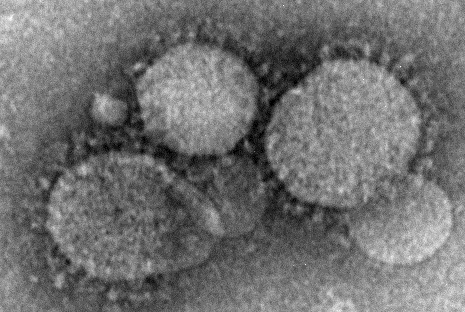User login
A little PT goes a long way in hospitalized COPD patients
MADRID – Adding physical therapy to standard care improved self-reported quality of life in patients hospitalized with an acute exacerbation of chronic obstructive pulmonary disease in a randomized, controlled trial.
Significant gains in health-related quality of life were seen at discharge on all of the EUROQol-5D questionnaire subscales including mobility (mean 2.00 vs. 1.29; P less than .001), self-care (mean 1.76 vs. 1.19; P = .004), usual activities (mean 2.14 vs. 1.43; P = .004), pain/discomfort (mean 1.71 vs. 1.24; P = .014), and anxiety/depression (mean 2.00 vs. 1.38; P less than .001).
Overall health, measured with the EUROQol-5D visual analog scale, also improved significantly from an average score of 57.0 to 74.4 (P = .006), Irene Torres-Sánchez, PT, reported at the world congress of the American College of Chest Physicians.
What stands out is that the average hospital length of stay was just 8.8 days.
The physical therapy protocol included 45 minutes of daily, individualized resistance training targeting the lower limbs and controlled breathing exercises including relaxation exercises, pursed lips breathing, and active expiration, explained Ms. Torres-Sánchez of University of Granada, Spain.
No significant differences were found between the 30 intervention patients and 30 controls at baseline in Saint George\'s Respiratory Questionnaire values (63.95 vs. 63.00). Their average age was 71 years and body mass index was 27.6 kg/m2.
Improvements were seen in the control group, but they were statistically significant, using a P value of less than .05, only for anxiety/depression (mean 1.96 vs. 1.46; P less than .001). Overall health did not improve significantly from baseline (55.42 vs. 58.96; P = .396), according to the poster presentation (Chest 2014;145:372A [doi:10.1378/chest.1823625]).
In two other posters reported during the same session, the investigators showed that adults hospitalized with acute COPD exacerbation walked only 255 steps per day on average (Chest 2014;145:385A [doi:10.1378/chest.1822986]).
Those who took part in the PT program, however, had improved muscle strength and steadiness and muscle endurance, although it was not uniformly significant for both legs (Chest 2014;145:369A [doi:10.1378/chest.1823630]).
The investigators reported no financial disclosures.
Dr. Eric Gartman, FCCP, comments: While further work would be needed in this area to confirm their data and ensure that the effects seen were not just due to the 45 minutes of daily attention the PT group received - it supports other recent studies in our field that the earlier we mobilize and encourage our patients to get out of bed, the better their functionality and outcomes. In addition to the benefit to patients, if such interventions were also shown to reduce COPD readmissions, formal implementation of these resources would become very attractive to health care teams and hospital administrators alike.
Dr. Eric Gartman, FCCP, comments: While further work would be needed in this area to confirm their data and ensure that the effects seen were not just due to the 45 minutes of daily attention the PT group received - it supports other recent studies in our field that the earlier we mobilize and encourage our patients to get out of bed, the better their functionality and outcomes. In addition to the benefit to patients, if such interventions were also shown to reduce COPD readmissions, formal implementation of these resources would become very attractive to health care teams and hospital administrators alike.
Dr. Eric Gartman, FCCP, comments: While further work would be needed in this area to confirm their data and ensure that the effects seen were not just due to the 45 minutes of daily attention the PT group received - it supports other recent studies in our field that the earlier we mobilize and encourage our patients to get out of bed, the better their functionality and outcomes. In addition to the benefit to patients, if such interventions were also shown to reduce COPD readmissions, formal implementation of these resources would become very attractive to health care teams and hospital administrators alike.
MADRID – Adding physical therapy to standard care improved self-reported quality of life in patients hospitalized with an acute exacerbation of chronic obstructive pulmonary disease in a randomized, controlled trial.
Significant gains in health-related quality of life were seen at discharge on all of the EUROQol-5D questionnaire subscales including mobility (mean 2.00 vs. 1.29; P less than .001), self-care (mean 1.76 vs. 1.19; P = .004), usual activities (mean 2.14 vs. 1.43; P = .004), pain/discomfort (mean 1.71 vs. 1.24; P = .014), and anxiety/depression (mean 2.00 vs. 1.38; P less than .001).
Overall health, measured with the EUROQol-5D visual analog scale, also improved significantly from an average score of 57.0 to 74.4 (P = .006), Irene Torres-Sánchez, PT, reported at the world congress of the American College of Chest Physicians.
What stands out is that the average hospital length of stay was just 8.8 days.
The physical therapy protocol included 45 minutes of daily, individualized resistance training targeting the lower limbs and controlled breathing exercises including relaxation exercises, pursed lips breathing, and active expiration, explained Ms. Torres-Sánchez of University of Granada, Spain.
No significant differences were found between the 30 intervention patients and 30 controls at baseline in Saint George\'s Respiratory Questionnaire values (63.95 vs. 63.00). Their average age was 71 years and body mass index was 27.6 kg/m2.
Improvements were seen in the control group, but they were statistically significant, using a P value of less than .05, only for anxiety/depression (mean 1.96 vs. 1.46; P less than .001). Overall health did not improve significantly from baseline (55.42 vs. 58.96; P = .396), according to the poster presentation (Chest 2014;145:372A [doi:10.1378/chest.1823625]).
In two other posters reported during the same session, the investigators showed that adults hospitalized with acute COPD exacerbation walked only 255 steps per day on average (Chest 2014;145:385A [doi:10.1378/chest.1822986]).
Those who took part in the PT program, however, had improved muscle strength and steadiness and muscle endurance, although it was not uniformly significant for both legs (Chest 2014;145:369A [doi:10.1378/chest.1823630]).
The investigators reported no financial disclosures.
MADRID – Adding physical therapy to standard care improved self-reported quality of life in patients hospitalized with an acute exacerbation of chronic obstructive pulmonary disease in a randomized, controlled trial.
Significant gains in health-related quality of life were seen at discharge on all of the EUROQol-5D questionnaire subscales including mobility (mean 2.00 vs. 1.29; P less than .001), self-care (mean 1.76 vs. 1.19; P = .004), usual activities (mean 2.14 vs. 1.43; P = .004), pain/discomfort (mean 1.71 vs. 1.24; P = .014), and anxiety/depression (mean 2.00 vs. 1.38; P less than .001).
Overall health, measured with the EUROQol-5D visual analog scale, also improved significantly from an average score of 57.0 to 74.4 (P = .006), Irene Torres-Sánchez, PT, reported at the world congress of the American College of Chest Physicians.
What stands out is that the average hospital length of stay was just 8.8 days.
The physical therapy protocol included 45 minutes of daily, individualized resistance training targeting the lower limbs and controlled breathing exercises including relaxation exercises, pursed lips breathing, and active expiration, explained Ms. Torres-Sánchez of University of Granada, Spain.
No significant differences were found between the 30 intervention patients and 30 controls at baseline in Saint George\'s Respiratory Questionnaire values (63.95 vs. 63.00). Their average age was 71 years and body mass index was 27.6 kg/m2.
Improvements were seen in the control group, but they were statistically significant, using a P value of less than .05, only for anxiety/depression (mean 1.96 vs. 1.46; P less than .001). Overall health did not improve significantly from baseline (55.42 vs. 58.96; P = .396), according to the poster presentation (Chest 2014;145:372A [doi:10.1378/chest.1823625]).
In two other posters reported during the same session, the investigators showed that adults hospitalized with acute COPD exacerbation walked only 255 steps per day on average (Chest 2014;145:385A [doi:10.1378/chest.1822986]).
Those who took part in the PT program, however, had improved muscle strength and steadiness and muscle endurance, although it was not uniformly significant for both legs (Chest 2014;145:369A [doi:10.1378/chest.1823630]).
The investigators reported no financial disclosures.
AT CHEST WORLD CONGRESS 2014
Key clinical point: Prescribing PT may reduce anxiety and depression in hospitalized COPD patients as well as improve muscle strength and steadiness.
Major finding: Overall health on the EUROQol-5D visual analog scale improved from 57.0 to 74.4 at discharge (P = .006).
Data source: A randomized, single-blind trial in 60 patients hospitalized with COPD.
Disclosures: The investigators reported no financial disclosures.
Walking disability raises red flag in diabetics with OA
PARIS – Walking disability from hip or knee osteoarthritis puts patients with comorbid diabetes at increased risk for serious diabetes complications, according to a retrospective cohort study.
After a median follow-up of 6.6 years among 437 patients with moderately severe symptomatic osteoarthritis (OA) and type 1 or type 2 diabetes, 37% experienced the composite diabetes-specific primary outcome of hospitalization for hypo- or hyperglycemia, soft tissue infection, amputation, or initiation of chronic dialysis.
Specifically, there were 51 hospitalizations for hypoglycemia, 11 for hyperglycemia, 127 for soft tissue infections, 10 amputations, and 4 patients who went on chronic dialysis.
After adjusting for age, sex, and preexisting cardiovascular disease, baseline walking disability was a significant independent predictor of risk for a non-CVD, diabetes-specific complication (Hazard ratio per unit increase in Health Assessment Questionnaire [HAQ] walking score, 1.26; P = .02), Dr. Gillian Hawker reported at the World Congress on Osteoarthritis.
In sensitivity analyses, the results were unchanged after additional adjustment for post-baseline receipt of a primary, elective hip or knee joint replacement or when retinopathy was included as an outcome.
"In people with diabetes, OA functional limitations may impede their ability to carry out diabetes self-management activities, increasing their risk for diabetes complications," noted Dr. Hawker, professor of medicine at the University of Toronto and physician-in-chief of medicine at Women’s College Hospital, Toronto.
This is particularly worrisome since the coprevalence of diabetes with OA is high, with as many as 50%-60% of patients with longstanding diabetes also having clinically evident hand, hip, or knee OA.
In an oral presentation at the same meeting, Dr. Hawker stressed the need for early identification of walking disability after reporting that greater walking disability was an independent predictor of all-cause death and major cardiovascular events in the overall cohort of patients with symptomatic hip or knee OA alone.
The retrospective cohort study linked provincial health administrative databases with surveys from a population cohort of 2,156 patients with at least moderately severe symptomatic hip or knee OA who were recruited from 1996 to 1998 through a screening survey in Ontario.
At baseline, 457 patients self-reported diabetes or met criteria for inclusion in the Ontario Diabetes Database (sensitivity 86%, specificity 97%) and were without preexisting retinopathy or renal failure. After censoring patients who died, emigrated, or had insufficient data, 434 patients were included in the current analysis. The study could not differentiate type 1 from type 2 diabetes.
Their mean age was 71.6 years (all were 55 years or older), 41% were obese, and 53.2% had preexisting CVD. Their median HAQ walking disability score was 2 on a 3-point scale, median HAQ grip score was 1 on a 3-point scale, and mean Western Ontario and McMaster Osteoarthritis Index (WOMAC) summary score was 43.4. Overall, 55.5% used a walking aid, and 39% reported using NSAIDs. (A walking disability score of 2 corresponds with walking outdoors on flat ground with much difficulty, whereas a score of 3 means the person is unable to do it. A grip score of 1 corresponds with some difficulty in just one of the following tasks: opening car doors, opening previously open jars, or turning faucets on and off.)
In multivariable analyses, no effect was found for baseline grip strength (adjusted HR, 1.16; P = .07), Dr. Hawker reported at the meeting, sponsored by the Osteoarthritis Research Society International.
However, in sensitivity analyses that further adjusted for receipt of a diabetes drug prescription or NSAID in patients at least 66 years old at baseline and thus eligible for drug benefits, both HAQ walking and grip scores were significant predictors of risk for a diabetes complication, she reported. Specific data were not shown in the poster presentation, but the adjusted hazard ratios per unit increase in HAQ were 1.36 (P = .003) and 1.26 (P = .01), respectively, according to the abstract.
"Controlling for confounders, reduced grip strength and increased walking disability were identified as potentially modifiable risk factors for serious diabetes complications in people with both OA and diabetes," the authors concluded. "Additional studies are warranted to confirm or refute our findings, and if confirmed, to elucidate potential mechanisms."
Dr. Hawker reported having no financial disclosures.
PARIS – Walking disability from hip or knee osteoarthritis puts patients with comorbid diabetes at increased risk for serious diabetes complications, according to a retrospective cohort study.
After a median follow-up of 6.6 years among 437 patients with moderately severe symptomatic osteoarthritis (OA) and type 1 or type 2 diabetes, 37% experienced the composite diabetes-specific primary outcome of hospitalization for hypo- or hyperglycemia, soft tissue infection, amputation, or initiation of chronic dialysis.
Specifically, there were 51 hospitalizations for hypoglycemia, 11 for hyperglycemia, 127 for soft tissue infections, 10 amputations, and 4 patients who went on chronic dialysis.
After adjusting for age, sex, and preexisting cardiovascular disease, baseline walking disability was a significant independent predictor of risk for a non-CVD, diabetes-specific complication (Hazard ratio per unit increase in Health Assessment Questionnaire [HAQ] walking score, 1.26; P = .02), Dr. Gillian Hawker reported at the World Congress on Osteoarthritis.
In sensitivity analyses, the results were unchanged after additional adjustment for post-baseline receipt of a primary, elective hip or knee joint replacement or when retinopathy was included as an outcome.
"In people with diabetes, OA functional limitations may impede their ability to carry out diabetes self-management activities, increasing their risk for diabetes complications," noted Dr. Hawker, professor of medicine at the University of Toronto and physician-in-chief of medicine at Women’s College Hospital, Toronto.
This is particularly worrisome since the coprevalence of diabetes with OA is high, with as many as 50%-60% of patients with longstanding diabetes also having clinically evident hand, hip, or knee OA.
In an oral presentation at the same meeting, Dr. Hawker stressed the need for early identification of walking disability after reporting that greater walking disability was an independent predictor of all-cause death and major cardiovascular events in the overall cohort of patients with symptomatic hip or knee OA alone.
The retrospective cohort study linked provincial health administrative databases with surveys from a population cohort of 2,156 patients with at least moderately severe symptomatic hip or knee OA who were recruited from 1996 to 1998 through a screening survey in Ontario.
At baseline, 457 patients self-reported diabetes or met criteria for inclusion in the Ontario Diabetes Database (sensitivity 86%, specificity 97%) and were without preexisting retinopathy or renal failure. After censoring patients who died, emigrated, or had insufficient data, 434 patients were included in the current analysis. The study could not differentiate type 1 from type 2 diabetes.
Their mean age was 71.6 years (all were 55 years or older), 41% were obese, and 53.2% had preexisting CVD. Their median HAQ walking disability score was 2 on a 3-point scale, median HAQ grip score was 1 on a 3-point scale, and mean Western Ontario and McMaster Osteoarthritis Index (WOMAC) summary score was 43.4. Overall, 55.5% used a walking aid, and 39% reported using NSAIDs. (A walking disability score of 2 corresponds with walking outdoors on flat ground with much difficulty, whereas a score of 3 means the person is unable to do it. A grip score of 1 corresponds with some difficulty in just one of the following tasks: opening car doors, opening previously open jars, or turning faucets on and off.)
In multivariable analyses, no effect was found for baseline grip strength (adjusted HR, 1.16; P = .07), Dr. Hawker reported at the meeting, sponsored by the Osteoarthritis Research Society International.
However, in sensitivity analyses that further adjusted for receipt of a diabetes drug prescription or NSAID in patients at least 66 years old at baseline and thus eligible for drug benefits, both HAQ walking and grip scores were significant predictors of risk for a diabetes complication, she reported. Specific data were not shown in the poster presentation, but the adjusted hazard ratios per unit increase in HAQ were 1.36 (P = .003) and 1.26 (P = .01), respectively, according to the abstract.
"Controlling for confounders, reduced grip strength and increased walking disability were identified as potentially modifiable risk factors for serious diabetes complications in people with both OA and diabetes," the authors concluded. "Additional studies are warranted to confirm or refute our findings, and if confirmed, to elucidate potential mechanisms."
Dr. Hawker reported having no financial disclosures.
PARIS – Walking disability from hip or knee osteoarthritis puts patients with comorbid diabetes at increased risk for serious diabetes complications, according to a retrospective cohort study.
After a median follow-up of 6.6 years among 437 patients with moderately severe symptomatic osteoarthritis (OA) and type 1 or type 2 diabetes, 37% experienced the composite diabetes-specific primary outcome of hospitalization for hypo- or hyperglycemia, soft tissue infection, amputation, or initiation of chronic dialysis.
Specifically, there were 51 hospitalizations for hypoglycemia, 11 for hyperglycemia, 127 for soft tissue infections, 10 amputations, and 4 patients who went on chronic dialysis.
After adjusting for age, sex, and preexisting cardiovascular disease, baseline walking disability was a significant independent predictor of risk for a non-CVD, diabetes-specific complication (Hazard ratio per unit increase in Health Assessment Questionnaire [HAQ] walking score, 1.26; P = .02), Dr. Gillian Hawker reported at the World Congress on Osteoarthritis.
In sensitivity analyses, the results were unchanged after additional adjustment for post-baseline receipt of a primary, elective hip or knee joint replacement or when retinopathy was included as an outcome.
"In people with diabetes, OA functional limitations may impede their ability to carry out diabetes self-management activities, increasing their risk for diabetes complications," noted Dr. Hawker, professor of medicine at the University of Toronto and physician-in-chief of medicine at Women’s College Hospital, Toronto.
This is particularly worrisome since the coprevalence of diabetes with OA is high, with as many as 50%-60% of patients with longstanding diabetes also having clinically evident hand, hip, or knee OA.
In an oral presentation at the same meeting, Dr. Hawker stressed the need for early identification of walking disability after reporting that greater walking disability was an independent predictor of all-cause death and major cardiovascular events in the overall cohort of patients with symptomatic hip or knee OA alone.
The retrospective cohort study linked provincial health administrative databases with surveys from a population cohort of 2,156 patients with at least moderately severe symptomatic hip or knee OA who were recruited from 1996 to 1998 through a screening survey in Ontario.
At baseline, 457 patients self-reported diabetes or met criteria for inclusion in the Ontario Diabetes Database (sensitivity 86%, specificity 97%) and were without preexisting retinopathy or renal failure. After censoring patients who died, emigrated, or had insufficient data, 434 patients were included in the current analysis. The study could not differentiate type 1 from type 2 diabetes.
Their mean age was 71.6 years (all were 55 years or older), 41% were obese, and 53.2% had preexisting CVD. Their median HAQ walking disability score was 2 on a 3-point scale, median HAQ grip score was 1 on a 3-point scale, and mean Western Ontario and McMaster Osteoarthritis Index (WOMAC) summary score was 43.4. Overall, 55.5% used a walking aid, and 39% reported using NSAIDs. (A walking disability score of 2 corresponds with walking outdoors on flat ground with much difficulty, whereas a score of 3 means the person is unable to do it. A grip score of 1 corresponds with some difficulty in just one of the following tasks: opening car doors, opening previously open jars, or turning faucets on and off.)
In multivariable analyses, no effect was found for baseline grip strength (adjusted HR, 1.16; P = .07), Dr. Hawker reported at the meeting, sponsored by the Osteoarthritis Research Society International.
However, in sensitivity analyses that further adjusted for receipt of a diabetes drug prescription or NSAID in patients at least 66 years old at baseline and thus eligible for drug benefits, both HAQ walking and grip scores were significant predictors of risk for a diabetes complication, she reported. Specific data were not shown in the poster presentation, but the adjusted hazard ratios per unit increase in HAQ were 1.36 (P = .003) and 1.26 (P = .01), respectively, according to the abstract.
"Controlling for confounders, reduced grip strength and increased walking disability were identified as potentially modifiable risk factors for serious diabetes complications in people with both OA and diabetes," the authors concluded. "Additional studies are warranted to confirm or refute our findings, and if confirmed, to elucidate potential mechanisms."
Dr. Hawker reported having no financial disclosures.
AT OARSI 2014
Key clinical point: Early identification of walking disability is important in patients with OA and diabetes to help them to carry out diabetes self-management activities.
Major finding: Baseline walking disability was associated with an increased risk for a non-CVD, diabetes-specific complication (adjusted HR, 1.26; P = .02).
Data source: A retrospective cohort study in 437 patients with OA and diabetes.
Disclosures: Dr. Hawker reported having no financial disclosures.
VIDEO: PCPs fuzzy on HCC surveillance in cirrhosis
CHICAGO – Several medical societies recommend that patients with cirrhosis undergo ultrasound surveillance for hepatocellular carcinoma every 6 months, but studies suggest less than 20% receive surveillance. The most common reason for failure to get the screening is lack of provider recommendation.
In a video interview with us at the annual Digestive Disease Week, Dr. Eimile Dalton-Fitzgerald of the University of Texas Southwestern Medical Center at Dallas delves into primary care provider (PCP) practice patterns, knowledge, and attitudes regarding hepatocellular carcinoma (HCC) surveillance gleaned from a survey of 77 PCPs in a large safety-net hospital in Dallas County, Texas.
Though most PCPs surveyed believe HCC surveillance is their responsibility (90%) and is effective for early tumor detection in cirrhosis (85%), many had misconceptions about the appropriate surveillance test choice.
PCPs also report several barriers to HCC surveillance, including not being up to date with surveillance guidelines (68%), difficulty with patient communication (56%), and having more important issues to manage in the clinic (52%).
A full 87% also said they’re influenced by a lack of recommendations from the U.S. Preventive Services Task Force, which has yet to weigh in on the issue.
Dr. Dalton-Fitzgerald reported no conflicting interests.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – Several medical societies recommend that patients with cirrhosis undergo ultrasound surveillance for hepatocellular carcinoma every 6 months, but studies suggest less than 20% receive surveillance. The most common reason for failure to get the screening is lack of provider recommendation.
In a video interview with us at the annual Digestive Disease Week, Dr. Eimile Dalton-Fitzgerald of the University of Texas Southwestern Medical Center at Dallas delves into primary care provider (PCP) practice patterns, knowledge, and attitudes regarding hepatocellular carcinoma (HCC) surveillance gleaned from a survey of 77 PCPs in a large safety-net hospital in Dallas County, Texas.
Though most PCPs surveyed believe HCC surveillance is their responsibility (90%) and is effective for early tumor detection in cirrhosis (85%), many had misconceptions about the appropriate surveillance test choice.
PCPs also report several barriers to HCC surveillance, including not being up to date with surveillance guidelines (68%), difficulty with patient communication (56%), and having more important issues to manage in the clinic (52%).
A full 87% also said they’re influenced by a lack of recommendations from the U.S. Preventive Services Task Force, which has yet to weigh in on the issue.
Dr. Dalton-Fitzgerald reported no conflicting interests.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – Several medical societies recommend that patients with cirrhosis undergo ultrasound surveillance for hepatocellular carcinoma every 6 months, but studies suggest less than 20% receive surveillance. The most common reason for failure to get the screening is lack of provider recommendation.
In a video interview with us at the annual Digestive Disease Week, Dr. Eimile Dalton-Fitzgerald of the University of Texas Southwestern Medical Center at Dallas delves into primary care provider (PCP) practice patterns, knowledge, and attitudes regarding hepatocellular carcinoma (HCC) surveillance gleaned from a survey of 77 PCPs in a large safety-net hospital in Dallas County, Texas.
Though most PCPs surveyed believe HCC surveillance is their responsibility (90%) and is effective for early tumor detection in cirrhosis (85%), many had misconceptions about the appropriate surveillance test choice.
PCPs also report several barriers to HCC surveillance, including not being up to date with surveillance guidelines (68%), difficulty with patient communication (56%), and having more important issues to manage in the clinic (52%).
A full 87% also said they’re influenced by a lack of recommendations from the U.S. Preventive Services Task Force, which has yet to weigh in on the issue.
Dr. Dalton-Fitzgerald reported no conflicting interests.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT DDW 2014
RM-131 makes headway against diabetic gastroparesis
CHICAGO – The investigational ghrelin agonist RM-131 significantly improved gastric emptying and vomiting in patients with diabetic gastroparesis in a phase II, double-blind study.
Gastric emptying improved by an average of 23 minutes from baseline after 4 weeks of twice-daily subcutaneous injections of RM-131 10 mcg (P less than .001). This compares with nonsignificant improvements of 7.5 minutes for placebo and 5.9 minutes for once-daily RM-131 10 mcg, Dr. Anthony Lembo reported in a late-breaking abstract session at the annual Digestive Disease Week.
Twice-daily RM-131 also reduced weekly vomiting episodes by 63% (P = .033) and vomiting severity by 58% (P = .005), compared with placebo.
RM-131, also known as relamorelin, has been granted fast-track review status by the Food and Drug Administration for the treatment of diabetic gastroparesis. The novel ghrelin pentapeptide agonist is 15- to 130-fold more potent than ghrelin, a hormone produced in the stomach that stimulates gastrointestinal activity, according to Dr. Lembo, with Beth Israel Deaconess Medical Center in Boston.
The study enrolled 204 patients with type 1 or type 2 diabetes and a hemoglobin A1c value of less than 11%; a history of ongoing gastroparesis symptoms; nausea and/or vomiting at least once in the 2 weeks prior to enrollment; and a screening gastric emptying time of more than 79 minutes. Their mean age was 55 years.
A post hoc analysis on patients with baseline vomiting showed that twice-daily RM-131 was effective on all endpoints, including four subjective patient-reported symptoms – nausea, abdominal pain, bloating, and early satiety, Dr. Lembo said.
Gastric emptying, as demonstrated by the Gastric Emptying Breath Test, improved by 30.6 minutes from baseline (P = .02), weekly vomiting episodes by 63% (P = .041), and vomiting severity by 59% (P = .006).
The four-symptom composite score improved numerically, but not significantly, in the overall population, whereas there was a clear and significant improvement in 1 week in the subgroup with vomiting (6.36 points; P less than .043), he said.
The subgroup with vomiting comprised about 60% of the study population, and had 5.6 to 6.4 baseline vomiting episodes per week versus 3.2 to 3.5 per week in the overall population. Vomiting is the most bothersome symptom in diabetic gastroparesis and often brings patients to the hospital for treatment, Dr. Lembo observed.
The subset with vomiting also had more severe overall gastric emptying symptoms at baseline. Initial analyses, however, suggest a weak correlation between improvement in gastric emptying and symptom improvement, he said.
Session cochair Dr. John Inadomi, professor of medicine and head of gastroenterology, University of Washington, Seattle, said in an interview that the weak correlation "is not surprising given what we know about gastroparesis and the pathogenesis of symptoms, but it does raise questions about where the most effective therapy is going to come from. Is it going to come from trying to accelerate gastric emptying or trying to go after some other way to relieve symptoms? Nevertheless, this is the first study of a so-called pure prokinetic that has had at least some clinical benefit, but again, it was in the subset of patients with vomiting."
Treatment-emergent adverse events occurred in 30 of 69 patients (43.5%) on placebo, 32 of 67 patients (47.8%) on once-daily RM-131, and 25 of 68 patients (36.8%) on twice-daily dosing. No adverse event was more common with RM-131 and likewise, there were no concerns about labs, glucose, weight, ECGs, physical exam, or injection-site reactions, Dr. Lembo said.
Rhythm Pharmaceuticals sponsored the study. Dr. Lembo reported relationships with Salix Pharmaceuticals, AstraZeneca, and other companies. Dr. Inadomi reported relationships with Given Imaging, ChemImage, and other companies.
Increased public awareness and the recent release of updated clinical guidelines have made the diagnosis of gastroparesis easier for clinicians. The treatment of gastroparesis, however, remains problematic for both patients and providers for a number of reasons. One, gastroparesis is not a single disorder; rather, the pathophysiology of gastroparesis is multifactorial, and patients with identical symptoms often have different underlying pathophysiologic processes responsible for symptom generation. Two, treating the underlying pathophysiology (i.e., delayed gastric emptying) does not necessarily translate into an improvement in symptoms. Three, metoclopramide is the only medication currently approved by the Food and Drug Administration for the treatment of diabetic gastroparesis, although it is not recommended for long-term use and may cause side effects in up to 30% of diabetic patients. Finally, distinguishing idiopathic gastroparesis from functional dyspepsia can be difficult, further complicating treatment.

|
| Dr. Brian E. Lacy |
The authors also reported that RM-131 improved gastric emptying at the end of the 4-week study, measured by a breath test. Other individual symptoms of gastroparesis were not improved in the total group, although in a subgroup of patients with more severe vomiting symptoms, RM-131 appeared to provide the greatest improvement in symptoms. As in other studies in gastroparetic patients, improvement in gastric emptying did not predict an improvement in symptoms.
Although the precise mechanism by which RM-131 improves symptoms, especially in the subset of patients with predominant vomiting, is unknown, the current study is important, as it signals interest in an entirely new class of therapeutic agents for diabetic gastroparesis. Larger prospective studies, subtyped based on predominant symptoms and the extent of delayed gastric emptying, are eagerly awaited.
Dr. Brian E. Lacy is professor of medicine, Geisel School of Medicine at Dartmouth, and chief of the section of gastroenterology and hepatology at Dartmouth-Hitchcock Medical Center, Hanover, N.H. He is on the scientific advisory boards of Ironwood, Takeda, Prometheus, and Salix.
Increased public awareness and the recent release of updated clinical guidelines have made the diagnosis of gastroparesis easier for clinicians. The treatment of gastroparesis, however, remains problematic for both patients and providers for a number of reasons. One, gastroparesis is not a single disorder; rather, the pathophysiology of gastroparesis is multifactorial, and patients with identical symptoms often have different underlying pathophysiologic processes responsible for symptom generation. Two, treating the underlying pathophysiology (i.e., delayed gastric emptying) does not necessarily translate into an improvement in symptoms. Three, metoclopramide is the only medication currently approved by the Food and Drug Administration for the treatment of diabetic gastroparesis, although it is not recommended for long-term use and may cause side effects in up to 30% of diabetic patients. Finally, distinguishing idiopathic gastroparesis from functional dyspepsia can be difficult, further complicating treatment.

|
| Dr. Brian E. Lacy |
The authors also reported that RM-131 improved gastric emptying at the end of the 4-week study, measured by a breath test. Other individual symptoms of gastroparesis were not improved in the total group, although in a subgroup of patients with more severe vomiting symptoms, RM-131 appeared to provide the greatest improvement in symptoms. As in other studies in gastroparetic patients, improvement in gastric emptying did not predict an improvement in symptoms.
Although the precise mechanism by which RM-131 improves symptoms, especially in the subset of patients with predominant vomiting, is unknown, the current study is important, as it signals interest in an entirely new class of therapeutic agents for diabetic gastroparesis. Larger prospective studies, subtyped based on predominant symptoms and the extent of delayed gastric emptying, are eagerly awaited.
Dr. Brian E. Lacy is professor of medicine, Geisel School of Medicine at Dartmouth, and chief of the section of gastroenterology and hepatology at Dartmouth-Hitchcock Medical Center, Hanover, N.H. He is on the scientific advisory boards of Ironwood, Takeda, Prometheus, and Salix.
Increased public awareness and the recent release of updated clinical guidelines have made the diagnosis of gastroparesis easier for clinicians. The treatment of gastroparesis, however, remains problematic for both patients and providers for a number of reasons. One, gastroparesis is not a single disorder; rather, the pathophysiology of gastroparesis is multifactorial, and patients with identical symptoms often have different underlying pathophysiologic processes responsible for symptom generation. Two, treating the underlying pathophysiology (i.e., delayed gastric emptying) does not necessarily translate into an improvement in symptoms. Three, metoclopramide is the only medication currently approved by the Food and Drug Administration for the treatment of diabetic gastroparesis, although it is not recommended for long-term use and may cause side effects in up to 30% of diabetic patients. Finally, distinguishing idiopathic gastroparesis from functional dyspepsia can be difficult, further complicating treatment.

|
| Dr. Brian E. Lacy |
The authors also reported that RM-131 improved gastric emptying at the end of the 4-week study, measured by a breath test. Other individual symptoms of gastroparesis were not improved in the total group, although in a subgroup of patients with more severe vomiting symptoms, RM-131 appeared to provide the greatest improvement in symptoms. As in other studies in gastroparetic patients, improvement in gastric emptying did not predict an improvement in symptoms.
Although the precise mechanism by which RM-131 improves symptoms, especially in the subset of patients with predominant vomiting, is unknown, the current study is important, as it signals interest in an entirely new class of therapeutic agents for diabetic gastroparesis. Larger prospective studies, subtyped based on predominant symptoms and the extent of delayed gastric emptying, are eagerly awaited.
Dr. Brian E. Lacy is professor of medicine, Geisel School of Medicine at Dartmouth, and chief of the section of gastroenterology and hepatology at Dartmouth-Hitchcock Medical Center, Hanover, N.H. He is on the scientific advisory boards of Ironwood, Takeda, Prometheus, and Salix.
CHICAGO – The investigational ghrelin agonist RM-131 significantly improved gastric emptying and vomiting in patients with diabetic gastroparesis in a phase II, double-blind study.
Gastric emptying improved by an average of 23 minutes from baseline after 4 weeks of twice-daily subcutaneous injections of RM-131 10 mcg (P less than .001). This compares with nonsignificant improvements of 7.5 minutes for placebo and 5.9 minutes for once-daily RM-131 10 mcg, Dr. Anthony Lembo reported in a late-breaking abstract session at the annual Digestive Disease Week.
Twice-daily RM-131 also reduced weekly vomiting episodes by 63% (P = .033) and vomiting severity by 58% (P = .005), compared with placebo.
RM-131, also known as relamorelin, has been granted fast-track review status by the Food and Drug Administration for the treatment of diabetic gastroparesis. The novel ghrelin pentapeptide agonist is 15- to 130-fold more potent than ghrelin, a hormone produced in the stomach that stimulates gastrointestinal activity, according to Dr. Lembo, with Beth Israel Deaconess Medical Center in Boston.
The study enrolled 204 patients with type 1 or type 2 diabetes and a hemoglobin A1c value of less than 11%; a history of ongoing gastroparesis symptoms; nausea and/or vomiting at least once in the 2 weeks prior to enrollment; and a screening gastric emptying time of more than 79 minutes. Their mean age was 55 years.
A post hoc analysis on patients with baseline vomiting showed that twice-daily RM-131 was effective on all endpoints, including four subjective patient-reported symptoms – nausea, abdominal pain, bloating, and early satiety, Dr. Lembo said.
Gastric emptying, as demonstrated by the Gastric Emptying Breath Test, improved by 30.6 minutes from baseline (P = .02), weekly vomiting episodes by 63% (P = .041), and vomiting severity by 59% (P = .006).
The four-symptom composite score improved numerically, but not significantly, in the overall population, whereas there was a clear and significant improvement in 1 week in the subgroup with vomiting (6.36 points; P less than .043), he said.
The subgroup with vomiting comprised about 60% of the study population, and had 5.6 to 6.4 baseline vomiting episodes per week versus 3.2 to 3.5 per week in the overall population. Vomiting is the most bothersome symptom in diabetic gastroparesis and often brings patients to the hospital for treatment, Dr. Lembo observed.
The subset with vomiting also had more severe overall gastric emptying symptoms at baseline. Initial analyses, however, suggest a weak correlation between improvement in gastric emptying and symptom improvement, he said.
Session cochair Dr. John Inadomi, professor of medicine and head of gastroenterology, University of Washington, Seattle, said in an interview that the weak correlation "is not surprising given what we know about gastroparesis and the pathogenesis of symptoms, but it does raise questions about where the most effective therapy is going to come from. Is it going to come from trying to accelerate gastric emptying or trying to go after some other way to relieve symptoms? Nevertheless, this is the first study of a so-called pure prokinetic that has had at least some clinical benefit, but again, it was in the subset of patients with vomiting."
Treatment-emergent adverse events occurred in 30 of 69 patients (43.5%) on placebo, 32 of 67 patients (47.8%) on once-daily RM-131, and 25 of 68 patients (36.8%) on twice-daily dosing. No adverse event was more common with RM-131 and likewise, there were no concerns about labs, glucose, weight, ECGs, physical exam, or injection-site reactions, Dr. Lembo said.
Rhythm Pharmaceuticals sponsored the study. Dr. Lembo reported relationships with Salix Pharmaceuticals, AstraZeneca, and other companies. Dr. Inadomi reported relationships with Given Imaging, ChemImage, and other companies.
CHICAGO – The investigational ghrelin agonist RM-131 significantly improved gastric emptying and vomiting in patients with diabetic gastroparesis in a phase II, double-blind study.
Gastric emptying improved by an average of 23 minutes from baseline after 4 weeks of twice-daily subcutaneous injections of RM-131 10 mcg (P less than .001). This compares with nonsignificant improvements of 7.5 minutes for placebo and 5.9 minutes for once-daily RM-131 10 mcg, Dr. Anthony Lembo reported in a late-breaking abstract session at the annual Digestive Disease Week.
Twice-daily RM-131 also reduced weekly vomiting episodes by 63% (P = .033) and vomiting severity by 58% (P = .005), compared with placebo.
RM-131, also known as relamorelin, has been granted fast-track review status by the Food and Drug Administration for the treatment of diabetic gastroparesis. The novel ghrelin pentapeptide agonist is 15- to 130-fold more potent than ghrelin, a hormone produced in the stomach that stimulates gastrointestinal activity, according to Dr. Lembo, with Beth Israel Deaconess Medical Center in Boston.
The study enrolled 204 patients with type 1 or type 2 diabetes and a hemoglobin A1c value of less than 11%; a history of ongoing gastroparesis symptoms; nausea and/or vomiting at least once in the 2 weeks prior to enrollment; and a screening gastric emptying time of more than 79 minutes. Their mean age was 55 years.
A post hoc analysis on patients with baseline vomiting showed that twice-daily RM-131 was effective on all endpoints, including four subjective patient-reported symptoms – nausea, abdominal pain, bloating, and early satiety, Dr. Lembo said.
Gastric emptying, as demonstrated by the Gastric Emptying Breath Test, improved by 30.6 minutes from baseline (P = .02), weekly vomiting episodes by 63% (P = .041), and vomiting severity by 59% (P = .006).
The four-symptom composite score improved numerically, but not significantly, in the overall population, whereas there was a clear and significant improvement in 1 week in the subgroup with vomiting (6.36 points; P less than .043), he said.
The subgroup with vomiting comprised about 60% of the study population, and had 5.6 to 6.4 baseline vomiting episodes per week versus 3.2 to 3.5 per week in the overall population. Vomiting is the most bothersome symptom in diabetic gastroparesis and often brings patients to the hospital for treatment, Dr. Lembo observed.
The subset with vomiting also had more severe overall gastric emptying symptoms at baseline. Initial analyses, however, suggest a weak correlation between improvement in gastric emptying and symptom improvement, he said.
Session cochair Dr. John Inadomi, professor of medicine and head of gastroenterology, University of Washington, Seattle, said in an interview that the weak correlation "is not surprising given what we know about gastroparesis and the pathogenesis of symptoms, but it does raise questions about where the most effective therapy is going to come from. Is it going to come from trying to accelerate gastric emptying or trying to go after some other way to relieve symptoms? Nevertheless, this is the first study of a so-called pure prokinetic that has had at least some clinical benefit, but again, it was in the subset of patients with vomiting."
Treatment-emergent adverse events occurred in 30 of 69 patients (43.5%) on placebo, 32 of 67 patients (47.8%) on once-daily RM-131, and 25 of 68 patients (36.8%) on twice-daily dosing. No adverse event was more common with RM-131 and likewise, there were no concerns about labs, glucose, weight, ECGs, physical exam, or injection-site reactions, Dr. Lembo said.
Rhythm Pharmaceuticals sponsored the study. Dr. Lembo reported relationships with Salix Pharmaceuticals, AstraZeneca, and other companies. Dr. Inadomi reported relationships with Given Imaging, ChemImage, and other companies.
AT DDW 2014
Major finding: Twice-daily RM-131 reduced overall weekly vomiting episodes by 63% (P = .033) and vomiting severity by 58% (P = .005).
Data source: A phase II, double-blind, randomized study in 204 diabetic gastroparesis patients.
Disclosures: Rhythm Pharmaceuticals sponsored the study. Dr. Lembo reported relationships with Salix Pharmaceuticals, AstraZeneca, and other companies.
VIDEO – IBD meds found safe during pregnancy
CHICAGO – Medications to treat inflammatory bowel disease were well tolerated in pregnancy and weren’t linked to worse maternal-fetal outcomes, according to a study of more than 1,000 pregnant women.
Despite many physicians’ concerns about the safety of azathioprine and biologic agents during pregnancy, maternal exposure to those IBD medications was not associated with an increase in adverse outcomes such as preterm birth or congenital anomalies, explained Dr. Uma Mahadevan of the University of California, San Francisco Center for Colitis and Crohn’s Disease.
"If you continue [the medications] and control your disease and have a healthy pregnancy, that’s the key to having a healthy baby," Dr. Mahadevan observed.
In a video interview at the annual Digestive Disease Week, Dr. Mahadevan discussed the study’s findings, whether combination therapy has an impact on the risk of preterm birth, and what effect IBD medication exposure may have had on children’s achievement of developmental milestones up to age 4 years.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – Medications to treat inflammatory bowel disease were well tolerated in pregnancy and weren’t linked to worse maternal-fetal outcomes, according to a study of more than 1,000 pregnant women.
Despite many physicians’ concerns about the safety of azathioprine and biologic agents during pregnancy, maternal exposure to those IBD medications was not associated with an increase in adverse outcomes such as preterm birth or congenital anomalies, explained Dr. Uma Mahadevan of the University of California, San Francisco Center for Colitis and Crohn’s Disease.
"If you continue [the medications] and control your disease and have a healthy pregnancy, that’s the key to having a healthy baby," Dr. Mahadevan observed.
In a video interview at the annual Digestive Disease Week, Dr. Mahadevan discussed the study’s findings, whether combination therapy has an impact on the risk of preterm birth, and what effect IBD medication exposure may have had on children’s achievement of developmental milestones up to age 4 years.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – Medications to treat inflammatory bowel disease were well tolerated in pregnancy and weren’t linked to worse maternal-fetal outcomes, according to a study of more than 1,000 pregnant women.
Despite many physicians’ concerns about the safety of azathioprine and biologic agents during pregnancy, maternal exposure to those IBD medications was not associated with an increase in adverse outcomes such as preterm birth or congenital anomalies, explained Dr. Uma Mahadevan of the University of California, San Francisco Center for Colitis and Crohn’s Disease.
"If you continue [the medications] and control your disease and have a healthy pregnancy, that’s the key to having a healthy baby," Dr. Mahadevan observed.
In a video interview at the annual Digestive Disease Week, Dr. Mahadevan discussed the study’s findings, whether combination therapy has an impact on the risk of preterm birth, and what effect IBD medication exposure may have had on children’s achievement of developmental milestones up to age 4 years.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
At DDW 2014
IBD Meds Found Safe During Pregnancy
CHICAGO – Medications to treat inflammatory bowel disease were well tolerated in pregnancy and weren’t linked to worse maternal-fetal outcomes, according to a study of more than 1,000 pregnant women.
Despite many physicians’ concerns about the safety of azathioprine and biologic agents during pregnancy, maternal exposure to those IBD medications was not associated with an increase in adverse outcomes such as preterm birth or congenital anomalies, explained Dr. Uma Mahadevan of the University of California, San Francisco Center for Colitis and Crohn’s Disease.
"If you continue [the medications] and control your disease and have a healthy pregnancy, that’s the key to having a healthy baby," Dr. Mahadevan observed.
In a video interview at the annual Digestive Disease Week, Dr. Mahadevan discussed the study’s findings, whether combination therapy has an impact on the risk of preterm birth, and what effect IBD medication exposure may have had on children’s achievement of developmental milestones up to age 4 years.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – Medications to treat inflammatory bowel disease were well tolerated in pregnancy and weren’t linked to worse maternal-fetal outcomes, according to a study of more than 1,000 pregnant women.
Despite many physicians’ concerns about the safety of azathioprine and biologic agents during pregnancy, maternal exposure to those IBD medications was not associated with an increase in adverse outcomes such as preterm birth or congenital anomalies, explained Dr. Uma Mahadevan of the University of California, San Francisco Center for Colitis and Crohn’s Disease.
"If you continue [the medications] and control your disease and have a healthy pregnancy, that’s the key to having a healthy baby," Dr. Mahadevan observed.
In a video interview at the annual Digestive Disease Week, Dr. Mahadevan discussed the study’s findings, whether combination therapy has an impact on the risk of preterm birth, and what effect IBD medication exposure may have had on children’s achievement of developmental milestones up to age 4 years.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – Medications to treat inflammatory bowel disease were well tolerated in pregnancy and weren’t linked to worse maternal-fetal outcomes, according to a study of more than 1,000 pregnant women.
Despite many physicians’ concerns about the safety of azathioprine and biologic agents during pregnancy, maternal exposure to those IBD medications was not associated with an increase in adverse outcomes such as preterm birth or congenital anomalies, explained Dr. Uma Mahadevan of the University of California, San Francisco Center for Colitis and Crohn’s Disease.
"If you continue [the medications] and control your disease and have a healthy pregnancy, that’s the key to having a healthy baby," Dr. Mahadevan observed.
In a video interview at the annual Digestive Disease Week, Dr. Mahadevan discussed the study’s findings, whether combination therapy has an impact on the risk of preterm birth, and what effect IBD medication exposure may have had on children’s achievement of developmental milestones up to age 4 years.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
At DDW 2014
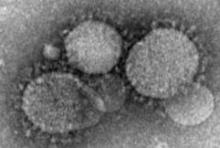
CDC Confirms First U.S. Case of Deadly Middle East Respiratory Syndrome
The Centers for Disease Control and Prevention has confirmed the first case of the deadly Middle East Respiratory Syndrome coronavirus in the United States.
The patient, a health care worker who recently returned from Riyadh, Saudi Arabia, is in stable condition in an Indiana hospital after being admitted with shortness of breath, cough, and fever on April 27.
No other cases have been reported beyond this single case, but investigators are just beginning to reach out to those who came in contact with the patient, said Dr. Anne Schuchat, assistant surgeon general and director of the CDC’s National Center for Immunization and Respiratory Diseases, during a press briefing.
That list could be quite long, as the patient flew on April 24 from Saudi Arabia to London, then on to Chicago, and then traveled by bus to Indiana. The patient is in good condition, and remains in isolation in Community Hospital in Munster, Indiana, according to a statement from the hospital.
Health officials would not release the name of the airline or bus line the patient traveled on.
Indiana state health officials took specimens from the patient, which were then confirmed late today by the CDC.
There is no specific treatment for the Middle East Respiratory Syndrome coronavirus (MERS-CoV), which has a case fatality rate of about 30% among symptomatic patients.
Since the virus was first reported in 2012 in Saudi Arabia, 263 cases have been confirmed in 12 countries, with 92 deaths. More than 100 other patients have been confirmed and reported by their ministries of health, but are not included in the World Health Organization tally, Dr. Schuchat said.
The Indiana case represents a "very low risk to the broader general public" because MERS-CoV has not shown the ability to spread easily from person to person in the community setting, she said.
"It is very concerning that the virus has spread in hospitals, and we should not be surprised if additional cases are identified among the health care workers who had close contact with this patient before the patient was isolated and special precautions were implemented," she went on to say.
An outbreak in the spring of 2013 involved 23 confirmed cases at four healthcare facilities, and another surge in cases beginning in March of this year has been identified.
It is unknown whether this represents seasonal variation or mutations in the virus that may make it more easily transmissible. At least one isolate has been sequenced by polymerase chain reaction from a patient in Saudi Arabia, and there were no changes in the virus, Dr. Schuchat said.
"With the SARS virus 10, 11 years ago, we did believe there was a change in the virus that led to more explosive transmission. So we’ll continue to look at that, but we don’t have data right now to confirm that that has happened," she said.
For now, the CDC is not advising people change their travel plans, but is recommending that people who develop a fever, cough, or shortness of breath within 14 days after traveling in or near the Arabian Peninsula see a health care provider, Dr. Schuchat said. Others that should monitor their symptoms include those who have close contact with someone who has these symptoms and travel history.
The CDC is also advising people to use basic respiratory illness precautions such as frequent hand washing, avoiding close contact with people who are sick, and disinfecting frequently touched surfaces.
No specific precautions were given for pediatricians or children, other than to ask for a travel history in patients with respiratory symptoms. The youngest case of MERS-CoV was in a 2 year-old, but the median age is around 51 years, she said.
Dr. Schuchat stressed that guidance may change as this "very fluid" situation evolves. The Indiana Department of Health and the CDC will share additional information on their Web sites and host additional press conferences, as needed, she said.
For more information on Middle East Respiratory Syndrome, go to http://www.cdc.gov/coronavirus/mers/index.html.
The Centers for Disease Control and Prevention has confirmed the first case of the deadly Middle East Respiratory Syndrome coronavirus in the United States.
The patient, a health care worker who recently returned from Riyadh, Saudi Arabia, is in stable condition in an Indiana hospital after being admitted with shortness of breath, cough, and fever on April 27.
No other cases have been reported beyond this single case, but investigators are just beginning to reach out to those who came in contact with the patient, said Dr. Anne Schuchat, assistant surgeon general and director of the CDC’s National Center for Immunization and Respiratory Diseases, during a press briefing.
That list could be quite long, as the patient flew on April 24 from Saudi Arabia to London, then on to Chicago, and then traveled by bus to Indiana. The patient is in good condition, and remains in isolation in Community Hospital in Munster, Indiana, according to a statement from the hospital.
Health officials would not release the name of the airline or bus line the patient traveled on.
Indiana state health officials took specimens from the patient, which were then confirmed late today by the CDC.
There is no specific treatment for the Middle East Respiratory Syndrome coronavirus (MERS-CoV), which has a case fatality rate of about 30% among symptomatic patients.
Since the virus was first reported in 2012 in Saudi Arabia, 263 cases have been confirmed in 12 countries, with 92 deaths. More than 100 other patients have been confirmed and reported by their ministries of health, but are not included in the World Health Organization tally, Dr. Schuchat said.
The Indiana case represents a "very low risk to the broader general public" because MERS-CoV has not shown the ability to spread easily from person to person in the community setting, she said.
"It is very concerning that the virus has spread in hospitals, and we should not be surprised if additional cases are identified among the health care workers who had close contact with this patient before the patient was isolated and special precautions were implemented," she went on to say.
An outbreak in the spring of 2013 involved 23 confirmed cases at four healthcare facilities, and another surge in cases beginning in March of this year has been identified.
It is unknown whether this represents seasonal variation or mutations in the virus that may make it more easily transmissible. At least one isolate has been sequenced by polymerase chain reaction from a patient in Saudi Arabia, and there were no changes in the virus, Dr. Schuchat said.
"With the SARS virus 10, 11 years ago, we did believe there was a change in the virus that led to more explosive transmission. So we’ll continue to look at that, but we don’t have data right now to confirm that that has happened," she said.
For now, the CDC is not advising people change their travel plans, but is recommending that people who develop a fever, cough, or shortness of breath within 14 days after traveling in or near the Arabian Peninsula see a health care provider, Dr. Schuchat said. Others that should monitor their symptoms include those who have close contact with someone who has these symptoms and travel history.
The CDC is also advising people to use basic respiratory illness precautions such as frequent hand washing, avoiding close contact with people who are sick, and disinfecting frequently touched surfaces.
No specific precautions were given for pediatricians or children, other than to ask for a travel history in patients with respiratory symptoms. The youngest case of MERS-CoV was in a 2 year-old, but the median age is around 51 years, she said.
Dr. Schuchat stressed that guidance may change as this "very fluid" situation evolves. The Indiana Department of Health and the CDC will share additional information on their Web sites and host additional press conferences, as needed, she said.
For more information on Middle East Respiratory Syndrome, go to http://www.cdc.gov/coronavirus/mers/index.html.
The Centers for Disease Control and Prevention has confirmed the first case of the deadly Middle East Respiratory Syndrome coronavirus in the United States.
The patient, a health care worker who recently returned from Riyadh, Saudi Arabia, is in stable condition in an Indiana hospital after being admitted with shortness of breath, cough, and fever on April 27.
No other cases have been reported beyond this single case, but investigators are just beginning to reach out to those who came in contact with the patient, said Dr. Anne Schuchat, assistant surgeon general and director of the CDC’s National Center for Immunization and Respiratory Diseases, during a press briefing.
That list could be quite long, as the patient flew on April 24 from Saudi Arabia to London, then on to Chicago, and then traveled by bus to Indiana. The patient is in good condition, and remains in isolation in Community Hospital in Munster, Indiana, according to a statement from the hospital.
Health officials would not release the name of the airline or bus line the patient traveled on.
Indiana state health officials took specimens from the patient, which were then confirmed late today by the CDC.
There is no specific treatment for the Middle East Respiratory Syndrome coronavirus (MERS-CoV), which has a case fatality rate of about 30% among symptomatic patients.
Since the virus was first reported in 2012 in Saudi Arabia, 263 cases have been confirmed in 12 countries, with 92 deaths. More than 100 other patients have been confirmed and reported by their ministries of health, but are not included in the World Health Organization tally, Dr. Schuchat said.
The Indiana case represents a "very low risk to the broader general public" because MERS-CoV has not shown the ability to spread easily from person to person in the community setting, she said.
"It is very concerning that the virus has spread in hospitals, and we should not be surprised if additional cases are identified among the health care workers who had close contact with this patient before the patient was isolated and special precautions were implemented," she went on to say.
An outbreak in the spring of 2013 involved 23 confirmed cases at four healthcare facilities, and another surge in cases beginning in March of this year has been identified.
It is unknown whether this represents seasonal variation or mutations in the virus that may make it more easily transmissible. At least one isolate has been sequenced by polymerase chain reaction from a patient in Saudi Arabia, and there were no changes in the virus, Dr. Schuchat said.
"With the SARS virus 10, 11 years ago, we did believe there was a change in the virus that led to more explosive transmission. So we’ll continue to look at that, but we don’t have data right now to confirm that that has happened," she said.
For now, the CDC is not advising people change their travel plans, but is recommending that people who develop a fever, cough, or shortness of breath within 14 days after traveling in or near the Arabian Peninsula see a health care provider, Dr. Schuchat said. Others that should monitor their symptoms include those who have close contact with someone who has these symptoms and travel history.
The CDC is also advising people to use basic respiratory illness precautions such as frequent hand washing, avoiding close contact with people who are sick, and disinfecting frequently touched surfaces.
No specific precautions were given for pediatricians or children, other than to ask for a travel history in patients with respiratory symptoms. The youngest case of MERS-CoV was in a 2 year-old, but the median age is around 51 years, she said.
Dr. Schuchat stressed that guidance may change as this "very fluid" situation evolves. The Indiana Department of Health and the CDC will share additional information on their Web sites and host additional press conferences, as needed, she said.
For more information on Middle East Respiratory Syndrome, go to http://www.cdc.gov/coronavirus/mers/index.html.
Patient satisfaction not always linked to hospital safety, effectiveness
BOSTON – Hospital size and operative volume were significantly associated with satisfaction among general surgery patients in an analysis of 171 U.S. hospitals.
Surprisingly, all other safety and effectiveness measures, with the exception of low hospital mortality index, did not reliably reflect patient satisfaction, "indicating that the system plays perhaps a bigger role than anything else we can do," Dr. Gregory D. Kennedy said at the annual meeting of the American Surgical Association.
Moreover, a clean room and well-controlled pain were the best predictors of high patient satisfaction.
If it’s "the quality of the hotel, not the quality of the surgeon that drives patient satisfaction," and given that this is tied to reimbursement, what should the message be to hospital CEOs? asked discussant Dr. John J. Ricotta, chief of surgery at MedStar Washington (D.C.) Hospital Center.
Dr. Kennedy said the message he takes to the C-suite is that patient satisfaction cannot be a surrogate marker for safety and effectiveness or the only measure of quality because, in doing the right thing, surgeons often make patients unhappy. As a colorectal surgeon, he said he has unhappy patients every day, and remarked that he sometimes feels like a used car salesman where the only thing that he worries about is whether the patient is having a good experience when they drive off the lot, not whether it’s a safe, reliable car.
Dr. Kennedy, vice chair of quality at the University of Wisconsin School of Medicine, Madison, suggested that future quality measures also may need to make the distinction between satisfied and engaged, well-informed patients because a disengaged patient can be highly satisfied, while a highly engaged patient may not.
For the current study, the investigators examined federal Hospital Consumer Assessment Healthcare Providers and Systems (HCAHPS) survey results from 171 hospitals in the University Health System Consortium database from 2011 to 2012. Patients can check one of four boxes for each question on the 27-item survey, with high satisfaction defined as median responses above the 75th percentile on the top box score. This cutoff was used because the Centers for Medicare & Medicaid Services, which developed the HCAHPS, uses only the top box score, Dr. Kennedy explained.
The median hospital size was 421 beds (range, 25-1,280 beds), the median operative volume was 6,341 cases (range, 192-24,258 cases), and the mortality index was 0.83 (range, 0-2.61).
In all, 62% of high-volume hospitals, defined as those with an operative volume above the median, achieved high patient satisfaction, compared with 38% of low-volume hospitals (P less than .001). Similar results were seen for operative volume, he said.
Other system measures such as number of ICU cases and Surgical Care Improvement Project (SCIP) compliance were not associated with high HCAHPS scores.
Among patient safety indicators, only low mortality index was associated with high satisfaction (P less than .001), while complications, early mortality, and overall mortality were not.
Interestingly, hospitals with a higher number of Patient Safety Indicator cases – those involving accidental puncture, laceration, and venous thromboembolism – had higher rates of patient satisfaction, "suggesting that unsafe care is perhaps correlated with high satisfaction," Dr. Kennedy said.
Discussant Dr. Fabrizio Michelassi, chair of surgery at Weill Cornell Medical College and surgeon-in-chief, New York–Presbyterian Hospital/Weill Cornell Medical Center, in New York City, questioned whether "unsafe care gives more options for physicians to show their compassionate side," and said the overall findings are not that surprising to practicing surgeons, who frequently hear patient complaints, despite having performed a quality operation.
Dr. Kennedy said a recent paper from the Cleveland Clinic (Dis. Colon Rectum. 2013;56:219-25) suggests that Patient Safety Indicator cases are really a reflection of surgical complexity and not unsafe care at all.
Finally, other discussants criticized the study for failing to tie satisfaction to patient outcomes; for failing to control for factors influencing patient satisfaction such as age, sex, or social status; and for not looking at geographic differences or nursing-to-staff ratios.
The complete manuscript of this study and its presentation at the American Surgical Association’s 134th Annual Meeting, April 2014, in Boston, is anticipated to be published in the Annals of Surgery, pending editorial review.
Dr. Kennedy reported no conflicting interests.
BOSTON – Hospital size and operative volume were significantly associated with satisfaction among general surgery patients in an analysis of 171 U.S. hospitals.
Surprisingly, all other safety and effectiveness measures, with the exception of low hospital mortality index, did not reliably reflect patient satisfaction, "indicating that the system plays perhaps a bigger role than anything else we can do," Dr. Gregory D. Kennedy said at the annual meeting of the American Surgical Association.
Moreover, a clean room and well-controlled pain were the best predictors of high patient satisfaction.
If it’s "the quality of the hotel, not the quality of the surgeon that drives patient satisfaction," and given that this is tied to reimbursement, what should the message be to hospital CEOs? asked discussant Dr. John J. Ricotta, chief of surgery at MedStar Washington (D.C.) Hospital Center.
Dr. Kennedy said the message he takes to the C-suite is that patient satisfaction cannot be a surrogate marker for safety and effectiveness or the only measure of quality because, in doing the right thing, surgeons often make patients unhappy. As a colorectal surgeon, he said he has unhappy patients every day, and remarked that he sometimes feels like a used car salesman where the only thing that he worries about is whether the patient is having a good experience when they drive off the lot, not whether it’s a safe, reliable car.
Dr. Kennedy, vice chair of quality at the University of Wisconsin School of Medicine, Madison, suggested that future quality measures also may need to make the distinction between satisfied and engaged, well-informed patients because a disengaged patient can be highly satisfied, while a highly engaged patient may not.
For the current study, the investigators examined federal Hospital Consumer Assessment Healthcare Providers and Systems (HCAHPS) survey results from 171 hospitals in the University Health System Consortium database from 2011 to 2012. Patients can check one of four boxes for each question on the 27-item survey, with high satisfaction defined as median responses above the 75th percentile on the top box score. This cutoff was used because the Centers for Medicare & Medicaid Services, which developed the HCAHPS, uses only the top box score, Dr. Kennedy explained.
The median hospital size was 421 beds (range, 25-1,280 beds), the median operative volume was 6,341 cases (range, 192-24,258 cases), and the mortality index was 0.83 (range, 0-2.61).
In all, 62% of high-volume hospitals, defined as those with an operative volume above the median, achieved high patient satisfaction, compared with 38% of low-volume hospitals (P less than .001). Similar results were seen for operative volume, he said.
Other system measures such as number of ICU cases and Surgical Care Improvement Project (SCIP) compliance were not associated with high HCAHPS scores.
Among patient safety indicators, only low mortality index was associated with high satisfaction (P less than .001), while complications, early mortality, and overall mortality were not.
Interestingly, hospitals with a higher number of Patient Safety Indicator cases – those involving accidental puncture, laceration, and venous thromboembolism – had higher rates of patient satisfaction, "suggesting that unsafe care is perhaps correlated with high satisfaction," Dr. Kennedy said.
Discussant Dr. Fabrizio Michelassi, chair of surgery at Weill Cornell Medical College and surgeon-in-chief, New York–Presbyterian Hospital/Weill Cornell Medical Center, in New York City, questioned whether "unsafe care gives more options for physicians to show their compassionate side," and said the overall findings are not that surprising to practicing surgeons, who frequently hear patient complaints, despite having performed a quality operation.
Dr. Kennedy said a recent paper from the Cleveland Clinic (Dis. Colon Rectum. 2013;56:219-25) suggests that Patient Safety Indicator cases are really a reflection of surgical complexity and not unsafe care at all.
Finally, other discussants criticized the study for failing to tie satisfaction to patient outcomes; for failing to control for factors influencing patient satisfaction such as age, sex, or social status; and for not looking at geographic differences or nursing-to-staff ratios.
The complete manuscript of this study and its presentation at the American Surgical Association’s 134th Annual Meeting, April 2014, in Boston, is anticipated to be published in the Annals of Surgery, pending editorial review.
Dr. Kennedy reported no conflicting interests.
BOSTON – Hospital size and operative volume were significantly associated with satisfaction among general surgery patients in an analysis of 171 U.S. hospitals.
Surprisingly, all other safety and effectiveness measures, with the exception of low hospital mortality index, did not reliably reflect patient satisfaction, "indicating that the system plays perhaps a bigger role than anything else we can do," Dr. Gregory D. Kennedy said at the annual meeting of the American Surgical Association.
Moreover, a clean room and well-controlled pain were the best predictors of high patient satisfaction.
If it’s "the quality of the hotel, not the quality of the surgeon that drives patient satisfaction," and given that this is tied to reimbursement, what should the message be to hospital CEOs? asked discussant Dr. John J. Ricotta, chief of surgery at MedStar Washington (D.C.) Hospital Center.
Dr. Kennedy said the message he takes to the C-suite is that patient satisfaction cannot be a surrogate marker for safety and effectiveness or the only measure of quality because, in doing the right thing, surgeons often make patients unhappy. As a colorectal surgeon, he said he has unhappy patients every day, and remarked that he sometimes feels like a used car salesman where the only thing that he worries about is whether the patient is having a good experience when they drive off the lot, not whether it’s a safe, reliable car.
Dr. Kennedy, vice chair of quality at the University of Wisconsin School of Medicine, Madison, suggested that future quality measures also may need to make the distinction between satisfied and engaged, well-informed patients because a disengaged patient can be highly satisfied, while a highly engaged patient may not.
For the current study, the investigators examined federal Hospital Consumer Assessment Healthcare Providers and Systems (HCAHPS) survey results from 171 hospitals in the University Health System Consortium database from 2011 to 2012. Patients can check one of four boxes for each question on the 27-item survey, with high satisfaction defined as median responses above the 75th percentile on the top box score. This cutoff was used because the Centers for Medicare & Medicaid Services, which developed the HCAHPS, uses only the top box score, Dr. Kennedy explained.
The median hospital size was 421 beds (range, 25-1,280 beds), the median operative volume was 6,341 cases (range, 192-24,258 cases), and the mortality index was 0.83 (range, 0-2.61).
In all, 62% of high-volume hospitals, defined as those with an operative volume above the median, achieved high patient satisfaction, compared with 38% of low-volume hospitals (P less than .001). Similar results were seen for operative volume, he said.
Other system measures such as number of ICU cases and Surgical Care Improvement Project (SCIP) compliance were not associated with high HCAHPS scores.
Among patient safety indicators, only low mortality index was associated with high satisfaction (P less than .001), while complications, early mortality, and overall mortality were not.
Interestingly, hospitals with a higher number of Patient Safety Indicator cases – those involving accidental puncture, laceration, and venous thromboembolism – had higher rates of patient satisfaction, "suggesting that unsafe care is perhaps correlated with high satisfaction," Dr. Kennedy said.
Discussant Dr. Fabrizio Michelassi, chair of surgery at Weill Cornell Medical College and surgeon-in-chief, New York–Presbyterian Hospital/Weill Cornell Medical Center, in New York City, questioned whether "unsafe care gives more options for physicians to show their compassionate side," and said the overall findings are not that surprising to practicing surgeons, who frequently hear patient complaints, despite having performed a quality operation.
Dr. Kennedy said a recent paper from the Cleveland Clinic (Dis. Colon Rectum. 2013;56:219-25) suggests that Patient Safety Indicator cases are really a reflection of surgical complexity and not unsafe care at all.
Finally, other discussants criticized the study for failing to tie satisfaction to patient outcomes; for failing to control for factors influencing patient satisfaction such as age, sex, or social status; and for not looking at geographic differences or nursing-to-staff ratios.
The complete manuscript of this study and its presentation at the American Surgical Association’s 134th Annual Meeting, April 2014, in Boston, is anticipated to be published in the Annals of Surgery, pending editorial review.
Dr. Kennedy reported no conflicting interests.
AT ASA 2014
Major finding: In the sample, 62% of high-volume hospitals achieved high patient satisfaction, vs. 38% of low-volume hospitals. Other system measures such as number of ICU cases and Surgical Care Improvement Project (compliance were not associated with high HCAHPS scores.
Data source: A retrospective analysis of HCAHPS surveys at 171 U.S. hospitals.
Disclosures: Dr. Kennedy reported no conflicting interests.
CDC confirms first U.S. case of deadly Middle East Respiratory Syndrome
The Centers for Disease Control and Prevention has confirmed the first case of the deadly Middle East Respiratory Syndrome coronavirus in the United States.
The patient, a health care worker who recently returned from Riyadh, Saudi Arabia, is in stable condition in an Indiana hospital after being admitted with shortness of breath, cough, and fever on April 27.
No other cases have been reported beyond this single case, but investigators are just beginning to reach out to those who came in contact with the patient, said Dr. Anne Schuchat, assistant surgeon general and director of the CDC’s National Center for Immunization and Respiratory Diseases, during a press briefing.
That list could be quite long, as the patient flew on April 24 from Saudi Arabia to London, then on to Chicago, and then traveled by bus to Indiana. The patient is in good condition, and remains in isolation in Community Hospital in Munster, Indiana, according to a statement from the hospital.
Health officials would not release the name of the airline or bus line the patient traveled on.
Indiana state health officials took specimens from the patient, which were then confirmed late today by the CDC.
There is no specific treatment for the Middle East Respiratory Syndrome coronavirus (MERS-CoV), which has a case fatality rate of about 30% among symptomatic patients.
Since the virus was first reported in 2012 in Saudi Arabia, 263 cases have been confirmed in 12 countries, with 92 deaths. More than 100 other patients have been confirmed and reported by their ministries of health, but are not included in the World Health Organization tally, Dr. Schuchat said.
The Indiana case represents a "very low risk to the broader general public" because MERS-CoV has not shown the ability to spread easily from person to person in the community setting, she said.
"It is very concerning that the virus has spread in hospitals, and we should not be surprised if additional cases are identified among the health care workers who had close contact with this patient before the patient was isolated and special precautions were implemented," she went on to say.
An outbreak in the spring of 2013 involved 23 confirmed cases at four healthcare facilities, and another surge in cases beginning in March of this year has been identified.
It is unknown whether this represents seasonal variation or mutations in the virus that may make it more easily transmissible. At least one isolate has been sequenced by polymerase chain reaction from a patient in Saudi Arabia, and there were no changes in the virus, Dr. Schuchat said.
"With the SARS virus 10, 11 years ago, we did believe there was a change in the virus that led to more explosive transmission. So we’ll continue to look at that, but we don’t have data right now to confirm that that has happened," she said.
For now, the CDC is not advising people change their travel plans, but is recommending that people who develop a fever, cough, or shortness of breath within 14 days after traveling in or near the Arabian Peninsula see a health care provider, Dr. Schuchat said. Others that should monitor their symptoms include those who have close contact with someone who has these symptoms and travel history.
The CDC is also advising people to use basic respiratory illness precautions such as frequent hand washing, avoiding close contact with people who are sick, and disinfecting frequently touched surfaces.
No specific precautions were given for pediatricians or children, other than to ask for a travel history in patients with respiratory symptoms. The youngest case of MERS-CoV was in a 2 year-old, but the median age is around 51 years, she said.
Dr. Schuchat stressed that guidance may change as this "very fluid" situation evolves. The Indiana Department of Health and the CDC will share additional information on their Web sites and host additional press conferences, as needed, she said.
For more information on Middle East Respiratory Syndrome, go to http://www.cdc.gov/coronavirus/mers/index.html.
The Centers for Disease Control and Prevention has confirmed the first case of the deadly Middle East Respiratory Syndrome coronavirus in the United States.
The patient, a health care worker who recently returned from Riyadh, Saudi Arabia, is in stable condition in an Indiana hospital after being admitted with shortness of breath, cough, and fever on April 27.
No other cases have been reported beyond this single case, but investigators are just beginning to reach out to those who came in contact with the patient, said Dr. Anne Schuchat, assistant surgeon general and director of the CDC’s National Center for Immunization and Respiratory Diseases, during a press briefing.
That list could be quite long, as the patient flew on April 24 from Saudi Arabia to London, then on to Chicago, and then traveled by bus to Indiana. The patient is in good condition, and remains in isolation in Community Hospital in Munster, Indiana, according to a statement from the hospital.
Health officials would not release the name of the airline or bus line the patient traveled on.
Indiana state health officials took specimens from the patient, which were then confirmed late today by the CDC.
There is no specific treatment for the Middle East Respiratory Syndrome coronavirus (MERS-CoV), which has a case fatality rate of about 30% among symptomatic patients.
Since the virus was first reported in 2012 in Saudi Arabia, 263 cases have been confirmed in 12 countries, with 92 deaths. More than 100 other patients have been confirmed and reported by their ministries of health, but are not included in the World Health Organization tally, Dr. Schuchat said.
The Indiana case represents a "very low risk to the broader general public" because MERS-CoV has not shown the ability to spread easily from person to person in the community setting, she said.
"It is very concerning that the virus has spread in hospitals, and we should not be surprised if additional cases are identified among the health care workers who had close contact with this patient before the patient was isolated and special precautions were implemented," she went on to say.
An outbreak in the spring of 2013 involved 23 confirmed cases at four healthcare facilities, and another surge in cases beginning in March of this year has been identified.
It is unknown whether this represents seasonal variation or mutations in the virus that may make it more easily transmissible. At least one isolate has been sequenced by polymerase chain reaction from a patient in Saudi Arabia, and there were no changes in the virus, Dr. Schuchat said.
"With the SARS virus 10, 11 years ago, we did believe there was a change in the virus that led to more explosive transmission. So we’ll continue to look at that, but we don’t have data right now to confirm that that has happened," she said.
For now, the CDC is not advising people change their travel plans, but is recommending that people who develop a fever, cough, or shortness of breath within 14 days after traveling in or near the Arabian Peninsula see a health care provider, Dr. Schuchat said. Others that should monitor their symptoms include those who have close contact with someone who has these symptoms and travel history.
The CDC is also advising people to use basic respiratory illness precautions such as frequent hand washing, avoiding close contact with people who are sick, and disinfecting frequently touched surfaces.
No specific precautions were given for pediatricians or children, other than to ask for a travel history in patients with respiratory symptoms. The youngest case of MERS-CoV was in a 2 year-old, but the median age is around 51 years, she said.
Dr. Schuchat stressed that guidance may change as this "very fluid" situation evolves. The Indiana Department of Health and the CDC will share additional information on their Web sites and host additional press conferences, as needed, she said.
For more information on Middle East Respiratory Syndrome, go to http://www.cdc.gov/coronavirus/mers/index.html.
The Centers for Disease Control and Prevention has confirmed the first case of the deadly Middle East Respiratory Syndrome coronavirus in the United States.
The patient, a health care worker who recently returned from Riyadh, Saudi Arabia, is in stable condition in an Indiana hospital after being admitted with shortness of breath, cough, and fever on April 27.
No other cases have been reported beyond this single case, but investigators are just beginning to reach out to those who came in contact with the patient, said Dr. Anne Schuchat, assistant surgeon general and director of the CDC’s National Center for Immunization and Respiratory Diseases, during a press briefing.
That list could be quite long, as the patient flew on April 24 from Saudi Arabia to London, then on to Chicago, and then traveled by bus to Indiana. The patient is in good condition, and remains in isolation in Community Hospital in Munster, Indiana, according to a statement from the hospital.
Health officials would not release the name of the airline or bus line the patient traveled on.
Indiana state health officials took specimens from the patient, which were then confirmed late today by the CDC.
There is no specific treatment for the Middle East Respiratory Syndrome coronavirus (MERS-CoV), which has a case fatality rate of about 30% among symptomatic patients.
Since the virus was first reported in 2012 in Saudi Arabia, 263 cases have been confirmed in 12 countries, with 92 deaths. More than 100 other patients have been confirmed and reported by their ministries of health, but are not included in the World Health Organization tally, Dr. Schuchat said.
The Indiana case represents a "very low risk to the broader general public" because MERS-CoV has not shown the ability to spread easily from person to person in the community setting, she said.
"It is very concerning that the virus has spread in hospitals, and we should not be surprised if additional cases are identified among the health care workers who had close contact with this patient before the patient was isolated and special precautions were implemented," she went on to say.
An outbreak in the spring of 2013 involved 23 confirmed cases at four healthcare facilities, and another surge in cases beginning in March of this year has been identified.
It is unknown whether this represents seasonal variation or mutations in the virus that may make it more easily transmissible. At least one isolate has been sequenced by polymerase chain reaction from a patient in Saudi Arabia, and there were no changes in the virus, Dr. Schuchat said.
"With the SARS virus 10, 11 years ago, we did believe there was a change in the virus that led to more explosive transmission. So we’ll continue to look at that, but we don’t have data right now to confirm that that has happened," she said.
For now, the CDC is not advising people change their travel plans, but is recommending that people who develop a fever, cough, or shortness of breath within 14 days after traveling in or near the Arabian Peninsula see a health care provider, Dr. Schuchat said. Others that should monitor their symptoms include those who have close contact with someone who has these symptoms and travel history.
The CDC is also advising people to use basic respiratory illness precautions such as frequent hand washing, avoiding close contact with people who are sick, and disinfecting frequently touched surfaces.
No specific precautions were given for pediatricians or children, other than to ask for a travel history in patients with respiratory symptoms. The youngest case of MERS-CoV was in a 2 year-old, but the median age is around 51 years, she said.
Dr. Schuchat stressed that guidance may change as this "very fluid" situation evolves. The Indiana Department of Health and the CDC will share additional information on their Web sites and host additional press conferences, as needed, she said.
For more information on Middle East Respiratory Syndrome, go to http://www.cdc.gov/coronavirus/mers/index.html.
FROM A CDC TELEBRIEFING
OA-related walking disability on par with diabetes as CVD risk factor
PARIS – Greater walking disability is an independent predictor of all-cause death and major cardiovascular events in patients with symptomatic hip or knee osteoarthritis, according to a population-based study that confirms previous research.
For the composite cardiovascular disease (CVD) outcome of hospitalization for angina, acute myocardial infarction, coronary revascularization, congestive heart failure, stroke, or transient ischemic attack, the risk associated with a Health Assessment Questionnaire (HAQ) walking disability score of 2 was the same as that of diabetes and pre-existing CVD.
Total joint replacement reduced those risks by about 40%, Dr. Gillian A. Hawker said at the World Congress on Osteoarthritis (OA).
"I think the key piece here is that these data at least suggest that walking disability is a cardiovascular risk factor similar to diabetes," she said. "When we’re talking about risk assessment for cardiovascular outcomes, which everyone does pretty routinely in medicine, walking disability, which is really osteoarthritis, is one of the risk factors that they should be assessing ... If we can just start asking, hopefully it will get us thinking about, ‘Wow, this person has OA,’ and we need to start assessing it."
Session comoderator Ana M. Valdes, Ph.D., of the University of Nottingham (England) commented that the results are fascinating because this is not the first study to identify these associations. In 2011, British investigators reported that all-cause mortality, particularly due to CVD causes, was significantly related to baseline walking disability in patients with knee or hip OA (Br. Med. J. 2011;342:d1165).
"One of the things that happens often with OA is that it’s seen as just pain and if we can give them plenty of relief, they’ll be OK, but it’s actually life-threatening because if we do not address their disability, they are at risk of dying," Dr. Valdes said in an interview. "I think this should be put on the front pages of the weekly medical journals, so that people are aware of the dangers of not treating osteoarthritis properly."
Comorbidity is a barrier to OA care, and 90% of those aged 65 years and older with OA have at least one other chronic condition, such as diabetes and heart disease, observed Dr. Hawker, professor of medicine at the University of Toronto and physician-in-chief of medicine, Women’s College Hospital in Toronto.
Her group reported in a separate presentation at the meeting that walking disability also raises the risk for diabetes complications in those with both OA and diabetes.
For the current population-based study, the investigators linked provincial health administrative databases to baseline surveys from a population cohort of 2,156 patients with symptomatic moderate to severe OA who were recruited in 1996-1998 through a screening survey in Ontario.
Their mean age was 71 years (all were 55 years or older), 72% were female, 34% obese, 20% had diabetes, and 40% had prebaseline CVD. Their average HAQ walking disability score was 2 on a 3-point scale, and they had a mean WOMAC (Western Ontario and McMaster Universities Osteoarthritis Index) summary score of 41. Overall, 44% used a walking aid. (A walking disability score of 2 corresponds with walking outdoors on flat ground with much difficulty, whereas a score of 3 means the person is unable to do it.)
After a median follow-up period of 13.2 years, 57% of patients had died, and 38% experienced the composite CVD outcome with a median of 9.2 years follow-up.
In multivariable analysis, there was a very clear dose-response, where increased walking disability was associated with increasing risk of all-cause death, Dr. Hawker said. The adjusted hazard ratio per unit increase in HAQ walking score was 1.30 (P less than .001), after controlling for confounders including age, sex, preexisting CVD, diabetes, hypertension, body mass index, and smoking status.
A HAQ walking score of 2 was associated with an adjusted hazard ratio of 1.69, which was actually greater than that associated with preexisting cardiovascular disease or diabetes, she said.
For the composite CVD outcome, there was also a significant association with HAQ walking score (adjusted HR, 1.17; P = .001).
Sensitivity analyses performed in 402 patients (18%) receiving postbaseline total joint arthroplasty (TJA ) showed that controlling for TJA did not change the effect of baseline disability but was protective for both all-cause death (HR, 0.62; P less than .001) and CVD events (HR, 0.66; P less than .001), Dr. Hawker said at the meeting sponsored by the Osteoarthritis Research Society International.
Although the data were not dichotomized, she noted that there was "beautiful discrimination" when they looked at the outcomes based on whether or not patients used a walking aid.
"Obviously, it’s nice to see a dose-response, but simply knowing that someone’s using a walking aid and getting them to the point where they’ve got improved walking ability, could go a long way," she said.
Though the study was not designed to address potential mechanisms, Dr. Hawker said she’s long argued that physical activity and mobility are a key player of downstream effects on fitness, blood pressure, glucose control, and ability to participate in self-management activities and physician visits. The potential role for pain, mood, and stress also can not be excluded.
One audience member asked whether walking disability may be a "cheap and dirty version" of the stress test.
Dr. Hawker responded that walking disability is a proxy for a lot of things and is definitely OA-related, adding, "Yes, I think it is a very cheap and dirty stress test. It’s a lovely thing that if we focused more explicitly on it in randomized trials, for instance as a primary outcome, or in rehabilitation strategies, it would have global benefit."
The authors reported no competing interests.
PARIS – Greater walking disability is an independent predictor of all-cause death and major cardiovascular events in patients with symptomatic hip or knee osteoarthritis, according to a population-based study that confirms previous research.
For the composite cardiovascular disease (CVD) outcome of hospitalization for angina, acute myocardial infarction, coronary revascularization, congestive heart failure, stroke, or transient ischemic attack, the risk associated with a Health Assessment Questionnaire (HAQ) walking disability score of 2 was the same as that of diabetes and pre-existing CVD.
Total joint replacement reduced those risks by about 40%, Dr. Gillian A. Hawker said at the World Congress on Osteoarthritis (OA).
"I think the key piece here is that these data at least suggest that walking disability is a cardiovascular risk factor similar to diabetes," she said. "When we’re talking about risk assessment for cardiovascular outcomes, which everyone does pretty routinely in medicine, walking disability, which is really osteoarthritis, is one of the risk factors that they should be assessing ... If we can just start asking, hopefully it will get us thinking about, ‘Wow, this person has OA,’ and we need to start assessing it."
Session comoderator Ana M. Valdes, Ph.D., of the University of Nottingham (England) commented that the results are fascinating because this is not the first study to identify these associations. In 2011, British investigators reported that all-cause mortality, particularly due to CVD causes, was significantly related to baseline walking disability in patients with knee or hip OA (Br. Med. J. 2011;342:d1165).
"One of the things that happens often with OA is that it’s seen as just pain and if we can give them plenty of relief, they’ll be OK, but it’s actually life-threatening because if we do not address their disability, they are at risk of dying," Dr. Valdes said in an interview. "I think this should be put on the front pages of the weekly medical journals, so that people are aware of the dangers of not treating osteoarthritis properly."
Comorbidity is a barrier to OA care, and 90% of those aged 65 years and older with OA have at least one other chronic condition, such as diabetes and heart disease, observed Dr. Hawker, professor of medicine at the University of Toronto and physician-in-chief of medicine, Women’s College Hospital in Toronto.
Her group reported in a separate presentation at the meeting that walking disability also raises the risk for diabetes complications in those with both OA and diabetes.
For the current population-based study, the investigators linked provincial health administrative databases to baseline surveys from a population cohort of 2,156 patients with symptomatic moderate to severe OA who were recruited in 1996-1998 through a screening survey in Ontario.
Their mean age was 71 years (all were 55 years or older), 72% were female, 34% obese, 20% had diabetes, and 40% had prebaseline CVD. Their average HAQ walking disability score was 2 on a 3-point scale, and they had a mean WOMAC (Western Ontario and McMaster Universities Osteoarthritis Index) summary score of 41. Overall, 44% used a walking aid. (A walking disability score of 2 corresponds with walking outdoors on flat ground with much difficulty, whereas a score of 3 means the person is unable to do it.)
After a median follow-up period of 13.2 years, 57% of patients had died, and 38% experienced the composite CVD outcome with a median of 9.2 years follow-up.
In multivariable analysis, there was a very clear dose-response, where increased walking disability was associated with increasing risk of all-cause death, Dr. Hawker said. The adjusted hazard ratio per unit increase in HAQ walking score was 1.30 (P less than .001), after controlling for confounders including age, sex, preexisting CVD, diabetes, hypertension, body mass index, and smoking status.
A HAQ walking score of 2 was associated with an adjusted hazard ratio of 1.69, which was actually greater than that associated with preexisting cardiovascular disease or diabetes, she said.
For the composite CVD outcome, there was also a significant association with HAQ walking score (adjusted HR, 1.17; P = .001).
Sensitivity analyses performed in 402 patients (18%) receiving postbaseline total joint arthroplasty (TJA ) showed that controlling for TJA did not change the effect of baseline disability but was protective for both all-cause death (HR, 0.62; P less than .001) and CVD events (HR, 0.66; P less than .001), Dr. Hawker said at the meeting sponsored by the Osteoarthritis Research Society International.
Although the data were not dichotomized, she noted that there was "beautiful discrimination" when they looked at the outcomes based on whether or not patients used a walking aid.
"Obviously, it’s nice to see a dose-response, but simply knowing that someone’s using a walking aid and getting them to the point where they’ve got improved walking ability, could go a long way," she said.
Though the study was not designed to address potential mechanisms, Dr. Hawker said she’s long argued that physical activity and mobility are a key player of downstream effects on fitness, blood pressure, glucose control, and ability to participate in self-management activities and physician visits. The potential role for pain, mood, and stress also can not be excluded.
One audience member asked whether walking disability may be a "cheap and dirty version" of the stress test.
Dr. Hawker responded that walking disability is a proxy for a lot of things and is definitely OA-related, adding, "Yes, I think it is a very cheap and dirty stress test. It’s a lovely thing that if we focused more explicitly on it in randomized trials, for instance as a primary outcome, or in rehabilitation strategies, it would have global benefit."
The authors reported no competing interests.
PARIS – Greater walking disability is an independent predictor of all-cause death and major cardiovascular events in patients with symptomatic hip or knee osteoarthritis, according to a population-based study that confirms previous research.
For the composite cardiovascular disease (CVD) outcome of hospitalization for angina, acute myocardial infarction, coronary revascularization, congestive heart failure, stroke, or transient ischemic attack, the risk associated with a Health Assessment Questionnaire (HAQ) walking disability score of 2 was the same as that of diabetes and pre-existing CVD.
Total joint replacement reduced those risks by about 40%, Dr. Gillian A. Hawker said at the World Congress on Osteoarthritis (OA).
"I think the key piece here is that these data at least suggest that walking disability is a cardiovascular risk factor similar to diabetes," she said. "When we’re talking about risk assessment for cardiovascular outcomes, which everyone does pretty routinely in medicine, walking disability, which is really osteoarthritis, is one of the risk factors that they should be assessing ... If we can just start asking, hopefully it will get us thinking about, ‘Wow, this person has OA,’ and we need to start assessing it."
Session comoderator Ana M. Valdes, Ph.D., of the University of Nottingham (England) commented that the results are fascinating because this is not the first study to identify these associations. In 2011, British investigators reported that all-cause mortality, particularly due to CVD causes, was significantly related to baseline walking disability in patients with knee or hip OA (Br. Med. J. 2011;342:d1165).
"One of the things that happens often with OA is that it’s seen as just pain and if we can give them plenty of relief, they’ll be OK, but it’s actually life-threatening because if we do not address their disability, they are at risk of dying," Dr. Valdes said in an interview. "I think this should be put on the front pages of the weekly medical journals, so that people are aware of the dangers of not treating osteoarthritis properly."
Comorbidity is a barrier to OA care, and 90% of those aged 65 years and older with OA have at least one other chronic condition, such as diabetes and heart disease, observed Dr. Hawker, professor of medicine at the University of Toronto and physician-in-chief of medicine, Women’s College Hospital in Toronto.
Her group reported in a separate presentation at the meeting that walking disability also raises the risk for diabetes complications in those with both OA and diabetes.
For the current population-based study, the investigators linked provincial health administrative databases to baseline surveys from a population cohort of 2,156 patients with symptomatic moderate to severe OA who were recruited in 1996-1998 through a screening survey in Ontario.
Their mean age was 71 years (all were 55 years or older), 72% were female, 34% obese, 20% had diabetes, and 40% had prebaseline CVD. Their average HAQ walking disability score was 2 on a 3-point scale, and they had a mean WOMAC (Western Ontario and McMaster Universities Osteoarthritis Index) summary score of 41. Overall, 44% used a walking aid. (A walking disability score of 2 corresponds with walking outdoors on flat ground with much difficulty, whereas a score of 3 means the person is unable to do it.)
After a median follow-up period of 13.2 years, 57% of patients had died, and 38% experienced the composite CVD outcome with a median of 9.2 years follow-up.
In multivariable analysis, there was a very clear dose-response, where increased walking disability was associated with increasing risk of all-cause death, Dr. Hawker said. The adjusted hazard ratio per unit increase in HAQ walking score was 1.30 (P less than .001), after controlling for confounders including age, sex, preexisting CVD, diabetes, hypertension, body mass index, and smoking status.
A HAQ walking score of 2 was associated with an adjusted hazard ratio of 1.69, which was actually greater than that associated with preexisting cardiovascular disease or diabetes, she said.
For the composite CVD outcome, there was also a significant association with HAQ walking score (adjusted HR, 1.17; P = .001).
Sensitivity analyses performed in 402 patients (18%) receiving postbaseline total joint arthroplasty (TJA ) showed that controlling for TJA did not change the effect of baseline disability but was protective for both all-cause death (HR, 0.62; P less than .001) and CVD events (HR, 0.66; P less than .001), Dr. Hawker said at the meeting sponsored by the Osteoarthritis Research Society International.
Although the data were not dichotomized, she noted that there was "beautiful discrimination" when they looked at the outcomes based on whether or not patients used a walking aid.
"Obviously, it’s nice to see a dose-response, but simply knowing that someone’s using a walking aid and getting them to the point where they’ve got improved walking ability, could go a long way," she said.
Though the study was not designed to address potential mechanisms, Dr. Hawker said she’s long argued that physical activity and mobility are a key player of downstream effects on fitness, blood pressure, glucose control, and ability to participate in self-management activities and physician visits. The potential role for pain, mood, and stress also can not be excluded.
One audience member asked whether walking disability may be a "cheap and dirty version" of the stress test.
Dr. Hawker responded that walking disability is a proxy for a lot of things and is definitely OA-related, adding, "Yes, I think it is a very cheap and dirty stress test. It’s a lovely thing that if we focused more explicitly on it in randomized trials, for instance as a primary outcome, or in rehabilitation strategies, it would have global benefit."
The authors reported no competing interests.
AT OARSI 2014
Key clinical point: A study confirming previous research suggests that a high level of walking disability in OA patients should be considered a strong risk factor for CVD.
Major finding: The adjusted hazard ratio per unit increase in HAQ walking score was 1.30 (P less than .001) for all-cause mortality and 1.17 (P = .001) for composite CVD events.
Data source: A population-based, cohort study in 2,156 hip and knee OA patients.
Disclosures: The authors reported no competing interests.