User login
Complication gap narrows between low- and high-volume bariatric centers
WASHINGTON – Annual case volume appears to be falling out as a safety factor in bariatric surgery, Dr. Oliver Varban said at the annual clinical congress of the American College of Surgeons.
In the early days of the procedure, facilities that performed more than 125 operations each year had significantly better safety outcomes than did those performing fewer operations. But that difference is fading, particularly as laparoscopy continues to supplant open surgery, said Dr. Varban of the University of Michigan Health Systems, Ann Arbor.
"Over time, safety is improving in both high- and low-volume centers, with lower rates of complications, morbidity, and mortality. The inverse relationship does still persist, but that effect is attenuating."
Dr. Varban conducted a review of more than 270,000 bariatric surgery procedures performed in 12 hospitals from 2004 to 2009. He separated the hospitals by annual case volume: 125 or more and less than 125 per year.
In each year, about two-thirds of the procedures were completed in high-volume hospitals. The type of surgery also varied over the study period. During 2004-2005, laparoscopic and open procedures were about equally common. By 2006-2007, laparoscopic operations made up 65% of all bariatric procedures, and that held steady through 2008-2009.
The type of procedure evolved as well, Dr. Varban noted. In 2004-2005, gastric banding comprised about 5% of the operations. By 2006-2007, that had risen to 20%, and by 2008-2009, the number was close to 30%.
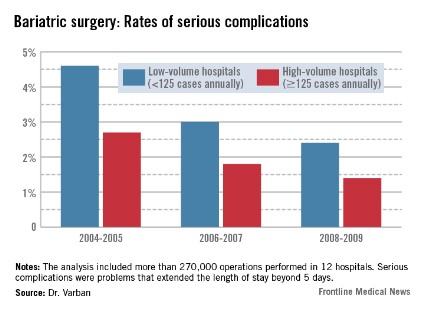
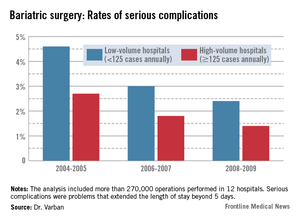
In the first era, low-volume hospitals had significantly higher rates of any complication than did high-volume centers (9.3% vs. 6%). By 2006-2007, the difference had narrowed but was still statistically significant (7% vs. 4.8%). By 2008-2009, the difference was no longer significant (5.6% vs. 4.5%).
A multivariate analysis that controlled for type of surgery and patient demographics found a similar trend. The risk of any complication remained significantly elevated at low-volume centers during all three periods (odds ratio, 1.33 in 2004-2005; 1.35 in 2006-2007; and 1.21 in 2008-2009).
The pattern of serious complications (problems that extended the length of stay beyond 5 days) was similar, with a rate of 4.6% vs. 2.7% in the early era; 3% vs. 1.8% in the middle era; and 2.4% vs. 1.4% in the final era. The risk of serious complications was significantly higher in the low-volume group in every era (OR 1.35, 1.43, and 1.44, respectively).
The rates of reoperation were higher in low-volume centers in every era, and declined as time went on. However, the difference between low- and high-volume centers in terms of reoperation rates was nonsignificant at every time point (1.5% vs. 1.06%; 1% vs. 0.75%; 0.78% vs. 0.67%). Similarly, the adjusted odds ratios were nonsignificant (OR 1.22, 1.28, and 1.20).
Although overall the mortality rates were low and remained low, they declined significantly in both groups over the study period (from 0.22% vs. 0.1% and 0.09% vs. 0.04%). Only in the first era was the difference statistically significant, with an adjusted OR of 1.71.
Dr. Varban had no financial disclosures.
WASHINGTON – Annual case volume appears to be falling out as a safety factor in bariatric surgery, Dr. Oliver Varban said at the annual clinical congress of the American College of Surgeons.
In the early days of the procedure, facilities that performed more than 125 operations each year had significantly better safety outcomes than did those performing fewer operations. But that difference is fading, particularly as laparoscopy continues to supplant open surgery, said Dr. Varban of the University of Michigan Health Systems, Ann Arbor.

"Over time, safety is improving in both high- and low-volume centers, with lower rates of complications, morbidity, and mortality. The inverse relationship does still persist, but that effect is attenuating."
Dr. Varban conducted a review of more than 270,000 bariatric surgery procedures performed in 12 hospitals from 2004 to 2009. He separated the hospitals by annual case volume: 125 or more and less than 125 per year.
In each year, about two-thirds of the procedures were completed in high-volume hospitals. The type of surgery also varied over the study period. During 2004-2005, laparoscopic and open procedures were about equally common. By 2006-2007, laparoscopic operations made up 65% of all bariatric procedures, and that held steady through 2008-2009.
The type of procedure evolved as well, Dr. Varban noted. In 2004-2005, gastric banding comprised about 5% of the operations. By 2006-2007, that had risen to 20%, and by 2008-2009, the number was close to 30%.
In the first era, low-volume hospitals had significantly higher rates of any complication than did high-volume centers (9.3% vs. 6%). By 2006-2007, the difference had narrowed but was still statistically significant (7% vs. 4.8%). By 2008-2009, the difference was no longer significant (5.6% vs. 4.5%).
A multivariate analysis that controlled for type of surgery and patient demographics found a similar trend. The risk of any complication remained significantly elevated at low-volume centers during all three periods (odds ratio, 1.33 in 2004-2005; 1.35 in 2006-2007; and 1.21 in 2008-2009).
The pattern of serious complications (problems that extended the length of stay beyond 5 days) was similar, with a rate of 4.6% vs. 2.7% in the early era; 3% vs. 1.8% in the middle era; and 2.4% vs. 1.4% in the final era. The risk of serious complications was significantly higher in the low-volume group in every era (OR 1.35, 1.43, and 1.44, respectively).
The rates of reoperation were higher in low-volume centers in every era, and declined as time went on. However, the difference between low- and high-volume centers in terms of reoperation rates was nonsignificant at every time point (1.5% vs. 1.06%; 1% vs. 0.75%; 0.78% vs. 0.67%). Similarly, the adjusted odds ratios were nonsignificant (OR 1.22, 1.28, and 1.20).
Although overall the mortality rates were low and remained low, they declined significantly in both groups over the study period (from 0.22% vs. 0.1% and 0.09% vs. 0.04%). Only in the first era was the difference statistically significant, with an adjusted OR of 1.71.
Dr. Varban had no financial disclosures.
WASHINGTON – Annual case volume appears to be falling out as a safety factor in bariatric surgery, Dr. Oliver Varban said at the annual clinical congress of the American College of Surgeons.
In the early days of the procedure, facilities that performed more than 125 operations each year had significantly better safety outcomes than did those performing fewer operations. But that difference is fading, particularly as laparoscopy continues to supplant open surgery, said Dr. Varban of the University of Michigan Health Systems, Ann Arbor.

"Over time, safety is improving in both high- and low-volume centers, with lower rates of complications, morbidity, and mortality. The inverse relationship does still persist, but that effect is attenuating."
Dr. Varban conducted a review of more than 270,000 bariatric surgery procedures performed in 12 hospitals from 2004 to 2009. He separated the hospitals by annual case volume: 125 or more and less than 125 per year.
In each year, about two-thirds of the procedures were completed in high-volume hospitals. The type of surgery also varied over the study period. During 2004-2005, laparoscopic and open procedures were about equally common. By 2006-2007, laparoscopic operations made up 65% of all bariatric procedures, and that held steady through 2008-2009.
The type of procedure evolved as well, Dr. Varban noted. In 2004-2005, gastric banding comprised about 5% of the operations. By 2006-2007, that had risen to 20%, and by 2008-2009, the number was close to 30%.
In the first era, low-volume hospitals had significantly higher rates of any complication than did high-volume centers (9.3% vs. 6%). By 2006-2007, the difference had narrowed but was still statistically significant (7% vs. 4.8%). By 2008-2009, the difference was no longer significant (5.6% vs. 4.5%).
A multivariate analysis that controlled for type of surgery and patient demographics found a similar trend. The risk of any complication remained significantly elevated at low-volume centers during all three periods (odds ratio, 1.33 in 2004-2005; 1.35 in 2006-2007; and 1.21 in 2008-2009).
The pattern of serious complications (problems that extended the length of stay beyond 5 days) was similar, with a rate of 4.6% vs. 2.7% in the early era; 3% vs. 1.8% in the middle era; and 2.4% vs. 1.4% in the final era. The risk of serious complications was significantly higher in the low-volume group in every era (OR 1.35, 1.43, and 1.44, respectively).
The rates of reoperation were higher in low-volume centers in every era, and declined as time went on. However, the difference between low- and high-volume centers in terms of reoperation rates was nonsignificant at every time point (1.5% vs. 1.06%; 1% vs. 0.75%; 0.78% vs. 0.67%). Similarly, the adjusted odds ratios were nonsignificant (OR 1.22, 1.28, and 1.20).
Although overall the mortality rates were low and remained low, they declined significantly in both groups over the study period (from 0.22% vs. 0.1% and 0.09% vs. 0.04%). Only in the first era was the difference statistically significant, with an adjusted OR of 1.71.
Dr. Varban had no financial disclosures.
AT THE ACS CLINICAL CONGRESS
Major finding: From 2004 to 2009, the rates of serious complications, reoperation, and mortality improved in low-volume hospitals, bringing their results closer to those seen in high-volume centers.
Data source: The study included data on more than 270,000 bariatric surgical procedures.
Disclosures: Dr. Varban had no financial disclosures.
Single-incision sling may help stress urinary incontinence
NATIONAL HARBOR, MD. – After 12 months, an adjustable, single-incision sling significantly improved clinical and quality of life measures associated with stress urinary incontinence in women.
The Altis sling decreased the Urinary Distress Index (UDI) from a mean of 55 to a mean of 10, Dr. Douglas Van Drie said at a meeting sponsored by the AAGL. The Incontinence Impact Questionnaire (ILQ-7) showed similar improvements at the interim assessment of the device’s 2-year safety and efficacy study.
The study was sponsored by Coloplast, which makes the sling, with input and monitoring by the Food and Drug Administration. Altis was approved in November 2012 based on an investigational device exemption study, which included implant data. The FDA efficacy requirement was a 50% decrease in pad weight by 6 months.
According to the company website, "Altis is a unique, minimally invasive sling that combines integrated two-way tensioning with lightweight sling material to provide strength, security, and adjustability."
Physicians are divided on their thoughts about a single-incision sling, said Dr. Van Drie, a urogynecologist in group practice in Grand Rapids, Mich. "Those [physicians] who use them are advocates for their safety and simplicity, and the ability to insert them in the office. They have been adopted in different areas of the world as an option for doing simplified, less costly incontinence surgery. The argument against is questions about their staying power – will the effect hold up long term?" he said.
Even though the Altis is a single-incision sling, it has a "very secure" anchoring system, Dr. Van Drie said. The anchor not only goes into the obturator internus, but into the membrane and the obturator externus.
The study involved 113 women, with a mean age of 54 years. Their histories included stress incontinence with hypermobility (81%), without hypermobility (19%), mixed incontinence (37%), and overactive bladder (5%).
Most of the procedures were performed during an inpatient hospital stay (59%); however, 24% were performed at an ambulatory surgical center and 17%, in the physician’s office. General anesthesia was used in 52%, spinal in 3%, and local in 45%.
At 12 months, 90% of patients had at least a 50% reduction in pad weight, and 90% had a negative cough stress test result. The UDI decreased by a mean of 46 points, and the IIQ-7 score, by a mean of 47 points.
There were 11 device-related failures in eight patients. These included one each of urinary retention, urinary tract infection, decreased urine stream, dyspareunia, inflammation, worsening of overactive bladder, and voiding dysfunction. There were four mesh extrusions (3.5%), all less than 3 cm. Two patients with extrusion were smokers, and one was diabetic.
Serious adverse events occurred in three patients: One with a hematoma, one patient who needed transfer to the operating room because of anxiety during repair of a 2-mm mesh extrusion, and one patient whose adverse event was changed to a severe adverse event when she moved out of the study. There were no unanticipated device effects, Dr. Van Drie noted.
Dr. Van Drie is a consultant for Coloplast and has received research money and grants from the company.
NATIONAL HARBOR, MD. – After 12 months, an adjustable, single-incision sling significantly improved clinical and quality of life measures associated with stress urinary incontinence in women.
The Altis sling decreased the Urinary Distress Index (UDI) from a mean of 55 to a mean of 10, Dr. Douglas Van Drie said at a meeting sponsored by the AAGL. The Incontinence Impact Questionnaire (ILQ-7) showed similar improvements at the interim assessment of the device’s 2-year safety and efficacy study.
The study was sponsored by Coloplast, which makes the sling, with input and monitoring by the Food and Drug Administration. Altis was approved in November 2012 based on an investigational device exemption study, which included implant data. The FDA efficacy requirement was a 50% decrease in pad weight by 6 months.
According to the company website, "Altis is a unique, minimally invasive sling that combines integrated two-way tensioning with lightweight sling material to provide strength, security, and adjustability."
Physicians are divided on their thoughts about a single-incision sling, said Dr. Van Drie, a urogynecologist in group practice in Grand Rapids, Mich. "Those [physicians] who use them are advocates for their safety and simplicity, and the ability to insert them in the office. They have been adopted in different areas of the world as an option for doing simplified, less costly incontinence surgery. The argument against is questions about their staying power – will the effect hold up long term?" he said.
Even though the Altis is a single-incision sling, it has a "very secure" anchoring system, Dr. Van Drie said. The anchor not only goes into the obturator internus, but into the membrane and the obturator externus.
The study involved 113 women, with a mean age of 54 years. Their histories included stress incontinence with hypermobility (81%), without hypermobility (19%), mixed incontinence (37%), and overactive bladder (5%).
Most of the procedures were performed during an inpatient hospital stay (59%); however, 24% were performed at an ambulatory surgical center and 17%, in the physician’s office. General anesthesia was used in 52%, spinal in 3%, and local in 45%.
At 12 months, 90% of patients had at least a 50% reduction in pad weight, and 90% had a negative cough stress test result. The UDI decreased by a mean of 46 points, and the IIQ-7 score, by a mean of 47 points.
There were 11 device-related failures in eight patients. These included one each of urinary retention, urinary tract infection, decreased urine stream, dyspareunia, inflammation, worsening of overactive bladder, and voiding dysfunction. There were four mesh extrusions (3.5%), all less than 3 cm. Two patients with extrusion were smokers, and one was diabetic.
Serious adverse events occurred in three patients: One with a hematoma, one patient who needed transfer to the operating room because of anxiety during repair of a 2-mm mesh extrusion, and one patient whose adverse event was changed to a severe adverse event when she moved out of the study. There were no unanticipated device effects, Dr. Van Drie noted.
Dr. Van Drie is a consultant for Coloplast and has received research money and grants from the company.
NATIONAL HARBOR, MD. – After 12 months, an adjustable, single-incision sling significantly improved clinical and quality of life measures associated with stress urinary incontinence in women.
The Altis sling decreased the Urinary Distress Index (UDI) from a mean of 55 to a mean of 10, Dr. Douglas Van Drie said at a meeting sponsored by the AAGL. The Incontinence Impact Questionnaire (ILQ-7) showed similar improvements at the interim assessment of the device’s 2-year safety and efficacy study.
The study was sponsored by Coloplast, which makes the sling, with input and monitoring by the Food and Drug Administration. Altis was approved in November 2012 based on an investigational device exemption study, which included implant data. The FDA efficacy requirement was a 50% decrease in pad weight by 6 months.
According to the company website, "Altis is a unique, minimally invasive sling that combines integrated two-way tensioning with lightweight sling material to provide strength, security, and adjustability."
Physicians are divided on their thoughts about a single-incision sling, said Dr. Van Drie, a urogynecologist in group practice in Grand Rapids, Mich. "Those [physicians] who use them are advocates for their safety and simplicity, and the ability to insert them in the office. They have been adopted in different areas of the world as an option for doing simplified, less costly incontinence surgery. The argument against is questions about their staying power – will the effect hold up long term?" he said.
Even though the Altis is a single-incision sling, it has a "very secure" anchoring system, Dr. Van Drie said. The anchor not only goes into the obturator internus, but into the membrane and the obturator externus.
The study involved 113 women, with a mean age of 54 years. Their histories included stress incontinence with hypermobility (81%), without hypermobility (19%), mixed incontinence (37%), and overactive bladder (5%).
Most of the procedures were performed during an inpatient hospital stay (59%); however, 24% were performed at an ambulatory surgical center and 17%, in the physician’s office. General anesthesia was used in 52%, spinal in 3%, and local in 45%.
At 12 months, 90% of patients had at least a 50% reduction in pad weight, and 90% had a negative cough stress test result. The UDI decreased by a mean of 46 points, and the IIQ-7 score, by a mean of 47 points.
There were 11 device-related failures in eight patients. These included one each of urinary retention, urinary tract infection, decreased urine stream, dyspareunia, inflammation, worsening of overactive bladder, and voiding dysfunction. There were four mesh extrusions (3.5%), all less than 3 cm. Two patients with extrusion were smokers, and one was diabetic.
Serious adverse events occurred in three patients: One with a hematoma, one patient who needed transfer to the operating room because of anxiety during repair of a 2-mm mesh extrusion, and one patient whose adverse event was changed to a severe adverse event when she moved out of the study. There were no unanticipated device effects, Dr. Van Drie noted.
Dr. Van Drie is a consultant for Coloplast and has received research money and grants from the company.
AT THE AAGL GLOBAL CONGRESS
Major finding: After receiving a single-incision incontinence sling, 90% of women had significant improvements in clinical and quality of life measures related to stress urinary incontinence.
Data source: A prospective study of 113 women.
Disclosures: Coloplast sponsored the study. Dr. Van Drie is a consultant for Coloplast and has received research money and grants from the company.
Patients report long-lasting benefit of thermal balloon endometrial ablation
NATIONAL HARBOR, MD. – A thermal balloon endometrial ablation provided long-lasting benefit to women with dysmenorrhea, with 86% saying the benefits lasted for up to 11 years, and 82% reporting that they needed no further treatment for the condition.
"Patient satisfaction with [the device] is high," Vinod Kumar, Ph.D., said at a meeting sponsored by the AAGL. "It appears to be an effective option for women with symptoms of heavy menstrual bleeding and a robust alternative to hysterectomy."
Dr. Kumar of the University Hospitals Birmingham, England, presented follow-up data of up to 11 years on a cohort of 192 women who responded to a mailed survey after undergoing thermal balloon endometrial ablation with the Gynecare ThermaChoice III Uterine Balloon Therapy System.
In the cohort, 18 patients were 10-11 years postprocedure; 82 were 5-10 years postprocedure; and 92 were up to 5 years postprocedure.
They were a mean of 44 years when ablation was performed. Most (77%) had a normal uterus; 15% had an intramural fibroid; 2% a submucosal fibroid of less than 3 cm; and 3%, a polyp. The findings were undisclosed for the remainder. Uterine size was more than 10 cm in 74%.
With a mean follow-up of 30 months, 56% reported amenorrhea. Flow was lighter than before the procedure in 39% of the women, and unchanged or worse in 5%. Of the 44% who still had menstrual bleeding, length of bleeding was less than 3 days in 14%, 3-5 days in 14%, 6-7 days in 9%, and more than 8 days in 7%.
Most women (86%) reported less pain or no pain at follow-up. For 9%, there was no pain change; the rest reported a worsening of pain.
The majority of women (86%) reported that the clinical benefit had been maintained over time.
No further treatment was necessary in 82% of the group. Four women received a levonorgestrel intrauterine device. Nine were using medical therapy, including hormone therapy. One woman had a repeat endometrial ablation, and 20 had hysterectomy.
Most women (86%) reported that they were either satisfied or very satisfied with the procedure.
Dr. Kumar had no financial disclosures.
NATIONAL HARBOR, MD. – A thermal balloon endometrial ablation provided long-lasting benefit to women with dysmenorrhea, with 86% saying the benefits lasted for up to 11 years, and 82% reporting that they needed no further treatment for the condition.
"Patient satisfaction with [the device] is high," Vinod Kumar, Ph.D., said at a meeting sponsored by the AAGL. "It appears to be an effective option for women with symptoms of heavy menstrual bleeding and a robust alternative to hysterectomy."
Dr. Kumar of the University Hospitals Birmingham, England, presented follow-up data of up to 11 years on a cohort of 192 women who responded to a mailed survey after undergoing thermal balloon endometrial ablation with the Gynecare ThermaChoice III Uterine Balloon Therapy System.
In the cohort, 18 patients were 10-11 years postprocedure; 82 were 5-10 years postprocedure; and 92 were up to 5 years postprocedure.
They were a mean of 44 years when ablation was performed. Most (77%) had a normal uterus; 15% had an intramural fibroid; 2% a submucosal fibroid of less than 3 cm; and 3%, a polyp. The findings were undisclosed for the remainder. Uterine size was more than 10 cm in 74%.
With a mean follow-up of 30 months, 56% reported amenorrhea. Flow was lighter than before the procedure in 39% of the women, and unchanged or worse in 5%. Of the 44% who still had menstrual bleeding, length of bleeding was less than 3 days in 14%, 3-5 days in 14%, 6-7 days in 9%, and more than 8 days in 7%.
Most women (86%) reported less pain or no pain at follow-up. For 9%, there was no pain change; the rest reported a worsening of pain.
The majority of women (86%) reported that the clinical benefit had been maintained over time.
No further treatment was necessary in 82% of the group. Four women received a levonorgestrel intrauterine device. Nine were using medical therapy, including hormone therapy. One woman had a repeat endometrial ablation, and 20 had hysterectomy.
Most women (86%) reported that they were either satisfied or very satisfied with the procedure.
Dr. Kumar had no financial disclosures.
NATIONAL HARBOR, MD. – A thermal balloon endometrial ablation provided long-lasting benefit to women with dysmenorrhea, with 86% saying the benefits lasted for up to 11 years, and 82% reporting that they needed no further treatment for the condition.
"Patient satisfaction with [the device] is high," Vinod Kumar, Ph.D., said at a meeting sponsored by the AAGL. "It appears to be an effective option for women with symptoms of heavy menstrual bleeding and a robust alternative to hysterectomy."
Dr. Kumar of the University Hospitals Birmingham, England, presented follow-up data of up to 11 years on a cohort of 192 women who responded to a mailed survey after undergoing thermal balloon endometrial ablation with the Gynecare ThermaChoice III Uterine Balloon Therapy System.
In the cohort, 18 patients were 10-11 years postprocedure; 82 were 5-10 years postprocedure; and 92 were up to 5 years postprocedure.
They were a mean of 44 years when ablation was performed. Most (77%) had a normal uterus; 15% had an intramural fibroid; 2% a submucosal fibroid of less than 3 cm; and 3%, a polyp. The findings were undisclosed for the remainder. Uterine size was more than 10 cm in 74%.
With a mean follow-up of 30 months, 56% reported amenorrhea. Flow was lighter than before the procedure in 39% of the women, and unchanged or worse in 5%. Of the 44% who still had menstrual bleeding, length of bleeding was less than 3 days in 14%, 3-5 days in 14%, 6-7 days in 9%, and more than 8 days in 7%.
Most women (86%) reported less pain or no pain at follow-up. For 9%, there was no pain change; the rest reported a worsening of pain.
The majority of women (86%) reported that the clinical benefit had been maintained over time.
No further treatment was necessary in 82% of the group. Four women received a levonorgestrel intrauterine device. Nine were using medical therapy, including hormone therapy. One woman had a repeat endometrial ablation, and 20 had hysterectomy.
Most women (86%) reported that they were either satisfied or very satisfied with the procedure.
Dr. Kumar had no financial disclosures.
AT THE AAGL GLOBAL CONGRESS
Major finding: After up to 11 years of follow-up, 86% of women who had a thermal balloon endometrial ablation said they were satisfied with the procedure, and 82% had needed no further treatment.
Data source: The findings were from a follow-up study of 192 women.
Disclosures: Dr. Kumar had no financial disclosures.
Bowel resection for endometriosis restores fertility in many women
NATIONAL HARBOR, MD. – Pregnancy occurred in more than half of infertile women who had a segmental laparoscopic bowel resection for endometriosis.
Of 155 women, 96 (62%) conceived after the surgery. Most of these pregnancies (71 of 96; 74%) were spontaneous, Dr. Rosa Maria Neme reported at a meeting sponsored by AAGL.
The prospective study included women treated during 2009-2012 at the University of São Paulo, Brazil, said Dr. Neme, director of the university’s endometriosis center. The patients were a mean of 32 years old.
Most (92%) had bowel symptoms, including pain during defecation, diarrhea and constipation, abdominal bloating, and dyschezia. Endometriosis-related symptoms were present in all, and included dysmenorrhea, nonmenstrual pelvic pain and dyspareunia, tenesmus, cyclic rectal bleeding, lower back pain, and asthenia.
An associated infertility was present in 62% (155), with a mean duration of 18 months. Most patients (78%) had already undergone some form of fertility treatment before surgery and 69% had undergone a previous surgery for pelvic pain.
All of the patients had a torus bowel resection. A number of other procedures also were performed during the surgery, including extensive ureterolysis (80%), ovarian cystectomy (70%), partial vaginal resection (20%), and appendectomy (20%).
The mean operative time was 117 minutes. There were no conversions to open surgery, no transfusions, and no intra- or postoperative complications. The mean length of stay was 3 days. The pathology of all resected lesions was stromal and glandular endometriosis.
After the surgery, 96 pregnancies occurred among the subgroup with infertility (62%). The median time to conception was 8 months. The majority of pregnancies were spontaneous (71), with the rest achieved by in vitro fertilization. There were four miscarriages.
A quality of life survey was conducted at 6 months. By that time, dysmenorrhea, dyspareunia and pain on defecation, intestinal cramping, diarrhea and constipation had disappeared in all women.
Dr. Neme had no financial disclosures.
NATIONAL HARBOR, MD. – Pregnancy occurred in more than half of infertile women who had a segmental laparoscopic bowel resection for endometriosis.
Of 155 women, 96 (62%) conceived after the surgery. Most of these pregnancies (71 of 96; 74%) were spontaneous, Dr. Rosa Maria Neme reported at a meeting sponsored by AAGL.
The prospective study included women treated during 2009-2012 at the University of São Paulo, Brazil, said Dr. Neme, director of the university’s endometriosis center. The patients were a mean of 32 years old.
Most (92%) had bowel symptoms, including pain during defecation, diarrhea and constipation, abdominal bloating, and dyschezia. Endometriosis-related symptoms were present in all, and included dysmenorrhea, nonmenstrual pelvic pain and dyspareunia, tenesmus, cyclic rectal bleeding, lower back pain, and asthenia.
An associated infertility was present in 62% (155), with a mean duration of 18 months. Most patients (78%) had already undergone some form of fertility treatment before surgery and 69% had undergone a previous surgery for pelvic pain.
All of the patients had a torus bowel resection. A number of other procedures also were performed during the surgery, including extensive ureterolysis (80%), ovarian cystectomy (70%), partial vaginal resection (20%), and appendectomy (20%).
The mean operative time was 117 minutes. There were no conversions to open surgery, no transfusions, and no intra- or postoperative complications. The mean length of stay was 3 days. The pathology of all resected lesions was stromal and glandular endometriosis.
After the surgery, 96 pregnancies occurred among the subgroup with infertility (62%). The median time to conception was 8 months. The majority of pregnancies were spontaneous (71), with the rest achieved by in vitro fertilization. There were four miscarriages.
A quality of life survey was conducted at 6 months. By that time, dysmenorrhea, dyspareunia and pain on defecation, intestinal cramping, diarrhea and constipation had disappeared in all women.
Dr. Neme had no financial disclosures.
NATIONAL HARBOR, MD. – Pregnancy occurred in more than half of infertile women who had a segmental laparoscopic bowel resection for endometriosis.
Of 155 women, 96 (62%) conceived after the surgery. Most of these pregnancies (71 of 96; 74%) were spontaneous, Dr. Rosa Maria Neme reported at a meeting sponsored by AAGL.
The prospective study included women treated during 2009-2012 at the University of São Paulo, Brazil, said Dr. Neme, director of the university’s endometriosis center. The patients were a mean of 32 years old.
Most (92%) had bowel symptoms, including pain during defecation, diarrhea and constipation, abdominal bloating, and dyschezia. Endometriosis-related symptoms were present in all, and included dysmenorrhea, nonmenstrual pelvic pain and dyspareunia, tenesmus, cyclic rectal bleeding, lower back pain, and asthenia.
An associated infertility was present in 62% (155), with a mean duration of 18 months. Most patients (78%) had already undergone some form of fertility treatment before surgery and 69% had undergone a previous surgery for pelvic pain.
All of the patients had a torus bowel resection. A number of other procedures also were performed during the surgery, including extensive ureterolysis (80%), ovarian cystectomy (70%), partial vaginal resection (20%), and appendectomy (20%).
The mean operative time was 117 minutes. There were no conversions to open surgery, no transfusions, and no intra- or postoperative complications. The mean length of stay was 3 days. The pathology of all resected lesions was stromal and glandular endometriosis.
After the surgery, 96 pregnancies occurred among the subgroup with infertility (62%). The median time to conception was 8 months. The majority of pregnancies were spontaneous (71), with the rest achieved by in vitro fertilization. There were four miscarriages.
A quality of life survey was conducted at 6 months. By that time, dysmenorrhea, dyspareunia and pain on defecation, intestinal cramping, diarrhea and constipation had disappeared in all women.
Dr. Neme had no financial disclosures.
AT THE AAGL GLOBAL CONGRESS
Major finding: Pregnancy occurred in 62% of women with bowel endometriosis who underwent a bowel resection.
Data source: The prospective study involved 250 women, 155 of whom had infertility.
Disclosures: Dr. Neme had no financial disclosures.
Extraperitoneal lymphadenectomy nets bigger node harvest in endometrial cancer
NATIONAL HARBOR, MD. – Extraperitoneal para-aortic lymphadenectomy can result in more harvested nodes than standard laparoscopic or robot-assisted laparoscopic staging of patients with endometrial cancer, according to Dr. Janelle Pakish.
Harvesting the nodes does take longer this way – about 50 minutes longer than a standard laparoscopic staging and 40 minutes longer than a robotic-assisted one. But a retrospective study of 194 cases found that it secured twice as many para-aortic nodes, with half the conversion rate to open surgery as with laparoscopic staging, Dr. Pakish said at a meeting sponsored by the AAGL.
"It is safe and feasible, and especially effective for obese patients," in whom the technique yielded the largest node retrieval, said Dr. Pakish, a gynecologic oncologist at Dartmouth-Hitchcock Medical Center, Lebanon, N.H.
The study included data on patients who underwent extra- or transperitoneal laparoscopic lymphadenectomy for endometrial cancer staging at M.D. Anderson Cancer Center in Houston, from 2007 to 2012. The cohort was divided into three groups: those who had extraperitoneal para-aortic sampling followed by robot-assisted total laparoscopic lymphadenectomy (extraperitoneal group, 34); those who had a transperitoneal para-aortic lymphadenectomy followed by robot-assisted total laparoscopic hysterectomy (transperitoneal robotic group, 52); and those who had transperitoneal laparoscopic lymphadenectomy followed by a total laparoscopic hysterectomy (transperitoneal laparoscopic group, 108).
Significantly more para-aortic nodes were harvested in the extraperitoneal group than in the transperitoneal laparoscopic or the transperitoneal robotic groups (median of 10 vs. 5 and 4.5, respectively). The number of pelvic nodes was not significantly different between the three groups (median of 14, 13, and 13.5, respectively).
The extraperitoneal method was particularly effective in patients with a body mass index of 35 kg/m2 or more, harvesting a median of four more nodes than transperitoneal laparoscopy and seven more than transperitoneal robot-assisted laparoscopy.
The method was more time consuming, however, Dr. Pakish said. The median operative time for extraperitoneal lymphadenectomy was 339 minutes, compared with 286 minutes for the transperitoneal laparoscopy group and 297 for the transperitoneal robotic group.
There were three conversions to transperitoneal laparoscopy and three to laparoscopy in the extraperitoneal group (8.8%). In the transperitoneal laparoscopy group, there were 17 conversions to laparotomy (15.7%). The transperitoneal robotic group had the lowest conversion rate (two patients, 3.8%).
Dr. Pakish said she had no relevant financial disclosures.
NATIONAL HARBOR, MD. – Extraperitoneal para-aortic lymphadenectomy can result in more harvested nodes than standard laparoscopic or robot-assisted laparoscopic staging of patients with endometrial cancer, according to Dr. Janelle Pakish.
Harvesting the nodes does take longer this way – about 50 minutes longer than a standard laparoscopic staging and 40 minutes longer than a robotic-assisted one. But a retrospective study of 194 cases found that it secured twice as many para-aortic nodes, with half the conversion rate to open surgery as with laparoscopic staging, Dr. Pakish said at a meeting sponsored by the AAGL.
"It is safe and feasible, and especially effective for obese patients," in whom the technique yielded the largest node retrieval, said Dr. Pakish, a gynecologic oncologist at Dartmouth-Hitchcock Medical Center, Lebanon, N.H.
The study included data on patients who underwent extra- or transperitoneal laparoscopic lymphadenectomy for endometrial cancer staging at M.D. Anderson Cancer Center in Houston, from 2007 to 2012. The cohort was divided into three groups: those who had extraperitoneal para-aortic sampling followed by robot-assisted total laparoscopic lymphadenectomy (extraperitoneal group, 34); those who had a transperitoneal para-aortic lymphadenectomy followed by robot-assisted total laparoscopic hysterectomy (transperitoneal robotic group, 52); and those who had transperitoneal laparoscopic lymphadenectomy followed by a total laparoscopic hysterectomy (transperitoneal laparoscopic group, 108).
Significantly more para-aortic nodes were harvested in the extraperitoneal group than in the transperitoneal laparoscopic or the transperitoneal robotic groups (median of 10 vs. 5 and 4.5, respectively). The number of pelvic nodes was not significantly different between the three groups (median of 14, 13, and 13.5, respectively).
The extraperitoneal method was particularly effective in patients with a body mass index of 35 kg/m2 or more, harvesting a median of four more nodes than transperitoneal laparoscopy and seven more than transperitoneal robot-assisted laparoscopy.
The method was more time consuming, however, Dr. Pakish said. The median operative time for extraperitoneal lymphadenectomy was 339 minutes, compared with 286 minutes for the transperitoneal laparoscopy group and 297 for the transperitoneal robotic group.
There were three conversions to transperitoneal laparoscopy and three to laparoscopy in the extraperitoneal group (8.8%). In the transperitoneal laparoscopy group, there were 17 conversions to laparotomy (15.7%). The transperitoneal robotic group had the lowest conversion rate (two patients, 3.8%).
Dr. Pakish said she had no relevant financial disclosures.
NATIONAL HARBOR, MD. – Extraperitoneal para-aortic lymphadenectomy can result in more harvested nodes than standard laparoscopic or robot-assisted laparoscopic staging of patients with endometrial cancer, according to Dr. Janelle Pakish.
Harvesting the nodes does take longer this way – about 50 minutes longer than a standard laparoscopic staging and 40 minutes longer than a robotic-assisted one. But a retrospective study of 194 cases found that it secured twice as many para-aortic nodes, with half the conversion rate to open surgery as with laparoscopic staging, Dr. Pakish said at a meeting sponsored by the AAGL.
"It is safe and feasible, and especially effective for obese patients," in whom the technique yielded the largest node retrieval, said Dr. Pakish, a gynecologic oncologist at Dartmouth-Hitchcock Medical Center, Lebanon, N.H.
The study included data on patients who underwent extra- or transperitoneal laparoscopic lymphadenectomy for endometrial cancer staging at M.D. Anderson Cancer Center in Houston, from 2007 to 2012. The cohort was divided into three groups: those who had extraperitoneal para-aortic sampling followed by robot-assisted total laparoscopic lymphadenectomy (extraperitoneal group, 34); those who had a transperitoneal para-aortic lymphadenectomy followed by robot-assisted total laparoscopic hysterectomy (transperitoneal robotic group, 52); and those who had transperitoneal laparoscopic lymphadenectomy followed by a total laparoscopic hysterectomy (transperitoneal laparoscopic group, 108).
Significantly more para-aortic nodes were harvested in the extraperitoneal group than in the transperitoneal laparoscopic or the transperitoneal robotic groups (median of 10 vs. 5 and 4.5, respectively). The number of pelvic nodes was not significantly different between the three groups (median of 14, 13, and 13.5, respectively).
The extraperitoneal method was particularly effective in patients with a body mass index of 35 kg/m2 or more, harvesting a median of four more nodes than transperitoneal laparoscopy and seven more than transperitoneal robot-assisted laparoscopy.
The method was more time consuming, however, Dr. Pakish said. The median operative time for extraperitoneal lymphadenectomy was 339 minutes, compared with 286 minutes for the transperitoneal laparoscopy group and 297 for the transperitoneal robotic group.
There were three conversions to transperitoneal laparoscopy and three to laparoscopy in the extraperitoneal group (8.8%). In the transperitoneal laparoscopy group, there were 17 conversions to laparotomy (15.7%). The transperitoneal robotic group had the lowest conversion rate (two patients, 3.8%).
Dr. Pakish said she had no relevant financial disclosures.
AT THE AAGL GLOBAL CONGRESS
Major finding: Extraperitoneal laparoscopic lymphadenectomy secured a median of 10 para-aortic lymph nodes during endometrial cancer staging – twice as many as did transperitoneal staging.
Data source: A retrospective study of 194 patients.
Disclosures: Dr. Pakish said she had no relevant financial disclosures.
Race, ethnicity influence chances of minimally invasive hysterectomy
NATIONAL HARBOR, MD. – Race and ethnicity still appear to play a role in determining which patients receive a minimally invasive hysterectomy, and which undergo a traditional abdominal procedure.
Black, Hispanic, and Asian women were up to 50% less likely to have either a laparoscopic or vaginal hysterectomy, compared with whites, Dr. Katharine Esselen reported at the AAGL global congress.
The findings remained statistically significant even after Dr. Esselen and her colleagues controlled for a variety of patient, financial, and hospital characteristics.
"Racial disparities exist in the mode of hysterectomy in endometrial and cervical cancer, and must be further investigated to better understand the contributing factors so that they may be eradicated," said Dr. Esselen, a clinical fellow in gynecologic oncology at Brigham and Women’s Hospital, Boston.
The researchers extracted their data from the 2009 National Inpatient Sample. It included 1,000 hospitals and more than 8 million patient stays – representing 20% of the discharges in the country for that year.
In 2009, there were 64,410 hysterectomies performed for gynecologic malignancy. More than half (54%) were for endometrial cancer, followed by cervical cancer (23%), and ovarian cancer (19%). Other cancers made up the remainder.
The majority of surgeries in all these categories were abdominal: 72% of the endometrial cases, 56% of the cervical cases, and 95% of the ovarian cases. Laparoscopic hysterectomy was the surgical mode in 26% of endometrial cases, 23% of cervical cases, and 4% of ovarian cases. Vaginal hysterectomies were performed for 2% of endometrial cases, 21% of vaginal cases, and just 1% of ovarian cases.
A multivariate regression analysis controlled for demographic factors (age, race/ethnicity, insurance); patient factors (cancer diagnosis, fibroids, endometriosis, prolapse, menstrual disorders, age, severity of comorbidities, obesity); and hospital factors (urban/rural, teaching status, size, and region of country).
After adjustment for all of these factors, black women were 43% less likely to have a minimally invasive hysterectomy for endometrial cancer than were white women – a significant difference. Hispanic and Asian women were also significantly less likely to have minimally invasive surgery (MIS), with odds ratios of 0.61 and 0.63, respectively. Native American women, however, were more than five times as likely to have such a procedure compared with white women (OR, 5.26).
Insurance also played a role, Dr. Esselen said. Those with Medicaid were significantly less likely to have a minimally invasive procedure (OR, 0.64) than were those with private insurance.
The findings were similar for ovarian cancer. Black and Asian women were significantly less likely to have MIS than were whites (OR, 0.41 and 0.44, respectively). There was no significant difference seen for Hispanic women.
Medical comorbidities were significantly related to the chance of MIS as well. MIS was significantly less likely in women with moderate loss of function due to comorbid conditions (endometrial OR, 0.47; cervical OR, 0.62; ovarian OR, 0.38). Those with major to extreme loss of function had an even smaller chance (endometrial OR, 0.23; cervical OR, 0.21; ovarian OR, 0.10). The P values on these were all less than .0001.
Obesity only affected MIS odds in endometrial cancer, significantly increasing the chance of such a procedure (OR, 1.27).
Dr. Esselen said she had no financial disclosures.
NATIONAL HARBOR, MD. – Race and ethnicity still appear to play a role in determining which patients receive a minimally invasive hysterectomy, and which undergo a traditional abdominal procedure.
Black, Hispanic, and Asian women were up to 50% less likely to have either a laparoscopic or vaginal hysterectomy, compared with whites, Dr. Katharine Esselen reported at the AAGL global congress.
The findings remained statistically significant even after Dr. Esselen and her colleagues controlled for a variety of patient, financial, and hospital characteristics.
"Racial disparities exist in the mode of hysterectomy in endometrial and cervical cancer, and must be further investigated to better understand the contributing factors so that they may be eradicated," said Dr. Esselen, a clinical fellow in gynecologic oncology at Brigham and Women’s Hospital, Boston.
The researchers extracted their data from the 2009 National Inpatient Sample. It included 1,000 hospitals and more than 8 million patient stays – representing 20% of the discharges in the country for that year.
In 2009, there were 64,410 hysterectomies performed for gynecologic malignancy. More than half (54%) were for endometrial cancer, followed by cervical cancer (23%), and ovarian cancer (19%). Other cancers made up the remainder.
The majority of surgeries in all these categories were abdominal: 72% of the endometrial cases, 56% of the cervical cases, and 95% of the ovarian cases. Laparoscopic hysterectomy was the surgical mode in 26% of endometrial cases, 23% of cervical cases, and 4% of ovarian cases. Vaginal hysterectomies were performed for 2% of endometrial cases, 21% of vaginal cases, and just 1% of ovarian cases.
A multivariate regression analysis controlled for demographic factors (age, race/ethnicity, insurance); patient factors (cancer diagnosis, fibroids, endometriosis, prolapse, menstrual disorders, age, severity of comorbidities, obesity); and hospital factors (urban/rural, teaching status, size, and region of country).
After adjustment for all of these factors, black women were 43% less likely to have a minimally invasive hysterectomy for endometrial cancer than were white women – a significant difference. Hispanic and Asian women were also significantly less likely to have minimally invasive surgery (MIS), with odds ratios of 0.61 and 0.63, respectively. Native American women, however, were more than five times as likely to have such a procedure compared with white women (OR, 5.26).
Insurance also played a role, Dr. Esselen said. Those with Medicaid were significantly less likely to have a minimally invasive procedure (OR, 0.64) than were those with private insurance.
The findings were similar for ovarian cancer. Black and Asian women were significantly less likely to have MIS than were whites (OR, 0.41 and 0.44, respectively). There was no significant difference seen for Hispanic women.
Medical comorbidities were significantly related to the chance of MIS as well. MIS was significantly less likely in women with moderate loss of function due to comorbid conditions (endometrial OR, 0.47; cervical OR, 0.62; ovarian OR, 0.38). Those with major to extreme loss of function had an even smaller chance (endometrial OR, 0.23; cervical OR, 0.21; ovarian OR, 0.10). The P values on these were all less than .0001.
Obesity only affected MIS odds in endometrial cancer, significantly increasing the chance of such a procedure (OR, 1.27).
Dr. Esselen said she had no financial disclosures.
NATIONAL HARBOR, MD. – Race and ethnicity still appear to play a role in determining which patients receive a minimally invasive hysterectomy, and which undergo a traditional abdominal procedure.
Black, Hispanic, and Asian women were up to 50% less likely to have either a laparoscopic or vaginal hysterectomy, compared with whites, Dr. Katharine Esselen reported at the AAGL global congress.
The findings remained statistically significant even after Dr. Esselen and her colleagues controlled for a variety of patient, financial, and hospital characteristics.
"Racial disparities exist in the mode of hysterectomy in endometrial and cervical cancer, and must be further investigated to better understand the contributing factors so that they may be eradicated," said Dr. Esselen, a clinical fellow in gynecologic oncology at Brigham and Women’s Hospital, Boston.
The researchers extracted their data from the 2009 National Inpatient Sample. It included 1,000 hospitals and more than 8 million patient stays – representing 20% of the discharges in the country for that year.
In 2009, there were 64,410 hysterectomies performed for gynecologic malignancy. More than half (54%) were for endometrial cancer, followed by cervical cancer (23%), and ovarian cancer (19%). Other cancers made up the remainder.
The majority of surgeries in all these categories were abdominal: 72% of the endometrial cases, 56% of the cervical cases, and 95% of the ovarian cases. Laparoscopic hysterectomy was the surgical mode in 26% of endometrial cases, 23% of cervical cases, and 4% of ovarian cases. Vaginal hysterectomies were performed for 2% of endometrial cases, 21% of vaginal cases, and just 1% of ovarian cases.
A multivariate regression analysis controlled for demographic factors (age, race/ethnicity, insurance); patient factors (cancer diagnosis, fibroids, endometriosis, prolapse, menstrual disorders, age, severity of comorbidities, obesity); and hospital factors (urban/rural, teaching status, size, and region of country).
After adjustment for all of these factors, black women were 43% less likely to have a minimally invasive hysterectomy for endometrial cancer than were white women – a significant difference. Hispanic and Asian women were also significantly less likely to have minimally invasive surgery (MIS), with odds ratios of 0.61 and 0.63, respectively. Native American women, however, were more than five times as likely to have such a procedure compared with white women (OR, 5.26).
Insurance also played a role, Dr. Esselen said. Those with Medicaid were significantly less likely to have a minimally invasive procedure (OR, 0.64) than were those with private insurance.
The findings were similar for ovarian cancer. Black and Asian women were significantly less likely to have MIS than were whites (OR, 0.41 and 0.44, respectively). There was no significant difference seen for Hispanic women.
Medical comorbidities were significantly related to the chance of MIS as well. MIS was significantly less likely in women with moderate loss of function due to comorbid conditions (endometrial OR, 0.47; cervical OR, 0.62; ovarian OR, 0.38). Those with major to extreme loss of function had an even smaller chance (endometrial OR, 0.23; cervical OR, 0.21; ovarian OR, 0.10). The P values on these were all less than .0001.
Obesity only affected MIS odds in endometrial cancer, significantly increasing the chance of such a procedure (OR, 1.27).
Dr. Esselen said she had no financial disclosures.
AT THE AAGL GLOBAL CONGRESS
Major finding: Black women were 43% less likely to have a minimally invasive hysterectomy for endometrial cancer than were white women. Hispanic and Asian women were also significantly less likely to have minimally invasive surgery (MIS), with odds ratios of 0.61 and 0.63, respectively.
Data source: The 2009 National Inpatient Sample, with data on more than 64,000 hysterectomies for gynecologic cancers.
Disclosures: Dr. Esselen said she had no financial disclosures.
Studies question benefits of induced hypothermia after cardiac arrest
Two new studies may cast some doubt upon the now widely held belief that core cooling improves outcomes in unconscious cardiac arrest patients.
The studies, presented at the American Heart Association scientific sessions, found that neither cooling to hypothermic levels, compared with normothermic, nor prehospital, compared with in-hospital, significantly improved mortality or neurologic outcomes in more 2,000 patients. The papers were simultaneously published – one in the New England Journal of Medicine, and the other in JAMA.
Induced hypothermia is now standard of care for unconscious survivors of out-of-hospital cardiac arrests. But some animal models suggest that the earlier cooling begins, the better outcomes result. Dr. Francis Kim and his colleagues, however, found almost identical outcomes in a group of 1,359 patients, whatever the timing of hypothermia induction (JAMA 2013 Nov. 17 [doi:10.1001/jama.2013.282173]).
Dr. Kim of the University of Washington, Seattle, and his coauthors randomized patients to induction in the field and during transport, or upon hospital arrival. The intervention group received an infusion of up to 2L of ice-cold normal saline, 7-10 mg pancuronium, and 1-2 mg diazepam, with a target temperature goal of 34°C.
In the control group, hypothermia induction occurred in the hospital, according to each site’s protocol, with either surface or intravascular regimens.
Patients were divided into two groups: those with ventricular fibrillation and those without. Patients without VF were older (68 vs. 62 years). Other baseline characteristics – including time from the call to the return of spontaneous circulation, heart rate, and blood pressure – were similar.
The in-field intervention decreased mean core temperature by about 1.2°C in patients with VF and 1.3°C in those without VF. These patients achieved the target core temperature of 34°C more than 1 hour sooner than patients cooled in the hospital.
Survival to discharge was not significantly different between the intervention and control groups (63% vs. 64% of those with VF; 19% vs. 16% of those without VF).
Nor were there significant differences in the neurologic status of full recovery or mild impairment at discharge, the investigators said. Among those with VF, good neurologic status occurred in 57% of the intervention group and 62% of the control group. For those patients without VF, good neurologic outcomes occurred in 14% of the intervention group and 13% of the control group.
There were no significant differences in the proportion of patients who awakened from coma or died without awakening in any of the groups. The median length of stay was within the VF group (9 days for both treatment groups), and among those without VF (about 11 days for each treatment group).
The intervention carried some evidence of increased harm, the authors noted. Significantly more of the intervention patients rearrested during transport (26% vs. 21% of the controls). They also had significantly lower oxygenation, increased pulmonary edema on the first chest x-ray, and greater use of diuretics in the first 12 hours of hospitalization.
"Importantly, the method of pre-hospital hypothermia may have been associated with early harm that could have masked subsequent improvement," the investigators noted. "Rearrest possibly worsened brain ischemia that did not affect early mortality, but manifested as increased risk of death later during the hospitalization."
The second study released at the AHA meeting questioned whether induced hypothermia confers significant benefit over maintaining a near-normothermic temperature. Patients cooled to 33°C had no better outcomes than those whose core temperatures were held at 36°C, Dr. Niklas Nielsen and colleagues reported Nov. 18 (N. Engl. J. Med. 2013 Nov. 17 [doi:10.1056/NEJMoa1310519]).
"Our trial does not provide evidence that targeting a body temperature of 33° C confers any benefit for unconscious patients admitted to the hospital after out-of-hospital cardiac arrest, as compared with targeting a body temperature of 36°C," wrote Dr. Nielsen of Helsingborg Hospital, Sweden, and his colleagues.
The team analyzed outcomes for 939 patients who had been randomized to the two cooling regimens. The study’s primary outcome was all-cause mortality by 180 days after enrollment. The secondary outcomes included a composite of death or poor neurologic outcome at 180 days as evaluated with the Cerebral Performance Category (CPC) and modified Rankin scales.
The intervention consisted of 36 hours of cooling and gradual rewarming. Treatment teams were allowed to use ice packs, ice-cold fluid, and intravascular or surface temperature management. Gradual rewarming to 37°C commenced after 28 hours; this was discontinued or tapered at 36 hours. After this, patients in both groups were treated with fever-control measures to prevent a rebound temperature of above 37.5°C.
The patients were a mean of 64 years old; most of them (about 75%) had VF as the first shocked rhythm. Spontaneous circulation returned a median of 25 minutes after the arrest. All were unconscious when they arrived at the hospital. The mean follow-up was 256 days.
Most (76%) had temperature management with a surface cooling system; the rest were cooled by an intravascular catheter. Three patients in the 33°C group and four in the 36°C group didn’t get the assigned intervention; 16 in the 33°C group were rewarmed sooner than the protocol, at the discretion of their physician.
During the first week of hospitalization, 247 patients (132 in the 33°C group and 115 in the 36°C group) had life support withdrawn. Reasons for withdrawal included brain death, multiorgan failure, and ethical concerns.
At final follow-up, 50% of patients in the 33°C group and 48% in the 36°C group had died – a nonsignificant difference. There were no significant differences in the composite outcome of death or poor neurologic outcome whether measured by the CPC or the modified Rankin scales.
These results were similar in all adjusted analyses, in the intent-to-treat population, and in the per-protocol population.
Adverse events were common, occurring in 93% of the 33°C group and 90% of the 36°C group. The most common was hypokalemia, which was more significantly more common in the 33°C group (19% vs. 13%).
The study differed in one important way from others that have supported the use of induced hypothermia – and this might have had a key impact on the overall finding.
"We did not allow the natural trajectory of temperature evolution in either group," the authors said. "We actively controlled the temperature during the intervention period and aimed to prevent fever during the first 3 days after cardiac arrest."
It’s difficult to compare these results with those of the Hypothermia After Cardiac Arrest Study – the 2006 trial which first found in favor of the practice, the authors said.
Mortality in both groups of the current study was lower than that seen in 2006 – probably because of the recent advances in prehospital and in-hospital critical care management for cardiac arrest patients.
"We did not find any harm with a targeted temperature of 33°C as compared with 36°C," the investigators wrote. "However, it is worth recognizing that for all outcomes, none of the point estimates were in the direction of a benefit for the 33°C group. On the basis of these results, decisions about which temperature to target after out-of-hospital cardiac arrest require careful consideration."
Dr. Kim had no financial disclosures. Several of his coauthors disclosed financial relationships with medical device companies. Dr. Nielsen had nothing to disclose. Five of his coauthors disclosed financial relationships with outside entities; however, none appeared to be related to the study.
Dr. Niklas Nielsen and his colleagues found no real benefit of induced hypothermia over near-normothermic temperature maintenance in patients with out-of-hospital cardiac arrest, Dr. Jon C. Rittenberger and Dr. Clifton W. Callaway wrote in an editorial published along with the study (N. Engl. J. Med. 2013 Nov. 17 [doi:10.1056/NEJMe1312700]).
"This superbly executed study is more than twice the size of the original trials combined (which enrolled a total of 352 patients) and was conducted with meticulous attention to modern intensive care," the colleagues wrote, saying that there are multiple possible explanations for its finding.
The largest factor among these, they said, is probably the immense progress made in emergency care since the original hypothermia studies were published nearly a decade ago. "There has been evolution of intensive care over the past decade and improvements in patient care may have reduced the potential incremental benefits of a single intervention. In addition, illness severity varies greatly among patients with cardiac arrest, and there may be subgroups of patients who do benefit from induced hypothermia but who were not designated in advance. Particularly if the degree or duration of hypothermia must be adjusted to match the severity of brain injury, the benefits to a subgroup may be missed in a trial of one regimen of hypothermia for all comers."The most important message of this trial, they wrote, "is that modern, aggressive care that includes attention to temperature works, making survival more likely than death when a patient is hospitalized after CPR.
"In contrast to a decade ago, one-half instead of one-third of patients with return of spontaneous circulation after CPR can expect to survive hospitalization. Few medical situations have enjoyed such absolute improvement over the same time period."
Dr. Clifton W. Callaway is the Ronald D Stewart Endowed Chair of Emergency Medicine Research at the University of Pittsburgh. He disclosed receiving speakers fees from several cardiac health groups and holding two patents on devices related to cardiac resuscitation. Dr. Jon C. Rittenberger is director of residency research in the emergency medicine department of the University of Pittsburgh. He has received honoraria and lecture fees for speaking on emergency medicine.
Dr. Niklas Nielsen and his colleagues found no real benefit of induced hypothermia over near-normothermic temperature maintenance in patients with out-of-hospital cardiac arrest, Dr. Jon C. Rittenberger and Dr. Clifton W. Callaway wrote in an editorial published along with the study (N. Engl. J. Med. 2013 Nov. 17 [doi:10.1056/NEJMe1312700]).
"This superbly executed study is more than twice the size of the original trials combined (which enrolled a total of 352 patients) and was conducted with meticulous attention to modern intensive care," the colleagues wrote, saying that there are multiple possible explanations for its finding.
The largest factor among these, they said, is probably the immense progress made in emergency care since the original hypothermia studies were published nearly a decade ago. "There has been evolution of intensive care over the past decade and improvements in patient care may have reduced the potential incremental benefits of a single intervention. In addition, illness severity varies greatly among patients with cardiac arrest, and there may be subgroups of patients who do benefit from induced hypothermia but who were not designated in advance. Particularly if the degree or duration of hypothermia must be adjusted to match the severity of brain injury, the benefits to a subgroup may be missed in a trial of one regimen of hypothermia for all comers."The most important message of this trial, they wrote, "is that modern, aggressive care that includes attention to temperature works, making survival more likely than death when a patient is hospitalized after CPR.
"In contrast to a decade ago, one-half instead of one-third of patients with return of spontaneous circulation after CPR can expect to survive hospitalization. Few medical situations have enjoyed such absolute improvement over the same time period."
Dr. Clifton W. Callaway is the Ronald D Stewart Endowed Chair of Emergency Medicine Research at the University of Pittsburgh. He disclosed receiving speakers fees from several cardiac health groups and holding two patents on devices related to cardiac resuscitation. Dr. Jon C. Rittenberger is director of residency research in the emergency medicine department of the University of Pittsburgh. He has received honoraria and lecture fees for speaking on emergency medicine.
Dr. Niklas Nielsen and his colleagues found no real benefit of induced hypothermia over near-normothermic temperature maintenance in patients with out-of-hospital cardiac arrest, Dr. Jon C. Rittenberger and Dr. Clifton W. Callaway wrote in an editorial published along with the study (N. Engl. J. Med. 2013 Nov. 17 [doi:10.1056/NEJMe1312700]).
"This superbly executed study is more than twice the size of the original trials combined (which enrolled a total of 352 patients) and was conducted with meticulous attention to modern intensive care," the colleagues wrote, saying that there are multiple possible explanations for its finding.
The largest factor among these, they said, is probably the immense progress made in emergency care since the original hypothermia studies were published nearly a decade ago. "There has been evolution of intensive care over the past decade and improvements in patient care may have reduced the potential incremental benefits of a single intervention. In addition, illness severity varies greatly among patients with cardiac arrest, and there may be subgroups of patients who do benefit from induced hypothermia but who were not designated in advance. Particularly if the degree or duration of hypothermia must be adjusted to match the severity of brain injury, the benefits to a subgroup may be missed in a trial of one regimen of hypothermia for all comers."The most important message of this trial, they wrote, "is that modern, aggressive care that includes attention to temperature works, making survival more likely than death when a patient is hospitalized after CPR.
"In contrast to a decade ago, one-half instead of one-third of patients with return of spontaneous circulation after CPR can expect to survive hospitalization. Few medical situations have enjoyed such absolute improvement over the same time period."
Dr. Clifton W. Callaway is the Ronald D Stewart Endowed Chair of Emergency Medicine Research at the University of Pittsburgh. He disclosed receiving speakers fees from several cardiac health groups and holding two patents on devices related to cardiac resuscitation. Dr. Jon C. Rittenberger is director of residency research in the emergency medicine department of the University of Pittsburgh. He has received honoraria and lecture fees for speaking on emergency medicine.
Two new studies may cast some doubt upon the now widely held belief that core cooling improves outcomes in unconscious cardiac arrest patients.
The studies, presented at the American Heart Association scientific sessions, found that neither cooling to hypothermic levels, compared with normothermic, nor prehospital, compared with in-hospital, significantly improved mortality or neurologic outcomes in more 2,000 patients. The papers were simultaneously published – one in the New England Journal of Medicine, and the other in JAMA.
Induced hypothermia is now standard of care for unconscious survivors of out-of-hospital cardiac arrests. But some animal models suggest that the earlier cooling begins, the better outcomes result. Dr. Francis Kim and his colleagues, however, found almost identical outcomes in a group of 1,359 patients, whatever the timing of hypothermia induction (JAMA 2013 Nov. 17 [doi:10.1001/jama.2013.282173]).
Dr. Kim of the University of Washington, Seattle, and his coauthors randomized patients to induction in the field and during transport, or upon hospital arrival. The intervention group received an infusion of up to 2L of ice-cold normal saline, 7-10 mg pancuronium, and 1-2 mg diazepam, with a target temperature goal of 34°C.
In the control group, hypothermia induction occurred in the hospital, according to each site’s protocol, with either surface or intravascular regimens.
Patients were divided into two groups: those with ventricular fibrillation and those without. Patients without VF were older (68 vs. 62 years). Other baseline characteristics – including time from the call to the return of spontaneous circulation, heart rate, and blood pressure – were similar.
The in-field intervention decreased mean core temperature by about 1.2°C in patients with VF and 1.3°C in those without VF. These patients achieved the target core temperature of 34°C more than 1 hour sooner than patients cooled in the hospital.
Survival to discharge was not significantly different between the intervention and control groups (63% vs. 64% of those with VF; 19% vs. 16% of those without VF).
Nor were there significant differences in the neurologic status of full recovery or mild impairment at discharge, the investigators said. Among those with VF, good neurologic status occurred in 57% of the intervention group and 62% of the control group. For those patients without VF, good neurologic outcomes occurred in 14% of the intervention group and 13% of the control group.
There were no significant differences in the proportion of patients who awakened from coma or died without awakening in any of the groups. The median length of stay was within the VF group (9 days for both treatment groups), and among those without VF (about 11 days for each treatment group).
The intervention carried some evidence of increased harm, the authors noted. Significantly more of the intervention patients rearrested during transport (26% vs. 21% of the controls). They also had significantly lower oxygenation, increased pulmonary edema on the first chest x-ray, and greater use of diuretics in the first 12 hours of hospitalization.
"Importantly, the method of pre-hospital hypothermia may have been associated with early harm that could have masked subsequent improvement," the investigators noted. "Rearrest possibly worsened brain ischemia that did not affect early mortality, but manifested as increased risk of death later during the hospitalization."
The second study released at the AHA meeting questioned whether induced hypothermia confers significant benefit over maintaining a near-normothermic temperature. Patients cooled to 33°C had no better outcomes than those whose core temperatures were held at 36°C, Dr. Niklas Nielsen and colleagues reported Nov. 18 (N. Engl. J. Med. 2013 Nov. 17 [doi:10.1056/NEJMoa1310519]).
"Our trial does not provide evidence that targeting a body temperature of 33° C confers any benefit for unconscious patients admitted to the hospital after out-of-hospital cardiac arrest, as compared with targeting a body temperature of 36°C," wrote Dr. Nielsen of Helsingborg Hospital, Sweden, and his colleagues.
The team analyzed outcomes for 939 patients who had been randomized to the two cooling regimens. The study’s primary outcome was all-cause mortality by 180 days after enrollment. The secondary outcomes included a composite of death or poor neurologic outcome at 180 days as evaluated with the Cerebral Performance Category (CPC) and modified Rankin scales.
The intervention consisted of 36 hours of cooling and gradual rewarming. Treatment teams were allowed to use ice packs, ice-cold fluid, and intravascular or surface temperature management. Gradual rewarming to 37°C commenced after 28 hours; this was discontinued or tapered at 36 hours. After this, patients in both groups were treated with fever-control measures to prevent a rebound temperature of above 37.5°C.
The patients were a mean of 64 years old; most of them (about 75%) had VF as the first shocked rhythm. Spontaneous circulation returned a median of 25 minutes after the arrest. All were unconscious when they arrived at the hospital. The mean follow-up was 256 days.
Most (76%) had temperature management with a surface cooling system; the rest were cooled by an intravascular catheter. Three patients in the 33°C group and four in the 36°C group didn’t get the assigned intervention; 16 in the 33°C group were rewarmed sooner than the protocol, at the discretion of their physician.
During the first week of hospitalization, 247 patients (132 in the 33°C group and 115 in the 36°C group) had life support withdrawn. Reasons for withdrawal included brain death, multiorgan failure, and ethical concerns.
At final follow-up, 50% of patients in the 33°C group and 48% in the 36°C group had died – a nonsignificant difference. There were no significant differences in the composite outcome of death or poor neurologic outcome whether measured by the CPC or the modified Rankin scales.
These results were similar in all adjusted analyses, in the intent-to-treat population, and in the per-protocol population.
Adverse events were common, occurring in 93% of the 33°C group and 90% of the 36°C group. The most common was hypokalemia, which was more significantly more common in the 33°C group (19% vs. 13%).
The study differed in one important way from others that have supported the use of induced hypothermia – and this might have had a key impact on the overall finding.
"We did not allow the natural trajectory of temperature evolution in either group," the authors said. "We actively controlled the temperature during the intervention period and aimed to prevent fever during the first 3 days after cardiac arrest."
It’s difficult to compare these results with those of the Hypothermia After Cardiac Arrest Study – the 2006 trial which first found in favor of the practice, the authors said.
Mortality in both groups of the current study was lower than that seen in 2006 – probably because of the recent advances in prehospital and in-hospital critical care management for cardiac arrest patients.
"We did not find any harm with a targeted temperature of 33°C as compared with 36°C," the investigators wrote. "However, it is worth recognizing that for all outcomes, none of the point estimates were in the direction of a benefit for the 33°C group. On the basis of these results, decisions about which temperature to target after out-of-hospital cardiac arrest require careful consideration."
Dr. Kim had no financial disclosures. Several of his coauthors disclosed financial relationships with medical device companies. Dr. Nielsen had nothing to disclose. Five of his coauthors disclosed financial relationships with outside entities; however, none appeared to be related to the study.
Two new studies may cast some doubt upon the now widely held belief that core cooling improves outcomes in unconscious cardiac arrest patients.
The studies, presented at the American Heart Association scientific sessions, found that neither cooling to hypothermic levels, compared with normothermic, nor prehospital, compared with in-hospital, significantly improved mortality or neurologic outcomes in more 2,000 patients. The papers were simultaneously published – one in the New England Journal of Medicine, and the other in JAMA.
Induced hypothermia is now standard of care for unconscious survivors of out-of-hospital cardiac arrests. But some animal models suggest that the earlier cooling begins, the better outcomes result. Dr. Francis Kim and his colleagues, however, found almost identical outcomes in a group of 1,359 patients, whatever the timing of hypothermia induction (JAMA 2013 Nov. 17 [doi:10.1001/jama.2013.282173]).
Dr. Kim of the University of Washington, Seattle, and his coauthors randomized patients to induction in the field and during transport, or upon hospital arrival. The intervention group received an infusion of up to 2L of ice-cold normal saline, 7-10 mg pancuronium, and 1-2 mg diazepam, with a target temperature goal of 34°C.
In the control group, hypothermia induction occurred in the hospital, according to each site’s protocol, with either surface or intravascular regimens.
Patients were divided into two groups: those with ventricular fibrillation and those without. Patients without VF were older (68 vs. 62 years). Other baseline characteristics – including time from the call to the return of spontaneous circulation, heart rate, and blood pressure – were similar.
The in-field intervention decreased mean core temperature by about 1.2°C in patients with VF and 1.3°C in those without VF. These patients achieved the target core temperature of 34°C more than 1 hour sooner than patients cooled in the hospital.
Survival to discharge was not significantly different between the intervention and control groups (63% vs. 64% of those with VF; 19% vs. 16% of those without VF).
Nor were there significant differences in the neurologic status of full recovery or mild impairment at discharge, the investigators said. Among those with VF, good neurologic status occurred in 57% of the intervention group and 62% of the control group. For those patients without VF, good neurologic outcomes occurred in 14% of the intervention group and 13% of the control group.
There were no significant differences in the proportion of patients who awakened from coma or died without awakening in any of the groups. The median length of stay was within the VF group (9 days for both treatment groups), and among those without VF (about 11 days for each treatment group).
The intervention carried some evidence of increased harm, the authors noted. Significantly more of the intervention patients rearrested during transport (26% vs. 21% of the controls). They also had significantly lower oxygenation, increased pulmonary edema on the first chest x-ray, and greater use of diuretics in the first 12 hours of hospitalization.
"Importantly, the method of pre-hospital hypothermia may have been associated with early harm that could have masked subsequent improvement," the investigators noted. "Rearrest possibly worsened brain ischemia that did not affect early mortality, but manifested as increased risk of death later during the hospitalization."
The second study released at the AHA meeting questioned whether induced hypothermia confers significant benefit over maintaining a near-normothermic temperature. Patients cooled to 33°C had no better outcomes than those whose core temperatures were held at 36°C, Dr. Niklas Nielsen and colleagues reported Nov. 18 (N. Engl. J. Med. 2013 Nov. 17 [doi:10.1056/NEJMoa1310519]).
"Our trial does not provide evidence that targeting a body temperature of 33° C confers any benefit for unconscious patients admitted to the hospital after out-of-hospital cardiac arrest, as compared with targeting a body temperature of 36°C," wrote Dr. Nielsen of Helsingborg Hospital, Sweden, and his colleagues.
The team analyzed outcomes for 939 patients who had been randomized to the two cooling regimens. The study’s primary outcome was all-cause mortality by 180 days after enrollment. The secondary outcomes included a composite of death or poor neurologic outcome at 180 days as evaluated with the Cerebral Performance Category (CPC) and modified Rankin scales.
The intervention consisted of 36 hours of cooling and gradual rewarming. Treatment teams were allowed to use ice packs, ice-cold fluid, and intravascular or surface temperature management. Gradual rewarming to 37°C commenced after 28 hours; this was discontinued or tapered at 36 hours. After this, patients in both groups were treated with fever-control measures to prevent a rebound temperature of above 37.5°C.
The patients were a mean of 64 years old; most of them (about 75%) had VF as the first shocked rhythm. Spontaneous circulation returned a median of 25 minutes after the arrest. All were unconscious when they arrived at the hospital. The mean follow-up was 256 days.
Most (76%) had temperature management with a surface cooling system; the rest were cooled by an intravascular catheter. Three patients in the 33°C group and four in the 36°C group didn’t get the assigned intervention; 16 in the 33°C group were rewarmed sooner than the protocol, at the discretion of their physician.
During the first week of hospitalization, 247 patients (132 in the 33°C group and 115 in the 36°C group) had life support withdrawn. Reasons for withdrawal included brain death, multiorgan failure, and ethical concerns.
At final follow-up, 50% of patients in the 33°C group and 48% in the 36°C group had died – a nonsignificant difference. There were no significant differences in the composite outcome of death or poor neurologic outcome whether measured by the CPC or the modified Rankin scales.
These results were similar in all adjusted analyses, in the intent-to-treat population, and in the per-protocol population.
Adverse events were common, occurring in 93% of the 33°C group and 90% of the 36°C group. The most common was hypokalemia, which was more significantly more common in the 33°C group (19% vs. 13%).
The study differed in one important way from others that have supported the use of induced hypothermia – and this might have had a key impact on the overall finding.
"We did not allow the natural trajectory of temperature evolution in either group," the authors said. "We actively controlled the temperature during the intervention period and aimed to prevent fever during the first 3 days after cardiac arrest."
It’s difficult to compare these results with those of the Hypothermia After Cardiac Arrest Study – the 2006 trial which first found in favor of the practice, the authors said.
Mortality in both groups of the current study was lower than that seen in 2006 – probably because of the recent advances in prehospital and in-hospital critical care management for cardiac arrest patients.
"We did not find any harm with a targeted temperature of 33°C as compared with 36°C," the investigators wrote. "However, it is worth recognizing that for all outcomes, none of the point estimates were in the direction of a benefit for the 33°C group. On the basis of these results, decisions about which temperature to target after out-of-hospital cardiac arrest require careful consideration."
Dr. Kim had no financial disclosures. Several of his coauthors disclosed financial relationships with medical device companies. Dr. Nielsen had nothing to disclose. Five of his coauthors disclosed financial relationships with outside entities; however, none appeared to be related to the study.
FROM THE AHA SCIENTIFIC SESSIONS
Major finding: Induced hypothermia after out-of-hospital cardiac arrest didn’t improve patient outcomes when compared with near-normal temperatures, or whether it was delivered before or during hospitalization.
Data source: One study randomized 939 patients to either early or in-hospital cooling; the other randomized 1,359 to a target temperature of 33° C or 36° C.
Disclosures: Dr. Kim had no financial disclosures. Several of his coauthors disclosed financial relationships with medical device companies. Dr. Nielsen had nothing to disclose. Five of his coauthors disclosed financial relationships with outside entities; however, none appeared to be related to the study.
Shingles vaccine most effective and cost effective when given to patients in their 60s
Because efficacy of the shingles vaccine appears to wane over time, it should not routinely be given to people in their 50s, according to information presented at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
"At this time, there is insufficient evidence supporting long-term protection of the vaccine, so if people younger than 60 [years] are vaccinated, they might not be protected when chance of disease is highest," said Dr. Jeff Duchin, chair of the ACIP’s herpes zoster working group. "We will continue to monitor data as they becomes available, but at this point we are reaffirming that vaccination be done in those age 60 [years] and older."
The committee made its recommendation after reviewing newly released data from Merck’s Long-Term Persistence Study for shingles. It found that, 10 years after vaccination, the overall incidence rate of shingles had increased from about 4/1,000 person-years to 11/1,000 person-years. The observed incidence rate of shingles in historical controls is about 13 cases/1,000 person-years.
Dr. Janie Parrino, director of vaccine clinical research at Merck Research Laboratories, presented results of the company’s latest study of the vaccine’s extended efficacy. The shingles Long-Term Persistence Study is an 8-year follow-up of the Shingles Prevention Study, in which subjects were randomized to Merck’s Zostavax vaccine or placebo. It showed that the vaccine significantly reduced the incidence of herpes zoster and postherpetic neuralgia (PHN) as well as the shingles burden of illness, in adults older than 60 years.
A short-term follow-up study appeared last year; it added 4 more years of follow-up data on more than 14,000 of the original cohort.
It found a gradual decrease in vaccine efficacy by 6-7 years after vaccination. "Analysis of vaccine efficacy in each year after vaccination for all outcomes showed a decrease after year 1, with a further decline thereafter," the investigators said. "Vaccine efficacy was statistically significant for the incidence of herpes zoster and herpes zoster burden of illness through year 5 after vaccination, but vaccine efficacy is uncertain beyond that point."
The long-term study presented at the Atlanta meeting involved up to 12 years of data on almost 7,000 people in the original cohort. It demonstrated a continuous time-bound waning of efficacy.
In addition to the increasing annual incidence of shingles, the study found an increasing incidence of PHN, from less than 1/1,000 person-years during the year of vaccination to about 1/1,000 person years by year 10. However, the incidence among placebo subjects and historical controls was more than 2/1,000 person years by that time. The shingles burden of illness (a measure of acute pain) also increased as time passed, Dr. Parrino noted.
Taking into account both clinical efficacy and cost, Ismael Ortega-Sanchez, Ph.D., a health economist with the CDC, concluded that the shingles vaccine is most effective when given to people in their 60s and 70s.
Vaccinating from age 50-59 years would prevent about 20,000 shingles cases annually. But vaccinating in the 60s would prevent 26,000 and, in the 70s, 21,000. The trend was similar for cases of PHN: prevention of 1,000 cases for vaccinating in the 50s, 4,000 for vaccinating in the 60s, and 8,000 for vaccinating in the 70s.
Other outcomes – emergency visits, ambulatory visits, hospitalizations, length of hospital stay, and prescriptions – also increased significantly over the 3 decades.
Savings tracked clinical outcomes in a value assessment model of 6 million people: 1 million vaccinated and unvaccinated in each of the three age cohorts.
Compared with not vaccinating, vaccinating those in their 50s would save $16.8 million annually. Vaccinating during the 60s would save about $24.3 million, and during the 70s, $31.9 million.
And, because younger people are likely to have more systemic reactions, they cost more to vaccinate. A model that accounted for the cost of the vaccine, administration, and treating local and systemic reactions, put the total price tag at $193 million for those in their 50s, and $190 million for each of the two older groups.
Finally, the number needed to treat to prevent one case of shingles, PHN, or non-PHN complication case was higher in those in their 50s than in those in their 60s or 70s (one shingles case – 51, 38, and 47 respectively; one PHN case – 988, 247, and 124).
"This isn’t intended to tell people not to get the vaccine early, but to make them aware of the risks and benefits, given that the greatest risk of the disease is later in life," Dr. Duchin said. Since there are no data on booster shingles vaccines, he said "we only have one shot at this and we need to focus our efforts on those at the greatest risk."
Because efficacy of the shingles vaccine appears to wane over time, it should not routinely be given to people in their 50s, according to information presented at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
"At this time, there is insufficient evidence supporting long-term protection of the vaccine, so if people younger than 60 [years] are vaccinated, they might not be protected when chance of disease is highest," said Dr. Jeff Duchin, chair of the ACIP’s herpes zoster working group. "We will continue to monitor data as they becomes available, but at this point we are reaffirming that vaccination be done in those age 60 [years] and older."
The committee made its recommendation after reviewing newly released data from Merck’s Long-Term Persistence Study for shingles. It found that, 10 years after vaccination, the overall incidence rate of shingles had increased from about 4/1,000 person-years to 11/1,000 person-years. The observed incidence rate of shingles in historical controls is about 13 cases/1,000 person-years.
Dr. Janie Parrino, director of vaccine clinical research at Merck Research Laboratories, presented results of the company’s latest study of the vaccine’s extended efficacy. The shingles Long-Term Persistence Study is an 8-year follow-up of the Shingles Prevention Study, in which subjects were randomized to Merck’s Zostavax vaccine or placebo. It showed that the vaccine significantly reduced the incidence of herpes zoster and postherpetic neuralgia (PHN) as well as the shingles burden of illness, in adults older than 60 years.
A short-term follow-up study appeared last year; it added 4 more years of follow-up data on more than 14,000 of the original cohort.
It found a gradual decrease in vaccine efficacy by 6-7 years after vaccination. "Analysis of vaccine efficacy in each year after vaccination for all outcomes showed a decrease after year 1, with a further decline thereafter," the investigators said. "Vaccine efficacy was statistically significant for the incidence of herpes zoster and herpes zoster burden of illness through year 5 after vaccination, but vaccine efficacy is uncertain beyond that point."
The long-term study presented at the Atlanta meeting involved up to 12 years of data on almost 7,000 people in the original cohort. It demonstrated a continuous time-bound waning of efficacy.
In addition to the increasing annual incidence of shingles, the study found an increasing incidence of PHN, from less than 1/1,000 person-years during the year of vaccination to about 1/1,000 person years by year 10. However, the incidence among placebo subjects and historical controls was more than 2/1,000 person years by that time. The shingles burden of illness (a measure of acute pain) also increased as time passed, Dr. Parrino noted.
Taking into account both clinical efficacy and cost, Ismael Ortega-Sanchez, Ph.D., a health economist with the CDC, concluded that the shingles vaccine is most effective when given to people in their 60s and 70s.
Vaccinating from age 50-59 years would prevent about 20,000 shingles cases annually. But vaccinating in the 60s would prevent 26,000 and, in the 70s, 21,000. The trend was similar for cases of PHN: prevention of 1,000 cases for vaccinating in the 50s, 4,000 for vaccinating in the 60s, and 8,000 for vaccinating in the 70s.
Other outcomes – emergency visits, ambulatory visits, hospitalizations, length of hospital stay, and prescriptions – also increased significantly over the 3 decades.
Savings tracked clinical outcomes in a value assessment model of 6 million people: 1 million vaccinated and unvaccinated in each of the three age cohorts.
Compared with not vaccinating, vaccinating those in their 50s would save $16.8 million annually. Vaccinating during the 60s would save about $24.3 million, and during the 70s, $31.9 million.
And, because younger people are likely to have more systemic reactions, they cost more to vaccinate. A model that accounted for the cost of the vaccine, administration, and treating local and systemic reactions, put the total price tag at $193 million for those in their 50s, and $190 million for each of the two older groups.
Finally, the number needed to treat to prevent one case of shingles, PHN, or non-PHN complication case was higher in those in their 50s than in those in their 60s or 70s (one shingles case – 51, 38, and 47 respectively; one PHN case – 988, 247, and 124).
"This isn’t intended to tell people not to get the vaccine early, but to make them aware of the risks and benefits, given that the greatest risk of the disease is later in life," Dr. Duchin said. Since there are no data on booster shingles vaccines, he said "we only have one shot at this and we need to focus our efforts on those at the greatest risk."
Because efficacy of the shingles vaccine appears to wane over time, it should not routinely be given to people in their 50s, according to information presented at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
"At this time, there is insufficient evidence supporting long-term protection of the vaccine, so if people younger than 60 [years] are vaccinated, they might not be protected when chance of disease is highest," said Dr. Jeff Duchin, chair of the ACIP’s herpes zoster working group. "We will continue to monitor data as they becomes available, but at this point we are reaffirming that vaccination be done in those age 60 [years] and older."
The committee made its recommendation after reviewing newly released data from Merck’s Long-Term Persistence Study for shingles. It found that, 10 years after vaccination, the overall incidence rate of shingles had increased from about 4/1,000 person-years to 11/1,000 person-years. The observed incidence rate of shingles in historical controls is about 13 cases/1,000 person-years.
Dr. Janie Parrino, director of vaccine clinical research at Merck Research Laboratories, presented results of the company’s latest study of the vaccine’s extended efficacy. The shingles Long-Term Persistence Study is an 8-year follow-up of the Shingles Prevention Study, in which subjects were randomized to Merck’s Zostavax vaccine or placebo. It showed that the vaccine significantly reduced the incidence of herpes zoster and postherpetic neuralgia (PHN) as well as the shingles burden of illness, in adults older than 60 years.
A short-term follow-up study appeared last year; it added 4 more years of follow-up data on more than 14,000 of the original cohort.
It found a gradual decrease in vaccine efficacy by 6-7 years after vaccination. "Analysis of vaccine efficacy in each year after vaccination for all outcomes showed a decrease after year 1, with a further decline thereafter," the investigators said. "Vaccine efficacy was statistically significant for the incidence of herpes zoster and herpes zoster burden of illness through year 5 after vaccination, but vaccine efficacy is uncertain beyond that point."
The long-term study presented at the Atlanta meeting involved up to 12 years of data on almost 7,000 people in the original cohort. It demonstrated a continuous time-bound waning of efficacy.
In addition to the increasing annual incidence of shingles, the study found an increasing incidence of PHN, from less than 1/1,000 person-years during the year of vaccination to about 1/1,000 person years by year 10. However, the incidence among placebo subjects and historical controls was more than 2/1,000 person years by that time. The shingles burden of illness (a measure of acute pain) also increased as time passed, Dr. Parrino noted.
Taking into account both clinical efficacy and cost, Ismael Ortega-Sanchez, Ph.D., a health economist with the CDC, concluded that the shingles vaccine is most effective when given to people in their 60s and 70s.
Vaccinating from age 50-59 years would prevent about 20,000 shingles cases annually. But vaccinating in the 60s would prevent 26,000 and, in the 70s, 21,000. The trend was similar for cases of PHN: prevention of 1,000 cases for vaccinating in the 50s, 4,000 for vaccinating in the 60s, and 8,000 for vaccinating in the 70s.
Other outcomes – emergency visits, ambulatory visits, hospitalizations, length of hospital stay, and prescriptions – also increased significantly over the 3 decades.
Savings tracked clinical outcomes in a value assessment model of 6 million people: 1 million vaccinated and unvaccinated in each of the three age cohorts.
Compared with not vaccinating, vaccinating those in their 50s would save $16.8 million annually. Vaccinating during the 60s would save about $24.3 million, and during the 70s, $31.9 million.
And, because younger people are likely to have more systemic reactions, they cost more to vaccinate. A model that accounted for the cost of the vaccine, administration, and treating local and systemic reactions, put the total price tag at $193 million for those in their 50s, and $190 million for each of the two older groups.
Finally, the number needed to treat to prevent one case of shingles, PHN, or non-PHN complication case was higher in those in their 50s than in those in their 60s or 70s (one shingles case – 51, 38, and 47 respectively; one PHN case – 988, 247, and 124).
"This isn’t intended to tell people not to get the vaccine early, but to make them aware of the risks and benefits, given that the greatest risk of the disease is later in life," Dr. Duchin said. Since there are no data on booster shingles vaccines, he said "we only have one shot at this and we need to focus our efforts on those at the greatest risk."
FROM ACIP
Small study: TBI leads to rapid deposition of brain amyloid
Brain imaging of patients with traumatic brain injury in conjunction with analyses of postmortem tissue appears to confirm that beta amyloid plaques form in some areas of the brain shortly after injury, according to findings published Nov. 11 in JAMA Neurology.
The finding suggests that PET with Pittsburgh compound B (PiB), which binds to amyloid beta plaques, could be useful in the post-TBI patient workup, reported Young T. Hong, Ph.D., of the Wolfson Brain Imaging Centre at the University of Cambridge, England, and colleagues.
"The use of [PET PiB] for amyloid imaging following TBI provides us with the potential for understanding the pathophysiology of TBI, for characterizing the mechanistic drivers of disease progression or suboptimal recovery in the subacute phase of TBI, for identifying patients at high risk of accelerated [Alzheimer’s disease], and for evaluating the potential of antiamyloid therapies," the investigators wrote (JAMA Neurol. 2013 Nov. 11 [doi:10.1001/jamaneurol.2013.4847]).
PET with PiB in 15 patients with a traumatic brain injury that occurred 1-361 days before showed more amyloid binding than did the scans of 11 healthy controls. Amyloid tended to appear rapidly – within 48 hours of injury in some patients. It was located with significantly greater concentration in the cortical gray matter and striatum of TBI patients, compared with controls, but not in the thalamus or white matter. However, most patients showed very little accumulation around the injury site or in places with high vasogenic edema. The medial temporal and hippocampal regions were likewise spared, the investigators noted.
They saw similar results in comparisons of postmortem tissue samples from 16 patients who had died 3 hours to 56 days after a TBI and 7 who had died of other, nonneurologic causes that were analyzed via autoradiography with tritium-labeled PiB and immunocytochemistry for amyloid beta. TBI patients had more frequent and denser amyloid in the neocortical gray matter than did controls. There was no cerebellar cortical amyloid binding among TBI patients except for a 61-year old woman who died 12 hours after a brain injury and had amyloid accumulation in the meningeal vessels.
In the imaging portion of the study, TBI patients and healthy controls had median ages of 33 years and 35 years, respectively. The postmortem analyses involved TBI patients and controls with median ages of 46 years and 61 years, respectively. Patients were matched for age within the cohorts.
Of the 15 TBI patients who underwent PET with PiB, 8 survived with good recovery, 2 with moderate disability, and 3 with severe disability; the other 2 remained in a vegetative state.
The amyloid biding in the TBI patients is reminiscent of the early amyloid deposition seen in some patients who have presenilin-1 gene mutations, which cause early-onset Alzheimer’s disease. The areas of PiB binding in the TBI patients "would therefore be concordant with currently proposed mechanism of overproduction leading to amyloid deposition in TBI," the authors noted.
The study’s biggest weaknesses are its small sample size and lack of serial imaging, the latter perhaps most important because the small proportion of late studies in the 15-patient cohort "limits inferences regarding the temporal pattern of [PiB] binding in vivo and requires confirmation in a larger cohort," they wrote.
The National Institute for Health Research Cambridge Biomedical Research Centre funded the study. Two authors – Dr. William E. Klunk and Chester A. Mathis, Ph.D. – are coinventors of PiB and have a financial interest in the licensing agreement for PiB between GE Healthcare and the University of Pittsburgh.
On Twitter @Alz_Gal
Brain imaging of patients with traumatic brain injury in conjunction with analyses of postmortem tissue appears to confirm that beta amyloid plaques form in some areas of the brain shortly after injury, according to findings published Nov. 11 in JAMA Neurology.
The finding suggests that PET with Pittsburgh compound B (PiB), which binds to amyloid beta plaques, could be useful in the post-TBI patient workup, reported Young T. Hong, Ph.D., of the Wolfson Brain Imaging Centre at the University of Cambridge, England, and colleagues.
"The use of [PET PiB] for amyloid imaging following TBI provides us with the potential for understanding the pathophysiology of TBI, for characterizing the mechanistic drivers of disease progression or suboptimal recovery in the subacute phase of TBI, for identifying patients at high risk of accelerated [Alzheimer’s disease], and for evaluating the potential of antiamyloid therapies," the investigators wrote (JAMA Neurol. 2013 Nov. 11 [doi:10.1001/jamaneurol.2013.4847]).
PET with PiB in 15 patients with a traumatic brain injury that occurred 1-361 days before showed more amyloid binding than did the scans of 11 healthy controls. Amyloid tended to appear rapidly – within 48 hours of injury in some patients. It was located with significantly greater concentration in the cortical gray matter and striatum of TBI patients, compared with controls, but not in the thalamus or white matter. However, most patients showed very little accumulation around the injury site or in places with high vasogenic edema. The medial temporal and hippocampal regions were likewise spared, the investigators noted.
They saw similar results in comparisons of postmortem tissue samples from 16 patients who had died 3 hours to 56 days after a TBI and 7 who had died of other, nonneurologic causes that were analyzed via autoradiography with tritium-labeled PiB and immunocytochemistry for amyloid beta. TBI patients had more frequent and denser amyloid in the neocortical gray matter than did controls. There was no cerebellar cortical amyloid binding among TBI patients except for a 61-year old woman who died 12 hours after a brain injury and had amyloid accumulation in the meningeal vessels.
In the imaging portion of the study, TBI patients and healthy controls had median ages of 33 years and 35 years, respectively. The postmortem analyses involved TBI patients and controls with median ages of 46 years and 61 years, respectively. Patients were matched for age within the cohorts.
Of the 15 TBI patients who underwent PET with PiB, 8 survived with good recovery, 2 with moderate disability, and 3 with severe disability; the other 2 remained in a vegetative state.
The amyloid biding in the TBI patients is reminiscent of the early amyloid deposition seen in some patients who have presenilin-1 gene mutations, which cause early-onset Alzheimer’s disease. The areas of PiB binding in the TBI patients "would therefore be concordant with currently proposed mechanism of overproduction leading to amyloid deposition in TBI," the authors noted.
The study’s biggest weaknesses are its small sample size and lack of serial imaging, the latter perhaps most important because the small proportion of late studies in the 15-patient cohort "limits inferences regarding the temporal pattern of [PiB] binding in vivo and requires confirmation in a larger cohort," they wrote.
The National Institute for Health Research Cambridge Biomedical Research Centre funded the study. Two authors – Dr. William E. Klunk and Chester A. Mathis, Ph.D. – are coinventors of PiB and have a financial interest in the licensing agreement for PiB between GE Healthcare and the University of Pittsburgh.
On Twitter @Alz_Gal
Brain imaging of patients with traumatic brain injury in conjunction with analyses of postmortem tissue appears to confirm that beta amyloid plaques form in some areas of the brain shortly after injury, according to findings published Nov. 11 in JAMA Neurology.
The finding suggests that PET with Pittsburgh compound B (PiB), which binds to amyloid beta plaques, could be useful in the post-TBI patient workup, reported Young T. Hong, Ph.D., of the Wolfson Brain Imaging Centre at the University of Cambridge, England, and colleagues.
"The use of [PET PiB] for amyloid imaging following TBI provides us with the potential for understanding the pathophysiology of TBI, for characterizing the mechanistic drivers of disease progression or suboptimal recovery in the subacute phase of TBI, for identifying patients at high risk of accelerated [Alzheimer’s disease], and for evaluating the potential of antiamyloid therapies," the investigators wrote (JAMA Neurol. 2013 Nov. 11 [doi:10.1001/jamaneurol.2013.4847]).
PET with PiB in 15 patients with a traumatic brain injury that occurred 1-361 days before showed more amyloid binding than did the scans of 11 healthy controls. Amyloid tended to appear rapidly – within 48 hours of injury in some patients. It was located with significantly greater concentration in the cortical gray matter and striatum of TBI patients, compared with controls, but not in the thalamus or white matter. However, most patients showed very little accumulation around the injury site or in places with high vasogenic edema. The medial temporal and hippocampal regions were likewise spared, the investigators noted.
They saw similar results in comparisons of postmortem tissue samples from 16 patients who had died 3 hours to 56 days after a TBI and 7 who had died of other, nonneurologic causes that were analyzed via autoradiography with tritium-labeled PiB and immunocytochemistry for amyloid beta. TBI patients had more frequent and denser amyloid in the neocortical gray matter than did controls. There was no cerebellar cortical amyloid binding among TBI patients except for a 61-year old woman who died 12 hours after a brain injury and had amyloid accumulation in the meningeal vessels.
In the imaging portion of the study, TBI patients and healthy controls had median ages of 33 years and 35 years, respectively. The postmortem analyses involved TBI patients and controls with median ages of 46 years and 61 years, respectively. Patients were matched for age within the cohorts.
Of the 15 TBI patients who underwent PET with PiB, 8 survived with good recovery, 2 with moderate disability, and 3 with severe disability; the other 2 remained in a vegetative state.
The amyloid biding in the TBI patients is reminiscent of the early amyloid deposition seen in some patients who have presenilin-1 gene mutations, which cause early-onset Alzheimer’s disease. The areas of PiB binding in the TBI patients "would therefore be concordant with currently proposed mechanism of overproduction leading to amyloid deposition in TBI," the authors noted.
The study’s biggest weaknesses are its small sample size and lack of serial imaging, the latter perhaps most important because the small proportion of late studies in the 15-patient cohort "limits inferences regarding the temporal pattern of [PiB] binding in vivo and requires confirmation in a larger cohort," they wrote.
The National Institute for Health Research Cambridge Biomedical Research Centre funded the study. Two authors – Dr. William E. Klunk and Chester A. Mathis, Ph.D. – are coinventors of PiB and have a financial interest in the licensing agreement for PiB between GE Healthcare and the University of Pittsburgh.
On Twitter @Alz_Gal
FROM JAMA NEUROLOGY
Major finding: Amyloid was located with significantly greater concentration in the cortical gray matter and striatum of TBI patients, compared with controls, but not in the thalamus or white matter.
Data source: The study compared brain imaging scans of 15 patients with recent TBI and 11 healthy controls, and postmortem brain tissue samples between 16 TBI patients and 7 controls.
Disclosures: The National Institute for Health Research Cambridge Biomedical Research Centre funded the study. Two authors – Dr. William E. Klunk and Chester A. Mathis, Ph.D. – are coinventors of PiB and have a financial interest in the licensing agreement for PiB between GE Healthcare and the University of Pittsburgh.
Oxycodone-naloxone nips RLS symptoms after other treatment failures
A combination of extended-release oxycodone-naloxone significantly improved the symptoms of restless legs syndrome among patients who had failed first-line therapy, in a double-blind, randomized placebo-controlled study.
By the end of 12 weeks, 57% of patients randomized to the treatment group were considered responders, compared with 31% of patients in the placebo group, and 42% of patients in the treatment group had remission of symptoms, vs. 19% in the placebo group, Dr. Claudia Trenkwalder and her colleagues reported (Lancet Neuro 2013;12:1141-50). All of these differences were statistically significant.
"Our findings suggest a new, much-needed, option for management of severe restless legs syndrome for patients who cannot tolerate dopaminergic drugs," for nonresponders, and for those who developed tolerance or augmentation when receiving dopaminergic medications, wrote Dr. Trenkwalder of the department of neurosurgery, University of Göttingen (Germany) and her colleagues.
The study randomized 304 patients with severe restless legs syndrome (RLS) to either placebo (154) or to prolonged-release oxycodone-naloxone (150). The starting dose was 5 mg oxycodone with 2.5 mg naloxone twice a day. This could be titrated up to a maximum of 40 mg oxycodone with 20 mg naloxone twice a day. A 40-week open-label extension trial followed.
Patients had a mean disease duration of 10 years. All had failed first-line therapy with a dopaminergic drug. The mean International RLS Study Group symptom severity score was 31.6 on a 40-point scale, with 40 points corresponding to very severe symptoms. Change on this scale was the primary endpoint.
Of the entire 304 patients, 204 completed the 12-week randomized trial. Significantly more dropouts occurred in the treatment group (13% vs. 7%).
The mean daily dose was 22 mg hydrocodone with 11 mg naloxone. Ten patients took the maximum dose at some point during the study.
Symptoms began to decrease after 1 week among those taking the study drug. By 12 weeks, the symptom score had decreased a mean of 16.6 points in the treatment group and 9.5 points in the placebo group.
During this part of the study, 13% of the active group and 7% of the placebo group withdrew because of adverse events. The most common side effects were fatigue, constipation, nausea, and headache.
There were 197 patients who entered the extension phase; 157 of these completed the 40-week treatment period. At the end of the extension phase, 43% were classified as being in remission, with 22% having no symptoms at all.
The investigators cautioned that the study does not support using oxycodone-naloxone as a first-line therapy for RLS.
The study was sponsored by Mundipharma Research. Dr. Trenkwalder is a consultant for the company and reported financial relationships with numerous pharmaceutical companies. Three of the study’s coinvestigators are also Mundipharma employees, and several coinvestigators reported relationships with numerous pharmaceutical companies.
A combination of extended-release oxycodone-naloxone significantly improved the symptoms of restless legs syndrome among patients who had failed first-line therapy, in a double-blind, randomized placebo-controlled study.
By the end of 12 weeks, 57% of patients randomized to the treatment group were considered responders, compared with 31% of patients in the placebo group, and 42% of patients in the treatment group had remission of symptoms, vs. 19% in the placebo group, Dr. Claudia Trenkwalder and her colleagues reported (Lancet Neuro 2013;12:1141-50). All of these differences were statistically significant.
"Our findings suggest a new, much-needed, option for management of severe restless legs syndrome for patients who cannot tolerate dopaminergic drugs," for nonresponders, and for those who developed tolerance or augmentation when receiving dopaminergic medications, wrote Dr. Trenkwalder of the department of neurosurgery, University of Göttingen (Germany) and her colleagues.
The study randomized 304 patients with severe restless legs syndrome (RLS) to either placebo (154) or to prolonged-release oxycodone-naloxone (150). The starting dose was 5 mg oxycodone with 2.5 mg naloxone twice a day. This could be titrated up to a maximum of 40 mg oxycodone with 20 mg naloxone twice a day. A 40-week open-label extension trial followed.
Patients had a mean disease duration of 10 years. All had failed first-line therapy with a dopaminergic drug. The mean International RLS Study Group symptom severity score was 31.6 on a 40-point scale, with 40 points corresponding to very severe symptoms. Change on this scale was the primary endpoint.
Of the entire 304 patients, 204 completed the 12-week randomized trial. Significantly more dropouts occurred in the treatment group (13% vs. 7%).
The mean daily dose was 22 mg hydrocodone with 11 mg naloxone. Ten patients took the maximum dose at some point during the study.
Symptoms began to decrease after 1 week among those taking the study drug. By 12 weeks, the symptom score had decreased a mean of 16.6 points in the treatment group and 9.5 points in the placebo group.
During this part of the study, 13% of the active group and 7% of the placebo group withdrew because of adverse events. The most common side effects were fatigue, constipation, nausea, and headache.
There were 197 patients who entered the extension phase; 157 of these completed the 40-week treatment period. At the end of the extension phase, 43% were classified as being in remission, with 22% having no symptoms at all.
The investigators cautioned that the study does not support using oxycodone-naloxone as a first-line therapy for RLS.
The study was sponsored by Mundipharma Research. Dr. Trenkwalder is a consultant for the company and reported financial relationships with numerous pharmaceutical companies. Three of the study’s coinvestigators are also Mundipharma employees, and several coinvestigators reported relationships with numerous pharmaceutical companies.
A combination of extended-release oxycodone-naloxone significantly improved the symptoms of restless legs syndrome among patients who had failed first-line therapy, in a double-blind, randomized placebo-controlled study.
By the end of 12 weeks, 57% of patients randomized to the treatment group were considered responders, compared with 31% of patients in the placebo group, and 42% of patients in the treatment group had remission of symptoms, vs. 19% in the placebo group, Dr. Claudia Trenkwalder and her colleagues reported (Lancet Neuro 2013;12:1141-50). All of these differences were statistically significant.
"Our findings suggest a new, much-needed, option for management of severe restless legs syndrome for patients who cannot tolerate dopaminergic drugs," for nonresponders, and for those who developed tolerance or augmentation when receiving dopaminergic medications, wrote Dr. Trenkwalder of the department of neurosurgery, University of Göttingen (Germany) and her colleagues.
The study randomized 304 patients with severe restless legs syndrome (RLS) to either placebo (154) or to prolonged-release oxycodone-naloxone (150). The starting dose was 5 mg oxycodone with 2.5 mg naloxone twice a day. This could be titrated up to a maximum of 40 mg oxycodone with 20 mg naloxone twice a day. A 40-week open-label extension trial followed.
Patients had a mean disease duration of 10 years. All had failed first-line therapy with a dopaminergic drug. The mean International RLS Study Group symptom severity score was 31.6 on a 40-point scale, with 40 points corresponding to very severe symptoms. Change on this scale was the primary endpoint.
Of the entire 304 patients, 204 completed the 12-week randomized trial. Significantly more dropouts occurred in the treatment group (13% vs. 7%).
The mean daily dose was 22 mg hydrocodone with 11 mg naloxone. Ten patients took the maximum dose at some point during the study.
Symptoms began to decrease after 1 week among those taking the study drug. By 12 weeks, the symptom score had decreased a mean of 16.6 points in the treatment group and 9.5 points in the placebo group.
During this part of the study, 13% of the active group and 7% of the placebo group withdrew because of adverse events. The most common side effects were fatigue, constipation, nausea, and headache.
There were 197 patients who entered the extension phase; 157 of these completed the 40-week treatment period. At the end of the extension phase, 43% were classified as being in remission, with 22% having no symptoms at all.
The investigators cautioned that the study does not support using oxycodone-naloxone as a first-line therapy for RLS.
The study was sponsored by Mundipharma Research. Dr. Trenkwalder is a consultant for the company and reported financial relationships with numerous pharmaceutical companies. Three of the study’s coinvestigators are also Mundipharma employees, and several coinvestigators reported relationships with numerous pharmaceutical companies.
FROM LANCET NEUROLOGY
Major finding: After 12 weeks of oxycodone-naloxone therapy, 42% of patients with treatment-resistant restless legs syndrome had symptom remission.
Data source: The 12-week, placebo-controlled trial comprised 304 patients.
Disclosures: The study was sponsored by Mundipharma Research. Dr. Trenkwalder is a consultant for the company and reported financial relationships with numerous pharmaceutical companies. Three of the study’s coinvestigators are also Mundipharma employees, and several coinvestigators reported relationships with numerous pharmaceutical companies.