User login
Defensive medicine’s stranglehold on the realities of practice
In the September 2017 issue of JAMA Neurology, Louis R. Caplan, MD, wrote an excellent editorial, “Patient care is all about stories.” He notes that we all hear from patients about a recurrence of their previous stroke deficits, typically caused by infections, medications, or metabolic changes.
His point is that, telling the difference between true vascular events and recrudescence of old deficits can be difficult, but generally can be gleaned by taking a thorough history. He also notes, quite correctly, that the generic, automated features of modern charting systems often make it harder to get the details you need from previous visits.
Obviously, being able to accurately tell the difference between them can save health care costs, too. In a study in the same issue, Mehmet Topcuoglo, MD, and his colleagues discuss methodologies to differentiate between the causes of recrudescence of stroke-related deficits. Currently, the main approach is to admit patients to the hospital, do a knee-jerk repeat work-up with MRI, magnetic resonance angiogram, and echocardiogram (typically ordered before the neurologist has even been told of the consult) and then conclude that nothing has changed neurologically and that it was all caused by a bladder infection.
Surely, if we had an accurate way of telling the difference between them with a careful history, we’d save a lot of time and money on unnecessary hospital admissions. Right?
It sounds good in principle, but, sadly, the answer is “probably not.”
This is where the idealism of medicine meets the reality of its practice.
In the world of the emergency department, time and resources are limited. Emergency medicine physicians don’t have the luxury of taking a detailed neurologic history, nor are they trained (or expected) to be able to do so. Their job is to decide what is (and isn’t) life-threatening and who does (or doesn’t) need to be admitted.
But probably the main reason why Dr. Topcuoglo and his colleagues’ methodologies will never be implemented is defensive medicine. It’s a heck of lot easier and safer for any doctor – emergency medicine, hospitalist, and neurologist – to admit the patient and order more studies than it is to get served for malpractice and have to defend why you didn’t do that.
People can bemoan defensive medicine and its costs all they want. But, if you’ve been sued, you won’t care. You’ll order any test to protect yourself. Claiming that you followed a guideline from a journal, no matter how well researched it was, will likely be worthless the one time a stroke was missed. It’s easy for a plaintiff’s attorney to find someone to say you fell below the standard of care for doing so.
For an example of where this stands, here’s something from personal experience: One of my patients went to the emergency department for recrudescence of an old left hemiparesis, likely caused by a urinary tract infection. This wasn’t the first time it had happened. A head CT was stable while a urine analysis was abnormal. Because of my schedule, I wasn’t in a position to go see him in the ED in an expedient fashion. The ED physician was planning on admitting him and called to notify me. Knowing the history, I suggested sending him home with treatment for the UTI and to follow up with me the next day.
I thought that seemed reasonable, but the ED doctor didn’t. He said, “If you want to do that, then I am going to document that it’s on your instructions, that you are assuming all responsibility for care and outcome if a stroke is missed, and that I entirely disagree with your decision.”
I’m sure another neurologist might have said, “Okay, tell him to come in here tomorrow,” and hung up, but I really don’t have that kind of fortitude or desire for conflict with another physician. So I backed down and let the person on the scene make the decision. I saw the patient later that day as a consult, all his tests (except the urine analysis in the ED) were fine, and he went home the next day. I’m sure the bill was at least $50,000 (what really got paid is another matter), and defensive medicine had, for better or worse, won out over probability and reason.
Dr. Caplan, quite correctly, emphasizes the importance of taking a careful history, and I absolutely agree with him. Unfortunately, the lack of time in the ED setting, and fears driven by legal consequences, often make a good history irrelevant. Even when it’s done, there are other forces that push it to the background in making medical decisions.
I’m not saying that’s a good thing – it isn’t. But that’s the way it is right now in American medicine, and this aspect of the system shows no sign of changing.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
In the September 2017 issue of JAMA Neurology, Louis R. Caplan, MD, wrote an excellent editorial, “Patient care is all about stories.” He notes that we all hear from patients about a recurrence of their previous stroke deficits, typically caused by infections, medications, or metabolic changes.
His point is that, telling the difference between true vascular events and recrudescence of old deficits can be difficult, but generally can be gleaned by taking a thorough history. He also notes, quite correctly, that the generic, automated features of modern charting systems often make it harder to get the details you need from previous visits.
Obviously, being able to accurately tell the difference between them can save health care costs, too. In a study in the same issue, Mehmet Topcuoglo, MD, and his colleagues discuss methodologies to differentiate between the causes of recrudescence of stroke-related deficits. Currently, the main approach is to admit patients to the hospital, do a knee-jerk repeat work-up with MRI, magnetic resonance angiogram, and echocardiogram (typically ordered before the neurologist has even been told of the consult) and then conclude that nothing has changed neurologically and that it was all caused by a bladder infection.
Surely, if we had an accurate way of telling the difference between them with a careful history, we’d save a lot of time and money on unnecessary hospital admissions. Right?
It sounds good in principle, but, sadly, the answer is “probably not.”
This is where the idealism of medicine meets the reality of its practice.
In the world of the emergency department, time and resources are limited. Emergency medicine physicians don’t have the luxury of taking a detailed neurologic history, nor are they trained (or expected) to be able to do so. Their job is to decide what is (and isn’t) life-threatening and who does (or doesn’t) need to be admitted.
But probably the main reason why Dr. Topcuoglo and his colleagues’ methodologies will never be implemented is defensive medicine. It’s a heck of lot easier and safer for any doctor – emergency medicine, hospitalist, and neurologist – to admit the patient and order more studies than it is to get served for malpractice and have to defend why you didn’t do that.
People can bemoan defensive medicine and its costs all they want. But, if you’ve been sued, you won’t care. You’ll order any test to protect yourself. Claiming that you followed a guideline from a journal, no matter how well researched it was, will likely be worthless the one time a stroke was missed. It’s easy for a plaintiff’s attorney to find someone to say you fell below the standard of care for doing so.
For an example of where this stands, here’s something from personal experience: One of my patients went to the emergency department for recrudescence of an old left hemiparesis, likely caused by a urinary tract infection. This wasn’t the first time it had happened. A head CT was stable while a urine analysis was abnormal. Because of my schedule, I wasn’t in a position to go see him in the ED in an expedient fashion. The ED physician was planning on admitting him and called to notify me. Knowing the history, I suggested sending him home with treatment for the UTI and to follow up with me the next day.
I thought that seemed reasonable, but the ED doctor didn’t. He said, “If you want to do that, then I am going to document that it’s on your instructions, that you are assuming all responsibility for care and outcome if a stroke is missed, and that I entirely disagree with your decision.”
I’m sure another neurologist might have said, “Okay, tell him to come in here tomorrow,” and hung up, but I really don’t have that kind of fortitude or desire for conflict with another physician. So I backed down and let the person on the scene make the decision. I saw the patient later that day as a consult, all his tests (except the urine analysis in the ED) were fine, and he went home the next day. I’m sure the bill was at least $50,000 (what really got paid is another matter), and defensive medicine had, for better or worse, won out over probability and reason.
Dr. Caplan, quite correctly, emphasizes the importance of taking a careful history, and I absolutely agree with him. Unfortunately, the lack of time in the ED setting, and fears driven by legal consequences, often make a good history irrelevant. Even when it’s done, there are other forces that push it to the background in making medical decisions.
I’m not saying that’s a good thing – it isn’t. But that’s the way it is right now in American medicine, and this aspect of the system shows no sign of changing.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
In the September 2017 issue of JAMA Neurology, Louis R. Caplan, MD, wrote an excellent editorial, “Patient care is all about stories.” He notes that we all hear from patients about a recurrence of their previous stroke deficits, typically caused by infections, medications, or metabolic changes.
His point is that, telling the difference between true vascular events and recrudescence of old deficits can be difficult, but generally can be gleaned by taking a thorough history. He also notes, quite correctly, that the generic, automated features of modern charting systems often make it harder to get the details you need from previous visits.
Obviously, being able to accurately tell the difference between them can save health care costs, too. In a study in the same issue, Mehmet Topcuoglo, MD, and his colleagues discuss methodologies to differentiate between the causes of recrudescence of stroke-related deficits. Currently, the main approach is to admit patients to the hospital, do a knee-jerk repeat work-up with MRI, magnetic resonance angiogram, and echocardiogram (typically ordered before the neurologist has even been told of the consult) and then conclude that nothing has changed neurologically and that it was all caused by a bladder infection.
Surely, if we had an accurate way of telling the difference between them with a careful history, we’d save a lot of time and money on unnecessary hospital admissions. Right?
It sounds good in principle, but, sadly, the answer is “probably not.”
This is where the idealism of medicine meets the reality of its practice.
In the world of the emergency department, time and resources are limited. Emergency medicine physicians don’t have the luxury of taking a detailed neurologic history, nor are they trained (or expected) to be able to do so. Their job is to decide what is (and isn’t) life-threatening and who does (or doesn’t) need to be admitted.
But probably the main reason why Dr. Topcuoglo and his colleagues’ methodologies will never be implemented is defensive medicine. It’s a heck of lot easier and safer for any doctor – emergency medicine, hospitalist, and neurologist – to admit the patient and order more studies than it is to get served for malpractice and have to defend why you didn’t do that.
People can bemoan defensive medicine and its costs all they want. But, if you’ve been sued, you won’t care. You’ll order any test to protect yourself. Claiming that you followed a guideline from a journal, no matter how well researched it was, will likely be worthless the one time a stroke was missed. It’s easy for a plaintiff’s attorney to find someone to say you fell below the standard of care for doing so.
For an example of where this stands, here’s something from personal experience: One of my patients went to the emergency department for recrudescence of an old left hemiparesis, likely caused by a urinary tract infection. This wasn’t the first time it had happened. A head CT was stable while a urine analysis was abnormal. Because of my schedule, I wasn’t in a position to go see him in the ED in an expedient fashion. The ED physician was planning on admitting him and called to notify me. Knowing the history, I suggested sending him home with treatment for the UTI and to follow up with me the next day.
I thought that seemed reasonable, but the ED doctor didn’t. He said, “If you want to do that, then I am going to document that it’s on your instructions, that you are assuming all responsibility for care and outcome if a stroke is missed, and that I entirely disagree with your decision.”
I’m sure another neurologist might have said, “Okay, tell him to come in here tomorrow,” and hung up, but I really don’t have that kind of fortitude or desire for conflict with another physician. So I backed down and let the person on the scene make the decision. I saw the patient later that day as a consult, all his tests (except the urine analysis in the ED) were fine, and he went home the next day. I’m sure the bill was at least $50,000 (what really got paid is another matter), and defensive medicine had, for better or worse, won out over probability and reason.
Dr. Caplan, quite correctly, emphasizes the importance of taking a careful history, and I absolutely agree with him. Unfortunately, the lack of time in the ED setting, and fears driven by legal consequences, often make a good history irrelevant. Even when it’s done, there are other forces that push it to the background in making medical decisions.
I’m not saying that’s a good thing – it isn’t. But that’s the way it is right now in American medicine, and this aspect of the system shows no sign of changing.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Risking it all on the miracle of teamwork
On Feb. 4, 2018, with his team narrowly leading the New England Patriots in Super Bowl 52, Philadelphia Eagles head coach Doug Pederson made an audacious 4th-and-goal call. At the suggestion of backup quarterback Nick Foles, Pederson chose to rely on his team’s ability to execute the “Philly Special.” This was a risky trick play that was rehearsed but never tested, and one which could prove disastrous unless executed just right. With 34 seconds left in the first half, the Eagles pulled it off. Foles caught the ball in the end zone, securing his team’s place in football history and becoming the first quarterback to both throw for and catch a touchdown in one Super Bowl. He was named MVP and led the team to its first NFL title in 58 years.
For those of us who call Philadelphia our home, Super Bowl 52 represented so much more than just a victory, it was a miracle. We have long endured the highs and lows of Philadelphia football, watching as year after year our hopes were dashed by coaches and players who showed such promise, yet demonstrated such disappointment. But this year everything changed.
True fans could sense something different in the weeks leading up to that cold February day in Minneapolis. As the Eagle’s chances of competing in the Super Bowl grew more and more possible, the narrative wasn’t about any star player or member of the coaching staff, but instead the story of an incredible team. Even after the injury of starting quarterback and football phenom Carson Wentz in week 14, players and fans never lost hope in the promise of victory. Finally, Philadelphia had the team that could, and would, pull off something that had heretofore seemed like only an impossible dream.
It occurs to us that physicians should find the story of the Philadelphia Eagles not only inspirational, but also aspirational, even more so after reading the original research published by Dr. Richard Young, et al. in the February issue of Family Medicine.1 In this article, Dr. Young and his colleagues observed physicians during 982 patient encounters. The group measured the total visit time, face-to-face time, non-face time, and EHR work time (before, during, and after patient hours). The results weren’t surprising: Physicians spend more time working in the EHR than they spend in face-to-face time with patients.
This study confirmed prior work done by Ardnt et al. published in the fall in Annals of Family Medicine,2 which demonstrated that “primary care physicians spend more than half their workday, nearly 6 hours, interacting with the EHR during and after clinic hours.” Sadly, despite improving technology, the chasm between interacting with computers and interacting with actual patients only seems to be widening. To preserve the sanctity of the physician-patient relationship, we are forced to consider a completely new approach to how we practice: team-based care.
Team-based care isn’t a new idea, but it is being embraced with new fervor in the era of electronic health records. This is because the blessing — and curse — of the EHR is the vast amount of information that can be stored and accessed while caring for patients. To take advantage of this, doctors have been forced to become the primary agents for data entry and retrieval, something that is nearly impossible to do effectively while performing the cognitive work of a highly educated clinician. Rather than allowing us to take better care of patients, EHRs seem to have a paradoxical effect, limiting “face-to-face” time and squelching our efforts to address anything outside the immediate issues at hand. To improve the experience for us and our patients, we need to begin to rely on others.
To start, consider how a team can help support your documentation. As we’ve written about before, scribe services can be a tremendous benefit but aren’t the only way to improve efficiency. Medical assistants and nursing staff need to be encouraged to operate at the top level of their license, documenting where allowable and even queueing up orders, medication refills, and preventative care interventions when appropriate. This can be tremendously useful during previsit planning and can ensure that nothing is missed during the patient encounter.
Team-based care can also extend far beyond the EHR. For example, care coordinators can be employed to focus on specific high- and rising-risk patient populations. These health care professionals (typically nurses) reach out directly to patients and review their care, and even schedule visits with patients independently of the physician. This establishes therapeutic relationships that have been shown to prevent disease exacerbations and hospital readmissions, greatly reducing the cost of care.
Some facilities are now also using scheduling advocates, charged with facilitating referrals, arranging specialist and diagnostic appointments, and following up with patients to make sure they’ve successfully navigated the health care landscape. Behavioral health specialists and clinical pharmacists are also making their way into physician practices to expand the scope of offerings and decompress the burden on physicians. While these all have an associated cost, changes in the way physicians get paid are making the extra support economical and often necessary to satisfy the requirements under risk-based and fee-for-value contracts. We also predict that practices that choose to eschew the team approach to care will lose a competitive advantage in a health care market that is becoming more and more consumer driven. Patients, as well as providers, see the benefits of highly effective care teams.
Following the Eagles’ dramatic Super Bowl victory, coach Doug Pederson addressed his team in the locker room with these words: “This is a team game. As we’ve said before, an individual can make a difference, but a team makes a miracle.” While we as physicians may easily become jaded by the “miracles” of modern medicine, our patients have not yet lost hope in our ability to deliver on the promise of victory. To meet their expectations, we need to acknowledge that we will no longer be able to make it into the end zone alone; instead — in the game of modern medicine — we’ll need a team to take us there.
- Young RA, Burge SK, Kumar KA, Wilson JM, Ortiz DF. A Time-Motion Study of Primary Care Physicians’ Work in the Electronic Health Record Era. Fam Med. 2018 February;50(2):91-9.
- Arndt BG, Beasley JW, Watkinson MD, Temte JL, Tuan W-J, Sinsky CA, Gilchrist VJ..Ann Fam Med. 2017 September/October;15(5)5:419-26.
Dr. Skolnik is a professor of family and community medicine at Jefferson Medical College, Philadelphia, and an associate director of the family medicine residency program at Abington (Pa.) Jefferson Health. Dr. Notte is a family physician and Associate Chief Medical Information Officer for Jefferson Health. Follow him on twitter (@doctornotte).
On Feb. 4, 2018, with his team narrowly leading the New England Patriots in Super Bowl 52, Philadelphia Eagles head coach Doug Pederson made an audacious 4th-and-goal call. At the suggestion of backup quarterback Nick Foles, Pederson chose to rely on his team’s ability to execute the “Philly Special.” This was a risky trick play that was rehearsed but never tested, and one which could prove disastrous unless executed just right. With 34 seconds left in the first half, the Eagles pulled it off. Foles caught the ball in the end zone, securing his team’s place in football history and becoming the first quarterback to both throw for and catch a touchdown in one Super Bowl. He was named MVP and led the team to its first NFL title in 58 years.
For those of us who call Philadelphia our home, Super Bowl 52 represented so much more than just a victory, it was a miracle. We have long endured the highs and lows of Philadelphia football, watching as year after year our hopes were dashed by coaches and players who showed such promise, yet demonstrated such disappointment. But this year everything changed.
True fans could sense something different in the weeks leading up to that cold February day in Minneapolis. As the Eagle’s chances of competing in the Super Bowl grew more and more possible, the narrative wasn’t about any star player or member of the coaching staff, but instead the story of an incredible team. Even after the injury of starting quarterback and football phenom Carson Wentz in week 14, players and fans never lost hope in the promise of victory. Finally, Philadelphia had the team that could, and would, pull off something that had heretofore seemed like only an impossible dream.
It occurs to us that physicians should find the story of the Philadelphia Eagles not only inspirational, but also aspirational, even more so after reading the original research published by Dr. Richard Young, et al. in the February issue of Family Medicine.1 In this article, Dr. Young and his colleagues observed physicians during 982 patient encounters. The group measured the total visit time, face-to-face time, non-face time, and EHR work time (before, during, and after patient hours). The results weren’t surprising: Physicians spend more time working in the EHR than they spend in face-to-face time with patients.
This study confirmed prior work done by Ardnt et al. published in the fall in Annals of Family Medicine,2 which demonstrated that “primary care physicians spend more than half their workday, nearly 6 hours, interacting with the EHR during and after clinic hours.” Sadly, despite improving technology, the chasm between interacting with computers and interacting with actual patients only seems to be widening. To preserve the sanctity of the physician-patient relationship, we are forced to consider a completely new approach to how we practice: team-based care.
Team-based care isn’t a new idea, but it is being embraced with new fervor in the era of electronic health records. This is because the blessing — and curse — of the EHR is the vast amount of information that can be stored and accessed while caring for patients. To take advantage of this, doctors have been forced to become the primary agents for data entry and retrieval, something that is nearly impossible to do effectively while performing the cognitive work of a highly educated clinician. Rather than allowing us to take better care of patients, EHRs seem to have a paradoxical effect, limiting “face-to-face” time and squelching our efforts to address anything outside the immediate issues at hand. To improve the experience for us and our patients, we need to begin to rely on others.
To start, consider how a team can help support your documentation. As we’ve written about before, scribe services can be a tremendous benefit but aren’t the only way to improve efficiency. Medical assistants and nursing staff need to be encouraged to operate at the top level of their license, documenting where allowable and even queueing up orders, medication refills, and preventative care interventions when appropriate. This can be tremendously useful during previsit planning and can ensure that nothing is missed during the patient encounter.
Team-based care can also extend far beyond the EHR. For example, care coordinators can be employed to focus on specific high- and rising-risk patient populations. These health care professionals (typically nurses) reach out directly to patients and review their care, and even schedule visits with patients independently of the physician. This establishes therapeutic relationships that have been shown to prevent disease exacerbations and hospital readmissions, greatly reducing the cost of care.
Some facilities are now also using scheduling advocates, charged with facilitating referrals, arranging specialist and diagnostic appointments, and following up with patients to make sure they’ve successfully navigated the health care landscape. Behavioral health specialists and clinical pharmacists are also making their way into physician practices to expand the scope of offerings and decompress the burden on physicians. While these all have an associated cost, changes in the way physicians get paid are making the extra support economical and often necessary to satisfy the requirements under risk-based and fee-for-value contracts. We also predict that practices that choose to eschew the team approach to care will lose a competitive advantage in a health care market that is becoming more and more consumer driven. Patients, as well as providers, see the benefits of highly effective care teams.
Following the Eagles’ dramatic Super Bowl victory, coach Doug Pederson addressed his team in the locker room with these words: “This is a team game. As we’ve said before, an individual can make a difference, but a team makes a miracle.” While we as physicians may easily become jaded by the “miracles” of modern medicine, our patients have not yet lost hope in our ability to deliver on the promise of victory. To meet their expectations, we need to acknowledge that we will no longer be able to make it into the end zone alone; instead — in the game of modern medicine — we’ll need a team to take us there.
- Young RA, Burge SK, Kumar KA, Wilson JM, Ortiz DF. A Time-Motion Study of Primary Care Physicians’ Work in the Electronic Health Record Era. Fam Med. 2018 February;50(2):91-9.
- Arndt BG, Beasley JW, Watkinson MD, Temte JL, Tuan W-J, Sinsky CA, Gilchrist VJ..Ann Fam Med. 2017 September/October;15(5)5:419-26.
Dr. Skolnik is a professor of family and community medicine at Jefferson Medical College, Philadelphia, and an associate director of the family medicine residency program at Abington (Pa.) Jefferson Health. Dr. Notte is a family physician and Associate Chief Medical Information Officer for Jefferson Health. Follow him on twitter (@doctornotte).
On Feb. 4, 2018, with his team narrowly leading the New England Patriots in Super Bowl 52, Philadelphia Eagles head coach Doug Pederson made an audacious 4th-and-goal call. At the suggestion of backup quarterback Nick Foles, Pederson chose to rely on his team’s ability to execute the “Philly Special.” This was a risky trick play that was rehearsed but never tested, and one which could prove disastrous unless executed just right. With 34 seconds left in the first half, the Eagles pulled it off. Foles caught the ball in the end zone, securing his team’s place in football history and becoming the first quarterback to both throw for and catch a touchdown in one Super Bowl. He was named MVP and led the team to its first NFL title in 58 years.
For those of us who call Philadelphia our home, Super Bowl 52 represented so much more than just a victory, it was a miracle. We have long endured the highs and lows of Philadelphia football, watching as year after year our hopes were dashed by coaches and players who showed such promise, yet demonstrated such disappointment. But this year everything changed.
True fans could sense something different in the weeks leading up to that cold February day in Minneapolis. As the Eagle’s chances of competing in the Super Bowl grew more and more possible, the narrative wasn’t about any star player or member of the coaching staff, but instead the story of an incredible team. Even after the injury of starting quarterback and football phenom Carson Wentz in week 14, players and fans never lost hope in the promise of victory. Finally, Philadelphia had the team that could, and would, pull off something that had heretofore seemed like only an impossible dream.
It occurs to us that physicians should find the story of the Philadelphia Eagles not only inspirational, but also aspirational, even more so after reading the original research published by Dr. Richard Young, et al. in the February issue of Family Medicine.1 In this article, Dr. Young and his colleagues observed physicians during 982 patient encounters. The group measured the total visit time, face-to-face time, non-face time, and EHR work time (before, during, and after patient hours). The results weren’t surprising: Physicians spend more time working in the EHR than they spend in face-to-face time with patients.
This study confirmed prior work done by Ardnt et al. published in the fall in Annals of Family Medicine,2 which demonstrated that “primary care physicians spend more than half their workday, nearly 6 hours, interacting with the EHR during and after clinic hours.” Sadly, despite improving technology, the chasm between interacting with computers and interacting with actual patients only seems to be widening. To preserve the sanctity of the physician-patient relationship, we are forced to consider a completely new approach to how we practice: team-based care.
Team-based care isn’t a new idea, but it is being embraced with new fervor in the era of electronic health records. This is because the blessing — and curse — of the EHR is the vast amount of information that can be stored and accessed while caring for patients. To take advantage of this, doctors have been forced to become the primary agents for data entry and retrieval, something that is nearly impossible to do effectively while performing the cognitive work of a highly educated clinician. Rather than allowing us to take better care of patients, EHRs seem to have a paradoxical effect, limiting “face-to-face” time and squelching our efforts to address anything outside the immediate issues at hand. To improve the experience for us and our patients, we need to begin to rely on others.
To start, consider how a team can help support your documentation. As we’ve written about before, scribe services can be a tremendous benefit but aren’t the only way to improve efficiency. Medical assistants and nursing staff need to be encouraged to operate at the top level of their license, documenting where allowable and even queueing up orders, medication refills, and preventative care interventions when appropriate. This can be tremendously useful during previsit planning and can ensure that nothing is missed during the patient encounter.
Team-based care can also extend far beyond the EHR. For example, care coordinators can be employed to focus on specific high- and rising-risk patient populations. These health care professionals (typically nurses) reach out directly to patients and review their care, and even schedule visits with patients independently of the physician. This establishes therapeutic relationships that have been shown to prevent disease exacerbations and hospital readmissions, greatly reducing the cost of care.
Some facilities are now also using scheduling advocates, charged with facilitating referrals, arranging specialist and diagnostic appointments, and following up with patients to make sure they’ve successfully navigated the health care landscape. Behavioral health specialists and clinical pharmacists are also making their way into physician practices to expand the scope of offerings and decompress the burden on physicians. While these all have an associated cost, changes in the way physicians get paid are making the extra support economical and often necessary to satisfy the requirements under risk-based and fee-for-value contracts. We also predict that practices that choose to eschew the team approach to care will lose a competitive advantage in a health care market that is becoming more and more consumer driven. Patients, as well as providers, see the benefits of highly effective care teams.
Following the Eagles’ dramatic Super Bowl victory, coach Doug Pederson addressed his team in the locker room with these words: “This is a team game. As we’ve said before, an individual can make a difference, but a team makes a miracle.” While we as physicians may easily become jaded by the “miracles” of modern medicine, our patients have not yet lost hope in our ability to deliver on the promise of victory. To meet their expectations, we need to acknowledge that we will no longer be able to make it into the end zone alone; instead — in the game of modern medicine — we’ll need a team to take us there.
- Young RA, Burge SK, Kumar KA, Wilson JM, Ortiz DF. A Time-Motion Study of Primary Care Physicians’ Work in the Electronic Health Record Era. Fam Med. 2018 February;50(2):91-9.
- Arndt BG, Beasley JW, Watkinson MD, Temte JL, Tuan W-J, Sinsky CA, Gilchrist VJ..Ann Fam Med. 2017 September/October;15(5)5:419-26.
Dr. Skolnik is a professor of family and community medicine at Jefferson Medical College, Philadelphia, and an associate director of the family medicine residency program at Abington (Pa.) Jefferson Health. Dr. Notte is a family physician and Associate Chief Medical Information Officer for Jefferson Health. Follow him on twitter (@doctornotte).
Facing the world
***
Edgar is 86. His COPD recently caught up with him, and he needs oxygen. The nasal prongs that deliver it are irritating, but what really bothers him about them is that he won’t attend any activities in his assisted living facility wearing nasal prongs and trailing a tank.
***
Brenda comes in with a large bandage on her forehead. She takes it off to show me a large, jagged, fully-healed scar. Four months ago she gashed her head on an appliance and didn’t get medical help in time to have the wound properly stitched.
“My bangs aren’t thick enough to cover it,” she says. “My daughter asks why I wear a Band-Aid all the time,” says Brenda. But Brenda would rather walk around with a large bandage on her forehead. Just as Edgar won’t let anyone see him sick and diminished, Brenda won’t let anyone see her face damaged.
***
Stella has lymphoma. While she was on chemotherapy, she stayed put at home and avoided crowds to avoid catching someone’s virus. Once chemo was done, she was able to fly to Tallahassee, Fla., to see her new granddaughter Genevieve.
Unfortunately, her lymphoma recurred sooner than she and her doctors had hoped. Now Stella is on a new drug. This seems to be helping, but it puts her back at risk for infections in crowds.
And on planes. “Will you be able to visit Genevieve in Florida?” I ask.
Her husband Ben interjects. “Her doctors say she can,” he said, “but she would have to wear a mask on the plane, and Stella won’t wear a mask.”
***
Malcolm comes in now and then for this and that. This time, he is here for a skin check. At each visit he brings me up to date on an endless family lawsuit over a contested estate. Its subplots could script a whole Netflix series.
When I’m done with the skin check, Malcolm says, “also, I’d like Botox on my forehead.”
“OK,” I say. I don’t ask why, but Malcolm answers anyway.
“The lawsuit is finally coming to a head,” he says. “One of the nephews contesting the will is flying in from Indonesia, and the trial gets underway in Kentucky next week. I never would have started this fight, but since my charming relatives did, I’m in it to win it.”
I wish him luck.
“That’s why I want Botox,” he says. “I’m going to testify, and I want to look my confident best.”
Go, Malcolm!
While we’re talking Botox, I recall Amy, a well-traveled consultant who gave lectures all over.
“I’m curious,” I once asked her. “What do people say to you after you get Botox? Do they notice?”
“That’s interesting,” she said. “When I speak to groups, my face is projected onto large screens. That makes my wrinkles look like the Grand Canyon.
“When I started doing Botox, a man came over to me after a lecture and said, ‘I’ve heard you before, but this time you were, somehow, more cogent and compelling.’
“I thanked him, of course,” said Amy with a smile. “But the speech he was praising was the exact same speech he’d heard the first time.”
***
The rest of us try to put on the best face we can, assisted (in descending order of importance) by: makers of cosmetics, estheticians, dentists, plastic surgeons, and – oh yes – even dermatologists.
Happy to assist!
Dr. Rockoff practices dermatology in Brookline, Mass., and is a longtime contributor to Dermatology News. He serves on the clinical faculty at Tufts University, Boston, and has taught senior medical students and other trainees for 30 years. His second book, “Act Like a Doctor, Think Like a Patient,” is available at amazon.com and barnesandnoble.com. Write to him at [email protected].
***
Edgar is 86. His COPD recently caught up with him, and he needs oxygen. The nasal prongs that deliver it are irritating, but what really bothers him about them is that he won’t attend any activities in his assisted living facility wearing nasal prongs and trailing a tank.
***
Brenda comes in with a large bandage on her forehead. She takes it off to show me a large, jagged, fully-healed scar. Four months ago she gashed her head on an appliance and didn’t get medical help in time to have the wound properly stitched.
“My bangs aren’t thick enough to cover it,” she says. “My daughter asks why I wear a Band-Aid all the time,” says Brenda. But Brenda would rather walk around with a large bandage on her forehead. Just as Edgar won’t let anyone see him sick and diminished, Brenda won’t let anyone see her face damaged.
***
Stella has lymphoma. While she was on chemotherapy, she stayed put at home and avoided crowds to avoid catching someone’s virus. Once chemo was done, she was able to fly to Tallahassee, Fla., to see her new granddaughter Genevieve.
Unfortunately, her lymphoma recurred sooner than she and her doctors had hoped. Now Stella is on a new drug. This seems to be helping, but it puts her back at risk for infections in crowds.
And on planes. “Will you be able to visit Genevieve in Florida?” I ask.
Her husband Ben interjects. “Her doctors say she can,” he said, “but she would have to wear a mask on the plane, and Stella won’t wear a mask.”
***
Malcolm comes in now and then for this and that. This time, he is here for a skin check. At each visit he brings me up to date on an endless family lawsuit over a contested estate. Its subplots could script a whole Netflix series.
When I’m done with the skin check, Malcolm says, “also, I’d like Botox on my forehead.”
“OK,” I say. I don’t ask why, but Malcolm answers anyway.
“The lawsuit is finally coming to a head,” he says. “One of the nephews contesting the will is flying in from Indonesia, and the trial gets underway in Kentucky next week. I never would have started this fight, but since my charming relatives did, I’m in it to win it.”
I wish him luck.
“That’s why I want Botox,” he says. “I’m going to testify, and I want to look my confident best.”
Go, Malcolm!
While we’re talking Botox, I recall Amy, a well-traveled consultant who gave lectures all over.
“I’m curious,” I once asked her. “What do people say to you after you get Botox? Do they notice?”
“That’s interesting,” she said. “When I speak to groups, my face is projected onto large screens. That makes my wrinkles look like the Grand Canyon.
“When I started doing Botox, a man came over to me after a lecture and said, ‘I’ve heard you before, but this time you were, somehow, more cogent and compelling.’
“I thanked him, of course,” said Amy with a smile. “But the speech he was praising was the exact same speech he’d heard the first time.”
***
The rest of us try to put on the best face we can, assisted (in descending order of importance) by: makers of cosmetics, estheticians, dentists, plastic surgeons, and – oh yes – even dermatologists.
Happy to assist!
Dr. Rockoff practices dermatology in Brookline, Mass., and is a longtime contributor to Dermatology News. He serves on the clinical faculty at Tufts University, Boston, and has taught senior medical students and other trainees for 30 years. His second book, “Act Like a Doctor, Think Like a Patient,” is available at amazon.com and barnesandnoble.com. Write to him at [email protected].
***
Edgar is 86. His COPD recently caught up with him, and he needs oxygen. The nasal prongs that deliver it are irritating, but what really bothers him about them is that he won’t attend any activities in his assisted living facility wearing nasal prongs and trailing a tank.
***
Brenda comes in with a large bandage on her forehead. She takes it off to show me a large, jagged, fully-healed scar. Four months ago she gashed her head on an appliance and didn’t get medical help in time to have the wound properly stitched.
“My bangs aren’t thick enough to cover it,” she says. “My daughter asks why I wear a Band-Aid all the time,” says Brenda. But Brenda would rather walk around with a large bandage on her forehead. Just as Edgar won’t let anyone see him sick and diminished, Brenda won’t let anyone see her face damaged.
***
Stella has lymphoma. While she was on chemotherapy, she stayed put at home and avoided crowds to avoid catching someone’s virus. Once chemo was done, she was able to fly to Tallahassee, Fla., to see her new granddaughter Genevieve.
Unfortunately, her lymphoma recurred sooner than she and her doctors had hoped. Now Stella is on a new drug. This seems to be helping, but it puts her back at risk for infections in crowds.
And on planes. “Will you be able to visit Genevieve in Florida?” I ask.
Her husband Ben interjects. “Her doctors say she can,” he said, “but she would have to wear a mask on the plane, and Stella won’t wear a mask.”
***
Malcolm comes in now and then for this and that. This time, he is here for a skin check. At each visit he brings me up to date on an endless family lawsuit over a contested estate. Its subplots could script a whole Netflix series.
When I’m done with the skin check, Malcolm says, “also, I’d like Botox on my forehead.”
“OK,” I say. I don’t ask why, but Malcolm answers anyway.
“The lawsuit is finally coming to a head,” he says. “One of the nephews contesting the will is flying in from Indonesia, and the trial gets underway in Kentucky next week. I never would have started this fight, but since my charming relatives did, I’m in it to win it.”
I wish him luck.
“That’s why I want Botox,” he says. “I’m going to testify, and I want to look my confident best.”
Go, Malcolm!
While we’re talking Botox, I recall Amy, a well-traveled consultant who gave lectures all over.
“I’m curious,” I once asked her. “What do people say to you after you get Botox? Do they notice?”
“That’s interesting,” she said. “When I speak to groups, my face is projected onto large screens. That makes my wrinkles look like the Grand Canyon.
“When I started doing Botox, a man came over to me after a lecture and said, ‘I’ve heard you before, but this time you were, somehow, more cogent and compelling.’
“I thanked him, of course,” said Amy with a smile. “But the speech he was praising was the exact same speech he’d heard the first time.”
***
The rest of us try to put on the best face we can, assisted (in descending order of importance) by: makers of cosmetics, estheticians, dentists, plastic surgeons, and – oh yes – even dermatologists.
Happy to assist!
Dr. Rockoff practices dermatology in Brookline, Mass., and is a longtime contributor to Dermatology News. He serves on the clinical faculty at Tufts University, Boston, and has taught senior medical students and other trainees for 30 years. His second book, “Act Like a Doctor, Think Like a Patient,” is available at amazon.com and barnesandnoble.com. Write to him at [email protected].
Letter from the Editor: IBD drugs, ‘liquid biopsies,’ and DDW
The coming months will provide us a welcome relief from health care politics as we turn our attention to the science of medicine. Digestive Disease Week® (DDW) will occur from June 2 to 5 in Washington, DC. Major themes already are emerging and implications for our clinical practices are exciting. In this month’s issue of GI & Hepatology News, we summarize a presentation about the IBD medication pipeline given by Dr. Bill Sandborn (UCSD) at the Crohn’s & Colitis CongressTM (a partnership between the Crohn’s & Colitis Foundation and AGA, in Las Vegas). The number of medications that will enter clinical practice is impressive and so is the variety of antigen targets. Over the last several decades, we have defined multiple inflammatory pathways that can lead to IBD and developed medications that modify abnormal immune responses. We are entering an era of precision medicine never before seen in our specialty. Most of these biological medications can be given orally or subcutaneously, precluding the need for infusion centers. I anticipate an enormous offering of IBD-related science at DDW®.
The Board of Editors appreciates the feedback that many of you sent us in our latest readership survey. Each month, we try hard to collect articles of clinical interest to the wide variety of clinicians and researchers that read GI & Hepatology News. We will continue to improve our offerings based on your valuable opinions.
John I. Allen, MD, MBA, AGAF
Editor in Chief
The coming months will provide us a welcome relief from health care politics as we turn our attention to the science of medicine. Digestive Disease Week® (DDW) will occur from June 2 to 5 in Washington, DC. Major themes already are emerging and implications for our clinical practices are exciting. In this month’s issue of GI & Hepatology News, we summarize a presentation about the IBD medication pipeline given by Dr. Bill Sandborn (UCSD) at the Crohn’s & Colitis CongressTM (a partnership between the Crohn’s & Colitis Foundation and AGA, in Las Vegas). The number of medications that will enter clinical practice is impressive and so is the variety of antigen targets. Over the last several decades, we have defined multiple inflammatory pathways that can lead to IBD and developed medications that modify abnormal immune responses. We are entering an era of precision medicine never before seen in our specialty. Most of these biological medications can be given orally or subcutaneously, precluding the need for infusion centers. I anticipate an enormous offering of IBD-related science at DDW®.
The Board of Editors appreciates the feedback that many of you sent us in our latest readership survey. Each month, we try hard to collect articles of clinical interest to the wide variety of clinicians and researchers that read GI & Hepatology News. We will continue to improve our offerings based on your valuable opinions.
John I. Allen, MD, MBA, AGAF
Editor in Chief
The coming months will provide us a welcome relief from health care politics as we turn our attention to the science of medicine. Digestive Disease Week® (DDW) will occur from June 2 to 5 in Washington, DC. Major themes already are emerging and implications for our clinical practices are exciting. In this month’s issue of GI & Hepatology News, we summarize a presentation about the IBD medication pipeline given by Dr. Bill Sandborn (UCSD) at the Crohn’s & Colitis CongressTM (a partnership between the Crohn’s & Colitis Foundation and AGA, in Las Vegas). The number of medications that will enter clinical practice is impressive and so is the variety of antigen targets. Over the last several decades, we have defined multiple inflammatory pathways that can lead to IBD and developed medications that modify abnormal immune responses. We are entering an era of precision medicine never before seen in our specialty. Most of these biological medications can be given orally or subcutaneously, precluding the need for infusion centers. I anticipate an enormous offering of IBD-related science at DDW®.
The Board of Editors appreciates the feedback that many of you sent us in our latest readership survey. Each month, we try hard to collect articles of clinical interest to the wide variety of clinicians and researchers that read GI & Hepatology News. We will continue to improve our offerings based on your valuable opinions.
John I. Allen, MD, MBA, AGAF
Editor in Chief
Enhancing the effects of microfocused ultrasound with cosmeceuticals
The use of microfocused ultrasound (MFUS) emerged in dermatology in 2009 as a minimally invasive approach to treating mild to moderate facial and neck laxity. Microfocused ultrasound with visualization (MFU-V), as represented by the device Ultherapy, adds high-resolution ultrasound imaging so that the user can see the targets for MFUS energy. Visualization also allows the user to choose the appropriate treatment depth and transducer.1 Since its introduction, Ultherapy has been investigated for efficacy and safety in tightening, lifting, and wrinkle reduction beyond the face and neck, specifically including the décolletage, abdomen, arms/elbows, knees, medial thighs, and buttocks.1-8 This column will focus on using cosmeceuticals to improve the skin-tightening outcomes of microfocused ultrasound.
Two weeks before the procedure
Ingredients that should be used prior to MFU-V include retinoids, such as tretinoin and retinol. Various studies have demonstrated that pretreatment with tretinoin increases collagen production and speeds wound healing.12-14 Kligman et al. assessed wound healing after punch biopsy and found that arm wounds pretreated with tretinoin cream 0.05%-0.1% were significantly smaller by 35%-37% on days 1 and 4 and by 47%-50% on days 6, 8, and 11 than were wounds on the untreated arms.15 The majority of studies on the subject recommend a 2- to 4-week tretinoin pretreatment regimen because peak epidermal hypertrophy occurs after 7 days of tretinoin application and normalizes after 14 days of continued treatment.16,17 This time frame allows for the skin to recover from any retinoid dermatitis prior to the procedure. Adapalene should be started 5-6 weeks prior to procedures because it has a longer half-life and requires an earlier initiation period.18 Although studies have not been performed evaluating the efficacy of topical ascorbic acid and hydroxy acids, pretreating skin with them also may accelerate collagen production after procedures such as MFU-V.19,20
Postprocedure skin care
Because no incision or ablation occurs with MFUS, the use of ascorbic acid, hydroxy acids, and retinoids can and should be continued after the procedure in addition to before the procedure. The combination of topical Arnica montana and Rhododendron tomentosum in a gel pad has been shown to mitigate postoperative ecchymosis and edema after oculofacial surgery.24 Topical curcumin has been shown to facilitate wound healing in animals, but its smell and color make it difficult to use topically. I recommend adding turmeric to food pre- and postprocedure.25
Adverse events
Late in 2017, Friedmann et al. offered a report on the nature of the rare complications from MFUS, which have included mild and fleeting ecchymosis, edema, erythema, and nerve paralysis. In this retrospective multicenter case series of five patients seen in the authors’ practice who experienced serious adverse reactions to Ultherapy, the authors reported that single sessions of MFUS yielded blistering, erosion/ulceration, or cutaneous or subcutaneous tissue edema with resulting atrophy and/or cutaneous necrosis. The authors concluded that while serious adverse events following MFUS are rare, such reactions might be underreported and should be prepared for with early management to diminish inflammation.26 Other adverse events, which are transient and rare, may include discomfort and mild bruising.
Educating patients on pre- and postprocedure instructions can help minimize adverse events. Avoiding foods that decrease platelet function, like ginger, green tea, alcohol (red wine), salmon, and flax seeds, can reduce the risk of bruising. Use of topical and oral antioxidants before and after treatments also may help reduce inflammation and edema.
Conclusion
Please email me at [email protected] if you have any comments, suggestions, or anecdotal reports to share on using cosmeceuticals and nutraceuticals before and after procedures. I will share your responses on my LinkedIn account.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote two textbooks, “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002) and “Cosmeceuticals and Cosmetic Ingredients” (New York: McGraw-Hill, 2014); she also wrote a New York Times Best Sellers book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Evolus, Galderma, and Revance. She is the founder and CEO of Skin Type Solutions Franchise Systems.
References
1. Clin Cosmet Investig Dermatol. 2017 Oct 30;10:423-9.
2. J Am Acad Dermatol. 2013 Dec;69(6):965-71.
3. Dermatol Surg. 2015 Mar;41(3):327-35.
4. J Cosmet Dermatol Sci Appl. 2012;2(2A):108-16.
5. Dermatol Surg. 2015 Jul;41(7):821-6.
6. J Cosmet Laser Ther. 2014 Oct;16(5):225-9.
7. Dermatol Surg. 2012 May;38(5):754-9.
8. Dermatol Surg. 2014 Oct;40(10):1113-7.
9. Clin Cosmet Investig Dermatol. 2015; 8: 47-52.
10. Dermatol Surg. 2017 Sep 8. doi: 10.1097/DSS.0000000000001216. [Epub ahead of print]
11. Arch Dermatol. 2004 Feb;140(2):204-9.
12. Aesthetic Plast Surg. 1995 May-Jun;19(3):243-6.
13. Plast Reconstr Surg. 2011 Mar;127(3):1343-5.
14. J Am Acad Dermatol. 1998 Aug;39(2 Pt 3):S79-81.
15. Popp C et al. Br J Dermatol. 1995 Jan;132(1):46-53.
16. J Am Acad Dermatol. 2004 Dec;51(6):940-6.
17. J Korean Med Sci. 1996 Aug;11(4):335-41.
18. Eur J Dermatol. 2002 Mar-Apr;12(2):145-8.
19. Proc Natl Acad Sci U S A. 1981 May; 78(5): 2879-82.
20. Exp Dermatol. 2003;12 Suppl 2:57-63.
21. Dermatol Surg. 2001 Feb;27(2):137-42.
22. Br J Dermatol. 1992 Sep;127(3):247-53.
23. J Invest Dermatol. 1991;96:587.
24. Ophthal Plast Reconstr Surg. 2017 Jan/Feb;33(1):47-52.
25. Wound Repair Regen. 1998 Mar-Apr;6(2):167-77.
26. Lasers Surg Med. 2018 Jan;50(1):13-9.
The use of microfocused ultrasound (MFUS) emerged in dermatology in 2009 as a minimally invasive approach to treating mild to moderate facial and neck laxity. Microfocused ultrasound with visualization (MFU-V), as represented by the device Ultherapy, adds high-resolution ultrasound imaging so that the user can see the targets for MFUS energy. Visualization also allows the user to choose the appropriate treatment depth and transducer.1 Since its introduction, Ultherapy has been investigated for efficacy and safety in tightening, lifting, and wrinkle reduction beyond the face and neck, specifically including the décolletage, abdomen, arms/elbows, knees, medial thighs, and buttocks.1-8 This column will focus on using cosmeceuticals to improve the skin-tightening outcomes of microfocused ultrasound.
Two weeks before the procedure
Ingredients that should be used prior to MFU-V include retinoids, such as tretinoin and retinol. Various studies have demonstrated that pretreatment with tretinoin increases collagen production and speeds wound healing.12-14 Kligman et al. assessed wound healing after punch biopsy and found that arm wounds pretreated with tretinoin cream 0.05%-0.1% were significantly smaller by 35%-37% on days 1 and 4 and by 47%-50% on days 6, 8, and 11 than were wounds on the untreated arms.15 The majority of studies on the subject recommend a 2- to 4-week tretinoin pretreatment regimen because peak epidermal hypertrophy occurs after 7 days of tretinoin application and normalizes after 14 days of continued treatment.16,17 This time frame allows for the skin to recover from any retinoid dermatitis prior to the procedure. Adapalene should be started 5-6 weeks prior to procedures because it has a longer half-life and requires an earlier initiation period.18 Although studies have not been performed evaluating the efficacy of topical ascorbic acid and hydroxy acids, pretreating skin with them also may accelerate collagen production after procedures such as MFU-V.19,20
Postprocedure skin care
Because no incision or ablation occurs with MFUS, the use of ascorbic acid, hydroxy acids, and retinoids can and should be continued after the procedure in addition to before the procedure. The combination of topical Arnica montana and Rhododendron tomentosum in a gel pad has been shown to mitigate postoperative ecchymosis and edema after oculofacial surgery.24 Topical curcumin has been shown to facilitate wound healing in animals, but its smell and color make it difficult to use topically. I recommend adding turmeric to food pre- and postprocedure.25
Adverse events
Late in 2017, Friedmann et al. offered a report on the nature of the rare complications from MFUS, which have included mild and fleeting ecchymosis, edema, erythema, and nerve paralysis. In this retrospective multicenter case series of five patients seen in the authors’ practice who experienced serious adverse reactions to Ultherapy, the authors reported that single sessions of MFUS yielded blistering, erosion/ulceration, or cutaneous or subcutaneous tissue edema with resulting atrophy and/or cutaneous necrosis. The authors concluded that while serious adverse events following MFUS are rare, such reactions might be underreported and should be prepared for with early management to diminish inflammation.26 Other adverse events, which are transient and rare, may include discomfort and mild bruising.
Educating patients on pre- and postprocedure instructions can help minimize adverse events. Avoiding foods that decrease platelet function, like ginger, green tea, alcohol (red wine), salmon, and flax seeds, can reduce the risk of bruising. Use of topical and oral antioxidants before and after treatments also may help reduce inflammation and edema.
Conclusion
Please email me at [email protected] if you have any comments, suggestions, or anecdotal reports to share on using cosmeceuticals and nutraceuticals before and after procedures. I will share your responses on my LinkedIn account.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote two textbooks, “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002) and “Cosmeceuticals and Cosmetic Ingredients” (New York: McGraw-Hill, 2014); she also wrote a New York Times Best Sellers book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Evolus, Galderma, and Revance. She is the founder and CEO of Skin Type Solutions Franchise Systems.
References
1. Clin Cosmet Investig Dermatol. 2017 Oct 30;10:423-9.
2. J Am Acad Dermatol. 2013 Dec;69(6):965-71.
3. Dermatol Surg. 2015 Mar;41(3):327-35.
4. J Cosmet Dermatol Sci Appl. 2012;2(2A):108-16.
5. Dermatol Surg. 2015 Jul;41(7):821-6.
6. J Cosmet Laser Ther. 2014 Oct;16(5):225-9.
7. Dermatol Surg. 2012 May;38(5):754-9.
8. Dermatol Surg. 2014 Oct;40(10):1113-7.
9. Clin Cosmet Investig Dermatol. 2015; 8: 47-52.
10. Dermatol Surg. 2017 Sep 8. doi: 10.1097/DSS.0000000000001216. [Epub ahead of print]
11. Arch Dermatol. 2004 Feb;140(2):204-9.
12. Aesthetic Plast Surg. 1995 May-Jun;19(3):243-6.
13. Plast Reconstr Surg. 2011 Mar;127(3):1343-5.
14. J Am Acad Dermatol. 1998 Aug;39(2 Pt 3):S79-81.
15. Popp C et al. Br J Dermatol. 1995 Jan;132(1):46-53.
16. J Am Acad Dermatol. 2004 Dec;51(6):940-6.
17. J Korean Med Sci. 1996 Aug;11(4):335-41.
18. Eur J Dermatol. 2002 Mar-Apr;12(2):145-8.
19. Proc Natl Acad Sci U S A. 1981 May; 78(5): 2879-82.
20. Exp Dermatol. 2003;12 Suppl 2:57-63.
21. Dermatol Surg. 2001 Feb;27(2):137-42.
22. Br J Dermatol. 1992 Sep;127(3):247-53.
23. J Invest Dermatol. 1991;96:587.
24. Ophthal Plast Reconstr Surg. 2017 Jan/Feb;33(1):47-52.
25. Wound Repair Regen. 1998 Mar-Apr;6(2):167-77.
26. Lasers Surg Med. 2018 Jan;50(1):13-9.
The use of microfocused ultrasound (MFUS) emerged in dermatology in 2009 as a minimally invasive approach to treating mild to moderate facial and neck laxity. Microfocused ultrasound with visualization (MFU-V), as represented by the device Ultherapy, adds high-resolution ultrasound imaging so that the user can see the targets for MFUS energy. Visualization also allows the user to choose the appropriate treatment depth and transducer.1 Since its introduction, Ultherapy has been investigated for efficacy and safety in tightening, lifting, and wrinkle reduction beyond the face and neck, specifically including the décolletage, abdomen, arms/elbows, knees, medial thighs, and buttocks.1-8 This column will focus on using cosmeceuticals to improve the skin-tightening outcomes of microfocused ultrasound.
Two weeks before the procedure
Ingredients that should be used prior to MFU-V include retinoids, such as tretinoin and retinol. Various studies have demonstrated that pretreatment with tretinoin increases collagen production and speeds wound healing.12-14 Kligman et al. assessed wound healing after punch biopsy and found that arm wounds pretreated with tretinoin cream 0.05%-0.1% were significantly smaller by 35%-37% on days 1 and 4 and by 47%-50% on days 6, 8, and 11 than were wounds on the untreated arms.15 The majority of studies on the subject recommend a 2- to 4-week tretinoin pretreatment regimen because peak epidermal hypertrophy occurs after 7 days of tretinoin application and normalizes after 14 days of continued treatment.16,17 This time frame allows for the skin to recover from any retinoid dermatitis prior to the procedure. Adapalene should be started 5-6 weeks prior to procedures because it has a longer half-life and requires an earlier initiation period.18 Although studies have not been performed evaluating the efficacy of topical ascorbic acid and hydroxy acids, pretreating skin with them also may accelerate collagen production after procedures such as MFU-V.19,20
Postprocedure skin care
Because no incision or ablation occurs with MFUS, the use of ascorbic acid, hydroxy acids, and retinoids can and should be continued after the procedure in addition to before the procedure. The combination of topical Arnica montana and Rhododendron tomentosum in a gel pad has been shown to mitigate postoperative ecchymosis and edema after oculofacial surgery.24 Topical curcumin has been shown to facilitate wound healing in animals, but its smell and color make it difficult to use topically. I recommend adding turmeric to food pre- and postprocedure.25
Adverse events
Late in 2017, Friedmann et al. offered a report on the nature of the rare complications from MFUS, which have included mild and fleeting ecchymosis, edema, erythema, and nerve paralysis. In this retrospective multicenter case series of five patients seen in the authors’ practice who experienced serious adverse reactions to Ultherapy, the authors reported that single sessions of MFUS yielded blistering, erosion/ulceration, or cutaneous or subcutaneous tissue edema with resulting atrophy and/or cutaneous necrosis. The authors concluded that while serious adverse events following MFUS are rare, such reactions might be underreported and should be prepared for with early management to diminish inflammation.26 Other adverse events, which are transient and rare, may include discomfort and mild bruising.
Educating patients on pre- and postprocedure instructions can help minimize adverse events. Avoiding foods that decrease platelet function, like ginger, green tea, alcohol (red wine), salmon, and flax seeds, can reduce the risk of bruising. Use of topical and oral antioxidants before and after treatments also may help reduce inflammation and edema.
Conclusion
Please email me at [email protected] if you have any comments, suggestions, or anecdotal reports to share on using cosmeceuticals and nutraceuticals before and after procedures. I will share your responses on my LinkedIn account.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote two textbooks, “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002) and “Cosmeceuticals and Cosmetic Ingredients” (New York: McGraw-Hill, 2014); she also wrote a New York Times Best Sellers book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Evolus, Galderma, and Revance. She is the founder and CEO of Skin Type Solutions Franchise Systems.
References
1. Clin Cosmet Investig Dermatol. 2017 Oct 30;10:423-9.
2. J Am Acad Dermatol. 2013 Dec;69(6):965-71.
3. Dermatol Surg. 2015 Mar;41(3):327-35.
4. J Cosmet Dermatol Sci Appl. 2012;2(2A):108-16.
5. Dermatol Surg. 2015 Jul;41(7):821-6.
6. J Cosmet Laser Ther. 2014 Oct;16(5):225-9.
7. Dermatol Surg. 2012 May;38(5):754-9.
8. Dermatol Surg. 2014 Oct;40(10):1113-7.
9. Clin Cosmet Investig Dermatol. 2015; 8: 47-52.
10. Dermatol Surg. 2017 Sep 8. doi: 10.1097/DSS.0000000000001216. [Epub ahead of print]
11. Arch Dermatol. 2004 Feb;140(2):204-9.
12. Aesthetic Plast Surg. 1995 May-Jun;19(3):243-6.
13. Plast Reconstr Surg. 2011 Mar;127(3):1343-5.
14. J Am Acad Dermatol. 1998 Aug;39(2 Pt 3):S79-81.
15. Popp C et al. Br J Dermatol. 1995 Jan;132(1):46-53.
16. J Am Acad Dermatol. 2004 Dec;51(6):940-6.
17. J Korean Med Sci. 1996 Aug;11(4):335-41.
18. Eur J Dermatol. 2002 Mar-Apr;12(2):145-8.
19. Proc Natl Acad Sci U S A. 1981 May; 78(5): 2879-82.
20. Exp Dermatol. 2003;12 Suppl 2:57-63.
21. Dermatol Surg. 2001 Feb;27(2):137-42.
22. Br J Dermatol. 1992 Sep;127(3):247-53.
23. J Invest Dermatol. 1991;96:587.
24. Ophthal Plast Reconstr Surg. 2017 Jan/Feb;33(1):47-52.
25. Wound Repair Regen. 1998 Mar-Apr;6(2):167-77.
26. Lasers Surg Med. 2018 Jan;50(1):13-9.
Make the Diagnosis - March 2018
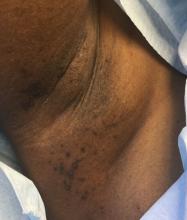
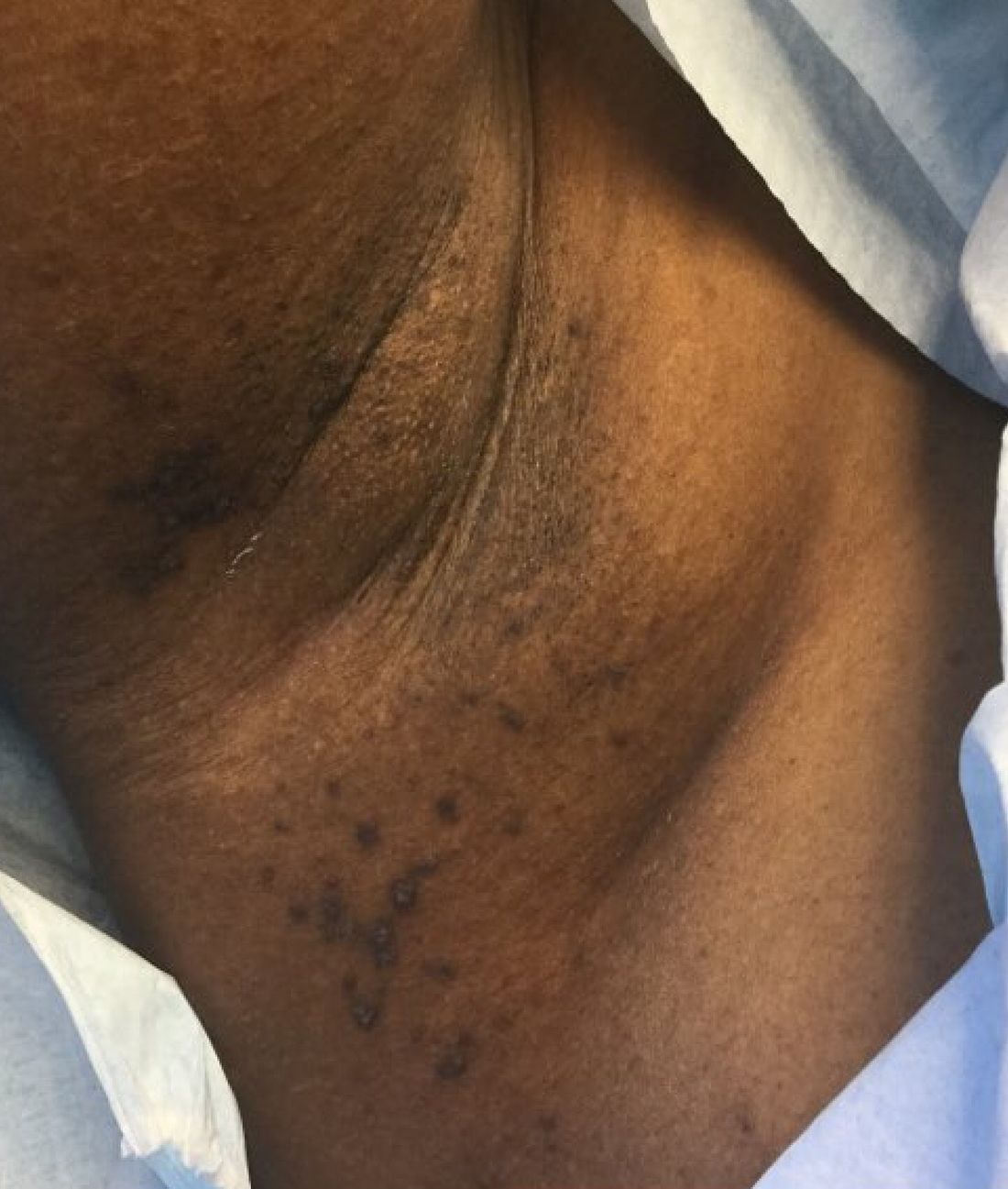
Familial benign chronic pemphigus, also known as Hailey-Hailey disease, is an uncommon autosomal dominant genetic condition. A mutation in the calcium ATPase (ATP2C1) gene on chromosome 3q21 interferes with calcium signaling and results in a loss of keratinocyte adhesion. Generally, the onset of the condition is in the second or third decade. There are two clinical subtypes of the disease: segmental type 1 and segmental type 2.
Histology reveals groups of acantholytic cells that resemble a “dilapidated brick wall.” Direct immunofluorescence is negative, unlike pemphigus vulgaris.
As hyperhidrosis is a known aggravating factor, injection with botulinum toxin (this is off-label use not yet approved by the Food and Drug Administration) in affected areas to decrease sweating has been reported to be effective.
This case and photo were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at edermatologynews.com. To submit a case for possible publication, send an email to [email protected].
Familial benign chronic pemphigus, also known as Hailey-Hailey disease, is an uncommon autosomal dominant genetic condition. A mutation in the calcium ATPase (ATP2C1) gene on chromosome 3q21 interferes with calcium signaling and results in a loss of keratinocyte adhesion. Generally, the onset of the condition is in the second or third decade. There are two clinical subtypes of the disease: segmental type 1 and segmental type 2.
Histology reveals groups of acantholytic cells that resemble a “dilapidated brick wall.” Direct immunofluorescence is negative, unlike pemphigus vulgaris.
As hyperhidrosis is a known aggravating factor, injection with botulinum toxin (this is off-label use not yet approved by the Food and Drug Administration) in affected areas to decrease sweating has been reported to be effective.
This case and photo were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at edermatologynews.com. To submit a case for possible publication, send an email to [email protected].
Familial benign chronic pemphigus, also known as Hailey-Hailey disease, is an uncommon autosomal dominant genetic condition. A mutation in the calcium ATPase (ATP2C1) gene on chromosome 3q21 interferes with calcium signaling and results in a loss of keratinocyte adhesion. Generally, the onset of the condition is in the second or third decade. There are two clinical subtypes of the disease: segmental type 1 and segmental type 2.
Histology reveals groups of acantholytic cells that resemble a “dilapidated brick wall.” Direct immunofluorescence is negative, unlike pemphigus vulgaris.
As hyperhidrosis is a known aggravating factor, injection with botulinum toxin (this is off-label use not yet approved by the Food and Drug Administration) in affected areas to decrease sweating has been reported to be effective.
This case and photo were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at edermatologynews.com. To submit a case for possible publication, send an email to [email protected].
A 39-year-old healthy black woman presented with itchy, painful lesions in the bilateral axillae and groin. The lesions have come and gone for 15 years and flare when the patient perspires. Her mother and grandmother have the same condition.
DDSEP® 8 Quick Quiz - March 2018 Question 2
Correct Answer: B
Rationale
The patient has a favorable anatomy for a surgical drainage procedure such as a lateral pancreaticojejunostomy (Peustow procedure). Surgery has been noted to provide superior pain relief over 5 years compared with endoscopy. Hospital costs and length of stay were similar between the groups. Continued medical therapy is unlikely to add further benefit on top of what she has already achieved. EUS-guided celiac plexus block will only provide temporary pain relief. There are limited long-term data on the effectiveness of total pancreatectomy with islet autotransplantation in alleviating pain.
References
1. Cahen D.L., Gouma D.J., Laramée P., et al. Gastroenterology. 2011;141(5):1690-5.
2. Conwell D.L., Lee L.S., Yadav D., et al. American pancreatic association practice guidelines in chronic pancreatitis. Pancreas. 2014;43:1143-62.
Correct Answer: B
Rationale
The patient has a favorable anatomy for a surgical drainage procedure such as a lateral pancreaticojejunostomy (Peustow procedure). Surgery has been noted to provide superior pain relief over 5 years compared with endoscopy. Hospital costs and length of stay were similar between the groups. Continued medical therapy is unlikely to add further benefit on top of what she has already achieved. EUS-guided celiac plexus block will only provide temporary pain relief. There are limited long-term data on the effectiveness of total pancreatectomy with islet autotransplantation in alleviating pain.
References
1. Cahen D.L., Gouma D.J., Laramée P., et al. Gastroenterology. 2011;141(5):1690-5.
2. Conwell D.L., Lee L.S., Yadav D., et al. American pancreatic association practice guidelines in chronic pancreatitis. Pancreas. 2014;43:1143-62.
Correct Answer: B
Rationale
The patient has a favorable anatomy for a surgical drainage procedure such as a lateral pancreaticojejunostomy (Peustow procedure). Surgery has been noted to provide superior pain relief over 5 years compared with endoscopy. Hospital costs and length of stay were similar between the groups. Continued medical therapy is unlikely to add further benefit on top of what she has already achieved. EUS-guided celiac plexus block will only provide temporary pain relief. There are limited long-term data on the effectiveness of total pancreatectomy with islet autotransplantation in alleviating pain.
References
1. Cahen D.L., Gouma D.J., Laramée P., et al. Gastroenterology. 2011;141(5):1690-5.
2. Conwell D.L., Lee L.S., Yadav D., et al. American pancreatic association practice guidelines in chronic pancreatitis. Pancreas. 2014;43:1143-62.
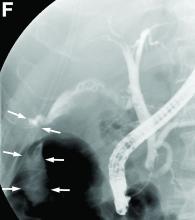
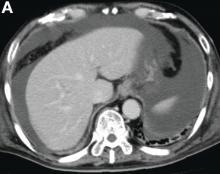
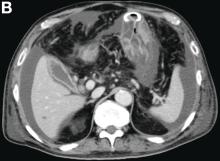
A 68-year-old woman with alcoholic chronic pancreatitis has constant, disabling pain. She has previously tried gabapentin, celecoxib, and antioxidants with partial improvement. She currently takes nonenteric coated pancrealipase (90,000 IU per meal) and controlled-release oxycontin. CT of the abdomen demonstrates a few small punctate calcifications in the head of the pancreas, a 1-cm calculus in the genu with a markedly dilated pancreatic duct in the body and tail, and moderate distal atrophy. There are no pseudocysts. She discusses further options to treat her pain.
Which intervention will most likely improve her pain and quality of life over the next 5 years?
DDSEP® 8 Quick Quiz - March 2018 Question 1
Q1. Correct answer: C
Rationale
The initial management of dyspepsia depends on symptoms and presence of any “alarm features.” In patients without “alarm features” presenting with symptoms suggestive of hepatobiliary or pancreatic causes, the initial diagnostic tests should include liver/pancreatic blood tests and abdominal imaging. For other dyspeptic patients without alarm features, initial management would include H. pylori testing (breath, stool antigen, or antibody) and/or empiric antisecretory (PPI) therapy. However, for patients who present with “alarm features” such as dysphagia, anemia, GI bleeding, anorexia, significant weight loss, etc., an upper endoscopy should be performed to evaluate for the presence of any upper GI tract malignancy. In this patient, the presence of microcytic anemia is an alarm feature. Tricyclic antidepressants such as amitriptyline may be used as treatment for functional dyspepsia, after organic causes have been ruled out.
Reference
1. Talley N.J., Vakil N.B., Moayyedi P. American Gastroenterological Association technical review on the evaluation of dyspepsia. Gastroenterology 2005;129:1756-80.
Q1. Correct answer: C
Rationale
The initial management of dyspepsia depends on symptoms and presence of any “alarm features.” In patients without “alarm features” presenting with symptoms suggestive of hepatobiliary or pancreatic causes, the initial diagnostic tests should include liver/pancreatic blood tests and abdominal imaging. For other dyspeptic patients without alarm features, initial management would include H. pylori testing (breath, stool antigen, or antibody) and/or empiric antisecretory (PPI) therapy. However, for patients who present with “alarm features” such as dysphagia, anemia, GI bleeding, anorexia, significant weight loss, etc., an upper endoscopy should be performed to evaluate for the presence of any upper GI tract malignancy. In this patient, the presence of microcytic anemia is an alarm feature. Tricyclic antidepressants such as amitriptyline may be used as treatment for functional dyspepsia, after organic causes have been ruled out.
Reference
1. Talley N.J., Vakil N.B., Moayyedi P. American Gastroenterological Association technical review on the evaluation of dyspepsia. Gastroenterology 2005;129:1756-80.
Q1. Correct answer: C
Rationale
The initial management of dyspepsia depends on symptoms and presence of any “alarm features.” In patients without “alarm features” presenting with symptoms suggestive of hepatobiliary or pancreatic causes, the initial diagnostic tests should include liver/pancreatic blood tests and abdominal imaging. For other dyspeptic patients without alarm features, initial management would include H. pylori testing (breath, stool antigen, or antibody) and/or empiric antisecretory (PPI) therapy. However, for patients who present with “alarm features” such as dysphagia, anemia, GI bleeding, anorexia, significant weight loss, etc., an upper endoscopy should be performed to evaluate for the presence of any upper GI tract malignancy. In this patient, the presence of microcytic anemia is an alarm feature. Tricyclic antidepressants such as amitriptyline may be used as treatment for functional dyspepsia, after organic causes have been ruled out.
Reference
1. Talley N.J., Vakil N.B., Moayyedi P. American Gastroenterological Association technical review on the evaluation of dyspepsia. Gastroenterology 2005;129:1756-80.
A 37-year-old man with no significant past medical history presents with a dull, nonradiating epigastric pain for 3 months. The pain is not associated with eating or positional changes. He denies any heartburn, regurgitation, chest pain, nausea, vomiting, dysphagia, odynophagia, or weight loss. He currently does not take any medications or supplements. Social and family history are not significant. Physical examination reveals minimal tenderness to deep palpation in the epigastrum, but otherwise it is unremarkable. A complete blood count reveals a white blood cell count of 6, hemoglobin 10 g/dL, MCV 72 fL, and platelet count of 200 x 103/mcL. What is the most important next step of management?
Clinical Challenges - March 2018 What's your diagnosis?
The diagnosis: Spontaneous gallbladder perforation
References
1. Ausania, F., Guzman Suarez, S., Alvarez Garcia, H. et al. Gallbladder perforation: morbidity, mortality and preoperative risk prediction. Surg Endosc. 2015;29:955-60.
2. Niemeier, O.W. Acute free perforation of the gall-bladder. Ann Surg. 1934;99:922-4.
3. Hyodo, T., Kumano, S., Kushihata, F. et al. CT and MR cholangiography: advantages and pitfalls in perioperative evaluation of biliary tree. Br J Radiol. 2012;85:887-96.
The diagnosis: Spontaneous gallbladder perforation
References
1. Ausania, F., Guzman Suarez, S., Alvarez Garcia, H. et al. Gallbladder perforation: morbidity, mortality and preoperative risk prediction. Surg Endosc. 2015;29:955-60.
2. Niemeier, O.W. Acute free perforation of the gall-bladder. Ann Surg. 1934;99:922-4.
3. Hyodo, T., Kumano, S., Kushihata, F. et al. CT and MR cholangiography: advantages and pitfalls in perioperative evaluation of biliary tree. Br J Radiol. 2012;85:887-96.
The diagnosis: Spontaneous gallbladder perforation
References
1. Ausania, F., Guzman Suarez, S., Alvarez Garcia, H. et al. Gallbladder perforation: morbidity, mortality and preoperative risk prediction. Surg Endosc. 2015;29:955-60.
2. Niemeier, O.W. Acute free perforation of the gall-bladder. Ann Surg. 1934;99:922-4.
3. Hyodo, T., Kumano, S., Kushihata, F. et al. CT and MR cholangiography: advantages and pitfalls in perioperative evaluation of biliary tree. Br J Radiol. 2012;85:887-96.
Published previously in Gastroenterology (2016;151[1]:40-2).
What is your diagnosis and treatment?
Mental health reporting laws: A false answer to gun violence
The tragic Feb. 14 massacre at Marjory Stoneman Douglas High School in Parkland, Fla., which left 17 people dead and 15 hospitalized, was the 34th1 U.S. mass shooting of 2018. Last year was a record year, with 346 mass shootings.2 In response to these tragedies, mental health providers are being looked at to help solve the problem. In fact, the day after the Florida shooting, President Trump tweeted: “So many signs that the Florida shooter was mentally disturbed ... Must always report such instances to authorities, again and again!”3
This response is reminiscent of how the state of New York reacted to the 2012 Sandy Hook massacre in Newtown, Conn. One month after the shooting, the NY SAFE (New York Secure Ammunition and Firearms Enforcement) Act was passed, which, among its measures, requires mental health providers to report individuals “likely” to engage in harm.4 The act represented an attempt to empower mental health providers to report high-risk, potentially violent individuals to the appropriate authorities.
However, we must acknowledge that some practical difficulties arise with such mandates. First is the reality that the overwhelming majority of patients in therapy who endorse violent impulses do not go on to commit harm toward themselves or others, nor do they break the law. Up to 25% of teenagers have thoughts of killing themselves6; 41% of depressed mothers have experienced thoughts of harming their children7; and up to 68% of individuals have homicidal fantasies.8 It would be difficult to argue that we would be doing our due diligence in reporting all patients who share such thoughts. Even if we agree that we ought to report only those who are at high risk for potential future violence, few guidelines are useful in stratifying risk for future violence outside of forensic settings. Prognostication is particularly difficult in cases where there is no history of previous criminal activity, as was the case with the confessed9 gunman in the Florida case. We also must acknowledge that evidence is scant suggesting that mental health treatment as a whole can reduce the incidence of mass murders. Evidence does suggest, however, that patients with serious mental illness are not involved in a majority of cases.10
Second, these types of mandates can perpetuate stigma in mental health, both potentially driving away patients who could benefit from therapy, and straining the relationships between patient and provider for those in treatment. Given the high degree of effort it takes for some populations to engage in mental health services, Indeed, it has been demonstrated many times that nothing matters more to the outcome of mental health treatment than the relationship between the providers and their patients,11 an effect that is true even for medication management.12 Being more wary of one’s treating provider may, therefore, limit the effect of treatment from the patient’s perspective. In the same vein, some therapists may rightly pause to consider whether they should be more vigilant and dutifully extract statements confirming a nonviolent mindset at every meeting, much in the way we perform suicide risk assessment to assess safety. Or, therapists could hesitate to probe for violent themes out of an underlying wish to avoid discovering something that they would have to report. Such tensions created within the therapist also may lend to suboptimal treatment.
Dr. Badre is a forensic psychiatrist in San Diego and an expert in correctional mental health. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Dr. Joshi is a research track psychiatry resident at the University of California, San Diego. His current research focuses on developing novel therapeutic strategies to target cognitive impairment in schizophrenia. His interests include graduate medical education and applied bioethics.
References
1. Gun Violence Archive http://www.gunviolencearchive.org/reports/mass-shooting
2. https://www.abc15.com/news/data/mass-shootings-in-the-us-when-where-they-have-occurred-in-2018
3. https://twitter.com/realDonaldTrump/status/964110212885106689
4. http://www.nyspsych.org/index.php?option=com_content&view=article&id=73
5. https://www.politico.com/states/new-york/albany/story/2018/02/15/after-florida-school-shooting-cuomo-again-touts-safe-act-256104
6. MMWR 2004;53(2):1-96
7. J Affect Disord. 1999 Jul;54(1-2):21-8
8. Ethol Sociobiol. 1993;14(4):231-48
9. http://www.dcf.state.fl.us/newsroom/publicdocuments/Headquarters/Records20180219/Petition%20to%20Publically%20Release%20DCF%20Records.pdf
10. Ann Rev Clin Psychol. 2017 May 8;13:445-69
11. Psychotherapy Theory Res Prac. 2001;38(4):357-61
12. J Affect Disord. 2006 Jun;92(2-3):287-90
The tragic Feb. 14 massacre at Marjory Stoneman Douglas High School in Parkland, Fla., which left 17 people dead and 15 hospitalized, was the 34th1 U.S. mass shooting of 2018. Last year was a record year, with 346 mass shootings.2 In response to these tragedies, mental health providers are being looked at to help solve the problem. In fact, the day after the Florida shooting, President Trump tweeted: “So many signs that the Florida shooter was mentally disturbed ... Must always report such instances to authorities, again and again!”3
This response is reminiscent of how the state of New York reacted to the 2012 Sandy Hook massacre in Newtown, Conn. One month after the shooting, the NY SAFE (New York Secure Ammunition and Firearms Enforcement) Act was passed, which, among its measures, requires mental health providers to report individuals “likely” to engage in harm.4 The act represented an attempt to empower mental health providers to report high-risk, potentially violent individuals to the appropriate authorities.
However, we must acknowledge that some practical difficulties arise with such mandates. First is the reality that the overwhelming majority of patients in therapy who endorse violent impulses do not go on to commit harm toward themselves or others, nor do they break the law. Up to 25% of teenagers have thoughts of killing themselves6; 41% of depressed mothers have experienced thoughts of harming their children7; and up to 68% of individuals have homicidal fantasies.8 It would be difficult to argue that we would be doing our due diligence in reporting all patients who share such thoughts. Even if we agree that we ought to report only those who are at high risk for potential future violence, few guidelines are useful in stratifying risk for future violence outside of forensic settings. Prognostication is particularly difficult in cases where there is no history of previous criminal activity, as was the case with the confessed9 gunman in the Florida case. We also must acknowledge that evidence is scant suggesting that mental health treatment as a whole can reduce the incidence of mass murders. Evidence does suggest, however, that patients with serious mental illness are not involved in a majority of cases.10
Second, these types of mandates can perpetuate stigma in mental health, both potentially driving away patients who could benefit from therapy, and straining the relationships between patient and provider for those in treatment. Given the high degree of effort it takes for some populations to engage in mental health services, Indeed, it has been demonstrated many times that nothing matters more to the outcome of mental health treatment than the relationship between the providers and their patients,11 an effect that is true even for medication management.12 Being more wary of one’s treating provider may, therefore, limit the effect of treatment from the patient’s perspective. In the same vein, some therapists may rightly pause to consider whether they should be more vigilant and dutifully extract statements confirming a nonviolent mindset at every meeting, much in the way we perform suicide risk assessment to assess safety. Or, therapists could hesitate to probe for violent themes out of an underlying wish to avoid discovering something that they would have to report. Such tensions created within the therapist also may lend to suboptimal treatment.
Dr. Badre is a forensic psychiatrist in San Diego and an expert in correctional mental health. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Dr. Joshi is a research track psychiatry resident at the University of California, San Diego. His current research focuses on developing novel therapeutic strategies to target cognitive impairment in schizophrenia. His interests include graduate medical education and applied bioethics.
References
1. Gun Violence Archive http://www.gunviolencearchive.org/reports/mass-shooting
2. https://www.abc15.com/news/data/mass-shootings-in-the-us-when-where-they-have-occurred-in-2018
3. https://twitter.com/realDonaldTrump/status/964110212885106689
4. http://www.nyspsych.org/index.php?option=com_content&view=article&id=73
5. https://www.politico.com/states/new-york/albany/story/2018/02/15/after-florida-school-shooting-cuomo-again-touts-safe-act-256104
6. MMWR 2004;53(2):1-96
7. J Affect Disord. 1999 Jul;54(1-2):21-8
8. Ethol Sociobiol. 1993;14(4):231-48
9. http://www.dcf.state.fl.us/newsroom/publicdocuments/Headquarters/Records20180219/Petition%20to%20Publically%20Release%20DCF%20Records.pdf
10. Ann Rev Clin Psychol. 2017 May 8;13:445-69
11. Psychotherapy Theory Res Prac. 2001;38(4):357-61
12. J Affect Disord. 2006 Jun;92(2-3):287-90
The tragic Feb. 14 massacre at Marjory Stoneman Douglas High School in Parkland, Fla., which left 17 people dead and 15 hospitalized, was the 34th1 U.S. mass shooting of 2018. Last year was a record year, with 346 mass shootings.2 In response to these tragedies, mental health providers are being looked at to help solve the problem. In fact, the day after the Florida shooting, President Trump tweeted: “So many signs that the Florida shooter was mentally disturbed ... Must always report such instances to authorities, again and again!”3
This response is reminiscent of how the state of New York reacted to the 2012 Sandy Hook massacre in Newtown, Conn. One month after the shooting, the NY SAFE (New York Secure Ammunition and Firearms Enforcement) Act was passed, which, among its measures, requires mental health providers to report individuals “likely” to engage in harm.4 The act represented an attempt to empower mental health providers to report high-risk, potentially violent individuals to the appropriate authorities.
However, we must acknowledge that some practical difficulties arise with such mandates. First is the reality that the overwhelming majority of patients in therapy who endorse violent impulses do not go on to commit harm toward themselves or others, nor do they break the law. Up to 25% of teenagers have thoughts of killing themselves6; 41% of depressed mothers have experienced thoughts of harming their children7; and up to 68% of individuals have homicidal fantasies.8 It would be difficult to argue that we would be doing our due diligence in reporting all patients who share such thoughts. Even if we agree that we ought to report only those who are at high risk for potential future violence, few guidelines are useful in stratifying risk for future violence outside of forensic settings. Prognostication is particularly difficult in cases where there is no history of previous criminal activity, as was the case with the confessed9 gunman in the Florida case. We also must acknowledge that evidence is scant suggesting that mental health treatment as a whole can reduce the incidence of mass murders. Evidence does suggest, however, that patients with serious mental illness are not involved in a majority of cases.10
Second, these types of mandates can perpetuate stigma in mental health, both potentially driving away patients who could benefit from therapy, and straining the relationships between patient and provider for those in treatment. Given the high degree of effort it takes for some populations to engage in mental health services, Indeed, it has been demonstrated many times that nothing matters more to the outcome of mental health treatment than the relationship between the providers and their patients,11 an effect that is true even for medication management.12 Being more wary of one’s treating provider may, therefore, limit the effect of treatment from the patient’s perspective. In the same vein, some therapists may rightly pause to consider whether they should be more vigilant and dutifully extract statements confirming a nonviolent mindset at every meeting, much in the way we perform suicide risk assessment to assess safety. Or, therapists could hesitate to probe for violent themes out of an underlying wish to avoid discovering something that they would have to report. Such tensions created within the therapist also may lend to suboptimal treatment.
Dr. Badre is a forensic psychiatrist in San Diego and an expert in correctional mental health. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Dr. Joshi is a research track psychiatry resident at the University of California, San Diego. His current research focuses on developing novel therapeutic strategies to target cognitive impairment in schizophrenia. His interests include graduate medical education and applied bioethics.
References
1. Gun Violence Archive http://www.gunviolencearchive.org/reports/mass-shooting
2. https://www.abc15.com/news/data/mass-shootings-in-the-us-when-where-they-have-occurred-in-2018
3. https://twitter.com/realDonaldTrump/status/964110212885106689
4. http://www.nyspsych.org/index.php?option=com_content&view=article&id=73
5. https://www.politico.com/states/new-york/albany/story/2018/02/15/after-florida-school-shooting-cuomo-again-touts-safe-act-256104
6. MMWR 2004;53(2):1-96
7. J Affect Disord. 1999 Jul;54(1-2):21-8
8. Ethol Sociobiol. 1993;14(4):231-48
9. http://www.dcf.state.fl.us/newsroom/publicdocuments/Headquarters/Records20180219/Petition%20to%20Publically%20Release%20DCF%20Records.pdf
10. Ann Rev Clin Psychol. 2017 May 8;13:445-69
11. Psychotherapy Theory Res Prac. 2001;38(4):357-61
12. J Affect Disord. 2006 Jun;92(2-3):287-90