User login
Beta-blocker therapy post AMI
reported that the treatment of AMI in patients with propranolol, the beta-blocker of the day, reduced mortality by over 25% over 3 years (JAMA. 1981 Nov 6;246[18]:2073-4), and a European trial of timolol confirmed those findings.
New medications have been developed, including ACE inhibitors, statins, and a variety of drugs that modify the thrombotic process that occurs with the event. In addition, endovascular procedures have modified the obstructive coronary vascular anatomy that precipitated the event. At the same time, the definition of an AMI has changed dramatically, now depending in many instances on transitory elevation of the highly sensitive troponins. The BHAT definition depended largely on electrocardiographic changes associated with the event, which were ST-segment elevations in 79% of the occurrences, or significant ST-segment changes associated often with elevation of the insensitive enzyme, serum glutamic transaminase, and significant symptoms. The characteristics of the BHAT patient only faintly resemble the patients who we now classify with AMIs, and its definition expanded well beyond the BHAT patients. And yet beta-blocker therapy is still part of the class I or II recommendations for the treatment of an AMI.
Reasonable questions have been raised as to the wisdom of throwing the full bag of therapy at this population rather than a more selective choice of drugs. In order to gain further insight into the benefits of beta-blocker therapy in the total spectrum of post MI therapy, large populations studies using a variety of statistical gymnastics have tried to identify the unique benefit that can be attributed to beta-blocker therapy relative to the other drugs and surgical interventions used in the post-AMI populations. The result has been a mixed message without any clear guidance. One can make a good case for carrying out clinical trials in specific subsets of the currently defined post-AMI population. Such an endeavor is unlikely. Pharma is clearly not interested in old drug research and the National Heart, Lung, and Blood Institute doesn’t have the funds. So it comes down to clinician’s decisions.
As an avowed beta-blocker enthusiast, I use it in most of my post-AMI patients, particularly if they have left ventricular dysfunction, and in patients with concomitant hypertension. I do not use it in patents with transitory troponin elevations without convincing evidence of clinical symptoms. I will continue beta-blockade as long as I am able to write the prescription or until someone has answered the question.
Dr. Goldstein, medical editor of Cardiology News, is professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies.
reported that the treatment of AMI in patients with propranolol, the beta-blocker of the day, reduced mortality by over 25% over 3 years (JAMA. 1981 Nov 6;246[18]:2073-4), and a European trial of timolol confirmed those findings.
New medications have been developed, including ACE inhibitors, statins, and a variety of drugs that modify the thrombotic process that occurs with the event. In addition, endovascular procedures have modified the obstructive coronary vascular anatomy that precipitated the event. At the same time, the definition of an AMI has changed dramatically, now depending in many instances on transitory elevation of the highly sensitive troponins. The BHAT definition depended largely on electrocardiographic changes associated with the event, which were ST-segment elevations in 79% of the occurrences, or significant ST-segment changes associated often with elevation of the insensitive enzyme, serum glutamic transaminase, and significant symptoms. The characteristics of the BHAT patient only faintly resemble the patients who we now classify with AMIs, and its definition expanded well beyond the BHAT patients. And yet beta-blocker therapy is still part of the class I or II recommendations for the treatment of an AMI.
Reasonable questions have been raised as to the wisdom of throwing the full bag of therapy at this population rather than a more selective choice of drugs. In order to gain further insight into the benefits of beta-blocker therapy in the total spectrum of post MI therapy, large populations studies using a variety of statistical gymnastics have tried to identify the unique benefit that can be attributed to beta-blocker therapy relative to the other drugs and surgical interventions used in the post-AMI populations. The result has been a mixed message without any clear guidance. One can make a good case for carrying out clinical trials in specific subsets of the currently defined post-AMI population. Such an endeavor is unlikely. Pharma is clearly not interested in old drug research and the National Heart, Lung, and Blood Institute doesn’t have the funds. So it comes down to clinician’s decisions.
As an avowed beta-blocker enthusiast, I use it in most of my post-AMI patients, particularly if they have left ventricular dysfunction, and in patients with concomitant hypertension. I do not use it in patents with transitory troponin elevations without convincing evidence of clinical symptoms. I will continue beta-blockade as long as I am able to write the prescription or until someone has answered the question.
Dr. Goldstein, medical editor of Cardiology News, is professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies.
reported that the treatment of AMI in patients with propranolol, the beta-blocker of the day, reduced mortality by over 25% over 3 years (JAMA. 1981 Nov 6;246[18]:2073-4), and a European trial of timolol confirmed those findings.
New medications have been developed, including ACE inhibitors, statins, and a variety of drugs that modify the thrombotic process that occurs with the event. In addition, endovascular procedures have modified the obstructive coronary vascular anatomy that precipitated the event. At the same time, the definition of an AMI has changed dramatically, now depending in many instances on transitory elevation of the highly sensitive troponins. The BHAT definition depended largely on electrocardiographic changes associated with the event, which were ST-segment elevations in 79% of the occurrences, or significant ST-segment changes associated often with elevation of the insensitive enzyme, serum glutamic transaminase, and significant symptoms. The characteristics of the BHAT patient only faintly resemble the patients who we now classify with AMIs, and its definition expanded well beyond the BHAT patients. And yet beta-blocker therapy is still part of the class I or II recommendations for the treatment of an AMI.
Reasonable questions have been raised as to the wisdom of throwing the full bag of therapy at this population rather than a more selective choice of drugs. In order to gain further insight into the benefits of beta-blocker therapy in the total spectrum of post MI therapy, large populations studies using a variety of statistical gymnastics have tried to identify the unique benefit that can be attributed to beta-blocker therapy relative to the other drugs and surgical interventions used in the post-AMI populations. The result has been a mixed message without any clear guidance. One can make a good case for carrying out clinical trials in specific subsets of the currently defined post-AMI population. Such an endeavor is unlikely. Pharma is clearly not interested in old drug research and the National Heart, Lung, and Blood Institute doesn’t have the funds. So it comes down to clinician’s decisions.
As an avowed beta-blocker enthusiast, I use it in most of my post-AMI patients, particularly if they have left ventricular dysfunction, and in patients with concomitant hypertension. I do not use it in patents with transitory troponin elevations without convincing evidence of clinical symptoms. I will continue beta-blockade as long as I am able to write the prescription or until someone has answered the question.
Dr. Goldstein, medical editor of Cardiology News, is professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies.
Warn patients about the risk of misusing OTC pain meds
Roughly 8 in 10 Americans routinely reach for over-the-counter (OTC) pain pills to relieve headaches, backaches, sore muscles, fevers, or colds, according to a national poll by the American Gastroenterological Association (AGA). Most are unaware that these medications, if used incorrectly, can be just as dangerous as prescription drugs.
In my GI practice, I often see cases of accidental OTC pain medicine overdose that have caused stomach bleeding, ulcers, liver damage, and even liver failure. And, I am not alone – the poll found that gastroenterologists see, on average, nearly two patients per week with complications from OTC pain pills.
As the nation’s attention is focused on the opioid crisis, and with good reason, we cannot forget about the risks associated with OTC pain medicines. While it may seem harmless to take more OTC medications than indicated, the body is not capable of absorbing higher doses of pain medicine ingredients at a faster rate.
According to the AGA’s survey, many people are confident they can manage symptoms on their own, without consulting a doctor. Yet, the same poll found that 39% of Americans knowingly took more than the recommended dosage. In most cases, they falsely believed that taking more OTC pain medicine than what was indicated on the label would help them “feel better faster.”
A short time ago, I treated a woman in her 20s who recently had dental surgery. She was taking Lortab, which is a combination of acetaminophen and hydrocodone, an opioid. But when she still felt pain, she took additional OTC acetaminophen to try to find faster relief. When I saw her, her liver tests were abnormal, her acetaminophen level was elevated, and she was feeling very nauseated. In trying to get faster pain relief, she unintentionally overdosed on OTC pain medicine.
To help patients avoid this kind of medication mishap, it’s vital that health care providers initiate conversations at every visit about the dangers of OTC pain medicine overdose. The following tips have helped me advise my patients and educate them on the associated risks:
• Encourage your patients to read and follow all medicine labels, even on OTC drugs – every time they reach for something in the medicine cabinet.
• Talk to your patients about the two main types of oral OTC pain medicines and make sure they know to take only one product at a time containing the same type of active ingredient.
• Ask about all medicines your patients take, including OTC medicines, as they may not know to tell you.
• Patients may not realize that their current health situation, age, and/or medical history can impact their risk for OTC pain medicine overdose. Let them know that products that worked in the past may no longer be the right choice for them now.
If more health care providers emphasized the dangers of incorrect usage of OTC pain medicines, we could easily help patients avoid the dangerous side effects of taking too much.
Dr. Wilcox is professor of medicine in the division of gastroenterology and hepatology at the University of Alabama at Birmingham and a chair of AGA’s Gut Check: Know Your Medicine campaign.
Roughly 8 in 10 Americans routinely reach for over-the-counter (OTC) pain pills to relieve headaches, backaches, sore muscles, fevers, or colds, according to a national poll by the American Gastroenterological Association (AGA). Most are unaware that these medications, if used incorrectly, can be just as dangerous as prescription drugs.
In my GI practice, I often see cases of accidental OTC pain medicine overdose that have caused stomach bleeding, ulcers, liver damage, and even liver failure. And, I am not alone – the poll found that gastroenterologists see, on average, nearly two patients per week with complications from OTC pain pills.
As the nation’s attention is focused on the opioid crisis, and with good reason, we cannot forget about the risks associated with OTC pain medicines. While it may seem harmless to take more OTC medications than indicated, the body is not capable of absorbing higher doses of pain medicine ingredients at a faster rate.
According to the AGA’s survey, many people are confident they can manage symptoms on their own, without consulting a doctor. Yet, the same poll found that 39% of Americans knowingly took more than the recommended dosage. In most cases, they falsely believed that taking more OTC pain medicine than what was indicated on the label would help them “feel better faster.”
A short time ago, I treated a woman in her 20s who recently had dental surgery. She was taking Lortab, which is a combination of acetaminophen and hydrocodone, an opioid. But when she still felt pain, she took additional OTC acetaminophen to try to find faster relief. When I saw her, her liver tests were abnormal, her acetaminophen level was elevated, and she was feeling very nauseated. In trying to get faster pain relief, she unintentionally overdosed on OTC pain medicine.
To help patients avoid this kind of medication mishap, it’s vital that health care providers initiate conversations at every visit about the dangers of OTC pain medicine overdose. The following tips have helped me advise my patients and educate them on the associated risks:
• Encourage your patients to read and follow all medicine labels, even on OTC drugs – every time they reach for something in the medicine cabinet.
• Talk to your patients about the two main types of oral OTC pain medicines and make sure they know to take only one product at a time containing the same type of active ingredient.
• Ask about all medicines your patients take, including OTC medicines, as they may not know to tell you.
• Patients may not realize that their current health situation, age, and/or medical history can impact their risk for OTC pain medicine overdose. Let them know that products that worked in the past may no longer be the right choice for them now.
If more health care providers emphasized the dangers of incorrect usage of OTC pain medicines, we could easily help patients avoid the dangerous side effects of taking too much.
Dr. Wilcox is professor of medicine in the division of gastroenterology and hepatology at the University of Alabama at Birmingham and a chair of AGA’s Gut Check: Know Your Medicine campaign.
Roughly 8 in 10 Americans routinely reach for over-the-counter (OTC) pain pills to relieve headaches, backaches, sore muscles, fevers, or colds, according to a national poll by the American Gastroenterological Association (AGA). Most are unaware that these medications, if used incorrectly, can be just as dangerous as prescription drugs.
In my GI practice, I often see cases of accidental OTC pain medicine overdose that have caused stomach bleeding, ulcers, liver damage, and even liver failure. And, I am not alone – the poll found that gastroenterologists see, on average, nearly two patients per week with complications from OTC pain pills.
As the nation’s attention is focused on the opioid crisis, and with good reason, we cannot forget about the risks associated with OTC pain medicines. While it may seem harmless to take more OTC medications than indicated, the body is not capable of absorbing higher doses of pain medicine ingredients at a faster rate.
According to the AGA’s survey, many people are confident they can manage symptoms on their own, without consulting a doctor. Yet, the same poll found that 39% of Americans knowingly took more than the recommended dosage. In most cases, they falsely believed that taking more OTC pain medicine than what was indicated on the label would help them “feel better faster.”
A short time ago, I treated a woman in her 20s who recently had dental surgery. She was taking Lortab, which is a combination of acetaminophen and hydrocodone, an opioid. But when she still felt pain, she took additional OTC acetaminophen to try to find faster relief. When I saw her, her liver tests were abnormal, her acetaminophen level was elevated, and she was feeling very nauseated. In trying to get faster pain relief, she unintentionally overdosed on OTC pain medicine.
To help patients avoid this kind of medication mishap, it’s vital that health care providers initiate conversations at every visit about the dangers of OTC pain medicine overdose. The following tips have helped me advise my patients and educate them on the associated risks:
• Encourage your patients to read and follow all medicine labels, even on OTC drugs – every time they reach for something in the medicine cabinet.
• Talk to your patients about the two main types of oral OTC pain medicines and make sure they know to take only one product at a time containing the same type of active ingredient.
• Ask about all medicines your patients take, including OTC medicines, as they may not know to tell you.
• Patients may not realize that their current health situation, age, and/or medical history can impact their risk for OTC pain medicine overdose. Let them know that products that worked in the past may no longer be the right choice for them now.
If more health care providers emphasized the dangers of incorrect usage of OTC pain medicines, we could easily help patients avoid the dangerous side effects of taking too much.
Dr. Wilcox is professor of medicine in the division of gastroenterology and hepatology at the University of Alabama at Birmingham and a chair of AGA’s Gut Check: Know Your Medicine campaign.
Anxiety disorders: Psychopharmacologic treatment update
Anxiety disorders, including separation anxiety disorder, social anxiety disorder, and generalized anxiety disorder, are some of the most common psychiatric conditions of childhood and adolescence, affecting up to 20% of youth.1 Patients commonly present with a mix of symptoms that often span multiple anxiety disorder diagnoses. While this pattern can present somewhat of a diagnostic conundrum, it can be reassuring to know that such constellations of symptoms are the rule rather than the exception. Further, given that both the pharmacologic and nonpharmacologic treatment strategies don’t change much among the various anxiety disorders, the lack of a definitive single diagnosis should not delay intervention. Be alert to the possibility that anxiety and anxiety disorders can be the engine that drives what on the surface appears to be more disruptive and oppositional behavior.
Although medications can be a useful part of treatment, they are not recommended as a stand-alone intervention. Nonpharmacologic treatments generally should be tried before medications are considered. Among the different types of psychotherapy, cognitive-behavioral therapy (CBT) has the most empirical support from research trials, although other modalities such as mindfulness-based treatments show some promise. As anxiety disorders often run in families, it also can be very useful to explore the possibility that one or more parents also struggle with an anxiety disorder, which, if untreated, might complicate the child’s course.
With regard to medications, it is being increasingly appreciated that, despite SSRIs being most popularly known as antidepressants, these medications actually may be as efficacious or even more efficacious in the management of anxiety disorders. This class remains the cornerstone of medication treatment, and a brief review of current options follows.
SSRIs and SNRIs
A 2015 meta-analysis that examined nine randomized controlled trials of SSRIs and serotonin and norepinephrine reuptake inhibitors (SNRIs) for pediatric anxiety disorders concluded that these agents provided a benefit of modest effect size. No significant increase in treatment-emergent suicidality was found, and the medications were generally well tolerated.2 This analysis also found some evidence that greater efficacy was related to a medication with more specific serotonergic properties, suggesting improved response with “true” SSRIs versus SNRIs such as venlafaxine and duloxetine. One major study using sertraline found that, at least in the short term, combined use of sertraline with CBT resulted in better efficacy than either treatment alone.3 Dosing of SSRIs should start low: A general rule is to begin at half of the smallest dosage made, depending on the age and size of the patient. One question that often comes up after a successful trial is how long to continue the medications. A recent meta-analysis in adults concluded that there was evidence that stopping medication prior to 1 year resulted in an increased risk of relapse with little to guide clinicians after that 1-year mark.4
Benzodiazepines
Even though benzodiazepines have been around for a long time, data supporting their efficacy and safety in pediatric populations remain extremely limited, and what has been reported has not been particularly positive. Thus, most experts do not suggest using benzodiazepines for anxiety disorders, with the exception of helping children through single or rare events, such as medical procedures or enabling an adolescent who has been fearful of attending school to get to the building on the first day back after a long absence.
Guanfacine
In a recent exploratory trial of guanfacine for children with mixed anxiety disorders,5 the medication was well tolerated overall but did not result in statistically significant improvement relative to placebo on primary anxiety rating scales. However, a higher number of children were rated as improved on a clinician-rated scale. This medication is usually started at 0.5 mg/day and increased as tolerated, while checking vital signs, to a maximum of 4 mg/day.
Atomoxetine
A randomized control trial of pediatric patients with both ADHD and an anxiety disorder showed reductions in both symptom domains with atomoxetine dosed at an average of 1.3 mg/kg per day.6 There is little evidence to suggest its use in primary anxiety disorders without comorbid ADHD.
Buspirone
This 5-hydroxytryptamine 1a agonist has Food and Drug Administration approval for generalized anxiety disorder in adults and is generally well tolerated. Unfortunately, two randomized controlled studies in children and adolescents did not find statistically significant improvement relative to placebo, although some methodological problems may have played a role.7
Antipsychotics
Although sometimes used to augment an SSRI in adult anxiety disorders, there are little data to support the use of antipsychotics in pediatric populations, especially given the antecedent risks of the drugs.
Summary
Pharmacotherapy for anxiety disorders often includes the advice that, if medications are indicated in conjunction with psychotherapy, to start with an SSRI; and if that is not effective to try a different one.7 An SNRI such as venlafaxine or duloxetine may then be a third-line alternative, although for youth with comorbid ADHD, consideration of either atomoxetine or guanfacine is also reasonable. Beyond that point, there unfortunately are little systematic data to guide pharmacologic decision making, and increased potential risks of other classes of medications suggest the need for caution and consultation.
Looking for more mental health training? Attend the 12th annual Child Psychiatry in Primary Care conference in Burlington, on May 4, 2018,organized by the University of Vermont with Dr. Rettew as course director. Go to http://www.med.uvm.edu/cme/conferences.
References
1. Merikangas KR et al. J Am Acad Child Adolesc Psychiatry. 2010 Oct;49(10):980-9.
2. Strawn JR et al. Depress Anxiety. 2015 Mar;32(3):149-57.
3. Walkup J et al. N Engl J Med. 2008 Dec 25;359(26):2753-66.
4. Batelaan N et al. BMJ. 2017 Sep 13;358:j3927.
5. Strawn JR et al. J Child Adolesc Psychopharm. 2017 Feb;27(1): 29-37..
6. Geller D et al. J Am Acad Child Adolesc Psychiatry. 2007 Sep;46(9):1119-27.
7. Strawn JR et al. J Child Adolesc Psychopharm. 2017 Feb;28(1): 2-9.
Anxiety disorders, including separation anxiety disorder, social anxiety disorder, and generalized anxiety disorder, are some of the most common psychiatric conditions of childhood and adolescence, affecting up to 20% of youth.1 Patients commonly present with a mix of symptoms that often span multiple anxiety disorder diagnoses. While this pattern can present somewhat of a diagnostic conundrum, it can be reassuring to know that such constellations of symptoms are the rule rather than the exception. Further, given that both the pharmacologic and nonpharmacologic treatment strategies don’t change much among the various anxiety disorders, the lack of a definitive single diagnosis should not delay intervention. Be alert to the possibility that anxiety and anxiety disorders can be the engine that drives what on the surface appears to be more disruptive and oppositional behavior.
Although medications can be a useful part of treatment, they are not recommended as a stand-alone intervention. Nonpharmacologic treatments generally should be tried before medications are considered. Among the different types of psychotherapy, cognitive-behavioral therapy (CBT) has the most empirical support from research trials, although other modalities such as mindfulness-based treatments show some promise. As anxiety disorders often run in families, it also can be very useful to explore the possibility that one or more parents also struggle with an anxiety disorder, which, if untreated, might complicate the child’s course.
With regard to medications, it is being increasingly appreciated that, despite SSRIs being most popularly known as antidepressants, these medications actually may be as efficacious or even more efficacious in the management of anxiety disorders. This class remains the cornerstone of medication treatment, and a brief review of current options follows.
SSRIs and SNRIs
A 2015 meta-analysis that examined nine randomized controlled trials of SSRIs and serotonin and norepinephrine reuptake inhibitors (SNRIs) for pediatric anxiety disorders concluded that these agents provided a benefit of modest effect size. No significant increase in treatment-emergent suicidality was found, and the medications were generally well tolerated.2 This analysis also found some evidence that greater efficacy was related to a medication with more specific serotonergic properties, suggesting improved response with “true” SSRIs versus SNRIs such as venlafaxine and duloxetine. One major study using sertraline found that, at least in the short term, combined use of sertraline with CBT resulted in better efficacy than either treatment alone.3 Dosing of SSRIs should start low: A general rule is to begin at half of the smallest dosage made, depending on the age and size of the patient. One question that often comes up after a successful trial is how long to continue the medications. A recent meta-analysis in adults concluded that there was evidence that stopping medication prior to 1 year resulted in an increased risk of relapse with little to guide clinicians after that 1-year mark.4
Benzodiazepines
Even though benzodiazepines have been around for a long time, data supporting their efficacy and safety in pediatric populations remain extremely limited, and what has been reported has not been particularly positive. Thus, most experts do not suggest using benzodiazepines for anxiety disorders, with the exception of helping children through single or rare events, such as medical procedures or enabling an adolescent who has been fearful of attending school to get to the building on the first day back after a long absence.
Guanfacine
In a recent exploratory trial of guanfacine for children with mixed anxiety disorders,5 the medication was well tolerated overall but did not result in statistically significant improvement relative to placebo on primary anxiety rating scales. However, a higher number of children were rated as improved on a clinician-rated scale. This medication is usually started at 0.5 mg/day and increased as tolerated, while checking vital signs, to a maximum of 4 mg/day.
Atomoxetine
A randomized control trial of pediatric patients with both ADHD and an anxiety disorder showed reductions in both symptom domains with atomoxetine dosed at an average of 1.3 mg/kg per day.6 There is little evidence to suggest its use in primary anxiety disorders without comorbid ADHD.
Buspirone
This 5-hydroxytryptamine 1a agonist has Food and Drug Administration approval for generalized anxiety disorder in adults and is generally well tolerated. Unfortunately, two randomized controlled studies in children and adolescents did not find statistically significant improvement relative to placebo, although some methodological problems may have played a role.7
Antipsychotics
Although sometimes used to augment an SSRI in adult anxiety disorders, there are little data to support the use of antipsychotics in pediatric populations, especially given the antecedent risks of the drugs.
Summary
Pharmacotherapy for anxiety disorders often includes the advice that, if medications are indicated in conjunction with psychotherapy, to start with an SSRI; and if that is not effective to try a different one.7 An SNRI such as venlafaxine or duloxetine may then be a third-line alternative, although for youth with comorbid ADHD, consideration of either atomoxetine or guanfacine is also reasonable. Beyond that point, there unfortunately are little systematic data to guide pharmacologic decision making, and increased potential risks of other classes of medications suggest the need for caution and consultation.
Looking for more mental health training? Attend the 12th annual Child Psychiatry in Primary Care conference in Burlington, on May 4, 2018,organized by the University of Vermont with Dr. Rettew as course director. Go to http://www.med.uvm.edu/cme/conferences.
References
1. Merikangas KR et al. J Am Acad Child Adolesc Psychiatry. 2010 Oct;49(10):980-9.
2. Strawn JR et al. Depress Anxiety. 2015 Mar;32(3):149-57.
3. Walkup J et al. N Engl J Med. 2008 Dec 25;359(26):2753-66.
4. Batelaan N et al. BMJ. 2017 Sep 13;358:j3927.
5. Strawn JR et al. J Child Adolesc Psychopharm. 2017 Feb;27(1): 29-37..
6. Geller D et al. J Am Acad Child Adolesc Psychiatry. 2007 Sep;46(9):1119-27.
7. Strawn JR et al. J Child Adolesc Psychopharm. 2017 Feb;28(1): 2-9.
Anxiety disorders, including separation anxiety disorder, social anxiety disorder, and generalized anxiety disorder, are some of the most common psychiatric conditions of childhood and adolescence, affecting up to 20% of youth.1 Patients commonly present with a mix of symptoms that often span multiple anxiety disorder diagnoses. While this pattern can present somewhat of a diagnostic conundrum, it can be reassuring to know that such constellations of symptoms are the rule rather than the exception. Further, given that both the pharmacologic and nonpharmacologic treatment strategies don’t change much among the various anxiety disorders, the lack of a definitive single diagnosis should not delay intervention. Be alert to the possibility that anxiety and anxiety disorders can be the engine that drives what on the surface appears to be more disruptive and oppositional behavior.
Although medications can be a useful part of treatment, they are not recommended as a stand-alone intervention. Nonpharmacologic treatments generally should be tried before medications are considered. Among the different types of psychotherapy, cognitive-behavioral therapy (CBT) has the most empirical support from research trials, although other modalities such as mindfulness-based treatments show some promise. As anxiety disorders often run in families, it also can be very useful to explore the possibility that one or more parents also struggle with an anxiety disorder, which, if untreated, might complicate the child’s course.
With regard to medications, it is being increasingly appreciated that, despite SSRIs being most popularly known as antidepressants, these medications actually may be as efficacious or even more efficacious in the management of anxiety disorders. This class remains the cornerstone of medication treatment, and a brief review of current options follows.
SSRIs and SNRIs
A 2015 meta-analysis that examined nine randomized controlled trials of SSRIs and serotonin and norepinephrine reuptake inhibitors (SNRIs) for pediatric anxiety disorders concluded that these agents provided a benefit of modest effect size. No significant increase in treatment-emergent suicidality was found, and the medications were generally well tolerated.2 This analysis also found some evidence that greater efficacy was related to a medication with more specific serotonergic properties, suggesting improved response with “true” SSRIs versus SNRIs such as venlafaxine and duloxetine. One major study using sertraline found that, at least in the short term, combined use of sertraline with CBT resulted in better efficacy than either treatment alone.3 Dosing of SSRIs should start low: A general rule is to begin at half of the smallest dosage made, depending on the age and size of the patient. One question that often comes up after a successful trial is how long to continue the medications. A recent meta-analysis in adults concluded that there was evidence that stopping medication prior to 1 year resulted in an increased risk of relapse with little to guide clinicians after that 1-year mark.4
Benzodiazepines
Even though benzodiazepines have been around for a long time, data supporting their efficacy and safety in pediatric populations remain extremely limited, and what has been reported has not been particularly positive. Thus, most experts do not suggest using benzodiazepines for anxiety disorders, with the exception of helping children through single or rare events, such as medical procedures or enabling an adolescent who has been fearful of attending school to get to the building on the first day back after a long absence.
Guanfacine
In a recent exploratory trial of guanfacine for children with mixed anxiety disorders,5 the medication was well tolerated overall but did not result in statistically significant improvement relative to placebo on primary anxiety rating scales. However, a higher number of children were rated as improved on a clinician-rated scale. This medication is usually started at 0.5 mg/day and increased as tolerated, while checking vital signs, to a maximum of 4 mg/day.
Atomoxetine
A randomized control trial of pediatric patients with both ADHD and an anxiety disorder showed reductions in both symptom domains with atomoxetine dosed at an average of 1.3 mg/kg per day.6 There is little evidence to suggest its use in primary anxiety disorders without comorbid ADHD.
Buspirone
This 5-hydroxytryptamine 1a agonist has Food and Drug Administration approval for generalized anxiety disorder in adults and is generally well tolerated. Unfortunately, two randomized controlled studies in children and adolescents did not find statistically significant improvement relative to placebo, although some methodological problems may have played a role.7
Antipsychotics
Although sometimes used to augment an SSRI in adult anxiety disorders, there are little data to support the use of antipsychotics in pediatric populations, especially given the antecedent risks of the drugs.
Summary
Pharmacotherapy for anxiety disorders often includes the advice that, if medications are indicated in conjunction with psychotherapy, to start with an SSRI; and if that is not effective to try a different one.7 An SNRI such as venlafaxine or duloxetine may then be a third-line alternative, although for youth with comorbid ADHD, consideration of either atomoxetine or guanfacine is also reasonable. Beyond that point, there unfortunately are little systematic data to guide pharmacologic decision making, and increased potential risks of other classes of medications suggest the need for caution and consultation.
Looking for more mental health training? Attend the 12th annual Child Psychiatry in Primary Care conference in Burlington, on May 4, 2018,organized by the University of Vermont with Dr. Rettew as course director. Go to http://www.med.uvm.edu/cme/conferences.
References
1. Merikangas KR et al. J Am Acad Child Adolesc Psychiatry. 2010 Oct;49(10):980-9.
2. Strawn JR et al. Depress Anxiety. 2015 Mar;32(3):149-57.
3. Walkup J et al. N Engl J Med. 2008 Dec 25;359(26):2753-66.
4. Batelaan N et al. BMJ. 2017 Sep 13;358:j3927.
5. Strawn JR et al. J Child Adolesc Psychopharm. 2017 Feb;27(1): 29-37..
6. Geller D et al. J Am Acad Child Adolesc Psychiatry. 2007 Sep;46(9):1119-27.
7. Strawn JR et al. J Child Adolesc Psychopharm. 2017 Feb;28(1): 2-9.
How to help children process, overcome horrific traumas
Unfathomable. Unspeakable.
These are among the terms used to describe children’s extreme traumatic experiences such as severe abuse and neglect. It is often most shocking when these acts are perpetrated by the children’s parents – the very ones that children should be able to depend on for their protection and safety.
Many believe that, in addition to the cumulative and serious nature of repetitive interpersonal traumas themselves, this betrayal of trust will result in irreparable psychological damage. Fortunately, this does not have to be the case. Children are more resilient than we realize; with safety, support, and effective treatment, they can recover from even the most extreme traumas and live healthy, productive lives.
and allow them to live in safe, stable, supportive settings while minimizing traumatic separation from siblings or further disruptions in their living situation. Acute medical problems need to be stabilized, and a thorough mental health assessment should be conducted.
Evidence-based trauma-focused psychotherapy is the first-line treatment for addressing pediatric posttraumatic stress disorder (J Am Acad Child Adolesc Psychiatry. 2010 Apr;49[4]:414-30). Several treatments are currently available. A few examples are trauma-focused cognitive-behavioral therapy, or TF-CBT, for children aged 3-18 years; child-parent psychotherapy (CPP) for children aged 0-6 years; a group school-based model, cognitive behavioral interventions for trauma in schools (CBITS); and Trauma Affect Regulation: Guide for Education and Therapy for teens (TARGET).
Common elements of evidence-based trauma-focused treatments are: 1) nonperpetrating caregivers are included in therapy to enhance support and understanding of the child’s trauma responses, and to address trauma-related behavioral problems; 2) skills are provided to the youth and caregiver for coping with negative trauma-related thoughts, feelings, and behaviors; and 3) children are supported to directly talk about and make meaning of their trauma experiences.
Through trauma-focused treatment, children become able to cope with their traumatic experiences and memories – make sense out of them. These traumas are no longer “unfathomable or “unspeakable,” but rather, manageable memories of bad experiences that the children have had the courage to face and master.
Children can recover from even extreme trauma experiences when they receive effective trauma-focused treatment in the context of a supportive environment. More information about evidence-based treatments is available from the National Child Traumatic Stress Network at www.nctsn.org/resources/topics/treatments-that-work/promising-practices.
Unfathomable. Unspeakable.
These are among the terms used to describe children’s extreme traumatic experiences such as severe abuse and neglect. It is often most shocking when these acts are perpetrated by the children’s parents – the very ones that children should be able to depend on for their protection and safety.
Many believe that, in addition to the cumulative and serious nature of repetitive interpersonal traumas themselves, this betrayal of trust will result in irreparable psychological damage. Fortunately, this does not have to be the case. Children are more resilient than we realize; with safety, support, and effective treatment, they can recover from even the most extreme traumas and live healthy, productive lives.
and allow them to live in safe, stable, supportive settings while minimizing traumatic separation from siblings or further disruptions in their living situation. Acute medical problems need to be stabilized, and a thorough mental health assessment should be conducted.
Evidence-based trauma-focused psychotherapy is the first-line treatment for addressing pediatric posttraumatic stress disorder (J Am Acad Child Adolesc Psychiatry. 2010 Apr;49[4]:414-30). Several treatments are currently available. A few examples are trauma-focused cognitive-behavioral therapy, or TF-CBT, for children aged 3-18 years; child-parent psychotherapy (CPP) for children aged 0-6 years; a group school-based model, cognitive behavioral interventions for trauma in schools (CBITS); and Trauma Affect Regulation: Guide for Education and Therapy for teens (TARGET).
Common elements of evidence-based trauma-focused treatments are: 1) nonperpetrating caregivers are included in therapy to enhance support and understanding of the child’s trauma responses, and to address trauma-related behavioral problems; 2) skills are provided to the youth and caregiver for coping with negative trauma-related thoughts, feelings, and behaviors; and 3) children are supported to directly talk about and make meaning of their trauma experiences.
Through trauma-focused treatment, children become able to cope with their traumatic experiences and memories – make sense out of them. These traumas are no longer “unfathomable or “unspeakable,” but rather, manageable memories of bad experiences that the children have had the courage to face and master.
Children can recover from even extreme trauma experiences when they receive effective trauma-focused treatment in the context of a supportive environment. More information about evidence-based treatments is available from the National Child Traumatic Stress Network at www.nctsn.org/resources/topics/treatments-that-work/promising-practices.
Unfathomable. Unspeakable.
These are among the terms used to describe children’s extreme traumatic experiences such as severe abuse and neglect. It is often most shocking when these acts are perpetrated by the children’s parents – the very ones that children should be able to depend on for their protection and safety.
Many believe that, in addition to the cumulative and serious nature of repetitive interpersonal traumas themselves, this betrayal of trust will result in irreparable psychological damage. Fortunately, this does not have to be the case. Children are more resilient than we realize; with safety, support, and effective treatment, they can recover from even the most extreme traumas and live healthy, productive lives.
and allow them to live in safe, stable, supportive settings while minimizing traumatic separation from siblings or further disruptions in their living situation. Acute medical problems need to be stabilized, and a thorough mental health assessment should be conducted.
Evidence-based trauma-focused psychotherapy is the first-line treatment for addressing pediatric posttraumatic stress disorder (J Am Acad Child Adolesc Psychiatry. 2010 Apr;49[4]:414-30). Several treatments are currently available. A few examples are trauma-focused cognitive-behavioral therapy, or TF-CBT, for children aged 3-18 years; child-parent psychotherapy (CPP) for children aged 0-6 years; a group school-based model, cognitive behavioral interventions for trauma in schools (CBITS); and Trauma Affect Regulation: Guide for Education and Therapy for teens (TARGET).
Common elements of evidence-based trauma-focused treatments are: 1) nonperpetrating caregivers are included in therapy to enhance support and understanding of the child’s trauma responses, and to address trauma-related behavioral problems; 2) skills are provided to the youth and caregiver for coping with negative trauma-related thoughts, feelings, and behaviors; and 3) children are supported to directly talk about and make meaning of their trauma experiences.
Through trauma-focused treatment, children become able to cope with their traumatic experiences and memories – make sense out of them. These traumas are no longer “unfathomable or “unspeakable,” but rather, manageable memories of bad experiences that the children have had the courage to face and master.
Children can recover from even extreme trauma experiences when they receive effective trauma-focused treatment in the context of a supportive environment. More information about evidence-based treatments is available from the National Child Traumatic Stress Network at www.nctsn.org/resources/topics/treatments-that-work/promising-practices.
Implementing enhanced recovery protocols for gynecologic surgery
“Enhanced Recovery After Surgery” (ERAS) practices and protocols have been increasingly refined and adopted for the field of gynecology, and there is hope among gynecologic surgeons – and some recent evidence – that, with the ERAS movement, we are improving patient recoveries and outcomes and minimizing the need for opioids.
This applies not only to open surgeries but also to the minimally invasive procedures that already are prized for significant reductions in morbidity and length of stay. The overarching and guiding principle of ERAS is that any surgery – whether open or minimally invasive, major or minor – places stress on the body and is associated with risks and morbidity.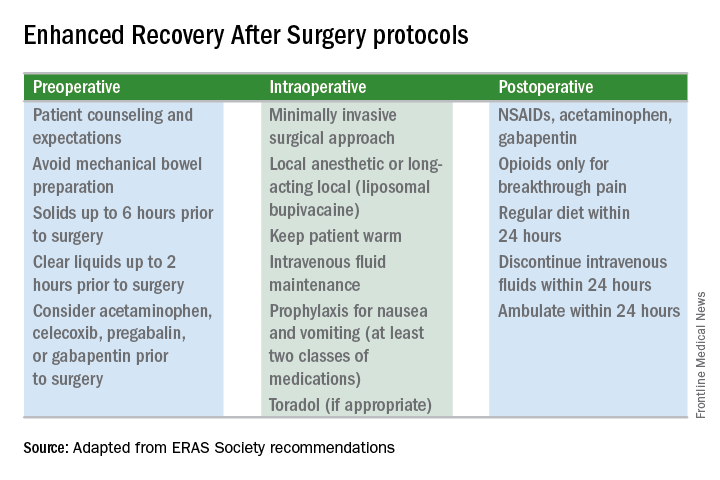
Enhanced recovery protocols are multidisciplinary, perioperative approaches designed to lessen the body’s stress response to surgery. The protocols and pathways offer us a menu of small changes that, in the aggregate, can lead to large and demonstrable benefits – especially when these small changes are chosen across the preoperative, intraoperative, and postoperative arenas and then standardized in one’s practice. Among the major components of ERAS practices and protocols are limiting preoperative fasting, employing multimodal analgesia, encouraging early ambulation and early postsurgical feeding, and creating culture shift that includes greater emphasis on patient expectations.
In our practice, we are incorporating ERAS practices not only in hopes of reducing the stress of all surgeries before, during, and after, but also with the goal of achieving a postoperative opioid-free hysterectomy, myomectomy, and extensive endometriosis surgery. (All of our advanced procedures are performed laparoscopically or robotically.)
Over the past 7 or so years, we have adopted a multimodal approach to pain control that includes a bundle of preoperative analgesics – acetaminophen, pregabalin, and celecoxib (we call it “TLC” for Tylenol, Lyrica, and Celebrex) – and the use of liposomal bupivacaine in our robotic surgeries. We are now turning toward ERAS nutritional changes, most of which run counter to traditional paradigms for surgical care. And in other areas, such as dedicated preoperative counseling, we continue to refine and improve our practices.
Improved Outcomes
The ERAS mindset notably intersected gynecology with the publication in 2016 of a two-part series of guidelines for gynecology/oncology surgery from the ERAS Society. The 8-year-old society has its roots in a study group of European surgeons and others who decided to examine surgical practices and the concept of multimodal surgical care put forth in the 1990s by Henrik Kehlet, MD, PhD, then a professor at the University of Copenhagen.
The first set of recommendations addressed pre- and intraoperative care (Gynecol Oncol. 2016 Feb;140[2]:313-22), and the second set addressed postoperative care (Gynecol Oncol. 2016 Feb;140[2]:323-32). Similar evidence-based recommendations were previously written for colonic resections, rectal and pelvic surgery, and other surgical specialties.
Most of the published outcomes of enhanced recovery protocols come from colorectal surgery. As noted in the ERAS Society gynecology/oncology guidelines, the benefits include an average reduction in length of stay of 2.5 days and a decrease in complications by as much as 50%.
There is growing evidence, however, that ERAS programs are also beneficial for patients undergoing laparoscopic surgery, and outcomes from gynecology – including minimally invasive surgery – are also being reported.
For instance, a retrospective case-control study of 55 consecutive gynecologic oncology patients treated at the University of California, San Francisco, with laparoscopic or robotic surgery and an enhanced recovery pathway – and 110 historical control patients matched on the basis of age and surgery type – found significant improvements in recovery time, decreased pain despite reduced opioid use, and overall lower hospital costs (Obstet Gynecol. 2016 Jul;128[1]:138-44).
The enhanced recovery pathway included patient education, multimodal antiemetics, multimodal analgesia, and balanced fluid administration. Early catheter removal, ambulation, and feeding were also components. Analgesia included routine preoperative gabapentin, diclofenac, and acetaminophen; routine postoperative gabapentin, NSAIDs, and acetaminophen; and transversus abdominis plane blocks in 32 of the ERAS patients.
ERAS patients were significantly more likely to be discharged on day 1 (91%, compared with 60% in the control group). Opioid use decreased by 30%, and pain scores on postoperative day 1 were significantly lower.
Another study looking at the effect of enhanced recovery implementation in gynecologic surgeries at the University of Virginia, Charlottesville, (gynecologic oncology, urogynecology, and general gynecology) similarly reported benefits for vaginal and minimally invasive procedures, as well as for open procedures (Obstet Gynecol. 2016 Sep;128[3]:457-66).
In the minimally invasive group, investigators compared 324 patients before ERAS implementation with 249 patients afterward and found that the median length of stay was unchanged (1 day). However, intraoperative and postoperative opioid consumption decreased significantly and – even though actual pain scores improved only slightly – patient satisfaction scores improved markedly among post-ERAS patients. Patients gave higher marks, for instance, to questions regarding pain control (“how well your pain was controlled”) and teamwork (“staff worked together to care for you”).
Reducing Opioids
New opioid use that persists after a surgical procedure is a postsurgical complication that we all should be working to prevent. It has been estimated that 6% of surgical patients – even those who’ve had relatively minor surgical procedures – will become long-term opioid users, developing a dependence on the drugs prescribed to them for postsurgical pain.
This was shown last year in a national study of insurance claims data from between 2013 and 2014; investigators identified adults without opioid use in the year prior to surgery (including hysterectomy) and found that 5.9%-6.5% were filling opioid prescriptions 90-180 days after their surgical procedure. The incidence in a nonoperative control cohort was 0.4% (JAMA Surg. 2017 Jun 21;152[6]:e170504). Notably, this prolonged use was greatest in patients with prior pain conditions, substance abuse, and mental health disorders – a finding that may have implications for the counseling we provide prior to surgery.
It’s not clear what the optimal analgesic regimen is for minimally invasive or other gynecologic surgeries. What is clearly recommended, however, is that the approach be multifaceted. In our practice, we believe that the preoperative use of acetaminophen, pregabalin, and celecoxib plays an important role in reducing postoperative pain and opioid use. But we also have striven to create a practice-wide culture shift (throughout the operating and recovery rooms), for instance, that encourages using the least amounts of narcotics possible and using the shortest-acting formulations possible.
Transversus abdominis plane (TAP) blocks are also often part of ERAS protocols; they have been shown in at least two randomized controlled trials of abdominal hysterectomy to reduce intraoperative fentanyl requirements and to reduce immediate postoperative pain scores and postoperative morphine requirements (Anesth Analg. 2008 Dec;107[6]:2056-60; J Anaesthesiol Clin Pharmacol. 2014 Jul-Sep;30[3]:391-6).
More recently, liposomal bupivacaine, which is slowly released over several days, has gained traction as a substitute for standard bupivacaine and other agents in TAP blocks. In one recent retrospective study, abdominal incision infiltration with liposomal bupivacaine was associated with less opioid use (with no change in pain scores), compared with bupivacaine hydrochloride after laparotomy for gynecologic malignancies (Obstet Gynecol. 2016 Nov;128[5]:1009-17). It’s significantly more expensive, however, making it likely that the formulation is being used more judiciously in minimally invasive gynecologic surgery than in open surgeries.
Because of costs, we currently are restricted to using liposomal bupivacaine in our robotic surgeries only. In our practice, the single 20 mL vial (266 mg of liposomal bupivacaine) is diluted with 20 mL of normal saline, but it can be further diluted without loss of efficacy. With a 16-gauge needle, the liposomal bupivacaine is distributed across the incisions (usually 20 mL in the umbilicus with a larger incision and 10 mL in each of the two lateral incisions). Patients are counseled that they may have more discomfort after 3 days, but by this point most are mobile and feeling relatively well with a combination of NSAIDs and acetaminophen.
With growing visibility of the problem of narcotic dependence in the United States, patients seem increasingly receptive and even eager to limit or avoid the use of opioids. Patients should be counseled that minimizing or avoiding opioids may also speed recovery. Narcotics cause gut motility to slow down, which may hinder mobilization. Early mobilization (within 24 hours) is among the enhanced recovery elements that the ERAS Society guidelines say is “of particular value” for minimally invasive surgery, along with maintenance of normothermia and normovolemia with maintenance of adequate cardiac output.
Selecting Steps
Our practice is also trying to reduce preoperative bowel preparation and preoperative fasting, both of which have been found to be stressful for the body without evidence of benefit. These practices can lead to insulin resistance and hyperglycemia, which are associated with increased morbidity and length of stay.
It is now recommended that clear fluids be allowed up to 2 hours before surgery and solids up to 6 hours before. Some health systems and practices also recommend presurgical carbohydrate loading (for example, 10 ounces of apple juice 2 hours before surgery) – another small change on the ERAS menu – to further reduce postoperative insulin resistance and help the body cope with its stress response to surgery.
Along with nutritional changes are also various measures aimed at optimizing the body’s functionality before surgery (“prehabilitation”), from walking 30 minutes a day to abstaining from alcohol for patients who drink heavily.
Throughout the country, enhanced recovery protocols are taking shape in gynecologic surgery. ERAS was featured in an aptly titled panel session at the 2017 annual meeting of the American Association of Gynecologic Laparoscopists: “Outpatient Hysterectomy, ERAS, and Same-Day Discharge: The Next Big Thing in Gyn Surgery.” Others are applying ERAS to scheduled cesarean sections. And in our practice, I believe that if we continue making small changes, we will reach our goal of opioid-free recoveries and a better surgical experience for our patients.
Kirsten Sasaki, MD, is an associate of the Advanced Gynecologic Surgery Institute. She reported that she has no disclosures relevant to this Master Class.
“Enhanced Recovery After Surgery” (ERAS) practices and protocols have been increasingly refined and adopted for the field of gynecology, and there is hope among gynecologic surgeons – and some recent evidence – that, with the ERAS movement, we are improving patient recoveries and outcomes and minimizing the need for opioids.
This applies not only to open surgeries but also to the minimally invasive procedures that already are prized for significant reductions in morbidity and length of stay. The overarching and guiding principle of ERAS is that any surgery – whether open or minimally invasive, major or minor – places stress on the body and is associated with risks and morbidity.
Enhanced recovery protocols are multidisciplinary, perioperative approaches designed to lessen the body’s stress response to surgery. The protocols and pathways offer us a menu of small changes that, in the aggregate, can lead to large and demonstrable benefits – especially when these small changes are chosen across the preoperative, intraoperative, and postoperative arenas and then standardized in one’s practice. Among the major components of ERAS practices and protocols are limiting preoperative fasting, employing multimodal analgesia, encouraging early ambulation and early postsurgical feeding, and creating culture shift that includes greater emphasis on patient expectations.
In our practice, we are incorporating ERAS practices not only in hopes of reducing the stress of all surgeries before, during, and after, but also with the goal of achieving a postoperative opioid-free hysterectomy, myomectomy, and extensive endometriosis surgery. (All of our advanced procedures are performed laparoscopically or robotically.)
Over the past 7 or so years, we have adopted a multimodal approach to pain control that includes a bundle of preoperative analgesics – acetaminophen, pregabalin, and celecoxib (we call it “TLC” for Tylenol, Lyrica, and Celebrex) – and the use of liposomal bupivacaine in our robotic surgeries. We are now turning toward ERAS nutritional changes, most of which run counter to traditional paradigms for surgical care. And in other areas, such as dedicated preoperative counseling, we continue to refine and improve our practices.
Improved Outcomes
The ERAS mindset notably intersected gynecology with the publication in 2016 of a two-part series of guidelines for gynecology/oncology surgery from the ERAS Society. The 8-year-old society has its roots in a study group of European surgeons and others who decided to examine surgical practices and the concept of multimodal surgical care put forth in the 1990s by Henrik Kehlet, MD, PhD, then a professor at the University of Copenhagen.
The first set of recommendations addressed pre- and intraoperative care (Gynecol Oncol. 2016 Feb;140[2]:313-22), and the second set addressed postoperative care (Gynecol Oncol. 2016 Feb;140[2]:323-32). Similar evidence-based recommendations were previously written for colonic resections, rectal and pelvic surgery, and other surgical specialties.
Most of the published outcomes of enhanced recovery protocols come from colorectal surgery. As noted in the ERAS Society gynecology/oncology guidelines, the benefits include an average reduction in length of stay of 2.5 days and a decrease in complications by as much as 50%.
There is growing evidence, however, that ERAS programs are also beneficial for patients undergoing laparoscopic surgery, and outcomes from gynecology – including minimally invasive surgery – are also being reported.
For instance, a retrospective case-control study of 55 consecutive gynecologic oncology patients treated at the University of California, San Francisco, with laparoscopic or robotic surgery and an enhanced recovery pathway – and 110 historical control patients matched on the basis of age and surgery type – found significant improvements in recovery time, decreased pain despite reduced opioid use, and overall lower hospital costs (Obstet Gynecol. 2016 Jul;128[1]:138-44).
The enhanced recovery pathway included patient education, multimodal antiemetics, multimodal analgesia, and balanced fluid administration. Early catheter removal, ambulation, and feeding were also components. Analgesia included routine preoperative gabapentin, diclofenac, and acetaminophen; routine postoperative gabapentin, NSAIDs, and acetaminophen; and transversus abdominis plane blocks in 32 of the ERAS patients.
ERAS patients were significantly more likely to be discharged on day 1 (91%, compared with 60% in the control group). Opioid use decreased by 30%, and pain scores on postoperative day 1 were significantly lower.
Another study looking at the effect of enhanced recovery implementation in gynecologic surgeries at the University of Virginia, Charlottesville, (gynecologic oncology, urogynecology, and general gynecology) similarly reported benefits for vaginal and minimally invasive procedures, as well as for open procedures (Obstet Gynecol. 2016 Sep;128[3]:457-66).
In the minimally invasive group, investigators compared 324 patients before ERAS implementation with 249 patients afterward and found that the median length of stay was unchanged (1 day). However, intraoperative and postoperative opioid consumption decreased significantly and – even though actual pain scores improved only slightly – patient satisfaction scores improved markedly among post-ERAS patients. Patients gave higher marks, for instance, to questions regarding pain control (“how well your pain was controlled”) and teamwork (“staff worked together to care for you”).
Reducing Opioids
New opioid use that persists after a surgical procedure is a postsurgical complication that we all should be working to prevent. It has been estimated that 6% of surgical patients – even those who’ve had relatively minor surgical procedures – will become long-term opioid users, developing a dependence on the drugs prescribed to them for postsurgical pain.
This was shown last year in a national study of insurance claims data from between 2013 and 2014; investigators identified adults without opioid use in the year prior to surgery (including hysterectomy) and found that 5.9%-6.5% were filling opioid prescriptions 90-180 days after their surgical procedure. The incidence in a nonoperative control cohort was 0.4% (JAMA Surg. 2017 Jun 21;152[6]:e170504). Notably, this prolonged use was greatest in patients with prior pain conditions, substance abuse, and mental health disorders – a finding that may have implications for the counseling we provide prior to surgery.
It’s not clear what the optimal analgesic regimen is for minimally invasive or other gynecologic surgeries. What is clearly recommended, however, is that the approach be multifaceted. In our practice, we believe that the preoperative use of acetaminophen, pregabalin, and celecoxib plays an important role in reducing postoperative pain and opioid use. But we also have striven to create a practice-wide culture shift (throughout the operating and recovery rooms), for instance, that encourages using the least amounts of narcotics possible and using the shortest-acting formulations possible.
Transversus abdominis plane (TAP) blocks are also often part of ERAS protocols; they have been shown in at least two randomized controlled trials of abdominal hysterectomy to reduce intraoperative fentanyl requirements and to reduce immediate postoperative pain scores and postoperative morphine requirements (Anesth Analg. 2008 Dec;107[6]:2056-60; J Anaesthesiol Clin Pharmacol. 2014 Jul-Sep;30[3]:391-6).
More recently, liposomal bupivacaine, which is slowly released over several days, has gained traction as a substitute for standard bupivacaine and other agents in TAP blocks. In one recent retrospective study, abdominal incision infiltration with liposomal bupivacaine was associated with less opioid use (with no change in pain scores), compared with bupivacaine hydrochloride after laparotomy for gynecologic malignancies (Obstet Gynecol. 2016 Nov;128[5]:1009-17). It’s significantly more expensive, however, making it likely that the formulation is being used more judiciously in minimally invasive gynecologic surgery than in open surgeries.
Because of costs, we currently are restricted to using liposomal bupivacaine in our robotic surgeries only. In our practice, the single 20 mL vial (266 mg of liposomal bupivacaine) is diluted with 20 mL of normal saline, but it can be further diluted without loss of efficacy. With a 16-gauge needle, the liposomal bupivacaine is distributed across the incisions (usually 20 mL in the umbilicus with a larger incision and 10 mL in each of the two lateral incisions). Patients are counseled that they may have more discomfort after 3 days, but by this point most are mobile and feeling relatively well with a combination of NSAIDs and acetaminophen.
With growing visibility of the problem of narcotic dependence in the United States, patients seem increasingly receptive and even eager to limit or avoid the use of opioids. Patients should be counseled that minimizing or avoiding opioids may also speed recovery. Narcotics cause gut motility to slow down, which may hinder mobilization. Early mobilization (within 24 hours) is among the enhanced recovery elements that the ERAS Society guidelines say is “of particular value” for minimally invasive surgery, along with maintenance of normothermia and normovolemia with maintenance of adequate cardiac output.
Selecting Steps
Our practice is also trying to reduce preoperative bowel preparation and preoperative fasting, both of which have been found to be stressful for the body without evidence of benefit. These practices can lead to insulin resistance and hyperglycemia, which are associated with increased morbidity and length of stay.
It is now recommended that clear fluids be allowed up to 2 hours before surgery and solids up to 6 hours before. Some health systems and practices also recommend presurgical carbohydrate loading (for example, 10 ounces of apple juice 2 hours before surgery) – another small change on the ERAS menu – to further reduce postoperative insulin resistance and help the body cope with its stress response to surgery.
Along with nutritional changes are also various measures aimed at optimizing the body’s functionality before surgery (“prehabilitation”), from walking 30 minutes a day to abstaining from alcohol for patients who drink heavily.
Throughout the country, enhanced recovery protocols are taking shape in gynecologic surgery. ERAS was featured in an aptly titled panel session at the 2017 annual meeting of the American Association of Gynecologic Laparoscopists: “Outpatient Hysterectomy, ERAS, and Same-Day Discharge: The Next Big Thing in Gyn Surgery.” Others are applying ERAS to scheduled cesarean sections. And in our practice, I believe that if we continue making small changes, we will reach our goal of opioid-free recoveries and a better surgical experience for our patients.
Kirsten Sasaki, MD, is an associate of the Advanced Gynecologic Surgery Institute. She reported that she has no disclosures relevant to this Master Class.
“Enhanced Recovery After Surgery” (ERAS) practices and protocols have been increasingly refined and adopted for the field of gynecology, and there is hope among gynecologic surgeons – and some recent evidence – that, with the ERAS movement, we are improving patient recoveries and outcomes and minimizing the need for opioids.
This applies not only to open surgeries but also to the minimally invasive procedures that already are prized for significant reductions in morbidity and length of stay. The overarching and guiding principle of ERAS is that any surgery – whether open or minimally invasive, major or minor – places stress on the body and is associated with risks and morbidity.
Enhanced recovery protocols are multidisciplinary, perioperative approaches designed to lessen the body’s stress response to surgery. The protocols and pathways offer us a menu of small changes that, in the aggregate, can lead to large and demonstrable benefits – especially when these small changes are chosen across the preoperative, intraoperative, and postoperative arenas and then standardized in one’s practice. Among the major components of ERAS practices and protocols are limiting preoperative fasting, employing multimodal analgesia, encouraging early ambulation and early postsurgical feeding, and creating culture shift that includes greater emphasis on patient expectations.
In our practice, we are incorporating ERAS practices not only in hopes of reducing the stress of all surgeries before, during, and after, but also with the goal of achieving a postoperative opioid-free hysterectomy, myomectomy, and extensive endometriosis surgery. (All of our advanced procedures are performed laparoscopically or robotically.)
Over the past 7 or so years, we have adopted a multimodal approach to pain control that includes a bundle of preoperative analgesics – acetaminophen, pregabalin, and celecoxib (we call it “TLC” for Tylenol, Lyrica, and Celebrex) – and the use of liposomal bupivacaine in our robotic surgeries. We are now turning toward ERAS nutritional changes, most of which run counter to traditional paradigms for surgical care. And in other areas, such as dedicated preoperative counseling, we continue to refine and improve our practices.
Improved Outcomes
The ERAS mindset notably intersected gynecology with the publication in 2016 of a two-part series of guidelines for gynecology/oncology surgery from the ERAS Society. The 8-year-old society has its roots in a study group of European surgeons and others who decided to examine surgical practices and the concept of multimodal surgical care put forth in the 1990s by Henrik Kehlet, MD, PhD, then a professor at the University of Copenhagen.
The first set of recommendations addressed pre- and intraoperative care (Gynecol Oncol. 2016 Feb;140[2]:313-22), and the second set addressed postoperative care (Gynecol Oncol. 2016 Feb;140[2]:323-32). Similar evidence-based recommendations were previously written for colonic resections, rectal and pelvic surgery, and other surgical specialties.
Most of the published outcomes of enhanced recovery protocols come from colorectal surgery. As noted in the ERAS Society gynecology/oncology guidelines, the benefits include an average reduction in length of stay of 2.5 days and a decrease in complications by as much as 50%.
There is growing evidence, however, that ERAS programs are also beneficial for patients undergoing laparoscopic surgery, and outcomes from gynecology – including minimally invasive surgery – are also being reported.
For instance, a retrospective case-control study of 55 consecutive gynecologic oncology patients treated at the University of California, San Francisco, with laparoscopic or robotic surgery and an enhanced recovery pathway – and 110 historical control patients matched on the basis of age and surgery type – found significant improvements in recovery time, decreased pain despite reduced opioid use, and overall lower hospital costs (Obstet Gynecol. 2016 Jul;128[1]:138-44).
The enhanced recovery pathway included patient education, multimodal antiemetics, multimodal analgesia, and balanced fluid administration. Early catheter removal, ambulation, and feeding were also components. Analgesia included routine preoperative gabapentin, diclofenac, and acetaminophen; routine postoperative gabapentin, NSAIDs, and acetaminophen; and transversus abdominis plane blocks in 32 of the ERAS patients.
ERAS patients were significantly more likely to be discharged on day 1 (91%, compared with 60% in the control group). Opioid use decreased by 30%, and pain scores on postoperative day 1 were significantly lower.
Another study looking at the effect of enhanced recovery implementation in gynecologic surgeries at the University of Virginia, Charlottesville, (gynecologic oncology, urogynecology, and general gynecology) similarly reported benefits for vaginal and minimally invasive procedures, as well as for open procedures (Obstet Gynecol. 2016 Sep;128[3]:457-66).
In the minimally invasive group, investigators compared 324 patients before ERAS implementation with 249 patients afterward and found that the median length of stay was unchanged (1 day). However, intraoperative and postoperative opioid consumption decreased significantly and – even though actual pain scores improved only slightly – patient satisfaction scores improved markedly among post-ERAS patients. Patients gave higher marks, for instance, to questions regarding pain control (“how well your pain was controlled”) and teamwork (“staff worked together to care for you”).
Reducing Opioids
New opioid use that persists after a surgical procedure is a postsurgical complication that we all should be working to prevent. It has been estimated that 6% of surgical patients – even those who’ve had relatively minor surgical procedures – will become long-term opioid users, developing a dependence on the drugs prescribed to them for postsurgical pain.
This was shown last year in a national study of insurance claims data from between 2013 and 2014; investigators identified adults without opioid use in the year prior to surgery (including hysterectomy) and found that 5.9%-6.5% were filling opioid prescriptions 90-180 days after their surgical procedure. The incidence in a nonoperative control cohort was 0.4% (JAMA Surg. 2017 Jun 21;152[6]:e170504). Notably, this prolonged use was greatest in patients with prior pain conditions, substance abuse, and mental health disorders – a finding that may have implications for the counseling we provide prior to surgery.
It’s not clear what the optimal analgesic regimen is for minimally invasive or other gynecologic surgeries. What is clearly recommended, however, is that the approach be multifaceted. In our practice, we believe that the preoperative use of acetaminophen, pregabalin, and celecoxib plays an important role in reducing postoperative pain and opioid use. But we also have striven to create a practice-wide culture shift (throughout the operating and recovery rooms), for instance, that encourages using the least amounts of narcotics possible and using the shortest-acting formulations possible.
Transversus abdominis plane (TAP) blocks are also often part of ERAS protocols; they have been shown in at least two randomized controlled trials of abdominal hysterectomy to reduce intraoperative fentanyl requirements and to reduce immediate postoperative pain scores and postoperative morphine requirements (Anesth Analg. 2008 Dec;107[6]:2056-60; J Anaesthesiol Clin Pharmacol. 2014 Jul-Sep;30[3]:391-6).
More recently, liposomal bupivacaine, which is slowly released over several days, has gained traction as a substitute for standard bupivacaine and other agents in TAP blocks. In one recent retrospective study, abdominal incision infiltration with liposomal bupivacaine was associated with less opioid use (with no change in pain scores), compared with bupivacaine hydrochloride after laparotomy for gynecologic malignancies (Obstet Gynecol. 2016 Nov;128[5]:1009-17). It’s significantly more expensive, however, making it likely that the formulation is being used more judiciously in minimally invasive gynecologic surgery than in open surgeries.
Because of costs, we currently are restricted to using liposomal bupivacaine in our robotic surgeries only. In our practice, the single 20 mL vial (266 mg of liposomal bupivacaine) is diluted with 20 mL of normal saline, but it can be further diluted without loss of efficacy. With a 16-gauge needle, the liposomal bupivacaine is distributed across the incisions (usually 20 mL in the umbilicus with a larger incision and 10 mL in each of the two lateral incisions). Patients are counseled that they may have more discomfort after 3 days, but by this point most are mobile and feeling relatively well with a combination of NSAIDs and acetaminophen.
With growing visibility of the problem of narcotic dependence in the United States, patients seem increasingly receptive and even eager to limit or avoid the use of opioids. Patients should be counseled that minimizing or avoiding opioids may also speed recovery. Narcotics cause gut motility to slow down, which may hinder mobilization. Early mobilization (within 24 hours) is among the enhanced recovery elements that the ERAS Society guidelines say is “of particular value” for minimally invasive surgery, along with maintenance of normothermia and normovolemia with maintenance of adequate cardiac output.
Selecting Steps
Our practice is also trying to reduce preoperative bowel preparation and preoperative fasting, both of which have been found to be stressful for the body without evidence of benefit. These practices can lead to insulin resistance and hyperglycemia, which are associated with increased morbidity and length of stay.
It is now recommended that clear fluids be allowed up to 2 hours before surgery and solids up to 6 hours before. Some health systems and practices also recommend presurgical carbohydrate loading (for example, 10 ounces of apple juice 2 hours before surgery) – another small change on the ERAS menu – to further reduce postoperative insulin resistance and help the body cope with its stress response to surgery.
Along with nutritional changes are also various measures aimed at optimizing the body’s functionality before surgery (“prehabilitation”), from walking 30 minutes a day to abstaining from alcohol for patients who drink heavily.
Throughout the country, enhanced recovery protocols are taking shape in gynecologic surgery. ERAS was featured in an aptly titled panel session at the 2017 annual meeting of the American Association of Gynecologic Laparoscopists: “Outpatient Hysterectomy, ERAS, and Same-Day Discharge: The Next Big Thing in Gyn Surgery.” Others are applying ERAS to scheduled cesarean sections. And in our practice, I believe that if we continue making small changes, we will reach our goal of opioid-free recoveries and a better surgical experience for our patients.
Kirsten Sasaki, MD, is an associate of the Advanced Gynecologic Surgery Institute. She reported that she has no disclosures relevant to this Master Class.
You paid how much for that medicine?
This is the first article in a series on the cost of medications.
While it’s not news that some pharmaceuticals are exorbitantly expensive and therefore unavailable to our patients, I have learned that there are ways around the obstacle of cost for at least some medications. I want to tell you what I’ve learned about the high cost of two medications: aripiprazole, the generic of Abilify, and modafinil, the generic of Provigil, but the lessons learned may apply to other psychotropics – and Smokey Robinson and the Miracles said it best in a line from their 1960 hit song, “You’d better shop around.”
Over the past few years, the price of aripiprazole has come down considerably, or so I believed. A patient recently complained that his copay after insurance for a 1-month supply of 2-mg tablets was hundreds of dollars, and he showed me a bill where the cost before insurance was more than $2,000! Another patient, also someone with commercial insurance, said he couldn’t afford aripiprazole and asked me to phone in a prescription to healthwarehouse.com. The medication was mailed to him for about $35 a month. Finally, a third patient with Medicare used an online service called Blink Health. He explained that he paid for the medicine online with a credit card – about $80, far less than the price quoted by the pharmacy. He was then given some type of code to present to the pharmacist, who then supplied the medications. In this case, the same pills, the same pharmacy, at a fraction of the cost. With that, I called several pharmacies and asked about the price of generic Abilify, 5 mg, 30 tablets.
I also wrote a column about the tremendous difficulty I had trying to get preauthorization for modafinil, in “Preauthorization of medications: Who oversees the placement of the hoops?” In that case, I spent weeks trying to get approval for the medication, and in the end, it was not approved, and the patient was not able to get it. Soon after, I learned on a Psychiatry Network Facebook discussion that generic Provigil is not expensive at all! Once again, I fired up my phone and called around. Those prices and those I found for Abilify are listed in the chart.

So these are cash prices; they do not take into account the cost with insurance. My patients have educated me after they could not afford the insurance copays. I wondered about my own coverage, and signed on to my pharmacy benefits manager account to look up the cost of both Abilify and Provigil. I’m sorry to say that I can’t report back: With my health insurance, Empire Blue Cross and Express Scripts, both medications require Coverage Review. It was also noted that Abilify requires Step treatment; I must first fail other medications. Should I ever need either of these medications, you’ll know where to find me: in line at the Costco pharmacy. I’ll be the one dancing to Smokey Robinson and the Miracles. https://www.youtube.com/watch?v=AQGXa3FiXKM
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016).
This is the first article in a series on the cost of medications.
While it’s not news that some pharmaceuticals are exorbitantly expensive and therefore unavailable to our patients, I have learned that there are ways around the obstacle of cost for at least some medications. I want to tell you what I’ve learned about the high cost of two medications: aripiprazole, the generic of Abilify, and modafinil, the generic of Provigil, but the lessons learned may apply to other psychotropics – and Smokey Robinson and the Miracles said it best in a line from their 1960 hit song, “You’d better shop around.”
Over the past few years, the price of aripiprazole has come down considerably, or so I believed. A patient recently complained that his copay after insurance for a 1-month supply of 2-mg tablets was hundreds of dollars, and he showed me a bill where the cost before insurance was more than $2,000! Another patient, also someone with commercial insurance, said he couldn’t afford aripiprazole and asked me to phone in a prescription to healthwarehouse.com. The medication was mailed to him for about $35 a month. Finally, a third patient with Medicare used an online service called Blink Health. He explained that he paid for the medicine online with a credit card – about $80, far less than the price quoted by the pharmacy. He was then given some type of code to present to the pharmacist, who then supplied the medications. In this case, the same pills, the same pharmacy, at a fraction of the cost. With that, I called several pharmacies and asked about the price of generic Abilify, 5 mg, 30 tablets.
I also wrote a column about the tremendous difficulty I had trying to get preauthorization for modafinil, in “Preauthorization of medications: Who oversees the placement of the hoops?” In that case, I spent weeks trying to get approval for the medication, and in the end, it was not approved, and the patient was not able to get it. Soon after, I learned on a Psychiatry Network Facebook discussion that generic Provigil is not expensive at all! Once again, I fired up my phone and called around. Those prices and those I found for Abilify are listed in the chart.

So these are cash prices; they do not take into account the cost with insurance. My patients have educated me after they could not afford the insurance copays. I wondered about my own coverage, and signed on to my pharmacy benefits manager account to look up the cost of both Abilify and Provigil. I’m sorry to say that I can’t report back: With my health insurance, Empire Blue Cross and Express Scripts, both medications require Coverage Review. It was also noted that Abilify requires Step treatment; I must first fail other medications. Should I ever need either of these medications, you’ll know where to find me: in line at the Costco pharmacy. I’ll be the one dancing to Smokey Robinson and the Miracles. https://www.youtube.com/watch?v=AQGXa3FiXKM
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016).
This is the first article in a series on the cost of medications.
While it’s not news that some pharmaceuticals are exorbitantly expensive and therefore unavailable to our patients, I have learned that there are ways around the obstacle of cost for at least some medications. I want to tell you what I’ve learned about the high cost of two medications: aripiprazole, the generic of Abilify, and modafinil, the generic of Provigil, but the lessons learned may apply to other psychotropics – and Smokey Robinson and the Miracles said it best in a line from their 1960 hit song, “You’d better shop around.”
Over the past few years, the price of aripiprazole has come down considerably, or so I believed. A patient recently complained that his copay after insurance for a 1-month supply of 2-mg tablets was hundreds of dollars, and he showed me a bill where the cost before insurance was more than $2,000! Another patient, also someone with commercial insurance, said he couldn’t afford aripiprazole and asked me to phone in a prescription to healthwarehouse.com. The medication was mailed to him for about $35 a month. Finally, a third patient with Medicare used an online service called Blink Health. He explained that he paid for the medicine online with a credit card – about $80, far less than the price quoted by the pharmacy. He was then given some type of code to present to the pharmacist, who then supplied the medications. In this case, the same pills, the same pharmacy, at a fraction of the cost. With that, I called several pharmacies and asked about the price of generic Abilify, 5 mg, 30 tablets.
I also wrote a column about the tremendous difficulty I had trying to get preauthorization for modafinil, in “Preauthorization of medications: Who oversees the placement of the hoops?” In that case, I spent weeks trying to get approval for the medication, and in the end, it was not approved, and the patient was not able to get it. Soon after, I learned on a Psychiatry Network Facebook discussion that generic Provigil is not expensive at all! Once again, I fired up my phone and called around. Those prices and those I found for Abilify are listed in the chart.

So these are cash prices; they do not take into account the cost with insurance. My patients have educated me after they could not afford the insurance copays. I wondered about my own coverage, and signed on to my pharmacy benefits manager account to look up the cost of both Abilify and Provigil. I’m sorry to say that I can’t report back: With my health insurance, Empire Blue Cross and Express Scripts, both medications require Coverage Review. It was also noted that Abilify requires Step treatment; I must first fail other medications. Should I ever need either of these medications, you’ll know where to find me: in line at the Costco pharmacy. I’ll be the one dancing to Smokey Robinson and the Miracles. https://www.youtube.com/watch?v=AQGXa3FiXKM
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016).
Hungry or what?
“She will eat when she is hungry.” That in so many words is the mantra of grandparents blessed with experience and common sense and of most pediatricians when consulting parents challenged with a picky eater. From birth, children understand the simple equation that to survive they must eat. With rare exception, the motivating power of hunger can be leveraged for success even with infants who have spent their first months relying on enteral feedings. I have written an entire book based solely on the premise that if you present a young child food she will eat it ... eventually (“Coping With a Picky Eater: A Guide for the Perplexed Parent” New York: Simon and Schuster, 1998).
But if we reverse the words to read, “When she is eating, she is hungry,” do we have an equally valid observation? I think we have ample evidence that it is not.
The result was a year-long odyssey of pumping that included consultations with five different lactation consultants in the first frustrating month and a half. She eventually received some comforting advice from a pediatrician who reassured her that there was little research to guide her and to “just feed him; trust your instincts.”
While it is unfortunately true that there is very little good science we can fall back on when counseling women who are struggling with breastfeeding, I wonder about the wisdom of telling this mother to trust her instincts. I guess my hesitancy is based on 40 years of primary care pediatrics in which I could generally count on the instincts of young children, but their parents’ not so much. While maternal intuition is generally superior to the paternal version, I am hesitant to rely totally on either when facing a clinical dilemma such as defining hunger.
What about the fussy baby who is comforted by just a pacifier? What is the difference? There are several explanations, but it will require introducing the concept of nutrition deficiency.
Most babies who are satisfied with just a nipple, be it silicone or flesh, simply find sucking a comfort measure. A few, and I am sure you have seen some of them, are overly patient. They seem to be saying, “I need the calories, but you’re a good mom and I enjoy sucking. I can wait. Some day, you may make more milk or give me a bottle.” In the worst-case scenarios, their patience leaves these babies so nutritionally deficient that they can slip into apathy and die.
There are no easy answers. As pediatricians, our job is to sort out those fussy “hungry” babies whose behavior means they are overtired from those who are nutritionally deficient, from those with a dysfunctional satiety center. Making the differentiation is difficult but much easier than helping parents ignore one of their instincts.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
“She will eat when she is hungry.” That in so many words is the mantra of grandparents blessed with experience and common sense and of most pediatricians when consulting parents challenged with a picky eater. From birth, children understand the simple equation that to survive they must eat. With rare exception, the motivating power of hunger can be leveraged for success even with infants who have spent their first months relying on enteral feedings. I have written an entire book based solely on the premise that if you present a young child food she will eat it ... eventually (“Coping With a Picky Eater: A Guide for the Perplexed Parent” New York: Simon and Schuster, 1998).
But if we reverse the words to read, “When she is eating, she is hungry,” do we have an equally valid observation? I think we have ample evidence that it is not.
The result was a year-long odyssey of pumping that included consultations with five different lactation consultants in the first frustrating month and a half. She eventually received some comforting advice from a pediatrician who reassured her that there was little research to guide her and to “just feed him; trust your instincts.”
While it is unfortunately true that there is very little good science we can fall back on when counseling women who are struggling with breastfeeding, I wonder about the wisdom of telling this mother to trust her instincts. I guess my hesitancy is based on 40 years of primary care pediatrics in which I could generally count on the instincts of young children, but their parents’ not so much. While maternal intuition is generally superior to the paternal version, I am hesitant to rely totally on either when facing a clinical dilemma such as defining hunger.
What about the fussy baby who is comforted by just a pacifier? What is the difference? There are several explanations, but it will require introducing the concept of nutrition deficiency.
Most babies who are satisfied with just a nipple, be it silicone or flesh, simply find sucking a comfort measure. A few, and I am sure you have seen some of them, are overly patient. They seem to be saying, “I need the calories, but you’re a good mom and I enjoy sucking. I can wait. Some day, you may make more milk or give me a bottle.” In the worst-case scenarios, their patience leaves these babies so nutritionally deficient that they can slip into apathy and die.
There are no easy answers. As pediatricians, our job is to sort out those fussy “hungry” babies whose behavior means they are overtired from those who are nutritionally deficient, from those with a dysfunctional satiety center. Making the differentiation is difficult but much easier than helping parents ignore one of their instincts.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
“She will eat when she is hungry.” That in so many words is the mantra of grandparents blessed with experience and common sense and of most pediatricians when consulting parents challenged with a picky eater. From birth, children understand the simple equation that to survive they must eat. With rare exception, the motivating power of hunger can be leveraged for success even with infants who have spent their first months relying on enteral feedings. I have written an entire book based solely on the premise that if you present a young child food she will eat it ... eventually (“Coping With a Picky Eater: A Guide for the Perplexed Parent” New York: Simon and Schuster, 1998).
But if we reverse the words to read, “When she is eating, she is hungry,” do we have an equally valid observation? I think we have ample evidence that it is not.
The result was a year-long odyssey of pumping that included consultations with five different lactation consultants in the first frustrating month and a half. She eventually received some comforting advice from a pediatrician who reassured her that there was little research to guide her and to “just feed him; trust your instincts.”
While it is unfortunately true that there is very little good science we can fall back on when counseling women who are struggling with breastfeeding, I wonder about the wisdom of telling this mother to trust her instincts. I guess my hesitancy is based on 40 years of primary care pediatrics in which I could generally count on the instincts of young children, but their parents’ not so much. While maternal intuition is generally superior to the paternal version, I am hesitant to rely totally on either when facing a clinical dilemma such as defining hunger.
What about the fussy baby who is comforted by just a pacifier? What is the difference? There are several explanations, but it will require introducing the concept of nutrition deficiency.
Most babies who are satisfied with just a nipple, be it silicone or flesh, simply find sucking a comfort measure. A few, and I am sure you have seen some of them, are overly patient. They seem to be saying, “I need the calories, but you’re a good mom and I enjoy sucking. I can wait. Some day, you may make more milk or give me a bottle.” In the worst-case scenarios, their patience leaves these babies so nutritionally deficient that they can slip into apathy and die.
There are no easy answers. As pediatricians, our job is to sort out those fussy “hungry” babies whose behavior means they are overtired from those who are nutritionally deficient, from those with a dysfunctional satiety center. Making the differentiation is difficult but much easier than helping parents ignore one of their instincts.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
Protocols to reduce opioid use and shorten length of stay
While originally pioneered by European anesthesiologists and surgeons in Europe in the 1990s, enhanced recovery after surgery (ERAS) programs, also known as enhanced recovery protocols or fast-track surgery, have now gained popularity across the surgical spectrum within the United States. The goal of these programs is to utilize multidisciplinary and multimodal interventions to minimize the physiologic changes associated with surgery and thereby enhance the perioperative experience – reduced morbidity and mortality, shorter length of stay, less postoperative opioid use, and faster resumption to normal activity, at a decreased cost of care.
1. Enhanced patient education, including managing expectations.
2. Decreased perioperative fasting periods.
3. Blood volume and temperature maintenance intraoperatively.
4. Postoperative mobilization early and often.
5. Multimodal pain relief and nausea/vomiting prophylaxis.
6. Use of postoperative drains and catheters only as long as required.
Today, I have asked Kirsten Sasaki, MD, to discuss some of these ERAS concepts. I have asked Dr. Sasaki to especially focus on decreasing opioid utilization. For a thorough discussion on ERAS recommendations using an evidence-based approach, one can review two excellent papers by Nelson et al. (Gynecol Oncol. 2016 Feb;140[2]:313-22; Gynecol Oncol. 2016 Feb;140[2]:323-32).
Dr. Sasaki completed her internship and residency at Tufts Medical Center, Boston. Dr. Sasaki then went on to become our second fellow at the Fellowship in Minimally Invasive Gynecologic Surgery in affiliation with AAGL and Society of Reproductive Surgeons at Advocate Lutheran General Hospital, Park Ridge, Ill. As a Fellow, Dr. Sasaki was recognized for her excellent teaching and research capabilities. Ultimately, however, it was her tremendous surgical skills and surgical sense that led me to invite her to join my practice in 2014.
Dr. Miller is a clinical associate professor at the University of Illinois in Chicago and past president of the AAGL. He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in metropolitan Chicago and the director of minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill. He has no disclosures relevant to this Master Class.
While originally pioneered by European anesthesiologists and surgeons in Europe in the 1990s, enhanced recovery after surgery (ERAS) programs, also known as enhanced recovery protocols or fast-track surgery, have now gained popularity across the surgical spectrum within the United States. The goal of these programs is to utilize multidisciplinary and multimodal interventions to minimize the physiologic changes associated with surgery and thereby enhance the perioperative experience – reduced morbidity and mortality, shorter length of stay, less postoperative opioid use, and faster resumption to normal activity, at a decreased cost of care.
1. Enhanced patient education, including managing expectations.
2. Decreased perioperative fasting periods.
3. Blood volume and temperature maintenance intraoperatively.
4. Postoperative mobilization early and often.
5. Multimodal pain relief and nausea/vomiting prophylaxis.
6. Use of postoperative drains and catheters only as long as required.
Today, I have asked Kirsten Sasaki, MD, to discuss some of these ERAS concepts. I have asked Dr. Sasaki to especially focus on decreasing opioid utilization. For a thorough discussion on ERAS recommendations using an evidence-based approach, one can review two excellent papers by Nelson et al. (Gynecol Oncol. 2016 Feb;140[2]:313-22; Gynecol Oncol. 2016 Feb;140[2]:323-32).
Dr. Sasaki completed her internship and residency at Tufts Medical Center, Boston. Dr. Sasaki then went on to become our second fellow at the Fellowship in Minimally Invasive Gynecologic Surgery in affiliation with AAGL and Society of Reproductive Surgeons at Advocate Lutheran General Hospital, Park Ridge, Ill. As a Fellow, Dr. Sasaki was recognized for her excellent teaching and research capabilities. Ultimately, however, it was her tremendous surgical skills and surgical sense that led me to invite her to join my practice in 2014.
Dr. Miller is a clinical associate professor at the University of Illinois in Chicago and past president of the AAGL. He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in metropolitan Chicago and the director of minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill. He has no disclosures relevant to this Master Class.
While originally pioneered by European anesthesiologists and surgeons in Europe in the 1990s, enhanced recovery after surgery (ERAS) programs, also known as enhanced recovery protocols or fast-track surgery, have now gained popularity across the surgical spectrum within the United States. The goal of these programs is to utilize multidisciplinary and multimodal interventions to minimize the physiologic changes associated with surgery and thereby enhance the perioperative experience – reduced morbidity and mortality, shorter length of stay, less postoperative opioid use, and faster resumption to normal activity, at a decreased cost of care.
1. Enhanced patient education, including managing expectations.
2. Decreased perioperative fasting periods.
3. Blood volume and temperature maintenance intraoperatively.
4. Postoperative mobilization early and often.
5. Multimodal pain relief and nausea/vomiting prophylaxis.
6. Use of postoperative drains and catheters only as long as required.
Today, I have asked Kirsten Sasaki, MD, to discuss some of these ERAS concepts. I have asked Dr. Sasaki to especially focus on decreasing opioid utilization. For a thorough discussion on ERAS recommendations using an evidence-based approach, one can review two excellent papers by Nelson et al. (Gynecol Oncol. 2016 Feb;140[2]:313-22; Gynecol Oncol. 2016 Feb;140[2]:323-32).
Dr. Sasaki completed her internship and residency at Tufts Medical Center, Boston. Dr. Sasaki then went on to become our second fellow at the Fellowship in Minimally Invasive Gynecologic Surgery in affiliation with AAGL and Society of Reproductive Surgeons at Advocate Lutheran General Hospital, Park Ridge, Ill. As a Fellow, Dr. Sasaki was recognized for her excellent teaching and research capabilities. Ultimately, however, it was her tremendous surgical skills and surgical sense that led me to invite her to join my practice in 2014.
Dr. Miller is a clinical associate professor at the University of Illinois in Chicago and past president of the AAGL. He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in metropolitan Chicago and the director of minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill. He has no disclosures relevant to this Master Class.
Psychiatry Innovation Lab nurtures young businesses
The future of mental health is in our own hands. As psychiatrists, we have a wealth of experience to fuel transformation in our field and improve care for our patients. What we lack is the space in which to share our ideas and turn them into tangible solutions like sustainable businesses, diagnostic devices, mobile apps, or prevention campaigns.
The Psychiatry Innovation Lab provides just that – it is an incubator nurturing early-stage ideas and ventures by investing in them through mentorship, education, funding, and collaboration opportunities within a community of mental health innovators.
The lab’s main event is on Sunday, May 6, 2018, when leaders in psychiatry will gather alongside industry leaders at the Jacob Javits Convention Center in New York City during the American Psychiatric Association meeting for this fourth annual live event . At the event, finalists will pitch ideas “Shark Tank” style to a panel of expert judges. Finalists will work closely with industry leaders and audience members to fine-tune their ideas before pitching their revisions to the judges as well as to a live, voting audience, who together select the winners of cash awards and other prizes.
The Psychiatry Innovation Lab was founded in 2015 by Nina Vasan, MD, a Stanford (Calif.) University psychiatrist and author of Do Good Well: Your Guide to Leadership, Action, and Social Innovation (San Francisco: Jossey-Bass, 2013), with the purpose of catalyzing more entrepreneurship and innovation in mental health.
“The most meaningful part of the lab has been the impact that these ventures have had on the patients that we serve,” said Dr. Vasan, chair of the Psychiatry Innovation Lab and director of Brainstorm, the Stanford Laboratory for Brain Health Innovation and Entrepreneurship. “Year after year, psychiatrists come back and tell us that they are looking at their own patient care differently now. They’re looking for ways to be innovative, and we’re giving them the tools necessary to turn their clinical expertise into successful ventures.”
Applications are currently open online to pitch an idea in this year’s lab. Applications consist of brief answers to questions and a short YouTube video. The deadline for submissions is Friday, March 9, 2018. Anyone with innovative ideas, regardless of the stage, is invited to apply. The APA Workgroup on Psychiatry Innovation, which oversees the lab, welcomes ideas from people pitching for the first time to those working on their ideas full time. They noted that past finalists have come from all experience levels and a wide range of backgrounds, including students, CEOs, engineers, and psychiatrists.
Past winners and participants have come from a range of stages, from early start-ups to established companies, including NeuroLex Labs, which uses voice samples to detect behavioral health illness; Spring, which uses machine learning to determine the best depression treatment; Muse, which creates an EEG sensing headband to aid mindfulness practice; and Overdose Recovery Bracelet, which builds a wristband to deliver naloxone to people who have overdosed on opiates.
The diversity of ideas has been remarkable; ideas in the past have ranged from traditional for-profit businesses to nonprofits, educational and public health programs, and proposals for government reform. They address problems at all parts of the health care spectrum: prevention, diagnosis, treatment, systems, EMRs, and more.
“Many past ideas have revolved around technology such as artificial intelligence, social networking, or telemedicine,” Dr. Vasan said. “. Those ideas are especially important in solving the most pressing problems in our field today.”
In addition to those competing, anyone registered to attend the APA annual meeting can attend the event as an audience member or join a team and help as an innovation leader during the coaching portion of the program. Last year alone, more than 70% of the participants in the lab said that it was better than other educational events they have attended, 95% of participants found it useful, and participants made an average of six new contacts through the event.
“The Psychiatry Innovation Lab connected me to people who continue to inspire and educate me toward making a real impact with my own endeavors,” said Cody Rall, MD, founder of the YouTube channel Techforpsych and a 2016 semifinalist.
“The lab has grown dramatically. When we started we worked hard to find just one sponsor; last year we had 10 sponsors and over $25,000 in cash and prizes.” Dr. Vasan said. “We also had our first international finalist from Pakistan. The commitment to solving problems in mental health is taking off, and we want to ensure that psychiatrists are front and center in leading the change.”
For more information about joining the innovation lab or other ways to be involved, including sponsorship and judging, visit https://www.psychiatry.org/innovation or email Nina Taylor, APA deputy director of education, at [email protected].
This article was updated January 23, 2018.
Dr. Chaudhary is a board member of the APA Psychiatry Innovation Lab and the APA Workgroup on Psychiatry Innovation; chief resident in child and adolescent psychiatry at Massachusetts General Hospital/McLean and clinical fellow at Harvard Medical School, both in Boston; and founding partner of Brainstorm: Stanford Laboratory for Brain Health Innovation and Entrepreneurship. Dr. Ghomi is a board member of the APA Psychiatry Innovation Lab and the APA Workgroup on Psychiatry Innovation; resident in psychiatry at the University of Washington, Seattle; chief medical officer at NeuroLex; and founding partner of Brainstorm: Stanford Laboratory for Brain Health Innovation and Entrepreneurship.
The future of mental health is in our own hands. As psychiatrists, we have a wealth of experience to fuel transformation in our field and improve care for our patients. What we lack is the space in which to share our ideas and turn them into tangible solutions like sustainable businesses, diagnostic devices, mobile apps, or prevention campaigns.
The Psychiatry Innovation Lab provides just that – it is an incubator nurturing early-stage ideas and ventures by investing in them through mentorship, education, funding, and collaboration opportunities within a community of mental health innovators.
The lab’s main event is on Sunday, May 6, 2018, when leaders in psychiatry will gather alongside industry leaders at the Jacob Javits Convention Center in New York City during the American Psychiatric Association meeting for this fourth annual live event . At the event, finalists will pitch ideas “Shark Tank” style to a panel of expert judges. Finalists will work closely with industry leaders and audience members to fine-tune their ideas before pitching their revisions to the judges as well as to a live, voting audience, who together select the winners of cash awards and other prizes.
The Psychiatry Innovation Lab was founded in 2015 by Nina Vasan, MD, a Stanford (Calif.) University psychiatrist and author of Do Good Well: Your Guide to Leadership, Action, and Social Innovation (San Francisco: Jossey-Bass, 2013), with the purpose of catalyzing more entrepreneurship and innovation in mental health.
“The most meaningful part of the lab has been the impact that these ventures have had on the patients that we serve,” said Dr. Vasan, chair of the Psychiatry Innovation Lab and director of Brainstorm, the Stanford Laboratory for Brain Health Innovation and Entrepreneurship. “Year after year, psychiatrists come back and tell us that they are looking at their own patient care differently now. They’re looking for ways to be innovative, and we’re giving them the tools necessary to turn their clinical expertise into successful ventures.”
Applications are currently open online to pitch an idea in this year’s lab. Applications consist of brief answers to questions and a short YouTube video. The deadline for submissions is Friday, March 9, 2018. Anyone with innovative ideas, regardless of the stage, is invited to apply. The APA Workgroup on Psychiatry Innovation, which oversees the lab, welcomes ideas from people pitching for the first time to those working on their ideas full time. They noted that past finalists have come from all experience levels and a wide range of backgrounds, including students, CEOs, engineers, and psychiatrists.
Past winners and participants have come from a range of stages, from early start-ups to established companies, including NeuroLex Labs, which uses voice samples to detect behavioral health illness; Spring, which uses machine learning to determine the best depression treatment; Muse, which creates an EEG sensing headband to aid mindfulness practice; and Overdose Recovery Bracelet, which builds a wristband to deliver naloxone to people who have overdosed on opiates.
The diversity of ideas has been remarkable; ideas in the past have ranged from traditional for-profit businesses to nonprofits, educational and public health programs, and proposals for government reform. They address problems at all parts of the health care spectrum: prevention, diagnosis, treatment, systems, EMRs, and more.
“Many past ideas have revolved around technology such as artificial intelligence, social networking, or telemedicine,” Dr. Vasan said. “. Those ideas are especially important in solving the most pressing problems in our field today.”
In addition to those competing, anyone registered to attend the APA annual meeting can attend the event as an audience member or join a team and help as an innovation leader during the coaching portion of the program. Last year alone, more than 70% of the participants in the lab said that it was better than other educational events they have attended, 95% of participants found it useful, and participants made an average of six new contacts through the event.
“The Psychiatry Innovation Lab connected me to people who continue to inspire and educate me toward making a real impact with my own endeavors,” said Cody Rall, MD, founder of the YouTube channel Techforpsych and a 2016 semifinalist.
“The lab has grown dramatically. When we started we worked hard to find just one sponsor; last year we had 10 sponsors and over $25,000 in cash and prizes.” Dr. Vasan said. “We also had our first international finalist from Pakistan. The commitment to solving problems in mental health is taking off, and we want to ensure that psychiatrists are front and center in leading the change.”
For more information about joining the innovation lab or other ways to be involved, including sponsorship and judging, visit https://www.psychiatry.org/innovation or email Nina Taylor, APA deputy director of education, at [email protected].
This article was updated January 23, 2018.
Dr. Chaudhary is a board member of the APA Psychiatry Innovation Lab and the APA Workgroup on Psychiatry Innovation; chief resident in child and adolescent psychiatry at Massachusetts General Hospital/McLean and clinical fellow at Harvard Medical School, both in Boston; and founding partner of Brainstorm: Stanford Laboratory for Brain Health Innovation and Entrepreneurship. Dr. Ghomi is a board member of the APA Psychiatry Innovation Lab and the APA Workgroup on Psychiatry Innovation; resident in psychiatry at the University of Washington, Seattle; chief medical officer at NeuroLex; and founding partner of Brainstorm: Stanford Laboratory for Brain Health Innovation and Entrepreneurship.
The future of mental health is in our own hands. As psychiatrists, we have a wealth of experience to fuel transformation in our field and improve care for our patients. What we lack is the space in which to share our ideas and turn them into tangible solutions like sustainable businesses, diagnostic devices, mobile apps, or prevention campaigns.
The Psychiatry Innovation Lab provides just that – it is an incubator nurturing early-stage ideas and ventures by investing in them through mentorship, education, funding, and collaboration opportunities within a community of mental health innovators.
The lab’s main event is on Sunday, May 6, 2018, when leaders in psychiatry will gather alongside industry leaders at the Jacob Javits Convention Center in New York City during the American Psychiatric Association meeting for this fourth annual live event . At the event, finalists will pitch ideas “Shark Tank” style to a panel of expert judges. Finalists will work closely with industry leaders and audience members to fine-tune their ideas before pitching their revisions to the judges as well as to a live, voting audience, who together select the winners of cash awards and other prizes.
The Psychiatry Innovation Lab was founded in 2015 by Nina Vasan, MD, a Stanford (Calif.) University psychiatrist and author of Do Good Well: Your Guide to Leadership, Action, and Social Innovation (San Francisco: Jossey-Bass, 2013), with the purpose of catalyzing more entrepreneurship and innovation in mental health.
“The most meaningful part of the lab has been the impact that these ventures have had on the patients that we serve,” said Dr. Vasan, chair of the Psychiatry Innovation Lab and director of Brainstorm, the Stanford Laboratory for Brain Health Innovation and Entrepreneurship. “Year after year, psychiatrists come back and tell us that they are looking at their own patient care differently now. They’re looking for ways to be innovative, and we’re giving them the tools necessary to turn their clinical expertise into successful ventures.”
Applications are currently open online to pitch an idea in this year’s lab. Applications consist of brief answers to questions and a short YouTube video. The deadline for submissions is Friday, March 9, 2018. Anyone with innovative ideas, regardless of the stage, is invited to apply. The APA Workgroup on Psychiatry Innovation, which oversees the lab, welcomes ideas from people pitching for the first time to those working on their ideas full time. They noted that past finalists have come from all experience levels and a wide range of backgrounds, including students, CEOs, engineers, and psychiatrists.
Past winners and participants have come from a range of stages, from early start-ups to established companies, including NeuroLex Labs, which uses voice samples to detect behavioral health illness; Spring, which uses machine learning to determine the best depression treatment; Muse, which creates an EEG sensing headband to aid mindfulness practice; and Overdose Recovery Bracelet, which builds a wristband to deliver naloxone to people who have overdosed on opiates.
The diversity of ideas has been remarkable; ideas in the past have ranged from traditional for-profit businesses to nonprofits, educational and public health programs, and proposals for government reform. They address problems at all parts of the health care spectrum: prevention, diagnosis, treatment, systems, EMRs, and more.
“Many past ideas have revolved around technology such as artificial intelligence, social networking, or telemedicine,” Dr. Vasan said. “. Those ideas are especially important in solving the most pressing problems in our field today.”
In addition to those competing, anyone registered to attend the APA annual meeting can attend the event as an audience member or join a team and help as an innovation leader during the coaching portion of the program. Last year alone, more than 70% of the participants in the lab said that it was better than other educational events they have attended, 95% of participants found it useful, and participants made an average of six new contacts through the event.
“The Psychiatry Innovation Lab connected me to people who continue to inspire and educate me toward making a real impact with my own endeavors,” said Cody Rall, MD, founder of the YouTube channel Techforpsych and a 2016 semifinalist.
“The lab has grown dramatically. When we started we worked hard to find just one sponsor; last year we had 10 sponsors and over $25,000 in cash and prizes.” Dr. Vasan said. “We also had our first international finalist from Pakistan. The commitment to solving problems in mental health is taking off, and we want to ensure that psychiatrists are front and center in leading the change.”
For more information about joining the innovation lab or other ways to be involved, including sponsorship and judging, visit https://www.psychiatry.org/innovation or email Nina Taylor, APA deputy director of education, at [email protected].
This article was updated January 23, 2018.
Dr. Chaudhary is a board member of the APA Psychiatry Innovation Lab and the APA Workgroup on Psychiatry Innovation; chief resident in child and adolescent psychiatry at Massachusetts General Hospital/McLean and clinical fellow at Harvard Medical School, both in Boston; and founding partner of Brainstorm: Stanford Laboratory for Brain Health Innovation and Entrepreneurship. Dr. Ghomi is a board member of the APA Psychiatry Innovation Lab and the APA Workgroup on Psychiatry Innovation; resident in psychiatry at the University of Washington, Seattle; chief medical officer at NeuroLex; and founding partner of Brainstorm: Stanford Laboratory for Brain Health Innovation and Entrepreneurship.
Sentinel node biopsy: Who needs it?
More than 17 years ago, I published an article that was largely ignored, predicating that patient benefit from the sentinel node biopsy procedure was unlikely.
I asserted that the lymph nodes are not a reliable filter for melanoma cells, lymphatic drainage is capricious, and many individuals (especially younger ones) have benign neval rests in their lymph nodes that cannot be distinguished from melanoma deposits, since they are both positive for the S100 protein (Int J Dermatol. 2000 Nov;39[11]:807-11). In addition, multiple uncontrolled studies had shown that locating sentinel nodes, followed by a complete lymph node dissection, had no survival benefit. At the time, I argued that sentinel node biopsy should be performed only if the patient was going to be enrolled in a clinical study.
Many surgical oncologists have built their careers around the flawed premise that removing the draining nodes would cure melanoma. It doesn’t. It is past time to admit it and move on.
So, if completion node dissection does not save lives, why do a sentinel node biopsy? I recently asked a dermatologist friend, who is a committed acolyte of the sentinel node biopsy school, why he continues to recommend sentinel node biopsy if there is no benefit from complete node dissection. His quick response was that patients want to know if they are at higher risk of metastatic disease so that they can be followed closely with high-resolution ultrasound at a major cancer center and can be eligible for clinical trials. His reply gave me pause, so I asked why completion node dissection was still being recommended. I was told that some patients with positive sentinel nodes lived far away, and if they would not make regular follow-up visits for high-resolution ultrasound, the surgical oncologists do completion node dissection to ensure “local nodal control.” Yipes! You’re going to rip my groin out because I like quiet county living?
I doubt that patients would be enthusiastic if told beforehand that sentinel node biopsies costs $14,000-$18,000, and has a 9% complication rate, and one-third of those patients who have complications end up with permanent lymphedema. I wondered if the patients were told they could have a genetic test done on their already excised melanoma tissue that would tell them if they were in a high-risk group without having an additional invasive surgical procedure. I wondered if they were told that 10%-30% of people with negative sentinel nodes go on to develop metastatic disease. I also wondered if they had been told they would have to walk around with their melanoma, which could spread at any time, for several additional weeks, while waiting for the results of their sentinel node biopsy, instead of having the melanoma immediately removed by their dermatologist. I also wondered if they had been told that high-resolution ultrasound has not definitively been shown to be superior to clinical palpation of the lymph nodes.
I looked into the possibility of clinical trials for patients with positive sentinel nodes, as well. Based on my search of clinical trials.gov in January, there are 33 trials in the United States studying patients with stage 3 (positive sentinel node) or greater disease. If I had a positive sentinel node, I would look for a study in which I had a chance of getting nivolumab, which recently has been shown to be superior to ipilimumab in the phase 3 Checkmate 238 trial published in 2017 (N Engl J Med 2017 Nov 9; 377:1824-35).
But I am getting ahead of myself.
As a thinking man, if I had a thick melanoma (that was less than 2 mm), I would opt for a genetic test of my already excised melanoma tissue. If the results of that genetic test (which has near identical sensitivity and specificity for developing metastatic disease as a sentinel node) put me in the low-risk group, I would pass on the sentinel node biopsy. This would eliminate a lot of unnecessary surgery. If I fell into the high-risk group, I would consider a sentinel node biopsy so I could get into a study, or determine if I needed to find a way to get my insurance to pay, or if I could personally afford nivolumab. Even if I opted not to take the drug, because of the potential risk of high-grade side effects, the high-risk genetic profile tells me I would still need more frequent follow-up.
These are exciting times. I am looking forward to clinical trials that allow a patient with a high-risk genetic profile to go directly into a trial. We are moving into the realm of individualized genomic medicine in which metastatic melanoma truly becomes a curable disease.
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Dr. Coldiron has no financial or other conflicts of interest with Castle Biosciences, the manufacturer of the DecisionDx-Melanoma genetic expression profile test. Email him at [email protected].
More than 17 years ago, I published an article that was largely ignored, predicating that patient benefit from the sentinel node biopsy procedure was unlikely.
I asserted that the lymph nodes are not a reliable filter for melanoma cells, lymphatic drainage is capricious, and many individuals (especially younger ones) have benign neval rests in their lymph nodes that cannot be distinguished from melanoma deposits, since they are both positive for the S100 protein (Int J Dermatol. 2000 Nov;39[11]:807-11). In addition, multiple uncontrolled studies had shown that locating sentinel nodes, followed by a complete lymph node dissection, had no survival benefit. At the time, I argued that sentinel node biopsy should be performed only if the patient was going to be enrolled in a clinical study.
Many surgical oncologists have built their careers around the flawed premise that removing the draining nodes would cure melanoma. It doesn’t. It is past time to admit it and move on.
So, if completion node dissection does not save lives, why do a sentinel node biopsy? I recently asked a dermatologist friend, who is a committed acolyte of the sentinel node biopsy school, why he continues to recommend sentinel node biopsy if there is no benefit from complete node dissection. His quick response was that patients want to know if they are at higher risk of metastatic disease so that they can be followed closely with high-resolution ultrasound at a major cancer center and can be eligible for clinical trials. His reply gave me pause, so I asked why completion node dissection was still being recommended. I was told that some patients with positive sentinel nodes lived far away, and if they would not make regular follow-up visits for high-resolution ultrasound, the surgical oncologists do completion node dissection to ensure “local nodal control.” Yipes! You’re going to rip my groin out because I like quiet county living?
I doubt that patients would be enthusiastic if told beforehand that sentinel node biopsies costs $14,000-$18,000, and has a 9% complication rate, and one-third of those patients who have complications end up with permanent lymphedema. I wondered if the patients were told they could have a genetic test done on their already excised melanoma tissue that would tell them if they were in a high-risk group without having an additional invasive surgical procedure. I wondered if they were told that 10%-30% of people with negative sentinel nodes go on to develop metastatic disease. I also wondered if they had been told they would have to walk around with their melanoma, which could spread at any time, for several additional weeks, while waiting for the results of their sentinel node biopsy, instead of having the melanoma immediately removed by their dermatologist. I also wondered if they had been told that high-resolution ultrasound has not definitively been shown to be superior to clinical palpation of the lymph nodes.
I looked into the possibility of clinical trials for patients with positive sentinel nodes, as well. Based on my search of clinical trials.gov in January, there are 33 trials in the United States studying patients with stage 3 (positive sentinel node) or greater disease. If I had a positive sentinel node, I would look for a study in which I had a chance of getting nivolumab, which recently has been shown to be superior to ipilimumab in the phase 3 Checkmate 238 trial published in 2017 (N Engl J Med 2017 Nov 9; 377:1824-35).
But I am getting ahead of myself.
As a thinking man, if I had a thick melanoma (that was less than 2 mm), I would opt for a genetic test of my already excised melanoma tissue. If the results of that genetic test (which has near identical sensitivity and specificity for developing metastatic disease as a sentinel node) put me in the low-risk group, I would pass on the sentinel node biopsy. This would eliminate a lot of unnecessary surgery. If I fell into the high-risk group, I would consider a sentinel node biopsy so I could get into a study, or determine if I needed to find a way to get my insurance to pay, or if I could personally afford nivolumab. Even if I opted not to take the drug, because of the potential risk of high-grade side effects, the high-risk genetic profile tells me I would still need more frequent follow-up.
These are exciting times. I am looking forward to clinical trials that allow a patient with a high-risk genetic profile to go directly into a trial. We are moving into the realm of individualized genomic medicine in which metastatic melanoma truly becomes a curable disease.
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Dr. Coldiron has no financial or other conflicts of interest with Castle Biosciences, the manufacturer of the DecisionDx-Melanoma genetic expression profile test. Email him at [email protected].
More than 17 years ago, I published an article that was largely ignored, predicating that patient benefit from the sentinel node biopsy procedure was unlikely.
I asserted that the lymph nodes are not a reliable filter for melanoma cells, lymphatic drainage is capricious, and many individuals (especially younger ones) have benign neval rests in their lymph nodes that cannot be distinguished from melanoma deposits, since they are both positive for the S100 protein (Int J Dermatol. 2000 Nov;39[11]:807-11). In addition, multiple uncontrolled studies had shown that locating sentinel nodes, followed by a complete lymph node dissection, had no survival benefit. At the time, I argued that sentinel node biopsy should be performed only if the patient was going to be enrolled in a clinical study.
Many surgical oncologists have built their careers around the flawed premise that removing the draining nodes would cure melanoma. It doesn’t. It is past time to admit it and move on.
So, if completion node dissection does not save lives, why do a sentinel node biopsy? I recently asked a dermatologist friend, who is a committed acolyte of the sentinel node biopsy school, why he continues to recommend sentinel node biopsy if there is no benefit from complete node dissection. His quick response was that patients want to know if they are at higher risk of metastatic disease so that they can be followed closely with high-resolution ultrasound at a major cancer center and can be eligible for clinical trials. His reply gave me pause, so I asked why completion node dissection was still being recommended. I was told that some patients with positive sentinel nodes lived far away, and if they would not make regular follow-up visits for high-resolution ultrasound, the surgical oncologists do completion node dissection to ensure “local nodal control.” Yipes! You’re going to rip my groin out because I like quiet county living?
I doubt that patients would be enthusiastic if told beforehand that sentinel node biopsies costs $14,000-$18,000, and has a 9% complication rate, and one-third of those patients who have complications end up with permanent lymphedema. I wondered if the patients were told they could have a genetic test done on their already excised melanoma tissue that would tell them if they were in a high-risk group without having an additional invasive surgical procedure. I wondered if they were told that 10%-30% of people with negative sentinel nodes go on to develop metastatic disease. I also wondered if they had been told they would have to walk around with their melanoma, which could spread at any time, for several additional weeks, while waiting for the results of their sentinel node biopsy, instead of having the melanoma immediately removed by their dermatologist. I also wondered if they had been told that high-resolution ultrasound has not definitively been shown to be superior to clinical palpation of the lymph nodes.
I looked into the possibility of clinical trials for patients with positive sentinel nodes, as well. Based on my search of clinical trials.gov in January, there are 33 trials in the United States studying patients with stage 3 (positive sentinel node) or greater disease. If I had a positive sentinel node, I would look for a study in which I had a chance of getting nivolumab, which recently has been shown to be superior to ipilimumab in the phase 3 Checkmate 238 trial published in 2017 (N Engl J Med 2017 Nov 9; 377:1824-35).
But I am getting ahead of myself.
As a thinking man, if I had a thick melanoma (that was less than 2 mm), I would opt for a genetic test of my already excised melanoma tissue. If the results of that genetic test (which has near identical sensitivity and specificity for developing metastatic disease as a sentinel node) put me in the low-risk group, I would pass on the sentinel node biopsy. This would eliminate a lot of unnecessary surgery. If I fell into the high-risk group, I would consider a sentinel node biopsy so I could get into a study, or determine if I needed to find a way to get my insurance to pay, or if I could personally afford nivolumab. Even if I opted not to take the drug, because of the potential risk of high-grade side effects, the high-risk genetic profile tells me I would still need more frequent follow-up.
These are exciting times. I am looking forward to clinical trials that allow a patient with a high-risk genetic profile to go directly into a trial. We are moving into the realm of individualized genomic medicine in which metastatic melanoma truly becomes a curable disease.
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Dr. Coldiron has no financial or other conflicts of interest with Castle Biosciences, the manufacturer of the DecisionDx-Melanoma genetic expression profile test. Email him at [email protected].





















