User login
Evidence Suggests Pregnancies Can Survive Maternal Cancer Treatment
Emerging data on pregnancy and cancer can now help women and their doctors chart a safer course between effective treatment and protecting the developing fetus.
Two registries – one in the United States and another in Europe – agree: It’s not only possible to save a pregnancy in many situations, but children born to these women appear to be largely unaffected by in utero chemotherapy exposure. The combined studies followed more than 200 exposed children for up to 18 years; neither one found any elevated risk of congenital anomaly or any kind of cognitive or developmental delay.
"This is practice-changing information," Dr. Frédéric Amant, primary author on the European paper, said in an interview. "Until now, physicians were reluctant to administer chemotherapy and usually opted for termination, or at least for a delay in treatment or premature delivery in order to get treatment going," he said.
Dr. Amant’s study leads off a special section on cancer in pregnancy, published in the March issue of Lancet Oncology. The paper examined evidence that both supports and questions the clinical wisdom of treating a disease that threatens two very different patients – an adult woman and the fetus she carries.
Every case is different, depending on the type of cancer, its grade and potential aggressiveness, the stage of pregnancy, and the woman’s own desires, said Dr. Elyce Cardonick, an ob.gyn. at Cooper University Hospital in Camden, N.J., and the lead investigator of the Pregnancy and Cancer Registry.
"With solid tumors you usually have time to do the surgery, get the pathologic diagnosis, and let the patient recover. But if there is leukemia, for instance, you have to move faster. The first question should be ‘Could we delay treatment for this patient if she was not pregnant?’ I don’t want to limit their treatment, but at the same time, it’s true that they might not be getting the most up-to-date treatment – the newest agent on the block – because you would want to go with something there is at least some experience with. But this doesn’t necessarily mean they are going to do worse."
Cancer occurs in about 1 in 1,000 pregnancies, said Dr. Sarah Temkin, a gynecologic oncologist at the University of Maryland, Baltimore. Many of her patients are referred after a routine screening during early pregnancy finds something abnormal, or when a woman with an existing cancer is incidentally found to be pregnant. But signs that might raise a red flag in other situations don’t necessarily alert physicians to danger in pregnant women, she said in an interview. Pregnancy could obfuscate some symptoms, which might be further downplayed in light of a mother’s relatively young age. Breast cancer is a prime example.
"There are two problems, especially for breast cancers, which are the most common ones we see in pregnancy. First, a woman’s breasts are changing anyway during that time. Breast cancer is so rare in women of earlier childbearing age that both the patient and the doctor tend to disregard any new lumps and bumps."
But despite its rarity, cancer in all forms appears to be increasing among pregnant women, she said. This is probably a direct relation to age. "Cancer rates increase with increasing age, and women are becoming mothers at older and older ages."
When cancer coincides with pregnancy, Dr. Temkin views the mother’s health as paramount. "The mother is the person with cancer, and she deserves whatever the standard of care is for that particular cancer – the best care that would be offered to her if she was not pregnant."
Chemotherapy and Fetal Outcomes
To examine long-term neurodevelopmental risks associated with maternal cancer treatment, Dr. Amant, a gynecologic oncologist at the Leuven (Belgium) Cancer Institute, and his colleagues, are following 70 children from the age of 18 months until 18 years. In the newly published interim analysis, the mean follow-up time is 22 months. All the children had intrauterine exposure to chemotherapy, radiation, oncologic surgery, or combinations of these.
The analysis, which also appeared in Lancet Oncology’s special issue, includes data on all the children, including 18 who are now older than 9 years (Lancet Oncol. 2012;13:256-64).
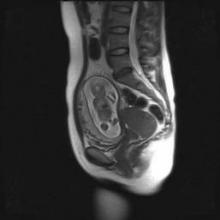
The children – 68 singletons and one set of twins – were born from pregnancies exposed to a total of 236 chemotherapy cycles. Exposure varied by the mother’s cancer type and its stage at diagnosis. In all, 34 mothers were treated with only chemotherapy, 27 had chemotherapy and surgery, 1 received chemotherapy and radiation, and 6 women were treated with all three modalities. Most of the children also had in utero exposure to multiple imaging studies, including MRI, ultrasound, echocardiography, CT, and mammography. Chemotherapy regimens included doxorubicin, epirubicin, idarubicin and daunorubicin.
Fetuses also were exposed to a variety of other drugs, including antibiotics, antiemetics, pain medications, colony stimulating factors, and anxiolytics.
Breast cancer was the most common disease type (35). There were 18 cases of hematologic cancers, 6 ovarian cancers, 4 cervical cancers, and 1 each of basal cell carcinoma, brain tumor, Ewing’s sarcoma, colorectal cancer, and nasopharyngeal cancer.
The mean gestational age at cancer diagnosis was 18 weeks, although fetuses ranged in age from 2 to 33 weeks when their mothers were diagnosed. About a third of the babies (23) were born at term.
Seven were born at 28-32 weeks, nine at 32-34 weeks, and 31 at 34-37 weeks. Weight for gestational age was below the 10th percentile in 14 children (21%).
Seven congenital anomalies were found in the group of 70 children (10%) – a rate not significantly different from that in the background population. There were only two major malformations, for a 2.9% rate. Malformations included the following:
• Hip subluxation, pectus excavatum and hemangioma, associated with chemotherapy only.
• Bilateral partial syndactyly, associated with chemotherapy plus radiotherapy.
• Bilateral small protuberance on one finger, and rectal atresia, associated with chemotherapy plus surgery.
• Bilateral double cartilage ring in a child exposed to chemotherapy, surgery and radiotherapy.
None of the children showed any congenital cardiac issues.
All of the children showed neurocognitive development that was within normal range, except for the set of twins, who were delivered by cesarean section at 32.5 weeks after a preterm premature rupture of membranes. These children were so delayed that they were not able to complete cognitive testing. Their mother developed an acute myeloid leukemia – one of the true "emergency" cancers diagnosed in pregnancy, Dr. Amant said. The babies had been exposed to idarubicin and cytosine arabinoside at 15.5, 21.5, 26.5, and 31.5 weeks’ gestation.
The boy, who weighed 1,640 g at birth, had a normal karyotype but, at 3 years, brain imaging showed a unilateral polymicrogyria in the left perisylvian area. He showed an early developmental delay; at age 9 years, he had the developmental capacity of a 12-month old.
The girl, who weighed 1,390 g at birth, also had an early developmental delay, but at age 9 years she attended school with support. Her parents refused brain imaging.
"The other 68 children did well," Dr. Amant said. "This doesn’t mean they were all normal in every way, but in any population you will see learning and developmental delay issues. We think the problem for the delayed children was not related to chemotherapy exposure, but more likely to their prematurity."
Dr. Amant saw a direct correlation between gestational age and intelligence quota. "When we controlled for age, gender, and country of birth, we found that the IQ score increased by almost 12 points for each additional month of gestation."
The U.S. Experience
Dr. Cardonick has found similar results. Her registry now contains information on 280 women who were enrolled over a 13-year period. The children born from these pregnancies have been assessed annually since birth. It also includes 70 controls – children whose mothers had cancer but who were not exposed to chemotherapy.
She published interim results of the registry in 2010 (Cancer J. 2010;16:76-82). At that point, it contained information on 231 women and 157 children. The most common malignancy was breast cancer (128); the mean gestational age at diagnosis was 13 weeks. About a third of the women (54) were advised to terminate their pregnancy; 12 did so.
Among those who continued both pregnancy and treatment, neonatal outcomes were generally good. There were nine premature deliveries related to preterm labor or premature rupture of membranes. The congenital anomaly rate born was 4%, which was in line with the normal background rate and slightly lower than that seen in Dr. Amant’s cohort.
The infants’ mean birth weight was 2,647 g, which was significantly less than the mean 2,873 g in the control group, but probably clinically irrelevant, Dr. Cardonick wrote in the paper.
She continues to follow these children annually. At this point, the children are a mean 5 years old; her oldest subject is 14 years. So far, the rate of neurocognitive issues in the group is no different than would be observed in any other group, and none of the children has developed any health problem that could be conclusively tied to intrauterine chemotherapy exposure.
Her experience stresses several key factors that must be considered in this situation. Her patients received chemotherapy at a mean of 20 weeks’ gestation – safely outside the critical early period. Only two women received chemotherapy before 10 weeks; both were treated before they knew they were pregnant. Both children born of these pregnancies were considered well. One with intrauterine exposure to cytarabine was developmentally normal at age 7 years. The other, who was exposed to oxaliplatin and capecitabine, was normal at age 2 years.
Treatment also was stopped a mean of 40 days before delivery, to allow the mother’s bone marrow to fully recover before giving birth.
A second U.S. study was reported at the 2011 meeting of the American Society of Clinical Oncology. A poster by Dr. Jennifer Litton and her colleagues examined physiological outcomes in 81 children exposed to chemotherapy for maternal breast cancer. The mothers had taken a standardized chemotherapy regimen of 5-fluorouracil, doxorubicin and cyclophosphamide (FAC) given during the second and third trimesters (J. Clin. Oncol. 2011;29[May 20 suppl.]:abstract 1099).
One child was born with Down syndrome, one with a club foot, and one with ureteral reflux. Three parents reported language delay in later follow-up surveys. Other reported health issues included 15 children with allergies and/or eczema, 2 with asthma, and 1 with absence seizures.
Dr. Litton, a breast oncologist at the University of Texas M.D. Anderson Cancer Center in Houston, also cowrote a 2010 review of breast cancer treatment in pregnancy, in which she discusses maternal and fetal outcomes from several cohorts, and the possible impact of intrauterine exposure to a variety of chemotherapy agents (Oncologist 2010;15:1238-47).
Risks Vary With Cancer Type
Breast cancer during pregnancy may be the simplest to treat. If the cancer is caught very early, it may be reasonable to delay treatment until the fetus has passed the critical first trimester, waiting until organs are formed and the risk of chemically induced damage is reduced, Dr. Temkin said. "It’s safe to do breast surgery during pregnancy and it’s safe to give chemotherapy after the first trimester."
But physicians can miss a new breast tumor during a prenatal exam, so some present at a more advanced stage, according to Dr. Amant, who is also the lead author of the Lancet’s breast cancer report (Lancet 2012;379:570-9).
Infiltrating ductal adenocarcinomas account for more than 70% of the breast cancers diagnosed during pregnancy. These can be aggressive, said Dr. Amant. Estrogen receptor status is probably no different in pregnant and nonpregnant women.
If the tumor is discovered early and is pathologically favorable, chemotherapy probably can be delayed until 14 weeks’ gestation, allowing nearly complete fetal organogenesis without worsening the mother’s outcome. Women also may elect an early termination if the pathology is unfavorable, or for other personal reasons, Dr. Temkin said. "I think a lot of it depends on when the cancer is diagnosed. Patients of mine who already have a diagnosis and then become pregnant almost always elect to terminate. But if the cancer is discovered when the pregnancy is farther along, most will continue, especially if the woman is highly emotionally invested," she noted.
Tougher Cancers, Tougher Choices
Treating gynecologic cancers during pregnancy often comes down to a choice between the mother’s health and maintenance of the pregnancy, Dr. Temkin said. "The standard of care for ovarian cancers is surgery or radiation to the pelvis, where the fetus is. Cervical cancer is treated with a hysterectomy or radiation, and neither treatment is compatible with keeping a pregnancy. Neoadjuvant therapy is not considered standard of care for these tumors. These are complex decisions for the patient: ‘Do I accept a different treatment [that might not be as effective] or maintain the pregnancy?’ "
In early cervical cancers without nodal spread, the most common tactic is close observation with periodic imaging to rule out spread; therapy is given after delivery, Dr. Phillippe Morice wrote in the Lancet section’s review on gynecologic malignancies (Lancet 2012;379:558-69).
"Delayed treatment until fetal maturation for patients with stage IA disease has an excellent prognosis and is now the standard of care," wrote Dr. Morice of the Institut de Cancérologie Gustave-Roussy in Villejuif, France.
Locally advanced disease is often not compatible with pregnancy. "The main treatment choice is either neoadjuvant chemotherapy or chemotherapy and radiotherapy. In pregnant patients, this approach means that the pregnancy must be ended before the initiation of therapy, but in exceptional cases in which surgery to end the pregnancy is not technically feasible ([that is], a bulky cervical tumor), radiation therapy can be delivered with the fetus in utero, resulting in a spontaneous abortion in about 3 weeks," he wrote.
Ovarian tumors can be surgically staged and – if it is of low malignant potential – can be laparoscopically removed, usually without endangering the pregnancy. Large tumors or those with aggressive pathology, like epithelial tumors, are much more difficult. Advanced or large tumors often have uterine and pelvic involvement, and treatment usually means a hysterectomy.
The literature contains reports of a very few women who have undergone chemotherapy to control peritoneal spread while keeping a pregnancy. However, despite giving birth to normally developed children, a number of these women died from recurrent disease, Dr. Morice noted.
Hematologic Cancers: True Emergency
Cancers of the blood are rare in pregnancy, occurring in only 1 of every 6,000. But when they do occur, they can be devastating, Dr. Benjamin Brenner wrote in the special series (Lancet 2012;379:580-7).
Pregnant patients with Hodgkin’s lymphoma generally do as well as their nonpregnant counterparts and can receive the same chemotherapy regimens, observing the first-trimester delay to favor the fetus.
Those who present with non-Hodgkin’s lymphoma are likely to have a very poor outlook. This disease is very rare in pregnant women, and symptoms can overlap with Hodgkin’s. Those factors, combined with a desire to avoid imaging, can delay diagnosis until the cancer is more advanced, said Dr. Brenner of the Rambam Health Care Campus, Haifa, Israel.
Acute leukemia is also rare, but demands urgent attention regardless of gestational stage, Dr. Brenner warned. "Patients diagnosed with acute leukemia during the first trimester are recommended to terminate the pregnancy, in view of the high risk of toxic effects on the fetus and mother, along with the expected need for further intensive treatment including stem-cell transplantation, which is absolutely contraindicated during gestation."
Talking It Out
Despite the emerging positive evidence, treating cancer during pregnancy can be a tough sell, Dr. Amant said. "Women have been told over and over to avoid taking so much as an aspirin. It’s very difficult to convince them that a fetus can not only survive a mother’s cancer treatment, but have a good chance of developing normally."
The stress of a cancer diagnosis during a desired pregnancy is very hard on patients, Dr. Temkin added. "Pregnancy is a time when many women come to grips with their own mortality as well as that of giving new life. Adding a diagnosis of cancer of top of that – especially in the face of a much-desired pregnancy – can be devastating."
These women are faced with two options: terminate the pregnancy and concentrate on their own treatment, or continue the pregnancy knowing that their unborn child will be exposed to the possible risks of radiation, chemotherapy, and surgery. Either option can "inflict terrible guilt on a pregnant woman. We can try to minimize that to some degree, but it’s important to know from the outset that what is the right solution for one patient is not right for the next."
Connecting with other women who have experienced the same situation can be of immense value, Dr. Cardonick said. She participates in an online support group called "Hope for Two."
The organization’s main goal is to link new patients with survivors who can help educate them as well as lend emotional support. Patients call in or fill out a secure online request for a personal match-up with a survivor, who is often a woman who has had the same type of cancer.
The website also contains links to news and medical articles, books, and financial assistance sources, and allows new patients to securely contact Dr. Cardonick’s pregnancy registry. "We keep in touch with the baby’s pediatrician and the mom every year, to see how things are going and [to] collect information," she said. "The best way to treat these women in the future depends on the information we continue to gather in the present."
None of the researchers interviewed for this article had any relevant financial disclosures. Dr. Litton noted that she had no financial disclosures for her 2011 ASCO poster.
Emerging data on pregnancy and cancer can now help women and their doctors chart a safer course between effective treatment and protecting the developing fetus.
Two registries – one in the United States and another in Europe – agree: It’s not only possible to save a pregnancy in many situations, but children born to these women appear to be largely unaffected by in utero chemotherapy exposure. The combined studies followed more than 200 exposed children for up to 18 years; neither one found any elevated risk of congenital anomaly or any kind of cognitive or developmental delay.
"This is practice-changing information," Dr. Frédéric Amant, primary author on the European paper, said in an interview. "Until now, physicians were reluctant to administer chemotherapy and usually opted for termination, or at least for a delay in treatment or premature delivery in order to get treatment going," he said.
Dr. Amant’s study leads off a special section on cancer in pregnancy, published in the March issue of Lancet Oncology. The paper examined evidence that both supports and questions the clinical wisdom of treating a disease that threatens two very different patients – an adult woman and the fetus she carries.
Every case is different, depending on the type of cancer, its grade and potential aggressiveness, the stage of pregnancy, and the woman’s own desires, said Dr. Elyce Cardonick, an ob.gyn. at Cooper University Hospital in Camden, N.J., and the lead investigator of the Pregnancy and Cancer Registry.
"With solid tumors you usually have time to do the surgery, get the pathologic diagnosis, and let the patient recover. But if there is leukemia, for instance, you have to move faster. The first question should be ‘Could we delay treatment for this patient if she was not pregnant?’ I don’t want to limit their treatment, but at the same time, it’s true that they might not be getting the most up-to-date treatment – the newest agent on the block – because you would want to go with something there is at least some experience with. But this doesn’t necessarily mean they are going to do worse."
Cancer occurs in about 1 in 1,000 pregnancies, said Dr. Sarah Temkin, a gynecologic oncologist at the University of Maryland, Baltimore. Many of her patients are referred after a routine screening during early pregnancy finds something abnormal, or when a woman with an existing cancer is incidentally found to be pregnant. But signs that might raise a red flag in other situations don’t necessarily alert physicians to danger in pregnant women, she said in an interview. Pregnancy could obfuscate some symptoms, which might be further downplayed in light of a mother’s relatively young age. Breast cancer is a prime example.
"There are two problems, especially for breast cancers, which are the most common ones we see in pregnancy. First, a woman’s breasts are changing anyway during that time. Breast cancer is so rare in women of earlier childbearing age that both the patient and the doctor tend to disregard any new lumps and bumps."
But despite its rarity, cancer in all forms appears to be increasing among pregnant women, she said. This is probably a direct relation to age. "Cancer rates increase with increasing age, and women are becoming mothers at older and older ages."
When cancer coincides with pregnancy, Dr. Temkin views the mother’s health as paramount. "The mother is the person with cancer, and she deserves whatever the standard of care is for that particular cancer – the best care that would be offered to her if she was not pregnant."
Chemotherapy and Fetal Outcomes
To examine long-term neurodevelopmental risks associated with maternal cancer treatment, Dr. Amant, a gynecologic oncologist at the Leuven (Belgium) Cancer Institute, and his colleagues, are following 70 children from the age of 18 months until 18 years. In the newly published interim analysis, the mean follow-up time is 22 months. All the children had intrauterine exposure to chemotherapy, radiation, oncologic surgery, or combinations of these.
The analysis, which also appeared in Lancet Oncology’s special issue, includes data on all the children, including 18 who are now older than 9 years (Lancet Oncol. 2012;13:256-64).
The children – 68 singletons and one set of twins – were born from pregnancies exposed to a total of 236 chemotherapy cycles. Exposure varied by the mother’s cancer type and its stage at diagnosis. In all, 34 mothers were treated with only chemotherapy, 27 had chemotherapy and surgery, 1 received chemotherapy and radiation, and 6 women were treated with all three modalities. Most of the children also had in utero exposure to multiple imaging studies, including MRI, ultrasound, echocardiography, CT, and mammography. Chemotherapy regimens included doxorubicin, epirubicin, idarubicin and daunorubicin.
Fetuses also were exposed to a variety of other drugs, including antibiotics, antiemetics, pain medications, colony stimulating factors, and anxiolytics.
Breast cancer was the most common disease type (35). There were 18 cases of hematologic cancers, 6 ovarian cancers, 4 cervical cancers, and 1 each of basal cell carcinoma, brain tumor, Ewing’s sarcoma, colorectal cancer, and nasopharyngeal cancer.
The mean gestational age at cancer diagnosis was 18 weeks, although fetuses ranged in age from 2 to 33 weeks when their mothers were diagnosed. About a third of the babies (23) were born at term.
Seven were born at 28-32 weeks, nine at 32-34 weeks, and 31 at 34-37 weeks. Weight for gestational age was below the 10th percentile in 14 children (21%).
Seven congenital anomalies were found in the group of 70 children (10%) – a rate not significantly different from that in the background population. There were only two major malformations, for a 2.9% rate. Malformations included the following:
• Hip subluxation, pectus excavatum and hemangioma, associated with chemotherapy only.
• Bilateral partial syndactyly, associated with chemotherapy plus radiotherapy.
• Bilateral small protuberance on one finger, and rectal atresia, associated with chemotherapy plus surgery.
• Bilateral double cartilage ring in a child exposed to chemotherapy, surgery and radiotherapy.
None of the children showed any congenital cardiac issues.
All of the children showed neurocognitive development that was within normal range, except for the set of twins, who were delivered by cesarean section at 32.5 weeks after a preterm premature rupture of membranes. These children were so delayed that they were not able to complete cognitive testing. Their mother developed an acute myeloid leukemia – one of the true "emergency" cancers diagnosed in pregnancy, Dr. Amant said. The babies had been exposed to idarubicin and cytosine arabinoside at 15.5, 21.5, 26.5, and 31.5 weeks’ gestation.
The boy, who weighed 1,640 g at birth, had a normal karyotype but, at 3 years, brain imaging showed a unilateral polymicrogyria in the left perisylvian area. He showed an early developmental delay; at age 9 years, he had the developmental capacity of a 12-month old.
The girl, who weighed 1,390 g at birth, also had an early developmental delay, but at age 9 years she attended school with support. Her parents refused brain imaging.
"The other 68 children did well," Dr. Amant said. "This doesn’t mean they were all normal in every way, but in any population you will see learning and developmental delay issues. We think the problem for the delayed children was not related to chemotherapy exposure, but more likely to their prematurity."
Dr. Amant saw a direct correlation between gestational age and intelligence quota. "When we controlled for age, gender, and country of birth, we found that the IQ score increased by almost 12 points for each additional month of gestation."
The U.S. Experience
Dr. Cardonick has found similar results. Her registry now contains information on 280 women who were enrolled over a 13-year period. The children born from these pregnancies have been assessed annually since birth. It also includes 70 controls – children whose mothers had cancer but who were not exposed to chemotherapy.
She published interim results of the registry in 2010 (Cancer J. 2010;16:76-82). At that point, it contained information on 231 women and 157 children. The most common malignancy was breast cancer (128); the mean gestational age at diagnosis was 13 weeks. About a third of the women (54) were advised to terminate their pregnancy; 12 did so.
Among those who continued both pregnancy and treatment, neonatal outcomes were generally good. There were nine premature deliveries related to preterm labor or premature rupture of membranes. The congenital anomaly rate born was 4%, which was in line with the normal background rate and slightly lower than that seen in Dr. Amant’s cohort.
The infants’ mean birth weight was 2,647 g, which was significantly less than the mean 2,873 g in the control group, but probably clinically irrelevant, Dr. Cardonick wrote in the paper.
She continues to follow these children annually. At this point, the children are a mean 5 years old; her oldest subject is 14 years. So far, the rate of neurocognitive issues in the group is no different than would be observed in any other group, and none of the children has developed any health problem that could be conclusively tied to intrauterine chemotherapy exposure.
Her experience stresses several key factors that must be considered in this situation. Her patients received chemotherapy at a mean of 20 weeks’ gestation – safely outside the critical early period. Only two women received chemotherapy before 10 weeks; both were treated before they knew they were pregnant. Both children born of these pregnancies were considered well. One with intrauterine exposure to cytarabine was developmentally normal at age 7 years. The other, who was exposed to oxaliplatin and capecitabine, was normal at age 2 years.
Treatment also was stopped a mean of 40 days before delivery, to allow the mother’s bone marrow to fully recover before giving birth.
A second U.S. study was reported at the 2011 meeting of the American Society of Clinical Oncology. A poster by Dr. Jennifer Litton and her colleagues examined physiological outcomes in 81 children exposed to chemotherapy for maternal breast cancer. The mothers had taken a standardized chemotherapy regimen of 5-fluorouracil, doxorubicin and cyclophosphamide (FAC) given during the second and third trimesters (J. Clin. Oncol. 2011;29[May 20 suppl.]:abstract 1099).
One child was born with Down syndrome, one with a club foot, and one with ureteral reflux. Three parents reported language delay in later follow-up surveys. Other reported health issues included 15 children with allergies and/or eczema, 2 with asthma, and 1 with absence seizures.
Dr. Litton, a breast oncologist at the University of Texas M.D. Anderson Cancer Center in Houston, also cowrote a 2010 review of breast cancer treatment in pregnancy, in which she discusses maternal and fetal outcomes from several cohorts, and the possible impact of intrauterine exposure to a variety of chemotherapy agents (Oncologist 2010;15:1238-47).
Risks Vary With Cancer Type
Breast cancer during pregnancy may be the simplest to treat. If the cancer is caught very early, it may be reasonable to delay treatment until the fetus has passed the critical first trimester, waiting until organs are formed and the risk of chemically induced damage is reduced, Dr. Temkin said. "It’s safe to do breast surgery during pregnancy and it’s safe to give chemotherapy after the first trimester."
But physicians can miss a new breast tumor during a prenatal exam, so some present at a more advanced stage, according to Dr. Amant, who is also the lead author of the Lancet’s breast cancer report (Lancet 2012;379:570-9).
Infiltrating ductal adenocarcinomas account for more than 70% of the breast cancers diagnosed during pregnancy. These can be aggressive, said Dr. Amant. Estrogen receptor status is probably no different in pregnant and nonpregnant women.
If the tumor is discovered early and is pathologically favorable, chemotherapy probably can be delayed until 14 weeks’ gestation, allowing nearly complete fetal organogenesis without worsening the mother’s outcome. Women also may elect an early termination if the pathology is unfavorable, or for other personal reasons, Dr. Temkin said. "I think a lot of it depends on when the cancer is diagnosed. Patients of mine who already have a diagnosis and then become pregnant almost always elect to terminate. But if the cancer is discovered when the pregnancy is farther along, most will continue, especially if the woman is highly emotionally invested," she noted.
Tougher Cancers, Tougher Choices
Treating gynecologic cancers during pregnancy often comes down to a choice between the mother’s health and maintenance of the pregnancy, Dr. Temkin said. "The standard of care for ovarian cancers is surgery or radiation to the pelvis, where the fetus is. Cervical cancer is treated with a hysterectomy or radiation, and neither treatment is compatible with keeping a pregnancy. Neoadjuvant therapy is not considered standard of care for these tumors. These are complex decisions for the patient: ‘Do I accept a different treatment [that might not be as effective] or maintain the pregnancy?’ "
In early cervical cancers without nodal spread, the most common tactic is close observation with periodic imaging to rule out spread; therapy is given after delivery, Dr. Phillippe Morice wrote in the Lancet section’s review on gynecologic malignancies (Lancet 2012;379:558-69).
"Delayed treatment until fetal maturation for patients with stage IA disease has an excellent prognosis and is now the standard of care," wrote Dr. Morice of the Institut de Cancérologie Gustave-Roussy in Villejuif, France.
Locally advanced disease is often not compatible with pregnancy. "The main treatment choice is either neoadjuvant chemotherapy or chemotherapy and radiotherapy. In pregnant patients, this approach means that the pregnancy must be ended before the initiation of therapy, but in exceptional cases in which surgery to end the pregnancy is not technically feasible ([that is], a bulky cervical tumor), radiation therapy can be delivered with the fetus in utero, resulting in a spontaneous abortion in about 3 weeks," he wrote.
Ovarian tumors can be surgically staged and – if it is of low malignant potential – can be laparoscopically removed, usually without endangering the pregnancy. Large tumors or those with aggressive pathology, like epithelial tumors, are much more difficult. Advanced or large tumors often have uterine and pelvic involvement, and treatment usually means a hysterectomy.
The literature contains reports of a very few women who have undergone chemotherapy to control peritoneal spread while keeping a pregnancy. However, despite giving birth to normally developed children, a number of these women died from recurrent disease, Dr. Morice noted.
Hematologic Cancers: True Emergency
Cancers of the blood are rare in pregnancy, occurring in only 1 of every 6,000. But when they do occur, they can be devastating, Dr. Benjamin Brenner wrote in the special series (Lancet 2012;379:580-7).
Pregnant patients with Hodgkin’s lymphoma generally do as well as their nonpregnant counterparts and can receive the same chemotherapy regimens, observing the first-trimester delay to favor the fetus.
Those who present with non-Hodgkin’s lymphoma are likely to have a very poor outlook. This disease is very rare in pregnant women, and symptoms can overlap with Hodgkin’s. Those factors, combined with a desire to avoid imaging, can delay diagnosis until the cancer is more advanced, said Dr. Brenner of the Rambam Health Care Campus, Haifa, Israel.
Acute leukemia is also rare, but demands urgent attention regardless of gestational stage, Dr. Brenner warned. "Patients diagnosed with acute leukemia during the first trimester are recommended to terminate the pregnancy, in view of the high risk of toxic effects on the fetus and mother, along with the expected need for further intensive treatment including stem-cell transplantation, which is absolutely contraindicated during gestation."
Talking It Out
Despite the emerging positive evidence, treating cancer during pregnancy can be a tough sell, Dr. Amant said. "Women have been told over and over to avoid taking so much as an aspirin. It’s very difficult to convince them that a fetus can not only survive a mother’s cancer treatment, but have a good chance of developing normally."
The stress of a cancer diagnosis during a desired pregnancy is very hard on patients, Dr. Temkin added. "Pregnancy is a time when many women come to grips with their own mortality as well as that of giving new life. Adding a diagnosis of cancer of top of that – especially in the face of a much-desired pregnancy – can be devastating."
These women are faced with two options: terminate the pregnancy and concentrate on their own treatment, or continue the pregnancy knowing that their unborn child will be exposed to the possible risks of radiation, chemotherapy, and surgery. Either option can "inflict terrible guilt on a pregnant woman. We can try to minimize that to some degree, but it’s important to know from the outset that what is the right solution for one patient is not right for the next."
Connecting with other women who have experienced the same situation can be of immense value, Dr. Cardonick said. She participates in an online support group called "Hope for Two."
The organization’s main goal is to link new patients with survivors who can help educate them as well as lend emotional support. Patients call in or fill out a secure online request for a personal match-up with a survivor, who is often a woman who has had the same type of cancer.
The website also contains links to news and medical articles, books, and financial assistance sources, and allows new patients to securely contact Dr. Cardonick’s pregnancy registry. "We keep in touch with the baby’s pediatrician and the mom every year, to see how things are going and [to] collect information," she said. "The best way to treat these women in the future depends on the information we continue to gather in the present."
None of the researchers interviewed for this article had any relevant financial disclosures. Dr. Litton noted that she had no financial disclosures for her 2011 ASCO poster.
Emerging data on pregnancy and cancer can now help women and their doctors chart a safer course between effective treatment and protecting the developing fetus.
Two registries – one in the United States and another in Europe – agree: It’s not only possible to save a pregnancy in many situations, but children born to these women appear to be largely unaffected by in utero chemotherapy exposure. The combined studies followed more than 200 exposed children for up to 18 years; neither one found any elevated risk of congenital anomaly or any kind of cognitive or developmental delay.
"This is practice-changing information," Dr. Frédéric Amant, primary author on the European paper, said in an interview. "Until now, physicians were reluctant to administer chemotherapy and usually opted for termination, or at least for a delay in treatment or premature delivery in order to get treatment going," he said.
Dr. Amant’s study leads off a special section on cancer in pregnancy, published in the March issue of Lancet Oncology. The paper examined evidence that both supports and questions the clinical wisdom of treating a disease that threatens two very different patients – an adult woman and the fetus she carries.
Every case is different, depending on the type of cancer, its grade and potential aggressiveness, the stage of pregnancy, and the woman’s own desires, said Dr. Elyce Cardonick, an ob.gyn. at Cooper University Hospital in Camden, N.J., and the lead investigator of the Pregnancy and Cancer Registry.
"With solid tumors you usually have time to do the surgery, get the pathologic diagnosis, and let the patient recover. But if there is leukemia, for instance, you have to move faster. The first question should be ‘Could we delay treatment for this patient if she was not pregnant?’ I don’t want to limit their treatment, but at the same time, it’s true that they might not be getting the most up-to-date treatment – the newest agent on the block – because you would want to go with something there is at least some experience with. But this doesn’t necessarily mean they are going to do worse."
Cancer occurs in about 1 in 1,000 pregnancies, said Dr. Sarah Temkin, a gynecologic oncologist at the University of Maryland, Baltimore. Many of her patients are referred after a routine screening during early pregnancy finds something abnormal, or when a woman with an existing cancer is incidentally found to be pregnant. But signs that might raise a red flag in other situations don’t necessarily alert physicians to danger in pregnant women, she said in an interview. Pregnancy could obfuscate some symptoms, which might be further downplayed in light of a mother’s relatively young age. Breast cancer is a prime example.
"There are two problems, especially for breast cancers, which are the most common ones we see in pregnancy. First, a woman’s breasts are changing anyway during that time. Breast cancer is so rare in women of earlier childbearing age that both the patient and the doctor tend to disregard any new lumps and bumps."
But despite its rarity, cancer in all forms appears to be increasing among pregnant women, she said. This is probably a direct relation to age. "Cancer rates increase with increasing age, and women are becoming mothers at older and older ages."
When cancer coincides with pregnancy, Dr. Temkin views the mother’s health as paramount. "The mother is the person with cancer, and she deserves whatever the standard of care is for that particular cancer – the best care that would be offered to her if she was not pregnant."
Chemotherapy and Fetal Outcomes
To examine long-term neurodevelopmental risks associated with maternal cancer treatment, Dr. Amant, a gynecologic oncologist at the Leuven (Belgium) Cancer Institute, and his colleagues, are following 70 children from the age of 18 months until 18 years. In the newly published interim analysis, the mean follow-up time is 22 months. All the children had intrauterine exposure to chemotherapy, radiation, oncologic surgery, or combinations of these.
The analysis, which also appeared in Lancet Oncology’s special issue, includes data on all the children, including 18 who are now older than 9 years (Lancet Oncol. 2012;13:256-64).
The children – 68 singletons and one set of twins – were born from pregnancies exposed to a total of 236 chemotherapy cycles. Exposure varied by the mother’s cancer type and its stage at diagnosis. In all, 34 mothers were treated with only chemotherapy, 27 had chemotherapy and surgery, 1 received chemotherapy and radiation, and 6 women were treated with all three modalities. Most of the children also had in utero exposure to multiple imaging studies, including MRI, ultrasound, echocardiography, CT, and mammography. Chemotherapy regimens included doxorubicin, epirubicin, idarubicin and daunorubicin.
Fetuses also were exposed to a variety of other drugs, including antibiotics, antiemetics, pain medications, colony stimulating factors, and anxiolytics.
Breast cancer was the most common disease type (35). There were 18 cases of hematologic cancers, 6 ovarian cancers, 4 cervical cancers, and 1 each of basal cell carcinoma, brain tumor, Ewing’s sarcoma, colorectal cancer, and nasopharyngeal cancer.
The mean gestational age at cancer diagnosis was 18 weeks, although fetuses ranged in age from 2 to 33 weeks when their mothers were diagnosed. About a third of the babies (23) were born at term.
Seven were born at 28-32 weeks, nine at 32-34 weeks, and 31 at 34-37 weeks. Weight for gestational age was below the 10th percentile in 14 children (21%).
Seven congenital anomalies were found in the group of 70 children (10%) – a rate not significantly different from that in the background population. There were only two major malformations, for a 2.9% rate. Malformations included the following:
• Hip subluxation, pectus excavatum and hemangioma, associated with chemotherapy only.
• Bilateral partial syndactyly, associated with chemotherapy plus radiotherapy.
• Bilateral small protuberance on one finger, and rectal atresia, associated with chemotherapy plus surgery.
• Bilateral double cartilage ring in a child exposed to chemotherapy, surgery and radiotherapy.
None of the children showed any congenital cardiac issues.
All of the children showed neurocognitive development that was within normal range, except for the set of twins, who were delivered by cesarean section at 32.5 weeks after a preterm premature rupture of membranes. These children were so delayed that they were not able to complete cognitive testing. Their mother developed an acute myeloid leukemia – one of the true "emergency" cancers diagnosed in pregnancy, Dr. Amant said. The babies had been exposed to idarubicin and cytosine arabinoside at 15.5, 21.5, 26.5, and 31.5 weeks’ gestation.
The boy, who weighed 1,640 g at birth, had a normal karyotype but, at 3 years, brain imaging showed a unilateral polymicrogyria in the left perisylvian area. He showed an early developmental delay; at age 9 years, he had the developmental capacity of a 12-month old.
The girl, who weighed 1,390 g at birth, also had an early developmental delay, but at age 9 years she attended school with support. Her parents refused brain imaging.
"The other 68 children did well," Dr. Amant said. "This doesn’t mean they were all normal in every way, but in any population you will see learning and developmental delay issues. We think the problem for the delayed children was not related to chemotherapy exposure, but more likely to their prematurity."
Dr. Amant saw a direct correlation between gestational age and intelligence quota. "When we controlled for age, gender, and country of birth, we found that the IQ score increased by almost 12 points for each additional month of gestation."
The U.S. Experience
Dr. Cardonick has found similar results. Her registry now contains information on 280 women who were enrolled over a 13-year period. The children born from these pregnancies have been assessed annually since birth. It also includes 70 controls – children whose mothers had cancer but who were not exposed to chemotherapy.
She published interim results of the registry in 2010 (Cancer J. 2010;16:76-82). At that point, it contained information on 231 women and 157 children. The most common malignancy was breast cancer (128); the mean gestational age at diagnosis was 13 weeks. About a third of the women (54) were advised to terminate their pregnancy; 12 did so.
Among those who continued both pregnancy and treatment, neonatal outcomes were generally good. There were nine premature deliveries related to preterm labor or premature rupture of membranes. The congenital anomaly rate born was 4%, which was in line with the normal background rate and slightly lower than that seen in Dr. Amant’s cohort.
The infants’ mean birth weight was 2,647 g, which was significantly less than the mean 2,873 g in the control group, but probably clinically irrelevant, Dr. Cardonick wrote in the paper.
She continues to follow these children annually. At this point, the children are a mean 5 years old; her oldest subject is 14 years. So far, the rate of neurocognitive issues in the group is no different than would be observed in any other group, and none of the children has developed any health problem that could be conclusively tied to intrauterine chemotherapy exposure.
Her experience stresses several key factors that must be considered in this situation. Her patients received chemotherapy at a mean of 20 weeks’ gestation – safely outside the critical early period. Only two women received chemotherapy before 10 weeks; both were treated before they knew they were pregnant. Both children born of these pregnancies were considered well. One with intrauterine exposure to cytarabine was developmentally normal at age 7 years. The other, who was exposed to oxaliplatin and capecitabine, was normal at age 2 years.
Treatment also was stopped a mean of 40 days before delivery, to allow the mother’s bone marrow to fully recover before giving birth.
A second U.S. study was reported at the 2011 meeting of the American Society of Clinical Oncology. A poster by Dr. Jennifer Litton and her colleagues examined physiological outcomes in 81 children exposed to chemotherapy for maternal breast cancer. The mothers had taken a standardized chemotherapy regimen of 5-fluorouracil, doxorubicin and cyclophosphamide (FAC) given during the second and third trimesters (J. Clin. Oncol. 2011;29[May 20 suppl.]:abstract 1099).
One child was born with Down syndrome, one with a club foot, and one with ureteral reflux. Three parents reported language delay in later follow-up surveys. Other reported health issues included 15 children with allergies and/or eczema, 2 with asthma, and 1 with absence seizures.
Dr. Litton, a breast oncologist at the University of Texas M.D. Anderson Cancer Center in Houston, also cowrote a 2010 review of breast cancer treatment in pregnancy, in which she discusses maternal and fetal outcomes from several cohorts, and the possible impact of intrauterine exposure to a variety of chemotherapy agents (Oncologist 2010;15:1238-47).
Risks Vary With Cancer Type
Breast cancer during pregnancy may be the simplest to treat. If the cancer is caught very early, it may be reasonable to delay treatment until the fetus has passed the critical first trimester, waiting until organs are formed and the risk of chemically induced damage is reduced, Dr. Temkin said. "It’s safe to do breast surgery during pregnancy and it’s safe to give chemotherapy after the first trimester."
But physicians can miss a new breast tumor during a prenatal exam, so some present at a more advanced stage, according to Dr. Amant, who is also the lead author of the Lancet’s breast cancer report (Lancet 2012;379:570-9).
Infiltrating ductal adenocarcinomas account for more than 70% of the breast cancers diagnosed during pregnancy. These can be aggressive, said Dr. Amant. Estrogen receptor status is probably no different in pregnant and nonpregnant women.
If the tumor is discovered early and is pathologically favorable, chemotherapy probably can be delayed until 14 weeks’ gestation, allowing nearly complete fetal organogenesis without worsening the mother’s outcome. Women also may elect an early termination if the pathology is unfavorable, or for other personal reasons, Dr. Temkin said. "I think a lot of it depends on when the cancer is diagnosed. Patients of mine who already have a diagnosis and then become pregnant almost always elect to terminate. But if the cancer is discovered when the pregnancy is farther along, most will continue, especially if the woman is highly emotionally invested," she noted.
Tougher Cancers, Tougher Choices
Treating gynecologic cancers during pregnancy often comes down to a choice between the mother’s health and maintenance of the pregnancy, Dr. Temkin said. "The standard of care for ovarian cancers is surgery or radiation to the pelvis, where the fetus is. Cervical cancer is treated with a hysterectomy or radiation, and neither treatment is compatible with keeping a pregnancy. Neoadjuvant therapy is not considered standard of care for these tumors. These are complex decisions for the patient: ‘Do I accept a different treatment [that might not be as effective] or maintain the pregnancy?’ "
In early cervical cancers without nodal spread, the most common tactic is close observation with periodic imaging to rule out spread; therapy is given after delivery, Dr. Phillippe Morice wrote in the Lancet section’s review on gynecologic malignancies (Lancet 2012;379:558-69).
"Delayed treatment until fetal maturation for patients with stage IA disease has an excellent prognosis and is now the standard of care," wrote Dr. Morice of the Institut de Cancérologie Gustave-Roussy in Villejuif, France.
Locally advanced disease is often not compatible with pregnancy. "The main treatment choice is either neoadjuvant chemotherapy or chemotherapy and radiotherapy. In pregnant patients, this approach means that the pregnancy must be ended before the initiation of therapy, but in exceptional cases in which surgery to end the pregnancy is not technically feasible ([that is], a bulky cervical tumor), radiation therapy can be delivered with the fetus in utero, resulting in a spontaneous abortion in about 3 weeks," he wrote.
Ovarian tumors can be surgically staged and – if it is of low malignant potential – can be laparoscopically removed, usually without endangering the pregnancy. Large tumors or those with aggressive pathology, like epithelial tumors, are much more difficult. Advanced or large tumors often have uterine and pelvic involvement, and treatment usually means a hysterectomy.
The literature contains reports of a very few women who have undergone chemotherapy to control peritoneal spread while keeping a pregnancy. However, despite giving birth to normally developed children, a number of these women died from recurrent disease, Dr. Morice noted.
Hematologic Cancers: True Emergency
Cancers of the blood are rare in pregnancy, occurring in only 1 of every 6,000. But when they do occur, they can be devastating, Dr. Benjamin Brenner wrote in the special series (Lancet 2012;379:580-7).
Pregnant patients with Hodgkin’s lymphoma generally do as well as their nonpregnant counterparts and can receive the same chemotherapy regimens, observing the first-trimester delay to favor the fetus.
Those who present with non-Hodgkin’s lymphoma are likely to have a very poor outlook. This disease is very rare in pregnant women, and symptoms can overlap with Hodgkin’s. Those factors, combined with a desire to avoid imaging, can delay diagnosis until the cancer is more advanced, said Dr. Brenner of the Rambam Health Care Campus, Haifa, Israel.
Acute leukemia is also rare, but demands urgent attention regardless of gestational stage, Dr. Brenner warned. "Patients diagnosed with acute leukemia during the first trimester are recommended to terminate the pregnancy, in view of the high risk of toxic effects on the fetus and mother, along with the expected need for further intensive treatment including stem-cell transplantation, which is absolutely contraindicated during gestation."
Talking It Out
Despite the emerging positive evidence, treating cancer during pregnancy can be a tough sell, Dr. Amant said. "Women have been told over and over to avoid taking so much as an aspirin. It’s very difficult to convince them that a fetus can not only survive a mother’s cancer treatment, but have a good chance of developing normally."
The stress of a cancer diagnosis during a desired pregnancy is very hard on patients, Dr. Temkin added. "Pregnancy is a time when many women come to grips with their own mortality as well as that of giving new life. Adding a diagnosis of cancer of top of that – especially in the face of a much-desired pregnancy – can be devastating."
These women are faced with two options: terminate the pregnancy and concentrate on their own treatment, or continue the pregnancy knowing that their unborn child will be exposed to the possible risks of radiation, chemotherapy, and surgery. Either option can "inflict terrible guilt on a pregnant woman. We can try to minimize that to some degree, but it’s important to know from the outset that what is the right solution for one patient is not right for the next."
Connecting with other women who have experienced the same situation can be of immense value, Dr. Cardonick said. She participates in an online support group called "Hope for Two."
The organization’s main goal is to link new patients with survivors who can help educate them as well as lend emotional support. Patients call in or fill out a secure online request for a personal match-up with a survivor, who is often a woman who has had the same type of cancer.
The website also contains links to news and medical articles, books, and financial assistance sources, and allows new patients to securely contact Dr. Cardonick’s pregnancy registry. "We keep in touch with the baby’s pediatrician and the mom every year, to see how things are going and [to] collect information," she said. "The best way to treat these women in the future depends on the information we continue to gather in the present."
None of the researchers interviewed for this article had any relevant financial disclosures. Dr. Litton noted that she had no financial disclosures for her 2011 ASCO poster.
Amyloid Imaging Agent May Aid Alzheimer's Diagnosis
The Food and Drug Administration’s approval of the first amyloid plaque-imaging agent, florbetapir F18, may usher in a "new age" in the diagnosis of Alzheimer’s disease, according to Dr. Daniel Skovronsky.
"We are entering a new age in which we can evaluate patients [with cognitive decline] based on the molecular pathology in the brain," rather than on symptoms alone, Dr. Skovronsky, president of Avid Radiopharmaceuticals, said in a press briefing on April 9. "We hope this will also lead to the generation of therapies that don’t only target the symptoms but also the molecular pathology of the disease. It’s our long-term vision that through early detection and interventions, someday we can make Alzheimer’s a preventable disease."
Florbetapir (Amyvid), which was approved on April 6, binds to beta-amyloid plaques in the brain, the diagnostic hallmark of Alzheimer’s. Until now, clinicians have based the diagnosis on symptoms and cognitive testing, although they have been estimated to misdiagnose about a fifth of patients with probable Alzheimer’s based on definitive diagnosis established at autopsy. A florbetapir scan can now reliably exclude a diagnosis of Alzheimer’s, Dr. Skovronsky said.
"A positive scan does not mean the patients have Alzheimer’s, since neuritic plaques can be seen in other diseases and are also sometimes present in cognitively normal people as they age," he noted.
The scans should be used in conjunction with clinical findings to help shape a patient’s treatment, he added.
But the ability to pinpoint amyloid status may create more problems for a population already struggling with a difficult diagnosis, according to the Alzheimer’s Association. Although the group supports the approval, it called florbetapir’s approval a "double-edged sword," and expressed concerns about how florbetapir could be used.
"On the one hand ... this product will expand the clinical and research opportunities for amyloid imaging by making this brain imaging tool more widely available to the field. On the other hand, the fact that all of the potential uses of this product are not crystal clear tempers our enthusiasm. Again, additional research is needed to clarify the role of florbetapir-PET imaging in Alzheimer’s," the association said.
The product could be used to exploit patients as well. "The Alzheimer’s Association is also concerned about the possibility of less-than-scrupulous operators offering imaging services and making unrealistic promises about the value of florbetapir imaging to sometimes vulnerable and worried individuals. As such, we recommend that people get access to this test only in the context of a complete evaluation of medical/neurological status and with appropriate expert consultation."
Last year, an FDA advisory panel voted against immediate approval of florbetapir because of concerns about interreader variability and the lack of a single reading technique. Since then, Avid Radiopharmaceuticals and its parent company, Eli Lilly, have created a reader training tool, which has been shown to improve scan interpretation, Dr. Skovronsky said. The training program will be available online and in person to clinicians who will be interpreting the scans.
He said Lilly did not compare inter-reader reproducibility before and after the training program. But after the training, readers who re-evaluated scans in the three pivotal approval trials agreed on the results 83% of the time, according to a Lilly spokesperson.
The company compared online and in-person training strategies in one of the three trials, which assessed positive scans in relation to a pathologic diagnosis of Alzheimer’s. Scans evaluated by readers trained in person showed a median sensitivity of 92% and a specificity of 95%. Those evaluated by readers trained online were slightly less accurate, with a median 82% sensitivity and 95% specificity. The company did not say whether these differences were statistically significant or clinically meaningful.
Florbetapir is also playing a role in research. The lack of a reliable biomarker of Alzheimer’s disease progression has hampered research into disease-modifying therapies. Imaging compounds, including florbetapir, are being investigated for their ability to provide visual markers that could show any response to drugs designed to affect beta-amyloid plaque levels.
(Click here for the full prescribing information for florbetapir.)
The Food and Drug Administration’s approval of the first amyloid plaque-imaging agent, florbetapir F18, may usher in a "new age" in the diagnosis of Alzheimer’s disease, according to Dr. Daniel Skovronsky.
"We are entering a new age in which we can evaluate patients [with cognitive decline] based on the molecular pathology in the brain," rather than on symptoms alone, Dr. Skovronsky, president of Avid Radiopharmaceuticals, said in a press briefing on April 9. "We hope this will also lead to the generation of therapies that don’t only target the symptoms but also the molecular pathology of the disease. It’s our long-term vision that through early detection and interventions, someday we can make Alzheimer’s a preventable disease."
Florbetapir (Amyvid), which was approved on April 6, binds to beta-amyloid plaques in the brain, the diagnostic hallmark of Alzheimer’s. Until now, clinicians have based the diagnosis on symptoms and cognitive testing, although they have been estimated to misdiagnose about a fifth of patients with probable Alzheimer’s based on definitive diagnosis established at autopsy. A florbetapir scan can now reliably exclude a diagnosis of Alzheimer’s, Dr. Skovronsky said.
"A positive scan does not mean the patients have Alzheimer’s, since neuritic plaques can be seen in other diseases and are also sometimes present in cognitively normal people as they age," he noted.
The scans should be used in conjunction with clinical findings to help shape a patient’s treatment, he added.
But the ability to pinpoint amyloid status may create more problems for a population already struggling with a difficult diagnosis, according to the Alzheimer’s Association. Although the group supports the approval, it called florbetapir’s approval a "double-edged sword," and expressed concerns about how florbetapir could be used.
"On the one hand ... this product will expand the clinical and research opportunities for amyloid imaging by making this brain imaging tool more widely available to the field. On the other hand, the fact that all of the potential uses of this product are not crystal clear tempers our enthusiasm. Again, additional research is needed to clarify the role of florbetapir-PET imaging in Alzheimer’s," the association said.
The product could be used to exploit patients as well. "The Alzheimer’s Association is also concerned about the possibility of less-than-scrupulous operators offering imaging services and making unrealistic promises about the value of florbetapir imaging to sometimes vulnerable and worried individuals. As such, we recommend that people get access to this test only in the context of a complete evaluation of medical/neurological status and with appropriate expert consultation."
Last year, an FDA advisory panel voted against immediate approval of florbetapir because of concerns about interreader variability and the lack of a single reading technique. Since then, Avid Radiopharmaceuticals and its parent company, Eli Lilly, have created a reader training tool, which has been shown to improve scan interpretation, Dr. Skovronsky said. The training program will be available online and in person to clinicians who will be interpreting the scans.
He said Lilly did not compare inter-reader reproducibility before and after the training program. But after the training, readers who re-evaluated scans in the three pivotal approval trials agreed on the results 83% of the time, according to a Lilly spokesperson.
The company compared online and in-person training strategies in one of the three trials, which assessed positive scans in relation to a pathologic diagnosis of Alzheimer’s. Scans evaluated by readers trained in person showed a median sensitivity of 92% and a specificity of 95%. Those evaluated by readers trained online were slightly less accurate, with a median 82% sensitivity and 95% specificity. The company did not say whether these differences were statistically significant or clinically meaningful.
Florbetapir is also playing a role in research. The lack of a reliable biomarker of Alzheimer’s disease progression has hampered research into disease-modifying therapies. Imaging compounds, including florbetapir, are being investigated for their ability to provide visual markers that could show any response to drugs designed to affect beta-amyloid plaque levels.
(Click here for the full prescribing information for florbetapir.)
The Food and Drug Administration’s approval of the first amyloid plaque-imaging agent, florbetapir F18, may usher in a "new age" in the diagnosis of Alzheimer’s disease, according to Dr. Daniel Skovronsky.
"We are entering a new age in which we can evaluate patients [with cognitive decline] based on the molecular pathology in the brain," rather than on symptoms alone, Dr. Skovronsky, president of Avid Radiopharmaceuticals, said in a press briefing on April 9. "We hope this will also lead to the generation of therapies that don’t only target the symptoms but also the molecular pathology of the disease. It’s our long-term vision that through early detection and interventions, someday we can make Alzheimer’s a preventable disease."
Florbetapir (Amyvid), which was approved on April 6, binds to beta-amyloid plaques in the brain, the diagnostic hallmark of Alzheimer’s. Until now, clinicians have based the diagnosis on symptoms and cognitive testing, although they have been estimated to misdiagnose about a fifth of patients with probable Alzheimer’s based on definitive diagnosis established at autopsy. A florbetapir scan can now reliably exclude a diagnosis of Alzheimer’s, Dr. Skovronsky said.
"A positive scan does not mean the patients have Alzheimer’s, since neuritic plaques can be seen in other diseases and are also sometimes present in cognitively normal people as they age," he noted.
The scans should be used in conjunction with clinical findings to help shape a patient’s treatment, he added.
But the ability to pinpoint amyloid status may create more problems for a population already struggling with a difficult diagnosis, according to the Alzheimer’s Association. Although the group supports the approval, it called florbetapir’s approval a "double-edged sword," and expressed concerns about how florbetapir could be used.
"On the one hand ... this product will expand the clinical and research opportunities for amyloid imaging by making this brain imaging tool more widely available to the field. On the other hand, the fact that all of the potential uses of this product are not crystal clear tempers our enthusiasm. Again, additional research is needed to clarify the role of florbetapir-PET imaging in Alzheimer’s," the association said.
The product could be used to exploit patients as well. "The Alzheimer’s Association is also concerned about the possibility of less-than-scrupulous operators offering imaging services and making unrealistic promises about the value of florbetapir imaging to sometimes vulnerable and worried individuals. As such, we recommend that people get access to this test only in the context of a complete evaluation of medical/neurological status and with appropriate expert consultation."
Last year, an FDA advisory panel voted against immediate approval of florbetapir because of concerns about interreader variability and the lack of a single reading technique. Since then, Avid Radiopharmaceuticals and its parent company, Eli Lilly, have created a reader training tool, which has been shown to improve scan interpretation, Dr. Skovronsky said. The training program will be available online and in person to clinicians who will be interpreting the scans.
He said Lilly did not compare inter-reader reproducibility before and after the training program. But after the training, readers who re-evaluated scans in the three pivotal approval trials agreed on the results 83% of the time, according to a Lilly spokesperson.
The company compared online and in-person training strategies in one of the three trials, which assessed positive scans in relation to a pathologic diagnosis of Alzheimer’s. Scans evaluated by readers trained in person showed a median sensitivity of 92% and a specificity of 95%. Those evaluated by readers trained online were slightly less accurate, with a median 82% sensitivity and 95% specificity. The company did not say whether these differences were statistically significant or clinically meaningful.
Florbetapir is also playing a role in research. The lack of a reliable biomarker of Alzheimer’s disease progression has hampered research into disease-modifying therapies. Imaging compounds, including florbetapir, are being investigated for their ability to provide visual markers that could show any response to drugs designed to affect beta-amyloid plaque levels.
(Click here for the full prescribing information for florbetapir.)
Small Changes Yield Big Bang for Chlamydia Screening Buck
MINNEAPOLIS – A few simple administrative changes made some big differences at clinics that screen for chlamydia, increasing the number of screenings and saving more than $40,000 in just 1 year.
Chlamydia is the most common nationally reportable infectious disease, with 1.4 million cases diagnosed in the U.S. in 2011. Yet it’s also one of the most underdiagnosed, Elizabeth Torrone, Ph.D. said at a conference on STD prevention sponsored by the Centers for Disease Control and Prevention.
Despite its ability to hide in plain sight, chlamydia can have serious health consequences for young women, said Dr. Torrone, an epidemiologist at the CDC.
"It’s most common in young women and it’s usually asymptomatic, but 1 in 10 infections results in pelvic inflammatory disease, which can have adverse effects on reproduction. CDC recommends annual screening for all sexually active women aged 25 or younger, and for older women who are at high risk for infection," such as those with new or multiple sexual partners.
Chlamydia rates seem to have increased significantly in the past decade, but that’s most likely the result of increased screening. "The more we look for it, the more we find." But screening still doesn’t match up to the CDC guidelines, she said. "Since 2001, screening among females aged 16-24 years has increased to 60% of those targeted compared to 30% before 2001. But older women still have higher screening rates than necessary, because only about 1% of the positives occur in women aged 25-39 years."
She described how three health departments have tackled the problem, and the benefits they reaped from a few low-cost changes in their screening routine.
San Francisco Enforces the Guidelines
In San Francisco, health department family planning clinics were charged with screening any woman younger than 25 years, and all women who were pregnant or getting an intrauterine birth control device inserted. But clinicians still could screen other groups: The lab requisition slip had an "other" indication, where clinicians could write in any other reason they thought a screen was necessary.
In 2008, Dr. Torrone said, 64% of the chlamydia screening in these clinics was done on women older than 26 years. "This led us to conclude they were over-screening the older population," she said.
To address the problem, the health department simply removed the "other" box on the lab slip, leaving only three choices: younger than 26 years, pregnant, or IUD insertion. The labs rejected any specimen without one of these valid reasons for testing marked.
"The only way you could screen a woman older than 26 was if she was pregnant or having an IUD inserted. So this really forced our clinicians to adhere to the screening guidelines," she said.
When they compared screening in 2008 with 2009 – after the new slips came out – there was a 25% decrease in the number of tests on women 26 and older, and a 4% increase in positive tests – indicating that more young women were being screened.
The change also dramatically decreased costs, Dr. Torrone said. "For every screen, the cost decreased by 3.5%, resulting in a savings of almost $40,000."
Kentucky’s Experience
In 2009, to assure that clinics were screening appropriately and to make screening financially feasible, CDC implemented a program designed to produce at least a 3% positivity rate at any clinic that screened for chlamydia.
"In Kentucky, 75 sites participating in [the program] had a positivity rate of less than 3% in women younger than 26, so CDC offered some assistance in getting that increased," she said.
The plan included site-specific feedback to those clinics not meeting the 3% rule. Feedback included the clinic’s positivity rate, implementing risk-based screening criteria, and individual technical assistance to help out in some sites.
By 2010, 54 sites were reporting positivity rates of less than 3%. * "Obviously Kentucky still has some work to do, but they plan to target high-volume sites and intensify communication and teamwork with providers."
Specimen Pooling in Idaho
Specimen pooling proved effective in Idaho, a state with a relatively low number of positive chlamydia tests. Specimen pooling is a two-step lab procedure that batches samples. Each batch has a single test. If the result is negative then every sample in that batch is negative, eliminating individual sample testing. If the batch tests positive, then every sample in that group must be tested.
The technique saves money on every specimen that goes into a negative batch, Dr. Torrone said.
It’s most cost-effective in areas with low chlamydia prevalence.
In 2009, the Idaho Bureau of Laboratories instituted the pooling technique. In July of that year, 1,300 samples were submitted. Using the pooling technique, the lab ran 727 tests to identify all the positive samples.
"Assuming a cost of $14.47 per test, this saved 47% on the positive tests," Dr. Torrone said. "The lab estimated that they were able to test 15,201 samples for the price of 8,866 samples."
Dr. Torrone reported having no financial disclosures.
*Correction, 4/13/12: An earlier version of this story misreported the percentage of the reduction in the number of clinics in Kentucky reporting positivity rates of less than 3%.
MINNEAPOLIS – A few simple administrative changes made some big differences at clinics that screen for chlamydia, increasing the number of screenings and saving more than $40,000 in just 1 year.
Chlamydia is the most common nationally reportable infectious disease, with 1.4 million cases diagnosed in the U.S. in 2011. Yet it’s also one of the most underdiagnosed, Elizabeth Torrone, Ph.D. said at a conference on STD prevention sponsored by the Centers for Disease Control and Prevention.
Despite its ability to hide in plain sight, chlamydia can have serious health consequences for young women, said Dr. Torrone, an epidemiologist at the CDC.
"It’s most common in young women and it’s usually asymptomatic, but 1 in 10 infections results in pelvic inflammatory disease, which can have adverse effects on reproduction. CDC recommends annual screening for all sexually active women aged 25 or younger, and for older women who are at high risk for infection," such as those with new or multiple sexual partners.
Chlamydia rates seem to have increased significantly in the past decade, but that’s most likely the result of increased screening. "The more we look for it, the more we find." But screening still doesn’t match up to the CDC guidelines, she said. "Since 2001, screening among females aged 16-24 years has increased to 60% of those targeted compared to 30% before 2001. But older women still have higher screening rates than necessary, because only about 1% of the positives occur in women aged 25-39 years."
She described how three health departments have tackled the problem, and the benefits they reaped from a few low-cost changes in their screening routine.
San Francisco Enforces the Guidelines
In San Francisco, health department family planning clinics were charged with screening any woman younger than 25 years, and all women who were pregnant or getting an intrauterine birth control device inserted. But clinicians still could screen other groups: The lab requisition slip had an "other" indication, where clinicians could write in any other reason they thought a screen was necessary.
In 2008, Dr. Torrone said, 64% of the chlamydia screening in these clinics was done on women older than 26 years. "This led us to conclude they were over-screening the older population," she said.
To address the problem, the health department simply removed the "other" box on the lab slip, leaving only three choices: younger than 26 years, pregnant, or IUD insertion. The labs rejected any specimen without one of these valid reasons for testing marked.
"The only way you could screen a woman older than 26 was if she was pregnant or having an IUD inserted. So this really forced our clinicians to adhere to the screening guidelines," she said.
When they compared screening in 2008 with 2009 – after the new slips came out – there was a 25% decrease in the number of tests on women 26 and older, and a 4% increase in positive tests – indicating that more young women were being screened.
The change also dramatically decreased costs, Dr. Torrone said. "For every screen, the cost decreased by 3.5%, resulting in a savings of almost $40,000."
Kentucky’s Experience
In 2009, to assure that clinics were screening appropriately and to make screening financially feasible, CDC implemented a program designed to produce at least a 3% positivity rate at any clinic that screened for chlamydia.
"In Kentucky, 75 sites participating in [the program] had a positivity rate of less than 3% in women younger than 26, so CDC offered some assistance in getting that increased," she said.
The plan included site-specific feedback to those clinics not meeting the 3% rule. Feedback included the clinic’s positivity rate, implementing risk-based screening criteria, and individual technical assistance to help out in some sites.
By 2010, 54 sites were reporting positivity rates of less than 3%. * "Obviously Kentucky still has some work to do, but they plan to target high-volume sites and intensify communication and teamwork with providers."
Specimen Pooling in Idaho
Specimen pooling proved effective in Idaho, a state with a relatively low number of positive chlamydia tests. Specimen pooling is a two-step lab procedure that batches samples. Each batch has a single test. If the result is negative then every sample in that batch is negative, eliminating individual sample testing. If the batch tests positive, then every sample in that group must be tested.
The technique saves money on every specimen that goes into a negative batch, Dr. Torrone said.
It’s most cost-effective in areas with low chlamydia prevalence.
In 2009, the Idaho Bureau of Laboratories instituted the pooling technique. In July of that year, 1,300 samples were submitted. Using the pooling technique, the lab ran 727 tests to identify all the positive samples.
"Assuming a cost of $14.47 per test, this saved 47% on the positive tests," Dr. Torrone said. "The lab estimated that they were able to test 15,201 samples for the price of 8,866 samples."
Dr. Torrone reported having no financial disclosures.
*Correction, 4/13/12: An earlier version of this story misreported the percentage of the reduction in the number of clinics in Kentucky reporting positivity rates of less than 3%.
MINNEAPOLIS – A few simple administrative changes made some big differences at clinics that screen for chlamydia, increasing the number of screenings and saving more than $40,000 in just 1 year.
Chlamydia is the most common nationally reportable infectious disease, with 1.4 million cases diagnosed in the U.S. in 2011. Yet it’s also one of the most underdiagnosed, Elizabeth Torrone, Ph.D. said at a conference on STD prevention sponsored by the Centers for Disease Control and Prevention.
Despite its ability to hide in plain sight, chlamydia can have serious health consequences for young women, said Dr. Torrone, an epidemiologist at the CDC.
"It’s most common in young women and it’s usually asymptomatic, but 1 in 10 infections results in pelvic inflammatory disease, which can have adverse effects on reproduction. CDC recommends annual screening for all sexually active women aged 25 or younger, and for older women who are at high risk for infection," such as those with new or multiple sexual partners.
Chlamydia rates seem to have increased significantly in the past decade, but that’s most likely the result of increased screening. "The more we look for it, the more we find." But screening still doesn’t match up to the CDC guidelines, she said. "Since 2001, screening among females aged 16-24 years has increased to 60% of those targeted compared to 30% before 2001. But older women still have higher screening rates than necessary, because only about 1% of the positives occur in women aged 25-39 years."
She described how three health departments have tackled the problem, and the benefits they reaped from a few low-cost changes in their screening routine.
San Francisco Enforces the Guidelines
In San Francisco, health department family planning clinics were charged with screening any woman younger than 25 years, and all women who were pregnant or getting an intrauterine birth control device inserted. But clinicians still could screen other groups: The lab requisition slip had an "other" indication, where clinicians could write in any other reason they thought a screen was necessary.
In 2008, Dr. Torrone said, 64% of the chlamydia screening in these clinics was done on women older than 26 years. "This led us to conclude they were over-screening the older population," she said.
To address the problem, the health department simply removed the "other" box on the lab slip, leaving only three choices: younger than 26 years, pregnant, or IUD insertion. The labs rejected any specimen without one of these valid reasons for testing marked.
"The only way you could screen a woman older than 26 was if she was pregnant or having an IUD inserted. So this really forced our clinicians to adhere to the screening guidelines," she said.
When they compared screening in 2008 with 2009 – after the new slips came out – there was a 25% decrease in the number of tests on women 26 and older, and a 4% increase in positive tests – indicating that more young women were being screened.
The change also dramatically decreased costs, Dr. Torrone said. "For every screen, the cost decreased by 3.5%, resulting in a savings of almost $40,000."
Kentucky’s Experience
In 2009, to assure that clinics were screening appropriately and to make screening financially feasible, CDC implemented a program designed to produce at least a 3% positivity rate at any clinic that screened for chlamydia.
"In Kentucky, 75 sites participating in [the program] had a positivity rate of less than 3% in women younger than 26, so CDC offered some assistance in getting that increased," she said.
The plan included site-specific feedback to those clinics not meeting the 3% rule. Feedback included the clinic’s positivity rate, implementing risk-based screening criteria, and individual technical assistance to help out in some sites.
By 2010, 54 sites were reporting positivity rates of less than 3%. * "Obviously Kentucky still has some work to do, but they plan to target high-volume sites and intensify communication and teamwork with providers."
Specimen Pooling in Idaho
Specimen pooling proved effective in Idaho, a state with a relatively low number of positive chlamydia tests. Specimen pooling is a two-step lab procedure that batches samples. Each batch has a single test. If the result is negative then every sample in that batch is negative, eliminating individual sample testing. If the batch tests positive, then every sample in that group must be tested.
The technique saves money on every specimen that goes into a negative batch, Dr. Torrone said.
It’s most cost-effective in areas with low chlamydia prevalence.
In 2009, the Idaho Bureau of Laboratories instituted the pooling technique. In July of that year, 1,300 samples were submitted. Using the pooling technique, the lab ran 727 tests to identify all the positive samples.
"Assuming a cost of $14.47 per test, this saved 47% on the positive tests," Dr. Torrone said. "The lab estimated that they were able to test 15,201 samples for the price of 8,866 samples."
Dr. Torrone reported having no financial disclosures.
*Correction, 4/13/12: An earlier version of this story misreported the percentage of the reduction in the number of clinics in Kentucky reporting positivity rates of less than 3%.
FROM A CONFERENCE ON STD PREVENTION SPONSORED BY THE CENTERS FOR DISEASE CONTROL AND PREVENTION
Gonorrhea Continues to Outsmart Antibiotics
MINNEAPOLIS – Neisseria gonorrhoeae is probing the final frontier of antimicrobial treatment.
The persistent and versatile bacteria has proved its mettle against every antibiotic humans have thrown at it over the past 70 years, and now shows signs of figuring out the cephalosporins – the last bastion of effective antimicrobial treatment.
The Centers for Disease Control and Prevention have not seen any case of cephalosporin treatment failures in the United States yet. But researchers at the National STD Conference said it’s only a matter of time.
"We have already seen oral cephalosporin treatment failures in Asia and, in the last few years, in Europe," said Mark Pandori, Ph.D, of the San Francisco Department of Public Health. "None have yet been recorded in the U.S. but it’s wise to assume that it is on our doorstep, if not already here."
The bacteria developed fluoroquinolone resistance in the early 2000s, prompting CDC to drop that drug from its treatment recommendation in 2010. Now the agency recommends dual therapy: a 250-mg injection of ceftriaxone or a 400-mg dose of cefixeme, plus a 1-g dose of azithromycin or a 7-day course of doxycycline.
But now azithromycin resistance is becoming more common. Last July, the agency issued an alert about a cluster of five azithromycin-resistant isolates from a group of men who have sex with men (MSM) in San Diego.
In fact, one case of azithromycin treatment failure has occurred in the U.S., Olusegun Soge, Ph.D. reported at the meeting. After 12 days of treatment with the drug, the bacteria had already developed an increased resistance level, said Dr. Soge, an epidemiologist at the University of Washington, Seattle.
The patient was a 26-year-old MSM who received 2 grams of azithromycin. A culture showed a high level of azithromycin resistance. After 12 days of treatment, the patient was still experiencing symptoms consistent with a gonorrhea infection. He denied any sexual relations during treatment.
"We needed to know if this was a possible treatment failure, or a re-exposure," Dr. Soge said. Nucleic acid amplification testing (NAAT) determined that both isolates showed the same genetic blueprint, but the new specimen had an even higher resistance level: a minimum inhibitory concentration (MIC) of 8 mcg/mL compared to 0.5 mcg/mL in the first sample.
"The emergence of a resistant variant in an individual patient during treatment is alarming and may predict rapid emergence of gonococcal resistance," he said. He recommended that any patient who receives azithromycin as a single treatment should return for a test of cure.
At this point, cephalosporins are really the only treatment with certain effectiveness, said Dr. Sarah Guerry, medical director, of the sexually transmitted disease program at the Los Angeles County Public Health. "Not only have we had to increase the recommended dosage of ceftriaxone, but now this is our therapy of last resort," she said at the meeting.
Many STD clinics, however, are only able to distribute oral medications. Dr. Guerry and her colleagues examined whether complying with the new treatment recommendations of intramuscular ceftriaxone was delaying treatment.
She reviewed all urogenital and anorectal gonorrhea cases diagnosed in Los Angeles County from April-August 2010 – before the new treatment guidelines – and from the same period in 2011. The review focused on clinics that treated less than 10% of cases with any dose of ceftriaxone injection, and compared the overall and median time to treatment, as well as compliance with the new recommendations.
There were 508 gonorrhea cases in 2010 and 401 in 2011. Overall, 11% of the clinics surveyed were considered "low injectors."
Almost three-fourths of the clinics (72%) dispensed some form of the treatment recommended for that period. This did not change after the new guidelines came out, indicating that most clinics adopted the new guideline. But in 2010, only 13% of the facilities were giving the injected ceftriaxone. In 2011, there was a dramatic change, with 90% of the facilities giving the injection.
Time to treatment, however, was not significantly altered, indicating that switching to the injected form did not delay treatment. Overall, the time from diagnosis to treatment increased from 5 to 7 days.
Still, there is plenty of room for improvement in delivering the recommended treatment, according to Roxanne Kerani, Ph.D., an epidemiologist at the Seattle and King County Public Health Department, Wash. She collaborated with CDC researchers to estimate how frequently clinicians across the country are delivering treatment according to the new CDC guidelines. They polled clinics included in the national STD Surveillance Network: Washington, California, Colorado, Baltimore, and San Francisco.
Overall, 80% of patients with gonorrhea received ceftriaxone as part of their treatment. But only 45% received the recommended dual therapy. About a third got ceftriaxone alone.
The incidence of dual treatment varied significantly according to the type of site, she said. STD clinics were most likely to administer dual therapy (88%), which was significantly more than hospitals (79%), private providers (71%) or family planning clinics. Men, particularly MSM, were significantly more likely to receive the recommended therapy than were women, regardless of their sexual preference.
"We believe that continued surveillance of treatment practices is important, given the changing antibiotic susceptibility patterns we’re seeing, and the limited alternative treatment options for gonorrhea," she said.
Despite consternation over the creeping threat of resistance, gonorrhea is not nearly as common as it once was, said Mark Stenger, an epidemiologist at the CDC.
"We are in a good epidemiologic moment with gonorrhea rates in the U.S. right now. There have been incredible declines in rates overall."
According to the CDC, the national gonorrhea rate declined by 74% from 1975 to 1997 – largely due to a national gonorrhea control program instituted in the mid-70s, Dr. Stenger said. In 2010, the overall rate was 101 per 100,000 persons, with a total of 309,341 cases diagnosed that year.
"This is the lowest rate since we began recording it," he said.
Healthy People 2020 sets what Mr. Stenger called "realistic goals" for gonorrhea incidence: 257 per 100,000 persons for women and 198 per 100,000 persons for men by the end of that year.
"When we look at incidence among males in the 2011 unpublished data, there are a number of counties that are not meeting that goal right now ... and a number of counties that are. We need to work in those areas that don’t meet it and continue to do whatever we are doing well to keep cases low in the areas that have already met this target."
None of the presenters said they have any financial disclosures.
MINNEAPOLIS – Neisseria gonorrhoeae is probing the final frontier of antimicrobial treatment.
The persistent and versatile bacteria has proved its mettle against every antibiotic humans have thrown at it over the past 70 years, and now shows signs of figuring out the cephalosporins – the last bastion of effective antimicrobial treatment.
The Centers for Disease Control and Prevention have not seen any case of cephalosporin treatment failures in the United States yet. But researchers at the National STD Conference said it’s only a matter of time.
"We have already seen oral cephalosporin treatment failures in Asia and, in the last few years, in Europe," said Mark Pandori, Ph.D, of the San Francisco Department of Public Health. "None have yet been recorded in the U.S. but it’s wise to assume that it is on our doorstep, if not already here."
The bacteria developed fluoroquinolone resistance in the early 2000s, prompting CDC to drop that drug from its treatment recommendation in 2010. Now the agency recommends dual therapy: a 250-mg injection of ceftriaxone or a 400-mg dose of cefixeme, plus a 1-g dose of azithromycin or a 7-day course of doxycycline.
But now azithromycin resistance is becoming more common. Last July, the agency issued an alert about a cluster of five azithromycin-resistant isolates from a group of men who have sex with men (MSM) in San Diego.
In fact, one case of azithromycin treatment failure has occurred in the U.S., Olusegun Soge, Ph.D. reported at the meeting. After 12 days of treatment with the drug, the bacteria had already developed an increased resistance level, said Dr. Soge, an epidemiologist at the University of Washington, Seattle.
The patient was a 26-year-old MSM who received 2 grams of azithromycin. A culture showed a high level of azithromycin resistance. After 12 days of treatment, the patient was still experiencing symptoms consistent with a gonorrhea infection. He denied any sexual relations during treatment.
"We needed to know if this was a possible treatment failure, or a re-exposure," Dr. Soge said. Nucleic acid amplification testing (NAAT) determined that both isolates showed the same genetic blueprint, but the new specimen had an even higher resistance level: a minimum inhibitory concentration (MIC) of 8 mcg/mL compared to 0.5 mcg/mL in the first sample.
"The emergence of a resistant variant in an individual patient during treatment is alarming and may predict rapid emergence of gonococcal resistance," he said. He recommended that any patient who receives azithromycin as a single treatment should return for a test of cure.
At this point, cephalosporins are really the only treatment with certain effectiveness, said Dr. Sarah Guerry, medical director, of the sexually transmitted disease program at the Los Angeles County Public Health. "Not only have we had to increase the recommended dosage of ceftriaxone, but now this is our therapy of last resort," she said at the meeting.
Many STD clinics, however, are only able to distribute oral medications. Dr. Guerry and her colleagues examined whether complying with the new treatment recommendations of intramuscular ceftriaxone was delaying treatment.
She reviewed all urogenital and anorectal gonorrhea cases diagnosed in Los Angeles County from April-August 2010 – before the new treatment guidelines – and from the same period in 2011. The review focused on clinics that treated less than 10% of cases with any dose of ceftriaxone injection, and compared the overall and median time to treatment, as well as compliance with the new recommendations.
There were 508 gonorrhea cases in 2010 and 401 in 2011. Overall, 11% of the clinics surveyed were considered "low injectors."
Almost three-fourths of the clinics (72%) dispensed some form of the treatment recommended for that period. This did not change after the new guidelines came out, indicating that most clinics adopted the new guideline. But in 2010, only 13% of the facilities were giving the injected ceftriaxone. In 2011, there was a dramatic change, with 90% of the facilities giving the injection.
Time to treatment, however, was not significantly altered, indicating that switching to the injected form did not delay treatment. Overall, the time from diagnosis to treatment increased from 5 to 7 days.
Still, there is plenty of room for improvement in delivering the recommended treatment, according to Roxanne Kerani, Ph.D., an epidemiologist at the Seattle and King County Public Health Department, Wash. She collaborated with CDC researchers to estimate how frequently clinicians across the country are delivering treatment according to the new CDC guidelines. They polled clinics included in the national STD Surveillance Network: Washington, California, Colorado, Baltimore, and San Francisco.
Overall, 80% of patients with gonorrhea received ceftriaxone as part of their treatment. But only 45% received the recommended dual therapy. About a third got ceftriaxone alone.
The incidence of dual treatment varied significantly according to the type of site, she said. STD clinics were most likely to administer dual therapy (88%), which was significantly more than hospitals (79%), private providers (71%) or family planning clinics. Men, particularly MSM, were significantly more likely to receive the recommended therapy than were women, regardless of their sexual preference.
"We believe that continued surveillance of treatment practices is important, given the changing antibiotic susceptibility patterns we’re seeing, and the limited alternative treatment options for gonorrhea," she said.
Despite consternation over the creeping threat of resistance, gonorrhea is not nearly as common as it once was, said Mark Stenger, an epidemiologist at the CDC.
"We are in a good epidemiologic moment with gonorrhea rates in the U.S. right now. There have been incredible declines in rates overall."
According to the CDC, the national gonorrhea rate declined by 74% from 1975 to 1997 – largely due to a national gonorrhea control program instituted in the mid-70s, Dr. Stenger said. In 2010, the overall rate was 101 per 100,000 persons, with a total of 309,341 cases diagnosed that year.
"This is the lowest rate since we began recording it," he said.
Healthy People 2020 sets what Mr. Stenger called "realistic goals" for gonorrhea incidence: 257 per 100,000 persons for women and 198 per 100,000 persons for men by the end of that year.
"When we look at incidence among males in the 2011 unpublished data, there are a number of counties that are not meeting that goal right now ... and a number of counties that are. We need to work in those areas that don’t meet it and continue to do whatever we are doing well to keep cases low in the areas that have already met this target."
None of the presenters said they have any financial disclosures.
MINNEAPOLIS – Neisseria gonorrhoeae is probing the final frontier of antimicrobial treatment.
The persistent and versatile bacteria has proved its mettle against every antibiotic humans have thrown at it over the past 70 years, and now shows signs of figuring out the cephalosporins – the last bastion of effective antimicrobial treatment.
The Centers for Disease Control and Prevention have not seen any case of cephalosporin treatment failures in the United States yet. But researchers at the National STD Conference said it’s only a matter of time.
"We have already seen oral cephalosporin treatment failures in Asia and, in the last few years, in Europe," said Mark Pandori, Ph.D, of the San Francisco Department of Public Health. "None have yet been recorded in the U.S. but it’s wise to assume that it is on our doorstep, if not already here."
The bacteria developed fluoroquinolone resistance in the early 2000s, prompting CDC to drop that drug from its treatment recommendation in 2010. Now the agency recommends dual therapy: a 250-mg injection of ceftriaxone or a 400-mg dose of cefixeme, plus a 1-g dose of azithromycin or a 7-day course of doxycycline.
But now azithromycin resistance is becoming more common. Last July, the agency issued an alert about a cluster of five azithromycin-resistant isolates from a group of men who have sex with men (MSM) in San Diego.
In fact, one case of azithromycin treatment failure has occurred in the U.S., Olusegun Soge, Ph.D. reported at the meeting. After 12 days of treatment with the drug, the bacteria had already developed an increased resistance level, said Dr. Soge, an epidemiologist at the University of Washington, Seattle.
The patient was a 26-year-old MSM who received 2 grams of azithromycin. A culture showed a high level of azithromycin resistance. After 12 days of treatment, the patient was still experiencing symptoms consistent with a gonorrhea infection. He denied any sexual relations during treatment.
"We needed to know if this was a possible treatment failure, or a re-exposure," Dr. Soge said. Nucleic acid amplification testing (NAAT) determined that both isolates showed the same genetic blueprint, but the new specimen had an even higher resistance level: a minimum inhibitory concentration (MIC) of 8 mcg/mL compared to 0.5 mcg/mL in the first sample.
"The emergence of a resistant variant in an individual patient during treatment is alarming and may predict rapid emergence of gonococcal resistance," he said. He recommended that any patient who receives azithromycin as a single treatment should return for a test of cure.
At this point, cephalosporins are really the only treatment with certain effectiveness, said Dr. Sarah Guerry, medical director, of the sexually transmitted disease program at the Los Angeles County Public Health. "Not only have we had to increase the recommended dosage of ceftriaxone, but now this is our therapy of last resort," she said at the meeting.
Many STD clinics, however, are only able to distribute oral medications. Dr. Guerry and her colleagues examined whether complying with the new treatment recommendations of intramuscular ceftriaxone was delaying treatment.
She reviewed all urogenital and anorectal gonorrhea cases diagnosed in Los Angeles County from April-August 2010 – before the new treatment guidelines – and from the same period in 2011. The review focused on clinics that treated less than 10% of cases with any dose of ceftriaxone injection, and compared the overall and median time to treatment, as well as compliance with the new recommendations.
There were 508 gonorrhea cases in 2010 and 401 in 2011. Overall, 11% of the clinics surveyed were considered "low injectors."
Almost three-fourths of the clinics (72%) dispensed some form of the treatment recommended for that period. This did not change after the new guidelines came out, indicating that most clinics adopted the new guideline. But in 2010, only 13% of the facilities were giving the injected ceftriaxone. In 2011, there was a dramatic change, with 90% of the facilities giving the injection.
Time to treatment, however, was not significantly altered, indicating that switching to the injected form did not delay treatment. Overall, the time from diagnosis to treatment increased from 5 to 7 days.
Still, there is plenty of room for improvement in delivering the recommended treatment, according to Roxanne Kerani, Ph.D., an epidemiologist at the Seattle and King County Public Health Department, Wash. She collaborated with CDC researchers to estimate how frequently clinicians across the country are delivering treatment according to the new CDC guidelines. They polled clinics included in the national STD Surveillance Network: Washington, California, Colorado, Baltimore, and San Francisco.
Overall, 80% of patients with gonorrhea received ceftriaxone as part of their treatment. But only 45% received the recommended dual therapy. About a third got ceftriaxone alone.
The incidence of dual treatment varied significantly according to the type of site, she said. STD clinics were most likely to administer dual therapy (88%), which was significantly more than hospitals (79%), private providers (71%) or family planning clinics. Men, particularly MSM, were significantly more likely to receive the recommended therapy than were women, regardless of their sexual preference.
"We believe that continued surveillance of treatment practices is important, given the changing antibiotic susceptibility patterns we’re seeing, and the limited alternative treatment options for gonorrhea," she said.
Despite consternation over the creeping threat of resistance, gonorrhea is not nearly as common as it once was, said Mark Stenger, an epidemiologist at the CDC.
"We are in a good epidemiologic moment with gonorrhea rates in the U.S. right now. There have been incredible declines in rates overall."
According to the CDC, the national gonorrhea rate declined by 74% from 1975 to 1997 – largely due to a national gonorrhea control program instituted in the mid-70s, Dr. Stenger said. In 2010, the overall rate was 101 per 100,000 persons, with a total of 309,341 cases diagnosed that year.
"This is the lowest rate since we began recording it," he said.
Healthy People 2020 sets what Mr. Stenger called "realistic goals" for gonorrhea incidence: 257 per 100,000 persons for women and 198 per 100,000 persons for men by the end of that year.
"When we look at incidence among males in the 2011 unpublished data, there are a number of counties that are not meeting that goal right now ... and a number of counties that are. We need to work in those areas that don’t meet it and continue to do whatever we are doing well to keep cases low in the areas that have already met this target."
None of the presenters said they have any financial disclosures.
EXPERT ANALYSIS FROM THE NATIONAL STD CONFERENCE
Azithromycin, Doxycycline Quell Nongonococcal Urethritis in Men
MINNEAPOLIS – Azithromycin and doxycycline appear to be equally effective for men with nongonococcal urethritis.
In a randomized placebo-controlled trial, both drugs had good overall effectiveness, with an 84% clinical cure rate for azithromycin and a 78% rate for doxycycline. But some pathogens were more resistant to the drugs than others, Lisa Manhart, Ph.D., said at a conference on STD prevention sponsored by the Centers for Disease Control and Prevention.
The Mycoplasma Genitalium Antibiotic Susceptibility and Treatment (MEGA) trial enrolled 606 men with nongonococcal urethritis from 2007 to 2011. The main end points of the trial were clinical and microbiological cure of urethritis associated with M. genitalium infection, when treated with the standard therapy recommended by the Centers for Disease Control and Prevention: a single dose of 1 g of azithromycin, or a 7-day regimen of 100 mg of doxycycline taken twice a day.
Several pathogens cause nongonococcal urethritis, said Dr. Manhart, an epidemiologist at the University of Washington, Seattle. Among them are Chlamydia trachomatis, M. genitalium, the recently differentiated Ureaplasma urealyticum, and Trichomonas vaginalis. In some infections, no pathogen can be identified. In addition to presenting the end points for M. genitalium infections, Dr. Manhart broke the results out by other pathogens.
The mean age of the men in the trial was 34 years. Most (67%) were heterosexual, 28% were homosexual, and about 5% were bisexual. They reported a mean of six sexual partners in the past 12 months.
The most common infectious pathogen was chlamydia (26%), followed by U. urealyticum (24%). M. genitalium was present in 14% of the study group, and T. vaginalis in 2%. The remainder of the group had an idiopathic urethritis.
Treatment consisted of either a placebo azithromycin and active doxycycline or active azithromycin and placebo doxycycline. If men still showed symptoms or microbiological failure after treatment, they were offered reverse therapy (i.e., a treatment pack with the opposite active and placebo drugs), and returned for a follow-up visit in 3 weeks. If they were still not clinically or microbiologically cured, they received a course of moxifloxacin.
Overall, there was no statistically significant difference in cure rates between the two drugs. Nor were there significant differences among men with chlamydia infections, with a clinical cure rate of 87% for azithromycin and 78% for doxycycline. For microbiological cure, the rates were 86% and 90%, respectively.
Both clinical and microbiological cures rates were much lower in men infected with M. genitalium, but the rates were not significantly different between the two drugs. Clinical cure rates were 68% for azithromycin and 48% for doxycycline. For microbiological cure, the rates were 39% and 30%, respectively.
Men with U. urealyticum infections fared a little better, but again, the cure rates were not significantly different between the drugs. A clinical cure occurred in 85% of the azithromycin group and 75% of the doxycycline group. Microbiological cure rates were 75% and 69%, respectively.
For those with idiopathic urethritis, the cure rate for both drugs was identical: 87%.
Reverse therapy was still not effective in some men with U. urealyticum or M. genitalium infections. Three weeks after that treatment, 51% of those with M. genitalium and 57% of those with U. urealyticum were still positive for the pathogen. These men received a course of moxifloxacin.
"Moxifloxacin was quite effective, but at the end of the trial, we still had two men with M. genitalium and four with U. urealyticum who did not clear their infections," Dr. Manhart said.
Moxifloxacin, although highly effective, is too expensive to be used as first-time therapy in a public health department setting, she noted. "Its preinsurance cost runs up to $150, so we are still faced with a conundrum when treating these infections."
Dr. Manhart had no financial declarations.
MINNEAPOLIS – Azithromycin and doxycycline appear to be equally effective for men with nongonococcal urethritis.
In a randomized placebo-controlled trial, both drugs had good overall effectiveness, with an 84% clinical cure rate for azithromycin and a 78% rate for doxycycline. But some pathogens were more resistant to the drugs than others, Lisa Manhart, Ph.D., said at a conference on STD prevention sponsored by the Centers for Disease Control and Prevention.
The Mycoplasma Genitalium Antibiotic Susceptibility and Treatment (MEGA) trial enrolled 606 men with nongonococcal urethritis from 2007 to 2011. The main end points of the trial were clinical and microbiological cure of urethritis associated with M. genitalium infection, when treated with the standard therapy recommended by the Centers for Disease Control and Prevention: a single dose of 1 g of azithromycin, or a 7-day regimen of 100 mg of doxycycline taken twice a day.
Several pathogens cause nongonococcal urethritis, said Dr. Manhart, an epidemiologist at the University of Washington, Seattle. Among them are Chlamydia trachomatis, M. genitalium, the recently differentiated Ureaplasma urealyticum, and Trichomonas vaginalis. In some infections, no pathogen can be identified. In addition to presenting the end points for M. genitalium infections, Dr. Manhart broke the results out by other pathogens.
The mean age of the men in the trial was 34 years. Most (67%) were heterosexual, 28% were homosexual, and about 5% were bisexual. They reported a mean of six sexual partners in the past 12 months.
The most common infectious pathogen was chlamydia (26%), followed by U. urealyticum (24%). M. genitalium was present in 14% of the study group, and T. vaginalis in 2%. The remainder of the group had an idiopathic urethritis.
Treatment consisted of either a placebo azithromycin and active doxycycline or active azithromycin and placebo doxycycline. If men still showed symptoms or microbiological failure after treatment, they were offered reverse therapy (i.e., a treatment pack with the opposite active and placebo drugs), and returned for a follow-up visit in 3 weeks. If they were still not clinically or microbiologically cured, they received a course of moxifloxacin.
Overall, there was no statistically significant difference in cure rates between the two drugs. Nor were there significant differences among men with chlamydia infections, with a clinical cure rate of 87% for azithromycin and 78% for doxycycline. For microbiological cure, the rates were 86% and 90%, respectively.
Both clinical and microbiological cures rates were much lower in men infected with M. genitalium, but the rates were not significantly different between the two drugs. Clinical cure rates were 68% for azithromycin and 48% for doxycycline. For microbiological cure, the rates were 39% and 30%, respectively.
Men with U. urealyticum infections fared a little better, but again, the cure rates were not significantly different between the drugs. A clinical cure occurred in 85% of the azithromycin group and 75% of the doxycycline group. Microbiological cure rates were 75% and 69%, respectively.
For those with idiopathic urethritis, the cure rate for both drugs was identical: 87%.
Reverse therapy was still not effective in some men with U. urealyticum or M. genitalium infections. Three weeks after that treatment, 51% of those with M. genitalium and 57% of those with U. urealyticum were still positive for the pathogen. These men received a course of moxifloxacin.
"Moxifloxacin was quite effective, but at the end of the trial, we still had two men with M. genitalium and four with U. urealyticum who did not clear their infections," Dr. Manhart said.
Moxifloxacin, although highly effective, is too expensive to be used as first-time therapy in a public health department setting, she noted. "Its preinsurance cost runs up to $150, so we are still faced with a conundrum when treating these infections."
Dr. Manhart had no financial declarations.
MINNEAPOLIS – Azithromycin and doxycycline appear to be equally effective for men with nongonococcal urethritis.
In a randomized placebo-controlled trial, both drugs had good overall effectiveness, with an 84% clinical cure rate for azithromycin and a 78% rate for doxycycline. But some pathogens were more resistant to the drugs than others, Lisa Manhart, Ph.D., said at a conference on STD prevention sponsored by the Centers for Disease Control and Prevention.
The Mycoplasma Genitalium Antibiotic Susceptibility and Treatment (MEGA) trial enrolled 606 men with nongonococcal urethritis from 2007 to 2011. The main end points of the trial were clinical and microbiological cure of urethritis associated with M. genitalium infection, when treated with the standard therapy recommended by the Centers for Disease Control and Prevention: a single dose of 1 g of azithromycin, or a 7-day regimen of 100 mg of doxycycline taken twice a day.
Several pathogens cause nongonococcal urethritis, said Dr. Manhart, an epidemiologist at the University of Washington, Seattle. Among them are Chlamydia trachomatis, M. genitalium, the recently differentiated Ureaplasma urealyticum, and Trichomonas vaginalis. In some infections, no pathogen can be identified. In addition to presenting the end points for M. genitalium infections, Dr. Manhart broke the results out by other pathogens.
The mean age of the men in the trial was 34 years. Most (67%) were heterosexual, 28% were homosexual, and about 5% were bisexual. They reported a mean of six sexual partners in the past 12 months.
The most common infectious pathogen was chlamydia (26%), followed by U. urealyticum (24%). M. genitalium was present in 14% of the study group, and T. vaginalis in 2%. The remainder of the group had an idiopathic urethritis.
Treatment consisted of either a placebo azithromycin and active doxycycline or active azithromycin and placebo doxycycline. If men still showed symptoms or microbiological failure after treatment, they were offered reverse therapy (i.e., a treatment pack with the opposite active and placebo drugs), and returned for a follow-up visit in 3 weeks. If they were still not clinically or microbiologically cured, they received a course of moxifloxacin.
Overall, there was no statistically significant difference in cure rates between the two drugs. Nor were there significant differences among men with chlamydia infections, with a clinical cure rate of 87% for azithromycin and 78% for doxycycline. For microbiological cure, the rates were 86% and 90%, respectively.
Both clinical and microbiological cures rates were much lower in men infected with M. genitalium, but the rates were not significantly different between the two drugs. Clinical cure rates were 68% for azithromycin and 48% for doxycycline. For microbiological cure, the rates were 39% and 30%, respectively.
Men with U. urealyticum infections fared a little better, but again, the cure rates were not significantly different between the drugs. A clinical cure occurred in 85% of the azithromycin group and 75% of the doxycycline group. Microbiological cure rates were 75% and 69%, respectively.
For those with idiopathic urethritis, the cure rate for both drugs was identical: 87%.
Reverse therapy was still not effective in some men with U. urealyticum or M. genitalium infections. Three weeks after that treatment, 51% of those with M. genitalium and 57% of those with U. urealyticum were still positive for the pathogen. These men received a course of moxifloxacin.
"Moxifloxacin was quite effective, but at the end of the trial, we still had two men with M. genitalium and four with U. urealyticum who did not clear their infections," Dr. Manhart said.
Moxifloxacin, although highly effective, is too expensive to be used as first-time therapy in a public health department setting, she noted. "Its preinsurance cost runs up to $150, so we are still faced with a conundrum when treating these infections."
Dr. Manhart had no financial declarations.
FROM A CONFERENCE ON STD PREVENTION SPONSORED BY THE CENTERS FOR DISEASE CONTROL AND PREVENTION
When Diagnosed, Women Help Treat Partners for Chlamydia
MINNEAPOLIS – Researchers at the National STD Prevention Conference agreed: Not enough women are getting tested for chlamydia, and health care providers are missing valuable opportunities to test them.
Among those lost opportunities is the ability to stem the spread of the disease, as new findings show that women who are diagnosed with chlamydia are likely to provide treatment to their partners.
Women with chlamydia infection are usually asymptomatic, and often consult a clinician for some other sexual health concern. Most of these visits provide a perfect opportunity to add a chlamydia screening test, said Dr. Joan Chow, chief of epidemiology in the STD control branch of the California Department of Public Health.
"Annual chlamydia screening has increased during the past 10 years, but still, about 50% of young women who should be screened aren’t being screened," she said at a conference on STD prevention sponsored by the Centers for Disease Control and Prevention.
She and her colleagues examined data from California’s Family PACT program, which provides comprehensive family planning services to low-income women.
More than 1.5 million women are enrolled in the program, which includes more than 2,000 medical providers and uses the services of about 200 labs in the state. Chlamydia testing is part of the PACT program benefits, and is recommended for all sexually active women aged 25 years or younger.
The study period spanned 2008-2010, focusing on women aged 25 or younger who were seen at by Family PACT providers in 2009. Dr. Chow compared the chlamydia testing these women received with any testing they had in the prior 12 months, or in the following 12 months.
During 2009, 219,319 young women accessed PACT services; 74% of those had a chlamydia test in the past 12 months, leaving 25% (55,915) untested at the 2009 visit.
Overall, 89% had urine collected for either a pregnancy test or to evaluate for a urinary tract infection. Of those patients, 43% had other services that included an easy opportunity to test, including a pelvic exam.
Most of these young women (75%) visited a PACT office only once during the study period. Only half (52%) returned to an office in the following 12 months, and of those, only 50% were tested for chlamydia, despite not being tested at the previous visit.
In all, only a quarter of the untested young women were tested in the following 12 months, Dr. Chow said. "Since most had only one visit, opportunities to test were not abundant."
But most patients who are diagnosed do help treat their sexual partners if they have access to the expedited treatment packs some clinics provide.
Typically, the packs contain 1 g of azithromycin, condoms, educational material about chlamydia and other STDs, and contact information for all local STD clinics, said Meighan Rogers of the New York City Department of Health’s STD branch.
She and her colleagues conducted a follow-up study of 671 patients diagnosed with chlamydia during a 12-month period from February 2011 through February 2012. Patients could receive up to three partner packs at the visit, with three additional packs available by prescription. The analysis included only patients from heterosexual couples.
A health department representative called each patient 5 days after treatment to determine if the pack had been given to partners, and if the partners had taken the medication. The survey also asked about any adverse effects of the antibiotic and ascertained the written materials’ clarity.
Most of the patients (74%) were women. Of all those eligible for the patient packs, 52% accepted them. Surveyors were able to contact 174 of these patients; 122 of those reported having a total of 150 partners who were locatable. About 44% of these (66) were successfully contacted.
Most (74%) of the partners reported getting the packs; 94% of those said they took the medication. Two partners reported having medical problems from the medication, although the analysis did not specify what those were.
About a quarter of the partners (23%) said they visited a physician afterward. Of those who did not see a doctor, 100% said they planned to. Women partners were significantly more likely to plan a doctor’s visit than men (46% vs. 13%).
That’s good news, said Ms. Rogers. "It’s very hopeful that almost half of the women were planning to see a physician, since this might translate into a decreased risk of pelvic inflammatory disease and its reproductive sequelae."
As state and federal employees, the presenters had no financial disclosures.
MINNEAPOLIS – Researchers at the National STD Prevention Conference agreed: Not enough women are getting tested for chlamydia, and health care providers are missing valuable opportunities to test them.
Among those lost opportunities is the ability to stem the spread of the disease, as new findings show that women who are diagnosed with chlamydia are likely to provide treatment to their partners.
Women with chlamydia infection are usually asymptomatic, and often consult a clinician for some other sexual health concern. Most of these visits provide a perfect opportunity to add a chlamydia screening test, said Dr. Joan Chow, chief of epidemiology in the STD control branch of the California Department of Public Health.
"Annual chlamydia screening has increased during the past 10 years, but still, about 50% of young women who should be screened aren’t being screened," she said at a conference on STD prevention sponsored by the Centers for Disease Control and Prevention.
She and her colleagues examined data from California’s Family PACT program, which provides comprehensive family planning services to low-income women.
More than 1.5 million women are enrolled in the program, which includes more than 2,000 medical providers and uses the services of about 200 labs in the state. Chlamydia testing is part of the PACT program benefits, and is recommended for all sexually active women aged 25 years or younger.
The study period spanned 2008-2010, focusing on women aged 25 or younger who were seen at by Family PACT providers in 2009. Dr. Chow compared the chlamydia testing these women received with any testing they had in the prior 12 months, or in the following 12 months.
During 2009, 219,319 young women accessed PACT services; 74% of those had a chlamydia test in the past 12 months, leaving 25% (55,915) untested at the 2009 visit.
Overall, 89% had urine collected for either a pregnancy test or to evaluate for a urinary tract infection. Of those patients, 43% had other services that included an easy opportunity to test, including a pelvic exam.
Most of these young women (75%) visited a PACT office only once during the study period. Only half (52%) returned to an office in the following 12 months, and of those, only 50% were tested for chlamydia, despite not being tested at the previous visit.
In all, only a quarter of the untested young women were tested in the following 12 months, Dr. Chow said. "Since most had only one visit, opportunities to test were not abundant."
But most patients who are diagnosed do help treat their sexual partners if they have access to the expedited treatment packs some clinics provide.
Typically, the packs contain 1 g of azithromycin, condoms, educational material about chlamydia and other STDs, and contact information for all local STD clinics, said Meighan Rogers of the New York City Department of Health’s STD branch.
She and her colleagues conducted a follow-up study of 671 patients diagnosed with chlamydia during a 12-month period from February 2011 through February 2012. Patients could receive up to three partner packs at the visit, with three additional packs available by prescription. The analysis included only patients from heterosexual couples.
A health department representative called each patient 5 days after treatment to determine if the pack had been given to partners, and if the partners had taken the medication. The survey also asked about any adverse effects of the antibiotic and ascertained the written materials’ clarity.
Most of the patients (74%) were women. Of all those eligible for the patient packs, 52% accepted them. Surveyors were able to contact 174 of these patients; 122 of those reported having a total of 150 partners who were locatable. About 44% of these (66) were successfully contacted.
Most (74%) of the partners reported getting the packs; 94% of those said they took the medication. Two partners reported having medical problems from the medication, although the analysis did not specify what those were.
About a quarter of the partners (23%) said they visited a physician afterward. Of those who did not see a doctor, 100% said they planned to. Women partners were significantly more likely to plan a doctor’s visit than men (46% vs. 13%).
That’s good news, said Ms. Rogers. "It’s very hopeful that almost half of the women were planning to see a physician, since this might translate into a decreased risk of pelvic inflammatory disease and its reproductive sequelae."
As state and federal employees, the presenters had no financial disclosures.
MINNEAPOLIS – Researchers at the National STD Prevention Conference agreed: Not enough women are getting tested for chlamydia, and health care providers are missing valuable opportunities to test them.
Among those lost opportunities is the ability to stem the spread of the disease, as new findings show that women who are diagnosed with chlamydia are likely to provide treatment to their partners.
Women with chlamydia infection are usually asymptomatic, and often consult a clinician for some other sexual health concern. Most of these visits provide a perfect opportunity to add a chlamydia screening test, said Dr. Joan Chow, chief of epidemiology in the STD control branch of the California Department of Public Health.
"Annual chlamydia screening has increased during the past 10 years, but still, about 50% of young women who should be screened aren’t being screened," she said at a conference on STD prevention sponsored by the Centers for Disease Control and Prevention.
She and her colleagues examined data from California’s Family PACT program, which provides comprehensive family planning services to low-income women.
More than 1.5 million women are enrolled in the program, which includes more than 2,000 medical providers and uses the services of about 200 labs in the state. Chlamydia testing is part of the PACT program benefits, and is recommended for all sexually active women aged 25 years or younger.
The study period spanned 2008-2010, focusing on women aged 25 or younger who were seen at by Family PACT providers in 2009. Dr. Chow compared the chlamydia testing these women received with any testing they had in the prior 12 months, or in the following 12 months.
During 2009, 219,319 young women accessed PACT services; 74% of those had a chlamydia test in the past 12 months, leaving 25% (55,915) untested at the 2009 visit.
Overall, 89% had urine collected for either a pregnancy test or to evaluate for a urinary tract infection. Of those patients, 43% had other services that included an easy opportunity to test, including a pelvic exam.
Most of these young women (75%) visited a PACT office only once during the study period. Only half (52%) returned to an office in the following 12 months, and of those, only 50% were tested for chlamydia, despite not being tested at the previous visit.
In all, only a quarter of the untested young women were tested in the following 12 months, Dr. Chow said. "Since most had only one visit, opportunities to test were not abundant."
But most patients who are diagnosed do help treat their sexual partners if they have access to the expedited treatment packs some clinics provide.
Typically, the packs contain 1 g of azithromycin, condoms, educational material about chlamydia and other STDs, and contact information for all local STD clinics, said Meighan Rogers of the New York City Department of Health’s STD branch.
She and her colleagues conducted a follow-up study of 671 patients diagnosed with chlamydia during a 12-month period from February 2011 through February 2012. Patients could receive up to three partner packs at the visit, with three additional packs available by prescription. The analysis included only patients from heterosexual couples.
A health department representative called each patient 5 days after treatment to determine if the pack had been given to partners, and if the partners had taken the medication. The survey also asked about any adverse effects of the antibiotic and ascertained the written materials’ clarity.
Most of the patients (74%) were women. Of all those eligible for the patient packs, 52% accepted them. Surveyors were able to contact 174 of these patients; 122 of those reported having a total of 150 partners who were locatable. About 44% of these (66) were successfully contacted.
Most (74%) of the partners reported getting the packs; 94% of those said they took the medication. Two partners reported having medical problems from the medication, although the analysis did not specify what those were.
About a quarter of the partners (23%) said they visited a physician afterward. Of those who did not see a doctor, 100% said they planned to. Women partners were significantly more likely to plan a doctor’s visit than men (46% vs. 13%).
That’s good news, said Ms. Rogers. "It’s very hopeful that almost half of the women were planning to see a physician, since this might translate into a decreased risk of pelvic inflammatory disease and its reproductive sequelae."
As state and federal employees, the presenters had no financial disclosures.
FROM A CONFERENCE ON STD PREVENTION SPONSORED BY THE CENTERS FOR DISEASE CONTROL AND PREVENTION
Embolectomy May Help Despite Infarct Size
NEW ORLEANS – Improving the outcomes of endovascular stroke intervention lies – at least in part – in selecting the best candidates.
But even those who are at the highest risk of harm from the treatment still may reap some benefit when compared with the grim alternative, researchers said at the International Stroke Conference, which was sponsored by the American Heart Association.
The oldest patients and those with large ischemic infarcts are at exceedingly high risk of very poor outcomes with mechanical embolectomy, according to Dr. Albert Yoo and Dr. Yogesh Moradiya. But their analyses of stroke study cohorts indicate that clinicians might be able to predict and screen for patients who will benefit most from endovascular stroke treatment.
Patients with the lowest scores on the ASPECTS (Alberta Stroke Program Early CT Score) assessment are known to have "dismal outcomes, and should probably be excluded from this therapy," said Dr. Yoo, a vascular radiologist at Massachusetts General Hospital, Boston. But, he said, his analysis identified a subgroup of patients he now believes should be treated, despite their prior reputation as barely salvageable.
"If you take the wrong person, and the procedure goes bad and they die on the table, this is not helping the brain."
In the prediction of outcome by ASPECTS, patients with ischemic stroke are grouped by the size of their initial infarct, as seen on noncontrast CT scan. The 10-point score has conventionally dichotomized patients into those destined to have poor outcomes (ASPECTS 0-7) and those with much better outcomes (APSECTS 8-10).
Dr. Yoo examined outcomes in two stroke study cohorts that used ASPECTS as a predictor of outcome. The pooled analysis included 215 patients with a mean age of 66 years. Their mean National Institutes of Health Stroke Scale score was 17. Most of the strokes (77%) were in the middle cerebral artery.
The pretreatment ASPECTS score was 0-4 in 16% of patients, 5-7 in 34%, and 8-10 in the remainder. The median ASPECTS score was 7, which is the widely accepted cutoff level for using mechanical embolectomy with a reasonable hope of good outcome.
The procedure was successful in most patients, with 83% achieving a TIMI (thrombolysis in myocardial infarction) score of 2 or 3. Most of the reperfusions occurred in a timely manner as well (within 6 hours for 27% and shortly thereafter for the rest).
Dr. Yoo said that, generally, those with the lowest scores on ASPECTS fared significantly worse, as expected. In the conventional split, 24% of those with scores of 0-7 had a good 90-day outcome (defined as discharge home with self-care), compared with 44% of those with scores of 8-10. Time to reperfusion did not affect outcomes in the lower-scoring group. The patients who reperfused early had almost exactly the same poor outcome as did those who reperfused late and those who never received the intervention.
But when Dr. Yoo split the scores of the cohorts into three groups, he found that the vast majority of poor outcomes occurred in the 0- to 4-point range. Among these patients, only 4% had good outcomes, and no one in the 0- to 3-point group had a good outcome.
Patients who had a score of 5, 6, or 7 points on ASPECTS, however, fared significantly better, with good outcomes in about 20%, 30%, and 40%, respectively. Time to reperfusion was important in these patients: Outcomes were good in 53% of early reperfusions, 31% of late reperfusions, and 20% of those who were not reperfused.
The results for patients with 8-10 points on ASPECTS – generally considered the group most likely to benefit – were similar to those seen in the 5- to 7-point group. Good outcomes occurred in 54% of those in the 8- to 10-point group with early perfusion. The rates were 46% in those with later reperfusions and 17% in those with no reperfusion.
When Dr. Yoo combined patients with 5-7 points and 8-10 points on ASPECTS, the outcomes still looked positive. "Almost all [85%] of the patients in these groups who achieved a TIMI 2-3 reperfusion had good outcomes," he said. This was a threefold increase over similar patients with a TIMI score of 0-1.
"The ASPECTS score does identify a group of patients highly likely to have dismal outcomes who probably will not benefit from reperfusion therapy and should be excluded from it," he said. "But patients with ASPECTS scores of 5-7 do appear to benefit, and treatment should not be withheld from this group."
Age as the determining factor of who should undergo reperfusion therapy proved a tougher sell for Dr. Yogesh Moradiya, a neurology fellow at the SUNY Downstate Medical Center, Brooklyn, N.Y.
He examined the relationship between advanced age and functional outcome after ischemic stroke treatment in a sample of 6,700 patients in the Nationwide Inpatient Sample. Of those patients, 20% were older than 80 years.
"Not surprisingly, most [68%] of these elderly old were female," said Dr. Moradiya. Those older than 80 years also had more comorbidity, leading to a higher Elixhauser comorbidity index than that in young patients (mean, 3.4 vs. 1.6).
Mortality was significantly higher in the older patients than the younger patients (31% vs. 22%), although there were no differences between the age groups in the rates of symptomatic and asymptomatic intracranial hemorrhage, mechanical ventilation, and tracheostomy.
The older patients were more likely to receive intravenous or intra-arterial thrombolysis in addition to embolectomy, probably because of an unsuccessful initial endovascular procedure. But the octogenarians did not withstand their treatment and hospitalization very well. Compared with younger patients, the older patients had significantly higher rates of heart attack (7% vs. 5%), acute kidney injury (9% vs. 6%), gastrointestinal bleeding (3% vs. 2%), urinary tract infections (22% vs. 15%), and transfusions (10% vs. 8%).
Patients older than 80 years had several comorbidities that significantly predicted mortality after ischemic stroke, including the following:
• Chronic pulmonary disease (odds ratio, 2.7).
• Acute kidney injury (OR, 3.8).
• Myocardial infarction (OR, 3.2).
• Intracranial hemorrhage (OR, 3).
• Sepsis (OR, 8).
Thrombolytic treatment lowered the risk of death by 68% in older patients, but this was not significantly different from the treatment effect in younger patients. Dr. Moradiya suggested that selecting younger or healthier patients would improve endovascular stroke treatment outcomes. "This is screening in the sense that people with this higher comorbidity profile should not undergo embolectomy, compared to the more healthy elderly."
This summation rankled some audience members, including Dr. James L. Frey, director of the stroke center at Barrow Neurological Institute, Phoenix.
"I don’t like this concept of reducing complications by screening people for these procedures," he said during the discussion. "It seems obvious that the medical complications are the problem, and not the procedure itself, so when you talk about screening, how are you going to do that?"
Very elderly stroke patients want treatment just as much as younger patients do, he said. "They don’t like what’s going wrong with their brain and they want someone to do something about it. They don’t mind so much if you try this and they die, but they do not want to live with a brain hemorrhage. It sounds like you’re saying they are better off having a clot in their middle cerebral artery than having someone try to remove it. Is this what you’re saying?"
Dr. Felipe Albuquerque, a session moderator, added his take on the issue.
"Absolutely, the brain is paramount and all other organs are secondary, so we should do all we can for the brain. But if you take the wrong person, and the procedure goes bad and they die on the table, this is not helping the brain. There are simply patients who are too high risk, and for these you might think about a different procedure."
"I don’t do these procedures with anesthesia on board," because of the extra burden it places on an elderly patient, said Dr. Albuquerque, a neurosurgeon who is also with the Barrow Neurological Institute.
"So in that sense, screening does help your approach and should perhaps obviate intervention for people who are going to die, no matter what you do."
Dr. Moradiya had no financial disclosures. Dr. Yoo said that he has received research funding from Penumbra Inc. Dr. Frey and Dr. Albuquerque said they had no relevant disclosures.
NEW ORLEANS – Improving the outcomes of endovascular stroke intervention lies – at least in part – in selecting the best candidates.
But even those who are at the highest risk of harm from the treatment still may reap some benefit when compared with the grim alternative, researchers said at the International Stroke Conference, which was sponsored by the American Heart Association.
The oldest patients and those with large ischemic infarcts are at exceedingly high risk of very poor outcomes with mechanical embolectomy, according to Dr. Albert Yoo and Dr. Yogesh Moradiya. But their analyses of stroke study cohorts indicate that clinicians might be able to predict and screen for patients who will benefit most from endovascular stroke treatment.
Patients with the lowest scores on the ASPECTS (Alberta Stroke Program Early CT Score) assessment are known to have "dismal outcomes, and should probably be excluded from this therapy," said Dr. Yoo, a vascular radiologist at Massachusetts General Hospital, Boston. But, he said, his analysis identified a subgroup of patients he now believes should be treated, despite their prior reputation as barely salvageable.
"If you take the wrong person, and the procedure goes bad and they die on the table, this is not helping the brain."
In the prediction of outcome by ASPECTS, patients with ischemic stroke are grouped by the size of their initial infarct, as seen on noncontrast CT scan. The 10-point score has conventionally dichotomized patients into those destined to have poor outcomes (ASPECTS 0-7) and those with much better outcomes (APSECTS 8-10).
Dr. Yoo examined outcomes in two stroke study cohorts that used ASPECTS as a predictor of outcome. The pooled analysis included 215 patients with a mean age of 66 years. Their mean National Institutes of Health Stroke Scale score was 17. Most of the strokes (77%) were in the middle cerebral artery.
The pretreatment ASPECTS score was 0-4 in 16% of patients, 5-7 in 34%, and 8-10 in the remainder. The median ASPECTS score was 7, which is the widely accepted cutoff level for using mechanical embolectomy with a reasonable hope of good outcome.
The procedure was successful in most patients, with 83% achieving a TIMI (thrombolysis in myocardial infarction) score of 2 or 3. Most of the reperfusions occurred in a timely manner as well (within 6 hours for 27% and shortly thereafter for the rest).
Dr. Yoo said that, generally, those with the lowest scores on ASPECTS fared significantly worse, as expected. In the conventional split, 24% of those with scores of 0-7 had a good 90-day outcome (defined as discharge home with self-care), compared with 44% of those with scores of 8-10. Time to reperfusion did not affect outcomes in the lower-scoring group. The patients who reperfused early had almost exactly the same poor outcome as did those who reperfused late and those who never received the intervention.
But when Dr. Yoo split the scores of the cohorts into three groups, he found that the vast majority of poor outcomes occurred in the 0- to 4-point range. Among these patients, only 4% had good outcomes, and no one in the 0- to 3-point group had a good outcome.
Patients who had a score of 5, 6, or 7 points on ASPECTS, however, fared significantly better, with good outcomes in about 20%, 30%, and 40%, respectively. Time to reperfusion was important in these patients: Outcomes were good in 53% of early reperfusions, 31% of late reperfusions, and 20% of those who were not reperfused.
The results for patients with 8-10 points on ASPECTS – generally considered the group most likely to benefit – were similar to those seen in the 5- to 7-point group. Good outcomes occurred in 54% of those in the 8- to 10-point group with early perfusion. The rates were 46% in those with later reperfusions and 17% in those with no reperfusion.
When Dr. Yoo combined patients with 5-7 points and 8-10 points on ASPECTS, the outcomes still looked positive. "Almost all [85%] of the patients in these groups who achieved a TIMI 2-3 reperfusion had good outcomes," he said. This was a threefold increase over similar patients with a TIMI score of 0-1.
"The ASPECTS score does identify a group of patients highly likely to have dismal outcomes who probably will not benefit from reperfusion therapy and should be excluded from it," he said. "But patients with ASPECTS scores of 5-7 do appear to benefit, and treatment should not be withheld from this group."
Age as the determining factor of who should undergo reperfusion therapy proved a tougher sell for Dr. Yogesh Moradiya, a neurology fellow at the SUNY Downstate Medical Center, Brooklyn, N.Y.
He examined the relationship between advanced age and functional outcome after ischemic stroke treatment in a sample of 6,700 patients in the Nationwide Inpatient Sample. Of those patients, 20% were older than 80 years.
"Not surprisingly, most [68%] of these elderly old were female," said Dr. Moradiya. Those older than 80 years also had more comorbidity, leading to a higher Elixhauser comorbidity index than that in young patients (mean, 3.4 vs. 1.6).
Mortality was significantly higher in the older patients than the younger patients (31% vs. 22%), although there were no differences between the age groups in the rates of symptomatic and asymptomatic intracranial hemorrhage, mechanical ventilation, and tracheostomy.
The older patients were more likely to receive intravenous or intra-arterial thrombolysis in addition to embolectomy, probably because of an unsuccessful initial endovascular procedure. But the octogenarians did not withstand their treatment and hospitalization very well. Compared with younger patients, the older patients had significantly higher rates of heart attack (7% vs. 5%), acute kidney injury (9% vs. 6%), gastrointestinal bleeding (3% vs. 2%), urinary tract infections (22% vs. 15%), and transfusions (10% vs. 8%).
Patients older than 80 years had several comorbidities that significantly predicted mortality after ischemic stroke, including the following:
• Chronic pulmonary disease (odds ratio, 2.7).
• Acute kidney injury (OR, 3.8).
• Myocardial infarction (OR, 3.2).
• Intracranial hemorrhage (OR, 3).
• Sepsis (OR, 8).
Thrombolytic treatment lowered the risk of death by 68% in older patients, but this was not significantly different from the treatment effect in younger patients. Dr. Moradiya suggested that selecting younger or healthier patients would improve endovascular stroke treatment outcomes. "This is screening in the sense that people with this higher comorbidity profile should not undergo embolectomy, compared to the more healthy elderly."
This summation rankled some audience members, including Dr. James L. Frey, director of the stroke center at Barrow Neurological Institute, Phoenix.
"I don’t like this concept of reducing complications by screening people for these procedures," he said during the discussion. "It seems obvious that the medical complications are the problem, and not the procedure itself, so when you talk about screening, how are you going to do that?"
Very elderly stroke patients want treatment just as much as younger patients do, he said. "They don’t like what’s going wrong with their brain and they want someone to do something about it. They don’t mind so much if you try this and they die, but they do not want to live with a brain hemorrhage. It sounds like you’re saying they are better off having a clot in their middle cerebral artery than having someone try to remove it. Is this what you’re saying?"
Dr. Felipe Albuquerque, a session moderator, added his take on the issue.
"Absolutely, the brain is paramount and all other organs are secondary, so we should do all we can for the brain. But if you take the wrong person, and the procedure goes bad and they die on the table, this is not helping the brain. There are simply patients who are too high risk, and for these you might think about a different procedure."
"I don’t do these procedures with anesthesia on board," because of the extra burden it places on an elderly patient, said Dr. Albuquerque, a neurosurgeon who is also with the Barrow Neurological Institute.
"So in that sense, screening does help your approach and should perhaps obviate intervention for people who are going to die, no matter what you do."
Dr. Moradiya had no financial disclosures. Dr. Yoo said that he has received research funding from Penumbra Inc. Dr. Frey and Dr. Albuquerque said they had no relevant disclosures.
NEW ORLEANS – Improving the outcomes of endovascular stroke intervention lies – at least in part – in selecting the best candidates.
But even those who are at the highest risk of harm from the treatment still may reap some benefit when compared with the grim alternative, researchers said at the International Stroke Conference, which was sponsored by the American Heart Association.
The oldest patients and those with large ischemic infarcts are at exceedingly high risk of very poor outcomes with mechanical embolectomy, according to Dr. Albert Yoo and Dr. Yogesh Moradiya. But their analyses of stroke study cohorts indicate that clinicians might be able to predict and screen for patients who will benefit most from endovascular stroke treatment.
Patients with the lowest scores on the ASPECTS (Alberta Stroke Program Early CT Score) assessment are known to have "dismal outcomes, and should probably be excluded from this therapy," said Dr. Yoo, a vascular radiologist at Massachusetts General Hospital, Boston. But, he said, his analysis identified a subgroup of patients he now believes should be treated, despite their prior reputation as barely salvageable.
"If you take the wrong person, and the procedure goes bad and they die on the table, this is not helping the brain."
In the prediction of outcome by ASPECTS, patients with ischemic stroke are grouped by the size of their initial infarct, as seen on noncontrast CT scan. The 10-point score has conventionally dichotomized patients into those destined to have poor outcomes (ASPECTS 0-7) and those with much better outcomes (APSECTS 8-10).
Dr. Yoo examined outcomes in two stroke study cohorts that used ASPECTS as a predictor of outcome. The pooled analysis included 215 patients with a mean age of 66 years. Their mean National Institutes of Health Stroke Scale score was 17. Most of the strokes (77%) were in the middle cerebral artery.
The pretreatment ASPECTS score was 0-4 in 16% of patients, 5-7 in 34%, and 8-10 in the remainder. The median ASPECTS score was 7, which is the widely accepted cutoff level for using mechanical embolectomy with a reasonable hope of good outcome.
The procedure was successful in most patients, with 83% achieving a TIMI (thrombolysis in myocardial infarction) score of 2 or 3. Most of the reperfusions occurred in a timely manner as well (within 6 hours for 27% and shortly thereafter for the rest).
Dr. Yoo said that, generally, those with the lowest scores on ASPECTS fared significantly worse, as expected. In the conventional split, 24% of those with scores of 0-7 had a good 90-day outcome (defined as discharge home with self-care), compared with 44% of those with scores of 8-10. Time to reperfusion did not affect outcomes in the lower-scoring group. The patients who reperfused early had almost exactly the same poor outcome as did those who reperfused late and those who never received the intervention.
But when Dr. Yoo split the scores of the cohorts into three groups, he found that the vast majority of poor outcomes occurred in the 0- to 4-point range. Among these patients, only 4% had good outcomes, and no one in the 0- to 3-point group had a good outcome.
Patients who had a score of 5, 6, or 7 points on ASPECTS, however, fared significantly better, with good outcomes in about 20%, 30%, and 40%, respectively. Time to reperfusion was important in these patients: Outcomes were good in 53% of early reperfusions, 31% of late reperfusions, and 20% of those who were not reperfused.
The results for patients with 8-10 points on ASPECTS – generally considered the group most likely to benefit – were similar to those seen in the 5- to 7-point group. Good outcomes occurred in 54% of those in the 8- to 10-point group with early perfusion. The rates were 46% in those with later reperfusions and 17% in those with no reperfusion.
When Dr. Yoo combined patients with 5-7 points and 8-10 points on ASPECTS, the outcomes still looked positive. "Almost all [85%] of the patients in these groups who achieved a TIMI 2-3 reperfusion had good outcomes," he said. This was a threefold increase over similar patients with a TIMI score of 0-1.
"The ASPECTS score does identify a group of patients highly likely to have dismal outcomes who probably will not benefit from reperfusion therapy and should be excluded from it," he said. "But patients with ASPECTS scores of 5-7 do appear to benefit, and treatment should not be withheld from this group."
Age as the determining factor of who should undergo reperfusion therapy proved a tougher sell for Dr. Yogesh Moradiya, a neurology fellow at the SUNY Downstate Medical Center, Brooklyn, N.Y.
He examined the relationship between advanced age and functional outcome after ischemic stroke treatment in a sample of 6,700 patients in the Nationwide Inpatient Sample. Of those patients, 20% were older than 80 years.
"Not surprisingly, most [68%] of these elderly old were female," said Dr. Moradiya. Those older than 80 years also had more comorbidity, leading to a higher Elixhauser comorbidity index than that in young patients (mean, 3.4 vs. 1.6).
Mortality was significantly higher in the older patients than the younger patients (31% vs. 22%), although there were no differences between the age groups in the rates of symptomatic and asymptomatic intracranial hemorrhage, mechanical ventilation, and tracheostomy.
The older patients were more likely to receive intravenous or intra-arterial thrombolysis in addition to embolectomy, probably because of an unsuccessful initial endovascular procedure. But the octogenarians did not withstand their treatment and hospitalization very well. Compared with younger patients, the older patients had significantly higher rates of heart attack (7% vs. 5%), acute kidney injury (9% vs. 6%), gastrointestinal bleeding (3% vs. 2%), urinary tract infections (22% vs. 15%), and transfusions (10% vs. 8%).
Patients older than 80 years had several comorbidities that significantly predicted mortality after ischemic stroke, including the following:
• Chronic pulmonary disease (odds ratio, 2.7).
• Acute kidney injury (OR, 3.8).
• Myocardial infarction (OR, 3.2).
• Intracranial hemorrhage (OR, 3).
• Sepsis (OR, 8).
Thrombolytic treatment lowered the risk of death by 68% in older patients, but this was not significantly different from the treatment effect in younger patients. Dr. Moradiya suggested that selecting younger or healthier patients would improve endovascular stroke treatment outcomes. "This is screening in the sense that people with this higher comorbidity profile should not undergo embolectomy, compared to the more healthy elderly."
This summation rankled some audience members, including Dr. James L. Frey, director of the stroke center at Barrow Neurological Institute, Phoenix.
"I don’t like this concept of reducing complications by screening people for these procedures," he said during the discussion. "It seems obvious that the medical complications are the problem, and not the procedure itself, so when you talk about screening, how are you going to do that?"
Very elderly stroke patients want treatment just as much as younger patients do, he said. "They don’t like what’s going wrong with their brain and they want someone to do something about it. They don’t mind so much if you try this and they die, but they do not want to live with a brain hemorrhage. It sounds like you’re saying they are better off having a clot in their middle cerebral artery than having someone try to remove it. Is this what you’re saying?"
Dr. Felipe Albuquerque, a session moderator, added his take on the issue.
"Absolutely, the brain is paramount and all other organs are secondary, so we should do all we can for the brain. But if you take the wrong person, and the procedure goes bad and they die on the table, this is not helping the brain. There are simply patients who are too high risk, and for these you might think about a different procedure."
"I don’t do these procedures with anesthesia on board," because of the extra burden it places on an elderly patient, said Dr. Albuquerque, a neurosurgeon who is also with the Barrow Neurological Institute.
"So in that sense, screening does help your approach and should perhaps obviate intervention for people who are going to die, no matter what you do."
Dr. Moradiya had no financial disclosures. Dr. Yoo said that he has received research funding from Penumbra Inc. Dr. Frey and Dr. Albuquerque said they had no relevant disclosures.
FROM THE INTERNATIONAL STROKE CONFERENCE
Major Finding: Embolectomy could help up to 70% of patients whose ischemic stroke infarct size may have previously disqualified them from treatment.
Data Source: Two database studies identified additional patients who could benefit from mechanical embolectomy.
Disclosures: Dr. Moradiya had no financial disclosures. Dr. Yoo said that he has received research funding from Penumbra. Dr. Frey and Dr. Albuquerque said they had no relevant disclosures.
Targeted Support Better for Those With Schizophrenia
Older adults with schizophrenia got more – and better – jobs using an individualized employment support program than with conventional job counseling, a study has shown.
Within a year after beginning each program, more than half of those in the individualized program had worked, compared with less than a third of those who received the typical counseling, Elizabeth W. Twamley, Ph.D., and her colleagues wrote in the March issue of Schizophrenia Research (2012;135:100-4).
The 12-month study randomized 58 middle-aged and older adults with schizophrenia to the programs. The individualized placement and support (IPS) program used counselors with a caseload of no more than 25 clients. Caseworkers made an effort to integrate employment counseling with psychiatric treatment. The program also emphasized obtaining competitive work (that is, jobs not reserved for disabled workers) and offered all clients unlimited support during their job search and subsequent employment.
Caseworkers in the conventional vocational rehabilitation (CVR) arm had 35 clients. The study staff provided extra support with job-readiness and prevocational coaching classes and with making appointments. For the three job appointments, the staff provided reminders and transportation.
The clients’ mean age was 51 years; their average duration of illness was 24 years. About half had a diagnosis of schizoaffective disorder; the remainder had schizophrenia. Most (79%) had been without paid work for 2 years, although 86% had at one time held the same job for at least 12 months.
The study had a 21% attrition rate (12 clients). These participants were significantly less likely to be taking atypical antipsychotics (42% vs. 80%), were more likely to have a diagnosis of schizophrenia (67% vs. 33%), and had been unemployed for a significantly longer period (13 vs. 5 years).
Those involved in the IPS program had significantly better employment outcomes by the end of 12 months. More than half (57%) held competitive jobs, compared with 29% in the conventional arm. Most of the IPS clients (70%) obtained any kind of paid work, whether competitive or reserved, compared with 36% of CVR clients.
Those in the IPS group also worked for a significantly longer period (mean, 12 weeks, vs. 5) and earned significantly more money (mean, $2,047, vs. $607).
"The [number needed to treat] for competitive work (3.56) indicated that providing IPS to about three people would result in one more competitive job than would CVR," wrote Dr. Twamley of the University of California, San Diego, and her coauthors.
Several factors significantly affected employment outcomes. In the IPS group, longer prior unemployment was associated with a lower chance of obtaining employment and with a lower income when working. The higher the baseline functional measures, the better the chance of competitive employment and higher earnings.
In the conventional group, higher depression scores on the Hamilton Rating Scale for Depression predicted worse employment outcomes, "perhaps due to lack of integration between CVR and psychiatric treatment."
The National Institute of Mental Health and the National Alliance for Research of Schizophrenia and Depression funded the study. None of the authors reported any relevant financial conflicts.
Older adults with schizophrenia got more – and better – jobs using an individualized employment support program than with conventional job counseling, a study has shown.
Within a year after beginning each program, more than half of those in the individualized program had worked, compared with less than a third of those who received the typical counseling, Elizabeth W. Twamley, Ph.D., and her colleagues wrote in the March issue of Schizophrenia Research (2012;135:100-4).
The 12-month study randomized 58 middle-aged and older adults with schizophrenia to the programs. The individualized placement and support (IPS) program used counselors with a caseload of no more than 25 clients. Caseworkers made an effort to integrate employment counseling with psychiatric treatment. The program also emphasized obtaining competitive work (that is, jobs not reserved for disabled workers) and offered all clients unlimited support during their job search and subsequent employment.
Caseworkers in the conventional vocational rehabilitation (CVR) arm had 35 clients. The study staff provided extra support with job-readiness and prevocational coaching classes and with making appointments. For the three job appointments, the staff provided reminders and transportation.
The clients’ mean age was 51 years; their average duration of illness was 24 years. About half had a diagnosis of schizoaffective disorder; the remainder had schizophrenia. Most (79%) had been without paid work for 2 years, although 86% had at one time held the same job for at least 12 months.
The study had a 21% attrition rate (12 clients). These participants were significantly less likely to be taking atypical antipsychotics (42% vs. 80%), were more likely to have a diagnosis of schizophrenia (67% vs. 33%), and had been unemployed for a significantly longer period (13 vs. 5 years).
Those involved in the IPS program had significantly better employment outcomes by the end of 12 months. More than half (57%) held competitive jobs, compared with 29% in the conventional arm. Most of the IPS clients (70%) obtained any kind of paid work, whether competitive or reserved, compared with 36% of CVR clients.
Those in the IPS group also worked for a significantly longer period (mean, 12 weeks, vs. 5) and earned significantly more money (mean, $2,047, vs. $607).
"The [number needed to treat] for competitive work (3.56) indicated that providing IPS to about three people would result in one more competitive job than would CVR," wrote Dr. Twamley of the University of California, San Diego, and her coauthors.
Several factors significantly affected employment outcomes. In the IPS group, longer prior unemployment was associated with a lower chance of obtaining employment and with a lower income when working. The higher the baseline functional measures, the better the chance of competitive employment and higher earnings.
In the conventional group, higher depression scores on the Hamilton Rating Scale for Depression predicted worse employment outcomes, "perhaps due to lack of integration between CVR and psychiatric treatment."
The National Institute of Mental Health and the National Alliance for Research of Schizophrenia and Depression funded the study. None of the authors reported any relevant financial conflicts.
Older adults with schizophrenia got more – and better – jobs using an individualized employment support program than with conventional job counseling, a study has shown.
Within a year after beginning each program, more than half of those in the individualized program had worked, compared with less than a third of those who received the typical counseling, Elizabeth W. Twamley, Ph.D., and her colleagues wrote in the March issue of Schizophrenia Research (2012;135:100-4).
The 12-month study randomized 58 middle-aged and older adults with schizophrenia to the programs. The individualized placement and support (IPS) program used counselors with a caseload of no more than 25 clients. Caseworkers made an effort to integrate employment counseling with psychiatric treatment. The program also emphasized obtaining competitive work (that is, jobs not reserved for disabled workers) and offered all clients unlimited support during their job search and subsequent employment.
Caseworkers in the conventional vocational rehabilitation (CVR) arm had 35 clients. The study staff provided extra support with job-readiness and prevocational coaching classes and with making appointments. For the three job appointments, the staff provided reminders and transportation.
The clients’ mean age was 51 years; their average duration of illness was 24 years. About half had a diagnosis of schizoaffective disorder; the remainder had schizophrenia. Most (79%) had been without paid work for 2 years, although 86% had at one time held the same job for at least 12 months.
The study had a 21% attrition rate (12 clients). These participants were significantly less likely to be taking atypical antipsychotics (42% vs. 80%), were more likely to have a diagnosis of schizophrenia (67% vs. 33%), and had been unemployed for a significantly longer period (13 vs. 5 years).
Those involved in the IPS program had significantly better employment outcomes by the end of 12 months. More than half (57%) held competitive jobs, compared with 29% in the conventional arm. Most of the IPS clients (70%) obtained any kind of paid work, whether competitive or reserved, compared with 36% of CVR clients.
Those in the IPS group also worked for a significantly longer period (mean, 12 weeks, vs. 5) and earned significantly more money (mean, $2,047, vs. $607).
"The [number needed to treat] for competitive work (3.56) indicated that providing IPS to about three people would result in one more competitive job than would CVR," wrote Dr. Twamley of the University of California, San Diego, and her coauthors.
Several factors significantly affected employment outcomes. In the IPS group, longer prior unemployment was associated with a lower chance of obtaining employment and with a lower income when working. The higher the baseline functional measures, the better the chance of competitive employment and higher earnings.
In the conventional group, higher depression scores on the Hamilton Rating Scale for Depression predicted worse employment outcomes, "perhaps due to lack of integration between CVR and psychiatric treatment."
The National Institute of Mental Health and the National Alliance for Research of Schizophrenia and Depression funded the study. None of the authors reported any relevant financial conflicts.
FROM SCHIZOPHRENIA RESEARCH
Major Finding: An individualized employment counseling program for clients with schizophrenia helped 57% obtain competitive employment – significantly more than the 29% who got jobs while enrolled in a conventional program.
Data Source: The study randomized 58 people with schizophrenia or schizoaffective disorder to either the individualized or the conventional programs.
Disclosures: The National Institute of Mental Health and the National Alliance for Research of Schizophrenia and Depression funded the study. None of the authors reported any relevant financial conflicts.
Toddlers' Autistic Behaviors Linked to Preteen Psychosis
Autistic-like behaviors in toddlers might predict psychotic events in adolescence, a retrospective study has determined.
Speech problems and ritualistic behaviors in 3- and 7-year-olds showed a particularly strong influence, increasing the risk of psychosis in preteens by up to 300%, Rhys Bevan Jones, Ph.D., and colleagues wrote in the March issue of Schizophrenia Research (Schizophr Res. 2012;135:164-9).
In the study, nearly 20% of children with these characteristics at age 7 went on to develop psychotic episodes by age 12.
"The findings of this study suggest that clinicians should ask about psychotic experiences in those with autistic traits," wrote Dr. Jones of Cardiff (Wales) University. "Clinicians should also assess for traits of autism in those who develop psychotic experiences (23% of those with psychotic experiences in adolescence had at least one autistic trait at the age of 3, and 10% had at least one psychotic trait at the age of 7). Clinical care is likely to be enhanced by careful consideration of premorbid and comorbid autistic traits that might be impacting on patient function."
"It is also possible that [autism spectrum disorders] and psychotic disorders represent part of the same disorder that is manifest differently at different stages of development."
The authors examined data from the Avon Longitudinal Study of Parents and Children. The cohort includes 14,541 children who were born from April 1991 to December 1992, and assessed annually. Dr. Jones’s study comprised 6,439 with childhood data on autistic traits and who were interviewed at age 12 for any psychotic episode.
Psychosis was measured with the psychosis-like symptoms interview, which includes 12 core questions about hallucinations, delusions, and experiences of thought interference over the prior 6 months.
At age 3, 14% of the entire group had speech problems, 9% social problems, and 3% ritualistic behaviors. By age 7, many of these had disappeared, leaving 4% with speech problems, 3% with social problems, and 2% with ritualistic behaviors.
At age 12, 744 children (11.5%) had experienced a psychotic episode. Of these, 483 had no autistic traits. One trait was present in 115; 24 had two traits, and three children had all three of the autistic characteristics.
The investigators performed a multivariate analysis that controlled for male gender, family history of mental illness, low IQ, and parental education and income. Another confounder – peer bullying – was examined separately.
Compared to children without toddler-age language concerns, those with speech problems at age 3 were 58% more likely to have a psychotic episode by age 12 – a significant increase. But children who still had speech problems by age 7 were twice as likely to have a psychotic episode (odds ratio, 2.11).
Ritualistic behaviors were also significantly associated with preteen psychosis. Children who showed those behaviors at age 3 were 74% more likely to experience a psychotic episode. Children who still showed ritualistic behavior at age 7 were 300% more likely to have experienced psychosis by age 12 (OR, 3.05). Social difficulties at both ages 3 and 7 did not significantly increase the risk of late psychosis.
The risk of psychosis increased by 33% for every autistic characteristic in a toddler, and by 66% for every characteristic still present at age 7.
Peer victimization also increased the risk of psychosis, the authors noted. Having autistic traits at age 7 increased the risk of bullying by 38%, and bullying at age 8 was associated with a 69% increase in the risk of psychosis.
It’s unclear whether the associations are driven by a common neurobiological underpinning, social stressors associated with autistic characteristics, or a combination of both, the authors wrote.
[Autism spectrum disorders] can lead to a number of potential stressors, including social rejection or isolation, peer victimization, and stresses from academic failure. It is possible that these stresses mediate an increased risk of future psychotic experiences. "It is also possible that [autism spectrum disorders] and psychotic disorders represent part of the same disorder that is manifest differently at different stages of development," they noted.
The study was sponsored by the U.K. Medical Research Council, the Wellcome Trust, and the University of Bristol (England). None of the authors had any financial declarations.
Autistic-like behaviors in toddlers might predict psychotic events in adolescence, a retrospective study has determined.
Speech problems and ritualistic behaviors in 3- and 7-year-olds showed a particularly strong influence, increasing the risk of psychosis in preteens by up to 300%, Rhys Bevan Jones, Ph.D., and colleagues wrote in the March issue of Schizophrenia Research (Schizophr Res. 2012;135:164-9).
In the study, nearly 20% of children with these characteristics at age 7 went on to develop psychotic episodes by age 12.
"The findings of this study suggest that clinicians should ask about psychotic experiences in those with autistic traits," wrote Dr. Jones of Cardiff (Wales) University. "Clinicians should also assess for traits of autism in those who develop psychotic experiences (23% of those with psychotic experiences in adolescence had at least one autistic trait at the age of 3, and 10% had at least one psychotic trait at the age of 7). Clinical care is likely to be enhanced by careful consideration of premorbid and comorbid autistic traits that might be impacting on patient function."
"It is also possible that [autism spectrum disorders] and psychotic disorders represent part of the same disorder that is manifest differently at different stages of development."
The authors examined data from the Avon Longitudinal Study of Parents and Children. The cohort includes 14,541 children who were born from April 1991 to December 1992, and assessed annually. Dr. Jones’s study comprised 6,439 with childhood data on autistic traits and who were interviewed at age 12 for any psychotic episode.
Psychosis was measured with the psychosis-like symptoms interview, which includes 12 core questions about hallucinations, delusions, and experiences of thought interference over the prior 6 months.
At age 3, 14% of the entire group had speech problems, 9% social problems, and 3% ritualistic behaviors. By age 7, many of these had disappeared, leaving 4% with speech problems, 3% with social problems, and 2% with ritualistic behaviors.
At age 12, 744 children (11.5%) had experienced a psychotic episode. Of these, 483 had no autistic traits. One trait was present in 115; 24 had two traits, and three children had all three of the autistic characteristics.
The investigators performed a multivariate analysis that controlled for male gender, family history of mental illness, low IQ, and parental education and income. Another confounder – peer bullying – was examined separately.
Compared to children without toddler-age language concerns, those with speech problems at age 3 were 58% more likely to have a psychotic episode by age 12 – a significant increase. But children who still had speech problems by age 7 were twice as likely to have a psychotic episode (odds ratio, 2.11).
Ritualistic behaviors were also significantly associated with preteen psychosis. Children who showed those behaviors at age 3 were 74% more likely to experience a psychotic episode. Children who still showed ritualistic behavior at age 7 were 300% more likely to have experienced psychosis by age 12 (OR, 3.05). Social difficulties at both ages 3 and 7 did not significantly increase the risk of late psychosis.
The risk of psychosis increased by 33% for every autistic characteristic in a toddler, and by 66% for every characteristic still present at age 7.
Peer victimization also increased the risk of psychosis, the authors noted. Having autistic traits at age 7 increased the risk of bullying by 38%, and bullying at age 8 was associated with a 69% increase in the risk of psychosis.
It’s unclear whether the associations are driven by a common neurobiological underpinning, social stressors associated with autistic characteristics, or a combination of both, the authors wrote.
[Autism spectrum disorders] can lead to a number of potential stressors, including social rejection or isolation, peer victimization, and stresses from academic failure. It is possible that these stresses mediate an increased risk of future psychotic experiences. "It is also possible that [autism spectrum disorders] and psychotic disorders represent part of the same disorder that is manifest differently at different stages of development," they noted.
The study was sponsored by the U.K. Medical Research Council, the Wellcome Trust, and the University of Bristol (England). None of the authors had any financial declarations.
Autistic-like behaviors in toddlers might predict psychotic events in adolescence, a retrospective study has determined.
Speech problems and ritualistic behaviors in 3- and 7-year-olds showed a particularly strong influence, increasing the risk of psychosis in preteens by up to 300%, Rhys Bevan Jones, Ph.D., and colleagues wrote in the March issue of Schizophrenia Research (Schizophr Res. 2012;135:164-9).
In the study, nearly 20% of children with these characteristics at age 7 went on to develop psychotic episodes by age 12.
"The findings of this study suggest that clinicians should ask about psychotic experiences in those with autistic traits," wrote Dr. Jones of Cardiff (Wales) University. "Clinicians should also assess for traits of autism in those who develop psychotic experiences (23% of those with psychotic experiences in adolescence had at least one autistic trait at the age of 3, and 10% had at least one psychotic trait at the age of 7). Clinical care is likely to be enhanced by careful consideration of premorbid and comorbid autistic traits that might be impacting on patient function."
"It is also possible that [autism spectrum disorders] and psychotic disorders represent part of the same disorder that is manifest differently at different stages of development."
The authors examined data from the Avon Longitudinal Study of Parents and Children. The cohort includes 14,541 children who were born from April 1991 to December 1992, and assessed annually. Dr. Jones’s study comprised 6,439 with childhood data on autistic traits and who were interviewed at age 12 for any psychotic episode.
Psychosis was measured with the psychosis-like symptoms interview, which includes 12 core questions about hallucinations, delusions, and experiences of thought interference over the prior 6 months.
At age 3, 14% of the entire group had speech problems, 9% social problems, and 3% ritualistic behaviors. By age 7, many of these had disappeared, leaving 4% with speech problems, 3% with social problems, and 2% with ritualistic behaviors.
At age 12, 744 children (11.5%) had experienced a psychotic episode. Of these, 483 had no autistic traits. One trait was present in 115; 24 had two traits, and three children had all three of the autistic characteristics.
The investigators performed a multivariate analysis that controlled for male gender, family history of mental illness, low IQ, and parental education and income. Another confounder – peer bullying – was examined separately.
Compared to children without toddler-age language concerns, those with speech problems at age 3 were 58% more likely to have a psychotic episode by age 12 – a significant increase. But children who still had speech problems by age 7 were twice as likely to have a psychotic episode (odds ratio, 2.11).
Ritualistic behaviors were also significantly associated with preteen psychosis. Children who showed those behaviors at age 3 were 74% more likely to experience a psychotic episode. Children who still showed ritualistic behavior at age 7 were 300% more likely to have experienced psychosis by age 12 (OR, 3.05). Social difficulties at both ages 3 and 7 did not significantly increase the risk of late psychosis.
The risk of psychosis increased by 33% for every autistic characteristic in a toddler, and by 66% for every characteristic still present at age 7.
Peer victimization also increased the risk of psychosis, the authors noted. Having autistic traits at age 7 increased the risk of bullying by 38%, and bullying at age 8 was associated with a 69% increase in the risk of psychosis.
It’s unclear whether the associations are driven by a common neurobiological underpinning, social stressors associated with autistic characteristics, or a combination of both, the authors wrote.
[Autism spectrum disorders] can lead to a number of potential stressors, including social rejection or isolation, peer victimization, and stresses from academic failure. It is possible that these stresses mediate an increased risk of future psychotic experiences. "It is also possible that [autism spectrum disorders] and psychotic disorders represent part of the same disorder that is manifest differently at different stages of development," they noted.
The study was sponsored by the U.K. Medical Research Council, the Wellcome Trust, and the University of Bristol (England). None of the authors had any financial declarations.
FROM SCHIZOPHRENIA RESEARCH
Major Finding: Characteristics of autism at ages 3 and 7 are associated with up to a 300% increase in the risk of having a psychotic episode by age 12.
Data Source: The retrospective study included data on 6,439 children who had been followed since birth and interviewed at age 12 for any evidence of psychosis.
Disclosures: The study was sponsored by the U.K. Medical Research Council, the Wellcome Trust, and the University of Bristol (England). None of the authors had any financial declarations.
Antipsychotics Can Spur 'Metabolic Train Wreck'
NEW YORK – With new national data finding that 17% of the country's youngsters are obese, physicians who treat children with antipsychotics face even tougher medication choices.
Because many of the drugs are tied to increases in weight and obesity-driven diseases, it's not hard to see the looming metabolic train wreck, Dr. Christoph U. Correll said at the meeting.
The newest NHANES (National Health and Nutrition Survey), released in January, found that more than one-third of U.S. adults and almost 17% of youth were classified as obese in 2009-2010.
“We are now seeing obese children with type 2 diabetes, and we know that this disease has the same 8-year risk as a myocardial infarction for [another] heart attack,” said Dr. Correll of the Zucker Hillside Hospital in Glen Oak, N.Y. “We will see obese children who will not outlive their parents, and we must admit that our treatments are contributing to this.”
Studies show that many antipsychotic medications are associated with significant weight gain, much of that leading to metabolic syndrome, Dr. Correll said. A 2011 meta-analysis of 3,000 children found that those taking olanzapine gained almost 4 kg within just 1 month of starting the medication (Euro. Psychiatry 2011:26:144-58). Quetiapine was associated with a gain of 2 kg over 4 weeks, and risperidone with a gain of 2 kg over 7 weeks. Patients taking aripiprazole gained 1 kg over 6 weeks, whereas those taking a placebo had no weight change.
This phenomenon is directly related to the onset of metabolic syndrome, and its associated cardiovascular risks of lipid abnormalities and hypertension, Dr. Correll said. “The longer you treat, the higher are these rates. And the longer you have cardiovascular risk factors, the greater your chance of death.”
A 2006 study illustrates his point. By 10 years of treatment, 21% of patients had metabolic syndrome; by 20 years of treatment, 34%, and after 20 years, 39%.
Age played a significant role, as expected, but a significant difference was found between the general population and patients taking the drugs. Among patients aged 35-45 years, 35% had metabolic syndrome, compared with 6.5% of nonpatients. By 55 years, the prevalence had doubled in the general population, to about 14%, while it increased significantly less in patients, to 38%.
“This shows that we are shifting the metabolic burden to earlier in life. These patients are prematurely metabolically demented, and we are doing this by not taking care of their weight and metabolic problems.”
This leaves psychiatrists on the horns of a dilemma, Dr. Correll said. Patients need treatment, but the drugs come with a high physiological price. To decrease patients' exposure to metabolic risks, he advises starting treatment with the lowest metabolically driven medications, and moving to other drugs only if the less risky ones aren't controlling symptoms.
Studies suggest that, in most patients, olanzapine confers the greatest risk of weight gain, and risperidone the lowest. Lurasidone is doing “quite well” in adult studies, as far as weight gain goes (Drugs Today [Barc.] 2011;47:807–16), but it has not been examined in children or adolescents. “I think this is something we as pediatric psychiatrists should be trying to get our hands on,” Dr. Correll said.
A newly published, randomized trial suggests that valproate might change some of the metabolic activity that drives weight gain in patients taking antipsychotic medications.
Patients with treatment-resistant schizophrenia, schizoaffective disorder, or bipolar disorder were randomized to olanzapine or risperidone alone, or to either of the drugs plus valproate as a mood stabilizer. Every one of those taking olanzapine plus valproate experienced significant weight gains, whereas the weight gain occurred in 60% of those taking olanzapine alone.
Weight gain was less common in both risperidone groups. But the addition of valproate seemed to enhance risperidone's weight-neutral effect: Some 15% of those taking the combination gained significant amounts of weight – significantly fewer than the 30% who gained weight on risperidone alone (J. Clin. Psychiatry 2011;72:1602-10).
The same pattern held with other metabolic measures, including changes in body mass index, glycosylated hemoglobin, total cholesterol, and the triglyceride/HDL cholesterol ratio.
The findings point out just how much remains unknown about the metabolic effects of antipsychotic medications. For now, Dr. Correll said, the best path is to start with the lowest metabolically active drug appropriate for that patient, and closely monitor any metabolic changes.
“At each visit, obtain both weight and body mass index,” he said. “Measure waist circumference at least once a year, and provide counseling on diet and exercise.”
Dr. Correll disclosed that he is a consultant for and is on the advisory boards of numerous drug companies, and also receives research support and honoraria from many of them.
NEW YORK – With new national data finding that 17% of the country's youngsters are obese, physicians who treat children with antipsychotics face even tougher medication choices.
Because many of the drugs are tied to increases in weight and obesity-driven diseases, it's not hard to see the looming metabolic train wreck, Dr. Christoph U. Correll said at the meeting.
The newest NHANES (National Health and Nutrition Survey), released in January, found that more than one-third of U.S. adults and almost 17% of youth were classified as obese in 2009-2010.
“We are now seeing obese children with type 2 diabetes, and we know that this disease has the same 8-year risk as a myocardial infarction for [another] heart attack,” said Dr. Correll of the Zucker Hillside Hospital in Glen Oak, N.Y. “We will see obese children who will not outlive their parents, and we must admit that our treatments are contributing to this.”
Studies show that many antipsychotic medications are associated with significant weight gain, much of that leading to metabolic syndrome, Dr. Correll said. A 2011 meta-analysis of 3,000 children found that those taking olanzapine gained almost 4 kg within just 1 month of starting the medication (Euro. Psychiatry 2011:26:144-58). Quetiapine was associated with a gain of 2 kg over 4 weeks, and risperidone with a gain of 2 kg over 7 weeks. Patients taking aripiprazole gained 1 kg over 6 weeks, whereas those taking a placebo had no weight change.
This phenomenon is directly related to the onset of metabolic syndrome, and its associated cardiovascular risks of lipid abnormalities and hypertension, Dr. Correll said. “The longer you treat, the higher are these rates. And the longer you have cardiovascular risk factors, the greater your chance of death.”
A 2006 study illustrates his point. By 10 years of treatment, 21% of patients had metabolic syndrome; by 20 years of treatment, 34%, and after 20 years, 39%.
Age played a significant role, as expected, but a significant difference was found between the general population and patients taking the drugs. Among patients aged 35-45 years, 35% had metabolic syndrome, compared with 6.5% of nonpatients. By 55 years, the prevalence had doubled in the general population, to about 14%, while it increased significantly less in patients, to 38%.
“This shows that we are shifting the metabolic burden to earlier in life. These patients are prematurely metabolically demented, and we are doing this by not taking care of their weight and metabolic problems.”
This leaves psychiatrists on the horns of a dilemma, Dr. Correll said. Patients need treatment, but the drugs come with a high physiological price. To decrease patients' exposure to metabolic risks, he advises starting treatment with the lowest metabolically driven medications, and moving to other drugs only if the less risky ones aren't controlling symptoms.
Studies suggest that, in most patients, olanzapine confers the greatest risk of weight gain, and risperidone the lowest. Lurasidone is doing “quite well” in adult studies, as far as weight gain goes (Drugs Today [Barc.] 2011;47:807–16), but it has not been examined in children or adolescents. “I think this is something we as pediatric psychiatrists should be trying to get our hands on,” Dr. Correll said.
A newly published, randomized trial suggests that valproate might change some of the metabolic activity that drives weight gain in patients taking antipsychotic medications.
Patients with treatment-resistant schizophrenia, schizoaffective disorder, or bipolar disorder were randomized to olanzapine or risperidone alone, or to either of the drugs plus valproate as a mood stabilizer. Every one of those taking olanzapine plus valproate experienced significant weight gains, whereas the weight gain occurred in 60% of those taking olanzapine alone.
Weight gain was less common in both risperidone groups. But the addition of valproate seemed to enhance risperidone's weight-neutral effect: Some 15% of those taking the combination gained significant amounts of weight – significantly fewer than the 30% who gained weight on risperidone alone (J. Clin. Psychiatry 2011;72:1602-10).
The same pattern held with other metabolic measures, including changes in body mass index, glycosylated hemoglobin, total cholesterol, and the triglyceride/HDL cholesterol ratio.
The findings point out just how much remains unknown about the metabolic effects of antipsychotic medications. For now, Dr. Correll said, the best path is to start with the lowest metabolically active drug appropriate for that patient, and closely monitor any metabolic changes.
“At each visit, obtain both weight and body mass index,” he said. “Measure waist circumference at least once a year, and provide counseling on diet and exercise.”
Dr. Correll disclosed that he is a consultant for and is on the advisory boards of numerous drug companies, and also receives research support and honoraria from many of them.
NEW YORK – With new national data finding that 17% of the country's youngsters are obese, physicians who treat children with antipsychotics face even tougher medication choices.
Because many of the drugs are tied to increases in weight and obesity-driven diseases, it's not hard to see the looming metabolic train wreck, Dr. Christoph U. Correll said at the meeting.
The newest NHANES (National Health and Nutrition Survey), released in January, found that more than one-third of U.S. adults and almost 17% of youth were classified as obese in 2009-2010.
“We are now seeing obese children with type 2 diabetes, and we know that this disease has the same 8-year risk as a myocardial infarction for [another] heart attack,” said Dr. Correll of the Zucker Hillside Hospital in Glen Oak, N.Y. “We will see obese children who will not outlive their parents, and we must admit that our treatments are contributing to this.”
Studies show that many antipsychotic medications are associated with significant weight gain, much of that leading to metabolic syndrome, Dr. Correll said. A 2011 meta-analysis of 3,000 children found that those taking olanzapine gained almost 4 kg within just 1 month of starting the medication (Euro. Psychiatry 2011:26:144-58). Quetiapine was associated with a gain of 2 kg over 4 weeks, and risperidone with a gain of 2 kg over 7 weeks. Patients taking aripiprazole gained 1 kg over 6 weeks, whereas those taking a placebo had no weight change.
This phenomenon is directly related to the onset of metabolic syndrome, and its associated cardiovascular risks of lipid abnormalities and hypertension, Dr. Correll said. “The longer you treat, the higher are these rates. And the longer you have cardiovascular risk factors, the greater your chance of death.”
A 2006 study illustrates his point. By 10 years of treatment, 21% of patients had metabolic syndrome; by 20 years of treatment, 34%, and after 20 years, 39%.
Age played a significant role, as expected, but a significant difference was found between the general population and patients taking the drugs. Among patients aged 35-45 years, 35% had metabolic syndrome, compared with 6.5% of nonpatients. By 55 years, the prevalence had doubled in the general population, to about 14%, while it increased significantly less in patients, to 38%.
“This shows that we are shifting the metabolic burden to earlier in life. These patients are prematurely metabolically demented, and we are doing this by not taking care of their weight and metabolic problems.”
This leaves psychiatrists on the horns of a dilemma, Dr. Correll said. Patients need treatment, but the drugs come with a high physiological price. To decrease patients' exposure to metabolic risks, he advises starting treatment with the lowest metabolically driven medications, and moving to other drugs only if the less risky ones aren't controlling symptoms.
Studies suggest that, in most patients, olanzapine confers the greatest risk of weight gain, and risperidone the lowest. Lurasidone is doing “quite well” in adult studies, as far as weight gain goes (Drugs Today [Barc.] 2011;47:807–16), but it has not been examined in children or adolescents. “I think this is something we as pediatric psychiatrists should be trying to get our hands on,” Dr. Correll said.
A newly published, randomized trial suggests that valproate might change some of the metabolic activity that drives weight gain in patients taking antipsychotic medications.
Patients with treatment-resistant schizophrenia, schizoaffective disorder, or bipolar disorder were randomized to olanzapine or risperidone alone, or to either of the drugs plus valproate as a mood stabilizer. Every one of those taking olanzapine plus valproate experienced significant weight gains, whereas the weight gain occurred in 60% of those taking olanzapine alone.
Weight gain was less common in both risperidone groups. But the addition of valproate seemed to enhance risperidone's weight-neutral effect: Some 15% of those taking the combination gained significant amounts of weight – significantly fewer than the 30% who gained weight on risperidone alone (J. Clin. Psychiatry 2011;72:1602-10).
The same pattern held with other metabolic measures, including changes in body mass index, glycosylated hemoglobin, total cholesterol, and the triglyceride/HDL cholesterol ratio.
The findings point out just how much remains unknown about the metabolic effects of antipsychotic medications. For now, Dr. Correll said, the best path is to start with the lowest metabolically active drug appropriate for that patient, and closely monitor any metabolic changes.
“At each visit, obtain both weight and body mass index,” he said. “Measure waist circumference at least once a year, and provide counseling on diet and exercise.”
Dr. Correll disclosed that he is a consultant for and is on the advisory boards of numerous drug companies, and also receives research support and honoraria from many of them.
EXPERT ANALYSIS FROM A PSYCHOPHARMACOLOGY UPDATE SPONSORED BY THE AMERICAN ACADEMY OF CHILD AND ADOLESCENT PSYCHIATRY