User login
How to integrate mental health care into primary care
During my training as a child and adolescent psychiatry fellow, I “lived” down the hall from 10 other people just like me who had similar offices and training. Our pace was tailored to pediatric psychiatry. Appointments were 30 minutes or more. Our goal was to provide the most comprehensive mental health care for the families whom we grew to know and love.
The impetus to create an integrated mental health care approach has been well elucidated by the American Academy of Child and Adolescent Psychiatry (AACAP) in its report, Collaborative mental health care in pediatric primary care. It is based on some telling statistics: Fifty percent of all cases of mental illness begin before age 14 years and 75% begin by age 24. Half of all pediatric office visits involve behavioral, psychosocial, or educational concerns. The American Academy of Pediatrics’ Task Force on Mental Health similarly has stated that primary care clinicians can and should be able to provide mental health services to children and adolescents in a primary care setting.
Integrative psychiatry and primary care treatment comes in three forms: classic consultation, in which a specialist sees a patient and refers back to the PCP with recommendations; colocation, in which mental health specialists practice in the same office but essentially are “ships crossing in the night” with PCPs; and the most-lauded form, collaborative/integrative care, in which back-and-forth consultation and discussions of a case occur between mental health specialists and PCPs, with in-person follow-up as needed.
Several institutions offer programs to address the AACAP and AAP imperatives, most prominently the University of Washington, Seattle, and the University of Massachusetts, Worcester. Both offer resources on how to create an integrated care model (University of Washington AIMS Resource Center; The University of Massachusetts Center for Integrated Care).
What can one do in a busy pediatric primary care practice to address mental health imperatives on the individual provider level? Often PCPs can, as I do, offer families some resources by having a set of mental health handouts and resources. I have gathered useful handouts for families throughout my residency to use as shortcuts and visual aids to promote mental health. I use the AACAP Facts for Families for handouts on mental health diagnoses and topics. I use the National Sleep Foundation for its sleep hygiene tips. I also offer some low-cost mindfulness resources to help kids and parents with their anxiety, such as the Calm app and Headspace app. If parents have difficulty with access to parent management training (the first-line treatment to manage aggression in children), I often recommend “The Defiant Child: A Parent’s Guide to Oppositional Defiant Disorder” (Lanham, Md.: Taylor Trade Publishing, 1997), which shows how to create a rewards system in the home to promote positive behavior. “How to Talk So Kids Will Listen & Listen So Kids Will Talk” (New York: Scribner, 2012 ) is a beloved book for parents (and there is a teenager version) that I recommend when parents launch into questions about how to talk to kids and teens about difficult topics so that, ultimately, they can improve their relationship.
Dr. Pawlowski is an adult, adolescent, and child psychiatrist at the University of Vermont Medical Center and an assistant professor of psychiatry at UVM, both in Burlington. Email her at [email protected].
Resources
The AACAP website has materials to help clinicians develop a collaborative mental health care model in the primary care setting: Search for “collaboration with primary care.”
The journal Pediatrics also has a useful resource: Improving mental health services in primary care: Reducing administrative and financial barriers to access and collaboration. (2009;123;1248-51).
During my training as a child and adolescent psychiatry fellow, I “lived” down the hall from 10 other people just like me who had similar offices and training. Our pace was tailored to pediatric psychiatry. Appointments were 30 minutes or more. Our goal was to provide the most comprehensive mental health care for the families whom we grew to know and love.
The impetus to create an integrated mental health care approach has been well elucidated by the American Academy of Child and Adolescent Psychiatry (AACAP) in its report, Collaborative mental health care in pediatric primary care. It is based on some telling statistics: Fifty percent of all cases of mental illness begin before age 14 years and 75% begin by age 24. Half of all pediatric office visits involve behavioral, psychosocial, or educational concerns. The American Academy of Pediatrics’ Task Force on Mental Health similarly has stated that primary care clinicians can and should be able to provide mental health services to children and adolescents in a primary care setting.
Integrative psychiatry and primary care treatment comes in three forms: classic consultation, in which a specialist sees a patient and refers back to the PCP with recommendations; colocation, in which mental health specialists practice in the same office but essentially are “ships crossing in the night” with PCPs; and the most-lauded form, collaborative/integrative care, in which back-and-forth consultation and discussions of a case occur between mental health specialists and PCPs, with in-person follow-up as needed.
Several institutions offer programs to address the AACAP and AAP imperatives, most prominently the University of Washington, Seattle, and the University of Massachusetts, Worcester. Both offer resources on how to create an integrated care model (University of Washington AIMS Resource Center; The University of Massachusetts Center for Integrated Care).
What can one do in a busy pediatric primary care practice to address mental health imperatives on the individual provider level? Often PCPs can, as I do, offer families some resources by having a set of mental health handouts and resources. I have gathered useful handouts for families throughout my residency to use as shortcuts and visual aids to promote mental health. I use the AACAP Facts for Families for handouts on mental health diagnoses and topics. I use the National Sleep Foundation for its sleep hygiene tips. I also offer some low-cost mindfulness resources to help kids and parents with their anxiety, such as the Calm app and Headspace app. If parents have difficulty with access to parent management training (the first-line treatment to manage aggression in children), I often recommend “The Defiant Child: A Parent’s Guide to Oppositional Defiant Disorder” (Lanham, Md.: Taylor Trade Publishing, 1997), which shows how to create a rewards system in the home to promote positive behavior. “How to Talk So Kids Will Listen & Listen So Kids Will Talk” (New York: Scribner, 2012 ) is a beloved book for parents (and there is a teenager version) that I recommend when parents launch into questions about how to talk to kids and teens about difficult topics so that, ultimately, they can improve their relationship.
Dr. Pawlowski is an adult, adolescent, and child psychiatrist at the University of Vermont Medical Center and an assistant professor of psychiatry at UVM, both in Burlington. Email her at [email protected].
Resources
The AACAP website has materials to help clinicians develop a collaborative mental health care model in the primary care setting: Search for “collaboration with primary care.”
The journal Pediatrics also has a useful resource: Improving mental health services in primary care: Reducing administrative and financial barriers to access and collaboration. (2009;123;1248-51).
During my training as a child and adolescent psychiatry fellow, I “lived” down the hall from 10 other people just like me who had similar offices and training. Our pace was tailored to pediatric psychiatry. Appointments were 30 minutes or more. Our goal was to provide the most comprehensive mental health care for the families whom we grew to know and love.
The impetus to create an integrated mental health care approach has been well elucidated by the American Academy of Child and Adolescent Psychiatry (AACAP) in its report, Collaborative mental health care in pediatric primary care. It is based on some telling statistics: Fifty percent of all cases of mental illness begin before age 14 years and 75% begin by age 24. Half of all pediatric office visits involve behavioral, psychosocial, or educational concerns. The American Academy of Pediatrics’ Task Force on Mental Health similarly has stated that primary care clinicians can and should be able to provide mental health services to children and adolescents in a primary care setting.
Integrative psychiatry and primary care treatment comes in three forms: classic consultation, in which a specialist sees a patient and refers back to the PCP with recommendations; colocation, in which mental health specialists practice in the same office but essentially are “ships crossing in the night” with PCPs; and the most-lauded form, collaborative/integrative care, in which back-and-forth consultation and discussions of a case occur between mental health specialists and PCPs, with in-person follow-up as needed.
Several institutions offer programs to address the AACAP and AAP imperatives, most prominently the University of Washington, Seattle, and the University of Massachusetts, Worcester. Both offer resources on how to create an integrated care model (University of Washington AIMS Resource Center; The University of Massachusetts Center for Integrated Care).
What can one do in a busy pediatric primary care practice to address mental health imperatives on the individual provider level? Often PCPs can, as I do, offer families some resources by having a set of mental health handouts and resources. I have gathered useful handouts for families throughout my residency to use as shortcuts and visual aids to promote mental health. I use the AACAP Facts for Families for handouts on mental health diagnoses and topics. I use the National Sleep Foundation for its sleep hygiene tips. I also offer some low-cost mindfulness resources to help kids and parents with their anxiety, such as the Calm app and Headspace app. If parents have difficulty with access to parent management training (the first-line treatment to manage aggression in children), I often recommend “The Defiant Child: A Parent’s Guide to Oppositional Defiant Disorder” (Lanham, Md.: Taylor Trade Publishing, 1997), which shows how to create a rewards system in the home to promote positive behavior. “How to Talk So Kids Will Listen & Listen So Kids Will Talk” (New York: Scribner, 2012 ) is a beloved book for parents (and there is a teenager version) that I recommend when parents launch into questions about how to talk to kids and teens about difficult topics so that, ultimately, they can improve their relationship.
Dr. Pawlowski is an adult, adolescent, and child psychiatrist at the University of Vermont Medical Center and an assistant professor of psychiatry at UVM, both in Burlington. Email her at [email protected].
Resources
The AACAP website has materials to help clinicians develop a collaborative mental health care model in the primary care setting: Search for “collaboration with primary care.”
The journal Pediatrics also has a useful resource: Improving mental health services in primary care: Reducing administrative and financial barriers to access and collaboration. (2009;123;1248-51).
Serotonin syndrome warnings magnify its rare probability
Serotonin syndrome.
What do those words make you think of? A rare neurological condition? The differential diagnosis you got at 2:00 a.m. from an overly enthusiastic resident? Or a fax from a pharmacy that you get a few times a week?
I’ll say the last.
Triptans are not as common, but are still out there in large numbers. To date, they’re the most effective migraine treatment we have.
Inevitably, these roads will cross, especially because SNRIs, and their older cousins the tricylic antidepressants, are commonly used for migraine prevention. And that’s where the fun begins. There’s a suspected incidence of serotonin syndrome when the two are combined, which became widespread knowledge following a 2006 Food and Drug Administration alert. This is a fact drilled into us by multiple call-backs and faxes from pharmacies, terrified patients who use Google, and electronic prescribing systems that flag our attempts to combine them to make sure we know THIS IS DANGEROUS!
But a study published Feb. 26 in JAMA Neurology found that it’s rarer than anyone suspected (JAMA Neurol. 2018 Feb 26. doi: 10.1001/jamaneurol.2017.5144). Breaking down 14 years’ worth of patient data with more than 19,000 patients on both triptans and serotonergic drugs, there were only two cases (0.01%) that clearly met criteria for serotonin syndrome.
I also understand where the warnings come from. When things go bad, medicine becomes a blame game as each side points at another. The pharmacy wants to say they warned us. The e-prescribing system company wants to say they warned us. The patients want to know why no one warned them when a Google search makes it sound common. And the malpractice lawyers want to blame everyone.
But there are more serious side effects out there. Dilantin has been linked to lymphoma. Sinemet (possibly) to melanoma. But do you remember the last time you had to click or sign off on a pharmacy warning for those? Me neither.
Using any drug is a balance of risks and benefits. We make our judgments, discuss concerns with the patients, and roll the dice every day. Side effects aren’t uncommon. Most serious side effects are rare. But warnings that magnify issues with a rare probability don’t help the situation and can keep patients from receiving the help they need.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Serotonin syndrome.
What do those words make you think of? A rare neurological condition? The differential diagnosis you got at 2:00 a.m. from an overly enthusiastic resident? Or a fax from a pharmacy that you get a few times a week?
I’ll say the last.
Triptans are not as common, but are still out there in large numbers. To date, they’re the most effective migraine treatment we have.
Inevitably, these roads will cross, especially because SNRIs, and their older cousins the tricylic antidepressants, are commonly used for migraine prevention. And that’s where the fun begins. There’s a suspected incidence of serotonin syndrome when the two are combined, which became widespread knowledge following a 2006 Food and Drug Administration alert. This is a fact drilled into us by multiple call-backs and faxes from pharmacies, terrified patients who use Google, and electronic prescribing systems that flag our attempts to combine them to make sure we know THIS IS DANGEROUS!
But a study published Feb. 26 in JAMA Neurology found that it’s rarer than anyone suspected (JAMA Neurol. 2018 Feb 26. doi: 10.1001/jamaneurol.2017.5144). Breaking down 14 years’ worth of patient data with more than 19,000 patients on both triptans and serotonergic drugs, there were only two cases (0.01%) that clearly met criteria for serotonin syndrome.
I also understand where the warnings come from. When things go bad, medicine becomes a blame game as each side points at another. The pharmacy wants to say they warned us. The e-prescribing system company wants to say they warned us. The patients want to know why no one warned them when a Google search makes it sound common. And the malpractice lawyers want to blame everyone.
But there are more serious side effects out there. Dilantin has been linked to lymphoma. Sinemet (possibly) to melanoma. But do you remember the last time you had to click or sign off on a pharmacy warning for those? Me neither.
Using any drug is a balance of risks and benefits. We make our judgments, discuss concerns with the patients, and roll the dice every day. Side effects aren’t uncommon. Most serious side effects are rare. But warnings that magnify issues with a rare probability don’t help the situation and can keep patients from receiving the help they need.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Serotonin syndrome.
What do those words make you think of? A rare neurological condition? The differential diagnosis you got at 2:00 a.m. from an overly enthusiastic resident? Or a fax from a pharmacy that you get a few times a week?
I’ll say the last.
Triptans are not as common, but are still out there in large numbers. To date, they’re the most effective migraine treatment we have.
Inevitably, these roads will cross, especially because SNRIs, and their older cousins the tricylic antidepressants, are commonly used for migraine prevention. And that’s where the fun begins. There’s a suspected incidence of serotonin syndrome when the two are combined, which became widespread knowledge following a 2006 Food and Drug Administration alert. This is a fact drilled into us by multiple call-backs and faxes from pharmacies, terrified patients who use Google, and electronic prescribing systems that flag our attempts to combine them to make sure we know THIS IS DANGEROUS!
But a study published Feb. 26 in JAMA Neurology found that it’s rarer than anyone suspected (JAMA Neurol. 2018 Feb 26. doi: 10.1001/jamaneurol.2017.5144). Breaking down 14 years’ worth of patient data with more than 19,000 patients on both triptans and serotonergic drugs, there were only two cases (0.01%) that clearly met criteria for serotonin syndrome.
I also understand where the warnings come from. When things go bad, medicine becomes a blame game as each side points at another. The pharmacy wants to say they warned us. The e-prescribing system company wants to say they warned us. The patients want to know why no one warned them when a Google search makes it sound common. And the malpractice lawyers want to blame everyone.
But there are more serious side effects out there. Dilantin has been linked to lymphoma. Sinemet (possibly) to melanoma. But do you remember the last time you had to click or sign off on a pharmacy warning for those? Me neither.
Using any drug is a balance of risks and benefits. We make our judgments, discuss concerns with the patients, and roll the dice every day. Side effects aren’t uncommon. Most serious side effects are rare. But warnings that magnify issues with a rare probability don’t help the situation and can keep patients from receiving the help they need.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Pediatric Dermatology Consult - February 2018
The patient was diagnosed with pityriasis rosea (PR) on the basis of the clinical findings; a biopsy was not performed. The patient’s pruritus was treated with oral hydroxyzine and topical 1% triamcinolone ointment. She experienced itch relief with these treatments. On follow-up at 3 months, the patient’s lesions had mostly resolved with some postinflammatory hyperpigmentation.
In some patients, flu-like symptoms precede the onset of skin lesions; this has led to speculation regarding a viral etiology for PR. This prodrome, which is present in as many as half of all cases,can include mild headache, low-grade fever, joint aches, or malaise.2 Pityriasis rosea is thought to occur secondary to a systemic activation of human herpesviruses (HHV) 6 and/or HHV-7. Three cases of PR have been reported in the setting of H1N1 influenza virus infection.3 In one small study, HHV-8 was detected by polymerase chain reaction in approximately 20% of biopsy samples of lesional skin in patients with PR.4 However, most research on a viral etiology for pityriasis rosea has focused on HHV-6 and to a lesser extent HHV-7. DNA from both viruses has been isolated from PR lesions, but at varying detection rates.5,6 Furthermore, HHV-7 DNA has been isolated in as many as 14% of normal individuals without pityriasis rosea, suggesting that the presence of this virus on the skin is fairly common.7
Pityriasis rosea occurs in males and females of all ethnicities, with a slight female predominance. It is rare in young children and older adults. Most cases occur in adolescents and in adults in their twenties and early thirties. Cases occur most frequently in fall and spring.8
The herald patch of pityriasis rosea is typically solitary, but cases with multiple herald patches have been described. The herald patch can range in size from 1-10 cm and usually contains the best example of trailing scale – scale seen on the inside edge of the annular lesion. The satellite lesions of pityriasis rosea are typically papules or plaques with a collarette of scale. These lesions usually are oriented along the Langer cleavage lines, giving them a “Christmas tree” configuration when they appear on the posterior trunk.
Mimics
The herald patch of pityriasis rosea can resemble tinea corporis, and if there is any doubt as to the diagnosis, potassium hydroxide examination (also known as a KOH test) and/or fungal culture should be done to rule out a fungal etiology. However, certain features of this case, particularly the subsequent development of satellite lesions, are more consistent with pityriasis rosea.
Secondary syphilis should be considered in patients who are sexually active. The lesions of secondary syphilis are not typically pruritic, and involvement of the palms and soles is common (whereas such involvement is rare in pityriasis rosea).
Like pityriasis rosea, pityriasis lichenoides et varioliformis acuta (PLEVA) is characterized by papular lesions that resolve spontaneously; the lesions of PLEVA usually evolve to vesicular, necrotic, and purpuric papules that take longer to resolve than PR lesions. The lesions of PLEVA are more erythematous, pustular, and crusting than the lesions of pityriasis rosea.
Guttate psoriasis, which occurs following streptococcal pharyngitis in over 50% of patients, does not present with a herald lesion or distribution along Langer’s lines.10 If guttate psoriasis is suspected, rapid streptococcal testing of the throat or perianal area may be considered.
Nummular eczema presents as papules that enlarge to form erythematous, lichenified plaques that measure 1-2 cm in diameter. A relatively sudden eruption, such as this patient’s, would be unusual for nummular eczema. Also, nummular eczema typically occurs on xerotic skin, more often on the extremities than the trunk.
Diagnostic tests, treatment
Most patients do not require specific therapy for pityriasis rosea. Patients should be reassured that PR is typically a self-limited disease without long-term sequelae. Pregnant patients who develop pityriasis rosea in the first trimester may be at higher risk for spontaneous abortion,although data on the subject are sorely lacking.11 Oral antihistamines are useful in reducing pruritus associated with PR, and some patients experience relief by applying a low-potency topical corticosteroid.
In more severe cases, or in cases in which the patient is greatly distressed by the lesions, both broadband and narrowband UVB phototherapy effectively improve severity of lesions and reduces symptoms.12 These observations suggest that moderate sun exposure can help to reduce severity of PR lesions and hasten their resolution, but no studies assessing the effect of sun exposure on pityriasis rosea symptoms have been performed.
Furthermore, the possible role of the HHV-6 in PR has led some investigators to explore the utility of acyclovir in managing pityriasis rosea.13 One group recently found that 400 mg of acyclovir three times per day for 7 days decreased the number of lesions and pruritus associated with pityriasis rosea, compared those seen in controls, at 1-month follow-up.13
Timely recognition of the diagnosis, consideration of mimics, and ample reassurance are appropriate when approaching this disease.
Mr. Kusari is with the division of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, and the departments of dermatology and pediatrics, University of California, San Diego. Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. They have no relevant financial disclosures. Email them at [email protected].
References
1. Dermatology. 2015;231(1):9-14.
2. World J Clin Cases. 2017 Jun 16;5(6):203-11.
3. Pediatr Dermatol. 2011 May-Jun;28(3):341-2.
4. J Eur Acad Dermatol Venereol. 2006 Jul;20(6):667-71.
5. Dermatology. 1997;195(4):374-8.
6. J Invest Dermatol. 2005 Jun;124(6):1234-40.
7. Arch Dermatol. 1999 Sep;135(9):1070-2.
8. J Am Acad Dermatol. 1982 Jul;7(1):80-9.
9. Iran J Pediatr. 2010 Jun;20(2):237-41.
10. J Pediatr. 1988 Dec;113(6):1037-9.
11. J Am Acad Dermatol. 2008 May;58(5 Suppl 1):S78-83.
12. J Am Acad Dermatol. 1995 Dec;33(6):996-9.
13. Indian Dermatol Online J. 2015 May-Jun;6(3):181-4.
The patient was diagnosed with pityriasis rosea (PR) on the basis of the clinical findings; a biopsy was not performed. The patient’s pruritus was treated with oral hydroxyzine and topical 1% triamcinolone ointment. She experienced itch relief with these treatments. On follow-up at 3 months, the patient’s lesions had mostly resolved with some postinflammatory hyperpigmentation.
In some patients, flu-like symptoms precede the onset of skin lesions; this has led to speculation regarding a viral etiology for PR. This prodrome, which is present in as many as half of all cases,can include mild headache, low-grade fever, joint aches, or malaise.2 Pityriasis rosea is thought to occur secondary to a systemic activation of human herpesviruses (HHV) 6 and/or HHV-7. Three cases of PR have been reported in the setting of H1N1 influenza virus infection.3 In one small study, HHV-8 was detected by polymerase chain reaction in approximately 20% of biopsy samples of lesional skin in patients with PR.4 However, most research on a viral etiology for pityriasis rosea has focused on HHV-6 and to a lesser extent HHV-7. DNA from both viruses has been isolated from PR lesions, but at varying detection rates.5,6 Furthermore, HHV-7 DNA has been isolated in as many as 14% of normal individuals without pityriasis rosea, suggesting that the presence of this virus on the skin is fairly common.7
Pityriasis rosea occurs in males and females of all ethnicities, with a slight female predominance. It is rare in young children and older adults. Most cases occur in adolescents and in adults in their twenties and early thirties. Cases occur most frequently in fall and spring.8
The herald patch of pityriasis rosea is typically solitary, but cases with multiple herald patches have been described. The herald patch can range in size from 1-10 cm and usually contains the best example of trailing scale – scale seen on the inside edge of the annular lesion. The satellite lesions of pityriasis rosea are typically papules or plaques with a collarette of scale. These lesions usually are oriented along the Langer cleavage lines, giving them a “Christmas tree” configuration when they appear on the posterior trunk.
Mimics
The herald patch of pityriasis rosea can resemble tinea corporis, and if there is any doubt as to the diagnosis, potassium hydroxide examination (also known as a KOH test) and/or fungal culture should be done to rule out a fungal etiology. However, certain features of this case, particularly the subsequent development of satellite lesions, are more consistent with pityriasis rosea.
Secondary syphilis should be considered in patients who are sexually active. The lesions of secondary syphilis are not typically pruritic, and involvement of the palms and soles is common (whereas such involvement is rare in pityriasis rosea).
Like pityriasis rosea, pityriasis lichenoides et varioliformis acuta (PLEVA) is characterized by papular lesions that resolve spontaneously; the lesions of PLEVA usually evolve to vesicular, necrotic, and purpuric papules that take longer to resolve than PR lesions. The lesions of PLEVA are more erythematous, pustular, and crusting than the lesions of pityriasis rosea.
Guttate psoriasis, which occurs following streptococcal pharyngitis in over 50% of patients, does not present with a herald lesion or distribution along Langer’s lines.10 If guttate psoriasis is suspected, rapid streptococcal testing of the throat or perianal area may be considered.
Nummular eczema presents as papules that enlarge to form erythematous, lichenified plaques that measure 1-2 cm in diameter. A relatively sudden eruption, such as this patient’s, would be unusual for nummular eczema. Also, nummular eczema typically occurs on xerotic skin, more often on the extremities than the trunk.
Diagnostic tests, treatment
Most patients do not require specific therapy for pityriasis rosea. Patients should be reassured that PR is typically a self-limited disease without long-term sequelae. Pregnant patients who develop pityriasis rosea in the first trimester may be at higher risk for spontaneous abortion,although data on the subject are sorely lacking.11 Oral antihistamines are useful in reducing pruritus associated with PR, and some patients experience relief by applying a low-potency topical corticosteroid.
In more severe cases, or in cases in which the patient is greatly distressed by the lesions, both broadband and narrowband UVB phototherapy effectively improve severity of lesions and reduces symptoms.12 These observations suggest that moderate sun exposure can help to reduce severity of PR lesions and hasten their resolution, but no studies assessing the effect of sun exposure on pityriasis rosea symptoms have been performed.
Furthermore, the possible role of the HHV-6 in PR has led some investigators to explore the utility of acyclovir in managing pityriasis rosea.13 One group recently found that 400 mg of acyclovir three times per day for 7 days decreased the number of lesions and pruritus associated with pityriasis rosea, compared those seen in controls, at 1-month follow-up.13
Timely recognition of the diagnosis, consideration of mimics, and ample reassurance are appropriate when approaching this disease.
Mr. Kusari is with the division of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, and the departments of dermatology and pediatrics, University of California, San Diego. Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. They have no relevant financial disclosures. Email them at [email protected].
References
1. Dermatology. 2015;231(1):9-14.
2. World J Clin Cases. 2017 Jun 16;5(6):203-11.
3. Pediatr Dermatol. 2011 May-Jun;28(3):341-2.
4. J Eur Acad Dermatol Venereol. 2006 Jul;20(6):667-71.
5. Dermatology. 1997;195(4):374-8.
6. J Invest Dermatol. 2005 Jun;124(6):1234-40.
7. Arch Dermatol. 1999 Sep;135(9):1070-2.
8. J Am Acad Dermatol. 1982 Jul;7(1):80-9.
9. Iran J Pediatr. 2010 Jun;20(2):237-41.
10. J Pediatr. 1988 Dec;113(6):1037-9.
11. J Am Acad Dermatol. 2008 May;58(5 Suppl 1):S78-83.
12. J Am Acad Dermatol. 1995 Dec;33(6):996-9.
13. Indian Dermatol Online J. 2015 May-Jun;6(3):181-4.
The patient was diagnosed with pityriasis rosea (PR) on the basis of the clinical findings; a biopsy was not performed. The patient’s pruritus was treated with oral hydroxyzine and topical 1% triamcinolone ointment. She experienced itch relief with these treatments. On follow-up at 3 months, the patient’s lesions had mostly resolved with some postinflammatory hyperpigmentation.
In some patients, flu-like symptoms precede the onset of skin lesions; this has led to speculation regarding a viral etiology for PR. This prodrome, which is present in as many as half of all cases,can include mild headache, low-grade fever, joint aches, or malaise.2 Pityriasis rosea is thought to occur secondary to a systemic activation of human herpesviruses (HHV) 6 and/or HHV-7. Three cases of PR have been reported in the setting of H1N1 influenza virus infection.3 In one small study, HHV-8 was detected by polymerase chain reaction in approximately 20% of biopsy samples of lesional skin in patients with PR.4 However, most research on a viral etiology for pityriasis rosea has focused on HHV-6 and to a lesser extent HHV-7. DNA from both viruses has been isolated from PR lesions, but at varying detection rates.5,6 Furthermore, HHV-7 DNA has been isolated in as many as 14% of normal individuals without pityriasis rosea, suggesting that the presence of this virus on the skin is fairly common.7
Pityriasis rosea occurs in males and females of all ethnicities, with a slight female predominance. It is rare in young children and older adults. Most cases occur in adolescents and in adults in their twenties and early thirties. Cases occur most frequently in fall and spring.8
The herald patch of pityriasis rosea is typically solitary, but cases with multiple herald patches have been described. The herald patch can range in size from 1-10 cm and usually contains the best example of trailing scale – scale seen on the inside edge of the annular lesion. The satellite lesions of pityriasis rosea are typically papules or plaques with a collarette of scale. These lesions usually are oriented along the Langer cleavage lines, giving them a “Christmas tree” configuration when they appear on the posterior trunk.
Mimics
The herald patch of pityriasis rosea can resemble tinea corporis, and if there is any doubt as to the diagnosis, potassium hydroxide examination (also known as a KOH test) and/or fungal culture should be done to rule out a fungal etiology. However, certain features of this case, particularly the subsequent development of satellite lesions, are more consistent with pityriasis rosea.
Secondary syphilis should be considered in patients who are sexually active. The lesions of secondary syphilis are not typically pruritic, and involvement of the palms and soles is common (whereas such involvement is rare in pityriasis rosea).
Like pityriasis rosea, pityriasis lichenoides et varioliformis acuta (PLEVA) is characterized by papular lesions that resolve spontaneously; the lesions of PLEVA usually evolve to vesicular, necrotic, and purpuric papules that take longer to resolve than PR lesions. The lesions of PLEVA are more erythematous, pustular, and crusting than the lesions of pityriasis rosea.
Guttate psoriasis, which occurs following streptococcal pharyngitis in over 50% of patients, does not present with a herald lesion or distribution along Langer’s lines.10 If guttate psoriasis is suspected, rapid streptococcal testing of the throat or perianal area may be considered.
Nummular eczema presents as papules that enlarge to form erythematous, lichenified plaques that measure 1-2 cm in diameter. A relatively sudden eruption, such as this patient’s, would be unusual for nummular eczema. Also, nummular eczema typically occurs on xerotic skin, more often on the extremities than the trunk.
Diagnostic tests, treatment
Most patients do not require specific therapy for pityriasis rosea. Patients should be reassured that PR is typically a self-limited disease without long-term sequelae. Pregnant patients who develop pityriasis rosea in the first trimester may be at higher risk for spontaneous abortion,although data on the subject are sorely lacking.11 Oral antihistamines are useful in reducing pruritus associated with PR, and some patients experience relief by applying a low-potency topical corticosteroid.
In more severe cases, or in cases in which the patient is greatly distressed by the lesions, both broadband and narrowband UVB phototherapy effectively improve severity of lesions and reduces symptoms.12 These observations suggest that moderate sun exposure can help to reduce severity of PR lesions and hasten their resolution, but no studies assessing the effect of sun exposure on pityriasis rosea symptoms have been performed.
Furthermore, the possible role of the HHV-6 in PR has led some investigators to explore the utility of acyclovir in managing pityriasis rosea.13 One group recently found that 400 mg of acyclovir three times per day for 7 days decreased the number of lesions and pruritus associated with pityriasis rosea, compared those seen in controls, at 1-month follow-up.13
Timely recognition of the diagnosis, consideration of mimics, and ample reassurance are appropriate when approaching this disease.
Mr. Kusari is with the division of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, and the departments of dermatology and pediatrics, University of California, San Diego. Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. They have no relevant financial disclosures. Email them at [email protected].
References
1. Dermatology. 2015;231(1):9-14.
2. World J Clin Cases. 2017 Jun 16;5(6):203-11.
3. Pediatr Dermatol. 2011 May-Jun;28(3):341-2.
4. J Eur Acad Dermatol Venereol. 2006 Jul;20(6):667-71.
5. Dermatology. 1997;195(4):374-8.
6. J Invest Dermatol. 2005 Jun;124(6):1234-40.
7. Arch Dermatol. 1999 Sep;135(9):1070-2.
8. J Am Acad Dermatol. 1982 Jul;7(1):80-9.
9. Iran J Pediatr. 2010 Jun;20(2):237-41.
10. J Pediatr. 1988 Dec;113(6):1037-9.
11. J Am Acad Dermatol. 2008 May;58(5 Suppl 1):S78-83.
12. J Am Acad Dermatol. 1995 Dec;33(6):996-9.
13. Indian Dermatol Online J. 2015 May-Jun;6(3):181-4.
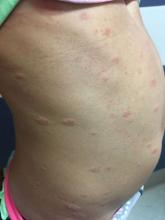
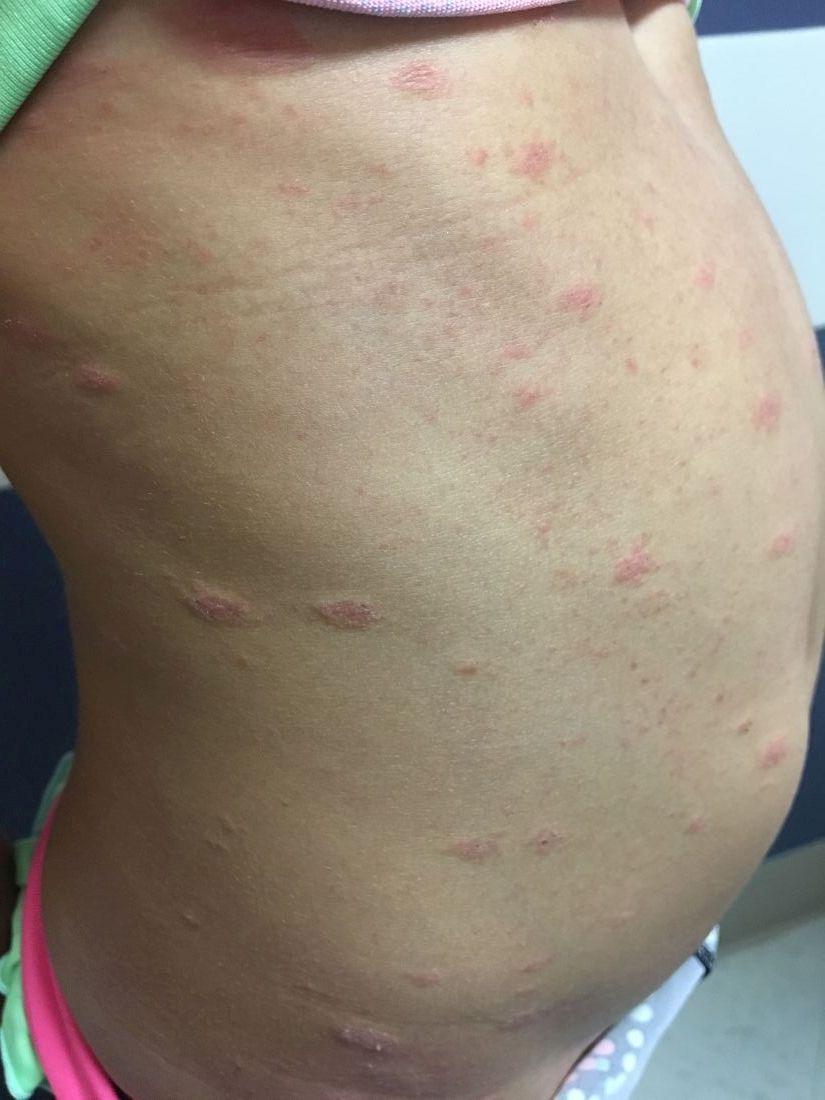
A 6-year-old female presents to the pediatric dermatology office with a 2-day history of a slightly itchy skin lesion on her back. Her birthday was a week prior, and her mother gave her a new kitten, and since then she has been playing with the kitten daily. She has tried some over-the-counter antifungal cream since the lesion first appeared, but there hasn’t been much improvement. The night prior to presenting to the office, the mother noticed more lesions developing on the child’s torso, and because of this, she became worried.
On physical exam, the patient is well appearing, and vital signs are normal. She has multiple scaly, pink, oval plaques and papules on her torso. There are no oral lesions, and her palms and soles are spared.
Is 17-OHPC effective for reducing risk of preterm birth?
In 2003, the Eunice Kennedy Shriver National Institute of Child Health and Human Development’s Maternal-Fetal Medicine Units (MFMU) Network reported on a placebo-controlled randomized study of 17–alpha hydroxyprogesterone caproate (17-OHPC) in women with a history of spontaneous preterm delivery. The study demonstrated a 33% reduction in recurrent preterm birth after weekly treatment with 17-OHPC, which was initiated at 16-20 weeks of gestation.
This landmark study, led by Paul Meis, MD, validated what had been suggested in an earlier meta-analysis (1990) by Mark Keirse, MD – and it quickly altered clinical practice. It set into motion a string of studies on the use of 17-OHPC and other progestational compounds in women with a variety of conditions associated with an increased risk for preterm birth.
It is not surprising, then, that the literature has become muddied and full of contradictory findings since publication of the Meis study and the initial studies on vaginal progesterone in women with a midtrimester short cervix. Further confounding our ability to judge a treatment’s effectiveness is the fact that spontaneous preterm birth is increasingly understood to be a multifactorial, highly heterogeneous condition. We cannot, with a broad stroke, say that all women with a prior preterm birth, for instance, will respond to progestogens in a similar manner or are at the same level of risk of recurrent spontaneous preterm birth (sPTB).
The number of large, randomized clinical trials evaluating progestins is actually quite small but opinions abound about the data from these studies. Below, I have categorized these treatments according to my view at this time of the currently available data.
Consensus
One area in which there is agreement concerns the use of 17-OHPC intramuscular injections in multifetal gestations. Two randomized clinical trials undertaken by the MFMU Network – one in twins and one in triplets – concluded that 17-OHPC is ineffective in reducing the rate of preterm birth. Moreover, in another, more recent MFMU Network study, there was a negative linear relationship between concentrations of 17-OHPC and gestational age at delivery. Women with twin gestations who had higher concentrations of 17-OHPC delivered at earlier gestational ages than women with lower concentrations (Am J Obstet Gynecol. 2012;207[5]:396.e1-8).
Other investigators have similarly shown in clinical trials that the preterm birth rate actually seems to be worsened in multifetal gestations when 17-OHPC is used. There is now widespread agreement that the compound should not be used in these patients.
In addition, an MFMU Network study led by William A. Grobman, MD, demonstrated that 17-OHPC (250-mg injections) does not provide any benefit to nulliparous women with a sonographic cervical length less than 30 mm (Am J Obstet Gynecol. 2012;207[5]:390.e1-8). Other studies utilizing higher doses of 17-OHPC similarly found no benefit. There is also agreement that 17-OHPC has no benefit in treating women with preterm premature rupture of the membranes, preterm labor, or as a maintenance treatment after an episode of preterm labor.
General agreement without consensus
There is general agreement that women with a singleton gestation and a prior spontaneous preterm birth should be offered 17-OHPC, and that women with a singleton gestation and a midtrimester shortened cervical length should be offered vaginal progesterone and not 17-OHPC. However, even in these populations, there are questions about efficacy, dosing, and other issues.
In the Meis study (N Engl J Med. 2003;348:2379-85), treatment with 17-OHPC in women with a singleton gestation and a prior preterm delivery significantly reduced the risk of another preterm birth at less than 37 weeks’ gestation (36.3% in the progesterone group vs. 54.9% in the placebo group; relative risk, 0.66), at less than 35 weeks’ gestation (RR, 0.67), and at less than 32 weeks’ gestation (RR, 0.58). The exceptionally high rate of preterm delivery in the placebo group, however, prompted other investigators to express concern in published correspondence that the study was potentially flawed.
We reported an inverse relationship between 17-OHPC concentration and spontaneous preterm birth as part of a study conducted with the MFMU Network and the Obstetrical-Fetal Pharmacology Research Units Network. All women in the study had singleton gestations and received 250 mg weekly 17-OHPC (the broader study was designed to evaluate the benefit of omega-3 supplementation). We measured plasma concentrations of 17-OHPC and found that women with concentrations in the lowest quartile had a significantly higher risk of preterm birth and delivered at significantly earlier gestational ages than did women in the second through fourth quartiles (Am J Obstet Gynecol. 2014;210[2]:128.e1-6).
Other studies/abstracts similarly evaluating the relationship between 17-OHPC concentrations and preterm birth have reported mixed results, with both validation and refutation of our findings.
Research underway may help settle the controversy. In an ongoing, open-label pharmacology study being conducted by the Obstetrical-Fetal Pharmacology Research Units Network, women with singleton pregnancies and a history of prior preterm birth are being randomly assigned to receive either 250 mg (the empirically chosen, currently recommended dose) or 500 mg 17-OHPC. A relationship between the plasma concentration of 17-OHPC at 26-30 weeks’ gestation and the incidence of preterm birth would offer proof of efficacy and could help elucidate the therapeutic dosing; if there is no relationship, we revert to the question of whether the agent really works. Based on current evidence, both the Society for Maternal-Fetal Medicine (SMFM) and the American College of Obstetricians and Gynecologists (ACOG) support the use of 17-OHPC for prevention of sPTB in women with a prior sPTB.
Questions about vaginal progesterone have also been somewhat unsettled. Eduardo B. Fonseca, MD, reported in 2007 that asymptomatic women with a short cervix (defined as 15 mm or less) who were randomized to receive vaginal progesterone at a median of 22 weeks’ gestation had a significantly lower rate of preterm birth before 34 weeks’ gestation than those who received placebo (RR, 0.56; N Engl J Med. 2007;357[5]:462-9). Research that followed offered mixed conclusions, with a study by Sonia S. Hassan, MD, showing benefit and a study by Jane E. Norman, MD, showing no benefit. Notably, in 2012, the Food and Drug Administration voted against approval of a sustained-release progesterone vaginal gel, citing research results that were not sufficiently compelling.
Still, vaginal progesterone has been endorsed by both ACOG and by the SMFM for women with a short cervical length in the midtrimester. This is supported by a new review and meta-analysis of individual patient data by Roberto Romero, MD, in which vaginal progesterone was found to significantly decrease the risk of preterm birth in singleton gestations with a midtrimester cervical length of 25 mm or less. The reduction occurred over a wide range of gestational ages, including at less than 33 weeks of gestation (RR, 0.62; Am J Obstet Gynecol. 2018 Feb;218[2]:161-80).
Disagreement
Some have argued that vaginal progesterone should be offered to women with a history of prior spontaneous preterm birth, but the largest study to look at this application – a randomized multinational trial reported by John M. O’Brien, MD, and his colleagues in 2007 – found that use of the compound did not reduce the frequency of recurrent preterm birth at or before 32 weeks. Others have argued that vaginal progesterone is of benefit in this group of women based on a combination of multiple subgroup analyses. There is disagreement between ACOG and SMFM on this issue. ACOG supports the use of vaginal progesterone for women with a prior preterm birth but the SMFM strongly rejects this treatment and only endorses 17-OHPC for this indication.
Unresolved
The value of vaginal progesterone supplementation in reducing preterm births in women with twin gestations is under continuing investigation, including a study of women with twin gestation and a short cervix. This MFMU Network randomized trial, now underway, is evaluating the effectiveness of vaginal progesterone or pessary, compared with placebo, in preventing early preterm birth in women carrying twins who have a cervical length less than 30 mm.
Another question about the use of progesterone concerns the woman who delivered preterm during a twin gestation and is now pregnant with a singleton gestation. Should anything be offered to her? This is a question that has not yet been addressed in the literature.
What does seem clear is that spontaneous preterm birth is a multifactorial condition with numerous causes, and quite possibly an interaction between genetics, maternal characteristics, and the environment surrounding each pregnancy (Semin Perinatol. 2016;40[5]:273-80). Certainly, there are different pathways and mechanisms at play in patients who deliver at 35-36 weeks, for instance, compared with those who deliver at 25-26 weeks.
We recently obtained cervical fluid from pregnant women with prior preterm births and analyzed the samples for concentrations of cytokines and matrix metalloproteinases. Women with a prior early preterm delivery at less than 26 weeks had elevations in five cervical cytokines – an inflammatory signature, in essence – while those whose prior preterm birth occurred at a later gestational age had no elevations of these cytokines (Am J Perinatol. 2017 Nov 15. doi: 10.1055/s-0037-1608631).
Hopefully, we soon will be able to identify subpopulations of pregnant women who will benefit more from progesterone supplementation. More research needs to be done at a granular level, with more narrowly defined populations – and with consideration of various pharmacologic, genetic and environmental factors – in order to develop a more specific treatment approach. In the meantime, it is important to appreciate the unknowns that underlie the highly variable clinical responses and outcomes seen in our clinical trials.
Dr. Caritis is professor of obstetrics, gynecology, and reproductive sciences at Magee-Womens Hospital, University of Pittsburgh. He has no disclosures relevant to this Master Class.
In 2003, the Eunice Kennedy Shriver National Institute of Child Health and Human Development’s Maternal-Fetal Medicine Units (MFMU) Network reported on a placebo-controlled randomized study of 17–alpha hydroxyprogesterone caproate (17-OHPC) in women with a history of spontaneous preterm delivery. The study demonstrated a 33% reduction in recurrent preterm birth after weekly treatment with 17-OHPC, which was initiated at 16-20 weeks of gestation.
This landmark study, led by Paul Meis, MD, validated what had been suggested in an earlier meta-analysis (1990) by Mark Keirse, MD – and it quickly altered clinical practice. It set into motion a string of studies on the use of 17-OHPC and other progestational compounds in women with a variety of conditions associated with an increased risk for preterm birth.
It is not surprising, then, that the literature has become muddied and full of contradictory findings since publication of the Meis study and the initial studies on vaginal progesterone in women with a midtrimester short cervix. Further confounding our ability to judge a treatment’s effectiveness is the fact that spontaneous preterm birth is increasingly understood to be a multifactorial, highly heterogeneous condition. We cannot, with a broad stroke, say that all women with a prior preterm birth, for instance, will respond to progestogens in a similar manner or are at the same level of risk of recurrent spontaneous preterm birth (sPTB).
The number of large, randomized clinical trials evaluating progestins is actually quite small but opinions abound about the data from these studies. Below, I have categorized these treatments according to my view at this time of the currently available data.
Consensus
One area in which there is agreement concerns the use of 17-OHPC intramuscular injections in multifetal gestations. Two randomized clinical trials undertaken by the MFMU Network – one in twins and one in triplets – concluded that 17-OHPC is ineffective in reducing the rate of preterm birth. Moreover, in another, more recent MFMU Network study, there was a negative linear relationship between concentrations of 17-OHPC and gestational age at delivery. Women with twin gestations who had higher concentrations of 17-OHPC delivered at earlier gestational ages than women with lower concentrations (Am J Obstet Gynecol. 2012;207[5]:396.e1-8).
Other investigators have similarly shown in clinical trials that the preterm birth rate actually seems to be worsened in multifetal gestations when 17-OHPC is used. There is now widespread agreement that the compound should not be used in these patients.
In addition, an MFMU Network study led by William A. Grobman, MD, demonstrated that 17-OHPC (250-mg injections) does not provide any benefit to nulliparous women with a sonographic cervical length less than 30 mm (Am J Obstet Gynecol. 2012;207[5]:390.e1-8). Other studies utilizing higher doses of 17-OHPC similarly found no benefit. There is also agreement that 17-OHPC has no benefit in treating women with preterm premature rupture of the membranes, preterm labor, or as a maintenance treatment after an episode of preterm labor.
General agreement without consensus
There is general agreement that women with a singleton gestation and a prior spontaneous preterm birth should be offered 17-OHPC, and that women with a singleton gestation and a midtrimester shortened cervical length should be offered vaginal progesterone and not 17-OHPC. However, even in these populations, there are questions about efficacy, dosing, and other issues.
In the Meis study (N Engl J Med. 2003;348:2379-85), treatment with 17-OHPC in women with a singleton gestation and a prior preterm delivery significantly reduced the risk of another preterm birth at less than 37 weeks’ gestation (36.3% in the progesterone group vs. 54.9% in the placebo group; relative risk, 0.66), at less than 35 weeks’ gestation (RR, 0.67), and at less than 32 weeks’ gestation (RR, 0.58). The exceptionally high rate of preterm delivery in the placebo group, however, prompted other investigators to express concern in published correspondence that the study was potentially flawed.
We reported an inverse relationship between 17-OHPC concentration and spontaneous preterm birth as part of a study conducted with the MFMU Network and the Obstetrical-Fetal Pharmacology Research Units Network. All women in the study had singleton gestations and received 250 mg weekly 17-OHPC (the broader study was designed to evaluate the benefit of omega-3 supplementation). We measured plasma concentrations of 17-OHPC and found that women with concentrations in the lowest quartile had a significantly higher risk of preterm birth and delivered at significantly earlier gestational ages than did women in the second through fourth quartiles (Am J Obstet Gynecol. 2014;210[2]:128.e1-6).
Other studies/abstracts similarly evaluating the relationship between 17-OHPC concentrations and preterm birth have reported mixed results, with both validation and refutation of our findings.
Research underway may help settle the controversy. In an ongoing, open-label pharmacology study being conducted by the Obstetrical-Fetal Pharmacology Research Units Network, women with singleton pregnancies and a history of prior preterm birth are being randomly assigned to receive either 250 mg (the empirically chosen, currently recommended dose) or 500 mg 17-OHPC. A relationship between the plasma concentration of 17-OHPC at 26-30 weeks’ gestation and the incidence of preterm birth would offer proof of efficacy and could help elucidate the therapeutic dosing; if there is no relationship, we revert to the question of whether the agent really works. Based on current evidence, both the Society for Maternal-Fetal Medicine (SMFM) and the American College of Obstetricians and Gynecologists (ACOG) support the use of 17-OHPC for prevention of sPTB in women with a prior sPTB.
Questions about vaginal progesterone have also been somewhat unsettled. Eduardo B. Fonseca, MD, reported in 2007 that asymptomatic women with a short cervix (defined as 15 mm or less) who were randomized to receive vaginal progesterone at a median of 22 weeks’ gestation had a significantly lower rate of preterm birth before 34 weeks’ gestation than those who received placebo (RR, 0.56; N Engl J Med. 2007;357[5]:462-9). Research that followed offered mixed conclusions, with a study by Sonia S. Hassan, MD, showing benefit and a study by Jane E. Norman, MD, showing no benefit. Notably, in 2012, the Food and Drug Administration voted against approval of a sustained-release progesterone vaginal gel, citing research results that were not sufficiently compelling.
Still, vaginal progesterone has been endorsed by both ACOG and by the SMFM for women with a short cervical length in the midtrimester. This is supported by a new review and meta-analysis of individual patient data by Roberto Romero, MD, in which vaginal progesterone was found to significantly decrease the risk of preterm birth in singleton gestations with a midtrimester cervical length of 25 mm or less. The reduction occurred over a wide range of gestational ages, including at less than 33 weeks of gestation (RR, 0.62; Am J Obstet Gynecol. 2018 Feb;218[2]:161-80).
Disagreement
Some have argued that vaginal progesterone should be offered to women with a history of prior spontaneous preterm birth, but the largest study to look at this application – a randomized multinational trial reported by John M. O’Brien, MD, and his colleagues in 2007 – found that use of the compound did not reduce the frequency of recurrent preterm birth at or before 32 weeks. Others have argued that vaginal progesterone is of benefit in this group of women based on a combination of multiple subgroup analyses. There is disagreement between ACOG and SMFM on this issue. ACOG supports the use of vaginal progesterone for women with a prior preterm birth but the SMFM strongly rejects this treatment and only endorses 17-OHPC for this indication.
Unresolved
The value of vaginal progesterone supplementation in reducing preterm births in women with twin gestations is under continuing investigation, including a study of women with twin gestation and a short cervix. This MFMU Network randomized trial, now underway, is evaluating the effectiveness of vaginal progesterone or pessary, compared with placebo, in preventing early preterm birth in women carrying twins who have a cervical length less than 30 mm.
Another question about the use of progesterone concerns the woman who delivered preterm during a twin gestation and is now pregnant with a singleton gestation. Should anything be offered to her? This is a question that has not yet been addressed in the literature.
What does seem clear is that spontaneous preterm birth is a multifactorial condition with numerous causes, and quite possibly an interaction between genetics, maternal characteristics, and the environment surrounding each pregnancy (Semin Perinatol. 2016;40[5]:273-80). Certainly, there are different pathways and mechanisms at play in patients who deliver at 35-36 weeks, for instance, compared with those who deliver at 25-26 weeks.
We recently obtained cervical fluid from pregnant women with prior preterm births and analyzed the samples for concentrations of cytokines and matrix metalloproteinases. Women with a prior early preterm delivery at less than 26 weeks had elevations in five cervical cytokines – an inflammatory signature, in essence – while those whose prior preterm birth occurred at a later gestational age had no elevations of these cytokines (Am J Perinatol. 2017 Nov 15. doi: 10.1055/s-0037-1608631).
Hopefully, we soon will be able to identify subpopulations of pregnant women who will benefit more from progesterone supplementation. More research needs to be done at a granular level, with more narrowly defined populations – and with consideration of various pharmacologic, genetic and environmental factors – in order to develop a more specific treatment approach. In the meantime, it is important to appreciate the unknowns that underlie the highly variable clinical responses and outcomes seen in our clinical trials.
Dr. Caritis is professor of obstetrics, gynecology, and reproductive sciences at Magee-Womens Hospital, University of Pittsburgh. He has no disclosures relevant to this Master Class.
In 2003, the Eunice Kennedy Shriver National Institute of Child Health and Human Development’s Maternal-Fetal Medicine Units (MFMU) Network reported on a placebo-controlled randomized study of 17–alpha hydroxyprogesterone caproate (17-OHPC) in women with a history of spontaneous preterm delivery. The study demonstrated a 33% reduction in recurrent preterm birth after weekly treatment with 17-OHPC, which was initiated at 16-20 weeks of gestation.
This landmark study, led by Paul Meis, MD, validated what had been suggested in an earlier meta-analysis (1990) by Mark Keirse, MD – and it quickly altered clinical practice. It set into motion a string of studies on the use of 17-OHPC and other progestational compounds in women with a variety of conditions associated with an increased risk for preterm birth.
It is not surprising, then, that the literature has become muddied and full of contradictory findings since publication of the Meis study and the initial studies on vaginal progesterone in women with a midtrimester short cervix. Further confounding our ability to judge a treatment’s effectiveness is the fact that spontaneous preterm birth is increasingly understood to be a multifactorial, highly heterogeneous condition. We cannot, with a broad stroke, say that all women with a prior preterm birth, for instance, will respond to progestogens in a similar manner or are at the same level of risk of recurrent spontaneous preterm birth (sPTB).
The number of large, randomized clinical trials evaluating progestins is actually quite small but opinions abound about the data from these studies. Below, I have categorized these treatments according to my view at this time of the currently available data.
Consensus
One area in which there is agreement concerns the use of 17-OHPC intramuscular injections in multifetal gestations. Two randomized clinical trials undertaken by the MFMU Network – one in twins and one in triplets – concluded that 17-OHPC is ineffective in reducing the rate of preterm birth. Moreover, in another, more recent MFMU Network study, there was a negative linear relationship between concentrations of 17-OHPC and gestational age at delivery. Women with twin gestations who had higher concentrations of 17-OHPC delivered at earlier gestational ages than women with lower concentrations (Am J Obstet Gynecol. 2012;207[5]:396.e1-8).
Other investigators have similarly shown in clinical trials that the preterm birth rate actually seems to be worsened in multifetal gestations when 17-OHPC is used. There is now widespread agreement that the compound should not be used in these patients.
In addition, an MFMU Network study led by William A. Grobman, MD, demonstrated that 17-OHPC (250-mg injections) does not provide any benefit to nulliparous women with a sonographic cervical length less than 30 mm (Am J Obstet Gynecol. 2012;207[5]:390.e1-8). Other studies utilizing higher doses of 17-OHPC similarly found no benefit. There is also agreement that 17-OHPC has no benefit in treating women with preterm premature rupture of the membranes, preterm labor, or as a maintenance treatment after an episode of preterm labor.
General agreement without consensus
There is general agreement that women with a singleton gestation and a prior spontaneous preterm birth should be offered 17-OHPC, and that women with a singleton gestation and a midtrimester shortened cervical length should be offered vaginal progesterone and not 17-OHPC. However, even in these populations, there are questions about efficacy, dosing, and other issues.
In the Meis study (N Engl J Med. 2003;348:2379-85), treatment with 17-OHPC in women with a singleton gestation and a prior preterm delivery significantly reduced the risk of another preterm birth at less than 37 weeks’ gestation (36.3% in the progesterone group vs. 54.9% in the placebo group; relative risk, 0.66), at less than 35 weeks’ gestation (RR, 0.67), and at less than 32 weeks’ gestation (RR, 0.58). The exceptionally high rate of preterm delivery in the placebo group, however, prompted other investigators to express concern in published correspondence that the study was potentially flawed.
We reported an inverse relationship between 17-OHPC concentration and spontaneous preterm birth as part of a study conducted with the MFMU Network and the Obstetrical-Fetal Pharmacology Research Units Network. All women in the study had singleton gestations and received 250 mg weekly 17-OHPC (the broader study was designed to evaluate the benefit of omega-3 supplementation). We measured plasma concentrations of 17-OHPC and found that women with concentrations in the lowest quartile had a significantly higher risk of preterm birth and delivered at significantly earlier gestational ages than did women in the second through fourth quartiles (Am J Obstet Gynecol. 2014;210[2]:128.e1-6).
Other studies/abstracts similarly evaluating the relationship between 17-OHPC concentrations and preterm birth have reported mixed results, with both validation and refutation of our findings.
Research underway may help settle the controversy. In an ongoing, open-label pharmacology study being conducted by the Obstetrical-Fetal Pharmacology Research Units Network, women with singleton pregnancies and a history of prior preterm birth are being randomly assigned to receive either 250 mg (the empirically chosen, currently recommended dose) or 500 mg 17-OHPC. A relationship between the plasma concentration of 17-OHPC at 26-30 weeks’ gestation and the incidence of preterm birth would offer proof of efficacy and could help elucidate the therapeutic dosing; if there is no relationship, we revert to the question of whether the agent really works. Based on current evidence, both the Society for Maternal-Fetal Medicine (SMFM) and the American College of Obstetricians and Gynecologists (ACOG) support the use of 17-OHPC for prevention of sPTB in women with a prior sPTB.
Questions about vaginal progesterone have also been somewhat unsettled. Eduardo B. Fonseca, MD, reported in 2007 that asymptomatic women with a short cervix (defined as 15 mm or less) who were randomized to receive vaginal progesterone at a median of 22 weeks’ gestation had a significantly lower rate of preterm birth before 34 weeks’ gestation than those who received placebo (RR, 0.56; N Engl J Med. 2007;357[5]:462-9). Research that followed offered mixed conclusions, with a study by Sonia S. Hassan, MD, showing benefit and a study by Jane E. Norman, MD, showing no benefit. Notably, in 2012, the Food and Drug Administration voted against approval of a sustained-release progesterone vaginal gel, citing research results that were not sufficiently compelling.
Still, vaginal progesterone has been endorsed by both ACOG and by the SMFM for women with a short cervical length in the midtrimester. This is supported by a new review and meta-analysis of individual patient data by Roberto Romero, MD, in which vaginal progesterone was found to significantly decrease the risk of preterm birth in singleton gestations with a midtrimester cervical length of 25 mm or less. The reduction occurred over a wide range of gestational ages, including at less than 33 weeks of gestation (RR, 0.62; Am J Obstet Gynecol. 2018 Feb;218[2]:161-80).
Disagreement
Some have argued that vaginal progesterone should be offered to women with a history of prior spontaneous preterm birth, but the largest study to look at this application – a randomized multinational trial reported by John M. O’Brien, MD, and his colleagues in 2007 – found that use of the compound did not reduce the frequency of recurrent preterm birth at or before 32 weeks. Others have argued that vaginal progesterone is of benefit in this group of women based on a combination of multiple subgroup analyses. There is disagreement between ACOG and SMFM on this issue. ACOG supports the use of vaginal progesterone for women with a prior preterm birth but the SMFM strongly rejects this treatment and only endorses 17-OHPC for this indication.
Unresolved
The value of vaginal progesterone supplementation in reducing preterm births in women with twin gestations is under continuing investigation, including a study of women with twin gestation and a short cervix. This MFMU Network randomized trial, now underway, is evaluating the effectiveness of vaginal progesterone or pessary, compared with placebo, in preventing early preterm birth in women carrying twins who have a cervical length less than 30 mm.
Another question about the use of progesterone concerns the woman who delivered preterm during a twin gestation and is now pregnant with a singleton gestation. Should anything be offered to her? This is a question that has not yet been addressed in the literature.
What does seem clear is that spontaneous preterm birth is a multifactorial condition with numerous causes, and quite possibly an interaction between genetics, maternal characteristics, and the environment surrounding each pregnancy (Semin Perinatol. 2016;40[5]:273-80). Certainly, there are different pathways and mechanisms at play in patients who deliver at 35-36 weeks, for instance, compared with those who deliver at 25-26 weeks.
We recently obtained cervical fluid from pregnant women with prior preterm births and analyzed the samples for concentrations of cytokines and matrix metalloproteinases. Women with a prior early preterm delivery at less than 26 weeks had elevations in five cervical cytokines – an inflammatory signature, in essence – while those whose prior preterm birth occurred at a later gestational age had no elevations of these cytokines (Am J Perinatol. 2017 Nov 15. doi: 10.1055/s-0037-1608631).
Hopefully, we soon will be able to identify subpopulations of pregnant women who will benefit more from progesterone supplementation. More research needs to be done at a granular level, with more narrowly defined populations – and with consideration of various pharmacologic, genetic and environmental factors – in order to develop a more specific treatment approach. In the meantime, it is important to appreciate the unknowns that underlie the highly variable clinical responses and outcomes seen in our clinical trials.
Dr. Caritis is professor of obstetrics, gynecology, and reproductive sciences at Magee-Womens Hospital, University of Pittsburgh. He has no disclosures relevant to this Master Class.
For preterm birth, we must avoid being too quick to prescribe therapeutic measures
As ob.gyns., our decisions not only deeply affect the health and well-being of our patients, but can also dramatically impact their children and families. Perhaps nowhere else is the gravity of our medical choices more felt than in the management of premature labor. Premature birth is one of the major drivers of infant mortality, which remains a significant public health problem in the United States where the rate of infant mortality is nearly 6 of every 1,000 live births.
Therefore, when the two seminal studies were published that showed using injectable or vaginal progesterone successfully delayed labor with fewer neonatal complications, the findings were quickly embraced and applied clinically. However, subsequent studies indicated that progesterone is only beneficial to a certain subset of patients – those with singleton pregnancies and a short cervix. The variance in the results of this research highlights an important point: We must treat each patient as an individual, based on her unique medical history, circumstances, and, yes, symptoms. One size does not fit all.
Equally important is a greater need across our practice to avoid being too quick to prescribe therapeutic measures that do not treat the root of the problem. We must instead provide guidance based on rigorously conducted research and analysis. However, even very promising results should not necessarily be used to guide all of clinical practice, and certainly not without scrutiny and considerable analysis.
To dissect the available data and present the most current findings regarding progesterone use to prevent preterm labor, we have invited Steve Caritis, MD, professor of obstetrics, gynecology, and reproductive sciences at Magee-Womens Hospital, University of Pittsburgh, to be the guest author for this month’s Master Class.
Dr. Reece, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. Dr. Reece said he had no relevant financial disclosures. He is the medical editor of this column. Contact him at [email protected].
As ob.gyns., our decisions not only deeply affect the health and well-being of our patients, but can also dramatically impact their children and families. Perhaps nowhere else is the gravity of our medical choices more felt than in the management of premature labor. Premature birth is one of the major drivers of infant mortality, which remains a significant public health problem in the United States where the rate of infant mortality is nearly 6 of every 1,000 live births.
Therefore, when the two seminal studies were published that showed using injectable or vaginal progesterone successfully delayed labor with fewer neonatal complications, the findings were quickly embraced and applied clinically. However, subsequent studies indicated that progesterone is only beneficial to a certain subset of patients – those with singleton pregnancies and a short cervix. The variance in the results of this research highlights an important point: We must treat each patient as an individual, based on her unique medical history, circumstances, and, yes, symptoms. One size does not fit all.
Equally important is a greater need across our practice to avoid being too quick to prescribe therapeutic measures that do not treat the root of the problem. We must instead provide guidance based on rigorously conducted research and analysis. However, even very promising results should not necessarily be used to guide all of clinical practice, and certainly not without scrutiny and considerable analysis.
To dissect the available data and present the most current findings regarding progesterone use to prevent preterm labor, we have invited Steve Caritis, MD, professor of obstetrics, gynecology, and reproductive sciences at Magee-Womens Hospital, University of Pittsburgh, to be the guest author for this month’s Master Class.
Dr. Reece, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. Dr. Reece said he had no relevant financial disclosures. He is the medical editor of this column. Contact him at [email protected].
As ob.gyns., our decisions not only deeply affect the health and well-being of our patients, but can also dramatically impact their children and families. Perhaps nowhere else is the gravity of our medical choices more felt than in the management of premature labor. Premature birth is one of the major drivers of infant mortality, which remains a significant public health problem in the United States where the rate of infant mortality is nearly 6 of every 1,000 live births.
Therefore, when the two seminal studies were published that showed using injectable or vaginal progesterone successfully delayed labor with fewer neonatal complications, the findings were quickly embraced and applied clinically. However, subsequent studies indicated that progesterone is only beneficial to a certain subset of patients – those with singleton pregnancies and a short cervix. The variance in the results of this research highlights an important point: We must treat each patient as an individual, based on her unique medical history, circumstances, and, yes, symptoms. One size does not fit all.
Equally important is a greater need across our practice to avoid being too quick to prescribe therapeutic measures that do not treat the root of the problem. We must instead provide guidance based on rigorously conducted research and analysis. However, even very promising results should not necessarily be used to guide all of clinical practice, and certainly not without scrutiny and considerable analysis.
To dissect the available data and present the most current findings regarding progesterone use to prevent preterm labor, we have invited Steve Caritis, MD, professor of obstetrics, gynecology, and reproductive sciences at Magee-Womens Hospital, University of Pittsburgh, to be the guest author for this month’s Master Class.
Dr. Reece, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. Dr. Reece said he had no relevant financial disclosures. He is the medical editor of this column. Contact him at [email protected].
Learning from the 2017 Oscar fiasco
It was a “never event.” At the very end of the 2017 Academy Awards presentation, the winner for Best Picture was announced. It was wrong. Two and a half minutes later it was corrected. The true winner was “Moonlight,” not “La La Land.” But by then much damage had been done.
I watched it happen live on TV and reviewed it again on YouTube. Several news agencies investigated and reported on what happened. I don’t have any inside information beyond that, but my engineering perspective can illuminate how to reduce mistakes.
The first lesson is how quickly people seek to assign blame after something goes wrong. I saw various online news agencies say Warren Beatty had announced the wrong winner. While he opened the envelope, it was Faye Dunaway who actually made the announcement of “La La Land.” Furthermore, Warren and Faye were merely reading the card. Warren had been given the wrong envelope, as high resolution photographs prove. The envelope was a duplicate for the prize announced just before them for the Best Actress award. The card said Emma Stone and in a smaller font “La La Land,” the film in which she starred. Warren hesitated because of how this was written on the card. Faye thought he was trying to pause as a shtick to increase suspense so she glanced at the card and blurted out “La La Land.”
Experts in quality improvement have learned that the best way to reduce errors is to resist this tendency to assign blame. A better approach is to assume, absent evidence to the contrary, that everyone is acting responsibly and sincerely to help the patient. Hear both sides of the story before jumping to any conclusions. Find systemic factors that contributed to a human error. Then focus on ameliorating systemic weaknesses.
One contributing factor for the error at the Oscars was that there were two copies of the set of award envelopes, with one set available on each side of the stage. This way the presenters can enter from either side of the stage. They are handed an envelope by one of the two auditors from PricewaterhouseCoopers, who are the only ones who know the contents.
A key component of safety is having check backs. The envelopes have the name of the award on the outside. One might hope the presenter would double check that they are being given the correct envelope by the auditor. But backstage is a very nervous and hectic place for the presenters. Actors are not professionals dedicated to safety.
Medical care is different. Before giving a transfusion, one nurse reads the number on the bag of blood to another nurse, who confirms that it matches a paper form. That simple act can prevent mistakes. Perhaps the auditor handing the envelope to the Oscar presenter should ask the presenter, who knows which award s/he is scheduled to announce, to read out loud the award title on the front of the envelope.
Clearly, Warren Beatty was confused by the contents of the envelope. He was expecting a card to have the name of a film, not the name of an actress with the film’s name in small print below it. He didn’t know what action to take and hesitated. Faye Dunaway plunged forward and misinterpreted the card. A key component of quality is making it safe for anyone, if they are not confident in what is happening, to stop the proceeding, ask questions, and challenge plans. For example, there are time-outs prior to surgery. A second component is presenting information in a form less likely to be misinterpreted. Medicine has a problem with many sound-alike and look-alike drug names, so sometimes these words are spelled with particular letters capitalized, to distinguish them. I wish EHRs would present lab results in large, bold font.
Another contributing factor here was that Faye misinterpreted Warren’s behaviors as a joke. Major airlines utilize the “sterile cockpit.” During the few minutes that they are running through the preflight checklist, the pilot and copilot do not discuss last night’s football game, crack jokes, or engage in any other extraneous conversations. They avoid interruptions and distractions, focusing solely on the task. Sign outs in medicine need to adopt this habit.
There is a concern that one of the auditors tweeted a picture of Emma Stone backstage holding her Oscar at the same time the fiasco was happening on stage. In the modern world, cell phones and selfies are a key source of distraction, errors, and car accidents.
Per the Army, “Prior planning prevents poor performance.” A couple days before the Oscar fiasco, the auditors were interviewed and they revealed that they didn’t have an action plan to deal with the situation of a mistaken announcement. They figured it was extremely unlikely and that the circumstances would determine the best response.
Experience has shown that in the hours leading up to a pediatric code, there may be several opportunities to recognize the risk and intervene so that blame cannot be assigned to a single person or action. Mock codes prepare people to think on their feet. And it is important to have a clearly designated person in charge of a code. Leadership matters.
In the Oscar fiasco, the damage was quickly limited by the gracious words of a “La La Land” producer He assessed the situation, announced the mistake, beckoned the “Moonlight” cast and crew to the stage, graciously complimented them, showed the correct award envelope and card to the camera, and offered the statue to the correct producer. Then he hastened his team off the stage. These actions of responsibility, truthfulness, transparency, and grace staunched the bleeding, minimized the damage, and as best as possible, remediated the error. Movie producers are experts at dealing with crises and catastrophes. Medical staff, when revealing errors to patients, can learn from this role model.
Dr. Powell is a pediatric hospitalist and clinical ethics consultant living in St. Louis. Email him at [email protected].
It was a “never event.” At the very end of the 2017 Academy Awards presentation, the winner for Best Picture was announced. It was wrong. Two and a half minutes later it was corrected. The true winner was “Moonlight,” not “La La Land.” But by then much damage had been done.
I watched it happen live on TV and reviewed it again on YouTube. Several news agencies investigated and reported on what happened. I don’t have any inside information beyond that, but my engineering perspective can illuminate how to reduce mistakes.
The first lesson is how quickly people seek to assign blame after something goes wrong. I saw various online news agencies say Warren Beatty had announced the wrong winner. While he opened the envelope, it was Faye Dunaway who actually made the announcement of “La La Land.” Furthermore, Warren and Faye were merely reading the card. Warren had been given the wrong envelope, as high resolution photographs prove. The envelope was a duplicate for the prize announced just before them for the Best Actress award. The card said Emma Stone and in a smaller font “La La Land,” the film in which she starred. Warren hesitated because of how this was written on the card. Faye thought he was trying to pause as a shtick to increase suspense so she glanced at the card and blurted out “La La Land.”
Experts in quality improvement have learned that the best way to reduce errors is to resist this tendency to assign blame. A better approach is to assume, absent evidence to the contrary, that everyone is acting responsibly and sincerely to help the patient. Hear both sides of the story before jumping to any conclusions. Find systemic factors that contributed to a human error. Then focus on ameliorating systemic weaknesses.
One contributing factor for the error at the Oscars was that there were two copies of the set of award envelopes, with one set available on each side of the stage. This way the presenters can enter from either side of the stage. They are handed an envelope by one of the two auditors from PricewaterhouseCoopers, who are the only ones who know the contents.
A key component of safety is having check backs. The envelopes have the name of the award on the outside. One might hope the presenter would double check that they are being given the correct envelope by the auditor. But backstage is a very nervous and hectic place for the presenters. Actors are not professionals dedicated to safety.
Medical care is different. Before giving a transfusion, one nurse reads the number on the bag of blood to another nurse, who confirms that it matches a paper form. That simple act can prevent mistakes. Perhaps the auditor handing the envelope to the Oscar presenter should ask the presenter, who knows which award s/he is scheduled to announce, to read out loud the award title on the front of the envelope.
Clearly, Warren Beatty was confused by the contents of the envelope. He was expecting a card to have the name of a film, not the name of an actress with the film’s name in small print below it. He didn’t know what action to take and hesitated. Faye Dunaway plunged forward and misinterpreted the card. A key component of quality is making it safe for anyone, if they are not confident in what is happening, to stop the proceeding, ask questions, and challenge plans. For example, there are time-outs prior to surgery. A second component is presenting information in a form less likely to be misinterpreted. Medicine has a problem with many sound-alike and look-alike drug names, so sometimes these words are spelled with particular letters capitalized, to distinguish them. I wish EHRs would present lab results in large, bold font.
Another contributing factor here was that Faye misinterpreted Warren’s behaviors as a joke. Major airlines utilize the “sterile cockpit.” During the few minutes that they are running through the preflight checklist, the pilot and copilot do not discuss last night’s football game, crack jokes, or engage in any other extraneous conversations. They avoid interruptions and distractions, focusing solely on the task. Sign outs in medicine need to adopt this habit.
There is a concern that one of the auditors tweeted a picture of Emma Stone backstage holding her Oscar at the same time the fiasco was happening on stage. In the modern world, cell phones and selfies are a key source of distraction, errors, and car accidents.
Per the Army, “Prior planning prevents poor performance.” A couple days before the Oscar fiasco, the auditors were interviewed and they revealed that they didn’t have an action plan to deal with the situation of a mistaken announcement. They figured it was extremely unlikely and that the circumstances would determine the best response.
Experience has shown that in the hours leading up to a pediatric code, there may be several opportunities to recognize the risk and intervene so that blame cannot be assigned to a single person or action. Mock codes prepare people to think on their feet. And it is important to have a clearly designated person in charge of a code. Leadership matters.
In the Oscar fiasco, the damage was quickly limited by the gracious words of a “La La Land” producer He assessed the situation, announced the mistake, beckoned the “Moonlight” cast and crew to the stage, graciously complimented them, showed the correct award envelope and card to the camera, and offered the statue to the correct producer. Then he hastened his team off the stage. These actions of responsibility, truthfulness, transparency, and grace staunched the bleeding, minimized the damage, and as best as possible, remediated the error. Movie producers are experts at dealing with crises and catastrophes. Medical staff, when revealing errors to patients, can learn from this role model.
Dr. Powell is a pediatric hospitalist and clinical ethics consultant living in St. Louis. Email him at [email protected].
It was a “never event.” At the very end of the 2017 Academy Awards presentation, the winner for Best Picture was announced. It was wrong. Two and a half minutes later it was corrected. The true winner was “Moonlight,” not “La La Land.” But by then much damage had been done.
I watched it happen live on TV and reviewed it again on YouTube. Several news agencies investigated and reported on what happened. I don’t have any inside information beyond that, but my engineering perspective can illuminate how to reduce mistakes.
The first lesson is how quickly people seek to assign blame after something goes wrong. I saw various online news agencies say Warren Beatty had announced the wrong winner. While he opened the envelope, it was Faye Dunaway who actually made the announcement of “La La Land.” Furthermore, Warren and Faye were merely reading the card. Warren had been given the wrong envelope, as high resolution photographs prove. The envelope was a duplicate for the prize announced just before them for the Best Actress award. The card said Emma Stone and in a smaller font “La La Land,” the film in which she starred. Warren hesitated because of how this was written on the card. Faye thought he was trying to pause as a shtick to increase suspense so she glanced at the card and blurted out “La La Land.”
Experts in quality improvement have learned that the best way to reduce errors is to resist this tendency to assign blame. A better approach is to assume, absent evidence to the contrary, that everyone is acting responsibly and sincerely to help the patient. Hear both sides of the story before jumping to any conclusions. Find systemic factors that contributed to a human error. Then focus on ameliorating systemic weaknesses.
One contributing factor for the error at the Oscars was that there were two copies of the set of award envelopes, with one set available on each side of the stage. This way the presenters can enter from either side of the stage. They are handed an envelope by one of the two auditors from PricewaterhouseCoopers, who are the only ones who know the contents.
A key component of safety is having check backs. The envelopes have the name of the award on the outside. One might hope the presenter would double check that they are being given the correct envelope by the auditor. But backstage is a very nervous and hectic place for the presenters. Actors are not professionals dedicated to safety.
Medical care is different. Before giving a transfusion, one nurse reads the number on the bag of blood to another nurse, who confirms that it matches a paper form. That simple act can prevent mistakes. Perhaps the auditor handing the envelope to the Oscar presenter should ask the presenter, who knows which award s/he is scheduled to announce, to read out loud the award title on the front of the envelope.
Clearly, Warren Beatty was confused by the contents of the envelope. He was expecting a card to have the name of a film, not the name of an actress with the film’s name in small print below it. He didn’t know what action to take and hesitated. Faye Dunaway plunged forward and misinterpreted the card. A key component of quality is making it safe for anyone, if they are not confident in what is happening, to stop the proceeding, ask questions, and challenge plans. For example, there are time-outs prior to surgery. A second component is presenting information in a form less likely to be misinterpreted. Medicine has a problem with many sound-alike and look-alike drug names, so sometimes these words are spelled with particular letters capitalized, to distinguish them. I wish EHRs would present lab results in large, bold font.
Another contributing factor here was that Faye misinterpreted Warren’s behaviors as a joke. Major airlines utilize the “sterile cockpit.” During the few minutes that they are running through the preflight checklist, the pilot and copilot do not discuss last night’s football game, crack jokes, or engage in any other extraneous conversations. They avoid interruptions and distractions, focusing solely on the task. Sign outs in medicine need to adopt this habit.
There is a concern that one of the auditors tweeted a picture of Emma Stone backstage holding her Oscar at the same time the fiasco was happening on stage. In the modern world, cell phones and selfies are a key source of distraction, errors, and car accidents.
Per the Army, “Prior planning prevents poor performance.” A couple days before the Oscar fiasco, the auditors were interviewed and they revealed that they didn’t have an action plan to deal with the situation of a mistaken announcement. They figured it was extremely unlikely and that the circumstances would determine the best response.
Experience has shown that in the hours leading up to a pediatric code, there may be several opportunities to recognize the risk and intervene so that blame cannot be assigned to a single person or action. Mock codes prepare people to think on their feet. And it is important to have a clearly designated person in charge of a code. Leadership matters.
In the Oscar fiasco, the damage was quickly limited by the gracious words of a “La La Land” producer He assessed the situation, announced the mistake, beckoned the “Moonlight” cast and crew to the stage, graciously complimented them, showed the correct award envelope and card to the camera, and offered the statue to the correct producer. Then he hastened his team off the stage. These actions of responsibility, truthfulness, transparency, and grace staunched the bleeding, minimized the damage, and as best as possible, remediated the error. Movie producers are experts at dealing with crises and catastrophes. Medical staff, when revealing errors to patients, can learn from this role model.
Dr. Powell is a pediatric hospitalist and clinical ethics consultant living in St. Louis. Email him at [email protected].
Telling her she has cancer: A patient-centered approach to breaking bad news
Hearing a diagnosis of cancer is one of the most significant moments of a patient’s life and informing a patient of her diagnosis is an emotionally and technically challenging task for an obstetrician gynecologist who is frequently on the front line of making this diagnosis. In this column, we will explore some patient-centered strategies to perform this difficult task well so that patients come away informed but with the highest chance for positive emotional adjustment.
Fewer than 10% of physicians report receiving formal training in techniques of breaking bad news. For the majority of clinicians concerns are centered on being honest and not taking away hope, and in responding to a patient’s emotions.1 The SPIKES approach was developed to arm physicians with strategies to discuss a cancer diagnosis with their patients. This approach includes six key elements to incorporate during the encounter. These strategies are not meant to be formulaic but rather consistent principles that can be adjusted for individual patient needs.
Setting up the discussion
Breaking bad news should not be a one-size-fits-all approach. Age, educational level, culture, religion, race and ethnicity, and socioeconomic opportunities each affects what and how patients may want to have this kind of information communicated to them. So how do you know how best to deliver a patient-centered approach for your patient? I recommend this simple strategy: Ask her. When ordering a test or performing a biopsy, let the patient know then why you are ordering the test and inform her of the possibility that the results may show cancer. Ask her how she would like for you to communicate that result. Would she like to be called by phone, the benefit of which is quick dissemination of information? Or would she like to receive the information face to face in the office? Research supports that most patients prefer to learn the result in the office.2 If so, I recommend scheduling a follow-up appointment in advance to prevent delays. Ask her if she would like a family member or a supportive friend to be present for the conveying of results so that she will have time to make these arrangements. Ask her if she would prefer for an alternate person to be provided with the results on her behalf.
When preparing to speak with the patient, it is valuable to mentally rehearse the words that you’ll use. Arrange for privacy and manage time constraints and interruptions (silence pagers and phones, ensure there is adequate time allocated in the schedule). Sit down to deliver the news and make a connection with eye contact and, if appropriate, touch.
Assessing the patient’s perception. Before you tell, ask. For example, “what is your understanding about why we did the biopsy?” This will guide you in where her head and heart are and can ensure you meet her wherever she is.
Obtaining the patient’s invitation. Ask the patient what she would like to be told and how much information. What would she like you to focus on? What does she not want to hear?
Giving knowledge and information to the patient. Especially now, it is important to avoid jargon and use nontechnical terms. However, do not shy away from using specific words like “cancer” by substituting them for more vague and confusing terms such as “malignancy” or “tumor.” It is important to find the balance between expressing information without being overly emotive, while avoiding excessive bluntness. Word choice is critical. Communication styles in the breaking of bad news can be separated broadly into three styles: disease centered, emotion centered, and patient-centered.3 The patient-centered approach is achieved by balancing emotional connection, information sharing, nondominance, and conveying hope. (For example, “I have some disappointing news to share. Shall we talk about the next steps in treatment? I understand this is that this is difficult for you.”) In general, this approach is most valued by patients and is associated with better information recall.
Addressing the patient’s emotions with empathetic responses. It is important that physicians take a moment to pause after communicating the test result. Even if prepared, most patients will still have a moment of shock, and their minds will likely spin through a multitude of thoughts preventing them from being able to “hear” and focus on the subsequent information. This is a moment to reflect on her reactions, her body language, and nonverbal communications to guide you on how to approach the rest of the encounter. Offer her your comfort and condolence in whichever way feels appropriate for you and her.
Beware of your own inclinations to “soften the blow.” It is a natural, compassionate instinct to follow-up giving a bad piece of information by balancing a good piece of information. For example, after just telling a woman that she has endometrial cancer, following with a statement such as “but it’s just stage 1 and is curable with surgery.” While this certainly may have immediate comforting effects, it has a couple of unintended consequences. First, it can result in difficulties later adjusting to a change in diagnosis when more information comes in (for example, upstaging after surgery or imaging). It is better to be honest and tell patients only what you know for sure in these immediate first moments of diagnosis when complete information is lacking. A more general statement such as “for most women, this is found at an early stage and is highly treatable” may be more appropriate and still provide some comfort. Second, attempts to soften the blow with a qualifying statement of positivity, such as “this is a good kind of cancer to have” might be interpreted by some patients as failing to acknowledge their devastation. She may feel that you are minimizing her condition and not allowing her to grieve or be distressed.
Strategy and summary. Patients who leave the encounter with some kind of plan for the future feel less distressed and anxious. The direction at this point of the encounter should be led by the patient. What are her greatest concerns (such as mortality, loss of fertility, time off work for treatment), and what does she want to know right now? Most patients express a desire to know more about treatment or prognosis.2,4 Unfortunately, it often is not possible to furnish this yet, particularly if this falls into the realm of a subspecialist, and prognostication typically requires more information than a provider has at initial diagnosis. However, leaving these questions unanswered is likely to result in a patient feeling helpless. For example, if an ob.gyn. discovers an apparent advanced ovarian cancer on a CT scan, tell her that, despite its apparent advanced case, it is usually treatable and that a gynecologic oncologist will discuss those best treatment options with her. Assure her that you will expeditiously refer her to a specialist who will provide her with those specifics.
The aftermath
That interval between initial diagnosis and specialist consultation is extraordinarily difficult and a high anxiety time. It is not unreasonable, in such cases, to recommend the patient to reputable online information sources, such as the Society of Gynecologic Oncology or American Cancer Society websites so that she and her family can do some research prior to that visit in order to prepare them better and give them a sense of understanding in their disease.
It is a particularly compassionate touch to reach out to the patient in the days following her cancer diagnosis, even if she has moved on to a specialist. Patients often tell me that they felt enormous reassurance and appreciation when their ob.gyn. reached out to them to “check on how they are doing.” This can usually reasonably be done by phone. This second contact serves another critical purpose: it allows for repetition of the diagnosis and initial plan, and the ability to fill in the blanks of what the patient may have missed during the prior visit, if her mind was, naturally, elsewhere. It also, quite simply, shows that you care.
Ultimately, none of us can break bad news perfectly every time. We all need to be insightful with each of these encounters as to what we did well, what we did not, and how we can adjust in the future. With respect to the SPIKES approach, patients report that physicians struggle most with the “perception,” “invitation,” and “strategy and summary” components.5 Our objective should be keeping the patient’s needs in mind, rather than our own, to maximize the chance of doing a good job. If this task is done well, not only are patients more likely to have positive emotional adjustments to their diagnosis but also more adherence with future therapies.4 In the end, it is the patient who has the final say on whether it was done well or not.
Dr. Rossi is an assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She reports no relevant financial disclosures.
References
1. Baile WF et al. Oncologist. 2000;5(4):302-11.
2. Girgis A et al. Behav Med. 1999 Summer;25(2):69-77.
3. Schmid MM et al. Patient Educ Couns. 2005 Sep;58(3):244-51.
4. Girgis A et al. J Clin Oncol. 1995 Sep;13(9):2449-56.
5. Marscholiek P et al. J Cancer Educ. 2018 Feb 5. doi: 10.1007/s13187-017-1315-3.
Hearing a diagnosis of cancer is one of the most significant moments of a patient’s life and informing a patient of her diagnosis is an emotionally and technically challenging task for an obstetrician gynecologist who is frequently on the front line of making this diagnosis. In this column, we will explore some patient-centered strategies to perform this difficult task well so that patients come away informed but with the highest chance for positive emotional adjustment.
Fewer than 10% of physicians report receiving formal training in techniques of breaking bad news. For the majority of clinicians concerns are centered on being honest and not taking away hope, and in responding to a patient’s emotions.1 The SPIKES approach was developed to arm physicians with strategies to discuss a cancer diagnosis with their patients. This approach includes six key elements to incorporate during the encounter. These strategies are not meant to be formulaic but rather consistent principles that can be adjusted for individual patient needs.
Setting up the discussion
Breaking bad news should not be a one-size-fits-all approach. Age, educational level, culture, religion, race and ethnicity, and socioeconomic opportunities each affects what and how patients may want to have this kind of information communicated to them. So how do you know how best to deliver a patient-centered approach for your patient? I recommend this simple strategy: Ask her. When ordering a test or performing a biopsy, let the patient know then why you are ordering the test and inform her of the possibility that the results may show cancer. Ask her how she would like for you to communicate that result. Would she like to be called by phone, the benefit of which is quick dissemination of information? Or would she like to receive the information face to face in the office? Research supports that most patients prefer to learn the result in the office.2 If so, I recommend scheduling a follow-up appointment in advance to prevent delays. Ask her if she would like a family member or a supportive friend to be present for the conveying of results so that she will have time to make these arrangements. Ask her if she would prefer for an alternate person to be provided with the results on her behalf.
When preparing to speak with the patient, it is valuable to mentally rehearse the words that you’ll use. Arrange for privacy and manage time constraints and interruptions (silence pagers and phones, ensure there is adequate time allocated in the schedule). Sit down to deliver the news and make a connection with eye contact and, if appropriate, touch.
Assessing the patient’s perception. Before you tell, ask. For example, “what is your understanding about why we did the biopsy?” This will guide you in where her head and heart are and can ensure you meet her wherever she is.
Obtaining the patient’s invitation. Ask the patient what she would like to be told and how much information. What would she like you to focus on? What does she not want to hear?
Giving knowledge and information to the patient. Especially now, it is important to avoid jargon and use nontechnical terms. However, do not shy away from using specific words like “cancer” by substituting them for more vague and confusing terms such as “malignancy” or “tumor.” It is important to find the balance between expressing information without being overly emotive, while avoiding excessive bluntness. Word choice is critical. Communication styles in the breaking of bad news can be separated broadly into three styles: disease centered, emotion centered, and patient-centered.3 The patient-centered approach is achieved by balancing emotional connection, information sharing, nondominance, and conveying hope. (For example, “I have some disappointing news to share. Shall we talk about the next steps in treatment? I understand this is that this is difficult for you.”) In general, this approach is most valued by patients and is associated with better information recall.
Addressing the patient’s emotions with empathetic responses. It is important that physicians take a moment to pause after communicating the test result. Even if prepared, most patients will still have a moment of shock, and their minds will likely spin through a multitude of thoughts preventing them from being able to “hear” and focus on the subsequent information. This is a moment to reflect on her reactions, her body language, and nonverbal communications to guide you on how to approach the rest of the encounter. Offer her your comfort and condolence in whichever way feels appropriate for you and her.
Beware of your own inclinations to “soften the blow.” It is a natural, compassionate instinct to follow-up giving a bad piece of information by balancing a good piece of information. For example, after just telling a woman that she has endometrial cancer, following with a statement such as “but it’s just stage 1 and is curable with surgery.” While this certainly may have immediate comforting effects, it has a couple of unintended consequences. First, it can result in difficulties later adjusting to a change in diagnosis when more information comes in (for example, upstaging after surgery or imaging). It is better to be honest and tell patients only what you know for sure in these immediate first moments of diagnosis when complete information is lacking. A more general statement such as “for most women, this is found at an early stage and is highly treatable” may be more appropriate and still provide some comfort. Second, attempts to soften the blow with a qualifying statement of positivity, such as “this is a good kind of cancer to have” might be interpreted by some patients as failing to acknowledge their devastation. She may feel that you are minimizing her condition and not allowing her to grieve or be distressed.
Strategy and summary. Patients who leave the encounter with some kind of plan for the future feel less distressed and anxious. The direction at this point of the encounter should be led by the patient. What are her greatest concerns (such as mortality, loss of fertility, time off work for treatment), and what does she want to know right now? Most patients express a desire to know more about treatment or prognosis.2,4 Unfortunately, it often is not possible to furnish this yet, particularly if this falls into the realm of a subspecialist, and prognostication typically requires more information than a provider has at initial diagnosis. However, leaving these questions unanswered is likely to result in a patient feeling helpless. For example, if an ob.gyn. discovers an apparent advanced ovarian cancer on a CT scan, tell her that, despite its apparent advanced case, it is usually treatable and that a gynecologic oncologist will discuss those best treatment options with her. Assure her that you will expeditiously refer her to a specialist who will provide her with those specifics.
The aftermath
That interval between initial diagnosis and specialist consultation is extraordinarily difficult and a high anxiety time. It is not unreasonable, in such cases, to recommend the patient to reputable online information sources, such as the Society of Gynecologic Oncology or American Cancer Society websites so that she and her family can do some research prior to that visit in order to prepare them better and give them a sense of understanding in their disease.
It is a particularly compassionate touch to reach out to the patient in the days following her cancer diagnosis, even if she has moved on to a specialist. Patients often tell me that they felt enormous reassurance and appreciation when their ob.gyn. reached out to them to “check on how they are doing.” This can usually reasonably be done by phone. This second contact serves another critical purpose: it allows for repetition of the diagnosis and initial plan, and the ability to fill in the blanks of what the patient may have missed during the prior visit, if her mind was, naturally, elsewhere. It also, quite simply, shows that you care.
Ultimately, none of us can break bad news perfectly every time. We all need to be insightful with each of these encounters as to what we did well, what we did not, and how we can adjust in the future. With respect to the SPIKES approach, patients report that physicians struggle most with the “perception,” “invitation,” and “strategy and summary” components.5 Our objective should be keeping the patient’s needs in mind, rather than our own, to maximize the chance of doing a good job. If this task is done well, not only are patients more likely to have positive emotional adjustments to their diagnosis but also more adherence with future therapies.4 In the end, it is the patient who has the final say on whether it was done well or not.
Dr. Rossi is an assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She reports no relevant financial disclosures.
References
1. Baile WF et al. Oncologist. 2000;5(4):302-11.
2. Girgis A et al. Behav Med. 1999 Summer;25(2):69-77.
3. Schmid MM et al. Patient Educ Couns. 2005 Sep;58(3):244-51.
4. Girgis A et al. J Clin Oncol. 1995 Sep;13(9):2449-56.
5. Marscholiek P et al. J Cancer Educ. 2018 Feb 5. doi: 10.1007/s13187-017-1315-3.
Hearing a diagnosis of cancer is one of the most significant moments of a patient’s life and informing a patient of her diagnosis is an emotionally and technically challenging task for an obstetrician gynecologist who is frequently on the front line of making this diagnosis. In this column, we will explore some patient-centered strategies to perform this difficult task well so that patients come away informed but with the highest chance for positive emotional adjustment.
Fewer than 10% of physicians report receiving formal training in techniques of breaking bad news. For the majority of clinicians concerns are centered on being honest and not taking away hope, and in responding to a patient’s emotions.1 The SPIKES approach was developed to arm physicians with strategies to discuss a cancer diagnosis with their patients. This approach includes six key elements to incorporate during the encounter. These strategies are not meant to be formulaic but rather consistent principles that can be adjusted for individual patient needs.
Setting up the discussion
Breaking bad news should not be a one-size-fits-all approach. Age, educational level, culture, religion, race and ethnicity, and socioeconomic opportunities each affects what and how patients may want to have this kind of information communicated to them. So how do you know how best to deliver a patient-centered approach for your patient? I recommend this simple strategy: Ask her. When ordering a test or performing a biopsy, let the patient know then why you are ordering the test and inform her of the possibility that the results may show cancer. Ask her how she would like for you to communicate that result. Would she like to be called by phone, the benefit of which is quick dissemination of information? Or would she like to receive the information face to face in the office? Research supports that most patients prefer to learn the result in the office.2 If so, I recommend scheduling a follow-up appointment in advance to prevent delays. Ask her if she would like a family member or a supportive friend to be present for the conveying of results so that she will have time to make these arrangements. Ask her if she would prefer for an alternate person to be provided with the results on her behalf.
When preparing to speak with the patient, it is valuable to mentally rehearse the words that you’ll use. Arrange for privacy and manage time constraints and interruptions (silence pagers and phones, ensure there is adequate time allocated in the schedule). Sit down to deliver the news and make a connection with eye contact and, if appropriate, touch.
Assessing the patient’s perception. Before you tell, ask. For example, “what is your understanding about why we did the biopsy?” This will guide you in where her head and heart are and can ensure you meet her wherever she is.
Obtaining the patient’s invitation. Ask the patient what she would like to be told and how much information. What would she like you to focus on? What does she not want to hear?
Giving knowledge and information to the patient. Especially now, it is important to avoid jargon and use nontechnical terms. However, do not shy away from using specific words like “cancer” by substituting them for more vague and confusing terms such as “malignancy” or “tumor.” It is important to find the balance between expressing information without being overly emotive, while avoiding excessive bluntness. Word choice is critical. Communication styles in the breaking of bad news can be separated broadly into three styles: disease centered, emotion centered, and patient-centered.3 The patient-centered approach is achieved by balancing emotional connection, information sharing, nondominance, and conveying hope. (For example, “I have some disappointing news to share. Shall we talk about the next steps in treatment? I understand this is that this is difficult for you.”) In general, this approach is most valued by patients and is associated with better information recall.
Addressing the patient’s emotions with empathetic responses. It is important that physicians take a moment to pause after communicating the test result. Even if prepared, most patients will still have a moment of shock, and their minds will likely spin through a multitude of thoughts preventing them from being able to “hear” and focus on the subsequent information. This is a moment to reflect on her reactions, her body language, and nonverbal communications to guide you on how to approach the rest of the encounter. Offer her your comfort and condolence in whichever way feels appropriate for you and her.
Beware of your own inclinations to “soften the blow.” It is a natural, compassionate instinct to follow-up giving a bad piece of information by balancing a good piece of information. For example, after just telling a woman that she has endometrial cancer, following with a statement such as “but it’s just stage 1 and is curable with surgery.” While this certainly may have immediate comforting effects, it has a couple of unintended consequences. First, it can result in difficulties later adjusting to a change in diagnosis when more information comes in (for example, upstaging after surgery or imaging). It is better to be honest and tell patients only what you know for sure in these immediate first moments of diagnosis when complete information is lacking. A more general statement such as “for most women, this is found at an early stage and is highly treatable” may be more appropriate and still provide some comfort. Second, attempts to soften the blow with a qualifying statement of positivity, such as “this is a good kind of cancer to have” might be interpreted by some patients as failing to acknowledge their devastation. She may feel that you are minimizing her condition and not allowing her to grieve or be distressed.
Strategy and summary. Patients who leave the encounter with some kind of plan for the future feel less distressed and anxious. The direction at this point of the encounter should be led by the patient. What are her greatest concerns (such as mortality, loss of fertility, time off work for treatment), and what does she want to know right now? Most patients express a desire to know more about treatment or prognosis.2,4 Unfortunately, it often is not possible to furnish this yet, particularly if this falls into the realm of a subspecialist, and prognostication typically requires more information than a provider has at initial diagnosis. However, leaving these questions unanswered is likely to result in a patient feeling helpless. For example, if an ob.gyn. discovers an apparent advanced ovarian cancer on a CT scan, tell her that, despite its apparent advanced case, it is usually treatable and that a gynecologic oncologist will discuss those best treatment options with her. Assure her that you will expeditiously refer her to a specialist who will provide her with those specifics.
The aftermath
That interval between initial diagnosis and specialist consultation is extraordinarily difficult and a high anxiety time. It is not unreasonable, in such cases, to recommend the patient to reputable online information sources, such as the Society of Gynecologic Oncology or American Cancer Society websites so that she and her family can do some research prior to that visit in order to prepare them better and give them a sense of understanding in their disease.
It is a particularly compassionate touch to reach out to the patient in the days following her cancer diagnosis, even if she has moved on to a specialist. Patients often tell me that they felt enormous reassurance and appreciation when their ob.gyn. reached out to them to “check on how they are doing.” This can usually reasonably be done by phone. This second contact serves another critical purpose: it allows for repetition of the diagnosis and initial plan, and the ability to fill in the blanks of what the patient may have missed during the prior visit, if her mind was, naturally, elsewhere. It also, quite simply, shows that you care.
Ultimately, none of us can break bad news perfectly every time. We all need to be insightful with each of these encounters as to what we did well, what we did not, and how we can adjust in the future. With respect to the SPIKES approach, patients report that physicians struggle most with the “perception,” “invitation,” and “strategy and summary” components.5 Our objective should be keeping the patient’s needs in mind, rather than our own, to maximize the chance of doing a good job. If this task is done well, not only are patients more likely to have positive emotional adjustments to their diagnosis but also more adherence with future therapies.4 In the end, it is the patient who has the final say on whether it was done well or not.
Dr. Rossi is an assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She reports no relevant financial disclosures.
References
1. Baile WF et al. Oncologist. 2000;5(4):302-11.
2. Girgis A et al. Behav Med. 1999 Summer;25(2):69-77.
3. Schmid MM et al. Patient Educ Couns. 2005 Sep;58(3):244-51.
4. Girgis A et al. J Clin Oncol. 1995 Sep;13(9):2449-56.
5. Marscholiek P et al. J Cancer Educ. 2018 Feb 5. doi: 10.1007/s13187-017-1315-3.
Making social media work for your practice
Social media use is ubiquitous and, in the digital age, it is the ascendant form of communication. Individuals and organizations, digital immigrants (those born before the widespread adoption of digital technology), and digital natives alike are leveraging social media platforms, such as blogs, Facebook, Twitter, YouTube, and LinkedIn, to curate, consume, and share information across the spectrum of demographics and target audiences. In the United States, 7 in 10 Americans are using social media and, although young adults were early adopters, use among older adults is increasing rapidly.1
Furthermore, social media has cultivated remarkable opportunities in the dissemination of health information and disrupted traditional methods of patient–provider communication. The days when medically trained health professionals were the gatekeepers of health information are long gone. Approximately 50% of Americans seek health information online before seeing a physician.2 Patients and other consumers regularly access social media to search for information about diseases and treatments, engage with other patients, identify providers, and to express or rate their satisfaction with providers, clinics, and health systems.3-5 In addition, they trust online health information from doctors more than that from hospitals, health insurers, and drug companies.6 Not surprisingly, this has led to tremendous growth in use of social media by health care providers, hospitals, and health centers. More than 90% of US hospitals have a Facebook page and 50% have a Twitter account.7
There is ample opportunity to close the gap between patient and health care provider engagement in Social media, equip providers with the tools they need to be competent consumers and sharers of information in this digital exchange, and increase the pool of evidence-based information on GI and liver diseases on social media.12 However, there is limited published literature tailored to gastroenterologists and hepatologists. The goal of this article, therefore, is to provide a broad overview of best practices in the professional use of social media and highlight examples of novel applications in clinical practice.
Best practices: Getting started and maintaining a presence on social media
Social media can magnify your professional image, amplify your voice, and extend your reach and influence much faster than other methods. It also can be damaging if not used responsibly. Thus, we recommend the following approaches to responsible use of social media and cultivating your social media presence based on current evidence, professional organizations’ policy statements, and our combined experience. We initially presented these strategies during a Meet-the-Professor Luncheon at Digestive Disease Week® in Chicago (http://www.ddw.org/education/session-recordings).
Second, as with other aspects of medical training and practice, find a mentor to provide hands-on advice. This is particularly true if your general familiarity with the social media platforms is limited. If this is not available through your network of colleagues or workplace, we recommend exploring opportunities offered through your professional organization(s) such as the aforementioned Meet-the-Professor Luncheon at Digestive Diseases Week.
Third, know the privacy setting options on your social media platform(s) of choice and use them to your advantage. For example, on Facebook and Twitter, you can select an option that requests your permission before a friend or follower is added to your network. You also can tailor who (such as friends or followers only) can access your posted content directly. However, know that your content still may be made public if it is shared by one of your friends or followers.
Fourth, nurture your social media presence by sharing credible content deliberately, regularly, and, when appropriate, with attribution.
Fifth, diversify your content within the realm of your predefined objectives and/or goals and avoid a singular focus of self-promotion or the appearance of self-promotion. Top social media users suggest, and the authors agree, that your content should be only 25%-33% of your posts.
Sixth, thoroughly vet all content that you share. Avoid automatically sharing articles or posts because of a catchy headline. Read them before you post them. There may be details buried in them that are not credible or with which you do not agree.
Seventh, build community by connecting and engaging with other users on your social media platform(s) of choice.
Eighth, integrate multiple media (i.e., photos, videos, infographics) and/or social media platforms (i.e., embed link to YouTube or a website) to increase engagement.
Ninth, adhere to the code of ethics, governance, and privacy of the profession and of your employer.
Best practices: Privacy and governance in patient-oriented communication on social media
Two factors that have been of pivotal concern with the adoption of social media in the health care arena and led to many health care professionals being laggards as opposed to early adopters are privacy and governance. Will it violate the patient–provider relationship? What about the Health Insurance Portability and Accountability Act? How do I maintain boundaries between myself and the public at large? These are just a few of the questions that commonly are asked by those who are unfamiliar with social media etiquette for health care professionals. We highly recommend reviewing the position paper regarding online medical professionalism issued by the American College of Physicians and the Federation of State Medical Boards as a starting point.13 We believe the following to be contemporary guiding principles for GI health providers for maintaining a digital footprint on social media that reflects the ethical and professional standards of the field.
First, avoid sharing information that could be construed as a patient identifier without documented consent. This includes, but is not limited to, an identifiable specimen or photograph, and stories of care, rare conditions, and complications. Note that dates and location of care can lead to identification of a patient or care episode.
Second, recognize that personal and professional online profiles/pages are discoverable. Many advocate for separating the two as a means of shielding the public from elements of a private persona (i.e., family pictures and controversial opinions). However, the capacity to share and find comments and images on social media is much more powerful than the privacy settings on the various social media platforms. If you establish distinct personal and professional profiles, exercise caution before accepting friend or follow requests from patients on your personal profile. In addition, be cautious with your posts on private social media accounts because they rarely truly are private.
Third, avoid providing specific medical recommendations to individuals. This creates a patient–provider relationship and legal duty. Instead, recommend consultation with a health care provider and consider providing a link to general information on the topic (e.g., AGA information for patients at www.gastro.org/patientinfo).
Fourth, declare conflicts of interest, if applicable, when sharing information involving your clinical, research, and/or business practice.
Fifth, routinely monitor your online presence for accuracy and appropriateness of content posted by you and by others in reference to you. Know that our profession’s ethical standards for behavior extend to social media and we can be held accountable to colleagues and our employer if we violate them.
Many employers have become savvy to issues of governance in use of social media and institute policy recommendations to which employees are expected to adhere. If you are an employee, we recommend checking with your marketing and/or human resources department(s) in regards to this. If you are an employer and do not have such a policy on online professionalism, it is our hope that this article serves as a launching pad.
Novel applications for social media in clinical practice
Social media has been shown to be an effective medium for medical education through virtual journal clubs, moderated discussions or chats, and video sharing for teaching procedures, to name a few applications. Social media is used to collect data via polls or surveys, and to disseminate and track the views and downloads of published works. It is also a source for unsolicited, real-time feedback on patient experience and engagement through data-mining techniques, such as natural language processing and, more simply, for solicited feedback for patient satisfaction ratings. However, its role in academic promotion is less clear and is an area for which we see a great opportunity for growth.
Summary
We have outlined a high-level overview for why you should consider establishing and maintaining a professional presence on social media and how to accomplish this. These reasons include sharing information with colleagues, patients, and the public; amplifying the voice of physicians, a view that has diminished in the often-volatile health care environment; and promotion of the value of your work, be it patient care, advocacy, research, or education. You will have a smoother experience if you learn your local rules and policies and abide by our suggestions to avoid adverse outcomes. You will be most effective if you establish goals for your social media participation and revisit these goals over time for continued relevance and success and if you have consistent and valuable output that will support attainment of these goals. Welcome to the GI social media community! Be sure to follow Clinical Gastroenterology and Hepatology and the American Gastroenterological Association on Facebook (facebook.com/cghjournal and facebook.com/amergastroassn) and Twitter (@AGA_CGH and @AmerGastroAssn), and the coauthors (@DMGrayMD and @DrDeborahFisher) on Twitter.
References
1. Social Media Fact Sheet. Pew Research Center [updated January 12, 2017]. Available from http://www.pewinternet.org/fact-sheet/social-media/. Accessed: June 20, 2017.
2. Hesse B.W., Nelson D.E., Kreps G.L., et al. Trust and sources of health information: the impact of the Internet and its implications for health care providers: findings from the first Health Information National Trends Survey. Arch Intern Med. 2005;165:2618-24.
3. Moorhead S.A., Hazlett D.E., Harrison L., et al. A new dimension of health care: systematic review of the uses, benefits, and limitations of social media for health communication. J Med Internet Res. 2013;15:e85.
4. Chou W.Y., Hunt Y.M., Beckjord E.B. et al. Social media use in the United States: implications for health communication. J Med Internet Res. 2009;11:e48.
5. Chretien K.C., Kind T. Social media and clinical care: ethical, professional, and social implications. Circulation. 2013;27:1413-21.
6. Social Media ‘likes’ Healthcare. PwC Health Research Institute; 2012. Available from https://www.pwc.com/us/en/health-industries/health-research-institute/publications/pdf/health-care-social-media-report.pdf. Accessed: June 20, 2017.
7. Griffis H.M., Kilaru A.S., Werner R.M., et al. Use of social media across US hospitals: descriptive analysis of adoption and utilization. J Med Internet Res. 2014;16:e264.
8. Davis E.D., Tang S.J., Glover P.H., et al. Impact of social media on gastroenterologists in the United States. Dig Liver Dis. 2015;47:258-9.
9. Chiang A.L., Vartabedian B., Spiegel B. Harnessing the hashtag: a standard approach to GI dialogue on social media. Am J Gastroenterol. 2016;111:1082-4.
10. Reich J., Guo L., Hall J., et al. A survey of social media use and preferences in patients with inflammatory bowel disease. Inflamm Bowel Dis. 2016;22:2678-87.
11. Timms C., Forton D.M., Poullis A. Social media use in patients with inflammatory bowel disease and chronic viral hepatitis. Clin Med. 2014;14:215.
12. Prasad B. Social media, health care, and social networking. Gastrointest Endosc. 2013;77:492-5.
13. Farnan J.M., Snyder Sulmasy L., Worster B.K., et al. Online medical professionalism: patient and public relationships: policy statement from the American College of Physicians and the Federation of State Medical Boards. Ann Intern Med. 2013;158:620-7.
14. Cabrera D., Vartabedian B.S., Spinner R.J., et al. More than likes and tweets: creating social media portfolios for academic promotion and tenure. J Grad Med Educ. 2017;9:421-5.
15. Cabrera D. Mayo Clinic includes social media scholarship activities in academic advancement. Available from https://socialmedia.mayoclinic.org/2016/05/25/mayo-clinic-includes-social-media-scholarship-activities-in-academic-advancement/
Date: May 26, 2016. (Accessed: July 1, 2017).
16. Freitag C.E., Arnold M.A., Gardner J.M., et al. If you are not on social media, here’s what you’re missing! #DoTheThing. Arch Pathol Lab Med. 2017; (Epub ahead of print).
17. Stukus D.R. How I used twitter to get promoted in academic medicine. Available from http://www.kevinmd.com/blog/2016/10/used-twitter-get-promoted-academic-medicine.html. Date: October 9, 2016. (Accessed: July 1, 2017).
Dr. Gray is in the division of gastroenterology, hepatology, and nutrition, department of medicine, The Ohio State University College of Medicine, Columbus; Dr. Fisher is in the division of gastroenterology, department of medicine, Duke University, Durham, N.C. The authors disclose no conflicts of interest.
Social media use is ubiquitous and, in the digital age, it is the ascendant form of communication. Individuals and organizations, digital immigrants (those born before the widespread adoption of digital technology), and digital natives alike are leveraging social media platforms, such as blogs, Facebook, Twitter, YouTube, and LinkedIn, to curate, consume, and share information across the spectrum of demographics and target audiences. In the United States, 7 in 10 Americans are using social media and, although young adults were early adopters, use among older adults is increasing rapidly.1
Furthermore, social media has cultivated remarkable opportunities in the dissemination of health information and disrupted traditional methods of patient–provider communication. The days when medically trained health professionals were the gatekeepers of health information are long gone. Approximately 50% of Americans seek health information online before seeing a physician.2 Patients and other consumers regularly access social media to search for information about diseases and treatments, engage with other patients, identify providers, and to express or rate their satisfaction with providers, clinics, and health systems.3-5 In addition, they trust online health information from doctors more than that from hospitals, health insurers, and drug companies.6 Not surprisingly, this has led to tremendous growth in use of social media by health care providers, hospitals, and health centers. More than 90% of US hospitals have a Facebook page and 50% have a Twitter account.7
There is ample opportunity to close the gap between patient and health care provider engagement in Social media, equip providers with the tools they need to be competent consumers and sharers of information in this digital exchange, and increase the pool of evidence-based information on GI and liver diseases on social media.12 However, there is limited published literature tailored to gastroenterologists and hepatologists. The goal of this article, therefore, is to provide a broad overview of best practices in the professional use of social media and highlight examples of novel applications in clinical practice.
Best practices: Getting started and maintaining a presence on social media
Social media can magnify your professional image, amplify your voice, and extend your reach and influence much faster than other methods. It also can be damaging if not used responsibly. Thus, we recommend the following approaches to responsible use of social media and cultivating your social media presence based on current evidence, professional organizations’ policy statements, and our combined experience. We initially presented these strategies during a Meet-the-Professor Luncheon at Digestive Disease Week® in Chicago (http://www.ddw.org/education/session-recordings).
Second, as with other aspects of medical training and practice, find a mentor to provide hands-on advice. This is particularly true if your general familiarity with the social media platforms is limited. If this is not available through your network of colleagues or workplace, we recommend exploring opportunities offered through your professional organization(s) such as the aforementioned Meet-the-Professor Luncheon at Digestive Diseases Week.
Third, know the privacy setting options on your social media platform(s) of choice and use them to your advantage. For example, on Facebook and Twitter, you can select an option that requests your permission before a friend or follower is added to your network. You also can tailor who (such as friends or followers only) can access your posted content directly. However, know that your content still may be made public if it is shared by one of your friends or followers.
Fourth, nurture your social media presence by sharing credible content deliberately, regularly, and, when appropriate, with attribution.
Fifth, diversify your content within the realm of your predefined objectives and/or goals and avoid a singular focus of self-promotion or the appearance of self-promotion. Top social media users suggest, and the authors agree, that your content should be only 25%-33% of your posts.
Sixth, thoroughly vet all content that you share. Avoid automatically sharing articles or posts because of a catchy headline. Read them before you post them. There may be details buried in them that are not credible or with which you do not agree.
Seventh, build community by connecting and engaging with other users on your social media platform(s) of choice.
Eighth, integrate multiple media (i.e., photos, videos, infographics) and/or social media platforms (i.e., embed link to YouTube or a website) to increase engagement.
Ninth, adhere to the code of ethics, governance, and privacy of the profession and of your employer.
Best practices: Privacy and governance in patient-oriented communication on social media
Two factors that have been of pivotal concern with the adoption of social media in the health care arena and led to many health care professionals being laggards as opposed to early adopters are privacy and governance. Will it violate the patient–provider relationship? What about the Health Insurance Portability and Accountability Act? How do I maintain boundaries between myself and the public at large? These are just a few of the questions that commonly are asked by those who are unfamiliar with social media etiquette for health care professionals. We highly recommend reviewing the position paper regarding online medical professionalism issued by the American College of Physicians and the Federation of State Medical Boards as a starting point.13 We believe the following to be contemporary guiding principles for GI health providers for maintaining a digital footprint on social media that reflects the ethical and professional standards of the field.
First, avoid sharing information that could be construed as a patient identifier without documented consent. This includes, but is not limited to, an identifiable specimen or photograph, and stories of care, rare conditions, and complications. Note that dates and location of care can lead to identification of a patient or care episode.
Second, recognize that personal and professional online profiles/pages are discoverable. Many advocate for separating the two as a means of shielding the public from elements of a private persona (i.e., family pictures and controversial opinions). However, the capacity to share and find comments and images on social media is much more powerful than the privacy settings on the various social media platforms. If you establish distinct personal and professional profiles, exercise caution before accepting friend or follow requests from patients on your personal profile. In addition, be cautious with your posts on private social media accounts because they rarely truly are private.
Third, avoid providing specific medical recommendations to individuals. This creates a patient–provider relationship and legal duty. Instead, recommend consultation with a health care provider and consider providing a link to general information on the topic (e.g., AGA information for patients at www.gastro.org/patientinfo).
Fourth, declare conflicts of interest, if applicable, when sharing information involving your clinical, research, and/or business practice.
Fifth, routinely monitor your online presence for accuracy and appropriateness of content posted by you and by others in reference to you. Know that our profession’s ethical standards for behavior extend to social media and we can be held accountable to colleagues and our employer if we violate them.
Many employers have become savvy to issues of governance in use of social media and institute policy recommendations to which employees are expected to adhere. If you are an employee, we recommend checking with your marketing and/or human resources department(s) in regards to this. If you are an employer and do not have such a policy on online professionalism, it is our hope that this article serves as a launching pad.
Novel applications for social media in clinical practice
Social media has been shown to be an effective medium for medical education through virtual journal clubs, moderated discussions or chats, and video sharing for teaching procedures, to name a few applications. Social media is used to collect data via polls or surveys, and to disseminate and track the views and downloads of published works. It is also a source for unsolicited, real-time feedback on patient experience and engagement through data-mining techniques, such as natural language processing and, more simply, for solicited feedback for patient satisfaction ratings. However, its role in academic promotion is less clear and is an area for which we see a great opportunity for growth.

Summary
We have outlined a high-level overview for why you should consider establishing and maintaining a professional presence on social media and how to accomplish this. These reasons include sharing information with colleagues, patients, and the public; amplifying the voice of physicians, a view that has diminished in the often-volatile health care environment; and promotion of the value of your work, be it patient care, advocacy, research, or education. You will have a smoother experience if you learn your local rules and policies and abide by our suggestions to avoid adverse outcomes. You will be most effective if you establish goals for your social media participation and revisit these goals over time for continued relevance and success and if you have consistent and valuable output that will support attainment of these goals. Welcome to the GI social media community! Be sure to follow Clinical Gastroenterology and Hepatology and the American Gastroenterological Association on Facebook (facebook.com/cghjournal and facebook.com/amergastroassn) and Twitter (@AGA_CGH and @AmerGastroAssn), and the coauthors (@DMGrayMD and @DrDeborahFisher) on Twitter.
References
1. Social Media Fact Sheet. Pew Research Center [updated January 12, 2017]. Available from http://www.pewinternet.org/fact-sheet/social-media/. Accessed: June 20, 2017.
2. Hesse B.W., Nelson D.E., Kreps G.L., et al. Trust and sources of health information: the impact of the Internet and its implications for health care providers: findings from the first Health Information National Trends Survey. Arch Intern Med. 2005;165:2618-24.
3. Moorhead S.A., Hazlett D.E., Harrison L., et al. A new dimension of health care: systematic review of the uses, benefits, and limitations of social media for health communication. J Med Internet Res. 2013;15:e85.
4. Chou W.Y., Hunt Y.M., Beckjord E.B. et al. Social media use in the United States: implications for health communication. J Med Internet Res. 2009;11:e48.
5. Chretien K.C., Kind T. Social media and clinical care: ethical, professional, and social implications. Circulation. 2013;27:1413-21.
6. Social Media ‘likes’ Healthcare. PwC Health Research Institute; 2012. Available from https://www.pwc.com/us/en/health-industries/health-research-institute/publications/pdf/health-care-social-media-report.pdf. Accessed: June 20, 2017.
7. Griffis H.M., Kilaru A.S., Werner R.M., et al. Use of social media across US hospitals: descriptive analysis of adoption and utilization. J Med Internet Res. 2014;16:e264.
8. Davis E.D., Tang S.J., Glover P.H., et al. Impact of social media on gastroenterologists in the United States. Dig Liver Dis. 2015;47:258-9.
9. Chiang A.L., Vartabedian B., Spiegel B. Harnessing the hashtag: a standard approach to GI dialogue on social media. Am J Gastroenterol. 2016;111:1082-4.
10. Reich J., Guo L., Hall J., et al. A survey of social media use and preferences in patients with inflammatory bowel disease. Inflamm Bowel Dis. 2016;22:2678-87.
11. Timms C., Forton D.M., Poullis A. Social media use in patients with inflammatory bowel disease and chronic viral hepatitis. Clin Med. 2014;14:215.
12. Prasad B. Social media, health care, and social networking. Gastrointest Endosc. 2013;77:492-5.
13. Farnan J.M., Snyder Sulmasy L., Worster B.K., et al. Online medical professionalism: patient and public relationships: policy statement from the American College of Physicians and the Federation of State Medical Boards. Ann Intern Med. 2013;158:620-7.
14. Cabrera D., Vartabedian B.S., Spinner R.J., et al. More than likes and tweets: creating social media portfolios for academic promotion and tenure. J Grad Med Educ. 2017;9:421-5.
15. Cabrera D. Mayo Clinic includes social media scholarship activities in academic advancement. Available from https://socialmedia.mayoclinic.org/2016/05/25/mayo-clinic-includes-social-media-scholarship-activities-in-academic-advancement/
Date: May 26, 2016. (Accessed: July 1, 2017).
16. Freitag C.E., Arnold M.A., Gardner J.M., et al. If you are not on social media, here’s what you’re missing! #DoTheThing. Arch Pathol Lab Med. 2017; (Epub ahead of print).
17. Stukus D.R. How I used twitter to get promoted in academic medicine. Available from http://www.kevinmd.com/blog/2016/10/used-twitter-get-promoted-academic-medicine.html. Date: October 9, 2016. (Accessed: July 1, 2017).
Dr. Gray is in the division of gastroenterology, hepatology, and nutrition, department of medicine, The Ohio State University College of Medicine, Columbus; Dr. Fisher is in the division of gastroenterology, department of medicine, Duke University, Durham, N.C. The authors disclose no conflicts of interest.
Social media use is ubiquitous and, in the digital age, it is the ascendant form of communication. Individuals and organizations, digital immigrants (those born before the widespread adoption of digital technology), and digital natives alike are leveraging social media platforms, such as blogs, Facebook, Twitter, YouTube, and LinkedIn, to curate, consume, and share information across the spectrum of demographics and target audiences. In the United States, 7 in 10 Americans are using social media and, although young adults were early adopters, use among older adults is increasing rapidly.1
Furthermore, social media has cultivated remarkable opportunities in the dissemination of health information and disrupted traditional methods of patient–provider communication. The days when medically trained health professionals were the gatekeepers of health information are long gone. Approximately 50% of Americans seek health information online before seeing a physician.2 Patients and other consumers regularly access social media to search for information about diseases and treatments, engage with other patients, identify providers, and to express or rate their satisfaction with providers, clinics, and health systems.3-5 In addition, they trust online health information from doctors more than that from hospitals, health insurers, and drug companies.6 Not surprisingly, this has led to tremendous growth in use of social media by health care providers, hospitals, and health centers. More than 90% of US hospitals have a Facebook page and 50% have a Twitter account.7
There is ample opportunity to close the gap between patient and health care provider engagement in Social media, equip providers with the tools they need to be competent consumers and sharers of information in this digital exchange, and increase the pool of evidence-based information on GI and liver diseases on social media.12 However, there is limited published literature tailored to gastroenterologists and hepatologists. The goal of this article, therefore, is to provide a broad overview of best practices in the professional use of social media and highlight examples of novel applications in clinical practice.
Best practices: Getting started and maintaining a presence on social media
Social media can magnify your professional image, amplify your voice, and extend your reach and influence much faster than other methods. It also can be damaging if not used responsibly. Thus, we recommend the following approaches to responsible use of social media and cultivating your social media presence based on current evidence, professional organizations’ policy statements, and our combined experience. We initially presented these strategies during a Meet-the-Professor Luncheon at Digestive Disease Week® in Chicago (http://www.ddw.org/education/session-recordings).
Second, as with other aspects of medical training and practice, find a mentor to provide hands-on advice. This is particularly true if your general familiarity with the social media platforms is limited. If this is not available through your network of colleagues or workplace, we recommend exploring opportunities offered through your professional organization(s) such as the aforementioned Meet-the-Professor Luncheon at Digestive Diseases Week.
Third, know the privacy setting options on your social media platform(s) of choice and use them to your advantage. For example, on Facebook and Twitter, you can select an option that requests your permission before a friend or follower is added to your network. You also can tailor who (such as friends or followers only) can access your posted content directly. However, know that your content still may be made public if it is shared by one of your friends or followers.
Fourth, nurture your social media presence by sharing credible content deliberately, regularly, and, when appropriate, with attribution.
Fifth, diversify your content within the realm of your predefined objectives and/or goals and avoid a singular focus of self-promotion or the appearance of self-promotion. Top social media users suggest, and the authors agree, that your content should be only 25%-33% of your posts.
Sixth, thoroughly vet all content that you share. Avoid automatically sharing articles or posts because of a catchy headline. Read them before you post them. There may be details buried in them that are not credible or with which you do not agree.
Seventh, build community by connecting and engaging with other users on your social media platform(s) of choice.
Eighth, integrate multiple media (i.e., photos, videos, infographics) and/or social media platforms (i.e., embed link to YouTube or a website) to increase engagement.
Ninth, adhere to the code of ethics, governance, and privacy of the profession and of your employer.
Best practices: Privacy and governance in patient-oriented communication on social media
Two factors that have been of pivotal concern with the adoption of social media in the health care arena and led to many health care professionals being laggards as opposed to early adopters are privacy and governance. Will it violate the patient–provider relationship? What about the Health Insurance Portability and Accountability Act? How do I maintain boundaries between myself and the public at large? These are just a few of the questions that commonly are asked by those who are unfamiliar with social media etiquette for health care professionals. We highly recommend reviewing the position paper regarding online medical professionalism issued by the American College of Physicians and the Federation of State Medical Boards as a starting point.13 We believe the following to be contemporary guiding principles for GI health providers for maintaining a digital footprint on social media that reflects the ethical and professional standards of the field.
First, avoid sharing information that could be construed as a patient identifier without documented consent. This includes, but is not limited to, an identifiable specimen or photograph, and stories of care, rare conditions, and complications. Note that dates and location of care can lead to identification of a patient or care episode.
Second, recognize that personal and professional online profiles/pages are discoverable. Many advocate for separating the two as a means of shielding the public from elements of a private persona (i.e., family pictures and controversial opinions). However, the capacity to share and find comments and images on social media is much more powerful than the privacy settings on the various social media platforms. If you establish distinct personal and professional profiles, exercise caution before accepting friend or follow requests from patients on your personal profile. In addition, be cautious with your posts on private social media accounts because they rarely truly are private.
Third, avoid providing specific medical recommendations to individuals. This creates a patient–provider relationship and legal duty. Instead, recommend consultation with a health care provider and consider providing a link to general information on the topic (e.g., AGA information for patients at www.gastro.org/patientinfo).
Fourth, declare conflicts of interest, if applicable, when sharing information involving your clinical, research, and/or business practice.
Fifth, routinely monitor your online presence for accuracy and appropriateness of content posted by you and by others in reference to you. Know that our profession’s ethical standards for behavior extend to social media and we can be held accountable to colleagues and our employer if we violate them.
Many employers have become savvy to issues of governance in use of social media and institute policy recommendations to which employees are expected to adhere. If you are an employee, we recommend checking with your marketing and/or human resources department(s) in regards to this. If you are an employer and do not have such a policy on online professionalism, it is our hope that this article serves as a launching pad.
Novel applications for social media in clinical practice
Social media has been shown to be an effective medium for medical education through virtual journal clubs, moderated discussions or chats, and video sharing for teaching procedures, to name a few applications. Social media is used to collect data via polls or surveys, and to disseminate and track the views and downloads of published works. It is also a source for unsolicited, real-time feedback on patient experience and engagement through data-mining techniques, such as natural language processing and, more simply, for solicited feedback for patient satisfaction ratings. However, its role in academic promotion is less clear and is an area for which we see a great opportunity for growth.

Summary
We have outlined a high-level overview for why you should consider establishing and maintaining a professional presence on social media and how to accomplish this. These reasons include sharing information with colleagues, patients, and the public; amplifying the voice of physicians, a view that has diminished in the often-volatile health care environment; and promotion of the value of your work, be it patient care, advocacy, research, or education. You will have a smoother experience if you learn your local rules and policies and abide by our suggestions to avoid adverse outcomes. You will be most effective if you establish goals for your social media participation and revisit these goals over time for continued relevance and success and if you have consistent and valuable output that will support attainment of these goals. Welcome to the GI social media community! Be sure to follow Clinical Gastroenterology and Hepatology and the American Gastroenterological Association on Facebook (facebook.com/cghjournal and facebook.com/amergastroassn) and Twitter (@AGA_CGH and @AmerGastroAssn), and the coauthors (@DMGrayMD and @DrDeborahFisher) on Twitter.
References
1. Social Media Fact Sheet. Pew Research Center [updated January 12, 2017]. Available from http://www.pewinternet.org/fact-sheet/social-media/. Accessed: June 20, 2017.
2. Hesse B.W., Nelson D.E., Kreps G.L., et al. Trust and sources of health information: the impact of the Internet and its implications for health care providers: findings from the first Health Information National Trends Survey. Arch Intern Med. 2005;165:2618-24.
3. Moorhead S.A., Hazlett D.E., Harrison L., et al. A new dimension of health care: systematic review of the uses, benefits, and limitations of social media for health communication. J Med Internet Res. 2013;15:e85.
4. Chou W.Y., Hunt Y.M., Beckjord E.B. et al. Social media use in the United States: implications for health communication. J Med Internet Res. 2009;11:e48.
5. Chretien K.C., Kind T. Social media and clinical care: ethical, professional, and social implications. Circulation. 2013;27:1413-21.
6. Social Media ‘likes’ Healthcare. PwC Health Research Institute; 2012. Available from https://www.pwc.com/us/en/health-industries/health-research-institute/publications/pdf/health-care-social-media-report.pdf. Accessed: June 20, 2017.
7. Griffis H.M., Kilaru A.S., Werner R.M., et al. Use of social media across US hospitals: descriptive analysis of adoption and utilization. J Med Internet Res. 2014;16:e264.
8. Davis E.D., Tang S.J., Glover P.H., et al. Impact of social media on gastroenterologists in the United States. Dig Liver Dis. 2015;47:258-9.
9. Chiang A.L., Vartabedian B., Spiegel B. Harnessing the hashtag: a standard approach to GI dialogue on social media. Am J Gastroenterol. 2016;111:1082-4.
10. Reich J., Guo L., Hall J., et al. A survey of social media use and preferences in patients with inflammatory bowel disease. Inflamm Bowel Dis. 2016;22:2678-87.
11. Timms C., Forton D.M., Poullis A. Social media use in patients with inflammatory bowel disease and chronic viral hepatitis. Clin Med. 2014;14:215.
12. Prasad B. Social media, health care, and social networking. Gastrointest Endosc. 2013;77:492-5.
13. Farnan J.M., Snyder Sulmasy L., Worster B.K., et al. Online medical professionalism: patient and public relationships: policy statement from the American College of Physicians and the Federation of State Medical Boards. Ann Intern Med. 2013;158:620-7.
14. Cabrera D., Vartabedian B.S., Spinner R.J., et al. More than likes and tweets: creating social media portfolios for academic promotion and tenure. J Grad Med Educ. 2017;9:421-5.
15. Cabrera D. Mayo Clinic includes social media scholarship activities in academic advancement. Available from https://socialmedia.mayoclinic.org/2016/05/25/mayo-clinic-includes-social-media-scholarship-activities-in-academic-advancement/
Date: May 26, 2016. (Accessed: July 1, 2017).
16. Freitag C.E., Arnold M.A., Gardner J.M., et al. If you are not on social media, here’s what you’re missing! #DoTheThing. Arch Pathol Lab Med. 2017; (Epub ahead of print).
17. Stukus D.R. How I used twitter to get promoted in academic medicine. Available from http://www.kevinmd.com/blog/2016/10/used-twitter-get-promoted-academic-medicine.html. Date: October 9, 2016. (Accessed: July 1, 2017).
Dr. Gray is in the division of gastroenterology, hepatology, and nutrition, department of medicine, The Ohio State University College of Medicine, Columbus; Dr. Fisher is in the division of gastroenterology, department of medicine, Duke University, Durham, N.C. The authors disclose no conflicts of interest.
How to advise adolescents ISO drugs on the ‘dark web’
There was a time, not so long ago, when in the popular imagination, a drug deal involved an aging hippie in a tie-dyed shirt and love beads, copping a joint at a Dead concert. Today, however, in the age of the Internet and smartphones, a teenager in his bedroom can select, order, and have delivered to his door illicit drugs via the marketplaces on the “dark web.”
As the name implies, the dark web is a subterranean layer of the Internet that is mysterious, ominous, and sometimes lawless. It lies “beneath” the surface web – the layer where grandmothers post on Facebook and purchase on Amazon.
The dark web largely was the brainchild of three mathematicians at the Naval Research Laboratory as a means of encrypting messages exchanged by the intelligence community. They dubbed their project “Tor,” for “The Onion Router,” as the system consists of layer after layer of random relays, permitting anonymity on the Internet with little risk of tracking or surveillance.
This online underworld first came to my attention several years ago. The father of a teen who was being treated for disruptive mood dysregulation, attention-deficit disorder, and alcohol and cannabis use disorder called to inform me that his son had been arrested at Lollapalooza with hundreds of Adderall tablets and Xanax bars. Several weeks later, in session, the young man disclosed to me that he had found simple instructions online about installing Tor, creating a VPN (a virtual private network), accessing the dark web, transacting with bitcoin, and identifying drug marketplaces. He also demonstrated a detailed knowledge of chemical manufacturing in China, pill pressing in Canada, and money laundering in Switzerland.
With the air of an insider sharing “trade secrets,” this young man described how dealers on the dark web avoid detection and ensure secure delivery of the goods: latex gloves, vacuum sealing, and bleach dipping to obviate fingerprints – human and chemical. He said that dealers will send “dummy” packages to throw off the authorities and that buyers often will use the address of a clueless or absent neighbor. In his case, however, the parcels were delivered to his doorstep.
. For one thing, acquiring drugs in this way can seem less risky, as there is no chance of being robbed at gunpoint in a sketchy neighborhood or being busted for possession during a routine traffic stop.
Crossing over to the world of the dark web also can give an adolescent a sense of being clever and rebellious and of pulling a “fast one” on the parents – and on us, the clinicians who are treating them. And a teen who is interested in using illicit substances and plays “Call of Duty” from the comfort of his family’s basement without actual injury or death might assume that he can attain illicit drugs that are safe and inexpensive.
There are counterarguments to misinformation about the dark web. For example, contrary to the notion that buying drugs on the dark web minimizes interdiction or arrest, clinicians should point out that since international law enforcement shut down Silk Road and incarcerated its founder, Ross Ulbricht (also known as “Dread Pirate Roberts”) in 2013, hundreds of other dark web marketplaces such as AlphaBay and Hansa have been silenced and their operators prosecuted.
Moreover, the U.S. Department of Justice recently launched the Joint Criminal Opioid Darknet Enforcement (J-CODE) group, and the U.S. Postal Service Inspection Service reportedly has been hiring cybercrime and dark web specialists to combat drug trafficking. It might come as a surprise to a teen that, in a state where recreational marijuana is legal, transport and delivery of cannabis by the postal service elevates purchase and possession to the level of a violation of federal law. Informing even the most oblivious or oppositional adolescent that a drug felony can disqualify him for college grants and loans, impede his search for gainful employment, or prohibit him from obtaining a professional license, might give him a moment’s pause.
Adolescents seeking to buy drugs on the dark web should brace themselves for another shock. Whether lulled by custom after years of shopping on Amazon or using PayPal, or simply dulled by addiction, they might have a blind trust that bitcoin tumblers and “dark” escrow accounts will secure their payments. It will be rude awakening when they learn that the transfer and holding of currency on the dark web is vulnerable to hackers and to operators of the marketplaces – who are known to simply abscond with funds.
Currently, the drug marketplaces on the dark web represent a thin slice of the total illicit drug trade. These marketplaces, however, are growing quickly and offer buyers a virtual smorgasbord: the leading prescription drugs bought are Xanax and OxyContin, whereas 3,4-methylenedioxymethamphetamine (MDMA)/ecstasy and cannabis are the most commonly purchased controlled Schedule I substances, according to an article in The Economist. The estimated annual sales for 2016 ranged from $100 million to $200 million, but even more alarming is the percentage of substance abusers who have purchased drugs on the dark web: A recent article in The Independent reported that 13.2% of U.S. respondents self-reported making at least one purchase online, whereas in the United Kingdom, the self-reported percentage was 25.3 %, and in Finland, it was 41.4%.
Evidence abounds that drugs purchased on the dark web often are counterfeit and sometimes “dirty.” There is a report of “Viagra” containing cement dust, of “Ambien” containing haloperidol, and “Xanax” laced with fentanyl, the latter having been linked to several deaths and hospital admissions in the Bay Area in 2016. (Big Pharma, in fact, is engaged in surveillance and investigation of the sale of knockoffs online, The Telegraph reported in article about the proliferation of fake drugs available on the surface web).Unfortunately, attempting to reduce teen substance abuse using law enforcement measures directed at dark web marketplaces might be a game of whack-a-mole: As soon as one supply source is staunched, another surfaces. Indeed, addiction should be viewed not simply in terms of the biopsychosocial model, but also as an economic activity. Thus, it might be more beneficial for clinicians to concentrate our resources and efforts on curtailing the demand through education and treatment.
Dr. Marseille is a psychiatrist who works on the staff at a clinic in Winfield, Ill. His special interests include adolescent and addiction medicine, eating disorders, trauma, bipolar disorder, and the psychiatric manifestations of acute and chronic medical conditions.
There was a time, not so long ago, when in the popular imagination, a drug deal involved an aging hippie in a tie-dyed shirt and love beads, copping a joint at a Dead concert. Today, however, in the age of the Internet and smartphones, a teenager in his bedroom can select, order, and have delivered to his door illicit drugs via the marketplaces on the “dark web.”
As the name implies, the dark web is a subterranean layer of the Internet that is mysterious, ominous, and sometimes lawless. It lies “beneath” the surface web – the layer where grandmothers post on Facebook and purchase on Amazon.
The dark web largely was the brainchild of three mathematicians at the Naval Research Laboratory as a means of encrypting messages exchanged by the intelligence community. They dubbed their project “Tor,” for “The Onion Router,” as the system consists of layer after layer of random relays, permitting anonymity on the Internet with little risk of tracking or surveillance.
This online underworld first came to my attention several years ago. The father of a teen who was being treated for disruptive mood dysregulation, attention-deficit disorder, and alcohol and cannabis use disorder called to inform me that his son had been arrested at Lollapalooza with hundreds of Adderall tablets and Xanax bars. Several weeks later, in session, the young man disclosed to me that he had found simple instructions online about installing Tor, creating a VPN (a virtual private network), accessing the dark web, transacting with bitcoin, and identifying drug marketplaces. He also demonstrated a detailed knowledge of chemical manufacturing in China, pill pressing in Canada, and money laundering in Switzerland.
With the air of an insider sharing “trade secrets,” this young man described how dealers on the dark web avoid detection and ensure secure delivery of the goods: latex gloves, vacuum sealing, and bleach dipping to obviate fingerprints – human and chemical. He said that dealers will send “dummy” packages to throw off the authorities and that buyers often will use the address of a clueless or absent neighbor. In his case, however, the parcels were delivered to his doorstep.
. For one thing, acquiring drugs in this way can seem less risky, as there is no chance of being robbed at gunpoint in a sketchy neighborhood or being busted for possession during a routine traffic stop.
Crossing over to the world of the dark web also can give an adolescent a sense of being clever and rebellious and of pulling a “fast one” on the parents – and on us, the clinicians who are treating them. And a teen who is interested in using illicit substances and plays “Call of Duty” from the comfort of his family’s basement without actual injury or death might assume that he can attain illicit drugs that are safe and inexpensive.
There are counterarguments to misinformation about the dark web. For example, contrary to the notion that buying drugs on the dark web minimizes interdiction or arrest, clinicians should point out that since international law enforcement shut down Silk Road and incarcerated its founder, Ross Ulbricht (also known as “Dread Pirate Roberts”) in 2013, hundreds of other dark web marketplaces such as AlphaBay and Hansa have been silenced and their operators prosecuted.
Moreover, the U.S. Department of Justice recently launched the Joint Criminal Opioid Darknet Enforcement (J-CODE) group, and the U.S. Postal Service Inspection Service reportedly has been hiring cybercrime and dark web specialists to combat drug trafficking. It might come as a surprise to a teen that, in a state where recreational marijuana is legal, transport and delivery of cannabis by the postal service elevates purchase and possession to the level of a violation of federal law. Informing even the most oblivious or oppositional adolescent that a drug felony can disqualify him for college grants and loans, impede his search for gainful employment, or prohibit him from obtaining a professional license, might give him a moment’s pause.
Adolescents seeking to buy drugs on the dark web should brace themselves for another shock. Whether lulled by custom after years of shopping on Amazon or using PayPal, or simply dulled by addiction, they might have a blind trust that bitcoin tumblers and “dark” escrow accounts will secure their payments. It will be rude awakening when they learn that the transfer and holding of currency on the dark web is vulnerable to hackers and to operators of the marketplaces – who are known to simply abscond with funds.
Currently, the drug marketplaces on the dark web represent a thin slice of the total illicit drug trade. These marketplaces, however, are growing quickly and offer buyers a virtual smorgasbord: the leading prescription drugs bought are Xanax and OxyContin, whereas 3,4-methylenedioxymethamphetamine (MDMA)/ecstasy and cannabis are the most commonly purchased controlled Schedule I substances, according to an article in The Economist. The estimated annual sales for 2016 ranged from $100 million to $200 million, but even more alarming is the percentage of substance abusers who have purchased drugs on the dark web: A recent article in The Independent reported that 13.2% of U.S. respondents self-reported making at least one purchase online, whereas in the United Kingdom, the self-reported percentage was 25.3 %, and in Finland, it was 41.4%.
Evidence abounds that drugs purchased on the dark web often are counterfeit and sometimes “dirty.” There is a report of “Viagra” containing cement dust, of “Ambien” containing haloperidol, and “Xanax” laced with fentanyl, the latter having been linked to several deaths and hospital admissions in the Bay Area in 2016. (Big Pharma, in fact, is engaged in surveillance and investigation of the sale of knockoffs online, The Telegraph reported in article about the proliferation of fake drugs available on the surface web).Unfortunately, attempting to reduce teen substance abuse using law enforcement measures directed at dark web marketplaces might be a game of whack-a-mole: As soon as one supply source is staunched, another surfaces. Indeed, addiction should be viewed not simply in terms of the biopsychosocial model, but also as an economic activity. Thus, it might be more beneficial for clinicians to concentrate our resources and efforts on curtailing the demand through education and treatment.
Dr. Marseille is a psychiatrist who works on the staff at a clinic in Winfield, Ill. His special interests include adolescent and addiction medicine, eating disorders, trauma, bipolar disorder, and the psychiatric manifestations of acute and chronic medical conditions.
There was a time, not so long ago, when in the popular imagination, a drug deal involved an aging hippie in a tie-dyed shirt and love beads, copping a joint at a Dead concert. Today, however, in the age of the Internet and smartphones, a teenager in his bedroom can select, order, and have delivered to his door illicit drugs via the marketplaces on the “dark web.”
As the name implies, the dark web is a subterranean layer of the Internet that is mysterious, ominous, and sometimes lawless. It lies “beneath” the surface web – the layer where grandmothers post on Facebook and purchase on Amazon.
The dark web largely was the brainchild of three mathematicians at the Naval Research Laboratory as a means of encrypting messages exchanged by the intelligence community. They dubbed their project “Tor,” for “The Onion Router,” as the system consists of layer after layer of random relays, permitting anonymity on the Internet with little risk of tracking or surveillance.
This online underworld first came to my attention several years ago. The father of a teen who was being treated for disruptive mood dysregulation, attention-deficit disorder, and alcohol and cannabis use disorder called to inform me that his son had been arrested at Lollapalooza with hundreds of Adderall tablets and Xanax bars. Several weeks later, in session, the young man disclosed to me that he had found simple instructions online about installing Tor, creating a VPN (a virtual private network), accessing the dark web, transacting with bitcoin, and identifying drug marketplaces. He also demonstrated a detailed knowledge of chemical manufacturing in China, pill pressing in Canada, and money laundering in Switzerland.
With the air of an insider sharing “trade secrets,” this young man described how dealers on the dark web avoid detection and ensure secure delivery of the goods: latex gloves, vacuum sealing, and bleach dipping to obviate fingerprints – human and chemical. He said that dealers will send “dummy” packages to throw off the authorities and that buyers often will use the address of a clueless or absent neighbor. In his case, however, the parcels were delivered to his doorstep.
. For one thing, acquiring drugs in this way can seem less risky, as there is no chance of being robbed at gunpoint in a sketchy neighborhood or being busted for possession during a routine traffic stop.
Crossing over to the world of the dark web also can give an adolescent a sense of being clever and rebellious and of pulling a “fast one” on the parents – and on us, the clinicians who are treating them. And a teen who is interested in using illicit substances and plays “Call of Duty” from the comfort of his family’s basement without actual injury or death might assume that he can attain illicit drugs that are safe and inexpensive.
There are counterarguments to misinformation about the dark web. For example, contrary to the notion that buying drugs on the dark web minimizes interdiction or arrest, clinicians should point out that since international law enforcement shut down Silk Road and incarcerated its founder, Ross Ulbricht (also known as “Dread Pirate Roberts”) in 2013, hundreds of other dark web marketplaces such as AlphaBay and Hansa have been silenced and their operators prosecuted.
Moreover, the U.S. Department of Justice recently launched the Joint Criminal Opioid Darknet Enforcement (J-CODE) group, and the U.S. Postal Service Inspection Service reportedly has been hiring cybercrime and dark web specialists to combat drug trafficking. It might come as a surprise to a teen that, in a state where recreational marijuana is legal, transport and delivery of cannabis by the postal service elevates purchase and possession to the level of a violation of federal law. Informing even the most oblivious or oppositional adolescent that a drug felony can disqualify him for college grants and loans, impede his search for gainful employment, or prohibit him from obtaining a professional license, might give him a moment’s pause.
Adolescents seeking to buy drugs on the dark web should brace themselves for another shock. Whether lulled by custom after years of shopping on Amazon or using PayPal, or simply dulled by addiction, they might have a blind trust that bitcoin tumblers and “dark” escrow accounts will secure their payments. It will be rude awakening when they learn that the transfer and holding of currency on the dark web is vulnerable to hackers and to operators of the marketplaces – who are known to simply abscond with funds.
Currently, the drug marketplaces on the dark web represent a thin slice of the total illicit drug trade. These marketplaces, however, are growing quickly and offer buyers a virtual smorgasbord: the leading prescription drugs bought are Xanax and OxyContin, whereas 3,4-methylenedioxymethamphetamine (MDMA)/ecstasy and cannabis are the most commonly purchased controlled Schedule I substances, according to an article in The Economist. The estimated annual sales for 2016 ranged from $100 million to $200 million, but even more alarming is the percentage of substance abusers who have purchased drugs on the dark web: A recent article in The Independent reported that 13.2% of U.S. respondents self-reported making at least one purchase online, whereas in the United Kingdom, the self-reported percentage was 25.3 %, and in Finland, it was 41.4%.
Evidence abounds that drugs purchased on the dark web often are counterfeit and sometimes “dirty.” There is a report of “Viagra” containing cement dust, of “Ambien” containing haloperidol, and “Xanax” laced with fentanyl, the latter having been linked to several deaths and hospital admissions in the Bay Area in 2016. (Big Pharma, in fact, is engaged in surveillance and investigation of the sale of knockoffs online, The Telegraph reported in article about the proliferation of fake drugs available on the surface web).Unfortunately, attempting to reduce teen substance abuse using law enforcement measures directed at dark web marketplaces might be a game of whack-a-mole: As soon as one supply source is staunched, another surfaces. Indeed, addiction should be viewed not simply in terms of the biopsychosocial model, but also as an economic activity. Thus, it might be more beneficial for clinicians to concentrate our resources and efforts on curtailing the demand through education and treatment.
Dr. Marseille is a psychiatrist who works on the staff at a clinic in Winfield, Ill. His special interests include adolescent and addiction medicine, eating disorders, trauma, bipolar disorder, and the psychiatric manifestations of acute and chronic medical conditions.
Inaccurate depictions of inpatient psychiatry foster stigma
When it comes to the portrayal of physicians in popular culture, psychiatrists are second only to surgeons.1 Far too often, these portrayals of psychiatry have misrepresented our specialty – and stigmatized our patients. Some of the content produced by entertainment giants Netflix and Marvel is a case in point.
That Netflix-Marvel collaboration has proven fruitful, resulting in six television series spanning seven seasons of entertainment to date. Last year alone saw the release of three Netflix-Marvel productions, including Marvel’s “Iron Fist” in March, “The Defenders” in August, and “The Punisher” in November. Given the popularity and ease of streaming Marvel’s Netflix productions, these series have the potential to entertain and inform a wide audience. Unfortunately, however, part of their influence might be to perpetuate stigma and fear surrounding inpatient psychiatric care. Take the inaccurate portrayal of psychiatry in “Iron Fist” compared with the reality of modern psychiatric care, for example.
While hospitalized, Danny is repeatedly shown in restraints, including four-point restraints, a belt, and a straitjacket. The use of restraints is sometimes portrayed as unprovoked, without evidence of aggression on the part of Danny. In addition, it appears that he is left in restraints for extended periods of time, as he is shown, for example, waking up in the night still restrained. While the use of restraints may be warranted in instances of extreme aggression or violence, the current culture of inpatient psychiatric care has shifted toward safely minimizing the use of restraints and seclusion.2 Straitjackets might be an icon of a bygone era of psychiatric care, but they no longer are a mainstream form of restraint used in the United States. Modern best practices would not result in a nonthreatening patient being placed in restraints and left in them for an extended period of time as shown in “Iron Fist.”
Danny is shown being forcibly administered medications, even in nonthreatening situations. These medications are given via parenteral injection as well as orally, with a psychiatric technician shown roughly inserting a tongue depressor deep in Danny’s mouth, pouring pills into his oral cavity, and manually holding his mouth shut to ensure ingestion. In truth, psychiatric patients are sometimes given intramuscular injections of calming agents when their level of agitation threatens to harm others or themselves. For patients to be given an involuntary injection when not acutely threatening, typically states require some form of legal application for forced medication (a process that does not appear to have been observed in “Iron Fist”). As we know, forced oral medications never should be undertaken given the significant risk to patient (possible choking) as well as staff (bitten fingers).
Staff supervision of patients at the fictional Birch Psychiatric Hospital is extremely poor. At one point, a fellow patient enters Danny’s room dressed in a white coat, pretending to be his doctor. As the conversation progresses, the patient grabs a fork from Danny’s food tray and holds it to his throat, threatening to kill him before being gruffly dragged off by hospital staff. At another point, Danny is shown in four-point restraints, and a patient simply walks into his room and removes the restraints for him. By contrast, in a real, modern inpatient psychiatric facility unit, staff would be routinely providing safety checks on patients throughout the day. If a patient is at risk for violence, sharp metal cutlery would not be included on accessible food trays. Patient attire would be subject to hospital inspection and approval, so it is unclear how or where a patient would have access to a physician’s coat to pull off such an impersonation. And finally, if a patient were sufficiently agitated to require the use of four-point restraints, he or she would be closely supervised and not left alone in an open area where other patients could remove the restraints.
The hospital stays described in “Iron Fist” are very long, and it is strongly implied that psychiatric diagnoses are invented to prolong inpatient care indefinitely. Referring to the duration of his initial involuntary hold, one patient tells Danny: “That what they tell you? Seventy-two hours? (laughs) Me, I had a little incident inside a pharmacy. Seventy-two hours later, I’m a bipolar with mixed affective episodes layered atop a substance abuse disorder. That was 2 years ago. Billy was living under a bridge. Seventy-two hours later, he’s a paranoid personality disorder. That was just over a year ago. And Jimmy was screaming at people in Times Square. Seventy-two hours later, he’s a schizoaffective disorder. He’s been here almost 15 years. Don’t think you’ll be any different.”
Most modern inpatient psychiatric care is designed around short-term hospitalization (days to weeks rather than months to years) with a goal of reintegrating patients back into the community with ongoing outpatient care as soon as they can safely make that transition. In addition, to insinuate that psychiatrists invent diagnoses to keep patients “locked up” insults the integrity of the many dedicated mental health workers who provide care for an ill and often overlooked population.
The most egregious examples of poor psychiatric care portrayed in “Iron Fist” involve illegal or criminal activities. Video cameras placed throughout the hospital transmit a live feed to a shadowy figure who has no role in patient care. A psychiatric technician escorts Danny to a room full of patients hired to kill him. These particular concerns are outlandish enough that perhaps they don’t even need to be directly addressed, but for the sake of completeness it is worth noting that psychiatric hospitals are subject to rigorous oversight from numerous regulatory bodies to ensure that patient care is delivered in a safe and respectable manner and that all protected health information is accessible only by those whose treatment role necessitates such access.
Marvel’s “Iron Fist” seeks to entertain its audience, but it does a poor job of showing viewers a realistic portrayal of inpatient psychiatric care. The show presents a psychiatric hospital as the setting for inappropriate use of restraints, unwarranted use of forced oral and injectable medications, a lack of supervision leading to violence between patients, and even attempted murder accommodated by a hospital employee. Also, the show strongly implies that psychiatric diagnoses are invented for the purpose of continuing inpatient care indefinitely.
In sum, the psychiatric hospital is seen as an inhumane form of imprisonment from which one can only hope to escape with the benefit of a glowing, magical fist. Although this is fiction, these kinds of narratives can have very real consequences in perpetuating stigma against psychiatric care. Ultimately, such storylines undermine the public’s confidence in clinicians seeking to provide caring and compassionate care.
Dr. Weber is psychiatry department chair at Logan (Utah) Regional Hospital with Intermountain Healthcare.
References
1 J Nat Med Assoc. 2002 Jul. 94[7]:635-58.
2 Aggress Violent Behav. 2017;34:139-46.
When it comes to the portrayal of physicians in popular culture, psychiatrists are second only to surgeons.1 Far too often, these portrayals of psychiatry have misrepresented our specialty – and stigmatized our patients. Some of the content produced by entertainment giants Netflix and Marvel is a case in point.
That Netflix-Marvel collaboration has proven fruitful, resulting in six television series spanning seven seasons of entertainment to date. Last year alone saw the release of three Netflix-Marvel productions, including Marvel’s “Iron Fist” in March, “The Defenders” in August, and “The Punisher” in November. Given the popularity and ease of streaming Marvel’s Netflix productions, these series have the potential to entertain and inform a wide audience. Unfortunately, however, part of their influence might be to perpetuate stigma and fear surrounding inpatient psychiatric care. Take the inaccurate portrayal of psychiatry in “Iron Fist” compared with the reality of modern psychiatric care, for example.
While hospitalized, Danny is repeatedly shown in restraints, including four-point restraints, a belt, and a straitjacket. The use of restraints is sometimes portrayed as unprovoked, without evidence of aggression on the part of Danny. In addition, it appears that he is left in restraints for extended periods of time, as he is shown, for example, waking up in the night still restrained. While the use of restraints may be warranted in instances of extreme aggression or violence, the current culture of inpatient psychiatric care has shifted toward safely minimizing the use of restraints and seclusion.2 Straitjackets might be an icon of a bygone era of psychiatric care, but they no longer are a mainstream form of restraint used in the United States. Modern best practices would not result in a nonthreatening patient being placed in restraints and left in them for an extended period of time as shown in “Iron Fist.”
Danny is shown being forcibly administered medications, even in nonthreatening situations. These medications are given via parenteral injection as well as orally, with a psychiatric technician shown roughly inserting a tongue depressor deep in Danny’s mouth, pouring pills into his oral cavity, and manually holding his mouth shut to ensure ingestion. In truth, psychiatric patients are sometimes given intramuscular injections of calming agents when their level of agitation threatens to harm others or themselves. For patients to be given an involuntary injection when not acutely threatening, typically states require some form of legal application for forced medication (a process that does not appear to have been observed in “Iron Fist”). As we know, forced oral medications never should be undertaken given the significant risk to patient (possible choking) as well as staff (bitten fingers).
Staff supervision of patients at the fictional Birch Psychiatric Hospital is extremely poor. At one point, a fellow patient enters Danny’s room dressed in a white coat, pretending to be his doctor. As the conversation progresses, the patient grabs a fork from Danny’s food tray and holds it to his throat, threatening to kill him before being gruffly dragged off by hospital staff. At another point, Danny is shown in four-point restraints, and a patient simply walks into his room and removes the restraints for him. By contrast, in a real, modern inpatient psychiatric facility unit, staff would be routinely providing safety checks on patients throughout the day. If a patient is at risk for violence, sharp metal cutlery would not be included on accessible food trays. Patient attire would be subject to hospital inspection and approval, so it is unclear how or where a patient would have access to a physician’s coat to pull off such an impersonation. And finally, if a patient were sufficiently agitated to require the use of four-point restraints, he or she would be closely supervised and not left alone in an open area where other patients could remove the restraints.
The hospital stays described in “Iron Fist” are very long, and it is strongly implied that psychiatric diagnoses are invented to prolong inpatient care indefinitely. Referring to the duration of his initial involuntary hold, one patient tells Danny: “That what they tell you? Seventy-two hours? (laughs) Me, I had a little incident inside a pharmacy. Seventy-two hours later, I’m a bipolar with mixed affective episodes layered atop a substance abuse disorder. That was 2 years ago. Billy was living under a bridge. Seventy-two hours later, he’s a paranoid personality disorder. That was just over a year ago. And Jimmy was screaming at people in Times Square. Seventy-two hours later, he’s a schizoaffective disorder. He’s been here almost 15 years. Don’t think you’ll be any different.”
Most modern inpatient psychiatric care is designed around short-term hospitalization (days to weeks rather than months to years) with a goal of reintegrating patients back into the community with ongoing outpatient care as soon as they can safely make that transition. In addition, to insinuate that psychiatrists invent diagnoses to keep patients “locked up” insults the integrity of the many dedicated mental health workers who provide care for an ill and often overlooked population.
The most egregious examples of poor psychiatric care portrayed in “Iron Fist” involve illegal or criminal activities. Video cameras placed throughout the hospital transmit a live feed to a shadowy figure who has no role in patient care. A psychiatric technician escorts Danny to a room full of patients hired to kill him. These particular concerns are outlandish enough that perhaps they don’t even need to be directly addressed, but for the sake of completeness it is worth noting that psychiatric hospitals are subject to rigorous oversight from numerous regulatory bodies to ensure that patient care is delivered in a safe and respectable manner and that all protected health information is accessible only by those whose treatment role necessitates such access.
Marvel’s “Iron Fist” seeks to entertain its audience, but it does a poor job of showing viewers a realistic portrayal of inpatient psychiatric care. The show presents a psychiatric hospital as the setting for inappropriate use of restraints, unwarranted use of forced oral and injectable medications, a lack of supervision leading to violence between patients, and even attempted murder accommodated by a hospital employee. Also, the show strongly implies that psychiatric diagnoses are invented for the purpose of continuing inpatient care indefinitely.
In sum, the psychiatric hospital is seen as an inhumane form of imprisonment from which one can only hope to escape with the benefit of a glowing, magical fist. Although this is fiction, these kinds of narratives can have very real consequences in perpetuating stigma against psychiatric care. Ultimately, such storylines undermine the public’s confidence in clinicians seeking to provide caring and compassionate care.
Dr. Weber is psychiatry department chair at Logan (Utah) Regional Hospital with Intermountain Healthcare.
References
1 J Nat Med Assoc. 2002 Jul. 94[7]:635-58.
2 Aggress Violent Behav. 2017;34:139-46.
When it comes to the portrayal of physicians in popular culture, psychiatrists are second only to surgeons.1 Far too often, these portrayals of psychiatry have misrepresented our specialty – and stigmatized our patients. Some of the content produced by entertainment giants Netflix and Marvel is a case in point.
That Netflix-Marvel collaboration has proven fruitful, resulting in six television series spanning seven seasons of entertainment to date. Last year alone saw the release of three Netflix-Marvel productions, including Marvel’s “Iron Fist” in March, “The Defenders” in August, and “The Punisher” in November. Given the popularity and ease of streaming Marvel’s Netflix productions, these series have the potential to entertain and inform a wide audience. Unfortunately, however, part of their influence might be to perpetuate stigma and fear surrounding inpatient psychiatric care. Take the inaccurate portrayal of psychiatry in “Iron Fist” compared with the reality of modern psychiatric care, for example.
While hospitalized, Danny is repeatedly shown in restraints, including four-point restraints, a belt, and a straitjacket. The use of restraints is sometimes portrayed as unprovoked, without evidence of aggression on the part of Danny. In addition, it appears that he is left in restraints for extended periods of time, as he is shown, for example, waking up in the night still restrained. While the use of restraints may be warranted in instances of extreme aggression or violence, the current culture of inpatient psychiatric care has shifted toward safely minimizing the use of restraints and seclusion.2 Straitjackets might be an icon of a bygone era of psychiatric care, but they no longer are a mainstream form of restraint used in the United States. Modern best practices would not result in a nonthreatening patient being placed in restraints and left in them for an extended period of time as shown in “Iron Fist.”
Danny is shown being forcibly administered medications, even in nonthreatening situations. These medications are given via parenteral injection as well as orally, with a psychiatric technician shown roughly inserting a tongue depressor deep in Danny’s mouth, pouring pills into his oral cavity, and manually holding his mouth shut to ensure ingestion. In truth, psychiatric patients are sometimes given intramuscular injections of calming agents when their level of agitation threatens to harm others or themselves. For patients to be given an involuntary injection when not acutely threatening, typically states require some form of legal application for forced medication (a process that does not appear to have been observed in “Iron Fist”). As we know, forced oral medications never should be undertaken given the significant risk to patient (possible choking) as well as staff (bitten fingers).
Staff supervision of patients at the fictional Birch Psychiatric Hospital is extremely poor. At one point, a fellow patient enters Danny’s room dressed in a white coat, pretending to be his doctor. As the conversation progresses, the patient grabs a fork from Danny’s food tray and holds it to his throat, threatening to kill him before being gruffly dragged off by hospital staff. At another point, Danny is shown in four-point restraints, and a patient simply walks into his room and removes the restraints for him. By contrast, in a real, modern inpatient psychiatric facility unit, staff would be routinely providing safety checks on patients throughout the day. If a patient is at risk for violence, sharp metal cutlery would not be included on accessible food trays. Patient attire would be subject to hospital inspection and approval, so it is unclear how or where a patient would have access to a physician’s coat to pull off such an impersonation. And finally, if a patient were sufficiently agitated to require the use of four-point restraints, he or she would be closely supervised and not left alone in an open area where other patients could remove the restraints.
The hospital stays described in “Iron Fist” are very long, and it is strongly implied that psychiatric diagnoses are invented to prolong inpatient care indefinitely. Referring to the duration of his initial involuntary hold, one patient tells Danny: “That what they tell you? Seventy-two hours? (laughs) Me, I had a little incident inside a pharmacy. Seventy-two hours later, I’m a bipolar with mixed affective episodes layered atop a substance abuse disorder. That was 2 years ago. Billy was living under a bridge. Seventy-two hours later, he’s a paranoid personality disorder. That was just over a year ago. And Jimmy was screaming at people in Times Square. Seventy-two hours later, he’s a schizoaffective disorder. He’s been here almost 15 years. Don’t think you’ll be any different.”
Most modern inpatient psychiatric care is designed around short-term hospitalization (days to weeks rather than months to years) with a goal of reintegrating patients back into the community with ongoing outpatient care as soon as they can safely make that transition. In addition, to insinuate that psychiatrists invent diagnoses to keep patients “locked up” insults the integrity of the many dedicated mental health workers who provide care for an ill and often overlooked population.
The most egregious examples of poor psychiatric care portrayed in “Iron Fist” involve illegal or criminal activities. Video cameras placed throughout the hospital transmit a live feed to a shadowy figure who has no role in patient care. A psychiatric technician escorts Danny to a room full of patients hired to kill him. These particular concerns are outlandish enough that perhaps they don’t even need to be directly addressed, but for the sake of completeness it is worth noting that psychiatric hospitals are subject to rigorous oversight from numerous regulatory bodies to ensure that patient care is delivered in a safe and respectable manner and that all protected health information is accessible only by those whose treatment role necessitates such access.
Marvel’s “Iron Fist” seeks to entertain its audience, but it does a poor job of showing viewers a realistic portrayal of inpatient psychiatric care. The show presents a psychiatric hospital as the setting for inappropriate use of restraints, unwarranted use of forced oral and injectable medications, a lack of supervision leading to violence between patients, and even attempted murder accommodated by a hospital employee. Also, the show strongly implies that psychiatric diagnoses are invented for the purpose of continuing inpatient care indefinitely.
In sum, the psychiatric hospital is seen as an inhumane form of imprisonment from which one can only hope to escape with the benefit of a glowing, magical fist. Although this is fiction, these kinds of narratives can have very real consequences in perpetuating stigma against psychiatric care. Ultimately, such storylines undermine the public’s confidence in clinicians seeking to provide caring and compassionate care.
Dr. Weber is psychiatry department chair at Logan (Utah) Regional Hospital with Intermountain Healthcare.
References
1 J Nat Med Assoc. 2002 Jul. 94[7]:635-58.
2 Aggress Violent Behav. 2017;34:139-46.