User login
Urologists Back PSA Screening; Rail Against USPSTF's Position
ATLANTA – U.S. Preventive Services Task Force recommendations against using the prostate specific antigen to universally screen men for prostate cancer are unlikely to be last word on the controversial topic– as evidenced by several thousand irked urologists at the annual meeting of the American Urological Association.
Corridors and elevators at the AUA meeting were abuzz with physicians railing against the USPSTF’s finalized recommendation. Scores more piled into a standing-room only auditorium to collectively air their grievances at a town hall meeting convened on PSA screening.
AUA spokesman Dr. Ian Thompson of the University of Texas Health Sciences Center, San Antonio, opened the forum asking: "How many of you do not support the new screening recommendation?" Nearly every member raised a hand – and some added a vocal exclamation point to their reply. Less than 30 hands shot up in support of the USPSTF’s stance against universal PSA screening.
The focal point: whether the USPSTF correctly interpreted the two pivotal trials on which it based its latest recommendations – the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial (N. Engl. J. Med. 2009;360:1310-9) and the European Randomized Study for Prostate Cancer (ERSCP) (N. Engl. J. Med. 2012;366:981-90).
The USPSTF’s new position replaces the 2008 recommendations, which cited insufficient evidence to support improved health outcomes associated with prostate cancer screening for men younger than 75 years and more conclusive evidence pointing to more harm than benefit for men aged 75 years or older.
Dr. Fritz Schroder, a primary investigator on the ERSCP, noted that in fact an updated analysis presents a much stronger case for screening than in 2009, when the study was first published. Initially, the study found a 20% decreased risk of prostate cancer mortality, but also a high rate of overdiagnosis. However, it also reflected a relatively short follow-up of 9 years* – not nearly long enough to fully evaluate the effect of screening, said the professor of urology at Erasmus University in Rotterdam, the Netherlands.
"Our subjects were about 63 [years old] when they entered, with an average life expectancy of 15 more years. So we have to wait. We need at least 15 years of data, which we won’t see for several more years. At this point, only 30% of men in both arms have died from anything, and lots of things can happen in the years ahead before we reach that other 70%."
The latest update to ERSCP showed an ever-increasing separation of the mortality curves, with a relative reduction in risk of prostate cancer death of 21% in the PSA screening group. After adjustment, the relative risk reduction was 29% in the screening group. "While the increase [to 29%] is not large, the statistical significance has improved considerably," he said, from a P-value of .04 to a P value of .001. "I believe the argument in favor of screening has more strength, mainly because we now have a 29% reduction in mortality and a highly significant difference."
Ruth Etzioni, Ph.D., agreed. "In any screening trial, the benefit grows over time," said the biostatistician who specialized in modeling prostate cancer outcomes at the Fred Hutchinson Cancer Research Center at the University of Washington, Seattle. "Recognizing that we need to go beyond the data to get numbers relevant for policy making, we must conclude that the picture [in favor of screening], is somewhat more positive than the short-term outcomes suggested."
USPSTF member Timothy J. Wilt did not deviate from his group’s position. He said the task force took the updated evidence into consideration while shaping the final document.
"While there’s no magic number that makes screening beneficial, we do look at the other recommendations we’ve made," with breast cancer and colorectal cancer screening. "For prostate cancer, if there’s any benefit at all, it’s extremely small, saving less that 1 in 1,000 men screened from prostate cancer death, but there are frequent, severe harms," Dr. Wilt said.
The AUA panel agreed that the current PSA test is far from perfect. However, until something better comes along, smarter screening is better than abandoning screening, said Dr. John T. Wei, a urologist at the University of Michigan, Ann Arbor. "It’s clearly helped clinically, but a great number of men also suffer because of it. We need new, better, and more-specific tests so all this harm can be reduced."
Dr. Thompson expressed his own frustration in an interview after the meeting.
"We are just so disappointed that the task force reached this decision. There are foibles with the PSA screen, and we all know that most men will have some abnormality if you look hard enough. But it’s also true that prostate cancer is one of the most common causes of cancer death in men."
PSA testing is a fine line that physician and patient must walk together, he said. "Every single organization in the world has said mass screening is inappropriate and that it should be done in the context of a single man and his understanding of his own risk factors and the risks and benefits of screening. The problem with this recommendation is that it takes all the nuance and complexity of this interaction and simply says, ‘Don’t have the discussion.’ The American public makes risk-based decisions all the time, after looking at the pros and cons of an issue – and I think people can weigh this out themselves without having someone else to say ‘Follow my priorities instead of your own.’ "
Dr. Brian McNeil, chief of urology at the University Hospital Brooklyn, N.Y., said that individual risk assessment is the key to using PSA wisely. During the meeting, Dr. McNeil presented a case series of 148 black men in their 60s, all of whom came to him with symptoms that turned out to be metastatic prostate cancer.
"A white male with no family history and no other risk factors can probably safely delay a PSA test until age 50," he said in an interview. "But some men you will want to begin screening at age 45 or even age 40."
The prostate cancer patient's quality of life is something that "gets lost in this screening argument," he said. "Nobody ever talks about how it feels to have a bony metastasis or a big cancerous prostate obstructing your bladder. Screening can catch these cancers earlier so we can save our patients from this kind of thing."
Dr. Dilip Doctor, who practices in Flushing, N.Y., said doctors have a "moral and legal obligation" to screen for prostate cancer. If physicians neglect to provide this inexpensive test, he said, they could experience some legal difficulties.
"No patient is going to forgive you for missing his cancer that could have been diagnosed with a PSA," he said in an interview. "You might read this article and think about it. But listen, those guys are not going to come down and defend you in a lawsuit. The only thing to protect you is to practice good medicine and that means doing the only test available and that is a PSA. If you don’t do it, it’s negligent."
None of the physicians interviewed for this article declared having any financial relationships.
* Correction (5/25/12): An earlier posted version of this story incorrectly stated the follow-up period of the original ERSCP study published in 2009. At that time, the follow-up period was 9 years.
ATLANTA – U.S. Preventive Services Task Force recommendations against using the prostate specific antigen to universally screen men for prostate cancer are unlikely to be last word on the controversial topic– as evidenced by several thousand irked urologists at the annual meeting of the American Urological Association.
Corridors and elevators at the AUA meeting were abuzz with physicians railing against the USPSTF’s finalized recommendation. Scores more piled into a standing-room only auditorium to collectively air their grievances at a town hall meeting convened on PSA screening.
AUA spokesman Dr. Ian Thompson of the University of Texas Health Sciences Center, San Antonio, opened the forum asking: "How many of you do not support the new screening recommendation?" Nearly every member raised a hand – and some added a vocal exclamation point to their reply. Less than 30 hands shot up in support of the USPSTF’s stance against universal PSA screening.
The focal point: whether the USPSTF correctly interpreted the two pivotal trials on which it based its latest recommendations – the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial (N. Engl. J. Med. 2009;360:1310-9) and the European Randomized Study for Prostate Cancer (ERSCP) (N. Engl. J. Med. 2012;366:981-90).
The USPSTF’s new position replaces the 2008 recommendations, which cited insufficient evidence to support improved health outcomes associated with prostate cancer screening for men younger than 75 years and more conclusive evidence pointing to more harm than benefit for men aged 75 years or older.
Dr. Fritz Schroder, a primary investigator on the ERSCP, noted that in fact an updated analysis presents a much stronger case for screening than in 2009, when the study was first published. Initially, the study found a 20% decreased risk of prostate cancer mortality, but also a high rate of overdiagnosis. However, it also reflected a relatively short follow-up of 9 years* – not nearly long enough to fully evaluate the effect of screening, said the professor of urology at Erasmus University in Rotterdam, the Netherlands.
"Our subjects were about 63 [years old] when they entered, with an average life expectancy of 15 more years. So we have to wait. We need at least 15 years of data, which we won’t see for several more years. At this point, only 30% of men in both arms have died from anything, and lots of things can happen in the years ahead before we reach that other 70%."
The latest update to ERSCP showed an ever-increasing separation of the mortality curves, with a relative reduction in risk of prostate cancer death of 21% in the PSA screening group. After adjustment, the relative risk reduction was 29% in the screening group. "While the increase [to 29%] is not large, the statistical significance has improved considerably," he said, from a P-value of .04 to a P value of .001. "I believe the argument in favor of screening has more strength, mainly because we now have a 29% reduction in mortality and a highly significant difference."
Ruth Etzioni, Ph.D., agreed. "In any screening trial, the benefit grows over time," said the biostatistician who specialized in modeling prostate cancer outcomes at the Fred Hutchinson Cancer Research Center at the University of Washington, Seattle. "Recognizing that we need to go beyond the data to get numbers relevant for policy making, we must conclude that the picture [in favor of screening], is somewhat more positive than the short-term outcomes suggested."
USPSTF member Timothy J. Wilt did not deviate from his group’s position. He said the task force took the updated evidence into consideration while shaping the final document.
"While there’s no magic number that makes screening beneficial, we do look at the other recommendations we’ve made," with breast cancer and colorectal cancer screening. "For prostate cancer, if there’s any benefit at all, it’s extremely small, saving less that 1 in 1,000 men screened from prostate cancer death, but there are frequent, severe harms," Dr. Wilt said.
The AUA panel agreed that the current PSA test is far from perfect. However, until something better comes along, smarter screening is better than abandoning screening, said Dr. John T. Wei, a urologist at the University of Michigan, Ann Arbor. "It’s clearly helped clinically, but a great number of men also suffer because of it. We need new, better, and more-specific tests so all this harm can be reduced."
Dr. Thompson expressed his own frustration in an interview after the meeting.
"We are just so disappointed that the task force reached this decision. There are foibles with the PSA screen, and we all know that most men will have some abnormality if you look hard enough. But it’s also true that prostate cancer is one of the most common causes of cancer death in men."
PSA testing is a fine line that physician and patient must walk together, he said. "Every single organization in the world has said mass screening is inappropriate and that it should be done in the context of a single man and his understanding of his own risk factors and the risks and benefits of screening. The problem with this recommendation is that it takes all the nuance and complexity of this interaction and simply says, ‘Don’t have the discussion.’ The American public makes risk-based decisions all the time, after looking at the pros and cons of an issue – and I think people can weigh this out themselves without having someone else to say ‘Follow my priorities instead of your own.’ "
Dr. Brian McNeil, chief of urology at the University Hospital Brooklyn, N.Y., said that individual risk assessment is the key to using PSA wisely. During the meeting, Dr. McNeil presented a case series of 148 black men in their 60s, all of whom came to him with symptoms that turned out to be metastatic prostate cancer.
"A white male with no family history and no other risk factors can probably safely delay a PSA test until age 50," he said in an interview. "But some men you will want to begin screening at age 45 or even age 40."
The prostate cancer patient's quality of life is something that "gets lost in this screening argument," he said. "Nobody ever talks about how it feels to have a bony metastasis or a big cancerous prostate obstructing your bladder. Screening can catch these cancers earlier so we can save our patients from this kind of thing."
Dr. Dilip Doctor, who practices in Flushing, N.Y., said doctors have a "moral and legal obligation" to screen for prostate cancer. If physicians neglect to provide this inexpensive test, he said, they could experience some legal difficulties.
"No patient is going to forgive you for missing his cancer that could have been diagnosed with a PSA," he said in an interview. "You might read this article and think about it. But listen, those guys are not going to come down and defend you in a lawsuit. The only thing to protect you is to practice good medicine and that means doing the only test available and that is a PSA. If you don’t do it, it’s negligent."
None of the physicians interviewed for this article declared having any financial relationships.
* Correction (5/25/12): An earlier posted version of this story incorrectly stated the follow-up period of the original ERSCP study published in 2009. At that time, the follow-up period was 9 years.
ATLANTA – U.S. Preventive Services Task Force recommendations against using the prostate specific antigen to universally screen men for prostate cancer are unlikely to be last word on the controversial topic– as evidenced by several thousand irked urologists at the annual meeting of the American Urological Association.
Corridors and elevators at the AUA meeting were abuzz with physicians railing against the USPSTF’s finalized recommendation. Scores more piled into a standing-room only auditorium to collectively air their grievances at a town hall meeting convened on PSA screening.
AUA spokesman Dr. Ian Thompson of the University of Texas Health Sciences Center, San Antonio, opened the forum asking: "How many of you do not support the new screening recommendation?" Nearly every member raised a hand – and some added a vocal exclamation point to their reply. Less than 30 hands shot up in support of the USPSTF’s stance against universal PSA screening.
The focal point: whether the USPSTF correctly interpreted the two pivotal trials on which it based its latest recommendations – the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial (N. Engl. J. Med. 2009;360:1310-9) and the European Randomized Study for Prostate Cancer (ERSCP) (N. Engl. J. Med. 2012;366:981-90).
The USPSTF’s new position replaces the 2008 recommendations, which cited insufficient evidence to support improved health outcomes associated with prostate cancer screening for men younger than 75 years and more conclusive evidence pointing to more harm than benefit for men aged 75 years or older.
Dr. Fritz Schroder, a primary investigator on the ERSCP, noted that in fact an updated analysis presents a much stronger case for screening than in 2009, when the study was first published. Initially, the study found a 20% decreased risk of prostate cancer mortality, but also a high rate of overdiagnosis. However, it also reflected a relatively short follow-up of 9 years* – not nearly long enough to fully evaluate the effect of screening, said the professor of urology at Erasmus University in Rotterdam, the Netherlands.
"Our subjects were about 63 [years old] when they entered, with an average life expectancy of 15 more years. So we have to wait. We need at least 15 years of data, which we won’t see for several more years. At this point, only 30% of men in both arms have died from anything, and lots of things can happen in the years ahead before we reach that other 70%."
The latest update to ERSCP showed an ever-increasing separation of the mortality curves, with a relative reduction in risk of prostate cancer death of 21% in the PSA screening group. After adjustment, the relative risk reduction was 29% in the screening group. "While the increase [to 29%] is not large, the statistical significance has improved considerably," he said, from a P-value of .04 to a P value of .001. "I believe the argument in favor of screening has more strength, mainly because we now have a 29% reduction in mortality and a highly significant difference."
Ruth Etzioni, Ph.D., agreed. "In any screening trial, the benefit grows over time," said the biostatistician who specialized in modeling prostate cancer outcomes at the Fred Hutchinson Cancer Research Center at the University of Washington, Seattle. "Recognizing that we need to go beyond the data to get numbers relevant for policy making, we must conclude that the picture [in favor of screening], is somewhat more positive than the short-term outcomes suggested."
USPSTF member Timothy J. Wilt did not deviate from his group’s position. He said the task force took the updated evidence into consideration while shaping the final document.
"While there’s no magic number that makes screening beneficial, we do look at the other recommendations we’ve made," with breast cancer and colorectal cancer screening. "For prostate cancer, if there’s any benefit at all, it’s extremely small, saving less that 1 in 1,000 men screened from prostate cancer death, but there are frequent, severe harms," Dr. Wilt said.
The AUA panel agreed that the current PSA test is far from perfect. However, until something better comes along, smarter screening is better than abandoning screening, said Dr. John T. Wei, a urologist at the University of Michigan, Ann Arbor. "It’s clearly helped clinically, but a great number of men also suffer because of it. We need new, better, and more-specific tests so all this harm can be reduced."
Dr. Thompson expressed his own frustration in an interview after the meeting.
"We are just so disappointed that the task force reached this decision. There are foibles with the PSA screen, and we all know that most men will have some abnormality if you look hard enough. But it’s also true that prostate cancer is one of the most common causes of cancer death in men."
PSA testing is a fine line that physician and patient must walk together, he said. "Every single organization in the world has said mass screening is inappropriate and that it should be done in the context of a single man and his understanding of his own risk factors and the risks and benefits of screening. The problem with this recommendation is that it takes all the nuance and complexity of this interaction and simply says, ‘Don’t have the discussion.’ The American public makes risk-based decisions all the time, after looking at the pros and cons of an issue – and I think people can weigh this out themselves without having someone else to say ‘Follow my priorities instead of your own.’ "
Dr. Brian McNeil, chief of urology at the University Hospital Brooklyn, N.Y., said that individual risk assessment is the key to using PSA wisely. During the meeting, Dr. McNeil presented a case series of 148 black men in their 60s, all of whom came to him with symptoms that turned out to be metastatic prostate cancer.
"A white male with no family history and no other risk factors can probably safely delay a PSA test until age 50," he said in an interview. "But some men you will want to begin screening at age 45 or even age 40."
The prostate cancer patient's quality of life is something that "gets lost in this screening argument," he said. "Nobody ever talks about how it feels to have a bony metastasis or a big cancerous prostate obstructing your bladder. Screening can catch these cancers earlier so we can save our patients from this kind of thing."
Dr. Dilip Doctor, who practices in Flushing, N.Y., said doctors have a "moral and legal obligation" to screen for prostate cancer. If physicians neglect to provide this inexpensive test, he said, they could experience some legal difficulties.
"No patient is going to forgive you for missing his cancer that could have been diagnosed with a PSA," he said in an interview. "You might read this article and think about it. But listen, those guys are not going to come down and defend you in a lawsuit. The only thing to protect you is to practice good medicine and that means doing the only test available and that is a PSA. If you don’t do it, it’s negligent."
None of the physicians interviewed for this article declared having any financial relationships.
* Correction (5/25/12): An earlier posted version of this story incorrectly stated the follow-up period of the original ERSCP study published in 2009. At that time, the follow-up period was 9 years.
FROM THE ANNUAL MEETING OF THE AMERICAN UROLOGICAL ASSOCIATION
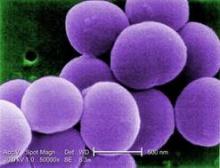
Chlorhexidine-Resistant S. aureus Infections on the Rise
BOSTON – It’s no surprise that antibiotic resistance continues to grow, but now some Staphylococcus aureus are showing off a new trick – resistance to chlorhexidine, the antiseptic relied upon to prevent staph infections.
In a review of isolates from pediatric cancer patients, an increasing number of S. aureus became resistant to the antiseptic, Dr. J. Chase McNeil said at the annual meeting of the Pediatric Academic Societies. The jump from susceptible to resistant occurred around 2006 – 2 years after the Texas Children’s Hospital began using chlorhexidine in the weekly central line dressing changes for its cancer patients, and a year after the facility introduced chlorhexidine mouthwash up to four times each day for patients with acute myeloid leukemia.
"It’s very interesting to see this upward trend [in resistance]," he said in an interview. "Before 2007, we had none, and since then we’ve seen an increase every year."
While the clinical significance of this phenomenon remains unclear, it’s very clear that the bacteria are changing, he added.
Dr. Chase reported a review of a prospectively acquired data set, which includes all children treated at the university’s pediatric oncology facility. He and his coinvestigators looked for infections caused by S. aureus and their related complications. They also assessed the emergence of staph isolates showing the qacA/B gene, which confers a higher minimum inhibitory concentration and minimum bactericidal concentration to chlorhexidine and other quaternary ammonium compound (QAC) antiseptics. The study also examined rates of methicillin resistance.
From 2001 through 2011, 213 S. aureus infections developed in 179 patients. Infections were most commonly associated with acute lymphoblastic leukemia (43%). Other cancers were primary central nervous system malignancies (11%), acute myeloid leukemia (AML), and hematopoietic stem cell transplant (HSCT) patients (16%). Among the infections were bacteremias (40%), skin and soft tissue infections (36%) and surgical site infections (15%).
Most of the infections were methicillin-susceptible S. aureus (147); the remaining infections were methicillin resistant. Most of the methicillin-resistant S. aureus isolates were also the USA300 strain, a particularly resistant strain associated with serious, rapidly progressing infections. The USA300 isolates were responsible for 59% of the skin/soft tissue infections and 25% of the bacteremias.
Overall, 8% of the isolates were also qacA/B positive. These were more likely to be resistant to ciprofloxacin than the qacA/B-negative isolates (50% vs. 15%).
Chlorhexidine-cleansed dressing changes and catheter cleansings began in 2004 in response to a sharp increase in staph infections in AML and HSCT patients, Dr. McNeil said. These infections did drop precipitously in the following years. In 2005, AML patients began using a chlorhexidine mouthwash up to four times each day.
However, in 2006, just as the staph infection rate was improving, qacA/B resistance suddenly appeared. By 2009, 10% of infections were positive for qacA/B, and by 2011, this had risen to about 22%.
"We can’t say if the change in the microbiology of these is caused by anything, or causing anything significant, but there is definitely a temporal association," he said.
Chlorhexidine continues to be relied upon in the oncology ward, he said. By 2011, daily chlorhexidine baths became part of the standard care for neutropenic AML patients.
Among the entire group of patients with infections, 19% (37) developed a total of 58 complications. Bacteremias were associated with most of the complications (70%). A multivariate analysis showed a significant association between complicated bacteremias and AMC patients with a low lymphocyte count.
Thirteen patients with bacteremia also developed pulmonary nodules. All of these were associated with methicillin-susceptible S. aureus isolates. The nodules developed rapidly, appearing a median of 5 days after bacteremia onset. Six patients were biopsied, with S. aureus cultured from five. One nodule was a metastatic tumor. One other patient with nodules died before culture. This patient had an invasive pulmonary fungal infection.
Dr. McNeil said several factors were significantly associated with staph infections and pulmonary nodules, including HSCT, a low lymphocyte count, and low platelets.
"This isn’t surprising because it’s well known that children with malignancies are at a high risk for staph disease because of their immune compromise and a high exposure to empiric antibiotics and antiseptics."
Dr. McNeil said he had no financial declarations.
BOSTON – It’s no surprise that antibiotic resistance continues to grow, but now some Staphylococcus aureus are showing off a new trick – resistance to chlorhexidine, the antiseptic relied upon to prevent staph infections.
In a review of isolates from pediatric cancer patients, an increasing number of S. aureus became resistant to the antiseptic, Dr. J. Chase McNeil said at the annual meeting of the Pediatric Academic Societies. The jump from susceptible to resistant occurred around 2006 – 2 years after the Texas Children’s Hospital began using chlorhexidine in the weekly central line dressing changes for its cancer patients, and a year after the facility introduced chlorhexidine mouthwash up to four times each day for patients with acute myeloid leukemia.
"It’s very interesting to see this upward trend [in resistance]," he said in an interview. "Before 2007, we had none, and since then we’ve seen an increase every year."
While the clinical significance of this phenomenon remains unclear, it’s very clear that the bacteria are changing, he added.
Dr. Chase reported a review of a prospectively acquired data set, which includes all children treated at the university’s pediatric oncology facility. He and his coinvestigators looked for infections caused by S. aureus and their related complications. They also assessed the emergence of staph isolates showing the qacA/B gene, which confers a higher minimum inhibitory concentration and minimum bactericidal concentration to chlorhexidine and other quaternary ammonium compound (QAC) antiseptics. The study also examined rates of methicillin resistance.
From 2001 through 2011, 213 S. aureus infections developed in 179 patients. Infections were most commonly associated with acute lymphoblastic leukemia (43%). Other cancers were primary central nervous system malignancies (11%), acute myeloid leukemia (AML), and hematopoietic stem cell transplant (HSCT) patients (16%). Among the infections were bacteremias (40%), skin and soft tissue infections (36%) and surgical site infections (15%).
Most of the infections were methicillin-susceptible S. aureus (147); the remaining infections were methicillin resistant. Most of the methicillin-resistant S. aureus isolates were also the USA300 strain, a particularly resistant strain associated with serious, rapidly progressing infections. The USA300 isolates were responsible for 59% of the skin/soft tissue infections and 25% of the bacteremias.
Overall, 8% of the isolates were also qacA/B positive. These were more likely to be resistant to ciprofloxacin than the qacA/B-negative isolates (50% vs. 15%).
Chlorhexidine-cleansed dressing changes and catheter cleansings began in 2004 in response to a sharp increase in staph infections in AML and HSCT patients, Dr. McNeil said. These infections did drop precipitously in the following years. In 2005, AML patients began using a chlorhexidine mouthwash up to four times each day.
However, in 2006, just as the staph infection rate was improving, qacA/B resistance suddenly appeared. By 2009, 10% of infections were positive for qacA/B, and by 2011, this had risen to about 22%.
"We can’t say if the change in the microbiology of these is caused by anything, or causing anything significant, but there is definitely a temporal association," he said.
Chlorhexidine continues to be relied upon in the oncology ward, he said. By 2011, daily chlorhexidine baths became part of the standard care for neutropenic AML patients.
Among the entire group of patients with infections, 19% (37) developed a total of 58 complications. Bacteremias were associated with most of the complications (70%). A multivariate analysis showed a significant association between complicated bacteremias and AMC patients with a low lymphocyte count.
Thirteen patients with bacteremia also developed pulmonary nodules. All of these were associated with methicillin-susceptible S. aureus isolates. The nodules developed rapidly, appearing a median of 5 days after bacteremia onset. Six patients were biopsied, with S. aureus cultured from five. One nodule was a metastatic tumor. One other patient with nodules died before culture. This patient had an invasive pulmonary fungal infection.
Dr. McNeil said several factors were significantly associated with staph infections and pulmonary nodules, including HSCT, a low lymphocyte count, and low platelets.
"This isn’t surprising because it’s well known that children with malignancies are at a high risk for staph disease because of their immune compromise and a high exposure to empiric antibiotics and antiseptics."
Dr. McNeil said he had no financial declarations.
BOSTON – It’s no surprise that antibiotic resistance continues to grow, but now some Staphylococcus aureus are showing off a new trick – resistance to chlorhexidine, the antiseptic relied upon to prevent staph infections.
In a review of isolates from pediatric cancer patients, an increasing number of S. aureus became resistant to the antiseptic, Dr. J. Chase McNeil said at the annual meeting of the Pediatric Academic Societies. The jump from susceptible to resistant occurred around 2006 – 2 years after the Texas Children’s Hospital began using chlorhexidine in the weekly central line dressing changes for its cancer patients, and a year after the facility introduced chlorhexidine mouthwash up to four times each day for patients with acute myeloid leukemia.
"It’s very interesting to see this upward trend [in resistance]," he said in an interview. "Before 2007, we had none, and since then we’ve seen an increase every year."
While the clinical significance of this phenomenon remains unclear, it’s very clear that the bacteria are changing, he added.
Dr. Chase reported a review of a prospectively acquired data set, which includes all children treated at the university’s pediatric oncology facility. He and his coinvestigators looked for infections caused by S. aureus and their related complications. They also assessed the emergence of staph isolates showing the qacA/B gene, which confers a higher minimum inhibitory concentration and minimum bactericidal concentration to chlorhexidine and other quaternary ammonium compound (QAC) antiseptics. The study also examined rates of methicillin resistance.
From 2001 through 2011, 213 S. aureus infections developed in 179 patients. Infections were most commonly associated with acute lymphoblastic leukemia (43%). Other cancers were primary central nervous system malignancies (11%), acute myeloid leukemia (AML), and hematopoietic stem cell transplant (HSCT) patients (16%). Among the infections were bacteremias (40%), skin and soft tissue infections (36%) and surgical site infections (15%).
Most of the infections were methicillin-susceptible S. aureus (147); the remaining infections were methicillin resistant. Most of the methicillin-resistant S. aureus isolates were also the USA300 strain, a particularly resistant strain associated with serious, rapidly progressing infections. The USA300 isolates were responsible for 59% of the skin/soft tissue infections and 25% of the bacteremias.
Overall, 8% of the isolates were also qacA/B positive. These were more likely to be resistant to ciprofloxacin than the qacA/B-negative isolates (50% vs. 15%).
Chlorhexidine-cleansed dressing changes and catheter cleansings began in 2004 in response to a sharp increase in staph infections in AML and HSCT patients, Dr. McNeil said. These infections did drop precipitously in the following years. In 2005, AML patients began using a chlorhexidine mouthwash up to four times each day.
However, in 2006, just as the staph infection rate was improving, qacA/B resistance suddenly appeared. By 2009, 10% of infections were positive for qacA/B, and by 2011, this had risen to about 22%.
"We can’t say if the change in the microbiology of these is caused by anything, or causing anything significant, but there is definitely a temporal association," he said.
Chlorhexidine continues to be relied upon in the oncology ward, he said. By 2011, daily chlorhexidine baths became part of the standard care for neutropenic AML patients.
Among the entire group of patients with infections, 19% (37) developed a total of 58 complications. Bacteremias were associated with most of the complications (70%). A multivariate analysis showed a significant association between complicated bacteremias and AMC patients with a low lymphocyte count.
Thirteen patients with bacteremia also developed pulmonary nodules. All of these were associated with methicillin-susceptible S. aureus isolates. The nodules developed rapidly, appearing a median of 5 days after bacteremia onset. Six patients were biopsied, with S. aureus cultured from five. One nodule was a metastatic tumor. One other patient with nodules died before culture. This patient had an invasive pulmonary fungal infection.
Dr. McNeil said several factors were significantly associated with staph infections and pulmonary nodules, including HSCT, a low lymphocyte count, and low platelets.
"This isn’t surprising because it’s well known that children with malignancies are at a high risk for staph disease because of their immune compromise and a high exposure to empiric antibiotics and antiseptics."
Dr. McNeil said he had no financial declarations.
FROM THE ANNUAL MEETING OF THE PEDIATRIC ACADEMIC SOCIETIES
Major Finding: In a pediatric oncology ward, chlorhexidine-resistant S. aureus–associated infections increased from 0 in 2007 to more than 20% of staph infections in 2011.
Data Source: The data were drawn from an 11-year prospectively acquired series.
Disclosures: Dr. J. Chase McNeil had no financial disclosures.
PSA Level Could Determine Screening Frequency in 40s
ATLANTA – Men in their 40s with a low prostate specific antigen can probably safely delay additional testing for 10-15 years.
Young men with a higher baseline level, however, are twice as likely to develop prostate cancer over the same time period and should probably have their PSA tested at regular intervals, Dr. Christopher Weight said at the annual meeting of the American Urologic Association.
His prospective study of 268 men in their 40s showed that none of the men with a baseline PSA of 1.0 ng/mL or less developed high-risk disease by 10 years and only 3% developed it by 15 years. The findings could provide an effective way to risk-stratify young populations, reducing unnecessary testing and the consequences that sometimes follow it, he said.
"We have to admit that we overdiagnose and overtreat men," Dr. Weight said at a press briefing. "But there is danger in completely throwing out the PSA test. Testing men early can help us identify those who can safely delay additional testing and those who will benefit from more frequent tests."
Dr. Weight, a urology oncology fellow at the Mayo Clinic, Rochester, Minn., turned to the Olmsted County cohort for the study data. Since 1990, most of the residents in the county have received their medical care through the Mayo Clinic and its affiliate centers. A linked health records database provides information for long-term population-based studies.
His analysis included 268 men, all of whom had a baseline PSA drawn sometime during their 40s (median age 45 years). The men also had a transrectal ultrasound and digital rectal exam. They have been followed now for up to 20 years, with a median time of 16 years.
Among the cohort, 192 had a baseline PSA of 1.0 ng/mL or lower and 76 had a level of more than 1.0 ng/mL. There were no significant between-group differences in either family history or the results of the rectal exam.
Over the full follow-up period, men with the lower PSA level had a significantly lower risk of exceeding the age-specific cut points for PSA than did men with the higher levels (10% vs. 50%).
By the end of the follow-up period, there were six incident cases of prostate cancer in the low-PSA group, all of which were low-risk disease. This translated to an incidence rate of 1.6 per 1,000 patient/ years, with a mean of 15 years until diagnosis.
Twice as many men in the high-PSA group developed prostate cancer (12). Of these cases, 10 were low-risk disease and 2 high-risk. This translated into a rate of 8/1,000 patient-years, with a mean of 10 years to diagnosis.
The baseline measurement was fairly predictive of 15-year outcomes, Dr. Weight said. A cutoff of 1.0 ng/mL at the initial test had a sensitivity of 67% and a specificity of 74% for predicting the occurrence of prostate cancer. Changing the cutoff to 0.7 ng/mL on the initial test resulted in a sensitivity of 83% and a specificity of 46%
The results show that this single, early PSA level may be helpful in counseling patients about follow-up, said Dr. Scott Eggener, who moderated the briefing.
"The goals of any test are to identify the cohort of people most likely to benefit, and those people in whom screening can be limited to minimize the potential harms of the test," said Dr. Eggener, director of urology outcomes and translational research at the University of Chicago Medical Center. "If a young man has a very low PSA, we can feel comfortable in recommending that he have another test in several years, somewhat like what’s done with a screening colonoscopy. If the level is higher, this patient probably needs to be followed more frequently."
Neither Dr. Weight nor Dr. Eggener had any financial disclosures.
ATLANTA – Men in their 40s with a low prostate specific antigen can probably safely delay additional testing for 10-15 years.
Young men with a higher baseline level, however, are twice as likely to develop prostate cancer over the same time period and should probably have their PSA tested at regular intervals, Dr. Christopher Weight said at the annual meeting of the American Urologic Association.
His prospective study of 268 men in their 40s showed that none of the men with a baseline PSA of 1.0 ng/mL or less developed high-risk disease by 10 years and only 3% developed it by 15 years. The findings could provide an effective way to risk-stratify young populations, reducing unnecessary testing and the consequences that sometimes follow it, he said.
"We have to admit that we overdiagnose and overtreat men," Dr. Weight said at a press briefing. "But there is danger in completely throwing out the PSA test. Testing men early can help us identify those who can safely delay additional testing and those who will benefit from more frequent tests."
Dr. Weight, a urology oncology fellow at the Mayo Clinic, Rochester, Minn., turned to the Olmsted County cohort for the study data. Since 1990, most of the residents in the county have received their medical care through the Mayo Clinic and its affiliate centers. A linked health records database provides information for long-term population-based studies.
His analysis included 268 men, all of whom had a baseline PSA drawn sometime during their 40s (median age 45 years). The men also had a transrectal ultrasound and digital rectal exam. They have been followed now for up to 20 years, with a median time of 16 years.
Among the cohort, 192 had a baseline PSA of 1.0 ng/mL or lower and 76 had a level of more than 1.0 ng/mL. There were no significant between-group differences in either family history or the results of the rectal exam.
Over the full follow-up period, men with the lower PSA level had a significantly lower risk of exceeding the age-specific cut points for PSA than did men with the higher levels (10% vs. 50%).
By the end of the follow-up period, there were six incident cases of prostate cancer in the low-PSA group, all of which were low-risk disease. This translated to an incidence rate of 1.6 per 1,000 patient/ years, with a mean of 15 years until diagnosis.
Twice as many men in the high-PSA group developed prostate cancer (12). Of these cases, 10 were low-risk disease and 2 high-risk. This translated into a rate of 8/1,000 patient-years, with a mean of 10 years to diagnosis.
The baseline measurement was fairly predictive of 15-year outcomes, Dr. Weight said. A cutoff of 1.0 ng/mL at the initial test had a sensitivity of 67% and a specificity of 74% for predicting the occurrence of prostate cancer. Changing the cutoff to 0.7 ng/mL on the initial test resulted in a sensitivity of 83% and a specificity of 46%
The results show that this single, early PSA level may be helpful in counseling patients about follow-up, said Dr. Scott Eggener, who moderated the briefing.
"The goals of any test are to identify the cohort of people most likely to benefit, and those people in whom screening can be limited to minimize the potential harms of the test," said Dr. Eggener, director of urology outcomes and translational research at the University of Chicago Medical Center. "If a young man has a very low PSA, we can feel comfortable in recommending that he have another test in several years, somewhat like what’s done with a screening colonoscopy. If the level is higher, this patient probably needs to be followed more frequently."
Neither Dr. Weight nor Dr. Eggener had any financial disclosures.
ATLANTA – Men in their 40s with a low prostate specific antigen can probably safely delay additional testing for 10-15 years.
Young men with a higher baseline level, however, are twice as likely to develop prostate cancer over the same time period and should probably have their PSA tested at regular intervals, Dr. Christopher Weight said at the annual meeting of the American Urologic Association.
His prospective study of 268 men in their 40s showed that none of the men with a baseline PSA of 1.0 ng/mL or less developed high-risk disease by 10 years and only 3% developed it by 15 years. The findings could provide an effective way to risk-stratify young populations, reducing unnecessary testing and the consequences that sometimes follow it, he said.
"We have to admit that we overdiagnose and overtreat men," Dr. Weight said at a press briefing. "But there is danger in completely throwing out the PSA test. Testing men early can help us identify those who can safely delay additional testing and those who will benefit from more frequent tests."
Dr. Weight, a urology oncology fellow at the Mayo Clinic, Rochester, Minn., turned to the Olmsted County cohort for the study data. Since 1990, most of the residents in the county have received their medical care through the Mayo Clinic and its affiliate centers. A linked health records database provides information for long-term population-based studies.
His analysis included 268 men, all of whom had a baseline PSA drawn sometime during their 40s (median age 45 years). The men also had a transrectal ultrasound and digital rectal exam. They have been followed now for up to 20 years, with a median time of 16 years.
Among the cohort, 192 had a baseline PSA of 1.0 ng/mL or lower and 76 had a level of more than 1.0 ng/mL. There were no significant between-group differences in either family history or the results of the rectal exam.
Over the full follow-up period, men with the lower PSA level had a significantly lower risk of exceeding the age-specific cut points for PSA than did men with the higher levels (10% vs. 50%).
By the end of the follow-up period, there were six incident cases of prostate cancer in the low-PSA group, all of which were low-risk disease. This translated to an incidence rate of 1.6 per 1,000 patient/ years, with a mean of 15 years until diagnosis.
Twice as many men in the high-PSA group developed prostate cancer (12). Of these cases, 10 were low-risk disease and 2 high-risk. This translated into a rate of 8/1,000 patient-years, with a mean of 10 years to diagnosis.
The baseline measurement was fairly predictive of 15-year outcomes, Dr. Weight said. A cutoff of 1.0 ng/mL at the initial test had a sensitivity of 67% and a specificity of 74% for predicting the occurrence of prostate cancer. Changing the cutoff to 0.7 ng/mL on the initial test resulted in a sensitivity of 83% and a specificity of 46%
The results show that this single, early PSA level may be helpful in counseling patients about follow-up, said Dr. Scott Eggener, who moderated the briefing.
"The goals of any test are to identify the cohort of people most likely to benefit, and those people in whom screening can be limited to minimize the potential harms of the test," said Dr. Eggener, director of urology outcomes and translational research at the University of Chicago Medical Center. "If a young man has a very low PSA, we can feel comfortable in recommending that he have another test in several years, somewhat like what’s done with a screening colonoscopy. If the level is higher, this patient probably needs to be followed more frequently."
Neither Dr. Weight nor Dr. Eggener had any financial disclosures.
FROM THE ANNUAL MEETING OF THE AMERICAN UROLOGICAL ASSOCIATION
Readmission 40% More Likely After Hospital-Acquired Infections
Hospitalized patients who pick up a nosocomial infection are 40% more likely to be readmitted within a year of discharge than patients who are not infected during their stay.
Ramped-up infection control could benefit not only patients but the hospital itself, Jon Furuno, Ph.D., and his colleagues wrote in the June issue of Infection Control and Hospital Epidemiology.
"Efforts to reduce healthcare-associated infections may have the additional benefit of reducing the incidence of hospital readmission and increased healthcare costs," wrote Dr. Furuno, an infectious disease specialist at the University of Maryland, Baltimore, and Oregon Health and Science University, Portland, and his coauthors. "Furthermore, patients with a positive clinical culture result obtained more than 48 hours after hospital admission could be targeted to receive additional discharge planning resources designed to reduce the likelihood of hospital readmission" (Infect. Contr. Hosp. Epidem. 2012;33:539-44 [doi: 10.1086/665725]).
Dr. Furuno and his associates examined inpatient infections from Jan. 1, 2001, to Dec. 31, 2008, at the University of Maryland Medical Center. The team focused on methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococci (VRE), and Clostridium difficile infections. The primary end point was time to readmission.
The study group included 136,513 admissions over the 8-year period. The patients’ mean age was 51 years, and the mean length of stay was 6 days (range, 3-13 days).
An infection was deemed to be hospital-acquired if a positive culture was obtained more than 48 hours after admission.
Only 3% of the study group had a positive culture that was hospital acquired. MRSA was cultured from 49% of the patients, C. difficile from 35%, and VRE from 25%. Some patients (383) had more than one pathogenic culture.
Compared with noninfected patients, infected patients were significantly older (mean 52 vs. 51 years), had higher Charlson comorbidity indices, and more likely to be male. They were also more likely to be admitted to the intensive care unit (ICU) than were those without infection (46% vs. 22%), and had a significantly longer hospital stay (24 vs. 6 days).
Within the next year, 35% of the study group was readmitted to the hospital. Rehospitalization was significantly more common among patients with the past infection than among those without (hazard ratio, 1.40). Those with VRE had the highest risk of readmission (HR, 1.67), followed by those with MRSA (HR, 1.30) and C. difficile (HR, 1.35).
There were also significant differences within the readmission group. Patients under age 65 were significantly more likely to be readmitted than were older patients (HR, 1.45). Those who had an ICU stay during the index hospitalization were significantly less likely to be readmitted (HR, 0.87). Each 1-point increase on the Charlson comorbidity index was associated with a significant 11% increase in the risk of readmission (HR, 1.11).
"Furthermore, the median time to readmission for [previously infected] patients was almost half that for other patients," they noted.
The authors reported having no relevant disclosures. The study was sponsored by the Agency for Healthcare Research and Quality and the National Institutes of Health.

|
|
I don’t think that hospital-acquired infections actually cause readmissions after discharge, although that may represent a small percentage of these cases. Instead, these infections are more likely to be a marker for a patient with factors that put them at risk for infection – older patients and those with medical comorbidities, for example, who are more susceptible to decompensation. Patients with a chronic disease are also vulnerable. They live on a teeter-totter, and small imbalances can tip the whole thing over. It doesn’t take much for these patients to land back in the hospital.
Getting a hospital-acquired infection increases recovery time and the need for rehabilitation, and all of this increases the likelihood of readmission.
Dr. Frank Michota is the director of academic affairs in the department of hospital medicine at the Cleveland Clinic. He reports having no relevant conflicts of interest.

|
|
I don’t think that hospital-acquired infections actually cause readmissions after discharge, although that may represent a small percentage of these cases. Instead, these infections are more likely to be a marker for a patient with factors that put them at risk for infection – older patients and those with medical comorbidities, for example, who are more susceptible to decompensation. Patients with a chronic disease are also vulnerable. They live on a teeter-totter, and small imbalances can tip the whole thing over. It doesn’t take much for these patients to land back in the hospital.
Getting a hospital-acquired infection increases recovery time and the need for rehabilitation, and all of this increases the likelihood of readmission.
Dr. Frank Michota is the director of academic affairs in the department of hospital medicine at the Cleveland Clinic. He reports having no relevant conflicts of interest.

|
|
I don’t think that hospital-acquired infections actually cause readmissions after discharge, although that may represent a small percentage of these cases. Instead, these infections are more likely to be a marker for a patient with factors that put them at risk for infection – older patients and those with medical comorbidities, for example, who are more susceptible to decompensation. Patients with a chronic disease are also vulnerable. They live on a teeter-totter, and small imbalances can tip the whole thing over. It doesn’t take much for these patients to land back in the hospital.
Getting a hospital-acquired infection increases recovery time and the need for rehabilitation, and all of this increases the likelihood of readmission.
Dr. Frank Michota is the director of academic affairs in the department of hospital medicine at the Cleveland Clinic. He reports having no relevant conflicts of interest.
Hospitalized patients who pick up a nosocomial infection are 40% more likely to be readmitted within a year of discharge than patients who are not infected during their stay.
Ramped-up infection control could benefit not only patients but the hospital itself, Jon Furuno, Ph.D., and his colleagues wrote in the June issue of Infection Control and Hospital Epidemiology.
"Efforts to reduce healthcare-associated infections may have the additional benefit of reducing the incidence of hospital readmission and increased healthcare costs," wrote Dr. Furuno, an infectious disease specialist at the University of Maryland, Baltimore, and Oregon Health and Science University, Portland, and his coauthors. "Furthermore, patients with a positive clinical culture result obtained more than 48 hours after hospital admission could be targeted to receive additional discharge planning resources designed to reduce the likelihood of hospital readmission" (Infect. Contr. Hosp. Epidem. 2012;33:539-44 [doi: 10.1086/665725]).
Dr. Furuno and his associates examined inpatient infections from Jan. 1, 2001, to Dec. 31, 2008, at the University of Maryland Medical Center. The team focused on methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococci (VRE), and Clostridium difficile infections. The primary end point was time to readmission.
The study group included 136,513 admissions over the 8-year period. The patients’ mean age was 51 years, and the mean length of stay was 6 days (range, 3-13 days).
An infection was deemed to be hospital-acquired if a positive culture was obtained more than 48 hours after admission.
Only 3% of the study group had a positive culture that was hospital acquired. MRSA was cultured from 49% of the patients, C. difficile from 35%, and VRE from 25%. Some patients (383) had more than one pathogenic culture.
Compared with noninfected patients, infected patients were significantly older (mean 52 vs. 51 years), had higher Charlson comorbidity indices, and more likely to be male. They were also more likely to be admitted to the intensive care unit (ICU) than were those without infection (46% vs. 22%), and had a significantly longer hospital stay (24 vs. 6 days).
Within the next year, 35% of the study group was readmitted to the hospital. Rehospitalization was significantly more common among patients with the past infection than among those without (hazard ratio, 1.40). Those with VRE had the highest risk of readmission (HR, 1.67), followed by those with MRSA (HR, 1.30) and C. difficile (HR, 1.35).
There were also significant differences within the readmission group. Patients under age 65 were significantly more likely to be readmitted than were older patients (HR, 1.45). Those who had an ICU stay during the index hospitalization were significantly less likely to be readmitted (HR, 0.87). Each 1-point increase on the Charlson comorbidity index was associated with a significant 11% increase in the risk of readmission (HR, 1.11).
"Furthermore, the median time to readmission for [previously infected] patients was almost half that for other patients," they noted.
The authors reported having no relevant disclosures. The study was sponsored by the Agency for Healthcare Research and Quality and the National Institutes of Health.
Hospitalized patients who pick up a nosocomial infection are 40% more likely to be readmitted within a year of discharge than patients who are not infected during their stay.
Ramped-up infection control could benefit not only patients but the hospital itself, Jon Furuno, Ph.D., and his colleagues wrote in the June issue of Infection Control and Hospital Epidemiology.
"Efforts to reduce healthcare-associated infections may have the additional benefit of reducing the incidence of hospital readmission and increased healthcare costs," wrote Dr. Furuno, an infectious disease specialist at the University of Maryland, Baltimore, and Oregon Health and Science University, Portland, and his coauthors. "Furthermore, patients with a positive clinical culture result obtained more than 48 hours after hospital admission could be targeted to receive additional discharge planning resources designed to reduce the likelihood of hospital readmission" (Infect. Contr. Hosp. Epidem. 2012;33:539-44 [doi: 10.1086/665725]).
Dr. Furuno and his associates examined inpatient infections from Jan. 1, 2001, to Dec. 31, 2008, at the University of Maryland Medical Center. The team focused on methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococci (VRE), and Clostridium difficile infections. The primary end point was time to readmission.
The study group included 136,513 admissions over the 8-year period. The patients’ mean age was 51 years, and the mean length of stay was 6 days (range, 3-13 days).
An infection was deemed to be hospital-acquired if a positive culture was obtained more than 48 hours after admission.
Only 3% of the study group had a positive culture that was hospital acquired. MRSA was cultured from 49% of the patients, C. difficile from 35%, and VRE from 25%. Some patients (383) had more than one pathogenic culture.
Compared with noninfected patients, infected patients were significantly older (mean 52 vs. 51 years), had higher Charlson comorbidity indices, and more likely to be male. They were also more likely to be admitted to the intensive care unit (ICU) than were those without infection (46% vs. 22%), and had a significantly longer hospital stay (24 vs. 6 days).
Within the next year, 35% of the study group was readmitted to the hospital. Rehospitalization was significantly more common among patients with the past infection than among those without (hazard ratio, 1.40). Those with VRE had the highest risk of readmission (HR, 1.67), followed by those with MRSA (HR, 1.30) and C. difficile (HR, 1.35).
There were also significant differences within the readmission group. Patients under age 65 were significantly more likely to be readmitted than were older patients (HR, 1.45). Those who had an ICU stay during the index hospitalization were significantly less likely to be readmitted (HR, 0.87). Each 1-point increase on the Charlson comorbidity index was associated with a significant 11% increase in the risk of readmission (HR, 1.11).
"Furthermore, the median time to readmission for [previously infected] patients was almost half that for other patients," they noted.
The authors reported having no relevant disclosures. The study was sponsored by the Agency for Healthcare Research and Quality and the National Institutes of Health.
FROM INFECTION CONTROL AND HOSPITAL EPIDEMIOLOGY
Fever Work-Up for Infants May Be Safely Shortened
BOSTON – Many infants with a fever of unknown origin can be safely discharged from the hospital sooner than the routinely recommended 48 hours, a study has shown.
A review of 11 years’ worth of data found that just 0.5% of these infants’ blood and cerebrospinal fluid cultures took more than 24 hours to come back positive. These patients all had concerning signs on their clinical exam or initial labs, suggesting that infants with a low-risk profile can probably be safely discharged by 24 hours.
"Institutions that are hospitalizing all of these infants for 48 hours can think about changing that to 36 or even 24 hours for infants who have normal labs and look clinically well," Vikram Fielding-Singh said at the annual meeting of the Pediatric Academic Societies.
Mr. Fielding-Singh, a medical student at Stanford (Calif.) University, reviewed the university’s inpatient records from 1999 to 2010. All patients in his analysis were 30 days old or younger, and underwent a diagnostic work-up for fever of unknown origin; tests included blood and CSF cultures.
The study comprised 1,880 cultures taken from 1,145 infants. Of these patients, 483 were hospitalized for fever without a known source.
Of the total group, 31 infants (3%) had positive blood or CSF cultures. Only six (0.5%) of these took more than 24 hours to return a positive result. In the subset of infants hospitalized for a fever work-up, 24 (5%) had positive cultures. Again, just six (1%) returned a positive result after 24 hours.
All of these infants had warning signs of a serious illness, either on clinical exam or in other lab work, Mr. Fielding-Singh said. No infant with normal labs at presentation had a positive culture that took more than 24 hours to return a positive result.
Five infants had bacteremia – four with a concurrent urinary tract infection – and one had bacterial meningitis. All of these patients had concerning lab findings – a positive urine culture result, elevated white blood cell levels, or an increased absolute band count. Five also had other abnormal findings, including pleocytosis, a chest radiograph showing infiltrate, or an elevated C-reactive protein level.
Mr. Fielding-Singh also performed a sequential review of one of every four hospitalizations (121) for the fever of unknown origin work-up. More than half (56%) were considered low risk according to their labs and clinical evaluation, he said.
Dr. Alan R. Schroeder, who supervised the project, said it showed that physicians can "safely do less" with many patients.
"It’s our moral obligation to look at practices that put children at risk and that aren’t necessarily evidence based," said Dr. Schroeder, a pediatric critical care specialist at the Santa Clara Valley Medical Center, San Jose, Calif. "This is a practice that is almost rote for many institutions. These babies get all kinds of tests and are hospitalized for 48 hours almost without much thought."
A routine 48-hour admission for every infant with a presumed fever without source "puts a pretty big burden" on hospital resources, Dr. Schroeder said in an interview, not to mention putting small patients at great risk for nosocomial complications.
"If you could lessen the hospital stay [for] more than half of these kids, that should have a positive impact on resource utilization and certainly on the iatrogenic harms that can occur to these little babies," he said.
Neither Mr. Fielding-Singh nor Dr. Schroeder reported having any relevant financial disclosures.
BOSTON – Many infants with a fever of unknown origin can be safely discharged from the hospital sooner than the routinely recommended 48 hours, a study has shown.
A review of 11 years’ worth of data found that just 0.5% of these infants’ blood and cerebrospinal fluid cultures took more than 24 hours to come back positive. These patients all had concerning signs on their clinical exam or initial labs, suggesting that infants with a low-risk profile can probably be safely discharged by 24 hours.
"Institutions that are hospitalizing all of these infants for 48 hours can think about changing that to 36 or even 24 hours for infants who have normal labs and look clinically well," Vikram Fielding-Singh said at the annual meeting of the Pediatric Academic Societies.
Mr. Fielding-Singh, a medical student at Stanford (Calif.) University, reviewed the university’s inpatient records from 1999 to 2010. All patients in his analysis were 30 days old or younger, and underwent a diagnostic work-up for fever of unknown origin; tests included blood and CSF cultures.
The study comprised 1,880 cultures taken from 1,145 infants. Of these patients, 483 were hospitalized for fever without a known source.
Of the total group, 31 infants (3%) had positive blood or CSF cultures. Only six (0.5%) of these took more than 24 hours to return a positive result. In the subset of infants hospitalized for a fever work-up, 24 (5%) had positive cultures. Again, just six (1%) returned a positive result after 24 hours.
All of these infants had warning signs of a serious illness, either on clinical exam or in other lab work, Mr. Fielding-Singh said. No infant with normal labs at presentation had a positive culture that took more than 24 hours to return a positive result.
Five infants had bacteremia – four with a concurrent urinary tract infection – and one had bacterial meningitis. All of these patients had concerning lab findings – a positive urine culture result, elevated white blood cell levels, or an increased absolute band count. Five also had other abnormal findings, including pleocytosis, a chest radiograph showing infiltrate, or an elevated C-reactive protein level.
Mr. Fielding-Singh also performed a sequential review of one of every four hospitalizations (121) for the fever of unknown origin work-up. More than half (56%) were considered low risk according to their labs and clinical evaluation, he said.
Dr. Alan R. Schroeder, who supervised the project, said it showed that physicians can "safely do less" with many patients.
"It’s our moral obligation to look at practices that put children at risk and that aren’t necessarily evidence based," said Dr. Schroeder, a pediatric critical care specialist at the Santa Clara Valley Medical Center, San Jose, Calif. "This is a practice that is almost rote for many institutions. These babies get all kinds of tests and are hospitalized for 48 hours almost without much thought."
A routine 48-hour admission for every infant with a presumed fever without source "puts a pretty big burden" on hospital resources, Dr. Schroeder said in an interview, not to mention putting small patients at great risk for nosocomial complications.
"If you could lessen the hospital stay [for] more than half of these kids, that should have a positive impact on resource utilization and certainly on the iatrogenic harms that can occur to these little babies," he said.
Neither Mr. Fielding-Singh nor Dr. Schroeder reported having any relevant financial disclosures.
BOSTON – Many infants with a fever of unknown origin can be safely discharged from the hospital sooner than the routinely recommended 48 hours, a study has shown.
A review of 11 years’ worth of data found that just 0.5% of these infants’ blood and cerebrospinal fluid cultures took more than 24 hours to come back positive. These patients all had concerning signs on their clinical exam or initial labs, suggesting that infants with a low-risk profile can probably be safely discharged by 24 hours.
"Institutions that are hospitalizing all of these infants for 48 hours can think about changing that to 36 or even 24 hours for infants who have normal labs and look clinically well," Vikram Fielding-Singh said at the annual meeting of the Pediatric Academic Societies.
Mr. Fielding-Singh, a medical student at Stanford (Calif.) University, reviewed the university’s inpatient records from 1999 to 2010. All patients in his analysis were 30 days old or younger, and underwent a diagnostic work-up for fever of unknown origin; tests included blood and CSF cultures.
The study comprised 1,880 cultures taken from 1,145 infants. Of these patients, 483 were hospitalized for fever without a known source.
Of the total group, 31 infants (3%) had positive blood or CSF cultures. Only six (0.5%) of these took more than 24 hours to return a positive result. In the subset of infants hospitalized for a fever work-up, 24 (5%) had positive cultures. Again, just six (1%) returned a positive result after 24 hours.
All of these infants had warning signs of a serious illness, either on clinical exam or in other lab work, Mr. Fielding-Singh said. No infant with normal labs at presentation had a positive culture that took more than 24 hours to return a positive result.
Five infants had bacteremia – four with a concurrent urinary tract infection – and one had bacterial meningitis. All of these patients had concerning lab findings – a positive urine culture result, elevated white blood cell levels, or an increased absolute band count. Five also had other abnormal findings, including pleocytosis, a chest radiograph showing infiltrate, or an elevated C-reactive protein level.
Mr. Fielding-Singh also performed a sequential review of one of every four hospitalizations (121) for the fever of unknown origin work-up. More than half (56%) were considered low risk according to their labs and clinical evaluation, he said.
Dr. Alan R. Schroeder, who supervised the project, said it showed that physicians can "safely do less" with many patients.
"It’s our moral obligation to look at practices that put children at risk and that aren’t necessarily evidence based," said Dr. Schroeder, a pediatric critical care specialist at the Santa Clara Valley Medical Center, San Jose, Calif. "This is a practice that is almost rote for many institutions. These babies get all kinds of tests and are hospitalized for 48 hours almost without much thought."
A routine 48-hour admission for every infant with a presumed fever without source "puts a pretty big burden" on hospital resources, Dr. Schroeder said in an interview, not to mention putting small patients at great risk for nosocomial complications.
"If you could lessen the hospital stay [for] more than half of these kids, that should have a positive impact on resource utilization and certainly on the iatrogenic harms that can occur to these little babies," he said.
Neither Mr. Fielding-Singh nor Dr. Schroeder reported having any relevant financial disclosures.
FROM THE ANNUAL MEETING OF THE PEDIATRIC ACADEMIC SOCIETIES
Major Finding: In a group of infants hospitalized for a fever work-up, fewer than 1% of cultures took more than 24 hours to return a positive result.
Data Source: A review of 1,880 blood and CSF cultures from 1,145 infants was conducted.
Disclosures: Neither Mr. Fielding-Singh nor Dr. Schroeder reported having any relevant financial disclosures.
Neonatal Candidiasis Decreases With Prophylactic Antifungals
BOSTON – Preventive antifungal therapy and changes in broad-spectrum antibiotic use were associated with a significant decrease in the incidence of invasive candidiasis in neonatal intensive care units.
As these changes took place in a large group of neonatal intensive care units, Candida infections dropped from 4 to 1 per 1,000 patients. In the smallest infants – those weighing less than 750 grams – incidence dropped from 83 to 24 per 1,000, Dr. Sofia Aliaga said at the annual meeting of the Pediatric Academic Societies.
Dr. Aliaga, a neonatologist at the University of North Carolina at Chapel Hill, and her colleagues used a large administrative database to determine the incidence of invasive candidiasis over a 14-year period (1997-2010). In addition to disease incidence, they looked at changes in both antifungal prophylaxis and empirical treatment, and the use of broad-spectrum antibiotics in the NICUs.
The database contained information on 709,325 infants seen at 322 units, all of which were managed by a single medical group. There were 2,101 episodes of invasive candidiasis in 2,063 infants.
The researchers divided the cohort into four groups by weight: less than 750 g, 750-999 g, 1,000-1,499 g, and 1,500 g or heavier.
Over the study period, antifungal prophylaxis increased significantly among the smallest babies, from 4 to 119 admissions per 1,000. At the same time, empirical antifungal therapy for these infants also increased, from 4 to 11 admissions per 1,000 overall. The biggest change with empirical therapy occurred in the smallest babies, increasing from 4 to 111 per 1,000.
Finally, the units decreased their use of broad-spectrum antibiotics for all admissions. Use fell from 276 to 48 admissions per 1,000. This change was seen across all weight groups.
The incidence of invasive candidiasis fell from 4 to 1 per 1,000 admissions. The greatest decrease occurred among babies weighing less than 750 g. In this group, the infection fell from 83 to 24 per 1,000.
Overall changes in NICU management also might have influenced the incidence of candidiasis, Dr. Aliaga acknowledged.
"During this time, there was a big push to do quality improvement projects with central line catheters to decrease infection rates ... Babies in the NICU also spend a lot less time on ventilators now than they did 10 years ago, so with less time with an endotracheal tube there are fewer ventilator-associated infections. But I do think the decrease in disease we’re seeing is real, and related to the changes we have made."
Dr. Aliaga said she had no relevant financial disclosures.
BOSTON – Preventive antifungal therapy and changes in broad-spectrum antibiotic use were associated with a significant decrease in the incidence of invasive candidiasis in neonatal intensive care units.
As these changes took place in a large group of neonatal intensive care units, Candida infections dropped from 4 to 1 per 1,000 patients. In the smallest infants – those weighing less than 750 grams – incidence dropped from 83 to 24 per 1,000, Dr. Sofia Aliaga said at the annual meeting of the Pediatric Academic Societies.
Dr. Aliaga, a neonatologist at the University of North Carolina at Chapel Hill, and her colleagues used a large administrative database to determine the incidence of invasive candidiasis over a 14-year period (1997-2010). In addition to disease incidence, they looked at changes in both antifungal prophylaxis and empirical treatment, and the use of broad-spectrum antibiotics in the NICUs.
The database contained information on 709,325 infants seen at 322 units, all of which were managed by a single medical group. There were 2,101 episodes of invasive candidiasis in 2,063 infants.
The researchers divided the cohort into four groups by weight: less than 750 g, 750-999 g, 1,000-1,499 g, and 1,500 g or heavier.
Over the study period, antifungal prophylaxis increased significantly among the smallest babies, from 4 to 119 admissions per 1,000. At the same time, empirical antifungal therapy for these infants also increased, from 4 to 11 admissions per 1,000 overall. The biggest change with empirical therapy occurred in the smallest babies, increasing from 4 to 111 per 1,000.
Finally, the units decreased their use of broad-spectrum antibiotics for all admissions. Use fell from 276 to 48 admissions per 1,000. This change was seen across all weight groups.
The incidence of invasive candidiasis fell from 4 to 1 per 1,000 admissions. The greatest decrease occurred among babies weighing less than 750 g. In this group, the infection fell from 83 to 24 per 1,000.
Overall changes in NICU management also might have influenced the incidence of candidiasis, Dr. Aliaga acknowledged.
"During this time, there was a big push to do quality improvement projects with central line catheters to decrease infection rates ... Babies in the NICU also spend a lot less time on ventilators now than they did 10 years ago, so with less time with an endotracheal tube there are fewer ventilator-associated infections. But I do think the decrease in disease we’re seeing is real, and related to the changes we have made."
Dr. Aliaga said she had no relevant financial disclosures.
BOSTON – Preventive antifungal therapy and changes in broad-spectrum antibiotic use were associated with a significant decrease in the incidence of invasive candidiasis in neonatal intensive care units.
As these changes took place in a large group of neonatal intensive care units, Candida infections dropped from 4 to 1 per 1,000 patients. In the smallest infants – those weighing less than 750 grams – incidence dropped from 83 to 24 per 1,000, Dr. Sofia Aliaga said at the annual meeting of the Pediatric Academic Societies.
Dr. Aliaga, a neonatologist at the University of North Carolina at Chapel Hill, and her colleagues used a large administrative database to determine the incidence of invasive candidiasis over a 14-year period (1997-2010). In addition to disease incidence, they looked at changes in both antifungal prophylaxis and empirical treatment, and the use of broad-spectrum antibiotics in the NICUs.
The database contained information on 709,325 infants seen at 322 units, all of which were managed by a single medical group. There were 2,101 episodes of invasive candidiasis in 2,063 infants.
The researchers divided the cohort into four groups by weight: less than 750 g, 750-999 g, 1,000-1,499 g, and 1,500 g or heavier.
Over the study period, antifungal prophylaxis increased significantly among the smallest babies, from 4 to 119 admissions per 1,000. At the same time, empirical antifungal therapy for these infants also increased, from 4 to 11 admissions per 1,000 overall. The biggest change with empirical therapy occurred in the smallest babies, increasing from 4 to 111 per 1,000.
Finally, the units decreased their use of broad-spectrum antibiotics for all admissions. Use fell from 276 to 48 admissions per 1,000. This change was seen across all weight groups.
The incidence of invasive candidiasis fell from 4 to 1 per 1,000 admissions. The greatest decrease occurred among babies weighing less than 750 g. In this group, the infection fell from 83 to 24 per 1,000.
Overall changes in NICU management also might have influenced the incidence of candidiasis, Dr. Aliaga acknowledged.
"During this time, there was a big push to do quality improvement projects with central line catheters to decrease infection rates ... Babies in the NICU also spend a lot less time on ventilators now than they did 10 years ago, so with less time with an endotracheal tube there are fewer ventilator-associated infections. But I do think the decrease in disease we’re seeing is real, and related to the changes we have made."
Dr. Aliaga said she had no relevant financial disclosures.
FROM THE ANNUAL MEETING OF THE PEDIATRIC ACADEMIC SOCIETIES
Major Finding: The incidence of invasive candidiasis dropped by 75% after neonatal units instituted a program of prophylactic antifungal treatment.
Data Source: Dr. Aliaga extracted her data from a large administrative database.
Disclosures: Dr. Aliaga said she had no relevant financial disclosures.
National Plan Aims to Eliminate Alzheimer's By 2025
BETHESDA, MD. – A new plan marshals the forces of funding, research, and education to fight the battle against Alzheimer’s.
The National Plan to Address Alzheimer’s Disease has an ambitious, overarching goal: to prevent and effectively treat Alzheimer’s by 2025. But while focusing on the future, it does not neglect the present. The plan calls for the resources necessary to ease burdens the disease places on families and caregivers, to improve the quality of care for patients, and to engage the public in a national debate over a condition that threatens to overwhelm national health care systems by 2040.
A unified strategy is the only way to stop a tidal wave that threatens not only America, but the entire world, said Health and Human Services Secretary Kathleen Sebelius.
Americans are living longer and longer, Ms. Sebelius said when she unveiled the plan at a research summit sponsored by the National Institute on Aging. "But with that come additional challenges, and one of the biggest is Alzheimer’s. We know that as many as 5 million Americans carry this diagnosis now. And as those of us in the baby boomer generation age, the numbers of people with Alzheimer’s will more than double in the next few decades. What we know is that a lot more needs to be done, and done right now."
The 2011 National Alzheimer’s Project Act called for the creation of such a plan. Drawing on the experiences and opinions of researchers, policy makers, advocacy groups, and families, it began to take shape that year. In February, with a draft version, the Obama administration allocated $156 million to support its goals. This move allowed the National Institutes of Health to earmark an additional $50 million for Alzheimer’s research this fiscal year.
"Our goal is that everyone can get the care and help they need."
The proposed 2013 federal budget includes $100 million for efforts to combat Alzheimer’s. In addition to $80 million earmarked for research, the proposed budget allocates monies for public awareness campaigns ($4.2 million), provider education programs ($4 million), caregiver support ($10.5 million), and improved data collection ($1.3 million).
This year’s research allocation will fund two groundbreaking studies: the first-ever primary prevention trial in patients at high risk for the disease and a study to determine the effect of inhaled insulin on cognition, function, and biomarkers in people with mild cognitive impairment or early Alzheimer’s.
"Research has provided valuable insights into the disease in the last few years, but no breakthroughs leading to treatment," Ms. Sebelius said. "Clinical trials have been disappointing. We have no way of preventing Alzheimer’s – our ultimate goal. We have a general lack of understanding of Alzheimer’s as well. Many caregivers have to ‘go it’ alone, and health care professionals with limited time may not be able to address dementia in the most effective ways."
But the Alzheimer’s plan is not only about research, Ms. Sebelius said. It brings unprecedented resources to those on the front lines of the fight: patients, families, caregivers, and health care workers.
"Our goal is that everyone can get the care and help they need. This is a true national plan based on a strong partnership with the Alzheimer’s community, and clearly we will not be able to carry it off without them."
A new government-sponsored information clearinghouse will offer immediate answers to at least some of the problems those dealing with the disease face on a daily basis.
The website, www.alzheimers.gov, is devoted to all aspects of care that patients with Alzheimer’s disease require. It provides information on current treatment options, living situations, research studies, and the financial challenges of the disease, as well as direct links to the national plan and the National Alzheimer’s Project Act.
The time is right for such a bold and aggressive agenda, said researcher Suzanne Craft, Ph.D., director of the memory disorders clinic at the American Lakes division of the VA Puget Sound Health Care System, Tacoma, Wash., and lead investigator on the inhaled insulin trial.
"It’s time to draw national attention to the need for additional research funding and additional programs to support caregivers, and to educate the primary care physicians who take care of these patients. Alzheimer’s places the greatest financial burden of any disease on our health care system, and one of the greatest emotional burdens on families and caregivers. This overarching approach has been lacking, and this is a very important step in the right direction."
Dr. Craft had no financial disclosures.
BETHESDA, MD. – A new plan marshals the forces of funding, research, and education to fight the battle against Alzheimer’s.
The National Plan to Address Alzheimer’s Disease has an ambitious, overarching goal: to prevent and effectively treat Alzheimer’s by 2025. But while focusing on the future, it does not neglect the present. The plan calls for the resources necessary to ease burdens the disease places on families and caregivers, to improve the quality of care for patients, and to engage the public in a national debate over a condition that threatens to overwhelm national health care systems by 2040.
A unified strategy is the only way to stop a tidal wave that threatens not only America, but the entire world, said Health and Human Services Secretary Kathleen Sebelius.
Americans are living longer and longer, Ms. Sebelius said when she unveiled the plan at a research summit sponsored by the National Institute on Aging. "But with that come additional challenges, and one of the biggest is Alzheimer’s. We know that as many as 5 million Americans carry this diagnosis now. And as those of us in the baby boomer generation age, the numbers of people with Alzheimer’s will more than double in the next few decades. What we know is that a lot more needs to be done, and done right now."
The 2011 National Alzheimer’s Project Act called for the creation of such a plan. Drawing on the experiences and opinions of researchers, policy makers, advocacy groups, and families, it began to take shape that year. In February, with a draft version, the Obama administration allocated $156 million to support its goals. This move allowed the National Institutes of Health to earmark an additional $50 million for Alzheimer’s research this fiscal year.
"Our goal is that everyone can get the care and help they need."
The proposed 2013 federal budget includes $100 million for efforts to combat Alzheimer’s. In addition to $80 million earmarked for research, the proposed budget allocates monies for public awareness campaigns ($4.2 million), provider education programs ($4 million), caregiver support ($10.5 million), and improved data collection ($1.3 million).
This year’s research allocation will fund two groundbreaking studies: the first-ever primary prevention trial in patients at high risk for the disease and a study to determine the effect of inhaled insulin on cognition, function, and biomarkers in people with mild cognitive impairment or early Alzheimer’s.
"Research has provided valuable insights into the disease in the last few years, but no breakthroughs leading to treatment," Ms. Sebelius said. "Clinical trials have been disappointing. We have no way of preventing Alzheimer’s – our ultimate goal. We have a general lack of understanding of Alzheimer’s as well. Many caregivers have to ‘go it’ alone, and health care professionals with limited time may not be able to address dementia in the most effective ways."
But the Alzheimer’s plan is not only about research, Ms. Sebelius said. It brings unprecedented resources to those on the front lines of the fight: patients, families, caregivers, and health care workers.
"Our goal is that everyone can get the care and help they need. This is a true national plan based on a strong partnership with the Alzheimer’s community, and clearly we will not be able to carry it off without them."
A new government-sponsored information clearinghouse will offer immediate answers to at least some of the problems those dealing with the disease face on a daily basis.
The website, www.alzheimers.gov, is devoted to all aspects of care that patients with Alzheimer’s disease require. It provides information on current treatment options, living situations, research studies, and the financial challenges of the disease, as well as direct links to the national plan and the National Alzheimer’s Project Act.
The time is right for such a bold and aggressive agenda, said researcher Suzanne Craft, Ph.D., director of the memory disorders clinic at the American Lakes division of the VA Puget Sound Health Care System, Tacoma, Wash., and lead investigator on the inhaled insulin trial.
"It’s time to draw national attention to the need for additional research funding and additional programs to support caregivers, and to educate the primary care physicians who take care of these patients. Alzheimer’s places the greatest financial burden of any disease on our health care system, and one of the greatest emotional burdens on families and caregivers. This overarching approach has been lacking, and this is a very important step in the right direction."
Dr. Craft had no financial disclosures.
BETHESDA, MD. – A new plan marshals the forces of funding, research, and education to fight the battle against Alzheimer’s.
The National Plan to Address Alzheimer’s Disease has an ambitious, overarching goal: to prevent and effectively treat Alzheimer’s by 2025. But while focusing on the future, it does not neglect the present. The plan calls for the resources necessary to ease burdens the disease places on families and caregivers, to improve the quality of care for patients, and to engage the public in a national debate over a condition that threatens to overwhelm national health care systems by 2040.
A unified strategy is the only way to stop a tidal wave that threatens not only America, but the entire world, said Health and Human Services Secretary Kathleen Sebelius.
Americans are living longer and longer, Ms. Sebelius said when she unveiled the plan at a research summit sponsored by the National Institute on Aging. "But with that come additional challenges, and one of the biggest is Alzheimer’s. We know that as many as 5 million Americans carry this diagnosis now. And as those of us in the baby boomer generation age, the numbers of people with Alzheimer’s will more than double in the next few decades. What we know is that a lot more needs to be done, and done right now."
The 2011 National Alzheimer’s Project Act called for the creation of such a plan. Drawing on the experiences and opinions of researchers, policy makers, advocacy groups, and families, it began to take shape that year. In February, with a draft version, the Obama administration allocated $156 million to support its goals. This move allowed the National Institutes of Health to earmark an additional $50 million for Alzheimer’s research this fiscal year.
"Our goal is that everyone can get the care and help they need."
The proposed 2013 federal budget includes $100 million for efforts to combat Alzheimer’s. In addition to $80 million earmarked for research, the proposed budget allocates monies for public awareness campaigns ($4.2 million), provider education programs ($4 million), caregiver support ($10.5 million), and improved data collection ($1.3 million).
This year’s research allocation will fund two groundbreaking studies: the first-ever primary prevention trial in patients at high risk for the disease and a study to determine the effect of inhaled insulin on cognition, function, and biomarkers in people with mild cognitive impairment or early Alzheimer’s.
"Research has provided valuable insights into the disease in the last few years, but no breakthroughs leading to treatment," Ms. Sebelius said. "Clinical trials have been disappointing. We have no way of preventing Alzheimer’s – our ultimate goal. We have a general lack of understanding of Alzheimer’s as well. Many caregivers have to ‘go it’ alone, and health care professionals with limited time may not be able to address dementia in the most effective ways."
But the Alzheimer’s plan is not only about research, Ms. Sebelius said. It brings unprecedented resources to those on the front lines of the fight: patients, families, caregivers, and health care workers.
"Our goal is that everyone can get the care and help they need. This is a true national plan based on a strong partnership with the Alzheimer’s community, and clearly we will not be able to carry it off without them."
A new government-sponsored information clearinghouse will offer immediate answers to at least some of the problems those dealing with the disease face on a daily basis.
The website, www.alzheimers.gov, is devoted to all aspects of care that patients with Alzheimer’s disease require. It provides information on current treatment options, living situations, research studies, and the financial challenges of the disease, as well as direct links to the national plan and the National Alzheimer’s Project Act.
The time is right for such a bold and aggressive agenda, said researcher Suzanne Craft, Ph.D., director of the memory disorders clinic at the American Lakes division of the VA Puget Sound Health Care System, Tacoma, Wash., and lead investigator on the inhaled insulin trial.
"It’s time to draw national attention to the need for additional research funding and additional programs to support caregivers, and to educate the primary care physicians who take care of these patients. Alzheimer’s places the greatest financial burden of any disease on our health care system, and one of the greatest emotional burdens on families and caregivers. This overarching approach has been lacking, and this is a very important step in the right direction."
Dr. Craft had no financial disclosures.
FROM A RESEARCH SUMMIT SPONSORED BY THE NATIONAL INSTITUTE ON AGING
Federal Funds Launch First Alzheimer's Prevention Study
BETHESDA, MD. – The first-ever primary prevention trial for Alzheimer’s disease will investigate whether immunotherapy can delay – or even stop – the disease in a group of subjects who are genetically destined to develop it.
The $100 million project is one of the first to receive a special federal allocation for Alzheimer’s research, as envisioned in President Obama’s 2011 National Alzheimer’s Project Act.
Federal dollars will also support a second major clinical trial – a placebo-controlled study designed to evaluate the effect of intranasal insulin on cognition, function, and biomarkers in individuals with mild cognitive impairment and early Alzheimer’s.
These two projects will consume about half of the additional $50 million in Alzheimer’s project funding allocated by Congress for this fiscal year. Next year’s additional federal boost of $80 million will support more new studies, according to Dr. Ronald Petersen, chairman of the National Alzheimer’s Project Act’s Advisory Council on Alzheimer’s Research, Care, and Services.
"More studies are coming," he said at a research summit sponsored by the National Institute on Aging. These will include experiments with pluripotent stem cells, which have recently been differentiated into functional human Alzheimer’s neurons, and a large longitudinal study of the link between Alzheimer’s and cardiovascular disease.
Dr. Eric Reiman is one of three primary investigators on the 5-year immunotherapy trial. The placebo-controlled trial will give the humanized monoclonal antibody crenezumab to a group of about 300 members of an extended family in Colombia before they show symptoms of the disease. Many in this family express a mutation in the presenilin-1 gene that causes very early onset Alzheimer’s, with symptoms beginning before the age of 45 years. The trial will also include a smaller number of family members in the United States.
Half of the individuals will receive infusions of the drug, and half will receive a placebo. An additional group of individuals without the mutation will also receive a placebo, he added.
The participants will be followed for up to 5 years, with periodic cognitive, biomarker, and imaging assessments, said Dr. Reiman, executive director of the Banner Alzheimer’s Institute, Phoenix. After 2 years, a data safety monitoring committee will evaluate any progress and determine whether the trial should proceed.
Immunotherapy has walked a rocky path in Alzheimer’s drug development. The first active immunization clinical trial, which tested AN1792* in AD patients, was halted when a subset of patients developed meningoencephalitis. The first passive immunotherapy trial with bapineuzumab, showed vasogenic edema and microhemorrhages in 12 patients, but this drug continues to be studied.
The crenezumab study is unique in one other way, Dr. Reiman said in an interview: All of the data generated will become publicly available as soon as the trial is complete.
"We’ll be sharing all the data and samples in the hope of faster development of preventive treatments. We’re all in this fight together and given the stakes, it’s time for a paradigm shift that can – hopefully – launch a new era in Alzheimer’s prevention research."
In addition to its $16 million federal grant, the study will draw on $15 million in philanthropic funds from the Banner Institute, Dr. Reiman added.
Suzanne Craft, Ph.D., is heading the insulin study. The 5-year study will randomize 240 subjects with mild cognitive impairment or early Alzheimer’s to intranasal insulin or placebo for 12 months. At the end of that time, placebo patients will receive insulin in a 12-month open-label study. Subjects will be assessed periodically for changes in cognition, function, and cerebrospinal biomarkers.
An $8 million federal grant makes the study possible, said Dr. Craft, professor of psychiatry and behavioral sciences at the University of Washington, Seattle, and director of the Memory Disorders Clinic at the American Lakes division of the VA Puget Sound Health Care System.
Her earlier work with insulin showed tantalizing promise. A 104-subject, placebo-controlled study found that doses of both 20 and 40 IU of intranasal insulin preserved cognition and function in patients with mild cognitive impairment and early Alzheimer’s (Arch. Neurol. 2012;69:29-38). There was also some hint that these changes were associated with improved beta-amyloid biomarkers in cerebrospinal fluid. Patients assigned to placebo also showed declines in neuronal metabolism compared with those taking insulin.
Data from the new insulin study will also go into the public domain, Dr. Craft said in an interview.
"I think that as we make these data available to people with good ideas, we’ll see a proliferation of results from them," she said. "It’s essential to bring together investigators from all different disciplines to look at the question from different angles. It’s a crucial part of advancing our knowledge about this terrible disease."
Genentech is sponsoring the crenezumab study. Dr. Reiman had no personal financial disclosures. Dr. Craft had no financial disclosures.
*Correction 5/20/12: An earlier version of this story misidentified the investigational immunotherapy drug that was associated with adverse events in Alzheimer’s disease patients.
BETHESDA, MD. – The first-ever primary prevention trial for Alzheimer’s disease will investigate whether immunotherapy can delay – or even stop – the disease in a group of subjects who are genetically destined to develop it.
The $100 million project is one of the first to receive a special federal allocation for Alzheimer’s research, as envisioned in President Obama’s 2011 National Alzheimer’s Project Act.
Federal dollars will also support a second major clinical trial – a placebo-controlled study designed to evaluate the effect of intranasal insulin on cognition, function, and biomarkers in individuals with mild cognitive impairment and early Alzheimer’s.
These two projects will consume about half of the additional $50 million in Alzheimer’s project funding allocated by Congress for this fiscal year. Next year’s additional federal boost of $80 million will support more new studies, according to Dr. Ronald Petersen, chairman of the National Alzheimer’s Project Act’s Advisory Council on Alzheimer’s Research, Care, and Services.
"More studies are coming," he said at a research summit sponsored by the National Institute on Aging. These will include experiments with pluripotent stem cells, which have recently been differentiated into functional human Alzheimer’s neurons, and a large longitudinal study of the link between Alzheimer’s and cardiovascular disease.
Dr. Eric Reiman is one of three primary investigators on the 5-year immunotherapy trial. The placebo-controlled trial will give the humanized monoclonal antibody crenezumab to a group of about 300 members of an extended family in Colombia before they show symptoms of the disease. Many in this family express a mutation in the presenilin-1 gene that causes very early onset Alzheimer’s, with symptoms beginning before the age of 45 years. The trial will also include a smaller number of family members in the United States.
Half of the individuals will receive infusions of the drug, and half will receive a placebo. An additional group of individuals without the mutation will also receive a placebo, he added.
The participants will be followed for up to 5 years, with periodic cognitive, biomarker, and imaging assessments, said Dr. Reiman, executive director of the Banner Alzheimer’s Institute, Phoenix. After 2 years, a data safety monitoring committee will evaluate any progress and determine whether the trial should proceed.
Immunotherapy has walked a rocky path in Alzheimer’s drug development. The first active immunization clinical trial, which tested AN1792* in AD patients, was halted when a subset of patients developed meningoencephalitis. The first passive immunotherapy trial with bapineuzumab, showed vasogenic edema and microhemorrhages in 12 patients, but this drug continues to be studied.
The crenezumab study is unique in one other way, Dr. Reiman said in an interview: All of the data generated will become publicly available as soon as the trial is complete.
"We’ll be sharing all the data and samples in the hope of faster development of preventive treatments. We’re all in this fight together and given the stakes, it’s time for a paradigm shift that can – hopefully – launch a new era in Alzheimer’s prevention research."
In addition to its $16 million federal grant, the study will draw on $15 million in philanthropic funds from the Banner Institute, Dr. Reiman added.
Suzanne Craft, Ph.D., is heading the insulin study. The 5-year study will randomize 240 subjects with mild cognitive impairment or early Alzheimer’s to intranasal insulin or placebo for 12 months. At the end of that time, placebo patients will receive insulin in a 12-month open-label study. Subjects will be assessed periodically for changes in cognition, function, and cerebrospinal biomarkers.
An $8 million federal grant makes the study possible, said Dr. Craft, professor of psychiatry and behavioral sciences at the University of Washington, Seattle, and director of the Memory Disorders Clinic at the American Lakes division of the VA Puget Sound Health Care System.
Her earlier work with insulin showed tantalizing promise. A 104-subject, placebo-controlled study found that doses of both 20 and 40 IU of intranasal insulin preserved cognition and function in patients with mild cognitive impairment and early Alzheimer’s (Arch. Neurol. 2012;69:29-38). There was also some hint that these changes were associated with improved beta-amyloid biomarkers in cerebrospinal fluid. Patients assigned to placebo also showed declines in neuronal metabolism compared with those taking insulin.
Data from the new insulin study will also go into the public domain, Dr. Craft said in an interview.
"I think that as we make these data available to people with good ideas, we’ll see a proliferation of results from them," she said. "It’s essential to bring together investigators from all different disciplines to look at the question from different angles. It’s a crucial part of advancing our knowledge about this terrible disease."
Genentech is sponsoring the crenezumab study. Dr. Reiman had no personal financial disclosures. Dr. Craft had no financial disclosures.
*Correction 5/20/12: An earlier version of this story misidentified the investigational immunotherapy drug that was associated with adverse events in Alzheimer’s disease patients.
BETHESDA, MD. – The first-ever primary prevention trial for Alzheimer’s disease will investigate whether immunotherapy can delay – or even stop – the disease in a group of subjects who are genetically destined to develop it.
The $100 million project is one of the first to receive a special federal allocation for Alzheimer’s research, as envisioned in President Obama’s 2011 National Alzheimer’s Project Act.
Federal dollars will also support a second major clinical trial – a placebo-controlled study designed to evaluate the effect of intranasal insulin on cognition, function, and biomarkers in individuals with mild cognitive impairment and early Alzheimer’s.
These two projects will consume about half of the additional $50 million in Alzheimer’s project funding allocated by Congress for this fiscal year. Next year’s additional federal boost of $80 million will support more new studies, according to Dr. Ronald Petersen, chairman of the National Alzheimer’s Project Act’s Advisory Council on Alzheimer’s Research, Care, and Services.
"More studies are coming," he said at a research summit sponsored by the National Institute on Aging. These will include experiments with pluripotent stem cells, which have recently been differentiated into functional human Alzheimer’s neurons, and a large longitudinal study of the link between Alzheimer’s and cardiovascular disease.
Dr. Eric Reiman is one of three primary investigators on the 5-year immunotherapy trial. The placebo-controlled trial will give the humanized monoclonal antibody crenezumab to a group of about 300 members of an extended family in Colombia before they show symptoms of the disease. Many in this family express a mutation in the presenilin-1 gene that causes very early onset Alzheimer’s, with symptoms beginning before the age of 45 years. The trial will also include a smaller number of family members in the United States.
Half of the individuals will receive infusions of the drug, and half will receive a placebo. An additional group of individuals without the mutation will also receive a placebo, he added.
The participants will be followed for up to 5 years, with periodic cognitive, biomarker, and imaging assessments, said Dr. Reiman, executive director of the Banner Alzheimer’s Institute, Phoenix. After 2 years, a data safety monitoring committee will evaluate any progress and determine whether the trial should proceed.
Immunotherapy has walked a rocky path in Alzheimer’s drug development. The first active immunization clinical trial, which tested AN1792* in AD patients, was halted when a subset of patients developed meningoencephalitis. The first passive immunotherapy trial with bapineuzumab, showed vasogenic edema and microhemorrhages in 12 patients, but this drug continues to be studied.
The crenezumab study is unique in one other way, Dr. Reiman said in an interview: All of the data generated will become publicly available as soon as the trial is complete.
"We’ll be sharing all the data and samples in the hope of faster development of preventive treatments. We’re all in this fight together and given the stakes, it’s time for a paradigm shift that can – hopefully – launch a new era in Alzheimer’s prevention research."
In addition to its $16 million federal grant, the study will draw on $15 million in philanthropic funds from the Banner Institute, Dr. Reiman added.
Suzanne Craft, Ph.D., is heading the insulin study. The 5-year study will randomize 240 subjects with mild cognitive impairment or early Alzheimer’s to intranasal insulin or placebo for 12 months. At the end of that time, placebo patients will receive insulin in a 12-month open-label study. Subjects will be assessed periodically for changes in cognition, function, and cerebrospinal biomarkers.
An $8 million federal grant makes the study possible, said Dr. Craft, professor of psychiatry and behavioral sciences at the University of Washington, Seattle, and director of the Memory Disorders Clinic at the American Lakes division of the VA Puget Sound Health Care System.
Her earlier work with insulin showed tantalizing promise. A 104-subject, placebo-controlled study found that doses of both 20 and 40 IU of intranasal insulin preserved cognition and function in patients with mild cognitive impairment and early Alzheimer’s (Arch. Neurol. 2012;69:29-38). There was also some hint that these changes were associated with improved beta-amyloid biomarkers in cerebrospinal fluid. Patients assigned to placebo also showed declines in neuronal metabolism compared with those taking insulin.
Data from the new insulin study will also go into the public domain, Dr. Craft said in an interview.
"I think that as we make these data available to people with good ideas, we’ll see a proliferation of results from them," she said. "It’s essential to bring together investigators from all different disciplines to look at the question from different angles. It’s a crucial part of advancing our knowledge about this terrible disease."
Genentech is sponsoring the crenezumab study. Dr. Reiman had no personal financial disclosures. Dr. Craft had no financial disclosures.
*Correction 5/20/12: An earlier version of this story misidentified the investigational immunotherapy drug that was associated with adverse events in Alzheimer’s disease patients.
FROM A RESEARCH SUMMIT SPONSORED BY THE NATIONAL INSTITUTE ON AGING
CDC: Indoor Tanning, Sunburns Still Common in Young Adults
Despite the documented increased use of sunscreen and protective clothing in recent years, up to 66% of young adults are still getting sunburned at least once a year.
The use of tanning beds also continues, according to the May 11 issue of the Morbidity and Mortality Report. A review of the National Health Information Survey found that up to 44% of women in the subgroup analysis reported using the devices in 2010, and that those who tan indoors do so up to 28 times each year. Even people with a family history of skin cancer or with recent sunburns are using the devices, said Anne Hartman, a biostatistician with the National Cancer Institute.
The findings are worrisome on a number of levels, said Dr. Daniel M. Siegel, president of the American Academy of Dermatology.
"It is distressing that the study found tanning bed use is higher in those with a family history of skin cancer and that sunburn prevalence remains high," he said in an interview. "This emphasizes how important it is to educate the public and encourage them to change their behaviors as they do not necessarily understand the dangers of sun exposure and indoor tanning. If they do, the risks may not be discouraging the behavior."
The sun protection study extracted data from 5 years of the National Health Interview Survey (NHIS). The survey includes detailed questions on sun exposure, the use of sunscreens and protective clothing, and other sun-protective behaviors from the 2000-2010 surveys (MMWR Morb. Mortal. Wkly. Rep. 2012;61:317-22).
Overall, 50% of respondents reported at least one sunburn during the past 12 months. This was highest among whites (66% in 2010) and lowest among blacks (11% in 2010). A similar number of men and women reported getting sunburned, and the prevalence of burning did not differ from 2000 to 2010 (about 50% each year).
Women were most likely to report sunscreen use and staying in the shade (37% and 35%, respectively). Sunscreen use increased significantly from 2000 to 2010, as did the use of clothing to the ankles (21% in 2000 and 26% in 2010). There were also significant increases in the practice of seeking shade (29% and 35%, respectively). However, there were no significant increases in the use of wide-brimmed hats or long-sleeved shirts.
White women were more likely to report shade use, with a significant increase each year. Black women were least likely to report using sunscreens each year.
Men most often reported using long clothing and staying in the shade (33% and 26%, respectively). Shade use among men increased from 18% in 2000 to 26% in 2010. Long protective clothing use increased from 28% to 33%, but changes in sunscreen use were not significant over that time, nor were changes in wearing a long-sleeved shirt or wide-brimmed hat.
Clinicians and policy-makers can help improve these behaviors by continuing to stress the importance of sun protection, Ms. Hartman said. "Evidence from a recent review by the U.S. Preventive Services Task Force suggests that behavioral counseling can increase sun protective behaviors, particularly among persons aged 10-24 years. Additionally, environmental and policy changes (for example, the provision of shade and sunscreen in recreational setting) could be promising strategies for creating social and physical environments that routinely promote sun protection for younger adults and persons of all ages" (Ann. Intern. Med. 2012 May 8 [Epub ahead of print]).
The indoor tanning study used NHIS data from 2010 and included information from 25,233 respondents aged 18 years and older.
Overall, 6% of respondents reported indoor tanning in the past 12 months, reported Ms. Hartman. Tanning was most common among young adults, with 12% of those aged 18-25 years and 9% of those aged 26-29 years using it.
A total of 9% of those with a family history of skin cancer reported indoor tanning, as did 8% of those who had a sunburn within the past 12 months. The associations with cancer and sunburn suggest that many people still don’t understand the dangers of indoor tanning, Ms. Hartman said.
Tanning was also related to socioeconomic status; 7% of those with some college or technical school and 7% of those with an income of more than 200% of the federal poverty level reporting indoor tanning.
The highest rates were in white women aged 18-21 years (32%), particularly those from the Midwest (44%). A total of 36% of Southern women aged 22-25 years also reported indoor tanning.
When white women did tan inside, they did so frequently, the report found. Overall, women reported an average of 20 times per year, with 58% reporting that they tanned 10 or more times per year. White women aged 18-21 years had the highest frequency of tanning (28 sessions per year), with 68% reporting a frequency of 10 or more times per year.
The findings from both studies suggest the need for even more education about sun protective behaviors, Dr. Marcus Plescia said in a press statement.
"More public health efforts, including providing shade and sunscreen in recreational settings, are needed to raise awareness of the importance of sun protection and sunburn prevention to reduce the burden of skin cancer," said Dr. Plescia, director of cancer prevention and control at the Centers for Disease Control and Prevention. "We must accelerate our efforts to educate young adults about the dangers of indoor tanning."
Ms. Hartman, Dr. Siegel, and Dr. Plescia have no relevant financial disclosures.
Despite the documented increased use of sunscreen and protective clothing in recent years, up to 66% of young adults are still getting sunburned at least once a year.
The use of tanning beds also continues, according to the May 11 issue of the Morbidity and Mortality Report. A review of the National Health Information Survey found that up to 44% of women in the subgroup analysis reported using the devices in 2010, and that those who tan indoors do so up to 28 times each year. Even people with a family history of skin cancer or with recent sunburns are using the devices, said Anne Hartman, a biostatistician with the National Cancer Institute.
The findings are worrisome on a number of levels, said Dr. Daniel M. Siegel, president of the American Academy of Dermatology.
"It is distressing that the study found tanning bed use is higher in those with a family history of skin cancer and that sunburn prevalence remains high," he said in an interview. "This emphasizes how important it is to educate the public and encourage them to change their behaviors as they do not necessarily understand the dangers of sun exposure and indoor tanning. If they do, the risks may not be discouraging the behavior."
The sun protection study extracted data from 5 years of the National Health Interview Survey (NHIS). The survey includes detailed questions on sun exposure, the use of sunscreens and protective clothing, and other sun-protective behaviors from the 2000-2010 surveys (MMWR Morb. Mortal. Wkly. Rep. 2012;61:317-22).
Overall, 50% of respondents reported at least one sunburn during the past 12 months. This was highest among whites (66% in 2010) and lowest among blacks (11% in 2010). A similar number of men and women reported getting sunburned, and the prevalence of burning did not differ from 2000 to 2010 (about 50% each year).
Women were most likely to report sunscreen use and staying in the shade (37% and 35%, respectively). Sunscreen use increased significantly from 2000 to 2010, as did the use of clothing to the ankles (21% in 2000 and 26% in 2010). There were also significant increases in the practice of seeking shade (29% and 35%, respectively). However, there were no significant increases in the use of wide-brimmed hats or long-sleeved shirts.
White women were more likely to report shade use, with a significant increase each year. Black women were least likely to report using sunscreens each year.
Men most often reported using long clothing and staying in the shade (33% and 26%, respectively). Shade use among men increased from 18% in 2000 to 26% in 2010. Long protective clothing use increased from 28% to 33%, but changes in sunscreen use were not significant over that time, nor were changes in wearing a long-sleeved shirt or wide-brimmed hat.
Clinicians and policy-makers can help improve these behaviors by continuing to stress the importance of sun protection, Ms. Hartman said. "Evidence from a recent review by the U.S. Preventive Services Task Force suggests that behavioral counseling can increase sun protective behaviors, particularly among persons aged 10-24 years. Additionally, environmental and policy changes (for example, the provision of shade and sunscreen in recreational setting) could be promising strategies for creating social and physical environments that routinely promote sun protection for younger adults and persons of all ages" (Ann. Intern. Med. 2012 May 8 [Epub ahead of print]).
The indoor tanning study used NHIS data from 2010 and included information from 25,233 respondents aged 18 years and older.
Overall, 6% of respondents reported indoor tanning in the past 12 months, reported Ms. Hartman. Tanning was most common among young adults, with 12% of those aged 18-25 years and 9% of those aged 26-29 years using it.
A total of 9% of those with a family history of skin cancer reported indoor tanning, as did 8% of those who had a sunburn within the past 12 months. The associations with cancer and sunburn suggest that many people still don’t understand the dangers of indoor tanning, Ms. Hartman said.
Tanning was also related to socioeconomic status; 7% of those with some college or technical school and 7% of those with an income of more than 200% of the federal poverty level reporting indoor tanning.
The highest rates were in white women aged 18-21 years (32%), particularly those from the Midwest (44%). A total of 36% of Southern women aged 22-25 years also reported indoor tanning.
When white women did tan inside, they did so frequently, the report found. Overall, women reported an average of 20 times per year, with 58% reporting that they tanned 10 or more times per year. White women aged 18-21 years had the highest frequency of tanning (28 sessions per year), with 68% reporting a frequency of 10 or more times per year.
The findings from both studies suggest the need for even more education about sun protective behaviors, Dr. Marcus Plescia said in a press statement.
"More public health efforts, including providing shade and sunscreen in recreational settings, are needed to raise awareness of the importance of sun protection and sunburn prevention to reduce the burden of skin cancer," said Dr. Plescia, director of cancer prevention and control at the Centers for Disease Control and Prevention. "We must accelerate our efforts to educate young adults about the dangers of indoor tanning."
Ms. Hartman, Dr. Siegel, and Dr. Plescia have no relevant financial disclosures.
Despite the documented increased use of sunscreen and protective clothing in recent years, up to 66% of young adults are still getting sunburned at least once a year.
The use of tanning beds also continues, according to the May 11 issue of the Morbidity and Mortality Report. A review of the National Health Information Survey found that up to 44% of women in the subgroup analysis reported using the devices in 2010, and that those who tan indoors do so up to 28 times each year. Even people with a family history of skin cancer or with recent sunburns are using the devices, said Anne Hartman, a biostatistician with the National Cancer Institute.
The findings are worrisome on a number of levels, said Dr. Daniel M. Siegel, president of the American Academy of Dermatology.
"It is distressing that the study found tanning bed use is higher in those with a family history of skin cancer and that sunburn prevalence remains high," he said in an interview. "This emphasizes how important it is to educate the public and encourage them to change their behaviors as they do not necessarily understand the dangers of sun exposure and indoor tanning. If they do, the risks may not be discouraging the behavior."
The sun protection study extracted data from 5 years of the National Health Interview Survey (NHIS). The survey includes detailed questions on sun exposure, the use of sunscreens and protective clothing, and other sun-protective behaviors from the 2000-2010 surveys (MMWR Morb. Mortal. Wkly. Rep. 2012;61:317-22).
Overall, 50% of respondents reported at least one sunburn during the past 12 months. This was highest among whites (66% in 2010) and lowest among blacks (11% in 2010). A similar number of men and women reported getting sunburned, and the prevalence of burning did not differ from 2000 to 2010 (about 50% each year).
Women were most likely to report sunscreen use and staying in the shade (37% and 35%, respectively). Sunscreen use increased significantly from 2000 to 2010, as did the use of clothing to the ankles (21% in 2000 and 26% in 2010). There were also significant increases in the practice of seeking shade (29% and 35%, respectively). However, there were no significant increases in the use of wide-brimmed hats or long-sleeved shirts.
White women were more likely to report shade use, with a significant increase each year. Black women were least likely to report using sunscreens each year.
Men most often reported using long clothing and staying in the shade (33% and 26%, respectively). Shade use among men increased from 18% in 2000 to 26% in 2010. Long protective clothing use increased from 28% to 33%, but changes in sunscreen use were not significant over that time, nor were changes in wearing a long-sleeved shirt or wide-brimmed hat.
Clinicians and policy-makers can help improve these behaviors by continuing to stress the importance of sun protection, Ms. Hartman said. "Evidence from a recent review by the U.S. Preventive Services Task Force suggests that behavioral counseling can increase sun protective behaviors, particularly among persons aged 10-24 years. Additionally, environmental and policy changes (for example, the provision of shade and sunscreen in recreational setting) could be promising strategies for creating social and physical environments that routinely promote sun protection for younger adults and persons of all ages" (Ann. Intern. Med. 2012 May 8 [Epub ahead of print]).
The indoor tanning study used NHIS data from 2010 and included information from 25,233 respondents aged 18 years and older.
Overall, 6% of respondents reported indoor tanning in the past 12 months, reported Ms. Hartman. Tanning was most common among young adults, with 12% of those aged 18-25 years and 9% of those aged 26-29 years using it.
A total of 9% of those with a family history of skin cancer reported indoor tanning, as did 8% of those who had a sunburn within the past 12 months. The associations with cancer and sunburn suggest that many people still don’t understand the dangers of indoor tanning, Ms. Hartman said.
Tanning was also related to socioeconomic status; 7% of those with some college or technical school and 7% of those with an income of more than 200% of the federal poverty level reporting indoor tanning.
The highest rates were in white women aged 18-21 years (32%), particularly those from the Midwest (44%). A total of 36% of Southern women aged 22-25 years also reported indoor tanning.
When white women did tan inside, they did so frequently, the report found. Overall, women reported an average of 20 times per year, with 58% reporting that they tanned 10 or more times per year. White women aged 18-21 years had the highest frequency of tanning (28 sessions per year), with 68% reporting a frequency of 10 or more times per year.
The findings from both studies suggest the need for even more education about sun protective behaviors, Dr. Marcus Plescia said in a press statement.
"More public health efforts, including providing shade and sunscreen in recreational settings, are needed to raise awareness of the importance of sun protection and sunburn prevention to reduce the burden of skin cancer," said Dr. Plescia, director of cancer prevention and control at the Centers for Disease Control and Prevention. "We must accelerate our efforts to educate young adults about the dangers of indoor tanning."
Ms. Hartman, Dr. Siegel, and Dr. Plescia have no relevant financial disclosures.
FROM THE MORBIDITY AND MORTALITY REPORT
Major Finding: Up to 65% of young adults reported at least one sunburn and up to 44% of women reported using a tanning bed in 2010.
Data Source: The studies included data from 11 years of the National Health Interview Survey.
Disclosures: Ms. Hartman, Dr. Siegel, and Dr. Plescia have no relevant financial disclosures.
For Infants With Bronchiolitis, Nasogastric Feeds Have Advantages
BOSTON – Nasogastric feeding is safe and feasible for infants with viral bronchiolitis.
Infants who received nutrition by NG tube did not experience any exacerbation of their respiratory illness and recovered just as quickly as those who received intravenous fluids, Dr. Amir Kugelman said at the annual meeting of the Pediatric Academic Societies.
Although his randomized controlled study wasn’t designed to evaluate nutritional outcomes, Dr. Kugelman, director of the pediatric pulmonary unit at Bnai Zion Medical Center, Haifa, Israel, said that NG feeding offers some advantages over intravenous fluids.
Intravenous fluids provide limited calories and no lipids or proteins, which can lead to a catabolic state during a time of increased nutritional needs. "Gastric tube feeding is more physiologic and allows mothers to continue breast-feeding" while their infant is unable to tolerate oral intake, said Dr. Kugelman. "There is also the possibility that better nutrition might enhance recovery."
His study included 51 infants (mean age, 2.5 months) who had moderate viral bronchiolitis. They could not sustain oral feeding because of an increased respiratory rate of at least 60 breaths per minute. Most of them (41) were positive for respiratory syncytial virus; 11 had developed pneumonia. There were no significant baseline differences except for length of illness – those randomized to the intravenous group had been ill an average of 4 days, compared with 3 days in the NG group.
The IV group received a continuous infusion of a standard 5% dextrose in normal saline solution. The NG group received a slow drip of breast milk or infant formula. Three infants on NG feeds were switched to IV because of vomiting, and one on an IV was switched to NG feeds because of poor IV access and fluid extravasation.
There were no significant differences in the duration of oxygen needed, the time to full oral feeds, or the length of stay, Dr. Kugelman said. There were no incidences of aspiration. Infants in the IV group had a trend toward a shorter length of stay (100 hours vs. 120 hours), but this is probably because they had a longer duration of illness before hospitalization, he said.
Dr. Kugelman had no financial disclosures.
BOSTON – Nasogastric feeding is safe and feasible for infants with viral bronchiolitis.
Infants who received nutrition by NG tube did not experience any exacerbation of their respiratory illness and recovered just as quickly as those who received intravenous fluids, Dr. Amir Kugelman said at the annual meeting of the Pediatric Academic Societies.
Although his randomized controlled study wasn’t designed to evaluate nutritional outcomes, Dr. Kugelman, director of the pediatric pulmonary unit at Bnai Zion Medical Center, Haifa, Israel, said that NG feeding offers some advantages over intravenous fluids.
Intravenous fluids provide limited calories and no lipids or proteins, which can lead to a catabolic state during a time of increased nutritional needs. "Gastric tube feeding is more physiologic and allows mothers to continue breast-feeding" while their infant is unable to tolerate oral intake, said Dr. Kugelman. "There is also the possibility that better nutrition might enhance recovery."
His study included 51 infants (mean age, 2.5 months) who had moderate viral bronchiolitis. They could not sustain oral feeding because of an increased respiratory rate of at least 60 breaths per minute. Most of them (41) were positive for respiratory syncytial virus; 11 had developed pneumonia. There were no significant baseline differences except for length of illness – those randomized to the intravenous group had been ill an average of 4 days, compared with 3 days in the NG group.
The IV group received a continuous infusion of a standard 5% dextrose in normal saline solution. The NG group received a slow drip of breast milk or infant formula. Three infants on NG feeds were switched to IV because of vomiting, and one on an IV was switched to NG feeds because of poor IV access and fluid extravasation.
There were no significant differences in the duration of oxygen needed, the time to full oral feeds, or the length of stay, Dr. Kugelman said. There were no incidences of aspiration. Infants in the IV group had a trend toward a shorter length of stay (100 hours vs. 120 hours), but this is probably because they had a longer duration of illness before hospitalization, he said.
Dr. Kugelman had no financial disclosures.
BOSTON – Nasogastric feeding is safe and feasible for infants with viral bronchiolitis.
Infants who received nutrition by NG tube did not experience any exacerbation of their respiratory illness and recovered just as quickly as those who received intravenous fluids, Dr. Amir Kugelman said at the annual meeting of the Pediatric Academic Societies.
Although his randomized controlled study wasn’t designed to evaluate nutritional outcomes, Dr. Kugelman, director of the pediatric pulmonary unit at Bnai Zion Medical Center, Haifa, Israel, said that NG feeding offers some advantages over intravenous fluids.
Intravenous fluids provide limited calories and no lipids or proteins, which can lead to a catabolic state during a time of increased nutritional needs. "Gastric tube feeding is more physiologic and allows mothers to continue breast-feeding" while their infant is unable to tolerate oral intake, said Dr. Kugelman. "There is also the possibility that better nutrition might enhance recovery."
His study included 51 infants (mean age, 2.5 months) who had moderate viral bronchiolitis. They could not sustain oral feeding because of an increased respiratory rate of at least 60 breaths per minute. Most of them (41) were positive for respiratory syncytial virus; 11 had developed pneumonia. There were no significant baseline differences except for length of illness – those randomized to the intravenous group had been ill an average of 4 days, compared with 3 days in the NG group.
The IV group received a continuous infusion of a standard 5% dextrose in normal saline solution. The NG group received a slow drip of breast milk or infant formula. Three infants on NG feeds were switched to IV because of vomiting, and one on an IV was switched to NG feeds because of poor IV access and fluid extravasation.
There were no significant differences in the duration of oxygen needed, the time to full oral feeds, or the length of stay, Dr. Kugelman said. There were no incidences of aspiration. Infants in the IV group had a trend toward a shorter length of stay (100 hours vs. 120 hours), but this is probably because they had a longer duration of illness before hospitalization, he said.
Dr. Kugelman had no financial disclosures.
FROM THE ANNUAL MEETING OF THE PEDIATRIC ACADEMIC SOCIETIES
Major Finding: Infants with moderate bronchiolitis who received nasogastric feeding recovered as well as did those with intravenous fluids.
Data Source: This randomized controlled trial included 51 infants.
Disclosures: Dr. Kugelman had no financial disclosures.