User login
Doug Brunk is a San Diego-based award-winning reporter who began covering health care in 1991. Before joining the company, he wrote for the health sciences division of Columbia University and was an associate editor at Contemporary Long Term Care magazine when it won a Jesse H. Neal Award. His work has been syndicated by the Los Angeles Times and he is the author of two books related to the University of Kentucky Wildcats men's basketball program. Doug has a master’s degree in magazine journalism from the S.I. Newhouse School of Public Communications at Syracuse University. Follow him on Twitter @dougbrunk.
Time to scrap LMWH for prevention of placenta-mediated pregnancy complications?
SAN DIEGO – Low molecular weight heparin does not appear to reduce the risk of recurrent placenta-mediated pregnancy complications in women with prior such complications, according to Marc Rodger, MD.
“It’s time to put the needles away for pregnant patients,” he said at the biennial summit of the Thrombosis & Hemostasis Societies of North America.
The pathophysiology of placenta-mediated pregnancy complications includes placental thrombosis. Thrombophilias predispose to the development of thrombosis in slow-flow circulation of the placenta. “It’s possible that the etiology mix of placental-mediated pregnancy complications includes thrombophilias, and by extension, that anticoagulants would prevent these complications,” said Dr. Rodger, a senior scientist at the hospital and professor at the University of Ottawa.
In a study from 1999, researchers demonstrated that patients with pregnancy-mediated placental complications were 8.2 times more likely to develop thrombophilia, compared with controls (N Engl J Med. 1999;340:9-13). “But as with positive initial case-control studies, subsequent work downplayed this association,” Dr. Rodger said. “Now, we’re at a point where we recognize that thrombophilias are weakly associated with recurrent early loss, late pregnancy loss, and severe preeclampsia ([odds ratio] of about 1.5-2.0 for all associations), while thrombophilias are not associated with nonsevere preeclampsia and small for gestational age.”
Currently, low-molecular-weight heparin (LMWH) is the preferred pharmacoprophylaxis in pregnancy. Unfractionated heparin, meanwhile, requires b.i.d. or t.i.d. injections, and has a 10-fold higher risk of heparin-induced thrombocytopenia and a greater than 10-fold higher risk of osteoporotic fracture. Warfarin is teratogenic antepartum and inconvenient postpartum, while direct oral anticoagulants cross the placenta and enter breast milk.
Downsides of LMWH include the burden of self-injections and costs of over $10,000 per antepartum period, Dr. Rodger said. Common side effects include minor bleeding and elevated liver function tests, and it complicates regional anesthetic options at term. Uncommon side effects include major bleeding, skin reactions, and postpartum wound complications, while rare but serious complications include heparin-induced thrombocytopenia and osteoporotic fractures.
He offered a hypothetical case. A 32-year-old woman with prior severe preeclampsia who delivered at 32 weeks asks you, “Should I be treated with LMWH in my next pregnancy?” What should you tell her? To answer this question, Dr. Rodger and his associates conducted a study-level meta-analysis of six randomized controlled trials that included 848 pregnant women with prior placenta-mediated pregnancy complications (Blood. 2014;123[6]:822-8). The primary objective was to determine the effect of LMWH in preventing placenta-mediated pregnancy complications in women with prior late placenta-mediated pregnancy complications. This included patients with or without thrombophilia who were treated with or without LMWH. The primary outcome was a composite of preeclampsia, birth of an SGA newborn, placental abruption, or pregnancy loss greater than 20 weeks. Overall, 67 (18.7%) of 358 of women being given prophylactic LMWH had recurrent severe placenta-mediated pregnancy complications, compared with 127 (42.9%) of 296 women with no LMWH (relative risk reduction, 0.52; P = .01, indicating moderate heterogeneity). They identified similar relative risk reductions with LMWH for individual outcomes, including any preeclampsia, severe preeclampsia, SGA below the 10th percentile, SGA below the 5th percentile, preterm delivery less than 37 weeks, and preterm delivery less than 34 weeks with minimal heterogeneity. They concluded that LMWH “may be a promising therapy for recurrent, especially severe, placenta-mediated pregnancy complications, but further research is required.”
At the meeting, Dr. Rodger noted that the positive studies in the analysis were single-center trials, “which are generally acknowledged to be of a lesser methodologic quality, and the majority of patients in these single-center trials are from a small area in the south of France. Multicenter trials don’t show an effect, so is it single-centeredness or is it something else? The other feature that’s distinct is that the positive trials recruited patients with prior severe complications only, while the negative trials included patients with nonsevere complications. So maybe LMWH works in patients who have a very strong phenotype that have had very bad prior complications. We can’t tease that out with a study-level meta-analysis because we’re getting average effects over heterogeneous groups of patients.”
To expand on the study-level meta-analysis, Dr. Rodger and his associates conducted a systematic review and individual patient data meta-analysis of eight randomized trials of 963 patients conducted between 2000 and 2013 of LMWH to prevent recurrent placenta-mediated pregnancy complications (Lancet. 2016;388:2629-41). “In this approach you get individual patient data from the trials, and you create a new randomized, controlled data set,” he explained. “That way we could tease out the patients who have had the prior severe complications and whether their mild or severe outcomes are being prevented or not.”
The study’s composite primary outcome was one or more of the following: early-onset or severe preeclampsia, SGA newborn below the 5th percentile, late pregnancy loss (over 20 weeks), or placental abruption. Dr. Rodger and his associates found that LMWH did not significantly reduce the risk of recurrent placenta-mediated pregnancy complications, compared with patients who did not receive LMWH (14% vs. 22%, respectively; P = .09). In subgroup analyses, however, LMWH in multicenter trials reduced the primary outcome in women with previous abruption (P = .006) but not in any of the other subgroups of previous complications. “There were small numbers of patients in this subgroup, though, so I would use caution,” Dr. Rodger said. Two recent randomized, controlled trials from separate investigators further support the overall null findings of the individual patient data meta-analysis (Obstet Gynecol. 2016;128[5]:1053-63 and Am J Obstet Gynecol. 2017 Mar;216[3]:296.e1-296.e14).
Revisiting the hypothetical case of a 32-year-old woman with prior severe preeclampsia who delivered at 32 weeks, Dr. Rodger said that he would “definitely not” recommend LMWH during her next pregnancy.
He acknowledged limitations of the systematic review, including the limited numbers of patients in subgroups and the large differences between single-center and multicenter trials. “We still can’t explain this, and it remains an open question that bugs me,” he said. “This has been seen in many disease areas. Empirically, single-centeredness leans toward positivity.”
He called for more research in women with recurrent pregnancy loss. Dr. Rodger reported having no financial disclosures.
SAN DIEGO – Low molecular weight heparin does not appear to reduce the risk of recurrent placenta-mediated pregnancy complications in women with prior such complications, according to Marc Rodger, MD.
“It’s time to put the needles away for pregnant patients,” he said at the biennial summit of the Thrombosis & Hemostasis Societies of North America.
The pathophysiology of placenta-mediated pregnancy complications includes placental thrombosis. Thrombophilias predispose to the development of thrombosis in slow-flow circulation of the placenta. “It’s possible that the etiology mix of placental-mediated pregnancy complications includes thrombophilias, and by extension, that anticoagulants would prevent these complications,” said Dr. Rodger, a senior scientist at the hospital and professor at the University of Ottawa.
In a study from 1999, researchers demonstrated that patients with pregnancy-mediated placental complications were 8.2 times more likely to develop thrombophilia, compared with controls (N Engl J Med. 1999;340:9-13). “But as with positive initial case-control studies, subsequent work downplayed this association,” Dr. Rodger said. “Now, we’re at a point where we recognize that thrombophilias are weakly associated with recurrent early loss, late pregnancy loss, and severe preeclampsia ([odds ratio] of about 1.5-2.0 for all associations), while thrombophilias are not associated with nonsevere preeclampsia and small for gestational age.”
Currently, low-molecular-weight heparin (LMWH) is the preferred pharmacoprophylaxis in pregnancy. Unfractionated heparin, meanwhile, requires b.i.d. or t.i.d. injections, and has a 10-fold higher risk of heparin-induced thrombocytopenia and a greater than 10-fold higher risk of osteoporotic fracture. Warfarin is teratogenic antepartum and inconvenient postpartum, while direct oral anticoagulants cross the placenta and enter breast milk.
Downsides of LMWH include the burden of self-injections and costs of over $10,000 per antepartum period, Dr. Rodger said. Common side effects include minor bleeding and elevated liver function tests, and it complicates regional anesthetic options at term. Uncommon side effects include major bleeding, skin reactions, and postpartum wound complications, while rare but serious complications include heparin-induced thrombocytopenia and osteoporotic fractures.
He offered a hypothetical case. A 32-year-old woman with prior severe preeclampsia who delivered at 32 weeks asks you, “Should I be treated with LMWH in my next pregnancy?” What should you tell her? To answer this question, Dr. Rodger and his associates conducted a study-level meta-analysis of six randomized controlled trials that included 848 pregnant women with prior placenta-mediated pregnancy complications (Blood. 2014;123[6]:822-8). The primary objective was to determine the effect of LMWH in preventing placenta-mediated pregnancy complications in women with prior late placenta-mediated pregnancy complications. This included patients with or without thrombophilia who were treated with or without LMWH. The primary outcome was a composite of preeclampsia, birth of an SGA newborn, placental abruption, or pregnancy loss greater than 20 weeks. Overall, 67 (18.7%) of 358 of women being given prophylactic LMWH had recurrent severe placenta-mediated pregnancy complications, compared with 127 (42.9%) of 296 women with no LMWH (relative risk reduction, 0.52; P = .01, indicating moderate heterogeneity). They identified similar relative risk reductions with LMWH for individual outcomes, including any preeclampsia, severe preeclampsia, SGA below the 10th percentile, SGA below the 5th percentile, preterm delivery less than 37 weeks, and preterm delivery less than 34 weeks with minimal heterogeneity. They concluded that LMWH “may be a promising therapy for recurrent, especially severe, placenta-mediated pregnancy complications, but further research is required.”
At the meeting, Dr. Rodger noted that the positive studies in the analysis were single-center trials, “which are generally acknowledged to be of a lesser methodologic quality, and the majority of patients in these single-center trials are from a small area in the south of France. Multicenter trials don’t show an effect, so is it single-centeredness or is it something else? The other feature that’s distinct is that the positive trials recruited patients with prior severe complications only, while the negative trials included patients with nonsevere complications. So maybe LMWH works in patients who have a very strong phenotype that have had very bad prior complications. We can’t tease that out with a study-level meta-analysis because we’re getting average effects over heterogeneous groups of patients.”
To expand on the study-level meta-analysis, Dr. Rodger and his associates conducted a systematic review and individual patient data meta-analysis of eight randomized trials of 963 patients conducted between 2000 and 2013 of LMWH to prevent recurrent placenta-mediated pregnancy complications (Lancet. 2016;388:2629-41). “In this approach you get individual patient data from the trials, and you create a new randomized, controlled data set,” he explained. “That way we could tease out the patients who have had the prior severe complications and whether their mild or severe outcomes are being prevented or not.”
The study’s composite primary outcome was one or more of the following: early-onset or severe preeclampsia, SGA newborn below the 5th percentile, late pregnancy loss (over 20 weeks), or placental abruption. Dr. Rodger and his associates found that LMWH did not significantly reduce the risk of recurrent placenta-mediated pregnancy complications, compared with patients who did not receive LMWH (14% vs. 22%, respectively; P = .09). In subgroup analyses, however, LMWH in multicenter trials reduced the primary outcome in women with previous abruption (P = .006) but not in any of the other subgroups of previous complications. “There were small numbers of patients in this subgroup, though, so I would use caution,” Dr. Rodger said. Two recent randomized, controlled trials from separate investigators further support the overall null findings of the individual patient data meta-analysis (Obstet Gynecol. 2016;128[5]:1053-63 and Am J Obstet Gynecol. 2017 Mar;216[3]:296.e1-296.e14).
Revisiting the hypothetical case of a 32-year-old woman with prior severe preeclampsia who delivered at 32 weeks, Dr. Rodger said that he would “definitely not” recommend LMWH during her next pregnancy.
He acknowledged limitations of the systematic review, including the limited numbers of patients in subgroups and the large differences between single-center and multicenter trials. “We still can’t explain this, and it remains an open question that bugs me,” he said. “This has been seen in many disease areas. Empirically, single-centeredness leans toward positivity.”
He called for more research in women with recurrent pregnancy loss. Dr. Rodger reported having no financial disclosures.
SAN DIEGO – Low molecular weight heparin does not appear to reduce the risk of recurrent placenta-mediated pregnancy complications in women with prior such complications, according to Marc Rodger, MD.
“It’s time to put the needles away for pregnant patients,” he said at the biennial summit of the Thrombosis & Hemostasis Societies of North America.
The pathophysiology of placenta-mediated pregnancy complications includes placental thrombosis. Thrombophilias predispose to the development of thrombosis in slow-flow circulation of the placenta. “It’s possible that the etiology mix of placental-mediated pregnancy complications includes thrombophilias, and by extension, that anticoagulants would prevent these complications,” said Dr. Rodger, a senior scientist at the hospital and professor at the University of Ottawa.
In a study from 1999, researchers demonstrated that patients with pregnancy-mediated placental complications were 8.2 times more likely to develop thrombophilia, compared with controls (N Engl J Med. 1999;340:9-13). “But as with positive initial case-control studies, subsequent work downplayed this association,” Dr. Rodger said. “Now, we’re at a point where we recognize that thrombophilias are weakly associated with recurrent early loss, late pregnancy loss, and severe preeclampsia ([odds ratio] of about 1.5-2.0 for all associations), while thrombophilias are not associated with nonsevere preeclampsia and small for gestational age.”
Currently, low-molecular-weight heparin (LMWH) is the preferred pharmacoprophylaxis in pregnancy. Unfractionated heparin, meanwhile, requires b.i.d. or t.i.d. injections, and has a 10-fold higher risk of heparin-induced thrombocytopenia and a greater than 10-fold higher risk of osteoporotic fracture. Warfarin is teratogenic antepartum and inconvenient postpartum, while direct oral anticoagulants cross the placenta and enter breast milk.
Downsides of LMWH include the burden of self-injections and costs of over $10,000 per antepartum period, Dr. Rodger said. Common side effects include minor bleeding and elevated liver function tests, and it complicates regional anesthetic options at term. Uncommon side effects include major bleeding, skin reactions, and postpartum wound complications, while rare but serious complications include heparin-induced thrombocytopenia and osteoporotic fractures.
He offered a hypothetical case. A 32-year-old woman with prior severe preeclampsia who delivered at 32 weeks asks you, “Should I be treated with LMWH in my next pregnancy?” What should you tell her? To answer this question, Dr. Rodger and his associates conducted a study-level meta-analysis of six randomized controlled trials that included 848 pregnant women with prior placenta-mediated pregnancy complications (Blood. 2014;123[6]:822-8). The primary objective was to determine the effect of LMWH in preventing placenta-mediated pregnancy complications in women with prior late placenta-mediated pregnancy complications. This included patients with or without thrombophilia who were treated with or without LMWH. The primary outcome was a composite of preeclampsia, birth of an SGA newborn, placental abruption, or pregnancy loss greater than 20 weeks. Overall, 67 (18.7%) of 358 of women being given prophylactic LMWH had recurrent severe placenta-mediated pregnancy complications, compared with 127 (42.9%) of 296 women with no LMWH (relative risk reduction, 0.52; P = .01, indicating moderate heterogeneity). They identified similar relative risk reductions with LMWH for individual outcomes, including any preeclampsia, severe preeclampsia, SGA below the 10th percentile, SGA below the 5th percentile, preterm delivery less than 37 weeks, and preterm delivery less than 34 weeks with minimal heterogeneity. They concluded that LMWH “may be a promising therapy for recurrent, especially severe, placenta-mediated pregnancy complications, but further research is required.”
At the meeting, Dr. Rodger noted that the positive studies in the analysis were single-center trials, “which are generally acknowledged to be of a lesser methodologic quality, and the majority of patients in these single-center trials are from a small area in the south of France. Multicenter trials don’t show an effect, so is it single-centeredness or is it something else? The other feature that’s distinct is that the positive trials recruited patients with prior severe complications only, while the negative trials included patients with nonsevere complications. So maybe LMWH works in patients who have a very strong phenotype that have had very bad prior complications. We can’t tease that out with a study-level meta-analysis because we’re getting average effects over heterogeneous groups of patients.”
To expand on the study-level meta-analysis, Dr. Rodger and his associates conducted a systematic review and individual patient data meta-analysis of eight randomized trials of 963 patients conducted between 2000 and 2013 of LMWH to prevent recurrent placenta-mediated pregnancy complications (Lancet. 2016;388:2629-41). “In this approach you get individual patient data from the trials, and you create a new randomized, controlled data set,” he explained. “That way we could tease out the patients who have had the prior severe complications and whether their mild or severe outcomes are being prevented or not.”
The study’s composite primary outcome was one or more of the following: early-onset or severe preeclampsia, SGA newborn below the 5th percentile, late pregnancy loss (over 20 weeks), or placental abruption. Dr. Rodger and his associates found that LMWH did not significantly reduce the risk of recurrent placenta-mediated pregnancy complications, compared with patients who did not receive LMWH (14% vs. 22%, respectively; P = .09). In subgroup analyses, however, LMWH in multicenter trials reduced the primary outcome in women with previous abruption (P = .006) but not in any of the other subgroups of previous complications. “There were small numbers of patients in this subgroup, though, so I would use caution,” Dr. Rodger said. Two recent randomized, controlled trials from separate investigators further support the overall null findings of the individual patient data meta-analysis (Obstet Gynecol. 2016;128[5]:1053-63 and Am J Obstet Gynecol. 2017 Mar;216[3]:296.e1-296.e14).
Revisiting the hypothetical case of a 32-year-old woman with prior severe preeclampsia who delivered at 32 weeks, Dr. Rodger said that he would “definitely not” recommend LMWH during her next pregnancy.
He acknowledged limitations of the systematic review, including the limited numbers of patients in subgroups and the large differences between single-center and multicenter trials. “We still can’t explain this, and it remains an open question that bugs me,” he said. “This has been seen in many disease areas. Empirically, single-centeredness leans toward positivity.”
He called for more research in women with recurrent pregnancy loss. Dr. Rodger reported having no financial disclosures.
REPORTING FROM THSNA 2018
Survey sheds light on consumer preferences regarding cosmetic procedures
DALLAS – The most important referral sources for cosmetic procedures are physicians and family members and friends, but there appears to be a knowledge gap as to which cosmetic providers are actually medical doctors, results from an online survey found.
“There are approximately 16 million cosmetic procedures performed in the U.S., and that number is growing rapidly,” study author Adam J. Wulkan, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “They’re performed by dermatologists, plastic surgeons, facial plastic surgeons, nurses, aestheticians, dentists, and more. Yet little is known regarding how consumers choose cosmetic procedures and providers.”
In an effort to elucidate how consumers research, self-educate, and choose cosmetic surgery procedures and providers, Dr. Wulkan and his associates used Survey Monkey to send a 20-item survey to 931 individuals in the United States. Respondents qualified for participation if they acknowledged having obtained or considered obtaining a cosmetic procedure. Of the 931 individuals polled, 323 (35%) met inclusion criteria; 84 (9%) had received a cosmetic procedure, and 239 (26%) had considered one. Nearly three-quarters of respondents (73%) were female; 22% of respondents were aged 18-29 years, 25% were aged 30-44 years, 29% were aged 45-59 years, and 24% were aged 60 years and older.
The top three sources for referral to cosmetic procedures/providers were physicians (67%), family or friends (57%), and Google searches (51%). However, fewer than half of respondents (42.5%) had a procedure performed after having a consultation. Reasons for this could be related to several factors, Dr. Wulkan said, including the cost of the procedure, fear of adverse events, or not being an appropriate candidate for treatment at the time of consultation.
The most popular cosmetic procedures were laser hair removal (28%), laser/light therapy (25%), abdominoplasty (25%), injectables (24%), and noninvasive fat reduction (24%). The survey also asked whether certain providers were medical doctors or not, and 89% of respondents thought plastic surgeons were medical doctors, and 82% thought that dermatologists were medical doctors; the respondents also thought dentists (52%), aestheticians (20%), and nurses (11%) were medical doctors. “There’s an education gap regarding which providers are medical doctors,” said Dr. Wulkan, who is a cosmetic laser and dermatology fellow at Massachusetts General Hospital, Boston. “With the growing number of nonphysician aesthetic providers, consumer education might be a valuable tool.”
Most respondents (82%) checked physician credentials prior to treatment. In addition, they were most likely to have their cosmetic procedures performed by either a plastic surgeon or by a dermatologist.
Dr. Wulkan acknowledged certain limitations of the study, including the sample size and reliability of the answers. He reported having no financial disclosures. One study coauthor had various ties to industry companies, including Allergan, Revance Therapeutics, and Cytrellis Biosystems.
DALLAS – The most important referral sources for cosmetic procedures are physicians and family members and friends, but there appears to be a knowledge gap as to which cosmetic providers are actually medical doctors, results from an online survey found.
“There are approximately 16 million cosmetic procedures performed in the U.S., and that number is growing rapidly,” study author Adam J. Wulkan, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “They’re performed by dermatologists, plastic surgeons, facial plastic surgeons, nurses, aestheticians, dentists, and more. Yet little is known regarding how consumers choose cosmetic procedures and providers.”
In an effort to elucidate how consumers research, self-educate, and choose cosmetic surgery procedures and providers, Dr. Wulkan and his associates used Survey Monkey to send a 20-item survey to 931 individuals in the United States. Respondents qualified for participation if they acknowledged having obtained or considered obtaining a cosmetic procedure. Of the 931 individuals polled, 323 (35%) met inclusion criteria; 84 (9%) had received a cosmetic procedure, and 239 (26%) had considered one. Nearly three-quarters of respondents (73%) were female; 22% of respondents were aged 18-29 years, 25% were aged 30-44 years, 29% were aged 45-59 years, and 24% were aged 60 years and older.
The top three sources for referral to cosmetic procedures/providers were physicians (67%), family or friends (57%), and Google searches (51%). However, fewer than half of respondents (42.5%) had a procedure performed after having a consultation. Reasons for this could be related to several factors, Dr. Wulkan said, including the cost of the procedure, fear of adverse events, or not being an appropriate candidate for treatment at the time of consultation.
The most popular cosmetic procedures were laser hair removal (28%), laser/light therapy (25%), abdominoplasty (25%), injectables (24%), and noninvasive fat reduction (24%). The survey also asked whether certain providers were medical doctors or not, and 89% of respondents thought plastic surgeons were medical doctors, and 82% thought that dermatologists were medical doctors; the respondents also thought dentists (52%), aestheticians (20%), and nurses (11%) were medical doctors. “There’s an education gap regarding which providers are medical doctors,” said Dr. Wulkan, who is a cosmetic laser and dermatology fellow at Massachusetts General Hospital, Boston. “With the growing number of nonphysician aesthetic providers, consumer education might be a valuable tool.”
Most respondents (82%) checked physician credentials prior to treatment. In addition, they were most likely to have their cosmetic procedures performed by either a plastic surgeon or by a dermatologist.
Dr. Wulkan acknowledged certain limitations of the study, including the sample size and reliability of the answers. He reported having no financial disclosures. One study coauthor had various ties to industry companies, including Allergan, Revance Therapeutics, and Cytrellis Biosystems.
DALLAS – The most important referral sources for cosmetic procedures are physicians and family members and friends, but there appears to be a knowledge gap as to which cosmetic providers are actually medical doctors, results from an online survey found.
“There are approximately 16 million cosmetic procedures performed in the U.S., and that number is growing rapidly,” study author Adam J. Wulkan, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “They’re performed by dermatologists, plastic surgeons, facial plastic surgeons, nurses, aestheticians, dentists, and more. Yet little is known regarding how consumers choose cosmetic procedures and providers.”
In an effort to elucidate how consumers research, self-educate, and choose cosmetic surgery procedures and providers, Dr. Wulkan and his associates used Survey Monkey to send a 20-item survey to 931 individuals in the United States. Respondents qualified for participation if they acknowledged having obtained or considered obtaining a cosmetic procedure. Of the 931 individuals polled, 323 (35%) met inclusion criteria; 84 (9%) had received a cosmetic procedure, and 239 (26%) had considered one. Nearly three-quarters of respondents (73%) were female; 22% of respondents were aged 18-29 years, 25% were aged 30-44 years, 29% were aged 45-59 years, and 24% were aged 60 years and older.
The top three sources for referral to cosmetic procedures/providers were physicians (67%), family or friends (57%), and Google searches (51%). However, fewer than half of respondents (42.5%) had a procedure performed after having a consultation. Reasons for this could be related to several factors, Dr. Wulkan said, including the cost of the procedure, fear of adverse events, or not being an appropriate candidate for treatment at the time of consultation.
The most popular cosmetic procedures were laser hair removal (28%), laser/light therapy (25%), abdominoplasty (25%), injectables (24%), and noninvasive fat reduction (24%). The survey also asked whether certain providers were medical doctors or not, and 89% of respondents thought plastic surgeons were medical doctors, and 82% thought that dermatologists were medical doctors; the respondents also thought dentists (52%), aestheticians (20%), and nurses (11%) were medical doctors. “There’s an education gap regarding which providers are medical doctors,” said Dr. Wulkan, who is a cosmetic laser and dermatology fellow at Massachusetts General Hospital, Boston. “With the growing number of nonphysician aesthetic providers, consumer education might be a valuable tool.”
Most respondents (82%) checked physician credentials prior to treatment. In addition, they were most likely to have their cosmetic procedures performed by either a plastic surgeon or by a dermatologist.
Dr. Wulkan acknowledged certain limitations of the study, including the sample size and reliability of the answers. He reported having no financial disclosures. One study coauthor had various ties to industry companies, including Allergan, Revance Therapeutics, and Cytrellis Biosystems.
REPORTING FROM ASLMS 2018
Key clinical point: An education gap exists regarding which cosmetic surgery providers are medical doctors.
Major finding: Physicians were the top source of referral to cosmetic procedures/providers, yet only 82% of consumers believe that dermatologists are medical doctors.
Study details: Responses from 323 consumers who completed a 20-item online survey about cosmetic surgery procedures and providers.
Disclosures: Dr. Wulkan reported having no financial disclosures. One study coauthor had various ties to industry companies, including Allergan, Revance, and Cytrellis.
VIDEO: Assessing consumer knowledge about cosmetic procedures
DALLAS – Most people rely on physicians, family, and friends to obtain relevant information about cosmetic procedures, but a knowledge gap exists regarding which cosmetic providers are medical doctors.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Those are two key findings from a national survey that set out to assess how consumers research, educate themselves, and choose cosmetic procedures and providers. At the annual conference of the American Society for Laser Medicine and Surgery, study author Adam J. Wulkan, MD, discussed results from the 20-item survey, which was based on responses from 323 people who have obtained or have considered obtaining a cosmetic procedure such as laser hair removal.
Dr. Wulkan is a dermatologist at Massachusetts General Hospital, Boston. He reported having no financial disclosures. Study coauthor Mathew Avram, MD, serves on the medical advisory board of Sciton and on the scientific advisory boards of Sienna Biopharmaceuticals, Cytrellis, and Allergan. He also is consultant for Merz Aesthetics, Allergan, Soliton, Invasix, and Revance, and has intellectual property with Cytrellis. He also holds stock options with Cytrellis, Invasix, and Zalea.
DALLAS – Most people rely on physicians, family, and friends to obtain relevant information about cosmetic procedures, but a knowledge gap exists regarding which cosmetic providers are medical doctors.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Those are two key findings from a national survey that set out to assess how consumers research, educate themselves, and choose cosmetic procedures and providers. At the annual conference of the American Society for Laser Medicine and Surgery, study author Adam J. Wulkan, MD, discussed results from the 20-item survey, which was based on responses from 323 people who have obtained or have considered obtaining a cosmetic procedure such as laser hair removal.
Dr. Wulkan is a dermatologist at Massachusetts General Hospital, Boston. He reported having no financial disclosures. Study coauthor Mathew Avram, MD, serves on the medical advisory board of Sciton and on the scientific advisory boards of Sienna Biopharmaceuticals, Cytrellis, and Allergan. He also is consultant for Merz Aesthetics, Allergan, Soliton, Invasix, and Revance, and has intellectual property with Cytrellis. He also holds stock options with Cytrellis, Invasix, and Zalea.
DALLAS – Most people rely on physicians, family, and friends to obtain relevant information about cosmetic procedures, but a knowledge gap exists regarding which cosmetic providers are medical doctors.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Those are two key findings from a national survey that set out to assess how consumers research, educate themselves, and choose cosmetic procedures and providers. At the annual conference of the American Society for Laser Medicine and Surgery, study author Adam J. Wulkan, MD, discussed results from the 20-item survey, which was based on responses from 323 people who have obtained or have considered obtaining a cosmetic procedure such as laser hair removal.
Dr. Wulkan is a dermatologist at Massachusetts General Hospital, Boston. He reported having no financial disclosures. Study coauthor Mathew Avram, MD, serves on the medical advisory board of Sciton and on the scientific advisory boards of Sienna Biopharmaceuticals, Cytrellis, and Allergan. He also is consultant for Merz Aesthetics, Allergan, Soliton, Invasix, and Revance, and has intellectual property with Cytrellis. He also holds stock options with Cytrellis, Invasix, and Zalea.
REPORTING FROM ASLMS 2018
Experts offer guidance on use of emicizumab
SAN DIEGO – Emicizumab is a safe and effective new therapy for individuals with hemophilia A and inhibitor antibodies that will likely provide a paradigm shift for managing this patient population, according to Michael U. Callaghan, MD.
“It’s a safe drug, but you do have to be cautious about treating breakthrough bleeds with activated prothrombin complex concentrate (aPCC) resistance in particular,” Dr. Callaghan, a pediatric hematologist/oncologist at Children’s Hospital of Michigan, Detroit, said in an interview at the biennial summit of the Thrombosis & Hemostasis Societies of North America. “Patients require laboratory monitoring, and you need to educate anyone who’s going to see the patient about how the drug affects laboratory tests.”
Approved in November 2017, emicizumab (Hemlibra) is a recombinant, humanized bispecific immunoglobulin G4 monoclonal antibody that mimics the cofactor function of activated factor VIII (FVIIIa) by bridging activated factor IX and factor X. After 4 weeks of a loading dose of 3 mg/kg, subcutaneous once weekly dosing at 1.5 mg/kg demonstrated significant reduction in annualized bleeding rates in patients of all ages with congenital hemophilia A and inhibitors. But treatment-related adverse events occurred during the pivotal trials.
In an effort to provide recommendations on use of emicizumab beyond information contained in the agent’s package insert, Dr. Callaghan and his associates reviewed published literature, meeting abstracts, and expert experience with emicizumab on clinical trials.
Since emicizumab is highly selective for human FIXa and FX, only chromogenic FVIII assays using human reagents can assess emicizumab activity but those assays are not widely available, the researchers noted in their abstract. “In contrast, emicizumab does not affect chromogenic assays that contain bovine reagents and thus both native and infused factor FVIII levels as well as inhibitor titers (modified Bethesda assay) can be measured using this platform.”
In a phase 3 trial of emicizumab known as HAVEN 1, serious adverse events included three cases of thrombotic microangiopathy (TMA) and two thrombotic events (TE) (N Engl J Med 2017;377:809-18).* To prevent breakthrough bleeding, aPCC should be avoided unless there are no effective alternatives to control bleeding, Dr. Callaghan said. Treatment options for bleeding include bypassing agents such as human or recombinant porcine FVIII.
To prevent, monitor, and treat TMA and TE, prior to starting emicizumab, patients should be informed that baseline hemostasis is increased with the agent and that there is an increased risk of pathologic thrombosis with bypassing agents.
Patients should also be informed about the risk of TE/TMA and the signs and symptoms of TE/TMA. “If repeated dosing of bypass agents is needed, particularly aPCC, patients should contact their hemophilia treatment center,” the researchers wrote. “If TE/TMA is suspected, platelet count, creatinine, d-dimer, and fibrinogen should be monitored. If TE/TMA occur, emicizumab should be held and aPCC discontinued until resolution. Upon resolution of TE/TMA, consideration should be given to restarting emicizumab on a case-by-case basis.”
As for laboratory considerations, the researchers noted that results of activated partial prothrombin time (aPTT) will be shortened in patients on emicizumab, often into the normal range even at low concentrations. In addition, one-stage aPTT based factor VIII activity assays will yield high factor VIII activities, even at low concentrations of the drug. “Health care providers including dentists, surgeons, and emergency room staff need to be informed of the effects of emicizumab on laboratory tests,” they wrote in a poster at THSNA 2018.
HAVEN 1 and HAVEN 2 showed that 22 patients underwent 29 surgical procedures: tooth extractions (6), CVAD procedures (9), and other procedures (14). Of the 29 surgical procedures, 9 (31%) were managed with prophylactic bypassing agents, and one treated bleed occurred. At the same time, 20 procedures (69%) were managed without prophylactic bypassing agents, and two treated bleeds occurred.
The researchers concluded that additional studies are needed to inform the use of emicizumab in people with hemophilia A, with and without inhibitor antibodies.
Dr. Callaghan reported having no financial disclosures.
*Correction, 4/26/2018: An earlier version of this story misstated the number of cases of thrombotic microangiopathy.
SAN DIEGO – Emicizumab is a safe and effective new therapy for individuals with hemophilia A and inhibitor antibodies that will likely provide a paradigm shift for managing this patient population, according to Michael U. Callaghan, MD.
“It’s a safe drug, but you do have to be cautious about treating breakthrough bleeds with activated prothrombin complex concentrate (aPCC) resistance in particular,” Dr. Callaghan, a pediatric hematologist/oncologist at Children’s Hospital of Michigan, Detroit, said in an interview at the biennial summit of the Thrombosis & Hemostasis Societies of North America. “Patients require laboratory monitoring, and you need to educate anyone who’s going to see the patient about how the drug affects laboratory tests.”
Approved in November 2017, emicizumab (Hemlibra) is a recombinant, humanized bispecific immunoglobulin G4 monoclonal antibody that mimics the cofactor function of activated factor VIII (FVIIIa) by bridging activated factor IX and factor X. After 4 weeks of a loading dose of 3 mg/kg, subcutaneous once weekly dosing at 1.5 mg/kg demonstrated significant reduction in annualized bleeding rates in patients of all ages with congenital hemophilia A and inhibitors. But treatment-related adverse events occurred during the pivotal trials.
In an effort to provide recommendations on use of emicizumab beyond information contained in the agent’s package insert, Dr. Callaghan and his associates reviewed published literature, meeting abstracts, and expert experience with emicizumab on clinical trials.
Since emicizumab is highly selective for human FIXa and FX, only chromogenic FVIII assays using human reagents can assess emicizumab activity but those assays are not widely available, the researchers noted in their abstract. “In contrast, emicizumab does not affect chromogenic assays that contain bovine reagents and thus both native and infused factor FVIII levels as well as inhibitor titers (modified Bethesda assay) can be measured using this platform.”
In a phase 3 trial of emicizumab known as HAVEN 1, serious adverse events included three cases of thrombotic microangiopathy (TMA) and two thrombotic events (TE) (N Engl J Med 2017;377:809-18).* To prevent breakthrough bleeding, aPCC should be avoided unless there are no effective alternatives to control bleeding, Dr. Callaghan said. Treatment options for bleeding include bypassing agents such as human or recombinant porcine FVIII.
To prevent, monitor, and treat TMA and TE, prior to starting emicizumab, patients should be informed that baseline hemostasis is increased with the agent and that there is an increased risk of pathologic thrombosis with bypassing agents.
Patients should also be informed about the risk of TE/TMA and the signs and symptoms of TE/TMA. “If repeated dosing of bypass agents is needed, particularly aPCC, patients should contact their hemophilia treatment center,” the researchers wrote. “If TE/TMA is suspected, platelet count, creatinine, d-dimer, and fibrinogen should be monitored. If TE/TMA occur, emicizumab should be held and aPCC discontinued until resolution. Upon resolution of TE/TMA, consideration should be given to restarting emicizumab on a case-by-case basis.”
As for laboratory considerations, the researchers noted that results of activated partial prothrombin time (aPTT) will be shortened in patients on emicizumab, often into the normal range even at low concentrations. In addition, one-stage aPTT based factor VIII activity assays will yield high factor VIII activities, even at low concentrations of the drug. “Health care providers including dentists, surgeons, and emergency room staff need to be informed of the effects of emicizumab on laboratory tests,” they wrote in a poster at THSNA 2018.
HAVEN 1 and HAVEN 2 showed that 22 patients underwent 29 surgical procedures: tooth extractions (6), CVAD procedures (9), and other procedures (14). Of the 29 surgical procedures, 9 (31%) were managed with prophylactic bypassing agents, and one treated bleed occurred. At the same time, 20 procedures (69%) were managed without prophylactic bypassing agents, and two treated bleeds occurred.
The researchers concluded that additional studies are needed to inform the use of emicizumab in people with hemophilia A, with and without inhibitor antibodies.
Dr. Callaghan reported having no financial disclosures.
*Correction, 4/26/2018: An earlier version of this story misstated the number of cases of thrombotic microangiopathy.
SAN DIEGO – Emicizumab is a safe and effective new therapy for individuals with hemophilia A and inhibitor antibodies that will likely provide a paradigm shift for managing this patient population, according to Michael U. Callaghan, MD.
“It’s a safe drug, but you do have to be cautious about treating breakthrough bleeds with activated prothrombin complex concentrate (aPCC) resistance in particular,” Dr. Callaghan, a pediatric hematologist/oncologist at Children’s Hospital of Michigan, Detroit, said in an interview at the biennial summit of the Thrombosis & Hemostasis Societies of North America. “Patients require laboratory monitoring, and you need to educate anyone who’s going to see the patient about how the drug affects laboratory tests.”
Approved in November 2017, emicizumab (Hemlibra) is a recombinant, humanized bispecific immunoglobulin G4 monoclonal antibody that mimics the cofactor function of activated factor VIII (FVIIIa) by bridging activated factor IX and factor X. After 4 weeks of a loading dose of 3 mg/kg, subcutaneous once weekly dosing at 1.5 mg/kg demonstrated significant reduction in annualized bleeding rates in patients of all ages with congenital hemophilia A and inhibitors. But treatment-related adverse events occurred during the pivotal trials.
In an effort to provide recommendations on use of emicizumab beyond information contained in the agent’s package insert, Dr. Callaghan and his associates reviewed published literature, meeting abstracts, and expert experience with emicizumab on clinical trials.
Since emicizumab is highly selective for human FIXa and FX, only chromogenic FVIII assays using human reagents can assess emicizumab activity but those assays are not widely available, the researchers noted in their abstract. “In contrast, emicizumab does not affect chromogenic assays that contain bovine reagents and thus both native and infused factor FVIII levels as well as inhibitor titers (modified Bethesda assay) can be measured using this platform.”
In a phase 3 trial of emicizumab known as HAVEN 1, serious adverse events included three cases of thrombotic microangiopathy (TMA) and two thrombotic events (TE) (N Engl J Med 2017;377:809-18).* To prevent breakthrough bleeding, aPCC should be avoided unless there are no effective alternatives to control bleeding, Dr. Callaghan said. Treatment options for bleeding include bypassing agents such as human or recombinant porcine FVIII.
To prevent, monitor, and treat TMA and TE, prior to starting emicizumab, patients should be informed that baseline hemostasis is increased with the agent and that there is an increased risk of pathologic thrombosis with bypassing agents.
Patients should also be informed about the risk of TE/TMA and the signs and symptoms of TE/TMA. “If repeated dosing of bypass agents is needed, particularly aPCC, patients should contact their hemophilia treatment center,” the researchers wrote. “If TE/TMA is suspected, platelet count, creatinine, d-dimer, and fibrinogen should be monitored. If TE/TMA occur, emicizumab should be held and aPCC discontinued until resolution. Upon resolution of TE/TMA, consideration should be given to restarting emicizumab on a case-by-case basis.”
As for laboratory considerations, the researchers noted that results of activated partial prothrombin time (aPTT) will be shortened in patients on emicizumab, often into the normal range even at low concentrations. In addition, one-stage aPTT based factor VIII activity assays will yield high factor VIII activities, even at low concentrations of the drug. “Health care providers including dentists, surgeons, and emergency room staff need to be informed of the effects of emicizumab on laboratory tests,” they wrote in a poster at THSNA 2018.
HAVEN 1 and HAVEN 2 showed that 22 patients underwent 29 surgical procedures: tooth extractions (6), CVAD procedures (9), and other procedures (14). Of the 29 surgical procedures, 9 (31%) were managed with prophylactic bypassing agents, and one treated bleed occurred. At the same time, 20 procedures (69%) were managed without prophylactic bypassing agents, and two treated bleeds occurred.
The researchers concluded that additional studies are needed to inform the use of emicizumab in people with hemophilia A, with and without inhibitor antibodies.
Dr. Callaghan reported having no financial disclosures.
*Correction, 4/26/2018: An earlier version of this story misstated the number of cases of thrombotic microangiopathy.
EXPERT ANALYSIS FROM THSNA 2018
Probe linked to smartphone found effective in diagnosing oral cancer
DALLAS – A low-cost , in a clinical study of 92 people.
“Oral cancer is the sixth most common cancer in the world, but it’s the only major cancer whose outcome has not improved in the last 50 years,” study author Petra Wilder-Smith DDS, PhD, said in an interview following the annual conference of the American Society for Laser Medicine and Surgery Inc. “The main challenge is that over two-thirds of oral cancers are detected after they’ve metastasized. When you get spread like that, your survival is about 20% at 5 years, whereas if you detect it before spread, your survival is about 80% at 5 years.”
At the meeting, Vania Firmalino, an undergraduate student at the University of California, Irvine, discussed efforts by Dr. Wilder-Smith, Rongguang Liang, PhD, of the College of Optical Sciences at the University of Arizona, Tucson, and their colleagues to develop and evaluate the screening performance of a novel, low-cost smartphone-based mini probe for oral cancer screening and oral potentially premalignant lesions (OPMLs). The device provides high-resolution polarized white light images in combination with autofluorescence (AF) imaging capability.
The researchers found that inter-subject variation at each location was small, but inter-site differences were considerable. For example, optical data from OPMLs and oral cancer sites differed from normal with regard to white-light reflectance intensities, vascular homogeneity, and standard deviation. The AF signal in OPMLs and oral cancers shifted progressively to the red, together with a diminished green fluorescence signal. The cloud-based diagnostic algorithm based on these properties performed well, with an agreement with standard-of-care diagnosis of 80.6%.
“Artificial intelligence improves with data,” said Dr. Wilder-Smith, who is also a senior fellow at the university’s Chao Family Comprehensive Cancer Center. “We trained this system on about 200 images. When you’re up to 1,000 images per condition, that’s when you really start to get the benefits of artificial intelligence and machine learning. There’s huge potential here, especially when you think that 40% of the world’s risk for oral cancer is in India, which has good cell phone coverage. India also has a government-financed public health program whereby they already send health care workers to the remote areas of India to screen for basic diseases.”
The study won an award for best overall clinical abstract at the meeting. Dr. Wilder-Smith reported having no financial disclosures. The project was supported with funding from the National Institute of Biomedical Imaging and Bioengineering and the Beckman Foundation.
DALLAS – A low-cost , in a clinical study of 92 people.
“Oral cancer is the sixth most common cancer in the world, but it’s the only major cancer whose outcome has not improved in the last 50 years,” study author Petra Wilder-Smith DDS, PhD, said in an interview following the annual conference of the American Society for Laser Medicine and Surgery Inc. “The main challenge is that over two-thirds of oral cancers are detected after they’ve metastasized. When you get spread like that, your survival is about 20% at 5 years, whereas if you detect it before spread, your survival is about 80% at 5 years.”
At the meeting, Vania Firmalino, an undergraduate student at the University of California, Irvine, discussed efforts by Dr. Wilder-Smith, Rongguang Liang, PhD, of the College of Optical Sciences at the University of Arizona, Tucson, and their colleagues to develop and evaluate the screening performance of a novel, low-cost smartphone-based mini probe for oral cancer screening and oral potentially premalignant lesions (OPMLs). The device provides high-resolution polarized white light images in combination with autofluorescence (AF) imaging capability.
The researchers found that inter-subject variation at each location was small, but inter-site differences were considerable. For example, optical data from OPMLs and oral cancer sites differed from normal with regard to white-light reflectance intensities, vascular homogeneity, and standard deviation. The AF signal in OPMLs and oral cancers shifted progressively to the red, together with a diminished green fluorescence signal. The cloud-based diagnostic algorithm based on these properties performed well, with an agreement with standard-of-care diagnosis of 80.6%.
“Artificial intelligence improves with data,” said Dr. Wilder-Smith, who is also a senior fellow at the university’s Chao Family Comprehensive Cancer Center. “We trained this system on about 200 images. When you’re up to 1,000 images per condition, that’s when you really start to get the benefits of artificial intelligence and machine learning. There’s huge potential here, especially when you think that 40% of the world’s risk for oral cancer is in India, which has good cell phone coverage. India also has a government-financed public health program whereby they already send health care workers to the remote areas of India to screen for basic diseases.”
The study won an award for best overall clinical abstract at the meeting. Dr. Wilder-Smith reported having no financial disclosures. The project was supported with funding from the National Institute of Biomedical Imaging and Bioengineering and the Beckman Foundation.
DALLAS – A low-cost , in a clinical study of 92 people.
“Oral cancer is the sixth most common cancer in the world, but it’s the only major cancer whose outcome has not improved in the last 50 years,” study author Petra Wilder-Smith DDS, PhD, said in an interview following the annual conference of the American Society for Laser Medicine and Surgery Inc. “The main challenge is that over two-thirds of oral cancers are detected after they’ve metastasized. When you get spread like that, your survival is about 20% at 5 years, whereas if you detect it before spread, your survival is about 80% at 5 years.”
At the meeting, Vania Firmalino, an undergraduate student at the University of California, Irvine, discussed efforts by Dr. Wilder-Smith, Rongguang Liang, PhD, of the College of Optical Sciences at the University of Arizona, Tucson, and their colleagues to develop and evaluate the screening performance of a novel, low-cost smartphone-based mini probe for oral cancer screening and oral potentially premalignant lesions (OPMLs). The device provides high-resolution polarized white light images in combination with autofluorescence (AF) imaging capability.
The researchers found that inter-subject variation at each location was small, but inter-site differences were considerable. For example, optical data from OPMLs and oral cancer sites differed from normal with regard to white-light reflectance intensities, vascular homogeneity, and standard deviation. The AF signal in OPMLs and oral cancers shifted progressively to the red, together with a diminished green fluorescence signal. The cloud-based diagnostic algorithm based on these properties performed well, with an agreement with standard-of-care diagnosis of 80.6%.
“Artificial intelligence improves with data,” said Dr. Wilder-Smith, who is also a senior fellow at the university’s Chao Family Comprehensive Cancer Center. “We trained this system on about 200 images. When you’re up to 1,000 images per condition, that’s when you really start to get the benefits of artificial intelligence and machine learning. There’s huge potential here, especially when you think that 40% of the world’s risk for oral cancer is in India, which has good cell phone coverage. India also has a government-financed public health program whereby they already send health care workers to the remote areas of India to screen for basic diseases.”
The study won an award for best overall clinical abstract at the meeting. Dr. Wilder-Smith reported having no financial disclosures. The project was supported with funding from the National Institute of Biomedical Imaging and Bioengineering and the Beckman Foundation.
REPORTING FROM ASLMS 2018
Key clinical point: A compact oral probe that links to a smartphone was able to detect oral cancer.
Major finding: The optical diagnostic probe had a high rate of agreement (80.6%) with standard-of-care diagnosis.
Study details: A clinical analysis of 92 people with visually healthy oral mucosa or oral leukoplakia, erythroplakia, or ulceration.
Disclosures: Dr. Wilder-Smith reported having no financial disclosures. The National Institute of Biomedical Imaging and Bioengineering and the Beckman Foundation funded the project.
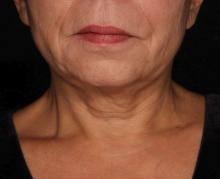
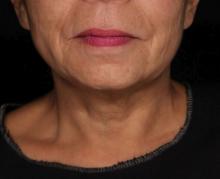
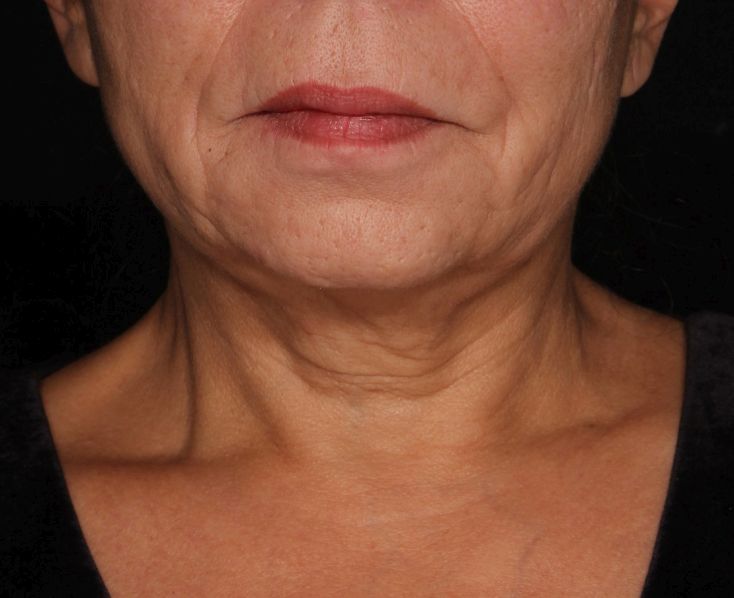
Picosecond 755-nm laser found effective for neck rejuvenation
DALLAS –
“It’s important to note that response was variable, but many patients were satisfied with the treatment,” Hana Jeon, MD said at the annual conference of the American Society for Laser Medicine and Surgery, Inc. “Further studies are needed to identify the clinical characteristics of neck laxity that would most benefit from this treatment.”
The researchers enrolled 25 patients with an average age of 58 years. The laser treatment settings were a 6-mm spot side-delivered at a fluence of 0.71 J/cm2 in a pulse width of 750 picoseconds. The patients were treated five times on the neck every 2-4 weeks, and follow-up visits were scheduled for 1 month and 3 months after the last treatment. Digital photos were taken at each visit. Formal assessment tools included patient and physician satisfaction scores and the Global Aesthetic Improvement Scale. In all, 21 women and 3 men completed the study. The majority (72%) had Fitzpatrick skin type II, while 16% had type III, 8% had type I, and 4% had type IV. An average of 5,042 pulses were delivered during each treatment session. The majority of patients (84%) required no anesthesia, while the rest used topical numbing medicine from 30 minutes to an hour prior to the procedure.
Dr. Jeon reported that the average pain score during the procedure was 4.7 on a 10-point scale. Forced-air cooling was used for comfort, and on average, mild redness following the treatment lasted less than 1 day (a mean of 0.6 days, with a range of 0-5 days). Mild pain also lasted less than 1 day (a mean of 0.1 days, with a range of 0-2 days). No swelling, crusting, bruising, bleeding, infection, blistering, scarring, burn, or dyspigmentation occurred.
On the Global Aesthetic Improvement Scale at 1 and 3 months, physicians described 43% and 23% of cases, respectively, as “improved,” 17% and 18% of cases as “much improved,” and 4% and 9% of cases as “extremely improved.”
At 3 months, 35% of patients said they would be “somewhat likely” to recommend the procedure, and 30% said they would be “extremely likely” to recommend it.
Dr. Jeon reported having no financial disclosures.
DALLAS –
“It’s important to note that response was variable, but many patients were satisfied with the treatment,” Hana Jeon, MD said at the annual conference of the American Society for Laser Medicine and Surgery, Inc. “Further studies are needed to identify the clinical characteristics of neck laxity that would most benefit from this treatment.”
The researchers enrolled 25 patients with an average age of 58 years. The laser treatment settings were a 6-mm spot side-delivered at a fluence of 0.71 J/cm2 in a pulse width of 750 picoseconds. The patients were treated five times on the neck every 2-4 weeks, and follow-up visits were scheduled for 1 month and 3 months after the last treatment. Digital photos were taken at each visit. Formal assessment tools included patient and physician satisfaction scores and the Global Aesthetic Improvement Scale. In all, 21 women and 3 men completed the study. The majority (72%) had Fitzpatrick skin type II, while 16% had type III, 8% had type I, and 4% had type IV. An average of 5,042 pulses were delivered during each treatment session. The majority of patients (84%) required no anesthesia, while the rest used topical numbing medicine from 30 minutes to an hour prior to the procedure.
Dr. Jeon reported that the average pain score during the procedure was 4.7 on a 10-point scale. Forced-air cooling was used for comfort, and on average, mild redness following the treatment lasted less than 1 day (a mean of 0.6 days, with a range of 0-5 days). Mild pain also lasted less than 1 day (a mean of 0.1 days, with a range of 0-2 days). No swelling, crusting, bruising, bleeding, infection, blistering, scarring, burn, or dyspigmentation occurred.
On the Global Aesthetic Improvement Scale at 1 and 3 months, physicians described 43% and 23% of cases, respectively, as “improved,” 17% and 18% of cases as “much improved,” and 4% and 9% of cases as “extremely improved.”
At 3 months, 35% of patients said they would be “somewhat likely” to recommend the procedure, and 30% said they would be “extremely likely” to recommend it.
Dr. Jeon reported having no financial disclosures.
DALLAS –
“It’s important to note that response was variable, but many patients were satisfied with the treatment,” Hana Jeon, MD said at the annual conference of the American Society for Laser Medicine and Surgery, Inc. “Further studies are needed to identify the clinical characteristics of neck laxity that would most benefit from this treatment.”
The researchers enrolled 25 patients with an average age of 58 years. The laser treatment settings were a 6-mm spot side-delivered at a fluence of 0.71 J/cm2 in a pulse width of 750 picoseconds. The patients were treated five times on the neck every 2-4 weeks, and follow-up visits were scheduled for 1 month and 3 months after the last treatment. Digital photos were taken at each visit. Formal assessment tools included patient and physician satisfaction scores and the Global Aesthetic Improvement Scale. In all, 21 women and 3 men completed the study. The majority (72%) had Fitzpatrick skin type II, while 16% had type III, 8% had type I, and 4% had type IV. An average of 5,042 pulses were delivered during each treatment session. The majority of patients (84%) required no anesthesia, while the rest used topical numbing medicine from 30 minutes to an hour prior to the procedure.
Dr. Jeon reported that the average pain score during the procedure was 4.7 on a 10-point scale. Forced-air cooling was used for comfort, and on average, mild redness following the treatment lasted less than 1 day (a mean of 0.6 days, with a range of 0-5 days). Mild pain also lasted less than 1 day (a mean of 0.1 days, with a range of 0-2 days). No swelling, crusting, bruising, bleeding, infection, blistering, scarring, burn, or dyspigmentation occurred.
On the Global Aesthetic Improvement Scale at 1 and 3 months, physicians described 43% and 23% of cases, respectively, as “improved,” 17% and 18% of cases as “much improved,” and 4% and 9% of cases as “extremely improved.”
At 3 months, 35% of patients said they would be “somewhat likely” to recommend the procedure, and 30% said they would be “extremely likely” to recommend it.
Dr. Jeon reported having no financial disclosures.
REPORTING FROM ASLMS 2018
Key clinical point: Response to using a picosecond 755-nm laser with focus lens array for neck rejuvenation was variable.
Major finding: On the Global Aesthetic Improvement Scale at 1 and 3 months, physicians described 43% and 23% of cases, respectively, as “improved.”
Study details: A single-center study of 25 patients treated for neck laxity.
Disclosures: Dr. Jeon reported having no financial disclosures.
CO2 laser guided by confocal microscopy effectively treated superficial BCC
DALLAS – The use of CO2 laser ablation guided by reflectance confocal microscopy is an effective, minimally invasive treatment for superficial and early nodular basal cell carcinoma (BCC), according to results from an ongoing study.
“While surgery is the gold standard for many basal cell carcinomas, nonsurgical therapies may be a good option for the superficial and early nodular subtypes,” lead study author Anthony M. Rossi, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “Laser ablation was used many years ago, so this is not a novel concept, but we’re bringing it back and we’re trying to use confocal microscopy to hone in on the basal cell and selectively target the tumor.”
For the current analysis, he and his associates used with a mean age of 55 years. Of the 20 lesions, 18 were located on the limbs and trunk, while two were on the head and neck. The median lesion diameter was 7 mm. Prior to laser ablation, the researchers performed reflectance confocal microscopy to define lateral and deep margins and define the laser parameters.
The median number of laser passes was three, and ranged from two to eight, delivered at a fluence of 7.5 J/cm2. Reflectance confocal microscopy was repeated immediately after the laser treatment to the skin wound margins and deep margins, and it was performed every 3-6 months thereafter. “If you do confocal microscopy too early, you’ll see mainly inflammation and you may see residual tumor that hasn’t been fully resolved yet,” Dr. Rossi said.
As for future directions, he and his colleagues are developing contrast agents to enhance the ability to detect BCC tumors in vivo, to highlight tumor islands, and to differentiate sebaceous glands and hair follicles. Dr. Rossi reported having no relevant disclosures.
DALLAS – The use of CO2 laser ablation guided by reflectance confocal microscopy is an effective, minimally invasive treatment for superficial and early nodular basal cell carcinoma (BCC), according to results from an ongoing study.
“While surgery is the gold standard for many basal cell carcinomas, nonsurgical therapies may be a good option for the superficial and early nodular subtypes,” lead study author Anthony M. Rossi, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “Laser ablation was used many years ago, so this is not a novel concept, but we’re bringing it back and we’re trying to use confocal microscopy to hone in on the basal cell and selectively target the tumor.”
For the current analysis, he and his associates used with a mean age of 55 years. Of the 20 lesions, 18 were located on the limbs and trunk, while two were on the head and neck. The median lesion diameter was 7 mm. Prior to laser ablation, the researchers performed reflectance confocal microscopy to define lateral and deep margins and define the laser parameters.
The median number of laser passes was three, and ranged from two to eight, delivered at a fluence of 7.5 J/cm2. Reflectance confocal microscopy was repeated immediately after the laser treatment to the skin wound margins and deep margins, and it was performed every 3-6 months thereafter. “If you do confocal microscopy too early, you’ll see mainly inflammation and you may see residual tumor that hasn’t been fully resolved yet,” Dr. Rossi said.
As for future directions, he and his colleagues are developing contrast agents to enhance the ability to detect BCC tumors in vivo, to highlight tumor islands, and to differentiate sebaceous glands and hair follicles. Dr. Rossi reported having no relevant disclosures.
DALLAS – The use of CO2 laser ablation guided by reflectance confocal microscopy is an effective, minimally invasive treatment for superficial and early nodular basal cell carcinoma (BCC), according to results from an ongoing study.
“While surgery is the gold standard for many basal cell carcinomas, nonsurgical therapies may be a good option for the superficial and early nodular subtypes,” lead study author Anthony M. Rossi, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “Laser ablation was used many years ago, so this is not a novel concept, but we’re bringing it back and we’re trying to use confocal microscopy to hone in on the basal cell and selectively target the tumor.”
For the current analysis, he and his associates used with a mean age of 55 years. Of the 20 lesions, 18 were located on the limbs and trunk, while two were on the head and neck. The median lesion diameter was 7 mm. Prior to laser ablation, the researchers performed reflectance confocal microscopy to define lateral and deep margins and define the laser parameters.
The median number of laser passes was three, and ranged from two to eight, delivered at a fluence of 7.5 J/cm2. Reflectance confocal microscopy was repeated immediately after the laser treatment to the skin wound margins and deep margins, and it was performed every 3-6 months thereafter. “If you do confocal microscopy too early, you’ll see mainly inflammation and you may see residual tumor that hasn’t been fully resolved yet,” Dr. Rossi said.
As for future directions, he and his colleagues are developing contrast agents to enhance the ability to detect BCC tumors in vivo, to highlight tumor islands, and to differentiate sebaceous glands and hair follicles. Dr. Rossi reported having no relevant disclosures.
REPORTING FROM ASLMS 2018
Key clinical point: Reflectance confocal microscopy-guided CO2 laser ablation of basal cell carcinoma (BCC) was found to be effective.
Major finding: After an average follow-up of 17 months, no recurrence of BCC has been detected clinically, dermoscopically, or by reflectance confocal microscopy.
Study details: A clinical analysis of seven adults with superficial BCC who were treated with a CO2 laser guided by confocal microscopy.
Disclosures: Dr. Rossi reported having no financial disclosures.
Noninvasive 1060-nm diode laser found effective for axillary hyperhidrosis
DALLAS – , a condition that affects an estimated 1.4% people in the United States.
“Newer treatments such as microwave technology, botulinum toxin injections, and lasers have emerged as effective methods,” lead study author Bruce E. Katz, MD, who directs the Juva Skin & Laser Center in New York, said at the annual conference of the American Society for Laser Medicine and Surgery Inc.
In a prospective pilot study, Dr. Katz and his associates examined the use of a noninvasive 1060-nm diode laser (SculpSure, Cynosure) in 10 subjects with moderate to severe hyperhidrosis of the axilla, which was defined as having Hyperhidrosis Disease Severity Scale (HDSS) scores of 3 or 4. The subjects, whose mean age was 42 years, received two laser treatments on the axillary area at 6-week intervals, and the researchers performed starch iodine tests and took two-dimensional photography of the axilla before and after treatment. Subjects were asked to rate their conditions on the HDSS and satisfaction with treatment. Two blinded dermatologist evaluators graded the reduction in sweat production on photos of starch iodine tests. The researchers also performed a retrospective analysis on all subjects to assess safety and efficacy of treatment.
The 1060-nm diode laser used for the study features four flat, nonsuction applicators. Dr. Katz and his associates positioned two applicators on the axilla simultaneously for 25 minutes. “The 1060-nm wavelength has minimal absorption in the melanin, so we can really treat any skin type,” he said. “It has a high affinity for adipose tissue, and we believe that its targeted effect at the level of the eccrine and apocrine glands should help improve hyperhidrosis. It works by elevating the tissue temperature to about 42 to 47 degrees Centigrade, without damaging surrounding tissue. The device has a sapphire cooling plate, so it’s comfortable for the patient.”
The retrospective analysis revealed minimal adverse events such as pain, mild erythema, and edema, which were transient and resolved without intervention in a day or two. To date, no cases have recurred, “which we were surprised by,” Dr. Katz said. “We were surprised that the effect lasted that long.”
SOURCE: Katz et. al. ASLMS 2018.
DALLAS – , a condition that affects an estimated 1.4% people in the United States.
“Newer treatments such as microwave technology, botulinum toxin injections, and lasers have emerged as effective methods,” lead study author Bruce E. Katz, MD, who directs the Juva Skin & Laser Center in New York, said at the annual conference of the American Society for Laser Medicine and Surgery Inc.
In a prospective pilot study, Dr. Katz and his associates examined the use of a noninvasive 1060-nm diode laser (SculpSure, Cynosure) in 10 subjects with moderate to severe hyperhidrosis of the axilla, which was defined as having Hyperhidrosis Disease Severity Scale (HDSS) scores of 3 or 4. The subjects, whose mean age was 42 years, received two laser treatments on the axillary area at 6-week intervals, and the researchers performed starch iodine tests and took two-dimensional photography of the axilla before and after treatment. Subjects were asked to rate their conditions on the HDSS and satisfaction with treatment. Two blinded dermatologist evaluators graded the reduction in sweat production on photos of starch iodine tests. The researchers also performed a retrospective analysis on all subjects to assess safety and efficacy of treatment.
The 1060-nm diode laser used for the study features four flat, nonsuction applicators. Dr. Katz and his associates positioned two applicators on the axilla simultaneously for 25 minutes. “The 1060-nm wavelength has minimal absorption in the melanin, so we can really treat any skin type,” he said. “It has a high affinity for adipose tissue, and we believe that its targeted effect at the level of the eccrine and apocrine glands should help improve hyperhidrosis. It works by elevating the tissue temperature to about 42 to 47 degrees Centigrade, without damaging surrounding tissue. The device has a sapphire cooling plate, so it’s comfortable for the patient.”
The retrospective analysis revealed minimal adverse events such as pain, mild erythema, and edema, which were transient and resolved without intervention in a day or two. To date, no cases have recurred, “which we were surprised by,” Dr. Katz said. “We were surprised that the effect lasted that long.”
SOURCE: Katz et. al. ASLMS 2018.
DALLAS – , a condition that affects an estimated 1.4% people in the United States.
“Newer treatments such as microwave technology, botulinum toxin injections, and lasers have emerged as effective methods,” lead study author Bruce E. Katz, MD, who directs the Juva Skin & Laser Center in New York, said at the annual conference of the American Society for Laser Medicine and Surgery Inc.
In a prospective pilot study, Dr. Katz and his associates examined the use of a noninvasive 1060-nm diode laser (SculpSure, Cynosure) in 10 subjects with moderate to severe hyperhidrosis of the axilla, which was defined as having Hyperhidrosis Disease Severity Scale (HDSS) scores of 3 or 4. The subjects, whose mean age was 42 years, received two laser treatments on the axillary area at 6-week intervals, and the researchers performed starch iodine tests and took two-dimensional photography of the axilla before and after treatment. Subjects were asked to rate their conditions on the HDSS and satisfaction with treatment. Two blinded dermatologist evaluators graded the reduction in sweat production on photos of starch iodine tests. The researchers also performed a retrospective analysis on all subjects to assess safety and efficacy of treatment.
The 1060-nm diode laser used for the study features four flat, nonsuction applicators. Dr. Katz and his associates positioned two applicators on the axilla simultaneously for 25 minutes. “The 1060-nm wavelength has minimal absorption in the melanin, so we can really treat any skin type,” he said. “It has a high affinity for adipose tissue, and we believe that its targeted effect at the level of the eccrine and apocrine glands should help improve hyperhidrosis. It works by elevating the tissue temperature to about 42 to 47 degrees Centigrade, without damaging surrounding tissue. The device has a sapphire cooling plate, so it’s comfortable for the patient.”
The retrospective analysis revealed minimal adverse events such as pain, mild erythema, and edema, which were transient and resolved without intervention in a day or two. To date, no cases have recurred, “which we were surprised by,” Dr. Katz said. “We were surprised that the effect lasted that long.”
SOURCE: Katz et. al. ASLMS 2018.
REPORTING FROM ASLMS 2018
Few physicians report adverse events related to body contouring devices
DALLAS – Most physicians do not report adverse events related to noninvasive body contouring devices to the Food and Drug Administration, a database review showed.
“The FDA mandates that manufacturers and device operators disclose medical device reports to monitor suspected injuries and device malfunctions to the Manufacturer and User Facility Device Experience [MAUDE] database,” study author Adam J. Wulkan, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “Given the rapid growth in the noninvasive fat reduction market, it is essential physicians be aware of associated adverse events.”
“Given that this is the largest database for adverse events, I was surprised how few reports were done by physicians,” Dr. Wulkan said in an interview.
He reported results from 83 of the 98 MDRs. A total of 26 MDRs reported on cryolipolysis and included newly diagnosed or exacerbation of prior umbilical hernias (10), neuropathies (5), paradoxical fat hyperplasias (4), blisters (3), rashes (1), flu-like symptoms (1), gastroenteritis (1), and prolapsed bladder/uterus/rectum (1). Of the 11 MDRs for radio-frequency devices, 10 were burns/blisters and 1 fire.
Dr. Wulkan went on to note that 17 MDRs were reported for 1060-nm laser lipolysis, including burns/blisters (5), nodules (4), pain (3), cellulitis/abscesses (2), excessive swelling (1), neuropathy (1), and vomiting (1). There were four cases of burns on MDRs from focused/pulsed ultrasound procedures. One was on a non–FDA approved body site and another after combination radio-frequency/infrared treatment for cellulite reduction.
There were eight MDRs reported with 1440-nm laser cellulite reduction, including burn (5), unacceptable cosmesis (2), and faulty power supply (1). In addition, there were 12 MDRs reported on high-frequency ultrasound, including burns/blisters (9), subcutaneous nodules (2), and excoriation (1). Finally, vacuum-assisted submission was associated with five MDRs, including festooning/unacceptable cosmesis (2), seroma/hematoma (1), laceration (1), and nodules (1).
Dr. Wulkan acknowledged certain limitations of the study, including the fact that not all MDRs are reported to MAUDE, and that the reliability of patient reporting can be questionable. “We have to figure out if there is a natural causation between the adverse event and the treatment itself,” he said at the meeting. “The number of MDRs for any given device must be correlated to the number of procedures performed before we jump to conclusions about the safety of any given device.”
He reported having no financial disclosures. One study coauthor holds positions on advisory boards of, is a consultant for, and has intellectual property and/or stock options with various industry companies, such as Soliton and Allergan.
SOURCE: Wulkan et. al., ASLMS 2018.
DALLAS – Most physicians do not report adverse events related to noninvasive body contouring devices to the Food and Drug Administration, a database review showed.
“The FDA mandates that manufacturers and device operators disclose medical device reports to monitor suspected injuries and device malfunctions to the Manufacturer and User Facility Device Experience [MAUDE] database,” study author Adam J. Wulkan, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “Given the rapid growth in the noninvasive fat reduction market, it is essential physicians be aware of associated adverse events.”
“Given that this is the largest database for adverse events, I was surprised how few reports were done by physicians,” Dr. Wulkan said in an interview.
He reported results from 83 of the 98 MDRs. A total of 26 MDRs reported on cryolipolysis and included newly diagnosed or exacerbation of prior umbilical hernias (10), neuropathies (5), paradoxical fat hyperplasias (4), blisters (3), rashes (1), flu-like symptoms (1), gastroenteritis (1), and prolapsed bladder/uterus/rectum (1). Of the 11 MDRs for radio-frequency devices, 10 were burns/blisters and 1 fire.
Dr. Wulkan went on to note that 17 MDRs were reported for 1060-nm laser lipolysis, including burns/blisters (5), nodules (4), pain (3), cellulitis/abscesses (2), excessive swelling (1), neuropathy (1), and vomiting (1). There were four cases of burns on MDRs from focused/pulsed ultrasound procedures. One was on a non–FDA approved body site and another after combination radio-frequency/infrared treatment for cellulite reduction.
There were eight MDRs reported with 1440-nm laser cellulite reduction, including burn (5), unacceptable cosmesis (2), and faulty power supply (1). In addition, there were 12 MDRs reported on high-frequency ultrasound, including burns/blisters (9), subcutaneous nodules (2), and excoriation (1). Finally, vacuum-assisted submission was associated with five MDRs, including festooning/unacceptable cosmesis (2), seroma/hematoma (1), laceration (1), and nodules (1).
Dr. Wulkan acknowledged certain limitations of the study, including the fact that not all MDRs are reported to MAUDE, and that the reliability of patient reporting can be questionable. “We have to figure out if there is a natural causation between the adverse event and the treatment itself,” he said at the meeting. “The number of MDRs for any given device must be correlated to the number of procedures performed before we jump to conclusions about the safety of any given device.”
He reported having no financial disclosures. One study coauthor holds positions on advisory boards of, is a consultant for, and has intellectual property and/or stock options with various industry companies, such as Soliton and Allergan.
SOURCE: Wulkan et. al., ASLMS 2018.
DALLAS – Most physicians do not report adverse events related to noninvasive body contouring devices to the Food and Drug Administration, a database review showed.
“The FDA mandates that manufacturers and device operators disclose medical device reports to monitor suspected injuries and device malfunctions to the Manufacturer and User Facility Device Experience [MAUDE] database,” study author Adam J. Wulkan, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “Given the rapid growth in the noninvasive fat reduction market, it is essential physicians be aware of associated adverse events.”
“Given that this is the largest database for adverse events, I was surprised how few reports were done by physicians,” Dr. Wulkan said in an interview.
He reported results from 83 of the 98 MDRs. A total of 26 MDRs reported on cryolipolysis and included newly diagnosed or exacerbation of prior umbilical hernias (10), neuropathies (5), paradoxical fat hyperplasias (4), blisters (3), rashes (1), flu-like symptoms (1), gastroenteritis (1), and prolapsed bladder/uterus/rectum (1). Of the 11 MDRs for radio-frequency devices, 10 were burns/blisters and 1 fire.
Dr. Wulkan went on to note that 17 MDRs were reported for 1060-nm laser lipolysis, including burns/blisters (5), nodules (4), pain (3), cellulitis/abscesses (2), excessive swelling (1), neuropathy (1), and vomiting (1). There were four cases of burns on MDRs from focused/pulsed ultrasound procedures. One was on a non–FDA approved body site and another after combination radio-frequency/infrared treatment for cellulite reduction.
There were eight MDRs reported with 1440-nm laser cellulite reduction, including burn (5), unacceptable cosmesis (2), and faulty power supply (1). In addition, there were 12 MDRs reported on high-frequency ultrasound, including burns/blisters (9), subcutaneous nodules (2), and excoriation (1). Finally, vacuum-assisted submission was associated with five MDRs, including festooning/unacceptable cosmesis (2), seroma/hematoma (1), laceration (1), and nodules (1).
Dr. Wulkan acknowledged certain limitations of the study, including the fact that not all MDRs are reported to MAUDE, and that the reliability of patient reporting can be questionable. “We have to figure out if there is a natural causation between the adverse event and the treatment itself,” he said at the meeting. “The number of MDRs for any given device must be correlated to the number of procedures performed before we jump to conclusions about the safety of any given device.”
He reported having no financial disclosures. One study coauthor holds positions on advisory boards of, is a consultant for, and has intellectual property and/or stock options with various industry companies, such as Soliton and Allergan.
SOURCE: Wulkan et. al., ASLMS 2018.
REPORTING FROM ASLMS 2018
Key clinical point: Most physicians do not report adverse events related to noninvasive body contouring devices to the FDA .
Major finding: Of 83 medical device reports, 26 were related to cryolipolysis.
Study details: An analysis of 83 complications of noninvasive fat reduction and cellulite reduction devices reported to the FDA.
Disclosures: Dr. Wulkan reported having no financial disclosures. One study coauthor holds positions on advisory boards of, is a consultant for, and has intellectual property and/or stock options with various industry companies, such as Soliton and Allergan.
Source: Wulkan et al. ASLMS 2018.
Laser treatment of port wine stains in infancy found safe, effective
DALLAS – Laser treatment of port wine stains in infancy is both safe and effective, with no incidence of scarring or pigmentary changes, according to results from a single-center analysis.
“Early intervention allows for treatment without general anesthesia, with faster and more complete clearance than what has been reported for treatments begun at older ages,” Hana Jeon, MD, said at the annual conference of the American Society for Laser Medicine and Surgery.
A recent Food and Drug Administration Drug Safety Communication warned that “repeated or lengthy used of general anesthetic and sedation drugs during surgeries or procedures in children younger than 3 years or in pregnant women during their third trimester may affect the development of children’s brains.” Dr. Jeon, a dermatologist in private practice in New York, noted that the FDA warning “places a greater importance on the already controversial topic of when to initiate port wine stain [PWS] treatments in pediatric patients, which requires repeated treatments and are often performed with general anesthesia. Without treatment, these lesions tend to get larger and thicker with time. Starting the treatment during infancy has the potential to limit the use of general anesthesia and to facilitate clearing.”
In what she said is the largest retrospective study of its kind to date, Dr. Jeon and her associates evaluated the success and safety of treating PWSs with a pulsed dye laser at the age of 1 year or younger in the office setting without general anesthesia. The patients received their first PWS treatment at their center during 2000-2017. They reviewed the charts of 197 patients to extract relevant data, including demographic information, age at the time of procedure, and treatment dates. The data cutoff was at 1 year following the initial treatment. Four physicians independently reviewed before and after photos and used the visual analogue scale to grade them.
The pulsed dye laser with dynamic cooling spray was used to minimize patient discomfort. No topical, local, or general anesthesia was used. Patients were immobilized by ancillary staff with parents present in most cases. Ocular shields were placed to allow treatment of periocular lesions.
Of the 197 patients, 63% were female, 90.1% had Fitzpatrick skin types I-III, 8.1% had type IV skin, and the rest had type V-VI skin. Most of the lesions were facial (75.6%), and 41.1% had periocular involvement. The average lesion size was 61 cm2.
The treatment settings for the pulsed dye laser were a 10-12 mm spot size delivered at a fluence of 6.5-9 J/cm2 in a pulse duration of 0.45-1.5 milliseconds. The average age at the time of first treatment was 3.4 months (range, 5-355 days), and the average number of treatments was 9.8 (range, 2-23). Most of the patients (116) were aged 0-3 months at the time of first treatment, followed by 51 aged 3-6 months, 19 aged 6-9 months, and 11 aged 9-12 months.
According to the averaged scores on the visual analogue scale assigned by physicians, 27.4% showed a 100% clearance, 39.1% showed 76%-99% improvement, 15.1% showed 51%-75% improvement, 10.7% showed 26%-50% improvement, and 7.7% showed 0%-25% improvement. No scarring or pigmentary changes were observed.
An analysis of dermatomal distribution revealed that the presence of a V1 lesion was a statistically significant predictor of a higher clearance rate, while the presence of a V3 lesion was a statistically significant predictor of a low clearance rate. “Regardless of where the lesion was, all patients did well,” she said.
Given the small proportion of patients who received their first treatment between the ages of 6 and 12 months of age, Dr. Jeon said that “further studies would be needed to elucidate whether earlier intervention helps to achieve better results.”*
Advantages of early treatment, she said, include the ability to “treat lesions before they get larger and thicker, which also decreases the risk of spontaneous bleeding. Thinner skin allows for better penetration of the laser beam, and we can also avoid using general anesthesia. Psychologically, if we’re able to clear these lesions, it can result in improved quality of life and self-esteem; not just for the patient but for the family as well.”
Dr. Jeon acknowledged certain limitations of the study, including its retrospective design, variable follow-up times, and non-standardization of photographs.
She reported having no financial disclosures.
SOURCE: Jeon H et al. ASLMS 2018.
Correction, 4/18/18: An earlier version of this article misstated the age range of the study participants.
DALLAS – Laser treatment of port wine stains in infancy is both safe and effective, with no incidence of scarring or pigmentary changes, according to results from a single-center analysis.
“Early intervention allows for treatment without general anesthesia, with faster and more complete clearance than what has been reported for treatments begun at older ages,” Hana Jeon, MD, said at the annual conference of the American Society for Laser Medicine and Surgery.
A recent Food and Drug Administration Drug Safety Communication warned that “repeated or lengthy used of general anesthetic and sedation drugs during surgeries or procedures in children younger than 3 years or in pregnant women during their third trimester may affect the development of children’s brains.” Dr. Jeon, a dermatologist in private practice in New York, noted that the FDA warning “places a greater importance on the already controversial topic of when to initiate port wine stain [PWS] treatments in pediatric patients, which requires repeated treatments and are often performed with general anesthesia. Without treatment, these lesions tend to get larger and thicker with time. Starting the treatment during infancy has the potential to limit the use of general anesthesia and to facilitate clearing.”
In what she said is the largest retrospective study of its kind to date, Dr. Jeon and her associates evaluated the success and safety of treating PWSs with a pulsed dye laser at the age of 1 year or younger in the office setting without general anesthesia. The patients received their first PWS treatment at their center during 2000-2017. They reviewed the charts of 197 patients to extract relevant data, including demographic information, age at the time of procedure, and treatment dates. The data cutoff was at 1 year following the initial treatment. Four physicians independently reviewed before and after photos and used the visual analogue scale to grade them.
The pulsed dye laser with dynamic cooling spray was used to minimize patient discomfort. No topical, local, or general anesthesia was used. Patients were immobilized by ancillary staff with parents present in most cases. Ocular shields were placed to allow treatment of periocular lesions.
Of the 197 patients, 63% were female, 90.1% had Fitzpatrick skin types I-III, 8.1% had type IV skin, and the rest had type V-VI skin. Most of the lesions were facial (75.6%), and 41.1% had periocular involvement. The average lesion size was 61 cm2.
The treatment settings for the pulsed dye laser were a 10-12 mm spot size delivered at a fluence of 6.5-9 J/cm2 in a pulse duration of 0.45-1.5 milliseconds. The average age at the time of first treatment was 3.4 months (range, 5-355 days), and the average number of treatments was 9.8 (range, 2-23). Most of the patients (116) were aged 0-3 months at the time of first treatment, followed by 51 aged 3-6 months, 19 aged 6-9 months, and 11 aged 9-12 months.
According to the averaged scores on the visual analogue scale assigned by physicians, 27.4% showed a 100% clearance, 39.1% showed 76%-99% improvement, 15.1% showed 51%-75% improvement, 10.7% showed 26%-50% improvement, and 7.7% showed 0%-25% improvement. No scarring or pigmentary changes were observed.
An analysis of dermatomal distribution revealed that the presence of a V1 lesion was a statistically significant predictor of a higher clearance rate, while the presence of a V3 lesion was a statistically significant predictor of a low clearance rate. “Regardless of where the lesion was, all patients did well,” she said.
Given the small proportion of patients who received their first treatment between the ages of 6 and 12 months of age, Dr. Jeon said that “further studies would be needed to elucidate whether earlier intervention helps to achieve better results.”*
Advantages of early treatment, she said, include the ability to “treat lesions before they get larger and thicker, which also decreases the risk of spontaneous bleeding. Thinner skin allows for better penetration of the laser beam, and we can also avoid using general anesthesia. Psychologically, if we’re able to clear these lesions, it can result in improved quality of life and self-esteem; not just for the patient but for the family as well.”
Dr. Jeon acknowledged certain limitations of the study, including its retrospective design, variable follow-up times, and non-standardization of photographs.
She reported having no financial disclosures.
SOURCE: Jeon H et al. ASLMS 2018.
Correction, 4/18/18: An earlier version of this article misstated the age range of the study participants.
DALLAS – Laser treatment of port wine stains in infancy is both safe and effective, with no incidence of scarring or pigmentary changes, according to results from a single-center analysis.
“Early intervention allows for treatment without general anesthesia, with faster and more complete clearance than what has been reported for treatments begun at older ages,” Hana Jeon, MD, said at the annual conference of the American Society for Laser Medicine and Surgery.
A recent Food and Drug Administration Drug Safety Communication warned that “repeated or lengthy used of general anesthetic and sedation drugs during surgeries or procedures in children younger than 3 years or in pregnant women during their third trimester may affect the development of children’s brains.” Dr. Jeon, a dermatologist in private practice in New York, noted that the FDA warning “places a greater importance on the already controversial topic of when to initiate port wine stain [PWS] treatments in pediatric patients, which requires repeated treatments and are often performed with general anesthesia. Without treatment, these lesions tend to get larger and thicker with time. Starting the treatment during infancy has the potential to limit the use of general anesthesia and to facilitate clearing.”
In what she said is the largest retrospective study of its kind to date, Dr. Jeon and her associates evaluated the success and safety of treating PWSs with a pulsed dye laser at the age of 1 year or younger in the office setting without general anesthesia. The patients received their first PWS treatment at their center during 2000-2017. They reviewed the charts of 197 patients to extract relevant data, including demographic information, age at the time of procedure, and treatment dates. The data cutoff was at 1 year following the initial treatment. Four physicians independently reviewed before and after photos and used the visual analogue scale to grade them.
The pulsed dye laser with dynamic cooling spray was used to minimize patient discomfort. No topical, local, or general anesthesia was used. Patients were immobilized by ancillary staff with parents present in most cases. Ocular shields were placed to allow treatment of periocular lesions.
Of the 197 patients, 63% were female, 90.1% had Fitzpatrick skin types I-III, 8.1% had type IV skin, and the rest had type V-VI skin. Most of the lesions were facial (75.6%), and 41.1% had periocular involvement. The average lesion size was 61 cm2.
The treatment settings for the pulsed dye laser were a 10-12 mm spot size delivered at a fluence of 6.5-9 J/cm2 in a pulse duration of 0.45-1.5 milliseconds. The average age at the time of first treatment was 3.4 months (range, 5-355 days), and the average number of treatments was 9.8 (range, 2-23). Most of the patients (116) were aged 0-3 months at the time of first treatment, followed by 51 aged 3-6 months, 19 aged 6-9 months, and 11 aged 9-12 months.
According to the averaged scores on the visual analogue scale assigned by physicians, 27.4% showed a 100% clearance, 39.1% showed 76%-99% improvement, 15.1% showed 51%-75% improvement, 10.7% showed 26%-50% improvement, and 7.7% showed 0%-25% improvement. No scarring or pigmentary changes were observed.
An analysis of dermatomal distribution revealed that the presence of a V1 lesion was a statistically significant predictor of a higher clearance rate, while the presence of a V3 lesion was a statistically significant predictor of a low clearance rate. “Regardless of where the lesion was, all patients did well,” she said.
Given the small proportion of patients who received their first treatment between the ages of 6 and 12 months of age, Dr. Jeon said that “further studies would be needed to elucidate whether earlier intervention helps to achieve better results.”*
Advantages of early treatment, she said, include the ability to “treat lesions before they get larger and thicker, which also decreases the risk of spontaneous bleeding. Thinner skin allows for better penetration of the laser beam, and we can also avoid using general anesthesia. Psychologically, if we’re able to clear these lesions, it can result in improved quality of life and self-esteem; not just for the patient but for the family as well.”
Dr. Jeon acknowledged certain limitations of the study, including its retrospective design, variable follow-up times, and non-standardization of photographs.
She reported having no financial disclosures.
SOURCE: Jeon H et al. ASLMS 2018.
Correction, 4/18/18: An earlier version of this article misstated the age range of the study participants.
REPORTING FROM ASLMS 2018
Key clinical point:
Major finding: On the visual analogue scale, 27.4% of infants achieved complete clearance of their port wine lesion after treatment.
Study details: An analysis of 197 infants aged 1 year or younger with port wine stains who were treated with a pulsed dye laser during 2000-2017.
Disclosures: Dr. Jeon reported having no financial disclosures.
Source: Jeon H et al. ASLMS 2018.