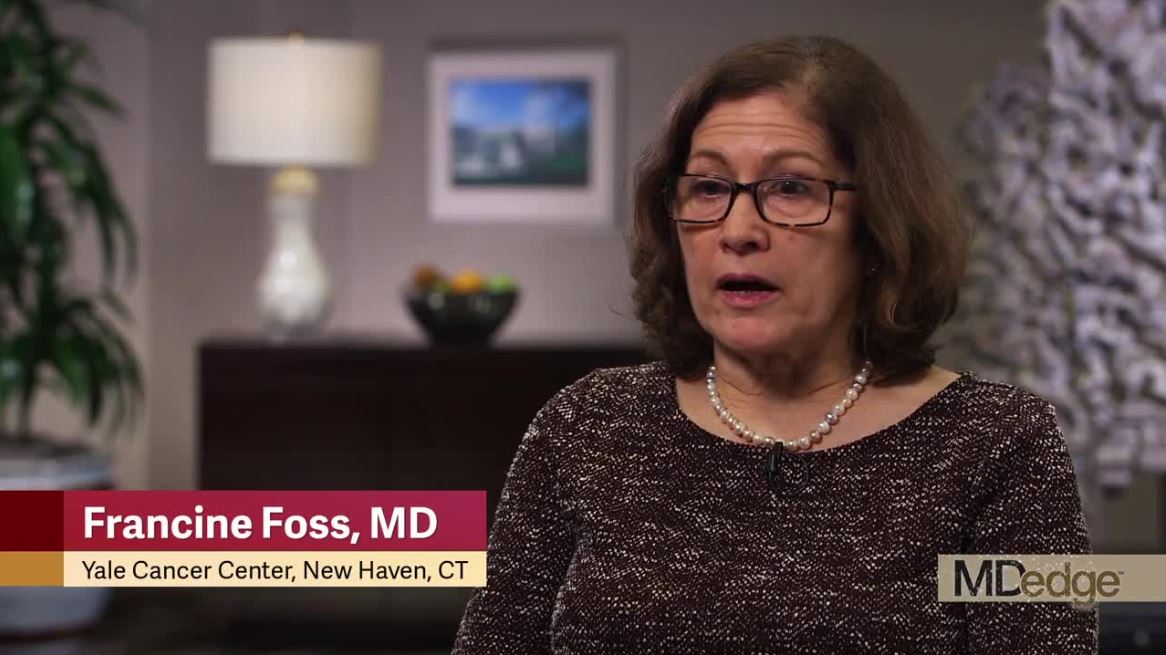User login
How to surf the rosacea treatment algorithm
WAIKOLOA, HAWAII – according to Linda Stein Gold, MD, director of dermatology research at Henry Ford Hospital in Detroit.

Papules and pustules need an oral or topical anti-inflammatory drug. Background erythema requires an alpha adrenergic agonist. Telangiectasia is best handled by a laser device, and if a patient has a phyma, “you’ve got to use a surgical approach,” she said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation. It sounds simple, but there are decisions to be made about what drugs and formulations to use, and when, and when to combine them.
In an interview, Dr. Stein Gold shared her approach to treatment, along with the latest on using ivermectin and brimonidine together, plus her thoughts on new medications under development and the role of the Demodex mite in rosacea.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – according to Linda Stein Gold, MD, director of dermatology research at Henry Ford Hospital in Detroit.

Papules and pustules need an oral or topical anti-inflammatory drug. Background erythema requires an alpha adrenergic agonist. Telangiectasia is best handled by a laser device, and if a patient has a phyma, “you’ve got to use a surgical approach,” she said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation. It sounds simple, but there are decisions to be made about what drugs and formulations to use, and when, and when to combine them.
In an interview, Dr. Stein Gold shared her approach to treatment, along with the latest on using ivermectin and brimonidine together, plus her thoughts on new medications under development and the role of the Demodex mite in rosacea.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – according to Linda Stein Gold, MD, director of dermatology research at Henry Ford Hospital in Detroit.

Papules and pustules need an oral or topical anti-inflammatory drug. Background erythema requires an alpha adrenergic agonist. Telangiectasia is best handled by a laser device, and if a patient has a phyma, “you’ve got to use a surgical approach,” she said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation. It sounds simple, but there are decisions to be made about what drugs and formulations to use, and when, and when to combine them.
In an interview, Dr. Stein Gold shared her approach to treatment, along with the latest on using ivermectin and brimonidine together, plus her thoughts on new medications under development and the role of the Demodex mite in rosacea.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Don’t fear spironolactone, isotretinoin, OCs for acne
WAIKOLOA, HAWAII – There’s really , of the department of dermatology at the University of Alabama at Birmingham.
There have been concerns with all three in the past, but most of the worries have been recently laid to rest.
The news hasn’t reached everyone, though, so, by and large, they are “tools I think we are not using enough of,” Dr. Harper said in an interview. With isotretinoin, for instance, it really isn’t necessary to do blood work for lipids and liver function every month, a daunting prospect for patients; baseline testing with a repeat at 2 months is sufficient, as long as there’s no dose escalation and results are acceptable, with the exception of a monthly pregnancy test for women, she noted. Meanwhile, there’s no evidence of a link with inflammatory bowel disease, and wound healing isn’t as much of an issue as once thought.
It’s the same story with spironolactone. Hyperkalemia is a long-standing concern, but it turns out that “in healthy young women taking spironolactone for acne, we don’t need to be checking potassium.” As far as breast cancer goes, the potential risk with spironolactone hasn’t panned out in the literature, and there may not be “a link at all,” Dr. Harper said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
There are caveats, of course. Hormonal treatments shouldn’t be used in young women until they’ve established their menstrual cycle. OCs should not be used in smokers, or people who have hypertension or migraines, among other conditions. Also, elevated triglycerides remain a concern with isotretinoin. “The number I would want people to remember is 500 [mg/dL],” the threshold when triglycerides become a problem.
In the interview, Dr. Harper explained the new thinking on these three options, and shared her treatment tips, including what to do if patients’ triglycerides hit the 500 mg/dL mark.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – There’s really , of the department of dermatology at the University of Alabama at Birmingham.
There have been concerns with all three in the past, but most of the worries have been recently laid to rest.
The news hasn’t reached everyone, though, so, by and large, they are “tools I think we are not using enough of,” Dr. Harper said in an interview. With isotretinoin, for instance, it really isn’t necessary to do blood work for lipids and liver function every month, a daunting prospect for patients; baseline testing with a repeat at 2 months is sufficient, as long as there’s no dose escalation and results are acceptable, with the exception of a monthly pregnancy test for women, she noted. Meanwhile, there’s no evidence of a link with inflammatory bowel disease, and wound healing isn’t as much of an issue as once thought.
It’s the same story with spironolactone. Hyperkalemia is a long-standing concern, but it turns out that “in healthy young women taking spironolactone for acne, we don’t need to be checking potassium.” As far as breast cancer goes, the potential risk with spironolactone hasn’t panned out in the literature, and there may not be “a link at all,” Dr. Harper said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
There are caveats, of course. Hormonal treatments shouldn’t be used in young women until they’ve established their menstrual cycle. OCs should not be used in smokers, or people who have hypertension or migraines, among other conditions. Also, elevated triglycerides remain a concern with isotretinoin. “The number I would want people to remember is 500 [mg/dL],” the threshold when triglycerides become a problem.
In the interview, Dr. Harper explained the new thinking on these three options, and shared her treatment tips, including what to do if patients’ triglycerides hit the 500 mg/dL mark.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – There’s really , of the department of dermatology at the University of Alabama at Birmingham.
There have been concerns with all three in the past, but most of the worries have been recently laid to rest.
The news hasn’t reached everyone, though, so, by and large, they are “tools I think we are not using enough of,” Dr. Harper said in an interview. With isotretinoin, for instance, it really isn’t necessary to do blood work for lipids and liver function every month, a daunting prospect for patients; baseline testing with a repeat at 2 months is sufficient, as long as there’s no dose escalation and results are acceptable, with the exception of a monthly pregnancy test for women, she noted. Meanwhile, there’s no evidence of a link with inflammatory bowel disease, and wound healing isn’t as much of an issue as once thought.
It’s the same story with spironolactone. Hyperkalemia is a long-standing concern, but it turns out that “in healthy young women taking spironolactone for acne, we don’t need to be checking potassium.” As far as breast cancer goes, the potential risk with spironolactone hasn’t panned out in the literature, and there may not be “a link at all,” Dr. Harper said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
There are caveats, of course. Hormonal treatments shouldn’t be used in young women until they’ve established their menstrual cycle. OCs should not be used in smokers, or people who have hypertension or migraines, among other conditions. Also, elevated triglycerides remain a concern with isotretinoin. “The number I would want people to remember is 500 [mg/dL],” the threshold when triglycerides become a problem.
In the interview, Dr. Harper explained the new thinking on these three options, and shared her treatment tips, including what to do if patients’ triglycerides hit the 500 mg/dL mark.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
February 2019 Highlights
Increased risk of second cancers in mycosis fungoides
LA JOLLA, CALIF. – A retrospective study suggests patients with mycosis fungoides (MF) have an increased risk of developing hematologic and solid tumor malignancies.

Researchers found the risk of second malignancy was highest among MF patients aged 30 to 50 years and patients who had tumor stage or advanced stage MF.
The increased risk was present during the entire period after MF diagnosis, but it was greatest in the first 6 months after diagnosis and roughly a dozen years later.
Amrita Goyal, MD, of the University of Minnesota in Minneapolis, and her colleagues presented these findings at the annual T-cell Lymphoma Forum.
The researchers first assessed the risk of second malignancy in 172 MF patients treated at UMN from 2005 to 2017, comparing this cohort to a control group of 172 patients with seborrheic dermatitis.
Second malignancies occurred in 24 MF patients and three controls, which was a significant difference (P = .0045). The most common second malignancies among the MF patients were melanoma (n = 4), prostate cancer (n = 3), and renal cell carcinoma (n = 3).
Further analyses revealed that MF patients were more likely to develop a second malignancy if they had tumor stage disease (P = .0024) or stage IIB or higher disease (P = .03).
To corroborate and expand upon these results, Dr. Goyal and her colleagues analyzed data from the Surveillance, Epidemiology, and End Results (SEER) database on patients diagnosed with MF from 2000 to 2014.
Among the 6,196 MF patients in this cohort, there were 514 second cancers.
“We found that MF patients were, overall, 10 times more likely to develop a second malignancy [compared with the general population],” Dr. Goyal said.
Specifically, the standardized incidence ratio was 10.15 for all malignancies, 7.33 for solid tumors, and 41.72 for hematologic malignancies.
Standardized incidence ratios for individual malignancies were:
- 69.8 for Hodgkin lymphoma.
- 46.5 for non-Hodgkin lymphoma.
- 8.6 for leukemia.
- 7.2 for melanoma.
- 6.2 for lung cancer.
- 7.9 for female breast cancer.
- 5.2 for colon cancer.
- 4.1 for prostate cancer.
- 3.9 for renal cell carcinoma.
- 3.8 for pancreatic cancer.
- 3.6 for bladder cancer.
“We found there is an increased risk [of second malignancy] during the first 6 months after diagnosis of MF, likely related to patients being in contact with the health care system more,” Dr. Goyal said. “Over time, patients have about a 7- to 10-fold increased risk over baseline, until they reach about 12 or 13 years after diagnosis, at which point, there is an increase in risk.”
The researchers found the greatest risk of second malignancy was among patients aged 30 to 50 years, although there was an increased risk for all age groups.
“The reason we think patients are experiencing an increased risk of cancers is we believe this may be due to immune suppression secondary to the mycosis fungoides, although further studies need to be performed to determine if that’s accurate,” Dr. Goyal said.
To that end, she and her colleagues are planning gene expression studies in patients from the UMN cohort. The researchers plan to examine genes involved in the pathogenesis of second malignancies and MF progression in tissue samples from 36 MF patients, 12 who developed second malignancies and 24 who did not.
The current research was funded by the American Society of Hematology. Dr. Goyal reported having no relevant financial disclosures. The T-cell Lymphoma Forum is organized by Jonathan Wood & Associates, which is owned by the same company as this news organization.
LA JOLLA, CALIF. – A retrospective study suggests patients with mycosis fungoides (MF) have an increased risk of developing hematologic and solid tumor malignancies.

Researchers found the risk of second malignancy was highest among MF patients aged 30 to 50 years and patients who had tumor stage or advanced stage MF.
The increased risk was present during the entire period after MF diagnosis, but it was greatest in the first 6 months after diagnosis and roughly a dozen years later.
Amrita Goyal, MD, of the University of Minnesota in Minneapolis, and her colleagues presented these findings at the annual T-cell Lymphoma Forum.
The researchers first assessed the risk of second malignancy in 172 MF patients treated at UMN from 2005 to 2017, comparing this cohort to a control group of 172 patients with seborrheic dermatitis.
Second malignancies occurred in 24 MF patients and three controls, which was a significant difference (P = .0045). The most common second malignancies among the MF patients were melanoma (n = 4), prostate cancer (n = 3), and renal cell carcinoma (n = 3).
Further analyses revealed that MF patients were more likely to develop a second malignancy if they had tumor stage disease (P = .0024) or stage IIB or higher disease (P = .03).
To corroborate and expand upon these results, Dr. Goyal and her colleagues analyzed data from the Surveillance, Epidemiology, and End Results (SEER) database on patients diagnosed with MF from 2000 to 2014.
Among the 6,196 MF patients in this cohort, there were 514 second cancers.
“We found that MF patients were, overall, 10 times more likely to develop a second malignancy [compared with the general population],” Dr. Goyal said.
Specifically, the standardized incidence ratio was 10.15 for all malignancies, 7.33 for solid tumors, and 41.72 for hematologic malignancies.
Standardized incidence ratios for individual malignancies were:
- 69.8 for Hodgkin lymphoma.
- 46.5 for non-Hodgkin lymphoma.
- 8.6 for leukemia.
- 7.2 for melanoma.
- 6.2 for lung cancer.
- 7.9 for female breast cancer.
- 5.2 for colon cancer.
- 4.1 for prostate cancer.
- 3.9 for renal cell carcinoma.
- 3.8 for pancreatic cancer.
- 3.6 for bladder cancer.
“We found there is an increased risk [of second malignancy] during the first 6 months after diagnosis of MF, likely related to patients being in contact with the health care system more,” Dr. Goyal said. “Over time, patients have about a 7- to 10-fold increased risk over baseline, until they reach about 12 or 13 years after diagnosis, at which point, there is an increase in risk.”
The researchers found the greatest risk of second malignancy was among patients aged 30 to 50 years, although there was an increased risk for all age groups.
“The reason we think patients are experiencing an increased risk of cancers is we believe this may be due to immune suppression secondary to the mycosis fungoides, although further studies need to be performed to determine if that’s accurate,” Dr. Goyal said.
To that end, she and her colleagues are planning gene expression studies in patients from the UMN cohort. The researchers plan to examine genes involved in the pathogenesis of second malignancies and MF progression in tissue samples from 36 MF patients, 12 who developed second malignancies and 24 who did not.
The current research was funded by the American Society of Hematology. Dr. Goyal reported having no relevant financial disclosures. The T-cell Lymphoma Forum is organized by Jonathan Wood & Associates, which is owned by the same company as this news organization.
LA JOLLA, CALIF. – A retrospective study suggests patients with mycosis fungoides (MF) have an increased risk of developing hematologic and solid tumor malignancies.

Researchers found the risk of second malignancy was highest among MF patients aged 30 to 50 years and patients who had tumor stage or advanced stage MF.
The increased risk was present during the entire period after MF diagnosis, but it was greatest in the first 6 months after diagnosis and roughly a dozen years later.
Amrita Goyal, MD, of the University of Minnesota in Minneapolis, and her colleagues presented these findings at the annual T-cell Lymphoma Forum.
The researchers first assessed the risk of second malignancy in 172 MF patients treated at UMN from 2005 to 2017, comparing this cohort to a control group of 172 patients with seborrheic dermatitis.
Second malignancies occurred in 24 MF patients and three controls, which was a significant difference (P = .0045). The most common second malignancies among the MF patients were melanoma (n = 4), prostate cancer (n = 3), and renal cell carcinoma (n = 3).
Further analyses revealed that MF patients were more likely to develop a second malignancy if they had tumor stage disease (P = .0024) or stage IIB or higher disease (P = .03).
To corroborate and expand upon these results, Dr. Goyal and her colleagues analyzed data from the Surveillance, Epidemiology, and End Results (SEER) database on patients diagnosed with MF from 2000 to 2014.
Among the 6,196 MF patients in this cohort, there were 514 second cancers.
“We found that MF patients were, overall, 10 times more likely to develop a second malignancy [compared with the general population],” Dr. Goyal said.
Specifically, the standardized incidence ratio was 10.15 for all malignancies, 7.33 for solid tumors, and 41.72 for hematologic malignancies.
Standardized incidence ratios for individual malignancies were:
- 69.8 for Hodgkin lymphoma.
- 46.5 for non-Hodgkin lymphoma.
- 8.6 for leukemia.
- 7.2 for melanoma.
- 6.2 for lung cancer.
- 7.9 for female breast cancer.
- 5.2 for colon cancer.
- 4.1 for prostate cancer.
- 3.9 for renal cell carcinoma.
- 3.8 for pancreatic cancer.
- 3.6 for bladder cancer.
“We found there is an increased risk [of second malignancy] during the first 6 months after diagnosis of MF, likely related to patients being in contact with the health care system more,” Dr. Goyal said. “Over time, patients have about a 7- to 10-fold increased risk over baseline, until they reach about 12 or 13 years after diagnosis, at which point, there is an increase in risk.”
The researchers found the greatest risk of second malignancy was among patients aged 30 to 50 years, although there was an increased risk for all age groups.
“The reason we think patients are experiencing an increased risk of cancers is we believe this may be due to immune suppression secondary to the mycosis fungoides, although further studies need to be performed to determine if that’s accurate,” Dr. Goyal said.
To that end, she and her colleagues are planning gene expression studies in patients from the UMN cohort. The researchers plan to examine genes involved in the pathogenesis of second malignancies and MF progression in tissue samples from 36 MF patients, 12 who developed second malignancies and 24 who did not.
The current research was funded by the American Society of Hematology. Dr. Goyal reported having no relevant financial disclosures. The T-cell Lymphoma Forum is organized by Jonathan Wood & Associates, which is owned by the same company as this news organization.
REPORTING FROM TCLF 2019
Key clinical point:
Major finding: In a cohort of MF patients from the SEER database, the standardized incidence ratio was 10.15 for all malignancies, 7.33 for solid tumors, and 41.72 for hematologic malignancies.
Study details: Retrospective study of 6,196 MF patients from the SEER database, and a single-center cohort of 172 MF patients who were matched to 172 patients with seborrheic dermatitis.
Disclosures: This research was funded by the American Society of Hematology. Dr. Goyal reported having no relevant financial disclosures.
Cobomarsen shows early promise for treating ATLL
LA JOLLA, CALIF. – Phase 1 results suggest cobomarsen is well tolerated and can maintain or improve responses in patients with previously treated adult T-cell leukemia/lymphoma (ATLL).
Five of eight ATLL patients studied experienced disease stabilization or improvement while receiving cobomarsen (MRG-106), an inhibitor of microRNA-155.
There were no grade 3/4 adverse events (AEs) or serious AEs related to cobomarsen in these patients.
Francine Foss, MD, of Yale Cancer Center in New Haven, Conn., and her colleagues presented these results at the annual T-cell Lymphoma Forum.
In this ongoing trial (NCT02580552), researchers are evaluating cobomarsen in patients with B- and T-cell lymphomas, including mycosis fungoides and ATLL.
Results are available for eight patients with previously treated ATLL. These patients had received a median of 4 (range, 1-10) prior systemic therapies, and they had a median age of 51 years (range, 40-68).
The patients received three loading doses of cobomarsen during the first week of cycle 1, followed by weekly dosing. All patients have received cobomarsen as a 600 mg intravenous infusion. They can remain on cobomarsen until they progress, experience clinically significant side effects, cannot tolerate the drug, or the trial is terminated.
The researchers have measured efficacy at least monthly by monitoring tumor cell burden in the peripheral blood and lymph nodes, as well as evaluating changes in skin involvement.
Stabilization and response
“Initially, we saw some very good responses in patients who had escalating disease. In other words, their disease was progressing after conventional chemotherapy,” Dr. Foss said. “They went on this microRNA, [and] their disease stabilized and then regressed. We saw, subsequently, in another three or four patients, the same pattern of activity.”
In all, five patients achieved or maintained a response while on cobomarsen. All five were still receiving the drug at the data cutoff on Dec. 13, 2018.
Two of these patients had acute disease and were in partial response (PR) at baseline. These patients had received cobomarsen for 87 days and 401 days as of the data cutoff.
The other three patients still receiving cobomarsen at the cutoff had lymphomatous disease. At baseline, two of the patients were in PR and one had stable disease.
The two patients in PR at baseline had received cobomarsen for 80 days and 366 days at the data cutoff. The patient with stable disease had received the drug for 161 days.
Progression and withdrawal
There were three patients who withdrew from the study because of disease progression. Two of these patients were relapsing with significant skin involvement at baseline.
One of the patients discontinued cobomarsen after 23 days of treatment. The other patient received cobomarsen for 91 days and left the study, then re-enrolled and received cobomarsen for another 42 days before withdrawing from the study again.
The third patient had relapsed lymphomatous disease at baseline. This patient had a mixed response to cobomarsen, with some nodes decreasing in size and others increasing. She discontinued cobomarsen after 9 days.
“It’s still early on in our experience with ATLL, so we don’t really know yet who the patient is that’s going to respond – what are the clinical features that would predict response in these patients,” Dr. Foss said. “And we’re still really trying to understand how we give the drug to these patients, for how long, and whether or not we can change the dosing interval. But, nevertheless, we have some very interesting data.”
Safety
There were no dose-limiting toxicities, AE-related discontinuations, treatment-related grade 3/4 AEs, or new opportunistic infections observed.
“[I] have to say, in using this drug now for over a year in two of my patients – and that’s with weekly administration – we really haven’t seen anything as far as adverse events,” Dr. Foss said.
She noted that one patient has reported transient diarrhea after dosing.
Two serious AEs – febrile neutropenia and pyrexia – occurred in one patient, but neither of these events were considered related to cobomarsen. The AEs occurred after the patient had stopped cobomarsen, and both events resolved.
There were no on-treatment deaths. One patient (the one who received cobomarsen for 9 days) died from disease progression approximately 2 months after stopping cobomarsen and while on a different therapy.
Dr. Foss said, based on their results, she and her colleagues are hoping to accrue more ATLL patients in this trial.
The trial is sponsored by miRagen Therapeutics. Dr. Foss is a cochair of the T-cell Lymphoma Forum. The T-cell Lymphoma Forum is organized by Jonathan Wood & Associates, which is owned by the same company as this news organization.
LA JOLLA, CALIF. – Phase 1 results suggest cobomarsen is well tolerated and can maintain or improve responses in patients with previously treated adult T-cell leukemia/lymphoma (ATLL).
Five of eight ATLL patients studied experienced disease stabilization or improvement while receiving cobomarsen (MRG-106), an inhibitor of microRNA-155.
There were no grade 3/4 adverse events (AEs) or serious AEs related to cobomarsen in these patients.
Francine Foss, MD, of Yale Cancer Center in New Haven, Conn., and her colleagues presented these results at the annual T-cell Lymphoma Forum.
In this ongoing trial (NCT02580552), researchers are evaluating cobomarsen in patients with B- and T-cell lymphomas, including mycosis fungoides and ATLL.
Results are available for eight patients with previously treated ATLL. These patients had received a median of 4 (range, 1-10) prior systemic therapies, and they had a median age of 51 years (range, 40-68).
The patients received three loading doses of cobomarsen during the first week of cycle 1, followed by weekly dosing. All patients have received cobomarsen as a 600 mg intravenous infusion. They can remain on cobomarsen until they progress, experience clinically significant side effects, cannot tolerate the drug, or the trial is terminated.
The researchers have measured efficacy at least monthly by monitoring tumor cell burden in the peripheral blood and lymph nodes, as well as evaluating changes in skin involvement.
Stabilization and response
“Initially, we saw some very good responses in patients who had escalating disease. In other words, their disease was progressing after conventional chemotherapy,” Dr. Foss said. “They went on this microRNA, [and] their disease stabilized and then regressed. We saw, subsequently, in another three or four patients, the same pattern of activity.”
In all, five patients achieved or maintained a response while on cobomarsen. All five were still receiving the drug at the data cutoff on Dec. 13, 2018.
Two of these patients had acute disease and were in partial response (PR) at baseline. These patients had received cobomarsen for 87 days and 401 days as of the data cutoff.
The other three patients still receiving cobomarsen at the cutoff had lymphomatous disease. At baseline, two of the patients were in PR and one had stable disease.
The two patients in PR at baseline had received cobomarsen for 80 days and 366 days at the data cutoff. The patient with stable disease had received the drug for 161 days.
Progression and withdrawal
There were three patients who withdrew from the study because of disease progression. Two of these patients were relapsing with significant skin involvement at baseline.
One of the patients discontinued cobomarsen after 23 days of treatment. The other patient received cobomarsen for 91 days and left the study, then re-enrolled and received cobomarsen for another 42 days before withdrawing from the study again.
The third patient had relapsed lymphomatous disease at baseline. This patient had a mixed response to cobomarsen, with some nodes decreasing in size and others increasing. She discontinued cobomarsen after 9 days.
“It’s still early on in our experience with ATLL, so we don’t really know yet who the patient is that’s going to respond – what are the clinical features that would predict response in these patients,” Dr. Foss said. “And we’re still really trying to understand how we give the drug to these patients, for how long, and whether or not we can change the dosing interval. But, nevertheless, we have some very interesting data.”
Safety
There were no dose-limiting toxicities, AE-related discontinuations, treatment-related grade 3/4 AEs, or new opportunistic infections observed.
“[I] have to say, in using this drug now for over a year in two of my patients – and that’s with weekly administration – we really haven’t seen anything as far as adverse events,” Dr. Foss said.
She noted that one patient has reported transient diarrhea after dosing.
Two serious AEs – febrile neutropenia and pyrexia – occurred in one patient, but neither of these events were considered related to cobomarsen. The AEs occurred after the patient had stopped cobomarsen, and both events resolved.
There were no on-treatment deaths. One patient (the one who received cobomarsen for 9 days) died from disease progression approximately 2 months after stopping cobomarsen and while on a different therapy.
Dr. Foss said, based on their results, she and her colleagues are hoping to accrue more ATLL patients in this trial.
The trial is sponsored by miRagen Therapeutics. Dr. Foss is a cochair of the T-cell Lymphoma Forum. The T-cell Lymphoma Forum is organized by Jonathan Wood & Associates, which is owned by the same company as this news organization.
LA JOLLA, CALIF. – Phase 1 results suggest cobomarsen is well tolerated and can maintain or improve responses in patients with previously treated adult T-cell leukemia/lymphoma (ATLL).
Five of eight ATLL patients studied experienced disease stabilization or improvement while receiving cobomarsen (MRG-106), an inhibitor of microRNA-155.
There were no grade 3/4 adverse events (AEs) or serious AEs related to cobomarsen in these patients.
Francine Foss, MD, of Yale Cancer Center in New Haven, Conn., and her colleagues presented these results at the annual T-cell Lymphoma Forum.
In this ongoing trial (NCT02580552), researchers are evaluating cobomarsen in patients with B- and T-cell lymphomas, including mycosis fungoides and ATLL.
Results are available for eight patients with previously treated ATLL. These patients had received a median of 4 (range, 1-10) prior systemic therapies, and they had a median age of 51 years (range, 40-68).
The patients received three loading doses of cobomarsen during the first week of cycle 1, followed by weekly dosing. All patients have received cobomarsen as a 600 mg intravenous infusion. They can remain on cobomarsen until they progress, experience clinically significant side effects, cannot tolerate the drug, or the trial is terminated.
The researchers have measured efficacy at least monthly by monitoring tumor cell burden in the peripheral blood and lymph nodes, as well as evaluating changes in skin involvement.
Stabilization and response
“Initially, we saw some very good responses in patients who had escalating disease. In other words, their disease was progressing after conventional chemotherapy,” Dr. Foss said. “They went on this microRNA, [and] their disease stabilized and then regressed. We saw, subsequently, in another three or four patients, the same pattern of activity.”
In all, five patients achieved or maintained a response while on cobomarsen. All five were still receiving the drug at the data cutoff on Dec. 13, 2018.
Two of these patients had acute disease and were in partial response (PR) at baseline. These patients had received cobomarsen for 87 days and 401 days as of the data cutoff.
The other three patients still receiving cobomarsen at the cutoff had lymphomatous disease. At baseline, two of the patients were in PR and one had stable disease.
The two patients in PR at baseline had received cobomarsen for 80 days and 366 days at the data cutoff. The patient with stable disease had received the drug for 161 days.
Progression and withdrawal
There were three patients who withdrew from the study because of disease progression. Two of these patients were relapsing with significant skin involvement at baseline.
One of the patients discontinued cobomarsen after 23 days of treatment. The other patient received cobomarsen for 91 days and left the study, then re-enrolled and received cobomarsen for another 42 days before withdrawing from the study again.
The third patient had relapsed lymphomatous disease at baseline. This patient had a mixed response to cobomarsen, with some nodes decreasing in size and others increasing. She discontinued cobomarsen after 9 days.
“It’s still early on in our experience with ATLL, so we don’t really know yet who the patient is that’s going to respond – what are the clinical features that would predict response in these patients,” Dr. Foss said. “And we’re still really trying to understand how we give the drug to these patients, for how long, and whether or not we can change the dosing interval. But, nevertheless, we have some very interesting data.”
Safety
There were no dose-limiting toxicities, AE-related discontinuations, treatment-related grade 3/4 AEs, or new opportunistic infections observed.
“[I] have to say, in using this drug now for over a year in two of my patients – and that’s with weekly administration – we really haven’t seen anything as far as adverse events,” Dr. Foss said.
She noted that one patient has reported transient diarrhea after dosing.
Two serious AEs – febrile neutropenia and pyrexia – occurred in one patient, but neither of these events were considered related to cobomarsen. The AEs occurred after the patient had stopped cobomarsen, and both events resolved.
There were no on-treatment deaths. One patient (the one who received cobomarsen for 9 days) died from disease progression approximately 2 months after stopping cobomarsen and while on a different therapy.
Dr. Foss said, based on their results, she and her colleagues are hoping to accrue more ATLL patients in this trial.
The trial is sponsored by miRagen Therapeutics. Dr. Foss is a cochair of the T-cell Lymphoma Forum. The T-cell Lymphoma Forum is organized by Jonathan Wood & Associates, which is owned by the same company as this news organization.
REPORTING FROM TCLF 2019
Key clinical point:
Major finding: Five of eight ATLL patients studied experienced disease stabilization or improvement while receiving cobomarsen.
Study details: Phase 1 trial including eight ATLL patients.
Disclosures: The trial is sponsored by miRagen Therapeutics.
Cerdulatinib yields ‘encouraging’ results in CTCL, PTCL
LA JOLLA, CALIF. – The spleen tyrosine kinase/Janus kinase inhibitor cerdulatinib has demonstrated activity against relapsed and refractory T-cell lymphomas.

In a phase 2 trial, cerdulatinib produced responses in 34% of patients with peripheral T-cell lymphoma (PTCL) and 26% of those with cutaneous T-cell lymphoma (CTCL).
The best responders were patients with angioimmunoblastic T-cell lymphoma, half of whom achieved a complete response (CR).
The most common grade 3 or higher adverse events (AEs) were amylase increase and lipase increase. However, these increases resolved with dose reduction or interruption, and there were no cases of clinical pancreatitis.
“The data is very encouraging,” said Tatyana Feldman, MD, of the John Theurer Cancer Center in Hackensack, N.J.
Dr. Feldman and her colleagues previously presented results from the phase 2 trial of cerdulatinib (NCT01994382) at the 2018 annual congress of the European Hematology Association.
Dr. Feldman and her colleagues presented data from expansion cohorts of the ongoing trial at the annual T-cell Lymphoma Forum. The cohorts included patients with PTCL or CTCL who had received at least one prior systemic therapy.
PTCL cohort
The 45 PTCL patients had a median age of 65 years (range, 21-84). They had received a median of 3 (range, 1-12) prior therapeutic regimens, 51% were refractory to their last therapy, and 27% had undergone stem cell transplant (SCT).
The patients received cerdulatinib at 30 mg orally twice a day until progression or intolerance, and 41 patients were evaluable for response.
The overall response rate was 34% (n = 14). Eleven patients had a CR, three had a partial response (PR), and nine had stable disease.
Responses according to subtype were as follows:
- 7 CRs and 1 PR in angioimmunoblastic T-cell lymphoma.
- 2 CRs in PTCL not otherwise specified.
- 1 CR in gamma-delta T-cell lymphoma.
- 1 PR in ALK-negative anaplastic large-cell lymphoma.
- 1 CR and 1 PR in adult T-cell leukemia/lymphoma.
Eight responders have remained on cerdulatinib for anywhere from 3 months to more than 12 months. Five patients have had a response lasting at least 6 months. One patient went on to SCT after achieving a CR.
The most common grade 3 or higher AEs observed in PTCL patients were amylase increase (n = 8), lipase increase (n = 6), pneumonia/lung infection (n = 5), neutropenia (n = 4), diarrhea (n = 4), febrile neutropenia (n = 4), abdominal pain (n = 4), sepsis/bacteremia (n = 3), anemia (n = 3), fatigue (n = 2), and pain (n = 1).
There were two grade 5 AEs – acute respiratory distress syndrome and pneumonia.
CTCL cohort
The 29 CTCL patients had a median age of 62 years (range, 24-79). They had received a median of 4 (range, 1-13) prior therapies, 55% were refractory to their last therapy, and 3% had undergone SCT.
The patients received cerdulatinib at 30 mg orally twice a day until progression or intolerance, and 27 were evaluable for response.
The overall response rate was 26% (n = 7). Two patients achieved a CR, five achieved a PR, and nine had stable disease. Responses occurred in mycosis fungoides and Sézary syndrome.
Eleven of 23 patients (48%) achieved at least a 50% reduction in skin lesions, and the researchers observed rapid improvements in pruritus.
“I saw patients who would take the first pill, and they would call me and say, ‘I no longer itch,’ ” Dr. Feldman said.
The most common grade 3 or higher AEs in CTCL patients were lipase increase (n = 11), amylase increase (n = 5), sepsis/bacteremia (n = 3), pain (n = 2), fatigue (n = 1), neutropenia (n = 1), and diarrhea (n = 1).
“It’s a very well-tolerated drug,” Dr. Feldman said, adding that there were “really no severe side effects which would prohibit the use of the drug.”
She noted that cerdulatinib’s “favorable” side effect profile might make it a promising candidate for use in combination regimens.
“I think it will be possible to combine it with other drugs in development in T-cell lymphoma. … immunological checkpoint inhibitors, epigenetic modulators such as HDAC [histone deacetylase] inhibitors, methylating agents, and PI3 kinase inhibitors,” Dr. Feldman said.
She reported having no disclosures relevant to this study. The trial is sponsored by Portola Pharmaceuticals.
The T-cell Lymphoma Forum is organized by Jonathan Wood & Associates, which is owned by the same company as this news organization.
LA JOLLA, CALIF. – The spleen tyrosine kinase/Janus kinase inhibitor cerdulatinib has demonstrated activity against relapsed and refractory T-cell lymphomas.

In a phase 2 trial, cerdulatinib produced responses in 34% of patients with peripheral T-cell lymphoma (PTCL) and 26% of those with cutaneous T-cell lymphoma (CTCL).
The best responders were patients with angioimmunoblastic T-cell lymphoma, half of whom achieved a complete response (CR).
The most common grade 3 or higher adverse events (AEs) were amylase increase and lipase increase. However, these increases resolved with dose reduction or interruption, and there were no cases of clinical pancreatitis.
“The data is very encouraging,” said Tatyana Feldman, MD, of the John Theurer Cancer Center in Hackensack, N.J.
Dr. Feldman and her colleagues previously presented results from the phase 2 trial of cerdulatinib (NCT01994382) at the 2018 annual congress of the European Hematology Association.
Dr. Feldman and her colleagues presented data from expansion cohorts of the ongoing trial at the annual T-cell Lymphoma Forum. The cohorts included patients with PTCL or CTCL who had received at least one prior systemic therapy.
PTCL cohort
The 45 PTCL patients had a median age of 65 years (range, 21-84). They had received a median of 3 (range, 1-12) prior therapeutic regimens, 51% were refractory to their last therapy, and 27% had undergone stem cell transplant (SCT).
The patients received cerdulatinib at 30 mg orally twice a day until progression or intolerance, and 41 patients were evaluable for response.
The overall response rate was 34% (n = 14). Eleven patients had a CR, three had a partial response (PR), and nine had stable disease.
Responses according to subtype were as follows:
- 7 CRs and 1 PR in angioimmunoblastic T-cell lymphoma.
- 2 CRs in PTCL not otherwise specified.
- 1 CR in gamma-delta T-cell lymphoma.
- 1 PR in ALK-negative anaplastic large-cell lymphoma.
- 1 CR and 1 PR in adult T-cell leukemia/lymphoma.
Eight responders have remained on cerdulatinib for anywhere from 3 months to more than 12 months. Five patients have had a response lasting at least 6 months. One patient went on to SCT after achieving a CR.
The most common grade 3 or higher AEs observed in PTCL patients were amylase increase (n = 8), lipase increase (n = 6), pneumonia/lung infection (n = 5), neutropenia (n = 4), diarrhea (n = 4), febrile neutropenia (n = 4), abdominal pain (n = 4), sepsis/bacteremia (n = 3), anemia (n = 3), fatigue (n = 2), and pain (n = 1).
There were two grade 5 AEs – acute respiratory distress syndrome and pneumonia.
CTCL cohort
The 29 CTCL patients had a median age of 62 years (range, 24-79). They had received a median of 4 (range, 1-13) prior therapies, 55% were refractory to their last therapy, and 3% had undergone SCT.
The patients received cerdulatinib at 30 mg orally twice a day until progression or intolerance, and 27 were evaluable for response.
The overall response rate was 26% (n = 7). Two patients achieved a CR, five achieved a PR, and nine had stable disease. Responses occurred in mycosis fungoides and Sézary syndrome.
Eleven of 23 patients (48%) achieved at least a 50% reduction in skin lesions, and the researchers observed rapid improvements in pruritus.
“I saw patients who would take the first pill, and they would call me and say, ‘I no longer itch,’ ” Dr. Feldman said.
The most common grade 3 or higher AEs in CTCL patients were lipase increase (n = 11), amylase increase (n = 5), sepsis/bacteremia (n = 3), pain (n = 2), fatigue (n = 1), neutropenia (n = 1), and diarrhea (n = 1).
“It’s a very well-tolerated drug,” Dr. Feldman said, adding that there were “really no severe side effects which would prohibit the use of the drug.”
She noted that cerdulatinib’s “favorable” side effect profile might make it a promising candidate for use in combination regimens.
“I think it will be possible to combine it with other drugs in development in T-cell lymphoma. … immunological checkpoint inhibitors, epigenetic modulators such as HDAC [histone deacetylase] inhibitors, methylating agents, and PI3 kinase inhibitors,” Dr. Feldman said.
She reported having no disclosures relevant to this study. The trial is sponsored by Portola Pharmaceuticals.
The T-cell Lymphoma Forum is organized by Jonathan Wood & Associates, which is owned by the same company as this news organization.
LA JOLLA, CALIF. – The spleen tyrosine kinase/Janus kinase inhibitor cerdulatinib has demonstrated activity against relapsed and refractory T-cell lymphomas.

In a phase 2 trial, cerdulatinib produced responses in 34% of patients with peripheral T-cell lymphoma (PTCL) and 26% of those with cutaneous T-cell lymphoma (CTCL).
The best responders were patients with angioimmunoblastic T-cell lymphoma, half of whom achieved a complete response (CR).
The most common grade 3 or higher adverse events (AEs) were amylase increase and lipase increase. However, these increases resolved with dose reduction or interruption, and there were no cases of clinical pancreatitis.
“The data is very encouraging,” said Tatyana Feldman, MD, of the John Theurer Cancer Center in Hackensack, N.J.
Dr. Feldman and her colleagues previously presented results from the phase 2 trial of cerdulatinib (NCT01994382) at the 2018 annual congress of the European Hematology Association.
Dr. Feldman and her colleagues presented data from expansion cohorts of the ongoing trial at the annual T-cell Lymphoma Forum. The cohorts included patients with PTCL or CTCL who had received at least one prior systemic therapy.
PTCL cohort
The 45 PTCL patients had a median age of 65 years (range, 21-84). They had received a median of 3 (range, 1-12) prior therapeutic regimens, 51% were refractory to their last therapy, and 27% had undergone stem cell transplant (SCT).
The patients received cerdulatinib at 30 mg orally twice a day until progression or intolerance, and 41 patients were evaluable for response.
The overall response rate was 34% (n = 14). Eleven patients had a CR, three had a partial response (PR), and nine had stable disease.
Responses according to subtype were as follows:
- 7 CRs and 1 PR in angioimmunoblastic T-cell lymphoma.
- 2 CRs in PTCL not otherwise specified.
- 1 CR in gamma-delta T-cell lymphoma.
- 1 PR in ALK-negative anaplastic large-cell lymphoma.
- 1 CR and 1 PR in adult T-cell leukemia/lymphoma.
Eight responders have remained on cerdulatinib for anywhere from 3 months to more than 12 months. Five patients have had a response lasting at least 6 months. One patient went on to SCT after achieving a CR.
The most common grade 3 or higher AEs observed in PTCL patients were amylase increase (n = 8), lipase increase (n = 6), pneumonia/lung infection (n = 5), neutropenia (n = 4), diarrhea (n = 4), febrile neutropenia (n = 4), abdominal pain (n = 4), sepsis/bacteremia (n = 3), anemia (n = 3), fatigue (n = 2), and pain (n = 1).
There were two grade 5 AEs – acute respiratory distress syndrome and pneumonia.
CTCL cohort
The 29 CTCL patients had a median age of 62 years (range, 24-79). They had received a median of 4 (range, 1-13) prior therapies, 55% were refractory to their last therapy, and 3% had undergone SCT.
The patients received cerdulatinib at 30 mg orally twice a day until progression or intolerance, and 27 were evaluable for response.
The overall response rate was 26% (n = 7). Two patients achieved a CR, five achieved a PR, and nine had stable disease. Responses occurred in mycosis fungoides and Sézary syndrome.
Eleven of 23 patients (48%) achieved at least a 50% reduction in skin lesions, and the researchers observed rapid improvements in pruritus.
“I saw patients who would take the first pill, and they would call me and say, ‘I no longer itch,’ ” Dr. Feldman said.
The most common grade 3 or higher AEs in CTCL patients were lipase increase (n = 11), amylase increase (n = 5), sepsis/bacteremia (n = 3), pain (n = 2), fatigue (n = 1), neutropenia (n = 1), and diarrhea (n = 1).
“It’s a very well-tolerated drug,” Dr. Feldman said, adding that there were “really no severe side effects which would prohibit the use of the drug.”
She noted that cerdulatinib’s “favorable” side effect profile might make it a promising candidate for use in combination regimens.
“I think it will be possible to combine it with other drugs in development in T-cell lymphoma. … immunological checkpoint inhibitors, epigenetic modulators such as HDAC [histone deacetylase] inhibitors, methylating agents, and PI3 kinase inhibitors,” Dr. Feldman said.
She reported having no disclosures relevant to this study. The trial is sponsored by Portola Pharmaceuticals.
The T-cell Lymphoma Forum is organized by Jonathan Wood & Associates, which is owned by the same company as this news organization.
REPORTING FROM TCLF 2019
Key clinical point:
Major finding: The overall response rate was 34% in patients with peripheral T-cell lymphoma (PTCL) and 26% in patients with cutaneous T-cell lymphoma (CTCL).
Study details: Expansion cohorts of a phase 2 trial including 45 PTCL patients and 29 CTCL patients
Disclosures: The study was funded by Portola Pharmaceuticals. The investigator reported having no relevant conflicts.
Combo emerges as bridge to transplant in rel/ref PTCL
LA JOLLA, CALIF. – The combination of duvelisib and romidepsin is active and can provide a bridge to transplant in relapsed or refractory peripheral T-cell lymphoma (PTCL), according to researchers.

In a phase 1 trial, duvelisib plus romidepsin produced an overall response rate (ORR) of 59% in patients with PTCL. Sixteen patients achieved a response, nine had a complete response (CR), and six complete responders went on to transplant.
“So we think that you can achieve remission deep enough to then move on to a potentially curative approach,” said study investigator Neha Mehta-Shah, MD, of Washington University in St. Louis.
She and her colleagues evaluated romidepsin plus duvelisib, as well as bortezomib plus duvelisib, in a phase 1 trial (NCT02783625) of patients with relapsed or refractory PTCL or cutaneous T-cell lymphoma (CTCL).
Dr. Mehta-Shah presented the results at the annual T-cell Lymphoma Forum.
She reported results in 80 patients – 51 with PTCL and 29 with CTCL. The patients’ median age was 64 years (range, 28-83), and 57% of the study population were men. Patients had received a median of 3 (range, 1-16) prior therapies, and 16% had received a prior transplant.
Treatment
Dr. Mehta-Shah noted that patients and providers could choose whether patients would receive romidepsin or bortezomib.
Patients in the romidepsin arm received romidepsin at 10 mg/m2 on days 1, 8, and 15 of each 28-day cycle. Patients in the bortezomib arm received bortezomib at 1 mg/m2 on days 1, 4, 8, and 11 of each cycle.
Duvelisib dosing was escalated, so patients received duvelisib at 25 mg, 50 mg, or 75 mg twice daily.
In the bortezomib arm, there was one dose-limiting toxicity – grade 3 neutropenia – in a patient who received duvelisib at the 25-mg dose. There were no dose-limiting toxicities in the romidepsin arm.
The researchers determined that the maximum tolerated dose (MTD) of duvelisib was 75 mg twice daily in the romidepsin arm and 25 mg twice daily in the bortezomib arm.
Lead-in phase
The study also had a lead-in phase during which patients could receive single-agent duvelisib.
“Because the original phase 1 study of duvelisib did not collect as many prospective tumor biopsies or on-treatment biopsies, we built into this study a lead-in phase so that we could characterize on-treatment biopsies to better understand mechanisms of response or resistance,” Dr. Mehta-Shah said.
Patients and providers could choose to be part of the lead-in phase, she noted. Patients who did not achieve a CR during this phase went on to receive either combination therapy, which was predetermined before the monotherapy began.
There were 14 patients who received duvelisib monotherapy at 75 mg twice daily. Four of them achieved a CR, and three had a partial response (PR). Ten patients went on to receive romidepsin as well. One of them achieved a CR, and three had a PR.
There were 12 patients who received duvelisib monotherapy at 25 mg twice daily. Three of them achieved a CR, and two had a PR. Nine patients went on to receive bortezomib as well. This combination produced one CR and two PRs.
Efficacy with romidepsin
Among all evaluable PTCL patients in the romidepsin arm, the ORR was 59% (16/27), and the CR rate was 33% (9/27).
Responses occurred in seven patients with PTCL not otherwise specified (NOS), six with angioimmunoblastic T-cell lymphoma (AITL), one with hepatosplenic T-cell lymphoma, one with aggressive epidermotropic CD8+ T-cell lymphoma, and one with primary cutaneous PTCL.
CRs occurred in five patients with AITL and four with PTCL-NOS. Six patients who achieved a CR went on to transplant.
Among evaluable CTCL patients in the romidepsin arm, the ORR was 45% (5/11), and there were no CRs. Responses occurred in three patients with mycosis fungoides and two with Sézary syndrome.
The median progression-free survival was 5.41 months in CTCL patients and 6.72 months in PTCL patients.
Efficacy with bortezomib
Among evaluable PTCL patients in the bortezomib arm, the ORR was 44% (7/16), and the CR rate was 25% (4/16).
Responses occurred in three patients with AITL and four with PTCL-NOS. CRs occurred in two patients with each subtype.
Among evaluable CTCL patients in the bortezomib arm, the ORR was 27% (4/15), and there were no CRs. Responses occurred in one patient with mycosis fungoides and three with Sézary syndrome. One CTCL patient went on to transplant.
The median progression-free survival was 4.56 months among CTCL patients and 4.39 months in PTCL patients.
Safety
Dr. Mehta-Shah said both combinations were considered safe and well tolerated. However, there was a grade 5 adverse event (AE) – Stevens-Johnson syndrome – that occurred in the bortezomib arm and was considered possibly related to treatment.
Grade 3/4 AEs observed in the 31 patients treated at the MTD in the romidepsin arm were transaminase increase (n = 7), diarrhea (n = 6), hyponatremia (n = 4), neutrophil count decrease (n = 10), and platelet count decrease (n = 3).
Grade 3/4 AEs observed in the 23 patients treated at the MTD in the bortezomib arm were transaminase increase (n = 2) and neutrophil count decrease (n = 5).
Grade 3/4 transaminitis seemed to be more common among patients who received duvelisib alone during the lead-in phase, Dr. Mehta-Shah said.
Among patients treated at the MTD in the romidepsin arm, grade 3/4 transaminitis occurred in four patients treated during the lead-in phase and three who began receiving romidepsin and duvelisib together. In the bortezomib arm, grade 3/4 transaminitis occurred in two patients treated at the MTD, both of whom received duvelisib alone during the lead-in phase.
Based on these results, Dr. Mehta-Shah and her colleagues are planning to expand the romidepsin arm to an additional 25 patients. By testing the combination in more patients, the researchers hope to better understand the occurrence of transaminitis and assess the durability of response.
This study is supported by Verastem. Dr. Shah reported relationships with Celgene, Kyowa Kirin, Bristol-Myers Squibb, Verastem, and Genentech.
The T-cell Lymphoma Forum is held by Jonathan Wood & Associates, which is owned by the same company as this news organization.
LA JOLLA, CALIF. – The combination of duvelisib and romidepsin is active and can provide a bridge to transplant in relapsed or refractory peripheral T-cell lymphoma (PTCL), according to researchers.

In a phase 1 trial, duvelisib plus romidepsin produced an overall response rate (ORR) of 59% in patients with PTCL. Sixteen patients achieved a response, nine had a complete response (CR), and six complete responders went on to transplant.
“So we think that you can achieve remission deep enough to then move on to a potentially curative approach,” said study investigator Neha Mehta-Shah, MD, of Washington University in St. Louis.
She and her colleagues evaluated romidepsin plus duvelisib, as well as bortezomib plus duvelisib, in a phase 1 trial (NCT02783625) of patients with relapsed or refractory PTCL or cutaneous T-cell lymphoma (CTCL).
Dr. Mehta-Shah presented the results at the annual T-cell Lymphoma Forum.
She reported results in 80 patients – 51 with PTCL and 29 with CTCL. The patients’ median age was 64 years (range, 28-83), and 57% of the study population were men. Patients had received a median of 3 (range, 1-16) prior therapies, and 16% had received a prior transplant.
Treatment
Dr. Mehta-Shah noted that patients and providers could choose whether patients would receive romidepsin or bortezomib.
Patients in the romidepsin arm received romidepsin at 10 mg/m2 on days 1, 8, and 15 of each 28-day cycle. Patients in the bortezomib arm received bortezomib at 1 mg/m2 on days 1, 4, 8, and 11 of each cycle.
Duvelisib dosing was escalated, so patients received duvelisib at 25 mg, 50 mg, or 75 mg twice daily.
In the bortezomib arm, there was one dose-limiting toxicity – grade 3 neutropenia – in a patient who received duvelisib at the 25-mg dose. There were no dose-limiting toxicities in the romidepsin arm.
The researchers determined that the maximum tolerated dose (MTD) of duvelisib was 75 mg twice daily in the romidepsin arm and 25 mg twice daily in the bortezomib arm.
Lead-in phase
The study also had a lead-in phase during which patients could receive single-agent duvelisib.
“Because the original phase 1 study of duvelisib did not collect as many prospective tumor biopsies or on-treatment biopsies, we built into this study a lead-in phase so that we could characterize on-treatment biopsies to better understand mechanisms of response or resistance,” Dr. Mehta-Shah said.
Patients and providers could choose to be part of the lead-in phase, she noted. Patients who did not achieve a CR during this phase went on to receive either combination therapy, which was predetermined before the monotherapy began.
There were 14 patients who received duvelisib monotherapy at 75 mg twice daily. Four of them achieved a CR, and three had a partial response (PR). Ten patients went on to receive romidepsin as well. One of them achieved a CR, and three had a PR.
There were 12 patients who received duvelisib monotherapy at 25 mg twice daily. Three of them achieved a CR, and two had a PR. Nine patients went on to receive bortezomib as well. This combination produced one CR and two PRs.
Efficacy with romidepsin
Among all evaluable PTCL patients in the romidepsin arm, the ORR was 59% (16/27), and the CR rate was 33% (9/27).
Responses occurred in seven patients with PTCL not otherwise specified (NOS), six with angioimmunoblastic T-cell lymphoma (AITL), one with hepatosplenic T-cell lymphoma, one with aggressive epidermotropic CD8+ T-cell lymphoma, and one with primary cutaneous PTCL.
CRs occurred in five patients with AITL and four with PTCL-NOS. Six patients who achieved a CR went on to transplant.
Among evaluable CTCL patients in the romidepsin arm, the ORR was 45% (5/11), and there were no CRs. Responses occurred in three patients with mycosis fungoides and two with Sézary syndrome.
The median progression-free survival was 5.41 months in CTCL patients and 6.72 months in PTCL patients.
Efficacy with bortezomib
Among evaluable PTCL patients in the bortezomib arm, the ORR was 44% (7/16), and the CR rate was 25% (4/16).
Responses occurred in three patients with AITL and four with PTCL-NOS. CRs occurred in two patients with each subtype.
Among evaluable CTCL patients in the bortezomib arm, the ORR was 27% (4/15), and there were no CRs. Responses occurred in one patient with mycosis fungoides and three with Sézary syndrome. One CTCL patient went on to transplant.
The median progression-free survival was 4.56 months among CTCL patients and 4.39 months in PTCL patients.
Safety
Dr. Mehta-Shah said both combinations were considered safe and well tolerated. However, there was a grade 5 adverse event (AE) – Stevens-Johnson syndrome – that occurred in the bortezomib arm and was considered possibly related to treatment.
Grade 3/4 AEs observed in the 31 patients treated at the MTD in the romidepsin arm were transaminase increase (n = 7), diarrhea (n = 6), hyponatremia (n = 4), neutrophil count decrease (n = 10), and platelet count decrease (n = 3).
Grade 3/4 AEs observed in the 23 patients treated at the MTD in the bortezomib arm were transaminase increase (n = 2) and neutrophil count decrease (n = 5).
Grade 3/4 transaminitis seemed to be more common among patients who received duvelisib alone during the lead-in phase, Dr. Mehta-Shah said.
Among patients treated at the MTD in the romidepsin arm, grade 3/4 transaminitis occurred in four patients treated during the lead-in phase and three who began receiving romidepsin and duvelisib together. In the bortezomib arm, grade 3/4 transaminitis occurred in two patients treated at the MTD, both of whom received duvelisib alone during the lead-in phase.
Based on these results, Dr. Mehta-Shah and her colleagues are planning to expand the romidepsin arm to an additional 25 patients. By testing the combination in more patients, the researchers hope to better understand the occurrence of transaminitis and assess the durability of response.
This study is supported by Verastem. Dr. Shah reported relationships with Celgene, Kyowa Kirin, Bristol-Myers Squibb, Verastem, and Genentech.
The T-cell Lymphoma Forum is held by Jonathan Wood & Associates, which is owned by the same company as this news organization.
LA JOLLA, CALIF. – The combination of duvelisib and romidepsin is active and can provide a bridge to transplant in relapsed or refractory peripheral T-cell lymphoma (PTCL), according to researchers.

In a phase 1 trial, duvelisib plus romidepsin produced an overall response rate (ORR) of 59% in patients with PTCL. Sixteen patients achieved a response, nine had a complete response (CR), and six complete responders went on to transplant.
“So we think that you can achieve remission deep enough to then move on to a potentially curative approach,” said study investigator Neha Mehta-Shah, MD, of Washington University in St. Louis.
She and her colleagues evaluated romidepsin plus duvelisib, as well as bortezomib plus duvelisib, in a phase 1 trial (NCT02783625) of patients with relapsed or refractory PTCL or cutaneous T-cell lymphoma (CTCL).
Dr. Mehta-Shah presented the results at the annual T-cell Lymphoma Forum.
She reported results in 80 patients – 51 with PTCL and 29 with CTCL. The patients’ median age was 64 years (range, 28-83), and 57% of the study population were men. Patients had received a median of 3 (range, 1-16) prior therapies, and 16% had received a prior transplant.
Treatment
Dr. Mehta-Shah noted that patients and providers could choose whether patients would receive romidepsin or bortezomib.
Patients in the romidepsin arm received romidepsin at 10 mg/m2 on days 1, 8, and 15 of each 28-day cycle. Patients in the bortezomib arm received bortezomib at 1 mg/m2 on days 1, 4, 8, and 11 of each cycle.
Duvelisib dosing was escalated, so patients received duvelisib at 25 mg, 50 mg, or 75 mg twice daily.
In the bortezomib arm, there was one dose-limiting toxicity – grade 3 neutropenia – in a patient who received duvelisib at the 25-mg dose. There were no dose-limiting toxicities in the romidepsin arm.
The researchers determined that the maximum tolerated dose (MTD) of duvelisib was 75 mg twice daily in the romidepsin arm and 25 mg twice daily in the bortezomib arm.
Lead-in phase
The study also had a lead-in phase during which patients could receive single-agent duvelisib.
“Because the original phase 1 study of duvelisib did not collect as many prospective tumor biopsies or on-treatment biopsies, we built into this study a lead-in phase so that we could characterize on-treatment biopsies to better understand mechanisms of response or resistance,” Dr. Mehta-Shah said.
Patients and providers could choose to be part of the lead-in phase, she noted. Patients who did not achieve a CR during this phase went on to receive either combination therapy, which was predetermined before the monotherapy began.
There were 14 patients who received duvelisib monotherapy at 75 mg twice daily. Four of them achieved a CR, and three had a partial response (PR). Ten patients went on to receive romidepsin as well. One of them achieved a CR, and three had a PR.
There were 12 patients who received duvelisib monotherapy at 25 mg twice daily. Three of them achieved a CR, and two had a PR. Nine patients went on to receive bortezomib as well. This combination produced one CR and two PRs.
Efficacy with romidepsin
Among all evaluable PTCL patients in the romidepsin arm, the ORR was 59% (16/27), and the CR rate was 33% (9/27).
Responses occurred in seven patients with PTCL not otherwise specified (NOS), six with angioimmunoblastic T-cell lymphoma (AITL), one with hepatosplenic T-cell lymphoma, one with aggressive epidermotropic CD8+ T-cell lymphoma, and one with primary cutaneous PTCL.
CRs occurred in five patients with AITL and four with PTCL-NOS. Six patients who achieved a CR went on to transplant.
Among evaluable CTCL patients in the romidepsin arm, the ORR was 45% (5/11), and there were no CRs. Responses occurred in three patients with mycosis fungoides and two with Sézary syndrome.
The median progression-free survival was 5.41 months in CTCL patients and 6.72 months in PTCL patients.
Efficacy with bortezomib
Among evaluable PTCL patients in the bortezomib arm, the ORR was 44% (7/16), and the CR rate was 25% (4/16).
Responses occurred in three patients with AITL and four with PTCL-NOS. CRs occurred in two patients with each subtype.
Among evaluable CTCL patients in the bortezomib arm, the ORR was 27% (4/15), and there were no CRs. Responses occurred in one patient with mycosis fungoides and three with Sézary syndrome. One CTCL patient went on to transplant.
The median progression-free survival was 4.56 months among CTCL patients and 4.39 months in PTCL patients.
Safety
Dr. Mehta-Shah said both combinations were considered safe and well tolerated. However, there was a grade 5 adverse event (AE) – Stevens-Johnson syndrome – that occurred in the bortezomib arm and was considered possibly related to treatment.
Grade 3/4 AEs observed in the 31 patients treated at the MTD in the romidepsin arm were transaminase increase (n = 7), diarrhea (n = 6), hyponatremia (n = 4), neutrophil count decrease (n = 10), and platelet count decrease (n = 3).
Grade 3/4 AEs observed in the 23 patients treated at the MTD in the bortezomib arm were transaminase increase (n = 2) and neutrophil count decrease (n = 5).
Grade 3/4 transaminitis seemed to be more common among patients who received duvelisib alone during the lead-in phase, Dr. Mehta-Shah said.
Among patients treated at the MTD in the romidepsin arm, grade 3/4 transaminitis occurred in four patients treated during the lead-in phase and three who began receiving romidepsin and duvelisib together. In the bortezomib arm, grade 3/4 transaminitis occurred in two patients treated at the MTD, both of whom received duvelisib alone during the lead-in phase.
Based on these results, Dr. Mehta-Shah and her colleagues are planning to expand the romidepsin arm to an additional 25 patients. By testing the combination in more patients, the researchers hope to better understand the occurrence of transaminitis and assess the durability of response.
This study is supported by Verastem. Dr. Shah reported relationships with Celgene, Kyowa Kirin, Bristol-Myers Squibb, Verastem, and Genentech.
The T-cell Lymphoma Forum is held by Jonathan Wood & Associates, which is owned by the same company as this news organization.
REPORTING FROM TCLF 2019
Key clinical point:
Major finding: The overall response rate was 59%, and six of nine complete responders went on to transplant.
Study details: Phase 1 trial of 80 patients that included 27 evaluable PTCL patients who received romidepsin and duvelisib.
Disclosures: The study is supported by Verastem. Dr. Shah reported relationships with Celgene, Kyowa Kirin, Bristol-Myers Squibb, Verastem, and Genentech.
Applying ECHELON-2 results to clinical practice
LA JOLLA, CALIF. – Results from the ECHELON-2 trial led to the U.S. approval of brentuximab vedotin (BV) in combination with cyclophosphamide, doxorubicin, and prednisone (CHP), but there are still questions about how to apply the trial results to practice.

At the annual T-cell Lymphoma Forum, trial investigators and other physicians debated the best use of this combination.
BV-CHP is approved to treat patients with previously untreated systemic anaplastic large-cell lymphoma (sALCL) or other CD30-expressing peripheral T-cell lymphomas (PTCLs), including angioimmunoblastic T-cell lymphoma (AITL) and PTCL not otherwise specified (NOS).
Patients who received BV-CHP in ECHELON-2 had superior progression-free survival (PFS) and overall survival (OS) compared to patients who received cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP).
These results were initially presented at the 2018 annual meeting of the American Society of Hematology and simultaneously published in The Lancet (2019 Jan 19;393[10168]:229-40).
ECHELON-2 investigator Owen O’Connor, MD, PhD, of Columbia University Medical Center in New York, also presented details on the trial at the T-cell Lymphoma Forum. His presentation was followed by a discussion with meeting attendees about applying the trial results to clinical practice.
CD30 expression
One of the issues discussed was the importance of CD30 expression in deciding which patients should receive BV.
For a patient to be eligible for ECHELON-2, the diagnostic biopsy had to show at least 10% of the neoplastic cells were CD30-positive. However, the Food and Drug Administration (FDA) has not made a similar requirement for prescribing BV. PTCL patients with any level of CD30 expression are eligible for treatment with BV-CHP, according to the FDA.
“[I]t’s still a matter of great debate and controversy as to whether we have good enough data to suggest that there’s a threshold effect with regard to the expression of CD30 and responsiveness or sensitivity to brentuximab vedotin,” Dr. O’Connor said.
“This has been an issue from the very first day with this drug, which is, ‘Just how much CD30 do you need to get a response?’ I can’t speak on behalf of the FDA, but I think they are not absolutely convinced that there’s a threshold. They take [CD30-] positive as ‘good enough’ across the board.”
“The FDA has said, ‘The data we’ve seen says there’s a lot of heterogeneity [with biopsies].’ You may do a biopsy and find 30% [of cells are CD30-positive], and you may do another biopsy [in the same patient] and find less than 10%. I don’t think the regulatory agencies are convinced that a single biopsy looking at CD30 ... is representative of the entire tumor burden.”
Andrei Shustov, MD, an ECHELON-2 investigator from the University of Washington in Seattle, questioned whether CD30 expression should be considered when deciding on the use of BV in PTCL.
“Is CD30 staining relevant at all, or should we default back to studies, say, in colon cancer where we didn’t even care about EGFR because we might be missing it by current techniques?” Dr. Shustov asked. “Should we even worry about CD30 expression ... because we cannot reliably detect low levels of CD30?”
Some attendees echoed this sentiment, questioning the utility of assessing CD30 expression. Other attendees said they would defer to the trial data and only treat patients with BV-CHP if they had at least 10% CD30.
PTCL subtypes
Meeting attendees also discussed the value of BV in different PTCL subtypes.
At the request of European regulatory agencies, ECHELON-2 was largely focused on patients with sALCL. They made up 70% of the total trial population, while 16% of patients had PTCL-NOS, 12% had AITL, and a small number of patients had other subtypes. These numbers meant ECHELON-2 was not powered to determine differences in OS or PFS in non-sALCL subtypes.
As a result, some attendees expressed concerns about using BV-CHP to treat PTCL-NOS or AITL. They argued that it wasn’t clear whether patients with these subtypes would derive more benefit from BV-CHP, CHOP, or CHOP plus etoposide (CHOEP).
Other attendees said they would feel comfortable using BV-CHP in patients with PTCL-NOS or AITL based on ECHELON-2 results.
CHOP vs. CHOEP
The use of CHOP in ECHELON-2 was another point of discussion. Some attendees said CHOEP should have been used as the comparator instead.
A few individuals mentioned retrospective data suggesting CHOEP may confer a benefit over CHOP in PTCL (Blood. 2010 Nov 4;116[18]:3418-25).
Marek Trneny, MD, of Charles University General Hospital in Prague, referenced new data from the Czech National Lymphoma Registry, which showed that patients newly diagnosed with PTCL had superior PFS and OS when they received CHOEP rather than CHOP.
Based on these findings, Dr. Trneny said he would consider treating CD30-positive PTCL patients with CHOEP plus BV rather than BV-CHP.
However, most other attendees said they would not consider adding BV to CHOEP due to the absence of clinical trial data supporting this approach.
Some attendees did say they would use CHOEP instead of BV-CHP, particularly in patients with PTCL-NOS or AITL and in patients with CD30 expression below 10%.
ECHELON-2 was funded by Seattle Genetics and Millennium Pharmaceuticals, a wholly owned subsidiary of Takeda Pharmaceutical Company.
Dr. O’Connor and Dr. Shustov were investigators on ECHELON-2. Dr. O’Connor is a cochair of the T-cell Lymphoma Forum. The T-cell Lymphoma Forum is organized by Jonathan Wood & Associates, which is owned by the same company as this news organization.
LA JOLLA, CALIF. – Results from the ECHELON-2 trial led to the U.S. approval of brentuximab vedotin (BV) in combination with cyclophosphamide, doxorubicin, and prednisone (CHP), but there are still questions about how to apply the trial results to practice.

At the annual T-cell Lymphoma Forum, trial investigators and other physicians debated the best use of this combination.
BV-CHP is approved to treat patients with previously untreated systemic anaplastic large-cell lymphoma (sALCL) or other CD30-expressing peripheral T-cell lymphomas (PTCLs), including angioimmunoblastic T-cell lymphoma (AITL) and PTCL not otherwise specified (NOS).
Patients who received BV-CHP in ECHELON-2 had superior progression-free survival (PFS) and overall survival (OS) compared to patients who received cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP).
These results were initially presented at the 2018 annual meeting of the American Society of Hematology and simultaneously published in The Lancet (2019 Jan 19;393[10168]:229-40).
ECHELON-2 investigator Owen O’Connor, MD, PhD, of Columbia University Medical Center in New York, also presented details on the trial at the T-cell Lymphoma Forum. His presentation was followed by a discussion with meeting attendees about applying the trial results to clinical practice.
CD30 expression
One of the issues discussed was the importance of CD30 expression in deciding which patients should receive BV.
For a patient to be eligible for ECHELON-2, the diagnostic biopsy had to show at least 10% of the neoplastic cells were CD30-positive. However, the Food and Drug Administration (FDA) has not made a similar requirement for prescribing BV. PTCL patients with any level of CD30 expression are eligible for treatment with BV-CHP, according to the FDA.
“[I]t’s still a matter of great debate and controversy as to whether we have good enough data to suggest that there’s a threshold effect with regard to the expression of CD30 and responsiveness or sensitivity to brentuximab vedotin,” Dr. O’Connor said.
“This has been an issue from the very first day with this drug, which is, ‘Just how much CD30 do you need to get a response?’ I can’t speak on behalf of the FDA, but I think they are not absolutely convinced that there’s a threshold. They take [CD30-] positive as ‘good enough’ across the board.”
“The FDA has said, ‘The data we’ve seen says there’s a lot of heterogeneity [with biopsies].’ You may do a biopsy and find 30% [of cells are CD30-positive], and you may do another biopsy [in the same patient] and find less than 10%. I don’t think the regulatory agencies are convinced that a single biopsy looking at CD30 ... is representative of the entire tumor burden.”
Andrei Shustov, MD, an ECHELON-2 investigator from the University of Washington in Seattle, questioned whether CD30 expression should be considered when deciding on the use of BV in PTCL.
“Is CD30 staining relevant at all, or should we default back to studies, say, in colon cancer where we didn’t even care about EGFR because we might be missing it by current techniques?” Dr. Shustov asked. “Should we even worry about CD30 expression ... because we cannot reliably detect low levels of CD30?”
Some attendees echoed this sentiment, questioning the utility of assessing CD30 expression. Other attendees said they would defer to the trial data and only treat patients with BV-CHP if they had at least 10% CD30.
PTCL subtypes
Meeting attendees also discussed the value of BV in different PTCL subtypes.
At the request of European regulatory agencies, ECHELON-2 was largely focused on patients with sALCL. They made up 70% of the total trial population, while 16% of patients had PTCL-NOS, 12% had AITL, and a small number of patients had other subtypes. These numbers meant ECHELON-2 was not powered to determine differences in OS or PFS in non-sALCL subtypes.
As a result, some attendees expressed concerns about using BV-CHP to treat PTCL-NOS or AITL. They argued that it wasn’t clear whether patients with these subtypes would derive more benefit from BV-CHP, CHOP, or CHOP plus etoposide (CHOEP).
Other attendees said they would feel comfortable using BV-CHP in patients with PTCL-NOS or AITL based on ECHELON-2 results.
CHOP vs. CHOEP
The use of CHOP in ECHELON-2 was another point of discussion. Some attendees said CHOEP should have been used as the comparator instead.
A few individuals mentioned retrospective data suggesting CHOEP may confer a benefit over CHOP in PTCL (Blood. 2010 Nov 4;116[18]:3418-25).
Marek Trneny, MD, of Charles University General Hospital in Prague, referenced new data from the Czech National Lymphoma Registry, which showed that patients newly diagnosed with PTCL had superior PFS and OS when they received CHOEP rather than CHOP.
Based on these findings, Dr. Trneny said he would consider treating CD30-positive PTCL patients with CHOEP plus BV rather than BV-CHP.
However, most other attendees said they would not consider adding BV to CHOEP due to the absence of clinical trial data supporting this approach.
Some attendees did say they would use CHOEP instead of BV-CHP, particularly in patients with PTCL-NOS or AITL and in patients with CD30 expression below 10%.
ECHELON-2 was funded by Seattle Genetics and Millennium Pharmaceuticals, a wholly owned subsidiary of Takeda Pharmaceutical Company.
Dr. O’Connor and Dr. Shustov were investigators on ECHELON-2. Dr. O’Connor is a cochair of the T-cell Lymphoma Forum. The T-cell Lymphoma Forum is organized by Jonathan Wood & Associates, which is owned by the same company as this news organization.
LA JOLLA, CALIF. – Results from the ECHELON-2 trial led to the U.S. approval of brentuximab vedotin (BV) in combination with cyclophosphamide, doxorubicin, and prednisone (CHP), but there are still questions about how to apply the trial results to practice.

At the annual T-cell Lymphoma Forum, trial investigators and other physicians debated the best use of this combination.
BV-CHP is approved to treat patients with previously untreated systemic anaplastic large-cell lymphoma (sALCL) or other CD30-expressing peripheral T-cell lymphomas (PTCLs), including angioimmunoblastic T-cell lymphoma (AITL) and PTCL not otherwise specified (NOS).
Patients who received BV-CHP in ECHELON-2 had superior progression-free survival (PFS) and overall survival (OS) compared to patients who received cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP).
These results were initially presented at the 2018 annual meeting of the American Society of Hematology and simultaneously published in The Lancet (2019 Jan 19;393[10168]:229-40).
ECHELON-2 investigator Owen O’Connor, MD, PhD, of Columbia University Medical Center in New York, also presented details on the trial at the T-cell Lymphoma Forum. His presentation was followed by a discussion with meeting attendees about applying the trial results to clinical practice.
CD30 expression
One of the issues discussed was the importance of CD30 expression in deciding which patients should receive BV.
For a patient to be eligible for ECHELON-2, the diagnostic biopsy had to show at least 10% of the neoplastic cells were CD30-positive. However, the Food and Drug Administration (FDA) has not made a similar requirement for prescribing BV. PTCL patients with any level of CD30 expression are eligible for treatment with BV-CHP, according to the FDA.
“[I]t’s still a matter of great debate and controversy as to whether we have good enough data to suggest that there’s a threshold effect with regard to the expression of CD30 and responsiveness or sensitivity to brentuximab vedotin,” Dr. O’Connor said.
“This has been an issue from the very first day with this drug, which is, ‘Just how much CD30 do you need to get a response?’ I can’t speak on behalf of the FDA, but I think they are not absolutely convinced that there’s a threshold. They take [CD30-] positive as ‘good enough’ across the board.”
“The FDA has said, ‘The data we’ve seen says there’s a lot of heterogeneity [with biopsies].’ You may do a biopsy and find 30% [of cells are CD30-positive], and you may do another biopsy [in the same patient] and find less than 10%. I don’t think the regulatory agencies are convinced that a single biopsy looking at CD30 ... is representative of the entire tumor burden.”
Andrei Shustov, MD, an ECHELON-2 investigator from the University of Washington in Seattle, questioned whether CD30 expression should be considered when deciding on the use of BV in PTCL.
“Is CD30 staining relevant at all, or should we default back to studies, say, in colon cancer where we didn’t even care about EGFR because we might be missing it by current techniques?” Dr. Shustov asked. “Should we even worry about CD30 expression ... because we cannot reliably detect low levels of CD30?”
Some attendees echoed this sentiment, questioning the utility of assessing CD30 expression. Other attendees said they would defer to the trial data and only treat patients with BV-CHP if they had at least 10% CD30.
PTCL subtypes
Meeting attendees also discussed the value of BV in different PTCL subtypes.
At the request of European regulatory agencies, ECHELON-2 was largely focused on patients with sALCL. They made up 70% of the total trial population, while 16% of patients had PTCL-NOS, 12% had AITL, and a small number of patients had other subtypes. These numbers meant ECHELON-2 was not powered to determine differences in OS or PFS in non-sALCL subtypes.
As a result, some attendees expressed concerns about using BV-CHP to treat PTCL-NOS or AITL. They argued that it wasn’t clear whether patients with these subtypes would derive more benefit from BV-CHP, CHOP, or CHOP plus etoposide (CHOEP).
Other attendees said they would feel comfortable using BV-CHP in patients with PTCL-NOS or AITL based on ECHELON-2 results.
CHOP vs. CHOEP
The use of CHOP in ECHELON-2 was another point of discussion. Some attendees said CHOEP should have been used as the comparator instead.
A few individuals mentioned retrospective data suggesting CHOEP may confer a benefit over CHOP in PTCL (Blood. 2010 Nov 4;116[18]:3418-25).
Marek Trneny, MD, of Charles University General Hospital in Prague, referenced new data from the Czech National Lymphoma Registry, which showed that patients newly diagnosed with PTCL had superior PFS and OS when they received CHOEP rather than CHOP.
Based on these findings, Dr. Trneny said he would consider treating CD30-positive PTCL patients with CHOEP plus BV rather than BV-CHP.
However, most other attendees said they would not consider adding BV to CHOEP due to the absence of clinical trial data supporting this approach.
Some attendees did say they would use CHOEP instead of BV-CHP, particularly in patients with PTCL-NOS or AITL and in patients with CD30 expression below 10%.
ECHELON-2 was funded by Seattle Genetics and Millennium Pharmaceuticals, a wholly owned subsidiary of Takeda Pharmaceutical Company.
Dr. O’Connor and Dr. Shustov were investigators on ECHELON-2. Dr. O’Connor is a cochair of the T-cell Lymphoma Forum. The T-cell Lymphoma Forum is organized by Jonathan Wood & Associates, which is owned by the same company as this news organization.
EXPERT ANALYSIS FROM TCLF 2019
(Sponsored) Video Roundtable: Basal Insulin Pharmacology

Pharmacokinetic (PK) and pharmacodynamic (PD) properties of drugs form the basis for the development of drugs used in everyday clinical practice, such as commonly used insulin products. PK measures the concentration of a drug in the body, and reflects the rates and amounts absorbed and processed. PD is the biologic effect of a drug in the body, including the time-course of when the biologic effect starts, peaks, and ends. While the determination of PK/PD parameters is important and foundational for the development of different insulin products, studies are often complex and can be difficult to translate into real-world clinical practice. In this roundtable, the speakers discuss PK/PD concepts, focusing on the differentiation of basal insulin analogs and their use in individualized diabetes therapy.
First, the speakers discuss the euglycemic glucose clamp methodology—the standard technique for evaluating PK/PD of insulin—including how it is performed, what parameters it measures (and how they can be interpreted), and its limitations.
Next, the speakers discuss how PK/PD impacts drug development, with particular focus on PK/PD studies used in the development of the second-generation basal insulin analogs insulin glargine 300 U/mL (Gla-300) and insulin degludec.
Finally, the speakers discuss how PK/PD data translate into clinical practice, including the relationship between PK/PD and drug efficacy and safety, and how it influences dosing strategies, hypoglycemia risk, and patient education. Further, the speakers discuss how the PK/PD profile of basal insulins can inform primary care providers when selecting appropriate individualized therapy for patients.
Financial support provided by Sanofi US, Inc.

Pharmacokinetic (PK) and pharmacodynamic (PD) properties of drugs form the basis for the development of drugs used in everyday clinical practice, such as commonly used insulin products. PK measures the concentration of a drug in the body, and reflects the rates and amounts absorbed and processed. PD is the biologic effect of a drug in the body, including the time-course of when the biologic effect starts, peaks, and ends. While the determination of PK/PD parameters is important and foundational for the development of different insulin products, studies are often complex and can be difficult to translate into real-world clinical practice. In this roundtable, the speakers discuss PK/PD concepts, focusing on the differentiation of basal insulin analogs and their use in individualized diabetes therapy.
First, the speakers discuss the euglycemic glucose clamp methodology—the standard technique for evaluating PK/PD of insulin—including how it is performed, what parameters it measures (and how they can be interpreted), and its limitations.
Next, the speakers discuss how PK/PD impacts drug development, with particular focus on PK/PD studies used in the development of the second-generation basal insulin analogs insulin glargine 300 U/mL (Gla-300) and insulin degludec.
Finally, the speakers discuss how PK/PD data translate into clinical practice, including the relationship between PK/PD and drug efficacy and safety, and how it influences dosing strategies, hypoglycemia risk, and patient education. Further, the speakers discuss how the PK/PD profile of basal insulins can inform primary care providers when selecting appropriate individualized therapy for patients.
Financial support provided by Sanofi US, Inc.

Pharmacokinetic (PK) and pharmacodynamic (PD) properties of drugs form the basis for the development of drugs used in everyday clinical practice, such as commonly used insulin products. PK measures the concentration of a drug in the body, and reflects the rates and amounts absorbed and processed. PD is the biologic effect of a drug in the body, including the time-course of when the biologic effect starts, peaks, and ends. While the determination of PK/PD parameters is important and foundational for the development of different insulin products, studies are often complex and can be difficult to translate into real-world clinical practice. In this roundtable, the speakers discuss PK/PD concepts, focusing on the differentiation of basal insulin analogs and their use in individualized diabetes therapy.
First, the speakers discuss the euglycemic glucose clamp methodology—the standard technique for evaluating PK/PD of insulin—including how it is performed, what parameters it measures (and how they can be interpreted), and its limitations.
Next, the speakers discuss how PK/PD impacts drug development, with particular focus on PK/PD studies used in the development of the second-generation basal insulin analogs insulin glargine 300 U/mL (Gla-300) and insulin degludec.
Finally, the speakers discuss how PK/PD data translate into clinical practice, including the relationship between PK/PD and drug efficacy and safety, and how it influences dosing strategies, hypoglycemia risk, and patient education. Further, the speakers discuss how the PK/PD profile of basal insulins can inform primary care providers when selecting appropriate individualized therapy for patients.
Financial support provided by Sanofi US, Inc.
Bedbugs in the Workplace