User login
Official Newspaper of the American College of Surgeons
FRAIL scale found to predict 1-year functional status of geriatric trauma patients
WAIKOLOA, HAWAII – The FRAIL scale questionnaire predicts functional status and mortality at 1 year among geriatric trauma patients and is a useful tool for bedside screening by clinicians, results from a single-center study demonstrated.
“Over the past 2 years, the implications of frailty among the geriatric trauma population have gained much attention in the trauma community,” Cathy A. Maxwell, PhD, RN, said in an interview in advance of the annual meeting of the American Association for the Surgery of Trauma. “This work highlights the clinical utility of the FRAIL scale for screening injured older patients who are admitted to trauma centers and other acute care hospitals. Hopefully, it will encourage trauma care providers to use the instrument to identify older patients’ pre-injury/baseline status and to obtain a frailty risk adjustment measure for quality improvement efforts.”
Developed by the International Association of Nutrition and Aging, the validated five-item FRAIL scale requires answers to questions about fatigue, resistance, ambulation, illnesses, and loss of weight (J Nutr Health Aging. 2012;16[7]:601-8). In an effort to examine the influence of pre-injury physical frailty (as measured by FRAIL) on 1-year outcomes, Dr. Maxwell, of the Vanderbilt University, Nashville, Tenn., and her associates evaluated injured patients aged 65 and older who were admitted through the ED between October 2013 and March 2014 and who participated in a prior study (J Trauma Acute Care Surg. 2016;80[2]:195-203). The researchers identified the five items of the FRAIL instrument from that study and created a pre-injury FRAIL score for each patient.
Dr. Maxwell reported results from 188 patients with a median age of 77, a median Injury Severity Score of 10, and a median comorbidity index of 3. Upon admission to the ED, 63 patients (34%) screened as frail (defined as a FRAIL score of 3 or greater), 71 (38%) screened as pre-frail (defined as a FRAIL score of 1-2), and 54 (29%) screened as non-frail (defined as a FRAIL score of zero). Frequencies for components of the FRAIL score were as follows: fatigue (65%), resistance (32%), ambulation (40%), illnesses (27%), and loss of weight (6%).
After the researchers controlled for age, comorbidities, injury severity, and cognitive status via the Ascertain Dementia 8-item Informant Questionnaire (AD8), they found that pre-injury FRAIL scores explained about 13% of the variability in physical function as measured by the Barthel Index (P less than .001). A total of 47 patients (26%) died within 1 year of admission. Logistic regression analysis revealed that after adjustment for these same variables, the higher the pre-injury FRAIL score, the greater the likelihood of mortality within 1 year (odds ratio, 1.74; P = .001).
“The FRAIL scale predicts functional decline and mortality in geriatric trauma patients and is a useful tool for clinicians,” Dr. Maxwell concluded. “Bedside nurses in our trauma unit at Vanderbilt University Medical Center are currently using this instrument to screen our older patients. We have seen an increase in earlier geriatric palliative care consultations as a result of our screening efforts.”
She acknowledged certain limitations of the study, including the fact that it was a secondary analysis. “We created FRAIL scale scores for 188 patients from six different data sources, thus, the created scores may not accurately represent actual prospectively collected FRAIL scores,” Dr. Maxwell said. “That being said, we compared the frailty frequencies from this study with actual FRAIL scale scores (from current bedside FRAIL screens) and we are seeing similar percentages of patients in non-frail, pre-frail and frail categories. This strengthens the findings of this study.”
She reported having no financial disclosures.
WAIKOLOA, HAWAII – The FRAIL scale questionnaire predicts functional status and mortality at 1 year among geriatric trauma patients and is a useful tool for bedside screening by clinicians, results from a single-center study demonstrated.
“Over the past 2 years, the implications of frailty among the geriatric trauma population have gained much attention in the trauma community,” Cathy A. Maxwell, PhD, RN, said in an interview in advance of the annual meeting of the American Association for the Surgery of Trauma. “This work highlights the clinical utility of the FRAIL scale for screening injured older patients who are admitted to trauma centers and other acute care hospitals. Hopefully, it will encourage trauma care providers to use the instrument to identify older patients’ pre-injury/baseline status and to obtain a frailty risk adjustment measure for quality improvement efforts.”
Developed by the International Association of Nutrition and Aging, the validated five-item FRAIL scale requires answers to questions about fatigue, resistance, ambulation, illnesses, and loss of weight (J Nutr Health Aging. 2012;16[7]:601-8). In an effort to examine the influence of pre-injury physical frailty (as measured by FRAIL) on 1-year outcomes, Dr. Maxwell, of the Vanderbilt University, Nashville, Tenn., and her associates evaluated injured patients aged 65 and older who were admitted through the ED between October 2013 and March 2014 and who participated in a prior study (J Trauma Acute Care Surg. 2016;80[2]:195-203). The researchers identified the five items of the FRAIL instrument from that study and created a pre-injury FRAIL score for each patient.
Dr. Maxwell reported results from 188 patients with a median age of 77, a median Injury Severity Score of 10, and a median comorbidity index of 3. Upon admission to the ED, 63 patients (34%) screened as frail (defined as a FRAIL score of 3 or greater), 71 (38%) screened as pre-frail (defined as a FRAIL score of 1-2), and 54 (29%) screened as non-frail (defined as a FRAIL score of zero). Frequencies for components of the FRAIL score were as follows: fatigue (65%), resistance (32%), ambulation (40%), illnesses (27%), and loss of weight (6%).
After the researchers controlled for age, comorbidities, injury severity, and cognitive status via the Ascertain Dementia 8-item Informant Questionnaire (AD8), they found that pre-injury FRAIL scores explained about 13% of the variability in physical function as measured by the Barthel Index (P less than .001). A total of 47 patients (26%) died within 1 year of admission. Logistic regression analysis revealed that after adjustment for these same variables, the higher the pre-injury FRAIL score, the greater the likelihood of mortality within 1 year (odds ratio, 1.74; P = .001).
“The FRAIL scale predicts functional decline and mortality in geriatric trauma patients and is a useful tool for clinicians,” Dr. Maxwell concluded. “Bedside nurses in our trauma unit at Vanderbilt University Medical Center are currently using this instrument to screen our older patients. We have seen an increase in earlier geriatric palliative care consultations as a result of our screening efforts.”
She acknowledged certain limitations of the study, including the fact that it was a secondary analysis. “We created FRAIL scale scores for 188 patients from six different data sources, thus, the created scores may not accurately represent actual prospectively collected FRAIL scores,” Dr. Maxwell said. “That being said, we compared the frailty frequencies from this study with actual FRAIL scale scores (from current bedside FRAIL screens) and we are seeing similar percentages of patients in non-frail, pre-frail and frail categories. This strengthens the findings of this study.”
She reported having no financial disclosures.
WAIKOLOA, HAWAII – The FRAIL scale questionnaire predicts functional status and mortality at 1 year among geriatric trauma patients and is a useful tool for bedside screening by clinicians, results from a single-center study demonstrated.
“Over the past 2 years, the implications of frailty among the geriatric trauma population have gained much attention in the trauma community,” Cathy A. Maxwell, PhD, RN, said in an interview in advance of the annual meeting of the American Association for the Surgery of Trauma. “This work highlights the clinical utility of the FRAIL scale for screening injured older patients who are admitted to trauma centers and other acute care hospitals. Hopefully, it will encourage trauma care providers to use the instrument to identify older patients’ pre-injury/baseline status and to obtain a frailty risk adjustment measure for quality improvement efforts.”
Developed by the International Association of Nutrition and Aging, the validated five-item FRAIL scale requires answers to questions about fatigue, resistance, ambulation, illnesses, and loss of weight (J Nutr Health Aging. 2012;16[7]:601-8). In an effort to examine the influence of pre-injury physical frailty (as measured by FRAIL) on 1-year outcomes, Dr. Maxwell, of the Vanderbilt University, Nashville, Tenn., and her associates evaluated injured patients aged 65 and older who were admitted through the ED between October 2013 and March 2014 and who participated in a prior study (J Trauma Acute Care Surg. 2016;80[2]:195-203). The researchers identified the five items of the FRAIL instrument from that study and created a pre-injury FRAIL score for each patient.
Dr. Maxwell reported results from 188 patients with a median age of 77, a median Injury Severity Score of 10, and a median comorbidity index of 3. Upon admission to the ED, 63 patients (34%) screened as frail (defined as a FRAIL score of 3 or greater), 71 (38%) screened as pre-frail (defined as a FRAIL score of 1-2), and 54 (29%) screened as non-frail (defined as a FRAIL score of zero). Frequencies for components of the FRAIL score were as follows: fatigue (65%), resistance (32%), ambulation (40%), illnesses (27%), and loss of weight (6%).
After the researchers controlled for age, comorbidities, injury severity, and cognitive status via the Ascertain Dementia 8-item Informant Questionnaire (AD8), they found that pre-injury FRAIL scores explained about 13% of the variability in physical function as measured by the Barthel Index (P less than .001). A total of 47 patients (26%) died within 1 year of admission. Logistic regression analysis revealed that after adjustment for these same variables, the higher the pre-injury FRAIL score, the greater the likelihood of mortality within 1 year (odds ratio, 1.74; P = .001).
“The FRAIL scale predicts functional decline and mortality in geriatric trauma patients and is a useful tool for clinicians,” Dr. Maxwell concluded. “Bedside nurses in our trauma unit at Vanderbilt University Medical Center are currently using this instrument to screen our older patients. We have seen an increase in earlier geriatric palliative care consultations as a result of our screening efforts.”
She acknowledged certain limitations of the study, including the fact that it was a secondary analysis. “We created FRAIL scale scores for 188 patients from six different data sources, thus, the created scores may not accurately represent actual prospectively collected FRAIL scores,” Dr. Maxwell said. “That being said, we compared the frailty frequencies from this study with actual FRAIL scale scores (from current bedside FRAIL screens) and we are seeing similar percentages of patients in non-frail, pre-frail and frail categories. This strengthens the findings of this study.”
She reported having no financial disclosures.
AT THE AAST ANNUAL MEETING
Key clinical point: The FRAIL scale is a useful tool for bedside screening of geriatric trauma patients.
Major finding: On logistic regression analysis, the higher the pre-injury FRAIL score, the greater the likelihood of mortality within 1 year (OR = 1.74; P = .001).
Data source: A secondary analysis of 188 injured patients aged 65 and older who were admitted through the ED between October 2013 and March 2014.
Disclosures: Dr. Maxwell reported having no financial disclosures.
Damage control laparotomy rates fell after QI project
WAIKOLOA, HI. – If you openly share your institution’s use of damage control laparotomy, it is possible to safely decrease use of the procedure, according to results from a 2-year, single-center quality improvement project.
“Damage control laparotomy (DCL) is currently overused, both in trauma and general surgery,” John A. Harvin, MD, said in an interview in advance of the annual meeting of the American Association for the Surgery of Trauma. “One of the barriers to discussing the overuse is that no one knows what the ‘right’ rate of DCL should be. The vast majority of papers reporting findings regarding DCL fail to provide a denominator, thus actual rates of DCL across the country are not known. It is only by word of mouth that the overuse comes out.”
In a quality improvement (QI) project designed to decrease the rate of DCL at the University of Texas Health Science Center, Houston, Dr. Harvin and his associates prospectively evaluated all emergent laparotomies performed at the institution from November 2013 to October 2015. During year 1 of the QI project, trauma faculty completed report cards immediately following every DCL. During year 2, the researchers collectively reviewed DCLs every other month to determine which patients may have safely undergone definitive laparotomy. They prospectively compared the morbidity and mortality of patients in the quality improvement group with that of a published historical cohort group of patients who underwent emergent laparotomy between January 2011 and October 2013 (Am. J. Surg. 2016;212[1]:34-9).
Dr. Harvin of the division of acute care surgery at the university, reported that the rate of DCL among the historical control group was 39%. Soon after the QI project was implemented the DCL rate fell to 23% (P less than .05), and it declined further following completion of the project, to 18%. “This was accomplished by open discussion among our faculty on who and why DCL was done,” he said. “Over the course of the 2-year QI project, approximately 70 DCLs were avoided without an increase in complications or mortality.”
Many of the findings surprised the researchers, including the fact that the indications for DCL did not differ before and after implementation of the QI project. “So, surgeons did not necessarily abandon certain indications but used all indications more selectively,” Dr. Harvin noted. “That being said, we were able to identify a few indications for DCL that may or may not be necessary. Lastly, despite a significant reduction in the overall use of DCL, we saw no change in rates of complications. This flies in the face of many papers reporting an association between DCL and all kinds of morbidities.”
He acknowledged certain limitations of the study, including the fact that the researchers identified a Hawthorne effect after implementation of the QI intervention. “Despite this, there was a sustained reduction in the rate of DCL that persisted following termination of the project,” Dr. Harvin said. “Additionally, this is not a randomized clinical trial, but a before and after trial which is subject to the usual methodological flaws such as confounding by temporal change and differences in baseline characteristics of the patient populations.” He reported having no financial disclosures.
WAIKOLOA, HI. – If you openly share your institution’s use of damage control laparotomy, it is possible to safely decrease use of the procedure, according to results from a 2-year, single-center quality improvement project.
“Damage control laparotomy (DCL) is currently overused, both in trauma and general surgery,” John A. Harvin, MD, said in an interview in advance of the annual meeting of the American Association for the Surgery of Trauma. “One of the barriers to discussing the overuse is that no one knows what the ‘right’ rate of DCL should be. The vast majority of papers reporting findings regarding DCL fail to provide a denominator, thus actual rates of DCL across the country are not known. It is only by word of mouth that the overuse comes out.”
In a quality improvement (QI) project designed to decrease the rate of DCL at the University of Texas Health Science Center, Houston, Dr. Harvin and his associates prospectively evaluated all emergent laparotomies performed at the institution from November 2013 to October 2015. During year 1 of the QI project, trauma faculty completed report cards immediately following every DCL. During year 2, the researchers collectively reviewed DCLs every other month to determine which patients may have safely undergone definitive laparotomy. They prospectively compared the morbidity and mortality of patients in the quality improvement group with that of a published historical cohort group of patients who underwent emergent laparotomy between January 2011 and October 2013 (Am. J. Surg. 2016;212[1]:34-9).
Dr. Harvin of the division of acute care surgery at the university, reported that the rate of DCL among the historical control group was 39%. Soon after the QI project was implemented the DCL rate fell to 23% (P less than .05), and it declined further following completion of the project, to 18%. “This was accomplished by open discussion among our faculty on who and why DCL was done,” he said. “Over the course of the 2-year QI project, approximately 70 DCLs were avoided without an increase in complications or mortality.”
Many of the findings surprised the researchers, including the fact that the indications for DCL did not differ before and after implementation of the QI project. “So, surgeons did not necessarily abandon certain indications but used all indications more selectively,” Dr. Harvin noted. “That being said, we were able to identify a few indications for DCL that may or may not be necessary. Lastly, despite a significant reduction in the overall use of DCL, we saw no change in rates of complications. This flies in the face of many papers reporting an association between DCL and all kinds of morbidities.”
He acknowledged certain limitations of the study, including the fact that the researchers identified a Hawthorne effect after implementation of the QI intervention. “Despite this, there was a sustained reduction in the rate of DCL that persisted following termination of the project,” Dr. Harvin said. “Additionally, this is not a randomized clinical trial, but a before and after trial which is subject to the usual methodological flaws such as confounding by temporal change and differences in baseline characteristics of the patient populations.” He reported having no financial disclosures.
WAIKOLOA, HI. – If you openly share your institution’s use of damage control laparotomy, it is possible to safely decrease use of the procedure, according to results from a 2-year, single-center quality improvement project.
“Damage control laparotomy (DCL) is currently overused, both in trauma and general surgery,” John A. Harvin, MD, said in an interview in advance of the annual meeting of the American Association for the Surgery of Trauma. “One of the barriers to discussing the overuse is that no one knows what the ‘right’ rate of DCL should be. The vast majority of papers reporting findings regarding DCL fail to provide a denominator, thus actual rates of DCL across the country are not known. It is only by word of mouth that the overuse comes out.”
In a quality improvement (QI) project designed to decrease the rate of DCL at the University of Texas Health Science Center, Houston, Dr. Harvin and his associates prospectively evaluated all emergent laparotomies performed at the institution from November 2013 to October 2015. During year 1 of the QI project, trauma faculty completed report cards immediately following every DCL. During year 2, the researchers collectively reviewed DCLs every other month to determine which patients may have safely undergone definitive laparotomy. They prospectively compared the morbidity and mortality of patients in the quality improvement group with that of a published historical cohort group of patients who underwent emergent laparotomy between January 2011 and October 2013 (Am. J. Surg. 2016;212[1]:34-9).
Dr. Harvin of the division of acute care surgery at the university, reported that the rate of DCL among the historical control group was 39%. Soon after the QI project was implemented the DCL rate fell to 23% (P less than .05), and it declined further following completion of the project, to 18%. “This was accomplished by open discussion among our faculty on who and why DCL was done,” he said. “Over the course of the 2-year QI project, approximately 70 DCLs were avoided without an increase in complications or mortality.”
Many of the findings surprised the researchers, including the fact that the indications for DCL did not differ before and after implementation of the QI project. “So, surgeons did not necessarily abandon certain indications but used all indications more selectively,” Dr. Harvin noted. “That being said, we were able to identify a few indications for DCL that may or may not be necessary. Lastly, despite a significant reduction in the overall use of DCL, we saw no change in rates of complications. This flies in the face of many papers reporting an association between DCL and all kinds of morbidities.”
He acknowledged certain limitations of the study, including the fact that the researchers identified a Hawthorne effect after implementation of the QI intervention. “Despite this, there was a sustained reduction in the rate of DCL that persisted following termination of the project,” Dr. Harvin said. “Additionally, this is not a randomized clinical trial, but a before and after trial which is subject to the usual methodological flaws such as confounding by temporal change and differences in baseline characteristics of the patient populations.” He reported having no financial disclosures.
AT THE AAST ANNUAL MEETING
Key clinical point: Implementation of a quality improvement project led to significantly decreased rates of damage control laparotomy.
Major finding: Soon after the QI project was implemented, the damage control laparotomy (DCL) rate fell from 39% to 23% (P less than .05), and it declined further following completion of the project, to 18%.
Data source: A prospective evaluation of all emergent laparotomies performed at the University of Texas Health Science Center, Houston, from November 2013 to October 2015.
Disclosures: Dr. Harvin reported having no financial disclosures.
Think outside the ‘cardiac box’ to predict cardiac injury
WAIKOLOA, HI. – For gunshot wounds, the current “cardiac box” was the poorest predictor of cardiac injury, results from a single-center retrospective study demonstrated.
“We determined that, from a statistical standpoint, the cardiac box should be redefined to include the area of the thorax that extends from the clavicle to xiphoid and from the anterior midline to the posterior midline of the left thorax,” Bryan C. Morse, MD, said in an interview in advance of the annual meeting of the American Association for the Surgery of Trauma. “The classic cardiac box is inadequate to discriminate whether a gunshot wound will create a cardiac injury.”
Dr. Morse of Emory University and Grady Memorial Hospital, Atlanta, and his associates recently published their experience with penetrating cardiac injuries over the past 36 years and documented an increase in the number of cardiac injuries from gunshots over the past 10 years (J. Trauma Acute Care Surg. 2016 Jul 6. doi: 10.1097/TA.0000000000001165). They also noted that several of these injuries were caused by penetrating thoracic wounds outside the cardiac box.
The cardiac box is currently defined as the area of the chest overlying the heart, bounded by the midclavicular lines (laterally) and from the clavicles to the tip of the xiphoid. “Surgical teaching dictates that penetrating injuries (i.e. stab wounds and gunshot wounds) in the box have the highest likelihood of cardiac injury and thereby mandate further evaluation,” Dr. Morse said. “These studies, however, are based on small patient sample sizes in which the majority were stab wound victims and underwent minimal statistical scrutiny.”
In what he said is the largest study of its kind, Dr. Morse and his associates conducted a retrospective review of trauma registry data from Grady’s trauma center and autopsy reports to identify patients with penetrating thoracic gunshot wounds and cardiac injury from 2011 to 2013 and to evaluate the relationship between penetrating injuries and the likelihood of a cardiac injury. Using a circumferential grid system around the thorax, the researchers employed logistic regression analysis to compare differences in rates of cardiac injury from entrance/exit wounds in the cardiac box, versus outside the box. They repeated the process to identify potential regions that yield improved predictions for cardiac injury over the current definition of the cardiac box.
Over the 3-year study period, 263 patients sustained 735 penetrating thoracic wounds, of which 80% were gunshot wounds (GSWs). Most of the patients were males (89%) with a median of two injuries each. After stab wounds were excluded, 277 GSWs to the thorax were included for study and 95 (34%) injured the heart. Of the 233 GSWs entering the cardiac box, 30% caused cardiac injury while, of the 44 GSWs outside the cardiac box, 32% penetrated the heart, suggesting that the current cardiac box is a poor predictor of cardiac injury relative to the thoracic non–cardiac box regions (OR 1.1; P = .71).
The researchers observed that the regions from the anterior to the posterior midline of the left thorax provided the highest positive predictive value, with a sensitivity of 90% and a specificity of 31%, making this region the most statistically significant discriminator of cardiac injury (OR, 4.4; P less than .01). This finding was primarily based on the fact that gunshots to the left lateral chest (an area not currently included in the box) had a high rate of cardiac injury (41%; OR, 1.4).
“The current cardiac box is unable to discriminate between gunshot wounds that will cause a cardiac injury and those that will not,” Dr. Morse said. “Any gunshot wound to the chest can cause a cardiac injury. While clinically relevant box borders would include the left chest, the bottom line for surgeons is to think outside the current cardiac box.”
The improved cardiac box that he and his associates proposed includes the area from the clavicles to the xiphoid and from the anterior to the posterior midline over the left thorax. “While this may be intuitive, it is not what we as surgeons have been teaching,” he said. “Finally, gunshots to areas such as the right posterior and posterolateral chest were associated with rates of cardiac injury greater than 30% despite their distance from the heart. This led us to conclude that a gunshot anywhere to the chest should be considered to potentially cause a cardiac injury.”
Dr. Morse acknowledged certain limitations of the study, including the fact that the study excluded graze wounds and gunshots above the clavicles and below the xiphoid. “However, a small percentage of these did cause cardiac injuries, which emphasizes the point that gunshot wounds from any entrance can cause cardiac injury.”
Invited discussant Nicholas Namias, MD, professor and chief of the division of acute care surgery at Jackson Memorial Hospital, Miami, said that the study by Dr. Morse and his associates “confirms what Dr. [Grace] Rozycki showed 20 years ago: Forget the [cardiac] box; it’s dead. Just throw an ultrasound probe on.”
Dr. Morse reported having no relevant financial disclosures.
WAIKOLOA, HI. – For gunshot wounds, the current “cardiac box” was the poorest predictor of cardiac injury, results from a single-center retrospective study demonstrated.
“We determined that, from a statistical standpoint, the cardiac box should be redefined to include the area of the thorax that extends from the clavicle to xiphoid and from the anterior midline to the posterior midline of the left thorax,” Bryan C. Morse, MD, said in an interview in advance of the annual meeting of the American Association for the Surgery of Trauma. “The classic cardiac box is inadequate to discriminate whether a gunshot wound will create a cardiac injury.”
Dr. Morse of Emory University and Grady Memorial Hospital, Atlanta, and his associates recently published their experience with penetrating cardiac injuries over the past 36 years and documented an increase in the number of cardiac injuries from gunshots over the past 10 years (J. Trauma Acute Care Surg. 2016 Jul 6. doi: 10.1097/TA.0000000000001165). They also noted that several of these injuries were caused by penetrating thoracic wounds outside the cardiac box.
The cardiac box is currently defined as the area of the chest overlying the heart, bounded by the midclavicular lines (laterally) and from the clavicles to the tip of the xiphoid. “Surgical teaching dictates that penetrating injuries (i.e. stab wounds and gunshot wounds) in the box have the highest likelihood of cardiac injury and thereby mandate further evaluation,” Dr. Morse said. “These studies, however, are based on small patient sample sizes in which the majority were stab wound victims and underwent minimal statistical scrutiny.”
In what he said is the largest study of its kind, Dr. Morse and his associates conducted a retrospective review of trauma registry data from Grady’s trauma center and autopsy reports to identify patients with penetrating thoracic gunshot wounds and cardiac injury from 2011 to 2013 and to evaluate the relationship between penetrating injuries and the likelihood of a cardiac injury. Using a circumferential grid system around the thorax, the researchers employed logistic regression analysis to compare differences in rates of cardiac injury from entrance/exit wounds in the cardiac box, versus outside the box. They repeated the process to identify potential regions that yield improved predictions for cardiac injury over the current definition of the cardiac box.
Over the 3-year study period, 263 patients sustained 735 penetrating thoracic wounds, of which 80% were gunshot wounds (GSWs). Most of the patients were males (89%) with a median of two injuries each. After stab wounds were excluded, 277 GSWs to the thorax were included for study and 95 (34%) injured the heart. Of the 233 GSWs entering the cardiac box, 30% caused cardiac injury while, of the 44 GSWs outside the cardiac box, 32% penetrated the heart, suggesting that the current cardiac box is a poor predictor of cardiac injury relative to the thoracic non–cardiac box regions (OR 1.1; P = .71).
The researchers observed that the regions from the anterior to the posterior midline of the left thorax provided the highest positive predictive value, with a sensitivity of 90% and a specificity of 31%, making this region the most statistically significant discriminator of cardiac injury (OR, 4.4; P less than .01). This finding was primarily based on the fact that gunshots to the left lateral chest (an area not currently included in the box) had a high rate of cardiac injury (41%; OR, 1.4).
“The current cardiac box is unable to discriminate between gunshot wounds that will cause a cardiac injury and those that will not,” Dr. Morse said. “Any gunshot wound to the chest can cause a cardiac injury. While clinically relevant box borders would include the left chest, the bottom line for surgeons is to think outside the current cardiac box.”
The improved cardiac box that he and his associates proposed includes the area from the clavicles to the xiphoid and from the anterior to the posterior midline over the left thorax. “While this may be intuitive, it is not what we as surgeons have been teaching,” he said. “Finally, gunshots to areas such as the right posterior and posterolateral chest were associated with rates of cardiac injury greater than 30% despite their distance from the heart. This led us to conclude that a gunshot anywhere to the chest should be considered to potentially cause a cardiac injury.”
Dr. Morse acknowledged certain limitations of the study, including the fact that the study excluded graze wounds and gunshots above the clavicles and below the xiphoid. “However, a small percentage of these did cause cardiac injuries, which emphasizes the point that gunshot wounds from any entrance can cause cardiac injury.”
Invited discussant Nicholas Namias, MD, professor and chief of the division of acute care surgery at Jackson Memorial Hospital, Miami, said that the study by Dr. Morse and his associates “confirms what Dr. [Grace] Rozycki showed 20 years ago: Forget the [cardiac] box; it’s dead. Just throw an ultrasound probe on.”
Dr. Morse reported having no relevant financial disclosures.
WAIKOLOA, HI. – For gunshot wounds, the current “cardiac box” was the poorest predictor of cardiac injury, results from a single-center retrospective study demonstrated.
“We determined that, from a statistical standpoint, the cardiac box should be redefined to include the area of the thorax that extends from the clavicle to xiphoid and from the anterior midline to the posterior midline of the left thorax,” Bryan C. Morse, MD, said in an interview in advance of the annual meeting of the American Association for the Surgery of Trauma. “The classic cardiac box is inadequate to discriminate whether a gunshot wound will create a cardiac injury.”
Dr. Morse of Emory University and Grady Memorial Hospital, Atlanta, and his associates recently published their experience with penetrating cardiac injuries over the past 36 years and documented an increase in the number of cardiac injuries from gunshots over the past 10 years (J. Trauma Acute Care Surg. 2016 Jul 6. doi: 10.1097/TA.0000000000001165). They also noted that several of these injuries were caused by penetrating thoracic wounds outside the cardiac box.
The cardiac box is currently defined as the area of the chest overlying the heart, bounded by the midclavicular lines (laterally) and from the clavicles to the tip of the xiphoid. “Surgical teaching dictates that penetrating injuries (i.e. stab wounds and gunshot wounds) in the box have the highest likelihood of cardiac injury and thereby mandate further evaluation,” Dr. Morse said. “These studies, however, are based on small patient sample sizes in which the majority were stab wound victims and underwent minimal statistical scrutiny.”
In what he said is the largest study of its kind, Dr. Morse and his associates conducted a retrospective review of trauma registry data from Grady’s trauma center and autopsy reports to identify patients with penetrating thoracic gunshot wounds and cardiac injury from 2011 to 2013 and to evaluate the relationship between penetrating injuries and the likelihood of a cardiac injury. Using a circumferential grid system around the thorax, the researchers employed logistic regression analysis to compare differences in rates of cardiac injury from entrance/exit wounds in the cardiac box, versus outside the box. They repeated the process to identify potential regions that yield improved predictions for cardiac injury over the current definition of the cardiac box.
Over the 3-year study period, 263 patients sustained 735 penetrating thoracic wounds, of which 80% were gunshot wounds (GSWs). Most of the patients were males (89%) with a median of two injuries each. After stab wounds were excluded, 277 GSWs to the thorax were included for study and 95 (34%) injured the heart. Of the 233 GSWs entering the cardiac box, 30% caused cardiac injury while, of the 44 GSWs outside the cardiac box, 32% penetrated the heart, suggesting that the current cardiac box is a poor predictor of cardiac injury relative to the thoracic non–cardiac box regions (OR 1.1; P = .71).
The researchers observed that the regions from the anterior to the posterior midline of the left thorax provided the highest positive predictive value, with a sensitivity of 90% and a specificity of 31%, making this region the most statistically significant discriminator of cardiac injury (OR, 4.4; P less than .01). This finding was primarily based on the fact that gunshots to the left lateral chest (an area not currently included in the box) had a high rate of cardiac injury (41%; OR, 1.4).
“The current cardiac box is unable to discriminate between gunshot wounds that will cause a cardiac injury and those that will not,” Dr. Morse said. “Any gunshot wound to the chest can cause a cardiac injury. While clinically relevant box borders would include the left chest, the bottom line for surgeons is to think outside the current cardiac box.”
The improved cardiac box that he and his associates proposed includes the area from the clavicles to the xiphoid and from the anterior to the posterior midline over the left thorax. “While this may be intuitive, it is not what we as surgeons have been teaching,” he said. “Finally, gunshots to areas such as the right posterior and posterolateral chest were associated with rates of cardiac injury greater than 30% despite their distance from the heart. This led us to conclude that a gunshot anywhere to the chest should be considered to potentially cause a cardiac injury.”
Dr. Morse acknowledged certain limitations of the study, including the fact that the study excluded graze wounds and gunshots above the clavicles and below the xiphoid. “However, a small percentage of these did cause cardiac injuries, which emphasizes the point that gunshot wounds from any entrance can cause cardiac injury.”
Invited discussant Nicholas Namias, MD, professor and chief of the division of acute care surgery at Jackson Memorial Hospital, Miami, said that the study by Dr. Morse and his associates “confirms what Dr. [Grace] Rozycki showed 20 years ago: Forget the [cardiac] box; it’s dead. Just throw an ultrasound probe on.”
Dr. Morse reported having no relevant financial disclosures.
AT THE AAST ANNUAL MEETING
Key clinical point: The current cardiac box is inadequate to discriminate whether a gunshot wound will create a cardiac injury.
Major finding: Of the 233 gunshot wounds entering the cardiac box, 30% caused cardiac injury while, of the 44 GSWs outside the cardiac box, 32% penetrated the heart, suggesting that the current cardiac box is a poor predictor of cardiac injury relative to the thoracic non–cardiac box regions (OR 1.1; P = .71).
Data source: A retrospective review of 236 patients with penetrating thoracic gunshot wounds and cardiac injury from 2011 to 2013.
Disclosures: Dr. Morse reported having no relevant financial disclosures.
Pain starting in knee later arises in other joints
People who develop knee pain associated with osteoarthritis often subsequently develop pain in other joints, according to a study of two observational, community-based cohorts that could not discern any pattern of new pain sites.
In the “first investigation of the association of knee pain with pain in multiple other sites,” David T. Felson, MD, of Boston University and his colleagues reported that the regions where pain developed after first appearing in the knee varied from person to person and occurred in both upper and lower extremities, which goes against the hypothesis that adjacent joints are most often affected by knee pain.
The study involved patients from the MOST (Multicenter Osteoarthritis Study) trial, including 281 with knee pain at the index visit (168 unilaterally) and 852 without, as well as patients from OAI (the Osteoarthritis Initiative), including 412 with knee pain at the index visit (241 unilaterally), and 1,941 without. The investigators assessed the patients’ data for 14 total joints outside of the knees: 2 each of feet, ankles, hips, hands, wrists, elbows, and shoulders (Arthritis Rheumatol. 2016 Sep 2. doi: 10.1002/art.39848).
Patients with new-onset knee pain at the index visit reported a mean of 2.3 painful joints outside the knee, compared with a significantly lower number of 1.3 reported by those without knee pain. The mean number of nonknee joints with pain was higher among patients with bilateral knee pain, compared with unilateral knee pain. The percentage of patients who reported pain outside the knee rose with the number of painful knees: 80% for two, 64% for one, and 50% for none.
The patients who developed new unilateral knee pain at the index visit also experienced an increase in prevalent joint pain in multiple joints in upper- and lower-extremity sites. In particular, the investigators noted that ipsilateral prevalent hip joint pain, which they characterized as pain in the groin or front of the thigh, was more than twice as likely to occur among those with new unilateral knee pain at the index visit, but the odds for contralateral hip joint pain did not reach statistical significance. The comparisons were adjusted for age, sex, body mass index, depression at the index visit, study (MOST or OAI), and count of painful upper and lower limb joints at the index visit (excluding knees).
When examining only patients with new-onset joint pain outside of the knee, the odds of patients with new knee pain to later develop new-onset joint pain outside the knee were 30% higher than for those without knee pain. Patients with new knee pain had a mean 2.6 new painful joints out of 12.1 eligible joints, compared with 2.0 new painful joints in those without knee pain out of 12.7 eligible joints. (Joint regions with prevalent symptoms at the index visit were excluded as incident painful sites.) Patients with knee pain also had a consistently higher rate of new-onset pain in nonknee joints when compared with patients without knee pain in at least half of the follow-up visits over the course of the MOST and OAI studies. Sensitivity analyses indicated that the association between knee pain and subsequent pain in other joints was not driven by the inclusion of patients with widespread pain.
“There was no clear-cut predilection for pain in any specific lower-extremity joint region,” the investigators wrote.
The investigators noted that other researchers have suggested that patients with knee pain may be at higher risk for lower-extremity joint pain because of changes to their gait that gradually cause damage to other joints, but evidence in this study doesn’t “necessarily support the argument that in persons with knee pain, aberrant loading by altered movement patterns induces pain in only nearby joints. Our findings suggest that the sites affected are more than just hip and ankle and that there is no special predilection for pain in these locations.”
While the investigators cannot differentiate underlying mechanisms for their study’s finding of multiple co-occurring sites of joint pain in people with new-onset knee pain, they suggested that it “supports either a predilection for osteoarthritic changes at multiple joint sites and/or raises the possibility that nervous system–driven pain sensitization increases the risk not only of widespread pain but even of regional pain. Since symptomatic OA is unusual in some of these painful sites (e.g., elbow, shoulder, ankle), pain sensitization would seem a more likely explanation.”
Some of the study’s limitations described by the investigators included the uncertainty surrounding whether new-onset knee pain was truly new onset or whether it was a reoccurrence, and also the fact that most of the people in the two cohorts had multiple sites of joint pain at both the baseline and the index visit and there were too few people with no sites of pain outside the knee to carry out subanalyses in that group, which “speaks to the high prevalence of multiple joint pains in older adult cohorts.”
The research was supported by grants from the National Institutes of Health. The authors had no disclosures to report.
People who develop knee pain associated with osteoarthritis often subsequently develop pain in other joints, according to a study of two observational, community-based cohorts that could not discern any pattern of new pain sites.
In the “first investigation of the association of knee pain with pain in multiple other sites,” David T. Felson, MD, of Boston University and his colleagues reported that the regions where pain developed after first appearing in the knee varied from person to person and occurred in both upper and lower extremities, which goes against the hypothesis that adjacent joints are most often affected by knee pain.
The study involved patients from the MOST (Multicenter Osteoarthritis Study) trial, including 281 with knee pain at the index visit (168 unilaterally) and 852 without, as well as patients from OAI (the Osteoarthritis Initiative), including 412 with knee pain at the index visit (241 unilaterally), and 1,941 without. The investigators assessed the patients’ data for 14 total joints outside of the knees: 2 each of feet, ankles, hips, hands, wrists, elbows, and shoulders (Arthritis Rheumatol. 2016 Sep 2. doi: 10.1002/art.39848).
Patients with new-onset knee pain at the index visit reported a mean of 2.3 painful joints outside the knee, compared with a significantly lower number of 1.3 reported by those without knee pain. The mean number of nonknee joints with pain was higher among patients with bilateral knee pain, compared with unilateral knee pain. The percentage of patients who reported pain outside the knee rose with the number of painful knees: 80% for two, 64% for one, and 50% for none.
The patients who developed new unilateral knee pain at the index visit also experienced an increase in prevalent joint pain in multiple joints in upper- and lower-extremity sites. In particular, the investigators noted that ipsilateral prevalent hip joint pain, which they characterized as pain in the groin or front of the thigh, was more than twice as likely to occur among those with new unilateral knee pain at the index visit, but the odds for contralateral hip joint pain did not reach statistical significance. The comparisons were adjusted for age, sex, body mass index, depression at the index visit, study (MOST or OAI), and count of painful upper and lower limb joints at the index visit (excluding knees).
When examining only patients with new-onset joint pain outside of the knee, the odds of patients with new knee pain to later develop new-onset joint pain outside the knee were 30% higher than for those without knee pain. Patients with new knee pain had a mean 2.6 new painful joints out of 12.1 eligible joints, compared with 2.0 new painful joints in those without knee pain out of 12.7 eligible joints. (Joint regions with prevalent symptoms at the index visit were excluded as incident painful sites.) Patients with knee pain also had a consistently higher rate of new-onset pain in nonknee joints when compared with patients without knee pain in at least half of the follow-up visits over the course of the MOST and OAI studies. Sensitivity analyses indicated that the association between knee pain and subsequent pain in other joints was not driven by the inclusion of patients with widespread pain.
“There was no clear-cut predilection for pain in any specific lower-extremity joint region,” the investigators wrote.
The investigators noted that other researchers have suggested that patients with knee pain may be at higher risk for lower-extremity joint pain because of changes to their gait that gradually cause damage to other joints, but evidence in this study doesn’t “necessarily support the argument that in persons with knee pain, aberrant loading by altered movement patterns induces pain in only nearby joints. Our findings suggest that the sites affected are more than just hip and ankle and that there is no special predilection for pain in these locations.”
While the investigators cannot differentiate underlying mechanisms for their study’s finding of multiple co-occurring sites of joint pain in people with new-onset knee pain, they suggested that it “supports either a predilection for osteoarthritic changes at multiple joint sites and/or raises the possibility that nervous system–driven pain sensitization increases the risk not only of widespread pain but even of regional pain. Since symptomatic OA is unusual in some of these painful sites (e.g., elbow, shoulder, ankle), pain sensitization would seem a more likely explanation.”
Some of the study’s limitations described by the investigators included the uncertainty surrounding whether new-onset knee pain was truly new onset or whether it was a reoccurrence, and also the fact that most of the people in the two cohorts had multiple sites of joint pain at both the baseline and the index visit and there were too few people with no sites of pain outside the knee to carry out subanalyses in that group, which “speaks to the high prevalence of multiple joint pains in older adult cohorts.”
The research was supported by grants from the National Institutes of Health. The authors had no disclosures to report.
People who develop knee pain associated with osteoarthritis often subsequently develop pain in other joints, according to a study of two observational, community-based cohorts that could not discern any pattern of new pain sites.
In the “first investigation of the association of knee pain with pain in multiple other sites,” David T. Felson, MD, of Boston University and his colleagues reported that the regions where pain developed after first appearing in the knee varied from person to person and occurred in both upper and lower extremities, which goes against the hypothesis that adjacent joints are most often affected by knee pain.
The study involved patients from the MOST (Multicenter Osteoarthritis Study) trial, including 281 with knee pain at the index visit (168 unilaterally) and 852 without, as well as patients from OAI (the Osteoarthritis Initiative), including 412 with knee pain at the index visit (241 unilaterally), and 1,941 without. The investigators assessed the patients’ data for 14 total joints outside of the knees: 2 each of feet, ankles, hips, hands, wrists, elbows, and shoulders (Arthritis Rheumatol. 2016 Sep 2. doi: 10.1002/art.39848).
Patients with new-onset knee pain at the index visit reported a mean of 2.3 painful joints outside the knee, compared with a significantly lower number of 1.3 reported by those without knee pain. The mean number of nonknee joints with pain was higher among patients with bilateral knee pain, compared with unilateral knee pain. The percentage of patients who reported pain outside the knee rose with the number of painful knees: 80% for two, 64% for one, and 50% for none.
The patients who developed new unilateral knee pain at the index visit also experienced an increase in prevalent joint pain in multiple joints in upper- and lower-extremity sites. In particular, the investigators noted that ipsilateral prevalent hip joint pain, which they characterized as pain in the groin or front of the thigh, was more than twice as likely to occur among those with new unilateral knee pain at the index visit, but the odds for contralateral hip joint pain did not reach statistical significance. The comparisons were adjusted for age, sex, body mass index, depression at the index visit, study (MOST or OAI), and count of painful upper and lower limb joints at the index visit (excluding knees).
When examining only patients with new-onset joint pain outside of the knee, the odds of patients with new knee pain to later develop new-onset joint pain outside the knee were 30% higher than for those without knee pain. Patients with new knee pain had a mean 2.6 new painful joints out of 12.1 eligible joints, compared with 2.0 new painful joints in those without knee pain out of 12.7 eligible joints. (Joint regions with prevalent symptoms at the index visit were excluded as incident painful sites.) Patients with knee pain also had a consistently higher rate of new-onset pain in nonknee joints when compared with patients without knee pain in at least half of the follow-up visits over the course of the MOST and OAI studies. Sensitivity analyses indicated that the association between knee pain and subsequent pain in other joints was not driven by the inclusion of patients with widespread pain.
“There was no clear-cut predilection for pain in any specific lower-extremity joint region,” the investigators wrote.
The investigators noted that other researchers have suggested that patients with knee pain may be at higher risk for lower-extremity joint pain because of changes to their gait that gradually cause damage to other joints, but evidence in this study doesn’t “necessarily support the argument that in persons with knee pain, aberrant loading by altered movement patterns induces pain in only nearby joints. Our findings suggest that the sites affected are more than just hip and ankle and that there is no special predilection for pain in these locations.”
While the investigators cannot differentiate underlying mechanisms for their study’s finding of multiple co-occurring sites of joint pain in people with new-onset knee pain, they suggested that it “supports either a predilection for osteoarthritic changes at multiple joint sites and/or raises the possibility that nervous system–driven pain sensitization increases the risk not only of widespread pain but even of regional pain. Since symptomatic OA is unusual in some of these painful sites (e.g., elbow, shoulder, ankle), pain sensitization would seem a more likely explanation.”
Some of the study’s limitations described by the investigators included the uncertainty surrounding whether new-onset knee pain was truly new onset or whether it was a reoccurrence, and also the fact that most of the people in the two cohorts had multiple sites of joint pain at both the baseline and the index visit and there were too few people with no sites of pain outside the knee to carry out subanalyses in that group, which “speaks to the high prevalence of multiple joint pains in older adult cohorts.”
The research was supported by grants from the National Institutes of Health. The authors had no disclosures to report.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point:People with frequently painful knees often develop pain in joints outside the knee, and the sites vary from person to person.
Major finding: The odds of patients with new knee pain to later develop joint pain outside the knee were 30% higher than for those without knee pain.
Data source: A study of 693 persons with index visit knee pain and 2,793 without it from two community-based cohorts.
Disclosures: The research was supported by grants from the National Institutes of Health. The authors had no disclosures to report.
United States an expensive place for knee, hip replacement
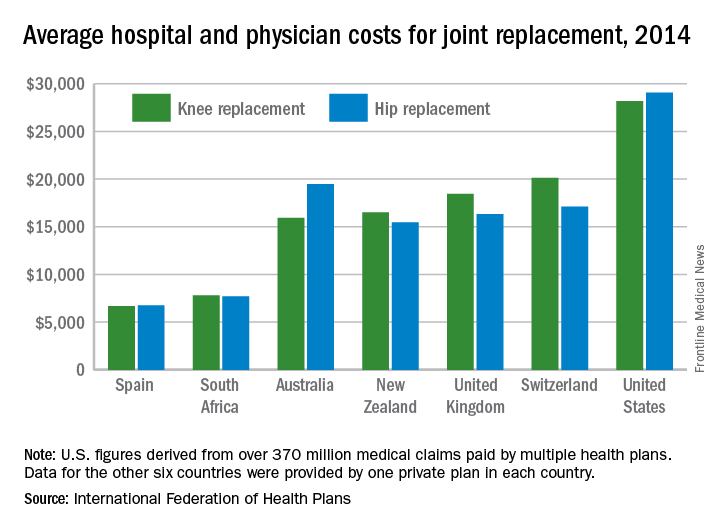
Knee and hip replacement surgeries were more expensive in the United States than in a group of six other industrialized countries in 2014, according to a report from the International Federation of Health Plans.
The U.S. average for total hospital and physician costs was $28,184 for knee replacement and $29,067 for hip replacement. Switzerland was the next most expensive country for knee replacements at $20,132, and Australia was second for hip replacements at $19,484. Spain had the lowest average cost for both surgeries: $6,687 for knee replacement and $6,757 for hip replacement, the IFHP reported.
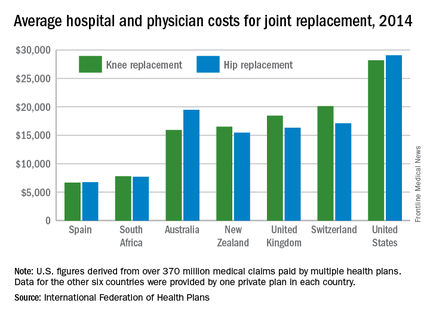
“We look at these numbers every year and it’s always a shocking demonstration of how much procedures and prescription drugs actually cost,” IFHP Chief Executive Tom Sackville said in a written statement. “There is no reason why identical procedures and products should vary in price so much across countries: it illustrates the damaging effects of an inadequately regulated health care market.”
The IFHP consists of 80 member companies in 25 countries. For the survey, costs for each country were submitted by participating member plans. Costs for the United States are derived from over 370 million employer-sponsored medical claims incurred from Jan. 1, 2014, to Dec. 31, 2014, and paid by multiple health plans. Cost data for the other six countries were provided by one private plan in each country.
Knee and hip replacement surgeries were more expensive in the United States than in a group of six other industrialized countries in 2014, according to a report from the International Federation of Health Plans.
The U.S. average for total hospital and physician costs was $28,184 for knee replacement and $29,067 for hip replacement. Switzerland was the next most expensive country for knee replacements at $20,132, and Australia was second for hip replacements at $19,484. Spain had the lowest average cost for both surgeries: $6,687 for knee replacement and $6,757 for hip replacement, the IFHP reported.

“We look at these numbers every year and it’s always a shocking demonstration of how much procedures and prescription drugs actually cost,” IFHP Chief Executive Tom Sackville said in a written statement. “There is no reason why identical procedures and products should vary in price so much across countries: it illustrates the damaging effects of an inadequately regulated health care market.”
The IFHP consists of 80 member companies in 25 countries. For the survey, costs for each country were submitted by participating member plans. Costs for the United States are derived from over 370 million employer-sponsored medical claims incurred from Jan. 1, 2014, to Dec. 31, 2014, and paid by multiple health plans. Cost data for the other six countries were provided by one private plan in each country.
Knee and hip replacement surgeries were more expensive in the United States than in a group of six other industrialized countries in 2014, according to a report from the International Federation of Health Plans.
The U.S. average for total hospital and physician costs was $28,184 for knee replacement and $29,067 for hip replacement. Switzerland was the next most expensive country for knee replacements at $20,132, and Australia was second for hip replacements at $19,484. Spain had the lowest average cost for both surgeries: $6,687 for knee replacement and $6,757 for hip replacement, the IFHP reported.

“We look at these numbers every year and it’s always a shocking demonstration of how much procedures and prescription drugs actually cost,” IFHP Chief Executive Tom Sackville said in a written statement. “There is no reason why identical procedures and products should vary in price so much across countries: it illustrates the damaging effects of an inadequately regulated health care market.”
The IFHP consists of 80 member companies in 25 countries. For the survey, costs for each country were submitted by participating member plans. Costs for the United States are derived from over 370 million employer-sponsored medical claims incurred from Jan. 1, 2014, to Dec. 31, 2014, and paid by multiple health plans. Cost data for the other six countries were provided by one private plan in each country.
VIDEO: Medical apps will disrupt health care in a good way
BOSTON – Smartphones coupled with the rapidly growing number of medical applications are the latest disruptive technologies reshaping the way physicians conduct business and interact with patients, according to Paul Alan Wetter, MD, founder and chairman of the Society of Laparoendoscopic Surgeons.
“The message really is to the physicians out there: We need to be prepared and understand as much about this as we can,” said Dr. Wetter, clinical professor emeritus at the University of Miami. “We don’t want to be, 5 years from now ... wondering what’s going, what is this change?”
In a video interview, Dr. Wetter explained how mobile devices and apps could potentially improve technology-based tools that doctors already use, such as electronic health records, by allowing patients to carry accurate and up-to-date medical information with them.
Dr. Wetter spoke at the annual Minimally Invasive Surgery Week, held by the Society of Laparoendoscopic Surgeons. He did not report any relevant financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
BOSTON – Smartphones coupled with the rapidly growing number of medical applications are the latest disruptive technologies reshaping the way physicians conduct business and interact with patients, according to Paul Alan Wetter, MD, founder and chairman of the Society of Laparoendoscopic Surgeons.
“The message really is to the physicians out there: We need to be prepared and understand as much about this as we can,” said Dr. Wetter, clinical professor emeritus at the University of Miami. “We don’t want to be, 5 years from now ... wondering what’s going, what is this change?”
In a video interview, Dr. Wetter explained how mobile devices and apps could potentially improve technology-based tools that doctors already use, such as electronic health records, by allowing patients to carry accurate and up-to-date medical information with them.
Dr. Wetter spoke at the annual Minimally Invasive Surgery Week, held by the Society of Laparoendoscopic Surgeons. He did not report any relevant financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
BOSTON – Smartphones coupled with the rapidly growing number of medical applications are the latest disruptive technologies reshaping the way physicians conduct business and interact with patients, according to Paul Alan Wetter, MD, founder and chairman of the Society of Laparoendoscopic Surgeons.
“The message really is to the physicians out there: We need to be prepared and understand as much about this as we can,” said Dr. Wetter, clinical professor emeritus at the University of Miami. “We don’t want to be, 5 years from now ... wondering what’s going, what is this change?”
In a video interview, Dr. Wetter explained how mobile devices and apps could potentially improve technology-based tools that doctors already use, such as electronic health records, by allowing patients to carry accurate and up-to-date medical information with them.
Dr. Wetter spoke at the annual Minimally Invasive Surgery Week, held by the Society of Laparoendoscopic Surgeons. He did not report any relevant financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT MINIMALLY INVASIVE SURGERY WEEK
Four factors raise risk of post-TAVR endocarditis
Four factors – younger patient age, male sex, diabetes, and moderate to severe residual aortic regurgitation – are associated with a significantly increased risk of infective endocarditis after transcatheter aortic valve replacement, according to a report published online Sept. 13 in JAMA.
Until now, data pertaining to endocarditis following TAVR “have been limited to case reports and relatively small series with limited follow-up,” said Ander Regueiro, MD, of Laval University, Quebec City, and his associates.
They performed a retrospective analysis of data in a large international registry of TAVR cases to better characterize post-TAVR endocarditis.
Dr. Regueiro and his colleagues focused on 20,006 TAVR procedures done at 47 medical centers in Europe, North America, and South America during a 10-year period. The median time to symptom onset was 5.3 months after the procedure.
Infective endocarditis was definitively diagnosed in 250 of these cases. This incidence is similar to that reported for endocarditis following surgical aortic valve replacement, indicating that TAVR is no less predisposing to endocarditis despite being a less invasive approach.
The mean age of patients who developed post-TAVR endocarditis was 78.9 years, compared with 81.8 years for those who did not (HR, 0.97). The reason for this association is unclear, but it is possible that younger patients chosen for TAVR because of their prohibitive surgical risk carry a higher burden of comorbidity than do older patients. Similarly, 62% of endocarditis cases arose in men (HR, 1.69), and sex differences in comorbid conditions may explain the higher risk among men.
More patients who developed endocarditis had diabetes (41.7%), compared with those who did not develop endocarditis (30%), for an HR of 1.52. And patients who had moderate to severe residual aortic regurgitation after TAVR also were at much higher risk for endocarditis than were those who did not (HR, 2.05), the investigators noted (JAMA. 2016 Sep 13;316[10]:1083-92).
In contrast, factors that were not associated with endocarditis risk included chronic pulmonary disease, type of valve (self-expandable or balloon-expandable), and setting of the procedure (catheterization lab vs. operating room).
The bacteria that most commonly caused infective endocarditis were Enterococci species (24.6% of cases), Staphylococcus aureus (23.8%), and coagulase-negative staphylococci (16.8%). This should be taken into consideration when selecting antibiotics for prophylaxis before TAVR and when choosing empirical antibiotics for treatment while waiting for blood culture results, wrote Dr. Regueiro and his associates.
“This information may help clinicians identify patients at higher risk [for endocarditis] and aid in implementing appropriate preventive measures,” they noted.
This study was supported by a grant from the Alfonso Martin Escudero Foundation. Dr. Regueiro reported having no relevant financial disclosures; his associates reported ties to numerous industry sources.
Four factors – younger patient age, male sex, diabetes, and moderate to severe residual aortic regurgitation – are associated with a significantly increased risk of infective endocarditis after transcatheter aortic valve replacement, according to a report published online Sept. 13 in JAMA.
Until now, data pertaining to endocarditis following TAVR “have been limited to case reports and relatively small series with limited follow-up,” said Ander Regueiro, MD, of Laval University, Quebec City, and his associates.
They performed a retrospective analysis of data in a large international registry of TAVR cases to better characterize post-TAVR endocarditis.
Dr. Regueiro and his colleagues focused on 20,006 TAVR procedures done at 47 medical centers in Europe, North America, and South America during a 10-year period. The median time to symptom onset was 5.3 months after the procedure.
Infective endocarditis was definitively diagnosed in 250 of these cases. This incidence is similar to that reported for endocarditis following surgical aortic valve replacement, indicating that TAVR is no less predisposing to endocarditis despite being a less invasive approach.
The mean age of patients who developed post-TAVR endocarditis was 78.9 years, compared with 81.8 years for those who did not (HR, 0.97). The reason for this association is unclear, but it is possible that younger patients chosen for TAVR because of their prohibitive surgical risk carry a higher burden of comorbidity than do older patients. Similarly, 62% of endocarditis cases arose in men (HR, 1.69), and sex differences in comorbid conditions may explain the higher risk among men.
More patients who developed endocarditis had diabetes (41.7%), compared with those who did not develop endocarditis (30%), for an HR of 1.52. And patients who had moderate to severe residual aortic regurgitation after TAVR also were at much higher risk for endocarditis than were those who did not (HR, 2.05), the investigators noted (JAMA. 2016 Sep 13;316[10]:1083-92).
In contrast, factors that were not associated with endocarditis risk included chronic pulmonary disease, type of valve (self-expandable or balloon-expandable), and setting of the procedure (catheterization lab vs. operating room).
The bacteria that most commonly caused infective endocarditis were Enterococci species (24.6% of cases), Staphylococcus aureus (23.8%), and coagulase-negative staphylococci (16.8%). This should be taken into consideration when selecting antibiotics for prophylaxis before TAVR and when choosing empirical antibiotics for treatment while waiting for blood culture results, wrote Dr. Regueiro and his associates.
“This information may help clinicians identify patients at higher risk [for endocarditis] and aid in implementing appropriate preventive measures,” they noted.
This study was supported by a grant from the Alfonso Martin Escudero Foundation. Dr. Regueiro reported having no relevant financial disclosures; his associates reported ties to numerous industry sources.
Four factors – younger patient age, male sex, diabetes, and moderate to severe residual aortic regurgitation – are associated with a significantly increased risk of infective endocarditis after transcatheter aortic valve replacement, according to a report published online Sept. 13 in JAMA.
Until now, data pertaining to endocarditis following TAVR “have been limited to case reports and relatively small series with limited follow-up,” said Ander Regueiro, MD, of Laval University, Quebec City, and his associates.
They performed a retrospective analysis of data in a large international registry of TAVR cases to better characterize post-TAVR endocarditis.
Dr. Regueiro and his colleagues focused on 20,006 TAVR procedures done at 47 medical centers in Europe, North America, and South America during a 10-year period. The median time to symptom onset was 5.3 months after the procedure.
Infective endocarditis was definitively diagnosed in 250 of these cases. This incidence is similar to that reported for endocarditis following surgical aortic valve replacement, indicating that TAVR is no less predisposing to endocarditis despite being a less invasive approach.
The mean age of patients who developed post-TAVR endocarditis was 78.9 years, compared with 81.8 years for those who did not (HR, 0.97). The reason for this association is unclear, but it is possible that younger patients chosen for TAVR because of their prohibitive surgical risk carry a higher burden of comorbidity than do older patients. Similarly, 62% of endocarditis cases arose in men (HR, 1.69), and sex differences in comorbid conditions may explain the higher risk among men.
More patients who developed endocarditis had diabetes (41.7%), compared with those who did not develop endocarditis (30%), for an HR of 1.52. And patients who had moderate to severe residual aortic regurgitation after TAVR also were at much higher risk for endocarditis than were those who did not (HR, 2.05), the investigators noted (JAMA. 2016 Sep 13;316[10]:1083-92).
In contrast, factors that were not associated with endocarditis risk included chronic pulmonary disease, type of valve (self-expandable or balloon-expandable), and setting of the procedure (catheterization lab vs. operating room).
The bacteria that most commonly caused infective endocarditis were Enterococci species (24.6% of cases), Staphylococcus aureus (23.8%), and coagulase-negative staphylococci (16.8%). This should be taken into consideration when selecting antibiotics for prophylaxis before TAVR and when choosing empirical antibiotics for treatment while waiting for blood culture results, wrote Dr. Regueiro and his associates.
“This information may help clinicians identify patients at higher risk [for endocarditis] and aid in implementing appropriate preventive measures,” they noted.
This study was supported by a grant from the Alfonso Martin Escudero Foundation. Dr. Regueiro reported having no relevant financial disclosures; his associates reported ties to numerous industry sources.
FROM JAMA
Key clinical point: Four factors raise the risk that patients undergoing transcatheter aortic valve replacement will develop infective endocarditis.
Major finding: The mean age of patients who developed post-TAVR endocarditis was 78.9 years, compared with 81.8 years for those who didn’t (HR, 0.97).
Data source: A retrospective analysis of data in an international registry involving 20,006 patients who underwent TAVR at 47 medical centers during a 10-year period.
Disclosures: This study was supported by a grant from the Alfonso Martin Escudero Foundation. Dr. Regueiro reported having no relevant financial disclosures; his associates reported ties to numerous industry sources.
Health sector claims 4 spots among top 10 lobbyers in 2016
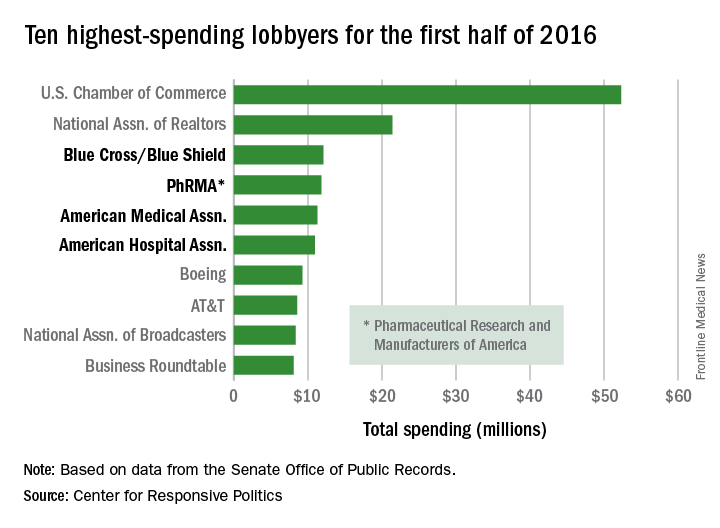
Four of the 10 highest-spending lobbyers for the first half of 2016 were in the health sector, with Blue Cross/Blue Shield occupying the sector’s top spot by a relatively small margin, according to the Center for Responsive Politics.
The four health-sectors concerns filled spots 3-6 in the overall top 10. Blue Cross/Blue Shield spent almost $12.1 million on lobbying in the first half of the year, putting it just ahead of the Pharmaceutical Research and Manufacturers of America (PhRMA), which spent $11.8 million. The American Medical Association was next at $11.3 million, followed by the American Hospital Association at $10.9 million, the center reported on OpenSecrets.org.
After those four, the next-highest health-sector spender was Pfizer, which put up almost $6.2 million in lobbying – good for 18th place for the first half of 2016. The health sector itself was the highest spending of the 121 ranked, taking a $266 million bite out of the total $1.6 billion lobbying pie for the year so far, according to the center’s analysis of data downloaded from the Senate Office of Public Records on Aug. 9.
The perennial leading spender on lobbying, the U.S. Chamber of Commerce, was well ahead of second place, with its $52.3 million more than doubling the $21.4 million spent by the National Association of Realtors. The two groups have finished 1-2 in lobbying spending every year since 2012, and the Chamber of Commerce has been the leading spender since 2001, data on OpenSecrets show.
Four of the 10 highest-spending lobbyers for the first half of 2016 were in the health sector, with Blue Cross/Blue Shield occupying the sector’s top spot by a relatively small margin, according to the Center for Responsive Politics.
The four health-sectors concerns filled spots 3-6 in the overall top 10. Blue Cross/Blue Shield spent almost $12.1 million on lobbying in the first half of the year, putting it just ahead of the Pharmaceutical Research and Manufacturers of America (PhRMA), which spent $11.8 million. The American Medical Association was next at $11.3 million, followed by the American Hospital Association at $10.9 million, the center reported on OpenSecrets.org.
After those four, the next-highest health-sector spender was Pfizer, which put up almost $6.2 million in lobbying – good for 18th place for the first half of 2016. The health sector itself was the highest spending of the 121 ranked, taking a $266 million bite out of the total $1.6 billion lobbying pie for the year so far, according to the center’s analysis of data downloaded from the Senate Office of Public Records on Aug. 9.
The perennial leading spender on lobbying, the U.S. Chamber of Commerce, was well ahead of second place, with its $52.3 million more than doubling the $21.4 million spent by the National Association of Realtors. The two groups have finished 1-2 in lobbying spending every year since 2012, and the Chamber of Commerce has been the leading spender since 2001, data on OpenSecrets show.
Four of the 10 highest-spending lobbyers for the first half of 2016 were in the health sector, with Blue Cross/Blue Shield occupying the sector’s top spot by a relatively small margin, according to the Center for Responsive Politics.
The four health-sectors concerns filled spots 3-6 in the overall top 10. Blue Cross/Blue Shield spent almost $12.1 million on lobbying in the first half of the year, putting it just ahead of the Pharmaceutical Research and Manufacturers of America (PhRMA), which spent $11.8 million. The American Medical Association was next at $11.3 million, followed by the American Hospital Association at $10.9 million, the center reported on OpenSecrets.org.
After those four, the next-highest health-sector spender was Pfizer, which put up almost $6.2 million in lobbying – good for 18th place for the first half of 2016. The health sector itself was the highest spending of the 121 ranked, taking a $266 million bite out of the total $1.6 billion lobbying pie for the year so far, according to the center’s analysis of data downloaded from the Senate Office of Public Records on Aug. 9.
The perennial leading spender on lobbying, the U.S. Chamber of Commerce, was well ahead of second place, with its $52.3 million more than doubling the $21.4 million spent by the National Association of Realtors. The two groups have finished 1-2 in lobbying spending every year since 2012, and the Chamber of Commerce has been the leading spender since 2001, data on OpenSecrets show.
WHO updates ranking of critically important antimicrobials
In light of increasing antibiotic resistance among pathogens, the World Health Organization has revised its global rankings of critically important antimicrobials used in human medicine, designating quinolones, third- and fourth-generation cephalosporins, macrolides and ketolides, and glycopeptides as among the highest-priority drugs in the world.
Peter C. Collignon, MBBS, of Canberra (Australia) Hospital and his colleagues on the WHO Advisory Group on Integrated Surveillance of Antimicrobial Resistance, created the rankings for use in developing risk management strategies related to antimicrobial use in food production animals. According to Dr. Collignon and his coauthors, the rankings are intended to help regulators and other stakeholders know which types of antimicrobials used in animals present potentially higher risks to human populations and help inform how this use might be better managed (e.g. restriction to single-animal therapy or prohibition of mass treatment and extra-label use) to minimize the risk of transmission of resistance to the human population.
WHO studies previously suggested that antimicrobials which currently have no veterinary equivalent (for example, carbapenems) “as well as any new class of antimicrobial developed for human therapy should not be used in animals.” Dr. Collignon’s WHO Advisory Group followed two essential criteria to designate antimicrobials of utmost importance to human health in the new study: 1. antimicrobials that are the sole, or one of limited available therapies, to treat serious bacterial infections in people and 2. antimicrobials used to treat infections in people caused by either (a) bacteria that may be transmitted to humans from nonhuman sources or (b) bacteria that may acquire resistance genes from nonhuman sources.
The highest-priority and most critically important antimicrobials are those which meet the criteria listed above and that are used in greatest volume or highest frequency by humans. Another criteria for prioritization involves antimicrobial classes where evidence suggests that the “transmission of resistant bacteria or resistance genes from nonhuman sources is already occurring, or has occurred previously.” Quinolones, third- and fourth-generation cephalosporins, macrolides and ketolides, and glycopeptides were the only antimicrobials that met all criteria for prioritization.
“Antimicrobial resistance remains a threat to human health and drivers of resistance act in all sectors; human, animal, and the environment,” the WHO Advisory Group concluded. “Prioritizing the antimicrobials that are critically important for humans is a valuable and strategic risk-management tool and will be improved with the evidence-based approach which is currently underway.”
Read the full study in Clinical Infectious Diseases (doi: 10.1093/cid/ciw475).
In light of increasing antibiotic resistance among pathogens, the World Health Organization has revised its global rankings of critically important antimicrobials used in human medicine, designating quinolones, third- and fourth-generation cephalosporins, macrolides and ketolides, and glycopeptides as among the highest-priority drugs in the world.
Peter C. Collignon, MBBS, of Canberra (Australia) Hospital and his colleagues on the WHO Advisory Group on Integrated Surveillance of Antimicrobial Resistance, created the rankings for use in developing risk management strategies related to antimicrobial use in food production animals. According to Dr. Collignon and his coauthors, the rankings are intended to help regulators and other stakeholders know which types of antimicrobials used in animals present potentially higher risks to human populations and help inform how this use might be better managed (e.g. restriction to single-animal therapy or prohibition of mass treatment and extra-label use) to minimize the risk of transmission of resistance to the human population.
WHO studies previously suggested that antimicrobials which currently have no veterinary equivalent (for example, carbapenems) “as well as any new class of antimicrobial developed for human therapy should not be used in animals.” Dr. Collignon’s WHO Advisory Group followed two essential criteria to designate antimicrobials of utmost importance to human health in the new study: 1. antimicrobials that are the sole, or one of limited available therapies, to treat serious bacterial infections in people and 2. antimicrobials used to treat infections in people caused by either (a) bacteria that may be transmitted to humans from nonhuman sources or (b) bacteria that may acquire resistance genes from nonhuman sources.
The highest-priority and most critically important antimicrobials are those which meet the criteria listed above and that are used in greatest volume or highest frequency by humans. Another criteria for prioritization involves antimicrobial classes where evidence suggests that the “transmission of resistant bacteria or resistance genes from nonhuman sources is already occurring, or has occurred previously.” Quinolones, third- and fourth-generation cephalosporins, macrolides and ketolides, and glycopeptides were the only antimicrobials that met all criteria for prioritization.
“Antimicrobial resistance remains a threat to human health and drivers of resistance act in all sectors; human, animal, and the environment,” the WHO Advisory Group concluded. “Prioritizing the antimicrobials that are critically important for humans is a valuable and strategic risk-management tool and will be improved with the evidence-based approach which is currently underway.”
Read the full study in Clinical Infectious Diseases (doi: 10.1093/cid/ciw475).
In light of increasing antibiotic resistance among pathogens, the World Health Organization has revised its global rankings of critically important antimicrobials used in human medicine, designating quinolones, third- and fourth-generation cephalosporins, macrolides and ketolides, and glycopeptides as among the highest-priority drugs in the world.
Peter C. Collignon, MBBS, of Canberra (Australia) Hospital and his colleagues on the WHO Advisory Group on Integrated Surveillance of Antimicrobial Resistance, created the rankings for use in developing risk management strategies related to antimicrobial use in food production animals. According to Dr. Collignon and his coauthors, the rankings are intended to help regulators and other stakeholders know which types of antimicrobials used in animals present potentially higher risks to human populations and help inform how this use might be better managed (e.g. restriction to single-animal therapy or prohibition of mass treatment and extra-label use) to minimize the risk of transmission of resistance to the human population.
WHO studies previously suggested that antimicrobials which currently have no veterinary equivalent (for example, carbapenems) “as well as any new class of antimicrobial developed for human therapy should not be used in animals.” Dr. Collignon’s WHO Advisory Group followed two essential criteria to designate antimicrobials of utmost importance to human health in the new study: 1. antimicrobials that are the sole, or one of limited available therapies, to treat serious bacterial infections in people and 2. antimicrobials used to treat infections in people caused by either (a) bacteria that may be transmitted to humans from nonhuman sources or (b) bacteria that may acquire resistance genes from nonhuman sources.
The highest-priority and most critically important antimicrobials are those which meet the criteria listed above and that are used in greatest volume or highest frequency by humans. Another criteria for prioritization involves antimicrobial classes where evidence suggests that the “transmission of resistant bacteria or resistance genes from nonhuman sources is already occurring, or has occurred previously.” Quinolones, third- and fourth-generation cephalosporins, macrolides and ketolides, and glycopeptides were the only antimicrobials that met all criteria for prioritization.
“Antimicrobial resistance remains a threat to human health and drivers of resistance act in all sectors; human, animal, and the environment,” the WHO Advisory Group concluded. “Prioritizing the antimicrobials that are critically important for humans is a valuable and strategic risk-management tool and will be improved with the evidence-based approach which is currently underway.”
Read the full study in Clinical Infectious Diseases (doi: 10.1093/cid/ciw475).
FROM CLINICAL INFECTIOUS DISEASES
ANTARCTIC results chill enthusiasm for platelet monitoring
ROME – Measuring platelet function in order to tailor antiplatelet therapy in elderly patients undergoing percutaneous coronary intervention for acute coronary syndromes did not improve their clinical outcomes in the randomized ANTARCTIC trial, Gilles Montalescot, MD, reported at the annual congress of the European Society of Cardiology.
“We found absolutely no benefit for this strategy of adjustment of antiplatelet therapy based upon platelet function testing. The study was completely neutral on all types of endpoints, ischemic as well as bleeding,” said Dr. Montalescot, professor of cardiology at the University of Paris VI and director of the cardiac care unit at Pitié-Salpêtrière Hospital.
This was a disappointing result in what was the largest-ever randomized clinical trial involving PCI in elderly patients, he said. This was a high-risk population, not only by virtue of everyone being over age 75 years, but because they all presented with ACS. Indeed, one-third of ANTARCTIC participants underwent primary PCI for ST-segment elevation myocardial infarction.
ANTARCTIC (Assessment of a Normal Versus Tailored Dose of Prasugrel After Stenting in Patients Aged Over 75 Years to Reduce the Composite of Bleeding, Stent Thrombosis, and Ischemic Complications) was carried out as a follow-up to the earlier ARCTIC randomized trial, also conducted by Dr. Montalescot and his coinvestigators. Like ANTARCTIC, ARCTIC, too, showed no clinical benefit for platelet function testing in order to adjust antiplatelet therapy (N Engl J Med. 2012;367:2100-9). At the time, ARCTIC’s critics argued that this individualized strategy didn’t achieve the expected improved outcomes because the trial was conducted in low-risk, stable patients undergoing elective scheduled PCI. In contrast, if there was ever a high-risk population in which platelet function testing and tailored antiplatelet therapy should work, it was in the very high-risk ANTARCTIC population, he said.
ANTARCTIC included 877 elderly patients undergoing urgent PCI for ACS who were placed on low-dose aspirin and randomized to standard antiplatelet therapy with prasugrel (Effient) at 5 mg/day, the European approved dose for long-term maintenance therapy in elderly patients, or to tailored antiplatelet therapy.
Patients in the tailored therapy arm received prasugrel at 5 mg/day for the first 14 days, then underwent platelet function testing with the VerifyNow P2Y12 system. If they demonstrated high on-drug platelet activity, defined as at least 208 P2Y12 reaction units (PRU), their prasugrel was bumped up to 10 mg/day. If their PRU measurement was in what is considered the optimal range for quelling ischemia without promoting bleeding – that is, less than 208 but more than 85 PRU – they remained on prasugrel at 5 mg/day. And if they scored less than 85 PRU, exposing them to excess bleeding risk due to high suppression of platelets, they were switched to clopidogrel (Plavix) at 75 mg/day, a less potent antiplatelet regimen.
Two weeks after their first platelet function measurement, participants in the tailored therapy arm returned for a second round of platelet activity testing, with their antiplatelet regimen once again being adjusted on the basis of the results.
The primary study endpoint was net clinical benefit over a 12-month follow-up period. This was defined as the composite of cardiovascular death, MI, stroke, urgent revascularization, stent thrombosis, and Bleeding Academic Research Consortium (BARC) types 2, 3, or 5. This composite endpoint occurred in 27.6% of the platelet monitoring group and a near-identical 27.8% of conventionally managed patients.
Of note, 42% of patients in the actively monitored group were within the target platelet inhibition range when tested 14 days into the study. At study’s end, 55% of patients remained on prasugrel at 5 mg/day, 39% were on clopidogrel at 75 mg/day, and less than 4% were on prasugrel at 10 mg/day. Thus, most patients who underwent a dose adjustment on the basis of their VerifyNow results were downgraded to a less-potent antiplatelet regimen. Very few required enhanced platelet suppression in the form of 10 mg/day of prasugrel.
“Platelet function monitoring is difficult to use. Patients have to come back twice to be monitored. It’s costly. It’s time consuming. And platelet function monitoring clearly does not help,” the cardiologist said.
The ANTARCTIC results will likely lead to a revision of the American and European guidelines, which currently give a class IIb/level of evidence C recommendation for platelet function testing in high-risk situations.
“There is a huge literature showing that platelet reactivity affects clinical outcomes,” Dr. Montalescot continued. “One hypothesis now is that platelet reactivity may be only a marker of risk; you can modify it, but that has no impact on patient outcomes. We may be in the same situation here as with HDL cholesterol, for example.”
Discussant Steen Dalby Kristensen, MD, noted that ANTARCTIC is just the latest in a slew of negative randomized clinical trials of individualized antiplatelet therapy for coronary artery disease. In addition to ARCTIC, others include GRAVITAS, TRIGGER PCI, and ASCET. One study, the German/Austrian TROPICAL ACS trial, remains ongoing.
“It really is an intriguing concept that many of us have been fascinated by for years: to identify the sweet spot where, by measuring platelet aggregation and maybe changing the therapy, we can find just the right balance between bleeding and ischemia. The ANTARCTIC results are quite disappointing for platelet-monitoring enthusiasts. Is the whole concept wrong?” said Dr. Kristensen, professor of cardiology and head of the cardiovascular research center at Aarhus (Denmark) University Hospital.
“I think even more disappointing for me than the lack of impact on ischemic events was the bleeding. I would have anticipated that maybe bleeding could be avoided by adjusting the dose, but this was not the case,” he added.
But Stephan Gielen, MD, saw a silver lining in the negative results for ANTARCTIC.
“From my perspective as a clinical interventionalist, I’m happy that you ended up in the way you did. Putting things positively, this study confirms the safety of dual-platelet inhibition with prasugrel at the reduced dose of 5 mg in an elderly population. There is no need to go to the trouble of monitoring platelet function even in this elderly population, which I think for clinical practice is a good message,” said Dr. Gielen of Detmold (Germany) Hospital, who cochaired a press conference where Dr. Montalescot presented the ANTARCTIC results.
Simultaneously with Dr. Montalescot’s presentation at ESC 2016 in Rome, the ANTARCTIC results were published online (Lancet. 2016 Aug 26. doi: 10.1016/S0140-6736(16)31323-X).
ANTARCTIC was funded by Eli Lilly, Daiichi Sankyo, Stentys, Accriva Diagnostics, Medtronic, and the French Foundation for Heart Research. The presenter reported receiving research grants from and/or serving as a consultant to those organizations and numerous others.
ROME – Measuring platelet function in order to tailor antiplatelet therapy in elderly patients undergoing percutaneous coronary intervention for acute coronary syndromes did not improve their clinical outcomes in the randomized ANTARCTIC trial, Gilles Montalescot, MD, reported at the annual congress of the European Society of Cardiology.
“We found absolutely no benefit for this strategy of adjustment of antiplatelet therapy based upon platelet function testing. The study was completely neutral on all types of endpoints, ischemic as well as bleeding,” said Dr. Montalescot, professor of cardiology at the University of Paris VI and director of the cardiac care unit at Pitié-Salpêtrière Hospital.
This was a disappointing result in what was the largest-ever randomized clinical trial involving PCI in elderly patients, he said. This was a high-risk population, not only by virtue of everyone being over age 75 years, but because they all presented with ACS. Indeed, one-third of ANTARCTIC participants underwent primary PCI for ST-segment elevation myocardial infarction.
ANTARCTIC (Assessment of a Normal Versus Tailored Dose of Prasugrel After Stenting in Patients Aged Over 75 Years to Reduce the Composite of Bleeding, Stent Thrombosis, and Ischemic Complications) was carried out as a follow-up to the earlier ARCTIC randomized trial, also conducted by Dr. Montalescot and his coinvestigators. Like ANTARCTIC, ARCTIC, too, showed no clinical benefit for platelet function testing in order to adjust antiplatelet therapy (N Engl J Med. 2012;367:2100-9). At the time, ARCTIC’s critics argued that this individualized strategy didn’t achieve the expected improved outcomes because the trial was conducted in low-risk, stable patients undergoing elective scheduled PCI. In contrast, if there was ever a high-risk population in which platelet function testing and tailored antiplatelet therapy should work, it was in the very high-risk ANTARCTIC population, he said.
ANTARCTIC included 877 elderly patients undergoing urgent PCI for ACS who were placed on low-dose aspirin and randomized to standard antiplatelet therapy with prasugrel (Effient) at 5 mg/day, the European approved dose for long-term maintenance therapy in elderly patients, or to tailored antiplatelet therapy.
Patients in the tailored therapy arm received prasugrel at 5 mg/day for the first 14 days, then underwent platelet function testing with the VerifyNow P2Y12 system. If they demonstrated high on-drug platelet activity, defined as at least 208 P2Y12 reaction units (PRU), their prasugrel was bumped up to 10 mg/day. If their PRU measurement was in what is considered the optimal range for quelling ischemia without promoting bleeding – that is, less than 208 but more than 85 PRU – they remained on prasugrel at 5 mg/day. And if they scored less than 85 PRU, exposing them to excess bleeding risk due to high suppression of platelets, they were switched to clopidogrel (Plavix) at 75 mg/day, a less potent antiplatelet regimen.
Two weeks after their first platelet function measurement, participants in the tailored therapy arm returned for a second round of platelet activity testing, with their antiplatelet regimen once again being adjusted on the basis of the results.
The primary study endpoint was net clinical benefit over a 12-month follow-up period. This was defined as the composite of cardiovascular death, MI, stroke, urgent revascularization, stent thrombosis, and Bleeding Academic Research Consortium (BARC) types 2, 3, or 5. This composite endpoint occurred in 27.6% of the platelet monitoring group and a near-identical 27.8% of conventionally managed patients.
Of note, 42% of patients in the actively monitored group were within the target platelet inhibition range when tested 14 days into the study. At study’s end, 55% of patients remained on prasugrel at 5 mg/day, 39% were on clopidogrel at 75 mg/day, and less than 4% were on prasugrel at 10 mg/day. Thus, most patients who underwent a dose adjustment on the basis of their VerifyNow results were downgraded to a less-potent antiplatelet regimen. Very few required enhanced platelet suppression in the form of 10 mg/day of prasugrel.
“Platelet function monitoring is difficult to use. Patients have to come back twice to be monitored. It’s costly. It’s time consuming. And platelet function monitoring clearly does not help,” the cardiologist said.
The ANTARCTIC results will likely lead to a revision of the American and European guidelines, which currently give a class IIb/level of evidence C recommendation for platelet function testing in high-risk situations.
“There is a huge literature showing that platelet reactivity affects clinical outcomes,” Dr. Montalescot continued. “One hypothesis now is that platelet reactivity may be only a marker of risk; you can modify it, but that has no impact on patient outcomes. We may be in the same situation here as with HDL cholesterol, for example.”
Discussant Steen Dalby Kristensen, MD, noted that ANTARCTIC is just the latest in a slew of negative randomized clinical trials of individualized antiplatelet therapy for coronary artery disease. In addition to ARCTIC, others include GRAVITAS, TRIGGER PCI, and ASCET. One study, the German/Austrian TROPICAL ACS trial, remains ongoing.
“It really is an intriguing concept that many of us have been fascinated by for years: to identify the sweet spot where, by measuring platelet aggregation and maybe changing the therapy, we can find just the right balance between bleeding and ischemia. The ANTARCTIC results are quite disappointing for platelet-monitoring enthusiasts. Is the whole concept wrong?” said Dr. Kristensen, professor of cardiology and head of the cardiovascular research center at Aarhus (Denmark) University Hospital.
“I think even more disappointing for me than the lack of impact on ischemic events was the bleeding. I would have anticipated that maybe bleeding could be avoided by adjusting the dose, but this was not the case,” he added.
But Stephan Gielen, MD, saw a silver lining in the negative results for ANTARCTIC.
“From my perspective as a clinical interventionalist, I’m happy that you ended up in the way you did. Putting things positively, this study confirms the safety of dual-platelet inhibition with prasugrel at the reduced dose of 5 mg in an elderly population. There is no need to go to the trouble of monitoring platelet function even in this elderly population, which I think for clinical practice is a good message,” said Dr. Gielen of Detmold (Germany) Hospital, who cochaired a press conference where Dr. Montalescot presented the ANTARCTIC results.
Simultaneously with Dr. Montalescot’s presentation at ESC 2016 in Rome, the ANTARCTIC results were published online (Lancet. 2016 Aug 26. doi: 10.1016/S0140-6736(16)31323-X).
ANTARCTIC was funded by Eli Lilly, Daiichi Sankyo, Stentys, Accriva Diagnostics, Medtronic, and the French Foundation for Heart Research. The presenter reported receiving research grants from and/or serving as a consultant to those organizations and numerous others.
ROME – Measuring platelet function in order to tailor antiplatelet therapy in elderly patients undergoing percutaneous coronary intervention for acute coronary syndromes did not improve their clinical outcomes in the randomized ANTARCTIC trial, Gilles Montalescot, MD, reported at the annual congress of the European Society of Cardiology.
“We found absolutely no benefit for this strategy of adjustment of antiplatelet therapy based upon platelet function testing. The study was completely neutral on all types of endpoints, ischemic as well as bleeding,” said Dr. Montalescot, professor of cardiology at the University of Paris VI and director of the cardiac care unit at Pitié-Salpêtrière Hospital.
This was a disappointing result in what was the largest-ever randomized clinical trial involving PCI in elderly patients, he said. This was a high-risk population, not only by virtue of everyone being over age 75 years, but because they all presented with ACS. Indeed, one-third of ANTARCTIC participants underwent primary PCI for ST-segment elevation myocardial infarction.
ANTARCTIC (Assessment of a Normal Versus Tailored Dose of Prasugrel After Stenting in Patients Aged Over 75 Years to Reduce the Composite of Bleeding, Stent Thrombosis, and Ischemic Complications) was carried out as a follow-up to the earlier ARCTIC randomized trial, also conducted by Dr. Montalescot and his coinvestigators. Like ANTARCTIC, ARCTIC, too, showed no clinical benefit for platelet function testing in order to adjust antiplatelet therapy (N Engl J Med. 2012;367:2100-9). At the time, ARCTIC’s critics argued that this individualized strategy didn’t achieve the expected improved outcomes because the trial was conducted in low-risk, stable patients undergoing elective scheduled PCI. In contrast, if there was ever a high-risk population in which platelet function testing and tailored antiplatelet therapy should work, it was in the very high-risk ANTARCTIC population, he said.
ANTARCTIC included 877 elderly patients undergoing urgent PCI for ACS who were placed on low-dose aspirin and randomized to standard antiplatelet therapy with prasugrel (Effient) at 5 mg/day, the European approved dose for long-term maintenance therapy in elderly patients, or to tailored antiplatelet therapy.
Patients in the tailored therapy arm received prasugrel at 5 mg/day for the first 14 days, then underwent platelet function testing with the VerifyNow P2Y12 system. If they demonstrated high on-drug platelet activity, defined as at least 208 P2Y12 reaction units (PRU), their prasugrel was bumped up to 10 mg/day. If their PRU measurement was in what is considered the optimal range for quelling ischemia without promoting bleeding – that is, less than 208 but more than 85 PRU – they remained on prasugrel at 5 mg/day. And if they scored less than 85 PRU, exposing them to excess bleeding risk due to high suppression of platelets, they were switched to clopidogrel (Plavix) at 75 mg/day, a less potent antiplatelet regimen.
Two weeks after their first platelet function measurement, participants in the tailored therapy arm returned for a second round of platelet activity testing, with their antiplatelet regimen once again being adjusted on the basis of the results.
The primary study endpoint was net clinical benefit over a 12-month follow-up period. This was defined as the composite of cardiovascular death, MI, stroke, urgent revascularization, stent thrombosis, and Bleeding Academic Research Consortium (BARC) types 2, 3, or 5. This composite endpoint occurred in 27.6% of the platelet monitoring group and a near-identical 27.8% of conventionally managed patients.
Of note, 42% of patients in the actively monitored group were within the target platelet inhibition range when tested 14 days into the study. At study’s end, 55% of patients remained on prasugrel at 5 mg/day, 39% were on clopidogrel at 75 mg/day, and less than 4% were on prasugrel at 10 mg/day. Thus, most patients who underwent a dose adjustment on the basis of their VerifyNow results were downgraded to a less-potent antiplatelet regimen. Very few required enhanced platelet suppression in the form of 10 mg/day of prasugrel.
“Platelet function monitoring is difficult to use. Patients have to come back twice to be monitored. It’s costly. It’s time consuming. And platelet function monitoring clearly does not help,” the cardiologist said.
The ANTARCTIC results will likely lead to a revision of the American and European guidelines, which currently give a class IIb/level of evidence C recommendation for platelet function testing in high-risk situations.
“There is a huge literature showing that platelet reactivity affects clinical outcomes,” Dr. Montalescot continued. “One hypothesis now is that platelet reactivity may be only a marker of risk; you can modify it, but that has no impact on patient outcomes. We may be in the same situation here as with HDL cholesterol, for example.”
Discussant Steen Dalby Kristensen, MD, noted that ANTARCTIC is just the latest in a slew of negative randomized clinical trials of individualized antiplatelet therapy for coronary artery disease. In addition to ARCTIC, others include GRAVITAS, TRIGGER PCI, and ASCET. One study, the German/Austrian TROPICAL ACS trial, remains ongoing.
“It really is an intriguing concept that many of us have been fascinated by for years: to identify the sweet spot where, by measuring platelet aggregation and maybe changing the therapy, we can find just the right balance between bleeding and ischemia. The ANTARCTIC results are quite disappointing for platelet-monitoring enthusiasts. Is the whole concept wrong?” said Dr. Kristensen, professor of cardiology and head of the cardiovascular research center at Aarhus (Denmark) University Hospital.
“I think even more disappointing for me than the lack of impact on ischemic events was the bleeding. I would have anticipated that maybe bleeding could be avoided by adjusting the dose, but this was not the case,” he added.
But Stephan Gielen, MD, saw a silver lining in the negative results for ANTARCTIC.
“From my perspective as a clinical interventionalist, I’m happy that you ended up in the way you did. Putting things positively, this study confirms the safety of dual-platelet inhibition with prasugrel at the reduced dose of 5 mg in an elderly population. There is no need to go to the trouble of monitoring platelet function even in this elderly population, which I think for clinical practice is a good message,” said Dr. Gielen of Detmold (Germany) Hospital, who cochaired a press conference where Dr. Montalescot presented the ANTARCTIC results.
Simultaneously with Dr. Montalescot’s presentation at ESC 2016 in Rome, the ANTARCTIC results were published online (Lancet. 2016 Aug 26. doi: 10.1016/S0140-6736(16)31323-X).
ANTARCTIC was funded by Eli Lilly, Daiichi Sankyo, Stentys, Accriva Diagnostics, Medtronic, and the French Foundation for Heart Research. The presenter reported receiving research grants from and/or serving as a consultant to those organizations and numerous others.
AT THE ESC CONGRESS 2016
Key clinical point: Researchers have just about given up on the notion that monitoring platelet function in order to individualize antiplatelet therapy in patients undergoing PCI provides any clinical benefit.
Major finding: Individualized antiplatelet therapy based upon serial measurements of platelet function did not result in improved outcomes in elderly patients undergoing PCI for acute coronary syndrome (27.6% in the platelet monitoring group and 27.8% in conventionally managed patients).
Data source: ANTARCTIC was an open-label, blinded-endpoint randomized trial of tailored versus standard antiplatelet therapy in 877 elderly patients undergoing PCI for ACS.
Disclosures: ANTARCTIC was funded by Eli Lilly, Daiichi Sankyo, Stentys, Accriva Diagnostics, Medtronic, and the French Foundation for Heart Research. The presenter reported receiving research grants from and/or serving as a consultant to those organizations and numerous others.