User login
Venous thromboembolism: What to do after anticoagulation is started
Deep vein thrombosis and pulmonary embolism are collectively referred to as venous thromboembolic (VTE) disease. They affect approximately 100,000 to 300,000 patients per year in the United States.1 Although patients with deep vein thrombosis can be treated as outpatients, many are admitted for the initiation of anticoagulation. Initial anticoagulation usually requires the overlap of a parenteral anticoagulant (unfractionated heparin, low-molecular-weight heparin [LMWH] or fondaparinux) with warfarin for a minimum of 5 days and until the international normalized ratio (INR) of the prothrombin time is above 2.0 for at least 24 hours.2
Three clinical issues need to be addressed after the initiation of anticoagulation for VTE:
- Determination of the length of anticoagulation with the correct anticoagulant
- Prevention of postthrombotic syndrome
- Appropriate screening for occult malignancy.
HOW LONG SHOULD VTE BE TREATED?
The duration of anticoagulation has been a matter of debate.
The risk of recurrent VTE appears related to clinical risk factors that a patient has at the time of the initial thrombotic event. An epidemiologic study3 found that patients with VTE treated for approximately 6 months had a low rate of recurrence (0% at 2 years of follow-up) if surgery was the risk factor. The risk climbed to 9% if the risk factor was nonsurgical and to 19% if there were no provoking risk factors.
The likelihood of VTE recurrence and therefore the recommended duration of treatment depend on whether the VTE event was provoked, cancer-related, recurrent, thrombophilia-related, or idiopathic. We address each of these scenarios below.
HOW LONG TO TREAT PROVOKED VTE
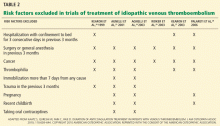
A VTE event is considered provoked if the patient had a clear inciting risk factor. As defined in various clinical trials, these risk factors include:
- Hospitalization with confinement to bed for 3 or more consecutive days in the last 3 months
- Surgery or general anesthesia in the last 3 months
- Immobilization for more than 7 days, regardless of the cause
- Trauma in the last 3 months
- Pregnancy
- Use of an oral contraceptive, regardless of which estrogen or progesterone analogue it contains
- Travel for more than 4 hours
- Recent childbirth.
However, the trials that tested different lengths of anticoagulation have varied markedly in how they defined provoked deep vein thrombosis.4–7
Recommendation: Warfarin or equivalent for 3 months
The American College of Chest Physicians (ACCP) recommends 3 months of anticoagulation with warfarin or another vitamin K antagonist for patients with VTE secondary to a transient (reversible) risk factor,2 and we agree.
HOW LONG TO TREAT CANCER-RELATED VTE
Patients with cancer are at higher risk of developing VTE. Furthermore, in one study,9 compared with other patients with VTE, patients with cancer were three times more likely to have another episode of VTE, with a cumulative rate of recurrence at 1 year of 21% vs 7%. Cancer patients were also twice as likely to suffer major bleeding complications while on anticoagulation.9
Warfarin is a difficult drug to manage because it has many interactions with foods, diseases, and other drugs. These difficulties are amplified in many cancer patients during chemotherapy.
Warfarin was compared with a LMWH in four randomized trials in cancer patients, and a meta-analysis10 found a 50% relative reduction in the rates of recurrent deep vein thrombosis and pulmonary embolism with LMWH treatment. These results were driven primarily by the CLOT trial (Comparison of Low-Molecular-Weight Heparin Versus Oral Anticoagulant Therapy for the Prevention of Recurrent Venous Thromboembolism in Patients With Cancer),11 which showed an 8% absolute risk reduction (number needed to treat 13) without an increase in major bleeding when cancer-related VTE was treated with an LMWH—ie, dalteparin (Fragmin)—for 6 months compared with warfarin.
Current thinking suggests that VTE should be treated until the cancer is resolved. However, this hypothesis has not been adequately tested, and consequently, the ACCP gives it only a level 1C recommendation.2 The largest of the four trials comparing warfarin and an LMWH lasted only 6 months. The safety of extending LMWH treatment beyond 6 months is currently unknown but is under investigation (clinicaltrials.gov identifier NCT00942968).
Recommendation: LMWH therapy for at least 6 months
The ACCP guidelines recommend LMWH therapy for 3 to 6 months, followed by warfarin or another vitamin K antagonist or continued LMWH treatment until the cancer is resolved.2
The National Comprehensive Cancer Network guidelines recommend an LMWH for 6 months as monotherapy and indefinite anticoagulation if the cancer is still active.12
The American Society of Clinical Oncology guidelines recommend an LMWH for at least 6 months and indefinite anticoagulant therapy for selected patients with active cancer.13
We agree that patients with active cancer should receive an LMWH for at least 6 months and indefinite anticoagulation until the cancer is resolved.
In our experience, many patients are reluctant to give themselves the daily injections that LMWH therapy requires, and so they need to be well-informed about the marked decrease in VTE recurrence with this less-convenient and more-expensive therapy. Many patients face insurance barriers to cover the cost of LMWH therapy; however, careful attention to preauthorization can usually overcome this obstacle.
HOW LONG TO TREAT RECURRENT VTE
It makes clinical sense that patients who have a second VTE event should be treated indefinitely. This theory was tested in a randomized clinical trial14 in which patients with provoked or unprovoked VTE were randomized after their second event to receive anticoagulation for 6 months vs indefinitely.
After 4 years of follow-up, the recurrence rate was 21% in patients assigned to 6 months of treatment and only 3% in patients who continued anticoagulation throughout the trial. On the other hand, major hemorrhage occurred in 3% of patients treated for 6 months and in 9% in patients who continued anticoagulation indefinitely.
Of note, most of the patients in this trial had unprovoked (idiopathic) VTE, so the results should not be extrapolated to patients with provoked VTE, who accounted for only 20% of the study population.14
Recommendation: Long-term anticoagulation
We agree with the ACCP recommendation2 that patients who have had a second episode of unprovoked VTE should receive long-term anticoagulation. Because of a lack of data, the duration of therapy for patients with a second episode of provoked VTE should be individualized.
HOW LONG TO TREAT THROMBOPHILIA-RELATED VTE
Inherited thrombophilias
Patients with VTE that is not related to a clear provoking risk factor or cancer frequently have testing to evaluate for a hypercoagulable state. This workup traditionally includes the most common inherited thrombophilias for gene mutations for factor V and prothrombin as well as for deficiencies in protein C, protein S, antithrombin and the acquired antiphospholipid syndrome.
The key questions that should be asked prior to embarking on this workup are:
- Will the results change the length of therapy for the patient?
- Will testing the patient help with genetic counseling and possible testing of family members?
- Will the results change the targeted INR range for warfarin or other vitamin K antagonist therapy?
Patients with inherited thrombophilia have a greater risk of developing an initial VTE event; however, these tests do not help predict the recurrence of VTE in patients with established disease more than clinical risk factors do. A prospective study demonstrated this by looking at the effect of thrombophilia and clinical factors on the recurrence of venous thrombosis and found that inherited prothrombotic abnormalities do not appear to play an important role in the risk of a recurrent event.15 On the other hand, clinical factors, such as whether the first event was idiopathic or provoked, appear more important in determining the duration of anticoagulation therapy.15 A systematic review of the common inherited thrombophilias showed the VTE recurrence rate of patients with factor V Leiden was higher than in patients without the mutation; however, the absolute rates of recurrence were not much different than what would be expected in patients with idiopathic VTE.16
A retrospective study involving a large cohort of families of patients who already had experienced a first episode of either idiopathic or provoked VTE showed high annual risks of recurrent VTE associated with hereditary deficiencies of protein S (8.4%), protein C (6.0%), and antithrombin (10%).17 However, for the more commonly occurring genetic thrombophilias, the factor V Leiden and prothrombin G20210A mutations, family members with either gene abnormality had low rates of VTE, suggesting that testing of relatives of probands is not clinically useful.16
Antiphospholipid syndrome
Antiphospholipid syndrome is an acquired thrombophilia. A patient has thrombotic antiphospholipid syndrome when there is a history of vascular thrombosis in the presence of persistently positive tests (at least 12 weeks apart) for lupus anticoagulants, anticardiolipin antibodies, or anti-beta-2 glycoprotein I. A prospective study of 412 patients with a first episode of VTE found that 15% were positive for anticardiolipin antibody at the end of 6 months of anticoagulation. The risk of recurrent VTE after 4 years was 29% in patients with antibodies and 14% in those without antibodies (relative risk 2.1; 95% confidence interval [CI] 1.3–3.3; P =.0013).18
Recent reviews advise indefinite warfarin anticoagulation in patients with VTE and persistence of antiphospholipid antibodies.19 However, the optimal duration of anticoagulation is uncertain. Until well-designed clinical trials are done, the current general consensus is to anticoagulate these patients indefinitely.20,21 Retrospective studies had suggested that patients with antiphospholipid antibodies required a higher therapeutic INR range; however, this observation was tested in two trials that found no difference in thromboembolic rates when patients were randomized to an INR of 2.0–3.0 vs 3.1–4.0,22 or 2.0–3.0 vs 3.0–4.5.23
No formal recommendations
In the absence of strong evidence, the ACCP guidelines do not include a recommendation on the duration of anticoagulation treatment specific to inherited thrombophilias. We believe that clinical factors are more important than inherited thrombophilias for deciding the duration of anticoagulation, and that testing is almost never indicated or useful. However, patients with antiphospholipid syndrome are at high risk of recurrence, and it is our practice to anticoagulate these patients indefinitely.
HOW LONG TO TREAT UNPROVOKED (IDIOPATHIC) VTE
A VTE event is thought to be idiopathic if it occurs without a clearly identified provoking factor.
In an observational study,3 patients with oral contraceptive use, transient illness, immobilization, or a history of travel had an 8.8% risk of recurrence vs 19.4% in patients with unprovoked VTE. The meta-analysis discussed above (Table 1)8 also shows that patients with these nonsurgical risk factors have a lower rate of recurrence than patients with idiopathic VTE.
The high rate of recurrence of idiopathic VTE (4% to 27% after 3 months of anticoagulation24–26) suggests that a longer duration of treatment is reasonable. However, increasing the length of therapy from 3 to 12 months delays but does not prevent recurrence, the risk of which begins to accumulate once anticoagulation is stopped.24,25
Three promising strategies to identify subgroups of patients with idiopathic VTE who are at highest risk of recurrence and who would benefit the most from prolonged anticoagulation are d-dimer testing, evaluation for residual vein thrombosis in patients who present with a deep vein thrombosis, and clinical prediction rules.
d-dimer testing
d-dimer is a degradation product of fibrin and is an indirect marker of residual thrombosis.30
In a systematic review of patients with a first episode of unprovoked VTE,31 a normal d-dimer concentration at the end of at least 3 months of anticoagulation was associated with a 3.5% annual risk of recurrence, whereas an elevated d-dimer level at that time was associated with an annual risk of 8.9%. These results were confirmed in a systematic review of individual patient data.32
In a randomized trial,28 patients with an idiopathic VTE event who received anticoagulation for at least 3 months had their d-dimer level measured 1 month after cessation of treatment. Those with an elevated level were randomized to either resume anticoagulation or not. Patients who resumed anticoagulation had an annual recurrence rate of 2%; however, those who were allocated not to restart anticoagulation had a recurrence rate of 10.9% per year. There was no difference in the rate of major bleeding between the two groups. Patients in this clinical trial who had a normal d-dimer level did not restart anticoagulation and had an annual recurrence rate of 4.4%.
Evaluation for residual thrombosis
A randomized trial34 in patients with both provoked and idiopathic deep vein thrombosis showed a reduction in recurrence when those who had residual vein thrombosis were given extended anticoagulation. In the subset of patients whose deep vein thrombosis was idiopathic, the recurrence rate was 17% per year when treatment lasted only 3 months and 10% when it was extended for up to 1 year.
Another trial35 randomized patients with provoked and idiopathic deep vein thrombosis to receive anticoagulation for the usual duration or to continue treatment until recanalization of the residual thrombus was demonstrated on follow-up Doppler ultrasonography. Patients who received this ultrasonography-tailored treatment had a lower rate of recurrence of VTE; however, the absolute reductions in recurrence rates cannot be calculated from this report for patients with idiopathic deep vein thrombosis.
A prospective observational study36 of the predictive value of d-dimer status and residual vein thrombus found that only d-dimer was an independent risk factor for recurrent VTE after vitamin K antagonist withdrawal.
A clinical prediction rule: ‘Men and HERDOO2’
A promising tool for predicting if a patient is at low risk of recurrent VTE after the first episode of proximal deep vein thrombosis or pulmonary embolism is known by the mnemonic device “Men and HERDOO2.” It is based on data prospectively derived by Rodger et al37 to identify patients with less than a 3% annual risk of recurrent VTE after their first event of idiopathic proximal deep vein thrombosis or pulmonary embolism. Risk factors for recurrent VTE were male sex (the “men” of “Men and HERDOO2”), signs of postthrombotic syndrome, including hyperpigmentation of the lower extremities, edema or redness of either leg, a d-dimer level > 250 μg/L, obesity (body mass index > 30 kg/m2, and older age (> 65 years).
Overall, one-fourth of the population were women with no risk factors or one risk factor, and their risk of recurrence was 1.6% per year. Men and women who had two or more risk factors for postthrombotic syndrome (hyperpigmentation, edema, or redness), elevated d-dimer, obesity, or older age were predicted to be at higher risk of recurrent VTE. Patients such as this should be considered for indefinite anticoagulation.
Ideally, clinical prediction rules should be validated in a separate group of patients before they are used routinely in practice,38 and this clinical prediction rule is currently being tested in the REVERSE II study. If the results are consistent, this will be an easy-to-use tool to help identify patients who likely can safely stop anticoagulation therapy after 3 to 6 months (clinicaltrials.gov Identifier: NCT00967304).
The location of the thrombosis also influences the likelihood of recurrence. Patients with isolated distal (calf) deep vein thrombosis are less likely to suffer recurrent VTE than those who present with proximal deep vein thrombosis. However, trials focusing specifically on the precise subset of idiopathic isolated distal deep vein thrombosis are lacking. In a randomized trial39 comparing 6 vs 12 weeks of anticoagulation for isolated distal deep vein thrombosis and 12 vs 24 weeks for proximal deep vein thrombosis, the annual rates of recurrence after 12 weeks of treatment were approximately 3.4% for isolated distal and 8.1% for proximal deep vein thrombosis.39
Recommendation: At least 3 months of warfarin or equivalent
We agree with the ACCP recommendation2 that patients with unprovoked VTE should receive at least 3 months of anticoagulation with a vitamin K antagonist.
If the patient has no risk factors for bleeding and good anticoagulant monitoring is achievable, we agree with long-term anticoagulation for proximal unprovoked deep vein thrombosis or pulmonary embolism, and 3 months of therapy for isolated distal unprovoked deep vein thrombosis.
Patient preferences and the risk of recurrence vs the risk of bleeding should be discussed with patients when contemplating indefinite anticoagulation.
If testing is being considered to assist in the decision to prescribe indefinite anticoagulation, we prefer using d-dimer levels rather than ultrasonography to detect residual venous thrombosis because of its ease of use and the strength of the current evidence.
PREVENTING POSTTHROMBOTIC SYNDROME
The postthrombotic (postphlebitic) syndrome is a chronic and burdensome consequence of deep vein thrombosis that occurs despite anticoagulation therapy. It is estimated to affect 23% to 60% of patients and typically manifests in the first 2 years.40 It is not only costly in clinical terms, with decreased quality of life for the patient, but health care expenditures have been estimated to range from $400 per year in a Brazilian study to $7,000 per year in a US study.40
Typical symptoms include leg pain, heaviness, swelling, and cramping. In severe cases, chronic venous ulcers can occur and are difficult to treat.41
The definition of postthrombotic syndrome has been unclear over the years, and six different scales that measure signs and symptoms have been reported.42
The Villalta scale has been proposed by the International Society of Thrombosis and Hemostasis as a diagnostic standard to define postthrombotic syndrome.42 This validated scale is based on five clinical symptoms, six clinical signs, and the presence or absence of venous ulcers. Each clinical symptom and sign is scored as mild (1 point), moderate (2 points), or severe (3 points). Symptoms include pain, cramps, heaviness, paresthesia, and pruritus; the six clinical signs are pretibial edema, skin induration, hyperpigmentation, redness, venous ectasia, and pain on calf compression.
According to the International Society of Thrombosis and Hemostasis, postthrombotic syndrome is present if the Villalta score is 5 or greater or if a venous ulcer is present in a leg with previous deep vein thrombosis. Further, using the Villalta scale, postthrombotic syndrome can be categorized as mild (score 5–9), moderate (10–14), or severe (≥ 15).
A limitation of the Villalta scale is that the presence or absence of a venous ulcer has not been assigned a score. Since a venous ulcer requires more aggressive measures, the society defines postthrombotic syndrome as severe if venous ulcers are present.42
Acute symptoms of deep vein thrombosis may take months to resolve and, indeed, acute symptoms may transition to chronic symptoms without a symptom-free interval. It is recommended that postthrombotic syndrome not be diagnosed before 3 months to avoid inappropriately attributing acute symptoms and signs of deep vein thrombosis to the postthrombotic syndrome.42
Studies of stockings
A systematic review of three randomized trials44 concluded that elastic compression stockings reduce the risk of postthrombotic syndrome (any severity) from 43% to 20% and severe postthrombotic syndrome from 15% to 7%.43
The first of these trials44 randomized patients soon after the diagnosis of deep vein thrombosis to receive made-to-order compression stockings that were rated at 30 to 40 mm Hg or to be in a control group that did not receive stockings. The second trial45 randomized patients 1 year after the index event of deep vein thrombosis to receive 20- to 30-mm Hg stockings or stockings that were two sizes too large (the control group). The third study46 randomly allocated patients to receive “off-the-shelf” stockings (30–40 mm Hg) or no stockings. Each study used its own definition of postthrombotic syndrome.
Although these studies strongly suggest compression stockings prevent postthrombotic syndrome, several methodologic issues remain:
- A standard definition of postthrombotic syndrome was not used
- The amount of compression varied between studies
- The studies were not blinded.
Lack of blinding becomes most significant when an outcome is based on subjective findings, like the symptoms that make up a large part of the diagnosis of postthrombotic syndrome.
The SOX trial, currently under way, is designed to address these methodologic issues and should be completed in 2012 (clinicaltrials.gov Identifier: NCT00143598).
Recommendation: Stockings for at least 2 years
We agree with the ACCP recommendation that a patient who has had a symptomatic proximal deep vein thrombosis should wear an elastic compression stocking with an ankle pressure gradient of 30 to 40 mm Hg as soon as possible after starting anticoagulant therapy and continuing for a minimum of 2 years.2
SCREENING FOR OCCULT MALIGNANCY
VTE can be the first manifestation of cancer.
French physician Armand Trousseau, in the 1860s, was the first to describe disseminated intravascular coagulation closely associated with adenocarcinoma. Ironically, several years later, after suffering for weeks from abdominal pain, he declared to one of his students that he had developed thrombosis, and he died of gastric cancer shortly thereafter.47
Since cancer is a well-known risk factor for VTE, it is logical to screen for cancer as an explanation for an idiopathic VTE event.48 To make an informed decision, one needs to understand the rate of occult cancer at the time VTE is diagnosed, the risk of future development of cancer, and the utility of extensive cancer screening.
The clinical efficacy, side effects, and cost-effectiveness of cancer screening in patients with idiopathic VTE are unknown. However, a systematic review47 of 34 studies found that, in patients with idiopathic VTE, cancer was diagnosed within 1 month in 6.1%, within 6 months in 8.6%, and within 1 year in 10.0% (95% CI 8.6–11.3).
A subset of studies compared two strategies for screening soon after the diagnosis of idiopathic VTE: a strategy limited to the history, physical examination, basic blood work, and chest radiography vs an extensive screening strategy that also included serum tumor markers or abdominal ultrasonography or computed tomography. Limited screening detected 49% of the prevalent cancers; extensive screening increased this rate to 70%. Stated another way, the detection rate for prevalent cancers was 5% with limited screening and 7% with extensive screening soon after the diagnosis of idiopathic VTE.47
Patients with idiopathic VTE had higher rates of cancer within 1 month of diagnosis than patients with provoked VTE (6.1% vs 1.9%), and this difference persisted at 1 year (10.0% vs 2.6%).47
Recommendation: Individualized cancer screening
Patients with idiopathic VTE have a significant risk of occult cancer within the first year after diagnosis, and cancer screening should be considered. Our practice for patients with idiopathic VTE is to perform a history and physical examination and ensure that the patient is up to date on age- and sex-specific cancer screening.
The use of additional imaging or biomarkers should be discussed with patients so they can balance the risks (radiation and potential false-positive results with their downstream consequences), costs, and potential benefits, given the lack of proven survival benefit or cost-effectiveness.
ORAL ANTICOAGULANT MANAGEMENT
Warfarin’s multiple interactions, along with the need for INR monitoring, make it a difficult medication to manage.
The Joint Commission, the US organization for health service accreditation and certification, has defined National Patient Safety Goals and quality measures for the management of anticoagulation.49 Organized anticoagulation management services, dosing algorithms, and patient self-testing using capillary INR meters or patient self-management of warfarin were recommended as tools to improve the time patients spend in the therapeutic INR range.50
Two new oral anticoagulants
The limitations of warfarin have stimulated the search for newer oral anticoagulants that do not require laboratory monitoring or have as many diet and drug interactions.
Two trials have been published with experimental oral anticoagulants that had similar efficacy and safety as warfarin in the treatment of VTE.
The study of dabigatran (Pradaxa) vs warfarin in the treatment of acute VTE (the RECOVER trial)51 randomized 2,539 patients with acute VTE to receive the oral direct thrombin inhibitor dabigatran or warfarin for approximately 6 months. Of note, each treatment group received a median of 6 days of heparin, LMWH, or fondaparinux at the beginning of blinded therapy. The rates of recurrent VTE and major bleeding were similar between the treatment arms, and overall bleeding was less with dabigatran. Dabigatran was approved in the United States in October 2010 for stroke prevention in atrial fibrillation but has yet to be approved for the treatment of VTE pending further study (clinicaltrials.gov Identifier: NCT00680186).
A study of oral rivaroxaban (Xarelto) for symptomatic VTE (the EINSTEIN-DVT trial) 52 randomized 3,449 patients with acute deep vein thrombosis to rivaroxaban or enoxaparin (Lovenox) overlapped with warfarin or another vitamin K antagonist in the usual manner. No difference was noted between the treatments in the rate of recurrence of VTE or of major bleeding. Of note, patients randomized to rivaroxaban received 15 mg twice a day for the first 3 weeks of treatment and then 20 mg per day for the remainder of their therapy and did not require parenteral anticoagulant overlap.
The long-awaited promise of easier-to-use oral anticoagulants for the treatment of VTE is drawing near and has the potential to revolutionize the treatment of this common disorder. In the meantime, close monitoring of warfarin and careful patient education regarding its use are essential. And even with the development of new drugs in the future, it is still imperative that patients with acute VTE receive the correct length of anticoagulation treatment, are prescribed stockings to prevent postthrombotic syndrome, and are updated on routine cancer screening.
- Spencer FA, Emery C, Lessard D, et al. The Worcester Venous Thromboembolism study: a population-based study of the clinical epidemiology of venous thromboembolism. J Gen Intern Med 2006; 21:722–727.
- Kearon C, Kahn SR, Agnelli G, Goldhaber S, Raskob GE, Comerota AJ; American College of Chest Physicians. Antithrombotic therapy for venous thromboembolic disease: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest 2008; 133(suppl 6):454S–545S.
- Baglin T, Luddington R, Brown K, Baglin C. Incidence of recurrent venous thromboembolism in relation to clinical and thrombophilic risk factors: prospective cohort study. Lancet 2003; 362:523–526.
- Schulman S, Lockner D, Juhlin-Dannfelt A. The duration of oral anticoagulation after deep vein thrombosis. A randomized study. Acta Med Scand 1985; 217:547–552.
- Optimum duration of anticoagulation for deep-vein thrombosis and pulmonary embolism. Research Committee of the British Thoracic Society. Lancet 1992; 340:873–876.
- Schulman S, Rhedin AS, Lindmarker P, et al. A comparison of six weeks with six months of oral anticoagulant therapy after a first episode of venous thromboembolism. Duration of Anticoagulation Trial Study Group. N Engl J Med 1995; 332:1661–1665.
- Kearon C, Ginsberg JS, Anderson DR, et al. Comparison of 1 month with 3 months of anticoagulation for a first episode of venous thromboembolism associated with a transient risk factor. J Thromb Haemost 2004; 2:743–749.
- Iorio A, Kearon C, Filippucci E, et al. Risk of recurrence after a first episode of symptomatic venous thromboembolism provoked by a transient risk factor: a systematic review. Arch Intern Med 2010; 170:1710–1716.
- Prandoni P, Lensing AW, Piccioli A, et al. Recurrent venous thromboembolism and bleeding complications during anticoagulant treatment in patients with cancer and venous thrombosis. Blood 2002; 100:3484–3488.
- Hull RD, Pineo GF, Brant RF, et al; LITE Trial Investigators. Long-term low-molecular-weight heparin versus usual care in proximal-vein thrombosis patients with cancer. Am J Med 2006; 119:1062–1072.
- Lee AY, Levine MN, Baker RI, et al; Randomized Comparison of Low-Molecular-Weight Heparin versus Oral Anticoagulant Therapy for the Prevention of Recurrent Venous Thromboembolism in Patients with Cancer (CLOT) Investigators. Low-molecular-weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N Engl J Med 2003; 349:146–153.
- National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology, Venous Thromboembolic Disease. http://www.nccn.org/professionals/physician_gls/pdf/vte.pdf. Accessed August 3, 2011.
- Lyman GH, Khorana AA, Falanga A, et al; American Society of Clinical Oncology. American Society of Clinical Oncology guideline: recommendations for venous thromboembolism prophylaxis and treatment in patients with cancer. J Clin Oncol 2007; 25:5490–5505.
- Schulman S, Granqvist S, Holmström M, et al. The duration of oral anticoagulant therapy after a second episode of venous thromboembolism. The Duration of Anticoagulation Trial Study Group. N Engl J Med 1997; 336:393–398.
- Christiansen SC, Cannegieter SC, Koster T, Vandenbroucke JP, Rosendaal FR. Thrombophilia, clinical factors, and recurrent venous thrombotic events. JAMA 2005; 293:2352–2361.
- Segal JB, Brotman DJ, Necochea AJ, et al. Predictive value of factor V Leiden and prothrombin G20210A in adults with venous thromboembolism and in family members of those with a mutation: a systematic review. JAMA 2009; 301:2472–2485.
- Brouwer JL, Lijfering WM, Ten Kate MK, Kluin-Nelemans HC, Veeger NJ, van der Meer J. High long-term absolute risk of recurrent venous thromboembolism in patients with hereditary deficiencies of protein S, protein C or antithrombin. Thromb Haemost 2009; 101:93–99.
- Schulman S, Svenungsson E, Granqvist S. Anticardiolipin antibodies predict early recurrence of thromboembolism and death among patients with venous thromboembolism following anticoagulant therapy. Duration of Anticoagulation Study Group. Am J Med 1998; 104:332–338.
- Derksen RH, de Groot PG. Towards evidence-based treatment of thrombotic antiphospholipid syndrome. Lupus 2010; 19:470–474.
- Lim W, Crowther MA, Eikelboom JW. Management of antiphospholipid antibody syndrome: a systematic review. JAMA 2006; 295:1050–1057.
- Fonseca AG, D’Cruz DP. Controversies in the antiphospholipid syndrome: can we ever stop warfarin? J Autoimmune Dis 2008; 5:6.
- Crowther MA, Ginsberg JS, Julian J, et al. A comparison of two intensities of warfarin for the prevention of recurrent thrombosis in patients with the antiphospholipid antibody syndrome. N Engl J Med 2003; 349:1133–1138.
- Finazzi G, Marchioli R, Brancaccio V, et al. A randomized clinical trial of high-intensity warfarin vs. conventional antithrombotic therapy for the prevention of recurrent thrombosis in patients with the antiphospholipid syndrome (WAPS). J Thromb Haemost 2005; 3:848–853.
- Agnelli G, Prandoni P, Becattini C, et al; Warfarin Optimal Duration Italian Trial Investigators. Extended oral anticoagulant therapy after a first episode of pulmonary embolism. Ann Intern Med 2003; 139:19–25.
- Agnelli G, Prandoni P, Santamaria MG, et al; Warfarin Optimal Duration Italian Trial Investigators. Three months versus one year of oral anticoagulant therapy for idiopathic deep venous thrombosis. Warfarin Optimal Duration Italian Trial Investigators. N Engl J Med 2001; 345:165–169.
- Kearon C, Gent M, Hirsh J, et al. A comparison of three months of anticoagulation with extended anticoagulation for a first episode of idiopathic venous thromboembolism. N Engl J Med 1999; 340:901–907.
- Kearon C, Ginsberg JS, Kovacs MJ, et al; Extended Low-Intensity Anticoagulation for Thrombo-Embolism Investigators. Comparison of low-intensity warfarin therapy with conventional-intensity warfarin therapy for long-term prevention of recurrent venous thromboembolism. N Engl J Med 2003; 349:631–639.
- Palareti G, Cosmi B, Legnani C, et al; PROLONG Investigators. D-dimer testing to determine the duration of anticoagulation therapy. N Engl J Med 2006; 355:1780–1789.
- Ridker PM, Goldhaber SZ, Glynn RJ. Low-intensity versus conventional-intensity warfarin for prevention of recurrent venous thromboembolism. N Engl J Med 2003; 349:2164–2167.
- Bockenstedt P. D-dimer in venous thromboembolism. N Engl J Med 2003; 349:1203–1204.
- Verhovsek M, Douketis JD, Yi Q, et al. Systematic review: D-dimer to predict recurrent disease after stopping anticoagulant therapy for unprovoked venous thromboembolism. Ann Intern Med 2008; 149:481–490,W94.
- Douketis J, Tosetto A, Marcucci M, et al. Patient-level metaanalysis: effect of measurement timing, threshold, and patient age on ability of D-dimer testing to assess recurrence risk after unprovoked venous thromboembolism. Ann Intern Med 2010; 153:523–531.
- Prandoni P, Lensing AW, Prins MH, et al. Residual venous thrombosis as a predictive factor of recurrent venous thromboembolism. Ann Intern Med 2002; 137:955–960.
- Siragusa S, Malato A, Anastasio R, et al. Residual vein thrombosis to establish duration of anticoagulation after a first episode of deep vein thrombosis: the Duration of Anticoagulation based on Compression UltraSonography (DACUS) study. Blood 2008; 112:511–515.
- Prandoni P, Prins MH, Lensing AW, et al; AESOPUS Investigators. Residual thrombosis on ultrasonography to guide the duration of anticoagulation in patients with deep venous thrombosis: a randomized trial. Ann Intern Med 2009; 150:577–585.
- Cosmi B, Legnani C, Cini M, Guazzaloca G, Palareti G. D-dimer levels in combination with residual venous obstruction and the risk of recurrence after anticoagulation withdrawal for a first idiopathic deep vein thrombosis. Thromb Haemost 2005; 94:969–974.
- Rodger MA, Kahn SR, Wells PS, et al. Identifying unprovoked thromboembolism patients at low risk for recurrence who can discontinue anticoagulant therapy. CMAJ 2008; 179:417–426.
- McGinn TG, Guyatt GH, Wyer PC, Naylor CD, Stiell IG, Richardson WS. Users’ guides to the medical literature: XXII: how to use articles about clinical decision rules. Evidence-Based Medicine Working Group. JAMA 2000; 284:79–84.
- Pinede L, Ninet J, Duhaut P, et al; Investigators of the “Durée Optimale du Traitement AntiVitamines K” (DOTAVK) Study. Comparison of 3 and 6 months of oral anticoagulant therapy after a first episode of proximal deep vein thrombosis or pulmonary embolism and comparison of 6 and 12 weeks of therapy after isolated calf deep vein thrombosis. Circulation 2001; 103:2453–2460.
- Ashrani AA, Heit JA. Incidence and cost burden of postthrombotic syndrome. J Thromb Thrombolysis 2009; 28:465–476.
- Kahn SR, Shrier I, Julian JA, et al. Determinants and time course of the postthrombotic syndrome after acute deep venous thrombosis. Ann Intern Med 2008; 149:698–707.
- Kahn SR, Partsch H, Vedantham S, Prandoni P, Kearon C; Subcommittee on Control of Anticoagulation of the Scientific and Standardization Committee of the International Society on Thrombosis and Haemostasis. Definition of post-thrombotic syndrome of the leg for use in clinical investigations: a recommendation for standardization. J Thromb Haemost 2009; 7:879–883.
- Kolbach DN, Sandbrink MW, Hamulyak K, Neumann HA, Prins MH. Non-pharmaceutical measures for prevention of post-thrombotic syndrome. Cochrane Database Syst Rev 2004;CD004174.
- Brandjes DP, Büller HR, Heijboer H, et al. Randomised trial of effect of compression stockings in patients with symptomatic proximal-vein thrombosis. Lancet 1997; 349:759–762.
- Ginsberg JS, Hirsh J, Julian J, et al. Prevention and treatment of postphlebitic syndrome: results of a 3-part study. Arch Intern Med 2001; 161:2105–2109.
- Prandoni P, Lensing AW, Prins MH, et al. Below-knee elastic compression stockings to prevent the post-thrombotic syndrome: a randomized, controlled trial. Ann Intern Med 2004; 141:249–256.
- Carrier M, Le Gal G, Wells PS, Fergusson D, Ramsay T, Rodger MA. Systematic review: the Trousseau syndrome revisited: should we screen extensively for cancer in patients with venous thromboembolism? Ann Intern Med 2008; 149:323–333.
- Blom JW, Doggen CJ, Osanto S, Rosendaal FR. Malignancies, prothrombotic mutations, and the risk of venous thrombosis. JAMA 2005; 293:715–722.
- Kaatz S. Impact on patient care: patient case through the continuum of care. J Thromb Thrombolysis 2010; 29:167–170.
- Ansell J, Hirsh J, Hylek E, Jacobson A, Crowther M, Palareti G; American College of Chest Physicians. Pharmacology and management of the vitamin K antagonists: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest 2008; 133(suppl 6):160S–198S.
- Schulman S, Kearon C, Kakkar AK, et al; for the RE-COVER Study Group. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med 2009; 361:2342–2452.
- The EINSTEIN Investigators. Oral rivaroxaban for symptomatic venous thromboembolism. N Engl J Med 2010; 363;2499–2510.
Deep vein thrombosis and pulmonary embolism are collectively referred to as venous thromboembolic (VTE) disease. They affect approximately 100,000 to 300,000 patients per year in the United States.1 Although patients with deep vein thrombosis can be treated as outpatients, many are admitted for the initiation of anticoagulation. Initial anticoagulation usually requires the overlap of a parenteral anticoagulant (unfractionated heparin, low-molecular-weight heparin [LMWH] or fondaparinux) with warfarin for a minimum of 5 days and until the international normalized ratio (INR) of the prothrombin time is above 2.0 for at least 24 hours.2
Three clinical issues need to be addressed after the initiation of anticoagulation for VTE:
- Determination of the length of anticoagulation with the correct anticoagulant
- Prevention of postthrombotic syndrome
- Appropriate screening for occult malignancy.
HOW LONG SHOULD VTE BE TREATED?
The duration of anticoagulation has been a matter of debate.
The risk of recurrent VTE appears related to clinical risk factors that a patient has at the time of the initial thrombotic event. An epidemiologic study3 found that patients with VTE treated for approximately 6 months had a low rate of recurrence (0% at 2 years of follow-up) if surgery was the risk factor. The risk climbed to 9% if the risk factor was nonsurgical and to 19% if there were no provoking risk factors.
The likelihood of VTE recurrence and therefore the recommended duration of treatment depend on whether the VTE event was provoked, cancer-related, recurrent, thrombophilia-related, or idiopathic. We address each of these scenarios below.
HOW LONG TO TREAT PROVOKED VTE
A VTE event is considered provoked if the patient had a clear inciting risk factor. As defined in various clinical trials, these risk factors include:
- Hospitalization with confinement to bed for 3 or more consecutive days in the last 3 months
- Surgery or general anesthesia in the last 3 months
- Immobilization for more than 7 days, regardless of the cause
- Trauma in the last 3 months
- Pregnancy
- Use of an oral contraceptive, regardless of which estrogen or progesterone analogue it contains
- Travel for more than 4 hours
- Recent childbirth.
However, the trials that tested different lengths of anticoagulation have varied markedly in how they defined provoked deep vein thrombosis.4–7
Recommendation: Warfarin or equivalent for 3 months
The American College of Chest Physicians (ACCP) recommends 3 months of anticoagulation with warfarin or another vitamin K antagonist for patients with VTE secondary to a transient (reversible) risk factor,2 and we agree.
HOW LONG TO TREAT CANCER-RELATED VTE
Patients with cancer are at higher risk of developing VTE. Furthermore, in one study,9 compared with other patients with VTE, patients with cancer were three times more likely to have another episode of VTE, with a cumulative rate of recurrence at 1 year of 21% vs 7%. Cancer patients were also twice as likely to suffer major bleeding complications while on anticoagulation.9
Warfarin is a difficult drug to manage because it has many interactions with foods, diseases, and other drugs. These difficulties are amplified in many cancer patients during chemotherapy.
Warfarin was compared with a LMWH in four randomized trials in cancer patients, and a meta-analysis10 found a 50% relative reduction in the rates of recurrent deep vein thrombosis and pulmonary embolism with LMWH treatment. These results were driven primarily by the CLOT trial (Comparison of Low-Molecular-Weight Heparin Versus Oral Anticoagulant Therapy for the Prevention of Recurrent Venous Thromboembolism in Patients With Cancer),11 which showed an 8% absolute risk reduction (number needed to treat 13) without an increase in major bleeding when cancer-related VTE was treated with an LMWH—ie, dalteparin (Fragmin)—for 6 months compared with warfarin.
Current thinking suggests that VTE should be treated until the cancer is resolved. However, this hypothesis has not been adequately tested, and consequently, the ACCP gives it only a level 1C recommendation.2 The largest of the four trials comparing warfarin and an LMWH lasted only 6 months. The safety of extending LMWH treatment beyond 6 months is currently unknown but is under investigation (clinicaltrials.gov identifier NCT00942968).
Recommendation: LMWH therapy for at least 6 months
The ACCP guidelines recommend LMWH therapy for 3 to 6 months, followed by warfarin or another vitamin K antagonist or continued LMWH treatment until the cancer is resolved.2
The National Comprehensive Cancer Network guidelines recommend an LMWH for 6 months as monotherapy and indefinite anticoagulation if the cancer is still active.12
The American Society of Clinical Oncology guidelines recommend an LMWH for at least 6 months and indefinite anticoagulant therapy for selected patients with active cancer.13
We agree that patients with active cancer should receive an LMWH for at least 6 months and indefinite anticoagulation until the cancer is resolved.
In our experience, many patients are reluctant to give themselves the daily injections that LMWH therapy requires, and so they need to be well-informed about the marked decrease in VTE recurrence with this less-convenient and more-expensive therapy. Many patients face insurance barriers to cover the cost of LMWH therapy; however, careful attention to preauthorization can usually overcome this obstacle.
HOW LONG TO TREAT RECURRENT VTE
It makes clinical sense that patients who have a second VTE event should be treated indefinitely. This theory was tested in a randomized clinical trial14 in which patients with provoked or unprovoked VTE were randomized after their second event to receive anticoagulation for 6 months vs indefinitely.
After 4 years of follow-up, the recurrence rate was 21% in patients assigned to 6 months of treatment and only 3% in patients who continued anticoagulation throughout the trial. On the other hand, major hemorrhage occurred in 3% of patients treated for 6 months and in 9% in patients who continued anticoagulation indefinitely.
Of note, most of the patients in this trial had unprovoked (idiopathic) VTE, so the results should not be extrapolated to patients with provoked VTE, who accounted for only 20% of the study population.14
Recommendation: Long-term anticoagulation
We agree with the ACCP recommendation2 that patients who have had a second episode of unprovoked VTE should receive long-term anticoagulation. Because of a lack of data, the duration of therapy for patients with a second episode of provoked VTE should be individualized.
HOW LONG TO TREAT THROMBOPHILIA-RELATED VTE
Inherited thrombophilias
Patients with VTE that is not related to a clear provoking risk factor or cancer frequently have testing to evaluate for a hypercoagulable state. This workup traditionally includes the most common inherited thrombophilias for gene mutations for factor V and prothrombin as well as for deficiencies in protein C, protein S, antithrombin and the acquired antiphospholipid syndrome.
The key questions that should be asked prior to embarking on this workup are:
- Will the results change the length of therapy for the patient?
- Will testing the patient help with genetic counseling and possible testing of family members?
- Will the results change the targeted INR range for warfarin or other vitamin K antagonist therapy?
Patients with inherited thrombophilia have a greater risk of developing an initial VTE event; however, these tests do not help predict the recurrence of VTE in patients with established disease more than clinical risk factors do. A prospective study demonstrated this by looking at the effect of thrombophilia and clinical factors on the recurrence of venous thrombosis and found that inherited prothrombotic abnormalities do not appear to play an important role in the risk of a recurrent event.15 On the other hand, clinical factors, such as whether the first event was idiopathic or provoked, appear more important in determining the duration of anticoagulation therapy.15 A systematic review of the common inherited thrombophilias showed the VTE recurrence rate of patients with factor V Leiden was higher than in patients without the mutation; however, the absolute rates of recurrence were not much different than what would be expected in patients with idiopathic VTE.16
A retrospective study involving a large cohort of families of patients who already had experienced a first episode of either idiopathic or provoked VTE showed high annual risks of recurrent VTE associated with hereditary deficiencies of protein S (8.4%), protein C (6.0%), and antithrombin (10%).17 However, for the more commonly occurring genetic thrombophilias, the factor V Leiden and prothrombin G20210A mutations, family members with either gene abnormality had low rates of VTE, suggesting that testing of relatives of probands is not clinically useful.16
Antiphospholipid syndrome
Antiphospholipid syndrome is an acquired thrombophilia. A patient has thrombotic antiphospholipid syndrome when there is a history of vascular thrombosis in the presence of persistently positive tests (at least 12 weeks apart) for lupus anticoagulants, anticardiolipin antibodies, or anti-beta-2 glycoprotein I. A prospective study of 412 patients with a first episode of VTE found that 15% were positive for anticardiolipin antibody at the end of 6 months of anticoagulation. The risk of recurrent VTE after 4 years was 29% in patients with antibodies and 14% in those without antibodies (relative risk 2.1; 95% confidence interval [CI] 1.3–3.3; P =.0013).18
Recent reviews advise indefinite warfarin anticoagulation in patients with VTE and persistence of antiphospholipid antibodies.19 However, the optimal duration of anticoagulation is uncertain. Until well-designed clinical trials are done, the current general consensus is to anticoagulate these patients indefinitely.20,21 Retrospective studies had suggested that patients with antiphospholipid antibodies required a higher therapeutic INR range; however, this observation was tested in two trials that found no difference in thromboembolic rates when patients were randomized to an INR of 2.0–3.0 vs 3.1–4.0,22 or 2.0–3.0 vs 3.0–4.5.23
No formal recommendations
In the absence of strong evidence, the ACCP guidelines do not include a recommendation on the duration of anticoagulation treatment specific to inherited thrombophilias. We believe that clinical factors are more important than inherited thrombophilias for deciding the duration of anticoagulation, and that testing is almost never indicated or useful. However, patients with antiphospholipid syndrome are at high risk of recurrence, and it is our practice to anticoagulate these patients indefinitely.
HOW LONG TO TREAT UNPROVOKED (IDIOPATHIC) VTE
A VTE event is thought to be idiopathic if it occurs without a clearly identified provoking factor.
In an observational study,3 patients with oral contraceptive use, transient illness, immobilization, or a history of travel had an 8.8% risk of recurrence vs 19.4% in patients with unprovoked VTE. The meta-analysis discussed above (Table 1)8 also shows that patients with these nonsurgical risk factors have a lower rate of recurrence than patients with idiopathic VTE.
The high rate of recurrence of idiopathic VTE (4% to 27% after 3 months of anticoagulation24–26) suggests that a longer duration of treatment is reasonable. However, increasing the length of therapy from 3 to 12 months delays but does not prevent recurrence, the risk of which begins to accumulate once anticoagulation is stopped.24,25
Three promising strategies to identify subgroups of patients with idiopathic VTE who are at highest risk of recurrence and who would benefit the most from prolonged anticoagulation are d-dimer testing, evaluation for residual vein thrombosis in patients who present with a deep vein thrombosis, and clinical prediction rules.
d-dimer testing
d-dimer is a degradation product of fibrin and is an indirect marker of residual thrombosis.30
In a systematic review of patients with a first episode of unprovoked VTE,31 a normal d-dimer concentration at the end of at least 3 months of anticoagulation was associated with a 3.5% annual risk of recurrence, whereas an elevated d-dimer level at that time was associated with an annual risk of 8.9%. These results were confirmed in a systematic review of individual patient data.32
In a randomized trial,28 patients with an idiopathic VTE event who received anticoagulation for at least 3 months had their d-dimer level measured 1 month after cessation of treatment. Those with an elevated level were randomized to either resume anticoagulation or not. Patients who resumed anticoagulation had an annual recurrence rate of 2%; however, those who were allocated not to restart anticoagulation had a recurrence rate of 10.9% per year. There was no difference in the rate of major bleeding between the two groups. Patients in this clinical trial who had a normal d-dimer level did not restart anticoagulation and had an annual recurrence rate of 4.4%.
Evaluation for residual thrombosis
A randomized trial34 in patients with both provoked and idiopathic deep vein thrombosis showed a reduction in recurrence when those who had residual vein thrombosis were given extended anticoagulation. In the subset of patients whose deep vein thrombosis was idiopathic, the recurrence rate was 17% per year when treatment lasted only 3 months and 10% when it was extended for up to 1 year.
Another trial35 randomized patients with provoked and idiopathic deep vein thrombosis to receive anticoagulation for the usual duration or to continue treatment until recanalization of the residual thrombus was demonstrated on follow-up Doppler ultrasonography. Patients who received this ultrasonography-tailored treatment had a lower rate of recurrence of VTE; however, the absolute reductions in recurrence rates cannot be calculated from this report for patients with idiopathic deep vein thrombosis.
A prospective observational study36 of the predictive value of d-dimer status and residual vein thrombus found that only d-dimer was an independent risk factor for recurrent VTE after vitamin K antagonist withdrawal.
A clinical prediction rule: ‘Men and HERDOO2’
A promising tool for predicting if a patient is at low risk of recurrent VTE after the first episode of proximal deep vein thrombosis or pulmonary embolism is known by the mnemonic device “Men and HERDOO2.” It is based on data prospectively derived by Rodger et al37 to identify patients with less than a 3% annual risk of recurrent VTE after their first event of idiopathic proximal deep vein thrombosis or pulmonary embolism. Risk factors for recurrent VTE were male sex (the “men” of “Men and HERDOO2”), signs of postthrombotic syndrome, including hyperpigmentation of the lower extremities, edema or redness of either leg, a d-dimer level > 250 μg/L, obesity (body mass index > 30 kg/m2, and older age (> 65 years).
Overall, one-fourth of the population were women with no risk factors or one risk factor, and their risk of recurrence was 1.6% per year. Men and women who had two or more risk factors for postthrombotic syndrome (hyperpigmentation, edema, or redness), elevated d-dimer, obesity, or older age were predicted to be at higher risk of recurrent VTE. Patients such as this should be considered for indefinite anticoagulation.
Ideally, clinical prediction rules should be validated in a separate group of patients before they are used routinely in practice,38 and this clinical prediction rule is currently being tested in the REVERSE II study. If the results are consistent, this will be an easy-to-use tool to help identify patients who likely can safely stop anticoagulation therapy after 3 to 6 months (clinicaltrials.gov Identifier: NCT00967304).
The location of the thrombosis also influences the likelihood of recurrence. Patients with isolated distal (calf) deep vein thrombosis are less likely to suffer recurrent VTE than those who present with proximal deep vein thrombosis. However, trials focusing specifically on the precise subset of idiopathic isolated distal deep vein thrombosis are lacking. In a randomized trial39 comparing 6 vs 12 weeks of anticoagulation for isolated distal deep vein thrombosis and 12 vs 24 weeks for proximal deep vein thrombosis, the annual rates of recurrence after 12 weeks of treatment were approximately 3.4% for isolated distal and 8.1% for proximal deep vein thrombosis.39
Recommendation: At least 3 months of warfarin or equivalent
We agree with the ACCP recommendation2 that patients with unprovoked VTE should receive at least 3 months of anticoagulation with a vitamin K antagonist.
If the patient has no risk factors for bleeding and good anticoagulant monitoring is achievable, we agree with long-term anticoagulation for proximal unprovoked deep vein thrombosis or pulmonary embolism, and 3 months of therapy for isolated distal unprovoked deep vein thrombosis.
Patient preferences and the risk of recurrence vs the risk of bleeding should be discussed with patients when contemplating indefinite anticoagulation.
If testing is being considered to assist in the decision to prescribe indefinite anticoagulation, we prefer using d-dimer levels rather than ultrasonography to detect residual venous thrombosis because of its ease of use and the strength of the current evidence.
PREVENTING POSTTHROMBOTIC SYNDROME
The postthrombotic (postphlebitic) syndrome is a chronic and burdensome consequence of deep vein thrombosis that occurs despite anticoagulation therapy. It is estimated to affect 23% to 60% of patients and typically manifests in the first 2 years.40 It is not only costly in clinical terms, with decreased quality of life for the patient, but health care expenditures have been estimated to range from $400 per year in a Brazilian study to $7,000 per year in a US study.40
Typical symptoms include leg pain, heaviness, swelling, and cramping. In severe cases, chronic venous ulcers can occur and are difficult to treat.41
The definition of postthrombotic syndrome has been unclear over the years, and six different scales that measure signs and symptoms have been reported.42
The Villalta scale has been proposed by the International Society of Thrombosis and Hemostasis as a diagnostic standard to define postthrombotic syndrome.42 This validated scale is based on five clinical symptoms, six clinical signs, and the presence or absence of venous ulcers. Each clinical symptom and sign is scored as mild (1 point), moderate (2 points), or severe (3 points). Symptoms include pain, cramps, heaviness, paresthesia, and pruritus; the six clinical signs are pretibial edema, skin induration, hyperpigmentation, redness, venous ectasia, and pain on calf compression.
According to the International Society of Thrombosis and Hemostasis, postthrombotic syndrome is present if the Villalta score is 5 or greater or if a venous ulcer is present in a leg with previous deep vein thrombosis. Further, using the Villalta scale, postthrombotic syndrome can be categorized as mild (score 5–9), moderate (10–14), or severe (≥ 15).
A limitation of the Villalta scale is that the presence or absence of a venous ulcer has not been assigned a score. Since a venous ulcer requires more aggressive measures, the society defines postthrombotic syndrome as severe if venous ulcers are present.42
Acute symptoms of deep vein thrombosis may take months to resolve and, indeed, acute symptoms may transition to chronic symptoms without a symptom-free interval. It is recommended that postthrombotic syndrome not be diagnosed before 3 months to avoid inappropriately attributing acute symptoms and signs of deep vein thrombosis to the postthrombotic syndrome.42
Studies of stockings
A systematic review of three randomized trials44 concluded that elastic compression stockings reduce the risk of postthrombotic syndrome (any severity) from 43% to 20% and severe postthrombotic syndrome from 15% to 7%.43
The first of these trials44 randomized patients soon after the diagnosis of deep vein thrombosis to receive made-to-order compression stockings that were rated at 30 to 40 mm Hg or to be in a control group that did not receive stockings. The second trial45 randomized patients 1 year after the index event of deep vein thrombosis to receive 20- to 30-mm Hg stockings or stockings that were two sizes too large (the control group). The third study46 randomly allocated patients to receive “off-the-shelf” stockings (30–40 mm Hg) or no stockings. Each study used its own definition of postthrombotic syndrome.
Although these studies strongly suggest compression stockings prevent postthrombotic syndrome, several methodologic issues remain:
- A standard definition of postthrombotic syndrome was not used
- The amount of compression varied between studies
- The studies were not blinded.
Lack of blinding becomes most significant when an outcome is based on subjective findings, like the symptoms that make up a large part of the diagnosis of postthrombotic syndrome.
The SOX trial, currently under way, is designed to address these methodologic issues and should be completed in 2012 (clinicaltrials.gov Identifier: NCT00143598).
Recommendation: Stockings for at least 2 years
We agree with the ACCP recommendation that a patient who has had a symptomatic proximal deep vein thrombosis should wear an elastic compression stocking with an ankle pressure gradient of 30 to 40 mm Hg as soon as possible after starting anticoagulant therapy and continuing for a minimum of 2 years.2
SCREENING FOR OCCULT MALIGNANCY
VTE can be the first manifestation of cancer.
French physician Armand Trousseau, in the 1860s, was the first to describe disseminated intravascular coagulation closely associated with adenocarcinoma. Ironically, several years later, after suffering for weeks from abdominal pain, he declared to one of his students that he had developed thrombosis, and he died of gastric cancer shortly thereafter.47
Since cancer is a well-known risk factor for VTE, it is logical to screen for cancer as an explanation for an idiopathic VTE event.48 To make an informed decision, one needs to understand the rate of occult cancer at the time VTE is diagnosed, the risk of future development of cancer, and the utility of extensive cancer screening.
The clinical efficacy, side effects, and cost-effectiveness of cancer screening in patients with idiopathic VTE are unknown. However, a systematic review47 of 34 studies found that, in patients with idiopathic VTE, cancer was diagnosed within 1 month in 6.1%, within 6 months in 8.6%, and within 1 year in 10.0% (95% CI 8.6–11.3).
A subset of studies compared two strategies for screening soon after the diagnosis of idiopathic VTE: a strategy limited to the history, physical examination, basic blood work, and chest radiography vs an extensive screening strategy that also included serum tumor markers or abdominal ultrasonography or computed tomography. Limited screening detected 49% of the prevalent cancers; extensive screening increased this rate to 70%. Stated another way, the detection rate for prevalent cancers was 5% with limited screening and 7% with extensive screening soon after the diagnosis of idiopathic VTE.47
Patients with idiopathic VTE had higher rates of cancer within 1 month of diagnosis than patients with provoked VTE (6.1% vs 1.9%), and this difference persisted at 1 year (10.0% vs 2.6%).47
Recommendation: Individualized cancer screening
Patients with idiopathic VTE have a significant risk of occult cancer within the first year after diagnosis, and cancer screening should be considered. Our practice for patients with idiopathic VTE is to perform a history and physical examination and ensure that the patient is up to date on age- and sex-specific cancer screening.
The use of additional imaging or biomarkers should be discussed with patients so they can balance the risks (radiation and potential false-positive results with their downstream consequences), costs, and potential benefits, given the lack of proven survival benefit or cost-effectiveness.
ORAL ANTICOAGULANT MANAGEMENT
Warfarin’s multiple interactions, along with the need for INR monitoring, make it a difficult medication to manage.
The Joint Commission, the US organization for health service accreditation and certification, has defined National Patient Safety Goals and quality measures for the management of anticoagulation.49 Organized anticoagulation management services, dosing algorithms, and patient self-testing using capillary INR meters or patient self-management of warfarin were recommended as tools to improve the time patients spend in the therapeutic INR range.50
Two new oral anticoagulants
The limitations of warfarin have stimulated the search for newer oral anticoagulants that do not require laboratory monitoring or have as many diet and drug interactions.
Two trials have been published with experimental oral anticoagulants that had similar efficacy and safety as warfarin in the treatment of VTE.
The study of dabigatran (Pradaxa) vs warfarin in the treatment of acute VTE (the RECOVER trial)51 randomized 2,539 patients with acute VTE to receive the oral direct thrombin inhibitor dabigatran or warfarin for approximately 6 months. Of note, each treatment group received a median of 6 days of heparin, LMWH, or fondaparinux at the beginning of blinded therapy. The rates of recurrent VTE and major bleeding were similar between the treatment arms, and overall bleeding was less with dabigatran. Dabigatran was approved in the United States in October 2010 for stroke prevention in atrial fibrillation but has yet to be approved for the treatment of VTE pending further study (clinicaltrials.gov Identifier: NCT00680186).
A study of oral rivaroxaban (Xarelto) for symptomatic VTE (the EINSTEIN-DVT trial) 52 randomized 3,449 patients with acute deep vein thrombosis to rivaroxaban or enoxaparin (Lovenox) overlapped with warfarin or another vitamin K antagonist in the usual manner. No difference was noted between the treatments in the rate of recurrence of VTE or of major bleeding. Of note, patients randomized to rivaroxaban received 15 mg twice a day for the first 3 weeks of treatment and then 20 mg per day for the remainder of their therapy and did not require parenteral anticoagulant overlap.
The long-awaited promise of easier-to-use oral anticoagulants for the treatment of VTE is drawing near and has the potential to revolutionize the treatment of this common disorder. In the meantime, close monitoring of warfarin and careful patient education regarding its use are essential. And even with the development of new drugs in the future, it is still imperative that patients with acute VTE receive the correct length of anticoagulation treatment, are prescribed stockings to prevent postthrombotic syndrome, and are updated on routine cancer screening.
Deep vein thrombosis and pulmonary embolism are collectively referred to as venous thromboembolic (VTE) disease. They affect approximately 100,000 to 300,000 patients per year in the United States.1 Although patients with deep vein thrombosis can be treated as outpatients, many are admitted for the initiation of anticoagulation. Initial anticoagulation usually requires the overlap of a parenteral anticoagulant (unfractionated heparin, low-molecular-weight heparin [LMWH] or fondaparinux) with warfarin for a minimum of 5 days and until the international normalized ratio (INR) of the prothrombin time is above 2.0 for at least 24 hours.2
Three clinical issues need to be addressed after the initiation of anticoagulation for VTE:
- Determination of the length of anticoagulation with the correct anticoagulant
- Prevention of postthrombotic syndrome
- Appropriate screening for occult malignancy.
HOW LONG SHOULD VTE BE TREATED?
The duration of anticoagulation has been a matter of debate.
The risk of recurrent VTE appears related to clinical risk factors that a patient has at the time of the initial thrombotic event. An epidemiologic study3 found that patients with VTE treated for approximately 6 months had a low rate of recurrence (0% at 2 years of follow-up) if surgery was the risk factor. The risk climbed to 9% if the risk factor was nonsurgical and to 19% if there were no provoking risk factors.
The likelihood of VTE recurrence and therefore the recommended duration of treatment depend on whether the VTE event was provoked, cancer-related, recurrent, thrombophilia-related, or idiopathic. We address each of these scenarios below.
HOW LONG TO TREAT PROVOKED VTE
A VTE event is considered provoked if the patient had a clear inciting risk factor. As defined in various clinical trials, these risk factors include:
- Hospitalization with confinement to bed for 3 or more consecutive days in the last 3 months
- Surgery or general anesthesia in the last 3 months
- Immobilization for more than 7 days, regardless of the cause
- Trauma in the last 3 months
- Pregnancy
- Use of an oral contraceptive, regardless of which estrogen or progesterone analogue it contains
- Travel for more than 4 hours
- Recent childbirth.
However, the trials that tested different lengths of anticoagulation have varied markedly in how they defined provoked deep vein thrombosis.4–7
Recommendation: Warfarin or equivalent for 3 months
The American College of Chest Physicians (ACCP) recommends 3 months of anticoagulation with warfarin or another vitamin K antagonist for patients with VTE secondary to a transient (reversible) risk factor,2 and we agree.
HOW LONG TO TREAT CANCER-RELATED VTE
Patients with cancer are at higher risk of developing VTE. Furthermore, in one study,9 compared with other patients with VTE, patients with cancer were three times more likely to have another episode of VTE, with a cumulative rate of recurrence at 1 year of 21% vs 7%. Cancer patients were also twice as likely to suffer major bleeding complications while on anticoagulation.9
Warfarin is a difficult drug to manage because it has many interactions with foods, diseases, and other drugs. These difficulties are amplified in many cancer patients during chemotherapy.
Warfarin was compared with a LMWH in four randomized trials in cancer patients, and a meta-analysis10 found a 50% relative reduction in the rates of recurrent deep vein thrombosis and pulmonary embolism with LMWH treatment. These results were driven primarily by the CLOT trial (Comparison of Low-Molecular-Weight Heparin Versus Oral Anticoagulant Therapy for the Prevention of Recurrent Venous Thromboembolism in Patients With Cancer),11 which showed an 8% absolute risk reduction (number needed to treat 13) without an increase in major bleeding when cancer-related VTE was treated with an LMWH—ie, dalteparin (Fragmin)—for 6 months compared with warfarin.
Current thinking suggests that VTE should be treated until the cancer is resolved. However, this hypothesis has not been adequately tested, and consequently, the ACCP gives it only a level 1C recommendation.2 The largest of the four trials comparing warfarin and an LMWH lasted only 6 months. The safety of extending LMWH treatment beyond 6 months is currently unknown but is under investigation (clinicaltrials.gov identifier NCT00942968).
Recommendation: LMWH therapy for at least 6 months
The ACCP guidelines recommend LMWH therapy for 3 to 6 months, followed by warfarin or another vitamin K antagonist or continued LMWH treatment until the cancer is resolved.2
The National Comprehensive Cancer Network guidelines recommend an LMWH for 6 months as monotherapy and indefinite anticoagulation if the cancer is still active.12
The American Society of Clinical Oncology guidelines recommend an LMWH for at least 6 months and indefinite anticoagulant therapy for selected patients with active cancer.13
We agree that patients with active cancer should receive an LMWH for at least 6 months and indefinite anticoagulation until the cancer is resolved.
In our experience, many patients are reluctant to give themselves the daily injections that LMWH therapy requires, and so they need to be well-informed about the marked decrease in VTE recurrence with this less-convenient and more-expensive therapy. Many patients face insurance barriers to cover the cost of LMWH therapy; however, careful attention to preauthorization can usually overcome this obstacle.
HOW LONG TO TREAT RECURRENT VTE
It makes clinical sense that patients who have a second VTE event should be treated indefinitely. This theory was tested in a randomized clinical trial14 in which patients with provoked or unprovoked VTE were randomized after their second event to receive anticoagulation for 6 months vs indefinitely.
After 4 years of follow-up, the recurrence rate was 21% in patients assigned to 6 months of treatment and only 3% in patients who continued anticoagulation throughout the trial. On the other hand, major hemorrhage occurred in 3% of patients treated for 6 months and in 9% in patients who continued anticoagulation indefinitely.
Of note, most of the patients in this trial had unprovoked (idiopathic) VTE, so the results should not be extrapolated to patients with provoked VTE, who accounted for only 20% of the study population.14
Recommendation: Long-term anticoagulation
We agree with the ACCP recommendation2 that patients who have had a second episode of unprovoked VTE should receive long-term anticoagulation. Because of a lack of data, the duration of therapy for patients with a second episode of provoked VTE should be individualized.
HOW LONG TO TREAT THROMBOPHILIA-RELATED VTE
Inherited thrombophilias
Patients with VTE that is not related to a clear provoking risk factor or cancer frequently have testing to evaluate for a hypercoagulable state. This workup traditionally includes the most common inherited thrombophilias for gene mutations for factor V and prothrombin as well as for deficiencies in protein C, protein S, antithrombin and the acquired antiphospholipid syndrome.
The key questions that should be asked prior to embarking on this workup are:
- Will the results change the length of therapy for the patient?
- Will testing the patient help with genetic counseling and possible testing of family members?
- Will the results change the targeted INR range for warfarin or other vitamin K antagonist therapy?
Patients with inherited thrombophilia have a greater risk of developing an initial VTE event; however, these tests do not help predict the recurrence of VTE in patients with established disease more than clinical risk factors do. A prospective study demonstrated this by looking at the effect of thrombophilia and clinical factors on the recurrence of venous thrombosis and found that inherited prothrombotic abnormalities do not appear to play an important role in the risk of a recurrent event.15 On the other hand, clinical factors, such as whether the first event was idiopathic or provoked, appear more important in determining the duration of anticoagulation therapy.15 A systematic review of the common inherited thrombophilias showed the VTE recurrence rate of patients with factor V Leiden was higher than in patients without the mutation; however, the absolute rates of recurrence were not much different than what would be expected in patients with idiopathic VTE.16
A retrospective study involving a large cohort of families of patients who already had experienced a first episode of either idiopathic or provoked VTE showed high annual risks of recurrent VTE associated with hereditary deficiencies of protein S (8.4%), protein C (6.0%), and antithrombin (10%).17 However, for the more commonly occurring genetic thrombophilias, the factor V Leiden and prothrombin G20210A mutations, family members with either gene abnormality had low rates of VTE, suggesting that testing of relatives of probands is not clinically useful.16
Antiphospholipid syndrome
Antiphospholipid syndrome is an acquired thrombophilia. A patient has thrombotic antiphospholipid syndrome when there is a history of vascular thrombosis in the presence of persistently positive tests (at least 12 weeks apart) for lupus anticoagulants, anticardiolipin antibodies, or anti-beta-2 glycoprotein I. A prospective study of 412 patients with a first episode of VTE found that 15% were positive for anticardiolipin antibody at the end of 6 months of anticoagulation. The risk of recurrent VTE after 4 years was 29% in patients with antibodies and 14% in those without antibodies (relative risk 2.1; 95% confidence interval [CI] 1.3–3.3; P =.0013).18
Recent reviews advise indefinite warfarin anticoagulation in patients with VTE and persistence of antiphospholipid antibodies.19 However, the optimal duration of anticoagulation is uncertain. Until well-designed clinical trials are done, the current general consensus is to anticoagulate these patients indefinitely.20,21 Retrospective studies had suggested that patients with antiphospholipid antibodies required a higher therapeutic INR range; however, this observation was tested in two trials that found no difference in thromboembolic rates when patients were randomized to an INR of 2.0–3.0 vs 3.1–4.0,22 or 2.0–3.0 vs 3.0–4.5.23
No formal recommendations
In the absence of strong evidence, the ACCP guidelines do not include a recommendation on the duration of anticoagulation treatment specific to inherited thrombophilias. We believe that clinical factors are more important than inherited thrombophilias for deciding the duration of anticoagulation, and that testing is almost never indicated or useful. However, patients with antiphospholipid syndrome are at high risk of recurrence, and it is our practice to anticoagulate these patients indefinitely.
HOW LONG TO TREAT UNPROVOKED (IDIOPATHIC) VTE
A VTE event is thought to be idiopathic if it occurs without a clearly identified provoking factor.
In an observational study,3 patients with oral contraceptive use, transient illness, immobilization, or a history of travel had an 8.8% risk of recurrence vs 19.4% in patients with unprovoked VTE. The meta-analysis discussed above (Table 1)8 also shows that patients with these nonsurgical risk factors have a lower rate of recurrence than patients with idiopathic VTE.
The high rate of recurrence of idiopathic VTE (4% to 27% after 3 months of anticoagulation24–26) suggests that a longer duration of treatment is reasonable. However, increasing the length of therapy from 3 to 12 months delays but does not prevent recurrence, the risk of which begins to accumulate once anticoagulation is stopped.24,25
Three promising strategies to identify subgroups of patients with idiopathic VTE who are at highest risk of recurrence and who would benefit the most from prolonged anticoagulation are d-dimer testing, evaluation for residual vein thrombosis in patients who present with a deep vein thrombosis, and clinical prediction rules.
d-dimer testing
d-dimer is a degradation product of fibrin and is an indirect marker of residual thrombosis.30
In a systematic review of patients with a first episode of unprovoked VTE,31 a normal d-dimer concentration at the end of at least 3 months of anticoagulation was associated with a 3.5% annual risk of recurrence, whereas an elevated d-dimer level at that time was associated with an annual risk of 8.9%. These results were confirmed in a systematic review of individual patient data.32
In a randomized trial,28 patients with an idiopathic VTE event who received anticoagulation for at least 3 months had their d-dimer level measured 1 month after cessation of treatment. Those with an elevated level were randomized to either resume anticoagulation or not. Patients who resumed anticoagulation had an annual recurrence rate of 2%; however, those who were allocated not to restart anticoagulation had a recurrence rate of 10.9% per year. There was no difference in the rate of major bleeding between the two groups. Patients in this clinical trial who had a normal d-dimer level did not restart anticoagulation and had an annual recurrence rate of 4.4%.
Evaluation for residual thrombosis
A randomized trial34 in patients with both provoked and idiopathic deep vein thrombosis showed a reduction in recurrence when those who had residual vein thrombosis were given extended anticoagulation. In the subset of patients whose deep vein thrombosis was idiopathic, the recurrence rate was 17% per year when treatment lasted only 3 months and 10% when it was extended for up to 1 year.
Another trial35 randomized patients with provoked and idiopathic deep vein thrombosis to receive anticoagulation for the usual duration or to continue treatment until recanalization of the residual thrombus was demonstrated on follow-up Doppler ultrasonography. Patients who received this ultrasonography-tailored treatment had a lower rate of recurrence of VTE; however, the absolute reductions in recurrence rates cannot be calculated from this report for patients with idiopathic deep vein thrombosis.
A prospective observational study36 of the predictive value of d-dimer status and residual vein thrombus found that only d-dimer was an independent risk factor for recurrent VTE after vitamin K antagonist withdrawal.
A clinical prediction rule: ‘Men and HERDOO2’
A promising tool for predicting if a patient is at low risk of recurrent VTE after the first episode of proximal deep vein thrombosis or pulmonary embolism is known by the mnemonic device “Men and HERDOO2.” It is based on data prospectively derived by Rodger et al37 to identify patients with less than a 3% annual risk of recurrent VTE after their first event of idiopathic proximal deep vein thrombosis or pulmonary embolism. Risk factors for recurrent VTE were male sex (the “men” of “Men and HERDOO2”), signs of postthrombotic syndrome, including hyperpigmentation of the lower extremities, edema or redness of either leg, a d-dimer level > 250 μg/L, obesity (body mass index > 30 kg/m2, and older age (> 65 years).
Overall, one-fourth of the population were women with no risk factors or one risk factor, and their risk of recurrence was 1.6% per year. Men and women who had two or more risk factors for postthrombotic syndrome (hyperpigmentation, edema, or redness), elevated d-dimer, obesity, or older age were predicted to be at higher risk of recurrent VTE. Patients such as this should be considered for indefinite anticoagulation.
Ideally, clinical prediction rules should be validated in a separate group of patients before they are used routinely in practice,38 and this clinical prediction rule is currently being tested in the REVERSE II study. If the results are consistent, this will be an easy-to-use tool to help identify patients who likely can safely stop anticoagulation therapy after 3 to 6 months (clinicaltrials.gov Identifier: NCT00967304).
The location of the thrombosis also influences the likelihood of recurrence. Patients with isolated distal (calf) deep vein thrombosis are less likely to suffer recurrent VTE than those who present with proximal deep vein thrombosis. However, trials focusing specifically on the precise subset of idiopathic isolated distal deep vein thrombosis are lacking. In a randomized trial39 comparing 6 vs 12 weeks of anticoagulation for isolated distal deep vein thrombosis and 12 vs 24 weeks for proximal deep vein thrombosis, the annual rates of recurrence after 12 weeks of treatment were approximately 3.4% for isolated distal and 8.1% for proximal deep vein thrombosis.39
Recommendation: At least 3 months of warfarin or equivalent
We agree with the ACCP recommendation2 that patients with unprovoked VTE should receive at least 3 months of anticoagulation with a vitamin K antagonist.
If the patient has no risk factors for bleeding and good anticoagulant monitoring is achievable, we agree with long-term anticoagulation for proximal unprovoked deep vein thrombosis or pulmonary embolism, and 3 months of therapy for isolated distal unprovoked deep vein thrombosis.
Patient preferences and the risk of recurrence vs the risk of bleeding should be discussed with patients when contemplating indefinite anticoagulation.
If testing is being considered to assist in the decision to prescribe indefinite anticoagulation, we prefer using d-dimer levels rather than ultrasonography to detect residual venous thrombosis because of its ease of use and the strength of the current evidence.
PREVENTING POSTTHROMBOTIC SYNDROME
The postthrombotic (postphlebitic) syndrome is a chronic and burdensome consequence of deep vein thrombosis that occurs despite anticoagulation therapy. It is estimated to affect 23% to 60% of patients and typically manifests in the first 2 years.40 It is not only costly in clinical terms, with decreased quality of life for the patient, but health care expenditures have been estimated to range from $400 per year in a Brazilian study to $7,000 per year in a US study.40
Typical symptoms include leg pain, heaviness, swelling, and cramping. In severe cases, chronic venous ulcers can occur and are difficult to treat.41
The definition of postthrombotic syndrome has been unclear over the years, and six different scales that measure signs and symptoms have been reported.42
The Villalta scale has been proposed by the International Society of Thrombosis and Hemostasis as a diagnostic standard to define postthrombotic syndrome.42 This validated scale is based on five clinical symptoms, six clinical signs, and the presence or absence of venous ulcers. Each clinical symptom and sign is scored as mild (1 point), moderate (2 points), or severe (3 points). Symptoms include pain, cramps, heaviness, paresthesia, and pruritus; the six clinical signs are pretibial edema, skin induration, hyperpigmentation, redness, venous ectasia, and pain on calf compression.
According to the International Society of Thrombosis and Hemostasis, postthrombotic syndrome is present if the Villalta score is 5 or greater or if a venous ulcer is present in a leg with previous deep vein thrombosis. Further, using the Villalta scale, postthrombotic syndrome can be categorized as mild (score 5–9), moderate (10–14), or severe (≥ 15).
A limitation of the Villalta scale is that the presence or absence of a venous ulcer has not been assigned a score. Since a venous ulcer requires more aggressive measures, the society defines postthrombotic syndrome as severe if venous ulcers are present.42
Acute symptoms of deep vein thrombosis may take months to resolve and, indeed, acute symptoms may transition to chronic symptoms without a symptom-free interval. It is recommended that postthrombotic syndrome not be diagnosed before 3 months to avoid inappropriately attributing acute symptoms and signs of deep vein thrombosis to the postthrombotic syndrome.42
Studies of stockings
A systematic review of three randomized trials44 concluded that elastic compression stockings reduce the risk of postthrombotic syndrome (any severity) from 43% to 20% and severe postthrombotic syndrome from 15% to 7%.43
The first of these trials44 randomized patients soon after the diagnosis of deep vein thrombosis to receive made-to-order compression stockings that were rated at 30 to 40 mm Hg or to be in a control group that did not receive stockings. The second trial45 randomized patients 1 year after the index event of deep vein thrombosis to receive 20- to 30-mm Hg stockings or stockings that were two sizes too large (the control group). The third study46 randomly allocated patients to receive “off-the-shelf” stockings (30–40 mm Hg) or no stockings. Each study used its own definition of postthrombotic syndrome.
Although these studies strongly suggest compression stockings prevent postthrombotic syndrome, several methodologic issues remain:
- A standard definition of postthrombotic syndrome was not used
- The amount of compression varied between studies
- The studies were not blinded.
Lack of blinding becomes most significant when an outcome is based on subjective findings, like the symptoms that make up a large part of the diagnosis of postthrombotic syndrome.
The SOX trial, currently under way, is designed to address these methodologic issues and should be completed in 2012 (clinicaltrials.gov Identifier: NCT00143598).
Recommendation: Stockings for at least 2 years
We agree with the ACCP recommendation that a patient who has had a symptomatic proximal deep vein thrombosis should wear an elastic compression stocking with an ankle pressure gradient of 30 to 40 mm Hg as soon as possible after starting anticoagulant therapy and continuing for a minimum of 2 years.2
SCREENING FOR OCCULT MALIGNANCY
VTE can be the first manifestation of cancer.
French physician Armand Trousseau, in the 1860s, was the first to describe disseminated intravascular coagulation closely associated with adenocarcinoma. Ironically, several years later, after suffering for weeks from abdominal pain, he declared to one of his students that he had developed thrombosis, and he died of gastric cancer shortly thereafter.47
Since cancer is a well-known risk factor for VTE, it is logical to screen for cancer as an explanation for an idiopathic VTE event.48 To make an informed decision, one needs to understand the rate of occult cancer at the time VTE is diagnosed, the risk of future development of cancer, and the utility of extensive cancer screening.
The clinical efficacy, side effects, and cost-effectiveness of cancer screening in patients with idiopathic VTE are unknown. However, a systematic review47 of 34 studies found that, in patients with idiopathic VTE, cancer was diagnosed within 1 month in 6.1%, within 6 months in 8.6%, and within 1 year in 10.0% (95% CI 8.6–11.3).
A subset of studies compared two strategies for screening soon after the diagnosis of idiopathic VTE: a strategy limited to the history, physical examination, basic blood work, and chest radiography vs an extensive screening strategy that also included serum tumor markers or abdominal ultrasonography or computed tomography. Limited screening detected 49% of the prevalent cancers; extensive screening increased this rate to 70%. Stated another way, the detection rate for prevalent cancers was 5% with limited screening and 7% with extensive screening soon after the diagnosis of idiopathic VTE.47
Patients with idiopathic VTE had higher rates of cancer within 1 month of diagnosis than patients with provoked VTE (6.1% vs 1.9%), and this difference persisted at 1 year (10.0% vs 2.6%).47
Recommendation: Individualized cancer screening
Patients with idiopathic VTE have a significant risk of occult cancer within the first year after diagnosis, and cancer screening should be considered. Our practice for patients with idiopathic VTE is to perform a history and physical examination and ensure that the patient is up to date on age- and sex-specific cancer screening.
The use of additional imaging or biomarkers should be discussed with patients so they can balance the risks (radiation and potential false-positive results with their downstream consequences), costs, and potential benefits, given the lack of proven survival benefit or cost-effectiveness.
ORAL ANTICOAGULANT MANAGEMENT
Warfarin’s multiple interactions, along with the need for INR monitoring, make it a difficult medication to manage.
The Joint Commission, the US organization for health service accreditation and certification, has defined National Patient Safety Goals and quality measures for the management of anticoagulation.49 Organized anticoagulation management services, dosing algorithms, and patient self-testing using capillary INR meters or patient self-management of warfarin were recommended as tools to improve the time patients spend in the therapeutic INR range.50
Two new oral anticoagulants
The limitations of warfarin have stimulated the search for newer oral anticoagulants that do not require laboratory monitoring or have as many diet and drug interactions.
Two trials have been published with experimental oral anticoagulants that had similar efficacy and safety as warfarin in the treatment of VTE.
The study of dabigatran (Pradaxa) vs warfarin in the treatment of acute VTE (the RECOVER trial)51 randomized 2,539 patients with acute VTE to receive the oral direct thrombin inhibitor dabigatran or warfarin for approximately 6 months. Of note, each treatment group received a median of 6 days of heparin, LMWH, or fondaparinux at the beginning of blinded therapy. The rates of recurrent VTE and major bleeding were similar between the treatment arms, and overall bleeding was less with dabigatran. Dabigatran was approved in the United States in October 2010 for stroke prevention in atrial fibrillation but has yet to be approved for the treatment of VTE pending further study (clinicaltrials.gov Identifier: NCT00680186).
A study of oral rivaroxaban (Xarelto) for symptomatic VTE (the EINSTEIN-DVT trial) 52 randomized 3,449 patients with acute deep vein thrombosis to rivaroxaban or enoxaparin (Lovenox) overlapped with warfarin or another vitamin K antagonist in the usual manner. No difference was noted between the treatments in the rate of recurrence of VTE or of major bleeding. Of note, patients randomized to rivaroxaban received 15 mg twice a day for the first 3 weeks of treatment and then 20 mg per day for the remainder of their therapy and did not require parenteral anticoagulant overlap.
The long-awaited promise of easier-to-use oral anticoagulants for the treatment of VTE is drawing near and has the potential to revolutionize the treatment of this common disorder. In the meantime, close monitoring of warfarin and careful patient education regarding its use are essential. And even with the development of new drugs in the future, it is still imperative that patients with acute VTE receive the correct length of anticoagulation treatment, are prescribed stockings to prevent postthrombotic syndrome, and are updated on routine cancer screening.
- Spencer FA, Emery C, Lessard D, et al. The Worcester Venous Thromboembolism study: a population-based study of the clinical epidemiology of venous thromboembolism. J Gen Intern Med 2006; 21:722–727.
- Kearon C, Kahn SR, Agnelli G, Goldhaber S, Raskob GE, Comerota AJ; American College of Chest Physicians. Antithrombotic therapy for venous thromboembolic disease: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest 2008; 133(suppl 6):454S–545S.
- Baglin T, Luddington R, Brown K, Baglin C. Incidence of recurrent venous thromboembolism in relation to clinical and thrombophilic risk factors: prospective cohort study. Lancet 2003; 362:523–526.
- Schulman S, Lockner D, Juhlin-Dannfelt A. The duration of oral anticoagulation after deep vein thrombosis. A randomized study. Acta Med Scand 1985; 217:547–552.
- Optimum duration of anticoagulation for deep-vein thrombosis and pulmonary embolism. Research Committee of the British Thoracic Society. Lancet 1992; 340:873–876.
- Schulman S, Rhedin AS, Lindmarker P, et al. A comparison of six weeks with six months of oral anticoagulant therapy after a first episode of venous thromboembolism. Duration of Anticoagulation Trial Study Group. N Engl J Med 1995; 332:1661–1665.
- Kearon C, Ginsberg JS, Anderson DR, et al. Comparison of 1 month with 3 months of anticoagulation for a first episode of venous thromboembolism associated with a transient risk factor. J Thromb Haemost 2004; 2:743–749.
- Iorio A, Kearon C, Filippucci E, et al. Risk of recurrence after a first episode of symptomatic venous thromboembolism provoked by a transient risk factor: a systematic review. Arch Intern Med 2010; 170:1710–1716.
- Prandoni P, Lensing AW, Piccioli A, et al. Recurrent venous thromboembolism and bleeding complications during anticoagulant treatment in patients with cancer and venous thrombosis. Blood 2002; 100:3484–3488.
- Hull RD, Pineo GF, Brant RF, et al; LITE Trial Investigators. Long-term low-molecular-weight heparin versus usual care in proximal-vein thrombosis patients with cancer. Am J Med 2006; 119:1062–1072.
- Lee AY, Levine MN, Baker RI, et al; Randomized Comparison of Low-Molecular-Weight Heparin versus Oral Anticoagulant Therapy for the Prevention of Recurrent Venous Thromboembolism in Patients with Cancer (CLOT) Investigators. Low-molecular-weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N Engl J Med 2003; 349:146–153.
- National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology, Venous Thromboembolic Disease. http://www.nccn.org/professionals/physician_gls/pdf/vte.pdf. Accessed August 3, 2011.
- Lyman GH, Khorana AA, Falanga A, et al; American Society of Clinical Oncology. American Society of Clinical Oncology guideline: recommendations for venous thromboembolism prophylaxis and treatment in patients with cancer. J Clin Oncol 2007; 25:5490–5505.
- Schulman S, Granqvist S, Holmström M, et al. The duration of oral anticoagulant therapy after a second episode of venous thromboembolism. The Duration of Anticoagulation Trial Study Group. N Engl J Med 1997; 336:393–398.
- Christiansen SC, Cannegieter SC, Koster T, Vandenbroucke JP, Rosendaal FR. Thrombophilia, clinical factors, and recurrent venous thrombotic events. JAMA 2005; 293:2352–2361.
- Segal JB, Brotman DJ, Necochea AJ, et al. Predictive value of factor V Leiden and prothrombin G20210A in adults with venous thromboembolism and in family members of those with a mutation: a systematic review. JAMA 2009; 301:2472–2485.
- Brouwer JL, Lijfering WM, Ten Kate MK, Kluin-Nelemans HC, Veeger NJ, van der Meer J. High long-term absolute risk of recurrent venous thromboembolism in patients with hereditary deficiencies of protein S, protein C or antithrombin. Thromb Haemost 2009; 101:93–99.
- Schulman S, Svenungsson E, Granqvist S. Anticardiolipin antibodies predict early recurrence of thromboembolism and death among patients with venous thromboembolism following anticoagulant therapy. Duration of Anticoagulation Study Group. Am J Med 1998; 104:332–338.
- Derksen RH, de Groot PG. Towards evidence-based treatment of thrombotic antiphospholipid syndrome. Lupus 2010; 19:470–474.
- Lim W, Crowther MA, Eikelboom JW. Management of antiphospholipid antibody syndrome: a systematic review. JAMA 2006; 295:1050–1057.
- Fonseca AG, D’Cruz DP. Controversies in the antiphospholipid syndrome: can we ever stop warfarin? J Autoimmune Dis 2008; 5:6.
- Crowther MA, Ginsberg JS, Julian J, et al. A comparison of two intensities of warfarin for the prevention of recurrent thrombosis in patients with the antiphospholipid antibody syndrome. N Engl J Med 2003; 349:1133–1138.
- Finazzi G, Marchioli R, Brancaccio V, et al. A randomized clinical trial of high-intensity warfarin vs. conventional antithrombotic therapy for the prevention of recurrent thrombosis in patients with the antiphospholipid syndrome (WAPS). J Thromb Haemost 2005; 3:848–853.
- Agnelli G, Prandoni P, Becattini C, et al; Warfarin Optimal Duration Italian Trial Investigators. Extended oral anticoagulant therapy after a first episode of pulmonary embolism. Ann Intern Med 2003; 139:19–25.
- Agnelli G, Prandoni P, Santamaria MG, et al; Warfarin Optimal Duration Italian Trial Investigators. Three months versus one year of oral anticoagulant therapy for idiopathic deep venous thrombosis. Warfarin Optimal Duration Italian Trial Investigators. N Engl J Med 2001; 345:165–169.
- Kearon C, Gent M, Hirsh J, et al. A comparison of three months of anticoagulation with extended anticoagulation for a first episode of idiopathic venous thromboembolism. N Engl J Med 1999; 340:901–907.
- Kearon C, Ginsberg JS, Kovacs MJ, et al; Extended Low-Intensity Anticoagulation for Thrombo-Embolism Investigators. Comparison of low-intensity warfarin therapy with conventional-intensity warfarin therapy for long-term prevention of recurrent venous thromboembolism. N Engl J Med 2003; 349:631–639.
- Palareti G, Cosmi B, Legnani C, et al; PROLONG Investigators. D-dimer testing to determine the duration of anticoagulation therapy. N Engl J Med 2006; 355:1780–1789.
- Ridker PM, Goldhaber SZ, Glynn RJ. Low-intensity versus conventional-intensity warfarin for prevention of recurrent venous thromboembolism. N Engl J Med 2003; 349:2164–2167.
- Bockenstedt P. D-dimer in venous thromboembolism. N Engl J Med 2003; 349:1203–1204.
- Verhovsek M, Douketis JD, Yi Q, et al. Systematic review: D-dimer to predict recurrent disease after stopping anticoagulant therapy for unprovoked venous thromboembolism. Ann Intern Med 2008; 149:481–490,W94.
- Douketis J, Tosetto A, Marcucci M, et al. Patient-level metaanalysis: effect of measurement timing, threshold, and patient age on ability of D-dimer testing to assess recurrence risk after unprovoked venous thromboembolism. Ann Intern Med 2010; 153:523–531.
- Prandoni P, Lensing AW, Prins MH, et al. Residual venous thrombosis as a predictive factor of recurrent venous thromboembolism. Ann Intern Med 2002; 137:955–960.
- Siragusa S, Malato A, Anastasio R, et al. Residual vein thrombosis to establish duration of anticoagulation after a first episode of deep vein thrombosis: the Duration of Anticoagulation based on Compression UltraSonography (DACUS) study. Blood 2008; 112:511–515.
- Prandoni P, Prins MH, Lensing AW, et al; AESOPUS Investigators. Residual thrombosis on ultrasonography to guide the duration of anticoagulation in patients with deep venous thrombosis: a randomized trial. Ann Intern Med 2009; 150:577–585.
- Cosmi B, Legnani C, Cini M, Guazzaloca G, Palareti G. D-dimer levels in combination with residual venous obstruction and the risk of recurrence after anticoagulation withdrawal for a first idiopathic deep vein thrombosis. Thromb Haemost 2005; 94:969–974.
- Rodger MA, Kahn SR, Wells PS, et al. Identifying unprovoked thromboembolism patients at low risk for recurrence who can discontinue anticoagulant therapy. CMAJ 2008; 179:417–426.
- McGinn TG, Guyatt GH, Wyer PC, Naylor CD, Stiell IG, Richardson WS. Users’ guides to the medical literature: XXII: how to use articles about clinical decision rules. Evidence-Based Medicine Working Group. JAMA 2000; 284:79–84.
- Pinede L, Ninet J, Duhaut P, et al; Investigators of the “Durée Optimale du Traitement AntiVitamines K” (DOTAVK) Study. Comparison of 3 and 6 months of oral anticoagulant therapy after a first episode of proximal deep vein thrombosis or pulmonary embolism and comparison of 6 and 12 weeks of therapy after isolated calf deep vein thrombosis. Circulation 2001; 103:2453–2460.
- Ashrani AA, Heit JA. Incidence and cost burden of postthrombotic syndrome. J Thromb Thrombolysis 2009; 28:465–476.
- Kahn SR, Shrier I, Julian JA, et al. Determinants and time course of the postthrombotic syndrome after acute deep venous thrombosis. Ann Intern Med 2008; 149:698–707.
- Kahn SR, Partsch H, Vedantham S, Prandoni P, Kearon C; Subcommittee on Control of Anticoagulation of the Scientific and Standardization Committee of the International Society on Thrombosis and Haemostasis. Definition of post-thrombotic syndrome of the leg for use in clinical investigations: a recommendation for standardization. J Thromb Haemost 2009; 7:879–883.
- Kolbach DN, Sandbrink MW, Hamulyak K, Neumann HA, Prins MH. Non-pharmaceutical measures for prevention of post-thrombotic syndrome. Cochrane Database Syst Rev 2004;CD004174.
- Brandjes DP, Büller HR, Heijboer H, et al. Randomised trial of effect of compression stockings in patients with symptomatic proximal-vein thrombosis. Lancet 1997; 349:759–762.
- Ginsberg JS, Hirsh J, Julian J, et al. Prevention and treatment of postphlebitic syndrome: results of a 3-part study. Arch Intern Med 2001; 161:2105–2109.
- Prandoni P, Lensing AW, Prins MH, et al. Below-knee elastic compression stockings to prevent the post-thrombotic syndrome: a randomized, controlled trial. Ann Intern Med 2004; 141:249–256.
- Carrier M, Le Gal G, Wells PS, Fergusson D, Ramsay T, Rodger MA. Systematic review: the Trousseau syndrome revisited: should we screen extensively for cancer in patients with venous thromboembolism? Ann Intern Med 2008; 149:323–333.
- Blom JW, Doggen CJ, Osanto S, Rosendaal FR. Malignancies, prothrombotic mutations, and the risk of venous thrombosis. JAMA 2005; 293:715–722.
- Kaatz S. Impact on patient care: patient case through the continuum of care. J Thromb Thrombolysis 2010; 29:167–170.
- Ansell J, Hirsh J, Hylek E, Jacobson A, Crowther M, Palareti G; American College of Chest Physicians. Pharmacology and management of the vitamin K antagonists: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest 2008; 133(suppl 6):160S–198S.
- Schulman S, Kearon C, Kakkar AK, et al; for the RE-COVER Study Group. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med 2009; 361:2342–2452.
- The EINSTEIN Investigators. Oral rivaroxaban for symptomatic venous thromboembolism. N Engl J Med 2010; 363;2499–2510.
- Spencer FA, Emery C, Lessard D, et al. The Worcester Venous Thromboembolism study: a population-based study of the clinical epidemiology of venous thromboembolism. J Gen Intern Med 2006; 21:722–727.
- Kearon C, Kahn SR, Agnelli G, Goldhaber S, Raskob GE, Comerota AJ; American College of Chest Physicians. Antithrombotic therapy for venous thromboembolic disease: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest 2008; 133(suppl 6):454S–545S.
- Baglin T, Luddington R, Brown K, Baglin C. Incidence of recurrent venous thromboembolism in relation to clinical and thrombophilic risk factors: prospective cohort study. Lancet 2003; 362:523–526.
- Schulman S, Lockner D, Juhlin-Dannfelt A. The duration of oral anticoagulation after deep vein thrombosis. A randomized study. Acta Med Scand 1985; 217:547–552.
- Optimum duration of anticoagulation for deep-vein thrombosis and pulmonary embolism. Research Committee of the British Thoracic Society. Lancet 1992; 340:873–876.
- Schulman S, Rhedin AS, Lindmarker P, et al. A comparison of six weeks with six months of oral anticoagulant therapy after a first episode of venous thromboembolism. Duration of Anticoagulation Trial Study Group. N Engl J Med 1995; 332:1661–1665.
- Kearon C, Ginsberg JS, Anderson DR, et al. Comparison of 1 month with 3 months of anticoagulation for a first episode of venous thromboembolism associated with a transient risk factor. J Thromb Haemost 2004; 2:743–749.
- Iorio A, Kearon C, Filippucci E, et al. Risk of recurrence after a first episode of symptomatic venous thromboembolism provoked by a transient risk factor: a systematic review. Arch Intern Med 2010; 170:1710–1716.
- Prandoni P, Lensing AW, Piccioli A, et al. Recurrent venous thromboembolism and bleeding complications during anticoagulant treatment in patients with cancer and venous thrombosis. Blood 2002; 100:3484–3488.
- Hull RD, Pineo GF, Brant RF, et al; LITE Trial Investigators. Long-term low-molecular-weight heparin versus usual care in proximal-vein thrombosis patients with cancer. Am J Med 2006; 119:1062–1072.
- Lee AY, Levine MN, Baker RI, et al; Randomized Comparison of Low-Molecular-Weight Heparin versus Oral Anticoagulant Therapy for the Prevention of Recurrent Venous Thromboembolism in Patients with Cancer (CLOT) Investigators. Low-molecular-weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N Engl J Med 2003; 349:146–153.
- National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology, Venous Thromboembolic Disease. http://www.nccn.org/professionals/physician_gls/pdf/vte.pdf. Accessed August 3, 2011.
- Lyman GH, Khorana AA, Falanga A, et al; American Society of Clinical Oncology. American Society of Clinical Oncology guideline: recommendations for venous thromboembolism prophylaxis and treatment in patients with cancer. J Clin Oncol 2007; 25:5490–5505.
- Schulman S, Granqvist S, Holmström M, et al. The duration of oral anticoagulant therapy after a second episode of venous thromboembolism. The Duration of Anticoagulation Trial Study Group. N Engl J Med 1997; 336:393–398.
- Christiansen SC, Cannegieter SC, Koster T, Vandenbroucke JP, Rosendaal FR. Thrombophilia, clinical factors, and recurrent venous thrombotic events. JAMA 2005; 293:2352–2361.
- Segal JB, Brotman DJ, Necochea AJ, et al. Predictive value of factor V Leiden and prothrombin G20210A in adults with venous thromboembolism and in family members of those with a mutation: a systematic review. JAMA 2009; 301:2472–2485.
- Brouwer JL, Lijfering WM, Ten Kate MK, Kluin-Nelemans HC, Veeger NJ, van der Meer J. High long-term absolute risk of recurrent venous thromboembolism in patients with hereditary deficiencies of protein S, protein C or antithrombin. Thromb Haemost 2009; 101:93–99.
- Schulman S, Svenungsson E, Granqvist S. Anticardiolipin antibodies predict early recurrence of thromboembolism and death among patients with venous thromboembolism following anticoagulant therapy. Duration of Anticoagulation Study Group. Am J Med 1998; 104:332–338.
- Derksen RH, de Groot PG. Towards evidence-based treatment of thrombotic antiphospholipid syndrome. Lupus 2010; 19:470–474.
- Lim W, Crowther MA, Eikelboom JW. Management of antiphospholipid antibody syndrome: a systematic review. JAMA 2006; 295:1050–1057.
- Fonseca AG, D’Cruz DP. Controversies in the antiphospholipid syndrome: can we ever stop warfarin? J Autoimmune Dis 2008; 5:6.
- Crowther MA, Ginsberg JS, Julian J, et al. A comparison of two intensities of warfarin for the prevention of recurrent thrombosis in patients with the antiphospholipid antibody syndrome. N Engl J Med 2003; 349:1133–1138.
- Finazzi G, Marchioli R, Brancaccio V, et al. A randomized clinical trial of high-intensity warfarin vs. conventional antithrombotic therapy for the prevention of recurrent thrombosis in patients with the antiphospholipid syndrome (WAPS). J Thromb Haemost 2005; 3:848–853.
- Agnelli G, Prandoni P, Becattini C, et al; Warfarin Optimal Duration Italian Trial Investigators. Extended oral anticoagulant therapy after a first episode of pulmonary embolism. Ann Intern Med 2003; 139:19–25.
- Agnelli G, Prandoni P, Santamaria MG, et al; Warfarin Optimal Duration Italian Trial Investigators. Three months versus one year of oral anticoagulant therapy for idiopathic deep venous thrombosis. Warfarin Optimal Duration Italian Trial Investigators. N Engl J Med 2001; 345:165–169.
- Kearon C, Gent M, Hirsh J, et al. A comparison of three months of anticoagulation with extended anticoagulation for a first episode of idiopathic venous thromboembolism. N Engl J Med 1999; 340:901–907.
- Kearon C, Ginsberg JS, Kovacs MJ, et al; Extended Low-Intensity Anticoagulation for Thrombo-Embolism Investigators. Comparison of low-intensity warfarin therapy with conventional-intensity warfarin therapy for long-term prevention of recurrent venous thromboembolism. N Engl J Med 2003; 349:631–639.
- Palareti G, Cosmi B, Legnani C, et al; PROLONG Investigators. D-dimer testing to determine the duration of anticoagulation therapy. N Engl J Med 2006; 355:1780–1789.
- Ridker PM, Goldhaber SZ, Glynn RJ. Low-intensity versus conventional-intensity warfarin for prevention of recurrent venous thromboembolism. N Engl J Med 2003; 349:2164–2167.
- Bockenstedt P. D-dimer in venous thromboembolism. N Engl J Med 2003; 349:1203–1204.
- Verhovsek M, Douketis JD, Yi Q, et al. Systematic review: D-dimer to predict recurrent disease after stopping anticoagulant therapy for unprovoked venous thromboembolism. Ann Intern Med 2008; 149:481–490,W94.
- Douketis J, Tosetto A, Marcucci M, et al. Patient-level metaanalysis: effect of measurement timing, threshold, and patient age on ability of D-dimer testing to assess recurrence risk after unprovoked venous thromboembolism. Ann Intern Med 2010; 153:523–531.
- Prandoni P, Lensing AW, Prins MH, et al. Residual venous thrombosis as a predictive factor of recurrent venous thromboembolism. Ann Intern Med 2002; 137:955–960.
- Siragusa S, Malato A, Anastasio R, et al. Residual vein thrombosis to establish duration of anticoagulation after a first episode of deep vein thrombosis: the Duration of Anticoagulation based on Compression UltraSonography (DACUS) study. Blood 2008; 112:511–515.
- Prandoni P, Prins MH, Lensing AW, et al; AESOPUS Investigators. Residual thrombosis on ultrasonography to guide the duration of anticoagulation in patients with deep venous thrombosis: a randomized trial. Ann Intern Med 2009; 150:577–585.
- Cosmi B, Legnani C, Cini M, Guazzaloca G, Palareti G. D-dimer levels in combination with residual venous obstruction and the risk of recurrence after anticoagulation withdrawal for a first idiopathic deep vein thrombosis. Thromb Haemost 2005; 94:969–974.
- Rodger MA, Kahn SR, Wells PS, et al. Identifying unprovoked thromboembolism patients at low risk for recurrence who can discontinue anticoagulant therapy. CMAJ 2008; 179:417–426.
- McGinn TG, Guyatt GH, Wyer PC, Naylor CD, Stiell IG, Richardson WS. Users’ guides to the medical literature: XXII: how to use articles about clinical decision rules. Evidence-Based Medicine Working Group. JAMA 2000; 284:79–84.
- Pinede L, Ninet J, Duhaut P, et al; Investigators of the “Durée Optimale du Traitement AntiVitamines K” (DOTAVK) Study. Comparison of 3 and 6 months of oral anticoagulant therapy after a first episode of proximal deep vein thrombosis or pulmonary embolism and comparison of 6 and 12 weeks of therapy after isolated calf deep vein thrombosis. Circulation 2001; 103:2453–2460.
- Ashrani AA, Heit JA. Incidence and cost burden of postthrombotic syndrome. J Thromb Thrombolysis 2009; 28:465–476.
- Kahn SR, Shrier I, Julian JA, et al. Determinants and time course of the postthrombotic syndrome after acute deep venous thrombosis. Ann Intern Med 2008; 149:698–707.
- Kahn SR, Partsch H, Vedantham S, Prandoni P, Kearon C; Subcommittee on Control of Anticoagulation of the Scientific and Standardization Committee of the International Society on Thrombosis and Haemostasis. Definition of post-thrombotic syndrome of the leg for use in clinical investigations: a recommendation for standardization. J Thromb Haemost 2009; 7:879–883.
- Kolbach DN, Sandbrink MW, Hamulyak K, Neumann HA, Prins MH. Non-pharmaceutical measures for prevention of post-thrombotic syndrome. Cochrane Database Syst Rev 2004;CD004174.
- Brandjes DP, Büller HR, Heijboer H, et al. Randomised trial of effect of compression stockings in patients with symptomatic proximal-vein thrombosis. Lancet 1997; 349:759–762.
- Ginsberg JS, Hirsh J, Julian J, et al. Prevention and treatment of postphlebitic syndrome: results of a 3-part study. Arch Intern Med 2001; 161:2105–2109.
- Prandoni P, Lensing AW, Prins MH, et al. Below-knee elastic compression stockings to prevent the post-thrombotic syndrome: a randomized, controlled trial. Ann Intern Med 2004; 141:249–256.
- Carrier M, Le Gal G, Wells PS, Fergusson D, Ramsay T, Rodger MA. Systematic review: the Trousseau syndrome revisited: should we screen extensively for cancer in patients with venous thromboembolism? Ann Intern Med 2008; 149:323–333.
- Blom JW, Doggen CJ, Osanto S, Rosendaal FR. Malignancies, prothrombotic mutations, and the risk of venous thrombosis. JAMA 2005; 293:715–722.
- Kaatz S. Impact on patient care: patient case through the continuum of care. J Thromb Thrombolysis 2010; 29:167–170.
- Ansell J, Hirsh J, Hylek E, Jacobson A, Crowther M, Palareti G; American College of Chest Physicians. Pharmacology and management of the vitamin K antagonists: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest 2008; 133(suppl 6):160S–198S.
- Schulman S, Kearon C, Kakkar AK, et al; for the RE-COVER Study Group. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med 2009; 361:2342–2452.
- The EINSTEIN Investigators. Oral rivaroxaban for symptomatic venous thromboembolism. N Engl J Med 2010; 363;2499–2510.
KEY POINTS
- A low-molecular-weight heparin for at least 6 months is the treatment of choice for cancer-related VTE.
- We recommend 3 months of anticoagulation for VTE caused by a reversible risk factor and indefinite treatment for idiopathic VTE in patients without risk factors for bleeding who can get anticoagulation monitoring.
- Clinical factors are more important in deciding the duration of anticoagulation therapy than evidence of an inherited thrombophilic state.
- Elastic compression stockings reduce the risk of postthrombotic syndrome substantially.
- Patients with idiopathic VTE should have a basic screening for malignancy.
Pharmacist‐Directed Anticoagulation
Anticoagulants are one of the most common drug classes involved in medication errors and adverse events. Warfarin, an anticoagulant that plays a key role in the management of many disease states, is implicated in approximately 30% of reported anticoagulant‐related errors.1 Anticoagulation with warfarin is complicated by inter‐individual variability in response to therapy, clinically significant drug interactions, a narrow therapeutic window, and the need for frequent and lifelong monitoring.2
In the hospital setting, warfarin use is complicated due to patient handoff among health care providers, and acute illnesses that impact sensitivity and response to warfarin. Common causes of errors with anticoagulants are knowledge deficits, failure to follow policy/procedure/protocol, and communication issues.1 An added opportunity for warfarin‐related medication errors is the risk associated with the transition from the inpatient‐to‐outpatient setting. Due to the risk and complexity associated with anticoagulant medications, the Joint Commission instituted National Patient Safety Goal (NPSG) 03.05.01 (formerly NPSG 3E): a series of requirements intended to Reduce the likelihood of patient harm with the use of anticoagulation therapy.3 In order to optimally address this National Patient Safety Goal, a systematic intervention would be required to impact each step of the medication use process for anticoagulants.
Several studies have suggested that dedicated anticoagulation management services or clinics improve anticoagulation management in the outpatient setting.2 Non‐physician providers, primarily pharmacists and nurses, frequently manage outpatient anticoagulation management services or clinics. However, very few studies have evaluated the impact of a warfarin management service in the inpatient hospital setting.48 While the few available studies suggest some benefit associated with an inpatient anticoagulation management service, a minority of these studies have assessed the role of these services in facilitating the transition of the anticoagulated patient to the outpatient setting.7
In order to improve anticoagulation management and safety, our institution implemented an inpatient Pharmacist‐Directed Anticoagulation Service (PDAS). The purpose of this study was to evaluate the impact of this service on both transition of care and safety of patients receiving warfarin anticoagulation.
METHODS
This study was completed at Henry Ford Hospital, an 802‐bed, tertiary care, level 1 trauma and academic medical center in Detroit, MI. The study was carried out between November 2007 and June 2009. The study was approved by the Henry Ford Hospital Institutional Review Board with waiver of consent.
Patients
This was a prospective cluster randomized study. All patients admitted to two internal medicine units (IM1 or IM2) or two cardiology units (Card1 or Card2), who received at least one inpatient dose of warfarin, were eligible for inclusion. Patients were included regardless of whether warfarin was newly initiated during the index admission (newly initiated patients) or was continuation of existing anticoagulation (existing warfarin patients). In order to ensure that patient data following discharge would be available for analysis, patients were excluded from this analysis if they were not scheduled to follow‐up in the Henry Ford Medical Group outpatient anticoagulation clinics after discharge, however, these patients were cared for by the PDAS service in the usual manner.
Study Design
Prior to implementation of the PDAS, one internal medicine and one cardiology unit was randomly selected to receive the PDAS intervention (IM1 and Card1), while the other two units (IM2 and Card2) served as control units. These hospital units were selected because anticoagulants are frequently used on these units and the patient population is generally similar between the two internal medicine and two cardiology unitswith exception that Card1 unit also contains a specialized service for advanced heart failure and left ventricular assist device (LVAD) patients. Of note, there was significant expansion of the heart failure service and LVAD program during the time frame of the study, accounting for a greater number of more complicated patients on the Card1 (PDAS) unit.
Specific responsibilities of the PDAS related to warfarin are detailed in Table 1. The PDAS was implemented in September 2007 as a system‐based change to improve anticoagulant safety at our institution. The goals of this service were to improve communication regarding anticoagulation; to improve safety as patients transition from the inpatient‐to‐outpatient settings; and to standardize anticoagulant dosing, monitoring, and patient education. For patients taking warfarin, who are cared for by a health system‐affiliated physician, the PDAS collaborates with our outpatient anticoagulation clinics in order to facilitate transition from the inpatient‐to‐outpatient setting. The Henry Ford Health System has an established, multisite outpatient Anticoagulation Clinic with >5000 patients actively receiving warfarin dosing and monitoring. The anticoagulation clinics are staffed by nurses and pharmacists who provide standardized management of warfarin for patients of all physicians within our health system and provide consistent high‐quality care (average time in international normalized ratio [INR] goal range = 68.2%). The anticoagulation clinics have been in existence since 1992. The PDAS is comprised of three full‐time and two part‐time pharmacists whose responsibilities are limited to the management of anticoagulation throughout the hospital.
| Inpatient Care | Patient Education | Transition of Care |
|---|---|---|
| ||
| Initial dose selection and daily dose adjustments after warfarin is initiated by primary team | Comprehensive education provided verbally and via written communication utilizing the Krames database. | Contact anticoagulation‐responsible physician and anticoagulation clinic via phone. |
| Provide written dosing regimen to patient and provide date for first INR postdischarge. | ||
| Daily laboratory monitoring | Education provided is standardized between inpatient and outpatient settings. | Create electronic Anticoagulation Discharge Summary. Document communication with the outpatient clinicians, reason for admission, steps taken to manage warfarin drug interactions, and warfarin doses administered during stay, discharge warfarin dose and follow‐up date. |
The PDAS was staffed by repurposing pharmacist staff. All pharmacists had either several years of general medicine‐based clinical practice experience or residency training, or both. Pharmacists were oriented to service responsibilities by spending approximately one week in the outpatient anticoagulation clinic and completing focused review of internal and external anticoagulation guidelines.
In the control group, management of anticoagulation and transition of care occurred at the discretion of the primary care team. The primary team had access to a clinical pharmacist, who was not part of the PDAS, seven days per week. However, the primary team was not able to consult the PDAS.
This study was primarily designed to assess the impact of the PDAS on both transition of care and patient safety. For study endpoint purposes, transition of care was assessed by satisfactory completion and documentation of four important metrics: 1) appropriate enrollment in the anticoagulation clinic; 2) documented communication between the inpatient service responsible for anticoagulation and the outpatient anticoagulation clinic prior to patient discharge; 3) documented communication between the inpatient service responsible for anticoagulation and the physician responsible for outpatient management of the patient; 4) INR drawn within five days of hospital discharge. Documentation of communication for metric #2 and #3 was obtained by reviewing the electronic medical record system, particularly electronic discharge summaries and telephone encounter notes.
The primary safety endpoint was defined as a composite of any INR >5, any episode of major bleeding, or development of new thrombosis. This endpoint was met if any of these events occurred either during the index hospitalization or within 30 days of hospital discharge. Major bleeding was identified by review of outpatient anticoagulation clinic encounters and the patient's electronic medical record (includes all inpatient and outpatient encounters within Henry Ford Health System) by using the International Society of Thrombosis and Haemostasis standard and was defined as fatal bleeding or symptomatic bleeding in a critical area or organ (intracranial, intraspinal, intraocular, retroperitoneal, intraarticular, pericardial, or intramuscular with compartment syndrome), or bleeding causing a reduction in hemoglobin levels of 2 g/dL or more, or leading to transfusion of two or more units of blood or red cells.9 New thrombosis was defined as documentation of any of the following: deep vein thrombosis, pulmonary embolism, or cardioembolic stroke. Need for dose adjustment at the first anticoagulation clinic visit after discharge was evaluated as a secondary endpoint.
All analyses compared the PDAS to the control group. In addition, a planned comparison of patients in the PDAS and control groups who were newly initiated to warfarin during the study hospitalization (newly initiated subgroup) and those who were taking warfarin on admission (existing warfarin subgroup) was also undertaken. It was expected that these subgroup analyses would likely be underpowered, however, the potential implications of a service such as this could differ based on history of warfarin use. Therefore, these analyses were planned for exploratory purposes. In order to determine the impact of risk factors for altered warfarin pharmacodynamic response on the safety endpoint, post hoc subgroup analyses were performed based on demographics and clinical characteristics.
Data Analysis
Data are presented as mean standard deviation or proportion, as appropriate. A P‐value of less than 0.05 was considered significant for all comparisons and all tests were two‐tailed.
Intervention and control groups were compared with Student's t test, MannWhitney U test, chi‐square or Fishers exact test, as appropriate. Relative risk (RR) and 95% confidence intervals (CI) were calculated for all primary analyses. All statistical analyses were performed with SPSS v.12.0 (SPSS Inc, Chicago, IL).
It was estimated that a sample size of 250 patients per group would provide greater than 80% power to detect at least a 50% improvement in both the transition of care and primary safety endpoints, with implementation of the PDAS. This calculation is based on the following assumptions: alpha = 0.05; expected control group achievement of the four transition of care metrics = 50%; rate of safety endpoint for the control group = 20%.4
RESULTS
Baseline Characteristics
During the study period, 1360 patients were admitted to the study units. A total of 377 and 483 patients were found to be ineligible for inclusion on the PDAS and control units, respectively. These patients were ineligible because they did not follow up in the Henry Ford Medical Group outpatient anticoagulation clinic. In total, 500 patients were included in the analysis. Patients (n = 145) who were newly initiated on warfarin made up 29% of the total population. Table 2 presents baseline clinical characteristics for patients in the PDAS and control groups, showing increased age, and a greater proportion of patients with heart failure and LVADs in the PDAS group. Patients in the PDAS group had significantly longer hospital stays, however, these increases were driven by a longer length of stay among the advanced heart failure service patients that were managed by the PDAS.
| PDAS (n = 250) | Control (n = 250) | P Value | |
|---|---|---|---|
| |||
| Demographic data | |||
| Age (mean SD) | 64.1 15.6 | 68.0 14.9 | 0.004 |
| Male gender | 54.0% | 56.4% | 0.589 |
| Caucasian race | 44.4% | 50.4% | 0.179 |
| Admitted to a cardiology unit | 78.8% | 74.8% | 0.289 |
| Length of stay (mean SD) | 8.13 7.04 | 6.29 5.63 | 0.001 |
| No heart failure history: length of stay (mean SD) | 6.83 4.53 | 6.15 5.14 | 0.288 |
| Heart failure history: length of stay (mean SD) | 9.09 8.31 | 6.45 6.15 | 0.004 |
| History of heart failure* | 57.6% | 47.6% | 0.025 |
| Heart failure with an LVAD | 14.0% | 0.4% | <0.001 |
| Indication for anticoagulation | |||
| Venous thromboembolism | 21.6% | 18.4% | 0.371 |
| Atrial fibrillation | 54.4% | 66.4% | 0.006 |
| Other | 24.0% | 15.2% | 0.013 |
| Primary admission diagnosis* | |||
| Heart failure | 25.6%* | 21.6% | 0.292 |
| Atrial fibrillation | 16.4% | 20.8% | 0.206 |
| Acute coronary syndrome | 13.6% | 17.6% | 0.218 |
| Venous thromboembolism | 4.8% | 4.8% | 1.00 |
| Infection | 12.4% | 10.0% | 0.395 |
| Bleeding | 1.6% | 1.2% | 0.703 |
Early Warfarin Management
Warfarin management metrics are presented in Table 3. The number of inpatient days prescribed warfarin was increased in the PDAS group by greater than one day while PDAS patients required significantly less dosage adjustment at first outpatient follow‐up visit. Similar to increases noted with length of stay, increases in inpatient warfarin days were likely driven by patients with severe heart failure managed by the PDAS.
| Warfarin Dosing | PDAS (n = 250) | Control (n = 250) | P Value |
|---|---|---|---|
| |||
| Initial dose (mean SD) | 5.23 2.37 | 4.99 2.07 | 0.245 |
| Discharge dose (mean SD) | 5.15 2.52 | 4.91 2.14 | 0.258 |
| INR at discharge (mean SD) | 2.07 0.73 | 2.04 0.73 | 0.660 |
| Therapeutic INR at discharge | 40.8% | 38.0% | 0.522 |
| Inpatient warfarin days (mean SD) | 4.97 4.30 | 3.68 2.69 | <0.001 |
| No heart failure: inpatient warfarin days (mean SD) | 4.09 2.49 | 3.60 2.67 | 0.148 |
| Heart failure: inpatient warfarin days (mean SD) | 5.62 5.16 | 3.76 2.71 | <0.001 |
| Dose change required at first follow‐up visit | 44.8% | 72.6% | <0.001 |
Transition of Care
Transition of care results are presented in Table 4. Full compliance and achievement of the transition of care metrics occurred significantly more often in the PDAS versus control patients with markedly increased rates of documented communication between inpatient providers and both outpatient anticoagulation clinic staff and outpatient physicians. Early follow‐up INR monitoring also occurred more frequently in the PDAS patients. The PDAS patients experienced greater compliance with the transition of care metrics regardless of whether they were in the newly initiated or existing warfarin subgroups (data not shown).
| Transition of Care | PDAS (n = 250) | Control (n = 250) | Relative Risk (95% CI) | P Value |
|---|---|---|---|---|
| ||||
| 100% Communication bundle* compliance, % (n) | 75.6% (189) | 2.8% (7) | 27.0 (13.056.2) | <0.001 |
| Appropriately enrolled in the AC clinic, % (n) | 97.2% (243) | 95.2% (238) | 1.02 (0.991.06) | 0.242 |
| Communication: inpatient service and outpatient physician, % (n) | 99.6% (249) | 12.4% (31) | 8.03 (5.7811.2) | <0.001 |
| Communication: inpatient clinicians and AC clinic staff, % (n) | 98.8% (247) | 14.8% (37) | 6.68 (4.969.00) | <0.001 |
| INR drawn within five days of hospital discharge, % (n) | 78.4% (196) | 66.4% (166) | 1.18 (1.061.32) | 0.003 |
| 30‐Day Composite safety endpoint, % (n) | 10.0% (25) | 14.8% (37) | 0.68 (0.421.09) | 0.103 |
| Inpatient + 30‐day INR >5, % (n) | 9.6% (24) | 14.8% (37) | 0.65 (0.401.05) | 0.076 |
| Inpatient + 30‐day major bleeding, % (n) | 0.8% (2) | 0.4% (1) | 2.00 (0.1821.9) | 0.563 |
| Inpatient + 30‐day thrombosis, % (n) | 0% (0) | 0% (0) | N/A | N/A |
Anticoagulant Safety
Safety endpoint data is presented in Table 4. The composite safety outcome of INR >5, major bleeding event, or thrombosis occurred in 12.4% of all patients with no early thrombotic events and only three major bleeding events recorded. Excessive INR values >5 occurred less frequently in the PDAS patients, however, differences in this metric and the composite safety outcome were not significantly different. Safety endpoint results in the overall population were driven by a reduction in INR values >5 among newly initiated warfarin patients in the PDAS group (PDAS: 9.5% vs control: 19.7%; P = 0.079; Figure 1). Other subgroup analyses relating to the safety endpoint are presented in Figure 1.
DISCUSSION
This article describes a systematic intervention designed to improve anticoagulation safety and efficacy in the hospital and during the transition to the postdischarge setting. Implementation of a PDAS did not impact patient bleeding and thrombotic outcomes, but did result in improved coordination and documentation of warfarin management and subsequent enhancement in the transition of the anticoagulated patient from the inpatient‐to‐outpatient setting with the Pharmacist‐Directed Anticoagulation Service.
Limited previous work has investigated the role of an anticoagulation service in inpatient management of anticoagulation.48 Only one published study has investigated the impact of this type of service on transition of care issues with warfarin, as was done in our study.7 In that study, management by an inpatient anticoagulation service resulted in a greater proportion of patients referred to an anticoagulation clinic for management (P = 0.001), more patients presenting to the anticoagulation clinic with a therapeutic INR (P = 0.001), and fewer patients presenting to the clinic with supratherapeutic levels of anticoagulation (P = 0.002). These results are somewhat analogous to our findings, in that patients in our study were less likely to require a dose change at the first clinic follow‐up visit after discharge or to have INR values 5.
We completed several subgroup analyses to thoroughly explore the impact of the PDAS on the safety endpoint. While firm conclusions cannot be drawn from these subgroup analyses, some hypothesis‐generating observations can be made. First, there was a greater impact of the PDAS on the safety endpoint in patients who are usually more sensitive to the effects of warfarin and therefore more challenging to manage.2 The impact of the PDAS was also greater among patients whose length of stay was more than five days (population median). This is significant because it suggests that when the opportunity for adverse events and miscommunication is greatest (ie, during hospitalizations of longer duration), there appears to be improvement in the safety endpoint with the PDAS.
To our knowledge, this study was the first to explore the impact of an inpatient anticoagulation service on the care of both newly initiated and existing warfarin patients, rather than only patients newly initiated on warfarin. As expected, the greatest influence of the PDAS on the safety endpoint was observed among the newly initiated patients. While the safety impact of the PDAS was noted most significantly among the newly initiated patients, the PDAS had a positive effect on the transition of care metrics regardless of previous warfarin use.
A limitation of our study should be mentioned. While we employed a design in which randomly selected units were exposed to the PDAS, individual patients were not randomized to the service. This cluster randomized design was chosen because it mimics quality improvement processes that would be rolled out to a hospital nursing unit. While lack of randomization at the patient level is a limitation, our cluster randomized study design is pragmatic and represents an improvement over the existing published before and after quasi‐experimental studies in this area.48
An improved system for documentation of communication was built into the PDAS processes on implementation of the service. However, it should also be noted that inadequate communication between inpatient and outpatient providers was an identified root cause for adverse events with warfarin in our institution prior to implementation of the PDAS. Therefore, improvement in the transition of care metrics with the PDAS were likely due to a combination of both improved documentation and true improvement in communication.
The approach of the PDAS was refined in several ways early after implementation of the service. Some major notable improvements included the development of a systematic approach to ensuring appropriate follow‐up and transition of care for patients being discharged to a skilled nursing facility, and creation of a system that required a mandatory conversation between the PDAS and a surgical service if anticoagulation is ordered within 48 hours of a major procedure. An upcoming improvement to the service will be to transition from a home grown electronic database, which was built for the purposes of streamlining clinical workflow and data collection, to a commercially available software program that has recently become available for management of inpatient anticoagulation. The major advantage of the new program will be the clinical decision support capabilities that will help to further streamline the service and allow for greater efficiency.
To understand implications of our PDAS intervention, it is important to remember that the majority of study patients were prescribed warfarin prior to hospital admission, that patients in both study groups were enrolled in an established, multisite anticoagulation clinic, and that patients in both groups were managed with a comprehensive inpatientoutpatient electronic medical record. Therefore, all providers caring for these patients had real‐time access to all warfarin dosing and dose adjustments, INR results, and anticoagulation clinic encounters, even though formal communication between providers was infrequently documented for control group patients in our electronic medical record. Study patients had low rates of bleeding and no adverse thrombotic outcomes across both treatment groups. Therefore, this type of model is likely to produce larger gains in communication and safety outcomes in health care systems without established anticoagulation clinics or comprehensive electronic medical records.
The PDAS was enthusiastically accepted by providers at our institution and expanded hospital‐wide after completion of this pilot. The PDAS model is a viable approach to standardize anticoagulant management with a goal of improving anticoagulant safety in the inpatient setting. Assessment of the effectiveness of models such as the PDAS for improving anticoagulant safety in the inpatient setting is particularly relevant with the current expectations for hospitals set by The Joint Commission's NPSG.03.05.01.3 More importantly the PDAS model can be an option for improving the transition of the anticoagulated patient from the inpatient‐to‐outpatient setting. Follow‐up with the anticoagulation clinic occurred earlier with the PDAS and, while this study was not designed to evaluate the impact of this new service on rehospitalization, recent literature suggests that earlier follow‐up after discharge leads to less rehospitalization.10 Finally, it may be possible to adapt this model to provide more intensive medication therapy management and monitoring for hospitalized patients with other complicated medication regimens or chronic disease.
CONCLUSION
The clinical pharmacist is uniquely prepared to manage inter‐individual variability in pharmacodynamic response to drug therapy, as well as to provide high‐quality patient education. This study evaluated a new model of inpatient warfarin management, in which warfarin dosing, monitoring, patient education, and transition of care was coordinated by a specialized team of clinical pharmacists that worked in collaboration with physicians and outpatient anticoagulation clinic staff. Safety and efficiency of the care provided by this new service was improved in certain subsets of more complex patients. The major advantage of this service was improvement in patient handoff, improved communication, and earlier patient follow‐up after discharge. Therefore, implementation of a Pharmacist‐Directed Anticoagulation Service provides a net improvement in quality of care for the patient taking warfarin in the inpatient setting.
Acknowledgements
The authors acknowledge the efforts of the PDAS staff: Nassif Abi‐Samra, Pam Holland, Sara Lanfear, and Gail Washington. This work would not have been possible without the dedication of these pharmacists. All authors had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
- U.S. Pharmacopeia. USP Patient Safety CapsLink: January 2008. Available at: http://www.usp.org/pdf/EN/patientSafety/capsLink2008–01‐01.pdf. Accessed March 19,2010.
- ,,,,,.Pharmacology and management of the vitamin K antagonists: American College of Chest Physicians evidence‐based clinical practice guidelines (8th ed.).Chest.2008;133:160S–198S.
- The Joint Commission. 2009 National Patient Safety Goals. Available at: http://www.jointcommission.org/NR/rdonlyres/31666E86‐E7F4–423E‐9BE8‐F05BD1CB0AA8/0/HAP_NPSG.pdf. Accessed May 6,2010.
- ,,, et al.Optimization of inpatient warfarin therapy: impact of a daily consultation by a pharmacist‐managed anticoagulation service.Ann Pharmacother.2000;34:567–572.
- ,,, et al.Pharmacy‐managed protocol for warfarin use in orthopedic surgery patients.Am J Health‐Syst Pharm.1995;52:1310–1316.
- .Pharmacist involvement with warfarin dosing for inpatients.Pharm World Sci.2001;23:31–35.
- ,,.Evaluation of a pharmacy‐managed warfarin‐monitoring service to coordinate inpatient and outpatient therapy.Am J Hosp Pharm.1992;49:387–394.
- ,.Implementation and evaluation of a pharmacist‐assisted warfarin dosing program.Can J Hosp Pharm.1997;50:169–175.
- ,.Definition of major bleeding in clinical investigations of antihemostatic medicinal products in non‐surgical patients.J Thromb Haemostasis.2005;3:692–694.
- ,,.Rehospitalizations among patients in the Medicare fee‐for‐service program.N Engl J Med.2009;360:1418–1428.
Anticoagulants are one of the most common drug classes involved in medication errors and adverse events. Warfarin, an anticoagulant that plays a key role in the management of many disease states, is implicated in approximately 30% of reported anticoagulant‐related errors.1 Anticoagulation with warfarin is complicated by inter‐individual variability in response to therapy, clinically significant drug interactions, a narrow therapeutic window, and the need for frequent and lifelong monitoring.2
In the hospital setting, warfarin use is complicated due to patient handoff among health care providers, and acute illnesses that impact sensitivity and response to warfarin. Common causes of errors with anticoagulants are knowledge deficits, failure to follow policy/procedure/protocol, and communication issues.1 An added opportunity for warfarin‐related medication errors is the risk associated with the transition from the inpatient‐to‐outpatient setting. Due to the risk and complexity associated with anticoagulant medications, the Joint Commission instituted National Patient Safety Goal (NPSG) 03.05.01 (formerly NPSG 3E): a series of requirements intended to Reduce the likelihood of patient harm with the use of anticoagulation therapy.3 In order to optimally address this National Patient Safety Goal, a systematic intervention would be required to impact each step of the medication use process for anticoagulants.
Several studies have suggested that dedicated anticoagulation management services or clinics improve anticoagulation management in the outpatient setting.2 Non‐physician providers, primarily pharmacists and nurses, frequently manage outpatient anticoagulation management services or clinics. However, very few studies have evaluated the impact of a warfarin management service in the inpatient hospital setting.48 While the few available studies suggest some benefit associated with an inpatient anticoagulation management service, a minority of these studies have assessed the role of these services in facilitating the transition of the anticoagulated patient to the outpatient setting.7
In order to improve anticoagulation management and safety, our institution implemented an inpatient Pharmacist‐Directed Anticoagulation Service (PDAS). The purpose of this study was to evaluate the impact of this service on both transition of care and safety of patients receiving warfarin anticoagulation.
METHODS
This study was completed at Henry Ford Hospital, an 802‐bed, tertiary care, level 1 trauma and academic medical center in Detroit, MI. The study was carried out between November 2007 and June 2009. The study was approved by the Henry Ford Hospital Institutional Review Board with waiver of consent.
Patients
This was a prospective cluster randomized study. All patients admitted to two internal medicine units (IM1 or IM2) or two cardiology units (Card1 or Card2), who received at least one inpatient dose of warfarin, were eligible for inclusion. Patients were included regardless of whether warfarin was newly initiated during the index admission (newly initiated patients) or was continuation of existing anticoagulation (existing warfarin patients). In order to ensure that patient data following discharge would be available for analysis, patients were excluded from this analysis if they were not scheduled to follow‐up in the Henry Ford Medical Group outpatient anticoagulation clinics after discharge, however, these patients were cared for by the PDAS service in the usual manner.
Study Design
Prior to implementation of the PDAS, one internal medicine and one cardiology unit was randomly selected to receive the PDAS intervention (IM1 and Card1), while the other two units (IM2 and Card2) served as control units. These hospital units were selected because anticoagulants are frequently used on these units and the patient population is generally similar between the two internal medicine and two cardiology unitswith exception that Card1 unit also contains a specialized service for advanced heart failure and left ventricular assist device (LVAD) patients. Of note, there was significant expansion of the heart failure service and LVAD program during the time frame of the study, accounting for a greater number of more complicated patients on the Card1 (PDAS) unit.
Specific responsibilities of the PDAS related to warfarin are detailed in Table 1. The PDAS was implemented in September 2007 as a system‐based change to improve anticoagulant safety at our institution. The goals of this service were to improve communication regarding anticoagulation; to improve safety as patients transition from the inpatient‐to‐outpatient settings; and to standardize anticoagulant dosing, monitoring, and patient education. For patients taking warfarin, who are cared for by a health system‐affiliated physician, the PDAS collaborates with our outpatient anticoagulation clinics in order to facilitate transition from the inpatient‐to‐outpatient setting. The Henry Ford Health System has an established, multisite outpatient Anticoagulation Clinic with >5000 patients actively receiving warfarin dosing and monitoring. The anticoagulation clinics are staffed by nurses and pharmacists who provide standardized management of warfarin for patients of all physicians within our health system and provide consistent high‐quality care (average time in international normalized ratio [INR] goal range = 68.2%). The anticoagulation clinics have been in existence since 1992. The PDAS is comprised of three full‐time and two part‐time pharmacists whose responsibilities are limited to the management of anticoagulation throughout the hospital.
| Inpatient Care | Patient Education | Transition of Care |
|---|---|---|
| ||
| Initial dose selection and daily dose adjustments after warfarin is initiated by primary team | Comprehensive education provided verbally and via written communication utilizing the Krames database. | Contact anticoagulation‐responsible physician and anticoagulation clinic via phone. |
| Provide written dosing regimen to patient and provide date for first INR postdischarge. | ||
| Daily laboratory monitoring | Education provided is standardized between inpatient and outpatient settings. | Create electronic Anticoagulation Discharge Summary. Document communication with the outpatient clinicians, reason for admission, steps taken to manage warfarin drug interactions, and warfarin doses administered during stay, discharge warfarin dose and follow‐up date. |
The PDAS was staffed by repurposing pharmacist staff. All pharmacists had either several years of general medicine‐based clinical practice experience or residency training, or both. Pharmacists were oriented to service responsibilities by spending approximately one week in the outpatient anticoagulation clinic and completing focused review of internal and external anticoagulation guidelines.
In the control group, management of anticoagulation and transition of care occurred at the discretion of the primary care team. The primary team had access to a clinical pharmacist, who was not part of the PDAS, seven days per week. However, the primary team was not able to consult the PDAS.
This study was primarily designed to assess the impact of the PDAS on both transition of care and patient safety. For study endpoint purposes, transition of care was assessed by satisfactory completion and documentation of four important metrics: 1) appropriate enrollment in the anticoagulation clinic; 2) documented communication between the inpatient service responsible for anticoagulation and the outpatient anticoagulation clinic prior to patient discharge; 3) documented communication between the inpatient service responsible for anticoagulation and the physician responsible for outpatient management of the patient; 4) INR drawn within five days of hospital discharge. Documentation of communication for metric #2 and #3 was obtained by reviewing the electronic medical record system, particularly electronic discharge summaries and telephone encounter notes.
The primary safety endpoint was defined as a composite of any INR >5, any episode of major bleeding, or development of new thrombosis. This endpoint was met if any of these events occurred either during the index hospitalization or within 30 days of hospital discharge. Major bleeding was identified by review of outpatient anticoagulation clinic encounters and the patient's electronic medical record (includes all inpatient and outpatient encounters within Henry Ford Health System) by using the International Society of Thrombosis and Haemostasis standard and was defined as fatal bleeding or symptomatic bleeding in a critical area or organ (intracranial, intraspinal, intraocular, retroperitoneal, intraarticular, pericardial, or intramuscular with compartment syndrome), or bleeding causing a reduction in hemoglobin levels of 2 g/dL or more, or leading to transfusion of two or more units of blood or red cells.9 New thrombosis was defined as documentation of any of the following: deep vein thrombosis, pulmonary embolism, or cardioembolic stroke. Need for dose adjustment at the first anticoagulation clinic visit after discharge was evaluated as a secondary endpoint.
All analyses compared the PDAS to the control group. In addition, a planned comparison of patients in the PDAS and control groups who were newly initiated to warfarin during the study hospitalization (newly initiated subgroup) and those who were taking warfarin on admission (existing warfarin subgroup) was also undertaken. It was expected that these subgroup analyses would likely be underpowered, however, the potential implications of a service such as this could differ based on history of warfarin use. Therefore, these analyses were planned for exploratory purposes. In order to determine the impact of risk factors for altered warfarin pharmacodynamic response on the safety endpoint, post hoc subgroup analyses were performed based on demographics and clinical characteristics.
Data Analysis
Data are presented as mean standard deviation or proportion, as appropriate. A P‐value of less than 0.05 was considered significant for all comparisons and all tests were two‐tailed.
Intervention and control groups were compared with Student's t test, MannWhitney U test, chi‐square or Fishers exact test, as appropriate. Relative risk (RR) and 95% confidence intervals (CI) were calculated for all primary analyses. All statistical analyses were performed with SPSS v.12.0 (SPSS Inc, Chicago, IL).
It was estimated that a sample size of 250 patients per group would provide greater than 80% power to detect at least a 50% improvement in both the transition of care and primary safety endpoints, with implementation of the PDAS. This calculation is based on the following assumptions: alpha = 0.05; expected control group achievement of the four transition of care metrics = 50%; rate of safety endpoint for the control group = 20%.4
RESULTS
Baseline Characteristics
During the study period, 1360 patients were admitted to the study units. A total of 377 and 483 patients were found to be ineligible for inclusion on the PDAS and control units, respectively. These patients were ineligible because they did not follow up in the Henry Ford Medical Group outpatient anticoagulation clinic. In total, 500 patients were included in the analysis. Patients (n = 145) who were newly initiated on warfarin made up 29% of the total population. Table 2 presents baseline clinical characteristics for patients in the PDAS and control groups, showing increased age, and a greater proportion of patients with heart failure and LVADs in the PDAS group. Patients in the PDAS group had significantly longer hospital stays, however, these increases were driven by a longer length of stay among the advanced heart failure service patients that were managed by the PDAS.
| PDAS (n = 250) | Control (n = 250) | P Value | |
|---|---|---|---|
| |||
| Demographic data | |||
| Age (mean SD) | 64.1 15.6 | 68.0 14.9 | 0.004 |
| Male gender | 54.0% | 56.4% | 0.589 |
| Caucasian race | 44.4% | 50.4% | 0.179 |
| Admitted to a cardiology unit | 78.8% | 74.8% | 0.289 |
| Length of stay (mean SD) | 8.13 7.04 | 6.29 5.63 | 0.001 |
| No heart failure history: length of stay (mean SD) | 6.83 4.53 | 6.15 5.14 | 0.288 |
| Heart failure history: length of stay (mean SD) | 9.09 8.31 | 6.45 6.15 | 0.004 |
| History of heart failure* | 57.6% | 47.6% | 0.025 |
| Heart failure with an LVAD | 14.0% | 0.4% | <0.001 |
| Indication for anticoagulation | |||
| Venous thromboembolism | 21.6% | 18.4% | 0.371 |
| Atrial fibrillation | 54.4% | 66.4% | 0.006 |
| Other | 24.0% | 15.2% | 0.013 |
| Primary admission diagnosis* | |||
| Heart failure | 25.6%* | 21.6% | 0.292 |
| Atrial fibrillation | 16.4% | 20.8% | 0.206 |
| Acute coronary syndrome | 13.6% | 17.6% | 0.218 |
| Venous thromboembolism | 4.8% | 4.8% | 1.00 |
| Infection | 12.4% | 10.0% | 0.395 |
| Bleeding | 1.6% | 1.2% | 0.703 |
Early Warfarin Management
Warfarin management metrics are presented in Table 3. The number of inpatient days prescribed warfarin was increased in the PDAS group by greater than one day while PDAS patients required significantly less dosage adjustment at first outpatient follow‐up visit. Similar to increases noted with length of stay, increases in inpatient warfarin days were likely driven by patients with severe heart failure managed by the PDAS.
| Warfarin Dosing | PDAS (n = 250) | Control (n = 250) | P Value |
|---|---|---|---|
| |||
| Initial dose (mean SD) | 5.23 2.37 | 4.99 2.07 | 0.245 |
| Discharge dose (mean SD) | 5.15 2.52 | 4.91 2.14 | 0.258 |
| INR at discharge (mean SD) | 2.07 0.73 | 2.04 0.73 | 0.660 |
| Therapeutic INR at discharge | 40.8% | 38.0% | 0.522 |
| Inpatient warfarin days (mean SD) | 4.97 4.30 | 3.68 2.69 | <0.001 |
| No heart failure: inpatient warfarin days (mean SD) | 4.09 2.49 | 3.60 2.67 | 0.148 |
| Heart failure: inpatient warfarin days (mean SD) | 5.62 5.16 | 3.76 2.71 | <0.001 |
| Dose change required at first follow‐up visit | 44.8% | 72.6% | <0.001 |
Transition of Care
Transition of care results are presented in Table 4. Full compliance and achievement of the transition of care metrics occurred significantly more often in the PDAS versus control patients with markedly increased rates of documented communication between inpatient providers and both outpatient anticoagulation clinic staff and outpatient physicians. Early follow‐up INR monitoring also occurred more frequently in the PDAS patients. The PDAS patients experienced greater compliance with the transition of care metrics regardless of whether they were in the newly initiated or existing warfarin subgroups (data not shown).
| Transition of Care | PDAS (n = 250) | Control (n = 250) | Relative Risk (95% CI) | P Value |
|---|---|---|---|---|
| ||||
| 100% Communication bundle* compliance, % (n) | 75.6% (189) | 2.8% (7) | 27.0 (13.056.2) | <0.001 |
| Appropriately enrolled in the AC clinic, % (n) | 97.2% (243) | 95.2% (238) | 1.02 (0.991.06) | 0.242 |
| Communication: inpatient service and outpatient physician, % (n) | 99.6% (249) | 12.4% (31) | 8.03 (5.7811.2) | <0.001 |
| Communication: inpatient clinicians and AC clinic staff, % (n) | 98.8% (247) | 14.8% (37) | 6.68 (4.969.00) | <0.001 |
| INR drawn within five days of hospital discharge, % (n) | 78.4% (196) | 66.4% (166) | 1.18 (1.061.32) | 0.003 |
| 30‐Day Composite safety endpoint, % (n) | 10.0% (25) | 14.8% (37) | 0.68 (0.421.09) | 0.103 |
| Inpatient + 30‐day INR >5, % (n) | 9.6% (24) | 14.8% (37) | 0.65 (0.401.05) | 0.076 |
| Inpatient + 30‐day major bleeding, % (n) | 0.8% (2) | 0.4% (1) | 2.00 (0.1821.9) | 0.563 |
| Inpatient + 30‐day thrombosis, % (n) | 0% (0) | 0% (0) | N/A | N/A |
Anticoagulant Safety
Safety endpoint data is presented in Table 4. The composite safety outcome of INR >5, major bleeding event, or thrombosis occurred in 12.4% of all patients with no early thrombotic events and only three major bleeding events recorded. Excessive INR values >5 occurred less frequently in the PDAS patients, however, differences in this metric and the composite safety outcome were not significantly different. Safety endpoint results in the overall population were driven by a reduction in INR values >5 among newly initiated warfarin patients in the PDAS group (PDAS: 9.5% vs control: 19.7%; P = 0.079; Figure 1). Other subgroup analyses relating to the safety endpoint are presented in Figure 1.

DISCUSSION
This article describes a systematic intervention designed to improve anticoagulation safety and efficacy in the hospital and during the transition to the postdischarge setting. Implementation of a PDAS did not impact patient bleeding and thrombotic outcomes, but did result in improved coordination and documentation of warfarin management and subsequent enhancement in the transition of the anticoagulated patient from the inpatient‐to‐outpatient setting with the Pharmacist‐Directed Anticoagulation Service.
Limited previous work has investigated the role of an anticoagulation service in inpatient management of anticoagulation.48 Only one published study has investigated the impact of this type of service on transition of care issues with warfarin, as was done in our study.7 In that study, management by an inpatient anticoagulation service resulted in a greater proportion of patients referred to an anticoagulation clinic for management (P = 0.001), more patients presenting to the anticoagulation clinic with a therapeutic INR (P = 0.001), and fewer patients presenting to the clinic with supratherapeutic levels of anticoagulation (P = 0.002). These results are somewhat analogous to our findings, in that patients in our study were less likely to require a dose change at the first clinic follow‐up visit after discharge or to have INR values 5.
We completed several subgroup analyses to thoroughly explore the impact of the PDAS on the safety endpoint. While firm conclusions cannot be drawn from these subgroup analyses, some hypothesis‐generating observations can be made. First, there was a greater impact of the PDAS on the safety endpoint in patients who are usually more sensitive to the effects of warfarin and therefore more challenging to manage.2 The impact of the PDAS was also greater among patients whose length of stay was more than five days (population median). This is significant because it suggests that when the opportunity for adverse events and miscommunication is greatest (ie, during hospitalizations of longer duration), there appears to be improvement in the safety endpoint with the PDAS.
To our knowledge, this study was the first to explore the impact of an inpatient anticoagulation service on the care of both newly initiated and existing warfarin patients, rather than only patients newly initiated on warfarin. As expected, the greatest influence of the PDAS on the safety endpoint was observed among the newly initiated patients. While the safety impact of the PDAS was noted most significantly among the newly initiated patients, the PDAS had a positive effect on the transition of care metrics regardless of previous warfarin use.
A limitation of our study should be mentioned. While we employed a design in which randomly selected units were exposed to the PDAS, individual patients were not randomized to the service. This cluster randomized design was chosen because it mimics quality improvement processes that would be rolled out to a hospital nursing unit. While lack of randomization at the patient level is a limitation, our cluster randomized study design is pragmatic and represents an improvement over the existing published before and after quasi‐experimental studies in this area.48
An improved system for documentation of communication was built into the PDAS processes on implementation of the service. However, it should also be noted that inadequate communication between inpatient and outpatient providers was an identified root cause for adverse events with warfarin in our institution prior to implementation of the PDAS. Therefore, improvement in the transition of care metrics with the PDAS were likely due to a combination of both improved documentation and true improvement in communication.
The approach of the PDAS was refined in several ways early after implementation of the service. Some major notable improvements included the development of a systematic approach to ensuring appropriate follow‐up and transition of care for patients being discharged to a skilled nursing facility, and creation of a system that required a mandatory conversation between the PDAS and a surgical service if anticoagulation is ordered within 48 hours of a major procedure. An upcoming improvement to the service will be to transition from a home grown electronic database, which was built for the purposes of streamlining clinical workflow and data collection, to a commercially available software program that has recently become available for management of inpatient anticoagulation. The major advantage of the new program will be the clinical decision support capabilities that will help to further streamline the service and allow for greater efficiency.
To understand implications of our PDAS intervention, it is important to remember that the majority of study patients were prescribed warfarin prior to hospital admission, that patients in both study groups were enrolled in an established, multisite anticoagulation clinic, and that patients in both groups were managed with a comprehensive inpatientoutpatient electronic medical record. Therefore, all providers caring for these patients had real‐time access to all warfarin dosing and dose adjustments, INR results, and anticoagulation clinic encounters, even though formal communication between providers was infrequently documented for control group patients in our electronic medical record. Study patients had low rates of bleeding and no adverse thrombotic outcomes across both treatment groups. Therefore, this type of model is likely to produce larger gains in communication and safety outcomes in health care systems without established anticoagulation clinics or comprehensive electronic medical records.
The PDAS was enthusiastically accepted by providers at our institution and expanded hospital‐wide after completion of this pilot. The PDAS model is a viable approach to standardize anticoagulant management with a goal of improving anticoagulant safety in the inpatient setting. Assessment of the effectiveness of models such as the PDAS for improving anticoagulant safety in the inpatient setting is particularly relevant with the current expectations for hospitals set by The Joint Commission's NPSG.03.05.01.3 More importantly the PDAS model can be an option for improving the transition of the anticoagulated patient from the inpatient‐to‐outpatient setting. Follow‐up with the anticoagulation clinic occurred earlier with the PDAS and, while this study was not designed to evaluate the impact of this new service on rehospitalization, recent literature suggests that earlier follow‐up after discharge leads to less rehospitalization.10 Finally, it may be possible to adapt this model to provide more intensive medication therapy management and monitoring for hospitalized patients with other complicated medication regimens or chronic disease.
CONCLUSION
The clinical pharmacist is uniquely prepared to manage inter‐individual variability in pharmacodynamic response to drug therapy, as well as to provide high‐quality patient education. This study evaluated a new model of inpatient warfarin management, in which warfarin dosing, monitoring, patient education, and transition of care was coordinated by a specialized team of clinical pharmacists that worked in collaboration with physicians and outpatient anticoagulation clinic staff. Safety and efficiency of the care provided by this new service was improved in certain subsets of more complex patients. The major advantage of this service was improvement in patient handoff, improved communication, and earlier patient follow‐up after discharge. Therefore, implementation of a Pharmacist‐Directed Anticoagulation Service provides a net improvement in quality of care for the patient taking warfarin in the inpatient setting.
Acknowledgements
The authors acknowledge the efforts of the PDAS staff: Nassif Abi‐Samra, Pam Holland, Sara Lanfear, and Gail Washington. This work would not have been possible without the dedication of these pharmacists. All authors had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Anticoagulants are one of the most common drug classes involved in medication errors and adverse events. Warfarin, an anticoagulant that plays a key role in the management of many disease states, is implicated in approximately 30% of reported anticoagulant‐related errors.1 Anticoagulation with warfarin is complicated by inter‐individual variability in response to therapy, clinically significant drug interactions, a narrow therapeutic window, and the need for frequent and lifelong monitoring.2
In the hospital setting, warfarin use is complicated due to patient handoff among health care providers, and acute illnesses that impact sensitivity and response to warfarin. Common causes of errors with anticoagulants are knowledge deficits, failure to follow policy/procedure/protocol, and communication issues.1 An added opportunity for warfarin‐related medication errors is the risk associated with the transition from the inpatient‐to‐outpatient setting. Due to the risk and complexity associated with anticoagulant medications, the Joint Commission instituted National Patient Safety Goal (NPSG) 03.05.01 (formerly NPSG 3E): a series of requirements intended to Reduce the likelihood of patient harm with the use of anticoagulation therapy.3 In order to optimally address this National Patient Safety Goal, a systematic intervention would be required to impact each step of the medication use process for anticoagulants.
Several studies have suggested that dedicated anticoagulation management services or clinics improve anticoagulation management in the outpatient setting.2 Non‐physician providers, primarily pharmacists and nurses, frequently manage outpatient anticoagulation management services or clinics. However, very few studies have evaluated the impact of a warfarin management service in the inpatient hospital setting.48 While the few available studies suggest some benefit associated with an inpatient anticoagulation management service, a minority of these studies have assessed the role of these services in facilitating the transition of the anticoagulated patient to the outpatient setting.7
In order to improve anticoagulation management and safety, our institution implemented an inpatient Pharmacist‐Directed Anticoagulation Service (PDAS). The purpose of this study was to evaluate the impact of this service on both transition of care and safety of patients receiving warfarin anticoagulation.
METHODS
This study was completed at Henry Ford Hospital, an 802‐bed, tertiary care, level 1 trauma and academic medical center in Detroit, MI. The study was carried out between November 2007 and June 2009. The study was approved by the Henry Ford Hospital Institutional Review Board with waiver of consent.
Patients
This was a prospective cluster randomized study. All patients admitted to two internal medicine units (IM1 or IM2) or two cardiology units (Card1 or Card2), who received at least one inpatient dose of warfarin, were eligible for inclusion. Patients were included regardless of whether warfarin was newly initiated during the index admission (newly initiated patients) or was continuation of existing anticoagulation (existing warfarin patients). In order to ensure that patient data following discharge would be available for analysis, patients were excluded from this analysis if they were not scheduled to follow‐up in the Henry Ford Medical Group outpatient anticoagulation clinics after discharge, however, these patients were cared for by the PDAS service in the usual manner.
Study Design
Prior to implementation of the PDAS, one internal medicine and one cardiology unit was randomly selected to receive the PDAS intervention (IM1 and Card1), while the other two units (IM2 and Card2) served as control units. These hospital units were selected because anticoagulants are frequently used on these units and the patient population is generally similar between the two internal medicine and two cardiology unitswith exception that Card1 unit also contains a specialized service for advanced heart failure and left ventricular assist device (LVAD) patients. Of note, there was significant expansion of the heart failure service and LVAD program during the time frame of the study, accounting for a greater number of more complicated patients on the Card1 (PDAS) unit.
Specific responsibilities of the PDAS related to warfarin are detailed in Table 1. The PDAS was implemented in September 2007 as a system‐based change to improve anticoagulant safety at our institution. The goals of this service were to improve communication regarding anticoagulation; to improve safety as patients transition from the inpatient‐to‐outpatient settings; and to standardize anticoagulant dosing, monitoring, and patient education. For patients taking warfarin, who are cared for by a health system‐affiliated physician, the PDAS collaborates with our outpatient anticoagulation clinics in order to facilitate transition from the inpatient‐to‐outpatient setting. The Henry Ford Health System has an established, multisite outpatient Anticoagulation Clinic with >5000 patients actively receiving warfarin dosing and monitoring. The anticoagulation clinics are staffed by nurses and pharmacists who provide standardized management of warfarin for patients of all physicians within our health system and provide consistent high‐quality care (average time in international normalized ratio [INR] goal range = 68.2%). The anticoagulation clinics have been in existence since 1992. The PDAS is comprised of three full‐time and two part‐time pharmacists whose responsibilities are limited to the management of anticoagulation throughout the hospital.
| Inpatient Care | Patient Education | Transition of Care |
|---|---|---|
| ||
| Initial dose selection and daily dose adjustments after warfarin is initiated by primary team | Comprehensive education provided verbally and via written communication utilizing the Krames database. | Contact anticoagulation‐responsible physician and anticoagulation clinic via phone. |
| Provide written dosing regimen to patient and provide date for first INR postdischarge. | ||
| Daily laboratory monitoring | Education provided is standardized between inpatient and outpatient settings. | Create electronic Anticoagulation Discharge Summary. Document communication with the outpatient clinicians, reason for admission, steps taken to manage warfarin drug interactions, and warfarin doses administered during stay, discharge warfarin dose and follow‐up date. |
The PDAS was staffed by repurposing pharmacist staff. All pharmacists had either several years of general medicine‐based clinical practice experience or residency training, or both. Pharmacists were oriented to service responsibilities by spending approximately one week in the outpatient anticoagulation clinic and completing focused review of internal and external anticoagulation guidelines.
In the control group, management of anticoagulation and transition of care occurred at the discretion of the primary care team. The primary team had access to a clinical pharmacist, who was not part of the PDAS, seven days per week. However, the primary team was not able to consult the PDAS.
This study was primarily designed to assess the impact of the PDAS on both transition of care and patient safety. For study endpoint purposes, transition of care was assessed by satisfactory completion and documentation of four important metrics: 1) appropriate enrollment in the anticoagulation clinic; 2) documented communication between the inpatient service responsible for anticoagulation and the outpatient anticoagulation clinic prior to patient discharge; 3) documented communication between the inpatient service responsible for anticoagulation and the physician responsible for outpatient management of the patient; 4) INR drawn within five days of hospital discharge. Documentation of communication for metric #2 and #3 was obtained by reviewing the electronic medical record system, particularly electronic discharge summaries and telephone encounter notes.
The primary safety endpoint was defined as a composite of any INR >5, any episode of major bleeding, or development of new thrombosis. This endpoint was met if any of these events occurred either during the index hospitalization or within 30 days of hospital discharge. Major bleeding was identified by review of outpatient anticoagulation clinic encounters and the patient's electronic medical record (includes all inpatient and outpatient encounters within Henry Ford Health System) by using the International Society of Thrombosis and Haemostasis standard and was defined as fatal bleeding or symptomatic bleeding in a critical area or organ (intracranial, intraspinal, intraocular, retroperitoneal, intraarticular, pericardial, or intramuscular with compartment syndrome), or bleeding causing a reduction in hemoglobin levels of 2 g/dL or more, or leading to transfusion of two or more units of blood or red cells.9 New thrombosis was defined as documentation of any of the following: deep vein thrombosis, pulmonary embolism, or cardioembolic stroke. Need for dose adjustment at the first anticoagulation clinic visit after discharge was evaluated as a secondary endpoint.
All analyses compared the PDAS to the control group. In addition, a planned comparison of patients in the PDAS and control groups who were newly initiated to warfarin during the study hospitalization (newly initiated subgroup) and those who were taking warfarin on admission (existing warfarin subgroup) was also undertaken. It was expected that these subgroup analyses would likely be underpowered, however, the potential implications of a service such as this could differ based on history of warfarin use. Therefore, these analyses were planned for exploratory purposes. In order to determine the impact of risk factors for altered warfarin pharmacodynamic response on the safety endpoint, post hoc subgroup analyses were performed based on demographics and clinical characteristics.
Data Analysis
Data are presented as mean standard deviation or proportion, as appropriate. A P‐value of less than 0.05 was considered significant for all comparisons and all tests were two‐tailed.
Intervention and control groups were compared with Student's t test, MannWhitney U test, chi‐square or Fishers exact test, as appropriate. Relative risk (RR) and 95% confidence intervals (CI) were calculated for all primary analyses. All statistical analyses were performed with SPSS v.12.0 (SPSS Inc, Chicago, IL).
It was estimated that a sample size of 250 patients per group would provide greater than 80% power to detect at least a 50% improvement in both the transition of care and primary safety endpoints, with implementation of the PDAS. This calculation is based on the following assumptions: alpha = 0.05; expected control group achievement of the four transition of care metrics = 50%; rate of safety endpoint for the control group = 20%.4
RESULTS
Baseline Characteristics
During the study period, 1360 patients were admitted to the study units. A total of 377 and 483 patients were found to be ineligible for inclusion on the PDAS and control units, respectively. These patients were ineligible because they did not follow up in the Henry Ford Medical Group outpatient anticoagulation clinic. In total, 500 patients were included in the analysis. Patients (n = 145) who were newly initiated on warfarin made up 29% of the total population. Table 2 presents baseline clinical characteristics for patients in the PDAS and control groups, showing increased age, and a greater proportion of patients with heart failure and LVADs in the PDAS group. Patients in the PDAS group had significantly longer hospital stays, however, these increases were driven by a longer length of stay among the advanced heart failure service patients that were managed by the PDAS.
| PDAS (n = 250) | Control (n = 250) | P Value | |
|---|---|---|---|
| |||
| Demographic data | |||
| Age (mean SD) | 64.1 15.6 | 68.0 14.9 | 0.004 |
| Male gender | 54.0% | 56.4% | 0.589 |
| Caucasian race | 44.4% | 50.4% | 0.179 |
| Admitted to a cardiology unit | 78.8% | 74.8% | 0.289 |
| Length of stay (mean SD) | 8.13 7.04 | 6.29 5.63 | 0.001 |
| No heart failure history: length of stay (mean SD) | 6.83 4.53 | 6.15 5.14 | 0.288 |
| Heart failure history: length of stay (mean SD) | 9.09 8.31 | 6.45 6.15 | 0.004 |
| History of heart failure* | 57.6% | 47.6% | 0.025 |
| Heart failure with an LVAD | 14.0% | 0.4% | <0.001 |
| Indication for anticoagulation | |||
| Venous thromboembolism | 21.6% | 18.4% | 0.371 |
| Atrial fibrillation | 54.4% | 66.4% | 0.006 |
| Other | 24.0% | 15.2% | 0.013 |
| Primary admission diagnosis* | |||
| Heart failure | 25.6%* | 21.6% | 0.292 |
| Atrial fibrillation | 16.4% | 20.8% | 0.206 |
| Acute coronary syndrome | 13.6% | 17.6% | 0.218 |
| Venous thromboembolism | 4.8% | 4.8% | 1.00 |
| Infection | 12.4% | 10.0% | 0.395 |
| Bleeding | 1.6% | 1.2% | 0.703 |
Early Warfarin Management
Warfarin management metrics are presented in Table 3. The number of inpatient days prescribed warfarin was increased in the PDAS group by greater than one day while PDAS patients required significantly less dosage adjustment at first outpatient follow‐up visit. Similar to increases noted with length of stay, increases in inpatient warfarin days were likely driven by patients with severe heart failure managed by the PDAS.
| Warfarin Dosing | PDAS (n = 250) | Control (n = 250) | P Value |
|---|---|---|---|
| |||
| Initial dose (mean SD) | 5.23 2.37 | 4.99 2.07 | 0.245 |
| Discharge dose (mean SD) | 5.15 2.52 | 4.91 2.14 | 0.258 |
| INR at discharge (mean SD) | 2.07 0.73 | 2.04 0.73 | 0.660 |
| Therapeutic INR at discharge | 40.8% | 38.0% | 0.522 |
| Inpatient warfarin days (mean SD) | 4.97 4.30 | 3.68 2.69 | <0.001 |
| No heart failure: inpatient warfarin days (mean SD) | 4.09 2.49 | 3.60 2.67 | 0.148 |
| Heart failure: inpatient warfarin days (mean SD) | 5.62 5.16 | 3.76 2.71 | <0.001 |
| Dose change required at first follow‐up visit | 44.8% | 72.6% | <0.001 |
Transition of Care
Transition of care results are presented in Table 4. Full compliance and achievement of the transition of care metrics occurred significantly more often in the PDAS versus control patients with markedly increased rates of documented communication between inpatient providers and both outpatient anticoagulation clinic staff and outpatient physicians. Early follow‐up INR monitoring also occurred more frequently in the PDAS patients. The PDAS patients experienced greater compliance with the transition of care metrics regardless of whether they were in the newly initiated or existing warfarin subgroups (data not shown).
| Transition of Care | PDAS (n = 250) | Control (n = 250) | Relative Risk (95% CI) | P Value |
|---|---|---|---|---|
| ||||
| 100% Communication bundle* compliance, % (n) | 75.6% (189) | 2.8% (7) | 27.0 (13.056.2) | <0.001 |
| Appropriately enrolled in the AC clinic, % (n) | 97.2% (243) | 95.2% (238) | 1.02 (0.991.06) | 0.242 |
| Communication: inpatient service and outpatient physician, % (n) | 99.6% (249) | 12.4% (31) | 8.03 (5.7811.2) | <0.001 |
| Communication: inpatient clinicians and AC clinic staff, % (n) | 98.8% (247) | 14.8% (37) | 6.68 (4.969.00) | <0.001 |
| INR drawn within five days of hospital discharge, % (n) | 78.4% (196) | 66.4% (166) | 1.18 (1.061.32) | 0.003 |
| 30‐Day Composite safety endpoint, % (n) | 10.0% (25) | 14.8% (37) | 0.68 (0.421.09) | 0.103 |
| Inpatient + 30‐day INR >5, % (n) | 9.6% (24) | 14.8% (37) | 0.65 (0.401.05) | 0.076 |
| Inpatient + 30‐day major bleeding, % (n) | 0.8% (2) | 0.4% (1) | 2.00 (0.1821.9) | 0.563 |
| Inpatient + 30‐day thrombosis, % (n) | 0% (0) | 0% (0) | N/A | N/A |
Anticoagulant Safety
Safety endpoint data is presented in Table 4. The composite safety outcome of INR >5, major bleeding event, or thrombosis occurred in 12.4% of all patients with no early thrombotic events and only three major bleeding events recorded. Excessive INR values >5 occurred less frequently in the PDAS patients, however, differences in this metric and the composite safety outcome were not significantly different. Safety endpoint results in the overall population were driven by a reduction in INR values >5 among newly initiated warfarin patients in the PDAS group (PDAS: 9.5% vs control: 19.7%; P = 0.079; Figure 1). Other subgroup analyses relating to the safety endpoint are presented in Figure 1.

DISCUSSION
This article describes a systematic intervention designed to improve anticoagulation safety and efficacy in the hospital and during the transition to the postdischarge setting. Implementation of a PDAS did not impact patient bleeding and thrombotic outcomes, but did result in improved coordination and documentation of warfarin management and subsequent enhancement in the transition of the anticoagulated patient from the inpatient‐to‐outpatient setting with the Pharmacist‐Directed Anticoagulation Service.
Limited previous work has investigated the role of an anticoagulation service in inpatient management of anticoagulation.48 Only one published study has investigated the impact of this type of service on transition of care issues with warfarin, as was done in our study.7 In that study, management by an inpatient anticoagulation service resulted in a greater proportion of patients referred to an anticoagulation clinic for management (P = 0.001), more patients presenting to the anticoagulation clinic with a therapeutic INR (P = 0.001), and fewer patients presenting to the clinic with supratherapeutic levels of anticoagulation (P = 0.002). These results are somewhat analogous to our findings, in that patients in our study were less likely to require a dose change at the first clinic follow‐up visit after discharge or to have INR values 5.
We completed several subgroup analyses to thoroughly explore the impact of the PDAS on the safety endpoint. While firm conclusions cannot be drawn from these subgroup analyses, some hypothesis‐generating observations can be made. First, there was a greater impact of the PDAS on the safety endpoint in patients who are usually more sensitive to the effects of warfarin and therefore more challenging to manage.2 The impact of the PDAS was also greater among patients whose length of stay was more than five days (population median). This is significant because it suggests that when the opportunity for adverse events and miscommunication is greatest (ie, during hospitalizations of longer duration), there appears to be improvement in the safety endpoint with the PDAS.
To our knowledge, this study was the first to explore the impact of an inpatient anticoagulation service on the care of both newly initiated and existing warfarin patients, rather than only patients newly initiated on warfarin. As expected, the greatest influence of the PDAS on the safety endpoint was observed among the newly initiated patients. While the safety impact of the PDAS was noted most significantly among the newly initiated patients, the PDAS had a positive effect on the transition of care metrics regardless of previous warfarin use.
A limitation of our study should be mentioned. While we employed a design in which randomly selected units were exposed to the PDAS, individual patients were not randomized to the service. This cluster randomized design was chosen because it mimics quality improvement processes that would be rolled out to a hospital nursing unit. While lack of randomization at the patient level is a limitation, our cluster randomized study design is pragmatic and represents an improvement over the existing published before and after quasi‐experimental studies in this area.48
An improved system for documentation of communication was built into the PDAS processes on implementation of the service. However, it should also be noted that inadequate communication between inpatient and outpatient providers was an identified root cause for adverse events with warfarin in our institution prior to implementation of the PDAS. Therefore, improvement in the transition of care metrics with the PDAS were likely due to a combination of both improved documentation and true improvement in communication.
The approach of the PDAS was refined in several ways early after implementation of the service. Some major notable improvements included the development of a systematic approach to ensuring appropriate follow‐up and transition of care for patients being discharged to a skilled nursing facility, and creation of a system that required a mandatory conversation between the PDAS and a surgical service if anticoagulation is ordered within 48 hours of a major procedure. An upcoming improvement to the service will be to transition from a home grown electronic database, which was built for the purposes of streamlining clinical workflow and data collection, to a commercially available software program that has recently become available for management of inpatient anticoagulation. The major advantage of the new program will be the clinical decision support capabilities that will help to further streamline the service and allow for greater efficiency.
To understand implications of our PDAS intervention, it is important to remember that the majority of study patients were prescribed warfarin prior to hospital admission, that patients in both study groups were enrolled in an established, multisite anticoagulation clinic, and that patients in both groups were managed with a comprehensive inpatientoutpatient electronic medical record. Therefore, all providers caring for these patients had real‐time access to all warfarin dosing and dose adjustments, INR results, and anticoagulation clinic encounters, even though formal communication between providers was infrequently documented for control group patients in our electronic medical record. Study patients had low rates of bleeding and no adverse thrombotic outcomes across both treatment groups. Therefore, this type of model is likely to produce larger gains in communication and safety outcomes in health care systems without established anticoagulation clinics or comprehensive electronic medical records.
The PDAS was enthusiastically accepted by providers at our institution and expanded hospital‐wide after completion of this pilot. The PDAS model is a viable approach to standardize anticoagulant management with a goal of improving anticoagulant safety in the inpatient setting. Assessment of the effectiveness of models such as the PDAS for improving anticoagulant safety in the inpatient setting is particularly relevant with the current expectations for hospitals set by The Joint Commission's NPSG.03.05.01.3 More importantly the PDAS model can be an option for improving the transition of the anticoagulated patient from the inpatient‐to‐outpatient setting. Follow‐up with the anticoagulation clinic occurred earlier with the PDAS and, while this study was not designed to evaluate the impact of this new service on rehospitalization, recent literature suggests that earlier follow‐up after discharge leads to less rehospitalization.10 Finally, it may be possible to adapt this model to provide more intensive medication therapy management and monitoring for hospitalized patients with other complicated medication regimens or chronic disease.
CONCLUSION
The clinical pharmacist is uniquely prepared to manage inter‐individual variability in pharmacodynamic response to drug therapy, as well as to provide high‐quality patient education. This study evaluated a new model of inpatient warfarin management, in which warfarin dosing, monitoring, patient education, and transition of care was coordinated by a specialized team of clinical pharmacists that worked in collaboration with physicians and outpatient anticoagulation clinic staff. Safety and efficiency of the care provided by this new service was improved in certain subsets of more complex patients. The major advantage of this service was improvement in patient handoff, improved communication, and earlier patient follow‐up after discharge. Therefore, implementation of a Pharmacist‐Directed Anticoagulation Service provides a net improvement in quality of care for the patient taking warfarin in the inpatient setting.
Acknowledgements
The authors acknowledge the efforts of the PDAS staff: Nassif Abi‐Samra, Pam Holland, Sara Lanfear, and Gail Washington. This work would not have been possible without the dedication of these pharmacists. All authors had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
- U.S. Pharmacopeia. USP Patient Safety CapsLink: January 2008. Available at: http://www.usp.org/pdf/EN/patientSafety/capsLink2008–01‐01.pdf. Accessed March 19,2010.
- ,,,,,.Pharmacology and management of the vitamin K antagonists: American College of Chest Physicians evidence‐based clinical practice guidelines (8th ed.).Chest.2008;133:160S–198S.
- The Joint Commission. 2009 National Patient Safety Goals. Available at: http://www.jointcommission.org/NR/rdonlyres/31666E86‐E7F4–423E‐9BE8‐F05BD1CB0AA8/0/HAP_NPSG.pdf. Accessed May 6,2010.
- ,,, et al.Optimization of inpatient warfarin therapy: impact of a daily consultation by a pharmacist‐managed anticoagulation service.Ann Pharmacother.2000;34:567–572.
- ,,, et al.Pharmacy‐managed protocol for warfarin use in orthopedic surgery patients.Am J Health‐Syst Pharm.1995;52:1310–1316.
- .Pharmacist involvement with warfarin dosing for inpatients.Pharm World Sci.2001;23:31–35.
- ,,.Evaluation of a pharmacy‐managed warfarin‐monitoring service to coordinate inpatient and outpatient therapy.Am J Hosp Pharm.1992;49:387–394.
- ,.Implementation and evaluation of a pharmacist‐assisted warfarin dosing program.Can J Hosp Pharm.1997;50:169–175.
- ,.Definition of major bleeding in clinical investigations of antihemostatic medicinal products in non‐surgical patients.J Thromb Haemostasis.2005;3:692–694.
- ,,.Rehospitalizations among patients in the Medicare fee‐for‐service program.N Engl J Med.2009;360:1418–1428.
- U.S. Pharmacopeia. USP Patient Safety CapsLink: January 2008. Available at: http://www.usp.org/pdf/EN/patientSafety/capsLink2008–01‐01.pdf. Accessed March 19,2010.
- ,,,,,.Pharmacology and management of the vitamin K antagonists: American College of Chest Physicians evidence‐based clinical practice guidelines (8th ed.).Chest.2008;133:160S–198S.
- The Joint Commission. 2009 National Patient Safety Goals. Available at: http://www.jointcommission.org/NR/rdonlyres/31666E86‐E7F4–423E‐9BE8‐F05BD1CB0AA8/0/HAP_NPSG.pdf. Accessed May 6,2010.
- ,,, et al.Optimization of inpatient warfarin therapy: impact of a daily consultation by a pharmacist‐managed anticoagulation service.Ann Pharmacother.2000;34:567–572.
- ,,, et al.Pharmacy‐managed protocol for warfarin use in orthopedic surgery patients.Am J Health‐Syst Pharm.1995;52:1310–1316.
- .Pharmacist involvement with warfarin dosing for inpatients.Pharm World Sci.2001;23:31–35.
- ,,.Evaluation of a pharmacy‐managed warfarin‐monitoring service to coordinate inpatient and outpatient therapy.Am J Hosp Pharm.1992;49:387–394.
- ,.Implementation and evaluation of a pharmacist‐assisted warfarin dosing program.Can J Hosp Pharm.1997;50:169–175.
- ,.Definition of major bleeding in clinical investigations of antihemostatic medicinal products in non‐surgical patients.J Thromb Haemostasis.2005;3:692–694.
- ,,.Rehospitalizations among patients in the Medicare fee‐for‐service program.N Engl J Med.2009;360:1418–1428.
Copyright © 2011 Society of Hospital Medicine