User login
Barriers to CAR T use in the spotlight at first European meeting
outcomes data suggest.
For that reason, and because bone marrow units are profit centers and CAR T-cell therapy reimbursement remains problematic, CAR T in the United States is “effectively being used as a bridge to transplant” – at a cost of more than $1 million per dose, economist Duane Schulthess told attendees at a recent, first-of-its-kind joint European CAR T-cell meeting in Paris, which was cosponsored by the European Hematology Association (EHA) and the European Society for Blood and Marrow Transplantation (EBMT).
“This is the way clinical practice is evolving right now; the price is not allowing enough experimentation for CAR T to flow up and be used in the less-diseased population,” said Mr. Schulthess, managing director of Vital Transformation, a consulting company based in Wezembeek-Oppem, Belgium.
In Europe, there is a slightly different problem in that health technology assessment bodies (HTAs) “have to figure out what they want to do” given the 2018 approvals of the first CAR T therapies there, he said, explaining that the data he presented was from a study commissioned by the Dutch government to help determine “what [CAR T] looks like from an effectiveness standpoint while they’re trying to figure out how much it’s worth and what they should pay.”
“Increasingly these are the big issues,” Mr. Schulthess said.
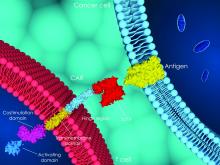
In August, the European Commission approved tisagenlecleucel (Kymriah) and axicabtagene ciloleucel (Yescarta) on the recommendation of the European Medicines Agency. Kymriah was approved for pediatric and young adult patients up to age 25 years with refractory B-cell acute lymphoblastic leukemia in relapse after transplant or in second or later relapse, as well as for adults patients with relapsed/refractory diffuse large B-cell lymphoma after failing at least two lines of systemic therapy, and Yescarta was approved for the latter and for the treatment of primary refractory mediastinal large B-cell lymphoma after at least two lines of systemic therapy.
The approvals have researchers and clinicians there clamoring for information about the therapy, which is revolutionizing the field of hematologic malignancies, according to Christian Chabannon, MD, PhD, chair of the EBMT Cellular Therapy & Immunobiology Working Party and vice-chair of the EBMT Scientific Council.
“An increasing number of European institutions are starting to administer this new category of medicinal products and increasingly contribute to ongoing clinical protocols and preclinical studies,” Dr. Chabannon said in an interview, explaining the urgency in planning the 1st European CAR T Cell Meeting just 6 months after the CAR T approvals in Europe.
EHA and EBMT brought together patient advocates, young investigators, and experts from across the globe to present the latest relevant information and data on topics ranging from current trials and experience, CAR T implementation and management, the preclinical and clinical pipelines, various CAR T applications, industry perspectives, and relevant economic issues, he said.
The latter is where Mr. Schulthess came in.
His research involved patient-level treatment pathway data from a database of more than 3 million patients treated with either allogeneic hematopoietic stem cell transplant (allo-HCT) or CAR T therapy across 5 years of experience. The data showed up to 85% response rates for each in the first-line setting. He and his colleagues then looked at therapy choices for those who failed to respond to second-line therapies and at how decisions were made regarding transplant and CAR T therapy – and specifically whether CAR T can be a substitute for transplant.
Ultimately, they looked at 29 allo-HCT recipients and 14 CAR T therapy recipients for a head-to-head comparison of the two treatments and performed an in-depth cost-efficacy analysis using a novel “visual pathology” methodology to account for limitations in the data.
The 3-year relapse-free survival probability was nearly 68% in the transplant recipients and 46% with CAR T.
“Now why is that? [Because] ... these populations are not the same; the CAR T population has a much higher disease burden,” Mr. Schulthess said. “So what we’re seeing [among] actual clinical doctors doing this for real – they are defaulting to bone marrow transplants, except in those cases where they do not have enough time or the patient does not respond. Then and only then are they giving CAR T.”
And that comes back to the fact that bone marrow units make money, he said.
CAR T is costly, and reimbursement can be problematic; these are disincentives for doctors to use CAR T therapy, at least in the United States, and while this is currently “being worked out,” the choice more often is “giving bone marrow transplant first and seeing what happens,” Mr. Schulthess said.
In Europe, that creates “a tough choice” for the HTAs, he said, noting that, in the absence of evidence of CAR T being curative in the subpopulation of patients with high disease burden who fail transplant and given the high cost, there is a push to determine at what point it begins to make sense economically.
“We think that you gain efficiency at ... roughly $277,000 [per dose] because [at that cost] you can do more CAR Ts than you can do bone marrow transplants. [CAR T] is less invasive, it’s lighter touch, it’s more efficient,” he said. “So if we were to see an efficiency cost of between $222,000 and $277,000, we think that works.”
Another recent study came to similar conclusions based on quality assessments, he said (J Clin Oncol. 2018 Sep 13. doi: 10.1200/JCO.2018.79.0642).
“We think that’s where this is going to end up, so we think that, if someone starts producing this for a couple hundred thousand bucks, then – certainly in Europe – it will make sense for this to start drifting up and being used as a substitute [to transplant],” he added.
Mr. Schulthess was one of scores of experts and investigators who presented at the EHA/EBMT joint meeting, which included numerous U.S. pioneers in the field and young European investigators, among others, Dr. Chabannon said.
Attesting to the enthusiasm in Europe regarding CAR T, Dr. Chabannon said that there were “more requests for registration than the venue could safely accommodate, a long waiting list, and a high number of individuals on the waiting list who registered for the live streaming” of the event.
“The field of CAR T cells is growing at a fast pace since the first clinical successes reported in the early 2010s, and one can wonder whether the expectations are not in excess of what reality will deliver,” he said. “Nevertheless, CAR T cells represent an essential innovation, not an incremental progress in biomedical sciences. They combine new mechanisms of action, clinical activity in advanced malignancies (and possibly beyond the field of cancer), transfer of manufacturing of human cell-based therapeutics to the industry, and potentially the first commercial success for a gene therapy.”
Surveys conducted by various professional associations, including EBMT, have clearly identified the potential for clinical successes that CAR T cells represent and the tremendous challenges raised by these innovations, he said, noting that “these include fulfilling specific educational needs.”
Therefore, EBMT and EHA have already announced that a second edition of the meeting is planned for Jan. 30 – Feb. 1, 2020, he noted.
Mr. Schulthess reported that his research was funded by the Dutch government.
outcomes data suggest.
For that reason, and because bone marrow units are profit centers and CAR T-cell therapy reimbursement remains problematic, CAR T in the United States is “effectively being used as a bridge to transplant” – at a cost of more than $1 million per dose, economist Duane Schulthess told attendees at a recent, first-of-its-kind joint European CAR T-cell meeting in Paris, which was cosponsored by the European Hematology Association (EHA) and the European Society for Blood and Marrow Transplantation (EBMT).
“This is the way clinical practice is evolving right now; the price is not allowing enough experimentation for CAR T to flow up and be used in the less-diseased population,” said Mr. Schulthess, managing director of Vital Transformation, a consulting company based in Wezembeek-Oppem, Belgium.
In Europe, there is a slightly different problem in that health technology assessment bodies (HTAs) “have to figure out what they want to do” given the 2018 approvals of the first CAR T therapies there, he said, explaining that the data he presented was from a study commissioned by the Dutch government to help determine “what [CAR T] looks like from an effectiveness standpoint while they’re trying to figure out how much it’s worth and what they should pay.”
“Increasingly these are the big issues,” Mr. Schulthess said.
In August, the European Commission approved tisagenlecleucel (Kymriah) and axicabtagene ciloleucel (Yescarta) on the recommendation of the European Medicines Agency. Kymriah was approved for pediatric and young adult patients up to age 25 years with refractory B-cell acute lymphoblastic leukemia in relapse after transplant or in second or later relapse, as well as for adults patients with relapsed/refractory diffuse large B-cell lymphoma after failing at least two lines of systemic therapy, and Yescarta was approved for the latter and for the treatment of primary refractory mediastinal large B-cell lymphoma after at least two lines of systemic therapy.
The approvals have researchers and clinicians there clamoring for information about the therapy, which is revolutionizing the field of hematologic malignancies, according to Christian Chabannon, MD, PhD, chair of the EBMT Cellular Therapy & Immunobiology Working Party and vice-chair of the EBMT Scientific Council.
“An increasing number of European institutions are starting to administer this new category of medicinal products and increasingly contribute to ongoing clinical protocols and preclinical studies,” Dr. Chabannon said in an interview, explaining the urgency in planning the 1st European CAR T Cell Meeting just 6 months after the CAR T approvals in Europe.
EHA and EBMT brought together patient advocates, young investigators, and experts from across the globe to present the latest relevant information and data on topics ranging from current trials and experience, CAR T implementation and management, the preclinical and clinical pipelines, various CAR T applications, industry perspectives, and relevant economic issues, he said.
The latter is where Mr. Schulthess came in.
His research involved patient-level treatment pathway data from a database of more than 3 million patients treated with either allogeneic hematopoietic stem cell transplant (allo-HCT) or CAR T therapy across 5 years of experience. The data showed up to 85% response rates for each in the first-line setting. He and his colleagues then looked at therapy choices for those who failed to respond to second-line therapies and at how decisions were made regarding transplant and CAR T therapy – and specifically whether CAR T can be a substitute for transplant.
Ultimately, they looked at 29 allo-HCT recipients and 14 CAR T therapy recipients for a head-to-head comparison of the two treatments and performed an in-depth cost-efficacy analysis using a novel “visual pathology” methodology to account for limitations in the data.
The 3-year relapse-free survival probability was nearly 68% in the transplant recipients and 46% with CAR T.
“Now why is that? [Because] ... these populations are not the same; the CAR T population has a much higher disease burden,” Mr. Schulthess said. “So what we’re seeing [among] actual clinical doctors doing this for real – they are defaulting to bone marrow transplants, except in those cases where they do not have enough time or the patient does not respond. Then and only then are they giving CAR T.”
And that comes back to the fact that bone marrow units make money, he said.
CAR T is costly, and reimbursement can be problematic; these are disincentives for doctors to use CAR T therapy, at least in the United States, and while this is currently “being worked out,” the choice more often is “giving bone marrow transplant first and seeing what happens,” Mr. Schulthess said.
In Europe, that creates “a tough choice” for the HTAs, he said, noting that, in the absence of evidence of CAR T being curative in the subpopulation of patients with high disease burden who fail transplant and given the high cost, there is a push to determine at what point it begins to make sense economically.
“We think that you gain efficiency at ... roughly $277,000 [per dose] because [at that cost] you can do more CAR Ts than you can do bone marrow transplants. [CAR T] is less invasive, it’s lighter touch, it’s more efficient,” he said. “So if we were to see an efficiency cost of between $222,000 and $277,000, we think that works.”
Another recent study came to similar conclusions based on quality assessments, he said (J Clin Oncol. 2018 Sep 13. doi: 10.1200/JCO.2018.79.0642).
“We think that’s where this is going to end up, so we think that, if someone starts producing this for a couple hundred thousand bucks, then – certainly in Europe – it will make sense for this to start drifting up and being used as a substitute [to transplant],” he added.
Mr. Schulthess was one of scores of experts and investigators who presented at the EHA/EBMT joint meeting, which included numerous U.S. pioneers in the field and young European investigators, among others, Dr. Chabannon said.
Attesting to the enthusiasm in Europe regarding CAR T, Dr. Chabannon said that there were “more requests for registration than the venue could safely accommodate, a long waiting list, and a high number of individuals on the waiting list who registered for the live streaming” of the event.
“The field of CAR T cells is growing at a fast pace since the first clinical successes reported in the early 2010s, and one can wonder whether the expectations are not in excess of what reality will deliver,” he said. “Nevertheless, CAR T cells represent an essential innovation, not an incremental progress in biomedical sciences. They combine new mechanisms of action, clinical activity in advanced malignancies (and possibly beyond the field of cancer), transfer of manufacturing of human cell-based therapeutics to the industry, and potentially the first commercial success for a gene therapy.”
Surveys conducted by various professional associations, including EBMT, have clearly identified the potential for clinical successes that CAR T cells represent and the tremendous challenges raised by these innovations, he said, noting that “these include fulfilling specific educational needs.”
Therefore, EBMT and EHA have already announced that a second edition of the meeting is planned for Jan. 30 – Feb. 1, 2020, he noted.
Mr. Schulthess reported that his research was funded by the Dutch government.
outcomes data suggest.
For that reason, and because bone marrow units are profit centers and CAR T-cell therapy reimbursement remains problematic, CAR T in the United States is “effectively being used as a bridge to transplant” – at a cost of more than $1 million per dose, economist Duane Schulthess told attendees at a recent, first-of-its-kind joint European CAR T-cell meeting in Paris, which was cosponsored by the European Hematology Association (EHA) and the European Society for Blood and Marrow Transplantation (EBMT).
“This is the way clinical practice is evolving right now; the price is not allowing enough experimentation for CAR T to flow up and be used in the less-diseased population,” said Mr. Schulthess, managing director of Vital Transformation, a consulting company based in Wezembeek-Oppem, Belgium.
In Europe, there is a slightly different problem in that health technology assessment bodies (HTAs) “have to figure out what they want to do” given the 2018 approvals of the first CAR T therapies there, he said, explaining that the data he presented was from a study commissioned by the Dutch government to help determine “what [CAR T] looks like from an effectiveness standpoint while they’re trying to figure out how much it’s worth and what they should pay.”
“Increasingly these are the big issues,” Mr. Schulthess said.
In August, the European Commission approved tisagenlecleucel (Kymriah) and axicabtagene ciloleucel (Yescarta) on the recommendation of the European Medicines Agency. Kymriah was approved for pediatric and young adult patients up to age 25 years with refractory B-cell acute lymphoblastic leukemia in relapse after transplant or in second or later relapse, as well as for adults patients with relapsed/refractory diffuse large B-cell lymphoma after failing at least two lines of systemic therapy, and Yescarta was approved for the latter and for the treatment of primary refractory mediastinal large B-cell lymphoma after at least two lines of systemic therapy.
The approvals have researchers and clinicians there clamoring for information about the therapy, which is revolutionizing the field of hematologic malignancies, according to Christian Chabannon, MD, PhD, chair of the EBMT Cellular Therapy & Immunobiology Working Party and vice-chair of the EBMT Scientific Council.
“An increasing number of European institutions are starting to administer this new category of medicinal products and increasingly contribute to ongoing clinical protocols and preclinical studies,” Dr. Chabannon said in an interview, explaining the urgency in planning the 1st European CAR T Cell Meeting just 6 months after the CAR T approvals in Europe.
EHA and EBMT brought together patient advocates, young investigators, and experts from across the globe to present the latest relevant information and data on topics ranging from current trials and experience, CAR T implementation and management, the preclinical and clinical pipelines, various CAR T applications, industry perspectives, and relevant economic issues, he said.
The latter is where Mr. Schulthess came in.
His research involved patient-level treatment pathway data from a database of more than 3 million patients treated with either allogeneic hematopoietic stem cell transplant (allo-HCT) or CAR T therapy across 5 years of experience. The data showed up to 85% response rates for each in the first-line setting. He and his colleagues then looked at therapy choices for those who failed to respond to second-line therapies and at how decisions were made regarding transplant and CAR T therapy – and specifically whether CAR T can be a substitute for transplant.
Ultimately, they looked at 29 allo-HCT recipients and 14 CAR T therapy recipients for a head-to-head comparison of the two treatments and performed an in-depth cost-efficacy analysis using a novel “visual pathology” methodology to account for limitations in the data.
The 3-year relapse-free survival probability was nearly 68% in the transplant recipients and 46% with CAR T.
“Now why is that? [Because] ... these populations are not the same; the CAR T population has a much higher disease burden,” Mr. Schulthess said. “So what we’re seeing [among] actual clinical doctors doing this for real – they are defaulting to bone marrow transplants, except in those cases where they do not have enough time or the patient does not respond. Then and only then are they giving CAR T.”
And that comes back to the fact that bone marrow units make money, he said.
CAR T is costly, and reimbursement can be problematic; these are disincentives for doctors to use CAR T therapy, at least in the United States, and while this is currently “being worked out,” the choice more often is “giving bone marrow transplant first and seeing what happens,” Mr. Schulthess said.
In Europe, that creates “a tough choice” for the HTAs, he said, noting that, in the absence of evidence of CAR T being curative in the subpopulation of patients with high disease burden who fail transplant and given the high cost, there is a push to determine at what point it begins to make sense economically.
“We think that you gain efficiency at ... roughly $277,000 [per dose] because [at that cost] you can do more CAR Ts than you can do bone marrow transplants. [CAR T] is less invasive, it’s lighter touch, it’s more efficient,” he said. “So if we were to see an efficiency cost of between $222,000 and $277,000, we think that works.”
Another recent study came to similar conclusions based on quality assessments, he said (J Clin Oncol. 2018 Sep 13. doi: 10.1200/JCO.2018.79.0642).
“We think that’s where this is going to end up, so we think that, if someone starts producing this for a couple hundred thousand bucks, then – certainly in Europe – it will make sense for this to start drifting up and being used as a substitute [to transplant],” he added.
Mr. Schulthess was one of scores of experts and investigators who presented at the EHA/EBMT joint meeting, which included numerous U.S. pioneers in the field and young European investigators, among others, Dr. Chabannon said.
Attesting to the enthusiasm in Europe regarding CAR T, Dr. Chabannon said that there were “more requests for registration than the venue could safely accommodate, a long waiting list, and a high number of individuals on the waiting list who registered for the live streaming” of the event.
“The field of CAR T cells is growing at a fast pace since the first clinical successes reported in the early 2010s, and one can wonder whether the expectations are not in excess of what reality will deliver,” he said. “Nevertheless, CAR T cells represent an essential innovation, not an incremental progress in biomedical sciences. They combine new mechanisms of action, clinical activity in advanced malignancies (and possibly beyond the field of cancer), transfer of manufacturing of human cell-based therapeutics to the industry, and potentially the first commercial success for a gene therapy.”
Surveys conducted by various professional associations, including EBMT, have clearly identified the potential for clinical successes that CAR T cells represent and the tremendous challenges raised by these innovations, he said, noting that “these include fulfilling specific educational needs.”
Therefore, EBMT and EHA have already announced that a second edition of the meeting is planned for Jan. 30 – Feb. 1, 2020, he noted.
Mr. Schulthess reported that his research was funded by the Dutch government.
Spinal cord atrophy found to be accelerated in subset of RRMS patients
DALLAS – The rate of spinal cord atrophy at the C1 level is promising as a prognostic biomarker for the future conversion to secondary progressive disease in patients with relapsing remitting multiple sclerosis (RRMS), results from a novel, single-center study suggest.
“Among all magnetic resonance imaging measures, spinal cord area shows the strongest correlations with MS disability and has been shown to discriminate progressive from relapsing remitting disease subtypes,” lead study author Antje Bischof, MD, said in an interview in advance of the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “In our work, we used a novel method to accurately measure upper cervical cord area at C1 vertebral level from brain MRI. This enabled us to show for the first time that compared to a matched group of patients who remained relapsing remitting MS over 2 decades.”
Dr. Bischof, a postdoctoral research fellow in the department of neurology at the University of California, San Francisco, and her colleagues matched 54 RRMS patients who converted to secondary progressive MS (SPMS) during the 12-year observation period with 54 RRMS patients who remained RRMS during the observation period, based on demographic and clinical criteria. Additionally, they evaluated 54 age- and sex-matched healthy controls at baseline. From routine T1-weighted brain MRI, they analyzed brain measures and spinal cord area at C1 level over 12 years to evaluate their potential to discriminate between the two matched groups during the preconversion period.
Subjects who developed SPMS showed higher rates of spinal cord atrophy (–2.2% per year; standard error, 0.2) before conversion to a secondary progressive course, compared with their RRMS matches who did not convert to SPMS (–0.7% per year; SE, 0.2; P less than .0001). Their data suggest that this difference exists at least 4 years before conversion to SPMS. “None of the commonly used measures of the brain including global brain volumes like white matter and gray matter, regional brain volumes like thalamus, and MS lesion volumes, discriminated between the patients with relapsing remitting MS who later converted to secondary progressive disease and the patients who remained RRMS,” Dr. Bischof said.
She acknowledged certain limitations of the study, including the small sample size and the fact that the results require confirmation in a second MS cohort in order to be generalizable. “These results suggest cervical cord atrophy rate at C1 level as a prognostic biomarker for the conversion to secondary progressive MS and could be useful for treatment decisions early in the disease course, and for the study of genetic, epidemiologic, and immune variables in MS,” Dr. Bischof concluded.
She reported having no financial disclosures.
SOURCE: Bischof A et al. ACTRIMS Forum 2019, Abstract P157.
DALLAS – The rate of spinal cord atrophy at the C1 level is promising as a prognostic biomarker for the future conversion to secondary progressive disease in patients with relapsing remitting multiple sclerosis (RRMS), results from a novel, single-center study suggest.
“Among all magnetic resonance imaging measures, spinal cord area shows the strongest correlations with MS disability and has been shown to discriminate progressive from relapsing remitting disease subtypes,” lead study author Antje Bischof, MD, said in an interview in advance of the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “In our work, we used a novel method to accurately measure upper cervical cord area at C1 vertebral level from brain MRI. This enabled us to show for the first time that compared to a matched group of patients who remained relapsing remitting MS over 2 decades.”
Dr. Bischof, a postdoctoral research fellow in the department of neurology at the University of California, San Francisco, and her colleagues matched 54 RRMS patients who converted to secondary progressive MS (SPMS) during the 12-year observation period with 54 RRMS patients who remained RRMS during the observation period, based on demographic and clinical criteria. Additionally, they evaluated 54 age- and sex-matched healthy controls at baseline. From routine T1-weighted brain MRI, they analyzed brain measures and spinal cord area at C1 level over 12 years to evaluate their potential to discriminate between the two matched groups during the preconversion period.
Subjects who developed SPMS showed higher rates of spinal cord atrophy (–2.2% per year; standard error, 0.2) before conversion to a secondary progressive course, compared with their RRMS matches who did not convert to SPMS (–0.7% per year; SE, 0.2; P less than .0001). Their data suggest that this difference exists at least 4 years before conversion to SPMS. “None of the commonly used measures of the brain including global brain volumes like white matter and gray matter, regional brain volumes like thalamus, and MS lesion volumes, discriminated between the patients with relapsing remitting MS who later converted to secondary progressive disease and the patients who remained RRMS,” Dr. Bischof said.
She acknowledged certain limitations of the study, including the small sample size and the fact that the results require confirmation in a second MS cohort in order to be generalizable. “These results suggest cervical cord atrophy rate at C1 level as a prognostic biomarker for the conversion to secondary progressive MS and could be useful for treatment decisions early in the disease course, and for the study of genetic, epidemiologic, and immune variables in MS,” Dr. Bischof concluded.
She reported having no financial disclosures.
SOURCE: Bischof A et al. ACTRIMS Forum 2019, Abstract P157.
DALLAS – The rate of spinal cord atrophy at the C1 level is promising as a prognostic biomarker for the future conversion to secondary progressive disease in patients with relapsing remitting multiple sclerosis (RRMS), results from a novel, single-center study suggest.
“Among all magnetic resonance imaging measures, spinal cord area shows the strongest correlations with MS disability and has been shown to discriminate progressive from relapsing remitting disease subtypes,” lead study author Antje Bischof, MD, said in an interview in advance of the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “In our work, we used a novel method to accurately measure upper cervical cord area at C1 vertebral level from brain MRI. This enabled us to show for the first time that compared to a matched group of patients who remained relapsing remitting MS over 2 decades.”
Dr. Bischof, a postdoctoral research fellow in the department of neurology at the University of California, San Francisco, and her colleagues matched 54 RRMS patients who converted to secondary progressive MS (SPMS) during the 12-year observation period with 54 RRMS patients who remained RRMS during the observation period, based on demographic and clinical criteria. Additionally, they evaluated 54 age- and sex-matched healthy controls at baseline. From routine T1-weighted brain MRI, they analyzed brain measures and spinal cord area at C1 level over 12 years to evaluate their potential to discriminate between the two matched groups during the preconversion period.
Subjects who developed SPMS showed higher rates of spinal cord atrophy (–2.2% per year; standard error, 0.2) before conversion to a secondary progressive course, compared with their RRMS matches who did not convert to SPMS (–0.7% per year; SE, 0.2; P less than .0001). Their data suggest that this difference exists at least 4 years before conversion to SPMS. “None of the commonly used measures of the brain including global brain volumes like white matter and gray matter, regional brain volumes like thalamus, and MS lesion volumes, discriminated between the patients with relapsing remitting MS who later converted to secondary progressive disease and the patients who remained RRMS,” Dr. Bischof said.
She acknowledged certain limitations of the study, including the small sample size and the fact that the results require confirmation in a second MS cohort in order to be generalizable. “These results suggest cervical cord atrophy rate at C1 level as a prognostic biomarker for the conversion to secondary progressive MS and could be useful for treatment decisions early in the disease course, and for the study of genetic, epidemiologic, and immune variables in MS,” Dr. Bischof concluded.
She reported having no financial disclosures.
SOURCE: Bischof A et al. ACTRIMS Forum 2019, Abstract P157.
REPORTING FROM ACTRIMS FORUM 2019
Key clinical point: In patients with relapsing remitting MS (RRMS), upper cervical cord atrophy, as obtained from routine T1-weighted brain MRI, is a strong indicator of impending conversion to secondary progressive MS (SPMS).
Major finding: Subjects who developed SPMS showed higher rates of spinal cord atrophy (–2.2% per year; standard error, 0.2) before conversion to a secondary progressive course, compared with their RRMS matches who did not convert to SPMS (–0.7% per year; SE, 0.2; P less than .0001).
Study details: A single-center, observational study of 54 RRMS patients who converted to SPMS during the 12-year observation and 54 RRMS patients who remained RRMS during the observation period.
Disclosures: Dr. Bischof reported having no financial disclosures.
Source: Bischof A et al. ACTRIMS Forum 2019, Abstract P157.
CLL, GVHD may raise risk for skin cancer after allo-HCT
Previously unknown risk factors for secondary skin cancer linked with allogeneic hematopoietic cell transplantation (HCT) have been identified, researchers report after a retrospective analysis.
“We confirmed [graft-versus-host disease] as a risk factor, identified [chronic lymphocytic leukemia] as an additional risk factor, and found that patients who received myeloablative transplants in adulthood had fewer [basal cell carcinomas] than their counterparts,” Peggy A. Wu, MD, of the Beth Israel Deaconess Medical Center in Boston, and her colleagues wrote in the Journal of Investigative Dermatology.
The team analyzed 1,974 patients who underwent transplantation for various types of hematologic cancer and survived for a minimum of 100 days following transplant. Among this cohort, 119 patients developed various forms of skin cancer, including basal and squamous cell carcinoma.
Reports of skin malignancy were confirmed using physician records and pathology reports. Dr. Wu and her colleagues excluded patients whose indication for transplant was a primary immunodeficiency or Fanconi anemia.
“Reflecting advances that allow older patients to be eligible for HCT, the median age at transplantation of our cohort was one of the oldest (51.1 years) in the literature,” the researchers wrote.
In univariable models, the researchers found that prior chronic lymphocytic leukemia (CLL) (hazard ratio, 2.2; 95% CI, 1.3-3.7), chronic graft-versus-host disease (GVHD) (HR, 3.1; 95% CI, 1.7-5.4), and age at transplant of more than 60 years (HR, 10.8; 95% CI, 3.3-35.6) were all linked to an increased risk for squamous cell carcinomas. A multivariable analysis found that these factors continued as significant risk factors.
For basal cell carcinomas, the risk factors identified were prior CLL (HR, 3.5; 95% CI, 2.0-6.4), acute GVHD (HR, 1.9; 95% CI, 1.1-3.3), and chronic GVHD (HR, 3.2; 95% CI, 1.6-6.5) using univariable models. These factors all continued to be significant in multivariable analysis.
Additionally, the researchers found that a myeloablative conditioning regimen and total body irradiation were protective against development of basal cell carcinomas in univariable models. However, the protective effect continued for myeloablative condition in the multivariable model only.
“To our knowledge, previously unreported risk factors in this contemporary cohort include prior CLL for squamous cell carcinoma and basal cell carcinoma and reduced-intensity conditioning for basal cell carcinoma,” the researchers wrote.
The study was supported by the Skin Cancer Foundation, Women’s Dermatologic Society, Harvard Catalyst, and Harvard University. The authors reported having no conflicts of interest.
SOURCE: Wu PA et al. J Invest Dermatol. 2019 Mar;139(3):591-9.
Previously unknown risk factors for secondary skin cancer linked with allogeneic hematopoietic cell transplantation (HCT) have been identified, researchers report after a retrospective analysis.
“We confirmed [graft-versus-host disease] as a risk factor, identified [chronic lymphocytic leukemia] as an additional risk factor, and found that patients who received myeloablative transplants in adulthood had fewer [basal cell carcinomas] than their counterparts,” Peggy A. Wu, MD, of the Beth Israel Deaconess Medical Center in Boston, and her colleagues wrote in the Journal of Investigative Dermatology.
The team analyzed 1,974 patients who underwent transplantation for various types of hematologic cancer and survived for a minimum of 100 days following transplant. Among this cohort, 119 patients developed various forms of skin cancer, including basal and squamous cell carcinoma.
Reports of skin malignancy were confirmed using physician records and pathology reports. Dr. Wu and her colleagues excluded patients whose indication for transplant was a primary immunodeficiency or Fanconi anemia.
“Reflecting advances that allow older patients to be eligible for HCT, the median age at transplantation of our cohort was one of the oldest (51.1 years) in the literature,” the researchers wrote.
In univariable models, the researchers found that prior chronic lymphocytic leukemia (CLL) (hazard ratio, 2.2; 95% CI, 1.3-3.7), chronic graft-versus-host disease (GVHD) (HR, 3.1; 95% CI, 1.7-5.4), and age at transplant of more than 60 years (HR, 10.8; 95% CI, 3.3-35.6) were all linked to an increased risk for squamous cell carcinomas. A multivariable analysis found that these factors continued as significant risk factors.
For basal cell carcinomas, the risk factors identified were prior CLL (HR, 3.5; 95% CI, 2.0-6.4), acute GVHD (HR, 1.9; 95% CI, 1.1-3.3), and chronic GVHD (HR, 3.2; 95% CI, 1.6-6.5) using univariable models. These factors all continued to be significant in multivariable analysis.
Additionally, the researchers found that a myeloablative conditioning regimen and total body irradiation were protective against development of basal cell carcinomas in univariable models. However, the protective effect continued for myeloablative condition in the multivariable model only.
“To our knowledge, previously unreported risk factors in this contemporary cohort include prior CLL for squamous cell carcinoma and basal cell carcinoma and reduced-intensity conditioning for basal cell carcinoma,” the researchers wrote.
The study was supported by the Skin Cancer Foundation, Women’s Dermatologic Society, Harvard Catalyst, and Harvard University. The authors reported having no conflicts of interest.
SOURCE: Wu PA et al. J Invest Dermatol. 2019 Mar;139(3):591-9.
Previously unknown risk factors for secondary skin cancer linked with allogeneic hematopoietic cell transplantation (HCT) have been identified, researchers report after a retrospective analysis.
“We confirmed [graft-versus-host disease] as a risk factor, identified [chronic lymphocytic leukemia] as an additional risk factor, and found that patients who received myeloablative transplants in adulthood had fewer [basal cell carcinomas] than their counterparts,” Peggy A. Wu, MD, of the Beth Israel Deaconess Medical Center in Boston, and her colleagues wrote in the Journal of Investigative Dermatology.
The team analyzed 1,974 patients who underwent transplantation for various types of hematologic cancer and survived for a minimum of 100 days following transplant. Among this cohort, 119 patients developed various forms of skin cancer, including basal and squamous cell carcinoma.
Reports of skin malignancy were confirmed using physician records and pathology reports. Dr. Wu and her colleagues excluded patients whose indication for transplant was a primary immunodeficiency or Fanconi anemia.
“Reflecting advances that allow older patients to be eligible for HCT, the median age at transplantation of our cohort was one of the oldest (51.1 years) in the literature,” the researchers wrote.
In univariable models, the researchers found that prior chronic lymphocytic leukemia (CLL) (hazard ratio, 2.2; 95% CI, 1.3-3.7), chronic graft-versus-host disease (GVHD) (HR, 3.1; 95% CI, 1.7-5.4), and age at transplant of more than 60 years (HR, 10.8; 95% CI, 3.3-35.6) were all linked to an increased risk for squamous cell carcinomas. A multivariable analysis found that these factors continued as significant risk factors.
For basal cell carcinomas, the risk factors identified were prior CLL (HR, 3.5; 95% CI, 2.0-6.4), acute GVHD (HR, 1.9; 95% CI, 1.1-3.3), and chronic GVHD (HR, 3.2; 95% CI, 1.6-6.5) using univariable models. These factors all continued to be significant in multivariable analysis.
Additionally, the researchers found that a myeloablative conditioning regimen and total body irradiation were protective against development of basal cell carcinomas in univariable models. However, the protective effect continued for myeloablative condition in the multivariable model only.
“To our knowledge, previously unreported risk factors in this contemporary cohort include prior CLL for squamous cell carcinoma and basal cell carcinoma and reduced-intensity conditioning for basal cell carcinoma,” the researchers wrote.
The study was supported by the Skin Cancer Foundation, Women’s Dermatologic Society, Harvard Catalyst, and Harvard University. The authors reported having no conflicts of interest.
SOURCE: Wu PA et al. J Invest Dermatol. 2019 Mar;139(3):591-9.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
ZUMA-1 update: Axi-cel responses persist at 2 years
HOUSTON – With a median follow-up now exceeding 2 years, 39% of refractory large B-cell lymphoma patients enrolled in the pivotal ZUMA-1 trial have maintained ongoing response to axicabtagene ciloleucel, according to an investigator involved in the study.
Median duration of response to axi-cel and median overall survival have not yet been reached, while a recent subset analysis showed that nearly half of patients with certain high-risk characteristics had a durable response, said investigator Sattva S. Neelapu, MD, of the University of Texas MD Anderson Cancer Center, Houston.
Evidence of B-cell recovery and a decrease in detectable, gene-marked CAR T cells have been noted in further follow-up, suggesting that functional CAR T-cell persistence may not be required for long-term remissions, Dr. Neelapu added.
“These data support [the conclusion] that axi-cel induces durable remissions in patients with large B-cell lymphoma who otherwise lack curative options,” Dr. Neelapu said at the Transplantation & Cellular Therapy Meetings.
The update on the phase 1/2 ZUMA-1 study included 108 patients with refractory large B-cell lymphoma who received axi-cel, the CD19-directed autologous chimeric antigen receptor (CAR) T-cell therapy.
In a previously reported 1-year update on the trial, 42% of patients had ongoing responses, Dr. Neelapu said. In the present update, with a median follow-up of 27.1 months, ongoing responses were seen in 39%, most of whom (37%) were in complete response, according to the data presented.
Thirty-three patients in the phase 2 portion of ZUMA-1 were known to have double-expressor or high-grade B-cell lymphoma, according to the investigator. In this high-risk subset, 48% were in ongoing complete response at the 2-year follow-up.
Progression-free survival in ZUMA-1 plateaued at the 6 month-follow-up, according to Dr. Neelapu, who said that plateau has been largely maintained, with just 10 patients progressing since then. Median progression-free survival is 5.9 months and median overall survival has not been reached, with a 24-month overall survival of 51%.
Late-onset serious adverse events mainly consisted of manageable infections, none of which were considered related to axi-cel treatment, according to Dr. Neelapu.
The proportion of ongoing responders with detectable CAR T-cells has decreased over time, from 95% at 3 months to 66% at 24 months, Dr. Neelapu reported. Meanwhile, the proportion of ongoing responders with detectable B cells after axi-cel treatment has gone from 17% to 75%.
More details on the 2-year follow-up data from ZUMA-1 were reported recently in the Lancet Oncology (2019 Jan;20[1]:31-42).
Funding for ZUMA-1 came from Kite and the Leukemia & Lymphoma Society. Dr. Neelapu reported disclosures related to Kite, Celgene, Cellectis, Merck, Poseida, Acerta, Karus, Bristol-Myers Squibb, Novartis, and Unum Therapeutics.
The meeting was held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research. At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
SOURCE: Neelapu SS et al. TCT 2019, Abstract 82.
HOUSTON – With a median follow-up now exceeding 2 years, 39% of refractory large B-cell lymphoma patients enrolled in the pivotal ZUMA-1 trial have maintained ongoing response to axicabtagene ciloleucel, according to an investigator involved in the study.
Median duration of response to axi-cel and median overall survival have not yet been reached, while a recent subset analysis showed that nearly half of patients with certain high-risk characteristics had a durable response, said investigator Sattva S. Neelapu, MD, of the University of Texas MD Anderson Cancer Center, Houston.
Evidence of B-cell recovery and a decrease in detectable, gene-marked CAR T cells have been noted in further follow-up, suggesting that functional CAR T-cell persistence may not be required for long-term remissions, Dr. Neelapu added.
“These data support [the conclusion] that axi-cel induces durable remissions in patients with large B-cell lymphoma who otherwise lack curative options,” Dr. Neelapu said at the Transplantation & Cellular Therapy Meetings.
The update on the phase 1/2 ZUMA-1 study included 108 patients with refractory large B-cell lymphoma who received axi-cel, the CD19-directed autologous chimeric antigen receptor (CAR) T-cell therapy.
In a previously reported 1-year update on the trial, 42% of patients had ongoing responses, Dr. Neelapu said. In the present update, with a median follow-up of 27.1 months, ongoing responses were seen in 39%, most of whom (37%) were in complete response, according to the data presented.
Thirty-three patients in the phase 2 portion of ZUMA-1 were known to have double-expressor or high-grade B-cell lymphoma, according to the investigator. In this high-risk subset, 48% were in ongoing complete response at the 2-year follow-up.
Progression-free survival in ZUMA-1 plateaued at the 6 month-follow-up, according to Dr. Neelapu, who said that plateau has been largely maintained, with just 10 patients progressing since then. Median progression-free survival is 5.9 months and median overall survival has not been reached, with a 24-month overall survival of 51%.
Late-onset serious adverse events mainly consisted of manageable infections, none of which were considered related to axi-cel treatment, according to Dr. Neelapu.
The proportion of ongoing responders with detectable CAR T-cells has decreased over time, from 95% at 3 months to 66% at 24 months, Dr. Neelapu reported. Meanwhile, the proportion of ongoing responders with detectable B cells after axi-cel treatment has gone from 17% to 75%.
More details on the 2-year follow-up data from ZUMA-1 were reported recently in the Lancet Oncology (2019 Jan;20[1]:31-42).
Funding for ZUMA-1 came from Kite and the Leukemia & Lymphoma Society. Dr. Neelapu reported disclosures related to Kite, Celgene, Cellectis, Merck, Poseida, Acerta, Karus, Bristol-Myers Squibb, Novartis, and Unum Therapeutics.
The meeting was held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research. At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
SOURCE: Neelapu SS et al. TCT 2019, Abstract 82.
HOUSTON – With a median follow-up now exceeding 2 years, 39% of refractory large B-cell lymphoma patients enrolled in the pivotal ZUMA-1 trial have maintained ongoing response to axicabtagene ciloleucel, according to an investigator involved in the study.
Median duration of response to axi-cel and median overall survival have not yet been reached, while a recent subset analysis showed that nearly half of patients with certain high-risk characteristics had a durable response, said investigator Sattva S. Neelapu, MD, of the University of Texas MD Anderson Cancer Center, Houston.
Evidence of B-cell recovery and a decrease in detectable, gene-marked CAR T cells have been noted in further follow-up, suggesting that functional CAR T-cell persistence may not be required for long-term remissions, Dr. Neelapu added.
“These data support [the conclusion] that axi-cel induces durable remissions in patients with large B-cell lymphoma who otherwise lack curative options,” Dr. Neelapu said at the Transplantation & Cellular Therapy Meetings.
The update on the phase 1/2 ZUMA-1 study included 108 patients with refractory large B-cell lymphoma who received axi-cel, the CD19-directed autologous chimeric antigen receptor (CAR) T-cell therapy.
In a previously reported 1-year update on the trial, 42% of patients had ongoing responses, Dr. Neelapu said. In the present update, with a median follow-up of 27.1 months, ongoing responses were seen in 39%, most of whom (37%) were in complete response, according to the data presented.
Thirty-three patients in the phase 2 portion of ZUMA-1 were known to have double-expressor or high-grade B-cell lymphoma, according to the investigator. In this high-risk subset, 48% were in ongoing complete response at the 2-year follow-up.
Progression-free survival in ZUMA-1 plateaued at the 6 month-follow-up, according to Dr. Neelapu, who said that plateau has been largely maintained, with just 10 patients progressing since then. Median progression-free survival is 5.9 months and median overall survival has not been reached, with a 24-month overall survival of 51%.
Late-onset serious adverse events mainly consisted of manageable infections, none of which were considered related to axi-cel treatment, according to Dr. Neelapu.
The proportion of ongoing responders with detectable CAR T-cells has decreased over time, from 95% at 3 months to 66% at 24 months, Dr. Neelapu reported. Meanwhile, the proportion of ongoing responders with detectable B cells after axi-cel treatment has gone from 17% to 75%.
More details on the 2-year follow-up data from ZUMA-1 were reported recently in the Lancet Oncology (2019 Jan;20[1]:31-42).
Funding for ZUMA-1 came from Kite and the Leukemia & Lymphoma Society. Dr. Neelapu reported disclosures related to Kite, Celgene, Cellectis, Merck, Poseida, Acerta, Karus, Bristol-Myers Squibb, Novartis, and Unum Therapeutics.
The meeting was held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research. At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
SOURCE: Neelapu SS et al. TCT 2019, Abstract 82.
REPORTING FROM TCT 2019
Dual-targeted CAR T shows ‘clinical signal’ in NHL
HOUSTON – A dual-targeted, locally manufactured, anti-CD19/CD20 chimeric antigen receptor (CAR) T-cell therapy was safe and produced ongoing complete responses in a phase 1 study of heavily pretreated non-Hodgkin lymphoma patients, an investigator reported.
The bispecific CAR T-cell product, designed to limit relapses due to loss of target antigen, was produced at the point of care with a 100% success rate for these heavily pretreated patients, the first of whom has now maintained a complete response for 19 months, said Parameswaran Hari, MD, of the Medical College of Wisconsin, Milwaukee.
“So far, this trial has demonstrated safety for this bispecific vector and suggests a clinical signal, with 7 out of 12 patients with ongoing CR, and with minimal toxicity,” Dr. Hari said at the Transplantation & Cellular Therapy Meetings.
“Point of care delivery, we think, allowed patients to have fresh infusion of CAR T cells, with the avoidance of cryopreservation,” added Dr. Hari, who presented the results on behalf of coinvestigators at the Medical College of Wisconsin and Lentigen Technology.
There was no grade 3 or 4 neurotoxicity or cytokine release syndrome among the 12 patients reported to date in the phase 1, dose-escalation trial, and no patient required intensive care, according to Dr. Hari. Grade 1 and 2 neurotoxicity occurred in two and one patients, respectively, while grade 1 and 2 cytokine release syndrome was observed in three patients each.
Among the 12 patients treated to date, the overall response rate was 81% at day 28, Dr. Hari said, noting that of 6 patients treated at the goal dose of 2.5 x 106 cells/kg, 5 remain in ongoing complete remission.
The median age of patients enrolled in the study was 55 years. Six patients had diffuse large B-cell lymphoma, five had mantle cell lymphoma, and two had chronic lymphocytic leukemia. All but one patient underwent fludarabine/cyclophosphamide lymphodepletion prior to receiving the inpatient CAR T-cell infusions, given over the course of 2 days.
Dr. Hari specifically highlighted the mantle cell lymphoma patient subset, noting that four out of five patients were in complete remission at day 28, and remained in ongoing complete remission at times ranging from 1 to 16 months.
With a set manufacturing time of 14 days, production was successful in all 12 patients, and 10 were able to receive fresh product, while 3 received cryopreserved product due to illness-related delays and a holiday, according to the investigators.
“The time to actual delivery of CAR T cell in the patient is actually shortened dramatically,” Dr. Hari said. “We hope to get it down to day 10.”
Local manufacturing can also reduce some costs associated with CAR T-cell production, such as shipping and courier costs, he added.
Taken together, these findings suggest that locally manufactured anti-CD19/CD20 CAR T cells could improve clinical outcomes for patients with relapsed and refractory B-cell NHL, with efficiency through point-of-care delivery, Dr. Hari concluded.
Further studies are planned to evaluate the efficacy of the product and to investigate the mechanism of relapse or progression in patients who experience treatment failure, he said.
Dr. Hari reported disclosures related to Juno, Kite, Spectrum, Janssen, Takeda, Celgene, and BMS. Several study coauthors reported that they were employed by Lentigen Technology, a Miltenyi Biotec Company.
The meeting was held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research. At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
SOURCE: Shah NN et al. TCT 2019, Abstract 80.
HOUSTON – A dual-targeted, locally manufactured, anti-CD19/CD20 chimeric antigen receptor (CAR) T-cell therapy was safe and produced ongoing complete responses in a phase 1 study of heavily pretreated non-Hodgkin lymphoma patients, an investigator reported.
The bispecific CAR T-cell product, designed to limit relapses due to loss of target antigen, was produced at the point of care with a 100% success rate for these heavily pretreated patients, the first of whom has now maintained a complete response for 19 months, said Parameswaran Hari, MD, of the Medical College of Wisconsin, Milwaukee.
“So far, this trial has demonstrated safety for this bispecific vector and suggests a clinical signal, with 7 out of 12 patients with ongoing CR, and with minimal toxicity,” Dr. Hari said at the Transplantation & Cellular Therapy Meetings.
“Point of care delivery, we think, allowed patients to have fresh infusion of CAR T cells, with the avoidance of cryopreservation,” added Dr. Hari, who presented the results on behalf of coinvestigators at the Medical College of Wisconsin and Lentigen Technology.
There was no grade 3 or 4 neurotoxicity or cytokine release syndrome among the 12 patients reported to date in the phase 1, dose-escalation trial, and no patient required intensive care, according to Dr. Hari. Grade 1 and 2 neurotoxicity occurred in two and one patients, respectively, while grade 1 and 2 cytokine release syndrome was observed in three patients each.
Among the 12 patients treated to date, the overall response rate was 81% at day 28, Dr. Hari said, noting that of 6 patients treated at the goal dose of 2.5 x 106 cells/kg, 5 remain in ongoing complete remission.
The median age of patients enrolled in the study was 55 years. Six patients had diffuse large B-cell lymphoma, five had mantle cell lymphoma, and two had chronic lymphocytic leukemia. All but one patient underwent fludarabine/cyclophosphamide lymphodepletion prior to receiving the inpatient CAR T-cell infusions, given over the course of 2 days.
Dr. Hari specifically highlighted the mantle cell lymphoma patient subset, noting that four out of five patients were in complete remission at day 28, and remained in ongoing complete remission at times ranging from 1 to 16 months.
With a set manufacturing time of 14 days, production was successful in all 12 patients, and 10 were able to receive fresh product, while 3 received cryopreserved product due to illness-related delays and a holiday, according to the investigators.
“The time to actual delivery of CAR T cell in the patient is actually shortened dramatically,” Dr. Hari said. “We hope to get it down to day 10.”
Local manufacturing can also reduce some costs associated with CAR T-cell production, such as shipping and courier costs, he added.
Taken together, these findings suggest that locally manufactured anti-CD19/CD20 CAR T cells could improve clinical outcomes for patients with relapsed and refractory B-cell NHL, with efficiency through point-of-care delivery, Dr. Hari concluded.
Further studies are planned to evaluate the efficacy of the product and to investigate the mechanism of relapse or progression in patients who experience treatment failure, he said.
Dr. Hari reported disclosures related to Juno, Kite, Spectrum, Janssen, Takeda, Celgene, and BMS. Several study coauthors reported that they were employed by Lentigen Technology, a Miltenyi Biotec Company.
The meeting was held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research. At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
SOURCE: Shah NN et al. TCT 2019, Abstract 80.
HOUSTON – A dual-targeted, locally manufactured, anti-CD19/CD20 chimeric antigen receptor (CAR) T-cell therapy was safe and produced ongoing complete responses in a phase 1 study of heavily pretreated non-Hodgkin lymphoma patients, an investigator reported.
The bispecific CAR T-cell product, designed to limit relapses due to loss of target antigen, was produced at the point of care with a 100% success rate for these heavily pretreated patients, the first of whom has now maintained a complete response for 19 months, said Parameswaran Hari, MD, of the Medical College of Wisconsin, Milwaukee.
“So far, this trial has demonstrated safety for this bispecific vector and suggests a clinical signal, with 7 out of 12 patients with ongoing CR, and with minimal toxicity,” Dr. Hari said at the Transplantation & Cellular Therapy Meetings.
“Point of care delivery, we think, allowed patients to have fresh infusion of CAR T cells, with the avoidance of cryopreservation,” added Dr. Hari, who presented the results on behalf of coinvestigators at the Medical College of Wisconsin and Lentigen Technology.
There was no grade 3 or 4 neurotoxicity or cytokine release syndrome among the 12 patients reported to date in the phase 1, dose-escalation trial, and no patient required intensive care, according to Dr. Hari. Grade 1 and 2 neurotoxicity occurred in two and one patients, respectively, while grade 1 and 2 cytokine release syndrome was observed in three patients each.
Among the 12 patients treated to date, the overall response rate was 81% at day 28, Dr. Hari said, noting that of 6 patients treated at the goal dose of 2.5 x 106 cells/kg, 5 remain in ongoing complete remission.
The median age of patients enrolled in the study was 55 years. Six patients had diffuse large B-cell lymphoma, five had mantle cell lymphoma, and two had chronic lymphocytic leukemia. All but one patient underwent fludarabine/cyclophosphamide lymphodepletion prior to receiving the inpatient CAR T-cell infusions, given over the course of 2 days.
Dr. Hari specifically highlighted the mantle cell lymphoma patient subset, noting that four out of five patients were in complete remission at day 28, and remained in ongoing complete remission at times ranging from 1 to 16 months.
With a set manufacturing time of 14 days, production was successful in all 12 patients, and 10 were able to receive fresh product, while 3 received cryopreserved product due to illness-related delays and a holiday, according to the investigators.
“The time to actual delivery of CAR T cell in the patient is actually shortened dramatically,” Dr. Hari said. “We hope to get it down to day 10.”
Local manufacturing can also reduce some costs associated with CAR T-cell production, such as shipping and courier costs, he added.
Taken together, these findings suggest that locally manufactured anti-CD19/CD20 CAR T cells could improve clinical outcomes for patients with relapsed and refractory B-cell NHL, with efficiency through point-of-care delivery, Dr. Hari concluded.
Further studies are planned to evaluate the efficacy of the product and to investigate the mechanism of relapse or progression in patients who experience treatment failure, he said.
Dr. Hari reported disclosures related to Juno, Kite, Spectrum, Janssen, Takeda, Celgene, and BMS. Several study coauthors reported that they were employed by Lentigen Technology, a Miltenyi Biotec Company.
The meeting was held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research. At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
SOURCE: Shah NN et al. TCT 2019, Abstract 80.
REPORTING FROM TCT 2019
Experts Weigh in on Medication Overuse Headache
Following the American Headache Society’s Scottsdale Headache Symposium in November 2018, MedPage Today posted an article which shared differing opinions from Drs. Hans-Christoph Diener and Elizabeth Loder on medication overuse headache (MOH). While Dr. Diener noted that “we can identify people with chronic migraine who are at risk to have medication overuse,” and that following successful withdrawal treatment “the majority of patients…revert to episodic migraine,” Dr. Loder pointed out that while MOH may exist and contribute to chronic migraine, “it is over-emphasized and the evidence in support of these concepts is weak.”
For this article, I’ve asked Drs. Marcelo Bigal, Rob Cowan, Jack Schim, and Stewart J. Tepper to share their perspectives on this topic. I also asked both Dr. Diener and Dr. Loder to expand on their comments. We will see Dr. Diener’s response to the article, but we did not hear back from Dr. Loder. Lastly, I will weigh in on the MOH discussion.
Marcelo Bigal, MD, PhD
Chief Medical Officer, Purdue Pharma
The issue of medication overuse headache (MOH) needs to be disentangled into a few separate but related issues. First, do excessive medications make migraine worse? Second, should MOH be considered a distinct form of headache? And how can evidence inform clinical practice?
Robust evidence supports the fact that excessive acute medication use is associated with increased headache frequency among migraineurs. In a large epidemiological study, we demonstrated that exposure (medication) precedes outcome (increased headache frequency).1 The risk was higher for barbiturates, followed by opioids and triptans, and was not increased by nonsteroidal anti-inflammatory drugs (NSAIDs). Dose response and critical exposure levels were identified. Based on this study and several others, we argued that criteria of causality had been demonstrated beyond reasonable doubt.2
However, since the effect is specific to migraine in that the exposure only increases the risk in migraineurs, not in individuals with other types of pain, we do not consider MOH a distinct entity. Instead, we believe that excessive medication is a risk factor for chronic migraine (CM). Therefore, we should be able to subdivide CM into 2 groups, one with and one without excessive medication use.
From a clinical perspective, physicians should monitor acute medication consumption in individuals with migraine and should be liberal in starting preventive therapy. In those with CM and excessive acute medication use we don’t advocate abrupt discontinuation of acute medications, since some preventive medications, especially the newer anti-calcitonin gene-related peptide (CGRP) antibodies, seem to work equally well in individuals with and without excessive use of medication,3 allowing more natural and gradual control of acute medication consumption without the need for detoxification.
Rob Cowan, MD, FAAN, FAHS
Higgins Professor of Neurology and Neurosciences
Chief, Division of Headache Medicine, Dept. of Neurology and Neurosciences
Director, Stanford University School of Medicine
As is often the case when 2 smart people take artificially imposed opposite positions, the truth will lie somewhere in the middle. I doubt either of the debaters would argue either extreme position: MOH does not exist, or MOH when present is solely responsible for chronic daily headache (CDH). The argument that the absence of controlled studies negates the proposition fails the common-sense test: Without a controlled study, we can’t be sure that wearing a helmet when bicycling is better than not. In such a case, observational data is sufficient. Could there be confounders (eg, helmet wearers are more inclined to ride safely)? Of course, but is that important? Similarly, does the fact that some MOH patients continue with CDH after cessation of medication overuse warrant a general de-emphasis? Certainly not for the third-to-half of patients who benefit from limiting medication use.
I suspect both Drs. Diener and Loder would agree that we would benefit from better markers of chronification and that earlier intervention with at-risk patients (eg, patients with increasing headache frequency, severity or duration but still in the episodic phase).
Jack Schim, MD
Co-Director, The Headache Center of Southern California
There has long been recognition that overuse of analgesic medications can be linked to progression of headache disorders. MOH was initially described by Dr. Lee Kudrow in 1982, in a chapter entitled, “Paradoxical effects of frequent analgesic use.”4 The most recent edition of the International Classification of Headache Disorders (ICHD-3) description does not entail features that imply causality. While there is epidemiologic observation of correlation between frequent analgesic use and progression of primary headache disorders, the causal relationship is often obscured by the facts. Overuse of acute medications is quite common in individuals with CM, but not all with CM overuse medications.
In the article being discussed, Drs. Diener and Loder reviewed facts and opinions. They helped clarify that while MOH is widely recognized, much of what is known is descriptive, and not based on solid science. From their presentations, we can conclude that we can recognize MOH based on ICHD-3 criteria, but we cannot tell an individual with chronic headache whether we can best help them by educating them, or by adjusting preventives, or both. The call to action is clear; we need to evaluate best therapeutic approaches in an empiric fashion. Our best new therapies for migraine prevention, CGRP mAbs, work for the majority of patients, with minimal side effects, even in the face of what has been considered MOH. Now, we need to strategize how best to approach these clinically challenged individuals. We need to avoid further stigmatizing our patients. Let’s recognize that our patients do not fail preventives, the prior preventives have failed our patients. Can the introduction of highly effective, well tolerated preventives at an earlier stage help avoid chronification that may drive medication overuse?
Stewart J. Tepper, MD, FAHS
Professor of Neurology, Geisel School of Medicine at Dartmouth
It is clear that overuse of some acute medication is detrimental to patient health. Examples of this include analgesic nephropathy or peptic ulcer disease, and exacerbation of depression with overuse of barbiturate compounds or benzodiazepines. Few doubt the health merits of reduction of acute medication overuse, regardless of whether the acute medications can be proven to transform episodic migraine (EM) to CM.
The good news is that the issues of the existence of true MOH and its proper management are rapidly becoming less important. OnabotulinumtoxinA use decreases triptan use in multiple randomized controlled trials for CM prevention.
Each of the anti-CGRP and anti-CGRP receptor monoclonal antibodies (mAbs) have been effective in preventing CM with medication overuse. All have lowered acute medication use, both triptans and analgesics. It is worth noting, however, that in both the OnabotulinumtoxinA and mAb trials, over-users of opioids and barbiturates were excluded. The mAbs converted patients from acute medication overuse to non-overuse, and from CM with medication overuse to EM without medication. These changes occurred without specific plans for weaning acute medication in place.
Accordingly, patients with CM with acute medication overuse should be treated with optimal prevention, and the evidence is strongest for use of the mAbs to both reduce mean monthly migraine days and all acute medication use, both triptans and analgesics. The new monoclonal antibody effectiveness may make the old arguments moot.
Hans-Christoph Diener, MD, PhD
University of Essen, Germany
I think no one doubts that MOH exists. The worldwide prevalence is between 1% and 2% (Table). The dilemma is that the diagnosis can only be made after the intake of acute medication has been reduced. There are confounders: migraine can improve irrespective of the reduction of acute medication and many physicians will implement migraine prevention at the time of withdrawal. No randomized trial compared the continuation of unchanged intake of medication to treat migraine attacks with reduction or withdrawal.
| Author (year) (reference) | Country | Age Group | Prevalence of MOH |
Castillo et al. (1999)7 | Spain | ≥ 14 | 1,2% |
Wang et al. (2000)8 | Taiwan | ≥ 65 | 1,0% |
Lu et al. (2001)9 | Taiwan | ≥ 15 | 1,1% |
Pascual et al. (2001)10 | Review |
| 1,0-1,9% |
Prencipe et al. (2001)11 | France | ≥ 65 | 1,7% |
Colas et al. (2004)12 | Spain | ≥ 14 | 1,5% |
Zwart et al. (2004)13 | Norway | ≥ 20 | 0,9-1,0% |
Dyb et al. (2006)14 | Norway | 13-18 | 0,2% |
Wang et al. (2006)15 | Taiwan | 12-14 | 0,3% |
Wiendels (2006)16 | Netherlands | 25-55 | 2,6% |
Stovner et al.(2007)17 | Review |
| 0,5-1,0% |
Aaseth et al. (2008, 2009)18,19 | Norway | 30-44 | 1,7% |
Rueda-Sanchez & Diaz-Martinez (2008)20 | Columbia | 18-65 | 4,5% |
Katsarava et al. (2009)21 | Georgia | ≥ 16 | 0,9% |
Da Silva et al. (2009)22 | Brazil | 10-93 | 1,6% |
Straube et al. (2010)23 | Germany | 18-88 | 1,0% |
Jonsson et al. (2011, 2012)24,25 | Sweden | ≥ 15 | 1,8% |
Linde et al. (2011)26 | Norway | ≥ 20 | 1,0% |
Lipton et al. (2011)27 | USA | 12-17 | 1,0% |
Ayzenberg et al. (2012)28 | Russia | 18-65 | 7,2% |
Ertas et al. (2012)29 | Turkey | 18-65 | 2,1% |
Hagen et al. (2012)30 | Norway | ≥ 20 | 0,8% |
Yu et al. (2012)31 | China | 18-65 | 0,9% |
Shahbeigi et al. (2013)32 | Iran | ≥ 10 | 4,9% |
Schramm et al. (2013)33 | Germany | 18-65 | 0,7% |
Park et al. (2014)34 | South Korea | 19-69 | 0,5% |
Kristoffersen & Lundqvist (2014)35 | Multinational summary |
| 1,0-2,0% |
Steiner (2014)36 | Multinational summary |
| 1,0-2,0% |
Westergaard et al. (2015)37 | Denmark |
| 0,5-7,2% |
Bravo (2015)38 | Multinational | Older | 1,0-7,1% |
Mbewe et al. (2015)39 | Zambia | 18-65 | 12,7% (adj. 7,1%) |
Kulkarni et al. (2015)40 | India | 18-65 | 1,2% |
Westergaard et al. (2016)41 | Denmark | ≥ 16 | 1,6% (adj. 1,8%) |
Manandhar et al. (2016)42 | Nepal | 18-65 | 2,2% |
Zebenigus et al. (2016)43 | Ethiopia | 18-65 | 0,8% (adj. 0,7%) |
Al-Hashel et al. (2017)44 | Kuwait | 18-65 | 2,4% |
Rastenyte et al. (2017)45 | Lithuania | 18-65 | 3,5% (adj. 3,2%) |
Henning et al. (2018)46 | Germany | 18-65 | 0,7% |
Global Burden of Disease 201747 | Global |
| 0,8% |
+++
Commentary by Alan M. Rapoport, MD
The above comments by my associates are very informative and help the reader to better understand the arguments about MOH. When Dr. Lee Kudrow taught me and my partner Dr. Fred Sheftell about the entity of analgesic and ergotamine overuse headaches in 1979, we set out to find those patients, observe and treat them. We did not have to wait long as so many patients with frequent and severe headaches came to see us with what we now term “medication overuse headache.” They told us that they had fewer headaches several years before and increased the use of acute care medications as their headaches increased in frequency. They were unaware of the probability that their headaches increased in frequency because their medication did. Some would argue that they increased their intake to feel better as the headache increased on their own.
Of course, we were not sure of the cause and effect, but we saw the result of tapering the acute care medications, whether or not we used preventives, hospitalized those patients or treated them with behavioral medicine approaches, etc. We observed that the combination of these treatments seemed to work better than just detox, but we did not do the proper studies to prove it. We also noticed that about 30% to 40% of patients did not improve as well as others, and daily or near-daily headaches continued, often of a lesser intensity. Almost all felt better in general and had fewer adverse events from the medication. The decrease in those medications was undoubtedly better for their brain function, livers and kidneys.
I do believe that medication overuse makes most patients with frequent EM or CM worse and we should educate patients to avoid it. I agree with Dr. Bigal that preventive medications may help some patients to improve despite the excessive use of acute care medication, but I am not sure that the older preventives work as well and certainly not as quickly as the newer ones. Recently I have seen the anti-CGRP mAbs work wonders with some of my patients who could not decrease their triptan intake. They just stop using the triptans as their headaches decrease on these therapies.
There is another interesting phenomenon that I have seen in practice—mostly with butalbital products, which I no longer prescribe. Forty years ago, patients would say that they only had 4 headaches per month lasting 1 to 2 days and I would prescribe 10 pills for them. They would call in 2 weeks and say they needed more. When queried they would invariably say it worked so well on the bad headaches and made them feel so much better, that they took 1 or 2 on days they thought they were going to get a headache and it prevented them from forming. They were soon taking it frequently and over time they were dependent on the medication, and then it stopped working and was difficult to withdraw.
Dr. Loder’s point that the studies on MOH have not proven that medication overuse causes it may be technically true; but it would be unethical to start patients on too much medication and randomize some to stay on and some to taper off. Dr. Kudrow came the closest by taking existing MOH patients and treating half with withdrawal and half of each with preventives. His breakthrough study in 1982 “proved” the existence of analgesic rebound and directed us to the best treatment at that time.4 This was the first study to examine the effect of stopping the overuse of medication to see the results.
Finally, I am unhappy that ICHD-3 has defined MOH only by number of days of medication use, not at all considering whether or not the patient develops a new type of headache, a worse type or more frequent headache (the way it was in previous versions). We have all seen patients taking 3 triptans per day on their own or with our suggestion, and many actually do better with no headache, for a period of time. This is medication overuse by definition, but not MOH, as they have little headache. But we do not recognize this entity.
The good news is, with education and anti-CGRP therapies, we will probably see less MOH in the future, or at least know better how to treat it.
+++
References
1. Bigal ME, Serrano D, Buse D, et al. Acute migraine medications and evolution from episodic to chronic migraine: a longitudinal population-based study. Headache. 2008;48(8):1157-68. doi: 10.1111/j.1526-4610.2008.01217.x. PubMed PMID: 18808500.
2. Bigal ME, Lipton RB. Overuse of acute migraine medications and migraine chronification. Curr Pain Headache Rep. 2009;13(4):301-7. PubMed PMID: 19586594.
3. Bigal ME, Edvinsson L, Rapoport AM, et al. Safety, tolerability, and efficacy of TEV-48125 for preventive treatment of chronic migraine: a multicentre, randomised, double-blind, placebo-controlled, phase 2b study. Lancet Neurol. 2015;14(11):1091-100. doi: 10.1016/S1474-4422(15)00245-8. PubMed PMID: 26432181.
4. Kudrow L. Paradoxical Effects of Frequent Analgesic Use. Advances in Neurology. 1982;33:335-341.
5. Diener HC, Holle D, Dresler T, Gaul C. Chronic Headache Due to Overuse of Analgesics and Anti-Migraine Agents. Dtsch Arztebl Int. 2018;115(22):365-370.
6. Westergaard ML, Glumer C, Hansen EH, Jensen RH. Prevalence of chronic headache with and without medication overuse: associations with socioeconomic position and physical and mental health status. Pain. 2014;155(10):2005-2013.
7. Castillo J, Munoz P, Guitera V, Pascual J. Epidemiology of chronic daily headache in the general population. Headache. 1999;39:190-196.
8. Wang SJ, Fuh JL, Lu SR, et al. Chronic daily headache in chinese elderly - prevalence, risk factors, and biannual follow-up. Neurology. 2000;54:314-319.
9. Lu SR, Fuh JL, Chen WT, Juang KD, Wang SJ. Chronic daily headache in Taipei, Taiwan: prevalence, follow-up and outcome predictors. Cephalalgia. 2001;21:980-986.
10. Pascual J, Colas R, Castillo J. Epidemiology of chronic daily headache. Curr Pain Headache Rep. 2001;5(6):529-536.
11. Prencipe M, Casini AR, Ferretti C, et al. Prevalence of headache in an elderly population: attack frequency, disability, and use of medication. J Neurol Neurosurg Psychiatry. 2001;70(3):377-381.
12. Colas R, Munoz P, Temprano R, Gomez C, Pascual J. Chronic daily headache with analgesic overuse: epidemiology and impact on quality of life. Neurology. 2004;62:1338-1342.
13. Zwart J, Dyb G, Hagen K, Svebak S, Stovner L, Holmen J. Analgesic overuse among subjects with headache, neck, and low back pain. Neurology. 2004;62:1540-1544.
14. Dyb G, Holmen TL, Zwart JA. Analgesic overuse among adolescents with headache: the Head-HUNT-Youth Study. Neurology. 2006;66(2):198-201.
15. Wang SJ, Fuh JL, Lu SR, Juang KD. Chronic daily headache in adolescents: prevalence, impact, and medication overuse. Neurology. 2006;66(2):193-197.
16. Wiendels NJ, Knuistingh NA, Rosendaal FR, et al. Chronic frequent headache in the general population: prevalence and associated factors. Cephalalgia. 2006;26(12):1434-1442.
17. Stovner L, Hagen K, Jensen R, et al. The global burden of headache: a documentation of headache prevalence and disability worldwide. Cephalalgia. 2007;27(3):193-210.
18. Aaseth K, Grande RB, Kvaerner KJ, Gulbrandsen P, Lundqvist C, Russell MB. Prevalence of secondary chronic headaches in a population-based sample of 30-44-year-old persons. The Akershus study of chronic headache. Cephalalgia. 2008;28(7):705-713.
19. Aaseth K, Grande RB, Lundqvist C, Russell MB. What is chronic headache in the general population? The Akershus study of chronic headache. Acta Neurol Scand Suppl. 2009;(189):30-32.
20. Rueda-Sanchez M, Diaz-Martinez LA. Prevalence and associated factors for episodic and chronic daily headache in the Colombian population. Cephalalgia. 2008;28(3):216-225.
21. Katsarava Z, Dzagnidze A, Kukava M, et al. Primary headache disorders in the Republic of Georgia: prevalence and risk factors. Neurology. 2009;73(21):1796-1803.
22. da Silva A, Jr., Costa EC, Gomes JB, et al. Chronic headache and comorbidities: a two-phase, population-based, cross-sectional study. Headache. 2010;50(8):1306-1312.
23. Straube A, Pfaffenrath V, Ladwig KH, et al. Prevalence of chronic migraine and medication overuse headache in Germany--the German DMKG headache study. Cephalalgia. 2010;30(2):207-213.
24. Jonsson P, Hedenrud T, Linde M. Epidemiology of medication overuse headache in the general Swedish population. Cephalalgia. 2011;31(9):1015-1022.
25. Jonsson P, Linde M, Hensing G, Hedenrud T. Sociodemographic differences in medication use, health-care contacts and sickness absence among individuals with medication-overuse headache. J Headache Pain. 2012;13(4):281-290.
26. Linde M, Stovner LJ, Zwart JA, Hagen K. Time trends in the prevalence of headache disorders. The Nord-Trondelag Health Studies (HUNT 2 and HUNT 3). Cephalalgia. 2011;31(5):585-596.
27. Lipton RB, Manack A, Ricci JA, Chee E, Turkel CC, Winner P. Prevalence and burden of chronic migraine in adolescents: results of the chronic daily headache in adolescents study (C-dAS). Headache. 2011;51(5):693-706.
28. Ayzenberg I, Katsarava Z, Sborowski A, et al. The prevalence of primary headache disorders in Russia: a countrywide survey. Cephalalgia. 2012;32(5):373-381.
29. Ertas M, Baykan B, Orhan EK, et al. One-year prevalence and the impact of migraine and tension-type headache in Turkey: a nationwide home-based study in adults. J Headache Pain. 2012;13(2):147-157.
30. Hagen K, Linde M, Steiner TJ, Stovner LJ, Zwart JA. Risk factors for medication-overuse headache: an 11-year follow-up study. The Nord-Trondelag Health Studies. Pain. 2012;153(1):56-61.
31. Yu S, Liu R, Zhao G, et al. The prevalence and burden of primary headaches in China: a population-based door-to-door survey. Headache. 2012;52(4):582-591.
32. Shahbeigi S, Fereshtehnejad SM, Mohammadi N, et al. Epidemiology of headaches in Tehran urban area: a population-based cross-sectional study in district 8, year 2010. Neurol Sci. 2013;34(7):1157-66.
33. Schramm SH, Obermann M, Katsarava Z, Diener HC, Moebus S, Yoon MS. Epidemiological profiles of patients with chronic migraine and chronic tension-type headache. J Headache Pain. 2013;14:40.
34. Park JW, Moon HS, Kim JM, Lee KS, Chu MK. Chronic daily headache in Korea: prevalence, clinical characteristics, medical consultation and management. J Clin Neurol. 2014;10(3):236-243.
35. Kristoffersen ES, Lundqvist C. Medication-overuse headache: epidemiology, diagnosis and treatment. Ther Adv Drug Saf. 2014;5(2):87-99.
36. Steiner TJ, Stovner LJ, Katsarava Z, et al. The impact of headache in Europe: principal results of the Eurolight project. J Headache Pain. 2014;15:31.
37. Westergaard ML, Hansen EH, Glumer C, Jensen RH. Prescription pain medications and chronic headache in Denmark: implications for preventing medication overuse. Eur J Clin Pharmacol. 2015;71(7):851-860.
38. Bravo TP. Headaches of the elderly. Curr Neurol Neurosci Rep. 2015;15(6):30.
39. Mbewe E, Zairemthiama P, Yeh HH, Paul R, Birbeck GL, Steiner TJ. The epidemiology of primary headache disorders in Zambia: a population-based door-to-door survey. J Headache Pain. 2015;16:515.
40. Kulkarni GB, Rao GN, Gururaj G, Stovner LJ, Steiner TJ. Headache disorders and public ill-health in India: prevalence estimates in Karnataka State. J Headache Pain. 2015;16:67.
41. Westergaard ML, Glumer C, Hansen EH, Jensen RH. Medication overuse, healthy lifestyle behaviour and stress in chronic headache: Results from a population-based representative survey. Cephalalgia. 2016;36(1):15-28.
42. Manandhar K, Risal A, Linde M, Steiner TJ. The burden of headache disorders in Nepal: estimates from a population-based survey. J Headache Pain. 2015;17:3.
43. Zebenigus M, Tekle-Haimanot R, Worku DK, Thomas H, Steiner TJ. The prevalence of primary headache disorders in Ethiopia. J Headache Pain. 2016;17(1):110.
44. Al-Hashel JY, Ahmed SF, Alroughani R. Prevalence of Primary Headache Disorders in Kuwait. Neuroepidemiology. 2017;48(3-4):138-146.
45. Rastenyte D, Mickeviciene D, Stovner LJ, Thomas H, Andree C, Steiner TJ. Prevalence and burden of headache disorders in Lithuania and their public-health and policy implications: a population-based study within the Eurolight Project. J Headache Pain. 2017;18(1):53.
46. Henning V, Katsarava Z, Obermann M, Moebus S, Schramm S. Remission of chronic headache: Rates, potential predictors and the role of medication, follow-up results of the German Headache Consortium (GHC) Study. Cephalalgia. 2018;38(3):551-560.
47. Global Burden of Disease Neurological Disorders Collaborator Group. Global, regional, and national burden of neurological disorders during 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Neurol. 2017;16(11):877-897.
Following the American Headache Society’s Scottsdale Headache Symposium in November 2018, MedPage Today posted an article which shared differing opinions from Drs. Hans-Christoph Diener and Elizabeth Loder on medication overuse headache (MOH). While Dr. Diener noted that “we can identify people with chronic migraine who are at risk to have medication overuse,” and that following successful withdrawal treatment “the majority of patients…revert to episodic migraine,” Dr. Loder pointed out that while MOH may exist and contribute to chronic migraine, “it is over-emphasized and the evidence in support of these concepts is weak.”
For this article, I’ve asked Drs. Marcelo Bigal, Rob Cowan, Jack Schim, and Stewart J. Tepper to share their perspectives on this topic. I also asked both Dr. Diener and Dr. Loder to expand on their comments. We will see Dr. Diener’s response to the article, but we did not hear back from Dr. Loder. Lastly, I will weigh in on the MOH discussion.
Marcelo Bigal, MD, PhD
Chief Medical Officer, Purdue Pharma
The issue of medication overuse headache (MOH) needs to be disentangled into a few separate but related issues. First, do excessive medications make migraine worse? Second, should MOH be considered a distinct form of headache? And how can evidence inform clinical practice?
Robust evidence supports the fact that excessive acute medication use is associated with increased headache frequency among migraineurs. In a large epidemiological study, we demonstrated that exposure (medication) precedes outcome (increased headache frequency).1 The risk was higher for barbiturates, followed by opioids and triptans, and was not increased by nonsteroidal anti-inflammatory drugs (NSAIDs). Dose response and critical exposure levels were identified. Based on this study and several others, we argued that criteria of causality had been demonstrated beyond reasonable doubt.2
However, since the effect is specific to migraine in that the exposure only increases the risk in migraineurs, not in individuals with other types of pain, we do not consider MOH a distinct entity. Instead, we believe that excessive medication is a risk factor for chronic migraine (CM). Therefore, we should be able to subdivide CM into 2 groups, one with and one without excessive medication use.
From a clinical perspective, physicians should monitor acute medication consumption in individuals with migraine and should be liberal in starting preventive therapy. In those with CM and excessive acute medication use we don’t advocate abrupt discontinuation of acute medications, since some preventive medications, especially the newer anti-calcitonin gene-related peptide (CGRP) antibodies, seem to work equally well in individuals with and without excessive use of medication,3 allowing more natural and gradual control of acute medication consumption without the need for detoxification.
Rob Cowan, MD, FAAN, FAHS
Higgins Professor of Neurology and Neurosciences
Chief, Division of Headache Medicine, Dept. of Neurology and Neurosciences
Director, Stanford University School of Medicine
As is often the case when 2 smart people take artificially imposed opposite positions, the truth will lie somewhere in the middle. I doubt either of the debaters would argue either extreme position: MOH does not exist, or MOH when present is solely responsible for chronic daily headache (CDH). The argument that the absence of controlled studies negates the proposition fails the common-sense test: Without a controlled study, we can’t be sure that wearing a helmet when bicycling is better than not. In such a case, observational data is sufficient. Could there be confounders (eg, helmet wearers are more inclined to ride safely)? Of course, but is that important? Similarly, does the fact that some MOH patients continue with CDH after cessation of medication overuse warrant a general de-emphasis? Certainly not for the third-to-half of patients who benefit from limiting medication use.
I suspect both Drs. Diener and Loder would agree that we would benefit from better markers of chronification and that earlier intervention with at-risk patients (eg, patients with increasing headache frequency, severity or duration but still in the episodic phase).
Jack Schim, MD
Co-Director, The Headache Center of Southern California
There has long been recognition that overuse of analgesic medications can be linked to progression of headache disorders. MOH was initially described by Dr. Lee Kudrow in 1982, in a chapter entitled, “Paradoxical effects of frequent analgesic use.”4 The most recent edition of the International Classification of Headache Disorders (ICHD-3) description does not entail features that imply causality. While there is epidemiologic observation of correlation between frequent analgesic use and progression of primary headache disorders, the causal relationship is often obscured by the facts. Overuse of acute medications is quite common in individuals with CM, but not all with CM overuse medications.
In the article being discussed, Drs. Diener and Loder reviewed facts and opinions. They helped clarify that while MOH is widely recognized, much of what is known is descriptive, and not based on solid science. From their presentations, we can conclude that we can recognize MOH based on ICHD-3 criteria, but we cannot tell an individual with chronic headache whether we can best help them by educating them, or by adjusting preventives, or both. The call to action is clear; we need to evaluate best therapeutic approaches in an empiric fashion. Our best new therapies for migraine prevention, CGRP mAbs, work for the majority of patients, with minimal side effects, even in the face of what has been considered MOH. Now, we need to strategize how best to approach these clinically challenged individuals. We need to avoid further stigmatizing our patients. Let’s recognize that our patients do not fail preventives, the prior preventives have failed our patients. Can the introduction of highly effective, well tolerated preventives at an earlier stage help avoid chronification that may drive medication overuse?
Stewart J. Tepper, MD, FAHS
Professor of Neurology, Geisel School of Medicine at Dartmouth
It is clear that overuse of some acute medication is detrimental to patient health. Examples of this include analgesic nephropathy or peptic ulcer disease, and exacerbation of depression with overuse of barbiturate compounds or benzodiazepines. Few doubt the health merits of reduction of acute medication overuse, regardless of whether the acute medications can be proven to transform episodic migraine (EM) to CM.
The good news is that the issues of the existence of true MOH and its proper management are rapidly becoming less important. OnabotulinumtoxinA use decreases triptan use in multiple randomized controlled trials for CM prevention.
Each of the anti-CGRP and anti-CGRP receptor monoclonal antibodies (mAbs) have been effective in preventing CM with medication overuse. All have lowered acute medication use, both triptans and analgesics. It is worth noting, however, that in both the OnabotulinumtoxinA and mAb trials, over-users of opioids and barbiturates were excluded. The mAbs converted patients from acute medication overuse to non-overuse, and from CM with medication overuse to EM without medication. These changes occurred without specific plans for weaning acute medication in place.
Accordingly, patients with CM with acute medication overuse should be treated with optimal prevention, and the evidence is strongest for use of the mAbs to both reduce mean monthly migraine days and all acute medication use, both triptans and analgesics. The new monoclonal antibody effectiveness may make the old arguments moot.
Hans-Christoph Diener, MD, PhD
University of Essen, Germany
I think no one doubts that MOH exists. The worldwide prevalence is between 1% and 2% (Table). The dilemma is that the diagnosis can only be made after the intake of acute medication has been reduced. There are confounders: migraine can improve irrespective of the reduction of acute medication and many physicians will implement migraine prevention at the time of withdrawal. No randomized trial compared the continuation of unchanged intake of medication to treat migraine attacks with reduction or withdrawal.
| Author (year) (reference) | Country | Age Group | Prevalence of MOH |
Castillo et al. (1999)7 | Spain | ≥ 14 | 1,2% |
Wang et al. (2000)8 | Taiwan | ≥ 65 | 1,0% |
Lu et al. (2001)9 | Taiwan | ≥ 15 | 1,1% |
Pascual et al. (2001)10 | Review |
| 1,0-1,9% |
Prencipe et al. (2001)11 | France | ≥ 65 | 1,7% |
Colas et al. (2004)12 | Spain | ≥ 14 | 1,5% |
Zwart et al. (2004)13 | Norway | ≥ 20 | 0,9-1,0% |
Dyb et al. (2006)14 | Norway | 13-18 | 0,2% |
Wang et al. (2006)15 | Taiwan | 12-14 | 0,3% |
Wiendels (2006)16 | Netherlands | 25-55 | 2,6% |
Stovner et al.(2007)17 | Review |
| 0,5-1,0% |
Aaseth et al. (2008, 2009)18,19 | Norway | 30-44 | 1,7% |
Rueda-Sanchez & Diaz-Martinez (2008)20 | Columbia | 18-65 | 4,5% |
Katsarava et al. (2009)21 | Georgia | ≥ 16 | 0,9% |
Da Silva et al. (2009)22 | Brazil | 10-93 | 1,6% |
Straube et al. (2010)23 | Germany | 18-88 | 1,0% |
Jonsson et al. (2011, 2012)24,25 | Sweden | ≥ 15 | 1,8% |
Linde et al. (2011)26 | Norway | ≥ 20 | 1,0% |
Lipton et al. (2011)27 | USA | 12-17 | 1,0% |
Ayzenberg et al. (2012)28 | Russia | 18-65 | 7,2% |
Ertas et al. (2012)29 | Turkey | 18-65 | 2,1% |
Hagen et al. (2012)30 | Norway | ≥ 20 | 0,8% |
Yu et al. (2012)31 | China | 18-65 | 0,9% |
Shahbeigi et al. (2013)32 | Iran | ≥ 10 | 4,9% |
Schramm et al. (2013)33 | Germany | 18-65 | 0,7% |
Park et al. (2014)34 | South Korea | 19-69 | 0,5% |
Kristoffersen & Lundqvist (2014)35 | Multinational summary |
| 1,0-2,0% |
Steiner (2014)36 | Multinational summary |
| 1,0-2,0% |
Westergaard et al. (2015)37 | Denmark |
| 0,5-7,2% |
Bravo (2015)38 | Multinational | Older | 1,0-7,1% |
Mbewe et al. (2015)39 | Zambia | 18-65 | 12,7% (adj. 7,1%) |
Kulkarni et al. (2015)40 | India | 18-65 | 1,2% |
Westergaard et al. (2016)41 | Denmark | ≥ 16 | 1,6% (adj. 1,8%) |
Manandhar et al. (2016)42 | Nepal | 18-65 | 2,2% |
Zebenigus et al. (2016)43 | Ethiopia | 18-65 | 0,8% (adj. 0,7%) |
Al-Hashel et al. (2017)44 | Kuwait | 18-65 | 2,4% |
Rastenyte et al. (2017)45 | Lithuania | 18-65 | 3,5% (adj. 3,2%) |
Henning et al. (2018)46 | Germany | 18-65 | 0,7% |
Global Burden of Disease 201747 | Global |
| 0,8% |
+++
Commentary by Alan M. Rapoport, MD
The above comments by my associates are very informative and help the reader to better understand the arguments about MOH. When Dr. Lee Kudrow taught me and my partner Dr. Fred Sheftell about the entity of analgesic and ergotamine overuse headaches in 1979, we set out to find those patients, observe and treat them. We did not have to wait long as so many patients with frequent and severe headaches came to see us with what we now term “medication overuse headache.” They told us that they had fewer headaches several years before and increased the use of acute care medications as their headaches increased in frequency. They were unaware of the probability that their headaches increased in frequency because their medication did. Some would argue that they increased their intake to feel better as the headache increased on their own.
Of course, we were not sure of the cause and effect, but we saw the result of tapering the acute care medications, whether or not we used preventives, hospitalized those patients or treated them with behavioral medicine approaches, etc. We observed that the combination of these treatments seemed to work better than just detox, but we did not do the proper studies to prove it. We also noticed that about 30% to 40% of patients did not improve as well as others, and daily or near-daily headaches continued, often of a lesser intensity. Almost all felt better in general and had fewer adverse events from the medication. The decrease in those medications was undoubtedly better for their brain function, livers and kidneys.
I do believe that medication overuse makes most patients with frequent EM or CM worse and we should educate patients to avoid it. I agree with Dr. Bigal that preventive medications may help some patients to improve despite the excessive use of acute care medication, but I am not sure that the older preventives work as well and certainly not as quickly as the newer ones. Recently I have seen the anti-CGRP mAbs work wonders with some of my patients who could not decrease their triptan intake. They just stop using the triptans as their headaches decrease on these therapies.
There is another interesting phenomenon that I have seen in practice—mostly with butalbital products, which I no longer prescribe. Forty years ago, patients would say that they only had 4 headaches per month lasting 1 to 2 days and I would prescribe 10 pills for them. They would call in 2 weeks and say they needed more. When queried they would invariably say it worked so well on the bad headaches and made them feel so much better, that they took 1 or 2 on days they thought they were going to get a headache and it prevented them from forming. They were soon taking it frequently and over time they were dependent on the medication, and then it stopped working and was difficult to withdraw.
Dr. Loder’s point that the studies on MOH have not proven that medication overuse causes it may be technically true; but it would be unethical to start patients on too much medication and randomize some to stay on and some to taper off. Dr. Kudrow came the closest by taking existing MOH patients and treating half with withdrawal and half of each with preventives. His breakthrough study in 1982 “proved” the existence of analgesic rebound and directed us to the best treatment at that time.4 This was the first study to examine the effect of stopping the overuse of medication to see the results.
Finally, I am unhappy that ICHD-3 has defined MOH only by number of days of medication use, not at all considering whether or not the patient develops a new type of headache, a worse type or more frequent headache (the way it was in previous versions). We have all seen patients taking 3 triptans per day on their own or with our suggestion, and many actually do better with no headache, for a period of time. This is medication overuse by definition, but not MOH, as they have little headache. But we do not recognize this entity.
The good news is, with education and anti-CGRP therapies, we will probably see less MOH in the future, or at least know better how to treat it.
+++
References
1. Bigal ME, Serrano D, Buse D, et al. Acute migraine medications and evolution from episodic to chronic migraine: a longitudinal population-based study. Headache. 2008;48(8):1157-68. doi: 10.1111/j.1526-4610.2008.01217.x. PubMed PMID: 18808500.
2. Bigal ME, Lipton RB. Overuse of acute migraine medications and migraine chronification. Curr Pain Headache Rep. 2009;13(4):301-7. PubMed PMID: 19586594.
3. Bigal ME, Edvinsson L, Rapoport AM, et al. Safety, tolerability, and efficacy of TEV-48125 for preventive treatment of chronic migraine: a multicentre, randomised, double-blind, placebo-controlled, phase 2b study. Lancet Neurol. 2015;14(11):1091-100. doi: 10.1016/S1474-4422(15)00245-8. PubMed PMID: 26432181.
4. Kudrow L. Paradoxical Effects of Frequent Analgesic Use. Advances in Neurology. 1982;33:335-341.
5. Diener HC, Holle D, Dresler T, Gaul C. Chronic Headache Due to Overuse of Analgesics and Anti-Migraine Agents. Dtsch Arztebl Int. 2018;115(22):365-370.
6. Westergaard ML, Glumer C, Hansen EH, Jensen RH. Prevalence of chronic headache with and without medication overuse: associations with socioeconomic position and physical and mental health status. Pain. 2014;155(10):2005-2013.
7. Castillo J, Munoz P, Guitera V, Pascual J. Epidemiology of chronic daily headache in the general population. Headache. 1999;39:190-196.
8. Wang SJ, Fuh JL, Lu SR, et al. Chronic daily headache in chinese elderly - prevalence, risk factors, and biannual follow-up. Neurology. 2000;54:314-319.
9. Lu SR, Fuh JL, Chen WT, Juang KD, Wang SJ. Chronic daily headache in Taipei, Taiwan: prevalence, follow-up and outcome predictors. Cephalalgia. 2001;21:980-986.
10. Pascual J, Colas R, Castillo J. Epidemiology of chronic daily headache. Curr Pain Headache Rep. 2001;5(6):529-536.
11. Prencipe M, Casini AR, Ferretti C, et al. Prevalence of headache in an elderly population: attack frequency, disability, and use of medication. J Neurol Neurosurg Psychiatry. 2001;70(3):377-381.
12. Colas R, Munoz P, Temprano R, Gomez C, Pascual J. Chronic daily headache with analgesic overuse: epidemiology and impact on quality of life. Neurology. 2004;62:1338-1342.
13. Zwart J, Dyb G, Hagen K, Svebak S, Stovner L, Holmen J. Analgesic overuse among subjects with headache, neck, and low back pain. Neurology. 2004;62:1540-1544.
14. Dyb G, Holmen TL, Zwart JA. Analgesic overuse among adolescents with headache: the Head-HUNT-Youth Study. Neurology. 2006;66(2):198-201.
15. Wang SJ, Fuh JL, Lu SR, Juang KD. Chronic daily headache in adolescents: prevalence, impact, and medication overuse. Neurology. 2006;66(2):193-197.
16. Wiendels NJ, Knuistingh NA, Rosendaal FR, et al. Chronic frequent headache in the general population: prevalence and associated factors. Cephalalgia. 2006;26(12):1434-1442.
17. Stovner L, Hagen K, Jensen R, et al. The global burden of headache: a documentation of headache prevalence and disability worldwide. Cephalalgia. 2007;27(3):193-210.
18. Aaseth K, Grande RB, Kvaerner KJ, Gulbrandsen P, Lundqvist C, Russell MB. Prevalence of secondary chronic headaches in a population-based sample of 30-44-year-old persons. The Akershus study of chronic headache. Cephalalgia. 2008;28(7):705-713.
19. Aaseth K, Grande RB, Lundqvist C, Russell MB. What is chronic headache in the general population? The Akershus study of chronic headache. Acta Neurol Scand Suppl. 2009;(189):30-32.
20. Rueda-Sanchez M, Diaz-Martinez LA. Prevalence and associated factors for episodic and chronic daily headache in the Colombian population. Cephalalgia. 2008;28(3):216-225.
21. Katsarava Z, Dzagnidze A, Kukava M, et al. Primary headache disorders in the Republic of Georgia: prevalence and risk factors. Neurology. 2009;73(21):1796-1803.
22. da Silva A, Jr., Costa EC, Gomes JB, et al. Chronic headache and comorbidities: a two-phase, population-based, cross-sectional study. Headache. 2010;50(8):1306-1312.
23. Straube A, Pfaffenrath V, Ladwig KH, et al. Prevalence of chronic migraine and medication overuse headache in Germany--the German DMKG headache study. Cephalalgia. 2010;30(2):207-213.
24. Jonsson P, Hedenrud T, Linde M. Epidemiology of medication overuse headache in the general Swedish population. Cephalalgia. 2011;31(9):1015-1022.
25. Jonsson P, Linde M, Hensing G, Hedenrud T. Sociodemographic differences in medication use, health-care contacts and sickness absence among individuals with medication-overuse headache. J Headache Pain. 2012;13(4):281-290.
26. Linde M, Stovner LJ, Zwart JA, Hagen K. Time trends in the prevalence of headache disorders. The Nord-Trondelag Health Studies (HUNT 2 and HUNT 3). Cephalalgia. 2011;31(5):585-596.
27. Lipton RB, Manack A, Ricci JA, Chee E, Turkel CC, Winner P. Prevalence and burden of chronic migraine in adolescents: results of the chronic daily headache in adolescents study (C-dAS). Headache. 2011;51(5):693-706.
28. Ayzenberg I, Katsarava Z, Sborowski A, et al. The prevalence of primary headache disorders in Russia: a countrywide survey. Cephalalgia. 2012;32(5):373-381.
29. Ertas M, Baykan B, Orhan EK, et al. One-year prevalence and the impact of migraine and tension-type headache in Turkey: a nationwide home-based study in adults. J Headache Pain. 2012;13(2):147-157.
30. Hagen K, Linde M, Steiner TJ, Stovner LJ, Zwart JA. Risk factors for medication-overuse headache: an 11-year follow-up study. The Nord-Trondelag Health Studies. Pain. 2012;153(1):56-61.
31. Yu S, Liu R, Zhao G, et al. The prevalence and burden of primary headaches in China: a population-based door-to-door survey. Headache. 2012;52(4):582-591.
32. Shahbeigi S, Fereshtehnejad SM, Mohammadi N, et al. Epidemiology of headaches in Tehran urban area: a population-based cross-sectional study in district 8, year 2010. Neurol Sci. 2013;34(7):1157-66.
33. Schramm SH, Obermann M, Katsarava Z, Diener HC, Moebus S, Yoon MS. Epidemiological profiles of patients with chronic migraine and chronic tension-type headache. J Headache Pain. 2013;14:40.
34. Park JW, Moon HS, Kim JM, Lee KS, Chu MK. Chronic daily headache in Korea: prevalence, clinical characteristics, medical consultation and management. J Clin Neurol. 2014;10(3):236-243.
35. Kristoffersen ES, Lundqvist C. Medication-overuse headache: epidemiology, diagnosis and treatment. Ther Adv Drug Saf. 2014;5(2):87-99.
36. Steiner TJ, Stovner LJ, Katsarava Z, et al. The impact of headache in Europe: principal results of the Eurolight project. J Headache Pain. 2014;15:31.
37. Westergaard ML, Hansen EH, Glumer C, Jensen RH. Prescription pain medications and chronic headache in Denmark: implications for preventing medication overuse. Eur J Clin Pharmacol. 2015;71(7):851-860.
38. Bravo TP. Headaches of the elderly. Curr Neurol Neurosci Rep. 2015;15(6):30.
39. Mbewe E, Zairemthiama P, Yeh HH, Paul R, Birbeck GL, Steiner TJ. The epidemiology of primary headache disorders in Zambia: a population-based door-to-door survey. J Headache Pain. 2015;16:515.
40. Kulkarni GB, Rao GN, Gururaj G, Stovner LJ, Steiner TJ. Headache disorders and public ill-health in India: prevalence estimates in Karnataka State. J Headache Pain. 2015;16:67.
41. Westergaard ML, Glumer C, Hansen EH, Jensen RH. Medication overuse, healthy lifestyle behaviour and stress in chronic headache: Results from a population-based representative survey. Cephalalgia. 2016;36(1):15-28.
42. Manandhar K, Risal A, Linde M, Steiner TJ. The burden of headache disorders in Nepal: estimates from a population-based survey. J Headache Pain. 2015;17:3.
43. Zebenigus M, Tekle-Haimanot R, Worku DK, Thomas H, Steiner TJ. The prevalence of primary headache disorders in Ethiopia. J Headache Pain. 2016;17(1):110.
44. Al-Hashel JY, Ahmed SF, Alroughani R. Prevalence of Primary Headache Disorders in Kuwait. Neuroepidemiology. 2017;48(3-4):138-146.
45. Rastenyte D, Mickeviciene D, Stovner LJ, Thomas H, Andree C, Steiner TJ. Prevalence and burden of headache disorders in Lithuania and their public-health and policy implications: a population-based study within the Eurolight Project. J Headache Pain. 2017;18(1):53.
46. Henning V, Katsarava Z, Obermann M, Moebus S, Schramm S. Remission of chronic headache: Rates, potential predictors and the role of medication, follow-up results of the German Headache Consortium (GHC) Study. Cephalalgia. 2018;38(3):551-560.
47. Global Burden of Disease Neurological Disorders Collaborator Group. Global, regional, and national burden of neurological disorders during 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Neurol. 2017;16(11):877-897.
Following the American Headache Society’s Scottsdale Headache Symposium in November 2018, MedPage Today posted an article which shared differing opinions from Drs. Hans-Christoph Diener and Elizabeth Loder on medication overuse headache (MOH). While Dr. Diener noted that “we can identify people with chronic migraine who are at risk to have medication overuse,” and that following successful withdrawal treatment “the majority of patients…revert to episodic migraine,” Dr. Loder pointed out that while MOH may exist and contribute to chronic migraine, “it is over-emphasized and the evidence in support of these concepts is weak.”
For this article, I’ve asked Drs. Marcelo Bigal, Rob Cowan, Jack Schim, and Stewart J. Tepper to share their perspectives on this topic. I also asked both Dr. Diener and Dr. Loder to expand on their comments. We will see Dr. Diener’s response to the article, but we did not hear back from Dr. Loder. Lastly, I will weigh in on the MOH discussion.
Marcelo Bigal, MD, PhD
Chief Medical Officer, Purdue Pharma
The issue of medication overuse headache (MOH) needs to be disentangled into a few separate but related issues. First, do excessive medications make migraine worse? Second, should MOH be considered a distinct form of headache? And how can evidence inform clinical practice?
Robust evidence supports the fact that excessive acute medication use is associated with increased headache frequency among migraineurs. In a large epidemiological study, we demonstrated that exposure (medication) precedes outcome (increased headache frequency).1 The risk was higher for barbiturates, followed by opioids and triptans, and was not increased by nonsteroidal anti-inflammatory drugs (NSAIDs). Dose response and critical exposure levels were identified. Based on this study and several others, we argued that criteria of causality had been demonstrated beyond reasonable doubt.2
However, since the effect is specific to migraine in that the exposure only increases the risk in migraineurs, not in individuals with other types of pain, we do not consider MOH a distinct entity. Instead, we believe that excessive medication is a risk factor for chronic migraine (CM). Therefore, we should be able to subdivide CM into 2 groups, one with and one without excessive medication use.
From a clinical perspective, physicians should monitor acute medication consumption in individuals with migraine and should be liberal in starting preventive therapy. In those with CM and excessive acute medication use we don’t advocate abrupt discontinuation of acute medications, since some preventive medications, especially the newer anti-calcitonin gene-related peptide (CGRP) antibodies, seem to work equally well in individuals with and without excessive use of medication,3 allowing more natural and gradual control of acute medication consumption without the need for detoxification.
Rob Cowan, MD, FAAN, FAHS
Higgins Professor of Neurology and Neurosciences
Chief, Division of Headache Medicine, Dept. of Neurology and Neurosciences
Director, Stanford University School of Medicine
As is often the case when 2 smart people take artificially imposed opposite positions, the truth will lie somewhere in the middle. I doubt either of the debaters would argue either extreme position: MOH does not exist, or MOH when present is solely responsible for chronic daily headache (CDH). The argument that the absence of controlled studies negates the proposition fails the common-sense test: Without a controlled study, we can’t be sure that wearing a helmet when bicycling is better than not. In such a case, observational data is sufficient. Could there be confounders (eg, helmet wearers are more inclined to ride safely)? Of course, but is that important? Similarly, does the fact that some MOH patients continue with CDH after cessation of medication overuse warrant a general de-emphasis? Certainly not for the third-to-half of patients who benefit from limiting medication use.
I suspect both Drs. Diener and Loder would agree that we would benefit from better markers of chronification and that earlier intervention with at-risk patients (eg, patients with increasing headache frequency, severity or duration but still in the episodic phase).
Jack Schim, MD
Co-Director, The Headache Center of Southern California
There has long been recognition that overuse of analgesic medications can be linked to progression of headache disorders. MOH was initially described by Dr. Lee Kudrow in 1982, in a chapter entitled, “Paradoxical effects of frequent analgesic use.”4 The most recent edition of the International Classification of Headache Disorders (ICHD-3) description does not entail features that imply causality. While there is epidemiologic observation of correlation between frequent analgesic use and progression of primary headache disorders, the causal relationship is often obscured by the facts. Overuse of acute medications is quite common in individuals with CM, but not all with CM overuse medications.
In the article being discussed, Drs. Diener and Loder reviewed facts and opinions. They helped clarify that while MOH is widely recognized, much of what is known is descriptive, and not based on solid science. From their presentations, we can conclude that we can recognize MOH based on ICHD-3 criteria, but we cannot tell an individual with chronic headache whether we can best help them by educating them, or by adjusting preventives, or both. The call to action is clear; we need to evaluate best therapeutic approaches in an empiric fashion. Our best new therapies for migraine prevention, CGRP mAbs, work for the majority of patients, with minimal side effects, even in the face of what has been considered MOH. Now, we need to strategize how best to approach these clinically challenged individuals. We need to avoid further stigmatizing our patients. Let’s recognize that our patients do not fail preventives, the prior preventives have failed our patients. Can the introduction of highly effective, well tolerated preventives at an earlier stage help avoid chronification that may drive medication overuse?
Stewart J. Tepper, MD, FAHS
Professor of Neurology, Geisel School of Medicine at Dartmouth
It is clear that overuse of some acute medication is detrimental to patient health. Examples of this include analgesic nephropathy or peptic ulcer disease, and exacerbation of depression with overuse of barbiturate compounds or benzodiazepines. Few doubt the health merits of reduction of acute medication overuse, regardless of whether the acute medications can be proven to transform episodic migraine (EM) to CM.
The good news is that the issues of the existence of true MOH and its proper management are rapidly becoming less important. OnabotulinumtoxinA use decreases triptan use in multiple randomized controlled trials for CM prevention.
Each of the anti-CGRP and anti-CGRP receptor monoclonal antibodies (mAbs) have been effective in preventing CM with medication overuse. All have lowered acute medication use, both triptans and analgesics. It is worth noting, however, that in both the OnabotulinumtoxinA and mAb trials, over-users of opioids and barbiturates were excluded. The mAbs converted patients from acute medication overuse to non-overuse, and from CM with medication overuse to EM without medication. These changes occurred without specific plans for weaning acute medication in place.
Accordingly, patients with CM with acute medication overuse should be treated with optimal prevention, and the evidence is strongest for use of the mAbs to both reduce mean monthly migraine days and all acute medication use, both triptans and analgesics. The new monoclonal antibody effectiveness may make the old arguments moot.
Hans-Christoph Diener, MD, PhD
University of Essen, Germany
I think no one doubts that MOH exists. The worldwide prevalence is between 1% and 2% (Table). The dilemma is that the diagnosis can only be made after the intake of acute medication has been reduced. There are confounders: migraine can improve irrespective of the reduction of acute medication and many physicians will implement migraine prevention at the time of withdrawal. No randomized trial compared the continuation of unchanged intake of medication to treat migraine attacks with reduction or withdrawal.
| Author (year) (reference) | Country | Age Group | Prevalence of MOH |
Castillo et al. (1999)7 | Spain | ≥ 14 | 1,2% |
Wang et al. (2000)8 | Taiwan | ≥ 65 | 1,0% |
Lu et al. (2001)9 | Taiwan | ≥ 15 | 1,1% |
Pascual et al. (2001)10 | Review |
| 1,0-1,9% |
Prencipe et al. (2001)11 | France | ≥ 65 | 1,7% |
Colas et al. (2004)12 | Spain | ≥ 14 | 1,5% |
Zwart et al. (2004)13 | Norway | ≥ 20 | 0,9-1,0% |
Dyb et al. (2006)14 | Norway | 13-18 | 0,2% |
Wang et al. (2006)15 | Taiwan | 12-14 | 0,3% |
Wiendels (2006)16 | Netherlands | 25-55 | 2,6% |
Stovner et al.(2007)17 | Review |
| 0,5-1,0% |
Aaseth et al. (2008, 2009)18,19 | Norway | 30-44 | 1,7% |
Rueda-Sanchez & Diaz-Martinez (2008)20 | Columbia | 18-65 | 4,5% |
Katsarava et al. (2009)21 | Georgia | ≥ 16 | 0,9% |
Da Silva et al. (2009)22 | Brazil | 10-93 | 1,6% |
Straube et al. (2010)23 | Germany | 18-88 | 1,0% |
Jonsson et al. (2011, 2012)24,25 | Sweden | ≥ 15 | 1,8% |
Linde et al. (2011)26 | Norway | ≥ 20 | 1,0% |
Lipton et al. (2011)27 | USA | 12-17 | 1,0% |
Ayzenberg et al. (2012)28 | Russia | 18-65 | 7,2% |
Ertas et al. (2012)29 | Turkey | 18-65 | 2,1% |
Hagen et al. (2012)30 | Norway | ≥ 20 | 0,8% |
Yu et al. (2012)31 | China | 18-65 | 0,9% |
Shahbeigi et al. (2013)32 | Iran | ≥ 10 | 4,9% |
Schramm et al. (2013)33 | Germany | 18-65 | 0,7% |
Park et al. (2014)34 | South Korea | 19-69 | 0,5% |
Kristoffersen & Lundqvist (2014)35 | Multinational summary |
| 1,0-2,0% |
Steiner (2014)36 | Multinational summary |
| 1,0-2,0% |
Westergaard et al. (2015)37 | Denmark |
| 0,5-7,2% |
Bravo (2015)38 | Multinational | Older | 1,0-7,1% |
Mbewe et al. (2015)39 | Zambia | 18-65 | 12,7% (adj. 7,1%) |
Kulkarni et al. (2015)40 | India | 18-65 | 1,2% |
Westergaard et al. (2016)41 | Denmark | ≥ 16 | 1,6% (adj. 1,8%) |
Manandhar et al. (2016)42 | Nepal | 18-65 | 2,2% |
Zebenigus et al. (2016)43 | Ethiopia | 18-65 | 0,8% (adj. 0,7%) |
Al-Hashel et al. (2017)44 | Kuwait | 18-65 | 2,4% |
Rastenyte et al. (2017)45 | Lithuania | 18-65 | 3,5% (adj. 3,2%) |
Henning et al. (2018)46 | Germany | 18-65 | 0,7% |
Global Burden of Disease 201747 | Global |
| 0,8% |
+++
Commentary by Alan M. Rapoport, MD
The above comments by my associates are very informative and help the reader to better understand the arguments about MOH. When Dr. Lee Kudrow taught me and my partner Dr. Fred Sheftell about the entity of analgesic and ergotamine overuse headaches in 1979, we set out to find those patients, observe and treat them. We did not have to wait long as so many patients with frequent and severe headaches came to see us with what we now term “medication overuse headache.” They told us that they had fewer headaches several years before and increased the use of acute care medications as their headaches increased in frequency. They were unaware of the probability that their headaches increased in frequency because their medication did. Some would argue that they increased their intake to feel better as the headache increased on their own.
Of course, we were not sure of the cause and effect, but we saw the result of tapering the acute care medications, whether or not we used preventives, hospitalized those patients or treated them with behavioral medicine approaches, etc. We observed that the combination of these treatments seemed to work better than just detox, but we did not do the proper studies to prove it. We also noticed that about 30% to 40% of patients did not improve as well as others, and daily or near-daily headaches continued, often of a lesser intensity. Almost all felt better in general and had fewer adverse events from the medication. The decrease in those medications was undoubtedly better for their brain function, livers and kidneys.
I do believe that medication overuse makes most patients with frequent EM or CM worse and we should educate patients to avoid it. I agree with Dr. Bigal that preventive medications may help some patients to improve despite the excessive use of acute care medication, but I am not sure that the older preventives work as well and certainly not as quickly as the newer ones. Recently I have seen the anti-CGRP mAbs work wonders with some of my patients who could not decrease their triptan intake. They just stop using the triptans as their headaches decrease on these therapies.
There is another interesting phenomenon that I have seen in practice—mostly with butalbital products, which I no longer prescribe. Forty years ago, patients would say that they only had 4 headaches per month lasting 1 to 2 days and I would prescribe 10 pills for them. They would call in 2 weeks and say they needed more. When queried they would invariably say it worked so well on the bad headaches and made them feel so much better, that they took 1 or 2 on days they thought they were going to get a headache and it prevented them from forming. They were soon taking it frequently and over time they were dependent on the medication, and then it stopped working and was difficult to withdraw.
Dr. Loder’s point that the studies on MOH have not proven that medication overuse causes it may be technically true; but it would be unethical to start patients on too much medication and randomize some to stay on and some to taper off. Dr. Kudrow came the closest by taking existing MOH patients and treating half with withdrawal and half of each with preventives. His breakthrough study in 1982 “proved” the existence of analgesic rebound and directed us to the best treatment at that time.4 This was the first study to examine the effect of stopping the overuse of medication to see the results.
Finally, I am unhappy that ICHD-3 has defined MOH only by number of days of medication use, not at all considering whether or not the patient develops a new type of headache, a worse type or more frequent headache (the way it was in previous versions). We have all seen patients taking 3 triptans per day on their own or with our suggestion, and many actually do better with no headache, for a period of time. This is medication overuse by definition, but not MOH, as they have little headache. But we do not recognize this entity.
The good news is, with education and anti-CGRP therapies, we will probably see less MOH in the future, or at least know better how to treat it.
+++
References
1. Bigal ME, Serrano D, Buse D, et al. Acute migraine medications and evolution from episodic to chronic migraine: a longitudinal population-based study. Headache. 2008;48(8):1157-68. doi: 10.1111/j.1526-4610.2008.01217.x. PubMed PMID: 18808500.
2. Bigal ME, Lipton RB. Overuse of acute migraine medications and migraine chronification. Curr Pain Headache Rep. 2009;13(4):301-7. PubMed PMID: 19586594.
3. Bigal ME, Edvinsson L, Rapoport AM, et al. Safety, tolerability, and efficacy of TEV-48125 for preventive treatment of chronic migraine: a multicentre, randomised, double-blind, placebo-controlled, phase 2b study. Lancet Neurol. 2015;14(11):1091-100. doi: 10.1016/S1474-4422(15)00245-8. PubMed PMID: 26432181.
4. Kudrow L. Paradoxical Effects of Frequent Analgesic Use. Advances in Neurology. 1982;33:335-341.
5. Diener HC, Holle D, Dresler T, Gaul C. Chronic Headache Due to Overuse of Analgesics and Anti-Migraine Agents. Dtsch Arztebl Int. 2018;115(22):365-370.
6. Westergaard ML, Glumer C, Hansen EH, Jensen RH. Prevalence of chronic headache with and without medication overuse: associations with socioeconomic position and physical and mental health status. Pain. 2014;155(10):2005-2013.
7. Castillo J, Munoz P, Guitera V, Pascual J. Epidemiology of chronic daily headache in the general population. Headache. 1999;39:190-196.
8. Wang SJ, Fuh JL, Lu SR, et al. Chronic daily headache in chinese elderly - prevalence, risk factors, and biannual follow-up. Neurology. 2000;54:314-319.
9. Lu SR, Fuh JL, Chen WT, Juang KD, Wang SJ. Chronic daily headache in Taipei, Taiwan: prevalence, follow-up and outcome predictors. Cephalalgia. 2001;21:980-986.
10. Pascual J, Colas R, Castillo J. Epidemiology of chronic daily headache. Curr Pain Headache Rep. 2001;5(6):529-536.
11. Prencipe M, Casini AR, Ferretti C, et al. Prevalence of headache in an elderly population: attack frequency, disability, and use of medication. J Neurol Neurosurg Psychiatry. 2001;70(3):377-381.
12. Colas R, Munoz P, Temprano R, Gomez C, Pascual J. Chronic daily headache with analgesic overuse: epidemiology and impact on quality of life. Neurology. 2004;62:1338-1342.
13. Zwart J, Dyb G, Hagen K, Svebak S, Stovner L, Holmen J. Analgesic overuse among subjects with headache, neck, and low back pain. Neurology. 2004;62:1540-1544.
14. Dyb G, Holmen TL, Zwart JA. Analgesic overuse among adolescents with headache: the Head-HUNT-Youth Study. Neurology. 2006;66(2):198-201.
15. Wang SJ, Fuh JL, Lu SR, Juang KD. Chronic daily headache in adolescents: prevalence, impact, and medication overuse. Neurology. 2006;66(2):193-197.
16. Wiendels NJ, Knuistingh NA, Rosendaal FR, et al. Chronic frequent headache in the general population: prevalence and associated factors. Cephalalgia. 2006;26(12):1434-1442.
17. Stovner L, Hagen K, Jensen R, et al. The global burden of headache: a documentation of headache prevalence and disability worldwide. Cephalalgia. 2007;27(3):193-210.
18. Aaseth K, Grande RB, Kvaerner KJ, Gulbrandsen P, Lundqvist C, Russell MB. Prevalence of secondary chronic headaches in a population-based sample of 30-44-year-old persons. The Akershus study of chronic headache. Cephalalgia. 2008;28(7):705-713.
19. Aaseth K, Grande RB, Lundqvist C, Russell MB. What is chronic headache in the general population? The Akershus study of chronic headache. Acta Neurol Scand Suppl. 2009;(189):30-32.
20. Rueda-Sanchez M, Diaz-Martinez LA. Prevalence and associated factors for episodic and chronic daily headache in the Colombian population. Cephalalgia. 2008;28(3):216-225.
21. Katsarava Z, Dzagnidze A, Kukava M, et al. Primary headache disorders in the Republic of Georgia: prevalence and risk factors. Neurology. 2009;73(21):1796-1803.
22. da Silva A, Jr., Costa EC, Gomes JB, et al. Chronic headache and comorbidities: a two-phase, population-based, cross-sectional study. Headache. 2010;50(8):1306-1312.
23. Straube A, Pfaffenrath V, Ladwig KH, et al. Prevalence of chronic migraine and medication overuse headache in Germany--the German DMKG headache study. Cephalalgia. 2010;30(2):207-213.
24. Jonsson P, Hedenrud T, Linde M. Epidemiology of medication overuse headache in the general Swedish population. Cephalalgia. 2011;31(9):1015-1022.
25. Jonsson P, Linde M, Hensing G, Hedenrud T. Sociodemographic differences in medication use, health-care contacts and sickness absence among individuals with medication-overuse headache. J Headache Pain. 2012;13(4):281-290.
26. Linde M, Stovner LJ, Zwart JA, Hagen K. Time trends in the prevalence of headache disorders. The Nord-Trondelag Health Studies (HUNT 2 and HUNT 3). Cephalalgia. 2011;31(5):585-596.
27. Lipton RB, Manack A, Ricci JA, Chee E, Turkel CC, Winner P. Prevalence and burden of chronic migraine in adolescents: results of the chronic daily headache in adolescents study (C-dAS). Headache. 2011;51(5):693-706.
28. Ayzenberg I, Katsarava Z, Sborowski A, et al. The prevalence of primary headache disorders in Russia: a countrywide survey. Cephalalgia. 2012;32(5):373-381.
29. Ertas M, Baykan B, Orhan EK, et al. One-year prevalence and the impact of migraine and tension-type headache in Turkey: a nationwide home-based study in adults. J Headache Pain. 2012;13(2):147-157.
30. Hagen K, Linde M, Steiner TJ, Stovner LJ, Zwart JA. Risk factors for medication-overuse headache: an 11-year follow-up study. The Nord-Trondelag Health Studies. Pain. 2012;153(1):56-61.
31. Yu S, Liu R, Zhao G, et al. The prevalence and burden of primary headaches in China: a population-based door-to-door survey. Headache. 2012;52(4):582-591.
32. Shahbeigi S, Fereshtehnejad SM, Mohammadi N, et al. Epidemiology of headaches in Tehran urban area: a population-based cross-sectional study in district 8, year 2010. Neurol Sci. 2013;34(7):1157-66.
33. Schramm SH, Obermann M, Katsarava Z, Diener HC, Moebus S, Yoon MS. Epidemiological profiles of patients with chronic migraine and chronic tension-type headache. J Headache Pain. 2013;14:40.
34. Park JW, Moon HS, Kim JM, Lee KS, Chu MK. Chronic daily headache in Korea: prevalence, clinical characteristics, medical consultation and management. J Clin Neurol. 2014;10(3):236-243.
35. Kristoffersen ES, Lundqvist C. Medication-overuse headache: epidemiology, diagnosis and treatment. Ther Adv Drug Saf. 2014;5(2):87-99.
36. Steiner TJ, Stovner LJ, Katsarava Z, et al. The impact of headache in Europe: principal results of the Eurolight project. J Headache Pain. 2014;15:31.
37. Westergaard ML, Hansen EH, Glumer C, Jensen RH. Prescription pain medications and chronic headache in Denmark: implications for preventing medication overuse. Eur J Clin Pharmacol. 2015;71(7):851-860.
38. Bravo TP. Headaches of the elderly. Curr Neurol Neurosci Rep. 2015;15(6):30.
39. Mbewe E, Zairemthiama P, Yeh HH, Paul R, Birbeck GL, Steiner TJ. The epidemiology of primary headache disorders in Zambia: a population-based door-to-door survey. J Headache Pain. 2015;16:515.
40. Kulkarni GB, Rao GN, Gururaj G, Stovner LJ, Steiner TJ. Headache disorders and public ill-health in India: prevalence estimates in Karnataka State. J Headache Pain. 2015;16:67.
41. Westergaard ML, Glumer C, Hansen EH, Jensen RH. Medication overuse, healthy lifestyle behaviour and stress in chronic headache: Results from a population-based representative survey. Cephalalgia. 2016;36(1):15-28.
42. Manandhar K, Risal A, Linde M, Steiner TJ. The burden of headache disorders in Nepal: estimates from a population-based survey. J Headache Pain. 2015;17:3.
43. Zebenigus M, Tekle-Haimanot R, Worku DK, Thomas H, Steiner TJ. The prevalence of primary headache disorders in Ethiopia. J Headache Pain. 2016;17(1):110.
44. Al-Hashel JY, Ahmed SF, Alroughani R. Prevalence of Primary Headache Disorders in Kuwait. Neuroepidemiology. 2017;48(3-4):138-146.
45. Rastenyte D, Mickeviciene D, Stovner LJ, Thomas H, Andree C, Steiner TJ. Prevalence and burden of headache disorders in Lithuania and their public-health and policy implications: a population-based study within the Eurolight Project. J Headache Pain. 2017;18(1):53.
46. Henning V, Katsarava Z, Obermann M, Moebus S, Schramm S. Remission of chronic headache: Rates, potential predictors and the role of medication, follow-up results of the German Headache Consortium (GHC) Study. Cephalalgia. 2018;38(3):551-560.
47. Global Burden of Disease Neurological Disorders Collaborator Group. Global, regional, and national burden of neurological disorders during 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Neurol. 2017;16(11):877-897.
Haplo-HCT shows viability in DLBCL
For patients with diffuse large B-cell lymphoma (DLBCL) who need allogeneic hematopoietic cell transplantation (allo-HCT), a haploidentical family member could be a viable donor, according to a retrospective study of 1,438 patients.
When combined with nonmyeloablative/reduced intensity conditioning (NMC/RIC) and posttransplant cyclophosphamide (PTCy), patients treated with haploidentical HCT (haplo-HCT) had outcomes similar to those seen in patients with matched donors, reported Peter Dreger, MD, of the University of Heidelberg (Germany) and his colleagues.
“Using well-matched sibling donors (MSDs) or unrelated donors (MUDs), allo-HCT can result in sustained disease control in 30% to 45% of patients with DLBCL who have early disease recurrence after standard chemoimmunotherapy or have failed auto-HCT [autologous HCT],” the investigators wrote in Blood Advances. “However, the search for a well-matched unrelated donor could be time-consuming and unsuccessful in up to 50% of the patients in need.”
But the present findings suggest that haplo-HCT may one day improve these odds by providing a larger pool of potential donors.
The patients in the study were divided into four treatment groups: haplo-HCT (n = 132), MSD (n = 525), MUD with T-cell depletion (n = 403), and MUD without T-cell depletion (n = 378). For graft-versus-host disease (GVHD) prophylaxis, patients in the haplo-HCT group received PTCy, with or without a calcineurin inhibitor and mycophenolate mofetil, whereas all patients with matched donors received a calcineurin inhibitor. T-cell depletion was accomplished by in vivo antithymocyte globulin and alemtuzumab.
The primary end point was overall survival (OS). Secondary end points were progression-free survival (PFS), progression/relapse, and nonrelapse mortality (NRM).
After a median follow-up of 4.1 years, all groups had similar outcomes, without statistical differences in multivariable analysis.
In the haplo-HCT group, the 3-year OS rate was 46%, the NRM rate was 22%, the PFS rate was 38%, and the relapse/progression rate was 41%.
Of note, patients receiving haplo-HCT did have a lower cumulative incidence of chronic GVHD, at 15% after 1 year and 18% after 2 years. These rates were significantly lower than the other groups’ 1- and 2-year GVHD rates, which were as follows: MSD, 41% and 48%; MUD with T-cell depletion, 23% and 27%; and MUD without T-cell depletion, 48% and 57%.
The investigators noted that these disparities may actually be caused by the use of bone marrow grafts in the haplo-HCT group instead of peripheral blood grafts, which were used in most of the patients in the other groups.
Overall, the findings were encouraging, but the investigators cautioned that “additional studies are needed before haploidentical donors can be considered as equivalent to well-matched related or unrelated donors in patients with DLBCL.”
The study was funded by the Center for International Blood & Marrow Transplant Research (CIBMTR) and the European Society for Blood and Marrow Transplantation. CIBMTR is supported by grants from the U.S. government and the pharmaceutical industry. The authors reported having no competing financial interests.
SOURCE: Dreger P et al. Blood Adv. 2019 Feb 12;3(3):360-9.
For patients with diffuse large B-cell lymphoma (DLBCL) who need allogeneic hematopoietic cell transplantation (allo-HCT), a haploidentical family member could be a viable donor, according to a retrospective study of 1,438 patients.
When combined with nonmyeloablative/reduced intensity conditioning (NMC/RIC) and posttransplant cyclophosphamide (PTCy), patients treated with haploidentical HCT (haplo-HCT) had outcomes similar to those seen in patients with matched donors, reported Peter Dreger, MD, of the University of Heidelberg (Germany) and his colleagues.
“Using well-matched sibling donors (MSDs) or unrelated donors (MUDs), allo-HCT can result in sustained disease control in 30% to 45% of patients with DLBCL who have early disease recurrence after standard chemoimmunotherapy or have failed auto-HCT [autologous HCT],” the investigators wrote in Blood Advances. “However, the search for a well-matched unrelated donor could be time-consuming and unsuccessful in up to 50% of the patients in need.”
But the present findings suggest that haplo-HCT may one day improve these odds by providing a larger pool of potential donors.
The patients in the study were divided into four treatment groups: haplo-HCT (n = 132), MSD (n = 525), MUD with T-cell depletion (n = 403), and MUD without T-cell depletion (n = 378). For graft-versus-host disease (GVHD) prophylaxis, patients in the haplo-HCT group received PTCy, with or without a calcineurin inhibitor and mycophenolate mofetil, whereas all patients with matched donors received a calcineurin inhibitor. T-cell depletion was accomplished by in vivo antithymocyte globulin and alemtuzumab.
The primary end point was overall survival (OS). Secondary end points were progression-free survival (PFS), progression/relapse, and nonrelapse mortality (NRM).
After a median follow-up of 4.1 years, all groups had similar outcomes, without statistical differences in multivariable analysis.
In the haplo-HCT group, the 3-year OS rate was 46%, the NRM rate was 22%, the PFS rate was 38%, and the relapse/progression rate was 41%.
Of note, patients receiving haplo-HCT did have a lower cumulative incidence of chronic GVHD, at 15% after 1 year and 18% after 2 years. These rates were significantly lower than the other groups’ 1- and 2-year GVHD rates, which were as follows: MSD, 41% and 48%; MUD with T-cell depletion, 23% and 27%; and MUD without T-cell depletion, 48% and 57%.
The investigators noted that these disparities may actually be caused by the use of bone marrow grafts in the haplo-HCT group instead of peripheral blood grafts, which were used in most of the patients in the other groups.
Overall, the findings were encouraging, but the investigators cautioned that “additional studies are needed before haploidentical donors can be considered as equivalent to well-matched related or unrelated donors in patients with DLBCL.”
The study was funded by the Center for International Blood & Marrow Transplant Research (CIBMTR) and the European Society for Blood and Marrow Transplantation. CIBMTR is supported by grants from the U.S. government and the pharmaceutical industry. The authors reported having no competing financial interests.
SOURCE: Dreger P et al. Blood Adv. 2019 Feb 12;3(3):360-9.
For patients with diffuse large B-cell lymphoma (DLBCL) who need allogeneic hematopoietic cell transplantation (allo-HCT), a haploidentical family member could be a viable donor, according to a retrospective study of 1,438 patients.
When combined with nonmyeloablative/reduced intensity conditioning (NMC/RIC) and posttransplant cyclophosphamide (PTCy), patients treated with haploidentical HCT (haplo-HCT) had outcomes similar to those seen in patients with matched donors, reported Peter Dreger, MD, of the University of Heidelberg (Germany) and his colleagues.
“Using well-matched sibling donors (MSDs) or unrelated donors (MUDs), allo-HCT can result in sustained disease control in 30% to 45% of patients with DLBCL who have early disease recurrence after standard chemoimmunotherapy or have failed auto-HCT [autologous HCT],” the investigators wrote in Blood Advances. “However, the search for a well-matched unrelated donor could be time-consuming and unsuccessful in up to 50% of the patients in need.”
But the present findings suggest that haplo-HCT may one day improve these odds by providing a larger pool of potential donors.
The patients in the study were divided into four treatment groups: haplo-HCT (n = 132), MSD (n = 525), MUD with T-cell depletion (n = 403), and MUD without T-cell depletion (n = 378). For graft-versus-host disease (GVHD) prophylaxis, patients in the haplo-HCT group received PTCy, with or without a calcineurin inhibitor and mycophenolate mofetil, whereas all patients with matched donors received a calcineurin inhibitor. T-cell depletion was accomplished by in vivo antithymocyte globulin and alemtuzumab.
The primary end point was overall survival (OS). Secondary end points were progression-free survival (PFS), progression/relapse, and nonrelapse mortality (NRM).
After a median follow-up of 4.1 years, all groups had similar outcomes, without statistical differences in multivariable analysis.
In the haplo-HCT group, the 3-year OS rate was 46%, the NRM rate was 22%, the PFS rate was 38%, and the relapse/progression rate was 41%.
Of note, patients receiving haplo-HCT did have a lower cumulative incidence of chronic GVHD, at 15% after 1 year and 18% after 2 years. These rates were significantly lower than the other groups’ 1- and 2-year GVHD rates, which were as follows: MSD, 41% and 48%; MUD with T-cell depletion, 23% and 27%; and MUD without T-cell depletion, 48% and 57%.
The investigators noted that these disparities may actually be caused by the use of bone marrow grafts in the haplo-HCT group instead of peripheral blood grafts, which were used in most of the patients in the other groups.
Overall, the findings were encouraging, but the investigators cautioned that “additional studies are needed before haploidentical donors can be considered as equivalent to well-matched related or unrelated donors in patients with DLBCL.”
The study was funded by the Center for International Blood & Marrow Transplant Research (CIBMTR) and the European Society for Blood and Marrow Transplantation. CIBMTR is supported by grants from the U.S. government and the pharmaceutical industry. The authors reported having no competing financial interests.
SOURCE: Dreger P et al. Blood Adv. 2019 Feb 12;3(3):360-9.
FROM BLOOD ADVANCES
New Topical Treatments for Psoriasis



FDA grants priority review to polatuzumab vedotin for DLBCL
With this BLA, Genentech is seeking approval for polatuzumab vedotin in combination with bendamustine and rituximab (BR) to treat patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL).
The FDA grants priority review to applications for products that are expected to provide significant improvements in the treatment, diagnosis, or prevention of serious conditions. The agency aims to take action on a priority review application within 6 months, rather than the standard 10 months.
The FDA is expected to make a decision on this BLA by Aug. 19, 2019.
The BLA is supported by a phase 1b/2 trial (NCT02257567) of patients with relapsed or refractory follicular lymphoma or DLBCL who received polatuzumab vedotin in combination with BR or obinutuzumab.
The trial’s phase 2 stage included 80 DLBCL patients who were randomized to receive BR or BR plus polatuzumab vedotin, according to Genentech.
The complete response rate was 40% in the polatuzumab vedotin arm and 18% in the BR arm. The median duration of response was 10.3 months and 4.1 months, respectively (hazard ratio [HR] = 0.44).
The median progression-free survival was 7.6 months in the polatuzumab vedotin arm and 2.0 months in the BR arm (HR = 0.34).
Among patients who were ineligible for a transplant, the median overall survival (an exploratory endpoint) was 12.4 months in the polatuzumab vedotin arm and 4.7 months in the BR arm (HR = 0.42).
Patients who received polatuzumab vedotin had higher rates of grade 3-4 cytopenias, compared with patients who received BR alone. Rates of infection and transfusion were similar between the arms.
With this BLA, Genentech is seeking approval for polatuzumab vedotin in combination with bendamustine and rituximab (BR) to treat patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL).
The FDA grants priority review to applications for products that are expected to provide significant improvements in the treatment, diagnosis, or prevention of serious conditions. The agency aims to take action on a priority review application within 6 months, rather than the standard 10 months.
The FDA is expected to make a decision on this BLA by Aug. 19, 2019.
The BLA is supported by a phase 1b/2 trial (NCT02257567) of patients with relapsed or refractory follicular lymphoma or DLBCL who received polatuzumab vedotin in combination with BR or obinutuzumab.
The trial’s phase 2 stage included 80 DLBCL patients who were randomized to receive BR or BR plus polatuzumab vedotin, according to Genentech.
The complete response rate was 40% in the polatuzumab vedotin arm and 18% in the BR arm. The median duration of response was 10.3 months and 4.1 months, respectively (hazard ratio [HR] = 0.44).
The median progression-free survival was 7.6 months in the polatuzumab vedotin arm and 2.0 months in the BR arm (HR = 0.34).
Among patients who were ineligible for a transplant, the median overall survival (an exploratory endpoint) was 12.4 months in the polatuzumab vedotin arm and 4.7 months in the BR arm (HR = 0.42).
Patients who received polatuzumab vedotin had higher rates of grade 3-4 cytopenias, compared with patients who received BR alone. Rates of infection and transfusion were similar between the arms.
With this BLA, Genentech is seeking approval for polatuzumab vedotin in combination with bendamustine and rituximab (BR) to treat patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL).
The FDA grants priority review to applications for products that are expected to provide significant improvements in the treatment, diagnosis, or prevention of serious conditions. The agency aims to take action on a priority review application within 6 months, rather than the standard 10 months.
The FDA is expected to make a decision on this BLA by Aug. 19, 2019.
The BLA is supported by a phase 1b/2 trial (NCT02257567) of patients with relapsed or refractory follicular lymphoma or DLBCL who received polatuzumab vedotin in combination with BR or obinutuzumab.
The trial’s phase 2 stage included 80 DLBCL patients who were randomized to receive BR or BR plus polatuzumab vedotin, according to Genentech.
The complete response rate was 40% in the polatuzumab vedotin arm and 18% in the BR arm. The median duration of response was 10.3 months and 4.1 months, respectively (hazard ratio [HR] = 0.44).
The median progression-free survival was 7.6 months in the polatuzumab vedotin arm and 2.0 months in the BR arm (HR = 0.34).
Among patients who were ineligible for a transplant, the median overall survival (an exploratory endpoint) was 12.4 months in the polatuzumab vedotin arm and 4.7 months in the BR arm (HR = 0.42).
Patients who received polatuzumab vedotin had higher rates of grade 3-4 cytopenias, compared with patients who received BR alone. Rates of infection and transfusion were similar between the arms.
Early intensive treatment of MS may benefit patients
First-line treatment of multiple sclerosis with a high-efficacy therapy may produce better long-term outcomes than does an escalation treatment approach, data from a real-world cohort study suggest.
In a population-based cohort of patients with multiple sclerosis (MS) in southeast Wales, those who initiated treatment with a high-efficacy therapy had a smaller average increase in Expanded Disability Status Scale (EDSS) score after 5 years, compared with patients who started on moderate-efficacy therapy, researchers reported Feb. 18 in JAMA Neurology. These outcomes occurred “despite clinical surveillance and targeted escalation” in the group of patients who started on moderate-efficacy drugs, said first author Katharine Harding, PhD, of Cardiff University and the University Hospital of Wales in Cardiff and the Royal Gwent Hospital, Newport, Wales, and her colleagues. “These findings suggest that real-world escalation approaches may be inadequate to prevent unfavorable long-term outcomes and support the need for a prospective clinical trial to compare disease-modifying therapy algorithms.”
The investigators analyzed data collected between January 1998 and December 2016 from 592 patients with MS. Of the 592 patients, 104 initiated treatment with alemtuzumab (Lemtrada) or natalizumab (Tysabri), which the researchers classified as high-efficacy therapies (i.e., early intensive treatment), and 488 initiated treatment with interferons, glatiramer acetate (Copaxone), dimethyl fumarate (Tecfidera), fingolimod (Gilenya), or teriflunomide (Aubagio), which were considered moderate-efficacy therapies (i.e., escalation approach).
At baseline, patients who received early intensive treatment had higher average EDSS scores, compared with patients treated with an escalation approach (4.2 vs. 3.5). After 5 years, the average increase in EDSS score was lower among patients who received early intensive treatment, compared with patients treated with an escalation approach (0.3 vs. 1.2). The researchers adjusted for patients’ sex, age at treatment, year of starting treatment, and escalation to high-efficacy treatment in the escalation treatment approach group.
Median time to sustained accumulation of disability was 6.0 years for the early intensive therapy group and 3.1 years for the escalation therapy group, but the risk of sustained accumulation of disability did not differ between the groups after adjustment for covariates.
“Although patients were selected to receive early intensive treatment on the basis of poor prognostic factors, including more active disease, it was this patient group that had better long-term outcomes,” Dr. Harding and her colleagues wrote.
There were no treatment-related deaths in the study. Among patients who received alemtuzumab, 87% developed infusion-related adverse events, and 47% developed autoimmunity. Among patients receiving natalizumab, there were no serious adverse events and no cases of progressive multifocal leukoencephalopathy. In patients receiving moderate-efficacy disease-modifying therapies, there were seven serious adverse events (1.4%).
Dr. Harding disclosed grants from Novartis UK outside the present study. Coauthors reported honoraria, support to attend educational meetings, and travel expenses, as well as grants and salary outside the present study, from various pharmaceutical companies, including Biogen, Teva, Roche, MedDay Pharma, Merck, Genzyme, and Novartis.
SOURCE: Harding K et al. JAMA Neurol. 2019 Feb 18. doi: 10.1001/jamaneurol.2018.4905
First-line treatment of multiple sclerosis with a high-efficacy therapy may produce better long-term outcomes than does an escalation treatment approach, data from a real-world cohort study suggest.
In a population-based cohort of patients with multiple sclerosis (MS) in southeast Wales, those who initiated treatment with a high-efficacy therapy had a smaller average increase in Expanded Disability Status Scale (EDSS) score after 5 years, compared with patients who started on moderate-efficacy therapy, researchers reported Feb. 18 in JAMA Neurology. These outcomes occurred “despite clinical surveillance and targeted escalation” in the group of patients who started on moderate-efficacy drugs, said first author Katharine Harding, PhD, of Cardiff University and the University Hospital of Wales in Cardiff and the Royal Gwent Hospital, Newport, Wales, and her colleagues. “These findings suggest that real-world escalation approaches may be inadequate to prevent unfavorable long-term outcomes and support the need for a prospective clinical trial to compare disease-modifying therapy algorithms.”
The investigators analyzed data collected between January 1998 and December 2016 from 592 patients with MS. Of the 592 patients, 104 initiated treatment with alemtuzumab (Lemtrada) or natalizumab (Tysabri), which the researchers classified as high-efficacy therapies (i.e., early intensive treatment), and 488 initiated treatment with interferons, glatiramer acetate (Copaxone), dimethyl fumarate (Tecfidera), fingolimod (Gilenya), or teriflunomide (Aubagio), which were considered moderate-efficacy therapies (i.e., escalation approach).
At baseline, patients who received early intensive treatment had higher average EDSS scores, compared with patients treated with an escalation approach (4.2 vs. 3.5). After 5 years, the average increase in EDSS score was lower among patients who received early intensive treatment, compared with patients treated with an escalation approach (0.3 vs. 1.2). The researchers adjusted for patients’ sex, age at treatment, year of starting treatment, and escalation to high-efficacy treatment in the escalation treatment approach group.
Median time to sustained accumulation of disability was 6.0 years for the early intensive therapy group and 3.1 years for the escalation therapy group, but the risk of sustained accumulation of disability did not differ between the groups after adjustment for covariates.
“Although patients were selected to receive early intensive treatment on the basis of poor prognostic factors, including more active disease, it was this patient group that had better long-term outcomes,” Dr. Harding and her colleagues wrote.
There were no treatment-related deaths in the study. Among patients who received alemtuzumab, 87% developed infusion-related adverse events, and 47% developed autoimmunity. Among patients receiving natalizumab, there were no serious adverse events and no cases of progressive multifocal leukoencephalopathy. In patients receiving moderate-efficacy disease-modifying therapies, there were seven serious adverse events (1.4%).
Dr. Harding disclosed grants from Novartis UK outside the present study. Coauthors reported honoraria, support to attend educational meetings, and travel expenses, as well as grants and salary outside the present study, from various pharmaceutical companies, including Biogen, Teva, Roche, MedDay Pharma, Merck, Genzyme, and Novartis.
SOURCE: Harding K et al. JAMA Neurol. 2019 Feb 18. doi: 10.1001/jamaneurol.2018.4905
First-line treatment of multiple sclerosis with a high-efficacy therapy may produce better long-term outcomes than does an escalation treatment approach, data from a real-world cohort study suggest.
In a population-based cohort of patients with multiple sclerosis (MS) in southeast Wales, those who initiated treatment with a high-efficacy therapy had a smaller average increase in Expanded Disability Status Scale (EDSS) score after 5 years, compared with patients who started on moderate-efficacy therapy, researchers reported Feb. 18 in JAMA Neurology. These outcomes occurred “despite clinical surveillance and targeted escalation” in the group of patients who started on moderate-efficacy drugs, said first author Katharine Harding, PhD, of Cardiff University and the University Hospital of Wales in Cardiff and the Royal Gwent Hospital, Newport, Wales, and her colleagues. “These findings suggest that real-world escalation approaches may be inadequate to prevent unfavorable long-term outcomes and support the need for a prospective clinical trial to compare disease-modifying therapy algorithms.”
The investigators analyzed data collected between January 1998 and December 2016 from 592 patients with MS. Of the 592 patients, 104 initiated treatment with alemtuzumab (Lemtrada) or natalizumab (Tysabri), which the researchers classified as high-efficacy therapies (i.e., early intensive treatment), and 488 initiated treatment with interferons, glatiramer acetate (Copaxone), dimethyl fumarate (Tecfidera), fingolimod (Gilenya), or teriflunomide (Aubagio), which were considered moderate-efficacy therapies (i.e., escalation approach).
At baseline, patients who received early intensive treatment had higher average EDSS scores, compared with patients treated with an escalation approach (4.2 vs. 3.5). After 5 years, the average increase in EDSS score was lower among patients who received early intensive treatment, compared with patients treated with an escalation approach (0.3 vs. 1.2). The researchers adjusted for patients’ sex, age at treatment, year of starting treatment, and escalation to high-efficacy treatment in the escalation treatment approach group.
Median time to sustained accumulation of disability was 6.0 years for the early intensive therapy group and 3.1 years for the escalation therapy group, but the risk of sustained accumulation of disability did not differ between the groups after adjustment for covariates.
“Although patients were selected to receive early intensive treatment on the basis of poor prognostic factors, including more active disease, it was this patient group that had better long-term outcomes,” Dr. Harding and her colleagues wrote.
There were no treatment-related deaths in the study. Among patients who received alemtuzumab, 87% developed infusion-related adverse events, and 47% developed autoimmunity. Among patients receiving natalizumab, there were no serious adverse events and no cases of progressive multifocal leukoencephalopathy. In patients receiving moderate-efficacy disease-modifying therapies, there were seven serious adverse events (1.4%).
Dr. Harding disclosed grants from Novartis UK outside the present study. Coauthors reported honoraria, support to attend educational meetings, and travel expenses, as well as grants and salary outside the present study, from various pharmaceutical companies, including Biogen, Teva, Roche, MedDay Pharma, Merck, Genzyme, and Novartis.
SOURCE: Harding K et al. JAMA Neurol. 2019 Feb 18. doi: 10.1001/jamaneurol.2018.4905
FROM JAMA NEUROLOGY
Key clinical point:
Major finding: After 5 years, the average increase in Expanded Disability Status Scale score was lower among patients who received early intensive treatment, compared with patients treated with an escalation approach (0.3 vs. 1.2).
Study details: A population-based cohort study of 592 patients with MS in southeast Wales.
Disclosures: Dr. Harding disclosed grants from Novartis UK outside the present study. Coauthors reported honoraria, support to attend educational meetings, and travel expenses, as well as grants and salary outside the present study, from various pharmaceutical companies, including Biogen, Teva, Roche, MedDay Pharma, Merck, Genzyme, and Novartis.
Source: Harding K et al. JAMA Neurol. 2019 Feb 18. doi: 10.1001/jamaneurol.2018.4905.