User login
Some providers quicker to tube feed end-of-life elderly
Subspecialists who care for dementia patients near the end of life are far more likely to introduce a feeding tube than hospitalists who follow such patients.
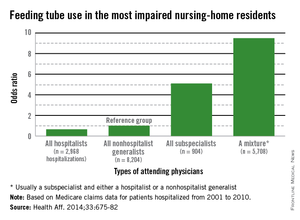
Compared with nonhospital generalists, hospitalists were 22% less likely to tube feed hospitalized nursing-home residents - and even less likely to tube feed patients who were the most severely impaired (35%). In contrast, subspecialists were five times more likely to insert a tube. When a mixed group of physicians was on the case, rates were even higher, with a 9-fold increase overall and a 9.5-fold increase for severely demented patients.
The findings clearly illustrate that many physicians could benefit from some education about the most appropriate interventions when patients near the end of life enter a hospital, Dr. Joan Teno and her associates reported in the April issue of Health Affairs (2014;33:675-82).
"It may be that subspecialists do not have adequate knowledge about the risks and benefits of using feeding tubes in people with advanced dementia," said Dr. Teno of Brown University, Providence, R.I., and her coauthors. "Hospitals should educate physicians about the lack of efficacy of PEG [percutaneous endoscopic gastrostomy] feeding tubes, compared with hand feeding, in prolonging survival and preventing aspiration pneumonias and pressure ulcers in people with advanced dementia. In addition, hospitals should examine how they staff the role of attending physician and ensure coordination of care when patient hand-offs are made between different types of attending physicians."Such education would bring all physicians up to speed with position statements against tube feeding for this group of patients. The issue sits atop the Choosing Wisely lists of both the American Academy of Hospice and Palliative Medicine and the American Geriatrics Society. The American Academy of Hospice and Palliative Medicine states that "feeding tubes do not result in improved survival, prevention of aspiration pneumonia, or improved healing of pressure ulcers. Feeding tube use in such patients has actually been associated with pressure ulcer development, use of physical and pharmacological restraints, and patient distress about the tube itself."
Dr. Eric G. Tangalos agrees with the concept that tube feeding can impose even more distress on both these patients and their families. "As a medical profession and a society, we have yet to accept some of the futility of our actions and continue to ignore the burdens tube feedings place on patients, families, and the health care system once a hospitalization has come to its conclusion," he said in an interview.
Who's the attending?
Dr. Teno and her team looked at the rate of feeding tube insertion in fee-for-service Medicare patients with advanced dementia who were within 90 days of death and hospitalized with a diagnosis of urinary tract infection, sepsis, pneumonia, or dehydration. The study examined decisions made by four groups of physicians who cared for these patients: hospitalists, nonhospitalist generalists (geriatricians, general practitioners, internists, and family physicians), subspecialists, and mixed groups that included a subspecialist and either a hospitalist or nonhospitalist generalist.
The cohort comprised 53,492 patients hospitalized from 2001 to 2010. The patients' mean age was 85 years. About 60% had a do not resuscitate order, and 10% had an order against tube feeding.
The rate of hospitalists as attending physicians increased from 11% in 2001 to 28% in 2010. The portion of patients seen by a mixture of attending physicians increased from 29% in 2001 to 38% in 2010.
The rates of tube feeding were lowest when a hospitalist or nonhospitalist generalist was the attending physician (1.6% and 2.2%, respectively). Subspecialists had significantly higher rates (11%). The highest rate occurred when there were mixed groups of physicians involved in the patient's care (15.6%).
Using the nonhospitalist generalists as a reference group, the researchers found that hospitalists were 22% less likely to insert a tube overall and 35% less likely to do so when the patient had very severe cognitive and physical impairment.
Conversely, subspecialists were five times more likely to commence tube feeding for all patients and for very severely impaired patients. The mixed groups were the most likely to begin tube feeding - almost 9 times more likely overall and 9.5 times more likely for the most severely impaired patients.
"Our finding that subspecialists had a higher rate of insertions of PEG feeding tubes might reflect their lack of experience in providing care for people with advanced dementia," the authors wrote.
The fragmention factor
The mixed-physician group could be seen as a proxy for discontinuity of care among the attending physicians, they noted. Prior studies have found that such discontinuity was associated with longer hospital stays.
"There may be a lack of care coordination during patient hand offs between attending physicians that begins a cascade of events, ending with the insertion of a PEG feeding tube."
Dr. Diane E. Meier, professor of geriatrics and palliative medicine at Icahn School of Medicine at Mount Sinai, New York, and director of the Center to Advance Palliative Care, agreed that group care without a leader creates confusion.
"One of the hallmarks of modern medicine in the U.S. is fragmentation. It is typical for a person with dementia to have a different specialist for every organ system, a problem compounded in the hospital when a completely new group of specialists is brought into the care team. The problem with this abundance of doctors is that no one is really in charge of the whole patient and what makes the most sense for the patient as a person.
"Organ- and specialty-specific decision making leads to bad practices - including trying to 'solve' a feeding difficulty as if it is an isolated problem when the real issue is progressive brain failure - a terminal illness that cannot be fixed with a feeding tube."
The study questions not only the feeding tube issue, but also the wisdom of repeatedly hospitalizing elderly patients with severe dementia who could be in the last phase of life - especially for conditions that are expected complications of severe dementia. The authors suggested that there may be financial motives to admit fee-for-service patients.
"The fee-for-service system provides incentives to hospitalize nursing-home residents with severe dementia because such hospitalizations qualify the patients for skilled nursing-home services. Bundling of payments and institutional special needs plans that reverse these financial incentives may reduce health care expenditures and improve the quality of care for nursing-home residents with advanced dementia by avoiding burdensome transitions between facilities and the stress of relocation."
The National Institute on Aging funded the study. Dr. Teno made no financial declarations.
Dr. Daniel Ouellette, FCCP, comments: The Choosing Wisely campaign sponsored by Consumer Reports and the American Board of Internal Medicine Foundation provides recommendations to help patients and providers make wise decisions about appropriate care. Both the American Academy of Hospice and Palliative Medicine and the American Geriatrics Society target the inappropriate use of feeding tubes in persons at the end of life, suggesting that such treatments do not improve patient outcomes. The news that subspecialists and primary care physicians order feeding tubes much more frequently than hospitalists is sobering information for pulmonary and critical care physicians taking care of such patients. However, future investigations focused on nutritional support and involving the patient population that pulmonologists frequently manage is likely to be complicated by the facts that such patients are or have been critically ill, and are likely to implicate a broader array of end-of-life decision making than just the provision of feeding.
Dr. Daniel Ouellette, FCCP, comments: The Choosing Wisely campaign sponsored by Consumer Reports and the American Board of Internal Medicine Foundation provides recommendations to help patients and providers make wise decisions about appropriate care. Both the American Academy of Hospice and Palliative Medicine and the American Geriatrics Society target the inappropriate use of feeding tubes in persons at the end of life, suggesting that such treatments do not improve patient outcomes. The news that subspecialists and primary care physicians order feeding tubes much more frequently than hospitalists is sobering information for pulmonary and critical care physicians taking care of such patients. However, future investigations focused on nutritional support and involving the patient population that pulmonologists frequently manage is likely to be complicated by the facts that such patients are or have been critically ill, and are likely to implicate a broader array of end-of-life decision making than just the provision of feeding.
Dr. Daniel Ouellette, FCCP, comments: The Choosing Wisely campaign sponsored by Consumer Reports and the American Board of Internal Medicine Foundation provides recommendations to help patients and providers make wise decisions about appropriate care. Both the American Academy of Hospice and Palliative Medicine and the American Geriatrics Society target the inappropriate use of feeding tubes in persons at the end of life, suggesting that such treatments do not improve patient outcomes. The news that subspecialists and primary care physicians order feeding tubes much more frequently than hospitalists is sobering information for pulmonary and critical care physicians taking care of such patients. However, future investigations focused on nutritional support and involving the patient population that pulmonologists frequently manage is likely to be complicated by the facts that such patients are or have been critically ill, and are likely to implicate a broader array of end-of-life decision making than just the provision of feeding.
Subspecialists who care for dementia patients near the end of life are far more likely to introduce a feeding tube than hospitalists who follow such patients.
Compared with nonhospital generalists, hospitalists were 22% less likely to tube feed hospitalized nursing-home residents - and even less likely to tube feed patients who were the most severely impaired (35%). In contrast, subspecialists were five times more likely to insert a tube. When a mixed group of physicians was on the case, rates were even higher, with a 9-fold increase overall and a 9.5-fold increase for severely demented patients.
The findings clearly illustrate that many physicians could benefit from some education about the most appropriate interventions when patients near the end of life enter a hospital, Dr. Joan Teno and her associates reported in the April issue of Health Affairs (2014;33:675-82).

"It may be that subspecialists do not have adequate knowledge about the risks and benefits of using feeding tubes in people with advanced dementia," said Dr. Teno of Brown University, Providence, R.I., and her coauthors. "Hospitals should educate physicians about the lack of efficacy of PEG [percutaneous endoscopic gastrostomy] feeding tubes, compared with hand feeding, in prolonging survival and preventing aspiration pneumonias and pressure ulcers in people with advanced dementia. In addition, hospitals should examine how they staff the role of attending physician and ensure coordination of care when patient hand-offs are made between different types of attending physicians."Such education would bring all physicians up to speed with position statements against tube feeding for this group of patients. The issue sits atop the Choosing Wisely lists of both the American Academy of Hospice and Palliative Medicine and the American Geriatrics Society. The American Academy of Hospice and Palliative Medicine states that "feeding tubes do not result in improved survival, prevention of aspiration pneumonia, or improved healing of pressure ulcers. Feeding tube use in such patients has actually been associated with pressure ulcer development, use of physical and pharmacological restraints, and patient distress about the tube itself."
Dr. Eric G. Tangalos agrees with the concept that tube feeding can impose even more distress on both these patients and their families. "As a medical profession and a society, we have yet to accept some of the futility of our actions and continue to ignore the burdens tube feedings place on patients, families, and the health care system once a hospitalization has come to its conclusion," he said in an interview.
Who's the attending?
Dr. Teno and her team looked at the rate of feeding tube insertion in fee-for-service Medicare patients with advanced dementia who were within 90 days of death and hospitalized with a diagnosis of urinary tract infection, sepsis, pneumonia, or dehydration. The study examined decisions made by four groups of physicians who cared for these patients: hospitalists, nonhospitalist generalists (geriatricians, general practitioners, internists, and family physicians), subspecialists, and mixed groups that included a subspecialist and either a hospitalist or nonhospitalist generalist.
The cohort comprised 53,492 patients hospitalized from 2001 to 2010. The patients' mean age was 85 years. About 60% had a do not resuscitate order, and 10% had an order against tube feeding.
The rate of hospitalists as attending physicians increased from 11% in 2001 to 28% in 2010. The portion of patients seen by a mixture of attending physicians increased from 29% in 2001 to 38% in 2010.
The rates of tube feeding were lowest when a hospitalist or nonhospitalist generalist was the attending physician (1.6% and 2.2%, respectively). Subspecialists had significantly higher rates (11%). The highest rate occurred when there were mixed groups of physicians involved in the patient's care (15.6%).
Using the nonhospitalist generalists as a reference group, the researchers found that hospitalists were 22% less likely to insert a tube overall and 35% less likely to do so when the patient had very severe cognitive and physical impairment.
Conversely, subspecialists were five times more likely to commence tube feeding for all patients and for very severely impaired patients. The mixed groups were the most likely to begin tube feeding - almost 9 times more likely overall and 9.5 times more likely for the most severely impaired patients.
"Our finding that subspecialists had a higher rate of insertions of PEG feeding tubes might reflect their lack of experience in providing care for people with advanced dementia," the authors wrote.
The fragmention factor
The mixed-physician group could be seen as a proxy for discontinuity of care among the attending physicians, they noted. Prior studies have found that such discontinuity was associated with longer hospital stays.
"There may be a lack of care coordination during patient hand offs between attending physicians that begins a cascade of events, ending with the insertion of a PEG feeding tube."
Dr. Diane E. Meier, professor of geriatrics and palliative medicine at Icahn School of Medicine at Mount Sinai, New York, and director of the Center to Advance Palliative Care, agreed that group care without a leader creates confusion.
"One of the hallmarks of modern medicine in the U.S. is fragmentation. It is typical for a person with dementia to have a different specialist for every organ system, a problem compounded in the hospital when a completely new group of specialists is brought into the care team. The problem with this abundance of doctors is that no one is really in charge of the whole patient and what makes the most sense for the patient as a person.
"Organ- and specialty-specific decision making leads to bad practices - including trying to 'solve' a feeding difficulty as if it is an isolated problem when the real issue is progressive brain failure - a terminal illness that cannot be fixed with a feeding tube."
The study questions not only the feeding tube issue, but also the wisdom of repeatedly hospitalizing elderly patients with severe dementia who could be in the last phase of life - especially for conditions that are expected complications of severe dementia. The authors suggested that there may be financial motives to admit fee-for-service patients.
"The fee-for-service system provides incentives to hospitalize nursing-home residents with severe dementia because such hospitalizations qualify the patients for skilled nursing-home services. Bundling of payments and institutional special needs plans that reverse these financial incentives may reduce health care expenditures and improve the quality of care for nursing-home residents with advanced dementia by avoiding burdensome transitions between facilities and the stress of relocation."
The National Institute on Aging funded the study. Dr. Teno made no financial declarations.
Subspecialists who care for dementia patients near the end of life are far more likely to introduce a feeding tube than hospitalists who follow such patients.
Compared with nonhospital generalists, hospitalists were 22% less likely to tube feed hospitalized nursing-home residents - and even less likely to tube feed patients who were the most severely impaired (35%). In contrast, subspecialists were five times more likely to insert a tube. When a mixed group of physicians was on the case, rates were even higher, with a 9-fold increase overall and a 9.5-fold increase for severely demented patients.
The findings clearly illustrate that many physicians could benefit from some education about the most appropriate interventions when patients near the end of life enter a hospital, Dr. Joan Teno and her associates reported in the April issue of Health Affairs (2014;33:675-82).

"It may be that subspecialists do not have adequate knowledge about the risks and benefits of using feeding tubes in people with advanced dementia," said Dr. Teno of Brown University, Providence, R.I., and her coauthors. "Hospitals should educate physicians about the lack of efficacy of PEG [percutaneous endoscopic gastrostomy] feeding tubes, compared with hand feeding, in prolonging survival and preventing aspiration pneumonias and pressure ulcers in people with advanced dementia. In addition, hospitals should examine how they staff the role of attending physician and ensure coordination of care when patient hand-offs are made between different types of attending physicians."Such education would bring all physicians up to speed with position statements against tube feeding for this group of patients. The issue sits atop the Choosing Wisely lists of both the American Academy of Hospice and Palliative Medicine and the American Geriatrics Society. The American Academy of Hospice and Palliative Medicine states that "feeding tubes do not result in improved survival, prevention of aspiration pneumonia, or improved healing of pressure ulcers. Feeding tube use in such patients has actually been associated with pressure ulcer development, use of physical and pharmacological restraints, and patient distress about the tube itself."
Dr. Eric G. Tangalos agrees with the concept that tube feeding can impose even more distress on both these patients and their families. "As a medical profession and a society, we have yet to accept some of the futility of our actions and continue to ignore the burdens tube feedings place on patients, families, and the health care system once a hospitalization has come to its conclusion," he said in an interview.
Who's the attending?
Dr. Teno and her team looked at the rate of feeding tube insertion in fee-for-service Medicare patients with advanced dementia who were within 90 days of death and hospitalized with a diagnosis of urinary tract infection, sepsis, pneumonia, or dehydration. The study examined decisions made by four groups of physicians who cared for these patients: hospitalists, nonhospitalist generalists (geriatricians, general practitioners, internists, and family physicians), subspecialists, and mixed groups that included a subspecialist and either a hospitalist or nonhospitalist generalist.
The cohort comprised 53,492 patients hospitalized from 2001 to 2010. The patients' mean age was 85 years. About 60% had a do not resuscitate order, and 10% had an order against tube feeding.
The rate of hospitalists as attending physicians increased from 11% in 2001 to 28% in 2010. The portion of patients seen by a mixture of attending physicians increased from 29% in 2001 to 38% in 2010.
The rates of tube feeding were lowest when a hospitalist or nonhospitalist generalist was the attending physician (1.6% and 2.2%, respectively). Subspecialists had significantly higher rates (11%). The highest rate occurred when there were mixed groups of physicians involved in the patient's care (15.6%).
Using the nonhospitalist generalists as a reference group, the researchers found that hospitalists were 22% less likely to insert a tube overall and 35% less likely to do so when the patient had very severe cognitive and physical impairment.
Conversely, subspecialists were five times more likely to commence tube feeding for all patients and for very severely impaired patients. The mixed groups were the most likely to begin tube feeding - almost 9 times more likely overall and 9.5 times more likely for the most severely impaired patients.
"Our finding that subspecialists had a higher rate of insertions of PEG feeding tubes might reflect their lack of experience in providing care for people with advanced dementia," the authors wrote.
The fragmention factor
The mixed-physician group could be seen as a proxy for discontinuity of care among the attending physicians, they noted. Prior studies have found that such discontinuity was associated with longer hospital stays.
"There may be a lack of care coordination during patient hand offs between attending physicians that begins a cascade of events, ending with the insertion of a PEG feeding tube."
Dr. Diane E. Meier, professor of geriatrics and palliative medicine at Icahn School of Medicine at Mount Sinai, New York, and director of the Center to Advance Palliative Care, agreed that group care without a leader creates confusion.
"One of the hallmarks of modern medicine in the U.S. is fragmentation. It is typical for a person with dementia to have a different specialist for every organ system, a problem compounded in the hospital when a completely new group of specialists is brought into the care team. The problem with this abundance of doctors is that no one is really in charge of the whole patient and what makes the most sense for the patient as a person.
"Organ- and specialty-specific decision making leads to bad practices - including trying to 'solve' a feeding difficulty as if it is an isolated problem when the real issue is progressive brain failure - a terminal illness that cannot be fixed with a feeding tube."
The study questions not only the feeding tube issue, but also the wisdom of repeatedly hospitalizing elderly patients with severe dementia who could be in the last phase of life - especially for conditions that are expected complications of severe dementia. The authors suggested that there may be financial motives to admit fee-for-service patients.
"The fee-for-service system provides incentives to hospitalize nursing-home residents with severe dementia because such hospitalizations qualify the patients for skilled nursing-home services. Bundling of payments and institutional special needs plans that reverse these financial incentives may reduce health care expenditures and improve the quality of care for nursing-home residents with advanced dementia by avoiding burdensome transitions between facilities and the stress of relocation."
The National Institute on Aging funded the study. Dr. Teno made no financial declarations.
FROM HEALTH AFFAIRS
Major finding: Primary care doctors and subspecialists were up
to 9 times more likely to initiate tube feeding in hospitalized
patients with severe dementia.
Data source: Data are from a review of more than 53,000 Medicare fee-for-service nursing home patients.
Disclosures: The National Institute on Aging sponsored the study. Dr. Teno had no financial disclosures.
Proposed neuromyelitis optica diagnostic criteria reflect new disease understanding
PHILADELPHIA – A proposed revision of the neuromyelitis optica diagnostic criteria takes into account newly appreciated variations in how the disease can present clinically.
If adopted, the new criteria would offer diagnostic pathways for patients who have symptoms, but who might or might not have the serum antibodies usually associated with the disorder, Dr. Dean Wingerchuk said at the annual meeting of the American Academy of Neurology.
They reflect the current understanding of neuromyelitis optica as a spectrum of clinical symptoms, said Dr. Wingerchuk, professor of neurology at the Mayo Clinic, Scottsdale, Ariz. Neuromyelitis optica spectrum disorder (NMOSD) was identified in 2007 – 1 year after the existing diagnostic criteria were published.
In the new guidelines, "we wanted to encompass all patients who would have previously been diagnosed as having neuromyelitis optica or NMOSD," he said. A new stratification of antibody positive or antibody negative reflects the fact that not all patients are seropositive at presentation, particularly early in the disease course; that antibody testing is not available or reliable everywhere; and that as-yet-unidentified antibodies might be implicated in the disorder.
The workgroup that authored the document consisted of 18 members from nine countries. It began its work in 2011. The proposed criteria still need to be prospectively validated before they could be widely adopted, noted Dr. Wingerchuk, who was a primary author of the 2006 criteria.
The existing criteria require the presence of transverse myelitis, optic neuritis, and at least two of the following:
• Brain MRI imaging findings that are nondiagnostic for multiple sclerosis.
• A spinal cord lesion extending over three or more vertebral segments.
• Seropositivity for NMO-IgG.
The newly proposed criteria have been expanded to include six different core characteristics: optic neuritis; acute myelitis; area postrema syndrome (nausea, vomiting, and hiccups); other brain stem syndromes; symptomatic narcolepsy or acute diencephalic syndrome with MRI findings; and symptomatic cerebral syndrome with MRI findings.
Antibody-positive patients need to show at least one of these core characteristics, with no other better explanation for their symptoms.
The bar is a little higher for antibody-negative patients. They need to show at least two of the core characteristics, meeting the following requirements:
• At least one of the core symptoms must be optic neuritis, myelitis, or area postrema syndrome.
• The core characteristics must be disseminated in space.
• MRI findings must distinguish NMOSD from multiple sclerosis or other demyelinating disorders.
Prospective validation will require follow-up of patients who are seropositive at diagnosis but present with less common syndromes, and detailed descriptions of seronegative groups to determine whether they eventually convert to a clinical NMOSD, Dr. Wingerchuk said.
The project is being funded by the Guthy-Jackson Charitable Foundation. Dr. Wingerchuk disclosed that he has received financial compensation on an adjudication committee for an NMO trial that was sponsored by MedImmune.
On Twitter @alz_gal
PHILADELPHIA – A proposed revision of the neuromyelitis optica diagnostic criteria takes into account newly appreciated variations in how the disease can present clinically.
If adopted, the new criteria would offer diagnostic pathways for patients who have symptoms, but who might or might not have the serum antibodies usually associated with the disorder, Dr. Dean Wingerchuk said at the annual meeting of the American Academy of Neurology.
They reflect the current understanding of neuromyelitis optica as a spectrum of clinical symptoms, said Dr. Wingerchuk, professor of neurology at the Mayo Clinic, Scottsdale, Ariz. Neuromyelitis optica spectrum disorder (NMOSD) was identified in 2007 – 1 year after the existing diagnostic criteria were published.
In the new guidelines, "we wanted to encompass all patients who would have previously been diagnosed as having neuromyelitis optica or NMOSD," he said. A new stratification of antibody positive or antibody negative reflects the fact that not all patients are seropositive at presentation, particularly early in the disease course; that antibody testing is not available or reliable everywhere; and that as-yet-unidentified antibodies might be implicated in the disorder.
The workgroup that authored the document consisted of 18 members from nine countries. It began its work in 2011. The proposed criteria still need to be prospectively validated before they could be widely adopted, noted Dr. Wingerchuk, who was a primary author of the 2006 criteria.
The existing criteria require the presence of transverse myelitis, optic neuritis, and at least two of the following:
• Brain MRI imaging findings that are nondiagnostic for multiple sclerosis.
• A spinal cord lesion extending over three or more vertebral segments.
• Seropositivity for NMO-IgG.
The newly proposed criteria have been expanded to include six different core characteristics: optic neuritis; acute myelitis; area postrema syndrome (nausea, vomiting, and hiccups); other brain stem syndromes; symptomatic narcolepsy or acute diencephalic syndrome with MRI findings; and symptomatic cerebral syndrome with MRI findings.
Antibody-positive patients need to show at least one of these core characteristics, with no other better explanation for their symptoms.
The bar is a little higher for antibody-negative patients. They need to show at least two of the core characteristics, meeting the following requirements:
• At least one of the core symptoms must be optic neuritis, myelitis, or area postrema syndrome.
• The core characteristics must be disseminated in space.
• MRI findings must distinguish NMOSD from multiple sclerosis or other demyelinating disorders.
Prospective validation will require follow-up of patients who are seropositive at diagnosis but present with less common syndromes, and detailed descriptions of seronegative groups to determine whether they eventually convert to a clinical NMOSD, Dr. Wingerchuk said.
The project is being funded by the Guthy-Jackson Charitable Foundation. Dr. Wingerchuk disclosed that he has received financial compensation on an adjudication committee for an NMO trial that was sponsored by MedImmune.
On Twitter @alz_gal
PHILADELPHIA – A proposed revision of the neuromyelitis optica diagnostic criteria takes into account newly appreciated variations in how the disease can present clinically.
If adopted, the new criteria would offer diagnostic pathways for patients who have symptoms, but who might or might not have the serum antibodies usually associated with the disorder, Dr. Dean Wingerchuk said at the annual meeting of the American Academy of Neurology.
They reflect the current understanding of neuromyelitis optica as a spectrum of clinical symptoms, said Dr. Wingerchuk, professor of neurology at the Mayo Clinic, Scottsdale, Ariz. Neuromyelitis optica spectrum disorder (NMOSD) was identified in 2007 – 1 year after the existing diagnostic criteria were published.
In the new guidelines, "we wanted to encompass all patients who would have previously been diagnosed as having neuromyelitis optica or NMOSD," he said. A new stratification of antibody positive or antibody negative reflects the fact that not all patients are seropositive at presentation, particularly early in the disease course; that antibody testing is not available or reliable everywhere; and that as-yet-unidentified antibodies might be implicated in the disorder.
The workgroup that authored the document consisted of 18 members from nine countries. It began its work in 2011. The proposed criteria still need to be prospectively validated before they could be widely adopted, noted Dr. Wingerchuk, who was a primary author of the 2006 criteria.
The existing criteria require the presence of transverse myelitis, optic neuritis, and at least two of the following:
• Brain MRI imaging findings that are nondiagnostic for multiple sclerosis.
• A spinal cord lesion extending over three or more vertebral segments.
• Seropositivity for NMO-IgG.
The newly proposed criteria have been expanded to include six different core characteristics: optic neuritis; acute myelitis; area postrema syndrome (nausea, vomiting, and hiccups); other brain stem syndromes; symptomatic narcolepsy or acute diencephalic syndrome with MRI findings; and symptomatic cerebral syndrome with MRI findings.
Antibody-positive patients need to show at least one of these core characteristics, with no other better explanation for their symptoms.
The bar is a little higher for antibody-negative patients. They need to show at least two of the core characteristics, meeting the following requirements:
• At least one of the core symptoms must be optic neuritis, myelitis, or area postrema syndrome.
• The core characteristics must be disseminated in space.
• MRI findings must distinguish NMOSD from multiple sclerosis or other demyelinating disorders.
Prospective validation will require follow-up of patients who are seropositive at diagnosis but present with less common syndromes, and detailed descriptions of seronegative groups to determine whether they eventually convert to a clinical NMOSD, Dr. Wingerchuk said.
The project is being funded by the Guthy-Jackson Charitable Foundation. Dr. Wingerchuk disclosed that he has received financial compensation on an adjudication committee for an NMO trial that was sponsored by MedImmune.
On Twitter @alz_gal
AT THE AAN 2014 ANNUAL MEETING
Childhood obesity predicts adult cardiovascular risks
Teens who are overweight, but metabolically healthy, still run a significantly higher risk of developing increased carotid intimal-medial thickness and other cardiovascular risk factors as adults.
A study of more than 1,600 Finns who were followed for up to 25 years found that, while childhood metabolic risk factors significantly contributed to adult cardiovascular risks, obesity in isolation was also a very strong predictor, Dr. Juha Koskinen and his colleagues reported online April 17 in Diabetes Care (Diab. Care 2014 April 14 [doi: 10.2337/dc14-0008]).
"The findings demonstrate an increased risk of future raised intimal-medial thickness in pediatric overweight individuals, even in those without metabolic derangement in youth – a finding that assists with clinical decision making in screening and pediatric overweight and obesity management," wrote Dr. Koskinen of the University of Turku, Finland, and his coauthors.
The ongoing Cardiovascular Risk in Young Finns Study is one of the largest follow-up studies tracking cardiovascular risk from childhood to adulthood. Its main aim is to determine how childhood lifestyle, as well as biological and psychological measures, affects the risk of future cardiovascular diseases. Initiated in 1980, it has since tracked a dwindling number of the 3,596 young people originally enrolled when they were 3-18 years old. In 2007, a total of 2,204 subjects were examined. The most recent follow-up was performed in 2011 and included blood samples and questionnaires but not ultrasound data.
For this study, the researchers focused on 1,617 youths who were a mean of 16 years old in 1986. The researchers divided this cohort into four groups according to baseline cardiovascular risk factors:
• Group 1: normal weight, no metabolic problems.
• Group 2: normal weight, at least one metabolic problem, such as elevated blood pressure, glucose, triglycerides, low HDL cholesterol, and high LDL cholesterol.
• Group 3: overweight/obese, no metabolic problems.
• Group 4: overweight/obese, at least one metabolic problem.
They assessed the 21-year risk of subclinical atherosclerosis (as determined by ultrasonographic measurement of carotid intimal-medial thickness), and the 21- to 25-year risks of developing type 2 diabetes and metabolic syndrome.
By the 21-year follow-up, carotid intimal-medial thickness had increased linearly according to the baseline groupings, with overweight/obese subjects (groups 3 and 4) having significantly greater values than group 1. After adjusting for baseline glucose, triglycerides, lipids, systolic blood pressure, smoking, and family history of heart disease, the difference between groups 1 and 2 was not significant, but the differences between all of the other groups remained so.
By 25 years, compared with subjects with normal weight and metabolic status, those who were overweight but metabolically normal at baseline were twice as likely to have metabolic syndrome and six times as likely to have diabetes.
Subjects who were overweight and had metabolic problems at baseline were more than three times as likely to have metabolic syndrome and three times as likely to have diabetes.
"Our analysis ... supports the encouragement of young individuals to avoid being overweight/ obese, irrespective of their metabolic profile," the authors concluded.
The Cardiovascular Risk in Young Finns Study is supported by several national and private grants. None of the authors reported any financial disclosures.
Teens who are overweight, but metabolically healthy, still run a significantly higher risk of developing increased carotid intimal-medial thickness and other cardiovascular risk factors as adults.
A study of more than 1,600 Finns who were followed for up to 25 years found that, while childhood metabolic risk factors significantly contributed to adult cardiovascular risks, obesity in isolation was also a very strong predictor, Dr. Juha Koskinen and his colleagues reported online April 17 in Diabetes Care (Diab. Care 2014 April 14 [doi: 10.2337/dc14-0008]).
"The findings demonstrate an increased risk of future raised intimal-medial thickness in pediatric overweight individuals, even in those without metabolic derangement in youth – a finding that assists with clinical decision making in screening and pediatric overweight and obesity management," wrote Dr. Koskinen of the University of Turku, Finland, and his coauthors.
The ongoing Cardiovascular Risk in Young Finns Study is one of the largest follow-up studies tracking cardiovascular risk from childhood to adulthood. Its main aim is to determine how childhood lifestyle, as well as biological and psychological measures, affects the risk of future cardiovascular diseases. Initiated in 1980, it has since tracked a dwindling number of the 3,596 young people originally enrolled when they were 3-18 years old. In 2007, a total of 2,204 subjects were examined. The most recent follow-up was performed in 2011 and included blood samples and questionnaires but not ultrasound data.
For this study, the researchers focused on 1,617 youths who were a mean of 16 years old in 1986. The researchers divided this cohort into four groups according to baseline cardiovascular risk factors:
• Group 1: normal weight, no metabolic problems.
• Group 2: normal weight, at least one metabolic problem, such as elevated blood pressure, glucose, triglycerides, low HDL cholesterol, and high LDL cholesterol.
• Group 3: overweight/obese, no metabolic problems.
• Group 4: overweight/obese, at least one metabolic problem.
They assessed the 21-year risk of subclinical atherosclerosis (as determined by ultrasonographic measurement of carotid intimal-medial thickness), and the 21- to 25-year risks of developing type 2 diabetes and metabolic syndrome.
By the 21-year follow-up, carotid intimal-medial thickness had increased linearly according to the baseline groupings, with overweight/obese subjects (groups 3 and 4) having significantly greater values than group 1. After adjusting for baseline glucose, triglycerides, lipids, systolic blood pressure, smoking, and family history of heart disease, the difference between groups 1 and 2 was not significant, but the differences between all of the other groups remained so.
By 25 years, compared with subjects with normal weight and metabolic status, those who were overweight but metabolically normal at baseline were twice as likely to have metabolic syndrome and six times as likely to have diabetes.
Subjects who were overweight and had metabolic problems at baseline were more than three times as likely to have metabolic syndrome and three times as likely to have diabetes.
"Our analysis ... supports the encouragement of young individuals to avoid being overweight/ obese, irrespective of their metabolic profile," the authors concluded.
The Cardiovascular Risk in Young Finns Study is supported by several national and private grants. None of the authors reported any financial disclosures.
Teens who are overweight, but metabolically healthy, still run a significantly higher risk of developing increased carotid intimal-medial thickness and other cardiovascular risk factors as adults.
A study of more than 1,600 Finns who were followed for up to 25 years found that, while childhood metabolic risk factors significantly contributed to adult cardiovascular risks, obesity in isolation was also a very strong predictor, Dr. Juha Koskinen and his colleagues reported online April 17 in Diabetes Care (Diab. Care 2014 April 14 [doi: 10.2337/dc14-0008]).
"The findings demonstrate an increased risk of future raised intimal-medial thickness in pediatric overweight individuals, even in those without metabolic derangement in youth – a finding that assists with clinical decision making in screening and pediatric overweight and obesity management," wrote Dr. Koskinen of the University of Turku, Finland, and his coauthors.
The ongoing Cardiovascular Risk in Young Finns Study is one of the largest follow-up studies tracking cardiovascular risk from childhood to adulthood. Its main aim is to determine how childhood lifestyle, as well as biological and psychological measures, affects the risk of future cardiovascular diseases. Initiated in 1980, it has since tracked a dwindling number of the 3,596 young people originally enrolled when they were 3-18 years old. In 2007, a total of 2,204 subjects were examined. The most recent follow-up was performed in 2011 and included blood samples and questionnaires but not ultrasound data.
For this study, the researchers focused on 1,617 youths who were a mean of 16 years old in 1986. The researchers divided this cohort into four groups according to baseline cardiovascular risk factors:
• Group 1: normal weight, no metabolic problems.
• Group 2: normal weight, at least one metabolic problem, such as elevated blood pressure, glucose, triglycerides, low HDL cholesterol, and high LDL cholesterol.
• Group 3: overweight/obese, no metabolic problems.
• Group 4: overweight/obese, at least one metabolic problem.
They assessed the 21-year risk of subclinical atherosclerosis (as determined by ultrasonographic measurement of carotid intimal-medial thickness), and the 21- to 25-year risks of developing type 2 diabetes and metabolic syndrome.
By the 21-year follow-up, carotid intimal-medial thickness had increased linearly according to the baseline groupings, with overweight/obese subjects (groups 3 and 4) having significantly greater values than group 1. After adjusting for baseline glucose, triglycerides, lipids, systolic blood pressure, smoking, and family history of heart disease, the difference between groups 1 and 2 was not significant, but the differences between all of the other groups remained so.
By 25 years, compared with subjects with normal weight and metabolic status, those who were overweight but metabolically normal at baseline were twice as likely to have metabolic syndrome and six times as likely to have diabetes.
Subjects who were overweight and had metabolic problems at baseline were more than three times as likely to have metabolic syndrome and three times as likely to have diabetes.
"Our analysis ... supports the encouragement of young individuals to avoid being overweight/ obese, irrespective of their metabolic profile," the authors concluded.
The Cardiovascular Risk in Young Finns Study is supported by several national and private grants. None of the authors reported any financial disclosures.
FROM DIABETES CARE
Major finding: Overweight but metabolically normal children were twice as likely to develop diabetes and six times as likely to develop metabolic syndrome as adults, compared with normal-weight children.
Data source: The study tracked more than 1,600 children for up to 25 years.
Disclosures: The Cardiovascular Risk in Young Finns Study is supported by several national and private grants. None of the authors reported any financial disclosures.
Orexin antagonist improved sleep in phase III studies
PHILADELPHIA – An orexin receptor antagonist – the first drug to affect a neural system that promotes wakefulness – has proven safe and effective for up to 1 year in a pooled analysis of three phase III studies of patients with insomnia.
The drug (suvorexant) significantly improved both subjective and objective measures of sleep, including sleep onset, total sleep time, and wakefulness after sleep, Dr. W. Joseph Herring said at the annual meeting of the American Academy of Neurology.
Based on these data, the manufacturer, Merck, proceeded last year to preliminary talks with the Food and Drug Administration. The committee considering suvorexant recommended that Merck focus on the smaller of two proposed dose ranges – 20 mg for people up to 64 years of age, and 15 mg for those aged 65 years and older.
Although the proposed higher doses of 40 mg for younger patients and 30 mg for older patients, were deemed safe and effective, the lower dose recommendation by the FDA is consistent with its "usual recommendations to go with lower doses of sleep medications," said Dr. Herring, Merck’s executive director of neuroscience clinical research. "So this 15/20-mg regimen is the most likely to be clinically relevant to prescribers."
The orexin neural system was discovered in the late 1990s, Dr. Herring noted. Orexin neurons release two neuropeptides that interact with downstream receptors that promote wakefulness. Secretion follows a circadian rhythm. Suvorexant orexin antagonists block the activity of this wake-signaling system, allowing sleep to occur.
So far, studies have investigated the drug in almost 3,000 patients; 160 of these were treated for at least a year. The three phase III trials comprised more than 275,000 exposure nights. Most of the patients were women; 46% of the patients were aged 65 years or older.
Sleep was measured by subjective assessment and objective scales, including polysomnography.
In the two efficacy studies, the higher doses decreased time to persistent sleep by 15 minutes on the first night; the lower doses did so by 10 minutes. By 3 months, the high-dose group’s decreased time to persistent sleep was 5 minutes; the low dose group’s time was lowered to 4 minutes.
On the first night, the high-dose drug reduced wakefulness after sleep by 40 minutes; the low-dose drug did so by 35 minutes. After 3 months, the reductions were similar (about 25 minutes).
When the night was divided into thirds, both doses decreased wakefulness significantly and about equally, especially in the second and third fractions of the night.
In the subgroup of 160 patients who were treated for at least 12 months, the drugs showed persistent efficacy overall, although the high doses were somewhat more effective, Dr. Herring said.
A multivariate regression analysis found that, compared with placebo, patients who took the high dose were 2.4 times more likely to be considered responders at 1 month and 1.8 times more likely to be responders at 3 months. Those who took the low dose were 1.8 times more likely to respond at 1 and 3 months.
"The drugs really proved about equal in the chances of a good response," said Dr. Herring.
Although there were more adverse events in the active groups than in the placebo groups – and more in the high-dose groups, compared with the low-dose groups – suvorexant was considered safe, he said. The discontinuation rates for drug-related adverse events in the high-dose, low-dose, and placebo groups were 4%, 3%, and 2%, respectively.
The most common issues were next-day somnolence (3% for placebo, 7% with the low dose, and 11% for the high dose, respectively); headache (6%, 7%, and 7%, respectively); and fatigue (2%, 2%, and 4%, respectively).
Few serious adverse events were reported. These included one case of suicidal ideation each in the placebo and low-dose groups, and eight cases in the high-dose group. Dr. Herring said these were mild to moderate and of short duration.
There were no cases of falls or cataplexy, although two patients taking the high dose showed complex sleep behaviors.
Neither of the doses was associated with withdrawal symptoms or clinically significant insomnia rebound during the washout periods. "The drug appears to have a low potential for abuse," Dr. Herring noted.
Merck sponsored the studies. Dr. Herring is a full-time employee of the company.
On Twitter @alz_gal
PHILADELPHIA – An orexin receptor antagonist – the first drug to affect a neural system that promotes wakefulness – has proven safe and effective for up to 1 year in a pooled analysis of three phase III studies of patients with insomnia.
The drug (suvorexant) significantly improved both subjective and objective measures of sleep, including sleep onset, total sleep time, and wakefulness after sleep, Dr. W. Joseph Herring said at the annual meeting of the American Academy of Neurology.
Based on these data, the manufacturer, Merck, proceeded last year to preliminary talks with the Food and Drug Administration. The committee considering suvorexant recommended that Merck focus on the smaller of two proposed dose ranges – 20 mg for people up to 64 years of age, and 15 mg for those aged 65 years and older.
Although the proposed higher doses of 40 mg for younger patients and 30 mg for older patients, were deemed safe and effective, the lower dose recommendation by the FDA is consistent with its "usual recommendations to go with lower doses of sleep medications," said Dr. Herring, Merck’s executive director of neuroscience clinical research. "So this 15/20-mg regimen is the most likely to be clinically relevant to prescribers."
The orexin neural system was discovered in the late 1990s, Dr. Herring noted. Orexin neurons release two neuropeptides that interact with downstream receptors that promote wakefulness. Secretion follows a circadian rhythm. Suvorexant orexin antagonists block the activity of this wake-signaling system, allowing sleep to occur.
So far, studies have investigated the drug in almost 3,000 patients; 160 of these were treated for at least a year. The three phase III trials comprised more than 275,000 exposure nights. Most of the patients were women; 46% of the patients were aged 65 years or older.
Sleep was measured by subjective assessment and objective scales, including polysomnography.
In the two efficacy studies, the higher doses decreased time to persistent sleep by 15 minutes on the first night; the lower doses did so by 10 minutes. By 3 months, the high-dose group’s decreased time to persistent sleep was 5 minutes; the low dose group’s time was lowered to 4 minutes.
On the first night, the high-dose drug reduced wakefulness after sleep by 40 minutes; the low-dose drug did so by 35 minutes. After 3 months, the reductions were similar (about 25 minutes).
When the night was divided into thirds, both doses decreased wakefulness significantly and about equally, especially in the second and third fractions of the night.
In the subgroup of 160 patients who were treated for at least 12 months, the drugs showed persistent efficacy overall, although the high doses were somewhat more effective, Dr. Herring said.
A multivariate regression analysis found that, compared with placebo, patients who took the high dose were 2.4 times more likely to be considered responders at 1 month and 1.8 times more likely to be responders at 3 months. Those who took the low dose were 1.8 times more likely to respond at 1 and 3 months.
"The drugs really proved about equal in the chances of a good response," said Dr. Herring.
Although there were more adverse events in the active groups than in the placebo groups – and more in the high-dose groups, compared with the low-dose groups – suvorexant was considered safe, he said. The discontinuation rates for drug-related adverse events in the high-dose, low-dose, and placebo groups were 4%, 3%, and 2%, respectively.
The most common issues were next-day somnolence (3% for placebo, 7% with the low dose, and 11% for the high dose, respectively); headache (6%, 7%, and 7%, respectively); and fatigue (2%, 2%, and 4%, respectively).
Few serious adverse events were reported. These included one case of suicidal ideation each in the placebo and low-dose groups, and eight cases in the high-dose group. Dr. Herring said these were mild to moderate and of short duration.
There were no cases of falls or cataplexy, although two patients taking the high dose showed complex sleep behaviors.
Neither of the doses was associated with withdrawal symptoms or clinically significant insomnia rebound during the washout periods. "The drug appears to have a low potential for abuse," Dr. Herring noted.
Merck sponsored the studies. Dr. Herring is a full-time employee of the company.
On Twitter @alz_gal
PHILADELPHIA – An orexin receptor antagonist – the first drug to affect a neural system that promotes wakefulness – has proven safe and effective for up to 1 year in a pooled analysis of three phase III studies of patients with insomnia.
The drug (suvorexant) significantly improved both subjective and objective measures of sleep, including sleep onset, total sleep time, and wakefulness after sleep, Dr. W. Joseph Herring said at the annual meeting of the American Academy of Neurology.
Based on these data, the manufacturer, Merck, proceeded last year to preliminary talks with the Food and Drug Administration. The committee considering suvorexant recommended that Merck focus on the smaller of two proposed dose ranges – 20 mg for people up to 64 years of age, and 15 mg for those aged 65 years and older.
Although the proposed higher doses of 40 mg for younger patients and 30 mg for older patients, were deemed safe and effective, the lower dose recommendation by the FDA is consistent with its "usual recommendations to go with lower doses of sleep medications," said Dr. Herring, Merck’s executive director of neuroscience clinical research. "So this 15/20-mg regimen is the most likely to be clinically relevant to prescribers."
The orexin neural system was discovered in the late 1990s, Dr. Herring noted. Orexin neurons release two neuropeptides that interact with downstream receptors that promote wakefulness. Secretion follows a circadian rhythm. Suvorexant orexin antagonists block the activity of this wake-signaling system, allowing sleep to occur.
So far, studies have investigated the drug in almost 3,000 patients; 160 of these were treated for at least a year. The three phase III trials comprised more than 275,000 exposure nights. Most of the patients were women; 46% of the patients were aged 65 years or older.
Sleep was measured by subjective assessment and objective scales, including polysomnography.
In the two efficacy studies, the higher doses decreased time to persistent sleep by 15 minutes on the first night; the lower doses did so by 10 minutes. By 3 months, the high-dose group’s decreased time to persistent sleep was 5 minutes; the low dose group’s time was lowered to 4 minutes.
On the first night, the high-dose drug reduced wakefulness after sleep by 40 minutes; the low-dose drug did so by 35 minutes. After 3 months, the reductions were similar (about 25 minutes).
When the night was divided into thirds, both doses decreased wakefulness significantly and about equally, especially in the second and third fractions of the night.
In the subgroup of 160 patients who were treated for at least 12 months, the drugs showed persistent efficacy overall, although the high doses were somewhat more effective, Dr. Herring said.
A multivariate regression analysis found that, compared with placebo, patients who took the high dose were 2.4 times more likely to be considered responders at 1 month and 1.8 times more likely to be responders at 3 months. Those who took the low dose were 1.8 times more likely to respond at 1 and 3 months.
"The drugs really proved about equal in the chances of a good response," said Dr. Herring.
Although there were more adverse events in the active groups than in the placebo groups – and more in the high-dose groups, compared with the low-dose groups – suvorexant was considered safe, he said. The discontinuation rates for drug-related adverse events in the high-dose, low-dose, and placebo groups were 4%, 3%, and 2%, respectively.
The most common issues were next-day somnolence (3% for placebo, 7% with the low dose, and 11% for the high dose, respectively); headache (6%, 7%, and 7%, respectively); and fatigue (2%, 2%, and 4%, respectively).
Few serious adverse events were reported. These included one case of suicidal ideation each in the placebo and low-dose groups, and eight cases in the high-dose group. Dr. Herring said these were mild to moderate and of short duration.
There were no cases of falls or cataplexy, although two patients taking the high dose showed complex sleep behaviors.
Neither of the doses was associated with withdrawal symptoms or clinically significant insomnia rebound during the washout periods. "The drug appears to have a low potential for abuse," Dr. Herring noted.
Merck sponsored the studies. Dr. Herring is a full-time employee of the company.
On Twitter @alz_gal
AT THE AAN 2014 ANNUAL MEETING
Key clinical point: Orexin antagonist suvorexant improved sleep based on both subjective and objective measures, with minimal side effects.
Major finding: In patients with insomnia, suvorexant reduced the time to sleep onset by up to 10 minutes, and wakefulness after sleep by up to 40 minutes.
Data source: Three phase III studies involving approximately 3,000 patients.
Disclosures: Dr. Herring is the director of neuroscience clinical research at Merck, which sponsored the studies.
Simple screening tool improves headache management in primary care
PHILADELPHIA – A simple, three-item screening tool significantly increased headache diagnoses and triptan prescriptions, and significantly decreased opioid prescriptions, based on data from a 5-week intervention at two primary care clinics.
The screen can be administered by a medical assistant and is highly accurate for diagnosing migraine, Dr. Kevin Brennan said at the annual meeting of the American Academy of Neurology.
Of more than 16,000 patients with headache who were referred to a specialty headache clinic from primary care, up to 65% were already taking opioid pain medications, while as few as 10% were taking triptans, said Dr. Brennan, director of the Headache Physiology Laboratory at the University of Utah, Salt Lake City. The problem is inaccurate diagnosis and coding, he said.
"The predominant code for these referred patients was 784, which is a symptom code for headache, and provides no diagnostic utility. If you don’t diagnose, there is a much lower likelihood of appropriately treating," he explained.
The university’s headache clinic receives most of its patients from 10 primary care practices in the region. Over a 2-year period (2008-2010), there were more than 50,000 headache visits in these practices. Of these, approximately 17,000 arrived at the specialty clinic with an actual headache diagnosis, Dr. Brennan said.
The numbers were revealed after an anecdotal observation that many patients were arriving for specialty treatment with a symptom code instead of a diagnosis code, and already taking inappropriate medications.
Dr. Brennan and his colleagues launched a quality improvement project to attempt to boost both appropriate diagnoses and appropriate treatments.
About a third of the referred patients were taking opiates at the time of referral. Other inappropriate medications were antianxiety drugs, barbiturates, antidepressants, antiemetics, nonsteroidal anti-inflammatories, benzodiazepines, decongestants, and muscle relaxants. Only 9% of the referred patients were taking a triptan.
"We wanted to see why we were getting so many of these patients," Dr. Brennan said. "And when we started looking at the medications and diagnostic codes, our initial reaction was ... something I can’t say."
The data showed that headache simply was not being diagnosed. Instead, the most common code was the one that identifies headache as a symptom, Dr. Brennan explained.
To address the problem, the headache research team implemented an easy migraine screen into the patients’ electronic medical records. The ID Migraine algorithm is a three-item screen that can be administered by a medical assistant. It showed 81% sensitivity and 75% specificity for migraine in a validation study.
If a patient answers yes to the three questions (headache disability, nausea, and sensitivity to light), the likelihood of migraine is very, very high, Dr. Brennan said. If the screen is positive, a treatment and medication algorithm is displayed.
The screen was piloted at two clinics during a 5-week period in 2013. The clinics were chosen because they had the best rates of headache diagnostic coding. Prior to the intervention, 41% of the codes were the 784 headache symptom code, 17% of patients received a triptan, and 18% of patients received an opioid.
Five weeks later, the symptom code was reduced to 33% of headache encounters – a 20% improvement. There was a 36% relative increase in the number of triptan prescriptions and a 37% decrease in the number of opioid prescriptions.
The underdiagnosis of headache at the primary care level boils down to one thing, Dr. Brennan said: Time – or the lack of it.
"There’s not enough time in medical school to learn about headache," he said, noting that doctors in training spend an average of 3 hours on headache during an entire school career. Neither do most primary care doctors have enough time during a typical office visit to address headaches. And the idea remains entrenched that headache patients are "difficult" and that migraine is hard to diagnose, he added.
"Right now, we’re herding cats," Dr. Brennan said. "We’re trying to work with the medical directors and get them to implement [screening]. But they still come back saying, ‘Migraine is so hard to diagnose.’ With this system, it’s not hard," he said. "Headache is primary care. It’s more common than asthma and diabetes combined. There’s no excuse for not diagnosing it."
There was no outside funding for the study. Dr. Brennan had no financial disclosures.
PHILADELPHIA – A simple, three-item screening tool significantly increased headache diagnoses and triptan prescriptions, and significantly decreased opioid prescriptions, based on data from a 5-week intervention at two primary care clinics.
The screen can be administered by a medical assistant and is highly accurate for diagnosing migraine, Dr. Kevin Brennan said at the annual meeting of the American Academy of Neurology.
Of more than 16,000 patients with headache who were referred to a specialty headache clinic from primary care, up to 65% were already taking opioid pain medications, while as few as 10% were taking triptans, said Dr. Brennan, director of the Headache Physiology Laboratory at the University of Utah, Salt Lake City. The problem is inaccurate diagnosis and coding, he said.
"The predominant code for these referred patients was 784, which is a symptom code for headache, and provides no diagnostic utility. If you don’t diagnose, there is a much lower likelihood of appropriately treating," he explained.
The university’s headache clinic receives most of its patients from 10 primary care practices in the region. Over a 2-year period (2008-2010), there were more than 50,000 headache visits in these practices. Of these, approximately 17,000 arrived at the specialty clinic with an actual headache diagnosis, Dr. Brennan said.
The numbers were revealed after an anecdotal observation that many patients were arriving for specialty treatment with a symptom code instead of a diagnosis code, and already taking inappropriate medications.
Dr. Brennan and his colleagues launched a quality improvement project to attempt to boost both appropriate diagnoses and appropriate treatments.
About a third of the referred patients were taking opiates at the time of referral. Other inappropriate medications were antianxiety drugs, barbiturates, antidepressants, antiemetics, nonsteroidal anti-inflammatories, benzodiazepines, decongestants, and muscle relaxants. Only 9% of the referred patients were taking a triptan.
"We wanted to see why we were getting so many of these patients," Dr. Brennan said. "And when we started looking at the medications and diagnostic codes, our initial reaction was ... something I can’t say."
The data showed that headache simply was not being diagnosed. Instead, the most common code was the one that identifies headache as a symptom, Dr. Brennan explained.
To address the problem, the headache research team implemented an easy migraine screen into the patients’ electronic medical records. The ID Migraine algorithm is a three-item screen that can be administered by a medical assistant. It showed 81% sensitivity and 75% specificity for migraine in a validation study.
If a patient answers yes to the three questions (headache disability, nausea, and sensitivity to light), the likelihood of migraine is very, very high, Dr. Brennan said. If the screen is positive, a treatment and medication algorithm is displayed.
The screen was piloted at two clinics during a 5-week period in 2013. The clinics were chosen because they had the best rates of headache diagnostic coding. Prior to the intervention, 41% of the codes were the 784 headache symptom code, 17% of patients received a triptan, and 18% of patients received an opioid.
Five weeks later, the symptom code was reduced to 33% of headache encounters – a 20% improvement. There was a 36% relative increase in the number of triptan prescriptions and a 37% decrease in the number of opioid prescriptions.
The underdiagnosis of headache at the primary care level boils down to one thing, Dr. Brennan said: Time – or the lack of it.
"There’s not enough time in medical school to learn about headache," he said, noting that doctors in training spend an average of 3 hours on headache during an entire school career. Neither do most primary care doctors have enough time during a typical office visit to address headaches. And the idea remains entrenched that headache patients are "difficult" and that migraine is hard to diagnose, he added.
"Right now, we’re herding cats," Dr. Brennan said. "We’re trying to work with the medical directors and get them to implement [screening]. But they still come back saying, ‘Migraine is so hard to diagnose.’ With this system, it’s not hard," he said. "Headache is primary care. It’s more common than asthma and diabetes combined. There’s no excuse for not diagnosing it."
There was no outside funding for the study. Dr. Brennan had no financial disclosures.
PHILADELPHIA – A simple, three-item screening tool significantly increased headache diagnoses and triptan prescriptions, and significantly decreased opioid prescriptions, based on data from a 5-week intervention at two primary care clinics.
The screen can be administered by a medical assistant and is highly accurate for diagnosing migraine, Dr. Kevin Brennan said at the annual meeting of the American Academy of Neurology.
Of more than 16,000 patients with headache who were referred to a specialty headache clinic from primary care, up to 65% were already taking opioid pain medications, while as few as 10% were taking triptans, said Dr. Brennan, director of the Headache Physiology Laboratory at the University of Utah, Salt Lake City. The problem is inaccurate diagnosis and coding, he said.
"The predominant code for these referred patients was 784, which is a symptom code for headache, and provides no diagnostic utility. If you don’t diagnose, there is a much lower likelihood of appropriately treating," he explained.
The university’s headache clinic receives most of its patients from 10 primary care practices in the region. Over a 2-year period (2008-2010), there were more than 50,000 headache visits in these practices. Of these, approximately 17,000 arrived at the specialty clinic with an actual headache diagnosis, Dr. Brennan said.
The numbers were revealed after an anecdotal observation that many patients were arriving for specialty treatment with a symptom code instead of a diagnosis code, and already taking inappropriate medications.
Dr. Brennan and his colleagues launched a quality improvement project to attempt to boost both appropriate diagnoses and appropriate treatments.
About a third of the referred patients were taking opiates at the time of referral. Other inappropriate medications were antianxiety drugs, barbiturates, antidepressants, antiemetics, nonsteroidal anti-inflammatories, benzodiazepines, decongestants, and muscle relaxants. Only 9% of the referred patients were taking a triptan.
"We wanted to see why we were getting so many of these patients," Dr. Brennan said. "And when we started looking at the medications and diagnostic codes, our initial reaction was ... something I can’t say."
The data showed that headache simply was not being diagnosed. Instead, the most common code was the one that identifies headache as a symptom, Dr. Brennan explained.
To address the problem, the headache research team implemented an easy migraine screen into the patients’ electronic medical records. The ID Migraine algorithm is a three-item screen that can be administered by a medical assistant. It showed 81% sensitivity and 75% specificity for migraine in a validation study.
If a patient answers yes to the three questions (headache disability, nausea, and sensitivity to light), the likelihood of migraine is very, very high, Dr. Brennan said. If the screen is positive, a treatment and medication algorithm is displayed.
The screen was piloted at two clinics during a 5-week period in 2013. The clinics were chosen because they had the best rates of headache diagnostic coding. Prior to the intervention, 41% of the codes were the 784 headache symptom code, 17% of patients received a triptan, and 18% of patients received an opioid.
Five weeks later, the symptom code was reduced to 33% of headache encounters – a 20% improvement. There was a 36% relative increase in the number of triptan prescriptions and a 37% decrease in the number of opioid prescriptions.
The underdiagnosis of headache at the primary care level boils down to one thing, Dr. Brennan said: Time – or the lack of it.
"There’s not enough time in medical school to learn about headache," he said, noting that doctors in training spend an average of 3 hours on headache during an entire school career. Neither do most primary care doctors have enough time during a typical office visit to address headaches. And the idea remains entrenched that headache patients are "difficult" and that migraine is hard to diagnose, he added.
"Right now, we’re herding cats," Dr. Brennan said. "We’re trying to work with the medical directors and get them to implement [screening]. But they still come back saying, ‘Migraine is so hard to diagnose.’ With this system, it’s not hard," he said. "Headache is primary care. It’s more common than asthma and diabetes combined. There’s no excuse for not diagnosing it."
There was no outside funding for the study. Dr. Brennan had no financial disclosures.
AT THE AAN 2014 ANNUAL MEETING
Key clinical point: Use of a headache screening tool in a primary care setting can reduce the number of incorrect diagnoses and inappropriate medications in patients presenting to a specialty clinic.
Major finding: A simple, three-item headache screen in primary care settings increased triptan prescriptions by 36% and decreased opioid prescriptions by 37% over 5 weeks.
Data source: An intervention project at two primary care clinics.
Disclosures: There was no outside funding for the study. Dr. Brennan had no financial disclosures.
Many migraine patients quit meds by 12 months
PHILADELPHIA - Adherence to migraine prophylactics drops off sharply within a few months of the initial prescription and falls even farther by 1 year, based on claims data from approximately 8,700 patients.
By 6 months, only 30% of patients were adherent to their preventive medications, defined as taking them as directed at least 80% of the time, Zsolt Hepp, Pharm.D., said at the annual meeting of the American Academy of Neurology. By 12 months, adherence dropped to about 20%; fewer than 1 in 5 patients.
"Adherence rates were alarmingly low," said Dr. Hepp, a researcher for Allergan. "And no matter what class of drugs we looked at, the rates were similar."
Dr. Hepp and his colleagues conducted a retrospective analysis using 2007-2012 claims data from the Truven Health Analytics MarketScan Database. The system contains Medicare, supplemental, and Medicaid claims for more 40 million unique patients per year. Patients, who were at least 18 years old, were diagnosed with chronic migraine, and who initiated an oral migraine prophylactic medication between 2008-2012 were included in the analysis.
The initial study group comprised more than 76,000 patients, but exclusion criteria narrowed it to about 8,700, as the researchers tried to pinpoint prescriptions that were solely for migraine. Since most of the 14 medications investigated have different primary indications, the team excluded patients with a diagnosis for that another indication (depression, for example) within a year prior to the migraine diagnosis.
The analysis also included only patients who had full, unlapsed insurance coverage during the year of interest; many in the original cohort were excluded because of such lapses.
The 14 commonly prescribed migraine medications in the study included four recommended by the American Academy of Neurology as first-line preventives.
The drugs examined were antidepressants (nortriptyline and amitriptyline, citalopram, sertraline, fluoxetine, paroxetine, and venlafaxine), beta-blockers (propranolol, metoprolol, atenolol, and nadolol), and anticonvulsants (divalproex, gabapentin, and topiramate).
The majority of patients were women, with a mean age of 40 years. Most (59%) were employed full time. Common comorbidities included nonmigraine headache (54%), cancer (22%), hypertension (18%), depression (18%), sleep disorders (11%), and gastroesophageal reflux disease (11%).
The adherence rates were surprisingly similar across drug classes, Dr. Hepp noted. Overall adherence rates using the Medication Possession Ratio hovered at 28%-29% at 6 months and 19%-21% at 12 months.
When broken down by individual drug, the antidepressants had the highest rates of adherence at 6 months. Venlafaxine and fluoxetine topped the list, with 37% of patients adherent at 6 months. However, by 12 months, adherence had dropped to 28% for venlafaxine and 22% for fluoxetine.
"All of these medications need to be taken at least once daily – and sometimes several times a day – to really maintain their effectiveness," Dr. Hepp noted. "This is the first real-world analysis of the U.S. migraine population, and we feel the results are generalizable to that population."
Unfortunately, he said, a claims database doesn’t give any insight into why patients stop taking their medication. One view came up during the discussion period, however.
"People don’t want to take drugs that make them gain weight or feel stupid," one discussant said bluntly.
Dr. Hepp added that this low adherence was not surprising, in light of research that shows similar problems with other chronic diseases.
A 2009 study examined six different chronic conditions, including hypertension and diabetes, he said. "By far the best adherence rates were for diabetes, and that was only about 60% by 12 months."
Although the claims database revealed nothing about the factors driving the lack of adherence, data from previous studies suggest that side effects and dissatisfaction are likely suspects, Dr. Hepp said. His own previous study on the reasons for discontinuation in clinical trials of three migraine treatments showed that "a significant portion of the discontinued patients decided to stop treatment due to side effects," he said in an interview. I actually have another publication that was a systematic literature review of adherence manuscripts.
"Another study by Blumenfeld et al. 2013 found that top reasons for discontinuation of oral migraine prophyalctics were side effects and treatment satisfaction," he said. "It’s likely that these other studies highlight the very reason(s) why we saw the level of nonadherence in our study," he said. "Our results most certainly call attention to the large gap in the treatment of this highly burdened population, and at the very least this study calls for further research and hopefully the integration of adherence into future clincial trials for migraine prevention tretaments."
Allergan sponsored the study. Dr Hepp is a full-time employee of that company.
On Twitter @alz_gal
While this analysis doesn’t allow us to determine why adherence to the preventive medications was so poor (16% at 1 year), we may hazard some guesses based on our experience with how patients use these medications. Based on my clinical experience, the adverse side effect profile and/or the lack of efficacy are the two biggest reasons [for poor adherence].
Sometimes, the adverse event profile prevents increasing the dose of the medications to levels that may be therapeutic, so we never know if the medication might have been effective. However, it’s worth remembering that the best that the currently available preventive drugs can accomplish is to reduce headache days by half or more in about half the patients who take them.
Finally, this cohort had chronic migraine – that means they were a more severely affected group and perhaps less responsive to preventive medications.
Dr. David Dodick is a neurologist and headache specialist at the Mayo Clinic, Scottsdale, Az. He disclosed financial relationships with multiple pharmaceutical companies.
While this analysis doesn’t allow us to determine why adherence to the preventive medications was so poor (16% at 1 year), we may hazard some guesses based on our experience with how patients use these medications. Based on my clinical experience, the adverse side effect profile and/or the lack of efficacy are the two biggest reasons [for poor adherence].
Sometimes, the adverse event profile prevents increasing the dose of the medications to levels that may be therapeutic, so we never know if the medication might have been effective. However, it’s worth remembering that the best that the currently available preventive drugs can accomplish is to reduce headache days by half or more in about half the patients who take them.
Finally, this cohort had chronic migraine – that means they were a more severely affected group and perhaps less responsive to preventive medications.
Dr. David Dodick is a neurologist and headache specialist at the Mayo Clinic, Scottsdale, Az. He disclosed financial relationships with multiple pharmaceutical companies.
While this analysis doesn’t allow us to determine why adherence to the preventive medications was so poor (16% at 1 year), we may hazard some guesses based on our experience with how patients use these medications. Based on my clinical experience, the adverse side effect profile and/or the lack of efficacy are the two biggest reasons [for poor adherence].
Sometimes, the adverse event profile prevents increasing the dose of the medications to levels that may be therapeutic, so we never know if the medication might have been effective. However, it’s worth remembering that the best that the currently available preventive drugs can accomplish is to reduce headache days by half or more in about half the patients who take them.
Finally, this cohort had chronic migraine – that means they were a more severely affected group and perhaps less responsive to preventive medications.
Dr. David Dodick is a neurologist and headache specialist at the Mayo Clinic, Scottsdale, Az. He disclosed financial relationships with multiple pharmaceutical companies.
PHILADELPHIA - Adherence to migraine prophylactics drops off sharply within a few months of the initial prescription and falls even farther by 1 year, based on claims data from approximately 8,700 patients.
By 6 months, only 30% of patients were adherent to their preventive medications, defined as taking them as directed at least 80% of the time, Zsolt Hepp, Pharm.D., said at the annual meeting of the American Academy of Neurology. By 12 months, adherence dropped to about 20%; fewer than 1 in 5 patients.
"Adherence rates were alarmingly low," said Dr. Hepp, a researcher for Allergan. "And no matter what class of drugs we looked at, the rates were similar."
Dr. Hepp and his colleagues conducted a retrospective analysis using 2007-2012 claims data from the Truven Health Analytics MarketScan Database. The system contains Medicare, supplemental, and Medicaid claims for more 40 million unique patients per year. Patients, who were at least 18 years old, were diagnosed with chronic migraine, and who initiated an oral migraine prophylactic medication between 2008-2012 were included in the analysis.
The initial study group comprised more than 76,000 patients, but exclusion criteria narrowed it to about 8,700, as the researchers tried to pinpoint prescriptions that were solely for migraine. Since most of the 14 medications investigated have different primary indications, the team excluded patients with a diagnosis for that another indication (depression, for example) within a year prior to the migraine diagnosis.
The analysis also included only patients who had full, unlapsed insurance coverage during the year of interest; many in the original cohort were excluded because of such lapses.
The 14 commonly prescribed migraine medications in the study included four recommended by the American Academy of Neurology as first-line preventives.
The drugs examined were antidepressants (nortriptyline and amitriptyline, citalopram, sertraline, fluoxetine, paroxetine, and venlafaxine), beta-blockers (propranolol, metoprolol, atenolol, and nadolol), and anticonvulsants (divalproex, gabapentin, and topiramate).
The majority of patients were women, with a mean age of 40 years. Most (59%) were employed full time. Common comorbidities included nonmigraine headache (54%), cancer (22%), hypertension (18%), depression (18%), sleep disorders (11%), and gastroesophageal reflux disease (11%).
The adherence rates were surprisingly similar across drug classes, Dr. Hepp noted. Overall adherence rates using the Medication Possession Ratio hovered at 28%-29% at 6 months and 19%-21% at 12 months.
When broken down by individual drug, the antidepressants had the highest rates of adherence at 6 months. Venlafaxine and fluoxetine topped the list, with 37% of patients adherent at 6 months. However, by 12 months, adherence had dropped to 28% for venlafaxine and 22% for fluoxetine.
"All of these medications need to be taken at least once daily – and sometimes several times a day – to really maintain their effectiveness," Dr. Hepp noted. "This is the first real-world analysis of the U.S. migraine population, and we feel the results are generalizable to that population."
Unfortunately, he said, a claims database doesn’t give any insight into why patients stop taking their medication. One view came up during the discussion period, however.
"People don’t want to take drugs that make them gain weight or feel stupid," one discussant said bluntly.
Dr. Hepp added that this low adherence was not surprising, in light of research that shows similar problems with other chronic diseases.
A 2009 study examined six different chronic conditions, including hypertension and diabetes, he said. "By far the best adherence rates were for diabetes, and that was only about 60% by 12 months."
Although the claims database revealed nothing about the factors driving the lack of adherence, data from previous studies suggest that side effects and dissatisfaction are likely suspects, Dr. Hepp said. His own previous study on the reasons for discontinuation in clinical trials of three migraine treatments showed that "a significant portion of the discontinued patients decided to stop treatment due to side effects," he said in an interview. I actually have another publication that was a systematic literature review of adherence manuscripts.
"Another study by Blumenfeld et al. 2013 found that top reasons for discontinuation of oral migraine prophyalctics were side effects and treatment satisfaction," he said. "It’s likely that these other studies highlight the very reason(s) why we saw the level of nonadherence in our study," he said. "Our results most certainly call attention to the large gap in the treatment of this highly burdened population, and at the very least this study calls for further research and hopefully the integration of adherence into future clincial trials for migraine prevention tretaments."
Allergan sponsored the study. Dr Hepp is a full-time employee of that company.
On Twitter @alz_gal
PHILADELPHIA - Adherence to migraine prophylactics drops off sharply within a few months of the initial prescription and falls even farther by 1 year, based on claims data from approximately 8,700 patients.
By 6 months, only 30% of patients were adherent to their preventive medications, defined as taking them as directed at least 80% of the time, Zsolt Hepp, Pharm.D., said at the annual meeting of the American Academy of Neurology. By 12 months, adherence dropped to about 20%; fewer than 1 in 5 patients.
"Adherence rates were alarmingly low," said Dr. Hepp, a researcher for Allergan. "And no matter what class of drugs we looked at, the rates were similar."
Dr. Hepp and his colleagues conducted a retrospective analysis using 2007-2012 claims data from the Truven Health Analytics MarketScan Database. The system contains Medicare, supplemental, and Medicaid claims for more 40 million unique patients per year. Patients, who were at least 18 years old, were diagnosed with chronic migraine, and who initiated an oral migraine prophylactic medication between 2008-2012 were included in the analysis.
The initial study group comprised more than 76,000 patients, but exclusion criteria narrowed it to about 8,700, as the researchers tried to pinpoint prescriptions that were solely for migraine. Since most of the 14 medications investigated have different primary indications, the team excluded patients with a diagnosis for that another indication (depression, for example) within a year prior to the migraine diagnosis.
The analysis also included only patients who had full, unlapsed insurance coverage during the year of interest; many in the original cohort were excluded because of such lapses.
The 14 commonly prescribed migraine medications in the study included four recommended by the American Academy of Neurology as first-line preventives.
The drugs examined were antidepressants (nortriptyline and amitriptyline, citalopram, sertraline, fluoxetine, paroxetine, and venlafaxine), beta-blockers (propranolol, metoprolol, atenolol, and nadolol), and anticonvulsants (divalproex, gabapentin, and topiramate).
The majority of patients were women, with a mean age of 40 years. Most (59%) were employed full time. Common comorbidities included nonmigraine headache (54%), cancer (22%), hypertension (18%), depression (18%), sleep disorders (11%), and gastroesophageal reflux disease (11%).
The adherence rates were surprisingly similar across drug classes, Dr. Hepp noted. Overall adherence rates using the Medication Possession Ratio hovered at 28%-29% at 6 months and 19%-21% at 12 months.
When broken down by individual drug, the antidepressants had the highest rates of adherence at 6 months. Venlafaxine and fluoxetine topped the list, with 37% of patients adherent at 6 months. However, by 12 months, adherence had dropped to 28% for venlafaxine and 22% for fluoxetine.
"All of these medications need to be taken at least once daily – and sometimes several times a day – to really maintain their effectiveness," Dr. Hepp noted. "This is the first real-world analysis of the U.S. migraine population, and we feel the results are generalizable to that population."
Unfortunately, he said, a claims database doesn’t give any insight into why patients stop taking their medication. One view came up during the discussion period, however.
"People don’t want to take drugs that make them gain weight or feel stupid," one discussant said bluntly.
Dr. Hepp added that this low adherence was not surprising, in light of research that shows similar problems with other chronic diseases.
A 2009 study examined six different chronic conditions, including hypertension and diabetes, he said. "By far the best adherence rates were for diabetes, and that was only about 60% by 12 months."
Although the claims database revealed nothing about the factors driving the lack of adherence, data from previous studies suggest that side effects and dissatisfaction are likely suspects, Dr. Hepp said. His own previous study on the reasons for discontinuation in clinical trials of three migraine treatments showed that "a significant portion of the discontinued patients decided to stop treatment due to side effects," he said in an interview. I actually have another publication that was a systematic literature review of adherence manuscripts.
"Another study by Blumenfeld et al. 2013 found that top reasons for discontinuation of oral migraine prophyalctics were side effects and treatment satisfaction," he said. "It’s likely that these other studies highlight the very reason(s) why we saw the level of nonadherence in our study," he said. "Our results most certainly call attention to the large gap in the treatment of this highly burdened population, and at the very least this study calls for further research and hopefully the integration of adherence into future clincial trials for migraine prevention tretaments."
Allergan sponsored the study. Dr Hepp is a full-time employee of that company.
On Twitter @alz_gal
AT THE AAN 2014 ANNUAL MEETING
Key clinical point: This study is a real-world analysis of migraine patients in the United States., with generalizable results, according to the researchers.
Major finding: Adherence to migraine preventives dropped to about 30% by 12 months, and 20% by 6 months.
Data source: A retrospective study of approximately 8,700 patients.
Disclosures: Dr. Hepp is a full-time employee of Allergan, which sponsored the study.
Telemedicine gives small hospitals the biggest boost in rTPA use for stroke
PHILADELPHIA – A telemedicine program for patients with acute ischemic stroke increased the use of recombinant tissue plasminogen activator by as much as 13% in the year after implementation.
Most of the 13 hospitals in the retrospective study significantly increased their recombinant tissue plasminogen activator (rTPA) use, Dr. Jeffrey C. Wagner said at the annual meeting of the American Academy of Neurology. Three didn’t benefit, however, and one hospital actually experienced a significant decrease in rTPA administration.
rTPA "is the only treatment proven to benefit patients during acute ischemic stroke," said Dr. Wagner, a stroke specialist at Blue Sky Neurology in Denver.
"We obviously need more research to discover why these hospitals didn’t benefit more dramatically," he said.
The study population included patients aged 18 years and older who were admitted with a primary diagnosis of acute ischemic stroke and discharged between July 1, 2006, and Dec. 31, 2012; more than 15,000 patients were included in the retrospective analysis. Outcomes were extracted from a national claims database, and the hospitals had a minimum of 1 year of data available before and after implementing a telemedicine program. The hospitals represented a variety of patient demographics. About two-thirds were rural; approximately half were small, defined as fewer than 200 beds. The hospitals were located in the Northeastern, Southern, and Western portions of the United States.
The primary outcome was the percentage of patients treated with rTPA before and after telemedicine implementation.
Overall, rTPA administration increased significantly, from 4.5% to 7.3% after a telemedicine program was introduced. The increase was similar whether patients were admitted to the hospital or whether they were transferred to a stroke center.
There were, however, significant outcome differences among the hospitals according to size. Overall the use of rTPA in smaller hospitals increased from 1% to 7% after implementing a telemedicine program, compared with an increase from 5.4% to 7% in larger hospitals. Those results were similar when patients were stratified as inpatients or transferred patients.
Two hospitals increased rTPA use by more than 10%; five hospitals increased rTPA use by 5%-10%, and three hospitals increased rTPA use by 0-5%.
However, the rTPA use in three hospitals remained virtually identical; two administered no rTPA either before or after implementing a telemedicine program, and rTPA use in the third hospital increase from none to less than 2%. In addition, one of the largest hospitals in the study actually saw a 14% decrease in rTPA use, a finding Dr. Wagner was hard pressed to explain.
"In my experience, there’s a huge integrated system required for telemedicine to work well, from the EMS [emergency medical services] people calling in a stroke alert in advance to the emergency doctors activating the telemedicine system. My theory would be that there might not have been complete buy-in from the staff."
The three hospitals that didn’t experience any benefit were the smallest in the cohort – a factor that probably contributed to their lack of results, he added.
"My theory here is that they spanned the largest area of geography, so having telemedicine might not improve rTPA usage, because the real issue was not being able to get patients there in time," he said.
The study was funded by Genentech. Dr. Wagner had no additional disclosures.
On Twitter @alz_gal
PHILADELPHIA – A telemedicine program for patients with acute ischemic stroke increased the use of recombinant tissue plasminogen activator by as much as 13% in the year after implementation.
Most of the 13 hospitals in the retrospective study significantly increased their recombinant tissue plasminogen activator (rTPA) use, Dr. Jeffrey C. Wagner said at the annual meeting of the American Academy of Neurology. Three didn’t benefit, however, and one hospital actually experienced a significant decrease in rTPA administration.
rTPA "is the only treatment proven to benefit patients during acute ischemic stroke," said Dr. Wagner, a stroke specialist at Blue Sky Neurology in Denver.
"We obviously need more research to discover why these hospitals didn’t benefit more dramatically," he said.
The study population included patients aged 18 years and older who were admitted with a primary diagnosis of acute ischemic stroke and discharged between July 1, 2006, and Dec. 31, 2012; more than 15,000 patients were included in the retrospective analysis. Outcomes were extracted from a national claims database, and the hospitals had a minimum of 1 year of data available before and after implementing a telemedicine program. The hospitals represented a variety of patient demographics. About two-thirds were rural; approximately half were small, defined as fewer than 200 beds. The hospitals were located in the Northeastern, Southern, and Western portions of the United States.
The primary outcome was the percentage of patients treated with rTPA before and after telemedicine implementation.
Overall, rTPA administration increased significantly, from 4.5% to 7.3% after a telemedicine program was introduced. The increase was similar whether patients were admitted to the hospital or whether they were transferred to a stroke center.
There were, however, significant outcome differences among the hospitals according to size. Overall the use of rTPA in smaller hospitals increased from 1% to 7% after implementing a telemedicine program, compared with an increase from 5.4% to 7% in larger hospitals. Those results were similar when patients were stratified as inpatients or transferred patients.
Two hospitals increased rTPA use by more than 10%; five hospitals increased rTPA use by 5%-10%, and three hospitals increased rTPA use by 0-5%.
However, the rTPA use in three hospitals remained virtually identical; two administered no rTPA either before or after implementing a telemedicine program, and rTPA use in the third hospital increase from none to less than 2%. In addition, one of the largest hospitals in the study actually saw a 14% decrease in rTPA use, a finding Dr. Wagner was hard pressed to explain.
"In my experience, there’s a huge integrated system required for telemedicine to work well, from the EMS [emergency medical services] people calling in a stroke alert in advance to the emergency doctors activating the telemedicine system. My theory would be that there might not have been complete buy-in from the staff."
The three hospitals that didn’t experience any benefit were the smallest in the cohort – a factor that probably contributed to their lack of results, he added.
"My theory here is that they spanned the largest area of geography, so having telemedicine might not improve rTPA usage, because the real issue was not being able to get patients there in time," he said.
The study was funded by Genentech. Dr. Wagner had no additional disclosures.
On Twitter @alz_gal
PHILADELPHIA – A telemedicine program for patients with acute ischemic stroke increased the use of recombinant tissue plasminogen activator by as much as 13% in the year after implementation.
Most of the 13 hospitals in the retrospective study significantly increased their recombinant tissue plasminogen activator (rTPA) use, Dr. Jeffrey C. Wagner said at the annual meeting of the American Academy of Neurology. Three didn’t benefit, however, and one hospital actually experienced a significant decrease in rTPA administration.
rTPA "is the only treatment proven to benefit patients during acute ischemic stroke," said Dr. Wagner, a stroke specialist at Blue Sky Neurology in Denver.
"We obviously need more research to discover why these hospitals didn’t benefit more dramatically," he said.
The study population included patients aged 18 years and older who were admitted with a primary diagnosis of acute ischemic stroke and discharged between July 1, 2006, and Dec. 31, 2012; more than 15,000 patients were included in the retrospective analysis. Outcomes were extracted from a national claims database, and the hospitals had a minimum of 1 year of data available before and after implementing a telemedicine program. The hospitals represented a variety of patient demographics. About two-thirds were rural; approximately half were small, defined as fewer than 200 beds. The hospitals were located in the Northeastern, Southern, and Western portions of the United States.
The primary outcome was the percentage of patients treated with rTPA before and after telemedicine implementation.
Overall, rTPA administration increased significantly, from 4.5% to 7.3% after a telemedicine program was introduced. The increase was similar whether patients were admitted to the hospital or whether they were transferred to a stroke center.
There were, however, significant outcome differences among the hospitals according to size. Overall the use of rTPA in smaller hospitals increased from 1% to 7% after implementing a telemedicine program, compared with an increase from 5.4% to 7% in larger hospitals. Those results were similar when patients were stratified as inpatients or transferred patients.
Two hospitals increased rTPA use by more than 10%; five hospitals increased rTPA use by 5%-10%, and three hospitals increased rTPA use by 0-5%.
However, the rTPA use in three hospitals remained virtually identical; two administered no rTPA either before or after implementing a telemedicine program, and rTPA use in the third hospital increase from none to less than 2%. In addition, one of the largest hospitals in the study actually saw a 14% decrease in rTPA use, a finding Dr. Wagner was hard pressed to explain.
"In my experience, there’s a huge integrated system required for telemedicine to work well, from the EMS [emergency medical services] people calling in a stroke alert in advance to the emergency doctors activating the telemedicine system. My theory would be that there might not have been complete buy-in from the staff."
The three hospitals that didn’t experience any benefit were the smallest in the cohort – a factor that probably contributed to their lack of results, he added.
"My theory here is that they spanned the largest area of geography, so having telemedicine might not improve rTPA usage, because the real issue was not being able to get patients there in time," he said.
The study was funded by Genentech. Dr. Wagner had no additional disclosures.
On Twitter @alz_gal
AT THE AAN 2014 ANNUAL MEETING
Major finding: Telemedicine programs increased rTPA usage by up to 13% during the year after implementation.
Data source: The retrospective study comprised 13 hospitals and approximately 15,000 patients.
Disclosures: The study was funded by Genentech. Dr. Wagner had no additional financial disclosures.
Young children with OCD respond to family-based therapy
A family-based program of cognitive-behavioral therapy eased symptoms of obsessive-compulsive disorder in young children significantly better than did a traditional relaxation-based treatment.
Almost three-fourths of the youngsters who underwent the family-based therapy were "very much" or "much" improved after 14 weeks of treatment, Jennifer Freeman, Ph.D., and her colleagues wrote in a recently published report (JAMA Psychiatry 2014 April 23 [doi:10.1001/jamapsychiatry.2014.170]).
The number needed to treat to achieve one additional responder over the relaxation-based program was about three, wrote Dr. Freeman, codirector of the pediatric anxiety research clinic at Bradley-Hasbro Children’s Research Center, Providence, R.I., and her coauthors.
The Pediatric Obsessive-Compulsive Disorder Treatment Study for Young Children (POTS Jr.) study randomized 127 children aged 5-8 years to either the family-based or relaxation-based treatments. More than 90% of the participants were white, 1.6% were black, 2.4% were Asian, 3.1% were mixed race, and 3.1% were unreported. Meanwhile, 95.3% were not Hispanic, and 4.7% were Hispanic. The participants’ families’ annual income range was $70,000-$80,000.
The family-based treatment was made up of four components: "g1) psychoeducation; 2) behavior management skills training (parent tools); 3) externalizing OCD and [exposure plus response prevention] (child tools); and 4) family process components."h for both parents and children. Parents learn about different ways to manage their child’s OCD behaviors – ignoring and rewords for example – as well as modeling positive behavior and helping the child learn self-regulation through exposure and response prevention. The cognitive-behavioral skills are simplified to improve the child’s ability to understand and employ them.
The relaxation-based program focuses on stress reduction and on teaching children how to recognize anxiety and other negative feelings, as well as positive feelings.
Assessments were conducted at baseline and 5, 9, and 14 weeks with a symptoms scale and the Clinical Global Impressions Severity and Improvement scales.
Children were a mean of 7 years old and had a mean 2-year duration of symptoms. By the end of the study, 72% of those in the family-based group were much or very much improved, compared with 41% of those in the relaxation group – a statistically significant difference. An effects size analysis determined that the difference also was clinically significant.
Dr. Freeman and her associates cited several limitations. For example, the enrollment of children from nonwhite and non–English speaking families was limited, which raises questions about whether the conclusions are generalizable, they said. "gSimilarly, the lack of socioeconomic diversity in the sample is a concern,"h they wrote.
Because the program is individually tailored to a child’s unique developmental level, it introduces the idea that treatment for obsessive-compulsive disorder can begin early, possibly interrupting symptom acceleration, the author said.
"With appropriate parental support, young children with OCD who undergo family-based [therapy] can make significant gains beyond what can be expected from having parents attempt to teach relaxation strategies to their children with OCD. Hopefully, these gains in overall development can decrease the chronicity and morbidity of this debilitating illness."h
The study was supported by several universities and the National Institute of Mental Health. Dr. Freeman had no financial disclosures, but one coauthor reported financial relationships with numerous pharmaceutical companies.
On Twitter @alz_gal
A family-based program of cognitive-behavioral therapy eased symptoms of obsessive-compulsive disorder in young children significantly better than did a traditional relaxation-based treatment.
Almost three-fourths of the youngsters who underwent the family-based therapy were "very much" or "much" improved after 14 weeks of treatment, Jennifer Freeman, Ph.D., and her colleagues wrote in a recently published report (JAMA Psychiatry 2014 April 23 [doi:10.1001/jamapsychiatry.2014.170]).
The number needed to treat to achieve one additional responder over the relaxation-based program was about three, wrote Dr. Freeman, codirector of the pediatric anxiety research clinic at Bradley-Hasbro Children’s Research Center, Providence, R.I., and her coauthors.
The Pediatric Obsessive-Compulsive Disorder Treatment Study for Young Children (POTS Jr.) study randomized 127 children aged 5-8 years to either the family-based or relaxation-based treatments. More than 90% of the participants were white, 1.6% were black, 2.4% were Asian, 3.1% were mixed race, and 3.1% were unreported. Meanwhile, 95.3% were not Hispanic, and 4.7% were Hispanic. The participants’ families’ annual income range was $70,000-$80,000.
The family-based treatment was made up of four components: "g1) psychoeducation; 2) behavior management skills training (parent tools); 3) externalizing OCD and [exposure plus response prevention] (child tools); and 4) family process components."h for both parents and children. Parents learn about different ways to manage their child’s OCD behaviors – ignoring and rewords for example – as well as modeling positive behavior and helping the child learn self-regulation through exposure and response prevention. The cognitive-behavioral skills are simplified to improve the child’s ability to understand and employ them.
The relaxation-based program focuses on stress reduction and on teaching children how to recognize anxiety and other negative feelings, as well as positive feelings.
Assessments were conducted at baseline and 5, 9, and 14 weeks with a symptoms scale and the Clinical Global Impressions Severity and Improvement scales.
Children were a mean of 7 years old and had a mean 2-year duration of symptoms. By the end of the study, 72% of those in the family-based group were much or very much improved, compared with 41% of those in the relaxation group – a statistically significant difference. An effects size analysis determined that the difference also was clinically significant.
Dr. Freeman and her associates cited several limitations. For example, the enrollment of children from nonwhite and non–English speaking families was limited, which raises questions about whether the conclusions are generalizable, they said. "gSimilarly, the lack of socioeconomic diversity in the sample is a concern,"h they wrote.
Because the program is individually tailored to a child’s unique developmental level, it introduces the idea that treatment for obsessive-compulsive disorder can begin early, possibly interrupting symptom acceleration, the author said.
"With appropriate parental support, young children with OCD who undergo family-based [therapy] can make significant gains beyond what can be expected from having parents attempt to teach relaxation strategies to their children with OCD. Hopefully, these gains in overall development can decrease the chronicity and morbidity of this debilitating illness."h
The study was supported by several universities and the National Institute of Mental Health. Dr. Freeman had no financial disclosures, but one coauthor reported financial relationships with numerous pharmaceutical companies.
On Twitter @alz_gal
A family-based program of cognitive-behavioral therapy eased symptoms of obsessive-compulsive disorder in young children significantly better than did a traditional relaxation-based treatment.
Almost three-fourths of the youngsters who underwent the family-based therapy were "very much" or "much" improved after 14 weeks of treatment, Jennifer Freeman, Ph.D., and her colleagues wrote in a recently published report (JAMA Psychiatry 2014 April 23 [doi:10.1001/jamapsychiatry.2014.170]).
The number needed to treat to achieve one additional responder over the relaxation-based program was about three, wrote Dr. Freeman, codirector of the pediatric anxiety research clinic at Bradley-Hasbro Children’s Research Center, Providence, R.I., and her coauthors.
The Pediatric Obsessive-Compulsive Disorder Treatment Study for Young Children (POTS Jr.) study randomized 127 children aged 5-8 years to either the family-based or relaxation-based treatments. More than 90% of the participants were white, 1.6% were black, 2.4% were Asian, 3.1% were mixed race, and 3.1% were unreported. Meanwhile, 95.3% were not Hispanic, and 4.7% were Hispanic. The participants’ families’ annual income range was $70,000-$80,000.
The family-based treatment was made up of four components: "g1) psychoeducation; 2) behavior management skills training (parent tools); 3) externalizing OCD and [exposure plus response prevention] (child tools); and 4) family process components."h for both parents and children. Parents learn about different ways to manage their child’s OCD behaviors – ignoring and rewords for example – as well as modeling positive behavior and helping the child learn self-regulation through exposure and response prevention. The cognitive-behavioral skills are simplified to improve the child’s ability to understand and employ them.
The relaxation-based program focuses on stress reduction and on teaching children how to recognize anxiety and other negative feelings, as well as positive feelings.
Assessments were conducted at baseline and 5, 9, and 14 weeks with a symptoms scale and the Clinical Global Impressions Severity and Improvement scales.
Children were a mean of 7 years old and had a mean 2-year duration of symptoms. By the end of the study, 72% of those in the family-based group were much or very much improved, compared with 41% of those in the relaxation group – a statistically significant difference. An effects size analysis determined that the difference also was clinically significant.
Dr. Freeman and her associates cited several limitations. For example, the enrollment of children from nonwhite and non–English speaking families was limited, which raises questions about whether the conclusions are generalizable, they said. "gSimilarly, the lack of socioeconomic diversity in the sample is a concern,"h they wrote.
Because the program is individually tailored to a child’s unique developmental level, it introduces the idea that treatment for obsessive-compulsive disorder can begin early, possibly interrupting symptom acceleration, the author said.
"With appropriate parental support, young children with OCD who undergo family-based [therapy] can make significant gains beyond what can be expected from having parents attempt to teach relaxation strategies to their children with OCD. Hopefully, these gains in overall development can decrease the chronicity and morbidity of this debilitating illness."h
The study was supported by several universities and the National Institute of Mental Health. Dr. Freeman had no financial disclosures, but one coauthor reported financial relationships with numerous pharmaceutical companies.
On Twitter @alz_gal
FROM JAMA PSYCHIATRY
Key clinical point: Families are key in helping young children with OCD experience less-intense symptoms.
Major finding: A family-based treatment for pediatric OCD affected improvement in 72% of children, compared to 41% who improved with a traditional relaxation-based treatment.
Data source: The randomized trial comprised 127 children aged 5-8 years.
Disclosures: The study was supported by several universities and the National Institute of Mental Health. Dr. Freeman had no financial disclosures, but one co-author reported financial relationships with numerous pharmaceutical companies.
B-cell inhibitor might prevent new brain lesions in multiple sclerosis
A drug that inhibits early-stage B-lymphocyte activation reduced disease activity in patients with relapsing-remitting multiple sclerosis, and showed potential to significantly decrease the annual number of new brain lesions.
The "intriguing" early results of a phase II randomized study suggest that ofatumumab’s targeting of peripheral B cells may be beneficial in treating the disease, according to Daren Austin, Ph.D., head of biopharm clinical pharmacology and biometrics at GlaxoSmithKline, Uxbridge, U.K. He will present these data Wednesday, April 30, at the annual meeting of the American Academy of Neurology.
"These results need to be validated, of course, but the findings are interesting," Dr. Austin said in a press statement. "They provide new insight into the mechanism of B cells in MS [multiple sclerosis] and present a possible new target threshold for exploring the potential benefit of anti-B-cell therapy."
Ofatumumab (Arzerra) is a fully humanized monoclonal antibody against B-lymphocyte antigen CD20, which optimizes B-cell antibody response. On April 17, the U.S. Food and Drug Administration approved the agent for use in combination with chlorambucil in previously untreated patients with chronic lymphocytic leukemia, for whom fludarabine-based therapy is considered inappropriate.
The drug also has shown potential in treating follicular non-Hodgkin’s lymphoma, diffuse large B-cell lymphoma, and rheumatoid arthritis.
The data Dr. Austin will report are part of an ongoing dose-ranging evaluation of 3 to 180 mg of ofatumumab for relapsing-remitting MS. The drug is administered by subcutaneous injection.
The current study included 231 patients who were randomized to placebo or ofatumumab for 24 weeks. The primary endpoint was the correlation of CD19 B-cell count to new gadolinium-enhancing brain lesions (a proxy measure for disease activity). Brain imaging was conducted every 4 weeks throughout the trial.
All of the groups – including placebo – showed lesion activity in the first 4 weeks. However, patients in the active groups showed some lesion suppression after that time. The beneficial effect seemed to appear when patients’ CD19 count fell below 64 cells/mcL, Dr. Austin said.
Researchers correlated B-cell levels to the total number of new brain lesions that appeared on magnetic resonance imaging. If CD19 levels were maintained in the range of 32-64 cells/mcL, patients taking the drug would develop fewer than 1 new lesion per year, compared with 16 new lesions per year in untreated patients, Dr. Austin said in the statement.
The most common side effects, which occurred twice as often in the active group as in the placebo group, were injection-site reaction, dizziness, anxiety, fever, respiratory tract infection, and neuropathy.
Ofatumumab is not approved anywhere in the world for use in the treatment of MS, Dr. Austin added.
GlaxoSmithKline sponsored the study. Dr. Austin is an employee of the company.
A drug that inhibits early-stage B-lymphocyte activation reduced disease activity in patients with relapsing-remitting multiple sclerosis, and showed potential to significantly decrease the annual number of new brain lesions.
The "intriguing" early results of a phase II randomized study suggest that ofatumumab’s targeting of peripheral B cells may be beneficial in treating the disease, according to Daren Austin, Ph.D., head of biopharm clinical pharmacology and biometrics at GlaxoSmithKline, Uxbridge, U.K. He will present these data Wednesday, April 30, at the annual meeting of the American Academy of Neurology.
"These results need to be validated, of course, but the findings are interesting," Dr. Austin said in a press statement. "They provide new insight into the mechanism of B cells in MS [multiple sclerosis] and present a possible new target threshold for exploring the potential benefit of anti-B-cell therapy."
Ofatumumab (Arzerra) is a fully humanized monoclonal antibody against B-lymphocyte antigen CD20, which optimizes B-cell antibody response. On April 17, the U.S. Food and Drug Administration approved the agent for use in combination with chlorambucil in previously untreated patients with chronic lymphocytic leukemia, for whom fludarabine-based therapy is considered inappropriate.
The drug also has shown potential in treating follicular non-Hodgkin’s lymphoma, diffuse large B-cell lymphoma, and rheumatoid arthritis.
The data Dr. Austin will report are part of an ongoing dose-ranging evaluation of 3 to 180 mg of ofatumumab for relapsing-remitting MS. The drug is administered by subcutaneous injection.
The current study included 231 patients who were randomized to placebo or ofatumumab for 24 weeks. The primary endpoint was the correlation of CD19 B-cell count to new gadolinium-enhancing brain lesions (a proxy measure for disease activity). Brain imaging was conducted every 4 weeks throughout the trial.
All of the groups – including placebo – showed lesion activity in the first 4 weeks. However, patients in the active groups showed some lesion suppression after that time. The beneficial effect seemed to appear when patients’ CD19 count fell below 64 cells/mcL, Dr. Austin said.
Researchers correlated B-cell levels to the total number of new brain lesions that appeared on magnetic resonance imaging. If CD19 levels were maintained in the range of 32-64 cells/mcL, patients taking the drug would develop fewer than 1 new lesion per year, compared with 16 new lesions per year in untreated patients, Dr. Austin said in the statement.
The most common side effects, which occurred twice as often in the active group as in the placebo group, were injection-site reaction, dizziness, anxiety, fever, respiratory tract infection, and neuropathy.
Ofatumumab is not approved anywhere in the world for use in the treatment of MS, Dr. Austin added.
GlaxoSmithKline sponsored the study. Dr. Austin is an employee of the company.
A drug that inhibits early-stage B-lymphocyte activation reduced disease activity in patients with relapsing-remitting multiple sclerosis, and showed potential to significantly decrease the annual number of new brain lesions.
The "intriguing" early results of a phase II randomized study suggest that ofatumumab’s targeting of peripheral B cells may be beneficial in treating the disease, according to Daren Austin, Ph.D., head of biopharm clinical pharmacology and biometrics at GlaxoSmithKline, Uxbridge, U.K. He will present these data Wednesday, April 30, at the annual meeting of the American Academy of Neurology.
"These results need to be validated, of course, but the findings are interesting," Dr. Austin said in a press statement. "They provide new insight into the mechanism of B cells in MS [multiple sclerosis] and present a possible new target threshold for exploring the potential benefit of anti-B-cell therapy."
Ofatumumab (Arzerra) is a fully humanized monoclonal antibody against B-lymphocyte antigen CD20, which optimizes B-cell antibody response. On April 17, the U.S. Food and Drug Administration approved the agent for use in combination with chlorambucil in previously untreated patients with chronic lymphocytic leukemia, for whom fludarabine-based therapy is considered inappropriate.
The drug also has shown potential in treating follicular non-Hodgkin’s lymphoma, diffuse large B-cell lymphoma, and rheumatoid arthritis.
The data Dr. Austin will report are part of an ongoing dose-ranging evaluation of 3 to 180 mg of ofatumumab for relapsing-remitting MS. The drug is administered by subcutaneous injection.
The current study included 231 patients who were randomized to placebo or ofatumumab for 24 weeks. The primary endpoint was the correlation of CD19 B-cell count to new gadolinium-enhancing brain lesions (a proxy measure for disease activity). Brain imaging was conducted every 4 weeks throughout the trial.
All of the groups – including placebo – showed lesion activity in the first 4 weeks. However, patients in the active groups showed some lesion suppression after that time. The beneficial effect seemed to appear when patients’ CD19 count fell below 64 cells/mcL, Dr. Austin said.
Researchers correlated B-cell levels to the total number of new brain lesions that appeared on magnetic resonance imaging. If CD19 levels were maintained in the range of 32-64 cells/mcL, patients taking the drug would develop fewer than 1 new lesion per year, compared with 16 new lesions per year in untreated patients, Dr. Austin said in the statement.
The most common side effects, which occurred twice as often in the active group as in the placebo group, were injection-site reaction, dizziness, anxiety, fever, respiratory tract infection, and neuropathy.
Ofatumumab is not approved anywhere in the world for use in the treatment of MS, Dr. Austin added.
GlaxoSmithKline sponsored the study. Dr. Austin is an employee of the company.
FROM THE AAN 2014 ANNUAL MEETING
Major finding: Patients receiving the biologic agent ofatumumab experienced suppression of multiple sclerosis lesions when their CD19 B-cell counts fell below 64 cells/mcL.
Data source: A phase II, randomized, dose-ranging study of 231 patients.
Disclosures: GlaxoSmithKline sponsored the study. Dr. Austin is an employee of the company.
MCI confers increased risk of death
Patients with mild cognitive impairment were 80% more likely to die over a 6-year period than cognitively normal patients.
The risks were significantly elevated for both patients with amnestic and nonamnestic mild cognitive impairment (MCI), Dr. Maria Vassilaki and her colleagues reported in a study released April 23 in advance of the annual meeting of the American Academy of Neurology.
"Our findings build on the previous awareness of the higher mortality rate associated with MCI [mild cognitive impairment] and could promote greater vigilance in the management of these patients, enhancing timely management of any medical issues that might arise," said Dr. Vassilaki of the Mayo Clinic, Rochester, Minn. "In parallel, this information could serve as a reminder to individuals diagnosed with MCI to consult promptly with their health care team in any instances where health-related issues arise."
The findings were from a substudy of the Mayo Clinic Study on Aging (Neuroepidemiology 2008;30:58-69). The study accrued information on more than 2,000 residents of Olmstead County, Minn., who were aged 70-89 years at the time of enrollment.
Over a median follow-up of about 6 years, 331 of the 862 subjects with MCI died as did 224 of the 1,292 cognitively normal subjects.
Mortality was significantly increased whether the subjects later developed dementia (hazard ratio, 1.47). But it was much higher among those who had amnestic MCI than among the nonamnestic group (HR, 1.68 and 2.26, respectively).
"We still need to continue the research for a longer follow-up period to see whether such differences in death rates persist," Dr. Vassilaki said in an interview. "We also need to understand the factors that lead to greater mortality in individuals with MCI and to determine why individuals with nonamnestic MCI had a higher mortality rate than [did] those with amnestic MCI, compared with cognitively normal individuals."
The causal relationship is still under investigation, she said.
"There is another step to this investigation underway, to look at the causes of death. We just retrieved data on the underlying causes of death and will compare those in the different MCI subgroups and in cognitively normal individuals. This might shed some light," Dr. Vassilaki said.
She noted that previous studies have reported that individuals with MCI (either with amnestic MCI or nonamnestic MCI, depending on what the study investigated) were more likely to have dementia as an underlying cause of death, compared with cognitively normal individuals.
Dr. Vassilaki noted that Dr. Ronald Peterson and Dr. Rosebud O. Roberts are primary investigators on the Mayo Clinic Study on Aging.
She reported having no financial conflicts of interest. The study was supported with a grant from the National Institute on Aging.
On Twitter @alz_gal
Patients with mild cognitive impairment were 80% more likely to die over a 6-year period than cognitively normal patients.
The risks were significantly elevated for both patients with amnestic and nonamnestic mild cognitive impairment (MCI), Dr. Maria Vassilaki and her colleagues reported in a study released April 23 in advance of the annual meeting of the American Academy of Neurology.
"Our findings build on the previous awareness of the higher mortality rate associated with MCI [mild cognitive impairment] and could promote greater vigilance in the management of these patients, enhancing timely management of any medical issues that might arise," said Dr. Vassilaki of the Mayo Clinic, Rochester, Minn. "In parallel, this information could serve as a reminder to individuals diagnosed with MCI to consult promptly with their health care team in any instances where health-related issues arise."
The findings were from a substudy of the Mayo Clinic Study on Aging (Neuroepidemiology 2008;30:58-69). The study accrued information on more than 2,000 residents of Olmstead County, Minn., who were aged 70-89 years at the time of enrollment.
Over a median follow-up of about 6 years, 331 of the 862 subjects with MCI died as did 224 of the 1,292 cognitively normal subjects.
Mortality was significantly increased whether the subjects later developed dementia (hazard ratio, 1.47). But it was much higher among those who had amnestic MCI than among the nonamnestic group (HR, 1.68 and 2.26, respectively).
"We still need to continue the research for a longer follow-up period to see whether such differences in death rates persist," Dr. Vassilaki said in an interview. "We also need to understand the factors that lead to greater mortality in individuals with MCI and to determine why individuals with nonamnestic MCI had a higher mortality rate than [did] those with amnestic MCI, compared with cognitively normal individuals."
The causal relationship is still under investigation, she said.
"There is another step to this investigation underway, to look at the causes of death. We just retrieved data on the underlying causes of death and will compare those in the different MCI subgroups and in cognitively normal individuals. This might shed some light," Dr. Vassilaki said.
She noted that previous studies have reported that individuals with MCI (either with amnestic MCI or nonamnestic MCI, depending on what the study investigated) were more likely to have dementia as an underlying cause of death, compared with cognitively normal individuals.
Dr. Vassilaki noted that Dr. Ronald Peterson and Dr. Rosebud O. Roberts are primary investigators on the Mayo Clinic Study on Aging.
She reported having no financial conflicts of interest. The study was supported with a grant from the National Institute on Aging.
On Twitter @alz_gal
Patients with mild cognitive impairment were 80% more likely to die over a 6-year period than cognitively normal patients.
The risks were significantly elevated for both patients with amnestic and nonamnestic mild cognitive impairment (MCI), Dr. Maria Vassilaki and her colleagues reported in a study released April 23 in advance of the annual meeting of the American Academy of Neurology.
"Our findings build on the previous awareness of the higher mortality rate associated with MCI [mild cognitive impairment] and could promote greater vigilance in the management of these patients, enhancing timely management of any medical issues that might arise," said Dr. Vassilaki of the Mayo Clinic, Rochester, Minn. "In parallel, this information could serve as a reminder to individuals diagnosed with MCI to consult promptly with their health care team in any instances where health-related issues arise."
The findings were from a substudy of the Mayo Clinic Study on Aging (Neuroepidemiology 2008;30:58-69). The study accrued information on more than 2,000 residents of Olmstead County, Minn., who were aged 70-89 years at the time of enrollment.
Over a median follow-up of about 6 years, 331 of the 862 subjects with MCI died as did 224 of the 1,292 cognitively normal subjects.
Mortality was significantly increased whether the subjects later developed dementia (hazard ratio, 1.47). But it was much higher among those who had amnestic MCI than among the nonamnestic group (HR, 1.68 and 2.26, respectively).
"We still need to continue the research for a longer follow-up period to see whether such differences in death rates persist," Dr. Vassilaki said in an interview. "We also need to understand the factors that lead to greater mortality in individuals with MCI and to determine why individuals with nonamnestic MCI had a higher mortality rate than [did] those with amnestic MCI, compared with cognitively normal individuals."
The causal relationship is still under investigation, she said.
"There is another step to this investigation underway, to look at the causes of death. We just retrieved data on the underlying causes of death and will compare those in the different MCI subgroups and in cognitively normal individuals. This might shed some light," Dr. Vassilaki said.
She noted that previous studies have reported that individuals with MCI (either with amnestic MCI or nonamnestic MCI, depending on what the study investigated) were more likely to have dementia as an underlying cause of death, compared with cognitively normal individuals.
Dr. Vassilaki noted that Dr. Ronald Peterson and Dr. Rosebud O. Roberts are primary investigators on the Mayo Clinic Study on Aging.
She reported having no financial conflicts of interest. The study was supported with a grant from the National Institute on Aging.
On Twitter @alz_gal
AT THE AAN 2014 ANNUAL MEETING
Major finding: Over 6 years of follow-up, 331 of 862 patients with MCI died vs. 224 of 1,292 cognitively normal patients.
Data source: A 2,000 patient subset of the Mayo Clinic Study on Aging, which enrolled residents of Olmsted County, Minn., aged 70-89 years.
Disclosures: Dr. Vassilaki reported no conflicts of interest. The study was supported by the National Institute on Aging.