User login
Official Newspaper of the American College of Surgeons
Legislators: Investigate Medicare fraud before paying doctors
Republican leaders in Congress are calling on CMS to impose stricter safeguards against fraudulent Medicare billing by physicians.
The chairmen of the Senate Finance Committee, the House Ways and Means and Energy and Commerce Committees, and the chairmen of key House subcommittees said that the Centers for Medicare & Medicaid Services relies too heavily on the “outdated” pay and chase method and should focus more energy on preventing payment for potential fraudulent claims.
“Improper payments remain an enormous problem for the Medicare program,” the chairmen wrote in a Sept. 12 letter to Acting CMS Administrator Andy Slavitt. “In 2015, the Medicare program had an error rate of 12.1% or $43.3 billion dollars. The billions of dollars lost to Medicare fraud each year underscore the importance of stopping potentially fraudulent payments before they’re made,”
Some health law experts, however, argue that CMS already has a process for in place for pre-identifying inaccurate claims via prepayment audits and reviews. Such efforts can be devastating for physicians who come under scrutiny for unintentional mistakes, said Daniel F. Shay, a Philadelphia health law attorney.
“I can understand why, particularly in an election year, elected officials might send a letter reiterating the need to curb ‘waste, fraud, and abuse,’” Mr. Shay said in an interview. “It’s true that it’s more efficient for the government to investigate a physician’s claim for reimbursement first, and then pay. However, I think we have to take into account the physicians’ perspective, especially physicians in smaller, independent practices.”
The legislators’ letter acknowledges that CMS has taken some proactive steps to prevent health fraud, including creation of the Fraud Prevention System (FPS), which highlights questionable billing patterns and identifies providers who pose high risk to the program. FPS runs analytics on 4.5 million claims daily and has led to more than $820 million in savings, according to CMS. However, legislators said they are still concerned that CMS too often pays claims before investigating whether they’re false. The letter requests that CMS clarify its implementation of the FPS program, including details on fraud investigations and how the agency monitors FPS’s effectiveness.
Houston, Tex.–based health law attorney Michael E. Clark disagrees that CMS is overusing the pay-and-chase method. Quite the contrary, he said.
“The federal government cannot seem to find the right balance on how to address program fraud,” Mr. Clark said in an interview. “While ‘pay and chase’ once was a problem, now the government can effectively destroy health care service providers under a very low threshold without the businesses having a meaningful right to appeal that determination.”
Specifically, CMS can withhold Medicare reimbursement from health providers under an amended 2011 law that permits payments to be suppressed when “credible” allegations of fraud have been made, but are disputed. The term “credible” is a new, lower standard for the administrative action, which was meant to address the pay-and-chase problem, Mr. Clark said. The law defines a “credible allegation of fraud” as an allegation from any source, including but not limited to fraud hotline complaints, data mining of claims, patterns identified through provider audits, civil false claims cases, and law enforcement investigations.
“That standard is easy to meet and agencies have every incentive to claim they’ve got so-called credible allegations of fraud in order to avoid being criticized later on for not preventing the monies from being dissipated,” he said. Because the law precludes health providers from appealing the fraud allegation to a federal court until all administrative remedies have been exhausted, “a health care services provider can quickly be put out of business, even if it turns out that the investigation proves not to be actionable.”
Prepayment reviews of claims can drag on for months, severely impacting a physician’s income, Mr. Shay added. In his experience, the majority of physicians under investigation are not trying to game the system, but rather don’t understand all of the administrative requirements related to filing claims. In some cases, the physicians’ notes are not complete, their bills are too high for services provided, or not enough documentation exists to support medical necessity.
“In the midst of that, you have doctors who are likely well-meaning, who have provided a service to a patient in need, and who are facing real economic hardship without an effective mechanism to challenge or end the prepayment review process,” he said.
Rather than more prepayment investigations, Mr. Shay would like to see CMS focus on physician education.
There needs to be “more emphasis on provider education in terms of compliance with program requirements,” he said. “It shouldn’t require a lawyer getting involved to find out what specifically [CMS] wants them to do. That should be part of the process as a standard.”
On Twitter @legal_med
Republican leaders in Congress are calling on CMS to impose stricter safeguards against fraudulent Medicare billing by physicians.
The chairmen of the Senate Finance Committee, the House Ways and Means and Energy and Commerce Committees, and the chairmen of key House subcommittees said that the Centers for Medicare & Medicaid Services relies too heavily on the “outdated” pay and chase method and should focus more energy on preventing payment for potential fraudulent claims.
“Improper payments remain an enormous problem for the Medicare program,” the chairmen wrote in a Sept. 12 letter to Acting CMS Administrator Andy Slavitt. “In 2015, the Medicare program had an error rate of 12.1% or $43.3 billion dollars. The billions of dollars lost to Medicare fraud each year underscore the importance of stopping potentially fraudulent payments before they’re made,”
Some health law experts, however, argue that CMS already has a process for in place for pre-identifying inaccurate claims via prepayment audits and reviews. Such efforts can be devastating for physicians who come under scrutiny for unintentional mistakes, said Daniel F. Shay, a Philadelphia health law attorney.
“I can understand why, particularly in an election year, elected officials might send a letter reiterating the need to curb ‘waste, fraud, and abuse,’” Mr. Shay said in an interview. “It’s true that it’s more efficient for the government to investigate a physician’s claim for reimbursement first, and then pay. However, I think we have to take into account the physicians’ perspective, especially physicians in smaller, independent practices.”
The legislators’ letter acknowledges that CMS has taken some proactive steps to prevent health fraud, including creation of the Fraud Prevention System (FPS), which highlights questionable billing patterns and identifies providers who pose high risk to the program. FPS runs analytics on 4.5 million claims daily and has led to more than $820 million in savings, according to CMS. However, legislators said they are still concerned that CMS too often pays claims before investigating whether they’re false. The letter requests that CMS clarify its implementation of the FPS program, including details on fraud investigations and how the agency monitors FPS’s effectiveness.
Houston, Tex.–based health law attorney Michael E. Clark disagrees that CMS is overusing the pay-and-chase method. Quite the contrary, he said.
“The federal government cannot seem to find the right balance on how to address program fraud,” Mr. Clark said in an interview. “While ‘pay and chase’ once was a problem, now the government can effectively destroy health care service providers under a very low threshold without the businesses having a meaningful right to appeal that determination.”
Specifically, CMS can withhold Medicare reimbursement from health providers under an amended 2011 law that permits payments to be suppressed when “credible” allegations of fraud have been made, but are disputed. The term “credible” is a new, lower standard for the administrative action, which was meant to address the pay-and-chase problem, Mr. Clark said. The law defines a “credible allegation of fraud” as an allegation from any source, including but not limited to fraud hotline complaints, data mining of claims, patterns identified through provider audits, civil false claims cases, and law enforcement investigations.
“That standard is easy to meet and agencies have every incentive to claim they’ve got so-called credible allegations of fraud in order to avoid being criticized later on for not preventing the monies from being dissipated,” he said. Because the law precludes health providers from appealing the fraud allegation to a federal court until all administrative remedies have been exhausted, “a health care services provider can quickly be put out of business, even if it turns out that the investigation proves not to be actionable.”
Prepayment reviews of claims can drag on for months, severely impacting a physician’s income, Mr. Shay added. In his experience, the majority of physicians under investigation are not trying to game the system, but rather don’t understand all of the administrative requirements related to filing claims. In some cases, the physicians’ notes are not complete, their bills are too high for services provided, or not enough documentation exists to support medical necessity.
“In the midst of that, you have doctors who are likely well-meaning, who have provided a service to a patient in need, and who are facing real economic hardship without an effective mechanism to challenge or end the prepayment review process,” he said.
Rather than more prepayment investigations, Mr. Shay would like to see CMS focus on physician education.
There needs to be “more emphasis on provider education in terms of compliance with program requirements,” he said. “It shouldn’t require a lawyer getting involved to find out what specifically [CMS] wants them to do. That should be part of the process as a standard.”
On Twitter @legal_med
Republican leaders in Congress are calling on CMS to impose stricter safeguards against fraudulent Medicare billing by physicians.
The chairmen of the Senate Finance Committee, the House Ways and Means and Energy and Commerce Committees, and the chairmen of key House subcommittees said that the Centers for Medicare & Medicaid Services relies too heavily on the “outdated” pay and chase method and should focus more energy on preventing payment for potential fraudulent claims.
“Improper payments remain an enormous problem for the Medicare program,” the chairmen wrote in a Sept. 12 letter to Acting CMS Administrator Andy Slavitt. “In 2015, the Medicare program had an error rate of 12.1% or $43.3 billion dollars. The billions of dollars lost to Medicare fraud each year underscore the importance of stopping potentially fraudulent payments before they’re made,”
Some health law experts, however, argue that CMS already has a process for in place for pre-identifying inaccurate claims via prepayment audits and reviews. Such efforts can be devastating for physicians who come under scrutiny for unintentional mistakes, said Daniel F. Shay, a Philadelphia health law attorney.
“I can understand why, particularly in an election year, elected officials might send a letter reiterating the need to curb ‘waste, fraud, and abuse,’” Mr. Shay said in an interview. “It’s true that it’s more efficient for the government to investigate a physician’s claim for reimbursement first, and then pay. However, I think we have to take into account the physicians’ perspective, especially physicians in smaller, independent practices.”
The legislators’ letter acknowledges that CMS has taken some proactive steps to prevent health fraud, including creation of the Fraud Prevention System (FPS), which highlights questionable billing patterns and identifies providers who pose high risk to the program. FPS runs analytics on 4.5 million claims daily and has led to more than $820 million in savings, according to CMS. However, legislators said they are still concerned that CMS too often pays claims before investigating whether they’re false. The letter requests that CMS clarify its implementation of the FPS program, including details on fraud investigations and how the agency monitors FPS’s effectiveness.
Houston, Tex.–based health law attorney Michael E. Clark disagrees that CMS is overusing the pay-and-chase method. Quite the contrary, he said.
“The federal government cannot seem to find the right balance on how to address program fraud,” Mr. Clark said in an interview. “While ‘pay and chase’ once was a problem, now the government can effectively destroy health care service providers under a very low threshold without the businesses having a meaningful right to appeal that determination.”
Specifically, CMS can withhold Medicare reimbursement from health providers under an amended 2011 law that permits payments to be suppressed when “credible” allegations of fraud have been made, but are disputed. The term “credible” is a new, lower standard for the administrative action, which was meant to address the pay-and-chase problem, Mr. Clark said. The law defines a “credible allegation of fraud” as an allegation from any source, including but not limited to fraud hotline complaints, data mining of claims, patterns identified through provider audits, civil false claims cases, and law enforcement investigations.
“That standard is easy to meet and agencies have every incentive to claim they’ve got so-called credible allegations of fraud in order to avoid being criticized later on for not preventing the monies from being dissipated,” he said. Because the law precludes health providers from appealing the fraud allegation to a federal court until all administrative remedies have been exhausted, “a health care services provider can quickly be put out of business, even if it turns out that the investigation proves not to be actionable.”
Prepayment reviews of claims can drag on for months, severely impacting a physician’s income, Mr. Shay added. In his experience, the majority of physicians under investigation are not trying to game the system, but rather don’t understand all of the administrative requirements related to filing claims. In some cases, the physicians’ notes are not complete, their bills are too high for services provided, or not enough documentation exists to support medical necessity.
“In the midst of that, you have doctors who are likely well-meaning, who have provided a service to a patient in need, and who are facing real economic hardship without an effective mechanism to challenge or end the prepayment review process,” he said.
Rather than more prepayment investigations, Mr. Shay would like to see CMS focus on physician education.
There needs to be “more emphasis on provider education in terms of compliance with program requirements,” he said. “It shouldn’t require a lawyer getting involved to find out what specifically [CMS] wants them to do. That should be part of the process as a standard.”
On Twitter @legal_med
Guideline recommends optimal periop management of geriatric patients
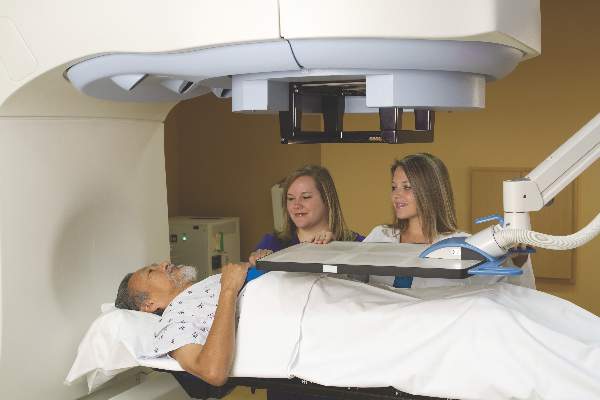
SAN DIEGO – As the number of surgery patients over the age of 65 continues to burgeon, clinicians have a resource to help them provide optimal perioperative care to this patient population.
At the American College of Surgeons/National Surgical Quality Improvement Program National Conference, Ronnie A. Rosenthal, MD, discussed highlights from “Optimal Perioperative Management of the Geriatric Patient: A Best Practice Guideline from the ACS NSQIP/American Geriatrics Society,” which was published in January 2016.
Work on the guideline began in 2013, when a 28-member multidisciplinary panel began to conduct a structured search of Medline to identify systematic reviews, meta-analyses, practice guidelines, and clinical trials on the topic. The panel included experts from ACS, the ACS Geriatric Surgery Task Force, the American Society of Anesthesiologists, the American Geriatrics Society, and the AGS’ Geriatrics for Specialists Initiative. The 61-page document is divided into four categories: immediate preoperative period, intraoperative management, postoperative care, and care transitions.
Working with patients on goals
As noted in the guideline, a primary goal of the immediate preoperative period is to discuss with the patient his or her goals and expectations. Patient expectations are influenced by their treatment preferences. In fact, researchers have found that older patients are less likely to want a treatment – even if it results in cure – that may result in severe functional or cognitive impairment. For patients with existing advanced directives, organizations representing nurses, anesthesiologists, and surgeons all agree that there must be a “reconsideration” of these directives prior to surgery. A discussion that includes the new risks of the procedure must be conducted to ensure that the approach to potential life-threatening problems is consistent with the patient’s values.
Preoperative management of medications
Another recommendation for the preoperative period is to ensure that older patients have shorter fasts, have appropriate prophylactic antibiotics, continue medications with withdrawal potential, and discontinue medications that are not essential. The latter point is based on the Beers Criteria, a list of medications that are inappropriate or potentially inappropriate to use in older adults (J Am Geriatr Soc. 2015 Nov;63[11]:2227-46). “You want to discontinue as many inappropriate medications as possible, because one of the main side effects of their use is delirium, and you want to avoid that,” said Dr. Rosenthal, professor of surgery at the Yale University, New Haven, Conn., and one of the guideline authors.
Anesthesia and pain management
Intraoperative management strategies contained in the guideline include establishing an anesthetic approach and a perioperative analgesia pain plan, preventing postoperative nausea and vomiting, assessing patient safety in the OR, preventing predictable complications, and optimizing fluid management. Physiologic effects of anesthesia medications include changes in systemic vascular resistance, cardiac preload, baroreceptor responses, lung mechanics, oxygen diffusion, neurotransmitter function, and end-organ blood flow, among others. “These physiologic changes of aging have significant clinical implications,” Dr. Rosenthal noted. “These are variable among individuals and variable among organ systems, and it’s important that we pay attention to that. Because of this variability, there is insufficient evidence to recommend a single ‘best’ anesthetic plan for all older adults.”
The guideline recommends that each patient have an individualized pain plan that consists of a directed pain history and physical exam and is appropriately titrated for increased sensitivity. “It should include a prophylactic bowel regimen for anybody who’s on an opioid in particular,” she said. “We should avoid inappropriate medications like benzodiazepines, and we should use a multimodal therapy with opioid-sparing and regional techniques.”
Pulmonary considerations for anesthesia include susceptibility to hypocarbia and hypoxemia, and susceptibility to residual anesthetic effects. “Because of physiologic changes, the anesthesia medications aren’t metabolized in the same way,” she said. “Older people may have lower drug requirements and may not recover as quickly from the effects of these drugs. This can lead to respiratory compromise and also can increase the risk of aspiration.” Strategies to prevent pulmonary complications include using regional anesthesia when possible and avoiding the use of intermediate- and long-acting neuromuscular blocking agents. Dr. Rosenthal said that there is insufficient evidence in the current medical literature to recommend a single “best” intraoperative fluid management plan for all older adults. “Part of the reason it’s so difficult is because of the cardiac physiologic changes [with aging],” she explained. “Older people are susceptible to volume overload. On the other hand, they also may have an exaggerated decline in cardiac function if you give them too little fluid and they have insufficient preload. It’s a very fine line and that’s why it’s hard to recommend a single best strategy.”
Be alert to postoperative delirium
Postoperatively, the guideline recommends that care plans include controlling perioperative acute pain; addressing delirium/cognitive issues; preventing functional decline, falls, pressure ulcers, and urinary track infections; maintaining adequate nutrition; and avoiding pulmonary complications. Dr. Rosenthal underscored the importance of using the four-question Short Confusion Assessment Method (Short CAM) to assess for delirium. “For it to be delirium, there has to be evidence of acute change in mental status from baseline; it has to be acute and fluctuating, and characterized by inattention,” she said. “The patient also has to have either disorganized thinking or an altered level of consciousness.”
Many of the precipitating factors of delirium can be prevented by treating pain, watching medications, preventing dehydration and undernutrition, removing catheters and other devices when possible, preventing constipation, and using minimally invasive techniques to reduce the physiologic stress of surgery. “Sometimes symptoms of delirium are a warning sign that something else is going on, such as an infection, hypoxemia, electrolyte imbalance, neurological events, and major organ dysfunction,” she said. The first-line therapy for treating delirium as recommended in the guideline is a multicomponent intervention that focuses on frequent reorientation with voice, calendars, and clocks; eliminating use of restraints; having familiar objects in the room; and ensuring the use of assistive devices. The second-line therapy is antipsychotic medications at the lowest effective dose. “The mantra is start low and go slow,” she said.
Preventing postoperative functional decline
Another postoperative strategy in the guideline involves targeted fall prevention, such as having an assistive device at the bedside if used as an outpatient and prescribing early physical therapy focused on maintaining mobility as the primary event. “Every day an older patient is immobilized it takes at least 3 days to regain the lost function,” Dr. Rosenthal said. “And for older surgical patients, one in four experiences a significant decline in function by hospital discharge and 60% experience some loss of independence.” (The latter statistic comes from a study published online July 13, 2016, in JAMA Surgery: doi:10.1001/jamasurg.2016.1689.) Interventions for preventing functional decline include promotion of family participation in care, early mobilization, early physical/occupational therapy referral, geriatric consultation, comprehensive discharge planning, and nutritional support. She pointed out that an estimated 40% of community-dwelling elders and two-thirds of nursing home residents are either malnourished or “at risk” of malnutrition.
Transition of care
The final category in the guideline, transition of care, recommends an assessment of social support/home health needs, complete medication review, predischarge geriatric assessment, formal written discharge instructions, and communication with the patient’s primary care physician. “Common models of transitional care involve good coordination with the primary care physician,” she said. “There’s good data to show that people who see their primary care physician within 2 weeks of discharge do better in terms of readmission.”
Dr. Rosenthal reported having no financial disclosures.
SAN DIEGO – As the number of surgery patients over the age of 65 continues to burgeon, clinicians have a resource to help them provide optimal perioperative care to this patient population.
At the American College of Surgeons/National Surgical Quality Improvement Program National Conference, Ronnie A. Rosenthal, MD, discussed highlights from “Optimal Perioperative Management of the Geriatric Patient: A Best Practice Guideline from the ACS NSQIP/American Geriatrics Society,” which was published in January 2016.
Work on the guideline began in 2013, when a 28-member multidisciplinary panel began to conduct a structured search of Medline to identify systematic reviews, meta-analyses, practice guidelines, and clinical trials on the topic. The panel included experts from ACS, the ACS Geriatric Surgery Task Force, the American Society of Anesthesiologists, the American Geriatrics Society, and the AGS’ Geriatrics for Specialists Initiative. The 61-page document is divided into four categories: immediate preoperative period, intraoperative management, postoperative care, and care transitions.
Working with patients on goals
As noted in the guideline, a primary goal of the immediate preoperative period is to discuss with the patient his or her goals and expectations. Patient expectations are influenced by their treatment preferences. In fact, researchers have found that older patients are less likely to want a treatment – even if it results in cure – that may result in severe functional or cognitive impairment. For patients with existing advanced directives, organizations representing nurses, anesthesiologists, and surgeons all agree that there must be a “reconsideration” of these directives prior to surgery. A discussion that includes the new risks of the procedure must be conducted to ensure that the approach to potential life-threatening problems is consistent with the patient’s values.
Preoperative management of medications
Another recommendation for the preoperative period is to ensure that older patients have shorter fasts, have appropriate prophylactic antibiotics, continue medications with withdrawal potential, and discontinue medications that are not essential. The latter point is based on the Beers Criteria, a list of medications that are inappropriate or potentially inappropriate to use in older adults (J Am Geriatr Soc. 2015 Nov;63[11]:2227-46). “You want to discontinue as many inappropriate medications as possible, because one of the main side effects of their use is delirium, and you want to avoid that,” said Dr. Rosenthal, professor of surgery at the Yale University, New Haven, Conn., and one of the guideline authors.
Anesthesia and pain management
Intraoperative management strategies contained in the guideline include establishing an anesthetic approach and a perioperative analgesia pain plan, preventing postoperative nausea and vomiting, assessing patient safety in the OR, preventing predictable complications, and optimizing fluid management. Physiologic effects of anesthesia medications include changes in systemic vascular resistance, cardiac preload, baroreceptor responses, lung mechanics, oxygen diffusion, neurotransmitter function, and end-organ blood flow, among others. “These physiologic changes of aging have significant clinical implications,” Dr. Rosenthal noted. “These are variable among individuals and variable among organ systems, and it’s important that we pay attention to that. Because of this variability, there is insufficient evidence to recommend a single ‘best’ anesthetic plan for all older adults.”
The guideline recommends that each patient have an individualized pain plan that consists of a directed pain history and physical exam and is appropriately titrated for increased sensitivity. “It should include a prophylactic bowel regimen for anybody who’s on an opioid in particular,” she said. “We should avoid inappropriate medications like benzodiazepines, and we should use a multimodal therapy with opioid-sparing and regional techniques.”
Pulmonary considerations for anesthesia include susceptibility to hypocarbia and hypoxemia, and susceptibility to residual anesthetic effects. “Because of physiologic changes, the anesthesia medications aren’t metabolized in the same way,” she said. “Older people may have lower drug requirements and may not recover as quickly from the effects of these drugs. This can lead to respiratory compromise and also can increase the risk of aspiration.” Strategies to prevent pulmonary complications include using regional anesthesia when possible and avoiding the use of intermediate- and long-acting neuromuscular blocking agents. Dr. Rosenthal said that there is insufficient evidence in the current medical literature to recommend a single “best” intraoperative fluid management plan for all older adults. “Part of the reason it’s so difficult is because of the cardiac physiologic changes [with aging],” she explained. “Older people are susceptible to volume overload. On the other hand, they also may have an exaggerated decline in cardiac function if you give them too little fluid and they have insufficient preload. It’s a very fine line and that’s why it’s hard to recommend a single best strategy.”
Be alert to postoperative delirium
Postoperatively, the guideline recommends that care plans include controlling perioperative acute pain; addressing delirium/cognitive issues; preventing functional decline, falls, pressure ulcers, and urinary track infections; maintaining adequate nutrition; and avoiding pulmonary complications. Dr. Rosenthal underscored the importance of using the four-question Short Confusion Assessment Method (Short CAM) to assess for delirium. “For it to be delirium, there has to be evidence of acute change in mental status from baseline; it has to be acute and fluctuating, and characterized by inattention,” she said. “The patient also has to have either disorganized thinking or an altered level of consciousness.”
Many of the precipitating factors of delirium can be prevented by treating pain, watching medications, preventing dehydration and undernutrition, removing catheters and other devices when possible, preventing constipation, and using minimally invasive techniques to reduce the physiologic stress of surgery. “Sometimes symptoms of delirium are a warning sign that something else is going on, such as an infection, hypoxemia, electrolyte imbalance, neurological events, and major organ dysfunction,” she said. The first-line therapy for treating delirium as recommended in the guideline is a multicomponent intervention that focuses on frequent reorientation with voice, calendars, and clocks; eliminating use of restraints; having familiar objects in the room; and ensuring the use of assistive devices. The second-line therapy is antipsychotic medications at the lowest effective dose. “The mantra is start low and go slow,” she said.
Preventing postoperative functional decline
Another postoperative strategy in the guideline involves targeted fall prevention, such as having an assistive device at the bedside if used as an outpatient and prescribing early physical therapy focused on maintaining mobility as the primary event. “Every day an older patient is immobilized it takes at least 3 days to regain the lost function,” Dr. Rosenthal said. “And for older surgical patients, one in four experiences a significant decline in function by hospital discharge and 60% experience some loss of independence.” (The latter statistic comes from a study published online July 13, 2016, in JAMA Surgery: doi:10.1001/jamasurg.2016.1689.) Interventions for preventing functional decline include promotion of family participation in care, early mobilization, early physical/occupational therapy referral, geriatric consultation, comprehensive discharge planning, and nutritional support. She pointed out that an estimated 40% of community-dwelling elders and two-thirds of nursing home residents are either malnourished or “at risk” of malnutrition.
Transition of care
The final category in the guideline, transition of care, recommends an assessment of social support/home health needs, complete medication review, predischarge geriatric assessment, formal written discharge instructions, and communication with the patient’s primary care physician. “Common models of transitional care involve good coordination with the primary care physician,” she said. “There’s good data to show that people who see their primary care physician within 2 weeks of discharge do better in terms of readmission.”
Dr. Rosenthal reported having no financial disclosures.
SAN DIEGO – As the number of surgery patients over the age of 65 continues to burgeon, clinicians have a resource to help them provide optimal perioperative care to this patient population.
At the American College of Surgeons/National Surgical Quality Improvement Program National Conference, Ronnie A. Rosenthal, MD, discussed highlights from “Optimal Perioperative Management of the Geriatric Patient: A Best Practice Guideline from the ACS NSQIP/American Geriatrics Society,” which was published in January 2016.
Work on the guideline began in 2013, when a 28-member multidisciplinary panel began to conduct a structured search of Medline to identify systematic reviews, meta-analyses, practice guidelines, and clinical trials on the topic. The panel included experts from ACS, the ACS Geriatric Surgery Task Force, the American Society of Anesthesiologists, the American Geriatrics Society, and the AGS’ Geriatrics for Specialists Initiative. The 61-page document is divided into four categories: immediate preoperative period, intraoperative management, postoperative care, and care transitions.
Working with patients on goals
As noted in the guideline, a primary goal of the immediate preoperative period is to discuss with the patient his or her goals and expectations. Patient expectations are influenced by their treatment preferences. In fact, researchers have found that older patients are less likely to want a treatment – even if it results in cure – that may result in severe functional or cognitive impairment. For patients with existing advanced directives, organizations representing nurses, anesthesiologists, and surgeons all agree that there must be a “reconsideration” of these directives prior to surgery. A discussion that includes the new risks of the procedure must be conducted to ensure that the approach to potential life-threatening problems is consistent with the patient’s values.
Preoperative management of medications
Another recommendation for the preoperative period is to ensure that older patients have shorter fasts, have appropriate prophylactic antibiotics, continue medications with withdrawal potential, and discontinue medications that are not essential. The latter point is based on the Beers Criteria, a list of medications that are inappropriate or potentially inappropriate to use in older adults (J Am Geriatr Soc. 2015 Nov;63[11]:2227-46). “You want to discontinue as many inappropriate medications as possible, because one of the main side effects of their use is delirium, and you want to avoid that,” said Dr. Rosenthal, professor of surgery at the Yale University, New Haven, Conn., and one of the guideline authors.
Anesthesia and pain management
Intraoperative management strategies contained in the guideline include establishing an anesthetic approach and a perioperative analgesia pain plan, preventing postoperative nausea and vomiting, assessing patient safety in the OR, preventing predictable complications, and optimizing fluid management. Physiologic effects of anesthesia medications include changes in systemic vascular resistance, cardiac preload, baroreceptor responses, lung mechanics, oxygen diffusion, neurotransmitter function, and end-organ blood flow, among others. “These physiologic changes of aging have significant clinical implications,” Dr. Rosenthal noted. “These are variable among individuals and variable among organ systems, and it’s important that we pay attention to that. Because of this variability, there is insufficient evidence to recommend a single ‘best’ anesthetic plan for all older adults.”
The guideline recommends that each patient have an individualized pain plan that consists of a directed pain history and physical exam and is appropriately titrated for increased sensitivity. “It should include a prophylactic bowel regimen for anybody who’s on an opioid in particular,” she said. “We should avoid inappropriate medications like benzodiazepines, and we should use a multimodal therapy with opioid-sparing and regional techniques.”
Pulmonary considerations for anesthesia include susceptibility to hypocarbia and hypoxemia, and susceptibility to residual anesthetic effects. “Because of physiologic changes, the anesthesia medications aren’t metabolized in the same way,” she said. “Older people may have lower drug requirements and may not recover as quickly from the effects of these drugs. This can lead to respiratory compromise and also can increase the risk of aspiration.” Strategies to prevent pulmonary complications include using regional anesthesia when possible and avoiding the use of intermediate- and long-acting neuromuscular blocking agents. Dr. Rosenthal said that there is insufficient evidence in the current medical literature to recommend a single “best” intraoperative fluid management plan for all older adults. “Part of the reason it’s so difficult is because of the cardiac physiologic changes [with aging],” she explained. “Older people are susceptible to volume overload. On the other hand, they also may have an exaggerated decline in cardiac function if you give them too little fluid and they have insufficient preload. It’s a very fine line and that’s why it’s hard to recommend a single best strategy.”
Be alert to postoperative delirium
Postoperatively, the guideline recommends that care plans include controlling perioperative acute pain; addressing delirium/cognitive issues; preventing functional decline, falls, pressure ulcers, and urinary track infections; maintaining adequate nutrition; and avoiding pulmonary complications. Dr. Rosenthal underscored the importance of using the four-question Short Confusion Assessment Method (Short CAM) to assess for delirium. “For it to be delirium, there has to be evidence of acute change in mental status from baseline; it has to be acute and fluctuating, and characterized by inattention,” she said. “The patient also has to have either disorganized thinking or an altered level of consciousness.”
Many of the precipitating factors of delirium can be prevented by treating pain, watching medications, preventing dehydration and undernutrition, removing catheters and other devices when possible, preventing constipation, and using minimally invasive techniques to reduce the physiologic stress of surgery. “Sometimes symptoms of delirium are a warning sign that something else is going on, such as an infection, hypoxemia, electrolyte imbalance, neurological events, and major organ dysfunction,” she said. The first-line therapy for treating delirium as recommended in the guideline is a multicomponent intervention that focuses on frequent reorientation with voice, calendars, and clocks; eliminating use of restraints; having familiar objects in the room; and ensuring the use of assistive devices. The second-line therapy is antipsychotic medications at the lowest effective dose. “The mantra is start low and go slow,” she said.
Preventing postoperative functional decline
Another postoperative strategy in the guideline involves targeted fall prevention, such as having an assistive device at the bedside if used as an outpatient and prescribing early physical therapy focused on maintaining mobility as the primary event. “Every day an older patient is immobilized it takes at least 3 days to regain the lost function,” Dr. Rosenthal said. “And for older surgical patients, one in four experiences a significant decline in function by hospital discharge and 60% experience some loss of independence.” (The latter statistic comes from a study published online July 13, 2016, in JAMA Surgery: doi:10.1001/jamasurg.2016.1689.) Interventions for preventing functional decline include promotion of family participation in care, early mobilization, early physical/occupational therapy referral, geriatric consultation, comprehensive discharge planning, and nutritional support. She pointed out that an estimated 40% of community-dwelling elders and two-thirds of nursing home residents are either malnourished or “at risk” of malnutrition.
Transition of care
The final category in the guideline, transition of care, recommends an assessment of social support/home health needs, complete medication review, predischarge geriatric assessment, formal written discharge instructions, and communication with the patient’s primary care physician. “Common models of transitional care involve good coordination with the primary care physician,” she said. “There’s good data to show that people who see their primary care physician within 2 weeks of discharge do better in terms of readmission.”
Dr. Rosenthal reported having no financial disclosures.
EXPERT ANALYSIS AT THE ACS NSQIP NATIONAL CONFERENCE
Palliative cancer surgery: Prioritize patient values
Risk-assessment tools can give surgeons a clinical framework to help inform decisions about palliative care surgery in patients with advanced malignancies, but cannot replace nuanced clinical judgment that incorporates patients’ priorities, according to results of a meta-analysis.
Ian W. Folkert, MD, and Robert E. Roses, MD, of the department of surgery at the Hospital of the University of Pennsylvania, Philadelphia, reviewed the available research on the indications for palliative surgery for patients with advanced disease and risk-assessment tools for patient selection. Emergent and palliative surgery in such situations require a careful consideration of many clinical factors such as overall prognosis and risk of a surgical approach, compared with nonsurgical interventions (J Surg Oncol. 2016;114:[3]311-15). But the investigators concluded that while an evidence-based approach to patient selection for palliative cancer surgery can offer some guidance on the potential for achieving clinical goals, ultimately the decision to proceed must prioritize patient values and orientation to treatment.
Tumor bleeding
Tumor-related complications often initiate the question of palliative surgical and nonsurgical interventions.
Studies of acute hemorrhage from malignancies indicate that bleeding originating from a tumor is rarely massive and usually can be managed endoscopically (Clin Endosc. 2015 Mar;48[2]:121-7; Aliment Pharmacol Ther. 2013;38:144-50; Mayo Clin Proc. 1994;69[8]:736-40). Transcatheter arterial embolization also is used successfully to manage tumor bleeding (J Vasc Interv Radiol. 2015 Sep;26[9]:1297-304; Indian J Cancer. 2014 Feb; 51[6]56-9). The investigators stated, “Although tumor rebleeding may be frequent, repeat endoscopy is often effective and is self-recommending given the high risk of major morbidity after a palliative foregut resection ... [and] all efforts should be made to avoid emergency gastrectomy or esophagectomy.”
Obstructing tumors
Patients with acute colonic obstruction because of colon cancer typically have been treated with a proximal diverting colostomy, but palliative self-expanding metallic stent placement (SEMS) has emerged as an option. Recent studies have shown both short- and long-term clinical success of SEMS, but rates of major morbidity and mortality for emergent surgery and SEMS were similar, as were rates of overall survival (Surg Endosc. 2015;29[6];1580-5; World J Gastroenterol. 2013 Sep 7;19[33]:5565-74). SEMS-related mortality was primarily because of perforation (Endoscopy. 2008 Mar;40[3]:184-91). Stenting for esophageal and gastroesophageal (GE) function obstruction is also emerging as a nonsurgical option. The investigators noted, “There is a very limited role for palliative surgery for esophageal and GE junction tumors.” Gastric outlet obstruction, proximal duodenal obstruction, and biliary tract obstruction are treated palliatively with stents, but “gastrojejunostomy and other bypass operations may provide effective palliation in carefully selected patients.”
Tumor perforation
Few nonsurgical treatment options are available for tumor perforation. Palliative surgical intervention often is undertaken in the context of neutropenia and abdominal pain, the investigators said. These patients are at high risk for morbidity and mortality. One study reviewed found that “prolonged neutropenia and severe sepsis were associated with poor outcomes in all patients, while surgical management was associated with improved survival (Ann Surg. 2008;248[1];104-9), but nonoperative management and comfort care were deemed appropriate for those patients with advanced disease and for those in whom surgery is high risk.
Patient selection for palliative surgery
The studies examined suggest that patient selection for palliative surgical intervention requires the weighing of clinical variables of frailty, morbidity, and mortality. The investigators reviewed a variety of risk-assessment tools developed to help surgeons with that decision (J Am Coll Surg. 2003;197[1];16-21; J Palliat Med. 2014;17:37-42; Ann Surg. 2011;254[2]:333-8). Among the factors considered are the amplified risks of mortality in these patients, the high cost of emergent operations, and most importantly, the chances of extending survival. The complexity of palliative and emergent surgical indications means that risk-assessment studies are “frequently too reductive to provide meaningful guidance” to the surgeon.
Dr. Folkert and Dr. Roses concluded that risk-assessment tools underscore the poor outcomes associated with operations in this setting, and, to some extent, guide decision making, but “they do not supplant clinical judgment, nor do they account for patient values and orientation toward treatment. It remains impossible to place a uniform value on length and quality of life, and patients’ values are paramount in informing treatment decisions.”
The authors had no conflicts to disclose.
Risk-assessment tools can give surgeons a clinical framework to help inform decisions about palliative care surgery in patients with advanced malignancies, but cannot replace nuanced clinical judgment that incorporates patients’ priorities, according to results of a meta-analysis.
Ian W. Folkert, MD, and Robert E. Roses, MD, of the department of surgery at the Hospital of the University of Pennsylvania, Philadelphia, reviewed the available research on the indications for palliative surgery for patients with advanced disease and risk-assessment tools for patient selection. Emergent and palliative surgery in such situations require a careful consideration of many clinical factors such as overall prognosis and risk of a surgical approach, compared with nonsurgical interventions (J Surg Oncol. 2016;114:[3]311-15). But the investigators concluded that while an evidence-based approach to patient selection for palliative cancer surgery can offer some guidance on the potential for achieving clinical goals, ultimately the decision to proceed must prioritize patient values and orientation to treatment.
Tumor bleeding
Tumor-related complications often initiate the question of palliative surgical and nonsurgical interventions.
Studies of acute hemorrhage from malignancies indicate that bleeding originating from a tumor is rarely massive and usually can be managed endoscopically (Clin Endosc. 2015 Mar;48[2]:121-7; Aliment Pharmacol Ther. 2013;38:144-50; Mayo Clin Proc. 1994;69[8]:736-40). Transcatheter arterial embolization also is used successfully to manage tumor bleeding (J Vasc Interv Radiol. 2015 Sep;26[9]:1297-304; Indian J Cancer. 2014 Feb; 51[6]56-9). The investigators stated, “Although tumor rebleeding may be frequent, repeat endoscopy is often effective and is self-recommending given the high risk of major morbidity after a palliative foregut resection ... [and] all efforts should be made to avoid emergency gastrectomy or esophagectomy.”
Obstructing tumors
Patients with acute colonic obstruction because of colon cancer typically have been treated with a proximal diverting colostomy, but palliative self-expanding metallic stent placement (SEMS) has emerged as an option. Recent studies have shown both short- and long-term clinical success of SEMS, but rates of major morbidity and mortality for emergent surgery and SEMS were similar, as were rates of overall survival (Surg Endosc. 2015;29[6];1580-5; World J Gastroenterol. 2013 Sep 7;19[33]:5565-74). SEMS-related mortality was primarily because of perforation (Endoscopy. 2008 Mar;40[3]:184-91). Stenting for esophageal and gastroesophageal (GE) function obstruction is also emerging as a nonsurgical option. The investigators noted, “There is a very limited role for palliative surgery for esophageal and GE junction tumors.” Gastric outlet obstruction, proximal duodenal obstruction, and biliary tract obstruction are treated palliatively with stents, but “gastrojejunostomy and other bypass operations may provide effective palliation in carefully selected patients.”
Tumor perforation
Few nonsurgical treatment options are available for tumor perforation. Palliative surgical intervention often is undertaken in the context of neutropenia and abdominal pain, the investigators said. These patients are at high risk for morbidity and mortality. One study reviewed found that “prolonged neutropenia and severe sepsis were associated with poor outcomes in all patients, while surgical management was associated with improved survival (Ann Surg. 2008;248[1];104-9), but nonoperative management and comfort care were deemed appropriate for those patients with advanced disease and for those in whom surgery is high risk.
Patient selection for palliative surgery
The studies examined suggest that patient selection for palliative surgical intervention requires the weighing of clinical variables of frailty, morbidity, and mortality. The investigators reviewed a variety of risk-assessment tools developed to help surgeons with that decision (J Am Coll Surg. 2003;197[1];16-21; J Palliat Med. 2014;17:37-42; Ann Surg. 2011;254[2]:333-8). Among the factors considered are the amplified risks of mortality in these patients, the high cost of emergent operations, and most importantly, the chances of extending survival. The complexity of palliative and emergent surgical indications means that risk-assessment studies are “frequently too reductive to provide meaningful guidance” to the surgeon.
Dr. Folkert and Dr. Roses concluded that risk-assessment tools underscore the poor outcomes associated with operations in this setting, and, to some extent, guide decision making, but “they do not supplant clinical judgment, nor do they account for patient values and orientation toward treatment. It remains impossible to place a uniform value on length and quality of life, and patients’ values are paramount in informing treatment decisions.”
The authors had no conflicts to disclose.
Risk-assessment tools can give surgeons a clinical framework to help inform decisions about palliative care surgery in patients with advanced malignancies, but cannot replace nuanced clinical judgment that incorporates patients’ priorities, according to results of a meta-analysis.
Ian W. Folkert, MD, and Robert E. Roses, MD, of the department of surgery at the Hospital of the University of Pennsylvania, Philadelphia, reviewed the available research on the indications for palliative surgery for patients with advanced disease and risk-assessment tools for patient selection. Emergent and palliative surgery in such situations require a careful consideration of many clinical factors such as overall prognosis and risk of a surgical approach, compared with nonsurgical interventions (J Surg Oncol. 2016;114:[3]311-15). But the investigators concluded that while an evidence-based approach to patient selection for palliative cancer surgery can offer some guidance on the potential for achieving clinical goals, ultimately the decision to proceed must prioritize patient values and orientation to treatment.
Tumor bleeding
Tumor-related complications often initiate the question of palliative surgical and nonsurgical interventions.
Studies of acute hemorrhage from malignancies indicate that bleeding originating from a tumor is rarely massive and usually can be managed endoscopically (Clin Endosc. 2015 Mar;48[2]:121-7; Aliment Pharmacol Ther. 2013;38:144-50; Mayo Clin Proc. 1994;69[8]:736-40). Transcatheter arterial embolization also is used successfully to manage tumor bleeding (J Vasc Interv Radiol. 2015 Sep;26[9]:1297-304; Indian J Cancer. 2014 Feb; 51[6]56-9). The investigators stated, “Although tumor rebleeding may be frequent, repeat endoscopy is often effective and is self-recommending given the high risk of major morbidity after a palliative foregut resection ... [and] all efforts should be made to avoid emergency gastrectomy or esophagectomy.”
Obstructing tumors
Patients with acute colonic obstruction because of colon cancer typically have been treated with a proximal diverting colostomy, but palliative self-expanding metallic stent placement (SEMS) has emerged as an option. Recent studies have shown both short- and long-term clinical success of SEMS, but rates of major morbidity and mortality for emergent surgery and SEMS were similar, as were rates of overall survival (Surg Endosc. 2015;29[6];1580-5; World J Gastroenterol. 2013 Sep 7;19[33]:5565-74). SEMS-related mortality was primarily because of perforation (Endoscopy. 2008 Mar;40[3]:184-91). Stenting for esophageal and gastroesophageal (GE) function obstruction is also emerging as a nonsurgical option. The investigators noted, “There is a very limited role for palliative surgery for esophageal and GE junction tumors.” Gastric outlet obstruction, proximal duodenal obstruction, and biliary tract obstruction are treated palliatively with stents, but “gastrojejunostomy and other bypass operations may provide effective palliation in carefully selected patients.”
Tumor perforation
Few nonsurgical treatment options are available for tumor perforation. Palliative surgical intervention often is undertaken in the context of neutropenia and abdominal pain, the investigators said. These patients are at high risk for morbidity and mortality. One study reviewed found that “prolonged neutropenia and severe sepsis were associated with poor outcomes in all patients, while surgical management was associated with improved survival (Ann Surg. 2008;248[1];104-9), but nonoperative management and comfort care were deemed appropriate for those patients with advanced disease and for those in whom surgery is high risk.
Patient selection for palliative surgery
The studies examined suggest that patient selection for palliative surgical intervention requires the weighing of clinical variables of frailty, morbidity, and mortality. The investigators reviewed a variety of risk-assessment tools developed to help surgeons with that decision (J Am Coll Surg. 2003;197[1];16-21; J Palliat Med. 2014;17:37-42; Ann Surg. 2011;254[2]:333-8). Among the factors considered are the amplified risks of mortality in these patients, the high cost of emergent operations, and most importantly, the chances of extending survival. The complexity of palliative and emergent surgical indications means that risk-assessment studies are “frequently too reductive to provide meaningful guidance” to the surgeon.
Dr. Folkert and Dr. Roses concluded that risk-assessment tools underscore the poor outcomes associated with operations in this setting, and, to some extent, guide decision making, but “they do not supplant clinical judgment, nor do they account for patient values and orientation toward treatment. It remains impossible to place a uniform value on length and quality of life, and patients’ values are paramount in informing treatment decisions.”
The authors had no conflicts to disclose.
FROM THE JOURNAL OF SURGICAL ONCOLOGY
10-year follow-up: Localized prostate cancer treatments offer similar efficacy
The three main approaches for treating localized prostate cancer – surgery, radiotherapy, or active monitoring – yield similar efficacy outcomes but different quality-of-life outcomes, according to two reports published on the New England Journal of Medicine.
Both reports present the findings of the ongoing Protect (Prostate Testing for Cancer and Treatment) trial, a large prospective, randomized trial in the United Kingdom comparing mortality and other health outcomes in men with PSA-detected localized disease. The trial involved 82,429 men aged 50-69 years who had a PSA test between 1999 and 2009, of whom 2,664 were found to have localized prostate cancer. A total of 1,643 of these participants agreed to be randomly assigned to radical prostatectomy (553 men), radical radiotherapy (545 men), or active monitoring (545 men).
“Active monitoring” involved avoiding any immediate therapy and regularly monitoring disease progression so that radical treatment with curative intent could be given if the need arose. Patients were monitored every 3 months for the first year, then every 6-12 months thereafter. This differs from “watchful waiting,” which doesn’t involve any plan for curative radical treatment if disease progresses.
The first report focused on mortality and disease progression in these 1,643 participants at a median of 10 years of follow-up. The primary outcome measure, prostate cancer–specific survival, was 98.8% or greater across all three study groups, and there was no significant difference among them. Thus, all three approaches yielded the same efficacy: prostate cancer–specific mortality of approximately 1%, said Freddie C. Hamdy, MD, of the Nuffield Department of Surgical Sciences, University of Oxford, and his associates.
However, the rate of disease progression among men assigned to surgery (8.9/1,000 person-years) or to radiotherapy (9.0/1,000 person-years) was less than half the rate among men assigned to active monitoring (22.9/1,000 person-years). The rate of metastasis followed this same pattern (2.4, 3.0, and 6.3 per 1,000 person-years, respectively).
“These differences show the effectiveness of immediate radical therapy over active monitoring, but they have not translated into significant differences – nor have they ruled out equivalence – in disease-specific or all-cause mortality; thus, longer-term follow-up is necessary,” Dr. Hamdy and his associates said (N Engl J Med. 2016 Sep 14. doi: 10.1056/NEJMoa1606220).
The trade-off was that 44% of the men assigned to active monitoring were able to forgo both radical surgery and radical radiotherapy, avoiding the adverse effects of those treatments. These included nine thromboembolic or cardiovascular events, 14 transfusions, one rectal injury, and nine anastomotic problems requiring intervention, they noted.
It is important to remember that approximately one-fourth of the men assigned to active monitoring went on to undergo radical treatment within 3 years, and that more than half did so by the 10-year follow-up date, the investigators added.
The second report focused on patient-reported outcomes concerning urinary, bowel, sexual, and quality-of-life issues in the 1,643 participants at 6 years of follow-up. These differed markedly among the three study groups, said Jenny L. Donovan, PhD, of the School of Social and Community Medicine, University of Bristol, U.K., and her associates.
Prostatectomy had a clear negative effect on urinary continence and sexual function, particularly erectile function, compared with radiotherapy and active monitoring. This peaked at 6 months after surgery, and though some patients recovered some function over time, urinary incontinence remained worse in the prostatectomy group than in the other two groups throughout follow-up. The use of absorbent pads rose from 1% at baseline to 46% at 6 months in the prostatectomy group. In comparison, the 6-month rate in the radiotherapy group rose to only 5% and that in the active-monitoring group to only 4%.
Radiotherapy plus neoadjuvant androgen-deprivation therapy had more of a negative effect on bowel function, urinary voiding, and nocturia than did the other two treatment approaches. However, many patients eventually showed considerable recovery on most of these measures, except that they continued to have bloody stools more frequently than did men who had prostatectomy or active monitoring.
Men in the active-monitoring group had substantially less difficulty with urinary, sexual, and bowel function, as expected. However, this gradually worsened over time as increasing numbers of these men eventually underwent radical treatments, Dr. Donovan and her associates wrote (N Engl J Med. 2016 Sep 14. doi: 10.1056/NEJMoa1606221).
Quality-of-life measures generally reflected these differences among the three study groups, “with some evidence of accommodation to changes over time.” General mental and physical health, cancer-specific quality of life, and anxiety and depression all were similar across the three groups at 6 years.
“Follow-up for an additional 5-10 years is required to fully inform decisions involving the trade-off between the shorter-term effects of the management strategies shown here and the longer course of progression and treatment of prostate cancer in the context of other life-threatening conditions,” they said.
The Protect trial was supported by the U.K. National Institute for Health Research Health Technology Assessment Programme, the University of Oxford, University Hospitals Bristol, the Oxford NIHR Biomedical Research Centre, and the Cancer Research U.K. Oxford Centre. Dr. Hamdy and Dr. Donovan and their associates reported having no relevant financial disclosures.
As both groups of researchers noted, longer follow-up is needed to definitively assess outcomes in the Protect trial. For now, however, we can conclude that active monitoring leads to increased metastasis, compared with either surgery or radiotherapy.
So if a man wants to avoid metastatic prostate cancer and the adverse effects of its treatment, active monitoring should be considered only if he has life-shortening, coexisting disease and his life expectancy is less than the 10-year median follow-up of this study.
Men who have low- or intermediate-risk prostate cancer should feel free to select either surgery or radiotherapy on the basis of the treatments’ QOL profiles, since the mortality profiles are equivalent.
Anthony V. D’Amico, MD, is in the department of radiation oncology at Brigham and Women’s Hospital and the Dana-Farber Cancer Institute, both in Boston. He reported having no relevant financial disclosures. Dr. D’Amico made these remarks in an editorial accompanying the two reports on the Protect trial (N Engl J Med. 2016 Sep 14. doi: 10.1056/NEHMe1610395).
As both groups of researchers noted, longer follow-up is needed to definitively assess outcomes in the Protect trial. For now, however, we can conclude that active monitoring leads to increased metastasis, compared with either surgery or radiotherapy.
So if a man wants to avoid metastatic prostate cancer and the adverse effects of its treatment, active monitoring should be considered only if he has life-shortening, coexisting disease and his life expectancy is less than the 10-year median follow-up of this study.
Men who have low- or intermediate-risk prostate cancer should feel free to select either surgery or radiotherapy on the basis of the treatments’ QOL profiles, since the mortality profiles are equivalent.
Anthony V. D’Amico, MD, is in the department of radiation oncology at Brigham and Women’s Hospital and the Dana-Farber Cancer Institute, both in Boston. He reported having no relevant financial disclosures. Dr. D’Amico made these remarks in an editorial accompanying the two reports on the Protect trial (N Engl J Med. 2016 Sep 14. doi: 10.1056/NEHMe1610395).
As both groups of researchers noted, longer follow-up is needed to definitively assess outcomes in the Protect trial. For now, however, we can conclude that active monitoring leads to increased metastasis, compared with either surgery or radiotherapy.
So if a man wants to avoid metastatic prostate cancer and the adverse effects of its treatment, active monitoring should be considered only if he has life-shortening, coexisting disease and his life expectancy is less than the 10-year median follow-up of this study.
Men who have low- or intermediate-risk prostate cancer should feel free to select either surgery or radiotherapy on the basis of the treatments’ QOL profiles, since the mortality profiles are equivalent.
Anthony V. D’Amico, MD, is in the department of radiation oncology at Brigham and Women’s Hospital and the Dana-Farber Cancer Institute, both in Boston. He reported having no relevant financial disclosures. Dr. D’Amico made these remarks in an editorial accompanying the two reports on the Protect trial (N Engl J Med. 2016 Sep 14. doi: 10.1056/NEHMe1610395).
The three main approaches for treating localized prostate cancer – surgery, radiotherapy, or active monitoring – yield similar efficacy outcomes but different quality-of-life outcomes, according to two reports published on the New England Journal of Medicine.
Both reports present the findings of the ongoing Protect (Prostate Testing for Cancer and Treatment) trial, a large prospective, randomized trial in the United Kingdom comparing mortality and other health outcomes in men with PSA-detected localized disease. The trial involved 82,429 men aged 50-69 years who had a PSA test between 1999 and 2009, of whom 2,664 were found to have localized prostate cancer. A total of 1,643 of these participants agreed to be randomly assigned to radical prostatectomy (553 men), radical radiotherapy (545 men), or active monitoring (545 men).
“Active monitoring” involved avoiding any immediate therapy and regularly monitoring disease progression so that radical treatment with curative intent could be given if the need arose. Patients were monitored every 3 months for the first year, then every 6-12 months thereafter. This differs from “watchful waiting,” which doesn’t involve any plan for curative radical treatment if disease progresses.
The first report focused on mortality and disease progression in these 1,643 participants at a median of 10 years of follow-up. The primary outcome measure, prostate cancer–specific survival, was 98.8% or greater across all three study groups, and there was no significant difference among them. Thus, all three approaches yielded the same efficacy: prostate cancer–specific mortality of approximately 1%, said Freddie C. Hamdy, MD, of the Nuffield Department of Surgical Sciences, University of Oxford, and his associates.
However, the rate of disease progression among men assigned to surgery (8.9/1,000 person-years) or to radiotherapy (9.0/1,000 person-years) was less than half the rate among men assigned to active monitoring (22.9/1,000 person-years). The rate of metastasis followed this same pattern (2.4, 3.0, and 6.3 per 1,000 person-years, respectively).
“These differences show the effectiveness of immediate radical therapy over active monitoring, but they have not translated into significant differences – nor have they ruled out equivalence – in disease-specific or all-cause mortality; thus, longer-term follow-up is necessary,” Dr. Hamdy and his associates said (N Engl J Med. 2016 Sep 14. doi: 10.1056/NEJMoa1606220).
The trade-off was that 44% of the men assigned to active monitoring were able to forgo both radical surgery and radical radiotherapy, avoiding the adverse effects of those treatments. These included nine thromboembolic or cardiovascular events, 14 transfusions, one rectal injury, and nine anastomotic problems requiring intervention, they noted.
It is important to remember that approximately one-fourth of the men assigned to active monitoring went on to undergo radical treatment within 3 years, and that more than half did so by the 10-year follow-up date, the investigators added.
The second report focused on patient-reported outcomes concerning urinary, bowel, sexual, and quality-of-life issues in the 1,643 participants at 6 years of follow-up. These differed markedly among the three study groups, said Jenny L. Donovan, PhD, of the School of Social and Community Medicine, University of Bristol, U.K., and her associates.
Prostatectomy had a clear negative effect on urinary continence and sexual function, particularly erectile function, compared with radiotherapy and active monitoring. This peaked at 6 months after surgery, and though some patients recovered some function over time, urinary incontinence remained worse in the prostatectomy group than in the other two groups throughout follow-up. The use of absorbent pads rose from 1% at baseline to 46% at 6 months in the prostatectomy group. In comparison, the 6-month rate in the radiotherapy group rose to only 5% and that in the active-monitoring group to only 4%.
Radiotherapy plus neoadjuvant androgen-deprivation therapy had more of a negative effect on bowel function, urinary voiding, and nocturia than did the other two treatment approaches. However, many patients eventually showed considerable recovery on most of these measures, except that they continued to have bloody stools more frequently than did men who had prostatectomy or active monitoring.
Men in the active-monitoring group had substantially less difficulty with urinary, sexual, and bowel function, as expected. However, this gradually worsened over time as increasing numbers of these men eventually underwent radical treatments, Dr. Donovan and her associates wrote (N Engl J Med. 2016 Sep 14. doi: 10.1056/NEJMoa1606221).
Quality-of-life measures generally reflected these differences among the three study groups, “with some evidence of accommodation to changes over time.” General mental and physical health, cancer-specific quality of life, and anxiety and depression all were similar across the three groups at 6 years.
“Follow-up for an additional 5-10 years is required to fully inform decisions involving the trade-off between the shorter-term effects of the management strategies shown here and the longer course of progression and treatment of prostate cancer in the context of other life-threatening conditions,” they said.
The Protect trial was supported by the U.K. National Institute for Health Research Health Technology Assessment Programme, the University of Oxford, University Hospitals Bristol, the Oxford NIHR Biomedical Research Centre, and the Cancer Research U.K. Oxford Centre. Dr. Hamdy and Dr. Donovan and their associates reported having no relevant financial disclosures.
The three main approaches for treating localized prostate cancer – surgery, radiotherapy, or active monitoring – yield similar efficacy outcomes but different quality-of-life outcomes, according to two reports published on the New England Journal of Medicine.
Both reports present the findings of the ongoing Protect (Prostate Testing for Cancer and Treatment) trial, a large prospective, randomized trial in the United Kingdom comparing mortality and other health outcomes in men with PSA-detected localized disease. The trial involved 82,429 men aged 50-69 years who had a PSA test between 1999 and 2009, of whom 2,664 were found to have localized prostate cancer. A total of 1,643 of these participants agreed to be randomly assigned to radical prostatectomy (553 men), radical radiotherapy (545 men), or active monitoring (545 men).
“Active monitoring” involved avoiding any immediate therapy and regularly monitoring disease progression so that radical treatment with curative intent could be given if the need arose. Patients were monitored every 3 months for the first year, then every 6-12 months thereafter. This differs from “watchful waiting,” which doesn’t involve any plan for curative radical treatment if disease progresses.
The first report focused on mortality and disease progression in these 1,643 participants at a median of 10 years of follow-up. The primary outcome measure, prostate cancer–specific survival, was 98.8% or greater across all three study groups, and there was no significant difference among them. Thus, all three approaches yielded the same efficacy: prostate cancer–specific mortality of approximately 1%, said Freddie C. Hamdy, MD, of the Nuffield Department of Surgical Sciences, University of Oxford, and his associates.
However, the rate of disease progression among men assigned to surgery (8.9/1,000 person-years) or to radiotherapy (9.0/1,000 person-years) was less than half the rate among men assigned to active monitoring (22.9/1,000 person-years). The rate of metastasis followed this same pattern (2.4, 3.0, and 6.3 per 1,000 person-years, respectively).
“These differences show the effectiveness of immediate radical therapy over active monitoring, but they have not translated into significant differences – nor have they ruled out equivalence – in disease-specific or all-cause mortality; thus, longer-term follow-up is necessary,” Dr. Hamdy and his associates said (N Engl J Med. 2016 Sep 14. doi: 10.1056/NEJMoa1606220).
The trade-off was that 44% of the men assigned to active monitoring were able to forgo both radical surgery and radical radiotherapy, avoiding the adverse effects of those treatments. These included nine thromboembolic or cardiovascular events, 14 transfusions, one rectal injury, and nine anastomotic problems requiring intervention, they noted.
It is important to remember that approximately one-fourth of the men assigned to active monitoring went on to undergo radical treatment within 3 years, and that more than half did so by the 10-year follow-up date, the investigators added.
The second report focused on patient-reported outcomes concerning urinary, bowel, sexual, and quality-of-life issues in the 1,643 participants at 6 years of follow-up. These differed markedly among the three study groups, said Jenny L. Donovan, PhD, of the School of Social and Community Medicine, University of Bristol, U.K., and her associates.
Prostatectomy had a clear negative effect on urinary continence and sexual function, particularly erectile function, compared with radiotherapy and active monitoring. This peaked at 6 months after surgery, and though some patients recovered some function over time, urinary incontinence remained worse in the prostatectomy group than in the other two groups throughout follow-up. The use of absorbent pads rose from 1% at baseline to 46% at 6 months in the prostatectomy group. In comparison, the 6-month rate in the radiotherapy group rose to only 5% and that in the active-monitoring group to only 4%.
Radiotherapy plus neoadjuvant androgen-deprivation therapy had more of a negative effect on bowel function, urinary voiding, and nocturia than did the other two treatment approaches. However, many patients eventually showed considerable recovery on most of these measures, except that they continued to have bloody stools more frequently than did men who had prostatectomy or active monitoring.
Men in the active-monitoring group had substantially less difficulty with urinary, sexual, and bowel function, as expected. However, this gradually worsened over time as increasing numbers of these men eventually underwent radical treatments, Dr. Donovan and her associates wrote (N Engl J Med. 2016 Sep 14. doi: 10.1056/NEJMoa1606221).
Quality-of-life measures generally reflected these differences among the three study groups, “with some evidence of accommodation to changes over time.” General mental and physical health, cancer-specific quality of life, and anxiety and depression all were similar across the three groups at 6 years.
“Follow-up for an additional 5-10 years is required to fully inform decisions involving the trade-off between the shorter-term effects of the management strategies shown here and the longer course of progression and treatment of prostate cancer in the context of other life-threatening conditions,” they said.
The Protect trial was supported by the U.K. National Institute for Health Research Health Technology Assessment Programme, the University of Oxford, University Hospitals Bristol, the Oxford NIHR Biomedical Research Centre, and the Cancer Research U.K. Oxford Centre. Dr. Hamdy and Dr. Donovan and their associates reported having no relevant financial disclosures.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: The three main approaches for treating localized prostate cancer yield similar efficacy outcomes but different quality-of-life outcomes.
Major finding: All three treatment approaches – surgery, radiotherapy, and active monitoring – yielded the same efficacy: 10-year prostate cancer–specific mortality of approximately 1%.
Data source: The Protect trial, a prospective randomized study involving 1,643 prostate cancer patients in the U.K. followed for 10 years.
Disclosures: The Protect trial was supported by the U.K. National Institute for Health Research Health Technology Assessment Programme, the University of Oxford, University Hospitals Bristol, the Oxford NIHR Biomedical Research Centre, and the Cancer Research U.K. Oxford Centre. Dr. Hamdy and Dr. Donovan and their associates reported having no relevant financial disclosures.
Study eyes anastomotic failure in stapled vs. hand-sewn techniques
WAIKOLOA, HAWAII – The risk of anastomotic failure among emergency general surgery patients requiring bowel resection and anastomosis stands at 12.5% and is similar between stapled and hand-sewn techniques, results from a multicenter analysis demonstrated.
Surgeons participating in the study, known as Stapled vs. Handsewn: A Prospective Emergency Surgery Study (SHAPES), “appear to be performing hand-sewn techniques in patients who have a higher burden of disease,” Brandon R. Bruns, MD, said at the annual meeting of the American Association for the Surgery of Trauma. “Without adjustment and despite being performed in a more ill population, there was no difference in failure rate between hand-sewn and stapled techniques. After modeling, only being managed with an open abdomen and contamination at initial operation were associated with anastomotic failure.”
For SHAPES, which is the largest study of its kind and was sponsored by the AAST Multi-Institutional Studies Committee, Dr. Bruns and his associates set out to prospectively evaluate anastomotic failure rates for hand-sewn and stapled anastomoses in patients undergoing urgent/emergent operations. A 1999 study by Seattle researchers found that stapled techniques seemed to be associated with anastomotic failure (J Trauma. 1999;47[3]:500-08). Two years later, the same researchers pooled 4-year retrospective data from four trauma centers and concluded that again, hand-sewn techniques appeared to be superior to stapled techniques after traumatic bowel resection and anastomosis (J Trauma. 2001;51[6]:1054-61). Also in 2001, AAST sponsored a multi-institutional study that examined the same question, this time in penetrative colonic injury. Investigators found no difference in complications between the two groups (J Trauma. 2002;52[1]:117-21). They did, however, find a 22.7% overall incidence of colon-related complications.
“With this background we hypothesized that anastomotic failure rate would be high for emergency general surgery patients undergoing bowel resection and anastomosis,” said Dr. Bruns, an acute care surgeon at the R. Adams Cowley Shock Trauma Center at the University of Maryland Medical Center, Baltimore. “We also hypothesized that failure rates would be higher for stapled techniques, compared with hand-sewn.”
The SHAPES researchers prospectively enrolled 595 patients at 15 medical centers in the United States who underwent urgent/emergent bowel resection for emergency general surgery pathology between July 22, 2013, and Dec. 31, 2015. The patients were grouped by hand-sewn vs. stapled anastomoses and demographic and clinical variables were collected. The primary outcome was anastomotic failure. As in other studies, anastomotic failure was evaluated at the anastomosis level. Multivariable logistic regression was done, controlling for age and risk factors for anastomotic failure.
Dr. Bruns reported that the 595 patients had 649 anastomoses. Of these, 61% were stapled and 39% were hand-sewn. The mean age of patients was 62 years, 51% were male, and the overall mortality rate was 7.7%. More than two-thirds of the patients (35%) had more than one indication for operative intervention. The most common single indication for operation was small bowel obstruction, at 23%. The majority of the anastomoses were small bowel to small bowel (72%), while 21% were small bowel to large bowel, and 7% were large bowel to large bowel. There were a total of 81 anastomotic failures, for a rate of 12.5%.
When the researchers compared the hand-sewn and stapled groups, they were similar with respect to sex and age, with higher Charlson comorbidity indices in stapled and a higher body mass index in the hand-sewn group. They also observed a lower hemoglobin, higher INR, higher lactate, lower albumin, and worse renal function in the hand-sewn group, compared with the stapled group. Moreover, hand-sewn anastomotic techniques were performed more frequently in patients that were on vasopressor support. “Despite patients receiving hand-sewn techniques, having worse laboratory values, and more intraoperative vasopressors, the two techniques had equivalent failure rates, at 15.4% for hand-sewn and 10.6% for stapled,” Dr. Bruns said. “Patients with hand-sewn techniques had longer hospital length of stay, longer ICU length of stay, and a significantly higher mortality, compared with those who underwent stapled techniques. Interestingly and different from most other studies on the topic, operating time between the two groups was equivalent.”
On multivariate regression, the presence of contamination at initial bowel resection (OR 1.96) and the patient being managed with an open abdomen (OR 2.53) were independently associated with anastomotic failure, while the type of anastomosis (hand-sewn vs. stapled) was not.
The researchers also conducted a subanalysis of 165 patients managed with an open abdomen who had bowel resection and anastomosis. These patients had higher BMIs, higher lactates, higher INRs, and more negative base deficits, compared with those who were not managed with an open abdomen. “Perhaps not unexpectedly, open abdomen patients were more likely to be on vasopressor agents,” Dr. Bruns said. “They had longer hospital lengths of stay, more ICU days, and an 18.2% overall mortality. Overall there was an almost 22% anastomotic failure rate in patients managed with an open abdomen, compared with an 8.5% rate in patients managed with non-open techniques.” Comparing hand-sewn and stapled techniques, there was no difference in failure rate in patients managed with an open abdomen (25.2% vs. 17.5%, respectively; P = .20).
“An overall mortality rate of 8% and an anastomotic complication rate of 12.5% should emphasize the dire needs for these operations and the need for meticulous operative as well as surgically directed perioperative care in these patients,” the invited discussant, Gregory Jurkovich, MD, professor of surgery at the Davis Medical Center, University of California, said at the meeting. “We as surgeons must pay attention to all aspects of care in these patients.”
Dr. Bruns acknowledged certain limitations of the study, including the fact that it was not a randomized, controlled trial. Also, “surgeon preference did dictate the type of anastomosis that was created, and the patient and surgeon populations were heterogeneous,” he said. “The multivariable model was limited by missing laboratory data, likely given the urgent nature of some of the operative procedures.”
He reported having no financial disclosures.
WAIKOLOA, HAWAII – The risk of anastomotic failure among emergency general surgery patients requiring bowel resection and anastomosis stands at 12.5% and is similar between stapled and hand-sewn techniques, results from a multicenter analysis demonstrated.
Surgeons participating in the study, known as Stapled vs. Handsewn: A Prospective Emergency Surgery Study (SHAPES), “appear to be performing hand-sewn techniques in patients who have a higher burden of disease,” Brandon R. Bruns, MD, said at the annual meeting of the American Association for the Surgery of Trauma. “Without adjustment and despite being performed in a more ill population, there was no difference in failure rate between hand-sewn and stapled techniques. After modeling, only being managed with an open abdomen and contamination at initial operation were associated with anastomotic failure.”
For SHAPES, which is the largest study of its kind and was sponsored by the AAST Multi-Institutional Studies Committee, Dr. Bruns and his associates set out to prospectively evaluate anastomotic failure rates for hand-sewn and stapled anastomoses in patients undergoing urgent/emergent operations. A 1999 study by Seattle researchers found that stapled techniques seemed to be associated with anastomotic failure (J Trauma. 1999;47[3]:500-08). Two years later, the same researchers pooled 4-year retrospective data from four trauma centers and concluded that again, hand-sewn techniques appeared to be superior to stapled techniques after traumatic bowel resection and anastomosis (J Trauma. 2001;51[6]:1054-61). Also in 2001, AAST sponsored a multi-institutional study that examined the same question, this time in penetrative colonic injury. Investigators found no difference in complications between the two groups (J Trauma. 2002;52[1]:117-21). They did, however, find a 22.7% overall incidence of colon-related complications.
“With this background we hypothesized that anastomotic failure rate would be high for emergency general surgery patients undergoing bowel resection and anastomosis,” said Dr. Bruns, an acute care surgeon at the R. Adams Cowley Shock Trauma Center at the University of Maryland Medical Center, Baltimore. “We also hypothesized that failure rates would be higher for stapled techniques, compared with hand-sewn.”
The SHAPES researchers prospectively enrolled 595 patients at 15 medical centers in the United States who underwent urgent/emergent bowel resection for emergency general surgery pathology between July 22, 2013, and Dec. 31, 2015. The patients were grouped by hand-sewn vs. stapled anastomoses and demographic and clinical variables were collected. The primary outcome was anastomotic failure. As in other studies, anastomotic failure was evaluated at the anastomosis level. Multivariable logistic regression was done, controlling for age and risk factors for anastomotic failure.
Dr. Bruns reported that the 595 patients had 649 anastomoses. Of these, 61% were stapled and 39% were hand-sewn. The mean age of patients was 62 years, 51% were male, and the overall mortality rate was 7.7%. More than two-thirds of the patients (35%) had more than one indication for operative intervention. The most common single indication for operation was small bowel obstruction, at 23%. The majority of the anastomoses were small bowel to small bowel (72%), while 21% were small bowel to large bowel, and 7% were large bowel to large bowel. There were a total of 81 anastomotic failures, for a rate of 12.5%.
When the researchers compared the hand-sewn and stapled groups, they were similar with respect to sex and age, with higher Charlson comorbidity indices in stapled and a higher body mass index in the hand-sewn group. They also observed a lower hemoglobin, higher INR, higher lactate, lower albumin, and worse renal function in the hand-sewn group, compared with the stapled group. Moreover, hand-sewn anastomotic techniques were performed more frequently in patients that were on vasopressor support. “Despite patients receiving hand-sewn techniques, having worse laboratory values, and more intraoperative vasopressors, the two techniques had equivalent failure rates, at 15.4% for hand-sewn and 10.6% for stapled,” Dr. Bruns said. “Patients with hand-sewn techniques had longer hospital length of stay, longer ICU length of stay, and a significantly higher mortality, compared with those who underwent stapled techniques. Interestingly and different from most other studies on the topic, operating time between the two groups was equivalent.”
On multivariate regression, the presence of contamination at initial bowel resection (OR 1.96) and the patient being managed with an open abdomen (OR 2.53) were independently associated with anastomotic failure, while the type of anastomosis (hand-sewn vs. stapled) was not.
The researchers also conducted a subanalysis of 165 patients managed with an open abdomen who had bowel resection and anastomosis. These patients had higher BMIs, higher lactates, higher INRs, and more negative base deficits, compared with those who were not managed with an open abdomen. “Perhaps not unexpectedly, open abdomen patients were more likely to be on vasopressor agents,” Dr. Bruns said. “They had longer hospital lengths of stay, more ICU days, and an 18.2% overall mortality. Overall there was an almost 22% anastomotic failure rate in patients managed with an open abdomen, compared with an 8.5% rate in patients managed with non-open techniques.” Comparing hand-sewn and stapled techniques, there was no difference in failure rate in patients managed with an open abdomen (25.2% vs. 17.5%, respectively; P = .20).
“An overall mortality rate of 8% and an anastomotic complication rate of 12.5% should emphasize the dire needs for these operations and the need for meticulous operative as well as surgically directed perioperative care in these patients,” the invited discussant, Gregory Jurkovich, MD, professor of surgery at the Davis Medical Center, University of California, said at the meeting. “We as surgeons must pay attention to all aspects of care in these patients.”
Dr. Bruns acknowledged certain limitations of the study, including the fact that it was not a randomized, controlled trial. Also, “surgeon preference did dictate the type of anastomosis that was created, and the patient and surgeon populations were heterogeneous,” he said. “The multivariable model was limited by missing laboratory data, likely given the urgent nature of some of the operative procedures.”
He reported having no financial disclosures.
WAIKOLOA, HAWAII – The risk of anastomotic failure among emergency general surgery patients requiring bowel resection and anastomosis stands at 12.5% and is similar between stapled and hand-sewn techniques, results from a multicenter analysis demonstrated.
Surgeons participating in the study, known as Stapled vs. Handsewn: A Prospective Emergency Surgery Study (SHAPES), “appear to be performing hand-sewn techniques in patients who have a higher burden of disease,” Brandon R. Bruns, MD, said at the annual meeting of the American Association for the Surgery of Trauma. “Without adjustment and despite being performed in a more ill population, there was no difference in failure rate between hand-sewn and stapled techniques. After modeling, only being managed with an open abdomen and contamination at initial operation were associated with anastomotic failure.”
For SHAPES, which is the largest study of its kind and was sponsored by the AAST Multi-Institutional Studies Committee, Dr. Bruns and his associates set out to prospectively evaluate anastomotic failure rates for hand-sewn and stapled anastomoses in patients undergoing urgent/emergent operations. A 1999 study by Seattle researchers found that stapled techniques seemed to be associated with anastomotic failure (J Trauma. 1999;47[3]:500-08). Two years later, the same researchers pooled 4-year retrospective data from four trauma centers and concluded that again, hand-sewn techniques appeared to be superior to stapled techniques after traumatic bowel resection and anastomosis (J Trauma. 2001;51[6]:1054-61). Also in 2001, AAST sponsored a multi-institutional study that examined the same question, this time in penetrative colonic injury. Investigators found no difference in complications between the two groups (J Trauma. 2002;52[1]:117-21). They did, however, find a 22.7% overall incidence of colon-related complications.
“With this background we hypothesized that anastomotic failure rate would be high for emergency general surgery patients undergoing bowel resection and anastomosis,” said Dr. Bruns, an acute care surgeon at the R. Adams Cowley Shock Trauma Center at the University of Maryland Medical Center, Baltimore. “We also hypothesized that failure rates would be higher for stapled techniques, compared with hand-sewn.”
The SHAPES researchers prospectively enrolled 595 patients at 15 medical centers in the United States who underwent urgent/emergent bowel resection for emergency general surgery pathology between July 22, 2013, and Dec. 31, 2015. The patients were grouped by hand-sewn vs. stapled anastomoses and demographic and clinical variables were collected. The primary outcome was anastomotic failure. As in other studies, anastomotic failure was evaluated at the anastomosis level. Multivariable logistic regression was done, controlling for age and risk factors for anastomotic failure.
Dr. Bruns reported that the 595 patients had 649 anastomoses. Of these, 61% were stapled and 39% were hand-sewn. The mean age of patients was 62 years, 51% were male, and the overall mortality rate was 7.7%. More than two-thirds of the patients (35%) had more than one indication for operative intervention. The most common single indication for operation was small bowel obstruction, at 23%. The majority of the anastomoses were small bowel to small bowel (72%), while 21% were small bowel to large bowel, and 7% were large bowel to large bowel. There were a total of 81 anastomotic failures, for a rate of 12.5%.
When the researchers compared the hand-sewn and stapled groups, they were similar with respect to sex and age, with higher Charlson comorbidity indices in stapled and a higher body mass index in the hand-sewn group. They also observed a lower hemoglobin, higher INR, higher lactate, lower albumin, and worse renal function in the hand-sewn group, compared with the stapled group. Moreover, hand-sewn anastomotic techniques were performed more frequently in patients that were on vasopressor support. “Despite patients receiving hand-sewn techniques, having worse laboratory values, and more intraoperative vasopressors, the two techniques had equivalent failure rates, at 15.4% for hand-sewn and 10.6% for stapled,” Dr. Bruns said. “Patients with hand-sewn techniques had longer hospital length of stay, longer ICU length of stay, and a significantly higher mortality, compared with those who underwent stapled techniques. Interestingly and different from most other studies on the topic, operating time between the two groups was equivalent.”
On multivariate regression, the presence of contamination at initial bowel resection (OR 1.96) and the patient being managed with an open abdomen (OR 2.53) were independently associated with anastomotic failure, while the type of anastomosis (hand-sewn vs. stapled) was not.
The researchers also conducted a subanalysis of 165 patients managed with an open abdomen who had bowel resection and anastomosis. These patients had higher BMIs, higher lactates, higher INRs, and more negative base deficits, compared with those who were not managed with an open abdomen. “Perhaps not unexpectedly, open abdomen patients were more likely to be on vasopressor agents,” Dr. Bruns said. “They had longer hospital lengths of stay, more ICU days, and an 18.2% overall mortality. Overall there was an almost 22% anastomotic failure rate in patients managed with an open abdomen, compared with an 8.5% rate in patients managed with non-open techniques.” Comparing hand-sewn and stapled techniques, there was no difference in failure rate in patients managed with an open abdomen (25.2% vs. 17.5%, respectively; P = .20).
“An overall mortality rate of 8% and an anastomotic complication rate of 12.5% should emphasize the dire needs for these operations and the need for meticulous operative as well as surgically directed perioperative care in these patients,” the invited discussant, Gregory Jurkovich, MD, professor of surgery at the Davis Medical Center, University of California, said at the meeting. “We as surgeons must pay attention to all aspects of care in these patients.”
Dr. Bruns acknowledged certain limitations of the study, including the fact that it was not a randomized, controlled trial. Also, “surgeon preference did dictate the type of anastomosis that was created, and the patient and surgeon populations were heterogeneous,” he said. “The multivariable model was limited by missing laboratory data, likely given the urgent nature of some of the operative procedures.”
He reported having no financial disclosures.
AT THE AAST ANNUAL MEETING
Key clinical point: In patients requiring emergency bowel resection and anastomosis, surgeons appear to be performing hand-sewn techniques in patients who have a higher burden of disease.
Major finding: There were 81 anastomotic failures in the study group, for a rate of 12.5%.
Data source: A prospective evaluation of 595 patients at 15 medical centers in the United States who underwent urgent/emergent bowel resection for emergency general surgery pathology between July 22, 2013, and Dec. 31, 2015.
Disclosures: Dr. Bruns reported having no financial disclosures.
CSF lactate concentration identifies postneurosurgical bacterial meningitis
The concentration of lactate in the cerebrospinal fluid accurately identifies bacterial meningitis that develops after neurosurgery, distinguishing it from other conditions, according to a report published in BMC Infectious Diseases.
In patients who have undergone neurosurgery, failure to promptly identify and treat bacterial meningitis is associated with patient mortality as high as 50%, reported Xiong Xiao of the department of neurosurgery and the China National Clinical Research Center for Neurological Diseases at Beijing Tiantan Hospital and Capital Medical University, Beijing, and associates.
A recent small study suggested that cerebrospinal fluid (CSF) lactate was accurate at differentiating postoperative bacterial meningitis from aseptic meningitis. To examine this possibility in a larger patient population, Dr. Xiao and associates reviewed 1,672 articles in the medical literature. They found few high-quality studies of this topic, but were able to perform a meta-analysis and pool the data from five studies involving 404 postneurosurgical patients treated during a 15-year period.
CSF lactate concentration identified bacterial meningitis with a pooled sensitivity of 92% and a pooled specificity of 88%. “Moreover, this test is fast, simple, objective, and affordable, and can be widely applied in hospitals,” the investigators wrote (BMC Infect Dis. 2016;16:483. doi: 10.1186/s12879-016-1818-2).Larger prospective studies are needed to confirm this finding and to provide a more thorough understanding of the indicators of postneurosurgical meningitis, Dr. Xiao and associates added.
This study was supported by Beijing Tiantan Hospital Funds for Young Scholars, the Ministry of Science and Technology of China, and the Beijing Talents Fund. Dr. Xiao and associates reported having no relevant financial disclosures.
The concentration of lactate in the cerebrospinal fluid accurately identifies bacterial meningitis that develops after neurosurgery, distinguishing it from other conditions, according to a report published in BMC Infectious Diseases.
In patients who have undergone neurosurgery, failure to promptly identify and treat bacterial meningitis is associated with patient mortality as high as 50%, reported Xiong Xiao of the department of neurosurgery and the China National Clinical Research Center for Neurological Diseases at Beijing Tiantan Hospital and Capital Medical University, Beijing, and associates.
A recent small study suggested that cerebrospinal fluid (CSF) lactate was accurate at differentiating postoperative bacterial meningitis from aseptic meningitis. To examine this possibility in a larger patient population, Dr. Xiao and associates reviewed 1,672 articles in the medical literature. They found few high-quality studies of this topic, but were able to perform a meta-analysis and pool the data from five studies involving 404 postneurosurgical patients treated during a 15-year period.
CSF lactate concentration identified bacterial meningitis with a pooled sensitivity of 92% and a pooled specificity of 88%. “Moreover, this test is fast, simple, objective, and affordable, and can be widely applied in hospitals,” the investigators wrote (BMC Infect Dis. 2016;16:483. doi: 10.1186/s12879-016-1818-2).Larger prospective studies are needed to confirm this finding and to provide a more thorough understanding of the indicators of postneurosurgical meningitis, Dr. Xiao and associates added.
This study was supported by Beijing Tiantan Hospital Funds for Young Scholars, the Ministry of Science and Technology of China, and the Beijing Talents Fund. Dr. Xiao and associates reported having no relevant financial disclosures.
The concentration of lactate in the cerebrospinal fluid accurately identifies bacterial meningitis that develops after neurosurgery, distinguishing it from other conditions, according to a report published in BMC Infectious Diseases.
In patients who have undergone neurosurgery, failure to promptly identify and treat bacterial meningitis is associated with patient mortality as high as 50%, reported Xiong Xiao of the department of neurosurgery and the China National Clinical Research Center for Neurological Diseases at Beijing Tiantan Hospital and Capital Medical University, Beijing, and associates.
A recent small study suggested that cerebrospinal fluid (CSF) lactate was accurate at differentiating postoperative bacterial meningitis from aseptic meningitis. To examine this possibility in a larger patient population, Dr. Xiao and associates reviewed 1,672 articles in the medical literature. They found few high-quality studies of this topic, but were able to perform a meta-analysis and pool the data from five studies involving 404 postneurosurgical patients treated during a 15-year period.
CSF lactate concentration identified bacterial meningitis with a pooled sensitivity of 92% and a pooled specificity of 88%. “Moreover, this test is fast, simple, objective, and affordable, and can be widely applied in hospitals,” the investigators wrote (BMC Infect Dis. 2016;16:483. doi: 10.1186/s12879-016-1818-2).Larger prospective studies are needed to confirm this finding and to provide a more thorough understanding of the indicators of postneurosurgical meningitis, Dr. Xiao and associates added.
This study was supported by Beijing Tiantan Hospital Funds for Young Scholars, the Ministry of Science and Technology of China, and the Beijing Talents Fund. Dr. Xiao and associates reported having no relevant financial disclosures.
FROM BMC INFECTIOUS DISEASES
Key clinical point: CSF lactate concentration accurately identifies bacterial meningitis that develops after neurosurgery.
Major finding: CSF lactate concentration identified bacterial meningitis with a pooled sensitivity of 92% and a pooled specificity of 88%.
Data source: A meta-analysis of five published studies involving 404 patients during a 15-year period.
Disclosures: This study was supported by Beijing Tiantan Hospital Funds for Young Scholars, the Ministry of Science and Technology of China, and the Beijing Talents Fund. Dr. Xiao and associates reported having no relevant financial disclosures.
Endobariatrics: Coming to a clinic near you
SAN DIEGO – Device companies are working hard to bring obesity management to the endoscopy suite.
The field is called endobariatrics, and its goal is to fill the gap between surgery and pharmacotherapy. Drugs and lifestyle counseling don’t work for all patients, but many patients resist having surgery. These devices are meant to fill the need for nonsurgical, “noninvasive” solutions to obesity management.
Endobariatrics has the potential to be a boon for both obese patients and gastroenterology practices. Surgeons, however, approach these innovations with caution, due in part to past experiences of rescuing patients with failed devices. “Some innovations get disseminated without adequate studies to demonstrate whether they are superior, or even equal in efficacy and safety to, well-established procedures for the same condition. Even when they have been conducted, negative studies showing no difference may end up being rejected by journals and not published,” noted Karen Deveney, MD, FACS, professor of surgery, vice-chair for education in the department of surgery at Oregon Health & Science University, Portland, and co-Editor of ACS Surgery News.
Several new investigational devices and approaches were showcased at the annual Digestive Disease Week; some “are beginning to approach the kind of results we see with surgical techniques,” said Steven Edmundowicz, MD, medical director of the University of Colorado Digestive Health Center, Aurora.
“We are seeing a tremendous amount of development in this space, but it’s early, and we have to be cautious,” he said. There have already been a few disappointments, including the EndoBarrier, a fluoropolymer liner anchored in the duodenal bulb and unfurled down the duodenum to block food absorption. A key U.S. trial was recently halted due to liver abscesses.
Dr. Edmundowicz reviewed the latest developments presented at DDW.Self-assembling magnets for dual-path enteral anastomoses
The goal of the GI Windows system is to create a partial jejunoileal, side to side bypass without surgery. A 28-mm magnet ring is introduced to the ileum by colonoscopy, and a second ring to the jejunum by endoscopy. The rings snap together and tissue caught between them dies from pressure necrosis, leaving patients with a jejunoileal communication. Once food reaches that point, it either diverts through the anastomosis or continues past it down the digestive track. The magnets pass after the anastomosis forms in a week or so.
In a 6-month feasibility study from the Czech Republic, 10 obese patients lost 28.3% of their excess weight without diet restrictions. Those with diabetes had a mean hemoglobin A1c drop of 1.8%, and normalization of fasting blood glucose levels. The procedure took just over an hour and a half after the first five cases.
“I am very excited about [this]; I really want to see where the data are going,” Dr. Edmundowicz said.
Duodenal mucosal resurfacing
The idea of the Revita System (Fractyl) is to ablate “diabetic mucosa” in the duodenum so that normal mucosa can replace it. Saline is injected endoscopically under a portion of the duodenal mucosa to lift it off the muscularis; once isolated, the mucosa is destroyed – some in the audience thought “cooked” was a better word – by exposure to a hot water balloon catheter threaded into the lumen.
Thirty-nine overweight or obese type 2 diabetics had a 1.2% improvement at 6 months from a baseline hemoglobin A1c of 9.6% in a series from Santiago, Chile. Weight loss was modest in the trial; the system is being developed for type 2 diabetics.
There is some histologic support for the notion of a diabetic mucosa with both structural and hormonal aberrations, but it’s unclear if it’s a sign or cause of sickness. Even so, “the mucosa regenerates” and won’t be diabetic “for a while” after the procedure, said investigator Manoel Galvao Neto, MD, of the Gastro Obeso Center, São Paulo.
Gastric balloons
Inflating a balloon in the stomach to make people feel full isn’t new, but the notion of putting the balloon into a capsule that patients can swallow and inflating it through a tether is a more recent notion.
The Obalon Therapeutics is one such device. In an unblinded, sham-controlled trial with 336 obese patients, subjects who got the 250-mL, nitrogen-filled Obalons – most received three – lost about 3% more of their total body weight at 24 weeks than those who did not. Although swallowed, Obalon is removed endoscopically. Meanwhile, 34 obese patients who swallowed the 550-mL, fluid-filled Elipse balloon (Allurion) had a total body weight loss of 9.5% and mean excess weight loss of 37.2% at 4 months, by which time Elipse deflates on its own and passes without endoscopic retrieval.
“This is a very promising approach. I am very excited about digested balloons,” said Dr. Edmundowicz, an investigator in the Obalon study.
Endoscopic sleeve gastroplasty
Endoscopic sleeve gastroplasty duplicates sleeve gastrectomy with stitches placed endoscopically to seal off the greater curvature of the stomach; functionally, patients are left with a narrow sleeve of a stomach. In a multicenter series presented at DDW, 242 patients had a mean total body weight loss of 19.8% at 18 months, with a low incidence of complications. “Weight loss appears to be continuing,” Dr. Edmundowicz said. Investigators used the Apollo OverStitch (Apollo Endosurgery) to place the sutures.
Aspiration therapy
With Food and Drug Administration approval on June 14, AspireAssist (Aspire Bariatrics) is probably the best known of the newer approaches. Patients drain a portion of their meals through an endoscopically placed percutaneous gastrostomy tube a half hour or so after eating. It takes 5-10 minutes. The agency is eager to keep it out of the hands of bulimics.
One-year data were reported at DDW; 111 obese AspireAssist subjects lost a mean of 37.2% of their excess weight versus 13% in 60 patients randomized to lifestyle counseling alone.
“It may not be aesthetically pleasing, but it certainly works. It’s a viable technology,” said Dr. Edmundowicz, who was an investigator.
The studies were funded by companies developing the devices and techniques. Dr. Edmundowicz has stock options, or is a consultant or researcher, Aspire, Obalon, GI Dynamics, Elira, and other firms.
Caveat emptor, all over again
Although many of these innovations will eventually achieve acceptable clinical results in trials and be adopted by both gastroenterologists and surgeons, a note of caution is in order, argues Tyler G. Hughes, MD, FACS, is clinical professor in the department of surgery and director of medical education at the Kansas University School of Medicine, Salina Campus, coeditor of ACS Surgery News.
“The recent uptick in ‘non-surgical’ obesity treatment reverberates as history gets ready to repeat itself. If surgeons don’t take the lead, two things are likely to happen. First, ‘interventionists,’ ignorant of the lessons of yesterday will introduce foreign bodies into the soft GI tract only to see these ‘safe’ devices create surgical havoc or malabsorptive disasters. Second, instead of doing a complete spectrum of obesity procedures, surgeons will be left with the worst cases and fixing complications while the ‘interventionist’ (and I do not limit that to gastroenterologists) do the easy cases at prices three times the cost of the old way. Will this really benefit the patients?
Rather than permitting a race to the newest unproven device to satisfy public clamor, while conveniently making a tidy profit for anyone able to claim the title ‘interventionist,’ bariatric surgeons need to grab the endoscope with both hands and do the research and reporting for these new procedures. It starts with the need for every surgical resident to be proficient with the GI endoscopes and ends with surgeons seeing surgery in a wider view than through an incision,” Dr. Hughes said.
SAN DIEGO – Device companies are working hard to bring obesity management to the endoscopy suite.
The field is called endobariatrics, and its goal is to fill the gap between surgery and pharmacotherapy. Drugs and lifestyle counseling don’t work for all patients, but many patients resist having surgery. These devices are meant to fill the need for nonsurgical, “noninvasive” solutions to obesity management.
Endobariatrics has the potential to be a boon for both obese patients and gastroenterology practices. Surgeons, however, approach these innovations with caution, due in part to past experiences of rescuing patients with failed devices. “Some innovations get disseminated without adequate studies to demonstrate whether they are superior, or even equal in efficacy and safety to, well-established procedures for the same condition. Even when they have been conducted, negative studies showing no difference may end up being rejected by journals and not published,” noted Karen Deveney, MD, FACS, professor of surgery, vice-chair for education in the department of surgery at Oregon Health & Science University, Portland, and co-Editor of ACS Surgery News.
Several new investigational devices and approaches were showcased at the annual Digestive Disease Week; some “are beginning to approach the kind of results we see with surgical techniques,” said Steven Edmundowicz, MD, medical director of the University of Colorado Digestive Health Center, Aurora.
“We are seeing a tremendous amount of development in this space, but it’s early, and we have to be cautious,” he said. There have already been a few disappointments, including the EndoBarrier, a fluoropolymer liner anchored in the duodenal bulb and unfurled down the duodenum to block food absorption. A key U.S. trial was recently halted due to liver abscesses.
Dr. Edmundowicz reviewed the latest developments presented at DDW.Self-assembling magnets for dual-path enteral anastomoses
The goal of the GI Windows system is to create a partial jejunoileal, side to side bypass without surgery. A 28-mm magnet ring is introduced to the ileum by colonoscopy, and a second ring to the jejunum by endoscopy. The rings snap together and tissue caught between them dies from pressure necrosis, leaving patients with a jejunoileal communication. Once food reaches that point, it either diverts through the anastomosis or continues past it down the digestive track. The magnets pass after the anastomosis forms in a week or so.
In a 6-month feasibility study from the Czech Republic, 10 obese patients lost 28.3% of their excess weight without diet restrictions. Those with diabetes had a mean hemoglobin A1c drop of 1.8%, and normalization of fasting blood glucose levels. The procedure took just over an hour and a half after the first five cases.
“I am very excited about [this]; I really want to see where the data are going,” Dr. Edmundowicz said.
Duodenal mucosal resurfacing
The idea of the Revita System (Fractyl) is to ablate “diabetic mucosa” in the duodenum so that normal mucosa can replace it. Saline is injected endoscopically under a portion of the duodenal mucosa to lift it off the muscularis; once isolated, the mucosa is destroyed – some in the audience thought “cooked” was a better word – by exposure to a hot water balloon catheter threaded into the lumen.
Thirty-nine overweight or obese type 2 diabetics had a 1.2% improvement at 6 months from a baseline hemoglobin A1c of 9.6% in a series from Santiago, Chile. Weight loss was modest in the trial; the system is being developed for type 2 diabetics.
There is some histologic support for the notion of a diabetic mucosa with both structural and hormonal aberrations, but it’s unclear if it’s a sign or cause of sickness. Even so, “the mucosa regenerates” and won’t be diabetic “for a while” after the procedure, said investigator Manoel Galvao Neto, MD, of the Gastro Obeso Center, São Paulo.
Gastric balloons
Inflating a balloon in the stomach to make people feel full isn’t new, but the notion of putting the balloon into a capsule that patients can swallow and inflating it through a tether is a more recent notion.
The Obalon Therapeutics is one such device. In an unblinded, sham-controlled trial with 336 obese patients, subjects who got the 250-mL, nitrogen-filled Obalons – most received three – lost about 3% more of their total body weight at 24 weeks than those who did not. Although swallowed, Obalon is removed endoscopically. Meanwhile, 34 obese patients who swallowed the 550-mL, fluid-filled Elipse balloon (Allurion) had a total body weight loss of 9.5% and mean excess weight loss of 37.2% at 4 months, by which time Elipse deflates on its own and passes without endoscopic retrieval.
“This is a very promising approach. I am very excited about digested balloons,” said Dr. Edmundowicz, an investigator in the Obalon study.
Endoscopic sleeve gastroplasty
Endoscopic sleeve gastroplasty duplicates sleeve gastrectomy with stitches placed endoscopically to seal off the greater curvature of the stomach; functionally, patients are left with a narrow sleeve of a stomach. In a multicenter series presented at DDW, 242 patients had a mean total body weight loss of 19.8% at 18 months, with a low incidence of complications. “Weight loss appears to be continuing,” Dr. Edmundowicz said. Investigators used the Apollo OverStitch (Apollo Endosurgery) to place the sutures.
Aspiration therapy
With Food and Drug Administration approval on June 14, AspireAssist (Aspire Bariatrics) is probably the best known of the newer approaches. Patients drain a portion of their meals through an endoscopically placed percutaneous gastrostomy tube a half hour or so after eating. It takes 5-10 minutes. The agency is eager to keep it out of the hands of bulimics.
One-year data were reported at DDW; 111 obese AspireAssist subjects lost a mean of 37.2% of their excess weight versus 13% in 60 patients randomized to lifestyle counseling alone.
“It may not be aesthetically pleasing, but it certainly works. It’s a viable technology,” said Dr. Edmundowicz, who was an investigator.
The studies were funded by companies developing the devices and techniques. Dr. Edmundowicz has stock options, or is a consultant or researcher, Aspire, Obalon, GI Dynamics, Elira, and other firms.
Caveat emptor, all over again
Although many of these innovations will eventually achieve acceptable clinical results in trials and be adopted by both gastroenterologists and surgeons, a note of caution is in order, argues Tyler G. Hughes, MD, FACS, is clinical professor in the department of surgery and director of medical education at the Kansas University School of Medicine, Salina Campus, coeditor of ACS Surgery News.
“The recent uptick in ‘non-surgical’ obesity treatment reverberates as history gets ready to repeat itself. If surgeons don’t take the lead, two things are likely to happen. First, ‘interventionists,’ ignorant of the lessons of yesterday will introduce foreign bodies into the soft GI tract only to see these ‘safe’ devices create surgical havoc or malabsorptive disasters. Second, instead of doing a complete spectrum of obesity procedures, surgeons will be left with the worst cases and fixing complications while the ‘interventionist’ (and I do not limit that to gastroenterologists) do the easy cases at prices three times the cost of the old way. Will this really benefit the patients?
Rather than permitting a race to the newest unproven device to satisfy public clamor, while conveniently making a tidy profit for anyone able to claim the title ‘interventionist,’ bariatric surgeons need to grab the endoscope with both hands and do the research and reporting for these new procedures. It starts with the need for every surgical resident to be proficient with the GI endoscopes and ends with surgeons seeing surgery in a wider view than through an incision,” Dr. Hughes said.
SAN DIEGO – Device companies are working hard to bring obesity management to the endoscopy suite.
The field is called endobariatrics, and its goal is to fill the gap between surgery and pharmacotherapy. Drugs and lifestyle counseling don’t work for all patients, but many patients resist having surgery. These devices are meant to fill the need for nonsurgical, “noninvasive” solutions to obesity management.
Endobariatrics has the potential to be a boon for both obese patients and gastroenterology practices. Surgeons, however, approach these innovations with caution, due in part to past experiences of rescuing patients with failed devices. “Some innovations get disseminated without adequate studies to demonstrate whether they are superior, or even equal in efficacy and safety to, well-established procedures for the same condition. Even when they have been conducted, negative studies showing no difference may end up being rejected by journals and not published,” noted Karen Deveney, MD, FACS, professor of surgery, vice-chair for education in the department of surgery at Oregon Health & Science University, Portland, and co-Editor of ACS Surgery News.
Several new investigational devices and approaches were showcased at the annual Digestive Disease Week; some “are beginning to approach the kind of results we see with surgical techniques,” said Steven Edmundowicz, MD, medical director of the University of Colorado Digestive Health Center, Aurora.
“We are seeing a tremendous amount of development in this space, but it’s early, and we have to be cautious,” he said. There have already been a few disappointments, including the EndoBarrier, a fluoropolymer liner anchored in the duodenal bulb and unfurled down the duodenum to block food absorption. A key U.S. trial was recently halted due to liver abscesses.
Dr. Edmundowicz reviewed the latest developments presented at DDW.Self-assembling magnets for dual-path enteral anastomoses
The goal of the GI Windows system is to create a partial jejunoileal, side to side bypass without surgery. A 28-mm magnet ring is introduced to the ileum by colonoscopy, and a second ring to the jejunum by endoscopy. The rings snap together and tissue caught between them dies from pressure necrosis, leaving patients with a jejunoileal communication. Once food reaches that point, it either diverts through the anastomosis or continues past it down the digestive track. The magnets pass after the anastomosis forms in a week or so.
In a 6-month feasibility study from the Czech Republic, 10 obese patients lost 28.3% of their excess weight without diet restrictions. Those with diabetes had a mean hemoglobin A1c drop of 1.8%, and normalization of fasting blood glucose levels. The procedure took just over an hour and a half after the first five cases.
“I am very excited about [this]; I really want to see where the data are going,” Dr. Edmundowicz said.
Duodenal mucosal resurfacing
The idea of the Revita System (Fractyl) is to ablate “diabetic mucosa” in the duodenum so that normal mucosa can replace it. Saline is injected endoscopically under a portion of the duodenal mucosa to lift it off the muscularis; once isolated, the mucosa is destroyed – some in the audience thought “cooked” was a better word – by exposure to a hot water balloon catheter threaded into the lumen.
Thirty-nine overweight or obese type 2 diabetics had a 1.2% improvement at 6 months from a baseline hemoglobin A1c of 9.6% in a series from Santiago, Chile. Weight loss was modest in the trial; the system is being developed for type 2 diabetics.
There is some histologic support for the notion of a diabetic mucosa with both structural and hormonal aberrations, but it’s unclear if it’s a sign or cause of sickness. Even so, “the mucosa regenerates” and won’t be diabetic “for a while” after the procedure, said investigator Manoel Galvao Neto, MD, of the Gastro Obeso Center, São Paulo.
Gastric balloons
Inflating a balloon in the stomach to make people feel full isn’t new, but the notion of putting the balloon into a capsule that patients can swallow and inflating it through a tether is a more recent notion.
The Obalon Therapeutics is one such device. In an unblinded, sham-controlled trial with 336 obese patients, subjects who got the 250-mL, nitrogen-filled Obalons – most received three – lost about 3% more of their total body weight at 24 weeks than those who did not. Although swallowed, Obalon is removed endoscopically. Meanwhile, 34 obese patients who swallowed the 550-mL, fluid-filled Elipse balloon (Allurion) had a total body weight loss of 9.5% and mean excess weight loss of 37.2% at 4 months, by which time Elipse deflates on its own and passes without endoscopic retrieval.
“This is a very promising approach. I am very excited about digested balloons,” said Dr. Edmundowicz, an investigator in the Obalon study.
Endoscopic sleeve gastroplasty
Endoscopic sleeve gastroplasty duplicates sleeve gastrectomy with stitches placed endoscopically to seal off the greater curvature of the stomach; functionally, patients are left with a narrow sleeve of a stomach. In a multicenter series presented at DDW, 242 patients had a mean total body weight loss of 19.8% at 18 months, with a low incidence of complications. “Weight loss appears to be continuing,” Dr. Edmundowicz said. Investigators used the Apollo OverStitch (Apollo Endosurgery) to place the sutures.
Aspiration therapy
With Food and Drug Administration approval on June 14, AspireAssist (Aspire Bariatrics) is probably the best known of the newer approaches. Patients drain a portion of their meals through an endoscopically placed percutaneous gastrostomy tube a half hour or so after eating. It takes 5-10 minutes. The agency is eager to keep it out of the hands of bulimics.
One-year data were reported at DDW; 111 obese AspireAssist subjects lost a mean of 37.2% of their excess weight versus 13% in 60 patients randomized to lifestyle counseling alone.
“It may not be aesthetically pleasing, but it certainly works. It’s a viable technology,” said Dr. Edmundowicz, who was an investigator.
The studies were funded by companies developing the devices and techniques. Dr. Edmundowicz has stock options, or is a consultant or researcher, Aspire, Obalon, GI Dynamics, Elira, and other firms.
Caveat emptor, all over again
Although many of these innovations will eventually achieve acceptable clinical results in trials and be adopted by both gastroenterologists and surgeons, a note of caution is in order, argues Tyler G. Hughes, MD, FACS, is clinical professor in the department of surgery and director of medical education at the Kansas University School of Medicine, Salina Campus, coeditor of ACS Surgery News.
“The recent uptick in ‘non-surgical’ obesity treatment reverberates as history gets ready to repeat itself. If surgeons don’t take the lead, two things are likely to happen. First, ‘interventionists,’ ignorant of the lessons of yesterday will introduce foreign bodies into the soft GI tract only to see these ‘safe’ devices create surgical havoc or malabsorptive disasters. Second, instead of doing a complete spectrum of obesity procedures, surgeons will be left with the worst cases and fixing complications while the ‘interventionist’ (and I do not limit that to gastroenterologists) do the easy cases at prices three times the cost of the old way. Will this really benefit the patients?
Rather than permitting a race to the newest unproven device to satisfy public clamor, while conveniently making a tidy profit for anyone able to claim the title ‘interventionist,’ bariatric surgeons need to grab the endoscope with both hands and do the research and reporting for these new procedures. It starts with the need for every surgical resident to be proficient with the GI endoscopes and ends with surgeons seeing surgery in a wider view than through an incision,” Dr. Hughes said.
AT DDW 2016
Commentary: New treatments for new patients – A call to action for bariatric surgeons
How many common bile duct operations have you done lately? How about laparotomies for bleeding duodenal ulcers? How about central venous access catheters? How many procedures that you as a general surgeon may have done a few decades ago are now done primarily by endoscopists, radiologists, or others using minimal access for their procedures?
Now hold that thought, as we turn to the field of bariatric surgery.
Twenty years ago, in 1996, there were approximately 12,000 bariatric operations performed in the United States. From 1998 to 2004, that number increased to almost 136,000 per year (J Am Coll Surg. 2011;213:261-6). What was the reason? While other factors likely had some influence, the overwhelming factor was that laparoscopic bariatric surgery became available. Patients perceived this approach as less invasive. Primary care referring physicians did as well.
The rapid rise in laparoscopy boosted the popularity of bariatric surgery but since that bump, the bariatric surgery numbers have remained flat. Currently, less than 2% of eligible patients who are morbidly obese opt for surgical treatment each year – despite the fact that the safety of bariatric surgery has dramatically improved over the past 15 years. Currently the mortality rate for laparoscopic gastric bypass is at 0.15% (Ann Surg. 2014;259:123-30) and sleeve gastrectomy mortality is lower than that. Only appendectomy has a lower mortality rate among major abdominal operations. Despite the safety record, and despite over a decade of publications demonstrating the effectiveness of bariatric surgery in prolonging life, improving or eliminating comorbid medical problems of obesity, improving the quality of life of patients, and decreasing the cost of their medical care, there still has been no major new shift toward surgery by patients who would benefit from it.
What we are offering is not what these patients want. We as bariatric and metabolic surgeons must face the reality of that fact.
While endoscopic bariatric procedures are not new, their use to date has been limited to modifying existing operations, such as narrowing the anastomosis after gastric bypass for patients who are regaining weight. Such procedures have enjoyed at best mild to moderate short-term success, but poor long-term success.
The performance of a successful endoscopic sleeve gastrectomy, however, is a different issue. The sleeve gastrectomy has rapidly become the most popular bariatric operation. Its successful performance endoscopically (Endoscopy. 2015;47:164-6) should serve notice to bariatric surgeons that the time has come to learn to do endoscopic bariatric surgery. If effective, it almost certainly will be what patients seek in the future.
Societal stigmas, patient expectations, and our culture all drive the perception that obesity is a problem that individuals should be able to solve on their own. It is this firmly entrenched belief that is the foundation of the multi-billion dollar diet products industry. Yet the concept of having surgery to treat severe obesity is one that most severely obese patients do not easily embrace. A first-hand successful experience of a friend or relative is often needed for these individuals to consider a surgical procedure. While most patients with ultrasound-proven gallstones who are symptomatic will be referred to a surgeon by their primary care physician, how often is this true for the patient with a body mass index over 35?
The appeal of endoscopic procedures for patients and referring physicians is, of course, that these procedures are perceived as not really surgery. They are minimally invasive endoscopic procedures. The risk profile is very low. Why not consider it? patients may ask themselves. After all, you are not really having surgery.
Many of my bariatric colleagues will likely disagree with this recommendation to embrace endoscopic procedures. After all, the track record to date of many of these procedures and devices has not been impressive. All devices to date that have involved endoscopic treatment of obesity have either failed and been removed from the market, or are in their infancy still looking to establish efficacy (Clin Gastroenterol Hepatol. 2016;14:507-15). I have been vocally critical at symposia and national meetings of the concept of using an endoscopic suturing device to narrow the anastomosis of patients with a gastric bypass who are regaining weight. I have argued that the procedure is doomed to long-term minimal success or more likely flat-out failure. However, such a conservative approach that demands proof of long-term efficacy could predictably place us conservative curmudgeons on the sidelines of treating obesity when successful endoscopic procedures are available and become the sought-after option, just as laparoscopy became the sought-after option 17 years ago. How many surgeons offered primarily open bariatric operations after about 2005?
If we are to reach more than 1%-2% of the eligible patients who would benefit from treatment for morbid obesity and its related medical problems, then we need to take a different approach. We have about maximized how well we can do the surgical approach we now offer. It isn’t gaining in popularity among those who need it most. While we should not abandon the procedures that have been proven so effective, we should embrace new options for our patients.
It is certainly possible that the stigma of having surgery will resolve if a patient has an endoscopic procedure that is successful but only transiently. A more definitive operative procedure then may follow. Is that not ultimately better for that patient than having him or her never pursue a treatment for obesity? While this argument seems appropriate for the long-term overall patient good, the short-term increase in cost may make it a difficult sell to payers. But insurance companies are already doing their very best not to pay for highly medically effective and proven cost-effective bariatric surgery now. Only public pressure will force them to change – perhaps the public pressure of demand for endoscopic procedures.
Bariatric surgeons: It’s time to become bariatric endoscopists. If we want new patients, we need to adopt new treatments.
Dr. Schirmer is the Stephen H. Watts Professor of Surgery at the University of Virginia Health Sciences Center, Charlottesville.
How many common bile duct operations have you done lately? How about laparotomies for bleeding duodenal ulcers? How about central venous access catheters? How many procedures that you as a general surgeon may have done a few decades ago are now done primarily by endoscopists, radiologists, or others using minimal access for their procedures?
Now hold that thought, as we turn to the field of bariatric surgery.
Twenty years ago, in 1996, there were approximately 12,000 bariatric operations performed in the United States. From 1998 to 2004, that number increased to almost 136,000 per year (J Am Coll Surg. 2011;213:261-6). What was the reason? While other factors likely had some influence, the overwhelming factor was that laparoscopic bariatric surgery became available. Patients perceived this approach as less invasive. Primary care referring physicians did as well.
The rapid rise in laparoscopy boosted the popularity of bariatric surgery but since that bump, the bariatric surgery numbers have remained flat. Currently, less than 2% of eligible patients who are morbidly obese opt for surgical treatment each year – despite the fact that the safety of bariatric surgery has dramatically improved over the past 15 years. Currently the mortality rate for laparoscopic gastric bypass is at 0.15% (Ann Surg. 2014;259:123-30) and sleeve gastrectomy mortality is lower than that. Only appendectomy has a lower mortality rate among major abdominal operations. Despite the safety record, and despite over a decade of publications demonstrating the effectiveness of bariatric surgery in prolonging life, improving or eliminating comorbid medical problems of obesity, improving the quality of life of patients, and decreasing the cost of their medical care, there still has been no major new shift toward surgery by patients who would benefit from it.
What we are offering is not what these patients want. We as bariatric and metabolic surgeons must face the reality of that fact.
While endoscopic bariatric procedures are not new, their use to date has been limited to modifying existing operations, such as narrowing the anastomosis after gastric bypass for patients who are regaining weight. Such procedures have enjoyed at best mild to moderate short-term success, but poor long-term success.
The performance of a successful endoscopic sleeve gastrectomy, however, is a different issue. The sleeve gastrectomy has rapidly become the most popular bariatric operation. Its successful performance endoscopically (Endoscopy. 2015;47:164-6) should serve notice to bariatric surgeons that the time has come to learn to do endoscopic bariatric surgery. If effective, it almost certainly will be what patients seek in the future.
Societal stigmas, patient expectations, and our culture all drive the perception that obesity is a problem that individuals should be able to solve on their own. It is this firmly entrenched belief that is the foundation of the multi-billion dollar diet products industry. Yet the concept of having surgery to treat severe obesity is one that most severely obese patients do not easily embrace. A first-hand successful experience of a friend or relative is often needed for these individuals to consider a surgical procedure. While most patients with ultrasound-proven gallstones who are symptomatic will be referred to a surgeon by their primary care physician, how often is this true for the patient with a body mass index over 35?
The appeal of endoscopic procedures for patients and referring physicians is, of course, that these procedures are perceived as not really surgery. They are minimally invasive endoscopic procedures. The risk profile is very low. Why not consider it? patients may ask themselves. After all, you are not really having surgery.
Many of my bariatric colleagues will likely disagree with this recommendation to embrace endoscopic procedures. After all, the track record to date of many of these procedures and devices has not been impressive. All devices to date that have involved endoscopic treatment of obesity have either failed and been removed from the market, or are in their infancy still looking to establish efficacy (Clin Gastroenterol Hepatol. 2016;14:507-15). I have been vocally critical at symposia and national meetings of the concept of using an endoscopic suturing device to narrow the anastomosis of patients with a gastric bypass who are regaining weight. I have argued that the procedure is doomed to long-term minimal success or more likely flat-out failure. However, such a conservative approach that demands proof of long-term efficacy could predictably place us conservative curmudgeons on the sidelines of treating obesity when successful endoscopic procedures are available and become the sought-after option, just as laparoscopy became the sought-after option 17 years ago. How many surgeons offered primarily open bariatric operations after about 2005?
If we are to reach more than 1%-2% of the eligible patients who would benefit from treatment for morbid obesity and its related medical problems, then we need to take a different approach. We have about maximized how well we can do the surgical approach we now offer. It isn’t gaining in popularity among those who need it most. While we should not abandon the procedures that have been proven so effective, we should embrace new options for our patients.
It is certainly possible that the stigma of having surgery will resolve if a patient has an endoscopic procedure that is successful but only transiently. A more definitive operative procedure then may follow. Is that not ultimately better for that patient than having him or her never pursue a treatment for obesity? While this argument seems appropriate for the long-term overall patient good, the short-term increase in cost may make it a difficult sell to payers. But insurance companies are already doing their very best not to pay for highly medically effective and proven cost-effective bariatric surgery now. Only public pressure will force them to change – perhaps the public pressure of demand for endoscopic procedures.
Bariatric surgeons: It’s time to become bariatric endoscopists. If we want new patients, we need to adopt new treatments.
Dr. Schirmer is the Stephen H. Watts Professor of Surgery at the University of Virginia Health Sciences Center, Charlottesville.
How many common bile duct operations have you done lately? How about laparotomies for bleeding duodenal ulcers? How about central venous access catheters? How many procedures that you as a general surgeon may have done a few decades ago are now done primarily by endoscopists, radiologists, or others using minimal access for their procedures?
Now hold that thought, as we turn to the field of bariatric surgery.
Twenty years ago, in 1996, there were approximately 12,000 bariatric operations performed in the United States. From 1998 to 2004, that number increased to almost 136,000 per year (J Am Coll Surg. 2011;213:261-6). What was the reason? While other factors likely had some influence, the overwhelming factor was that laparoscopic bariatric surgery became available. Patients perceived this approach as less invasive. Primary care referring physicians did as well.
The rapid rise in laparoscopy boosted the popularity of bariatric surgery but since that bump, the bariatric surgery numbers have remained flat. Currently, less than 2% of eligible patients who are morbidly obese opt for surgical treatment each year – despite the fact that the safety of bariatric surgery has dramatically improved over the past 15 years. Currently the mortality rate for laparoscopic gastric bypass is at 0.15% (Ann Surg. 2014;259:123-30) and sleeve gastrectomy mortality is lower than that. Only appendectomy has a lower mortality rate among major abdominal operations. Despite the safety record, and despite over a decade of publications demonstrating the effectiveness of bariatric surgery in prolonging life, improving or eliminating comorbid medical problems of obesity, improving the quality of life of patients, and decreasing the cost of their medical care, there still has been no major new shift toward surgery by patients who would benefit from it.
What we are offering is not what these patients want. We as bariatric and metabolic surgeons must face the reality of that fact.
While endoscopic bariatric procedures are not new, their use to date has been limited to modifying existing operations, such as narrowing the anastomosis after gastric bypass for patients who are regaining weight. Such procedures have enjoyed at best mild to moderate short-term success, but poor long-term success.
The performance of a successful endoscopic sleeve gastrectomy, however, is a different issue. The sleeve gastrectomy has rapidly become the most popular bariatric operation. Its successful performance endoscopically (Endoscopy. 2015;47:164-6) should serve notice to bariatric surgeons that the time has come to learn to do endoscopic bariatric surgery. If effective, it almost certainly will be what patients seek in the future.
Societal stigmas, patient expectations, and our culture all drive the perception that obesity is a problem that individuals should be able to solve on their own. It is this firmly entrenched belief that is the foundation of the multi-billion dollar diet products industry. Yet the concept of having surgery to treat severe obesity is one that most severely obese patients do not easily embrace. A first-hand successful experience of a friend or relative is often needed for these individuals to consider a surgical procedure. While most patients with ultrasound-proven gallstones who are symptomatic will be referred to a surgeon by their primary care physician, how often is this true for the patient with a body mass index over 35?
The appeal of endoscopic procedures for patients and referring physicians is, of course, that these procedures are perceived as not really surgery. They are minimally invasive endoscopic procedures. The risk profile is very low. Why not consider it? patients may ask themselves. After all, you are not really having surgery.
Many of my bariatric colleagues will likely disagree with this recommendation to embrace endoscopic procedures. After all, the track record to date of many of these procedures and devices has not been impressive. All devices to date that have involved endoscopic treatment of obesity have either failed and been removed from the market, or are in their infancy still looking to establish efficacy (Clin Gastroenterol Hepatol. 2016;14:507-15). I have been vocally critical at symposia and national meetings of the concept of using an endoscopic suturing device to narrow the anastomosis of patients with a gastric bypass who are regaining weight. I have argued that the procedure is doomed to long-term minimal success or more likely flat-out failure. However, such a conservative approach that demands proof of long-term efficacy could predictably place us conservative curmudgeons on the sidelines of treating obesity when successful endoscopic procedures are available and become the sought-after option, just as laparoscopy became the sought-after option 17 years ago. How many surgeons offered primarily open bariatric operations after about 2005?
If we are to reach more than 1%-2% of the eligible patients who would benefit from treatment for morbid obesity and its related medical problems, then we need to take a different approach. We have about maximized how well we can do the surgical approach we now offer. It isn’t gaining in popularity among those who need it most. While we should not abandon the procedures that have been proven so effective, we should embrace new options for our patients.
It is certainly possible that the stigma of having surgery will resolve if a patient has an endoscopic procedure that is successful but only transiently. A more definitive operative procedure then may follow. Is that not ultimately better for that patient than having him or her never pursue a treatment for obesity? While this argument seems appropriate for the long-term overall patient good, the short-term increase in cost may make it a difficult sell to payers. But insurance companies are already doing their very best not to pay for highly medically effective and proven cost-effective bariatric surgery now. Only public pressure will force them to change – perhaps the public pressure of demand for endoscopic procedures.
Bariatric surgeons: It’s time to become bariatric endoscopists. If we want new patients, we need to adopt new treatments.
Dr. Schirmer is the Stephen H. Watts Professor of Surgery at the University of Virginia Health Sciences Center, Charlottesville.
The future of surgery
Predicting the future has been a favorite topic of surgeons through the ages for addresses to august surgical societies.
Confident predictions of the future of surgery, however, have not always stood the test of time. Speaking at the University of Manchester’s Centenary celebration in 1973 at an international symposium on “Medicine in the 21st Century,” the noted surgeon J. Englebert Dunphy correctly predicted the prominence of joint-replacement procedures, but incorrectly asserted that medical advances would virtually eliminate the need for cholecystectomy through dissolution of gallstones and the need for surgical approaches to atherosclerosis through plaque prevention and dissolution. He accurately predicted that infections and sepsis would remain serious problems. But he missed the mark when he predicted that surgical pain would be eliminated by a pill that would block somatic nerve impulses without any respiratory or circulatory effects (Surgery. 1974 Mar;75:332-7). Technologic advances such as laparoscopic cholecystectomy, which emerged less than 20 years into the future, were not on his radar screen.
Change and disruptive technology
To be sure, the surgical procedures and methods have changed markedly since the time I trained in surgery in the 1970s. The most obvious change is the shift from large incisions to small ones, with the commensurate quick recovery and short hospital stays. This change is primarily because of the emergence of disruptive technology, a concept that has pervaded every avenue of our current lives, not just surgery: Think Uber, robotics in industry, the Internet, smartphones, and the miniaturization of just about every communication means. In medicine, these disruptive technologies have led to the emergence of the electronic health record, new imaging modalities, percutaneous interventional techniques, fiber optic endoscopy, laparoscopy, and endovascular surgery.
Maintaining capacity to do infrequent operations
During my training, H2 receptor antagonists and Helicobacter pylori came on the scene, with the result that ulcer operations almost disappeared from our surgical armamentarium; one of my most frequent operations as a senior and chief resident has become a rarity 40 years later for our trainees. And yet, a general surgeon must know how to perform an ulcer operation and which type is the best for a given circumstance, since perforated and bleeding ulcers are still seen, if infrequently. The perforated ulcer is seen most often in patients with multiple comorbidities who can least tolerate a complication and need one effective and well-executed operation if they are going to survive. How do we continue to teach residents these procedures when they have become infrequent?
There is perhaps some utility in keeping some aging surgeons around to teach residents on fresh cadavers, or to call them out of their assisted living facilities when needed for consultation! Now that the majority of operations in most places are being performed by minimally invasive surgical (MIS) methods, we may not need MIS fellowships or training much longer for trainees to become proficient in these skills, because it is the open operations that are less frequent. Our chief residents are much less secure about performing an open cholecystectomy, of which they have performed perhaps 5, than they are about performing laparoscopic cholecystectomy, of which they have performed 105.
Technologies on the verge of disrupting
Mark Aeder, MD, FACS, recently asked an important question in the ACS Communities thread on the future of surgery: What will the future disruptive innovations be? Which areas of surgery will bloom next?
There are many game-changing emerging technologies that could well turn out to be future “disruptors” of surgery as we know it. Cancer surgery is on track to be transformed by the development of genomics and personalized medicine and immunotherapy for melanoma and lung cancer.
The areas most likely to remain in the surgical realm are trauma, infections, and inflammation. Though safer cars, seatbelt laws, and helmet laws for motorcyclists have already decreased motor vehicle accidents and injury severity, we have still not produced a cure for stupidity or bad luck. Traumatic injuries will always be with us, and surgeons well trained in trauma management will continue to be needed. Appendicitis, cholecystitis, and diverticulitis will continue to require operations, even though inroads have begun with the studies showing success of antibiotic treatment for appendicitis and diverticulitis.
Keep current, avoid bandwagons
The key lesson, not only for our young surgeons-in-training but also for our seasoned surgeons, is to keep learning, keep networking, and keep adopting new techniques as soon as they show true evidence of success.
The best way for surgeons to remain prepared for whatever the future will bring is to stay current with innovations coming on the scene but not jump on the bandwagon too early and adopt new fads without substantial evidence of their soundness. A few retrospective case series reporting success with a new operation is insufficient evidence to try it out on the unsuspecting public. Although the completion of well-designed randomized trials with adequate follow-up takes time, it is better to stick with well-established and evidence-based techniques than rush to embrace an inadequately vetted procedure that may end up harming a patient.
Not all innovative devices or procedures rise to the level of being so truly “experimental” that they would require an institutional review board, and this ought to prompt an extra measure of caution by surgeons. Some “innovations,” such as the endobariatric devices reported elsewhere in this issue, represent novel variations of previous procedures but are as yet unvalidated by long-term studies. Responsible adoption of these novel procedures requires a full disclosure to the patient that the procedure or device is novel, without long-term evidence of success, and may entail unknown risks. Don’t let your enthusiasm to be an early adopter overcome your scientific and historic obligation to prevent harm to the patient.
Dr. Deveney is professor of surgery and vice chair of education in the department of surgery, Oregon Health & Science University, Portland. She is the co-Editor of ACS Surgery News.
Predicting the future has been a favorite topic of surgeons through the ages for addresses to august surgical societies.
Confident predictions of the future of surgery, however, have not always stood the test of time. Speaking at the University of Manchester’s Centenary celebration in 1973 at an international symposium on “Medicine in the 21st Century,” the noted surgeon J. Englebert Dunphy correctly predicted the prominence of joint-replacement procedures, but incorrectly asserted that medical advances would virtually eliminate the need for cholecystectomy through dissolution of gallstones and the need for surgical approaches to atherosclerosis through plaque prevention and dissolution. He accurately predicted that infections and sepsis would remain serious problems. But he missed the mark when he predicted that surgical pain would be eliminated by a pill that would block somatic nerve impulses without any respiratory or circulatory effects (Surgery. 1974 Mar;75:332-7). Technologic advances such as laparoscopic cholecystectomy, which emerged less than 20 years into the future, were not on his radar screen.
Change and disruptive technology
To be sure, the surgical procedures and methods have changed markedly since the time I trained in surgery in the 1970s. The most obvious change is the shift from large incisions to small ones, with the commensurate quick recovery and short hospital stays. This change is primarily because of the emergence of disruptive technology, a concept that has pervaded every avenue of our current lives, not just surgery: Think Uber, robotics in industry, the Internet, smartphones, and the miniaturization of just about every communication means. In medicine, these disruptive technologies have led to the emergence of the electronic health record, new imaging modalities, percutaneous interventional techniques, fiber optic endoscopy, laparoscopy, and endovascular surgery.
Maintaining capacity to do infrequent operations
During my training, H2 receptor antagonists and Helicobacter pylori came on the scene, with the result that ulcer operations almost disappeared from our surgical armamentarium; one of my most frequent operations as a senior and chief resident has become a rarity 40 years later for our trainees. And yet, a general surgeon must know how to perform an ulcer operation and which type is the best for a given circumstance, since perforated and bleeding ulcers are still seen, if infrequently. The perforated ulcer is seen most often in patients with multiple comorbidities who can least tolerate a complication and need one effective and well-executed operation if they are going to survive. How do we continue to teach residents these procedures when they have become infrequent?
There is perhaps some utility in keeping some aging surgeons around to teach residents on fresh cadavers, or to call them out of their assisted living facilities when needed for consultation! Now that the majority of operations in most places are being performed by minimally invasive surgical (MIS) methods, we may not need MIS fellowships or training much longer for trainees to become proficient in these skills, because it is the open operations that are less frequent. Our chief residents are much less secure about performing an open cholecystectomy, of which they have performed perhaps 5, than they are about performing laparoscopic cholecystectomy, of which they have performed 105.
Technologies on the verge of disrupting
Mark Aeder, MD, FACS, recently asked an important question in the ACS Communities thread on the future of surgery: What will the future disruptive innovations be? Which areas of surgery will bloom next?
There are many game-changing emerging technologies that could well turn out to be future “disruptors” of surgery as we know it. Cancer surgery is on track to be transformed by the development of genomics and personalized medicine and immunotherapy for melanoma and lung cancer.
The areas most likely to remain in the surgical realm are trauma, infections, and inflammation. Though safer cars, seatbelt laws, and helmet laws for motorcyclists have already decreased motor vehicle accidents and injury severity, we have still not produced a cure for stupidity or bad luck. Traumatic injuries will always be with us, and surgeons well trained in trauma management will continue to be needed. Appendicitis, cholecystitis, and diverticulitis will continue to require operations, even though inroads have begun with the studies showing success of antibiotic treatment for appendicitis and diverticulitis.
Keep current, avoid bandwagons
The key lesson, not only for our young surgeons-in-training but also for our seasoned surgeons, is to keep learning, keep networking, and keep adopting new techniques as soon as they show true evidence of success.
The best way for surgeons to remain prepared for whatever the future will bring is to stay current with innovations coming on the scene but not jump on the bandwagon too early and adopt new fads without substantial evidence of their soundness. A few retrospective case series reporting success with a new operation is insufficient evidence to try it out on the unsuspecting public. Although the completion of well-designed randomized trials with adequate follow-up takes time, it is better to stick with well-established and evidence-based techniques than rush to embrace an inadequately vetted procedure that may end up harming a patient.
Not all innovative devices or procedures rise to the level of being so truly “experimental” that they would require an institutional review board, and this ought to prompt an extra measure of caution by surgeons. Some “innovations,” such as the endobariatric devices reported elsewhere in this issue, represent novel variations of previous procedures but are as yet unvalidated by long-term studies. Responsible adoption of these novel procedures requires a full disclosure to the patient that the procedure or device is novel, without long-term evidence of success, and may entail unknown risks. Don’t let your enthusiasm to be an early adopter overcome your scientific and historic obligation to prevent harm to the patient.
Dr. Deveney is professor of surgery and vice chair of education in the department of surgery, Oregon Health & Science University, Portland. She is the co-Editor of ACS Surgery News.
Predicting the future has been a favorite topic of surgeons through the ages for addresses to august surgical societies.
Confident predictions of the future of surgery, however, have not always stood the test of time. Speaking at the University of Manchester’s Centenary celebration in 1973 at an international symposium on “Medicine in the 21st Century,” the noted surgeon J. Englebert Dunphy correctly predicted the prominence of joint-replacement procedures, but incorrectly asserted that medical advances would virtually eliminate the need for cholecystectomy through dissolution of gallstones and the need for surgical approaches to atherosclerosis through plaque prevention and dissolution. He accurately predicted that infections and sepsis would remain serious problems. But he missed the mark when he predicted that surgical pain would be eliminated by a pill that would block somatic nerve impulses without any respiratory or circulatory effects (Surgery. 1974 Mar;75:332-7). Technologic advances such as laparoscopic cholecystectomy, which emerged less than 20 years into the future, were not on his radar screen.
Change and disruptive technology
To be sure, the surgical procedures and methods have changed markedly since the time I trained in surgery in the 1970s. The most obvious change is the shift from large incisions to small ones, with the commensurate quick recovery and short hospital stays. This change is primarily because of the emergence of disruptive technology, a concept that has pervaded every avenue of our current lives, not just surgery: Think Uber, robotics in industry, the Internet, smartphones, and the miniaturization of just about every communication means. In medicine, these disruptive technologies have led to the emergence of the electronic health record, new imaging modalities, percutaneous interventional techniques, fiber optic endoscopy, laparoscopy, and endovascular surgery.
Maintaining capacity to do infrequent operations
During my training, H2 receptor antagonists and Helicobacter pylori came on the scene, with the result that ulcer operations almost disappeared from our surgical armamentarium; one of my most frequent operations as a senior and chief resident has become a rarity 40 years later for our trainees. And yet, a general surgeon must know how to perform an ulcer operation and which type is the best for a given circumstance, since perforated and bleeding ulcers are still seen, if infrequently. The perforated ulcer is seen most often in patients with multiple comorbidities who can least tolerate a complication and need one effective and well-executed operation if they are going to survive. How do we continue to teach residents these procedures when they have become infrequent?
There is perhaps some utility in keeping some aging surgeons around to teach residents on fresh cadavers, or to call them out of their assisted living facilities when needed for consultation! Now that the majority of operations in most places are being performed by minimally invasive surgical (MIS) methods, we may not need MIS fellowships or training much longer for trainees to become proficient in these skills, because it is the open operations that are less frequent. Our chief residents are much less secure about performing an open cholecystectomy, of which they have performed perhaps 5, than they are about performing laparoscopic cholecystectomy, of which they have performed 105.
Technologies on the verge of disrupting
Mark Aeder, MD, FACS, recently asked an important question in the ACS Communities thread on the future of surgery: What will the future disruptive innovations be? Which areas of surgery will bloom next?
There are many game-changing emerging technologies that could well turn out to be future “disruptors” of surgery as we know it. Cancer surgery is on track to be transformed by the development of genomics and personalized medicine and immunotherapy for melanoma and lung cancer.
The areas most likely to remain in the surgical realm are trauma, infections, and inflammation. Though safer cars, seatbelt laws, and helmet laws for motorcyclists have already decreased motor vehicle accidents and injury severity, we have still not produced a cure for stupidity or bad luck. Traumatic injuries will always be with us, and surgeons well trained in trauma management will continue to be needed. Appendicitis, cholecystitis, and diverticulitis will continue to require operations, even though inroads have begun with the studies showing success of antibiotic treatment for appendicitis and diverticulitis.
Keep current, avoid bandwagons
The key lesson, not only for our young surgeons-in-training but also for our seasoned surgeons, is to keep learning, keep networking, and keep adopting new techniques as soon as they show true evidence of success.
The best way for surgeons to remain prepared for whatever the future will bring is to stay current with innovations coming on the scene but not jump on the bandwagon too early and adopt new fads without substantial evidence of their soundness. A few retrospective case series reporting success with a new operation is insufficient evidence to try it out on the unsuspecting public. Although the completion of well-designed randomized trials with adequate follow-up takes time, it is better to stick with well-established and evidence-based techniques than rush to embrace an inadequately vetted procedure that may end up harming a patient.
Not all innovative devices or procedures rise to the level of being so truly “experimental” that they would require an institutional review board, and this ought to prompt an extra measure of caution by surgeons. Some “innovations,” such as the endobariatric devices reported elsewhere in this issue, represent novel variations of previous procedures but are as yet unvalidated by long-term studies. Responsible adoption of these novel procedures requires a full disclosure to the patient that the procedure or device is novel, without long-term evidence of success, and may entail unknown risks. Don’t let your enthusiasm to be an early adopter overcome your scientific and historic obligation to prevent harm to the patient.
Dr. Deveney is professor of surgery and vice chair of education in the department of surgery, Oregon Health & Science University, Portland. She is the co-Editor of ACS Surgery News.
Coming your way ... MIPS
In this column, I will return to the topic of the Quality Payment Program (QPP), Centers for Medicare & Medicaid Services’ new payment program, currently scheduled to go into effect January 2017. The QPP has two tracks, the Merit-Based Incentive Payment System (MIPS) and advanced Alternative Payment Models (APMs). Again, it is widely expected that for the first several years of the program, the vast majority of surgeons will participate in the QPP via the MIPS track.
To briefly review from the July edition of this column, MIPS consists of four components:
1) Quality.
2) Resource Use.
3) Advancing Care Information (ACI).
4) Clinical Practice Improvement Activities (CPIA).
The Quality component replaces the Physician Quality Reporting System (PQRS), the Resource Use component replaces the Value-Based Modifier (VBM), the Advancing Care Information (ACI) component replaces the Electronic Health Record Meaningful Use (EHR-MU) program and is proposed to simplify the program by modifying requirements. The fourth component of MIPS, the Clinical Practice Improvement Activities (CPIA), is new.
MIPS participants will be assigned a Composite Performance Score based on their performance in all four components. As proposed for 2017, 50% of the MIPS Composite Performance Score will be based on performance in the Quality component, 25% will be based on the ACI component, 15% will be based on the CPIA, and 10% will be based on Resource Use. Thus, 75% of one’s score will be determined by performance in the Quality and ACI components. With that in mind, the remainder of this month’s column will be dedicated to the ACI with next month’s column focusing on the Quality component.
The Advancing Care Component (ACI)
As discussed above, the ACI component modifies and replaces the Electronic Health Record Meaningful Use (EHR-MU) program. As currently proposed, (and subject to change with release of the final rule in early November 2016), the score for ACI will be derived in two parts: a base score (50 points) and a performance score.
The threshold for achieving the base score remains an “all or nothing” proposition. Achieving the threshold is based upon reporting a yes/no statement OR the appropriate numerator and denominator for each measure, as required, for the following six objectives:
1) Protect Patient Health Information.
2) Electronic Prescribing.
3) Patient Electronic Access to Health Information.
4) Coordination of Care through Patient Engagement.
5) Health Information Exchange.
6) Public Health and Clinical Data Reporting.
Only after meeting the requirements for the base score is one eligible to receive additional performance score credit based on the level of performance on a subset of three of the objectives required to achieve the base score. Those three are:
1) Patient Electronic Access to Health Information.
2) Coordination of Care through Patient Engagement.
3) Health Information Exchange
There are a total of eight measures for these three objectives.
The two measures specific to the Patient Electronic Access objective are:
a) Patient Access Measure.
b) Patient-Specific Education Measure.
Coordination of Care through Patient Engagement is assessed based upon the following three measures:
a) View, Download, Transmit Measure.
b) Secure Messaging Measure.
c) Patient-Generated Health Data Measures.
For the Health Information Exchange objective, assessment is also based on three measures. They are:
a) Patient Record Exchange Measure.
b) Request/Accept Patient Care Record Measure.
c) Clinical Information Reconciliation Measure.
Scoring for each of these eight measures is based upon reporting both a numerator and denominator, thus facilitating the calculation of percentage score to which a point value is assigned. For example, a performance rate of 85 on one of the listed measures would earn 8.5 points toward the performance score. The sum of the points accumulated for these eight measures is added to Threshold Score to determine the final score for ACI. Again, 25% of one’s MIPS Composite Score is derived from performance in the ACI component.
It warrants repeating and re-emphasis that the above information is subject to modification pending the release of the MIPS Final Rule, expected in early November.
As mentioned above, the Quality component of MIPS replaces the PQRS. Quality assessment will comprise 50% of the MIPS Composite Score. In next month’s column, I will provide an outline of the changes CMS is proposing for the Quality assessment. As a preview, some good news can be found in the fact that the current MIPS Quality component proposal will require providers to report on a total of six measures as opposed to the previous PQRS requirement to report on nine.
Until next month …
In this column, I will return to the topic of the Quality Payment Program (QPP), Centers for Medicare & Medicaid Services’ new payment program, currently scheduled to go into effect January 2017. The QPP has two tracks, the Merit-Based Incentive Payment System (MIPS) and advanced Alternative Payment Models (APMs). Again, it is widely expected that for the first several years of the program, the vast majority of surgeons will participate in the QPP via the MIPS track.
To briefly review from the July edition of this column, MIPS consists of four components:
1) Quality.
2) Resource Use.
3) Advancing Care Information (ACI).
4) Clinical Practice Improvement Activities (CPIA).
The Quality component replaces the Physician Quality Reporting System (PQRS), the Resource Use component replaces the Value-Based Modifier (VBM), the Advancing Care Information (ACI) component replaces the Electronic Health Record Meaningful Use (EHR-MU) program and is proposed to simplify the program by modifying requirements. The fourth component of MIPS, the Clinical Practice Improvement Activities (CPIA), is new.
MIPS participants will be assigned a Composite Performance Score based on their performance in all four components. As proposed for 2017, 50% of the MIPS Composite Performance Score will be based on performance in the Quality component, 25% will be based on the ACI component, 15% will be based on the CPIA, and 10% will be based on Resource Use. Thus, 75% of one’s score will be determined by performance in the Quality and ACI components. With that in mind, the remainder of this month’s column will be dedicated to the ACI with next month’s column focusing on the Quality component.
The Advancing Care Component (ACI)
As discussed above, the ACI component modifies and replaces the Electronic Health Record Meaningful Use (EHR-MU) program. As currently proposed, (and subject to change with release of the final rule in early November 2016), the score for ACI will be derived in two parts: a base score (50 points) and a performance score.

The threshold for achieving the base score remains an “all or nothing” proposition. Achieving the threshold is based upon reporting a yes/no statement OR the appropriate numerator and denominator for each measure, as required, for the following six objectives:
1) Protect Patient Health Information.
2) Electronic Prescribing.
3) Patient Electronic Access to Health Information.
4) Coordination of Care through Patient Engagement.
5) Health Information Exchange.
6) Public Health and Clinical Data Reporting.
Only after meeting the requirements for the base score is one eligible to receive additional performance score credit based on the level of performance on a subset of three of the objectives required to achieve the base score. Those three are:
1) Patient Electronic Access to Health Information.
2) Coordination of Care through Patient Engagement.
3) Health Information Exchange
There are a total of eight measures for these three objectives.
The two measures specific to the Patient Electronic Access objective are:
a) Patient Access Measure.
b) Patient-Specific Education Measure.
Coordination of Care through Patient Engagement is assessed based upon the following three measures:
a) View, Download, Transmit Measure.
b) Secure Messaging Measure.
c) Patient-Generated Health Data Measures.
For the Health Information Exchange objective, assessment is also based on three measures. They are:
a) Patient Record Exchange Measure.
b) Request/Accept Patient Care Record Measure.
c) Clinical Information Reconciliation Measure.
Scoring for each of these eight measures is based upon reporting both a numerator and denominator, thus facilitating the calculation of percentage score to which a point value is assigned. For example, a performance rate of 85 on one of the listed measures would earn 8.5 points toward the performance score. The sum of the points accumulated for these eight measures is added to Threshold Score to determine the final score for ACI. Again, 25% of one’s MIPS Composite Score is derived from performance in the ACI component.
It warrants repeating and re-emphasis that the above information is subject to modification pending the release of the MIPS Final Rule, expected in early November.
As mentioned above, the Quality component of MIPS replaces the PQRS. Quality assessment will comprise 50% of the MIPS Composite Score. In next month’s column, I will provide an outline of the changes CMS is proposing for the Quality assessment. As a preview, some good news can be found in the fact that the current MIPS Quality component proposal will require providers to report on a total of six measures as opposed to the previous PQRS requirement to report on nine.
Until next month …
In this column, I will return to the topic of the Quality Payment Program (QPP), Centers for Medicare & Medicaid Services’ new payment program, currently scheduled to go into effect January 2017. The QPP has two tracks, the Merit-Based Incentive Payment System (MIPS) and advanced Alternative Payment Models (APMs). Again, it is widely expected that for the first several years of the program, the vast majority of surgeons will participate in the QPP via the MIPS track.
To briefly review from the July edition of this column, MIPS consists of four components:
1) Quality.
2) Resource Use.
3) Advancing Care Information (ACI).
4) Clinical Practice Improvement Activities (CPIA).
The Quality component replaces the Physician Quality Reporting System (PQRS), the Resource Use component replaces the Value-Based Modifier (VBM), the Advancing Care Information (ACI) component replaces the Electronic Health Record Meaningful Use (EHR-MU) program and is proposed to simplify the program by modifying requirements. The fourth component of MIPS, the Clinical Practice Improvement Activities (CPIA), is new.
MIPS participants will be assigned a Composite Performance Score based on their performance in all four components. As proposed for 2017, 50% of the MIPS Composite Performance Score will be based on performance in the Quality component, 25% will be based on the ACI component, 15% will be based on the CPIA, and 10% will be based on Resource Use. Thus, 75% of one’s score will be determined by performance in the Quality and ACI components. With that in mind, the remainder of this month’s column will be dedicated to the ACI with next month’s column focusing on the Quality component.
The Advancing Care Component (ACI)
As discussed above, the ACI component modifies and replaces the Electronic Health Record Meaningful Use (EHR-MU) program. As currently proposed, (and subject to change with release of the final rule in early November 2016), the score for ACI will be derived in two parts: a base score (50 points) and a performance score.

The threshold for achieving the base score remains an “all or nothing” proposition. Achieving the threshold is based upon reporting a yes/no statement OR the appropriate numerator and denominator for each measure, as required, for the following six objectives:
1) Protect Patient Health Information.
2) Electronic Prescribing.
3) Patient Electronic Access to Health Information.
4) Coordination of Care through Patient Engagement.
5) Health Information Exchange.
6) Public Health and Clinical Data Reporting.
Only after meeting the requirements for the base score is one eligible to receive additional performance score credit based on the level of performance on a subset of three of the objectives required to achieve the base score. Those three are:
1) Patient Electronic Access to Health Information.
2) Coordination of Care through Patient Engagement.
3) Health Information Exchange
There are a total of eight measures for these three objectives.
The two measures specific to the Patient Electronic Access objective are:
a) Patient Access Measure.
b) Patient-Specific Education Measure.
Coordination of Care through Patient Engagement is assessed based upon the following three measures:
a) View, Download, Transmit Measure.
b) Secure Messaging Measure.
c) Patient-Generated Health Data Measures.
For the Health Information Exchange objective, assessment is also based on three measures. They are:
a) Patient Record Exchange Measure.
b) Request/Accept Patient Care Record Measure.
c) Clinical Information Reconciliation Measure.
Scoring for each of these eight measures is based upon reporting both a numerator and denominator, thus facilitating the calculation of percentage score to which a point value is assigned. For example, a performance rate of 85 on one of the listed measures would earn 8.5 points toward the performance score. The sum of the points accumulated for these eight measures is added to Threshold Score to determine the final score for ACI. Again, 25% of one’s MIPS Composite Score is derived from performance in the ACI component.
It warrants repeating and re-emphasis that the above information is subject to modification pending the release of the MIPS Final Rule, expected in early November.
As mentioned above, the Quality component of MIPS replaces the PQRS. Quality assessment will comprise 50% of the MIPS Composite Score. In next month’s column, I will provide an outline of the changes CMS is proposing for the Quality assessment. As a preview, some good news can be found in the fact that the current MIPS Quality component proposal will require providers to report on a total of six measures as opposed to the previous PQRS requirement to report on nine.
Until next month …