User login
VIDEO: Congress examines observation status policies
Medicare rules surrounding payment for observation status aren’t well understood outside of hospital walls, but the topic has started to make its way to Capitol Hill.
As Medicare patients increasingly get hit with large outpatient bills for hospitals stays that they thought were covered as inpatient care, lawmakers are taking notice.
Over the summer, members of Congress held hearings looking at how Medicare’s payment rules are affecting patients’ pocketbooks and their quality of care. Dr. Ann M. Sheehy, a hospitalist who has researched the use of observation status, testified before the Senate Special Committee on Aging. Watch her full testimony online.
Medicare rules surrounding payment for observation status aren’t well understood outside of hospital walls, but the topic has started to make its way to Capitol Hill.
As Medicare patients increasingly get hit with large outpatient bills for hospitals stays that they thought were covered as inpatient care, lawmakers are taking notice.
Over the summer, members of Congress held hearings looking at how Medicare’s payment rules are affecting patients’ pocketbooks and their quality of care. Dr. Ann M. Sheehy, a hospitalist who has researched the use of observation status, testified before the Senate Special Committee on Aging. Watch her full testimony online.
Medicare rules surrounding payment for observation status aren’t well understood outside of hospital walls, but the topic has started to make its way to Capitol Hill.
As Medicare patients increasingly get hit with large outpatient bills for hospitals stays that they thought were covered as inpatient care, lawmakers are taking notice.
Over the summer, members of Congress held hearings looking at how Medicare’s payment rules are affecting patients’ pocketbooks and their quality of care. Dr. Ann M. Sheehy, a hospitalist who has researched the use of observation status, testified before the Senate Special Committee on Aging. Watch her full testimony online.
Cancer Survivorship Care Plans
Julie Beck, MSN, MPH, APRN, presented during one of the breakout sessions at the AVAHO 2014 Meeting in Portland, Oregon. Beck's presentation focuses on the goals, problems encountered, and tools used when the VA Connecticut Healthcare System was implementing a lung survivorship program for patients.
According to Beck, implementation of a survivorship care plan across all disciplines in VA facilities can greatly improve health care provider understanding of patient needs and help better guide treatment.
Watch this video to find learn more about the program at the Connecticut VA:
Julie Beck, MSN, MPH, APRN, presented during one of the breakout sessions at the AVAHO 2014 Meeting in Portland, Oregon. Beck's presentation focuses on the goals, problems encountered, and tools used when the VA Connecticut Healthcare System was implementing a lung survivorship program for patients.
According to Beck, implementation of a survivorship care plan across all disciplines in VA facilities can greatly improve health care provider understanding of patient needs and help better guide treatment.
Watch this video to find learn more about the program at the Connecticut VA:
Julie Beck, MSN, MPH, APRN, presented during one of the breakout sessions at the AVAHO 2014 Meeting in Portland, Oregon. Beck's presentation focuses on the goals, problems encountered, and tools used when the VA Connecticut Healthcare System was implementing a lung survivorship program for patients.
According to Beck, implementation of a survivorship care plan across all disciplines in VA facilities can greatly improve health care provider understanding of patient needs and help better guide treatment.
Watch this video to find learn more about the program at the Connecticut VA:
VIDEO: Low recurrence with high HER2 expression and a GP2 peptide vaccine
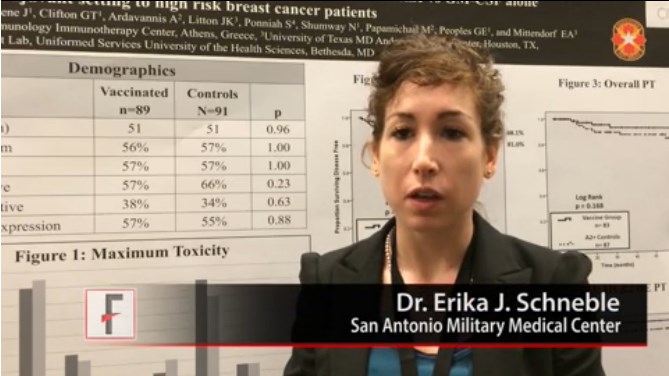
SAN FRANCISCO – A GP2 peptide vaccine to prevent breast cancer recurrence appears most effective in women whose tumors had the highest HER2 overexpression. In a video interview, Dr. Erika J. Schneble of the San Antonio (Tex.) Military Medical Center describes the phase II adjuvant vaccine study she presented at a breast cancer symposium sponsored by the American Society of Clinical Oncology.
Results of an intent-to-treat analysis of all patients showed that 88% of 89 vaccinated women and 81% of 91 patients in the control group remained disease free after a median follow-up of 34 months (P = .43). Among patients who completed treatment, disease-free survival rates were 94% among 83 patients in the vaccine group and 85% among 87 patients in the control group, the investigators reported (P = .17).
In prespecified subgroup analyses based on the level of expression of human epidermal growth factor receptor 2 (HER2), the greatest potential effect was seen in those with the highest HER2 overexpression. In this subgroup, disease-free survival rates were 94% in 51 patients in the vaccine group and 89% in 50 patients in the control group, in an intent-to-treat analysis (P = .86). Excluding two of these patients who developed early recurrences during the primary vaccine series and one patient who developed a non-breast malignancy, however, no recurrences were seen in the vaccine group. Disease-free survival rates were 100% in 48 women in the vaccine group and 89% in 50 women in the control group (P = .08).
Dr. Schneble conducted the 180-patient trial with primary investigator Dr. Elizabeth A. Mittendorf, a surgical oncologist at the University of Texas MD Anderson Cancer Center, Houston, and her associates.
Also in the video, Dr. Hope S. Rugo, director of the Breast Oncology Clinical Trials Program at the University of California, San Francisco, gives her perspective on the study findings. She was not involved with the trial.
Dr. Schneble reporting having no financial disclosures. Dr. Mittendorf’s institution receives per-patient support to enroll patients in vaccine trials sponsored by Galena Biopharma, Antigen Express, and Norwell. One of their coinvestigators has partial inventor rights to GP2. Dr. Rugo disclosed financial associations with Genomic Health, Plexxikon, Merck, and Novartis. She is on the scientific advisory board of Galena Biopharma.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @sherryboschert
SAN FRANCISCO – A GP2 peptide vaccine to prevent breast cancer recurrence appears most effective in women whose tumors had the highest HER2 overexpression. In a video interview, Dr. Erika J. Schneble of the San Antonio (Tex.) Military Medical Center describes the phase II adjuvant vaccine study she presented at a breast cancer symposium sponsored by the American Society of Clinical Oncology.
Results of an intent-to-treat analysis of all patients showed that 88% of 89 vaccinated women and 81% of 91 patients in the control group remained disease free after a median follow-up of 34 months (P = .43). Among patients who completed treatment, disease-free survival rates were 94% among 83 patients in the vaccine group and 85% among 87 patients in the control group, the investigators reported (P = .17).
In prespecified subgroup analyses based on the level of expression of human epidermal growth factor receptor 2 (HER2), the greatest potential effect was seen in those with the highest HER2 overexpression. In this subgroup, disease-free survival rates were 94% in 51 patients in the vaccine group and 89% in 50 patients in the control group, in an intent-to-treat analysis (P = .86). Excluding two of these patients who developed early recurrences during the primary vaccine series and one patient who developed a non-breast malignancy, however, no recurrences were seen in the vaccine group. Disease-free survival rates were 100% in 48 women in the vaccine group and 89% in 50 women in the control group (P = .08).
Dr. Schneble conducted the 180-patient trial with primary investigator Dr. Elizabeth A. Mittendorf, a surgical oncologist at the University of Texas MD Anderson Cancer Center, Houston, and her associates.
Also in the video, Dr. Hope S. Rugo, director of the Breast Oncology Clinical Trials Program at the University of California, San Francisco, gives her perspective on the study findings. She was not involved with the trial.
Dr. Schneble reporting having no financial disclosures. Dr. Mittendorf’s institution receives per-patient support to enroll patients in vaccine trials sponsored by Galena Biopharma, Antigen Express, and Norwell. One of their coinvestigators has partial inventor rights to GP2. Dr. Rugo disclosed financial associations with Genomic Health, Plexxikon, Merck, and Novartis. She is on the scientific advisory board of Galena Biopharma.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @sherryboschert
SAN FRANCISCO – A GP2 peptide vaccine to prevent breast cancer recurrence appears most effective in women whose tumors had the highest HER2 overexpression. In a video interview, Dr. Erika J. Schneble of the San Antonio (Tex.) Military Medical Center describes the phase II adjuvant vaccine study she presented at a breast cancer symposium sponsored by the American Society of Clinical Oncology.
Results of an intent-to-treat analysis of all patients showed that 88% of 89 vaccinated women and 81% of 91 patients in the control group remained disease free after a median follow-up of 34 months (P = .43). Among patients who completed treatment, disease-free survival rates were 94% among 83 patients in the vaccine group and 85% among 87 patients in the control group, the investigators reported (P = .17).
In prespecified subgroup analyses based on the level of expression of human epidermal growth factor receptor 2 (HER2), the greatest potential effect was seen in those with the highest HER2 overexpression. In this subgroup, disease-free survival rates were 94% in 51 patients in the vaccine group and 89% in 50 patients in the control group, in an intent-to-treat analysis (P = .86). Excluding two of these patients who developed early recurrences during the primary vaccine series and one patient who developed a non-breast malignancy, however, no recurrences were seen in the vaccine group. Disease-free survival rates were 100% in 48 women in the vaccine group and 89% in 50 women in the control group (P = .08).
Dr. Schneble conducted the 180-patient trial with primary investigator Dr. Elizabeth A. Mittendorf, a surgical oncologist at the University of Texas MD Anderson Cancer Center, Houston, and her associates.
Also in the video, Dr. Hope S. Rugo, director of the Breast Oncology Clinical Trials Program at the University of California, San Francisco, gives her perspective on the study findings. She was not involved with the trial.
Dr. Schneble reporting having no financial disclosures. Dr. Mittendorf’s institution receives per-patient support to enroll patients in vaccine trials sponsored by Galena Biopharma, Antigen Express, and Norwell. One of their coinvestigators has partial inventor rights to GP2. Dr. Rugo disclosed financial associations with Genomic Health, Plexxikon, Merck, and Novartis. She is on the scientific advisory board of Galena Biopharma.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @sherryboschert
AT THE ASCO BREAST CANCER SYMPOSIUM
Letizia Leocani, MD, PhD
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
VIDEO: Switch ANCA-associated vasculitis patients to maintenance therapy after 6 months
LAS VEGAS – After 3-6 months of remission induction therapy with high-dose steroids and cyclophosphamide, it’s time to switch ANCA-associated vasculitis patients to maintenance therapy.
Recent studies have shown it’s not necessary to go beyond that point with induction treatment; patients do well by then with less toxic maintenance agents.
Dr. Brian Mandell explained how he and his colleagues at the Cleveland Clinic handle the matter and what they use for maintenance therapy, at the conference held by Global Academy for Medical Education. Global Academy for Medical Education and this news organization are owned by Frontline Medical Communications.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
LAS VEGAS – After 3-6 months of remission induction therapy with high-dose steroids and cyclophosphamide, it’s time to switch ANCA-associated vasculitis patients to maintenance therapy.
Recent studies have shown it’s not necessary to go beyond that point with induction treatment; patients do well by then with less toxic maintenance agents.
Dr. Brian Mandell explained how he and his colleagues at the Cleveland Clinic handle the matter and what they use for maintenance therapy, at the conference held by Global Academy for Medical Education. Global Academy for Medical Education and this news organization are owned by Frontline Medical Communications.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
LAS VEGAS – After 3-6 months of remission induction therapy with high-dose steroids and cyclophosphamide, it’s time to switch ANCA-associated vasculitis patients to maintenance therapy.
Recent studies have shown it’s not necessary to go beyond that point with induction treatment; patients do well by then with less toxic maintenance agents.
Dr. Brian Mandell explained how he and his colleagues at the Cleveland Clinic handle the matter and what they use for maintenance therapy, at the conference held by Global Academy for Medical Education. Global Academy for Medical Education and this news organization are owned by Frontline Medical Communications.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE ANNUAL PERSPECTIVES IN RHEUMATIC DISEASES
VIDEO: ASCO Breast Cancer Symposium final wrap-up with Dr. Hope S. Rugo
SAN FRANCISCO – Can we figure out who doesn’t need excision of atypical ductal hyperplasia and intraepithelial neoplasia? Should we screen all women with low–estrogen receptor positivity (1%-9%) for BRCA mutations? Dr. Hope S. Rugo discusses these questions and others that came up during the final sessions at the breast cancer symposium sponsored by the American Society of Clinical Oncology.
The meeting closed out with discussions of evolving standards in the treatment of breast cancer, how to apply these standards to clinical practice, and talks on reversing hormone resistance, including new agents to improve response to hormone therapy.
Dr. Rugo, director of the Breast Oncology Clinical Trials Program at the University of California, San Francisco, also summarizes data presented in the final presentations, including results from a randomized phase II trial of a GP2 vaccine in the adjuvant setting for high-risk breast cancer patients, suggesting that it may reduce recurrence rates in some women with HER2-positive cancer. “It’s very encouraging because we’ve been sort of barking up the vaccine alley for a long, long time and not gotten anywhere. I think it’s quite encouraging data,” she says.
Dr. Rugo disclosed financial associations with Genomic Health, Plexxikon, Merck, and Novartis. She is on the scientific advisory board of Galena Biopharma, which is developing the breast cancer vaccines.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @sherryboschert
SAN FRANCISCO – Can we figure out who doesn’t need excision of atypical ductal hyperplasia and intraepithelial neoplasia? Should we screen all women with low–estrogen receptor positivity (1%-9%) for BRCA mutations? Dr. Hope S. Rugo discusses these questions and others that came up during the final sessions at the breast cancer symposium sponsored by the American Society of Clinical Oncology.
The meeting closed out with discussions of evolving standards in the treatment of breast cancer, how to apply these standards to clinical practice, and talks on reversing hormone resistance, including new agents to improve response to hormone therapy.
Dr. Rugo, director of the Breast Oncology Clinical Trials Program at the University of California, San Francisco, also summarizes data presented in the final presentations, including results from a randomized phase II trial of a GP2 vaccine in the adjuvant setting for high-risk breast cancer patients, suggesting that it may reduce recurrence rates in some women with HER2-positive cancer. “It’s very encouraging because we’ve been sort of barking up the vaccine alley for a long, long time and not gotten anywhere. I think it’s quite encouraging data,” she says.
Dr. Rugo disclosed financial associations with Genomic Health, Plexxikon, Merck, and Novartis. She is on the scientific advisory board of Galena Biopharma, which is developing the breast cancer vaccines.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @sherryboschert
SAN FRANCISCO – Can we figure out who doesn’t need excision of atypical ductal hyperplasia and intraepithelial neoplasia? Should we screen all women with low–estrogen receptor positivity (1%-9%) for BRCA mutations? Dr. Hope S. Rugo discusses these questions and others that came up during the final sessions at the breast cancer symposium sponsored by the American Society of Clinical Oncology.
The meeting closed out with discussions of evolving standards in the treatment of breast cancer, how to apply these standards to clinical practice, and talks on reversing hormone resistance, including new agents to improve response to hormone therapy.
Dr. Rugo, director of the Breast Oncology Clinical Trials Program at the University of California, San Francisco, also summarizes data presented in the final presentations, including results from a randomized phase II trial of a GP2 vaccine in the adjuvant setting for high-risk breast cancer patients, suggesting that it may reduce recurrence rates in some women with HER2-positive cancer. “It’s very encouraging because we’ve been sort of barking up the vaccine alley for a long, long time and not gotten anywhere. I think it’s quite encouraging data,” she says.
Dr. Rugo disclosed financial associations with Genomic Health, Plexxikon, Merck, and Novartis. She is on the scientific advisory board of Galena Biopharma, which is developing the breast cancer vaccines.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @sherryboschert
AT THE ASCO BREAST CANCER SYMPOSIUM
VIDEO: A fib screening finds 5% of elderly undiagnosed
BARCELONA – Roughly 5% of people aged 75 years or older have undiagnosed atrial fibrillation, based on a population-based screening study in Sweden that has assessed nearly 7,000 people, Dr. Mårten Rosenqvist said during an interview at the annual congress of the European Society of Cardiology.
Once diagnosed with atrial fibrillation, all these people immediately qualified for anticoagulant treatment because of their age-related stroke risk. The StrokeStop study will follow all the screened people for 5 years, as well as a concurrently assembled cohort of unscreened controls, to determine the benefit from screening for preventing strokes. “If we can reduce the rate of stroke, it would be a reason to implement a national atrial fibrillation screening program” for all people aged 75 years and older, said Dr. Rosenqvist, professor of cardiology at the Karolinska Institute in Stockholm.
Dr. Rosenqvist said that he is a consultant to Zenicor, a company that markets an ECG-based device for diagnosing atrial fibrillation being used in the StrokeStop study. He also is a consultant to several drug companies.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter@mitchelzoler
BARCELONA – Roughly 5% of people aged 75 years or older have undiagnosed atrial fibrillation, based on a population-based screening study in Sweden that has assessed nearly 7,000 people, Dr. Mårten Rosenqvist said during an interview at the annual congress of the European Society of Cardiology.
Once diagnosed with atrial fibrillation, all these people immediately qualified for anticoagulant treatment because of their age-related stroke risk. The StrokeStop study will follow all the screened people for 5 years, as well as a concurrently assembled cohort of unscreened controls, to determine the benefit from screening for preventing strokes. “If we can reduce the rate of stroke, it would be a reason to implement a national atrial fibrillation screening program” for all people aged 75 years and older, said Dr. Rosenqvist, professor of cardiology at the Karolinska Institute in Stockholm.
Dr. Rosenqvist said that he is a consultant to Zenicor, a company that markets an ECG-based device for diagnosing atrial fibrillation being used in the StrokeStop study. He also is a consultant to several drug companies.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter@mitchelzoler
BARCELONA – Roughly 5% of people aged 75 years or older have undiagnosed atrial fibrillation, based on a population-based screening study in Sweden that has assessed nearly 7,000 people, Dr. Mårten Rosenqvist said during an interview at the annual congress of the European Society of Cardiology.
Once diagnosed with atrial fibrillation, all these people immediately qualified for anticoagulant treatment because of their age-related stroke risk. The StrokeStop study will follow all the screened people for 5 years, as well as a concurrently assembled cohort of unscreened controls, to determine the benefit from screening for preventing strokes. “If we can reduce the rate of stroke, it would be a reason to implement a national atrial fibrillation screening program” for all people aged 75 years and older, said Dr. Rosenqvist, professor of cardiology at the Karolinska Institute in Stockholm.
Dr. Rosenqvist said that he is a consultant to Zenicor, a company that markets an ECG-based device for diagnosing atrial fibrillation being used in the StrokeStop study. He also is a consultant to several drug companies.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter@mitchelzoler
AT THE ESC CONGRESS 2014
VIDEO: Screen scleroderma patients annually for pulmonary hypertension
LAS VEGAS – Scleroderma patients should be screened every year for pulmonary hypertension even if they are recently diagnosed and have no respiratory symptoms.
Noninvasive tests can catch pulmonary problems early, and treatments are available to slow the progression of scleroderma. Dr. Dinesh Khanna, director of the University of Michigan’s Scleroderma Program in Ann Arbor, discussed how to perform scleroderma screening at a conference held by Global Academy for Medical Education. Global Academy for Medical Education and this news organization are owned by Frontline Medical Communications.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
LAS VEGAS – Scleroderma patients should be screened every year for pulmonary hypertension even if they are recently diagnosed and have no respiratory symptoms.
Noninvasive tests can catch pulmonary problems early, and treatments are available to slow the progression of scleroderma. Dr. Dinesh Khanna, director of the University of Michigan’s Scleroderma Program in Ann Arbor, discussed how to perform scleroderma screening at a conference held by Global Academy for Medical Education. Global Academy for Medical Education and this news organization are owned by Frontline Medical Communications.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
LAS VEGAS – Scleroderma patients should be screened every year for pulmonary hypertension even if they are recently diagnosed and have no respiratory symptoms.
Noninvasive tests can catch pulmonary problems early, and treatments are available to slow the progression of scleroderma. Dr. Dinesh Khanna, director of the University of Michigan’s Scleroderma Program in Ann Arbor, discussed how to perform scleroderma screening at a conference held by Global Academy for Medical Education. Global Academy for Medical Education and this news organization are owned by Frontline Medical Communications.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Alasdair Coles, MD
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
VIDEO: NCI’s Dr. Steven Rosenberg talks TILs in breast cancer and much more
BETHESDA, MD. – We visited the office of Dr. Steven A. Rosenberg, chief of the Surgery Branch at the National Cancer Institute, and a pioneer of adoptive cell transfer using tumor infiltrating lymphocytes (TILs) in melanoma patients. We asked for his thoughts on recently published work on TILs in breast cancer and on the future of immunotherapy for breast cancer.
He explained that the success of immunotherapy for breast cancer, he believes, lies in being able to identify the mutations driving tumor growth and manipulating the immune system to recognize those mutations. Dr. Rosenberg began studies in July to investigate this hypothesis.
Listen to the wide-ranging discussion with Dr. Rosenberg on issues from the immunogenicity of epithelial tumors to checkpoint inhibitors and the future of chemotherapy.
Dr. Rosenberg had no relevant disclosures.
On Twitter @whitneymcknight
BETHESDA, MD. – We visited the office of Dr. Steven A. Rosenberg, chief of the Surgery Branch at the National Cancer Institute, and a pioneer of adoptive cell transfer using tumor infiltrating lymphocytes (TILs) in melanoma patients. We asked for his thoughts on recently published work on TILs in breast cancer and on the future of immunotherapy for breast cancer.
He explained that the success of immunotherapy for breast cancer, he believes, lies in being able to identify the mutations driving tumor growth and manipulating the immune system to recognize those mutations. Dr. Rosenberg began studies in July to investigate this hypothesis.
Listen to the wide-ranging discussion with Dr. Rosenberg on issues from the immunogenicity of epithelial tumors to checkpoint inhibitors and the future of chemotherapy.
Dr. Rosenberg had no relevant disclosures.
On Twitter @whitneymcknight
BETHESDA, MD. – We visited the office of Dr. Steven A. Rosenberg, chief of the Surgery Branch at the National Cancer Institute, and a pioneer of adoptive cell transfer using tumor infiltrating lymphocytes (TILs) in melanoma patients. We asked for his thoughts on recently published work on TILs in breast cancer and on the future of immunotherapy for breast cancer.
He explained that the success of immunotherapy for breast cancer, he believes, lies in being able to identify the mutations driving tumor growth and manipulating the immune system to recognize those mutations. Dr. Rosenberg began studies in July to investigate this hypothesis.
Listen to the wide-ranging discussion with Dr. Rosenberg on issues from the immunogenicity of epithelial tumors to checkpoint inhibitors and the future of chemotherapy.
Dr. Rosenberg had no relevant disclosures.
On Twitter @whitneymcknight
AT THE NATIONAL CANCER INSTITUTE