User login
Learning and feedback reduce cesarean section rate
NEW ORLEANS – A training program that continually reinforced best practices for labor and delivery reduced the risk of cesarean section by 10% overall, and by up to 32% in a subgroup of high-risk pregnancies.
The program also significantly improved newborn outcomes, with a 12% decrease in the rate of major neonatal morbidity, Dr. Nils Chaillet reported at the Pregnancy Meeting, sponsored by the Society of Maternal-Fetal Medicine. The neonatal benefit remained significant even after excluding all preterm births from the analysis, said Dr. Chaillet of the Sainte-Justine University Hospital Research Center, Montreal.
The QUARISMA (Quality of Care, Management of Obstetrical Risk, and Mode of Delivery in Quebec) study examined the effects of a clinician training program designed to promote optimal management of labor and delivery. QUARISMA was a multifaceted evidence-medicine program based on audit, feedback, and educational activities that followed the World Health Organization guidelines for quality obstetrical care.
A 2-day training session at each participating hospital introduced the program. Throughout the 3.5-year study, a local audit committee, headed by a peer-selected leader, regularly evaluated performance and provided feedback on results and suggestions on quality improvement. These audits were conducted every 3 months. They included an analysis of each cesarean case, centering on whether and how it could have been prevented.
The study randomized 32 public hospitals in Quebec to the intervention or to a control arm of usual practice. The hospitals all had a cesarean section rate of at least 17% and no prior program aimed at reducing that rate. They had to perform more than 300 deliveries annually. All women who gave birth to an infant of at least 500 grams and 24 weeks’ gestation were included in the study.
A total of 105,351 deliveries occurred. In the control group, the baseline rate of cesarean section was 23%; that remained unchanged. The rate did decrease in the intervention group, dropping from 22.5% at baseline to 21.8% at study’s end – a 10% risk reduction that was statistically significant.
Women with low-risk pregnancies experienced a considerable benefit, Dr. Chaillet noted, with an overall risk reduction of 20%. That varied among hospital type. It was smallest (2%) at community hospitals, followed by 21% at regional hospitals and 24% at tertiary care hospitals.
Among high-risk pregnancies, the risk of a cesarean decreased by 4% overall. It did not change in tertiary care centers, but decreased by 5% at regional hospitals and 32% at community hospitals.
Medical interventions were a secondary endpoint. There were no changes in the rates of episiotomy, epidurals, or artificial rupture of membranes. However, the risk of operative vaginal delivery dropped by 12%, and pharmacologic labor induction by 18%. The odds of using oxytocin during labor increased by16%.
There were no significant changes in the composite measures of minor or major maternal morbidities. But neonatal outcomes did improve. The risk of a minor neonatal morbidity decreased by a significant 12%; the risk of a major problem decreased by 19%.
Dr. Chaillet made no financial disclosures.
On Twitter @alz_gal
NEW ORLEANS – A training program that continually reinforced best practices for labor and delivery reduced the risk of cesarean section by 10% overall, and by up to 32% in a subgroup of high-risk pregnancies.
The program also significantly improved newborn outcomes, with a 12% decrease in the rate of major neonatal morbidity, Dr. Nils Chaillet reported at the Pregnancy Meeting, sponsored by the Society of Maternal-Fetal Medicine. The neonatal benefit remained significant even after excluding all preterm births from the analysis, said Dr. Chaillet of the Sainte-Justine University Hospital Research Center, Montreal.
The QUARISMA (Quality of Care, Management of Obstetrical Risk, and Mode of Delivery in Quebec) study examined the effects of a clinician training program designed to promote optimal management of labor and delivery. QUARISMA was a multifaceted evidence-medicine program based on audit, feedback, and educational activities that followed the World Health Organization guidelines for quality obstetrical care.
A 2-day training session at each participating hospital introduced the program. Throughout the 3.5-year study, a local audit committee, headed by a peer-selected leader, regularly evaluated performance and provided feedback on results and suggestions on quality improvement. These audits were conducted every 3 months. They included an analysis of each cesarean case, centering on whether and how it could have been prevented.
The study randomized 32 public hospitals in Quebec to the intervention or to a control arm of usual practice. The hospitals all had a cesarean section rate of at least 17% and no prior program aimed at reducing that rate. They had to perform more than 300 deliveries annually. All women who gave birth to an infant of at least 500 grams and 24 weeks’ gestation were included in the study.
A total of 105,351 deliveries occurred. In the control group, the baseline rate of cesarean section was 23%; that remained unchanged. The rate did decrease in the intervention group, dropping from 22.5% at baseline to 21.8% at study’s end – a 10% risk reduction that was statistically significant.
Women with low-risk pregnancies experienced a considerable benefit, Dr. Chaillet noted, with an overall risk reduction of 20%. That varied among hospital type. It was smallest (2%) at community hospitals, followed by 21% at regional hospitals and 24% at tertiary care hospitals.
Among high-risk pregnancies, the risk of a cesarean decreased by 4% overall. It did not change in tertiary care centers, but decreased by 5% at regional hospitals and 32% at community hospitals.
Medical interventions were a secondary endpoint. There were no changes in the rates of episiotomy, epidurals, or artificial rupture of membranes. However, the risk of operative vaginal delivery dropped by 12%, and pharmacologic labor induction by 18%. The odds of using oxytocin during labor increased by16%.
There were no significant changes in the composite measures of minor or major maternal morbidities. But neonatal outcomes did improve. The risk of a minor neonatal morbidity decreased by a significant 12%; the risk of a major problem decreased by 19%.
Dr. Chaillet made no financial disclosures.
On Twitter @alz_gal
NEW ORLEANS – A training program that continually reinforced best practices for labor and delivery reduced the risk of cesarean section by 10% overall, and by up to 32% in a subgroup of high-risk pregnancies.
The program also significantly improved newborn outcomes, with a 12% decrease in the rate of major neonatal morbidity, Dr. Nils Chaillet reported at the Pregnancy Meeting, sponsored by the Society of Maternal-Fetal Medicine. The neonatal benefit remained significant even after excluding all preterm births from the analysis, said Dr. Chaillet of the Sainte-Justine University Hospital Research Center, Montreal.
The QUARISMA (Quality of Care, Management of Obstetrical Risk, and Mode of Delivery in Quebec) study examined the effects of a clinician training program designed to promote optimal management of labor and delivery. QUARISMA was a multifaceted evidence-medicine program based on audit, feedback, and educational activities that followed the World Health Organization guidelines for quality obstetrical care.
A 2-day training session at each participating hospital introduced the program. Throughout the 3.5-year study, a local audit committee, headed by a peer-selected leader, regularly evaluated performance and provided feedback on results and suggestions on quality improvement. These audits were conducted every 3 months. They included an analysis of each cesarean case, centering on whether and how it could have been prevented.
The study randomized 32 public hospitals in Quebec to the intervention or to a control arm of usual practice. The hospitals all had a cesarean section rate of at least 17% and no prior program aimed at reducing that rate. They had to perform more than 300 deliveries annually. All women who gave birth to an infant of at least 500 grams and 24 weeks’ gestation were included in the study.
A total of 105,351 deliveries occurred. In the control group, the baseline rate of cesarean section was 23%; that remained unchanged. The rate did decrease in the intervention group, dropping from 22.5% at baseline to 21.8% at study’s end – a 10% risk reduction that was statistically significant.
Women with low-risk pregnancies experienced a considerable benefit, Dr. Chaillet noted, with an overall risk reduction of 20%. That varied among hospital type. It was smallest (2%) at community hospitals, followed by 21% at regional hospitals and 24% at tertiary care hospitals.
Among high-risk pregnancies, the risk of a cesarean decreased by 4% overall. It did not change in tertiary care centers, but decreased by 5% at regional hospitals and 32% at community hospitals.
Medical interventions were a secondary endpoint. There were no changes in the rates of episiotomy, epidurals, or artificial rupture of membranes. However, the risk of operative vaginal delivery dropped by 12%, and pharmacologic labor induction by 18%. The odds of using oxytocin during labor increased by16%.
There were no significant changes in the composite measures of minor or major maternal morbidities. But neonatal outcomes did improve. The risk of a minor neonatal morbidity decreased by a significant 12%; the risk of a major problem decreased by 19%.
Dr. Chaillet made no financial disclosures.
On Twitter @alz_gal
AT THE PREGNANCY MEETING
Major finding: Among high-risk pregnancies, the risk of a cesarean decreased by 4% overall. It did not change in tertiary care centers, but decreased by 5% at regional hospitals and 32% at community hospitals.
Data source: The study randomized 32 hospitals and tracked outcomes in more than 100,000 births.
Disclosures: Dr. Chaillet had no financial disclosures.
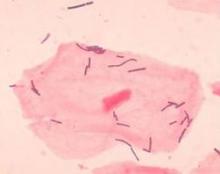
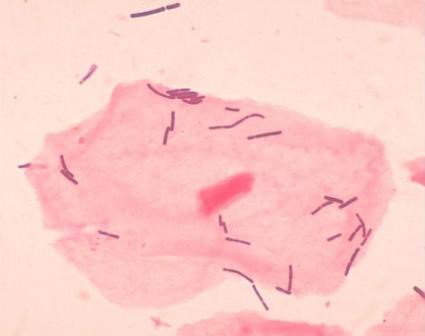
Intra-amniotic debris associated with preterm birth, not growth restriction
NEW ORLEANS – Intra-amniotic debris can portend a preterm birth, but doesn’t seem to be associated with any other adverse pregnancy outcomes, including small-for-gestational-age infants.
The debris – a mix of bacterial and inflammatory cells encased in an antibiotic-resistant biofilm – may actually trigger an early birth to help protect the fetus, Dr. George R. Saade said at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
The debris is associated with placental senescence, "which leads to preterm birth before there can be further fetal compromise and growth restriction," said Dr. Saade, director of maternal-fetal medicine at the University of Texas Medical Branch, Galveston.
He presented a secondary analysis of a study that examined the effect of progesterone injections to prevent preterm birth in nulliparous women with a short cervix.
The study was halted in October 2012, after preliminary results showed no treatment benefit.
The main outcome for the study was a composite of fetal death, small-for-gestational-age infants, placental abruption, and gestational hypertension/preeclampsia. Of the 657 randomized to progesterone or placebo, 78 were found to have intra-amniotic debris on ultrasound.
These women were significantly older than were those without debris (23 vs. 22 years), had a significantly higher prepregnancy body mass index (28 kg/m2 vs. 25 kg/m2), and a significantly shorter cervical length on ultrasound (19 vs. 24 cm).
There were significantly more infants born at less than 37 weeks’ gestation in women with debris (35% vs. 23%). But there was no significant between-group difference in the composite outcome. When Dr. Saade looked at the individual outcome measures, he found only one significant difference. Despite the larger number of preterm births, there were no small-for-gestational-age infants born to mothers with the debris. However, 5% of those without debris did have small-for-dates babies.
Dr. Saade had no financial declarations.
NEW ORLEANS – Intra-amniotic debris can portend a preterm birth, but doesn’t seem to be associated with any other adverse pregnancy outcomes, including small-for-gestational-age infants.
The debris – a mix of bacterial and inflammatory cells encased in an antibiotic-resistant biofilm – may actually trigger an early birth to help protect the fetus, Dr. George R. Saade said at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
The debris is associated with placental senescence, "which leads to preterm birth before there can be further fetal compromise and growth restriction," said Dr. Saade, director of maternal-fetal medicine at the University of Texas Medical Branch, Galveston.
He presented a secondary analysis of a study that examined the effect of progesterone injections to prevent preterm birth in nulliparous women with a short cervix.
The study was halted in October 2012, after preliminary results showed no treatment benefit.
The main outcome for the study was a composite of fetal death, small-for-gestational-age infants, placental abruption, and gestational hypertension/preeclampsia. Of the 657 randomized to progesterone or placebo, 78 were found to have intra-amniotic debris on ultrasound.
These women were significantly older than were those without debris (23 vs. 22 years), had a significantly higher prepregnancy body mass index (28 kg/m2 vs. 25 kg/m2), and a significantly shorter cervical length on ultrasound (19 vs. 24 cm).
There were significantly more infants born at less than 37 weeks’ gestation in women with debris (35% vs. 23%). But there was no significant between-group difference in the composite outcome. When Dr. Saade looked at the individual outcome measures, he found only one significant difference. Despite the larger number of preterm births, there were no small-for-gestational-age infants born to mothers with the debris. However, 5% of those without debris did have small-for-dates babies.
Dr. Saade had no financial declarations.
NEW ORLEANS – Intra-amniotic debris can portend a preterm birth, but doesn’t seem to be associated with any other adverse pregnancy outcomes, including small-for-gestational-age infants.
The debris – a mix of bacterial and inflammatory cells encased in an antibiotic-resistant biofilm – may actually trigger an early birth to help protect the fetus, Dr. George R. Saade said at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
The debris is associated with placental senescence, "which leads to preterm birth before there can be further fetal compromise and growth restriction," said Dr. Saade, director of maternal-fetal medicine at the University of Texas Medical Branch, Galveston.
He presented a secondary analysis of a study that examined the effect of progesterone injections to prevent preterm birth in nulliparous women with a short cervix.
The study was halted in October 2012, after preliminary results showed no treatment benefit.
The main outcome for the study was a composite of fetal death, small-for-gestational-age infants, placental abruption, and gestational hypertension/preeclampsia. Of the 657 randomized to progesterone or placebo, 78 were found to have intra-amniotic debris on ultrasound.
These women were significantly older than were those without debris (23 vs. 22 years), had a significantly higher prepregnancy body mass index (28 kg/m2 vs. 25 kg/m2), and a significantly shorter cervical length on ultrasound (19 vs. 24 cm).
There were significantly more infants born at less than 37 weeks’ gestation in women with debris (35% vs. 23%). But there was no significant between-group difference in the composite outcome. When Dr. Saade looked at the individual outcome measures, he found only one significant difference. Despite the larger number of preterm births, there were no small-for-gestational-age infants born to mothers with the debris. However, 5% of those without debris did have small-for-dates babies.
Dr. Saade had no financial declarations.
AT THE PREGNANCY MEETING
Major finding: Preterm birth occurred in 35% of women who had intra-amniotic debris, but none of those infants were small for gestational age.
Data source: The analysis comprised 657 women.
Disclosures: Dr. George Saade had no financial disclosures.
Treating pediatric bipolar disorder means leaning on adult data
NEW YORK – Treating children and teens with refractory bipolar disorder requires both persistence and faith, according to Dr. Gabrielle A. Carlson.
Although new data are trickling out, few of the medications available for treating both mania and depression have been extensively studied in children and teens, Dr. Carlson said at a psychopharmacology update held by the American Academy of Child and Adolescent Psychiatry.
"The first-generation antipsychotics have been grandfathered in with [Food and Drug Administration] approval. There are very little data for youngsters, and it’s unlikely that there ever will be, since they are generic now and no one will be spending the money" to further investigate them.
A few pediatric trials are in progress for some of the newer, second-generation antipsychotics, but most efficacy data are still from adult studies, said Dr. Carlson, professor of psychiatry and pediatrics at Stony Brook University, New York.
Most recently, paloperidone was found effective as an acute treatment for mania in children with bipolar disorder. But the open-label study was very small – just 15 patients – and was terminated early because of the slow enrollment. The study tested two doses (3 mg and 12 mg). There was a significant mean decline of almost 19 points on the Young Mania Rating Scale after 8 weeks of treatment. Children gained a mean of 4 pounds, however (Psychopharmacology [Berl.] 2013;227:449-58).
Sublingual treatment
Asenapine "is the new kid on the block," Dr. Carlson said. It was approved in 2009 for the acute treatment of schizophrenia and manic or mixed bipolar episodes in adults. It’s administered as a sublingual tablet that takes about 10 minutes to dissolve.
"This is good news if you’ve got a kid who has trouble swallowing pills. The bad news is that you have to hold it under your tongue for 10 minutes, and if you’ve got an aggressive, agitated kid – well, good luck with that. If he does it, though, you can get a good treatment response pretty quickly, because it has a peak concentration of 30-90 minutes and a half-life of about 24 hours."
The adult studies of asenapine showed a significant benefit in about 1 week, with many bipolar patients seeing a 50% reduction in symptoms. Two placebo-controlled trials in children are just wrapping up. The first, comprising 400 children, is complete, but no data have yet been published.
The second, a 50-week open-label study, is ongoing; about 300 children are enrolled.
In the adult studies of asenapine, the number needed to treat was about eight. "The NNT for many other drugs is four or five, so to me this looks like a contender, but not anything great. The sublingual route is the biggest benefit," although, Dr. Carlson cautioned, "it does make noncompliance a lot easier, just by swallowing it." The bioavailability when swallowed is only about 35%.
Asenapine is sedating, so it might be a good choice for as-needed treatment; the weight gain profile is similar to that of olanzapine. The twice-daily dosing is weight based, Dr. Carlson said. "Younger children are more sensitive to the initial dose, so they need a lower starting point and shorter up-titration, which isn’t necessary in the older kids."
For children with bipolar depression, there’s little evidence to support the use of quetiapine. There are no data in youngsters and few data to support its use in adults. A 2012 meta-analysis found that the drug is no better than placebo.
The combination of olanzapine and fluoxetine has been examined in children aged 10-17 years. The industry-sponsored placebo-controlled study treated 160 children for 8 weeks.
At the end of the study, remission rates were 59% in the active group and 43% in the placebo group – a significant difference. Children taking the combination gained significantly more weight than those taking placebo – an average of 4 vs. 0.5 kg. There were also significant increases in liver enzymes and prolactin in those taking the drugs, as well as a higher chance of prolonged QTc intervals.
Unpublished results of another study hint that lamotrigine also might be effective in children with bipolar depression, Dr. Carlson said.
The 8-week study looked at what happened to children aged 10-17 years who had lamotrigine withdrawn after they had responded to it. These patients were given the option of continuing the drug or switching to placebo.
"So far they have 250 enrolled," Dr. Carlson said. "Of these, 136 were willing to be randomized, meaning that at least 60% got better in the open-label phase."
There are no published pediatric data on lurasidone or ketamine, although both seem promising. Lurasidone has been approved for adults, and a study being done in youngsters is not specific for bipolar disorder. Children with schizophrenia, psychosis, autism disorders, Asperger’s syndrome, and refractory major depressive disorder also can enroll.
A recently published adult study, PREVAIL 2 (Program to Evaluate the Antidepressant Impact of Lurasidone), comprised 505 patients with bipolar I depression. They were randomized to once-daily, flexibly dosed lurasidone at either 20-60 mg/day or 80-120 mg/day, or to placebo.
After 6 weeks, depression scores fell in both the 20- to 60-mg/day group (–15.4; effect size = 0.51) and the 80- to 120-mg/day group (–15.4; effect size =0.51), compared with placebo (–10.7).
Similarly, lurasidone treatment resulted in significantly greater endpoint reduction in Clinical Global Impressions-Bipolar Scale depression severity scores for both the 20- to 60-mg/day group (–1.8; effect size = 0.61) and the 80- to 120-mg/day group (–1.7; effect size = 0.50), compared with placebo (–1.1). Both lurasidone groups also experienced significant improvements compared with placebo in anxiety symptoms, and in patient-reported measures of quality of life and functional impairment. Response rates were 53% for the active group and 30% for the placebo group, with remission rates of 42% vs. 40%.
"The effect size was similar for the high and low doses, about 0.5, which is not terrible," Dr. Carlson said. "Discontinuation rates were fairly low, suggesting that it may be worth pursuing."
The main side effects were nausea, headache, akathisia, and somnolence, but very few changes were found in weight, lipids, or measures of glycemic control.
"Ketamine is something we’ve been hearing a lot about," Dr. Carlson said. "If we can come up with a way to give it that’s not intravenous and make it last for a while, it could be really valuable."
One study found that adults randomized to ketamine had significantly greater improvements in depression scores after 24 hours than did those who had midazolam.
The study comprised 73 patients. Response rates at 24 hours were 64% vs. 28% for placebo – with patients taking ketamine more than twice as likely to respond in the adjusted analysis.
The response was highest in the first 3 days after infusion; by day 7, it had dropped to about 45%. A review also highlighted ketamine's 'Achilles heel' – it just hasn’t got staying power. The paper looked at effect sizes in two depression studies and found that they peaked around 2 or 3 days after infusion. But 2 weeks later, the effect size was just about the same as with placebo.
"It’s a really interesting drug, and it does appear to hold some promise," Dr. Carlson said.
Dr. Carlson noted that she has received research funding from Bristol-Myers Squibb, GlaxoSmithKline, Merck & Co., Otsuka America Pharmaceutical, Pfizer, and Schering-Plough.
NEW YORK – Treating children and teens with refractory bipolar disorder requires both persistence and faith, according to Dr. Gabrielle A. Carlson.
Although new data are trickling out, few of the medications available for treating both mania and depression have been extensively studied in children and teens, Dr. Carlson said at a psychopharmacology update held by the American Academy of Child and Adolescent Psychiatry.
"The first-generation antipsychotics have been grandfathered in with [Food and Drug Administration] approval. There are very little data for youngsters, and it’s unlikely that there ever will be, since they are generic now and no one will be spending the money" to further investigate them.
A few pediatric trials are in progress for some of the newer, second-generation antipsychotics, but most efficacy data are still from adult studies, said Dr. Carlson, professor of psychiatry and pediatrics at Stony Brook University, New York.
Most recently, paloperidone was found effective as an acute treatment for mania in children with bipolar disorder. But the open-label study was very small – just 15 patients – and was terminated early because of the slow enrollment. The study tested two doses (3 mg and 12 mg). There was a significant mean decline of almost 19 points on the Young Mania Rating Scale after 8 weeks of treatment. Children gained a mean of 4 pounds, however (Psychopharmacology [Berl.] 2013;227:449-58).
Sublingual treatment
Asenapine "is the new kid on the block," Dr. Carlson said. It was approved in 2009 for the acute treatment of schizophrenia and manic or mixed bipolar episodes in adults. It’s administered as a sublingual tablet that takes about 10 minutes to dissolve.
"This is good news if you’ve got a kid who has trouble swallowing pills. The bad news is that you have to hold it under your tongue for 10 minutes, and if you’ve got an aggressive, agitated kid – well, good luck with that. If he does it, though, you can get a good treatment response pretty quickly, because it has a peak concentration of 30-90 minutes and a half-life of about 24 hours."
The adult studies of asenapine showed a significant benefit in about 1 week, with many bipolar patients seeing a 50% reduction in symptoms. Two placebo-controlled trials in children are just wrapping up. The first, comprising 400 children, is complete, but no data have yet been published.
The second, a 50-week open-label study, is ongoing; about 300 children are enrolled.
In the adult studies of asenapine, the number needed to treat was about eight. "The NNT for many other drugs is four or five, so to me this looks like a contender, but not anything great. The sublingual route is the biggest benefit," although, Dr. Carlson cautioned, "it does make noncompliance a lot easier, just by swallowing it." The bioavailability when swallowed is only about 35%.
Asenapine is sedating, so it might be a good choice for as-needed treatment; the weight gain profile is similar to that of olanzapine. The twice-daily dosing is weight based, Dr. Carlson said. "Younger children are more sensitive to the initial dose, so they need a lower starting point and shorter up-titration, which isn’t necessary in the older kids."
For children with bipolar depression, there’s little evidence to support the use of quetiapine. There are no data in youngsters and few data to support its use in adults. A 2012 meta-analysis found that the drug is no better than placebo.
The combination of olanzapine and fluoxetine has been examined in children aged 10-17 years. The industry-sponsored placebo-controlled study treated 160 children for 8 weeks.
At the end of the study, remission rates were 59% in the active group and 43% in the placebo group – a significant difference. Children taking the combination gained significantly more weight than those taking placebo – an average of 4 vs. 0.5 kg. There were also significant increases in liver enzymes and prolactin in those taking the drugs, as well as a higher chance of prolonged QTc intervals.
Unpublished results of another study hint that lamotrigine also might be effective in children with bipolar depression, Dr. Carlson said.
The 8-week study looked at what happened to children aged 10-17 years who had lamotrigine withdrawn after they had responded to it. These patients were given the option of continuing the drug or switching to placebo.
"So far they have 250 enrolled," Dr. Carlson said. "Of these, 136 were willing to be randomized, meaning that at least 60% got better in the open-label phase."
There are no published pediatric data on lurasidone or ketamine, although both seem promising. Lurasidone has been approved for adults, and a study being done in youngsters is not specific for bipolar disorder. Children with schizophrenia, psychosis, autism disorders, Asperger’s syndrome, and refractory major depressive disorder also can enroll.
A recently published adult study, PREVAIL 2 (Program to Evaluate the Antidepressant Impact of Lurasidone), comprised 505 patients with bipolar I depression. They were randomized to once-daily, flexibly dosed lurasidone at either 20-60 mg/day or 80-120 mg/day, or to placebo.
After 6 weeks, depression scores fell in both the 20- to 60-mg/day group (–15.4; effect size = 0.51) and the 80- to 120-mg/day group (–15.4; effect size =0.51), compared with placebo (–10.7).
Similarly, lurasidone treatment resulted in significantly greater endpoint reduction in Clinical Global Impressions-Bipolar Scale depression severity scores for both the 20- to 60-mg/day group (–1.8; effect size = 0.61) and the 80- to 120-mg/day group (–1.7; effect size = 0.50), compared with placebo (–1.1). Both lurasidone groups also experienced significant improvements compared with placebo in anxiety symptoms, and in patient-reported measures of quality of life and functional impairment. Response rates were 53% for the active group and 30% for the placebo group, with remission rates of 42% vs. 40%.
"The effect size was similar for the high and low doses, about 0.5, which is not terrible," Dr. Carlson said. "Discontinuation rates were fairly low, suggesting that it may be worth pursuing."
The main side effects were nausea, headache, akathisia, and somnolence, but very few changes were found in weight, lipids, or measures of glycemic control.
"Ketamine is something we’ve been hearing a lot about," Dr. Carlson said. "If we can come up with a way to give it that’s not intravenous and make it last for a while, it could be really valuable."
One study found that adults randomized to ketamine had significantly greater improvements in depression scores after 24 hours than did those who had midazolam.
The study comprised 73 patients. Response rates at 24 hours were 64% vs. 28% for placebo – with patients taking ketamine more than twice as likely to respond in the adjusted analysis.
The response was highest in the first 3 days after infusion; by day 7, it had dropped to about 45%. A review also highlighted ketamine's 'Achilles heel' – it just hasn’t got staying power. The paper looked at effect sizes in two depression studies and found that they peaked around 2 or 3 days after infusion. But 2 weeks later, the effect size was just about the same as with placebo.
"It’s a really interesting drug, and it does appear to hold some promise," Dr. Carlson said.
Dr. Carlson noted that she has received research funding from Bristol-Myers Squibb, GlaxoSmithKline, Merck & Co., Otsuka America Pharmaceutical, Pfizer, and Schering-Plough.
NEW YORK – Treating children and teens with refractory bipolar disorder requires both persistence and faith, according to Dr. Gabrielle A. Carlson.
Although new data are trickling out, few of the medications available for treating both mania and depression have been extensively studied in children and teens, Dr. Carlson said at a psychopharmacology update held by the American Academy of Child and Adolescent Psychiatry.
"The first-generation antipsychotics have been grandfathered in with [Food and Drug Administration] approval. There are very little data for youngsters, and it’s unlikely that there ever will be, since they are generic now and no one will be spending the money" to further investigate them.
A few pediatric trials are in progress for some of the newer, second-generation antipsychotics, but most efficacy data are still from adult studies, said Dr. Carlson, professor of psychiatry and pediatrics at Stony Brook University, New York.
Most recently, paloperidone was found effective as an acute treatment for mania in children with bipolar disorder. But the open-label study was very small – just 15 patients – and was terminated early because of the slow enrollment. The study tested two doses (3 mg and 12 mg). There was a significant mean decline of almost 19 points on the Young Mania Rating Scale after 8 weeks of treatment. Children gained a mean of 4 pounds, however (Psychopharmacology [Berl.] 2013;227:449-58).
Sublingual treatment
Asenapine "is the new kid on the block," Dr. Carlson said. It was approved in 2009 for the acute treatment of schizophrenia and manic or mixed bipolar episodes in adults. It’s administered as a sublingual tablet that takes about 10 minutes to dissolve.
"This is good news if you’ve got a kid who has trouble swallowing pills. The bad news is that you have to hold it under your tongue for 10 minutes, and if you’ve got an aggressive, agitated kid – well, good luck with that. If he does it, though, you can get a good treatment response pretty quickly, because it has a peak concentration of 30-90 minutes and a half-life of about 24 hours."
The adult studies of asenapine showed a significant benefit in about 1 week, with many bipolar patients seeing a 50% reduction in symptoms. Two placebo-controlled trials in children are just wrapping up. The first, comprising 400 children, is complete, but no data have yet been published.
The second, a 50-week open-label study, is ongoing; about 300 children are enrolled.
In the adult studies of asenapine, the number needed to treat was about eight. "The NNT for many other drugs is four or five, so to me this looks like a contender, but not anything great. The sublingual route is the biggest benefit," although, Dr. Carlson cautioned, "it does make noncompliance a lot easier, just by swallowing it." The bioavailability when swallowed is only about 35%.
Asenapine is sedating, so it might be a good choice for as-needed treatment; the weight gain profile is similar to that of olanzapine. The twice-daily dosing is weight based, Dr. Carlson said. "Younger children are more sensitive to the initial dose, so they need a lower starting point and shorter up-titration, which isn’t necessary in the older kids."
For children with bipolar depression, there’s little evidence to support the use of quetiapine. There are no data in youngsters and few data to support its use in adults. A 2012 meta-analysis found that the drug is no better than placebo.
The combination of olanzapine and fluoxetine has been examined in children aged 10-17 years. The industry-sponsored placebo-controlled study treated 160 children for 8 weeks.
At the end of the study, remission rates were 59% in the active group and 43% in the placebo group – a significant difference. Children taking the combination gained significantly more weight than those taking placebo – an average of 4 vs. 0.5 kg. There were also significant increases in liver enzymes and prolactin in those taking the drugs, as well as a higher chance of prolonged QTc intervals.
Unpublished results of another study hint that lamotrigine also might be effective in children with bipolar depression, Dr. Carlson said.
The 8-week study looked at what happened to children aged 10-17 years who had lamotrigine withdrawn after they had responded to it. These patients were given the option of continuing the drug or switching to placebo.
"So far they have 250 enrolled," Dr. Carlson said. "Of these, 136 were willing to be randomized, meaning that at least 60% got better in the open-label phase."
There are no published pediatric data on lurasidone or ketamine, although both seem promising. Lurasidone has been approved for adults, and a study being done in youngsters is not specific for bipolar disorder. Children with schizophrenia, psychosis, autism disorders, Asperger’s syndrome, and refractory major depressive disorder also can enroll.
A recently published adult study, PREVAIL 2 (Program to Evaluate the Antidepressant Impact of Lurasidone), comprised 505 patients with bipolar I depression. They were randomized to once-daily, flexibly dosed lurasidone at either 20-60 mg/day or 80-120 mg/day, or to placebo.
After 6 weeks, depression scores fell in both the 20- to 60-mg/day group (–15.4; effect size = 0.51) and the 80- to 120-mg/day group (–15.4; effect size =0.51), compared with placebo (–10.7).
Similarly, lurasidone treatment resulted in significantly greater endpoint reduction in Clinical Global Impressions-Bipolar Scale depression severity scores for both the 20- to 60-mg/day group (–1.8; effect size = 0.61) and the 80- to 120-mg/day group (–1.7; effect size = 0.50), compared with placebo (–1.1). Both lurasidone groups also experienced significant improvements compared with placebo in anxiety symptoms, and in patient-reported measures of quality of life and functional impairment. Response rates were 53% for the active group and 30% for the placebo group, with remission rates of 42% vs. 40%.
"The effect size was similar for the high and low doses, about 0.5, which is not terrible," Dr. Carlson said. "Discontinuation rates were fairly low, suggesting that it may be worth pursuing."
The main side effects were nausea, headache, akathisia, and somnolence, but very few changes were found in weight, lipids, or measures of glycemic control.
"Ketamine is something we’ve been hearing a lot about," Dr. Carlson said. "If we can come up with a way to give it that’s not intravenous and make it last for a while, it could be really valuable."
One study found that adults randomized to ketamine had significantly greater improvements in depression scores after 24 hours than did those who had midazolam.
The study comprised 73 patients. Response rates at 24 hours were 64% vs. 28% for placebo – with patients taking ketamine more than twice as likely to respond in the adjusted analysis.
The response was highest in the first 3 days after infusion; by day 7, it had dropped to about 45%. A review also highlighted ketamine's 'Achilles heel' – it just hasn’t got staying power. The paper looked at effect sizes in two depression studies and found that they peaked around 2 or 3 days after infusion. But 2 weeks later, the effect size was just about the same as with placebo.
"It’s a really interesting drug, and it does appear to hold some promise," Dr. Carlson said.
Dr. Carlson noted that she has received research funding from Bristol-Myers Squibb, GlaxoSmithKline, Merck & Co., Otsuka America Pharmaceutical, Pfizer, and Schering-Plough.
AT THE PSYCHOPHARMACOLOGY UPDATE INSTITUTE
For women with prior cesareans, 39-week delivery is not optimal
NEW ORLEANS – The safest time to deliver a woman with prior cesarean sections may be up to 2 weeks earlier than the current recommendation.
Guidelines from a number of national groups, including the Society for Maternal-Fetal Medicine, recommend planning a cesarean delivery at 39 weeks for these women. But a retrospective study has determined that maternal risk increases sharply after 37 weeks for women who have had three or more cesarean deliveries – a full 2 weeks before there is any increase in fetal risk.
"The data clearly show that the optimal time for delivery in women with three or more prior cesareans is 37 weeks," Dr. Laura Hart said at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
Dr. Hart of the University of Texas Health Science Center, Houston, extracted her data from the Maternal-Fetal Medicine Units (MFMU) Cesarean Section Registry. It contains prospective data on more than 24,000 deliveries performed after prior cesarean. She examined information on 6,435 deliveries that followed at least two prior cesareans (80% after two sections and 20% after three or more).
Dr. Hart analyzed both maternal and fetal outcomes. Each was a single composite comprising several endpoints. The maternal composite took into account transfusion, hysterectomy, operative injury, thromboembolic event, pulmonary edema, and death. The perinatal composite included respiratory distress syndrome, necrotizing enterocolitis (grade 2 or 3), intraventricular hemorrhage (grade 3 or 4), hypoxic ischemic injury, seizure, and stillbirth or neonatal death.
The results were expressed as a risk of the composite event relative to the number of ongoing pregnancies.
Less than half of the women who had two prior cesareans delivered at 39 weeks (46%); 11% delivered at 37 weeks, 31% at 38 weeks, and 13% at 40 weeks or more.
For these women, the risk of maternal complications was less than 5 per 1,000 ongoing pregnancies at 37 weeks. It remained low at 38 weeks (about 7/1,000). But between 38 and 39 weeks, it rose sharply, to 16/1,000. The risk remained similarly elevated at 40 weeks and beyond.
The perinatal outcomes risk was low at 37 weeks (7/1,000) and remained low at 38 and 39 weeks (7 and 9/1,000, respectively). The risk also increased sharply between 38 and 39 weeks, rising to about 20/1,000. The maternal and perinatal risk curves intersected between weeks 37 and 38, indicating that 38 weeks was probably the optimal time for delivery.
Less than half of women who had at least three prior cesareans also made it to 39 weeks (41%). About a third (35%) delivered at 38 weeks, 15% at 37 weeks, and 9% at 40 weeks or more.
Again, maternal risk was very low at 37 weeks (about 4/1,000 ongoing pregnancies). But in contrast to the risk for women with two prior deliveries, the risk for these women increased sharply and linearly over the final weeks of gestation. By 38 weeks, it had jumped to about 16/1,000. By 39 weeks, it was close to 50/1,000, and by 40 weeks and beyond, more than 50/1,000.
Conversely, perinatal risk remained fairly low in weeks 37, 38, and 39 (9, 7, and 10/1,000, respectively). Risk accelerated between weeks 39 and 40; by week 40 and beyond, it hovered just above 25/1,000. The risk curves intersected between weeks 37 and 38, indicating that 37 weeks is the optimal time for delivery.
Dr. Hart added that maternal transfusion was the main risk driver for women, followed closely by operative injury and hysterectomy.
These data were published in a supplement to the American Journal of Obstetrics and Gynecology (Amer. J. Obstet. Gynecol. 2014;210(suppl.):S27).
Dr. Hart had no financial disclosures.
NEW ORLEANS – The safest time to deliver a woman with prior cesarean sections may be up to 2 weeks earlier than the current recommendation.
Guidelines from a number of national groups, including the Society for Maternal-Fetal Medicine, recommend planning a cesarean delivery at 39 weeks for these women. But a retrospective study has determined that maternal risk increases sharply after 37 weeks for women who have had three or more cesarean deliveries – a full 2 weeks before there is any increase in fetal risk.
"The data clearly show that the optimal time for delivery in women with three or more prior cesareans is 37 weeks," Dr. Laura Hart said at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
Dr. Hart of the University of Texas Health Science Center, Houston, extracted her data from the Maternal-Fetal Medicine Units (MFMU) Cesarean Section Registry. It contains prospective data on more than 24,000 deliveries performed after prior cesarean. She examined information on 6,435 deliveries that followed at least two prior cesareans (80% after two sections and 20% after three or more).
Dr. Hart analyzed both maternal and fetal outcomes. Each was a single composite comprising several endpoints. The maternal composite took into account transfusion, hysterectomy, operative injury, thromboembolic event, pulmonary edema, and death. The perinatal composite included respiratory distress syndrome, necrotizing enterocolitis (grade 2 or 3), intraventricular hemorrhage (grade 3 or 4), hypoxic ischemic injury, seizure, and stillbirth or neonatal death.
The results were expressed as a risk of the composite event relative to the number of ongoing pregnancies.
Less than half of the women who had two prior cesareans delivered at 39 weeks (46%); 11% delivered at 37 weeks, 31% at 38 weeks, and 13% at 40 weeks or more.
For these women, the risk of maternal complications was less than 5 per 1,000 ongoing pregnancies at 37 weeks. It remained low at 38 weeks (about 7/1,000). But between 38 and 39 weeks, it rose sharply, to 16/1,000. The risk remained similarly elevated at 40 weeks and beyond.
The perinatal outcomes risk was low at 37 weeks (7/1,000) and remained low at 38 and 39 weeks (7 and 9/1,000, respectively). The risk also increased sharply between 38 and 39 weeks, rising to about 20/1,000. The maternal and perinatal risk curves intersected between weeks 37 and 38, indicating that 38 weeks was probably the optimal time for delivery.
Less than half of women who had at least three prior cesareans also made it to 39 weeks (41%). About a third (35%) delivered at 38 weeks, 15% at 37 weeks, and 9% at 40 weeks or more.
Again, maternal risk was very low at 37 weeks (about 4/1,000 ongoing pregnancies). But in contrast to the risk for women with two prior deliveries, the risk for these women increased sharply and linearly over the final weeks of gestation. By 38 weeks, it had jumped to about 16/1,000. By 39 weeks, it was close to 50/1,000, and by 40 weeks and beyond, more than 50/1,000.
Conversely, perinatal risk remained fairly low in weeks 37, 38, and 39 (9, 7, and 10/1,000, respectively). Risk accelerated between weeks 39 and 40; by week 40 and beyond, it hovered just above 25/1,000. The risk curves intersected between weeks 37 and 38, indicating that 37 weeks is the optimal time for delivery.
Dr. Hart added that maternal transfusion was the main risk driver for women, followed closely by operative injury and hysterectomy.
These data were published in a supplement to the American Journal of Obstetrics and Gynecology (Amer. J. Obstet. Gynecol. 2014;210(suppl.):S27).
Dr. Hart had no financial disclosures.
NEW ORLEANS – The safest time to deliver a woman with prior cesarean sections may be up to 2 weeks earlier than the current recommendation.
Guidelines from a number of national groups, including the Society for Maternal-Fetal Medicine, recommend planning a cesarean delivery at 39 weeks for these women. But a retrospective study has determined that maternal risk increases sharply after 37 weeks for women who have had three or more cesarean deliveries – a full 2 weeks before there is any increase in fetal risk.
"The data clearly show that the optimal time for delivery in women with three or more prior cesareans is 37 weeks," Dr. Laura Hart said at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
Dr. Hart of the University of Texas Health Science Center, Houston, extracted her data from the Maternal-Fetal Medicine Units (MFMU) Cesarean Section Registry. It contains prospective data on more than 24,000 deliveries performed after prior cesarean. She examined information on 6,435 deliveries that followed at least two prior cesareans (80% after two sections and 20% after three or more).
Dr. Hart analyzed both maternal and fetal outcomes. Each was a single composite comprising several endpoints. The maternal composite took into account transfusion, hysterectomy, operative injury, thromboembolic event, pulmonary edema, and death. The perinatal composite included respiratory distress syndrome, necrotizing enterocolitis (grade 2 or 3), intraventricular hemorrhage (grade 3 or 4), hypoxic ischemic injury, seizure, and stillbirth or neonatal death.
The results were expressed as a risk of the composite event relative to the number of ongoing pregnancies.
Less than half of the women who had two prior cesareans delivered at 39 weeks (46%); 11% delivered at 37 weeks, 31% at 38 weeks, and 13% at 40 weeks or more.
For these women, the risk of maternal complications was less than 5 per 1,000 ongoing pregnancies at 37 weeks. It remained low at 38 weeks (about 7/1,000). But between 38 and 39 weeks, it rose sharply, to 16/1,000. The risk remained similarly elevated at 40 weeks and beyond.
The perinatal outcomes risk was low at 37 weeks (7/1,000) and remained low at 38 and 39 weeks (7 and 9/1,000, respectively). The risk also increased sharply between 38 and 39 weeks, rising to about 20/1,000. The maternal and perinatal risk curves intersected between weeks 37 and 38, indicating that 38 weeks was probably the optimal time for delivery.
Less than half of women who had at least three prior cesareans also made it to 39 weeks (41%). About a third (35%) delivered at 38 weeks, 15% at 37 weeks, and 9% at 40 weeks or more.
Again, maternal risk was very low at 37 weeks (about 4/1,000 ongoing pregnancies). But in contrast to the risk for women with two prior deliveries, the risk for these women increased sharply and linearly over the final weeks of gestation. By 38 weeks, it had jumped to about 16/1,000. By 39 weeks, it was close to 50/1,000, and by 40 weeks and beyond, more than 50/1,000.
Conversely, perinatal risk remained fairly low in weeks 37, 38, and 39 (9, 7, and 10/1,000, respectively). Risk accelerated between weeks 39 and 40; by week 40 and beyond, it hovered just above 25/1,000. The risk curves intersected between weeks 37 and 38, indicating that 37 weeks is the optimal time for delivery.
Dr. Hart added that maternal transfusion was the main risk driver for women, followed closely by operative injury and hysterectomy.
These data were published in a supplement to the American Journal of Obstetrics and Gynecology (Amer. J. Obstet. Gynecol. 2014;210(suppl.):S27).
Dr. Hart had no financial disclosures.
AT THE PREGNANCY MEETING
Major finding: The optimal time for delivery of women with two prior cesarean sections is 38 weeks; for women with three or more prior sections, it is 37 weeks.
Data source: The retrospective analysis comprised 6,435 women.
Disclosures: Dr. Laura Hart had no financial disclosures.
Preterm birth prevention – let’s get the bugs out
Screening for and treating bacterial vaginosis is a routine part of preterm birth prevention. A quick course of antibiotics for women with proven infection is easy and inexpensive – and if it works, a bullet with a huge bang for the buck.
Unfortunately, it just doesn’t seem to work.
A randomized, double-blind, placebo controlled trial presented Feb. 6 at the annual Pregnancy Meeting sponsored by the Society of Maternal Fetal Medicine, found that clindamycin conferred no benefit on women with bacterial vaginosis (BV)who were at high risk for preterm birth.
The trial, held from May 2006 through June 2011, randomized 2,869 pregnant women at low risk for preterm birth to receive either one of two oral clindamycin regimens or placebo before 15 weeks’ gestation.
The study was complicated by the unexpectedly low preterm birth rate in the group that wasn’t treated, Dr. Gilles Brabant said at the meeting.
The study design assumed a 2% rate in the treated group and 4% rate in the nontreated group. The actual rate was extremely low and almost identical (1.2% in the treatment groups vs. 1% in the comparator group).
However, the power of calculation held in the final analysis, leading Dr. Brabant, of the University Hospital of Lille, France, to question just why the antibiotic wasn’t improving outcomes.
"We to have to ask ourselves, is BV the guilty party or might it be something else? We need to look further – to genetics, inflammatory response, and molecular biology – and perhaps even the microbiome."
Dr. Michal Elovitz said that she couldn’t agree more. In fact, during her presentation she said that it’s time to rethink the whole pathological paradigm of preterm birth.
The current concept starts with bacterial colonization that ascends to the cervix from the vagina, instigating an inflammatory response. The bacteria then propagate in the placenta, provoking another inflammatory response in the decidua and uterus. From there, they travel through the umbilical cord, getting the baby into the inflammation act.
"All of these serve to promote uterine contractions and degradation of the fetal membrane," said Dr. Elovitz of the University of Pennsylvania, Philadelphia. "In this model, contractions are the primary event leading to delivery – cervical remodeling is a secondary event."
But this model doesn’t explain why antibiotic trials fail to demonstrate any effect on preterm birth. In the face of these repeat failures, Dr. Elovitz said, "We need to ask if bacterial ascension into the uterus is a necessary pathogenesis for preterm birth."
She’s not letting bacteria off the hook, but she is suggesting that they may affect pregnancy outcomes in a much different way – through the collective influence of their communities at the cervicovaginal junction.
Dr. Elovitz’s research has identified four distinct communities that can populate the cervicovaginal space. Three of these are dominated by different species of lactobacilli – a species never before associated with preterm birth. The fourth lacks lactobacilli; it’s largely composed of the anaerobes that have typically been associated with BV.
She said she thinks it’s time for a new pathogenic model for preterm birth.
"We propose that the microbiota – specifically a dysbiotic community in the cervicovaginal space – may induce cervical remodeling through the interaction of the microbiota, the cervical-epithelial barrier, and the immune system," she said.
She investigated this by looking at 77 cervicovaginal samples*. Of these, 21 belonged to women who went on to have a preterm birth. The samples were obtained at 20-24 weeks and 24-28 weeks’ gestation, and genotyped to discover the exact nature of all their bacterial inhabitants. She found 168 phylotypes, and went on to focus on the 50 that were most frequently present in all samples.
The women who had term and preterm deliveries were demographically similar, except that in the first gestational period, cervical length was already significantly shorter in women destined for a preterm delivery – a hint that something was going on months before any symptoms developed.
It turns out that a single species of lactobacillus – L. iners – dominated the samples of almost all of the women who eventually had a preterm delivery. Women who had term pregnancies were significantly less likely to have that group. In fact, their samples were about evenly split between groups headed by L. crispatus and the Lactobacilli-absent anaerobes.
Lactobacilli generally aren’t thought of as bad guys, Dr. Elovitz said. But emerging data suggest that some strains aren’t so mild-mannered. "We know from nonpregnant data that there are different strains that are either pro- or negative immune mediators," which may play into the preterm birth puzzle.
"Lactobacillus also has mutations that can produce a much less favorable mucin, and if you have those mutations you are much more prone to acquire sexually transmitted diseases and HIV infections."
The good news is that while antibiotics may not be the key to this puzzle, the microbiome can be modified. For example, a plant-based diet has been found to change the microbiota associated with inflammatory bowel disease.
It’s not clear yet if, or how, the cervicovaginal communities could be modulated to protect against preterm birth. But it is clear that the well-trod path of antibiotics just isn’t getting the results researchers, doctors, mothers, and babies need.
Neither Dr. Brabant nor Dr. Elovitz had any financial declarations.
*Correction, 2/7/2014: An earlier version of this story incorrectly stated the number of cervicovaginal samples.
Screening for and treating bacterial vaginosis is a routine part of preterm birth prevention. A quick course of antibiotics for women with proven infection is easy and inexpensive – and if it works, a bullet with a huge bang for the buck.
Unfortunately, it just doesn’t seem to work.
A randomized, double-blind, placebo controlled trial presented Feb. 6 at the annual Pregnancy Meeting sponsored by the Society of Maternal Fetal Medicine, found that clindamycin conferred no benefit on women with bacterial vaginosis (BV)who were at high risk for preterm birth.
The trial, held from May 2006 through June 2011, randomized 2,869 pregnant women at low risk for preterm birth to receive either one of two oral clindamycin regimens or placebo before 15 weeks’ gestation.
The study was complicated by the unexpectedly low preterm birth rate in the group that wasn’t treated, Dr. Gilles Brabant said at the meeting.
The study design assumed a 2% rate in the treated group and 4% rate in the nontreated group. The actual rate was extremely low and almost identical (1.2% in the treatment groups vs. 1% in the comparator group).
However, the power of calculation held in the final analysis, leading Dr. Brabant, of the University Hospital of Lille, France, to question just why the antibiotic wasn’t improving outcomes.
"We to have to ask ourselves, is BV the guilty party or might it be something else? We need to look further – to genetics, inflammatory response, and molecular biology – and perhaps even the microbiome."
Dr. Michal Elovitz said that she couldn’t agree more. In fact, during her presentation she said that it’s time to rethink the whole pathological paradigm of preterm birth.
The current concept starts with bacterial colonization that ascends to the cervix from the vagina, instigating an inflammatory response. The bacteria then propagate in the placenta, provoking another inflammatory response in the decidua and uterus. From there, they travel through the umbilical cord, getting the baby into the inflammation act.
"All of these serve to promote uterine contractions and degradation of the fetal membrane," said Dr. Elovitz of the University of Pennsylvania, Philadelphia. "In this model, contractions are the primary event leading to delivery – cervical remodeling is a secondary event."
But this model doesn’t explain why antibiotic trials fail to demonstrate any effect on preterm birth. In the face of these repeat failures, Dr. Elovitz said, "We need to ask if bacterial ascension into the uterus is a necessary pathogenesis for preterm birth."
She’s not letting bacteria off the hook, but she is suggesting that they may affect pregnancy outcomes in a much different way – through the collective influence of their communities at the cervicovaginal junction.
Dr. Elovitz’s research has identified four distinct communities that can populate the cervicovaginal space. Three of these are dominated by different species of lactobacilli – a species never before associated with preterm birth. The fourth lacks lactobacilli; it’s largely composed of the anaerobes that have typically been associated with BV.
She said she thinks it’s time for a new pathogenic model for preterm birth.
"We propose that the microbiota – specifically a dysbiotic community in the cervicovaginal space – may induce cervical remodeling through the interaction of the microbiota, the cervical-epithelial barrier, and the immune system," she said.
She investigated this by looking at 77 cervicovaginal samples*. Of these, 21 belonged to women who went on to have a preterm birth. The samples were obtained at 20-24 weeks and 24-28 weeks’ gestation, and genotyped to discover the exact nature of all their bacterial inhabitants. She found 168 phylotypes, and went on to focus on the 50 that were most frequently present in all samples.
The women who had term and preterm deliveries were demographically similar, except that in the first gestational period, cervical length was already significantly shorter in women destined for a preterm delivery – a hint that something was going on months before any symptoms developed.
It turns out that a single species of lactobacillus – L. iners – dominated the samples of almost all of the women who eventually had a preterm delivery. Women who had term pregnancies were significantly less likely to have that group. In fact, their samples were about evenly split between groups headed by L. crispatus and the Lactobacilli-absent anaerobes.
Lactobacilli generally aren’t thought of as bad guys, Dr. Elovitz said. But emerging data suggest that some strains aren’t so mild-mannered. "We know from nonpregnant data that there are different strains that are either pro- or negative immune mediators," which may play into the preterm birth puzzle.
"Lactobacillus also has mutations that can produce a much less favorable mucin, and if you have those mutations you are much more prone to acquire sexually transmitted diseases and HIV infections."
The good news is that while antibiotics may not be the key to this puzzle, the microbiome can be modified. For example, a plant-based diet has been found to change the microbiota associated with inflammatory bowel disease.
It’s not clear yet if, or how, the cervicovaginal communities could be modulated to protect against preterm birth. But it is clear that the well-trod path of antibiotics just isn’t getting the results researchers, doctors, mothers, and babies need.
Neither Dr. Brabant nor Dr. Elovitz had any financial declarations.
*Correction, 2/7/2014: An earlier version of this story incorrectly stated the number of cervicovaginal samples.
Screening for and treating bacterial vaginosis is a routine part of preterm birth prevention. A quick course of antibiotics for women with proven infection is easy and inexpensive – and if it works, a bullet with a huge bang for the buck.
Unfortunately, it just doesn’t seem to work.
A randomized, double-blind, placebo controlled trial presented Feb. 6 at the annual Pregnancy Meeting sponsored by the Society of Maternal Fetal Medicine, found that clindamycin conferred no benefit on women with bacterial vaginosis (BV)who were at high risk for preterm birth.
The trial, held from May 2006 through June 2011, randomized 2,869 pregnant women at low risk for preterm birth to receive either one of two oral clindamycin regimens or placebo before 15 weeks’ gestation.
The study was complicated by the unexpectedly low preterm birth rate in the group that wasn’t treated, Dr. Gilles Brabant said at the meeting.
The study design assumed a 2% rate in the treated group and 4% rate in the nontreated group. The actual rate was extremely low and almost identical (1.2% in the treatment groups vs. 1% in the comparator group).
However, the power of calculation held in the final analysis, leading Dr. Brabant, of the University Hospital of Lille, France, to question just why the antibiotic wasn’t improving outcomes.
"We to have to ask ourselves, is BV the guilty party or might it be something else? We need to look further – to genetics, inflammatory response, and molecular biology – and perhaps even the microbiome."
Dr. Michal Elovitz said that she couldn’t agree more. In fact, during her presentation she said that it’s time to rethink the whole pathological paradigm of preterm birth.
The current concept starts with bacterial colonization that ascends to the cervix from the vagina, instigating an inflammatory response. The bacteria then propagate in the placenta, provoking another inflammatory response in the decidua and uterus. From there, they travel through the umbilical cord, getting the baby into the inflammation act.
"All of these serve to promote uterine contractions and degradation of the fetal membrane," said Dr. Elovitz of the University of Pennsylvania, Philadelphia. "In this model, contractions are the primary event leading to delivery – cervical remodeling is a secondary event."
But this model doesn’t explain why antibiotic trials fail to demonstrate any effect on preterm birth. In the face of these repeat failures, Dr. Elovitz said, "We need to ask if bacterial ascension into the uterus is a necessary pathogenesis for preterm birth."
She’s not letting bacteria off the hook, but she is suggesting that they may affect pregnancy outcomes in a much different way – through the collective influence of their communities at the cervicovaginal junction.
Dr. Elovitz’s research has identified four distinct communities that can populate the cervicovaginal space. Three of these are dominated by different species of lactobacilli – a species never before associated with preterm birth. The fourth lacks lactobacilli; it’s largely composed of the anaerobes that have typically been associated with BV.
She said she thinks it’s time for a new pathogenic model for preterm birth.
"We propose that the microbiota – specifically a dysbiotic community in the cervicovaginal space – may induce cervical remodeling through the interaction of the microbiota, the cervical-epithelial barrier, and the immune system," she said.
She investigated this by looking at 77 cervicovaginal samples*. Of these, 21 belonged to women who went on to have a preterm birth. The samples were obtained at 20-24 weeks and 24-28 weeks’ gestation, and genotyped to discover the exact nature of all their bacterial inhabitants. She found 168 phylotypes, and went on to focus on the 50 that were most frequently present in all samples.
The women who had term and preterm deliveries were demographically similar, except that in the first gestational period, cervical length was already significantly shorter in women destined for a preterm delivery – a hint that something was going on months before any symptoms developed.
It turns out that a single species of lactobacillus – L. iners – dominated the samples of almost all of the women who eventually had a preterm delivery. Women who had term pregnancies were significantly less likely to have that group. In fact, their samples were about evenly split between groups headed by L. crispatus and the Lactobacilli-absent anaerobes.
Lactobacilli generally aren’t thought of as bad guys, Dr. Elovitz said. But emerging data suggest that some strains aren’t so mild-mannered. "We know from nonpregnant data that there are different strains that are either pro- or negative immune mediators," which may play into the preterm birth puzzle.
"Lactobacillus also has mutations that can produce a much less favorable mucin, and if you have those mutations you are much more prone to acquire sexually transmitted diseases and HIV infections."
The good news is that while antibiotics may not be the key to this puzzle, the microbiome can be modified. For example, a plant-based diet has been found to change the microbiota associated with inflammatory bowel disease.
It’s not clear yet if, or how, the cervicovaginal communities could be modulated to protect against preterm birth. But it is clear that the well-trod path of antibiotics just isn’t getting the results researchers, doctors, mothers, and babies need.
Neither Dr. Brabant nor Dr. Elovitz had any financial declarations.
*Correction, 2/7/2014: An earlier version of this story incorrectly stated the number of cervicovaginal samples.
New stroke guidelines focus on women’s risks
Newly released guidelines provide the first evidence-based recommendations for preventing stroke in women.
The document addresses the issues that uniquely increase stroke risk in women – pregnancy, hormonal therapy, contraception, and migraine – along with factors like atrial fibrillation and obesity, Dr. Cheryl Bushnell and her colleagues wrote in the February issue of Stroke.
"If you are a woman, you share many of the same risk factors for stroke with men, but your risk is also influenced by hormones, reproductive health, pregnancy, childbirth, and other sex-related factors," Dr. Bushnell noted in a press statement.
The document – created by the American Heart Association and American Stroke Association – is the first to look at these gender-specific issues, wrote Dr. Bushnell, director of the Stroke Center at Wake Forest Baptist Medical Center in Winston-Salem, N.C. (Stroke 2014 [doi:10.1161/01.str.0000442009.06663.48]).
It provides graded evidence for preventive strategies in a number of risk categories. Evidence was obtained by examining dozens of studies numbering hundreds of thousands of women. But despite the extant literature, Dr. Bushnell and her colleagues said more research needs to be conducted.
"There is a need for recognition of women’s unique, sex-specific stroke risk factors, and a risk score that includes these factors would thereby identify women at risk," they wrote. "Similarly, it is important to improve stroke awareness and provide more rigorous education to women at younger ages, including childbearing ages."
The guidelines are aimed at primary care providers, who have the biggest interface with women at a prevention level – and intended to help them forge an active partnership with patients.
"More importantly," the authors wrote, "this guideline may empower women and their families to understand their own risk and how they can minimize the chances of having a stroke."
Pregnancy
For recommendations on pregnancy outcomes and stroke related to preeclampsia, the guidelines drew on evidence from 17 studies.
For women with chronic primary or secondary hypertension, or with a history of pregnancy-related hypertension, Level A evidence supports using low-dose aspirin during the second and third trimester. Level A evidence also supports calcium supplementation to prevent preeclampsia in women with low dietary intake.
There was also a Level A recommendation to treat severe hypertension during pregnancy with safe antihypertensives (methyldopa, labetalol, and nifedipine). Level B evidence supported treating moderate hypertension. The use of atenolol, angiotensin receptor blockers, and direct renin inhibitors is contraindicated because of teratogenicity.
Because preeclampsia increases lifelong stroke risk, the guidelines also recommended evaluating these women within 1 year of giving birth, and, based on their individual and family risk factors, possibly treating them for cardiovascular risk factors.
Oral contraceptives
Four studies comprising about 800,000 women examined the risk of stroke in women using hormonal birth control.
Level A evidence did not support routine screening for prothrombotic mutations before starting oral contraception. But there was Level B evidence that oral contraceptives may be harmful in women who had risk factors, including cigarette use and prior thromboembolic events.
Menopause-related hormone therapy
Seven studies – including the Women’s Health Initiative – examined the links between stroke and hormone therapy in about 37,000 women. Two recommendations supported by Level A evidence were made.
Hormone therapy should not be used for either primary or secondary stroke prevention in postmenopausal women.
Selective estrogen receptor modulators (raloxifene, tamoxifen, and tibolone) should not be used for primary prevention of stroke.
Migraine with aura
There is scant literature examining the link between migraine with aura and stroke, although what does exist suggests that the risk may be doubled overall. The addition of another factor, like pregnancy or preeclampsia, dramatically increases the risk. But because these data are low in number, the recommendations are the same as they are for men.
Level B evidence supports smoking cessation in women with migraine and aura. Level C evidence suggests that treatments that reduce the frequency of migraine may also reduce the risk of stroke.
Obesity and metabolic syndrome
A healthy lifestyle of eating whole foods, exercise, and abstaining from tobacco has been shown to lower stroke incidence in both women and men. But subgroup analyses hint that men derive the most benefit. Women-only studies of these interventions have posted mixed results about their ability to reduce stroke in women.
The authors said much more research is necessary to target interventions that are especially beneficial for women. Until then, Level B evidence supports maintaining a lifestyle of exercise, healthy eating, no tobacco use, and moderate alcohol intake (a drink a day or less) for women who aren’t pregnant.
Atrial fibrillation
Overall, similar numbers of women and men have atrial fibrillation. But the condition becomes more common with age, and women have a longer life expectancy than do men. Therefore, the authors noted, atrial fibrillation will become more common as the population of elderly women increases.
They recommend that primary care physicians actively screen women for atrial fibrillation once they reach age 75 years. The screening method, supported by Level B evidence, should be pulse followed by an electrocardiogram.
For women aged 65 years and younger who have atrial fibrillation but no other risk factors, there is no evidence supporting oral anticoagulation. Level B evidence does support antiplatelet therapy.
Dr. Bushnell had no financial disclosures. One of the 16 coauthors reported relationships with several pharmaceutical companies
On Twitter @alz_gal
Newly released guidelines provide the first evidence-based recommendations for preventing stroke in women.
The document addresses the issues that uniquely increase stroke risk in women – pregnancy, hormonal therapy, contraception, and migraine – along with factors like atrial fibrillation and obesity, Dr. Cheryl Bushnell and her colleagues wrote in the February issue of Stroke.
"If you are a woman, you share many of the same risk factors for stroke with men, but your risk is also influenced by hormones, reproductive health, pregnancy, childbirth, and other sex-related factors," Dr. Bushnell noted in a press statement.
The document – created by the American Heart Association and American Stroke Association – is the first to look at these gender-specific issues, wrote Dr. Bushnell, director of the Stroke Center at Wake Forest Baptist Medical Center in Winston-Salem, N.C. (Stroke 2014 [doi:10.1161/01.str.0000442009.06663.48]).
It provides graded evidence for preventive strategies in a number of risk categories. Evidence was obtained by examining dozens of studies numbering hundreds of thousands of women. But despite the extant literature, Dr. Bushnell and her colleagues said more research needs to be conducted.
"There is a need for recognition of women’s unique, sex-specific stroke risk factors, and a risk score that includes these factors would thereby identify women at risk," they wrote. "Similarly, it is important to improve stroke awareness and provide more rigorous education to women at younger ages, including childbearing ages."
The guidelines are aimed at primary care providers, who have the biggest interface with women at a prevention level – and intended to help them forge an active partnership with patients.
"More importantly," the authors wrote, "this guideline may empower women and their families to understand their own risk and how they can minimize the chances of having a stroke."
Pregnancy
For recommendations on pregnancy outcomes and stroke related to preeclampsia, the guidelines drew on evidence from 17 studies.
For women with chronic primary or secondary hypertension, or with a history of pregnancy-related hypertension, Level A evidence supports using low-dose aspirin during the second and third trimester. Level A evidence also supports calcium supplementation to prevent preeclampsia in women with low dietary intake.
There was also a Level A recommendation to treat severe hypertension during pregnancy with safe antihypertensives (methyldopa, labetalol, and nifedipine). Level B evidence supported treating moderate hypertension. The use of atenolol, angiotensin receptor blockers, and direct renin inhibitors is contraindicated because of teratogenicity.
Because preeclampsia increases lifelong stroke risk, the guidelines also recommended evaluating these women within 1 year of giving birth, and, based on their individual and family risk factors, possibly treating them for cardiovascular risk factors.
Oral contraceptives
Four studies comprising about 800,000 women examined the risk of stroke in women using hormonal birth control.
Level A evidence did not support routine screening for prothrombotic mutations before starting oral contraception. But there was Level B evidence that oral contraceptives may be harmful in women who had risk factors, including cigarette use and prior thromboembolic events.
Menopause-related hormone therapy
Seven studies – including the Women’s Health Initiative – examined the links between stroke and hormone therapy in about 37,000 women. Two recommendations supported by Level A evidence were made.
Hormone therapy should not be used for either primary or secondary stroke prevention in postmenopausal women.
Selective estrogen receptor modulators (raloxifene, tamoxifen, and tibolone) should not be used for primary prevention of stroke.
Migraine with aura
There is scant literature examining the link between migraine with aura and stroke, although what does exist suggests that the risk may be doubled overall. The addition of another factor, like pregnancy or preeclampsia, dramatically increases the risk. But because these data are low in number, the recommendations are the same as they are for men.
Level B evidence supports smoking cessation in women with migraine and aura. Level C evidence suggests that treatments that reduce the frequency of migraine may also reduce the risk of stroke.
Obesity and metabolic syndrome
A healthy lifestyle of eating whole foods, exercise, and abstaining from tobacco has been shown to lower stroke incidence in both women and men. But subgroup analyses hint that men derive the most benefit. Women-only studies of these interventions have posted mixed results about their ability to reduce stroke in women.
The authors said much more research is necessary to target interventions that are especially beneficial for women. Until then, Level B evidence supports maintaining a lifestyle of exercise, healthy eating, no tobacco use, and moderate alcohol intake (a drink a day or less) for women who aren’t pregnant.
Atrial fibrillation
Overall, similar numbers of women and men have atrial fibrillation. But the condition becomes more common with age, and women have a longer life expectancy than do men. Therefore, the authors noted, atrial fibrillation will become more common as the population of elderly women increases.
They recommend that primary care physicians actively screen women for atrial fibrillation once they reach age 75 years. The screening method, supported by Level B evidence, should be pulse followed by an electrocardiogram.
For women aged 65 years and younger who have atrial fibrillation but no other risk factors, there is no evidence supporting oral anticoagulation. Level B evidence does support antiplatelet therapy.
Dr. Bushnell had no financial disclosures. One of the 16 coauthors reported relationships with several pharmaceutical companies
On Twitter @alz_gal
Newly released guidelines provide the first evidence-based recommendations for preventing stroke in women.
The document addresses the issues that uniquely increase stroke risk in women – pregnancy, hormonal therapy, contraception, and migraine – along with factors like atrial fibrillation and obesity, Dr. Cheryl Bushnell and her colleagues wrote in the February issue of Stroke.
"If you are a woman, you share many of the same risk factors for stroke with men, but your risk is also influenced by hormones, reproductive health, pregnancy, childbirth, and other sex-related factors," Dr. Bushnell noted in a press statement.
The document – created by the American Heart Association and American Stroke Association – is the first to look at these gender-specific issues, wrote Dr. Bushnell, director of the Stroke Center at Wake Forest Baptist Medical Center in Winston-Salem, N.C. (Stroke 2014 [doi:10.1161/01.str.0000442009.06663.48]).
It provides graded evidence for preventive strategies in a number of risk categories. Evidence was obtained by examining dozens of studies numbering hundreds of thousands of women. But despite the extant literature, Dr. Bushnell and her colleagues said more research needs to be conducted.
"There is a need for recognition of women’s unique, sex-specific stroke risk factors, and a risk score that includes these factors would thereby identify women at risk," they wrote. "Similarly, it is important to improve stroke awareness and provide more rigorous education to women at younger ages, including childbearing ages."
The guidelines are aimed at primary care providers, who have the biggest interface with women at a prevention level – and intended to help them forge an active partnership with patients.
"More importantly," the authors wrote, "this guideline may empower women and their families to understand their own risk and how they can minimize the chances of having a stroke."
Pregnancy
For recommendations on pregnancy outcomes and stroke related to preeclampsia, the guidelines drew on evidence from 17 studies.
For women with chronic primary or secondary hypertension, or with a history of pregnancy-related hypertension, Level A evidence supports using low-dose aspirin during the second and third trimester. Level A evidence also supports calcium supplementation to prevent preeclampsia in women with low dietary intake.
There was also a Level A recommendation to treat severe hypertension during pregnancy with safe antihypertensives (methyldopa, labetalol, and nifedipine). Level B evidence supported treating moderate hypertension. The use of atenolol, angiotensin receptor blockers, and direct renin inhibitors is contraindicated because of teratogenicity.
Because preeclampsia increases lifelong stroke risk, the guidelines also recommended evaluating these women within 1 year of giving birth, and, based on their individual and family risk factors, possibly treating them for cardiovascular risk factors.
Oral contraceptives
Four studies comprising about 800,000 women examined the risk of stroke in women using hormonal birth control.
Level A evidence did not support routine screening for prothrombotic mutations before starting oral contraception. But there was Level B evidence that oral contraceptives may be harmful in women who had risk factors, including cigarette use and prior thromboembolic events.
Menopause-related hormone therapy
Seven studies – including the Women’s Health Initiative – examined the links between stroke and hormone therapy in about 37,000 women. Two recommendations supported by Level A evidence were made.
Hormone therapy should not be used for either primary or secondary stroke prevention in postmenopausal women.
Selective estrogen receptor modulators (raloxifene, tamoxifen, and tibolone) should not be used for primary prevention of stroke.
Migraine with aura
There is scant literature examining the link between migraine with aura and stroke, although what does exist suggests that the risk may be doubled overall. The addition of another factor, like pregnancy or preeclampsia, dramatically increases the risk. But because these data are low in number, the recommendations are the same as they are for men.
Level B evidence supports smoking cessation in women with migraine and aura. Level C evidence suggests that treatments that reduce the frequency of migraine may also reduce the risk of stroke.
Obesity and metabolic syndrome
A healthy lifestyle of eating whole foods, exercise, and abstaining from tobacco has been shown to lower stroke incidence in both women and men. But subgroup analyses hint that men derive the most benefit. Women-only studies of these interventions have posted mixed results about their ability to reduce stroke in women.
The authors said much more research is necessary to target interventions that are especially beneficial for women. Until then, Level B evidence supports maintaining a lifestyle of exercise, healthy eating, no tobacco use, and moderate alcohol intake (a drink a day or less) for women who aren’t pregnant.
Atrial fibrillation
Overall, similar numbers of women and men have atrial fibrillation. But the condition becomes more common with age, and women have a longer life expectancy than do men. Therefore, the authors noted, atrial fibrillation will become more common as the population of elderly women increases.
They recommend that primary care physicians actively screen women for atrial fibrillation once they reach age 75 years. The screening method, supported by Level B evidence, should be pulse followed by an electrocardiogram.
For women aged 65 years and younger who have atrial fibrillation but no other risk factors, there is no evidence supporting oral anticoagulation. Level B evidence does support antiplatelet therapy.
Dr. Bushnell had no financial disclosures. One of the 16 coauthors reported relationships with several pharmaceutical companies
On Twitter @alz_gal
FROM STROKE
2014 adult vaccine schedule includes recombinant flu vaccine
The recombinant influenza vaccine is now recommended as an option for adults with egg allergies, according to the 2014 recommended vaccine schedule for adults.
Approved last year, the recombinant influenza vaccine (RIV) is manufactured with the baculovirus. It contains no egg protein, and can be safely administered to those who have an egg allergy of any severity, according to the schedule published Feb. 3 (Ann. Int. Med. 2014;160:190-8).
RIV is a good addition to the schedule, Dr. Jonathan Temte, chair of the Centers for Disease Control and Prevention’s Advisory Committee of Immunization Practices (ACIP), said in an interview.
This provides an option for people who have a serious egg allergy. Many people with egg allergies have only mild allergies – hives – and can receive influenza vaccine," Dr. Temte said.
Other updates to the schedule include the following:
• Adults who have undergone successful hematopoietic stem cell transplant should get a three-dose series of Haemophilus influenzae type B vaccine (Hib) 6-12 months after the procedure, regardless of prior vaccination. Hib is no longer recommended for previously unvaccinated HIV-infected adults, because their risk of infection is low.
• Zoster and human papillomavirus vaccines (either HPV2 or HPV4) are no longer specifically indicated for health care workers; instead, each should be provided to health care professionals and other patients who meet age and other indications.
• Previously unvaccinated patients 11 years old or older should have a single dose of tetanus, diphtheria, and acellular pertussis (Tdap), with a tetanus, diphtheria (Td) booster every 10 years. Pregnant women should get a dose of Tdap during each pregnancy (preferably during 27-36 weeks’ gestation) regardless of when they last received Tdap or Td.
• Timing of the HPV vaccine series was clarified. Both HPV vaccines have a recommended three-dose series, with the second dose administered 4-8 weeks after the first and the third dose administered at least 12 weeks after the second.
• Recommendations on the meningococcal vaccine clarify which patients need either one or two doses as well as who should receive the meningococcal conjugate (MenACWY-D) and who should have the meningococcal polysaccharide (MenACWY-CRM) quadrivalent vaccines.
You can view the 2014 schedule at our Resources section here.
The recombinant influenza vaccine is now recommended as an option for adults with egg allergies, according to the 2014 recommended vaccine schedule for adults.
Approved last year, the recombinant influenza vaccine (RIV) is manufactured with the baculovirus. It contains no egg protein, and can be safely administered to those who have an egg allergy of any severity, according to the schedule published Feb. 3 (Ann. Int. Med. 2014;160:190-8).
RIV is a good addition to the schedule, Dr. Jonathan Temte, chair of the Centers for Disease Control and Prevention’s Advisory Committee of Immunization Practices (ACIP), said in an interview.
This provides an option for people who have a serious egg allergy. Many people with egg allergies have only mild allergies – hives – and can receive influenza vaccine," Dr. Temte said.
Other updates to the schedule include the following:
• Adults who have undergone successful hematopoietic stem cell transplant should get a three-dose series of Haemophilus influenzae type B vaccine (Hib) 6-12 months after the procedure, regardless of prior vaccination. Hib is no longer recommended for previously unvaccinated HIV-infected adults, because their risk of infection is low.
• Zoster and human papillomavirus vaccines (either HPV2 or HPV4) are no longer specifically indicated for health care workers; instead, each should be provided to health care professionals and other patients who meet age and other indications.
• Previously unvaccinated patients 11 years old or older should have a single dose of tetanus, diphtheria, and acellular pertussis (Tdap), with a tetanus, diphtheria (Td) booster every 10 years. Pregnant women should get a dose of Tdap during each pregnancy (preferably during 27-36 weeks’ gestation) regardless of when they last received Tdap or Td.
• Timing of the HPV vaccine series was clarified. Both HPV vaccines have a recommended three-dose series, with the second dose administered 4-8 weeks after the first and the third dose administered at least 12 weeks after the second.
• Recommendations on the meningococcal vaccine clarify which patients need either one or two doses as well as who should receive the meningococcal conjugate (MenACWY-D) and who should have the meningococcal polysaccharide (MenACWY-CRM) quadrivalent vaccines.
You can view the 2014 schedule at our Resources section here.
The recombinant influenza vaccine is now recommended as an option for adults with egg allergies, according to the 2014 recommended vaccine schedule for adults.
Approved last year, the recombinant influenza vaccine (RIV) is manufactured with the baculovirus. It contains no egg protein, and can be safely administered to those who have an egg allergy of any severity, according to the schedule published Feb. 3 (Ann. Int. Med. 2014;160:190-8).
RIV is a good addition to the schedule, Dr. Jonathan Temte, chair of the Centers for Disease Control and Prevention’s Advisory Committee of Immunization Practices (ACIP), said in an interview.
This provides an option for people who have a serious egg allergy. Many people with egg allergies have only mild allergies – hives – and can receive influenza vaccine," Dr. Temte said.
Other updates to the schedule include the following:
• Adults who have undergone successful hematopoietic stem cell transplant should get a three-dose series of Haemophilus influenzae type B vaccine (Hib) 6-12 months after the procedure, regardless of prior vaccination. Hib is no longer recommended for previously unvaccinated HIV-infected adults, because their risk of infection is low.
• Zoster and human papillomavirus vaccines (either HPV2 or HPV4) are no longer specifically indicated for health care workers; instead, each should be provided to health care professionals and other patients who meet age and other indications.
• Previously unvaccinated patients 11 years old or older should have a single dose of tetanus, diphtheria, and acellular pertussis (Tdap), with a tetanus, diphtheria (Td) booster every 10 years. Pregnant women should get a dose of Tdap during each pregnancy (preferably during 27-36 weeks’ gestation) regardless of when they last received Tdap or Td.
• Timing of the HPV vaccine series was clarified. Both HPV vaccines have a recommended three-dose series, with the second dose administered 4-8 weeks after the first and the third dose administered at least 12 weeks after the second.
• Recommendations on the meningococcal vaccine clarify which patients need either one or two doses as well as who should receive the meningococcal conjugate (MenACWY-D) and who should have the meningococcal polysaccharide (MenACWY-CRM) quadrivalent vaccines.
You can view the 2014 schedule at our Resources section here.
FROM ANNALS OF INTERNAL MEDICINE
2014 adult vaccine schedule includes recombinant flu vaccine
The recombinant influenza vaccine is now recommended as an option for adults with egg allergies, according to the 2014 recommended vaccine schedule for adults.
Approved last year, the recombinant influenza vaccine (RIV) is manufactured with the baculovirus. It contains no egg protein, and can be safely administered to those who have an egg allergy of any severity, according to the schedule published Feb. 3 (Ann. Int. Med. 2014;160:190-8).
RIV is a good addition to the schedule, Dr. Jonathan Temte, chair of the Centers for Disease Control and Prevention’s Advisory Committee of Immunization Practices (ACIP), said in an interview.
This provides an option for people who have a serious egg allergy. Many people with egg allergies have only mild allergies – hives – and can receive influenza vaccine," Dr. Temte said.
Other updates to the schedule include the following:
• Adults who have undergone successful hematopoietic stem cell transplant should get a three-dose series of Haemophilus influenzae type B vaccine (Hib) 6-12 months after the procedure, regardless of prior vaccination. Hib is no longer recommended for previously unvaccinated HIV-infected adults, because their risk of infection is low.
• Zoster and human papillomavirus vaccines (either HPV2 or HPV4) are no longer specifically indicated for health care workers; instead, each should be provided to health care professionals and other patients who meet age and other indications.
• Previously unvaccinated patients 11 years old or older should have a single dose of tetanus, diphtheria, and acellular pertussis (Tdap), with a tetanus, diphtheria (Td) booster every 10 years. Pregnant women should get a dose of Tdap during each pregnancy (preferably during 27-36 weeks’ gestation) regardless of when they last received Tdap or Td.
• Timing of the HPV vaccine series was clarified. Both HPV vaccines have a recommended three-dose series, with the second dose administered 4-8 weeks after the first and the third dose administered at least 12 weeks after the second.
• Recommendations on the meningococcal vaccine clarify which patients need either one or two doses as well as who should receive the meningococcal conjugate (MenACWY-D) and who should have the meningococcal polysaccharide (MenACWY-CRM) quadrivalent vaccines.
You can view the 2014 schedule at our Resources section here.
The recombinant influenza vaccine is now recommended as an option for adults with egg allergies, according to the 2014 recommended vaccine schedule for adults.
Approved last year, the recombinant influenza vaccine (RIV) is manufactured with the baculovirus. It contains no egg protein, and can be safely administered to those who have an egg allergy of any severity, according to the schedule published Feb. 3 (Ann. Int. Med. 2014;160:190-8).
RIV is a good addition to the schedule, Dr. Jonathan Temte, chair of the Centers for Disease Control and Prevention’s Advisory Committee of Immunization Practices (ACIP), said in an interview.
This provides an option for people who have a serious egg allergy. Many people with egg allergies have only mild allergies – hives – and can receive influenza vaccine," Dr. Temte said.
Other updates to the schedule include the following:
• Adults who have undergone successful hematopoietic stem cell transplant should get a three-dose series of Haemophilus influenzae type B vaccine (Hib) 6-12 months after the procedure, regardless of prior vaccination. Hib is no longer recommended for previously unvaccinated HIV-infected adults, because their risk of infection is low.
• Zoster and human papillomavirus vaccines (either HPV2 or HPV4) are no longer specifically indicated for health care workers; instead, each should be provided to health care professionals and other patients who meet age and other indications.
• Previously unvaccinated patients 11 years old or older should have a single dose of tetanus, diphtheria, and acellular pertussis (Tdap), with a tetanus, diphtheria (Td) booster every 10 years. Pregnant women should get a dose of Tdap during each pregnancy (preferably during 27-36 weeks’ gestation) regardless of when they last received Tdap or Td.
• Timing of the HPV vaccine series was clarified. Both HPV vaccines have a recommended three-dose series, with the second dose administered 4-8 weeks after the first and the third dose administered at least 12 weeks after the second.
• Recommendations on the meningococcal vaccine clarify which patients need either one or two doses as well as who should receive the meningococcal conjugate (MenACWY-D) and who should have the meningococcal polysaccharide (MenACWY-CRM) quadrivalent vaccines.
You can view the 2014 schedule at our Resources section here.
The recombinant influenza vaccine is now recommended as an option for adults with egg allergies, according to the 2014 recommended vaccine schedule for adults.
Approved last year, the recombinant influenza vaccine (RIV) is manufactured with the baculovirus. It contains no egg protein, and can be safely administered to those who have an egg allergy of any severity, according to the schedule published Feb. 3 (Ann. Int. Med. 2014;160:190-8).
RIV is a good addition to the schedule, Dr. Jonathan Temte, chair of the Centers for Disease Control and Prevention’s Advisory Committee of Immunization Practices (ACIP), said in an interview.
This provides an option for people who have a serious egg allergy. Many people with egg allergies have only mild allergies – hives – and can receive influenza vaccine," Dr. Temte said.
Other updates to the schedule include the following:
• Adults who have undergone successful hematopoietic stem cell transplant should get a three-dose series of Haemophilus influenzae type B vaccine (Hib) 6-12 months after the procedure, regardless of prior vaccination. Hib is no longer recommended for previously unvaccinated HIV-infected adults, because their risk of infection is low.
• Zoster and human papillomavirus vaccines (either HPV2 or HPV4) are no longer specifically indicated for health care workers; instead, each should be provided to health care professionals and other patients who meet age and other indications.
• Previously unvaccinated patients 11 years old or older should have a single dose of tetanus, diphtheria, and acellular pertussis (Tdap), with a tetanus, diphtheria (Td) booster every 10 years. Pregnant women should get a dose of Tdap during each pregnancy (preferably during 27-36 weeks’ gestation) regardless of when they last received Tdap or Td.
• Timing of the HPV vaccine series was clarified. Both HPV vaccines have a recommended three-dose series, with the second dose administered 4-8 weeks after the first and the third dose administered at least 12 weeks after the second.
• Recommendations on the meningococcal vaccine clarify which patients need either one or two doses as well as who should receive the meningococcal conjugate (MenACWY-D) and who should have the meningococcal polysaccharide (MenACWY-CRM) quadrivalent vaccines.
You can view the 2014 schedule at our Resources section here.
FROM ANNALS OF INTERNAL MEDICINE
Physicians want to vaccinate, but money gets in the way
While almost all internists and family physicians feel a responsibility to ask about patients’ immunization status at least annually, most refer them to another source for all but basic seasonal vaccines.
A national survey determined that less than a third of these primary care providers stocked all 11 of the recommended adult vaccines. Financial issues were the biggest barrier to keeping all of the vaccines on hand and administering them, Dr. Laura Hurley and her colleagues wrote in the February issue of Annals of Internal Medicine (Ann. Int. Med. 2014;160:161-70).
"Although primary care physicians appear motivated to ensure that patients are up to date on vaccinations, many barriers exist. Implementation of system changes, including adopting practices that improve communication between primary care physicians and alternate vaccinators, more widespread use of effective tools ... and removing policy-related barriers, could improve adult vaccination in the United States," wrote Dr. Hurley of the Wellington E. Webb Center for Primary Care, Denver, and her coauthors.
Conducted by the Vaccine Policy Collaborative Initiative (VPCI), the survey assessed how responsible primary care physicians feel about adult vaccination, how often they assess and provide it, and what the barriers are to how vaccination is handled. The survey was mailed to 443 general internists and 409 family physicians, of whom 352 and 255 physicians, respectively, responded.
Ninety-seven percent of both groups said they assessed vaccination status at annual visits, but only about a third reported doing so at each visit.
Automated systems were fairly low down on the list of assessment techniques, although more family doctors than internists reported using them (36% vs. 8%). About half of all the respondents said it was moderately to very difficult to get up-to-date information on anything but the seasonal influenza vaccine.
In addition to the flu vaccine, most physicians checked on the status of pneumococcal; tetanus, diphtheria, and pertussis (Td and Tdap); and shingles vaccines. Family doctors were more likely to check on hepatitis A and B; MMR; and varicella vaccines.
Most physicians reported stocking influenza, pneumococcal, Td, and Tdap vaccines. Family physicians were more likely to stock both hepatitis vaccines, as well as those for human papillomavirus, varicella, meningitis, and shingles. Thirty-one percent of family physicians and 20% of internists said they routinely stocked all 11 of the recommended adult vaccines.
Money issues were the biggest barriers to stocking and administering vaccines, the respondents said. Commonly cited problems were lack of reimbursement for purchasing and administering; lack of insurance coverage for vaccines; and the up-front cost of buying the vaccine. Some providers worried the vaccine would expire before it could all be used, and others said patients simply refused them because they couldn’t pay.
Physicians in private and small group practices reported the biggest financial barriers, as did those who served a large number of Medicare Part D patients.
But even if they couldn’t provide the service themselves, respondents were sending patients to places that could – mostly pharmacies and public health departments. Still, financial issues were the biggest reasons for referrals; lack of insurance coverage or low insurance reimbursement figured prominently.
That can be a tricky proposition, however, said Dr. Jonathan Temte, chair of the Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices.
Section 317 of the Public Health Service Act provides federally purchased vaccine for free immunizations for qualifying patients, including uninsured adults. But underinsured adults or those who have very high insurance deductibles can’t get free immunizations with 317-purchased vaccines.
"Changes in the federal 317 rules prohibit adult patients who are insured from receiving vaccines without charge through public health agencies," Dr. Temte said in an interview. "Accordingly, the practice of ‘referring adults to public health’ for immunization doesn’t work too well."
When they did send patients to another source, most respondents were able to follow up on vaccination status. The most common way was to communicate with the vaccination provider. About a third of the two groups used an immunization information system. The information was usually recorded in the patient’s electronic health record or in a written chart or vaccine record.
"Of note," the authors said, "almost all physicians agreed that it was the primary care physician’s responsibility to see that patients receive recommended vaccines, even if they did so elsewhere."
The CDC sponsored the survey; none of the authors had any financial disclosures.
While almost all internists and family physicians feel a responsibility to ask about patients’ immunization status at least annually, most refer them to another source for all but basic seasonal vaccines.
A national survey determined that less than a third of these primary care providers stocked all 11 of the recommended adult vaccines. Financial issues were the biggest barrier to keeping all of the vaccines on hand and administering them, Dr. Laura Hurley and her colleagues wrote in the February issue of Annals of Internal Medicine (Ann. Int. Med. 2014;160:161-70).
"Although primary care physicians appear motivated to ensure that patients are up to date on vaccinations, many barriers exist. Implementation of system changes, including adopting practices that improve communication between primary care physicians and alternate vaccinators, more widespread use of effective tools ... and removing policy-related barriers, could improve adult vaccination in the United States," wrote Dr. Hurley of the Wellington E. Webb Center for Primary Care, Denver, and her coauthors.
Conducted by the Vaccine Policy Collaborative Initiative (VPCI), the survey assessed how responsible primary care physicians feel about adult vaccination, how often they assess and provide it, and what the barriers are to how vaccination is handled. The survey was mailed to 443 general internists and 409 family physicians, of whom 352 and 255 physicians, respectively, responded.
Ninety-seven percent of both groups said they assessed vaccination status at annual visits, but only about a third reported doing so at each visit.
Automated systems were fairly low down on the list of assessment techniques, although more family doctors than internists reported using them (36% vs. 8%). About half of all the respondents said it was moderately to very difficult to get up-to-date information on anything but the seasonal influenza vaccine.
In addition to the flu vaccine, most physicians checked on the status of pneumococcal; tetanus, diphtheria, and pertussis (Td and Tdap); and shingles vaccines. Family doctors were more likely to check on hepatitis A and B; MMR; and varicella vaccines.
Most physicians reported stocking influenza, pneumococcal, Td, and Tdap vaccines. Family physicians were more likely to stock both hepatitis vaccines, as well as those for human papillomavirus, varicella, meningitis, and shingles. Thirty-one percent of family physicians and 20% of internists said they routinely stocked all 11 of the recommended adult vaccines.
Money issues were the biggest barriers to stocking and administering vaccines, the respondents said. Commonly cited problems were lack of reimbursement for purchasing and administering; lack of insurance coverage for vaccines; and the up-front cost of buying the vaccine. Some providers worried the vaccine would expire before it could all be used, and others said patients simply refused them because they couldn’t pay.
Physicians in private and small group practices reported the biggest financial barriers, as did those who served a large number of Medicare Part D patients.
But even if they couldn’t provide the service themselves, respondents were sending patients to places that could – mostly pharmacies and public health departments. Still, financial issues were the biggest reasons for referrals; lack of insurance coverage or low insurance reimbursement figured prominently.
That can be a tricky proposition, however, said Dr. Jonathan Temte, chair of the Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices.
Section 317 of the Public Health Service Act provides federally purchased vaccine for free immunizations for qualifying patients, including uninsured adults. But underinsured adults or those who have very high insurance deductibles can’t get free immunizations with 317-purchased vaccines.
"Changes in the federal 317 rules prohibit adult patients who are insured from receiving vaccines without charge through public health agencies," Dr. Temte said in an interview. "Accordingly, the practice of ‘referring adults to public health’ for immunization doesn’t work too well."
When they did send patients to another source, most respondents were able to follow up on vaccination status. The most common way was to communicate with the vaccination provider. About a third of the two groups used an immunization information system. The information was usually recorded in the patient’s electronic health record or in a written chart or vaccine record.
"Of note," the authors said, "almost all physicians agreed that it was the primary care physician’s responsibility to see that patients receive recommended vaccines, even if they did so elsewhere."
The CDC sponsored the survey; none of the authors had any financial disclosures.
While almost all internists and family physicians feel a responsibility to ask about patients’ immunization status at least annually, most refer them to another source for all but basic seasonal vaccines.
A national survey determined that less than a third of these primary care providers stocked all 11 of the recommended adult vaccines. Financial issues were the biggest barrier to keeping all of the vaccines on hand and administering them, Dr. Laura Hurley and her colleagues wrote in the February issue of Annals of Internal Medicine (Ann. Int. Med. 2014;160:161-70).
"Although primary care physicians appear motivated to ensure that patients are up to date on vaccinations, many barriers exist. Implementation of system changes, including adopting practices that improve communication between primary care physicians and alternate vaccinators, more widespread use of effective tools ... and removing policy-related barriers, could improve adult vaccination in the United States," wrote Dr. Hurley of the Wellington E. Webb Center for Primary Care, Denver, and her coauthors.
Conducted by the Vaccine Policy Collaborative Initiative (VPCI), the survey assessed how responsible primary care physicians feel about adult vaccination, how often they assess and provide it, and what the barriers are to how vaccination is handled. The survey was mailed to 443 general internists and 409 family physicians, of whom 352 and 255 physicians, respectively, responded.
Ninety-seven percent of both groups said they assessed vaccination status at annual visits, but only about a third reported doing so at each visit.
Automated systems were fairly low down on the list of assessment techniques, although more family doctors than internists reported using them (36% vs. 8%). About half of all the respondents said it was moderately to very difficult to get up-to-date information on anything but the seasonal influenza vaccine.
In addition to the flu vaccine, most physicians checked on the status of pneumococcal; tetanus, diphtheria, and pertussis (Td and Tdap); and shingles vaccines. Family doctors were more likely to check on hepatitis A and B; MMR; and varicella vaccines.
Most physicians reported stocking influenza, pneumococcal, Td, and Tdap vaccines. Family physicians were more likely to stock both hepatitis vaccines, as well as those for human papillomavirus, varicella, meningitis, and shingles. Thirty-one percent of family physicians and 20% of internists said they routinely stocked all 11 of the recommended adult vaccines.
Money issues were the biggest barriers to stocking and administering vaccines, the respondents said. Commonly cited problems were lack of reimbursement for purchasing and administering; lack of insurance coverage for vaccines; and the up-front cost of buying the vaccine. Some providers worried the vaccine would expire before it could all be used, and others said patients simply refused them because they couldn’t pay.
Physicians in private and small group practices reported the biggest financial barriers, as did those who served a large number of Medicare Part D patients.
But even if they couldn’t provide the service themselves, respondents were sending patients to places that could – mostly pharmacies and public health departments. Still, financial issues were the biggest reasons for referrals; lack of insurance coverage or low insurance reimbursement figured prominently.
That can be a tricky proposition, however, said Dr. Jonathan Temte, chair of the Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices.
Section 317 of the Public Health Service Act provides federally purchased vaccine for free immunizations for qualifying patients, including uninsured adults. But underinsured adults or those who have very high insurance deductibles can’t get free immunizations with 317-purchased vaccines.
"Changes in the federal 317 rules prohibit adult patients who are insured from receiving vaccines without charge through public health agencies," Dr. Temte said in an interview. "Accordingly, the practice of ‘referring adults to public health’ for immunization doesn’t work too well."
When they did send patients to another source, most respondents were able to follow up on vaccination status. The most common way was to communicate with the vaccination provider. About a third of the two groups used an immunization information system. The information was usually recorded in the patient’s electronic health record or in a written chart or vaccine record.
"Of note," the authors said, "almost all physicians agreed that it was the primary care physician’s responsibility to see that patients receive recommended vaccines, even if they did so elsewhere."
The CDC sponsored the survey; none of the authors had any financial disclosures.
FROM ANNALS OF INTERNAL MEDICINE
Major finding: Although about 90% of primary care providers assessed patients’ vaccination status at least once a year, less than a third stocked all 11 of the recommended adult vaccines.
Data source: The survey was mailed to 443 general internists and 409 family physicians.
Disclosures: The CDC sponsored the survey; none of the authors had any financial disclosures.
2014 adult vaccine schedule includes recombinant flu vaccine
The recombinant influenza vaccine is now recommended as an option for adults with egg allergies, according to the 2014 recommended vaccine schedule for adults.
Approved last year, the recombinant influenza vaccine (RIV) is manufactured with the baculovirus. It contains no egg protein, and can be safely administered to those who have an egg allergy of any severity, according to the schedule published Feb. 3 (Ann. Int. Med. 2014;160:190-8).
RIV is a good addition to the schedule, Dr. Jonathan Temte, chair of the Centers for Disease Control and Prevention’s Advisory Committee of Immunization Practices (ACIP), said in an interview.
This provides an option for people who have a serious egg allergy. Many people with egg allergies have only mild allergies – hives – and can receive influenza vaccine," Dr. Temte said.
Other updates to the schedule include the following:
• Adults who have undergone successful hematopoietic stem cell transplant should get a three-dose series of Haemophilus influenzae type B vaccine (Hib) 6-12 months after the procedure, regardless of prior vaccination. Hib is no longer recommended for previously unvaccinated HIV-infected adults, because their risk of infection is low.
• Zoster and human papillomavirus vaccines (either HPV2 or HPV4) are no longer specifically indicated for health care workers; instead, each should be provided to health care professionals and other patients who meet age and other indications.
• Previously unvaccinated patients 11 years old or older should have a single dose of tetanus, diphtheria, and acellular pertussis (Tdap), with a tetanus, diphtheria (Td) booster every 10 years. Pregnant women should get a dose of Tdap during each pregnancy (preferably during 27-36 weeks’ gestation) regardless of when they last received Tdap or Td.
• Timing of the HPV vaccine series was clarified. Both HPV vaccines have a recommended three-dose series, with the second dose administered 4-8 weeks after the first and the third dose administered at least 12 weeks after the second.
• Recommendations on the meningococcal vaccine clarify which patients need either one or two doses as well as who should receive the meningococcal conjugate (MenACWY-D) and who should have the meningococcal polysaccharide (MenACWY-CRM) quadrivalent vaccines.
You can view the 2014 schedule at our Resources section here.
The recombinant influenza vaccine is now recommended as an option for adults with egg allergies, according to the 2014 recommended vaccine schedule for adults.
Approved last year, the recombinant influenza vaccine (RIV) is manufactured with the baculovirus. It contains no egg protein, and can be safely administered to those who have an egg allergy of any severity, according to the schedule published Feb. 3 (Ann. Int. Med. 2014;160:190-8).
RIV is a good addition to the schedule, Dr. Jonathan Temte, chair of the Centers for Disease Control and Prevention’s Advisory Committee of Immunization Practices (ACIP), said in an interview.
This provides an option for people who have a serious egg allergy. Many people with egg allergies have only mild allergies – hives – and can receive influenza vaccine," Dr. Temte said.
Other updates to the schedule include the following:
• Adults who have undergone successful hematopoietic stem cell transplant should get a three-dose series of Haemophilus influenzae type B vaccine (Hib) 6-12 months after the procedure, regardless of prior vaccination. Hib is no longer recommended for previously unvaccinated HIV-infected adults, because their risk of infection is low.
• Zoster and human papillomavirus vaccines (either HPV2 or HPV4) are no longer specifically indicated for health care workers; instead, each should be provided to health care professionals and other patients who meet age and other indications.
• Previously unvaccinated patients 11 years old or older should have a single dose of tetanus, diphtheria, and acellular pertussis (Tdap), with a tetanus, diphtheria (Td) booster every 10 years. Pregnant women should get a dose of Tdap during each pregnancy (preferably during 27-36 weeks’ gestation) regardless of when they last received Tdap or Td.
• Timing of the HPV vaccine series was clarified. Both HPV vaccines have a recommended three-dose series, with the second dose administered 4-8 weeks after the first and the third dose administered at least 12 weeks after the second.
• Recommendations on the meningococcal vaccine clarify which patients need either one or two doses as well as who should receive the meningococcal conjugate (MenACWY-D) and who should have the meningococcal polysaccharide (MenACWY-CRM) quadrivalent vaccines.
You can view the 2014 schedule at our Resources section here.
The recombinant influenza vaccine is now recommended as an option for adults with egg allergies, according to the 2014 recommended vaccine schedule for adults.
Approved last year, the recombinant influenza vaccine (RIV) is manufactured with the baculovirus. It contains no egg protein, and can be safely administered to those who have an egg allergy of any severity, according to the schedule published Feb. 3 (Ann. Int. Med. 2014;160:190-8).
RIV is a good addition to the schedule, Dr. Jonathan Temte, chair of the Centers for Disease Control and Prevention’s Advisory Committee of Immunization Practices (ACIP), said in an interview.
This provides an option for people who have a serious egg allergy. Many people with egg allergies have only mild allergies – hives – and can receive influenza vaccine," Dr. Temte said.
Other updates to the schedule include the following:
• Adults who have undergone successful hematopoietic stem cell transplant should get a three-dose series of Haemophilus influenzae type B vaccine (Hib) 6-12 months after the procedure, regardless of prior vaccination. Hib is no longer recommended for previously unvaccinated HIV-infected adults, because their risk of infection is low.
• Zoster and human papillomavirus vaccines (either HPV2 or HPV4) are no longer specifically indicated for health care workers; instead, each should be provided to health care professionals and other patients who meet age and other indications.
• Previously unvaccinated patients 11 years old or older should have a single dose of tetanus, diphtheria, and acellular pertussis (Tdap), with a tetanus, diphtheria (Td) booster every 10 years. Pregnant women should get a dose of Tdap during each pregnancy (preferably during 27-36 weeks’ gestation) regardless of when they last received Tdap or Td.
• Timing of the HPV vaccine series was clarified. Both HPV vaccines have a recommended three-dose series, with the second dose administered 4-8 weeks after the first and the third dose administered at least 12 weeks after the second.
• Recommendations on the meningococcal vaccine clarify which patients need either one or two doses as well as who should receive the meningococcal conjugate (MenACWY-D) and who should have the meningococcal polysaccharide (MenACWY-CRM) quadrivalent vaccines.
You can view the 2014 schedule at our Resources section here.
FROM ANNALS OF INTERNAL MEDICINE