User login
Official Newspaper of the American College of Surgeons
Operating with pain: Reader response
The feature article “Operating with pain: Surgeon workplace injury underrecognized" touched a nerve with our readers! The following comments appeared in the ACS Communities on the topic of pain and occupational injury among surgeons:
I recently was forced to undergo an anterior cervical decompression and fusion (ACDF) involving C4,5,6 and 7 due to worsening radicular pain and weakness due to severe cervical spinal stenosis. This problem was likely initiated by multiple injuries that I sustained as a wrestler in my younger days, but was no doubt exacerbated by 23 years bent over the OR table in extended periods of flexion, ignoring pain, and working every day no matter how I felt with no time for nuisances like physical therapy. Such is the mentality of the surgeon. In any case, my experience demonstrates that surgery, unlike many other medical specialties, takes a physical toll on its practitioners and also requires a certain level of fitness for surgeons to practice well.
The concept of wellness among surgeons is relatively new, in my opinion. Historically, surgical training was notorious for long hours, extended periods of sleep deprivation, irregular eating habits, strained interpersonal relationships, and frankly, sometimes an emotionally abusive environment. Many changes have been made to adapt training to be more livable, but these changes have been predominantly in the areas of work hours, sleep, and time off. Little has been done to teach adaptive strategies for the physical demands of performing surgery day after day.
Do we need a formal “plan” to educate surgeons how to save their backs and necks? Perhaps not, but surgeons do need to be aware that the cumulative “wear and tear” on our bodies can definitely affect how well we do our jobs, the number of years we are able to do our jobs, and the enjoyment with which we do our jobs. So tell the resident to stand up straight, teach them to operate with the table at the correct height, to hold the instruments in an ergonomic fashion, etc. Let’s begin to make proper ergonomics a part of our surgical culture so we may serve our patients for many years to come.
Bryan K. Richmond, MD, MBA, FACS
Charleston, W.Va.
I remember attending the ACS meeting as a chief resident. There was a laparoscopic instrument rep taking a survey about ergonomics, especially arm, shoulder, neck, and back pain during surgery. I just remember laughing and saying, “Heck no, no issues for me.” Well, now as a 50-year-old, I’ve got issues! I find that I have to be very aware of my posture during procedures and not spend too much time in one position. Hand cramps are not an infrequent problem during longer surgeries as well. Getting used to wearing slightly looser gloves has helped some.
Peter Krone, MD, FACS
Granbury, Tex.
As much as we all love surgery, it seems operating is definitely taking its toll on most every surgeon I know. Short of changing how we operate (i.e., lap vs. robotic, etc.), it seems there is little we can do to protect ourselves. I learned of gel mats a few years ago. They are awesome for longer cases. For me personally, it has been bilateral carpal tunnel releases and a C5,6/6,7 ACDF for degenerative changes causing radiculopathy. Fortunately, both operations were 100% successful.
I applaud you for looking at the (virtual lack of) ergonomics in surgery.
Brent C. Jackson, MD, FACS
Sacramento, Calif.
I finished my vascular surgical fellowship in 1991. Being old school, I continue to do some general surgery along with my comprehensive vascular surgery. In 2008, I had an urgent ACDF. Shortly thereafter, I attended the Southern Association of Vascular Surgery meeting and took an informal survey. I found that at least 60% of vascular surgeons in practice for 10 years had required an ACDF obviously secondary to loupes. Now in the endovascular cases wearing lead, lumbar back issues are also becoming very common in our field. This is a hugely important topic, and ergonomic study and training should become an integral part of training and retraining.
Thomas Appleby, MD, FACS
Charleston, S.C.
I am naturally right handed and have had the opposite experience of having left-handed surgeons teach me how to operate with my left hand throughout training. It is something I continue to do today. It comes in quite handy when helping one of my colleagues since they feel I am standing on the ‘wrong’ side of the table for most open procedures – but that was best for the left-handed approaches I learned.
Laparoscopic instruments are not friendly to anyone’s hands. Also, how fun is it to stand on your left foot for an entire case while operating a right foot control for your instruments?
Colette Whitby, MD, FACS
Southbridge, Mass.
Ergonomics in the OR is one of the subjects being considered at the Governor’s Competency and Wellness work group. More information is available, but certainly more has to be done.
David Welsh, MD, FACS
Batesville, IN
The feature article “Operating with pain: Surgeon workplace injury underrecognized" touched a nerve with our readers! The following comments appeared in the ACS Communities on the topic of pain and occupational injury among surgeons:
I recently was forced to undergo an anterior cervical decompression and fusion (ACDF) involving C4,5,6 and 7 due to worsening radicular pain and weakness due to severe cervical spinal stenosis. This problem was likely initiated by multiple injuries that I sustained as a wrestler in my younger days, but was no doubt exacerbated by 23 years bent over the OR table in extended periods of flexion, ignoring pain, and working every day no matter how I felt with no time for nuisances like physical therapy. Such is the mentality of the surgeon. In any case, my experience demonstrates that surgery, unlike many other medical specialties, takes a physical toll on its practitioners and also requires a certain level of fitness for surgeons to practice well.
The concept of wellness among surgeons is relatively new, in my opinion. Historically, surgical training was notorious for long hours, extended periods of sleep deprivation, irregular eating habits, strained interpersonal relationships, and frankly, sometimes an emotionally abusive environment. Many changes have been made to adapt training to be more livable, but these changes have been predominantly in the areas of work hours, sleep, and time off. Little has been done to teach adaptive strategies for the physical demands of performing surgery day after day.
Do we need a formal “plan” to educate surgeons how to save their backs and necks? Perhaps not, but surgeons do need to be aware that the cumulative “wear and tear” on our bodies can definitely affect how well we do our jobs, the number of years we are able to do our jobs, and the enjoyment with which we do our jobs. So tell the resident to stand up straight, teach them to operate with the table at the correct height, to hold the instruments in an ergonomic fashion, etc. Let’s begin to make proper ergonomics a part of our surgical culture so we may serve our patients for many years to come.
Bryan K. Richmond, MD, MBA, FACS
Charleston, W.Va.
I remember attending the ACS meeting as a chief resident. There was a laparoscopic instrument rep taking a survey about ergonomics, especially arm, shoulder, neck, and back pain during surgery. I just remember laughing and saying, “Heck no, no issues for me.” Well, now as a 50-year-old, I’ve got issues! I find that I have to be very aware of my posture during procedures and not spend too much time in one position. Hand cramps are not an infrequent problem during longer surgeries as well. Getting used to wearing slightly looser gloves has helped some.
Peter Krone, MD, FACS
Granbury, Tex.
As much as we all love surgery, it seems operating is definitely taking its toll on most every surgeon I know. Short of changing how we operate (i.e., lap vs. robotic, etc.), it seems there is little we can do to protect ourselves. I learned of gel mats a few years ago. They are awesome for longer cases. For me personally, it has been bilateral carpal tunnel releases and a C5,6/6,7 ACDF for degenerative changes causing radiculopathy. Fortunately, both operations were 100% successful.
I applaud you for looking at the (virtual lack of) ergonomics in surgery.
Brent C. Jackson, MD, FACS
Sacramento, Calif.
I finished my vascular surgical fellowship in 1991. Being old school, I continue to do some general surgery along with my comprehensive vascular surgery. In 2008, I had an urgent ACDF. Shortly thereafter, I attended the Southern Association of Vascular Surgery meeting and took an informal survey. I found that at least 60% of vascular surgeons in practice for 10 years had required an ACDF obviously secondary to loupes. Now in the endovascular cases wearing lead, lumbar back issues are also becoming very common in our field. This is a hugely important topic, and ergonomic study and training should become an integral part of training and retraining.
Thomas Appleby, MD, FACS
Charleston, S.C.
I am naturally right handed and have had the opposite experience of having left-handed surgeons teach me how to operate with my left hand throughout training. It is something I continue to do today. It comes in quite handy when helping one of my colleagues since they feel I am standing on the ‘wrong’ side of the table for most open procedures – but that was best for the left-handed approaches I learned.
Laparoscopic instruments are not friendly to anyone’s hands. Also, how fun is it to stand on your left foot for an entire case while operating a right foot control for your instruments?
Colette Whitby, MD, FACS
Southbridge, Mass.
Ergonomics in the OR is one of the subjects being considered at the Governor’s Competency and Wellness work group. More information is available, but certainly more has to be done.
David Welsh, MD, FACS
Batesville, IN
The feature article “Operating with pain: Surgeon workplace injury underrecognized" touched a nerve with our readers! The following comments appeared in the ACS Communities on the topic of pain and occupational injury among surgeons:
I recently was forced to undergo an anterior cervical decompression and fusion (ACDF) involving C4,5,6 and 7 due to worsening radicular pain and weakness due to severe cervical spinal stenosis. This problem was likely initiated by multiple injuries that I sustained as a wrestler in my younger days, but was no doubt exacerbated by 23 years bent over the OR table in extended periods of flexion, ignoring pain, and working every day no matter how I felt with no time for nuisances like physical therapy. Such is the mentality of the surgeon. In any case, my experience demonstrates that surgery, unlike many other medical specialties, takes a physical toll on its practitioners and also requires a certain level of fitness for surgeons to practice well.
The concept of wellness among surgeons is relatively new, in my opinion. Historically, surgical training was notorious for long hours, extended periods of sleep deprivation, irregular eating habits, strained interpersonal relationships, and frankly, sometimes an emotionally abusive environment. Many changes have been made to adapt training to be more livable, but these changes have been predominantly in the areas of work hours, sleep, and time off. Little has been done to teach adaptive strategies for the physical demands of performing surgery day after day.
Do we need a formal “plan” to educate surgeons how to save their backs and necks? Perhaps not, but surgeons do need to be aware that the cumulative “wear and tear” on our bodies can definitely affect how well we do our jobs, the number of years we are able to do our jobs, and the enjoyment with which we do our jobs. So tell the resident to stand up straight, teach them to operate with the table at the correct height, to hold the instruments in an ergonomic fashion, etc. Let’s begin to make proper ergonomics a part of our surgical culture so we may serve our patients for many years to come.
Bryan K. Richmond, MD, MBA, FACS
Charleston, W.Va.
I remember attending the ACS meeting as a chief resident. There was a laparoscopic instrument rep taking a survey about ergonomics, especially arm, shoulder, neck, and back pain during surgery. I just remember laughing and saying, “Heck no, no issues for me.” Well, now as a 50-year-old, I’ve got issues! I find that I have to be very aware of my posture during procedures and not spend too much time in one position. Hand cramps are not an infrequent problem during longer surgeries as well. Getting used to wearing slightly looser gloves has helped some.
Peter Krone, MD, FACS
Granbury, Tex.
As much as we all love surgery, it seems operating is definitely taking its toll on most every surgeon I know. Short of changing how we operate (i.e., lap vs. robotic, etc.), it seems there is little we can do to protect ourselves. I learned of gel mats a few years ago. They are awesome for longer cases. For me personally, it has been bilateral carpal tunnel releases and a C5,6/6,7 ACDF for degenerative changes causing radiculopathy. Fortunately, both operations were 100% successful.
I applaud you for looking at the (virtual lack of) ergonomics in surgery.
Brent C. Jackson, MD, FACS
Sacramento, Calif.
I finished my vascular surgical fellowship in 1991. Being old school, I continue to do some general surgery along with my comprehensive vascular surgery. In 2008, I had an urgent ACDF. Shortly thereafter, I attended the Southern Association of Vascular Surgery meeting and took an informal survey. I found that at least 60% of vascular surgeons in practice for 10 years had required an ACDF obviously secondary to loupes. Now in the endovascular cases wearing lead, lumbar back issues are also becoming very common in our field. This is a hugely important topic, and ergonomic study and training should become an integral part of training and retraining.
Thomas Appleby, MD, FACS
Charleston, S.C.
I am naturally right handed and have had the opposite experience of having left-handed surgeons teach me how to operate with my left hand throughout training. It is something I continue to do today. It comes in quite handy when helping one of my colleagues since they feel I am standing on the ‘wrong’ side of the table for most open procedures – but that was best for the left-handed approaches I learned.
Laparoscopic instruments are not friendly to anyone’s hands. Also, how fun is it to stand on your left foot for an entire case while operating a right foot control for your instruments?
Colette Whitby, MD, FACS
Southbridge, Mass.
Ergonomics in the OR is one of the subjects being considered at the Governor’s Competency and Wellness work group. More information is available, but certainly more has to be done.
David Welsh, MD, FACS
Batesville, IN
Delaying surgery for 11 weeks after RCT does not increase pCR rates for rectal cancer
Among patients with rectal cancer, delaying surgery for 11 weeks after the end of radiochemotherapy does not improve pathologic complete response rates, investigators reported.
Previously, the Lyon trial, the only randomized controlled study to investigate the effects of delaying surgery following the end of radiochemotherapy (RCT), found that compared with a 2-week delay, a 6-week delay significantly increased the number of patients who experienced complete response (53.1% vs. 71.7%, P = .007). The purpose of the current study was to evaluate the effect of a longer interval between RCT and surgery on pathologic complete response (pCR) rates.
For the phase III, multicenter, randomized trial, 265 patients with mid or lower rectal cancer were randomized to receive surgery at 7 weeks (n = 133) or 11 weeks (n = 132) following the end of RCT.
Baseline tumor characteristics and patient demographics were similar between the two study arms; the majority of patients had stage cT3 rectal cancer (82%).
There was no significant difference in pathologic complete response rate between the study arms (15% for 7-week group vs. 17.4% for 11-week group, P = .5983), reported Jeremie Lefevre, MD, of Hopital Saint-Antoine, Paris, and his associates (J Clin Oncol. 2016 July. doi: 10.1200/JCO.2016.67.6049).
Overall morbidity was significantly increased in the 11-week group (44.5% v 32%; P = .04), primarily explained by an increase in medical complications (32.8% vs. 19.2%; P = .01), the investigators wrote.
The French Ministry of Health funded the study. Dr. Lefevre and seven of his associates reported serving in advisory roles, receiving financial compensation, or participating in the speakers bureau for multiple companies.
On Twitter @jessnicolecraig
Among patients with rectal cancer, delaying surgery for 11 weeks after the end of radiochemotherapy does not improve pathologic complete response rates, investigators reported.
Previously, the Lyon trial, the only randomized controlled study to investigate the effects of delaying surgery following the end of radiochemotherapy (RCT), found that compared with a 2-week delay, a 6-week delay significantly increased the number of patients who experienced complete response (53.1% vs. 71.7%, P = .007). The purpose of the current study was to evaluate the effect of a longer interval between RCT and surgery on pathologic complete response (pCR) rates.
For the phase III, multicenter, randomized trial, 265 patients with mid or lower rectal cancer were randomized to receive surgery at 7 weeks (n = 133) or 11 weeks (n = 132) following the end of RCT.
Baseline tumor characteristics and patient demographics were similar between the two study arms; the majority of patients had stage cT3 rectal cancer (82%).
There was no significant difference in pathologic complete response rate between the study arms (15% for 7-week group vs. 17.4% for 11-week group, P = .5983), reported Jeremie Lefevre, MD, of Hopital Saint-Antoine, Paris, and his associates (J Clin Oncol. 2016 July. doi: 10.1200/JCO.2016.67.6049).
Overall morbidity was significantly increased in the 11-week group (44.5% v 32%; P = .04), primarily explained by an increase in medical complications (32.8% vs. 19.2%; P = .01), the investigators wrote.
The French Ministry of Health funded the study. Dr. Lefevre and seven of his associates reported serving in advisory roles, receiving financial compensation, or participating in the speakers bureau for multiple companies.
On Twitter @jessnicolecraig
Among patients with rectal cancer, delaying surgery for 11 weeks after the end of radiochemotherapy does not improve pathologic complete response rates, investigators reported.
Previously, the Lyon trial, the only randomized controlled study to investigate the effects of delaying surgery following the end of radiochemotherapy (RCT), found that compared with a 2-week delay, a 6-week delay significantly increased the number of patients who experienced complete response (53.1% vs. 71.7%, P = .007). The purpose of the current study was to evaluate the effect of a longer interval between RCT and surgery on pathologic complete response (pCR) rates.
For the phase III, multicenter, randomized trial, 265 patients with mid or lower rectal cancer were randomized to receive surgery at 7 weeks (n = 133) or 11 weeks (n = 132) following the end of RCT.
Baseline tumor characteristics and patient demographics were similar between the two study arms; the majority of patients had stage cT3 rectal cancer (82%).
There was no significant difference in pathologic complete response rate between the study arms (15% for 7-week group vs. 17.4% for 11-week group, P = .5983), reported Jeremie Lefevre, MD, of Hopital Saint-Antoine, Paris, and his associates (J Clin Oncol. 2016 July. doi: 10.1200/JCO.2016.67.6049).
Overall morbidity was significantly increased in the 11-week group (44.5% v 32%; P = .04), primarily explained by an increase in medical complications (32.8% vs. 19.2%; P = .01), the investigators wrote.
The French Ministry of Health funded the study. Dr. Lefevre and seven of his associates reported serving in advisory roles, receiving financial compensation, or participating in the speakers bureau for multiple companies.
On Twitter @jessnicolecraig
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Delaying surgery for 11 weeks after the end of radiochemotherapy does not improve pathologic complete response rates in patients with rectal cancer, compared with a delay of 7 weeks.
Major finding: There was no significant difference in pathologic complete response rate between the study arms (15% for 7-week group vs. 17.4% for 11-week group, P = .5983).
Data source: A phase III, multicenter, randomized trial involving 265 patients with varying stages of rectal cancer.
Disclosures: The French Ministry of Health funded the study. Dr. Lefevre and seven of his associates reported serving in advisory roles, receiving financial compensation, or participating in the speakers bureau for multiple companies.
Common surgeries linked to chronic opioid use among opioid-naive patients
Common surgeries increase the risk of chronic opioid use in opioid-naive adults, especially those using antidepressants or benzodiazepines before their operations, and those with substance abuse histories, according to an insurance claims analysis from Stanford (Calif.) University.
The researchers reviewed opioid prescribing in the first postop year – excluding the first 90 days – for 641,941 patients and compared that information with opioid prescribing for 18,011,137 adult patients who did not have surgery. None of the subjects had filled an opioid prescription in the previous year (JAMA Intern Med. 2016 Jul 11. doi: 10.1001/jamainternmed.2016.3298).
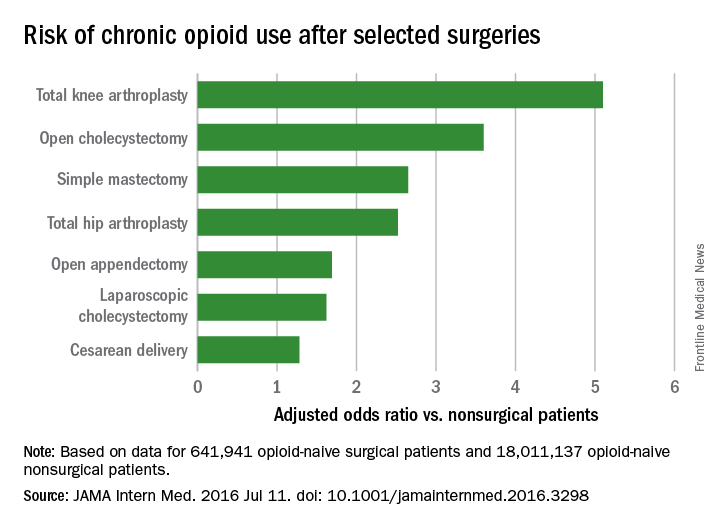
Chronic opioid use, defined as filling at least 120 days of opioid prescriptions within the first year of surgery, ranged up to 1.41% for total knee replacement, versus 0.136% in the nonsurgical controls. After adjustment for potential confounders, knee replacement increased the risk fivefold; open cholecystectomy almost fourfold; total hip replacement and simple mastectomy almost threefold; and laparoscopic cholecystectomy and open appendectomy almost twofold. Cesarean delivery increased the risk of chronic use by 28%.
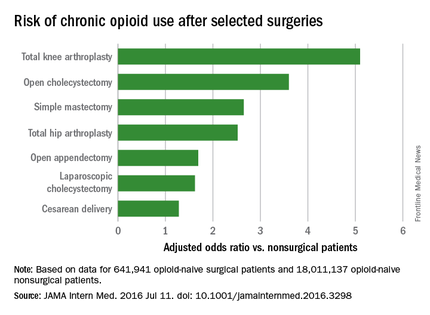
With the exception of knee and hip replacements, “these procedures are not indicated to relieve pain and are not thought to place patients at risk for long-term pain ... Our results suggest that primary care clinicians and surgeons should monitor opioid use closely in the postsurgical period,” wrote Eric C. Sun, MD, PhD, of the department of anesthesiology, perioperative and pain medicine at Stanford (Calif.) University, and his colleagues.
Preoperative antidepressants and benzodiazepines carried about the same risk of chronic use as alcohol abuse (odds ratio 1.83; P less than .001), while drug abuse history increased the risk even more (OR 3.15; P less than .001). Male sex, age over 50 years, and history of depression were also associated with chronic use on multivariate analysis. Meanwhile, transurethral prostatectomy, laparoscopic appendectomy, functional endoscopic sinus surgery, and cataract surgery did not increase chronic use risk.
“Surgical patients, particularly those at higher risk for chronic opioid use, may benefit from techniques to reduce the risk such as multimodal analgesia and regional anesthesia, particularly in light of literature suggesting that these interventions may improve other perioperative outcomes ... Patients may also benefit from other preoperative and postoperative interventions, such as evidence-based psychobehavioral pain management skills,” the investigators said.
It wasn’t clear until now that even opioid-naive patients are at risk for opioid problems after surgery. Stanford’s investigation is not the first to link surgery and opioid abuse, but previous studies tended to focus on patients with preexisting use and more painful operations.
The study included prescriptions for oral and patch fentanyl, hydrocodone, oral hydromorphone, methadone, morphine, oxymorphone, and oxycodone. Hydrocodone cough remedies and acetaminophen/codeine analgesics were excluded.
Nonsurgical patients tended to be younger than their surgical peers (mean 42 vs. 44 years) and more likely to be male (49% vs. 26%).
The authors had no disclosures. The work was funded in part by the Foundation for Anesthesia Education and Research and the Anesthesia Quality Institute. Claims data came from MarketScan (Truven Health Analytics).
Common surgeries increase the risk of chronic opioid use in opioid-naive adults, especially those using antidepressants or benzodiazepines before their operations, and those with substance abuse histories, according to an insurance claims analysis from Stanford (Calif.) University.
The researchers reviewed opioid prescribing in the first postop year – excluding the first 90 days – for 641,941 patients and compared that information with opioid prescribing for 18,011,137 adult patients who did not have surgery. None of the subjects had filled an opioid prescription in the previous year (JAMA Intern Med. 2016 Jul 11. doi: 10.1001/jamainternmed.2016.3298).
Chronic opioid use, defined as filling at least 120 days of opioid prescriptions within the first year of surgery, ranged up to 1.41% for total knee replacement, versus 0.136% in the nonsurgical controls. After adjustment for potential confounders, knee replacement increased the risk fivefold; open cholecystectomy almost fourfold; total hip replacement and simple mastectomy almost threefold; and laparoscopic cholecystectomy and open appendectomy almost twofold. Cesarean delivery increased the risk of chronic use by 28%.

With the exception of knee and hip replacements, “these procedures are not indicated to relieve pain and are not thought to place patients at risk for long-term pain ... Our results suggest that primary care clinicians and surgeons should monitor opioid use closely in the postsurgical period,” wrote Eric C. Sun, MD, PhD, of the department of anesthesiology, perioperative and pain medicine at Stanford (Calif.) University, and his colleagues.
Preoperative antidepressants and benzodiazepines carried about the same risk of chronic use as alcohol abuse (odds ratio 1.83; P less than .001), while drug abuse history increased the risk even more (OR 3.15; P less than .001). Male sex, age over 50 years, and history of depression were also associated with chronic use on multivariate analysis. Meanwhile, transurethral prostatectomy, laparoscopic appendectomy, functional endoscopic sinus surgery, and cataract surgery did not increase chronic use risk.
“Surgical patients, particularly those at higher risk for chronic opioid use, may benefit from techniques to reduce the risk such as multimodal analgesia and regional anesthesia, particularly in light of literature suggesting that these interventions may improve other perioperative outcomes ... Patients may also benefit from other preoperative and postoperative interventions, such as evidence-based psychobehavioral pain management skills,” the investigators said.
It wasn’t clear until now that even opioid-naive patients are at risk for opioid problems after surgery. Stanford’s investigation is not the first to link surgery and opioid abuse, but previous studies tended to focus on patients with preexisting use and more painful operations.
The study included prescriptions for oral and patch fentanyl, hydrocodone, oral hydromorphone, methadone, morphine, oxymorphone, and oxycodone. Hydrocodone cough remedies and acetaminophen/codeine analgesics were excluded.
Nonsurgical patients tended to be younger than their surgical peers (mean 42 vs. 44 years) and more likely to be male (49% vs. 26%).
The authors had no disclosures. The work was funded in part by the Foundation for Anesthesia Education and Research and the Anesthesia Quality Institute. Claims data came from MarketScan (Truven Health Analytics).
Common surgeries increase the risk of chronic opioid use in opioid-naive adults, especially those using antidepressants or benzodiazepines before their operations, and those with substance abuse histories, according to an insurance claims analysis from Stanford (Calif.) University.
The researchers reviewed opioid prescribing in the first postop year – excluding the first 90 days – for 641,941 patients and compared that information with opioid prescribing for 18,011,137 adult patients who did not have surgery. None of the subjects had filled an opioid prescription in the previous year (JAMA Intern Med. 2016 Jul 11. doi: 10.1001/jamainternmed.2016.3298).
Chronic opioid use, defined as filling at least 120 days of opioid prescriptions within the first year of surgery, ranged up to 1.41% for total knee replacement, versus 0.136% in the nonsurgical controls. After adjustment for potential confounders, knee replacement increased the risk fivefold; open cholecystectomy almost fourfold; total hip replacement and simple mastectomy almost threefold; and laparoscopic cholecystectomy and open appendectomy almost twofold. Cesarean delivery increased the risk of chronic use by 28%.

With the exception of knee and hip replacements, “these procedures are not indicated to relieve pain and are not thought to place patients at risk for long-term pain ... Our results suggest that primary care clinicians and surgeons should monitor opioid use closely in the postsurgical period,” wrote Eric C. Sun, MD, PhD, of the department of anesthesiology, perioperative and pain medicine at Stanford (Calif.) University, and his colleagues.
Preoperative antidepressants and benzodiazepines carried about the same risk of chronic use as alcohol abuse (odds ratio 1.83; P less than .001), while drug abuse history increased the risk even more (OR 3.15; P less than .001). Male sex, age over 50 years, and history of depression were also associated with chronic use on multivariate analysis. Meanwhile, transurethral prostatectomy, laparoscopic appendectomy, functional endoscopic sinus surgery, and cataract surgery did not increase chronic use risk.
“Surgical patients, particularly those at higher risk for chronic opioid use, may benefit from techniques to reduce the risk such as multimodal analgesia and regional anesthesia, particularly in light of literature suggesting that these interventions may improve other perioperative outcomes ... Patients may also benefit from other preoperative and postoperative interventions, such as evidence-based psychobehavioral pain management skills,” the investigators said.
It wasn’t clear until now that even opioid-naive patients are at risk for opioid problems after surgery. Stanford’s investigation is not the first to link surgery and opioid abuse, but previous studies tended to focus on patients with preexisting use and more painful operations.
The study included prescriptions for oral and patch fentanyl, hydrocodone, oral hydromorphone, methadone, morphine, oxymorphone, and oxycodone. Hydrocodone cough remedies and acetaminophen/codeine analgesics were excluded.
Nonsurgical patients tended to be younger than their surgical peers (mean 42 vs. 44 years) and more likely to be male (49% vs. 26%).
The authors had no disclosures. The work was funded in part by the Foundation for Anesthesia Education and Research and the Anesthesia Quality Institute. Claims data came from MarketScan (Truven Health Analytics).
FROM JAMA INTERNAL MEDICINE
Key clinical point: Common surgeries increase the risk of chronic opioid use in opioid-naive adults, especially among those using antidepressants or benzodiazepines before their operations, and those with substance abuse histories.
Major finding: After adjustment for potential confounders, knee replacement increased the risk fivefold; open cholecystectomy almost fourfold; and total hip replacement and simple mastectomy almost threefold.
Data source: Insurance claims of more than 18 million people.
Disclosures: The authors had no disclosures. The work was funded in part by the Foundation for Anesthesia Education and Research and the Anesthesia Quality Institute. Claims data came from MarketScan (Truven Health Analytics).
Cost of end-of-life care peaks at age 73 years
The cost of end-of-life care for Americans on traditional Medicare is higher for those in their early 70s than for beneficiaries in their 80s or 90s, according to the Kaiser Family Foundation.
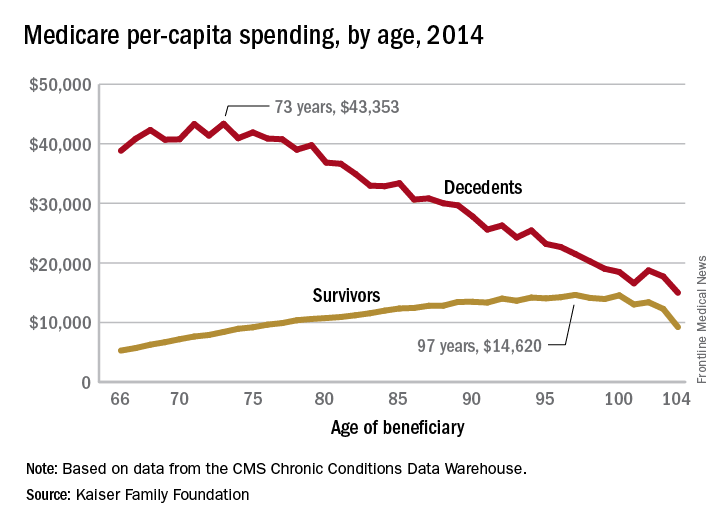
In 2014, the per-capita cost of care peaked at $43,353 for those who died at age 73, compared with $36,841 who died at age 80 and $27,779 for 90-year-old decedents, Kaiser found in its analysis of claims data from the Centers for Medicare & Medicaid Services Chronic Conditions Data Warehouse.
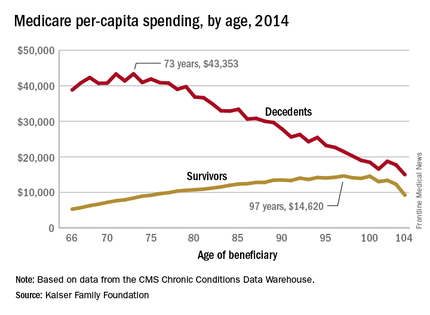
“This is a pattern we weren’t really expecting to see,” Juliette Cubanski, associate director of the program on Medicare policy for Kaiser, said in an article on the findings distributed by Kaiser Health News. “It kind of goes against the notion that doctors are throwing everything including the kitchen sink at people at the end of life regardless of how old they are,” she added.
The trend was quite different, and much less costly, for those who lived through the entire year. Their per-capita cost of care started at $5,271 for 66-year-olds and peaked at $14,620 for those aged 97. Medicare per capita spending for all decedents was nearly four times higher, at $34,529, than the $9,121 spent for each beneficiary who survived the year, the Kaiser report showed.
The largest share of that difference came from inpatient hospital care, which was 51% of decedents’ per-capita cost but only 27% for survivors. The cost for each group: $17,574 for decedents and $2,497 for survivors, according to Kaiser, which pointed out that its analysis covered only traditional Medicare beneficiaries during the calendar year in which they died and did not include spending in the full 12 months before their deaths.
The gap between decedents and survivors has narrowed in recent years. The growth rate from 2000 – when spending was $19,130 – to 2014 was 4.3% a year for decedents, while spending for survivors rose 5.5% annually from its $4,322 starting level at the turn of the century, the report noted.
The cost of end-of-life care for Americans on traditional Medicare is higher for those in their early 70s than for beneficiaries in their 80s or 90s, according to the Kaiser Family Foundation.
In 2014, the per-capita cost of care peaked at $43,353 for those who died at age 73, compared with $36,841 who died at age 80 and $27,779 for 90-year-old decedents, Kaiser found in its analysis of claims data from the Centers for Medicare & Medicaid Services Chronic Conditions Data Warehouse.

“This is a pattern we weren’t really expecting to see,” Juliette Cubanski, associate director of the program on Medicare policy for Kaiser, said in an article on the findings distributed by Kaiser Health News. “It kind of goes against the notion that doctors are throwing everything including the kitchen sink at people at the end of life regardless of how old they are,” she added.
The trend was quite different, and much less costly, for those who lived through the entire year. Their per-capita cost of care started at $5,271 for 66-year-olds and peaked at $14,620 for those aged 97. Medicare per capita spending for all decedents was nearly four times higher, at $34,529, than the $9,121 spent for each beneficiary who survived the year, the Kaiser report showed.
The largest share of that difference came from inpatient hospital care, which was 51% of decedents’ per-capita cost but only 27% for survivors. The cost for each group: $17,574 for decedents and $2,497 for survivors, according to Kaiser, which pointed out that its analysis covered only traditional Medicare beneficiaries during the calendar year in which they died and did not include spending in the full 12 months before their deaths.
The gap between decedents and survivors has narrowed in recent years. The growth rate from 2000 – when spending was $19,130 – to 2014 was 4.3% a year for decedents, while spending for survivors rose 5.5% annually from its $4,322 starting level at the turn of the century, the report noted.
The cost of end-of-life care for Americans on traditional Medicare is higher for those in their early 70s than for beneficiaries in their 80s or 90s, according to the Kaiser Family Foundation.
In 2014, the per-capita cost of care peaked at $43,353 for those who died at age 73, compared with $36,841 who died at age 80 and $27,779 for 90-year-old decedents, Kaiser found in its analysis of claims data from the Centers for Medicare & Medicaid Services Chronic Conditions Data Warehouse.

“This is a pattern we weren’t really expecting to see,” Juliette Cubanski, associate director of the program on Medicare policy for Kaiser, said in an article on the findings distributed by Kaiser Health News. “It kind of goes against the notion that doctors are throwing everything including the kitchen sink at people at the end of life regardless of how old they are,” she added.
The trend was quite different, and much less costly, for those who lived through the entire year. Their per-capita cost of care started at $5,271 for 66-year-olds and peaked at $14,620 for those aged 97. Medicare per capita spending for all decedents was nearly four times higher, at $34,529, than the $9,121 spent for each beneficiary who survived the year, the Kaiser report showed.
The largest share of that difference came from inpatient hospital care, which was 51% of decedents’ per-capita cost but only 27% for survivors. The cost for each group: $17,574 for decedents and $2,497 for survivors, according to Kaiser, which pointed out that its analysis covered only traditional Medicare beneficiaries during the calendar year in which they died and did not include spending in the full 12 months before their deaths.
The gap between decedents and survivors has narrowed in recent years. The growth rate from 2000 – when spending was $19,130 – to 2014 was 4.3% a year for decedents, while spending for survivors rose 5.5% annually from its $4,322 starting level at the turn of the century, the report noted.
Good Reading – Surgeon writers share their experiences with a wider audience
Are you casting about for a good book for yourself – maybe something to take on a long plane ride? Or are you looking for something for a young person interested in a career in surgery? Consider reading (or giving) a book written by a fellow surgeon.
To find such books, visit the ACS Surgeon Writers topic on the ACS Communities site. After just 6 months, this community has grown to 180 active members who share information on writing contests and conferences, pass on tips on mutual problems, and celebrate publications ranging from articles to full-length books. To participate in the dialogue, add your voice (and your publications) to our membership list. To find a book by a fellow surgeon, go through the files that form a sort of virtual bookshelf under the “Library” section. Don’t worry, you won’t find any thick surgical textbooks there, but rather books written for a wider audience. Fantasy, fiction, patient education, and memoir are all represented.
I’ve picked three memoirs to get you started. One is an anthology of pieces written by female surgeons. The second will take you into the world of transplant surgery. In the third, you accompany the surgeon-author to South Sudan on a mission for Doctors Without Borders (MSF). These true accounts, written by fellow surgeons, have the power to transport you into a world similar to, yet different from, your own surgical milieu.
Being a Woman Surgeon: Sixty Women Share Their Stories is a generous anthology collected and edited by Preeti John, MD, FACS. These short chapters are bite-sized reading tidbits that can be enjoyed in a few moments of spare time. You can read the book from cover to cover or dip into it randomly. It’s a great book to give to that young woman in your life – daughter, granddaughter, or mentee – who is thinking of a career in any of the surgical specialties (including, of course, general surgery). Female pediatric surgeons, orthopedic surgeons, general surgeons, and some leaders in the field of surgery contributed to this book.
Dr. John organized them by topic and by specialty, and included some interviews and poems at the end. It’s a generous slice of life. Surgeons share formative experiences from their training, the evolution of their careers, choice of paths, and the unfolding of their lives.
Last Night in the OR: A Transplant Surgeon’s Odyssey by Bud Shaw, MD, FACS, takes the reader along on a journey from the early days of liver transplant into the modern era. Many things in this book will resonate with the surgeon-reader. Three chapters aptly subtitled “Initiation” open the book. The year is 1981, and Dr. Shaw has just completed his surgical residency and begun a transplant fellowship in Pittsburgh. The humbling transformation from confident chief resident to beginner will ring true with any surgeon who has done a challenging fellowship. After an account of the first days’ chaotic, blinding confusion he ends with the admission that even abusive words, spoken in the heat of the moment, became phrases that he would “…in the distance of time and place, yearn to hear again.” Many who trained under the giants, in an era remote from work-hour limitations and political correctness, can identify with this sentiment.
The book threads nonlinearly, like memory itself, through time and space. Shaw includes his own experience with illness, and recounts how his surgeon-father reduced an inguinal hernia that could have been an ominous inguinal lymph node for his son. His father “was ninety years old then and he couldn’t remember what he’d had for breakfast, but he could still fix me with his hands.”
Ajak’s Song by Kenneth Waxman, MD, FACS, takes the reader to South Sudan with MSF. The account captures the frustrations and uncertainties of working in such an austere environment. General surgeons contemplating such a tour of duty will be interested in the medical details, including management of chronic osteomyelitis. From one such case comes the title of the book. Ajak, a young woman, develops a chronic open wound with exposed tibia after surviving a snake bite. Her path to the MSF hospital staffed by Dr. Waxman is circuitous, and she has already endured considerable treatment through an escalating series of healers. Amputation seems inevitable, but a plan is made to attempt to clean and heal the wound. Multiple operations are required. After her first procedure (and each subsequent one), Ajak awakens from anesthesia with a smile on her face, singing a song of thanks. As the small team waits with their young patient until she is ready to return to the ward, “Ajak repeatedly sings her lovely song.” By the end of the book, the reader will come to hear Ajak’s song as well.
For more good reading, go to the ACS Surgeon Writers Community Library. All the books listed are available through online booksellers and many are in bricks-and-mortar stores as well.
Dr. Scott-Conner is professor emeritus of surgery at the University of Iowa Carver College of Medicine. Visit Dr. Scott-Conner’s website (www.scott-conner.com) for information on ordering her works of fiction and nonfiction.
Are you casting about for a good book for yourself – maybe something to take on a long plane ride? Or are you looking for something for a young person interested in a career in surgery? Consider reading (or giving) a book written by a fellow surgeon.
To find such books, visit the ACS Surgeon Writers topic on the ACS Communities site. After just 6 months, this community has grown to 180 active members who share information on writing contests and conferences, pass on tips on mutual problems, and celebrate publications ranging from articles to full-length books. To participate in the dialogue, add your voice (and your publications) to our membership list. To find a book by a fellow surgeon, go through the files that form a sort of virtual bookshelf under the “Library” section. Don’t worry, you won’t find any thick surgical textbooks there, but rather books written for a wider audience. Fantasy, fiction, patient education, and memoir are all represented.
I’ve picked three memoirs to get you started. One is an anthology of pieces written by female surgeons. The second will take you into the world of transplant surgery. In the third, you accompany the surgeon-author to South Sudan on a mission for Doctors Without Borders (MSF). These true accounts, written by fellow surgeons, have the power to transport you into a world similar to, yet different from, your own surgical milieu.
Being a Woman Surgeon: Sixty Women Share Their Stories is a generous anthology collected and edited by Preeti John, MD, FACS. These short chapters are bite-sized reading tidbits that can be enjoyed in a few moments of spare time. You can read the book from cover to cover or dip into it randomly. It’s a great book to give to that young woman in your life – daughter, granddaughter, or mentee – who is thinking of a career in any of the surgical specialties (including, of course, general surgery). Female pediatric surgeons, orthopedic surgeons, general surgeons, and some leaders in the field of surgery contributed to this book.
Dr. John organized them by topic and by specialty, and included some interviews and poems at the end. It’s a generous slice of life. Surgeons share formative experiences from their training, the evolution of their careers, choice of paths, and the unfolding of their lives.
Last Night in the OR: A Transplant Surgeon’s Odyssey by Bud Shaw, MD, FACS, takes the reader along on a journey from the early days of liver transplant into the modern era. Many things in this book will resonate with the surgeon-reader. Three chapters aptly subtitled “Initiation” open the book. The year is 1981, and Dr. Shaw has just completed his surgical residency and begun a transplant fellowship in Pittsburgh. The humbling transformation from confident chief resident to beginner will ring true with any surgeon who has done a challenging fellowship. After an account of the first days’ chaotic, blinding confusion he ends with the admission that even abusive words, spoken in the heat of the moment, became phrases that he would “…in the distance of time and place, yearn to hear again.” Many who trained under the giants, in an era remote from work-hour limitations and political correctness, can identify with this sentiment.
The book threads nonlinearly, like memory itself, through time and space. Shaw includes his own experience with illness, and recounts how his surgeon-father reduced an inguinal hernia that could have been an ominous inguinal lymph node for his son. His father “was ninety years old then and he couldn’t remember what he’d had for breakfast, but he could still fix me with his hands.”
Ajak’s Song by Kenneth Waxman, MD, FACS, takes the reader to South Sudan with MSF. The account captures the frustrations and uncertainties of working in such an austere environment. General surgeons contemplating such a tour of duty will be interested in the medical details, including management of chronic osteomyelitis. From one such case comes the title of the book. Ajak, a young woman, develops a chronic open wound with exposed tibia after surviving a snake bite. Her path to the MSF hospital staffed by Dr. Waxman is circuitous, and she has already endured considerable treatment through an escalating series of healers. Amputation seems inevitable, but a plan is made to attempt to clean and heal the wound. Multiple operations are required. After her first procedure (and each subsequent one), Ajak awakens from anesthesia with a smile on her face, singing a song of thanks. As the small team waits with their young patient until she is ready to return to the ward, “Ajak repeatedly sings her lovely song.” By the end of the book, the reader will come to hear Ajak’s song as well.
For more good reading, go to the ACS Surgeon Writers Community Library. All the books listed are available through online booksellers and many are in bricks-and-mortar stores as well.
Dr. Scott-Conner is professor emeritus of surgery at the University of Iowa Carver College of Medicine. Visit Dr. Scott-Conner’s website (www.scott-conner.com) for information on ordering her works of fiction and nonfiction.
Are you casting about for a good book for yourself – maybe something to take on a long plane ride? Or are you looking for something for a young person interested in a career in surgery? Consider reading (or giving) a book written by a fellow surgeon.
To find such books, visit the ACS Surgeon Writers topic on the ACS Communities site. After just 6 months, this community has grown to 180 active members who share information on writing contests and conferences, pass on tips on mutual problems, and celebrate publications ranging from articles to full-length books. To participate in the dialogue, add your voice (and your publications) to our membership list. To find a book by a fellow surgeon, go through the files that form a sort of virtual bookshelf under the “Library” section. Don’t worry, you won’t find any thick surgical textbooks there, but rather books written for a wider audience. Fantasy, fiction, patient education, and memoir are all represented.
I’ve picked three memoirs to get you started. One is an anthology of pieces written by female surgeons. The second will take you into the world of transplant surgery. In the third, you accompany the surgeon-author to South Sudan on a mission for Doctors Without Borders (MSF). These true accounts, written by fellow surgeons, have the power to transport you into a world similar to, yet different from, your own surgical milieu.
Being a Woman Surgeon: Sixty Women Share Their Stories is a generous anthology collected and edited by Preeti John, MD, FACS. These short chapters are bite-sized reading tidbits that can be enjoyed in a few moments of spare time. You can read the book from cover to cover or dip into it randomly. It’s a great book to give to that young woman in your life – daughter, granddaughter, or mentee – who is thinking of a career in any of the surgical specialties (including, of course, general surgery). Female pediatric surgeons, orthopedic surgeons, general surgeons, and some leaders in the field of surgery contributed to this book.
Dr. John organized them by topic and by specialty, and included some interviews and poems at the end. It’s a generous slice of life. Surgeons share formative experiences from their training, the evolution of their careers, choice of paths, and the unfolding of their lives.
Last Night in the OR: A Transplant Surgeon’s Odyssey by Bud Shaw, MD, FACS, takes the reader along on a journey from the early days of liver transplant into the modern era. Many things in this book will resonate with the surgeon-reader. Three chapters aptly subtitled “Initiation” open the book. The year is 1981, and Dr. Shaw has just completed his surgical residency and begun a transplant fellowship in Pittsburgh. The humbling transformation from confident chief resident to beginner will ring true with any surgeon who has done a challenging fellowship. After an account of the first days’ chaotic, blinding confusion he ends with the admission that even abusive words, spoken in the heat of the moment, became phrases that he would “…in the distance of time and place, yearn to hear again.” Many who trained under the giants, in an era remote from work-hour limitations and political correctness, can identify with this sentiment.
The book threads nonlinearly, like memory itself, through time and space. Shaw includes his own experience with illness, and recounts how his surgeon-father reduced an inguinal hernia that could have been an ominous inguinal lymph node for his son. His father “was ninety years old then and he couldn’t remember what he’d had for breakfast, but he could still fix me with his hands.”
Ajak’s Song by Kenneth Waxman, MD, FACS, takes the reader to South Sudan with MSF. The account captures the frustrations and uncertainties of working in such an austere environment. General surgeons contemplating such a tour of duty will be interested in the medical details, including management of chronic osteomyelitis. From one such case comes the title of the book. Ajak, a young woman, develops a chronic open wound with exposed tibia after surviving a snake bite. Her path to the MSF hospital staffed by Dr. Waxman is circuitous, and she has already endured considerable treatment through an escalating series of healers. Amputation seems inevitable, but a plan is made to attempt to clean and heal the wound. Multiple operations are required. After her first procedure (and each subsequent one), Ajak awakens from anesthesia with a smile on her face, singing a song of thanks. As the small team waits with their young patient until she is ready to return to the ward, “Ajak repeatedly sings her lovely song.” By the end of the book, the reader will come to hear Ajak’s song as well.
For more good reading, go to the ACS Surgeon Writers Community Library. All the books listed are available through online booksellers and many are in bricks-and-mortar stores as well.
Dr. Scott-Conner is professor emeritus of surgery at the University of Iowa Carver College of Medicine. Visit Dr. Scott-Conner’s website (www.scott-conner.com) for information on ordering her works of fiction and nonfiction.
From the Washington Office: The operationalization of MACRA
On April 27, 2016, the Centers for Medicare and Medicaid Services (CMS) released its proposed rule on the Medicare Access and CHIP Reauthorization Act (MACRA). Fellows will remember that the MACRA legislation, passed in April of 2015, permanently repealed the Sustainable Growth Rate (SGR) formula and thus, represents the greatest sea change in Medicare physician payment since the establishment of the RBRVS (Resource-Based Relative Value Scale) in 1992.
In broadest policy terms, the law continues to advance the CMS policy goal of basing payment on quality and value over volume. From a granular perspective, the law combines Medicare’s three current quality programs into one new system. CMS published this 982-page proposed rule after reviewing the comments submitted by ACS and other interested parties in response to its request for information last fall. As I write, staff of the Division of Advocacy and Health Policy are in the process of crafting the ACS response to the proposed rule. Comments were due on June 27, 2016. It is anticipated that CMS will publish the final rule later this year, likely in late October or early November. Accordingly, the following description of the implementation of MACRA is based on the current understanding from the proposed rule, and is likely to change in some respects with the final rule.
CMS has designated the payment program operationalizing the MACRA law as the Quality Payment Program (QPP). The QPP has two tracks, the Merit-based Incentive Payment System (MIPS) and advanced Alternative Payment Models (APMs). For the first several years of the program, it is widely expected that the vast majority of physicians will participate in the QPP via the MIPS track. As such, I will direct the remainder of the text this month to the MIPS program.
MIPS: Merit-Based Incentive Payment System
The Merit-based Incentive Payment System, MIPS, consists of four components. They are: Quality, Resource Use, Advancing Care Information (ACI) and the Clinical Practice Improvement Activities (CPIA). Though the names have changed, Fellows are familiar with the substance of three of the components. For example, the Quality component replaces the Physician Quality Reporting System (PQRS); the Resource Use component replaces the Value-Based Modifier (VBM); and the Advancing Care Information component modifies and replaces the Electronic Health Record Meaningful Use (EHR-MU) program. The fourth component of MIPS is the new Clinical Practice Improvement Activities, which the legislation designates as intended to provide “credit for work to improve practice and facilitates future participation in alternative payment models.”
Composite Performance Score: MIPS participants will be assigned a Composite Performance Score based on their performance in all four components. For 2017, the first year for assessment under the QPP, 50 percent of the score will be based on performance in the Quality component, 10 percent will be based on the Resource Use component, 25 percent will be based on the Advancing Care Information component, and 15 percent will be based on the Clinical Practice Improvement Activities. In future years, there will be a gradual increase in the relative value of the Resource Use component with an equal and accompanying decrease in the value of the Quality component. As proposed, by the third assessment year (2019), the Quality and Resource Use components are expected to each account for 30 percent of the Composite Performance Score.
MIPS Quality Component: Though the Quality component of MIPS replaces the PQRS, CMS is proposing some changes that Fellows will welcome. As opposed to the previous PQRS requirement to report nine measures, the MIPS Quality component requires providers to report only six measures. One of these six must be an “Outcome” measure and another must be a “Cross-cutting” measure. While the reporting threshold for the percentage of patients on which reports will be required is proposed to increase substantially, ACS and other physician groups will be advocating that the required percentage published in the final rule be close to the 50 percent level found in current law.
Resources Use Component: There is also some good news relative to the Resource Use component in that there are NO reporting requirements. CMS will calculate this component from Medicare claims data and base its assessment of individual provider performance on the resource measures currently used for the Value-Based Modifier. Namely, those are the VBM Total per Capita Cost measure and the VBM Medicare Spending per Beneficiary measure. In addition, CMS will also be taking into account measures that specifically focus on episodes of care, something for which the College has previously advocated. Beginning in 2018, CMS plans to also take into consideration factors of patient condition and patient relation in order to address physician concerns about risk adjustment and attribution.
Advancing Care Information Component: This modifies and replaces the Electronic Health Record Meaningful Use program. The score for this component is derived in two parts, a Base score (50 percent) and a Performance score (up to an additional 50 percent). The threshold for achieving the Base score remains “all or nothing.” Only after meeting the requirements for the Base score is one eligible to receive additional Performance score credit based on the level of performance on a subset of the same measures required to achieve the Base score. Assessment in 2017 will be based on the EHR-MU requirements published in the 2015 Final Rule for the EHR-MU program.
Clinical Practice Improvement Component: The fourth component of MIPS is the Clinical Practice Improvement Activities component. As mentioned previously, this is a new requirement with no prior analogous program requirement. As such, it is very much in evolution. In the first year of MIPS assessment (2017), achieving full credit for the CPIA component should not pose much additional administrative burden as reporting will be by simple attestation. Physicians will chose from a list of 94 activities assigned two different weighted values. In order to receive full credit for the CPIA component, most providers will need to attest that they have participated in a minimum of three and a maximum of six of the 94 activities, depending on the weight of the activities selected, for 90 days.
As outlined above, the final rule on the Quality Payment Program is expected to be released in late October or early November. As proposed, assessment under the provisions of that final rule would begin in January of 2017. This leaves all providers with a very short time window in which to become familiar with the program that will impact their Medicare payment beginning in 2019.
Accordingly, in the next several editions of this column, I will provide more specific information about each of the four MIPS components, the scoring mechanism for MIPS assessment and the aforementioned alternate track to MIPS, the Alternative Payment Models. While it is easy to understand how many Fellows could initially find this change daunting and overwhelming, I am confident that with a minimal investment of time all can develop adequate working knowledge of the MIPS and APMs to participate successfully in the QPP.
Until next month...
Dr. Bailey is an ACS Fellow, a pediatric surgeon, and Medical Director, Advocacy, for the Division of Advocacy and Health Policy, in the ACS offices in Washington.
On April 27, 2016, the Centers for Medicare and Medicaid Services (CMS) released its proposed rule on the Medicare Access and CHIP Reauthorization Act (MACRA). Fellows will remember that the MACRA legislation, passed in April of 2015, permanently repealed the Sustainable Growth Rate (SGR) formula and thus, represents the greatest sea change in Medicare physician payment since the establishment of the RBRVS (Resource-Based Relative Value Scale) in 1992.
In broadest policy terms, the law continues to advance the CMS policy goal of basing payment on quality and value over volume. From a granular perspective, the law combines Medicare’s three current quality programs into one new system. CMS published this 982-page proposed rule after reviewing the comments submitted by ACS and other interested parties in response to its request for information last fall. As I write, staff of the Division of Advocacy and Health Policy are in the process of crafting the ACS response to the proposed rule. Comments were due on June 27, 2016. It is anticipated that CMS will publish the final rule later this year, likely in late October or early November. Accordingly, the following description of the implementation of MACRA is based on the current understanding from the proposed rule, and is likely to change in some respects with the final rule.
CMS has designated the payment program operationalizing the MACRA law as the Quality Payment Program (QPP). The QPP has two tracks, the Merit-based Incentive Payment System (MIPS) and advanced Alternative Payment Models (APMs). For the first several years of the program, it is widely expected that the vast majority of physicians will participate in the QPP via the MIPS track. As such, I will direct the remainder of the text this month to the MIPS program.
MIPS: Merit-Based Incentive Payment System
The Merit-based Incentive Payment System, MIPS, consists of four components. They are: Quality, Resource Use, Advancing Care Information (ACI) and the Clinical Practice Improvement Activities (CPIA). Though the names have changed, Fellows are familiar with the substance of three of the components. For example, the Quality component replaces the Physician Quality Reporting System (PQRS); the Resource Use component replaces the Value-Based Modifier (VBM); and the Advancing Care Information component modifies and replaces the Electronic Health Record Meaningful Use (EHR-MU) program. The fourth component of MIPS is the new Clinical Practice Improvement Activities, which the legislation designates as intended to provide “credit for work to improve practice and facilitates future participation in alternative payment models.”
Composite Performance Score: MIPS participants will be assigned a Composite Performance Score based on their performance in all four components. For 2017, the first year for assessment under the QPP, 50 percent of the score will be based on performance in the Quality component, 10 percent will be based on the Resource Use component, 25 percent will be based on the Advancing Care Information component, and 15 percent will be based on the Clinical Practice Improvement Activities. In future years, there will be a gradual increase in the relative value of the Resource Use component with an equal and accompanying decrease in the value of the Quality component. As proposed, by the third assessment year (2019), the Quality and Resource Use components are expected to each account for 30 percent of the Composite Performance Score.
MIPS Quality Component: Though the Quality component of MIPS replaces the PQRS, CMS is proposing some changes that Fellows will welcome. As opposed to the previous PQRS requirement to report nine measures, the MIPS Quality component requires providers to report only six measures. One of these six must be an “Outcome” measure and another must be a “Cross-cutting” measure. While the reporting threshold for the percentage of patients on which reports will be required is proposed to increase substantially, ACS and other physician groups will be advocating that the required percentage published in the final rule be close to the 50 percent level found in current law.
Resources Use Component: There is also some good news relative to the Resource Use component in that there are NO reporting requirements. CMS will calculate this component from Medicare claims data and base its assessment of individual provider performance on the resource measures currently used for the Value-Based Modifier. Namely, those are the VBM Total per Capita Cost measure and the VBM Medicare Spending per Beneficiary measure. In addition, CMS will also be taking into account measures that specifically focus on episodes of care, something for which the College has previously advocated. Beginning in 2018, CMS plans to also take into consideration factors of patient condition and patient relation in order to address physician concerns about risk adjustment and attribution.
Advancing Care Information Component: This modifies and replaces the Electronic Health Record Meaningful Use program. The score for this component is derived in two parts, a Base score (50 percent) and a Performance score (up to an additional 50 percent). The threshold for achieving the Base score remains “all or nothing.” Only after meeting the requirements for the Base score is one eligible to receive additional Performance score credit based on the level of performance on a subset of the same measures required to achieve the Base score. Assessment in 2017 will be based on the EHR-MU requirements published in the 2015 Final Rule for the EHR-MU program.
Clinical Practice Improvement Component: The fourth component of MIPS is the Clinical Practice Improvement Activities component. As mentioned previously, this is a new requirement with no prior analogous program requirement. As such, it is very much in evolution. In the first year of MIPS assessment (2017), achieving full credit for the CPIA component should not pose much additional administrative burden as reporting will be by simple attestation. Physicians will chose from a list of 94 activities assigned two different weighted values. In order to receive full credit for the CPIA component, most providers will need to attest that they have participated in a minimum of three and a maximum of six of the 94 activities, depending on the weight of the activities selected, for 90 days.
As outlined above, the final rule on the Quality Payment Program is expected to be released in late October or early November. As proposed, assessment under the provisions of that final rule would begin in January of 2017. This leaves all providers with a very short time window in which to become familiar with the program that will impact their Medicare payment beginning in 2019.
Accordingly, in the next several editions of this column, I will provide more specific information about each of the four MIPS components, the scoring mechanism for MIPS assessment and the aforementioned alternate track to MIPS, the Alternative Payment Models. While it is easy to understand how many Fellows could initially find this change daunting and overwhelming, I am confident that with a minimal investment of time all can develop adequate working knowledge of the MIPS and APMs to participate successfully in the QPP.
Until next month...
Dr. Bailey is an ACS Fellow, a pediatric surgeon, and Medical Director, Advocacy, for the Division of Advocacy and Health Policy, in the ACS offices in Washington.
On April 27, 2016, the Centers for Medicare and Medicaid Services (CMS) released its proposed rule on the Medicare Access and CHIP Reauthorization Act (MACRA). Fellows will remember that the MACRA legislation, passed in April of 2015, permanently repealed the Sustainable Growth Rate (SGR) formula and thus, represents the greatest sea change in Medicare physician payment since the establishment of the RBRVS (Resource-Based Relative Value Scale) in 1992.
In broadest policy terms, the law continues to advance the CMS policy goal of basing payment on quality and value over volume. From a granular perspective, the law combines Medicare’s three current quality programs into one new system. CMS published this 982-page proposed rule after reviewing the comments submitted by ACS and other interested parties in response to its request for information last fall. As I write, staff of the Division of Advocacy and Health Policy are in the process of crafting the ACS response to the proposed rule. Comments were due on June 27, 2016. It is anticipated that CMS will publish the final rule later this year, likely in late October or early November. Accordingly, the following description of the implementation of MACRA is based on the current understanding from the proposed rule, and is likely to change in some respects with the final rule.
CMS has designated the payment program operationalizing the MACRA law as the Quality Payment Program (QPP). The QPP has two tracks, the Merit-based Incentive Payment System (MIPS) and advanced Alternative Payment Models (APMs). For the first several years of the program, it is widely expected that the vast majority of physicians will participate in the QPP via the MIPS track. As such, I will direct the remainder of the text this month to the MIPS program.
MIPS: Merit-Based Incentive Payment System
The Merit-based Incentive Payment System, MIPS, consists of four components. They are: Quality, Resource Use, Advancing Care Information (ACI) and the Clinical Practice Improvement Activities (CPIA). Though the names have changed, Fellows are familiar with the substance of three of the components. For example, the Quality component replaces the Physician Quality Reporting System (PQRS); the Resource Use component replaces the Value-Based Modifier (VBM); and the Advancing Care Information component modifies and replaces the Electronic Health Record Meaningful Use (EHR-MU) program. The fourth component of MIPS is the new Clinical Practice Improvement Activities, which the legislation designates as intended to provide “credit for work to improve practice and facilitates future participation in alternative payment models.”
Composite Performance Score: MIPS participants will be assigned a Composite Performance Score based on their performance in all four components. For 2017, the first year for assessment under the QPP, 50 percent of the score will be based on performance in the Quality component, 10 percent will be based on the Resource Use component, 25 percent will be based on the Advancing Care Information component, and 15 percent will be based on the Clinical Practice Improvement Activities. In future years, there will be a gradual increase in the relative value of the Resource Use component with an equal and accompanying decrease in the value of the Quality component. As proposed, by the third assessment year (2019), the Quality and Resource Use components are expected to each account for 30 percent of the Composite Performance Score.
MIPS Quality Component: Though the Quality component of MIPS replaces the PQRS, CMS is proposing some changes that Fellows will welcome. As opposed to the previous PQRS requirement to report nine measures, the MIPS Quality component requires providers to report only six measures. One of these six must be an “Outcome” measure and another must be a “Cross-cutting” measure. While the reporting threshold for the percentage of patients on which reports will be required is proposed to increase substantially, ACS and other physician groups will be advocating that the required percentage published in the final rule be close to the 50 percent level found in current law.
Resources Use Component: There is also some good news relative to the Resource Use component in that there are NO reporting requirements. CMS will calculate this component from Medicare claims data and base its assessment of individual provider performance on the resource measures currently used for the Value-Based Modifier. Namely, those are the VBM Total per Capita Cost measure and the VBM Medicare Spending per Beneficiary measure. In addition, CMS will also be taking into account measures that specifically focus on episodes of care, something for which the College has previously advocated. Beginning in 2018, CMS plans to also take into consideration factors of patient condition and patient relation in order to address physician concerns about risk adjustment and attribution.
Advancing Care Information Component: This modifies and replaces the Electronic Health Record Meaningful Use program. The score for this component is derived in two parts, a Base score (50 percent) and a Performance score (up to an additional 50 percent). The threshold for achieving the Base score remains “all or nothing.” Only after meeting the requirements for the Base score is one eligible to receive additional Performance score credit based on the level of performance on a subset of the same measures required to achieve the Base score. Assessment in 2017 will be based on the EHR-MU requirements published in the 2015 Final Rule for the EHR-MU program.
Clinical Practice Improvement Component: The fourth component of MIPS is the Clinical Practice Improvement Activities component. As mentioned previously, this is a new requirement with no prior analogous program requirement. As such, it is very much in evolution. In the first year of MIPS assessment (2017), achieving full credit for the CPIA component should not pose much additional administrative burden as reporting will be by simple attestation. Physicians will chose from a list of 94 activities assigned two different weighted values. In order to receive full credit for the CPIA component, most providers will need to attest that they have participated in a minimum of three and a maximum of six of the 94 activities, depending on the weight of the activities selected, for 90 days.
As outlined above, the final rule on the Quality Payment Program is expected to be released in late October or early November. As proposed, assessment under the provisions of that final rule would begin in January of 2017. This leaves all providers with a very short time window in which to become familiar with the program that will impact their Medicare payment beginning in 2019.
Accordingly, in the next several editions of this column, I will provide more specific information about each of the four MIPS components, the scoring mechanism for MIPS assessment and the aforementioned alternate track to MIPS, the Alternative Payment Models. While it is easy to understand how many Fellows could initially find this change daunting and overwhelming, I am confident that with a minimal investment of time all can develop adequate working knowledge of the MIPS and APMs to participate successfully in the QPP.
Until next month...
Dr. Bailey is an ACS Fellow, a pediatric surgeon, and Medical Director, Advocacy, for the Division of Advocacy and Health Policy, in the ACS offices in Washington.
Syndecan-1 may predict kidney injury after ped heart surgery
Acute kidney injury is a common complication after pediatric cardiac surgery, but measuring for a specific genetic protein immediately after cardiac surgery may improve cardiac surgeons’ ability to predict patients at higher risk of AKI, according to researchers from Brazil. The study results are in the July issue of the Journal of Thoracic and Cardiovascular Surgery (2016;152-178-86).
“Plasma syndecan-1 levels measured early in the postoperative period were independently associated with severe acute kidney injury,” wrote Candice Torres de Melo Bezerra Cavalcante, MD, of Heart Hospital of Messejana and Federal University of Ceará.
Their prospective cohort study involved 289 pediatric patients who had cardiac surgery at their institution between September 2013 and December 2014.
Dr. Cavalcante and colleagues acknowledged that the traditional biomarker for renal function, serum creatinine, only increases appreciably after the glomerular filtration rate declines 50%, impairing physicians’ ability to detect AKI early enough to treat it. “This delay can explain, in part the, negative results in AKI therapeutic clinical trials,” they wrote.
They evaluated two different endothelial biomarkers in addition to syndecan-1 with regard to their capacity for predicting severe AKI: plasma ICAM-1, a marker of endothelial cell activation; and E-selectin, an endothelial cell adhesion molecule. Syndecan-1 works as a biomarker of injury to the glycocalyx protein that surrounds endothelial cell membranes that acts as a permeability barrier and prevents the cells from adhering to blood. They found that median syndecan-1 levels soon after surgery were higher in patients with severe AKI, 103.6 vs. 42.3 ng/mL.
“Although syndecan-1 is not a renal-specific biomarker, there has been recent increasing evidence that endothelial injury has an important role in AKI pathophysiology,” the researchers noted.
Study results showed the higher the level of syndecan-1, the greater the adjusted odds ratio (OR) for severe AKI. Levels of less than 17 ng/mL were considered normal; 17.1-46.7 ng/mL carried an adjusted OR of 1.42; 47.4-93.1 ng/mL had an adjusted OR of 2.05; and levels 96.3 or greater had an OR of 8.87.
“Maintenance of endothelial glycocalyx integrity can be a therapeutic target to reduce AKI in this setting,” the researchers wrote.
The authors acknowledged that the study was done at a single center that had dialysis and death rates three and five times higher, respectively, than those of developed countries; and it measured syndecan-1 at only one time point almost immediately after the operation.
“Adding postoperative syndecan-1, even when using a clinical model that already incorporates variables from renal angina index, results in significant improvement in the capacity to predict severe AKI,” Dr. Cavalcante and colleagues concluded.
They had no financial relationships to disclose.
Results of AKI in heart surgery patients have been “sobering,” with up to 56% of these patients being diagnosed with AKI, but research such as that by Dr. Cavalcante and colleagues represents a new approach to improving outcomes by combining clinical risk factors with specific biomarkers to identify patients at risk, Petros V. Anagnostopoulos, MD, of American Family Children’s Hospital, University of Wisconsin, said in his invited commentary (J Thorac Cardiovasc Surg. 2016;152[1]:187-8).
Dr. Anagnostopoulos acknowledged problems with traditional markers for renal function. “An ideal biomarker should be sensitive, easy to measure, reproducible, and inexpensive,” he said. “Finally, when combined with clinical prediction models, it should potentiate the discrimination of these models.”
Syndecan-1 answers that call, he said. “It peaks early and is cheap, fast, and easy to measure with readily available methods, which makes it an ideal early biomarker of AKI,” Dr. Anagnostopoulos said. Even so, he pointed out potential shortcomings of syndecan-1: It is not renal specific and it does not increase before the operation.
But he applauded Dr. Cavalcante and colleagues for pursuing research to combine clinical risk factors with specific biomarkers. “It is very likely that this type of clinical research will become prevalent in the near future and will hopefully produce results that will allow better individual patient-specific risk stratification,” Dr. Anagnostopoulos said.
He had no financial relationships to disclose.
Results of AKI in heart surgery patients have been “sobering,” with up to 56% of these patients being diagnosed with AKI, but research such as that by Dr. Cavalcante and colleagues represents a new approach to improving outcomes by combining clinical risk factors with specific biomarkers to identify patients at risk, Petros V. Anagnostopoulos, MD, of American Family Children’s Hospital, University of Wisconsin, said in his invited commentary (J Thorac Cardiovasc Surg. 2016;152[1]:187-8).
Dr. Anagnostopoulos acknowledged problems with traditional markers for renal function. “An ideal biomarker should be sensitive, easy to measure, reproducible, and inexpensive,” he said. “Finally, when combined with clinical prediction models, it should potentiate the discrimination of these models.”
Syndecan-1 answers that call, he said. “It peaks early and is cheap, fast, and easy to measure with readily available methods, which makes it an ideal early biomarker of AKI,” Dr. Anagnostopoulos said. Even so, he pointed out potential shortcomings of syndecan-1: It is not renal specific and it does not increase before the operation.
But he applauded Dr. Cavalcante and colleagues for pursuing research to combine clinical risk factors with specific biomarkers. “It is very likely that this type of clinical research will become prevalent in the near future and will hopefully produce results that will allow better individual patient-specific risk stratification,” Dr. Anagnostopoulos said.
He had no financial relationships to disclose.
Results of AKI in heart surgery patients have been “sobering,” with up to 56% of these patients being diagnosed with AKI, but research such as that by Dr. Cavalcante and colleagues represents a new approach to improving outcomes by combining clinical risk factors with specific biomarkers to identify patients at risk, Petros V. Anagnostopoulos, MD, of American Family Children’s Hospital, University of Wisconsin, said in his invited commentary (J Thorac Cardiovasc Surg. 2016;152[1]:187-8).
Dr. Anagnostopoulos acknowledged problems with traditional markers for renal function. “An ideal biomarker should be sensitive, easy to measure, reproducible, and inexpensive,” he said. “Finally, when combined with clinical prediction models, it should potentiate the discrimination of these models.”
Syndecan-1 answers that call, he said. “It peaks early and is cheap, fast, and easy to measure with readily available methods, which makes it an ideal early biomarker of AKI,” Dr. Anagnostopoulos said. Even so, he pointed out potential shortcomings of syndecan-1: It is not renal specific and it does not increase before the operation.
But he applauded Dr. Cavalcante and colleagues for pursuing research to combine clinical risk factors with specific biomarkers. “It is very likely that this type of clinical research will become prevalent in the near future and will hopefully produce results that will allow better individual patient-specific risk stratification,” Dr. Anagnostopoulos said.
He had no financial relationships to disclose.
Acute kidney injury is a common complication after pediatric cardiac surgery, but measuring for a specific genetic protein immediately after cardiac surgery may improve cardiac surgeons’ ability to predict patients at higher risk of AKI, according to researchers from Brazil. The study results are in the July issue of the Journal of Thoracic and Cardiovascular Surgery (2016;152-178-86).
“Plasma syndecan-1 levels measured early in the postoperative period were independently associated with severe acute kidney injury,” wrote Candice Torres de Melo Bezerra Cavalcante, MD, of Heart Hospital of Messejana and Federal University of Ceará.
Their prospective cohort study involved 289 pediatric patients who had cardiac surgery at their institution between September 2013 and December 2014.
Dr. Cavalcante and colleagues acknowledged that the traditional biomarker for renal function, serum creatinine, only increases appreciably after the glomerular filtration rate declines 50%, impairing physicians’ ability to detect AKI early enough to treat it. “This delay can explain, in part the, negative results in AKI therapeutic clinical trials,” they wrote.
They evaluated two different endothelial biomarkers in addition to syndecan-1 with regard to their capacity for predicting severe AKI: plasma ICAM-1, a marker of endothelial cell activation; and E-selectin, an endothelial cell adhesion molecule. Syndecan-1 works as a biomarker of injury to the glycocalyx protein that surrounds endothelial cell membranes that acts as a permeability barrier and prevents the cells from adhering to blood. They found that median syndecan-1 levels soon after surgery were higher in patients with severe AKI, 103.6 vs. 42.3 ng/mL.
“Although syndecan-1 is not a renal-specific biomarker, there has been recent increasing evidence that endothelial injury has an important role in AKI pathophysiology,” the researchers noted.
Study results showed the higher the level of syndecan-1, the greater the adjusted odds ratio (OR) for severe AKI. Levels of less than 17 ng/mL were considered normal; 17.1-46.7 ng/mL carried an adjusted OR of 1.42; 47.4-93.1 ng/mL had an adjusted OR of 2.05; and levels 96.3 or greater had an OR of 8.87.
“Maintenance of endothelial glycocalyx integrity can be a therapeutic target to reduce AKI in this setting,” the researchers wrote.
The authors acknowledged that the study was done at a single center that had dialysis and death rates three and five times higher, respectively, than those of developed countries; and it measured syndecan-1 at only one time point almost immediately after the operation.
“Adding postoperative syndecan-1, even when using a clinical model that already incorporates variables from renal angina index, results in significant improvement in the capacity to predict severe AKI,” Dr. Cavalcante and colleagues concluded.
They had no financial relationships to disclose.
Acute kidney injury is a common complication after pediatric cardiac surgery, but measuring for a specific genetic protein immediately after cardiac surgery may improve cardiac surgeons’ ability to predict patients at higher risk of AKI, according to researchers from Brazil. The study results are in the July issue of the Journal of Thoracic and Cardiovascular Surgery (2016;152-178-86).
“Plasma syndecan-1 levels measured early in the postoperative period were independently associated with severe acute kidney injury,” wrote Candice Torres de Melo Bezerra Cavalcante, MD, of Heart Hospital of Messejana and Federal University of Ceará.
Their prospective cohort study involved 289 pediatric patients who had cardiac surgery at their institution between September 2013 and December 2014.
Dr. Cavalcante and colleagues acknowledged that the traditional biomarker for renal function, serum creatinine, only increases appreciably after the glomerular filtration rate declines 50%, impairing physicians’ ability to detect AKI early enough to treat it. “This delay can explain, in part the, negative results in AKI therapeutic clinical trials,” they wrote.
They evaluated two different endothelial biomarkers in addition to syndecan-1 with regard to their capacity for predicting severe AKI: plasma ICAM-1, a marker of endothelial cell activation; and E-selectin, an endothelial cell adhesion molecule. Syndecan-1 works as a biomarker of injury to the glycocalyx protein that surrounds endothelial cell membranes that acts as a permeability barrier and prevents the cells from adhering to blood. They found that median syndecan-1 levels soon after surgery were higher in patients with severe AKI, 103.6 vs. 42.3 ng/mL.
“Although syndecan-1 is not a renal-specific biomarker, there has been recent increasing evidence that endothelial injury has an important role in AKI pathophysiology,” the researchers noted.
Study results showed the higher the level of syndecan-1, the greater the adjusted odds ratio (OR) for severe AKI. Levels of less than 17 ng/mL were considered normal; 17.1-46.7 ng/mL carried an adjusted OR of 1.42; 47.4-93.1 ng/mL had an adjusted OR of 2.05; and levels 96.3 or greater had an OR of 8.87.
“Maintenance of endothelial glycocalyx integrity can be a therapeutic target to reduce AKI in this setting,” the researchers wrote.
The authors acknowledged that the study was done at a single center that had dialysis and death rates three and five times higher, respectively, than those of developed countries; and it measured syndecan-1 at only one time point almost immediately after the operation.
“Adding postoperative syndecan-1, even when using a clinical model that already incorporates variables from renal angina index, results in significant improvement in the capacity to predict severe AKI,” Dr. Cavalcante and colleagues concluded.
They had no financial relationships to disclose.
FROM THE JOURNAL OF THORACIC AND CARDIOVASCULAR SURGERY
Key clinical point: The biomarker syndecan-1 may aid in determining acute kidney injury risk for children having cardiac surgery.
Major finding: Children with elevated levels of syndecan-1 had a two- to ninefold greater risk of acute kidney injury.
Data source: Single-institution, prospective cohort study of 289 pediatric patients who had cardiac surgery from September 2013 to December 2014.
Disclosures: Dr. Cavalcante and coauthors had no financial relationships to disclose.
Making the case for CABG using bilateral thoracic arteries
Cardiac surgeons have been slow to embrace bilateral internal thoracic arteries (ITAs) for coronary artery bypass grafting (CABG) despite accumulating evidence that this technique achieves better long-term survival than the single-artery technique, perhaps because they think the bilateral technique is more difficult. However, investigators from Johns Hopkins University have found no difference in results between four different bilateral ITAs techniques regardless of complexity.
Their single-center study analyzed outcomes from 762 patients at Johns Hopkins who had CABG by way of one of four different bilateral ITA (BITA) techniques between 1997 and 2014. The results are in the July issue of the Journal of Thoracic and Cardiovascular Surgery (2016;152:120-7).
“We found no significant difference in terms of long-term survival or freedom from repeat revascularization between different configurations of BITA use,” wrote J. Trent Magruder, MD, and his colleagues.
Dr. Magruder and his coauthors cited 13 reports that found BITA achieved better graft patency and long-term survival than did single internal thoracic arteries, but they noted the lack of reports comparing different BITA techniques. “Given the paucity of comparative data on long-term outcomes of various BITA configurations, we sought to study differences in mortality and the need for repeat revascularization among patients receiving varying BITA graft configurations at our institution,” they said.
The four groups and types of BITA procedures they analyzed were:
LL/RL group, in situ left ITA (LITA) anastomosed to the left anterior descending artery (LAD) with in situ right ITA (RITA) anastomosed to the left coronary circulation (n = 239).
LL/RR group, in situ LITA-LAD and in situ RITA-right coronary circulation (n = 239 patients).
RL/LL group, in situ RITA-LAD with in situ LITA-left coronary circulation (n = 185 patients).
Y group, in situ LITA-LAD with a free RITA as a composite graft with inflow from the LITA or a saphenous vein graft (n = 99 patients).
BITA cases comprised 5.7% of all 14,502 CABG procedures Johns Hopkins cardiac surgeons performed through the study period (60 BITA cases were dropped from the analysis because of incomplete data). That rate is about in line with a previously reported use rate of 4% of CABG procedures in the United States (Circulation. 2009;120:935-40).
Among the reasons Dr. Magruder and his coauthors cited for the lack of uptake of BITA among cardiac surgeons are discrepancies in survival data, a perceived high risk of complications such as sternal surgical site infections in patients with diabetes or chronic obstructive pulmonary disease or in those who are obese, and increased operative time and risk of bleeding.
With regard to the operation itself, the mean cross-clamp and coronary bypass times and number of bypass grafts were similar among all four groups, the latter ranging from 3.0 for the RL/LL group to 3.4 for the Y group. However, the researchers did find appreciable differences in rates of transfusions during the operation and skeletonization of the RITA at harvest. The Y group had the highest rates for both – 57.6% had transfusions and 72.7% had skeletonized RITA at harvest – followed by the RL/LL with rates of 43.2% for transfusions and 31.4% with skeletonized RITA. Rates for both intraoperative transfusion and RITA skeletonization were 24.7% and 8%, respectively, in the LL/RL group; and 37.7% and 18%, respectively, in the LL/RR group.
In-hospital complications, including reintervention for bleeding, heart attack, stroke, inflammation of the mediastinum, and death, were similar among all four groups. There were no in-hospital heart attacks. The only statistically significant difference was in hospital stay, ranging from an average of 6.1 days for the LL/RL group to 7.4 for both the LL/RR and RL/LL groups.
Through the duration of follow-up, the overall rate for repeat percutaneous coronary intervention was 7.6% – highest among the RL/LL (9.2%) and Y groups (9%). Those in the LL/RR group had the highest rates of repeat CABG: 1.7% vs. 0.8% for LL/RL group, 0.5% for the RL/LL group and 0% for group Y. Rates of late cardiac death were around 5% for the first three groups, but none were reported in group Y.
Dr. Magruder and colleagues acknowledged their study used a limited sample size for each procedure, but that their findings show that cardiac surgeons should choose their BITA configuration based on individual patient factors. “In general, the technically simplest operations should be selected because more complex procedures offer no additional benefit,” they said.
Dr. Magruder and his coauthors had no financial disclosures.
The take-home message of the study by Dr. Magruder and colleagues is that using bilateral internal thoracic arteries (BITA) is more important than the specific configuration, Saswata Deb, MD, BSc, and Stephen E. Fremes, MD, MSc, BSc, of the University of Toronto wrote in their invited commentary (J Thorac Cardiovasc Surg. 2016;152:128-30). “In other words: BITA – just do it!” they wrote.
Because the survival advantage of BITA in CABG typically becomes apparent 10 years or more after the operation, the Johns Hopkins study, along with the Arterial Revascularization Trial that compares BITA with single ITA (Semin Thorac Cardiovasc Surg. 2014;26:76-94), can help redefine how cardiac surgeons select conduits for CABG, the commentators said.
“What does this particular study add?” they asked. “Point estimates for the adjusted hazard ratio of death or repeat revascularization were close to unity for each of the primary grafting hypothesis comparisons.”
Dr. Deb and Dr. Fremes had no financial relationships to disclose.
The take-home message of the study by Dr. Magruder and colleagues is that using bilateral internal thoracic arteries (BITA) is more important than the specific configuration, Saswata Deb, MD, BSc, and Stephen E. Fremes, MD, MSc, BSc, of the University of Toronto wrote in their invited commentary (J Thorac Cardiovasc Surg. 2016;152:128-30). “In other words: BITA – just do it!” they wrote.
Because the survival advantage of BITA in CABG typically becomes apparent 10 years or more after the operation, the Johns Hopkins study, along with the Arterial Revascularization Trial that compares BITA with single ITA (Semin Thorac Cardiovasc Surg. 2014;26:76-94), can help redefine how cardiac surgeons select conduits for CABG, the commentators said.
“What does this particular study add?” they asked. “Point estimates for the adjusted hazard ratio of death or repeat revascularization were close to unity for each of the primary grafting hypothesis comparisons.”
Dr. Deb and Dr. Fremes had no financial relationships to disclose.
The take-home message of the study by Dr. Magruder and colleagues is that using bilateral internal thoracic arteries (BITA) is more important than the specific configuration, Saswata Deb, MD, BSc, and Stephen E. Fremes, MD, MSc, BSc, of the University of Toronto wrote in their invited commentary (J Thorac Cardiovasc Surg. 2016;152:128-30). “In other words: BITA – just do it!” they wrote.
Because the survival advantage of BITA in CABG typically becomes apparent 10 years or more after the operation, the Johns Hopkins study, along with the Arterial Revascularization Trial that compares BITA with single ITA (Semin Thorac Cardiovasc Surg. 2014;26:76-94), can help redefine how cardiac surgeons select conduits for CABG, the commentators said.
“What does this particular study add?” they asked. “Point estimates for the adjusted hazard ratio of death or repeat revascularization were close to unity for each of the primary grafting hypothesis comparisons.”
Dr. Deb and Dr. Fremes had no financial relationships to disclose.
Cardiac surgeons have been slow to embrace bilateral internal thoracic arteries (ITAs) for coronary artery bypass grafting (CABG) despite accumulating evidence that this technique achieves better long-term survival than the single-artery technique, perhaps because they think the bilateral technique is more difficult. However, investigators from Johns Hopkins University have found no difference in results between four different bilateral ITAs techniques regardless of complexity.
Their single-center study analyzed outcomes from 762 patients at Johns Hopkins who had CABG by way of one of four different bilateral ITA (BITA) techniques between 1997 and 2014. The results are in the July issue of the Journal of Thoracic and Cardiovascular Surgery (2016;152:120-7).
“We found no significant difference in terms of long-term survival or freedom from repeat revascularization between different configurations of BITA use,” wrote J. Trent Magruder, MD, and his colleagues.
Dr. Magruder and his coauthors cited 13 reports that found BITA achieved better graft patency and long-term survival than did single internal thoracic arteries, but they noted the lack of reports comparing different BITA techniques. “Given the paucity of comparative data on long-term outcomes of various BITA configurations, we sought to study differences in mortality and the need for repeat revascularization among patients receiving varying BITA graft configurations at our institution,” they said.
The four groups and types of BITA procedures they analyzed were:
LL/RL group, in situ left ITA (LITA) anastomosed to the left anterior descending artery (LAD) with in situ right ITA (RITA) anastomosed to the left coronary circulation (n = 239).
LL/RR group, in situ LITA-LAD and in situ RITA-right coronary circulation (n = 239 patients).
RL/LL group, in situ RITA-LAD with in situ LITA-left coronary circulation (n = 185 patients).
Y group, in situ LITA-LAD with a free RITA as a composite graft with inflow from the LITA or a saphenous vein graft (n = 99 patients).
BITA cases comprised 5.7% of all 14,502 CABG procedures Johns Hopkins cardiac surgeons performed through the study period (60 BITA cases were dropped from the analysis because of incomplete data). That rate is about in line with a previously reported use rate of 4% of CABG procedures in the United States (Circulation. 2009;120:935-40).
Among the reasons Dr. Magruder and his coauthors cited for the lack of uptake of BITA among cardiac surgeons are discrepancies in survival data, a perceived high risk of complications such as sternal surgical site infections in patients with diabetes or chronic obstructive pulmonary disease or in those who are obese, and increased operative time and risk of bleeding.
With regard to the operation itself, the mean cross-clamp and coronary bypass times and number of bypass grafts were similar among all four groups, the latter ranging from 3.0 for the RL/LL group to 3.4 for the Y group. However, the researchers did find appreciable differences in rates of transfusions during the operation and skeletonization of the RITA at harvest. The Y group had the highest rates for both – 57.6% had transfusions and 72.7% had skeletonized RITA at harvest – followed by the RL/LL with rates of 43.2% for transfusions and 31.4% with skeletonized RITA. Rates for both intraoperative transfusion and RITA skeletonization were 24.7% and 8%, respectively, in the LL/RL group; and 37.7% and 18%, respectively, in the LL/RR group.
In-hospital complications, including reintervention for bleeding, heart attack, stroke, inflammation of the mediastinum, and death, were similar among all four groups. There were no in-hospital heart attacks. The only statistically significant difference was in hospital stay, ranging from an average of 6.1 days for the LL/RL group to 7.4 for both the LL/RR and RL/LL groups.
Through the duration of follow-up, the overall rate for repeat percutaneous coronary intervention was 7.6% – highest among the RL/LL (9.2%) and Y groups (9%). Those in the LL/RR group had the highest rates of repeat CABG: 1.7% vs. 0.8% for LL/RL group, 0.5% for the RL/LL group and 0% for group Y. Rates of late cardiac death were around 5% for the first three groups, but none were reported in group Y.
Dr. Magruder and colleagues acknowledged their study used a limited sample size for each procedure, but that their findings show that cardiac surgeons should choose their BITA configuration based on individual patient factors. “In general, the technically simplest operations should be selected because more complex procedures offer no additional benefit,” they said.
Dr. Magruder and his coauthors had no financial disclosures.
Cardiac surgeons have been slow to embrace bilateral internal thoracic arteries (ITAs) for coronary artery bypass grafting (CABG) despite accumulating evidence that this technique achieves better long-term survival than the single-artery technique, perhaps because they think the bilateral technique is more difficult. However, investigators from Johns Hopkins University have found no difference in results between four different bilateral ITAs techniques regardless of complexity.
Their single-center study analyzed outcomes from 762 patients at Johns Hopkins who had CABG by way of one of four different bilateral ITA (BITA) techniques between 1997 and 2014. The results are in the July issue of the Journal of Thoracic and Cardiovascular Surgery (2016;152:120-7).
“We found no significant difference in terms of long-term survival or freedom from repeat revascularization between different configurations of BITA use,” wrote J. Trent Magruder, MD, and his colleagues.
Dr. Magruder and his coauthors cited 13 reports that found BITA achieved better graft patency and long-term survival than did single internal thoracic arteries, but they noted the lack of reports comparing different BITA techniques. “Given the paucity of comparative data on long-term outcomes of various BITA configurations, we sought to study differences in mortality and the need for repeat revascularization among patients receiving varying BITA graft configurations at our institution,” they said.
The four groups and types of BITA procedures they analyzed were:
LL/RL group, in situ left ITA (LITA) anastomosed to the left anterior descending artery (LAD) with in situ right ITA (RITA) anastomosed to the left coronary circulation (n = 239).
LL/RR group, in situ LITA-LAD and in situ RITA-right coronary circulation (n = 239 patients).
RL/LL group, in situ RITA-LAD with in situ LITA-left coronary circulation (n = 185 patients).
Y group, in situ LITA-LAD with a free RITA as a composite graft with inflow from the LITA or a saphenous vein graft (n = 99 patients).
BITA cases comprised 5.7% of all 14,502 CABG procedures Johns Hopkins cardiac surgeons performed through the study period (60 BITA cases were dropped from the analysis because of incomplete data). That rate is about in line with a previously reported use rate of 4% of CABG procedures in the United States (Circulation. 2009;120:935-40).
Among the reasons Dr. Magruder and his coauthors cited for the lack of uptake of BITA among cardiac surgeons are discrepancies in survival data, a perceived high risk of complications such as sternal surgical site infections in patients with diabetes or chronic obstructive pulmonary disease or in those who are obese, and increased operative time and risk of bleeding.
With regard to the operation itself, the mean cross-clamp and coronary bypass times and number of bypass grafts were similar among all four groups, the latter ranging from 3.0 for the RL/LL group to 3.4 for the Y group. However, the researchers did find appreciable differences in rates of transfusions during the operation and skeletonization of the RITA at harvest. The Y group had the highest rates for both – 57.6% had transfusions and 72.7% had skeletonized RITA at harvest – followed by the RL/LL with rates of 43.2% for transfusions and 31.4% with skeletonized RITA. Rates for both intraoperative transfusion and RITA skeletonization were 24.7% and 8%, respectively, in the LL/RL group; and 37.7% and 18%, respectively, in the LL/RR group.
In-hospital complications, including reintervention for bleeding, heart attack, stroke, inflammation of the mediastinum, and death, were similar among all four groups. There were no in-hospital heart attacks. The only statistically significant difference was in hospital stay, ranging from an average of 6.1 days for the LL/RL group to 7.4 for both the LL/RR and RL/LL groups.
Through the duration of follow-up, the overall rate for repeat percutaneous coronary intervention was 7.6% – highest among the RL/LL (9.2%) and Y groups (9%). Those in the LL/RR group had the highest rates of repeat CABG: 1.7% vs. 0.8% for LL/RL group, 0.5% for the RL/LL group and 0% for group Y. Rates of late cardiac death were around 5% for the first three groups, but none were reported in group Y.
Dr. Magruder and colleagues acknowledged their study used a limited sample size for each procedure, but that their findings show that cardiac surgeons should choose their BITA configuration based on individual patient factors. “In general, the technically simplest operations should be selected because more complex procedures offer no additional benefit,” they said.
Dr. Magruder and his coauthors had no financial disclosures.
FROM THE JOURNAL OF THORACIC AND CARDIOVASCULAR SURGERY
Key clinical point: No specific technique for bilateral internal thoracic artery (BITA) graft in coronary artery bypass grafting (CABG) has an advantage over other techniques.
Major finding: Among four different BITA configurations, no in-hospital deaths occurred and the late cardiac death rate was around 5% for three of the four groups and 0% for the fourth group.
Data source: Single-center, retrospective study of 762 patients who had CABG with use of BITA from 1997 to 2014.
Disclosures: Dr. Magruder and colleagues reported having no financial disclosures.
Four-branched arch replacement gets acceptable results
NEW YORK – A total aortic arch replacement approach that uses a four-branched graft with antegrade cerebral perfusion can be done with low rates of in-hospital death and complications, a large series from two institutions in Japan showed.
Kenji Minatoya, MD, of the National Cerebral and Cardiovascular Center in Osaka, Japan, reported that his institution’s approach for total arch replacement (TAR) had an in-hospital death rate of 5.2%.
Dr. Minatoya and his colleagues started using four-branch TAR in the 1980s, switching from retrograde to antegrade cerebral perfusion to protect the brain later on. “The study purpose was to investigate the results of total arch replacement using the four-branch graft as a benchmark in the endovascular era,” he said at the meeting, sponsored by the American Association for Thoracic Surgery.
The study involved 1,005 cases of total arch replacement performed at Dr. Minatoya’s center and at Tokyo Medical University from 2001 to 2016.
The study population included a cohort of 152 people in their 80s. The in-hospital death rate in this group was 11.8%, Dr. Minatoya said. The over-80 group mostly underwent thoracic endovascular aortic repair (TEVAR) beginning in 2008, he said, but in recent years some had open total arch replacement operations.
The univariate analysis showed that chronic kidney disease, long operation times, long durations for coronary bypass and circulatory arrest, and extended time on mechanical ventilation were risk factors for in-hospital death in octogenarians, Dr. Minatoya said. The multivariate analysis showed that male gender along with extended mechanical ventilation were risk factors for in-hospital death in this group, he said.
The overall population included 252 emergent operations, 224 of which were for acute aortic dissections, Dr. Minatoya said. The in-hospital death rate was 4.5% for elective operations and 7.1% for emergent cases, he said. The death rate for isolated, elective total arch replacement was 3.4%.
Focusing on acute aortic dissections, Dr. Minatoya said, “We have adopted an aggressive strategy for entry-site resection, including total arch replacement, in patients with arch tears.” Almost 50% of patients with acute aortic dissection had total arch replacement, he said, with identical 4.9% rates for in-hospital mortality rate and permanent neurological deficit in this group.
The leading overall causes of in-hospital death were low-output syndrome (38.5%), sepsis (25%), respiratory failure (21%) and rupture of the residual aneurysm (9%), Dr. Minatoya said.
Fifteen patients (1.5%) underwent second operations for arch grafts, he said: 11 for pseudoaneurysm; three for hemolysis and one for infection. Other overall measures in the analysis were a permanent neurological dysfunction rate of 3.6%, a temporary neurological dysfunction rate of 6.4%, and no spinal cord complications. Overall 5-year survival was 80.7% and 10-year survival was 63.1%, Dr. Minatoya said.
A total of 311 patients had concomitant procedures. They included aortic valve operations (64); aortic root replacement (38); mitral valve replacement (13); and coronary artery bypass grafting (196).
The typical operation in the study population took about 8 hours, Dr. Minatoya said (482 minutes). Timing of key operative steps were cardiopulmonary time of 254 minutes, cardiac arrest time of 146 minutes, antegrade cerebral perfusion time of 160 minutes and lower-body circulatory arrest time of 62 minutes.
“Since the mean age was 70 years old, we think the survival rate was acceptable,” Dr. Minatoya said, regarding overall study results. Overall risk factors for in-hospital death were short stature, long pump time, chronic kidney disease, and age of 80 and up, he said. Short stature was a risk factor for permanent neurological deficit, and males over age 80 had a higher risk for total arch replacement.
“Total arch replacement using the four-branched graft with antegrade cerebral perfusion could be accomplished with acceptable early and late results,” Dr. Minatoya said. “The branched-arch TEVAR may be a good option for octogenarians and patients with chronic kidney disease.”
Dr. Minatoya had no financial relationships to disclose.
NEW YORK – A total aortic arch replacement approach that uses a four-branched graft with antegrade cerebral perfusion can be done with low rates of in-hospital death and complications, a large series from two institutions in Japan showed.
Kenji Minatoya, MD, of the National Cerebral and Cardiovascular Center in Osaka, Japan, reported that his institution’s approach for total arch replacement (TAR) had an in-hospital death rate of 5.2%.
Dr. Minatoya and his colleagues started using four-branch TAR in the 1980s, switching from retrograde to antegrade cerebral perfusion to protect the brain later on. “The study purpose was to investigate the results of total arch replacement using the four-branch graft as a benchmark in the endovascular era,” he said at the meeting, sponsored by the American Association for Thoracic Surgery.
The study involved 1,005 cases of total arch replacement performed at Dr. Minatoya’s center and at Tokyo Medical University from 2001 to 2016.
The study population included a cohort of 152 people in their 80s. The in-hospital death rate in this group was 11.8%, Dr. Minatoya said. The over-80 group mostly underwent thoracic endovascular aortic repair (TEVAR) beginning in 2008, he said, but in recent years some had open total arch replacement operations.
The univariate analysis showed that chronic kidney disease, long operation times, long durations for coronary bypass and circulatory arrest, and extended time on mechanical ventilation were risk factors for in-hospital death in octogenarians, Dr. Minatoya said. The multivariate analysis showed that male gender along with extended mechanical ventilation were risk factors for in-hospital death in this group, he said.
The overall population included 252 emergent operations, 224 of which were for acute aortic dissections, Dr. Minatoya said. The in-hospital death rate was 4.5% for elective operations and 7.1% for emergent cases, he said. The death rate for isolated, elective total arch replacement was 3.4%.
Focusing on acute aortic dissections, Dr. Minatoya said, “We have adopted an aggressive strategy for entry-site resection, including total arch replacement, in patients with arch tears.” Almost 50% of patients with acute aortic dissection had total arch replacement, he said, with identical 4.9% rates for in-hospital mortality rate and permanent neurological deficit in this group.
The leading overall causes of in-hospital death were low-output syndrome (38.5%), sepsis (25%), respiratory failure (21%) and rupture of the residual aneurysm (9%), Dr. Minatoya said.
Fifteen patients (1.5%) underwent second operations for arch grafts, he said: 11 for pseudoaneurysm; three for hemolysis and one for infection. Other overall measures in the analysis were a permanent neurological dysfunction rate of 3.6%, a temporary neurological dysfunction rate of 6.4%, and no spinal cord complications. Overall 5-year survival was 80.7% and 10-year survival was 63.1%, Dr. Minatoya said.
A total of 311 patients had concomitant procedures. They included aortic valve operations (64); aortic root replacement (38); mitral valve replacement (13); and coronary artery bypass grafting (196).
The typical operation in the study population took about 8 hours, Dr. Minatoya said (482 minutes). Timing of key operative steps were cardiopulmonary time of 254 minutes, cardiac arrest time of 146 minutes, antegrade cerebral perfusion time of 160 minutes and lower-body circulatory arrest time of 62 minutes.
“Since the mean age was 70 years old, we think the survival rate was acceptable,” Dr. Minatoya said, regarding overall study results. Overall risk factors for in-hospital death were short stature, long pump time, chronic kidney disease, and age of 80 and up, he said. Short stature was a risk factor for permanent neurological deficit, and males over age 80 had a higher risk for total arch replacement.
“Total arch replacement using the four-branched graft with antegrade cerebral perfusion could be accomplished with acceptable early and late results,” Dr. Minatoya said. “The branched-arch TEVAR may be a good option for octogenarians and patients with chronic kidney disease.”
Dr. Minatoya had no financial relationships to disclose.
NEW YORK – A total aortic arch replacement approach that uses a four-branched graft with antegrade cerebral perfusion can be done with low rates of in-hospital death and complications, a large series from two institutions in Japan showed.
Kenji Minatoya, MD, of the National Cerebral and Cardiovascular Center in Osaka, Japan, reported that his institution’s approach for total arch replacement (TAR) had an in-hospital death rate of 5.2%.
Dr. Minatoya and his colleagues started using four-branch TAR in the 1980s, switching from retrograde to antegrade cerebral perfusion to protect the brain later on. “The study purpose was to investigate the results of total arch replacement using the four-branch graft as a benchmark in the endovascular era,” he said at the meeting, sponsored by the American Association for Thoracic Surgery.
The study involved 1,005 cases of total arch replacement performed at Dr. Minatoya’s center and at Tokyo Medical University from 2001 to 2016.
The study population included a cohort of 152 people in their 80s. The in-hospital death rate in this group was 11.8%, Dr. Minatoya said. The over-80 group mostly underwent thoracic endovascular aortic repair (TEVAR) beginning in 2008, he said, but in recent years some had open total arch replacement operations.
The univariate analysis showed that chronic kidney disease, long operation times, long durations for coronary bypass and circulatory arrest, and extended time on mechanical ventilation were risk factors for in-hospital death in octogenarians, Dr. Minatoya said. The multivariate analysis showed that male gender along with extended mechanical ventilation were risk factors for in-hospital death in this group, he said.
The overall population included 252 emergent operations, 224 of which were for acute aortic dissections, Dr. Minatoya said. The in-hospital death rate was 4.5% for elective operations and 7.1% for emergent cases, he said. The death rate for isolated, elective total arch replacement was 3.4%.
Focusing on acute aortic dissections, Dr. Minatoya said, “We have adopted an aggressive strategy for entry-site resection, including total arch replacement, in patients with arch tears.” Almost 50% of patients with acute aortic dissection had total arch replacement, he said, with identical 4.9% rates for in-hospital mortality rate and permanent neurological deficit in this group.
The leading overall causes of in-hospital death were low-output syndrome (38.5%), sepsis (25%), respiratory failure (21%) and rupture of the residual aneurysm (9%), Dr. Minatoya said.
Fifteen patients (1.5%) underwent second operations for arch grafts, he said: 11 for pseudoaneurysm; three for hemolysis and one for infection. Other overall measures in the analysis were a permanent neurological dysfunction rate of 3.6%, a temporary neurological dysfunction rate of 6.4%, and no spinal cord complications. Overall 5-year survival was 80.7% and 10-year survival was 63.1%, Dr. Minatoya said.
A total of 311 patients had concomitant procedures. They included aortic valve operations (64); aortic root replacement (38); mitral valve replacement (13); and coronary artery bypass grafting (196).
The typical operation in the study population took about 8 hours, Dr. Minatoya said (482 minutes). Timing of key operative steps were cardiopulmonary time of 254 minutes, cardiac arrest time of 146 minutes, antegrade cerebral perfusion time of 160 minutes and lower-body circulatory arrest time of 62 minutes.
“Since the mean age was 70 years old, we think the survival rate was acceptable,” Dr. Minatoya said, regarding overall study results. Overall risk factors for in-hospital death were short stature, long pump time, chronic kidney disease, and age of 80 and up, he said. Short stature was a risk factor for permanent neurological deficit, and males over age 80 had a higher risk for total arch replacement.
“Total arch replacement using the four-branched graft with antegrade cerebral perfusion could be accomplished with acceptable early and late results,” Dr. Minatoya said. “The branched-arch TEVAR may be a good option for octogenarians and patients with chronic kidney disease.”
Dr. Minatoya had no financial relationships to disclose.
AT THE AATS AORTIC SYMPOSIUM 2016
Key clinical point: Four-branched total aortic arch replacement can achieve acceptable early and late results.
Major finding: Overall in-hospital mortality was 5.2% and 5-year survival was 80.7%.
Data source: Consecutive series of 1,005 patients who had total arch replacement between 2001 and 2016 at two centers in Japan.
Disclosures: Dr. Minatoya reported having no financial disclosures.
Pediatric autologous aortic repair built to last
NEW YORK – With more than 1 million adults living today with congenital aortic disease, cardiovascular surgeons must think of outcomes in terms of decades, not years, when performing aortic arch repair in newborns, infants, and children, according to Charles D. Fraser Jr., M.D.
To that end, an all-autologous approach to aortic arch repair is key in preserving problem-free aortic function in adulthood, said Dr. Fraser, surgeon-in-chief at Texas Children’s Hospital in Houston.
Dr. Fraser reported on his center’s experience with all-autologous aortic arch repair techniques. He reviewed the following five principles that guide aortic arch repair in newborns, infants, and children at Texas Children’s Hospital:
• Use of autologous tissue reconstruction and avoidance of prosthetic material.
• Concomitant intracardiac repair.
• Use of anatomic reconstruction.
• Optimization of ventriculoarterial coupling.
• Preservation of laryngeal nerve function.
“The principles we developed at Texas Children’s Hospital we hope will translate into fewer of these patients that surgeons caring for adults with aortic disease will have to take care of later in life,” Dr. Fraser said at the meeting sponsored by the American Association for Thoracic Surgery. He reviewed cases in which he explained techniques he and his colleagues developed to address long-term outcomes.
The first challenge is to determine when to perform aortic repair in pediatric patients. “A question often asked is how small is too small when assessing the aortic arch in association with significant periductal coarctation?” he said. “Our rule of thumb has been that the arch diameter measured in millimeters should be at least the patient’s weight in kilograms plus one.” In other words, a 3-kg baby should have an aortic arch of at least 4 mm in diameter, he said.
He described the case of a 3.8-kg male baby on prostaglandin E1 who had aortic arch advancement repair and closure of atrial and ventricular septal defects at 8 days of age. The patient had an early origin of the left common carotid artery and a small proximal aortic arch. “This is the kind of patient in which we would do a complete aortic arch reconstruction, again with the autologous technique,” Dr. Fraser said.
In such a patient, Dr. Fraser and his colleagues at Texas Children’s Hospital support the circulation to the brain with antegrade cerebral perfusion, using transcranial Doppler and near-infrared spectroscopy to guide their profusion strategy, before putting the child on cardiac bypass and “profound” hypothermia. Careful planning before cannulation is important to perform the aortic transection at the correct level, he said
He also explained the ascending sliding arch aortoplasty, also known as the “Texas slide,” first described by E. Dean McKenzie, M.D., at Texas Children’s Hospital in 2011 (Ann. Thorac. Surg. 2011;91:805-10) This technique involves sliding a tongue-shaped piece of the ascending aorta underneath the aortic arch to construct an all-autologous repair.
“In patients with bicuspid aortic valves, we often observe that the ascending aorta is extremely elongated,” he said. “The idea is to take advantage of that and slide the ascending aorta completely up underneath the aortic arch and construct an all-autologous arch advancement type of repair.”
He presented the case of a 4-year-old boy with coarctation of the aorta in whom the Texas slide was indicated. “If this patient were treated with a simple coarctectomy, the patient would be subject to a life with a moderately hypoplastic aortic arch, and over the course of time, this could be problematic,” Dr. Fraser said. “The sliding reconstruction has relevance not only to the status of the aortic arch over the long term but it also has a profound effect on ventricular function.”
He noted a single-center, retrospective study from the United Kingdom that demonstrated that the quality of the aortic arch reconstruction, and the related opportunity for ventricular arterial coupling, directly correlate with long-term performance of the aortic arch in patients with hypoplastic left heart syndrome (J Thorac Cardiovasc Surg. 2014;148:1526-33).
“This is very important as part of the growing population of these patients who need long-term management, most of whom we’re anticipating managing not just for years, but for decades,” Dr. Fraser said.
He said he had no relevant financial disclosures.
NEW YORK – With more than 1 million adults living today with congenital aortic disease, cardiovascular surgeons must think of outcomes in terms of decades, not years, when performing aortic arch repair in newborns, infants, and children, according to Charles D. Fraser Jr., M.D.
To that end, an all-autologous approach to aortic arch repair is key in preserving problem-free aortic function in adulthood, said Dr. Fraser, surgeon-in-chief at Texas Children’s Hospital in Houston.
Dr. Fraser reported on his center’s experience with all-autologous aortic arch repair techniques. He reviewed the following five principles that guide aortic arch repair in newborns, infants, and children at Texas Children’s Hospital:
• Use of autologous tissue reconstruction and avoidance of prosthetic material.
• Concomitant intracardiac repair.
• Use of anatomic reconstruction.
• Optimization of ventriculoarterial coupling.
• Preservation of laryngeal nerve function.
“The principles we developed at Texas Children’s Hospital we hope will translate into fewer of these patients that surgeons caring for adults with aortic disease will have to take care of later in life,” Dr. Fraser said at the meeting sponsored by the American Association for Thoracic Surgery. He reviewed cases in which he explained techniques he and his colleagues developed to address long-term outcomes.
The first challenge is to determine when to perform aortic repair in pediatric patients. “A question often asked is how small is too small when assessing the aortic arch in association with significant periductal coarctation?” he said. “Our rule of thumb has been that the arch diameter measured in millimeters should be at least the patient’s weight in kilograms plus one.” In other words, a 3-kg baby should have an aortic arch of at least 4 mm in diameter, he said.
He described the case of a 3.8-kg male baby on prostaglandin E1 who had aortic arch advancement repair and closure of atrial and ventricular septal defects at 8 days of age. The patient had an early origin of the left common carotid artery and a small proximal aortic arch. “This is the kind of patient in which we would do a complete aortic arch reconstruction, again with the autologous technique,” Dr. Fraser said.
In such a patient, Dr. Fraser and his colleagues at Texas Children’s Hospital support the circulation to the brain with antegrade cerebral perfusion, using transcranial Doppler and near-infrared spectroscopy to guide their profusion strategy, before putting the child on cardiac bypass and “profound” hypothermia. Careful planning before cannulation is important to perform the aortic transection at the correct level, he said
He also explained the ascending sliding arch aortoplasty, also known as the “Texas slide,” first described by E. Dean McKenzie, M.D., at Texas Children’s Hospital in 2011 (Ann. Thorac. Surg. 2011;91:805-10) This technique involves sliding a tongue-shaped piece of the ascending aorta underneath the aortic arch to construct an all-autologous repair.
“In patients with bicuspid aortic valves, we often observe that the ascending aorta is extremely elongated,” he said. “The idea is to take advantage of that and slide the ascending aorta completely up underneath the aortic arch and construct an all-autologous arch advancement type of repair.”
He presented the case of a 4-year-old boy with coarctation of the aorta in whom the Texas slide was indicated. “If this patient were treated with a simple coarctectomy, the patient would be subject to a life with a moderately hypoplastic aortic arch, and over the course of time, this could be problematic,” Dr. Fraser said. “The sliding reconstruction has relevance not only to the status of the aortic arch over the long term but it also has a profound effect on ventricular function.”
He noted a single-center, retrospective study from the United Kingdom that demonstrated that the quality of the aortic arch reconstruction, and the related opportunity for ventricular arterial coupling, directly correlate with long-term performance of the aortic arch in patients with hypoplastic left heart syndrome (J Thorac Cardiovasc Surg. 2014;148:1526-33).
“This is very important as part of the growing population of these patients who need long-term management, most of whom we’re anticipating managing not just for years, but for decades,” Dr. Fraser said.
He said he had no relevant financial disclosures.
NEW YORK – With more than 1 million adults living today with congenital aortic disease, cardiovascular surgeons must think of outcomes in terms of decades, not years, when performing aortic arch repair in newborns, infants, and children, according to Charles D. Fraser Jr., M.D.
To that end, an all-autologous approach to aortic arch repair is key in preserving problem-free aortic function in adulthood, said Dr. Fraser, surgeon-in-chief at Texas Children’s Hospital in Houston.
Dr. Fraser reported on his center’s experience with all-autologous aortic arch repair techniques. He reviewed the following five principles that guide aortic arch repair in newborns, infants, and children at Texas Children’s Hospital:
• Use of autologous tissue reconstruction and avoidance of prosthetic material.
• Concomitant intracardiac repair.
• Use of anatomic reconstruction.
• Optimization of ventriculoarterial coupling.
• Preservation of laryngeal nerve function.
“The principles we developed at Texas Children’s Hospital we hope will translate into fewer of these patients that surgeons caring for adults with aortic disease will have to take care of later in life,” Dr. Fraser said at the meeting sponsored by the American Association for Thoracic Surgery. He reviewed cases in which he explained techniques he and his colleagues developed to address long-term outcomes.
The first challenge is to determine when to perform aortic repair in pediatric patients. “A question often asked is how small is too small when assessing the aortic arch in association with significant periductal coarctation?” he said. “Our rule of thumb has been that the arch diameter measured in millimeters should be at least the patient’s weight in kilograms plus one.” In other words, a 3-kg baby should have an aortic arch of at least 4 mm in diameter, he said.
He described the case of a 3.8-kg male baby on prostaglandin E1 who had aortic arch advancement repair and closure of atrial and ventricular septal defects at 8 days of age. The patient had an early origin of the left common carotid artery and a small proximal aortic arch. “This is the kind of patient in which we would do a complete aortic arch reconstruction, again with the autologous technique,” Dr. Fraser said.
In such a patient, Dr. Fraser and his colleagues at Texas Children’s Hospital support the circulation to the brain with antegrade cerebral perfusion, using transcranial Doppler and near-infrared spectroscopy to guide their profusion strategy, before putting the child on cardiac bypass and “profound” hypothermia. Careful planning before cannulation is important to perform the aortic transection at the correct level, he said
He also explained the ascending sliding arch aortoplasty, also known as the “Texas slide,” first described by E. Dean McKenzie, M.D., at Texas Children’s Hospital in 2011 (Ann. Thorac. Surg. 2011;91:805-10) This technique involves sliding a tongue-shaped piece of the ascending aorta underneath the aortic arch to construct an all-autologous repair.
“In patients with bicuspid aortic valves, we often observe that the ascending aorta is extremely elongated,” he said. “The idea is to take advantage of that and slide the ascending aorta completely up underneath the aortic arch and construct an all-autologous arch advancement type of repair.”
He presented the case of a 4-year-old boy with coarctation of the aorta in whom the Texas slide was indicated. “If this patient were treated with a simple coarctectomy, the patient would be subject to a life with a moderately hypoplastic aortic arch, and over the course of time, this could be problematic,” Dr. Fraser said. “The sliding reconstruction has relevance not only to the status of the aortic arch over the long term but it also has a profound effect on ventricular function.”
He noted a single-center, retrospective study from the United Kingdom that demonstrated that the quality of the aortic arch reconstruction, and the related opportunity for ventricular arterial coupling, directly correlate with long-term performance of the aortic arch in patients with hypoplastic left heart syndrome (J Thorac Cardiovasc Surg. 2014;148:1526-33).
“This is very important as part of the growing population of these patients who need long-term management, most of whom we’re anticipating managing not just for years, but for decades,” Dr. Fraser said.
He said he had no relevant financial disclosures.
AT AATS AORTIC SYMPOSIUM 2016
Key clinical point: Surgeons must think of outcomes for operations to correct aortic arch disease in children in the context of decades, not years.
Major finding: Five principles should guide autologous aortic arch repair in newborns, infants, and children.
Data source: Case studies from Texas Children’s Hospital.
Disclosures: Dr. Fraser reported having no relevant financial disclosures.