User login
Considering the true costs of clinical trials
This transcript has been edited for clarity.
We need to think about the ways that participating in clinical trials results in increased out-of-pocket costs to our patients and how that limits the ability of marginalized groups to participate. That should be a problem for us.
There are many subtle and some egregious ways that participating in clinical trials can result in increased costs. We may ask patients to come to the clinic more frequently. That may mean costs for transportation, wear and tear on your car, and gas prices. It may also mean that if you work in a job where you don’t have time off, and if you’re not at work, you don’t get paid. That’s a major hit to your take-home pay.
We also need to take a close and more honest look at our study budgets and what we consider standard of care. Now, this becomes a slippery slope because there are clear recommendations that we would all agree, but there are also differences of practice and differences of opinion.
How often should patients with advanced disease, who clinically are doing well, have scans to evaluate their disease status and look for subtle evidence of progression? Are laboratory studies part of the follow-up in patients in the adjuvant setting? Did you really need a urinalysis in somebody who’s going to be starting chemotherapy? Do you need an EKG if you’re going to be giving them a drug that doesn’t have potential cardiac toxicity, for which QTc prolongation is not a problem?
Those are often included in our clinical trials. In some cases, they might be paid for by the trial. In other cases, they’re billed to the insurance provider, which means they’ll contribute to deductibles and copays will apply. It is very likely that they will cost your patient something out of pocket.
Now, this becomes important because many of our consent forms would specifically say that things that are only done for the study are paid for by the study. How we define standard of care becomes vitally important. These issues have not been linked in this way frequently.
Clinical trials are how we make progress. The more patients who are able to participate in clinical trials, the better it is for all of us and all our future patients.
Kathy D. Miller, MD, is associate director of clinical research and codirector of the breast cancer program at the Melvin and Bren Simon Cancer Center at Indiana University, Indianapolis. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
We need to think about the ways that participating in clinical trials results in increased out-of-pocket costs to our patients and how that limits the ability of marginalized groups to participate. That should be a problem for us.
There are many subtle and some egregious ways that participating in clinical trials can result in increased costs. We may ask patients to come to the clinic more frequently. That may mean costs for transportation, wear and tear on your car, and gas prices. It may also mean that if you work in a job where you don’t have time off, and if you’re not at work, you don’t get paid. That’s a major hit to your take-home pay.
We also need to take a close and more honest look at our study budgets and what we consider standard of care. Now, this becomes a slippery slope because there are clear recommendations that we would all agree, but there are also differences of practice and differences of opinion.
How often should patients with advanced disease, who clinically are doing well, have scans to evaluate their disease status and look for subtle evidence of progression? Are laboratory studies part of the follow-up in patients in the adjuvant setting? Did you really need a urinalysis in somebody who’s going to be starting chemotherapy? Do you need an EKG if you’re going to be giving them a drug that doesn’t have potential cardiac toxicity, for which QTc prolongation is not a problem?
Those are often included in our clinical trials. In some cases, they might be paid for by the trial. In other cases, they’re billed to the insurance provider, which means they’ll contribute to deductibles and copays will apply. It is very likely that they will cost your patient something out of pocket.
Now, this becomes important because many of our consent forms would specifically say that things that are only done for the study are paid for by the study. How we define standard of care becomes vitally important. These issues have not been linked in this way frequently.
Clinical trials are how we make progress. The more patients who are able to participate in clinical trials, the better it is for all of us and all our future patients.
Kathy D. Miller, MD, is associate director of clinical research and codirector of the breast cancer program at the Melvin and Bren Simon Cancer Center at Indiana University, Indianapolis. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
We need to think about the ways that participating in clinical trials results in increased out-of-pocket costs to our patients and how that limits the ability of marginalized groups to participate. That should be a problem for us.
There are many subtle and some egregious ways that participating in clinical trials can result in increased costs. We may ask patients to come to the clinic more frequently. That may mean costs for transportation, wear and tear on your car, and gas prices. It may also mean that if you work in a job where you don’t have time off, and if you’re not at work, you don’t get paid. That’s a major hit to your take-home pay.
We also need to take a close and more honest look at our study budgets and what we consider standard of care. Now, this becomes a slippery slope because there are clear recommendations that we would all agree, but there are also differences of practice and differences of opinion.
How often should patients with advanced disease, who clinically are doing well, have scans to evaluate their disease status and look for subtle evidence of progression? Are laboratory studies part of the follow-up in patients in the adjuvant setting? Did you really need a urinalysis in somebody who’s going to be starting chemotherapy? Do you need an EKG if you’re going to be giving them a drug that doesn’t have potential cardiac toxicity, for which QTc prolongation is not a problem?
Those are often included in our clinical trials. In some cases, they might be paid for by the trial. In other cases, they’re billed to the insurance provider, which means they’ll contribute to deductibles and copays will apply. It is very likely that they will cost your patient something out of pocket.
Now, this becomes important because many of our consent forms would specifically say that things that are only done for the study are paid for by the study. How we define standard of care becomes vitally important. These issues have not been linked in this way frequently.
Clinical trials are how we make progress. The more patients who are able to participate in clinical trials, the better it is for all of us and all our future patients.
Kathy D. Miller, MD, is associate director of clinical research and codirector of the breast cancer program at the Melvin and Bren Simon Cancer Center at Indiana University, Indianapolis. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
A step forward in diabetic foot disease management
As we navigate the ever-evolving landscape of diabetic foot disease management, The goal is to create a common language of risk that is easily related from clinician to clinician to patient.
Whatever language we use, though, the problem we face is vast:
- Diabetic foot ulcers affect approximately 18.6 million people worldwide and 1.6 million in the United States each year.
- They are associated with high rates of premature death, with a 5-year mortality rate of 30%. This rate is greater than 70% for those with above-foot amputations, worse than all but the most aggressive cancers.
- The direct costs of treating diabetic foot ulcers in the United States is estimated at $9 billion-$13 billion annually.
- Over 550 million people worldwide have diabetes, with 18.6 million developing foot ulcers annually. Up to 34% of those with diabetes will develop a foot ulcer.
- About 20% of those with a diabetic foot ulcer will undergo amputation, a major cause of which is infection, which affects 50% of foot ulcers.
- Up to 20% of those with a foot ulcer require hospitalization, with 15%-20% undergoing amputation. Inequities exist in diabetes-related foot complications:
- –Rates of major amputation are higher in non-Hispanic Black, Hispanic, and Native American populations, compared with non-Hispanic White populations.
- –Non-Hispanic Black and Hispanic populations present with more advanced ulcers and peripheral artery disease, and are more likely to undergo amputation without revascularization attempt.
The IWGDF, a multidisciplinary team of international experts, has recently updated its guidelines. This team, comprising endocrinologists, internal medicine physicians, physiatrists, podiatrists, and vascular surgeons from across the globe, has worked tirelessly to provide us with a comprehensive guide to managing diabetes-related foot ulcers.
The updated guidelines address five critical clinical questions, each with up to 13 important outcomes. The systematic review that underpins these guidelines identified 149 eligible studies, assessing 28 different systems. This exhaustive research has led to the development of seven key recommendations that address the clinical questions and consider the existence of different clinical settings.
One of the significant updates in the 2023 guidelines is the recommendation of SINBAD – site, ischemia, neuropathy, bacterial infection, area, and depth – as the priority wound classification system for people with diabetes and a foot ulcer. This system is particularly useful for interprofessional communication, describing each composite variable, and conducting clinical audits using the full score. However, the guidelines also recommend the use of other, more specific assessment systems for infection and peripheral artery disease from the Infectious Diseases Society of America/IWGDF when resources and an appropriate level of expertise exist.
The introduction of the Wound, Ischemia and Foot Infection (WIfI) classification system in the guidelines is also a noteworthy development. This system is crucial in assessing perfusion and the likely benefit of revascularization in a person with diabetes and a foot ulcer. By assessing the level of wound ischemia and infection, we can make informed decisions about the need for vascular intervention, which can significantly affect the patient’s outcome. This can be done simply by classifying each of the three categories of wound, ischemia, or foot infection as none, mild, moderate, or severe. By simplifying the very dynamic comorbidities of tissue loss, ischemia, and infection into a usable and predictive scale, it helps us to communicate risk across disciplines. This has been found to be highly predictive of healing, amputation, and mortality.
We use WIfI every day across our system. An example might include a patient we recently treated:
A 76-year-old woman presented with a wound to her left foot. Her past medical history revealed type 2 diabetes, peripheral neuropathy, and documented peripheral artery disease with prior bilateral femoral-popliteal bypass conducted at an external facility. In addition to gangrenous changes to her fourth toe, she displayed erythema and lymphangitic streaking up her dorsal foot. While she was afebrile, her white cell count was 13,000/mcL. Radiographic examinations did not show signs of osteomyelitis. Noninvasive vascular evaluations revealed an ankle brachial index of 0.4 and a toe pressure of 10 mm Hg. An aortogram with a lower-extremity runoff arteriogram confirmed the obstruction of her left femoral-popliteal bypass.
Taking these results into account, her WIfI score was determined as: wound 2 (moderate), ischemia 3 (severe), foot infection 2 (moderate, no sepsis), translating to a clinical stage 4. This denotes a high risk for major amputation.
Following a team discussion, she was taken to the operating room for an initial debridement of her infection which consisted of a partial fourth ray resection to the level of the mid-metatarsal. Following control of the infection, she received a vascular assessment which ultimately constituted a femoral to distal anterior tibial bypass. Following both of these, she was discharged on a negative-pressure wound therapy device, receiving a split-thickness skin graft 4 weeks later.
The guidelines also emphasize the need for specific training, skills, and experience to ensure the accuracy of the recommended systems for characterizing foot ulcers. The person applying these systems should be appropriately trained and, according to their national or regional standards, should have the knowledge, expertise, and skills necessary to manage people with a diabetes-related foot ulcer.
As we continue to navigate the complexities of diabetes-related foot disease, these guidelines serve as a valuable compass, guiding our decisions and actions. They remind us of the importance of continuous learning, collaboration, and the application of evidence-based practice in our work.
I encourage you to delve into these guidelines. Let’s use them to improve our practice, enhance our communication, and, ultimately, provide better care for our patients.
Dr. Armstrong is professor of surgery, director of limb preservation, University of Southern California, Los Angeles. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
As we navigate the ever-evolving landscape of diabetic foot disease management, The goal is to create a common language of risk that is easily related from clinician to clinician to patient.
Whatever language we use, though, the problem we face is vast:
- Diabetic foot ulcers affect approximately 18.6 million people worldwide and 1.6 million in the United States each year.
- They are associated with high rates of premature death, with a 5-year mortality rate of 30%. This rate is greater than 70% for those with above-foot amputations, worse than all but the most aggressive cancers.
- The direct costs of treating diabetic foot ulcers in the United States is estimated at $9 billion-$13 billion annually.
- Over 550 million people worldwide have diabetes, with 18.6 million developing foot ulcers annually. Up to 34% of those with diabetes will develop a foot ulcer.
- About 20% of those with a diabetic foot ulcer will undergo amputation, a major cause of which is infection, which affects 50% of foot ulcers.
- Up to 20% of those with a foot ulcer require hospitalization, with 15%-20% undergoing amputation. Inequities exist in diabetes-related foot complications:
- –Rates of major amputation are higher in non-Hispanic Black, Hispanic, and Native American populations, compared with non-Hispanic White populations.
- –Non-Hispanic Black and Hispanic populations present with more advanced ulcers and peripheral artery disease, and are more likely to undergo amputation without revascularization attempt.
The IWGDF, a multidisciplinary team of international experts, has recently updated its guidelines. This team, comprising endocrinologists, internal medicine physicians, physiatrists, podiatrists, and vascular surgeons from across the globe, has worked tirelessly to provide us with a comprehensive guide to managing diabetes-related foot ulcers.
The updated guidelines address five critical clinical questions, each with up to 13 important outcomes. The systematic review that underpins these guidelines identified 149 eligible studies, assessing 28 different systems. This exhaustive research has led to the development of seven key recommendations that address the clinical questions and consider the existence of different clinical settings.
One of the significant updates in the 2023 guidelines is the recommendation of SINBAD – site, ischemia, neuropathy, bacterial infection, area, and depth – as the priority wound classification system for people with diabetes and a foot ulcer. This system is particularly useful for interprofessional communication, describing each composite variable, and conducting clinical audits using the full score. However, the guidelines also recommend the use of other, more specific assessment systems for infection and peripheral artery disease from the Infectious Diseases Society of America/IWGDF when resources and an appropriate level of expertise exist.
The introduction of the Wound, Ischemia and Foot Infection (WIfI) classification system in the guidelines is also a noteworthy development. This system is crucial in assessing perfusion and the likely benefit of revascularization in a person with diabetes and a foot ulcer. By assessing the level of wound ischemia and infection, we can make informed decisions about the need for vascular intervention, which can significantly affect the patient’s outcome. This can be done simply by classifying each of the three categories of wound, ischemia, or foot infection as none, mild, moderate, or severe. By simplifying the very dynamic comorbidities of tissue loss, ischemia, and infection into a usable and predictive scale, it helps us to communicate risk across disciplines. This has been found to be highly predictive of healing, amputation, and mortality.
We use WIfI every day across our system. An example might include a patient we recently treated:
A 76-year-old woman presented with a wound to her left foot. Her past medical history revealed type 2 diabetes, peripheral neuropathy, and documented peripheral artery disease with prior bilateral femoral-popliteal bypass conducted at an external facility. In addition to gangrenous changes to her fourth toe, she displayed erythema and lymphangitic streaking up her dorsal foot. While she was afebrile, her white cell count was 13,000/mcL. Radiographic examinations did not show signs of osteomyelitis. Noninvasive vascular evaluations revealed an ankle brachial index of 0.4 and a toe pressure of 10 mm Hg. An aortogram with a lower-extremity runoff arteriogram confirmed the obstruction of her left femoral-popliteal bypass.
Taking these results into account, her WIfI score was determined as: wound 2 (moderate), ischemia 3 (severe), foot infection 2 (moderate, no sepsis), translating to a clinical stage 4. This denotes a high risk for major amputation.
Following a team discussion, she was taken to the operating room for an initial debridement of her infection which consisted of a partial fourth ray resection to the level of the mid-metatarsal. Following control of the infection, she received a vascular assessment which ultimately constituted a femoral to distal anterior tibial bypass. Following both of these, she was discharged on a negative-pressure wound therapy device, receiving a split-thickness skin graft 4 weeks later.
The guidelines also emphasize the need for specific training, skills, and experience to ensure the accuracy of the recommended systems for characterizing foot ulcers. The person applying these systems should be appropriately trained and, according to their national or regional standards, should have the knowledge, expertise, and skills necessary to manage people with a diabetes-related foot ulcer.
As we continue to navigate the complexities of diabetes-related foot disease, these guidelines serve as a valuable compass, guiding our decisions and actions. They remind us of the importance of continuous learning, collaboration, and the application of evidence-based practice in our work.
I encourage you to delve into these guidelines. Let’s use them to improve our practice, enhance our communication, and, ultimately, provide better care for our patients.
Dr. Armstrong is professor of surgery, director of limb preservation, University of Southern California, Los Angeles. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
As we navigate the ever-evolving landscape of diabetic foot disease management, The goal is to create a common language of risk that is easily related from clinician to clinician to patient.
Whatever language we use, though, the problem we face is vast:
- Diabetic foot ulcers affect approximately 18.6 million people worldwide and 1.6 million in the United States each year.
- They are associated with high rates of premature death, with a 5-year mortality rate of 30%. This rate is greater than 70% for those with above-foot amputations, worse than all but the most aggressive cancers.
- The direct costs of treating diabetic foot ulcers in the United States is estimated at $9 billion-$13 billion annually.
- Over 550 million people worldwide have diabetes, with 18.6 million developing foot ulcers annually. Up to 34% of those with diabetes will develop a foot ulcer.
- About 20% of those with a diabetic foot ulcer will undergo amputation, a major cause of which is infection, which affects 50% of foot ulcers.
- Up to 20% of those with a foot ulcer require hospitalization, with 15%-20% undergoing amputation. Inequities exist in diabetes-related foot complications:
- –Rates of major amputation are higher in non-Hispanic Black, Hispanic, and Native American populations, compared with non-Hispanic White populations.
- –Non-Hispanic Black and Hispanic populations present with more advanced ulcers and peripheral artery disease, and are more likely to undergo amputation without revascularization attempt.
The IWGDF, a multidisciplinary team of international experts, has recently updated its guidelines. This team, comprising endocrinologists, internal medicine physicians, physiatrists, podiatrists, and vascular surgeons from across the globe, has worked tirelessly to provide us with a comprehensive guide to managing diabetes-related foot ulcers.
The updated guidelines address five critical clinical questions, each with up to 13 important outcomes. The systematic review that underpins these guidelines identified 149 eligible studies, assessing 28 different systems. This exhaustive research has led to the development of seven key recommendations that address the clinical questions and consider the existence of different clinical settings.
One of the significant updates in the 2023 guidelines is the recommendation of SINBAD – site, ischemia, neuropathy, bacterial infection, area, and depth – as the priority wound classification system for people with diabetes and a foot ulcer. This system is particularly useful for interprofessional communication, describing each composite variable, and conducting clinical audits using the full score. However, the guidelines also recommend the use of other, more specific assessment systems for infection and peripheral artery disease from the Infectious Diseases Society of America/IWGDF when resources and an appropriate level of expertise exist.
The introduction of the Wound, Ischemia and Foot Infection (WIfI) classification system in the guidelines is also a noteworthy development. This system is crucial in assessing perfusion and the likely benefit of revascularization in a person with diabetes and a foot ulcer. By assessing the level of wound ischemia and infection, we can make informed decisions about the need for vascular intervention, which can significantly affect the patient’s outcome. This can be done simply by classifying each of the three categories of wound, ischemia, or foot infection as none, mild, moderate, or severe. By simplifying the very dynamic comorbidities of tissue loss, ischemia, and infection into a usable and predictive scale, it helps us to communicate risk across disciplines. This has been found to be highly predictive of healing, amputation, and mortality.
We use WIfI every day across our system. An example might include a patient we recently treated:
A 76-year-old woman presented with a wound to her left foot. Her past medical history revealed type 2 diabetes, peripheral neuropathy, and documented peripheral artery disease with prior bilateral femoral-popliteal bypass conducted at an external facility. In addition to gangrenous changes to her fourth toe, she displayed erythema and lymphangitic streaking up her dorsal foot. While she was afebrile, her white cell count was 13,000/mcL. Radiographic examinations did not show signs of osteomyelitis. Noninvasive vascular evaluations revealed an ankle brachial index of 0.4 and a toe pressure of 10 mm Hg. An aortogram with a lower-extremity runoff arteriogram confirmed the obstruction of her left femoral-popliteal bypass.
Taking these results into account, her WIfI score was determined as: wound 2 (moderate), ischemia 3 (severe), foot infection 2 (moderate, no sepsis), translating to a clinical stage 4. This denotes a high risk for major amputation.
Following a team discussion, she was taken to the operating room for an initial debridement of her infection which consisted of a partial fourth ray resection to the level of the mid-metatarsal. Following control of the infection, she received a vascular assessment which ultimately constituted a femoral to distal anterior tibial bypass. Following both of these, she was discharged on a negative-pressure wound therapy device, receiving a split-thickness skin graft 4 weeks later.
The guidelines also emphasize the need for specific training, skills, and experience to ensure the accuracy of the recommended systems for characterizing foot ulcers. The person applying these systems should be appropriately trained and, according to their national or regional standards, should have the knowledge, expertise, and skills necessary to manage people with a diabetes-related foot ulcer.
As we continue to navigate the complexities of diabetes-related foot disease, these guidelines serve as a valuable compass, guiding our decisions and actions. They remind us of the importance of continuous learning, collaboration, and the application of evidence-based practice in our work.
I encourage you to delve into these guidelines. Let’s use them to improve our practice, enhance our communication, and, ultimately, provide better care for our patients.
Dr. Armstrong is professor of surgery, director of limb preservation, University of Southern California, Los Angeles. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
The bloated medical record
Until the 19th century there was nothing even resembling our current conception of the medical record. A few physicians may have kept personal notes, observations, and some sketches of their patients primarily to be used in teaching medical students or as part of their own curiosity-driven research. However, around 1800 the Governor Council of the State of New York adopted a proposition that all home doctors should register their medical cases again to be used as an educational tool. By 1830 these registries became annual reporting requirements that included admissions and discharges, treatment results, and expenditures. It shouldn’t surprise you learn that a review of these entries could be linked to a doctor’s prospects for promotion.
In 1919 the American College of Surgeons attempted to standardize its members’ “treatment diaries” to look something more like our current medical records with a history, lab tests, diagnosis, treatment plan, and something akin to daily progress notes. However, as late as the 1970s, when I began primary care practice, there were very few dictates on what our office notes should contain. A few (not including myself) had been trained to use a S.O.A.P. format (Subjective, Objective, Assessment, and Plan) to organize their observations. Back then I viewed my office records as primarily a mnemonic device and only because I had a partner did I make any passing attempt at legibility.
With AI staring us in the face and threatening to expand what has become an already bloated medical record,
Although there was a time when a doctor’s notes simply functioned as a mnemonic, few physicians today practice in isolation and their records must now serve as a vehicle to communicate with covering physicians and consultants.
How detailed do those notes need to be? Do we need more than the hard data – the numbers, the prescriptions, the biometrics, the chronology of the patient’s procedures? As a covering physician or consultant, I’m not really that interested in your subjective observations. It’s not that I don’t trust you, but like any good physician I’m going to take my own history directly from the patient and do my own physical exam. You may have missed something and I owe the patient a fresh look and listen before I render an opinion or prescribe a management plan.
The medical record has become a detailed invoice to be attached to your bill to third-party payers. You need to prove to them that your service has some value. It’s not that the third-party payers don’t trust you ... well maybe that’s the issue. They don’t. So you have to prove to them that you really did something. Since they weren’t in the exam room, you must document that you asked the patient questions, did a thorough exam, and spent a specified amount of time at it. Of course that assumes that there is a direct correlation between the amount of time you spent with the patient and the quality of care. Which isn’t always the case. One sentence merely stating that you are a well-trained professional and did a thorough job doesn’t seem to be good enough. It works for the plumber and the electrician. But again, it’s that trust thing.
Of course there are the licensing and certification organizations that have a legitimate interest in the quality of your work. Because having an observer following you around for a day or two is impractical (which I still think is a good idea), you need to include evidence in your chart that you practice the standard of care by following accepted screening measures and treating according to standard guidelines.
And finally, while we are talking about trust, there is the whole risk management thing – maybe the most potent inflater of medical records. The lawyer-promoted myth “if you didn’t document it, it didn’t happen” encourages doctors to use voluminous verbiage merely to give your lawyer ammunition when you find yourself in a he-said/she-said situation.
Of course all of this needs to be carefully worded because the patient now has and deserves the right to review his or her medical records. And this might be the only good news. AI can be taught to create a medical record that is complete and more easily read and digested by the patient. This could make the records even more voluminous and as more patients become familiar with their own health records they may begin to demand that they become more concise and actually reflect what went on in the visit.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Until the 19th century there was nothing even resembling our current conception of the medical record. A few physicians may have kept personal notes, observations, and some sketches of their patients primarily to be used in teaching medical students or as part of their own curiosity-driven research. However, around 1800 the Governor Council of the State of New York adopted a proposition that all home doctors should register their medical cases again to be used as an educational tool. By 1830 these registries became annual reporting requirements that included admissions and discharges, treatment results, and expenditures. It shouldn’t surprise you learn that a review of these entries could be linked to a doctor’s prospects for promotion.
In 1919 the American College of Surgeons attempted to standardize its members’ “treatment diaries” to look something more like our current medical records with a history, lab tests, diagnosis, treatment plan, and something akin to daily progress notes. However, as late as the 1970s, when I began primary care practice, there were very few dictates on what our office notes should contain. A few (not including myself) had been trained to use a S.O.A.P. format (Subjective, Objective, Assessment, and Plan) to organize their observations. Back then I viewed my office records as primarily a mnemonic device and only because I had a partner did I make any passing attempt at legibility.
With AI staring us in the face and threatening to expand what has become an already bloated medical record,
Although there was a time when a doctor’s notes simply functioned as a mnemonic, few physicians today practice in isolation and their records must now serve as a vehicle to communicate with covering physicians and consultants.
How detailed do those notes need to be? Do we need more than the hard data – the numbers, the prescriptions, the biometrics, the chronology of the patient’s procedures? As a covering physician or consultant, I’m not really that interested in your subjective observations. It’s not that I don’t trust you, but like any good physician I’m going to take my own history directly from the patient and do my own physical exam. You may have missed something and I owe the patient a fresh look and listen before I render an opinion or prescribe a management plan.
The medical record has become a detailed invoice to be attached to your bill to third-party payers. You need to prove to them that your service has some value. It’s not that the third-party payers don’t trust you ... well maybe that’s the issue. They don’t. So you have to prove to them that you really did something. Since they weren’t in the exam room, you must document that you asked the patient questions, did a thorough exam, and spent a specified amount of time at it. Of course that assumes that there is a direct correlation between the amount of time you spent with the patient and the quality of care. Which isn’t always the case. One sentence merely stating that you are a well-trained professional and did a thorough job doesn’t seem to be good enough. It works for the plumber and the electrician. But again, it’s that trust thing.
Of course there are the licensing and certification organizations that have a legitimate interest in the quality of your work. Because having an observer following you around for a day or two is impractical (which I still think is a good idea), you need to include evidence in your chart that you practice the standard of care by following accepted screening measures and treating according to standard guidelines.
And finally, while we are talking about trust, there is the whole risk management thing – maybe the most potent inflater of medical records. The lawyer-promoted myth “if you didn’t document it, it didn’t happen” encourages doctors to use voluminous verbiage merely to give your lawyer ammunition when you find yourself in a he-said/she-said situation.
Of course all of this needs to be carefully worded because the patient now has and deserves the right to review his or her medical records. And this might be the only good news. AI can be taught to create a medical record that is complete and more easily read and digested by the patient. This could make the records even more voluminous and as more patients become familiar with their own health records they may begin to demand that they become more concise and actually reflect what went on in the visit.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Until the 19th century there was nothing even resembling our current conception of the medical record. A few physicians may have kept personal notes, observations, and some sketches of their patients primarily to be used in teaching medical students or as part of their own curiosity-driven research. However, around 1800 the Governor Council of the State of New York adopted a proposition that all home doctors should register their medical cases again to be used as an educational tool. By 1830 these registries became annual reporting requirements that included admissions and discharges, treatment results, and expenditures. It shouldn’t surprise you learn that a review of these entries could be linked to a doctor’s prospects for promotion.
In 1919 the American College of Surgeons attempted to standardize its members’ “treatment diaries” to look something more like our current medical records with a history, lab tests, diagnosis, treatment plan, and something akin to daily progress notes. However, as late as the 1970s, when I began primary care practice, there were very few dictates on what our office notes should contain. A few (not including myself) had been trained to use a S.O.A.P. format (Subjective, Objective, Assessment, and Plan) to organize their observations. Back then I viewed my office records as primarily a mnemonic device and only because I had a partner did I make any passing attempt at legibility.
With AI staring us in the face and threatening to expand what has become an already bloated medical record,
Although there was a time when a doctor’s notes simply functioned as a mnemonic, few physicians today practice in isolation and their records must now serve as a vehicle to communicate with covering physicians and consultants.
How detailed do those notes need to be? Do we need more than the hard data – the numbers, the prescriptions, the biometrics, the chronology of the patient’s procedures? As a covering physician or consultant, I’m not really that interested in your subjective observations. It’s not that I don’t trust you, but like any good physician I’m going to take my own history directly from the patient and do my own physical exam. You may have missed something and I owe the patient a fresh look and listen before I render an opinion or prescribe a management plan.
The medical record has become a detailed invoice to be attached to your bill to third-party payers. You need to prove to them that your service has some value. It’s not that the third-party payers don’t trust you ... well maybe that’s the issue. They don’t. So you have to prove to them that you really did something. Since they weren’t in the exam room, you must document that you asked the patient questions, did a thorough exam, and spent a specified amount of time at it. Of course that assumes that there is a direct correlation between the amount of time you spent with the patient and the quality of care. Which isn’t always the case. One sentence merely stating that you are a well-trained professional and did a thorough job doesn’t seem to be good enough. It works for the plumber and the electrician. But again, it’s that trust thing.
Of course there are the licensing and certification organizations that have a legitimate interest in the quality of your work. Because having an observer following you around for a day or two is impractical (which I still think is a good idea), you need to include evidence in your chart that you practice the standard of care by following accepted screening measures and treating according to standard guidelines.
And finally, while we are talking about trust, there is the whole risk management thing – maybe the most potent inflater of medical records. The lawyer-promoted myth “if you didn’t document it, it didn’t happen” encourages doctors to use voluminous verbiage merely to give your lawyer ammunition when you find yourself in a he-said/she-said situation.
Of course all of this needs to be carefully worded because the patient now has and deserves the right to review his or her medical records. And this might be the only good news. AI can be taught to create a medical record that is complete and more easily read and digested by the patient. This could make the records even more voluminous and as more patients become familiar with their own health records they may begin to demand that they become more concise and actually reflect what went on in the visit.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
The best CRC screening test is still this one
I’m Dr. Kenny Lin. I am a family physician and associate director of the Lancaster General Hospital Family Medicine Residency, and I blog at Common Sense Family Doctor.
I’m 47 years old. Two years ago, when the U.S. Preventive Services Task Force (USPSTF) followed the American Cancer Society and lowered the starting age for colorectal cancer (CRC) screening from 50 to 45, my family physician brought up screening options at a health maintenance visit. Although I had expressed some skepticism about this change when the ACS updated its screening guideline in 2018, I generally follow the USPSTF recommendations in my own clinical practice, so I dutifully selected a test that, fortunately, came out negative.
Not everyone in the primary care community, however, is on board with screening average-risk adults in their late 40s for colorectal cancer. The American Academy of Family Physicians (AAFP) published a notable dissent, arguing that the evidence from modeling studies cited by the USPSTF to support lowering the starting age was insufficient. The AAFP also expressed concern that devoting screening resources to younger adults could come at the expense of improving screening rates in older adults who are at higher risk for CRC and increase health care costs without corresponding benefit.
Now, the American College of Physicians has joined the AAFP by releasing an updated guidance statement for CRC screening that discourages screening asymptomatic, average-risk adults between the ages of 45 and 49. In addition to the uncertainty surrounding benefits of screening adults in this age range, the ACP pointed out that starting screening at age 45, compared with age 50, would increase the number of colonoscopies and colonoscopy complications. My colleagues and I recently published a systematic review estimating that for every 10,000 screening colonoscopies performed, 8 people suffer a bowel perforation and 16 to 36 have severe bleeding requiring hospitalization. One in 3 patients undergoing colonoscopies report minor adverse events such as abdominal pain, bloating, and abdominal discomfort in the first 2 weeks following the procedure.
Other aspects of the ACP guidance differ from other colorectal cancer screening guidelines. Unlike the USPSTF, which made no distinctions between various recommended screening tests, the ACP preferentially endorsed fecal immunochemical or high-sensitivity fecal occult blood testing every 2 years, colonoscopy every 10 years, or flexible sigmoidoscopy every 10 years plus a fecal immunochemical test every 2 years. That leaves out stool DNA testing, which my patients increasingly request because they have seen television or online advertisements, and newer blood tests that detect methylation sequences in circulating tumor DNA.
Perhaps most controversial is the ACP’s suggestion that it is probably reasonable for some adults to start screening later than age 50 or undergo screening at longer intervals than currently recommended (for example, colonoscopy every 15 years). Recent data support extending the interval to repeat screening colonoscopy in selected populations; a large cross-sectional study found a low prevalence of advanced adenomas and colorectal cancers in colonoscopies performed 10 or more years after an initial negative colonoscopy, particularly in women and younger patients without gastrointestinal symptoms. A prominent BMJ guideline suggests that patients need not be screened until their estimated 15-year CRC risk is greater than 3% (which most people do not reach until their 60s) and then only need a single sigmoidoscopy or colonoscopy.
Despite some departures from other guidelines, it’s worth emphasizing that the ACP guideline agrees that screening for CRC is generally worthwhile between the ages of 50 and 75 years. On that front, we in primary care have more work to do; the Centers for Disease Control and Prevention estimates that 28% of American adults older than 50 are not up-to-date on CRC screening. And despite some recent debate about the relative benefits and harms of screening colonoscopy, compared with less invasive fecal tests, gastroenterologists and family physicians can agree that the best screening test is the test that gets done.
A version of this article first appeared on Medscape.com.
I’m Dr. Kenny Lin. I am a family physician and associate director of the Lancaster General Hospital Family Medicine Residency, and I blog at Common Sense Family Doctor.
I’m 47 years old. Two years ago, when the U.S. Preventive Services Task Force (USPSTF) followed the American Cancer Society and lowered the starting age for colorectal cancer (CRC) screening from 50 to 45, my family physician brought up screening options at a health maintenance visit. Although I had expressed some skepticism about this change when the ACS updated its screening guideline in 2018, I generally follow the USPSTF recommendations in my own clinical practice, so I dutifully selected a test that, fortunately, came out negative.
Not everyone in the primary care community, however, is on board with screening average-risk adults in their late 40s for colorectal cancer. The American Academy of Family Physicians (AAFP) published a notable dissent, arguing that the evidence from modeling studies cited by the USPSTF to support lowering the starting age was insufficient. The AAFP also expressed concern that devoting screening resources to younger adults could come at the expense of improving screening rates in older adults who are at higher risk for CRC and increase health care costs without corresponding benefit.
Now, the American College of Physicians has joined the AAFP by releasing an updated guidance statement for CRC screening that discourages screening asymptomatic, average-risk adults between the ages of 45 and 49. In addition to the uncertainty surrounding benefits of screening adults in this age range, the ACP pointed out that starting screening at age 45, compared with age 50, would increase the number of colonoscopies and colonoscopy complications. My colleagues and I recently published a systematic review estimating that for every 10,000 screening colonoscopies performed, 8 people suffer a bowel perforation and 16 to 36 have severe bleeding requiring hospitalization. One in 3 patients undergoing colonoscopies report minor adverse events such as abdominal pain, bloating, and abdominal discomfort in the first 2 weeks following the procedure.
Other aspects of the ACP guidance differ from other colorectal cancer screening guidelines. Unlike the USPSTF, which made no distinctions between various recommended screening tests, the ACP preferentially endorsed fecal immunochemical or high-sensitivity fecal occult blood testing every 2 years, colonoscopy every 10 years, or flexible sigmoidoscopy every 10 years plus a fecal immunochemical test every 2 years. That leaves out stool DNA testing, which my patients increasingly request because they have seen television or online advertisements, and newer blood tests that detect methylation sequences in circulating tumor DNA.
Perhaps most controversial is the ACP’s suggestion that it is probably reasonable for some adults to start screening later than age 50 or undergo screening at longer intervals than currently recommended (for example, colonoscopy every 15 years). Recent data support extending the interval to repeat screening colonoscopy in selected populations; a large cross-sectional study found a low prevalence of advanced adenomas and colorectal cancers in colonoscopies performed 10 or more years after an initial negative colonoscopy, particularly in women and younger patients without gastrointestinal symptoms. A prominent BMJ guideline suggests that patients need not be screened until their estimated 15-year CRC risk is greater than 3% (which most people do not reach until their 60s) and then only need a single sigmoidoscopy or colonoscopy.
Despite some departures from other guidelines, it’s worth emphasizing that the ACP guideline agrees that screening for CRC is generally worthwhile between the ages of 50 and 75 years. On that front, we in primary care have more work to do; the Centers for Disease Control and Prevention estimates that 28% of American adults older than 50 are not up-to-date on CRC screening. And despite some recent debate about the relative benefits and harms of screening colonoscopy, compared with less invasive fecal tests, gastroenterologists and family physicians can agree that the best screening test is the test that gets done.
A version of this article first appeared on Medscape.com.
I’m Dr. Kenny Lin. I am a family physician and associate director of the Lancaster General Hospital Family Medicine Residency, and I blog at Common Sense Family Doctor.
I’m 47 years old. Two years ago, when the U.S. Preventive Services Task Force (USPSTF) followed the American Cancer Society and lowered the starting age for colorectal cancer (CRC) screening from 50 to 45, my family physician brought up screening options at a health maintenance visit. Although I had expressed some skepticism about this change when the ACS updated its screening guideline in 2018, I generally follow the USPSTF recommendations in my own clinical practice, so I dutifully selected a test that, fortunately, came out negative.
Not everyone in the primary care community, however, is on board with screening average-risk adults in their late 40s for colorectal cancer. The American Academy of Family Physicians (AAFP) published a notable dissent, arguing that the evidence from modeling studies cited by the USPSTF to support lowering the starting age was insufficient. The AAFP also expressed concern that devoting screening resources to younger adults could come at the expense of improving screening rates in older adults who are at higher risk for CRC and increase health care costs without corresponding benefit.
Now, the American College of Physicians has joined the AAFP by releasing an updated guidance statement for CRC screening that discourages screening asymptomatic, average-risk adults between the ages of 45 and 49. In addition to the uncertainty surrounding benefits of screening adults in this age range, the ACP pointed out that starting screening at age 45, compared with age 50, would increase the number of colonoscopies and colonoscopy complications. My colleagues and I recently published a systematic review estimating that for every 10,000 screening colonoscopies performed, 8 people suffer a bowel perforation and 16 to 36 have severe bleeding requiring hospitalization. One in 3 patients undergoing colonoscopies report minor adverse events such as abdominal pain, bloating, and abdominal discomfort in the first 2 weeks following the procedure.
Other aspects of the ACP guidance differ from other colorectal cancer screening guidelines. Unlike the USPSTF, which made no distinctions between various recommended screening tests, the ACP preferentially endorsed fecal immunochemical or high-sensitivity fecal occult blood testing every 2 years, colonoscopy every 10 years, or flexible sigmoidoscopy every 10 years plus a fecal immunochemical test every 2 years. That leaves out stool DNA testing, which my patients increasingly request because they have seen television or online advertisements, and newer blood tests that detect methylation sequences in circulating tumor DNA.
Perhaps most controversial is the ACP’s suggestion that it is probably reasonable for some adults to start screening later than age 50 or undergo screening at longer intervals than currently recommended (for example, colonoscopy every 15 years). Recent data support extending the interval to repeat screening colonoscopy in selected populations; a large cross-sectional study found a low prevalence of advanced adenomas and colorectal cancers in colonoscopies performed 10 or more years after an initial negative colonoscopy, particularly in women and younger patients without gastrointestinal symptoms. A prominent BMJ guideline suggests that patients need not be screened until their estimated 15-year CRC risk is greater than 3% (which most people do not reach until their 60s) and then only need a single sigmoidoscopy or colonoscopy.
Despite some departures from other guidelines, it’s worth emphasizing that the ACP guideline agrees that screening for CRC is generally worthwhile between the ages of 50 and 75 years. On that front, we in primary care have more work to do; the Centers for Disease Control and Prevention estimates that 28% of American adults older than 50 are not up-to-date on CRC screening. And despite some recent debate about the relative benefits and harms of screening colonoscopy, compared with less invasive fecal tests, gastroenterologists and family physicians can agree that the best screening test is the test that gets done.
A version of this article first appeared on Medscape.com.
Unveiling the potential of prediction models in obstetrics
In the dawn of artificial intelligence’s potential to inform clinical practice, the importance of understanding the intent and interpretation of prediction tools is vital. In medicine, informed decision-making promotes patient autonomy and can lead to improved patient satisfaction and engagement in their own care.
This shared understanding enables patients to make informed choices about their care, reducing anxiety and increasing confidence in medical decision-making.
In obstetric clinical practice, prediction tools have been created to assess risk of primary cesarean delivery in gestational diabetes,1 cesarean delivery in hypertensive disorders of pregnancy,2 and failed induction of labor in nulliparous patients with an unfavorable cervix.3 By assessing a patient’s risk profile, clinicians can identify high-risk individuals who may require closer monitoring, early interventions, or specialized care. This allows for more timely interventions to optimize maternal and fetal health outcomes.
Other prediction tools are created to better elucidate to patients their individual risk of an outcome that may be modifiable, aiding physician counseling on mitigating factors to improve overall results. A relevant example is the American Diabetes Association’s risk of type 2 diabetes calculator used for counseling patients on risk reduction. This model includes both preexisting (ethnicity, family history, age, sex assigned at birth) and modifiable risk factors (body mass index, hypertension, physical activity) to predict risk of type 2 diabetes and is widely used in clinical practice to encourage integration of lifestyle changes to decrease risk.4 This model highlights the utility of prediction tools in counseling, providing quantitative data to clinicians to discuss a patient’s individual risk and how to mitigate that risk.
While predictive models clearly have many advantages and potential to improve personalized medicine, concerns have been raised that their interpretation and application can sometimes have unintended consequences as the complexity of these models can lead to variation in understanding among clinicians that impact decision-making. Different clinicians may assign different levels of importance to the predicted risks, resulting in differences in treatment plans and interventions. This variability can lead to disparities in care and outcomes, as patients with similar risk profiles may receive different management approaches based on the interpreting clinician.
Providers may either overly rely on prediction models or completely disregard them, depending on their level of trust or skepticism. Overreliance on prediction models may lead to the neglect of important clinical information or intuition, while disregarding the models may result in missed opportunities for early intervention or appropriate risk stratification. Achieving a balance between clinical judgment and the use of prediction models is crucial for optimal decision-making.
An example of how misinterpretation of the role of prediction tools in patient counseling can have far reaching consequences is the vaginal birth after cesarean (VBAC) calculator where race and ethnicity naturalized racial differences and likely contributed to cesarean overuse in Black pregnant people as non-White race was associated with a decreased chance of successful VBAC. Although the authors of the study that created the VBAC calculator intended it to be used as an adjunct to counseling, institutions and providers used low calculator scores to discourage or prohibit pregnant people from attempting a trial of labor after cesarean (TOLAC). This highlighted the importance of contextualizing the intent of prediction models within the broader clinical setting and individual patient circumstances and preferences.
This gap between intent and interpretation and subsequent application is influenced by individual clinician experience, training, personal biases, and subjective judgment. These subjective elements can introduce inconsistencies and variability in the utilization of prediction tools, leading to potential discrepancies in patient care. Inadequate understanding of prediction models and their statistical concepts can contribute to misinterpretation. It is this bias that prevents prediction models from serving their true purpose: to inform clinical decision-making, improve patient outcomes, and optimize resource allocation.
Clinicians may struggle with concepts such as predictive accuracy, overfitting, calibration, and external validation. Educational initiatives and enhanced training in statistical literacy can empower clinicians to better comprehend and apply prediction models in their practice. Researchers should make it clear that models should not be used in isolation, but rather integrated with clinical expertise and patient preferences. Understanding the limitations of prediction models and incorporating additional clinical information is essential.
Prediction models in obstetrics should undergo continuous evaluation and improvement to enhance their reliability and applicability. Regular updates, external validation, and recalibration are necessary to account for evolving clinical practices, changes in patient populations, and emerging evidence. Engaging clinicians in the evaluation process can foster ownership and promote a sense of trust in the models.
As machine learning and artificial intelligence improve the accuracy of prediction models, there is potential to revolutionize obstetric care by enabling more accurate individualized risk assessment and decision-making. Machine learning has the potential to significantly enhance prediction models in obstetrics by leveraging complex algorithms and advanced computational techniques. However, the unpredictable nature of clinician interpretation poses challenges to the effective utilization of these models.
By emphasizing communication, collaboration, education, and continuous evaluation, we can bridge the gap between prediction models and clinician interpretation that optimizes their use. This concerted effort will ultimately lead to improved patient care, enhanced clinical outcomes, and a more harmonious integration of these tools into obstetric practice.
Dr. Ramos is assistant professor of maternal fetal medicine and associate principal investigator at the Mother Infant Research Institute, Tufts University and Tufts Medical Center, Boston.
References
1. Ramos SZ et al. Predicting primary cesarean delivery in pregnancies complicated by gestational diabetes mellitus. Am J Obstet Gynecol. 2023 Jun 7;S0002-9378(23)00371-X. doi: 10.1016/j.ajog.2023.06.002.
2. Beninati MJ et al. Prediction model for vaginal birth after induction of labor in women with hypertensive disorders of pregnancy. Obstet Gynecol. 2020 Aug;136(2):402-410. doi: 10.1097/AOG.0000000000003938.
3. Levine LD et al. A validated calculator to estimate risk of cesarean after an induction of labor with an unfavorable cervix. Am J Obstet Gynecol. 2018 Feb;218(2):254.e1-254.e7. doi: 10.1016/j.ajog.2017.11.603.
4. American Diabetes Association. Our 60-Second Type 2 Diabetes Risk Test.
In the dawn of artificial intelligence’s potential to inform clinical practice, the importance of understanding the intent and interpretation of prediction tools is vital. In medicine, informed decision-making promotes patient autonomy and can lead to improved patient satisfaction and engagement in their own care.
This shared understanding enables patients to make informed choices about their care, reducing anxiety and increasing confidence in medical decision-making.
In obstetric clinical practice, prediction tools have been created to assess risk of primary cesarean delivery in gestational diabetes,1 cesarean delivery in hypertensive disorders of pregnancy,2 and failed induction of labor in nulliparous patients with an unfavorable cervix.3 By assessing a patient’s risk profile, clinicians can identify high-risk individuals who may require closer monitoring, early interventions, or specialized care. This allows for more timely interventions to optimize maternal and fetal health outcomes.
Other prediction tools are created to better elucidate to patients their individual risk of an outcome that may be modifiable, aiding physician counseling on mitigating factors to improve overall results. A relevant example is the American Diabetes Association’s risk of type 2 diabetes calculator used for counseling patients on risk reduction. This model includes both preexisting (ethnicity, family history, age, sex assigned at birth) and modifiable risk factors (body mass index, hypertension, physical activity) to predict risk of type 2 diabetes and is widely used in clinical practice to encourage integration of lifestyle changes to decrease risk.4 This model highlights the utility of prediction tools in counseling, providing quantitative data to clinicians to discuss a patient’s individual risk and how to mitigate that risk.
While predictive models clearly have many advantages and potential to improve personalized medicine, concerns have been raised that their interpretation and application can sometimes have unintended consequences as the complexity of these models can lead to variation in understanding among clinicians that impact decision-making. Different clinicians may assign different levels of importance to the predicted risks, resulting in differences in treatment plans and interventions. This variability can lead to disparities in care and outcomes, as patients with similar risk profiles may receive different management approaches based on the interpreting clinician.
Providers may either overly rely on prediction models or completely disregard them, depending on their level of trust or skepticism. Overreliance on prediction models may lead to the neglect of important clinical information or intuition, while disregarding the models may result in missed opportunities for early intervention or appropriate risk stratification. Achieving a balance between clinical judgment and the use of prediction models is crucial for optimal decision-making.
An example of how misinterpretation of the role of prediction tools in patient counseling can have far reaching consequences is the vaginal birth after cesarean (VBAC) calculator where race and ethnicity naturalized racial differences and likely contributed to cesarean overuse in Black pregnant people as non-White race was associated with a decreased chance of successful VBAC. Although the authors of the study that created the VBAC calculator intended it to be used as an adjunct to counseling, institutions and providers used low calculator scores to discourage or prohibit pregnant people from attempting a trial of labor after cesarean (TOLAC). This highlighted the importance of contextualizing the intent of prediction models within the broader clinical setting and individual patient circumstances and preferences.
This gap between intent and interpretation and subsequent application is influenced by individual clinician experience, training, personal biases, and subjective judgment. These subjective elements can introduce inconsistencies and variability in the utilization of prediction tools, leading to potential discrepancies in patient care. Inadequate understanding of prediction models and their statistical concepts can contribute to misinterpretation. It is this bias that prevents prediction models from serving their true purpose: to inform clinical decision-making, improve patient outcomes, and optimize resource allocation.
Clinicians may struggle with concepts such as predictive accuracy, overfitting, calibration, and external validation. Educational initiatives and enhanced training in statistical literacy can empower clinicians to better comprehend and apply prediction models in their practice. Researchers should make it clear that models should not be used in isolation, but rather integrated with clinical expertise and patient preferences. Understanding the limitations of prediction models and incorporating additional clinical information is essential.
Prediction models in obstetrics should undergo continuous evaluation and improvement to enhance their reliability and applicability. Regular updates, external validation, and recalibration are necessary to account for evolving clinical practices, changes in patient populations, and emerging evidence. Engaging clinicians in the evaluation process can foster ownership and promote a sense of trust in the models.
As machine learning and artificial intelligence improve the accuracy of prediction models, there is potential to revolutionize obstetric care by enabling more accurate individualized risk assessment and decision-making. Machine learning has the potential to significantly enhance prediction models in obstetrics by leveraging complex algorithms and advanced computational techniques. However, the unpredictable nature of clinician interpretation poses challenges to the effective utilization of these models.
By emphasizing communication, collaboration, education, and continuous evaluation, we can bridge the gap between prediction models and clinician interpretation that optimizes their use. This concerted effort will ultimately lead to improved patient care, enhanced clinical outcomes, and a more harmonious integration of these tools into obstetric practice.
Dr. Ramos is assistant professor of maternal fetal medicine and associate principal investigator at the Mother Infant Research Institute, Tufts University and Tufts Medical Center, Boston.
References
1. Ramos SZ et al. Predicting primary cesarean delivery in pregnancies complicated by gestational diabetes mellitus. Am J Obstet Gynecol. 2023 Jun 7;S0002-9378(23)00371-X. doi: 10.1016/j.ajog.2023.06.002.
2. Beninati MJ et al. Prediction model for vaginal birth after induction of labor in women with hypertensive disorders of pregnancy. Obstet Gynecol. 2020 Aug;136(2):402-410. doi: 10.1097/AOG.0000000000003938.
3. Levine LD et al. A validated calculator to estimate risk of cesarean after an induction of labor with an unfavorable cervix. Am J Obstet Gynecol. 2018 Feb;218(2):254.e1-254.e7. doi: 10.1016/j.ajog.2017.11.603.
4. American Diabetes Association. Our 60-Second Type 2 Diabetes Risk Test.
In the dawn of artificial intelligence’s potential to inform clinical practice, the importance of understanding the intent and interpretation of prediction tools is vital. In medicine, informed decision-making promotes patient autonomy and can lead to improved patient satisfaction and engagement in their own care.
This shared understanding enables patients to make informed choices about their care, reducing anxiety and increasing confidence in medical decision-making.
In obstetric clinical practice, prediction tools have been created to assess risk of primary cesarean delivery in gestational diabetes,1 cesarean delivery in hypertensive disorders of pregnancy,2 and failed induction of labor in nulliparous patients with an unfavorable cervix.3 By assessing a patient’s risk profile, clinicians can identify high-risk individuals who may require closer monitoring, early interventions, or specialized care. This allows for more timely interventions to optimize maternal and fetal health outcomes.
Other prediction tools are created to better elucidate to patients their individual risk of an outcome that may be modifiable, aiding physician counseling on mitigating factors to improve overall results. A relevant example is the American Diabetes Association’s risk of type 2 diabetes calculator used for counseling patients on risk reduction. This model includes both preexisting (ethnicity, family history, age, sex assigned at birth) and modifiable risk factors (body mass index, hypertension, physical activity) to predict risk of type 2 diabetes and is widely used in clinical practice to encourage integration of lifestyle changes to decrease risk.4 This model highlights the utility of prediction tools in counseling, providing quantitative data to clinicians to discuss a patient’s individual risk and how to mitigate that risk.
While predictive models clearly have many advantages and potential to improve personalized medicine, concerns have been raised that their interpretation and application can sometimes have unintended consequences as the complexity of these models can lead to variation in understanding among clinicians that impact decision-making. Different clinicians may assign different levels of importance to the predicted risks, resulting in differences in treatment plans and interventions. This variability can lead to disparities in care and outcomes, as patients with similar risk profiles may receive different management approaches based on the interpreting clinician.
Providers may either overly rely on prediction models or completely disregard them, depending on their level of trust or skepticism. Overreliance on prediction models may lead to the neglect of important clinical information or intuition, while disregarding the models may result in missed opportunities for early intervention or appropriate risk stratification. Achieving a balance between clinical judgment and the use of prediction models is crucial for optimal decision-making.
An example of how misinterpretation of the role of prediction tools in patient counseling can have far reaching consequences is the vaginal birth after cesarean (VBAC) calculator where race and ethnicity naturalized racial differences and likely contributed to cesarean overuse in Black pregnant people as non-White race was associated with a decreased chance of successful VBAC. Although the authors of the study that created the VBAC calculator intended it to be used as an adjunct to counseling, institutions and providers used low calculator scores to discourage or prohibit pregnant people from attempting a trial of labor after cesarean (TOLAC). This highlighted the importance of contextualizing the intent of prediction models within the broader clinical setting and individual patient circumstances and preferences.
This gap between intent and interpretation and subsequent application is influenced by individual clinician experience, training, personal biases, and subjective judgment. These subjective elements can introduce inconsistencies and variability in the utilization of prediction tools, leading to potential discrepancies in patient care. Inadequate understanding of prediction models and their statistical concepts can contribute to misinterpretation. It is this bias that prevents prediction models from serving their true purpose: to inform clinical decision-making, improve patient outcomes, and optimize resource allocation.
Clinicians may struggle with concepts such as predictive accuracy, overfitting, calibration, and external validation. Educational initiatives and enhanced training in statistical literacy can empower clinicians to better comprehend and apply prediction models in their practice. Researchers should make it clear that models should not be used in isolation, but rather integrated with clinical expertise and patient preferences. Understanding the limitations of prediction models and incorporating additional clinical information is essential.
Prediction models in obstetrics should undergo continuous evaluation and improvement to enhance their reliability and applicability. Regular updates, external validation, and recalibration are necessary to account for evolving clinical practices, changes in patient populations, and emerging evidence. Engaging clinicians in the evaluation process can foster ownership and promote a sense of trust in the models.
As machine learning and artificial intelligence improve the accuracy of prediction models, there is potential to revolutionize obstetric care by enabling more accurate individualized risk assessment and decision-making. Machine learning has the potential to significantly enhance prediction models in obstetrics by leveraging complex algorithms and advanced computational techniques. However, the unpredictable nature of clinician interpretation poses challenges to the effective utilization of these models.
By emphasizing communication, collaboration, education, and continuous evaluation, we can bridge the gap between prediction models and clinician interpretation that optimizes their use. This concerted effort will ultimately lead to improved patient care, enhanced clinical outcomes, and a more harmonious integration of these tools into obstetric practice.
Dr. Ramos is assistant professor of maternal fetal medicine and associate principal investigator at the Mother Infant Research Institute, Tufts University and Tufts Medical Center, Boston.
References
1. Ramos SZ et al. Predicting primary cesarean delivery in pregnancies complicated by gestational diabetes mellitus. Am J Obstet Gynecol. 2023 Jun 7;S0002-9378(23)00371-X. doi: 10.1016/j.ajog.2023.06.002.
2. Beninati MJ et al. Prediction model for vaginal birth after induction of labor in women with hypertensive disorders of pregnancy. Obstet Gynecol. 2020 Aug;136(2):402-410. doi: 10.1097/AOG.0000000000003938.
3. Levine LD et al. A validated calculator to estimate risk of cesarean after an induction of labor with an unfavorable cervix. Am J Obstet Gynecol. 2018 Feb;218(2):254.e1-254.e7. doi: 10.1016/j.ajog.2017.11.603.
4. American Diabetes Association. Our 60-Second Type 2 Diabetes Risk Test.
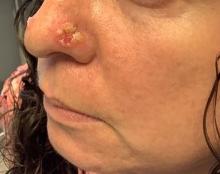
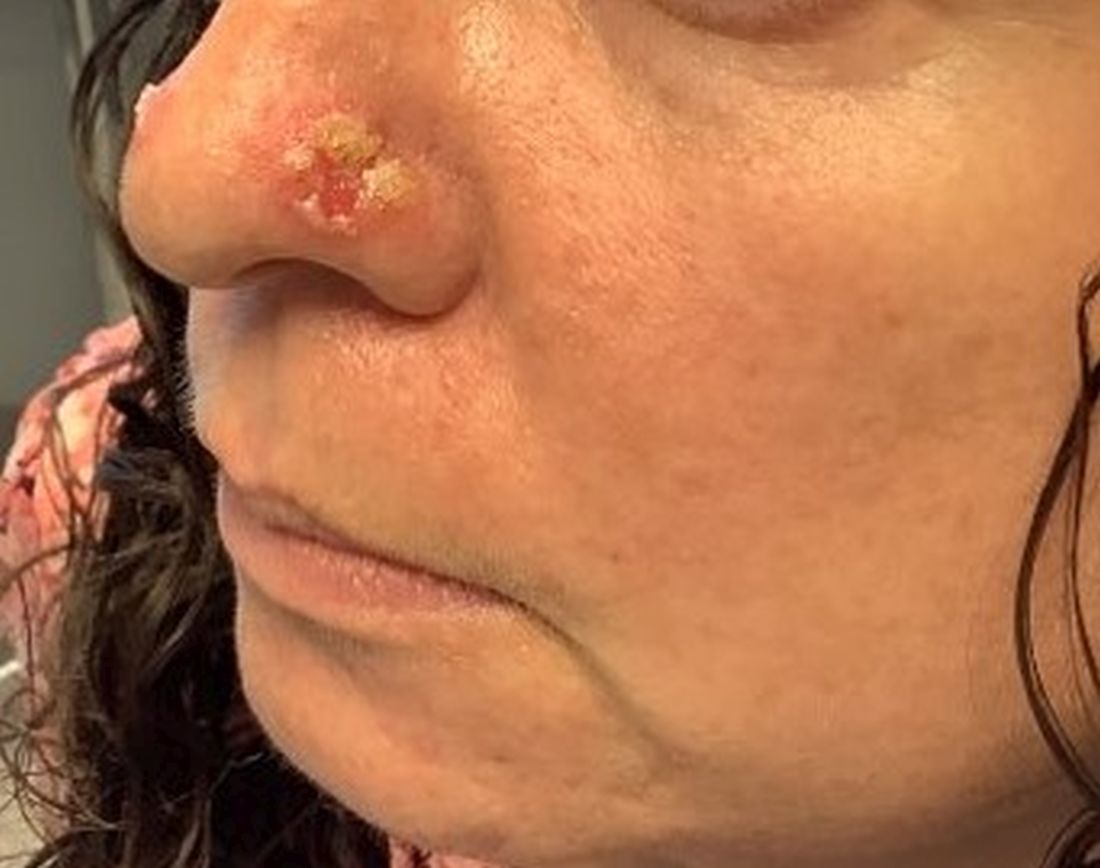
A 45-year-old White woman with no significant medical history presented with a 1-month history of lesions on the nose and right cheek
Cultures for bacteria, varicella zoster virus, herpes simplex virus, and mpox virus were all negative. A biopsy revealed suprabasilar acantholysis with follicular involvement in association with blister formation and inflammation. Direct immunofluorescence was positive for suprabasilar IgG and C3 deposition, consistent with pemphigus vulgaris (PV).
. There is likely a genetic predisposition. Medications that may induce pemphigus include penicillamine, nifedipine, or captopril.
Clinically, flaccid blistering lesions are present that may be cutaneous and/or mucosal. Bullae can progress to erosions and crusting, which then heal with pigment alteration but not scarring. The most commonly affected sites are the mouth, intertriginous areas, face, and neck. Mucosal lesions may involve the lips, esophagus, conjunctiva, and genitals.
Biopsy for histology and direct immunofluorescence is important in distinguishing between PV and other blistering disorders. Up to 75% of patients with active disease also have a positive indirect immunofluorescence with circulating IgG.
Treatment is generally immunosuppressive. Systemic therapy usually begins with prednisone and then is transitioned to a steroid-sparing agent such as mycophenolate mofetil. Other steroid-sparing agents include azathioprine, methotrexate, cyclophosphamide, and intravenous immunoglobulin. Secondary infections are possible and should be treated. Topical therapies aimed at reducing pain, especially in mucosal lesions, can be beneficial.
This case and the photos are from Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
Cultures for bacteria, varicella zoster virus, herpes simplex virus, and mpox virus were all negative. A biopsy revealed suprabasilar acantholysis with follicular involvement in association with blister formation and inflammation. Direct immunofluorescence was positive for suprabasilar IgG and C3 deposition, consistent with pemphigus vulgaris (PV).
. There is likely a genetic predisposition. Medications that may induce pemphigus include penicillamine, nifedipine, or captopril.
Clinically, flaccid blistering lesions are present that may be cutaneous and/or mucosal. Bullae can progress to erosions and crusting, which then heal with pigment alteration but not scarring. The most commonly affected sites are the mouth, intertriginous areas, face, and neck. Mucosal lesions may involve the lips, esophagus, conjunctiva, and genitals.
Biopsy for histology and direct immunofluorescence is important in distinguishing between PV and other blistering disorders. Up to 75% of patients with active disease also have a positive indirect immunofluorescence with circulating IgG.
Treatment is generally immunosuppressive. Systemic therapy usually begins with prednisone and then is transitioned to a steroid-sparing agent such as mycophenolate mofetil. Other steroid-sparing agents include azathioprine, methotrexate, cyclophosphamide, and intravenous immunoglobulin. Secondary infections are possible and should be treated. Topical therapies aimed at reducing pain, especially in mucosal lesions, can be beneficial.
This case and the photos are from Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
Cultures for bacteria, varicella zoster virus, herpes simplex virus, and mpox virus were all negative. A biopsy revealed suprabasilar acantholysis with follicular involvement in association with blister formation and inflammation. Direct immunofluorescence was positive for suprabasilar IgG and C3 deposition, consistent with pemphigus vulgaris (PV).
. There is likely a genetic predisposition. Medications that may induce pemphigus include penicillamine, nifedipine, or captopril.
Clinically, flaccid blistering lesions are present that may be cutaneous and/or mucosal. Bullae can progress to erosions and crusting, which then heal with pigment alteration but not scarring. The most commonly affected sites are the mouth, intertriginous areas, face, and neck. Mucosal lesions may involve the lips, esophagus, conjunctiva, and genitals.
Biopsy for histology and direct immunofluorescence is important in distinguishing between PV and other blistering disorders. Up to 75% of patients with active disease also have a positive indirect immunofluorescence with circulating IgG.
Treatment is generally immunosuppressive. Systemic therapy usually begins with prednisone and then is transitioned to a steroid-sparing agent such as mycophenolate mofetil. Other steroid-sparing agents include azathioprine, methotrexate, cyclophosphamide, and intravenous immunoglobulin. Secondary infections are possible and should be treated. Topical therapies aimed at reducing pain, especially in mucosal lesions, can be beneficial.
This case and the photos are from Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
For NSCLC, neoadjuvant, adjuvant, or both?
This transcript has been edited for clarity.
Dr. West: Here at ASCO 2023 [American Society of Clinical Oncology] in Chicago, we’ve seen some blockbuster presentations in thoracic oncology. Many of these have brought up some important questions about the clinical implications that we need to discuss further.
At ASCO, as well as in the couple or 3 months preceding ASCO, we’ve gotten more and more data on perioperative approaches. Of course, over the past couple of years, we’ve had some new options of postoperative immunotherapy for a year, say, after chemotherapy or possibly after chemotherapy.
We have also had very influential data, such as the CheckMate 816 trial that gave three cycles of chemotherapy with nivolumab vs. chemotherapy alone to patients with stage IB to IIIA disease, but largely, nearly two thirds, with IIIA disease. That showed a very clear improvement in the pathologic complete response (pCR) rate with nivolumab added to chemotherapy and also a highly significant improvement in event-free survival and a strong trend toward improved overall survival. This is FDA approved and has been increasingly adopted, I would say, maybe with some variability by geography and center, but really a good amount of enthusiasm.
Now, we have a bunch of trials that give chemotherapy with immunotherapy. We’ve got the AEGEAN trial with durvalumab. We have Neotorch with chemotherapy and toripalimab. At ASCO 2023, we had a highly prominent presentation of KEYNOTE-671, giving four cycles of chemotherapy with pembrolizumab vs. chemotherapy and placebo.
Then there’s the built-in postoperative component of a year of immunotherapy as well, in all these trials. The data for KEYNOTE-671 look quite good. Of course, the other trials also were significant. I would say the comparator now is not nothing or chemotherapy alone anymore; it’s really against what is the best current standard of care.
It certainly adds some cost, it adds some risk for toxicity, and it adds a year of a patient coming into the clinic and getting IV infusions all this time to get a treatment that the patient has already had for four cycles in most of these trials.
If your cancer is resistant, is there going to be an incremental benefit to giving more of it? What are your thoughts about the risk and benefit? Going to a patient in your own clinic, how are you going to counsel your patients? Will anything change after the presentation of all these data and how you approach preoperatively?
Dr. Rotow: I agree. In some sense, it’s an embarrassment of riches, right?
Dr. West: Yes.
Dr. Rotow: We have so many positive studies looking at perioperative immunotherapy for our patients. They all show improved outcomes, but of course, they all compare with the old control arm of chemotherapy alone in some form, and this is no longer a useful control in this space. The open question is, do you use neoadjuvant, do you use adjuvant, or do you use both?
My high-level takeaway from these data is that the neoadjuvant component appears to be important. I think the overall trend, comparing across studies, of course, is that outcomes seem to be better with the neoadjuvant component. You also get the advantage of potential downstaging and potential greater ease of surgical resection. We know they have lower morbidity resection and shorter surgeries. You can comment on that. You also get your pathologic response data.
Dr. West: You get the feedback.
Dr. Rotow: Exactly.
Dr. West: The deliverability is also a big issue. You know you can much more reliably deliver your intended treatment by doing neoadjuvant followed by surgery.
Dr. Rotow: Exactly.
Dr. West: We know there’s major drop-off if patients have surgery, and in the recovery room they hear you got it all, and then they need to come back and maybe get chemotherapy and immunotherapy for a year. They’d ask, “What for? I can’t see anything.”
Dr. Rotow: Exactly. I think there are many advantages to that neoadjuvant component. I think all or many of us now have integrated this into our routine practice. Now the question is, do you need the adjuvant element or not on top? That is challenging because no trial has compared adjuvant to nonadjuvant. I think we all advocate for the need for this trial to answer this in a more randomized, prospective fashion. Of course, that doesn’t help our clinic practice tomorrow when we see a patient.
Dr. West: Or for the next 4 years.
Dr. Rotow: Or for the next 4 years – exactly. There’s going to be the open question of who really needs this? In some sense, we may be guided by the path response during the surgery itself. I think there may be those who claim that if you have a pCR, do you really need additional therapy? We don’t know the answer, but it’s tempting to say we know the outcomes in event-free survival are extremely good with a pCR.
Dr. West: Which is only 20% or 25% of patients, so it’s not most.
Dr. Rotow: It’s not most, but it’s better than the 2% or so with chemotherapy alone. That’s real progress, and it’s nice to have that readout. For that 80% without a pCR, what to do? I suspect there will be variation from provider to provider and from patient to patient, depending on tolerability to prior therapy, the patient’s wishes around the goals of care, and the patient’s risk for autoimmune toxicities.
Maybe there’s a patient with underlying autoimmune disease who’s gotten their neoadjuvant therapy and done well. You don’t want to risk that ongoing risk of exposure. Perhaps a patient with no risk factors who desires very aggressive treatment might be interested in more treatment.
In KEYNOTE-671, I was interested in the PD-L1 subgroups. These did trend the way you expect, with better responses in PD-L1 high, but there were also good outcomes and benefit to immunotherapy with the perioperative strategy in PD-L1–negative patients.
Dr. West: That didn’t really exclude anybody.
Dr. Rotow: It didn’t exclude anybody. In CheckMate 816, everyone benefited, but the benefit was less with those PD-L1–negative patients.
Dr. West: True.
Dr. Rotow: Absent further data to guide me or any prospective data here comparing these strategies, I might lean toward a longer course of immunotherapy in that population in hopes of triggering a response. I suspect that there will be variation from clinician to clinician in that space.
Dr. West: This is a setting where I feel like I have equipoise. I really feel that the incremental benefit is pretty small.
Dr. Rotow: Small. I agree.
Dr. West: It’s, frankly, somewhat dubious. On the other hand, you’re in a situation where if you know that three of four patients will experience a relapse and less-than-amazing outcomes, it’s hard to leave something that’s FDA approved and studied and a well-sanctioned option on the table if this patient may have relapse later.
In the end, I feel like I’d like to offer this and discuss it with all my patients. I think it’s a great place for shared decision-making because if a patient hears about that and decides they’re not interested, I’ll be fine with that. I think that’s a very sensible approach, but I don’t want to make it unilaterally. Other patients may say they want every opportunity, and if it comes back, at least I’ll know I did everything we could.
Dr. Rotow: Exactly. I agree with your statement about equipoise. I truly think that this is present here in the situation, and that there’s room for discussion in both directions with patients.
Now, one caveat I’d like to add to all these data is that the data should not apply to patients with some of our classic nonsmoking-associated driver mutations. This is another piece to the neoadjuvant data that I think is worth commenting on – the need to get appropriate testing before initiation of therapy and the pitfalls of starting this kind of treatment without knowing full biomarker testing. I think that’s something we have to watch for in our clinical practice as well.
Dr. West: Perhaps especially if we’re talking about doing a year of postoperative and someone has an ALK rearrangement or an EGFR mutation and we didn’t know it. That is a group where we’re worried about a rapid transition and potentially prohibitive, even life-threatening, toxicities from not planning in advance for this. This is something you don’t want to give concurrently or one right on top of the other. You don’t want to give immunotherapy and then transition right to targeted therapy. It’s dangerous.
Dr. Rotow: Exactly. The stakes were already high with neoadjuvant alone, but at least you had that gap of the presurgical period, surgical recovery, and then initiation of adjuvant therapy, if needed, or at relapse. With a postoperative long adjuvant period, those stakes are elevated because the immunotherapy exposure continues, so it’s something to be mindful of.
Dr. West: We have a general sense that many, but not all, of the targets that we’re talking about are associated with low benefit from immunotherapy. It’s not that well studied. I think this is another place for individualized discussion of the pros and cons. They were included in the trial, but they probably benefit less.
Dr. Rotow: Exactly. I think with the best established, EGFR and ALK probably are not benefiting much. They were actually included in the trial. Many of the neoadjuvant studies do not allow them to enroll if they’re known. On the other end of that spectrum, I think KRAS is just fine to treat with immunotherapy.
Dr. West: Sure.
Dr. Rotow: It’s an actionable driver. It’s not a traditional nonsmoking-associated driver, and those do just fine.
Dr. West: The studies show that these patients benefit just as much, at least, as the other patients.
Dr. Rotow: Exactly. I would never withhold this form of therapy for a KRAS driver mutation. The others, I think, are still in a gray zone. Depending on the patient demographics and tobacco use, I may elicit more or less caution in that space.
Dr. West: Well, I think we’re going to have much to still tease apart, with room for judgment here without a strong sense of the data telling us exactly what to do.
Dr. Rotow: Exactly.
Dr. West: There’s a large amount of excitement and interest in these new data, but there are still many open questions. I hope we continue to mull it over as we get more data and more insight to shape our plans.
Dr. West is an associate professor at City of Hope Comprehensive Cancer Center in Duarte, Calif., and vice president of network strategy at AccessHope in Los Angeles. Dr. Rotow is the clinical director of the Lowe Center for Thoracic Oncology at the Dana-Farber Cancer Institute in Boston. Dr. West reported conflicts of interest with Ariad/Takeda, Bristol Myers Squibb, Boehringer Ingelheim, Spectrum, AstraZeneca, Celgene, Genentech/Roche, Pfizer, Merck, and Eli Lilly. Dr. Rotow reported conflicts of interest with Genentech, AstraZeneca,Guardant, and Janssen.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Dr. West: Here at ASCO 2023 [American Society of Clinical Oncology] in Chicago, we’ve seen some blockbuster presentations in thoracic oncology. Many of these have brought up some important questions about the clinical implications that we need to discuss further.
At ASCO, as well as in the couple or 3 months preceding ASCO, we’ve gotten more and more data on perioperative approaches. Of course, over the past couple of years, we’ve had some new options of postoperative immunotherapy for a year, say, after chemotherapy or possibly after chemotherapy.
We have also had very influential data, such as the CheckMate 816 trial that gave three cycles of chemotherapy with nivolumab vs. chemotherapy alone to patients with stage IB to IIIA disease, but largely, nearly two thirds, with IIIA disease. That showed a very clear improvement in the pathologic complete response (pCR) rate with nivolumab added to chemotherapy and also a highly significant improvement in event-free survival and a strong trend toward improved overall survival. This is FDA approved and has been increasingly adopted, I would say, maybe with some variability by geography and center, but really a good amount of enthusiasm.
Now, we have a bunch of trials that give chemotherapy with immunotherapy. We’ve got the AEGEAN trial with durvalumab. We have Neotorch with chemotherapy and toripalimab. At ASCO 2023, we had a highly prominent presentation of KEYNOTE-671, giving four cycles of chemotherapy with pembrolizumab vs. chemotherapy and placebo.
Then there’s the built-in postoperative component of a year of immunotherapy as well, in all these trials. The data for KEYNOTE-671 look quite good. Of course, the other trials also were significant. I would say the comparator now is not nothing or chemotherapy alone anymore; it’s really against what is the best current standard of care.
It certainly adds some cost, it adds some risk for toxicity, and it adds a year of a patient coming into the clinic and getting IV infusions all this time to get a treatment that the patient has already had for four cycles in most of these trials.
If your cancer is resistant, is there going to be an incremental benefit to giving more of it? What are your thoughts about the risk and benefit? Going to a patient in your own clinic, how are you going to counsel your patients? Will anything change after the presentation of all these data and how you approach preoperatively?
Dr. Rotow: I agree. In some sense, it’s an embarrassment of riches, right?
Dr. West: Yes.
Dr. Rotow: We have so many positive studies looking at perioperative immunotherapy for our patients. They all show improved outcomes, but of course, they all compare with the old control arm of chemotherapy alone in some form, and this is no longer a useful control in this space. The open question is, do you use neoadjuvant, do you use adjuvant, or do you use both?
My high-level takeaway from these data is that the neoadjuvant component appears to be important. I think the overall trend, comparing across studies, of course, is that outcomes seem to be better with the neoadjuvant component. You also get the advantage of potential downstaging and potential greater ease of surgical resection. We know they have lower morbidity resection and shorter surgeries. You can comment on that. You also get your pathologic response data.
Dr. West: You get the feedback.
Dr. Rotow: Exactly.
Dr. West: The deliverability is also a big issue. You know you can much more reliably deliver your intended treatment by doing neoadjuvant followed by surgery.
Dr. Rotow: Exactly.
Dr. West: We know there’s major drop-off if patients have surgery, and in the recovery room they hear you got it all, and then they need to come back and maybe get chemotherapy and immunotherapy for a year. They’d ask, “What for? I can’t see anything.”
Dr. Rotow: Exactly. I think there are many advantages to that neoadjuvant component. I think all or many of us now have integrated this into our routine practice. Now the question is, do you need the adjuvant element or not on top? That is challenging because no trial has compared adjuvant to nonadjuvant. I think we all advocate for the need for this trial to answer this in a more randomized, prospective fashion. Of course, that doesn’t help our clinic practice tomorrow when we see a patient.
Dr. West: Or for the next 4 years.
Dr. Rotow: Or for the next 4 years – exactly. There’s going to be the open question of who really needs this? In some sense, we may be guided by the path response during the surgery itself. I think there may be those who claim that if you have a pCR, do you really need additional therapy? We don’t know the answer, but it’s tempting to say we know the outcomes in event-free survival are extremely good with a pCR.
Dr. West: Which is only 20% or 25% of patients, so it’s not most.
Dr. Rotow: It’s not most, but it’s better than the 2% or so with chemotherapy alone. That’s real progress, and it’s nice to have that readout. For that 80% without a pCR, what to do? I suspect there will be variation from provider to provider and from patient to patient, depending on tolerability to prior therapy, the patient’s wishes around the goals of care, and the patient’s risk for autoimmune toxicities.
Maybe there’s a patient with underlying autoimmune disease who’s gotten their neoadjuvant therapy and done well. You don’t want to risk that ongoing risk of exposure. Perhaps a patient with no risk factors who desires very aggressive treatment might be interested in more treatment.
In KEYNOTE-671, I was interested in the PD-L1 subgroups. These did trend the way you expect, with better responses in PD-L1 high, but there were also good outcomes and benefit to immunotherapy with the perioperative strategy in PD-L1–negative patients.
Dr. West: That didn’t really exclude anybody.
Dr. Rotow: It didn’t exclude anybody. In CheckMate 816, everyone benefited, but the benefit was less with those PD-L1–negative patients.
Dr. West: True.
Dr. Rotow: Absent further data to guide me or any prospective data here comparing these strategies, I might lean toward a longer course of immunotherapy in that population in hopes of triggering a response. I suspect that there will be variation from clinician to clinician in that space.
Dr. West: This is a setting where I feel like I have equipoise. I really feel that the incremental benefit is pretty small.
Dr. Rotow: Small. I agree.
Dr. West: It’s, frankly, somewhat dubious. On the other hand, you’re in a situation where if you know that three of four patients will experience a relapse and less-than-amazing outcomes, it’s hard to leave something that’s FDA approved and studied and a well-sanctioned option on the table if this patient may have relapse later.
In the end, I feel like I’d like to offer this and discuss it with all my patients. I think it’s a great place for shared decision-making because if a patient hears about that and decides they’re not interested, I’ll be fine with that. I think that’s a very sensible approach, but I don’t want to make it unilaterally. Other patients may say they want every opportunity, and if it comes back, at least I’ll know I did everything we could.
Dr. Rotow: Exactly. I agree with your statement about equipoise. I truly think that this is present here in the situation, and that there’s room for discussion in both directions with patients.
Now, one caveat I’d like to add to all these data is that the data should not apply to patients with some of our classic nonsmoking-associated driver mutations. This is another piece to the neoadjuvant data that I think is worth commenting on – the need to get appropriate testing before initiation of therapy and the pitfalls of starting this kind of treatment without knowing full biomarker testing. I think that’s something we have to watch for in our clinical practice as well.
Dr. West: Perhaps especially if we’re talking about doing a year of postoperative and someone has an ALK rearrangement or an EGFR mutation and we didn’t know it. That is a group where we’re worried about a rapid transition and potentially prohibitive, even life-threatening, toxicities from not planning in advance for this. This is something you don’t want to give concurrently or one right on top of the other. You don’t want to give immunotherapy and then transition right to targeted therapy. It’s dangerous.
Dr. Rotow: Exactly. The stakes were already high with neoadjuvant alone, but at least you had that gap of the presurgical period, surgical recovery, and then initiation of adjuvant therapy, if needed, or at relapse. With a postoperative long adjuvant period, those stakes are elevated because the immunotherapy exposure continues, so it’s something to be mindful of.
Dr. West: We have a general sense that many, but not all, of the targets that we’re talking about are associated with low benefit from immunotherapy. It’s not that well studied. I think this is another place for individualized discussion of the pros and cons. They were included in the trial, but they probably benefit less.
Dr. Rotow: Exactly. I think with the best established, EGFR and ALK probably are not benefiting much. They were actually included in the trial. Many of the neoadjuvant studies do not allow them to enroll if they’re known. On the other end of that spectrum, I think KRAS is just fine to treat with immunotherapy.
Dr. West: Sure.
Dr. Rotow: It’s an actionable driver. It’s not a traditional nonsmoking-associated driver, and those do just fine.
Dr. West: The studies show that these patients benefit just as much, at least, as the other patients.
Dr. Rotow: Exactly. I would never withhold this form of therapy for a KRAS driver mutation. The others, I think, are still in a gray zone. Depending on the patient demographics and tobacco use, I may elicit more or less caution in that space.
Dr. West: Well, I think we’re going to have much to still tease apart, with room for judgment here without a strong sense of the data telling us exactly what to do.
Dr. Rotow: Exactly.
Dr. West: There’s a large amount of excitement and interest in these new data, but there are still many open questions. I hope we continue to mull it over as we get more data and more insight to shape our plans.
Dr. West is an associate professor at City of Hope Comprehensive Cancer Center in Duarte, Calif., and vice president of network strategy at AccessHope in Los Angeles. Dr. Rotow is the clinical director of the Lowe Center for Thoracic Oncology at the Dana-Farber Cancer Institute in Boston. Dr. West reported conflicts of interest with Ariad/Takeda, Bristol Myers Squibb, Boehringer Ingelheim, Spectrum, AstraZeneca, Celgene, Genentech/Roche, Pfizer, Merck, and Eli Lilly. Dr. Rotow reported conflicts of interest with Genentech, AstraZeneca,Guardant, and Janssen.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Dr. West: Here at ASCO 2023 [American Society of Clinical Oncology] in Chicago, we’ve seen some blockbuster presentations in thoracic oncology. Many of these have brought up some important questions about the clinical implications that we need to discuss further.
At ASCO, as well as in the couple or 3 months preceding ASCO, we’ve gotten more and more data on perioperative approaches. Of course, over the past couple of years, we’ve had some new options of postoperative immunotherapy for a year, say, after chemotherapy or possibly after chemotherapy.
We have also had very influential data, such as the CheckMate 816 trial that gave three cycles of chemotherapy with nivolumab vs. chemotherapy alone to patients with stage IB to IIIA disease, but largely, nearly two thirds, with IIIA disease. That showed a very clear improvement in the pathologic complete response (pCR) rate with nivolumab added to chemotherapy and also a highly significant improvement in event-free survival and a strong trend toward improved overall survival. This is FDA approved and has been increasingly adopted, I would say, maybe with some variability by geography and center, but really a good amount of enthusiasm.
Now, we have a bunch of trials that give chemotherapy with immunotherapy. We’ve got the AEGEAN trial with durvalumab. We have Neotorch with chemotherapy and toripalimab. At ASCO 2023, we had a highly prominent presentation of KEYNOTE-671, giving four cycles of chemotherapy with pembrolizumab vs. chemotherapy and placebo.
Then there’s the built-in postoperative component of a year of immunotherapy as well, in all these trials. The data for KEYNOTE-671 look quite good. Of course, the other trials also were significant. I would say the comparator now is not nothing or chemotherapy alone anymore; it’s really against what is the best current standard of care.
It certainly adds some cost, it adds some risk for toxicity, and it adds a year of a patient coming into the clinic and getting IV infusions all this time to get a treatment that the patient has already had for four cycles in most of these trials.
If your cancer is resistant, is there going to be an incremental benefit to giving more of it? What are your thoughts about the risk and benefit? Going to a patient in your own clinic, how are you going to counsel your patients? Will anything change after the presentation of all these data and how you approach preoperatively?
Dr. Rotow: I agree. In some sense, it’s an embarrassment of riches, right?
Dr. West: Yes.
Dr. Rotow: We have so many positive studies looking at perioperative immunotherapy for our patients. They all show improved outcomes, but of course, they all compare with the old control arm of chemotherapy alone in some form, and this is no longer a useful control in this space. The open question is, do you use neoadjuvant, do you use adjuvant, or do you use both?
My high-level takeaway from these data is that the neoadjuvant component appears to be important. I think the overall trend, comparing across studies, of course, is that outcomes seem to be better with the neoadjuvant component. You also get the advantage of potential downstaging and potential greater ease of surgical resection. We know they have lower morbidity resection and shorter surgeries. You can comment on that. You also get your pathologic response data.
Dr. West: You get the feedback.
Dr. Rotow: Exactly.
Dr. West: The deliverability is also a big issue. You know you can much more reliably deliver your intended treatment by doing neoadjuvant followed by surgery.
Dr. Rotow: Exactly.
Dr. West: We know there’s major drop-off if patients have surgery, and in the recovery room they hear you got it all, and then they need to come back and maybe get chemotherapy and immunotherapy for a year. They’d ask, “What for? I can’t see anything.”
Dr. Rotow: Exactly. I think there are many advantages to that neoadjuvant component. I think all or many of us now have integrated this into our routine practice. Now the question is, do you need the adjuvant element or not on top? That is challenging because no trial has compared adjuvant to nonadjuvant. I think we all advocate for the need for this trial to answer this in a more randomized, prospective fashion. Of course, that doesn’t help our clinic practice tomorrow when we see a patient.
Dr. West: Or for the next 4 years.
Dr. Rotow: Or for the next 4 years – exactly. There’s going to be the open question of who really needs this? In some sense, we may be guided by the path response during the surgery itself. I think there may be those who claim that if you have a pCR, do you really need additional therapy? We don’t know the answer, but it’s tempting to say we know the outcomes in event-free survival are extremely good with a pCR.
Dr. West: Which is only 20% or 25% of patients, so it’s not most.
Dr. Rotow: It’s not most, but it’s better than the 2% or so with chemotherapy alone. That’s real progress, and it’s nice to have that readout. For that 80% without a pCR, what to do? I suspect there will be variation from provider to provider and from patient to patient, depending on tolerability to prior therapy, the patient’s wishes around the goals of care, and the patient’s risk for autoimmune toxicities.
Maybe there’s a patient with underlying autoimmune disease who’s gotten their neoadjuvant therapy and done well. You don’t want to risk that ongoing risk of exposure. Perhaps a patient with no risk factors who desires very aggressive treatment might be interested in more treatment.
In KEYNOTE-671, I was interested in the PD-L1 subgroups. These did trend the way you expect, with better responses in PD-L1 high, but there were also good outcomes and benefit to immunotherapy with the perioperative strategy in PD-L1–negative patients.
Dr. West: That didn’t really exclude anybody.
Dr. Rotow: It didn’t exclude anybody. In CheckMate 816, everyone benefited, but the benefit was less with those PD-L1–negative patients.
Dr. West: True.
Dr. Rotow: Absent further data to guide me or any prospective data here comparing these strategies, I might lean toward a longer course of immunotherapy in that population in hopes of triggering a response. I suspect that there will be variation from clinician to clinician in that space.
Dr. West: This is a setting where I feel like I have equipoise. I really feel that the incremental benefit is pretty small.
Dr. Rotow: Small. I agree.
Dr. West: It’s, frankly, somewhat dubious. On the other hand, you’re in a situation where if you know that three of four patients will experience a relapse and less-than-amazing outcomes, it’s hard to leave something that’s FDA approved and studied and a well-sanctioned option on the table if this patient may have relapse later.
In the end, I feel like I’d like to offer this and discuss it with all my patients. I think it’s a great place for shared decision-making because if a patient hears about that and decides they’re not interested, I’ll be fine with that. I think that’s a very sensible approach, but I don’t want to make it unilaterally. Other patients may say they want every opportunity, and if it comes back, at least I’ll know I did everything we could.
Dr. Rotow: Exactly. I agree with your statement about equipoise. I truly think that this is present here in the situation, and that there’s room for discussion in both directions with patients.
Now, one caveat I’d like to add to all these data is that the data should not apply to patients with some of our classic nonsmoking-associated driver mutations. This is another piece to the neoadjuvant data that I think is worth commenting on – the need to get appropriate testing before initiation of therapy and the pitfalls of starting this kind of treatment without knowing full biomarker testing. I think that’s something we have to watch for in our clinical practice as well.
Dr. West: Perhaps especially if we’re talking about doing a year of postoperative and someone has an ALK rearrangement or an EGFR mutation and we didn’t know it. That is a group where we’re worried about a rapid transition and potentially prohibitive, even life-threatening, toxicities from not planning in advance for this. This is something you don’t want to give concurrently or one right on top of the other. You don’t want to give immunotherapy and then transition right to targeted therapy. It’s dangerous.
Dr. Rotow: Exactly. The stakes were already high with neoadjuvant alone, but at least you had that gap of the presurgical period, surgical recovery, and then initiation of adjuvant therapy, if needed, or at relapse. With a postoperative long adjuvant period, those stakes are elevated because the immunotherapy exposure continues, so it’s something to be mindful of.
Dr. West: We have a general sense that many, but not all, of the targets that we’re talking about are associated with low benefit from immunotherapy. It’s not that well studied. I think this is another place for individualized discussion of the pros and cons. They were included in the trial, but they probably benefit less.
Dr. Rotow: Exactly. I think with the best established, EGFR and ALK probably are not benefiting much. They were actually included in the trial. Many of the neoadjuvant studies do not allow them to enroll if they’re known. On the other end of that spectrum, I think KRAS is just fine to treat with immunotherapy.
Dr. West: Sure.
Dr. Rotow: It’s an actionable driver. It’s not a traditional nonsmoking-associated driver, and those do just fine.
Dr. West: The studies show that these patients benefit just as much, at least, as the other patients.
Dr. Rotow: Exactly. I would never withhold this form of therapy for a KRAS driver mutation. The others, I think, are still in a gray zone. Depending on the patient demographics and tobacco use, I may elicit more or less caution in that space.
Dr. West: Well, I think we’re going to have much to still tease apart, with room for judgment here without a strong sense of the data telling us exactly what to do.
Dr. Rotow: Exactly.
Dr. West: There’s a large amount of excitement and interest in these new data, but there are still many open questions. I hope we continue to mull it over as we get more data and more insight to shape our plans.
Dr. West is an associate professor at City of Hope Comprehensive Cancer Center in Duarte, Calif., and vice president of network strategy at AccessHope in Los Angeles. Dr. Rotow is the clinical director of the Lowe Center for Thoracic Oncology at the Dana-Farber Cancer Institute in Boston. Dr. West reported conflicts of interest with Ariad/Takeda, Bristol Myers Squibb, Boehringer Ingelheim, Spectrum, AstraZeneca, Celgene, Genentech/Roche, Pfizer, Merck, and Eli Lilly. Dr. Rotow reported conflicts of interest with Genentech, AstraZeneca,Guardant, and Janssen.
A version of this article first appeared on Medscape.com.
FROM ASCO 2023
Prior auth is a self-inflicted wound; is there a way out?
A few months ago, a close friend called me in a panic.
Her mother had been on an oral treatment for relapsed multiple myeloma for nearly 1 year. Originally, she had been prescribed the oral pill pomalidomide as part of her regimen, but it had caused cytopenias, constipation, and nausea even at lower doses. Now, she was tolerating her new pill, ixazomib, quite well.
Then, without so much as a letter from her insurance company, the patient’s pharmacy called to tell her that ixazomib was no longer covered without a peer-to-peer conversation given its cost.
The peer to peer left the oncology team and patient even more frustrated. After leaving the team on hold for nearly an hour, the insurance company denied the request for ixazomib. The hematologist conducting the peer-to-peer did not seem to understand why the patient had discontinued her previous medication.
Ultimately, the patient was forced to return to the old, less expensive drug. Since going back to pomalidomide, she has needed multiple dosing changes to manage the intense drug side effects.
I wish that stories like this were uncommon. I am often reminded of the ongoing challenges related to prior authorization when I see colleagues regaling the Twitterverse with their latest horror stories about an insurance company rejecting standard of care treatment or imaging. The data confirm the frequency of these experiences. Among a general cohort of physicians, the American Medical Association has reported that most practices complete 29 prior authorizations per physician every week and spend on average 14.6 hours, or nearly 2 full business days, of staff time each week on these requirements.
Given the growing burden of prior authorization, reform has now become a major policy issue at the federal and state level. Within the past year, organizations like the American Society of Clinical Oncology have released policy positions on prior authorization and are devoting sizable advocacy dollars to fight payers’ ability to enact it. Entire sections of the ASCO education book are devoted to detailing the negative effects of prior authorization, and recent ASCO-commissioned surveys have highlighted alarming statistics about how prior authorization harms patient care.
As a practicing oncologist, I welcome increased attention to the burdens of prior authorization. This attention shows that our professional organizations care about one of the largest contributors to burnout.
But as a researcher who studies systems of care, I worry that focusing our collective ire on health insurance companies is somewhat misplaced or unlikely to succeed.
Why? Because policy statements, Tweets, and legislation ignore the fact that prior authorization is a self-inflicted wound caused by decades of pharmaceutical companies, health systems, oncologists, and guideline bodies failing to factor costs of care into clinical practice.
Making cost-conscious oncology care a priority would be ideal, but it is also a tall order.
But, as costs continue to rise uncontrollably, the prior authorization system has ballooned out of control and reining in this system will be quite a challenge. Perhaps, we can look to my oncology practice at a Veterans Affairs Medical Center as a guide.
A self-inflicted wound
In oncology, most medications and imaging are subject to prior authorization, particularly higher-cost items, and dealing with prior authorization typically requires an army of staff. As enrollment in commercial health plans or Medicare Advantage skyrockets, prior authorization will only become more prevalent in oncology.
Why is this happening?
The lazy answer to this question is because health insurance companies put profits over patients.
However, prior authorization is a symptom, not a disease. And that symptom is exploding cancer care costs. The causes of high costs are varied: Pharmaceutical companies and device manufacturers set ungodly prices for drugs; oncologists and other specialists are often reimbursed for volume of care, not quality; and hospitals and health systems charge incredible amounts for routine labs and imaging, amounts that are orders of magnitude higher than what these services actually cost.
There is no easy solution to fix the drivers of such costs. One approach to rein in drug costs would be to allow Medicare to negotiate drug prices and set site-neutral pricing.
Another would be to incentivize doctors or health systems to reduce the use of high-cost services. But physicians are often unaware of the costs of a given treatment to a system or patient, and practices reimbursed by volume of care typically have little incentive to curb overall spending.
We are then left with payers, the primary means of reimbursing care and, specifically, utilization management, such as prior authorization and restrictive formularies, as the major mechanism to reduce this spending.
Prior authorization is broken
Here’s the rub though: Although payers are incentivized to curb costs through prior authorization, the data that they have to adjudicate therapeutic appropriateness are often terrible.
Administrative claims contain no data on biomarkers or performance status and are notoriously bad at reflecting basic information, such as a patient’s stage of cancer. For instance, the positive predictive value of claims-based algorithms to identify stage IV breast cancer is well below 50%, meaning that a claims algorithm that classifies a patient as stage IV is wrong more often than right.
If payers had better data, they could be more selective in their prior-authorization requirements. In other words, if payers could reliably identify who has metastatic breast cancer, an aromatase inhibitor, a common, well-accepted adjuvant therapy in hormone-positive, late-stage breast cancer, wouldn’t need prior authorization. What we have now is an inefficient system that sets prior authorization as a guardrail for most oncology care and then forces doctors to submit all relevant information about a patient to justify their treatment choices.
Keeping up with oncology treatment advances is also a challenge and requires tremendous expertise. Many insurers delegate their prior-authorization responsibilities to medication management companies, such as Magellan Rx or New Century Health, who maintain proprietary treatment pathways.
These companies anchor their prior authorization requirements to common guidelines. That would be fine if guideline-producing bodies like the National Comprehensive Cancer Network provided more firm recommendations on high-value treatments.
The NCCN recommendations, however, are expert-driven more than evidence or value driven. Often, therapies with less evidence make it into recommended treatment guidelines, and in some cases, the NCCN will equally recommend five options for treating a certain cancer, even when there is an obvious lower-cost option.
The effect of this, however, is that payers may then cover these five options, despite a 40-fold price difference among them, but then lean on requiring prior authorization for everything rather than being selective. Broad rather than targeted use of prior authorization alongside well-known issue like uncertain time lines, huge numbers of forms, and nonexperts doing peer to peers make for a huge mess.
What can we do?
How can we begin to solve the prior authorization crisis? A first step would be for guideline bodies to have more teeth in their recommendations. If NCCN and other guideline bodies, for instance, incorporated cost into their recommendations and designated these as “preferred” regimens, then clinicians could have better direction on therapy selection and payers could align their prior authorization policies with those recommendations. If patients had adverse effects with low-cost drugs, then a preferred alternative could be specified in such guidelines rather than subject patients, like my friend’s mother, to a toxic drug.
Second, payers could tailor the intensity of prior authorization requirements to the type of physician and clinical scenario at hand. Payers have rich data on practice patterns of oncologists. Payers should incentivize oncologists who follow guideline-based, high-value treatment pathways by lowering the need for frequent peer-to-peers or other prior authorization for “good performers.” This strategy, often termed gold carding, would use relief from prior authorization as a carrot.
Similarly, payers could reward practices that implement clinical pathways that enforce high-value care. For example, a practice could develop a treatment pathway that emphasizes access to urgent care to avoid hospitalizations as well as prioritizes access to relatively lower cost but equally effective options for therapy. If a payer reviewed and approved the pathway, perhaps payers could propose relief of future prior authorizations for practices whose oncologist practice on this pathway.
Third, payers could step up the intensity of prior authorization for certain high-cost or low-value treatments and lessen requirements for more routine services. For example, if every initial staging PET-CT required a peer to peer, oncologists would spend most of the day on the phone. Rather, lower-level tasks such as imaging may require a simple electronic EHR message, whereas high-cost items such as indefinite systemic therapy may require more frequent peer to peers.
Fourth, health systems and real-world data companies should devise better data sharing partnerships with payers so that payers could automatically examine attributes that clarify the choice of therapy. For example, if a payer could view that a patient had estrogen receptor/progesterone receptor–positive early-stage breast cancer post surgery, perhaps that payer would not require a prior authorization for an aromatase inhibitor. These real-time data sharing partnerships could reduce friction points in the system.
Finally, researchers and other groups should partner with payers to continually examine the effectiveness of any prior-authorization program. If a prior-authorization policy is no longer effective because evidence changes and evolves, then payers should consider retiring it.
In my primary oncology practice at a VA Medical Center, none of my treatments require an external prior authorization. Why? Because our local practice agreed to an established formulary, and national treatment pathways firmly specify a recommended treatment course.
Do I sometimes go off pathway? Yes, when I feel there’s a compelling reason. But that requires a structured electronic form to a central pharmacy body. I get a response within 24 hours, with no onerous prior authorization form or lengthy peer to peer.
Though there are plenty of unique qualities about the VA, the fact is that health systems and guideline bodies assuming the burden of cost containment could reduce prior-authorization requirements from payers.
Ultimately, the goal should be for oncologists to choose the highest-value treatment possible. Perhaps then, when the end-goal of cost-conscious oncology care with payers maintain an arm’s length from the patient-doctor relationship, we could all stop shouting at the wind about the burden of prior authorization.
Dr. Parikh is a medical oncologist and faculty member at the University of Pennsylvania, Philadelphia, and the Philadelphia VA Medical Center, an adjunct fellow at the Leonard Davis Institute of Health Economics, and senior clinical advisor at the Coalition to Transform Advanced Care. He reported conflicts of interest with GNS Healthcare, Nanology, Cancer Study Group, Embedded Healthcare, Veterans Affairs, PCF, National Palliative Care Research Center, MUSC, and Flatiron Health.
A version of this article first appeared on Medscape.com.
A few months ago, a close friend called me in a panic.
Her mother had been on an oral treatment for relapsed multiple myeloma for nearly 1 year. Originally, she had been prescribed the oral pill pomalidomide as part of her regimen, but it had caused cytopenias, constipation, and nausea even at lower doses. Now, she was tolerating her new pill, ixazomib, quite well.
Then, without so much as a letter from her insurance company, the patient’s pharmacy called to tell her that ixazomib was no longer covered without a peer-to-peer conversation given its cost.
The peer to peer left the oncology team and patient even more frustrated. After leaving the team on hold for nearly an hour, the insurance company denied the request for ixazomib. The hematologist conducting the peer-to-peer did not seem to understand why the patient had discontinued her previous medication.
Ultimately, the patient was forced to return to the old, less expensive drug. Since going back to pomalidomide, she has needed multiple dosing changes to manage the intense drug side effects.
I wish that stories like this were uncommon. I am often reminded of the ongoing challenges related to prior authorization when I see colleagues regaling the Twitterverse with their latest horror stories about an insurance company rejecting standard of care treatment or imaging. The data confirm the frequency of these experiences. Among a general cohort of physicians, the American Medical Association has reported that most practices complete 29 prior authorizations per physician every week and spend on average 14.6 hours, or nearly 2 full business days, of staff time each week on these requirements.
Given the growing burden of prior authorization, reform has now become a major policy issue at the federal and state level. Within the past year, organizations like the American Society of Clinical Oncology have released policy positions on prior authorization and are devoting sizable advocacy dollars to fight payers’ ability to enact it. Entire sections of the ASCO education book are devoted to detailing the negative effects of prior authorization, and recent ASCO-commissioned surveys have highlighted alarming statistics about how prior authorization harms patient care.
As a practicing oncologist, I welcome increased attention to the burdens of prior authorization. This attention shows that our professional organizations care about one of the largest contributors to burnout.
But as a researcher who studies systems of care, I worry that focusing our collective ire on health insurance companies is somewhat misplaced or unlikely to succeed.
Why? Because policy statements, Tweets, and legislation ignore the fact that prior authorization is a self-inflicted wound caused by decades of pharmaceutical companies, health systems, oncologists, and guideline bodies failing to factor costs of care into clinical practice.
Making cost-conscious oncology care a priority would be ideal, but it is also a tall order.
But, as costs continue to rise uncontrollably, the prior authorization system has ballooned out of control and reining in this system will be quite a challenge. Perhaps, we can look to my oncology practice at a Veterans Affairs Medical Center as a guide.
A self-inflicted wound
In oncology, most medications and imaging are subject to prior authorization, particularly higher-cost items, and dealing with prior authorization typically requires an army of staff. As enrollment in commercial health plans or Medicare Advantage skyrockets, prior authorization will only become more prevalent in oncology.
Why is this happening?
The lazy answer to this question is because health insurance companies put profits over patients.
However, prior authorization is a symptom, not a disease. And that symptom is exploding cancer care costs. The causes of high costs are varied: Pharmaceutical companies and device manufacturers set ungodly prices for drugs; oncologists and other specialists are often reimbursed for volume of care, not quality; and hospitals and health systems charge incredible amounts for routine labs and imaging, amounts that are orders of magnitude higher than what these services actually cost.
There is no easy solution to fix the drivers of such costs. One approach to rein in drug costs would be to allow Medicare to negotiate drug prices and set site-neutral pricing.
Another would be to incentivize doctors or health systems to reduce the use of high-cost services. But physicians are often unaware of the costs of a given treatment to a system or patient, and practices reimbursed by volume of care typically have little incentive to curb overall spending.
We are then left with payers, the primary means of reimbursing care and, specifically, utilization management, such as prior authorization and restrictive formularies, as the major mechanism to reduce this spending.
Prior authorization is broken
Here’s the rub though: Although payers are incentivized to curb costs through prior authorization, the data that they have to adjudicate therapeutic appropriateness are often terrible.
Administrative claims contain no data on biomarkers or performance status and are notoriously bad at reflecting basic information, such as a patient’s stage of cancer. For instance, the positive predictive value of claims-based algorithms to identify stage IV breast cancer is well below 50%, meaning that a claims algorithm that classifies a patient as stage IV is wrong more often than right.
If payers had better data, they could be more selective in their prior-authorization requirements. In other words, if payers could reliably identify who has metastatic breast cancer, an aromatase inhibitor, a common, well-accepted adjuvant therapy in hormone-positive, late-stage breast cancer, wouldn’t need prior authorization. What we have now is an inefficient system that sets prior authorization as a guardrail for most oncology care and then forces doctors to submit all relevant information about a patient to justify their treatment choices.
Keeping up with oncology treatment advances is also a challenge and requires tremendous expertise. Many insurers delegate their prior-authorization responsibilities to medication management companies, such as Magellan Rx or New Century Health, who maintain proprietary treatment pathways.
These companies anchor their prior authorization requirements to common guidelines. That would be fine if guideline-producing bodies like the National Comprehensive Cancer Network provided more firm recommendations on high-value treatments.
The NCCN recommendations, however, are expert-driven more than evidence or value driven. Often, therapies with less evidence make it into recommended treatment guidelines, and in some cases, the NCCN will equally recommend five options for treating a certain cancer, even when there is an obvious lower-cost option.
The effect of this, however, is that payers may then cover these five options, despite a 40-fold price difference among them, but then lean on requiring prior authorization for everything rather than being selective. Broad rather than targeted use of prior authorization alongside well-known issue like uncertain time lines, huge numbers of forms, and nonexperts doing peer to peers make for a huge mess.
What can we do?
How can we begin to solve the prior authorization crisis? A first step would be for guideline bodies to have more teeth in their recommendations. If NCCN and other guideline bodies, for instance, incorporated cost into their recommendations and designated these as “preferred” regimens, then clinicians could have better direction on therapy selection and payers could align their prior authorization policies with those recommendations. If patients had adverse effects with low-cost drugs, then a preferred alternative could be specified in such guidelines rather than subject patients, like my friend’s mother, to a toxic drug.
Second, payers could tailor the intensity of prior authorization requirements to the type of physician and clinical scenario at hand. Payers have rich data on practice patterns of oncologists. Payers should incentivize oncologists who follow guideline-based, high-value treatment pathways by lowering the need for frequent peer-to-peers or other prior authorization for “good performers.” This strategy, often termed gold carding, would use relief from prior authorization as a carrot.
Similarly, payers could reward practices that implement clinical pathways that enforce high-value care. For example, a practice could develop a treatment pathway that emphasizes access to urgent care to avoid hospitalizations as well as prioritizes access to relatively lower cost but equally effective options for therapy. If a payer reviewed and approved the pathway, perhaps payers could propose relief of future prior authorizations for practices whose oncologist practice on this pathway.
Third, payers could step up the intensity of prior authorization for certain high-cost or low-value treatments and lessen requirements for more routine services. For example, if every initial staging PET-CT required a peer to peer, oncologists would spend most of the day on the phone. Rather, lower-level tasks such as imaging may require a simple electronic EHR message, whereas high-cost items such as indefinite systemic therapy may require more frequent peer to peers.
Fourth, health systems and real-world data companies should devise better data sharing partnerships with payers so that payers could automatically examine attributes that clarify the choice of therapy. For example, if a payer could view that a patient had estrogen receptor/progesterone receptor–positive early-stage breast cancer post surgery, perhaps that payer would not require a prior authorization for an aromatase inhibitor. These real-time data sharing partnerships could reduce friction points in the system.
Finally, researchers and other groups should partner with payers to continually examine the effectiveness of any prior-authorization program. If a prior-authorization policy is no longer effective because evidence changes and evolves, then payers should consider retiring it.
In my primary oncology practice at a VA Medical Center, none of my treatments require an external prior authorization. Why? Because our local practice agreed to an established formulary, and national treatment pathways firmly specify a recommended treatment course.
Do I sometimes go off pathway? Yes, when I feel there’s a compelling reason. But that requires a structured electronic form to a central pharmacy body. I get a response within 24 hours, with no onerous prior authorization form or lengthy peer to peer.
Though there are plenty of unique qualities about the VA, the fact is that health systems and guideline bodies assuming the burden of cost containment could reduce prior-authorization requirements from payers.
Ultimately, the goal should be for oncologists to choose the highest-value treatment possible. Perhaps then, when the end-goal of cost-conscious oncology care with payers maintain an arm’s length from the patient-doctor relationship, we could all stop shouting at the wind about the burden of prior authorization.
Dr. Parikh is a medical oncologist and faculty member at the University of Pennsylvania, Philadelphia, and the Philadelphia VA Medical Center, an adjunct fellow at the Leonard Davis Institute of Health Economics, and senior clinical advisor at the Coalition to Transform Advanced Care. He reported conflicts of interest with GNS Healthcare, Nanology, Cancer Study Group, Embedded Healthcare, Veterans Affairs, PCF, National Palliative Care Research Center, MUSC, and Flatiron Health.
A version of this article first appeared on Medscape.com.
A few months ago, a close friend called me in a panic.
Her mother had been on an oral treatment for relapsed multiple myeloma for nearly 1 year. Originally, she had been prescribed the oral pill pomalidomide as part of her regimen, but it had caused cytopenias, constipation, and nausea even at lower doses. Now, she was tolerating her new pill, ixazomib, quite well.
Then, without so much as a letter from her insurance company, the patient’s pharmacy called to tell her that ixazomib was no longer covered without a peer-to-peer conversation given its cost.
The peer to peer left the oncology team and patient even more frustrated. After leaving the team on hold for nearly an hour, the insurance company denied the request for ixazomib. The hematologist conducting the peer-to-peer did not seem to understand why the patient had discontinued her previous medication.
Ultimately, the patient was forced to return to the old, less expensive drug. Since going back to pomalidomide, she has needed multiple dosing changes to manage the intense drug side effects.
I wish that stories like this were uncommon. I am often reminded of the ongoing challenges related to prior authorization when I see colleagues regaling the Twitterverse with their latest horror stories about an insurance company rejecting standard of care treatment or imaging. The data confirm the frequency of these experiences. Among a general cohort of physicians, the American Medical Association has reported that most practices complete 29 prior authorizations per physician every week and spend on average 14.6 hours, or nearly 2 full business days, of staff time each week on these requirements.
Given the growing burden of prior authorization, reform has now become a major policy issue at the federal and state level. Within the past year, organizations like the American Society of Clinical Oncology have released policy positions on prior authorization and are devoting sizable advocacy dollars to fight payers’ ability to enact it. Entire sections of the ASCO education book are devoted to detailing the negative effects of prior authorization, and recent ASCO-commissioned surveys have highlighted alarming statistics about how prior authorization harms patient care.
As a practicing oncologist, I welcome increased attention to the burdens of prior authorization. This attention shows that our professional organizations care about one of the largest contributors to burnout.
But as a researcher who studies systems of care, I worry that focusing our collective ire on health insurance companies is somewhat misplaced or unlikely to succeed.
Why? Because policy statements, Tweets, and legislation ignore the fact that prior authorization is a self-inflicted wound caused by decades of pharmaceutical companies, health systems, oncologists, and guideline bodies failing to factor costs of care into clinical practice.
Making cost-conscious oncology care a priority would be ideal, but it is also a tall order.
But, as costs continue to rise uncontrollably, the prior authorization system has ballooned out of control and reining in this system will be quite a challenge. Perhaps, we can look to my oncology practice at a Veterans Affairs Medical Center as a guide.
A self-inflicted wound
In oncology, most medications and imaging are subject to prior authorization, particularly higher-cost items, and dealing with prior authorization typically requires an army of staff. As enrollment in commercial health plans or Medicare Advantage skyrockets, prior authorization will only become more prevalent in oncology.
Why is this happening?
The lazy answer to this question is because health insurance companies put profits over patients.
However, prior authorization is a symptom, not a disease. And that symptom is exploding cancer care costs. The causes of high costs are varied: Pharmaceutical companies and device manufacturers set ungodly prices for drugs; oncologists and other specialists are often reimbursed for volume of care, not quality; and hospitals and health systems charge incredible amounts for routine labs and imaging, amounts that are orders of magnitude higher than what these services actually cost.
There is no easy solution to fix the drivers of such costs. One approach to rein in drug costs would be to allow Medicare to negotiate drug prices and set site-neutral pricing.
Another would be to incentivize doctors or health systems to reduce the use of high-cost services. But physicians are often unaware of the costs of a given treatment to a system or patient, and practices reimbursed by volume of care typically have little incentive to curb overall spending.
We are then left with payers, the primary means of reimbursing care and, specifically, utilization management, such as prior authorization and restrictive formularies, as the major mechanism to reduce this spending.
Prior authorization is broken
Here’s the rub though: Although payers are incentivized to curb costs through prior authorization, the data that they have to adjudicate therapeutic appropriateness are often terrible.
Administrative claims contain no data on biomarkers or performance status and are notoriously bad at reflecting basic information, such as a patient’s stage of cancer. For instance, the positive predictive value of claims-based algorithms to identify stage IV breast cancer is well below 50%, meaning that a claims algorithm that classifies a patient as stage IV is wrong more often than right.
If payers had better data, they could be more selective in their prior-authorization requirements. In other words, if payers could reliably identify who has metastatic breast cancer, an aromatase inhibitor, a common, well-accepted adjuvant therapy in hormone-positive, late-stage breast cancer, wouldn’t need prior authorization. What we have now is an inefficient system that sets prior authorization as a guardrail for most oncology care and then forces doctors to submit all relevant information about a patient to justify their treatment choices.
Keeping up with oncology treatment advances is also a challenge and requires tremendous expertise. Many insurers delegate their prior-authorization responsibilities to medication management companies, such as Magellan Rx or New Century Health, who maintain proprietary treatment pathways.
These companies anchor their prior authorization requirements to common guidelines. That would be fine if guideline-producing bodies like the National Comprehensive Cancer Network provided more firm recommendations on high-value treatments.
The NCCN recommendations, however, are expert-driven more than evidence or value driven. Often, therapies with less evidence make it into recommended treatment guidelines, and in some cases, the NCCN will equally recommend five options for treating a certain cancer, even when there is an obvious lower-cost option.
The effect of this, however, is that payers may then cover these five options, despite a 40-fold price difference among them, but then lean on requiring prior authorization for everything rather than being selective. Broad rather than targeted use of prior authorization alongside well-known issue like uncertain time lines, huge numbers of forms, and nonexperts doing peer to peers make for a huge mess.
What can we do?
How can we begin to solve the prior authorization crisis? A first step would be for guideline bodies to have more teeth in their recommendations. If NCCN and other guideline bodies, for instance, incorporated cost into their recommendations and designated these as “preferred” regimens, then clinicians could have better direction on therapy selection and payers could align their prior authorization policies with those recommendations. If patients had adverse effects with low-cost drugs, then a preferred alternative could be specified in such guidelines rather than subject patients, like my friend’s mother, to a toxic drug.
Second, payers could tailor the intensity of prior authorization requirements to the type of physician and clinical scenario at hand. Payers have rich data on practice patterns of oncologists. Payers should incentivize oncologists who follow guideline-based, high-value treatment pathways by lowering the need for frequent peer-to-peers or other prior authorization for “good performers.” This strategy, often termed gold carding, would use relief from prior authorization as a carrot.
Similarly, payers could reward practices that implement clinical pathways that enforce high-value care. For example, a practice could develop a treatment pathway that emphasizes access to urgent care to avoid hospitalizations as well as prioritizes access to relatively lower cost but equally effective options for therapy. If a payer reviewed and approved the pathway, perhaps payers could propose relief of future prior authorizations for practices whose oncologist practice on this pathway.
Third, payers could step up the intensity of prior authorization for certain high-cost or low-value treatments and lessen requirements for more routine services. For example, if every initial staging PET-CT required a peer to peer, oncologists would spend most of the day on the phone. Rather, lower-level tasks such as imaging may require a simple electronic EHR message, whereas high-cost items such as indefinite systemic therapy may require more frequent peer to peers.
Fourth, health systems and real-world data companies should devise better data sharing partnerships with payers so that payers could automatically examine attributes that clarify the choice of therapy. For example, if a payer could view that a patient had estrogen receptor/progesterone receptor–positive early-stage breast cancer post surgery, perhaps that payer would not require a prior authorization for an aromatase inhibitor. These real-time data sharing partnerships could reduce friction points in the system.
Finally, researchers and other groups should partner with payers to continually examine the effectiveness of any prior-authorization program. If a prior-authorization policy is no longer effective because evidence changes and evolves, then payers should consider retiring it.
In my primary oncology practice at a VA Medical Center, none of my treatments require an external prior authorization. Why? Because our local practice agreed to an established formulary, and national treatment pathways firmly specify a recommended treatment course.
Do I sometimes go off pathway? Yes, when I feel there’s a compelling reason. But that requires a structured electronic form to a central pharmacy body. I get a response within 24 hours, with no onerous prior authorization form or lengthy peer to peer.
Though there are plenty of unique qualities about the VA, the fact is that health systems and guideline bodies assuming the burden of cost containment could reduce prior-authorization requirements from payers.
Ultimately, the goal should be for oncologists to choose the highest-value treatment possible. Perhaps then, when the end-goal of cost-conscious oncology care with payers maintain an arm’s length from the patient-doctor relationship, we could all stop shouting at the wind about the burden of prior authorization.
Dr. Parikh is a medical oncologist and faculty member at the University of Pennsylvania, Philadelphia, and the Philadelphia VA Medical Center, an adjunct fellow at the Leonard Davis Institute of Health Economics, and senior clinical advisor at the Coalition to Transform Advanced Care. He reported conflicts of interest with GNS Healthcare, Nanology, Cancer Study Group, Embedded Healthcare, Veterans Affairs, PCF, National Palliative Care Research Center, MUSC, and Flatiron Health.
A version of this article first appeared on Medscape.com.
PRISm and nonspecific pattern: New insights in lung testing interpretation
The recent statement on interpretive strategies for lung testing uses the acronym PRISm for preserved ratio impaired spirometry. PRISm identifies patients with a normal forced expiratory volume in 1 second/forced vital capacity ratio but abnormal FEV1 and/or FVC (usually both). Most medical students are taught to call this a “restrictive pattern,” and every first-year pulmonary fellow orders full lung volumes when they see it. If total lung capacity (TLC) is normal, PRISm becomes the nonspecific pattern. If TLC is low, then the patient has “true” restriction, and if it’s elevated, then hyperinflation may be present.
The traditional classification scheme for basic spirometry interpretation (normal, restricted, obstructed, or mixed) is simple and conceptually clear. It turns out that many with this pattern won’t have an abnormal TLC, so the name is, in some ways, a misnomer and can be misleading. Enter PRISm, a more descriptive and inclusive term. The phrase also lends itself to a phonetic acronym that is fun to say, easy to remember, and likely to catch on with learners.
Information on occurrence and clinical behavior comes from large cohorts with basic spirometry, but without full lung volumes because PRISm no longer applies once TLC is determined. As may be expected, prevalence varies by the population studied. Estimates for general populations have been in the 7%-12% range; however, one study examining a database of patients with clinical spirometry referrals found a prevalence of 22.3%. Rates may be far higher in low- and middle-income countries. Identified risk factors include sex, tobacco use, and body mass index; the presence of PRISm is associated with respiratory symptoms and mortality. Thus, PRISm is common and it matters.
Along with PRISm, the nonspecific pattern is a new addition, if not a new concept, to the 2022 interpretative strategies statement. As with PRISm, the title is necessarily broad, though far less imaginative. Defined by reductions in FEV1 and FVC and a normal TLC, the nonspecific pattern has classically been considered a marker of early airway disease. The idea is that early, heterogeneous closure of distal segments of the bronchial tree can reduce total volume during a forced expiration before affecting the FEV1/FVC. The fact that the TLC is not a forced maneuver means there is proportionately less effect from more collapsible/susceptible smaller units. More recent data suggest that there are additional causes.
Because the nonspecific pattern requires full lung volumes, we have less population-level data than for PRISm. Estimated prevalence is approximately 9.5% in patients with complete test results. The two most common causes are obesity and airway obstruction, and the pattern is relatively stable over time. Notably, an increase in specific airway resistance or TLC minus alveolar volume difference predicts progression to frank obstruction on spirometry.
The physiologic changes that obesity inflicts on the lung have been well described. Patients with obesity breathe at lower lung volumes and are therefore susceptible to small airway closure at rest and during forced expiration. There is no doubt that the increased recognition of PRISm and the nonspecific pattern is in part related to the worldwide rise in obesity rates.
Key takeaways
In summary, PRISm and the nonspecific pattern are now part of the classification scheme we use for spirometry and full lung volumes, respectively. They should be included in interpretations given their diagnostic and predictive value. Airway disease and obesity are common causes and often coexist with either pattern. Many will not have a true, restrictive lung deficit, and a reductionist approach to interpretation is likely to lead to erroneous diagnoses. There were many important updates included in the 2022 iteration on lung testing interpretation that should not fly under the radar.
Dr. Holley is professor of medicine at Uniformed Services University in Bethesda, Md., and a pulmonary/sleep and critical care medicine physician at MedStar Washington Hospital Center in Washington. He disclosed ties with CHEST College, Metapharm, and WebMD.
A version of this article appeared on Medscape.com.
The recent statement on interpretive strategies for lung testing uses the acronym PRISm for preserved ratio impaired spirometry. PRISm identifies patients with a normal forced expiratory volume in 1 second/forced vital capacity ratio but abnormal FEV1 and/or FVC (usually both). Most medical students are taught to call this a “restrictive pattern,” and every first-year pulmonary fellow orders full lung volumes when they see it. If total lung capacity (TLC) is normal, PRISm becomes the nonspecific pattern. If TLC is low, then the patient has “true” restriction, and if it’s elevated, then hyperinflation may be present.
The traditional classification scheme for basic spirometry interpretation (normal, restricted, obstructed, or mixed) is simple and conceptually clear. It turns out that many with this pattern won’t have an abnormal TLC, so the name is, in some ways, a misnomer and can be misleading. Enter PRISm, a more descriptive and inclusive term. The phrase also lends itself to a phonetic acronym that is fun to say, easy to remember, and likely to catch on with learners.
Information on occurrence and clinical behavior comes from large cohorts with basic spirometry, but without full lung volumes because PRISm no longer applies once TLC is determined. As may be expected, prevalence varies by the population studied. Estimates for general populations have been in the 7%-12% range; however, one study examining a database of patients with clinical spirometry referrals found a prevalence of 22.3%. Rates may be far higher in low- and middle-income countries. Identified risk factors include sex, tobacco use, and body mass index; the presence of PRISm is associated with respiratory symptoms and mortality. Thus, PRISm is common and it matters.
Along with PRISm, the nonspecific pattern is a new addition, if not a new concept, to the 2022 interpretative strategies statement. As with PRISm, the title is necessarily broad, though far less imaginative. Defined by reductions in FEV1 and FVC and a normal TLC, the nonspecific pattern has classically been considered a marker of early airway disease. The idea is that early, heterogeneous closure of distal segments of the bronchial tree can reduce total volume during a forced expiration before affecting the FEV1/FVC. The fact that the TLC is not a forced maneuver means there is proportionately less effect from more collapsible/susceptible smaller units. More recent data suggest that there are additional causes.
Because the nonspecific pattern requires full lung volumes, we have less population-level data than for PRISm. Estimated prevalence is approximately 9.5% in patients with complete test results. The two most common causes are obesity and airway obstruction, and the pattern is relatively stable over time. Notably, an increase in specific airway resistance or TLC minus alveolar volume difference predicts progression to frank obstruction on spirometry.
The physiologic changes that obesity inflicts on the lung have been well described. Patients with obesity breathe at lower lung volumes and are therefore susceptible to small airway closure at rest and during forced expiration. There is no doubt that the increased recognition of PRISm and the nonspecific pattern is in part related to the worldwide rise in obesity rates.
Key takeaways
In summary, PRISm and the nonspecific pattern are now part of the classification scheme we use for spirometry and full lung volumes, respectively. They should be included in interpretations given their diagnostic and predictive value. Airway disease and obesity are common causes and often coexist with either pattern. Many will not have a true, restrictive lung deficit, and a reductionist approach to interpretation is likely to lead to erroneous diagnoses. There were many important updates included in the 2022 iteration on lung testing interpretation that should not fly under the radar.
Dr. Holley is professor of medicine at Uniformed Services University in Bethesda, Md., and a pulmonary/sleep and critical care medicine physician at MedStar Washington Hospital Center in Washington. He disclosed ties with CHEST College, Metapharm, and WebMD.
A version of this article appeared on Medscape.com.
The recent statement on interpretive strategies for lung testing uses the acronym PRISm for preserved ratio impaired spirometry. PRISm identifies patients with a normal forced expiratory volume in 1 second/forced vital capacity ratio but abnormal FEV1 and/or FVC (usually both). Most medical students are taught to call this a “restrictive pattern,” and every first-year pulmonary fellow orders full lung volumes when they see it. If total lung capacity (TLC) is normal, PRISm becomes the nonspecific pattern. If TLC is low, then the patient has “true” restriction, and if it’s elevated, then hyperinflation may be present.
The traditional classification scheme for basic spirometry interpretation (normal, restricted, obstructed, or mixed) is simple and conceptually clear. It turns out that many with this pattern won’t have an abnormal TLC, so the name is, in some ways, a misnomer and can be misleading. Enter PRISm, a more descriptive and inclusive term. The phrase also lends itself to a phonetic acronym that is fun to say, easy to remember, and likely to catch on with learners.
Information on occurrence and clinical behavior comes from large cohorts with basic spirometry, but without full lung volumes because PRISm no longer applies once TLC is determined. As may be expected, prevalence varies by the population studied. Estimates for general populations have been in the 7%-12% range; however, one study examining a database of patients with clinical spirometry referrals found a prevalence of 22.3%. Rates may be far higher in low- and middle-income countries. Identified risk factors include sex, tobacco use, and body mass index; the presence of PRISm is associated with respiratory symptoms and mortality. Thus, PRISm is common and it matters.
Along with PRISm, the nonspecific pattern is a new addition, if not a new concept, to the 2022 interpretative strategies statement. As with PRISm, the title is necessarily broad, though far less imaginative. Defined by reductions in FEV1 and FVC and a normal TLC, the nonspecific pattern has classically been considered a marker of early airway disease. The idea is that early, heterogeneous closure of distal segments of the bronchial tree can reduce total volume during a forced expiration before affecting the FEV1/FVC. The fact that the TLC is not a forced maneuver means there is proportionately less effect from more collapsible/susceptible smaller units. More recent data suggest that there are additional causes.
Because the nonspecific pattern requires full lung volumes, we have less population-level data than for PRISm. Estimated prevalence is approximately 9.5% in patients with complete test results. The two most common causes are obesity and airway obstruction, and the pattern is relatively stable over time. Notably, an increase in specific airway resistance or TLC minus alveolar volume difference predicts progression to frank obstruction on spirometry.
The physiologic changes that obesity inflicts on the lung have been well described. Patients with obesity breathe at lower lung volumes and are therefore susceptible to small airway closure at rest and during forced expiration. There is no doubt that the increased recognition of PRISm and the nonspecific pattern is in part related to the worldwide rise in obesity rates.
Key takeaways
In summary, PRISm and the nonspecific pattern are now part of the classification scheme we use for spirometry and full lung volumes, respectively. They should be included in interpretations given their diagnostic and predictive value. Airway disease and obesity are common causes and often coexist with either pattern. Many will not have a true, restrictive lung deficit, and a reductionist approach to interpretation is likely to lead to erroneous diagnoses. There were many important updates included in the 2022 iteration on lung testing interpretation that should not fly under the radar.
Dr. Holley is professor of medicine at Uniformed Services University in Bethesda, Md., and a pulmonary/sleep and critical care medicine physician at MedStar Washington Hospital Center in Washington. He disclosed ties with CHEST College, Metapharm, and WebMD.
A version of this article appeared on Medscape.com.
Scrubs & Heels Summit 2023: Filling a void for women in GI
.1-3 This gender disparity arises from a multitude of factors including lack of effective mentoring, unequal leadership and career advancement opportunities, and pay inequity. In this context, The Scrubs & Heels Leadership Summit (S&H) was launched in 2022 focused on the professional and personal development of women in gastroenterology.
I had the great pleasure and honor of attending the 2023 summit which took place in February in Rancho Palos Verdes, Calif. There were nearly 200 attendees ranging from trainees to midcareer and senior gastroenterologists and other health care professionals from both academia and private practices across the nation. The weekend course was directed by S&H cofounders, Dr. Aline Charabaty and Dr. Anita Afzali, and cochaired by Dr. Amy Oxentenko and Dr. Aja McCutchen.
The 2-day summit opened with a presentation by Sally Helgesen, author of How Women Rise, describing the 12 common habits that often hold women back in career advancement, promotion, or opportunities. Dr. Aline Charabaty addressed the myth of women needing to fulfill the role of superwoman or have suprahuman abilities. Attendees were challenged to reframe this societal construct and begin to find balance and the reasonable choice to switch to part-time work and, as Dr. Aja McCutch emphasized, dial-down responsibilities to maintain wellness when life has competing priorities.
Dr. Amy Oxentenko shared her personal journey to success and instilled the importance of engaging with community and society at large. We then heard from Dr. Neena Abraham on how to gracefully embrace transitions in our professional lives, whether intentionally sought or natural progressions of a career. She encouraged attendees to control our own narrative and seek challenges that promote growth. We explored different practice models with Dr. Caroline Hwang and learned strategies of switching from academics to private practice or vice versa. We also heard from cofounder Dr. Anita Afzali on becoming a physician executive and the importance of staying connected to patient care when rising in ranks of leadership.
The second day opened with a keynote address delivered by Dr. Marla Dubinsky detailing her journey of becoming a CEO of a publicly-traded company while retaining her role as professor and chief of pediatric gastroenterology in a large academic institution. Attendees were provided with a master class on discovering ways to inspire our inner entrepreneur and highlighted the benefit of physicians, especially women, in being effective business leaders. This talk was followed by a talk by Phil Schoenfeld, MD, FACS, editor-in-chief of Evidence-Based GI for the American College of Gastroenterology. He spoke on the importance of male allyship for women in GI and shared his personal experiences and challenges with allyship.
The summit included a breakout session by Dr. Rashmi Advani designed for residents to hear tips on how to have a successful fellowship match and for fellows to embrace a steep learning curve when starting and included tips for efficiency. Additional breakout sessions included learning ergonomic strategies for positioning and scope-holding, vocal-cord exercises before giving oral presentations, and how to formulate a business plan and negotiate a contract.
We ended the summit with uplifting advice from executive coaches Sonia Narang and Dr. Dawn Sears who taught us the art of leaning into opportunities, mansizing aspirations, finding coconspirators for amplification of female GI leaders, and supporting our colleagues personally and professionally.
Three key takeaway messages:
- Recognize your self-worth and the contributions you bring to your patients and community as a whole.
- Lean into the importance of vocalizing your asks, advocating for yourself, building your brand, and showcasing your accomplishments.
- Be mindful of the balance between the time and energy you dedicate towards goals that bring you recognition and fuel your passion and your mental, physical, and emotional health.
As a trainee, I benefited tremendously from attending and expanding my professional network of mentors, sponsors and colleagues. I am encouraged by this programming and hope to see more of it in the future.
Contributors to this article included: Rashmi Advani, MD2; Anita Afzali, MD3; Aline Charabaty, MD.4
Neither Dr. Syed, nor the article contributors, had financial conflicts of interest associated with this article. The AGA was represented at the Scrubs and Heels Summit as a society partner committed to the advancement of women in GI. AGA is building on years of efforts to bolster leadership, mentorship, and sponsorship among women in GI through its annual women’s leadership conference and most recently with its 2022 regional women in GI workshops held around the country that led to the development of a comprehensive gender equity strategy designed to build an environment of gender equity in the field of GI so that all can thrive.
Institutions and social media handle
1. Santa Clara Valley Medical Center (San Jose, Calif), @noorannemd
2. Cedars Sinai (Los Angeles), @AdvaniRashmiMD
3. University of Cincinnati, @IBD_Afzali
4. Johns Hopkins Medicine (Washington), @DCharabaty
References
Advani R et al. Gender-specific attitudes of internal medicine residents toward gastroenterology. Dig Dis Sci. 2022 Nov;67(11):5044-52.
American Association of Medical Colleges. Diversity in Medicine: Facts and Figures (2019).
Elta GH. The challenges of being a female gastroenterologist. Gastroenterol Clin North Am. 2011 Jun;40(2):441-7.
Burke CA et al. Gender disparity in the practice of gastroenterology: The first 5 years of a career. Am J Gastroenterol 2005;100:259-64
David, Yakira N. et al. Gender-specific factors influencing gastroenterologists to pursue careers in advanced endoscopy: perceptions vs reality. Journal of the American College of Gastroenterology, ACG 116.3 (2021):539-50.
Rabinowitz LG et al. Gender dynamics in education and practice of gastroenterology. Gastrointest Endosc. 2021;93:1047-56.
Rabinowitz LG et al. Survey finds gender disparities impact both women mentors and mentees in gastroenterology. Journal of the American College of Gastroenterology, ACG 2021;116:1876-84.
.1-3 This gender disparity arises from a multitude of factors including lack of effective mentoring, unequal leadership and career advancement opportunities, and pay inequity. In this context, The Scrubs & Heels Leadership Summit (S&H) was launched in 2022 focused on the professional and personal development of women in gastroenterology.
I had the great pleasure and honor of attending the 2023 summit which took place in February in Rancho Palos Verdes, Calif. There were nearly 200 attendees ranging from trainees to midcareer and senior gastroenterologists and other health care professionals from both academia and private practices across the nation. The weekend course was directed by S&H cofounders, Dr. Aline Charabaty and Dr. Anita Afzali, and cochaired by Dr. Amy Oxentenko and Dr. Aja McCutchen.
The 2-day summit opened with a presentation by Sally Helgesen, author of How Women Rise, describing the 12 common habits that often hold women back in career advancement, promotion, or opportunities. Dr. Aline Charabaty addressed the myth of women needing to fulfill the role of superwoman or have suprahuman abilities. Attendees were challenged to reframe this societal construct and begin to find balance and the reasonable choice to switch to part-time work and, as Dr. Aja McCutch emphasized, dial-down responsibilities to maintain wellness when life has competing priorities.
Dr. Amy Oxentenko shared her personal journey to success and instilled the importance of engaging with community and society at large. We then heard from Dr. Neena Abraham on how to gracefully embrace transitions in our professional lives, whether intentionally sought or natural progressions of a career. She encouraged attendees to control our own narrative and seek challenges that promote growth. We explored different practice models with Dr. Caroline Hwang and learned strategies of switching from academics to private practice or vice versa. We also heard from cofounder Dr. Anita Afzali on becoming a physician executive and the importance of staying connected to patient care when rising in ranks of leadership.
The second day opened with a keynote address delivered by Dr. Marla Dubinsky detailing her journey of becoming a CEO of a publicly-traded company while retaining her role as professor and chief of pediatric gastroenterology in a large academic institution. Attendees were provided with a master class on discovering ways to inspire our inner entrepreneur and highlighted the benefit of physicians, especially women, in being effective business leaders. This talk was followed by a talk by Phil Schoenfeld, MD, FACS, editor-in-chief of Evidence-Based GI for the American College of Gastroenterology. He spoke on the importance of male allyship for women in GI and shared his personal experiences and challenges with allyship.
The summit included a breakout session by Dr. Rashmi Advani designed for residents to hear tips on how to have a successful fellowship match and for fellows to embrace a steep learning curve when starting and included tips for efficiency. Additional breakout sessions included learning ergonomic strategies for positioning and scope-holding, vocal-cord exercises before giving oral presentations, and how to formulate a business plan and negotiate a contract.
We ended the summit with uplifting advice from executive coaches Sonia Narang and Dr. Dawn Sears who taught us the art of leaning into opportunities, mansizing aspirations, finding coconspirators for amplification of female GI leaders, and supporting our colleagues personally and professionally.
Three key takeaway messages:
- Recognize your self-worth and the contributions you bring to your patients and community as a whole.
- Lean into the importance of vocalizing your asks, advocating for yourself, building your brand, and showcasing your accomplishments.
- Be mindful of the balance between the time and energy you dedicate towards goals that bring you recognition and fuel your passion and your mental, physical, and emotional health.
As a trainee, I benefited tremendously from attending and expanding my professional network of mentors, sponsors and colleagues. I am encouraged by this programming and hope to see more of it in the future.
Contributors to this article included: Rashmi Advani, MD2; Anita Afzali, MD3; Aline Charabaty, MD.4
Neither Dr. Syed, nor the article contributors, had financial conflicts of interest associated with this article. The AGA was represented at the Scrubs and Heels Summit as a society partner committed to the advancement of women in GI. AGA is building on years of efforts to bolster leadership, mentorship, and sponsorship among women in GI through its annual women’s leadership conference and most recently with its 2022 regional women in GI workshops held around the country that led to the development of a comprehensive gender equity strategy designed to build an environment of gender equity in the field of GI so that all can thrive.
Institutions and social media handle
1. Santa Clara Valley Medical Center (San Jose, Calif), @noorannemd
2. Cedars Sinai (Los Angeles), @AdvaniRashmiMD
3. University of Cincinnati, @IBD_Afzali
4. Johns Hopkins Medicine (Washington), @DCharabaty
References
Advani R et al. Gender-specific attitudes of internal medicine residents toward gastroenterology. Dig Dis Sci. 2022 Nov;67(11):5044-52.
American Association of Medical Colleges. Diversity in Medicine: Facts and Figures (2019).
Elta GH. The challenges of being a female gastroenterologist. Gastroenterol Clin North Am. 2011 Jun;40(2):441-7.
Burke CA et al. Gender disparity in the practice of gastroenterology: The first 5 years of a career. Am J Gastroenterol 2005;100:259-64
David, Yakira N. et al. Gender-specific factors influencing gastroenterologists to pursue careers in advanced endoscopy: perceptions vs reality. Journal of the American College of Gastroenterology, ACG 116.3 (2021):539-50.
Rabinowitz LG et al. Gender dynamics in education and practice of gastroenterology. Gastrointest Endosc. 2021;93:1047-56.
Rabinowitz LG et al. Survey finds gender disparities impact both women mentors and mentees in gastroenterology. Journal of the American College of Gastroenterology, ACG 2021;116:1876-84.
.1-3 This gender disparity arises from a multitude of factors including lack of effective mentoring, unequal leadership and career advancement opportunities, and pay inequity. In this context, The Scrubs & Heels Leadership Summit (S&H) was launched in 2022 focused on the professional and personal development of women in gastroenterology.
I had the great pleasure and honor of attending the 2023 summit which took place in February in Rancho Palos Verdes, Calif. There were nearly 200 attendees ranging from trainees to midcareer and senior gastroenterologists and other health care professionals from both academia and private practices across the nation. The weekend course was directed by S&H cofounders, Dr. Aline Charabaty and Dr. Anita Afzali, and cochaired by Dr. Amy Oxentenko and Dr. Aja McCutchen.
The 2-day summit opened with a presentation by Sally Helgesen, author of How Women Rise, describing the 12 common habits that often hold women back in career advancement, promotion, or opportunities. Dr. Aline Charabaty addressed the myth of women needing to fulfill the role of superwoman or have suprahuman abilities. Attendees were challenged to reframe this societal construct and begin to find balance and the reasonable choice to switch to part-time work and, as Dr. Aja McCutch emphasized, dial-down responsibilities to maintain wellness when life has competing priorities.
Dr. Amy Oxentenko shared her personal journey to success and instilled the importance of engaging with community and society at large. We then heard from Dr. Neena Abraham on how to gracefully embrace transitions in our professional lives, whether intentionally sought or natural progressions of a career. She encouraged attendees to control our own narrative and seek challenges that promote growth. We explored different practice models with Dr. Caroline Hwang and learned strategies of switching from academics to private practice or vice versa. We also heard from cofounder Dr. Anita Afzali on becoming a physician executive and the importance of staying connected to patient care when rising in ranks of leadership.
The second day opened with a keynote address delivered by Dr. Marla Dubinsky detailing her journey of becoming a CEO of a publicly-traded company while retaining her role as professor and chief of pediatric gastroenterology in a large academic institution. Attendees were provided with a master class on discovering ways to inspire our inner entrepreneur and highlighted the benefit of physicians, especially women, in being effective business leaders. This talk was followed by a talk by Phil Schoenfeld, MD, FACS, editor-in-chief of Evidence-Based GI for the American College of Gastroenterology. He spoke on the importance of male allyship for women in GI and shared his personal experiences and challenges with allyship.
The summit included a breakout session by Dr. Rashmi Advani designed for residents to hear tips on how to have a successful fellowship match and for fellows to embrace a steep learning curve when starting and included tips for efficiency. Additional breakout sessions included learning ergonomic strategies for positioning and scope-holding, vocal-cord exercises before giving oral presentations, and how to formulate a business plan and negotiate a contract.
We ended the summit with uplifting advice from executive coaches Sonia Narang and Dr. Dawn Sears who taught us the art of leaning into opportunities, mansizing aspirations, finding coconspirators for amplification of female GI leaders, and supporting our colleagues personally and professionally.
Three key takeaway messages:
- Recognize your self-worth and the contributions you bring to your patients and community as a whole.
- Lean into the importance of vocalizing your asks, advocating for yourself, building your brand, and showcasing your accomplishments.
- Be mindful of the balance between the time and energy you dedicate towards goals that bring you recognition and fuel your passion and your mental, physical, and emotional health.
As a trainee, I benefited tremendously from attending and expanding my professional network of mentors, sponsors and colleagues. I am encouraged by this programming and hope to see more of it in the future.
Contributors to this article included: Rashmi Advani, MD2; Anita Afzali, MD3; Aline Charabaty, MD.4
Neither Dr. Syed, nor the article contributors, had financial conflicts of interest associated with this article. The AGA was represented at the Scrubs and Heels Summit as a society partner committed to the advancement of women in GI. AGA is building on years of efforts to bolster leadership, mentorship, and sponsorship among women in GI through its annual women’s leadership conference and most recently with its 2022 regional women in GI workshops held around the country that led to the development of a comprehensive gender equity strategy designed to build an environment of gender equity in the field of GI so that all can thrive.
Institutions and social media handle
1. Santa Clara Valley Medical Center (San Jose, Calif), @noorannemd
2. Cedars Sinai (Los Angeles), @AdvaniRashmiMD
3. University of Cincinnati, @IBD_Afzali
4. Johns Hopkins Medicine (Washington), @DCharabaty
References
Advani R et al. Gender-specific attitudes of internal medicine residents toward gastroenterology. Dig Dis Sci. 2022 Nov;67(11):5044-52.
American Association of Medical Colleges. Diversity in Medicine: Facts and Figures (2019).
Elta GH. The challenges of being a female gastroenterologist. Gastroenterol Clin North Am. 2011 Jun;40(2):441-7.
Burke CA et al. Gender disparity in the practice of gastroenterology: The first 5 years of a career. Am J Gastroenterol 2005;100:259-64
David, Yakira N. et al. Gender-specific factors influencing gastroenterologists to pursue careers in advanced endoscopy: perceptions vs reality. Journal of the American College of Gastroenterology, ACG 116.3 (2021):539-50.
Rabinowitz LG et al. Gender dynamics in education and practice of gastroenterology. Gastrointest Endosc. 2021;93:1047-56.
Rabinowitz LG et al. Survey finds gender disparities impact both women mentors and mentees in gastroenterology. Journal of the American College of Gastroenterology, ACG 2021;116:1876-84.