User login
Treating maternal subclinical hypothyroidism doesn’t improve childhood IQ
ATLANTA – Prenatal treatment of maternal subclinical hypothyroidism or hypothyroxinemia conferred no cognitive, behavioral, or neurodevelopmental benefit to children up to age 5.
The findings of two parallel randomized trials suggest that prenatal screening for these conditions is not necessary, Dr. Brian Casey said at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine. The data also support the American College of Obstetricians and Gynecologists’ 2007 recommendation against routine screening, said Dr. Casey, chief of maternal-fetal medicine and obstetrics at the University of Texas Southwestern Medical Center, Dallas.
The issue of whether to treat pregnant women for subclinical hypothyroidism has been debated for years. Studies in the early 2000s suggested that prenatal levothyroxine did improve child neurocognitive outcomes, but more recent studies, including a 2012 randomized trial, do not.
These data have led both the American College of Obstetricians and Gynecologists and the Endocrine Society to recommend against screening for subclinical hypothyroidism in pregnant women.
Dr. Casey and his colleagues conducted two large parallel randomized studies, one in women with subclinical hypothyroidism and another in women with hypothyroxinemia. Subclinical hypothyroidism was defined as a thyroid stimulating hormone (TSH) level of at least 4 mU/L with normal free T4. Hypothyroxinemia was defined as a normal TSH but a free T4 of less than 0.86 ng/dL.
The primary outcome in each group was child IQ at 5 years. Secondary outcomes were scores on neurodevelopmental and behavioral measures, including the presence of attention deficit hyperactivity disorder, conducted at ages 3, 4, and 5 years.
All women in the trials had a singleton pregnancy of less than 20 weeks’ gestation, with no known history of thyroid disease.
The subclinical hypothyroidism group comprised 677 women. They were a mean of 27 years old, with a mean gestational age of 16.5 weeks. Baseline TSH was 4.5 mU/L; baseline free T4 was 1 ng/dL. All had normal urinary iodide. They were randomized to placebo or to daily 100 mcg levothyroxine. The treatment goal was a TSH of 0.1-2.5 mU/L. Most (93%) achieved this by 21 weeks’ gestation.
There was no significant difference in the primary outcome of child IQ at 5 years between the treated and untreated groups (97 vs. 94). On the Bayley Scales of Infant and Toddler Development, scores of cognition, motor skills, and language were similar in each group at both 12 and 24 months. The Differential Ability Scales scores at 3 and 4 years were also similar. The Child Behavior Checklist scores at ages 4 years and 5 years were similar. There was no indication of an increase in ADHD.
The hypothyroxinemia trial comprised 467 women. They were randomized to placebo or to 50 mcg levothyroxine. These women were a mean of 28 years old with a mean gestational age of 18 weeks. Their baseline TSH was 1.5 mU/L, and baseline free T4 was 0.8 ng/dL. All had normal urinary iodide. The treatment goal was a free T4 of between 0.86 and 1.90 ng/dL. Most (83%) achieved this by 23 weeks’ gestation.
There was no difference on the primary outcome of child IQ at 5 years (94 vs. 91). On the Bayley Scales of Infant and Toddler Development, scores of cognition, motor skills, and language were similar in each group at both 12 and 24 months. The Differential Ability Scales scores at 3 years and 4 years were also similar. The Child Behavior Checklist scores at ages 4years and 5 years were similar. There was no indication of an increase in ADHD.
Dr. Casey added that there was no interaction between gestational age at baseline and treatment outcomes, suggesting that there may be little foundation to the argument that treating earlier in pregnancy is key.
He had no financial disclosures.
ATLANTA – Prenatal treatment of maternal subclinical hypothyroidism or hypothyroxinemia conferred no cognitive, behavioral, or neurodevelopmental benefit to children up to age 5.
The findings of two parallel randomized trials suggest that prenatal screening for these conditions is not necessary, Dr. Brian Casey said at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine. The data also support the American College of Obstetricians and Gynecologists’ 2007 recommendation against routine screening, said Dr. Casey, chief of maternal-fetal medicine and obstetrics at the University of Texas Southwestern Medical Center, Dallas.
The issue of whether to treat pregnant women for subclinical hypothyroidism has been debated for years. Studies in the early 2000s suggested that prenatal levothyroxine did improve child neurocognitive outcomes, but more recent studies, including a 2012 randomized trial, do not.
These data have led both the American College of Obstetricians and Gynecologists and the Endocrine Society to recommend against screening for subclinical hypothyroidism in pregnant women.
Dr. Casey and his colleagues conducted two large parallel randomized studies, one in women with subclinical hypothyroidism and another in women with hypothyroxinemia. Subclinical hypothyroidism was defined as a thyroid stimulating hormone (TSH) level of at least 4 mU/L with normal free T4. Hypothyroxinemia was defined as a normal TSH but a free T4 of less than 0.86 ng/dL.
The primary outcome in each group was child IQ at 5 years. Secondary outcomes were scores on neurodevelopmental and behavioral measures, including the presence of attention deficit hyperactivity disorder, conducted at ages 3, 4, and 5 years.
All women in the trials had a singleton pregnancy of less than 20 weeks’ gestation, with no known history of thyroid disease.
The subclinical hypothyroidism group comprised 677 women. They were a mean of 27 years old, with a mean gestational age of 16.5 weeks. Baseline TSH was 4.5 mU/L; baseline free T4 was 1 ng/dL. All had normal urinary iodide. They were randomized to placebo or to daily 100 mcg levothyroxine. The treatment goal was a TSH of 0.1-2.5 mU/L. Most (93%) achieved this by 21 weeks’ gestation.
There was no significant difference in the primary outcome of child IQ at 5 years between the treated and untreated groups (97 vs. 94). On the Bayley Scales of Infant and Toddler Development, scores of cognition, motor skills, and language were similar in each group at both 12 and 24 months. The Differential Ability Scales scores at 3 and 4 years were also similar. The Child Behavior Checklist scores at ages 4 years and 5 years were similar. There was no indication of an increase in ADHD.
The hypothyroxinemia trial comprised 467 women. They were randomized to placebo or to 50 mcg levothyroxine. These women were a mean of 28 years old with a mean gestational age of 18 weeks. Their baseline TSH was 1.5 mU/L, and baseline free T4 was 0.8 ng/dL. All had normal urinary iodide. The treatment goal was a free T4 of between 0.86 and 1.90 ng/dL. Most (83%) achieved this by 23 weeks’ gestation.
There was no difference on the primary outcome of child IQ at 5 years (94 vs. 91). On the Bayley Scales of Infant and Toddler Development, scores of cognition, motor skills, and language were similar in each group at both 12 and 24 months. The Differential Ability Scales scores at 3 years and 4 years were also similar. The Child Behavior Checklist scores at ages 4years and 5 years were similar. There was no indication of an increase in ADHD.
Dr. Casey added that there was no interaction between gestational age at baseline and treatment outcomes, suggesting that there may be little foundation to the argument that treating earlier in pregnancy is key.
He had no financial disclosures.
ATLANTA – Prenatal treatment of maternal subclinical hypothyroidism or hypothyroxinemia conferred no cognitive, behavioral, or neurodevelopmental benefit to children up to age 5.
The findings of two parallel randomized trials suggest that prenatal screening for these conditions is not necessary, Dr. Brian Casey said at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine. The data also support the American College of Obstetricians and Gynecologists’ 2007 recommendation against routine screening, said Dr. Casey, chief of maternal-fetal medicine and obstetrics at the University of Texas Southwestern Medical Center, Dallas.
The issue of whether to treat pregnant women for subclinical hypothyroidism has been debated for years. Studies in the early 2000s suggested that prenatal levothyroxine did improve child neurocognitive outcomes, but more recent studies, including a 2012 randomized trial, do not.
These data have led both the American College of Obstetricians and Gynecologists and the Endocrine Society to recommend against screening for subclinical hypothyroidism in pregnant women.
Dr. Casey and his colleagues conducted two large parallel randomized studies, one in women with subclinical hypothyroidism and another in women with hypothyroxinemia. Subclinical hypothyroidism was defined as a thyroid stimulating hormone (TSH) level of at least 4 mU/L with normal free T4. Hypothyroxinemia was defined as a normal TSH but a free T4 of less than 0.86 ng/dL.
The primary outcome in each group was child IQ at 5 years. Secondary outcomes were scores on neurodevelopmental and behavioral measures, including the presence of attention deficit hyperactivity disorder, conducted at ages 3, 4, and 5 years.
All women in the trials had a singleton pregnancy of less than 20 weeks’ gestation, with no known history of thyroid disease.
The subclinical hypothyroidism group comprised 677 women. They were a mean of 27 years old, with a mean gestational age of 16.5 weeks. Baseline TSH was 4.5 mU/L; baseline free T4 was 1 ng/dL. All had normal urinary iodide. They were randomized to placebo or to daily 100 mcg levothyroxine. The treatment goal was a TSH of 0.1-2.5 mU/L. Most (93%) achieved this by 21 weeks’ gestation.
There was no significant difference in the primary outcome of child IQ at 5 years between the treated and untreated groups (97 vs. 94). On the Bayley Scales of Infant and Toddler Development, scores of cognition, motor skills, and language were similar in each group at both 12 and 24 months. The Differential Ability Scales scores at 3 and 4 years were also similar. The Child Behavior Checklist scores at ages 4 years and 5 years were similar. There was no indication of an increase in ADHD.
The hypothyroxinemia trial comprised 467 women. They were randomized to placebo or to 50 mcg levothyroxine. These women were a mean of 28 years old with a mean gestational age of 18 weeks. Their baseline TSH was 1.5 mU/L, and baseline free T4 was 0.8 ng/dL. All had normal urinary iodide. The treatment goal was a free T4 of between 0.86 and 1.90 ng/dL. Most (83%) achieved this by 23 weeks’ gestation.
There was no difference on the primary outcome of child IQ at 5 years (94 vs. 91). On the Bayley Scales of Infant and Toddler Development, scores of cognition, motor skills, and language were similar in each group at both 12 and 24 months. The Differential Ability Scales scores at 3 years and 4 years were also similar. The Child Behavior Checklist scores at ages 4years and 5 years were similar. There was no indication of an increase in ADHD.
Dr. Casey added that there was no interaction between gestational age at baseline and treatment outcomes, suggesting that there may be little foundation to the argument that treating earlier in pregnancy is key.
He had no financial disclosures.
AT THE PREGNANCY MEETING
Key clinical point: Levothyroxine for women with subclinical hypothyroidism didn’t boost childhood cognition.
Major finding: Child IQ at 5 years old was 97 in the treated group and 94 in the placebo group – not a significant difference.
Data source: The parallel randomized controlled trials comprised a total of 1,144 women.
Disclosures: Dr. Casey had no financial disclosures.
VIDEO: Could noninvasive prenatal testing mean gene profiling for every pregnancy?
ATLANTA – The decision of whether to screen a pregnancy for genetic anomalies should not be taken lightly, especially in light of the invasive procedures required. But what if a simple maternal blood test could provide everything we needed to know about a fetus’ genetic health?
At the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine, Dr. Ronald J. Wapner of Columbia University, New York, said that time is quickly approaching, and that cell-free fetal DNA testing should – and will – become a routine part of prenatal care.
Watch an interview with Dr. Wapner here.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ATLANTA – The decision of whether to screen a pregnancy for genetic anomalies should not be taken lightly, especially in light of the invasive procedures required. But what if a simple maternal blood test could provide everything we needed to know about a fetus’ genetic health?
At the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine, Dr. Ronald J. Wapner of Columbia University, New York, said that time is quickly approaching, and that cell-free fetal DNA testing should – and will – become a routine part of prenatal care.
Watch an interview with Dr. Wapner here.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ATLANTA – The decision of whether to screen a pregnancy for genetic anomalies should not be taken lightly, especially in light of the invasive procedures required. But what if a simple maternal blood test could provide everything we needed to know about a fetus’ genetic health?
At the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine, Dr. Ronald J. Wapner of Columbia University, New York, said that time is quickly approaching, and that cell-free fetal DNA testing should – and will – become a routine part of prenatal care.
Watch an interview with Dr. Wapner here.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE PREGNANCY MEETING
VIDEO: To optimize children’s oral health, take care of moms first
ATLANTA – Oral health – whether poor or good – seems to run in families. Is it behavior, biology, or both?
At the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine, Dr. Renee Samelson, a maternal-fetal medicine specialist with a certification in preventive medicine, said that taking care of moms’ teeth and gums, even before pregnancy, gives children the best shot at maintaining a healthy mouth from infancy to old age.
Watch an interview with Dr. Samelson here.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ATLANTA – Oral health – whether poor or good – seems to run in families. Is it behavior, biology, or both?
At the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine, Dr. Renee Samelson, a maternal-fetal medicine specialist with a certification in preventive medicine, said that taking care of moms’ teeth and gums, even before pregnancy, gives children the best shot at maintaining a healthy mouth from infancy to old age.
Watch an interview with Dr. Samelson here.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ATLANTA – Oral health – whether poor or good – seems to run in families. Is it behavior, biology, or both?
At the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine, Dr. Renee Samelson, a maternal-fetal medicine specialist with a certification in preventive medicine, said that taking care of moms’ teeth and gums, even before pregnancy, gives children the best shot at maintaining a healthy mouth from infancy to old age.
Watch an interview with Dr. Samelson here.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE PREGNANCY MEETING
Inflammatory brain changes in familial Alzheimer’s appear decades before symptoms do
Inflammatory brain changes appear to develop 20 years before the onset of symptoms in people with some familial forms of Alzheimer’s disease, according to a longitudinal analysis of PET imaging biomarkers of astrocyte activation, amyloid-beta accumulation, and glucose metabolism in the brain.
Imaging studies demonstrated a sharp elevation in astrocyte activation – a response to neuronal insult – before amyloid-beta began to accumulate in the brain, and then steadily declined. These changes were eventually followed by a steady and spreading decrease in glucose metabolism that reached the hippocampus just 2 years before symptoms appeared, first author Elena Rodriguez-Vieitez, Ph.D., of the Karolinska Institute, Stockholm, and her colleagues reported (Brain. 2016 Jan 26. doi: 10.1093/brain/awv404).
The findings suggest that the disease may begin with an unknown insult that stimulates reactive astrocytosis – in familial forms of Alzheimer’s at least – and that the cells themselves could be a legitimate therapeutic target, wrote the investigators, led by Dr. Agneta Nordberg.
The initial astrocyte-alerting hit is still unknown, but it may be a reaction to soluble amyloid-beta. “Recent studies have supported the hypothesis that astrocytes have a beneficial role contributing to amyloid-beta clearance, but also that excess amyloid-beta can lead to oxidative stress and damage, and as a consequence to reduced astrocyte functionality, leading to reactive changes and decreased neuronal support, and thereby contributing to neurodegeneration.”
The decline in astrocyte activation with disease progression is another mystery, she said. It may be “an indication of a reduction in a certain type of astrocyte activation or functionality, a change of astrocyte activation phenotype, or possibly ‘astrodegeneration’ and astrocyte cell loss itself, as has been reported towards the late stages of Alzheimer’s disease.”
The study tracked brain PET imaging changes in 52 people: 27 from families with autosomal dominant Alzheimer’s disease (ADAD) mutations, including presenilin 1 or amyloid precursor protein genes, and 25 with sporadic Alzheimer’s disease or mild cognitive impairment (MCI). It employed three different PET tracers: Pittsburgh imaging compound B (PiB) to detect amyloid-beta; 18F-fluorodeoxyglucose (FDG) to detect glucose metabolism; and 11C-deuterium-L-deprenyl (DED) to detect astrocyte activation. Half had a follow-up visit after a mean of 2.8 years.
The ADAD cohort was divided into 16 noncarriers; 4 symptomatic carriers; and 7 presymptomatic carriers who were a mean of 10 years from symptom onset. The sporadic AD group was divided into 13 amyloid-positive and 4 amyloid-negative MCI patients, and 8 patients with an Alzheimer’s diagnosis. The 16 ADAD noncarriers served as a control group for PiB retention and FDG uptake, and 14 age-matched healthy controls served as controls for DED binding.
At baseline, all subjects underwent PET imaging with all three tracers, as well as cerebrospinal fluid biomarker analysis and neuropsychological assessments. Half of the subjects had additional clinical and imaging studies 3 years later. The investigators used historical data to estimate the age of symptom onset in the ADAD subjects, and thus extrapolated the imaging findings to reflect the pathologic course.
At baseline, there were significant between-group differences in all three imaging studies. PiB-positive MCI patients and Alzheimer’s patients from both the sporadic and ADAD cohorts had the highest PiB retention. But presymptomatic carriers had significantly higher DED binding than did any of the other groups. The increased astrocytosis that occurs with higher DED binding occurred in particular in 4 of 12 brain regions surveyed: the anterior cingulate cortex, thalamus, frontal, and parietal regions. Interestingly, the authors noted, DED binding in the sporadic Alzheimer’s and ADAD groups was not statistically different from that seen in a group of healthy control patients, except for a trend for higher binding in the frontal region.
In most brain regions at baseline, FDG uptake was greater in presymptomatic carriers and healthy controls than in either the PiB-positive MCI, sporadic Alzheimer’s, or ADAD groups, but there were no significant differences between the presymptomatic carriers and healthy controls or between the PiB-positive MCI, sporadic Alzheimer’s, or ADAD groups. FDG uptake at baseline was lower in the left parietal region of presymptomatic carriers than in the healthy controls and at follow-up this difference spread to the left posterior cingulate cortex and other parietal regions and to the left middle frontal gyrus and the bilateral cuneus. Presymptomatic carriers had significantly greater FDG uptake in the frontal and temporal regions and the right thalamus than did PiB-positive MCI patients, which persisted through follow-up.
The investigators used a linear mixed-effects model to estimate how astrocytosis, amyloid deposition, and glucose metabolism would change over time in both the ADAD and sporadic Alzheimer’s groups.
Comparing ADAD carriers to noncarriers, the model determined that amyloid began to aggregate in the putamen at about 17 years before symptom onset in carriers. From there, plaques spread outward, reaching the caudate nucleus and the anterior and posterior cingulate cortices by 15 years before symptoms, and the frontal cortex and other cortical regions about 14 years before.
Conversely, the model found that DED binding in ADAD presymptomatic carriers steadily declined from its peak at about 20 years before expected symptom onset. By the time symptoms appeared, astrocyte activation was similar to that observed in noncarriers. Among patients with sporadic AD/MCI, there were no significant temporal changes in DED binding.
Presymptomatic carriers also showed significant linear changes in glucose utilization. FDG uptake in the parietal and temporal regions began to decline about 7 years before symptom onset. Glucose metabolism was maintained in the caudate nucleus, thalamus, and hippocampus until it began to decline about 2 years before symptom onset.
Patients with amyloid-positive MCI or sporadic Alzheimer’s showed significant temporal increases in amyloid deposition, and declines in glucose metabolism in the anterior cingulate cortex, but no significant changes in astrocyte activation.
“Possible explanations for this finding include the heterogeneity of disease stage in MCI patients, the shorter time span investigated in the sporadic patients compared to the ADAD participants, or a possibly different progression of astrocyte activation in the sporadic compared to the autosomal dominant forms,” the authors noted.
The research was supported by grants from Swedish government entities, various Swedish foundations, and GE Healthcare. The authors had no financial disclosures.
Inflammatory brain changes appear to develop 20 years before the onset of symptoms in people with some familial forms of Alzheimer’s disease, according to a longitudinal analysis of PET imaging biomarkers of astrocyte activation, amyloid-beta accumulation, and glucose metabolism in the brain.
Imaging studies demonstrated a sharp elevation in astrocyte activation – a response to neuronal insult – before amyloid-beta began to accumulate in the brain, and then steadily declined. These changes were eventually followed by a steady and spreading decrease in glucose metabolism that reached the hippocampus just 2 years before symptoms appeared, first author Elena Rodriguez-Vieitez, Ph.D., of the Karolinska Institute, Stockholm, and her colleagues reported (Brain. 2016 Jan 26. doi: 10.1093/brain/awv404).
The findings suggest that the disease may begin with an unknown insult that stimulates reactive astrocytosis – in familial forms of Alzheimer’s at least – and that the cells themselves could be a legitimate therapeutic target, wrote the investigators, led by Dr. Agneta Nordberg.
The initial astrocyte-alerting hit is still unknown, but it may be a reaction to soluble amyloid-beta. “Recent studies have supported the hypothesis that astrocytes have a beneficial role contributing to amyloid-beta clearance, but also that excess amyloid-beta can lead to oxidative stress and damage, and as a consequence to reduced astrocyte functionality, leading to reactive changes and decreased neuronal support, and thereby contributing to neurodegeneration.”
The decline in astrocyte activation with disease progression is another mystery, she said. It may be “an indication of a reduction in a certain type of astrocyte activation or functionality, a change of astrocyte activation phenotype, or possibly ‘astrodegeneration’ and astrocyte cell loss itself, as has been reported towards the late stages of Alzheimer’s disease.”
The study tracked brain PET imaging changes in 52 people: 27 from families with autosomal dominant Alzheimer’s disease (ADAD) mutations, including presenilin 1 or amyloid precursor protein genes, and 25 with sporadic Alzheimer’s disease or mild cognitive impairment (MCI). It employed three different PET tracers: Pittsburgh imaging compound B (PiB) to detect amyloid-beta; 18F-fluorodeoxyglucose (FDG) to detect glucose metabolism; and 11C-deuterium-L-deprenyl (DED) to detect astrocyte activation. Half had a follow-up visit after a mean of 2.8 years.
The ADAD cohort was divided into 16 noncarriers; 4 symptomatic carriers; and 7 presymptomatic carriers who were a mean of 10 years from symptom onset. The sporadic AD group was divided into 13 amyloid-positive and 4 amyloid-negative MCI patients, and 8 patients with an Alzheimer’s diagnosis. The 16 ADAD noncarriers served as a control group for PiB retention and FDG uptake, and 14 age-matched healthy controls served as controls for DED binding.
At baseline, all subjects underwent PET imaging with all three tracers, as well as cerebrospinal fluid biomarker analysis and neuropsychological assessments. Half of the subjects had additional clinical and imaging studies 3 years later. The investigators used historical data to estimate the age of symptom onset in the ADAD subjects, and thus extrapolated the imaging findings to reflect the pathologic course.
At baseline, there were significant between-group differences in all three imaging studies. PiB-positive MCI patients and Alzheimer’s patients from both the sporadic and ADAD cohorts had the highest PiB retention. But presymptomatic carriers had significantly higher DED binding than did any of the other groups. The increased astrocytosis that occurs with higher DED binding occurred in particular in 4 of 12 brain regions surveyed: the anterior cingulate cortex, thalamus, frontal, and parietal regions. Interestingly, the authors noted, DED binding in the sporadic Alzheimer’s and ADAD groups was not statistically different from that seen in a group of healthy control patients, except for a trend for higher binding in the frontal region.
In most brain regions at baseline, FDG uptake was greater in presymptomatic carriers and healthy controls than in either the PiB-positive MCI, sporadic Alzheimer’s, or ADAD groups, but there were no significant differences between the presymptomatic carriers and healthy controls or between the PiB-positive MCI, sporadic Alzheimer’s, or ADAD groups. FDG uptake at baseline was lower in the left parietal region of presymptomatic carriers than in the healthy controls and at follow-up this difference spread to the left posterior cingulate cortex and other parietal regions and to the left middle frontal gyrus and the bilateral cuneus. Presymptomatic carriers had significantly greater FDG uptake in the frontal and temporal regions and the right thalamus than did PiB-positive MCI patients, which persisted through follow-up.
The investigators used a linear mixed-effects model to estimate how astrocytosis, amyloid deposition, and glucose metabolism would change over time in both the ADAD and sporadic Alzheimer’s groups.
Comparing ADAD carriers to noncarriers, the model determined that amyloid began to aggregate in the putamen at about 17 years before symptom onset in carriers. From there, plaques spread outward, reaching the caudate nucleus and the anterior and posterior cingulate cortices by 15 years before symptoms, and the frontal cortex and other cortical regions about 14 years before.
Conversely, the model found that DED binding in ADAD presymptomatic carriers steadily declined from its peak at about 20 years before expected symptom onset. By the time symptoms appeared, astrocyte activation was similar to that observed in noncarriers. Among patients with sporadic AD/MCI, there were no significant temporal changes in DED binding.
Presymptomatic carriers also showed significant linear changes in glucose utilization. FDG uptake in the parietal and temporal regions began to decline about 7 years before symptom onset. Glucose metabolism was maintained in the caudate nucleus, thalamus, and hippocampus until it began to decline about 2 years before symptom onset.
Patients with amyloid-positive MCI or sporadic Alzheimer’s showed significant temporal increases in amyloid deposition, and declines in glucose metabolism in the anterior cingulate cortex, but no significant changes in astrocyte activation.
“Possible explanations for this finding include the heterogeneity of disease stage in MCI patients, the shorter time span investigated in the sporadic patients compared to the ADAD participants, or a possibly different progression of astrocyte activation in the sporadic compared to the autosomal dominant forms,” the authors noted.
The research was supported by grants from Swedish government entities, various Swedish foundations, and GE Healthcare. The authors had no financial disclosures.
Inflammatory brain changes appear to develop 20 years before the onset of symptoms in people with some familial forms of Alzheimer’s disease, according to a longitudinal analysis of PET imaging biomarkers of astrocyte activation, amyloid-beta accumulation, and glucose metabolism in the brain.
Imaging studies demonstrated a sharp elevation in astrocyte activation – a response to neuronal insult – before amyloid-beta began to accumulate in the brain, and then steadily declined. These changes were eventually followed by a steady and spreading decrease in glucose metabolism that reached the hippocampus just 2 years before symptoms appeared, first author Elena Rodriguez-Vieitez, Ph.D., of the Karolinska Institute, Stockholm, and her colleagues reported (Brain. 2016 Jan 26. doi: 10.1093/brain/awv404).
The findings suggest that the disease may begin with an unknown insult that stimulates reactive astrocytosis – in familial forms of Alzheimer’s at least – and that the cells themselves could be a legitimate therapeutic target, wrote the investigators, led by Dr. Agneta Nordberg.
The initial astrocyte-alerting hit is still unknown, but it may be a reaction to soluble amyloid-beta. “Recent studies have supported the hypothesis that astrocytes have a beneficial role contributing to amyloid-beta clearance, but also that excess amyloid-beta can lead to oxidative stress and damage, and as a consequence to reduced astrocyte functionality, leading to reactive changes and decreased neuronal support, and thereby contributing to neurodegeneration.”
The decline in astrocyte activation with disease progression is another mystery, she said. It may be “an indication of a reduction in a certain type of astrocyte activation or functionality, a change of astrocyte activation phenotype, or possibly ‘astrodegeneration’ and astrocyte cell loss itself, as has been reported towards the late stages of Alzheimer’s disease.”
The study tracked brain PET imaging changes in 52 people: 27 from families with autosomal dominant Alzheimer’s disease (ADAD) mutations, including presenilin 1 or amyloid precursor protein genes, and 25 with sporadic Alzheimer’s disease or mild cognitive impairment (MCI). It employed three different PET tracers: Pittsburgh imaging compound B (PiB) to detect amyloid-beta; 18F-fluorodeoxyglucose (FDG) to detect glucose metabolism; and 11C-deuterium-L-deprenyl (DED) to detect astrocyte activation. Half had a follow-up visit after a mean of 2.8 years.
The ADAD cohort was divided into 16 noncarriers; 4 symptomatic carriers; and 7 presymptomatic carriers who were a mean of 10 years from symptom onset. The sporadic AD group was divided into 13 amyloid-positive and 4 amyloid-negative MCI patients, and 8 patients with an Alzheimer’s diagnosis. The 16 ADAD noncarriers served as a control group for PiB retention and FDG uptake, and 14 age-matched healthy controls served as controls for DED binding.
At baseline, all subjects underwent PET imaging with all three tracers, as well as cerebrospinal fluid biomarker analysis and neuropsychological assessments. Half of the subjects had additional clinical and imaging studies 3 years later. The investigators used historical data to estimate the age of symptom onset in the ADAD subjects, and thus extrapolated the imaging findings to reflect the pathologic course.
At baseline, there were significant between-group differences in all three imaging studies. PiB-positive MCI patients and Alzheimer’s patients from both the sporadic and ADAD cohorts had the highest PiB retention. But presymptomatic carriers had significantly higher DED binding than did any of the other groups. The increased astrocytosis that occurs with higher DED binding occurred in particular in 4 of 12 brain regions surveyed: the anterior cingulate cortex, thalamus, frontal, and parietal regions. Interestingly, the authors noted, DED binding in the sporadic Alzheimer’s and ADAD groups was not statistically different from that seen in a group of healthy control patients, except for a trend for higher binding in the frontal region.
In most brain regions at baseline, FDG uptake was greater in presymptomatic carriers and healthy controls than in either the PiB-positive MCI, sporadic Alzheimer’s, or ADAD groups, but there were no significant differences between the presymptomatic carriers and healthy controls or between the PiB-positive MCI, sporadic Alzheimer’s, or ADAD groups. FDG uptake at baseline was lower in the left parietal region of presymptomatic carriers than in the healthy controls and at follow-up this difference spread to the left posterior cingulate cortex and other parietal regions and to the left middle frontal gyrus and the bilateral cuneus. Presymptomatic carriers had significantly greater FDG uptake in the frontal and temporal regions and the right thalamus than did PiB-positive MCI patients, which persisted through follow-up.
The investigators used a linear mixed-effects model to estimate how astrocytosis, amyloid deposition, and glucose metabolism would change over time in both the ADAD and sporadic Alzheimer’s groups.
Comparing ADAD carriers to noncarriers, the model determined that amyloid began to aggregate in the putamen at about 17 years before symptom onset in carriers. From there, plaques spread outward, reaching the caudate nucleus and the anterior and posterior cingulate cortices by 15 years before symptoms, and the frontal cortex and other cortical regions about 14 years before.
Conversely, the model found that DED binding in ADAD presymptomatic carriers steadily declined from its peak at about 20 years before expected symptom onset. By the time symptoms appeared, astrocyte activation was similar to that observed in noncarriers. Among patients with sporadic AD/MCI, there were no significant temporal changes in DED binding.
Presymptomatic carriers also showed significant linear changes in glucose utilization. FDG uptake in the parietal and temporal regions began to decline about 7 years before symptom onset. Glucose metabolism was maintained in the caudate nucleus, thalamus, and hippocampus until it began to decline about 2 years before symptom onset.
Patients with amyloid-positive MCI or sporadic Alzheimer’s showed significant temporal increases in amyloid deposition, and declines in glucose metabolism in the anterior cingulate cortex, but no significant changes in astrocyte activation.
“Possible explanations for this finding include the heterogeneity of disease stage in MCI patients, the shorter time span investigated in the sporadic patients compared to the ADAD participants, or a possibly different progression of astrocyte activation in the sporadic compared to the autosomal dominant forms,” the authors noted.
The research was supported by grants from Swedish government entities, various Swedish foundations, and GE Healthcare. The authors had no financial disclosures.
FROM BRAIN
Key clinical point: Astrocytes are activated in the brains of Alzheimer’s disease patients 20 years before cognitive symptoms appear.
Major finding: Inflammatory brain changes appear to develop 20 years before the onset of symptoms in people with some familial forms of Alzheimer’s disease.
Data source: A longitudinal cohort study of 52 subjects, including 27 members of families with autosomal dominant Alzheimer’s disease and 25 with sporadic Alzheimer’s disease or mild cognitive impairment.
Disclosures: The research was supported by grants from Swedish government entities, various Swedish foundations, and GE Healthcare. The authors had no financial disclosures.
Chlorhexidine beats iodine for preventing C-section wound infections
ATLANTA – A chlorhexidine/alcohol skin antiseptic cut cesarean section surgical site infections by half, compared with a solution of iodine and alcohol.
The chlorhexidine solution significantly reduced the risk of both superficial and deep incisional infections, Dr. Methodius G. Tuuli reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine. The study was simultaneously published in the New England Journal of Medicine (2016 Feb 4. doi: 10.1056/NEJMoa1511048).
The randomized trial is the first to examine the two antiseptics in obstetric surgery, noted Dr. Tuuli of Washington University, St. Louis. The results echo those repeatedly found in the general surgical literature, and, he said, clearly show that chlorhexidine-based skin prep is more effective than the more often–employed iodine-based prep.
“We become comfortable doing the things we have always done, because that’s the way we were taught, and we see no reason to change,” he said in an interview. “I think now is the time to make a change for our patients.”
Dr. Tuuli’s study comprised 1,147 patients who delivered via cesarean section from 2011-2015. They were randomized to either a chlorhexidine/alcohol antiseptic (2% chlorhexidine gluconate with 70% isopropyl alcohol) or the iodine/alcohol combination (8.3% povidone-iodine with 72.5% isopropyl alcohol). Both groups received standard-of-care systemic antibiotic prophylaxis.
They were followed daily until discharge from the hospital, and then with a telephone call 30 days after delivery to assess whether a surgical site infection had occurred, as well as any visits to a physician’s office or emergency department that were related to a wound complication.
The co-primary endpoints were superficial and deep incisional infections. Secondary endpoints included length of hospital stay; physician office visits; hospital readmissions for infection-related complications; endometritis; positive wound culture; skin irritation; and allergic reaction.
Surgical site infections occurred in 23 patients in the chlorhexidine group and 42 in the iodine group (4.0% vs. 7.3%) – a significant 45% risk reduction (relative risk, 0.55). Superficial infections were significantly less common in the chlorhexidine group (3.0% vs. 4.9%), as were deep infections (1.0% vs. 2.4%).
A subgroup analysis examined unscheduled vs. scheduled cesarean; obese vs. nonobese patients; suture vs. staple closure; diabetes vs. no diabetes; and chronic comorbidities vs. none. Chlorhexidine was significantly more effective than iodine in each of these groups.
Antiseptic type did not affect rates of skin separation, seroma, hematoma, or cellulitis. Nor did it affect the rates of endometritis, hospitalization for infectious complications, or length of hospital stay. However, those in the chlorhexidine group were significantly less likely to visit a physician for wound care (7.9% vs. 12.5%)
Cultures were obtained on 32 patients with a confirmed infection; 27 of these specimens were positive. About half of the positive cultures were polymicrobial. The most common isolate was Staphylococcus aureus (37%). Methicillin-resistant S. aureus (MRSA) was present in 12% of cultures in the chlorhexidine group and 17% in the iodine group.
In an interview, Dr. Tuuli said that chlorhexidine has several properties that make it more effective than iodine. It is effective against both gram-negative and gram-positive organisms, including MRSA, and is not inactivated by organic matter. Although chlorhexidine is more likely than iodine to provoke an allergic reaction, none were observed in this study.
The study was supported by a grant from the National Institutes of Health. Dr. Tuuli reported having no financial disclosures; the antiseptics were procured and paid for by the medical center.
Watch Dr. Tuuli discuss the study results here.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ATLANTA – A chlorhexidine/alcohol skin antiseptic cut cesarean section surgical site infections by half, compared with a solution of iodine and alcohol.
The chlorhexidine solution significantly reduced the risk of both superficial and deep incisional infections, Dr. Methodius G. Tuuli reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine. The study was simultaneously published in the New England Journal of Medicine (2016 Feb 4. doi: 10.1056/NEJMoa1511048).
The randomized trial is the first to examine the two antiseptics in obstetric surgery, noted Dr. Tuuli of Washington University, St. Louis. The results echo those repeatedly found in the general surgical literature, and, he said, clearly show that chlorhexidine-based skin prep is more effective than the more often–employed iodine-based prep.
“We become comfortable doing the things we have always done, because that’s the way we were taught, and we see no reason to change,” he said in an interview. “I think now is the time to make a change for our patients.”
Dr. Tuuli’s study comprised 1,147 patients who delivered via cesarean section from 2011-2015. They were randomized to either a chlorhexidine/alcohol antiseptic (2% chlorhexidine gluconate with 70% isopropyl alcohol) or the iodine/alcohol combination (8.3% povidone-iodine with 72.5% isopropyl alcohol). Both groups received standard-of-care systemic antibiotic prophylaxis.
They were followed daily until discharge from the hospital, and then with a telephone call 30 days after delivery to assess whether a surgical site infection had occurred, as well as any visits to a physician’s office or emergency department that were related to a wound complication.
The co-primary endpoints were superficial and deep incisional infections. Secondary endpoints included length of hospital stay; physician office visits; hospital readmissions for infection-related complications; endometritis; positive wound culture; skin irritation; and allergic reaction.
Surgical site infections occurred in 23 patients in the chlorhexidine group and 42 in the iodine group (4.0% vs. 7.3%) – a significant 45% risk reduction (relative risk, 0.55). Superficial infections were significantly less common in the chlorhexidine group (3.0% vs. 4.9%), as were deep infections (1.0% vs. 2.4%).
A subgroup analysis examined unscheduled vs. scheduled cesarean; obese vs. nonobese patients; suture vs. staple closure; diabetes vs. no diabetes; and chronic comorbidities vs. none. Chlorhexidine was significantly more effective than iodine in each of these groups.
Antiseptic type did not affect rates of skin separation, seroma, hematoma, or cellulitis. Nor did it affect the rates of endometritis, hospitalization for infectious complications, or length of hospital stay. However, those in the chlorhexidine group were significantly less likely to visit a physician for wound care (7.9% vs. 12.5%)
Cultures were obtained on 32 patients with a confirmed infection; 27 of these specimens were positive. About half of the positive cultures were polymicrobial. The most common isolate was Staphylococcus aureus (37%). Methicillin-resistant S. aureus (MRSA) was present in 12% of cultures in the chlorhexidine group and 17% in the iodine group.
In an interview, Dr. Tuuli said that chlorhexidine has several properties that make it more effective than iodine. It is effective against both gram-negative and gram-positive organisms, including MRSA, and is not inactivated by organic matter. Although chlorhexidine is more likely than iodine to provoke an allergic reaction, none were observed in this study.
The study was supported by a grant from the National Institutes of Health. Dr. Tuuli reported having no financial disclosures; the antiseptics were procured and paid for by the medical center.
Watch Dr. Tuuli discuss the study results here.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ATLANTA – A chlorhexidine/alcohol skin antiseptic cut cesarean section surgical site infections by half, compared with a solution of iodine and alcohol.
The chlorhexidine solution significantly reduced the risk of both superficial and deep incisional infections, Dr. Methodius G. Tuuli reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine. The study was simultaneously published in the New England Journal of Medicine (2016 Feb 4. doi: 10.1056/NEJMoa1511048).
The randomized trial is the first to examine the two antiseptics in obstetric surgery, noted Dr. Tuuli of Washington University, St. Louis. The results echo those repeatedly found in the general surgical literature, and, he said, clearly show that chlorhexidine-based skin prep is more effective than the more often–employed iodine-based prep.
“We become comfortable doing the things we have always done, because that’s the way we were taught, and we see no reason to change,” he said in an interview. “I think now is the time to make a change for our patients.”
Dr. Tuuli’s study comprised 1,147 patients who delivered via cesarean section from 2011-2015. They were randomized to either a chlorhexidine/alcohol antiseptic (2% chlorhexidine gluconate with 70% isopropyl alcohol) or the iodine/alcohol combination (8.3% povidone-iodine with 72.5% isopropyl alcohol). Both groups received standard-of-care systemic antibiotic prophylaxis.
They were followed daily until discharge from the hospital, and then with a telephone call 30 days after delivery to assess whether a surgical site infection had occurred, as well as any visits to a physician’s office or emergency department that were related to a wound complication.
The co-primary endpoints were superficial and deep incisional infections. Secondary endpoints included length of hospital stay; physician office visits; hospital readmissions for infection-related complications; endometritis; positive wound culture; skin irritation; and allergic reaction.
Surgical site infections occurred in 23 patients in the chlorhexidine group and 42 in the iodine group (4.0% vs. 7.3%) – a significant 45% risk reduction (relative risk, 0.55). Superficial infections were significantly less common in the chlorhexidine group (3.0% vs. 4.9%), as were deep infections (1.0% vs. 2.4%).
A subgroup analysis examined unscheduled vs. scheduled cesarean; obese vs. nonobese patients; suture vs. staple closure; diabetes vs. no diabetes; and chronic comorbidities vs. none. Chlorhexidine was significantly more effective than iodine in each of these groups.
Antiseptic type did not affect rates of skin separation, seroma, hematoma, or cellulitis. Nor did it affect the rates of endometritis, hospitalization for infectious complications, or length of hospital stay. However, those in the chlorhexidine group were significantly less likely to visit a physician for wound care (7.9% vs. 12.5%)
Cultures were obtained on 32 patients with a confirmed infection; 27 of these specimens were positive. About half of the positive cultures were polymicrobial. The most common isolate was Staphylococcus aureus (37%). Methicillin-resistant S. aureus (MRSA) was present in 12% of cultures in the chlorhexidine group and 17% in the iodine group.
In an interview, Dr. Tuuli said that chlorhexidine has several properties that make it more effective than iodine. It is effective against both gram-negative and gram-positive organisms, including MRSA, and is not inactivated by organic matter. Although chlorhexidine is more likely than iodine to provoke an allergic reaction, none were observed in this study.
The study was supported by a grant from the National Institutes of Health. Dr. Tuuli reported having no financial disclosures; the antiseptics were procured and paid for by the medical center.
Watch Dr. Tuuli discuss the study results here.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE PREGNANCY MEETING
Key clinical point: A chlorhexidine/alcohol antiseptic was significantly more effective at preventing cesarean section incision infections than an iodine/alcohol antiseptic.
Major finding: The chlorhexidine solution decreased surgical site infections by half, compared with the iodine-based solution.
Data source: A randomized study of 1,147 women who delivered via cesarean from 2011-2015.
Disclosures: The study was supported by a grant from the National Institutes of Health. Dr. Tuuli reported having no financial disclosures; the antiseptics were procured and paid for by the medical center.
Betamethasone improves respiratory outcomes in late preterm birth
ATLANTA – Administration of betamethasone to mothers at risk of late preterm birth reduced the risk of perinatal respiratory complications – including supplemental oxygen, ventilation, and death – by 20%.
The steroid was also associated with a 33% reduction in the risk of severe respiratory complications, Dr. Cynthia Gyamfi-Bannerman reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine. The study was simultaneously published in the New England Journal of Medicine (2016; doi: 10.1056/NEJMoa1516783.).
“This finding is practice changing and it’s ready for prime time,” Dr. Gyamfi-Bannerman said in an interview. “Right now there are 300,000 late preterm deliveries every year in this country, and if we can do something now to improve their outcomes without imparting any harm, then I think we should do that right away.”
Although standard practice for early preterm infants, betamethasone use in the late preterm period has not been well studied since the 1970s, said Dr. Gyamfi-Bannerman of Columbia University, New York. At that time, it was determined to be unhelpful; since then, studies have focused on its use in earlier time points. However, she said, those early studies were small and had methodologic problems.
Dr. Gyamfi-Bannerman’s study is the first large, placebo-controlled trial to examine the steroid’s use in the late preterm period. It was conducted in 17 centers and comprised 2,831 women with a singleton pregnancy of 34 to 36 weeks, 5 days’ gestation. All women were at high risk of delivery during the late preterm period, which was considered up to 36 weeks, 6 days.
Women were assigned to receive two injections of either 12 mg betamethasone or placebo 24 hours apart. The primary neonatal outcome was a composite of treatments required during the first 72 postnatal hours, including positive airway pressure or high-flow nasal cannula for at least 2 hours; supplemental oxygen for at least 4 hours; extracorporeal membrane oxygenation or mechanical ventilation; or stillbirth or neonatal death within that 72-hour period.
The women were a mean of 28 years old. Labor with intact membranes was the most common risk factor for preterm delivery; followed by ruptured membranes, and gestational diabetes or preeclampsia. Other risks included expected delivery for intrauterine growth restriction or oligohydramnios.
Outcomes data were available for 2,827 infants. There were no stillbirths or deaths within the first 72 hours.
The primary outcome was significantly less common among those who received betamethasone (11.6% vs. 14.4%; relative risk, 0.80). The number needed to treat to prevent one outcome was 35.
A secondary analysis eliminated 32 infants with a major congenital anomaly that had been unrecognized before birth; the significant benefit of betamethasone was unchanged.
The rate of the composite outcome of severe respiratory complications was also significantly lower in the betamethasone group (8.1% vs. 12.1%; RR, 0.67). The number needed to treat to prevent one case of a severe respiratory complication was 25.
While rates of respiratory distress syndrome, apnea, and pneumonia were similar in the two groups, other disorders were significantly less common in the treated group, including transient tachypnea of the newborn (6.7% vs. 9.9%); bronchopulmonary dysplasia (0.1% vs. 0.6%), and the composite of the respiratory distress syndrome, transient tachypnea of the newborn, or apnea (13.9% vs. 17.8%).
Significantly fewer infants in the treatment group required resuscitation at birth (14.5% vs. 18.7%) and surfactant (1.8% vs. 3.1%).
The use of betamethasone was not associated with any clinically significant adverse neonatal effects with one exception, Dr. Gyamfi-Bannerman said. Neonatal hypoglycemia occurred in 24%, compared with 15% of those in the placebo group – a significant 60% increased relative risk. There were no hypoglycemia-related adverse events; nor did the condition affect length of stay. In fact, infants with hypoglycemia were discharged an average of 2 days earlier than were those without “suggesting that the condition was self-limiting.” It was probably associated with earlier feeding in these babies, she added.
Late preterm birth is one of the largest risk factors for neonatal hypoglycemia, she noted.
“To put this in perspective, our numbers were similar or even less than those reported in the literature,” she said. “Our nurses are familiar with this risk and these babies are being checked regularly. We should be looking for it, in the way that we normally would.”
The study was supported by grants from the National Institutes of Health. Dr. Gyamfi-Bannerman reported having no financial disclosures.
Watch Dr. Gyamfi-Bannerman discuss the study here.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ATLANTA – Administration of betamethasone to mothers at risk of late preterm birth reduced the risk of perinatal respiratory complications – including supplemental oxygen, ventilation, and death – by 20%.
The steroid was also associated with a 33% reduction in the risk of severe respiratory complications, Dr. Cynthia Gyamfi-Bannerman reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine. The study was simultaneously published in the New England Journal of Medicine (2016; doi: 10.1056/NEJMoa1516783.).
“This finding is practice changing and it’s ready for prime time,” Dr. Gyamfi-Bannerman said in an interview. “Right now there are 300,000 late preterm deliveries every year in this country, and if we can do something now to improve their outcomes without imparting any harm, then I think we should do that right away.”
Although standard practice for early preterm infants, betamethasone use in the late preterm period has not been well studied since the 1970s, said Dr. Gyamfi-Bannerman of Columbia University, New York. At that time, it was determined to be unhelpful; since then, studies have focused on its use in earlier time points. However, she said, those early studies were small and had methodologic problems.
Dr. Gyamfi-Bannerman’s study is the first large, placebo-controlled trial to examine the steroid’s use in the late preterm period. It was conducted in 17 centers and comprised 2,831 women with a singleton pregnancy of 34 to 36 weeks, 5 days’ gestation. All women were at high risk of delivery during the late preterm period, which was considered up to 36 weeks, 6 days.
Women were assigned to receive two injections of either 12 mg betamethasone or placebo 24 hours apart. The primary neonatal outcome was a composite of treatments required during the first 72 postnatal hours, including positive airway pressure or high-flow nasal cannula for at least 2 hours; supplemental oxygen for at least 4 hours; extracorporeal membrane oxygenation or mechanical ventilation; or stillbirth or neonatal death within that 72-hour period.
The women were a mean of 28 years old. Labor with intact membranes was the most common risk factor for preterm delivery; followed by ruptured membranes, and gestational diabetes or preeclampsia. Other risks included expected delivery for intrauterine growth restriction or oligohydramnios.
Outcomes data were available for 2,827 infants. There were no stillbirths or deaths within the first 72 hours.
The primary outcome was significantly less common among those who received betamethasone (11.6% vs. 14.4%; relative risk, 0.80). The number needed to treat to prevent one outcome was 35.
A secondary analysis eliminated 32 infants with a major congenital anomaly that had been unrecognized before birth; the significant benefit of betamethasone was unchanged.
The rate of the composite outcome of severe respiratory complications was also significantly lower in the betamethasone group (8.1% vs. 12.1%; RR, 0.67). The number needed to treat to prevent one case of a severe respiratory complication was 25.
While rates of respiratory distress syndrome, apnea, and pneumonia were similar in the two groups, other disorders were significantly less common in the treated group, including transient tachypnea of the newborn (6.7% vs. 9.9%); bronchopulmonary dysplasia (0.1% vs. 0.6%), and the composite of the respiratory distress syndrome, transient tachypnea of the newborn, or apnea (13.9% vs. 17.8%).
Significantly fewer infants in the treatment group required resuscitation at birth (14.5% vs. 18.7%) and surfactant (1.8% vs. 3.1%).
The use of betamethasone was not associated with any clinically significant adverse neonatal effects with one exception, Dr. Gyamfi-Bannerman said. Neonatal hypoglycemia occurred in 24%, compared with 15% of those in the placebo group – a significant 60% increased relative risk. There were no hypoglycemia-related adverse events; nor did the condition affect length of stay. In fact, infants with hypoglycemia were discharged an average of 2 days earlier than were those without “suggesting that the condition was self-limiting.” It was probably associated with earlier feeding in these babies, she added.
Late preterm birth is one of the largest risk factors for neonatal hypoglycemia, she noted.
“To put this in perspective, our numbers were similar or even less than those reported in the literature,” she said. “Our nurses are familiar with this risk and these babies are being checked regularly. We should be looking for it, in the way that we normally would.”
The study was supported by grants from the National Institutes of Health. Dr. Gyamfi-Bannerman reported having no financial disclosures.
Watch Dr. Gyamfi-Bannerman discuss the study here.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ATLANTA – Administration of betamethasone to mothers at risk of late preterm birth reduced the risk of perinatal respiratory complications – including supplemental oxygen, ventilation, and death – by 20%.
The steroid was also associated with a 33% reduction in the risk of severe respiratory complications, Dr. Cynthia Gyamfi-Bannerman reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine. The study was simultaneously published in the New England Journal of Medicine (2016; doi: 10.1056/NEJMoa1516783.).
“This finding is practice changing and it’s ready for prime time,” Dr. Gyamfi-Bannerman said in an interview. “Right now there are 300,000 late preterm deliveries every year in this country, and if we can do something now to improve their outcomes without imparting any harm, then I think we should do that right away.”
Although standard practice for early preterm infants, betamethasone use in the late preterm period has not been well studied since the 1970s, said Dr. Gyamfi-Bannerman of Columbia University, New York. At that time, it was determined to be unhelpful; since then, studies have focused on its use in earlier time points. However, she said, those early studies were small and had methodologic problems.
Dr. Gyamfi-Bannerman’s study is the first large, placebo-controlled trial to examine the steroid’s use in the late preterm period. It was conducted in 17 centers and comprised 2,831 women with a singleton pregnancy of 34 to 36 weeks, 5 days’ gestation. All women were at high risk of delivery during the late preterm period, which was considered up to 36 weeks, 6 days.
Women were assigned to receive two injections of either 12 mg betamethasone or placebo 24 hours apart. The primary neonatal outcome was a composite of treatments required during the first 72 postnatal hours, including positive airway pressure or high-flow nasal cannula for at least 2 hours; supplemental oxygen for at least 4 hours; extracorporeal membrane oxygenation or mechanical ventilation; or stillbirth or neonatal death within that 72-hour period.
The women were a mean of 28 years old. Labor with intact membranes was the most common risk factor for preterm delivery; followed by ruptured membranes, and gestational diabetes or preeclampsia. Other risks included expected delivery for intrauterine growth restriction or oligohydramnios.
Outcomes data were available for 2,827 infants. There were no stillbirths or deaths within the first 72 hours.
The primary outcome was significantly less common among those who received betamethasone (11.6% vs. 14.4%; relative risk, 0.80). The number needed to treat to prevent one outcome was 35.
A secondary analysis eliminated 32 infants with a major congenital anomaly that had been unrecognized before birth; the significant benefit of betamethasone was unchanged.
The rate of the composite outcome of severe respiratory complications was also significantly lower in the betamethasone group (8.1% vs. 12.1%; RR, 0.67). The number needed to treat to prevent one case of a severe respiratory complication was 25.
While rates of respiratory distress syndrome, apnea, and pneumonia were similar in the two groups, other disorders were significantly less common in the treated group, including transient tachypnea of the newborn (6.7% vs. 9.9%); bronchopulmonary dysplasia (0.1% vs. 0.6%), and the composite of the respiratory distress syndrome, transient tachypnea of the newborn, or apnea (13.9% vs. 17.8%).
Significantly fewer infants in the treatment group required resuscitation at birth (14.5% vs. 18.7%) and surfactant (1.8% vs. 3.1%).
The use of betamethasone was not associated with any clinically significant adverse neonatal effects with one exception, Dr. Gyamfi-Bannerman said. Neonatal hypoglycemia occurred in 24%, compared with 15% of those in the placebo group – a significant 60% increased relative risk. There were no hypoglycemia-related adverse events; nor did the condition affect length of stay. In fact, infants with hypoglycemia were discharged an average of 2 days earlier than were those without “suggesting that the condition was self-limiting.” It was probably associated with earlier feeding in these babies, she added.
Late preterm birth is one of the largest risk factors for neonatal hypoglycemia, she noted.
“To put this in perspective, our numbers were similar or even less than those reported in the literature,” she said. “Our nurses are familiar with this risk and these babies are being checked regularly. We should be looking for it, in the way that we normally would.”
The study was supported by grants from the National Institutes of Health. Dr. Gyamfi-Bannerman reported having no financial disclosures.
Watch Dr. Gyamfi-Bannerman discuss the study here.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE PREGNANCY MEETING
Key clinical point: Betamethasone improves neonatal respiratory outcomes in late preterm births.
Major finding: When administered in the later preterm period, betamethasone reduced the risk of severe perinatal respiratory complications by 20%, compared with placebo.
Data source: A randomized trial of 2,831 women with a singleton pregnancy.
Disclosures: The study was supported by grants from the National Institutes of Health. Dr. Gyamfi-Bannerman reported having no financial disclosures.
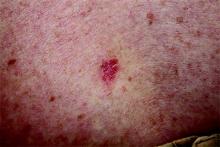
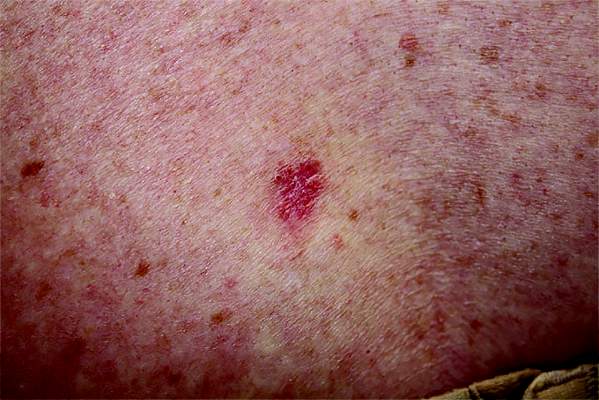
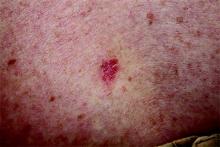
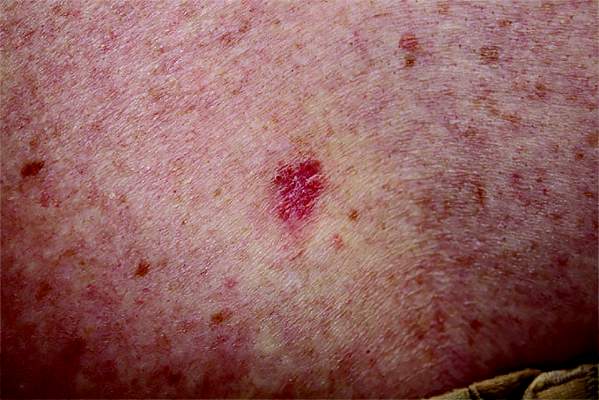
Organ Transplant Recipients Face Increased Risk for BCC
Recipients of a solid organ transplant face up to a sixfold increase in the risk of developing a basal cell carcinoma – a risk that seems to increase as time passes.
A pretransplant history of squamous cell carcinoma (SCC) increased this risk to 55 times that seen in the general population, Dr. Britta Krynitz and her colleagues reported (Br J Dermatol. 2015. doi: 10.1111/bjd.14153).
But even when the pretransplant SCC group was removed from the final analysis, the risk of basal cell carcinoma after transplant was five times that of the general population, “indicating that a pretransplant SCC has limited effect on BCC risk overall and that organ transplantation per se is a strong driver of posttransplant BCC risk,” wrote Dr. Krynitz of Karolinska Institute, Stockholm, and her coauthors.
“Our results strongly suggest tumor promoter effects of the immunosuppressive drugs in the pathogenesis of post-transplantation BCC,” the team said. “We speculate that calcineurin inhibitors and also antiproliferative drugs, often used in combination with corticosteroids, play a role.”
The researchers investigated the incidence of both BCC and SCC in a cohort of 4,023 patients who underwent solid organ transplant from 2004 to 2011. Their median age at the time of transplant was 53 years; most (59%) received a kidney. Other organs transplanted were liver (22%), heart and/or lung (15%), and other organs (4%). The median follow-up time was 3.4 years; the longest follow-up was 5.5 years.
Only 17 of patients had a history of melanoma, and 19 patients a history of SCC – less than 1% for each skin cancer. Seven percent (301) of patients had experienced some form of nonskin cancer.
By the end of follow-up, 341 BCCs had developed among 175 patients – an incidence of 6.7%. About half developed more than one BCC.
The researchers compared these patients to a group of almost 200,000 nontransplant patients who had developed BCC. Among these, the median age at BCC appearance was significantly older (71 years); 39% had more than one lesion.
The overall relative risk of BCC was increased sixfold in transplant recipients and was similar between the genders. However, the risk varied according to the type of organ received. Kidney recipients were at the highest risk (relative risk, 7.2), and those who received other organs had a lower risk (heart/lung: RR, 5.8; liver: RR, 2.6).
The risk also appeared to increase over time, the authors noted. From 0 to 2 years, it was 5.8; from 3 to 5 years, it increased to 7.0.
Among men, 54% of lesions appeared in the head/neck area and 35% on the trunk – a similar distribution to that seen in the nontransplant control group. Among women, there were differences between transplant patients and controls: 44% of lesions appeared on patients’ head/neck, compared with 60% in the control group, and 34% appeared on the truck, compared with 24% in the control group.
Histology was similar, as were the proportions of aggressive type II and highly aggressive type III lesions.
A total of 199 SCCs developed among 87 patients during follow-up, a ratio to BCC of 1:1.7. “The low ratio was probably due to the short follow-up in our study,” the authors noted.
The Welander Foundation, the Westerberg Foundation, and the Strategic Research Program in Epidemiology at Karolinska Institute sponsored the study. None of the authors had any financial declarations.
Recipients of a solid organ transplant face up to a sixfold increase in the risk of developing a basal cell carcinoma – a risk that seems to increase as time passes.
A pretransplant history of squamous cell carcinoma (SCC) increased this risk to 55 times that seen in the general population, Dr. Britta Krynitz and her colleagues reported (Br J Dermatol. 2015. doi: 10.1111/bjd.14153).
But even when the pretransplant SCC group was removed from the final analysis, the risk of basal cell carcinoma after transplant was five times that of the general population, “indicating that a pretransplant SCC has limited effect on BCC risk overall and that organ transplantation per se is a strong driver of posttransplant BCC risk,” wrote Dr. Krynitz of Karolinska Institute, Stockholm, and her coauthors.
“Our results strongly suggest tumor promoter effects of the immunosuppressive drugs in the pathogenesis of post-transplantation BCC,” the team said. “We speculate that calcineurin inhibitors and also antiproliferative drugs, often used in combination with corticosteroids, play a role.”
The researchers investigated the incidence of both BCC and SCC in a cohort of 4,023 patients who underwent solid organ transplant from 2004 to 2011. Their median age at the time of transplant was 53 years; most (59%) received a kidney. Other organs transplanted were liver (22%), heart and/or lung (15%), and other organs (4%). The median follow-up time was 3.4 years; the longest follow-up was 5.5 years.
Only 17 of patients had a history of melanoma, and 19 patients a history of SCC – less than 1% for each skin cancer. Seven percent (301) of patients had experienced some form of nonskin cancer.
By the end of follow-up, 341 BCCs had developed among 175 patients – an incidence of 6.7%. About half developed more than one BCC.
The researchers compared these patients to a group of almost 200,000 nontransplant patients who had developed BCC. Among these, the median age at BCC appearance was significantly older (71 years); 39% had more than one lesion.
The overall relative risk of BCC was increased sixfold in transplant recipients and was similar between the genders. However, the risk varied according to the type of organ received. Kidney recipients were at the highest risk (relative risk, 7.2), and those who received other organs had a lower risk (heart/lung: RR, 5.8; liver: RR, 2.6).
The risk also appeared to increase over time, the authors noted. From 0 to 2 years, it was 5.8; from 3 to 5 years, it increased to 7.0.
Among men, 54% of lesions appeared in the head/neck area and 35% on the trunk – a similar distribution to that seen in the nontransplant control group. Among women, there were differences between transplant patients and controls: 44% of lesions appeared on patients’ head/neck, compared with 60% in the control group, and 34% appeared on the truck, compared with 24% in the control group.
Histology was similar, as were the proportions of aggressive type II and highly aggressive type III lesions.
A total of 199 SCCs developed among 87 patients during follow-up, a ratio to BCC of 1:1.7. “The low ratio was probably due to the short follow-up in our study,” the authors noted.
The Welander Foundation, the Westerberg Foundation, and the Strategic Research Program in Epidemiology at Karolinska Institute sponsored the study. None of the authors had any financial declarations.
Recipients of a solid organ transplant face up to a sixfold increase in the risk of developing a basal cell carcinoma – a risk that seems to increase as time passes.
A pretransplant history of squamous cell carcinoma (SCC) increased this risk to 55 times that seen in the general population, Dr. Britta Krynitz and her colleagues reported (Br J Dermatol. 2015. doi: 10.1111/bjd.14153).
But even when the pretransplant SCC group was removed from the final analysis, the risk of basal cell carcinoma after transplant was five times that of the general population, “indicating that a pretransplant SCC has limited effect on BCC risk overall and that organ transplantation per se is a strong driver of posttransplant BCC risk,” wrote Dr. Krynitz of Karolinska Institute, Stockholm, and her coauthors.
“Our results strongly suggest tumor promoter effects of the immunosuppressive drugs in the pathogenesis of post-transplantation BCC,” the team said. “We speculate that calcineurin inhibitors and also antiproliferative drugs, often used in combination with corticosteroids, play a role.”
The researchers investigated the incidence of both BCC and SCC in a cohort of 4,023 patients who underwent solid organ transplant from 2004 to 2011. Their median age at the time of transplant was 53 years; most (59%) received a kidney. Other organs transplanted were liver (22%), heart and/or lung (15%), and other organs (4%). The median follow-up time was 3.4 years; the longest follow-up was 5.5 years.
Only 17 of patients had a history of melanoma, and 19 patients a history of SCC – less than 1% for each skin cancer. Seven percent (301) of patients had experienced some form of nonskin cancer.
By the end of follow-up, 341 BCCs had developed among 175 patients – an incidence of 6.7%. About half developed more than one BCC.
The researchers compared these patients to a group of almost 200,000 nontransplant patients who had developed BCC. Among these, the median age at BCC appearance was significantly older (71 years); 39% had more than one lesion.
The overall relative risk of BCC was increased sixfold in transplant recipients and was similar between the genders. However, the risk varied according to the type of organ received. Kidney recipients were at the highest risk (relative risk, 7.2), and those who received other organs had a lower risk (heart/lung: RR, 5.8; liver: RR, 2.6).
The risk also appeared to increase over time, the authors noted. From 0 to 2 years, it was 5.8; from 3 to 5 years, it increased to 7.0.
Among men, 54% of lesions appeared in the head/neck area and 35% on the trunk – a similar distribution to that seen in the nontransplant control group. Among women, there were differences between transplant patients and controls: 44% of lesions appeared on patients’ head/neck, compared with 60% in the control group, and 34% appeared on the truck, compared with 24% in the control group.
Histology was similar, as were the proportions of aggressive type II and highly aggressive type III lesions.
A total of 199 SCCs developed among 87 patients during follow-up, a ratio to BCC of 1:1.7. “The low ratio was probably due to the short follow-up in our study,” the authors noted.
The Welander Foundation, the Westerberg Foundation, and the Strategic Research Program in Epidemiology at Karolinska Institute sponsored the study. None of the authors had any financial declarations.
FROM BRITISH JOURNAL OF DERMATOLOGY
Organ transplant recipients face increased risk of BCC
Recipients of a solid organ transplant face up to a sixfold increase in the risk of developing a basal cell carcinoma – a risk that seems to increase as time passes.
A pretransplant history of squamous cell carcinoma (SCC) increased this risk to 55 times that seen in the general population, Dr. Britta Krynitz and her colleagues reported (Br J Dermatol. 2015. doi: 10.1111/bjd.14153).
But even when the pretransplant SCC group was removed from the final analysis, the risk of basal cell carcinoma after transplant was five times that of the general population, “indicating that a pretransplant SCC has limited effect on BCC risk overall and that organ transplantation per se is a strong driver of posttransplant BCC risk,” wrote Dr. Krynitz of Karolinska Institute, Stockholm, and her coauthors.
“Our results strongly suggest tumor promoter effects of the immunosuppressive drugs in the pathogenesis of post-transplantation BCC,” the team said. “We speculate that calcineurin inhibitors and also antiproliferative drugs, often used in combination with corticosteroids, play a role.”
The researchers investigated the incidence of both BCC and SCC in a cohort of 4,023 patients who underwent solid organ transplant from 2004 to 2011. Their median age at the time of transplant was 53 years; most (59%) received a kidney. Other organs transplanted were liver (22%), heart and/or lung (15%), and other organs (4%). The median follow-up time was 3.4 years; the longest follow-up was 5.5 years.
Only 17 of patients had a history of melanoma, and 19 patients a history of SCC – less than 1% for each skin cancer. Seven percent (301) of patients had experienced some form of nonskin cancer.
By the end of follow-up, 341 BCCs had developed among 175 patients – an incidence of 6.7%. About half developed more than one BCC.
The researchers compared these patients to a group of almost 200,000 nontransplant patients who had developed BCC. Among these, the median age at BCC appearance was significantly older (71 years); 39% had more than one lesion.
The overall relative risk of BCC was increased sixfold in transplant recipients and was similar between the genders. However, the risk varied according to the type of organ received. Kidney recipients were at the highest risk (relative risk, 7.2), and those who received other organs had a lower risk (heart/lung: RR, 5.8; liver: RR, 2.6).
The risk also appeared to increase over time, the authors noted. From 0 to 2 years, it was 5.8; from 3 to 5 years, it increased to 7.0.
Among men, 54% of lesions appeared in the head/neck area and 35% on the trunk – a similar distribution to that seen in the nontransplant control group. Among women, there were differences between transplant patients and controls: 44% of lesions appeared on patients’ head/neck, compared with 60% in the control group, and 34% appeared on the truck, compared with 24% in the control group.
Histology was similar, as were the proportions of aggressive type II and highly aggressive type III lesions.
A total of 199 SCCs developed among 87 patients during follow-up, a ratio to BCC of 1:1.7. “The low ratio was probably due to the short follow-up in our study,” the authors noted.
The Welander Foundation, the Westerberg Foundation, and the Strategic Research Program in Epidemiology at Karolinska Institute sponsored the study. None of the authors had any financial declarations.
Recipients of a solid organ transplant face up to a sixfold increase in the risk of developing a basal cell carcinoma – a risk that seems to increase as time passes.
A pretransplant history of squamous cell carcinoma (SCC) increased this risk to 55 times that seen in the general population, Dr. Britta Krynitz and her colleagues reported (Br J Dermatol. 2015. doi: 10.1111/bjd.14153).
But even when the pretransplant SCC group was removed from the final analysis, the risk of basal cell carcinoma after transplant was five times that of the general population, “indicating that a pretransplant SCC has limited effect on BCC risk overall and that organ transplantation per se is a strong driver of posttransplant BCC risk,” wrote Dr. Krynitz of Karolinska Institute, Stockholm, and her coauthors.
“Our results strongly suggest tumor promoter effects of the immunosuppressive drugs in the pathogenesis of post-transplantation BCC,” the team said. “We speculate that calcineurin inhibitors and also antiproliferative drugs, often used in combination with corticosteroids, play a role.”
The researchers investigated the incidence of both BCC and SCC in a cohort of 4,023 patients who underwent solid organ transplant from 2004 to 2011. Their median age at the time of transplant was 53 years; most (59%) received a kidney. Other organs transplanted were liver (22%), heart and/or lung (15%), and other organs (4%). The median follow-up time was 3.4 years; the longest follow-up was 5.5 years.
Only 17 of patients had a history of melanoma, and 19 patients a history of SCC – less than 1% for each skin cancer. Seven percent (301) of patients had experienced some form of nonskin cancer.
By the end of follow-up, 341 BCCs had developed among 175 patients – an incidence of 6.7%. About half developed more than one BCC.
The researchers compared these patients to a group of almost 200,000 nontransplant patients who had developed BCC. Among these, the median age at BCC appearance was significantly older (71 years); 39% had more than one lesion.
The overall relative risk of BCC was increased sixfold in transplant recipients and was similar between the genders. However, the risk varied according to the type of organ received. Kidney recipients were at the highest risk (relative risk, 7.2), and those who received other organs had a lower risk (heart/lung: RR, 5.8; liver: RR, 2.6).
The risk also appeared to increase over time, the authors noted. From 0 to 2 years, it was 5.8; from 3 to 5 years, it increased to 7.0.
Among men, 54% of lesions appeared in the head/neck area and 35% on the trunk – a similar distribution to that seen in the nontransplant control group. Among women, there were differences between transplant patients and controls: 44% of lesions appeared on patients’ head/neck, compared with 60% in the control group, and 34% appeared on the truck, compared with 24% in the control group.
Histology was similar, as were the proportions of aggressive type II and highly aggressive type III lesions.
A total of 199 SCCs developed among 87 patients during follow-up, a ratio to BCC of 1:1.7. “The low ratio was probably due to the short follow-up in our study,” the authors noted.
The Welander Foundation, the Westerberg Foundation, and the Strategic Research Program in Epidemiology at Karolinska Institute sponsored the study. None of the authors had any financial declarations.
Recipients of a solid organ transplant face up to a sixfold increase in the risk of developing a basal cell carcinoma – a risk that seems to increase as time passes.
A pretransplant history of squamous cell carcinoma (SCC) increased this risk to 55 times that seen in the general population, Dr. Britta Krynitz and her colleagues reported (Br J Dermatol. 2015. doi: 10.1111/bjd.14153).
But even when the pretransplant SCC group was removed from the final analysis, the risk of basal cell carcinoma after transplant was five times that of the general population, “indicating that a pretransplant SCC has limited effect on BCC risk overall and that organ transplantation per se is a strong driver of posttransplant BCC risk,” wrote Dr. Krynitz of Karolinska Institute, Stockholm, and her coauthors.
“Our results strongly suggest tumor promoter effects of the immunosuppressive drugs in the pathogenesis of post-transplantation BCC,” the team said. “We speculate that calcineurin inhibitors and also antiproliferative drugs, often used in combination with corticosteroids, play a role.”
The researchers investigated the incidence of both BCC and SCC in a cohort of 4,023 patients who underwent solid organ transplant from 2004 to 2011. Their median age at the time of transplant was 53 years; most (59%) received a kidney. Other organs transplanted were liver (22%), heart and/or lung (15%), and other organs (4%). The median follow-up time was 3.4 years; the longest follow-up was 5.5 years.
Only 17 of patients had a history of melanoma, and 19 patients a history of SCC – less than 1% for each skin cancer. Seven percent (301) of patients had experienced some form of nonskin cancer.
By the end of follow-up, 341 BCCs had developed among 175 patients – an incidence of 6.7%. About half developed more than one BCC.
The researchers compared these patients to a group of almost 200,000 nontransplant patients who had developed BCC. Among these, the median age at BCC appearance was significantly older (71 years); 39% had more than one lesion.
The overall relative risk of BCC was increased sixfold in transplant recipients and was similar between the genders. However, the risk varied according to the type of organ received. Kidney recipients were at the highest risk (relative risk, 7.2), and those who received other organs had a lower risk (heart/lung: RR, 5.8; liver: RR, 2.6).
The risk also appeared to increase over time, the authors noted. From 0 to 2 years, it was 5.8; from 3 to 5 years, it increased to 7.0.
Among men, 54% of lesions appeared in the head/neck area and 35% on the trunk – a similar distribution to that seen in the nontransplant control group. Among women, there were differences between transplant patients and controls: 44% of lesions appeared on patients’ head/neck, compared with 60% in the control group, and 34% appeared on the truck, compared with 24% in the control group.
Histology was similar, as were the proportions of aggressive type II and highly aggressive type III lesions.
A total of 199 SCCs developed among 87 patients during follow-up, a ratio to BCC of 1:1.7. “The low ratio was probably due to the short follow-up in our study,” the authors noted.
The Welander Foundation, the Westerberg Foundation, and the Strategic Research Program in Epidemiology at Karolinska Institute sponsored the study. None of the authors had any financial declarations.
FROM BRITISH JOURNAL OF DERMATOLOGY
Key clinical point: The risk of a basal cell carcinoma increases after solid organ transplant.
Major finding: Transplant patients have a sixfold increased risk of BCC.
Data source: A retrospective database study of 4,000 transplant patients and almost 200,000 controls.
Disclosures: The Welander Foundation, the Westerberg Foundation, and the Strategic Research Program in Epidemiology at Karolinska Institute sponsored the study. None of the authors had any financial declarations.
Zika could soon infect 4 million; U.S. impact likely to be much smaller
The Zika virus continues to spread, with the potential to infect up to 4 million people throughout the Americas.
But although locally transmitted infections are all almost certainly inevitable in the United States, federal health officials don’t expect large-scale infections.
“We do expect to see local transmission, but we are not likely to see widespread outbreaks,” Dr. Anthony S. Fauci, director of the National Institute of Allergy and Infectious Diseases, said during a press briefing on Jan. 28. “We think these would occur in pockets, similar to what we now see with dengue and chikungunya. We are not being cavalier about this; we are preparing. But large-scale outbreaks are not something we are likely to see, based on our experience with dengue and chikungunya.”
Dr. Fauci’s comments were in contrast to warnings issued simultaneously by the World Health Organization and the Pan American Health Organization (PAHO), WHO’s South American arm.
On Jan. 28, the WHO announced it would convene a meeting of its International Health Regulations Emergency Committee. WHO Director-General Margaret Chan said the virus is now present in 23 countries and territories in the region, as well as in parts of the South Pacific.
“The level of alarm is extremely high,” she said during the meeting. Although a causal link between the virus and outbreaks of microcephaly and Guillain-Barré syndrome is yet unproven, she said it is strongly suspected. “The possible links, only recently suspected, have rapidly changed the risk profile of Zika, from a mild threat to one of alarming proportions.”
Dr. Marcos Espinal, PAHO’s director of communicable diseases and health analysis, outlined the potential spread. His agency expects to see 3-4 million cases throughout North and South America, which could occur in any region that has endemic dengue fever. Both viruses are carried by mosquitoes of the Aedes genus. Two of these, the Asian tiger mosquito and the yellow fever mosquito, are commonly found in the United States, particularly in warmer regions.
Thus far, 31 cases of Zika virus have been confirmed in the United States (11 states and Washington), Dr. Anne Schuchat, principal deputy director for the Centers for Disease Control and Prevention, said in that agency’s briefing. All of these cases have been associated with travel to endemic areas, she said. She was unable to say how many cases had occurred in pregnant women.
There have been 19 confirmed cases in Puerto Rico and 1 in the U.S. Virgin Islands. These could be locally acquired cases, although that has not been confirmed. However, Zika infection is now a notifiable illness and any confirmed cases must be reported to CDC, she said.
Nearly a million cases have been laboratory confirmed in South America since 2015, Dr. Espinal noted. The vast majority of those have caused mild, transient symptoms. But in Brazil alone – the epicenter of the epidemic – there has been a substantial increase in newborn microcephaly. Almost 4,000 cases of this rare birth defect have been identified since October, when tracking began. Although the cases coincide with the rise of Zika infections, no one knows how many were related or what pathology could be mediating the association.
This is just one question muddying the Zika waters right now, Dr. Chan of the WHO said. Several difficult-to-address issues are hampering an effective response, including the widespread range of the mosquito vectors, which allows international spread; a lack of population immunity; and the absence of rapid diagnostic tests, effective treatment, and any vaccine.
Another concern is the potential for illness-potentiating coinfection. In the lab, Zika virus has shown cross-reactions with dengue types 1-4, yellow fever, and West Nile virus – all of which are carried by Aedes mosquitoes.
Scientists at the CDC and National Institutes of Health are working on the problem now, Dr. Fauci said. Efforts include the creation of animal models to study disease transmission and its effect on pre- and postnatal outcomes, and of diagnostic platforms that could easily and quickly identify Zika infections. These would quickly differentiate Zika from dengue infections, which are also caused by a strain of flavivirus.
An effective vaccine will not be quickly forthcoming, he cautioned, although a phase I trial is in the works and could begin later this year. The candidate is a DNA-based vaccine similar to the one now used to protect against dengue. It would almost certainly not be approved in less than 2 years, Dr Fauci added.
Until some of these questions have been answered, or until the epidemic slows, the CDC continues to caution pregnant women against travel to endemic areas. For those who do travel or who live in endemic areas, Dr. Schuchat reiterated CDC’s long-held advice to take precautions against mosquito bites: long-sleeved shirts and long pants; staying inside; and using an effective insect repellent. Those containing DEET are most effective and are safe for pregnant women to use, Dr. Schuchat said.
For now, the CDC’s Zika travel alert includes Puerto Rico, Barbados, Bolivia, Brazil, Cape Verde, Colombia, Ecuador, El Salvador, French Guiana, Guadeloupe, Guatemala, Guyana, Haiti, Honduras, Martinique, Mexico, Panama, Paraguay, Saint Martin, Samoa, Suriname, Venezuela, the U.S. Virgin Islands, and the Dominican Republic.
A transcript of the press briefing is available online.
This article was updated 2/1/2016.
On Twitter @Alz_Gal
The Zika virus continues to spread, with the potential to infect up to 4 million people throughout the Americas.
But although locally transmitted infections are all almost certainly inevitable in the United States, federal health officials don’t expect large-scale infections.
“We do expect to see local transmission, but we are not likely to see widespread outbreaks,” Dr. Anthony S. Fauci, director of the National Institute of Allergy and Infectious Diseases, said during a press briefing on Jan. 28. “We think these would occur in pockets, similar to what we now see with dengue and chikungunya. We are not being cavalier about this; we are preparing. But large-scale outbreaks are not something we are likely to see, based on our experience with dengue and chikungunya.”
Dr. Fauci’s comments were in contrast to warnings issued simultaneously by the World Health Organization and the Pan American Health Organization (PAHO), WHO’s South American arm.
On Jan. 28, the WHO announced it would convene a meeting of its International Health Regulations Emergency Committee. WHO Director-General Margaret Chan said the virus is now present in 23 countries and territories in the region, as well as in parts of the South Pacific.
“The level of alarm is extremely high,” she said during the meeting. Although a causal link between the virus and outbreaks of microcephaly and Guillain-Barré syndrome is yet unproven, she said it is strongly suspected. “The possible links, only recently suspected, have rapidly changed the risk profile of Zika, from a mild threat to one of alarming proportions.”
Dr. Marcos Espinal, PAHO’s director of communicable diseases and health analysis, outlined the potential spread. His agency expects to see 3-4 million cases throughout North and South America, which could occur in any region that has endemic dengue fever. Both viruses are carried by mosquitoes of the Aedes genus. Two of these, the Asian tiger mosquito and the yellow fever mosquito, are commonly found in the United States, particularly in warmer regions.
Thus far, 31 cases of Zika virus have been confirmed in the United States (11 states and Washington), Dr. Anne Schuchat, principal deputy director for the Centers for Disease Control and Prevention, said in that agency’s briefing. All of these cases have been associated with travel to endemic areas, she said. She was unable to say how many cases had occurred in pregnant women.
There have been 19 confirmed cases in Puerto Rico and 1 in the U.S. Virgin Islands. These could be locally acquired cases, although that has not been confirmed. However, Zika infection is now a notifiable illness and any confirmed cases must be reported to CDC, she said.
Nearly a million cases have been laboratory confirmed in South America since 2015, Dr. Espinal noted. The vast majority of those have caused mild, transient symptoms. But in Brazil alone – the epicenter of the epidemic – there has been a substantial increase in newborn microcephaly. Almost 4,000 cases of this rare birth defect have been identified since October, when tracking began. Although the cases coincide with the rise of Zika infections, no one knows how many were related or what pathology could be mediating the association.
This is just one question muddying the Zika waters right now, Dr. Chan of the WHO said. Several difficult-to-address issues are hampering an effective response, including the widespread range of the mosquito vectors, which allows international spread; a lack of population immunity; and the absence of rapid diagnostic tests, effective treatment, and any vaccine.
Another concern is the potential for illness-potentiating coinfection. In the lab, Zika virus has shown cross-reactions with dengue types 1-4, yellow fever, and West Nile virus – all of which are carried by Aedes mosquitoes.
Scientists at the CDC and National Institutes of Health are working on the problem now, Dr. Fauci said. Efforts include the creation of animal models to study disease transmission and its effect on pre- and postnatal outcomes, and of diagnostic platforms that could easily and quickly identify Zika infections. These would quickly differentiate Zika from dengue infections, which are also caused by a strain of flavivirus.
An effective vaccine will not be quickly forthcoming, he cautioned, although a phase I trial is in the works and could begin later this year. The candidate is a DNA-based vaccine similar to the one now used to protect against dengue. It would almost certainly not be approved in less than 2 years, Dr Fauci added.
Until some of these questions have been answered, or until the epidemic slows, the CDC continues to caution pregnant women against travel to endemic areas. For those who do travel or who live in endemic areas, Dr. Schuchat reiterated CDC’s long-held advice to take precautions against mosquito bites: long-sleeved shirts and long pants; staying inside; and using an effective insect repellent. Those containing DEET are most effective and are safe for pregnant women to use, Dr. Schuchat said.
For now, the CDC’s Zika travel alert includes Puerto Rico, Barbados, Bolivia, Brazil, Cape Verde, Colombia, Ecuador, El Salvador, French Guiana, Guadeloupe, Guatemala, Guyana, Haiti, Honduras, Martinique, Mexico, Panama, Paraguay, Saint Martin, Samoa, Suriname, Venezuela, the U.S. Virgin Islands, and the Dominican Republic.
A transcript of the press briefing is available online.
This article was updated 2/1/2016.
On Twitter @Alz_Gal
The Zika virus continues to spread, with the potential to infect up to 4 million people throughout the Americas.
But although locally transmitted infections are all almost certainly inevitable in the United States, federal health officials don’t expect large-scale infections.
“We do expect to see local transmission, but we are not likely to see widespread outbreaks,” Dr. Anthony S. Fauci, director of the National Institute of Allergy and Infectious Diseases, said during a press briefing on Jan. 28. “We think these would occur in pockets, similar to what we now see with dengue and chikungunya. We are not being cavalier about this; we are preparing. But large-scale outbreaks are not something we are likely to see, based on our experience with dengue and chikungunya.”
Dr. Fauci’s comments were in contrast to warnings issued simultaneously by the World Health Organization and the Pan American Health Organization (PAHO), WHO’s South American arm.
On Jan. 28, the WHO announced it would convene a meeting of its International Health Regulations Emergency Committee. WHO Director-General Margaret Chan said the virus is now present in 23 countries and territories in the region, as well as in parts of the South Pacific.
“The level of alarm is extremely high,” she said during the meeting. Although a causal link between the virus and outbreaks of microcephaly and Guillain-Barré syndrome is yet unproven, she said it is strongly suspected. “The possible links, only recently suspected, have rapidly changed the risk profile of Zika, from a mild threat to one of alarming proportions.”
Dr. Marcos Espinal, PAHO’s director of communicable diseases and health analysis, outlined the potential spread. His agency expects to see 3-4 million cases throughout North and South America, which could occur in any region that has endemic dengue fever. Both viruses are carried by mosquitoes of the Aedes genus. Two of these, the Asian tiger mosquito and the yellow fever mosquito, are commonly found in the United States, particularly in warmer regions.
Thus far, 31 cases of Zika virus have been confirmed in the United States (11 states and Washington), Dr. Anne Schuchat, principal deputy director for the Centers for Disease Control and Prevention, said in that agency’s briefing. All of these cases have been associated with travel to endemic areas, she said. She was unable to say how many cases had occurred in pregnant women.
There have been 19 confirmed cases in Puerto Rico and 1 in the U.S. Virgin Islands. These could be locally acquired cases, although that has not been confirmed. However, Zika infection is now a notifiable illness and any confirmed cases must be reported to CDC, she said.
Nearly a million cases have been laboratory confirmed in South America since 2015, Dr. Espinal noted. The vast majority of those have caused mild, transient symptoms. But in Brazil alone – the epicenter of the epidemic – there has been a substantial increase in newborn microcephaly. Almost 4,000 cases of this rare birth defect have been identified since October, when tracking began. Although the cases coincide with the rise of Zika infections, no one knows how many were related or what pathology could be mediating the association.
This is just one question muddying the Zika waters right now, Dr. Chan of the WHO said. Several difficult-to-address issues are hampering an effective response, including the widespread range of the mosquito vectors, which allows international spread; a lack of population immunity; and the absence of rapid diagnostic tests, effective treatment, and any vaccine.
Another concern is the potential for illness-potentiating coinfection. In the lab, Zika virus has shown cross-reactions with dengue types 1-4, yellow fever, and West Nile virus – all of which are carried by Aedes mosquitoes.
Scientists at the CDC and National Institutes of Health are working on the problem now, Dr. Fauci said. Efforts include the creation of animal models to study disease transmission and its effect on pre- and postnatal outcomes, and of diagnostic platforms that could easily and quickly identify Zika infections. These would quickly differentiate Zika from dengue infections, which are also caused by a strain of flavivirus.
An effective vaccine will not be quickly forthcoming, he cautioned, although a phase I trial is in the works and could begin later this year. The candidate is a DNA-based vaccine similar to the one now used to protect against dengue. It would almost certainly not be approved in less than 2 years, Dr Fauci added.
Until some of these questions have been answered, or until the epidemic slows, the CDC continues to caution pregnant women against travel to endemic areas. For those who do travel or who live in endemic areas, Dr. Schuchat reiterated CDC’s long-held advice to take precautions against mosquito bites: long-sleeved shirts and long pants; staying inside; and using an effective insect repellent. Those containing DEET are most effective and are safe for pregnant women to use, Dr. Schuchat said.
For now, the CDC’s Zika travel alert includes Puerto Rico, Barbados, Bolivia, Brazil, Cape Verde, Colombia, Ecuador, El Salvador, French Guiana, Guadeloupe, Guatemala, Guyana, Haiti, Honduras, Martinique, Mexico, Panama, Paraguay, Saint Martin, Samoa, Suriname, Venezuela, the U.S. Virgin Islands, and the Dominican Republic.
A transcript of the press briefing is available online.
This article was updated 2/1/2016.
On Twitter @Alz_Gal
AACE: New Algorithm Stresses Lifestyle Modification in Type 2 Diabetes
Lifestyle modification must be the cornerstone of any management plan for type 2 diabetes.
Effectively attending to basic problems – obesity, nutrition, and exercise – will dramatically increase the success of any long-term treatment plan for patients with type 2 diabetes (T2DM), and help prevent or delay disease development in those with prediabetes, according to a new treatment algorithm.
The document, published by the American Association of Clinical Endocrinology, is an annual update to its unique strategy of clarifying T2DM management.
The 2016 AACE/ACE Comprehensive Diabetes Management Algorithm presents an easy-to-follow stepwise decision model for diagnosis, blood glucose management, and medical management – including all of the currently approved oral diabetes medications and insulins (Endocr Pract. 2016 Jan;22[1]:84-113). The original algorithm was launched 10 years ago and was last updated in 2013.
This new iteration is the first to place lifestyle intervention as a foundation for the most effective medical management.
“Obviously, this is very important,” said Dr. Paul S. Jellinger, a member of the algorithm writing committee, and an endocrinologist in Fort Lauderdale, Fla. “Weight loss, fitness training, and nutritional management play important roles in the management of blood glucose, lipids, and blood pressure. Appropriate focus in this direction may reduce medication dosage and at times eliminate the need for pharmaceutical intervention. We have all experienced improved therapeutic results in patients who are engaged in effective lifestyle therapy.”
As well as being a text document replete with data-driven details, the algorithm is presented in a colorful poster format that is very helpful for both doctors and patients alike, said Dr. George Grunberger, AACE president. It’s an especially effective tool when considering treatment decisions in the 12 different drug classes used in T2DM management.
“It’s one thing to talk about risk and benefits but to see it graphically displayed is very helpful,” he said in an interview. “I have the poster in every exam room, and every time I am with a patient, I can point out where we are and where we are heading. Rather than me just talking, I can show exactly where we are and where we want to go.”
It is especially helpful for primary care physicians, who care for the vast majority of patients with T2DM, Dr. Grunberger said in an interview.
“We are trying to get better management information into primary care. Most people with diabetes will never see a specialist in their entire life. They need help with glycemic control, obesity, prediabetes, dyslipidemia, and these are the bread and butter of primary care. But there are so many new things going on in this field, and primary care doctors are already in over their heads with the amount of things they deal with. So we have sorted it out and provide practical, practice-oriented guidelines about how to get from A to B.”
Dr. Grunberger noted that most of the recommendations in the document are based on expert opinion. “There are no randomized, controlled trials for most of this stuff. Many of the medications are relatively new, and with 12 drug classes, there’s no way you can ever do a trial with every permutation.”
The new document is similar to the 2013 algorithm with regard to medical management, Dr. Grunberger said. Its focus on lifestyle modification as an integral part of treatment is new, however. “The initial algorithm 10 years ago was solely based on glycemic control. Now we’ve decided to look at more than blood sugar – at obesity, overweight, hypertension and dyslipidemia. You cannot ignore these things in a disease where the major morbidity and mortality are cardiovascular.”
The algorithm stresses that “lifestyle optimization” is essential for all patients with diabetes. “[It] is multifaceted, ongoing, and should engage the entire diabetes team.”
There are several key components to lifestyle modification. All of these should be addressed early.
Medical nutrition therapy
This is a fundamental issue that must be addressed. A primarily plant-based diet high in poly- and monounsaturated fats is recommended, with the goal of a 5%-10% reduction in body weight for overweight or obese patients. In addition to discussing foods that damage and promote metabolic health, patients may need help with carbohydrate and sugar intake. Structured counseling is an excellent way to achieve consistent results.
Physical activity
Regular exercise improves glucose control and lowers lipid and blood pressure levels. It decreases the chance of falls and fractures, promotes functional capacity, and reduces the risk of depression. The goal should be at least 150 minutes of moderate-intense exercise each week. Every patient – and particularly those with complications of diabetes and/or obesity – should have a thorough physical exam before embarking on an exercise program.
Adequate rest
Emerging data continue to confirm the importance of sleep in health and disease. Getting 6-9 hours each night is associated with a reduction in cardiometabolic risk factors. Sleep deprivation aggravates insulin resistance, hypertension, hyperglycemia, and dyslipidemia and increases proinflammatory cytokines. An evaluation for obstructive sleep apnea may be in order, especially for obese patients.
Behavioral support
It’s impossible to overstate the importance of support in a successful lifestyle modification program. Patients should be encouraged to join community groups that facilitate and teach healthy behaviors. Not only will doing so help improve compliance, but being part of a structured group also reaps social and cognitive benefits.
Smoking cessation
The final component of the program, smoking cessation, is critical. All forms of tobacco should be eliminated.
While lifestyle modification is crucial, it should not obviate prompt medical therapy. “Such efforts should not delay needed pharmacotherapy, which can be initiated simultaneously and adjusted based on patient response to lifestyle efforts,” the document notes. “The need for medical therapy should not be interpreted as a failure of lifestyle management but as an adjunct to it.”
Aggressive medical therapy really accelerates effective diabetes treatment, Dr. Jellinger said.
“Clinical inertia has been and remains a huge problem. Some studies demonstrate as much as a 2-year delay in advancing therapy while the patient still remains far from hemoglobin A1c goal. For decades a ‘treat to failure’ concept dominated, i.e., that we should advance therapy only after a prolonged period of failure on existing therapy. One of the major contributions of the earlier AACE algorithms as well as the current version has been the strong therapeutic mandate to re-evaluate the patient and make a therapeutic change in no longer than 3 months. This is a direct attempt to eliminate clinical inertia.”
Dr. Grunberger agreed.
“Why do we wait until people are sick and experiencing complications before we take them seriously? Preventing and dealing with overweight and obesity is complicated, but if you treat obesity, you are treating diabetes. We emphasize starting medical therapy early, going to combination therapy quickly because no one drug usually achieves the target, and trying to be aggressive. Get people on the right treatment as quickly as possible and sustain success – don’t go from one failure to another.”
Dr. Jellinger has received support from Amarin, Boehringer Ingelheim, Bristol-Myers Squibb/AstraZeneca, Janssen Pharmaceuticals, and Novo Nordisk.
Dr. Grunberger has received remuneration and research funding from Eli Lilly, BI-Lilly, Novo Nordisk, Sanofi, Janssen, AstraZeneca, Merck, Medtronic, and GlaxoSmithKline.
Lifestyle modification must be the cornerstone of any management plan for type 2 diabetes.
Effectively attending to basic problems – obesity, nutrition, and exercise – will dramatically increase the success of any long-term treatment plan for patients with type 2 diabetes (T2DM), and help prevent or delay disease development in those with prediabetes, according to a new treatment algorithm.
The document, published by the American Association of Clinical Endocrinology, is an annual update to its unique strategy of clarifying T2DM management.
The 2016 AACE/ACE Comprehensive Diabetes Management Algorithm presents an easy-to-follow stepwise decision model for diagnosis, blood glucose management, and medical management – including all of the currently approved oral diabetes medications and insulins (Endocr Pract. 2016 Jan;22[1]:84-113). The original algorithm was launched 10 years ago and was last updated in 2013.
This new iteration is the first to place lifestyle intervention as a foundation for the most effective medical management.
“Obviously, this is very important,” said Dr. Paul S. Jellinger, a member of the algorithm writing committee, and an endocrinologist in Fort Lauderdale, Fla. “Weight loss, fitness training, and nutritional management play important roles in the management of blood glucose, lipids, and blood pressure. Appropriate focus in this direction may reduce medication dosage and at times eliminate the need for pharmaceutical intervention. We have all experienced improved therapeutic results in patients who are engaged in effective lifestyle therapy.”
As well as being a text document replete with data-driven details, the algorithm is presented in a colorful poster format that is very helpful for both doctors and patients alike, said Dr. George Grunberger, AACE president. It’s an especially effective tool when considering treatment decisions in the 12 different drug classes used in T2DM management.
“It’s one thing to talk about risk and benefits but to see it graphically displayed is very helpful,” he said in an interview. “I have the poster in every exam room, and every time I am with a patient, I can point out where we are and where we are heading. Rather than me just talking, I can show exactly where we are and where we want to go.”
It is especially helpful for primary care physicians, who care for the vast majority of patients with T2DM, Dr. Grunberger said in an interview.
“We are trying to get better management information into primary care. Most people with diabetes will never see a specialist in their entire life. They need help with glycemic control, obesity, prediabetes, dyslipidemia, and these are the bread and butter of primary care. But there are so many new things going on in this field, and primary care doctors are already in over their heads with the amount of things they deal with. So we have sorted it out and provide practical, practice-oriented guidelines about how to get from A to B.”
Dr. Grunberger noted that most of the recommendations in the document are based on expert opinion. “There are no randomized, controlled trials for most of this stuff. Many of the medications are relatively new, and with 12 drug classes, there’s no way you can ever do a trial with every permutation.”
The new document is similar to the 2013 algorithm with regard to medical management, Dr. Grunberger said. Its focus on lifestyle modification as an integral part of treatment is new, however. “The initial algorithm 10 years ago was solely based on glycemic control. Now we’ve decided to look at more than blood sugar – at obesity, overweight, hypertension and dyslipidemia. You cannot ignore these things in a disease where the major morbidity and mortality are cardiovascular.”
The algorithm stresses that “lifestyle optimization” is essential for all patients with diabetes. “[It] is multifaceted, ongoing, and should engage the entire diabetes team.”
There are several key components to lifestyle modification. All of these should be addressed early.
Medical nutrition therapy
This is a fundamental issue that must be addressed. A primarily plant-based diet high in poly- and monounsaturated fats is recommended, with the goal of a 5%-10% reduction in body weight for overweight or obese patients. In addition to discussing foods that damage and promote metabolic health, patients may need help with carbohydrate and sugar intake. Structured counseling is an excellent way to achieve consistent results.
Physical activity
Regular exercise improves glucose control and lowers lipid and blood pressure levels. It decreases the chance of falls and fractures, promotes functional capacity, and reduces the risk of depression. The goal should be at least 150 minutes of moderate-intense exercise each week. Every patient – and particularly those with complications of diabetes and/or obesity – should have a thorough physical exam before embarking on an exercise program.
Adequate rest
Emerging data continue to confirm the importance of sleep in health and disease. Getting 6-9 hours each night is associated with a reduction in cardiometabolic risk factors. Sleep deprivation aggravates insulin resistance, hypertension, hyperglycemia, and dyslipidemia and increases proinflammatory cytokines. An evaluation for obstructive sleep apnea may be in order, especially for obese patients.
Behavioral support
It’s impossible to overstate the importance of support in a successful lifestyle modification program. Patients should be encouraged to join community groups that facilitate and teach healthy behaviors. Not only will doing so help improve compliance, but being part of a structured group also reaps social and cognitive benefits.
Smoking cessation
The final component of the program, smoking cessation, is critical. All forms of tobacco should be eliminated.
While lifestyle modification is crucial, it should not obviate prompt medical therapy. “Such efforts should not delay needed pharmacotherapy, which can be initiated simultaneously and adjusted based on patient response to lifestyle efforts,” the document notes. “The need for medical therapy should not be interpreted as a failure of lifestyle management but as an adjunct to it.”
Aggressive medical therapy really accelerates effective diabetes treatment, Dr. Jellinger said.
“Clinical inertia has been and remains a huge problem. Some studies demonstrate as much as a 2-year delay in advancing therapy while the patient still remains far from hemoglobin A1c goal. For decades a ‘treat to failure’ concept dominated, i.e., that we should advance therapy only after a prolonged period of failure on existing therapy. One of the major contributions of the earlier AACE algorithms as well as the current version has been the strong therapeutic mandate to re-evaluate the patient and make a therapeutic change in no longer than 3 months. This is a direct attempt to eliminate clinical inertia.”
Dr. Grunberger agreed.
“Why do we wait until people are sick and experiencing complications before we take them seriously? Preventing and dealing with overweight and obesity is complicated, but if you treat obesity, you are treating diabetes. We emphasize starting medical therapy early, going to combination therapy quickly because no one drug usually achieves the target, and trying to be aggressive. Get people on the right treatment as quickly as possible and sustain success – don’t go from one failure to another.”
Dr. Jellinger has received support from Amarin, Boehringer Ingelheim, Bristol-Myers Squibb/AstraZeneca, Janssen Pharmaceuticals, and Novo Nordisk.
Dr. Grunberger has received remuneration and research funding from Eli Lilly, BI-Lilly, Novo Nordisk, Sanofi, Janssen, AstraZeneca, Merck, Medtronic, and GlaxoSmithKline.
Lifestyle modification must be the cornerstone of any management plan for type 2 diabetes.
Effectively attending to basic problems – obesity, nutrition, and exercise – will dramatically increase the success of any long-term treatment plan for patients with type 2 diabetes (T2DM), and help prevent or delay disease development in those with prediabetes, according to a new treatment algorithm.
The document, published by the American Association of Clinical Endocrinology, is an annual update to its unique strategy of clarifying T2DM management.
The 2016 AACE/ACE Comprehensive Diabetes Management Algorithm presents an easy-to-follow stepwise decision model for diagnosis, blood glucose management, and medical management – including all of the currently approved oral diabetes medications and insulins (Endocr Pract. 2016 Jan;22[1]:84-113). The original algorithm was launched 10 years ago and was last updated in 2013.
This new iteration is the first to place lifestyle intervention as a foundation for the most effective medical management.
“Obviously, this is very important,” said Dr. Paul S. Jellinger, a member of the algorithm writing committee, and an endocrinologist in Fort Lauderdale, Fla. “Weight loss, fitness training, and nutritional management play important roles in the management of blood glucose, lipids, and blood pressure. Appropriate focus in this direction may reduce medication dosage and at times eliminate the need for pharmaceutical intervention. We have all experienced improved therapeutic results in patients who are engaged in effective lifestyle therapy.”
As well as being a text document replete with data-driven details, the algorithm is presented in a colorful poster format that is very helpful for both doctors and patients alike, said Dr. George Grunberger, AACE president. It’s an especially effective tool when considering treatment decisions in the 12 different drug classes used in T2DM management.
“It’s one thing to talk about risk and benefits but to see it graphically displayed is very helpful,” he said in an interview. “I have the poster in every exam room, and every time I am with a patient, I can point out where we are and where we are heading. Rather than me just talking, I can show exactly where we are and where we want to go.”
It is especially helpful for primary care physicians, who care for the vast majority of patients with T2DM, Dr. Grunberger said in an interview.
“We are trying to get better management information into primary care. Most people with diabetes will never see a specialist in their entire life. They need help with glycemic control, obesity, prediabetes, dyslipidemia, and these are the bread and butter of primary care. But there are so many new things going on in this field, and primary care doctors are already in over their heads with the amount of things they deal with. So we have sorted it out and provide practical, practice-oriented guidelines about how to get from A to B.”
Dr. Grunberger noted that most of the recommendations in the document are based on expert opinion. “There are no randomized, controlled trials for most of this stuff. Many of the medications are relatively new, and with 12 drug classes, there’s no way you can ever do a trial with every permutation.”
The new document is similar to the 2013 algorithm with regard to medical management, Dr. Grunberger said. Its focus on lifestyle modification as an integral part of treatment is new, however. “The initial algorithm 10 years ago was solely based on glycemic control. Now we’ve decided to look at more than blood sugar – at obesity, overweight, hypertension and dyslipidemia. You cannot ignore these things in a disease where the major morbidity and mortality are cardiovascular.”
The algorithm stresses that “lifestyle optimization” is essential for all patients with diabetes. “[It] is multifaceted, ongoing, and should engage the entire diabetes team.”
There are several key components to lifestyle modification. All of these should be addressed early.
Medical nutrition therapy
This is a fundamental issue that must be addressed. A primarily plant-based diet high in poly- and monounsaturated fats is recommended, with the goal of a 5%-10% reduction in body weight for overweight or obese patients. In addition to discussing foods that damage and promote metabolic health, patients may need help with carbohydrate and sugar intake. Structured counseling is an excellent way to achieve consistent results.
Physical activity
Regular exercise improves glucose control and lowers lipid and blood pressure levels. It decreases the chance of falls and fractures, promotes functional capacity, and reduces the risk of depression. The goal should be at least 150 minutes of moderate-intense exercise each week. Every patient – and particularly those with complications of diabetes and/or obesity – should have a thorough physical exam before embarking on an exercise program.
Adequate rest
Emerging data continue to confirm the importance of sleep in health and disease. Getting 6-9 hours each night is associated with a reduction in cardiometabolic risk factors. Sleep deprivation aggravates insulin resistance, hypertension, hyperglycemia, and dyslipidemia and increases proinflammatory cytokines. An evaluation for obstructive sleep apnea may be in order, especially for obese patients.
Behavioral support
It’s impossible to overstate the importance of support in a successful lifestyle modification program. Patients should be encouraged to join community groups that facilitate and teach healthy behaviors. Not only will doing so help improve compliance, but being part of a structured group also reaps social and cognitive benefits.
Smoking cessation
The final component of the program, smoking cessation, is critical. All forms of tobacco should be eliminated.
While lifestyle modification is crucial, it should not obviate prompt medical therapy. “Such efforts should not delay needed pharmacotherapy, which can be initiated simultaneously and adjusted based on patient response to lifestyle efforts,” the document notes. “The need for medical therapy should not be interpreted as a failure of lifestyle management but as an adjunct to it.”
Aggressive medical therapy really accelerates effective diabetes treatment, Dr. Jellinger said.
“Clinical inertia has been and remains a huge problem. Some studies demonstrate as much as a 2-year delay in advancing therapy while the patient still remains far from hemoglobin A1c goal. For decades a ‘treat to failure’ concept dominated, i.e., that we should advance therapy only after a prolonged period of failure on existing therapy. One of the major contributions of the earlier AACE algorithms as well as the current version has been the strong therapeutic mandate to re-evaluate the patient and make a therapeutic change in no longer than 3 months. This is a direct attempt to eliminate clinical inertia.”
Dr. Grunberger agreed.
“Why do we wait until people are sick and experiencing complications before we take them seriously? Preventing and dealing with overweight and obesity is complicated, but if you treat obesity, you are treating diabetes. We emphasize starting medical therapy early, going to combination therapy quickly because no one drug usually achieves the target, and trying to be aggressive. Get people on the right treatment as quickly as possible and sustain success – don’t go from one failure to another.”
Dr. Jellinger has received support from Amarin, Boehringer Ingelheim, Bristol-Myers Squibb/AstraZeneca, Janssen Pharmaceuticals, and Novo Nordisk.
Dr. Grunberger has received remuneration and research funding from Eli Lilly, BI-Lilly, Novo Nordisk, Sanofi, Janssen, AstraZeneca, Merck, Medtronic, and GlaxoSmithKline.