User login
Inclusivity needed in PHM fellowships
A year and a half ago, I found myself seated in a crowded hall at the national Pediatric Hospital Medicine (PHM) conference. Throughout the conference, trainees like me were warmly welcomed into small groups and lunch tables. I tried to keep my cool while PHM “celebrities” chatted with me in the elevator. Most sessions were prepared with plenty of chairs, and those that were not encouraged latecomers to grab a spot on the floor or the back wall – the more the merrier.
The intention of this “advice for applicants” meeting was to inspire and guide our next steps toward fellowship, but a discomforting reality loomed over us. It was the first year graduating pediatricians could not choose PHM board certification via the practice pathway – we needed an invitation in the form of a fellowship match.
The “hidden curriculum” was not subtle: People who scored a seat would keep their options open within the field of PHM, and those who did not had a murkier future. This message stood in stark contrast to the PHM inclusivity I had experienced all conference, and planted seeds of doubt: Was I welcome here? Did I “deserve” a seat?
I found the experience as a PHM fellowship applicant to be uncomfortable, and my all-too familiar friend “imposter syndrome” set up camp in my brain and made herself at home. I had no way of knowing how many programs to apply to, how many to interview at, or the chances of my matching at all. Once on the interview trail, I realized I was not alone in my discomfort – most applicants harbored some trepidation, and no one truly knew how the chips would fall on Match Day.
I am thrilled and relieved to have come out the other end in a great position. The team I work with and learn from is phenomenal. I am grateful that ACGME accreditation ensures structures are in place for fellows to be supported in their academic and educational efforts and have full confidence that the skills I gain in fellowship will help me contribute to progression of the field of PHM and improve my performance as a clinician-educator.
Sadly, each year PHM match day celebrations are dampened by the knowledge that a large portion of our colleagues are being left out in the cold with an “unmatched” notification in their inboxes. Approximately 200 graduating pediatricians become pediatric hospitalists each year,1 but only 68 fellowship positions were available in the United States for matriculation in 2020.2 In 2019, PHM fellowship candidates navigated the 6-month application journey with aspirations to further their training in the profession they love. Of the candidates who submitted a rank list committing to 2 or more years in PHM fellowship, 35% were denied.
Unfortunately, despite expansion of PHM fellowship programs and fifteen seats added from last year, we learned this December that there still are not enough positions to welcome qualified applicants with open arms: Thirty-three percent of candidates ranked PHM programs first in the NRMP but did not match – the highest unmatched percentage out of all pediatric subspecialties.3
The NRMP report shared a glimpse of our colleagues who received interview invitations and submitted a rank list, but this is likely an underestimation of pediatric graduates who wanted to obtain PHM board certification and wound up on a different path. Some residents anticipated the stiff competition and delayed their plans to apply for fellowship, while others matched into another subspecialty that was able to accommodate them. Many pediatric graduates joined the workforce directly as pediatric hospitalists knowing the practice pathway to certification is not available to them. Along with other physicians without board certification in PHM, they shoulder concerns of being withheld from professional advancement opportunities.
For the foreseeable future it is clear that pediatric hospitalists without board certification will be a large part of our community, and are crucial to providing high-quality care to hospitalized children nationally. In 2019, a national survey of pediatric hospital medicine groups revealed that 50% of pediatric hospitalist hires came directly out of residency, and only 8% of hires were fellowship trained.4 The same report revealed that 26% of physicians were board-certified.These percentages are likely to change over the next 5 years as the window of practice pathway certification closes and fellowship programs continue to expand. Only time will tell what the national prevalence of board-certified pediatric hospitalists settles out to be.
Historically, PHM fellowship graduates have assumed roles that include teaching and research responsibilities, and ACGME fellowship requirements have ensured that trainees graduate with skills in medical education and scholarship, and need only 4 weeks of training to be done in a community hospital.5 Pediatric hospitalists who do not pursue board certification are seeing the growing pool of PHM fellowship graduates prepared for positions in academic institutions. It is reasonable that they harbor concerns about being siloed toward primarily community hospital roles, and for community hospitalists to feel that this structure undervalues their role within the field of PHM.
At a time when inclusivity and community in medicine are receiving much-needed recognition, the current fellowship application climate has potential to create division within the PHM community. Newly graduating pediatric residents are among the populations disproportionately affected by the practice pathway cutoff. Like other subspecialties with ever-climbing steps up the “ivory tower” of academia and specialization within medicine, the inherent structure of the training pathway makes navigating it more difficult for pediatricians with professional, geographic, and economic diversity or constraints.
Med-Peds–trained colleagues have the added challenge of finding a fellowship position that is willing and able to support their concurrent internal medicine goals. International medical graduates make up about 20% of graduating residents each year, and just 11% of matched PHM fellows.3,6 Similarly, while DO medical graduates make up 20% of pediatric residents in the United States, only 10% of matched PHM fellows were DOs.3,6 New pediatricians with families or financial insecurity may be unable to invest in an expensive application process, move to a new city, and accept less than half of the average starting salary of a pediatric hospitalist for 2-3 years.7
The prevalence of implicit bias in medicine is well documented, and there is growing evidence that it negatively impacts candidate selection in medical education and contributes to minorities being underrepresented in the physician workforce.8 We must recognize the ways that adding a competitive costly hurdle may risk conflict with our mission to encourage diversity of representation within PHM leadership positions.
We have not yet successfully bridged the gap between qualified PHM fellowship candidates and available fellowship positions. I worry that this gap and the lack of transparency surrounding it is resulting in one portion of new pediatricians being welcomed by the subspecialty, and others feeling unsupported and alienated by the larger PHM community as early career physicians.
Right now, the only solution available is expansion of fellowship programs. We see progress with the new addition of fellowship positions every year, but finding funding for each position is often a lengthy endeavor, and the COVID-19 pandemic has tightened the purse strings of many children’s hospitals. It may be many years before the number of available fellowship positions more closely approximates the 200 pediatricians that become hospitalists each year.
The most equitable solution would be offering other avenues to board certification while this gap is being bridged, either by extending the practice pathway option, or making a third pathway that requires less institutional funding per fellow, but still incentivizes institutional investment in fellowship positions and resources (e.g., a pathway requiring some number of years in practice, plus 1 year in fellowship centered around a nonclinical academic curriculum).
In the absence of the solutions above, we collectively hold the responsibility of maintaining inclusivity and support of our PHM colleagues with and without board certification. One important strategy provided by Dr. Gregory Welsh9 is to incorporate community hospital medicine rotations into residency training. Sharing this side of PHM with residents may help some graduates avoid a training pathway they may not want or need. More importantly, it would raise trainee exposure and interest toward a service that is both expansive – approximately 70% of pediatric hospitalists practice in a community hospital – and crucial to children’s health nationally.
Pediatric hospitalists who are not eligible for board certification are vital and valued members of the PHM community, and as such need to maintain representation within PHM leadership. Professional development opportunities need to remain accessible outside of fellowship. The blossoming of virtual conferences and Zoom meet-ups in the face of the COVID-19 pandemic have shown us that with innovation (and a good Internet connection), networking and mentorship can be accomplished across thousands of miles.
While there’s great diversity within PHM, this subspecialty has a history of attracting pediatricians with some common core qualities: Grit, creativity, and the belief that a strong team is far greater than the sum of its parts. I have confidence that if we approach this PHM transition period with transparency about our goals and challenges, this community can emerge from it strong and united.
Dr. Ezzio is a first-year pediatric hospital medicine fellow at Helen DeVos Children’s Hospital in Grand Rapids, Mich. Her interests include medical education and advocacy. Dr. Ezzio would like to thank Dr. Jeri Kessenich and Dr. Rachel “Danielle” Fisher for their assistance in revising the article. To submit to, or for inquiries about, our PHM Fellows Column, please contact our Pediatrics Editor, Dr. Anika Kumar ([email protected]).
References
1. Leyenaar JK and Fritner MP. Graduating pediatric residents entering the hospital medicine workforce, 2006-2015. Acad Pediatr. 2018 Mar;18(2):200-7.
2. National Resident Matching Program. Results and data: Specialties matching service 2020 appointment year. Washington, DC 2020.
3. National Resident Matching Program. Results and data: Specialties matching service 2021 appointment year. Washington, DC 2021.
4. 2020 State of Hospital Medicine report. Society of Hospital Medicine. 2020.
5. Oshimura JM et al. Current roles and perceived needs of pediatric hospital medicine fellowship graduates. Hosp Pediatr. 2016;6(10):633-7.
6. National Resident Matching Program. Results and data: 2020 main residency match. Washington, DC 2020.
7. American Academy of Pediatrics Annual Survey of graduating residents 2003-2020.
8. Quinn Capers IV. How clinicians and educators can mitigate implicit bias in patient care and candidate selection in medical education. American Thoracic Society Scholar. 2020 Jun;1(3):211-17.
9. Welsh G. The importance of community pediatric hospital medicine. The Hospitalist. 2021 Jan;25(1):27.
A year and a half ago, I found myself seated in a crowded hall at the national Pediatric Hospital Medicine (PHM) conference. Throughout the conference, trainees like me were warmly welcomed into small groups and lunch tables. I tried to keep my cool while PHM “celebrities” chatted with me in the elevator. Most sessions were prepared with plenty of chairs, and those that were not encouraged latecomers to grab a spot on the floor or the back wall – the more the merrier.
The intention of this “advice for applicants” meeting was to inspire and guide our next steps toward fellowship, but a discomforting reality loomed over us. It was the first year graduating pediatricians could not choose PHM board certification via the practice pathway – we needed an invitation in the form of a fellowship match.
The “hidden curriculum” was not subtle: People who scored a seat would keep their options open within the field of PHM, and those who did not had a murkier future. This message stood in stark contrast to the PHM inclusivity I had experienced all conference, and planted seeds of doubt: Was I welcome here? Did I “deserve” a seat?
I found the experience as a PHM fellowship applicant to be uncomfortable, and my all-too familiar friend “imposter syndrome” set up camp in my brain and made herself at home. I had no way of knowing how many programs to apply to, how many to interview at, or the chances of my matching at all. Once on the interview trail, I realized I was not alone in my discomfort – most applicants harbored some trepidation, and no one truly knew how the chips would fall on Match Day.
I am thrilled and relieved to have come out the other end in a great position. The team I work with and learn from is phenomenal. I am grateful that ACGME accreditation ensures structures are in place for fellows to be supported in their academic and educational efforts and have full confidence that the skills I gain in fellowship will help me contribute to progression of the field of PHM and improve my performance as a clinician-educator.
Sadly, each year PHM match day celebrations are dampened by the knowledge that a large portion of our colleagues are being left out in the cold with an “unmatched” notification in their inboxes. Approximately 200 graduating pediatricians become pediatric hospitalists each year,1 but only 68 fellowship positions were available in the United States for matriculation in 2020.2 In 2019, PHM fellowship candidates navigated the 6-month application journey with aspirations to further their training in the profession they love. Of the candidates who submitted a rank list committing to 2 or more years in PHM fellowship, 35% were denied.
Unfortunately, despite expansion of PHM fellowship programs and fifteen seats added from last year, we learned this December that there still are not enough positions to welcome qualified applicants with open arms: Thirty-three percent of candidates ranked PHM programs first in the NRMP but did not match – the highest unmatched percentage out of all pediatric subspecialties.3
The NRMP report shared a glimpse of our colleagues who received interview invitations and submitted a rank list, but this is likely an underestimation of pediatric graduates who wanted to obtain PHM board certification and wound up on a different path. Some residents anticipated the stiff competition and delayed their plans to apply for fellowship, while others matched into another subspecialty that was able to accommodate them. Many pediatric graduates joined the workforce directly as pediatric hospitalists knowing the practice pathway to certification is not available to them. Along with other physicians without board certification in PHM, they shoulder concerns of being withheld from professional advancement opportunities.
For the foreseeable future it is clear that pediatric hospitalists without board certification will be a large part of our community, and are crucial to providing high-quality care to hospitalized children nationally. In 2019, a national survey of pediatric hospital medicine groups revealed that 50% of pediatric hospitalist hires came directly out of residency, and only 8% of hires were fellowship trained.4 The same report revealed that 26% of physicians were board-certified.These percentages are likely to change over the next 5 years as the window of practice pathway certification closes and fellowship programs continue to expand. Only time will tell what the national prevalence of board-certified pediatric hospitalists settles out to be.
Historically, PHM fellowship graduates have assumed roles that include teaching and research responsibilities, and ACGME fellowship requirements have ensured that trainees graduate with skills in medical education and scholarship, and need only 4 weeks of training to be done in a community hospital.5 Pediatric hospitalists who do not pursue board certification are seeing the growing pool of PHM fellowship graduates prepared for positions in academic institutions. It is reasonable that they harbor concerns about being siloed toward primarily community hospital roles, and for community hospitalists to feel that this structure undervalues their role within the field of PHM.
At a time when inclusivity and community in medicine are receiving much-needed recognition, the current fellowship application climate has potential to create division within the PHM community. Newly graduating pediatric residents are among the populations disproportionately affected by the practice pathway cutoff. Like other subspecialties with ever-climbing steps up the “ivory tower” of academia and specialization within medicine, the inherent structure of the training pathway makes navigating it more difficult for pediatricians with professional, geographic, and economic diversity or constraints.
Med-Peds–trained colleagues have the added challenge of finding a fellowship position that is willing and able to support their concurrent internal medicine goals. International medical graduates make up about 20% of graduating residents each year, and just 11% of matched PHM fellows.3,6 Similarly, while DO medical graduates make up 20% of pediatric residents in the United States, only 10% of matched PHM fellows were DOs.3,6 New pediatricians with families or financial insecurity may be unable to invest in an expensive application process, move to a new city, and accept less than half of the average starting salary of a pediatric hospitalist for 2-3 years.7
The prevalence of implicit bias in medicine is well documented, and there is growing evidence that it negatively impacts candidate selection in medical education and contributes to minorities being underrepresented in the physician workforce.8 We must recognize the ways that adding a competitive costly hurdle may risk conflict with our mission to encourage diversity of representation within PHM leadership positions.
We have not yet successfully bridged the gap between qualified PHM fellowship candidates and available fellowship positions. I worry that this gap and the lack of transparency surrounding it is resulting in one portion of new pediatricians being welcomed by the subspecialty, and others feeling unsupported and alienated by the larger PHM community as early career physicians.
Right now, the only solution available is expansion of fellowship programs. We see progress with the new addition of fellowship positions every year, but finding funding for each position is often a lengthy endeavor, and the COVID-19 pandemic has tightened the purse strings of many children’s hospitals. It may be many years before the number of available fellowship positions more closely approximates the 200 pediatricians that become hospitalists each year.
The most equitable solution would be offering other avenues to board certification while this gap is being bridged, either by extending the practice pathway option, or making a third pathway that requires less institutional funding per fellow, but still incentivizes institutional investment in fellowship positions and resources (e.g., a pathway requiring some number of years in practice, plus 1 year in fellowship centered around a nonclinical academic curriculum).
In the absence of the solutions above, we collectively hold the responsibility of maintaining inclusivity and support of our PHM colleagues with and without board certification. One important strategy provided by Dr. Gregory Welsh9 is to incorporate community hospital medicine rotations into residency training. Sharing this side of PHM with residents may help some graduates avoid a training pathway they may not want or need. More importantly, it would raise trainee exposure and interest toward a service that is both expansive – approximately 70% of pediatric hospitalists practice in a community hospital – and crucial to children’s health nationally.
Pediatric hospitalists who are not eligible for board certification are vital and valued members of the PHM community, and as such need to maintain representation within PHM leadership. Professional development opportunities need to remain accessible outside of fellowship. The blossoming of virtual conferences and Zoom meet-ups in the face of the COVID-19 pandemic have shown us that with innovation (and a good Internet connection), networking and mentorship can be accomplished across thousands of miles.
While there’s great diversity within PHM, this subspecialty has a history of attracting pediatricians with some common core qualities: Grit, creativity, and the belief that a strong team is far greater than the sum of its parts. I have confidence that if we approach this PHM transition period with transparency about our goals and challenges, this community can emerge from it strong and united.
Dr. Ezzio is a first-year pediatric hospital medicine fellow at Helen DeVos Children’s Hospital in Grand Rapids, Mich. Her interests include medical education and advocacy. Dr. Ezzio would like to thank Dr. Jeri Kessenich and Dr. Rachel “Danielle” Fisher for their assistance in revising the article. To submit to, or for inquiries about, our PHM Fellows Column, please contact our Pediatrics Editor, Dr. Anika Kumar ([email protected]).
References
1. Leyenaar JK and Fritner MP. Graduating pediatric residents entering the hospital medicine workforce, 2006-2015. Acad Pediatr. 2018 Mar;18(2):200-7.
2. National Resident Matching Program. Results and data: Specialties matching service 2020 appointment year. Washington, DC 2020.
3. National Resident Matching Program. Results and data: Specialties matching service 2021 appointment year. Washington, DC 2021.
4. 2020 State of Hospital Medicine report. Society of Hospital Medicine. 2020.
5. Oshimura JM et al. Current roles and perceived needs of pediatric hospital medicine fellowship graduates. Hosp Pediatr. 2016;6(10):633-7.
6. National Resident Matching Program. Results and data: 2020 main residency match. Washington, DC 2020.
7. American Academy of Pediatrics Annual Survey of graduating residents 2003-2020.
8. Quinn Capers IV. How clinicians and educators can mitigate implicit bias in patient care and candidate selection in medical education. American Thoracic Society Scholar. 2020 Jun;1(3):211-17.
9. Welsh G. The importance of community pediatric hospital medicine. The Hospitalist. 2021 Jan;25(1):27.
A year and a half ago, I found myself seated in a crowded hall at the national Pediatric Hospital Medicine (PHM) conference. Throughout the conference, trainees like me were warmly welcomed into small groups and lunch tables. I tried to keep my cool while PHM “celebrities” chatted with me in the elevator. Most sessions were prepared with plenty of chairs, and those that were not encouraged latecomers to grab a spot on the floor or the back wall – the more the merrier.
The intention of this “advice for applicants” meeting was to inspire and guide our next steps toward fellowship, but a discomforting reality loomed over us. It was the first year graduating pediatricians could not choose PHM board certification via the practice pathway – we needed an invitation in the form of a fellowship match.
The “hidden curriculum” was not subtle: People who scored a seat would keep their options open within the field of PHM, and those who did not had a murkier future. This message stood in stark contrast to the PHM inclusivity I had experienced all conference, and planted seeds of doubt: Was I welcome here? Did I “deserve” a seat?
I found the experience as a PHM fellowship applicant to be uncomfortable, and my all-too familiar friend “imposter syndrome” set up camp in my brain and made herself at home. I had no way of knowing how many programs to apply to, how many to interview at, or the chances of my matching at all. Once on the interview trail, I realized I was not alone in my discomfort – most applicants harbored some trepidation, and no one truly knew how the chips would fall on Match Day.
I am thrilled and relieved to have come out the other end in a great position. The team I work with and learn from is phenomenal. I am grateful that ACGME accreditation ensures structures are in place for fellows to be supported in their academic and educational efforts and have full confidence that the skills I gain in fellowship will help me contribute to progression of the field of PHM and improve my performance as a clinician-educator.
Sadly, each year PHM match day celebrations are dampened by the knowledge that a large portion of our colleagues are being left out in the cold with an “unmatched” notification in their inboxes. Approximately 200 graduating pediatricians become pediatric hospitalists each year,1 but only 68 fellowship positions were available in the United States for matriculation in 2020.2 In 2019, PHM fellowship candidates navigated the 6-month application journey with aspirations to further their training in the profession they love. Of the candidates who submitted a rank list committing to 2 or more years in PHM fellowship, 35% were denied.
Unfortunately, despite expansion of PHM fellowship programs and fifteen seats added from last year, we learned this December that there still are not enough positions to welcome qualified applicants with open arms: Thirty-three percent of candidates ranked PHM programs first in the NRMP but did not match – the highest unmatched percentage out of all pediatric subspecialties.3
The NRMP report shared a glimpse of our colleagues who received interview invitations and submitted a rank list, but this is likely an underestimation of pediatric graduates who wanted to obtain PHM board certification and wound up on a different path. Some residents anticipated the stiff competition and delayed their plans to apply for fellowship, while others matched into another subspecialty that was able to accommodate them. Many pediatric graduates joined the workforce directly as pediatric hospitalists knowing the practice pathway to certification is not available to them. Along with other physicians without board certification in PHM, they shoulder concerns of being withheld from professional advancement opportunities.
For the foreseeable future it is clear that pediatric hospitalists without board certification will be a large part of our community, and are crucial to providing high-quality care to hospitalized children nationally. In 2019, a national survey of pediatric hospital medicine groups revealed that 50% of pediatric hospitalist hires came directly out of residency, and only 8% of hires were fellowship trained.4 The same report revealed that 26% of physicians were board-certified.These percentages are likely to change over the next 5 years as the window of practice pathway certification closes and fellowship programs continue to expand. Only time will tell what the national prevalence of board-certified pediatric hospitalists settles out to be.
Historically, PHM fellowship graduates have assumed roles that include teaching and research responsibilities, and ACGME fellowship requirements have ensured that trainees graduate with skills in medical education and scholarship, and need only 4 weeks of training to be done in a community hospital.5 Pediatric hospitalists who do not pursue board certification are seeing the growing pool of PHM fellowship graduates prepared for positions in academic institutions. It is reasonable that they harbor concerns about being siloed toward primarily community hospital roles, and for community hospitalists to feel that this structure undervalues their role within the field of PHM.
At a time when inclusivity and community in medicine are receiving much-needed recognition, the current fellowship application climate has potential to create division within the PHM community. Newly graduating pediatric residents are among the populations disproportionately affected by the practice pathway cutoff. Like other subspecialties with ever-climbing steps up the “ivory tower” of academia and specialization within medicine, the inherent structure of the training pathway makes navigating it more difficult for pediatricians with professional, geographic, and economic diversity or constraints.
Med-Peds–trained colleagues have the added challenge of finding a fellowship position that is willing and able to support their concurrent internal medicine goals. International medical graduates make up about 20% of graduating residents each year, and just 11% of matched PHM fellows.3,6 Similarly, while DO medical graduates make up 20% of pediatric residents in the United States, only 10% of matched PHM fellows were DOs.3,6 New pediatricians with families or financial insecurity may be unable to invest in an expensive application process, move to a new city, and accept less than half of the average starting salary of a pediatric hospitalist for 2-3 years.7
The prevalence of implicit bias in medicine is well documented, and there is growing evidence that it negatively impacts candidate selection in medical education and contributes to minorities being underrepresented in the physician workforce.8 We must recognize the ways that adding a competitive costly hurdle may risk conflict with our mission to encourage diversity of representation within PHM leadership positions.
We have not yet successfully bridged the gap between qualified PHM fellowship candidates and available fellowship positions. I worry that this gap and the lack of transparency surrounding it is resulting in one portion of new pediatricians being welcomed by the subspecialty, and others feeling unsupported and alienated by the larger PHM community as early career physicians.
Right now, the only solution available is expansion of fellowship programs. We see progress with the new addition of fellowship positions every year, but finding funding for each position is often a lengthy endeavor, and the COVID-19 pandemic has tightened the purse strings of many children’s hospitals. It may be many years before the number of available fellowship positions more closely approximates the 200 pediatricians that become hospitalists each year.
The most equitable solution would be offering other avenues to board certification while this gap is being bridged, either by extending the practice pathway option, or making a third pathway that requires less institutional funding per fellow, but still incentivizes institutional investment in fellowship positions and resources (e.g., a pathway requiring some number of years in practice, plus 1 year in fellowship centered around a nonclinical academic curriculum).
In the absence of the solutions above, we collectively hold the responsibility of maintaining inclusivity and support of our PHM colleagues with and without board certification. One important strategy provided by Dr. Gregory Welsh9 is to incorporate community hospital medicine rotations into residency training. Sharing this side of PHM with residents may help some graduates avoid a training pathway they may not want or need. More importantly, it would raise trainee exposure and interest toward a service that is both expansive – approximately 70% of pediatric hospitalists practice in a community hospital – and crucial to children’s health nationally.
Pediatric hospitalists who are not eligible for board certification are vital and valued members of the PHM community, and as such need to maintain representation within PHM leadership. Professional development opportunities need to remain accessible outside of fellowship. The blossoming of virtual conferences and Zoom meet-ups in the face of the COVID-19 pandemic have shown us that with innovation (and a good Internet connection), networking and mentorship can be accomplished across thousands of miles.
While there’s great diversity within PHM, this subspecialty has a history of attracting pediatricians with some common core qualities: Grit, creativity, and the belief that a strong team is far greater than the sum of its parts. I have confidence that if we approach this PHM transition period with transparency about our goals and challenges, this community can emerge from it strong and united.
Dr. Ezzio is a first-year pediatric hospital medicine fellow at Helen DeVos Children’s Hospital in Grand Rapids, Mich. Her interests include medical education and advocacy. Dr. Ezzio would like to thank Dr. Jeri Kessenich and Dr. Rachel “Danielle” Fisher for their assistance in revising the article. To submit to, or for inquiries about, our PHM Fellows Column, please contact our Pediatrics Editor, Dr. Anika Kumar ([email protected]).
References
1. Leyenaar JK and Fritner MP. Graduating pediatric residents entering the hospital medicine workforce, 2006-2015. Acad Pediatr. 2018 Mar;18(2):200-7.
2. National Resident Matching Program. Results and data: Specialties matching service 2020 appointment year. Washington, DC 2020.
3. National Resident Matching Program. Results and data: Specialties matching service 2021 appointment year. Washington, DC 2021.
4. 2020 State of Hospital Medicine report. Society of Hospital Medicine. 2020.
5. Oshimura JM et al. Current roles and perceived needs of pediatric hospital medicine fellowship graduates. Hosp Pediatr. 2016;6(10):633-7.
6. National Resident Matching Program. Results and data: 2020 main residency match. Washington, DC 2020.
7. American Academy of Pediatrics Annual Survey of graduating residents 2003-2020.
8. Quinn Capers IV. How clinicians and educators can mitigate implicit bias in patient care and candidate selection in medical education. American Thoracic Society Scholar. 2020 Jun;1(3):211-17.
9. Welsh G. The importance of community pediatric hospital medicine. The Hospitalist. 2021 Jan;25(1):27.
I am the best. Sometimes.
The fifth and last time I was listed as Best of Boston was in 2019, when I shared honors with obstetrics, ice cream, interior design, and kitchenware.
My first time on that list was 10 years earlier, and came as a surprise. Though the magazine that runs the feature said that selections are “peer-generated,” I was never asked to evaluate any colleagues, so I don’t know who my admiring peers were or what they admired.
Three years later I was dropped from the list, for equally mysterious reasons. Maybe my acne patients did worse that year. Be that as it may, I was reinstated several years later. Perhaps my eczema outcomes surged.
How do you know when a doctor is good? I don’t need to remind you how many different ways we are evaluated. Hospitals and insurance companies monitor our prescribing practices and therapeutic outcomes. Many websites rate our performance. Read your own reviews, if you dare, penned by people who range from the totally disgruntled to the charmingly gruntled.
Often, their reasons are either beside the point or just wrong.
An example: (1 star out of 5): “Dr. Rockoff was terrible. He prescribed a very powerful regimen, and when I told him it was drying me out, he just insisted I keep using it.”
In fact – I was able to figure out who the patient was – my “powerful treatment” was over-the-counter benzoyl peroxide along with topical clindamycin. As for my insistence that she continue, she never came back for another visit. But she had called for refills.
You can surely come up with your own review tales.
But if patients don’t really understand how well we do, doctors are not necessarily much better at assessing colleagues. This came to mind recently when a close friend, increasingly hobbled by arthritis (you get more such friends as the years roll by) was looking into getting his knee replaced. He asked friends and family and got several names of orthopedists at respectable institutions. (I don’t know how many of them were Best of Boston, or even Best of Nashua, New Hampshire.)
The patients made these referrals because either they or people they knew had Dr. So-and-So replace their knee and had been pleased. That is nice to hear, but what does it prove? Even backup shortstops get on base sometimes.
So my friend called his rheumatologist, who recommended a knee specialist. My friend consulted that doctor, found her pleasant and personable, and liked what she had to say about the surgery and its expected aftermath.
My friend called back his rheumatologist to report his decision to go with his recommended doctor.
“I’m glad to hear that,” said the rheumatologist. “Three of my friends went to her and were very pleased.”
I am not in any way criticizing the rheumatologist. When people ask me for referrals – to internists, to plastic surgeons – I give them names of people I know or have sent patients to who had good experiences, or whom I just heard good things about. What can I really know about their diagnostic acumen or surgical dexterity?
A useful counterexample is what happened with my cousin who underwent back surgery a while back. He was considering several specialists when he had a discussion with a younger acquaintance who was chief resident in neurosurgery at a local medical center, and had actually operated with several of the surgeons under consideration. “Don’t go to Dr A,” said the young man. “It takes him 7 hours to do that procedure. Better go to Dr. B, who gets it done in under 3. The shorter operative time makes a big difference in speed of recovery.”
That is the kind of specialized and relevant knowledge that actually matters. How many referrals can you think of that you made or heard of about which the same can be said?
In the meantime, I will return to my own Bestness, which has been frequent, though intermittent. I like to think of myself as a vintage Chardonnay. Some years I am the best. Other years, not so much. Your best bet is to consult me in one of the former.
Preferably chilled.
Dr. Rockoff, who wrote the Dermatology News column “Under My Skin,” is now semiretired, after 40 years of practice in Brookline, Mass. He served on the clinical faculty at Tufts University, Boston, and taught senior medical students and other trainees for 30 years. His second book, “Act Like a Doctor, Think Like a Patient,” is available online. Write to him at [email protected].
The fifth and last time I was listed as Best of Boston was in 2019, when I shared honors with obstetrics, ice cream, interior design, and kitchenware.
My first time on that list was 10 years earlier, and came as a surprise. Though the magazine that runs the feature said that selections are “peer-generated,” I was never asked to evaluate any colleagues, so I don’t know who my admiring peers were or what they admired.
Three years later I was dropped from the list, for equally mysterious reasons. Maybe my acne patients did worse that year. Be that as it may, I was reinstated several years later. Perhaps my eczema outcomes surged.
How do you know when a doctor is good? I don’t need to remind you how many different ways we are evaluated. Hospitals and insurance companies monitor our prescribing practices and therapeutic outcomes. Many websites rate our performance. Read your own reviews, if you dare, penned by people who range from the totally disgruntled to the charmingly gruntled.
Often, their reasons are either beside the point or just wrong.
An example: (1 star out of 5): “Dr. Rockoff was terrible. He prescribed a very powerful regimen, and when I told him it was drying me out, he just insisted I keep using it.”
In fact – I was able to figure out who the patient was – my “powerful treatment” was over-the-counter benzoyl peroxide along with topical clindamycin. As for my insistence that she continue, she never came back for another visit. But she had called for refills.
You can surely come up with your own review tales.
But if patients don’t really understand how well we do, doctors are not necessarily much better at assessing colleagues. This came to mind recently when a close friend, increasingly hobbled by arthritis (you get more such friends as the years roll by) was looking into getting his knee replaced. He asked friends and family and got several names of orthopedists at respectable institutions. (I don’t know how many of them were Best of Boston, or even Best of Nashua, New Hampshire.)
The patients made these referrals because either they or people they knew had Dr. So-and-So replace their knee and had been pleased. That is nice to hear, but what does it prove? Even backup shortstops get on base sometimes.
So my friend called his rheumatologist, who recommended a knee specialist. My friend consulted that doctor, found her pleasant and personable, and liked what she had to say about the surgery and its expected aftermath.
My friend called back his rheumatologist to report his decision to go with his recommended doctor.
“I’m glad to hear that,” said the rheumatologist. “Three of my friends went to her and were very pleased.”
I am not in any way criticizing the rheumatologist. When people ask me for referrals – to internists, to plastic surgeons – I give them names of people I know or have sent patients to who had good experiences, or whom I just heard good things about. What can I really know about their diagnostic acumen or surgical dexterity?
A useful counterexample is what happened with my cousin who underwent back surgery a while back. He was considering several specialists when he had a discussion with a younger acquaintance who was chief resident in neurosurgery at a local medical center, and had actually operated with several of the surgeons under consideration. “Don’t go to Dr A,” said the young man. “It takes him 7 hours to do that procedure. Better go to Dr. B, who gets it done in under 3. The shorter operative time makes a big difference in speed of recovery.”
That is the kind of specialized and relevant knowledge that actually matters. How many referrals can you think of that you made or heard of about which the same can be said?
In the meantime, I will return to my own Bestness, which has been frequent, though intermittent. I like to think of myself as a vintage Chardonnay. Some years I am the best. Other years, not so much. Your best bet is to consult me in one of the former.
Preferably chilled.
Dr. Rockoff, who wrote the Dermatology News column “Under My Skin,” is now semiretired, after 40 years of practice in Brookline, Mass. He served on the clinical faculty at Tufts University, Boston, and taught senior medical students and other trainees for 30 years. His second book, “Act Like a Doctor, Think Like a Patient,” is available online. Write to him at [email protected].
The fifth and last time I was listed as Best of Boston was in 2019, when I shared honors with obstetrics, ice cream, interior design, and kitchenware.
My first time on that list was 10 years earlier, and came as a surprise. Though the magazine that runs the feature said that selections are “peer-generated,” I was never asked to evaluate any colleagues, so I don’t know who my admiring peers were or what they admired.
Three years later I was dropped from the list, for equally mysterious reasons. Maybe my acne patients did worse that year. Be that as it may, I was reinstated several years later. Perhaps my eczema outcomes surged.
How do you know when a doctor is good? I don’t need to remind you how many different ways we are evaluated. Hospitals and insurance companies monitor our prescribing practices and therapeutic outcomes. Many websites rate our performance. Read your own reviews, if you dare, penned by people who range from the totally disgruntled to the charmingly gruntled.
Often, their reasons are either beside the point or just wrong.
An example: (1 star out of 5): “Dr. Rockoff was terrible. He prescribed a very powerful regimen, and when I told him it was drying me out, he just insisted I keep using it.”
In fact – I was able to figure out who the patient was – my “powerful treatment” was over-the-counter benzoyl peroxide along with topical clindamycin. As for my insistence that she continue, she never came back for another visit. But she had called for refills.
You can surely come up with your own review tales.
But if patients don’t really understand how well we do, doctors are not necessarily much better at assessing colleagues. This came to mind recently when a close friend, increasingly hobbled by arthritis (you get more such friends as the years roll by) was looking into getting his knee replaced. He asked friends and family and got several names of orthopedists at respectable institutions. (I don’t know how many of them were Best of Boston, or even Best of Nashua, New Hampshire.)
The patients made these referrals because either they or people they knew had Dr. So-and-So replace their knee and had been pleased. That is nice to hear, but what does it prove? Even backup shortstops get on base sometimes.
So my friend called his rheumatologist, who recommended a knee specialist. My friend consulted that doctor, found her pleasant and personable, and liked what she had to say about the surgery and its expected aftermath.
My friend called back his rheumatologist to report his decision to go with his recommended doctor.
“I’m glad to hear that,” said the rheumatologist. “Three of my friends went to her and were very pleased.”
I am not in any way criticizing the rheumatologist. When people ask me for referrals – to internists, to plastic surgeons – I give them names of people I know or have sent patients to who had good experiences, or whom I just heard good things about. What can I really know about their diagnostic acumen or surgical dexterity?
A useful counterexample is what happened with my cousin who underwent back surgery a while back. He was considering several specialists when he had a discussion with a younger acquaintance who was chief resident in neurosurgery at a local medical center, and had actually operated with several of the surgeons under consideration. “Don’t go to Dr A,” said the young man. “It takes him 7 hours to do that procedure. Better go to Dr. B, who gets it done in under 3. The shorter operative time makes a big difference in speed of recovery.”
That is the kind of specialized and relevant knowledge that actually matters. How many referrals can you think of that you made or heard of about which the same can be said?
In the meantime, I will return to my own Bestness, which has been frequent, though intermittent. I like to think of myself as a vintage Chardonnay. Some years I am the best. Other years, not so much. Your best bet is to consult me in one of the former.
Preferably chilled.
Dr. Rockoff, who wrote the Dermatology News column “Under My Skin,” is now semiretired, after 40 years of practice in Brookline, Mass. He served on the clinical faculty at Tufts University, Boston, and taught senior medical students and other trainees for 30 years. His second book, “Act Like a Doctor, Think Like a Patient,” is available online. Write to him at [email protected].
Myth busting: SARS-CoV-2 vaccine
MYTH: I shouldn’t get the vaccine because of potential long-term side effects
We know that 68 million people in the United States and 244 million people worldwide have already received messenger RNA (mRNA) SARS-CoV-2 vaccines (Pfizer/BioNTech and Moderna). So for the short-term side effects we already know more than we would know about most vaccines.
What about the long-term side effects? There are myths that these vaccines somehow could cause autoimmunity. This came from three publications where the possibility of mRNA vaccines to produce autoimmunity was brought up as a discussion point.1-3 There was no evidence given in these publications, it was raised only as a hypothetical possibility.
There’s no evidence that mRNA or replication-defective DNA vaccines (AstraZeneca/Oxford and Johnson & Johnson) produce autoimmunity. Moreover, the mRNA and replication-defective DNA, once it’s inside of the muscle cell, is gone within a few days. What’s left after ribosome processing is the spike (S) protein as an immunogen. We’ve been vaccinating with proteins for 50 years and we haven’t seen autoimmunity.
MYTH: The vaccines aren’t safe because they were developed so quickly
These vaccines were developed at “warp speed” – that doesn’t mean they were developed without all the same safety safeguards that the Food and Drug Administration requires. The reason it happened so fast is because the seriousness of the pandemic allowed us, as a community, to enroll the patients into the studies fast. In a matter of months, we had all the studies filled. In a normal circumstance, that might take 2 or 3 years. And all of the regulatory agencies – the National Institutes of Health, the FDA, the Centers for Disease Control and Prevention – were ready to take the information and put a panel of specialists together and immediately review the data. No safety steps were missed. The same process that’s always required of phase 1, of phase 2, and then at phase 3 were accomplished.
The novelty of these vaccines was that they could be made so quickly. Messenger RNA vaccines can be made in a matter of days and then manufactured in a matter of 2 months. The DNA vaccines has a similar timeline trajectory.
MYTH: There’s no point in getting the vaccines because we still have to wear masks
Right now, out of an abundance of caution, until it’s proven that we don’t have to wear masks, it’s being recommended that we do so for the safety of others. Early data suggest that this will be temporary. In time, I suspect it will be shown that, after we receive the vaccine, it will be shown that we are not contagious to others and we’ll be able to get rid of our masks.
MYTH: I already had COVID-19 so I don’t need the vaccine
Some people have already caught the SARS-CoV-2 virus that causes this infection and so they feel that they’re immune and they don’t need to get the vaccine. Time will tell if that’s the case. Right now, we don’t know for sure. Early data suggest that a single dose of vaccine in persons who have had the infection may be sufficient. Over time, what happens in the vaccine field is we measure the immunity from the vaccine, and from people who’ve gotten the infection, and we find that there’s a measurement in the blood that correlates with protection. Right now, we don’t know that correlate of protection level. So, out of an abundance of caution, it’s being recommended that, even if you had the disease, maybe you didn’t develop enough immunity, and it’s better to get the vaccine than to get the illness a second time.
MYTH: The vaccines can give me SARS-CoV-2 infection
The new vaccines for COVID-19, released under emergency use Authorization, are mRNA and DNA vaccines. They are a blueprint for the Spike (S) protein of the virus. In order to become a protein, the mRNA, once it’s inside the cell, is processed by ribosomes. The product of the ribosome processing is a protein that cannot possibly cause harm as a virus. It’s a little piece of mRNA inside of a lipid nanoparticle, which is just a casing to protect the mRNA from breaking down until it’s injected in the body. The replication defective DNA vaccines (AstraZeneca/Oxford and Johnson & Johnson) are packaged inside of virus cells (adenoviruses). The DNA vaccines involve a three-step process:
- 1. The adenovirus, containing replication-defective DNA that encodes mRNA for the Spike (S) protein, is taken up by the host cells where it must make its way to the nucleus of the muscle cell.
- 2. The DNA is injected into the host cell nucleus and in the nucleus the DNA is decoded to an mRNA.
- 3. The mRNA is released from the nucleus and transported to the cell cytoplasm where the ribosomes process the mRNA in an identical manner as mRNA vaccines.
MYTH: The COVID-19 vaccines can alter my DNA
The mRNA and replication-defective DNA vaccines never interact with your DNA. mRNA vaccines never enter the nucleus. Replication-defective DNA vaccines cannot replicate and do not interact with host DNA. The vaccines can’t change your DNA.
Here is a link to YouTube videos I made on this topic: https://youtube.com/playlist?list=PLve-0UW04UMRKHfFbXyEpLY8GCm2WyJHD.
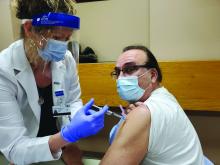
Here is a photo of me receiving my first SARS-CoV-2 shot (Moderna) in January 2021. I received my second shot in February. I am a lot less anxious. I hope my vaccine card will be a ticket to travel in the future.
Dr. Pichichero is a specialist in pediatric infectious diseases and director of the Research Institute at Rochester (N.Y.) General Hospital. He has no conflicts of interest to report.
References
1. Peck KM and Lauring AS. J Virol. 2018. doi: 10.1128/JVI.01031-17.
2. Pepini T et al. J Immunol. 2017 May 15. doi: 10.4049/jimmunol.1601877.
3. Theofilopoulos AN et al. Annu Rev Immunol. 2005. doi: 10.1146/annurev.immunol.23.021704.115843.
MYTH: I shouldn’t get the vaccine because of potential long-term side effects
We know that 68 million people in the United States and 244 million people worldwide have already received messenger RNA (mRNA) SARS-CoV-2 vaccines (Pfizer/BioNTech and Moderna). So for the short-term side effects we already know more than we would know about most vaccines.
What about the long-term side effects? There are myths that these vaccines somehow could cause autoimmunity. This came from three publications where the possibility of mRNA vaccines to produce autoimmunity was brought up as a discussion point.1-3 There was no evidence given in these publications, it was raised only as a hypothetical possibility.
There’s no evidence that mRNA or replication-defective DNA vaccines (AstraZeneca/Oxford and Johnson & Johnson) produce autoimmunity. Moreover, the mRNA and replication-defective DNA, once it’s inside of the muscle cell, is gone within a few days. What’s left after ribosome processing is the spike (S) protein as an immunogen. We’ve been vaccinating with proteins for 50 years and we haven’t seen autoimmunity.
MYTH: The vaccines aren’t safe because they were developed so quickly
These vaccines were developed at “warp speed” – that doesn’t mean they were developed without all the same safety safeguards that the Food and Drug Administration requires. The reason it happened so fast is because the seriousness of the pandemic allowed us, as a community, to enroll the patients into the studies fast. In a matter of months, we had all the studies filled. In a normal circumstance, that might take 2 or 3 years. And all of the regulatory agencies – the National Institutes of Health, the FDA, the Centers for Disease Control and Prevention – were ready to take the information and put a panel of specialists together and immediately review the data. No safety steps were missed. The same process that’s always required of phase 1, of phase 2, and then at phase 3 were accomplished.
The novelty of these vaccines was that they could be made so quickly. Messenger RNA vaccines can be made in a matter of days and then manufactured in a matter of 2 months. The DNA vaccines has a similar timeline trajectory.
MYTH: There’s no point in getting the vaccines because we still have to wear masks
Right now, out of an abundance of caution, until it’s proven that we don’t have to wear masks, it’s being recommended that we do so for the safety of others. Early data suggest that this will be temporary. In time, I suspect it will be shown that, after we receive the vaccine, it will be shown that we are not contagious to others and we’ll be able to get rid of our masks.
MYTH: I already had COVID-19 so I don’t need the vaccine
Some people have already caught the SARS-CoV-2 virus that causes this infection and so they feel that they’re immune and they don’t need to get the vaccine. Time will tell if that’s the case. Right now, we don’t know for sure. Early data suggest that a single dose of vaccine in persons who have had the infection may be sufficient. Over time, what happens in the vaccine field is we measure the immunity from the vaccine, and from people who’ve gotten the infection, and we find that there’s a measurement in the blood that correlates with protection. Right now, we don’t know that correlate of protection level. So, out of an abundance of caution, it’s being recommended that, even if you had the disease, maybe you didn’t develop enough immunity, and it’s better to get the vaccine than to get the illness a second time.
MYTH: The vaccines can give me SARS-CoV-2 infection
The new vaccines for COVID-19, released under emergency use Authorization, are mRNA and DNA vaccines. They are a blueprint for the Spike (S) protein of the virus. In order to become a protein, the mRNA, once it’s inside the cell, is processed by ribosomes. The product of the ribosome processing is a protein that cannot possibly cause harm as a virus. It’s a little piece of mRNA inside of a lipid nanoparticle, which is just a casing to protect the mRNA from breaking down until it’s injected in the body. The replication defective DNA vaccines (AstraZeneca/Oxford and Johnson & Johnson) are packaged inside of virus cells (adenoviruses). The DNA vaccines involve a three-step process:
- 1. The adenovirus, containing replication-defective DNA that encodes mRNA for the Spike (S) protein, is taken up by the host cells where it must make its way to the nucleus of the muscle cell.
- 2. The DNA is injected into the host cell nucleus and in the nucleus the DNA is decoded to an mRNA.
- 3. The mRNA is released from the nucleus and transported to the cell cytoplasm where the ribosomes process the mRNA in an identical manner as mRNA vaccines.
MYTH: The COVID-19 vaccines can alter my DNA
The mRNA and replication-defective DNA vaccines never interact with your DNA. mRNA vaccines never enter the nucleus. Replication-defective DNA vaccines cannot replicate and do not interact with host DNA. The vaccines can’t change your DNA.
Here is a link to YouTube videos I made on this topic: https://youtube.com/playlist?list=PLve-0UW04UMRKHfFbXyEpLY8GCm2WyJHD.
Here is a photo of me receiving my first SARS-CoV-2 shot (Moderna) in January 2021. I received my second shot in February. I am a lot less anxious. I hope my vaccine card will be a ticket to travel in the future.
Dr. Pichichero is a specialist in pediatric infectious diseases and director of the Research Institute at Rochester (N.Y.) General Hospital. He has no conflicts of interest to report.
References
1. Peck KM and Lauring AS. J Virol. 2018. doi: 10.1128/JVI.01031-17.
2. Pepini T et al. J Immunol. 2017 May 15. doi: 10.4049/jimmunol.1601877.
3. Theofilopoulos AN et al. Annu Rev Immunol. 2005. doi: 10.1146/annurev.immunol.23.021704.115843.
MYTH: I shouldn’t get the vaccine because of potential long-term side effects
We know that 68 million people in the United States and 244 million people worldwide have already received messenger RNA (mRNA) SARS-CoV-2 vaccines (Pfizer/BioNTech and Moderna). So for the short-term side effects we already know more than we would know about most vaccines.
What about the long-term side effects? There are myths that these vaccines somehow could cause autoimmunity. This came from three publications where the possibility of mRNA vaccines to produce autoimmunity was brought up as a discussion point.1-3 There was no evidence given in these publications, it was raised only as a hypothetical possibility.
There’s no evidence that mRNA or replication-defective DNA vaccines (AstraZeneca/Oxford and Johnson & Johnson) produce autoimmunity. Moreover, the mRNA and replication-defective DNA, once it’s inside of the muscle cell, is gone within a few days. What’s left after ribosome processing is the spike (S) protein as an immunogen. We’ve been vaccinating with proteins for 50 years and we haven’t seen autoimmunity.
MYTH: The vaccines aren’t safe because they were developed so quickly
These vaccines were developed at “warp speed” – that doesn’t mean they were developed without all the same safety safeguards that the Food and Drug Administration requires. The reason it happened so fast is because the seriousness of the pandemic allowed us, as a community, to enroll the patients into the studies fast. In a matter of months, we had all the studies filled. In a normal circumstance, that might take 2 or 3 years. And all of the regulatory agencies – the National Institutes of Health, the FDA, the Centers for Disease Control and Prevention – were ready to take the information and put a panel of specialists together and immediately review the data. No safety steps were missed. The same process that’s always required of phase 1, of phase 2, and then at phase 3 were accomplished.
The novelty of these vaccines was that they could be made so quickly. Messenger RNA vaccines can be made in a matter of days and then manufactured in a matter of 2 months. The DNA vaccines has a similar timeline trajectory.
MYTH: There’s no point in getting the vaccines because we still have to wear masks
Right now, out of an abundance of caution, until it’s proven that we don’t have to wear masks, it’s being recommended that we do so for the safety of others. Early data suggest that this will be temporary. In time, I suspect it will be shown that, after we receive the vaccine, it will be shown that we are not contagious to others and we’ll be able to get rid of our masks.
MYTH: I already had COVID-19 so I don’t need the vaccine
Some people have already caught the SARS-CoV-2 virus that causes this infection and so they feel that they’re immune and they don’t need to get the vaccine. Time will tell if that’s the case. Right now, we don’t know for sure. Early data suggest that a single dose of vaccine in persons who have had the infection may be sufficient. Over time, what happens in the vaccine field is we measure the immunity from the vaccine, and from people who’ve gotten the infection, and we find that there’s a measurement in the blood that correlates with protection. Right now, we don’t know that correlate of protection level. So, out of an abundance of caution, it’s being recommended that, even if you had the disease, maybe you didn’t develop enough immunity, and it’s better to get the vaccine than to get the illness a second time.
MYTH: The vaccines can give me SARS-CoV-2 infection
The new vaccines for COVID-19, released under emergency use Authorization, are mRNA and DNA vaccines. They are a blueprint for the Spike (S) protein of the virus. In order to become a protein, the mRNA, once it’s inside the cell, is processed by ribosomes. The product of the ribosome processing is a protein that cannot possibly cause harm as a virus. It’s a little piece of mRNA inside of a lipid nanoparticle, which is just a casing to protect the mRNA from breaking down until it’s injected in the body. The replication defective DNA vaccines (AstraZeneca/Oxford and Johnson & Johnson) are packaged inside of virus cells (adenoviruses). The DNA vaccines involve a three-step process:
- 1. The adenovirus, containing replication-defective DNA that encodes mRNA for the Spike (S) protein, is taken up by the host cells where it must make its way to the nucleus of the muscle cell.
- 2. The DNA is injected into the host cell nucleus and in the nucleus the DNA is decoded to an mRNA.
- 3. The mRNA is released from the nucleus and transported to the cell cytoplasm where the ribosomes process the mRNA in an identical manner as mRNA vaccines.
MYTH: The COVID-19 vaccines can alter my DNA
The mRNA and replication-defective DNA vaccines never interact with your DNA. mRNA vaccines never enter the nucleus. Replication-defective DNA vaccines cannot replicate and do not interact with host DNA. The vaccines can’t change your DNA.
Here is a link to YouTube videos I made on this topic: https://youtube.com/playlist?list=PLve-0UW04UMRKHfFbXyEpLY8GCm2WyJHD.
Here is a photo of me receiving my first SARS-CoV-2 shot (Moderna) in January 2021. I received my second shot in February. I am a lot less anxious. I hope my vaccine card will be a ticket to travel in the future.
Dr. Pichichero is a specialist in pediatric infectious diseases and director of the Research Institute at Rochester (N.Y.) General Hospital. He has no conflicts of interest to report.
References
1. Peck KM and Lauring AS. J Virol. 2018. doi: 10.1128/JVI.01031-17.
2. Pepini T et al. J Immunol. 2017 May 15. doi: 10.4049/jimmunol.1601877.
3. Theofilopoulos AN et al. Annu Rev Immunol. 2005. doi: 10.1146/annurev.immunol.23.021704.115843.
Palliative care and hospital medicine partnerships in the pandemic
Patients dying without their loved ones, families forced to remotely decide goals of care without the physical presence or human connection of the care team, overworked staff physically isolated from their critically ill patients, and at-risk community members with uncertain and undocumented goals for care are among the universal challenges confronted by hospitals and hospitalists during this COVID-19 pandemic. Partnerships among hospital medicine (HM) and palliative care (PC) teams at Dell Medical School/Dell Seton Medical Center thrive on mutually shared core values of patient centered care – compassion, empathy, and humanity.
A key PC-HM collaboration was adapting our multidisciplinary huddle to focus on communication effectiveness and efficiency in the medical intensive care unit (MICU). Expanded interprofessional and cross-specialty collaboration promoted streamlined, succinct, and standardized communication with patients’ families while their loved ones were critically ill with COVID-19. The PC team attended daily MICU multidisciplinary huddles, attentive to both the medical and psychosocial updates for each patient. During huddles, residents or HM providers were asked to end their presentation with a clinical status “headline” and solicited feedback from the multidisciplinary team before messaging to the family. The PC team then communicated with families a succinct and cohesive medical update and continuously explored goals of care. This allowed the HM team, often overwhelmed with admissions, co-managing intensive care patients, and facilitating safe discharges, to focus on urgent issues while PC provided continuity and personalized support for patients and families. PC’s ability to synthesize and summarize clinical information from multiple teams and then provide cohesive updates in patient-friendly language modeled important communication skills for learners and simultaneously benefited HM providers.
Our chaplains, too, were central to facilitating timely, proactive conversations and documentation of Medical Power of Attorney (MPOA) for patients with COVID-19 admitted to our hospital. HM prioritized early admission conversations with patients to counsel them on severity of illness, prognosis based on risk factors, to elucidate wishes for intubation or resuscitation, and to capture their desired medical decision maker. HM was notified of all COVID and PUI admissions, allowing us to speak with even critically ill patients in the ER or ICU prior to intubation in order to quickly and accurately capture patients’ wishes for treatment and delegate decision makers. Our chaplains supported and supplemented these efforts by diligently and dutifully soliciting, hearing, and documenting patient MPOA delegates, with over 50% MPOA completion by 24 hours of hospitalization.
Another early PC-HM project, “Meet My Loved One,” was adapted from the University of Alabama at Birmingham Palliative and Comfort Unit. The absence of families visiting the ICU and sharing pictures, stories, anecdotes of our patients left a deeply felt, dehumanizing void in the halls and rooms of our hospital. To fill this space with life and humanity, furloughed medical students on their “transition of care” electives contacted family members of their “continuity” patients focusing primarily on those patients expected to have prolonged ICU or hospital stays and solicited personal, humanizing information about our patients. Questions included: “What is your loved one’s preferred name or nickname?” and “What are three things we should know to take better care of your loved one?” With family permission, we posted this information on the door outside the patient’s room. Nursing staff, in particular, appreciated getting to know their patients more personally and families appreciated the staff’s desire to know their loved one as an individual.
It is also important to acknowledge setbacks. Early efforts to engage technology proved more foe than friend. We continue to struggle with using our iPads for video visits. Most of our families prefer “WhatsApp” for video communication, which is not compatible with operating systems on early versions of the iPad, which were generously and widely donated by local school systems. Desperate to allow families to connect, many providers resorted to using personal devices to facilitate video visits and family meetings. And we discovered that many video visits caused more not less family angst, especially for critically ill patients. Families often required preparation and coaching on what to expect and how to interact with intubated, sedated, proned, and paralyzed loved ones.
Our hospital medicine and palliative care teams have an established strong partnership. The COVID-19 pandemic created novel communication challenges but our shared mission toward patient-centered care allowed us to effectively collaborate to bring the patients goals of care to the forefront aligning patients, families, physicians, nurses, and staff during the COVID-19 surge.
Dr. Johnston is associate professor at Dell Medical School at The University of Texas in Austin. She practices hospital medicine and inpatient palliative care at Dell Seton Medical Center. Dr. Cooremans is a resident physician at Dell Medical School. Dr. Salib is the internal medicine clerkship director and an associate professor at Dell Medical School. Dr. Nieto is an assistant professor and associate chief of the Division of Hospital Medicine at Dell Medical School. Dr. Patel is an assistant professor at Dell Medical School. This article is part of a series written by members of the Division of Hospital Medicine at Dell Medical School, exploring lessons learned from the coronavirus pandemic and outlining an approach for creating COVID-19 Centers of Excellence. The article first appeared in The Hospital Leader, the official blog of SHM.
Patients dying without their loved ones, families forced to remotely decide goals of care without the physical presence or human connection of the care team, overworked staff physically isolated from their critically ill patients, and at-risk community members with uncertain and undocumented goals for care are among the universal challenges confronted by hospitals and hospitalists during this COVID-19 pandemic. Partnerships among hospital medicine (HM) and palliative care (PC) teams at Dell Medical School/Dell Seton Medical Center thrive on mutually shared core values of patient centered care – compassion, empathy, and humanity.
A key PC-HM collaboration was adapting our multidisciplinary huddle to focus on communication effectiveness and efficiency in the medical intensive care unit (MICU). Expanded interprofessional and cross-specialty collaboration promoted streamlined, succinct, and standardized communication with patients’ families while their loved ones were critically ill with COVID-19. The PC team attended daily MICU multidisciplinary huddles, attentive to both the medical and psychosocial updates for each patient. During huddles, residents or HM providers were asked to end their presentation with a clinical status “headline” and solicited feedback from the multidisciplinary team before messaging to the family. The PC team then communicated with families a succinct and cohesive medical update and continuously explored goals of care. This allowed the HM team, often overwhelmed with admissions, co-managing intensive care patients, and facilitating safe discharges, to focus on urgent issues while PC provided continuity and personalized support for patients and families. PC’s ability to synthesize and summarize clinical information from multiple teams and then provide cohesive updates in patient-friendly language modeled important communication skills for learners and simultaneously benefited HM providers.
Our chaplains, too, were central to facilitating timely, proactive conversations and documentation of Medical Power of Attorney (MPOA) for patients with COVID-19 admitted to our hospital. HM prioritized early admission conversations with patients to counsel them on severity of illness, prognosis based on risk factors, to elucidate wishes for intubation or resuscitation, and to capture their desired medical decision maker. HM was notified of all COVID and PUI admissions, allowing us to speak with even critically ill patients in the ER or ICU prior to intubation in order to quickly and accurately capture patients’ wishes for treatment and delegate decision makers. Our chaplains supported and supplemented these efforts by diligently and dutifully soliciting, hearing, and documenting patient MPOA delegates, with over 50% MPOA completion by 24 hours of hospitalization.
Another early PC-HM project, “Meet My Loved One,” was adapted from the University of Alabama at Birmingham Palliative and Comfort Unit. The absence of families visiting the ICU and sharing pictures, stories, anecdotes of our patients left a deeply felt, dehumanizing void in the halls and rooms of our hospital. To fill this space with life and humanity, furloughed medical students on their “transition of care” electives contacted family members of their “continuity” patients focusing primarily on those patients expected to have prolonged ICU or hospital stays and solicited personal, humanizing information about our patients. Questions included: “What is your loved one’s preferred name or nickname?” and “What are three things we should know to take better care of your loved one?” With family permission, we posted this information on the door outside the patient’s room. Nursing staff, in particular, appreciated getting to know their patients more personally and families appreciated the staff’s desire to know their loved one as an individual.
It is also important to acknowledge setbacks. Early efforts to engage technology proved more foe than friend. We continue to struggle with using our iPads for video visits. Most of our families prefer “WhatsApp” for video communication, which is not compatible with operating systems on early versions of the iPad, which were generously and widely donated by local school systems. Desperate to allow families to connect, many providers resorted to using personal devices to facilitate video visits and family meetings. And we discovered that many video visits caused more not less family angst, especially for critically ill patients. Families often required preparation and coaching on what to expect and how to interact with intubated, sedated, proned, and paralyzed loved ones.
Our hospital medicine and palliative care teams have an established strong partnership. The COVID-19 pandemic created novel communication challenges but our shared mission toward patient-centered care allowed us to effectively collaborate to bring the patients goals of care to the forefront aligning patients, families, physicians, nurses, and staff during the COVID-19 surge.
Dr. Johnston is associate professor at Dell Medical School at The University of Texas in Austin. She practices hospital medicine and inpatient palliative care at Dell Seton Medical Center. Dr. Cooremans is a resident physician at Dell Medical School. Dr. Salib is the internal medicine clerkship director and an associate professor at Dell Medical School. Dr. Nieto is an assistant professor and associate chief of the Division of Hospital Medicine at Dell Medical School. Dr. Patel is an assistant professor at Dell Medical School. This article is part of a series written by members of the Division of Hospital Medicine at Dell Medical School, exploring lessons learned from the coronavirus pandemic and outlining an approach for creating COVID-19 Centers of Excellence. The article first appeared in The Hospital Leader, the official blog of SHM.
Patients dying without their loved ones, families forced to remotely decide goals of care without the physical presence or human connection of the care team, overworked staff physically isolated from their critically ill patients, and at-risk community members with uncertain and undocumented goals for care are among the universal challenges confronted by hospitals and hospitalists during this COVID-19 pandemic. Partnerships among hospital medicine (HM) and palliative care (PC) teams at Dell Medical School/Dell Seton Medical Center thrive on mutually shared core values of patient centered care – compassion, empathy, and humanity.
A key PC-HM collaboration was adapting our multidisciplinary huddle to focus on communication effectiveness and efficiency in the medical intensive care unit (MICU). Expanded interprofessional and cross-specialty collaboration promoted streamlined, succinct, and standardized communication with patients’ families while their loved ones were critically ill with COVID-19. The PC team attended daily MICU multidisciplinary huddles, attentive to both the medical and psychosocial updates for each patient. During huddles, residents or HM providers were asked to end their presentation with a clinical status “headline” and solicited feedback from the multidisciplinary team before messaging to the family. The PC team then communicated with families a succinct and cohesive medical update and continuously explored goals of care. This allowed the HM team, often overwhelmed with admissions, co-managing intensive care patients, and facilitating safe discharges, to focus on urgent issues while PC provided continuity and personalized support for patients and families. PC’s ability to synthesize and summarize clinical information from multiple teams and then provide cohesive updates in patient-friendly language modeled important communication skills for learners and simultaneously benefited HM providers.
Our chaplains, too, were central to facilitating timely, proactive conversations and documentation of Medical Power of Attorney (MPOA) for patients with COVID-19 admitted to our hospital. HM prioritized early admission conversations with patients to counsel them on severity of illness, prognosis based on risk factors, to elucidate wishes for intubation or resuscitation, and to capture their desired medical decision maker. HM was notified of all COVID and PUI admissions, allowing us to speak with even critically ill patients in the ER or ICU prior to intubation in order to quickly and accurately capture patients’ wishes for treatment and delegate decision makers. Our chaplains supported and supplemented these efforts by diligently and dutifully soliciting, hearing, and documenting patient MPOA delegates, with over 50% MPOA completion by 24 hours of hospitalization.
Another early PC-HM project, “Meet My Loved One,” was adapted from the University of Alabama at Birmingham Palliative and Comfort Unit. The absence of families visiting the ICU and sharing pictures, stories, anecdotes of our patients left a deeply felt, dehumanizing void in the halls and rooms of our hospital. To fill this space with life and humanity, furloughed medical students on their “transition of care” electives contacted family members of their “continuity” patients focusing primarily on those patients expected to have prolonged ICU or hospital stays and solicited personal, humanizing information about our patients. Questions included: “What is your loved one’s preferred name or nickname?” and “What are three things we should know to take better care of your loved one?” With family permission, we posted this information on the door outside the patient’s room. Nursing staff, in particular, appreciated getting to know their patients more personally and families appreciated the staff’s desire to know their loved one as an individual.
It is also important to acknowledge setbacks. Early efforts to engage technology proved more foe than friend. We continue to struggle with using our iPads for video visits. Most of our families prefer “WhatsApp” for video communication, which is not compatible with operating systems on early versions of the iPad, which were generously and widely donated by local school systems. Desperate to allow families to connect, many providers resorted to using personal devices to facilitate video visits and family meetings. And we discovered that many video visits caused more not less family angst, especially for critically ill patients. Families often required preparation and coaching on what to expect and how to interact with intubated, sedated, proned, and paralyzed loved ones.
Our hospital medicine and palliative care teams have an established strong partnership. The COVID-19 pandemic created novel communication challenges but our shared mission toward patient-centered care allowed us to effectively collaborate to bring the patients goals of care to the forefront aligning patients, families, physicians, nurses, and staff during the COVID-19 surge.
Dr. Johnston is associate professor at Dell Medical School at The University of Texas in Austin. She practices hospital medicine and inpatient palliative care at Dell Seton Medical Center. Dr. Cooremans is a resident physician at Dell Medical School. Dr. Salib is the internal medicine clerkship director and an associate professor at Dell Medical School. Dr. Nieto is an assistant professor and associate chief of the Division of Hospital Medicine at Dell Medical School. Dr. Patel is an assistant professor at Dell Medical School. This article is part of a series written by members of the Division of Hospital Medicine at Dell Medical School, exploring lessons learned from the coronavirus pandemic and outlining an approach for creating COVID-19 Centers of Excellence. The article first appeared in The Hospital Leader, the official blog of SHM.
Who do you call in those late, quiet hours, when all seems lost?
I swear by Apollo Physician and Asclepius and Hygeia and Panacea and all the gods and goddesses, making them my witnesses, that I will fulfill according to my ability and judgment this oath and this covenant.
On my desk sits a bust of Hygeia, a mask from Venice, next to a small sculpture and a figurine of the plague doctor. Nearby, there is a Klimt closeup of Hygeia, a postcard portraying Asclepius, St. Sebastian paintings, and quotes from Maimonides. They whisper secrets and nod to the challenges of the past. These medical specters, ancient voices of the past, keep me grounded. They speak, listen, and elevate me, too. They bring life into my otherwise quiet room.
We all began our careers swearing to Apollo, Asclepius, Hygeia, and Panacea when we recited the Hippocratic Oath. I call upon them, and other gods and totems, and saints and ancient healers, now more than ever. As an atheist, I don’t appeal to them as prayers, but as Hippocrates intended. I look to their supernatural healing powers as a source of strength and as revealers of the natural and observable phenomena.
Apollo was one of the Twelve Olympians, a God of medicine, father of Asclepius. He was a healer, though his arrows also bore the plagues of the Gods.
For centuries, Apollo was found floating above the marble dissection table in the Bologna anatomical theater, guiding students who dove into the secrets of the human body.
Asclepius, son of Apollo, was hailed as a god of medicine. He healed many from plagues at his temples throughout the Ancient Greek and Roman empires. He was mentored in the healing arts by the centaur, Chiron. His many daughters and sons represent various aspects of medicine including cures, healing, recovery, sanitation, and beauty. To Asclepius, temples were places of healing, an ancient ancestor to modern hospitals.
Two of his daughters, Panacea and Hygeia, gave us the healing words of panacea and hygiene. Today, these acts of hygiene, handwashing, mask-wearing, and sanitation are discussed across the world louder than ever. While we’re all wishing for a panacea, we know it will take all the attributes of medicine to get us through this pandemic.
Hospitalists are part of the frontline teams facing this pandemic head-on. Gowning up for MRSA isolation seems quaint nowadays.
My attendings spoke of their fears, up against the unknown while on service in the 1980s, when HIV appeared. 2014 brought the Ebola biocontainment units. Now, this generation works daily against a modern plague, where every day is a risk of exposure. When every patient is in isolation, the garb begins to reflect the PPE that emerged during a 17th-century plague epidemics, the plague doctor outfit.
Godfather II fans recall the famous portrayal of the August 16th festival to San Rocco play out in the streets of New York. For those stricken with COVID-19 and recovered, you emulate San Rocco, in your continued return to service.
The Scuola Grande di San Rocco, in Venice, is the epitome of healing and greatness in one building. Tintoretto, the great Venetian painter, assembled the story of healing through art and portraits of San Rocco. The scuola, a confraternity, was a community of healers, gathered in one place to look after the less fortunate.
Hospitalists march into the hospital risking their lives. We always wear PPE for MRSA, ESBL, or C. diff. And enter reverse isolation rooms wearing N95s for possible TB cases. But those don’t elevate to the volume, to the same fear, as gowning up for COVID-19.
Hospitalists, frontline health care workers, embody the story of San Sebastian, another plague saint who absorbed the arrows, the symbolic plagues, onto his own shoulders so no one else had to bear them. San Sebastian was a Christian persecuted by a Roman emperor once his beliefs were discovered. He is often laden with arrows in spots where buboes would have appeared: the armpits and the groin. His sacrifice for others’ recovery became a symbol of absorbing the plague, the wounds, and the impact of the arrows.
This sacrifice epitomizes the daily work the frontline nurses, ER docs, intensivists, hospitalists, and the entire hospital staff perform daily, bearing the slung arrows of coronavirus.
One of the images I think of frequently during this time lies atop Castel San Angelo in Rome. Built in 161 AD, it has served as a mausoleum, prison, papal residence, and is currently a museum. Atop San’Angelo stands St. Michael, the destroyer of the dragon. He is sheathing his sword in representation of the end of the plague in 590.
The arrows flow, yet the sword will be sheathed. Evil will be halted. The stories of these ancient totems and strength can give us strength as they remind us of the work that was done for centuries: pestilence, famine, war. The great killers never go away completely.
Fast forward to today
These medical specters serve as reminders of what makes the field of medicine so inspiring: the selfless acts, the fortitude of spirit, the healers, the long history, and the shoulders of giants we stand upon. From these stories, we spring the healing waters we bathe in to give us the courage to wake up and care for our patients each day. These specters encourage us to defeat any and all of the scourges that come our way.
I hear and read stories about the frontline heroes, the vaccine makers, the PPE creators, the health care workers, grocery store clerks, and teachers. I’m honored to hear of these stories and your sacrifices. I’m inspired to continue upholding your essence, your fight, and your stories. In keeping with ancient empire metaphors, you are taking the slings of the diseased arrows flying to our brethren as you try to keep yourself and others safe.
The sheathing of this sword will come. These arrows will be silenced. But until then, I lean on these pictures, these stories, and these saints, to give us all the strength to wake up each morning and continue healing.
They serve as reminders of what makes the field of medicine so great: the selfless acts, the fortitude of spirit, the healers, the long history, and the shoulders of giants we stand upon. From these stories spring the healing waters we bathe in to give us the courage to wake up and care for our patients each day and defeat any and all scourges that come our way.
So, who do you call in those late, quiet hours, when all seems lost?
Dr. Messler is the executive director, quality initiatives at Glytec and works as a hospitalist at Morton Plant Hospitalist group in Clearwater, Fla. This essay appeared initially on The Hospital Leader, the official blog of SHM.
I swear by Apollo Physician and Asclepius and Hygeia and Panacea and all the gods and goddesses, making them my witnesses, that I will fulfill according to my ability and judgment this oath and this covenant.
On my desk sits a bust of Hygeia, a mask from Venice, next to a small sculpture and a figurine of the plague doctor. Nearby, there is a Klimt closeup of Hygeia, a postcard portraying Asclepius, St. Sebastian paintings, and quotes from Maimonides. They whisper secrets and nod to the challenges of the past. These medical specters, ancient voices of the past, keep me grounded. They speak, listen, and elevate me, too. They bring life into my otherwise quiet room.
We all began our careers swearing to Apollo, Asclepius, Hygeia, and Panacea when we recited the Hippocratic Oath. I call upon them, and other gods and totems, and saints and ancient healers, now more than ever. As an atheist, I don’t appeal to them as prayers, but as Hippocrates intended. I look to their supernatural healing powers as a source of strength and as revealers of the natural and observable phenomena.
Apollo was one of the Twelve Olympians, a God of medicine, father of Asclepius. He was a healer, though his arrows also bore the plagues of the Gods.
For centuries, Apollo was found floating above the marble dissection table in the Bologna anatomical theater, guiding students who dove into the secrets of the human body.
Asclepius, son of Apollo, was hailed as a god of medicine. He healed many from plagues at his temples throughout the Ancient Greek and Roman empires. He was mentored in the healing arts by the centaur, Chiron. His many daughters and sons represent various aspects of medicine including cures, healing, recovery, sanitation, and beauty. To Asclepius, temples were places of healing, an ancient ancestor to modern hospitals.
Two of his daughters, Panacea and Hygeia, gave us the healing words of panacea and hygiene. Today, these acts of hygiene, handwashing, mask-wearing, and sanitation are discussed across the world louder than ever. While we’re all wishing for a panacea, we know it will take all the attributes of medicine to get us through this pandemic.
Hospitalists are part of the frontline teams facing this pandemic head-on. Gowning up for MRSA isolation seems quaint nowadays.
My attendings spoke of their fears, up against the unknown while on service in the 1980s, when HIV appeared. 2014 brought the Ebola biocontainment units. Now, this generation works daily against a modern plague, where every day is a risk of exposure. When every patient is in isolation, the garb begins to reflect the PPE that emerged during a 17th-century plague epidemics, the plague doctor outfit.
Godfather II fans recall the famous portrayal of the August 16th festival to San Rocco play out in the streets of New York. For those stricken with COVID-19 and recovered, you emulate San Rocco, in your continued return to service.
The Scuola Grande di San Rocco, in Venice, is the epitome of healing and greatness in one building. Tintoretto, the great Venetian painter, assembled the story of healing through art and portraits of San Rocco. The scuola, a confraternity, was a community of healers, gathered in one place to look after the less fortunate.
Hospitalists march into the hospital risking their lives. We always wear PPE for MRSA, ESBL, or C. diff. And enter reverse isolation rooms wearing N95s for possible TB cases. But those don’t elevate to the volume, to the same fear, as gowning up for COVID-19.
Hospitalists, frontline health care workers, embody the story of San Sebastian, another plague saint who absorbed the arrows, the symbolic plagues, onto his own shoulders so no one else had to bear them. San Sebastian was a Christian persecuted by a Roman emperor once his beliefs were discovered. He is often laden with arrows in spots where buboes would have appeared: the armpits and the groin. His sacrifice for others’ recovery became a symbol of absorbing the plague, the wounds, and the impact of the arrows.
This sacrifice epitomizes the daily work the frontline nurses, ER docs, intensivists, hospitalists, and the entire hospital staff perform daily, bearing the slung arrows of coronavirus.
One of the images I think of frequently during this time lies atop Castel San Angelo in Rome. Built in 161 AD, it has served as a mausoleum, prison, papal residence, and is currently a museum. Atop San’Angelo stands St. Michael, the destroyer of the dragon. He is sheathing his sword in representation of the end of the plague in 590.
The arrows flow, yet the sword will be sheathed. Evil will be halted. The stories of these ancient totems and strength can give us strength as they remind us of the work that was done for centuries: pestilence, famine, war. The great killers never go away completely.
Fast forward to today
These medical specters serve as reminders of what makes the field of medicine so inspiring: the selfless acts, the fortitude of spirit, the healers, the long history, and the shoulders of giants we stand upon. From these stories, we spring the healing waters we bathe in to give us the courage to wake up and care for our patients each day. These specters encourage us to defeat any and all of the scourges that come our way.
I hear and read stories about the frontline heroes, the vaccine makers, the PPE creators, the health care workers, grocery store clerks, and teachers. I’m honored to hear of these stories and your sacrifices. I’m inspired to continue upholding your essence, your fight, and your stories. In keeping with ancient empire metaphors, you are taking the slings of the diseased arrows flying to our brethren as you try to keep yourself and others safe.
The sheathing of this sword will come. These arrows will be silenced. But until then, I lean on these pictures, these stories, and these saints, to give us all the strength to wake up each morning and continue healing.
They serve as reminders of what makes the field of medicine so great: the selfless acts, the fortitude of spirit, the healers, the long history, and the shoulders of giants we stand upon. From these stories spring the healing waters we bathe in to give us the courage to wake up and care for our patients each day and defeat any and all scourges that come our way.
So, who do you call in those late, quiet hours, when all seems lost?
Dr. Messler is the executive director, quality initiatives at Glytec and works as a hospitalist at Morton Plant Hospitalist group in Clearwater, Fla. This essay appeared initially on The Hospital Leader, the official blog of SHM.
I swear by Apollo Physician and Asclepius and Hygeia and Panacea and all the gods and goddesses, making them my witnesses, that I will fulfill according to my ability and judgment this oath and this covenant.
On my desk sits a bust of Hygeia, a mask from Venice, next to a small sculpture and a figurine of the plague doctor. Nearby, there is a Klimt closeup of Hygeia, a postcard portraying Asclepius, St. Sebastian paintings, and quotes from Maimonides. They whisper secrets and nod to the challenges of the past. These medical specters, ancient voices of the past, keep me grounded. They speak, listen, and elevate me, too. They bring life into my otherwise quiet room.
We all began our careers swearing to Apollo, Asclepius, Hygeia, and Panacea when we recited the Hippocratic Oath. I call upon them, and other gods and totems, and saints and ancient healers, now more than ever. As an atheist, I don’t appeal to them as prayers, but as Hippocrates intended. I look to their supernatural healing powers as a source of strength and as revealers of the natural and observable phenomena.
Apollo was one of the Twelve Olympians, a God of medicine, father of Asclepius. He was a healer, though his arrows also bore the plagues of the Gods.
For centuries, Apollo was found floating above the marble dissection table in the Bologna anatomical theater, guiding students who dove into the secrets of the human body.
Asclepius, son of Apollo, was hailed as a god of medicine. He healed many from plagues at his temples throughout the Ancient Greek and Roman empires. He was mentored in the healing arts by the centaur, Chiron. His many daughters and sons represent various aspects of medicine including cures, healing, recovery, sanitation, and beauty. To Asclepius, temples were places of healing, an ancient ancestor to modern hospitals.
Two of his daughters, Panacea and Hygeia, gave us the healing words of panacea and hygiene. Today, these acts of hygiene, handwashing, mask-wearing, and sanitation are discussed across the world louder than ever. While we’re all wishing for a panacea, we know it will take all the attributes of medicine to get us through this pandemic.
Hospitalists are part of the frontline teams facing this pandemic head-on. Gowning up for MRSA isolation seems quaint nowadays.
My attendings spoke of their fears, up against the unknown while on service in the 1980s, when HIV appeared. 2014 brought the Ebola biocontainment units. Now, this generation works daily against a modern plague, where every day is a risk of exposure. When every patient is in isolation, the garb begins to reflect the PPE that emerged during a 17th-century plague epidemics, the plague doctor outfit.
Godfather II fans recall the famous portrayal of the August 16th festival to San Rocco play out in the streets of New York. For those stricken with COVID-19 and recovered, you emulate San Rocco, in your continued return to service.
The Scuola Grande di San Rocco, in Venice, is the epitome of healing and greatness in one building. Tintoretto, the great Venetian painter, assembled the story of healing through art and portraits of San Rocco. The scuola, a confraternity, was a community of healers, gathered in one place to look after the less fortunate.
Hospitalists march into the hospital risking their lives. We always wear PPE for MRSA, ESBL, or C. diff. And enter reverse isolation rooms wearing N95s for possible TB cases. But those don’t elevate to the volume, to the same fear, as gowning up for COVID-19.
Hospitalists, frontline health care workers, embody the story of San Sebastian, another plague saint who absorbed the arrows, the symbolic plagues, onto his own shoulders so no one else had to bear them. San Sebastian was a Christian persecuted by a Roman emperor once his beliefs were discovered. He is often laden with arrows in spots where buboes would have appeared: the armpits and the groin. His sacrifice for others’ recovery became a symbol of absorbing the plague, the wounds, and the impact of the arrows.
This sacrifice epitomizes the daily work the frontline nurses, ER docs, intensivists, hospitalists, and the entire hospital staff perform daily, bearing the slung arrows of coronavirus.
One of the images I think of frequently during this time lies atop Castel San Angelo in Rome. Built in 161 AD, it has served as a mausoleum, prison, papal residence, and is currently a museum. Atop San’Angelo stands St. Michael, the destroyer of the dragon. He is sheathing his sword in representation of the end of the plague in 590.
The arrows flow, yet the sword will be sheathed. Evil will be halted. The stories of these ancient totems and strength can give us strength as they remind us of the work that was done for centuries: pestilence, famine, war. The great killers never go away completely.
Fast forward to today
These medical specters serve as reminders of what makes the field of medicine so inspiring: the selfless acts, the fortitude of spirit, the healers, the long history, and the shoulders of giants we stand upon. From these stories, we spring the healing waters we bathe in to give us the courage to wake up and care for our patients each day. These specters encourage us to defeat any and all of the scourges that come our way.
I hear and read stories about the frontline heroes, the vaccine makers, the PPE creators, the health care workers, grocery store clerks, and teachers. I’m honored to hear of these stories and your sacrifices. I’m inspired to continue upholding your essence, your fight, and your stories. In keeping with ancient empire metaphors, you are taking the slings of the diseased arrows flying to our brethren as you try to keep yourself and others safe.
The sheathing of this sword will come. These arrows will be silenced. But until then, I lean on these pictures, these stories, and these saints, to give us all the strength to wake up each morning and continue healing.
They serve as reminders of what makes the field of medicine so great: the selfless acts, the fortitude of spirit, the healers, the long history, and the shoulders of giants we stand upon. From these stories spring the healing waters we bathe in to give us the courage to wake up and care for our patients each day and defeat any and all scourges that come our way.
So, who do you call in those late, quiet hours, when all seems lost?
Dr. Messler is the executive director, quality initiatives at Glytec and works as a hospitalist at Morton Plant Hospitalist group in Clearwater, Fla. This essay appeared initially on The Hospital Leader, the official blog of SHM.
Mindful mentoring
Scenario
A GI faculty member is approached by two medical students who are planning careers in gastroenterology. They are interested in research projects and are very willing to dedicate the necessary time and energy. The faculty member is impressed by their desire and finds themselves recalling their own unsuccessful medical school search for a research mentor. Inspired by their enthusiasm and a desire to “give back,” the faculty member agrees to mentor them and helps them find suitable projects. Primarily because of the students’ hard work and fueled by their desire to produce results that will help their residency applications, the work progresses rapidly. Both students have separate abstracts accepted at a national meeting.
When COVID-19 hits, the faculty member is asked by their department to take on additional administrative and clinical work. They feel they cannot say no. Soon the faculty member finds it difficult to manage these new responsibilities on top of their many research projects, numerous clinical obligations, and additional pressures outside of work. They find they have no time for mentoring or even adequate sleep. Facing burnout, the faculty member is uncertain what to do for these hard-working and very gifted students. How would you recommend they manage their mentoring obligations?
Discussion
Mentorship is a cornerstone of academic medicine. In fact, it has been shown that academic clinicians who serve as mentors publish more papers, get more grants, are promoted faster, and are more likely to stay at their academic institutions with greater career satisfaction.1 However, not every mentor-mentee relationship is mutually beneficial. Usually, it’s the mentees that disproportionately suffer the consequences of a suboptimal relationship.2
Mentorship malpractice occurs when mentors’ behavior crosses a threshold that places the mentees’ success at risk.1,2 While the case above highlights a specific scenario where multiple issues are unfolding, the ability to recognize, address, and most importantly prevent mentorship malpractice ultimately benefits both mentees and mentors.
Understanding the various types of mentorship malpractice is helpful for prevention and course correction. As described by Chopra and colleagues, there are multiple types of passive and active mentorship malpractice.2 The passive forms are characterized by a lack of face-to-face meeting time with mentees and/or a lack of advocacy on the mentees’ behalf. Meanwhile, the active forms occur when the mentor exhibits self-serving behaviors. These can include listing themselves as first author on a mentee’s project or discouraging a mentee from working with other mentors. Mentors must be able to self-check, seek feedback from mentees, and encourage mentees to further their professional networks beyond the boundaries of what the mentor alone can offer. Doing so helps create new opportunities and helps ensure a mutually beneficial relationship.
A great initial step to prevent passive and active mentorship malpractice is to leverage the benefits of team mentorship.2,3 At its core, team mentorship capitalizes on the collective contributions of multiple mentors. Doing so not only provides security during uncertain times, but also allows for a diversity of perspectives, distribution of workload among mentors, and additional support for mentees.3,4 Team mentorship it is particularly important during this current global health crisis, and such an approach from the outset could have significantly improved the scenario above.
For the above scenario, likely a transition in mentorship would be needed. Such transitions, whether short term or long term, require transparency, honesty, and willingness to engage in difficult conversations with mentees. Whether the mentor in the above case engages another faculty to take on the mentees or chooses to find a colleague who will agree to take on other competing demands, it will require time, effort, and energy – all of which are in short supply. When team mentorship is established from the outset, such transitions of mentorship can occur seamlessly and with more ease for all.
Additional considerations for successful mentoring of medical students or early-career physicians include understanding generational differences between the mentor and their mentees. As outlined by Waljee and colleagues, the next generation of trainees and physicians may act in ways that deviate from the norms of academic medicine’s tradition. As a mentor, it is imperative to understand these actions are not intended to disrupt the traditions and norms of health systems.5 For example, the use of technology during rounds can often be misconstrued as disrespectful. However, the underlying intent in many cases is to answer a question or access a helpful reference.
Seeing behavior and actions from the perspective of the mentee is one of the many ways to support and sustain successful mentoring relationships. A mindful approach benefits both mentees and mentors; this includes reflecting on the underlying motives for mentorship and cultivating gratitude for the relationships formed.6 While these steps may seem trivial, gratitude promotes happiness, trust, motivation, and respect. It can be felt by others, including mentees.
As mentors continue to shape the future, they have an ethical obligation to care for themselves, in addition to their mentees. In addition to avoiding mentorship malpractice, engaging in team mentorship, and incorporating mindful mentoring, an emphasis on self-care is critical.7 Taking time to recharge is essential. It allows one to be fully present, while also setting an example for the mentee. Explicitly addressing self-care for both mentor and mentee is a part of mindful mentorship, with benefits for all.6
Three key points:
1. Awareness of mentorship malpractice
2. Importance of team mentorship
3. Benefits of mindful mentorship
Mr. Rodoni is with the University of Michigan Medical School and Stephen M. Ross School of Business, Ann Arbor, Mich. Dr. Fessel is a professor of radiology in the department of radiology at Michigan Medicine, Ann Arbor. They reported having no disclosures relevant to this article.
References:
1. Chopra V et al. JAMA Intern Med. 2018 Feb;178:175-6.
2. Chopra V et al. JAMA. 2016 Apr 12;315:1453-4.
3. Chopra V et al. The Mentoring Guide: Helping Mentors & Mentees Succeed. Ann Arbor: Michigan Publishing, 2019.
4. Rodoni BM et al. Annals of Surgery. 2020 Aug;272(2):e151-2.
5. Waljee JF et al. JAMA. 2018 Apr 17;319(15):1547-8.
6. Chopra V and Saint S. Healthc (Amst). 2020 Mar;8(1):100390.
7. Fessell D et al. “Mentoring During a Crisis.” Harvard Business Review. 2020 Oct 29.
Scenario
A GI faculty member is approached by two medical students who are planning careers in gastroenterology. They are interested in research projects and are very willing to dedicate the necessary time and energy. The faculty member is impressed by their desire and finds themselves recalling their own unsuccessful medical school search for a research mentor. Inspired by their enthusiasm and a desire to “give back,” the faculty member agrees to mentor them and helps them find suitable projects. Primarily because of the students’ hard work and fueled by their desire to produce results that will help their residency applications, the work progresses rapidly. Both students have separate abstracts accepted at a national meeting.
When COVID-19 hits, the faculty member is asked by their department to take on additional administrative and clinical work. They feel they cannot say no. Soon the faculty member finds it difficult to manage these new responsibilities on top of their many research projects, numerous clinical obligations, and additional pressures outside of work. They find they have no time for mentoring or even adequate sleep. Facing burnout, the faculty member is uncertain what to do for these hard-working and very gifted students. How would you recommend they manage their mentoring obligations?
Discussion
Mentorship is a cornerstone of academic medicine. In fact, it has been shown that academic clinicians who serve as mentors publish more papers, get more grants, are promoted faster, and are more likely to stay at their academic institutions with greater career satisfaction.1 However, not every mentor-mentee relationship is mutually beneficial. Usually, it’s the mentees that disproportionately suffer the consequences of a suboptimal relationship.2
Mentorship malpractice occurs when mentors’ behavior crosses a threshold that places the mentees’ success at risk.1,2 While the case above highlights a specific scenario where multiple issues are unfolding, the ability to recognize, address, and most importantly prevent mentorship malpractice ultimately benefits both mentees and mentors.
Understanding the various types of mentorship malpractice is helpful for prevention and course correction. As described by Chopra and colleagues, there are multiple types of passive and active mentorship malpractice.2 The passive forms are characterized by a lack of face-to-face meeting time with mentees and/or a lack of advocacy on the mentees’ behalf. Meanwhile, the active forms occur when the mentor exhibits self-serving behaviors. These can include listing themselves as first author on a mentee’s project or discouraging a mentee from working with other mentors. Mentors must be able to self-check, seek feedback from mentees, and encourage mentees to further their professional networks beyond the boundaries of what the mentor alone can offer. Doing so helps create new opportunities and helps ensure a mutually beneficial relationship.
A great initial step to prevent passive and active mentorship malpractice is to leverage the benefits of team mentorship.2,3 At its core, team mentorship capitalizes on the collective contributions of multiple mentors. Doing so not only provides security during uncertain times, but also allows for a diversity of perspectives, distribution of workload among mentors, and additional support for mentees.3,4 Team mentorship it is particularly important during this current global health crisis, and such an approach from the outset could have significantly improved the scenario above.
For the above scenario, likely a transition in mentorship would be needed. Such transitions, whether short term or long term, require transparency, honesty, and willingness to engage in difficult conversations with mentees. Whether the mentor in the above case engages another faculty to take on the mentees or chooses to find a colleague who will agree to take on other competing demands, it will require time, effort, and energy – all of which are in short supply. When team mentorship is established from the outset, such transitions of mentorship can occur seamlessly and with more ease for all.
Additional considerations for successful mentoring of medical students or early-career physicians include understanding generational differences between the mentor and their mentees. As outlined by Waljee and colleagues, the next generation of trainees and physicians may act in ways that deviate from the norms of academic medicine’s tradition. As a mentor, it is imperative to understand these actions are not intended to disrupt the traditions and norms of health systems.5 For example, the use of technology during rounds can often be misconstrued as disrespectful. However, the underlying intent in many cases is to answer a question or access a helpful reference.
Seeing behavior and actions from the perspective of the mentee is one of the many ways to support and sustain successful mentoring relationships. A mindful approach benefits both mentees and mentors; this includes reflecting on the underlying motives for mentorship and cultivating gratitude for the relationships formed.6 While these steps may seem trivial, gratitude promotes happiness, trust, motivation, and respect. It can be felt by others, including mentees.
As mentors continue to shape the future, they have an ethical obligation to care for themselves, in addition to their mentees. In addition to avoiding mentorship malpractice, engaging in team mentorship, and incorporating mindful mentoring, an emphasis on self-care is critical.7 Taking time to recharge is essential. It allows one to be fully present, while also setting an example for the mentee. Explicitly addressing self-care for both mentor and mentee is a part of mindful mentorship, with benefits for all.6
Three key points:
1. Awareness of mentorship malpractice
2. Importance of team mentorship
3. Benefits of mindful mentorship
Mr. Rodoni is with the University of Michigan Medical School and Stephen M. Ross School of Business, Ann Arbor, Mich. Dr. Fessel is a professor of radiology in the department of radiology at Michigan Medicine, Ann Arbor. They reported having no disclosures relevant to this article.
References:
1. Chopra V et al. JAMA Intern Med. 2018 Feb;178:175-6.
2. Chopra V et al. JAMA. 2016 Apr 12;315:1453-4.
3. Chopra V et al. The Mentoring Guide: Helping Mentors & Mentees Succeed. Ann Arbor: Michigan Publishing, 2019.
4. Rodoni BM et al. Annals of Surgery. 2020 Aug;272(2):e151-2.
5. Waljee JF et al. JAMA. 2018 Apr 17;319(15):1547-8.
6. Chopra V and Saint S. Healthc (Amst). 2020 Mar;8(1):100390.
7. Fessell D et al. “Mentoring During a Crisis.” Harvard Business Review. 2020 Oct 29.
Scenario
A GI faculty member is approached by two medical students who are planning careers in gastroenterology. They are interested in research projects and are very willing to dedicate the necessary time and energy. The faculty member is impressed by their desire and finds themselves recalling their own unsuccessful medical school search for a research mentor. Inspired by their enthusiasm and a desire to “give back,” the faculty member agrees to mentor them and helps them find suitable projects. Primarily because of the students’ hard work and fueled by their desire to produce results that will help their residency applications, the work progresses rapidly. Both students have separate abstracts accepted at a national meeting.
When COVID-19 hits, the faculty member is asked by their department to take on additional administrative and clinical work. They feel they cannot say no. Soon the faculty member finds it difficult to manage these new responsibilities on top of their many research projects, numerous clinical obligations, and additional pressures outside of work. They find they have no time for mentoring or even adequate sleep. Facing burnout, the faculty member is uncertain what to do for these hard-working and very gifted students. How would you recommend they manage their mentoring obligations?
Discussion
Mentorship is a cornerstone of academic medicine. In fact, it has been shown that academic clinicians who serve as mentors publish more papers, get more grants, are promoted faster, and are more likely to stay at their academic institutions with greater career satisfaction.1 However, not every mentor-mentee relationship is mutually beneficial. Usually, it’s the mentees that disproportionately suffer the consequences of a suboptimal relationship.2
Mentorship malpractice occurs when mentors’ behavior crosses a threshold that places the mentees’ success at risk.1,2 While the case above highlights a specific scenario where multiple issues are unfolding, the ability to recognize, address, and most importantly prevent mentorship malpractice ultimately benefits both mentees and mentors.
Understanding the various types of mentorship malpractice is helpful for prevention and course correction. As described by Chopra and colleagues, there are multiple types of passive and active mentorship malpractice.2 The passive forms are characterized by a lack of face-to-face meeting time with mentees and/or a lack of advocacy on the mentees’ behalf. Meanwhile, the active forms occur when the mentor exhibits self-serving behaviors. These can include listing themselves as first author on a mentee’s project or discouraging a mentee from working with other mentors. Mentors must be able to self-check, seek feedback from mentees, and encourage mentees to further their professional networks beyond the boundaries of what the mentor alone can offer. Doing so helps create new opportunities and helps ensure a mutually beneficial relationship.
A great initial step to prevent passive and active mentorship malpractice is to leverage the benefits of team mentorship.2,3 At its core, team mentorship capitalizes on the collective contributions of multiple mentors. Doing so not only provides security during uncertain times, but also allows for a diversity of perspectives, distribution of workload among mentors, and additional support for mentees.3,4 Team mentorship it is particularly important during this current global health crisis, and such an approach from the outset could have significantly improved the scenario above.
For the above scenario, likely a transition in mentorship would be needed. Such transitions, whether short term or long term, require transparency, honesty, and willingness to engage in difficult conversations with mentees. Whether the mentor in the above case engages another faculty to take on the mentees or chooses to find a colleague who will agree to take on other competing demands, it will require time, effort, and energy – all of which are in short supply. When team mentorship is established from the outset, such transitions of mentorship can occur seamlessly and with more ease for all.
Additional considerations for successful mentoring of medical students or early-career physicians include understanding generational differences between the mentor and their mentees. As outlined by Waljee and colleagues, the next generation of trainees and physicians may act in ways that deviate from the norms of academic medicine’s tradition. As a mentor, it is imperative to understand these actions are not intended to disrupt the traditions and norms of health systems.5 For example, the use of technology during rounds can often be misconstrued as disrespectful. However, the underlying intent in many cases is to answer a question or access a helpful reference.
Seeing behavior and actions from the perspective of the mentee is one of the many ways to support and sustain successful mentoring relationships. A mindful approach benefits both mentees and mentors; this includes reflecting on the underlying motives for mentorship and cultivating gratitude for the relationships formed.6 While these steps may seem trivial, gratitude promotes happiness, trust, motivation, and respect. It can be felt by others, including mentees.
As mentors continue to shape the future, they have an ethical obligation to care for themselves, in addition to their mentees. In addition to avoiding mentorship malpractice, engaging in team mentorship, and incorporating mindful mentoring, an emphasis on self-care is critical.7 Taking time to recharge is essential. It allows one to be fully present, while also setting an example for the mentee. Explicitly addressing self-care for both mentor and mentee is a part of mindful mentorship, with benefits for all.6
Three key points:
1. Awareness of mentorship malpractice
2. Importance of team mentorship
3. Benefits of mindful mentorship
Mr. Rodoni is with the University of Michigan Medical School and Stephen M. Ross School of Business, Ann Arbor, Mich. Dr. Fessel is a professor of radiology in the department of radiology at Michigan Medicine, Ann Arbor. They reported having no disclosures relevant to this article.
References:
1. Chopra V et al. JAMA Intern Med. 2018 Feb;178:175-6.
2. Chopra V et al. JAMA. 2016 Apr 12;315:1453-4.
3. Chopra V et al. The Mentoring Guide: Helping Mentors & Mentees Succeed. Ann Arbor: Michigan Publishing, 2019.
4. Rodoni BM et al. Annals of Surgery. 2020 Aug;272(2):e151-2.
5. Waljee JF et al. JAMA. 2018 Apr 17;319(15):1547-8.
6. Chopra V and Saint S. Healthc (Amst). 2020 Mar;8(1):100390.
7. Fessell D et al. “Mentoring During a Crisis.” Harvard Business Review. 2020 Oct 29.
Medical professional liability risk and mitigation: An overview for early-career gastroenterologists
Disclaimer: This article is for educational purposes only. All examples are hypothetical and aim to illustrate common clinical scenarios and challenges gastroenterologists may encounter within their scope of practice. The content herein should not be interpreted as legal advice for individual cases nor a substitute for seeking the advice of an attorney.
There are unique potential stressors faced by the gastroenterologist at each career stage, some more so early on. One such stressor, and one particularly important in a procedure-intensive specialty like GI, is medical professional liability (MPL), historically termed “medical malpractice.” Between 2009 and 2018, GI was the second-highest internal medicine subspecialty in both MPL claims made and claims paid,1 yet instruction on MPL risk and mitigation is scarce in fellowship, as is the available GI-related literature on the topic. This scarcity may generate untoward stress and unnecessarily expose gastroenterologists to avoidable MPL pitfalls. Therefore, it is vital for GI trainees, early-career gastroenterologists, and even seasoned gastroenterologists to have a working and updated knowledge of the general principles of MPL and GI-specific considerations. Such understanding can help preserve physician well-being, increase professional satisfaction, strengthen the doctor-patient relationship, and improve health care outcomes.2
To this end, we herein provide a focused review of the following: key MPL concepts, trends in MPL claims, GI-related MPL risk scenarios and considerations, adverse provider defensive mechanisms, documentation tenets, challenges posed by telemedicine, and the concept of “vicarious liability.”
Key MPL concepts
MPL falls under the umbrella of tort law, which itself falls under the umbrella of civil law; that is, civil (as opposed to criminal) justice governs torts – including but not limited to MPL claims – as well as other areas of law concerning noncriminal injury.3 A “tort” is a “civil wrong that unfairly causes another to experience loss or harm resulting in legal liability.”3 MPL claims assert the tort of negligence (similar to the concept of “incompetence”) and endeavor to compensate the harmed patient/individual while simultaneously dissuading suboptimal medical care by the provider in the future.4,5 A successful MPL claim must prove four overlapping elements: that the tortfeasor (here, the gastroenterologist) owed a duty of care to the injured party and breached that duty, which caused damages.6 Given that MPL cases exist within tort law rather than criminal law, the burden of proof for these cases is not “beyond a reasonable doubt”; instead, it’s “to a reasonable medical probability.”7
Trends in MPL claims
According to data compiled by the MPL Association, 278,220 MPL claims were made in the United States from 1985 to 2012.3,8-10 Among these, 1.8% involved gastroenterologists, which puts it at 17th place out of the 20 specialties surveyed.9 While the number of paid claims over this time frame decreased in GI by 34.6% (from 18.5 to 12.1 cases per 1,000 physician-years), there was a concurrent 23.3% increase in average claim compensation; essentially, there were fewer paid GI-related claims but there were higher payouts per paid claim.11,12 From 2009 to 2018, average legal defense costs for paid GI-related claims were $97,392, and average paid amount was $330,876.1
GI-related MPL risk scenarios and considerations
Many MPL claims relate to situations involving medical errors or adverse events (AEs), be they procedural or nonprocedural. However other aspects of GI also carry MPL risk.

Informed consent
MPL claims may be made not only on the grounds of inadequately informed consent but also inadequately informed refusal.5,13,14 While standards for adequate informed consent vary by state, most states apply the “reasonable patient standard,” i.e., assuming an average patient with enough information to be an active participant in the medical decision-making process. Generally, informed consent should ensure that the patient understands the nature of the procedure/treatment being proposed, there is a discussion of the risks and benefits of undergoing and not undergoing the procedure/treatment, reasonable alternatives are presented, the risks and benefits associated with these alternatives are discussed, and the patient’s comprehension of these things is assessed (Figure).15 Additionally, informed consent should be tailored to each patient and GI procedure/treatment on a case-by-case basis rather than using a one-size-fits-all approach. Moreover, documentation of the patient’s understanding of the (tailored) information provided can concurrently improve quality of the consent and potentially decrease MPL risk (Figure).16
Endoscopic procedures
Procedure-related MPL claims represent approximately 25% of all GI-related claims (8,17). Among these, 52% involve colonoscopy, 16% involve endoscopic retrograde cholangiopancreatography (ERCP), and 11% involve esophagogastroduodenoscopy.8 Albeit generally safe, colonoscopy, as with esophagogastroduodenoscopy, is subject to rare but serious AEs.18,19 Risk of these AEs may be accentuated in certain scenarios (such as severe colonic inflammation or coagulopathy) and, as discussed earlier, may merit tailored informed consent. Regardless of the procedure, in the event of postprocedural development of signs/symptoms (such as tachycardia, fever, chest or abdominal discomfort, or hypotension) indicating a potential AE, stabilizing measures and evaluation (such as blood work and imaging) should be undertaken, and hospital admission (if not already hospitalized) should be considered until discharge is deemed safe.19
ERCP-related MPL claims, for many years, have had the highest average compensation of any GI procedure.11 Though discussion of advanced procedures is beyond the scope of this article, it is worth mentioning the observation that most of such claims involve an allegation that the procedure was not indicated (for example, that it was performed based on inadequate evidence of pancreatobiliary pathology), or was for diagnostic purposes (for example, being done instead of noninvasive imaging) rather than therapeutic.20-23 This emphasizes the importance of appropriate procedure indications.
Percutaneous endoscopic gastrostomy (PEG) placement merits special mention given it can be complicated by ethical challenges (for example, needing a surrogate decision-maker’s consent or representing medical futility) and has a relatively high potential for MPL claims. PEG placement carries a low AE rate (0.1%-1%), but these AEs may result in high morbidity/mortality, in part because of the underlying comorbidities of patients needing PEG placement.24,25 Also, timing of a patient’s demise may coincide with PEG placement, thereby prompting (possibly unfounded) perceptions of causality.24-27 Therefore, such scenarios merit unique additional preprocedure safeguards. For instance, for patients lacking capacity to provide informed consent, especially when family members may differ on whether PEG should be placed, it is advisable to ask the family to select one surrogate decision-maker (if there’s no advance directive) to whom the gastroenterologist should discuss both the risks, benefits, and goals of PEG placement in the context of the patient’s overall clinical trajectory/life expectancy and the need for consent (or refusal) based on what the patient would have wished. In addition, having a medical professional witness this discussion may be useful.27
Antithrombotic agents
Periprocedural management of antithrombotics, including anticoagulants and antiplatelets, can pose challenges for the gastroenterologist. While clinical practice guidelines exist to guide decision-making in this regard, the variables involved may extend beyond the expertise of the gastroenterologist.28 For instance, in addition to the procedural risk for bleeding, the indication for antithrombotic therapy, risk of a thrombotic event, duration of action of the antithrombotic, and available bridging options should all be considered according to recommendations.28,29 While requiring more time on the part of the gastroenterologist, the optimal periprocedural management of antithrombotic agents would usually involve discussion with the provider managing antithrombotic therapy to best conduct a risk-benefit assessment regarding if (and how long) the antithrombotic therapy should be held (Figure). This shared decision-making, which should also include the patient, may help decrease MPL risk and improve outcomes.
Provider defense mechanisms
Physicians may engage in various defensive behaviors in an attempt to mitigate MPL risk; however, these behaviors may, paradoxically, increase risk.30,31
Assurance behaviors
Assurance behaviors refer to the practice of recommending or performing additional services (such as medications, imaging, procedures, and referrals) that are not clearly indicated.2,30,31 Assurance behaviors are driven by fear of MPL risk and/or missing a potential diagnosis. Recent studies have estimated that more than 50% of gastroenterologists worldwide have performed additional invasive procedures without clear indications, and that nearly one-third of endoscopic procedures annually have questionable indications.30,32 While assurance behaviors may seem likely to decrease MPL risk, overall, they may inadvertently increase AE and MPL risk, as well as health care expenditures.3,30,32
Avoidance behaviors
Avoidance behaviors refer to providers avoiding participation in potentially high-risk clinical interventions (for example, the actual procedures), including those for which they are credentialed/certified proficient.30,31 Two clinical scenarios that illustrate this behavior include the following: An advanced endoscopist credentialed to perform ERCP might refer a “high-risk” elderly patient with cholangitis to another provider to perform said ERCP or for percutaneous transhepatic drainage (in the absence of a clear benefit to such), or a gastroenterologist might refer a patient to interventional gastroenterology for resection of a large polyp even though gastroenterologists are usually proficient in this skill and may feel comfortable performing the resection themselves. Avoidance behaviors are driven by a fear of MPL risk and can have several negative consequences.33 For example, patients may not receive indicated interventions. Additionally, patients may have to wait longer for an intervention because they are referred to another provider, which also increases potential for loss to follow-up.2,30,31 This may be viewed as noncompliance with the standard of care, among other hazards, thereby increasing MPL risk.
Documentation tenets
Thorough documentation can decrease MPL risk, especially since it is often used as legal evidence.16 Documenting, for instance, preprocedure discussion of potential risk of AEs (such as bleeding or perforation) or procedural failure (for example, missed lesions)can protect gastroenterologists (Figure).16 While, as discussed previously, these should be covered in the informed consent process (which itself reduces MPL risk), proof of compliance in providing adequate informed consent must come in the form of documentation that indicates that the process took place and specifically what topics were discussed therein. MPL risk may be further decreased by documenting steps taken during a procedure and anatomic landmarks encountered to offer proof of technical competency and compliance with standards of care (Figure).16,34 In this context, it is worth recalling the adage: “If it’s not documented, it did not occur.”
Curbside consults versus consultation
Also germane here is the topic of whether documentation is needed for “curbside consults.” The uncertainty is, in part, semantic; that is, at what point does a “curbside” become a consultation? A curbside is a general question or query (such as anything that could also be answered by searching the Internet or reference materials) in response to which information is provided; once it involves provision of medical advice for a specific patient (for example, when patient identifiers have been shared or their EHR has been accessed), it constitutes a consultation. Based on these definitions, a curbside need not be documented, whereas a consultation – even if seemingly trivial – should be.
Consideration of language and cultural factors
Language barriers should be considered when the gastroenterologist is communicating with the patient, and such efforts, whenever made, should be documented to best protect against MPL.16,35 These considerations arise not only during the consent process but when obtaining a history, providing postprocedure instructions, and during follow-ups. To this end, 24/7 telephone interpreter services may assist the gastroenterologist (when one is communicating with non–English speakers and is not medically certified in the patient’s native/preferred language) and strengthen trust in the provider-patient relationship.36 Additionally, written materials (such as consent forms, procedural information) in patients’ native/preferred languages should be provided, when available, to enhance patient understanding and participation in care (Figure).35
Challenges posed by telemedicine
The COVID-19 pandemic has rapidly led to more virtual encounters. While increased utilization of telemedicine platforms may make health care more accessible, it does not lessen the clinicians’ duty to patients and may actually expose them to greater MPL risk.18,37,38 Therefore, the provider must be cognizant of two key principles to mitigate MPL risk in the context of telemedicine encounters. First, the same standard of care applies to virtual and in-person encounters.18,37,38 Second, patient privacy and HIPAA regulations are not waived during telemedicine encounters, and breaches of such may result in an MPL claim.18,37,38
With regard to the first principle, for patients who have not been physically examined (such as when a telemedicine visit was substituted for an in-person clinic encounter), gastroenterologists should not overlook requesting timely preprocedure anesthesia consultation or obtaining additional laboratory studies as needed to ensure safety and the same standard of care. Moreover, particularly in the context of pandemic-related decreased procedural capacity, triaging procedures can be especially challenging. Standardized institutional criteria which prioritize certain diagnoses/conditions over others, leaving room for justifiable exceptions, are advisable.
Vicarious liability
“Vicarious liability” is defined as that extending to persons who have not committed a wrong but on whose behalf wrongdoers acted.39 Therefore, gastroenterologists may be liable not only for their own actions but also for those of personnel they supervise (such as fellow trainees and non–physician practitioners).39 Vicarious liability aims to ensure that systemic checks and balances are in place so that, if failure occurs, harm can still be mitigated and/or avoided, as illustrated by Reason’s “Swiss Cheese Model.”40
Conclusion
Any gastroenterologist can experience an MPL claim. Such an experience can be especially stressful and confusing to early-career clinicians, especially if they’re unfamiliar with legal proceedings. Although MPL principles are not often taught in medical school or residency, it is important for gastroenterologists to be informed regarding tenets of MPL and cognizant of clinical situations which have relatively higher MPL risk. This can assuage untoward angst regarding MPL and highlight proactive risk-mitigation strategies. In general, gastroenterologist practices that can mitigate MPL risk include effective communication; adequate informed consent/refusal; documentation of preprocedure counseling, periprocedure events, and postprocedure recommendations; and maintenance of proper certification and privileging.
Dr. Azizian and Dr. Dalai are with the University of California, Los Angeles and the department of medicine at Olive View–UCLA Medical Center, Sylmar, Calif. They are co–first authors of this paper. Dr. Dalai is also with the division of gastroenterology at the University of New Mexico, Albuquerque. Dr. Adams is with the Center for Clinical Management Research in Veterans Affairs Ann Arbor Healthcare System, the division of gastroenterology at the University of Michigan Health System, and the Institute for Healthcare Policy and Innovation, all in Ann Arbor, Mich. Dr. Tabibian is with UCLA and the division of gastroenterology at Olive View–UCLA Medical Center. The authors have no conflicts of interest.
References
1. 2020 Data Sharing Project Gastroenterology 2009-2018. Inside Medical Liability: Second Quarter. Accessed 2020 Dec 6.
2. Mello MM et al. Health Aff (Millwood). 2004 Jul-Aug;23(4):42-53.
3. Adams MA et al. JAMA. 2014 Oct;312(13):1348-9.
4. Pegalis SE. American Law of Medical Malpractice 3d, Vol. 2. St. Paul, Minn.: Thomson Reuters, 2005.
5. Feld LD et al. Am J Gastroenterol. 2018 Nov;113(11):1577-9.
6. Sawyer v. Wight, 196 F. Supp. 2d 220, 226 (E.D.N.Y. 2002).
7. Michael A. Sita v. Long Island Jewish-Hillside Medical Center, 22 A.D.3d 743 (N.Y. App. Div. 2005).
8. Conklin LS et al. Clin Gastroenterol Hepatol. 2008 Jun;6(6):677-81.
9. Jena AB et al. N Engl J Med. 2011 Aug 18;365(7):629-36.
10. Kane CK. “Policy Research Perspectives Medical Liability Claim Frequency: A 2007-2008 Snapshot of Physicians.” Chicago: American Medical Association, 2010.
11. Hernandez LV et al. World J Gastrointest Endosc. 2013 Apr 16;5(4):169-73.
12. Schaffer AC et al. JAMA Intern Med. 2017 May 1;177(5):710-8.
13. Natanson v. Kline, 186 Kan. 393, 409, 350 P.2d 1093, 1106, decision clarified on denial of reh’g, 187 Kan. 186, 354 P.2d 670 (1960).
14. Truman v. Thomas, 27 Cal. 3d 285, 292, 611 P.2d 902, 906 (1980).
15. Shah P et al. Informed Consent, in “StatPearls.” Treasure Island, Fla.: StatPearls Publishing, 2020 Jan. Updated 2020 Aug 22.
16. Rex DK. Clin Gastroenterol Hepatol. 2013 Jul;11(7):768-73.
17. Gerstenberger PD, Plumeri PA. Gastrointest Endosc. Mar-Apr 1993;39(2):132-8.
18. Adams MA and Allen JI. Clin Gastroenterol Hepatol. 2019 Nov;17(12):2392-6.e1.
19. Ahlawat R et al. Esophagogastroduodenoscopy, in “StatPearls.” Treasure Island, Fla.: StatPearls Publishing, 2020 Jan. Updated 2020 Dec 9.
20. Cotton PB. Gastrointest Endosc. 2006 Mar;63(3):378-82.
21. Cotton PB. Gastrointest Endosc. 2010 Oct;72(4):904.
22. Adamson TE et al. West J Med. 1989 Mar;150(3):356-60.
23. Trap R et al. Endoscopy. 1999 Feb;31(2):125-30.
24. Funaki B. Semin Intervent Radiol. 2015 Mar;32(1):61-4.
25. Feeding Tube Nursing Home and Hospital Malpractice. Miller & Zois, Attorneys at Law. Accessed 2020 Jun 20.
26. Medical Malpractice Lawsuit Brings $750,000 Settlement: Death of 82-year-old woman from sepsis due to improper placement of feeding tube. Lubin & Meyers PC. Accessed 2020 Jun 20.
27. Brendel RW et al. Med Clin North Am. 2010 Nov;94(6):1229-40, xi-ii.
28. ASGE Standards of Practice Committee; Acosta RD et al. Gastrointest Endosc. 2016 Jan;83(1):3-16.
29. Saleem S and Thomas AL. Cureus. 2018 Jun 25;10(6):e2878.
30. Hiyama T et al. World J Gastroenterol. 2006 Dec 21;12(47):7671-5.
31. Studdert DM et al. JAMA. 2005 Jun 1;293(21):2609-17.
32. Shaheen NJ et al. Gastroenterology. 2018 May;154(7):1993-2003.
33. Oza VM et al. Clin Gastroenterol Hepatol. 2016 Feb;14(2):172-4.
34. Feld AD. Gastrointest Endosc Clin N Am. 2002 Jan;12(1):171-9, viii-ix.
35. Lee JS et al. J Gen Intern Med. 2017 Aug;32(8):863-70.
36. Forrow L and Kontrimas JC. J Gen Intern Med. 2017 Aug;32(8):855-7.
37. Moses RE et al. Am J Gastroenterol. 2014 Aug;109(8):1128-32.
38. Tabibian JH. “The Evolution of Telehealth.” Guidepoint: Legal Solutions Blog. Accessed 2020 Aug 12.
39. Feld AD. Am J Gastroenterol. 2004 Sep;99(9):1641-4.
40. Reason J. BMJ. 2000;320(7237):768‐70.
Disclaimer: This article is for educational purposes only. All examples are hypothetical and aim to illustrate common clinical scenarios and challenges gastroenterologists may encounter within their scope of practice. The content herein should not be interpreted as legal advice for individual cases nor a substitute for seeking the advice of an attorney.
There are unique potential stressors faced by the gastroenterologist at each career stage, some more so early on. One such stressor, and one particularly important in a procedure-intensive specialty like GI, is medical professional liability (MPL), historically termed “medical malpractice.” Between 2009 and 2018, GI was the second-highest internal medicine subspecialty in both MPL claims made and claims paid,1 yet instruction on MPL risk and mitigation is scarce in fellowship, as is the available GI-related literature on the topic. This scarcity may generate untoward stress and unnecessarily expose gastroenterologists to avoidable MPL pitfalls. Therefore, it is vital for GI trainees, early-career gastroenterologists, and even seasoned gastroenterologists to have a working and updated knowledge of the general principles of MPL and GI-specific considerations. Such understanding can help preserve physician well-being, increase professional satisfaction, strengthen the doctor-patient relationship, and improve health care outcomes.2
To this end, we herein provide a focused review of the following: key MPL concepts, trends in MPL claims, GI-related MPL risk scenarios and considerations, adverse provider defensive mechanisms, documentation tenets, challenges posed by telemedicine, and the concept of “vicarious liability.”
Key MPL concepts
MPL falls under the umbrella of tort law, which itself falls under the umbrella of civil law; that is, civil (as opposed to criminal) justice governs torts – including but not limited to MPL claims – as well as other areas of law concerning noncriminal injury.3 A “tort” is a “civil wrong that unfairly causes another to experience loss or harm resulting in legal liability.”3 MPL claims assert the tort of negligence (similar to the concept of “incompetence”) and endeavor to compensate the harmed patient/individual while simultaneously dissuading suboptimal medical care by the provider in the future.4,5 A successful MPL claim must prove four overlapping elements: that the tortfeasor (here, the gastroenterologist) owed a duty of care to the injured party and breached that duty, which caused damages.6 Given that MPL cases exist within tort law rather than criminal law, the burden of proof for these cases is not “beyond a reasonable doubt”; instead, it’s “to a reasonable medical probability.”7
Trends in MPL claims
According to data compiled by the MPL Association, 278,220 MPL claims were made in the United States from 1985 to 2012.3,8-10 Among these, 1.8% involved gastroenterologists, which puts it at 17th place out of the 20 specialties surveyed.9 While the number of paid claims over this time frame decreased in GI by 34.6% (from 18.5 to 12.1 cases per 1,000 physician-years), there was a concurrent 23.3% increase in average claim compensation; essentially, there were fewer paid GI-related claims but there were higher payouts per paid claim.11,12 From 2009 to 2018, average legal defense costs for paid GI-related claims were $97,392, and average paid amount was $330,876.1
GI-related MPL risk scenarios and considerations
Many MPL claims relate to situations involving medical errors or adverse events (AEs), be they procedural or nonprocedural. However other aspects of GI also carry MPL risk.

Informed consent
MPL claims may be made not only on the grounds of inadequately informed consent but also inadequately informed refusal.5,13,14 While standards for adequate informed consent vary by state, most states apply the “reasonable patient standard,” i.e., assuming an average patient with enough information to be an active participant in the medical decision-making process. Generally, informed consent should ensure that the patient understands the nature of the procedure/treatment being proposed, there is a discussion of the risks and benefits of undergoing and not undergoing the procedure/treatment, reasonable alternatives are presented, the risks and benefits associated with these alternatives are discussed, and the patient’s comprehension of these things is assessed (Figure).15 Additionally, informed consent should be tailored to each patient and GI procedure/treatment on a case-by-case basis rather than using a one-size-fits-all approach. Moreover, documentation of the patient’s understanding of the (tailored) information provided can concurrently improve quality of the consent and potentially decrease MPL risk (Figure).16
Endoscopic procedures
Procedure-related MPL claims represent approximately 25% of all GI-related claims (8,17). Among these, 52% involve colonoscopy, 16% involve endoscopic retrograde cholangiopancreatography (ERCP), and 11% involve esophagogastroduodenoscopy.8 Albeit generally safe, colonoscopy, as with esophagogastroduodenoscopy, is subject to rare but serious AEs.18,19 Risk of these AEs may be accentuated in certain scenarios (such as severe colonic inflammation or coagulopathy) and, as discussed earlier, may merit tailored informed consent. Regardless of the procedure, in the event of postprocedural development of signs/symptoms (such as tachycardia, fever, chest or abdominal discomfort, or hypotension) indicating a potential AE, stabilizing measures and evaluation (such as blood work and imaging) should be undertaken, and hospital admission (if not already hospitalized) should be considered until discharge is deemed safe.19
ERCP-related MPL claims, for many years, have had the highest average compensation of any GI procedure.11 Though discussion of advanced procedures is beyond the scope of this article, it is worth mentioning the observation that most of such claims involve an allegation that the procedure was not indicated (for example, that it was performed based on inadequate evidence of pancreatobiliary pathology), or was for diagnostic purposes (for example, being done instead of noninvasive imaging) rather than therapeutic.20-23 This emphasizes the importance of appropriate procedure indications.
Percutaneous endoscopic gastrostomy (PEG) placement merits special mention given it can be complicated by ethical challenges (for example, needing a surrogate decision-maker’s consent or representing medical futility) and has a relatively high potential for MPL claims. PEG placement carries a low AE rate (0.1%-1%), but these AEs may result in high morbidity/mortality, in part because of the underlying comorbidities of patients needing PEG placement.24,25 Also, timing of a patient’s demise may coincide with PEG placement, thereby prompting (possibly unfounded) perceptions of causality.24-27 Therefore, such scenarios merit unique additional preprocedure safeguards. For instance, for patients lacking capacity to provide informed consent, especially when family members may differ on whether PEG should be placed, it is advisable to ask the family to select one surrogate decision-maker (if there’s no advance directive) to whom the gastroenterologist should discuss both the risks, benefits, and goals of PEG placement in the context of the patient’s overall clinical trajectory/life expectancy and the need for consent (or refusal) based on what the patient would have wished. In addition, having a medical professional witness this discussion may be useful.27
Antithrombotic agents
Periprocedural management of antithrombotics, including anticoagulants and antiplatelets, can pose challenges for the gastroenterologist. While clinical practice guidelines exist to guide decision-making in this regard, the variables involved may extend beyond the expertise of the gastroenterologist.28 For instance, in addition to the procedural risk for bleeding, the indication for antithrombotic therapy, risk of a thrombotic event, duration of action of the antithrombotic, and available bridging options should all be considered according to recommendations.28,29 While requiring more time on the part of the gastroenterologist, the optimal periprocedural management of antithrombotic agents would usually involve discussion with the provider managing antithrombotic therapy to best conduct a risk-benefit assessment regarding if (and how long) the antithrombotic therapy should be held (Figure). This shared decision-making, which should also include the patient, may help decrease MPL risk and improve outcomes.
Provider defense mechanisms
Physicians may engage in various defensive behaviors in an attempt to mitigate MPL risk; however, these behaviors may, paradoxically, increase risk.30,31
Assurance behaviors
Assurance behaviors refer to the practice of recommending or performing additional services (such as medications, imaging, procedures, and referrals) that are not clearly indicated.2,30,31 Assurance behaviors are driven by fear of MPL risk and/or missing a potential diagnosis. Recent studies have estimated that more than 50% of gastroenterologists worldwide have performed additional invasive procedures without clear indications, and that nearly one-third of endoscopic procedures annually have questionable indications.30,32 While assurance behaviors may seem likely to decrease MPL risk, overall, they may inadvertently increase AE and MPL risk, as well as health care expenditures.3,30,32
Avoidance behaviors
Avoidance behaviors refer to providers avoiding participation in potentially high-risk clinical interventions (for example, the actual procedures), including those for which they are credentialed/certified proficient.30,31 Two clinical scenarios that illustrate this behavior include the following: An advanced endoscopist credentialed to perform ERCP might refer a “high-risk” elderly patient with cholangitis to another provider to perform said ERCP or for percutaneous transhepatic drainage (in the absence of a clear benefit to such), or a gastroenterologist might refer a patient to interventional gastroenterology for resection of a large polyp even though gastroenterologists are usually proficient in this skill and may feel comfortable performing the resection themselves. Avoidance behaviors are driven by a fear of MPL risk and can have several negative consequences.33 For example, patients may not receive indicated interventions. Additionally, patients may have to wait longer for an intervention because they are referred to another provider, which also increases potential for loss to follow-up.2,30,31 This may be viewed as noncompliance with the standard of care, among other hazards, thereby increasing MPL risk.
Documentation tenets
Thorough documentation can decrease MPL risk, especially since it is often used as legal evidence.16 Documenting, for instance, preprocedure discussion of potential risk of AEs (such as bleeding or perforation) or procedural failure (for example, missed lesions)can protect gastroenterologists (Figure).16 While, as discussed previously, these should be covered in the informed consent process (which itself reduces MPL risk), proof of compliance in providing adequate informed consent must come in the form of documentation that indicates that the process took place and specifically what topics were discussed therein. MPL risk may be further decreased by documenting steps taken during a procedure and anatomic landmarks encountered to offer proof of technical competency and compliance with standards of care (Figure).16,34 In this context, it is worth recalling the adage: “If it’s not documented, it did not occur.”
Curbside consults versus consultation
Also germane here is the topic of whether documentation is needed for “curbside consults.” The uncertainty is, in part, semantic; that is, at what point does a “curbside” become a consultation? A curbside is a general question or query (such as anything that could also be answered by searching the Internet or reference materials) in response to which information is provided; once it involves provision of medical advice for a specific patient (for example, when patient identifiers have been shared or their EHR has been accessed), it constitutes a consultation. Based on these definitions, a curbside need not be documented, whereas a consultation – even if seemingly trivial – should be.
Consideration of language and cultural factors
Language barriers should be considered when the gastroenterologist is communicating with the patient, and such efforts, whenever made, should be documented to best protect against MPL.16,35 These considerations arise not only during the consent process but when obtaining a history, providing postprocedure instructions, and during follow-ups. To this end, 24/7 telephone interpreter services may assist the gastroenterologist (when one is communicating with non–English speakers and is not medically certified in the patient’s native/preferred language) and strengthen trust in the provider-patient relationship.36 Additionally, written materials (such as consent forms, procedural information) in patients’ native/preferred languages should be provided, when available, to enhance patient understanding and participation in care (Figure).35
Challenges posed by telemedicine
The COVID-19 pandemic has rapidly led to more virtual encounters. While increased utilization of telemedicine platforms may make health care more accessible, it does not lessen the clinicians’ duty to patients and may actually expose them to greater MPL risk.18,37,38 Therefore, the provider must be cognizant of two key principles to mitigate MPL risk in the context of telemedicine encounters. First, the same standard of care applies to virtual and in-person encounters.18,37,38 Second, patient privacy and HIPAA regulations are not waived during telemedicine encounters, and breaches of such may result in an MPL claim.18,37,38
With regard to the first principle, for patients who have not been physically examined (such as when a telemedicine visit was substituted for an in-person clinic encounter), gastroenterologists should not overlook requesting timely preprocedure anesthesia consultation or obtaining additional laboratory studies as needed to ensure safety and the same standard of care. Moreover, particularly in the context of pandemic-related decreased procedural capacity, triaging procedures can be especially challenging. Standardized institutional criteria which prioritize certain diagnoses/conditions over others, leaving room for justifiable exceptions, are advisable.
Vicarious liability
“Vicarious liability” is defined as that extending to persons who have not committed a wrong but on whose behalf wrongdoers acted.39 Therefore, gastroenterologists may be liable not only for their own actions but also for those of personnel they supervise (such as fellow trainees and non–physician practitioners).39 Vicarious liability aims to ensure that systemic checks and balances are in place so that, if failure occurs, harm can still be mitigated and/or avoided, as illustrated by Reason’s “Swiss Cheese Model.”40
Conclusion
Any gastroenterologist can experience an MPL claim. Such an experience can be especially stressful and confusing to early-career clinicians, especially if they’re unfamiliar with legal proceedings. Although MPL principles are not often taught in medical school or residency, it is important for gastroenterologists to be informed regarding tenets of MPL and cognizant of clinical situations which have relatively higher MPL risk. This can assuage untoward angst regarding MPL and highlight proactive risk-mitigation strategies. In general, gastroenterologist practices that can mitigate MPL risk include effective communication; adequate informed consent/refusal; documentation of preprocedure counseling, periprocedure events, and postprocedure recommendations; and maintenance of proper certification and privileging.
Dr. Azizian and Dr. Dalai are with the University of California, Los Angeles and the department of medicine at Olive View–UCLA Medical Center, Sylmar, Calif. They are co–first authors of this paper. Dr. Dalai is also with the division of gastroenterology at the University of New Mexico, Albuquerque. Dr. Adams is with the Center for Clinical Management Research in Veterans Affairs Ann Arbor Healthcare System, the division of gastroenterology at the University of Michigan Health System, and the Institute for Healthcare Policy and Innovation, all in Ann Arbor, Mich. Dr. Tabibian is with UCLA and the division of gastroenterology at Olive View–UCLA Medical Center. The authors have no conflicts of interest.
References
1. 2020 Data Sharing Project Gastroenterology 2009-2018. Inside Medical Liability: Second Quarter. Accessed 2020 Dec 6.
2. Mello MM et al. Health Aff (Millwood). 2004 Jul-Aug;23(4):42-53.
3. Adams MA et al. JAMA. 2014 Oct;312(13):1348-9.
4. Pegalis SE. American Law of Medical Malpractice 3d, Vol. 2. St. Paul, Minn.: Thomson Reuters, 2005.
5. Feld LD et al. Am J Gastroenterol. 2018 Nov;113(11):1577-9.
6. Sawyer v. Wight, 196 F. Supp. 2d 220, 226 (E.D.N.Y. 2002).
7. Michael A. Sita v. Long Island Jewish-Hillside Medical Center, 22 A.D.3d 743 (N.Y. App. Div. 2005).
8. Conklin LS et al. Clin Gastroenterol Hepatol. 2008 Jun;6(6):677-81.
9. Jena AB et al. N Engl J Med. 2011 Aug 18;365(7):629-36.
10. Kane CK. “Policy Research Perspectives Medical Liability Claim Frequency: A 2007-2008 Snapshot of Physicians.” Chicago: American Medical Association, 2010.
11. Hernandez LV et al. World J Gastrointest Endosc. 2013 Apr 16;5(4):169-73.
12. Schaffer AC et al. JAMA Intern Med. 2017 May 1;177(5):710-8.
13. Natanson v. Kline, 186 Kan. 393, 409, 350 P.2d 1093, 1106, decision clarified on denial of reh’g, 187 Kan. 186, 354 P.2d 670 (1960).
14. Truman v. Thomas, 27 Cal. 3d 285, 292, 611 P.2d 902, 906 (1980).
15. Shah P et al. Informed Consent, in “StatPearls.” Treasure Island, Fla.: StatPearls Publishing, 2020 Jan. Updated 2020 Aug 22.
16. Rex DK. Clin Gastroenterol Hepatol. 2013 Jul;11(7):768-73.
17. Gerstenberger PD, Plumeri PA. Gastrointest Endosc. Mar-Apr 1993;39(2):132-8.
18. Adams MA and Allen JI. Clin Gastroenterol Hepatol. 2019 Nov;17(12):2392-6.e1.
19. Ahlawat R et al. Esophagogastroduodenoscopy, in “StatPearls.” Treasure Island, Fla.: StatPearls Publishing, 2020 Jan. Updated 2020 Dec 9.
20. Cotton PB. Gastrointest Endosc. 2006 Mar;63(3):378-82.
21. Cotton PB. Gastrointest Endosc. 2010 Oct;72(4):904.
22. Adamson TE et al. West J Med. 1989 Mar;150(3):356-60.
23. Trap R et al. Endoscopy. 1999 Feb;31(2):125-30.
24. Funaki B. Semin Intervent Radiol. 2015 Mar;32(1):61-4.
25. Feeding Tube Nursing Home and Hospital Malpractice. Miller & Zois, Attorneys at Law. Accessed 2020 Jun 20.
26. Medical Malpractice Lawsuit Brings $750,000 Settlement: Death of 82-year-old woman from sepsis due to improper placement of feeding tube. Lubin & Meyers PC. Accessed 2020 Jun 20.
27. Brendel RW et al. Med Clin North Am. 2010 Nov;94(6):1229-40, xi-ii.
28. ASGE Standards of Practice Committee; Acosta RD et al. Gastrointest Endosc. 2016 Jan;83(1):3-16.
29. Saleem S and Thomas AL. Cureus. 2018 Jun 25;10(6):e2878.
30. Hiyama T et al. World J Gastroenterol. 2006 Dec 21;12(47):7671-5.
31. Studdert DM et al. JAMA. 2005 Jun 1;293(21):2609-17.
32. Shaheen NJ et al. Gastroenterology. 2018 May;154(7):1993-2003.
33. Oza VM et al. Clin Gastroenterol Hepatol. 2016 Feb;14(2):172-4.
34. Feld AD. Gastrointest Endosc Clin N Am. 2002 Jan;12(1):171-9, viii-ix.
35. Lee JS et al. J Gen Intern Med. 2017 Aug;32(8):863-70.
36. Forrow L and Kontrimas JC. J Gen Intern Med. 2017 Aug;32(8):855-7.
37. Moses RE et al. Am J Gastroenterol. 2014 Aug;109(8):1128-32.
38. Tabibian JH. “The Evolution of Telehealth.” Guidepoint: Legal Solutions Blog. Accessed 2020 Aug 12.
39. Feld AD. Am J Gastroenterol. 2004 Sep;99(9):1641-4.
40. Reason J. BMJ. 2000;320(7237):768‐70.
Disclaimer: This article is for educational purposes only. All examples are hypothetical and aim to illustrate common clinical scenarios and challenges gastroenterologists may encounter within their scope of practice. The content herein should not be interpreted as legal advice for individual cases nor a substitute for seeking the advice of an attorney.
There are unique potential stressors faced by the gastroenterologist at each career stage, some more so early on. One such stressor, and one particularly important in a procedure-intensive specialty like GI, is medical professional liability (MPL), historically termed “medical malpractice.” Between 2009 and 2018, GI was the second-highest internal medicine subspecialty in both MPL claims made and claims paid,1 yet instruction on MPL risk and mitigation is scarce in fellowship, as is the available GI-related literature on the topic. This scarcity may generate untoward stress and unnecessarily expose gastroenterologists to avoidable MPL pitfalls. Therefore, it is vital for GI trainees, early-career gastroenterologists, and even seasoned gastroenterologists to have a working and updated knowledge of the general principles of MPL and GI-specific considerations. Such understanding can help preserve physician well-being, increase professional satisfaction, strengthen the doctor-patient relationship, and improve health care outcomes.2
To this end, we herein provide a focused review of the following: key MPL concepts, trends in MPL claims, GI-related MPL risk scenarios and considerations, adverse provider defensive mechanisms, documentation tenets, challenges posed by telemedicine, and the concept of “vicarious liability.”
Key MPL concepts
MPL falls under the umbrella of tort law, which itself falls under the umbrella of civil law; that is, civil (as opposed to criminal) justice governs torts – including but not limited to MPL claims – as well as other areas of law concerning noncriminal injury.3 A “tort” is a “civil wrong that unfairly causes another to experience loss or harm resulting in legal liability.”3 MPL claims assert the tort of negligence (similar to the concept of “incompetence”) and endeavor to compensate the harmed patient/individual while simultaneously dissuading suboptimal medical care by the provider in the future.4,5 A successful MPL claim must prove four overlapping elements: that the tortfeasor (here, the gastroenterologist) owed a duty of care to the injured party and breached that duty, which caused damages.6 Given that MPL cases exist within tort law rather than criminal law, the burden of proof for these cases is not “beyond a reasonable doubt”; instead, it’s “to a reasonable medical probability.”7
Trends in MPL claims
According to data compiled by the MPL Association, 278,220 MPL claims were made in the United States from 1985 to 2012.3,8-10 Among these, 1.8% involved gastroenterologists, which puts it at 17th place out of the 20 specialties surveyed.9 While the number of paid claims over this time frame decreased in GI by 34.6% (from 18.5 to 12.1 cases per 1,000 physician-years), there was a concurrent 23.3% increase in average claim compensation; essentially, there were fewer paid GI-related claims but there were higher payouts per paid claim.11,12 From 2009 to 2018, average legal defense costs for paid GI-related claims were $97,392, and average paid amount was $330,876.1
GI-related MPL risk scenarios and considerations
Many MPL claims relate to situations involving medical errors or adverse events (AEs), be they procedural or nonprocedural. However other aspects of GI also carry MPL risk.

Informed consent
MPL claims may be made not only on the grounds of inadequately informed consent but also inadequately informed refusal.5,13,14 While standards for adequate informed consent vary by state, most states apply the “reasonable patient standard,” i.e., assuming an average patient with enough information to be an active participant in the medical decision-making process. Generally, informed consent should ensure that the patient understands the nature of the procedure/treatment being proposed, there is a discussion of the risks and benefits of undergoing and not undergoing the procedure/treatment, reasonable alternatives are presented, the risks and benefits associated with these alternatives are discussed, and the patient’s comprehension of these things is assessed (Figure).15 Additionally, informed consent should be tailored to each patient and GI procedure/treatment on a case-by-case basis rather than using a one-size-fits-all approach. Moreover, documentation of the patient’s understanding of the (tailored) information provided can concurrently improve quality of the consent and potentially decrease MPL risk (Figure).16
Endoscopic procedures
Procedure-related MPL claims represent approximately 25% of all GI-related claims (8,17). Among these, 52% involve colonoscopy, 16% involve endoscopic retrograde cholangiopancreatography (ERCP), and 11% involve esophagogastroduodenoscopy.8 Albeit generally safe, colonoscopy, as with esophagogastroduodenoscopy, is subject to rare but serious AEs.18,19 Risk of these AEs may be accentuated in certain scenarios (such as severe colonic inflammation or coagulopathy) and, as discussed earlier, may merit tailored informed consent. Regardless of the procedure, in the event of postprocedural development of signs/symptoms (such as tachycardia, fever, chest or abdominal discomfort, or hypotension) indicating a potential AE, stabilizing measures and evaluation (such as blood work and imaging) should be undertaken, and hospital admission (if not already hospitalized) should be considered until discharge is deemed safe.19
ERCP-related MPL claims, for many years, have had the highest average compensation of any GI procedure.11 Though discussion of advanced procedures is beyond the scope of this article, it is worth mentioning the observation that most of such claims involve an allegation that the procedure was not indicated (for example, that it was performed based on inadequate evidence of pancreatobiliary pathology), or was for diagnostic purposes (for example, being done instead of noninvasive imaging) rather than therapeutic.20-23 This emphasizes the importance of appropriate procedure indications.
Percutaneous endoscopic gastrostomy (PEG) placement merits special mention given it can be complicated by ethical challenges (for example, needing a surrogate decision-maker’s consent or representing medical futility) and has a relatively high potential for MPL claims. PEG placement carries a low AE rate (0.1%-1%), but these AEs may result in high morbidity/mortality, in part because of the underlying comorbidities of patients needing PEG placement.24,25 Also, timing of a patient’s demise may coincide with PEG placement, thereby prompting (possibly unfounded) perceptions of causality.24-27 Therefore, such scenarios merit unique additional preprocedure safeguards. For instance, for patients lacking capacity to provide informed consent, especially when family members may differ on whether PEG should be placed, it is advisable to ask the family to select one surrogate decision-maker (if there’s no advance directive) to whom the gastroenterologist should discuss both the risks, benefits, and goals of PEG placement in the context of the patient’s overall clinical trajectory/life expectancy and the need for consent (or refusal) based on what the patient would have wished. In addition, having a medical professional witness this discussion may be useful.27
Antithrombotic agents
Periprocedural management of antithrombotics, including anticoagulants and antiplatelets, can pose challenges for the gastroenterologist. While clinical practice guidelines exist to guide decision-making in this regard, the variables involved may extend beyond the expertise of the gastroenterologist.28 For instance, in addition to the procedural risk for bleeding, the indication for antithrombotic therapy, risk of a thrombotic event, duration of action of the antithrombotic, and available bridging options should all be considered according to recommendations.28,29 While requiring more time on the part of the gastroenterologist, the optimal periprocedural management of antithrombotic agents would usually involve discussion with the provider managing antithrombotic therapy to best conduct a risk-benefit assessment regarding if (and how long) the antithrombotic therapy should be held (Figure). This shared decision-making, which should also include the patient, may help decrease MPL risk and improve outcomes.
Provider defense mechanisms
Physicians may engage in various defensive behaviors in an attempt to mitigate MPL risk; however, these behaviors may, paradoxically, increase risk.30,31
Assurance behaviors
Assurance behaviors refer to the practice of recommending or performing additional services (such as medications, imaging, procedures, and referrals) that are not clearly indicated.2,30,31 Assurance behaviors are driven by fear of MPL risk and/or missing a potential diagnosis. Recent studies have estimated that more than 50% of gastroenterologists worldwide have performed additional invasive procedures without clear indications, and that nearly one-third of endoscopic procedures annually have questionable indications.30,32 While assurance behaviors may seem likely to decrease MPL risk, overall, they may inadvertently increase AE and MPL risk, as well as health care expenditures.3,30,32
Avoidance behaviors
Avoidance behaviors refer to providers avoiding participation in potentially high-risk clinical interventions (for example, the actual procedures), including those for which they are credentialed/certified proficient.30,31 Two clinical scenarios that illustrate this behavior include the following: An advanced endoscopist credentialed to perform ERCP might refer a “high-risk” elderly patient with cholangitis to another provider to perform said ERCP or for percutaneous transhepatic drainage (in the absence of a clear benefit to such), or a gastroenterologist might refer a patient to interventional gastroenterology for resection of a large polyp even though gastroenterologists are usually proficient in this skill and may feel comfortable performing the resection themselves. Avoidance behaviors are driven by a fear of MPL risk and can have several negative consequences.33 For example, patients may not receive indicated interventions. Additionally, patients may have to wait longer for an intervention because they are referred to another provider, which also increases potential for loss to follow-up.2,30,31 This may be viewed as noncompliance with the standard of care, among other hazards, thereby increasing MPL risk.
Documentation tenets
Thorough documentation can decrease MPL risk, especially since it is often used as legal evidence.16 Documenting, for instance, preprocedure discussion of potential risk of AEs (such as bleeding or perforation) or procedural failure (for example, missed lesions)can protect gastroenterologists (Figure).16 While, as discussed previously, these should be covered in the informed consent process (which itself reduces MPL risk), proof of compliance in providing adequate informed consent must come in the form of documentation that indicates that the process took place and specifically what topics were discussed therein. MPL risk may be further decreased by documenting steps taken during a procedure and anatomic landmarks encountered to offer proof of technical competency and compliance with standards of care (Figure).16,34 In this context, it is worth recalling the adage: “If it’s not documented, it did not occur.”
Curbside consults versus consultation
Also germane here is the topic of whether documentation is needed for “curbside consults.” The uncertainty is, in part, semantic; that is, at what point does a “curbside” become a consultation? A curbside is a general question or query (such as anything that could also be answered by searching the Internet or reference materials) in response to which information is provided; once it involves provision of medical advice for a specific patient (for example, when patient identifiers have been shared or their EHR has been accessed), it constitutes a consultation. Based on these definitions, a curbside need not be documented, whereas a consultation – even if seemingly trivial – should be.
Consideration of language and cultural factors
Language barriers should be considered when the gastroenterologist is communicating with the patient, and such efforts, whenever made, should be documented to best protect against MPL.16,35 These considerations arise not only during the consent process but when obtaining a history, providing postprocedure instructions, and during follow-ups. To this end, 24/7 telephone interpreter services may assist the gastroenterologist (when one is communicating with non–English speakers and is not medically certified in the patient’s native/preferred language) and strengthen trust in the provider-patient relationship.36 Additionally, written materials (such as consent forms, procedural information) in patients’ native/preferred languages should be provided, when available, to enhance patient understanding and participation in care (Figure).35
Challenges posed by telemedicine
The COVID-19 pandemic has rapidly led to more virtual encounters. While increased utilization of telemedicine platforms may make health care more accessible, it does not lessen the clinicians’ duty to patients and may actually expose them to greater MPL risk.18,37,38 Therefore, the provider must be cognizant of two key principles to mitigate MPL risk in the context of telemedicine encounters. First, the same standard of care applies to virtual and in-person encounters.18,37,38 Second, patient privacy and HIPAA regulations are not waived during telemedicine encounters, and breaches of such may result in an MPL claim.18,37,38
With regard to the first principle, for patients who have not been physically examined (such as when a telemedicine visit was substituted for an in-person clinic encounter), gastroenterologists should not overlook requesting timely preprocedure anesthesia consultation or obtaining additional laboratory studies as needed to ensure safety and the same standard of care. Moreover, particularly in the context of pandemic-related decreased procedural capacity, triaging procedures can be especially challenging. Standardized institutional criteria which prioritize certain diagnoses/conditions over others, leaving room for justifiable exceptions, are advisable.
Vicarious liability
“Vicarious liability” is defined as that extending to persons who have not committed a wrong but on whose behalf wrongdoers acted.39 Therefore, gastroenterologists may be liable not only for their own actions but also for those of personnel they supervise (such as fellow trainees and non–physician practitioners).39 Vicarious liability aims to ensure that systemic checks and balances are in place so that, if failure occurs, harm can still be mitigated and/or avoided, as illustrated by Reason’s “Swiss Cheese Model.”40
Conclusion
Any gastroenterologist can experience an MPL claim. Such an experience can be especially stressful and confusing to early-career clinicians, especially if they’re unfamiliar with legal proceedings. Although MPL principles are not often taught in medical school or residency, it is important for gastroenterologists to be informed regarding tenets of MPL and cognizant of clinical situations which have relatively higher MPL risk. This can assuage untoward angst regarding MPL and highlight proactive risk-mitigation strategies. In general, gastroenterologist practices that can mitigate MPL risk include effective communication; adequate informed consent/refusal; documentation of preprocedure counseling, periprocedure events, and postprocedure recommendations; and maintenance of proper certification and privileging.
Dr. Azizian and Dr. Dalai are with the University of California, Los Angeles and the department of medicine at Olive View–UCLA Medical Center, Sylmar, Calif. They are co–first authors of this paper. Dr. Dalai is also with the division of gastroenterology at the University of New Mexico, Albuquerque. Dr. Adams is with the Center for Clinical Management Research in Veterans Affairs Ann Arbor Healthcare System, the division of gastroenterology at the University of Michigan Health System, and the Institute for Healthcare Policy and Innovation, all in Ann Arbor, Mich. Dr. Tabibian is with UCLA and the division of gastroenterology at Olive View–UCLA Medical Center. The authors have no conflicts of interest.
References
1. 2020 Data Sharing Project Gastroenterology 2009-2018. Inside Medical Liability: Second Quarter. Accessed 2020 Dec 6.
2. Mello MM et al. Health Aff (Millwood). 2004 Jul-Aug;23(4):42-53.
3. Adams MA et al. JAMA. 2014 Oct;312(13):1348-9.
4. Pegalis SE. American Law of Medical Malpractice 3d, Vol. 2. St. Paul, Minn.: Thomson Reuters, 2005.
5. Feld LD et al. Am J Gastroenterol. 2018 Nov;113(11):1577-9.
6. Sawyer v. Wight, 196 F. Supp. 2d 220, 226 (E.D.N.Y. 2002).
7. Michael A. Sita v. Long Island Jewish-Hillside Medical Center, 22 A.D.3d 743 (N.Y. App. Div. 2005).
8. Conklin LS et al. Clin Gastroenterol Hepatol. 2008 Jun;6(6):677-81.
9. Jena AB et al. N Engl J Med. 2011 Aug 18;365(7):629-36.
10. Kane CK. “Policy Research Perspectives Medical Liability Claim Frequency: A 2007-2008 Snapshot of Physicians.” Chicago: American Medical Association, 2010.
11. Hernandez LV et al. World J Gastrointest Endosc. 2013 Apr 16;5(4):169-73.
12. Schaffer AC et al. JAMA Intern Med. 2017 May 1;177(5):710-8.
13. Natanson v. Kline, 186 Kan. 393, 409, 350 P.2d 1093, 1106, decision clarified on denial of reh’g, 187 Kan. 186, 354 P.2d 670 (1960).
14. Truman v. Thomas, 27 Cal. 3d 285, 292, 611 P.2d 902, 906 (1980).
15. Shah P et al. Informed Consent, in “StatPearls.” Treasure Island, Fla.: StatPearls Publishing, 2020 Jan. Updated 2020 Aug 22.
16. Rex DK. Clin Gastroenterol Hepatol. 2013 Jul;11(7):768-73.
17. Gerstenberger PD, Plumeri PA. Gastrointest Endosc. Mar-Apr 1993;39(2):132-8.
18. Adams MA and Allen JI. Clin Gastroenterol Hepatol. 2019 Nov;17(12):2392-6.e1.
19. Ahlawat R et al. Esophagogastroduodenoscopy, in “StatPearls.” Treasure Island, Fla.: StatPearls Publishing, 2020 Jan. Updated 2020 Dec 9.
20. Cotton PB. Gastrointest Endosc. 2006 Mar;63(3):378-82.
21. Cotton PB. Gastrointest Endosc. 2010 Oct;72(4):904.
22. Adamson TE et al. West J Med. 1989 Mar;150(3):356-60.
23. Trap R et al. Endoscopy. 1999 Feb;31(2):125-30.
24. Funaki B. Semin Intervent Radiol. 2015 Mar;32(1):61-4.
25. Feeding Tube Nursing Home and Hospital Malpractice. Miller & Zois, Attorneys at Law. Accessed 2020 Jun 20.
26. Medical Malpractice Lawsuit Brings $750,000 Settlement: Death of 82-year-old woman from sepsis due to improper placement of feeding tube. Lubin & Meyers PC. Accessed 2020 Jun 20.
27. Brendel RW et al. Med Clin North Am. 2010 Nov;94(6):1229-40, xi-ii.
28. ASGE Standards of Practice Committee; Acosta RD et al. Gastrointest Endosc. 2016 Jan;83(1):3-16.
29. Saleem S and Thomas AL. Cureus. 2018 Jun 25;10(6):e2878.
30. Hiyama T et al. World J Gastroenterol. 2006 Dec 21;12(47):7671-5.
31. Studdert DM et al. JAMA. 2005 Jun 1;293(21):2609-17.
32. Shaheen NJ et al. Gastroenterology. 2018 May;154(7):1993-2003.
33. Oza VM et al. Clin Gastroenterol Hepatol. 2016 Feb;14(2):172-4.
34. Feld AD. Gastrointest Endosc Clin N Am. 2002 Jan;12(1):171-9, viii-ix.
35. Lee JS et al. J Gen Intern Med. 2017 Aug;32(8):863-70.
36. Forrow L and Kontrimas JC. J Gen Intern Med. 2017 Aug;32(8):855-7.
37. Moses RE et al. Am J Gastroenterol. 2014 Aug;109(8):1128-32.
38. Tabibian JH. “The Evolution of Telehealth.” Guidepoint: Legal Solutions Blog. Accessed 2020 Aug 12.
39. Feld AD. Am J Gastroenterol. 2004 Sep;99(9):1641-4.
40. Reason J. BMJ. 2000;320(7237):768‐70.
Is board recertification worth it?
I passed the neurology boards, for the first time, in 1998. Then again in 2009, and most recently in 2019.
So I’m up again in 2029. Regrettably, I missed grandfathering in for life by a few years.
Some people don’t study for them, but I’m a little too compulsive not to. I’d guess I put 40-50 hours into doing so in the 3 months beforehand. I didn’t want to fail and have to pay a hefty fee to retake them (the test fee for once is enough as it is).
I’ll be 63 when my next certification is due.
So I wonder (if I’m still in practice) will it even be worthwhile to do it all again? I like what I do, but certainly don’t plan on practicing forever.
Board certification looks good on paper, but certainly isn’t a requirement to practice. One of the best cardiologists I know has never bothered to get his board certification and I don’t think any less of him for it. He also isn’t wanting for patients, and those he has think he’s awesome.
That said, there are things, like being involved in research and legal work, where board certification is strongly recommended, if not mandatory. Since I do both, I certainly wouldn’t want to do anything that might affect my participating in them – if I’m still doing this in 8 years.
By the same token, my office lease runs out when I’m 62. At that point I’ll have been in the same place for 17 years. I don’t consider that a bad thing. I like my current office, and will be perfectly happy to wrap up my career here.
It brings up the same question, though, with logistics that are an even bigger PIA. The last thing I want to do is move my office as my career is winding down. But a lease extension for a few years can be negotiated, a board certification can’t.
I can’t help but wonder: If I’ve already passed it three times, hopefully that means I know what I’m doing. One side will argue that it’s purely greed, as the people who run the boards need money and a way to justify their existence. On the other side are those who argue that maintenance of certification, while not perfect, is the only way we have of making sure practicing physicians are staying up to snuff.
The truth, as always, is somewhere in between.
But it still raises a question that I, fortunately, have another 8 years to think about. Because I’m not in a position to debate if it’s right or wrong, I just have to play by the rules.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
I passed the neurology boards, for the first time, in 1998. Then again in 2009, and most recently in 2019.
So I’m up again in 2029. Regrettably, I missed grandfathering in for life by a few years.
Some people don’t study for them, but I’m a little too compulsive not to. I’d guess I put 40-50 hours into doing so in the 3 months beforehand. I didn’t want to fail and have to pay a hefty fee to retake them (the test fee for once is enough as it is).
I’ll be 63 when my next certification is due.
So I wonder (if I’m still in practice) will it even be worthwhile to do it all again? I like what I do, but certainly don’t plan on practicing forever.
Board certification looks good on paper, but certainly isn’t a requirement to practice. One of the best cardiologists I know has never bothered to get his board certification and I don’t think any less of him for it. He also isn’t wanting for patients, and those he has think he’s awesome.
That said, there are things, like being involved in research and legal work, where board certification is strongly recommended, if not mandatory. Since I do both, I certainly wouldn’t want to do anything that might affect my participating in them – if I’m still doing this in 8 years.
By the same token, my office lease runs out when I’m 62. At that point I’ll have been in the same place for 17 years. I don’t consider that a bad thing. I like my current office, and will be perfectly happy to wrap up my career here.
It brings up the same question, though, with logistics that are an even bigger PIA. The last thing I want to do is move my office as my career is winding down. But a lease extension for a few years can be negotiated, a board certification can’t.
I can’t help but wonder: If I’ve already passed it three times, hopefully that means I know what I’m doing. One side will argue that it’s purely greed, as the people who run the boards need money and a way to justify their existence. On the other side are those who argue that maintenance of certification, while not perfect, is the only way we have of making sure practicing physicians are staying up to snuff.
The truth, as always, is somewhere in between.
But it still raises a question that I, fortunately, have another 8 years to think about. Because I’m not in a position to debate if it’s right or wrong, I just have to play by the rules.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
I passed the neurology boards, for the first time, in 1998. Then again in 2009, and most recently in 2019.
So I’m up again in 2029. Regrettably, I missed grandfathering in for life by a few years.
Some people don’t study for them, but I’m a little too compulsive not to. I’d guess I put 40-50 hours into doing so in the 3 months beforehand. I didn’t want to fail and have to pay a hefty fee to retake them (the test fee for once is enough as it is).
I’ll be 63 when my next certification is due.
So I wonder (if I’m still in practice) will it even be worthwhile to do it all again? I like what I do, but certainly don’t plan on practicing forever.
Board certification looks good on paper, but certainly isn’t a requirement to practice. One of the best cardiologists I know has never bothered to get his board certification and I don’t think any less of him for it. He also isn’t wanting for patients, and those he has think he’s awesome.
That said, there are things, like being involved in research and legal work, where board certification is strongly recommended, if not mandatory. Since I do both, I certainly wouldn’t want to do anything that might affect my participating in them – if I’m still doing this in 8 years.
By the same token, my office lease runs out when I’m 62. At that point I’ll have been in the same place for 17 years. I don’t consider that a bad thing. I like my current office, and will be perfectly happy to wrap up my career here.
It brings up the same question, though, with logistics that are an even bigger PIA. The last thing I want to do is move my office as my career is winding down. But a lease extension for a few years can be negotiated, a board certification can’t.
I can’t help but wonder: If I’ve already passed it three times, hopefully that means I know what I’m doing. One side will argue that it’s purely greed, as the people who run the boards need money and a way to justify their existence. On the other side are those who argue that maintenance of certification, while not perfect, is the only way we have of making sure practicing physicians are staying up to snuff.
The truth, as always, is somewhere in between.
But it still raises a question that I, fortunately, have another 8 years to think about. Because I’m not in a position to debate if it’s right or wrong, I just have to play by the rules.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
The Physician Support Line: One psychiatrist strives to make a difference
Have you ever had a really good idea about how to improve the delivery of mental health services? An idea that would help people, but that would require passion, innovation, and hard work to implement, and one that immediately is beset with a list of reasons why it can not be implemented?
Mona Masood, DO, had an idea. The Pennsylvania psychiatrist was asked to help moderate a Facebook group started by one of her infectious disease colleagues last winter – a private Facebook group for physicians working with COVID-19 patients.
“The group was getting posts from frontline workers about how depressed and hopeless they were feeling.” Dr. Masood said. “People were posting about how they were having escape fantasies and how they regretted becoming physicians. It became clear that there was a need for more support.”
psychiatrist volunteers would take calls from physicians who needed someone to talk to – the psychiatrist would provide a sympathetic ear and have a list of resources, but this would be support, not treatment. There would be no prescriptions, no treatment relationship, no reporting to licensing boards or employers. The calls would be anonymous.
She posted her idea on the Facebook group, and the response was immediate. “There were a lot of emails – 200 psychiatrists responded saying: “Sign me up.” A Zoom meeting was set up, and the process was set in motion.
Dr. Masood used a Google document for weekly sign-ups so the volunteer psychiatrists could choose times. “We had to pay for an upgraded Google suite package for that many users. Getting this up and running was like the saying about building a plane as you fly it,” Dr. Masood said. “It forced so much so quickly because there was this acknowledgment that the need was there.”
Initially, the support line launched with a telehealth platform, but there was a problem. “Many doctors don’t want to be seen; they worry about being recognized.” Dr. Masood researched hotline phone services and was able to get one for a reduced fee. The volunteers have an App on their smartphones that enables them to log in at the start of their shifts and log out at the end. In addition to the logistics of coordinating the volunteers – now numbering over 700 – the group found a health care law firm that provided pro bono services to review the policies and procedures.
Now that the support line is running, Dr. Masood is able to set up the day’s volunteers for the support line connection in a few minutes each morning, but the beginning was not easy. Her private practice transitioned to telemedicine, and her two children were home with one in virtual school. “At first, it was like another full-time job.” She still remains available for trouble-shooting during the day. It’s a project she has taken on with passion.
The support line began as a response to watching colleagues struggle with COVID. Since it launched, there have been approximately 2,000 calls. Calls typically last for 20 to 90 minutes, and no one has called with a suicidal crisis. It is now open to doctors and medical students looking for support for any reason. “Physicians call with all kinds of issues. In the first 3 months, it was COVID, but then they called with other concerns – there were doctors who called with election anxiety, really anything that affects the general public also affects us.”
The group has also offered Saturday didactic sessions for volunteers and weekly debriefing sessions. Dr. Masood has been approached by Vibrant Emotional Health, the administrator of the National Suicide Prevention Lifeline, about resources to help with funding – until now, this endeavor has had no financing – and she is hopeful that their financial support will allow the support line to sustain itself and grow. Future directions include advocating for systemic change in how physician mental health and wellness issues are addressed.
The Physician Support Line was one psychiatrist’s vision for how to address a problem. Like so many things related to this pandemic, it happened quickly and with surprising efficiency. Implementing this service, however, was not easy – it required hard work, innovative thinking, and passion. Those looking for someone to listen can call 1-888-409-0141 and psychiatrists who wish to volunteer can sign up at physiciansupportline.com/volunteer-info.
Dr. Miller is coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Johns Hopkins University Press, 2018). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins University, both in Baltimore.
Have you ever had a really good idea about how to improve the delivery of mental health services? An idea that would help people, but that would require passion, innovation, and hard work to implement, and one that immediately is beset with a list of reasons why it can not be implemented?
Mona Masood, DO, had an idea. The Pennsylvania psychiatrist was asked to help moderate a Facebook group started by one of her infectious disease colleagues last winter – a private Facebook group for physicians working with COVID-19 patients.
“The group was getting posts from frontline workers about how depressed and hopeless they were feeling.” Dr. Masood said. “People were posting about how they were having escape fantasies and how they regretted becoming physicians. It became clear that there was a need for more support.”
psychiatrist volunteers would take calls from physicians who needed someone to talk to – the psychiatrist would provide a sympathetic ear and have a list of resources, but this would be support, not treatment. There would be no prescriptions, no treatment relationship, no reporting to licensing boards or employers. The calls would be anonymous.
She posted her idea on the Facebook group, and the response was immediate. “There were a lot of emails – 200 psychiatrists responded saying: “Sign me up.” A Zoom meeting was set up, and the process was set in motion.
Dr. Masood used a Google document for weekly sign-ups so the volunteer psychiatrists could choose times. “We had to pay for an upgraded Google suite package for that many users. Getting this up and running was like the saying about building a plane as you fly it,” Dr. Masood said. “It forced so much so quickly because there was this acknowledgment that the need was there.”
Initially, the support line launched with a telehealth platform, but there was a problem. “Many doctors don’t want to be seen; they worry about being recognized.” Dr. Masood researched hotline phone services and was able to get one for a reduced fee. The volunteers have an App on their smartphones that enables them to log in at the start of their shifts and log out at the end. In addition to the logistics of coordinating the volunteers – now numbering over 700 – the group found a health care law firm that provided pro bono services to review the policies and procedures.
Now that the support line is running, Dr. Masood is able to set up the day’s volunteers for the support line connection in a few minutes each morning, but the beginning was not easy. Her private practice transitioned to telemedicine, and her two children were home with one in virtual school. “At first, it was like another full-time job.” She still remains available for trouble-shooting during the day. It’s a project she has taken on with passion.
The support line began as a response to watching colleagues struggle with COVID. Since it launched, there have been approximately 2,000 calls. Calls typically last for 20 to 90 minutes, and no one has called with a suicidal crisis. It is now open to doctors and medical students looking for support for any reason. “Physicians call with all kinds of issues. In the first 3 months, it was COVID, but then they called with other concerns – there were doctors who called with election anxiety, really anything that affects the general public also affects us.”
The group has also offered Saturday didactic sessions for volunteers and weekly debriefing sessions. Dr. Masood has been approached by Vibrant Emotional Health, the administrator of the National Suicide Prevention Lifeline, about resources to help with funding – until now, this endeavor has had no financing – and she is hopeful that their financial support will allow the support line to sustain itself and grow. Future directions include advocating for systemic change in how physician mental health and wellness issues are addressed.
The Physician Support Line was one psychiatrist’s vision for how to address a problem. Like so many things related to this pandemic, it happened quickly and with surprising efficiency. Implementing this service, however, was not easy – it required hard work, innovative thinking, and passion. Those looking for someone to listen can call 1-888-409-0141 and psychiatrists who wish to volunteer can sign up at physiciansupportline.com/volunteer-info.
Dr. Miller is coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Johns Hopkins University Press, 2018). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins University, both in Baltimore.
Have you ever had a really good idea about how to improve the delivery of mental health services? An idea that would help people, but that would require passion, innovation, and hard work to implement, and one that immediately is beset with a list of reasons why it can not be implemented?
Mona Masood, DO, had an idea. The Pennsylvania psychiatrist was asked to help moderate a Facebook group started by one of her infectious disease colleagues last winter – a private Facebook group for physicians working with COVID-19 patients.
“The group was getting posts from frontline workers about how depressed and hopeless they were feeling.” Dr. Masood said. “People were posting about how they were having escape fantasies and how they regretted becoming physicians. It became clear that there was a need for more support.”
psychiatrist volunteers would take calls from physicians who needed someone to talk to – the psychiatrist would provide a sympathetic ear and have a list of resources, but this would be support, not treatment. There would be no prescriptions, no treatment relationship, no reporting to licensing boards or employers. The calls would be anonymous.
She posted her idea on the Facebook group, and the response was immediate. “There were a lot of emails – 200 psychiatrists responded saying: “Sign me up.” A Zoom meeting was set up, and the process was set in motion.
Dr. Masood used a Google document for weekly sign-ups so the volunteer psychiatrists could choose times. “We had to pay for an upgraded Google suite package for that many users. Getting this up and running was like the saying about building a plane as you fly it,” Dr. Masood said. “It forced so much so quickly because there was this acknowledgment that the need was there.”
Initially, the support line launched with a telehealth platform, but there was a problem. “Many doctors don’t want to be seen; they worry about being recognized.” Dr. Masood researched hotline phone services and was able to get one for a reduced fee. The volunteers have an App on their smartphones that enables them to log in at the start of their shifts and log out at the end. In addition to the logistics of coordinating the volunteers – now numbering over 700 – the group found a health care law firm that provided pro bono services to review the policies and procedures.
Now that the support line is running, Dr. Masood is able to set up the day’s volunteers for the support line connection in a few minutes each morning, but the beginning was not easy. Her private practice transitioned to telemedicine, and her two children were home with one in virtual school. “At first, it was like another full-time job.” She still remains available for trouble-shooting during the day. It’s a project she has taken on with passion.
The support line began as a response to watching colleagues struggle with COVID. Since it launched, there have been approximately 2,000 calls. Calls typically last for 20 to 90 minutes, and no one has called with a suicidal crisis. It is now open to doctors and medical students looking for support for any reason. “Physicians call with all kinds of issues. In the first 3 months, it was COVID, but then they called with other concerns – there were doctors who called with election anxiety, really anything that affects the general public also affects us.”
The group has also offered Saturday didactic sessions for volunteers and weekly debriefing sessions. Dr. Masood has been approached by Vibrant Emotional Health, the administrator of the National Suicide Prevention Lifeline, about resources to help with funding – until now, this endeavor has had no financing – and she is hopeful that their financial support will allow the support line to sustain itself and grow. Future directions include advocating for systemic change in how physician mental health and wellness issues are addressed.
The Physician Support Line was one psychiatrist’s vision for how to address a problem. Like so many things related to this pandemic, it happened quickly and with surprising efficiency. Implementing this service, however, was not easy – it required hard work, innovative thinking, and passion. Those looking for someone to listen can call 1-888-409-0141 and psychiatrists who wish to volunteer can sign up at physiciansupportline.com/volunteer-info.
Dr. Miller is coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Johns Hopkins University Press, 2018). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins University, both in Baltimore.
The psychiatrist’s role in navigating a toxic news cycle
This transcript has been edited for clarity.
I’m Dr. Gregory Scott Brown, an Austin (Tex.)-based psychiatrist and an affiliate faculty member at the University of Texas Dell Medical School.
Recently, a patient of mine told me that, because of the political environment we find ourselves in, he’s avoiding conversations with some of his longtime friends. Because of this, he’s feeling even more isolated than before.
We’re all coming off the heels of a tough year, and many of us expected that when we entered 2021 we’d quickly turn the page and life would get a whole lot easier. Since the reality is that we’re still dealing with deep divisions, social injustices, and the politicization of evidence-based medicine, emotions are naturally running high.
In listening to my patients over the past few weeks, there’s definitely a sense of optimism about coronavirus vaccines and getting back to life as usual. But there’s also a lingering sense of uncertainty and fear, especially when it comes to the possibility of saying the wrong thing, offending our friends, or just having conversations with people we may disagree with. I’m hearing concerns from my patients that 2020 exposed a dormant hatred that was brewing in the underbelly of our society, in our politics, and in our institutions. Patients are telling me that these concerns are making them anxious and some are avoiding interacting with people they may disagree with altogether because they’re afraid of the difficult conversations that may follow.
Since, like many of my patients as well as many of you, I follow the news, including stories of COVID-related deaths, economic hardships, peaceful protests gone bad, and political vitriol, I’ve had to remind myself about the importance of intentional kindness for effective communication and for supporting mental health.
I was reminded of the paper “Hate in the Counter-Transference” by the well-known pediatrician and psychoanalyst Donald Winnicott. He focuses on how to manage and sort through the strong emotions that may be experienced even during an encounter between a therapist and a patient. Although some of what he has to say doesn’t translate well to modern times, his recommendation that we acknowledge and try to normalize some of our feelings does. And this is how I’ve been starting a conversation with my patients – just by normalizing things a bit.
The past year or so has brought with it a range of intense emotions, including frustration, exhaustion, and some degree of sadness for most of us. When we’re self-aware about our feelings, we can make sense of them early on so that they don’t evolve into maladaptive ones like unhinged anger or hatred. My patients and I actively discuss how our feelings don’t have to get in the way of our ability to live and to interact with each other as we’d like to.
Considering the basic tenets of kindness is a good place to start. I recently spoke with Kelli Harding, MD, a psychiatrist and author of The Rabbit Effect: Live Longer, Happier, and Healthier with the Groundbreaking Science of Kindness. She pointed to research suggesting that kindness can benefit multiple areas of our health, from reducing cardiovascular events to improving mood and anxiety. In her book, she notes that
I tell my patients that we can’t always change our environment, but we can definitely change how we respond to it. This doesn’t mean it’s always an easy process, but there are things we can do. First, we have to acknowledge that some degree of conflict or cordial disagreement is inevitable and it doesn’t have to disrupt our mood.
An interesting study on conflict management pointed out that healthy conflict is actually beneficial in some cases. For instance, in the work environment, conflict can help with team development and better group decision-making. But it’s rigid personality differences, poor communication, emotional stress, and lack of candor that may contribute to so-called high-tension events, and this is where conflict can go awry. These are the areas that we can all focus on improving, not only for performance benefits but for our overall health and well-being as well.
The authors also recommend an active style of engagement as a technique to manage conflict, but in a way that feels both natural and safe. Other authors agree that so-called engaged coping is associated with a higher sense of control and overall improved psychological well-being. What this means is that avoidance may be necessary in the short term, but over time it may lead to more emotional stress and anxiety.
Overcoming the tendency to avoid requires both motivation and self-awareness. We need to know about patterns in our own behavior and how the behavior of others can push our buttons and spiral a healthy disagreement into a heated argument.
I like to recommend the hunger, angry, lonely, tired (HALT) model, which is often used as a self-care gauge in addiction treatment and to reduce medication errors. But I think it’s also a useful way to assess personal readiness for having a difficult conversation. I tell my patients to ask themselves in this moment: “Am I hungry, angry, lonely, or tired?” And if they are, perhaps it’s not the best time for the conversation.
Because 2020 brought with it a new set of challenges, it also forced many of us to focus on things that we just didn’t pay as much attention to before, including checking in on our feelings and the feelings of those around us. It also taught us to pay attention to the way and manner in which we communicate. I think that kindness is much easier to carry into difficult conversations if we approach them with a sense of curiosity before judgment. Ultimately, kindness is one of the best tools for balancing the intense emotions that many of us are feeling right now.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’m Dr. Gregory Scott Brown, an Austin (Tex.)-based psychiatrist and an affiliate faculty member at the University of Texas Dell Medical School.
Recently, a patient of mine told me that, because of the political environment we find ourselves in, he’s avoiding conversations with some of his longtime friends. Because of this, he’s feeling even more isolated than before.
We’re all coming off the heels of a tough year, and many of us expected that when we entered 2021 we’d quickly turn the page and life would get a whole lot easier. Since the reality is that we’re still dealing with deep divisions, social injustices, and the politicization of evidence-based medicine, emotions are naturally running high.
In listening to my patients over the past few weeks, there’s definitely a sense of optimism about coronavirus vaccines and getting back to life as usual. But there’s also a lingering sense of uncertainty and fear, especially when it comes to the possibility of saying the wrong thing, offending our friends, or just having conversations with people we may disagree with. I’m hearing concerns from my patients that 2020 exposed a dormant hatred that was brewing in the underbelly of our society, in our politics, and in our institutions. Patients are telling me that these concerns are making them anxious and some are avoiding interacting with people they may disagree with altogether because they’re afraid of the difficult conversations that may follow.
Since, like many of my patients as well as many of you, I follow the news, including stories of COVID-related deaths, economic hardships, peaceful protests gone bad, and political vitriol, I’ve had to remind myself about the importance of intentional kindness for effective communication and for supporting mental health.
I was reminded of the paper “Hate in the Counter-Transference” by the well-known pediatrician and psychoanalyst Donald Winnicott. He focuses on how to manage and sort through the strong emotions that may be experienced even during an encounter between a therapist and a patient. Although some of what he has to say doesn’t translate well to modern times, his recommendation that we acknowledge and try to normalize some of our feelings does. And this is how I’ve been starting a conversation with my patients – just by normalizing things a bit.
The past year or so has brought with it a range of intense emotions, including frustration, exhaustion, and some degree of sadness for most of us. When we’re self-aware about our feelings, we can make sense of them early on so that they don’t evolve into maladaptive ones like unhinged anger or hatred. My patients and I actively discuss how our feelings don’t have to get in the way of our ability to live and to interact with each other as we’d like to.
Considering the basic tenets of kindness is a good place to start. I recently spoke with Kelli Harding, MD, a psychiatrist and author of The Rabbit Effect: Live Longer, Happier, and Healthier with the Groundbreaking Science of Kindness. She pointed to research suggesting that kindness can benefit multiple areas of our health, from reducing cardiovascular events to improving mood and anxiety. In her book, she notes that
I tell my patients that we can’t always change our environment, but we can definitely change how we respond to it. This doesn’t mean it’s always an easy process, but there are things we can do. First, we have to acknowledge that some degree of conflict or cordial disagreement is inevitable and it doesn’t have to disrupt our mood.
An interesting study on conflict management pointed out that healthy conflict is actually beneficial in some cases. For instance, in the work environment, conflict can help with team development and better group decision-making. But it’s rigid personality differences, poor communication, emotional stress, and lack of candor that may contribute to so-called high-tension events, and this is where conflict can go awry. These are the areas that we can all focus on improving, not only for performance benefits but for our overall health and well-being as well.
The authors also recommend an active style of engagement as a technique to manage conflict, but in a way that feels both natural and safe. Other authors agree that so-called engaged coping is associated with a higher sense of control and overall improved psychological well-being. What this means is that avoidance may be necessary in the short term, but over time it may lead to more emotional stress and anxiety.
Overcoming the tendency to avoid requires both motivation and self-awareness. We need to know about patterns in our own behavior and how the behavior of others can push our buttons and spiral a healthy disagreement into a heated argument.
I like to recommend the hunger, angry, lonely, tired (HALT) model, which is often used as a self-care gauge in addiction treatment and to reduce medication errors. But I think it’s also a useful way to assess personal readiness for having a difficult conversation. I tell my patients to ask themselves in this moment: “Am I hungry, angry, lonely, or tired?” And if they are, perhaps it’s not the best time for the conversation.
Because 2020 brought with it a new set of challenges, it also forced many of us to focus on things that we just didn’t pay as much attention to before, including checking in on our feelings and the feelings of those around us. It also taught us to pay attention to the way and manner in which we communicate. I think that kindness is much easier to carry into difficult conversations if we approach them with a sense of curiosity before judgment. Ultimately, kindness is one of the best tools for balancing the intense emotions that many of us are feeling right now.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’m Dr. Gregory Scott Brown, an Austin (Tex.)-based psychiatrist and an affiliate faculty member at the University of Texas Dell Medical School.
Recently, a patient of mine told me that, because of the political environment we find ourselves in, he’s avoiding conversations with some of his longtime friends. Because of this, he’s feeling even more isolated than before.
We’re all coming off the heels of a tough year, and many of us expected that when we entered 2021 we’d quickly turn the page and life would get a whole lot easier. Since the reality is that we’re still dealing with deep divisions, social injustices, and the politicization of evidence-based medicine, emotions are naturally running high.
In listening to my patients over the past few weeks, there’s definitely a sense of optimism about coronavirus vaccines and getting back to life as usual. But there’s also a lingering sense of uncertainty and fear, especially when it comes to the possibility of saying the wrong thing, offending our friends, or just having conversations with people we may disagree with. I’m hearing concerns from my patients that 2020 exposed a dormant hatred that was brewing in the underbelly of our society, in our politics, and in our institutions. Patients are telling me that these concerns are making them anxious and some are avoiding interacting with people they may disagree with altogether because they’re afraid of the difficult conversations that may follow.
Since, like many of my patients as well as many of you, I follow the news, including stories of COVID-related deaths, economic hardships, peaceful protests gone bad, and political vitriol, I’ve had to remind myself about the importance of intentional kindness for effective communication and for supporting mental health.
I was reminded of the paper “Hate in the Counter-Transference” by the well-known pediatrician and psychoanalyst Donald Winnicott. He focuses on how to manage and sort through the strong emotions that may be experienced even during an encounter between a therapist and a patient. Although some of what he has to say doesn’t translate well to modern times, his recommendation that we acknowledge and try to normalize some of our feelings does. And this is how I’ve been starting a conversation with my patients – just by normalizing things a bit.
The past year or so has brought with it a range of intense emotions, including frustration, exhaustion, and some degree of sadness for most of us. When we’re self-aware about our feelings, we can make sense of them early on so that they don’t evolve into maladaptive ones like unhinged anger or hatred. My patients and I actively discuss how our feelings don’t have to get in the way of our ability to live and to interact with each other as we’d like to.
Considering the basic tenets of kindness is a good place to start. I recently spoke with Kelli Harding, MD, a psychiatrist and author of The Rabbit Effect: Live Longer, Happier, and Healthier with the Groundbreaking Science of Kindness. She pointed to research suggesting that kindness can benefit multiple areas of our health, from reducing cardiovascular events to improving mood and anxiety. In her book, she notes that
I tell my patients that we can’t always change our environment, but we can definitely change how we respond to it. This doesn’t mean it’s always an easy process, but there are things we can do. First, we have to acknowledge that some degree of conflict or cordial disagreement is inevitable and it doesn’t have to disrupt our mood.
An interesting study on conflict management pointed out that healthy conflict is actually beneficial in some cases. For instance, in the work environment, conflict can help with team development and better group decision-making. But it’s rigid personality differences, poor communication, emotional stress, and lack of candor that may contribute to so-called high-tension events, and this is where conflict can go awry. These are the areas that we can all focus on improving, not only for performance benefits but for our overall health and well-being as well.
The authors also recommend an active style of engagement as a technique to manage conflict, but in a way that feels both natural and safe. Other authors agree that so-called engaged coping is associated with a higher sense of control and overall improved psychological well-being. What this means is that avoidance may be necessary in the short term, but over time it may lead to more emotional stress and anxiety.
Overcoming the tendency to avoid requires both motivation and self-awareness. We need to know about patterns in our own behavior and how the behavior of others can push our buttons and spiral a healthy disagreement into a heated argument.
I like to recommend the hunger, angry, lonely, tired (HALT) model, which is often used as a self-care gauge in addiction treatment and to reduce medication errors. But I think it’s also a useful way to assess personal readiness for having a difficult conversation. I tell my patients to ask themselves in this moment: “Am I hungry, angry, lonely, or tired?” And if they are, perhaps it’s not the best time for the conversation.
Because 2020 brought with it a new set of challenges, it also forced many of us to focus on things that we just didn’t pay as much attention to before, including checking in on our feelings and the feelings of those around us. It also taught us to pay attention to the way and manner in which we communicate. I think that kindness is much easier to carry into difficult conversations if we approach them with a sense of curiosity before judgment. Ultimately, kindness is one of the best tools for balancing the intense emotions that many of us are feeling right now.
A version of this article first appeared on Medscape.com.