User login
Readmission after esophagectomy raises mortality
About one in five patients who have surgery to remove part or all of the esophagus return to the hospital for complications within 30 days, and when they do their chance of death increases fivefold, compared with those who don’t return to the hospital, investigators at the University of Virginia Health System reported in the Journal of Thoracic and Cardiovascular Surgery (2015;150:1254-60).
“Early recognition of life-threatening readmission diagnoses is essential in order to provide optimal care,” said lead author Dr. Yinin Hu and colleagues. Esophageal cancer is the fastest-growing cancer in the United States, so the study investigators set out to closely examine the reasons for readmissions and death after surgery.
The study identified 1,688 patients in the Surveillance, Epidemiology, and End Results (SEER)-Medicare database aged 66 or older who had surgery for esophageal cancer from 2000 to 2009. The overall 30-day mortality was 6.9%, and the 90-day mortality was 13.9%.
After excluding in-hospital deaths, the investigators’ readmission analysis included 1,543 patients. In this group, overall 90-day mortality following discharge was 6.4%, and the readmission rate within 30 days of discharge was 20.7%
The 90-day mortality for patients who were readmitted was more than four times that for those who were not readmitted, 16.3% vs. 3.8%; their in-hospital mortality was 8.8%. About one-third of readmissions were to facilities different from where patients had the index esophagectomy, and those patients were about seven times more likely to be transferred after readmission than patients admitted to the same facility, 15% vs. 1.9%. Risk-adjusted mortality did not vary significantly across providers.
The most frequent reasons for readmission were pneumonia (11.8%), malnutrition/dehydration (8.1%), pleural effusion (97.5%), and aspiration pneumonitis (6.8%). “Notably, more than one in five patients readmitted with a pulmonary diagnosis subsequently died within 90 days of the operation,” Dr. Hu and coauthors said, indicating that readmissions for pulmonary complications carried the worst prognosis.
This is the first study to demonstrate the gravity of pulmonary readmissions within 30 days of discharge, Dr. Hu and coauthors said. “Patients with nonspecific dyspneic symptoms or small pleural effusions should receive aggressive care upon readmission, as more than 20% will not survive the next few months,” Dr. Hu and coauthors said. “These results reinforce the notion that a fairly benign readmitting diagnosis is often an indicator of a much more severe root process.”
Among nonpulmonary reasons for readmission, dehydration and malnutrition carried the highest risk for death. “While there are many interventions that can promote postoperative nutrition, a readmission due to poor dietary tolerance often indicates other complications such as infection, stenosis, or anastomotic leak,” Dr. Hu and coauthors said. They suggested a thorough root-cause analysis should be part of every readmission.
The study also analyzed the hospital length of stay (LOS) as a predictor for readmission. The median LOS was 13 days, but the most common LOS was 9 days. “In general, the probability of readmission increases with increasing postoperative LOS,” Dr. Hu and colleagues said.
The authors reported no disclosures. Dr. Yinin Hu received funding from the National Institutes of Health and coauthor Dr. Benjamin Kozower received funding from the Agency for Healthcare Research and Quality.
The findings of this study may indicate that patients who stay in the hospital longer have underlying issues that did not surface during their admission for the operation, Dr. Anthony W. Kim of Yale University, New Haven, Conn., said in his invited commentary (J Thorac Cardiovasc Surg. 2015;150:1030-1). “Therefore, rather than employing a prevention of a failure-to-rescue strategy during a readmission, it may be worthwhile to enact a prevention-of-readmission strategy triggered by a longer than typical [length of stay],” Dr. Kim said. He suggested the root-cause analysis should begin in the hospital on the day after the patient exceeds the median length of stay.

|
Dr. Anthony W. Kim |
But citing the law of unintended consequences, Dr. Kim warns against using readmission as a quality metric as well a instrument to dictate reimbursement. “The law of unintended consequences dictates that when a readmission is taken out of context, using this measure raises the potential conflict of interest between doing what is right for the patient and achieving a specific milestone that may not be in the best interest of an individual patient,” he said. “Discharging a patient early perhaps to the exclusion of adequately addressing inpatient issues for the purposes of achieving a target [length of stay] is perhaps the prime example of these conflicting interests.”
Because of the difficult recovery course after esophagectomy, some readmissions are “necessary, beneficial and, unequivocally, the right decision,” Dr. Kim said. “Ironically, one of the unintended consequences of this article may be that it exposes the fact that until a better system of recording and scrutinizing readmissions exists, governing organizations should exercise considerable caution when assessing a surgeon, hospital, or system and their readmissions,” he said.
To paraphrase the sociologist Robert K. Merton, who devised the law of unintended consequences, the existing state of knowledge limits one’s ability to anticipate the consequences of action. The authors of this study “have added immensely to a body of knowledge that is still growing and deserves ongoing study if policy is to be based upon it,” Dr. Kim said.
Dr. Kim had no disclosures.
The findings of this study may indicate that patients who stay in the hospital longer have underlying issues that did not surface during their admission for the operation, Dr. Anthony W. Kim of Yale University, New Haven, Conn., said in his invited commentary (J Thorac Cardiovasc Surg. 2015;150:1030-1). “Therefore, rather than employing a prevention of a failure-to-rescue strategy during a readmission, it may be worthwhile to enact a prevention-of-readmission strategy triggered by a longer than typical [length of stay],” Dr. Kim said. He suggested the root-cause analysis should begin in the hospital on the day after the patient exceeds the median length of stay.

|
Dr. Anthony W. Kim |
But citing the law of unintended consequences, Dr. Kim warns against using readmission as a quality metric as well a instrument to dictate reimbursement. “The law of unintended consequences dictates that when a readmission is taken out of context, using this measure raises the potential conflict of interest between doing what is right for the patient and achieving a specific milestone that may not be in the best interest of an individual patient,” he said. “Discharging a patient early perhaps to the exclusion of adequately addressing inpatient issues for the purposes of achieving a target [length of stay] is perhaps the prime example of these conflicting interests.”
Because of the difficult recovery course after esophagectomy, some readmissions are “necessary, beneficial and, unequivocally, the right decision,” Dr. Kim said. “Ironically, one of the unintended consequences of this article may be that it exposes the fact that until a better system of recording and scrutinizing readmissions exists, governing organizations should exercise considerable caution when assessing a surgeon, hospital, or system and their readmissions,” he said.
To paraphrase the sociologist Robert K. Merton, who devised the law of unintended consequences, the existing state of knowledge limits one’s ability to anticipate the consequences of action. The authors of this study “have added immensely to a body of knowledge that is still growing and deserves ongoing study if policy is to be based upon it,” Dr. Kim said.
Dr. Kim had no disclosures.
The findings of this study may indicate that patients who stay in the hospital longer have underlying issues that did not surface during their admission for the operation, Dr. Anthony W. Kim of Yale University, New Haven, Conn., said in his invited commentary (J Thorac Cardiovasc Surg. 2015;150:1030-1). “Therefore, rather than employing a prevention of a failure-to-rescue strategy during a readmission, it may be worthwhile to enact a prevention-of-readmission strategy triggered by a longer than typical [length of stay],” Dr. Kim said. He suggested the root-cause analysis should begin in the hospital on the day after the patient exceeds the median length of stay.

|
Dr. Anthony W. Kim |
But citing the law of unintended consequences, Dr. Kim warns against using readmission as a quality metric as well a instrument to dictate reimbursement. “The law of unintended consequences dictates that when a readmission is taken out of context, using this measure raises the potential conflict of interest between doing what is right for the patient and achieving a specific milestone that may not be in the best interest of an individual patient,” he said. “Discharging a patient early perhaps to the exclusion of adequately addressing inpatient issues for the purposes of achieving a target [length of stay] is perhaps the prime example of these conflicting interests.”
Because of the difficult recovery course after esophagectomy, some readmissions are “necessary, beneficial and, unequivocally, the right decision,” Dr. Kim said. “Ironically, one of the unintended consequences of this article may be that it exposes the fact that until a better system of recording and scrutinizing readmissions exists, governing organizations should exercise considerable caution when assessing a surgeon, hospital, or system and their readmissions,” he said.
To paraphrase the sociologist Robert K. Merton, who devised the law of unintended consequences, the existing state of knowledge limits one’s ability to anticipate the consequences of action. The authors of this study “have added immensely to a body of knowledge that is still growing and deserves ongoing study if policy is to be based upon it,” Dr. Kim said.
Dr. Kim had no disclosures.
About one in five patients who have surgery to remove part or all of the esophagus return to the hospital for complications within 30 days, and when they do their chance of death increases fivefold, compared with those who don’t return to the hospital, investigators at the University of Virginia Health System reported in the Journal of Thoracic and Cardiovascular Surgery (2015;150:1254-60).
“Early recognition of life-threatening readmission diagnoses is essential in order to provide optimal care,” said lead author Dr. Yinin Hu and colleagues. Esophageal cancer is the fastest-growing cancer in the United States, so the study investigators set out to closely examine the reasons for readmissions and death after surgery.
The study identified 1,688 patients in the Surveillance, Epidemiology, and End Results (SEER)-Medicare database aged 66 or older who had surgery for esophageal cancer from 2000 to 2009. The overall 30-day mortality was 6.9%, and the 90-day mortality was 13.9%.
After excluding in-hospital deaths, the investigators’ readmission analysis included 1,543 patients. In this group, overall 90-day mortality following discharge was 6.4%, and the readmission rate within 30 days of discharge was 20.7%
The 90-day mortality for patients who were readmitted was more than four times that for those who were not readmitted, 16.3% vs. 3.8%; their in-hospital mortality was 8.8%. About one-third of readmissions were to facilities different from where patients had the index esophagectomy, and those patients were about seven times more likely to be transferred after readmission than patients admitted to the same facility, 15% vs. 1.9%. Risk-adjusted mortality did not vary significantly across providers.
The most frequent reasons for readmission were pneumonia (11.8%), malnutrition/dehydration (8.1%), pleural effusion (97.5%), and aspiration pneumonitis (6.8%). “Notably, more than one in five patients readmitted with a pulmonary diagnosis subsequently died within 90 days of the operation,” Dr. Hu and coauthors said, indicating that readmissions for pulmonary complications carried the worst prognosis.
This is the first study to demonstrate the gravity of pulmonary readmissions within 30 days of discharge, Dr. Hu and coauthors said. “Patients with nonspecific dyspneic symptoms or small pleural effusions should receive aggressive care upon readmission, as more than 20% will not survive the next few months,” Dr. Hu and coauthors said. “These results reinforce the notion that a fairly benign readmitting diagnosis is often an indicator of a much more severe root process.”
Among nonpulmonary reasons for readmission, dehydration and malnutrition carried the highest risk for death. “While there are many interventions that can promote postoperative nutrition, a readmission due to poor dietary tolerance often indicates other complications such as infection, stenosis, or anastomotic leak,” Dr. Hu and coauthors said. They suggested a thorough root-cause analysis should be part of every readmission.
The study also analyzed the hospital length of stay (LOS) as a predictor for readmission. The median LOS was 13 days, but the most common LOS was 9 days. “In general, the probability of readmission increases with increasing postoperative LOS,” Dr. Hu and colleagues said.
The authors reported no disclosures. Dr. Yinin Hu received funding from the National Institutes of Health and coauthor Dr. Benjamin Kozower received funding from the Agency for Healthcare Research and Quality.
About one in five patients who have surgery to remove part or all of the esophagus return to the hospital for complications within 30 days, and when they do their chance of death increases fivefold, compared with those who don’t return to the hospital, investigators at the University of Virginia Health System reported in the Journal of Thoracic and Cardiovascular Surgery (2015;150:1254-60).
“Early recognition of life-threatening readmission diagnoses is essential in order to provide optimal care,” said lead author Dr. Yinin Hu and colleagues. Esophageal cancer is the fastest-growing cancer in the United States, so the study investigators set out to closely examine the reasons for readmissions and death after surgery.
The study identified 1,688 patients in the Surveillance, Epidemiology, and End Results (SEER)-Medicare database aged 66 or older who had surgery for esophageal cancer from 2000 to 2009. The overall 30-day mortality was 6.9%, and the 90-day mortality was 13.9%.
After excluding in-hospital deaths, the investigators’ readmission analysis included 1,543 patients. In this group, overall 90-day mortality following discharge was 6.4%, and the readmission rate within 30 days of discharge was 20.7%
The 90-day mortality for patients who were readmitted was more than four times that for those who were not readmitted, 16.3% vs. 3.8%; their in-hospital mortality was 8.8%. About one-third of readmissions were to facilities different from where patients had the index esophagectomy, and those patients were about seven times more likely to be transferred after readmission than patients admitted to the same facility, 15% vs. 1.9%. Risk-adjusted mortality did not vary significantly across providers.
The most frequent reasons for readmission were pneumonia (11.8%), malnutrition/dehydration (8.1%), pleural effusion (97.5%), and aspiration pneumonitis (6.8%). “Notably, more than one in five patients readmitted with a pulmonary diagnosis subsequently died within 90 days of the operation,” Dr. Hu and coauthors said, indicating that readmissions for pulmonary complications carried the worst prognosis.
This is the first study to demonstrate the gravity of pulmonary readmissions within 30 days of discharge, Dr. Hu and coauthors said. “Patients with nonspecific dyspneic symptoms or small pleural effusions should receive aggressive care upon readmission, as more than 20% will not survive the next few months,” Dr. Hu and coauthors said. “These results reinforce the notion that a fairly benign readmitting diagnosis is often an indicator of a much more severe root process.”
Among nonpulmonary reasons for readmission, dehydration and malnutrition carried the highest risk for death. “While there are many interventions that can promote postoperative nutrition, a readmission due to poor dietary tolerance often indicates other complications such as infection, stenosis, or anastomotic leak,” Dr. Hu and coauthors said. They suggested a thorough root-cause analysis should be part of every readmission.
The study also analyzed the hospital length of stay (LOS) as a predictor for readmission. The median LOS was 13 days, but the most common LOS was 9 days. “In general, the probability of readmission increases with increasing postoperative LOS,” Dr. Hu and colleagues said.
The authors reported no disclosures. Dr. Yinin Hu received funding from the National Institutes of Health and coauthor Dr. Benjamin Kozower received funding from the Agency for Healthcare Research and Quality.
Key clinical point: Patients readmitted after esophagectomy are at a greater than fourfold higher risk of death than patients who do not need readmission.
Major finding: The 90-day mortality for patients who were readmitted was greater than four times that for those who were not readmitted, 16.3% vs. 3.8%.
Data source: Analysis of 1,688 patients in the SEER-Medicare database aged 66 or older who had surgery for esophageal cancer from 2000 to 2009.
Disclosures: The authors had no disclosures. Lead author Dr. Yinin Hu received funding from the National Institutes of Health and coauthor Dr. Benjamin Kozower received funding from the Agency for Healthcare Research and Quality.
Does position matter in ViV implantation?
With transcatheter valve-in-valve implantation emerging as a novel treatment for high-risk patients whose existing bioprostheses have deteriorated, a team of investigators at University Heart Center in Hamburg, Germany, has found that the procedure can be done successfully in four different anatomic positions with a variety of bioprostheses.
The findings from the single-center study were published in the December issue of the Journal of Thoracic and Cardiovascular Surgery. (J Thorac Cardiovasc Surg. 2015;150:1557-67). They retrospectively analyzed results of 75 patients who had transcatheter valve-in-valve (ViV) replacement at their institution from 2008 to 2014.
“ViV can be performed in all anatomic positions with acceptable hemodynamic and clinical outcome in high-risk patients,” wrote Dr. Lenard Conradi and coauthors. “Increasing importance of ViV can be anticipated considering growing use of surgical bioprostheses.”
Replacement of biological valves is becoming more common. For surgical aortic valve replacement (SAVR), biological procedures have largely replaced mechanical valve implantation, comprising 87% of all such procedures by 2014, according to data from the German Society for Thoracic and Cardiovascular Surgery (Thorac Cardiovasc Surg. 2014;62:380-92). “Therefore, increasing caseload of patients with deteriorated bioprostheses can be expected,” wrote Dr. Conradi and coauthors.
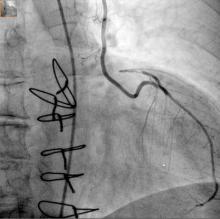
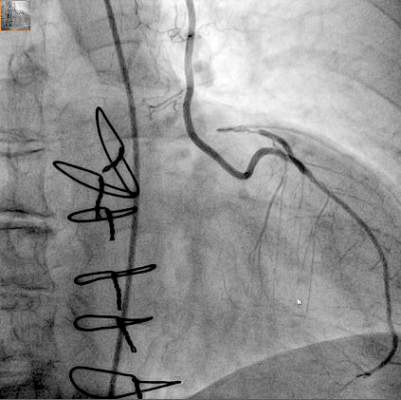
The four anatomic positions in which the investigators performed the procedures and their share of cases are: aortic (54 patients/72%), mitral (17/22.7%), and tricuspid and pulmonary positions (2/2.7% each). The average interval between the index procedure and ViV was 9 years, with a deviation of nearly 5 years among all procedures. Dr. Conradi and coinvestigators said their study focused on technical aspects of ViV procedures from each position to provide guidance for surgeons.
Overall, the study authors performed ViV successfully in 97.3% of patients, with two patients requiring sequential transcatheter heart valve implantation for initial malpositioning. Thirty-day mortality was 8%, which “ranged lower” than expected when compared to standard preoperative risk stratification, they wrote. Mortality was at 5.6% in the aortic group and 17.6% in the mitral group.
That none of the currently available surgical bioprostheses or transcatheter heart valves (THV) were designed for later ViV procedures in deteriorated bioprostheses – although the CoreValve and Sapein THV have approvals for the indication – “may explain some of the apparent shortcomings of ViV therapy,” the researchers wrote.
The most significant challenge of ViV therapy is dealing with elevated residual gradients, which positioning can influence, according to the study findings. “This is not so much an issue for mitral, tricuspid, or pulmonary positions since surgical bioprostheses implanted in these positions are usually of sufficient size to accommodate the THV,” the researchers noted. “However, in the aortic position, more severe spatial restrictions may apply.”
They cited other reports that described a reverse relationship between size of the bioprosthetic and resulting transvalvular gradient after ViV (JACC Cardiovasc Interv. 2011;4:1218-27; JAMA 2014;312:162-70).
To reduce gradients, the investigators used post-ballooning after aortic ViV with a self-expandable THV in 16 cases, succeeding in 12. “Likely, further THV expansion with active compression of soft leaflet and/or pannus tissue and tighter apposition of THV against the frames of surgical bioprostheses contributed to this desired effect,” wrote the researchers. Patient-prosthesis mismatch probably explained the four cases in which gradients could not be further reduced, they noted.
They issued one “word of caution” regarding aortic ViV in small-sized surgical bioprostheses: “Elevated postprocedural gradients have to be expected and must be weighed against expected benefits and against risk of repeat open heart surgery.”
The six transcatheter heart valves the investigators used were Edwards Sapien (XT)/Sapien3 (52%, 39/75); Medtronic CoreValve/CoreValveEvolut (34.7%, 26); St. Jude Portico and Boston Scientific Lotus (4%, three each); and JenaValve and Medtronic Engager (2.7%, two each). The study also looked at different access routes: transapical in 53.3% (40), transfemoral (transarterial or transvenous) in 42.7% (32), transaortic in 2.7% (2), and transjugular in 1.3% (1).
Dr. Conradi and coauthors Dr. Moritz Seiffert, Dr. Ulrich Schaefer, and Dr. Hendrik Treede disclosed ties with Edwards Lifesciences, JenaValve Technology, Medtronic, Symetis, and St. Jude Medical. Four other coauthors reported no disclosures.
As the population ages and younger patients choose bioprosthetic valves to avoid lifelong warfarin, surgeons are going to face more situations where they will have to decide whether to perform surgical or transcatheter reoperative valve surgery, Dr. Jessica Forcillo of Emory University, Atlanta, and coauthors wrote in an invited commentary (J Thorac Cardiovasc Surg. 2015;150:1568-9).
They called the 8% 30-day mortality rate in the Hamburg study “high” even though the average age of the study population was a “relatively young” 74 years. The Hamburg authors may have learned more had they evaluated fewer prostheses. “With a small number of patients and at the beginning of an experience, focusing on one or two available prostheses may have resulted in more accurate and reliable results,” noted Dr. Forcillo and her colleagues. That 53% of the procedures were done via the transapical approach may also explain the mortality rate, they said.
The overall 30-day mortality rate along with a 17.6% mortality in the mitral ViV group are causes for “some caution against overzealous performance of this procedure and continued monitoring of outcomes in other series,” wrote Dr. Forcillo and her colleagues.
But ViV implantation is a “transformative” technology, they said. “For the elderly, high-risk patients with [structural valve degeneration], transcatheter options may provide improved short-term outcomes,” they added. “The valve community eagerly awaits larger series with adjudicated outcomes of the transcatheter valve-in-valve procedure.”
Dr. Forcillo and coauthor Lillian Tsai had no disclosures. Dr. Vinod Thourani disclosed ties with St. Jude Medical, Edwards Lifesciences, Boston Scientific, Abbott Medical, Medtronic, Directflow, and Sorin Medical.
As the population ages and younger patients choose bioprosthetic valves to avoid lifelong warfarin, surgeons are going to face more situations where they will have to decide whether to perform surgical or transcatheter reoperative valve surgery, Dr. Jessica Forcillo of Emory University, Atlanta, and coauthors wrote in an invited commentary (J Thorac Cardiovasc Surg. 2015;150:1568-9).
They called the 8% 30-day mortality rate in the Hamburg study “high” even though the average age of the study population was a “relatively young” 74 years. The Hamburg authors may have learned more had they evaluated fewer prostheses. “With a small number of patients and at the beginning of an experience, focusing on one or two available prostheses may have resulted in more accurate and reliable results,” noted Dr. Forcillo and her colleagues. That 53% of the procedures were done via the transapical approach may also explain the mortality rate, they said.
The overall 30-day mortality rate along with a 17.6% mortality in the mitral ViV group are causes for “some caution against overzealous performance of this procedure and continued monitoring of outcomes in other series,” wrote Dr. Forcillo and her colleagues.
But ViV implantation is a “transformative” technology, they said. “For the elderly, high-risk patients with [structural valve degeneration], transcatheter options may provide improved short-term outcomes,” they added. “The valve community eagerly awaits larger series with adjudicated outcomes of the transcatheter valve-in-valve procedure.”
Dr. Forcillo and coauthor Lillian Tsai had no disclosures. Dr. Vinod Thourani disclosed ties with St. Jude Medical, Edwards Lifesciences, Boston Scientific, Abbott Medical, Medtronic, Directflow, and Sorin Medical.
As the population ages and younger patients choose bioprosthetic valves to avoid lifelong warfarin, surgeons are going to face more situations where they will have to decide whether to perform surgical or transcatheter reoperative valve surgery, Dr. Jessica Forcillo of Emory University, Atlanta, and coauthors wrote in an invited commentary (J Thorac Cardiovasc Surg. 2015;150:1568-9).
They called the 8% 30-day mortality rate in the Hamburg study “high” even though the average age of the study population was a “relatively young” 74 years. The Hamburg authors may have learned more had they evaluated fewer prostheses. “With a small number of patients and at the beginning of an experience, focusing on one or two available prostheses may have resulted in more accurate and reliable results,” noted Dr. Forcillo and her colleagues. That 53% of the procedures were done via the transapical approach may also explain the mortality rate, they said.
The overall 30-day mortality rate along with a 17.6% mortality in the mitral ViV group are causes for “some caution against overzealous performance of this procedure and continued monitoring of outcomes in other series,” wrote Dr. Forcillo and her colleagues.
But ViV implantation is a “transformative” technology, they said. “For the elderly, high-risk patients with [structural valve degeneration], transcatheter options may provide improved short-term outcomes,” they added. “The valve community eagerly awaits larger series with adjudicated outcomes of the transcatheter valve-in-valve procedure.”
Dr. Forcillo and coauthor Lillian Tsai had no disclosures. Dr. Vinod Thourani disclosed ties with St. Jude Medical, Edwards Lifesciences, Boston Scientific, Abbott Medical, Medtronic, Directflow, and Sorin Medical.
With transcatheter valve-in-valve implantation emerging as a novel treatment for high-risk patients whose existing bioprostheses have deteriorated, a team of investigators at University Heart Center in Hamburg, Germany, has found that the procedure can be done successfully in four different anatomic positions with a variety of bioprostheses.
The findings from the single-center study were published in the December issue of the Journal of Thoracic and Cardiovascular Surgery. (J Thorac Cardiovasc Surg. 2015;150:1557-67). They retrospectively analyzed results of 75 patients who had transcatheter valve-in-valve (ViV) replacement at their institution from 2008 to 2014.
“ViV can be performed in all anatomic positions with acceptable hemodynamic and clinical outcome in high-risk patients,” wrote Dr. Lenard Conradi and coauthors. “Increasing importance of ViV can be anticipated considering growing use of surgical bioprostheses.”
Replacement of biological valves is becoming more common. For surgical aortic valve replacement (SAVR), biological procedures have largely replaced mechanical valve implantation, comprising 87% of all such procedures by 2014, according to data from the German Society for Thoracic and Cardiovascular Surgery (Thorac Cardiovasc Surg. 2014;62:380-92). “Therefore, increasing caseload of patients with deteriorated bioprostheses can be expected,” wrote Dr. Conradi and coauthors.
The four anatomic positions in which the investigators performed the procedures and their share of cases are: aortic (54 patients/72%), mitral (17/22.7%), and tricuspid and pulmonary positions (2/2.7% each). The average interval between the index procedure and ViV was 9 years, with a deviation of nearly 5 years among all procedures. Dr. Conradi and coinvestigators said their study focused on technical aspects of ViV procedures from each position to provide guidance for surgeons.
Overall, the study authors performed ViV successfully in 97.3% of patients, with two patients requiring sequential transcatheter heart valve implantation for initial malpositioning. Thirty-day mortality was 8%, which “ranged lower” than expected when compared to standard preoperative risk stratification, they wrote. Mortality was at 5.6% in the aortic group and 17.6% in the mitral group.
That none of the currently available surgical bioprostheses or transcatheter heart valves (THV) were designed for later ViV procedures in deteriorated bioprostheses – although the CoreValve and Sapein THV have approvals for the indication – “may explain some of the apparent shortcomings of ViV therapy,” the researchers wrote.
The most significant challenge of ViV therapy is dealing with elevated residual gradients, which positioning can influence, according to the study findings. “This is not so much an issue for mitral, tricuspid, or pulmonary positions since surgical bioprostheses implanted in these positions are usually of sufficient size to accommodate the THV,” the researchers noted. “However, in the aortic position, more severe spatial restrictions may apply.”
They cited other reports that described a reverse relationship between size of the bioprosthetic and resulting transvalvular gradient after ViV (JACC Cardiovasc Interv. 2011;4:1218-27; JAMA 2014;312:162-70).
To reduce gradients, the investigators used post-ballooning after aortic ViV with a self-expandable THV in 16 cases, succeeding in 12. “Likely, further THV expansion with active compression of soft leaflet and/or pannus tissue and tighter apposition of THV against the frames of surgical bioprostheses contributed to this desired effect,” wrote the researchers. Patient-prosthesis mismatch probably explained the four cases in which gradients could not be further reduced, they noted.
They issued one “word of caution” regarding aortic ViV in small-sized surgical bioprostheses: “Elevated postprocedural gradients have to be expected and must be weighed against expected benefits and against risk of repeat open heart surgery.”
The six transcatheter heart valves the investigators used were Edwards Sapien (XT)/Sapien3 (52%, 39/75); Medtronic CoreValve/CoreValveEvolut (34.7%, 26); St. Jude Portico and Boston Scientific Lotus (4%, three each); and JenaValve and Medtronic Engager (2.7%, two each). The study also looked at different access routes: transapical in 53.3% (40), transfemoral (transarterial or transvenous) in 42.7% (32), transaortic in 2.7% (2), and transjugular in 1.3% (1).
Dr. Conradi and coauthors Dr. Moritz Seiffert, Dr. Ulrich Schaefer, and Dr. Hendrik Treede disclosed ties with Edwards Lifesciences, JenaValve Technology, Medtronic, Symetis, and St. Jude Medical. Four other coauthors reported no disclosures.
With transcatheter valve-in-valve implantation emerging as a novel treatment for high-risk patients whose existing bioprostheses have deteriorated, a team of investigators at University Heart Center in Hamburg, Germany, has found that the procedure can be done successfully in four different anatomic positions with a variety of bioprostheses.
The findings from the single-center study were published in the December issue of the Journal of Thoracic and Cardiovascular Surgery. (J Thorac Cardiovasc Surg. 2015;150:1557-67). They retrospectively analyzed results of 75 patients who had transcatheter valve-in-valve (ViV) replacement at their institution from 2008 to 2014.
“ViV can be performed in all anatomic positions with acceptable hemodynamic and clinical outcome in high-risk patients,” wrote Dr. Lenard Conradi and coauthors. “Increasing importance of ViV can be anticipated considering growing use of surgical bioprostheses.”
Replacement of biological valves is becoming more common. For surgical aortic valve replacement (SAVR), biological procedures have largely replaced mechanical valve implantation, comprising 87% of all such procedures by 2014, according to data from the German Society for Thoracic and Cardiovascular Surgery (Thorac Cardiovasc Surg. 2014;62:380-92). “Therefore, increasing caseload of patients with deteriorated bioprostheses can be expected,” wrote Dr. Conradi and coauthors.
The four anatomic positions in which the investigators performed the procedures and their share of cases are: aortic (54 patients/72%), mitral (17/22.7%), and tricuspid and pulmonary positions (2/2.7% each). The average interval between the index procedure and ViV was 9 years, with a deviation of nearly 5 years among all procedures. Dr. Conradi and coinvestigators said their study focused on technical aspects of ViV procedures from each position to provide guidance for surgeons.
Overall, the study authors performed ViV successfully in 97.3% of patients, with two patients requiring sequential transcatheter heart valve implantation for initial malpositioning. Thirty-day mortality was 8%, which “ranged lower” than expected when compared to standard preoperative risk stratification, they wrote. Mortality was at 5.6% in the aortic group and 17.6% in the mitral group.
That none of the currently available surgical bioprostheses or transcatheter heart valves (THV) were designed for later ViV procedures in deteriorated bioprostheses – although the CoreValve and Sapein THV have approvals for the indication – “may explain some of the apparent shortcomings of ViV therapy,” the researchers wrote.
The most significant challenge of ViV therapy is dealing with elevated residual gradients, which positioning can influence, according to the study findings. “This is not so much an issue for mitral, tricuspid, or pulmonary positions since surgical bioprostheses implanted in these positions are usually of sufficient size to accommodate the THV,” the researchers noted. “However, in the aortic position, more severe spatial restrictions may apply.”
They cited other reports that described a reverse relationship between size of the bioprosthetic and resulting transvalvular gradient after ViV (JACC Cardiovasc Interv. 2011;4:1218-27; JAMA 2014;312:162-70).
To reduce gradients, the investigators used post-ballooning after aortic ViV with a self-expandable THV in 16 cases, succeeding in 12. “Likely, further THV expansion with active compression of soft leaflet and/or pannus tissue and tighter apposition of THV against the frames of surgical bioprostheses contributed to this desired effect,” wrote the researchers. Patient-prosthesis mismatch probably explained the four cases in which gradients could not be further reduced, they noted.
They issued one “word of caution” regarding aortic ViV in small-sized surgical bioprostheses: “Elevated postprocedural gradients have to be expected and must be weighed against expected benefits and against risk of repeat open heart surgery.”
The six transcatheter heart valves the investigators used were Edwards Sapien (XT)/Sapien3 (52%, 39/75); Medtronic CoreValve/CoreValveEvolut (34.7%, 26); St. Jude Portico and Boston Scientific Lotus (4%, three each); and JenaValve and Medtronic Engager (2.7%, two each). The study also looked at different access routes: transapical in 53.3% (40), transfemoral (transarterial or transvenous) in 42.7% (32), transaortic in 2.7% (2), and transjugular in 1.3% (1).
Dr. Conradi and coauthors Dr. Moritz Seiffert, Dr. Ulrich Schaefer, and Dr. Hendrik Treede disclosed ties with Edwards Lifesciences, JenaValve Technology, Medtronic, Symetis, and St. Jude Medical. Four other coauthors reported no disclosures.
Key clinical point: Transcatheter valve-in-valve (ViV) implantation is a relatively safe treatment for patients with a deteriorated bioprostheses.
Major finding: A ViV implantation when performed in four different positions with six different transcatheter heart valves had a 30-day mortality of 8%.
Data source: Retrospective analysis of 75 consecutive patients receiving ViV procedures from 2008 to 2014 at a single institution.
Disclosures: Dr. Conradi and coauthors Dr. Moritz Seiffert, Dr. Ulrich Schaefer, and Dr. Hendrik Treede disclosed relationships with Edwards Lifesciences, JenaValve Technology, Medtronic, Symetis, and St. Jude Medical. Four other coauthors reported no disclosures.
Surgical ablation endures at 5 years
The Cox-Maze IV procedure (CMPIV) has become the standard for surgical ablation for atrial fibrillation (AF), yet little information has been available on how late outcomes compare with catheter-based ablation. A recent analysis of 576 procedures found that after 5 years, most people who had the procedure remained free of atrial tachyarrhythmias and anticoagulation.
The study, by investigators from Washington University, Barnes-Jewish Hospital in St. Louis, was published in the Journal of Thoracic and Cardiovascular Surgery (J Thorac Cardiovasc Surg. 2015;150:1168-78). The researchers first presented the study in April at the American Association for Thoracic Surgery meeting in Seattle.
“The results of the CMPIV remain superior to those reported for catheter ablation and other forms of surgical AF ablation, especially for patients with persistent or long-standing AF,” wrote Dr. Matthew C. Henn and his colleagues.
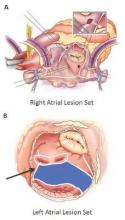
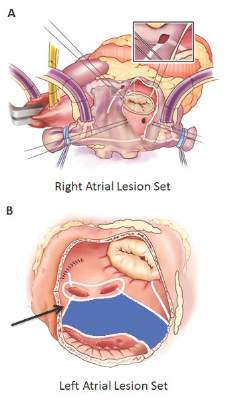
They set out to evaluate late outcomes after CMPIV using current consensus definitions of treatment failure, noting that such outcomes had yet to be reported. They followed 576 patients with atrial fibrillation who had a CMPIV from 2002 to 2014 and compared long-term freedom from atrial fibrillation on and off antiarrhythmic drugs (AADs) across various subgroups. They included the left-sided CMPIV lesion in the analysis because, they said, it had success rates similar to those of biatrial CMPIV.
The Cox-Maze procedure was first introduced by Dr. James Cox in 1987 and updated from the original “cut-and-sew” technique in 2002 to combine bipolar radiofrequency and cryothermal ablation lines in place of most surgical incisions. This iteration was called the Cox-Maze IV procedure. In 2005, CMPIV was modified to include a superior connecting lesion, which formed a “box lesion” by completely isolating the entire posterior left atrium. The study included 512 people who underwent the “box lesion” set procedure.
“The modifications of the CMPIV have allowed it to be performed through a right minithoracotomy (RMT) approach, which has further reduced major morbidity, mortality, and hospital stay compared to those who underwent sternotomy while enjoying equivalent outcomes with regards to freedom from AF,” wrote Dr. Henn and his coauthors.
In the entire cohort, the overall freedom from atrial tachyarrhythmias (ATAs) and anticoagulation were 92% at 1 year, 88% at 2 years, 87% at 3 years, 81% at 4 years, and 73% at 5 years. Overall freedom from ATAs off antiarrhythmic drugs for the entire cohort ranged from 81% at 1 year to 61% at 5 years, and freedom from anticoagulation ranged from 65% at 1 year to 55% at 5 years.
“Freedoms from ATAs on or off AADs were significantly higher in those who underwent box lesion sets when compared to those who did not at 5 years,” noted Dr. Henn and his coauthors. Among the box lesion set group, 78% of those on AADs remained free of ATAs vs. 45% in the non–box lesion set group, and for those off AADs, 66% had no ATAs at 5 years while 33% of the non–box lesion set group did.
Of the overall study population, 41% had paroxysmal AF and 58% had nonparoxysmal AF. Among the latter group, 20% had persistent and 80% had long-standing persistent AF. The nonparoxysmal AF group had a longer duration of preoperative AF, larger left atria and more failed catheter ablations, Dr. Henn and coauthors reported. But, the study showed no differences in freedom from atrial fibrillation on or off AADs at 5 years between patients with paroxysmal AF or persistent/long-standing persistent AF, or between those who underwent stand-alone procedure and those who received a concomitant Cox-Maze procedure. Among those who had a concomitant procedure, 50% had a concomitant mitral valve procedure and 23% had coronary artery bypass grafting.
“The CMPIV results in our series were better than what has been achieved with catheter ablation,” the researchers wrote. They cited studies that showed arrhythmia-free survival after a single ablation procedure ranging from 17% to 29% and “equally poor results.” (Circ Arrhythm Electrophysiol. 2015;8:18-24; J Am Coll Cardiol. 2011;57:160-166; J Am Heart Assoc. 2013;2:e004549.)
“The CMPIV remains the most successful surgical treatment for AF, even in patients with non-paroxysmal AF and regardless of the complexity of the concomitant procedures,” Dr. Henn and his coauthors concluded.
Inconsistencies in this study of the Cox-Maze IV procedure include differing types of atrial fibrillation, heterogeneous concomitant operations, multiple lesion sets and energy sources and inconsistent postablation monitoring, all of which make direct comparisons of surgical ablation strategies or even catheter ablation difficult, Dr. Robert Hawkins and Dr. Gorav Ailawadi of the University of Virginia noted in their invited commentary (J Thorac Cardiovasc Surg. 2015;150:1179-80). “Moreover, without controls or selection criteria, it is difficult to account for selection bias,” they wrote.
Yet, this study has “some important findings” despite its shortcomings, namely the “respectable” rates of atrial tachyarrhythmias off antiarrhythmic drugs. These results are superior to other clinical trials, “in part due to the expertise at Washington University,” noted Dr. Hawkins and Dr. Ailawadi.
Adding patients who had the box lesion set approach also improved 5-year outcomes in the study substantially, and left atrium (LA) ablation alone has good results in patients with paroxysmal AF, left atria less than 5.0 cm, and no right atrial enlargement. “Yet, a direct comparison between biatrial and LA lesion sets cannot be made due to the above listed limitations,” they wrote.
The study makes a case for surgical ablation when the preoperative duration of AF is less than 5-10 years and left atrium size is not a problem, and the lesion-set requires further investigation, they said. “Finally, this study highlights the continued need for rigorous monitoring and comparisons of homogeneous patient populations to make stronger conclusions.”
Dr. Ailawadi disclosed relationships with Abbot Vascular, Mitralign, Edwards Lifesciences and St. Jude Medical. Dr. Hawkins had no relationships to disclose.
Inconsistencies in this study of the Cox-Maze IV procedure include differing types of atrial fibrillation, heterogeneous concomitant operations, multiple lesion sets and energy sources and inconsistent postablation monitoring, all of which make direct comparisons of surgical ablation strategies or even catheter ablation difficult, Dr. Robert Hawkins and Dr. Gorav Ailawadi of the University of Virginia noted in their invited commentary (J Thorac Cardiovasc Surg. 2015;150:1179-80). “Moreover, without controls or selection criteria, it is difficult to account for selection bias,” they wrote.
Yet, this study has “some important findings” despite its shortcomings, namely the “respectable” rates of atrial tachyarrhythmias off antiarrhythmic drugs. These results are superior to other clinical trials, “in part due to the expertise at Washington University,” noted Dr. Hawkins and Dr. Ailawadi.
Adding patients who had the box lesion set approach also improved 5-year outcomes in the study substantially, and left atrium (LA) ablation alone has good results in patients with paroxysmal AF, left atria less than 5.0 cm, and no right atrial enlargement. “Yet, a direct comparison between biatrial and LA lesion sets cannot be made due to the above listed limitations,” they wrote.
The study makes a case for surgical ablation when the preoperative duration of AF is less than 5-10 years and left atrium size is not a problem, and the lesion-set requires further investigation, they said. “Finally, this study highlights the continued need for rigorous monitoring and comparisons of homogeneous patient populations to make stronger conclusions.”
Dr. Ailawadi disclosed relationships with Abbot Vascular, Mitralign, Edwards Lifesciences and St. Jude Medical. Dr. Hawkins had no relationships to disclose.
Inconsistencies in this study of the Cox-Maze IV procedure include differing types of atrial fibrillation, heterogeneous concomitant operations, multiple lesion sets and energy sources and inconsistent postablation monitoring, all of which make direct comparisons of surgical ablation strategies or even catheter ablation difficult, Dr. Robert Hawkins and Dr. Gorav Ailawadi of the University of Virginia noted in their invited commentary (J Thorac Cardiovasc Surg. 2015;150:1179-80). “Moreover, without controls or selection criteria, it is difficult to account for selection bias,” they wrote.
Yet, this study has “some important findings” despite its shortcomings, namely the “respectable” rates of atrial tachyarrhythmias off antiarrhythmic drugs. These results are superior to other clinical trials, “in part due to the expertise at Washington University,” noted Dr. Hawkins and Dr. Ailawadi.
Adding patients who had the box lesion set approach also improved 5-year outcomes in the study substantially, and left atrium (LA) ablation alone has good results in patients with paroxysmal AF, left atria less than 5.0 cm, and no right atrial enlargement. “Yet, a direct comparison between biatrial and LA lesion sets cannot be made due to the above listed limitations,” they wrote.
The study makes a case for surgical ablation when the preoperative duration of AF is less than 5-10 years and left atrium size is not a problem, and the lesion-set requires further investigation, they said. “Finally, this study highlights the continued need for rigorous monitoring and comparisons of homogeneous patient populations to make stronger conclusions.”
Dr. Ailawadi disclosed relationships with Abbot Vascular, Mitralign, Edwards Lifesciences and St. Jude Medical. Dr. Hawkins had no relationships to disclose.
The Cox-Maze IV procedure (CMPIV) has become the standard for surgical ablation for atrial fibrillation (AF), yet little information has been available on how late outcomes compare with catheter-based ablation. A recent analysis of 576 procedures found that after 5 years, most people who had the procedure remained free of atrial tachyarrhythmias and anticoagulation.
The study, by investigators from Washington University, Barnes-Jewish Hospital in St. Louis, was published in the Journal of Thoracic and Cardiovascular Surgery (J Thorac Cardiovasc Surg. 2015;150:1168-78). The researchers first presented the study in April at the American Association for Thoracic Surgery meeting in Seattle.
“The results of the CMPIV remain superior to those reported for catheter ablation and other forms of surgical AF ablation, especially for patients with persistent or long-standing AF,” wrote Dr. Matthew C. Henn and his colleagues.
They set out to evaluate late outcomes after CMPIV using current consensus definitions of treatment failure, noting that such outcomes had yet to be reported. They followed 576 patients with atrial fibrillation who had a CMPIV from 2002 to 2014 and compared long-term freedom from atrial fibrillation on and off antiarrhythmic drugs (AADs) across various subgroups. They included the left-sided CMPIV lesion in the analysis because, they said, it had success rates similar to those of biatrial CMPIV.
The Cox-Maze procedure was first introduced by Dr. James Cox in 1987 and updated from the original “cut-and-sew” technique in 2002 to combine bipolar radiofrequency and cryothermal ablation lines in place of most surgical incisions. This iteration was called the Cox-Maze IV procedure. In 2005, CMPIV was modified to include a superior connecting lesion, which formed a “box lesion” by completely isolating the entire posterior left atrium. The study included 512 people who underwent the “box lesion” set procedure.
“The modifications of the CMPIV have allowed it to be performed through a right minithoracotomy (RMT) approach, which has further reduced major morbidity, mortality, and hospital stay compared to those who underwent sternotomy while enjoying equivalent outcomes with regards to freedom from AF,” wrote Dr. Henn and his coauthors.
In the entire cohort, the overall freedom from atrial tachyarrhythmias (ATAs) and anticoagulation were 92% at 1 year, 88% at 2 years, 87% at 3 years, 81% at 4 years, and 73% at 5 years. Overall freedom from ATAs off antiarrhythmic drugs for the entire cohort ranged from 81% at 1 year to 61% at 5 years, and freedom from anticoagulation ranged from 65% at 1 year to 55% at 5 years.
“Freedoms from ATAs on or off AADs were significantly higher in those who underwent box lesion sets when compared to those who did not at 5 years,” noted Dr. Henn and his coauthors. Among the box lesion set group, 78% of those on AADs remained free of ATAs vs. 45% in the non–box lesion set group, and for those off AADs, 66% had no ATAs at 5 years while 33% of the non–box lesion set group did.
Of the overall study population, 41% had paroxysmal AF and 58% had nonparoxysmal AF. Among the latter group, 20% had persistent and 80% had long-standing persistent AF. The nonparoxysmal AF group had a longer duration of preoperative AF, larger left atria and more failed catheter ablations, Dr. Henn and coauthors reported. But, the study showed no differences in freedom from atrial fibrillation on or off AADs at 5 years between patients with paroxysmal AF or persistent/long-standing persistent AF, or between those who underwent stand-alone procedure and those who received a concomitant Cox-Maze procedure. Among those who had a concomitant procedure, 50% had a concomitant mitral valve procedure and 23% had coronary artery bypass grafting.
“The CMPIV results in our series were better than what has been achieved with catheter ablation,” the researchers wrote. They cited studies that showed arrhythmia-free survival after a single ablation procedure ranging from 17% to 29% and “equally poor results.” (Circ Arrhythm Electrophysiol. 2015;8:18-24; J Am Coll Cardiol. 2011;57:160-166; J Am Heart Assoc. 2013;2:e004549.)
“The CMPIV remains the most successful surgical treatment for AF, even in patients with non-paroxysmal AF and regardless of the complexity of the concomitant procedures,” Dr. Henn and his coauthors concluded.
The Cox-Maze IV procedure (CMPIV) has become the standard for surgical ablation for atrial fibrillation (AF), yet little information has been available on how late outcomes compare with catheter-based ablation. A recent analysis of 576 procedures found that after 5 years, most people who had the procedure remained free of atrial tachyarrhythmias and anticoagulation.
The study, by investigators from Washington University, Barnes-Jewish Hospital in St. Louis, was published in the Journal of Thoracic and Cardiovascular Surgery (J Thorac Cardiovasc Surg. 2015;150:1168-78). The researchers first presented the study in April at the American Association for Thoracic Surgery meeting in Seattle.
“The results of the CMPIV remain superior to those reported for catheter ablation and other forms of surgical AF ablation, especially for patients with persistent or long-standing AF,” wrote Dr. Matthew C. Henn and his colleagues.
They set out to evaluate late outcomes after CMPIV using current consensus definitions of treatment failure, noting that such outcomes had yet to be reported. They followed 576 patients with atrial fibrillation who had a CMPIV from 2002 to 2014 and compared long-term freedom from atrial fibrillation on and off antiarrhythmic drugs (AADs) across various subgroups. They included the left-sided CMPIV lesion in the analysis because, they said, it had success rates similar to those of biatrial CMPIV.
The Cox-Maze procedure was first introduced by Dr. James Cox in 1987 and updated from the original “cut-and-sew” technique in 2002 to combine bipolar radiofrequency and cryothermal ablation lines in place of most surgical incisions. This iteration was called the Cox-Maze IV procedure. In 2005, CMPIV was modified to include a superior connecting lesion, which formed a “box lesion” by completely isolating the entire posterior left atrium. The study included 512 people who underwent the “box lesion” set procedure.
“The modifications of the CMPIV have allowed it to be performed through a right minithoracotomy (RMT) approach, which has further reduced major morbidity, mortality, and hospital stay compared to those who underwent sternotomy while enjoying equivalent outcomes with regards to freedom from AF,” wrote Dr. Henn and his coauthors.
In the entire cohort, the overall freedom from atrial tachyarrhythmias (ATAs) and anticoagulation were 92% at 1 year, 88% at 2 years, 87% at 3 years, 81% at 4 years, and 73% at 5 years. Overall freedom from ATAs off antiarrhythmic drugs for the entire cohort ranged from 81% at 1 year to 61% at 5 years, and freedom from anticoagulation ranged from 65% at 1 year to 55% at 5 years.
“Freedoms from ATAs on or off AADs were significantly higher in those who underwent box lesion sets when compared to those who did not at 5 years,” noted Dr. Henn and his coauthors. Among the box lesion set group, 78% of those on AADs remained free of ATAs vs. 45% in the non–box lesion set group, and for those off AADs, 66% had no ATAs at 5 years while 33% of the non–box lesion set group did.
Of the overall study population, 41% had paroxysmal AF and 58% had nonparoxysmal AF. Among the latter group, 20% had persistent and 80% had long-standing persistent AF. The nonparoxysmal AF group had a longer duration of preoperative AF, larger left atria and more failed catheter ablations, Dr. Henn and coauthors reported. But, the study showed no differences in freedom from atrial fibrillation on or off AADs at 5 years between patients with paroxysmal AF or persistent/long-standing persistent AF, or between those who underwent stand-alone procedure and those who received a concomitant Cox-Maze procedure. Among those who had a concomitant procedure, 50% had a concomitant mitral valve procedure and 23% had coronary artery bypass grafting.
“The CMPIV results in our series were better than what has been achieved with catheter ablation,” the researchers wrote. They cited studies that showed arrhythmia-free survival after a single ablation procedure ranging from 17% to 29% and “equally poor results.” (Circ Arrhythm Electrophysiol. 2015;8:18-24; J Am Coll Cardiol. 2011;57:160-166; J Am Heart Assoc. 2013;2:e004549.)
“The CMPIV remains the most successful surgical treatment for AF, even in patients with non-paroxysmal AF and regardless of the complexity of the concomitant procedures,” Dr. Henn and his coauthors concluded.
Key clinical point: Outcomes with the Cox-Maze IV procedure for surgical ablation are superior to catheter ablation and other forms of surgical ablation for atrial fibrillation for up to 5 years duration.
Major finding: Seventy-three percent of the study population was free from atrial tachyarrhythmias and 55% were free from anticoagulation at 5 years.
Data source: Prospective analysis of 576 consecutive patients with atrial fibrillation who had Cox-Maze IV procedure or a left-sized Cox-Maze IV procedure from 2002 to 2014 at a single institution
Disclosures: The National Institutes of Health provided grants for the study. Coauthor Dr. Ralph J. Damiano Jr. disclosed research grants and educational funding from AtriCure and Edwards LifeSciences. The other authors had no disclosures.
Pediatric heart transplant results not improving
A 25-year study of heart transplants in children with congenital heart disease (CHD) at one institution has found that results haven’t improved over time despite advances in technology and techniques. To improve outcomes, transplant surgeons may need to do a better job of selecting patients and matching patients and donors, according to study in the December issue of the Journal of Thoracic and Cardiovascular Surgery (J Thorac Cardiovasc Surg. 2015;150:1455-62).
“Strategies to improve outcomes in CHD patients might need to address selection criteria, transplantation timing, pretransplant and posttransplant care,” noted Dr. Bahaaldin Alsoufi, of the division of cardiothoracic surgery, Children’s Healthcare of Atlanta, Emory University. “The effect of donor/recipient race mismatch warrants further investigation and might impact organ allocation algorithms or immunosuppression management,” wrote Dr. Alsoufi and his colleagues.
The researchers analyzed results of 124 children with CHD who had heart transplants from 1988 to 2013 at Emory University and Children’s Healthcare of Atlanta. Median age was 3.8 years; 61% were boys. Ten years after heart transplantation, 44% (54) of patients were alive without a second transplant, 13% (17) had a second transplant and 43% (53) died without a second transplant. After the second transplant, 9 of the 17 patients were alive, but 3 of them had gone onto a third transplant. Overall 15-year survival following the first transplant was 41% (51).
The study cited data from the Registry of the International Society for Heart and Lung Transplantation that reported more than 11,000 pediatric heart transplants worldwide in 2013, and CHD represents about 54% of all heart transplants in infants.
A multivariate analysis identified the following risk factors for early mortality after transplant: age younger than 12 months (hazard ration [HR] 7.2) and prolonged cardiopulmonary bypass (HR 5). Late-phase mortality risk factors were age younger than 12 months (HR 3) and donor/recipient race mismatch (HR 2.2).
“Survival was not affected by era, underlying anomaly, prior Fontan, sensitization or pulmonary artery augmentation,” wrote Dr. Alsoufi and his colleagues.
Among the risk factors, longer bypass times may be a surrogate for a more complicated operation, the authors said. But where prior sternotomy is a risk factor following a heart transplant in adults, the study found no such risk in children. Another risk factor previous reports identified is pulmonary artery augmentation, but, again, this study found no risk in the pediatric group.
The researchers looked at days on the waiting list, with a median wait of 39 days in the study group. In all, 175 children were listed for transplants, but 51 did not go through for various reasons. Most of the children with CHD who had a heart transplant had previous surgery; only 13% had a primary heart transplant, mostly in the earlier phase of the study.
Dr. Alsoufi and coauthors also identified African American race as a risk factor for lower survival, which is consistent with other reports. But this study agreed with a previous report that donor/recipient race mismatch was a significant risk factor in white and African American patients (Ann Thorac Surg. 2009;87:204-9). “While our finding might be anecdotal and specific to our geographic population, this warrants some investigation and might have some impact on future organ allocation algorithms and immunosuppression management,” the researchers wrote.
The authors had no relevant disclosures. Emory University School of Medicine, Children’s Healthcare of Atlanta provided study funding.
In his invited commentary, Dr. Robert D.B. Jaquiss of Duke University, Durham, N.C., took issue with the study authors’ “distress” at the lack of improvement in survival over the 25-year term of the study (J Thorac Cardiovasc Surg. 2015;150:1463-4) . Using the year 2000 as a demarcation line for early and late-phase results, Dr. Jaquiss said, “It must be pointed out that in the latter period recipients were much more ill.” He noted that 89% of post-2000 heart transplant patients had UNOS status 1 vs. 49% in the pre-2000 period.

|
Dr. Robert Jaquiss |
“Considering these between-era differences, an alternative, less ‘discouraging’ interpretation is that excellent outcomes were maintained despite the trend toward transplantation in sicker patients, undergoing more complex transplants, with longer ischemic times,” he said.
Dr. Jaquiss also cited “remarkably outstanding outcomes” in Fontan patients, reporting only one operative death in 33 patients. He found the lower survival for African-American patients in the study group “more sobering,” but also controversial because, among other reasons, “a complete mechanistic explanation remains elusive.” How these findings influence pediatric heart transplant practice “requires thoughtful and extensive investigation and discussion,” he said.
Wait-list mortality and mechanical bridge to transplant also deserve mention, he noted. “Though they are only briefly mentioned, the patients who died prior to transplant provide mute testimony to the lack of timely access to suitable donors,” Dr. Jaquiss said. Durable mechanical circulatory support can provide a bridge for these patients, but was not available through the majority of the study period.
“It is striking that no patient in this report was supported by a ventricular assist device (VAD), and only a small number (5%) had been on [extracorporeal membrane oxygenation] support,” Dr. Jaquiss said. “This is an unfortunate and unavoidable weakness of this report, given the recent introduction of VADs for pediatric heart transplant candidates.” The use of VAD in patients with CHD is “increasing rapidly,” he said.
Dr. Jaquiss had no disclosures.
In his invited commentary, Dr. Robert D.B. Jaquiss of Duke University, Durham, N.C., took issue with the study authors’ “distress” at the lack of improvement in survival over the 25-year term of the study (J Thorac Cardiovasc Surg. 2015;150:1463-4) . Using the year 2000 as a demarcation line for early and late-phase results, Dr. Jaquiss said, “It must be pointed out that in the latter period recipients were much more ill.” He noted that 89% of post-2000 heart transplant patients had UNOS status 1 vs. 49% in the pre-2000 period.

|
Dr. Robert Jaquiss |
“Considering these between-era differences, an alternative, less ‘discouraging’ interpretation is that excellent outcomes were maintained despite the trend toward transplantation in sicker patients, undergoing more complex transplants, with longer ischemic times,” he said.
Dr. Jaquiss also cited “remarkably outstanding outcomes” in Fontan patients, reporting only one operative death in 33 patients. He found the lower survival for African-American patients in the study group “more sobering,” but also controversial because, among other reasons, “a complete mechanistic explanation remains elusive.” How these findings influence pediatric heart transplant practice “requires thoughtful and extensive investigation and discussion,” he said.
Wait-list mortality and mechanical bridge to transplant also deserve mention, he noted. “Though they are only briefly mentioned, the patients who died prior to transplant provide mute testimony to the lack of timely access to suitable donors,” Dr. Jaquiss said. Durable mechanical circulatory support can provide a bridge for these patients, but was not available through the majority of the study period.
“It is striking that no patient in this report was supported by a ventricular assist device (VAD), and only a small number (5%) had been on [extracorporeal membrane oxygenation] support,” Dr. Jaquiss said. “This is an unfortunate and unavoidable weakness of this report, given the recent introduction of VADs for pediatric heart transplant candidates.” The use of VAD in patients with CHD is “increasing rapidly,” he said.
Dr. Jaquiss had no disclosures.
In his invited commentary, Dr. Robert D.B. Jaquiss of Duke University, Durham, N.C., took issue with the study authors’ “distress” at the lack of improvement in survival over the 25-year term of the study (J Thorac Cardiovasc Surg. 2015;150:1463-4) . Using the year 2000 as a demarcation line for early and late-phase results, Dr. Jaquiss said, “It must be pointed out that in the latter period recipients were much more ill.” He noted that 89% of post-2000 heart transplant patients had UNOS status 1 vs. 49% in the pre-2000 period.

|
Dr. Robert Jaquiss |
“Considering these between-era differences, an alternative, less ‘discouraging’ interpretation is that excellent outcomes were maintained despite the trend toward transplantation in sicker patients, undergoing more complex transplants, with longer ischemic times,” he said.
Dr. Jaquiss also cited “remarkably outstanding outcomes” in Fontan patients, reporting only one operative death in 33 patients. He found the lower survival for African-American patients in the study group “more sobering,” but also controversial because, among other reasons, “a complete mechanistic explanation remains elusive.” How these findings influence pediatric heart transplant practice “requires thoughtful and extensive investigation and discussion,” he said.
Wait-list mortality and mechanical bridge to transplant also deserve mention, he noted. “Though they are only briefly mentioned, the patients who died prior to transplant provide mute testimony to the lack of timely access to suitable donors,” Dr. Jaquiss said. Durable mechanical circulatory support can provide a bridge for these patients, but was not available through the majority of the study period.
“It is striking that no patient in this report was supported by a ventricular assist device (VAD), and only a small number (5%) had been on [extracorporeal membrane oxygenation] support,” Dr. Jaquiss said. “This is an unfortunate and unavoidable weakness of this report, given the recent introduction of VADs for pediatric heart transplant candidates.” The use of VAD in patients with CHD is “increasing rapidly,” he said.
Dr. Jaquiss had no disclosures.
A 25-year study of heart transplants in children with congenital heart disease (CHD) at one institution has found that results haven’t improved over time despite advances in technology and techniques. To improve outcomes, transplant surgeons may need to do a better job of selecting patients and matching patients and donors, according to study in the December issue of the Journal of Thoracic and Cardiovascular Surgery (J Thorac Cardiovasc Surg. 2015;150:1455-62).
“Strategies to improve outcomes in CHD patients might need to address selection criteria, transplantation timing, pretransplant and posttransplant care,” noted Dr. Bahaaldin Alsoufi, of the division of cardiothoracic surgery, Children’s Healthcare of Atlanta, Emory University. “The effect of donor/recipient race mismatch warrants further investigation and might impact organ allocation algorithms or immunosuppression management,” wrote Dr. Alsoufi and his colleagues.
The researchers analyzed results of 124 children with CHD who had heart transplants from 1988 to 2013 at Emory University and Children’s Healthcare of Atlanta. Median age was 3.8 years; 61% were boys. Ten years after heart transplantation, 44% (54) of patients were alive without a second transplant, 13% (17) had a second transplant and 43% (53) died without a second transplant. After the second transplant, 9 of the 17 patients were alive, but 3 of them had gone onto a third transplant. Overall 15-year survival following the first transplant was 41% (51).
The study cited data from the Registry of the International Society for Heart and Lung Transplantation that reported more than 11,000 pediatric heart transplants worldwide in 2013, and CHD represents about 54% of all heart transplants in infants.
A multivariate analysis identified the following risk factors for early mortality after transplant: age younger than 12 months (hazard ration [HR] 7.2) and prolonged cardiopulmonary bypass (HR 5). Late-phase mortality risk factors were age younger than 12 months (HR 3) and donor/recipient race mismatch (HR 2.2).
“Survival was not affected by era, underlying anomaly, prior Fontan, sensitization or pulmonary artery augmentation,” wrote Dr. Alsoufi and his colleagues.
Among the risk factors, longer bypass times may be a surrogate for a more complicated operation, the authors said. But where prior sternotomy is a risk factor following a heart transplant in adults, the study found no such risk in children. Another risk factor previous reports identified is pulmonary artery augmentation, but, again, this study found no risk in the pediatric group.
The researchers looked at days on the waiting list, with a median wait of 39 days in the study group. In all, 175 children were listed for transplants, but 51 did not go through for various reasons. Most of the children with CHD who had a heart transplant had previous surgery; only 13% had a primary heart transplant, mostly in the earlier phase of the study.
Dr. Alsoufi and coauthors also identified African American race as a risk factor for lower survival, which is consistent with other reports. But this study agreed with a previous report that donor/recipient race mismatch was a significant risk factor in white and African American patients (Ann Thorac Surg. 2009;87:204-9). “While our finding might be anecdotal and specific to our geographic population, this warrants some investigation and might have some impact on future organ allocation algorithms and immunosuppression management,” the researchers wrote.
The authors had no relevant disclosures. Emory University School of Medicine, Children’s Healthcare of Atlanta provided study funding.
A 25-year study of heart transplants in children with congenital heart disease (CHD) at one institution has found that results haven’t improved over time despite advances in technology and techniques. To improve outcomes, transplant surgeons may need to do a better job of selecting patients and matching patients and donors, according to study in the December issue of the Journal of Thoracic and Cardiovascular Surgery (J Thorac Cardiovasc Surg. 2015;150:1455-62).
“Strategies to improve outcomes in CHD patients might need to address selection criteria, transplantation timing, pretransplant and posttransplant care,” noted Dr. Bahaaldin Alsoufi, of the division of cardiothoracic surgery, Children’s Healthcare of Atlanta, Emory University. “The effect of donor/recipient race mismatch warrants further investigation and might impact organ allocation algorithms or immunosuppression management,” wrote Dr. Alsoufi and his colleagues.
The researchers analyzed results of 124 children with CHD who had heart transplants from 1988 to 2013 at Emory University and Children’s Healthcare of Atlanta. Median age was 3.8 years; 61% were boys. Ten years after heart transplantation, 44% (54) of patients were alive without a second transplant, 13% (17) had a second transplant and 43% (53) died without a second transplant. After the second transplant, 9 of the 17 patients were alive, but 3 of them had gone onto a third transplant. Overall 15-year survival following the first transplant was 41% (51).
The study cited data from the Registry of the International Society for Heart and Lung Transplantation that reported more than 11,000 pediatric heart transplants worldwide in 2013, and CHD represents about 54% of all heart transplants in infants.
A multivariate analysis identified the following risk factors for early mortality after transplant: age younger than 12 months (hazard ration [HR] 7.2) and prolonged cardiopulmonary bypass (HR 5). Late-phase mortality risk factors were age younger than 12 months (HR 3) and donor/recipient race mismatch (HR 2.2).
“Survival was not affected by era, underlying anomaly, prior Fontan, sensitization or pulmonary artery augmentation,” wrote Dr. Alsoufi and his colleagues.
Among the risk factors, longer bypass times may be a surrogate for a more complicated operation, the authors said. But where prior sternotomy is a risk factor following a heart transplant in adults, the study found no such risk in children. Another risk factor previous reports identified is pulmonary artery augmentation, but, again, this study found no risk in the pediatric group.
The researchers looked at days on the waiting list, with a median wait of 39 days in the study group. In all, 175 children were listed for transplants, but 51 did not go through for various reasons. Most of the children with CHD who had a heart transplant had previous surgery; only 13% had a primary heart transplant, mostly in the earlier phase of the study.
Dr. Alsoufi and coauthors also identified African American race as a risk factor for lower survival, which is consistent with other reports. But this study agreed with a previous report that donor/recipient race mismatch was a significant risk factor in white and African American patients (Ann Thorac Surg. 2009;87:204-9). “While our finding might be anecdotal and specific to our geographic population, this warrants some investigation and might have some impact on future organ allocation algorithms and immunosuppression management,” the researchers wrote.
The authors had no relevant disclosures. Emory University School of Medicine, Children’s Healthcare of Atlanta provided study funding.
Key clinical point: Pediatric heart transplantation outcomes for congenital heart disease haven’t improved in the current era, indicating ongoing challenges.
Major finding: Ten years following heart transplantation, 13% of patients had undergone retransplantation, 43% had died without retransplantation, and 44% were alive without retransplantation.
Data source: A review of 124 children with congenital heart disease who had heart transplantation at a single center.
Disclosures: The study authors had no relationships to disclose.
Conservative management for AR safe at 10 years
Whether to operate on patients with severe aortic regurgitation (AR) before or after symptoms appear has been a point of controversy among cardiothoracic surgeons, but a recent study has found that patients who have early surgery may not fare any better for up to 10 years than those who opt for a more conservative “watchful waiting” course of care.
Investigators from Belgium reported results from an analysis of 160 patients in the November issue of the Journal of Thoracic and Cardiovascular Surgery (2015;150:1100-08). “In asymptomatic severe AR, delaying surgery until the onset of class I/IIa operative triggers is safe, supporting current guidelines,” said Dr. Christophe de Meester and colleagues at the Catholic University of Louvain and St. Luc University Clinic in Brussels.
The goal of the study was to evaluate long-term outcomes and incidence of cardiac complications in patients with severe AR who did not have any signs and symptoms that called for surgery, and who either had surgery early on or entered conservative management and eventually had an operation when signs and symptoms did appear.
The study found that close follow-up and monitoring of patients with severe AR was a cornerstone of successful conservative management. “We found that survival was similar between the two groups,” Dr. De Meester and coauthors said. “Better survival was nonetheless observed in conservatively managed patients with regular as opposed to no or a looser follow-up.”
The most recent European Society of Cardiology (ESC) guidelines and American Heart Association/American College of Cardiology guidelines state that symptomatic severe AR is a class I indication for surgery regardless of left ventricular (LV) systolic function.
However, Dr. De Meester and colleagues said, the timing of that surgery is not so clear-cut. Earlier studies have shown that surgery could be delayed for patients with minimal symptoms, but more recent evidence has suggested the opposite, according to the study. Two factors favor surgery before symptoms arise – poor aortic valve repair outcomes in patients with symptoms of heart failure and long-standing severe AR, which eventually leads to LV dysfunction.
Yet, the latest ESC guidelines have been “reluctant” to make a strong case for early surgery before symptoms of LV dysfunction appear, and the AHA/ACC guidelines call for surgery only when symptoms of LV dysfunction or LV dilatation develop, Dr. de Meester and his coauthors said.
In the past, the risks of aortic valve replacement were too high to consider early surgery, the study authors said. “However, with the advent of aortic valve repair, operative mortality and long-term outcomes have improved to such an extent that early surgery has become a plausible option for patients.”
But the risk of these patients developing symptoms for surgery was nonetheless low over 10 years, the study found: 7.4% for developing severe LV dilatation; 0.6% for becoming symptomatic; and 0.9% for developing LV dysfunction. Overall, the rate of adverse events in the study population was 9.9% at 10 years.
In the study, 69 patients were initially managed conservatively, 49 of whom were in the watchful waiting group that visited a cardiologist at least annually and another 20 considered an “irregular follow-up subgroup.” Among the watchful waiting group, 31 developed symptoms for surgery (only two declined surgery). Watchful waiting patients had five- and 10-year survival of 100% and 95%, respectively, compared with 90% and 79% among those who had irregular follow-up.
Overall, the conservatively managed group had outcomes better than or equal to the early surgery group. Ten-year cardiovascular survival was 96% in both groups, whereas event-free survival was 92% at 10 years in the conservatively managed group vs. 81% in the early surgery group.
The study was supported by the Belgium National Fund for Scientific Research. The authors had no conflicts to disclose.
The design of the Belgium study “challenges” existing treatment guidelines for asymptomatic chronic aortic insufficiency in two ways, Dr. Leora Balsam and Dr. Abe deAndra Jr., both of the New York University-Langone Medical Center, write in their commentary (J Thorac Cardiovasc Surg. 2015;150:1108-10): first, by making aortic valve repair the preferred surgical treatment in the study and, secondly, by offering surgery to both symptomatic and asymptomatic patients.
“In the era of evidence-based medicine,” Dr. Balsam and Dr. deAndra wrote, “there remains a need for research and innovation even in areas where guidelines exist.”
While many authors have described aortic valve repair as an alternative to aortic valve replacement for chronic severe aortic insufficiency, Dr. Balsam and Dr. deAndra explained that the term aortic valve repair “encompasses a wide array of techniques,” among them valve-sparing aortic root replacement, subcommissural annuloplasty and “myriad” leaf resection, plication, and reconstruction techniques. Because of mounting reports of excellent results with aortic valve repair techniques, growing ranks of cardiothoracic surgeons have advocated for repair as an early intervention for aortic valve problems. But the question remains: “Have we identified the optimal triggers for intervention for aortic insufficiency?” they asked. “The answer is probably no, and that newer technology and diagnostic studies will better discriminate between patients that can benefit from intervention and those that will not.”
Dr. Balsam and Dr. deAndra had no disclosures.
The design of the Belgium study “challenges” existing treatment guidelines for asymptomatic chronic aortic insufficiency in two ways, Dr. Leora Balsam and Dr. Abe deAndra Jr., both of the New York University-Langone Medical Center, write in their commentary (J Thorac Cardiovasc Surg. 2015;150:1108-10): first, by making aortic valve repair the preferred surgical treatment in the study and, secondly, by offering surgery to both symptomatic and asymptomatic patients.
“In the era of evidence-based medicine,” Dr. Balsam and Dr. deAndra wrote, “there remains a need for research and innovation even in areas where guidelines exist.”
While many authors have described aortic valve repair as an alternative to aortic valve replacement for chronic severe aortic insufficiency, Dr. Balsam and Dr. deAndra explained that the term aortic valve repair “encompasses a wide array of techniques,” among them valve-sparing aortic root replacement, subcommissural annuloplasty and “myriad” leaf resection, plication, and reconstruction techniques. Because of mounting reports of excellent results with aortic valve repair techniques, growing ranks of cardiothoracic surgeons have advocated for repair as an early intervention for aortic valve problems. But the question remains: “Have we identified the optimal triggers for intervention for aortic insufficiency?” they asked. “The answer is probably no, and that newer technology and diagnostic studies will better discriminate between patients that can benefit from intervention and those that will not.”
Dr. Balsam and Dr. deAndra had no disclosures.
The design of the Belgium study “challenges” existing treatment guidelines for asymptomatic chronic aortic insufficiency in two ways, Dr. Leora Balsam and Dr. Abe deAndra Jr., both of the New York University-Langone Medical Center, write in their commentary (J Thorac Cardiovasc Surg. 2015;150:1108-10): first, by making aortic valve repair the preferred surgical treatment in the study and, secondly, by offering surgery to both symptomatic and asymptomatic patients.
“In the era of evidence-based medicine,” Dr. Balsam and Dr. deAndra wrote, “there remains a need for research and innovation even in areas where guidelines exist.”
While many authors have described aortic valve repair as an alternative to aortic valve replacement for chronic severe aortic insufficiency, Dr. Balsam and Dr. deAndra explained that the term aortic valve repair “encompasses a wide array of techniques,” among them valve-sparing aortic root replacement, subcommissural annuloplasty and “myriad” leaf resection, plication, and reconstruction techniques. Because of mounting reports of excellent results with aortic valve repair techniques, growing ranks of cardiothoracic surgeons have advocated for repair as an early intervention for aortic valve problems. But the question remains: “Have we identified the optimal triggers for intervention for aortic insufficiency?” they asked. “The answer is probably no, and that newer technology and diagnostic studies will better discriminate between patients that can benefit from intervention and those that will not.”
Dr. Balsam and Dr. deAndra had no disclosures.
Whether to operate on patients with severe aortic regurgitation (AR) before or after symptoms appear has been a point of controversy among cardiothoracic surgeons, but a recent study has found that patients who have early surgery may not fare any better for up to 10 years than those who opt for a more conservative “watchful waiting” course of care.
Investigators from Belgium reported results from an analysis of 160 patients in the November issue of the Journal of Thoracic and Cardiovascular Surgery (2015;150:1100-08). “In asymptomatic severe AR, delaying surgery until the onset of class I/IIa operative triggers is safe, supporting current guidelines,” said Dr. Christophe de Meester and colleagues at the Catholic University of Louvain and St. Luc University Clinic in Brussels.
The goal of the study was to evaluate long-term outcomes and incidence of cardiac complications in patients with severe AR who did not have any signs and symptoms that called for surgery, and who either had surgery early on or entered conservative management and eventually had an operation when signs and symptoms did appear.
The study found that close follow-up and monitoring of patients with severe AR was a cornerstone of successful conservative management. “We found that survival was similar between the two groups,” Dr. De Meester and coauthors said. “Better survival was nonetheless observed in conservatively managed patients with regular as opposed to no or a looser follow-up.”
The most recent European Society of Cardiology (ESC) guidelines and American Heart Association/American College of Cardiology guidelines state that symptomatic severe AR is a class I indication for surgery regardless of left ventricular (LV) systolic function.
However, Dr. De Meester and colleagues said, the timing of that surgery is not so clear-cut. Earlier studies have shown that surgery could be delayed for patients with minimal symptoms, but more recent evidence has suggested the opposite, according to the study. Two factors favor surgery before symptoms arise – poor aortic valve repair outcomes in patients with symptoms of heart failure and long-standing severe AR, which eventually leads to LV dysfunction.
Yet, the latest ESC guidelines have been “reluctant” to make a strong case for early surgery before symptoms of LV dysfunction appear, and the AHA/ACC guidelines call for surgery only when symptoms of LV dysfunction or LV dilatation develop, Dr. de Meester and his coauthors said.
In the past, the risks of aortic valve replacement were too high to consider early surgery, the study authors said. “However, with the advent of aortic valve repair, operative mortality and long-term outcomes have improved to such an extent that early surgery has become a plausible option for patients.”
But the risk of these patients developing symptoms for surgery was nonetheless low over 10 years, the study found: 7.4% for developing severe LV dilatation; 0.6% for becoming symptomatic; and 0.9% for developing LV dysfunction. Overall, the rate of adverse events in the study population was 9.9% at 10 years.
In the study, 69 patients were initially managed conservatively, 49 of whom were in the watchful waiting group that visited a cardiologist at least annually and another 20 considered an “irregular follow-up subgroup.” Among the watchful waiting group, 31 developed symptoms for surgery (only two declined surgery). Watchful waiting patients had five- and 10-year survival of 100% and 95%, respectively, compared with 90% and 79% among those who had irregular follow-up.
Overall, the conservatively managed group had outcomes better than or equal to the early surgery group. Ten-year cardiovascular survival was 96% in both groups, whereas event-free survival was 92% at 10 years in the conservatively managed group vs. 81% in the early surgery group.
The study was supported by the Belgium National Fund for Scientific Research. The authors had no conflicts to disclose.
Whether to operate on patients with severe aortic regurgitation (AR) before or after symptoms appear has been a point of controversy among cardiothoracic surgeons, but a recent study has found that patients who have early surgery may not fare any better for up to 10 years than those who opt for a more conservative “watchful waiting” course of care.
Investigators from Belgium reported results from an analysis of 160 patients in the November issue of the Journal of Thoracic and Cardiovascular Surgery (2015;150:1100-08). “In asymptomatic severe AR, delaying surgery until the onset of class I/IIa operative triggers is safe, supporting current guidelines,” said Dr. Christophe de Meester and colleagues at the Catholic University of Louvain and St. Luc University Clinic in Brussels.
The goal of the study was to evaluate long-term outcomes and incidence of cardiac complications in patients with severe AR who did not have any signs and symptoms that called for surgery, and who either had surgery early on or entered conservative management and eventually had an operation when signs and symptoms did appear.
The study found that close follow-up and monitoring of patients with severe AR was a cornerstone of successful conservative management. “We found that survival was similar between the two groups,” Dr. De Meester and coauthors said. “Better survival was nonetheless observed in conservatively managed patients with regular as opposed to no or a looser follow-up.”
The most recent European Society of Cardiology (ESC) guidelines and American Heart Association/American College of Cardiology guidelines state that symptomatic severe AR is a class I indication for surgery regardless of left ventricular (LV) systolic function.
However, Dr. De Meester and colleagues said, the timing of that surgery is not so clear-cut. Earlier studies have shown that surgery could be delayed for patients with minimal symptoms, but more recent evidence has suggested the opposite, according to the study. Two factors favor surgery before symptoms arise – poor aortic valve repair outcomes in patients with symptoms of heart failure and long-standing severe AR, which eventually leads to LV dysfunction.
Yet, the latest ESC guidelines have been “reluctant” to make a strong case for early surgery before symptoms of LV dysfunction appear, and the AHA/ACC guidelines call for surgery only when symptoms of LV dysfunction or LV dilatation develop, Dr. de Meester and his coauthors said.
In the past, the risks of aortic valve replacement were too high to consider early surgery, the study authors said. “However, with the advent of aortic valve repair, operative mortality and long-term outcomes have improved to such an extent that early surgery has become a plausible option for patients.”
But the risk of these patients developing symptoms for surgery was nonetheless low over 10 years, the study found: 7.4% for developing severe LV dilatation; 0.6% for becoming symptomatic; and 0.9% for developing LV dysfunction. Overall, the rate of adverse events in the study population was 9.9% at 10 years.
In the study, 69 patients were initially managed conservatively, 49 of whom were in the watchful waiting group that visited a cardiologist at least annually and another 20 considered an “irregular follow-up subgroup.” Among the watchful waiting group, 31 developed symptoms for surgery (only two declined surgery). Watchful waiting patients had five- and 10-year survival of 100% and 95%, respectively, compared with 90% and 79% among those who had irregular follow-up.
Overall, the conservatively managed group had outcomes better than or equal to the early surgery group. Ten-year cardiovascular survival was 96% in both groups, whereas event-free survival was 92% at 10 years in the conservatively managed group vs. 81% in the early surgery group.
The study was supported by the Belgium National Fund for Scientific Research. The authors had no conflicts to disclose.
FROM THE JOURNAL OF THORACIC AND CARDIOVASCULAR SURGERY
Key clinical point: Delaying surgery until the onset of symptoms of aortic insufficiency is safe, in support of current clinical guidelines.
Major finding: Ten-year cardiovascular survival was equal among conservatively managed and early-surgery groups, but event free survival was 92% at 10 years in the conservatively managed group vs. 81% in the early surgery group.
Data source: Analysis of 160 consecutive asymptomatic patients with severe aortic regurgitation who were assigned to either conservative management or early surgery and followed up for a median of 7.2 years.
Disclosures: The Belgium National Fund of Scientific Research supported the study. The authors had no disclosures.
Does LVAD inhibit cardio protection?
Placement of a left ventricular assist device (LVAD) after a heart attack has been found to suppress certain cellular signaling pathways that protect coronary tissue, but at the same time LVAD placement seemed to help normalize other protective properties in areas of the heart closest to the infarcted region, investigators reported in a recent study.
The findings could have implications in determining the best method for unloading and other medical therapies in the aftermath of a heart attack, Dr. Keshava Rajagopal of the University of Texas, Houston, and associates reported in the November issue of the Journal of Thoracic and Cardiovascular Surgery (2015;150:1332-41).
To study the effect of LVAD on cardiac tissue, the investigators induced myocardial infarction in sheep and then placed the animals on LVAD support for 2 weeks. After 10 more weeks of observation, the investigators harvested and analyzed the myocardial specimens. The principal goal of the study was to investigate how heart attack and subsequent short-term mechanical support of the left ventricle can influence signaling controlled by the protein beta-arrestin.
They found that an infarction of myocardial tissue caused activation of the beta-arrestin protein that regulates cellular pathways that can benefit cardiac cells. At the same time, LVAD support inhibited beta-arrestin activation, specifically in regulating pathways of two cardioprotective proteins: Akt, also called protein kinase B (PKB), and, to a lesser extent, ERK-1 and -2.
They also found that MI resulted in regional activation of load-induced signaling of cardiac G protein-coupled receptor (GPCR) via G proteins.
“These studies demonstrate that small platform catheter-based LVAD support exerts suppressive effects on cardioprotective beta-arrestin–mediated signal transduction, while normalizing the signaling networks of G-alpha-q–coupled cardiac GPCRS in the MI-adjacent zone,” Dr. Rajagopal and colleagues said.
They acknowledged that further studies are needed to better understand the roles that specific GPCRs in beta-arrestin–regulated signaling play in left ventricle dysfunction after a heart attack and to help define the optimal timing for LVAD based on signaling and genetic markers along with standard LV functional endpoints.
The authors had no disclosures.
The University of Maryland investigators in this study have joined the ranks of other investigators who have begun to unravel the consequences of mechanical unloading at the cellular level as well as its effect on the heart’s ability to handle calcium after infarction, Dr. William Hiesinger and Dr. Pavan Atluri of the University of Pennsylvania wrote in their invited commentary (J Thorac Cardiovasc Surg. 2015;150:1342-3).
“More broadly, these investigations are building the foundation of what will likely be the best platform for an efficacious bridge to recovery: multimodal therapy utilizing the titration of mechanical myocardial unloading,” they said. Dr. Hiesinger and Dr. Atluri commented on the limitations of the University of Maryland study, namely its small sample size and narrow scope. “This is, however, reflective more of the amazing complexity of the biologic and mechanical interactions between the heart and VAD and the need for further investigations of this kind than the quality of the research,” they said.
Understanding the molecular basis and metabolic function cardiac dysfunction after a heart attack is in the “nascent stages,” and even less is known about the effect ventricular loading has on these pathways, Dr. Hiesinger and Dr. Atluri said. “This study offers a concrete platform for both specific treatment and further study,” they wrote.
The University of Maryland investigators in this study have joined the ranks of other investigators who have begun to unravel the consequences of mechanical unloading at the cellular level as well as its effect on the heart’s ability to handle calcium after infarction, Dr. William Hiesinger and Dr. Pavan Atluri of the University of Pennsylvania wrote in their invited commentary (J Thorac Cardiovasc Surg. 2015;150:1342-3).
“More broadly, these investigations are building the foundation of what will likely be the best platform for an efficacious bridge to recovery: multimodal therapy utilizing the titration of mechanical myocardial unloading,” they said. Dr. Hiesinger and Dr. Atluri commented on the limitations of the University of Maryland study, namely its small sample size and narrow scope. “This is, however, reflective more of the amazing complexity of the biologic and mechanical interactions between the heart and VAD and the need for further investigations of this kind than the quality of the research,” they said.
Understanding the molecular basis and metabolic function cardiac dysfunction after a heart attack is in the “nascent stages,” and even less is known about the effect ventricular loading has on these pathways, Dr. Hiesinger and Dr. Atluri said. “This study offers a concrete platform for both specific treatment and further study,” they wrote.
The University of Maryland investigators in this study have joined the ranks of other investigators who have begun to unravel the consequences of mechanical unloading at the cellular level as well as its effect on the heart’s ability to handle calcium after infarction, Dr. William Hiesinger and Dr. Pavan Atluri of the University of Pennsylvania wrote in their invited commentary (J Thorac Cardiovasc Surg. 2015;150:1342-3).
“More broadly, these investigations are building the foundation of what will likely be the best platform for an efficacious bridge to recovery: multimodal therapy utilizing the titration of mechanical myocardial unloading,” they said. Dr. Hiesinger and Dr. Atluri commented on the limitations of the University of Maryland study, namely its small sample size and narrow scope. “This is, however, reflective more of the amazing complexity of the biologic and mechanical interactions between the heart and VAD and the need for further investigations of this kind than the quality of the research,” they said.
Understanding the molecular basis and metabolic function cardiac dysfunction after a heart attack is in the “nascent stages,” and even less is known about the effect ventricular loading has on these pathways, Dr. Hiesinger and Dr. Atluri said. “This study offers a concrete platform for both specific treatment and further study,” they wrote.
Placement of a left ventricular assist device (LVAD) after a heart attack has been found to suppress certain cellular signaling pathways that protect coronary tissue, but at the same time LVAD placement seemed to help normalize other protective properties in areas of the heart closest to the infarcted region, investigators reported in a recent study.
The findings could have implications in determining the best method for unloading and other medical therapies in the aftermath of a heart attack, Dr. Keshava Rajagopal of the University of Texas, Houston, and associates reported in the November issue of the Journal of Thoracic and Cardiovascular Surgery (2015;150:1332-41).
To study the effect of LVAD on cardiac tissue, the investigators induced myocardial infarction in sheep and then placed the animals on LVAD support for 2 weeks. After 10 more weeks of observation, the investigators harvested and analyzed the myocardial specimens. The principal goal of the study was to investigate how heart attack and subsequent short-term mechanical support of the left ventricle can influence signaling controlled by the protein beta-arrestin.
They found that an infarction of myocardial tissue caused activation of the beta-arrestin protein that regulates cellular pathways that can benefit cardiac cells. At the same time, LVAD support inhibited beta-arrestin activation, specifically in regulating pathways of two cardioprotective proteins: Akt, also called protein kinase B (PKB), and, to a lesser extent, ERK-1 and -2.
They also found that MI resulted in regional activation of load-induced signaling of cardiac G protein-coupled receptor (GPCR) via G proteins.
“These studies demonstrate that small platform catheter-based LVAD support exerts suppressive effects on cardioprotective beta-arrestin–mediated signal transduction, while normalizing the signaling networks of G-alpha-q–coupled cardiac GPCRS in the MI-adjacent zone,” Dr. Rajagopal and colleagues said.
They acknowledged that further studies are needed to better understand the roles that specific GPCRs in beta-arrestin–regulated signaling play in left ventricle dysfunction after a heart attack and to help define the optimal timing for LVAD based on signaling and genetic markers along with standard LV functional endpoints.
The authors had no disclosures.
Placement of a left ventricular assist device (LVAD) after a heart attack has been found to suppress certain cellular signaling pathways that protect coronary tissue, but at the same time LVAD placement seemed to help normalize other protective properties in areas of the heart closest to the infarcted region, investigators reported in a recent study.
The findings could have implications in determining the best method for unloading and other medical therapies in the aftermath of a heart attack, Dr. Keshava Rajagopal of the University of Texas, Houston, and associates reported in the November issue of the Journal of Thoracic and Cardiovascular Surgery (2015;150:1332-41).
To study the effect of LVAD on cardiac tissue, the investigators induced myocardial infarction in sheep and then placed the animals on LVAD support for 2 weeks. After 10 more weeks of observation, the investigators harvested and analyzed the myocardial specimens. The principal goal of the study was to investigate how heart attack and subsequent short-term mechanical support of the left ventricle can influence signaling controlled by the protein beta-arrestin.
They found that an infarction of myocardial tissue caused activation of the beta-arrestin protein that regulates cellular pathways that can benefit cardiac cells. At the same time, LVAD support inhibited beta-arrestin activation, specifically in regulating pathways of two cardioprotective proteins: Akt, also called protein kinase B (PKB), and, to a lesser extent, ERK-1 and -2.
They also found that MI resulted in regional activation of load-induced signaling of cardiac G protein-coupled receptor (GPCR) via G proteins.
“These studies demonstrate that small platform catheter-based LVAD support exerts suppressive effects on cardioprotective beta-arrestin–mediated signal transduction, while normalizing the signaling networks of G-alpha-q–coupled cardiac GPCRS in the MI-adjacent zone,” Dr. Rajagopal and colleagues said.
They acknowledged that further studies are needed to better understand the roles that specific GPCRs in beta-arrestin–regulated signaling play in left ventricle dysfunction after a heart attack and to help define the optimal timing for LVAD based on signaling and genetic markers along with standard LV functional endpoints.
The authors had no disclosures.
FROM THE JOURNAL OF THORACIC AND CARDIOVASCULAR SURGERY
Key clinical point: Left ventricular assist device (LVAD) support inhibits pathologic responses to mechanical loading but also can inhibit adaptive responses after myocardial infarction.
Major finding: LVAD support inhibited cardioprotective beta-arrestin–mediated signaling, but net benefits of normalization of load-induced G protein-coupled receptor (GPCR) signaling were observed in the MI-adjacent zone.
Data source: Sheep were induced with myocardial infarction and then placed on LVAD support for 2 weeks and observed for a total of 12 weeks. Then myocardial specimens were harvested and analyzed.
Disclosures: The study authors had no relationships to disclose.
Hybrid revascularization shows promise, but there are concerns
A hybrid coronary revascularization procedure that combines off-pump left internal mammary artery (LIMA) grafting with percutaneous coronary intervention (PCI) showed good results at 1 year after surgery, but nonetheless showed a rate of adverse events that may raise questions about the procedure.
In a study published in the November issue of the Journal of Thoracic and Cardiovascular Surgery, a team of investigators from Aarhus University Hospital in Denmark reported high rates of graft patency and low rates of death and stroke with the procedure 1 year after a series of 100 operations (J Thorac Cardiovasc Surg. 2015;150:1181-6).
“The high left internal mammary artery graft patency rate and low risk of death and stroke at 1 year seem promising for the long-term outcome of this revascularization strategy,” said Dr. Ivy Susanne Modrau and colleagues.
The single-center study evaluated 1-year clinical and angiographic results of 100 consecutive trial patients with multivessel disease who had the hybrid procedure between October 2010 and February 2012. “The rationale of hybrid coronary revascularization is to achieve the survival benefits of the LIMA to LAD (left anterior descending artery) graft with reduced invasiveness to minimize postprocedural discomfort and morbidity, in particular the risk of stroke,” Dr. Modrau and colleagues said.
The study used the LIMA to LAD graft performed off-pump through a reversed J-hemisternotomy “We chose this technique because of its excellent exposure of the heart, technical ease, low risk of complicating chronic pain, and applicability in virtually all patients,” Dr. Modrau said. Eighty-nine patients had surgery prior to PCI and 11 had PCI prior to surgery.
The primary endpoint was rate of major adverse cardiac or cerebrovascular events (MACCE), the composite of all-cause death, stroke, myocardial infarction, and repeat revascularization by PCI or coronary artery bypass grafting at 1 year. Secondary endpoints included individual components and status of stent and graft patency on angiography.
Overall, 20 patients met the 1-year primary endpoint of MACCE. One patient died, one other had a stroke, and three had heart attacks. Sixteen patients had repeat revascularization procedures, eight performed during the index hospitalization. Graft patency was 98% after 1 year.
Dr. Modrau and coauthors noted the MACCE rate of 20% “was higher than expected,” and certainly higher than results in the SYNTAX study (17.8% in the PCI group and 12.4% in the coronary artery bypass grafting [CABG] group) (Euro. Intervention. 2015;10:e1-e6). One possible reason the Danish investigators cited for higher than expected MACCE rates was that they may be attributed to the learning curve involved with LIMA grafting and the use of early angiography possibly revealing “clinically silent LIMA graft dysfunction due to technical errors.”
The number of repeat revascularizations in the study was more in line with the SYNTAX study: 7% in the Aarhus University study and 6% in the SYNTAX CABG group. However, a meta-analysis of six studies with 1,190 patients reported 1-year repeat revascularization rates of 3.8% after a hybrid procedure and 1.4% after CABG (Am Heart J. 2014;167:585-92).
Ultimately, the safety and efficacy of the hybrid revascularization approach will require long-term follow-up data and head-to-head comparison with conventional CABG and PCI in clinical trials. “Meanwhile, LIMA patency, the cornerstone of surgical revascularization, may be used as a surrogate endpoint for long-term survival after HCR,” Dr. Modrau and coauthors said.
They reported having no disclosures.
Hybrid revascularization procedures are “still not ready for prime time,” Dr. Carlos Mestres of Cleveland Clinic Abu Dhabi, United Arab Emirates, said in his invited commentary (J Thorac Cardiovasc Surg. 2015;150:1028-9).
The study illuminates two key points of concern, Dr. Mestres said: the “unexpectedly high” 20% rate of major adverse cardiac or cerebrovascular events (MACCE); and the in-hospital revascularization rate that was significantly higher than the 1% after CABG that the authors reported in their own institution. That calls into question the reason the investigators would change their own department strategy away from conventional CABG, where they had optimal results, he said.
Dr. Mestres also said the Danish investigators’ conclusion that the study results seemed promising for long-term outcomes of the hybrid procedure “are to be carefully dissected.”
He commended the investigators for collecting angiographic data at 1 year, but said that 1 year of follow-up “is simply not enough” to credibly compare staged procedures with CABG.
Dr. Mestres had no disclosures.
Hybrid revascularization procedures are “still not ready for prime time,” Dr. Carlos Mestres of Cleveland Clinic Abu Dhabi, United Arab Emirates, said in his invited commentary (J Thorac Cardiovasc Surg. 2015;150:1028-9).
The study illuminates two key points of concern, Dr. Mestres said: the “unexpectedly high” 20% rate of major adverse cardiac or cerebrovascular events (MACCE); and the in-hospital revascularization rate that was significantly higher than the 1% after CABG that the authors reported in their own institution. That calls into question the reason the investigators would change their own department strategy away from conventional CABG, where they had optimal results, he said.
Dr. Mestres also said the Danish investigators’ conclusion that the study results seemed promising for long-term outcomes of the hybrid procedure “are to be carefully dissected.”
He commended the investigators for collecting angiographic data at 1 year, but said that 1 year of follow-up “is simply not enough” to credibly compare staged procedures with CABG.
Dr. Mestres had no disclosures.
Hybrid revascularization procedures are “still not ready for prime time,” Dr. Carlos Mestres of Cleveland Clinic Abu Dhabi, United Arab Emirates, said in his invited commentary (J Thorac Cardiovasc Surg. 2015;150:1028-9).
The study illuminates two key points of concern, Dr. Mestres said: the “unexpectedly high” 20% rate of major adverse cardiac or cerebrovascular events (MACCE); and the in-hospital revascularization rate that was significantly higher than the 1% after CABG that the authors reported in their own institution. That calls into question the reason the investigators would change their own department strategy away from conventional CABG, where they had optimal results, he said.
Dr. Mestres also said the Danish investigators’ conclusion that the study results seemed promising for long-term outcomes of the hybrid procedure “are to be carefully dissected.”
He commended the investigators for collecting angiographic data at 1 year, but said that 1 year of follow-up “is simply not enough” to credibly compare staged procedures with CABG.
Dr. Mestres had no disclosures.
A hybrid coronary revascularization procedure that combines off-pump left internal mammary artery (LIMA) grafting with percutaneous coronary intervention (PCI) showed good results at 1 year after surgery, but nonetheless showed a rate of adverse events that may raise questions about the procedure.
In a study published in the November issue of the Journal of Thoracic and Cardiovascular Surgery, a team of investigators from Aarhus University Hospital in Denmark reported high rates of graft patency and low rates of death and stroke with the procedure 1 year after a series of 100 operations (J Thorac Cardiovasc Surg. 2015;150:1181-6).
“The high left internal mammary artery graft patency rate and low risk of death and stroke at 1 year seem promising for the long-term outcome of this revascularization strategy,” said Dr. Ivy Susanne Modrau and colleagues.
The single-center study evaluated 1-year clinical and angiographic results of 100 consecutive trial patients with multivessel disease who had the hybrid procedure between October 2010 and February 2012. “The rationale of hybrid coronary revascularization is to achieve the survival benefits of the LIMA to LAD (left anterior descending artery) graft with reduced invasiveness to minimize postprocedural discomfort and morbidity, in particular the risk of stroke,” Dr. Modrau and colleagues said.
The study used the LIMA to LAD graft performed off-pump through a reversed J-hemisternotomy “We chose this technique because of its excellent exposure of the heart, technical ease, low risk of complicating chronic pain, and applicability in virtually all patients,” Dr. Modrau said. Eighty-nine patients had surgery prior to PCI and 11 had PCI prior to surgery.
The primary endpoint was rate of major adverse cardiac or cerebrovascular events (MACCE), the composite of all-cause death, stroke, myocardial infarction, and repeat revascularization by PCI or coronary artery bypass grafting at 1 year. Secondary endpoints included individual components and status of stent and graft patency on angiography.
Overall, 20 patients met the 1-year primary endpoint of MACCE. One patient died, one other had a stroke, and three had heart attacks. Sixteen patients had repeat revascularization procedures, eight performed during the index hospitalization. Graft patency was 98% after 1 year.
Dr. Modrau and coauthors noted the MACCE rate of 20% “was higher than expected,” and certainly higher than results in the SYNTAX study (17.8% in the PCI group and 12.4% in the coronary artery bypass grafting [CABG] group) (Euro. Intervention. 2015;10:e1-e6). One possible reason the Danish investigators cited for higher than expected MACCE rates was that they may be attributed to the learning curve involved with LIMA grafting and the use of early angiography possibly revealing “clinically silent LIMA graft dysfunction due to technical errors.”
The number of repeat revascularizations in the study was more in line with the SYNTAX study: 7% in the Aarhus University study and 6% in the SYNTAX CABG group. However, a meta-analysis of six studies with 1,190 patients reported 1-year repeat revascularization rates of 3.8% after a hybrid procedure and 1.4% after CABG (Am Heart J. 2014;167:585-92).
Ultimately, the safety and efficacy of the hybrid revascularization approach will require long-term follow-up data and head-to-head comparison with conventional CABG and PCI in clinical trials. “Meanwhile, LIMA patency, the cornerstone of surgical revascularization, may be used as a surrogate endpoint for long-term survival after HCR,” Dr. Modrau and coauthors said.
They reported having no disclosures.
A hybrid coronary revascularization procedure that combines off-pump left internal mammary artery (LIMA) grafting with percutaneous coronary intervention (PCI) showed good results at 1 year after surgery, but nonetheless showed a rate of adverse events that may raise questions about the procedure.
In a study published in the November issue of the Journal of Thoracic and Cardiovascular Surgery, a team of investigators from Aarhus University Hospital in Denmark reported high rates of graft patency and low rates of death and stroke with the procedure 1 year after a series of 100 operations (J Thorac Cardiovasc Surg. 2015;150:1181-6).
“The high left internal mammary artery graft patency rate and low risk of death and stroke at 1 year seem promising for the long-term outcome of this revascularization strategy,” said Dr. Ivy Susanne Modrau and colleagues.
The single-center study evaluated 1-year clinical and angiographic results of 100 consecutive trial patients with multivessel disease who had the hybrid procedure between October 2010 and February 2012. “The rationale of hybrid coronary revascularization is to achieve the survival benefits of the LIMA to LAD (left anterior descending artery) graft with reduced invasiveness to minimize postprocedural discomfort and morbidity, in particular the risk of stroke,” Dr. Modrau and colleagues said.
The study used the LIMA to LAD graft performed off-pump through a reversed J-hemisternotomy “We chose this technique because of its excellent exposure of the heart, technical ease, low risk of complicating chronic pain, and applicability in virtually all patients,” Dr. Modrau said. Eighty-nine patients had surgery prior to PCI and 11 had PCI prior to surgery.
The primary endpoint was rate of major adverse cardiac or cerebrovascular events (MACCE), the composite of all-cause death, stroke, myocardial infarction, and repeat revascularization by PCI or coronary artery bypass grafting at 1 year. Secondary endpoints included individual components and status of stent and graft patency on angiography.
Overall, 20 patients met the 1-year primary endpoint of MACCE. One patient died, one other had a stroke, and three had heart attacks. Sixteen patients had repeat revascularization procedures, eight performed during the index hospitalization. Graft patency was 98% after 1 year.
Dr. Modrau and coauthors noted the MACCE rate of 20% “was higher than expected,” and certainly higher than results in the SYNTAX study (17.8% in the PCI group and 12.4% in the coronary artery bypass grafting [CABG] group) (Euro. Intervention. 2015;10:e1-e6). One possible reason the Danish investigators cited for higher than expected MACCE rates was that they may be attributed to the learning curve involved with LIMA grafting and the use of early angiography possibly revealing “clinically silent LIMA graft dysfunction due to technical errors.”
The number of repeat revascularizations in the study was more in line with the SYNTAX study: 7% in the Aarhus University study and 6% in the SYNTAX CABG group. However, a meta-analysis of six studies with 1,190 patients reported 1-year repeat revascularization rates of 3.8% after a hybrid procedure and 1.4% after CABG (Am Heart J. 2014;167:585-92).
Ultimately, the safety and efficacy of the hybrid revascularization approach will require long-term follow-up data and head-to-head comparison with conventional CABG and PCI in clinical trials. “Meanwhile, LIMA patency, the cornerstone of surgical revascularization, may be used as a surrogate endpoint for long-term survival after HCR,” Dr. Modrau and coauthors said.
They reported having no disclosures.
FROM THE JOURNAL OF THORACIC AND CARDIOVASCULAR SURGERY
Key clinical point:High 1-year left internal mammary artery (LIMA) graft patency and low risk of death and stroke seem promising for long-term outcome after HCR.
Major finding: At 1 year, 98% of patients had patent LIMA grafts but the 20% rate of major adverse cardiac or cerebrovascular events was “higher than expected.”
Data source: Prospective single arm clinical feasibility study including 100 consecutive patients with multivessel disease undergoing staged hybrid coronary revascularization.
Disclosures: The study authors had no disclosures.
Stem cell benefits endure 3 years in infants
Infants with congenital heart disease, specifically left heart syndrome, who had cardiac stem-cell therapy after surgery showed improved cardiac and brain function after 3 years when compared with infants who had standard therapy, according to latest results from a clinical trial of progenitor cell infusion in infants.
The prospective, controlled study, published in the November issue of the Journal of Thoracic and Cardiovascular Surgery (2015;150[5]:1198-1208), involved 14 infants with hypoplastic left heart syndrome (HLHS). Dr. Suguru Tarui and colleagues at Okayama University Hospital in Japan infused seven infants with intracoronary cardiosphere-derived cells (CDCs) 1 month after they had two- or three-stage palliative surgery. Seven controls had standard care alone. The trial is known TICAP (Three-year follow-up of the Transcoronary Infusion of Cardiac Progenitor Cells in Patients With Single-Ventricle Physiology) (ClinicalTrials.gov ID: NCT01273857).
Infants born with HLHS are known to have poor prognoses. HLHS has been associated with the highest mortality of all congenital heart lesions (Semin Thorac Cardiovasc Surg Pediatr Card Surg Annu. 2015;18[1]:2-6).
The Okayama investigators conducted TICAP in 2011 and demonstrated the feasibility and safety of the intracoronary delivery of CDCs in infants with HLHS after staged procedures. The latest report looks at the secondary outcome of cardiac function through 36 months of follow-up, and is the first clinical trial to report on the mid-term results of cardiac progenitor therapy in congenital heart disease.
“CDC infusion could improve right ventricular function from 3 through 36 months of observation,” Dr. Tarui and colleagues said. “Patients treated by CDCs showed an increase in somatic growth and reduced heart failure status in mid-term follow-up.”
Upon entry into the study, all subjects were of similar age, body weight, and risk profiles. After 36 months, the CDC group showed no complications and significantly improved right ventricular injection fraction compared with controls. As a result, the CDC group had reduced brain natriuretic peptide levels, lower rates of unplanned catheterizations, and higher weight for age. No adverse events occurred in the CDC group, while two patients in the control group had complications (one developed heart failure, another developed enteropathy).
The investigators looked at predictors of cardiac functional efficacy in the CDC group and determined that younger age was associated with greater improvement of right ventricular ejection fraction, as measured on echocardiography. The lower weight-for-age Z score and reduced ejection fraction at the time of infusion may be predictors of cardiac function improvements at 3 years.
“This therapeutic strategy may merit somatic growth enhancement and reduce the incidence of heart failure,” Dr. Tarui and coauthors said.
They noted a number of limitations of their small clinical trial. The study was nonrandomized, and the cardiac interventions were not blinded, among others. “Larger phase II studies focusing on changes in cardiac function, myocardial fibrosis, and quality of life and clinical event rates are warranted to confirm these effects of CDC administration in patients with single ventricular physiology,” they said.
The government of Japan through its ministries of Health, Labor and Welfare, and Education, Culture Sports, Science and Technology provided funding for the study, as did the Research Foundation of Okayama University Hospital. The authors had no disclosures.
The benefits of stem cell treatments for cardiac function beyond the initial follow-up period have “remained an unanswered question,” Dr. Sunjay Kaushal of University of Maryland, Baltimore, said in his invited commentary (J Thorac Cardiovasc Surg. 2015;150[5]:1209-11). “The 3-year follow-up data from the TICAP trial, therefore, offers one of the first opportunities to examine the durability of the outcomes found in stem cell-treated patients,” Dr. Kaushal said.
While TICAP provides clues into independent predictors of success with stem cell treatments, the small study population of seven patients makes it difficult to confirm those predictors, he said. “Multivariate analysis of results from future trials with larger sample size will be needed to verify these preliminary findings,” he noted.
Nonetheless, the study adds to the emerging evidence that younger, sicker patients may respond better to stem cell infusion, a concept Dr. Kaushal termed “intriguing.”
“Meanwhile, the TICAP trial and its three-year follow-up should garner enthusiasm for stem cell therapy, and establish the basis for non-ischemic ventricular dysfunction in pediatric patients as an emerging indication for stem cell therapy,” he said.
The benefits of stem cell treatments for cardiac function beyond the initial follow-up period have “remained an unanswered question,” Dr. Sunjay Kaushal of University of Maryland, Baltimore, said in his invited commentary (J Thorac Cardiovasc Surg. 2015;150[5]:1209-11). “The 3-year follow-up data from the TICAP trial, therefore, offers one of the first opportunities to examine the durability of the outcomes found in stem cell-treated patients,” Dr. Kaushal said.
While TICAP provides clues into independent predictors of success with stem cell treatments, the small study population of seven patients makes it difficult to confirm those predictors, he said. “Multivariate analysis of results from future trials with larger sample size will be needed to verify these preliminary findings,” he noted.
Nonetheless, the study adds to the emerging evidence that younger, sicker patients may respond better to stem cell infusion, a concept Dr. Kaushal termed “intriguing.”
“Meanwhile, the TICAP trial and its three-year follow-up should garner enthusiasm for stem cell therapy, and establish the basis for non-ischemic ventricular dysfunction in pediatric patients as an emerging indication for stem cell therapy,” he said.
The benefits of stem cell treatments for cardiac function beyond the initial follow-up period have “remained an unanswered question,” Dr. Sunjay Kaushal of University of Maryland, Baltimore, said in his invited commentary (J Thorac Cardiovasc Surg. 2015;150[5]:1209-11). “The 3-year follow-up data from the TICAP trial, therefore, offers one of the first opportunities to examine the durability of the outcomes found in stem cell-treated patients,” Dr. Kaushal said.
While TICAP provides clues into independent predictors of success with stem cell treatments, the small study population of seven patients makes it difficult to confirm those predictors, he said. “Multivariate analysis of results from future trials with larger sample size will be needed to verify these preliminary findings,” he noted.
Nonetheless, the study adds to the emerging evidence that younger, sicker patients may respond better to stem cell infusion, a concept Dr. Kaushal termed “intriguing.”
“Meanwhile, the TICAP trial and its three-year follow-up should garner enthusiasm for stem cell therapy, and establish the basis for non-ischemic ventricular dysfunction in pediatric patients as an emerging indication for stem cell therapy,” he said.
Infants with congenital heart disease, specifically left heart syndrome, who had cardiac stem-cell therapy after surgery showed improved cardiac and brain function after 3 years when compared with infants who had standard therapy, according to latest results from a clinical trial of progenitor cell infusion in infants.
The prospective, controlled study, published in the November issue of the Journal of Thoracic and Cardiovascular Surgery (2015;150[5]:1198-1208), involved 14 infants with hypoplastic left heart syndrome (HLHS). Dr. Suguru Tarui and colleagues at Okayama University Hospital in Japan infused seven infants with intracoronary cardiosphere-derived cells (CDCs) 1 month after they had two- or three-stage palliative surgery. Seven controls had standard care alone. The trial is known TICAP (Three-year follow-up of the Transcoronary Infusion of Cardiac Progenitor Cells in Patients With Single-Ventricle Physiology) (ClinicalTrials.gov ID: NCT01273857).
Infants born with HLHS are known to have poor prognoses. HLHS has been associated with the highest mortality of all congenital heart lesions (Semin Thorac Cardiovasc Surg Pediatr Card Surg Annu. 2015;18[1]:2-6).
The Okayama investigators conducted TICAP in 2011 and demonstrated the feasibility and safety of the intracoronary delivery of CDCs in infants with HLHS after staged procedures. The latest report looks at the secondary outcome of cardiac function through 36 months of follow-up, and is the first clinical trial to report on the mid-term results of cardiac progenitor therapy in congenital heart disease.
“CDC infusion could improve right ventricular function from 3 through 36 months of observation,” Dr. Tarui and colleagues said. “Patients treated by CDCs showed an increase in somatic growth and reduced heart failure status in mid-term follow-up.”
Upon entry into the study, all subjects were of similar age, body weight, and risk profiles. After 36 months, the CDC group showed no complications and significantly improved right ventricular injection fraction compared with controls. As a result, the CDC group had reduced brain natriuretic peptide levels, lower rates of unplanned catheterizations, and higher weight for age. No adverse events occurred in the CDC group, while two patients in the control group had complications (one developed heart failure, another developed enteropathy).
The investigators looked at predictors of cardiac functional efficacy in the CDC group and determined that younger age was associated with greater improvement of right ventricular ejection fraction, as measured on echocardiography. The lower weight-for-age Z score and reduced ejection fraction at the time of infusion may be predictors of cardiac function improvements at 3 years.
“This therapeutic strategy may merit somatic growth enhancement and reduce the incidence of heart failure,” Dr. Tarui and coauthors said.
They noted a number of limitations of their small clinical trial. The study was nonrandomized, and the cardiac interventions were not blinded, among others. “Larger phase II studies focusing on changes in cardiac function, myocardial fibrosis, and quality of life and clinical event rates are warranted to confirm these effects of CDC administration in patients with single ventricular physiology,” they said.
The government of Japan through its ministries of Health, Labor and Welfare, and Education, Culture Sports, Science and Technology provided funding for the study, as did the Research Foundation of Okayama University Hospital. The authors had no disclosures.
Infants with congenital heart disease, specifically left heart syndrome, who had cardiac stem-cell therapy after surgery showed improved cardiac and brain function after 3 years when compared with infants who had standard therapy, according to latest results from a clinical trial of progenitor cell infusion in infants.
The prospective, controlled study, published in the November issue of the Journal of Thoracic and Cardiovascular Surgery (2015;150[5]:1198-1208), involved 14 infants with hypoplastic left heart syndrome (HLHS). Dr. Suguru Tarui and colleagues at Okayama University Hospital in Japan infused seven infants with intracoronary cardiosphere-derived cells (CDCs) 1 month after they had two- or three-stage palliative surgery. Seven controls had standard care alone. The trial is known TICAP (Three-year follow-up of the Transcoronary Infusion of Cardiac Progenitor Cells in Patients With Single-Ventricle Physiology) (ClinicalTrials.gov ID: NCT01273857).
Infants born with HLHS are known to have poor prognoses. HLHS has been associated with the highest mortality of all congenital heart lesions (Semin Thorac Cardiovasc Surg Pediatr Card Surg Annu. 2015;18[1]:2-6).
The Okayama investigators conducted TICAP in 2011 and demonstrated the feasibility and safety of the intracoronary delivery of CDCs in infants with HLHS after staged procedures. The latest report looks at the secondary outcome of cardiac function through 36 months of follow-up, and is the first clinical trial to report on the mid-term results of cardiac progenitor therapy in congenital heart disease.
“CDC infusion could improve right ventricular function from 3 through 36 months of observation,” Dr. Tarui and colleagues said. “Patients treated by CDCs showed an increase in somatic growth and reduced heart failure status in mid-term follow-up.”
Upon entry into the study, all subjects were of similar age, body weight, and risk profiles. After 36 months, the CDC group showed no complications and significantly improved right ventricular injection fraction compared with controls. As a result, the CDC group had reduced brain natriuretic peptide levels, lower rates of unplanned catheterizations, and higher weight for age. No adverse events occurred in the CDC group, while two patients in the control group had complications (one developed heart failure, another developed enteropathy).
The investigators looked at predictors of cardiac functional efficacy in the CDC group and determined that younger age was associated with greater improvement of right ventricular ejection fraction, as measured on echocardiography. The lower weight-for-age Z score and reduced ejection fraction at the time of infusion may be predictors of cardiac function improvements at 3 years.
“This therapeutic strategy may merit somatic growth enhancement and reduce the incidence of heart failure,” Dr. Tarui and coauthors said.
They noted a number of limitations of their small clinical trial. The study was nonrandomized, and the cardiac interventions were not blinded, among others. “Larger phase II studies focusing on changes in cardiac function, myocardial fibrosis, and quality of life and clinical event rates are warranted to confirm these effects of CDC administration in patients with single ventricular physiology,” they said.
The government of Japan through its ministries of Health, Labor and Welfare, and Education, Culture Sports, Science and Technology provided funding for the study, as did the Research Foundation of Okayama University Hospital. The authors had no disclosures.
FROM JOURNAL OF THORACIC AND CARDIOVASCULAR SURGERY
Key clinical point: This is the first clinical trial to report the midterm safety and efficacy of cardiac progenitor therapy in congenital heart disease.
Major finding: After 36 months, infants who received stem cells for congenital heart failure had better outcomes than did infants who had standard treatment.
Data source: Prospective clinical trial of 14 infants enrolled in the Transcoronary Infusion of Cardiac Progenitor Cells in Hypoplastic Left Heat Syndrome (TICAP) trial, divided evenly between treatment and control groups.
Disclosures: The government of Japan through its ministries of Health, Labor and Welfare, and Education, Culture Sports, Science and Technology provided funding for the study, as did the Research Foundation of Okayama University Hospital. The authors had no disclosures.
‘Minimalist’ TAVR has short learning curve
As a “minimalist” approach to transcatheter aortic valve replacement – known as MA-TAVR – gains in popularity at high-volume centers, questions persist about the surgeon’s learning curve. A small series of MA-TAVR cases at Emory University in Atlanta has shown that the leaning curve may be like the TAVR approach itself: minimal.
Dr. Hanna Jensen and her associates reported on 151 consecutive patients who had MA-TAVR in the October issue of the Journal of Thoracic and Cardiovascular Surgery (J Thorac Cardiovasc Surg. 2015. doi: 10.1016/j.jtcvs.2015.07.078). They previously reported their findings at the annual meeting of the American Association for Thoracic Surgery in April in Seattle.
This study builds on an Emory study last year that reported the minimalist approach to TAVR cost about $10,000 less per patient than the standard transfemoral approach (JACC Cardiovasc Interv. 2014;7:898-904).
The operation the study authors evaluated is performed in the catheterization laboratory rather than the operating room, as in traditional TAVR. Both approaches use a femoral approach, but where traditional TAVR requires general anesthesia and transesophageal echocardiography (TEE), MA-TAVR uses local anesthesia, minimal conscious sedation, and transthoracic echocardiography (TTE).
The study authors acknowledged concerns that TTE may underestimate the severity of paravalvular leak after the procedure when compared with TEE. Their protocol relies on preoperative TTE and CT scans, or three-dimensional TEE if the case warrants it, to ensure optimal sizing of the transcatheter valve before the operation. “If any concerns arise, our threshold is low to perform intraoperative balloon-sizing,” Dr. Jensen and her coauthors said. They also use TTE, along with a root-angiogram after valve deployment, and invasively measure the aortic regurgitation index before and after deployment.
Most study patients were high-risk surgical candidates with a median Society of Thoracic Surgeons Predicted Risk of Mortality (STS PROM) score of 10%. The overall major stroke rate was 3.3%, while major vascular complications occurred in 3% of patients and the greater-than-mild paravalvular leak rate was 7%.
The study retrospectively evaluated 151 consecutive patients who were divided into three groups at different time points: May 2012 to January 2013, February to August 2013, and September 2013 to July 2014. Complications were similar among all three groups, but the third group had shorter hospital stays and less time in the intensive care unit (ICU).
The first group received only the first-generation SAPIEN valve system; use of the second-generation SAPIEN XT valve increased in latter two groups. The SAPIEN XT valve is available in 23, 26, or 29 mm, but the 29-mm size was not available in the first-generation SAPIEN implant.
A subgroup analysis looked at patients who were discharged within 48 hours of the operation or more than 48 hours afterward. The early-discharge patients had lower STS PROM scores (8.3% vs. 10.3%) and lower rates of diabetes (31% vs. 49%). They also had less need for postoperative pacemakers and less frequent rehospitalization. “This implies that in selected MA-TAVR patients early discharge is feasible and safe, but larger studies are required to identify the optimal profile of patients who can be sent home within the first two postoperative days,” Dr. Jensen and her colleagues said.
Early in the MA-TAVR protocol all patients were sent to the ICU. As the care team gained more experience with the procedure, the protocol changed to send all patients to a regular telemetry floor after surgery unless they had vascular issues or potential need for a pacemaker. The decreasing need for ICU “was the only indication of an institutional learning curve that was discovered, and demonstrated improved resource utilization over time,” the investigators said.
They encouraged other centers to pursue MA-TAVR. “As experience grows, we believe that this procedure can be done with less or no ICU support leading to a shorter hospital stay and improved resource utilization,” Dr. Jensen and her coauthors concluded. They called for further studies to determine the characteristics that make a patient most suitable for a short-admission MA-TAVR procedure.
Study coauthors Dr. Vasilis Babaliaros, Dr. Vinod Thourani, Amy Simone, and Patricia Keegan are research consultants with Edwards Lifesciences. The rest of the authors had no disclosures.
Calling this report an “early milestone in the relentless simplification” of transcatheter aortic valve replacement (TAVR), Dr. Craig Smith of Columbia University Medical Center/New York Presbyterian Hospital, wrote in his invited commentary that it nonetheless leaves a few questions unanswered – and may leave surgeons seeing their role in TAVR marginalized as the procedure moves from the operating room to the catheterization lab (J Thorac Cardiovasc Surg. 2015. doi: 10.1016/j.jtcvs.2015.07.082). .

|
Dr. Craig Smith |
One unanswered question revolves around the use of conscious sedation and transthoracic echocardiography (TTE) for the minimalist approach (MA), rather than general anesthesia and transesophageal echocardiography (TEE) of the traditional transfemoral approach. “MA requires reliance on [TTE] for assessment of paravalvular leak, and since TTE can’t be compared to TEE in the same patients and still be MA, the merits of this trade-off cannot be assessed in this population,” he said.
Further, he said that the study data do not conclusively link MA to early discharge because the early discharge patients had lower Society of Thoracic Surgery scores.
Another important unanswered question is whether endocarditis is more frequent in TAVR when it’s performed outside the operating room.
“While I suspect the answer will be ‘yes,’ this question will be left dangling until large numbers have been done in hybrid cath labs, because the frequency will be low, and because the forces propelling a ‘cath lab’ alternative to surgical or transcatheter valve replacement done in an operating room will be too powerful to retard on a hunch,” Dr. Smith wrote. “What will the departure of TAVR from operating rooms mean for the role of the surgeon? That is for surgeons to determine. Stay involved, or say goodbye!”
Calling this report an “early milestone in the relentless simplification” of transcatheter aortic valve replacement (TAVR), Dr. Craig Smith of Columbia University Medical Center/New York Presbyterian Hospital, wrote in his invited commentary that it nonetheless leaves a few questions unanswered – and may leave surgeons seeing their role in TAVR marginalized as the procedure moves from the operating room to the catheterization lab (J Thorac Cardiovasc Surg. 2015. doi: 10.1016/j.jtcvs.2015.07.082). .

|
Dr. Craig Smith |
One unanswered question revolves around the use of conscious sedation and transthoracic echocardiography (TTE) for the minimalist approach (MA), rather than general anesthesia and transesophageal echocardiography (TEE) of the traditional transfemoral approach. “MA requires reliance on [TTE] for assessment of paravalvular leak, and since TTE can’t be compared to TEE in the same patients and still be MA, the merits of this trade-off cannot be assessed in this population,” he said.
Further, he said that the study data do not conclusively link MA to early discharge because the early discharge patients had lower Society of Thoracic Surgery scores.
Another important unanswered question is whether endocarditis is more frequent in TAVR when it’s performed outside the operating room.
“While I suspect the answer will be ‘yes,’ this question will be left dangling until large numbers have been done in hybrid cath labs, because the frequency will be low, and because the forces propelling a ‘cath lab’ alternative to surgical or transcatheter valve replacement done in an operating room will be too powerful to retard on a hunch,” Dr. Smith wrote. “What will the departure of TAVR from operating rooms mean for the role of the surgeon? That is for surgeons to determine. Stay involved, or say goodbye!”
Calling this report an “early milestone in the relentless simplification” of transcatheter aortic valve replacement (TAVR), Dr. Craig Smith of Columbia University Medical Center/New York Presbyterian Hospital, wrote in his invited commentary that it nonetheless leaves a few questions unanswered – and may leave surgeons seeing their role in TAVR marginalized as the procedure moves from the operating room to the catheterization lab (J Thorac Cardiovasc Surg. 2015. doi: 10.1016/j.jtcvs.2015.07.082). .

|
Dr. Craig Smith |
One unanswered question revolves around the use of conscious sedation and transthoracic echocardiography (TTE) for the minimalist approach (MA), rather than general anesthesia and transesophageal echocardiography (TEE) of the traditional transfemoral approach. “MA requires reliance on [TTE] for assessment of paravalvular leak, and since TTE can’t be compared to TEE in the same patients and still be MA, the merits of this trade-off cannot be assessed in this population,” he said.
Further, he said that the study data do not conclusively link MA to early discharge because the early discharge patients had lower Society of Thoracic Surgery scores.
Another important unanswered question is whether endocarditis is more frequent in TAVR when it’s performed outside the operating room.
“While I suspect the answer will be ‘yes,’ this question will be left dangling until large numbers have been done in hybrid cath labs, because the frequency will be low, and because the forces propelling a ‘cath lab’ alternative to surgical or transcatheter valve replacement done in an operating room will be too powerful to retard on a hunch,” Dr. Smith wrote. “What will the departure of TAVR from operating rooms mean for the role of the surgeon? That is for surgeons to determine. Stay involved, or say goodbye!”
As a “minimalist” approach to transcatheter aortic valve replacement – known as MA-TAVR – gains in popularity at high-volume centers, questions persist about the surgeon’s learning curve. A small series of MA-TAVR cases at Emory University in Atlanta has shown that the leaning curve may be like the TAVR approach itself: minimal.
Dr. Hanna Jensen and her associates reported on 151 consecutive patients who had MA-TAVR in the October issue of the Journal of Thoracic and Cardiovascular Surgery (J Thorac Cardiovasc Surg. 2015. doi: 10.1016/j.jtcvs.2015.07.078). They previously reported their findings at the annual meeting of the American Association for Thoracic Surgery in April in Seattle.
This study builds on an Emory study last year that reported the minimalist approach to TAVR cost about $10,000 less per patient than the standard transfemoral approach (JACC Cardiovasc Interv. 2014;7:898-904).
The operation the study authors evaluated is performed in the catheterization laboratory rather than the operating room, as in traditional TAVR. Both approaches use a femoral approach, but where traditional TAVR requires general anesthesia and transesophageal echocardiography (TEE), MA-TAVR uses local anesthesia, minimal conscious sedation, and transthoracic echocardiography (TTE).
The study authors acknowledged concerns that TTE may underestimate the severity of paravalvular leak after the procedure when compared with TEE. Their protocol relies on preoperative TTE and CT scans, or three-dimensional TEE if the case warrants it, to ensure optimal sizing of the transcatheter valve before the operation. “If any concerns arise, our threshold is low to perform intraoperative balloon-sizing,” Dr. Jensen and her coauthors said. They also use TTE, along with a root-angiogram after valve deployment, and invasively measure the aortic regurgitation index before and after deployment.
Most study patients were high-risk surgical candidates with a median Society of Thoracic Surgeons Predicted Risk of Mortality (STS PROM) score of 10%. The overall major stroke rate was 3.3%, while major vascular complications occurred in 3% of patients and the greater-than-mild paravalvular leak rate was 7%.
The study retrospectively evaluated 151 consecutive patients who were divided into three groups at different time points: May 2012 to January 2013, February to August 2013, and September 2013 to July 2014. Complications were similar among all three groups, but the third group had shorter hospital stays and less time in the intensive care unit (ICU).
The first group received only the first-generation SAPIEN valve system; use of the second-generation SAPIEN XT valve increased in latter two groups. The SAPIEN XT valve is available in 23, 26, or 29 mm, but the 29-mm size was not available in the first-generation SAPIEN implant.
A subgroup analysis looked at patients who were discharged within 48 hours of the operation or more than 48 hours afterward. The early-discharge patients had lower STS PROM scores (8.3% vs. 10.3%) and lower rates of diabetes (31% vs. 49%). They also had less need for postoperative pacemakers and less frequent rehospitalization. “This implies that in selected MA-TAVR patients early discharge is feasible and safe, but larger studies are required to identify the optimal profile of patients who can be sent home within the first two postoperative days,” Dr. Jensen and her colleagues said.
Early in the MA-TAVR protocol all patients were sent to the ICU. As the care team gained more experience with the procedure, the protocol changed to send all patients to a regular telemetry floor after surgery unless they had vascular issues or potential need for a pacemaker. The decreasing need for ICU “was the only indication of an institutional learning curve that was discovered, and demonstrated improved resource utilization over time,” the investigators said.
They encouraged other centers to pursue MA-TAVR. “As experience grows, we believe that this procedure can be done with less or no ICU support leading to a shorter hospital stay and improved resource utilization,” Dr. Jensen and her coauthors concluded. They called for further studies to determine the characteristics that make a patient most suitable for a short-admission MA-TAVR procedure.
Study coauthors Dr. Vasilis Babaliaros, Dr. Vinod Thourani, Amy Simone, and Patricia Keegan are research consultants with Edwards Lifesciences. The rest of the authors had no disclosures.
As a “minimalist” approach to transcatheter aortic valve replacement – known as MA-TAVR – gains in popularity at high-volume centers, questions persist about the surgeon’s learning curve. A small series of MA-TAVR cases at Emory University in Atlanta has shown that the leaning curve may be like the TAVR approach itself: minimal.
Dr. Hanna Jensen and her associates reported on 151 consecutive patients who had MA-TAVR in the October issue of the Journal of Thoracic and Cardiovascular Surgery (J Thorac Cardiovasc Surg. 2015. doi: 10.1016/j.jtcvs.2015.07.078). They previously reported their findings at the annual meeting of the American Association for Thoracic Surgery in April in Seattle.
This study builds on an Emory study last year that reported the minimalist approach to TAVR cost about $10,000 less per patient than the standard transfemoral approach (JACC Cardiovasc Interv. 2014;7:898-904).
The operation the study authors evaluated is performed in the catheterization laboratory rather than the operating room, as in traditional TAVR. Both approaches use a femoral approach, but where traditional TAVR requires general anesthesia and transesophageal echocardiography (TEE), MA-TAVR uses local anesthesia, minimal conscious sedation, and transthoracic echocardiography (TTE).
The study authors acknowledged concerns that TTE may underestimate the severity of paravalvular leak after the procedure when compared with TEE. Their protocol relies on preoperative TTE and CT scans, or three-dimensional TEE if the case warrants it, to ensure optimal sizing of the transcatheter valve before the operation. “If any concerns arise, our threshold is low to perform intraoperative balloon-sizing,” Dr. Jensen and her coauthors said. They also use TTE, along with a root-angiogram after valve deployment, and invasively measure the aortic regurgitation index before and after deployment.
Most study patients were high-risk surgical candidates with a median Society of Thoracic Surgeons Predicted Risk of Mortality (STS PROM) score of 10%. The overall major stroke rate was 3.3%, while major vascular complications occurred in 3% of patients and the greater-than-mild paravalvular leak rate was 7%.
The study retrospectively evaluated 151 consecutive patients who were divided into three groups at different time points: May 2012 to January 2013, February to August 2013, and September 2013 to July 2014. Complications were similar among all three groups, but the third group had shorter hospital stays and less time in the intensive care unit (ICU).
The first group received only the first-generation SAPIEN valve system; use of the second-generation SAPIEN XT valve increased in latter two groups. The SAPIEN XT valve is available in 23, 26, or 29 mm, but the 29-mm size was not available in the first-generation SAPIEN implant.
A subgroup analysis looked at patients who were discharged within 48 hours of the operation or more than 48 hours afterward. The early-discharge patients had lower STS PROM scores (8.3% vs. 10.3%) and lower rates of diabetes (31% vs. 49%). They also had less need for postoperative pacemakers and less frequent rehospitalization. “This implies that in selected MA-TAVR patients early discharge is feasible and safe, but larger studies are required to identify the optimal profile of patients who can be sent home within the first two postoperative days,” Dr. Jensen and her colleagues said.
Early in the MA-TAVR protocol all patients were sent to the ICU. As the care team gained more experience with the procedure, the protocol changed to send all patients to a regular telemetry floor after surgery unless they had vascular issues or potential need for a pacemaker. The decreasing need for ICU “was the only indication of an institutional learning curve that was discovered, and demonstrated improved resource utilization over time,” the investigators said.
They encouraged other centers to pursue MA-TAVR. “As experience grows, we believe that this procedure can be done with less or no ICU support leading to a shorter hospital stay and improved resource utilization,” Dr. Jensen and her coauthors concluded. They called for further studies to determine the characteristics that make a patient most suitable for a short-admission MA-TAVR procedure.
Study coauthors Dr. Vasilis Babaliaros, Dr. Vinod Thourani, Amy Simone, and Patricia Keegan are research consultants with Edwards Lifesciences. The rest of the authors had no disclosures.
FROM THE JOURNAL OF THORACIC AND CARDIOVASCULAR SURGERY
Key clinical point: A minimalist approach to transcatheter aortic valve replacement (MA-TAVR) is feasible with acceptable outcomes.
Major finding: Transition to MA-TAVR in a high-volume center had a relatively small learning curve.
Data source: A review of 151 consecutive patients who had MA-TAVR at Emory University between May 2012 and July 2014.
Disclosures: Study coauthors Dr. Vasilis Babaliaros, Dr. Vinod Thourani, Amy Simone, and Patricia Keegan are research consultants with Edwards Lifesciences. The rest of the authors had no disclosures.
Mentoring that takes it up a notch
Everything seems to be extreme nowadays – “Extreme Makeover: Home Edition,” “Extreme Weight Loss,” even “Extreme Fishing” and “Extreme Couponing” – so it was only a matter of time that extreme came to cardiothoracic surgery.
Dr. Michael K. Pasque of Washington University in St. Louis explored “Extreme Mentoring in Cardiothoracic Surgery” in his commentary published online ahead of print for the October issue of the Journal of Thoracic and Cardiovascular Surgery (J Thorac Cardiovasc Surg. 2015 doi: 10.1016/j.jtcvs.2015.04.056).
Meaningful mentoring “carries with it considerable responsibility. Extreme mentoring comes only at a price – it is going to cost us,” Dr. Pasque wrote, calling on academic cardiothoracic surgical mentors to perform a self-appraisal of their commitment and mentoring skills. He even developed a self-appraisal checklist that involves 37 different markers in four different categories: general; goals, pathways, and meetings; milestone timelines and taking action; and clinical assistance.
The first step in extreme mentoring for the academic cardiothoracic surgeon is to focus exclusively on the mentee. “As cardiothoracic surgeons, we are used to having the attention focused on us,” Dr. Pasque noted, but mentoring is different: the “energy of our relationship” must focus on the mentee.
The next step involves an objective assessment of the mentee. “If we are to throw our support wholeheartedly behind our mentee, we must genuinely believe in them,” he said. This assessment leads to setting goals for the mentee. “My formula is to honestly estimate the surgical, research, teaching and academic life goals that are both desired by and within reach of our mentee – and then double them,” he said. “We must set very aggressive goals for our mentee.”
Achieving those goals involves directing mentees to the right pathway and then helping them stay on that pathway despite obstacles. “When their progress through these barriers is discussed – and that should be often – then ours should be the voice that reminds them that despite the momentary setbacks, the goals we have set are going to happen,” Dr. Pasque said.
The process involves frequent “and substantive” meetings between mentor and mentee and establishing timelines for achieving milestones and goals. The mentor must back up what happens in those meetings with action – both overt, like assisting them in surgery or introducing them to influential colleagues, and covert in ways the mentee may never know about.
One “clandestine” operation involves the mentor keeping an updated list of 10 individuals who have the most to offer the mentee, “especially in areas in which we have limited or no influence,” and habitually following up with them. The mentor must be willing to “pick a fight” so the mentee doesn’t get left behind on call while senior colleagues attend meetings.
“We must be the senior voice that speaks up for them,” Dr. Pasque wrote. “They need to attend these meetings, even if it is we who must stay behind in their place.”
The mentoring process involves being across the operating room table from them at key milestones in their surgical development and being on-call 24/7 for the mentee. That may seem like extreme handholding to some critics, but Dr. Pasque said that letting a patient suffer or die is inexcusable. “Our first priority is always the patient’s well-being.”
The mentor must show respect to the mentee and practice “extreme encouragement,” especially in the operating room. “There is something magical about being told you are a good surgeon,” he said. “You become one.” This isn’t about falsely building up the mentee, but instilling the confidence to act on the patient’s behalf. The mentee will face enough doubters. “We must be the voice that assures them otherwise,” he said.
Teaching leadership also is key for the mentor. Mentors teach leadership by modeling it. “The best leaders are always those who place the needs of others above their own,” Dr. Pasque pointed out, harkening back to putting the focus on the mentee. “We can’t teach them to put the needs of others above their own without putting their needs about ours.”
Ultimately, the mentor’s greatest desire should be that the mentee exceeds them, “that they make everyone forget about us,” Dr. Pasque said. That would be the “crowning achievement” that would make the mentor “most unforgettable.”
Dr. Pasque had no disclosures.
Everything seems to be extreme nowadays – “Extreme Makeover: Home Edition,” “Extreme Weight Loss,” even “Extreme Fishing” and “Extreme Couponing” – so it was only a matter of time that extreme came to cardiothoracic surgery.
Dr. Michael K. Pasque of Washington University in St. Louis explored “Extreme Mentoring in Cardiothoracic Surgery” in his commentary published online ahead of print for the October issue of the Journal of Thoracic and Cardiovascular Surgery (J Thorac Cardiovasc Surg. 2015 doi: 10.1016/j.jtcvs.2015.04.056).
Meaningful mentoring “carries with it considerable responsibility. Extreme mentoring comes only at a price – it is going to cost us,” Dr. Pasque wrote, calling on academic cardiothoracic surgical mentors to perform a self-appraisal of their commitment and mentoring skills. He even developed a self-appraisal checklist that involves 37 different markers in four different categories: general; goals, pathways, and meetings; milestone timelines and taking action; and clinical assistance.
The first step in extreme mentoring for the academic cardiothoracic surgeon is to focus exclusively on the mentee. “As cardiothoracic surgeons, we are used to having the attention focused on us,” Dr. Pasque noted, but mentoring is different: the “energy of our relationship” must focus on the mentee.
The next step involves an objective assessment of the mentee. “If we are to throw our support wholeheartedly behind our mentee, we must genuinely believe in them,” he said. This assessment leads to setting goals for the mentee. “My formula is to honestly estimate the surgical, research, teaching and academic life goals that are both desired by and within reach of our mentee – and then double them,” he said. “We must set very aggressive goals for our mentee.”
Achieving those goals involves directing mentees to the right pathway and then helping them stay on that pathway despite obstacles. “When their progress through these barriers is discussed – and that should be often – then ours should be the voice that reminds them that despite the momentary setbacks, the goals we have set are going to happen,” Dr. Pasque said.
The process involves frequent “and substantive” meetings between mentor and mentee and establishing timelines for achieving milestones and goals. The mentor must back up what happens in those meetings with action – both overt, like assisting them in surgery or introducing them to influential colleagues, and covert in ways the mentee may never know about.
One “clandestine” operation involves the mentor keeping an updated list of 10 individuals who have the most to offer the mentee, “especially in areas in which we have limited or no influence,” and habitually following up with them. The mentor must be willing to “pick a fight” so the mentee doesn’t get left behind on call while senior colleagues attend meetings.
“We must be the senior voice that speaks up for them,” Dr. Pasque wrote. “They need to attend these meetings, even if it is we who must stay behind in their place.”
The mentoring process involves being across the operating room table from them at key milestones in their surgical development and being on-call 24/7 for the mentee. That may seem like extreme handholding to some critics, but Dr. Pasque said that letting a patient suffer or die is inexcusable. “Our first priority is always the patient’s well-being.”
The mentor must show respect to the mentee and practice “extreme encouragement,” especially in the operating room. “There is something magical about being told you are a good surgeon,” he said. “You become one.” This isn’t about falsely building up the mentee, but instilling the confidence to act on the patient’s behalf. The mentee will face enough doubters. “We must be the voice that assures them otherwise,” he said.
Teaching leadership also is key for the mentor. Mentors teach leadership by modeling it. “The best leaders are always those who place the needs of others above their own,” Dr. Pasque pointed out, harkening back to putting the focus on the mentee. “We can’t teach them to put the needs of others above their own without putting their needs about ours.”
Ultimately, the mentor’s greatest desire should be that the mentee exceeds them, “that they make everyone forget about us,” Dr. Pasque said. That would be the “crowning achievement” that would make the mentor “most unforgettable.”
Dr. Pasque had no disclosures.
Everything seems to be extreme nowadays – “Extreme Makeover: Home Edition,” “Extreme Weight Loss,” even “Extreme Fishing” and “Extreme Couponing” – so it was only a matter of time that extreme came to cardiothoracic surgery.
Dr. Michael K. Pasque of Washington University in St. Louis explored “Extreme Mentoring in Cardiothoracic Surgery” in his commentary published online ahead of print for the October issue of the Journal of Thoracic and Cardiovascular Surgery (J Thorac Cardiovasc Surg. 2015 doi: 10.1016/j.jtcvs.2015.04.056).
Meaningful mentoring “carries with it considerable responsibility. Extreme mentoring comes only at a price – it is going to cost us,” Dr. Pasque wrote, calling on academic cardiothoracic surgical mentors to perform a self-appraisal of their commitment and mentoring skills. He even developed a self-appraisal checklist that involves 37 different markers in four different categories: general; goals, pathways, and meetings; milestone timelines and taking action; and clinical assistance.
The first step in extreme mentoring for the academic cardiothoracic surgeon is to focus exclusively on the mentee. “As cardiothoracic surgeons, we are used to having the attention focused on us,” Dr. Pasque noted, but mentoring is different: the “energy of our relationship” must focus on the mentee.
The next step involves an objective assessment of the mentee. “If we are to throw our support wholeheartedly behind our mentee, we must genuinely believe in them,” he said. This assessment leads to setting goals for the mentee. “My formula is to honestly estimate the surgical, research, teaching and academic life goals that are both desired by and within reach of our mentee – and then double them,” he said. “We must set very aggressive goals for our mentee.”
Achieving those goals involves directing mentees to the right pathway and then helping them stay on that pathway despite obstacles. “When their progress through these barriers is discussed – and that should be often – then ours should be the voice that reminds them that despite the momentary setbacks, the goals we have set are going to happen,” Dr. Pasque said.
The process involves frequent “and substantive” meetings between mentor and mentee and establishing timelines for achieving milestones and goals. The mentor must back up what happens in those meetings with action – both overt, like assisting them in surgery or introducing them to influential colleagues, and covert in ways the mentee may never know about.
One “clandestine” operation involves the mentor keeping an updated list of 10 individuals who have the most to offer the mentee, “especially in areas in which we have limited or no influence,” and habitually following up with them. The mentor must be willing to “pick a fight” so the mentee doesn’t get left behind on call while senior colleagues attend meetings.
“We must be the senior voice that speaks up for them,” Dr. Pasque wrote. “They need to attend these meetings, even if it is we who must stay behind in their place.”
The mentoring process involves being across the operating room table from them at key milestones in their surgical development and being on-call 24/7 for the mentee. That may seem like extreme handholding to some critics, but Dr. Pasque said that letting a patient suffer or die is inexcusable. “Our first priority is always the patient’s well-being.”
The mentor must show respect to the mentee and practice “extreme encouragement,” especially in the operating room. “There is something magical about being told you are a good surgeon,” he said. “You become one.” This isn’t about falsely building up the mentee, but instilling the confidence to act on the patient’s behalf. The mentee will face enough doubters. “We must be the voice that assures them otherwise,” he said.
Teaching leadership also is key for the mentor. Mentors teach leadership by modeling it. “The best leaders are always those who place the needs of others above their own,” Dr. Pasque pointed out, harkening back to putting the focus on the mentee. “We can’t teach them to put the needs of others above their own without putting their needs about ours.”
Ultimately, the mentor’s greatest desire should be that the mentee exceeds them, “that they make everyone forget about us,” Dr. Pasque said. That would be the “crowning achievement” that would make the mentor “most unforgettable.”
Dr. Pasque had no disclosures.