User login
Damian McNamara is a journalist for Medscape Medical News and MDedge. He worked full-time for MDedge as the Miami Bureau covering a dozen medical specialties during 2001-2012, then as a freelancer for Medscape and MDedge, before being hired on staff by Medscape in 2018. Now the two companies are one. He uses what he learned in school – Damian has a BS in chemistry and an MS in science, health and environmental reporting/journalism. He works out of a home office in Miami, with a 100-pound chocolate lab known to snore under his desk during work hours.
Pediatric Airway Clearance Strategies Lack Data
FORT LAUDERDALE, FLA. – Despite a lack of definitive evidence, airway clearance strategies are commonly employed in children with cystic fibrosis and other lung diseases, Dr. Veda L. Ackerman said.
"Unfortunately, we don’t really know very much about airway clearance. We do it a lot, but we don’t have a lot of data," Dr. Ackerman said at a seminar on pediatric pulmonology, which was sponsored by the American College of Chest Physicians and the American Academy of Pediatrics.
For instance, the only pediatric study comparing airway clearance to no such therapy was published in 1983 and assessed eight patients with cystic fibrosis (J. Pediatr. 2003;103:538-42).
"Intuitively and intellectually, airway clearance makes a lot of sense. But [it is important to] think about whether we are helping or hurting when we prescribe airway clearance," said Dr. Ackerman, a pediatric intensivist at Riley Hospital for Children and a pediatric pulmonologist in private practice in Indianapolis.
Oxygen desaturation, gastroesophageal reflux, aspiration, hyperventilation, and "guilt for the family from lack of adherence" are potential adverse events associated with prescription of airway clearance, Dr. Ackerman said.
There are "no data to support the use of one airway clearance technique over the other," Dr. Ackerman said. Chest physiotherapy (CPT), positive expiratory pressure (PEP) valve, Cardinal Health’s Flutter device, Smiths Medical’s Acapella system, Medical Acoustics’ Lung Flute device, airway clearance vests, and Smiths Medical’s EzPAP device are among the options.
No definitive data exist to support use of CPT in an asymptomatic child with cystic fibrosis, Dr. Ackerman said. This patient population is prone to adverse events, especially gastroesophageal reflux with or without aspiration. CPT also requires a significant time commitment on the part of families. Despite these concerns, "I still do recommend it" for some patients.
Airway clearance devices "jiggle, shake, or use sound waves to loosen mucus off the airway walls so secretions can be coughed up," Dr. Ackerman said. Success with these devices is often technique dependent.
The PEP valve is portable, takes 10-15 minutes to clear the airway, and can be used with aerosolized medications. However, Dr. Ackerman’s institution uses the Flutter "much more than the PEP valve," she said. This device "tends to be used for families who cannot put time into CPT or when the child goes to Grandma’s or on a sleepover." The device loosens mucus through expiratory oscillation, so it may be less effective at lower airflows, such as those used for small children or patients with more severe lung disease. The device has to be held at a precise angle to maximize oscillation, she added. Each use of the Flutter device takes about 10-15 minutes.
The Acapella system combines the benefits of the PEP valve and airway vibrations to mobilize secretions, Dr. Ackerman said. The mechanism of action is similar to that of the Flutter, except that the Acapella has a valve-magnet device to interrupt expiratory flow and thus can be used at any angle.
"All of these devices cost less than $100," Dr. Ackerman said. "These may – and I said may – be better than doing nothing at all."
Contraindications to the PEP valve, Flutter, and Acapella include pneumothorax, hemoptysis, and esophageal varices. Lung surgery is another contraindication, Dr. Ackerman said, because use of airway-clearance devices can cause an air leak or can break down an anastomosis site. A pulmonary embolus is another contraindication, but "fortunately we do not see this often in pediatrics." A perforated ear drum is also a contraindication to these airway devices "because it causes pain."
The Lung Flute uses a different strategy (acoustic waves) to increase mucociliary clearance. It vibrates the chest in a way that is similar to the way a reed instrument vibrates when it’s played, Dr. Ackerman said. "There are no pediatric data, but it is cheap and easy to use." The Lung Flute is used more commonly for patients with chronic obstructive pulmonary disorder and not as much in cystic fibrosis.
Airway clearance vests deliver pulses of air pressure to the chest wall. The vest loosens mucus through shearing at the air/mucus interface, and compression causes clearance through repetitive peak expiratory flows that expel mucus like small coughs.
"You should not get compression of the airway itself; only the chest wall is compressed," Dr. Ackerman said. In contrast, "if you blow hard enough with the Acapella, Flutter, or PEP, you could get airway collapse." An airway clearance vest costs approximately $10,000, and obtaining insurance approval can be difficult; reimbursement policies vary from state to state.
The EzPAP device clears airways through positive airway pressure in a way that is similar to intermittent positive pressure breathing. It is approved by the Food and Drug Administration for lung expansion therapy and the prevention and treatment of atelectasis. Although no peer-reviewed data are available, many children are using EzPAP because respiratory therapists believe in this device, Dr. Ackerman said.
For more information, Dr. Ackerman recommended the American College of Chest Physicians’ Evidence-Based Guidelines for Nonpharmacologic Airway Clearance Therapies (Chest 2006;129:250S-9S), which were published in 2006 but are still applicable in 2011, she added.
Dr. Ackerman said that she had no relevant financial disclosures.
FORT LAUDERDALE, FLA. – Despite a lack of definitive evidence, airway clearance strategies are commonly employed in children with cystic fibrosis and other lung diseases, Dr. Veda L. Ackerman said.
"Unfortunately, we don’t really know very much about airway clearance. We do it a lot, but we don’t have a lot of data," Dr. Ackerman said at a seminar on pediatric pulmonology, which was sponsored by the American College of Chest Physicians and the American Academy of Pediatrics.
For instance, the only pediatric study comparing airway clearance to no such therapy was published in 1983 and assessed eight patients with cystic fibrosis (J. Pediatr. 2003;103:538-42).
"Intuitively and intellectually, airway clearance makes a lot of sense. But [it is important to] think about whether we are helping or hurting when we prescribe airway clearance," said Dr. Ackerman, a pediatric intensivist at Riley Hospital for Children and a pediatric pulmonologist in private practice in Indianapolis.
Oxygen desaturation, gastroesophageal reflux, aspiration, hyperventilation, and "guilt for the family from lack of adherence" are potential adverse events associated with prescription of airway clearance, Dr. Ackerman said.
There are "no data to support the use of one airway clearance technique over the other," Dr. Ackerman said. Chest physiotherapy (CPT), positive expiratory pressure (PEP) valve, Cardinal Health’s Flutter device, Smiths Medical’s Acapella system, Medical Acoustics’ Lung Flute device, airway clearance vests, and Smiths Medical’s EzPAP device are among the options.
No definitive data exist to support use of CPT in an asymptomatic child with cystic fibrosis, Dr. Ackerman said. This patient population is prone to adverse events, especially gastroesophageal reflux with or without aspiration. CPT also requires a significant time commitment on the part of families. Despite these concerns, "I still do recommend it" for some patients.
Airway clearance devices "jiggle, shake, or use sound waves to loosen mucus off the airway walls so secretions can be coughed up," Dr. Ackerman said. Success with these devices is often technique dependent.
The PEP valve is portable, takes 10-15 minutes to clear the airway, and can be used with aerosolized medications. However, Dr. Ackerman’s institution uses the Flutter "much more than the PEP valve," she said. This device "tends to be used for families who cannot put time into CPT or when the child goes to Grandma’s or on a sleepover." The device loosens mucus through expiratory oscillation, so it may be less effective at lower airflows, such as those used for small children or patients with more severe lung disease. The device has to be held at a precise angle to maximize oscillation, she added. Each use of the Flutter device takes about 10-15 minutes.
The Acapella system combines the benefits of the PEP valve and airway vibrations to mobilize secretions, Dr. Ackerman said. The mechanism of action is similar to that of the Flutter, except that the Acapella has a valve-magnet device to interrupt expiratory flow and thus can be used at any angle.
"All of these devices cost less than $100," Dr. Ackerman said. "These may – and I said may – be better than doing nothing at all."
Contraindications to the PEP valve, Flutter, and Acapella include pneumothorax, hemoptysis, and esophageal varices. Lung surgery is another contraindication, Dr. Ackerman said, because use of airway-clearance devices can cause an air leak or can break down an anastomosis site. A pulmonary embolus is another contraindication, but "fortunately we do not see this often in pediatrics." A perforated ear drum is also a contraindication to these airway devices "because it causes pain."
The Lung Flute uses a different strategy (acoustic waves) to increase mucociliary clearance. It vibrates the chest in a way that is similar to the way a reed instrument vibrates when it’s played, Dr. Ackerman said. "There are no pediatric data, but it is cheap and easy to use." The Lung Flute is used more commonly for patients with chronic obstructive pulmonary disorder and not as much in cystic fibrosis.
Airway clearance vests deliver pulses of air pressure to the chest wall. The vest loosens mucus through shearing at the air/mucus interface, and compression causes clearance through repetitive peak expiratory flows that expel mucus like small coughs.
"You should not get compression of the airway itself; only the chest wall is compressed," Dr. Ackerman said. In contrast, "if you blow hard enough with the Acapella, Flutter, or PEP, you could get airway collapse." An airway clearance vest costs approximately $10,000, and obtaining insurance approval can be difficult; reimbursement policies vary from state to state.
The EzPAP device clears airways through positive airway pressure in a way that is similar to intermittent positive pressure breathing. It is approved by the Food and Drug Administration for lung expansion therapy and the prevention and treatment of atelectasis. Although no peer-reviewed data are available, many children are using EzPAP because respiratory therapists believe in this device, Dr. Ackerman said.
For more information, Dr. Ackerman recommended the American College of Chest Physicians’ Evidence-Based Guidelines for Nonpharmacologic Airway Clearance Therapies (Chest 2006;129:250S-9S), which were published in 2006 but are still applicable in 2011, she added.
Dr. Ackerman said that she had no relevant financial disclosures.
FORT LAUDERDALE, FLA. – Despite a lack of definitive evidence, airway clearance strategies are commonly employed in children with cystic fibrosis and other lung diseases, Dr. Veda L. Ackerman said.
"Unfortunately, we don’t really know very much about airway clearance. We do it a lot, but we don’t have a lot of data," Dr. Ackerman said at a seminar on pediatric pulmonology, which was sponsored by the American College of Chest Physicians and the American Academy of Pediatrics.
For instance, the only pediatric study comparing airway clearance to no such therapy was published in 1983 and assessed eight patients with cystic fibrosis (J. Pediatr. 2003;103:538-42).
"Intuitively and intellectually, airway clearance makes a lot of sense. But [it is important to] think about whether we are helping or hurting when we prescribe airway clearance," said Dr. Ackerman, a pediatric intensivist at Riley Hospital for Children and a pediatric pulmonologist in private practice in Indianapolis.
Oxygen desaturation, gastroesophageal reflux, aspiration, hyperventilation, and "guilt for the family from lack of adherence" are potential adverse events associated with prescription of airway clearance, Dr. Ackerman said.
There are "no data to support the use of one airway clearance technique over the other," Dr. Ackerman said. Chest physiotherapy (CPT), positive expiratory pressure (PEP) valve, Cardinal Health’s Flutter device, Smiths Medical’s Acapella system, Medical Acoustics’ Lung Flute device, airway clearance vests, and Smiths Medical’s EzPAP device are among the options.
No definitive data exist to support use of CPT in an asymptomatic child with cystic fibrosis, Dr. Ackerman said. This patient population is prone to adverse events, especially gastroesophageal reflux with or without aspiration. CPT also requires a significant time commitment on the part of families. Despite these concerns, "I still do recommend it" for some patients.
Airway clearance devices "jiggle, shake, or use sound waves to loosen mucus off the airway walls so secretions can be coughed up," Dr. Ackerman said. Success with these devices is often technique dependent.
The PEP valve is portable, takes 10-15 minutes to clear the airway, and can be used with aerosolized medications. However, Dr. Ackerman’s institution uses the Flutter "much more than the PEP valve," she said. This device "tends to be used for families who cannot put time into CPT or when the child goes to Grandma’s or on a sleepover." The device loosens mucus through expiratory oscillation, so it may be less effective at lower airflows, such as those used for small children or patients with more severe lung disease. The device has to be held at a precise angle to maximize oscillation, she added. Each use of the Flutter device takes about 10-15 minutes.
The Acapella system combines the benefits of the PEP valve and airway vibrations to mobilize secretions, Dr. Ackerman said. The mechanism of action is similar to that of the Flutter, except that the Acapella has a valve-magnet device to interrupt expiratory flow and thus can be used at any angle.
"All of these devices cost less than $100," Dr. Ackerman said. "These may – and I said may – be better than doing nothing at all."
Contraindications to the PEP valve, Flutter, and Acapella include pneumothorax, hemoptysis, and esophageal varices. Lung surgery is another contraindication, Dr. Ackerman said, because use of airway-clearance devices can cause an air leak or can break down an anastomosis site. A pulmonary embolus is another contraindication, but "fortunately we do not see this often in pediatrics." A perforated ear drum is also a contraindication to these airway devices "because it causes pain."
The Lung Flute uses a different strategy (acoustic waves) to increase mucociliary clearance. It vibrates the chest in a way that is similar to the way a reed instrument vibrates when it’s played, Dr. Ackerman said. "There are no pediatric data, but it is cheap and easy to use." The Lung Flute is used more commonly for patients with chronic obstructive pulmonary disorder and not as much in cystic fibrosis.
Airway clearance vests deliver pulses of air pressure to the chest wall. The vest loosens mucus through shearing at the air/mucus interface, and compression causes clearance through repetitive peak expiratory flows that expel mucus like small coughs.
"You should not get compression of the airway itself; only the chest wall is compressed," Dr. Ackerman said. In contrast, "if you blow hard enough with the Acapella, Flutter, or PEP, you could get airway collapse." An airway clearance vest costs approximately $10,000, and obtaining insurance approval can be difficult; reimbursement policies vary from state to state.
The EzPAP device clears airways through positive airway pressure in a way that is similar to intermittent positive pressure breathing. It is approved by the Food and Drug Administration for lung expansion therapy and the prevention and treatment of atelectasis. Although no peer-reviewed data are available, many children are using EzPAP because respiratory therapists believe in this device, Dr. Ackerman said.
For more information, Dr. Ackerman recommended the American College of Chest Physicians’ Evidence-Based Guidelines for Nonpharmacologic Airway Clearance Therapies (Chest 2006;129:250S-9S), which were published in 2006 but are still applicable in 2011, she added.
Dr. Ackerman said that she had no relevant financial disclosures.
Pulmonology Patients Can Travel by Air, but Restrictions Apply
FORT LAUDERDALE, FLA. – Your patients with cystic fibrosis or other pulmonary conditions may ask you if and when it’s safe for them to fly on an airplane.
How you respond can depend in part on their travel history, how long they will be exposed to increased cabin pressure, and if they are immunocompromised or have other risk factors for infection that are related to airborne pathogens, Dr. Susan L. Millard said.
Severe respiratory insufficiency, right heart failure or hemodynamic instability, and active pneumothorax are absolute contraindications to air travel, according to 30 experts who wrote a consensus statement for traveling with cystic fibrosis (J. Cyst. Fibros. 2010;9:385-99).
These first-ever European recommendations are useful because they address preparations for travel (for example, vaccinations and packing medication), important considerations during travel, and issues specific to the immunocompromised, Dr. Millard said at a pediatric pulmonology seminar, which was sponsored by the American College of Chest Physicians and the American Academy of Pediatrics.
Air travel for pulmonology patients can be difficult, Dr. Millard noted, because "the environment is very dangerous." The cabin is pressurized, alveolar partial pressure falls with increasing altitude, and the partial pressure of oxygen is inversely proportional to altitude. Because the Joint Aviation Authorities stipulated that mean cabin pressure match an altitude of 8,000 feet, "this means they want us to all have an oxygen saturation of about 90%."
Supplemental oxygen during air travel can help patients, but identification of appropriate candidates varies. Guidelines from the American Thoracic Society and British Thoracic Society (Thorax 2002;57:289-304)recommend that patients with chronic lung disease be able to maintain an arterial oxygen tension greater than 50 mm Hg or 6.6 kilopascals (kPa), Dr. Millard said. However, because they tend to be younger than COPD (chronic obstructive pulmonary disease) patients and generally have no increased cardiovascular risk, use of such a cutoff value could be an oversimplification for patients with cystic fibrosis, said Dr. Millard, a pediatric pulmonologist at Helen DeVos Children’s Hospital in Grand Rapids, Mich.
Hypoxia during flight is a major concern. Consider whether your patient will be able to sustain hyperventilation that is spurred by hypoxia while on the airplane. Significant bronchospasm, for example, could impede prolonged hyperventilation, Dr. Millard said.
Consider a hypoxia inhalation test in advance of travel. This test requires that patients breathe a hypoxic mixture of 15% oxygen with nitrogen for 20 minutes to predict their reaction to hypoxia at 8,000 feet. Supplemental oxygen is recommended if their arterial oxygen tension drops below 50-55 mm Hg or 6.6-7.4 kPa.
"The hypoxia inhalation test is found to be safe," Dr. Millard said. Applicability outside the clinic setting is a concern, however: "The problem is, they are sitting. This may not fully represent the physical stress and environmental variability of air travel," including Transportation Security Administration screening and walking long distances.
For this reason, some experts advise also screening patients with a walk test prior to their trip, Dr. Millard said. The American Thoracic Society provides guidelines for conducting a functional exercise evaluation called a 6-minute walk test, for example (Am. J. Resp. Crit. Care Med. 2002;166:111-7).
Patients who require supplemental oxygen are permitted to use their own approved portable oxygen concentrator (POC) on all airlines that operate in the United States. POCs weigh 8-10 pounds and batteries last an average of about 4 hours, Dr. Millard said. Also, some POCs are pulse generated, meaning the patient must be able to inspire strongly enough to get oxygen.
Advise your patients or their families to check in advance if their airline requires approval from a physician for POC use, Dr. Millard said. "I had a patient who gave me 48 hours notice that they were going to fly. I had to fill out a form ahead of time for the airline."
Pulmonology patients also may request a travel letter, "which is especially important if they are going through customs," Dr. Millard said. Include their insurance information, your contact information, the telephone number for the clinic, and a list of medications (and approximate quantities required).
Airborne infection risk is another major concern. Most commercial aircraft recirculate 50% of the air delivered to the passenger cabin, Dr. Millard said. Ideally, the aircraft features HEPA (high-efficiency particulate air) filters, although the U.S. Federal Aviation Authority (FAA) and the U.K. Civil Aviation Authority do not mandate this level of filtration.
Dr. Millard cited a study that supports transmission of H1N1 influenza during flight (Epidemiol. Health 2010;32:e2010006). Officials at the Korea Centers for Disease Control and Prevention determined that an infected woman who flew from Los Angeles to Seoul in 2009 infected other passengers. The study includes a seating map of the Boeing 747 that shows where she and other passengers who got sick were seated.
"People are trying to figure this out to make [air travel] safer," Dr. Millard said. For example, one set of researchers assessed the ability of commercially available biosensors to detect airborne pathogens on airplanes (PLoS One 2011;6:e14520). With the current technology, however, only steady-state bacteria concentrations were detected in cases in which at least seven infected passengers either coughed 20 times per hour or sneezed 4 times an hour. And no sensor in the study detected airborne viruses well. Sensors with improved sensitivity and/or the screening of individual patients for respiratory illnesses prior to boarding might reduce the infection risk, Dr. Millard said.
For a list of POC devices that have been approved by the FAA, visit www.faa.gov/about/initiatives/cabin_safety/portable_oxygen/.
For additional guidance from the TSA on traveling with supplemental oxygen or other medical devices, you can refer patients to www.tsa.gov/travelers/airtravel/specialneeds/editorial_1374.shtm.
Dr. Millard said that she had no relevant disclosures.
FORT LAUDERDALE, FLA. – Your patients with cystic fibrosis or other pulmonary conditions may ask you if and when it’s safe for them to fly on an airplane.
How you respond can depend in part on their travel history, how long they will be exposed to increased cabin pressure, and if they are immunocompromised or have other risk factors for infection that are related to airborne pathogens, Dr. Susan L. Millard said.
Severe respiratory insufficiency, right heart failure or hemodynamic instability, and active pneumothorax are absolute contraindications to air travel, according to 30 experts who wrote a consensus statement for traveling with cystic fibrosis (J. Cyst. Fibros. 2010;9:385-99).
These first-ever European recommendations are useful because they address preparations for travel (for example, vaccinations and packing medication), important considerations during travel, and issues specific to the immunocompromised, Dr. Millard said at a pediatric pulmonology seminar, which was sponsored by the American College of Chest Physicians and the American Academy of Pediatrics.
Air travel for pulmonology patients can be difficult, Dr. Millard noted, because "the environment is very dangerous." The cabin is pressurized, alveolar partial pressure falls with increasing altitude, and the partial pressure of oxygen is inversely proportional to altitude. Because the Joint Aviation Authorities stipulated that mean cabin pressure match an altitude of 8,000 feet, "this means they want us to all have an oxygen saturation of about 90%."
Supplemental oxygen during air travel can help patients, but identification of appropriate candidates varies. Guidelines from the American Thoracic Society and British Thoracic Society (Thorax 2002;57:289-304)recommend that patients with chronic lung disease be able to maintain an arterial oxygen tension greater than 50 mm Hg or 6.6 kilopascals (kPa), Dr. Millard said. However, because they tend to be younger than COPD (chronic obstructive pulmonary disease) patients and generally have no increased cardiovascular risk, use of such a cutoff value could be an oversimplification for patients with cystic fibrosis, said Dr. Millard, a pediatric pulmonologist at Helen DeVos Children’s Hospital in Grand Rapids, Mich.
Hypoxia during flight is a major concern. Consider whether your patient will be able to sustain hyperventilation that is spurred by hypoxia while on the airplane. Significant bronchospasm, for example, could impede prolonged hyperventilation, Dr. Millard said.
Consider a hypoxia inhalation test in advance of travel. This test requires that patients breathe a hypoxic mixture of 15% oxygen with nitrogen for 20 minutes to predict their reaction to hypoxia at 8,000 feet. Supplemental oxygen is recommended if their arterial oxygen tension drops below 50-55 mm Hg or 6.6-7.4 kPa.
"The hypoxia inhalation test is found to be safe," Dr. Millard said. Applicability outside the clinic setting is a concern, however: "The problem is, they are sitting. This may not fully represent the physical stress and environmental variability of air travel," including Transportation Security Administration screening and walking long distances.
For this reason, some experts advise also screening patients with a walk test prior to their trip, Dr. Millard said. The American Thoracic Society provides guidelines for conducting a functional exercise evaluation called a 6-minute walk test, for example (Am. J. Resp. Crit. Care Med. 2002;166:111-7).
Patients who require supplemental oxygen are permitted to use their own approved portable oxygen concentrator (POC) on all airlines that operate in the United States. POCs weigh 8-10 pounds and batteries last an average of about 4 hours, Dr. Millard said. Also, some POCs are pulse generated, meaning the patient must be able to inspire strongly enough to get oxygen.
Advise your patients or their families to check in advance if their airline requires approval from a physician for POC use, Dr. Millard said. "I had a patient who gave me 48 hours notice that they were going to fly. I had to fill out a form ahead of time for the airline."
Pulmonology patients also may request a travel letter, "which is especially important if they are going through customs," Dr. Millard said. Include their insurance information, your contact information, the telephone number for the clinic, and a list of medications (and approximate quantities required).
Airborne infection risk is another major concern. Most commercial aircraft recirculate 50% of the air delivered to the passenger cabin, Dr. Millard said. Ideally, the aircraft features HEPA (high-efficiency particulate air) filters, although the U.S. Federal Aviation Authority (FAA) and the U.K. Civil Aviation Authority do not mandate this level of filtration.
Dr. Millard cited a study that supports transmission of H1N1 influenza during flight (Epidemiol. Health 2010;32:e2010006). Officials at the Korea Centers for Disease Control and Prevention determined that an infected woman who flew from Los Angeles to Seoul in 2009 infected other passengers. The study includes a seating map of the Boeing 747 that shows where she and other passengers who got sick were seated.
"People are trying to figure this out to make [air travel] safer," Dr. Millard said. For example, one set of researchers assessed the ability of commercially available biosensors to detect airborne pathogens on airplanes (PLoS One 2011;6:e14520). With the current technology, however, only steady-state bacteria concentrations were detected in cases in which at least seven infected passengers either coughed 20 times per hour or sneezed 4 times an hour. And no sensor in the study detected airborne viruses well. Sensors with improved sensitivity and/or the screening of individual patients for respiratory illnesses prior to boarding might reduce the infection risk, Dr. Millard said.
For a list of POC devices that have been approved by the FAA, visit www.faa.gov/about/initiatives/cabin_safety/portable_oxygen/.
For additional guidance from the TSA on traveling with supplemental oxygen or other medical devices, you can refer patients to www.tsa.gov/travelers/airtravel/specialneeds/editorial_1374.shtm.
Dr. Millard said that she had no relevant disclosures.
FORT LAUDERDALE, FLA. – Your patients with cystic fibrosis or other pulmonary conditions may ask you if and when it’s safe for them to fly on an airplane.
How you respond can depend in part on their travel history, how long they will be exposed to increased cabin pressure, and if they are immunocompromised or have other risk factors for infection that are related to airborne pathogens, Dr. Susan L. Millard said.
Severe respiratory insufficiency, right heart failure or hemodynamic instability, and active pneumothorax are absolute contraindications to air travel, according to 30 experts who wrote a consensus statement for traveling with cystic fibrosis (J. Cyst. Fibros. 2010;9:385-99).
These first-ever European recommendations are useful because they address preparations for travel (for example, vaccinations and packing medication), important considerations during travel, and issues specific to the immunocompromised, Dr. Millard said at a pediatric pulmonology seminar, which was sponsored by the American College of Chest Physicians and the American Academy of Pediatrics.
Air travel for pulmonology patients can be difficult, Dr. Millard noted, because "the environment is very dangerous." The cabin is pressurized, alveolar partial pressure falls with increasing altitude, and the partial pressure of oxygen is inversely proportional to altitude. Because the Joint Aviation Authorities stipulated that mean cabin pressure match an altitude of 8,000 feet, "this means they want us to all have an oxygen saturation of about 90%."
Supplemental oxygen during air travel can help patients, but identification of appropriate candidates varies. Guidelines from the American Thoracic Society and British Thoracic Society (Thorax 2002;57:289-304)recommend that patients with chronic lung disease be able to maintain an arterial oxygen tension greater than 50 mm Hg or 6.6 kilopascals (kPa), Dr. Millard said. However, because they tend to be younger than COPD (chronic obstructive pulmonary disease) patients and generally have no increased cardiovascular risk, use of such a cutoff value could be an oversimplification for patients with cystic fibrosis, said Dr. Millard, a pediatric pulmonologist at Helen DeVos Children’s Hospital in Grand Rapids, Mich.
Hypoxia during flight is a major concern. Consider whether your patient will be able to sustain hyperventilation that is spurred by hypoxia while on the airplane. Significant bronchospasm, for example, could impede prolonged hyperventilation, Dr. Millard said.
Consider a hypoxia inhalation test in advance of travel. This test requires that patients breathe a hypoxic mixture of 15% oxygen with nitrogen for 20 minutes to predict their reaction to hypoxia at 8,000 feet. Supplemental oxygen is recommended if their arterial oxygen tension drops below 50-55 mm Hg or 6.6-7.4 kPa.
"The hypoxia inhalation test is found to be safe," Dr. Millard said. Applicability outside the clinic setting is a concern, however: "The problem is, they are sitting. This may not fully represent the physical stress and environmental variability of air travel," including Transportation Security Administration screening and walking long distances.
For this reason, some experts advise also screening patients with a walk test prior to their trip, Dr. Millard said. The American Thoracic Society provides guidelines for conducting a functional exercise evaluation called a 6-minute walk test, for example (Am. J. Resp. Crit. Care Med. 2002;166:111-7).
Patients who require supplemental oxygen are permitted to use their own approved portable oxygen concentrator (POC) on all airlines that operate in the United States. POCs weigh 8-10 pounds and batteries last an average of about 4 hours, Dr. Millard said. Also, some POCs are pulse generated, meaning the patient must be able to inspire strongly enough to get oxygen.
Advise your patients or their families to check in advance if their airline requires approval from a physician for POC use, Dr. Millard said. "I had a patient who gave me 48 hours notice that they were going to fly. I had to fill out a form ahead of time for the airline."
Pulmonology patients also may request a travel letter, "which is especially important if they are going through customs," Dr. Millard said. Include their insurance information, your contact information, the telephone number for the clinic, and a list of medications (and approximate quantities required).
Airborne infection risk is another major concern. Most commercial aircraft recirculate 50% of the air delivered to the passenger cabin, Dr. Millard said. Ideally, the aircraft features HEPA (high-efficiency particulate air) filters, although the U.S. Federal Aviation Authority (FAA) and the U.K. Civil Aviation Authority do not mandate this level of filtration.
Dr. Millard cited a study that supports transmission of H1N1 influenza during flight (Epidemiol. Health 2010;32:e2010006). Officials at the Korea Centers for Disease Control and Prevention determined that an infected woman who flew from Los Angeles to Seoul in 2009 infected other passengers. The study includes a seating map of the Boeing 747 that shows where she and other passengers who got sick were seated.
"People are trying to figure this out to make [air travel] safer," Dr. Millard said. For example, one set of researchers assessed the ability of commercially available biosensors to detect airborne pathogens on airplanes (PLoS One 2011;6:e14520). With the current technology, however, only steady-state bacteria concentrations were detected in cases in which at least seven infected passengers either coughed 20 times per hour or sneezed 4 times an hour. And no sensor in the study detected airborne viruses well. Sensors with improved sensitivity and/or the screening of individual patients for respiratory illnesses prior to boarding might reduce the infection risk, Dr. Millard said.
For a list of POC devices that have been approved by the FAA, visit www.faa.gov/about/initiatives/cabin_safety/portable_oxygen/.
For additional guidance from the TSA on traveling with supplemental oxygen or other medical devices, you can refer patients to www.tsa.gov/travelers/airtravel/specialneeds/editorial_1374.shtm.
Dr. Millard said that she had no relevant disclosures.
Pulmonology Patients Can Travel by Air, but Restrictions Apply
FORT LAUDERDALE, FLA. – Your patients with cystic fibrosis or other pulmonary conditions may ask you if and when it’s safe for them to fly on an airplane.
How you respond can depend in part on their travel history, how long they will be exposed to increased cabin pressure, and if they are immunocompromised or have other risk factors for infection that are related to airborne pathogens, Dr. Susan L. Millard said.
Severe respiratory insufficiency, right heart failure or hemodynamic instability, and active pneumothorax are absolute contraindications to air travel, according to 30 experts who wrote a consensus statement for traveling with cystic fibrosis (J. Cyst. Fibros. 2010;9:385-99).
These first-ever European recommendations are useful because they address preparations for travel (for example, vaccinations and packing medication), important considerations during travel, and issues specific to the immunocompromised, Dr. Millard said at a pediatric pulmonology seminar, which was sponsored by the American College of Chest Physicians and the American Academy of Pediatrics.
Air travel for pulmonology patients can be difficult, Dr. Millard noted, because "the environment is very dangerous." The cabin is pressurized, alveolar partial pressure falls with increasing altitude, and the partial pressure of oxygen is inversely proportional to altitude. Because the Joint Aviation Authorities stipulated that mean cabin pressure match an altitude of 8,000 feet, "this means they want us to all have an oxygen saturation of about 90%."
Supplemental oxygen during air travel can help patients, but identification of appropriate candidates varies. Guidelines from the American Thoracic Society and British Thoracic Society (Thorax 2002;57:289-304)recommend that patients with chronic lung disease be able to maintain an arterial oxygen tension greater than 50 mm Hg or 6.6 kilopascals (kPa), Dr. Millard said. However, because they tend to be younger than COPD (chronic obstructive pulmonary disease) patients and generally have no increased cardiovascular risk, use of such a cutoff value could be an oversimplification for patients with cystic fibrosis, said Dr. Millard, a pediatric pulmonologist at Helen DeVos Children’s Hospital in Grand Rapids, Mich.
Hypoxia during flight is a major concern. Consider whether your patient will be able to sustain hyperventilation that is spurred by hypoxia while on the airplane. Significant bronchospasm, for example, could impede prolonged hyperventilation, Dr. Millard said.
Consider a hypoxia inhalation test in advance of travel. This test requires that patients breathe a hypoxic mixture of 15% oxygen with nitrogen for 20 minutes to predict their reaction to hypoxia at 8,000 feet. Supplemental oxygen is recommended if their arterial oxygen tension drops below 50-55 mm Hg or 6.6-7.4 kPa.
"The hypoxia inhalation test is found to be safe," Dr. Millard said. Applicability outside the clinic setting is a concern, however: "The problem is, they are sitting. This may not fully represent the physical stress and environmental variability of air travel," including Transportation Security Administration screening and walking long distances.
For this reason, some experts advise also screening patients with a walk test prior to their trip, Dr. Millard said. The American Thoracic Society provides guidelines for conducting a functional exercise evaluation called a 6-minute walk test, for example (Am. J. Resp. Crit. Care Med. 2002;166:111-7).
Patients who require supplemental oxygen are permitted to use their own approved portable oxygen concentrator (POC) on all airlines that operate in the United States. POCs weigh 8-10 pounds and batteries last an average of about 4 hours, Dr. Millard said. Also, some POCs are pulse generated, meaning the patient must be able to inspire strongly enough to get oxygen.
Advise your patients or their families to check in advance if their airline requires approval from a physician for POC use, Dr. Millard said. "I had a patient who gave me 48 hours notice that they were going to fly. I had to fill out a form ahead of time for the airline."
Pulmonology patients also may request a travel letter, "which is especially important if they are going through customs," Dr. Millard said. Include their insurance information, your contact information, the telephone number for the clinic, and a list of medications (and approximate quantities required).
Airborne infection risk is another major concern. Most commercial aircraft recirculate 50% of the air delivered to the passenger cabin, Dr. Millard said. Ideally, the aircraft features HEPA (high-efficiency particulate air) filters, although the U.S. Federal Aviation Authority (FAA) and the U.K. Civil Aviation Authority do not mandate this level of filtration.
Dr. Millard cited a study that supports transmission of H1N1 influenza during flight (Epidemiol. Health 2010;32:e2010006). Officials at the Korea Centers for Disease Control and Prevention determined that an infected woman who flew from Los Angeles to Seoul in 2009 infected other passengers. The study includes a seating map of the Boeing 747 that shows where she and other passengers who got sick were seated.
"People are trying to figure this out to make [air travel] safer," Dr. Millard said. For example, one set of researchers assessed the ability of commercially available biosensors to detect airborne pathogens on airplanes (PLoS One 2011;6:e14520). With the current technology, however, only steady-state bacteria concentrations were detected in cases in which at least seven infected passengers either coughed 20 times per hour or sneezed 4 times an hour. And no sensor in the study detected airborne viruses well. Sensors with improved sensitivity and/or the screening of individual patients for respiratory illnesses prior to boarding might reduce the infection risk, Dr. Millard said.
For a list of POC devices that have been approved by the FAA, visit www.faa.gov/about/initiatives/cabin_safety/portable_oxygen/.
For additional guidance from the TSA on traveling with supplemental oxygen or other medical devices, you can refer patients to www.tsa.gov/travelers/airtravel/specialneeds/editorial_1374.shtm.
Dr. Millard said that she had no relevant disclosures.
FORT LAUDERDALE, FLA. – Your patients with cystic fibrosis or other pulmonary conditions may ask you if and when it’s safe for them to fly on an airplane.
How you respond can depend in part on their travel history, how long they will be exposed to increased cabin pressure, and if they are immunocompromised or have other risk factors for infection that are related to airborne pathogens, Dr. Susan L. Millard said.
Severe respiratory insufficiency, right heart failure or hemodynamic instability, and active pneumothorax are absolute contraindications to air travel, according to 30 experts who wrote a consensus statement for traveling with cystic fibrosis (J. Cyst. Fibros. 2010;9:385-99).
These first-ever European recommendations are useful because they address preparations for travel (for example, vaccinations and packing medication), important considerations during travel, and issues specific to the immunocompromised, Dr. Millard said at a pediatric pulmonology seminar, which was sponsored by the American College of Chest Physicians and the American Academy of Pediatrics.
Air travel for pulmonology patients can be difficult, Dr. Millard noted, because "the environment is very dangerous." The cabin is pressurized, alveolar partial pressure falls with increasing altitude, and the partial pressure of oxygen is inversely proportional to altitude. Because the Joint Aviation Authorities stipulated that mean cabin pressure match an altitude of 8,000 feet, "this means they want us to all have an oxygen saturation of about 90%."
Supplemental oxygen during air travel can help patients, but identification of appropriate candidates varies. Guidelines from the American Thoracic Society and British Thoracic Society (Thorax 2002;57:289-304)recommend that patients with chronic lung disease be able to maintain an arterial oxygen tension greater than 50 mm Hg or 6.6 kilopascals (kPa), Dr. Millard said. However, because they tend to be younger than COPD (chronic obstructive pulmonary disease) patients and generally have no increased cardiovascular risk, use of such a cutoff value could be an oversimplification for patients with cystic fibrosis, said Dr. Millard, a pediatric pulmonologist at Helen DeVos Children’s Hospital in Grand Rapids, Mich.
Hypoxia during flight is a major concern. Consider whether your patient will be able to sustain hyperventilation that is spurred by hypoxia while on the airplane. Significant bronchospasm, for example, could impede prolonged hyperventilation, Dr. Millard said.
Consider a hypoxia inhalation test in advance of travel. This test requires that patients breathe a hypoxic mixture of 15% oxygen with nitrogen for 20 minutes to predict their reaction to hypoxia at 8,000 feet. Supplemental oxygen is recommended if their arterial oxygen tension drops below 50-55 mm Hg or 6.6-7.4 kPa.
"The hypoxia inhalation test is found to be safe," Dr. Millard said. Applicability outside the clinic setting is a concern, however: "The problem is, they are sitting. This may not fully represent the physical stress and environmental variability of air travel," including Transportation Security Administration screening and walking long distances.
For this reason, some experts advise also screening patients with a walk test prior to their trip, Dr. Millard said. The American Thoracic Society provides guidelines for conducting a functional exercise evaluation called a 6-minute walk test, for example (Am. J. Resp. Crit. Care Med. 2002;166:111-7).
Patients who require supplemental oxygen are permitted to use their own approved portable oxygen concentrator (POC) on all airlines that operate in the United States. POCs weigh 8-10 pounds and batteries last an average of about 4 hours, Dr. Millard said. Also, some POCs are pulse generated, meaning the patient must be able to inspire strongly enough to get oxygen.
Advise your patients or their families to check in advance if their airline requires approval from a physician for POC use, Dr. Millard said. "I had a patient who gave me 48 hours notice that they were going to fly. I had to fill out a form ahead of time for the airline."
Pulmonology patients also may request a travel letter, "which is especially important if they are going through customs," Dr. Millard said. Include their insurance information, your contact information, the telephone number for the clinic, and a list of medications (and approximate quantities required).
Airborne infection risk is another major concern. Most commercial aircraft recirculate 50% of the air delivered to the passenger cabin, Dr. Millard said. Ideally, the aircraft features HEPA (high-efficiency particulate air) filters, although the U.S. Federal Aviation Authority (FAA) and the U.K. Civil Aviation Authority do not mandate this level of filtration.
Dr. Millard cited a study that supports transmission of H1N1 influenza during flight (Epidemiol. Health 2010;32:e2010006). Officials at the Korea Centers for Disease Control and Prevention determined that an infected woman who flew from Los Angeles to Seoul in 2009 infected other passengers. The study includes a seating map of the Boeing 747 that shows where she and other passengers who got sick were seated.
"People are trying to figure this out to make [air travel] safer," Dr. Millard said. For example, one set of researchers assessed the ability of commercially available biosensors to detect airborne pathogens on airplanes (PLoS One 2011;6:e14520). With the current technology, however, only steady-state bacteria concentrations were detected in cases in which at least seven infected passengers either coughed 20 times per hour or sneezed 4 times an hour. And no sensor in the study detected airborne viruses well. Sensors with improved sensitivity and/or the screening of individual patients for respiratory illnesses prior to boarding might reduce the infection risk, Dr. Millard said.
For a list of POC devices that have been approved by the FAA, visit www.faa.gov/about/initiatives/cabin_safety/portable_oxygen/.
For additional guidance from the TSA on traveling with supplemental oxygen or other medical devices, you can refer patients to www.tsa.gov/travelers/airtravel/specialneeds/editorial_1374.shtm.
Dr. Millard said that she had no relevant disclosures.
FORT LAUDERDALE, FLA. – Your patients with cystic fibrosis or other pulmonary conditions may ask you if and when it’s safe for them to fly on an airplane.
How you respond can depend in part on their travel history, how long they will be exposed to increased cabin pressure, and if they are immunocompromised or have other risk factors for infection that are related to airborne pathogens, Dr. Susan L. Millard said.
Severe respiratory insufficiency, right heart failure or hemodynamic instability, and active pneumothorax are absolute contraindications to air travel, according to 30 experts who wrote a consensus statement for traveling with cystic fibrosis (J. Cyst. Fibros. 2010;9:385-99).
These first-ever European recommendations are useful because they address preparations for travel (for example, vaccinations and packing medication), important considerations during travel, and issues specific to the immunocompromised, Dr. Millard said at a pediatric pulmonology seminar, which was sponsored by the American College of Chest Physicians and the American Academy of Pediatrics.
Air travel for pulmonology patients can be difficult, Dr. Millard noted, because "the environment is very dangerous." The cabin is pressurized, alveolar partial pressure falls with increasing altitude, and the partial pressure of oxygen is inversely proportional to altitude. Because the Joint Aviation Authorities stipulated that mean cabin pressure match an altitude of 8,000 feet, "this means they want us to all have an oxygen saturation of about 90%."
Supplemental oxygen during air travel can help patients, but identification of appropriate candidates varies. Guidelines from the American Thoracic Society and British Thoracic Society (Thorax 2002;57:289-304)recommend that patients with chronic lung disease be able to maintain an arterial oxygen tension greater than 50 mm Hg or 6.6 kilopascals (kPa), Dr. Millard said. However, because they tend to be younger than COPD (chronic obstructive pulmonary disease) patients and generally have no increased cardiovascular risk, use of such a cutoff value could be an oversimplification for patients with cystic fibrosis, said Dr. Millard, a pediatric pulmonologist at Helen DeVos Children’s Hospital in Grand Rapids, Mich.
Hypoxia during flight is a major concern. Consider whether your patient will be able to sustain hyperventilation that is spurred by hypoxia while on the airplane. Significant bronchospasm, for example, could impede prolonged hyperventilation, Dr. Millard said.
Consider a hypoxia inhalation test in advance of travel. This test requires that patients breathe a hypoxic mixture of 15% oxygen with nitrogen for 20 minutes to predict their reaction to hypoxia at 8,000 feet. Supplemental oxygen is recommended if their arterial oxygen tension drops below 50-55 mm Hg or 6.6-7.4 kPa.
"The hypoxia inhalation test is found to be safe," Dr. Millard said. Applicability outside the clinic setting is a concern, however: "The problem is, they are sitting. This may not fully represent the physical stress and environmental variability of air travel," including Transportation Security Administration screening and walking long distances.
For this reason, some experts advise also screening patients with a walk test prior to their trip, Dr. Millard said. The American Thoracic Society provides guidelines for conducting a functional exercise evaluation called a 6-minute walk test, for example (Am. J. Resp. Crit. Care Med. 2002;166:111-7).
Patients who require supplemental oxygen are permitted to use their own approved portable oxygen concentrator (POC) on all airlines that operate in the United States. POCs weigh 8-10 pounds and batteries last an average of about 4 hours, Dr. Millard said. Also, some POCs are pulse generated, meaning the patient must be able to inspire strongly enough to get oxygen.
Advise your patients or their families to check in advance if their airline requires approval from a physician for POC use, Dr. Millard said. "I had a patient who gave me 48 hours notice that they were going to fly. I had to fill out a form ahead of time for the airline."
Pulmonology patients also may request a travel letter, "which is especially important if they are going through customs," Dr. Millard said. Include their insurance information, your contact information, the telephone number for the clinic, and a list of medications (and approximate quantities required).
Airborne infection risk is another major concern. Most commercial aircraft recirculate 50% of the air delivered to the passenger cabin, Dr. Millard said. Ideally, the aircraft features HEPA (high-efficiency particulate air) filters, although the U.S. Federal Aviation Authority (FAA) and the U.K. Civil Aviation Authority do not mandate this level of filtration.
Dr. Millard cited a study that supports transmission of H1N1 influenza during flight (Epidemiol. Health 2010;32:e2010006). Officials at the Korea Centers for Disease Control and Prevention determined that an infected woman who flew from Los Angeles to Seoul in 2009 infected other passengers. The study includes a seating map of the Boeing 747 that shows where she and other passengers who got sick were seated.
"People are trying to figure this out to make [air travel] safer," Dr. Millard said. For example, one set of researchers assessed the ability of commercially available biosensors to detect airborne pathogens on airplanes (PLoS One 2011;6:e14520). With the current technology, however, only steady-state bacteria concentrations were detected in cases in which at least seven infected passengers either coughed 20 times per hour or sneezed 4 times an hour. And no sensor in the study detected airborne viruses well. Sensors with improved sensitivity and/or the screening of individual patients for respiratory illnesses prior to boarding might reduce the infection risk, Dr. Millard said.
For a list of POC devices that have been approved by the FAA, visit www.faa.gov/about/initiatives/cabin_safety/portable_oxygen/.
For additional guidance from the TSA on traveling with supplemental oxygen or other medical devices, you can refer patients to www.tsa.gov/travelers/airtravel/specialneeds/editorial_1374.shtm.
Dr. Millard said that she had no relevant disclosures.
Nanotechnology Vehicle Speeds Numbing of Topical Lidocaine
MIAMI BEACH – A 4% topical lidocaine cream with a nanotechnology-based vehicle in development provided superior pain relief before facial filler injections, compared with the most commonly-used topical anesthetic cream, according to patient and physician ratings in two split-face studies.
"This really has potential as the next topical anesthetic," Dr. Glynis Ablon said at the South Beach Symposium.
Although most dermatologists and pediatricians apply topical anesthetic products (such as commonly used LMX4 cream) approximately 20 minutes before needle-based procedures, the efficacy of NTL4 (Cutiecaine Cream) was seen in as little as 5 minutes, Dr. Ablon said.
NTL4 is formulated with a novel nanoparticle vehicle (INParT Drug Delivery System) that passively transports active compounds deeper into the skin. "It's a great new way to deliver lidocaine into the skin faster," said Dr. Ablon, a dermatologist in private practice in Manhattan Beach, Calif. The technology is also being assessed for new topical hyaluronic acid and topical botulinum toxins.
Dr. Ablon and her colleague Dr. Mark Nestor, who is in private practice in Aventura, Fla., conducted an initial study of 30 patients with NTL4 applied to one side of their face and over-the-counter LMX4 to the contralateral side, followed by a 20-second massage. After 20 minutes, Restylane was injected for facial rejuvenation. Participants and blinded raters assessed pain relief.
"The study was started to see if we could get that [numbing effect] in 20 minutes, and obviously faster numbing times could be a great thing for dermatologists as well as pediatricians," Dr. Ablon said.
Twenty patients (67%) reported minimal or no pain when injected on the NTL4 side of their face, compared with 12 patients (40%) who reported minimal or no pain on the LMX4 treated side. "Overall, patients significantly preferred treatment with NTL4 over LMX4," she said. The mean visual analog score (VAS) for pain was 1.99 in the NTL4 group vs. 3.08 in the LMX4 group immediately after injection; 0.21 vs. 0.74 at 1 hour; and 0.07 and 0.31 at 3 hours postinjection.
Blinded investigators also rated significantly less pain with NTL4, Dr. Ablon said.
Adverse events were minor and included some tenderness and bruising. Also, there was "some edema we believe was due to the actual injection of the Restylane."
In a second study, Dr. Ablon and Dr. Nestor assessed 5-, 10-, and 15-minute application times. The 20 participants had a 30-second massage of the topical anesthetic and subsequent Restylane injections of their nasolabial folds.
Sixteen patients (80%) treated with NTL4 reported minimal or no pain, compared with one patient (5%) treated with LMX4, according to pooled data for subjective pain ratings after 5-, 10-, and 15-minute application times.
"We did show that they had significantly less pain on injection with the NTL4 at the 5- and 15-minute incubation, with a trend favoring it at 10 minutes," Dr. Ablon said.
Mean VAS pain scores were significantly lower for the NTL4 group when injected 5 minutes after application: 1.72 for NTL4 vs. 4.20 for LMX4. Patients also reported a significant difference in pain when injected 15 minutes after injection: 1.92 with NTL4 vs. 4.67 with LMX4. There was a nonsignificant trend favoring lower VAS scores with NTL4 after a 10-minute application time(1.08 for NTL4 and 3.00 for LMX4).
All patients preferred the NTL4 side versus the LMX4 side for pain relief. Blinded investigators’ evaluation of pain was also statistically significant in favor of the NTL4, Dr. Ablon said.
Adverse events were minor in this study as well. "We did have one patient with edema that we think might have been an actual reaction to lidocaine," she noted.
"There is a lot going on with nanotechnology, and [it] is here to stay," Dr. Ablon said. This technology can be found in suntan lotions, age-defying makeup, and even toothpaste that coats damaged enamel.
The studies were funded through an unrestricted educational grant from Innovatech. Dr. Ablon and Dr. Nestor are consultants and investigators for Transdermal Corp.
MIAMI BEACH – A 4% topical lidocaine cream with a nanotechnology-based vehicle in development provided superior pain relief before facial filler injections, compared with the most commonly-used topical anesthetic cream, according to patient and physician ratings in two split-face studies.
"This really has potential as the next topical anesthetic," Dr. Glynis Ablon said at the South Beach Symposium.
Although most dermatologists and pediatricians apply topical anesthetic products (such as commonly used LMX4 cream) approximately 20 minutes before needle-based procedures, the efficacy of NTL4 (Cutiecaine Cream) was seen in as little as 5 minutes, Dr. Ablon said.
NTL4 is formulated with a novel nanoparticle vehicle (INParT Drug Delivery System) that passively transports active compounds deeper into the skin. "It's a great new way to deliver lidocaine into the skin faster," said Dr. Ablon, a dermatologist in private practice in Manhattan Beach, Calif. The technology is also being assessed for new topical hyaluronic acid and topical botulinum toxins.
Dr. Ablon and her colleague Dr. Mark Nestor, who is in private practice in Aventura, Fla., conducted an initial study of 30 patients with NTL4 applied to one side of their face and over-the-counter LMX4 to the contralateral side, followed by a 20-second massage. After 20 minutes, Restylane was injected for facial rejuvenation. Participants and blinded raters assessed pain relief.
"The study was started to see if we could get that [numbing effect] in 20 minutes, and obviously faster numbing times could be a great thing for dermatologists as well as pediatricians," Dr. Ablon said.
Twenty patients (67%) reported minimal or no pain when injected on the NTL4 side of their face, compared with 12 patients (40%) who reported minimal or no pain on the LMX4 treated side. "Overall, patients significantly preferred treatment with NTL4 over LMX4," she said. The mean visual analog score (VAS) for pain was 1.99 in the NTL4 group vs. 3.08 in the LMX4 group immediately after injection; 0.21 vs. 0.74 at 1 hour; and 0.07 and 0.31 at 3 hours postinjection.
Blinded investigators also rated significantly less pain with NTL4, Dr. Ablon said.
Adverse events were minor and included some tenderness and bruising. Also, there was "some edema we believe was due to the actual injection of the Restylane."
In a second study, Dr. Ablon and Dr. Nestor assessed 5-, 10-, and 15-minute application times. The 20 participants had a 30-second massage of the topical anesthetic and subsequent Restylane injections of their nasolabial folds.
Sixteen patients (80%) treated with NTL4 reported minimal or no pain, compared with one patient (5%) treated with LMX4, according to pooled data for subjective pain ratings after 5-, 10-, and 15-minute application times.
"We did show that they had significantly less pain on injection with the NTL4 at the 5- and 15-minute incubation, with a trend favoring it at 10 minutes," Dr. Ablon said.
Mean VAS pain scores were significantly lower for the NTL4 group when injected 5 minutes after application: 1.72 for NTL4 vs. 4.20 for LMX4. Patients also reported a significant difference in pain when injected 15 minutes after injection: 1.92 with NTL4 vs. 4.67 with LMX4. There was a nonsignificant trend favoring lower VAS scores with NTL4 after a 10-minute application time(1.08 for NTL4 and 3.00 for LMX4).
All patients preferred the NTL4 side versus the LMX4 side for pain relief. Blinded investigators’ evaluation of pain was also statistically significant in favor of the NTL4, Dr. Ablon said.
Adverse events were minor in this study as well. "We did have one patient with edema that we think might have been an actual reaction to lidocaine," she noted.
"There is a lot going on with nanotechnology, and [it] is here to stay," Dr. Ablon said. This technology can be found in suntan lotions, age-defying makeup, and even toothpaste that coats damaged enamel.
The studies were funded through an unrestricted educational grant from Innovatech. Dr. Ablon and Dr. Nestor are consultants and investigators for Transdermal Corp.
MIAMI BEACH – A 4% topical lidocaine cream with a nanotechnology-based vehicle in development provided superior pain relief before facial filler injections, compared with the most commonly-used topical anesthetic cream, according to patient and physician ratings in two split-face studies.
"This really has potential as the next topical anesthetic," Dr. Glynis Ablon said at the South Beach Symposium.
Although most dermatologists and pediatricians apply topical anesthetic products (such as commonly used LMX4 cream) approximately 20 minutes before needle-based procedures, the efficacy of NTL4 (Cutiecaine Cream) was seen in as little as 5 minutes, Dr. Ablon said.
NTL4 is formulated with a novel nanoparticle vehicle (INParT Drug Delivery System) that passively transports active compounds deeper into the skin. "It's a great new way to deliver lidocaine into the skin faster," said Dr. Ablon, a dermatologist in private practice in Manhattan Beach, Calif. The technology is also being assessed for new topical hyaluronic acid and topical botulinum toxins.
Dr. Ablon and her colleague Dr. Mark Nestor, who is in private practice in Aventura, Fla., conducted an initial study of 30 patients with NTL4 applied to one side of their face and over-the-counter LMX4 to the contralateral side, followed by a 20-second massage. After 20 minutes, Restylane was injected for facial rejuvenation. Participants and blinded raters assessed pain relief.
"The study was started to see if we could get that [numbing effect] in 20 minutes, and obviously faster numbing times could be a great thing for dermatologists as well as pediatricians," Dr. Ablon said.
Twenty patients (67%) reported minimal or no pain when injected on the NTL4 side of their face, compared with 12 patients (40%) who reported minimal or no pain on the LMX4 treated side. "Overall, patients significantly preferred treatment with NTL4 over LMX4," she said. The mean visual analog score (VAS) for pain was 1.99 in the NTL4 group vs. 3.08 in the LMX4 group immediately after injection; 0.21 vs. 0.74 at 1 hour; and 0.07 and 0.31 at 3 hours postinjection.
Blinded investigators also rated significantly less pain with NTL4, Dr. Ablon said.
Adverse events were minor and included some tenderness and bruising. Also, there was "some edema we believe was due to the actual injection of the Restylane."
In a second study, Dr. Ablon and Dr. Nestor assessed 5-, 10-, and 15-minute application times. The 20 participants had a 30-second massage of the topical anesthetic and subsequent Restylane injections of their nasolabial folds.
Sixteen patients (80%) treated with NTL4 reported minimal or no pain, compared with one patient (5%) treated with LMX4, according to pooled data for subjective pain ratings after 5-, 10-, and 15-minute application times.
"We did show that they had significantly less pain on injection with the NTL4 at the 5- and 15-minute incubation, with a trend favoring it at 10 minutes," Dr. Ablon said.
Mean VAS pain scores were significantly lower for the NTL4 group when injected 5 minutes after application: 1.72 for NTL4 vs. 4.20 for LMX4. Patients also reported a significant difference in pain when injected 15 minutes after injection: 1.92 with NTL4 vs. 4.67 with LMX4. There was a nonsignificant trend favoring lower VAS scores with NTL4 after a 10-minute application time(1.08 for NTL4 and 3.00 for LMX4).
All patients preferred the NTL4 side versus the LMX4 side for pain relief. Blinded investigators’ evaluation of pain was also statistically significant in favor of the NTL4, Dr. Ablon said.
Adverse events were minor in this study as well. "We did have one patient with edema that we think might have been an actual reaction to lidocaine," she noted.
"There is a lot going on with nanotechnology, and [it] is here to stay," Dr. Ablon said. This technology can be found in suntan lotions, age-defying makeup, and even toothpaste that coats damaged enamel.
The studies were funded through an unrestricted educational grant from Innovatech. Dr. Ablon and Dr. Nestor are consultants and investigators for Transdermal Corp.
FROM THE SOUTH BEACH SYMPOSIUM
Major Finding: Sixteen patients (80%) treated with NTL4 reported minimal or no pain, compared with one patient (5%) treated with LMX4. Mean visual analog scale scores were significantly lower for injection pain with a 5-minute application: 1.72 in the NTL4 group vs. 4.20 in the LMX4 group.
Data Source: Split-face comparison of 20 patients applying NTL4 or LMX4 for 5, 10, or 15 minutes prior to facial filler injections.
Disclosures: Dr. Ablon and Dr. Nestor are consultants and investigators for Transdermal Corp.
Lipolysis With Sodium Deoxycholate Reduces Submental Fat
MIAMI BEACH – Lipolysis with proprietary sodium deoxycholate injections significantly decreased the volume of submental fat in a randomized, vehicle-control, phase IIb study.
Investigators at 10 sites in the United States randomized 129 patients with moderate-to-severe "submental convexity" to low-dose or high-dose sodium deoxycholate (ATX-101, Kythera Biopharmaceuticals) or placebo. Administration was once a month for up to 5 months, Dr. Patricia S. Walker said at the South Beach Symposium.
Patients and clinicians rated changes at 16 weeks and 3 months after the final treatment, compared with baseline. At the same time, investigators also compared MRI volume measurements as an objective measure of fat changes.
MRI revealed significant reductions in submental fat at week 16 and week 32 from baseline, compared with placebo, for patients randomized to 2 mg/cm2 sodium deoxycholate (the high-dose group).
Seven dermatologists and three plastic surgeons at the 10 sites treated the participants and performed global assessments. The clinicians rated changes using a 5-point Clinician-Reported Submental Fat Impact Scale.
Again, the 2-mg/cm2 dose was more efficacious than placebo, with statistically significant reductions rated by doctors at week 16 and week 32. Although the effect on patients who received the 1-mg/cm2 dose (low-dose group) did not reach significance, this dose was also associated with improvement compared with placebo, suggesting a dose response, Dr. Walker said.
Patients also rated their own changes in submental fat using a 5-point Patient-Reported Submental Fat Impact Scale at baseline, week 16, and week 32.
Interestingly, all patients reported subjective improvements compared with baseline – even those assigned to vehicle-only injections. "Patients have a perception of self-improvement," Dr. Walker said. "There was very little change on MRI in the vehicle group, so it’s a true placebo effect – patients want to see a response."
Sodium deoxycholate selectively targets adipocytes. Histology showed infiltration of macrophages by day 3, with destruction and clearance of fat over time. "Twenty-eight days is about the right time for these injections to clear fat," said Dr. Walker, who is chief medical officer for Kythera Biopharmaceuticals.
Sodium deoxycholate is an endogenous, secondary bile acid. This is "how we break down fat in our diet," Dr. Walker said.
A total 114 patients completed the study. Fitzpatrick skin types ranged from I to VI, average age was 46 years (range 24-65 years), and the mean body mass index was 30.6 kg/m2 (range 19.5-48.8). "We found chin fat is not necessarily related to [body] size. It can be familial," Dr. Walker said.
Most adverse events were mild, transient, and limited to the chin area. "If you don’t have a safe product, you don’t have an aesthetic product," she said.
Treatment-related swelling, pain, and numbness were the most common adverse events. "There also was erythema in the vehicle group, so it was probably injection-technique related."
ATX-101 is a safe and effective treatment for submental fat reduction, Dr. Walker said. "It is nice to have an aesthetic product that everyone sees works."
Phase III trials are planned this year, according to Kythera Biopharmaceuticals.
Dr. Walker is an employee and shareholder of the company.
MIAMI BEACH – Lipolysis with proprietary sodium deoxycholate injections significantly decreased the volume of submental fat in a randomized, vehicle-control, phase IIb study.
Investigators at 10 sites in the United States randomized 129 patients with moderate-to-severe "submental convexity" to low-dose or high-dose sodium deoxycholate (ATX-101, Kythera Biopharmaceuticals) or placebo. Administration was once a month for up to 5 months, Dr. Patricia S. Walker said at the South Beach Symposium.
Patients and clinicians rated changes at 16 weeks and 3 months after the final treatment, compared with baseline. At the same time, investigators also compared MRI volume measurements as an objective measure of fat changes.
MRI revealed significant reductions in submental fat at week 16 and week 32 from baseline, compared with placebo, for patients randomized to 2 mg/cm2 sodium deoxycholate (the high-dose group).
Seven dermatologists and three plastic surgeons at the 10 sites treated the participants and performed global assessments. The clinicians rated changes using a 5-point Clinician-Reported Submental Fat Impact Scale.
Again, the 2-mg/cm2 dose was more efficacious than placebo, with statistically significant reductions rated by doctors at week 16 and week 32. Although the effect on patients who received the 1-mg/cm2 dose (low-dose group) did not reach significance, this dose was also associated with improvement compared with placebo, suggesting a dose response, Dr. Walker said.
Patients also rated their own changes in submental fat using a 5-point Patient-Reported Submental Fat Impact Scale at baseline, week 16, and week 32.
Interestingly, all patients reported subjective improvements compared with baseline – even those assigned to vehicle-only injections. "Patients have a perception of self-improvement," Dr. Walker said. "There was very little change on MRI in the vehicle group, so it’s a true placebo effect – patients want to see a response."
Sodium deoxycholate selectively targets adipocytes. Histology showed infiltration of macrophages by day 3, with destruction and clearance of fat over time. "Twenty-eight days is about the right time for these injections to clear fat," said Dr. Walker, who is chief medical officer for Kythera Biopharmaceuticals.
Sodium deoxycholate is an endogenous, secondary bile acid. This is "how we break down fat in our diet," Dr. Walker said.
A total 114 patients completed the study. Fitzpatrick skin types ranged from I to VI, average age was 46 years (range 24-65 years), and the mean body mass index was 30.6 kg/m2 (range 19.5-48.8). "We found chin fat is not necessarily related to [body] size. It can be familial," Dr. Walker said.
Most adverse events were mild, transient, and limited to the chin area. "If you don’t have a safe product, you don’t have an aesthetic product," she said.
Treatment-related swelling, pain, and numbness were the most common adverse events. "There also was erythema in the vehicle group, so it was probably injection-technique related."
ATX-101 is a safe and effective treatment for submental fat reduction, Dr. Walker said. "It is nice to have an aesthetic product that everyone sees works."
Phase III trials are planned this year, according to Kythera Biopharmaceuticals.
Dr. Walker is an employee and shareholder of the company.
MIAMI BEACH – Lipolysis with proprietary sodium deoxycholate injections significantly decreased the volume of submental fat in a randomized, vehicle-control, phase IIb study.
Investigators at 10 sites in the United States randomized 129 patients with moderate-to-severe "submental convexity" to low-dose or high-dose sodium deoxycholate (ATX-101, Kythera Biopharmaceuticals) or placebo. Administration was once a month for up to 5 months, Dr. Patricia S. Walker said at the South Beach Symposium.
Patients and clinicians rated changes at 16 weeks and 3 months after the final treatment, compared with baseline. At the same time, investigators also compared MRI volume measurements as an objective measure of fat changes.
MRI revealed significant reductions in submental fat at week 16 and week 32 from baseline, compared with placebo, for patients randomized to 2 mg/cm2 sodium deoxycholate (the high-dose group).
Seven dermatologists and three plastic surgeons at the 10 sites treated the participants and performed global assessments. The clinicians rated changes using a 5-point Clinician-Reported Submental Fat Impact Scale.
Again, the 2-mg/cm2 dose was more efficacious than placebo, with statistically significant reductions rated by doctors at week 16 and week 32. Although the effect on patients who received the 1-mg/cm2 dose (low-dose group) did not reach significance, this dose was also associated with improvement compared with placebo, suggesting a dose response, Dr. Walker said.
Patients also rated their own changes in submental fat using a 5-point Patient-Reported Submental Fat Impact Scale at baseline, week 16, and week 32.
Interestingly, all patients reported subjective improvements compared with baseline – even those assigned to vehicle-only injections. "Patients have a perception of self-improvement," Dr. Walker said. "There was very little change on MRI in the vehicle group, so it’s a true placebo effect – patients want to see a response."
Sodium deoxycholate selectively targets adipocytes. Histology showed infiltration of macrophages by day 3, with destruction and clearance of fat over time. "Twenty-eight days is about the right time for these injections to clear fat," said Dr. Walker, who is chief medical officer for Kythera Biopharmaceuticals.
Sodium deoxycholate is an endogenous, secondary bile acid. This is "how we break down fat in our diet," Dr. Walker said.
A total 114 patients completed the study. Fitzpatrick skin types ranged from I to VI, average age was 46 years (range 24-65 years), and the mean body mass index was 30.6 kg/m2 (range 19.5-48.8). "We found chin fat is not necessarily related to [body] size. It can be familial," Dr. Walker said.
Most adverse events were mild, transient, and limited to the chin area. "If you don’t have a safe product, you don’t have an aesthetic product," she said.
Treatment-related swelling, pain, and numbness were the most common adverse events. "There also was erythema in the vehicle group, so it was probably injection-technique related."
ATX-101 is a safe and effective treatment for submental fat reduction, Dr. Walker said. "It is nice to have an aesthetic product that everyone sees works."
Phase III trials are planned this year, according to Kythera Biopharmaceuticals.
Dr. Walker is an employee and shareholder of the company.
FROM THE SOUTH BEACH SYMPOSIUM
Major Finding: MRI revealed significant reductions in submental fat at week 16 and week 32 from baseline, compared with placebo, for patients randomized to 2 mg/cm2 sodium deoxycholate (the high-dose group).
Data Source: Investigators at 10 sites in the United States randomized 129 patients with moderate-to-severe "submental convexity" to low-dose or high-dose sodium deoxycholate or placebo.
Disclosures: Dr. Walker is an employee and shareholder of Kythera.
Picture Review: Dermoscopy Distinguishes Hair Disorders
ORLANDO - Dermoscopy can help with the diagnosis and management of hair and scale disorders.
In some instances, dermoscopy very rapidly reveal a characteristic sign of a hair or scalp condition. In presentations where dermoscopy only narrows down your differential diagnosis, further work-up with histology might be required, said Dr. Antonella Tosti, professor of clinical dermatology at the University of Miami.
How well do you know your dermoscopy diagnoses?
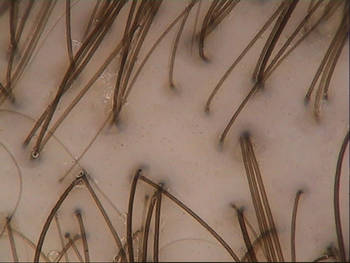
|
All photos courtesy Dr. Antonella Tosti Normal Scalp. Normally, hairs are arranged in follicular units, with up to three hair shafts from the same orifice. "You also see single hairs coming out from the scalp," Dr. Tosti said. Most hair disorders feature a high number of single hairs. A normal vascular pattern presents as interfollicular red loops and arborizing red lines. |
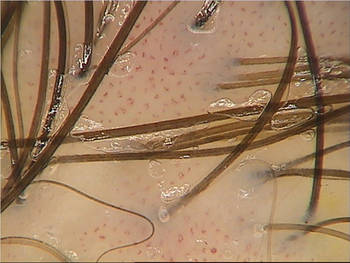
|
Psoriasis. Twisted, coiled loops are a sign of scalp psoriasis. Folliculitis decalvans is the only other scalp condition with a similar loop presentation. If you observe white scales, use high-magnification dermoscopy to look at the vessels before diagnosing psoriasis. White scales can also be seen in cicatricial alopecias such as lupus erythematosus and lichen planopilaris. "So you may need to take a biopsy when patient presents with wide, white scales. It's not always psoriasis," Dr. Tosti said. |
|
Alopecia areata. Alopecia areata and androgenetic alopecia each will feature yellow dots that correspond to follicular openings. "How often you see the yellow dots depends on the patients. They are seen often in [whites] and less commonly seen in darker-skinned patients," Dr. Tosti said. Hair diameter variability, a sign of androgenetic alopecia, can help you to distinguish these conditions. More than 20% variability in shaft thickness and an increase in the number of thinner hairs with advanced disease are diagnostic. "My suggestion is always use your dermatoscope when women complain of hair loss," Dr. Tosti said. "It is an easy and quick way to distinguish androgenetic alopecia." |
|
Discoid lupus erythematosus. A follicular red dot pattern is characteristic of this condition on dermoscopy. An atrophic epidermis can be seen in active lesions. The red dots are important to recognize because, if treated, hair can regrow in the affected area. "Make the diagnosis early and treat the patient immediately," Dr. Tosti said. Also, the red dot pattern will help you to distinguish this disorder from other conditions that can cause cicatricial alopecia. |

|
Cicatricial alopecia. A loss of follicular openings is an important clue on dermoscopy that indicates cicatricial alopecias |

|
Traction alopecia. Hair casts are a useful dermoscopy clue for this condition. "You can see them immediately," Dr. Tosti said. "Hair casts are also useful in African Americans if their hairstyle is associated with traction, even if they don't have alopecia. Sometimes they will deny a traction hairstyle." |
|
Tinea capitis. Dermoscopy facilitates this diagnosis. Look for comma-shaped hairs, a very specific sign seen in both ectothrix and endothrix T. capitis infections. "This is something you should look for in children. It's very fast," Dr. Tosti said. The comma hairs will be more numerous in recent infections, she added. |
|
Monilethrix. Dermoscopy can help you diagnose this condition very, very fast, Dr. Tosti said. The condition features beading of the hair shafts. The autosomal recessive form of monilethrix will appear much more severe on dermoscopy (J. Invest. Dermatol. 2006;126:1292-6). |
Dr. Tosti said that she had no relevant disclosures.
ORLANDO - Dermoscopy can help with the diagnosis and management of hair and scale disorders.
In some instances, dermoscopy very rapidly reveal a characteristic sign of a hair or scalp condition. In presentations where dermoscopy only narrows down your differential diagnosis, further work-up with histology might be required, said Dr. Antonella Tosti, professor of clinical dermatology at the University of Miami.
How well do you know your dermoscopy diagnoses?

|
All photos courtesy Dr. Antonella Tosti Normal Scalp. Normally, hairs are arranged in follicular units, with up to three hair shafts from the same orifice. "You also see single hairs coming out from the scalp," Dr. Tosti said. Most hair disorders feature a high number of single hairs. A normal vascular pattern presents as interfollicular red loops and arborizing red lines. |

|
Psoriasis. Twisted, coiled loops are a sign of scalp psoriasis. Folliculitis decalvans is the only other scalp condition with a similar loop presentation. If you observe white scales, use high-magnification dermoscopy to look at the vessels before diagnosing psoriasis. White scales can also be seen in cicatricial alopecias such as lupus erythematosus and lichen planopilaris. "So you may need to take a biopsy when patient presents with wide, white scales. It's not always psoriasis," Dr. Tosti said. |

|
Alopecia areata. Alopecia areata and androgenetic alopecia each will feature yellow dots that correspond to follicular openings. "How often you see the yellow dots depends on the patients. They are seen often in [whites] and less commonly seen in darker-skinned patients," Dr. Tosti said. Hair diameter variability, a sign of androgenetic alopecia, can help you to distinguish these conditions. More than 20% variability in shaft thickness and an increase in the number of thinner hairs with advanced disease are diagnostic. "My suggestion is always use your dermatoscope when women complain of hair loss," Dr. Tosti said. "It is an easy and quick way to distinguish androgenetic alopecia." |

|
Discoid lupus erythematosus. A follicular red dot pattern is characteristic of this condition on dermoscopy. An atrophic epidermis can be seen in active lesions. The red dots are important to recognize because, if treated, hair can regrow in the affected area. "Make the diagnosis early and treat the patient immediately," Dr. Tosti said. Also, the red dot pattern will help you to distinguish this disorder from other conditions that can cause cicatricial alopecia. |

|
Cicatricial alopecia. A loss of follicular openings is an important clue on dermoscopy that indicates cicatricial alopecias |

|
Traction alopecia. Hair casts are a useful dermoscopy clue for this condition. "You can see them immediately," Dr. Tosti said. "Hair casts are also useful in African Americans if their hairstyle is associated with traction, even if they don't have alopecia. Sometimes they will deny a traction hairstyle." |

|
Tinea capitis. Dermoscopy facilitates this diagnosis. Look for comma-shaped hairs, a very specific sign seen in both ectothrix and endothrix T. capitis infections. "This is something you should look for in children. It's very fast," Dr. Tosti said. The comma hairs will be more numerous in recent infections, she added. |

|
Monilethrix. Dermoscopy can help you diagnose this condition very, very fast, Dr. Tosti said. The condition features beading of the hair shafts. The autosomal recessive form of monilethrix will appear much more severe on dermoscopy (J. Invest. Dermatol. 2006;126:1292-6). |
Dr. Tosti said that she had no relevant disclosures.
ORLANDO - Dermoscopy can help with the diagnosis and management of hair and scale disorders.
In some instances, dermoscopy very rapidly reveal a characteristic sign of a hair or scalp condition. In presentations where dermoscopy only narrows down your differential diagnosis, further work-up with histology might be required, said Dr. Antonella Tosti, professor of clinical dermatology at the University of Miami.
How well do you know your dermoscopy diagnoses?

|
All photos courtesy Dr. Antonella Tosti Normal Scalp. Normally, hairs are arranged in follicular units, with up to three hair shafts from the same orifice. "You also see single hairs coming out from the scalp," Dr. Tosti said. Most hair disorders feature a high number of single hairs. A normal vascular pattern presents as interfollicular red loops and arborizing red lines. |

|
Psoriasis. Twisted, coiled loops are a sign of scalp psoriasis. Folliculitis decalvans is the only other scalp condition with a similar loop presentation. If you observe white scales, use high-magnification dermoscopy to look at the vessels before diagnosing psoriasis. White scales can also be seen in cicatricial alopecias such as lupus erythematosus and lichen planopilaris. "So you may need to take a biopsy when patient presents with wide, white scales. It's not always psoriasis," Dr. Tosti said. |

|
Alopecia areata. Alopecia areata and androgenetic alopecia each will feature yellow dots that correspond to follicular openings. "How often you see the yellow dots depends on the patients. They are seen often in [whites] and less commonly seen in darker-skinned patients," Dr. Tosti said. Hair diameter variability, a sign of androgenetic alopecia, can help you to distinguish these conditions. More than 20% variability in shaft thickness and an increase in the number of thinner hairs with advanced disease are diagnostic. "My suggestion is always use your dermatoscope when women complain of hair loss," Dr. Tosti said. "It is an easy and quick way to distinguish androgenetic alopecia." |

|
Discoid lupus erythematosus. A follicular red dot pattern is characteristic of this condition on dermoscopy. An atrophic epidermis can be seen in active lesions. The red dots are important to recognize because, if treated, hair can regrow in the affected area. "Make the diagnosis early and treat the patient immediately," Dr. Tosti said. Also, the red dot pattern will help you to distinguish this disorder from other conditions that can cause cicatricial alopecia. |

|
Cicatricial alopecia. A loss of follicular openings is an important clue on dermoscopy that indicates cicatricial alopecias |

|
Traction alopecia. Hair casts are a useful dermoscopy clue for this condition. "You can see them immediately," Dr. Tosti said. "Hair casts are also useful in African Americans if their hairstyle is associated with traction, even if they don't have alopecia. Sometimes they will deny a traction hairstyle." |

|
Tinea capitis. Dermoscopy facilitates this diagnosis. Look for comma-shaped hairs, a very specific sign seen in both ectothrix and endothrix T. capitis infections. "This is something you should look for in children. It's very fast," Dr. Tosti said. The comma hairs will be more numerous in recent infections, she added. |

|
Monilethrix. Dermoscopy can help you diagnose this condition very, very fast, Dr. Tosti said. The condition features beading of the hair shafts. The autosomal recessive form of monilethrix will appear much more severe on dermoscopy (J. Invest. Dermatol. 2006;126:1292-6). |
Dr. Tosti said that she had no relevant disclosures.
FROM THE ANNUAL MEETING OF THE FLORIDA SOCIETY OF DERMATOLOGIC SURGEONS
Picture Review: Dermoscopy Distinguishes Hair Disorders
ORLANDO - Dermoscopy can help with the diagnosis and management of hair and scale disorders.
In some instances, dermoscopy very rapidly reveal a characteristic sign of a hair or scalp condition. In presentations where dermoscopy only narrows down your differential diagnosis, further work-up with histology might be required, said Dr. Antonella Tosti, professor of clinical dermatology at the University of Miami.
How well do you know your dermoscopy diagnoses?
|
All photos courtesy Dr. Antonella Tosti Normal Scalp. Normally, hairs are arranged in follicular units, with up to three hair shafts from the same orifice. "You also see single hairs coming out from the scalp," Dr. Tosti said. Most hair disorders feature a high number of single hairs. A normal vascular pattern presents as interfollicular red loops and arborizing red lines. |
|
Psoriasis. Twisted, coiled loops are a sign of scalp psoriasis. Folliculitis decalvans is the only other scalp condition with a similar loop presentation. If you observe white scales, use high-magnification dermoscopy to look at the vessels before diagnosing psoriasis. White scales can also be seen in cicatricial alopecias such as lupus erythematosus and lichen planopilaris. "So you may need to take a biopsy when patient presents with wide, white scales. It's not always psoriasis," Dr. Tosti said. |
|
Alopecia areata. Alopecia areata and androgenetic alopecia each will feature yellow dots that correspond to follicular openings. "How often you see the yellow dots depends on the patients. They are seen often in [whites] and less commonly seen in darker-skinned patients," Dr. Tosti said. Hair diameter variability, a sign of androgenetic alopecia, can help you to distinguish these conditions. More than 20% variability in shaft thickness and an increase in the number of thinner hairs with advanced disease are diagnostic. "My suggestion is always use your dermatoscope when women complain of hair loss," Dr. Tosti said. "It is an easy and quick way to distinguish androgenetic alopecia." |
|
Discoid lupus erythematosus. A follicular red dot pattern is characteristic of this condition on dermoscopy. An atrophic epidermis can be seen in active lesions. The red dots are important to recognize because, if treated, hair can regrow in the affected area. "Make the diagnosis early and treat the patient immediately," Dr. Tosti said. Also, the red dot pattern will help you to distinguish this disorder from other conditions that can cause cicatricial alopecia. |

|
Cicatricial alopecia. A loss of follicular openings is an important clue on dermoscopy that indicates cicatricial alopecias |

|
Traction alopecia. Hair casts are a useful dermoscopy clue for this condition. "You can see them immediately," Dr. Tosti said. "Hair casts are also useful in African Americans if their hairstyle is associated with traction, even if they don't have alopecia. Sometimes they will deny a traction hairstyle." |
|
Tinea capitis. Dermoscopy facilitates this diagnosis. Look for comma-shaped hairs, a very specific sign seen in both ectothrix and endothrix T. capitis infections. "This is something you should look for in children. It's very fast," Dr. Tosti said. The comma hairs will be more numerous in recent infections, she added. |
|
Monilethrix. Dermoscopy can help you diagnose this condition very, very fast, Dr. Tosti said. The condition features beading of the hair shafts. The autosomal recessive form of monilethrix will appear much more severe on dermoscopy (J. Invest. Dermatol. 2006;126:1292-6). |
Dr. Tosti said that she had no relevant disclosures.
ORLANDO - Dermoscopy can help with the diagnosis and management of hair and scale disorders.
In some instances, dermoscopy very rapidly reveal a characteristic sign of a hair or scalp condition. In presentations where dermoscopy only narrows down your differential diagnosis, further work-up with histology might be required, said Dr. Antonella Tosti, professor of clinical dermatology at the University of Miami.
How well do you know your dermoscopy diagnoses?

|
All photos courtesy Dr. Antonella Tosti Normal Scalp. Normally, hairs are arranged in follicular units, with up to three hair shafts from the same orifice. "You also see single hairs coming out from the scalp," Dr. Tosti said. Most hair disorders feature a high number of single hairs. A normal vascular pattern presents as interfollicular red loops and arborizing red lines. |

|
Psoriasis. Twisted, coiled loops are a sign of scalp psoriasis. Folliculitis decalvans is the only other scalp condition with a similar loop presentation. If you observe white scales, use high-magnification dermoscopy to look at the vessels before diagnosing psoriasis. White scales can also be seen in cicatricial alopecias such as lupus erythematosus and lichen planopilaris. "So you may need to take a biopsy when patient presents with wide, white scales. It's not always psoriasis," Dr. Tosti said. |

|
Alopecia areata. Alopecia areata and androgenetic alopecia each will feature yellow dots that correspond to follicular openings. "How often you see the yellow dots depends on the patients. They are seen often in [whites] and less commonly seen in darker-skinned patients," Dr. Tosti said. Hair diameter variability, a sign of androgenetic alopecia, can help you to distinguish these conditions. More than 20% variability in shaft thickness and an increase in the number of thinner hairs with advanced disease are diagnostic. "My suggestion is always use your dermatoscope when women complain of hair loss," Dr. Tosti said. "It is an easy and quick way to distinguish androgenetic alopecia." |

|
Discoid lupus erythematosus. A follicular red dot pattern is characteristic of this condition on dermoscopy. An atrophic epidermis can be seen in active lesions. The red dots are important to recognize because, if treated, hair can regrow in the affected area. "Make the diagnosis early and treat the patient immediately," Dr. Tosti said. Also, the red dot pattern will help you to distinguish this disorder from other conditions that can cause cicatricial alopecia. |

|
Cicatricial alopecia. A loss of follicular openings is an important clue on dermoscopy that indicates cicatricial alopecias |

|
Traction alopecia. Hair casts are a useful dermoscopy clue for this condition. "You can see them immediately," Dr. Tosti said. "Hair casts are also useful in African Americans if their hairstyle is associated with traction, even if they don't have alopecia. Sometimes they will deny a traction hairstyle." |

|
Tinea capitis. Dermoscopy facilitates this diagnosis. Look for comma-shaped hairs, a very specific sign seen in both ectothrix and endothrix T. capitis infections. "This is something you should look for in children. It's very fast," Dr. Tosti said. The comma hairs will be more numerous in recent infections, she added. |

|
Monilethrix. Dermoscopy can help you diagnose this condition very, very fast, Dr. Tosti said. The condition features beading of the hair shafts. The autosomal recessive form of monilethrix will appear much more severe on dermoscopy (J. Invest. Dermatol. 2006;126:1292-6). |
Dr. Tosti said that she had no relevant disclosures.
ORLANDO - Dermoscopy can help with the diagnosis and management of hair and scale disorders.
In some instances, dermoscopy very rapidly reveal a characteristic sign of a hair or scalp condition. In presentations where dermoscopy only narrows down your differential diagnosis, further work-up with histology might be required, said Dr. Antonella Tosti, professor of clinical dermatology at the University of Miami.
How well do you know your dermoscopy diagnoses?

|
All photos courtesy Dr. Antonella Tosti Normal Scalp. Normally, hairs are arranged in follicular units, with up to three hair shafts from the same orifice. "You also see single hairs coming out from the scalp," Dr. Tosti said. Most hair disorders feature a high number of single hairs. A normal vascular pattern presents as interfollicular red loops and arborizing red lines. |

|
Psoriasis. Twisted, coiled loops are a sign of scalp psoriasis. Folliculitis decalvans is the only other scalp condition with a similar loop presentation. If you observe white scales, use high-magnification dermoscopy to look at the vessels before diagnosing psoriasis. White scales can also be seen in cicatricial alopecias such as lupus erythematosus and lichen planopilaris. "So you may need to take a biopsy when patient presents with wide, white scales. It's not always psoriasis," Dr. Tosti said. |

|
Alopecia areata. Alopecia areata and androgenetic alopecia each will feature yellow dots that correspond to follicular openings. "How often you see the yellow dots depends on the patients. They are seen often in [whites] and less commonly seen in darker-skinned patients," Dr. Tosti said. Hair diameter variability, a sign of androgenetic alopecia, can help you to distinguish these conditions. More than 20% variability in shaft thickness and an increase in the number of thinner hairs with advanced disease are diagnostic. "My suggestion is always use your dermatoscope when women complain of hair loss," Dr. Tosti said. "It is an easy and quick way to distinguish androgenetic alopecia." |

|
Discoid lupus erythematosus. A follicular red dot pattern is characteristic of this condition on dermoscopy. An atrophic epidermis can be seen in active lesions. The red dots are important to recognize because, if treated, hair can regrow in the affected area. "Make the diagnosis early and treat the patient immediately," Dr. Tosti said. Also, the red dot pattern will help you to distinguish this disorder from other conditions that can cause cicatricial alopecia. |

|
Cicatricial alopecia. A loss of follicular openings is an important clue on dermoscopy that indicates cicatricial alopecias |

|
Traction alopecia. Hair casts are a useful dermoscopy clue for this condition. "You can see them immediately," Dr. Tosti said. "Hair casts are also useful in African Americans if their hairstyle is associated with traction, even if they don't have alopecia. Sometimes they will deny a traction hairstyle." |

|
Tinea capitis. Dermoscopy facilitates this diagnosis. Look for comma-shaped hairs, a very specific sign seen in both ectothrix and endothrix T. capitis infections. "This is something you should look for in children. It's very fast," Dr. Tosti said. The comma hairs will be more numerous in recent infections, she added. |

|
Monilethrix. Dermoscopy can help you diagnose this condition very, very fast, Dr. Tosti said. The condition features beading of the hair shafts. The autosomal recessive form of monilethrix will appear much more severe on dermoscopy (J. Invest. Dermatol. 2006;126:1292-6). |
Dr. Tosti said that she had no relevant disclosures.
FROM THE ANNUAL MEETING OF THE FLORIDA SOCIETY OF DERMATOLOGIC SURGEONS
Picture Review: Dermoscopy Distinguishes Hair Disorders
ORLANDO - Dermoscopy can help with the diagnosis and management of hair and scale disorders.
In some instances, dermoscopy very rapidly reveal a characteristic sign of a hair or scalp condition. In presentations where dermoscopy only narrows down your differential diagnosis, further work-up with histology might be required, said Dr. Antonella Tosti, professor of clinical dermatology at the University of Miami.
How well do you know your dermoscopy diagnoses?

|
All photos courtesy Dr. Antonella Tosti Normal Scalp. Normally, hairs are arranged in follicular units, with up to three hair shafts from the same orifice. "You also see single hairs coming out from the scalp," Dr. Tosti said. Most hair disorders feature a high number of single hairs. A normal vascular pattern presents as interfollicular red loops and arborizing red lines. |
|
Psoriasis. Twisted, coiled loops are a sign of scalp psoriasis. Folliculitis decalvans is the only other scalp condition with a similar loop presentation. If you observe white scales, use high-magnification dermoscopy to look at the vessels before diagnosing psoriasis. White scales can also be seen in cicatricial alopecias such as lupus erythematosus and lichen planopilaris. "So you may need to take a biopsy when patient presents with wide, white scales. It's not always psoriasis," Dr. Tosti said. |
|
Alopecia areata. Alopecia areata and androgenetic alopecia each will feature yellow dots that correspond to follicular openings. "How often you see the yellow dots depends on the patients. They are seen often in [whites] and less commonly seen in darker-skinned patients," Dr. Tosti said. Hair diameter variability, a sign of androgenetic alopecia, can help you to distinguish these conditions. More than 20% variability in shaft thickness and an increase in the number of thinner hairs with advanced disease are diagnostic. "My suggestion is always use your dermatoscope when women complain of hair loss," Dr. Tosti said. "It is an easy and quick way to distinguish androgenetic alopecia." |
|
Discoid lupus erythematosus. A follicular red dot pattern is characteristic of this condition on dermoscopy. An atrophic epidermis can be seen in active lesions. The red dots are important to recognize because, if treated, hair can regrow in the affected area. "Make the diagnosis early and treat the patient immediately," Dr. Tosti said. Also, the red dot pattern will help you to distinguish this disorder from other conditions that can cause cicatricial alopecia. |

|
Cicatricial alopecia. A loss of follicular openings is an important clue on dermoscopy that indicates cicatricial alopecias |

|
Traction alopecia. Hair casts are a useful dermoscopy clue for this condition. "You can see them immediately," Dr. Tosti said. "Hair casts are also useful in African Americans if their hairstyle is associated with traction, even if they don't have alopecia. Sometimes they will deny a traction hairstyle." |
|
Tinea capitis. Dermoscopy facilitates this diagnosis. Look for comma-shaped hairs, a very specific sign seen in both ectothrix and endothrix T. capitis infections. "This is something you should look for in children. It's very fast," Dr. Tosti said. The comma hairs will be more numerous in recent infections, she added. |
|
Monilethrix. Dermoscopy can help you diagnose this condition very, very fast, Dr. Tosti said. The condition features beading of the hair shafts. The autosomal recessive form of monilethrix will appear much more severe on dermoscopy (J. Invest. Dermatol. 2006;126:1292-6). |
Dr. Tosti said that she had no relevant disclosures.
ORLANDO - Dermoscopy can help with the diagnosis and management of hair and scale disorders.
In some instances, dermoscopy very rapidly reveal a characteristic sign of a hair or scalp condition. In presentations where dermoscopy only narrows down your differential diagnosis, further work-up with histology might be required, said Dr. Antonella Tosti, professor of clinical dermatology at the University of Miami.
How well do you know your dermoscopy diagnoses?

|
All photos courtesy Dr. Antonella Tosti Normal Scalp. Normally, hairs are arranged in follicular units, with up to three hair shafts from the same orifice. "You also see single hairs coming out from the scalp," Dr. Tosti said. Most hair disorders feature a high number of single hairs. A normal vascular pattern presents as interfollicular red loops and arborizing red lines. |

|
Psoriasis. Twisted, coiled loops are a sign of scalp psoriasis. Folliculitis decalvans is the only other scalp condition with a similar loop presentation. If you observe white scales, use high-magnification dermoscopy to look at the vessels before diagnosing psoriasis. White scales can also be seen in cicatricial alopecias such as lupus erythematosus and lichen planopilaris. "So you may need to take a biopsy when patient presents with wide, white scales. It's not always psoriasis," Dr. Tosti said. |

|
Alopecia areata. Alopecia areata and androgenetic alopecia each will feature yellow dots that correspond to follicular openings. "How often you see the yellow dots depends on the patients. They are seen often in [whites] and less commonly seen in darker-skinned patients," Dr. Tosti said. Hair diameter variability, a sign of androgenetic alopecia, can help you to distinguish these conditions. More than 20% variability in shaft thickness and an increase in the number of thinner hairs with advanced disease are diagnostic. "My suggestion is always use your dermatoscope when women complain of hair loss," Dr. Tosti said. "It is an easy and quick way to distinguish androgenetic alopecia." |

|
Discoid lupus erythematosus. A follicular red dot pattern is characteristic of this condition on dermoscopy. An atrophic epidermis can be seen in active lesions. The red dots are important to recognize because, if treated, hair can regrow in the affected area. "Make the diagnosis early and treat the patient immediately," Dr. Tosti said. Also, the red dot pattern will help you to distinguish this disorder from other conditions that can cause cicatricial alopecia. |

|
Cicatricial alopecia. A loss of follicular openings is an important clue on dermoscopy that indicates cicatricial alopecias |

|
Traction alopecia. Hair casts are a useful dermoscopy clue for this condition. "You can see them immediately," Dr. Tosti said. "Hair casts are also useful in African Americans if their hairstyle is associated with traction, even if they don't have alopecia. Sometimes they will deny a traction hairstyle." |

|
Tinea capitis. Dermoscopy facilitates this diagnosis. Look for comma-shaped hairs, a very specific sign seen in both ectothrix and endothrix T. capitis infections. "This is something you should look for in children. It's very fast," Dr. Tosti said. The comma hairs will be more numerous in recent infections, she added. |

|
Monilethrix. Dermoscopy can help you diagnose this condition very, very fast, Dr. Tosti said. The condition features beading of the hair shafts. The autosomal recessive form of monilethrix will appear much more severe on dermoscopy (J. Invest. Dermatol. 2006;126:1292-6). |
Dr. Tosti said that she had no relevant disclosures.
ORLANDO - Dermoscopy can help with the diagnosis and management of hair and scale disorders.
In some instances, dermoscopy very rapidly reveal a characteristic sign of a hair or scalp condition. In presentations where dermoscopy only narrows down your differential diagnosis, further work-up with histology might be required, said Dr. Antonella Tosti, professor of clinical dermatology at the University of Miami.
How well do you know your dermoscopy diagnoses?

|
All photos courtesy Dr. Antonella Tosti Normal Scalp. Normally, hairs are arranged in follicular units, with up to three hair shafts from the same orifice. "You also see single hairs coming out from the scalp," Dr. Tosti said. Most hair disorders feature a high number of single hairs. A normal vascular pattern presents as interfollicular red loops and arborizing red lines. |

|
Psoriasis. Twisted, coiled loops are a sign of scalp psoriasis. Folliculitis decalvans is the only other scalp condition with a similar loop presentation. If you observe white scales, use high-magnification dermoscopy to look at the vessels before diagnosing psoriasis. White scales can also be seen in cicatricial alopecias such as lupus erythematosus and lichen planopilaris. "So you may need to take a biopsy when patient presents with wide, white scales. It's not always psoriasis," Dr. Tosti said. |

|
Alopecia areata. Alopecia areata and androgenetic alopecia each will feature yellow dots that correspond to follicular openings. "How often you see the yellow dots depends on the patients. They are seen often in [whites] and less commonly seen in darker-skinned patients," Dr. Tosti said. Hair diameter variability, a sign of androgenetic alopecia, can help you to distinguish these conditions. More than 20% variability in shaft thickness and an increase in the number of thinner hairs with advanced disease are diagnostic. "My suggestion is always use your dermatoscope when women complain of hair loss," Dr. Tosti said. "It is an easy and quick way to distinguish androgenetic alopecia." |

|
Discoid lupus erythematosus. A follicular red dot pattern is characteristic of this condition on dermoscopy. An atrophic epidermis can be seen in active lesions. The red dots are important to recognize because, if treated, hair can regrow in the affected area. "Make the diagnosis early and treat the patient immediately," Dr. Tosti said. Also, the red dot pattern will help you to distinguish this disorder from other conditions that can cause cicatricial alopecia. |

|
Cicatricial alopecia. A loss of follicular openings is an important clue on dermoscopy that indicates cicatricial alopecias |

|
Traction alopecia. Hair casts are a useful dermoscopy clue for this condition. "You can see them immediately," Dr. Tosti said. "Hair casts are also useful in African Americans if their hairstyle is associated with traction, even if they don't have alopecia. Sometimes they will deny a traction hairstyle." |

|
Tinea capitis. Dermoscopy facilitates this diagnosis. Look for comma-shaped hairs, a very specific sign seen in both ectothrix and endothrix T. capitis infections. "This is something you should look for in children. It's very fast," Dr. Tosti said. The comma hairs will be more numerous in recent infections, she added. |

|
Monilethrix. Dermoscopy can help you diagnose this condition very, very fast, Dr. Tosti said. The condition features beading of the hair shafts. The autosomal recessive form of monilethrix will appear much more severe on dermoscopy (J. Invest. Dermatol. 2006;126:1292-6). |
Dr. Tosti said that she had no relevant disclosures.
FROM THE ANNUAL MEETING OF THE FLORIDA SOCIETY OF DERMATOLOGIC SURGEONS
Study Suggests Wider Role for Neoadjuvant Chemo in Ovarian Cancer
ORLANDO – Neoadjuvant chemotherapy with interval debulking surgery yields similar overall and longer progression-free survival when compared with primary debulking surgery for women with stage IV epithelial ovarian cancer, according to a retrospective study.
"Our study suggests a potential wider role for neoadjuvant chemotherapy in management of stage IV ovarian cancer," Dr. Jose Alejandro Rauh-Hain said at the annual meeting of the Society of Gynecologic Oncologists.
Dr. Rauh-Hain and his associates assessed 221 newly diagnosed women with stage IV ovarian cancer. Overall survival was not significantly different at a median of 33 months with neoadjuvant chemotherapy-interval debulking surgery group (NACT-IDS) and 29 months with primary debulking surgery, said Dr. Rauh-Hain, a clinical fellow in obstetrics and gynecology at Massachusetts General Hospital and Brigham and Women’s Hospital, both in Boston.
Median progression-free survival was 14 months for the 45 women in the NACT-IDS group, compared with 10 months among the 176 women treated with primary debulking only.
Patient records were evaluated to subclassify stage IV disease, site of tumor, and dissemination at time of initial diagnosis. All women received primary therapy from Jan. 1, 1995, to Dec. 31, 2007, at either of the two institutions.
The strengths of the study are a large number of patients, treatment based on physician discretion, and an optimal debulking surgery rate of 58%, which is similar to other studies, said invited discussant Dr. Peter Rose. A retrospective design; an unbalanced comparison (because the majority had primary debulking surgery); and a lack of uniform criteria to select neoadjuvant chemotherapy are limitations.
"It seems like we are mixing a lot of different tomatoes, big red ones and small green ones, and what we are getting is tomato sauce," said Dr. Rose, section head, department of obstetrics and gynecology at the Cleveland Clinic in Ohio.
Optimal cytoreduction, defined as residual disease smaller than 1 cm, was not significantly different between groups: 71% of the NACT-IDS and 58% of the primary debulking cohorts (P = .1).
The rate of complete resection to no residual disease was significantly higher among women who underwent NACT-IDS, 27%, vs. 7.5% of women treated with primary debulking (P less than .001).
Median follow-up was 28 months. The longest median overall survival was observed for women who had primary debulking surgery and no residual disease (72 months), but Dr. Rauh-Hain pointed out that "only 7.5% of these patients who underwent primary debulking had no residual disease." This success rate is similar to 8% of 360 women debulked to no residual disease in another report ( J. Clin. Oncol. 2008;26:83-9).
Median overall survival reached 32 months for the primary surgery group who had optimal cytoreduction and to 20 months if cytoreduction was suboptimal.
Dr. Rauh-Hain and his associates also evaluated median progression-free and overall survival rates by site of distant metastatic disease, including pleural effusion, liver, abdomen, distant lymph nodes, and spleen. The only significant difference was in median overall survival among women who had liver metastases and NACT-IDS, 43 months vs. 27 months with primary debulking. Median progression-free survival in this group was 15 months vs. 13 months.
The superior overall survival with NACT-IDS among women who presented with parenchymal liver metastases suggests this therapeutic strategy may be the preferred option for these patients, Dr. Rauh-Hain said.
The NACT-IDS group had a shorter mean length of hospital stay, 8 days vs. 12 days in the primary debulking surgery group. There also was a trend toward fewer postoperative complications in the NACT-IDS cohort, 15% vs. 27%. Also, no woman in the NACT-IDS group died within 30 days of their initial surgery, compared with eight women (5%) of the primary debulking surgery group.
Possible limitations were the retrospective design of the study; lack of information regarding previous or subsequent care at nonaffiliated institutions; and a lack of standardized treatment were possible limitations, Dr. Rauh-Hain said. A lower postoperative mortality rate in the NACT-IDS group suggests there could be selection bias in terms of treatment selection, he added.
Dr. Rauh-Hain said that he had no relevant disclosures. Dr. Rose said he is a member of the Lilly speakers bureau.
ORLANDO – Neoadjuvant chemotherapy with interval debulking surgery yields similar overall and longer progression-free survival when compared with primary debulking surgery for women with stage IV epithelial ovarian cancer, according to a retrospective study.
"Our study suggests a potential wider role for neoadjuvant chemotherapy in management of stage IV ovarian cancer," Dr. Jose Alejandro Rauh-Hain said at the annual meeting of the Society of Gynecologic Oncologists.
Dr. Rauh-Hain and his associates assessed 221 newly diagnosed women with stage IV ovarian cancer. Overall survival was not significantly different at a median of 33 months with neoadjuvant chemotherapy-interval debulking surgery group (NACT-IDS) and 29 months with primary debulking surgery, said Dr. Rauh-Hain, a clinical fellow in obstetrics and gynecology at Massachusetts General Hospital and Brigham and Women’s Hospital, both in Boston.
Median progression-free survival was 14 months for the 45 women in the NACT-IDS group, compared with 10 months among the 176 women treated with primary debulking only.
Patient records were evaluated to subclassify stage IV disease, site of tumor, and dissemination at time of initial diagnosis. All women received primary therapy from Jan. 1, 1995, to Dec. 31, 2007, at either of the two institutions.
The strengths of the study are a large number of patients, treatment based on physician discretion, and an optimal debulking surgery rate of 58%, which is similar to other studies, said invited discussant Dr. Peter Rose. A retrospective design; an unbalanced comparison (because the majority had primary debulking surgery); and a lack of uniform criteria to select neoadjuvant chemotherapy are limitations.
"It seems like we are mixing a lot of different tomatoes, big red ones and small green ones, and what we are getting is tomato sauce," said Dr. Rose, section head, department of obstetrics and gynecology at the Cleveland Clinic in Ohio.
Optimal cytoreduction, defined as residual disease smaller than 1 cm, was not significantly different between groups: 71% of the NACT-IDS and 58% of the primary debulking cohorts (P = .1).
The rate of complete resection to no residual disease was significantly higher among women who underwent NACT-IDS, 27%, vs. 7.5% of women treated with primary debulking (P less than .001).
Median follow-up was 28 months. The longest median overall survival was observed for women who had primary debulking surgery and no residual disease (72 months), but Dr. Rauh-Hain pointed out that "only 7.5% of these patients who underwent primary debulking had no residual disease." This success rate is similar to 8% of 360 women debulked to no residual disease in another report ( J. Clin. Oncol. 2008;26:83-9).
Median overall survival reached 32 months for the primary surgery group who had optimal cytoreduction and to 20 months if cytoreduction was suboptimal.
Dr. Rauh-Hain and his associates also evaluated median progression-free and overall survival rates by site of distant metastatic disease, including pleural effusion, liver, abdomen, distant lymph nodes, and spleen. The only significant difference was in median overall survival among women who had liver metastases and NACT-IDS, 43 months vs. 27 months with primary debulking. Median progression-free survival in this group was 15 months vs. 13 months.
The superior overall survival with NACT-IDS among women who presented with parenchymal liver metastases suggests this therapeutic strategy may be the preferred option for these patients, Dr. Rauh-Hain said.
The NACT-IDS group had a shorter mean length of hospital stay, 8 days vs. 12 days in the primary debulking surgery group. There also was a trend toward fewer postoperative complications in the NACT-IDS cohort, 15% vs. 27%. Also, no woman in the NACT-IDS group died within 30 days of their initial surgery, compared with eight women (5%) of the primary debulking surgery group.
Possible limitations were the retrospective design of the study; lack of information regarding previous or subsequent care at nonaffiliated institutions; and a lack of standardized treatment were possible limitations, Dr. Rauh-Hain said. A lower postoperative mortality rate in the NACT-IDS group suggests there could be selection bias in terms of treatment selection, he added.
Dr. Rauh-Hain said that he had no relevant disclosures. Dr. Rose said he is a member of the Lilly speakers bureau.
ORLANDO – Neoadjuvant chemotherapy with interval debulking surgery yields similar overall and longer progression-free survival when compared with primary debulking surgery for women with stage IV epithelial ovarian cancer, according to a retrospective study.
"Our study suggests a potential wider role for neoadjuvant chemotherapy in management of stage IV ovarian cancer," Dr. Jose Alejandro Rauh-Hain said at the annual meeting of the Society of Gynecologic Oncologists.
Dr. Rauh-Hain and his associates assessed 221 newly diagnosed women with stage IV ovarian cancer. Overall survival was not significantly different at a median of 33 months with neoadjuvant chemotherapy-interval debulking surgery group (NACT-IDS) and 29 months with primary debulking surgery, said Dr. Rauh-Hain, a clinical fellow in obstetrics and gynecology at Massachusetts General Hospital and Brigham and Women’s Hospital, both in Boston.
Median progression-free survival was 14 months for the 45 women in the NACT-IDS group, compared with 10 months among the 176 women treated with primary debulking only.
Patient records were evaluated to subclassify stage IV disease, site of tumor, and dissemination at time of initial diagnosis. All women received primary therapy from Jan. 1, 1995, to Dec. 31, 2007, at either of the two institutions.
The strengths of the study are a large number of patients, treatment based on physician discretion, and an optimal debulking surgery rate of 58%, which is similar to other studies, said invited discussant Dr. Peter Rose. A retrospective design; an unbalanced comparison (because the majority had primary debulking surgery); and a lack of uniform criteria to select neoadjuvant chemotherapy are limitations.
"It seems like we are mixing a lot of different tomatoes, big red ones and small green ones, and what we are getting is tomato sauce," said Dr. Rose, section head, department of obstetrics and gynecology at the Cleveland Clinic in Ohio.
Optimal cytoreduction, defined as residual disease smaller than 1 cm, was not significantly different between groups: 71% of the NACT-IDS and 58% of the primary debulking cohorts (P = .1).
The rate of complete resection to no residual disease was significantly higher among women who underwent NACT-IDS, 27%, vs. 7.5% of women treated with primary debulking (P less than .001).
Median follow-up was 28 months. The longest median overall survival was observed for women who had primary debulking surgery and no residual disease (72 months), but Dr. Rauh-Hain pointed out that "only 7.5% of these patients who underwent primary debulking had no residual disease." This success rate is similar to 8% of 360 women debulked to no residual disease in another report ( J. Clin. Oncol. 2008;26:83-9).
Median overall survival reached 32 months for the primary surgery group who had optimal cytoreduction and to 20 months if cytoreduction was suboptimal.
Dr. Rauh-Hain and his associates also evaluated median progression-free and overall survival rates by site of distant metastatic disease, including pleural effusion, liver, abdomen, distant lymph nodes, and spleen. The only significant difference was in median overall survival among women who had liver metastases and NACT-IDS, 43 months vs. 27 months with primary debulking. Median progression-free survival in this group was 15 months vs. 13 months.
The superior overall survival with NACT-IDS among women who presented with parenchymal liver metastases suggests this therapeutic strategy may be the preferred option for these patients, Dr. Rauh-Hain said.
The NACT-IDS group had a shorter mean length of hospital stay, 8 days vs. 12 days in the primary debulking surgery group. There also was a trend toward fewer postoperative complications in the NACT-IDS cohort, 15% vs. 27%. Also, no woman in the NACT-IDS group died within 30 days of their initial surgery, compared with eight women (5%) of the primary debulking surgery group.
Possible limitations were the retrospective design of the study; lack of information regarding previous or subsequent care at nonaffiliated institutions; and a lack of standardized treatment were possible limitations, Dr. Rauh-Hain said. A lower postoperative mortality rate in the NACT-IDS group suggests there could be selection bias in terms of treatment selection, he added.
Dr. Rauh-Hain said that he had no relevant disclosures. Dr. Rose said he is a member of the Lilly speakers bureau.
FROM THE ANNUAL MEETING OF THE SOCIETY OF GYNECOLOGIC ONCOLOGISTS
Study Suggests Wider Role for Neoadjuvant Chemo in Ovarian Cancer
ORLANDO – Neoadjuvant chemotherapy with interval debulking surgery yields similar overall and longer progression-free survival when compared with primary debulking surgery for women with stage IV epithelial ovarian cancer, according to a retrospective study.
"Our study suggests a potential wider role for neoadjuvant chemotherapy in management of stage IV ovarian cancer," Dr. Jose Alejandro Rauh-Hain said at the annual meeting of the Society of Gynecologic Oncologists.
Dr. Rauh-Hain and his associates assessed 221 newly diagnosed women with stage IV ovarian cancer. Overall survival was not significantly different at a median of 33 months with neoadjuvant chemotherapy-interval debulking surgery group (NACT-IDS) and 29 months with primary debulking surgery, said Dr. Rauh-Hain, a clinical fellow in obstetrics and gynecology at Massachusetts General Hospital and Brigham and Women’s Hospital, both in Boston.
Median progression-free survival was 14 months for the 45 women in the NACT-IDS group, compared with 10 months among the 176 women treated with primary debulking only.
Patient records were evaluated to subclassify stage IV disease, site of tumor, and dissemination at time of initial diagnosis. All women received primary therapy from Jan. 1, 1995, to Dec. 31, 2007, at either of the two institutions.
The strengths of the study are a large number of patients, treatment based on physician discretion, and an optimal debulking surgery rate of 58%, which is similar to other studies, said invited discussant Dr. Peter Rose. A retrospective design; an unbalanced comparison (because the majority had primary debulking surgery); and a lack of uniform criteria to select neoadjuvant chemotherapy are limitations.
"It seems like we are mixing a lot of different tomatoes, big red ones and small green ones, and what we are getting is tomato sauce," said Dr. Rose, section head, department of obstetrics and gynecology at the Cleveland Clinic in Ohio.
Optimal cytoreduction, defined as residual disease smaller than 1 cm, was not significantly different between groups: 71% of the NACT-IDS and 58% of the primary debulking cohorts (P = .1).
The rate of complete resection to no residual disease was significantly higher among women who underwent NACT-IDS, 27%, vs. 7.5% of women treated with primary debulking (P less than .001).
Median follow-up was 28 months. The longest median overall survival was observed for women who had primary debulking surgery and no residual disease (72 months), but Dr. Rauh-Hain pointed out that "only 7.5% of these patients who underwent primary debulking had no residual disease." This success rate is similar to 8% of 360 women debulked to no residual disease in another report ( J. Clin. Oncol. 2008;26:83-9).
Median overall survival reached 32 months for the primary surgery group who had optimal cytoreduction and to 20 months if cytoreduction was suboptimal.
Dr. Rauh-Hain and his associates also evaluated median progression-free and overall survival rates by site of distant metastatic disease, including pleural effusion, liver, abdomen, distant lymph nodes, and spleen. The only significant difference was in median overall survival among women who had liver metastases and NACT-IDS, 43 months vs. 27 months with primary debulking. Median progression-free survival in this group was 15 months vs. 13 months.
The superior overall survival with NACT-IDS among women who presented with parenchymal liver metastases suggests this therapeutic strategy may be the preferred option for these patients, Dr. Rauh-Hain said.
The NACT-IDS group had a shorter mean length of hospital stay, 8 days vs. 12 days in the primary debulking surgery group. There also was a trend toward fewer postoperative complications in the NACT-IDS cohort, 15% vs. 27%. Also, no woman in the NACT-IDS group died within 30 days of their initial surgery, compared with eight women (5%) of the primary debulking surgery group.
Possible limitations were the retrospective design of the study; lack of information regarding previous or subsequent care at nonaffiliated institutions; and a lack of standardized treatment were possible limitations, Dr. Rauh-Hain said. A lower postoperative mortality rate in the NACT-IDS group suggests there could be selection bias in terms of treatment selection, he added.
Dr. Rauh-Hain said that he had no relevant disclosures. Dr. Rose said he is a member of the Lilly speakers bureau.
ORLANDO – Neoadjuvant chemotherapy with interval debulking surgery yields similar overall and longer progression-free survival when compared with primary debulking surgery for women with stage IV epithelial ovarian cancer, according to a retrospective study.
"Our study suggests a potential wider role for neoadjuvant chemotherapy in management of stage IV ovarian cancer," Dr. Jose Alejandro Rauh-Hain said at the annual meeting of the Society of Gynecologic Oncologists.
Dr. Rauh-Hain and his associates assessed 221 newly diagnosed women with stage IV ovarian cancer. Overall survival was not significantly different at a median of 33 months with neoadjuvant chemotherapy-interval debulking surgery group (NACT-IDS) and 29 months with primary debulking surgery, said Dr. Rauh-Hain, a clinical fellow in obstetrics and gynecology at Massachusetts General Hospital and Brigham and Women’s Hospital, both in Boston.
Median progression-free survival was 14 months for the 45 women in the NACT-IDS group, compared with 10 months among the 176 women treated with primary debulking only.
Patient records were evaluated to subclassify stage IV disease, site of tumor, and dissemination at time of initial diagnosis. All women received primary therapy from Jan. 1, 1995, to Dec. 31, 2007, at either of the two institutions.
The strengths of the study are a large number of patients, treatment based on physician discretion, and an optimal debulking surgery rate of 58%, which is similar to other studies, said invited discussant Dr. Peter Rose. A retrospective design; an unbalanced comparison (because the majority had primary debulking surgery); and a lack of uniform criteria to select neoadjuvant chemotherapy are limitations.
"It seems like we are mixing a lot of different tomatoes, big red ones and small green ones, and what we are getting is tomato sauce," said Dr. Rose, section head, department of obstetrics and gynecology at the Cleveland Clinic in Ohio.
Optimal cytoreduction, defined as residual disease smaller than 1 cm, was not significantly different between groups: 71% of the NACT-IDS and 58% of the primary debulking cohorts (P = .1).
The rate of complete resection to no residual disease was significantly higher among women who underwent NACT-IDS, 27%, vs. 7.5% of women treated with primary debulking (P less than .001).
Median follow-up was 28 months. The longest median overall survival was observed for women who had primary debulking surgery and no residual disease (72 months), but Dr. Rauh-Hain pointed out that "only 7.5% of these patients who underwent primary debulking had no residual disease." This success rate is similar to 8% of 360 women debulked to no residual disease in another report ( J. Clin. Oncol. 2008;26:83-9).
Median overall survival reached 32 months for the primary surgery group who had optimal cytoreduction and to 20 months if cytoreduction was suboptimal.
Dr. Rauh-Hain and his associates also evaluated median progression-free and overall survival rates by site of distant metastatic disease, including pleural effusion, liver, abdomen, distant lymph nodes, and spleen. The only significant difference was in median overall survival among women who had liver metastases and NACT-IDS, 43 months vs. 27 months with primary debulking. Median progression-free survival in this group was 15 months vs. 13 months.
The superior overall survival with NACT-IDS among women who presented with parenchymal liver metastases suggests this therapeutic strategy may be the preferred option for these patients, Dr. Rauh-Hain said.
The NACT-IDS group had a shorter mean length of hospital stay, 8 days vs. 12 days in the primary debulking surgery group. There also was a trend toward fewer postoperative complications in the NACT-IDS cohort, 15% vs. 27%. Also, no woman in the NACT-IDS group died within 30 days of their initial surgery, compared with eight women (5%) of the primary debulking surgery group.
Possible limitations were the retrospective design of the study; lack of information regarding previous or subsequent care at nonaffiliated institutions; and a lack of standardized treatment were possible limitations, Dr. Rauh-Hain said. A lower postoperative mortality rate in the NACT-IDS group suggests there could be selection bias in terms of treatment selection, he added.
Dr. Rauh-Hain said that he had no relevant disclosures. Dr. Rose said he is a member of the Lilly speakers bureau.
ORLANDO – Neoadjuvant chemotherapy with interval debulking surgery yields similar overall and longer progression-free survival when compared with primary debulking surgery for women with stage IV epithelial ovarian cancer, according to a retrospective study.
"Our study suggests a potential wider role for neoadjuvant chemotherapy in management of stage IV ovarian cancer," Dr. Jose Alejandro Rauh-Hain said at the annual meeting of the Society of Gynecologic Oncologists.
Dr. Rauh-Hain and his associates assessed 221 newly diagnosed women with stage IV ovarian cancer. Overall survival was not significantly different at a median of 33 months with neoadjuvant chemotherapy-interval debulking surgery group (NACT-IDS) and 29 months with primary debulking surgery, said Dr. Rauh-Hain, a clinical fellow in obstetrics and gynecology at Massachusetts General Hospital and Brigham and Women’s Hospital, both in Boston.
Median progression-free survival was 14 months for the 45 women in the NACT-IDS group, compared with 10 months among the 176 women treated with primary debulking only.
Patient records were evaluated to subclassify stage IV disease, site of tumor, and dissemination at time of initial diagnosis. All women received primary therapy from Jan. 1, 1995, to Dec. 31, 2007, at either of the two institutions.
The strengths of the study are a large number of patients, treatment based on physician discretion, and an optimal debulking surgery rate of 58%, which is similar to other studies, said invited discussant Dr. Peter Rose. A retrospective design; an unbalanced comparison (because the majority had primary debulking surgery); and a lack of uniform criteria to select neoadjuvant chemotherapy are limitations.
"It seems like we are mixing a lot of different tomatoes, big red ones and small green ones, and what we are getting is tomato sauce," said Dr. Rose, section head, department of obstetrics and gynecology at the Cleveland Clinic in Ohio.
Optimal cytoreduction, defined as residual disease smaller than 1 cm, was not significantly different between groups: 71% of the NACT-IDS and 58% of the primary debulking cohorts (P = .1).
The rate of complete resection to no residual disease was significantly higher among women who underwent NACT-IDS, 27%, vs. 7.5% of women treated with primary debulking (P less than .001).
Median follow-up was 28 months. The longest median overall survival was observed for women who had primary debulking surgery and no residual disease (72 months), but Dr. Rauh-Hain pointed out that "only 7.5% of these patients who underwent primary debulking had no residual disease." This success rate is similar to 8% of 360 women debulked to no residual disease in another report ( J. Clin. Oncol. 2008;26:83-9).
Median overall survival reached 32 months for the primary surgery group who had optimal cytoreduction and to 20 months if cytoreduction was suboptimal.
Dr. Rauh-Hain and his associates also evaluated median progression-free and overall survival rates by site of distant metastatic disease, including pleural effusion, liver, abdomen, distant lymph nodes, and spleen. The only significant difference was in median overall survival among women who had liver metastases and NACT-IDS, 43 months vs. 27 months with primary debulking. Median progression-free survival in this group was 15 months vs. 13 months.
The superior overall survival with NACT-IDS among women who presented with parenchymal liver metastases suggests this therapeutic strategy may be the preferred option for these patients, Dr. Rauh-Hain said.
The NACT-IDS group had a shorter mean length of hospital stay, 8 days vs. 12 days in the primary debulking surgery group. There also was a trend toward fewer postoperative complications in the NACT-IDS cohort, 15% vs. 27%. Also, no woman in the NACT-IDS group died within 30 days of their initial surgery, compared with eight women (5%) of the primary debulking surgery group.
Possible limitations were the retrospective design of the study; lack of information regarding previous or subsequent care at nonaffiliated institutions; and a lack of standardized treatment were possible limitations, Dr. Rauh-Hain said. A lower postoperative mortality rate in the NACT-IDS group suggests there could be selection bias in terms of treatment selection, he added.
Dr. Rauh-Hain said that he had no relevant disclosures. Dr. Rose said he is a member of the Lilly speakers bureau.
FROM THE ANNUAL MEETING OF THE SOCIETY OF GYNECOLOGIC ONCOLOGISTS
Major Finding: Overall survival was not significantly different at a median of 33 months with neoadjuvant chemotherapy-interval debulking surgery group (NACT-IDS) and 29 months with primary debulking surgery,
Data Source: Retrospective study of 221 women with newly diagnosed advanced ovarian cancer treated between 1995 and 2007.
Disclosures: Dr. Jose Alejandro Rauh-Hain said he had no disclosures. Dr. Peter Rose is a member of the speaker’s bureau for Lilly.



