001;10(1):19–28.
4. Holland JC, Alici Y. Management of distress in cancer patients. J Support Oncol. 2010;8(1):4-12.
5. Bulli F, Miccinesi G, Maruelli A, Katz M, Paci E. The measure of psychological distress in cancer patients: the use of distress thermometer in the oncological rehabilitation center of Florence. Support Care Cancer. 2009;17(7):771–779.
6. Jacobsen PB, Donovan KA, Trask PC, et al. Screening for psychologic distress in ambulatory cancer patients. Cancer. 2005;103(7):1494-1502.
7. Smith J, Berman S, Dimick J, et al. Distress Screening and Management in an Outpatient VA Cancer Clinic: A Pilot Project Involving Ambulatory Patients Across the Disease Trajectory. Fed Pract. 2017;34(Suppl 1):43S–50S.
8. Carlson LE, Waller A, Groff SL, Bultz BD. Screening for distress, the sixth vital sign, in lung cancer patients: effects on pain, fatigue, and common problems--secondary outcomes of a randomized controlled trial. Psychooncology. 2013;22(8):1880-1888.
9. Cooley ME, Short TH, Moriarty HJ. Symptom prevalence, distress, and change over time in adults receiving treatment for lung cancer. Psychooncology. 2003;12(7):694-708.
10. US Department of Veterans Affairs Office of Suicide Prevention. Suicide among veterans and other Americans 2001-2014. https://www.mentalhealth.va.gov/docs/2016suicidedatareport.pdf. Published August 3, 2016. Accessed April 13, 2020.
11. Aboumrad M, Shiner B, Riblet N, Mills, PD, Watts BV. Factors contributing to cancer-related suicide: a study of root-cause-analysis reports. Psychooncology. 2018;27(9):2237-2244.
12. US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention. National Strategy for Preventing Veteran Suicide 2018–2028. https://www.mentalhealth.va.gov/suicide_prevention/docs/Office-of-Mental-Health-and-Suicide-Prevention-National-Strategy-for-Preventing-Veterans-Suicide.pdf Published 2018. Accessed April 13, 2020.
13. Carlson LE, Waller A, Mitchell AJ. Screening for distress and unmet needs in patients with cancer: review and recommendations. J Clin Oncol. 2012;30(11):1160-1177.
14. Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606–613.
15. Martin A, Rief W, Klaiberg A, Braehler E. Validity of the brief patient health questionnaire mood scale (PHQ-9) in the general population. Gen Hosp Psychiatry. 2006;28(1):71-77.
16. Joiner TE. Why People Die by Suicide. Cambridge, MA: Harvard University Press, 2005.
17. Kleiman EM, Riskind JH, Schaefer KE. Social support and positive events as suicide resiliency factors: examination of synergistic buffering effects. Arch Suicide Res. 2014;18(2):144-155.
18. Van Orden KA, Witte TK, Gordon KH, Bender TW, Joiner TE Jr. Suicidal desire and the capability for suicide: tests of the interpersonal-psychological theory of suicidal behavior among adults. J Consult Clin Psychol. 2008;76(1):72–83.
19. Bryan CJ, Morrow CE, Anestis MD, Joiner TE. A preliminary test of the interpersonal -psychological theory of suicidal behavior in a military sample. Personal Individual Differ. 2010;48(3):347-350.
20. Miller SN, Monahan CJ, Phillips KM, Agliata D, Gironda RJ. Mental health utilization among veterans at risk for suicide: Data from a post-deployment clinic [published online ahead of print, 2018 Oct 8]. Psychol Serv. 2018;10.1037/ser0000311.
21. Galvão DA, Newton RU. Review of exercise intervention studies in cancer patients. J Clin Oncol. 2005;23(4):899-909.
22. Qaseem A, Kansagara D, Forciea MA, Cooke M, Denberg TD; Clinical Guidelines Committee of the American College of Physicians. Management of chronic insomnia disorder in adults: A clinical practice guideline from the American College of Physicians. Ann Intern Med. 2016;165(2):125-133.
23. Ngamkham S, Holden JE, Smith EL. A systematic review: Mindfulness intervention for cancer-related pain. Asia Pac J Oncol Nurs. 2019;6(2):161-169.
24. Granek L, Nakash O, Ben-David M, Shapira S, Ariad S. Oncologists’, nurses’, and social workers’ strategies and barriers to identifying suicide risk in cancer patients. Psychooncology. 2018;27(1):148-154.
Atrial Fibrillation and Bleeding in Patients With Chronic Lymphocytic Leukemia Treated with Ibrutinib in the Veterans Health Administration (FULL)
Chronic lymphocytic leukemia (CLL) is the most common leukemia diagnosed in developed countries, with an estimated 21,040 new diagnoses of CLL expected in the US in 2020. 1-3 CLL is an indolent cancer characterized by the accumulation of B-lymphocytes in the blood, marrow, and lymphoid tissues. 4 It has a heterogeneous clinical course; the majority of patients are observed or receive delayed treatment following diagnosis, while a minority of patients require immediate treatment. After first-line treatment, some patients experience prolonged remissions while others require retreatment within 1 or 2 years. Fortunately, advances in cancer biology and therapeutics in the last decade have increased the number of treatment options available for patients with CLL.
Until recently, most CLL treatments relied on a chemotherapy or a chemoimmunotherapy backbone; however, the last few years have seen novel therapies introduced, such as small molecule inhibitors to target molecular pathways that promote the normal development, expansion, and survival of B-cells.5 One such therapy is ibrutinib, a targeted Bruton tyrosine kinase inhibitor that received accelerated approval by the US Food and Drug Administration (FDA) in February 2014 for patients with CLL who received at least 1 prior therapy. The FDA later expanded this approval to include use of ibrutinib in patients with CLL with relapsed or refractory disease, with or without chromosome 17p deletion. In 2016, based on data from the RESONATE-17 study, the FDA approved ibrutinib for first-line therapy in patients with CLL.6
Ibrutinib’s efficacy, ease of administration and dosing (all doses are oral and fixed, rather than based on weight or body surface area), and relatively favorable safety profile have resulted in a rapid growth in its adoption.7 Since its adverse event (AE) profile is generally more tolerable than that of a typical chemoimmunotherapy, its use in older patients with CLL and patients with significant comorbidities is particularly appealing.8
However, the results of some clinical trials suggest an association between treatment with ibrutinib and an increased risk of bleeding-related events of any grade (44%) and major bleeding events (4%).7,8 The incidence of major bleeding events was reported to be higher (9%) in one clinical trial and at 5-year follow-up, although this trial did not exclude patients receiving concomitant oral anticoagulation with warfarin.6,9
Heterogeneity in clinical trials’ definitions of major bleeding confounded the ability to calculate bleeding risk in patients treated with ibrutinib in a systematic review and meta-analysis that called for more data.10 Additionally, patients with factors that might increase the risk of major bleeding with ibrutinib treatment were likely underrepresented in clinical trials, given the carefully selected nature of clinical trial subjects. These factors include renal or hepatic disease, gastrointestinal disease, and use of a number of concomitant medications such as antiplatelets or anticoagulant medications. Accounting for use of the latter is particularly important because patients who develop atrial fibrillation (Afib), one of the recognized AEs of treatment with ibrutinib, often are treated with anticoagulant medications in order to decrease the risk of stroke or other thromboembolic complications.
A single-site observational study of patients treated with ibrutinib reported a high utilization rate of antiplatelet medications (70%), anticoagulant medications (17%), or both (13%) with a concomitant major bleeding rate of 18% of patients.11 Prevalence of bleeding events seemed to be highly affected by the presence of concomitant medications: 78% of patients treated with ibrutinib while concurrently receiving both antiplatelet and anticoagulant medications developed a major bleeding event, while none of the patients who were not receiving antiplatelets, anticoagulants, or medications that interact with cytochrome P450 (an enzyme that metabolized chemotherapeutic agents used to treat cancer) experienced a major bleeding event.11
The prevalence of major bleeding events, comorbidities, and utilization of medications that could increase the risk of major bleeding in patients with CLL on ibrutinib in the Veterans Health Administration (VHA) is not known. The VHA is the largest integrated health care system in the US. To address these knowledge gaps, a retrospective observational study was conducted using data on demographics, comorbidities that could affect bleeding, use of anticoagulant and antiplatelet medications, and bleeding events in patients with CLL who were treated in the first year of ibrutinib availability from the VHA.
The first year of ibrutinib availability was chosen for this study since we anticipated that many health care providers would be unfamiliar with ibrutinib during that time given its novelty, and therefore more likely to codispense ibrutinib with medications that could increase the risk of a bleeding event. Since Afib is both an AE associated with ibrutinib treatment and a condition that often is treated with anticoagulants, the prevalence of Afib in this population was also included. For context, the incidence of bleeding and Afib and use of anticoagulant and antiplatelet medications during treatment in a cohort of patients with CLL treated with bendamustine + rituximab (BR) also was reported.
Methods
The VHA maintains the centralized US Department of Veterans Affairs Cancer Registry System (VACRS), with electronic medical record data and other sources captured in its Corporate Data Warehouse (CDW). The VHA CDW is a national repository comprising data from several VHA clinical and administrative systems. The CDW includes patient identifiers; demographics; vital status; lab information; administrative information (such as diagnostic International Statistical Classification of Diseases and Related Health Problems [ICD-9] codes); medication dispensation tables (such as outpatient fill); IV package information; and notes from radiology, pathology, outpatient and inpatient admission, discharge, and daily progress.
Registrars abstract all cancer cases within the VHA system (or diagnosed outside the VHA, if patients subsequently receive treatment in the VHA). It is estimated that VACRS captures 3% of cancer cases in the US.12 Like most registries, VACRS captures data such as diagnosis, age, gender, race, and vital status.
The study received approval from the University of Utah Institutional Review Board and used individual patient-level historical administrative, cancer registry, and electronic health care record data. Patients diagnosed and treated for CLL at the VHA from 2010 to 2014 were identified through the VACRS and CDW; patients with a prior malignancy were excluded. Patients who received ibrutinib or BR based on pharmacy dispensation information were selected. Patients were followed until December 31, 2016 or death; patients with documentation of another cancer or lack of utilization of the VHA hematology or oncology services (defined as absence of any hematology and/or oncology clinic visits for ≥ 18 months) were omitted from the final analysis (Figure).
Previous and concomitant utilization of antiplatelet (aspirin, clopidogrel) or anticoagulant (dalteparin, enoxaparin, fondaparinux, heparin, rivaroxaban, and warfarin) medications was extracted 6 months before and after the first dispensation of ibrutinib or BR using pharmacy dispensation records.
Study Definitions
Prevalence of comorbidities that could increase bleeding risk was determined using administrative ICD-9-CM codes. Liver disease was identified by presence of cirrhosis, hepatitis C virus, or alcoholic liver disease using administrative codes validated by Kramer and colleagues, who reported positive and negative predictive values of 90% and 87% for cirrhosis, 93% and 92% for hepatitis C virus, and 71% and 98% for alcoholic liver disease.13 Similarly, end-stage liver disease was identified using a validated coding algorithm developed by Goldberg and colleagues, with a positive predictive value of 89.3%.14 The presence of controlled or uncontrolled diabetes mellitus (DM) was identified using the procedure described by Guzman and colleagues.15 Quan’s algorithm was used to calculate Charlson Comorbidity Index (CCI) based on ICD-9-CM codes for inpatient and outpatient visits within a 6-month lookback period prior to treatment initiation.16
A major bleeding event was defined as a hospitalization with an ICD-9-CM code suggestive of major bleeding as the primary reason, as defined by Lane and colleagues in their study of major bleeding related to warfarin in a cohort of patients treated within the VHA.17 Incidence rates of major bleeding events were identified during the first 6 months of treatment. Incidence of Afib—defined as an inpatient or outpatient encounter with the 427.31 ICD-9-CM code—also was examined within the first 6 months after starting treatment. The period of 6 months was chosen because bendamustine must be discontinued after 6 months.
Study Analysis
Descriptive statistics were used to examine patient demographics, disease characteristics, and treatment history from initial CLL diagnosis through end of study observation period. Categorical variables were summarized using frequencies and accompanying proportions, while a mean and standard deviation were used to summarize continuous variables. For the means of continuous variables and of categorical data, 95% CIs were used. Proportions and accompanying 95% CIs characterized treatment patterns, including line of therapy, comorbidities, and bleeding events. Treatment duration was described using mean and accompanying 95% CI. Statistical tests were not conducted for comparisons among treatment groups. Patients were censored at the end of follow-up, defined as the earliest of the following scenarios: (1) end of study observation period (December 31, 2016); (2) development of a secondary cancer; or (3) last day of contact given absence of care within the VHA for ≥ 18 months (with care defined as oncology and/or oncology/hematology visit with an associated note). Analysis was performed using R 3.4.0.
Results
Between 2010 and 2014, 2,796 patients were diagnosed and received care for CLL within the VHA. Overall, all 172 patients who were treated with ibrutinib during our inclusion period were selected. These patients were treated between January 1, 2014 and December 31, 2016, following ibrutinib’s approval in early 2014. An additional 291 patients were selected who received BR (Table). Reflecting the predominantly male population of the VHA, 282 (97%) BR patients and 167 (97%) ibrutinib patients were male. The median age at diagnosis was 67 years for BR patients and 69 years for ibrutinib patients. About 76% of patients who received ibrutinib and 82% of patients who received BR were non-Hispanic white; 17% and 14% were African American, respectively.
Less than 10% of patients receiving either ibrutinib or BR had liver disease per criteria used by Kramer and colleagues, or end-stage liver disease using criteria developed by Goldberg and colleagues.12,13 About 5% of patients had a history of previous bleeding in the 6-month period prior to initiating either therapy. Mean CCI (excluding malignancy) score was 1.5 (range, 0-11) for the ibrutinib group, and 2.1 (range, 0-9) for the BR group. About 16% of the ibrutinib group had controlled DM and fewer than 10% had uncontrolled DM, while 4% of patients in the BR group met the criteria for controlled DM and another 4% met the criteria for uncontrolled DM.
There was very low utilization of anticoagulant or antiplatelet medication prior to initiation of ibrutinib (2.9% and 2.3%, respectively) or BR (< 1% each). In the first 6 months after treatment initiation, about 8% of patients in both ibrutinib and BR cohorts received anticoagulant medication while antiplatelet utilization was < 5% in either group.
In the BR group, 8 patients (2.7%) experienced a major bleeding event, while 14 patients (8.1%) in the ibrutinib group experienced a bleeding event (P = .008). While these numbers were too low to perform a formal statistical analysis of the association between clinical covariates and bleeding in either group, there did not seem to be an association between bleeding and liver disease or DM. Of patients who experienced a bleeding event, about 1 in 4 patients had had a prior bleeding event in both the ibrutinib and the BR groups. Interestingly, while none of the patients who experienced a bleeding event while receiving BR were taking concomitant anticoagulant medication, 3 of the 14 patients who experienced a bleeding event in the ibrutinib group showed evidence of anticoagulant utilization. Finally, the incidence of Afib (defined as patients with no evidence of Afib in the 6 months prior to treatment but with evidence of Afib in the 6 months following treatment initiation) was 4% in the BR group, and about 8% in the ibrutinib group (P = .003).
Discussion
To the authors’ knowledge, this study is the first to examine the real-world incidence of bleeding and Afib in veterans who received ibrutinib for CLL in the first year of its availability. The study found minimal use of anticoagulants and/or antiplatelet agents prior to receiving first-line ibrutinib or BR, and very low use of these agents in the first 6 months following the initiation of first-line treatment. This finding suggests a high awareness among VA providers of potential adverse effects (AEs) of ibrutinib and chemotherapy, and a careful selection of patients that lack risk factors for AEs.
In patients treated with first-line ibrutinib when compared with patients treated with first-line BR, moderate increases in bleeding (2.7% vs 8.1%, P = .008) and Afib (10.5% vs 3%, P = .003) also were observed. These results are concordant with previous findings examining the use of ibrutinib in patients with CLL.18-20
Limitations
The results of this study should be interpreted with caution, as some limitations must be considered. The study was conducted in the early days of ibrutinib adoption. Since then, more patients have been treated with ibrutinib and for longer durations. As clinicians gain more familiarity and with ibrutinib, and as additional novel therapeutics emerge, it is possible that the initial awareness about risks for possible AEs may diminish; patients with high comorbidity burdens and concomitant medications would be especially vulnerable in cases of reduced physician vigilance.
Another limitation of this study stems from the potential for dual system use among patients treated in the VHA. Concurrent or alternating use of multiple health care systems (use of VHA and private-sector facilities) may present gaps in the reconstruction of patient histories, resulting in missing data as patients transition between commercial, the Centers for Medicare and Medicaid Services, and VHA care. As a result, the results presented here do not reflect instances where a patient experienced a bleeding event treated outside the VA.
Problems with missing data also may occur due to incomplete extraction from the electronic health record; these issues were addressed by leveraging an understanding of the multiple data marts within the CDW environment to harmonize missing and/or erroneous information through use of other data marts when possible. Lastly, this research represents a population-level study of the VHA, thus all findings are directly relevant to the VHA. The generalizability of the findings outside the VHA would depend on the characteristics of the external population.
Conclusion
Real-world evidence from a nationwide cohort of veteran patients with CLL treated with ibrutinib suggest that, while there is an association of increased bleeding-related events and Afib, the risk is comparable to those reported in previous studies.18-20 These findings suggest that patients in real-world clinical care settings with higher levels of comorbidities may be at a slight increased risk for bleeding events and Afib.
1. Scarfò L, Ferreri AJ, Ghia P. Chronic lymphocytic leukaemia. Crit Rev Oncol Hematol. 2016;104:169-182.
2. Devereux S, Cuthill K. Chronic lymphocytic leukaemia. Medicine (Baltimore). 2017;45(5):292-296.
3. American Cancer Society. Cancer facts & figures 2020. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2020/cancer-facts-and-figures-2020.pdf. Accessed April 24, 2020.
4. Kipps TJ, Stevenson FK, Wu CJ, et al. Chronic lymphocytic leukaemia. Nat Rev Dis Primers. 2017;3:16096.
5. Owen C, Assouline S, Kuruvilla J, Uchida C, Bellingham C, Sehn L. Novel therapies for chronic lymphocytic leukemia: a Canadian perspective. Clin Lymphoma Myeloma Leuk. 2015;15(11):627-634.e5.
6. O’Brien S, Jones JA, Coutre SE, et al. Ibrutinib for patients with relapsed or refractory chronic lymphocytic leukaemia with 17p deletion (RESONATE-17): a phase 2, open-label, multicentre study. Lancet Oncol. 2016;17(10):1409–1418.
7. Burger JA, Tedeschi A, Barr PM, et al; RESONATE-2 Investigators. Ibrutinib as initial therapy for patients with chronic lymphocytic leukemia. N Engl J Med. 2015;373(25):2425-2437.
8. Byrd JC, Furman RR, Coutre SE, et al. Targeting BTK with ibrutinib in relapsed chronic lymphocytic leukemia. N Engl J Med. 2013;369(1):32-42.
9. O’Brien S, Furman R, Coutre S, et al. Single-agent ibrutinib in treatment-naive and relapsed/refractory chronic lymphocytic leukemia: a 5-year experience. Blood. 2018;131(17):1910-1919.
10. Caron F, Leong DP, Hillis C, Fraser G, Siegal D. Current understanding of bleeding with ibrutinib use: a systematic review and meta-analysis. Blood Adv. 2017;1(12):772-778.
11. Kunk PR, Mock J, Devitt ME, Palkimas S, et al. Major bleeding with ibrutinib: more than expected. Blood. 2016;128(22):3229.
12. Zullig LL, Jackson GL, Dorn RA, et al. Cancer incidence among patients of the U.S. Veterans Affairs Health Care System. Mil Med. 2012;177(6):693-701.
13. Kramer JR, Davila JA, Miller ED, Richardson P, Giordano TP, El-Serag HB. The validity of viral hepatitis and chronic liver disease diagnoses in Veterans Affairs administrative databases. Aliment Pharmacol Ther. 2008;27(3):274-282.
14. Goldberg D, Lewis JD, Halpern SD, Weiner M, Lo Re V 3rd. Validation of three coding algorithms to identify patients with end-stage liver disease in an administrative database. Pharmacoepidemiol Drug Saf. 2012;21(7):765-769.
15. Guzman JZ, Iatridis JC, Skovrlj B, et al. Outcomes and complications of diabetes mellitus on patients undergoing degenerative lumbar spine surgery. Spine (Phila Pa 1976). 2014;39(19):1596-1604.
16. Quan H, Sundararajan V, Halfon P, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43(11):1130-1139.
17. Lane MA, Zeringue A, McDonald JR. Serious bleeding events due to warfarin and antibiotic co-prescription in a cohort of veterans. Am J Med. 2014;127(7):657–663.e2.
18. Leong DP, Caron F, Hillis C, et al. The risk of atrial fibrillation with ibrutinib use: a systematic review and meta-analysis. Blood. 2016;128(1):138-140.
19. Lipsky AH, Farooqui MZ, Tian X, et al. Incidence and risk factors of bleeding-related adverse events in patients with chronic lymphocytic leukemia treated with ibrutinib. Haematologica. 2015;100(12):1571-1578.
20. Brown JR, Moslehi J, O’Brien S, et al. Characterization of atrial fibrillation adverse events reported in ibrutinib randomized controlled registration trials. Haematologica. 2017;102(10):1796-1805.
Chronic lymphocytic leukemia (CLL) is the most common leukemia diagnosed in developed countries, with an estimated 21,040 new diagnoses of CLL expected in the US in 2020. 1-3 CLL is an indolent cancer characterized by the accumulation of B-lymphocytes in the blood, marrow, and lymphoid tissues. 4 It has a heterogeneous clinical course; the majority of patients are observed or receive delayed treatment following diagnosis, while a minority of patients require immediate treatment. After first-line treatment, some patients experience prolonged remissions while others require retreatment within 1 or 2 years. Fortunately, advances in cancer biology and therapeutics in the last decade have increased the number of treatment options available for patients with CLL.
Until recently, most CLL treatments relied on a chemotherapy or a chemoimmunotherapy backbone; however, the last few years have seen novel therapies introduced, such as small molecule inhibitors to target molecular pathways that promote the normal development, expansion, and survival of B-cells.5 One such therapy is ibrutinib, a targeted Bruton tyrosine kinase inhibitor that received accelerated approval by the US Food and Drug Administration (FDA) in February 2014 for patients with CLL who received at least 1 prior therapy. The FDA later expanded this approval to include use of ibrutinib in patients with CLL with relapsed or refractory disease, with or without chromosome 17p deletion. In 2016, based on data from the RESONATE-17 study, the FDA approved ibrutinib for first-line therapy in patients with CLL.6
Ibrutinib’s efficacy, ease of administration and dosing (all doses are oral and fixed, rather than based on weight or body surface area), and relatively favorable safety profile have resulted in a rapid growth in its adoption.7 Since its adverse event (AE) profile is generally more tolerable than that of a typical chemoimmunotherapy, its use in older patients with CLL and patients with significant comorbidities is particularly appealing.8
However, the results of some clinical trials suggest an association between treatment with ibrutinib and an increased risk of bleeding-related events of any grade (44%) and major bleeding events (4%).7,8 The incidence of major bleeding events was reported to be higher (9%) in one clinical trial and at 5-year follow-up, although this trial did not exclude patients receiving concomitant oral anticoagulation with warfarin.6,9
Heterogeneity in clinical trials’ definitions of major bleeding confounded the ability to calculate bleeding risk in patients treated with ibrutinib in a systematic review and meta-analysis that called for more data.10 Additionally, patients with factors that might increase the risk of major bleeding with ibrutinib treatment were likely underrepresented in clinical trials, given the carefully selected nature of clinical trial subjects. These factors include renal or hepatic disease, gastrointestinal disease, and use of a number of concomitant medications such as antiplatelets or anticoagulant medications. Accounting for use of the latter is particularly important because patients who develop atrial fibrillation (Afib), one of the recognized AEs of treatment with ibrutinib, often are treated with anticoagulant medications in order to decrease the risk of stroke or other thromboembolic complications.
A single-site observational study of patients treated with ibrutinib reported a high utilization rate of antiplatelet medications (70%), anticoagulant medications (17%), or both (13%) with a concomitant major bleeding rate of 18% of patients.11 Prevalence of bleeding events seemed to be highly affected by the presence of concomitant medications: 78% of patients treated with ibrutinib while concurrently receiving both antiplatelet and anticoagulant medications developed a major bleeding event, while none of the patients who were not receiving antiplatelets, anticoagulants, or medications that interact with cytochrome P450 (an enzyme that metabolized chemotherapeutic agents used to treat cancer) experienced a major bleeding event.11
The prevalence of major bleeding events, comorbidities, and utilization of medications that could increase the risk of major bleeding in patients with CLL on ibrutinib in the Veterans Health Administration (VHA) is not known. The VHA is the largest integrated health care system in the US. To address these knowledge gaps, a retrospective observational study was conducted using data on demographics, comorbidities that could affect bleeding, use of anticoagulant and antiplatelet medications, and bleeding events in patients with CLL who were treated in the first year of ibrutinib availability from the VHA.
The first year of ibrutinib availability was chosen for this study since we anticipated that many health care providers would be unfamiliar with ibrutinib during that time given its novelty, and therefore more likely to codispense ibrutinib with medications that could increase the risk of a bleeding event. Since Afib is both an AE associated with ibrutinib treatment and a condition that often is treated with anticoagulants, the prevalence of Afib in this population was also included. For context, the incidence of bleeding and Afib and use of anticoagulant and antiplatelet medications during treatment in a cohort of patients with CLL treated with bendamustine + rituximab (BR) also was reported.
Methods
The VHA maintains the centralized US Department of Veterans Affairs Cancer Registry System (VACRS), with electronic medical record data and other sources captured in its Corporate Data Warehouse (CDW). The VHA CDW is a national repository comprising data from several VHA clinical and administrative systems. The CDW includes patient identifiers; demographics; vital status; lab information; administrative information (such as diagnostic International Statistical Classification of Diseases and Related Health Problems [ICD-9] codes); medication dispensation tables (such as outpatient fill); IV package information; and notes from radiology, pathology, outpatient and inpatient admission, discharge, and daily progress.
Registrars abstract all cancer cases within the VHA system (or diagnosed outside the VHA, if patients subsequently receive treatment in the VHA). It is estimated that VACRS captures 3% of cancer cases in the US.12 Like most registries, VACRS captures data such as diagnosis, age, gender, race, and vital status.
The study received approval from the University of Utah Institutional Review Board and used individual patient-level historical administrative, cancer registry, and electronic health care record data. Patients diagnosed and treated for CLL at the VHA from 2010 to 2014 were identified through the VACRS and CDW; patients with a prior malignancy were excluded. Patients who received ibrutinib or BR based on pharmacy dispensation information were selected. Patients were followed until December 31, 2016 or death; patients with documentation of another cancer or lack of utilization of the VHA hematology or oncology services (defined as absence of any hematology and/or oncology clinic visits for ≥ 18 months) were omitted from the final analysis (Figure).
Previous and concomitant utilization of antiplatelet (aspirin, clopidogrel) or anticoagulant (dalteparin, enoxaparin, fondaparinux, heparin, rivaroxaban, and warfarin) medications was extracted 6 months before and after the first dispensation of ibrutinib or BR using pharmacy dispensation records.
Study Definitions
Prevalence of comorbidities that could increase bleeding risk was determined using administrative ICD-9-CM codes. Liver disease was identified by presence of cirrhosis, hepatitis C virus, or alcoholic liver disease using administrative codes validated by Kramer and colleagues, who reported positive and negative predictive values of 90% and 87% for cirrhosis, 93% and 92% for hepatitis C virus, and 71% and 98% for alcoholic liver disease.13 Similarly, end-stage liver disease was identified using a validated coding algorithm developed by Goldberg and colleagues, with a positive predictive value of 89.3%.14 The presence of controlled or uncontrolled diabetes mellitus (DM) was identified using the procedure described by Guzman and colleagues.15 Quan’s algorithm was used to calculate Charlson Comorbidity Index (CCI) based on ICD-9-CM codes for inpatient and outpatient visits within a 6-month lookback period prior to treatment initiation.16
A major bleeding event was defined as a hospitalization with an ICD-9-CM code suggestive of major bleeding as the primary reason, as defined by Lane and colleagues in their study of major bleeding related to warfarin in a cohort of patients treated within the VHA.17 Incidence rates of major bleeding events were identified during the first 6 months of treatment. Incidence of Afib—defined as an inpatient or outpatient encounter with the 427.31 ICD-9-CM code—also was examined within the first 6 months after starting treatment. The period of 6 months was chosen because bendamustine must be discontinued after 6 months.
Study Analysis
Descriptive statistics were used to examine patient demographics, disease characteristics, and treatment history from initial CLL diagnosis through end of study observation period. Categorical variables were summarized using frequencies and accompanying proportions, while a mean and standard deviation were used to summarize continuous variables. For the means of continuous variables and of categorical data, 95% CIs were used. Proportions and accompanying 95% CIs characterized treatment patterns, including line of therapy, comorbidities, and bleeding events. Treatment duration was described using mean and accompanying 95% CI. Statistical tests were not conducted for comparisons among treatment groups. Patients were censored at the end of follow-up, defined as the earliest of the following scenarios: (1) end of study observation period (December 31, 2016); (2) development of a secondary cancer; or (3) last day of contact given absence of care within the VHA for ≥ 18 months (with care defined as oncology and/or oncology/hematology visit with an associated note). Analysis was performed using R 3.4.0.
Results
Between 2010 and 2014, 2,796 patients were diagnosed and received care for CLL within the VHA. Overall, all 172 patients who were treated with ibrutinib during our inclusion period were selected. These patients were treated between January 1, 2014 and December 31, 2016, following ibrutinib’s approval in early 2014. An additional 291 patients were selected who received BR (Table). Reflecting the predominantly male population of the VHA, 282 (97%) BR patients and 167 (97%) ibrutinib patients were male. The median age at diagnosis was 67 years for BR patients and 69 years for ibrutinib patients. About 76% of patients who received ibrutinib and 82% of patients who received BR were non-Hispanic white; 17% and 14% were African American, respectively.
Less than 10% of patients receiving either ibrutinib or BR had liver disease per criteria used by Kramer and colleagues, or end-stage liver disease using criteria developed by Goldberg and colleagues.12,13 About 5% of patients had a history of previous bleeding in the 6-month period prior to initiating either therapy. Mean CCI (excluding malignancy) score was 1.5 (range, 0-11) for the ibrutinib group, and 2.1 (range, 0-9) for the BR group. About 16% of the ibrutinib group had controlled DM and fewer than 10% had uncontrolled DM, while 4% of patients in the BR group met the criteria for controlled DM and another 4% met the criteria for uncontrolled DM.
There was very low utilization of anticoagulant or antiplatelet medication prior to initiation of ibrutinib (2.9% and 2.3%, respectively) or BR (< 1% each). In the first 6 months after treatment initiation, about 8% of patients in both ibrutinib and BR cohorts received anticoagulant medication while antiplatelet utilization was < 5% in either group.
In the BR group, 8 patients (2.7%) experienced a major bleeding event, while 14 patients (8.1%) in the ibrutinib group experienced a bleeding event (P = .008). While these numbers were too low to perform a formal statistical analysis of the association between clinical covariates and bleeding in either group, there did not seem to be an association between bleeding and liver disease or DM. Of patients who experienced a bleeding event, about 1 in 4 patients had had a prior bleeding event in both the ibrutinib and the BR groups. Interestingly, while none of the patients who experienced a bleeding event while receiving BR were taking concomitant anticoagulant medication, 3 of the 14 patients who experienced a bleeding event in the ibrutinib group showed evidence of anticoagulant utilization. Finally, the incidence of Afib (defined as patients with no evidence of Afib in the 6 months prior to treatment but with evidence of Afib in the 6 months following treatment initiation) was 4% in the BR group, and about 8% in the ibrutinib group (P = .003).
Discussion
To the authors’ knowledge, this study is the first to examine the real-world incidence of bleeding and Afib in veterans who received ibrutinib for CLL in the first year of its availability. The study found minimal use of anticoagulants and/or antiplatelet agents prior to receiving first-line ibrutinib or BR, and very low use of these agents in the first 6 months following the initiation of first-line treatment. This finding suggests a high awareness among VA providers of potential adverse effects (AEs) of ibrutinib and chemotherapy, and a careful selection of patients that lack risk factors for AEs.
In patients treated with first-line ibrutinib when compared with patients treated with first-line BR, moderate increases in bleeding (2.7% vs 8.1%, P = .008) and Afib (10.5% vs 3%, P = .003) also were observed. These results are concordant with previous findings examining the use of ibrutinib in patients with CLL.18-20
Limitations
The results of this study should be interpreted with caution, as some limitations must be considered. The study was conducted in the early days of ibrutinib adoption. Since then, more patients have been treated with ibrutinib and for longer durations. As clinicians gain more familiarity and with ibrutinib, and as additional novel therapeutics emerge, it is possible that the initial awareness about risks for possible AEs may diminish; patients with high comorbidity burdens and concomitant medications would be especially vulnerable in cases of reduced physician vigilance.
Another limitation of this study stems from the potential for dual system use among patients treated in the VHA. Concurrent or alternating use of multiple health care systems (use of VHA and private-sector facilities) may present gaps in the reconstruction of patient histories, resulting in missing data as patients transition between commercial, the Centers for Medicare and Medicaid Services, and VHA care. As a result, the results presented here do not reflect instances where a patient experienced a bleeding event treated outside the VA.
Problems with missing data also may occur due to incomplete extraction from the electronic health record; these issues were addressed by leveraging an understanding of the multiple data marts within the CDW environment to harmonize missing and/or erroneous information through use of other data marts when possible. Lastly, this research represents a population-level study of the VHA, thus all findings are directly relevant to the VHA. The generalizability of the findings outside the VHA would depend on the characteristics of the external population.
Conclusion
Real-world evidence from a nationwide cohort of veteran patients with CLL treated with ibrutinib suggest that, while there is an association of increased bleeding-related events and Afib, the risk is comparable to those reported in previous studies.18-20 These findings suggest that patients in real-world clinical care settings with higher levels of comorbidities may be at a slight increased risk for bleeding events and Afib.
Chronic lymphocytic leukemia (CLL) is the most common leukemia diagnosed in developed countries, with an estimated 21,040 new diagnoses of CLL expected in the US in 2020. 1-3 CLL is an indolent cancer characterized by the accumulation of B-lymphocytes in the blood, marrow, and lymphoid tissues. 4 It has a heterogeneous clinical course; the majority of patients are observed or receive delayed treatment following diagnosis, while a minority of patients require immediate treatment. After first-line treatment, some patients experience prolonged remissions while others require retreatment within 1 or 2 years. Fortunately, advances in cancer biology and therapeutics in the last decade have increased the number of treatment options available for patients with CLL.
Until recently, most CLL treatments relied on a chemotherapy or a chemoimmunotherapy backbone; however, the last few years have seen novel therapies introduced, such as small molecule inhibitors to target molecular pathways that promote the normal development, expansion, and survival of B-cells.5 One such therapy is ibrutinib, a targeted Bruton tyrosine kinase inhibitor that received accelerated approval by the US Food and Drug Administration (FDA) in February 2014 for patients with CLL who received at least 1 prior therapy. The FDA later expanded this approval to include use of ibrutinib in patients with CLL with relapsed or refractory disease, with or without chromosome 17p deletion. In 2016, based on data from the RESONATE-17 study, the FDA approved ibrutinib for first-line therapy in patients with CLL.6
Ibrutinib’s efficacy, ease of administration and dosing (all doses are oral and fixed, rather than based on weight or body surface area), and relatively favorable safety profile have resulted in a rapid growth in its adoption.7 Since its adverse event (AE) profile is generally more tolerable than that of a typical chemoimmunotherapy, its use in older patients with CLL and patients with significant comorbidities is particularly appealing.8
However, the results of some clinical trials suggest an association between treatment with ibrutinib and an increased risk of bleeding-related events of any grade (44%) and major bleeding events (4%).7,8 The incidence of major bleeding events was reported to be higher (9%) in one clinical trial and at 5-year follow-up, although this trial did not exclude patients receiving concomitant oral anticoagulation with warfarin.6,9
Heterogeneity in clinical trials’ definitions of major bleeding confounded the ability to calculate bleeding risk in patients treated with ibrutinib in a systematic review and meta-analysis that called for more data.10 Additionally, patients with factors that might increase the risk of major bleeding with ibrutinib treatment were likely underrepresented in clinical trials, given the carefully selected nature of clinical trial subjects. These factors include renal or hepatic disease, gastrointestinal disease, and use of a number of concomitant medications such as antiplatelets or anticoagulant medications. Accounting for use of the latter is particularly important because patients who develop atrial fibrillation (Afib), one of the recognized AEs of treatment with ibrutinib, often are treated with anticoagulant medications in order to decrease the risk of stroke or other thromboembolic complications.
A single-site observational study of patients treated with ibrutinib reported a high utilization rate of antiplatelet medications (70%), anticoagulant medications (17%), or both (13%) with a concomitant major bleeding rate of 18% of patients.11 Prevalence of bleeding events seemed to be highly affected by the presence of concomitant medications: 78% of patients treated with ibrutinib while concurrently receiving both antiplatelet and anticoagulant medications developed a major bleeding event, while none of the patients who were not receiving antiplatelets, anticoagulants, or medications that interact with cytochrome P450 (an enzyme that metabolized chemotherapeutic agents used to treat cancer) experienced a major bleeding event.11
The prevalence of major bleeding events, comorbidities, and utilization of medications that could increase the risk of major bleeding in patients with CLL on ibrutinib in the Veterans Health Administration (VHA) is not known. The VHA is the largest integrated health care system in the US. To address these knowledge gaps, a retrospective observational study was conducted using data on demographics, comorbidities that could affect bleeding, use of anticoagulant and antiplatelet medications, and bleeding events in patients with CLL who were treated in the first year of ibrutinib availability from the VHA.
The first year of ibrutinib availability was chosen for this study since we anticipated that many health care providers would be unfamiliar with ibrutinib during that time given its novelty, and therefore more likely to codispense ibrutinib with medications that could increase the risk of a bleeding event. Since Afib is both an AE associated with ibrutinib treatment and a condition that often is treated with anticoagulants, the prevalence of Afib in this population was also included. For context, the incidence of bleeding and Afib and use of anticoagulant and antiplatelet medications during treatment in a cohort of patients with CLL treated with bendamustine + rituximab (BR) also was reported.
Methods
The VHA maintains the centralized US Department of Veterans Affairs Cancer Registry System (VACRS), with electronic medical record data and other sources captured in its Corporate Data Warehouse (CDW). The VHA CDW is a national repository comprising data from several VHA clinical and administrative systems. The CDW includes patient identifiers; demographics; vital status; lab information; administrative information (such as diagnostic International Statistical Classification of Diseases and Related Health Problems [ICD-9] codes); medication dispensation tables (such as outpatient fill); IV package information; and notes from radiology, pathology, outpatient and inpatient admission, discharge, and daily progress.
Registrars abstract all cancer cases within the VHA system (or diagnosed outside the VHA, if patients subsequently receive treatment in the VHA). It is estimated that VACRS captures 3% of cancer cases in the US.12 Like most registries, VACRS captures data such as diagnosis, age, gender, race, and vital status.
The study received approval from the University of Utah Institutional Review Board and used individual patient-level historical administrative, cancer registry, and electronic health care record data. Patients diagnosed and treated for CLL at the VHA from 2010 to 2014 were identified through the VACRS and CDW; patients with a prior malignancy were excluded. Patients who received ibrutinib or BR based on pharmacy dispensation information were selected. Patients were followed until December 31, 2016 or death; patients with documentation of another cancer or lack of utilization of the VHA hematology or oncology services (defined as absence of any hematology and/or oncology clinic visits for ≥ 18 months) were omitted from the final analysis (Figure).
Previous and concomitant utilization of antiplatelet (aspirin, clopidogrel) or anticoagulant (dalteparin, enoxaparin, fondaparinux, heparin, rivaroxaban, and warfarin) medications was extracted 6 months before and after the first dispensation of ibrutinib or BR using pharmacy dispensation records.
Study Definitions
Prevalence of comorbidities that could increase bleeding risk was determined using administrative ICD-9-CM codes. Liver disease was identified by presence of cirrhosis, hepatitis C virus, or alcoholic liver disease using administrative codes validated by Kramer and colleagues, who reported positive and negative predictive values of 90% and 87% for cirrhosis, 93% and 92% for hepatitis C virus, and 71% and 98% for alcoholic liver disease.13 Similarly, end-stage liver disease was identified using a validated coding algorithm developed by Goldberg and colleagues, with a positive predictive value of 89.3%.14 The presence of controlled or uncontrolled diabetes mellitus (DM) was identified using the procedure described by Guzman and colleagues.15 Quan’s algorithm was used to calculate Charlson Comorbidity Index (CCI) based on ICD-9-CM codes for inpatient and outpatient visits within a 6-month lookback period prior to treatment initiation.16
A major bleeding event was defined as a hospitalization with an ICD-9-CM code suggestive of major bleeding as the primary reason, as defined by Lane and colleagues in their study of major bleeding related to warfarin in a cohort of patients treated within the VHA.17 Incidence rates of major bleeding events were identified during the first 6 months of treatment. Incidence of Afib—defined as an inpatient or outpatient encounter with the 427.31 ICD-9-CM code—also was examined within the first 6 months after starting treatment. The period of 6 months was chosen because bendamustine must be discontinued after 6 months.
Study Analysis
Descriptive statistics were used to examine patient demographics, disease characteristics, and treatment history from initial CLL diagnosis through end of study observation period. Categorical variables were summarized using frequencies and accompanying proportions, while a mean and standard deviation were used to summarize continuous variables. For the means of continuous variables and of categorical data, 95% CIs were used. Proportions and accompanying 95% CIs characterized treatment patterns, including line of therapy, comorbidities, and bleeding events. Treatment duration was described using mean and accompanying 95% CI. Statistical tests were not conducted for comparisons among treatment groups. Patients were censored at the end of follow-up, defined as the earliest of the following scenarios: (1) end of study observation period (December 31, 2016); (2) development of a secondary cancer; or (3) last day of contact given absence of care within the VHA for ≥ 18 months (with care defined as oncology and/or oncology/hematology visit with an associated note). Analysis was performed using R 3.4.0.
Results
Between 2010 and 2014, 2,796 patients were diagnosed and received care for CLL within the VHA. Overall, all 172 patients who were treated with ibrutinib during our inclusion period were selected. These patients were treated between January 1, 2014 and December 31, 2016, following ibrutinib’s approval in early 2014. An additional 291 patients were selected who received BR (Table). Reflecting the predominantly male population of the VHA, 282 (97%) BR patients and 167 (97%) ibrutinib patients were male. The median age at diagnosis was 67 years for BR patients and 69 years for ibrutinib patients. About 76% of patients who received ibrutinib and 82% of patients who received BR were non-Hispanic white; 17% and 14% were African American, respectively.
Less than 10% of patients receiving either ibrutinib or BR had liver disease per criteria used by Kramer and colleagues, or end-stage liver disease using criteria developed by Goldberg and colleagues.12,13 About 5% of patients had a history of previous bleeding in the 6-month period prior to initiating either therapy. Mean CCI (excluding malignancy) score was 1.5 (range, 0-11) for the ibrutinib group, and 2.1 (range, 0-9) for the BR group. About 16% of the ibrutinib group had controlled DM and fewer than 10% had uncontrolled DM, while 4% of patients in the BR group met the criteria for controlled DM and another 4% met the criteria for uncontrolled DM.
There was very low utilization of anticoagulant or antiplatelet medication prior to initiation of ibrutinib (2.9% and 2.3%, respectively) or BR (< 1% each). In the first 6 months after treatment initiation, about 8% of patients in both ibrutinib and BR cohorts received anticoagulant medication while antiplatelet utilization was < 5% in either group.
In the BR group, 8 patients (2.7%) experienced a major bleeding event, while 14 patients (8.1%) in the ibrutinib group experienced a bleeding event (P = .008). While these numbers were too low to perform a formal statistical analysis of the association between clinical covariates and bleeding in either group, there did not seem to be an association between bleeding and liver disease or DM. Of patients who experienced a bleeding event, about 1 in 4 patients had had a prior bleeding event in both the ibrutinib and the BR groups. Interestingly, while none of the patients who experienced a bleeding event while receiving BR were taking concomitant anticoagulant medication, 3 of the 14 patients who experienced a bleeding event in the ibrutinib group showed evidence of anticoagulant utilization. Finally, the incidence of Afib (defined as patients with no evidence of Afib in the 6 months prior to treatment but with evidence of Afib in the 6 months following treatment initiation) was 4% in the BR group, and about 8% in the ibrutinib group (P = .003).
Discussion
To the authors’ knowledge, this study is the first to examine the real-world incidence of bleeding and Afib in veterans who received ibrutinib for CLL in the first year of its availability. The study found minimal use of anticoagulants and/or antiplatelet agents prior to receiving first-line ibrutinib or BR, and very low use of these agents in the first 6 months following the initiation of first-line treatment. This finding suggests a high awareness among VA providers of potential adverse effects (AEs) of ibrutinib and chemotherapy, and a careful selection of patients that lack risk factors for AEs.
In patients treated with first-line ibrutinib when compared with patients treated with first-line BR, moderate increases in bleeding (2.7% vs 8.1%, P = .008) and Afib (10.5% vs 3%, P = .003) also were observed. These results are concordant with previous findings examining the use of ibrutinib in patients with CLL.18-20
Limitations
The results of this study should be interpreted with caution, as some limitations must be considered. The study was conducted in the early days of ibrutinib adoption. Since then, more patients have been treated with ibrutinib and for longer durations. As clinicians gain more familiarity and with ibrutinib, and as additional novel therapeutics emerge, it is possible that the initial awareness about risks for possible AEs may diminish; patients with high comorbidity burdens and concomitant medications would be especially vulnerable in cases of reduced physician vigilance.
Another limitation of this study stems from the potential for dual system use among patients treated in the VHA. Concurrent or alternating use of multiple health care systems (use of VHA and private-sector facilities) may present gaps in the reconstruction of patient histories, resulting in missing data as patients transition between commercial, the Centers for Medicare and Medicaid Services, and VHA care. As a result, the results presented here do not reflect instances where a patient experienced a bleeding event treated outside the VA.
Problems with missing data also may occur due to incomplete extraction from the electronic health record; these issues were addressed by leveraging an understanding of the multiple data marts within the CDW environment to harmonize missing and/or erroneous information through use of other data marts when possible. Lastly, this research represents a population-level study of the VHA, thus all findings are directly relevant to the VHA. The generalizability of the findings outside the VHA would depend on the characteristics of the external population.
Conclusion
Real-world evidence from a nationwide cohort of veteran patients with CLL treated with ibrutinib suggest that, while there is an association of increased bleeding-related events and Afib, the risk is comparable to those reported in previous studies.18-20 These findings suggest that patients in real-world clinical care settings with higher levels of comorbidities may be at a slight increased risk for bleeding events and Afib.
1. Scarfò L, Ferreri AJ, Ghia P. Chronic lymphocytic leukaemia. Crit Rev Oncol Hematol. 2016;104:169-182.
2. Devereux S, Cuthill K. Chronic lymphocytic leukaemia. Medicine (Baltimore). 2017;45(5):292-296.
3. American Cancer Society. Cancer facts & figures 2020. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2020/cancer-facts-and-figures-2020.pdf. Accessed April 24, 2020.
4. Kipps TJ, Stevenson FK, Wu CJ, et al. Chronic lymphocytic leukaemia. Nat Rev Dis Primers. 2017;3:16096.
5. Owen C, Assouline S, Kuruvilla J, Uchida C, Bellingham C, Sehn L. Novel therapies for chronic lymphocytic leukemia: a Canadian perspective. Clin Lymphoma Myeloma Leuk. 2015;15(11):627-634.e5.
6. O’Brien S, Jones JA, Coutre SE, et al. Ibrutinib for patients with relapsed or refractory chronic lymphocytic leukaemia with 17p deletion (RESONATE-17): a phase 2, open-label, multicentre study. Lancet Oncol. 2016;17(10):1409–1418.
7. Burger JA, Tedeschi A, Barr PM, et al; RESONATE-2 Investigators. Ibrutinib as initial therapy for patients with chronic lymphocytic leukemia. N Engl J Med. 2015;373(25):2425-2437.
8. Byrd JC, Furman RR, Coutre SE, et al. Targeting BTK with ibrutinib in relapsed chronic lymphocytic leukemia. N Engl J Med. 2013;369(1):32-42.
9. O’Brien S, Furman R, Coutre S, et al. Single-agent ibrutinib in treatment-naive and relapsed/refractory chronic lymphocytic leukemia: a 5-year experience. Blood. 2018;131(17):1910-1919.
10. Caron F, Leong DP, Hillis C, Fraser G, Siegal D. Current understanding of bleeding with ibrutinib use: a systematic review and meta-analysis. Blood Adv. 2017;1(12):772-778.
11. Kunk PR, Mock J, Devitt ME, Palkimas S, et al. Major bleeding with ibrutinib: more than expected. Blood. 2016;128(22):3229.
12. Zullig LL, Jackson GL, Dorn RA, et al. Cancer incidence among patients of the U.S. Veterans Affairs Health Care System. Mil Med. 2012;177(6):693-701.
13. Kramer JR, Davila JA, Miller ED, Richardson P, Giordano TP, El-Serag HB. The validity of viral hepatitis and chronic liver disease diagnoses in Veterans Affairs administrative databases. Aliment Pharmacol Ther. 2008;27(3):274-282.
14. Goldberg D, Lewis JD, Halpern SD, Weiner M, Lo Re V 3rd. Validation of three coding algorithms to identify patients with end-stage liver disease in an administrative database. Pharmacoepidemiol Drug Saf. 2012;21(7):765-769.
15. Guzman JZ, Iatridis JC, Skovrlj B, et al. Outcomes and complications of diabetes mellitus on patients undergoing degenerative lumbar spine surgery. Spine (Phila Pa 1976). 2014;39(19):1596-1604.
16. Quan H, Sundararajan V, Halfon P, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43(11):1130-1139.
17. Lane MA, Zeringue A, McDonald JR. Serious bleeding events due to warfarin and antibiotic co-prescription in a cohort of veterans. Am J Med. 2014;127(7):657–663.e2.
18. Leong DP, Caron F, Hillis C, et al. The risk of atrial fibrillation with ibrutinib use: a systematic review and meta-analysis. Blood. 2016;128(1):138-140.
19. Lipsky AH, Farooqui MZ, Tian X, et al. Incidence and risk factors of bleeding-related adverse events in patients with chronic lymphocytic leukemia treated with ibrutinib. Haematologica. 2015;100(12):1571-1578.
20. Brown JR, Moslehi J, O’Brien S, et al. Characterization of atrial fibrillation adverse events reported in ibrutinib randomized controlled registration trials. Haematologica. 2017;102(10):1796-1805.
1. Scarfò L, Ferreri AJ, Ghia P. Chronic lymphocytic leukaemia. Crit Rev Oncol Hematol. 2016;104:169-182.
2. Devereux S, Cuthill K. Chronic lymphocytic leukaemia. Medicine (Baltimore). 2017;45(5):292-296.
3. American Cancer Society. Cancer facts & figures 2020. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2020/cancer-facts-and-figures-2020.pdf. Accessed April 24, 2020.
4. Kipps TJ, Stevenson FK, Wu CJ, et al. Chronic lymphocytic leukaemia. Nat Rev Dis Primers. 2017;3:16096.
5. Owen C, Assouline S, Kuruvilla J, Uchida C, Bellingham C, Sehn L. Novel therapies for chronic lymphocytic leukemia: a Canadian perspective. Clin Lymphoma Myeloma Leuk. 2015;15(11):627-634.e5.
6. O’Brien S, Jones JA, Coutre SE, et al. Ibrutinib for patients with relapsed or refractory chronic lymphocytic leukaemia with 17p deletion (RESONATE-17): a phase 2, open-label, multicentre study. Lancet Oncol. 2016;17(10):1409–1418.
7. Burger JA, Tedeschi A, Barr PM, et al; RESONATE-2 Investigators. Ibrutinib as initial therapy for patients with chronic lymphocytic leukemia. N Engl J Med. 2015;373(25):2425-2437.
8. Byrd JC, Furman RR, Coutre SE, et al. Targeting BTK with ibrutinib in relapsed chronic lymphocytic leukemia. N Engl J Med. 2013;369(1):32-42.
9. O’Brien S, Furman R, Coutre S, et al. Single-agent ibrutinib in treatment-naive and relapsed/refractory chronic lymphocytic leukemia: a 5-year experience. Blood. 2018;131(17):1910-1919.
10. Caron F, Leong DP, Hillis C, Fraser G, Siegal D. Current understanding of bleeding with ibrutinib use: a systematic review and meta-analysis. Blood Adv. 2017;1(12):772-778.
11. Kunk PR, Mock J, Devitt ME, Palkimas S, et al. Major bleeding with ibrutinib: more than expected. Blood. 2016;128(22):3229.
12. Zullig LL, Jackson GL, Dorn RA, et al. Cancer incidence among patients of the U.S. Veterans Affairs Health Care System. Mil Med. 2012;177(6):693-701.
13. Kramer JR, Davila JA, Miller ED, Richardson P, Giordano TP, El-Serag HB. The validity of viral hepatitis and chronic liver disease diagnoses in Veterans Affairs administrative databases. Aliment Pharmacol Ther. 2008;27(3):274-282.
14. Goldberg D, Lewis JD, Halpern SD, Weiner M, Lo Re V 3rd. Validation of three coding algorithms to identify patients with end-stage liver disease in an administrative database. Pharmacoepidemiol Drug Saf. 2012;21(7):765-769.
15. Guzman JZ, Iatridis JC, Skovrlj B, et al. Outcomes and complications of diabetes mellitus on patients undergoing degenerative lumbar spine surgery. Spine (Phila Pa 1976). 2014;39(19):1596-1604.
16. Quan H, Sundararajan V, Halfon P, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43(11):1130-1139.
17. Lane MA, Zeringue A, McDonald JR. Serious bleeding events due to warfarin and antibiotic co-prescription in a cohort of veterans. Am J Med. 2014;127(7):657–663.e2.
18. Leong DP, Caron F, Hillis C, et al. The risk of atrial fibrillation with ibrutinib use: a systematic review and meta-analysis. Blood. 2016;128(1):138-140.
19. Lipsky AH, Farooqui MZ, Tian X, et al. Incidence and risk factors of bleeding-related adverse events in patients with chronic lymphocytic leukemia treated with ibrutinib. Haematologica. 2015;100(12):1571-1578.
20. Brown JR, Moslehi J, O’Brien S, et al. Characterization of atrial fibrillation adverse events reported in ibrutinib randomized controlled registration trials. Haematologica. 2017;102(10):1796-1805.
Radiotherapeutic Care of Patients With Stage IV Lung Cancer with Thoracic Symptoms in the Veterans Health Administration (FULL)
Lung cancer is the leading cause of cancer mortality both in the US and worldwide.1 Many patients diagnosed with lung cancer present with advanced disease with thoracic symptoms such as cough, hemoptysis, dyspnea, and chest pain.2-4 Palliative radiotherapy is routinely used in patients with locally advanced and metastatic lung cancer with the goal of relieving these symptoms and improving quality of life. Guidelines published by the American Society for Radiation Oncology (ASTRO) in 2011, and updated in 2018, provide recommendations on palliation of lung cancer with external beam radiotherapy (EBRT) and clarify the roles of concurrent chemotherapy and endobronchial brachytherapy (EBB) for palliation.5,6
After prostate cancer, lung cancer is the second most frequently diagnosed cancer in the Veterans Health Administration (VHA).7 The VHA consists of 172 medical centers and is the largest integrated health care system in the US. At the time of this study, 40 of these centers had onsite radiation facilities. The VHA Palliative Radiation Taskforce has conducted a series of surveys to evaluate use of palliative radiotherapy in the VHA, determine VHA practice concordance with ASTRO and American College of Radiology (ACR) guidelines, and direct educational efforts towards addressing gaps in knowledge. These efforts are directed at ensuring best practices throughout this large and heterogeneous healthcare system. In 2016 a survey was conducted to evaluate concordance of VHA radiation oncologist (RO) practice with the 2011 ASTRO guidelines on palliative thoracic radiotherapy for non-small cell lung cancer (NSCLC).
Methods
A survey instrument was generated by VHA National Palliative Radiotherapy Taskforce members. It was reviewed and approved for use by the VHA Patient Care Services office. In May of 2016, the online survey was sent to the 88 VHA ROs practicing at the 40 sites with onsite radiation facilities. The survey aimed to determine patterns of practice for palliation of thoracic symptoms secondary to lung cancer.
Demographic information obtained included years in practice, employment status, academic appointment, board certification, and familiarity with ASTRO lung cancer guidelines. Two clinical scenarios were presented to glean opinions on dose/fractionation schemes preferred, use of concurrent chemotherapy, and use of EBB and/or yttrium aluminum garnet (YAG) laser technology. Survey questions also assessed use of EBRT for palliation of hemoptysis, chest wall pain, and/or stridor as well as use of stereotactic body radiotherapy (SBRT) for palliation.
Survey results were assessed for concordance with published ASTRO guidelines. χ2 tests were run to test for associations between demographic factors such as academic appointment, years of practice, full time vs part time employment, and familiarity with ASTRO palliative lung cancer guidelines, with use of EBRT for palliation, dose and fractionation preference, use of concurrent chemotherapy, and strategy for management of endobronchial lesions.
Results
Of the 88 physicians surveyed, 54 responded for a response rate of 61%. Respondents represented 37 of the 40 (93%) VHA radiation oncology departments (Table 1). Among respondents, most were board certified (96%), held academic appointments (91%), and were full-time employees (85%). Forty-four percent of respondents were in practice for > 20 years, 19% for 11 to 20 years, 20% for 6 to 10 years, and 17% for < 6 years. A majority reported familiarity with the ASTRO guidelines (64%), while just 11% reported no familiarity with the guidelines.
When asked about use of SBRT for palliation of hemoptysis, stridor, and/or chest pain, the majority (87%) preferred conventional EBRT. Of the 13% who reported use of SBRT, most (11%) performed it onsite, with 2% of respondents referring offsite to non-VHA centers for the service. When asked about use of EBB for palliation, only 2% reported use of that procedure at their facilities, while 26% reported referral to non-VHA facilities for EBB. The remaining 72% of respondents favor use of conventional EBRT.
Respondents were presented with a case of a male patient aged 70 years who smoked and had widely metastatic NSCLC, a life expectancy of about 3 months, and 10/10 chest wall pain from direct tumor invasion. All respondents recommended palliative radiotherapy. The preferred fractionation was 20 Gray (Gy) in 5 fractions, which was recommended by 69% of respondents. The remainder recommended 30 Gy in 10 fractions (22%) or a single fraction of 10 Gy (9%). No respondent recommended the longer fractionation options of 60 Gy in 30 fractions, 45 Gy in 15 fractions, or 40 Gy in 20 fractions. The majority (98%) did not recommend concurrent chemotherapy.
When the above case was modified for an endobronchial lesion requiring palliation with associated lung collapse, rather than chest wall invasion, 20 respondents (38%) reported they would refer for EBB, and 20 respondents reported they would refer for YAG laser. As > 1 answer could be selected for this question, there were 12 respondents who selected both EBB and YAG laser; 8 selected only EBB, and 8 selected only YAG laser. Many respondents added comments about treating with EBRT, which had not been presented as an answer choice. Nearly half of respondents (49%) were amenable to referral for the use of EBB or YAG laser for lung reexpansion prior to radiotherapy. Three respondents mentioned referral for an endobronchial stent prior to palliative radiotherapy to address this question.
χ2 tests were used to evaluate for significant associations between demographic factors, such as number of years in practice, academic appointment, full-time vs part-time status, and familiarity with ASTRO guidelines with clinical management choices (Table 2). The χ2 analysis revealed that these demographic factors were not significantly associated with familiarity with ASTRO guidelines, offering SBRT for palliation, EBRT fractionation scheme preferred, use of concurrent chemotherapy, or use of EBB or YAG laser.
Discussion
This survey was conducted to evaluate concordance of management of metastatic lung cancer in the VHA with ASTRO guidelines. The relationship between respondents’ familiarity with the guidelines and responses also was evaluated to determine the impact such guidelines have on decision-making. The ASTRO guidelines for palliative thoracic radiation make recommendations regarding 3 issues: (1) radiation doses and fractionations for palliation; (2) the role of EBB; and (3) the use of concurrent chemotherapy.5,6
Radiation Dose and Fractionation for Palliation
A variety of dose/fractionation schemes are considered appropriate in the ASTRO guideline statement, including more prolonged courses such as 30 Gy/10 fractions as well as more hypofractionated regimens (ie, 20 Gy/5 fractions, 17 Gy/2 fractions, and a single fraction of 10 Gy). Higher dose regimens, such as 30 Gy/10 fractions, have been associated with prolonged survival, as well as increased toxicities such as radiation esophagitis.8 Therefore, the guidelines support use of 30 Gy/10 fractions for patients with good performance status while encouraging use of more hypofractionated regimens for patients with poor performance status. In considering more hypofractionated regimens, one must consider the possibility of adverse effects that can be associated with higher dose per fraction. For instance, 17 Gy/2 fractions has been associated with myelopathy; therefore it should be used with caution and careful treatment planning.9
For the survey case example (a male aged 70 years with a 3-month life expectancy who required palliation for chest wall pain), all respondents selected hypofractionated regimens; with no respondent selected the more prolonged fractionations of 60 Gy/30 fractions, 45 Gy/15 fractions, or 40 Gy/20 fractions. These more prolonged fractionations are not endorsed by the guidelines in general, and particularly not for a patient with poor life expectancy. All responses for this case selected by survey respondents are considered appropriate per the consensus guideline statement.
Role of Concurrent Chemotherapy
The ASTRO guidelines do not support use of concurrent chemotherapy for palliation of stage IV NSCLC.5,6 The 2018 updated guidelines established a role for concurrent chemotherapy for patients with stage III NSCLC with good performance status and life expectancy of > 3 months. This updated recommendation is based on data from 2 randomized trials demonstrating improvement in overall survival with the addition of chemotherapy for patients with stage III NSCLC undergoing palliative radiotherapy.10-12
These newer studies are in contrast to an older randomized study by Ball and colleagues that demonstrated greater toxicity from concurrent chemotherapy, with no improvement in outcomes such as palliation of symptoms, overall survival, or progression free survival.13 In contrast to the newer studies that included only patients with stage III NSCLC, about half of the patients in the Ball and colleagues study had known metastatic disease.10-13 Of note, staging for metastatic disease was not carried out routinely, so it is possible that a greater proportion of patients had metastatic disease that would have been seen on imaging. In concordance with the guidelines, 98% of the survey respondents did not recommend concurrent chemotherapy for palliation of intrathoracic symptom; only 1 respondent recommended use of chemotherapy for palliation.
Role of Endobronchial Brachytherapy
EBB involves implantation of radioactive sources for treatment of endobronchial lesions causing obstructive symptoms.14 Given the lack of randomized data that demonstrate a benefit of EBB over EBRT, the ASTRO guidelines do not endorse routine use of EBB for initial palliative management.15,16 The ASTRO guidelines reference a Cochrane Review of 13 trials that concluded that EBRT alone is superior to EBB alone for initial palliation of symptoms from endobronchial NSCLC.17
Of respondents surveyed, only 1 facility offered onsite EBB. The majority of respondents (72%) preferred the use of conventional EBRT techniques, while 26% refer to non-VHA centers for EBB. Lack of incorporation of EBB into routine VHA practice likely is a reflection of the unclear role of this technology based on the available literature and ASTRO guidelines. In the setting of a right lower lung collapse, more respondents (49%) would consider use of EBB or YAG laser technology for lung reexpansion prior to EBRT.
The ASTRO guidelines recommend that initial EBB in conjunction with EBRT be considered based on randomized data demonstrating significant improvement in lung reexpansion and in patient reported dyspnea with addition of EBB to EBRT over EBRT alone.18 However, the guidelines do not mandate the use of EBB in this situation. It is possible that targeted education regarding the role of EBB would improve knowledge of the potential benefit in the setting of lung collapse and increase the percentage of VHA ROs who would recommend this procedure.
Limitations
The study is limited by lack of generalizability of these findings to all ROs in the country. It is also possible that physician responses do not represent practice patterns with complete accuracy. The use of EBB varied among practitioners. Further study of this technology is necessary to clarify its role in the management of endobronchial obstructive symptoms and to determine whether efforts should be made to increase access to EBB within the VHA.
Conclusions
Most of the ROs who responded to our survey were cognizant and compliant with current ASTRO guidelines on management of lung cancer. Furthermore, familiarity with ASTRO guidelines and management choices were not associated with the respondents’ years in practice, academic appointment, full-time vs part-time status, or familiarity with ASTRO guidelines. This study is a nationwide survey of ROs in the VHA system that reflects the radiation-related care received by veterans with metastatic lung cancer. Responses were obtained from 93% of the 40 radiation oncology centers, so it is likely that the survey accurately represents the decision-making process at the majority of centers. It is possible that those who did not respond to the survey do not treat thoracic cases.
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015 65(2):87-108.
2. Kocher F, Hilbe W, Seeber A, et al. Longitudinal analysis of 2293 NSCLC patients: a comprehensive study from the TYROL registry. Lung Cancer. 2015;87(2):193-200.
3. Chute CG, Greenberg ER, Baron J, Korson R, Baker J, Yates J. Presenting conditions of 1539 population-based lung cancer patients by cell type and stage in New Hampshire and Vermont. Cancer. 1985;56(8):2107-2111.
4. Hyde L, Hyde Cl. Clinical manifestations of lung cancer. Chest. 1974;65(3):299-306.
5. Rodrigues G, Videtic GM, Sur R, et al. Palliative thoracic radiotherapy in lung cancer: An American Society for Radiation Oncology evidence-based clinical practice guideline. Pract Radiat Oncol. 2011;1(2):60-71.
6. Moeller B, Balagamwala EH, Chen A, et al. Palliative thoracic radiation therapy for non-small cell lung cancer: 2018 Update of an American Society for Radiation Oncology (ASTRO) Evidence-Based Guideline. Pract Radiat Oncol. 2018;8(4):245-250.
7. Zullig LL, Jackson GL, Dorn RA, et al. Cancer incidence among patients of the United States Veterans Affairs (VA) healthcare system. Mil Med. 2012;177(6):693-701.
8. Fairchild A, Harris K, Barnes E, et al. Palliative thoracic radiotherapy for lung cancer: a systematic review. J Clin Oncol. 2008;26(24):4001-4011.
9. A Medical Research Council (MRC) randomised trial of palliative radiotherapy with two fractions or a single fraction in patients with inoperable non-small-cell lung cancer (NSCLC) and poor performance status. Medical Research Council Lung Cancer Working Party. Br J Cancer. 1992;65(6):934-941.
10. Nawrocki S, Krzakowski M, Wasilewska-Tesluk E, et al. Concurrent chemotherapy and short course radiotherapy in patients with stage IIIA to IIIB non-small cell lung cancer not eligible for radical treatment: results of a randomized phase II study. J Thorac Oncol. 2010;5(8):1255-1262.
11. Strøm HH, Bremnes RM, Sundstrøm SH, Helbekkmo N, Fløtten O, Aasebø U. Concurrent palliative chemoradiation leads to survival and quality of life benefits in poor prognosis stage III non-small-cell lung cancer: a randomised trial by the Norwegian Lung Cancer Study Group. Br J Cancer. 2013;109(6):1467-1475.
12. Strøm HH, Bremnes RM, Sundstrøm SH, Helbekkmo N, Aasebø U. Poor prognosis patients with inoperable locally advanced NSCLC and large tumors benefit from palliative chemoradiotherapy: a subset analysis from a randomized clinical phase III trial. J Thorac Oncol. 2014;9(6):825-833.
13. Ball D, Smith J, Bishop J, et al. A phase III study of radiotherapy with and without continuous-infusion fluorouracil as palliation for non-small-cell lung cancer. Br J Cancer. 1997;75(5):690-697.
14. Stewart A, Parashar B, Patel M, et al. American Brachytherapy Society consensus guidelines for thoracic brachytherapy for lung cancer. Brachytherapy. 2016;15(1):1-11.
15. Sur R, Ahmed SN, Donde B, Morar R, Mohamed G, Sur M, Pacella JA, Van der Merwe E, Feldman C. Brachytherapy boost vs teletherapy boost in palliation of symptomatic, locally advanced non-small cell lung cancer: preliminary analysis of a randomized prospective study. J Brachytherapy Int. 2001;17(4):309-315.
16. Sur R, Donde B, Mohuiddin M, et al. Randomized prospective study on the role of high dose rate intraluminal brachytherapy (HDRILBT) in palliation of symptoms in advanced non-small cell lung cancer (NSCLC) treated with radiation alone. Int J Radiat Oncol Biol Phys. 2004;60(1):S205.
17. Ung YC, Yu E, Falkson C, et al. The role of high-dose-rate brachytherapy in the palliation of symptoms in patients with non-small cell lung cancer: a systematic review. Brachytherapy. 2006;5:189-202.
18. Langendijk H, de Jong J, Tjwa M, et al. External irradiation versus external irradiation plus endobronchial brachytherapy in inoperable non-small cell lung cancer: a prospective randomized study. Radiother Oncol. 2001;58(3):257-268.
Lung cancer is the leading cause of cancer mortality both in the US and worldwide.1 Many patients diagnosed with lung cancer present with advanced disease with thoracic symptoms such as cough, hemoptysis, dyspnea, and chest pain.2-4 Palliative radiotherapy is routinely used in patients with locally advanced and metastatic lung cancer with the goal of relieving these symptoms and improving quality of life. Guidelines published by the American Society for Radiation Oncology (ASTRO) in 2011, and updated in 2018, provide recommendations on palliation of lung cancer with external beam radiotherapy (EBRT) and clarify the roles of concurrent chemotherapy and endobronchial brachytherapy (EBB) for palliation.5,6
After prostate cancer, lung cancer is the second most frequently diagnosed cancer in the Veterans Health Administration (VHA).7 The VHA consists of 172 medical centers and is the largest integrated health care system in the US. At the time of this study, 40 of these centers had onsite radiation facilities. The VHA Palliative Radiation Taskforce has conducted a series of surveys to evaluate use of palliative radiotherapy in the VHA, determine VHA practice concordance with ASTRO and American College of Radiology (ACR) guidelines, and direct educational efforts towards addressing gaps in knowledge. These efforts are directed at ensuring best practices throughout this large and heterogeneous healthcare system. In 2016 a survey was conducted to evaluate concordance of VHA radiation oncologist (RO) practice with the 2011 ASTRO guidelines on palliative thoracic radiotherapy for non-small cell lung cancer (NSCLC).
Methods
A survey instrument was generated by VHA National Palliative Radiotherapy Taskforce members. It was reviewed and approved for use by the VHA Patient Care Services office. In May of 2016, the online survey was sent to the 88 VHA ROs practicing at the 40 sites with onsite radiation facilities. The survey aimed to determine patterns of practice for palliation of thoracic symptoms secondary to lung cancer.
Demographic information obtained included years in practice, employment status, academic appointment, board certification, and familiarity with ASTRO lung cancer guidelines. Two clinical scenarios were presented to glean opinions on dose/fractionation schemes preferred, use of concurrent chemotherapy, and use of EBB and/or yttrium aluminum garnet (YAG) laser technology. Survey questions also assessed use of EBRT for palliation of hemoptysis, chest wall pain, and/or stridor as well as use of stereotactic body radiotherapy (SBRT) for palliation.
Survey results were assessed for concordance with published ASTRO guidelines. χ2 tests were run to test for associations between demographic factors such as academic appointment, years of practice, full time vs part time employment, and familiarity with ASTRO palliative lung cancer guidelines, with use of EBRT for palliation, dose and fractionation preference, use of concurrent chemotherapy, and strategy for management of endobronchial lesions.
Results
Of the 88 physicians surveyed, 54 responded for a response rate of 61%. Respondents represented 37 of the 40 (93%) VHA radiation oncology departments (Table 1). Among respondents, most were board certified (96%), held academic appointments (91%), and were full-time employees (85%). Forty-four percent of respondents were in practice for > 20 years, 19% for 11 to 20 years, 20% for 6 to 10 years, and 17% for < 6 years. A majority reported familiarity with the ASTRO guidelines (64%), while just 11% reported no familiarity with the guidelines.
When asked about use of SBRT for palliation of hemoptysis, stridor, and/or chest pain, the majority (87%) preferred conventional EBRT. Of the 13% who reported use of SBRT, most (11%) performed it onsite, with 2% of respondents referring offsite to non-VHA centers for the service. When asked about use of EBB for palliation, only 2% reported use of that procedure at their facilities, while 26% reported referral to non-VHA facilities for EBB. The remaining 72% of respondents favor use of conventional EBRT.
Respondents were presented with a case of a male patient aged 70 years who smoked and had widely metastatic NSCLC, a life expectancy of about 3 months, and 10/10 chest wall pain from direct tumor invasion. All respondents recommended palliative radiotherapy. The preferred fractionation was 20 Gray (Gy) in 5 fractions, which was recommended by 69% of respondents. The remainder recommended 30 Gy in 10 fractions (22%) or a single fraction of 10 Gy (9%). No respondent recommended the longer fractionation options of 60 Gy in 30 fractions, 45 Gy in 15 fractions, or 40 Gy in 20 fractions. The majority (98%) did not recommend concurrent chemotherapy.
When the above case was modified for an endobronchial lesion requiring palliation with associated lung collapse, rather than chest wall invasion, 20 respondents (38%) reported they would refer for EBB, and 20 respondents reported they would refer for YAG laser. As > 1 answer could be selected for this question, there were 12 respondents who selected both EBB and YAG laser; 8 selected only EBB, and 8 selected only YAG laser. Many respondents added comments about treating with EBRT, which had not been presented as an answer choice. Nearly half of respondents (49%) were amenable to referral for the use of EBB or YAG laser for lung reexpansion prior to radiotherapy. Three respondents mentioned referral for an endobronchial stent prior to palliative radiotherapy to address this question.
χ2 tests were used to evaluate for significant associations between demographic factors, such as number of years in practice, academic appointment, full-time vs part-time status, and familiarity with ASTRO guidelines with clinical management choices (Table 2). The χ2 analysis revealed that these demographic factors were not significantly associated with familiarity with ASTRO guidelines, offering SBRT for palliation, EBRT fractionation scheme preferred, use of concurrent chemotherapy, or use of EBB or YAG laser.
Discussion
This survey was conducted to evaluate concordance of management of metastatic lung cancer in the VHA with ASTRO guidelines. The relationship between respondents’ familiarity with the guidelines and responses also was evaluated to determine the impact such guidelines have on decision-making. The ASTRO guidelines for palliative thoracic radiation make recommendations regarding 3 issues: (1) radiation doses and fractionations for palliation; (2) the role of EBB; and (3) the use of concurrent chemotherapy.5,6
Radiation Dose and Fractionation for Palliation
A variety of dose/fractionation schemes are considered appropriate in the ASTRO guideline statement, including more prolonged courses such as 30 Gy/10 fractions as well as more hypofractionated regimens (ie, 20 Gy/5 fractions, 17 Gy/2 fractions, and a single fraction of 10 Gy). Higher dose regimens, such as 30 Gy/10 fractions, have been associated with prolonged survival, as well as increased toxicities such as radiation esophagitis.8 Therefore, the guidelines support use of 30 Gy/10 fractions for patients with good performance status while encouraging use of more hypofractionated regimens for patients with poor performance status. In considering more hypofractionated regimens, one must consider the possibility of adverse effects that can be associated with higher dose per fraction. For instance, 17 Gy/2 fractions has been associated with myelopathy; therefore it should be used with caution and careful treatment planning.9
For the survey case example (a male aged 70 years with a 3-month life expectancy who required palliation for chest wall pain), all respondents selected hypofractionated regimens; with no respondent selected the more prolonged fractionations of 60 Gy/30 fractions, 45 Gy/15 fractions, or 40 Gy/20 fractions. These more prolonged fractionations are not endorsed by the guidelines in general, and particularly not for a patient with poor life expectancy. All responses for this case selected by survey respondents are considered appropriate per the consensus guideline statement.
Role of Concurrent Chemotherapy
The ASTRO guidelines do not support use of concurrent chemotherapy for palliation of stage IV NSCLC.5,6 The 2018 updated guidelines established a role for concurrent chemotherapy for patients with stage III NSCLC with good performance status and life expectancy of > 3 months. This updated recommendation is based on data from 2 randomized trials demonstrating improvement in overall survival with the addition of chemotherapy for patients with stage III NSCLC undergoing palliative radiotherapy.10-12
These newer studies are in contrast to an older randomized study by Ball and colleagues that demonstrated greater toxicity from concurrent chemotherapy, with no improvement in outcomes such as palliation of symptoms, overall survival, or progression free survival.13 In contrast to the newer studies that included only patients with stage III NSCLC, about half of the patients in the Ball and colleagues study had known metastatic disease.10-13 Of note, staging for metastatic disease was not carried out routinely, so it is possible that a greater proportion of patients had metastatic disease that would have been seen on imaging. In concordance with the guidelines, 98% of the survey respondents did not recommend concurrent chemotherapy for palliation of intrathoracic symptom; only 1 respondent recommended use of chemotherapy for palliation.
Role of Endobronchial Brachytherapy
EBB involves implantation of radioactive sources for treatment of endobronchial lesions causing obstructive symptoms.14 Given the lack of randomized data that demonstrate a benefit of EBB over EBRT, the ASTRO guidelines do not endorse routine use of EBB for initial palliative management.15,16 The ASTRO guidelines reference a Cochrane Review of 13 trials that concluded that EBRT alone is superior to EBB alone for initial palliation of symptoms from endobronchial NSCLC.17
Of respondents surveyed, only 1 facility offered onsite EBB. The majority of respondents (72%) preferred the use of conventional EBRT techniques, while 26% refer to non-VHA centers for EBB. Lack of incorporation of EBB into routine VHA practice likely is a reflection of the unclear role of this technology based on the available literature and ASTRO guidelines. In the setting of a right lower lung collapse, more respondents (49%) would consider use of EBB or YAG laser technology for lung reexpansion prior to EBRT.
The ASTRO guidelines recommend that initial EBB in conjunction with EBRT be considered based on randomized data demonstrating significant improvement in lung reexpansion and in patient reported dyspnea with addition of EBB to EBRT over EBRT alone.18 However, the guidelines do not mandate the use of EBB in this situation. It is possible that targeted education regarding the role of EBB would improve knowledge of the potential benefit in the setting of lung collapse and increase the percentage of VHA ROs who would recommend this procedure.
Limitations
The study is limited by lack of generalizability of these findings to all ROs in the country. It is also possible that physician responses do not represent practice patterns with complete accuracy. The use of EBB varied among practitioners. Further study of this technology is necessary to clarify its role in the management of endobronchial obstructive symptoms and to determine whether efforts should be made to increase access to EBB within the VHA.
Conclusions
Most of the ROs who responded to our survey were cognizant and compliant with current ASTRO guidelines on management of lung cancer. Furthermore, familiarity with ASTRO guidelines and management choices were not associated with the respondents’ years in practice, academic appointment, full-time vs part-time status, or familiarity with ASTRO guidelines. This study is a nationwide survey of ROs in the VHA system that reflects the radiation-related care received by veterans with metastatic lung cancer. Responses were obtained from 93% of the 40 radiation oncology centers, so it is likely that the survey accurately represents the decision-making process at the majority of centers. It is possible that those who did not respond to the survey do not treat thoracic cases.
Lung cancer is the leading cause of cancer mortality both in the US and worldwide.1 Many patients diagnosed with lung cancer present with advanced disease with thoracic symptoms such as cough, hemoptysis, dyspnea, and chest pain.2-4 Palliative radiotherapy is routinely used in patients with locally advanced and metastatic lung cancer with the goal of relieving these symptoms and improving quality of life. Guidelines published by the American Society for Radiation Oncology (ASTRO) in 2011, and updated in 2018, provide recommendations on palliation of lung cancer with external beam radiotherapy (EBRT) and clarify the roles of concurrent chemotherapy and endobronchial brachytherapy (EBB) for palliation.5,6
After prostate cancer, lung cancer is the second most frequently diagnosed cancer in the Veterans Health Administration (VHA).7 The VHA consists of 172 medical centers and is the largest integrated health care system in the US. At the time of this study, 40 of these centers had onsite radiation facilities. The VHA Palliative Radiation Taskforce has conducted a series of surveys to evaluate use of palliative radiotherapy in the VHA, determine VHA practice concordance with ASTRO and American College of Radiology (ACR) guidelines, and direct educational efforts towards addressing gaps in knowledge. These efforts are directed at ensuring best practices throughout this large and heterogeneous healthcare system. In 2016 a survey was conducted to evaluate concordance of VHA radiation oncologist (RO) practice with the 2011 ASTRO guidelines on palliative thoracic radiotherapy for non-small cell lung cancer (NSCLC).
Methods
A survey instrument was generated by VHA National Palliative Radiotherapy Taskforce members. It was reviewed and approved for use by the VHA Patient Care Services office. In May of 2016, the online survey was sent to the 88 VHA ROs practicing at the 40 sites with onsite radiation facilities. The survey aimed to determine patterns of practice for palliation of thoracic symptoms secondary to lung cancer.
Demographic information obtained included years in practice, employment status, academic appointment, board certification, and familiarity with ASTRO lung cancer guidelines. Two clinical scenarios were presented to glean opinions on dose/fractionation schemes preferred, use of concurrent chemotherapy, and use of EBB and/or yttrium aluminum garnet (YAG) laser technology. Survey questions also assessed use of EBRT for palliation of hemoptysis, chest wall pain, and/or stridor as well as use of stereotactic body radiotherapy (SBRT) for palliation.
Survey results were assessed for concordance with published ASTRO guidelines. χ2 tests were run to test for associations between demographic factors such as academic appointment, years of practice, full time vs part time employment, and familiarity with ASTRO palliative lung cancer guidelines, with use of EBRT for palliation, dose and fractionation preference, use of concurrent chemotherapy, and strategy for management of endobronchial lesions.
Results
Of the 88 physicians surveyed, 54 responded for a response rate of 61%. Respondents represented 37 of the 40 (93%) VHA radiation oncology departments (Table 1). Among respondents, most were board certified (96%), held academic appointments (91%), and were full-time employees (85%). Forty-four percent of respondents were in practice for > 20 years, 19% for 11 to 20 years, 20% for 6 to 10 years, and 17% for < 6 years. A majority reported familiarity with the ASTRO guidelines (64%), while just 11% reported no familiarity with the guidelines.
When asked about use of SBRT for palliation of hemoptysis, stridor, and/or chest pain, the majority (87%) preferred conventional EBRT. Of the 13% who reported use of SBRT, most (11%) performed it onsite, with 2% of respondents referring offsite to non-VHA centers for the service. When asked about use of EBB for palliation, only 2% reported use of that procedure at their facilities, while 26% reported referral to non-VHA facilities for EBB. The remaining 72% of respondents favor use of conventional EBRT.
Respondents were presented with a case of a male patient aged 70 years who smoked and had widely metastatic NSCLC, a life expectancy of about 3 months, and 10/10 chest wall pain from direct tumor invasion. All respondents recommended palliative radiotherapy. The preferred fractionation was 20 Gray (Gy) in 5 fractions, which was recommended by 69% of respondents. The remainder recommended 30 Gy in 10 fractions (22%) or a single fraction of 10 Gy (9%). No respondent recommended the longer fractionation options of 60 Gy in 30 fractions, 45 Gy in 15 fractions, or 40 Gy in 20 fractions. The majority (98%) did not recommend concurrent chemotherapy.
When the above case was modified for an endobronchial lesion requiring palliation with associated lung collapse, rather than chest wall invasion, 20 respondents (38%) reported they would refer for EBB, and 20 respondents reported they would refer for YAG laser. As > 1 answer could be selected for this question, there were 12 respondents who selected both EBB and YAG laser; 8 selected only EBB, and 8 selected only YAG laser. Many respondents added comments about treating with EBRT, which had not been presented as an answer choice. Nearly half of respondents (49%) were amenable to referral for the use of EBB or YAG laser for lung reexpansion prior to radiotherapy. Three respondents mentioned referral for an endobronchial stent prior to palliative radiotherapy to address this question.
χ2 tests were used to evaluate for significant associations between demographic factors, such as number of years in practice, academic appointment, full-time vs part-time status, and familiarity with ASTRO guidelines with clinical management choices (Table 2). The χ2 analysis revealed that these demographic factors were not significantly associated with familiarity with ASTRO guidelines, offering SBRT for palliation, EBRT fractionation scheme preferred, use of concurrent chemotherapy, or use of EBB or YAG laser.
Discussion
This survey was conducted to evaluate concordance of management of metastatic lung cancer in the VHA with ASTRO guidelines. The relationship between respondents’ familiarity with the guidelines and responses also was evaluated to determine the impact such guidelines have on decision-making. The ASTRO guidelines for palliative thoracic radiation make recommendations regarding 3 issues: (1) radiation doses and fractionations for palliation; (2) the role of EBB; and (3) the use of concurrent chemotherapy.5,6
Radiation Dose and Fractionation for Palliation
A variety of dose/fractionation schemes are considered appropriate in the ASTRO guideline statement, including more prolonged courses such as 30 Gy/10 fractions as well as more hypofractionated regimens (ie, 20 Gy/5 fractions, 17 Gy/2 fractions, and a single fraction of 10 Gy). Higher dose regimens, such as 30 Gy/10 fractions, have been associated with prolonged survival, as well as increased toxicities such as radiation esophagitis.8 Therefore, the guidelines support use of 30 Gy/10 fractions for patients with good performance status while encouraging use of more hypofractionated regimens for patients with poor performance status. In considering more hypofractionated regimens, one must consider the possibility of adverse effects that can be associated with higher dose per fraction. For instance, 17 Gy/2 fractions has been associated with myelopathy; therefore it should be used with caution and careful treatment planning.9
For the survey case example (a male aged 70 years with a 3-month life expectancy who required palliation for chest wall pain), all respondents selected hypofractionated regimens; with no respondent selected the more prolonged fractionations of 60 Gy/30 fractions, 45 Gy/15 fractions, or 40 Gy/20 fractions. These more prolonged fractionations are not endorsed by the guidelines in general, and particularly not for a patient with poor life expectancy. All responses for this case selected by survey respondents are considered appropriate per the consensus guideline statement.
Role of Concurrent Chemotherapy
The ASTRO guidelines do not support use of concurrent chemotherapy for palliation of stage IV NSCLC.5,6 The 2018 updated guidelines established a role for concurrent chemotherapy for patients with stage III NSCLC with good performance status and life expectancy of > 3 months. This updated recommendation is based on data from 2 randomized trials demonstrating improvement in overall survival with the addition of chemotherapy for patients with stage III NSCLC undergoing palliative radiotherapy.10-12
These newer studies are in contrast to an older randomized study by Ball and colleagues that demonstrated greater toxicity from concurrent chemotherapy, with no improvement in outcomes such as palliation of symptoms, overall survival, or progression free survival.13 In contrast to the newer studies that included only patients with stage III NSCLC, about half of the patients in the Ball and colleagues study had known metastatic disease.10-13 Of note, staging for metastatic disease was not carried out routinely, so it is possible that a greater proportion of patients had metastatic disease that would have been seen on imaging. In concordance with the guidelines, 98% of the survey respondents did not recommend concurrent chemotherapy for palliation of intrathoracic symptom; only 1 respondent recommended use of chemotherapy for palliation.
Role of Endobronchial Brachytherapy
EBB involves implantation of radioactive sources for treatment of endobronchial lesions causing obstructive symptoms.14 Given the lack of randomized data that demonstrate a benefit of EBB over EBRT, the ASTRO guidelines do not endorse routine use of EBB for initial palliative management.15,16 The ASTRO guidelines reference a Cochrane Review of 13 trials that concluded that EBRT alone is superior to EBB alone for initial palliation of symptoms from endobronchial NSCLC.17
Of respondents surveyed, only 1 facility offered onsite EBB. The majority of respondents (72%) preferred the use of conventional EBRT techniques, while 26% refer to non-VHA centers for EBB. Lack of incorporation of EBB into routine VHA practice likely is a reflection of the unclear role of this technology based on the available literature and ASTRO guidelines. In the setting of a right lower lung collapse, more respondents (49%) would consider use of EBB or YAG laser technology for lung reexpansion prior to EBRT.
The ASTRO guidelines recommend that initial EBB in conjunction with EBRT be considered based on randomized data demonstrating significant improvement in lung reexpansion and in patient reported dyspnea with addition of EBB to EBRT over EBRT alone.18 However, the guidelines do not mandate the use of EBB in this situation. It is possible that targeted education regarding the role of EBB would improve knowledge of the potential benefit in the setting of lung collapse and increase the percentage of VHA ROs who would recommend this procedure.
Limitations
The study is limited by lack of generalizability of these findings to all ROs in the country. It is also possible that physician responses do not represent practice patterns with complete accuracy. The use of EBB varied among practitioners. Further study of this technology is necessary to clarify its role in the management of endobronchial obstructive symptoms and to determine whether efforts should be made to increase access to EBB within the VHA.
Conclusions
Most of the ROs who responded to our survey were cognizant and compliant with current ASTRO guidelines on management of lung cancer. Furthermore, familiarity with ASTRO guidelines and management choices were not associated with the respondents’ years in practice, academic appointment, full-time vs part-time status, or familiarity with ASTRO guidelines. This study is a nationwide survey of ROs in the VHA system that reflects the radiation-related care received by veterans with metastatic lung cancer. Responses were obtained from 93% of the 40 radiation oncology centers, so it is likely that the survey accurately represents the decision-making process at the majority of centers. It is possible that those who did not respond to the survey do not treat thoracic cases.
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015 65(2):87-108.
2. Kocher F, Hilbe W, Seeber A, et al. Longitudinal analysis of 2293 NSCLC patients: a comprehensive study from the TYROL registry. Lung Cancer. 2015;87(2):193-200.
3. Chute CG, Greenberg ER, Baron J, Korson R, Baker J, Yates J. Presenting conditions of 1539 population-based lung cancer patients by cell type and stage in New Hampshire and Vermont. Cancer. 1985;56(8):2107-2111.
4. Hyde L, Hyde Cl. Clinical manifestations of lung cancer. Chest. 1974;65(3):299-306.
5. Rodrigues G, Videtic GM, Sur R, et al. Palliative thoracic radiotherapy in lung cancer: An American Society for Radiation Oncology evidence-based clinical practice guideline. Pract Radiat Oncol. 2011;1(2):60-71.
6. Moeller B, Balagamwala EH, Chen A, et al. Palliative thoracic radiation therapy for non-small cell lung cancer: 2018 Update of an American Society for Radiation Oncology (ASTRO) Evidence-Based Guideline. Pract Radiat Oncol. 2018;8(4):245-250.
7. Zullig LL, Jackson GL, Dorn RA, et al. Cancer incidence among patients of the United States Veterans Affairs (VA) healthcare system. Mil Med. 2012;177(6):693-701.
8. Fairchild A, Harris K, Barnes E, et al. Palliative thoracic radiotherapy for lung cancer: a systematic review. J Clin Oncol. 2008;26(24):4001-4011.
9. A Medical Research Council (MRC) randomised trial of palliative radiotherapy with two fractions or a single fraction in patients with inoperable non-small-cell lung cancer (NSCLC) and poor performance status. Medical Research Council Lung Cancer Working Party. Br J Cancer. 1992;65(6):934-941.
10. Nawrocki S, Krzakowski M, Wasilewska-Tesluk E, et al. Concurrent chemotherapy and short course radiotherapy in patients with stage IIIA to IIIB non-small cell lung cancer not eligible for radical treatment: results of a randomized phase II study. J Thorac Oncol. 2010;5(8):1255-1262.
11. Strøm HH, Bremnes RM, Sundstrøm SH, Helbekkmo N, Fløtten O, Aasebø U. Concurrent palliative chemoradiation leads to survival and quality of life benefits in poor prognosis stage III non-small-cell lung cancer: a randomised trial by the Norwegian Lung Cancer Study Group. Br J Cancer. 2013;109(6):1467-1475.
12. Strøm HH, Bremnes RM, Sundstrøm SH, Helbekkmo N, Aasebø U. Poor prognosis patients with inoperable locally advanced NSCLC and large tumors benefit from palliative chemoradiotherapy: a subset analysis from a randomized clinical phase III trial. J Thorac Oncol. 2014;9(6):825-833.
13. Ball D, Smith J, Bishop J, et al. A phase III study of radiotherapy with and without continuous-infusion fluorouracil as palliation for non-small-cell lung cancer. Br J Cancer. 1997;75(5):690-697.
14. Stewart A, Parashar B, Patel M, et al. American Brachytherapy Society consensus guidelines for thoracic brachytherapy for lung cancer. Brachytherapy. 2016;15(1):1-11.
15. Sur R, Ahmed SN, Donde B, Morar R, Mohamed G, Sur M, Pacella JA, Van der Merwe E, Feldman C. Brachytherapy boost vs teletherapy boost in palliation of symptomatic, locally advanced non-small cell lung cancer: preliminary analysis of a randomized prospective study. J Brachytherapy Int. 2001;17(4):309-315.
16. Sur R, Donde B, Mohuiddin M, et al. Randomized prospective study on the role of high dose rate intraluminal brachytherapy (HDRILBT) in palliation of symptoms in advanced non-small cell lung cancer (NSCLC) treated with radiation alone. Int J Radiat Oncol Biol Phys. 2004;60(1):S205.
17. Ung YC, Yu E, Falkson C, et al. The role of high-dose-rate brachytherapy in the palliation of symptoms in patients with non-small cell lung cancer: a systematic review. Brachytherapy. 2006;5:189-202.
18. Langendijk H, de Jong J, Tjwa M, et al. External irradiation versus external irradiation plus endobronchial brachytherapy in inoperable non-small cell lung cancer: a prospective randomized study. Radiother Oncol. 2001;58(3):257-268.
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015 65(2):87-108.
2. Kocher F, Hilbe W, Seeber A, et al. Longitudinal analysis of 2293 NSCLC patients: a comprehensive study from the TYROL registry. Lung Cancer. 2015;87(2):193-200.
3. Chute CG, Greenberg ER, Baron J, Korson R, Baker J, Yates J. Presenting conditions of 1539 population-based lung cancer patients by cell type and stage in New Hampshire and Vermont. Cancer. 1985;56(8):2107-2111.
4. Hyde L, Hyde Cl. Clinical manifestations of lung cancer. Chest. 1974;65(3):299-306.
5. Rodrigues G, Videtic GM, Sur R, et al. Palliative thoracic radiotherapy in lung cancer: An American Society for Radiation Oncology evidence-based clinical practice guideline. Pract Radiat Oncol. 2011;1(2):60-71.
6. Moeller B, Balagamwala EH, Chen A, et al. Palliative thoracic radiation therapy for non-small cell lung cancer: 2018 Update of an American Society for Radiation Oncology (ASTRO) Evidence-Based Guideline. Pract Radiat Oncol. 2018;8(4):245-250.
7. Zullig LL, Jackson GL, Dorn RA, et al. Cancer incidence among patients of the United States Veterans Affairs (VA) healthcare system. Mil Med. 2012;177(6):693-701.
8. Fairchild A, Harris K, Barnes E, et al. Palliative thoracic radiotherapy for lung cancer: a systematic review. J Clin Oncol. 2008;26(24):4001-4011.
9. A Medical Research Council (MRC) randomised trial of palliative radiotherapy with two fractions or a single fraction in patients with inoperable non-small-cell lung cancer (NSCLC) and poor performance status. Medical Research Council Lung Cancer Working Party. Br J Cancer. 1992;65(6):934-941.
10. Nawrocki S, Krzakowski M, Wasilewska-Tesluk E, et al. Concurrent chemotherapy and short course radiotherapy in patients with stage IIIA to IIIB non-small cell lung cancer not eligible for radical treatment: results of a randomized phase II study. J Thorac Oncol. 2010;5(8):1255-1262.
11. Strøm HH, Bremnes RM, Sundstrøm SH, Helbekkmo N, Fløtten O, Aasebø U. Concurrent palliative chemoradiation leads to survival and quality of life benefits in poor prognosis stage III non-small-cell lung cancer: a randomised trial by the Norwegian Lung Cancer Study Group. Br J Cancer. 2013;109(6):1467-1475.
12. Strøm HH, Bremnes RM, Sundstrøm SH, Helbekkmo N, Aasebø U. Poor prognosis patients with inoperable locally advanced NSCLC and large tumors benefit from palliative chemoradiotherapy: a subset analysis from a randomized clinical phase III trial. J Thorac Oncol. 2014;9(6):825-833.
13. Ball D, Smith J, Bishop J, et al. A phase III study of radiotherapy with and without continuous-infusion fluorouracil as palliation for non-small-cell lung cancer. Br J Cancer. 1997;75(5):690-697.
14. Stewart A, Parashar B, Patel M, et al. American Brachytherapy Society consensus guidelines for thoracic brachytherapy for lung cancer. Brachytherapy. 2016;15(1):1-11.
15. Sur R, Ahmed SN, Donde B, Morar R, Mohamed G, Sur M, Pacella JA, Van der Merwe E, Feldman C. Brachytherapy boost vs teletherapy boost in palliation of symptomatic, locally advanced non-small cell lung cancer: preliminary analysis of a randomized prospective study. J Brachytherapy Int. 2001;17(4):309-315.
16. Sur R, Donde B, Mohuiddin M, et al. Randomized prospective study on the role of high dose rate intraluminal brachytherapy (HDRILBT) in palliation of symptoms in advanced non-small cell lung cancer (NSCLC) treated with radiation alone. Int J Radiat Oncol Biol Phys. 2004;60(1):S205.
17. Ung YC, Yu E, Falkson C, et al. The role of high-dose-rate brachytherapy in the palliation of symptoms in patients with non-small cell lung cancer: a systematic review. Brachytherapy. 2006;5:189-202.
18. Langendijk H, de Jong J, Tjwa M, et al. External irradiation versus external irradiation plus endobronchial brachytherapy in inoperable non-small cell lung cancer: a prospective randomized study. Radiother Oncol. 2001;58(3):257-268.
Hyperprogression on immunotherapy: When outcomes are much worse
Immunotherapy with checkpoint inhibitors has ushered in a new era of cancer therapy, with some patients showing dramatic responses and significantly better outcomes than with other therapies across many cancer types. But some patients do worse, sometimes much worse.
A subset of patients who undergo immunotherapy experience unexpected, rapid disease progression, with a dramatic acceleration of disease trajectory. They also have a shorter progression-free survival and overall survival than would have been expected.
This has been described as hyperprogression and has been termed “hyperprogressive disease” (HPD). It has been seen in a variety of cancers; the incidence ranges from 4% to 29% in the studies reported to date.
There has been some debate over whether this is a real phenomenon or whether it is part of the natural course of disease.
HPD is a “provocative phenomenon,” wrote the authors of a recent commentary entitled “Hyperprogression and Immunotherapy: Fact, Fiction, or Alternative Fact?”
“This phenomenon has polarized oncologists who debate that this could still reflect the natural history of the disease,” said the author of another commentary.
But the tide is now turning toward acceptance of HPD, said Kartik Sehgal, MD, an oncologist at Dana-Farber Cancer Institute and Harvard University, both in Boston.
“With publication of multiple clinical reports of different cancer types worldwide, hyperprogression is now accepted by most oncologists to be a true phenomenon rather than natural progression of disease,” Dr. Sehgal said.
He authored an invited commentary in JAMA Network Openabout one of the latest meta-analyses (JAMA Netw Open. 2021;4[3]:e211136) to investigate HPD during immunotherapy. One of the biggest issues is that the studies that have reported on HPD have been retrospective, with a lack of comparator groups and a lack of a standardized definition of hyperprogression. Dr. Sehgal emphasized the need to study hyperprogression in well-designed prospective studies.
Existing data on HPD
HPD was described as “a new pattern of progression” seen in patients undergoing immune checkpoint inhibitor therapy in a 2017 article published in Clinical Cancer Research. Authors Stephane Champiat, MD, PhD, of Institut Gustave Roussy, Universite Paris Saclay, Villejuif, France, and colleagues cited “anecdotal occurrences” of HPD among patients in phase 1 trials of anti–PD-1/PD-L1 agents.
In that study, HPD was defined by tumor growth rate ratio. The incidence was 9% among 213 patients.
The findings raised concerns about treating elderly patients with anti–PD-1/PD-L1 monotherapy, according to the authors, who called for further study.
That same year, Roberto Ferrara, MD, and colleagues from the Insitut Gustave Roussy reported additional data indicating an incidence of HPD of 16% among 333 patients with non–small cell lung cancer who underwent immunotherapy at eight centers from 2012 to 2017. The findings, which were presented at the 2017 World Conference on Lung Cancer and reported at the time by this news organization, also showed that the incidence of HPD was higher with immunotherapy than with single-agent chemotherapy (5%).
Median overall survival (OS) was just 3.4 months among those with HPD, compared with 13 months in the overall study population – worse, even, than the median 5.4-month OS observed among patients with progressive disease who received immunotherapy.
In the wake of these findings, numerous researchers have attempted to better define HPD, its incidence, and patient factors associated with developing HPD while undergoing immunotherapy.
However, there is little so far to show for those efforts, Vivek Subbiah, MD, of the University of Texas MD Anderson Cancer Center, Houston, said in an interview.
“Many questions remain to be answered,” said Dr. Subbiah, clinical medical director of the Clinical Center for Targeted Therapy in the division of cancer medicine at MD Anderson. He was the senior author of the “Fact, Fiction, or Alternative Fact?” commentary.
Work is underway to elucidate biological mechanisms. Some groups have implicated the Fc region of antibodies. Another group has reported EGFR and MDM2/MDM4 amplifications in patients with HPD, Dr. Subbiah and colleagues noted.
Other “proposed contributing pathological mechanisms include modulation of tumor immune microenvironment through macrophages and regulatory T cells as well as activation of oncogenic signaling pathways,” noted Dr. Sehgal.
Both groups of authors emphasize the urgent need for prospective studies.
It is imperative to confirm underlying biology, predict which patients are at risk, and identify therapeutic directions for patients who experience HPD, Dr. Subbiah said.
The main challenge is defining HPD, he added. Definitions that have been proposed include tumor growth at least two times greater than in control persons, a 15% increase in tumor burden in a set period, and disease progression of 50% from the first evaluation before treatment, he said.
The recent meta-analysis by Hyo Jung Park, MD, PhD, and colleagues, which Dr. Sehgal addressed in his invited commentary, highlights the many approaches used for defining HPD.
Depending on the definition used, the incidence of HPD across 24 studies involving more than 3,100 patients ranged from 5.9% to 43.1%.
“Hyperprogressive disease could be overestimated or underestimated based on current assessment,” Dr. Park and colleagues concluded. They highlighted the importance of “establishing uniform and clinically relevant criteria based on currently available evidence.”
Steps for solving the HPD mystery
“I think we need to come up with consensus criteria for an HPD definition. We need a unified definition,” Dr. Subbiah said. “We also need to design prospective studies to prove or disprove the immunotherapy-HPD association.”
Prospective registries with independent review of patients with suspected immunotherapy-related HPD would be useful for assessing the true incidence and the biology of HPD among patients undergoing immunotherapy, he suggested.
“We need to know the immunologic signals of HPD. This can give us an idea if patients can be prospectively identified for being at risk,” he said. “We also need to know what to do if they are at risk.”
Dr. Sehgal also called for consensus on an HPD definition, with input from a multidisciplinary group that includes “colleagues from radiology, medical oncology, radiation oncology. Getting expertise from different disciplines would be helpful,” he said.
Dr. Park and colleagues suggested several key requirements for an optimal HP definition, such as the inclusion of multiple variables for measuring tumor growth acceleration, “sufficiently quantitative” criteria for determining time to failure, and establishment of a standardized measure of tumor growth acceleration.
The agreed-upon definition of HPD could be applied to patients in a prospective registry and to existing trial data, Dr. Sehgal said.
“Eventually, the goal of this exercise is to [determine] how we can help our patients the best, having a biomarker that can at least inform us in terms of being aware and being proactive in terms of looking for this ... so that interventions can be brought on earlier,” he said.
“If we know what may be a biological mechanism, we can design trials that are designed to look at how to overcome that HPD,” he said.
Dr. Sehgal said he believes HPD is triggered in some way by treatment, including immunotherapy, chemotherapy, and targeted therapy, but perhaps in different ways for each.
He estimated the true incidence of immunotherapy-related HPD will be in the 9%-10% range.
“This is a substantial number of patients, so it’s important that we try to understand this phenomenon, using, again, uniform criteria,” he said.
Current treatment decision-making
Until more is known, Dr. Sehgal said he considers the potential risk factors when treating patients with immunotherapy.
For example, the presence of MDM2 or MDM4 amplification on a genomic profile may factor into his treatment decision-making when it comes to using immunotherapy or immunotherapy in combination with chemotherapy, he said.
“Is that the only factor that is going to make me choose one thing or another? No,” Dr. Sehgal said. However, he said it would make him more “proactive in making sure the patient is doing clinically okay” and in determining when to obtain on-treatment imaging studies.
Dr. Subbiah emphasized the relative benefit of immunotherapy, noting that survival with chemotherapy for many difficult-to-treat cancers in the relapsed/refractory metastatic setting is less than 2 years.
Immunotherapy with checkpoint inhibitors has allowed some of these patients to live longer (with survival reported to be more than 10 years for patients with metastatic melanoma).
“Immunotherapy has been a game changer; it has been transformative in the lives of these patients,” Dr. Subbiah said. “So unless there is any other contraindication, the benefit of receiving immunotherapy for an approved indication far outweighs the risk of HPD.”
A version of this article first appeared on Medscape.com.
Immunotherapy with checkpoint inhibitors has ushered in a new era of cancer therapy, with some patients showing dramatic responses and significantly better outcomes than with other therapies across many cancer types. But some patients do worse, sometimes much worse.
A subset of patients who undergo immunotherapy experience unexpected, rapid disease progression, with a dramatic acceleration of disease trajectory. They also have a shorter progression-free survival and overall survival than would have been expected.
This has been described as hyperprogression and has been termed “hyperprogressive disease” (HPD). It has been seen in a variety of cancers; the incidence ranges from 4% to 29% in the studies reported to date.
There has been some debate over whether this is a real phenomenon or whether it is part of the natural course of disease.
HPD is a “provocative phenomenon,” wrote the authors of a recent commentary entitled “Hyperprogression and Immunotherapy: Fact, Fiction, or Alternative Fact?”
“This phenomenon has polarized oncologists who debate that this could still reflect the natural history of the disease,” said the author of another commentary.
But the tide is now turning toward acceptance of HPD, said Kartik Sehgal, MD, an oncologist at Dana-Farber Cancer Institute and Harvard University, both in Boston.
“With publication of multiple clinical reports of different cancer types worldwide, hyperprogression is now accepted by most oncologists to be a true phenomenon rather than natural progression of disease,” Dr. Sehgal said.
He authored an invited commentary in JAMA Network Openabout one of the latest meta-analyses (JAMA Netw Open. 2021;4[3]:e211136) to investigate HPD during immunotherapy. One of the biggest issues is that the studies that have reported on HPD have been retrospective, with a lack of comparator groups and a lack of a standardized definition of hyperprogression. Dr. Sehgal emphasized the need to study hyperprogression in well-designed prospective studies.
Existing data on HPD
HPD was described as “a new pattern of progression” seen in patients undergoing immune checkpoint inhibitor therapy in a 2017 article published in Clinical Cancer Research. Authors Stephane Champiat, MD, PhD, of Institut Gustave Roussy, Universite Paris Saclay, Villejuif, France, and colleagues cited “anecdotal occurrences” of HPD among patients in phase 1 trials of anti–PD-1/PD-L1 agents.
In that study, HPD was defined by tumor growth rate ratio. The incidence was 9% among 213 patients.
The findings raised concerns about treating elderly patients with anti–PD-1/PD-L1 monotherapy, according to the authors, who called for further study.
That same year, Roberto Ferrara, MD, and colleagues from the Insitut Gustave Roussy reported additional data indicating an incidence of HPD of 16% among 333 patients with non–small cell lung cancer who underwent immunotherapy at eight centers from 2012 to 2017. The findings, which were presented at the 2017 World Conference on Lung Cancer and reported at the time by this news organization, also showed that the incidence of HPD was higher with immunotherapy than with single-agent chemotherapy (5%).
Median overall survival (OS) was just 3.4 months among those with HPD, compared with 13 months in the overall study population – worse, even, than the median 5.4-month OS observed among patients with progressive disease who received immunotherapy.
In the wake of these findings, numerous researchers have attempted to better define HPD, its incidence, and patient factors associated with developing HPD while undergoing immunotherapy.
However, there is little so far to show for those efforts, Vivek Subbiah, MD, of the University of Texas MD Anderson Cancer Center, Houston, said in an interview.
“Many questions remain to be answered,” said Dr. Subbiah, clinical medical director of the Clinical Center for Targeted Therapy in the division of cancer medicine at MD Anderson. He was the senior author of the “Fact, Fiction, or Alternative Fact?” commentary.
Work is underway to elucidate biological mechanisms. Some groups have implicated the Fc region of antibodies. Another group has reported EGFR and MDM2/MDM4 amplifications in patients with HPD, Dr. Subbiah and colleagues noted.
Other “proposed contributing pathological mechanisms include modulation of tumor immune microenvironment through macrophages and regulatory T cells as well as activation of oncogenic signaling pathways,” noted Dr. Sehgal.
Both groups of authors emphasize the urgent need for prospective studies.
It is imperative to confirm underlying biology, predict which patients are at risk, and identify therapeutic directions for patients who experience HPD, Dr. Subbiah said.
The main challenge is defining HPD, he added. Definitions that have been proposed include tumor growth at least two times greater than in control persons, a 15% increase in tumor burden in a set period, and disease progression of 50% from the first evaluation before treatment, he said.
The recent meta-analysis by Hyo Jung Park, MD, PhD, and colleagues, which Dr. Sehgal addressed in his invited commentary, highlights the many approaches used for defining HPD.
Depending on the definition used, the incidence of HPD across 24 studies involving more than 3,100 patients ranged from 5.9% to 43.1%.
“Hyperprogressive disease could be overestimated or underestimated based on current assessment,” Dr. Park and colleagues concluded. They highlighted the importance of “establishing uniform and clinically relevant criteria based on currently available evidence.”
Steps for solving the HPD mystery
“I think we need to come up with consensus criteria for an HPD definition. We need a unified definition,” Dr. Subbiah said. “We also need to design prospective studies to prove or disprove the immunotherapy-HPD association.”
Prospective registries with independent review of patients with suspected immunotherapy-related HPD would be useful for assessing the true incidence and the biology of HPD among patients undergoing immunotherapy, he suggested.
“We need to know the immunologic signals of HPD. This can give us an idea if patients can be prospectively identified for being at risk,” he said. “We also need to know what to do if they are at risk.”
Dr. Sehgal also called for consensus on an HPD definition, with input from a multidisciplinary group that includes “colleagues from radiology, medical oncology, radiation oncology. Getting expertise from different disciplines would be helpful,” he said.
Dr. Park and colleagues suggested several key requirements for an optimal HP definition, such as the inclusion of multiple variables for measuring tumor growth acceleration, “sufficiently quantitative” criteria for determining time to failure, and establishment of a standardized measure of tumor growth acceleration.
The agreed-upon definition of HPD could be applied to patients in a prospective registry and to existing trial data, Dr. Sehgal said.
“Eventually, the goal of this exercise is to [determine] how we can help our patients the best, having a biomarker that can at least inform us in terms of being aware and being proactive in terms of looking for this ... so that interventions can be brought on earlier,” he said.
“If we know what may be a biological mechanism, we can design trials that are designed to look at how to overcome that HPD,” he said.
Dr. Sehgal said he believes HPD is triggered in some way by treatment, including immunotherapy, chemotherapy, and targeted therapy, but perhaps in different ways for each.
He estimated the true incidence of immunotherapy-related HPD will be in the 9%-10% range.
“This is a substantial number of patients, so it’s important that we try to understand this phenomenon, using, again, uniform criteria,” he said.
Current treatment decision-making
Until more is known, Dr. Sehgal said he considers the potential risk factors when treating patients with immunotherapy.
For example, the presence of MDM2 or MDM4 amplification on a genomic profile may factor into his treatment decision-making when it comes to using immunotherapy or immunotherapy in combination with chemotherapy, he said.
“Is that the only factor that is going to make me choose one thing or another? No,” Dr. Sehgal said. However, he said it would make him more “proactive in making sure the patient is doing clinically okay” and in determining when to obtain on-treatment imaging studies.
Dr. Subbiah emphasized the relative benefit of immunotherapy, noting that survival with chemotherapy for many difficult-to-treat cancers in the relapsed/refractory metastatic setting is less than 2 years.
Immunotherapy with checkpoint inhibitors has allowed some of these patients to live longer (with survival reported to be more than 10 years for patients with metastatic melanoma).
“Immunotherapy has been a game changer; it has been transformative in the lives of these patients,” Dr. Subbiah said. “So unless there is any other contraindication, the benefit of receiving immunotherapy for an approved indication far outweighs the risk of HPD.”
A version of this article first appeared on Medscape.com.
Immunotherapy with checkpoint inhibitors has ushered in a new era of cancer therapy, with some patients showing dramatic responses and significantly better outcomes than with other therapies across many cancer types. But some patients do worse, sometimes much worse.
A subset of patients who undergo immunotherapy experience unexpected, rapid disease progression, with a dramatic acceleration of disease trajectory. They also have a shorter progression-free survival and overall survival than would have been expected.
This has been described as hyperprogression and has been termed “hyperprogressive disease” (HPD). It has been seen in a variety of cancers; the incidence ranges from 4% to 29% in the studies reported to date.
There has been some debate over whether this is a real phenomenon or whether it is part of the natural course of disease.
HPD is a “provocative phenomenon,” wrote the authors of a recent commentary entitled “Hyperprogression and Immunotherapy: Fact, Fiction, or Alternative Fact?”
“This phenomenon has polarized oncologists who debate that this could still reflect the natural history of the disease,” said the author of another commentary.
But the tide is now turning toward acceptance of HPD, said Kartik Sehgal, MD, an oncologist at Dana-Farber Cancer Institute and Harvard University, both in Boston.
“With publication of multiple clinical reports of different cancer types worldwide, hyperprogression is now accepted by most oncologists to be a true phenomenon rather than natural progression of disease,” Dr. Sehgal said.
He authored an invited commentary in JAMA Network Openabout one of the latest meta-analyses (JAMA Netw Open. 2021;4[3]:e211136) to investigate HPD during immunotherapy. One of the biggest issues is that the studies that have reported on HPD have been retrospective, with a lack of comparator groups and a lack of a standardized definition of hyperprogression. Dr. Sehgal emphasized the need to study hyperprogression in well-designed prospective studies.
Existing data on HPD
HPD was described as “a new pattern of progression” seen in patients undergoing immune checkpoint inhibitor therapy in a 2017 article published in Clinical Cancer Research. Authors Stephane Champiat, MD, PhD, of Institut Gustave Roussy, Universite Paris Saclay, Villejuif, France, and colleagues cited “anecdotal occurrences” of HPD among patients in phase 1 trials of anti–PD-1/PD-L1 agents.
In that study, HPD was defined by tumor growth rate ratio. The incidence was 9% among 213 patients.
The findings raised concerns about treating elderly patients with anti–PD-1/PD-L1 monotherapy, according to the authors, who called for further study.
That same year, Roberto Ferrara, MD, and colleagues from the Insitut Gustave Roussy reported additional data indicating an incidence of HPD of 16% among 333 patients with non–small cell lung cancer who underwent immunotherapy at eight centers from 2012 to 2017. The findings, which were presented at the 2017 World Conference on Lung Cancer and reported at the time by this news organization, also showed that the incidence of HPD was higher with immunotherapy than with single-agent chemotherapy (5%).
Median overall survival (OS) was just 3.4 months among those with HPD, compared with 13 months in the overall study population – worse, even, than the median 5.4-month OS observed among patients with progressive disease who received immunotherapy.
In the wake of these findings, numerous researchers have attempted to better define HPD, its incidence, and patient factors associated with developing HPD while undergoing immunotherapy.
However, there is little so far to show for those efforts, Vivek Subbiah, MD, of the University of Texas MD Anderson Cancer Center, Houston, said in an interview.
“Many questions remain to be answered,” said Dr. Subbiah, clinical medical director of the Clinical Center for Targeted Therapy in the division of cancer medicine at MD Anderson. He was the senior author of the “Fact, Fiction, or Alternative Fact?” commentary.
Work is underway to elucidate biological mechanisms. Some groups have implicated the Fc region of antibodies. Another group has reported EGFR and MDM2/MDM4 amplifications in patients with HPD, Dr. Subbiah and colleagues noted.
Other “proposed contributing pathological mechanisms include modulation of tumor immune microenvironment through macrophages and regulatory T cells as well as activation of oncogenic signaling pathways,” noted Dr. Sehgal.
Both groups of authors emphasize the urgent need for prospective studies.
It is imperative to confirm underlying biology, predict which patients are at risk, and identify therapeutic directions for patients who experience HPD, Dr. Subbiah said.
The main challenge is defining HPD, he added. Definitions that have been proposed include tumor growth at least two times greater than in control persons, a 15% increase in tumor burden in a set period, and disease progression of 50% from the first evaluation before treatment, he said.
The recent meta-analysis by Hyo Jung Park, MD, PhD, and colleagues, which Dr. Sehgal addressed in his invited commentary, highlights the many approaches used for defining HPD.
Depending on the definition used, the incidence of HPD across 24 studies involving more than 3,100 patients ranged from 5.9% to 43.1%.
“Hyperprogressive disease could be overestimated or underestimated based on current assessment,” Dr. Park and colleagues concluded. They highlighted the importance of “establishing uniform and clinically relevant criteria based on currently available evidence.”
Steps for solving the HPD mystery
“I think we need to come up with consensus criteria for an HPD definition. We need a unified definition,” Dr. Subbiah said. “We also need to design prospective studies to prove or disprove the immunotherapy-HPD association.”
Prospective registries with independent review of patients with suspected immunotherapy-related HPD would be useful for assessing the true incidence and the biology of HPD among patients undergoing immunotherapy, he suggested.
“We need to know the immunologic signals of HPD. This can give us an idea if patients can be prospectively identified for being at risk,” he said. “We also need to know what to do if they are at risk.”
Dr. Sehgal also called for consensus on an HPD definition, with input from a multidisciplinary group that includes “colleagues from radiology, medical oncology, radiation oncology. Getting expertise from different disciplines would be helpful,” he said.
Dr. Park and colleagues suggested several key requirements for an optimal HP definition, such as the inclusion of multiple variables for measuring tumor growth acceleration, “sufficiently quantitative” criteria for determining time to failure, and establishment of a standardized measure of tumor growth acceleration.
The agreed-upon definition of HPD could be applied to patients in a prospective registry and to existing trial data, Dr. Sehgal said.
“Eventually, the goal of this exercise is to [determine] how we can help our patients the best, having a biomarker that can at least inform us in terms of being aware and being proactive in terms of looking for this ... so that interventions can be brought on earlier,” he said.
“If we know what may be a biological mechanism, we can design trials that are designed to look at how to overcome that HPD,” he said.
Dr. Sehgal said he believes HPD is triggered in some way by treatment, including immunotherapy, chemotherapy, and targeted therapy, but perhaps in different ways for each.
He estimated the true incidence of immunotherapy-related HPD will be in the 9%-10% range.
“This is a substantial number of patients, so it’s important that we try to understand this phenomenon, using, again, uniform criteria,” he said.
Current treatment decision-making
Until more is known, Dr. Sehgal said he considers the potential risk factors when treating patients with immunotherapy.
For example, the presence of MDM2 or MDM4 amplification on a genomic profile may factor into his treatment decision-making when it comes to using immunotherapy or immunotherapy in combination with chemotherapy, he said.
“Is that the only factor that is going to make me choose one thing or another? No,” Dr. Sehgal said. However, he said it would make him more “proactive in making sure the patient is doing clinically okay” and in determining when to obtain on-treatment imaging studies.
Dr. Subbiah emphasized the relative benefit of immunotherapy, noting that survival with chemotherapy for many difficult-to-treat cancers in the relapsed/refractory metastatic setting is less than 2 years.
Immunotherapy with checkpoint inhibitors has allowed some of these patients to live longer (with survival reported to be more than 10 years for patients with metastatic melanoma).
“Immunotherapy has been a game changer; it has been transformative in the lives of these patients,” Dr. Subbiah said. “So unless there is any other contraindication, the benefit of receiving immunotherapy for an approved indication far outweighs the risk of HPD.”
A version of this article first appeared on Medscape.com.
FDA panel votes against 2 cancer indications but backs 4 of 6
Federal advisers have supported the efforts of pharmaceutical companies in four of six cases in which these firms are fighting to maintain cancer indications for approved drugs. The advisers voted against the companies in two cases.
The staff of the Food and Drug Administration will now consider these votes as they decide what to do regarding the six cases of what they have termed “dangling” accelerated approvals.
“One of the reasons I think we’re convening today is to prevent these accelerated approvals from dangling ad infinitum,” commented one of the members of the advisory panel.
In these cases, companies have been unable to prove the expected benefits that led the FDA to grant accelerated approvals for these indications.
These accelerated approvals, which are often based on surrogate endpoints, such as overall response rates, are granted on the condition that further findings show a clinical benefit – such as in progression-free survival or overall survival – in larger trials.
The FDA tasked its Oncologic Drugs Advisory Committee (ODAC) with conducting the review of the six accelerated approvals for cancer indications at a 3-day meeting (April 27-29).
These reviews were only for specific cancer indications and will not lead to the removal of drugs from the market. These drugs have already been approved for several cancer indications. For example, one of the drugs that was reviewed, pembrolizumab (Keytruda), is approved in the United States for 28 indications.
The FDA is facing growing pains in its efforts to manage the rapidly changing landscape for these immune checkpoint inhibitors. This field of medicine has experienced an “unprecedented level of drug development” in recent years, FDA officials said in briefing materials, owing in part to the agency’s willingness to accept surrogate markers for accelerated approvals. Although some companies have struggled with these, others have built strong cases for the use of their checkpoint inhibitors for these indications.
The ODAC panelists, for example, noted the emergence of nivolumab (Opdivo) as an option for patients with gastric cancer as a reason for seeking to withdraw an indication for pembrolizumab (Keytruda) for this disease.
Just weeks before the meeting, on April 16, the FDA approved nivolumab plus chemotherapy as a first-line treatment for advanced or metastatic gastric cancer, gastroesophageal junction cancer, and esophageal adenocarcinoma. This was a full approval based on data showing an overall survival benefit from a phase 3 trial.
Votes by indication
On April 29, the last day of the meeting, the ODAC panel voted 6-2 against maintaining pembrolizumab’s indication as monotherapy for an advanced form of gastric cancer. This was an accelerated approval (granted in 2017) that was based on overall response rates from an open-label trial.
That last day of the meeting also saw another negative vote. On April 29, the ODAC panel voted 5-4 against maintaining an indication for nivolumab in patients with hepatocellular carcinoma (HCC) who were previously treated with sorafenib (Nexavar).
This accelerated approval for nivolumab was granted in 2017. The FDA said it had requested ODAC’s feedback on this indication because of the recent full approval of another checkpoint inhibitor for HCC, atezolizumab (Tecentriq), in combination with bevacizumab (Avastin) for patients with unresectable or metastatic diseases who have not received prior systemic therapy. This full approval (in May 2020) was based on an overall survival benefit.
There was one last vote on the third day of the meeting, and it was positive. The ODAC panel voted 8-0 in favor of maintaining the indication for the use of pembrolizumab as monotherapy for patients with HCC who have previously been treated with sorafenib.
The FDA altered the composition of the ODAC panel during the week, adding members in some cases who had expertise in particular cancers. That led to different totals for the week’s ODAC votes, as shown in the tallies summarized below.
On the first day of the meeting (April 27), the ODAC panel voted 7-2 in favor of maintaining a breast cancer indication for atezolizumab (Tecentriq). This covered use of the immunotherapy in combination with nab-paclitaxel for patients with unresectable locally advanced or metastatic triple-negative breast cancer whose tumors express PD-L1.
The second day of the meeting (April 28) also saw two positive votes. The ODAC panel voted 10-1 for maintaining the indication for atezolizumab for the first-line treatment of cisplatin-ineligible patients with advanced/metastatic urothelial carcinoma, pending final overall survival results from the IMvigor130 trial. The panel also voted 5-3 for maintaining the indication for pembrolizumab in patients with locally advanced or metastatic urothelial carcinoma who are not eligible for cisplatin-containing chemotherapy and whose tumors express PD-L1.
The FDA is not bound to follow the voting and recommendations of its advisory panels, but it usually does so.
Managing shifts in treatment
In both of the cases in which ODAC voted against maintaining indications, Richard Pazdur, MD, the FDA’s top regulator for cancer medicines, jumped into the debate. Dr. Pazdur countered arguments put forward by representatives of the manufacturers as they sought to maintain indications for their drugs.
Merck officials and representatives argued for pembrolizumab, saying that maintaining the gastric cancer indication might help patients whose disease has progressed despite earlier treatment.
Dr. Pazdur emphasized that the agency would help Merck and physicians to have access to pembrolizumab for these patients even if this one indication were to be withdrawn. But Dr. Pazdur and ODAC members also noted the recent shift in the landscape for gastric cancer, with the recent approval of a new indication for nivolumab.
“I want to emphasize to the patient community out there [that] we firmly believe in the role of checkpoint inhibitors in this disease,” Dr. Pazdur said during the discussion of the indication for pembrolizumab for gastric cancer. “We have to be cognizant of what is the appropriate setting for that, and it currently is in the first line.”
Dr. Pazdur noted that two studies had failed to confirm the expected benefit from pembrolizumab for patients with more advanced disease. Still, if “small numbers” of patients with advanced disease wanted access to Merck’s drug, the FDA and the company could accommodate them. The FDA could delay the removal of the gastric indication to allow patients to continue receiving it. The FDA also could work with physicians on other routes to provide the medicine, such as through single-patient investigational new drug applications or an expanded access program.
“Or Merck can alternatively give the drug gratis to patients,” Dr. Pazdur said.
#ProjectFacilitate for expanded access
One of Merck’s speakers at the ODAC meeting, Peter Enzinger, MD, of the Dana-Farber Cancer Institute, Boston, objected to Dr. Pazdur’s plan.
A loss of the gastric indication for pembrolizumab would result in patients with advanced cancer missing out on a chance to try this therapy. Some patients will not have had a chance to try a checkpoint inhibitor earlier in their treatment, and a loss of the indication would cost them that opportunity, he said.
“An expanded-access program sounds very nice, but the reality is that our patients are incredibly sick and that weeks matter,” Dr. Enzinger said, citing administrative hurdles as a barrier to treatment.
“Our patients just don’t have the time for that, and therefore I don’t think an expanded access program is the way to go,” Dr. Enzinger said.
Dr. Pazdur responded to these objections by highlighting an initiative called Project Facilitate at the FDA’s Oncology Center for Excellence. During the meeting, Dr. Pazdur’s division used its @FDAOncology Twitter handle to draw attention to this project.
ODAC panelist Diane Reidy-Lagunes, MD, of Memorial Sloan Kettering Cancer Center, New York, said she had struggled with this vote. She was one of the two panelists to vote in favor of keeping the indication.
“This is also incredibly hard for me. I actually changed it at the last minute,” she said of her vote.
But Dr. Reidy-Lagunes said she was concerned that some patients with advanced disease might not be able to get a checkpoint inhibitor.
“With disparities in healthcare and differences in the way that patients are treated throughout our country, I was nervous that they may not be able to get treated,” she said, noting that she shared her fellow panelists’ doubts about use of pembrolizumab as third-line treatment, owing to negative results in trials.
ODAC member David Mitchell, who served as a consumer representative, also said he found the vote on the gastric indication for pembrolizumab to be a difficult decision.
“As a patient with incurable cancer who’s now being given all three major classes of drugs to treat my disease in combination, these issues really cut close to home,” Mr. Mitchell said.
He said the expectation that the FDA’s expanded access program could help patients with advanced disease try pembrolizumab helped him decide to vote with the 6-2 majority against maintaining this gastric cancer approval.
His vote was based on “the changing treatment landscape.” There is general agreement that the patients in question should receive checkpoint inhibitors as first-line treatment, not third-line treatment, Mr. Mitchell said. The FDA should delay a withdrawal of the approval for pembrolizumab in this case and should allow a transition for those who missed out on treatment with a checkpoint inhibitor earlier in the disease course, he suggested.
“To protect the safety and well-being of patients, we have to base decisions on data,” Mr. Mitchell said. “The data don’t support maintaining the indication” for pembrolizumab.
Close split on nivolumab
In contrast to the 6-2 vote against maintaining the pembrolizumab indication, the ODAC panel split more closely, 5-4, on the question of maintaining an indication for the use as monotherapy of nivolumab in HCC.
ODAC panelist Philip C. Hoffman, MD, of the University of Chicago was among those who supported keeping the indication.
“There’s still an unmet need for second-line immunotherapy because there will always be some patients who are poor candidates for bevacizumab or who are not tolerating or responding to sorafenib,” he said.
ODAC panelist Mark A. Lewis, MD, of Intermountain Healthcare, Salt Lake City, said he voted “no” in part because he doubted that Bristol-Myers Squibb would be able to soon produce data for nivolumab that was needed to support this indication.
A version of this article first appeared on Medscape.com.
Federal advisers have supported the efforts of pharmaceutical companies in four of six cases in which these firms are fighting to maintain cancer indications for approved drugs. The advisers voted against the companies in two cases.
The staff of the Food and Drug Administration will now consider these votes as they decide what to do regarding the six cases of what they have termed “dangling” accelerated approvals.
“One of the reasons I think we’re convening today is to prevent these accelerated approvals from dangling ad infinitum,” commented one of the members of the advisory panel.
In these cases, companies have been unable to prove the expected benefits that led the FDA to grant accelerated approvals for these indications.
These accelerated approvals, which are often based on surrogate endpoints, such as overall response rates, are granted on the condition that further findings show a clinical benefit – such as in progression-free survival or overall survival – in larger trials.
The FDA tasked its Oncologic Drugs Advisory Committee (ODAC) with conducting the review of the six accelerated approvals for cancer indications at a 3-day meeting (April 27-29).
These reviews were only for specific cancer indications and will not lead to the removal of drugs from the market. These drugs have already been approved for several cancer indications. For example, one of the drugs that was reviewed, pembrolizumab (Keytruda), is approved in the United States for 28 indications.
The FDA is facing growing pains in its efforts to manage the rapidly changing landscape for these immune checkpoint inhibitors. This field of medicine has experienced an “unprecedented level of drug development” in recent years, FDA officials said in briefing materials, owing in part to the agency’s willingness to accept surrogate markers for accelerated approvals. Although some companies have struggled with these, others have built strong cases for the use of their checkpoint inhibitors for these indications.
The ODAC panelists, for example, noted the emergence of nivolumab (Opdivo) as an option for patients with gastric cancer as a reason for seeking to withdraw an indication for pembrolizumab (Keytruda) for this disease.
Just weeks before the meeting, on April 16, the FDA approved nivolumab plus chemotherapy as a first-line treatment for advanced or metastatic gastric cancer, gastroesophageal junction cancer, and esophageal adenocarcinoma. This was a full approval based on data showing an overall survival benefit from a phase 3 trial.
Votes by indication
On April 29, the last day of the meeting, the ODAC panel voted 6-2 against maintaining pembrolizumab’s indication as monotherapy for an advanced form of gastric cancer. This was an accelerated approval (granted in 2017) that was based on overall response rates from an open-label trial.
That last day of the meeting also saw another negative vote. On April 29, the ODAC panel voted 5-4 against maintaining an indication for nivolumab in patients with hepatocellular carcinoma (HCC) who were previously treated with sorafenib (Nexavar).
This accelerated approval for nivolumab was granted in 2017. The FDA said it had requested ODAC’s feedback on this indication because of the recent full approval of another checkpoint inhibitor for HCC, atezolizumab (Tecentriq), in combination with bevacizumab (Avastin) for patients with unresectable or metastatic diseases who have not received prior systemic therapy. This full approval (in May 2020) was based on an overall survival benefit.
There was one last vote on the third day of the meeting, and it was positive. The ODAC panel voted 8-0 in favor of maintaining the indication for the use of pembrolizumab as monotherapy for patients with HCC who have previously been treated with sorafenib.
The FDA altered the composition of the ODAC panel during the week, adding members in some cases who had expertise in particular cancers. That led to different totals for the week’s ODAC votes, as shown in the tallies summarized below.
On the first day of the meeting (April 27), the ODAC panel voted 7-2 in favor of maintaining a breast cancer indication for atezolizumab (Tecentriq). This covered use of the immunotherapy in combination with nab-paclitaxel for patients with unresectable locally advanced or metastatic triple-negative breast cancer whose tumors express PD-L1.
The second day of the meeting (April 28) also saw two positive votes. The ODAC panel voted 10-1 for maintaining the indication for atezolizumab for the first-line treatment of cisplatin-ineligible patients with advanced/metastatic urothelial carcinoma, pending final overall survival results from the IMvigor130 trial. The panel also voted 5-3 for maintaining the indication for pembrolizumab in patients with locally advanced or metastatic urothelial carcinoma who are not eligible for cisplatin-containing chemotherapy and whose tumors express PD-L1.
The FDA is not bound to follow the voting and recommendations of its advisory panels, but it usually does so.
Managing shifts in treatment
In both of the cases in which ODAC voted against maintaining indications, Richard Pazdur, MD, the FDA’s top regulator for cancer medicines, jumped into the debate. Dr. Pazdur countered arguments put forward by representatives of the manufacturers as they sought to maintain indications for their drugs.
Merck officials and representatives argued for pembrolizumab, saying that maintaining the gastric cancer indication might help patients whose disease has progressed despite earlier treatment.
Dr. Pazdur emphasized that the agency would help Merck and physicians to have access to pembrolizumab for these patients even if this one indication were to be withdrawn. But Dr. Pazdur and ODAC members also noted the recent shift in the landscape for gastric cancer, with the recent approval of a new indication for nivolumab.
“I want to emphasize to the patient community out there [that] we firmly believe in the role of checkpoint inhibitors in this disease,” Dr. Pazdur said during the discussion of the indication for pembrolizumab for gastric cancer. “We have to be cognizant of what is the appropriate setting for that, and it currently is in the first line.”
Dr. Pazdur noted that two studies had failed to confirm the expected benefit from pembrolizumab for patients with more advanced disease. Still, if “small numbers” of patients with advanced disease wanted access to Merck’s drug, the FDA and the company could accommodate them. The FDA could delay the removal of the gastric indication to allow patients to continue receiving it. The FDA also could work with physicians on other routes to provide the medicine, such as through single-patient investigational new drug applications or an expanded access program.
“Or Merck can alternatively give the drug gratis to patients,” Dr. Pazdur said.
#ProjectFacilitate for expanded access
One of Merck’s speakers at the ODAC meeting, Peter Enzinger, MD, of the Dana-Farber Cancer Institute, Boston, objected to Dr. Pazdur’s plan.
A loss of the gastric indication for pembrolizumab would result in patients with advanced cancer missing out on a chance to try this therapy. Some patients will not have had a chance to try a checkpoint inhibitor earlier in their treatment, and a loss of the indication would cost them that opportunity, he said.
“An expanded-access program sounds very nice, but the reality is that our patients are incredibly sick and that weeks matter,” Dr. Enzinger said, citing administrative hurdles as a barrier to treatment.
“Our patients just don’t have the time for that, and therefore I don’t think an expanded access program is the way to go,” Dr. Enzinger said.
Dr. Pazdur responded to these objections by highlighting an initiative called Project Facilitate at the FDA’s Oncology Center for Excellence. During the meeting, Dr. Pazdur’s division used its @FDAOncology Twitter handle to draw attention to this project.
ODAC panelist Diane Reidy-Lagunes, MD, of Memorial Sloan Kettering Cancer Center, New York, said she had struggled with this vote. She was one of the two panelists to vote in favor of keeping the indication.
“This is also incredibly hard for me. I actually changed it at the last minute,” she said of her vote.
But Dr. Reidy-Lagunes said she was concerned that some patients with advanced disease might not be able to get a checkpoint inhibitor.
“With disparities in healthcare and differences in the way that patients are treated throughout our country, I was nervous that they may not be able to get treated,” she said, noting that she shared her fellow panelists’ doubts about use of pembrolizumab as third-line treatment, owing to negative results in trials.
ODAC member David Mitchell, who served as a consumer representative, also said he found the vote on the gastric indication for pembrolizumab to be a difficult decision.
“As a patient with incurable cancer who’s now being given all three major classes of drugs to treat my disease in combination, these issues really cut close to home,” Mr. Mitchell said.
He said the expectation that the FDA’s expanded access program could help patients with advanced disease try pembrolizumab helped him decide to vote with the 6-2 majority against maintaining this gastric cancer approval.
His vote was based on “the changing treatment landscape.” There is general agreement that the patients in question should receive checkpoint inhibitors as first-line treatment, not third-line treatment, Mr. Mitchell said. The FDA should delay a withdrawal of the approval for pembrolizumab in this case and should allow a transition for those who missed out on treatment with a checkpoint inhibitor earlier in the disease course, he suggested.
“To protect the safety and well-being of patients, we have to base decisions on data,” Mr. Mitchell said. “The data don’t support maintaining the indication” for pembrolizumab.
Close split on nivolumab
In contrast to the 6-2 vote against maintaining the pembrolizumab indication, the ODAC panel split more closely, 5-4, on the question of maintaining an indication for the use as monotherapy of nivolumab in HCC.
ODAC panelist Philip C. Hoffman, MD, of the University of Chicago was among those who supported keeping the indication.
“There’s still an unmet need for second-line immunotherapy because there will always be some patients who are poor candidates for bevacizumab or who are not tolerating or responding to sorafenib,” he said.
ODAC panelist Mark A. Lewis, MD, of Intermountain Healthcare, Salt Lake City, said he voted “no” in part because he doubted that Bristol-Myers Squibb would be able to soon produce data for nivolumab that was needed to support this indication.
A version of this article first appeared on Medscape.com.
Federal advisers have supported the efforts of pharmaceutical companies in four of six cases in which these firms are fighting to maintain cancer indications for approved drugs. The advisers voted against the companies in two cases.
The staff of the Food and Drug Administration will now consider these votes as they decide what to do regarding the six cases of what they have termed “dangling” accelerated approvals.
“One of the reasons I think we’re convening today is to prevent these accelerated approvals from dangling ad infinitum,” commented one of the members of the advisory panel.
In these cases, companies have been unable to prove the expected benefits that led the FDA to grant accelerated approvals for these indications.
These accelerated approvals, which are often based on surrogate endpoints, such as overall response rates, are granted on the condition that further findings show a clinical benefit – such as in progression-free survival or overall survival – in larger trials.
The FDA tasked its Oncologic Drugs Advisory Committee (ODAC) with conducting the review of the six accelerated approvals for cancer indications at a 3-day meeting (April 27-29).
These reviews were only for specific cancer indications and will not lead to the removal of drugs from the market. These drugs have already been approved for several cancer indications. For example, one of the drugs that was reviewed, pembrolizumab (Keytruda), is approved in the United States for 28 indications.
The FDA is facing growing pains in its efforts to manage the rapidly changing landscape for these immune checkpoint inhibitors. This field of medicine has experienced an “unprecedented level of drug development” in recent years, FDA officials said in briefing materials, owing in part to the agency’s willingness to accept surrogate markers for accelerated approvals. Although some companies have struggled with these, others have built strong cases for the use of their checkpoint inhibitors for these indications.
The ODAC panelists, for example, noted the emergence of nivolumab (Opdivo) as an option for patients with gastric cancer as a reason for seeking to withdraw an indication for pembrolizumab (Keytruda) for this disease.
Just weeks before the meeting, on April 16, the FDA approved nivolumab plus chemotherapy as a first-line treatment for advanced or metastatic gastric cancer, gastroesophageal junction cancer, and esophageal adenocarcinoma. This was a full approval based on data showing an overall survival benefit from a phase 3 trial.
Votes by indication
On April 29, the last day of the meeting, the ODAC panel voted 6-2 against maintaining pembrolizumab’s indication as monotherapy for an advanced form of gastric cancer. This was an accelerated approval (granted in 2017) that was based on overall response rates from an open-label trial.
That last day of the meeting also saw another negative vote. On April 29, the ODAC panel voted 5-4 against maintaining an indication for nivolumab in patients with hepatocellular carcinoma (HCC) who were previously treated with sorafenib (Nexavar).
This accelerated approval for nivolumab was granted in 2017. The FDA said it had requested ODAC’s feedback on this indication because of the recent full approval of another checkpoint inhibitor for HCC, atezolizumab (Tecentriq), in combination with bevacizumab (Avastin) for patients with unresectable or metastatic diseases who have not received prior systemic therapy. This full approval (in May 2020) was based on an overall survival benefit.
There was one last vote on the third day of the meeting, and it was positive. The ODAC panel voted 8-0 in favor of maintaining the indication for the use of pembrolizumab as monotherapy for patients with HCC who have previously been treated with sorafenib.
The FDA altered the composition of the ODAC panel during the week, adding members in some cases who had expertise in particular cancers. That led to different totals for the week’s ODAC votes, as shown in the tallies summarized below.
On the first day of the meeting (April 27), the ODAC panel voted 7-2 in favor of maintaining a breast cancer indication for atezolizumab (Tecentriq). This covered use of the immunotherapy in combination with nab-paclitaxel for patients with unresectable locally advanced or metastatic triple-negative breast cancer whose tumors express PD-L1.
The second day of the meeting (April 28) also saw two positive votes. The ODAC panel voted 10-1 for maintaining the indication for atezolizumab for the first-line treatment of cisplatin-ineligible patients with advanced/metastatic urothelial carcinoma, pending final overall survival results from the IMvigor130 trial. The panel also voted 5-3 for maintaining the indication for pembrolizumab in patients with locally advanced or metastatic urothelial carcinoma who are not eligible for cisplatin-containing chemotherapy and whose tumors express PD-L1.
The FDA is not bound to follow the voting and recommendations of its advisory panels, but it usually does so.
Managing shifts in treatment
In both of the cases in which ODAC voted against maintaining indications, Richard Pazdur, MD, the FDA’s top regulator for cancer medicines, jumped into the debate. Dr. Pazdur countered arguments put forward by representatives of the manufacturers as they sought to maintain indications for their drugs.
Merck officials and representatives argued for pembrolizumab, saying that maintaining the gastric cancer indication might help patients whose disease has progressed despite earlier treatment.
Dr. Pazdur emphasized that the agency would help Merck and physicians to have access to pembrolizumab for these patients even if this one indication were to be withdrawn. But Dr. Pazdur and ODAC members also noted the recent shift in the landscape for gastric cancer, with the recent approval of a new indication for nivolumab.
“I want to emphasize to the patient community out there [that] we firmly believe in the role of checkpoint inhibitors in this disease,” Dr. Pazdur said during the discussion of the indication for pembrolizumab for gastric cancer. “We have to be cognizant of what is the appropriate setting for that, and it currently is in the first line.”
Dr. Pazdur noted that two studies had failed to confirm the expected benefit from pembrolizumab for patients with more advanced disease. Still, if “small numbers” of patients with advanced disease wanted access to Merck’s drug, the FDA and the company could accommodate them. The FDA could delay the removal of the gastric indication to allow patients to continue receiving it. The FDA also could work with physicians on other routes to provide the medicine, such as through single-patient investigational new drug applications or an expanded access program.
“Or Merck can alternatively give the drug gratis to patients,” Dr. Pazdur said.
#ProjectFacilitate for expanded access
One of Merck’s speakers at the ODAC meeting, Peter Enzinger, MD, of the Dana-Farber Cancer Institute, Boston, objected to Dr. Pazdur’s plan.
A loss of the gastric indication for pembrolizumab would result in patients with advanced cancer missing out on a chance to try this therapy. Some patients will not have had a chance to try a checkpoint inhibitor earlier in their treatment, and a loss of the indication would cost them that opportunity, he said.
“An expanded-access program sounds very nice, but the reality is that our patients are incredibly sick and that weeks matter,” Dr. Enzinger said, citing administrative hurdles as a barrier to treatment.
“Our patients just don’t have the time for that, and therefore I don’t think an expanded access program is the way to go,” Dr. Enzinger said.
Dr. Pazdur responded to these objections by highlighting an initiative called Project Facilitate at the FDA’s Oncology Center for Excellence. During the meeting, Dr. Pazdur’s division used its @FDAOncology Twitter handle to draw attention to this project.
ODAC panelist Diane Reidy-Lagunes, MD, of Memorial Sloan Kettering Cancer Center, New York, said she had struggled with this vote. She was one of the two panelists to vote in favor of keeping the indication.
“This is also incredibly hard for me. I actually changed it at the last minute,” she said of her vote.
But Dr. Reidy-Lagunes said she was concerned that some patients with advanced disease might not be able to get a checkpoint inhibitor.
“With disparities in healthcare and differences in the way that patients are treated throughout our country, I was nervous that they may not be able to get treated,” she said, noting that she shared her fellow panelists’ doubts about use of pembrolizumab as third-line treatment, owing to negative results in trials.
ODAC member David Mitchell, who served as a consumer representative, also said he found the vote on the gastric indication for pembrolizumab to be a difficult decision.
“As a patient with incurable cancer who’s now being given all three major classes of drugs to treat my disease in combination, these issues really cut close to home,” Mr. Mitchell said.
He said the expectation that the FDA’s expanded access program could help patients with advanced disease try pembrolizumab helped him decide to vote with the 6-2 majority against maintaining this gastric cancer approval.
His vote was based on “the changing treatment landscape.” There is general agreement that the patients in question should receive checkpoint inhibitors as first-line treatment, not third-line treatment, Mr. Mitchell said. The FDA should delay a withdrawal of the approval for pembrolizumab in this case and should allow a transition for those who missed out on treatment with a checkpoint inhibitor earlier in the disease course, he suggested.
“To protect the safety and well-being of patients, we have to base decisions on data,” Mr. Mitchell said. “The data don’t support maintaining the indication” for pembrolizumab.
Close split on nivolumab
In contrast to the 6-2 vote against maintaining the pembrolizumab indication, the ODAC panel split more closely, 5-4, on the question of maintaining an indication for the use as monotherapy of nivolumab in HCC.
ODAC panelist Philip C. Hoffman, MD, of the University of Chicago was among those who supported keeping the indication.
“There’s still an unmet need for second-line immunotherapy because there will always be some patients who are poor candidates for bevacizumab or who are not tolerating or responding to sorafenib,” he said.
ODAC panelist Mark A. Lewis, MD, of Intermountain Healthcare, Salt Lake City, said he voted “no” in part because he doubted that Bristol-Myers Squibb would be able to soon produce data for nivolumab that was needed to support this indication.
A version of this article first appeared on Medscape.com.
Basal ganglia microcircuits offer clues to Parkinson’s symptoms
, according to a new study using a mouse model of disease.
Parkinson’s disease is characterized by a range of cognitive and motor symptoms, which appear at different disease stages. While recent research has pointed to specific neuronal subpopulations, or microcircuits, operating in the basal ganglia, researchers lacked a clear understanding of how they might correspond with specific symptom domains.
In a study published online March 15 in Nature Neuroscience, lead author Varoth Lilascharoen, PhD, of the University of California, San Diego, and colleagues reported that two different neuronal subpopulations within the external globus pallidus, an important nucleus within the basal ganglia, are associated, respectively, with movement and with reversal learning (having to adapt to a reward pattern that is the reverse of a previous pattern). This is the first time, the investigators said, that the contributions of specific microcircuits in the basal ganglia have been linked to different behaviors.
Using electrophysiology, viral tracing, and other approaches, Dr. Lilascharoen and colleagues demonstrated that two microcircuits or populations of parvalbumin-expressing neurons could be manipulated to exacerbate or alleviate the motor or cognitive deficits in the dopamine-depleted mice.
One of these microcircuits, made up of substantia nigra pars reticulata-projecting GPe-PV neurons, could be manipulated in ways that promoted or inhibited the mice’s movement. The other, which comprises parafascicular thalamus-projecting GPe-PV neurons, could be manipulated to affect reversal learning, the researchers found. Activation or inhibition of either circuit was not seen affecting function in the other.
The results shed light on the functional organization of the different basal ganglia nuclei at the circuit level, and suggest, the authors argued, that differences in how different neuronal subpopulations adapt to dopamine loss could explain some of the patterns of progression seen in Parkinson’s disease.
The findings “establish the differential contributions from two distinct GPe-PV microcircuits in specific Parkinsonian-like behaviors linked to early and late stages of the disease,” Dr. Lilascharoen and colleagues wrote in their analysis. “[F]urther elucidation of the detailed connectivity of GPe subpopulations to their downstream targets … is needed to fully define the function of each microcircuit and design better therapeutic strategies for the various behavioral impairments of Parkinson’s disease.”
Commenting on the research, Stefan Lang, MD, PhD, of the University of Calgary in Alberta said, “While Parkinson’s disease is often referred to as a movement disorder, it is well known that nonmotor symptoms, including cognitive and behavioral impairment, are common and debilitating. Impairment of basal ganglia function is known to contribute to these different symptom domains, though the specific circuits have never been elucidated. [Dr.] Lilascharoen et al. tease apart specific basal ganglia circuits associated with motor and behavioral symptoms, thereby providing evidence that distinct microcircuits might contribute to unique behaviours. As technological advances in neuromodulatory therapies continue to improve the spatial and temporal resolution of stimulation, future treatments may allow for specific targeting of behavioral impairment symptoms in Parkinson’s disease.”
Dr. Lilascharoen and Dr. Lang did not report outside funding or conflicts of interest.
, according to a new study using a mouse model of disease.
Parkinson’s disease is characterized by a range of cognitive and motor symptoms, which appear at different disease stages. While recent research has pointed to specific neuronal subpopulations, or microcircuits, operating in the basal ganglia, researchers lacked a clear understanding of how they might correspond with specific symptom domains.
In a study published online March 15 in Nature Neuroscience, lead author Varoth Lilascharoen, PhD, of the University of California, San Diego, and colleagues reported that two different neuronal subpopulations within the external globus pallidus, an important nucleus within the basal ganglia, are associated, respectively, with movement and with reversal learning (having to adapt to a reward pattern that is the reverse of a previous pattern). This is the first time, the investigators said, that the contributions of specific microcircuits in the basal ganglia have been linked to different behaviors.
Using electrophysiology, viral tracing, and other approaches, Dr. Lilascharoen and colleagues demonstrated that two microcircuits or populations of parvalbumin-expressing neurons could be manipulated to exacerbate or alleviate the motor or cognitive deficits in the dopamine-depleted mice.
One of these microcircuits, made up of substantia nigra pars reticulata-projecting GPe-PV neurons, could be manipulated in ways that promoted or inhibited the mice’s movement. The other, which comprises parafascicular thalamus-projecting GPe-PV neurons, could be manipulated to affect reversal learning, the researchers found. Activation or inhibition of either circuit was not seen affecting function in the other.
The results shed light on the functional organization of the different basal ganglia nuclei at the circuit level, and suggest, the authors argued, that differences in how different neuronal subpopulations adapt to dopamine loss could explain some of the patterns of progression seen in Parkinson’s disease.
The findings “establish the differential contributions from two distinct GPe-PV microcircuits in specific Parkinsonian-like behaviors linked to early and late stages of the disease,” Dr. Lilascharoen and colleagues wrote in their analysis. “[F]urther elucidation of the detailed connectivity of GPe subpopulations to their downstream targets … is needed to fully define the function of each microcircuit and design better therapeutic strategies for the various behavioral impairments of Parkinson’s disease.”
Commenting on the research, Stefan Lang, MD, PhD, of the University of Calgary in Alberta said, “While Parkinson’s disease is often referred to as a movement disorder, it is well known that nonmotor symptoms, including cognitive and behavioral impairment, are common and debilitating. Impairment of basal ganglia function is known to contribute to these different symptom domains, though the specific circuits have never been elucidated. [Dr.] Lilascharoen et al. tease apart specific basal ganglia circuits associated with motor and behavioral symptoms, thereby providing evidence that distinct microcircuits might contribute to unique behaviours. As technological advances in neuromodulatory therapies continue to improve the spatial and temporal resolution of stimulation, future treatments may allow for specific targeting of behavioral impairment symptoms in Parkinson’s disease.”
Dr. Lilascharoen and Dr. Lang did not report outside funding or conflicts of interest.
, according to a new study using a mouse model of disease.
Parkinson’s disease is characterized by a range of cognitive and motor symptoms, which appear at different disease stages. While recent research has pointed to specific neuronal subpopulations, or microcircuits, operating in the basal ganglia, researchers lacked a clear understanding of how they might correspond with specific symptom domains.
In a study published online March 15 in Nature Neuroscience, lead author Varoth Lilascharoen, PhD, of the University of California, San Diego, and colleagues reported that two different neuronal subpopulations within the external globus pallidus, an important nucleus within the basal ganglia, are associated, respectively, with movement and with reversal learning (having to adapt to a reward pattern that is the reverse of a previous pattern). This is the first time, the investigators said, that the contributions of specific microcircuits in the basal ganglia have been linked to different behaviors.
Using electrophysiology, viral tracing, and other approaches, Dr. Lilascharoen and colleagues demonstrated that two microcircuits or populations of parvalbumin-expressing neurons could be manipulated to exacerbate or alleviate the motor or cognitive deficits in the dopamine-depleted mice.
One of these microcircuits, made up of substantia nigra pars reticulata-projecting GPe-PV neurons, could be manipulated in ways that promoted or inhibited the mice’s movement. The other, which comprises parafascicular thalamus-projecting GPe-PV neurons, could be manipulated to affect reversal learning, the researchers found. Activation or inhibition of either circuit was not seen affecting function in the other.
The results shed light on the functional organization of the different basal ganglia nuclei at the circuit level, and suggest, the authors argued, that differences in how different neuronal subpopulations adapt to dopamine loss could explain some of the patterns of progression seen in Parkinson’s disease.
The findings “establish the differential contributions from two distinct GPe-PV microcircuits in specific Parkinsonian-like behaviors linked to early and late stages of the disease,” Dr. Lilascharoen and colleagues wrote in their analysis. “[F]urther elucidation of the detailed connectivity of GPe subpopulations to their downstream targets … is needed to fully define the function of each microcircuit and design better therapeutic strategies for the various behavioral impairments of Parkinson’s disease.”
Commenting on the research, Stefan Lang, MD, PhD, of the University of Calgary in Alberta said, “While Parkinson’s disease is often referred to as a movement disorder, it is well known that nonmotor symptoms, including cognitive and behavioral impairment, are common and debilitating. Impairment of basal ganglia function is known to contribute to these different symptom domains, though the specific circuits have never been elucidated. [Dr.] Lilascharoen et al. tease apart specific basal ganglia circuits associated with motor and behavioral symptoms, thereby providing evidence that distinct microcircuits might contribute to unique behaviours. As technological advances in neuromodulatory therapies continue to improve the spatial and temporal resolution of stimulation, future treatments may allow for specific targeting of behavioral impairment symptoms in Parkinson’s disease.”
Dr. Lilascharoen and Dr. Lang did not report outside funding or conflicts of interest.
FROM NATURE NEUROSCIENCE
Adjuvant palbociclib fails in early breast cancer
Key clinical point: The addition of palbociclib to estrogen therapy for 1 year does not improve invasive disease-free survival (DFS) in high-risk patients with hormone-receptor–positive (HR+), human epidermal growth factor receptor (HER)-negative early breast cancer who had residual invasive disease after neoadjuvant chemotherapy.
Major finding: Palbociclib did not improve invasive DFS vs. placebo (stratified hazard ratio, 0.93; P = .525) after a median follow-up of 42.8 months. Incidences of grade 3-4 neutropenia and leukopenia were significantly higher in the palbociclib group. Eight fatal serious adverse events were reported.
Study details: A phase 3, double-blind, randomized PENELOPE-B study evaluated 1,250 high-risk patients with HR+, HER-negative early breast cancer who have residual invasive disease after neoadjuvant chemotherapy. Patients received estrogen therapy with either palbociclib or placebo.
Disclosures: The study was supported by Pfizer. The authors declared receiving consulting, honoraria, travel/accomodation expenses and research funding outside the study work. Some of the authors declared being employee of and/or stocks/ownership interests of various sources including Pfizer.
Source: Loibl S et al. J Clin Oncol. 2021 Apr 1. doi: 10.1200/JCO.20.03639.
Key clinical point: The addition of palbociclib to estrogen therapy for 1 year does not improve invasive disease-free survival (DFS) in high-risk patients with hormone-receptor–positive (HR+), human epidermal growth factor receptor (HER)-negative early breast cancer who had residual invasive disease after neoadjuvant chemotherapy.
Major finding: Palbociclib did not improve invasive DFS vs. placebo (stratified hazard ratio, 0.93; P = .525) after a median follow-up of 42.8 months. Incidences of grade 3-4 neutropenia and leukopenia were significantly higher in the palbociclib group. Eight fatal serious adverse events were reported.
Study details: A phase 3, double-blind, randomized PENELOPE-B study evaluated 1,250 high-risk patients with HR+, HER-negative early breast cancer who have residual invasive disease after neoadjuvant chemotherapy. Patients received estrogen therapy with either palbociclib or placebo.
Disclosures: The study was supported by Pfizer. The authors declared receiving consulting, honoraria, travel/accomodation expenses and research funding outside the study work. Some of the authors declared being employee of and/or stocks/ownership interests of various sources including Pfizer.
Source: Loibl S et al. J Clin Oncol. 2021 Apr 1. doi: 10.1200/JCO.20.03639.
Key clinical point: The addition of palbociclib to estrogen therapy for 1 year does not improve invasive disease-free survival (DFS) in high-risk patients with hormone-receptor–positive (HR+), human epidermal growth factor receptor (HER)-negative early breast cancer who had residual invasive disease after neoadjuvant chemotherapy.
Major finding: Palbociclib did not improve invasive DFS vs. placebo (stratified hazard ratio, 0.93; P = .525) after a median follow-up of 42.8 months. Incidences of grade 3-4 neutropenia and leukopenia were significantly higher in the palbociclib group. Eight fatal serious adverse events were reported.
Study details: A phase 3, double-blind, randomized PENELOPE-B study evaluated 1,250 high-risk patients with HR+, HER-negative early breast cancer who have residual invasive disease after neoadjuvant chemotherapy. Patients received estrogen therapy with either palbociclib or placebo.
Disclosures: The study was supported by Pfizer. The authors declared receiving consulting, honoraria, travel/accomodation expenses and research funding outside the study work. Some of the authors declared being employee of and/or stocks/ownership interests of various sources including Pfizer.
Source: Loibl S et al. J Clin Oncol. 2021 Apr 1. doi: 10.1200/JCO.20.03639.
Breast cancer: Axillary complete pathologic response varies by subtype
Key clinical point: The hormone receptor (HR)-negative/ human epidermal growth factor receptor 2 (HER2)-positive subtype was associated with the highest axillary pathologic complete response (pCR) rate and luminal A subtype was associated with the lowest axillary pCR rate.
Major finding: The axillary pCR rates were 60% for HR-negative/HER2-positive, 59% for HER2-positive, 48% for triple-negative, 45% for HR-positive/HER2-positive, 35% for luminal B, 18% for HR-positive/HER2-negative, and 13% for luminal A breast cancer subtypes.
Study details: A meta-analysis of 33 studies including 57,531 patients with breast cancer who received neoadjuvant systemic therapy.
Disclosures: The study was supported by a grant from the Dutch Cancer Society and Alpe d’Huzes Foundation. The authors received grants and personal fees outside this work.
Source: Samiei S et al. JAMA Surg. 2021 Apr 21. doi: 10.1001/jamasurg.2021.0891.
Key clinical point: The hormone receptor (HR)-negative/ human epidermal growth factor receptor 2 (HER2)-positive subtype was associated with the highest axillary pathologic complete response (pCR) rate and luminal A subtype was associated with the lowest axillary pCR rate.
Major finding: The axillary pCR rates were 60% for HR-negative/HER2-positive, 59% for HER2-positive, 48% for triple-negative, 45% for HR-positive/HER2-positive, 35% for luminal B, 18% for HR-positive/HER2-negative, and 13% for luminal A breast cancer subtypes.
Study details: A meta-analysis of 33 studies including 57,531 patients with breast cancer who received neoadjuvant systemic therapy.
Disclosures: The study was supported by a grant from the Dutch Cancer Society and Alpe d’Huzes Foundation. The authors received grants and personal fees outside this work.
Source: Samiei S et al. JAMA Surg. 2021 Apr 21. doi: 10.1001/jamasurg.2021.0891.
Key clinical point: The hormone receptor (HR)-negative/ human epidermal growth factor receptor 2 (HER2)-positive subtype was associated with the highest axillary pathologic complete response (pCR) rate and luminal A subtype was associated with the lowest axillary pCR rate.
Major finding: The axillary pCR rates were 60% for HR-negative/HER2-positive, 59% for HER2-positive, 48% for triple-negative, 45% for HR-positive/HER2-positive, 35% for luminal B, 18% for HR-positive/HER2-negative, and 13% for luminal A breast cancer subtypes.
Study details: A meta-analysis of 33 studies including 57,531 patients with breast cancer who received neoadjuvant systemic therapy.
Disclosures: The study was supported by a grant from the Dutch Cancer Society and Alpe d’Huzes Foundation. The authors received grants and personal fees outside this work.
Source: Samiei S et al. JAMA Surg. 2021 Apr 21. doi: 10.1001/jamasurg.2021.0891.