User login
Official Newspaper of the American College of Surgeons
What’s in store for CMS under Seema Verma?
WASHINGTON – Big changes could be in store for the Medicaid program under the potential leadership of Seema Verma as administrator of the Centers for Medicare & Medicaid Services.
In nominating Ms. Verma, President Trump called her a leading expert on Medicaid and Medicare who will help “transform our health care system for the benefit of all Americans.”
A relative unknown, Ms. Verma is a health policy consultant who has spent 20 years quietly designing policy projects involving Medicaid, including the crafting of Indiana’s Medicaid expansion under the Affordable Care Act in which she worked closely with Vice President Pence.
How the savvy consultant could alter the Medicaid program during her potential CMS tenure has been the source of much speculation. President Trump and Rep. Tom Price (R-Ga.), Health & Human Services secretary–designee, have called for the restructuring of Medicaid, including the possibility of block grants that would set limits on total annual spending regardless of enrollment or caps that would limit average spending per enrollee.
Ms. Verma’s role in the revamp will largely depend on what Congress allows, said Mark Polston, a Washington, D.C.–based health law attorney and former associate general counsel for litigation in the HHS Office of General Counsel, CMS division, under President George W. Bush.
“We don’t know what cards she has in her hands – let alone what’s in the deck for her to deal from,” Mr. Polston said at a meeting sponsored by the American Bar Association. “That’s really going to be set by Congress.”
“From a CMS perspective, the Medicaid program is in large part run by the states,” Mr. Blum said at the ABA meeting. “What the new nominee says to me is that there is going to be a very high priority placed upon working with states, working with governors to modify Medicaid programs and to shape them to meet different state priorities.”
Look no further than the Healthy Indiana Plan 2.0 to get an idea of what such plans could look like, said Leslie V. Norwalk, a Washington, D.C.–based health law attorney and a former acting CMS administrator under President George W. Bush. The Indiana plan requires patients to pay a small amount to receive health coverage and includes a lockout period if payments are missed. Ms. Verma could work with governors to develop similar model waivers, Ms. Norwalk said.
Mr. Blum said he believes a top priority for Ms. Verma will be limiting the disruption that could come if the ACA is repealed and finding ways to cover those who lose coverage.
“What the next team really needs to really realize [is] that those who sign up for coverage – with the exchanges or through the new state expansions – are sicker on average, have lower income on average, and they’re going to have to think very carefully about the transitions going on,” Mr. Blum said at the meeting. “How you think about the transitions, how you think about continuity of care, how you think about disruptions – those will be very real and very tangible for the next team.”
“If she’s smart, she’ll find a deputy who’s very strong on Medicare. She’ll probably also try to find a former Hill staffer who can help her on the Hill,” Mr. Scully said. “The best news for her is that they picked her really early, so she’s got a head start.”
Hiring a deputy with a strong insurance background will also be key, Ms. Norwalk said in an interview. Even if the exchanges are repealed, other CMS programs, such as Medicare Part D and Medicare Advantage rely heavily on insurers, she said.
Ms. Norwalk noted that Ms. Verma may be surprised to find that much of the CMS agenda is controlled by the requirements of Medicare’s regulatory cycle. “[It] will take up a lot of her time, perhaps more than she might anticipate,” she said. “But in addition, you’ll have governors coming in wanting to do waivers. ... The third component will be how much time she spends on Capitol Hill working on whether repeal is done or not, and certainly the replace function and how that works will be a critical component of what she does. [If she’s confirmed,] she’ll be a very busy lady.”
[email protected]
On Twitter @legal_med
WASHINGTON – Big changes could be in store for the Medicaid program under the potential leadership of Seema Verma as administrator of the Centers for Medicare & Medicaid Services.
In nominating Ms. Verma, President Trump called her a leading expert on Medicaid and Medicare who will help “transform our health care system for the benefit of all Americans.”
A relative unknown, Ms. Verma is a health policy consultant who has spent 20 years quietly designing policy projects involving Medicaid, including the crafting of Indiana’s Medicaid expansion under the Affordable Care Act in which she worked closely with Vice President Pence.
How the savvy consultant could alter the Medicaid program during her potential CMS tenure has been the source of much speculation. President Trump and Rep. Tom Price (R-Ga.), Health & Human Services secretary–designee, have called for the restructuring of Medicaid, including the possibility of block grants that would set limits on total annual spending regardless of enrollment or caps that would limit average spending per enrollee.
Ms. Verma’s role in the revamp will largely depend on what Congress allows, said Mark Polston, a Washington, D.C.–based health law attorney and former associate general counsel for litigation in the HHS Office of General Counsel, CMS division, under President George W. Bush.
“We don’t know what cards she has in her hands – let alone what’s in the deck for her to deal from,” Mr. Polston said at a meeting sponsored by the American Bar Association. “That’s really going to be set by Congress.”
“From a CMS perspective, the Medicaid program is in large part run by the states,” Mr. Blum said at the ABA meeting. “What the new nominee says to me is that there is going to be a very high priority placed upon working with states, working with governors to modify Medicaid programs and to shape them to meet different state priorities.”
Look no further than the Healthy Indiana Plan 2.0 to get an idea of what such plans could look like, said Leslie V. Norwalk, a Washington, D.C.–based health law attorney and a former acting CMS administrator under President George W. Bush. The Indiana plan requires patients to pay a small amount to receive health coverage and includes a lockout period if payments are missed. Ms. Verma could work with governors to develop similar model waivers, Ms. Norwalk said.
Mr. Blum said he believes a top priority for Ms. Verma will be limiting the disruption that could come if the ACA is repealed and finding ways to cover those who lose coverage.
“What the next team really needs to really realize [is] that those who sign up for coverage – with the exchanges or through the new state expansions – are sicker on average, have lower income on average, and they’re going to have to think very carefully about the transitions going on,” Mr. Blum said at the meeting. “How you think about the transitions, how you think about continuity of care, how you think about disruptions – those will be very real and very tangible for the next team.”
“If she’s smart, she’ll find a deputy who’s very strong on Medicare. She’ll probably also try to find a former Hill staffer who can help her on the Hill,” Mr. Scully said. “The best news for her is that they picked her really early, so she’s got a head start.”
Hiring a deputy with a strong insurance background will also be key, Ms. Norwalk said in an interview. Even if the exchanges are repealed, other CMS programs, such as Medicare Part D and Medicare Advantage rely heavily on insurers, she said.
Ms. Norwalk noted that Ms. Verma may be surprised to find that much of the CMS agenda is controlled by the requirements of Medicare’s regulatory cycle. “[It] will take up a lot of her time, perhaps more than she might anticipate,” she said. “But in addition, you’ll have governors coming in wanting to do waivers. ... The third component will be how much time she spends on Capitol Hill working on whether repeal is done or not, and certainly the replace function and how that works will be a critical component of what she does. [If she’s confirmed,] she’ll be a very busy lady.”
[email protected]
On Twitter @legal_med
WASHINGTON – Big changes could be in store for the Medicaid program under the potential leadership of Seema Verma as administrator of the Centers for Medicare & Medicaid Services.
In nominating Ms. Verma, President Trump called her a leading expert on Medicaid and Medicare who will help “transform our health care system for the benefit of all Americans.”
A relative unknown, Ms. Verma is a health policy consultant who has spent 20 years quietly designing policy projects involving Medicaid, including the crafting of Indiana’s Medicaid expansion under the Affordable Care Act in which she worked closely with Vice President Pence.
How the savvy consultant could alter the Medicaid program during her potential CMS tenure has been the source of much speculation. President Trump and Rep. Tom Price (R-Ga.), Health & Human Services secretary–designee, have called for the restructuring of Medicaid, including the possibility of block grants that would set limits on total annual spending regardless of enrollment or caps that would limit average spending per enrollee.
Ms. Verma’s role in the revamp will largely depend on what Congress allows, said Mark Polston, a Washington, D.C.–based health law attorney and former associate general counsel for litigation in the HHS Office of General Counsel, CMS division, under President George W. Bush.
“We don’t know what cards she has in her hands – let alone what’s in the deck for her to deal from,” Mr. Polston said at a meeting sponsored by the American Bar Association. “That’s really going to be set by Congress.”
“From a CMS perspective, the Medicaid program is in large part run by the states,” Mr. Blum said at the ABA meeting. “What the new nominee says to me is that there is going to be a very high priority placed upon working with states, working with governors to modify Medicaid programs and to shape them to meet different state priorities.”
Look no further than the Healthy Indiana Plan 2.0 to get an idea of what such plans could look like, said Leslie V. Norwalk, a Washington, D.C.–based health law attorney and a former acting CMS administrator under President George W. Bush. The Indiana plan requires patients to pay a small amount to receive health coverage and includes a lockout period if payments are missed. Ms. Verma could work with governors to develop similar model waivers, Ms. Norwalk said.
Mr. Blum said he believes a top priority for Ms. Verma will be limiting the disruption that could come if the ACA is repealed and finding ways to cover those who lose coverage.
“What the next team really needs to really realize [is] that those who sign up for coverage – with the exchanges or through the new state expansions – are sicker on average, have lower income on average, and they’re going to have to think very carefully about the transitions going on,” Mr. Blum said at the meeting. “How you think about the transitions, how you think about continuity of care, how you think about disruptions – those will be very real and very tangible for the next team.”
“If she’s smart, she’ll find a deputy who’s very strong on Medicare. She’ll probably also try to find a former Hill staffer who can help her on the Hill,” Mr. Scully said. “The best news for her is that they picked her really early, so she’s got a head start.”
Hiring a deputy with a strong insurance background will also be key, Ms. Norwalk said in an interview. Even if the exchanges are repealed, other CMS programs, such as Medicare Part D and Medicare Advantage rely heavily on insurers, she said.
Ms. Norwalk noted that Ms. Verma may be surprised to find that much of the CMS agenda is controlled by the requirements of Medicare’s regulatory cycle. “[It] will take up a lot of her time, perhaps more than she might anticipate,” she said. “But in addition, you’ll have governors coming in wanting to do waivers. ... The third component will be how much time she spends on Capitol Hill working on whether repeal is done or not, and certainly the replace function and how that works will be a critical component of what she does. [If she’s confirmed,] she’ll be a very busy lady.”
[email protected]
On Twitter @legal_med
AT THE AMERICAN BAR ASSOCIATION HEALTH LAW SUMMIT
High-intensity statins cut amputations and mortality in PAD
NEW ORLEANS – High-intensity statin therapy in patients with peripheral artery disease was associated with significant reductions in amputations as well as mortality during up to 5 years of follow-up in the first large study to examine the relationship, Shipra Arya, MD, reported at the American Heart Association scientific sessions.
Low- or moderate-intensity statin therapy also improved survival compared to no statin, albeit to a significantly lesser magnitude than high-intensity therapy. But high- and low/intermediate-intensity statins were similarly effective in reducing amputation risk, according to Dr. Arya, a vascular surgeon at Emory University in Atlanta.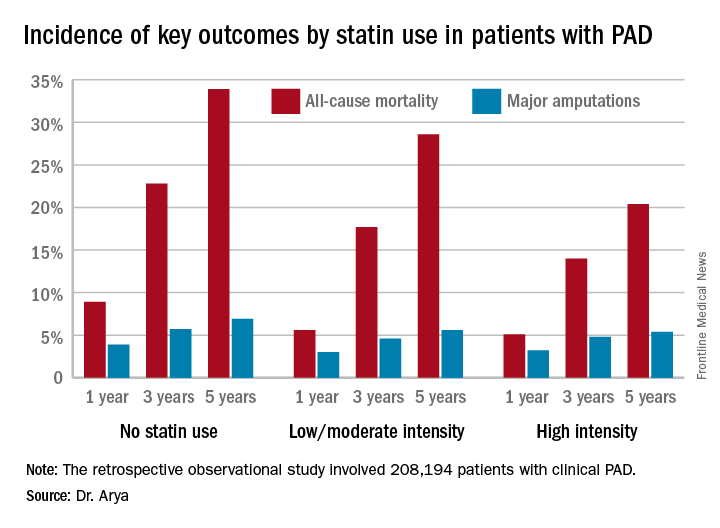
The 2013 AHA/American College of Cardiology treatment guidelines recommend high-intensity statins for all patients with clinical atherosclerotic disease, including those with PAD (Circulation. 2014 Jun 24;129[25 Suppl 2]:S1-45). (Updated PAD guidelines unveiled at the AHA meeting strongly recommend statin medication for all patients with PAD [Circulation. 2016 Nov 13. doi: 10.1161/CIR.0000000000000470]).However, the bulk of patients in Dr. Arya’s study were captured in the database prior to release of the 2013 guidelines. That may account for the sparse use of high-intensity statin therapy in the study cohort. Indeed, only 11.3% of the PAD patients were on a high-intensity statin. Another 36.2% were on moderate-intensity statin therapy, 3.5% were on low-intensity therapy, and 27.6% weren’t on a statin at all.
The relationship between statin therapy and mortality was strongly dose-dependent.
This study was funded by the AHA and the Atlanta Veterans Affairs Medical Center. Dr. Arya reported having no financial conflicts of interest.
NEW ORLEANS – High-intensity statin therapy in patients with peripheral artery disease was associated with significant reductions in amputations as well as mortality during up to 5 years of follow-up in the first large study to examine the relationship, Shipra Arya, MD, reported at the American Heart Association scientific sessions.
Low- or moderate-intensity statin therapy also improved survival compared to no statin, albeit to a significantly lesser magnitude than high-intensity therapy. But high- and low/intermediate-intensity statins were similarly effective in reducing amputation risk, according to Dr. Arya, a vascular surgeon at Emory University in Atlanta.
The 2013 AHA/American College of Cardiology treatment guidelines recommend high-intensity statins for all patients with clinical atherosclerotic disease, including those with PAD (Circulation. 2014 Jun 24;129[25 Suppl 2]:S1-45). (Updated PAD guidelines unveiled at the AHA meeting strongly recommend statin medication for all patients with PAD [Circulation. 2016 Nov 13. doi: 10.1161/CIR.0000000000000470]).However, the bulk of patients in Dr. Arya’s study were captured in the database prior to release of the 2013 guidelines. That may account for the sparse use of high-intensity statin therapy in the study cohort. Indeed, only 11.3% of the PAD patients were on a high-intensity statin. Another 36.2% were on moderate-intensity statin therapy, 3.5% were on low-intensity therapy, and 27.6% weren’t on a statin at all.
The relationship between statin therapy and mortality was strongly dose-dependent.
This study was funded by the AHA and the Atlanta Veterans Affairs Medical Center. Dr. Arya reported having no financial conflicts of interest.
NEW ORLEANS – High-intensity statin therapy in patients with peripheral artery disease was associated with significant reductions in amputations as well as mortality during up to 5 years of follow-up in the first large study to examine the relationship, Shipra Arya, MD, reported at the American Heart Association scientific sessions.
Low- or moderate-intensity statin therapy also improved survival compared to no statin, albeit to a significantly lesser magnitude than high-intensity therapy. But high- and low/intermediate-intensity statins were similarly effective in reducing amputation risk, according to Dr. Arya, a vascular surgeon at Emory University in Atlanta.
The 2013 AHA/American College of Cardiology treatment guidelines recommend high-intensity statins for all patients with clinical atherosclerotic disease, including those with PAD (Circulation. 2014 Jun 24;129[25 Suppl 2]:S1-45). (Updated PAD guidelines unveiled at the AHA meeting strongly recommend statin medication for all patients with PAD [Circulation. 2016 Nov 13. doi: 10.1161/CIR.0000000000000470]).However, the bulk of patients in Dr. Arya’s study were captured in the database prior to release of the 2013 guidelines. That may account for the sparse use of high-intensity statin therapy in the study cohort. Indeed, only 11.3% of the PAD patients were on a high-intensity statin. Another 36.2% were on moderate-intensity statin therapy, 3.5% were on low-intensity therapy, and 27.6% weren’t on a statin at all.
The relationship between statin therapy and mortality was strongly dose-dependent.
This study was funded by the AHA and the Atlanta Veterans Affairs Medical Center. Dr. Arya reported having no financial conflicts of interest.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point:
Major finding: The 5-year all-cause mortality rate after diagnosis of peripheral artery disease was 20.4% in patients on high-intensity statin therapy, 28.6% in those on a low- or moderate-intensity statin, and 33.9% in patients not on a statin.
Data source: A retrospective observational study of 208,194 patients with clinical peripheral artery disease in the national Veterans Affairs database for 2003-2014.
Disclosures: The AHA and the Atlanta Veterans Affairs Medical Center funded the study. The presenter reported having no financial conflicts of interest.
Longer follow-up needed to track mesh explantation trends
Explantation of mesh used in ventral hernia repair occurs in approximately 1 of every 1,000 surgeries, but it’s a “hidden” morbidity because it almost always happens well after the 30- to 90-day window in which postoperative complications are typically reported in most registries and surveillance systems, according to a report in the Journal of the American College of Surgeons.
Mesh explantation usually occurs 1-3 years after implantation and triples the operative costs of the original ventral hernia repair. The rate of 1 per 1,000 surgeries and the massive increase in cost are comparable with those of occult injury of the common bile duct during cholecystectomy that later requires biliary reconstruction. But mesh explantation doesn’t generate the “profound attention” accorded to bile duct injury, perhaps because it develops much later in the postoperative course.
“It is surprising that mesh complications have not yet prompted similar concern,” said Kristy Kummerow Broman, MD, of the department of surgery, Vanderbilt University Medical Center, Nashville, Tenn., and her associates.
Until now, the frequency and cost of mesh explantation after ventral hernia repair in the general population have not been known. To make a reasonable estimate, the investigators constructed a cohort of 619,751 patients using information from inpatient and surgery databases for New York, California, and Florida between 2005 and 2011. Most of these were open procedures (91%), while 9% were laparoscopic.
During a mean follow-up of 3 years, 438 patients (0.7 per 1,000) underwent mesh explantation. This is a clinically significant incidence, and is likely an underestimate because ICD-9 and CPT coding for mesh removal is highly variable, Dr. Broman and her associates said.
This rate, for just three states during 3 years of follow-up, is nearly twice as high as the rate of mesh-related complications voluntarily reported to the FDA in post-marketing surveillance for the entire country during a 7-year period, they noted (J Am Coll Surg. 2017 Jan;224:35-42).
“It is paramount” that surgeons, manufacturers, and regulatory groups advocate mandatory reporting and “extend the surveillance for at least 1-3 years after implantation of a mesh device,” Dr. Broman and her associates said.
In this study, the median time to explantation was approximately 1 year (range, 2 days to 6 years), and 80% of explantations occurred within 2 years.
The median cumulative operative cost – excluding physician fees, nonsurgical medical costs, and the costs of patient disability and lost productivity – were $21,889 for patients requiring mesh explantation, compared with only $6,579 for those who did not. This finding highlights “the profound long-term implications of implantable devices in abdominal wall reconstruction,” they noted.
To put their findings in context, the investigators reviewed the literature regarding major bile duct injury during cholecystectomy. One large study on cases from 2001 to 2011 found that the rate of biliary reconstruction was comparable with that of explantation, at 0.8 to 1.1 per 1,000. Similarly, reoperation for bile duct injury approximately tripled the operative costs ($9,061 for patients who required biliary reconstruction vs $2,689 for those who didn’t). However, the $21,000 for mesh reoperation far exceeds the $9,000 for biliary reoperation.
This study was supported by the Department of Veterans Affairs, the VA Tennessee Valley Healthcare System, and the Americas Hernia Society Quality Collaborative. Dr. Broman reported having no relevant financial disclosures; her associates reported ties to Intuitive Surgical Solutions, Bard Davol, Ariste Medical, and Pfizer.
Explantation of mesh used in ventral hernia repair occurs in approximately 1 of every 1,000 surgeries, but it’s a “hidden” morbidity because it almost always happens well after the 30- to 90-day window in which postoperative complications are typically reported in most registries and surveillance systems, according to a report in the Journal of the American College of Surgeons.
Mesh explantation usually occurs 1-3 years after implantation and triples the operative costs of the original ventral hernia repair. The rate of 1 per 1,000 surgeries and the massive increase in cost are comparable with those of occult injury of the common bile duct during cholecystectomy that later requires biliary reconstruction. But mesh explantation doesn’t generate the “profound attention” accorded to bile duct injury, perhaps because it develops much later in the postoperative course.
“It is surprising that mesh complications have not yet prompted similar concern,” said Kristy Kummerow Broman, MD, of the department of surgery, Vanderbilt University Medical Center, Nashville, Tenn., and her associates.
Until now, the frequency and cost of mesh explantation after ventral hernia repair in the general population have not been known. To make a reasonable estimate, the investigators constructed a cohort of 619,751 patients using information from inpatient and surgery databases for New York, California, and Florida between 2005 and 2011. Most of these were open procedures (91%), while 9% were laparoscopic.
During a mean follow-up of 3 years, 438 patients (0.7 per 1,000) underwent mesh explantation. This is a clinically significant incidence, and is likely an underestimate because ICD-9 and CPT coding for mesh removal is highly variable, Dr. Broman and her associates said.
This rate, for just three states during 3 years of follow-up, is nearly twice as high as the rate of mesh-related complications voluntarily reported to the FDA in post-marketing surveillance for the entire country during a 7-year period, they noted (J Am Coll Surg. 2017 Jan;224:35-42).
“It is paramount” that surgeons, manufacturers, and regulatory groups advocate mandatory reporting and “extend the surveillance for at least 1-3 years after implantation of a mesh device,” Dr. Broman and her associates said.
In this study, the median time to explantation was approximately 1 year (range, 2 days to 6 years), and 80% of explantations occurred within 2 years.
The median cumulative operative cost – excluding physician fees, nonsurgical medical costs, and the costs of patient disability and lost productivity – were $21,889 for patients requiring mesh explantation, compared with only $6,579 for those who did not. This finding highlights “the profound long-term implications of implantable devices in abdominal wall reconstruction,” they noted.
To put their findings in context, the investigators reviewed the literature regarding major bile duct injury during cholecystectomy. One large study on cases from 2001 to 2011 found that the rate of biliary reconstruction was comparable with that of explantation, at 0.8 to 1.1 per 1,000. Similarly, reoperation for bile duct injury approximately tripled the operative costs ($9,061 for patients who required biliary reconstruction vs $2,689 for those who didn’t). However, the $21,000 for mesh reoperation far exceeds the $9,000 for biliary reoperation.
This study was supported by the Department of Veterans Affairs, the VA Tennessee Valley Healthcare System, and the Americas Hernia Society Quality Collaborative. Dr. Broman reported having no relevant financial disclosures; her associates reported ties to Intuitive Surgical Solutions, Bard Davol, Ariste Medical, and Pfizer.
Explantation of mesh used in ventral hernia repair occurs in approximately 1 of every 1,000 surgeries, but it’s a “hidden” morbidity because it almost always happens well after the 30- to 90-day window in which postoperative complications are typically reported in most registries and surveillance systems, according to a report in the Journal of the American College of Surgeons.
Mesh explantation usually occurs 1-3 years after implantation and triples the operative costs of the original ventral hernia repair. The rate of 1 per 1,000 surgeries and the massive increase in cost are comparable with those of occult injury of the common bile duct during cholecystectomy that later requires biliary reconstruction. But mesh explantation doesn’t generate the “profound attention” accorded to bile duct injury, perhaps because it develops much later in the postoperative course.
“It is surprising that mesh complications have not yet prompted similar concern,” said Kristy Kummerow Broman, MD, of the department of surgery, Vanderbilt University Medical Center, Nashville, Tenn., and her associates.
Until now, the frequency and cost of mesh explantation after ventral hernia repair in the general population have not been known. To make a reasonable estimate, the investigators constructed a cohort of 619,751 patients using information from inpatient and surgery databases for New York, California, and Florida between 2005 and 2011. Most of these were open procedures (91%), while 9% were laparoscopic.
During a mean follow-up of 3 years, 438 patients (0.7 per 1,000) underwent mesh explantation. This is a clinically significant incidence, and is likely an underestimate because ICD-9 and CPT coding for mesh removal is highly variable, Dr. Broman and her associates said.
This rate, for just three states during 3 years of follow-up, is nearly twice as high as the rate of mesh-related complications voluntarily reported to the FDA in post-marketing surveillance for the entire country during a 7-year period, they noted (J Am Coll Surg. 2017 Jan;224:35-42).
“It is paramount” that surgeons, manufacturers, and regulatory groups advocate mandatory reporting and “extend the surveillance for at least 1-3 years after implantation of a mesh device,” Dr. Broman and her associates said.
In this study, the median time to explantation was approximately 1 year (range, 2 days to 6 years), and 80% of explantations occurred within 2 years.
The median cumulative operative cost – excluding physician fees, nonsurgical medical costs, and the costs of patient disability and lost productivity – were $21,889 for patients requiring mesh explantation, compared with only $6,579 for those who did not. This finding highlights “the profound long-term implications of implantable devices in abdominal wall reconstruction,” they noted.
To put their findings in context, the investigators reviewed the literature regarding major bile duct injury during cholecystectomy. One large study on cases from 2001 to 2011 found that the rate of biliary reconstruction was comparable with that of explantation, at 0.8 to 1.1 per 1,000. Similarly, reoperation for bile duct injury approximately tripled the operative costs ($9,061 for patients who required biliary reconstruction vs $2,689 for those who didn’t). However, the $21,000 for mesh reoperation far exceeds the $9,000 for biliary reoperation.
This study was supported by the Department of Veterans Affairs, the VA Tennessee Valley Healthcare System, and the Americas Hernia Society Quality Collaborative. Dr. Broman reported having no relevant financial disclosures; her associates reported ties to Intuitive Surgical Solutions, Bard Davol, Ariste Medical, and Pfizer.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF SURGEONS
Key clinical point: Explantation of mesh used in ventral hernia repair occurs in approximately 1 of every 1,000 surgeries, but it’s a “hidden” morbidity because it almost always happens well after the 30- to 90-day window in which postoperative complications are typically reported in most registries and surveillance systems.
Major finding: The 1/1,000 rate of mesh explantation, for three states during 3 years of follow-up, is nearly twice as high as the rate voluntarily reported to the Food and Drug Administration in post-marketing surveillance for the entire country during a 7-year period.
Data source: A longitudinal cohort study involving 619,751 adults undergoing ventral hernia repair in New York, California, and Florida who were followed for up to 6 years for mesh explantation.
Disclosures: This study was supported by the Department of Veterans Affairs, the VA Tennessee Valley Healthcare System, and the Americas Hernia Society Quality Collaborative. Dr. Broman reported having no relevant financial disclosures; her associates reported ties to Intuitive Surgical Solutions, Bard Davol, Ariste Medical, and Pfizer.
Lightweight mesh linked to longer LOS, worse QOL
Compared with midweight mesh, lightweight mesh was associated with more surgical site infection and longer hospital stay following open ventral hernia repair, according to a report published in the American Journal of Surgery.
In addition, lightweight mesh was associated with greater pain, more limitation of movement, and poorer quality of life for up to 2 years after the procedure, compared with midweight mesh.
Approximately 250,000 open ventral hernia repairs are performed in the Unites States each year, and mesh is used in 85% or more. Since heavyweight mesh was found to reduce abdominal wall mobility, which led to chronic discomfort in about 20% of cases, manufacturers turned to mesh that was more flexible, had reduced mass to decrease foreign-body reactions, but was strong enough to withstand the physiological stress that the abdominal wall is subjected to, the investigators noted.
They compared outcomes after hernia repairs using lightweight and midweight mesh by analyzing information in the International Hernia Mesh Registry database, which covers more than 30 medical centers in 10 countries. For this study, the researchers focused on 549 patients for whom surgeons had selected lightweight (34.2%) or midweight (47.7%) mesh. (The remaining 18.1% of cases used heavyweight mesh.)
Across the study groups, patients were similar for gender distribution; body mass index; race; and the presence of chronic obstructive pulmonary disease, asthma, and immunosuppression – factors that can heavily influence wound repair.
In an initial analysis of the data, midweight mesh was associated with significantly fewer superficial surgical site infections (1.2%) than lightweight mesh (4.8%), as well as a significantly shorter length of stay (3.6 days vs 5.3 days). However, rates of postoperative abdominal wall complications, abscesses, urinary tract infection, pneumonia, hematoma formation, seroma formation, ileus, deep vein thrombosis, and unplanned returns to the operating room were similar.
At 6-month follow-up, lightweight mesh was associated with significantly greater mesh sensation, abdominal discomfort, and movement limitation, as well as significantly worse overall quality of life (QOL), than midweight mesh. At 12 months, lightweight mesh was associated with significantly greater pain and limitation of movement and significantly worse QOL. At 24 months, lightweight mesh continued to be associated with movement limitation, but scores on other measures were similar to those with midweight mesh.
In a multivariate analysis that controlled for many potentially confounding variables, including smoking status, separation of the components of the mesh, the number of sutures anchoring the mesh, and the mesh location within the abdomen, midweight mesh was not associated with worse QOL scores at any time point. In contrast, lightweight mesh was associated with significantly worse QOL scores at 6 months, with an odds ratio of 2.64, and with significantly more pain at 12 months, with an OR of 2.58, Dr. Groene and his associates said (Am J Surg. 2016 Dec;212[6]:1054-62).
The investigators also noted that among their own hernia repair patients, lightweight mesh tends to fracture more easily than midweight mesh. Recent studies also have reported that over time, lightweight mesh is more likely to fail due to fracturing than midweight mesh, they added.
This study had no relevant financial relationships or sources of support. Dr. Groene and his associates reported having no financial conflicts of interest.
Compared with midweight mesh, lightweight mesh was associated with more surgical site infection and longer hospital stay following open ventral hernia repair, according to a report published in the American Journal of Surgery.
In addition, lightweight mesh was associated with greater pain, more limitation of movement, and poorer quality of life for up to 2 years after the procedure, compared with midweight mesh.
Approximately 250,000 open ventral hernia repairs are performed in the Unites States each year, and mesh is used in 85% or more. Since heavyweight mesh was found to reduce abdominal wall mobility, which led to chronic discomfort in about 20% of cases, manufacturers turned to mesh that was more flexible, had reduced mass to decrease foreign-body reactions, but was strong enough to withstand the physiological stress that the abdominal wall is subjected to, the investigators noted.
They compared outcomes after hernia repairs using lightweight and midweight mesh by analyzing information in the International Hernia Mesh Registry database, which covers more than 30 medical centers in 10 countries. For this study, the researchers focused on 549 patients for whom surgeons had selected lightweight (34.2%) or midweight (47.7%) mesh. (The remaining 18.1% of cases used heavyweight mesh.)
Across the study groups, patients were similar for gender distribution; body mass index; race; and the presence of chronic obstructive pulmonary disease, asthma, and immunosuppression – factors that can heavily influence wound repair.
In an initial analysis of the data, midweight mesh was associated with significantly fewer superficial surgical site infections (1.2%) than lightweight mesh (4.8%), as well as a significantly shorter length of stay (3.6 days vs 5.3 days). However, rates of postoperative abdominal wall complications, abscesses, urinary tract infection, pneumonia, hematoma formation, seroma formation, ileus, deep vein thrombosis, and unplanned returns to the operating room were similar.
At 6-month follow-up, lightweight mesh was associated with significantly greater mesh sensation, abdominal discomfort, and movement limitation, as well as significantly worse overall quality of life (QOL), than midweight mesh. At 12 months, lightweight mesh was associated with significantly greater pain and limitation of movement and significantly worse QOL. At 24 months, lightweight mesh continued to be associated with movement limitation, but scores on other measures were similar to those with midweight mesh.
In a multivariate analysis that controlled for many potentially confounding variables, including smoking status, separation of the components of the mesh, the number of sutures anchoring the mesh, and the mesh location within the abdomen, midweight mesh was not associated with worse QOL scores at any time point. In contrast, lightweight mesh was associated with significantly worse QOL scores at 6 months, with an odds ratio of 2.64, and with significantly more pain at 12 months, with an OR of 2.58, Dr. Groene and his associates said (Am J Surg. 2016 Dec;212[6]:1054-62).
The investigators also noted that among their own hernia repair patients, lightweight mesh tends to fracture more easily than midweight mesh. Recent studies also have reported that over time, lightweight mesh is more likely to fail due to fracturing than midweight mesh, they added.
This study had no relevant financial relationships or sources of support. Dr. Groene and his associates reported having no financial conflicts of interest.
Compared with midweight mesh, lightweight mesh was associated with more surgical site infection and longer hospital stay following open ventral hernia repair, according to a report published in the American Journal of Surgery.
In addition, lightweight mesh was associated with greater pain, more limitation of movement, and poorer quality of life for up to 2 years after the procedure, compared with midweight mesh.
Approximately 250,000 open ventral hernia repairs are performed in the Unites States each year, and mesh is used in 85% or more. Since heavyweight mesh was found to reduce abdominal wall mobility, which led to chronic discomfort in about 20% of cases, manufacturers turned to mesh that was more flexible, had reduced mass to decrease foreign-body reactions, but was strong enough to withstand the physiological stress that the abdominal wall is subjected to, the investigators noted.
They compared outcomes after hernia repairs using lightweight and midweight mesh by analyzing information in the International Hernia Mesh Registry database, which covers more than 30 medical centers in 10 countries. For this study, the researchers focused on 549 patients for whom surgeons had selected lightweight (34.2%) or midweight (47.7%) mesh. (The remaining 18.1% of cases used heavyweight mesh.)
Across the study groups, patients were similar for gender distribution; body mass index; race; and the presence of chronic obstructive pulmonary disease, asthma, and immunosuppression – factors that can heavily influence wound repair.
In an initial analysis of the data, midweight mesh was associated with significantly fewer superficial surgical site infections (1.2%) than lightweight mesh (4.8%), as well as a significantly shorter length of stay (3.6 days vs 5.3 days). However, rates of postoperative abdominal wall complications, abscesses, urinary tract infection, pneumonia, hematoma formation, seroma formation, ileus, deep vein thrombosis, and unplanned returns to the operating room were similar.
At 6-month follow-up, lightweight mesh was associated with significantly greater mesh sensation, abdominal discomfort, and movement limitation, as well as significantly worse overall quality of life (QOL), than midweight mesh. At 12 months, lightweight mesh was associated with significantly greater pain and limitation of movement and significantly worse QOL. At 24 months, lightweight mesh continued to be associated with movement limitation, but scores on other measures were similar to those with midweight mesh.
In a multivariate analysis that controlled for many potentially confounding variables, including smoking status, separation of the components of the mesh, the number of sutures anchoring the mesh, and the mesh location within the abdomen, midweight mesh was not associated with worse QOL scores at any time point. In contrast, lightweight mesh was associated with significantly worse QOL scores at 6 months, with an odds ratio of 2.64, and with significantly more pain at 12 months, with an OR of 2.58, Dr. Groene and his associates said (Am J Surg. 2016 Dec;212[6]:1054-62).
The investigators also noted that among their own hernia repair patients, lightweight mesh tends to fracture more easily than midweight mesh. Recent studies also have reported that over time, lightweight mesh is more likely to fail due to fracturing than midweight mesh, they added.
This study had no relevant financial relationships or sources of support. Dr. Groene and his associates reported having no financial conflicts of interest.
FROM THE AMERICAN JOURNAL OF SURGERY
Key clinical point: Compared with midweight mesh, lightweight mesh was associated with more surgical site infections and longer hospital stay in the short term and greater pain, more limitation of movement, and poorer quality of life for up to 2 years after open ventral hernia repair.
Major finding: In the short term, midweight mesh was associated with significantly fewer superficial surgical site infections (1.2%) than lightweight mesh (4.8%), as well as a significantly shorter length of stay (3.6 days vs 5.3 days).
Data source: An analysis of information in an international prospective registry of hernia mesh surgeries, which involved 549 adults.
Disclosures: This study had no relevant financial relationships or sources of support. Dr. Groene and his associates reported having no financial conflicts of interest.
Adjuvant chemo prolonged survival after radical nephroureterectomy
Adjuvant chemotherapy prolonged survival after radical nephroureterectomy by nearly a year, compared with observation alone, among patients with locally advanced or positive regional lymph node upper tract urothelial carcinoma, researchers reported.
After a median follow-up period of 49 months, median overall survival was 47 months with adjuvant chemotherapy and 36 months with observation alone (P less than .001), reported Thomas Seisen, MD, of Harvard Medical School, Boston, and his associates.
This analysis included 3,253 patients with pT3/T4 and/or pN+ upper tract urothelial carcinoma from the National Cancer Database. A total of 762 (23%) patients received adjuvant chemotherapy within 90 days after surgery, while 2,491 (77%) patients underwent observation only (J Clin Oncol. 2017 Jan 3. doi: 10.1200/JCO.2016.69.414).
Kaplan Meier analyses yielded 5-year adjusted overall survival rates of 44% and 36%, respectively. Adjuvant chemotherapy conferred a significant overall survival benefit in a Cox proportional hazards regression analysis (hazard ratio, 0.77; 95% confidence interval, 0.68 to 0.88), and the effect held up in tests designed to minimize selection bias – including propensity score adjustment (HR, 0.82; 0.73 to 0.93), stratification (HR, 0.84; 0.74 to 0.95), and matching (HR, 0.84; 0.75 to 0.95).
The effect persisted across subgroups stratified by age, gender, comorbidity burden, pathologic stage, and surgical margin status, and there was no significant variability in treatment effects, the researchers said. The findings are subject to “the usual biases related to the observational study design,” but pending level 1 evidence, they inform the management of patients with advanced upper tract urothelial carcinoma who undergo radical nephroureterectomy, the researchers concluded.
The work was supported by the Vattikuti Urology Institute, the Conquer Cancer Foundation of the American Society of Clinical Oncology, and the Prostate Cancer Foundation. Dr. Seisen had no relevant financial disclosures.
Adjuvant chemotherapy prolonged survival after radical nephroureterectomy by nearly a year, compared with observation alone, among patients with locally advanced or positive regional lymph node upper tract urothelial carcinoma, researchers reported.
After a median follow-up period of 49 months, median overall survival was 47 months with adjuvant chemotherapy and 36 months with observation alone (P less than .001), reported Thomas Seisen, MD, of Harvard Medical School, Boston, and his associates.
This analysis included 3,253 patients with pT3/T4 and/or pN+ upper tract urothelial carcinoma from the National Cancer Database. A total of 762 (23%) patients received adjuvant chemotherapy within 90 days after surgery, while 2,491 (77%) patients underwent observation only (J Clin Oncol. 2017 Jan 3. doi: 10.1200/JCO.2016.69.414).
Kaplan Meier analyses yielded 5-year adjusted overall survival rates of 44% and 36%, respectively. Adjuvant chemotherapy conferred a significant overall survival benefit in a Cox proportional hazards regression analysis (hazard ratio, 0.77; 95% confidence interval, 0.68 to 0.88), and the effect held up in tests designed to minimize selection bias – including propensity score adjustment (HR, 0.82; 0.73 to 0.93), stratification (HR, 0.84; 0.74 to 0.95), and matching (HR, 0.84; 0.75 to 0.95).
The effect persisted across subgroups stratified by age, gender, comorbidity burden, pathologic stage, and surgical margin status, and there was no significant variability in treatment effects, the researchers said. The findings are subject to “the usual biases related to the observational study design,” but pending level 1 evidence, they inform the management of patients with advanced upper tract urothelial carcinoma who undergo radical nephroureterectomy, the researchers concluded.
The work was supported by the Vattikuti Urology Institute, the Conquer Cancer Foundation of the American Society of Clinical Oncology, and the Prostate Cancer Foundation. Dr. Seisen had no relevant financial disclosures.
Adjuvant chemotherapy prolonged survival after radical nephroureterectomy by nearly a year, compared with observation alone, among patients with locally advanced or positive regional lymph node upper tract urothelial carcinoma, researchers reported.
After a median follow-up period of 49 months, median overall survival was 47 months with adjuvant chemotherapy and 36 months with observation alone (P less than .001), reported Thomas Seisen, MD, of Harvard Medical School, Boston, and his associates.
This analysis included 3,253 patients with pT3/T4 and/or pN+ upper tract urothelial carcinoma from the National Cancer Database. A total of 762 (23%) patients received adjuvant chemotherapy within 90 days after surgery, while 2,491 (77%) patients underwent observation only (J Clin Oncol. 2017 Jan 3. doi: 10.1200/JCO.2016.69.414).
Kaplan Meier analyses yielded 5-year adjusted overall survival rates of 44% and 36%, respectively. Adjuvant chemotherapy conferred a significant overall survival benefit in a Cox proportional hazards regression analysis (hazard ratio, 0.77; 95% confidence interval, 0.68 to 0.88), and the effect held up in tests designed to minimize selection bias – including propensity score adjustment (HR, 0.82; 0.73 to 0.93), stratification (HR, 0.84; 0.74 to 0.95), and matching (HR, 0.84; 0.75 to 0.95).
The effect persisted across subgroups stratified by age, gender, comorbidity burden, pathologic stage, and surgical margin status, and there was no significant variability in treatment effects, the researchers said. The findings are subject to “the usual biases related to the observational study design,” but pending level 1 evidence, they inform the management of patients with advanced upper tract urothelial carcinoma who undergo radical nephroureterectomy, the researchers concluded.
The work was supported by the Vattikuti Urology Institute, the Conquer Cancer Foundation of the American Society of Clinical Oncology, and the Prostate Cancer Foundation. Dr. Seisen had no relevant financial disclosures.
Key clinical point: Adjuvant chemotherapy prolonged survival after radical nephroureterectomy by nearly a year, compared with observation alone, among patients with locally advanced or positive regional lymph node upper tract urothelial carcinoma.
Major finding: After a median follow-up period of 49 months, median overall survival was 47 months with adjuvant chemotherapy and 36 months with observation alone (P less than .001).
Data source: An analysis of 3,253 patients with pT3/T4 and/or pN+ upper tract urothelial carcinoma from the National Cancer Database.
Disclosures: The work was supported by the Vattikuti Urology Institute, the Conquer Cancer Foundation of the American Society of Clinical Oncology, and the Prostate Cancer Foundation. Dr. Seisen had no relevant financial disclosures.
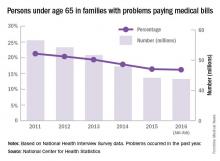
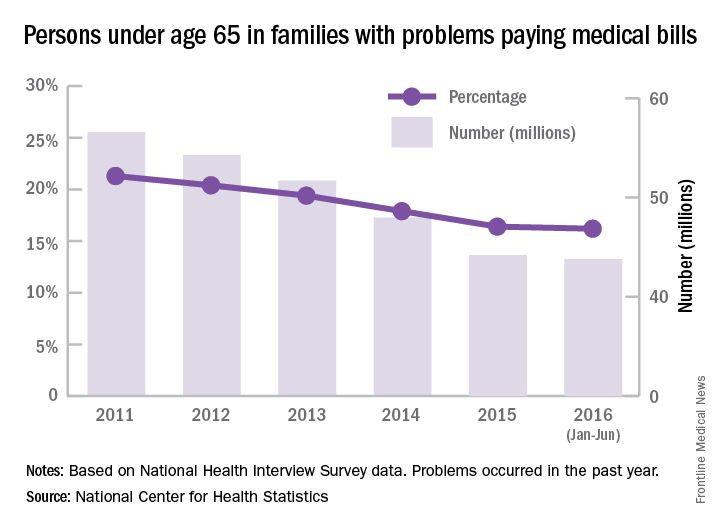
Fewer people having problems with medical bills
The number of people under age 65 years who were in families having trouble paying medical bills dropped by more than 22% from 2011 to 2016, according to the National Center for Health Statistics.
For the first 6 months of 2016, there were 43.8 million people, or 16.2% of the population under age 65 years, who were in families that had problems paying medical bills in the past year, which was down from 56.5 million (21.3 % of the population) in 2011, the NCHS reported.
The drop was consistent across race/ethnicity lines, but not the start and endpoints. The percentage of non-Hispanic blacks in families having trouble paying their medical bills dropped from 27.3% in 2011 to 23% in 2016, although there was actually a small increase from 2015 to 2016. Hispanics dropped from 24.3% in 2011 to 17.4% in 2016, non-Hispanic whites dropped from 19.8% to 15.1%, and non-Hispanic Asians went from 11% to 6%, according to data collected from 579,379 people for the National Health Interview Survey.
The number of people under age 65 years who were in families having trouble paying medical bills dropped by more than 22% from 2011 to 2016, according to the National Center for Health Statistics.
For the first 6 months of 2016, there were 43.8 million people, or 16.2% of the population under age 65 years, who were in families that had problems paying medical bills in the past year, which was down from 56.5 million (21.3 % of the population) in 2011, the NCHS reported.
The drop was consistent across race/ethnicity lines, but not the start and endpoints. The percentage of non-Hispanic blacks in families having trouble paying their medical bills dropped from 27.3% in 2011 to 23% in 2016, although there was actually a small increase from 2015 to 2016. Hispanics dropped from 24.3% in 2011 to 17.4% in 2016, non-Hispanic whites dropped from 19.8% to 15.1%, and non-Hispanic Asians went from 11% to 6%, according to data collected from 579,379 people for the National Health Interview Survey.
The number of people under age 65 years who were in families having trouble paying medical bills dropped by more than 22% from 2011 to 2016, according to the National Center for Health Statistics.
For the first 6 months of 2016, there were 43.8 million people, or 16.2% of the population under age 65 years, who were in families that had problems paying medical bills in the past year, which was down from 56.5 million (21.3 % of the population) in 2011, the NCHS reported.
The drop was consistent across race/ethnicity lines, but not the start and endpoints. The percentage of non-Hispanic blacks in families having trouble paying their medical bills dropped from 27.3% in 2011 to 23% in 2016, although there was actually a small increase from 2015 to 2016. Hispanics dropped from 24.3% in 2011 to 17.4% in 2016, non-Hispanic whites dropped from 19.8% to 15.1%, and non-Hispanic Asians went from 11% to 6%, according to data collected from 579,379 people for the National Health Interview Survey.
Diagnostic laparoscopy identifies ovarian cancers amenable to PCS
For women with suspected advanced epithelial ovarian cancer, diagnostic laparoscopy can help to distinguish between patients who could benefit from primary cytoreductive surgery (PCS) and those who might have better outcomes with neoadjuvant chemotherapy and interval cytoreductive surgery, according to investigators in the Netherlands.
In a randomized controlled trial exploring whether initial diagnostic laparoscopy could spare some patients from undergoing futile PCS, the investigators found that only 10% of patients assigned to diagnostic laparoscopy prior to PCS underwent a subsequent futile laparotomy, defined as residual disease greater than 1 cm following surgery. In contrast, 39% of women assigned to primary PCS had disease that might have been better treated by chemotherapy and interval surgery,
“In women with a plan for PCS, these data suggest that performance of diagnostic laparoscopy first is reasonable and that if cytoreduction to [less than] 1 cm of residual disease seems feasible, to proceed with PCS,” wrote Marrije R. Buist, MD of Academic Medical Center, Amsterdam, and colleagues.
Among women with International Federation of Gynecology and Obstetrics (FIGO) stage IIIC to IV epithelial ovarian cancer, survival depends largely on the ability of surgery to either completely remove disease, or to leave at best less than 1 cm of residual disease. However, aggressive surgery in patients with more extensive disease is associated with significant morbidities, the authors noted.
“If at PCS, extensive disease is present, surgery could be ceased, and neoadjuvant chemotherapy with interval surgery could be a good alternative treatment. Therefore, the identification of patients with extensive disease who are likely to have [more than] 1 cm of residual tumor after PCS, defined as a futile laparotomy, is important,” they wrote.
To test this idea, the investigators, from eight cancer centers in the Netherlands, enrolled 201 patients with suspected FIGO stage IIB ovarian cancer or higher, and randomly assigned them to undergo either initial diagnostic laparoscopy or PCS.
They found that 10 of the 102 patients (10%) assigned to diagnostic laparoscopy went on to undergo PCS that revealed residual disease greater than 1 cm, compared with 39 of the 99 patients (39%) assigned to PCS. This difference translated into a relative risk for futile laparotomy of 0.25 for diagnostic laparoscopy compared with PCS (P less than .001).
Only 3 (3%) patients in the diagnostic laparoscopy group went on to have both PCS and interval surgery, compared with 28 (28%) patients initially assigned to PCS (P less than .001).
The Dutch Organization for Health Research and Development supported the study. All but one coauthor reported having no potential conflicts of interest.
For women with suspected advanced epithelial ovarian cancer, diagnostic laparoscopy can help to distinguish between patients who could benefit from primary cytoreductive surgery (PCS) and those who might have better outcomes with neoadjuvant chemotherapy and interval cytoreductive surgery, according to investigators in the Netherlands.
In a randomized controlled trial exploring whether initial diagnostic laparoscopy could spare some patients from undergoing futile PCS, the investigators found that only 10% of patients assigned to diagnostic laparoscopy prior to PCS underwent a subsequent futile laparotomy, defined as residual disease greater than 1 cm following surgery. In contrast, 39% of women assigned to primary PCS had disease that might have been better treated by chemotherapy and interval surgery,
“In women with a plan for PCS, these data suggest that performance of diagnostic laparoscopy first is reasonable and that if cytoreduction to [less than] 1 cm of residual disease seems feasible, to proceed with PCS,” wrote Marrije R. Buist, MD of Academic Medical Center, Amsterdam, and colleagues.
Among women with International Federation of Gynecology and Obstetrics (FIGO) stage IIIC to IV epithelial ovarian cancer, survival depends largely on the ability of surgery to either completely remove disease, or to leave at best less than 1 cm of residual disease. However, aggressive surgery in patients with more extensive disease is associated with significant morbidities, the authors noted.
“If at PCS, extensive disease is present, surgery could be ceased, and neoadjuvant chemotherapy with interval surgery could be a good alternative treatment. Therefore, the identification of patients with extensive disease who are likely to have [more than] 1 cm of residual tumor after PCS, defined as a futile laparotomy, is important,” they wrote.
To test this idea, the investigators, from eight cancer centers in the Netherlands, enrolled 201 patients with suspected FIGO stage IIB ovarian cancer or higher, and randomly assigned them to undergo either initial diagnostic laparoscopy or PCS.
They found that 10 of the 102 patients (10%) assigned to diagnostic laparoscopy went on to undergo PCS that revealed residual disease greater than 1 cm, compared with 39 of the 99 patients (39%) assigned to PCS. This difference translated into a relative risk for futile laparotomy of 0.25 for diagnostic laparoscopy compared with PCS (P less than .001).
Only 3 (3%) patients in the diagnostic laparoscopy group went on to have both PCS and interval surgery, compared with 28 (28%) patients initially assigned to PCS (P less than .001).
The Dutch Organization for Health Research and Development supported the study. All but one coauthor reported having no potential conflicts of interest.
For women with suspected advanced epithelial ovarian cancer, diagnostic laparoscopy can help to distinguish between patients who could benefit from primary cytoreductive surgery (PCS) and those who might have better outcomes with neoadjuvant chemotherapy and interval cytoreductive surgery, according to investigators in the Netherlands.
In a randomized controlled trial exploring whether initial diagnostic laparoscopy could spare some patients from undergoing futile PCS, the investigators found that only 10% of patients assigned to diagnostic laparoscopy prior to PCS underwent a subsequent futile laparotomy, defined as residual disease greater than 1 cm following surgery. In contrast, 39% of women assigned to primary PCS had disease that might have been better treated by chemotherapy and interval surgery,
“In women with a plan for PCS, these data suggest that performance of diagnostic laparoscopy first is reasonable and that if cytoreduction to [less than] 1 cm of residual disease seems feasible, to proceed with PCS,” wrote Marrije R. Buist, MD of Academic Medical Center, Amsterdam, and colleagues.
Among women with International Federation of Gynecology and Obstetrics (FIGO) stage IIIC to IV epithelial ovarian cancer, survival depends largely on the ability of surgery to either completely remove disease, or to leave at best less than 1 cm of residual disease. However, aggressive surgery in patients with more extensive disease is associated with significant morbidities, the authors noted.
“If at PCS, extensive disease is present, surgery could be ceased, and neoadjuvant chemotherapy with interval surgery could be a good alternative treatment. Therefore, the identification of patients with extensive disease who are likely to have [more than] 1 cm of residual tumor after PCS, defined as a futile laparotomy, is important,” they wrote.
To test this idea, the investigators, from eight cancer centers in the Netherlands, enrolled 201 patients with suspected FIGO stage IIB ovarian cancer or higher, and randomly assigned them to undergo either initial diagnostic laparoscopy or PCS.
They found that 10 of the 102 patients (10%) assigned to diagnostic laparoscopy went on to undergo PCS that revealed residual disease greater than 1 cm, compared with 39 of the 99 patients (39%) assigned to PCS. This difference translated into a relative risk for futile laparotomy of 0.25 for diagnostic laparoscopy compared with PCS (P less than .001).
Only 3 (3%) patients in the diagnostic laparoscopy group went on to have both PCS and interval surgery, compared with 28 (28%) patients initially assigned to PCS (P less than .001).
The Dutch Organization for Health Research and Development supported the study. All but one coauthor reported having no potential conflicts of interest.
Key clinical point: Diagnostic laparoscopy can help to identify patients with advanced ovarian cancer who can best benefit from primary surgery or chemotherapy.
Major finding: Ten percent of women assigned to diagnostic laparoscopy underwent futile laparotomy, vs. 39% assigned to primary cytoreductive surgery.
Data source: Randomized controlled trial of 201 women with suspected FIGO stage IIB or greater disease.
Disclosures The Dutch Organization for Health Research and Development supported the study. All but one coauthor reported having no potential conflicts of interest.
Law & Medicine: How case law shapes EMTALA
(This is the second installment of a three-part series.)
Question: Emergency Medical Treatment and Labor Act (EMTALA) litigation has yielded which of the following rules of law?
A. The statute is applicable only when a patient is physically in the hospital’s emergency department (ED).
B. Directing an ambulance away from the ED is a violation of EMTALA.
C. All patients presenting to the ED must have an appropriate medical screening exam conforming to customary standard of care.
D. It is not what is performed in any medical screening exam, but whether it is applied evenly to all patients similarly situated.
E. The U.S. Supreme Court has held that an improper motive behind an unstable transfer is a prerequisite to an EMTALA violation.
Answer: D. In 1986, Congress enacted the Emergency Medical Treatment and Labor Act to ensure that all patients who present themselves to the emergency department are appropriately screened for an emergent medical condition, and if one is present, that they be stabilized prior to transfer or discharge.
In the 3 decades since its enactment, the statute has, as expected, spawned numerous lawsuits. Parts two and three of this series on EMTALA summarize the salient findings and rules of law in several interesting and impactful cases. These cases are neither encyclopedic nor necessarily representative of the types of litigation commonly encountered.
EMTALA is about events in the emergency department. They begin with the patient coming to the ED seeking treatment, and the statute specifically refers to “any individual ... [who] comes to the emergency department and a request is made on the individual’s behalf for examination or treatment for a medical condition.”
But what if the patient has yet to arrive, e.g., in an ambulance en route, and was diverted elsewhere in an unstable condition? In Hawaii’s case of Arrington v. Wong (237 F.3d 1066 [9th Cir. 2001]), the court was faced with whether the requirement that a patient must first “come to” the hospital means his or her literal physical presence in the hospital.
On May 5, 1996, Harold Arrington developed dyspnea while driving to his job as a security guard. En route to the closest medical facility, the Queen’s Medical Center, the ambulance personnel contacted Dr. Norbert Wong, the physician on call, describing the patient as being in severe respiratory distress, speaking one to two words at a time, and breathing about 50 times a minute.
Although Queen’s was not on diversionary status at the time, Dr. Wong thought it was okay for the ambulance to go instead to Tripler Hospital, a more distant hospital, as the patient’s doctor worked there. Unfortunately, by the time the ambulance arrived at Tripler, Mr. Arrington’s condition had deteriorated, and he was pronounced dead shortly after arrival.
The lower court ruled for the defendant, holding that the statute demanded an actual physical presence in the ED, but the 9th Circuit Court of Appeals reversed. It held that under EMTALA, a hospital may divert an ambulance that has contacted its emergency department and is on the way to that hospital only if the hospital is in diversionary status, because the diverting hospital then has a valid, treatment-related reason for doing so.
Such an interpretation of the law works no hardship on the hospital and is consistent with the Centers for Medicare & Medicaid Services’ regulation that “only requires hospitals that offer emergency services to provide screening and stabilizing treatment within the scope of their capabilities.” The 9th Circuit felt that this was consistent with the purpose and language of the EMTALA statute.
The next case, Summers v. Baptist Medical Center (91 F. 3d 1132 [8th Cir. 1996]), addressed the screening aspect of EMTALA, specifically on the distinction between disparity and adequacy in screening procedures. In Summers, the plaintiff fell from a tree while hunting and sustained bilateral hemothoraces, vertebral, rib, and sternal fractures. Incredibly, the diagnosis in the first hospital ED was muscle spasms, and the diagnoses only became clear when he checked into a second hospital 2 days later.
Still, the court ruled that there was no EMTALA violation, and that allegations of substandard care should be addressed under a negligence theory in state courts and not under EMTALA. The court reasoned that under the statute, an “inappropriate” screening is one that is performed in a disparate manner to similarly situated patients, and the hospital itself is usually left to define for itself what is within its capabilities. It is up to the hospital itself to determine what its screening procedures will be and to apply them alike to all patients with comparable complaints.
Likewise, in Vickers v. Nash (78 F.3d 139 [4th Cir. 1996]), an intoxicated patient who sustained a head injury following a fight died 4 days later from an epidural hematoma that was missed. He did have his head laceration treated in the ED, and was observed for 11 hours before discharge. The court ruled that the plaintiffs had failed to prove there was disparate treatment, and that hospitals can only be expected to stabilize emergency medical conditions known to them at the time.
Finally, in its first and thus far only EMTALA case, the U.S. Supreme Court in 1999 looked at whether an improper motive was a prerequisite for a finding of an EMTALA violation regarding stabilization and transfer.
In Roberts v. Galen of Virginia (119 S. Court 685 [1999]), the patient, injured in a truck accident, required a splenectomy and ventilator support. After a prolonged hospital stay, she was about to be moved to a nearby nursing home when she developed a high fever from an infection, and had to be transferred to an acute care facility. Her guardian, Roberts, brought suit, asserting violations of EMTALA’s stabilization and transfer requirements. The hospital argued that no material deterioration of the condition was likely to result from or occur during the transfer, and the district court determined her transfer was not prompted by an improper motive.
On appeal, the 6th Circuit Court of Appeals affirmed, extending its earlier holding that a showing of improper motive was required to make out an inadequate screening claim under EMTALA.
The hospital raised a number of important defenses, which included the physician lacking actual knowledge that the patient had an emergency medical condition, that EMTALA did not apply to in-hospital treatment and discharge decisions, and denying that EMTALA imposes minimum substantive standards of medical care.
The U.S. Supreme Court granted certiorari on the single issue whether the improper motive test should apply to an allegedly wrongful transfer. Overturning the appeals court, it held that Section 1395dd(b) (stabilization and transfer) contained no express or implied “improper motive” requirement. The Supreme Court declined to resolve broader issues under the statute.
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, and currently directs the St. Francis International Center for Healthcare Ethics in Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. Some of the articles in this series are adapted from the author’s 2006 book, “Medical Malpractice: Understanding the Law, Managing the Risk,” and his 2012 Halsbury treatise, “Medical Negligence and Professional Misconduct.” For additional information, readers may contact the author at [email protected].
(This is the second installment of a three-part series.)
Question: Emergency Medical Treatment and Labor Act (EMTALA) litigation has yielded which of the following rules of law?
A. The statute is applicable only when a patient is physically in the hospital’s emergency department (ED).
B. Directing an ambulance away from the ED is a violation of EMTALA.
C. All patients presenting to the ED must have an appropriate medical screening exam conforming to customary standard of care.
D. It is not what is performed in any medical screening exam, but whether it is applied evenly to all patients similarly situated.
E. The U.S. Supreme Court has held that an improper motive behind an unstable transfer is a prerequisite to an EMTALA violation.
Answer: D. In 1986, Congress enacted the Emergency Medical Treatment and Labor Act to ensure that all patients who present themselves to the emergency department are appropriately screened for an emergent medical condition, and if one is present, that they be stabilized prior to transfer or discharge.
In the 3 decades since its enactment, the statute has, as expected, spawned numerous lawsuits. Parts two and three of this series on EMTALA summarize the salient findings and rules of law in several interesting and impactful cases. These cases are neither encyclopedic nor necessarily representative of the types of litigation commonly encountered.
EMTALA is about events in the emergency department. They begin with the patient coming to the ED seeking treatment, and the statute specifically refers to “any individual ... [who] comes to the emergency department and a request is made on the individual’s behalf for examination or treatment for a medical condition.”
But what if the patient has yet to arrive, e.g., in an ambulance en route, and was diverted elsewhere in an unstable condition? In Hawaii’s case of Arrington v. Wong (237 F.3d 1066 [9th Cir. 2001]), the court was faced with whether the requirement that a patient must first “come to” the hospital means his or her literal physical presence in the hospital.
On May 5, 1996, Harold Arrington developed dyspnea while driving to his job as a security guard. En route to the closest medical facility, the Queen’s Medical Center, the ambulance personnel contacted Dr. Norbert Wong, the physician on call, describing the patient as being in severe respiratory distress, speaking one to two words at a time, and breathing about 50 times a minute.
Although Queen’s was not on diversionary status at the time, Dr. Wong thought it was okay for the ambulance to go instead to Tripler Hospital, a more distant hospital, as the patient’s doctor worked there. Unfortunately, by the time the ambulance arrived at Tripler, Mr. Arrington’s condition had deteriorated, and he was pronounced dead shortly after arrival.
The lower court ruled for the defendant, holding that the statute demanded an actual physical presence in the ED, but the 9th Circuit Court of Appeals reversed. It held that under EMTALA, a hospital may divert an ambulance that has contacted its emergency department and is on the way to that hospital only if the hospital is in diversionary status, because the diverting hospital then has a valid, treatment-related reason for doing so.
Such an interpretation of the law works no hardship on the hospital and is consistent with the Centers for Medicare & Medicaid Services’ regulation that “only requires hospitals that offer emergency services to provide screening and stabilizing treatment within the scope of their capabilities.” The 9th Circuit felt that this was consistent with the purpose and language of the EMTALA statute.
The next case, Summers v. Baptist Medical Center (91 F. 3d 1132 [8th Cir. 1996]), addressed the screening aspect of EMTALA, specifically on the distinction between disparity and adequacy in screening procedures. In Summers, the plaintiff fell from a tree while hunting and sustained bilateral hemothoraces, vertebral, rib, and sternal fractures. Incredibly, the diagnosis in the first hospital ED was muscle spasms, and the diagnoses only became clear when he checked into a second hospital 2 days later.
Still, the court ruled that there was no EMTALA violation, and that allegations of substandard care should be addressed under a negligence theory in state courts and not under EMTALA. The court reasoned that under the statute, an “inappropriate” screening is one that is performed in a disparate manner to similarly situated patients, and the hospital itself is usually left to define for itself what is within its capabilities. It is up to the hospital itself to determine what its screening procedures will be and to apply them alike to all patients with comparable complaints.
Likewise, in Vickers v. Nash (78 F.3d 139 [4th Cir. 1996]), an intoxicated patient who sustained a head injury following a fight died 4 days later from an epidural hematoma that was missed. He did have his head laceration treated in the ED, and was observed for 11 hours before discharge. The court ruled that the plaintiffs had failed to prove there was disparate treatment, and that hospitals can only be expected to stabilize emergency medical conditions known to them at the time.
Finally, in its first and thus far only EMTALA case, the U.S. Supreme Court in 1999 looked at whether an improper motive was a prerequisite for a finding of an EMTALA violation regarding stabilization and transfer.
In Roberts v. Galen of Virginia (119 S. Court 685 [1999]), the patient, injured in a truck accident, required a splenectomy and ventilator support. After a prolonged hospital stay, she was about to be moved to a nearby nursing home when she developed a high fever from an infection, and had to be transferred to an acute care facility. Her guardian, Roberts, brought suit, asserting violations of EMTALA’s stabilization and transfer requirements. The hospital argued that no material deterioration of the condition was likely to result from or occur during the transfer, and the district court determined her transfer was not prompted by an improper motive.
On appeal, the 6th Circuit Court of Appeals affirmed, extending its earlier holding that a showing of improper motive was required to make out an inadequate screening claim under EMTALA.
The hospital raised a number of important defenses, which included the physician lacking actual knowledge that the patient had an emergency medical condition, that EMTALA did not apply to in-hospital treatment and discharge decisions, and denying that EMTALA imposes minimum substantive standards of medical care.
The U.S. Supreme Court granted certiorari on the single issue whether the improper motive test should apply to an allegedly wrongful transfer. Overturning the appeals court, it held that Section 1395dd(b) (stabilization and transfer) contained no express or implied “improper motive” requirement. The Supreme Court declined to resolve broader issues under the statute.
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, and currently directs the St. Francis International Center for Healthcare Ethics in Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. Some of the articles in this series are adapted from the author’s 2006 book, “Medical Malpractice: Understanding the Law, Managing the Risk,” and his 2012 Halsbury treatise, “Medical Negligence and Professional Misconduct.” For additional information, readers may contact the author at [email protected].
(This is the second installment of a three-part series.)
Question: Emergency Medical Treatment and Labor Act (EMTALA) litigation has yielded which of the following rules of law?
A. The statute is applicable only when a patient is physically in the hospital’s emergency department (ED).
B. Directing an ambulance away from the ED is a violation of EMTALA.
C. All patients presenting to the ED must have an appropriate medical screening exam conforming to customary standard of care.
D. It is not what is performed in any medical screening exam, but whether it is applied evenly to all patients similarly situated.
E. The U.S. Supreme Court has held that an improper motive behind an unstable transfer is a prerequisite to an EMTALA violation.
Answer: D. In 1986, Congress enacted the Emergency Medical Treatment and Labor Act to ensure that all patients who present themselves to the emergency department are appropriately screened for an emergent medical condition, and if one is present, that they be stabilized prior to transfer or discharge.
In the 3 decades since its enactment, the statute has, as expected, spawned numerous lawsuits. Parts two and three of this series on EMTALA summarize the salient findings and rules of law in several interesting and impactful cases. These cases are neither encyclopedic nor necessarily representative of the types of litigation commonly encountered.
EMTALA is about events in the emergency department. They begin with the patient coming to the ED seeking treatment, and the statute specifically refers to “any individual ... [who] comes to the emergency department and a request is made on the individual’s behalf for examination or treatment for a medical condition.”
But what if the patient has yet to arrive, e.g., in an ambulance en route, and was diverted elsewhere in an unstable condition? In Hawaii’s case of Arrington v. Wong (237 F.3d 1066 [9th Cir. 2001]), the court was faced with whether the requirement that a patient must first “come to” the hospital means his or her literal physical presence in the hospital.
On May 5, 1996, Harold Arrington developed dyspnea while driving to his job as a security guard. En route to the closest medical facility, the Queen’s Medical Center, the ambulance personnel contacted Dr. Norbert Wong, the physician on call, describing the patient as being in severe respiratory distress, speaking one to two words at a time, and breathing about 50 times a minute.
Although Queen’s was not on diversionary status at the time, Dr. Wong thought it was okay for the ambulance to go instead to Tripler Hospital, a more distant hospital, as the patient’s doctor worked there. Unfortunately, by the time the ambulance arrived at Tripler, Mr. Arrington’s condition had deteriorated, and he was pronounced dead shortly after arrival.
The lower court ruled for the defendant, holding that the statute demanded an actual physical presence in the ED, but the 9th Circuit Court of Appeals reversed. It held that under EMTALA, a hospital may divert an ambulance that has contacted its emergency department and is on the way to that hospital only if the hospital is in diversionary status, because the diverting hospital then has a valid, treatment-related reason for doing so.
Such an interpretation of the law works no hardship on the hospital and is consistent with the Centers for Medicare & Medicaid Services’ regulation that “only requires hospitals that offer emergency services to provide screening and stabilizing treatment within the scope of their capabilities.” The 9th Circuit felt that this was consistent with the purpose and language of the EMTALA statute.
The next case, Summers v. Baptist Medical Center (91 F. 3d 1132 [8th Cir. 1996]), addressed the screening aspect of EMTALA, specifically on the distinction between disparity and adequacy in screening procedures. In Summers, the plaintiff fell from a tree while hunting and sustained bilateral hemothoraces, vertebral, rib, and sternal fractures. Incredibly, the diagnosis in the first hospital ED was muscle spasms, and the diagnoses only became clear when he checked into a second hospital 2 days later.
Still, the court ruled that there was no EMTALA violation, and that allegations of substandard care should be addressed under a negligence theory in state courts and not under EMTALA. The court reasoned that under the statute, an “inappropriate” screening is one that is performed in a disparate manner to similarly situated patients, and the hospital itself is usually left to define for itself what is within its capabilities. It is up to the hospital itself to determine what its screening procedures will be and to apply them alike to all patients with comparable complaints.
Likewise, in Vickers v. Nash (78 F.3d 139 [4th Cir. 1996]), an intoxicated patient who sustained a head injury following a fight died 4 days later from an epidural hematoma that was missed. He did have his head laceration treated in the ED, and was observed for 11 hours before discharge. The court ruled that the plaintiffs had failed to prove there was disparate treatment, and that hospitals can only be expected to stabilize emergency medical conditions known to them at the time.
Finally, in its first and thus far only EMTALA case, the U.S. Supreme Court in 1999 looked at whether an improper motive was a prerequisite for a finding of an EMTALA violation regarding stabilization and transfer.
In Roberts v. Galen of Virginia (119 S. Court 685 [1999]), the patient, injured in a truck accident, required a splenectomy and ventilator support. After a prolonged hospital stay, she was about to be moved to a nearby nursing home when she developed a high fever from an infection, and had to be transferred to an acute care facility. Her guardian, Roberts, brought suit, asserting violations of EMTALA’s stabilization and transfer requirements. The hospital argued that no material deterioration of the condition was likely to result from or occur during the transfer, and the district court determined her transfer was not prompted by an improper motive.
On appeal, the 6th Circuit Court of Appeals affirmed, extending its earlier holding that a showing of improper motive was required to make out an inadequate screening claim under EMTALA.
The hospital raised a number of important defenses, which included the physician lacking actual knowledge that the patient had an emergency medical condition, that EMTALA did not apply to in-hospital treatment and discharge decisions, and denying that EMTALA imposes minimum substantive standards of medical care.
The U.S. Supreme Court granted certiorari on the single issue whether the improper motive test should apply to an allegedly wrongful transfer. Overturning the appeals court, it held that Section 1395dd(b) (stabilization and transfer) contained no express or implied “improper motive” requirement. The Supreme Court declined to resolve broader issues under the statute.
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, and currently directs the St. Francis International Center for Healthcare Ethics in Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. Some of the articles in this series are adapted from the author’s 2006 book, “Medical Malpractice: Understanding the Law, Managing the Risk,” and his 2012 Halsbury treatise, “Medical Negligence and Professional Misconduct.” For additional information, readers may contact the author at [email protected].
Mesh fixation method had no impact on chronic postop pain in TAPP
WASHINGTON – Mesh fixation technique is not a factor in persistent pain after transabdominal preperitoneal (TAPP) groin hernia surgery, a study showed.
“Persistent pain is a well-known phenomenon after groin hernia surgery,” said Jakob Burcharth, MD, of University Hospital of Sjaelland Koge (Denmark). “The way that we fixate the mesh in laparoscopic surgery has been hypothesized to have an [impact] on the risk of persistent pain.”
In a study presented by Dr. Burcharth and his associates at the American College of Surgeons Clinical Congress, a total of 1,421 patients were examined. Each patient completed a validated pain questionnaire through the Danish Hernia Database from 2009 to 2012. The patients were divided into two groups: Group 1 had spray fibrin sealant (34%) for mesh fixation, and group 2 had tacks (66%). The results showed no difference between the groups in terms of pain in getting up from a chair, sitting or standing for more than 30 minutes, walking stairs, driving a car, or exercising, or in the need for postoperative analgesics or postoperative sick leave (all P greater than .20).
Dr. Burcharth concluded that a high number of patients reported persistent pain regardless of mesh fixation technique, which emphasizes the need for preoperative information.
Dr. Burcharth reported having no relevant financial disclosures.
WASHINGTON – Mesh fixation technique is not a factor in persistent pain after transabdominal preperitoneal (TAPP) groin hernia surgery, a study showed.
“Persistent pain is a well-known phenomenon after groin hernia surgery,” said Jakob Burcharth, MD, of University Hospital of Sjaelland Koge (Denmark). “The way that we fixate the mesh in laparoscopic surgery has been hypothesized to have an [impact] on the risk of persistent pain.”
In a study presented by Dr. Burcharth and his associates at the American College of Surgeons Clinical Congress, a total of 1,421 patients were examined. Each patient completed a validated pain questionnaire through the Danish Hernia Database from 2009 to 2012. The patients were divided into two groups: Group 1 had spray fibrin sealant (34%) for mesh fixation, and group 2 had tacks (66%). The results showed no difference between the groups in terms of pain in getting up from a chair, sitting or standing for more than 30 minutes, walking stairs, driving a car, or exercising, or in the need for postoperative analgesics or postoperative sick leave (all P greater than .20).
Dr. Burcharth concluded that a high number of patients reported persistent pain regardless of mesh fixation technique, which emphasizes the need for preoperative information.
Dr. Burcharth reported having no relevant financial disclosures.
WASHINGTON – Mesh fixation technique is not a factor in persistent pain after transabdominal preperitoneal (TAPP) groin hernia surgery, a study showed.
“Persistent pain is a well-known phenomenon after groin hernia surgery,” said Jakob Burcharth, MD, of University Hospital of Sjaelland Koge (Denmark). “The way that we fixate the mesh in laparoscopic surgery has been hypothesized to have an [impact] on the risk of persistent pain.”
In a study presented by Dr. Burcharth and his associates at the American College of Surgeons Clinical Congress, a total of 1,421 patients were examined. Each patient completed a validated pain questionnaire through the Danish Hernia Database from 2009 to 2012. The patients were divided into two groups: Group 1 had spray fibrin sealant (34%) for mesh fixation, and group 2 had tacks (66%). The results showed no difference between the groups in terms of pain in getting up from a chair, sitting or standing for more than 30 minutes, walking stairs, driving a car, or exercising, or in the need for postoperative analgesics or postoperative sick leave (all P greater than .20).
Dr. Burcharth concluded that a high number of patients reported persistent pain regardless of mesh fixation technique, which emphasizes the need for preoperative information.
Dr. Burcharth reported having no relevant financial disclosures.
Key clinical point:
Major finding: There was no difference between the groups in terms of pain in getting up from a chair, sitting or standing for more than 30 minutes, walking stairs, driving a car, or doing exercise, or in the need for postoperative analgesics or postoperative sick leave (all P greater than .20).
Data source: 1,421 patients who were examined and completed a validated pain questionnaire.
Disclosures: Dr. Burcharth reported having no relevant financial disclosures.
How to reduce readmissions after vascular procedures
CHICAGO – As Medicare ratchets up penalties for readmissions and hospitals scrutinize procedures such as carotid interventions and lower extremity bypass that have traditionally high readmission rates, a four-phase model that assesses readmission risks could help vascular surgeons and their institutions keep patients from returning after vascular procedures, according to a presentation at a symposium on vascular surgery sponsored by Northwestern University.
“The rate of readmissions for vascular surgery is 50% higher than all other surgical interventions,” said Karen Ho, MD, of Northwestern University, Chicago. She noted that the reported readmission rates for abdominal aortic aneurysm (AAA) repair were 13.3% for endovascular and 12.8% for open (Ann Surg. 2012;256:595-605). For carotid endarterectomy (CEA), a large study reported a readmission rate of 9.6% (J Am Coll Cardiol. 2015;65:1398-1408), while a sampling of the American College of Surgeons National Surgical Quality Improvement Program (ACS-NSQIP) database showed a 30-day readmission rate of 16.5% for lower extremity bypass (J Vasc Surg. 2014;60:1266-74).
Dr. Ho said one model vascular specialists and hospitals could employ to curtail readmissions was first reported in 2012 by Benjamin S. Brooke, MD, of Dartmouth-Hitchcock Medical Center, Lebanon, N.H., and his colleagues (J Vasc Surg. 2012;56:556-62). This model focuses on the following four phases: 1) develop a deeper understanding of the patient’s preexisting health conditions before surgery; 2) target the in-hospital postoperative period for possible intervention; 3) focus on discharge planning; and 4) determine at the actual readmission event itself if the patient should go to an alternative setting.
“I think as surgeons we often focus on the patients, the procedure, and what goes on in the hospital, but discharge planning and execution is potentially just as important in preventing readmissions,” Dr. Ho said. “This includes things like medication reconciliation, involvement of family, the primary care doctor, the nursing home or rehab facility, and the timing and scheduling of follow-up visits.”
However, she noted that unaccounted factors can also contribute to readmission risk. These can include availability of family to provide support; history of substance abuse; functional status; socioeconomic status; and medical history.
Dr. Ho noted that understanding the reasons for readmission can help vascular specialists gain a deeper understanding of their underlying causes. For example, wound complications top the list in readmissions of numerous vascular procedures, including AAA repair and lower extremity revascularization, but other causes are linked to specific procedures. “If you look at the endovascular repair group in AAA repairs, aneurysm and graft complications were the third most common reason for unplanned 30-day readmission,” she said. A multivariate analysis showed that while preoperative comorbidities had a modest effect on readmission rates after AAA repair, postoperative factors such as complications extending patients’ length of stay and discharge to a setting other than home had a profound effect (Ann Surg. 2012;256:595-605).
In carotid procedures, Dr. Ho noted that carotid artery stenting and CEA had 30-day readmission rates of around 10% (Stroke. 2012;43:2408-16), although CEA seemed to have a slight advantage. Cardiac complications, headache, and bleeding were the top reasons for readmissions for carotid procedures, Dr. Ho said. “In a multivariate analysis, a history of coronary artery bypass and any postoperative complication were associated with readmission,” she said (Vasc Endovascular Surg. 2014;48:217-23).
However, many risk factors for readmission are nonmodifiable, such as patient age 80 and up, or a history of renal failure, heart failure, or diabetes – all characteristics that made patients more prone to readmission after carotid procedures.
Likewise in lower extremity revascularization, nonmodifiable risk factors – end-stage renal disease, heart failure, or tissue loss indication – were prime culprits for readmissions, Dr. Ho noted (J Vasc Surg. 2013;57:955-62). “But also the strongest predictors for readmission included surgical site infections postoperatively as well as graft complications,” she said.
“Risk prediction models for readmissions perform poorly, which makes it difficult to identify high risk and to implement clinically actionable plans to reduce readmissions,” Dr. Ho said. “It also raises the question of whether other important variables, such as social determinants, which may disproportionately affect disadvantaged patients, maybe should be included in these risk prevention models to increase their predicative value.”
Until or if Medicare adjusts its risk evaluation measures accordingly to more accurately reflect the influence of underlying variables such as socioeconomic status and medical history, vascular specialists and their institutions will be pressed to develop programs to reduce readmissions.
Dr. Ho had no relevant financial relationships to disclose.
CHICAGO – As Medicare ratchets up penalties for readmissions and hospitals scrutinize procedures such as carotid interventions and lower extremity bypass that have traditionally high readmission rates, a four-phase model that assesses readmission risks could help vascular surgeons and their institutions keep patients from returning after vascular procedures, according to a presentation at a symposium on vascular surgery sponsored by Northwestern University.
“The rate of readmissions for vascular surgery is 50% higher than all other surgical interventions,” said Karen Ho, MD, of Northwestern University, Chicago. She noted that the reported readmission rates for abdominal aortic aneurysm (AAA) repair were 13.3% for endovascular and 12.8% for open (Ann Surg. 2012;256:595-605). For carotid endarterectomy (CEA), a large study reported a readmission rate of 9.6% (J Am Coll Cardiol. 2015;65:1398-1408), while a sampling of the American College of Surgeons National Surgical Quality Improvement Program (ACS-NSQIP) database showed a 30-day readmission rate of 16.5% for lower extremity bypass (J Vasc Surg. 2014;60:1266-74).
Dr. Ho said one model vascular specialists and hospitals could employ to curtail readmissions was first reported in 2012 by Benjamin S. Brooke, MD, of Dartmouth-Hitchcock Medical Center, Lebanon, N.H., and his colleagues (J Vasc Surg. 2012;56:556-62). This model focuses on the following four phases: 1) develop a deeper understanding of the patient’s preexisting health conditions before surgery; 2) target the in-hospital postoperative period for possible intervention; 3) focus on discharge planning; and 4) determine at the actual readmission event itself if the patient should go to an alternative setting.
“I think as surgeons we often focus on the patients, the procedure, and what goes on in the hospital, but discharge planning and execution is potentially just as important in preventing readmissions,” Dr. Ho said. “This includes things like medication reconciliation, involvement of family, the primary care doctor, the nursing home or rehab facility, and the timing and scheduling of follow-up visits.”
However, she noted that unaccounted factors can also contribute to readmission risk. These can include availability of family to provide support; history of substance abuse; functional status; socioeconomic status; and medical history.
Dr. Ho noted that understanding the reasons for readmission can help vascular specialists gain a deeper understanding of their underlying causes. For example, wound complications top the list in readmissions of numerous vascular procedures, including AAA repair and lower extremity revascularization, but other causes are linked to specific procedures. “If you look at the endovascular repair group in AAA repairs, aneurysm and graft complications were the third most common reason for unplanned 30-day readmission,” she said. A multivariate analysis showed that while preoperative comorbidities had a modest effect on readmission rates after AAA repair, postoperative factors such as complications extending patients’ length of stay and discharge to a setting other than home had a profound effect (Ann Surg. 2012;256:595-605).
In carotid procedures, Dr. Ho noted that carotid artery stenting and CEA had 30-day readmission rates of around 10% (Stroke. 2012;43:2408-16), although CEA seemed to have a slight advantage. Cardiac complications, headache, and bleeding were the top reasons for readmissions for carotid procedures, Dr. Ho said. “In a multivariate analysis, a history of coronary artery bypass and any postoperative complication were associated with readmission,” she said (Vasc Endovascular Surg. 2014;48:217-23).
However, many risk factors for readmission are nonmodifiable, such as patient age 80 and up, or a history of renal failure, heart failure, or diabetes – all characteristics that made patients more prone to readmission after carotid procedures.
Likewise in lower extremity revascularization, nonmodifiable risk factors – end-stage renal disease, heart failure, or tissue loss indication – were prime culprits for readmissions, Dr. Ho noted (J Vasc Surg. 2013;57:955-62). “But also the strongest predictors for readmission included surgical site infections postoperatively as well as graft complications,” she said.
“Risk prediction models for readmissions perform poorly, which makes it difficult to identify high risk and to implement clinically actionable plans to reduce readmissions,” Dr. Ho said. “It also raises the question of whether other important variables, such as social determinants, which may disproportionately affect disadvantaged patients, maybe should be included in these risk prevention models to increase their predicative value.”
Until or if Medicare adjusts its risk evaluation measures accordingly to more accurately reflect the influence of underlying variables such as socioeconomic status and medical history, vascular specialists and their institutions will be pressed to develop programs to reduce readmissions.
Dr. Ho had no relevant financial relationships to disclose.
CHICAGO – As Medicare ratchets up penalties for readmissions and hospitals scrutinize procedures such as carotid interventions and lower extremity bypass that have traditionally high readmission rates, a four-phase model that assesses readmission risks could help vascular surgeons and their institutions keep patients from returning after vascular procedures, according to a presentation at a symposium on vascular surgery sponsored by Northwestern University.
“The rate of readmissions for vascular surgery is 50% higher than all other surgical interventions,” said Karen Ho, MD, of Northwestern University, Chicago. She noted that the reported readmission rates for abdominal aortic aneurysm (AAA) repair were 13.3% for endovascular and 12.8% for open (Ann Surg. 2012;256:595-605). For carotid endarterectomy (CEA), a large study reported a readmission rate of 9.6% (J Am Coll Cardiol. 2015;65:1398-1408), while a sampling of the American College of Surgeons National Surgical Quality Improvement Program (ACS-NSQIP) database showed a 30-day readmission rate of 16.5% for lower extremity bypass (J Vasc Surg. 2014;60:1266-74).
Dr. Ho said one model vascular specialists and hospitals could employ to curtail readmissions was first reported in 2012 by Benjamin S. Brooke, MD, of Dartmouth-Hitchcock Medical Center, Lebanon, N.H., and his colleagues (J Vasc Surg. 2012;56:556-62). This model focuses on the following four phases: 1) develop a deeper understanding of the patient’s preexisting health conditions before surgery; 2) target the in-hospital postoperative period for possible intervention; 3) focus on discharge planning; and 4) determine at the actual readmission event itself if the patient should go to an alternative setting.
“I think as surgeons we often focus on the patients, the procedure, and what goes on in the hospital, but discharge planning and execution is potentially just as important in preventing readmissions,” Dr. Ho said. “This includes things like medication reconciliation, involvement of family, the primary care doctor, the nursing home or rehab facility, and the timing and scheduling of follow-up visits.”
However, she noted that unaccounted factors can also contribute to readmission risk. These can include availability of family to provide support; history of substance abuse; functional status; socioeconomic status; and medical history.
Dr. Ho noted that understanding the reasons for readmission can help vascular specialists gain a deeper understanding of their underlying causes. For example, wound complications top the list in readmissions of numerous vascular procedures, including AAA repair and lower extremity revascularization, but other causes are linked to specific procedures. “If you look at the endovascular repair group in AAA repairs, aneurysm and graft complications were the third most common reason for unplanned 30-day readmission,” she said. A multivariate analysis showed that while preoperative comorbidities had a modest effect on readmission rates after AAA repair, postoperative factors such as complications extending patients’ length of stay and discharge to a setting other than home had a profound effect (Ann Surg. 2012;256:595-605).
In carotid procedures, Dr. Ho noted that carotid artery stenting and CEA had 30-day readmission rates of around 10% (Stroke. 2012;43:2408-16), although CEA seemed to have a slight advantage. Cardiac complications, headache, and bleeding were the top reasons for readmissions for carotid procedures, Dr. Ho said. “In a multivariate analysis, a history of coronary artery bypass and any postoperative complication were associated with readmission,” she said (Vasc Endovascular Surg. 2014;48:217-23).
However, many risk factors for readmission are nonmodifiable, such as patient age 80 and up, or a history of renal failure, heart failure, or diabetes – all characteristics that made patients more prone to readmission after carotid procedures.
Likewise in lower extremity revascularization, nonmodifiable risk factors – end-stage renal disease, heart failure, or tissue loss indication – were prime culprits for readmissions, Dr. Ho noted (J Vasc Surg. 2013;57:955-62). “But also the strongest predictors for readmission included surgical site infections postoperatively as well as graft complications,” she said.
“Risk prediction models for readmissions perform poorly, which makes it difficult to identify high risk and to implement clinically actionable plans to reduce readmissions,” Dr. Ho said. “It also raises the question of whether other important variables, such as social determinants, which may disproportionately affect disadvantaged patients, maybe should be included in these risk prevention models to increase their predicative value.”
Until or if Medicare adjusts its risk evaluation measures accordingly to more accurately reflect the influence of underlying variables such as socioeconomic status and medical history, vascular specialists and their institutions will be pressed to develop programs to reduce readmissions.
Dr. Ho had no relevant financial relationships to disclose.
AT THE NORTHWESTERN VASCULAR SYMPOSIUM
Key clinical point: A four-phase model that assesses readmission risks could help vascular surgeons and their institutions keep patients from returning after procedures.
Major finding: Readmission rates for cardiac and vascular interventions range from around 13% for aortic aneurysm repair to 20% after lower extremity revascularization.
Data source: Literature review.
Disclosures: Dr. Ho reported having no financial disclosures.