User login
Use of non–vitamin K antagonist oral anticoagulants in the acute care, periprocedural settings
Non–vitamin K antagonist anticoagulants (NOACs, also called novel or direct oral anticoagulants) are commonly used to treat and prevent venous thromboembolism (VTE) and to prevent ischemic stroke in patients with nonvalvular atrial fibrillation. These agents, which include the factor Xa inhibitors rivaroxaban (Xarelto), apixaban (Eliquis), and edoxaban (Savaysa), and the competitive thrombin inhibitor dabigatran (Pradaxa), often are preferred over warfarin because of their more predictable pharmacokinetics, comparable efficacy, comparable or lower risk of major bleeding complications, fewer drug interactions, and lack of need for frequent monitoring. However, the acute care of patients taking NOACS can be challenging, because only dabigatran has an approved reversal agent, and none have readily available, reliable measurement assays. The American Heart Association (AHA) published a statement on the periprocedural and acute care management of patients taking NOACs. Here are the findings and recommendations of the AHA that are most relevant to primary care physicians.
Measurement
While all NOACs affect coagulation tests, their effect on prothrombin time and activated partial thromboplastin time is neither predictable nor an accurate reflection of the degree of anticoagulation. Instead, use the time of last drug ingestion and the patient’s creatinine clearance to estimate the anticoagulation effect. Dabigatran takes 1 hour to reach peak effect, or 2 hours if taken with food. Its half-life is 12-17 hours, on the higher end in the elderly and in those with moderate renal impairment. In those with severe renal impairment, half-life can be 28 hours. Rivaroxaban’s time to peak is 2-4 hours, and its half-life is 5-9 hours or up to 13 in the elderly. Apixaban’s time to peak is 3-4 hours and its half-life is about 12 hours. An antifactor Xa activity assay does provide a quantitative assessment of the factor Xa inhibitors.
Kidney injury
Acute kidney injury increases risk of bleeding while taking a NOAC. Monitor these patients closely and consider temporarily switching to a different anticoagulant in the setting of kidney injury.
Bleeding
Lack of reversibility is a common concern. Use 5 g of IV idarucizumab (Praxbind) to reverse dabigatran within minutes in a patient experiencing major bleeding. Hemodialysis, which removes about half of dabigatran in 4 hours, is a suitable option in acute kidney injury or in patients with a creatinine clearance under 30mL/min.
Options are more limited for the Xa inhibitors, because there are no available reversal agents and hemodialysis does not clear these highly protein-bound drugs. While data are limited, prothrombin complex concentrate may be given for patients on rivaroxaban, apixaban, or edoxaban who are experiencing an intracranial hemorrhage or other form of severe bleeding. Simply holding the NOAC is acceptable for minor bleeding.
Overdose
Activated charcoal to induce vomiting will work within 1-2 hours of drug ingestion.
Intracranial hemorrhage
Assume that a patient taking a NOAC who displays any acute neurologic change is experiencing an intracranial hemorrhage until proven otherwise. After CT confirmation, reverse dabigatran with idarucizumab, or give prothrombin complex concentrate to patients on other NOACs.
Ischemic stroke
Patients who suffer an ischemic stroke despite NOAC therapy are not candidates for tissue plasminogen activators.
The primary care physician is likely to be involved in the decision of whether, when, and for how long to resume anticoagulation therapy after a stroke. The statement says, “guidelines support withholding oral anticoagulation until 1-2 weeks after stroke among individuals with NVAF [nonvalvular atrial fibrillation], with shorter times for those with transient ischemic attack or small, nondisabling strokes and longer times for moderate to severe strokes.” In addition, it is worthwhile to consider medication nonadherence if no other etiology for the stroke is found; patients who miss doses may benefit more from warfarin because of its longer half-life.
Procedures and surgeries
Each year approximately 10% of patients on anticoagulation require surgery or other invasive procedures, and 20% require a minor procedure. To determine whether to interrupt NOAC therapy prior to a procedure, first determine the procedure’s bleeding risk. Patients undergoing procedures with low risk of bleeding, including minor dental, dermatologic, and ophthalmologic procedures, and endoscopies without biopsies, do not require interruption. For procedures with a moderate bleeding risk (including cardiac ablation, endoscopy with biopsies, radial artery catheterization) or high bleeding risk (including major surgery and cardiac catheterization via femoral artery), the patient’s thromboembolic risk should be evaluated using the medical history and the CHA2DS2 VASc score. NOACs should be stopped for 24-48 hours prior to the moderate to high-risk procedures. Dabigatran should be held for 72 hours for patients with creatinine clearance less than 50mL/min. Bridging therapy with heparin is not recommended for patients taking NOACS who are to have surgery. The decision about when to restart NOAC is based on the risk of thromboembolism and the bleeding risk of surgery.
Spinal or epidural anesthesia
Anesthesia guidelines recommend holding NOACs 3-5 days prior to the intervention, however, this increases risk of TE and studies have shown a very low incidence of hematoma in patients anticoagulated with a NOAC. For patients with a high risk of VTE, the NOAC can be resumed 12 hours post-procedure.
The bottom line
NOACS are commonly used for treatment and prophylaxis of VTE and atrial fibrillation and are often preferred over warfarin due to their more predictable pharmacokinetics, comparable efficacy, comparable or lower risk of major bleeding complications, fewer drug interactions, and lack of need for frequent monitoring. The AHA scientific statement gives guidance on managing NOACS in the face of acute bleeding as well as during and after procedures. NOACS should be stopped 24-48 hours prior to major surgeries and may be restarted based on weighing the risk of bleeding and risk of thromboembolism.
Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Thomas Jefferson University, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Jefferson Health. Dr. Oh is a third-year resident in the family medicine residency program at Abington Jefferson Health.
Reference
Raval AN et al. Management of patients on non–vitamin K antagonist oral anticoagulants in the acute care and periprocedural setting: A scientific statement from the American Heart Association. Circulation. 2017 Feb 6;135[10]:e604-e33. doi: 10.1161/CIR.0000000000000477
Non–vitamin K antagonist anticoagulants (NOACs, also called novel or direct oral anticoagulants) are commonly used to treat and prevent venous thromboembolism (VTE) and to prevent ischemic stroke in patients with nonvalvular atrial fibrillation. These agents, which include the factor Xa inhibitors rivaroxaban (Xarelto), apixaban (Eliquis), and edoxaban (Savaysa), and the competitive thrombin inhibitor dabigatran (Pradaxa), often are preferred over warfarin because of their more predictable pharmacokinetics, comparable efficacy, comparable or lower risk of major bleeding complications, fewer drug interactions, and lack of need for frequent monitoring. However, the acute care of patients taking NOACS can be challenging, because only dabigatran has an approved reversal agent, and none have readily available, reliable measurement assays. The American Heart Association (AHA) published a statement on the periprocedural and acute care management of patients taking NOACs. Here are the findings and recommendations of the AHA that are most relevant to primary care physicians.
Measurement
While all NOACs affect coagulation tests, their effect on prothrombin time and activated partial thromboplastin time is neither predictable nor an accurate reflection of the degree of anticoagulation. Instead, use the time of last drug ingestion and the patient’s creatinine clearance to estimate the anticoagulation effect. Dabigatran takes 1 hour to reach peak effect, or 2 hours if taken with food. Its half-life is 12-17 hours, on the higher end in the elderly and in those with moderate renal impairment. In those with severe renal impairment, half-life can be 28 hours. Rivaroxaban’s time to peak is 2-4 hours, and its half-life is 5-9 hours or up to 13 in the elderly. Apixaban’s time to peak is 3-4 hours and its half-life is about 12 hours. An antifactor Xa activity assay does provide a quantitative assessment of the factor Xa inhibitors.
Kidney injury
Acute kidney injury increases risk of bleeding while taking a NOAC. Monitor these patients closely and consider temporarily switching to a different anticoagulant in the setting of kidney injury.
Bleeding
Lack of reversibility is a common concern. Use 5 g of IV idarucizumab (Praxbind) to reverse dabigatran within minutes in a patient experiencing major bleeding. Hemodialysis, which removes about half of dabigatran in 4 hours, is a suitable option in acute kidney injury or in patients with a creatinine clearance under 30mL/min.
Options are more limited for the Xa inhibitors, because there are no available reversal agents and hemodialysis does not clear these highly protein-bound drugs. While data are limited, prothrombin complex concentrate may be given for patients on rivaroxaban, apixaban, or edoxaban who are experiencing an intracranial hemorrhage or other form of severe bleeding. Simply holding the NOAC is acceptable for minor bleeding.
Overdose
Activated charcoal to induce vomiting will work within 1-2 hours of drug ingestion.
Intracranial hemorrhage
Assume that a patient taking a NOAC who displays any acute neurologic change is experiencing an intracranial hemorrhage until proven otherwise. After CT confirmation, reverse dabigatran with idarucizumab, or give prothrombin complex concentrate to patients on other NOACs.
Ischemic stroke
Patients who suffer an ischemic stroke despite NOAC therapy are not candidates for tissue plasminogen activators.
The primary care physician is likely to be involved in the decision of whether, when, and for how long to resume anticoagulation therapy after a stroke. The statement says, “guidelines support withholding oral anticoagulation until 1-2 weeks after stroke among individuals with NVAF [nonvalvular atrial fibrillation], with shorter times for those with transient ischemic attack or small, nondisabling strokes and longer times for moderate to severe strokes.” In addition, it is worthwhile to consider medication nonadherence if no other etiology for the stroke is found; patients who miss doses may benefit more from warfarin because of its longer half-life.
Procedures and surgeries
Each year approximately 10% of patients on anticoagulation require surgery or other invasive procedures, and 20% require a minor procedure. To determine whether to interrupt NOAC therapy prior to a procedure, first determine the procedure’s bleeding risk. Patients undergoing procedures with low risk of bleeding, including minor dental, dermatologic, and ophthalmologic procedures, and endoscopies without biopsies, do not require interruption. For procedures with a moderate bleeding risk (including cardiac ablation, endoscopy with biopsies, radial artery catheterization) or high bleeding risk (including major surgery and cardiac catheterization via femoral artery), the patient’s thromboembolic risk should be evaluated using the medical history and the CHA2DS2 VASc score. NOACs should be stopped for 24-48 hours prior to the moderate to high-risk procedures. Dabigatran should be held for 72 hours for patients with creatinine clearance less than 50mL/min. Bridging therapy with heparin is not recommended for patients taking NOACS who are to have surgery. The decision about when to restart NOAC is based on the risk of thromboembolism and the bleeding risk of surgery.
Spinal or epidural anesthesia
Anesthesia guidelines recommend holding NOACs 3-5 days prior to the intervention, however, this increases risk of TE and studies have shown a very low incidence of hematoma in patients anticoagulated with a NOAC. For patients with a high risk of VTE, the NOAC can be resumed 12 hours post-procedure.
The bottom line
NOACS are commonly used for treatment and prophylaxis of VTE and atrial fibrillation and are often preferred over warfarin due to their more predictable pharmacokinetics, comparable efficacy, comparable or lower risk of major bleeding complications, fewer drug interactions, and lack of need for frequent monitoring. The AHA scientific statement gives guidance on managing NOACS in the face of acute bleeding as well as during and after procedures. NOACS should be stopped 24-48 hours prior to major surgeries and may be restarted based on weighing the risk of bleeding and risk of thromboembolism.
Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Thomas Jefferson University, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Jefferson Health. Dr. Oh is a third-year resident in the family medicine residency program at Abington Jefferson Health.
Reference
Raval AN et al. Management of patients on non–vitamin K antagonist oral anticoagulants in the acute care and periprocedural setting: A scientific statement from the American Heart Association. Circulation. 2017 Feb 6;135[10]:e604-e33. doi: 10.1161/CIR.0000000000000477
Non–vitamin K antagonist anticoagulants (NOACs, also called novel or direct oral anticoagulants) are commonly used to treat and prevent venous thromboembolism (VTE) and to prevent ischemic stroke in patients with nonvalvular atrial fibrillation. These agents, which include the factor Xa inhibitors rivaroxaban (Xarelto), apixaban (Eliquis), and edoxaban (Savaysa), and the competitive thrombin inhibitor dabigatran (Pradaxa), often are preferred over warfarin because of their more predictable pharmacokinetics, comparable efficacy, comparable or lower risk of major bleeding complications, fewer drug interactions, and lack of need for frequent monitoring. However, the acute care of patients taking NOACS can be challenging, because only dabigatran has an approved reversal agent, and none have readily available, reliable measurement assays. The American Heart Association (AHA) published a statement on the periprocedural and acute care management of patients taking NOACs. Here are the findings and recommendations of the AHA that are most relevant to primary care physicians.
Measurement
While all NOACs affect coagulation tests, their effect on prothrombin time and activated partial thromboplastin time is neither predictable nor an accurate reflection of the degree of anticoagulation. Instead, use the time of last drug ingestion and the patient’s creatinine clearance to estimate the anticoagulation effect. Dabigatran takes 1 hour to reach peak effect, or 2 hours if taken with food. Its half-life is 12-17 hours, on the higher end in the elderly and in those with moderate renal impairment. In those with severe renal impairment, half-life can be 28 hours. Rivaroxaban’s time to peak is 2-4 hours, and its half-life is 5-9 hours or up to 13 in the elderly. Apixaban’s time to peak is 3-4 hours and its half-life is about 12 hours. An antifactor Xa activity assay does provide a quantitative assessment of the factor Xa inhibitors.
Kidney injury
Acute kidney injury increases risk of bleeding while taking a NOAC. Monitor these patients closely and consider temporarily switching to a different anticoagulant in the setting of kidney injury.
Bleeding
Lack of reversibility is a common concern. Use 5 g of IV idarucizumab (Praxbind) to reverse dabigatran within minutes in a patient experiencing major bleeding. Hemodialysis, which removes about half of dabigatran in 4 hours, is a suitable option in acute kidney injury or in patients with a creatinine clearance under 30mL/min.
Options are more limited for the Xa inhibitors, because there are no available reversal agents and hemodialysis does not clear these highly protein-bound drugs. While data are limited, prothrombin complex concentrate may be given for patients on rivaroxaban, apixaban, or edoxaban who are experiencing an intracranial hemorrhage or other form of severe bleeding. Simply holding the NOAC is acceptable for minor bleeding.
Overdose
Activated charcoal to induce vomiting will work within 1-2 hours of drug ingestion.
Intracranial hemorrhage
Assume that a patient taking a NOAC who displays any acute neurologic change is experiencing an intracranial hemorrhage until proven otherwise. After CT confirmation, reverse dabigatran with idarucizumab, or give prothrombin complex concentrate to patients on other NOACs.
Ischemic stroke
Patients who suffer an ischemic stroke despite NOAC therapy are not candidates for tissue plasminogen activators.
The primary care physician is likely to be involved in the decision of whether, when, and for how long to resume anticoagulation therapy after a stroke. The statement says, “guidelines support withholding oral anticoagulation until 1-2 weeks after stroke among individuals with NVAF [nonvalvular atrial fibrillation], with shorter times for those with transient ischemic attack or small, nondisabling strokes and longer times for moderate to severe strokes.” In addition, it is worthwhile to consider medication nonadherence if no other etiology for the stroke is found; patients who miss doses may benefit more from warfarin because of its longer half-life.
Procedures and surgeries
Each year approximately 10% of patients on anticoagulation require surgery or other invasive procedures, and 20% require a minor procedure. To determine whether to interrupt NOAC therapy prior to a procedure, first determine the procedure’s bleeding risk. Patients undergoing procedures with low risk of bleeding, including minor dental, dermatologic, and ophthalmologic procedures, and endoscopies without biopsies, do not require interruption. For procedures with a moderate bleeding risk (including cardiac ablation, endoscopy with biopsies, radial artery catheterization) or high bleeding risk (including major surgery and cardiac catheterization via femoral artery), the patient’s thromboembolic risk should be evaluated using the medical history and the CHA2DS2 VASc score. NOACs should be stopped for 24-48 hours prior to the moderate to high-risk procedures. Dabigatran should be held for 72 hours for patients with creatinine clearance less than 50mL/min. Bridging therapy with heparin is not recommended for patients taking NOACS who are to have surgery. The decision about when to restart NOAC is based on the risk of thromboembolism and the bleeding risk of surgery.
Spinal or epidural anesthesia
Anesthesia guidelines recommend holding NOACs 3-5 days prior to the intervention, however, this increases risk of TE and studies have shown a very low incidence of hematoma in patients anticoagulated with a NOAC. For patients with a high risk of VTE, the NOAC can be resumed 12 hours post-procedure.
The bottom line
NOACS are commonly used for treatment and prophylaxis of VTE and atrial fibrillation and are often preferred over warfarin due to their more predictable pharmacokinetics, comparable efficacy, comparable or lower risk of major bleeding complications, fewer drug interactions, and lack of need for frequent monitoring. The AHA scientific statement gives guidance on managing NOACS in the face of acute bleeding as well as during and after procedures. NOACS should be stopped 24-48 hours prior to major surgeries and may be restarted based on weighing the risk of bleeding and risk of thromboembolism.
Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Thomas Jefferson University, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Jefferson Health. Dr. Oh is a third-year resident in the family medicine residency program at Abington Jefferson Health.
Reference
Raval AN et al. Management of patients on non–vitamin K antagonist oral anticoagulants in the acute care and periprocedural setting: A scientific statement from the American Heart Association. Circulation. 2017 Feb 6;135[10]:e604-e33. doi: 10.1161/CIR.0000000000000477
Our fascination with medication compliance
As a forensic psychiatrist, I follow news relating to mental illness and crime. Like many of the judges and lawyers with whom I work, the media appear to have an obsession with medication compliance in those with mental illness. Being “off medications” has become a threatening term suggestive of unbridled impulsivity and violence – a term that can explain any behavior, implying that without medication, humans are routinely capable of all things without warning.
The year 2018 already has provided two stark examples of this phenomenon. During the first week of the year, two articles with the following headlines were published: “ ‘He was off his meds’: Son charged in mother’s murder”1 and “A man lost his life in the subway after telling a teen off of his medication to ‘get away.’ ”2 Those two articles describe awful events that, as the headlines suggest, are best explained by a lack of compliance with a psychotropic medication regimen.
“A man lost his life in the subway after telling a teen off of his medication to ‘get away’ ” reports on a 65-year-old man who was pushed onto New York’s subway tracks after interacting with an 18-year-old who “did not take medication that day for his mental illness.” This article provides some limited details on the state of mind of the defendant, indicating that he had been “talking to himself.” However, to reinforce the message, the article informs the reader that he had been prescribed three psychotropics – insinuating a multiplier effect for the role of noncompliance. Furthermore, the article implies that missing a single day of psychotropics is an explanation for the incident.
The media routinely use this bias in favor of the medication explanation in its analysis – or lack thereof – of violent behavior in people with mental illness. Recent stories include a man killing his nephew3 and a man killing his girlfriend4, and both were incidents apparently best explained by medication noncompliance. Another story reports of police officers who were charged with assault after an altercation with a patient with mental illness led to the patient’s death. In the latter case, the article suggests that the simple fact that the victim had been acting erratically warrants the comment that he was “likely was off his medications.”
My work in the jail system also has been tainted by this overreliance on the unquestioned dogma of medication compliance. Discussions pertaining to punishment and privileges of inmates with mental illness often would lead to the question: “Is he taking his meds?” I have witnessed countless times when crucial decisions about placement in solitary confinement were predicated on questions of medication compliance. My answer was always the same: “Why does it matter? If the inmate is following the rules and behaving respectfully, how does taking a pill provide more important information?”
What was once thought to be a predictor of relapse risk, despite limited evidence, has become the outcome itself. As a society, we have falsely equated mental illness with violence; we have furthermore falsely equated remission and safety with medication compliance. The consequences of those beliefs are severe as we have limited attention, and our focus on medications blinds us to much clearer risk factors. 5
The court system is equally riveted with this question. Judges and lawyers associate medication compliance with legal competency, safety in probation or parole, and general well-being. I have witnessed agitated patients being reprimanded for their lack of medication adherence, leaving me to remind lawyers that the patient has been compliant. Conversely, patients are congratulated for their medication adherence when appearing well, until I remind the lawyers that the patient missed his last two visits for long-acting injectables.
As our field is reconciling new evidence questioning the long-term role of antipsychotics in schizophrenia, I am questioning whether society has accepted their value as a foregone conclusion. Lex Wunderink, MD, PhD, and his associates challenged accepted dogma when conducting a long-term, randomized trial of antipsychotics, in which patients on a dose reduction and discontinuation arm did better at 7 years than the patients on the continuation arm.6 The then National Institute of Mental Health director, Thomas Insel, MD, wrote in his blog that for some schizophrenia patients, “remaining on medication long term might impede a full return to wellness.”7
A Cochrane review of the literature found that, over time, antipsychotics had a diminishing effect on relapse prevention. After 2 years, the effect approached zero.8 In 2016, Nancy L. Sohler, PhD, and her colleagues looked at the literature on antipsychotic use in longer trials. The data were of poor quality and inconclusive.9 However, stories in the popular press suggest that the best explanation for violent behavior in the mentally ill population is medication noncompliance.
Inextricably bound up with this dogma regarding noncompliance is the incorporation of the pharmaceutical industry into mainstream psychiatry. The promise of psychopharmacology to treat mental illness was adopted in a wholesale manner for financial and practical reasons as well as a desperate optimism to relieve seemingly intractable problems. Sadly, the failure of psychopharmacology to produce on said promises has not produced a backlash. Instead, there is a doubling down on this belief, which can be seen, for example, in the creation of Abilify MyCite – with its promise to keep clinicians informed about their patients’ medication compliance.10 The alternative to this prescribing culture would be an attentive reckoning of the ongoing limitations inherent in the treatment of those with mental illness. If noncompliance cannot explain violence in people with mental illness, we are left with the same complex and subtle issues surrounding violence that frustrate easy journalistic explanations, and relatively cheap and easy interventions for the care of this population. It feels better and is more cost effective to blame the patients for not fitting our biological models by being drug nonresponders or noncompliant.
Blaming pills is facile. It is tangible and easier to measure than looking into someone’s mind. Psychiatrists have promoted this idea by teaching the public about chemical imbalances and by focusing on medication management. However, when the consequences are as severe as placing people in solitary confinement or explaining murder, the evidence needs to be equally solid as the severity of the punishment. This must start with psychiatrists reeducating the public on the role, the power, and the limitations of psychotropics.
Dr. Badre is a forensic psychiatrist in San Diego and an expert in correctional mental health. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Dr. Badre mentors residents on projects, including reduction in the use of solitary confinement of patients with mental illness and examination of the mentally ill offender. Dr. Badre can be reached at Badremd.com.
References
1. Worcester Patch. Jan. 2, 2018.
2. Rare News. Jan. 4, 2018.
3. WTSP.com. Dec. 31, 2017.
4. Wavy.com, Dec. 20, 2017.
5. U.S. Attorney General. 2016. U.S. Department of Justice Report and Recommendations Concerning the Use of Restrictive Housing.
6. JAMA Psychiatry. 2013 Sep;70(9):913-20.
7. Blogpost, by Thomas Insel, MD. Aug. 28, 2013.
8. Cochrane Database Syst Rev. 2012 May 16. doi: 10.1002/14651858.CD008016.pub2.
9. Am J Orthopsychiatry. 2016;86(5):477-85.
10. New York Times. Nov. 13, 2017.
As a forensic psychiatrist, I follow news relating to mental illness and crime. Like many of the judges and lawyers with whom I work, the media appear to have an obsession with medication compliance in those with mental illness. Being “off medications” has become a threatening term suggestive of unbridled impulsivity and violence – a term that can explain any behavior, implying that without medication, humans are routinely capable of all things without warning.
The year 2018 already has provided two stark examples of this phenomenon. During the first week of the year, two articles with the following headlines were published: “ ‘He was off his meds’: Son charged in mother’s murder”1 and “A man lost his life in the subway after telling a teen off of his medication to ‘get away.’ ”2 Those two articles describe awful events that, as the headlines suggest, are best explained by a lack of compliance with a psychotropic medication regimen.
“A man lost his life in the subway after telling a teen off of his medication to ‘get away’ ” reports on a 65-year-old man who was pushed onto New York’s subway tracks after interacting with an 18-year-old who “did not take medication that day for his mental illness.” This article provides some limited details on the state of mind of the defendant, indicating that he had been “talking to himself.” However, to reinforce the message, the article informs the reader that he had been prescribed three psychotropics – insinuating a multiplier effect for the role of noncompliance. Furthermore, the article implies that missing a single day of psychotropics is an explanation for the incident.
The media routinely use this bias in favor of the medication explanation in its analysis – or lack thereof – of violent behavior in people with mental illness. Recent stories include a man killing his nephew3 and a man killing his girlfriend4, and both were incidents apparently best explained by medication noncompliance. Another story reports of police officers who were charged with assault after an altercation with a patient with mental illness led to the patient’s death. In the latter case, the article suggests that the simple fact that the victim had been acting erratically warrants the comment that he was “likely was off his medications.”
My work in the jail system also has been tainted by this overreliance on the unquestioned dogma of medication compliance. Discussions pertaining to punishment and privileges of inmates with mental illness often would lead to the question: “Is he taking his meds?” I have witnessed countless times when crucial decisions about placement in solitary confinement were predicated on questions of medication compliance. My answer was always the same: “Why does it matter? If the inmate is following the rules and behaving respectfully, how does taking a pill provide more important information?”
What was once thought to be a predictor of relapse risk, despite limited evidence, has become the outcome itself. As a society, we have falsely equated mental illness with violence; we have furthermore falsely equated remission and safety with medication compliance. The consequences of those beliefs are severe as we have limited attention, and our focus on medications blinds us to much clearer risk factors. 5
The court system is equally riveted with this question. Judges and lawyers associate medication compliance with legal competency, safety in probation or parole, and general well-being. I have witnessed agitated patients being reprimanded for their lack of medication adherence, leaving me to remind lawyers that the patient has been compliant. Conversely, patients are congratulated for their medication adherence when appearing well, until I remind the lawyers that the patient missed his last two visits for long-acting injectables.
As our field is reconciling new evidence questioning the long-term role of antipsychotics in schizophrenia, I am questioning whether society has accepted their value as a foregone conclusion. Lex Wunderink, MD, PhD, and his associates challenged accepted dogma when conducting a long-term, randomized trial of antipsychotics, in which patients on a dose reduction and discontinuation arm did better at 7 years than the patients on the continuation arm.6 The then National Institute of Mental Health director, Thomas Insel, MD, wrote in his blog that for some schizophrenia patients, “remaining on medication long term might impede a full return to wellness.”7
A Cochrane review of the literature found that, over time, antipsychotics had a diminishing effect on relapse prevention. After 2 years, the effect approached zero.8 In 2016, Nancy L. Sohler, PhD, and her colleagues looked at the literature on antipsychotic use in longer trials. The data were of poor quality and inconclusive.9 However, stories in the popular press suggest that the best explanation for violent behavior in the mentally ill population is medication noncompliance.
Inextricably bound up with this dogma regarding noncompliance is the incorporation of the pharmaceutical industry into mainstream psychiatry. The promise of psychopharmacology to treat mental illness was adopted in a wholesale manner for financial and practical reasons as well as a desperate optimism to relieve seemingly intractable problems. Sadly, the failure of psychopharmacology to produce on said promises has not produced a backlash. Instead, there is a doubling down on this belief, which can be seen, for example, in the creation of Abilify MyCite – with its promise to keep clinicians informed about their patients’ medication compliance.10 The alternative to this prescribing culture would be an attentive reckoning of the ongoing limitations inherent in the treatment of those with mental illness. If noncompliance cannot explain violence in people with mental illness, we are left with the same complex and subtle issues surrounding violence that frustrate easy journalistic explanations, and relatively cheap and easy interventions for the care of this population. It feels better and is more cost effective to blame the patients for not fitting our biological models by being drug nonresponders or noncompliant.
Blaming pills is facile. It is tangible and easier to measure than looking into someone’s mind. Psychiatrists have promoted this idea by teaching the public about chemical imbalances and by focusing on medication management. However, when the consequences are as severe as placing people in solitary confinement or explaining murder, the evidence needs to be equally solid as the severity of the punishment. This must start with psychiatrists reeducating the public on the role, the power, and the limitations of psychotropics.
Dr. Badre is a forensic psychiatrist in San Diego and an expert in correctional mental health. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Dr. Badre mentors residents on projects, including reduction in the use of solitary confinement of patients with mental illness and examination of the mentally ill offender. Dr. Badre can be reached at Badremd.com.
References
1. Worcester Patch. Jan. 2, 2018.
2. Rare News. Jan. 4, 2018.
3. WTSP.com. Dec. 31, 2017.
4. Wavy.com, Dec. 20, 2017.
5. U.S. Attorney General. 2016. U.S. Department of Justice Report and Recommendations Concerning the Use of Restrictive Housing.
6. JAMA Psychiatry. 2013 Sep;70(9):913-20.
7. Blogpost, by Thomas Insel, MD. Aug. 28, 2013.
8. Cochrane Database Syst Rev. 2012 May 16. doi: 10.1002/14651858.CD008016.pub2.
9. Am J Orthopsychiatry. 2016;86(5):477-85.
10. New York Times. Nov. 13, 2017.
As a forensic psychiatrist, I follow news relating to mental illness and crime. Like many of the judges and lawyers with whom I work, the media appear to have an obsession with medication compliance in those with mental illness. Being “off medications” has become a threatening term suggestive of unbridled impulsivity and violence – a term that can explain any behavior, implying that without medication, humans are routinely capable of all things without warning.
The year 2018 already has provided two stark examples of this phenomenon. During the first week of the year, two articles with the following headlines were published: “ ‘He was off his meds’: Son charged in mother’s murder”1 and “A man lost his life in the subway after telling a teen off of his medication to ‘get away.’ ”2 Those two articles describe awful events that, as the headlines suggest, are best explained by a lack of compliance with a psychotropic medication regimen.
“A man lost his life in the subway after telling a teen off of his medication to ‘get away’ ” reports on a 65-year-old man who was pushed onto New York’s subway tracks after interacting with an 18-year-old who “did not take medication that day for his mental illness.” This article provides some limited details on the state of mind of the defendant, indicating that he had been “talking to himself.” However, to reinforce the message, the article informs the reader that he had been prescribed three psychotropics – insinuating a multiplier effect for the role of noncompliance. Furthermore, the article implies that missing a single day of psychotropics is an explanation for the incident.
The media routinely use this bias in favor of the medication explanation in its analysis – or lack thereof – of violent behavior in people with mental illness. Recent stories include a man killing his nephew3 and a man killing his girlfriend4, and both were incidents apparently best explained by medication noncompliance. Another story reports of police officers who were charged with assault after an altercation with a patient with mental illness led to the patient’s death. In the latter case, the article suggests that the simple fact that the victim had been acting erratically warrants the comment that he was “likely was off his medications.”
My work in the jail system also has been tainted by this overreliance on the unquestioned dogma of medication compliance. Discussions pertaining to punishment and privileges of inmates with mental illness often would lead to the question: “Is he taking his meds?” I have witnessed countless times when crucial decisions about placement in solitary confinement were predicated on questions of medication compliance. My answer was always the same: “Why does it matter? If the inmate is following the rules and behaving respectfully, how does taking a pill provide more important information?”
What was once thought to be a predictor of relapse risk, despite limited evidence, has become the outcome itself. As a society, we have falsely equated mental illness with violence; we have furthermore falsely equated remission and safety with medication compliance. The consequences of those beliefs are severe as we have limited attention, and our focus on medications blinds us to much clearer risk factors. 5
The court system is equally riveted with this question. Judges and lawyers associate medication compliance with legal competency, safety in probation or parole, and general well-being. I have witnessed agitated patients being reprimanded for their lack of medication adherence, leaving me to remind lawyers that the patient has been compliant. Conversely, patients are congratulated for their medication adherence when appearing well, until I remind the lawyers that the patient missed his last two visits for long-acting injectables.
As our field is reconciling new evidence questioning the long-term role of antipsychotics in schizophrenia, I am questioning whether society has accepted their value as a foregone conclusion. Lex Wunderink, MD, PhD, and his associates challenged accepted dogma when conducting a long-term, randomized trial of antipsychotics, in which patients on a dose reduction and discontinuation arm did better at 7 years than the patients on the continuation arm.6 The then National Institute of Mental Health director, Thomas Insel, MD, wrote in his blog that for some schizophrenia patients, “remaining on medication long term might impede a full return to wellness.”7
A Cochrane review of the literature found that, over time, antipsychotics had a diminishing effect on relapse prevention. After 2 years, the effect approached zero.8 In 2016, Nancy L. Sohler, PhD, and her colleagues looked at the literature on antipsychotic use in longer trials. The data were of poor quality and inconclusive.9 However, stories in the popular press suggest that the best explanation for violent behavior in the mentally ill population is medication noncompliance.
Inextricably bound up with this dogma regarding noncompliance is the incorporation of the pharmaceutical industry into mainstream psychiatry. The promise of psychopharmacology to treat mental illness was adopted in a wholesale manner for financial and practical reasons as well as a desperate optimism to relieve seemingly intractable problems. Sadly, the failure of psychopharmacology to produce on said promises has not produced a backlash. Instead, there is a doubling down on this belief, which can be seen, for example, in the creation of Abilify MyCite – with its promise to keep clinicians informed about their patients’ medication compliance.10 The alternative to this prescribing culture would be an attentive reckoning of the ongoing limitations inherent in the treatment of those with mental illness. If noncompliance cannot explain violence in people with mental illness, we are left with the same complex and subtle issues surrounding violence that frustrate easy journalistic explanations, and relatively cheap and easy interventions for the care of this population. It feels better and is more cost effective to blame the patients for not fitting our biological models by being drug nonresponders or noncompliant.
Blaming pills is facile. It is tangible and easier to measure than looking into someone’s mind. Psychiatrists have promoted this idea by teaching the public about chemical imbalances and by focusing on medication management. However, when the consequences are as severe as placing people in solitary confinement or explaining murder, the evidence needs to be equally solid as the severity of the punishment. This must start with psychiatrists reeducating the public on the role, the power, and the limitations of psychotropics.
Dr. Badre is a forensic psychiatrist in San Diego and an expert in correctional mental health. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Dr. Badre mentors residents on projects, including reduction in the use of solitary confinement of patients with mental illness and examination of the mentally ill offender. Dr. Badre can be reached at Badremd.com.
References
1. Worcester Patch. Jan. 2, 2018.
2. Rare News. Jan. 4, 2018.
3. WTSP.com. Dec. 31, 2017.
4. Wavy.com, Dec. 20, 2017.
5. U.S. Attorney General. 2016. U.S. Department of Justice Report and Recommendations Concerning the Use of Restrictive Housing.
6. JAMA Psychiatry. 2013 Sep;70(9):913-20.
7. Blogpost, by Thomas Insel, MD. Aug. 28, 2013.
8. Cochrane Database Syst Rev. 2012 May 16. doi: 10.1002/14651858.CD008016.pub2.
9. Am J Orthopsychiatry. 2016;86(5):477-85.
10. New York Times. Nov. 13, 2017.
From the Editors: Finding joy
While basking in the fading glow of the holidays, I have been reflecting on the dynamic of combined with the negativity local and world events engender that can push us toward burnout.
The holiday season is a time when people tend to engage in activities that have been shown to improve mood and outlook: connecting with old friends, sharing memories of a happier time, spending time with children and grandchildren, sitting around a warm fire, enjoying the sights and smells of bright decorations and fragrant candles, attending traditional holiday plays, concerts, and ballets. The holiday traditions, no matter what your ethnic or religious background, create community and warm feelings. The workweek may be shortened, people take a little time out, smile a bit more. For many of us, these activities can be a tonic.
At the same time, the days become shorter, colder, and grayer in most of North America. The normal frustrations of our day-to-day professional lives may seem more profound during the winter, and some experience SAD (seasonal affective disorder) or even burnout.
If we are not affected by these events, we may be missing the compassion gene. But I would suggest that an acute awareness of a world of trouble around us compounded with our own heavy load as surgeons is a recipe for burnout. It may not be within the capacity of any of us to alter the reality of our present world, and the surgical profession is not going to become a low-key occupation any time soon. But we can control our response to all this and take steps to attend our own emotional health.
I have found that the single most effective measure to combat negative feelings is to connect with colleagues, friends, and family to share positive, enjoyable experiences: a potluck dinner, a concert, a hike (or snowshoe trip) in the woods. We should seek out optimistic, glass-half-full individuals. We all have some of these folks in our lives and they do us a world of good.
With regard to professional stressors, reaching out to colleagues to work together in identifying remedies for a dysfunctional workplace may not only address the problem, but also allow you to recognize that you are not alone in your distress. Joining forces as a team to forge a solution can be satisfying and empowering.
Nevertheless, surgical practice remains intense, stressful, and demanding. As surgeons, we tend to be perfectionists, wanting to dot every “i” and cross every “t,” no matter how trivial. It is critical to set realistic expectations for how much you can achieve. Identify and prioritize personal and professional goals, make the most important goals take front and center, and delegate (or just allow to disappear) items that are less important. This may be the single most important strategy to avoid burnout: Prioritize what is essential and let the rest go.
A great deal has been written recently about resilience and mindfulness – facile concepts that don’t address the struggles of individuals feeling helpless and overwhelmed by the onslaught of demands on his/her time. Even though clichés about mindfulness can ring hollow, I have found that taking small steps to build my own inner reserves can help.
Here is my advice: Take a moment several times a day to appreciate something beautiful around you: a textured sky, a peaceful field, city lights, a nearby river with the ripples of wind on the water. Smile and greet someone on the street or in the hallway at work. Say a good word to someone on a job nicely done. Reflect on how doing these things affect you. Do they make you feel calmer and happier? “Rest your brain” every 2 hours for just a minute or two; cognitive fatigue occurs after 60-90 minutes and drains your energy if the “pause button” isn’t pushed.
Many of us neglect our personal health. It goes without saying that we are all far more likely to avoid burnout if we have a balanced diet, adequate sleep, and some exercise. We should all have a primary care provider for regular checkups and preventive exams. We speak with great authority when we counsel our patients to do this, so what possible excuse do we have for neglecting our own health?
One of the most important habits that I cultivate to improve my own mood is to end each day reflecting on three positive things that happened that day. Amid all of the calamities that occur every day in the world, it should not be difficult for those of us who live a life of relative privilege and plenty to find positive things in our lives. A strong association has been demonstrated between a sense of thankfulness and individual happiness and contentment. As surgeons, we have a ready source of positive reinforcers – the gratitude of our patients. I have a “feel good drawer” for “thank yous.” I open that drawer and read some of those messages from grateful patients. Reflecting on how we have been able to help our patients can do us all good when we are having doubts about our professional lives.
I want to encourage all surgeons to take a little better care of themselves this year. Take some specific steps to attend to your physical and emotional health. Do some activities the only purpose of which is to rest, to reflect, and to find joy.
Dr. Deveney is professor of surgery and vice chair of education in the department of surgery, Oregon Health & Science University, Portland. She is the coeditor of ACS Surgery News.
While basking in the fading glow of the holidays, I have been reflecting on the dynamic of combined with the negativity local and world events engender that can push us toward burnout.
The holiday season is a time when people tend to engage in activities that have been shown to improve mood and outlook: connecting with old friends, sharing memories of a happier time, spending time with children and grandchildren, sitting around a warm fire, enjoying the sights and smells of bright decorations and fragrant candles, attending traditional holiday plays, concerts, and ballets. The holiday traditions, no matter what your ethnic or religious background, create community and warm feelings. The workweek may be shortened, people take a little time out, smile a bit more. For many of us, these activities can be a tonic.
At the same time, the days become shorter, colder, and grayer in most of North America. The normal frustrations of our day-to-day professional lives may seem more profound during the winter, and some experience SAD (seasonal affective disorder) or even burnout.
If we are not affected by these events, we may be missing the compassion gene. But I would suggest that an acute awareness of a world of trouble around us compounded with our own heavy load as surgeons is a recipe for burnout. It may not be within the capacity of any of us to alter the reality of our present world, and the surgical profession is not going to become a low-key occupation any time soon. But we can control our response to all this and take steps to attend our own emotional health.
I have found that the single most effective measure to combat negative feelings is to connect with colleagues, friends, and family to share positive, enjoyable experiences: a potluck dinner, a concert, a hike (or snowshoe trip) in the woods. We should seek out optimistic, glass-half-full individuals. We all have some of these folks in our lives and they do us a world of good.
With regard to professional stressors, reaching out to colleagues to work together in identifying remedies for a dysfunctional workplace may not only address the problem, but also allow you to recognize that you are not alone in your distress. Joining forces as a team to forge a solution can be satisfying and empowering.
Nevertheless, surgical practice remains intense, stressful, and demanding. As surgeons, we tend to be perfectionists, wanting to dot every “i” and cross every “t,” no matter how trivial. It is critical to set realistic expectations for how much you can achieve. Identify and prioritize personal and professional goals, make the most important goals take front and center, and delegate (or just allow to disappear) items that are less important. This may be the single most important strategy to avoid burnout: Prioritize what is essential and let the rest go.
A great deal has been written recently about resilience and mindfulness – facile concepts that don’t address the struggles of individuals feeling helpless and overwhelmed by the onslaught of demands on his/her time. Even though clichés about mindfulness can ring hollow, I have found that taking small steps to build my own inner reserves can help.
Here is my advice: Take a moment several times a day to appreciate something beautiful around you: a textured sky, a peaceful field, city lights, a nearby river with the ripples of wind on the water. Smile and greet someone on the street or in the hallway at work. Say a good word to someone on a job nicely done. Reflect on how doing these things affect you. Do they make you feel calmer and happier? “Rest your brain” every 2 hours for just a minute or two; cognitive fatigue occurs after 60-90 minutes and drains your energy if the “pause button” isn’t pushed.
Many of us neglect our personal health. It goes without saying that we are all far more likely to avoid burnout if we have a balanced diet, adequate sleep, and some exercise. We should all have a primary care provider for regular checkups and preventive exams. We speak with great authority when we counsel our patients to do this, so what possible excuse do we have for neglecting our own health?
One of the most important habits that I cultivate to improve my own mood is to end each day reflecting on three positive things that happened that day. Amid all of the calamities that occur every day in the world, it should not be difficult for those of us who live a life of relative privilege and plenty to find positive things in our lives. A strong association has been demonstrated between a sense of thankfulness and individual happiness and contentment. As surgeons, we have a ready source of positive reinforcers – the gratitude of our patients. I have a “feel good drawer” for “thank yous.” I open that drawer and read some of those messages from grateful patients. Reflecting on how we have been able to help our patients can do us all good when we are having doubts about our professional lives.
I want to encourage all surgeons to take a little better care of themselves this year. Take some specific steps to attend to your physical and emotional health. Do some activities the only purpose of which is to rest, to reflect, and to find joy.
Dr. Deveney is professor of surgery and vice chair of education in the department of surgery, Oregon Health & Science University, Portland. She is the coeditor of ACS Surgery News.
While basking in the fading glow of the holidays, I have been reflecting on the dynamic of combined with the negativity local and world events engender that can push us toward burnout.
The holiday season is a time when people tend to engage in activities that have been shown to improve mood and outlook: connecting with old friends, sharing memories of a happier time, spending time with children and grandchildren, sitting around a warm fire, enjoying the sights and smells of bright decorations and fragrant candles, attending traditional holiday plays, concerts, and ballets. The holiday traditions, no matter what your ethnic or religious background, create community and warm feelings. The workweek may be shortened, people take a little time out, smile a bit more. For many of us, these activities can be a tonic.
At the same time, the days become shorter, colder, and grayer in most of North America. The normal frustrations of our day-to-day professional lives may seem more profound during the winter, and some experience SAD (seasonal affective disorder) or even burnout.
If we are not affected by these events, we may be missing the compassion gene. But I would suggest that an acute awareness of a world of trouble around us compounded with our own heavy load as surgeons is a recipe for burnout. It may not be within the capacity of any of us to alter the reality of our present world, and the surgical profession is not going to become a low-key occupation any time soon. But we can control our response to all this and take steps to attend our own emotional health.
I have found that the single most effective measure to combat negative feelings is to connect with colleagues, friends, and family to share positive, enjoyable experiences: a potluck dinner, a concert, a hike (or snowshoe trip) in the woods. We should seek out optimistic, glass-half-full individuals. We all have some of these folks in our lives and they do us a world of good.
With regard to professional stressors, reaching out to colleagues to work together in identifying remedies for a dysfunctional workplace may not only address the problem, but also allow you to recognize that you are not alone in your distress. Joining forces as a team to forge a solution can be satisfying and empowering.
Nevertheless, surgical practice remains intense, stressful, and demanding. As surgeons, we tend to be perfectionists, wanting to dot every “i” and cross every “t,” no matter how trivial. It is critical to set realistic expectations for how much you can achieve. Identify and prioritize personal and professional goals, make the most important goals take front and center, and delegate (or just allow to disappear) items that are less important. This may be the single most important strategy to avoid burnout: Prioritize what is essential and let the rest go.
A great deal has been written recently about resilience and mindfulness – facile concepts that don’t address the struggles of individuals feeling helpless and overwhelmed by the onslaught of demands on his/her time. Even though clichés about mindfulness can ring hollow, I have found that taking small steps to build my own inner reserves can help.
Here is my advice: Take a moment several times a day to appreciate something beautiful around you: a textured sky, a peaceful field, city lights, a nearby river with the ripples of wind on the water. Smile and greet someone on the street or in the hallway at work. Say a good word to someone on a job nicely done. Reflect on how doing these things affect you. Do they make you feel calmer and happier? “Rest your brain” every 2 hours for just a minute or two; cognitive fatigue occurs after 60-90 minutes and drains your energy if the “pause button” isn’t pushed.
Many of us neglect our personal health. It goes without saying that we are all far more likely to avoid burnout if we have a balanced diet, adequate sleep, and some exercise. We should all have a primary care provider for regular checkups and preventive exams. We speak with great authority when we counsel our patients to do this, so what possible excuse do we have for neglecting our own health?
One of the most important habits that I cultivate to improve my own mood is to end each day reflecting on three positive things that happened that day. Amid all of the calamities that occur every day in the world, it should not be difficult for those of us who live a life of relative privilege and plenty to find positive things in our lives. A strong association has been demonstrated between a sense of thankfulness and individual happiness and contentment. As surgeons, we have a ready source of positive reinforcers – the gratitude of our patients. I have a “feel good drawer” for “thank yous.” I open that drawer and read some of those messages from grateful patients. Reflecting on how we have been able to help our patients can do us all good when we are having doubts about our professional lives.
I want to encourage all surgeons to take a little better care of themselves this year. Take some specific steps to attend to your physical and emotional health. Do some activities the only purpose of which is to rest, to reflect, and to find joy.
Dr. Deveney is professor of surgery and vice chair of education in the department of surgery, Oregon Health & Science University, Portland. She is the coeditor of ACS Surgery News.
A trainee’s path to fighting addiction
When I came to this country, even before my current residency, I launched my addiction psychiatry career by researching nicotine addiction in schizophrenia patients. Those early experiences gave me a greater understanding of the health concerns and life experiences of people with addictions – and those more likely to develop them.
So imagine my excitement when I first became acquainted with the American Academy of Addiction Psychiatry (AAAP). I first learned about the AAAP, its mission, and activities at the 2017 American Psychiatric Association’s annual meeting in San Diego.
Getting in and involved
After returning to New York, I took those next steps and joined up, which opened the AAAP gates so I could receive its newsletters and submission calls, gain access to resources such as The American Journal on Addictions, survey the various joinable task forces, as well as discover who might be available to me as a mentor as part of the AAAP’s mentor-mentee program.
Sometime during my third-year residency training, I received a member-email advertising the AAAP 28th Annual Meeting and Scientific Symposium, and soon after that, received another email calling for research submissions to be presented there, as well as an invitation to apply for a trainee travel scholarship that would defray the cost for and allow its fellows to attend the meeting in San Diego. That alone was enticing enough to apply. But even more enticing was the opportunity to showcase the addiction work I had been doing during my residency, as well as to meet other members at various levels of the AAAP to determine whether I wanted to become more involved.
Pursuing experiences
I did not think twice about applying for the poster presentation and the travel scholarship. The AAAP’s online application forms for both were easy to understand and very well structured, which greatly helped me with filling out and formatting my applications. Taking the initiative toward even these first AAAP offerings brought more positive echoes. I was thrilled when the poster I proposed was accepted, mostly because it would give me the chance to present my recent addiction psych work from a higher platform. A few weeks later, I was thrilled again when I received an AAAP email congratulating me on being awarded the San Diego 28th annual meeting travel scholarship, which would waive the annual membership and conference registration fees, in addition to defraying my travel costs. Pacific breezes, here I come. And there I went. (Thanks to my extremely supportive training director, who first nominated me for the award.)
On the ground at 2017 AAAP
The 28th AAAP annual meeting opened on a balmy December Thursday, and that’s the day I arrived. I attended many addiction workshops and symposiums, which featured premier figures in addiction psychiatry. Of the numerous trainee-specific events I attended, the most informative was the “Fellowship Forum: Exploring the Field of Addiction Psychiatry.” At this forum, I learned the true benefits of doing an addiction psychiatry fellowship, while meeting many of the fellowship program directors of top institutions. Having them all under one “roof” made it easy to compare and contrast the specific training they offered.
Then came what were, for me, major highlights of the AAAP 2017. After I delivered my poster presentation and shared my research, I was able to receive very close, constructive feedback from the field’s most experienced professionals. And, finally, I met my AAAP mentors face to face: Dr. Amy Yule of Harvard Medical School, Boston; Dr. Thomas Penders, of East Carolina University, Greenville, N.C.; and Dr. Cornel Stanciu of Dartmouth-Hitchcock Medical Center, Lebanon, N.H.
One AAAP trainee’s takeaways
All AAAP trainees, fellows, presenters leave the meeting with their own conclusions, but my biggest takeaways were:
- Regarding barriers to buprenorphine, emerging research supports similar efficacy for long-acting injectable naltrexone.
- Various protocols for rapid implementation of naltrexone are being used, and these allow for smoother transition and shorter “washout” periods.
- We should not overlook the effects of tobacco use in our patient population – and should address it aggressively, regardless of psychiatric comorbidities.
- The cannabinoid CBD receptors that exist on the dopamine pathway strengthen and complicate their relationship with psychosis.
- , especially in rural and remote settings. The body of evidence supporting its efficacy is expanding.
- Synthetic cannabinoids are prevalent, and toxidromes exist – yet, trainees are not current on these.
The challenges facing those of us dedicated to fighting addiction have never been greater. I would urge more trainees and psychiatrists to join the AAAP in light of the opioid crisis and the potential fallout tied to marijuana legalization. I am grateful to have the opportunity to join my colleagues in this fight. Becoming part of the AAAP has led to a highly rewarding, career-enriching experience.
This article was updated 1/17/17.
Dr. Ahmed is a third-year resident in the department of psychiatry at Nassau University Medical Center, East Meadow, New York. Besides addiction psychiatry, his interests include public social psychiatry, health care policy, health disparities, and mental health stigma. Dr. Ahmed is a member of the American Psychiatric Association, the American Society of Clinical Psychopharmacology, and the American Association for Social Psychiatry.
When I came to this country, even before my current residency, I launched my addiction psychiatry career by researching nicotine addiction in schizophrenia patients. Those early experiences gave me a greater understanding of the health concerns and life experiences of people with addictions – and those more likely to develop them.
So imagine my excitement when I first became acquainted with the American Academy of Addiction Psychiatry (AAAP). I first learned about the AAAP, its mission, and activities at the 2017 American Psychiatric Association’s annual meeting in San Diego.
Getting in and involved
After returning to New York, I took those next steps and joined up, which opened the AAAP gates so I could receive its newsletters and submission calls, gain access to resources such as The American Journal on Addictions, survey the various joinable task forces, as well as discover who might be available to me as a mentor as part of the AAAP’s mentor-mentee program.
Sometime during my third-year residency training, I received a member-email advertising the AAAP 28th Annual Meeting and Scientific Symposium, and soon after that, received another email calling for research submissions to be presented there, as well as an invitation to apply for a trainee travel scholarship that would defray the cost for and allow its fellows to attend the meeting in San Diego. That alone was enticing enough to apply. But even more enticing was the opportunity to showcase the addiction work I had been doing during my residency, as well as to meet other members at various levels of the AAAP to determine whether I wanted to become more involved.
Pursuing experiences
I did not think twice about applying for the poster presentation and the travel scholarship. The AAAP’s online application forms for both were easy to understand and very well structured, which greatly helped me with filling out and formatting my applications. Taking the initiative toward even these first AAAP offerings brought more positive echoes. I was thrilled when the poster I proposed was accepted, mostly because it would give me the chance to present my recent addiction psych work from a higher platform. A few weeks later, I was thrilled again when I received an AAAP email congratulating me on being awarded the San Diego 28th annual meeting travel scholarship, which would waive the annual membership and conference registration fees, in addition to defraying my travel costs. Pacific breezes, here I come. And there I went. (Thanks to my extremely supportive training director, who first nominated me for the award.)
On the ground at 2017 AAAP
The 28th AAAP annual meeting opened on a balmy December Thursday, and that’s the day I arrived. I attended many addiction workshops and symposiums, which featured premier figures in addiction psychiatry. Of the numerous trainee-specific events I attended, the most informative was the “Fellowship Forum: Exploring the Field of Addiction Psychiatry.” At this forum, I learned the true benefits of doing an addiction psychiatry fellowship, while meeting many of the fellowship program directors of top institutions. Having them all under one “roof” made it easy to compare and contrast the specific training they offered.
Then came what were, for me, major highlights of the AAAP 2017. After I delivered my poster presentation and shared my research, I was able to receive very close, constructive feedback from the field’s most experienced professionals. And, finally, I met my AAAP mentors face to face: Dr. Amy Yule of Harvard Medical School, Boston; Dr. Thomas Penders, of East Carolina University, Greenville, N.C.; and Dr. Cornel Stanciu of Dartmouth-Hitchcock Medical Center, Lebanon, N.H.
One AAAP trainee’s takeaways
All AAAP trainees, fellows, presenters leave the meeting with their own conclusions, but my biggest takeaways were:
- Regarding barriers to buprenorphine, emerging research supports similar efficacy for long-acting injectable naltrexone.
- Various protocols for rapid implementation of naltrexone are being used, and these allow for smoother transition and shorter “washout” periods.
- We should not overlook the effects of tobacco use in our patient population – and should address it aggressively, regardless of psychiatric comorbidities.
- The cannabinoid CBD receptors that exist on the dopamine pathway strengthen and complicate their relationship with psychosis.
- , especially in rural and remote settings. The body of evidence supporting its efficacy is expanding.
- Synthetic cannabinoids are prevalent, and toxidromes exist – yet, trainees are not current on these.
The challenges facing those of us dedicated to fighting addiction have never been greater. I would urge more trainees and psychiatrists to join the AAAP in light of the opioid crisis and the potential fallout tied to marijuana legalization. I am grateful to have the opportunity to join my colleagues in this fight. Becoming part of the AAAP has led to a highly rewarding, career-enriching experience.
This article was updated 1/17/17.
Dr. Ahmed is a third-year resident in the department of psychiatry at Nassau University Medical Center, East Meadow, New York. Besides addiction psychiatry, his interests include public social psychiatry, health care policy, health disparities, and mental health stigma. Dr. Ahmed is a member of the American Psychiatric Association, the American Society of Clinical Psychopharmacology, and the American Association for Social Psychiatry.
When I came to this country, even before my current residency, I launched my addiction psychiatry career by researching nicotine addiction in schizophrenia patients. Those early experiences gave me a greater understanding of the health concerns and life experiences of people with addictions – and those more likely to develop them.
So imagine my excitement when I first became acquainted with the American Academy of Addiction Psychiatry (AAAP). I first learned about the AAAP, its mission, and activities at the 2017 American Psychiatric Association’s annual meeting in San Diego.
Getting in and involved
After returning to New York, I took those next steps and joined up, which opened the AAAP gates so I could receive its newsletters and submission calls, gain access to resources such as The American Journal on Addictions, survey the various joinable task forces, as well as discover who might be available to me as a mentor as part of the AAAP’s mentor-mentee program.
Sometime during my third-year residency training, I received a member-email advertising the AAAP 28th Annual Meeting and Scientific Symposium, and soon after that, received another email calling for research submissions to be presented there, as well as an invitation to apply for a trainee travel scholarship that would defray the cost for and allow its fellows to attend the meeting in San Diego. That alone was enticing enough to apply. But even more enticing was the opportunity to showcase the addiction work I had been doing during my residency, as well as to meet other members at various levels of the AAAP to determine whether I wanted to become more involved.
Pursuing experiences
I did not think twice about applying for the poster presentation and the travel scholarship. The AAAP’s online application forms for both were easy to understand and very well structured, which greatly helped me with filling out and formatting my applications. Taking the initiative toward even these first AAAP offerings brought more positive echoes. I was thrilled when the poster I proposed was accepted, mostly because it would give me the chance to present my recent addiction psych work from a higher platform. A few weeks later, I was thrilled again when I received an AAAP email congratulating me on being awarded the San Diego 28th annual meeting travel scholarship, which would waive the annual membership and conference registration fees, in addition to defraying my travel costs. Pacific breezes, here I come. And there I went. (Thanks to my extremely supportive training director, who first nominated me for the award.)
On the ground at 2017 AAAP
The 28th AAAP annual meeting opened on a balmy December Thursday, and that’s the day I arrived. I attended many addiction workshops and symposiums, which featured premier figures in addiction psychiatry. Of the numerous trainee-specific events I attended, the most informative was the “Fellowship Forum: Exploring the Field of Addiction Psychiatry.” At this forum, I learned the true benefits of doing an addiction psychiatry fellowship, while meeting many of the fellowship program directors of top institutions. Having them all under one “roof” made it easy to compare and contrast the specific training they offered.
Then came what were, for me, major highlights of the AAAP 2017. After I delivered my poster presentation and shared my research, I was able to receive very close, constructive feedback from the field’s most experienced professionals. And, finally, I met my AAAP mentors face to face: Dr. Amy Yule of Harvard Medical School, Boston; Dr. Thomas Penders, of East Carolina University, Greenville, N.C.; and Dr. Cornel Stanciu of Dartmouth-Hitchcock Medical Center, Lebanon, N.H.
One AAAP trainee’s takeaways
All AAAP trainees, fellows, presenters leave the meeting with their own conclusions, but my biggest takeaways were:
- Regarding barriers to buprenorphine, emerging research supports similar efficacy for long-acting injectable naltrexone.
- Various protocols for rapid implementation of naltrexone are being used, and these allow for smoother transition and shorter “washout” periods.
- We should not overlook the effects of tobacco use in our patient population – and should address it aggressively, regardless of psychiatric comorbidities.
- The cannabinoid CBD receptors that exist on the dopamine pathway strengthen and complicate their relationship with psychosis.
- , especially in rural and remote settings. The body of evidence supporting its efficacy is expanding.
- Synthetic cannabinoids are prevalent, and toxidromes exist – yet, trainees are not current on these.
The challenges facing those of us dedicated to fighting addiction have never been greater. I would urge more trainees and psychiatrists to join the AAAP in light of the opioid crisis and the potential fallout tied to marijuana legalization. I am grateful to have the opportunity to join my colleagues in this fight. Becoming part of the AAAP has led to a highly rewarding, career-enriching experience.
This article was updated 1/17/17.
Dr. Ahmed is a third-year resident in the department of psychiatry at Nassau University Medical Center, East Meadow, New York. Besides addiction psychiatry, his interests include public social psychiatry, health care policy, health disparities, and mental health stigma. Dr. Ahmed is a member of the American Psychiatric Association, the American Society of Clinical Psychopharmacology, and the American Association for Social Psychiatry.
How to set up your own RSS feed
In my last column, I reviewed the reasons why RSS news feeds can be a useful tool for keeping abreast on frequently updated information, including blog entries, news headlines, audio, and video, without having to check multiple Web pages every day.
This can help increase readership on your website, publicize a podcast, or keep your patients up to date on the latest treatments and procedures in your practice. And if your name appears in news or gossip sites, you will be alerted immediately.
Alternatively, many organizations that publish their own articles and news stories use a content management system (CMS) to organize, store, and publish their material, including RSS feeds. Examples include Drupal and Plone, which are both free, open source programs. Stand-alone RSS creation programs also exist; one popular example is RSS Builder, also a free and open source.
Disadvantages of free systems include advertisements (which can sometimes be removed for a monthly fee) and little or no technical support – and you will probably be limited to a single feed. You’ll also have to add and update headlines, links, and descriptive text manually. Your free feed can become quite expensive if you or staffers are forced to spend an inordinate amount of time maintaining it. Paid RSS editors like FeedForAll and NewzAlert Composer allow easier and less time-consuming content creation and maintenance.
Once you have picked a service or application, you can create your first feed, a process that will be different from program to program. But all feeds will need some basic data: a name (which should be the same as your practice or website); the URL for your website, to help viewers link back to your home page; and a description – a sentence or two describing the general content on the feed.
The next step is to populate the feed with content. Enter the title of each article, blog post, podcast episode, etc.; the URL that links directly to that content; and the publishing date. Each entry should have its own short, interesting description, which is what potential readers will see before they choose to click your entry in their RSS readers, and a global unique identifier (GUID), which the RSS readers use to detect changes or updates.
When all of your content is entered, all that remains is to export your feed to an extensible markup language (XML) file, which will allow visitors to subscribe to it. Upload the XML file to your website, place it on your home page, and click the “publish feed” button.
Once your feed is live, you’ll want to list it on some of the many RSS feed directories to maximize its visibility on search engines. There are literally hundreds of such directories; look for medically oriented ones that do not charge fees, and do not require a reciprocal link back to their website. Add each directory’s URL to your XML file.
Addendum: In my December 2017 column (“Your Online Reputation”), I suggested encouraging your most devoted patients to post favorable reviews about you on the “rating” websites. Several readers (including a practice consultant) have suggested making a laptop or tablet available in your office for that purpose. While that sounds like a great idea, most rating portals track incoming IP addresses, and automatically reject multiple reviews originating from the same computer.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News.
In my last column, I reviewed the reasons why RSS news feeds can be a useful tool for keeping abreast on frequently updated information, including blog entries, news headlines, audio, and video, without having to check multiple Web pages every day.
This can help increase readership on your website, publicize a podcast, or keep your patients up to date on the latest treatments and procedures in your practice. And if your name appears in news or gossip sites, you will be alerted immediately.
Alternatively, many organizations that publish their own articles and news stories use a content management system (CMS) to organize, store, and publish their material, including RSS feeds. Examples include Drupal and Plone, which are both free, open source programs. Stand-alone RSS creation programs also exist; one popular example is RSS Builder, also a free and open source.
Disadvantages of free systems include advertisements (which can sometimes be removed for a monthly fee) and little or no technical support – and you will probably be limited to a single feed. You’ll also have to add and update headlines, links, and descriptive text manually. Your free feed can become quite expensive if you or staffers are forced to spend an inordinate amount of time maintaining it. Paid RSS editors like FeedForAll and NewzAlert Composer allow easier and less time-consuming content creation and maintenance.
Once you have picked a service or application, you can create your first feed, a process that will be different from program to program. But all feeds will need some basic data: a name (which should be the same as your practice or website); the URL for your website, to help viewers link back to your home page; and a description – a sentence or two describing the general content on the feed.
The next step is to populate the feed with content. Enter the title of each article, blog post, podcast episode, etc.; the URL that links directly to that content; and the publishing date. Each entry should have its own short, interesting description, which is what potential readers will see before they choose to click your entry in their RSS readers, and a global unique identifier (GUID), which the RSS readers use to detect changes or updates.
When all of your content is entered, all that remains is to export your feed to an extensible markup language (XML) file, which will allow visitors to subscribe to it. Upload the XML file to your website, place it on your home page, and click the “publish feed” button.
Once your feed is live, you’ll want to list it on some of the many RSS feed directories to maximize its visibility on search engines. There are literally hundreds of such directories; look for medically oriented ones that do not charge fees, and do not require a reciprocal link back to their website. Add each directory’s URL to your XML file.
Addendum: In my December 2017 column (“Your Online Reputation”), I suggested encouraging your most devoted patients to post favorable reviews about you on the “rating” websites. Several readers (including a practice consultant) have suggested making a laptop or tablet available in your office for that purpose. While that sounds like a great idea, most rating portals track incoming IP addresses, and automatically reject multiple reviews originating from the same computer.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News.
In my last column, I reviewed the reasons why RSS news feeds can be a useful tool for keeping abreast on frequently updated information, including blog entries, news headlines, audio, and video, without having to check multiple Web pages every day.
This can help increase readership on your website, publicize a podcast, or keep your patients up to date on the latest treatments and procedures in your practice. And if your name appears in news or gossip sites, you will be alerted immediately.
Alternatively, many organizations that publish their own articles and news stories use a content management system (CMS) to organize, store, and publish their material, including RSS feeds. Examples include Drupal and Plone, which are both free, open source programs. Stand-alone RSS creation programs also exist; one popular example is RSS Builder, also a free and open source.
Disadvantages of free systems include advertisements (which can sometimes be removed for a monthly fee) and little or no technical support – and you will probably be limited to a single feed. You’ll also have to add and update headlines, links, and descriptive text manually. Your free feed can become quite expensive if you or staffers are forced to spend an inordinate amount of time maintaining it. Paid RSS editors like FeedForAll and NewzAlert Composer allow easier and less time-consuming content creation and maintenance.
Once you have picked a service or application, you can create your first feed, a process that will be different from program to program. But all feeds will need some basic data: a name (which should be the same as your practice or website); the URL for your website, to help viewers link back to your home page; and a description – a sentence or two describing the general content on the feed.
The next step is to populate the feed with content. Enter the title of each article, blog post, podcast episode, etc.; the URL that links directly to that content; and the publishing date. Each entry should have its own short, interesting description, which is what potential readers will see before they choose to click your entry in their RSS readers, and a global unique identifier (GUID), which the RSS readers use to detect changes or updates.
When all of your content is entered, all that remains is to export your feed to an extensible markup language (XML) file, which will allow visitors to subscribe to it. Upload the XML file to your website, place it on your home page, and click the “publish feed” button.
Once your feed is live, you’ll want to list it on some of the many RSS feed directories to maximize its visibility on search engines. There are literally hundreds of such directories; look for medically oriented ones that do not charge fees, and do not require a reciprocal link back to their website. Add each directory’s URL to your XML file.
Addendum: In my December 2017 column (“Your Online Reputation”), I suggested encouraging your most devoted patients to post favorable reviews about you on the “rating” websites. Several readers (including a practice consultant) have suggested making a laptop or tablet available in your office for that purpose. While that sounds like a great idea, most rating portals track incoming IP addresses, and automatically reject multiple reviews originating from the same computer.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News.
High-dose hydrogen peroxide for SKs
Formerly known as A-101, . It is a 40% hydrogen peroxide topical solution that is applied to raised SKs as an in-office procedure. As previously reported, SKs are composed of hyperadherent senescent cells that are arrested in the G1 phase of the cell cycle. They exhibit decreased apoptotic cell death, compared with normal skin.
Two double-blind vehicle-controlled studies demonstrated that more patients were clear or near clear of SKs after use of the 40% concentration solution than those in the vehicle group, according to the prescribing information. In the studies, patients with four raised SKs of the face, trunk or extremities were treated with Eskata at baseline and 3 weeks later, if necessary, or a vehicle. None of those in the vehicle group were cleared at follow-up (day 106), compared with 4% and 8% of those treated with Eskata, and at least three of the four lesions treated with Eskata had cleared in 13% and 23% at follow-up. Local skin reactions were mostly mild and transient, the most common being itching, stinging, crusting, swelling, redness, and scaling at the application site.
In the clinical studies, the solution was applied up to two times, on day 0 and again 3 weeks later on day 22. The lesion should first be cleansed with alcohol, and appropriate measures should be taken to ensure Eskata does not come in contact with the eyes. Nitrile or vinyl examination gloves should be used for application. Eskata is applied to the SKs with a pen-like applicator. The solution is applied uniformly in a circular motion with excess and the surrounding area patted dry with an absorbent wipe. Cotton gauze, tips, paper towels, or tissue should not be used as organic compounds can react with high concentrations of hydrogen peroxide. In a treatment session, one lesion may be treated up to four times, 1 minute apart. The applicator is used only once and may be discarded after lesions are treated. The packages may be stored at controlled room temperature (68° F to 77° F).
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Dr. Wesley has served on an advisory board panel for Aclaris, the manufacturer of Eskata. Dr. Talakoub had no related disclosures. Write to them at [email protected].
Formerly known as A-101, . It is a 40% hydrogen peroxide topical solution that is applied to raised SKs as an in-office procedure. As previously reported, SKs are composed of hyperadherent senescent cells that are arrested in the G1 phase of the cell cycle. They exhibit decreased apoptotic cell death, compared with normal skin.
Two double-blind vehicle-controlled studies demonstrated that more patients were clear or near clear of SKs after use of the 40% concentration solution than those in the vehicle group, according to the prescribing information. In the studies, patients with four raised SKs of the face, trunk or extremities were treated with Eskata at baseline and 3 weeks later, if necessary, or a vehicle. None of those in the vehicle group were cleared at follow-up (day 106), compared with 4% and 8% of those treated with Eskata, and at least three of the four lesions treated with Eskata had cleared in 13% and 23% at follow-up. Local skin reactions were mostly mild and transient, the most common being itching, stinging, crusting, swelling, redness, and scaling at the application site.
In the clinical studies, the solution was applied up to two times, on day 0 and again 3 weeks later on day 22. The lesion should first be cleansed with alcohol, and appropriate measures should be taken to ensure Eskata does not come in contact with the eyes. Nitrile or vinyl examination gloves should be used for application. Eskata is applied to the SKs with a pen-like applicator. The solution is applied uniformly in a circular motion with excess and the surrounding area patted dry with an absorbent wipe. Cotton gauze, tips, paper towels, or tissue should not be used as organic compounds can react with high concentrations of hydrogen peroxide. In a treatment session, one lesion may be treated up to four times, 1 minute apart. The applicator is used only once and may be discarded after lesions are treated. The packages may be stored at controlled room temperature (68° F to 77° F).
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Dr. Wesley has served on an advisory board panel for Aclaris, the manufacturer of Eskata. Dr. Talakoub had no related disclosures. Write to them at [email protected].
Formerly known as A-101, . It is a 40% hydrogen peroxide topical solution that is applied to raised SKs as an in-office procedure. As previously reported, SKs are composed of hyperadherent senescent cells that are arrested in the G1 phase of the cell cycle. They exhibit decreased apoptotic cell death, compared with normal skin.
Two double-blind vehicle-controlled studies demonstrated that more patients were clear or near clear of SKs after use of the 40% concentration solution than those in the vehicle group, according to the prescribing information. In the studies, patients with four raised SKs of the face, trunk or extremities were treated with Eskata at baseline and 3 weeks later, if necessary, or a vehicle. None of those in the vehicle group were cleared at follow-up (day 106), compared with 4% and 8% of those treated with Eskata, and at least three of the four lesions treated with Eskata had cleared in 13% and 23% at follow-up. Local skin reactions were mostly mild and transient, the most common being itching, stinging, crusting, swelling, redness, and scaling at the application site.
In the clinical studies, the solution was applied up to two times, on day 0 and again 3 weeks later on day 22. The lesion should first be cleansed with alcohol, and appropriate measures should be taken to ensure Eskata does not come in contact with the eyes. Nitrile or vinyl examination gloves should be used for application. Eskata is applied to the SKs with a pen-like applicator. The solution is applied uniformly in a circular motion with excess and the surrounding area patted dry with an absorbent wipe. Cotton gauze, tips, paper towels, or tissue should not be used as organic compounds can react with high concentrations of hydrogen peroxide. In a treatment session, one lesion may be treated up to four times, 1 minute apart. The applicator is used only once and may be discarded after lesions are treated. The packages may be stored at controlled room temperature (68° F to 77° F).
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Dr. Wesley has served on an advisory board panel for Aclaris, the manufacturer of Eskata. Dr. Talakoub had no related disclosures. Write to them at [email protected].
Experience may not always the best teacher
“In medicine, a lack of experience may not actually be a bad thing.” The words leapt off my tablet, and my blood pressure hit the stratosphere. How could a 30-year-old physician who is still doing his cardiology fellowship have the nerve to take a cheap shot at one of the cornerstones of my identity? (“For Doctors, Age May Be More Than a Number,” by Haider Javed Warraich, The New York Times, Jan. 6, 2018.) My reputation as a skilled pediatrician was built on the breadth and depth of my clinical experience. In the first 30 years of my professional career, I had participated in more than 300,000 patient encounters. Wasn’t that effort good for something?
He wrote that in a study of hospitalists (BMJ. 2017;357:j1797), the patient death rate increased steadily from 10.8% for physicians younger than 40 years to 12.1% for those 6o years and older, except those physicians treating a high volume of patients. He also cites a study in which the authors analyzed 62 studies of quality of care. The investigators found that slightly more than half of the studies “report decreasing performance with increasing years in practice” (Ann Int Med. 2005 Feb 15;142[4]:260-73).
Still in a defensive mode, I decided to chase down the references and take a closer look. In the study demonstrating patient mortality increasing with physician age, we must consider that these physicians were hospitalists and the patients were elderly. The observations may not be applicable to those seeing younger patients in a primary care office setting.
Another caveat listed by the authors is that the association between increasing patient mortality with increasing physician age did not hold true “among physicians with high volumes of patients.” As a high-volume practitioner myself, I found this comforting. It may be that there is some threshold effect in which a little experience is a negative but a ton of experience is a plus. Experience can teach efficiency. But, of course, efficiency doesn’t always equate with quality.
The study citing a decline in quality of care with physician age is a bit more troublesome. However, it may be that, rightly or wrongly, older physicians who have seen the tide go in and out enough times are more hesitant to accept and adhere to “evidence-based” quality measures that they see as ephemeral.
After what for me was an inflammatory opening, Dr. Warraich’s article closes with the observation that “mentorship is a two-way street.” Any physician who has worked with medical students and inquisitive house officers realizes that our surplus of experience always can stand to gain a little refreshing from those who have little of their own.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.”
“In medicine, a lack of experience may not actually be a bad thing.” The words leapt off my tablet, and my blood pressure hit the stratosphere. How could a 30-year-old physician who is still doing his cardiology fellowship have the nerve to take a cheap shot at one of the cornerstones of my identity? (“For Doctors, Age May Be More Than a Number,” by Haider Javed Warraich, The New York Times, Jan. 6, 2018.) My reputation as a skilled pediatrician was built on the breadth and depth of my clinical experience. In the first 30 years of my professional career, I had participated in more than 300,000 patient encounters. Wasn’t that effort good for something?
He wrote that in a study of hospitalists (BMJ. 2017;357:j1797), the patient death rate increased steadily from 10.8% for physicians younger than 40 years to 12.1% for those 6o years and older, except those physicians treating a high volume of patients. He also cites a study in which the authors analyzed 62 studies of quality of care. The investigators found that slightly more than half of the studies “report decreasing performance with increasing years in practice” (Ann Int Med. 2005 Feb 15;142[4]:260-73).
Still in a defensive mode, I decided to chase down the references and take a closer look. In the study demonstrating patient mortality increasing with physician age, we must consider that these physicians were hospitalists and the patients were elderly. The observations may not be applicable to those seeing younger patients in a primary care office setting.
Another caveat listed by the authors is that the association between increasing patient mortality with increasing physician age did not hold true “among physicians with high volumes of patients.” As a high-volume practitioner myself, I found this comforting. It may be that there is some threshold effect in which a little experience is a negative but a ton of experience is a plus. Experience can teach efficiency. But, of course, efficiency doesn’t always equate with quality.
The study citing a decline in quality of care with physician age is a bit more troublesome. However, it may be that, rightly or wrongly, older physicians who have seen the tide go in and out enough times are more hesitant to accept and adhere to “evidence-based” quality measures that they see as ephemeral.
After what for me was an inflammatory opening, Dr. Warraich’s article closes with the observation that “mentorship is a two-way street.” Any physician who has worked with medical students and inquisitive house officers realizes that our surplus of experience always can stand to gain a little refreshing from those who have little of their own.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.”
“In medicine, a lack of experience may not actually be a bad thing.” The words leapt off my tablet, and my blood pressure hit the stratosphere. How could a 30-year-old physician who is still doing his cardiology fellowship have the nerve to take a cheap shot at one of the cornerstones of my identity? (“For Doctors, Age May Be More Than a Number,” by Haider Javed Warraich, The New York Times, Jan. 6, 2018.) My reputation as a skilled pediatrician was built on the breadth and depth of my clinical experience. In the first 30 years of my professional career, I had participated in more than 300,000 patient encounters. Wasn’t that effort good for something?
He wrote that in a study of hospitalists (BMJ. 2017;357:j1797), the patient death rate increased steadily from 10.8% for physicians younger than 40 years to 12.1% for those 6o years and older, except those physicians treating a high volume of patients. He also cites a study in which the authors analyzed 62 studies of quality of care. The investigators found that slightly more than half of the studies “report decreasing performance with increasing years in practice” (Ann Int Med. 2005 Feb 15;142[4]:260-73).
Still in a defensive mode, I decided to chase down the references and take a closer look. In the study demonstrating patient mortality increasing with physician age, we must consider that these physicians were hospitalists and the patients were elderly. The observations may not be applicable to those seeing younger patients in a primary care office setting.
Another caveat listed by the authors is that the association between increasing patient mortality with increasing physician age did not hold true “among physicians with high volumes of patients.” As a high-volume practitioner myself, I found this comforting. It may be that there is some threshold effect in which a little experience is a negative but a ton of experience is a plus. Experience can teach efficiency. But, of course, efficiency doesn’t always equate with quality.
The study citing a decline in quality of care with physician age is a bit more troublesome. However, it may be that, rightly or wrongly, older physicians who have seen the tide go in and out enough times are more hesitant to accept and adhere to “evidence-based” quality measures that they see as ephemeral.
After what for me was an inflammatory opening, Dr. Warraich’s article closes with the observation that “mentorship is a two-way street.” Any physician who has worked with medical students and inquisitive house officers realizes that our surplus of experience always can stand to gain a little refreshing from those who have little of their own.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.”
Families are essential partners in treating substance use disorders
Addiction used to be considered a moral failing, and the family was blamed for keeping the relative with addictions sick, through behaviors labeled “codependency” and “enabling.” The opioid epidemic can take credit for putting a serious dent in these destructive and stigmatizing notions. When psychiatrists actively include families as educated treatment partners, fatalities are less likely, and the havoc created by addiction on families is mitigated.
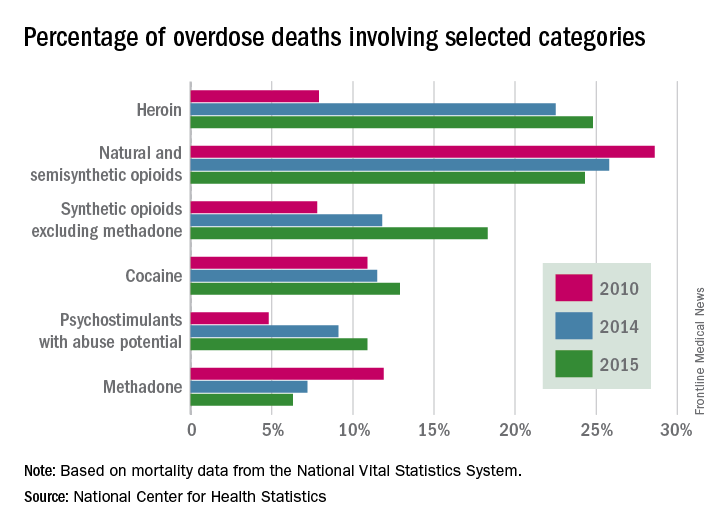
Genes and addiction
What causes addiction? Statistics show that Native Americans fare the worst of all minority groups, with death by opiates in whites and Native Americans double or triple the rates of African Americans and Latinos. Reasons put forward for Native American deaths are their vulnerability related to systemic racism, intergenerational trauma, and lack of access to health care. These “reasons” are well known to contribute to poor overall health status of impoverished communities.
Among impoverished white communities, the Monongahela Valley of Pennsylvania has been studied by Katherine McLean, PhD, as an example of postindustrial decay (Int J Drug Policy. 2016;[29]:19-26). Once a global center of steel production, the exodus of jobs, residents, and businesses since the early 1980s is thought to contribute to the high numbers of opioid deaths. A qualitative study of the people with addiction in the deteriorating mill city of McKeesport, Pa., characterized a risk environment hidden behind closed doors, and populated by unprepared, ambivalent overdose “assistants.” These people are “co-drug” users who themselves are reluctant to step forward because of fear of getting in trouble. The participants described the hopelessness and lack of opportunity as driving the use of heroin, with many stating that jobs and community reinvestment are needed to reduce fatalities. This certainly resonates with the Native American experience.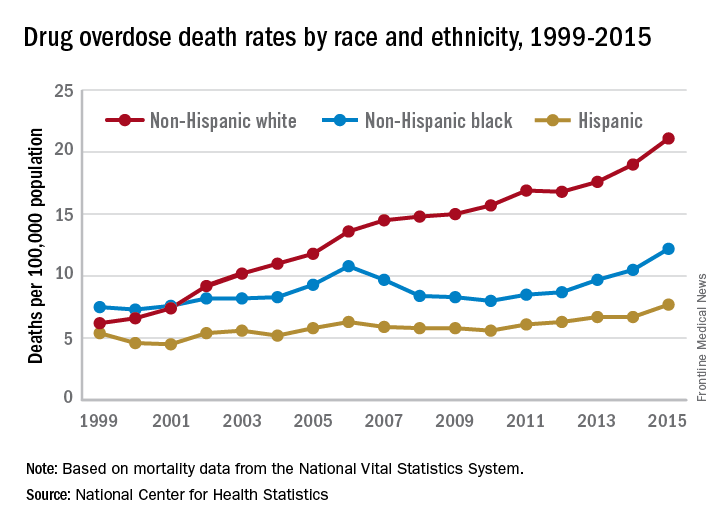
People with the AA variant of OXTR also have been shown to have less secure adult attachment and more social anxiety (World J Biol Psychiatry. 2016;17[1]:76-83). Comparing people with OXTR variants, the AA genotype was associated with a perceived negative social environment and significantly increased PTSD symptoms, whereas the GG genotype was protective.
However, for many decades, psychological theories about the defects of individuals and their moral failing have prevailed. In the family, aspersions have been cast on the family’s deficits in terms of setting limits and their enabling behaviors, mostly focusing on wives and mothers. The social mantra has been that since not all people get addicted, the strong resist and the weak succumb. Psychiatry has focused on providing psychotherapy to correct the personal deficits of the weak and addicted and, from a family perspective, on correcting negative personality traits in the caregivers, classified as codependency.
Roots of codependency
The concept of codependency began as a grassroots idea in the 1960s to describe family members, usually wives or mothers, who were deemed excessive in their caring for their husband or son with addiction. The term was used to describe women who had an overresponsibility for relationships, rather than a responsibility to self. Support groups for codependency, such as Co-Dependents Anonymous, Al-Anon/Alateen, Nar-Anon, and Adult Children of Alcoholics, based on the 12-step program model of Alcoholics Anonymous, were established. Codependents were negatively labeled as rescuers, supporters, and confidantes of their family member with substance use disorder. These helpers were described as dependent on the other person’s poor functioning to satisfy their own emotional needs.
In these early descriptions, there was a lack of discussion about possible deeper family dynamics: Inequality in financial independence of each partner, the desire for family stability focusing on the welfare of children, the real possibility that disrupting a relationship might result in violence if the woman was more assertive. Women were blamed for trying to love their spouse out of the addiction. One aspect of codependency is self-sacrifice, which used to be considered an important trait of the good wife but became a negative trait in the 1960s. Recovery from codependence was considered achieved when the wife or mother expressed healthy self-assertiveness. Some scholars did state, however, that they believed that codependency was not a negative trait, but rather a healthy personality trait taken to excess. In most studies, women with high codependency ratings have tended to be unemployed and with lower educations. Their behavior had been considered worthy of a DSM inclusion as a personality disorder, but luckily, this was thwarted.
A family is forced to make some accounting when an individual develops a substance use disorder. If the family tries to maintain equilibrium and keeps things as stable as possible for the sake of maintaining the family unit or the stability of the lives of the children, accommodation will be required. Accommodation minimizes the impact of the addiction: the sober spouse stepping up to complete the roles of the ill relative and in all ways reducing the impact of dysfunction on the family. If you swap out the illness and consider the ill family member as having cancer or respiratory distress, then you reframe the spouse’s “codependence” as the behavior of a caring spouse.
This long preamble is intended to illustrate how little family dynamics have to do with the etiology of addiction. Addiction is a chronic neurobiological disease, and like all diseases, individuals, and couples and families have the option of learning to cope well. We, as psychiatrists, must move away from blaming people (usually women who have less-than-optimal coping skills) and consider how best to engage partners and families in optimal programs.*
Families can benefit from specialist treatment and thus contribute to the recovery process. One example is a telepsychiatry program that aimed to improve family coping skills to cope with relatives who have substance use disorders. The Tele-intervention Model and Monitoring of Families of Drug Users is based on motivational interviewing and stages of change. Families were randomized into the intervention group (n = 163) or the usual treatment (n = 162). After 6 months of follow-up, the family members of the telepsychiatry group were twice as likely to modify their behavior (odds ratio; 2.08; 95% confidence interval, 1.18-3.65) (J Sub Use Misuse. 2017 Jan 28;52[2]:164-74). This model was organized so that each of nine calls had a specific goal to stimulate the family members in their process of change.
Family change and engagement also can occur through Alanon (Subst Use Misuse. 2015; 50[1]:62-71) and the use of the Arise program of Judith Landau, MD, (Landau J et al. Am J Drug Alcohol Abuse. 2000;26[3]:379-98).
In summary, the family can be engaged in treatment and can develop family coping skills to support their relative with this chronic neurobiological disease.
Dr. Heru is professor of psychiatry at the University of Colorado at Denver, Aurora. She is editor of “Working With Families in Medical Settings: A Multidisciplinary Guide for Psychiatrists and Other Health Professionals” (New York: Routledge, 2013). She has no conflicts of interest to disclose.
*Updated on January 12, 2018.
Addiction used to be considered a moral failing, and the family was blamed for keeping the relative with addictions sick, through behaviors labeled “codependency” and “enabling.” The opioid epidemic can take credit for putting a serious dent in these destructive and stigmatizing notions. When psychiatrists actively include families as educated treatment partners, fatalities are less likely, and the havoc created by addiction on families is mitigated.

Genes and addiction
What causes addiction? Statistics show that Native Americans fare the worst of all minority groups, with death by opiates in whites and Native Americans double or triple the rates of African Americans and Latinos. Reasons put forward for Native American deaths are their vulnerability related to systemic racism, intergenerational trauma, and lack of access to health care. These “reasons” are well known to contribute to poor overall health status of impoverished communities.
Among impoverished white communities, the Monongahela Valley of Pennsylvania has been studied by Katherine McLean, PhD, as an example of postindustrial decay (Int J Drug Policy. 2016;[29]:19-26). Once a global center of steel production, the exodus of jobs, residents, and businesses since the early 1980s is thought to contribute to the high numbers of opioid deaths. A qualitative study of the people with addiction in the deteriorating mill city of McKeesport, Pa., characterized a risk environment hidden behind closed doors, and populated by unprepared, ambivalent overdose “assistants.” These people are “co-drug” users who themselves are reluctant to step forward because of fear of getting in trouble. The participants described the hopelessness and lack of opportunity as driving the use of heroin, with many stating that jobs and community reinvestment are needed to reduce fatalities. This certainly resonates with the Native American experience.
People with the AA variant of OXTR also have been shown to have less secure adult attachment and more social anxiety (World J Biol Psychiatry. 2016;17[1]:76-83). Comparing people with OXTR variants, the AA genotype was associated with a perceived negative social environment and significantly increased PTSD symptoms, whereas the GG genotype was protective.
However, for many decades, psychological theories about the defects of individuals and their moral failing have prevailed. In the family, aspersions have been cast on the family’s deficits in terms of setting limits and their enabling behaviors, mostly focusing on wives and mothers. The social mantra has been that since not all people get addicted, the strong resist and the weak succumb. Psychiatry has focused on providing psychotherapy to correct the personal deficits of the weak and addicted and, from a family perspective, on correcting negative personality traits in the caregivers, classified as codependency.
Roots of codependency
The concept of codependency began as a grassroots idea in the 1960s to describe family members, usually wives or mothers, who were deemed excessive in their caring for their husband or son with addiction. The term was used to describe women who had an overresponsibility for relationships, rather than a responsibility to self. Support groups for codependency, such as Co-Dependents Anonymous, Al-Anon/Alateen, Nar-Anon, and Adult Children of Alcoholics, based on the 12-step program model of Alcoholics Anonymous, were established. Codependents were negatively labeled as rescuers, supporters, and confidantes of their family member with substance use disorder. These helpers were described as dependent on the other person’s poor functioning to satisfy their own emotional needs.
In these early descriptions, there was a lack of discussion about possible deeper family dynamics: Inequality in financial independence of each partner, the desire for family stability focusing on the welfare of children, the real possibility that disrupting a relationship might result in violence if the woman was more assertive. Women were blamed for trying to love their spouse out of the addiction. One aspect of codependency is self-sacrifice, which used to be considered an important trait of the good wife but became a negative trait in the 1960s. Recovery from codependence was considered achieved when the wife or mother expressed healthy self-assertiveness. Some scholars did state, however, that they believed that codependency was not a negative trait, but rather a healthy personality trait taken to excess. In most studies, women with high codependency ratings have tended to be unemployed and with lower educations. Their behavior had been considered worthy of a DSM inclusion as a personality disorder, but luckily, this was thwarted.
A family is forced to make some accounting when an individual develops a substance use disorder. If the family tries to maintain equilibrium and keeps things as stable as possible for the sake of maintaining the family unit or the stability of the lives of the children, accommodation will be required. Accommodation minimizes the impact of the addiction: the sober spouse stepping up to complete the roles of the ill relative and in all ways reducing the impact of dysfunction on the family. If you swap out the illness and consider the ill family member as having cancer or respiratory distress, then you reframe the spouse’s “codependence” as the behavior of a caring spouse.
This long preamble is intended to illustrate how little family dynamics have to do with the etiology of addiction. Addiction is a chronic neurobiological disease, and like all diseases, individuals, and couples and families have the option of learning to cope well. We, as psychiatrists, must move away from blaming people (usually women who have less-than-optimal coping skills) and consider how best to engage partners and families in optimal programs.*
Families can benefit from specialist treatment and thus contribute to the recovery process. One example is a telepsychiatry program that aimed to improve family coping skills to cope with relatives who have substance use disorders. The Tele-intervention Model and Monitoring of Families of Drug Users is based on motivational interviewing and stages of change. Families were randomized into the intervention group (n = 163) or the usual treatment (n = 162). After 6 months of follow-up, the family members of the telepsychiatry group were twice as likely to modify their behavior (odds ratio; 2.08; 95% confidence interval, 1.18-3.65) (J Sub Use Misuse. 2017 Jan 28;52[2]:164-74). This model was organized so that each of nine calls had a specific goal to stimulate the family members in their process of change.
Family change and engagement also can occur through Alanon (Subst Use Misuse. 2015; 50[1]:62-71) and the use of the Arise program of Judith Landau, MD, (Landau J et al. Am J Drug Alcohol Abuse. 2000;26[3]:379-98).
In summary, the family can be engaged in treatment and can develop family coping skills to support their relative with this chronic neurobiological disease.
Dr. Heru is professor of psychiatry at the University of Colorado at Denver, Aurora. She is editor of “Working With Families in Medical Settings: A Multidisciplinary Guide for Psychiatrists and Other Health Professionals” (New York: Routledge, 2013). She has no conflicts of interest to disclose.
*Updated on January 12, 2018.
Addiction used to be considered a moral failing, and the family was blamed for keeping the relative with addictions sick, through behaviors labeled “codependency” and “enabling.” The opioid epidemic can take credit for putting a serious dent in these destructive and stigmatizing notions. When psychiatrists actively include families as educated treatment partners, fatalities are less likely, and the havoc created by addiction on families is mitigated.

Genes and addiction
What causes addiction? Statistics show that Native Americans fare the worst of all minority groups, with death by opiates in whites and Native Americans double or triple the rates of African Americans and Latinos. Reasons put forward for Native American deaths are their vulnerability related to systemic racism, intergenerational trauma, and lack of access to health care. These “reasons” are well known to contribute to poor overall health status of impoverished communities.
Among impoverished white communities, the Monongahela Valley of Pennsylvania has been studied by Katherine McLean, PhD, as an example of postindustrial decay (Int J Drug Policy. 2016;[29]:19-26). Once a global center of steel production, the exodus of jobs, residents, and businesses since the early 1980s is thought to contribute to the high numbers of opioid deaths. A qualitative study of the people with addiction in the deteriorating mill city of McKeesport, Pa., characterized a risk environment hidden behind closed doors, and populated by unprepared, ambivalent overdose “assistants.” These people are “co-drug” users who themselves are reluctant to step forward because of fear of getting in trouble. The participants described the hopelessness and lack of opportunity as driving the use of heroin, with many stating that jobs and community reinvestment are needed to reduce fatalities. This certainly resonates with the Native American experience.
People with the AA variant of OXTR also have been shown to have less secure adult attachment and more social anxiety (World J Biol Psychiatry. 2016;17[1]:76-83). Comparing people with OXTR variants, the AA genotype was associated with a perceived negative social environment and significantly increased PTSD symptoms, whereas the GG genotype was protective.
However, for many decades, psychological theories about the defects of individuals and their moral failing have prevailed. In the family, aspersions have been cast on the family’s deficits in terms of setting limits and their enabling behaviors, mostly focusing on wives and mothers. The social mantra has been that since not all people get addicted, the strong resist and the weak succumb. Psychiatry has focused on providing psychotherapy to correct the personal deficits of the weak and addicted and, from a family perspective, on correcting negative personality traits in the caregivers, classified as codependency.
Roots of codependency
The concept of codependency began as a grassroots idea in the 1960s to describe family members, usually wives or mothers, who were deemed excessive in their caring for their husband or son with addiction. The term was used to describe women who had an overresponsibility for relationships, rather than a responsibility to self. Support groups for codependency, such as Co-Dependents Anonymous, Al-Anon/Alateen, Nar-Anon, and Adult Children of Alcoholics, based on the 12-step program model of Alcoholics Anonymous, were established. Codependents were negatively labeled as rescuers, supporters, and confidantes of their family member with substance use disorder. These helpers were described as dependent on the other person’s poor functioning to satisfy their own emotional needs.
In these early descriptions, there was a lack of discussion about possible deeper family dynamics: Inequality in financial independence of each partner, the desire for family stability focusing on the welfare of children, the real possibility that disrupting a relationship might result in violence if the woman was more assertive. Women were blamed for trying to love their spouse out of the addiction. One aspect of codependency is self-sacrifice, which used to be considered an important trait of the good wife but became a negative trait in the 1960s. Recovery from codependence was considered achieved when the wife or mother expressed healthy self-assertiveness. Some scholars did state, however, that they believed that codependency was not a negative trait, but rather a healthy personality trait taken to excess. In most studies, women with high codependency ratings have tended to be unemployed and with lower educations. Their behavior had been considered worthy of a DSM inclusion as a personality disorder, but luckily, this was thwarted.
A family is forced to make some accounting when an individual develops a substance use disorder. If the family tries to maintain equilibrium and keeps things as stable as possible for the sake of maintaining the family unit or the stability of the lives of the children, accommodation will be required. Accommodation minimizes the impact of the addiction: the sober spouse stepping up to complete the roles of the ill relative and in all ways reducing the impact of dysfunction on the family. If you swap out the illness and consider the ill family member as having cancer or respiratory distress, then you reframe the spouse’s “codependence” as the behavior of a caring spouse.
This long preamble is intended to illustrate how little family dynamics have to do with the etiology of addiction. Addiction is a chronic neurobiological disease, and like all diseases, individuals, and couples and families have the option of learning to cope well. We, as psychiatrists, must move away from blaming people (usually women who have less-than-optimal coping skills) and consider how best to engage partners and families in optimal programs.*
Families can benefit from specialist treatment and thus contribute to the recovery process. One example is a telepsychiatry program that aimed to improve family coping skills to cope with relatives who have substance use disorders. The Tele-intervention Model and Monitoring of Families of Drug Users is based on motivational interviewing and stages of change. Families were randomized into the intervention group (n = 163) or the usual treatment (n = 162). After 6 months of follow-up, the family members of the telepsychiatry group were twice as likely to modify their behavior (odds ratio; 2.08; 95% confidence interval, 1.18-3.65) (J Sub Use Misuse. 2017 Jan 28;52[2]:164-74). This model was organized so that each of nine calls had a specific goal to stimulate the family members in their process of change.
Family change and engagement also can occur through Alanon (Subst Use Misuse. 2015; 50[1]:62-71) and the use of the Arise program of Judith Landau, MD, (Landau J et al. Am J Drug Alcohol Abuse. 2000;26[3]:379-98).
In summary, the family can be engaged in treatment and can develop family coping skills to support their relative with this chronic neurobiological disease.
Dr. Heru is professor of psychiatry at the University of Colorado at Denver, Aurora. She is editor of “Working With Families in Medical Settings: A Multidisciplinary Guide for Psychiatrists and Other Health Professionals” (New York: Routledge, 2013). She has no conflicts of interest to disclose.
*Updated on January 12, 2018.
Improving Strength and Balance for Long-Term Care Residents At Risk for Falling: Suggestions for Practice
From the Geriatric Education and Research in Aging Sciences Centre, McMaster University Hamilton, ON (Dr. McArthur) and the University of Waterloo and Research Institute for Aging, Waterloo, ON (Dr. Giangregorio), Canada
Abstract
- Objective: To synthesize the available literature on exercise and falls reduction interventions in long-term care (LTC) and provide practical information for clinicians and other decision makers.
- Methods: Review of positive trials included in systematic reviews.
- Results: Falls are a major concern for residents, families, clinicians, and decision-makers in LTC. Exercise is recommended as part of a multifactorial falls prevention program for residents in LTC. Strength and balance exercises should be incorporated into the multifactorial falls prevention program. They should be challenging and progressed as the residents’ abilities improve. Evidence suggests that exercises should be completed 2 to 3 times per week for a period longer than 6 months. Exercise programs in LTC should be resident-centered and should consider residents’ potential physical and cognitive impairments. Exercises in standing should be prioritized where appropriate.
- Conclusion: Appropriately challenging and progressive strength and balance exercises should be included in a multifactorial falls prevention program for residents in LTC.
Key words: long-term care; nursing homes; falls reduction; exercise.
Falls are common in long-term care (LTC) homes: the estimated falls rate is 1.5 falls per bed per year, which is 3 times greater than that for older adults living in the community [1]. Falls can have significant consequences for residents in LTC, including functional disability, fractures, pain, reduced quality of life, and death [1–6]. Indeed, 25% of residents who are hospitalized after a fall die within 1 year [3]. Consequently, falls prevention programs are important to help in reducing falls and averting the associated negative consequences.
Exercise may address the circumstances and physical deconditioning that often contribute to falls in LTC residents. Weight shifting [7], walking, and transferring [8–10], are common activities that precede falls, suggesting that balance, gait, and functional mobility training may be possible targets for prevention. Additionally, it is estimated that LTC residents spend three quarters of their waking time in sedentary activities [11,12] and have a high prevalence of sarcopenia [13–16]. Challenging balance training and resistance exercise are well-known intervention for reducing falls [17] and improving muscle strength for community-dwelling older adults [18]. However, evidence around balance and strength training for preventing falls in LTC is mixed [17,19,20], and careful planning and modification of exercises is necessary to meet the needs of LTC residents.
Residents in LTC are often medically complex, with multiple comorbidities [21] that can affect their ability to meaningfully participate in exercise. In Canada, 56.3% of residents have a diagnosis of Alzheimer’s or other dementias, 25.0% have diabetes, 14.4% have chronic obstructive pulmonary disease, and 21.2% have experienced a stroke [21]. Residents also often have significant functional impairments. For example, 97% of residents require assistance with basic activities of daily living [21]. Therefore, the lack of effect of exercise as a single falls prevention strategy observed in previous studies may be because the often complex, multimorbid LTC population likely requires a multifactorial approach to fall prevention [17]. Additionally, organizational aspects of LTC homes (eg, specific funds dedicated to employing exercise professionals and to support exercise programming) can affect residents’ engagement in exercise [22,23]. Subsequently, prescribing exercises in the LTC context must consider both resident characteristics and organizational features of the LTC home (eg, professionals available to support exercise programming).
A comprehensive exercise prescription describes the elements of an appropriate exercise program to facilitate implementation of that program. The exercise prescription should include a description of the type (eg, balance, strength) and intensity of exercises (eg, subjective or objective measurement of how hard the resident is working) included in the program [24]. The prescription should also include a description of the dose of exercise: frequency of exercise participation (eg, 2 days per week), duration of individual exercise sessions (eg, 30-minute sessions), and duration of exercise program (eg, 12-week program) [24]. Lastly, the prescription should describe the setting of the exercise program (eg, group or individual basis) and the professional delivering the program (eg, physiotherapist, fitness instructor) [24].
Therefore, the objectives of this article are to (1) synthesize studies demonstrating a positive effect of exercise on reducing falls for residents in LTC; (2) provide an overview of the principles of balance and strength training to guide clinicians in designing appropriate exercise prescription; and (3) make suggestions for clinical practice regarding an appropriate strength and balance exercise protocol by considering the influence of the LTC context.
Methods
To provide clinicians and other policy-makers with a description of which balance and strength exercises may be effective for preventing falls, we synthesized trials that demonstrated a positive effect on reducing falls or falls risk for residents in LTC. Studies were identified through a database search for systematic reviews in PubMed, Ovid, and Google Scholar using the keywords falls, long-term care, nursing homes, exercise, strength, balance, and systematic reviews. Our purpose was to provide practical information on what works to prevent falls through balance and strength training for residents in LTC rather than to evaluate the available evidence. Therefore, only positive trials from systematic reviews were discussed, as we wanted to present exercises that seem to have a positive effect on decreasing falls. Positive trials were defined as those included in identified systematic reviews with a risk or rate ratio and confidence intervals below 1.0.
We first provide an overview of the conclusions of the systematic reviews found in our search. Next, for each positive trial we describe the following elements of the exercise component of the intervention: frequency, time of sessions, length of program, intensity, type of exercise including a description of the specific exercises performed, whether the intervention was delivered in a group or on an individual basis, the professional delivering the intervention, and any other features of the intervention aside from the exercise component. We used the ProFaNE taxonomy definitions [25] to identify and describe each element of the exercise interventions. Frequency is the number of times per week that residents engage in sessions, time of sessions is the amount allocated to each exercise session, duration of program is how long the resident participates in the exercise program, and intensity is the subjective or objective report of how hard the resident is working [25]. The types of exercises described were those targeting balance defined as “...the efficient transfer of bodyweight from one part of the body to another or challenges specific aspects of the balance systems (eg, vestibular system)” [25], and strength defined as “...contracting the muscles against a resistance to ‘overload’ and bring about a training effect in the muscular system” [25]. Strength could be either an external resistance (eg, dumbbell) or using body weight against gravity (eg, squat) [25].
Results
We found 3 systematic reviews that include exercise programs to reduce falls in LTC homes [17,19,20]. Overall, evidence suggests that exercise should be included as part of a multifactorial falls prevention program for residents in LTC. There is limited evidence that exercise as a single intervention prevents falls, and some trials, albeit underpowered, even demonstrate an increased risk of falling in the exercise group compared to control [19]. With regards to specific exercise programs, the Cochrane review found that gait, balance, and functional training decrease the rate of falls but not the risk of falling [26–28], and the 2013 review by Silva et al [20] concluded that combined exercise programs (ie, multiple types of exercise) that include balance tasks, are completed frequently (2–3 times per week), and over a long term (greater than 6 months) were most effective at preventing falls [20].
A more recent systematic review and meta-analysis [17] also concluded that there was no evidence that exercise as a single intervention can prevent falls for residents in LTC. Table 1 provides a description of the exercise component of the seven positive trials [29–35] that were included in the 3 systematic reviews we identified in our search.
Type of Exercise
Balance Exercises
There were 4 positive trials that included balance exercises in their intervention [31,33–35]. Trials that had a positive effect on reducing falls and included balance training employed mostly dynamic balance exercises in standing (Table 1). However, only 2 of the 7 trials provided a detailed description of their balance exercises (Table 1) [26,34]. Jensen et al [30] and Dyer et al [31] did not include a description of the balance training performed but stated that balance was part of the multicomponent exercise program. Becker et al [36] stated that participants performed standing balance exercises, while Schnelle et al [39] and Huang et al [32] did not include balance training in their trial.
Strength Exercises
Of the 7 positive trials included in this review, 6 included strength exercises [29–32,34,35]. The strength activities used in trials where exercise had a positive effect on decreasing falls included functional activities [29,31] and progressive resistance training [31,36] (Table 1). Functional activities are those that replicate what a resident might be required to do in their everyday life, such as performing sit-to-stands out of a chair (Figure)
Frequency, Time of Sessions, Duration of Program
In our description of positive trials, exercise was performed on 2 to 3 days per week for 20 to 75 minutes per session, for periods ranging from 4 to 52 weeks (Table 1).
Intensity
For the trials including balance exercises, one trial described the intensity as resident-specific [37] and another as individualized [33]. Two studies did not describe the intensity of their balance exercises [31,34]. The intensity of strength exercises included in the positive trials was individualized for one of the trial [29]. Two trials had participants complete 2 to 3 sets of 10 repetitions [32,35], with one indicating an intensity of 12–13 or “somewhat difficult” on the Borg Rating of Perceived Exertion Scale [32] and the other using a 10-rep max [35]. Two studies described their strength exercises as progressive [31,37], and one at a moderate to high intensity [30]. Lord et al prescribed 30 repetitions of each strength exercise [34].
Delivery of Intervention
Exercise was delivered in a group setting for 4 of the trials [31,32,34,36], individually for 2 of the trials [26,29], and the setting was not described for one of the trials (Table 1) [30]. Finally, only 3 of the 7 articles reported the professional delivering the intervention: one was research staff [29], one was geriatric nurses [32], and one was exercise assistants supported by a physiotherapist [31].
Discussion
There is limited evidence to support the use of strength and balance exercise as a single intervention to prevent falls in LTC. However, exercise should be included as part of a multifactorial falls prevention program. Trials that had a positive effect on decreasing falls training used dynamic balance exercises in standing, functional training, and progressive resistance training on 2 to 3 days per week, for 20 to 75 minutes per session, over 4 to 52 weeks. The intensity of balance exercises was individualized, and strength exercises were described as somewhat difficult or performed at a moderate to high intensity. Exercise was performed in a group or individually, and was delivered by research staff, geriatric nurses, exercise assistants supervised by physiotherapists, or more frequently, it was not reported who delivered the intervention.
Balance Training
Our work suggests that standing, dynamic balance exercises may be best to decrease falls. Example balance exercises include reducing the base of support (eg, standing with feet together instead of apart, or tandem with one foot in front), moving the center of gravity and control body position while standing (eg, reaching, weight shifting, stepping up or down), and standing without using arms for support or reducing reliance on the upper limbs for support (eg, use one hand on a handrail instead of two, or two fingers instead of the whole hand) [17]. It is well established that balance training programs, especially those including challenging exercises, can prevent falls in community-dwelling older adults [17]. However, the relationship is not as clear in LTC.
Strength Training
Reduced muscle strength has been identified as an important risk factor for falls [38]. There are also many psychological and metabolic benefits to strength training [39]. To induce change in muscular strength, resistance exercises need to be challenging and progressive. Our work suggests that strength training that is effective at decreasing falls is functional and progressive, and is completed at a moderate to high intensity. A resident should be able to do a strength exercise for one to two sets of 6 to 8 repetitions before being fatigued [40]. Once the resident can complete two sets of 13 to 15 repetitions easily the exercise should be progressed. Residents who are particularly deconditioned may need to begin with lower intensity strength exercises (eg, only do one set, with a lower resistance and progress to a higher resistance) [40]. Residents should perform resistance exercises for all major muscle groups [40]. Progression could include increasing the number of sets (eg, increase from one to two sets), the resistance (eg, holding dumbbells while squatting), or the intensity of the exercise (eg, squat lower or faster) [41].
Implementing Exercise Programs in LTC
Implementation of exercise programs into LTC homes should consider the dose of exercise (eg, time and frequency of sessions, duration of program), if they are delivered in a group or individual setting, and who is delivering them. First, trials included in this paper suggest that strength and balance exercises to prevent falls were delivered 2 to 3 times per week, for 20 to 75 minutes per session, over 4 to 52 weeks. Second, previous work has established that exercise programs delivered on 2 to 3 days per week over a period of more than 6 months are most effective at reducing falls in LTC [20]. Finally, a recent task force report from an international group of clinician researchers in LTC recommends twice weekly exercise sessions lasting 35 to 45 minutes each [40]. Therefore, strength and balance exercises to prevent falls in LTC should be delivered at least twice per week, for at least 20 minutes, for greater than 6 weeks’ duration.
Whether exercise should be performed in a group or individual setting remains unclear. Two of the 6 positive trials in this paper were completed individually, while 3 were in a group. The aforementioned task force also recommended that every resident who does not have contraindications to exercise must have an individualized exercise program as part of their health care plan [40]. However, whether the exercise program is provided on an individual basis or in a group setting was not delineated. Indeed, there are currently no recommendations concerning prioritizing group or individual exercise programs. Therefore, exercise programs being implemented into LTC homes should consider the residents’ preferences, the social benefits of group exercise, and the feasibility of individualizing exercises for the complex needs of residents in LTC in large group settings.
Finally, which professionals should deliver the exercise program is also uncertain. Only 3 of the positive trials in this paper described the professional delivering the intervention, with one being research staff, one geriatric nurses, and one exercise assistants supported by a physiotherapist. We suggest that professionals delivering an exercise program should be trained in exercise planning, delivery, and progression, be familiar with the principles of balance and strength training, and have training in working with older adults in LTC.
Modifications for Physical Impairments
Residents in LTC often have complex health needs, with multiple comorbidities (eg, stroke, Parkinson’s disease, multiple sclerosis) [21]. Modifications of strength and balance exercises may be required to accommodate for physical impairments (eg, hemiplegia, drop foot, freezing gait). For example, if a resident has hemiplegia and cannot fully activate the muscles of one arm, one can do resistance exercises with a dumbbell on the functioning side and active assisted range of motion (ie, the exercise provider assists the resident to achieve full range of motion against gravity) on the hemiparetic side. A resident with Parkinson’s disease who has freezing gait may need visual or rhythmical verbal cues to be able to accomplish standing balance tasks such as altered walking patterns (eg, wide or narrow stepping) [42].
Modifications for Cognitive Impairments
More than 80% of residents in LTC have some degree of cognitive impairment [21]. Cognitive impairment may be the result of stroke, depression, traumatic injuries, medications, and degenerative diseases such as Parkinson’s and Alzheimer’s disease [43]. A common misconception is that residents with cognitive impairment cannot benefit from exercise because they cannot learn new skills and have difficulty following directions. On the contrary, evidence suggests that exercise can improve functional mobility for residents with cognitive impairment [44,45].
Residents with cognitive impairment may require a different approach to facilitate participation in the desired exercises because of difficulty following multi-step directions, responsive behaviors, or increased distractibility [46]. Clear communication is key in improving the quality of interaction for residents with cognitive impairment. The Alzheimer Society of Ontario suggests 10 strategies for communicating with people with dementia [47], and we have provided suggestions of how to apply these communication strategies to the exercise context in LTC (Table 2). Other suggestions for engaging residents with cognitive impairment in strength and balance training include making the exercises functional (eg, ask them to pick something up of the floor to perform a squat, or reach a point on the wall to do calf raises) and playful (eg, toss a ball back and forth or sing a song about rowing to promote weight shifting) [48].
Standing versus Seated Exercises
Residents may not be able to participate in standing exercises for several reasons: perhaps the resident cannot stand or has severe balance impairments and a high falls risk; the resident may have poor insight into which exercises are safe to perform in standing versus sitting; or there may be limited supervision of a large group exercise class where the risk of falls is a concern. If balance impairments are a concern, where the risk of injury or falling while completing exercises in standing outweighs the benefit of doing the exercises, then seated exercises are appropriate. However, when residents are able, we recommend encouraging some or all exercises in standing, to facilitate carry over of strength gains into functional tasks such as being able to rise from a chair and walking. A recent study, comparing standing versus seated exercises for community dwelling older adults, saw greater functional gains for those who completed the standing exercises [49]. Therefore, strength and balance exercises should be performed in standing, where appropriate.
Resident-Centered Exercise for Falls Prevention
Putting the resident at the center of falls prevention is important. Previous work has found that older adults have expressed a strong preference for care that transcends traditional biomedical care and that values efficiency, consistency, and hierarchical decision making [50]. On the contrary, resident-centered care emphasizes well-being and quality of life as defined by the resident, values giving residents greater control over the nature of services they receive, and respects their rights to be involved in every day decision making [51,52]. Indeed, residents may choose to engage in risky behaviors that increase their risk of falls but also increases their quality of life. Previous work has found disconnects between residents’ perceived frailty and the potential ability of protective devices to prevent adverse events, such as falls and fractures [53]. Additionally, one study identified that older residents feared being labelled, so instead hid impairments and chose to refuse assistance and assistive devices [54]. For example, a resident with impaired balance and gait may choose to walk independently when they have been deemed as requiring a gait aid (eg, rollator walker). However, they may value walking without a gait aid and accept the increased risk of falling. Therefore, it is essential to find the delicate balance between respecting a resident’s right to make their own decisions and preventing adverse events, such as falls [52]. An example of this would be respecting a resident’s right to refuse to attend exercise programming even though the team may think they can benefit from strength and balance training.
There is limited evidence around falls prevention and resident-centered care. A recent systematic review [55] revealed that resident-centered care may increase falls rates [56,57]. However, the authors of the review attributed the increase in falls to differences in frailty between the control and intervention group [56], and to environmental factors (eg, slippery flooring material, lack of handrails) [57]. Additionally, these trials did not include an exercise program as part of the resident-centered care program. On the other hand, resident-centered care has been associated with reduction of boredom, helplessness, and depression [58,59]. Most studies included in the review were quasi-experimental, which significantly limits the evidence quality [55]. At this point in time, the evidence suggests that resident-centered care is important for mood and quality of life but may have a negative or no effect on reducing falls.
Multifactorial Falls Prevention Programs
While there are mixed results about the effect of exercise as a single intervention for reducing falls for residents in LTC, the literature clearly supports exercise as part of a multifactorial falls prevention program [17,20,60–62]. A 2015 umbrella review [62] of meta-analyses of randomized controlled trials of falls prevention interventions in LTC concluded that multifactorial interventions were the most effective at preventing falls in LTC. Additionally, recently developed recommendations for fracture prevention in LTC [61] suggest that balance, strength, and functional training should be included for residents who are not at high risk of fracture, while for those at high risk, exercise should be provided as part of a multifactorial falls prevention intervention. Clinicians must therefore incorporate elements aside from exercise into their falls prevention strategies. Interventions that have shown positive effects on reducing falls when delivered as part of multifactorial interventions include: staff and resident education [31,35,37], environmental modifications [31,35], supply/repair/provision of assistive devices [30], falls problem-solving conferences [30], urinary incontinence management [29], medication review [30], optician review [31], and cognitive behavioral therapy [32].
Conclusion and Suggestions for Clinical Practice
We suggest incorporating strength and balance exercises as part of a multifactorial falls prevention program for residents in LTC. Balance exercises should be challenging and dynamic (eg, weight shifting). Strength exercises should be of a moderate to high intensity (eg, can complete one to sets of 6 to 8 repetitions) and need to be progressed as the residents’ abilities improve. Residents should participate in strength and balance training on 2 to 3 days per week, for 30- to 45-minute sessions, for at least 6 months. Exercises in standing should be prioritized where appropriate. Exercise could be delivered in a group or individual format, but should consider the residents’ preferences, the social benefits of group exercise, and the feasibility of individualizing exercises for the complex needs of residents in LTC in large group settings. Professionals delivering an exercise program should be trained in exercise planning, delivery, and progression, be familiar with the principles of balance and strength training, and have training in working with older adults in LTC. Exercise programs in LTC should be resident-centered and consider residents’ potential physical and cognitive impairments.
Funding/support: Dr. Giangregorio was supported by grants from the Canadian Frailty Network and Canadian Institutes of Health Research.
1. Harris IA, Yong S, McEvoy L, Thorn L. A prospective study of the effect of nursing home residency on mortality following hip fracture. ANZ J Surg 2010;80:447–50.
2. Ooms ME, Vlasman P, Lips P, et al. The incidence of hip fractures in independent and institutionalized elderly people. Osteoporos Int 1994;4:6–10.
3. Ayoung-Chee P, McIntyre L, Ebel BE, et al. Long-term outcomes of ground-level falls in the elderly. J Trauma Acute Care Surg 2014;76:498–503.
4. Heinrich S, Rapp K, Rissmann U, et al. Cost of falls in old age: a systematic review. Osteoporos Int 2010;21: 891–902.
5. Rubenstein LZ, Josephson KR, Robbins AS. Falls in the nursing home. Ann Intern Med 1994;121:442–51.
6. Hartholt KA, van Beeck EF, Polinder S, et al. Societal consequences of falls in the older population: injuries, healthcare costs, and long-term reduced quality of life. J Trauma
2011;71:748–53.
7. Robinovitch SN, Feldman F, Yang Y, et al. Video capture of the circumstances of falls in elderly people residing in long-term care: an observational study. Lancet 2013;381:
47–54.
8. Rapp K, Becker C, Cameron ID, et al. Epidemiology of falls in residential aged care: analysis of more than 70,000 falls from residents of bavarian nursing homes. J Am Med Dir Assoc 2012;13:187.
9. Büchele G, Becker C, Cameron ID, et al. Predictors of serious consequences of falls in residential aged care: analysis of more than 70,000 falls from residents of Bavarian nursing homes. J Am Med Dir Assoc 2014;15:559–63.
10. McArthur C, Gonzalez DA, Roy E, Giangregorio L. What are the circumstances of falls and fractures in long-term care? Can J Aging / La Rev Can du Vieil 2016;35:491–8.
11. Chin A Paw MJM, van Poppel MNM, van Mechelen W. Effects of resistance and functional-skills training on habitual activity and constipation among older adults living in long-term care facilities: a randomized controlled trial. BMC Geriatr 2006;6:9.
12. Ikezoe T, Asakawa Y, Shima H, et al. Daytime physical activity patterns and physical fitness in institutionalized elderly women: an exploratory study. Arch Gerontol Geriatr 2013;57:221–5.
13. Senior HE, Henwood TR, Beller EM, et al. Prevalence and risk factors of sarcopenia among adults living in nursing homes. Maturitas 2015;82:418–23.
14. Smoliner C, Sieber CC, Wirth R. Prevalence of sarcopenia in geriatric hospitalized patients. J Am Med Dir Assoc 2014;15:267–72.
15. Landi F, Liperoti R, Fusco D, et al. Sarcopenia and mortality among older nursing home residents. J Am Med Dir Assoc 2012;13:121–6.
16. Yalcin A, Aras S, Atmis V, et al. Sarcopenia prevalence and factors associated with sarcopenia in older people living in a nursing home in Ankara Turkey. Geriatr Gerontol Int
2016;16:903–10.
17. Sherrington C, Michaleff ZA, Fairhall N, et al. Exercise to prevent falls in older adults: an updated systematic review and meta-analysis. Br J Sports Med October 2016.
18. Liu C, Latham NK. Progressive resistance strength training for improving physical function in older adults. In: Liu C, ed. Cochrane Database Syst Rev;2009:CD002759.
19. Cameron ID, Gillespie LD, Robertson MC, et al. Interventions for preventing falls in older people in care facilities and hospitals. Cochrane Database Syst Rev;2012:CD005465.
20. Silva RB, Eslick GD, Duque G. Exercise for falls and fracture prevention in long term care facilities: a systematic review and meta-analysis. J Am Med Dir Assoc 2013;14:685–9.
21. Hirdes JP, Mitchell L, Maxwell CJ, White N. Beyond the “iron lungs of gerontology”: Using evidence to shape the future of nursing homes in Canada. Can J Aging 2011;30: 371–90.
22. Benjamin K, Edwards N, Guitard P, et al. Factors that influence physical activity in long-term care: Perspectives of residents, staff, and significant others. Can J Aging 2011;30:247–58.
23. Benjamin K, Edwards N, Ploeg J, Legault F. Barriers to physical activity and restorative care for residents in long-term care: A review of the literature. J Aging Phys Act 2014;22:154–65.
24. American College of Sports Medicine. ACSM’s guidelines for exercise testing and prescription. 9th ed. American College of Sports Medicine; 2013.
25. Prevention of Falls Network Europe. Prevention of Falls Network Europe. Accessed 27 Nov 2017 at www.profane.eu.org/.
26. Sihvonen SE, Sipilä S, Era PA. Changes in postural balance in frail elderly women during a 4-week visual feedback training: a randomized controlled trial. Gerontology 2004;50:87–95.
27. Sakamoto K, Nakamura T, Hagino H, et al. Effects of unipedal standing balance exercise on the prevention of falls and hip fracture among clinically defined high-risk elderly individuals: a randomized controlled trial. J Orthop Sci 2006;11:467–72.
28. Shimada H, Obuchi S, Furuna T, Suzuki T. New intervention program for preventing falls among frail elderly people: the effects of perturbed walking exercise using a bilateral separated treadmill. Am J Phys Med Rehabil 2004;83:493–9.
29. Schnelle JF, Kapur K, Alessi C, et al. Does an exercise and incontinence intervention save healthcare costs in a nursing home population? J Am Geriatr Soc 2003;51:161–8.
30. Jensen J, Lundin-Olsson L, Nyberg L, Gustafson Y. Fall and injury prevention in older people living in residential care facilities: A cluster randomized trial. Ann Intern Med 2002;136:733–41.
31. Dyer CAE. Falls prevention in residential care homes: a randomised controlled trial. Age Ageing 2004;33:596–602.
32. Huang T-T, Chung M-L, Chen F-R, Chin Y-F, Wang B-H. Evaluation of a combined cognitive-behavioural and exercise intervention to manage fear of falling among elderly residents in nursing homes. Aging Ment Health 2016;20:2–12.
33. Sihvonen S, Sipilä S, Taskinen S, Era P. Fall incidence in frail older women after individualized visual feedback-based balance training. Gerontology 2004;50:411–6.
34. Lord SR, Castell S, Corcoran J, et al. The effect of group exercise on physical functioning and falls in frail older people living in retirement villages: a randomized, controlled trial. J Am Geriatr Soc 2003;51:1685–92.
35. Becker C, Kron M, Lindemann U, et al. Effectiveness of a multifaceted intervention on falls in nursing home residents. J Am Geriatr Soc 2003;51:306–13.
36. Becker C, Kron M, Lindemann U, et al. Effectiveness of a multifaceted intervention on falls in nursing home residents. J Am Geriatr Soc 2003;51:306–13.
37. Jensen J, Lundin-Olsson L, Nyberg L, Gustafson Y. Fall and injury prevention in older people living in residential care facilities. A cluster randomized trial. Ann Intern Med 2002;136:733–41.
38. Moreland JD, Richardson JA, Goldsmith CH, Clase CM. Muscle weakness and falls in older adults: a systematic review and meta-analysis. J Am Geriatr Soc 2004;52: 1121–9.
39. Chodzko-Zajko WJ, Proctor DN, Fiatarone Singh MA, et al. Exercise and physical activity for older adults. Med Sci Sport Exerc 2009;41:1510–30.
40. de Souto Barreto P, Morley JE, Chodzko-Zajko W, et al. Recommendations on physical activity and exercise for older adults living in long-term care facilities: a taskforce report. J Am Med Dir Assoc 2016;17:381–92.
41. American College of Sports Medicine. Progression models in resistance training for healthy adults. Med Sci Sport Exerc 2009;41:687–708.
42. Fietzek UM, Schroeteler FE, Ziegler K, et al. Randomized cross-over trial to investigate the efficacy of a two-week physiotherapy programme with repetitive exercises of cueing to reduce the severity of freezing of gait in patients with Parkinson’s disease. Clin Rehabil 2014;28:902–11.
43. Patterson C, Feightner J, Garcia A, MacKnight C. General risk factors for dementia: A systematic evidence review. Alzheimer Dement 2007;3:341–7.
44. Roach KE, Tappen RM, Kirk-Sanchez N, et al. A randomized controlled trial of an activity specific exercise program for individuals with alzheimer disease in long-term care settings. J Geriatr Phys Ther 2011;34:50–6.
45. Christofoletti G, Oliani MM, Gobbi S, et al. A controlled clinical trial on the effects of motor intervention on balance and cognition in institutionalized elderly patients with dementia. Clin Rehabil 2008;22:618–26.
46. van Alphen HJM, Hortobágyi T, van Heuvelen MJG. Barriers, motivators, and facilitators of physical activity in dementia patients: A systematic review. Arch Gerontol Geriatr 2016;66:109–18.
47. Alzheimer Society of Ontario. Rethink Dementia. Accessed 18 Sep 2017 at http://rethinkdementia.ca/.
48. Roach KE, Tappen RM, Kirk-Sanchez N, et al. A randomized controlled trial of an activity specific exercise program for individuals with Alzheimer disease in long-term care settings. J Geriatr Phys Ther 2011;34:50–6.
49. Brach JS, Perera S, Gilmore S, et al. Effectiveness of a timing and coordination group exercise program to improve mobility in community-dwelling older adults. JAMA Intern Med August 2017.
50. Rosher RB, Robinson S. Impact of the Eden alternative on family satisfaction. J Am Med Dir Assoc 2005;6:189–93.
51. Crandall LG, White DL, Schuldheis S, Talerico KA. Initiating person-centered care practices in long-term care facilities. J Gerontol Nurs 2007;33:47–56.
52. Sims-Gould J, McKay HA, Feldman F, et al. Autonomy, choice, patient-centered care, and hip protectors: the experience of residents and staff in long-term care. J Appl Gerontol 2014;33:690–709.
53. Robinovitch SN, Cronin T. Perception of postural limits in elderly nursing home and day care participants. J Gerontol A Biol Sci Med Sci 1999;54:B124-30.
54. Perkins MM, Ball MM, Whittington FJ, Hollingsworth C. Relational autonomy in assisted living: a focus on diverse care settings for older adults. J Aging Stud 2012;26:214–25.
55. Brownie S, Nancarrow S. Effects of person-centered care on residents and staff in aged-care facilities: a systematic review. Clin Interv Aging 2013;8:1–10.
56. Coleman MT, Looney S, O’Brien J, et al. The Eden Alternative: findings after 1 year of implementation. J Gerontol A Biol Sci Med Sci 2002;57:M422–7.
57. Chenoweth L, King MT, Jeon Y-H, et al. Caring for Aged Dementia Care Resident Study (CADRES) of personcentred care, dementia-care mapping, and usual care in dementia: a cluster-randomised trial. Lancet Neurol 2009;8: 317–25.
58. Bergman-Evans B. Beyond the basics. Effects of the Eden Alternative model on quality of life issues. J Gerontol Nurs 2004;30:27–34.
59. Robinson SB, Rosher RB. Tangling with the barriers to culture change: creating a resident-centered nursing home environment. J Gerontol Nurs 2006;32:19–25.
60. Cameron ID, Gillespie LD, Robertson MC, et al. Interventions for preventing falls in older people in care facilities and hospitals. Cochrane Database Syst Rev 2012;12.
61. Papaioannou A, Santesso N, Morin SN, et al. Recommendations for preventing fracture in long-term care. Can Med Assoc J 2015;187:1135–44.
62. Stubbs B, Denkinger MD, Brefka S, Dallmeier D. What works to prevent falls in older adults dwelling in long term care facilities and hospitals? An umbrella review of meta-analyses of randomised controlled trials. Maturitas 2015;81:335–42.
From the Geriatric Education and Research in Aging Sciences Centre, McMaster University Hamilton, ON (Dr. McArthur) and the University of Waterloo and Research Institute for Aging, Waterloo, ON (Dr. Giangregorio), Canada
Abstract
- Objective: To synthesize the available literature on exercise and falls reduction interventions in long-term care (LTC) and provide practical information for clinicians and other decision makers.
- Methods: Review of positive trials included in systematic reviews.
- Results: Falls are a major concern for residents, families, clinicians, and decision-makers in LTC. Exercise is recommended as part of a multifactorial falls prevention program for residents in LTC. Strength and balance exercises should be incorporated into the multifactorial falls prevention program. They should be challenging and progressed as the residents’ abilities improve. Evidence suggests that exercises should be completed 2 to 3 times per week for a period longer than 6 months. Exercise programs in LTC should be resident-centered and should consider residents’ potential physical and cognitive impairments. Exercises in standing should be prioritized where appropriate.
- Conclusion: Appropriately challenging and progressive strength and balance exercises should be included in a multifactorial falls prevention program for residents in LTC.
Key words: long-term care; nursing homes; falls reduction; exercise.
Falls are common in long-term care (LTC) homes: the estimated falls rate is 1.5 falls per bed per year, which is 3 times greater than that for older adults living in the community [1]. Falls can have significant consequences for residents in LTC, including functional disability, fractures, pain, reduced quality of life, and death [1–6]. Indeed, 25% of residents who are hospitalized after a fall die within 1 year [3]. Consequently, falls prevention programs are important to help in reducing falls and averting the associated negative consequences.
Exercise may address the circumstances and physical deconditioning that often contribute to falls in LTC residents. Weight shifting [7], walking, and transferring [8–10], are common activities that precede falls, suggesting that balance, gait, and functional mobility training may be possible targets for prevention. Additionally, it is estimated that LTC residents spend three quarters of their waking time in sedentary activities [11,12] and have a high prevalence of sarcopenia [13–16]. Challenging balance training and resistance exercise are well-known intervention for reducing falls [17] and improving muscle strength for community-dwelling older adults [18]. However, evidence around balance and strength training for preventing falls in LTC is mixed [17,19,20], and careful planning and modification of exercises is necessary to meet the needs of LTC residents.
Residents in LTC are often medically complex, with multiple comorbidities [21] that can affect their ability to meaningfully participate in exercise. In Canada, 56.3% of residents have a diagnosis of Alzheimer’s or other dementias, 25.0% have diabetes, 14.4% have chronic obstructive pulmonary disease, and 21.2% have experienced a stroke [21]. Residents also often have significant functional impairments. For example, 97% of residents require assistance with basic activities of daily living [21]. Therefore, the lack of effect of exercise as a single falls prevention strategy observed in previous studies may be because the often complex, multimorbid LTC population likely requires a multifactorial approach to fall prevention [17]. Additionally, organizational aspects of LTC homes (eg, specific funds dedicated to employing exercise professionals and to support exercise programming) can affect residents’ engagement in exercise [22,23]. Subsequently, prescribing exercises in the LTC context must consider both resident characteristics and organizational features of the LTC home (eg, professionals available to support exercise programming).
A comprehensive exercise prescription describes the elements of an appropriate exercise program to facilitate implementation of that program. The exercise prescription should include a description of the type (eg, balance, strength) and intensity of exercises (eg, subjective or objective measurement of how hard the resident is working) included in the program [24]. The prescription should also include a description of the dose of exercise: frequency of exercise participation (eg, 2 days per week), duration of individual exercise sessions (eg, 30-minute sessions), and duration of exercise program (eg, 12-week program) [24]. Lastly, the prescription should describe the setting of the exercise program (eg, group or individual basis) and the professional delivering the program (eg, physiotherapist, fitness instructor) [24].
Therefore, the objectives of this article are to (1) synthesize studies demonstrating a positive effect of exercise on reducing falls for residents in LTC; (2) provide an overview of the principles of balance and strength training to guide clinicians in designing appropriate exercise prescription; and (3) make suggestions for clinical practice regarding an appropriate strength and balance exercise protocol by considering the influence of the LTC context.
Methods
To provide clinicians and other policy-makers with a description of which balance and strength exercises may be effective for preventing falls, we synthesized trials that demonstrated a positive effect on reducing falls or falls risk for residents in LTC. Studies were identified through a database search for systematic reviews in PubMed, Ovid, and Google Scholar using the keywords falls, long-term care, nursing homes, exercise, strength, balance, and systematic reviews. Our purpose was to provide practical information on what works to prevent falls through balance and strength training for residents in LTC rather than to evaluate the available evidence. Therefore, only positive trials from systematic reviews were discussed, as we wanted to present exercises that seem to have a positive effect on decreasing falls. Positive trials were defined as those included in identified systematic reviews with a risk or rate ratio and confidence intervals below 1.0.
We first provide an overview of the conclusions of the systematic reviews found in our search. Next, for each positive trial we describe the following elements of the exercise component of the intervention: frequency, time of sessions, length of program, intensity, type of exercise including a description of the specific exercises performed, whether the intervention was delivered in a group or on an individual basis, the professional delivering the intervention, and any other features of the intervention aside from the exercise component. We used the ProFaNE taxonomy definitions [25] to identify and describe each element of the exercise interventions. Frequency is the number of times per week that residents engage in sessions, time of sessions is the amount allocated to each exercise session, duration of program is how long the resident participates in the exercise program, and intensity is the subjective or objective report of how hard the resident is working [25]. The types of exercises described were those targeting balance defined as “...the efficient transfer of bodyweight from one part of the body to another or challenges specific aspects of the balance systems (eg, vestibular system)” [25], and strength defined as “...contracting the muscles against a resistance to ‘overload’ and bring about a training effect in the muscular system” [25]. Strength could be either an external resistance (eg, dumbbell) or using body weight against gravity (eg, squat) [25].
Results
We found 3 systematic reviews that include exercise programs to reduce falls in LTC homes [17,19,20]. Overall, evidence suggests that exercise should be included as part of a multifactorial falls prevention program for residents in LTC. There is limited evidence that exercise as a single intervention prevents falls, and some trials, albeit underpowered, even demonstrate an increased risk of falling in the exercise group compared to control [19]. With regards to specific exercise programs, the Cochrane review found that gait, balance, and functional training decrease the rate of falls but not the risk of falling [26–28], and the 2013 review by Silva et al [20] concluded that combined exercise programs (ie, multiple types of exercise) that include balance tasks, are completed frequently (2–3 times per week), and over a long term (greater than 6 months) were most effective at preventing falls [20].
A more recent systematic review and meta-analysis [17] also concluded that there was no evidence that exercise as a single intervention can prevent falls for residents in LTC. Table 1 provides a description of the exercise component of the seven positive trials [29–35] that were included in the 3 systematic reviews we identified in our search.
Type of Exercise
Balance Exercises
There were 4 positive trials that included balance exercises in their intervention [31,33–35]. Trials that had a positive effect on reducing falls and included balance training employed mostly dynamic balance exercises in standing (Table 1). However, only 2 of the 7 trials provided a detailed description of their balance exercises (Table 1) [26,34]. Jensen et al [30] and Dyer et al [31] did not include a description of the balance training performed but stated that balance was part of the multicomponent exercise program. Becker et al [36] stated that participants performed standing balance exercises, while Schnelle et al [39] and Huang et al [32] did not include balance training in their trial.
Strength Exercises
Of the 7 positive trials included in this review, 6 included strength exercises [29–32,34,35]. The strength activities used in trials where exercise had a positive effect on decreasing falls included functional activities [29,31] and progressive resistance training [31,36] (Table 1). Functional activities are those that replicate what a resident might be required to do in their everyday life, such as performing sit-to-stands out of a chair (Figure)
Frequency, Time of Sessions, Duration of Program
In our description of positive trials, exercise was performed on 2 to 3 days per week for 20 to 75 minutes per session, for periods ranging from 4 to 52 weeks (Table 1).
Intensity
For the trials including balance exercises, one trial described the intensity as resident-specific [37] and another as individualized [33]. Two studies did not describe the intensity of their balance exercises [31,34]. The intensity of strength exercises included in the positive trials was individualized for one of the trial [29]. Two trials had participants complete 2 to 3 sets of 10 repetitions [32,35], with one indicating an intensity of 12–13 or “somewhat difficult” on the Borg Rating of Perceived Exertion Scale [32] and the other using a 10-rep max [35]. Two studies described their strength exercises as progressive [31,37], and one at a moderate to high intensity [30]. Lord et al prescribed 30 repetitions of each strength exercise [34].
Delivery of Intervention
Exercise was delivered in a group setting for 4 of the trials [31,32,34,36], individually for 2 of the trials [26,29], and the setting was not described for one of the trials (Table 1) [30]. Finally, only 3 of the 7 articles reported the professional delivering the intervention: one was research staff [29], one was geriatric nurses [32], and one was exercise assistants supported by a physiotherapist [31].
Discussion
There is limited evidence to support the use of strength and balance exercise as a single intervention to prevent falls in LTC. However, exercise should be included as part of a multifactorial falls prevention program. Trials that had a positive effect on decreasing falls training used dynamic balance exercises in standing, functional training, and progressive resistance training on 2 to 3 days per week, for 20 to 75 minutes per session, over 4 to 52 weeks. The intensity of balance exercises was individualized, and strength exercises were described as somewhat difficult or performed at a moderate to high intensity. Exercise was performed in a group or individually, and was delivered by research staff, geriatric nurses, exercise assistants supervised by physiotherapists, or more frequently, it was not reported who delivered the intervention.
Balance Training
Our work suggests that standing, dynamic balance exercises may be best to decrease falls. Example balance exercises include reducing the base of support (eg, standing with feet together instead of apart, or tandem with one foot in front), moving the center of gravity and control body position while standing (eg, reaching, weight shifting, stepping up or down), and standing without using arms for support or reducing reliance on the upper limbs for support (eg, use one hand on a handrail instead of two, or two fingers instead of the whole hand) [17]. It is well established that balance training programs, especially those including challenging exercises, can prevent falls in community-dwelling older adults [17]. However, the relationship is not as clear in LTC.
Strength Training
Reduced muscle strength has been identified as an important risk factor for falls [38]. There are also many psychological and metabolic benefits to strength training [39]. To induce change in muscular strength, resistance exercises need to be challenging and progressive. Our work suggests that strength training that is effective at decreasing falls is functional and progressive, and is completed at a moderate to high intensity. A resident should be able to do a strength exercise for one to two sets of 6 to 8 repetitions before being fatigued [40]. Once the resident can complete two sets of 13 to 15 repetitions easily the exercise should be progressed. Residents who are particularly deconditioned may need to begin with lower intensity strength exercises (eg, only do one set, with a lower resistance and progress to a higher resistance) [40]. Residents should perform resistance exercises for all major muscle groups [40]. Progression could include increasing the number of sets (eg, increase from one to two sets), the resistance (eg, holding dumbbells while squatting), or the intensity of the exercise (eg, squat lower or faster) [41].
Implementing Exercise Programs in LTC
Implementation of exercise programs into LTC homes should consider the dose of exercise (eg, time and frequency of sessions, duration of program), if they are delivered in a group or individual setting, and who is delivering them. First, trials included in this paper suggest that strength and balance exercises to prevent falls were delivered 2 to 3 times per week, for 20 to 75 minutes per session, over 4 to 52 weeks. Second, previous work has established that exercise programs delivered on 2 to 3 days per week over a period of more than 6 months are most effective at reducing falls in LTC [20]. Finally, a recent task force report from an international group of clinician researchers in LTC recommends twice weekly exercise sessions lasting 35 to 45 minutes each [40]. Therefore, strength and balance exercises to prevent falls in LTC should be delivered at least twice per week, for at least 20 minutes, for greater than 6 weeks’ duration.
Whether exercise should be performed in a group or individual setting remains unclear. Two of the 6 positive trials in this paper were completed individually, while 3 were in a group. The aforementioned task force also recommended that every resident who does not have contraindications to exercise must have an individualized exercise program as part of their health care plan [40]. However, whether the exercise program is provided on an individual basis or in a group setting was not delineated. Indeed, there are currently no recommendations concerning prioritizing group or individual exercise programs. Therefore, exercise programs being implemented into LTC homes should consider the residents’ preferences, the social benefits of group exercise, and the feasibility of individualizing exercises for the complex needs of residents in LTC in large group settings.
Finally, which professionals should deliver the exercise program is also uncertain. Only 3 of the positive trials in this paper described the professional delivering the intervention, with one being research staff, one geriatric nurses, and one exercise assistants supported by a physiotherapist. We suggest that professionals delivering an exercise program should be trained in exercise planning, delivery, and progression, be familiar with the principles of balance and strength training, and have training in working with older adults in LTC.
Modifications for Physical Impairments
Residents in LTC often have complex health needs, with multiple comorbidities (eg, stroke, Parkinson’s disease, multiple sclerosis) [21]. Modifications of strength and balance exercises may be required to accommodate for physical impairments (eg, hemiplegia, drop foot, freezing gait). For example, if a resident has hemiplegia and cannot fully activate the muscles of one arm, one can do resistance exercises with a dumbbell on the functioning side and active assisted range of motion (ie, the exercise provider assists the resident to achieve full range of motion against gravity) on the hemiparetic side. A resident with Parkinson’s disease who has freezing gait may need visual or rhythmical verbal cues to be able to accomplish standing balance tasks such as altered walking patterns (eg, wide or narrow stepping) [42].
Modifications for Cognitive Impairments
More than 80% of residents in LTC have some degree of cognitive impairment [21]. Cognitive impairment may be the result of stroke, depression, traumatic injuries, medications, and degenerative diseases such as Parkinson’s and Alzheimer’s disease [43]. A common misconception is that residents with cognitive impairment cannot benefit from exercise because they cannot learn new skills and have difficulty following directions. On the contrary, evidence suggests that exercise can improve functional mobility for residents with cognitive impairment [44,45].
Residents with cognitive impairment may require a different approach to facilitate participation in the desired exercises because of difficulty following multi-step directions, responsive behaviors, or increased distractibility [46]. Clear communication is key in improving the quality of interaction for residents with cognitive impairment. The Alzheimer Society of Ontario suggests 10 strategies for communicating with people with dementia [47], and we have provided suggestions of how to apply these communication strategies to the exercise context in LTC (Table 2). Other suggestions for engaging residents with cognitive impairment in strength and balance training include making the exercises functional (eg, ask them to pick something up of the floor to perform a squat, or reach a point on the wall to do calf raises) and playful (eg, toss a ball back and forth or sing a song about rowing to promote weight shifting) [48].
Standing versus Seated Exercises
Residents may not be able to participate in standing exercises for several reasons: perhaps the resident cannot stand or has severe balance impairments and a high falls risk; the resident may have poor insight into which exercises are safe to perform in standing versus sitting; or there may be limited supervision of a large group exercise class where the risk of falls is a concern. If balance impairments are a concern, where the risk of injury or falling while completing exercises in standing outweighs the benefit of doing the exercises, then seated exercises are appropriate. However, when residents are able, we recommend encouraging some or all exercises in standing, to facilitate carry over of strength gains into functional tasks such as being able to rise from a chair and walking. A recent study, comparing standing versus seated exercises for community dwelling older adults, saw greater functional gains for those who completed the standing exercises [49]. Therefore, strength and balance exercises should be performed in standing, where appropriate.
Resident-Centered Exercise for Falls Prevention
Putting the resident at the center of falls prevention is important. Previous work has found that older adults have expressed a strong preference for care that transcends traditional biomedical care and that values efficiency, consistency, and hierarchical decision making [50]. On the contrary, resident-centered care emphasizes well-being and quality of life as defined by the resident, values giving residents greater control over the nature of services they receive, and respects their rights to be involved in every day decision making [51,52]. Indeed, residents may choose to engage in risky behaviors that increase their risk of falls but also increases their quality of life. Previous work has found disconnects between residents’ perceived frailty and the potential ability of protective devices to prevent adverse events, such as falls and fractures [53]. Additionally, one study identified that older residents feared being labelled, so instead hid impairments and chose to refuse assistance and assistive devices [54]. For example, a resident with impaired balance and gait may choose to walk independently when they have been deemed as requiring a gait aid (eg, rollator walker). However, they may value walking without a gait aid and accept the increased risk of falling. Therefore, it is essential to find the delicate balance between respecting a resident’s right to make their own decisions and preventing adverse events, such as falls [52]. An example of this would be respecting a resident’s right to refuse to attend exercise programming even though the team may think they can benefit from strength and balance training.
There is limited evidence around falls prevention and resident-centered care. A recent systematic review [55] revealed that resident-centered care may increase falls rates [56,57]. However, the authors of the review attributed the increase in falls to differences in frailty between the control and intervention group [56], and to environmental factors (eg, slippery flooring material, lack of handrails) [57]. Additionally, these trials did not include an exercise program as part of the resident-centered care program. On the other hand, resident-centered care has been associated with reduction of boredom, helplessness, and depression [58,59]. Most studies included in the review were quasi-experimental, which significantly limits the evidence quality [55]. At this point in time, the evidence suggests that resident-centered care is important for mood and quality of life but may have a negative or no effect on reducing falls.
Multifactorial Falls Prevention Programs
While there are mixed results about the effect of exercise as a single intervention for reducing falls for residents in LTC, the literature clearly supports exercise as part of a multifactorial falls prevention program [17,20,60–62]. A 2015 umbrella review [62] of meta-analyses of randomized controlled trials of falls prevention interventions in LTC concluded that multifactorial interventions were the most effective at preventing falls in LTC. Additionally, recently developed recommendations for fracture prevention in LTC [61] suggest that balance, strength, and functional training should be included for residents who are not at high risk of fracture, while for those at high risk, exercise should be provided as part of a multifactorial falls prevention intervention. Clinicians must therefore incorporate elements aside from exercise into their falls prevention strategies. Interventions that have shown positive effects on reducing falls when delivered as part of multifactorial interventions include: staff and resident education [31,35,37], environmental modifications [31,35], supply/repair/provision of assistive devices [30], falls problem-solving conferences [30], urinary incontinence management [29], medication review [30], optician review [31], and cognitive behavioral therapy [32].
Conclusion and Suggestions for Clinical Practice
We suggest incorporating strength and balance exercises as part of a multifactorial falls prevention program for residents in LTC. Balance exercises should be challenging and dynamic (eg, weight shifting). Strength exercises should be of a moderate to high intensity (eg, can complete one to sets of 6 to 8 repetitions) and need to be progressed as the residents’ abilities improve. Residents should participate in strength and balance training on 2 to 3 days per week, for 30- to 45-minute sessions, for at least 6 months. Exercises in standing should be prioritized where appropriate. Exercise could be delivered in a group or individual format, but should consider the residents’ preferences, the social benefits of group exercise, and the feasibility of individualizing exercises for the complex needs of residents in LTC in large group settings. Professionals delivering an exercise program should be trained in exercise planning, delivery, and progression, be familiar with the principles of balance and strength training, and have training in working with older adults in LTC. Exercise programs in LTC should be resident-centered and consider residents’ potential physical and cognitive impairments.
Funding/support: Dr. Giangregorio was supported by grants from the Canadian Frailty Network and Canadian Institutes of Health Research.
From the Geriatric Education and Research in Aging Sciences Centre, McMaster University Hamilton, ON (Dr. McArthur) and the University of Waterloo and Research Institute for Aging, Waterloo, ON (Dr. Giangregorio), Canada
Abstract
- Objective: To synthesize the available literature on exercise and falls reduction interventions in long-term care (LTC) and provide practical information for clinicians and other decision makers.
- Methods: Review of positive trials included in systematic reviews.
- Results: Falls are a major concern for residents, families, clinicians, and decision-makers in LTC. Exercise is recommended as part of a multifactorial falls prevention program for residents in LTC. Strength and balance exercises should be incorporated into the multifactorial falls prevention program. They should be challenging and progressed as the residents’ abilities improve. Evidence suggests that exercises should be completed 2 to 3 times per week for a period longer than 6 months. Exercise programs in LTC should be resident-centered and should consider residents’ potential physical and cognitive impairments. Exercises in standing should be prioritized where appropriate.
- Conclusion: Appropriately challenging and progressive strength and balance exercises should be included in a multifactorial falls prevention program for residents in LTC.
Key words: long-term care; nursing homes; falls reduction; exercise.
Falls are common in long-term care (LTC) homes: the estimated falls rate is 1.5 falls per bed per year, which is 3 times greater than that for older adults living in the community [1]. Falls can have significant consequences for residents in LTC, including functional disability, fractures, pain, reduced quality of life, and death [1–6]. Indeed, 25% of residents who are hospitalized after a fall die within 1 year [3]. Consequently, falls prevention programs are important to help in reducing falls and averting the associated negative consequences.
Exercise may address the circumstances and physical deconditioning that often contribute to falls in LTC residents. Weight shifting [7], walking, and transferring [8–10], are common activities that precede falls, suggesting that balance, gait, and functional mobility training may be possible targets for prevention. Additionally, it is estimated that LTC residents spend three quarters of their waking time in sedentary activities [11,12] and have a high prevalence of sarcopenia [13–16]. Challenging balance training and resistance exercise are well-known intervention for reducing falls [17] and improving muscle strength for community-dwelling older adults [18]. However, evidence around balance and strength training for preventing falls in LTC is mixed [17,19,20], and careful planning and modification of exercises is necessary to meet the needs of LTC residents.
Residents in LTC are often medically complex, with multiple comorbidities [21] that can affect their ability to meaningfully participate in exercise. In Canada, 56.3% of residents have a diagnosis of Alzheimer’s or other dementias, 25.0% have diabetes, 14.4% have chronic obstructive pulmonary disease, and 21.2% have experienced a stroke [21]. Residents also often have significant functional impairments. For example, 97% of residents require assistance with basic activities of daily living [21]. Therefore, the lack of effect of exercise as a single falls prevention strategy observed in previous studies may be because the often complex, multimorbid LTC population likely requires a multifactorial approach to fall prevention [17]. Additionally, organizational aspects of LTC homes (eg, specific funds dedicated to employing exercise professionals and to support exercise programming) can affect residents’ engagement in exercise [22,23]. Subsequently, prescribing exercises in the LTC context must consider both resident characteristics and organizational features of the LTC home (eg, professionals available to support exercise programming).
A comprehensive exercise prescription describes the elements of an appropriate exercise program to facilitate implementation of that program. The exercise prescription should include a description of the type (eg, balance, strength) and intensity of exercises (eg, subjective or objective measurement of how hard the resident is working) included in the program [24]. The prescription should also include a description of the dose of exercise: frequency of exercise participation (eg, 2 days per week), duration of individual exercise sessions (eg, 30-minute sessions), and duration of exercise program (eg, 12-week program) [24]. Lastly, the prescription should describe the setting of the exercise program (eg, group or individual basis) and the professional delivering the program (eg, physiotherapist, fitness instructor) [24].
Therefore, the objectives of this article are to (1) synthesize studies demonstrating a positive effect of exercise on reducing falls for residents in LTC; (2) provide an overview of the principles of balance and strength training to guide clinicians in designing appropriate exercise prescription; and (3) make suggestions for clinical practice regarding an appropriate strength and balance exercise protocol by considering the influence of the LTC context.
Methods
To provide clinicians and other policy-makers with a description of which balance and strength exercises may be effective for preventing falls, we synthesized trials that demonstrated a positive effect on reducing falls or falls risk for residents in LTC. Studies were identified through a database search for systematic reviews in PubMed, Ovid, and Google Scholar using the keywords falls, long-term care, nursing homes, exercise, strength, balance, and systematic reviews. Our purpose was to provide practical information on what works to prevent falls through balance and strength training for residents in LTC rather than to evaluate the available evidence. Therefore, only positive trials from systematic reviews were discussed, as we wanted to present exercises that seem to have a positive effect on decreasing falls. Positive trials were defined as those included in identified systematic reviews with a risk or rate ratio and confidence intervals below 1.0.
We first provide an overview of the conclusions of the systematic reviews found in our search. Next, for each positive trial we describe the following elements of the exercise component of the intervention: frequency, time of sessions, length of program, intensity, type of exercise including a description of the specific exercises performed, whether the intervention was delivered in a group or on an individual basis, the professional delivering the intervention, and any other features of the intervention aside from the exercise component. We used the ProFaNE taxonomy definitions [25] to identify and describe each element of the exercise interventions. Frequency is the number of times per week that residents engage in sessions, time of sessions is the amount allocated to each exercise session, duration of program is how long the resident participates in the exercise program, and intensity is the subjective or objective report of how hard the resident is working [25]. The types of exercises described were those targeting balance defined as “...the efficient transfer of bodyweight from one part of the body to another or challenges specific aspects of the balance systems (eg, vestibular system)” [25], and strength defined as “...contracting the muscles against a resistance to ‘overload’ and bring about a training effect in the muscular system” [25]. Strength could be either an external resistance (eg, dumbbell) or using body weight against gravity (eg, squat) [25].
Results
We found 3 systematic reviews that include exercise programs to reduce falls in LTC homes [17,19,20]. Overall, evidence suggests that exercise should be included as part of a multifactorial falls prevention program for residents in LTC. There is limited evidence that exercise as a single intervention prevents falls, and some trials, albeit underpowered, even demonstrate an increased risk of falling in the exercise group compared to control [19]. With regards to specific exercise programs, the Cochrane review found that gait, balance, and functional training decrease the rate of falls but not the risk of falling [26–28], and the 2013 review by Silva et al [20] concluded that combined exercise programs (ie, multiple types of exercise) that include balance tasks, are completed frequently (2–3 times per week), and over a long term (greater than 6 months) were most effective at preventing falls [20].
A more recent systematic review and meta-analysis [17] also concluded that there was no evidence that exercise as a single intervention can prevent falls for residents in LTC. Table 1 provides a description of the exercise component of the seven positive trials [29–35] that were included in the 3 systematic reviews we identified in our search.
Type of Exercise
Balance Exercises
There were 4 positive trials that included balance exercises in their intervention [31,33–35]. Trials that had a positive effect on reducing falls and included balance training employed mostly dynamic balance exercises in standing (Table 1). However, only 2 of the 7 trials provided a detailed description of their balance exercises (Table 1) [26,34]. Jensen et al [30] and Dyer et al [31] did not include a description of the balance training performed but stated that balance was part of the multicomponent exercise program. Becker et al [36] stated that participants performed standing balance exercises, while Schnelle et al [39] and Huang et al [32] did not include balance training in their trial.
Strength Exercises
Of the 7 positive trials included in this review, 6 included strength exercises [29–32,34,35]. The strength activities used in trials where exercise had a positive effect on decreasing falls included functional activities [29,31] and progressive resistance training [31,36] (Table 1). Functional activities are those that replicate what a resident might be required to do in their everyday life, such as performing sit-to-stands out of a chair (Figure)
Frequency, Time of Sessions, Duration of Program
In our description of positive trials, exercise was performed on 2 to 3 days per week for 20 to 75 minutes per session, for periods ranging from 4 to 52 weeks (Table 1).
Intensity
For the trials including balance exercises, one trial described the intensity as resident-specific [37] and another as individualized [33]. Two studies did not describe the intensity of their balance exercises [31,34]. The intensity of strength exercises included in the positive trials was individualized for one of the trial [29]. Two trials had participants complete 2 to 3 sets of 10 repetitions [32,35], with one indicating an intensity of 12–13 or “somewhat difficult” on the Borg Rating of Perceived Exertion Scale [32] and the other using a 10-rep max [35]. Two studies described their strength exercises as progressive [31,37], and one at a moderate to high intensity [30]. Lord et al prescribed 30 repetitions of each strength exercise [34].
Delivery of Intervention
Exercise was delivered in a group setting for 4 of the trials [31,32,34,36], individually for 2 of the trials [26,29], and the setting was not described for one of the trials (Table 1) [30]. Finally, only 3 of the 7 articles reported the professional delivering the intervention: one was research staff [29], one was geriatric nurses [32], and one was exercise assistants supported by a physiotherapist [31].
Discussion
There is limited evidence to support the use of strength and balance exercise as a single intervention to prevent falls in LTC. However, exercise should be included as part of a multifactorial falls prevention program. Trials that had a positive effect on decreasing falls training used dynamic balance exercises in standing, functional training, and progressive resistance training on 2 to 3 days per week, for 20 to 75 minutes per session, over 4 to 52 weeks. The intensity of balance exercises was individualized, and strength exercises were described as somewhat difficult or performed at a moderate to high intensity. Exercise was performed in a group or individually, and was delivered by research staff, geriatric nurses, exercise assistants supervised by physiotherapists, or more frequently, it was not reported who delivered the intervention.
Balance Training
Our work suggests that standing, dynamic balance exercises may be best to decrease falls. Example balance exercises include reducing the base of support (eg, standing with feet together instead of apart, or tandem with one foot in front), moving the center of gravity and control body position while standing (eg, reaching, weight shifting, stepping up or down), and standing without using arms for support or reducing reliance on the upper limbs for support (eg, use one hand on a handrail instead of two, or two fingers instead of the whole hand) [17]. It is well established that balance training programs, especially those including challenging exercises, can prevent falls in community-dwelling older adults [17]. However, the relationship is not as clear in LTC.
Strength Training
Reduced muscle strength has been identified as an important risk factor for falls [38]. There are also many psychological and metabolic benefits to strength training [39]. To induce change in muscular strength, resistance exercises need to be challenging and progressive. Our work suggests that strength training that is effective at decreasing falls is functional and progressive, and is completed at a moderate to high intensity. A resident should be able to do a strength exercise for one to two sets of 6 to 8 repetitions before being fatigued [40]. Once the resident can complete two sets of 13 to 15 repetitions easily the exercise should be progressed. Residents who are particularly deconditioned may need to begin with lower intensity strength exercises (eg, only do one set, with a lower resistance and progress to a higher resistance) [40]. Residents should perform resistance exercises for all major muscle groups [40]. Progression could include increasing the number of sets (eg, increase from one to two sets), the resistance (eg, holding dumbbells while squatting), or the intensity of the exercise (eg, squat lower or faster) [41].
Implementing Exercise Programs in LTC
Implementation of exercise programs into LTC homes should consider the dose of exercise (eg, time and frequency of sessions, duration of program), if they are delivered in a group or individual setting, and who is delivering them. First, trials included in this paper suggest that strength and balance exercises to prevent falls were delivered 2 to 3 times per week, for 20 to 75 minutes per session, over 4 to 52 weeks. Second, previous work has established that exercise programs delivered on 2 to 3 days per week over a period of more than 6 months are most effective at reducing falls in LTC [20]. Finally, a recent task force report from an international group of clinician researchers in LTC recommends twice weekly exercise sessions lasting 35 to 45 minutes each [40]. Therefore, strength and balance exercises to prevent falls in LTC should be delivered at least twice per week, for at least 20 minutes, for greater than 6 weeks’ duration.
Whether exercise should be performed in a group or individual setting remains unclear. Two of the 6 positive trials in this paper were completed individually, while 3 were in a group. The aforementioned task force also recommended that every resident who does not have contraindications to exercise must have an individualized exercise program as part of their health care plan [40]. However, whether the exercise program is provided on an individual basis or in a group setting was not delineated. Indeed, there are currently no recommendations concerning prioritizing group or individual exercise programs. Therefore, exercise programs being implemented into LTC homes should consider the residents’ preferences, the social benefits of group exercise, and the feasibility of individualizing exercises for the complex needs of residents in LTC in large group settings.
Finally, which professionals should deliver the exercise program is also uncertain. Only 3 of the positive trials in this paper described the professional delivering the intervention, with one being research staff, one geriatric nurses, and one exercise assistants supported by a physiotherapist. We suggest that professionals delivering an exercise program should be trained in exercise planning, delivery, and progression, be familiar with the principles of balance and strength training, and have training in working with older adults in LTC.
Modifications for Physical Impairments
Residents in LTC often have complex health needs, with multiple comorbidities (eg, stroke, Parkinson’s disease, multiple sclerosis) [21]. Modifications of strength and balance exercises may be required to accommodate for physical impairments (eg, hemiplegia, drop foot, freezing gait). For example, if a resident has hemiplegia and cannot fully activate the muscles of one arm, one can do resistance exercises with a dumbbell on the functioning side and active assisted range of motion (ie, the exercise provider assists the resident to achieve full range of motion against gravity) on the hemiparetic side. A resident with Parkinson’s disease who has freezing gait may need visual or rhythmical verbal cues to be able to accomplish standing balance tasks such as altered walking patterns (eg, wide or narrow stepping) [42].
Modifications for Cognitive Impairments
More than 80% of residents in LTC have some degree of cognitive impairment [21]. Cognitive impairment may be the result of stroke, depression, traumatic injuries, medications, and degenerative diseases such as Parkinson’s and Alzheimer’s disease [43]. A common misconception is that residents with cognitive impairment cannot benefit from exercise because they cannot learn new skills and have difficulty following directions. On the contrary, evidence suggests that exercise can improve functional mobility for residents with cognitive impairment [44,45].
Residents with cognitive impairment may require a different approach to facilitate participation in the desired exercises because of difficulty following multi-step directions, responsive behaviors, or increased distractibility [46]. Clear communication is key in improving the quality of interaction for residents with cognitive impairment. The Alzheimer Society of Ontario suggests 10 strategies for communicating with people with dementia [47], and we have provided suggestions of how to apply these communication strategies to the exercise context in LTC (Table 2). Other suggestions for engaging residents with cognitive impairment in strength and balance training include making the exercises functional (eg, ask them to pick something up of the floor to perform a squat, or reach a point on the wall to do calf raises) and playful (eg, toss a ball back and forth or sing a song about rowing to promote weight shifting) [48].
Standing versus Seated Exercises
Residents may not be able to participate in standing exercises for several reasons: perhaps the resident cannot stand or has severe balance impairments and a high falls risk; the resident may have poor insight into which exercises are safe to perform in standing versus sitting; or there may be limited supervision of a large group exercise class where the risk of falls is a concern. If balance impairments are a concern, where the risk of injury or falling while completing exercises in standing outweighs the benefit of doing the exercises, then seated exercises are appropriate. However, when residents are able, we recommend encouraging some or all exercises in standing, to facilitate carry over of strength gains into functional tasks such as being able to rise from a chair and walking. A recent study, comparing standing versus seated exercises for community dwelling older adults, saw greater functional gains for those who completed the standing exercises [49]. Therefore, strength and balance exercises should be performed in standing, where appropriate.
Resident-Centered Exercise for Falls Prevention
Putting the resident at the center of falls prevention is important. Previous work has found that older adults have expressed a strong preference for care that transcends traditional biomedical care and that values efficiency, consistency, and hierarchical decision making [50]. On the contrary, resident-centered care emphasizes well-being and quality of life as defined by the resident, values giving residents greater control over the nature of services they receive, and respects their rights to be involved in every day decision making [51,52]. Indeed, residents may choose to engage in risky behaviors that increase their risk of falls but also increases their quality of life. Previous work has found disconnects between residents’ perceived frailty and the potential ability of protective devices to prevent adverse events, such as falls and fractures [53]. Additionally, one study identified that older residents feared being labelled, so instead hid impairments and chose to refuse assistance and assistive devices [54]. For example, a resident with impaired balance and gait may choose to walk independently when they have been deemed as requiring a gait aid (eg, rollator walker). However, they may value walking without a gait aid and accept the increased risk of falling. Therefore, it is essential to find the delicate balance between respecting a resident’s right to make their own decisions and preventing adverse events, such as falls [52]. An example of this would be respecting a resident’s right to refuse to attend exercise programming even though the team may think they can benefit from strength and balance training.
There is limited evidence around falls prevention and resident-centered care. A recent systematic review [55] revealed that resident-centered care may increase falls rates [56,57]. However, the authors of the review attributed the increase in falls to differences in frailty between the control and intervention group [56], and to environmental factors (eg, slippery flooring material, lack of handrails) [57]. Additionally, these trials did not include an exercise program as part of the resident-centered care program. On the other hand, resident-centered care has been associated with reduction of boredom, helplessness, and depression [58,59]. Most studies included in the review were quasi-experimental, which significantly limits the evidence quality [55]. At this point in time, the evidence suggests that resident-centered care is important for mood and quality of life but may have a negative or no effect on reducing falls.
Multifactorial Falls Prevention Programs
While there are mixed results about the effect of exercise as a single intervention for reducing falls for residents in LTC, the literature clearly supports exercise as part of a multifactorial falls prevention program [17,20,60–62]. A 2015 umbrella review [62] of meta-analyses of randomized controlled trials of falls prevention interventions in LTC concluded that multifactorial interventions were the most effective at preventing falls in LTC. Additionally, recently developed recommendations for fracture prevention in LTC [61] suggest that balance, strength, and functional training should be included for residents who are not at high risk of fracture, while for those at high risk, exercise should be provided as part of a multifactorial falls prevention intervention. Clinicians must therefore incorporate elements aside from exercise into their falls prevention strategies. Interventions that have shown positive effects on reducing falls when delivered as part of multifactorial interventions include: staff and resident education [31,35,37], environmental modifications [31,35], supply/repair/provision of assistive devices [30], falls problem-solving conferences [30], urinary incontinence management [29], medication review [30], optician review [31], and cognitive behavioral therapy [32].
Conclusion and Suggestions for Clinical Practice
We suggest incorporating strength and balance exercises as part of a multifactorial falls prevention program for residents in LTC. Balance exercises should be challenging and dynamic (eg, weight shifting). Strength exercises should be of a moderate to high intensity (eg, can complete one to sets of 6 to 8 repetitions) and need to be progressed as the residents’ abilities improve. Residents should participate in strength and balance training on 2 to 3 days per week, for 30- to 45-minute sessions, for at least 6 months. Exercises in standing should be prioritized where appropriate. Exercise could be delivered in a group or individual format, but should consider the residents’ preferences, the social benefits of group exercise, and the feasibility of individualizing exercises for the complex needs of residents in LTC in large group settings. Professionals delivering an exercise program should be trained in exercise planning, delivery, and progression, be familiar with the principles of balance and strength training, and have training in working with older adults in LTC. Exercise programs in LTC should be resident-centered and consider residents’ potential physical and cognitive impairments.
Funding/support: Dr. Giangregorio was supported by grants from the Canadian Frailty Network and Canadian Institutes of Health Research.
1. Harris IA, Yong S, McEvoy L, Thorn L. A prospective study of the effect of nursing home residency on mortality following hip fracture. ANZ J Surg 2010;80:447–50.
2. Ooms ME, Vlasman P, Lips P, et al. The incidence of hip fractures in independent and institutionalized elderly people. Osteoporos Int 1994;4:6–10.
3. Ayoung-Chee P, McIntyre L, Ebel BE, et al. Long-term outcomes of ground-level falls in the elderly. J Trauma Acute Care Surg 2014;76:498–503.
4. Heinrich S, Rapp K, Rissmann U, et al. Cost of falls in old age: a systematic review. Osteoporos Int 2010;21: 891–902.
5. Rubenstein LZ, Josephson KR, Robbins AS. Falls in the nursing home. Ann Intern Med 1994;121:442–51.
6. Hartholt KA, van Beeck EF, Polinder S, et al. Societal consequences of falls in the older population: injuries, healthcare costs, and long-term reduced quality of life. J Trauma
2011;71:748–53.
7. Robinovitch SN, Feldman F, Yang Y, et al. Video capture of the circumstances of falls in elderly people residing in long-term care: an observational study. Lancet 2013;381:
47–54.
8. Rapp K, Becker C, Cameron ID, et al. Epidemiology of falls in residential aged care: analysis of more than 70,000 falls from residents of bavarian nursing homes. J Am Med Dir Assoc 2012;13:187.
9. Büchele G, Becker C, Cameron ID, et al. Predictors of serious consequences of falls in residential aged care: analysis of more than 70,000 falls from residents of Bavarian nursing homes. J Am Med Dir Assoc 2014;15:559–63.
10. McArthur C, Gonzalez DA, Roy E, Giangregorio L. What are the circumstances of falls and fractures in long-term care? Can J Aging / La Rev Can du Vieil 2016;35:491–8.
11. Chin A Paw MJM, van Poppel MNM, van Mechelen W. Effects of resistance and functional-skills training on habitual activity and constipation among older adults living in long-term care facilities: a randomized controlled trial. BMC Geriatr 2006;6:9.
12. Ikezoe T, Asakawa Y, Shima H, et al. Daytime physical activity patterns and physical fitness in institutionalized elderly women: an exploratory study. Arch Gerontol Geriatr 2013;57:221–5.
13. Senior HE, Henwood TR, Beller EM, et al. Prevalence and risk factors of sarcopenia among adults living in nursing homes. Maturitas 2015;82:418–23.
14. Smoliner C, Sieber CC, Wirth R. Prevalence of sarcopenia in geriatric hospitalized patients. J Am Med Dir Assoc 2014;15:267–72.
15. Landi F, Liperoti R, Fusco D, et al. Sarcopenia and mortality among older nursing home residents. J Am Med Dir Assoc 2012;13:121–6.
16. Yalcin A, Aras S, Atmis V, et al. Sarcopenia prevalence and factors associated with sarcopenia in older people living in a nursing home in Ankara Turkey. Geriatr Gerontol Int
2016;16:903–10.
17. Sherrington C, Michaleff ZA, Fairhall N, et al. Exercise to prevent falls in older adults: an updated systematic review and meta-analysis. Br J Sports Med October 2016.
18. Liu C, Latham NK. Progressive resistance strength training for improving physical function in older adults. In: Liu C, ed. Cochrane Database Syst Rev;2009:CD002759.
19. Cameron ID, Gillespie LD, Robertson MC, et al. Interventions for preventing falls in older people in care facilities and hospitals. Cochrane Database Syst Rev;2012:CD005465.
20. Silva RB, Eslick GD, Duque G. Exercise for falls and fracture prevention in long term care facilities: a systematic review and meta-analysis. J Am Med Dir Assoc 2013;14:685–9.
21. Hirdes JP, Mitchell L, Maxwell CJ, White N. Beyond the “iron lungs of gerontology”: Using evidence to shape the future of nursing homes in Canada. Can J Aging 2011;30: 371–90.
22. Benjamin K, Edwards N, Guitard P, et al. Factors that influence physical activity in long-term care: Perspectives of residents, staff, and significant others. Can J Aging 2011;30:247–58.
23. Benjamin K, Edwards N, Ploeg J, Legault F. Barriers to physical activity and restorative care for residents in long-term care: A review of the literature. J Aging Phys Act 2014;22:154–65.
24. American College of Sports Medicine. ACSM’s guidelines for exercise testing and prescription. 9th ed. American College of Sports Medicine; 2013.
25. Prevention of Falls Network Europe. Prevention of Falls Network Europe. Accessed 27 Nov 2017 at www.profane.eu.org/.
26. Sihvonen SE, Sipilä S, Era PA. Changes in postural balance in frail elderly women during a 4-week visual feedback training: a randomized controlled trial. Gerontology 2004;50:87–95.
27. Sakamoto K, Nakamura T, Hagino H, et al. Effects of unipedal standing balance exercise on the prevention of falls and hip fracture among clinically defined high-risk elderly individuals: a randomized controlled trial. J Orthop Sci 2006;11:467–72.
28. Shimada H, Obuchi S, Furuna T, Suzuki T. New intervention program for preventing falls among frail elderly people: the effects of perturbed walking exercise using a bilateral separated treadmill. Am J Phys Med Rehabil 2004;83:493–9.
29. Schnelle JF, Kapur K, Alessi C, et al. Does an exercise and incontinence intervention save healthcare costs in a nursing home population? J Am Geriatr Soc 2003;51:161–8.
30. Jensen J, Lundin-Olsson L, Nyberg L, Gustafson Y. Fall and injury prevention in older people living in residential care facilities: A cluster randomized trial. Ann Intern Med 2002;136:733–41.
31. Dyer CAE. Falls prevention in residential care homes: a randomised controlled trial. Age Ageing 2004;33:596–602.
32. Huang T-T, Chung M-L, Chen F-R, Chin Y-F, Wang B-H. Evaluation of a combined cognitive-behavioural and exercise intervention to manage fear of falling among elderly residents in nursing homes. Aging Ment Health 2016;20:2–12.
33. Sihvonen S, Sipilä S, Taskinen S, Era P. Fall incidence in frail older women after individualized visual feedback-based balance training. Gerontology 2004;50:411–6.
34. Lord SR, Castell S, Corcoran J, et al. The effect of group exercise on physical functioning and falls in frail older people living in retirement villages: a randomized, controlled trial. J Am Geriatr Soc 2003;51:1685–92.
35. Becker C, Kron M, Lindemann U, et al. Effectiveness of a multifaceted intervention on falls in nursing home residents. J Am Geriatr Soc 2003;51:306–13.
36. Becker C, Kron M, Lindemann U, et al. Effectiveness of a multifaceted intervention on falls in nursing home residents. J Am Geriatr Soc 2003;51:306–13.
37. Jensen J, Lundin-Olsson L, Nyberg L, Gustafson Y. Fall and injury prevention in older people living in residential care facilities. A cluster randomized trial. Ann Intern Med 2002;136:733–41.
38. Moreland JD, Richardson JA, Goldsmith CH, Clase CM. Muscle weakness and falls in older adults: a systematic review and meta-analysis. J Am Geriatr Soc 2004;52: 1121–9.
39. Chodzko-Zajko WJ, Proctor DN, Fiatarone Singh MA, et al. Exercise and physical activity for older adults. Med Sci Sport Exerc 2009;41:1510–30.
40. de Souto Barreto P, Morley JE, Chodzko-Zajko W, et al. Recommendations on physical activity and exercise for older adults living in long-term care facilities: a taskforce report. J Am Med Dir Assoc 2016;17:381–92.
41. American College of Sports Medicine. Progression models in resistance training for healthy adults. Med Sci Sport Exerc 2009;41:687–708.
42. Fietzek UM, Schroeteler FE, Ziegler K, et al. Randomized cross-over trial to investigate the efficacy of a two-week physiotherapy programme with repetitive exercises of cueing to reduce the severity of freezing of gait in patients with Parkinson’s disease. Clin Rehabil 2014;28:902–11.
43. Patterson C, Feightner J, Garcia A, MacKnight C. General risk factors for dementia: A systematic evidence review. Alzheimer Dement 2007;3:341–7.
44. Roach KE, Tappen RM, Kirk-Sanchez N, et al. A randomized controlled trial of an activity specific exercise program for individuals with alzheimer disease in long-term care settings. J Geriatr Phys Ther 2011;34:50–6.
45. Christofoletti G, Oliani MM, Gobbi S, et al. A controlled clinical trial on the effects of motor intervention on balance and cognition in institutionalized elderly patients with dementia. Clin Rehabil 2008;22:618–26.
46. van Alphen HJM, Hortobágyi T, van Heuvelen MJG. Barriers, motivators, and facilitators of physical activity in dementia patients: A systematic review. Arch Gerontol Geriatr 2016;66:109–18.
47. Alzheimer Society of Ontario. Rethink Dementia. Accessed 18 Sep 2017 at http://rethinkdementia.ca/.
48. Roach KE, Tappen RM, Kirk-Sanchez N, et al. A randomized controlled trial of an activity specific exercise program for individuals with Alzheimer disease in long-term care settings. J Geriatr Phys Ther 2011;34:50–6.
49. Brach JS, Perera S, Gilmore S, et al. Effectiveness of a timing and coordination group exercise program to improve mobility in community-dwelling older adults. JAMA Intern Med August 2017.
50. Rosher RB, Robinson S. Impact of the Eden alternative on family satisfaction. J Am Med Dir Assoc 2005;6:189–93.
51. Crandall LG, White DL, Schuldheis S, Talerico KA. Initiating person-centered care practices in long-term care facilities. J Gerontol Nurs 2007;33:47–56.
52. Sims-Gould J, McKay HA, Feldman F, et al. Autonomy, choice, patient-centered care, and hip protectors: the experience of residents and staff in long-term care. J Appl Gerontol 2014;33:690–709.
53. Robinovitch SN, Cronin T. Perception of postural limits in elderly nursing home and day care participants. J Gerontol A Biol Sci Med Sci 1999;54:B124-30.
54. Perkins MM, Ball MM, Whittington FJ, Hollingsworth C. Relational autonomy in assisted living: a focus on diverse care settings for older adults. J Aging Stud 2012;26:214–25.
55. Brownie S, Nancarrow S. Effects of person-centered care on residents and staff in aged-care facilities: a systematic review. Clin Interv Aging 2013;8:1–10.
56. Coleman MT, Looney S, O’Brien J, et al. The Eden Alternative: findings after 1 year of implementation. J Gerontol A Biol Sci Med Sci 2002;57:M422–7.
57. Chenoweth L, King MT, Jeon Y-H, et al. Caring for Aged Dementia Care Resident Study (CADRES) of personcentred care, dementia-care mapping, and usual care in dementia: a cluster-randomised trial. Lancet Neurol 2009;8: 317–25.
58. Bergman-Evans B. Beyond the basics. Effects of the Eden Alternative model on quality of life issues. J Gerontol Nurs 2004;30:27–34.
59. Robinson SB, Rosher RB. Tangling with the barriers to culture change: creating a resident-centered nursing home environment. J Gerontol Nurs 2006;32:19–25.
60. Cameron ID, Gillespie LD, Robertson MC, et al. Interventions for preventing falls in older people in care facilities and hospitals. Cochrane Database Syst Rev 2012;12.
61. Papaioannou A, Santesso N, Morin SN, et al. Recommendations for preventing fracture in long-term care. Can Med Assoc J 2015;187:1135–44.
62. Stubbs B, Denkinger MD, Brefka S, Dallmeier D. What works to prevent falls in older adults dwelling in long term care facilities and hospitals? An umbrella review of meta-analyses of randomised controlled trials. Maturitas 2015;81:335–42.
1. Harris IA, Yong S, McEvoy L, Thorn L. A prospective study of the effect of nursing home residency on mortality following hip fracture. ANZ J Surg 2010;80:447–50.
2. Ooms ME, Vlasman P, Lips P, et al. The incidence of hip fractures in independent and institutionalized elderly people. Osteoporos Int 1994;4:6–10.
3. Ayoung-Chee P, McIntyre L, Ebel BE, et al. Long-term outcomes of ground-level falls in the elderly. J Trauma Acute Care Surg 2014;76:498–503.
4. Heinrich S, Rapp K, Rissmann U, et al. Cost of falls in old age: a systematic review. Osteoporos Int 2010;21: 891–902.
5. Rubenstein LZ, Josephson KR, Robbins AS. Falls in the nursing home. Ann Intern Med 1994;121:442–51.
6. Hartholt KA, van Beeck EF, Polinder S, et al. Societal consequences of falls in the older population: injuries, healthcare costs, and long-term reduced quality of life. J Trauma
2011;71:748–53.
7. Robinovitch SN, Feldman F, Yang Y, et al. Video capture of the circumstances of falls in elderly people residing in long-term care: an observational study. Lancet 2013;381:
47–54.
8. Rapp K, Becker C, Cameron ID, et al. Epidemiology of falls in residential aged care: analysis of more than 70,000 falls from residents of bavarian nursing homes. J Am Med Dir Assoc 2012;13:187.
9. Büchele G, Becker C, Cameron ID, et al. Predictors of serious consequences of falls in residential aged care: analysis of more than 70,000 falls from residents of Bavarian nursing homes. J Am Med Dir Assoc 2014;15:559–63.
10. McArthur C, Gonzalez DA, Roy E, Giangregorio L. What are the circumstances of falls and fractures in long-term care? Can J Aging / La Rev Can du Vieil 2016;35:491–8.
11. Chin A Paw MJM, van Poppel MNM, van Mechelen W. Effects of resistance and functional-skills training on habitual activity and constipation among older adults living in long-term care facilities: a randomized controlled trial. BMC Geriatr 2006;6:9.
12. Ikezoe T, Asakawa Y, Shima H, et al. Daytime physical activity patterns and physical fitness in institutionalized elderly women: an exploratory study. Arch Gerontol Geriatr 2013;57:221–5.
13. Senior HE, Henwood TR, Beller EM, et al. Prevalence and risk factors of sarcopenia among adults living in nursing homes. Maturitas 2015;82:418–23.
14. Smoliner C, Sieber CC, Wirth R. Prevalence of sarcopenia in geriatric hospitalized patients. J Am Med Dir Assoc 2014;15:267–72.
15. Landi F, Liperoti R, Fusco D, et al. Sarcopenia and mortality among older nursing home residents. J Am Med Dir Assoc 2012;13:121–6.
16. Yalcin A, Aras S, Atmis V, et al. Sarcopenia prevalence and factors associated with sarcopenia in older people living in a nursing home in Ankara Turkey. Geriatr Gerontol Int
2016;16:903–10.
17. Sherrington C, Michaleff ZA, Fairhall N, et al. Exercise to prevent falls in older adults: an updated systematic review and meta-analysis. Br J Sports Med October 2016.
18. Liu C, Latham NK. Progressive resistance strength training for improving physical function in older adults. In: Liu C, ed. Cochrane Database Syst Rev;2009:CD002759.
19. Cameron ID, Gillespie LD, Robertson MC, et al. Interventions for preventing falls in older people in care facilities and hospitals. Cochrane Database Syst Rev;2012:CD005465.
20. Silva RB, Eslick GD, Duque G. Exercise for falls and fracture prevention in long term care facilities: a systematic review and meta-analysis. J Am Med Dir Assoc 2013;14:685–9.
21. Hirdes JP, Mitchell L, Maxwell CJ, White N. Beyond the “iron lungs of gerontology”: Using evidence to shape the future of nursing homes in Canada. Can J Aging 2011;30: 371–90.
22. Benjamin K, Edwards N, Guitard P, et al. Factors that influence physical activity in long-term care: Perspectives of residents, staff, and significant others. Can J Aging 2011;30:247–58.
23. Benjamin K, Edwards N, Ploeg J, Legault F. Barriers to physical activity and restorative care for residents in long-term care: A review of the literature. J Aging Phys Act 2014;22:154–65.
24. American College of Sports Medicine. ACSM’s guidelines for exercise testing and prescription. 9th ed. American College of Sports Medicine; 2013.
25. Prevention of Falls Network Europe. Prevention of Falls Network Europe. Accessed 27 Nov 2017 at www.profane.eu.org/.
26. Sihvonen SE, Sipilä S, Era PA. Changes in postural balance in frail elderly women during a 4-week visual feedback training: a randomized controlled trial. Gerontology 2004;50:87–95.
27. Sakamoto K, Nakamura T, Hagino H, et al. Effects of unipedal standing balance exercise on the prevention of falls and hip fracture among clinically defined high-risk elderly individuals: a randomized controlled trial. J Orthop Sci 2006;11:467–72.
28. Shimada H, Obuchi S, Furuna T, Suzuki T. New intervention program for preventing falls among frail elderly people: the effects of perturbed walking exercise using a bilateral separated treadmill. Am J Phys Med Rehabil 2004;83:493–9.
29. Schnelle JF, Kapur K, Alessi C, et al. Does an exercise and incontinence intervention save healthcare costs in a nursing home population? J Am Geriatr Soc 2003;51:161–8.
30. Jensen J, Lundin-Olsson L, Nyberg L, Gustafson Y. Fall and injury prevention in older people living in residential care facilities: A cluster randomized trial. Ann Intern Med 2002;136:733–41.
31. Dyer CAE. Falls prevention in residential care homes: a randomised controlled trial. Age Ageing 2004;33:596–602.
32. Huang T-T, Chung M-L, Chen F-R, Chin Y-F, Wang B-H. Evaluation of a combined cognitive-behavioural and exercise intervention to manage fear of falling among elderly residents in nursing homes. Aging Ment Health 2016;20:2–12.
33. Sihvonen S, Sipilä S, Taskinen S, Era P. Fall incidence in frail older women after individualized visual feedback-based balance training. Gerontology 2004;50:411–6.
34. Lord SR, Castell S, Corcoran J, et al. The effect of group exercise on physical functioning and falls in frail older people living in retirement villages: a randomized, controlled trial. J Am Geriatr Soc 2003;51:1685–92.
35. Becker C, Kron M, Lindemann U, et al. Effectiveness of a multifaceted intervention on falls in nursing home residents. J Am Geriatr Soc 2003;51:306–13.
36. Becker C, Kron M, Lindemann U, et al. Effectiveness of a multifaceted intervention on falls in nursing home residents. J Am Geriatr Soc 2003;51:306–13.
37. Jensen J, Lundin-Olsson L, Nyberg L, Gustafson Y. Fall and injury prevention in older people living in residential care facilities. A cluster randomized trial. Ann Intern Med 2002;136:733–41.
38. Moreland JD, Richardson JA, Goldsmith CH, Clase CM. Muscle weakness and falls in older adults: a systematic review and meta-analysis. J Am Geriatr Soc 2004;52: 1121–9.
39. Chodzko-Zajko WJ, Proctor DN, Fiatarone Singh MA, et al. Exercise and physical activity for older adults. Med Sci Sport Exerc 2009;41:1510–30.
40. de Souto Barreto P, Morley JE, Chodzko-Zajko W, et al. Recommendations on physical activity and exercise for older adults living in long-term care facilities: a taskforce report. J Am Med Dir Assoc 2016;17:381–92.
41. American College of Sports Medicine. Progression models in resistance training for healthy adults. Med Sci Sport Exerc 2009;41:687–708.
42. Fietzek UM, Schroeteler FE, Ziegler K, et al. Randomized cross-over trial to investigate the efficacy of a two-week physiotherapy programme with repetitive exercises of cueing to reduce the severity of freezing of gait in patients with Parkinson’s disease. Clin Rehabil 2014;28:902–11.
43. Patterson C, Feightner J, Garcia A, MacKnight C. General risk factors for dementia: A systematic evidence review. Alzheimer Dement 2007;3:341–7.
44. Roach KE, Tappen RM, Kirk-Sanchez N, et al. A randomized controlled trial of an activity specific exercise program for individuals with alzheimer disease in long-term care settings. J Geriatr Phys Ther 2011;34:50–6.
45. Christofoletti G, Oliani MM, Gobbi S, et al. A controlled clinical trial on the effects of motor intervention on balance and cognition in institutionalized elderly patients with dementia. Clin Rehabil 2008;22:618–26.
46. van Alphen HJM, Hortobágyi T, van Heuvelen MJG. Barriers, motivators, and facilitators of physical activity in dementia patients: A systematic review. Arch Gerontol Geriatr 2016;66:109–18.
47. Alzheimer Society of Ontario. Rethink Dementia. Accessed 18 Sep 2017 at http://rethinkdementia.ca/.
48. Roach KE, Tappen RM, Kirk-Sanchez N, et al. A randomized controlled trial of an activity specific exercise program for individuals with Alzheimer disease in long-term care settings. J Geriatr Phys Ther 2011;34:50–6.
49. Brach JS, Perera S, Gilmore S, et al. Effectiveness of a timing and coordination group exercise program to improve mobility in community-dwelling older adults. JAMA Intern Med August 2017.
50. Rosher RB, Robinson S. Impact of the Eden alternative on family satisfaction. J Am Med Dir Assoc 2005;6:189–93.
51. Crandall LG, White DL, Schuldheis S, Talerico KA. Initiating person-centered care practices in long-term care facilities. J Gerontol Nurs 2007;33:47–56.
52. Sims-Gould J, McKay HA, Feldman F, et al. Autonomy, choice, patient-centered care, and hip protectors: the experience of residents and staff in long-term care. J Appl Gerontol 2014;33:690–709.
53. Robinovitch SN, Cronin T. Perception of postural limits in elderly nursing home and day care participants. J Gerontol A Biol Sci Med Sci 1999;54:B124-30.
54. Perkins MM, Ball MM, Whittington FJ, Hollingsworth C. Relational autonomy in assisted living: a focus on diverse care settings for older adults. J Aging Stud 2012;26:214–25.
55. Brownie S, Nancarrow S. Effects of person-centered care on residents and staff in aged-care facilities: a systematic review. Clin Interv Aging 2013;8:1–10.
56. Coleman MT, Looney S, O’Brien J, et al. The Eden Alternative: findings after 1 year of implementation. J Gerontol A Biol Sci Med Sci 2002;57:M422–7.
57. Chenoweth L, King MT, Jeon Y-H, et al. Caring for Aged Dementia Care Resident Study (CADRES) of personcentred care, dementia-care mapping, and usual care in dementia: a cluster-randomised trial. Lancet Neurol 2009;8: 317–25.
58. Bergman-Evans B. Beyond the basics. Effects of the Eden Alternative model on quality of life issues. J Gerontol Nurs 2004;30:27–34.
59. Robinson SB, Rosher RB. Tangling with the barriers to culture change: creating a resident-centered nursing home environment. J Gerontol Nurs 2006;32:19–25.
60. Cameron ID, Gillespie LD, Robertson MC, et al. Interventions for preventing falls in older people in care facilities and hospitals. Cochrane Database Syst Rev 2012;12.
61. Papaioannou A, Santesso N, Morin SN, et al. Recommendations for preventing fracture in long-term care. Can Med Assoc J 2015;187:1135–44.
62. Stubbs B, Denkinger MD, Brefka S, Dallmeier D. What works to prevent falls in older adults dwelling in long term care facilities and hospitals? An umbrella review of meta-analyses of randomised controlled trials. Maturitas 2015;81:335–42.
Homelessness: Whose job is it?
Despite programs to end homelessness, it remains a substantial and growing problem in many cities in the United States.1,2 In 2016, there were an estimated 10,550 homeless people living in my home state of Colorado, a 6% increase from the prior year.2 A recent point-estimate study found that there were more than 5,000 homeless individuals in the Denver metropolitan area on a single night in January 2017.3 Because of the relative scarcity of housing, a growing number of cities like Denver now utilize a practice known as vulnerability indexing to prioritize homeless persons at high risk of mortality from medical conditions for placement in permanent supportive housing.4
Although hospitalists like myself frequently care for vulnerable homeless patients in the hospital, most have little formal training in how best to care for and advocate for these individuals beyond treating their acute medical need, and little direct contact with community organizations with expertise in doing so. Instead, we have learned informally through experience. Hospital providers are often frustrated by the perceived lack of services and support available to these patients, and there is substantial variability in the extent to which providers engage patients and community partners during and after hospitalization. Despite the growing practice of vulnerability indexing in the community, hospital-based providers do not routinely assess vulnerability with respect to housing. Previous research indicates that housing status is assessed in only a minority of homeless patients during their hospital stay.12 Thus, hospitalization often represents a missed opportunity to identify vulnerability and utilize it to connect patients with housing and other resources.
Addressing the significant known health disparities faced by homeless persons is one of the greatest health equity challenges of our time.13 We need better ways of understanding, identifying, and addressing vulnerability among homeless patients who are hospitalized, paired with improved integration with local community organizations. This will require moving beyond the idea that homelessness is the social worker’s job to one of shared responsibility and advocacy.
Collaborative research and other partnerships that engage both community organizations and individuals affected by homelessness are crucial to further understand the specific needs, barriers, challenges, and opportunities for improving hospital care and care transitions in this population. As well-respected community members and systems thinkers who witness these inequities on a daily basis, hospitalists are well positioned to help lead this work.
Dr. Stella is a hospitalist at Denver Health and Hospital Authority, and an associate professor of medicine at the University of Colorado. She is a member of The Hospitalist editorial advisory board.
References
1. Ending Chronic Homelessness. (Aug 2017). U.S. Interagency Council on Homelessness. Available at: https://www.usich.gov/goals/chronicsness. Accessed: Oct 21, 2017.
2. 2016 Annual Homeless Assessment Report (AHAR) to Congress. (Nov 2016). U.S. Department of Housing and Urban Development Office of Community Planning and Development, Part 1. Available at: https://www.hudexchange.info/resources/documents/2016-AHAR-Part-1.pdf. Accessed: Oct 21, 2017.
3. 2017 Point-In-Time Report, Seven-County Metro Denver Region. Metro Denver Homeless Initiative. Available at: http://www.mdhi.org/2017_pit. Accessed Oct 22, 2017.
4. Henwood BF et al. Examining mortality among formerly homeless adults enrolled in Housing First: An observational study. BMC Public Health. 2015;15:1209.
5. Weinstein LC et al. Moving from street to home: Health status of entrants to a Housing First program. J Prim Care Community Health. 2011;2:11–5.
6. Kushel MB et al. Factors associated with the health care utilization of homeless persons. JAMA. 2001;285(2):200-6.
7. Kushel MB et al. Emergency department use among the homeless and marginally housed: Results from a community-based study. Am J Public Health. 2002;92(5):778-84.
8. Baggett TP et al. Mortality among homeless adults in Boston: Shifts in causes of death over a 15-year period. JAMA Intern Med. 2013 Feb 11;173(3):189–95.
9. Johnson et al. For many patients who use large amounts of health care services, the need is intense yet temporary. Health Aff (Millwood). 2015 Aug;34(8):1312-9.
10. Durfee J et al. The impact of tailored intervention services on charges and mortality for adult super-utilizers. Healthc (Amst). 2017 Aug 25. pii: S2213-0764(17)30057-X. doi: 10.1016/j.hjdsi.2017.08.004. [Epub ahead of print]
11. Rinehart DJ et al. Identifying subgroups of adult super utilizers in an urban safety-net system using latent class analysis: Implications for clinical practice. Med Care. 2016 Sep 14. doi: 10.1097/MLR.0000000000000628. [Epub ahead of print]
12. Greysen RS et al. Understanding transitions of care from hospital to homeless shelter: A mixed-methods, community-based participatory approach. J Gen Intern Med. 2012;27(11):1484-91.
13. National Health Care for the Homeless Council. (Oct 2012). Improving Care Transitions for People Experiencing Homelessness. (Lead author: Sabrina Edgington, policy and program specialist.) Available at: www.nhchc.org/wp-content/uploads/2012/12/Policy_Brief_Care_Transitions.pdf. Accessed Oct 21, 2017.
14. Koh HK et al. Improving healthcare for homeless people. JAMA. 2016;316(24):2586-7.
Despite programs to end homelessness, it remains a substantial and growing problem in many cities in the United States.1,2 In 2016, there were an estimated 10,550 homeless people living in my home state of Colorado, a 6% increase from the prior year.2 A recent point-estimate study found that there were more than 5,000 homeless individuals in the Denver metropolitan area on a single night in January 2017.3 Because of the relative scarcity of housing, a growing number of cities like Denver now utilize a practice known as vulnerability indexing to prioritize homeless persons at high risk of mortality from medical conditions for placement in permanent supportive housing.4
Although hospitalists like myself frequently care for vulnerable homeless patients in the hospital, most have little formal training in how best to care for and advocate for these individuals beyond treating their acute medical need, and little direct contact with community organizations with expertise in doing so. Instead, we have learned informally through experience. Hospital providers are often frustrated by the perceived lack of services and support available to these patients, and there is substantial variability in the extent to which providers engage patients and community partners during and after hospitalization. Despite the growing practice of vulnerability indexing in the community, hospital-based providers do not routinely assess vulnerability with respect to housing. Previous research indicates that housing status is assessed in only a minority of homeless patients during their hospital stay.12 Thus, hospitalization often represents a missed opportunity to identify vulnerability and utilize it to connect patients with housing and other resources.
Addressing the significant known health disparities faced by homeless persons is one of the greatest health equity challenges of our time.13 We need better ways of understanding, identifying, and addressing vulnerability among homeless patients who are hospitalized, paired with improved integration with local community organizations. This will require moving beyond the idea that homelessness is the social worker’s job to one of shared responsibility and advocacy.
Collaborative research and other partnerships that engage both community organizations and individuals affected by homelessness are crucial to further understand the specific needs, barriers, challenges, and opportunities for improving hospital care and care transitions in this population. As well-respected community members and systems thinkers who witness these inequities on a daily basis, hospitalists are well positioned to help lead this work.
Dr. Stella is a hospitalist at Denver Health and Hospital Authority, and an associate professor of medicine at the University of Colorado. She is a member of The Hospitalist editorial advisory board.
References
1. Ending Chronic Homelessness. (Aug 2017). U.S. Interagency Council on Homelessness. Available at: https://www.usich.gov/goals/chronicsness. Accessed: Oct 21, 2017.
2. 2016 Annual Homeless Assessment Report (AHAR) to Congress. (Nov 2016). U.S. Department of Housing and Urban Development Office of Community Planning and Development, Part 1. Available at: https://www.hudexchange.info/resources/documents/2016-AHAR-Part-1.pdf. Accessed: Oct 21, 2017.
3. 2017 Point-In-Time Report, Seven-County Metro Denver Region. Metro Denver Homeless Initiative. Available at: http://www.mdhi.org/2017_pit. Accessed Oct 22, 2017.
4. Henwood BF et al. Examining mortality among formerly homeless adults enrolled in Housing First: An observational study. BMC Public Health. 2015;15:1209.
5. Weinstein LC et al. Moving from street to home: Health status of entrants to a Housing First program. J Prim Care Community Health. 2011;2:11–5.
6. Kushel MB et al. Factors associated with the health care utilization of homeless persons. JAMA. 2001;285(2):200-6.
7. Kushel MB et al. Emergency department use among the homeless and marginally housed: Results from a community-based study. Am J Public Health. 2002;92(5):778-84.
8. Baggett TP et al. Mortality among homeless adults in Boston: Shifts in causes of death over a 15-year period. JAMA Intern Med. 2013 Feb 11;173(3):189–95.
9. Johnson et al. For many patients who use large amounts of health care services, the need is intense yet temporary. Health Aff (Millwood). 2015 Aug;34(8):1312-9.
10. Durfee J et al. The impact of tailored intervention services on charges and mortality for adult super-utilizers. Healthc (Amst). 2017 Aug 25. pii: S2213-0764(17)30057-X. doi: 10.1016/j.hjdsi.2017.08.004. [Epub ahead of print]
11. Rinehart DJ et al. Identifying subgroups of adult super utilizers in an urban safety-net system using latent class analysis: Implications for clinical practice. Med Care. 2016 Sep 14. doi: 10.1097/MLR.0000000000000628. [Epub ahead of print]
12. Greysen RS et al. Understanding transitions of care from hospital to homeless shelter: A mixed-methods, community-based participatory approach. J Gen Intern Med. 2012;27(11):1484-91.
13. National Health Care for the Homeless Council. (Oct 2012). Improving Care Transitions for People Experiencing Homelessness. (Lead author: Sabrina Edgington, policy and program specialist.) Available at: www.nhchc.org/wp-content/uploads/2012/12/Policy_Brief_Care_Transitions.pdf. Accessed Oct 21, 2017.
14. Koh HK et al. Improving healthcare for homeless people. JAMA. 2016;316(24):2586-7.
Despite programs to end homelessness, it remains a substantial and growing problem in many cities in the United States.1,2 In 2016, there were an estimated 10,550 homeless people living in my home state of Colorado, a 6% increase from the prior year.2 A recent point-estimate study found that there were more than 5,000 homeless individuals in the Denver metropolitan area on a single night in January 2017.3 Because of the relative scarcity of housing, a growing number of cities like Denver now utilize a practice known as vulnerability indexing to prioritize homeless persons at high risk of mortality from medical conditions for placement in permanent supportive housing.4
Although hospitalists like myself frequently care for vulnerable homeless patients in the hospital, most have little formal training in how best to care for and advocate for these individuals beyond treating their acute medical need, and little direct contact with community organizations with expertise in doing so. Instead, we have learned informally through experience. Hospital providers are often frustrated by the perceived lack of services and support available to these patients, and there is substantial variability in the extent to which providers engage patients and community partners during and after hospitalization. Despite the growing practice of vulnerability indexing in the community, hospital-based providers do not routinely assess vulnerability with respect to housing. Previous research indicates that housing status is assessed in only a minority of homeless patients during their hospital stay.12 Thus, hospitalization often represents a missed opportunity to identify vulnerability and utilize it to connect patients with housing and other resources.
Addressing the significant known health disparities faced by homeless persons is one of the greatest health equity challenges of our time.13 We need better ways of understanding, identifying, and addressing vulnerability among homeless patients who are hospitalized, paired with improved integration with local community organizations. This will require moving beyond the idea that homelessness is the social worker’s job to one of shared responsibility and advocacy.
Collaborative research and other partnerships that engage both community organizations and individuals affected by homelessness are crucial to further understand the specific needs, barriers, challenges, and opportunities for improving hospital care and care transitions in this population. As well-respected community members and systems thinkers who witness these inequities on a daily basis, hospitalists are well positioned to help lead this work.
Dr. Stella is a hospitalist at Denver Health and Hospital Authority, and an associate professor of medicine at the University of Colorado. She is a member of The Hospitalist editorial advisory board.
References
1. Ending Chronic Homelessness. (Aug 2017). U.S. Interagency Council on Homelessness. Available at: https://www.usich.gov/goals/chronicsness. Accessed: Oct 21, 2017.
2. 2016 Annual Homeless Assessment Report (AHAR) to Congress. (Nov 2016). U.S. Department of Housing and Urban Development Office of Community Planning and Development, Part 1. Available at: https://www.hudexchange.info/resources/documents/2016-AHAR-Part-1.pdf. Accessed: Oct 21, 2017.
3. 2017 Point-In-Time Report, Seven-County Metro Denver Region. Metro Denver Homeless Initiative. Available at: http://www.mdhi.org/2017_pit. Accessed Oct 22, 2017.
4. Henwood BF et al. Examining mortality among formerly homeless adults enrolled in Housing First: An observational study. BMC Public Health. 2015;15:1209.
5. Weinstein LC et al. Moving from street to home: Health status of entrants to a Housing First program. J Prim Care Community Health. 2011;2:11–5.
6. Kushel MB et al. Factors associated with the health care utilization of homeless persons. JAMA. 2001;285(2):200-6.
7. Kushel MB et al. Emergency department use among the homeless and marginally housed: Results from a community-based study. Am J Public Health. 2002;92(5):778-84.
8. Baggett TP et al. Mortality among homeless adults in Boston: Shifts in causes of death over a 15-year period. JAMA Intern Med. 2013 Feb 11;173(3):189–95.
9. Johnson et al. For many patients who use large amounts of health care services, the need is intense yet temporary. Health Aff (Millwood). 2015 Aug;34(8):1312-9.
10. Durfee J et al. The impact of tailored intervention services on charges and mortality for adult super-utilizers. Healthc (Amst). 2017 Aug 25. pii: S2213-0764(17)30057-X. doi: 10.1016/j.hjdsi.2017.08.004. [Epub ahead of print]
11. Rinehart DJ et al. Identifying subgroups of adult super utilizers in an urban safety-net system using latent class analysis: Implications for clinical practice. Med Care. 2016 Sep 14. doi: 10.1097/MLR.0000000000000628. [Epub ahead of print]
12. Greysen RS et al. Understanding transitions of care from hospital to homeless shelter: A mixed-methods, community-based participatory approach. J Gen Intern Med. 2012;27(11):1484-91.
13. National Health Care for the Homeless Council. (Oct 2012). Improving Care Transitions for People Experiencing Homelessness. (Lead author: Sabrina Edgington, policy and program specialist.) Available at: www.nhchc.org/wp-content/uploads/2012/12/Policy_Brief_Care_Transitions.pdf. Accessed Oct 21, 2017.
14. Koh HK et al. Improving healthcare for homeless people. JAMA. 2016;316(24):2586-7.