User login
Patients Hit Web Survey When TIAs Threaten
NEW ORLEANS – A brief online evaluation may prompt patients to seek quick medical attention if they experience symptoms of stroke or transient ischemic attack.
Researchers are evaluating the WebTIA Program, a 26-item questionnaire that identifies risk factors and symptoms common to stroke and TIA. The investigative deployment has enrolled more than 251 people from across the country, who came upon WebTIA when searching for information on symptoms, Dr. Anthony Kim said at an international stroke conference.
Immediate medical attention offers the best outcomes for patients, said Dr. Kim, medical director of the University of California, San Francisco, Stroke Center. But because TIA symptoms can come and go suddenly, they may not be enough to prompt a patient to visit to the emergency department, or even a call to the physician.
"There is a perception that because the symptoms resolved or improved, they are not as serious," he said at the meeting, sponsored by the American Stroke Association. "Even when the symptoms are correctly recognized as a TIA or stroke, people tend to underappreciate the need for urgent evaluation."
The Internet could improve this understanding, because it’s available 24 hours a day and there are no barriers to access, as there can be with a doctor’s or emergency department visit.
The ongoing WebTIA survey aims to test the utility of the Internet as a resource for helping people identify serious cerebrovascular symptoms. The survey page comes up as an option when people search for key words or phrases, such as "Am I having a ministroke." A click-through ad also appears on some other sites with stroke information.
The WebTIA site includes videos and text explanations of the TIA and stroke, and describes how taking the survey helps researchers create an effective Web-based risk-evaluation tool. The step-by-step program begins with six qualification questions and then guides visitors through the process of informed consent and explains Internet privacy terms. If patients agree, they answer a 26-item questionnaire that incorporates the ABCD2 score, as well as questions that give some hint about the brain territory affected. If visitors indicate that symptoms are occurring at the moment, the survey urges them to stop reading and seek emergency help.
The concluding page gives visitors the option to contact a study neurologist immediately, by phone or by computer, and reminds them that they’ll get a follow-up call from a neurologist participating in the study.
In the first 3 months, the WebTIA site received an average of 200 hits per day. More than 250 people completed the online interview; there were 175 follow-up interviews. The mean age was 59 years; the oldest visitor was aged 88 years, showing that elderly people can easily use the Internet to access information.
About two-thirds of the visitors had sought medical advice for their symptoms. Many noted that their symptoms had occurred within the prior 30 minutes and subsided. One man, vacationing in a recreational vehicle, completed the survey from his phone. About a quarter of visitors were sent to the hospital within minutes, hours, or a day.
Follow-up interviews determined that 43 (25%) had suffered a TIA and that 67 (39%) had either a TIA or a stroke. "There was good agreement between the online tool and our neurologists’ assessment," Dr. Kim remarked.
The ABCD2 score itself, however, was only moderately predictive, with a discrimination rate of 69%. "One of the 12 patients with a risk score of 0 had a cerebrovascular event of isolated diplopia," Dr. Kim said. Incorporating a question about diplopia and one about prior TIA or stroke improved the discrimination rate to 78%. By this calculation, seven patients had a risk score of 0, and none of those had an event.
"We identified a population of patients with possible TIA or stroke that might be a promising target for a computer-based public health intervention that could reduce delays to medical intervention." Such interventions would involve minimal cost and have the potential to reach a very large population, Dr. Kim said.
The project was funded by a grant from the National Stroke Association and Boehringer Ingelheim. Dr. Kim said he received a research grant from the company.
NEW ORLEANS – A brief online evaluation may prompt patients to seek quick medical attention if they experience symptoms of stroke or transient ischemic attack.
Researchers are evaluating the WebTIA Program, a 26-item questionnaire that identifies risk factors and symptoms common to stroke and TIA. The investigative deployment has enrolled more than 251 people from across the country, who came upon WebTIA when searching for information on symptoms, Dr. Anthony Kim said at an international stroke conference.
Immediate medical attention offers the best outcomes for patients, said Dr. Kim, medical director of the University of California, San Francisco, Stroke Center. But because TIA symptoms can come and go suddenly, they may not be enough to prompt a patient to visit to the emergency department, or even a call to the physician.
"There is a perception that because the symptoms resolved or improved, they are not as serious," he said at the meeting, sponsored by the American Stroke Association. "Even when the symptoms are correctly recognized as a TIA or stroke, people tend to underappreciate the need for urgent evaluation."
The Internet could improve this understanding, because it’s available 24 hours a day and there are no barriers to access, as there can be with a doctor’s or emergency department visit.
The ongoing WebTIA survey aims to test the utility of the Internet as a resource for helping people identify serious cerebrovascular symptoms. The survey page comes up as an option when people search for key words or phrases, such as "Am I having a ministroke." A click-through ad also appears on some other sites with stroke information.
The WebTIA site includes videos and text explanations of the TIA and stroke, and describes how taking the survey helps researchers create an effective Web-based risk-evaluation tool. The step-by-step program begins with six qualification questions and then guides visitors through the process of informed consent and explains Internet privacy terms. If patients agree, they answer a 26-item questionnaire that incorporates the ABCD2 score, as well as questions that give some hint about the brain territory affected. If visitors indicate that symptoms are occurring at the moment, the survey urges them to stop reading and seek emergency help.
The concluding page gives visitors the option to contact a study neurologist immediately, by phone or by computer, and reminds them that they’ll get a follow-up call from a neurologist participating in the study.
In the first 3 months, the WebTIA site received an average of 200 hits per day. More than 250 people completed the online interview; there were 175 follow-up interviews. The mean age was 59 years; the oldest visitor was aged 88 years, showing that elderly people can easily use the Internet to access information.
About two-thirds of the visitors had sought medical advice for their symptoms. Many noted that their symptoms had occurred within the prior 30 minutes and subsided. One man, vacationing in a recreational vehicle, completed the survey from his phone. About a quarter of visitors were sent to the hospital within minutes, hours, or a day.
Follow-up interviews determined that 43 (25%) had suffered a TIA and that 67 (39%) had either a TIA or a stroke. "There was good agreement between the online tool and our neurologists’ assessment," Dr. Kim remarked.
The ABCD2 score itself, however, was only moderately predictive, with a discrimination rate of 69%. "One of the 12 patients with a risk score of 0 had a cerebrovascular event of isolated diplopia," Dr. Kim said. Incorporating a question about diplopia and one about prior TIA or stroke improved the discrimination rate to 78%. By this calculation, seven patients had a risk score of 0, and none of those had an event.
"We identified a population of patients with possible TIA or stroke that might be a promising target for a computer-based public health intervention that could reduce delays to medical intervention." Such interventions would involve minimal cost and have the potential to reach a very large population, Dr. Kim said.
The project was funded by a grant from the National Stroke Association and Boehringer Ingelheim. Dr. Kim said he received a research grant from the company.
NEW ORLEANS – A brief online evaluation may prompt patients to seek quick medical attention if they experience symptoms of stroke or transient ischemic attack.
Researchers are evaluating the WebTIA Program, a 26-item questionnaire that identifies risk factors and symptoms common to stroke and TIA. The investigative deployment has enrolled more than 251 people from across the country, who came upon WebTIA when searching for information on symptoms, Dr. Anthony Kim said at an international stroke conference.
Immediate medical attention offers the best outcomes for patients, said Dr. Kim, medical director of the University of California, San Francisco, Stroke Center. But because TIA symptoms can come and go suddenly, they may not be enough to prompt a patient to visit to the emergency department, or even a call to the physician.
"There is a perception that because the symptoms resolved or improved, they are not as serious," he said at the meeting, sponsored by the American Stroke Association. "Even when the symptoms are correctly recognized as a TIA or stroke, people tend to underappreciate the need for urgent evaluation."
The Internet could improve this understanding, because it’s available 24 hours a day and there are no barriers to access, as there can be with a doctor’s or emergency department visit.
The ongoing WebTIA survey aims to test the utility of the Internet as a resource for helping people identify serious cerebrovascular symptoms. The survey page comes up as an option when people search for key words or phrases, such as "Am I having a ministroke." A click-through ad also appears on some other sites with stroke information.
The WebTIA site includes videos and text explanations of the TIA and stroke, and describes how taking the survey helps researchers create an effective Web-based risk-evaluation tool. The step-by-step program begins with six qualification questions and then guides visitors through the process of informed consent and explains Internet privacy terms. If patients agree, they answer a 26-item questionnaire that incorporates the ABCD2 score, as well as questions that give some hint about the brain territory affected. If visitors indicate that symptoms are occurring at the moment, the survey urges them to stop reading and seek emergency help.
The concluding page gives visitors the option to contact a study neurologist immediately, by phone or by computer, and reminds them that they’ll get a follow-up call from a neurologist participating in the study.
In the first 3 months, the WebTIA site received an average of 200 hits per day. More than 250 people completed the online interview; there were 175 follow-up interviews. The mean age was 59 years; the oldest visitor was aged 88 years, showing that elderly people can easily use the Internet to access information.
About two-thirds of the visitors had sought medical advice for their symptoms. Many noted that their symptoms had occurred within the prior 30 minutes and subsided. One man, vacationing in a recreational vehicle, completed the survey from his phone. About a quarter of visitors were sent to the hospital within minutes, hours, or a day.
Follow-up interviews determined that 43 (25%) had suffered a TIA and that 67 (39%) had either a TIA or a stroke. "There was good agreement between the online tool and our neurologists’ assessment," Dr. Kim remarked.
The ABCD2 score itself, however, was only moderately predictive, with a discrimination rate of 69%. "One of the 12 patients with a risk score of 0 had a cerebrovascular event of isolated diplopia," Dr. Kim said. Incorporating a question about diplopia and one about prior TIA or stroke improved the discrimination rate to 78%. By this calculation, seven patients had a risk score of 0, and none of those had an event.
"We identified a population of patients with possible TIA or stroke that might be a promising target for a computer-based public health intervention that could reduce delays to medical intervention." Such interventions would involve minimal cost and have the potential to reach a very large population, Dr. Kim said.
The project was funded by a grant from the National Stroke Association and Boehringer Ingelheim. Dr. Kim said he received a research grant from the company.
FROM THE INTERNATIONAL STROKE CONFERENCE
Major Finding: An investigational Web-based survey can identify symptoms of TIA or stroke in up to 78% of people who visit the site and fill out a questionnaire.
Data Source: The pilot WebTIA program, which has so far enrolled more than 250 patients.
Disclosures: The project was funded by a grant from the National Stroke Association and Boehringer Ingelheim. Dr. Kim said he received a research grant from the company
Haloperidol Doubles Risk of Death in Institutionalized Elderly
Compared with risperidone, haloperidol appeared to double the risk of death among elderly nursing home patients, while quetiapine was associated with a 20% decreased risk.
While some other antipsychotic medications appeared uniformly safe, haloperidol emerged as a dangerous way to manage an elderly patient with a behavioral problem, Krista F. Huybrechts, Ph.D., and her colleagues wrote Feb. 23 in BMJ (2012;344:e977 [doi:10.1136/bmj.e977]).
"The evidence accumulated so far implies that use of haloperidol in this vulnerable population cannot be justified because of the excess harm," wrote Dr. Huybrechts of Brigham and Women’s Hospital, Boston.
The findings emphasize the dangers of using drugs as a first-line therapy for elderly patients with behavioral problems. "If the clinician faces a situation in which the use of these drugs seem inevitable, our findings underscore the importance of always prescribing the lowest possible dose and of closely monitoring patients, especially shortly after the start of treatment."
Dr. Huybrechts and her coauthors reviewed medication and mortality data on 75,445 nursing home residents who were new users of antipsychotic medications from 2001-2005. About 60% of the group had some form of dementia. Other psychiatric diagnoses included depression, anxiety, delirium, and psychotic disorders. All of the patients were aged 65 years or older. Many also had physical comorbidities, including a history of heart attack, cardiac arrhythmias, ischemic heart disease, hypertension, cerebrovascular disease, diabetes, and Parkinson’s disease.
The study examined the use of haloperidol, aripiprazole, olanzapine, risperidone, and ziprasidone. Other medications, like thioridazine and chlorpromazine, were excluded from the analysis because they were infrequently prescribed in the group.
Most of the patients were censored from the record before 180 days. A discontinuation of treatment was the most common reason (57%), but some patients stopped because of a hospital admission (17%), a change in medication (13%), or death (12%).
There were 6,598 deaths (9% of the study cohort) during the first 180 days of antipsychotic treatment – a rate of 37 per person per year of treatment. About half of these were caused by circulatory disorders (49%). The other large causes were cerebrovascular diseases (10%) and respiratory disorders (15%); the remaining 26% of deaths were tied to illnesses unspecified in the analysis.
In a regression analysis, those taking haloperidol were twice as likely to die from any cause as those taking risperidone (hazard ratio, 2.07). The risk was strongest in the first 40 days of treatment (HR, 2.34) and declined thereafter. By 79 days, haloperidol patients were 32% more likely to have died; from 80-180 days, the increased risk was 46%.
Conversely, patients taking quetiapine were significantly less likely to die (HR, 0.81), and that risk remained fairly contrast throughout the 180-day study period.
No significant or clinically meaningful mortality trends were associated with the use of aripiprazole, olanzapine, risperidone, or ziprasidone.
The investigators found no significant interactions between the drug administered and the cause of death. "The increased risk of death with haloperidol and the decreased risk with quetiapine were observed for all causes examined," they said. However, the analysis suggested that olanzapine might be associated with a decrease in the risk of cerebrovascular death. There were insufficient data to draw any conclusions about mortality type in patients taking aripiprazole or ziprasidone.
Every drug except quetiapine showed a dose-response relationship with all-cause mortality. The relationship was most pronounced for haloperidol, with an 84% increased risk in those taking a high dose, and a 40% increased risk for those taking a medium dose, compared with those taking a low dose.
The study emphasizes the need to use alternative behavioral management tools for as long as possible in nursing home patients, the authors said.
"[It] reinforces the important risks associated with the use of these drugs and underscores the need to try alternative means of dealing with behavioral problems in older patients with dementia."
The Food and Drug Administration, and the Agency for Healthcare Research and Quality funded the study. None of the authors reported any financial conflicts.
Before prescribing any drug, the physician must consider both its risks and benefits, Dr. Jenny McCleery said in an accompanying editorial. But research continues to identify haloperidol as a uniquely dangerous drug for elderly institutionalized patients.
As a class, antipsychotic medications are rife with serious adverse events, especially for older patients, and most are only somewhat effective in controlling behavioral and psychological symptoms. Faced with agitated patients, worried families, and stressed staff, physicians often feel compelled to offer some hope, but seem conflicted about the value of first offering interventions other than medication.
"Guidelines universally agree that the first-line treatment for behavioral and psychological symptoms in dementia should be non–drug based, [but also] recommend the careful use of antipsychotics in the treatment of agitation, aggression, or psychosis that fails to respond to other measures and that reaches various severity thresholds, typically severe distress or serious risk to self or others."
Despite recommendations to limit the drugs’ use, however, antipsychotics are still widely prescribed for elderly patients with dementia or those who are institutionalized.
"Few clinical problems place doctors in as tangled a web of clinical evidence, social policy, and ethical concerns as how to manage behavioral problems in patients with dementia," Dr. McCleery wrote. Studies hint that doctors feel pressured to prescribe anything that might help and that they believe alternative therapies aren’t feasible or effective because they are hard to implement and require intensive clinician involvement.
"Where care homes or community care services are inadequate and local clinical resources cannot compensate for them, doctors face genuine dilemmas about how to respond to distressed patients, relatives, and carers, often in ethically complex situations that involve a variety of risks. It is probably fair to say that many doctors think that the evidence-based guidelines are not adequate for the day to day reality of practice."
Dr. McCleery is a consulting psychiatrist for the Oxford (England) Health National Health Systems Foundation Trust. She has acted as a local investigator for Eli Lilly (BMJ 2012;344:e1093).
Before prescribing any drug, the physician must consider both its risks and benefits, Dr. Jenny McCleery said in an accompanying editorial. But research continues to identify haloperidol as a uniquely dangerous drug for elderly institutionalized patients.
As a class, antipsychotic medications are rife with serious adverse events, especially for older patients, and most are only somewhat effective in controlling behavioral and psychological symptoms. Faced with agitated patients, worried families, and stressed staff, physicians often feel compelled to offer some hope, but seem conflicted about the value of first offering interventions other than medication.
"Guidelines universally agree that the first-line treatment for behavioral and psychological symptoms in dementia should be non–drug based, [but also] recommend the careful use of antipsychotics in the treatment of agitation, aggression, or psychosis that fails to respond to other measures and that reaches various severity thresholds, typically severe distress or serious risk to self or others."
Despite recommendations to limit the drugs’ use, however, antipsychotics are still widely prescribed for elderly patients with dementia or those who are institutionalized.
"Few clinical problems place doctors in as tangled a web of clinical evidence, social policy, and ethical concerns as how to manage behavioral problems in patients with dementia," Dr. McCleery wrote. Studies hint that doctors feel pressured to prescribe anything that might help and that they believe alternative therapies aren’t feasible or effective because they are hard to implement and require intensive clinician involvement.
"Where care homes or community care services are inadequate and local clinical resources cannot compensate for them, doctors face genuine dilemmas about how to respond to distressed patients, relatives, and carers, often in ethically complex situations that involve a variety of risks. It is probably fair to say that many doctors think that the evidence-based guidelines are not adequate for the day to day reality of practice."
Dr. McCleery is a consulting psychiatrist for the Oxford (England) Health National Health Systems Foundation Trust. She has acted as a local investigator for Eli Lilly (BMJ 2012;344:e1093).
Before prescribing any drug, the physician must consider both its risks and benefits, Dr. Jenny McCleery said in an accompanying editorial. But research continues to identify haloperidol as a uniquely dangerous drug for elderly institutionalized patients.
As a class, antipsychotic medications are rife with serious adverse events, especially for older patients, and most are only somewhat effective in controlling behavioral and psychological symptoms. Faced with agitated patients, worried families, and stressed staff, physicians often feel compelled to offer some hope, but seem conflicted about the value of first offering interventions other than medication.
"Guidelines universally agree that the first-line treatment for behavioral and psychological symptoms in dementia should be non–drug based, [but also] recommend the careful use of antipsychotics in the treatment of agitation, aggression, or psychosis that fails to respond to other measures and that reaches various severity thresholds, typically severe distress or serious risk to self or others."
Despite recommendations to limit the drugs’ use, however, antipsychotics are still widely prescribed for elderly patients with dementia or those who are institutionalized.
"Few clinical problems place doctors in as tangled a web of clinical evidence, social policy, and ethical concerns as how to manage behavioral problems in patients with dementia," Dr. McCleery wrote. Studies hint that doctors feel pressured to prescribe anything that might help and that they believe alternative therapies aren’t feasible or effective because they are hard to implement and require intensive clinician involvement.
"Where care homes or community care services are inadequate and local clinical resources cannot compensate for them, doctors face genuine dilemmas about how to respond to distressed patients, relatives, and carers, often in ethically complex situations that involve a variety of risks. It is probably fair to say that many doctors think that the evidence-based guidelines are not adequate for the day to day reality of practice."
Dr. McCleery is a consulting psychiatrist for the Oxford (England) Health National Health Systems Foundation Trust. She has acted as a local investigator for Eli Lilly (BMJ 2012;344:e1093).
Compared with risperidone, haloperidol appeared to double the risk of death among elderly nursing home patients, while quetiapine was associated with a 20% decreased risk.
While some other antipsychotic medications appeared uniformly safe, haloperidol emerged as a dangerous way to manage an elderly patient with a behavioral problem, Krista F. Huybrechts, Ph.D., and her colleagues wrote Feb. 23 in BMJ (2012;344:e977 [doi:10.1136/bmj.e977]).
"The evidence accumulated so far implies that use of haloperidol in this vulnerable population cannot be justified because of the excess harm," wrote Dr. Huybrechts of Brigham and Women’s Hospital, Boston.
The findings emphasize the dangers of using drugs as a first-line therapy for elderly patients with behavioral problems. "If the clinician faces a situation in which the use of these drugs seem inevitable, our findings underscore the importance of always prescribing the lowest possible dose and of closely monitoring patients, especially shortly after the start of treatment."
Dr. Huybrechts and her coauthors reviewed medication and mortality data on 75,445 nursing home residents who were new users of antipsychotic medications from 2001-2005. About 60% of the group had some form of dementia. Other psychiatric diagnoses included depression, anxiety, delirium, and psychotic disorders. All of the patients were aged 65 years or older. Many also had physical comorbidities, including a history of heart attack, cardiac arrhythmias, ischemic heart disease, hypertension, cerebrovascular disease, diabetes, and Parkinson’s disease.
The study examined the use of haloperidol, aripiprazole, olanzapine, risperidone, and ziprasidone. Other medications, like thioridazine and chlorpromazine, were excluded from the analysis because they were infrequently prescribed in the group.
Most of the patients were censored from the record before 180 days. A discontinuation of treatment was the most common reason (57%), but some patients stopped because of a hospital admission (17%), a change in medication (13%), or death (12%).
There were 6,598 deaths (9% of the study cohort) during the first 180 days of antipsychotic treatment – a rate of 37 per person per year of treatment. About half of these were caused by circulatory disorders (49%). The other large causes were cerebrovascular diseases (10%) and respiratory disorders (15%); the remaining 26% of deaths were tied to illnesses unspecified in the analysis.
In a regression analysis, those taking haloperidol were twice as likely to die from any cause as those taking risperidone (hazard ratio, 2.07). The risk was strongest in the first 40 days of treatment (HR, 2.34) and declined thereafter. By 79 days, haloperidol patients were 32% more likely to have died; from 80-180 days, the increased risk was 46%.
Conversely, patients taking quetiapine were significantly less likely to die (HR, 0.81), and that risk remained fairly contrast throughout the 180-day study period.
No significant or clinically meaningful mortality trends were associated with the use of aripiprazole, olanzapine, risperidone, or ziprasidone.
The investigators found no significant interactions between the drug administered and the cause of death. "The increased risk of death with haloperidol and the decreased risk with quetiapine were observed for all causes examined," they said. However, the analysis suggested that olanzapine might be associated with a decrease in the risk of cerebrovascular death. There were insufficient data to draw any conclusions about mortality type in patients taking aripiprazole or ziprasidone.
Every drug except quetiapine showed a dose-response relationship with all-cause mortality. The relationship was most pronounced for haloperidol, with an 84% increased risk in those taking a high dose, and a 40% increased risk for those taking a medium dose, compared with those taking a low dose.
The study emphasizes the need to use alternative behavioral management tools for as long as possible in nursing home patients, the authors said.
"[It] reinforces the important risks associated with the use of these drugs and underscores the need to try alternative means of dealing with behavioral problems in older patients with dementia."
The Food and Drug Administration, and the Agency for Healthcare Research and Quality funded the study. None of the authors reported any financial conflicts.
Compared with risperidone, haloperidol appeared to double the risk of death among elderly nursing home patients, while quetiapine was associated with a 20% decreased risk.
While some other antipsychotic medications appeared uniformly safe, haloperidol emerged as a dangerous way to manage an elderly patient with a behavioral problem, Krista F. Huybrechts, Ph.D., and her colleagues wrote Feb. 23 in BMJ (2012;344:e977 [doi:10.1136/bmj.e977]).
"The evidence accumulated so far implies that use of haloperidol in this vulnerable population cannot be justified because of the excess harm," wrote Dr. Huybrechts of Brigham and Women’s Hospital, Boston.
The findings emphasize the dangers of using drugs as a first-line therapy for elderly patients with behavioral problems. "If the clinician faces a situation in which the use of these drugs seem inevitable, our findings underscore the importance of always prescribing the lowest possible dose and of closely monitoring patients, especially shortly after the start of treatment."
Dr. Huybrechts and her coauthors reviewed medication and mortality data on 75,445 nursing home residents who were new users of antipsychotic medications from 2001-2005. About 60% of the group had some form of dementia. Other psychiatric diagnoses included depression, anxiety, delirium, and psychotic disorders. All of the patients were aged 65 years or older. Many also had physical comorbidities, including a history of heart attack, cardiac arrhythmias, ischemic heart disease, hypertension, cerebrovascular disease, diabetes, and Parkinson’s disease.
The study examined the use of haloperidol, aripiprazole, olanzapine, risperidone, and ziprasidone. Other medications, like thioridazine and chlorpromazine, were excluded from the analysis because they were infrequently prescribed in the group.
Most of the patients were censored from the record before 180 days. A discontinuation of treatment was the most common reason (57%), but some patients stopped because of a hospital admission (17%), a change in medication (13%), or death (12%).
There were 6,598 deaths (9% of the study cohort) during the first 180 days of antipsychotic treatment – a rate of 37 per person per year of treatment. About half of these were caused by circulatory disorders (49%). The other large causes were cerebrovascular diseases (10%) and respiratory disorders (15%); the remaining 26% of deaths were tied to illnesses unspecified in the analysis.
In a regression analysis, those taking haloperidol were twice as likely to die from any cause as those taking risperidone (hazard ratio, 2.07). The risk was strongest in the first 40 days of treatment (HR, 2.34) and declined thereafter. By 79 days, haloperidol patients were 32% more likely to have died; from 80-180 days, the increased risk was 46%.
Conversely, patients taking quetiapine were significantly less likely to die (HR, 0.81), and that risk remained fairly contrast throughout the 180-day study period.
No significant or clinically meaningful mortality trends were associated with the use of aripiprazole, olanzapine, risperidone, or ziprasidone.
The investigators found no significant interactions between the drug administered and the cause of death. "The increased risk of death with haloperidol and the decreased risk with quetiapine were observed for all causes examined," they said. However, the analysis suggested that olanzapine might be associated with a decrease in the risk of cerebrovascular death. There were insufficient data to draw any conclusions about mortality type in patients taking aripiprazole or ziprasidone.
Every drug except quetiapine showed a dose-response relationship with all-cause mortality. The relationship was most pronounced for haloperidol, with an 84% increased risk in those taking a high dose, and a 40% increased risk for those taking a medium dose, compared with those taking a low dose.
The study emphasizes the need to use alternative behavioral management tools for as long as possible in nursing home patients, the authors said.
"[It] reinforces the important risks associated with the use of these drugs and underscores the need to try alternative means of dealing with behavioral problems in older patients with dementia."
The Food and Drug Administration, and the Agency for Healthcare Research and Quality funded the study. None of the authors reported any financial conflicts.
FROM BMJ
Major Finding: Compared with risperidone, haloperidol increased the risk of death by 200% among a large cohort of elderly nursing home residents. Conversely, quetiapine was associated with a 20% decrease in the risk of death.
Data Source: A database review of 75,445 new users of the antipsychotic drugs haloperidol, aripiprazole, olanzapine, quetiapine, risperidone, and ziprasidone.
Disclosures: The Food and Drug Administration, and the Agency for Healthcare Research and Quality funded the study. None of the authors reported any financial conflicts.
Filgrastim Fails to Improve Ischemic Stroke Outcomes
NEW ORLEANS –A granulocyte colony-stimulating drug did not protect brain tissue or improve functional outcomes after ischemic stroke.
Researchers had high hopes for filgrastim, which is approved in Europe for chemotherapy-induced neutropenia. In vivo and in vitro studies indicated that filgrastim also had antiapoptotic properties, and stimulated arteriogenesis, neurogenesis and neurite outgrowth, said Dr. E. Bernd Ringelstein, a professor of neurology at the University Hospital of Münster (Germany).
Although the drug was safe and well tolerated, the results confirm the principal problem that has plagued the development of stroke drugs for years: They don’t make the leap from preclinical promise to clinical efficacy, he said. The results seem to echo a research saying from the 1980s: "The outlook for stroke therapy is excellent – if you’re a rat."
The negative findings were noted in the phase II AXIS 2 trial, which randomized 328 patients with acute ischemic stroke to placebo or filgrastim (AX200) at 135 mcg/kg over 72 hours.* Study participants had to have a middle cerebral artery infarct of at least 15 cc, and be younger than 86 years old. The intent-to-treat analysis included 323 patients, 64% of whom had received tissue plasminogen activator (TPA). There was a mean of 7 hours from onset of symptoms to administration of the study drug or placebo.
The study’s primary end point was 90-day function as measured by the modified Rankin Scale. Secondary end points included 30-day lesion volume and 90-day NIHSS (National Institutes of Health Stroke Scale) score and mortality. Imaging was graded by a core lab that was blinded to the treatment.
At 90 days, the modified Rankin Scale scores were 3.26 in the treatment group and 3.06 in the placebo group. The 90-day NIHSS scores were also similar (8.8 and 8.4, respectively). Pretreatment with TPA did not significantly affect outcomes in either group. Survival at 90 days was also not significantly different, at 78% in the treatment group and 82% in the placebo group.
The initial infarct volume was an average of 46 cc. The final lesion volumes were 59 cc in the treatment group and 66 cc in the placebo group, which were not significantly different.
The most common adverse events were cardiac, endocrine, and gastrointestinal disorders, none of which were significantly more common in the treated group. AX200 did significantly increase white blood cell and monocyte counts, and slightly decreased platelets relative to the placebo, as expected, Dr. Ringelstein said.
"In summary, AX200 was a very promising drug with a comprehensive activity package. It was safe and well tolerated, but it did not provide any benefit over placebo. At the moment, we do not know the reasons for this failure, but we are exploring this with additional analyses."
The study was sponsored by SYGNIS Biosience. Dr. Ringelstein disclosed that he was a member of the AXIS study steering board and a consultant and advisory board member for the company.
*Correction, 2/21/2012: An earlier version of this story misstated the phase of this trial.
NEW ORLEANS –A granulocyte colony-stimulating drug did not protect brain tissue or improve functional outcomes after ischemic stroke.
Researchers had high hopes for filgrastim, which is approved in Europe for chemotherapy-induced neutropenia. In vivo and in vitro studies indicated that filgrastim also had antiapoptotic properties, and stimulated arteriogenesis, neurogenesis and neurite outgrowth, said Dr. E. Bernd Ringelstein, a professor of neurology at the University Hospital of Münster (Germany).
Although the drug was safe and well tolerated, the results confirm the principal problem that has plagued the development of stroke drugs for years: They don’t make the leap from preclinical promise to clinical efficacy, he said. The results seem to echo a research saying from the 1980s: "The outlook for stroke therapy is excellent – if you’re a rat."
The negative findings were noted in the phase II AXIS 2 trial, which randomized 328 patients with acute ischemic stroke to placebo or filgrastim (AX200) at 135 mcg/kg over 72 hours.* Study participants had to have a middle cerebral artery infarct of at least 15 cc, and be younger than 86 years old. The intent-to-treat analysis included 323 patients, 64% of whom had received tissue plasminogen activator (TPA). There was a mean of 7 hours from onset of symptoms to administration of the study drug or placebo.
The study’s primary end point was 90-day function as measured by the modified Rankin Scale. Secondary end points included 30-day lesion volume and 90-day NIHSS (National Institutes of Health Stroke Scale) score and mortality. Imaging was graded by a core lab that was blinded to the treatment.
At 90 days, the modified Rankin Scale scores were 3.26 in the treatment group and 3.06 in the placebo group. The 90-day NIHSS scores were also similar (8.8 and 8.4, respectively). Pretreatment with TPA did not significantly affect outcomes in either group. Survival at 90 days was also not significantly different, at 78% in the treatment group and 82% in the placebo group.
The initial infarct volume was an average of 46 cc. The final lesion volumes were 59 cc in the treatment group and 66 cc in the placebo group, which were not significantly different.
The most common adverse events were cardiac, endocrine, and gastrointestinal disorders, none of which were significantly more common in the treated group. AX200 did significantly increase white blood cell and monocyte counts, and slightly decreased platelets relative to the placebo, as expected, Dr. Ringelstein said.
"In summary, AX200 was a very promising drug with a comprehensive activity package. It was safe and well tolerated, but it did not provide any benefit over placebo. At the moment, we do not know the reasons for this failure, but we are exploring this with additional analyses."
The study was sponsored by SYGNIS Biosience. Dr. Ringelstein disclosed that he was a member of the AXIS study steering board and a consultant and advisory board member for the company.
*Correction, 2/21/2012: An earlier version of this story misstated the phase of this trial.
NEW ORLEANS –A granulocyte colony-stimulating drug did not protect brain tissue or improve functional outcomes after ischemic stroke.
Researchers had high hopes for filgrastim, which is approved in Europe for chemotherapy-induced neutropenia. In vivo and in vitro studies indicated that filgrastim also had antiapoptotic properties, and stimulated arteriogenesis, neurogenesis and neurite outgrowth, said Dr. E. Bernd Ringelstein, a professor of neurology at the University Hospital of Münster (Germany).
Although the drug was safe and well tolerated, the results confirm the principal problem that has plagued the development of stroke drugs for years: They don’t make the leap from preclinical promise to clinical efficacy, he said. The results seem to echo a research saying from the 1980s: "The outlook for stroke therapy is excellent – if you’re a rat."
The negative findings were noted in the phase II AXIS 2 trial, which randomized 328 patients with acute ischemic stroke to placebo or filgrastim (AX200) at 135 mcg/kg over 72 hours.* Study participants had to have a middle cerebral artery infarct of at least 15 cc, and be younger than 86 years old. The intent-to-treat analysis included 323 patients, 64% of whom had received tissue plasminogen activator (TPA). There was a mean of 7 hours from onset of symptoms to administration of the study drug or placebo.
The study’s primary end point was 90-day function as measured by the modified Rankin Scale. Secondary end points included 30-day lesion volume and 90-day NIHSS (National Institutes of Health Stroke Scale) score and mortality. Imaging was graded by a core lab that was blinded to the treatment.
At 90 days, the modified Rankin Scale scores were 3.26 in the treatment group and 3.06 in the placebo group. The 90-day NIHSS scores were also similar (8.8 and 8.4, respectively). Pretreatment with TPA did not significantly affect outcomes in either group. Survival at 90 days was also not significantly different, at 78% in the treatment group and 82% in the placebo group.
The initial infarct volume was an average of 46 cc. The final lesion volumes were 59 cc in the treatment group and 66 cc in the placebo group, which were not significantly different.
The most common adverse events were cardiac, endocrine, and gastrointestinal disorders, none of which were significantly more common in the treated group. AX200 did significantly increase white blood cell and monocyte counts, and slightly decreased platelets relative to the placebo, as expected, Dr. Ringelstein said.
"In summary, AX200 was a very promising drug with a comprehensive activity package. It was safe and well tolerated, but it did not provide any benefit over placebo. At the moment, we do not know the reasons for this failure, but we are exploring this with additional analyses."
The study was sponsored by SYGNIS Biosience. Dr. Ringelstein disclosed that he was a member of the AXIS study steering board and a consultant and advisory board member for the company.
*Correction, 2/21/2012: An earlier version of this story misstated the phase of this trial.
FROM AN INTERNATIONAL STROKE CONFERENCE
Major Finding: At 90 days, the modified Rankin Scale scores were 3.26 in the treatment group and 3.06 in the placebo group. The 90-day NIHSS scores were 8.8 and 8.4, respectively. The final lesion volumes were 59 cc in the treatment group and 66 cc in the placebo group.
Data Source: AXIS 2 was a phase II, randomized, placebo-controlled trial with 323 patients in an intent-to-treat analysis.
Disclosures: The study was sponsored by SYGNIS Biosience. Dr. Ringelstein disclosed that he was a member of the AXIS study steering board and a consultant and advisory board member for the company.
Study Examines Safety of Stroke Treatment in Children
Thrombolytic drugs appear to be as safe for children with acute ischemic stroke as they are for adults, based on an analysis of more than 9,000 patients in the Kids’ Inpatient Database.
Overall, 4% of the children in the database who got the clot-busting drugs had a brain hemorrhage, which was not significantly different than the 6% rate seen in adults with similar treatment, according to Dr. Amer Alshekhlee, a neurology fellow at the St. Louis University.
The Kids Inpatient Database contains data on 3 million pediatric hospital stays from 1997 through 2009. Dr. Alshekhlee identified 9,367 children who were treated in hospitals for acute ischemic stroke. Only 75 of those – less than 1% – received thrombolysis.
The rate was not unexpectedly low, Dr. Alshekhlee, said in an interview. In fact, he said, some reviews have suggested avoiding thrombolytic treatment for children altogether.
"Children are not getting the treatment because the treatment is not recommended for them. The rate of using thrombolysis is also low in adults, about 2%, but this rate in children is even lower."
In the database, treated children were significantly older than the untreated (mean 13 years vs. 8 years).There were no significant demographic differences in sex, race, or family income.
An unadjusted analysis showed that treated children had a significantly greater incidence of both intracranial hemorrhage (4.0% vs. 0.38 %) and intracranial hemorrhage and in-hospital mortality combined (12% vs. 6%).
In a fully adjusted model, children who received thrombolysis had significantly greater odds of developing a brain bleed than were untreated children (odds ratio, 4.3), and those who did have a bleed had almost three and a half times greater odds of dying (OR, 3.4). However, thrombolysis itself was not associated with any significant increase in mortality.
Dr. Alshekhlee’s review of the database didn’t examine functional outcomes among the children who received thrombolytic medications.
Although the data used in the review are several years old, Dr. Alshekhlee said they probably accurately reflect today’s thrombolytic treatment in children.
From a practical standpoint, the study shows that the drugs are relatively safe for children, but can’t be construed as a practice changer, coauthor Dr. Salvador Cruz-Flores said in an interview.
"In the absence of any other alternative therapy for kids with severe stroke, perhaps physicians facing the situation should consider using it, but that will require informing parents of the uncertainty of its efficacy in the population and acknowledging the gap in knowledge we have," said Dr. Cruz-Flores, director of the Souers Stroke Institute at Saint Louis University.
The investigators are scheduled to present the full results of the study in late April at the annual meeting of the American Academy of Neurology.
The review was conducted without outside funding. Neither investigator had any relevant financial disclosures.
Thrombolytic drugs appear to be as safe for children with acute ischemic stroke as they are for adults, based on an analysis of more than 9,000 patients in the Kids’ Inpatient Database.
Overall, 4% of the children in the database who got the clot-busting drugs had a brain hemorrhage, which was not significantly different than the 6% rate seen in adults with similar treatment, according to Dr. Amer Alshekhlee, a neurology fellow at the St. Louis University.
The Kids Inpatient Database contains data on 3 million pediatric hospital stays from 1997 through 2009. Dr. Alshekhlee identified 9,367 children who were treated in hospitals for acute ischemic stroke. Only 75 of those – less than 1% – received thrombolysis.
The rate was not unexpectedly low, Dr. Alshekhlee, said in an interview. In fact, he said, some reviews have suggested avoiding thrombolytic treatment for children altogether.
"Children are not getting the treatment because the treatment is not recommended for them. The rate of using thrombolysis is also low in adults, about 2%, but this rate in children is even lower."
In the database, treated children were significantly older than the untreated (mean 13 years vs. 8 years).There were no significant demographic differences in sex, race, or family income.
An unadjusted analysis showed that treated children had a significantly greater incidence of both intracranial hemorrhage (4.0% vs. 0.38 %) and intracranial hemorrhage and in-hospital mortality combined (12% vs. 6%).
In a fully adjusted model, children who received thrombolysis had significantly greater odds of developing a brain bleed than were untreated children (odds ratio, 4.3), and those who did have a bleed had almost three and a half times greater odds of dying (OR, 3.4). However, thrombolysis itself was not associated with any significant increase in mortality.
Dr. Alshekhlee’s review of the database didn’t examine functional outcomes among the children who received thrombolytic medications.
Although the data used in the review are several years old, Dr. Alshekhlee said they probably accurately reflect today’s thrombolytic treatment in children.
From a practical standpoint, the study shows that the drugs are relatively safe for children, but can’t be construed as a practice changer, coauthor Dr. Salvador Cruz-Flores said in an interview.
"In the absence of any other alternative therapy for kids with severe stroke, perhaps physicians facing the situation should consider using it, but that will require informing parents of the uncertainty of its efficacy in the population and acknowledging the gap in knowledge we have," said Dr. Cruz-Flores, director of the Souers Stroke Institute at Saint Louis University.
The investigators are scheduled to present the full results of the study in late April at the annual meeting of the American Academy of Neurology.
The review was conducted without outside funding. Neither investigator had any relevant financial disclosures.
Thrombolytic drugs appear to be as safe for children with acute ischemic stroke as they are for adults, based on an analysis of more than 9,000 patients in the Kids’ Inpatient Database.
Overall, 4% of the children in the database who got the clot-busting drugs had a brain hemorrhage, which was not significantly different than the 6% rate seen in adults with similar treatment, according to Dr. Amer Alshekhlee, a neurology fellow at the St. Louis University.
The Kids Inpatient Database contains data on 3 million pediatric hospital stays from 1997 through 2009. Dr. Alshekhlee identified 9,367 children who were treated in hospitals for acute ischemic stroke. Only 75 of those – less than 1% – received thrombolysis.
The rate was not unexpectedly low, Dr. Alshekhlee, said in an interview. In fact, he said, some reviews have suggested avoiding thrombolytic treatment for children altogether.
"Children are not getting the treatment because the treatment is not recommended for them. The rate of using thrombolysis is also low in adults, about 2%, but this rate in children is even lower."
In the database, treated children were significantly older than the untreated (mean 13 years vs. 8 years).There were no significant demographic differences in sex, race, or family income.
An unadjusted analysis showed that treated children had a significantly greater incidence of both intracranial hemorrhage (4.0% vs. 0.38 %) and intracranial hemorrhage and in-hospital mortality combined (12% vs. 6%).
In a fully adjusted model, children who received thrombolysis had significantly greater odds of developing a brain bleed than were untreated children (odds ratio, 4.3), and those who did have a bleed had almost three and a half times greater odds of dying (OR, 3.4). However, thrombolysis itself was not associated with any significant increase in mortality.
Dr. Alshekhlee’s review of the database didn’t examine functional outcomes among the children who received thrombolytic medications.
Although the data used in the review are several years old, Dr. Alshekhlee said they probably accurately reflect today’s thrombolytic treatment in children.
From a practical standpoint, the study shows that the drugs are relatively safe for children, but can’t be construed as a practice changer, coauthor Dr. Salvador Cruz-Flores said in an interview.
"In the absence of any other alternative therapy for kids with severe stroke, perhaps physicians facing the situation should consider using it, but that will require informing parents of the uncertainty of its efficacy in the population and acknowledging the gap in knowledge we have," said Dr. Cruz-Flores, director of the Souers Stroke Institute at Saint Louis University.
The investigators are scheduled to present the full results of the study in late April at the annual meeting of the American Academy of Neurology.
The review was conducted without outside funding. Neither investigator had any relevant financial disclosures.
Major Finding: Children with acute ischemic stroke who received thrombolytic therapy had a 4% rate of intracranial hemorrhage, compared with 0.38% in untreated children.
Data Source: Data were extracted from the national Kids Inpatient Database and included more than 9,000 children with ischemic stroke
Disclosures: The research was conducted without outside funding. Neither researcher had any relevant financial disclosures.
Weight Lifting Improves Physical Function in Parkinson’s
A 2-year weight-lifting program improved physical symptoms of Parkinson’s disease – including tremor – and seemed to slow their progression.
The progressive resistance routine, which alternated between increasing strength and speed in weight-lifting, was significantly more effective than was an often-recommended program of strengthening, flexibility, and balance, according to Daniel Corcos, Ph.D., lead author of the comparative study. By the end of the study period, patients who were lifting weights maintained a significant level of improvement, but scores of the other group had fallen back to their baseline levels.
While weight lifting obviously benefits a patient by muscular strengthening, there seems to be a neuronal component as well, Dr. Corcos said in an interview.
"There is no question that part of the changes are related to what goes on in the brain. Every time you make a muscle contract, you involve the motor cortex and basal ganglia," he said. This repetitive experience appears to alter cortical excitability, improving the connection between brain and muscles, which deteriorates in Parkinson’s, said Dr. Corcos, a kinesiologist and professor of neurologic sciences at the University of Illinois, Chicago.
The investigation randomized 48 people with Parkinson’s disease to the progressive resistance program or to Fitness Counts, a program created by the National Parkinson’s Disease Foundation. Fitness Counts is performed without weights and includes 12 stretching exercises, 7 strengthening exercises, and a set of balancing exercises.
Patients in the weight-training program exercised upper and lower body muscle groups with ever-increasing weight. The initial step determined the heaviest weight a patient could tolerate. Resistance was set at 30%-40% of that maximum weight for the upper body exercises and 50%-60-% of the maximum for the lower body exercises. Weight was increased by at least 5% as soon as the exercise became easier. Every 8 weeks, the program alternated between strength training alone and the combination of strength and speed training, with emphasis on how quickly each repetition could be performed with good form.
The researchers expected each patient to complete 208 sessions by 24 months – usually 2 sessions each week. If they missed a two sessions in a row, the exercise study coordinator contacted the patient. The primary outcome was change on the Unified Parkinson’s Disease Rating Scale III (UPDRS-III) score off medication from baseline to the end of the study.
The patients’ mean age at study entry was 59 years. Most (38) completed the entire 2-year program.
Both the strength training and overall fitness programs had significantly reduced the UPDRS-III score at 6 months (by 6.42 and 5.38 points, respectively). But by the end of 2 years, the weight training group had maintained its improvement (7.3 points), while the fitness group’s score had dropped back to its baseline levels.
The study shows that patients with Parkinson’s can adhere to a sustained exercise regimen and reap valuable benefits from it, Dr. Corcos said. The average Parkinson’s patient experiences about a 30% decrease in normal activity, which provokes muscle deterioration even apart from disease progression. "Think about it. If you, a healthy person, stopped doing anything and laid up in bed for a year, you would still end up in a sorry state," Dr. Corcos said.
Depression probably contributes to reduced activity along with muscular problems. "Depression is part of Parkinson’s and although not every patient has it, it can be quite disabling. As it becomes harder and harder to move, people lose the motivation [to exercise]."
Patients also tend to put exercise at the bottom of their personal "to do" list, just as many healthy people do.
"As the medications lose effectiveness, patients have fewer periods during which they can get things done," Dr. Corcos said. "When it’s working well, they have so many things to do that they just don’t find the time for exercise. I hope this objective evidence may motivate patients to realize that exercise should be a top priority."
His is not the only study expanding the knowledge base in this area.
Fuzhong Li, Ph.D., of the Oregon Research Institute, Eugene, and his colleagues recently published the results of a randomized trial that explored the effect of a modified tai chi program on physical function in Parkinson’s patients. The study randomized 195 patients to tai chi, resistance training, or stretching for 24 weeks. The tai chi group experienced a significantly greater improvement in the UPDRS-III score than did the stretching group (but not the resistance training group), as well as significantly fewer falls than either of the other groups. They retained these benefits at 3 months after the intervention (N. Engl. J. Med. 2012;366:511-9).
Dr. Corcos is scheduled to present the full results of his study in late April at the annual meeting of the America Academy of Neurology.
The National Institutes of Health funded the study. Dr. Corcos had no financial disclosures.
A 2-year weight-lifting program improved physical symptoms of Parkinson’s disease – including tremor – and seemed to slow their progression.
The progressive resistance routine, which alternated between increasing strength and speed in weight-lifting, was significantly more effective than was an often-recommended program of strengthening, flexibility, and balance, according to Daniel Corcos, Ph.D., lead author of the comparative study. By the end of the study period, patients who were lifting weights maintained a significant level of improvement, but scores of the other group had fallen back to their baseline levels.
While weight lifting obviously benefits a patient by muscular strengthening, there seems to be a neuronal component as well, Dr. Corcos said in an interview.
"There is no question that part of the changes are related to what goes on in the brain. Every time you make a muscle contract, you involve the motor cortex and basal ganglia," he said. This repetitive experience appears to alter cortical excitability, improving the connection between brain and muscles, which deteriorates in Parkinson’s, said Dr. Corcos, a kinesiologist and professor of neurologic sciences at the University of Illinois, Chicago.
The investigation randomized 48 people with Parkinson’s disease to the progressive resistance program or to Fitness Counts, a program created by the National Parkinson’s Disease Foundation. Fitness Counts is performed without weights and includes 12 stretching exercises, 7 strengthening exercises, and a set of balancing exercises.
Patients in the weight-training program exercised upper and lower body muscle groups with ever-increasing weight. The initial step determined the heaviest weight a patient could tolerate. Resistance was set at 30%-40% of that maximum weight for the upper body exercises and 50%-60-% of the maximum for the lower body exercises. Weight was increased by at least 5% as soon as the exercise became easier. Every 8 weeks, the program alternated between strength training alone and the combination of strength and speed training, with emphasis on how quickly each repetition could be performed with good form.
The researchers expected each patient to complete 208 sessions by 24 months – usually 2 sessions each week. If they missed a two sessions in a row, the exercise study coordinator contacted the patient. The primary outcome was change on the Unified Parkinson’s Disease Rating Scale III (UPDRS-III) score off medication from baseline to the end of the study.
The patients’ mean age at study entry was 59 years. Most (38) completed the entire 2-year program.
Both the strength training and overall fitness programs had significantly reduced the UPDRS-III score at 6 months (by 6.42 and 5.38 points, respectively). But by the end of 2 years, the weight training group had maintained its improvement (7.3 points), while the fitness group’s score had dropped back to its baseline levels.
The study shows that patients with Parkinson’s can adhere to a sustained exercise regimen and reap valuable benefits from it, Dr. Corcos said. The average Parkinson’s patient experiences about a 30% decrease in normal activity, which provokes muscle deterioration even apart from disease progression. "Think about it. If you, a healthy person, stopped doing anything and laid up in bed for a year, you would still end up in a sorry state," Dr. Corcos said.
Depression probably contributes to reduced activity along with muscular problems. "Depression is part of Parkinson’s and although not every patient has it, it can be quite disabling. As it becomes harder and harder to move, people lose the motivation [to exercise]."
Patients also tend to put exercise at the bottom of their personal "to do" list, just as many healthy people do.
"As the medications lose effectiveness, patients have fewer periods during which they can get things done," Dr. Corcos said. "When it’s working well, they have so many things to do that they just don’t find the time for exercise. I hope this objective evidence may motivate patients to realize that exercise should be a top priority."
His is not the only study expanding the knowledge base in this area.
Fuzhong Li, Ph.D., of the Oregon Research Institute, Eugene, and his colleagues recently published the results of a randomized trial that explored the effect of a modified tai chi program on physical function in Parkinson’s patients. The study randomized 195 patients to tai chi, resistance training, or stretching for 24 weeks. The tai chi group experienced a significantly greater improvement in the UPDRS-III score than did the stretching group (but not the resistance training group), as well as significantly fewer falls than either of the other groups. They retained these benefits at 3 months after the intervention (N. Engl. J. Med. 2012;366:511-9).
Dr. Corcos is scheduled to present the full results of his study in late April at the annual meeting of the America Academy of Neurology.
The National Institutes of Health funded the study. Dr. Corcos had no financial disclosures.
A 2-year weight-lifting program improved physical symptoms of Parkinson’s disease – including tremor – and seemed to slow their progression.
The progressive resistance routine, which alternated between increasing strength and speed in weight-lifting, was significantly more effective than was an often-recommended program of strengthening, flexibility, and balance, according to Daniel Corcos, Ph.D., lead author of the comparative study. By the end of the study period, patients who were lifting weights maintained a significant level of improvement, but scores of the other group had fallen back to their baseline levels.
While weight lifting obviously benefits a patient by muscular strengthening, there seems to be a neuronal component as well, Dr. Corcos said in an interview.
"There is no question that part of the changes are related to what goes on in the brain. Every time you make a muscle contract, you involve the motor cortex and basal ganglia," he said. This repetitive experience appears to alter cortical excitability, improving the connection between brain and muscles, which deteriorates in Parkinson’s, said Dr. Corcos, a kinesiologist and professor of neurologic sciences at the University of Illinois, Chicago.
The investigation randomized 48 people with Parkinson’s disease to the progressive resistance program or to Fitness Counts, a program created by the National Parkinson’s Disease Foundation. Fitness Counts is performed without weights and includes 12 stretching exercises, 7 strengthening exercises, and a set of balancing exercises.
Patients in the weight-training program exercised upper and lower body muscle groups with ever-increasing weight. The initial step determined the heaviest weight a patient could tolerate. Resistance was set at 30%-40% of that maximum weight for the upper body exercises and 50%-60-% of the maximum for the lower body exercises. Weight was increased by at least 5% as soon as the exercise became easier. Every 8 weeks, the program alternated between strength training alone and the combination of strength and speed training, with emphasis on how quickly each repetition could be performed with good form.
The researchers expected each patient to complete 208 sessions by 24 months – usually 2 sessions each week. If they missed a two sessions in a row, the exercise study coordinator contacted the patient. The primary outcome was change on the Unified Parkinson’s Disease Rating Scale III (UPDRS-III) score off medication from baseline to the end of the study.
The patients’ mean age at study entry was 59 years. Most (38) completed the entire 2-year program.
Both the strength training and overall fitness programs had significantly reduced the UPDRS-III score at 6 months (by 6.42 and 5.38 points, respectively). But by the end of 2 years, the weight training group had maintained its improvement (7.3 points), while the fitness group’s score had dropped back to its baseline levels.
The study shows that patients with Parkinson’s can adhere to a sustained exercise regimen and reap valuable benefits from it, Dr. Corcos said. The average Parkinson’s patient experiences about a 30% decrease in normal activity, which provokes muscle deterioration even apart from disease progression. "Think about it. If you, a healthy person, stopped doing anything and laid up in bed for a year, you would still end up in a sorry state," Dr. Corcos said.
Depression probably contributes to reduced activity along with muscular problems. "Depression is part of Parkinson’s and although not every patient has it, it can be quite disabling. As it becomes harder and harder to move, people lose the motivation [to exercise]."
Patients also tend to put exercise at the bottom of their personal "to do" list, just as many healthy people do.
"As the medications lose effectiveness, patients have fewer periods during which they can get things done," Dr. Corcos said. "When it’s working well, they have so many things to do that they just don’t find the time for exercise. I hope this objective evidence may motivate patients to realize that exercise should be a top priority."
His is not the only study expanding the knowledge base in this area.
Fuzhong Li, Ph.D., of the Oregon Research Institute, Eugene, and his colleagues recently published the results of a randomized trial that explored the effect of a modified tai chi program on physical function in Parkinson’s patients. The study randomized 195 patients to tai chi, resistance training, or stretching for 24 weeks. The tai chi group experienced a significantly greater improvement in the UPDRS-III score than did the stretching group (but not the resistance training group), as well as significantly fewer falls than either of the other groups. They retained these benefits at 3 months after the intervention (N. Engl. J. Med. 2012;366:511-9).
Dr. Corcos is scheduled to present the full results of his study in late April at the annual meeting of the America Academy of Neurology.
The National Institutes of Health funded the study. Dr. Corcos had no financial disclosures.
Major Finding: Parkinson’s disease patients who lifted weights for 2 years decreased their UPDRS-III score by more than 7 points, compared with no change from baseline among patients who engaged in an overall fitness program.
Data Source: The study randomized 48 patients to a progressive resistance weight program or to the Fitness Counts program developed by the National Parkinson’s Disease Foundation
Disclosures: The National Institutes of Health funded the study. Dr. Corcos had no financial disclosures.
Scores Help Identify Stroke Patients at Risk of Brain Bleed
NEW ORLEANS – Two new risk assessment scores predicted the likelihood of a symptomatic intracranial hemorrhage after ischemic stroke treatment with more than 70% accuracy.
Predictive rules such as these are important because there is still no large-scale prospective study that clearly identifies which patients with ischemic stroke are more likely to develop a brain bleed after treatment with intravenous tissue plasminogen activator (TPA), Dr. Bijoy Menon said at the International Stroke Conference.
Dr. Menon, a clinical stroke fellow at the University of Calgary (Alta.), and his colleagues developed a 101-point score based on data extracted from the Get With The Guidelines stroke cohort. The cohort consisted of 10,242 patients with ischemic stroke who received TPA within 3 hours of the onset of stroke symptoms. A derivation cohort comprised 70% of the group; the rule was then validated in the remaining 30%.
The cohort’s mean age was 69 years. All patients experienced a moderate to severe ischemic stroke, with a mean National Institutes of Health Stroke Scale (NIHSS) score of 11. They received TPA at a mean of 1.35 hours after symptom onset.
About 5% of the group (496 patients) experienced a symptomatic intracranial hemorrhage (ICH), which the investigators defined as neurologic worsening within 36 hours of TPA administration.
In a multivariate regression model, Dr. Menon and his coinvestigators found six patient characteristics that were significantly associated with a brain bleed. They assigned each of these characteristics a point spread based on the range of measurements:
• Age. From age 60 years or younger (8 points) to older than 80 (17 points).
• NIHSS score. From 1-5 (25 points) to 20 and over (42 points).
• Systolic blood pressure. From less than 120 mm Hg (10 points) to 180 mm Hg or higher (21 points).
• Blood glucose level. From less than 100 mg/dL (2 points) to 150 mg/dL or more (8 points).
• Ethnicity. Asian, 9 points; all others, 0 points.
• Gender. Male, 4 points; female, 0 points.
Diabetes and a history of stroke were significantly associated with ICH in the initial analysis, but the P values were nonsignificant in the multivariate analysis, Dr. Menon noted.
"We also did not find any significantly increased risk associated with warfarin use or with the baseline international normalized ratio," he said at the meeting, sponsored by the American Heart Association.
The score accurately predicted ICH in 71% of the validation cohort, "comparable to most of the other scoring methods out there."
While the score is "well validated and evidence based," neither it nor any other single measure should be the sole factor in determining ischemic stroke treatment, Dr. Menon emphasized. "This is very important. It should not be used to infer which patients would benefit most or least from IV TPA. This is a cohort study, and because treatment was at the discretion of the individual physicians, there may be a selection bias present in it."
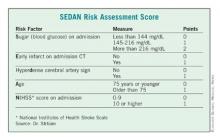
Dr. Daniel Strbian of the University of Helsinki presented a second score, dubbed SEDAN. Dr. Strbian and his colleagues developed their score based on a cohort of 972 ischemic stroke patients who developed an imaging-proven brain bleed after IV TPA treatment.
In a multivariate analysis, he and his associates found five factors that were independently associated with a significantly increased risk of symptomatic ICH: blood glucose level on admission, computed tomography (CT) imaging positive for ischemic stroke, cerebral artery hyperdensity, age, and NIHSS score.
Points were assigned based on the relative risk (RR) associated with each factor – the higher the total score, the greater the risk of symptomatic ICH. Each of the five risk-point categories generated a significant risk ratio:1 point, RR 0.19; 2 points, RR 0.40; 3 points, RR 1.85; 4 points, RR 3.7; 5 points, RR 5.6.
The score was validated in two additional groups totaling about 2,000 patients, resulting in an overall accuracy of about 77%.
"If we have a patient with a very high score, there is a very bad prognosis with a high chance of death or institutionalization at 3 months," Dr. Strbian said. "In this case, we know that the patient may die if we do nothing, but perhaps the SEDAN [score] can help us determine if an endovascular approach might be better than thrombolysis."
The SEDAN score is simple, easy, and quick to calculate, he said. But it’s only part of the treatment decision process.
"We can’t treat based on this score alone. If the patient fulfills the indications for TPA, it’s up to the physician and the patient to decide whether they are willing to accept the risk of an intracranial hemorrhage."
Dr. Menon and Dr. Strbian reported having no financial conflicts.
NEW ORLEANS – Two new risk assessment scores predicted the likelihood of a symptomatic intracranial hemorrhage after ischemic stroke treatment with more than 70% accuracy.
Predictive rules such as these are important because there is still no large-scale prospective study that clearly identifies which patients with ischemic stroke are more likely to develop a brain bleed after treatment with intravenous tissue plasminogen activator (TPA), Dr. Bijoy Menon said at the International Stroke Conference.
Dr. Menon, a clinical stroke fellow at the University of Calgary (Alta.), and his colleagues developed a 101-point score based on data extracted from the Get With The Guidelines stroke cohort. The cohort consisted of 10,242 patients with ischemic stroke who received TPA within 3 hours of the onset of stroke symptoms. A derivation cohort comprised 70% of the group; the rule was then validated in the remaining 30%.
The cohort’s mean age was 69 years. All patients experienced a moderate to severe ischemic stroke, with a mean National Institutes of Health Stroke Scale (NIHSS) score of 11. They received TPA at a mean of 1.35 hours after symptom onset.
About 5% of the group (496 patients) experienced a symptomatic intracranial hemorrhage (ICH), which the investigators defined as neurologic worsening within 36 hours of TPA administration.
In a multivariate regression model, Dr. Menon and his coinvestigators found six patient characteristics that were significantly associated with a brain bleed. They assigned each of these characteristics a point spread based on the range of measurements:
• Age. From age 60 years or younger (8 points) to older than 80 (17 points).
• NIHSS score. From 1-5 (25 points) to 20 and over (42 points).
• Systolic blood pressure. From less than 120 mm Hg (10 points) to 180 mm Hg or higher (21 points).
• Blood glucose level. From less than 100 mg/dL (2 points) to 150 mg/dL or more (8 points).
• Ethnicity. Asian, 9 points; all others, 0 points.
• Gender. Male, 4 points; female, 0 points.
Diabetes and a history of stroke were significantly associated with ICH in the initial analysis, but the P values were nonsignificant in the multivariate analysis, Dr. Menon noted.
"We also did not find any significantly increased risk associated with warfarin use or with the baseline international normalized ratio," he said at the meeting, sponsored by the American Heart Association.
The score accurately predicted ICH in 71% of the validation cohort, "comparable to most of the other scoring methods out there."
While the score is "well validated and evidence based," neither it nor any other single measure should be the sole factor in determining ischemic stroke treatment, Dr. Menon emphasized. "This is very important. It should not be used to infer which patients would benefit most or least from IV TPA. This is a cohort study, and because treatment was at the discretion of the individual physicians, there may be a selection bias present in it."
Dr. Daniel Strbian of the University of Helsinki presented a second score, dubbed SEDAN. Dr. Strbian and his colleagues developed their score based on a cohort of 972 ischemic stroke patients who developed an imaging-proven brain bleed after IV TPA treatment.
In a multivariate analysis, he and his associates found five factors that were independently associated with a significantly increased risk of symptomatic ICH: blood glucose level on admission, computed tomography (CT) imaging positive for ischemic stroke, cerebral artery hyperdensity, age, and NIHSS score.
Points were assigned based on the relative risk (RR) associated with each factor – the higher the total score, the greater the risk of symptomatic ICH. Each of the five risk-point categories generated a significant risk ratio:1 point, RR 0.19; 2 points, RR 0.40; 3 points, RR 1.85; 4 points, RR 3.7; 5 points, RR 5.6.
The score was validated in two additional groups totaling about 2,000 patients, resulting in an overall accuracy of about 77%.
"If we have a patient with a very high score, there is a very bad prognosis with a high chance of death or institutionalization at 3 months," Dr. Strbian said. "In this case, we know that the patient may die if we do nothing, but perhaps the SEDAN [score] can help us determine if an endovascular approach might be better than thrombolysis."
The SEDAN score is simple, easy, and quick to calculate, he said. But it’s only part of the treatment decision process.
"We can’t treat based on this score alone. If the patient fulfills the indications for TPA, it’s up to the physician and the patient to decide whether they are willing to accept the risk of an intracranial hemorrhage."
Dr. Menon and Dr. Strbian reported having no financial conflicts.
NEW ORLEANS – Two new risk assessment scores predicted the likelihood of a symptomatic intracranial hemorrhage after ischemic stroke treatment with more than 70% accuracy.
Predictive rules such as these are important because there is still no large-scale prospective study that clearly identifies which patients with ischemic stroke are more likely to develop a brain bleed after treatment with intravenous tissue plasminogen activator (TPA), Dr. Bijoy Menon said at the International Stroke Conference.
Dr. Menon, a clinical stroke fellow at the University of Calgary (Alta.), and his colleagues developed a 101-point score based on data extracted from the Get With The Guidelines stroke cohort. The cohort consisted of 10,242 patients with ischemic stroke who received TPA within 3 hours of the onset of stroke symptoms. A derivation cohort comprised 70% of the group; the rule was then validated in the remaining 30%.
The cohort’s mean age was 69 years. All patients experienced a moderate to severe ischemic stroke, with a mean National Institutes of Health Stroke Scale (NIHSS) score of 11. They received TPA at a mean of 1.35 hours after symptom onset.
About 5% of the group (496 patients) experienced a symptomatic intracranial hemorrhage (ICH), which the investigators defined as neurologic worsening within 36 hours of TPA administration.
In a multivariate regression model, Dr. Menon and his coinvestigators found six patient characteristics that were significantly associated with a brain bleed. They assigned each of these characteristics a point spread based on the range of measurements:
• Age. From age 60 years or younger (8 points) to older than 80 (17 points).
• NIHSS score. From 1-5 (25 points) to 20 and over (42 points).
• Systolic blood pressure. From less than 120 mm Hg (10 points) to 180 mm Hg or higher (21 points).
• Blood glucose level. From less than 100 mg/dL (2 points) to 150 mg/dL or more (8 points).
• Ethnicity. Asian, 9 points; all others, 0 points.
• Gender. Male, 4 points; female, 0 points.
Diabetes and a history of stroke were significantly associated with ICH in the initial analysis, but the P values were nonsignificant in the multivariate analysis, Dr. Menon noted.
"We also did not find any significantly increased risk associated with warfarin use or with the baseline international normalized ratio," he said at the meeting, sponsored by the American Heart Association.
The score accurately predicted ICH in 71% of the validation cohort, "comparable to most of the other scoring methods out there."
While the score is "well validated and evidence based," neither it nor any other single measure should be the sole factor in determining ischemic stroke treatment, Dr. Menon emphasized. "This is very important. It should not be used to infer which patients would benefit most or least from IV TPA. This is a cohort study, and because treatment was at the discretion of the individual physicians, there may be a selection bias present in it."
Dr. Daniel Strbian of the University of Helsinki presented a second score, dubbed SEDAN. Dr. Strbian and his colleagues developed their score based on a cohort of 972 ischemic stroke patients who developed an imaging-proven brain bleed after IV TPA treatment.
In a multivariate analysis, he and his associates found five factors that were independently associated with a significantly increased risk of symptomatic ICH: blood glucose level on admission, computed tomography (CT) imaging positive for ischemic stroke, cerebral artery hyperdensity, age, and NIHSS score.
Points were assigned based on the relative risk (RR) associated with each factor – the higher the total score, the greater the risk of symptomatic ICH. Each of the five risk-point categories generated a significant risk ratio:1 point, RR 0.19; 2 points, RR 0.40; 3 points, RR 1.85; 4 points, RR 3.7; 5 points, RR 5.6.
The score was validated in two additional groups totaling about 2,000 patients, resulting in an overall accuracy of about 77%.
"If we have a patient with a very high score, there is a very bad prognosis with a high chance of death or institutionalization at 3 months," Dr. Strbian said. "In this case, we know that the patient may die if we do nothing, but perhaps the SEDAN [score] can help us determine if an endovascular approach might be better than thrombolysis."
The SEDAN score is simple, easy, and quick to calculate, he said. But it’s only part of the treatment decision process.
"We can’t treat based on this score alone. If the patient fulfills the indications for TPA, it’s up to the physician and the patient to decide whether they are willing to accept the risk of an intracranial hemorrhage."
Dr. Menon and Dr. Strbian reported having no financial conflicts.
FROM THE INTERNATIONAL STROKE CONFERENCE
Major Finding: Two new scores helped predict intracranial hemorrhage after TPA treatment with more than 70% accuracy.
Data Source: Each score was derived from and validated in large, heterogeneous groups of patients who received intravenous TPA after an ischemic stroke.
Disclosures: Dr. Menon and Dr. Strbian reported having no financial conflicts.
Solitaire Trumped Merci in Stroke Clot Retrieval Trial
NEW ORLEANS – An investigational clot-retrieval device completely recanalized blocked cerebral arteries, improved neurologic outcomes, and significantly reduced mortality from acute ischemic stroke more often than did the widely used Merci device in a randomized, open-label trial.
The SWIFT (SOLITAIRE-FR With the Intention for Thrombectomy) study intended to randomize 200 patients with ischemic stroke to treatment with either of the devices, but after 18 months, the trial’s data safety monitoring board stopped the study with 144 patients treated, citing "an overwhelming benefit" of the Solitaire-Flow Restoration device over the Merci device, Dr. Jeffrey Saver said at the International Stroke Conference.
"With these results, we see for the first time a highly effective cerebral recanalization procedure for ischemic stroke," said Dr. Saver, director of the stroke unit at the University of California, Los Angeles, in an interview. "[Tissue plasminogen activator] opens the arteries partially 40% of the time and completely just 5% of the time. This device opens them completely 60% of the time. It’s a dramatic step forward and a device that will probably be a game-changer for our systems, once it becomes available."
Solitaire is a columnar metal cage which, when expanded, engages the clot at multiple points, facilitating extraction and decreasing the chance of symptomatic intracranial bleeding. The Merci device, a corkscrew-like coil, does release pressure on the atrial wall, "but has a tendency to uncoil and fail to engage the clot," he said.
The patients’ average age was 67 years; most (68%) were male. The median pre-stroke modified Rankin Scale score was 0.5. Time to first angiogram was 4-5 hours.
The primary end point – successful recanalization without symptomatic intracranial hemorrhage – occurred in 61% of the Solitaire group and 24% of the Merci group. This was a highly significant difference with a P value of .0001 in both noninferiority and superiority analyses.
The trial’s secondary end points also indicated the superiority and noninferiority of Solitaire in comparison with Merci:
• Use of rescue therapy (21% vs. 44%).
• Symptomatic intracranial hemorrhage (2% vs. 11%).
• All intracranial hemorrhage (17% vs. 38%).
• Good 90-day neurologic outcome (58% vs. 33%).
• 90-day mortality (17% vs. 38%).
The investigators defined a good neurologic outcome as a modified Rankin Scale score of 0-2 if a patient came in at that level, if the patient returned to their baseline score after coming in with a worse score, or for patients who achieved at least a 10-point increase in their National Institutes of Health Stroke Scale score also were assessed as having a good outcome.
The results demonstrate an excellent number-needed-to-treat analysis, Dr. Saver said.
"For every 2.7 patients treated, 1 additional patient had a successful recanalization with no brain bleed. For every five patients treated, one patient was saved from death. And for every four patients treated, there was one more with a good neurologic outcome."
Device fracture occurred in two instances with Solitaire but in no cases with Merci. There were two instances with each device in which the device could not be delivered appropriately. Air embolism occurred in one patient with each device. Vasospasms occurred in eight cases with Solitaire and six cases with Merci. Two patients undergoing interventions with the Merci device had vessel perforations, compared with none who were treated with the Solitaire device. Distal emboli occurred with Solitaire in two patients and with Merci in three patients.
The study design included a "roll-in" period in which the investigators learned how to perform the interventions; 31 patients were treated during this time, while the remaining 113 were randomized to treatment with the Merci device (55) or the Solitaire (58). All of the patients either were ineligible for tissue plasminogen activator or had failed a treatment.
Each patient received up to three passes of the assigned device. A separate lab, blinded to treatment arms, reviewed scans of the treated arteries. If the clot remained, physicians could try other appropriate treatments.
While SWIFT decisively proved Solitaire’s capability as a clot retriever, it didn’t have the chance to explore the device’s other possible indication as a stent, Dr. Saver added.
‘This device is designed to pull the clot out or, if that fails, you can detach the stent portion and leave it in place. But what we experienced in this study was that it was getting the clot out so often, we weren’t able to test it as a stenting device."
In fact, for some patients, Solitaire may be more suited as a stent than as a retrieval device. "I think that for a patient who has plaques in the intracranial arteries and a little bit of clot on top of the plaque, retrieval is not the best strategy. The devices tend to snag on the hard edges of the plaque. Those patients are best treated with a stenting approach, which may possible with his device."
Solitaire has already been approved in Europe. These new data should help propel it to a U.S. approval, he said.
"The data from this trial have been submitted to the Food and Drug Administration, and we are hopeful that within a few weeks or months these results will allow the FDA to approve it in the U.S."
Ev3, the company that makes Solitaire, funded the SWIFT trial. Dr. Saver disclosed that he is on the company’s speakers advisory board.
This trial brings us into the third generation of new and potentially better devices to open blocked arteries in acute ischemic stroke. Solitaire showed pretty impressive recanalization rates, compared with Merci, and improved clinical and functional outcomes.
This device also looks at little easier to deploy than Merci, and it seems to be somewhat more practical and better-designed.
Dr. Ralph L. Sacco |
I think the SWIFT trial will bring very important information to the FDA review, and hopefully we will have another device entering the market to help us treat acute stroke.
But even after interventionists get the Solitaire device, they must be well trained before they start using it. Additionally, devices like this are primarily used in comprehensive stroke centers. To increase the number of patients who might benefit from it, we need to increase the numbers of our comprehensive stroke centers.
Dr. Ralph L. Sacco is the immediate past president of the American Heart Association and chairman of neurology at the University of Miami. He had no relevant disclosures.
This trial brings us into the third generation of new and potentially better devices to open blocked arteries in acute ischemic stroke. Solitaire showed pretty impressive recanalization rates, compared with Merci, and improved clinical and functional outcomes.
This device also looks at little easier to deploy than Merci, and it seems to be somewhat more practical and better-designed.
Dr. Ralph L. Sacco |
I think the SWIFT trial will bring very important information to the FDA review, and hopefully we will have another device entering the market to help us treat acute stroke.
But even after interventionists get the Solitaire device, they must be well trained before they start using it. Additionally, devices like this are primarily used in comprehensive stroke centers. To increase the number of patients who might benefit from it, we need to increase the numbers of our comprehensive stroke centers.
Dr. Ralph L. Sacco is the immediate past president of the American Heart Association and chairman of neurology at the University of Miami. He had no relevant disclosures.
This trial brings us into the third generation of new and potentially better devices to open blocked arteries in acute ischemic stroke. Solitaire showed pretty impressive recanalization rates, compared with Merci, and improved clinical and functional outcomes.
This device also looks at little easier to deploy than Merci, and it seems to be somewhat more practical and better-designed.
Dr. Ralph L. Sacco |
I think the SWIFT trial will bring very important information to the FDA review, and hopefully we will have another device entering the market to help us treat acute stroke.
But even after interventionists get the Solitaire device, they must be well trained before they start using it. Additionally, devices like this are primarily used in comprehensive stroke centers. To increase the number of patients who might benefit from it, we need to increase the numbers of our comprehensive stroke centers.
Dr. Ralph L. Sacco is the immediate past president of the American Heart Association and chairman of neurology at the University of Miami. He had no relevant disclosures.
NEW ORLEANS – An investigational clot-retrieval device completely recanalized blocked cerebral arteries, improved neurologic outcomes, and significantly reduced mortality from acute ischemic stroke more often than did the widely used Merci device in a randomized, open-label trial.
The SWIFT (SOLITAIRE-FR With the Intention for Thrombectomy) study intended to randomize 200 patients with ischemic stroke to treatment with either of the devices, but after 18 months, the trial’s data safety monitoring board stopped the study with 144 patients treated, citing "an overwhelming benefit" of the Solitaire-Flow Restoration device over the Merci device, Dr. Jeffrey Saver said at the International Stroke Conference.
"With these results, we see for the first time a highly effective cerebral recanalization procedure for ischemic stroke," said Dr. Saver, director of the stroke unit at the University of California, Los Angeles, in an interview. "[Tissue plasminogen activator] opens the arteries partially 40% of the time and completely just 5% of the time. This device opens them completely 60% of the time. It’s a dramatic step forward and a device that will probably be a game-changer for our systems, once it becomes available."
Solitaire is a columnar metal cage which, when expanded, engages the clot at multiple points, facilitating extraction and decreasing the chance of symptomatic intracranial bleeding. The Merci device, a corkscrew-like coil, does release pressure on the atrial wall, "but has a tendency to uncoil and fail to engage the clot," he said.
The patients’ average age was 67 years; most (68%) were male. The median pre-stroke modified Rankin Scale score was 0.5. Time to first angiogram was 4-5 hours.
The primary end point – successful recanalization without symptomatic intracranial hemorrhage – occurred in 61% of the Solitaire group and 24% of the Merci group. This was a highly significant difference with a P value of .0001 in both noninferiority and superiority analyses.
The trial’s secondary end points also indicated the superiority and noninferiority of Solitaire in comparison with Merci:
• Use of rescue therapy (21% vs. 44%).
• Symptomatic intracranial hemorrhage (2% vs. 11%).
• All intracranial hemorrhage (17% vs. 38%).
• Good 90-day neurologic outcome (58% vs. 33%).
• 90-day mortality (17% vs. 38%).
The investigators defined a good neurologic outcome as a modified Rankin Scale score of 0-2 if a patient came in at that level, if the patient returned to their baseline score after coming in with a worse score, or for patients who achieved at least a 10-point increase in their National Institutes of Health Stroke Scale score also were assessed as having a good outcome.
The results demonstrate an excellent number-needed-to-treat analysis, Dr. Saver said.
"For every 2.7 patients treated, 1 additional patient had a successful recanalization with no brain bleed. For every five patients treated, one patient was saved from death. And for every four patients treated, there was one more with a good neurologic outcome."
Device fracture occurred in two instances with Solitaire but in no cases with Merci. There were two instances with each device in which the device could not be delivered appropriately. Air embolism occurred in one patient with each device. Vasospasms occurred in eight cases with Solitaire and six cases with Merci. Two patients undergoing interventions with the Merci device had vessel perforations, compared with none who were treated with the Solitaire device. Distal emboli occurred with Solitaire in two patients and with Merci in three patients.
The study design included a "roll-in" period in which the investigators learned how to perform the interventions; 31 patients were treated during this time, while the remaining 113 were randomized to treatment with the Merci device (55) or the Solitaire (58). All of the patients either were ineligible for tissue plasminogen activator or had failed a treatment.
Each patient received up to three passes of the assigned device. A separate lab, blinded to treatment arms, reviewed scans of the treated arteries. If the clot remained, physicians could try other appropriate treatments.
While SWIFT decisively proved Solitaire’s capability as a clot retriever, it didn’t have the chance to explore the device’s other possible indication as a stent, Dr. Saver added.
‘This device is designed to pull the clot out or, if that fails, you can detach the stent portion and leave it in place. But what we experienced in this study was that it was getting the clot out so often, we weren’t able to test it as a stenting device."
In fact, for some patients, Solitaire may be more suited as a stent than as a retrieval device. "I think that for a patient who has plaques in the intracranial arteries and a little bit of clot on top of the plaque, retrieval is not the best strategy. The devices tend to snag on the hard edges of the plaque. Those patients are best treated with a stenting approach, which may possible with his device."
Solitaire has already been approved in Europe. These new data should help propel it to a U.S. approval, he said.
"The data from this trial have been submitted to the Food and Drug Administration, and we are hopeful that within a few weeks or months these results will allow the FDA to approve it in the U.S."
Ev3, the company that makes Solitaire, funded the SWIFT trial. Dr. Saver disclosed that he is on the company’s speakers advisory board.
NEW ORLEANS – An investigational clot-retrieval device completely recanalized blocked cerebral arteries, improved neurologic outcomes, and significantly reduced mortality from acute ischemic stroke more often than did the widely used Merci device in a randomized, open-label trial.
The SWIFT (SOLITAIRE-FR With the Intention for Thrombectomy) study intended to randomize 200 patients with ischemic stroke to treatment with either of the devices, but after 18 months, the trial’s data safety monitoring board stopped the study with 144 patients treated, citing "an overwhelming benefit" of the Solitaire-Flow Restoration device over the Merci device, Dr. Jeffrey Saver said at the International Stroke Conference.
"With these results, we see for the first time a highly effective cerebral recanalization procedure for ischemic stroke," said Dr. Saver, director of the stroke unit at the University of California, Los Angeles, in an interview. "[Tissue plasminogen activator] opens the arteries partially 40% of the time and completely just 5% of the time. This device opens them completely 60% of the time. It’s a dramatic step forward and a device that will probably be a game-changer for our systems, once it becomes available."
Solitaire is a columnar metal cage which, when expanded, engages the clot at multiple points, facilitating extraction and decreasing the chance of symptomatic intracranial bleeding. The Merci device, a corkscrew-like coil, does release pressure on the atrial wall, "but has a tendency to uncoil and fail to engage the clot," he said.
The patients’ average age was 67 years; most (68%) were male. The median pre-stroke modified Rankin Scale score was 0.5. Time to first angiogram was 4-5 hours.
The primary end point – successful recanalization without symptomatic intracranial hemorrhage – occurred in 61% of the Solitaire group and 24% of the Merci group. This was a highly significant difference with a P value of .0001 in both noninferiority and superiority analyses.
The trial’s secondary end points also indicated the superiority and noninferiority of Solitaire in comparison with Merci:
• Use of rescue therapy (21% vs. 44%).
• Symptomatic intracranial hemorrhage (2% vs. 11%).
• All intracranial hemorrhage (17% vs. 38%).
• Good 90-day neurologic outcome (58% vs. 33%).
• 90-day mortality (17% vs. 38%).
The investigators defined a good neurologic outcome as a modified Rankin Scale score of 0-2 if a patient came in at that level, if the patient returned to their baseline score after coming in with a worse score, or for patients who achieved at least a 10-point increase in their National Institutes of Health Stroke Scale score also were assessed as having a good outcome.
The results demonstrate an excellent number-needed-to-treat analysis, Dr. Saver said.
"For every 2.7 patients treated, 1 additional patient had a successful recanalization with no brain bleed. For every five patients treated, one patient was saved from death. And for every four patients treated, there was one more with a good neurologic outcome."
Device fracture occurred in two instances with Solitaire but in no cases with Merci. There were two instances with each device in which the device could not be delivered appropriately. Air embolism occurred in one patient with each device. Vasospasms occurred in eight cases with Solitaire and six cases with Merci. Two patients undergoing interventions with the Merci device had vessel perforations, compared with none who were treated with the Solitaire device. Distal emboli occurred with Solitaire in two patients and with Merci in three patients.
The study design included a "roll-in" period in which the investigators learned how to perform the interventions; 31 patients were treated during this time, while the remaining 113 were randomized to treatment with the Merci device (55) or the Solitaire (58). All of the patients either were ineligible for tissue plasminogen activator or had failed a treatment.
Each patient received up to three passes of the assigned device. A separate lab, blinded to treatment arms, reviewed scans of the treated arteries. If the clot remained, physicians could try other appropriate treatments.
While SWIFT decisively proved Solitaire’s capability as a clot retriever, it didn’t have the chance to explore the device’s other possible indication as a stent, Dr. Saver added.
‘This device is designed to pull the clot out or, if that fails, you can detach the stent portion and leave it in place. But what we experienced in this study was that it was getting the clot out so often, we weren’t able to test it as a stenting device."
In fact, for some patients, Solitaire may be more suited as a stent than as a retrieval device. "I think that for a patient who has plaques in the intracranial arteries and a little bit of clot on top of the plaque, retrieval is not the best strategy. The devices tend to snag on the hard edges of the plaque. Those patients are best treated with a stenting approach, which may possible with his device."
Solitaire has already been approved in Europe. These new data should help propel it to a U.S. approval, he said.
"The data from this trial have been submitted to the Food and Drug Administration, and we are hopeful that within a few weeks or months these results will allow the FDA to approve it in the U.S."
Ev3, the company that makes Solitaire, funded the SWIFT trial. Dr. Saver disclosed that he is on the company’s speakers advisory board.
FROM THE INTERNATIONAL STROKE CONFERENCE
Major Finding: Successful recanalization without symptomatic intracranial hemorrhage occurred in a significantly larger proportion of patients who received treatment with Solitaire than did with Merci (61% vs. 24%).
Data Source: SWIFT randomized 113 patients with acute ischemic stroke to treatment with the investigational Solitaire device or the approved Merci device.
Disclosures: Ev3, the company that makes Solitaire, funded the SWIFT trial. Dr. Saver disclosed that he is on the company’s speakers advisory board.
Intracranial Bleed Risk Higher in Warfarin Than Rivaroxaban Users
NEW ORLEANS – Compared with warfarin, the new-generation anticoagulant rivaroxaban was associated with a 40% lower risk of intracranial hemorrhage in patients with atrial fibrillation who were taking the agents to prevent stroke.
Since its approval by the Food and Drug Administration last fall, rivaroxaban has been deemed an alternative to warfarin, especially among those patients who have trouble maintaining a stable international normalized ratio, who have warfarin side effects, or who find the constant warfarin monitoring untenable, Dr. Graeme Hankey said at an international stroke conference.
Rivaroxaban also doesn’t involve the medication and food interactions that can make warfarin problematic, said Dr. Hankey, head of the stroke unit at Royal Perth Hospital, Western Australia.
"Many patients don’t want to take warfarin for these reasons," he said in an interview. "Rivaroxaban doesn’t have these interactions. It has a stable, predictable effect with once-a-day dosing and doesn’t require this constant monitoring."
Dr. Hankey’s subanalysis of the pivotal ROCKET-AF study, which compared rivaroxaban with warfarin, also determined that some patients had up to a 4-fold increased risk of an intracranial bleed while taking either of the anticoagulants.
The initial study – Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared With Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation or ROCKET AF – included 14,264 patients with atrial fibrillation; a history of stroke, TIAs, or systemic embolism; and at least two of the following stroke risk factors: heart failure or a left ventricular ejection fraction of 35% or less; hypertension; an age of 75 years or more; or the presence of diabetes mellitus. Patients were randomized to receive dose-adjusted warfarin or rivaroxaban. Those with normal creatinine clearance received 20 mg daily of the study drug; those with a decreased clearance received 15 mg rivaroxaban daily.
The study’s main end point – a combination of stroke or systemic embolism – was similar in both groups (about 2% per year). The rates of myocardial infarction, and major bleeding also were not significantly different. Vascular mortality and all-cause mortality were nearly identical. However, there was a significant difference in intracranial hemorrhage, favoring rivaroxaban (0.8%; 55 vs. 1.2%; 84) (N. Engl. J. Med 2011;365:883-91). Fatal bleeding occurred in less than 1% of each group.
Dr. Hankey’s subanalysis examined the drug’s effect on intracranial hemorrhage, "probably the most-feared complication of anticoagulation," according to the investigator. It also examined independent risk factors for intracranial hemorrhage regardless of which drug the study patients took.
He and his colleagues found 172 intracranial hemorrhages in ROCKET-AF – a rate of about 7% per year. Of these bleeds, 128 were intracerebral, 5 subarachnoid, 38 subdural, and 1 extradural. Patients taking rivaroxaban were 40% less likely to have an intracranial hemorrhage than were those taking warfarin.
The subanalysis identified factors that significantly increased the risk of an intracranial bleed. The largest by far were race and concomitant use of clopidogrel. Compared with whites, blacks were more than 400% more likely to have a brain bleed (odds ratio, 4.2), while Asians were 200% more likely. Those taking clopidogrel plus either warfarin or rivaroxaban were 250% more likely to have a bleed than were those not taking an additional anticoagulant (OR, 2.50).
Other significant associations with brain bleeds were as follows:
• High diastolic blood pressure – 21% increased risk for every 10 mm/Hg above normal.
• Poor kidney function – 10% increased risk for every 10 mL/min decrease in creatinine clearance.
• History of stroke or transient ischemic attack – 55% increased risk.
• Low platelets – 8% increased risk;
• Low albumin – 37% increased risk.
The FDA approved rivaroxaban in 2011, to reduce the risk of stroke and systemic embolism in patients with nonvalvular atrial fibrillation.
ROCKET-AF was supported by Johnson & Johnson Pharmaceutical Researchand Development and by Bayer HealthCare. Dr. Hankey was an investigator on the trial and has received honoraria for speaking on the study drug. He is also a consultant for Boehringer Ingelheim and Bayer, which make dabigatran and rivaroxaban, respectively.
Although rivaroxaban is another tool in our armamentarium for patients at risk of stroke, I think it’s premature to make a paradigm shift away from warfarin.
Clearly, ROCKET-AF showed us that rivaroxaban was noninferior to warfarin. This subanalysis showed that rivaroxaban also had a decreased rate of intracranial hemorrhage, compared with warfarin, but we must keep in mind that the drug did not affect the ROCKET-AF trial’s primary end points of stroke or systemic embolism, nor did it decrease mortality, compared with warfarin.
One of the biggest concerns about rivaroxaban and some of the other new antithrombotic agents is that we have no established protocol to deal with bleeding, should it occur, as we do with warfarin. At this point, we really don’t know the best way to deal with a bleed. This is something we will learn only with more time. And, compared with the other new-generation anticoagulants apixaban and dabigatran, rivaroxaban seems to have a few more side effects, such as nausea and increased liver enzymes.
Rivaroxaban and these other drugs do have benefit of being fixed-dose medications taken once or twice a day, and which don’t require the monitoring that warfarin does. But they are going to be more expensive.
If a patient is under treatment with warfarin and is stable, and if the monitoring schedule is acceptable and there are no major INR fluctuations, I don’t think there is any indication to shift treatment. If the patient has difficulty maintaining a therapeutic INR or if the monitoring is inconvenient, then I suspect patients might welcome the use of one of these fixed-dose agents.
Dr. José Biller is chairman of neurology at Loyola University, Maywood, Ill. He is the lead author on a recently published review of the new-generation anticoagulation drugs (Expert Rev. Neurother. 2012;12:179-90). He had no disclosures.
Although rivaroxaban is another tool in our armamentarium for patients at risk of stroke, I think it’s premature to make a paradigm shift away from warfarin.
Clearly, ROCKET-AF showed us that rivaroxaban was noninferior to warfarin. This subanalysis showed that rivaroxaban also had a decreased rate of intracranial hemorrhage, compared with warfarin, but we must keep in mind that the drug did not affect the ROCKET-AF trial’s primary end points of stroke or systemic embolism, nor did it decrease mortality, compared with warfarin.
One of the biggest concerns about rivaroxaban and some of the other new antithrombotic agents is that we have no established protocol to deal with bleeding, should it occur, as we do with warfarin. At this point, we really don’t know the best way to deal with a bleed. This is something we will learn only with more time. And, compared with the other new-generation anticoagulants apixaban and dabigatran, rivaroxaban seems to have a few more side effects, such as nausea and increased liver enzymes.
Rivaroxaban and these other drugs do have benefit of being fixed-dose medications taken once or twice a day, and which don’t require the monitoring that warfarin does. But they are going to be more expensive.
If a patient is under treatment with warfarin and is stable, and if the monitoring schedule is acceptable and there are no major INR fluctuations, I don’t think there is any indication to shift treatment. If the patient has difficulty maintaining a therapeutic INR or if the monitoring is inconvenient, then I suspect patients might welcome the use of one of these fixed-dose agents.
Dr. José Biller is chairman of neurology at Loyola University, Maywood, Ill. He is the lead author on a recently published review of the new-generation anticoagulation drugs (Expert Rev. Neurother. 2012;12:179-90). He had no disclosures.
Although rivaroxaban is another tool in our armamentarium for patients at risk of stroke, I think it’s premature to make a paradigm shift away from warfarin.
Clearly, ROCKET-AF showed us that rivaroxaban was noninferior to warfarin. This subanalysis showed that rivaroxaban also had a decreased rate of intracranial hemorrhage, compared with warfarin, but we must keep in mind that the drug did not affect the ROCKET-AF trial’s primary end points of stroke or systemic embolism, nor did it decrease mortality, compared with warfarin.
One of the biggest concerns about rivaroxaban and some of the other new antithrombotic agents is that we have no established protocol to deal with bleeding, should it occur, as we do with warfarin. At this point, we really don’t know the best way to deal with a bleed. This is something we will learn only with more time. And, compared with the other new-generation anticoagulants apixaban and dabigatran, rivaroxaban seems to have a few more side effects, such as nausea and increased liver enzymes.
Rivaroxaban and these other drugs do have benefit of being fixed-dose medications taken once or twice a day, and which don’t require the monitoring that warfarin does. But they are going to be more expensive.
If a patient is under treatment with warfarin and is stable, and if the monitoring schedule is acceptable and there are no major INR fluctuations, I don’t think there is any indication to shift treatment. If the patient has difficulty maintaining a therapeutic INR or if the monitoring is inconvenient, then I suspect patients might welcome the use of one of these fixed-dose agents.
Dr. José Biller is chairman of neurology at Loyola University, Maywood, Ill. He is the lead author on a recently published review of the new-generation anticoagulation drugs (Expert Rev. Neurother. 2012;12:179-90). He had no disclosures.
NEW ORLEANS – Compared with warfarin, the new-generation anticoagulant rivaroxaban was associated with a 40% lower risk of intracranial hemorrhage in patients with atrial fibrillation who were taking the agents to prevent stroke.
Since its approval by the Food and Drug Administration last fall, rivaroxaban has been deemed an alternative to warfarin, especially among those patients who have trouble maintaining a stable international normalized ratio, who have warfarin side effects, or who find the constant warfarin monitoring untenable, Dr. Graeme Hankey said at an international stroke conference.
Rivaroxaban also doesn’t involve the medication and food interactions that can make warfarin problematic, said Dr. Hankey, head of the stroke unit at Royal Perth Hospital, Western Australia.
"Many patients don’t want to take warfarin for these reasons," he said in an interview. "Rivaroxaban doesn’t have these interactions. It has a stable, predictable effect with once-a-day dosing and doesn’t require this constant monitoring."
Dr. Hankey’s subanalysis of the pivotal ROCKET-AF study, which compared rivaroxaban with warfarin, also determined that some patients had up to a 4-fold increased risk of an intracranial bleed while taking either of the anticoagulants.
The initial study – Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared With Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation or ROCKET AF – included 14,264 patients with atrial fibrillation; a history of stroke, TIAs, or systemic embolism; and at least two of the following stroke risk factors: heart failure or a left ventricular ejection fraction of 35% or less; hypertension; an age of 75 years or more; or the presence of diabetes mellitus. Patients were randomized to receive dose-adjusted warfarin or rivaroxaban. Those with normal creatinine clearance received 20 mg daily of the study drug; those with a decreased clearance received 15 mg rivaroxaban daily.
The study’s main end point – a combination of stroke or systemic embolism – was similar in both groups (about 2% per year). The rates of myocardial infarction, and major bleeding also were not significantly different. Vascular mortality and all-cause mortality were nearly identical. However, there was a significant difference in intracranial hemorrhage, favoring rivaroxaban (0.8%; 55 vs. 1.2%; 84) (N. Engl. J. Med 2011;365:883-91). Fatal bleeding occurred in less than 1% of each group.
Dr. Hankey’s subanalysis examined the drug’s effect on intracranial hemorrhage, "probably the most-feared complication of anticoagulation," according to the investigator. It also examined independent risk factors for intracranial hemorrhage regardless of which drug the study patients took.
He and his colleagues found 172 intracranial hemorrhages in ROCKET-AF – a rate of about 7% per year. Of these bleeds, 128 were intracerebral, 5 subarachnoid, 38 subdural, and 1 extradural. Patients taking rivaroxaban were 40% less likely to have an intracranial hemorrhage than were those taking warfarin.
The subanalysis identified factors that significantly increased the risk of an intracranial bleed. The largest by far were race and concomitant use of clopidogrel. Compared with whites, blacks were more than 400% more likely to have a brain bleed (odds ratio, 4.2), while Asians were 200% more likely. Those taking clopidogrel plus either warfarin or rivaroxaban were 250% more likely to have a bleed than were those not taking an additional anticoagulant (OR, 2.50).
Other significant associations with brain bleeds were as follows:
• High diastolic blood pressure – 21% increased risk for every 10 mm/Hg above normal.
• Poor kidney function – 10% increased risk for every 10 mL/min decrease in creatinine clearance.
• History of stroke or transient ischemic attack – 55% increased risk.
• Low platelets – 8% increased risk;
• Low albumin – 37% increased risk.
The FDA approved rivaroxaban in 2011, to reduce the risk of stroke and systemic embolism in patients with nonvalvular atrial fibrillation.
ROCKET-AF was supported by Johnson & Johnson Pharmaceutical Researchand Development and by Bayer HealthCare. Dr. Hankey was an investigator on the trial and has received honoraria for speaking on the study drug. He is also a consultant for Boehringer Ingelheim and Bayer, which make dabigatran and rivaroxaban, respectively.
NEW ORLEANS – Compared with warfarin, the new-generation anticoagulant rivaroxaban was associated with a 40% lower risk of intracranial hemorrhage in patients with atrial fibrillation who were taking the agents to prevent stroke.
Since its approval by the Food and Drug Administration last fall, rivaroxaban has been deemed an alternative to warfarin, especially among those patients who have trouble maintaining a stable international normalized ratio, who have warfarin side effects, or who find the constant warfarin monitoring untenable, Dr. Graeme Hankey said at an international stroke conference.
Rivaroxaban also doesn’t involve the medication and food interactions that can make warfarin problematic, said Dr. Hankey, head of the stroke unit at Royal Perth Hospital, Western Australia.
"Many patients don’t want to take warfarin for these reasons," he said in an interview. "Rivaroxaban doesn’t have these interactions. It has a stable, predictable effect with once-a-day dosing and doesn’t require this constant monitoring."
Dr. Hankey’s subanalysis of the pivotal ROCKET-AF study, which compared rivaroxaban with warfarin, also determined that some patients had up to a 4-fold increased risk of an intracranial bleed while taking either of the anticoagulants.
The initial study – Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared With Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation or ROCKET AF – included 14,264 patients with atrial fibrillation; a history of stroke, TIAs, or systemic embolism; and at least two of the following stroke risk factors: heart failure or a left ventricular ejection fraction of 35% or less; hypertension; an age of 75 years or more; or the presence of diabetes mellitus. Patients were randomized to receive dose-adjusted warfarin or rivaroxaban. Those with normal creatinine clearance received 20 mg daily of the study drug; those with a decreased clearance received 15 mg rivaroxaban daily.
The study’s main end point – a combination of stroke or systemic embolism – was similar in both groups (about 2% per year). The rates of myocardial infarction, and major bleeding also were not significantly different. Vascular mortality and all-cause mortality were nearly identical. However, there was a significant difference in intracranial hemorrhage, favoring rivaroxaban (0.8%; 55 vs. 1.2%; 84) (N. Engl. J. Med 2011;365:883-91). Fatal bleeding occurred in less than 1% of each group.
Dr. Hankey’s subanalysis examined the drug’s effect on intracranial hemorrhage, "probably the most-feared complication of anticoagulation," according to the investigator. It also examined independent risk factors for intracranial hemorrhage regardless of which drug the study patients took.
He and his colleagues found 172 intracranial hemorrhages in ROCKET-AF – a rate of about 7% per year. Of these bleeds, 128 were intracerebral, 5 subarachnoid, 38 subdural, and 1 extradural. Patients taking rivaroxaban were 40% less likely to have an intracranial hemorrhage than were those taking warfarin.
The subanalysis identified factors that significantly increased the risk of an intracranial bleed. The largest by far were race and concomitant use of clopidogrel. Compared with whites, blacks were more than 400% more likely to have a brain bleed (odds ratio, 4.2), while Asians were 200% more likely. Those taking clopidogrel plus either warfarin or rivaroxaban were 250% more likely to have a bleed than were those not taking an additional anticoagulant (OR, 2.50).
Other significant associations with brain bleeds were as follows:
• High diastolic blood pressure – 21% increased risk for every 10 mm/Hg above normal.
• Poor kidney function – 10% increased risk for every 10 mL/min decrease in creatinine clearance.
• History of stroke or transient ischemic attack – 55% increased risk.
• Low platelets – 8% increased risk;
• Low albumin – 37% increased risk.
The FDA approved rivaroxaban in 2011, to reduce the risk of stroke and systemic embolism in patients with nonvalvular atrial fibrillation.
ROCKET-AF was supported by Johnson & Johnson Pharmaceutical Researchand Development and by Bayer HealthCare. Dr. Hankey was an investigator on the trial and has received honoraria for speaking on the study drug. He is also a consultant for Boehringer Ingelheim and Bayer, which make dabigatran and rivaroxaban, respectively.
FROM THE INTERNATIONAL STROKE CONFERENCE
Major Finding:In patients with atrial fibrillation and a high risk for
stroke, intracranial hemorrhage occurred in 1.2% of those taking warfarin for
stroke prevention and in 0.8% of those taking rivaroxaban, for an absolute risk
reduction of 0.4% and a relative risk reduction of 40%.
Data Source: A subanalysis of the ROCKET-AF study.
Disclosures: ROCKET-AF was supported by Johnson & Johnson Pharmaceutical Research and Development and by Bayer HealthCare. Dr. Hankey was an investigator on the trial and has received honoraria for speaking on the study drug. He is also a consultant for Boehringer Ingelheim and Bayer, which make dabigatran and rivaroxaban, respectively.
Memory Declines Years Before Stroke Strikes
NEW ORLEANS – Memory is already impaired years before a stroke occurs, declining most rapidly in people destined to suffer a fatal stroke, according to a population-based study of more than 11,000 people.
"For every year that passed [after age 50 years], those who survived a stroke lost their memory at twice the rate as controls. And for those who didn’t survive, their annual memory loss was 3 to 4 years faster than the controls," said Maria Glymour, Sc.D., of the Harvard University School of Public Health, Boston.*
Dr. Glymour and her colleagues examined the relationship between memory and stroke using data from the national Health and Retirement Study (HRS) cohort of people aged 50 years or older.
Interviews with the study’s participants occurred every 2 years, starting in 1998. In their 10-year follow-up study, the investigators focused on 1,456 people who had survived a stroke, 364 who died from a stroke, and 9,994 who did not have a stroke. An immediate and delayed word list recall task, included in the HRS, served as the proxy for working memory among the group.
As expected, Dr. Glymour and her associates found trajectories showing age-related memory decline among all subjects after their baseline interview.
There were also baseline differences between the groups. People who had a stroke – both survivors and nonsurvivors – had significantly poorer memory function at the beginning of the follow-up period.
"More than half of the memory gap between recent stroke survivors and stroke-free adults of similar age was evident before the stroke," she said during a press briefing at the International Stroke Conference.
The groups continued to separate as the years progressed, although individuals in all three groups had trajectories of at least some age-related memory decline during the 10-year follow-up period.
The study did not assess total cognitive ability, so Dr. Glymour could not say how many in each group already had some basis for memory impairment. But even among the stroke groups, many subjects started out with high function, she noted. "It was the rate of decline that was the key factor."
"We are really interested to know what’s going on in the brains of these people that seems to be related to having a stroke. My guess is that there is a physiologic basis – that there might be some subclinical strokes that have already occurred that affect memory."
Another possibility could be memory-related behavioral issues, she said. "If people are already having trouble with memory, they might have a harder time controlling their stroke risks – for example, taking their blood pressure medications."
Dr. Steven Greenberg, director of the Hemorrhagic Stroke Research Program at Massachusetts General Hospital, Boston, and an American Heart Association spokesman, agreed.
"The cause and effect are still a little unclear. But one possibility is this idea of unnoticed strokes going on over time that cause people to have such a bad trajectory of memory loss. When they do have a symptomatic stroke, they have very little reserve left."
The study was sponsored by the National Institutes of Health. None of the investigators or Dr. Greenberg had any relevant financial disclosures.
*This story was updated 2/2/2012.
NEW ORLEANS – Memory is already impaired years before a stroke occurs, declining most rapidly in people destined to suffer a fatal stroke, according to a population-based study of more than 11,000 people.
"For every year that passed [after age 50 years], those who survived a stroke lost their memory at twice the rate as controls. And for those who didn’t survive, their annual memory loss was 3 to 4 years faster than the controls," said Maria Glymour, Sc.D., of the Harvard University School of Public Health, Boston.*
Dr. Glymour and her colleagues examined the relationship between memory and stroke using data from the national Health and Retirement Study (HRS) cohort of people aged 50 years or older.
Interviews with the study’s participants occurred every 2 years, starting in 1998. In their 10-year follow-up study, the investigators focused on 1,456 people who had survived a stroke, 364 who died from a stroke, and 9,994 who did not have a stroke. An immediate and delayed word list recall task, included in the HRS, served as the proxy for working memory among the group.
As expected, Dr. Glymour and her associates found trajectories showing age-related memory decline among all subjects after their baseline interview.
There were also baseline differences between the groups. People who had a stroke – both survivors and nonsurvivors – had significantly poorer memory function at the beginning of the follow-up period.
"More than half of the memory gap between recent stroke survivors and stroke-free adults of similar age was evident before the stroke," she said during a press briefing at the International Stroke Conference.
The groups continued to separate as the years progressed, although individuals in all three groups had trajectories of at least some age-related memory decline during the 10-year follow-up period.
The study did not assess total cognitive ability, so Dr. Glymour could not say how many in each group already had some basis for memory impairment. But even among the stroke groups, many subjects started out with high function, she noted. "It was the rate of decline that was the key factor."
"We are really interested to know what’s going on in the brains of these people that seems to be related to having a stroke. My guess is that there is a physiologic basis – that there might be some subclinical strokes that have already occurred that affect memory."
Another possibility could be memory-related behavioral issues, she said. "If people are already having trouble with memory, they might have a harder time controlling their stroke risks – for example, taking their blood pressure medications."
Dr. Steven Greenberg, director of the Hemorrhagic Stroke Research Program at Massachusetts General Hospital, Boston, and an American Heart Association spokesman, agreed.
"The cause and effect are still a little unclear. But one possibility is this idea of unnoticed strokes going on over time that cause people to have such a bad trajectory of memory loss. When they do have a symptomatic stroke, they have very little reserve left."
The study was sponsored by the National Institutes of Health. None of the investigators or Dr. Greenberg had any relevant financial disclosures.
*This story was updated 2/2/2012.
NEW ORLEANS – Memory is already impaired years before a stroke occurs, declining most rapidly in people destined to suffer a fatal stroke, according to a population-based study of more than 11,000 people.
"For every year that passed [after age 50 years], those who survived a stroke lost their memory at twice the rate as controls. And for those who didn’t survive, their annual memory loss was 3 to 4 years faster than the controls," said Maria Glymour, Sc.D., of the Harvard University School of Public Health, Boston.*
Dr. Glymour and her colleagues examined the relationship between memory and stroke using data from the national Health and Retirement Study (HRS) cohort of people aged 50 years or older.
Interviews with the study’s participants occurred every 2 years, starting in 1998. In their 10-year follow-up study, the investigators focused on 1,456 people who had survived a stroke, 364 who died from a stroke, and 9,994 who did not have a stroke. An immediate and delayed word list recall task, included in the HRS, served as the proxy for working memory among the group.
As expected, Dr. Glymour and her associates found trajectories showing age-related memory decline among all subjects after their baseline interview.
There were also baseline differences between the groups. People who had a stroke – both survivors and nonsurvivors – had significantly poorer memory function at the beginning of the follow-up period.
"More than half of the memory gap between recent stroke survivors and stroke-free adults of similar age was evident before the stroke," she said during a press briefing at the International Stroke Conference.
The groups continued to separate as the years progressed, although individuals in all three groups had trajectories of at least some age-related memory decline during the 10-year follow-up period.
The study did not assess total cognitive ability, so Dr. Glymour could not say how many in each group already had some basis for memory impairment. But even among the stroke groups, many subjects started out with high function, she noted. "It was the rate of decline that was the key factor."
"We are really interested to know what’s going on in the brains of these people that seems to be related to having a stroke. My guess is that there is a physiologic basis – that there might be some subclinical strokes that have already occurred that affect memory."
Another possibility could be memory-related behavioral issues, she said. "If people are already having trouble with memory, they might have a harder time controlling their stroke risks – for example, taking their blood pressure medications."
Dr. Steven Greenberg, director of the Hemorrhagic Stroke Research Program at Massachusetts General Hospital, Boston, and an American Heart Association spokesman, agreed.
"The cause and effect are still a little unclear. But one possibility is this idea of unnoticed strokes going on over time that cause people to have such a bad trajectory of memory loss. When they do have a symptomatic stroke, they have very little reserve left."
The study was sponsored by the National Institutes of Health. None of the investigators or Dr. Greenberg had any relevant financial disclosures.
*This story was updated 2/2/2012.
FROM THE INTERNATIONAL STROKE CONFERENCE
Major Finding: Over a 10-year period, memory in stroke survivors declined twice as fast as in those who never had a stroke; the rate of decline was three times faster in people who eventually had a fatal stroke.
Data Source: The National Health Retirement study, a biennial survey of U.S. citizens aged 50 years and older.
Disclosures: The study was sponsored by the National Institutes of Health. None of the investigators or Dr. Greenberg had any relevant financial disclosures.
Most New ADHD Patients Don't Need Cardiac Testing
NEW YORK – An electrocadiogram* probably isn’t necessary for most youngsters starting a medication for attention-deficit/hyperactivity disorder.
Some easy screening questions should be enough to identify any children who have cardiovascular risks that a stimulant could exacerbate, Dr. James J. McGough said at a psychopharmacology update sponsored by the American Academy of Child and Adolescent Psychiatry.
"I am going to suggest that we end this neurosis" about cardiovascular risks during ADHD treatment. "The questions to ask before starting a kid out on any psychotropic medication are the same you would ask any child who is getting a sports physical," said Dr. McGough, chief of staff of the Resnick Neuropsychiatric Hospital at the University of California, Los Angeles.
The biggest fear is sudden cardiovascular death – an event usually associated with sport. "I don’t think a month goes by that I don’t hear about a soccer player, cheerleader, or basketball player who has had a sudden death. It’s tragic, but it happens. The bottom line is that there is a three times greater risk of sudden death associated with sports than with being on a stimulant."
In light of this statistic, he said: "There would be more sense in doing an EKG on every high school athlete, but no one is calling for that."
The key questions that should be in every ADHD assessment focus on family and personal history:
• Have you ever fainted while exercising?
• Is there a family history of sudden cardiac death?
• Is there any cardiac abnormality or a heart murmur?
If the answer to any of these is "yes," the patient should see a pediatric cardiologist before starting a stimulant medication. Otherwise, Dr. McGough said, "The benefit of an EKG is really not worth the cost."
The American Heart Association raised concerns about cardiac death and ADHD medications in 2008, when it recommended that every child have an EKG read by a pediatric cardiologist before beginning stimulant medication. However, the group noted, although the test was preferred, it should not be mandatory (Circ. 2008;117:2407-23).
A 2009 postmortem case-control study seemed to bolster the recommendation (Am. J. Psychiatry 2009;166:992-1001).
The authors examined amphetamine exposure among 1,128 children aged 7-19 years, half of whom had a sudden unexplained death and half of whom died in motor vehicle accidents. Ten of those in the sudden death group had amphetamine exposure compared to two in the accident group – a significant difference. The authors concluded that amphetamines could increase the risk of sudden death.
Despite this study, the Food and Drug Administration decided not to require cardiac testing for every ADHD start, Dr. McGough said. More recent studies suggest that the risk is very small or absent.
"There are about a half dozen papers on this now," including one published recently and two large retrospective cohort studies published last year.
The current study examined the risk of cardiovascular events in a database of 171,126 youngsters aged 6-21 years. There were three new cardiac symptoms and less than one cardiac event per 1 million days of current stimulant use. Compared to the reference group, the adjusted odds ratio of cardiac events during current use was 1.18, and with past use, 0.93 (J. Am. Acad. Child Adolesc. Psychiatry 2012;51:147-56).
Last November, the New England Journal of Medicine published a cohort study of 1.2 million children and young adults, 373,667 of whom were currently using ADHD medications. There were 81 serious cardiovascular events, a rate of 3 per 100,000 person-years (N. Engl. J. Med. 2011;365:1896-904).
Another study appeared in Pediatrics. The cohort consisted of 241,417 ADHD medication users. No significant associations were found with sudden death, ventricular arrhythmia, or all-cause mortality. None of the strokes identified during exposed time to ADHD agents were validated (Pediatrics 2011;127:1102-10).
"I think it’s time we put an end to this brouhaha," Dr. McGough said.
Dr. McGough disclosed that he is on the advisory boards of NextWave Pharmaceuticals, Noven Pharmaceuticals, Shiongi, Shire Pharmaceuticals, and Supernus Pharmaceuticals. He also is a consultant for Alexa Pharmaceuticals and MedImmune.
* Correction 2/27/2012: The technology mentioned in this sentence previously was misidentified.
NEW YORK – An electrocadiogram* probably isn’t necessary for most youngsters starting a medication for attention-deficit/hyperactivity disorder.
Some easy screening questions should be enough to identify any children who have cardiovascular risks that a stimulant could exacerbate, Dr. James J. McGough said at a psychopharmacology update sponsored by the American Academy of Child and Adolescent Psychiatry.
"I am going to suggest that we end this neurosis" about cardiovascular risks during ADHD treatment. "The questions to ask before starting a kid out on any psychotropic medication are the same you would ask any child who is getting a sports physical," said Dr. McGough, chief of staff of the Resnick Neuropsychiatric Hospital at the University of California, Los Angeles.
The biggest fear is sudden cardiovascular death – an event usually associated with sport. "I don’t think a month goes by that I don’t hear about a soccer player, cheerleader, or basketball player who has had a sudden death. It’s tragic, but it happens. The bottom line is that there is a three times greater risk of sudden death associated with sports than with being on a stimulant."
In light of this statistic, he said: "There would be more sense in doing an EKG on every high school athlete, but no one is calling for that."
The key questions that should be in every ADHD assessment focus on family and personal history:
• Have you ever fainted while exercising?
• Is there a family history of sudden cardiac death?
• Is there any cardiac abnormality or a heart murmur?
If the answer to any of these is "yes," the patient should see a pediatric cardiologist before starting a stimulant medication. Otherwise, Dr. McGough said, "The benefit of an EKG is really not worth the cost."
The American Heart Association raised concerns about cardiac death and ADHD medications in 2008, when it recommended that every child have an EKG read by a pediatric cardiologist before beginning stimulant medication. However, the group noted, although the test was preferred, it should not be mandatory (Circ. 2008;117:2407-23).
A 2009 postmortem case-control study seemed to bolster the recommendation (Am. J. Psychiatry 2009;166:992-1001).
The authors examined amphetamine exposure among 1,128 children aged 7-19 years, half of whom had a sudden unexplained death and half of whom died in motor vehicle accidents. Ten of those in the sudden death group had amphetamine exposure compared to two in the accident group – a significant difference. The authors concluded that amphetamines could increase the risk of sudden death.
Despite this study, the Food and Drug Administration decided not to require cardiac testing for every ADHD start, Dr. McGough said. More recent studies suggest that the risk is very small or absent.
"There are about a half dozen papers on this now," including one published recently and two large retrospective cohort studies published last year.
The current study examined the risk of cardiovascular events in a database of 171,126 youngsters aged 6-21 years. There were three new cardiac symptoms and less than one cardiac event per 1 million days of current stimulant use. Compared to the reference group, the adjusted odds ratio of cardiac events during current use was 1.18, and with past use, 0.93 (J. Am. Acad. Child Adolesc. Psychiatry 2012;51:147-56).
Last November, the New England Journal of Medicine published a cohort study of 1.2 million children and young adults, 373,667 of whom were currently using ADHD medications. There were 81 serious cardiovascular events, a rate of 3 per 100,000 person-years (N. Engl. J. Med. 2011;365:1896-904).
Another study appeared in Pediatrics. The cohort consisted of 241,417 ADHD medication users. No significant associations were found with sudden death, ventricular arrhythmia, or all-cause mortality. None of the strokes identified during exposed time to ADHD agents were validated (Pediatrics 2011;127:1102-10).
"I think it’s time we put an end to this brouhaha," Dr. McGough said.
Dr. McGough disclosed that he is on the advisory boards of NextWave Pharmaceuticals, Noven Pharmaceuticals, Shiongi, Shire Pharmaceuticals, and Supernus Pharmaceuticals. He also is a consultant for Alexa Pharmaceuticals and MedImmune.
* Correction 2/27/2012: The technology mentioned in this sentence previously was misidentified.
NEW YORK – An electrocadiogram* probably isn’t necessary for most youngsters starting a medication for attention-deficit/hyperactivity disorder.
Some easy screening questions should be enough to identify any children who have cardiovascular risks that a stimulant could exacerbate, Dr. James J. McGough said at a psychopharmacology update sponsored by the American Academy of Child and Adolescent Psychiatry.
"I am going to suggest that we end this neurosis" about cardiovascular risks during ADHD treatment. "The questions to ask before starting a kid out on any psychotropic medication are the same you would ask any child who is getting a sports physical," said Dr. McGough, chief of staff of the Resnick Neuropsychiatric Hospital at the University of California, Los Angeles.
The biggest fear is sudden cardiovascular death – an event usually associated with sport. "I don’t think a month goes by that I don’t hear about a soccer player, cheerleader, or basketball player who has had a sudden death. It’s tragic, but it happens. The bottom line is that there is a three times greater risk of sudden death associated with sports than with being on a stimulant."
In light of this statistic, he said: "There would be more sense in doing an EKG on every high school athlete, but no one is calling for that."
The key questions that should be in every ADHD assessment focus on family and personal history:
• Have you ever fainted while exercising?
• Is there a family history of sudden cardiac death?
• Is there any cardiac abnormality or a heart murmur?
If the answer to any of these is "yes," the patient should see a pediatric cardiologist before starting a stimulant medication. Otherwise, Dr. McGough said, "The benefit of an EKG is really not worth the cost."
The American Heart Association raised concerns about cardiac death and ADHD medications in 2008, when it recommended that every child have an EKG read by a pediatric cardiologist before beginning stimulant medication. However, the group noted, although the test was preferred, it should not be mandatory (Circ. 2008;117:2407-23).
A 2009 postmortem case-control study seemed to bolster the recommendation (Am. J. Psychiatry 2009;166:992-1001).
The authors examined amphetamine exposure among 1,128 children aged 7-19 years, half of whom had a sudden unexplained death and half of whom died in motor vehicle accidents. Ten of those in the sudden death group had amphetamine exposure compared to two in the accident group – a significant difference. The authors concluded that amphetamines could increase the risk of sudden death.
Despite this study, the Food and Drug Administration decided not to require cardiac testing for every ADHD start, Dr. McGough said. More recent studies suggest that the risk is very small or absent.
"There are about a half dozen papers on this now," including one published recently and two large retrospective cohort studies published last year.
The current study examined the risk of cardiovascular events in a database of 171,126 youngsters aged 6-21 years. There were three new cardiac symptoms and less than one cardiac event per 1 million days of current stimulant use. Compared to the reference group, the adjusted odds ratio of cardiac events during current use was 1.18, and with past use, 0.93 (J. Am. Acad. Child Adolesc. Psychiatry 2012;51:147-56).
Last November, the New England Journal of Medicine published a cohort study of 1.2 million children and young adults, 373,667 of whom were currently using ADHD medications. There were 81 serious cardiovascular events, a rate of 3 per 100,000 person-years (N. Engl. J. Med. 2011;365:1896-904).
Another study appeared in Pediatrics. The cohort consisted of 241,417 ADHD medication users. No significant associations were found with sudden death, ventricular arrhythmia, or all-cause mortality. None of the strokes identified during exposed time to ADHD agents were validated (Pediatrics 2011;127:1102-10).
"I think it’s time we put an end to this brouhaha," Dr. McGough said.
Dr. McGough disclosed that he is on the advisory boards of NextWave Pharmaceuticals, Noven Pharmaceuticals, Shiongi, Shire Pharmaceuticals, and Supernus Pharmaceuticals. He also is a consultant for Alexa Pharmaceuticals and MedImmune.
* Correction 2/27/2012: The technology mentioned in this sentence previously was misidentified.
EXPERT ANALYSIS FROM A PSYCHOPHARMACOLOGY UPDATE SPONSORED BY THE AMERICAN ACADEMY OF CHILD AND ADOLESCENT PSYCHIARISTS