User login
Official Newspaper of the American College of Surgeons
AMA: Growing patient responsibility leads to greater hassle
CHICAGO – Administrative hassles are taking too much time and money away from physician practices and the burden is likely to grow as employers ask workers to pay for more of their health care.
That’s the message in the most recent edition of the American Medical Association's National Health Insurer Report Card, released at the organization’s annual House of Delegates meeting.

Patients are being asked to cover as much as a quarter of their health care costs, according to the report card. The problem is exacerbated at the beginning of the year, when many must first satisfy a deductible.
Up to half of claims for which insurers pay $0 are those for which the patient owes the full amount, said Mark Reiger, vice president for payment and reimbursement strategy at NHXS, which helped develop the report card.
"As the burden shifts to the patients, more of your revenue is at risk," Mr. Reiger said, noting that "physicians are not very good at collecting patient responsibility."
Payment problems distract from patient care, said Dr. Barbara McAneny of the AMA Board of Trustees. The ABI study found that physician practices are spending as much as 14% of their revenues on getting paid.
You "should not have to divert as much as 14% of your gross revenue to ensure accurate insurance payments for your services," she said.
According to the report card, insurers have improved their performance since the AMA began monitoring it in 2008.
Dr. McAneny said that although things have gotten better, there’s still a lot of money being left on the table – about $43 billion over the last 3 years. That’s how much physicians and insurers have given up due to less than 100% accuracy in claims processing since 2010.
Even so, error rates have dropped from 20% in 2010 to 7% in the latest report.
Medicare was the most accurate payer (98%). UnitedHealthcare was the most accurate commercial insurer (97.5%), while Regence was the least (85%).
The insurers who participated in the report card are Aetna, Anthem, Cigna, Health Care Service Corp., Humana, Regence, UnitedHealthcare, and Medicare.
Insurers were rated according to how quickly they pay claims, how often they pay nothing on a claim, how often they match the contracted fee schedule, whether they disclose the reason for a denial, and a variety of other measures. The report found that the vast majority of claims are paid within 30 days, and most within 15 days. Medicare and Cigna paid 95% of claims within 15 days. In contrast, Aetna paid 66% in the same time frame.
The report shed light on another trend that could lead to more payment problems for physicians: Patients are being required to pay more out of pocket for care. Patients insured by Health Care Service Corp. pay for30% of their care, according to the report card. Most of the other insurers require 20%-25% copays from patients. Humana, on the low end, seeks 15% of the allowable amount from patients.
The AMA is asking payers and vendors to give physicians real-time estimates of the patient’s responsibility at the point of care, Dr. McAneny said.
Among the other findings: Claims denials decreased to 1.8% in 2013, compared with 3% in 2012. Medicare had the highest denial rate at almost 5%.
Insurers also responded more quickly to claims. The fastest, Humana, had a median response time of 6 days, while Aetna was the slowest, with a median response time of 14 days. Medicare matched Aetna, and has maintained that response time since 2008.
Along with the report card, the AMA unveiled its administrative burden index (ABI), which measures how much it costs a practice to rework rejected claims.
Medicare’s performance was not included in the ABI. The ABI score was based largely on the percentage of claims paid after 30 days. The AMA said that the tasks associated with avoidable errors, inefficiency, and waste in the claims process cost an average $2.36 per claim for physicians and insurers. Cigna had the best ABI cost per claim of $1.25 and HCSC had the worst, at $3.32, per claim.
Dr. McAneny said physicians can use the ABI data "to identify the cost of processing claims with the participating health insurers on the report card."
The report card data is based on 2.6 million claims submitted for February and March 2013. It is drawn from 41 states, 80 specialties, and more than 450 practices. It is based exclusively on claims that were submitted electronically and remittances received electronically; therefore, the results may not apply to practices that don’t use electronic submission.
On Twitter @aliciaault
CHICAGO – Administrative hassles are taking too much time and money away from physician practices and the burden is likely to grow as employers ask workers to pay for more of their health care.
That’s the message in the most recent edition of the American Medical Association's National Health Insurer Report Card, released at the organization’s annual House of Delegates meeting.

Patients are being asked to cover as much as a quarter of their health care costs, according to the report card. The problem is exacerbated at the beginning of the year, when many must first satisfy a deductible.
Up to half of claims for which insurers pay $0 are those for which the patient owes the full amount, said Mark Reiger, vice president for payment and reimbursement strategy at NHXS, which helped develop the report card.
"As the burden shifts to the patients, more of your revenue is at risk," Mr. Reiger said, noting that "physicians are not very good at collecting patient responsibility."
Payment problems distract from patient care, said Dr. Barbara McAneny of the AMA Board of Trustees. The ABI study found that physician practices are spending as much as 14% of their revenues on getting paid.
You "should not have to divert as much as 14% of your gross revenue to ensure accurate insurance payments for your services," she said.
According to the report card, insurers have improved their performance since the AMA began monitoring it in 2008.
Dr. McAneny said that although things have gotten better, there’s still a lot of money being left on the table – about $43 billion over the last 3 years. That’s how much physicians and insurers have given up due to less than 100% accuracy in claims processing since 2010.
Even so, error rates have dropped from 20% in 2010 to 7% in the latest report.
Medicare was the most accurate payer (98%). UnitedHealthcare was the most accurate commercial insurer (97.5%), while Regence was the least (85%).
The insurers who participated in the report card are Aetna, Anthem, Cigna, Health Care Service Corp., Humana, Regence, UnitedHealthcare, and Medicare.
Insurers were rated according to how quickly they pay claims, how often they pay nothing on a claim, how often they match the contracted fee schedule, whether they disclose the reason for a denial, and a variety of other measures. The report found that the vast majority of claims are paid within 30 days, and most within 15 days. Medicare and Cigna paid 95% of claims within 15 days. In contrast, Aetna paid 66% in the same time frame.
The report shed light on another trend that could lead to more payment problems for physicians: Patients are being required to pay more out of pocket for care. Patients insured by Health Care Service Corp. pay for30% of their care, according to the report card. Most of the other insurers require 20%-25% copays from patients. Humana, on the low end, seeks 15% of the allowable amount from patients.
The AMA is asking payers and vendors to give physicians real-time estimates of the patient’s responsibility at the point of care, Dr. McAneny said.
Among the other findings: Claims denials decreased to 1.8% in 2013, compared with 3% in 2012. Medicare had the highest denial rate at almost 5%.
Insurers also responded more quickly to claims. The fastest, Humana, had a median response time of 6 days, while Aetna was the slowest, with a median response time of 14 days. Medicare matched Aetna, and has maintained that response time since 2008.
Along with the report card, the AMA unveiled its administrative burden index (ABI), which measures how much it costs a practice to rework rejected claims.
Medicare’s performance was not included in the ABI. The ABI score was based largely on the percentage of claims paid after 30 days. The AMA said that the tasks associated with avoidable errors, inefficiency, and waste in the claims process cost an average $2.36 per claim for physicians and insurers. Cigna had the best ABI cost per claim of $1.25 and HCSC had the worst, at $3.32, per claim.
Dr. McAneny said physicians can use the ABI data "to identify the cost of processing claims with the participating health insurers on the report card."
The report card data is based on 2.6 million claims submitted for February and March 2013. It is drawn from 41 states, 80 specialties, and more than 450 practices. It is based exclusively on claims that were submitted electronically and remittances received electronically; therefore, the results may not apply to practices that don’t use electronic submission.
On Twitter @aliciaault
CHICAGO – Administrative hassles are taking too much time and money away from physician practices and the burden is likely to grow as employers ask workers to pay for more of their health care.
That’s the message in the most recent edition of the American Medical Association's National Health Insurer Report Card, released at the organization’s annual House of Delegates meeting.

Patients are being asked to cover as much as a quarter of their health care costs, according to the report card. The problem is exacerbated at the beginning of the year, when many must first satisfy a deductible.
Up to half of claims for which insurers pay $0 are those for which the patient owes the full amount, said Mark Reiger, vice president for payment and reimbursement strategy at NHXS, which helped develop the report card.
"As the burden shifts to the patients, more of your revenue is at risk," Mr. Reiger said, noting that "physicians are not very good at collecting patient responsibility."
Payment problems distract from patient care, said Dr. Barbara McAneny of the AMA Board of Trustees. The ABI study found that physician practices are spending as much as 14% of their revenues on getting paid.
You "should not have to divert as much as 14% of your gross revenue to ensure accurate insurance payments for your services," she said.
According to the report card, insurers have improved their performance since the AMA began monitoring it in 2008.
Dr. McAneny said that although things have gotten better, there’s still a lot of money being left on the table – about $43 billion over the last 3 years. That’s how much physicians and insurers have given up due to less than 100% accuracy in claims processing since 2010.
Even so, error rates have dropped from 20% in 2010 to 7% in the latest report.
Medicare was the most accurate payer (98%). UnitedHealthcare was the most accurate commercial insurer (97.5%), while Regence was the least (85%).
The insurers who participated in the report card are Aetna, Anthem, Cigna, Health Care Service Corp., Humana, Regence, UnitedHealthcare, and Medicare.
Insurers were rated according to how quickly they pay claims, how often they pay nothing on a claim, how often they match the contracted fee schedule, whether they disclose the reason for a denial, and a variety of other measures. The report found that the vast majority of claims are paid within 30 days, and most within 15 days. Medicare and Cigna paid 95% of claims within 15 days. In contrast, Aetna paid 66% in the same time frame.
The report shed light on another trend that could lead to more payment problems for physicians: Patients are being required to pay more out of pocket for care. Patients insured by Health Care Service Corp. pay for30% of their care, according to the report card. Most of the other insurers require 20%-25% copays from patients. Humana, on the low end, seeks 15% of the allowable amount from patients.
The AMA is asking payers and vendors to give physicians real-time estimates of the patient’s responsibility at the point of care, Dr. McAneny said.
Among the other findings: Claims denials decreased to 1.8% in 2013, compared with 3% in 2012. Medicare had the highest denial rate at almost 5%.
Insurers also responded more quickly to claims. The fastest, Humana, had a median response time of 6 days, while Aetna was the slowest, with a median response time of 14 days. Medicare matched Aetna, and has maintained that response time since 2008.
Along with the report card, the AMA unveiled its administrative burden index (ABI), which measures how much it costs a practice to rework rejected claims.
Medicare’s performance was not included in the ABI. The ABI score was based largely on the percentage of claims paid after 30 days. The AMA said that the tasks associated with avoidable errors, inefficiency, and waste in the claims process cost an average $2.36 per claim for physicians and insurers. Cigna had the best ABI cost per claim of $1.25 and HCSC had the worst, at $3.32, per claim.
Dr. McAneny said physicians can use the ABI data "to identify the cost of processing claims with the participating health insurers on the report card."
The report card data is based on 2.6 million claims submitted for February and March 2013. It is drawn from 41 states, 80 specialties, and more than 450 practices. It is based exclusively on claims that were submitted electronically and remittances received electronically; therefore, the results may not apply to practices that don’t use electronic submission.
On Twitter @aliciaault
AT THE AMA HOUSE OF DELEGATES
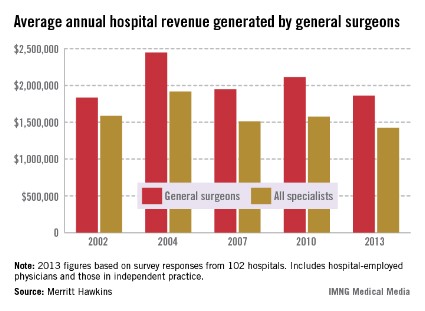
General surgeons and specialties generating less revenue for hospitals
Average annual hospital revenue generated by general surgeons dropped 11.9% from 2010 to 2013, according to a survey by physician job placement firm Merritt Hawkins.
The decrease is in line with the overall picture of net revenue earned by hospitals. The average net revenue generated for all physicians was $1.45 million – a drop of 6.2% from 2010 and, again, the lowest average since 2002, Merritt Hawkins said in its analysis.
The trend of dropping revenue "may be tied to changing venues of care as urgent care centers and occupational medicine centers absorb general surgery work previously done in hospitals," the survey authors wrote.
Average revenue of $1.86 million was reported for general surgeons in 2013, down from $2.11 million in 2010. By comparison, hospital revenue generated was almost $1.42 million for all specialists and $1.57 for all primary care physicians in 2013. The specialists’ average is the lowest Merritt Hawkins has reported since it started the survey in 2002 and is lower than the primary care average for the first time ever, the report noted.
The survey was sent to 5,500 hospitals in January 2013 and was completed by 102 hospital chief financial executives.
Average annual hospital revenue generated by general surgeons dropped 11.9% from 2010 to 2013, according to a survey by physician job placement firm Merritt Hawkins.
The decrease is in line with the overall picture of net revenue earned by hospitals. The average net revenue generated for all physicians was $1.45 million – a drop of 6.2% from 2010 and, again, the lowest average since 2002, Merritt Hawkins said in its analysis.
The trend of dropping revenue "may be tied to changing venues of care as urgent care centers and occupational medicine centers absorb general surgery work previously done in hospitals," the survey authors wrote.
Average revenue of $1.86 million was reported for general surgeons in 2013, down from $2.11 million in 2010. By comparison, hospital revenue generated was almost $1.42 million for all specialists and $1.57 for all primary care physicians in 2013. The specialists’ average is the lowest Merritt Hawkins has reported since it started the survey in 2002 and is lower than the primary care average for the first time ever, the report noted.
The survey was sent to 5,500 hospitals in January 2013 and was completed by 102 hospital chief financial executives.
Average annual hospital revenue generated by general surgeons dropped 11.9% from 2010 to 2013, according to a survey by physician job placement firm Merritt Hawkins.
The decrease is in line with the overall picture of net revenue earned by hospitals. The average net revenue generated for all physicians was $1.45 million – a drop of 6.2% from 2010 and, again, the lowest average since 2002, Merritt Hawkins said in its analysis.
The trend of dropping revenue "may be tied to changing venues of care as urgent care centers and occupational medicine centers absorb general surgery work previously done in hospitals," the survey authors wrote.
Average revenue of $1.86 million was reported for general surgeons in 2013, down from $2.11 million in 2010. By comparison, hospital revenue generated was almost $1.42 million for all specialists and $1.57 for all primary care physicians in 2013. The specialists’ average is the lowest Merritt Hawkins has reported since it started the survey in 2002 and is lower than the primary care average for the first time ever, the report noted.
The survey was sent to 5,500 hospitals in January 2013 and was completed by 102 hospital chief financial executives.
Two-year results from STABLE trial unveiled
SAN FRANCISCO – Endovascular repair of complicated type B aortic dissection using a composite device design demonstrates favorable aortic modeling, 2-year results from an ongoing trial showed.
However, patients treated in an acute phase may be prone to aortic growth and require close monitoring, Dr. Joseph V. Lombardi reported at Society for Vascular Surgery Annual Meeting.
Those are key findings the STABLE trial, a prospective, multicenter trial evaluating the safety and effectiveness of a composite endovascular system consisting of TX2 thoracic stent grafts and distal bare metal dissection stents (Zenith Dissection Endovascular System; Cook Medical) for the treatment of complicated type B aortic dissection. The system is commercially available in Europe but not in the United States.
The first published results from STABLE, which was comprised of 40 patients, found a 30-day mortality of 5% and a 1-year all-cause mortality of 10% (J. Vasc. Surg. 2012;55:629-40) "Positive aortic modeling was recognized as well," said Dr. Lombardi, chief of vascular surgery at Cooper University Hospital in Camden, N.J., and the principal STABLE investigator. "The goals for complicated type B aortic dissection are to recognize complications promptly, alleviate branch vessel malperfusion, reduce the risk of rupture, and minimize long-term complications."
Dr. Lombardi updated meeting attendees on 2-year data from the STABLE trial, which is being conducted in the United States, Europe, and Australia. He reported on results from 86 patients with a mean age of 59 years. Nearly three-quarters (73%) were male. The primary endpoint is 30-day mortality and follow-up through 5 years is ongoing.
The researchers observed a mean onset to treatment of 17 days. More than half (64%) presented in an acute phase, which they defined as within 0-14 days of symptom onset, while the remainder presented in a non-acute phase defined as greater than 14 days after symptom onset.
"The anatomical extent of the dissections extended through the visceral aorta in 99% of cases and in the common iliac artery or below in 80% of cases," Dr. Lombardi said. "There were a number of device combinations used depending on the patient’s anatomical presentation. Successful device deployment was seen in all patients, with 100% patency."
The all-cause mortality was 12% at 1 year and 15% at 2 years. The 30-day mortality rate for the 86 patients was 4.7%. Of the 55 patients who had acute presentations, the 30-day mortality rate was 5.5%.
"There were nine deaths between 1 month and 24 months, four of which were determined to be not related to the dissection," he said. "Combined, this translated into a dissection-related mortality of 11% at 24 months."
Between baseline and 2 years the true lumen diameter increased significantly in the descending thoracic aorta and in the more distal abdominal aorta, with a concomitant decrease in false lumen diameter, Dr. Lombardi said. Transaortic growth of 5 mm or greater was observed in the descending thoracic aorta in 21 patients (24%) and the abdominal aorta in 21 patients (24%).
Generalized linear model analysis revealed that the only significant factor for transaortic growth was duration from symptom onset to treatment (P = .05). "The acute phase treatment is associated with a risk of aortic dilation," Dr. Lombardi said. "Patients in this subgroup need to be closely monitored. Longer-term follow-up on STABLE is needed."
The STABLE Trial was sponsored by Cook Medical. Dr. Lombardi disclosed that he has received research grants from Cook Medical.
SAN FRANCISCO – Endovascular repair of complicated type B aortic dissection using a composite device design demonstrates favorable aortic modeling, 2-year results from an ongoing trial showed.
However, patients treated in an acute phase may be prone to aortic growth and require close monitoring, Dr. Joseph V. Lombardi reported at Society for Vascular Surgery Annual Meeting.
Those are key findings the STABLE trial, a prospective, multicenter trial evaluating the safety and effectiveness of a composite endovascular system consisting of TX2 thoracic stent grafts and distal bare metal dissection stents (Zenith Dissection Endovascular System; Cook Medical) for the treatment of complicated type B aortic dissection. The system is commercially available in Europe but not in the United States.
The first published results from STABLE, which was comprised of 40 patients, found a 30-day mortality of 5% and a 1-year all-cause mortality of 10% (J. Vasc. Surg. 2012;55:629-40) "Positive aortic modeling was recognized as well," said Dr. Lombardi, chief of vascular surgery at Cooper University Hospital in Camden, N.J., and the principal STABLE investigator. "The goals for complicated type B aortic dissection are to recognize complications promptly, alleviate branch vessel malperfusion, reduce the risk of rupture, and minimize long-term complications."
Dr. Lombardi updated meeting attendees on 2-year data from the STABLE trial, which is being conducted in the United States, Europe, and Australia. He reported on results from 86 patients with a mean age of 59 years. Nearly three-quarters (73%) were male. The primary endpoint is 30-day mortality and follow-up through 5 years is ongoing.
The researchers observed a mean onset to treatment of 17 days. More than half (64%) presented in an acute phase, which they defined as within 0-14 days of symptom onset, while the remainder presented in a non-acute phase defined as greater than 14 days after symptom onset.
"The anatomical extent of the dissections extended through the visceral aorta in 99% of cases and in the common iliac artery or below in 80% of cases," Dr. Lombardi said. "There were a number of device combinations used depending on the patient’s anatomical presentation. Successful device deployment was seen in all patients, with 100% patency."
The all-cause mortality was 12% at 1 year and 15% at 2 years. The 30-day mortality rate for the 86 patients was 4.7%. Of the 55 patients who had acute presentations, the 30-day mortality rate was 5.5%.
"There were nine deaths between 1 month and 24 months, four of which were determined to be not related to the dissection," he said. "Combined, this translated into a dissection-related mortality of 11% at 24 months."
Between baseline and 2 years the true lumen diameter increased significantly in the descending thoracic aorta and in the more distal abdominal aorta, with a concomitant decrease in false lumen diameter, Dr. Lombardi said. Transaortic growth of 5 mm or greater was observed in the descending thoracic aorta in 21 patients (24%) and the abdominal aorta in 21 patients (24%).
Generalized linear model analysis revealed that the only significant factor for transaortic growth was duration from symptom onset to treatment (P = .05). "The acute phase treatment is associated with a risk of aortic dilation," Dr. Lombardi said. "Patients in this subgroup need to be closely monitored. Longer-term follow-up on STABLE is needed."
The STABLE Trial was sponsored by Cook Medical. Dr. Lombardi disclosed that he has received research grants from Cook Medical.
SAN FRANCISCO – Endovascular repair of complicated type B aortic dissection using a composite device design demonstrates favorable aortic modeling, 2-year results from an ongoing trial showed.
However, patients treated in an acute phase may be prone to aortic growth and require close monitoring, Dr. Joseph V. Lombardi reported at Society for Vascular Surgery Annual Meeting.
Those are key findings the STABLE trial, a prospective, multicenter trial evaluating the safety and effectiveness of a composite endovascular system consisting of TX2 thoracic stent grafts and distal bare metal dissection stents (Zenith Dissection Endovascular System; Cook Medical) for the treatment of complicated type B aortic dissection. The system is commercially available in Europe but not in the United States.
The first published results from STABLE, which was comprised of 40 patients, found a 30-day mortality of 5% and a 1-year all-cause mortality of 10% (J. Vasc. Surg. 2012;55:629-40) "Positive aortic modeling was recognized as well," said Dr. Lombardi, chief of vascular surgery at Cooper University Hospital in Camden, N.J., and the principal STABLE investigator. "The goals for complicated type B aortic dissection are to recognize complications promptly, alleviate branch vessel malperfusion, reduce the risk of rupture, and minimize long-term complications."
Dr. Lombardi updated meeting attendees on 2-year data from the STABLE trial, which is being conducted in the United States, Europe, and Australia. He reported on results from 86 patients with a mean age of 59 years. Nearly three-quarters (73%) were male. The primary endpoint is 30-day mortality and follow-up through 5 years is ongoing.
The researchers observed a mean onset to treatment of 17 days. More than half (64%) presented in an acute phase, which they defined as within 0-14 days of symptom onset, while the remainder presented in a non-acute phase defined as greater than 14 days after symptom onset.
"The anatomical extent of the dissections extended through the visceral aorta in 99% of cases and in the common iliac artery or below in 80% of cases," Dr. Lombardi said. "There were a number of device combinations used depending on the patient’s anatomical presentation. Successful device deployment was seen in all patients, with 100% patency."
The all-cause mortality was 12% at 1 year and 15% at 2 years. The 30-day mortality rate for the 86 patients was 4.7%. Of the 55 patients who had acute presentations, the 30-day mortality rate was 5.5%.
"There were nine deaths between 1 month and 24 months, four of which were determined to be not related to the dissection," he said. "Combined, this translated into a dissection-related mortality of 11% at 24 months."
Between baseline and 2 years the true lumen diameter increased significantly in the descending thoracic aorta and in the more distal abdominal aorta, with a concomitant decrease in false lumen diameter, Dr. Lombardi said. Transaortic growth of 5 mm or greater was observed in the descending thoracic aorta in 21 patients (24%) and the abdominal aorta in 21 patients (24%).
Generalized linear model analysis revealed that the only significant factor for transaortic growth was duration from symptom onset to treatment (P = .05). "The acute phase treatment is associated with a risk of aortic dilation," Dr. Lombardi said. "Patients in this subgroup need to be closely monitored. Longer-term follow-up on STABLE is needed."
The STABLE Trial was sponsored by Cook Medical. Dr. Lombardi disclosed that he has received research grants from Cook Medical.
AT THE SVS ANNUAL MEETING
Major finding: Among patients who underwent endovascular repair of complicated type B aortic dissection with a composite device design, the 30-day mortality rate was 4.7%. Of the 55 patients who had acute presentations, the 30-day mortality rate was 5.5%.
Data source: Results from the ongoing STABLE Trial, a prospective, multicenter study evaluating the safety and effectiveness of a composite endovascular system consisting of TX2 thoracic stent grafts and distal bare metal dissection stents for the treatment of complicated type B aortic dissection.
Disclosures: The STABLE trial was sponsored by Cook Medical. Dr. Lombardi disclosed that he has received research grants from Cook Medical.
Aortic valve-sparing root surgery durable at 1 year
MINNEAPOLIS – Despite the complexity of aortic valve–sparing techniques, early outcomes and 1-year survival were similar to those achieved with valve replacing during root replacement surgery in patients with Marfan syndrome in a prospective registry study.
Major adverse valve-related events (MAVREs) at 1 year were also comparable, although there was an increase, of course, in aortic valve regurgitation with valve sparing (7% vs. 0%), Dr. Joseph Coselli said at the annual meeting of the American Association for Thoracic Surgery.
"Follow-up is needed for this particular incident because we don’t know exactly what’s going to happen to these 2-plus aortic regurgitations," he said. "It’s quite possible they may remain stable over a long period of time and don’t represent a failure of the concept."
In an initial report from the international registry, valve-sparing techniques were the most common, and provided comparable 30-day outcomes in 151 patients (J. Thorac. Cardiovasc. Surg. 2009;137:1124-32).
The current analysis involved 316 patients, aged 4-70 years, who underwent aortic valve–sparing (AVS) (n = 239) or aortic valve–replacing (AVR) (n = 63 mechanical and 14 tissue) root replacement surgery at 19 centers between March 2005 and November 2010. The type of operation was determined by clinical factors, and by surgeon and patient preference. AVR surgery was considered the only option in 17% of patients. At 1 year, clinical follow-up was complete in 98% and imaging follow-up in 93%.
"Interestingly, when we started out this particular collection of patients, over 30% were receiving aortic valve replacement, but toward the end of this observational study, late 2010, virtually almost all patients were receiving aortic valve sparing," said Dr. Coselli, chief of adult cardiac surgery at Baylor College of Medicine, Texas Heart Institute, Houston.
AVS patients were younger (33 vs. 39 years); had smaller sinuses of Valsalva (49 vs. 53 mm); and had less acute dissection (3% vs. 9%), chronic dissection (3% vs. 12%), and previous cardiovascular surgery (5% vs. 14%).
Aortic-sparing techniques required significantly longer cardiopulmonary bypass time (195 vs. 152 minutes) and aortic clamp time (156 vs. 115 minutes), but cut ICU time from 46 hours with AVR surgery to 26 hours, ventilator support time from 12 to 8 hours, and hospital length of stay from 7 to 6 days, Dr. Coselli reported.
At 30 days, MAVREs were reported in 6 patients in the AVR group and 15 in the AVS group (P = .4), with no differences in nonstructural dysfunction (1 vs. 7), embolism (1 vs. 3), or bleeding (2 vs. 3 events).
Two AVS patients required early reoperation: One required same-day reintervention because of coronary artery kinking after a Florida sleeve procedure, and the second needed reintervention because of a coronary pseudoaneurysm 6 days after a David-V procedure, he said. Two early deaths occurred, one in each group, but neither was valve related.
One-year survival rates were 98% after AVS surgery and 97% after AVR surgery (4 vs. 2 deaths; P = .06).
At 1 year, MAVREs occurred in 8 AVR and 35 AVS patients (P = .5). There was no significant difference between AVR and AVS in freedom from valve-related death (1 vs. 2), embolism (2 vs. 4 events), reintervention (0 vs. 1), endocarditis (1 vs. 0), valve thrombosis (0 both), or valve-related morbidity (7 vs. 28), Dr. Coselli said.
The AVS group had significantly more nonstructural dysfunction/structural valve deterioration (23 events vs. 1 event; P = .04), but significantly less bleeding (3 vs. 5 events; P = .01).
In a Cox regression analysis at 1 year, the type of surgery was not associated with overall survival, MAVREs, or any other valve-related outcome, he said.
Dr. Coselli reported research support from St. Jude Medical; an educational grant, consultancy, and royalties from Vascutek Terumo; and research support, speaking for, and steering committee membership with Medtronic. Two coauthors reported consultant/advisory board participation with Medtronic or Edwards Lifesciences.
MINNEAPOLIS – Despite the complexity of aortic valve–sparing techniques, early outcomes and 1-year survival were similar to those achieved with valve replacing during root replacement surgery in patients with Marfan syndrome in a prospective registry study.
Major adverse valve-related events (MAVREs) at 1 year were also comparable, although there was an increase, of course, in aortic valve regurgitation with valve sparing (7% vs. 0%), Dr. Joseph Coselli said at the annual meeting of the American Association for Thoracic Surgery.
"Follow-up is needed for this particular incident because we don’t know exactly what’s going to happen to these 2-plus aortic regurgitations," he said. "It’s quite possible they may remain stable over a long period of time and don’t represent a failure of the concept."
In an initial report from the international registry, valve-sparing techniques were the most common, and provided comparable 30-day outcomes in 151 patients (J. Thorac. Cardiovasc. Surg. 2009;137:1124-32).
The current analysis involved 316 patients, aged 4-70 years, who underwent aortic valve–sparing (AVS) (n = 239) or aortic valve–replacing (AVR) (n = 63 mechanical and 14 tissue) root replacement surgery at 19 centers between March 2005 and November 2010. The type of operation was determined by clinical factors, and by surgeon and patient preference. AVR surgery was considered the only option in 17% of patients. At 1 year, clinical follow-up was complete in 98% and imaging follow-up in 93%.
"Interestingly, when we started out this particular collection of patients, over 30% were receiving aortic valve replacement, but toward the end of this observational study, late 2010, virtually almost all patients were receiving aortic valve sparing," said Dr. Coselli, chief of adult cardiac surgery at Baylor College of Medicine, Texas Heart Institute, Houston.
AVS patients were younger (33 vs. 39 years); had smaller sinuses of Valsalva (49 vs. 53 mm); and had less acute dissection (3% vs. 9%), chronic dissection (3% vs. 12%), and previous cardiovascular surgery (5% vs. 14%).
Aortic-sparing techniques required significantly longer cardiopulmonary bypass time (195 vs. 152 minutes) and aortic clamp time (156 vs. 115 minutes), but cut ICU time from 46 hours with AVR surgery to 26 hours, ventilator support time from 12 to 8 hours, and hospital length of stay from 7 to 6 days, Dr. Coselli reported.
At 30 days, MAVREs were reported in 6 patients in the AVR group and 15 in the AVS group (P = .4), with no differences in nonstructural dysfunction (1 vs. 7), embolism (1 vs. 3), or bleeding (2 vs. 3 events).
Two AVS patients required early reoperation: One required same-day reintervention because of coronary artery kinking after a Florida sleeve procedure, and the second needed reintervention because of a coronary pseudoaneurysm 6 days after a David-V procedure, he said. Two early deaths occurred, one in each group, but neither was valve related.
One-year survival rates were 98% after AVS surgery and 97% after AVR surgery (4 vs. 2 deaths; P = .06).
At 1 year, MAVREs occurred in 8 AVR and 35 AVS patients (P = .5). There was no significant difference between AVR and AVS in freedom from valve-related death (1 vs. 2), embolism (2 vs. 4 events), reintervention (0 vs. 1), endocarditis (1 vs. 0), valve thrombosis (0 both), or valve-related morbidity (7 vs. 28), Dr. Coselli said.
The AVS group had significantly more nonstructural dysfunction/structural valve deterioration (23 events vs. 1 event; P = .04), but significantly less bleeding (3 vs. 5 events; P = .01).
In a Cox regression analysis at 1 year, the type of surgery was not associated with overall survival, MAVREs, or any other valve-related outcome, he said.
Dr. Coselli reported research support from St. Jude Medical; an educational grant, consultancy, and royalties from Vascutek Terumo; and research support, speaking for, and steering committee membership with Medtronic. Two coauthors reported consultant/advisory board participation with Medtronic or Edwards Lifesciences.
MINNEAPOLIS – Despite the complexity of aortic valve–sparing techniques, early outcomes and 1-year survival were similar to those achieved with valve replacing during root replacement surgery in patients with Marfan syndrome in a prospective registry study.
Major adverse valve-related events (MAVREs) at 1 year were also comparable, although there was an increase, of course, in aortic valve regurgitation with valve sparing (7% vs. 0%), Dr. Joseph Coselli said at the annual meeting of the American Association for Thoracic Surgery.
"Follow-up is needed for this particular incident because we don’t know exactly what’s going to happen to these 2-plus aortic regurgitations," he said. "It’s quite possible they may remain stable over a long period of time and don’t represent a failure of the concept."
In an initial report from the international registry, valve-sparing techniques were the most common, and provided comparable 30-day outcomes in 151 patients (J. Thorac. Cardiovasc. Surg. 2009;137:1124-32).
The current analysis involved 316 patients, aged 4-70 years, who underwent aortic valve–sparing (AVS) (n = 239) or aortic valve–replacing (AVR) (n = 63 mechanical and 14 tissue) root replacement surgery at 19 centers between March 2005 and November 2010. The type of operation was determined by clinical factors, and by surgeon and patient preference. AVR surgery was considered the only option in 17% of patients. At 1 year, clinical follow-up was complete in 98% and imaging follow-up in 93%.
"Interestingly, when we started out this particular collection of patients, over 30% were receiving aortic valve replacement, but toward the end of this observational study, late 2010, virtually almost all patients were receiving aortic valve sparing," said Dr. Coselli, chief of adult cardiac surgery at Baylor College of Medicine, Texas Heart Institute, Houston.
AVS patients were younger (33 vs. 39 years); had smaller sinuses of Valsalva (49 vs. 53 mm); and had less acute dissection (3% vs. 9%), chronic dissection (3% vs. 12%), and previous cardiovascular surgery (5% vs. 14%).
Aortic-sparing techniques required significantly longer cardiopulmonary bypass time (195 vs. 152 minutes) and aortic clamp time (156 vs. 115 minutes), but cut ICU time from 46 hours with AVR surgery to 26 hours, ventilator support time from 12 to 8 hours, and hospital length of stay from 7 to 6 days, Dr. Coselli reported.
At 30 days, MAVREs were reported in 6 patients in the AVR group and 15 in the AVS group (P = .4), with no differences in nonstructural dysfunction (1 vs. 7), embolism (1 vs. 3), or bleeding (2 vs. 3 events).
Two AVS patients required early reoperation: One required same-day reintervention because of coronary artery kinking after a Florida sleeve procedure, and the second needed reintervention because of a coronary pseudoaneurysm 6 days after a David-V procedure, he said. Two early deaths occurred, one in each group, but neither was valve related.
One-year survival rates were 98% after AVS surgery and 97% after AVR surgery (4 vs. 2 deaths; P = .06).
At 1 year, MAVREs occurred in 8 AVR and 35 AVS patients (P = .5). There was no significant difference between AVR and AVS in freedom from valve-related death (1 vs. 2), embolism (2 vs. 4 events), reintervention (0 vs. 1), endocarditis (1 vs. 0), valve thrombosis (0 both), or valve-related morbidity (7 vs. 28), Dr. Coselli said.
The AVS group had significantly more nonstructural dysfunction/structural valve deterioration (23 events vs. 1 event; P = .04), but significantly less bleeding (3 vs. 5 events; P = .01).
In a Cox regression analysis at 1 year, the type of surgery was not associated with overall survival, MAVREs, or any other valve-related outcome, he said.
Dr. Coselli reported research support from St. Jude Medical; an educational grant, consultancy, and royalties from Vascutek Terumo; and research support, speaking for, and steering committee membership with Medtronic. Two coauthors reported consultant/advisory board participation with Medtronic or Edwards Lifesciences.
AT THE AATS ANNUAL MEETING
Major finding: One-year survival rates were 98% after AVS surgery and 97% after AVR surgery (4 vs. 2 deaths; P = .06).
Data source: Prospective, international, observational registry study in 316 Marfan syndrome patients undergoing aortic root surgery.
Disclosures: Dr. Coselli reported research support from St. Jude Medical; an educational grant, consultancy, and royalties from Vascutek Terumo; and research support, speaking for, and steering committee membership with Medtronic. Two coauthors reported consultant/advisory board participation with Medtronic or Edwards Lifesciences.
Current but not past smokers at extra postoperative risk
Current smoking is associated with an increased risk of mortality and other adverse outcomes following major surgery, but past smoking is not, according to a report published online June 19 in JAMA Surgery.
Current smoking correlates with these adverse outcomes even in patients who don’t have obvious smoking-related disease such cardiovascular disease, chronic pulmonary disorders, or cancer, which suggests that smoking may exert its deleterious effects through acute or subclinical chronic vascular and respiratory pathologic mechanisms, said Dr. Khaled M. Musallam of the American University of Beirut (Lebanon) Medical Center and his associates.
Current smoking is associated with an increased risk of mortality and other adverse outcomes following major surgery, but past smoking is not, according to a recent report published in JAMA Surgery.
Since smoking cessation has clear benefits on morbidity and mortality in the surgical setting, "surgical teams should be more involved in the ongoing efforts to optimize measures for smoking control," they wrote.
"Surgery provides a teachable environment for smoking cessation. Unlike the long-term consequences of smoking, the acute consequences of smoking on patients’ postoperative outcomes can provide a strong motive for quitting," the investigators said.
Dr. Musallam and his colleagues examined the effect of smoking on surgical outcomes using data from the American College of Surgeons National Surgical Quality Improvement Program (NSQIP), which includes a registry that provides feedback to participating hospitals regarding 30-day risk-adjusted surgical morbidity and mortality.
For this study, they analyzed data on 607,558 patients undergoing major surgery at more than 200 participating hospitals during a 2-year period in the United States, Canada, Lebanon, and the United Arab Emirates. The mean age of the patients was 56 years (range, 16-90 years); 43% were men and 57% were women.
A total of 125,192 patients (21%) were current smokers and 78,763 (13%) were past smokers who had quit at least 1 year before surgery. The remaining patients had never smoked.
Only current smokers showed an increased likelihood of 30-day mortality. They also were at greater risk for adverse arterial events such as MI or stroke, as well as for adverse respiratory events such as pneumonia, need for intubation, and need for a ventilator, within 30 days of surgery, the investigators said (JAMA Surg. 2013 June 19 [doi:10.1001/jamasurg.2013.2360]).
The higher risk of these adverse outcomes occurred with smokers across all age groups but was particularly notable among those older than age 40 years. It was seen in both sexes, among those undergoing inpatient as well as outpatient procedures, in patients who had general as well as other types of anesthesia, across a variety of surgical subspecialties, and in both elective and emergency surgery cases.
The association between current smoking and adverse outcomes also remained robust in a sensitivity analysis, Dr. Musallam and his associates said.
There was a dose-response effect in an analysis of patients’ smoking history, with the likelihood of adverse arterial and respiratory events increasing in tandem with increasing pack-years of smoking, but even current "light" smokers who had fewer than 10 pack-years of smoking history were at increased risk for postoperative mortality and morbidity.
"These findings encourage ongoing efforts to implement smoking cessation programs," Dr. Musallam and his associates said.
"Early intervention in heavy smokers is warranted, especially because the effect of smoking on postoperative arterial and respiratory morbidity seems to be dose dependent. However, because smokers with a cigarette smoking history of less than 10 pack-years are also at risk of postoperative death, recent and light smokers should also be targeted," they suggested.
Dr. Musallam and his associates reported no financial conflicts of interest.
Current smoking is associated with an increased risk of mortality and other adverse outcomes following major surgery, but past smoking is not, according to a report published online June 19 in JAMA Surgery.
Current smoking correlates with these adverse outcomes even in patients who don’t have obvious smoking-related disease such cardiovascular disease, chronic pulmonary disorders, or cancer, which suggests that smoking may exert its deleterious effects through acute or subclinical chronic vascular and respiratory pathologic mechanisms, said Dr. Khaled M. Musallam of the American University of Beirut (Lebanon) Medical Center and his associates.
Current smoking is associated with an increased risk of mortality and other adverse outcomes following major surgery, but past smoking is not, according to a recent report published in JAMA Surgery.
Since smoking cessation has clear benefits on morbidity and mortality in the surgical setting, "surgical teams should be more involved in the ongoing efforts to optimize measures for smoking control," they wrote.
"Surgery provides a teachable environment for smoking cessation. Unlike the long-term consequences of smoking, the acute consequences of smoking on patients’ postoperative outcomes can provide a strong motive for quitting," the investigators said.
Dr. Musallam and his colleagues examined the effect of smoking on surgical outcomes using data from the American College of Surgeons National Surgical Quality Improvement Program (NSQIP), which includes a registry that provides feedback to participating hospitals regarding 30-day risk-adjusted surgical morbidity and mortality.
For this study, they analyzed data on 607,558 patients undergoing major surgery at more than 200 participating hospitals during a 2-year period in the United States, Canada, Lebanon, and the United Arab Emirates. The mean age of the patients was 56 years (range, 16-90 years); 43% were men and 57% were women.
A total of 125,192 patients (21%) were current smokers and 78,763 (13%) were past smokers who had quit at least 1 year before surgery. The remaining patients had never smoked.
Only current smokers showed an increased likelihood of 30-day mortality. They also were at greater risk for adverse arterial events such as MI or stroke, as well as for adverse respiratory events such as pneumonia, need for intubation, and need for a ventilator, within 30 days of surgery, the investigators said (JAMA Surg. 2013 June 19 [doi:10.1001/jamasurg.2013.2360]).
The higher risk of these adverse outcomes occurred with smokers across all age groups but was particularly notable among those older than age 40 years. It was seen in both sexes, among those undergoing inpatient as well as outpatient procedures, in patients who had general as well as other types of anesthesia, across a variety of surgical subspecialties, and in both elective and emergency surgery cases.
The association between current smoking and adverse outcomes also remained robust in a sensitivity analysis, Dr. Musallam and his associates said.
There was a dose-response effect in an analysis of patients’ smoking history, with the likelihood of adverse arterial and respiratory events increasing in tandem with increasing pack-years of smoking, but even current "light" smokers who had fewer than 10 pack-years of smoking history were at increased risk for postoperative mortality and morbidity.
"These findings encourage ongoing efforts to implement smoking cessation programs," Dr. Musallam and his associates said.
"Early intervention in heavy smokers is warranted, especially because the effect of smoking on postoperative arterial and respiratory morbidity seems to be dose dependent. However, because smokers with a cigarette smoking history of less than 10 pack-years are also at risk of postoperative death, recent and light smokers should also be targeted," they suggested.
Dr. Musallam and his associates reported no financial conflicts of interest.
Current smoking is associated with an increased risk of mortality and other adverse outcomes following major surgery, but past smoking is not, according to a report published online June 19 in JAMA Surgery.
Current smoking correlates with these adverse outcomes even in patients who don’t have obvious smoking-related disease such cardiovascular disease, chronic pulmonary disorders, or cancer, which suggests that smoking may exert its deleterious effects through acute or subclinical chronic vascular and respiratory pathologic mechanisms, said Dr. Khaled M. Musallam of the American University of Beirut (Lebanon) Medical Center and his associates.
Current smoking is associated with an increased risk of mortality and other adverse outcomes following major surgery, but past smoking is not, according to a recent report published in JAMA Surgery.
Since smoking cessation has clear benefits on morbidity and mortality in the surgical setting, "surgical teams should be more involved in the ongoing efforts to optimize measures for smoking control," they wrote.
"Surgery provides a teachable environment for smoking cessation. Unlike the long-term consequences of smoking, the acute consequences of smoking on patients’ postoperative outcomes can provide a strong motive for quitting," the investigators said.
Dr. Musallam and his colleagues examined the effect of smoking on surgical outcomes using data from the American College of Surgeons National Surgical Quality Improvement Program (NSQIP), which includes a registry that provides feedback to participating hospitals regarding 30-day risk-adjusted surgical morbidity and mortality.
For this study, they analyzed data on 607,558 patients undergoing major surgery at more than 200 participating hospitals during a 2-year period in the United States, Canada, Lebanon, and the United Arab Emirates. The mean age of the patients was 56 years (range, 16-90 years); 43% were men and 57% were women.
A total of 125,192 patients (21%) were current smokers and 78,763 (13%) were past smokers who had quit at least 1 year before surgery. The remaining patients had never smoked.
Only current smokers showed an increased likelihood of 30-day mortality. They also were at greater risk for adverse arterial events such as MI or stroke, as well as for adverse respiratory events such as pneumonia, need for intubation, and need for a ventilator, within 30 days of surgery, the investigators said (JAMA Surg. 2013 June 19 [doi:10.1001/jamasurg.2013.2360]).
The higher risk of these adverse outcomes occurred with smokers across all age groups but was particularly notable among those older than age 40 years. It was seen in both sexes, among those undergoing inpatient as well as outpatient procedures, in patients who had general as well as other types of anesthesia, across a variety of surgical subspecialties, and in both elective and emergency surgery cases.
The association between current smoking and adverse outcomes also remained robust in a sensitivity analysis, Dr. Musallam and his associates said.
There was a dose-response effect in an analysis of patients’ smoking history, with the likelihood of adverse arterial and respiratory events increasing in tandem with increasing pack-years of smoking, but even current "light" smokers who had fewer than 10 pack-years of smoking history were at increased risk for postoperative mortality and morbidity.
"These findings encourage ongoing efforts to implement smoking cessation programs," Dr. Musallam and his associates said.
"Early intervention in heavy smokers is warranted, especially because the effect of smoking on postoperative arterial and respiratory morbidity seems to be dose dependent. However, because smokers with a cigarette smoking history of less than 10 pack-years are also at risk of postoperative death, recent and light smokers should also be targeted," they suggested.
Dr. Musallam and his associates reported no financial conflicts of interest.
FROM JAMA SURGERY
Major Finding: Only current smokers, not past or never smokers, showed an increased likelihood of 30-day mortality, MI, stroke, pneumonia, the need for intubation, and the need for a ventilator.
Data Source: An analysis of data on 30-day mortality and morbidity in 607,558 adults undergoing major surgery in four countries during a 2-year period.
Disclosures: Dr. Musallam and his associates reported no financial conflicts of interest.
Wedge resection in NSCLC: Is 15 mm the magic margin?
MINNEAPOLIS – Increasing the surgical margin length up to 15 mm during wedge resection of small lung cancer tumors significantly lowered the risk of local recurrence among 474 consecutive patients.
No additional benefit was observed, however, beyond 15 mm, said Dr. Kamran Mohiuddin, a surgical research fellow at Brigham and Women’s Hospital, Boston.
Compared with a margin length of 5 mm, the adjusted risk of local recurrence was estimated to be 45% lower with a margin length of 10 mm (hazard ratio, 0.55), 59% lower with a 15-mm margin (HR, 0.41), and 54% lower with a 20-mm margin (HR, 0.46).
"The downward trend flattens out, indicating diminished benefit of increasing the margin length," he said at the annual meeting of the American Association for Thoracic Surgery.
Currently, the data are unclear regarding the optimal margin length for wedge resection of small non–small cell lung cancer (NSCLC) tumors of less than 2 cm. Wedge resections are associated with margins less than 1 cm and a high risk for locoregional recurrence (Ann. Surg. Oncol. 2007;14:2400-5), with a multicenter, prospective study suggesting that the optimal margin length should be larger than the maximum tumor diameter (Ann. Thorac. Surg. 2004;77:415-20).
When asked during a discussion of the analysis whether a more aggressive resection or segmentectomy would be performed if margins are found in the operating room to be inadequate based on the current results, senior author Dr. Scott J. Swanson, an ACS Fellow and director of minimally invasive thoracic surgery at the hospital, said they are taking the results forward into practice, but that it’s unclear whether 15 mm is the optimal number to target.
"Is 15 mm the correct margin? I am not sure we know the answer in all cases, but it is a useful number to keep in the surgeon’s head when we are doing resections for tumors that are 2 cm or less," he said in an interview. "A 15-mm margin seems to be a better target to aim for than margin length to tumor diameter ratio of greater than 1, as suggested by other investigators."
The current analysis included data from all patients, aged 21-85 years, who underwent wedge resection for NSCLC 2 cm or less at their institution between January 2001 and August 2011. Margin length, defined as the distance from the tumor to the closest stapled resection margin, was 0.1-0.5 cm in 36%, 0.6-1.0 cm in 25.5%, 1.1-2.0 cm in 28.5%, and greater than 2 cm in 10%.
The mean tumor size was 1.33 cm, the location of the tumor was the right upper lobe in the majority (36%), and video-assisted thoracic surgery (VATS) was used in 57.5%. The patients’ mean forced expiratory volume in 1 second (FEV1) was 79.8%, and the mean age was 68.5 years.
Perioperative death occurred in 1 patient and at least one major complication in 41 patients, Dr. Mohiuddin said.
The local recurrence rate was 5.8% at 1 year, 11.3% at 2 years, and 16.8% at 3 years. Median follow-up was 3.9 years.
In multivariate regression analysis, increased margin length was significantly associated with a lower risk of local recurrence, with evidence of diminished additional benefit beyond a length of 15 mm (P = .031), he said. The analysis adjusted for FEV1, chronic obstructive pulmonary disease, smoking, diabetes, tumor size, tumor lobe location, location within the hemothorax, surgeon, whether VATS or open surgery was used, and whether or not nodes were sampled.
Dr. Mohiuddin and his coauthors reported having no financial disclosures.
MINNEAPOLIS – Increasing the surgical margin length up to 15 mm during wedge resection of small lung cancer tumors significantly lowered the risk of local recurrence among 474 consecutive patients.
No additional benefit was observed, however, beyond 15 mm, said Dr. Kamran Mohiuddin, a surgical research fellow at Brigham and Women’s Hospital, Boston.
Compared with a margin length of 5 mm, the adjusted risk of local recurrence was estimated to be 45% lower with a margin length of 10 mm (hazard ratio, 0.55), 59% lower with a 15-mm margin (HR, 0.41), and 54% lower with a 20-mm margin (HR, 0.46).
"The downward trend flattens out, indicating diminished benefit of increasing the margin length," he said at the annual meeting of the American Association for Thoracic Surgery.
Currently, the data are unclear regarding the optimal margin length for wedge resection of small non–small cell lung cancer (NSCLC) tumors of less than 2 cm. Wedge resections are associated with margins less than 1 cm and a high risk for locoregional recurrence (Ann. Surg. Oncol. 2007;14:2400-5), with a multicenter, prospective study suggesting that the optimal margin length should be larger than the maximum tumor diameter (Ann. Thorac. Surg. 2004;77:415-20).
When asked during a discussion of the analysis whether a more aggressive resection or segmentectomy would be performed if margins are found in the operating room to be inadequate based on the current results, senior author Dr. Scott J. Swanson, an ACS Fellow and director of minimally invasive thoracic surgery at the hospital, said they are taking the results forward into practice, but that it’s unclear whether 15 mm is the optimal number to target.
"Is 15 mm the correct margin? I am not sure we know the answer in all cases, but it is a useful number to keep in the surgeon’s head when we are doing resections for tumors that are 2 cm or less," he said in an interview. "A 15-mm margin seems to be a better target to aim for than margin length to tumor diameter ratio of greater than 1, as suggested by other investigators."
The current analysis included data from all patients, aged 21-85 years, who underwent wedge resection for NSCLC 2 cm or less at their institution between January 2001 and August 2011. Margin length, defined as the distance from the tumor to the closest stapled resection margin, was 0.1-0.5 cm in 36%, 0.6-1.0 cm in 25.5%, 1.1-2.0 cm in 28.5%, and greater than 2 cm in 10%.
The mean tumor size was 1.33 cm, the location of the tumor was the right upper lobe in the majority (36%), and video-assisted thoracic surgery (VATS) was used in 57.5%. The patients’ mean forced expiratory volume in 1 second (FEV1) was 79.8%, and the mean age was 68.5 years.
Perioperative death occurred in 1 patient and at least one major complication in 41 patients, Dr. Mohiuddin said.
The local recurrence rate was 5.8% at 1 year, 11.3% at 2 years, and 16.8% at 3 years. Median follow-up was 3.9 years.
In multivariate regression analysis, increased margin length was significantly associated with a lower risk of local recurrence, with evidence of diminished additional benefit beyond a length of 15 mm (P = .031), he said. The analysis adjusted for FEV1, chronic obstructive pulmonary disease, smoking, diabetes, tumor size, tumor lobe location, location within the hemothorax, surgeon, whether VATS or open surgery was used, and whether or not nodes were sampled.
Dr. Mohiuddin and his coauthors reported having no financial disclosures.
MINNEAPOLIS – Increasing the surgical margin length up to 15 mm during wedge resection of small lung cancer tumors significantly lowered the risk of local recurrence among 474 consecutive patients.
No additional benefit was observed, however, beyond 15 mm, said Dr. Kamran Mohiuddin, a surgical research fellow at Brigham and Women’s Hospital, Boston.
Compared with a margin length of 5 mm, the adjusted risk of local recurrence was estimated to be 45% lower with a margin length of 10 mm (hazard ratio, 0.55), 59% lower with a 15-mm margin (HR, 0.41), and 54% lower with a 20-mm margin (HR, 0.46).
"The downward trend flattens out, indicating diminished benefit of increasing the margin length," he said at the annual meeting of the American Association for Thoracic Surgery.
Currently, the data are unclear regarding the optimal margin length for wedge resection of small non–small cell lung cancer (NSCLC) tumors of less than 2 cm. Wedge resections are associated with margins less than 1 cm and a high risk for locoregional recurrence (Ann. Surg. Oncol. 2007;14:2400-5), with a multicenter, prospective study suggesting that the optimal margin length should be larger than the maximum tumor diameter (Ann. Thorac. Surg. 2004;77:415-20).
When asked during a discussion of the analysis whether a more aggressive resection or segmentectomy would be performed if margins are found in the operating room to be inadequate based on the current results, senior author Dr. Scott J. Swanson, an ACS Fellow and director of minimally invasive thoracic surgery at the hospital, said they are taking the results forward into practice, but that it’s unclear whether 15 mm is the optimal number to target.
"Is 15 mm the correct margin? I am not sure we know the answer in all cases, but it is a useful number to keep in the surgeon’s head when we are doing resections for tumors that are 2 cm or less," he said in an interview. "A 15-mm margin seems to be a better target to aim for than margin length to tumor diameter ratio of greater than 1, as suggested by other investigators."
The current analysis included data from all patients, aged 21-85 years, who underwent wedge resection for NSCLC 2 cm or less at their institution between January 2001 and August 2011. Margin length, defined as the distance from the tumor to the closest stapled resection margin, was 0.1-0.5 cm in 36%, 0.6-1.0 cm in 25.5%, 1.1-2.0 cm in 28.5%, and greater than 2 cm in 10%.
The mean tumor size was 1.33 cm, the location of the tumor was the right upper lobe in the majority (36%), and video-assisted thoracic surgery (VATS) was used in 57.5%. The patients’ mean forced expiratory volume in 1 second (FEV1) was 79.8%, and the mean age was 68.5 years.
Perioperative death occurred in 1 patient and at least one major complication in 41 patients, Dr. Mohiuddin said.
The local recurrence rate was 5.8% at 1 year, 11.3% at 2 years, and 16.8% at 3 years. Median follow-up was 3.9 years.
In multivariate regression analysis, increased margin length was significantly associated with a lower risk of local recurrence, with evidence of diminished additional benefit beyond a length of 15 mm (P = .031), he said. The analysis adjusted for FEV1, chronic obstructive pulmonary disease, smoking, diabetes, tumor size, tumor lobe location, location within the hemothorax, surgeon, whether VATS or open surgery was used, and whether or not nodes were sampled.
Dr. Mohiuddin and his coauthors reported having no financial disclosures.
AT THE AATS ANNUAL MEETING
Major finding: Compared with a margin length of 5 mm, the adjusted risk of local recurrence was estimated to be 45% lower with a margin length of 10 mm (hazard ratio, 0.55), 59% lower with a margin length of 15 mm (HR, 0.41), and 54% lower with a 20-mm margin (HR, 0.46).
Data source: Retrospective analysis of 474 consecutive patients undergoing pulmonary wedge resection for non–small cell lung cancer tumors up to 2 cm.
Disclosures: Dr. Mohiuddin and his coauthors reported having no financial disclosures.
Improved presurgery impulse control screening needed in Parkinson’s
SYDNEY, AUSTRALIA – Many Parkinson’s disease centers performing deep brain stimulation surgery are not using formal, standardized screening for impulse control disorders in pre- or postsurgical patients, according to a large survey of Parkinson Study Group centers.
Deep brain stimulation surgery is known to increase impulsivity, and standard practice is to identify and treat impulse control disorders in patients before surgery, according to lead author Dr. Nawaz Hack, a junior fellow in movement disorders at the University of Florida, Gainesville, and his colleagues.
"Surgery will improve their motor symptoms, but it may make their impulsivity worse, especially if you don’t screen and appropriately identify it," Dr. Hack said. "But if you catch it early through a standardized screening, you can address it."
The researchers surveyed 48 Parkinson Study Group centers, 97% of which performed deep brain stimulation surgery and 67% of which said they served a population of over 500 patients a year.
The results showed that only 23% of sites employed a formal battery of tests for impulsive and compulsive behavior and that 7% did not report screening for impulse control disorders.
Speaking at a poster session at the international congress of Parkinson’s Disease and Movement Disorders, Dr. Hack said that the majority of sites were employing a more ad-hoc approach to screening for impulse control disorders, using questions that were not necessarily standardized.
The survey found that 80% of responding centers used a neuropsychologist to screen for potential behavioral issues but only 32% used a psychiatrist, suggesting that most are focused on identifying the problem but do not necessarily have the facilities to manage and treat it.
There was also a wide variety of approaches taken to manage impulse control issues in presurgical patients.
Seventy-nine percent of patients with an impulse control disorder were treated with medication reduction, although there were 10 different strategies employed across centers, the survey’s authors reported.
"This is what happens in the centers that understand Parkinson’s disease – these are centers that are knowledgeable – so if we’re deficient there, what’s happening?" Dr. Hack said.
The concern was that patients whose impulsivity becomes exacerbated by surgery were more likely to be lost to follow up because of the potential financial consequences of their behavioral disorder.
"If you’re impulsive before, and you’re not treated or screened, you will be seriously impulsive after, so they’ll leave the operating room, they’ll go home, and they’ll start doing behaviors that are literally destroying their family, their lives, and their finances," Dr. Hack said.
However, he said that identifying patients with impulse control disorders could be difficult because patients were sometimes reluctant to volunteer information on behaviors that might indicate the presence of impulsivity, such as hypersexuality or problem gambling.
Nearly three-quarters of centers (72%) reported observing impulse control disorders among deep brain stimulation surgery patients, and 68% reported it in postoperative patients.
Most centers (79%) did not feel that the choice of brain target, whether subthalamic nucleus or globus pallidus, was influential in behavioral disorders.
Dr. Hack did not declare any financial conflicts of interest with the research.
SYDNEY, AUSTRALIA – Many Parkinson’s disease centers performing deep brain stimulation surgery are not using formal, standardized screening for impulse control disorders in pre- or postsurgical patients, according to a large survey of Parkinson Study Group centers.
Deep brain stimulation surgery is known to increase impulsivity, and standard practice is to identify and treat impulse control disorders in patients before surgery, according to lead author Dr. Nawaz Hack, a junior fellow in movement disorders at the University of Florida, Gainesville, and his colleagues.
"Surgery will improve their motor symptoms, but it may make their impulsivity worse, especially if you don’t screen and appropriately identify it," Dr. Hack said. "But if you catch it early through a standardized screening, you can address it."
The researchers surveyed 48 Parkinson Study Group centers, 97% of which performed deep brain stimulation surgery and 67% of which said they served a population of over 500 patients a year.
The results showed that only 23% of sites employed a formal battery of tests for impulsive and compulsive behavior and that 7% did not report screening for impulse control disorders.
Speaking at a poster session at the international congress of Parkinson’s Disease and Movement Disorders, Dr. Hack said that the majority of sites were employing a more ad-hoc approach to screening for impulse control disorders, using questions that were not necessarily standardized.
The survey found that 80% of responding centers used a neuropsychologist to screen for potential behavioral issues but only 32% used a psychiatrist, suggesting that most are focused on identifying the problem but do not necessarily have the facilities to manage and treat it.
There was also a wide variety of approaches taken to manage impulse control issues in presurgical patients.
Seventy-nine percent of patients with an impulse control disorder were treated with medication reduction, although there were 10 different strategies employed across centers, the survey’s authors reported.
"This is what happens in the centers that understand Parkinson’s disease – these are centers that are knowledgeable – so if we’re deficient there, what’s happening?" Dr. Hack said.
The concern was that patients whose impulsivity becomes exacerbated by surgery were more likely to be lost to follow up because of the potential financial consequences of their behavioral disorder.
"If you’re impulsive before, and you’re not treated or screened, you will be seriously impulsive after, so they’ll leave the operating room, they’ll go home, and they’ll start doing behaviors that are literally destroying their family, their lives, and their finances," Dr. Hack said.
However, he said that identifying patients with impulse control disorders could be difficult because patients were sometimes reluctant to volunteer information on behaviors that might indicate the presence of impulsivity, such as hypersexuality or problem gambling.
Nearly three-quarters of centers (72%) reported observing impulse control disorders among deep brain stimulation surgery patients, and 68% reported it in postoperative patients.
Most centers (79%) did not feel that the choice of brain target, whether subthalamic nucleus or globus pallidus, was influential in behavioral disorders.
Dr. Hack did not declare any financial conflicts of interest with the research.
SYDNEY, AUSTRALIA – Many Parkinson’s disease centers performing deep brain stimulation surgery are not using formal, standardized screening for impulse control disorders in pre- or postsurgical patients, according to a large survey of Parkinson Study Group centers.
Deep brain stimulation surgery is known to increase impulsivity, and standard practice is to identify and treat impulse control disorders in patients before surgery, according to lead author Dr. Nawaz Hack, a junior fellow in movement disorders at the University of Florida, Gainesville, and his colleagues.
"Surgery will improve their motor symptoms, but it may make their impulsivity worse, especially if you don’t screen and appropriately identify it," Dr. Hack said. "But if you catch it early through a standardized screening, you can address it."
The researchers surveyed 48 Parkinson Study Group centers, 97% of which performed deep brain stimulation surgery and 67% of which said they served a population of over 500 patients a year.
The results showed that only 23% of sites employed a formal battery of tests for impulsive and compulsive behavior and that 7% did not report screening for impulse control disorders.
Speaking at a poster session at the international congress of Parkinson’s Disease and Movement Disorders, Dr. Hack said that the majority of sites were employing a more ad-hoc approach to screening for impulse control disorders, using questions that were not necessarily standardized.
The survey found that 80% of responding centers used a neuropsychologist to screen for potential behavioral issues but only 32% used a psychiatrist, suggesting that most are focused on identifying the problem but do not necessarily have the facilities to manage and treat it.
There was also a wide variety of approaches taken to manage impulse control issues in presurgical patients.
Seventy-nine percent of patients with an impulse control disorder were treated with medication reduction, although there were 10 different strategies employed across centers, the survey’s authors reported.
"This is what happens in the centers that understand Parkinson’s disease – these are centers that are knowledgeable – so if we’re deficient there, what’s happening?" Dr. Hack said.
The concern was that patients whose impulsivity becomes exacerbated by surgery were more likely to be lost to follow up because of the potential financial consequences of their behavioral disorder.
"If you’re impulsive before, and you’re not treated or screened, you will be seriously impulsive after, so they’ll leave the operating room, they’ll go home, and they’ll start doing behaviors that are literally destroying their family, their lives, and their finances," Dr. Hack said.
However, he said that identifying patients with impulse control disorders could be difficult because patients were sometimes reluctant to volunteer information on behaviors that might indicate the presence of impulsivity, such as hypersexuality or problem gambling.
Nearly three-quarters of centers (72%) reported observing impulse control disorders among deep brain stimulation surgery patients, and 68% reported it in postoperative patients.
Most centers (79%) did not feel that the choice of brain target, whether subthalamic nucleus or globus pallidus, was influential in behavioral disorders.
Dr. Hack did not declare any financial conflicts of interest with the research.
AT THE 2013 MDS INTERNATIONAL CONGRESS
Major finding: Only 23% of sites employed a formal battery of tests for impulsive and compulsive behavior and 7% did not report screening for impulse control disorders.
Data source: Survey of 48 Parkinson Study Group centers.
Disclosures: Dr. Hack did not declare any financial conflicts of interest.
Study IDs predictors of unplanned hospital readmission after CEA
SAN FRANCISCO – The 30-day unplanned readmission rate following carotid endarterectomy was 6.5% in a single-center study.
In addition, four variables were significantly associated with unplanned readmission: in-hospital postoperative congestive heart failure (CHF) exacerbation; in-hospital postoperative stroke; in-hospital postoperative hematoma; and prior coronary artery bypass graft (CABG).
"Whether these complications are completely avoidable is unknown, but we do identify a group of patients who would probably benefit from more comprehensive discharge planning and careful postdischarge care," Dr. Karen J. Ho said at the Society for Vascular Surgery annual meeting.
According to a study of Medicare claims data from 2003 to 2004, 20% of Medicare beneficiaries discharged from a hospital were rehospitalized within 30 days (N. Eng. J. Med. 2009;360:1418-28). The 30-day rehospitalization rate after vascular surgery was 24%, "the highest of all surgical specialties examined in the study," said Dr. Ho of the surgery department at Brigham and Women’s Hospital, Boston, who was not involved with the published study. "Medicare has started to decrease reimbursements for hospitals with excess readmissions after acute MI, heart failure, and pneumonia. Hip and knee replacements and chronic obstructive pulmonary disease will be added in 2014, and we anticipate that additional surgical procedures will be added thereafter," she said.
In an effort to determine the rate of 30-day unplanned readmission after carotid endarterectomy (CEA), Dr. Ho and her associates conducted a retrospective study of a prospectively collected vascular surgery database at Brigham and Women’s Hospital. The cohort included 896 consecutive CEAs performed between 2002 and 2011. Combined CABG/CEA procedures were excluded.
The primary endpoint was unplanned readmission within 30 days, defined as "any unanticipated, nonelective hospital readmission," she said. The secondary endpoint was 1-year survival.
The mean age of the patients was 70 years, 60% were male, and 95% were white. More than half (65%) had asymptomatic evidence of carotid artery disease.
Dr. Ho reported that the median postoperative length of stay was 1 day and that 9.9% of patients had at least one in-hospital complication. The most frequent in-hospital complication was bleeding/hematoma (4.1%), followed by arrhythmia (2.1%), dysphagia (1.7%), stroke (1.3%), and myocardial infarction (1.2%). Only 3% of patients required a reoperation, while most (94%) were discharged to home. The 30-day stroke rate was 1.7%, while the 30-day death rate was 0.6%.
The overall 30-day readmission rate was 8.6%, while the unplanned 30-day readmission rate was 6.5%. "Most of the overall readmissions (80%) occurred in the first 10 days, and the median time to unplanned readmission was 4 days," Dr. Ho said.
The most common reason for an unplanned readmission was a cardiac complication, followed by headache, bleeding/hematoma, stroke/transient ischemic attack/intracerebral hemorrhage, or other medical emergency. More than one-quarter of patients (27.5%) had more than one reason for an unplanned readmission, while 87.9% of patients had a CEA-related unplanned readmission.
When the researchers performed a univariate analysis followed by analysis with a multivariable Cox model for unplanned readmission, four variables were independently associated with unplanned readmission: in-hospital postoperative CHF exacerbation (hazard ratio, 15.1), in-hospital postoperative stroke (HR, 5.0), in-hospital postoperative hematoma (HR, 3.1), and prior CABG (HR, 2.0).
They also observed a significant difference in survival at 1 year between patients who had an unplanned readmission and those who did not (91% vs. 96%, respectively; P less than .01.) "It’s unclear whether these deaths in the unplanned readmission group were preventable or if they were related to carotid disease or to a procedure-related complication," Dr. Ho said. "Our guess is that the increased overall burden of comorbid disease in these patients, rather than the readmission itself, predicted decreased survival."
Limitations of the study included its retrospective design and the fact that it was conducted at a single center, she said, "but we do know that our unplanned readmission rate is comparable to estimates from recent Medicare data."
Dr. Ho said she had no relevant financial disclosures.
SAN FRANCISCO – The 30-day unplanned readmission rate following carotid endarterectomy was 6.5% in a single-center study.
In addition, four variables were significantly associated with unplanned readmission: in-hospital postoperative congestive heart failure (CHF) exacerbation; in-hospital postoperative stroke; in-hospital postoperative hematoma; and prior coronary artery bypass graft (CABG).
"Whether these complications are completely avoidable is unknown, but we do identify a group of patients who would probably benefit from more comprehensive discharge planning and careful postdischarge care," Dr. Karen J. Ho said at the Society for Vascular Surgery annual meeting.
According to a study of Medicare claims data from 2003 to 2004, 20% of Medicare beneficiaries discharged from a hospital were rehospitalized within 30 days (N. Eng. J. Med. 2009;360:1418-28). The 30-day rehospitalization rate after vascular surgery was 24%, "the highest of all surgical specialties examined in the study," said Dr. Ho of the surgery department at Brigham and Women’s Hospital, Boston, who was not involved with the published study. "Medicare has started to decrease reimbursements for hospitals with excess readmissions after acute MI, heart failure, and pneumonia. Hip and knee replacements and chronic obstructive pulmonary disease will be added in 2014, and we anticipate that additional surgical procedures will be added thereafter," she said.
In an effort to determine the rate of 30-day unplanned readmission after carotid endarterectomy (CEA), Dr. Ho and her associates conducted a retrospective study of a prospectively collected vascular surgery database at Brigham and Women’s Hospital. The cohort included 896 consecutive CEAs performed between 2002 and 2011. Combined CABG/CEA procedures were excluded.
The primary endpoint was unplanned readmission within 30 days, defined as "any unanticipated, nonelective hospital readmission," she said. The secondary endpoint was 1-year survival.
The mean age of the patients was 70 years, 60% were male, and 95% were white. More than half (65%) had asymptomatic evidence of carotid artery disease.
Dr. Ho reported that the median postoperative length of stay was 1 day and that 9.9% of patients had at least one in-hospital complication. The most frequent in-hospital complication was bleeding/hematoma (4.1%), followed by arrhythmia (2.1%), dysphagia (1.7%), stroke (1.3%), and myocardial infarction (1.2%). Only 3% of patients required a reoperation, while most (94%) were discharged to home. The 30-day stroke rate was 1.7%, while the 30-day death rate was 0.6%.
The overall 30-day readmission rate was 8.6%, while the unplanned 30-day readmission rate was 6.5%. "Most of the overall readmissions (80%) occurred in the first 10 days, and the median time to unplanned readmission was 4 days," Dr. Ho said.
The most common reason for an unplanned readmission was a cardiac complication, followed by headache, bleeding/hematoma, stroke/transient ischemic attack/intracerebral hemorrhage, or other medical emergency. More than one-quarter of patients (27.5%) had more than one reason for an unplanned readmission, while 87.9% of patients had a CEA-related unplanned readmission.
When the researchers performed a univariate analysis followed by analysis with a multivariable Cox model for unplanned readmission, four variables were independently associated with unplanned readmission: in-hospital postoperative CHF exacerbation (hazard ratio, 15.1), in-hospital postoperative stroke (HR, 5.0), in-hospital postoperative hematoma (HR, 3.1), and prior CABG (HR, 2.0).
They also observed a significant difference in survival at 1 year between patients who had an unplanned readmission and those who did not (91% vs. 96%, respectively; P less than .01.) "It’s unclear whether these deaths in the unplanned readmission group were preventable or if they were related to carotid disease or to a procedure-related complication," Dr. Ho said. "Our guess is that the increased overall burden of comorbid disease in these patients, rather than the readmission itself, predicted decreased survival."
Limitations of the study included its retrospective design and the fact that it was conducted at a single center, she said, "but we do know that our unplanned readmission rate is comparable to estimates from recent Medicare data."
Dr. Ho said she had no relevant financial disclosures.
SAN FRANCISCO – The 30-day unplanned readmission rate following carotid endarterectomy was 6.5% in a single-center study.
In addition, four variables were significantly associated with unplanned readmission: in-hospital postoperative congestive heart failure (CHF) exacerbation; in-hospital postoperative stroke; in-hospital postoperative hematoma; and prior coronary artery bypass graft (CABG).
"Whether these complications are completely avoidable is unknown, but we do identify a group of patients who would probably benefit from more comprehensive discharge planning and careful postdischarge care," Dr. Karen J. Ho said at the Society for Vascular Surgery annual meeting.
According to a study of Medicare claims data from 2003 to 2004, 20% of Medicare beneficiaries discharged from a hospital were rehospitalized within 30 days (N. Eng. J. Med. 2009;360:1418-28). The 30-day rehospitalization rate after vascular surgery was 24%, "the highest of all surgical specialties examined in the study," said Dr. Ho of the surgery department at Brigham and Women’s Hospital, Boston, who was not involved with the published study. "Medicare has started to decrease reimbursements for hospitals with excess readmissions after acute MI, heart failure, and pneumonia. Hip and knee replacements and chronic obstructive pulmonary disease will be added in 2014, and we anticipate that additional surgical procedures will be added thereafter," she said.
In an effort to determine the rate of 30-day unplanned readmission after carotid endarterectomy (CEA), Dr. Ho and her associates conducted a retrospective study of a prospectively collected vascular surgery database at Brigham and Women’s Hospital. The cohort included 896 consecutive CEAs performed between 2002 and 2011. Combined CABG/CEA procedures were excluded.
The primary endpoint was unplanned readmission within 30 days, defined as "any unanticipated, nonelective hospital readmission," she said. The secondary endpoint was 1-year survival.
The mean age of the patients was 70 years, 60% were male, and 95% were white. More than half (65%) had asymptomatic evidence of carotid artery disease.
Dr. Ho reported that the median postoperative length of stay was 1 day and that 9.9% of patients had at least one in-hospital complication. The most frequent in-hospital complication was bleeding/hematoma (4.1%), followed by arrhythmia (2.1%), dysphagia (1.7%), stroke (1.3%), and myocardial infarction (1.2%). Only 3% of patients required a reoperation, while most (94%) were discharged to home. The 30-day stroke rate was 1.7%, while the 30-day death rate was 0.6%.
The overall 30-day readmission rate was 8.6%, while the unplanned 30-day readmission rate was 6.5%. "Most of the overall readmissions (80%) occurred in the first 10 days, and the median time to unplanned readmission was 4 days," Dr. Ho said.
The most common reason for an unplanned readmission was a cardiac complication, followed by headache, bleeding/hematoma, stroke/transient ischemic attack/intracerebral hemorrhage, or other medical emergency. More than one-quarter of patients (27.5%) had more than one reason for an unplanned readmission, while 87.9% of patients had a CEA-related unplanned readmission.
When the researchers performed a univariate analysis followed by analysis with a multivariable Cox model for unplanned readmission, four variables were independently associated with unplanned readmission: in-hospital postoperative CHF exacerbation (hazard ratio, 15.1), in-hospital postoperative stroke (HR, 5.0), in-hospital postoperative hematoma (HR, 3.1), and prior CABG (HR, 2.0).
They also observed a significant difference in survival at 1 year between patients who had an unplanned readmission and those who did not (91% vs. 96%, respectively; P less than .01.) "It’s unclear whether these deaths in the unplanned readmission group were preventable or if they were related to carotid disease or to a procedure-related complication," Dr. Ho said. "Our guess is that the increased overall burden of comorbid disease in these patients, rather than the readmission itself, predicted decreased survival."
Limitations of the study included its retrospective design and the fact that it was conducted at a single center, she said, "but we do know that our unplanned readmission rate is comparable to estimates from recent Medicare data."
Dr. Ho said she had no relevant financial disclosures.
AT THE SVS ANNUAL MEETING
Major finding: Four variables were independently associated with unplanned readmission: in-hospital postoperative CHF exacerbation (hazard ratio, 15.1), in-hospital postoperative stroke (HR, 5.0), in-hospital postoperative hematoma (HR, 3.1), and prior CABG (HR, 2.0).
Data source: A study of 896 consecutive CEAs performed between 2002 and 2011 at Brigham and Women’s Hospital, Boston.
Disclosures: Dr. Ho said she had no relevant financial disclosures.
One-third of perioperative EVAR deaths occurred after discharge
SAN FRANCISCO – One-third of perioperative deaths and complications after elective endovascular repair of abdominal aortic aneurysms occur post discharge, results from a large analysis showed.
"Improved predischarge surveillance and close postdischarge follow-up of identified high-risk patients may further improve 30-day outcomes after EVAR," Dr. Prateek K. Gupta said at the Society for Vascular Surgery annual meeting.
Outcome improvement in the field of aortic surgery, specifically endovascular repair of abdominal aortic aneurysms, "has received much attention," said Dr. Gupta of the department of surgery at the University of Wisconsin Hospital and Clinics, Madison. "With EVAR, the index hospital stay after aortic surgery has decreased significantly, leaving a need for better understanding of postdischarge outcomes, which is necessary to improve quality and reduce readmission rates with implementation of targeted outpatient interventions."
In an effort to examine postdischarge 30-day outcomes after elective EVAR, Dr. Gupta and his associates identified 11,229 patients from the American College of Surgeons National Surgical Quality Improvement Program (NSQIP) database who underwent an elective EVAR for AAA between 2005 and 2010. The primary outcome of interest was postdischarge mortality, while the secondary outcome was postdischarge overall morbidity. The researchers performed univariate and multiple logistic regression analysis to assess factors associated with the primary and secondary study outcomes.
Of the 11,229 patient 83% were male and their mean age was 75 years. Dr. Gupta reported that 117 patients died within 30 days of EVAR, for a rate of 1%. Of these deaths, 31% occurred after hospital discharge, and the median time to death was 9 days. At the same time, 1,204 patients experienced complications within 30 days of EVAR, for a rate of 11%. Of these, 500 (40%) occurred post discharge, and the median time for a complication to occur was 3 days.
Only 20% of patients (7/36) who died post discharge experienced an in-hospital complication. Compared with patients who did not develop a postdischarge complication, those who had more than a sixfold likelihood of reoperation (20.4% vs. 3.1%, respectively; P less than .0001) and death (3.0% vs. 0.2%; P less than .0001) within 30 days of surgery.
Multivariable analysis revealed the following factors that were independently and significantly associated with postdischarge mortality: preoperative heart failure (adjusted odds ratio, 4.7), admission from a skilled nursing facility (AOR, 2.2), increase in age per year (AOR, 1.09), postdischarge renal failure requiring dialysis (AOR, 72.5), postdischarge cardiac arrest/MI (AOR, 46.6), and postdischarge pneumonia (AOR, 26.5).
Dr. Gupta reported that the 30-day postdischarge rate among patients admitted from a nursing facility or acute care was 2.5%. "In contrast to patients who survived after EVAR, patients who died post discharge were more likely to have been admitted from a nursing facility or acute care (13.9% vs. 1.8%; P less than .0001)," he said.
The 30-day post-discharge mortality was highest among patients who had postdischarge renal failure (27%),postdischarge MI (19%), and postdischarge pneumonia (15%).
The researchers also found that patients with a history of peripheral artery disease (PAD) had a significantly higher post-discharge complication rate after EVAR (7.1% vs. 4.3%; P = .001). This also correlated with a higher wound infection rate (3.2% vs. 1.7%; P = .01). A previous cardiac surgery also predisposed patients toward a higher overall postdischarge complication rate (5.3% vs. 4.2%; P = .007).
"Usually, patients undergoing EVAR are followed up at 2 weeks for wound evaluation, or at 1 month with a CT scan," Dr. Gupta said. "In the present study, the median occurrence for most of the postdischarge complications was within the first 10 days after surgery. The interquartile range was 11-22 days for the diagnosis of a wound infection after EVAR. These data suggest that earlier follow-up of high-risk patients may help identify and possibly prevent some of these complications and subsequently decrease readmissions. A standardized protocol for triage and surveillance of high-risk patients post EVAR is needed."
Limitations of the study include that fact that causality could not be determined because it was a retrospective analysis. "In addition, the timing of the operation is not specified in NSQIP," so it could either be a predischarge event or it could have occurred on readmission, Dr. Gupta said. "Data on readmission is not available from the 2005-2010 data sets."
Dr. Gupta said that he had no relevant financial disclosures to make.
Dr. Gupta and his colleagues have assessed postprocedure complications after elective EVAR based upon review of the NSQIP database, and concluded that earlier follow-up of high-risk patients might identify and prevent some of the complications. This study is limited by the database nature of the review. This study also does not provide us with data as to the size of the AAA in the high risk patients.
Most of the complications leading to increased risk of mortality were postoperative issues, such as renal failure or MI, which could not be identified at the time of procedure, or the time of discharge. The only identifiable preoperative risk factors for adverse outcomes were preoperative heart failure, admission from a skilled nursing facility and increasing age. While changing the timing of follow-up might be appropriate for the high-risk patients, other considerations would be changing to more percutaneous procedures , and use of other adjuncts, such as antibiotic irrigations or Prevena (negative pressure wound therapy for intact skin) to decrease the wound infection rates for those undergoing femoral exploration for EVAR. Further, any intervention on the elderly, especially nursing home patients, needs to be thoroughly considered, as EVAR is most often a preventive operation, assuming fitness and appropriate longevity remains for the patient.
The findings from this study are important, but mostly, should serve as a caution to properly assess patients to determine who will potentially benefit from EVAR, and which patients might be best managed by observation alone.
Dr. Linda Harris is the program director and division chief of vascular surgery at the State University of New York, Buffalo.
Dr. Gupta and his colleagues have assessed postprocedure complications after elective EVAR based upon review of the NSQIP database, and concluded that earlier follow-up of high-risk patients might identify and prevent some of the complications. This study is limited by the database nature of the review. This study also does not provide us with data as to the size of the AAA in the high risk patients.
Most of the complications leading to increased risk of mortality were postoperative issues, such as renal failure or MI, which could not be identified at the time of procedure, or the time of discharge. The only identifiable preoperative risk factors for adverse outcomes were preoperative heart failure, admission from a skilled nursing facility and increasing age. While changing the timing of follow-up might be appropriate for the high-risk patients, other considerations would be changing to more percutaneous procedures , and use of other adjuncts, such as antibiotic irrigations or Prevena (negative pressure wound therapy for intact skin) to decrease the wound infection rates for those undergoing femoral exploration for EVAR. Further, any intervention on the elderly, especially nursing home patients, needs to be thoroughly considered, as EVAR is most often a preventive operation, assuming fitness and appropriate longevity remains for the patient.
The findings from this study are important, but mostly, should serve as a caution to properly assess patients to determine who will potentially benefit from EVAR, and which patients might be best managed by observation alone.
Dr. Linda Harris is the program director and division chief of vascular surgery at the State University of New York, Buffalo.
Dr. Gupta and his colleagues have assessed postprocedure complications after elective EVAR based upon review of the NSQIP database, and concluded that earlier follow-up of high-risk patients might identify and prevent some of the complications. This study is limited by the database nature of the review. This study also does not provide us with data as to the size of the AAA in the high risk patients.
Most of the complications leading to increased risk of mortality were postoperative issues, such as renal failure or MI, which could not be identified at the time of procedure, or the time of discharge. The only identifiable preoperative risk factors for adverse outcomes were preoperative heart failure, admission from a skilled nursing facility and increasing age. While changing the timing of follow-up might be appropriate for the high-risk patients, other considerations would be changing to more percutaneous procedures , and use of other adjuncts, such as antibiotic irrigations or Prevena (negative pressure wound therapy for intact skin) to decrease the wound infection rates for those undergoing femoral exploration for EVAR. Further, any intervention on the elderly, especially nursing home patients, needs to be thoroughly considered, as EVAR is most often a preventive operation, assuming fitness and appropriate longevity remains for the patient.
The findings from this study are important, but mostly, should serve as a caution to properly assess patients to determine who will potentially benefit from EVAR, and which patients might be best managed by observation alone.
Dr. Linda Harris is the program director and division chief of vascular surgery at the State University of New York, Buffalo.
SAN FRANCISCO – One-third of perioperative deaths and complications after elective endovascular repair of abdominal aortic aneurysms occur post discharge, results from a large analysis showed.
"Improved predischarge surveillance and close postdischarge follow-up of identified high-risk patients may further improve 30-day outcomes after EVAR," Dr. Prateek K. Gupta said at the Society for Vascular Surgery annual meeting.
Outcome improvement in the field of aortic surgery, specifically endovascular repair of abdominal aortic aneurysms, "has received much attention," said Dr. Gupta of the department of surgery at the University of Wisconsin Hospital and Clinics, Madison. "With EVAR, the index hospital stay after aortic surgery has decreased significantly, leaving a need for better understanding of postdischarge outcomes, which is necessary to improve quality and reduce readmission rates with implementation of targeted outpatient interventions."
In an effort to examine postdischarge 30-day outcomes after elective EVAR, Dr. Gupta and his associates identified 11,229 patients from the American College of Surgeons National Surgical Quality Improvement Program (NSQIP) database who underwent an elective EVAR for AAA between 2005 and 2010. The primary outcome of interest was postdischarge mortality, while the secondary outcome was postdischarge overall morbidity. The researchers performed univariate and multiple logistic regression analysis to assess factors associated with the primary and secondary study outcomes.
Of the 11,229 patient 83% were male and their mean age was 75 years. Dr. Gupta reported that 117 patients died within 30 days of EVAR, for a rate of 1%. Of these deaths, 31% occurred after hospital discharge, and the median time to death was 9 days. At the same time, 1,204 patients experienced complications within 30 days of EVAR, for a rate of 11%. Of these, 500 (40%) occurred post discharge, and the median time for a complication to occur was 3 days.
Only 20% of patients (7/36) who died post discharge experienced an in-hospital complication. Compared with patients who did not develop a postdischarge complication, those who had more than a sixfold likelihood of reoperation (20.4% vs. 3.1%, respectively; P less than .0001) and death (3.0% vs. 0.2%; P less than .0001) within 30 days of surgery.
Multivariable analysis revealed the following factors that were independently and significantly associated with postdischarge mortality: preoperative heart failure (adjusted odds ratio, 4.7), admission from a skilled nursing facility (AOR, 2.2), increase in age per year (AOR, 1.09), postdischarge renal failure requiring dialysis (AOR, 72.5), postdischarge cardiac arrest/MI (AOR, 46.6), and postdischarge pneumonia (AOR, 26.5).
Dr. Gupta reported that the 30-day postdischarge rate among patients admitted from a nursing facility or acute care was 2.5%. "In contrast to patients who survived after EVAR, patients who died post discharge were more likely to have been admitted from a nursing facility or acute care (13.9% vs. 1.8%; P less than .0001)," he said.
The 30-day post-discharge mortality was highest among patients who had postdischarge renal failure (27%),postdischarge MI (19%), and postdischarge pneumonia (15%).
The researchers also found that patients with a history of peripheral artery disease (PAD) had a significantly higher post-discharge complication rate after EVAR (7.1% vs. 4.3%; P = .001). This also correlated with a higher wound infection rate (3.2% vs. 1.7%; P = .01). A previous cardiac surgery also predisposed patients toward a higher overall postdischarge complication rate (5.3% vs. 4.2%; P = .007).
"Usually, patients undergoing EVAR are followed up at 2 weeks for wound evaluation, or at 1 month with a CT scan," Dr. Gupta said. "In the present study, the median occurrence for most of the postdischarge complications was within the first 10 days after surgery. The interquartile range was 11-22 days for the diagnosis of a wound infection after EVAR. These data suggest that earlier follow-up of high-risk patients may help identify and possibly prevent some of these complications and subsequently decrease readmissions. A standardized protocol for triage and surveillance of high-risk patients post EVAR is needed."
Limitations of the study include that fact that causality could not be determined because it was a retrospective analysis. "In addition, the timing of the operation is not specified in NSQIP," so it could either be a predischarge event or it could have occurred on readmission, Dr. Gupta said. "Data on readmission is not available from the 2005-2010 data sets."
Dr. Gupta said that he had no relevant financial disclosures to make.
SAN FRANCISCO – One-third of perioperative deaths and complications after elective endovascular repair of abdominal aortic aneurysms occur post discharge, results from a large analysis showed.
"Improved predischarge surveillance and close postdischarge follow-up of identified high-risk patients may further improve 30-day outcomes after EVAR," Dr. Prateek K. Gupta said at the Society for Vascular Surgery annual meeting.
Outcome improvement in the field of aortic surgery, specifically endovascular repair of abdominal aortic aneurysms, "has received much attention," said Dr. Gupta of the department of surgery at the University of Wisconsin Hospital and Clinics, Madison. "With EVAR, the index hospital stay after aortic surgery has decreased significantly, leaving a need for better understanding of postdischarge outcomes, which is necessary to improve quality and reduce readmission rates with implementation of targeted outpatient interventions."
In an effort to examine postdischarge 30-day outcomes after elective EVAR, Dr. Gupta and his associates identified 11,229 patients from the American College of Surgeons National Surgical Quality Improvement Program (NSQIP) database who underwent an elective EVAR for AAA between 2005 and 2010. The primary outcome of interest was postdischarge mortality, while the secondary outcome was postdischarge overall morbidity. The researchers performed univariate and multiple logistic regression analysis to assess factors associated with the primary and secondary study outcomes.
Of the 11,229 patient 83% were male and their mean age was 75 years. Dr. Gupta reported that 117 patients died within 30 days of EVAR, for a rate of 1%. Of these deaths, 31% occurred after hospital discharge, and the median time to death was 9 days. At the same time, 1,204 patients experienced complications within 30 days of EVAR, for a rate of 11%. Of these, 500 (40%) occurred post discharge, and the median time for a complication to occur was 3 days.
Only 20% of patients (7/36) who died post discharge experienced an in-hospital complication. Compared with patients who did not develop a postdischarge complication, those who had more than a sixfold likelihood of reoperation (20.4% vs. 3.1%, respectively; P less than .0001) and death (3.0% vs. 0.2%; P less than .0001) within 30 days of surgery.
Multivariable analysis revealed the following factors that were independently and significantly associated with postdischarge mortality: preoperative heart failure (adjusted odds ratio, 4.7), admission from a skilled nursing facility (AOR, 2.2), increase in age per year (AOR, 1.09), postdischarge renal failure requiring dialysis (AOR, 72.5), postdischarge cardiac arrest/MI (AOR, 46.6), and postdischarge pneumonia (AOR, 26.5).
Dr. Gupta reported that the 30-day postdischarge rate among patients admitted from a nursing facility or acute care was 2.5%. "In contrast to patients who survived after EVAR, patients who died post discharge were more likely to have been admitted from a nursing facility or acute care (13.9% vs. 1.8%; P less than .0001)," he said.
The 30-day post-discharge mortality was highest among patients who had postdischarge renal failure (27%),postdischarge MI (19%), and postdischarge pneumonia (15%).
The researchers also found that patients with a history of peripheral artery disease (PAD) had a significantly higher post-discharge complication rate after EVAR (7.1% vs. 4.3%; P = .001). This also correlated with a higher wound infection rate (3.2% vs. 1.7%; P = .01). A previous cardiac surgery also predisposed patients toward a higher overall postdischarge complication rate (5.3% vs. 4.2%; P = .007).
"Usually, patients undergoing EVAR are followed up at 2 weeks for wound evaluation, or at 1 month with a CT scan," Dr. Gupta said. "In the present study, the median occurrence for most of the postdischarge complications was within the first 10 days after surgery. The interquartile range was 11-22 days for the diagnosis of a wound infection after EVAR. These data suggest that earlier follow-up of high-risk patients may help identify and possibly prevent some of these complications and subsequently decrease readmissions. A standardized protocol for triage and surveillance of high-risk patients post EVAR is needed."
Limitations of the study include that fact that causality could not be determined because it was a retrospective analysis. "In addition, the timing of the operation is not specified in NSQIP," so it could either be a predischarge event or it could have occurred on readmission, Dr. Gupta said. "Data on readmission is not available from the 2005-2010 data sets."
Dr. Gupta said that he had no relevant financial disclosures to make.
AT THE SVS ANNUAL MEETING
Major finding: Following endovascular repair of abdominal aortic aneurysms, 31% of deaths and 40% of complications occurred after hospital discharge.
Data source: An analysis of 11,229 patients from the American College of Surgeons National Surgical Quality Improvement Program (NSQIP) database who underwent an elective EVAR for AAA between 2005 and 2010.
Disclosures: Dr. Gupta said that he had no relevant financial conflicts to disclose.
Sunshine Act: Tracking of gifts, payments starts in August
CHICAGO – Worried that erroneous data about your relationships with industry will be publicly reported under the federal Sunshine Act? Officials from the Centers for Medicare and Medicaid Services spoke at the annual meeting of the American Medical Association House of Delegates June 17 in an effort to allay those fears.
The Sunshine Act, which requires manufacturers to report to the CMS almost all payments and gifts made to physicians and teaching hospitals, became law as part of the Affordable Care Act. Final rules for the ACA’s Sunshine Act provisions, which the government is now calling the Open Payments Program, were issued in February.
Once data have been collected and processed, physicians will have 45 days to dispute and correct manufacturers’ reports, the CMS officials said.
After that, the data will be made public. The 5 months of data that will be collected for this year – starting Aug. 1 – will be released publicly in September 2014. Going forward, the previous year’s data will be reported each June.
Although physicians will not report their data directly to the CMS, they will be expected to take an active role in ensuring the program is successful, according to Anita Griner, deputy director of the data sharing and partnership group at the agency’s Center for Program Integrity.
Manufacturers will start compiling data on Aug. 1 on payments to physicians for consulting, honoraria, grants, gifts, meals, travel, royalties, and speaking at events, including continuing education programs. In January, the CMS will encourage physicians and teaching hospitals to register with the agency, most likely through a web portal. Once registered, doctors will be notified when data about them has been submitted, Ms. Griner explained.
Providers will have 45 days to dispute the data. If they see something false, misleading, or wrong, they must indicate their dispute to the agency and then work out the dispute with the manufacturer. The data will be flagged as "disputed" on the public website.
If physicians don’t bring a dispute to the agency’s attention within the 45-day window, it could take months for the dispute to be noted on the website, Ms. Griner said, and if the dispute is not settled within a year, the manufacturer’s data will be the de facto report to the public.
"We encourage you to come in, get involved, and dispute things that are inaccurate," she said.
She and her colleague, Dr. Shantanu Agrawal, medical director of the agency’s Center for Program Integrity, urged physicians to track their own data.
In July, the CMS plans to release a free app for Android and Apple smartphones that will include all the categories that manufacturers must report. The agency also plans to release one for industry so that sales reps and others can upload data directly to physician contacts.
One physician questioned whether the public would "stampede" to get a look at the payment data, noting that he had never been asked by any patients whether a manufacturer had taken him to dinner. "I think it will be interesting to see if that occurs," said Dr. Agrawal.
On Twitter @aliciaault
CHICAGO – Worried that erroneous data about your relationships with industry will be publicly reported under the federal Sunshine Act? Officials from the Centers for Medicare and Medicaid Services spoke at the annual meeting of the American Medical Association House of Delegates June 17 in an effort to allay those fears.
The Sunshine Act, which requires manufacturers to report to the CMS almost all payments and gifts made to physicians and teaching hospitals, became law as part of the Affordable Care Act. Final rules for the ACA’s Sunshine Act provisions, which the government is now calling the Open Payments Program, were issued in February.
Once data have been collected and processed, physicians will have 45 days to dispute and correct manufacturers’ reports, the CMS officials said.
After that, the data will be made public. The 5 months of data that will be collected for this year – starting Aug. 1 – will be released publicly in September 2014. Going forward, the previous year’s data will be reported each June.
Although physicians will not report their data directly to the CMS, they will be expected to take an active role in ensuring the program is successful, according to Anita Griner, deputy director of the data sharing and partnership group at the agency’s Center for Program Integrity.
Manufacturers will start compiling data on Aug. 1 on payments to physicians for consulting, honoraria, grants, gifts, meals, travel, royalties, and speaking at events, including continuing education programs. In January, the CMS will encourage physicians and teaching hospitals to register with the agency, most likely through a web portal. Once registered, doctors will be notified when data about them has been submitted, Ms. Griner explained.
Providers will have 45 days to dispute the data. If they see something false, misleading, or wrong, they must indicate their dispute to the agency and then work out the dispute with the manufacturer. The data will be flagged as "disputed" on the public website.
If physicians don’t bring a dispute to the agency’s attention within the 45-day window, it could take months for the dispute to be noted on the website, Ms. Griner said, and if the dispute is not settled within a year, the manufacturer’s data will be the de facto report to the public.
"We encourage you to come in, get involved, and dispute things that are inaccurate," she said.
She and her colleague, Dr. Shantanu Agrawal, medical director of the agency’s Center for Program Integrity, urged physicians to track their own data.
In July, the CMS plans to release a free app for Android and Apple smartphones that will include all the categories that manufacturers must report. The agency also plans to release one for industry so that sales reps and others can upload data directly to physician contacts.
One physician questioned whether the public would "stampede" to get a look at the payment data, noting that he had never been asked by any patients whether a manufacturer had taken him to dinner. "I think it will be interesting to see if that occurs," said Dr. Agrawal.
On Twitter @aliciaault
CHICAGO – Worried that erroneous data about your relationships with industry will be publicly reported under the federal Sunshine Act? Officials from the Centers for Medicare and Medicaid Services spoke at the annual meeting of the American Medical Association House of Delegates June 17 in an effort to allay those fears.
The Sunshine Act, which requires manufacturers to report to the CMS almost all payments and gifts made to physicians and teaching hospitals, became law as part of the Affordable Care Act. Final rules for the ACA’s Sunshine Act provisions, which the government is now calling the Open Payments Program, were issued in February.
Once data have been collected and processed, physicians will have 45 days to dispute and correct manufacturers’ reports, the CMS officials said.
After that, the data will be made public. The 5 months of data that will be collected for this year – starting Aug. 1 – will be released publicly in September 2014. Going forward, the previous year’s data will be reported each June.
Although physicians will not report their data directly to the CMS, they will be expected to take an active role in ensuring the program is successful, according to Anita Griner, deputy director of the data sharing and partnership group at the agency’s Center for Program Integrity.
Manufacturers will start compiling data on Aug. 1 on payments to physicians for consulting, honoraria, grants, gifts, meals, travel, royalties, and speaking at events, including continuing education programs. In January, the CMS will encourage physicians and teaching hospitals to register with the agency, most likely through a web portal. Once registered, doctors will be notified when data about them has been submitted, Ms. Griner explained.
Providers will have 45 days to dispute the data. If they see something false, misleading, or wrong, they must indicate their dispute to the agency and then work out the dispute with the manufacturer. The data will be flagged as "disputed" on the public website.
If physicians don’t bring a dispute to the agency’s attention within the 45-day window, it could take months for the dispute to be noted on the website, Ms. Griner said, and if the dispute is not settled within a year, the manufacturer’s data will be the de facto report to the public.
"We encourage you to come in, get involved, and dispute things that are inaccurate," she said.
She and her colleague, Dr. Shantanu Agrawal, medical director of the agency’s Center for Program Integrity, urged physicians to track their own data.
In July, the CMS plans to release a free app for Android and Apple smartphones that will include all the categories that manufacturers must report. The agency also plans to release one for industry so that sales reps and others can upload data directly to physician contacts.
One physician questioned whether the public would "stampede" to get a look at the payment data, noting that he had never been asked by any patients whether a manufacturer had taken him to dinner. "I think it will be interesting to see if that occurs," said Dr. Agrawal.
On Twitter @aliciaault
AT THE AMA HOUSE OF DELEGATES