User login
Treating fractures in elderly patients: Beyond the broken bone
While half the fracture-prevention battle is getting people diagnosed with low bone density, nearly 80% of older Americans who suffer bone breaks are not tested or treated for osteoporosis. Fractures associated with aging and diminished bone mineral density exact an enormous toll on patients’ lives and cost the health care system billions of dollars annually according to Bone Health and Osteoporosis: A Report of the Surgeon General. But current gaps in patient education and bone density screening are huge.
“It’s concerning that older patients at risk for fracture are often not screened to determine their risk factors contributing to osteoporosis and patients are not educated about fracture prevention,” said Meryl S. LeBoff, MD, an endocrinologist at Brigham and Women’s Hospital, and chief of calcium and bone section, and professor of medicine, at Harvard Medical School, Boston. “Furthermore, the majority of highest-risk women and men who do have fractures are not screened and they do not receive effective, [Food and Drug Administration]–approved therapies.”
Recent guidelines
Screening with dual-energy x-ray absorptiometry (DEXA) is recommended for all women at age 65 and all men at age 70. But the occasion of a fracture in an older person who has not yet met these age thresholds should prompt a bone density assessment.
“Doctors need to stress that one in two women and one in four men over age 50 will have a fracture in their remaining lifetimes,” Dr. LeBoff said. ”Primary care doctors play a critical role in ordering timely bone densitometry for both sexes.
If an older patient has been treated for a fracture, the main goal going forward is to prevent another one, for which the risk is highest in the 2 years after the incident fracture.”
According to Kendall F. Moseley, MD, clinical director of the division of endocrinology, diabetes & metabolism at Johns Hopkins Medicine in Baltimore, “Elderly patients need to understand that a fracture at their age is like a heart attack of the bone,” she said, adding that just as cardiovascular risk factors such as high blood pressure and blood lipids are silent before a stroke or infarction, the bone thinning of old age is also silent.
Endocrinologist Jennifer J. Kelly, DO, director of the metabolic bone program and an associate professor at the University of Vermont Medical Center in Burlington, said a fracture in anyone over age 50 that appears not to have resulted from a traumatic blow, is a compelling reason to order a DEXA exam.
Nahid J. Rianon, MBBS/MD, DrPH, assistant professor of the division of geriatric medicine at the UTHealth McGovern Medical School, Houston, goes further: “Any fracture in someone age 50 and older warrants screening for osteoporosis. And if the fracture is nontraumatic, that is by definition a clinical diagnosis of osteoporosis regardless of normal results on bone density tests and they should be treated medically. There are aspects of bone that we still can’t measure in the clinical setting.”
If DEXA is not accessible, fracture risk over the next 10 years can be evaluated based on multiple patient characteristics and medical history using the online FRAX calculator.
Just a 3% risk of hip fracture on FRAX is considered an indication to begin medical osteoporosis treatment in the United States regardless of bone density test results, Dr. Rianon said.
Fracture management
Whether a senior suffers a traumatic fracture or an osteoporosis-related fragility fracture, older age can impede the healing process in some. Senescence may also increase systemic proinflammatory status, according to Clark and colleagues, writing in Current Osteoporosis Reports.
They called for research to develop more directed treatment options for the elderly population.
Dr. Rianon noted that healing may also be affected by a decrease in muscle mass, which plays a role in holding the bone in place. “But it is still controversial how changing metabolic factors affect bone healing in the elderly.”
However, countered Dr. Kelly, fractures in elderly patients are not necessarily less likely to mend – if osteoporosis is not present. “Many heal very well – it really depends more upon their overall health and medical history. Whether or not a person requires surgery depends more upon the extent of the fracture and if the bone is able to align and heal appropriately without surgery.”
Fracture sites
Spine. According to the American Academy of Orthopedic Surgeons the earliest and most frequent site of fragility fractures in the elderly is the spine. Most vertebral fracture pain improves within 3 months without specific treatment. A short period of rest, limited analgesic use, and possible back bracing may help as the fractures heal on their own. But if pain is severe and persistent, vertebral augmentation with percutaneous kyphoplasty or vertebroplasty may be an option. These procedures, however, can destabilize surrounding discs because of the greater thickness of the injected cement.
Hip. The most dangerous fractures occur in the hip. These carry at least a 20% risk of death in the first postoperative year and must be treated surgically. Those in the proximal femur, the head, or the femoral neck will usually need hip replacement, but if the break is farther down, it may be repaired with cement, screws, plates, and rods.
Distal radius. Outcomes of wrist fractures may be positive without surgical intervention, according to a recent retrospective analysis from Turkey by Yalin and colleagues. In a comparison of clinical outcomes in seniors aged 70-89 and assigned to cast immobilization or various surgical treatments for distal radius fractures, no statistically significant difference was found in patient-reported disability scores and range of motion values between casting and surgery in the first postoperative year.
Other sites. Fractures in the elderly are not uncommon in the shoulder, distal radius, cubitus, proximal humerus, and humerus. These fractures are often treated without surgery, but nevertheless signal a high risk for additional fractures.
Bone-enhancing medications
Even in the absence of diagnosed low bone density or osteoporosis, anabolic agents such as the synthetic human parathyroid hormones abaloparatide (Tymlos) and teriparatide (Forteo) may be used to help in some cases with a bad healing prognosis and may also be used for people undergoing surgeries such as a spinal fusion, but there are not clinical guidelines. “We receive referrals regularly for this treatment from our orthopedics colleagues, but it is considered an off-label use,” Dr. Kelly said.
The anabolics teriparatide and romosozumab (Evenity) have proved effective in lowering fractures in high-risk older women.
Post fracture
After recovering from a fracture, elderly people are strongly advised to make lifestyle changes to boost bone health and reduce risk of further fractures, said Willy M. Valencia, MD, a geriatrician-endocrinologist at the Cleveland Clinic. Apart from active daily living, he recommends several types of formal exercise to promote bone formation; increase muscle mass, strength, and flexibility; and improve endurance, balance, and gait. The National Institute on Aging outlines suitable exercise programs for seniors.
“These exercises will help reduce the risk of falling and to avoid more fractures,” he said. “Whether a patient has been exercising before the fracture or not, they may feel some reticence or reluctance to take up exercise afterwards because they’re afraid of having another fracture, but they should understand that their fracture risk increases if they remain sedentary. They should start slowly but they can’t be sitting all day.”
Even before it’s possible to exercise at the healing fracture site, added Dr. Rianon, its advisable to work other areas of the body. “Overall mobility is important, and exercising other parts of the body can stimulate strength and help prevent falling.”
In other postsurgical measures, a bone-friendly diet rich in calcium and vitamin D, as well as supplementation with these vital nutrients, is essential to lower the risk of falling.
Fall prevention is paramount, said Dr. Valencia. While exercise can improve, gait, balance, and endurance, logistical measures may also be necessary. Seniors may have to move to a one-floor domicile with no stairs to negotiate. At the very least, they need to fall-proof their daily lives by upgrading their eyeglasses and home lighting, eliminating obstacles and loose carpets, fixing bannisters, and installing bathroom handrails. Some may need assistive devices for walking, especially outdoors in slippery conditions.
At the end of the day, the role of the primary physician in screening for bone problems before fracture and postsurgical care is key. “Risk factors for osteoporosis and fracture risk must be added to the patient’s chart,” said Dr. Rianon. Added Dr. Moseley. “No matter how busy they are, my hope is that primary care physicians will not put patients’ bone health at the bottom of the clinical agenda.”
While half the fracture-prevention battle is getting people diagnosed with low bone density, nearly 80% of older Americans who suffer bone breaks are not tested or treated for osteoporosis. Fractures associated with aging and diminished bone mineral density exact an enormous toll on patients’ lives and cost the health care system billions of dollars annually according to Bone Health and Osteoporosis: A Report of the Surgeon General. But current gaps in patient education and bone density screening are huge.
“It’s concerning that older patients at risk for fracture are often not screened to determine their risk factors contributing to osteoporosis and patients are not educated about fracture prevention,” said Meryl S. LeBoff, MD, an endocrinologist at Brigham and Women’s Hospital, and chief of calcium and bone section, and professor of medicine, at Harvard Medical School, Boston. “Furthermore, the majority of highest-risk women and men who do have fractures are not screened and they do not receive effective, [Food and Drug Administration]–approved therapies.”
Recent guidelines
Screening with dual-energy x-ray absorptiometry (DEXA) is recommended for all women at age 65 and all men at age 70. But the occasion of a fracture in an older person who has not yet met these age thresholds should prompt a bone density assessment.
“Doctors need to stress that one in two women and one in four men over age 50 will have a fracture in their remaining lifetimes,” Dr. LeBoff said. ”Primary care doctors play a critical role in ordering timely bone densitometry for both sexes.
If an older patient has been treated for a fracture, the main goal going forward is to prevent another one, for which the risk is highest in the 2 years after the incident fracture.”
According to Kendall F. Moseley, MD, clinical director of the division of endocrinology, diabetes & metabolism at Johns Hopkins Medicine in Baltimore, “Elderly patients need to understand that a fracture at their age is like a heart attack of the bone,” she said, adding that just as cardiovascular risk factors such as high blood pressure and blood lipids are silent before a stroke or infarction, the bone thinning of old age is also silent.
Endocrinologist Jennifer J. Kelly, DO, director of the metabolic bone program and an associate professor at the University of Vermont Medical Center in Burlington, said a fracture in anyone over age 50 that appears not to have resulted from a traumatic blow, is a compelling reason to order a DEXA exam.
Nahid J. Rianon, MBBS/MD, DrPH, assistant professor of the division of geriatric medicine at the UTHealth McGovern Medical School, Houston, goes further: “Any fracture in someone age 50 and older warrants screening for osteoporosis. And if the fracture is nontraumatic, that is by definition a clinical diagnosis of osteoporosis regardless of normal results on bone density tests and they should be treated medically. There are aspects of bone that we still can’t measure in the clinical setting.”
If DEXA is not accessible, fracture risk over the next 10 years can be evaluated based on multiple patient characteristics and medical history using the online FRAX calculator.
Just a 3% risk of hip fracture on FRAX is considered an indication to begin medical osteoporosis treatment in the United States regardless of bone density test results, Dr. Rianon said.
Fracture management
Whether a senior suffers a traumatic fracture or an osteoporosis-related fragility fracture, older age can impede the healing process in some. Senescence may also increase systemic proinflammatory status, according to Clark and colleagues, writing in Current Osteoporosis Reports.
They called for research to develop more directed treatment options for the elderly population.
Dr. Rianon noted that healing may also be affected by a decrease in muscle mass, which plays a role in holding the bone in place. “But it is still controversial how changing metabolic factors affect bone healing in the elderly.”
However, countered Dr. Kelly, fractures in elderly patients are not necessarily less likely to mend – if osteoporosis is not present. “Many heal very well – it really depends more upon their overall health and medical history. Whether or not a person requires surgery depends more upon the extent of the fracture and if the bone is able to align and heal appropriately without surgery.”
Fracture sites
Spine. According to the American Academy of Orthopedic Surgeons the earliest and most frequent site of fragility fractures in the elderly is the spine. Most vertebral fracture pain improves within 3 months without specific treatment. A short period of rest, limited analgesic use, and possible back bracing may help as the fractures heal on their own. But if pain is severe and persistent, vertebral augmentation with percutaneous kyphoplasty or vertebroplasty may be an option. These procedures, however, can destabilize surrounding discs because of the greater thickness of the injected cement.
Hip. The most dangerous fractures occur in the hip. These carry at least a 20% risk of death in the first postoperative year and must be treated surgically. Those in the proximal femur, the head, or the femoral neck will usually need hip replacement, but if the break is farther down, it may be repaired with cement, screws, plates, and rods.
Distal radius. Outcomes of wrist fractures may be positive without surgical intervention, according to a recent retrospective analysis from Turkey by Yalin and colleagues. In a comparison of clinical outcomes in seniors aged 70-89 and assigned to cast immobilization or various surgical treatments for distal radius fractures, no statistically significant difference was found in patient-reported disability scores and range of motion values between casting and surgery in the first postoperative year.
Other sites. Fractures in the elderly are not uncommon in the shoulder, distal radius, cubitus, proximal humerus, and humerus. These fractures are often treated without surgery, but nevertheless signal a high risk for additional fractures.
Bone-enhancing medications
Even in the absence of diagnosed low bone density or osteoporosis, anabolic agents such as the synthetic human parathyroid hormones abaloparatide (Tymlos) and teriparatide (Forteo) may be used to help in some cases with a bad healing prognosis and may also be used for people undergoing surgeries such as a spinal fusion, but there are not clinical guidelines. “We receive referrals regularly for this treatment from our orthopedics colleagues, but it is considered an off-label use,” Dr. Kelly said.
The anabolics teriparatide and romosozumab (Evenity) have proved effective in lowering fractures in high-risk older women.
Post fracture
After recovering from a fracture, elderly people are strongly advised to make lifestyle changes to boost bone health and reduce risk of further fractures, said Willy M. Valencia, MD, a geriatrician-endocrinologist at the Cleveland Clinic. Apart from active daily living, he recommends several types of formal exercise to promote bone formation; increase muscle mass, strength, and flexibility; and improve endurance, balance, and gait. The National Institute on Aging outlines suitable exercise programs for seniors.
“These exercises will help reduce the risk of falling and to avoid more fractures,” he said. “Whether a patient has been exercising before the fracture or not, they may feel some reticence or reluctance to take up exercise afterwards because they’re afraid of having another fracture, but they should understand that their fracture risk increases if they remain sedentary. They should start slowly but they can’t be sitting all day.”
Even before it’s possible to exercise at the healing fracture site, added Dr. Rianon, its advisable to work other areas of the body. “Overall mobility is important, and exercising other parts of the body can stimulate strength and help prevent falling.”
In other postsurgical measures, a bone-friendly diet rich in calcium and vitamin D, as well as supplementation with these vital nutrients, is essential to lower the risk of falling.
Fall prevention is paramount, said Dr. Valencia. While exercise can improve, gait, balance, and endurance, logistical measures may also be necessary. Seniors may have to move to a one-floor domicile with no stairs to negotiate. At the very least, they need to fall-proof their daily lives by upgrading their eyeglasses and home lighting, eliminating obstacles and loose carpets, fixing bannisters, and installing bathroom handrails. Some may need assistive devices for walking, especially outdoors in slippery conditions.
At the end of the day, the role of the primary physician in screening for bone problems before fracture and postsurgical care is key. “Risk factors for osteoporosis and fracture risk must be added to the patient’s chart,” said Dr. Rianon. Added Dr. Moseley. “No matter how busy they are, my hope is that primary care physicians will not put patients’ bone health at the bottom of the clinical agenda.”
While half the fracture-prevention battle is getting people diagnosed with low bone density, nearly 80% of older Americans who suffer bone breaks are not tested or treated for osteoporosis. Fractures associated with aging and diminished bone mineral density exact an enormous toll on patients’ lives and cost the health care system billions of dollars annually according to Bone Health and Osteoporosis: A Report of the Surgeon General. But current gaps in patient education and bone density screening are huge.
“It’s concerning that older patients at risk for fracture are often not screened to determine their risk factors contributing to osteoporosis and patients are not educated about fracture prevention,” said Meryl S. LeBoff, MD, an endocrinologist at Brigham and Women’s Hospital, and chief of calcium and bone section, and professor of medicine, at Harvard Medical School, Boston. “Furthermore, the majority of highest-risk women and men who do have fractures are not screened and they do not receive effective, [Food and Drug Administration]–approved therapies.”
Recent guidelines
Screening with dual-energy x-ray absorptiometry (DEXA) is recommended for all women at age 65 and all men at age 70. But the occasion of a fracture in an older person who has not yet met these age thresholds should prompt a bone density assessment.
“Doctors need to stress that one in two women and one in four men over age 50 will have a fracture in their remaining lifetimes,” Dr. LeBoff said. ”Primary care doctors play a critical role in ordering timely bone densitometry for both sexes.
If an older patient has been treated for a fracture, the main goal going forward is to prevent another one, for which the risk is highest in the 2 years after the incident fracture.”
According to Kendall F. Moseley, MD, clinical director of the division of endocrinology, diabetes & metabolism at Johns Hopkins Medicine in Baltimore, “Elderly patients need to understand that a fracture at their age is like a heart attack of the bone,” she said, adding that just as cardiovascular risk factors such as high blood pressure and blood lipids are silent before a stroke or infarction, the bone thinning of old age is also silent.
Endocrinologist Jennifer J. Kelly, DO, director of the metabolic bone program and an associate professor at the University of Vermont Medical Center in Burlington, said a fracture in anyone over age 50 that appears not to have resulted from a traumatic blow, is a compelling reason to order a DEXA exam.
Nahid J. Rianon, MBBS/MD, DrPH, assistant professor of the division of geriatric medicine at the UTHealth McGovern Medical School, Houston, goes further: “Any fracture in someone age 50 and older warrants screening for osteoporosis. And if the fracture is nontraumatic, that is by definition a clinical diagnosis of osteoporosis regardless of normal results on bone density tests and they should be treated medically. There are aspects of bone that we still can’t measure in the clinical setting.”
If DEXA is not accessible, fracture risk over the next 10 years can be evaluated based on multiple patient characteristics and medical history using the online FRAX calculator.
Just a 3% risk of hip fracture on FRAX is considered an indication to begin medical osteoporosis treatment in the United States regardless of bone density test results, Dr. Rianon said.
Fracture management
Whether a senior suffers a traumatic fracture or an osteoporosis-related fragility fracture, older age can impede the healing process in some. Senescence may also increase systemic proinflammatory status, according to Clark and colleagues, writing in Current Osteoporosis Reports.
They called for research to develop more directed treatment options for the elderly population.
Dr. Rianon noted that healing may also be affected by a decrease in muscle mass, which plays a role in holding the bone in place. “But it is still controversial how changing metabolic factors affect bone healing in the elderly.”
However, countered Dr. Kelly, fractures in elderly patients are not necessarily less likely to mend – if osteoporosis is not present. “Many heal very well – it really depends more upon their overall health and medical history. Whether or not a person requires surgery depends more upon the extent of the fracture and if the bone is able to align and heal appropriately without surgery.”
Fracture sites
Spine. According to the American Academy of Orthopedic Surgeons the earliest and most frequent site of fragility fractures in the elderly is the spine. Most vertebral fracture pain improves within 3 months without specific treatment. A short period of rest, limited analgesic use, and possible back bracing may help as the fractures heal on their own. But if pain is severe and persistent, vertebral augmentation with percutaneous kyphoplasty or vertebroplasty may be an option. These procedures, however, can destabilize surrounding discs because of the greater thickness of the injected cement.
Hip. The most dangerous fractures occur in the hip. These carry at least a 20% risk of death in the first postoperative year and must be treated surgically. Those in the proximal femur, the head, or the femoral neck will usually need hip replacement, but if the break is farther down, it may be repaired with cement, screws, plates, and rods.
Distal radius. Outcomes of wrist fractures may be positive without surgical intervention, according to a recent retrospective analysis from Turkey by Yalin and colleagues. In a comparison of clinical outcomes in seniors aged 70-89 and assigned to cast immobilization or various surgical treatments for distal radius fractures, no statistically significant difference was found in patient-reported disability scores and range of motion values between casting and surgery in the first postoperative year.
Other sites. Fractures in the elderly are not uncommon in the shoulder, distal radius, cubitus, proximal humerus, and humerus. These fractures are often treated without surgery, but nevertheless signal a high risk for additional fractures.
Bone-enhancing medications
Even in the absence of diagnosed low bone density or osteoporosis, anabolic agents such as the synthetic human parathyroid hormones abaloparatide (Tymlos) and teriparatide (Forteo) may be used to help in some cases with a bad healing prognosis and may also be used for people undergoing surgeries such as a spinal fusion, but there are not clinical guidelines. “We receive referrals regularly for this treatment from our orthopedics colleagues, but it is considered an off-label use,” Dr. Kelly said.
The anabolics teriparatide and romosozumab (Evenity) have proved effective in lowering fractures in high-risk older women.
Post fracture
After recovering from a fracture, elderly people are strongly advised to make lifestyle changes to boost bone health and reduce risk of further fractures, said Willy M. Valencia, MD, a geriatrician-endocrinologist at the Cleveland Clinic. Apart from active daily living, he recommends several types of formal exercise to promote bone formation; increase muscle mass, strength, and flexibility; and improve endurance, balance, and gait. The National Institute on Aging outlines suitable exercise programs for seniors.
“These exercises will help reduce the risk of falling and to avoid more fractures,” he said. “Whether a patient has been exercising before the fracture or not, they may feel some reticence or reluctance to take up exercise afterwards because they’re afraid of having another fracture, but they should understand that their fracture risk increases if they remain sedentary. They should start slowly but they can’t be sitting all day.”
Even before it’s possible to exercise at the healing fracture site, added Dr. Rianon, its advisable to work other areas of the body. “Overall mobility is important, and exercising other parts of the body can stimulate strength and help prevent falling.”
In other postsurgical measures, a bone-friendly diet rich in calcium and vitamin D, as well as supplementation with these vital nutrients, is essential to lower the risk of falling.
Fall prevention is paramount, said Dr. Valencia. While exercise can improve, gait, balance, and endurance, logistical measures may also be necessary. Seniors may have to move to a one-floor domicile with no stairs to negotiate. At the very least, they need to fall-proof their daily lives by upgrading their eyeglasses and home lighting, eliminating obstacles and loose carpets, fixing bannisters, and installing bathroom handrails. Some may need assistive devices for walking, especially outdoors in slippery conditions.
At the end of the day, the role of the primary physician in screening for bone problems before fracture and postsurgical care is key. “Risk factors for osteoporosis and fracture risk must be added to the patient’s chart,” said Dr. Rianon. Added Dr. Moseley. “No matter how busy they are, my hope is that primary care physicians will not put patients’ bone health at the bottom of the clinical agenda.”
‘Missed opportunities’ for accurate diagnosing of women with vaginitis
Although the standard of care of diagnosing vaginitis is clinical evaluation, many practices do not perform accurate and comprehensive clinical examinations for a variety for reasons, and the Centers for Disease Control and Prevention currently recommends molecular testing, wrote Casey N. Pinto, PhD, of Penn State University, Hershey, and colleagues. The CDC also recommends testing women with vaginitis for Chlamydia trachomatis (CT) and Neissaria gonorrhoeae (NG) given the high rate of coinfections between vaginitis and these sexually transmitted infections, but data on cotesting in clinical practice are limited, they said.
In a study published in Sexually Transmitted Diseases, the researchers reviewed data from a commercial administrative claims database for 1,359,289 women aged 18-50 years who were diagnosed with vaginitis between 2012 and 2017.
The women were categorized into groups based on type of vaginitis diagnosis: nucleic amplification test (NAAT), DNA probe test, traditional lab test, and those diagnosed clinically at an index visit but with no CPT code for further testing.
Overall, nearly half of the women (49.2%) had no CPT code for further vaginitis testing beyond clinical diagnosis. Of those with CPT codes for testing, 50.9% underwent traditional point-of-care testing, wet mount, or culture, 23.5% had a DNA probe, and 20.6% had NAAT testing.
Approximately one-third (34%) of women were cotested for CT/NG. Testing rates varied widely across the type of vaginitis test, from 70.8% of women who received NAAT to 22.8% of women with no CPT code. In multivariate analysis including age, region, and the Charlson Comorbidity Index (CCI), those tested with NAAT were eight times more likely to be cotested for CT/NG than those with no CPT code (odds ratio, 8.77; P < .0001).
Women who received a traditional test or DNA probe test for vaginitis also were more likely to have CT/NG testing than women with no CPT code, but only 1.8-2.5 times as likely.
“Our data suggest that most clinicians are not engaging the standard of care for testing and diagnosing vaginitis, or not engaging in comprehensive care by cotesting for vaginitis and CT/NG when patients may be at risk, resulting in missed opportunities for accurate diagnosis and potential associated coinfections,” the researchers wrote in their discussion. The higher rates for CT/NG testing among women receiving either NAAT or DNA probe vaginitis testing could be attributed to bundled testing, they noted, and the lower rate of CT/NG testing for patients with no CPT code could stem from limited access to microscopy or clinician preference for clinical diagnosis only, they said.
The findings were limited by several factors, including the lack of data on testing and diagnoses prior to the study period and not billed to insurance, and by the inability to account for variables including race, ethnicity, and socioeconomic status, the researchers noted.
However, the results highlight the need for more comprehensive care in vaginitis testing to take advantage of opportunities to identify CT or NG in women diagnosed with vaginitis, they concluded.
The study was supported by Becton, Dickinson and Company. Lead author Dr. Pinto disclosed consulting for Becton, Dickinson and Company, and receiving an honorarium from Roche.
Although the standard of care of diagnosing vaginitis is clinical evaluation, many practices do not perform accurate and comprehensive clinical examinations for a variety for reasons, and the Centers for Disease Control and Prevention currently recommends molecular testing, wrote Casey N. Pinto, PhD, of Penn State University, Hershey, and colleagues. The CDC also recommends testing women with vaginitis for Chlamydia trachomatis (CT) and Neissaria gonorrhoeae (NG) given the high rate of coinfections between vaginitis and these sexually transmitted infections, but data on cotesting in clinical practice are limited, they said.
In a study published in Sexually Transmitted Diseases, the researchers reviewed data from a commercial administrative claims database for 1,359,289 women aged 18-50 years who were diagnosed with vaginitis between 2012 and 2017.
The women were categorized into groups based on type of vaginitis diagnosis: nucleic amplification test (NAAT), DNA probe test, traditional lab test, and those diagnosed clinically at an index visit but with no CPT code for further testing.
Overall, nearly half of the women (49.2%) had no CPT code for further vaginitis testing beyond clinical diagnosis. Of those with CPT codes for testing, 50.9% underwent traditional point-of-care testing, wet mount, or culture, 23.5% had a DNA probe, and 20.6% had NAAT testing.
Approximately one-third (34%) of women were cotested for CT/NG. Testing rates varied widely across the type of vaginitis test, from 70.8% of women who received NAAT to 22.8% of women with no CPT code. In multivariate analysis including age, region, and the Charlson Comorbidity Index (CCI), those tested with NAAT were eight times more likely to be cotested for CT/NG than those with no CPT code (odds ratio, 8.77; P < .0001).
Women who received a traditional test or DNA probe test for vaginitis also were more likely to have CT/NG testing than women with no CPT code, but only 1.8-2.5 times as likely.
“Our data suggest that most clinicians are not engaging the standard of care for testing and diagnosing vaginitis, or not engaging in comprehensive care by cotesting for vaginitis and CT/NG when patients may be at risk, resulting in missed opportunities for accurate diagnosis and potential associated coinfections,” the researchers wrote in their discussion. The higher rates for CT/NG testing among women receiving either NAAT or DNA probe vaginitis testing could be attributed to bundled testing, they noted, and the lower rate of CT/NG testing for patients with no CPT code could stem from limited access to microscopy or clinician preference for clinical diagnosis only, they said.
The findings were limited by several factors, including the lack of data on testing and diagnoses prior to the study period and not billed to insurance, and by the inability to account for variables including race, ethnicity, and socioeconomic status, the researchers noted.
However, the results highlight the need for more comprehensive care in vaginitis testing to take advantage of opportunities to identify CT or NG in women diagnosed with vaginitis, they concluded.
The study was supported by Becton, Dickinson and Company. Lead author Dr. Pinto disclosed consulting for Becton, Dickinson and Company, and receiving an honorarium from Roche.
Although the standard of care of diagnosing vaginitis is clinical evaluation, many practices do not perform accurate and comprehensive clinical examinations for a variety for reasons, and the Centers for Disease Control and Prevention currently recommends molecular testing, wrote Casey N. Pinto, PhD, of Penn State University, Hershey, and colleagues. The CDC also recommends testing women with vaginitis for Chlamydia trachomatis (CT) and Neissaria gonorrhoeae (NG) given the high rate of coinfections between vaginitis and these sexually transmitted infections, but data on cotesting in clinical practice are limited, they said.
In a study published in Sexually Transmitted Diseases, the researchers reviewed data from a commercial administrative claims database for 1,359,289 women aged 18-50 years who were diagnosed with vaginitis between 2012 and 2017.
The women were categorized into groups based on type of vaginitis diagnosis: nucleic amplification test (NAAT), DNA probe test, traditional lab test, and those diagnosed clinically at an index visit but with no CPT code for further testing.
Overall, nearly half of the women (49.2%) had no CPT code for further vaginitis testing beyond clinical diagnosis. Of those with CPT codes for testing, 50.9% underwent traditional point-of-care testing, wet mount, or culture, 23.5% had a DNA probe, and 20.6% had NAAT testing.
Approximately one-third (34%) of women were cotested for CT/NG. Testing rates varied widely across the type of vaginitis test, from 70.8% of women who received NAAT to 22.8% of women with no CPT code. In multivariate analysis including age, region, and the Charlson Comorbidity Index (CCI), those tested with NAAT were eight times more likely to be cotested for CT/NG than those with no CPT code (odds ratio, 8.77; P < .0001).
Women who received a traditional test or DNA probe test for vaginitis also were more likely to have CT/NG testing than women with no CPT code, but only 1.8-2.5 times as likely.
“Our data suggest that most clinicians are not engaging the standard of care for testing and diagnosing vaginitis, or not engaging in comprehensive care by cotesting for vaginitis and CT/NG when patients may be at risk, resulting in missed opportunities for accurate diagnosis and potential associated coinfections,” the researchers wrote in their discussion. The higher rates for CT/NG testing among women receiving either NAAT or DNA probe vaginitis testing could be attributed to bundled testing, they noted, and the lower rate of CT/NG testing for patients with no CPT code could stem from limited access to microscopy or clinician preference for clinical diagnosis only, they said.
The findings were limited by several factors, including the lack of data on testing and diagnoses prior to the study period and not billed to insurance, and by the inability to account for variables including race, ethnicity, and socioeconomic status, the researchers noted.
However, the results highlight the need for more comprehensive care in vaginitis testing to take advantage of opportunities to identify CT or NG in women diagnosed with vaginitis, they concluded.
The study was supported by Becton, Dickinson and Company. Lead author Dr. Pinto disclosed consulting for Becton, Dickinson and Company, and receiving an honorarium from Roche.
FROM SEXUALLY TRANSMITTED DISEASES
Underprescribed menopause relief: Women suffer needlessly
The result: Countless women grapple with the physical and emotional toll of this life transition.
These shortcomings have led to an influx of doctors moving from traditional practice to virtual startups that focus on women’s health issues, treating patients who come to them desperate and frustrated after years of unresolved issues.
The solution is often so simple it is almost maddening, specialists say: vaginal creams containing low-dose estrogen which can address the symptoms of menopause, from vaginal dryness to recurrent urinary tract infections.
“Hands down, this is one of the most meaningful interventions I’ve ever offered to a patient and yet it is underutilized,” said Ashley Winter, MD, chief medical officer and urologist at Odela Health, a digital women’s health clinic. “A lot of companies are blossoming in this menopause space because it is underserved by traditional health care – your gynecologist typically deals with reproduction, and typically when women are done with child-bearing, they’re kind of discharged from the care of their gynecologist.”
More than 1 million women in the United States go through menopause each year. According to a 2022 survey, 4 in 10 women report menopause symptoms that have been disruptive enough to interfere with their work performance on at least a weekly basis.
And yet, many women are not getting appropriate treatment.
Partially to blame is the harmful legacy of faulty data, doctors say. The early results of the federally funded Women’s Health Initiative, released in 2002, showed that hormone therapy (HT) led to increased risk for heart attacks, strokes, and breast cancer. But further analysis showed the opposite: Hormonal therapies have a helpful effect on cardiovascular and bone health and generally reduce risk of death in younger women or those in the early postmenopausal period.
Hormone therapy delivers estrogen, sometimes with progesterone, to the body through gels, creams, patches, pills, suppositories, or a device fitted inside the uterus. Systemic HT sends hormones into the bloodstream, while local HT – like vaginal estrogen cream – specifically treats vaginal symptoms of menopause.
Myths about the health risks linked to systemic and topical HT have long been debunked, and research on topical HT in particular shows it poses no risk for cancer or other chronic diseases.
Yet while 2 decades have passed since the misinformation first started to spread, people remain woefully uninformed about hormone treatments.
The FDA still requires that estrogen products carry a black-box warning on the early data, even though it has since been proven false.
“This is one of the most damaging PR misadventures of modern medicine in my opinion,” Dr. Winter said. “It has literally killed women, and it’s made them miserable.”
The public has a glaring lack of knowledge about menopause management, said Stephanie Faubion, MD, medical director for the North American Menopause Society and director of Mayo Clinic’s Center for Women’s Health.
Treating with low-dose estrogen isn’t a radical approach – in fact, it is the standard of care for women experiencing many menopause symptoms, Dr. Faubion said. But the topic does have nuance, and some people get lost in the specifics.
“I don’t think there’s a lot of knowledge on the risk-benefits of hormone therapy in general,” Dr. Faubion said. “New information comes out so frequently it’s difficult to keep track of. The answer is complicated and depends on dose, duration of treatment, what formulation you’re on. It’s difficult for a lot of people to understand.”
But Dr. Winter said the lack of public knowledge reflects a bigger problem: Knowledge gaps exist among doctors, too, stemming from insufficient training on menopause-related issues.
During her 6-year urology residency, she never learned the role of vaginal estrogen on urinary problems, Dr. Winter said. Only during a 1-year fellowship on sexual dysfunction did she hear about the treatment.
“Despite dealing with urinary issues, incontinence, blood in the urine – training to manage all those concerns – the role of local hormones in the vagina for managing all them was never taught, never discussed,” Dr. Winter said. “I never prescribed any of it.”
A year ago, Dr. Winter left her job at Kaiser Permanente to join Odela. After years of prescribing medications for overactive bladder with little to no results, she said, she now uses the knowledge she gained during her fellowship by helping women who have spent years battling debilitating symptoms.
Urologists are not the only clinicians who lack appropriate training. Obstetrics and gynecology residencies offer little knowledge on menopause treatments, said Ghazaleh Moayedi, DO, an ob.gyn. and complex family planning specialist for Texas-based Pegasus Health Justice Center.
The problem is partly a systems-based one, she said. Training programs often direct patients who are uninsured, or covered through public insurance, to medical residents. Patients who qualify for Medicaid or Medicare are often either pregnant or over 65, Dr. Moayedi said, so women actively going through the transition can slip through the cracks.
“What that means in a state like Texas where I’m based, where it is difficult to qualify for Medicaid, is that the people we see who do qualify are pregnant,” she said. “And you’re not on Medicare until you’re 65. So most ob.gyn. residents don’t graduate with expansive experience in menopause.”
According to Medicaid.gov, 80% of the national population covered by Medicaid is age 45 and younger.
When doctors have proper training and prescribe local hormones, patients don’t always follow the treatment plan, said Andrea Rapkin, MD, professor of obstetrics and gynecology at David Geffen School of Medicine at UCLA.
That failure to follow treatment is yet another example of remaining doubts from the misinformation spread through early research, Dr. Rapkin said.
“I’ll prescribe an estrogen product, and I’ll find out they didn’t take it even though I’ll reassure them,” she said. “I do think there are some lingering concerns, but I’m glad to see there is a growing interest in vaginal hormones.”
A version of this article first appeared on WebMD.com.
The result: Countless women grapple with the physical and emotional toll of this life transition.
These shortcomings have led to an influx of doctors moving from traditional practice to virtual startups that focus on women’s health issues, treating patients who come to them desperate and frustrated after years of unresolved issues.
The solution is often so simple it is almost maddening, specialists say: vaginal creams containing low-dose estrogen which can address the symptoms of menopause, from vaginal dryness to recurrent urinary tract infections.
“Hands down, this is one of the most meaningful interventions I’ve ever offered to a patient and yet it is underutilized,” said Ashley Winter, MD, chief medical officer and urologist at Odela Health, a digital women’s health clinic. “A lot of companies are blossoming in this menopause space because it is underserved by traditional health care – your gynecologist typically deals with reproduction, and typically when women are done with child-bearing, they’re kind of discharged from the care of their gynecologist.”
More than 1 million women in the United States go through menopause each year. According to a 2022 survey, 4 in 10 women report menopause symptoms that have been disruptive enough to interfere with their work performance on at least a weekly basis.
And yet, many women are not getting appropriate treatment.
Partially to blame is the harmful legacy of faulty data, doctors say. The early results of the federally funded Women’s Health Initiative, released in 2002, showed that hormone therapy (HT) led to increased risk for heart attacks, strokes, and breast cancer. But further analysis showed the opposite: Hormonal therapies have a helpful effect on cardiovascular and bone health and generally reduce risk of death in younger women or those in the early postmenopausal period.
Hormone therapy delivers estrogen, sometimes with progesterone, to the body through gels, creams, patches, pills, suppositories, or a device fitted inside the uterus. Systemic HT sends hormones into the bloodstream, while local HT – like vaginal estrogen cream – specifically treats vaginal symptoms of menopause.
Myths about the health risks linked to systemic and topical HT have long been debunked, and research on topical HT in particular shows it poses no risk for cancer or other chronic diseases.
Yet while 2 decades have passed since the misinformation first started to spread, people remain woefully uninformed about hormone treatments.
The FDA still requires that estrogen products carry a black-box warning on the early data, even though it has since been proven false.
“This is one of the most damaging PR misadventures of modern medicine in my opinion,” Dr. Winter said. “It has literally killed women, and it’s made them miserable.”
The public has a glaring lack of knowledge about menopause management, said Stephanie Faubion, MD, medical director for the North American Menopause Society and director of Mayo Clinic’s Center for Women’s Health.
Treating with low-dose estrogen isn’t a radical approach – in fact, it is the standard of care for women experiencing many menopause symptoms, Dr. Faubion said. But the topic does have nuance, and some people get lost in the specifics.
“I don’t think there’s a lot of knowledge on the risk-benefits of hormone therapy in general,” Dr. Faubion said. “New information comes out so frequently it’s difficult to keep track of. The answer is complicated and depends on dose, duration of treatment, what formulation you’re on. It’s difficult for a lot of people to understand.”
But Dr. Winter said the lack of public knowledge reflects a bigger problem: Knowledge gaps exist among doctors, too, stemming from insufficient training on menopause-related issues.
During her 6-year urology residency, she never learned the role of vaginal estrogen on urinary problems, Dr. Winter said. Only during a 1-year fellowship on sexual dysfunction did she hear about the treatment.
“Despite dealing with urinary issues, incontinence, blood in the urine – training to manage all those concerns – the role of local hormones in the vagina for managing all them was never taught, never discussed,” Dr. Winter said. “I never prescribed any of it.”
A year ago, Dr. Winter left her job at Kaiser Permanente to join Odela. After years of prescribing medications for overactive bladder with little to no results, she said, she now uses the knowledge she gained during her fellowship by helping women who have spent years battling debilitating symptoms.
Urologists are not the only clinicians who lack appropriate training. Obstetrics and gynecology residencies offer little knowledge on menopause treatments, said Ghazaleh Moayedi, DO, an ob.gyn. and complex family planning specialist for Texas-based Pegasus Health Justice Center.
The problem is partly a systems-based one, she said. Training programs often direct patients who are uninsured, or covered through public insurance, to medical residents. Patients who qualify for Medicaid or Medicare are often either pregnant or over 65, Dr. Moayedi said, so women actively going through the transition can slip through the cracks.
“What that means in a state like Texas where I’m based, where it is difficult to qualify for Medicaid, is that the people we see who do qualify are pregnant,” she said. “And you’re not on Medicare until you’re 65. So most ob.gyn. residents don’t graduate with expansive experience in menopause.”
According to Medicaid.gov, 80% of the national population covered by Medicaid is age 45 and younger.
When doctors have proper training and prescribe local hormones, patients don’t always follow the treatment plan, said Andrea Rapkin, MD, professor of obstetrics and gynecology at David Geffen School of Medicine at UCLA.
That failure to follow treatment is yet another example of remaining doubts from the misinformation spread through early research, Dr. Rapkin said.
“I’ll prescribe an estrogen product, and I’ll find out they didn’t take it even though I’ll reassure them,” she said. “I do think there are some lingering concerns, but I’m glad to see there is a growing interest in vaginal hormones.”
A version of this article first appeared on WebMD.com.
The result: Countless women grapple with the physical and emotional toll of this life transition.
These shortcomings have led to an influx of doctors moving from traditional practice to virtual startups that focus on women’s health issues, treating patients who come to them desperate and frustrated after years of unresolved issues.
The solution is often so simple it is almost maddening, specialists say: vaginal creams containing low-dose estrogen which can address the symptoms of menopause, from vaginal dryness to recurrent urinary tract infections.
“Hands down, this is one of the most meaningful interventions I’ve ever offered to a patient and yet it is underutilized,” said Ashley Winter, MD, chief medical officer and urologist at Odela Health, a digital women’s health clinic. “A lot of companies are blossoming in this menopause space because it is underserved by traditional health care – your gynecologist typically deals with reproduction, and typically when women are done with child-bearing, they’re kind of discharged from the care of their gynecologist.”
More than 1 million women in the United States go through menopause each year. According to a 2022 survey, 4 in 10 women report menopause symptoms that have been disruptive enough to interfere with their work performance on at least a weekly basis.
And yet, many women are not getting appropriate treatment.
Partially to blame is the harmful legacy of faulty data, doctors say. The early results of the federally funded Women’s Health Initiative, released in 2002, showed that hormone therapy (HT) led to increased risk for heart attacks, strokes, and breast cancer. But further analysis showed the opposite: Hormonal therapies have a helpful effect on cardiovascular and bone health and generally reduce risk of death in younger women or those in the early postmenopausal period.
Hormone therapy delivers estrogen, sometimes with progesterone, to the body through gels, creams, patches, pills, suppositories, or a device fitted inside the uterus. Systemic HT sends hormones into the bloodstream, while local HT – like vaginal estrogen cream – specifically treats vaginal symptoms of menopause.
Myths about the health risks linked to systemic and topical HT have long been debunked, and research on topical HT in particular shows it poses no risk for cancer or other chronic diseases.
Yet while 2 decades have passed since the misinformation first started to spread, people remain woefully uninformed about hormone treatments.
The FDA still requires that estrogen products carry a black-box warning on the early data, even though it has since been proven false.
“This is one of the most damaging PR misadventures of modern medicine in my opinion,” Dr. Winter said. “It has literally killed women, and it’s made them miserable.”
The public has a glaring lack of knowledge about menopause management, said Stephanie Faubion, MD, medical director for the North American Menopause Society and director of Mayo Clinic’s Center for Women’s Health.
Treating with low-dose estrogen isn’t a radical approach – in fact, it is the standard of care for women experiencing many menopause symptoms, Dr. Faubion said. But the topic does have nuance, and some people get lost in the specifics.
“I don’t think there’s a lot of knowledge on the risk-benefits of hormone therapy in general,” Dr. Faubion said. “New information comes out so frequently it’s difficult to keep track of. The answer is complicated and depends on dose, duration of treatment, what formulation you’re on. It’s difficult for a lot of people to understand.”
But Dr. Winter said the lack of public knowledge reflects a bigger problem: Knowledge gaps exist among doctors, too, stemming from insufficient training on menopause-related issues.
During her 6-year urology residency, she never learned the role of vaginal estrogen on urinary problems, Dr. Winter said. Only during a 1-year fellowship on sexual dysfunction did she hear about the treatment.
“Despite dealing with urinary issues, incontinence, blood in the urine – training to manage all those concerns – the role of local hormones in the vagina for managing all them was never taught, never discussed,” Dr. Winter said. “I never prescribed any of it.”
A year ago, Dr. Winter left her job at Kaiser Permanente to join Odela. After years of prescribing medications for overactive bladder with little to no results, she said, she now uses the knowledge she gained during her fellowship by helping women who have spent years battling debilitating symptoms.
Urologists are not the only clinicians who lack appropriate training. Obstetrics and gynecology residencies offer little knowledge on menopause treatments, said Ghazaleh Moayedi, DO, an ob.gyn. and complex family planning specialist for Texas-based Pegasus Health Justice Center.
The problem is partly a systems-based one, she said. Training programs often direct patients who are uninsured, or covered through public insurance, to medical residents. Patients who qualify for Medicaid or Medicare are often either pregnant or over 65, Dr. Moayedi said, so women actively going through the transition can slip through the cracks.
“What that means in a state like Texas where I’m based, where it is difficult to qualify for Medicaid, is that the people we see who do qualify are pregnant,” she said. “And you’re not on Medicare until you’re 65. So most ob.gyn. residents don’t graduate with expansive experience in menopause.”
According to Medicaid.gov, 80% of the national population covered by Medicaid is age 45 and younger.
When doctors have proper training and prescribe local hormones, patients don’t always follow the treatment plan, said Andrea Rapkin, MD, professor of obstetrics and gynecology at David Geffen School of Medicine at UCLA.
That failure to follow treatment is yet another example of remaining doubts from the misinformation spread through early research, Dr. Rapkin said.
“I’ll prescribe an estrogen product, and I’ll find out they didn’t take it even though I’ll reassure them,” she said. “I do think there are some lingering concerns, but I’m glad to see there is a growing interest in vaginal hormones.”
A version of this article first appeared on WebMD.com.
Mammography breast density reporting: What it means for clinicians
This transcript has been edited for clarity.
Today, I’m going to talk about the 2023 Food and Drug Administration regulation that requires breast density to be reported on all mammogram results nationwide, and for that report to go to both clinicians and patients. Previously this was the rule in some states, but not in others. This is important because 40%-50% of women have dense breasts. I’m going to discuss what that means for you, and for our patients.
First
Breast density describes the appearance of the breast on mammography. Appearance varies on the basis of breast tissue composition, with fibroglandular tissue being more dense than fatty tissue. Breast density is important because it relates to both the risk for cancer and the ability of mammography to detect cancer.
Breast density is defined and classified according to the American College of Radiology’s BI-RADS four-category scale. Categories 1 and 2 refer to breast tissue that is not dense, accounting for about 50% of the population. Categories 3 and 4 describe heterogeneously dense and extremely dense breast tissue, which occur in approximately 40% and 50% of women, respectively. When speaking about dense breast tissue readings on mammography, we are referring to categories 3 and 4.
Women with dense breast tissue have an increased risk of developing breast cancer and are less likely to have early breast cancer detected on mammography.
Let’s go over the details by category:
For women in categories 1 and 2 (considered not dense breast tissue), the sensitivity of mammography for detecting early breast cancer is 80%-90%. In categories 3 and 4, the sensitivity of mammography drops to 60%-70%.
Compared with women with average breast density, the risk of developing breast cancer is 20% higher in women with BI-RADS category 3 breasts, and more than twice as high (relative risk, 2.1) in those with BI-RADS category 4 breasts. Thus, the risk of developing breast cancer is higher, but the sensitivity of the test is lower.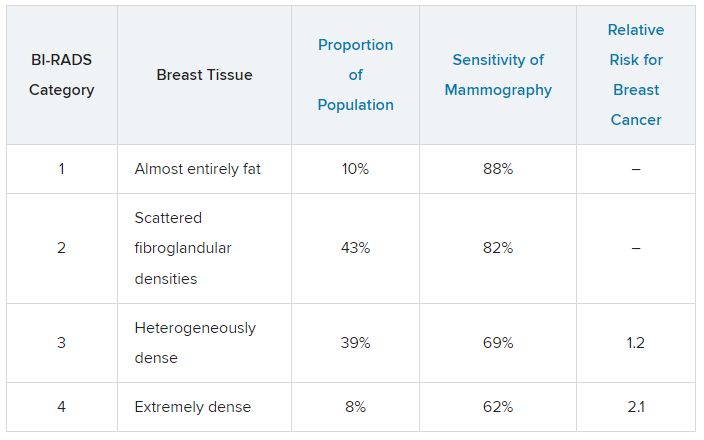
The clinical question is, what should we do about this? For women who have a normal mammogram with dense breasts, should follow-up testing be done, and if so, what test? The main follow-up testing options are either ultrasound or MRI, usually ultrasound. Additional testing will detect additional cancers that were not picked up on the initial mammogram and will also lead to additional biopsies for false-positive tests from the additional testing.
An American College of Gynecology and Obstetrics practice advisory nicely summarizes the evidence and clarifies that this decision is made in the context of a lack of published evidence demonstrating improved outcomes, specifically no reduction in breast cancer mortality, with supplemental testing. The official ACOG stance is that they “do not recommend routine use of alternative or adjunctive tests to screening mammography in women with dense breasts who are asymptomatic and have no additional risk factors.”
This is an area where it is important to understand the data. We are all going to be getting test results back that indicate level of breast density, and those test results will also be sent to our patients, so we are going to be asked about this by interested patients. Should this be something that we talk to patients about, utilizing shared decision-making to decide about whether follow-up testing is necessary in women with dense breasts? That is something each clinician will need to decide, and knowing the data is a critically important step in that decision.
Neil Skolnik, MD, is a professor, department of family medicine, at Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, and associate director, department of family medicine, Abington (Pennsylvania) Jefferson Health.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Today, I’m going to talk about the 2023 Food and Drug Administration regulation that requires breast density to be reported on all mammogram results nationwide, and for that report to go to both clinicians and patients. Previously this was the rule in some states, but not in others. This is important because 40%-50% of women have dense breasts. I’m going to discuss what that means for you, and for our patients.
First
Breast density describes the appearance of the breast on mammography. Appearance varies on the basis of breast tissue composition, with fibroglandular tissue being more dense than fatty tissue. Breast density is important because it relates to both the risk for cancer and the ability of mammography to detect cancer.
Breast density is defined and classified according to the American College of Radiology’s BI-RADS four-category scale. Categories 1 and 2 refer to breast tissue that is not dense, accounting for about 50% of the population. Categories 3 and 4 describe heterogeneously dense and extremely dense breast tissue, which occur in approximately 40% and 50% of women, respectively. When speaking about dense breast tissue readings on mammography, we are referring to categories 3 and 4.
Women with dense breast tissue have an increased risk of developing breast cancer and are less likely to have early breast cancer detected on mammography.
Let’s go over the details by category:
For women in categories 1 and 2 (considered not dense breast tissue), the sensitivity of mammography for detecting early breast cancer is 80%-90%. In categories 3 and 4, the sensitivity of mammography drops to 60%-70%.
Compared with women with average breast density, the risk of developing breast cancer is 20% higher in women with BI-RADS category 3 breasts, and more than twice as high (relative risk, 2.1) in those with BI-RADS category 4 breasts. Thus, the risk of developing breast cancer is higher, but the sensitivity of the test is lower.
The clinical question is, what should we do about this? For women who have a normal mammogram with dense breasts, should follow-up testing be done, and if so, what test? The main follow-up testing options are either ultrasound or MRI, usually ultrasound. Additional testing will detect additional cancers that were not picked up on the initial mammogram and will also lead to additional biopsies for false-positive tests from the additional testing.
An American College of Gynecology and Obstetrics practice advisory nicely summarizes the evidence and clarifies that this decision is made in the context of a lack of published evidence demonstrating improved outcomes, specifically no reduction in breast cancer mortality, with supplemental testing. The official ACOG stance is that they “do not recommend routine use of alternative or adjunctive tests to screening mammography in women with dense breasts who are asymptomatic and have no additional risk factors.”
This is an area where it is important to understand the data. We are all going to be getting test results back that indicate level of breast density, and those test results will also be sent to our patients, so we are going to be asked about this by interested patients. Should this be something that we talk to patients about, utilizing shared decision-making to decide about whether follow-up testing is necessary in women with dense breasts? That is something each clinician will need to decide, and knowing the data is a critically important step in that decision.
Neil Skolnik, MD, is a professor, department of family medicine, at Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, and associate director, department of family medicine, Abington (Pennsylvania) Jefferson Health.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Today, I’m going to talk about the 2023 Food and Drug Administration regulation that requires breast density to be reported on all mammogram results nationwide, and for that report to go to both clinicians and patients. Previously this was the rule in some states, but not in others. This is important because 40%-50% of women have dense breasts. I’m going to discuss what that means for you, and for our patients.
First
Breast density describes the appearance of the breast on mammography. Appearance varies on the basis of breast tissue composition, with fibroglandular tissue being more dense than fatty tissue. Breast density is important because it relates to both the risk for cancer and the ability of mammography to detect cancer.
Breast density is defined and classified according to the American College of Radiology’s BI-RADS four-category scale. Categories 1 and 2 refer to breast tissue that is not dense, accounting for about 50% of the population. Categories 3 and 4 describe heterogeneously dense and extremely dense breast tissue, which occur in approximately 40% and 50% of women, respectively. When speaking about dense breast tissue readings on mammography, we are referring to categories 3 and 4.
Women with dense breast tissue have an increased risk of developing breast cancer and are less likely to have early breast cancer detected on mammography.
Let’s go over the details by category:
For women in categories 1 and 2 (considered not dense breast tissue), the sensitivity of mammography for detecting early breast cancer is 80%-90%. In categories 3 and 4, the sensitivity of mammography drops to 60%-70%.
Compared with women with average breast density, the risk of developing breast cancer is 20% higher in women with BI-RADS category 3 breasts, and more than twice as high (relative risk, 2.1) in those with BI-RADS category 4 breasts. Thus, the risk of developing breast cancer is higher, but the sensitivity of the test is lower.
The clinical question is, what should we do about this? For women who have a normal mammogram with dense breasts, should follow-up testing be done, and if so, what test? The main follow-up testing options are either ultrasound or MRI, usually ultrasound. Additional testing will detect additional cancers that were not picked up on the initial mammogram and will also lead to additional biopsies for false-positive tests from the additional testing.
An American College of Gynecology and Obstetrics practice advisory nicely summarizes the evidence and clarifies that this decision is made in the context of a lack of published evidence demonstrating improved outcomes, specifically no reduction in breast cancer mortality, with supplemental testing. The official ACOG stance is that they “do not recommend routine use of alternative or adjunctive tests to screening mammography in women with dense breasts who are asymptomatic and have no additional risk factors.”
This is an area where it is important to understand the data. We are all going to be getting test results back that indicate level of breast density, and those test results will also be sent to our patients, so we are going to be asked about this by interested patients. Should this be something that we talk to patients about, utilizing shared decision-making to decide about whether follow-up testing is necessary in women with dense breasts? That is something each clinician will need to decide, and knowing the data is a critically important step in that decision.
Neil Skolnik, MD, is a professor, department of family medicine, at Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, and associate director, department of family medicine, Abington (Pennsylvania) Jefferson Health.
A version of this article first appeared on Medscape.com.
Weight loss linked to mortality risk in older women
Weight loss of at least 5% over a 3-year period was associated with significantly increased mortality in women at age 90, 95, and 100 years compared with those whose weight remained stable, based on data from more than 50,000 individuals.
Previous studies of later-life weight changes and mortality have yielded inconsistent results driven by considerations of weight loss intentionality, and data on older adults in particular are limited, wrote Aladdin H. Shadyab, PhD, of the University of California, San Diego, and colleagues.
In a study published in the Journals of Gerontology: Medical Sciences, the researchers reviewed data from the Women’s Health Initiative, a prospective study of factors affecting chronic disease development in postmenopausal women. The study population included 54,437 women who entered the WHI between 1993 and 1998 at ages 50-79 years. The mean baseline age was 69.8 years; 89.5% of the participants were White, 5.7% were Black, 2.7% were Asian, 2.5% were Hispanic/Latino, and the remaining 1.0% were multiracial, American Indian/Alaskan Native, Native Hawaiian/Other Pacific Islander, or unknown.
The primary outcomes were the associations of short-term (3-year) and long-term (10-year) weight changes with survival to ages 90, 95, and 100 years.
A total of 30,647 women survived to at least 90 years (56.3%).
Overall, women with a short-term weight loss of 5% or more of body weight were 33% less likely to survive to age 90 years, 35% less likely to survive to age 95 years, and 38% less likely to survive to age 100 years than were those whose weight remained stable (odds ratios, 0.67, 0.65, and 0.62, respectively).
The associations were stronger in cases of unintentional short-term weight loss. Intentional weight loss from baseline to year 3 was associated with 17% lower odds of survival to age 90 compared to stable weight (OR, 0.83), but unintentional weight loss was associated with 51% lower odds of survival to age 90 (OR, 0.49).
Similarly, women with 10-year weight loss of at least 5% were 40% less likely to survive to 90 years and 49% less likely to survive to 95 years (OR, 0.60 and OR, 0.51, respectively). The sample size was too small to assess the relation of 10-year weight loss with survival to 100 years, and intentionality was not assessed for 10-year weight changes.
By contrast, weight gain of at least 5% had no significant effect on survival to ages 90, 95, or 100 years, but stable weight over time increased the odds of living to ages 90 to 100 years by 1.2-fold to 2-fold compared to either intentional or unintentional weight loss of at least 5%.
The trends in results were similar across body weight categories (normal weight, overweight, and obese as defined by body mass index). Baseline age and smoking status had no significant effect on the results.
Some of the proportion of self-reported intentional weight loss in the study population may have been unintentional, the researchers wrote in their discussion.
“It is important to note that perceived intentionality of weight loss may be influenced by the many societal pressures to lose weight, especially among women, and therefore overestimate the behavioral changes underlying experienced weight loss in older adults,” they said.
The findings were limited by several factors including the potential for inaccurate self-reported weight loss intention, and the likelihood that the mean older age of the population at baseline (older than 60 years) meant that they were more likely to live longer regardless of weight changes, the researchers noted. Other limitations included the primarily White study population, and other residual confounding factors such as ill health that might drive weight loss, the researchers noted.
However, the results were strengthened by the large sample size and long follow-up period, and suggest that “blanket recommendations for weight loss in older women are unlikely to lead to better survival at advanced ages,” they concluded.
Data support weight monitoring
The investigators acknowledged that their data do not affect clinical recommendations for moderate weight loss in older women to improve health outcomes, especially in those with overweight or obesity, but instead “support close monitoring of the amount and speed of weight loss, particularly when unintentional, as an indicator of underlying poor health and predictor of decreased lifespan in older women.”
Neil Skolnik, MD, professor of family and community medicine at the Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, agreed with this conclusion. The current study suggests that when older women lose a significant amount of weight unintentionally, it could be a sign of failing health, he said.
Weight gain or loss in old age is very different from weight issues in younger people, where clinicians may be encouraging weight loss to improve health outcomes, Dr. Skolnik said in an interview.
A key take-home message for clinicians, in addition to monitoring weight in older patients, is to emphasize nutrition for individuals in their 80s, 90s, and beyond, he said.
The study was supported by the National Heart, Lung, and Blood Institute. Dr. Shadyab had no financial conflicts to disclose. Dr. Skolnik had no financial conflicts to disclose and serves on the editorial advisory board of Family Practice News.
Weight loss of at least 5% over a 3-year period was associated with significantly increased mortality in women at age 90, 95, and 100 years compared with those whose weight remained stable, based on data from more than 50,000 individuals.
Previous studies of later-life weight changes and mortality have yielded inconsistent results driven by considerations of weight loss intentionality, and data on older adults in particular are limited, wrote Aladdin H. Shadyab, PhD, of the University of California, San Diego, and colleagues.
In a study published in the Journals of Gerontology: Medical Sciences, the researchers reviewed data from the Women’s Health Initiative, a prospective study of factors affecting chronic disease development in postmenopausal women. The study population included 54,437 women who entered the WHI between 1993 and 1998 at ages 50-79 years. The mean baseline age was 69.8 years; 89.5% of the participants were White, 5.7% were Black, 2.7% were Asian, 2.5% were Hispanic/Latino, and the remaining 1.0% were multiracial, American Indian/Alaskan Native, Native Hawaiian/Other Pacific Islander, or unknown.
The primary outcomes were the associations of short-term (3-year) and long-term (10-year) weight changes with survival to ages 90, 95, and 100 years.
A total of 30,647 women survived to at least 90 years (56.3%).
Overall, women with a short-term weight loss of 5% or more of body weight were 33% less likely to survive to age 90 years, 35% less likely to survive to age 95 years, and 38% less likely to survive to age 100 years than were those whose weight remained stable (odds ratios, 0.67, 0.65, and 0.62, respectively).
The associations were stronger in cases of unintentional short-term weight loss. Intentional weight loss from baseline to year 3 was associated with 17% lower odds of survival to age 90 compared to stable weight (OR, 0.83), but unintentional weight loss was associated with 51% lower odds of survival to age 90 (OR, 0.49).
Similarly, women with 10-year weight loss of at least 5% were 40% less likely to survive to 90 years and 49% less likely to survive to 95 years (OR, 0.60 and OR, 0.51, respectively). The sample size was too small to assess the relation of 10-year weight loss with survival to 100 years, and intentionality was not assessed for 10-year weight changes.
By contrast, weight gain of at least 5% had no significant effect on survival to ages 90, 95, or 100 years, but stable weight over time increased the odds of living to ages 90 to 100 years by 1.2-fold to 2-fold compared to either intentional or unintentional weight loss of at least 5%.
The trends in results were similar across body weight categories (normal weight, overweight, and obese as defined by body mass index). Baseline age and smoking status had no significant effect on the results.
Some of the proportion of self-reported intentional weight loss in the study population may have been unintentional, the researchers wrote in their discussion.
“It is important to note that perceived intentionality of weight loss may be influenced by the many societal pressures to lose weight, especially among women, and therefore overestimate the behavioral changes underlying experienced weight loss in older adults,” they said.
The findings were limited by several factors including the potential for inaccurate self-reported weight loss intention, and the likelihood that the mean older age of the population at baseline (older than 60 years) meant that they were more likely to live longer regardless of weight changes, the researchers noted. Other limitations included the primarily White study population, and other residual confounding factors such as ill health that might drive weight loss, the researchers noted.
However, the results were strengthened by the large sample size and long follow-up period, and suggest that “blanket recommendations for weight loss in older women are unlikely to lead to better survival at advanced ages,” they concluded.
Data support weight monitoring
The investigators acknowledged that their data do not affect clinical recommendations for moderate weight loss in older women to improve health outcomes, especially in those with overweight or obesity, but instead “support close monitoring of the amount and speed of weight loss, particularly when unintentional, as an indicator of underlying poor health and predictor of decreased lifespan in older women.”
Neil Skolnik, MD, professor of family and community medicine at the Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, agreed with this conclusion. The current study suggests that when older women lose a significant amount of weight unintentionally, it could be a sign of failing health, he said.
Weight gain or loss in old age is very different from weight issues in younger people, where clinicians may be encouraging weight loss to improve health outcomes, Dr. Skolnik said in an interview.
A key take-home message for clinicians, in addition to monitoring weight in older patients, is to emphasize nutrition for individuals in their 80s, 90s, and beyond, he said.
The study was supported by the National Heart, Lung, and Blood Institute. Dr. Shadyab had no financial conflicts to disclose. Dr. Skolnik had no financial conflicts to disclose and serves on the editorial advisory board of Family Practice News.
Weight loss of at least 5% over a 3-year period was associated with significantly increased mortality in women at age 90, 95, and 100 years compared with those whose weight remained stable, based on data from more than 50,000 individuals.
Previous studies of later-life weight changes and mortality have yielded inconsistent results driven by considerations of weight loss intentionality, and data on older adults in particular are limited, wrote Aladdin H. Shadyab, PhD, of the University of California, San Diego, and colleagues.
In a study published in the Journals of Gerontology: Medical Sciences, the researchers reviewed data from the Women’s Health Initiative, a prospective study of factors affecting chronic disease development in postmenopausal women. The study population included 54,437 women who entered the WHI between 1993 and 1998 at ages 50-79 years. The mean baseline age was 69.8 years; 89.5% of the participants were White, 5.7% were Black, 2.7% were Asian, 2.5% were Hispanic/Latino, and the remaining 1.0% were multiracial, American Indian/Alaskan Native, Native Hawaiian/Other Pacific Islander, or unknown.
The primary outcomes were the associations of short-term (3-year) and long-term (10-year) weight changes with survival to ages 90, 95, and 100 years.
A total of 30,647 women survived to at least 90 years (56.3%).
Overall, women with a short-term weight loss of 5% or more of body weight were 33% less likely to survive to age 90 years, 35% less likely to survive to age 95 years, and 38% less likely to survive to age 100 years than were those whose weight remained stable (odds ratios, 0.67, 0.65, and 0.62, respectively).
The associations were stronger in cases of unintentional short-term weight loss. Intentional weight loss from baseline to year 3 was associated with 17% lower odds of survival to age 90 compared to stable weight (OR, 0.83), but unintentional weight loss was associated with 51% lower odds of survival to age 90 (OR, 0.49).
Similarly, women with 10-year weight loss of at least 5% were 40% less likely to survive to 90 years and 49% less likely to survive to 95 years (OR, 0.60 and OR, 0.51, respectively). The sample size was too small to assess the relation of 10-year weight loss with survival to 100 years, and intentionality was not assessed for 10-year weight changes.
By contrast, weight gain of at least 5% had no significant effect on survival to ages 90, 95, or 100 years, but stable weight over time increased the odds of living to ages 90 to 100 years by 1.2-fold to 2-fold compared to either intentional or unintentional weight loss of at least 5%.
The trends in results were similar across body weight categories (normal weight, overweight, and obese as defined by body mass index). Baseline age and smoking status had no significant effect on the results.
Some of the proportion of self-reported intentional weight loss in the study population may have been unintentional, the researchers wrote in their discussion.
“It is important to note that perceived intentionality of weight loss may be influenced by the many societal pressures to lose weight, especially among women, and therefore overestimate the behavioral changes underlying experienced weight loss in older adults,” they said.
The findings were limited by several factors including the potential for inaccurate self-reported weight loss intention, and the likelihood that the mean older age of the population at baseline (older than 60 years) meant that they were more likely to live longer regardless of weight changes, the researchers noted. Other limitations included the primarily White study population, and other residual confounding factors such as ill health that might drive weight loss, the researchers noted.
However, the results were strengthened by the large sample size and long follow-up period, and suggest that “blanket recommendations for weight loss in older women are unlikely to lead to better survival at advanced ages,” they concluded.
Data support weight monitoring
The investigators acknowledged that their data do not affect clinical recommendations for moderate weight loss in older women to improve health outcomes, especially in those with overweight or obesity, but instead “support close monitoring of the amount and speed of weight loss, particularly when unintentional, as an indicator of underlying poor health and predictor of decreased lifespan in older women.”
Neil Skolnik, MD, professor of family and community medicine at the Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, agreed with this conclusion. The current study suggests that when older women lose a significant amount of weight unintentionally, it could be a sign of failing health, he said.
Weight gain or loss in old age is very different from weight issues in younger people, where clinicians may be encouraging weight loss to improve health outcomes, Dr. Skolnik said in an interview.
A key take-home message for clinicians, in addition to monitoring weight in older patients, is to emphasize nutrition for individuals in their 80s, 90s, and beyond, he said.
The study was supported by the National Heart, Lung, and Blood Institute. Dr. Shadyab had no financial conflicts to disclose. Dr. Skolnik had no financial conflicts to disclose and serves on the editorial advisory board of Family Practice News.
FROM THE JOURNALS OF GERONTOLOGY: MEDICAL SCIENCES
A nurse’s view: Blood test for severe preeclampsia will save lives
There is amazing news for the world of obstetrics and for all pregnant women. Severe preeclampsia is a critical obstetrical condition that can have serious outcomes for a mother and baby. It can lead to eclampsia, an obstetrical emergency, which often results in death of the mother and/or baby.
Based on research published in the Journal of the American Heart Association, the incidence of new‐onset hypertensive disorders of pregnancy (gestational hypertension and preeclampsia/eclampsia) have nearly doubled in the United States from 2007 to 2019. And they continue to climb.
According to the Preeclampsia Foundation, 5%-8% of all pregnancies in the United States will result in preeclampsia. Black women are at a 60% higher risk than white women, and according to various sources, other risk groups include those who became pregnant via in vitro fertilization, mothers of multiples (twins and triplets), women with gestational diabetes, women over age 35, women with chronic hypertension, obesity, polycystic ovary syndrome, sickle cell disease, rheumatoid arthritis, lupus, migraines, antiphospholipid syndrome, previous pregnancy with preeclampsia, family history, and scleroderma.
Screening and treatment
Preeclampsia is a multiorgan disease of pregnancy, and can be mild, but may quickly progress to severe, which can be life-threatening for mother and baby. It was previously referred to as toxemia or the high blood pressure disease of pregnancy. It primarily involves the cardiovascular, neurologic and renal systems, and the liver. Patients typically present with elevated blood pressures, but other symptoms may include headache, swelling of hands and feet, blurry/double vision or seeing spots, nausea/vomiting, and epigastric pain. It is diagnosed with elevated blood pressures, blood work, and protein in the urine.
Early screening for preeclampsia is done in the first trimester. Presently, a combination of prenatal blood work, blood pressure monitoring, and recognition of high-risk groups is used to determine a treatment plan going forward. The American Congress of Obstetricians and Gynecologists recommends women that fall into this group for potentially developing preeclampsia take daily aspirin as a preventative measure.
In its milder form, a pregnant woman can be observed as an outpatient – monitored with antepartum testing, lab work, and patient education to report significant symptoms as listed above. Teaching patients about fetal kick counts to monitor their baby’s movements is equally important. Women with mild preeclampsia usually can safely deliver at term, being induced between 37-39 weeks’ gestation.
On the other hand, if mild preeclampsia progresses to severe preeclampsia, delivery may be preterm for the safety of mother and baby. Severe preeclampsia can lead to maternal organ damage, seizures, and even death of mother and/or baby.
About 20% of women with severe preeclampsia will develop HELLP (Hemolysis, Elevated Liver enzymes, and Low Platelets) syndrome, a life-threatening disease that often warrants immediate delivery. According to the National Library of Medicine, the mortality rate of women with HELLP syndrome is up to 24% and the perinatal death rate is up as high as 37%. These serious conditions can cause ineffective maternal clotting, liver rupture, placental abruption, and postpartum hemorrhage. It is most prevalent in the third trimester but can occur within 48 hours of delivery.
The only cure for preeclampsia in any form is delivery.
Patients with severe preeclampsia are hospitalized until delivery – sometimes a few days to a couple of weeks. Mother and baby are closely watched for further progression, including signs of organ damage in the mother and changes to the well-being of the baby. If the mother’s health is severely compromised, then the baby will be compromised as well. A preterm delivery may be necessary.
Impact of the new test
The National Institute of Health states that preterm babies born from preeclamptic mothers can suffer many health problems including cerebral palsy, deafness, blindness, epilepsy, and a host of other respiratory, cardiovascular, and endocrine issues. But the biggest issue is preterm birth, defined as birth before 37 weeks gestation. Being born preterm can require a long stay in the intensive care nursery.
This is where the first-of-its-kind prognostic blood test comes into play. The test’s ability to predict severe preeclampsia within 2 weeks can help save lives. The test can offer health care providers the ability to administer steroids for fetal lung maturity before delivery and be more prepared to care for what could be a very compromised newborn.
The blood test, which is recommended between 23-35 weeks gestation, involves analyzing a ratio between two proteins from the placenta, sFlt1 and PIGF. The higher the ratio, the higher the risk that severe preeclampsia will develop. Results can be available within 30 minutes, which is critical when contemplating treatment.
An example of the use of this ratio is illustrated with chronic hypertension in pregnancy, which is defined as elevated blood pressure before 20 weeks or even before conception. Since chronic hypertension can be a primary precursor to preeclampsia, patients with this condition are at higher risk. The FDA-approved blood test would be helpful in determining the plan of care; that is, delivery versus hospitalization versus monitor as an outpatient.
With a positive test result, a pregnant woman can be immediately hospitalized where she can get the care she and baby need as they await delivery. Since health care providers already know the high-risk groups, surveillance can begin early, utilizing this blood test to predict the progression to severe preeclampsia. Conversely, if the test is negative, a treatment plan can be made as an outpatient and the pregnancy continues.
Not all hospitals are equipped to care for premature babies. If delivery is not imminent, providers can use this blood test to identify those that should be transferred to a tertiary center for observation and monitoring. Mother and baby would then not be separated after birth.
We really don’t know who will develop severe preeclampsia and who won’t. This new blood test will be a critical tool as pregnant patients go through their second and third trimesters. It will be especially pivotal for these women, but important for all pregnant women in reducing maternal and fetal mortality and morbidity.
Ms. Barnett is a registered nurse in the department of obstetrics, Mills-Peninsula Medical Center, Burlingame, Calif. She has disclosed no relevant financial relationships.
There is amazing news for the world of obstetrics and for all pregnant women. Severe preeclampsia is a critical obstetrical condition that can have serious outcomes for a mother and baby. It can lead to eclampsia, an obstetrical emergency, which often results in death of the mother and/or baby.
Based on research published in the Journal of the American Heart Association, the incidence of new‐onset hypertensive disorders of pregnancy (gestational hypertension and preeclampsia/eclampsia) have nearly doubled in the United States from 2007 to 2019. And they continue to climb.
According to the Preeclampsia Foundation, 5%-8% of all pregnancies in the United States will result in preeclampsia. Black women are at a 60% higher risk than white women, and according to various sources, other risk groups include those who became pregnant via in vitro fertilization, mothers of multiples (twins and triplets), women with gestational diabetes, women over age 35, women with chronic hypertension, obesity, polycystic ovary syndrome, sickle cell disease, rheumatoid arthritis, lupus, migraines, antiphospholipid syndrome, previous pregnancy with preeclampsia, family history, and scleroderma.
Screening and treatment
Preeclampsia is a multiorgan disease of pregnancy, and can be mild, but may quickly progress to severe, which can be life-threatening for mother and baby. It was previously referred to as toxemia or the high blood pressure disease of pregnancy. It primarily involves the cardiovascular, neurologic and renal systems, and the liver. Patients typically present with elevated blood pressures, but other symptoms may include headache, swelling of hands and feet, blurry/double vision or seeing spots, nausea/vomiting, and epigastric pain. It is diagnosed with elevated blood pressures, blood work, and protein in the urine.
Early screening for preeclampsia is done in the first trimester. Presently, a combination of prenatal blood work, blood pressure monitoring, and recognition of high-risk groups is used to determine a treatment plan going forward. The American Congress of Obstetricians and Gynecologists recommends women that fall into this group for potentially developing preeclampsia take daily aspirin as a preventative measure.
In its milder form, a pregnant woman can be observed as an outpatient – monitored with antepartum testing, lab work, and patient education to report significant symptoms as listed above. Teaching patients about fetal kick counts to monitor their baby’s movements is equally important. Women with mild preeclampsia usually can safely deliver at term, being induced between 37-39 weeks’ gestation.
On the other hand, if mild preeclampsia progresses to severe preeclampsia, delivery may be preterm for the safety of mother and baby. Severe preeclampsia can lead to maternal organ damage, seizures, and even death of mother and/or baby.
About 20% of women with severe preeclampsia will develop HELLP (Hemolysis, Elevated Liver enzymes, and Low Platelets) syndrome, a life-threatening disease that often warrants immediate delivery. According to the National Library of Medicine, the mortality rate of women with HELLP syndrome is up to 24% and the perinatal death rate is up as high as 37%. These serious conditions can cause ineffective maternal clotting, liver rupture, placental abruption, and postpartum hemorrhage. It is most prevalent in the third trimester but can occur within 48 hours of delivery.
The only cure for preeclampsia in any form is delivery.
Patients with severe preeclampsia are hospitalized until delivery – sometimes a few days to a couple of weeks. Mother and baby are closely watched for further progression, including signs of organ damage in the mother and changes to the well-being of the baby. If the mother’s health is severely compromised, then the baby will be compromised as well. A preterm delivery may be necessary.
Impact of the new test
The National Institute of Health states that preterm babies born from preeclamptic mothers can suffer many health problems including cerebral palsy, deafness, blindness, epilepsy, and a host of other respiratory, cardiovascular, and endocrine issues. But the biggest issue is preterm birth, defined as birth before 37 weeks gestation. Being born preterm can require a long stay in the intensive care nursery.
This is where the first-of-its-kind prognostic blood test comes into play. The test’s ability to predict severe preeclampsia within 2 weeks can help save lives. The test can offer health care providers the ability to administer steroids for fetal lung maturity before delivery and be more prepared to care for what could be a very compromised newborn.
The blood test, which is recommended between 23-35 weeks gestation, involves analyzing a ratio between two proteins from the placenta, sFlt1 and PIGF. The higher the ratio, the higher the risk that severe preeclampsia will develop. Results can be available within 30 minutes, which is critical when contemplating treatment.
An example of the use of this ratio is illustrated with chronic hypertension in pregnancy, which is defined as elevated blood pressure before 20 weeks or even before conception. Since chronic hypertension can be a primary precursor to preeclampsia, patients with this condition are at higher risk. The FDA-approved blood test would be helpful in determining the plan of care; that is, delivery versus hospitalization versus monitor as an outpatient.
With a positive test result, a pregnant woman can be immediately hospitalized where she can get the care she and baby need as they await delivery. Since health care providers already know the high-risk groups, surveillance can begin early, utilizing this blood test to predict the progression to severe preeclampsia. Conversely, if the test is negative, a treatment plan can be made as an outpatient and the pregnancy continues.
Not all hospitals are equipped to care for premature babies. If delivery is not imminent, providers can use this blood test to identify those that should be transferred to a tertiary center for observation and monitoring. Mother and baby would then not be separated after birth.
We really don’t know who will develop severe preeclampsia and who won’t. This new blood test will be a critical tool as pregnant patients go through their second and third trimesters. It will be especially pivotal for these women, but important for all pregnant women in reducing maternal and fetal mortality and morbidity.
Ms. Barnett is a registered nurse in the department of obstetrics, Mills-Peninsula Medical Center, Burlingame, Calif. She has disclosed no relevant financial relationships.
There is amazing news for the world of obstetrics and for all pregnant women. Severe preeclampsia is a critical obstetrical condition that can have serious outcomes for a mother and baby. It can lead to eclampsia, an obstetrical emergency, which often results in death of the mother and/or baby.
Based on research published in the Journal of the American Heart Association, the incidence of new‐onset hypertensive disorders of pregnancy (gestational hypertension and preeclampsia/eclampsia) have nearly doubled in the United States from 2007 to 2019. And they continue to climb.
According to the Preeclampsia Foundation, 5%-8% of all pregnancies in the United States will result in preeclampsia. Black women are at a 60% higher risk than white women, and according to various sources, other risk groups include those who became pregnant via in vitro fertilization, mothers of multiples (twins and triplets), women with gestational diabetes, women over age 35, women with chronic hypertension, obesity, polycystic ovary syndrome, sickle cell disease, rheumatoid arthritis, lupus, migraines, antiphospholipid syndrome, previous pregnancy with preeclampsia, family history, and scleroderma.
Screening and treatment
Preeclampsia is a multiorgan disease of pregnancy, and can be mild, but may quickly progress to severe, which can be life-threatening for mother and baby. It was previously referred to as toxemia or the high blood pressure disease of pregnancy. It primarily involves the cardiovascular, neurologic and renal systems, and the liver. Patients typically present with elevated blood pressures, but other symptoms may include headache, swelling of hands and feet, blurry/double vision or seeing spots, nausea/vomiting, and epigastric pain. It is diagnosed with elevated blood pressures, blood work, and protein in the urine.
Early screening for preeclampsia is done in the first trimester. Presently, a combination of prenatal blood work, blood pressure monitoring, and recognition of high-risk groups is used to determine a treatment plan going forward. The American Congress of Obstetricians and Gynecologists recommends women that fall into this group for potentially developing preeclampsia take daily aspirin as a preventative measure.
In its milder form, a pregnant woman can be observed as an outpatient – monitored with antepartum testing, lab work, and patient education to report significant symptoms as listed above. Teaching patients about fetal kick counts to monitor their baby’s movements is equally important. Women with mild preeclampsia usually can safely deliver at term, being induced between 37-39 weeks’ gestation.
On the other hand, if mild preeclampsia progresses to severe preeclampsia, delivery may be preterm for the safety of mother and baby. Severe preeclampsia can lead to maternal organ damage, seizures, and even death of mother and/or baby.
About 20% of women with severe preeclampsia will develop HELLP (Hemolysis, Elevated Liver enzymes, and Low Platelets) syndrome, a life-threatening disease that often warrants immediate delivery. According to the National Library of Medicine, the mortality rate of women with HELLP syndrome is up to 24% and the perinatal death rate is up as high as 37%. These serious conditions can cause ineffective maternal clotting, liver rupture, placental abruption, and postpartum hemorrhage. It is most prevalent in the third trimester but can occur within 48 hours of delivery.
The only cure for preeclampsia in any form is delivery.
Patients with severe preeclampsia are hospitalized until delivery – sometimes a few days to a couple of weeks. Mother and baby are closely watched for further progression, including signs of organ damage in the mother and changes to the well-being of the baby. If the mother’s health is severely compromised, then the baby will be compromised as well. A preterm delivery may be necessary.
Impact of the new test
The National Institute of Health states that preterm babies born from preeclamptic mothers can suffer many health problems including cerebral palsy, deafness, blindness, epilepsy, and a host of other respiratory, cardiovascular, and endocrine issues. But the biggest issue is preterm birth, defined as birth before 37 weeks gestation. Being born preterm can require a long stay in the intensive care nursery.
This is where the first-of-its-kind prognostic blood test comes into play. The test’s ability to predict severe preeclampsia within 2 weeks can help save lives. The test can offer health care providers the ability to administer steroids for fetal lung maturity before delivery and be more prepared to care for what could be a very compromised newborn.
The blood test, which is recommended between 23-35 weeks gestation, involves analyzing a ratio between two proteins from the placenta, sFlt1 and PIGF. The higher the ratio, the higher the risk that severe preeclampsia will develop. Results can be available within 30 minutes, which is critical when contemplating treatment.
An example of the use of this ratio is illustrated with chronic hypertension in pregnancy, which is defined as elevated blood pressure before 20 weeks or even before conception. Since chronic hypertension can be a primary precursor to preeclampsia, patients with this condition are at higher risk. The FDA-approved blood test would be helpful in determining the plan of care; that is, delivery versus hospitalization versus monitor as an outpatient.
With a positive test result, a pregnant woman can be immediately hospitalized where she can get the care she and baby need as they await delivery. Since health care providers already know the high-risk groups, surveillance can begin early, utilizing this blood test to predict the progression to severe preeclampsia. Conversely, if the test is negative, a treatment plan can be made as an outpatient and the pregnancy continues.
Not all hospitals are equipped to care for premature babies. If delivery is not imminent, providers can use this blood test to identify those that should be transferred to a tertiary center for observation and monitoring. Mother and baby would then not be separated after birth.
We really don’t know who will develop severe preeclampsia and who won’t. This new blood test will be a critical tool as pregnant patients go through their second and third trimesters. It will be especially pivotal for these women, but important for all pregnant women in reducing maternal and fetal mortality and morbidity.
Ms. Barnett is a registered nurse in the department of obstetrics, Mills-Peninsula Medical Center, Burlingame, Calif. She has disclosed no relevant financial relationships.
Continuous glucose monitors for pregnant patients?
Patients with pregestational diabetes may benefit from use of a continuous subcutaneous insulin infusion pump paired with a continuous glucose monitor. Use of the tools has been associated with a reduction in maternal and neonatal morbidity, a recent study found.
“We were seeing an unacceptable burden of both maternal and fetal disease in our diabetic population,” said Neil Hamill, MD, a maternal-fetal medicine specialist at Methodist Women’s Hospital, Omaha, Neb., and an author of the study. “We thought the success with this technology in the nonpregnant population would and should translate into the pregnant population.”
Dr. Hamill and his colleagues analyzed data from 55 pregnant patients who received care at the Women’s Hospital Perinatal Center at the Nebraska Methodist Health System between October 2019 and October 2022. Everyone in the cohort had pregestational diabetes and required insulin prior to week 20 of pregnancy. They used CGMs for more than 2 weeks. The study set blood glucose levels of less than 140 mg/dL as a healthy benchmark.
Participants who had severe preeclampsia, who had delivered preterm, who had delivered a neonate with respiratory distress syndrome, and/or who had given birth to a larger-than-expected infant spent less time in the safe zone – having a blood glucose level below 140 mg/dL – than women who did not have those risk factors.
“When blood sugar control is better, maternal and fetal outcomes are improved,” Dr. Hamill said.
Neetu Sodhi, MD, an ob.gyn. at Providence Cedars-Sinai Tarzana Medical Center, Los Angeles, expressed optimism that use of blood glucose monitors and insulin pumps can improve outcomes for pregnant patients with pregestational diabetes.
“This is just another case for why it’s so important for patients to have access to these types of devices that really, really improve their outcomes and their health, and now it’s proven in the case of pregnancy outcomes too – or at least suggested strongly with this data,” Dr. Sodhi said.
Mark Ebell, MD, a professor of epidemiology at the University of Georgia, Athens, was more skeptical, pointing out that study participants might have used other methods in addition to the technology to lower their blood sugar levels.
The findings suggest that insulin pumps are more manageable than multiple, daily self-injections. About 1 in 9 women have diabetes in the United States, and 35% of people newly diagnosed with the condition are women of reproductive age.
Dr. Hamill said that in future research, use of a stricter criterion for baseline blood sugar levels (< 140 mg/dL) would be helpful, as would exploring how much time patients need to spend below that level for optimal outcomes.
“Those questions are really absent in the literature,” Dr. Hamill said. “Most of our obstetrical literature is comparing treatment types. All those things are secondary. It’s the blood sugar that confers the risk, and if we get the blood sugar better, risk is reduced.”
Dr. Hamill added that the benefits of these technologies for patients with gestational diabetes are unclear in consideration of the limited duration of the disease and the time required to implant or install a monitor and pump, as well as associated risks and the cost of the devices.
Dr. Sodhi said clinicians who see patients during family planning visits should review morbidities and medical problems related to diabetes.
“I think this is a study that’s maybe too early,” Dr. Sodhi said. “They did ‘guesstimates’ on what target blood glucose ranges to be looking at, but I think over time, we might, with more studies like this, be building a case to try to put these types of monitors in for patients who are young for the purpose of optimizing pregnancy outcomes.”
The authors have disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Patients with pregestational diabetes may benefit from use of a continuous subcutaneous insulin infusion pump paired with a continuous glucose monitor. Use of the tools has been associated with a reduction in maternal and neonatal morbidity, a recent study found.
“We were seeing an unacceptable burden of both maternal and fetal disease in our diabetic population,” said Neil Hamill, MD, a maternal-fetal medicine specialist at Methodist Women’s Hospital, Omaha, Neb., and an author of the study. “We thought the success with this technology in the nonpregnant population would and should translate into the pregnant population.”
Dr. Hamill and his colleagues analyzed data from 55 pregnant patients who received care at the Women’s Hospital Perinatal Center at the Nebraska Methodist Health System between October 2019 and October 2022. Everyone in the cohort had pregestational diabetes and required insulin prior to week 20 of pregnancy. They used CGMs for more than 2 weeks. The study set blood glucose levels of less than 140 mg/dL as a healthy benchmark.
Participants who had severe preeclampsia, who had delivered preterm, who had delivered a neonate with respiratory distress syndrome, and/or who had given birth to a larger-than-expected infant spent less time in the safe zone – having a blood glucose level below 140 mg/dL – than women who did not have those risk factors.
“When blood sugar control is better, maternal and fetal outcomes are improved,” Dr. Hamill said.
Neetu Sodhi, MD, an ob.gyn. at Providence Cedars-Sinai Tarzana Medical Center, Los Angeles, expressed optimism that use of blood glucose monitors and insulin pumps can improve outcomes for pregnant patients with pregestational diabetes.
“This is just another case for why it’s so important for patients to have access to these types of devices that really, really improve their outcomes and their health, and now it’s proven in the case of pregnancy outcomes too – or at least suggested strongly with this data,” Dr. Sodhi said.
Mark Ebell, MD, a professor of epidemiology at the University of Georgia, Athens, was more skeptical, pointing out that study participants might have used other methods in addition to the technology to lower their blood sugar levels.
The findings suggest that insulin pumps are more manageable than multiple, daily self-injections. About 1 in 9 women have diabetes in the United States, and 35% of people newly diagnosed with the condition are women of reproductive age.
Dr. Hamill said that in future research, use of a stricter criterion for baseline blood sugar levels (< 140 mg/dL) would be helpful, as would exploring how much time patients need to spend below that level for optimal outcomes.
“Those questions are really absent in the literature,” Dr. Hamill said. “Most of our obstetrical literature is comparing treatment types. All those things are secondary. It’s the blood sugar that confers the risk, and if we get the blood sugar better, risk is reduced.”
Dr. Hamill added that the benefits of these technologies for patients with gestational diabetes are unclear in consideration of the limited duration of the disease and the time required to implant or install a monitor and pump, as well as associated risks and the cost of the devices.
Dr. Sodhi said clinicians who see patients during family planning visits should review morbidities and medical problems related to diabetes.
“I think this is a study that’s maybe too early,” Dr. Sodhi said. “They did ‘guesstimates’ on what target blood glucose ranges to be looking at, but I think over time, we might, with more studies like this, be building a case to try to put these types of monitors in for patients who are young for the purpose of optimizing pregnancy outcomes.”
The authors have disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Patients with pregestational diabetes may benefit from use of a continuous subcutaneous insulin infusion pump paired with a continuous glucose monitor. Use of the tools has been associated with a reduction in maternal and neonatal morbidity, a recent study found.
“We were seeing an unacceptable burden of both maternal and fetal disease in our diabetic population,” said Neil Hamill, MD, a maternal-fetal medicine specialist at Methodist Women’s Hospital, Omaha, Neb., and an author of the study. “We thought the success with this technology in the nonpregnant population would and should translate into the pregnant population.”
Dr. Hamill and his colleagues analyzed data from 55 pregnant patients who received care at the Women’s Hospital Perinatal Center at the Nebraska Methodist Health System between October 2019 and October 2022. Everyone in the cohort had pregestational diabetes and required insulin prior to week 20 of pregnancy. They used CGMs for more than 2 weeks. The study set blood glucose levels of less than 140 mg/dL as a healthy benchmark.
Participants who had severe preeclampsia, who had delivered preterm, who had delivered a neonate with respiratory distress syndrome, and/or who had given birth to a larger-than-expected infant spent less time in the safe zone – having a blood glucose level below 140 mg/dL – than women who did not have those risk factors.
“When blood sugar control is better, maternal and fetal outcomes are improved,” Dr. Hamill said.
Neetu Sodhi, MD, an ob.gyn. at Providence Cedars-Sinai Tarzana Medical Center, Los Angeles, expressed optimism that use of blood glucose monitors and insulin pumps can improve outcomes for pregnant patients with pregestational diabetes.
“This is just another case for why it’s so important for patients to have access to these types of devices that really, really improve their outcomes and their health, and now it’s proven in the case of pregnancy outcomes too – or at least suggested strongly with this data,” Dr. Sodhi said.
Mark Ebell, MD, a professor of epidemiology at the University of Georgia, Athens, was more skeptical, pointing out that study participants might have used other methods in addition to the technology to lower their blood sugar levels.
The findings suggest that insulin pumps are more manageable than multiple, daily self-injections. About 1 in 9 women have diabetes in the United States, and 35% of people newly diagnosed with the condition are women of reproductive age.
Dr. Hamill said that in future research, use of a stricter criterion for baseline blood sugar levels (< 140 mg/dL) would be helpful, as would exploring how much time patients need to spend below that level for optimal outcomes.
“Those questions are really absent in the literature,” Dr. Hamill said. “Most of our obstetrical literature is comparing treatment types. All those things are secondary. It’s the blood sugar that confers the risk, and if we get the blood sugar better, risk is reduced.”
Dr. Hamill added that the benefits of these technologies for patients with gestational diabetes are unclear in consideration of the limited duration of the disease and the time required to implant or install a monitor and pump, as well as associated risks and the cost of the devices.
Dr. Sodhi said clinicians who see patients during family planning visits should review morbidities and medical problems related to diabetes.
“I think this is a study that’s maybe too early,” Dr. Sodhi said. “They did ‘guesstimates’ on what target blood glucose ranges to be looking at, but I think over time, we might, with more studies like this, be building a case to try to put these types of monitors in for patients who are young for the purpose of optimizing pregnancy outcomes.”
The authors have disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Atopic dermatitis may be a risk factor for GBS colonization in pregnancy
suggest.
“The rate of GBS colonization among pregnant females with a history of AD has not been previously reported, but AD could be a risk factor for maternal carriage of GBS,” corresponding author David J. Margolis, MD, PhD, of the department of dermatology at the University of Pennsylvania, Philadelphia, and colleagues wrote in the study, which was published as a letter to the editor online in the Journal of Investigative Dermatology. “GBS reporting in a large administrative database represents a unique opportunity to conduct a population-based evaluation of GBS carriage with AD. Understanding this association could expand our understanding of microbial changes associated with AD,” they noted.
To determine if an association between GBS and AD in pregnant women exists, the researchers performed a cross-sectional study using a random sample from an Optum administrative database of pregnant women who had vaginal deliveries between May of 2007 and September 2021. The primary outcome of interest was the presence of GBS based on American College of Obstetricians and Gynecologists–recommended codes for GBS during 36 0/7 to 37 6/7 weeks of pregnancy. They used descriptive statistics to summarize categorical and continuous variables as proportions and means, and logistic regression to examine the association between AD and GBS status.
The cohort included 566,467 pregnant women with an average age of 38.8 years. Of these, 2.9% had a diagnosis of AD or a history of AD, and 24.9% had diagnoses of asthma, seasonal allergies, or both. Women with AD had an increased odds ratio of asthma (OR, 2.55), seasonal allergies (OR, 3.39), or both (OR, 5.35), compared with those without AD.
GBS was reported in 20.6% of the cohort. The median time of follow-up for those with and without GBS was 494 days and 468 days, respectively (P = .134). Among the women with AD, 24.1% had GBS, compared with 20.51% of the women without AD (P <.0001), which translated into an OR of 1.23 (95% confidence interval, 1.18-1.27).
Among the women with GBS, the OR of asthma was 1.08 (95% CI, 1.06-1.10) and was 1.07 (95% CI, 1.05-1.09) among those with seasonal allergies. When adjusted for potential confounders, these findings did not change substantively.
“It is not apparent why pregnant females with AD are more likely to specifically carry GBS,” the authors wrote. “However, several studies have shown that individuals with AD are more likely to carry [Staphylococcus] aureus and that individuals with AD might be deficient in host defenses against S. aureus and other pathogens,” they added.
“Individuals with AD frequently receive antibiotics as part of their AD treatment and this might alter their resident microbiome. Carriage rates may be enhanced by the inhibition of an important barrier protein called filaggrin (FLG) and FLG loss of function genetic variation is known to decrease barrier proteins thought to inhibit the colonization of S. aureus and other pathogens,” the researchers wrote.
They acknowledged certain limitations of their study, including its reliance on an administrative database that does not contain information on past disease.
Asked to comment on the results, Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was not involved with the study, characterized AD as “the poster child for cutaneous dysbiosis – an altered petri dish, so to speak, [that] facilitates survival of the few, leading to decreased microbial diversity that can both enable potential pathogen invasion and immune dysregulation.”
Though it’s not surprising that pregnant AD patients have dysbiosis, the focus on GBS, “which can be a bad actor in the perinatal period, is an interesting connection,” he said. “Will this change practices? Pregnant women should be screened for GBS regardless, but maybe more attention or counseling can be offered to AD patients about the importance of screening. Would decolonization regimens be employed early in pregnancy? This study can’t answer that but certainly raises good questions.”
Dr. Margolis disclosed that he is or recently has been a consultant for Pfizer, Leo, and Sanofi with respect to studies of atopic dermatitis and served on an advisory board for the National Eczema Association. Another author disclosed receiving grants from companies related to work with AD; other authors had no disclosures. Dr. Friedman reported having no relevant disclosures.
suggest.
“The rate of GBS colonization among pregnant females with a history of AD has not been previously reported, but AD could be a risk factor for maternal carriage of GBS,” corresponding author David J. Margolis, MD, PhD, of the department of dermatology at the University of Pennsylvania, Philadelphia, and colleagues wrote in the study, which was published as a letter to the editor online in the Journal of Investigative Dermatology. “GBS reporting in a large administrative database represents a unique opportunity to conduct a population-based evaluation of GBS carriage with AD. Understanding this association could expand our understanding of microbial changes associated with AD,” they noted.
To determine if an association between GBS and AD in pregnant women exists, the researchers performed a cross-sectional study using a random sample from an Optum administrative database of pregnant women who had vaginal deliveries between May of 2007 and September 2021. The primary outcome of interest was the presence of GBS based on American College of Obstetricians and Gynecologists–recommended codes for GBS during 36 0/7 to 37 6/7 weeks of pregnancy. They used descriptive statistics to summarize categorical and continuous variables as proportions and means, and logistic regression to examine the association between AD and GBS status.
The cohort included 566,467 pregnant women with an average age of 38.8 years. Of these, 2.9% had a diagnosis of AD or a history of AD, and 24.9% had diagnoses of asthma, seasonal allergies, or both. Women with AD had an increased odds ratio of asthma (OR, 2.55), seasonal allergies (OR, 3.39), or both (OR, 5.35), compared with those without AD.
GBS was reported in 20.6% of the cohort. The median time of follow-up for those with and without GBS was 494 days and 468 days, respectively (P = .134). Among the women with AD, 24.1% had GBS, compared with 20.51% of the women without AD (P <.0001), which translated into an OR of 1.23 (95% confidence interval, 1.18-1.27).
Among the women with GBS, the OR of asthma was 1.08 (95% CI, 1.06-1.10) and was 1.07 (95% CI, 1.05-1.09) among those with seasonal allergies. When adjusted for potential confounders, these findings did not change substantively.
“It is not apparent why pregnant females with AD are more likely to specifically carry GBS,” the authors wrote. “However, several studies have shown that individuals with AD are more likely to carry [Staphylococcus] aureus and that individuals with AD might be deficient in host defenses against S. aureus and other pathogens,” they added.
“Individuals with AD frequently receive antibiotics as part of their AD treatment and this might alter their resident microbiome. Carriage rates may be enhanced by the inhibition of an important barrier protein called filaggrin (FLG) and FLG loss of function genetic variation is known to decrease barrier proteins thought to inhibit the colonization of S. aureus and other pathogens,” the researchers wrote.
They acknowledged certain limitations of their study, including its reliance on an administrative database that does not contain information on past disease.
Asked to comment on the results, Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was not involved with the study, characterized AD as “the poster child for cutaneous dysbiosis – an altered petri dish, so to speak, [that] facilitates survival of the few, leading to decreased microbial diversity that can both enable potential pathogen invasion and immune dysregulation.”
Though it’s not surprising that pregnant AD patients have dysbiosis, the focus on GBS, “which can be a bad actor in the perinatal period, is an interesting connection,” he said. “Will this change practices? Pregnant women should be screened for GBS regardless, but maybe more attention or counseling can be offered to AD patients about the importance of screening. Would decolonization regimens be employed early in pregnancy? This study can’t answer that but certainly raises good questions.”
Dr. Margolis disclosed that he is or recently has been a consultant for Pfizer, Leo, and Sanofi with respect to studies of atopic dermatitis and served on an advisory board for the National Eczema Association. Another author disclosed receiving grants from companies related to work with AD; other authors had no disclosures. Dr. Friedman reported having no relevant disclosures.
suggest.
“The rate of GBS colonization among pregnant females with a history of AD has not been previously reported, but AD could be a risk factor for maternal carriage of GBS,” corresponding author David J. Margolis, MD, PhD, of the department of dermatology at the University of Pennsylvania, Philadelphia, and colleagues wrote in the study, which was published as a letter to the editor online in the Journal of Investigative Dermatology. “GBS reporting in a large administrative database represents a unique opportunity to conduct a population-based evaluation of GBS carriage with AD. Understanding this association could expand our understanding of microbial changes associated with AD,” they noted.
To determine if an association between GBS and AD in pregnant women exists, the researchers performed a cross-sectional study using a random sample from an Optum administrative database of pregnant women who had vaginal deliveries between May of 2007 and September 2021. The primary outcome of interest was the presence of GBS based on American College of Obstetricians and Gynecologists–recommended codes for GBS during 36 0/7 to 37 6/7 weeks of pregnancy. They used descriptive statistics to summarize categorical and continuous variables as proportions and means, and logistic regression to examine the association between AD and GBS status.
The cohort included 566,467 pregnant women with an average age of 38.8 years. Of these, 2.9% had a diagnosis of AD or a history of AD, and 24.9% had diagnoses of asthma, seasonal allergies, or both. Women with AD had an increased odds ratio of asthma (OR, 2.55), seasonal allergies (OR, 3.39), or both (OR, 5.35), compared with those without AD.
GBS was reported in 20.6% of the cohort. The median time of follow-up for those with and without GBS was 494 days and 468 days, respectively (P = .134). Among the women with AD, 24.1% had GBS, compared with 20.51% of the women without AD (P <.0001), which translated into an OR of 1.23 (95% confidence interval, 1.18-1.27).
Among the women with GBS, the OR of asthma was 1.08 (95% CI, 1.06-1.10) and was 1.07 (95% CI, 1.05-1.09) among those with seasonal allergies. When adjusted for potential confounders, these findings did not change substantively.
“It is not apparent why pregnant females with AD are more likely to specifically carry GBS,” the authors wrote. “However, several studies have shown that individuals with AD are more likely to carry [Staphylococcus] aureus and that individuals with AD might be deficient in host defenses against S. aureus and other pathogens,” they added.
“Individuals with AD frequently receive antibiotics as part of their AD treatment and this might alter their resident microbiome. Carriage rates may be enhanced by the inhibition of an important barrier protein called filaggrin (FLG) and FLG loss of function genetic variation is known to decrease barrier proteins thought to inhibit the colonization of S. aureus and other pathogens,” the researchers wrote.
They acknowledged certain limitations of their study, including its reliance on an administrative database that does not contain information on past disease.
Asked to comment on the results, Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was not involved with the study, characterized AD as “the poster child for cutaneous dysbiosis – an altered petri dish, so to speak, [that] facilitates survival of the few, leading to decreased microbial diversity that can both enable potential pathogen invasion and immune dysregulation.”
Though it’s not surprising that pregnant AD patients have dysbiosis, the focus on GBS, “which can be a bad actor in the perinatal period, is an interesting connection,” he said. “Will this change practices? Pregnant women should be screened for GBS regardless, but maybe more attention or counseling can be offered to AD patients about the importance of screening. Would decolonization regimens be employed early in pregnancy? This study can’t answer that but certainly raises good questions.”
Dr. Margolis disclosed that he is or recently has been a consultant for Pfizer, Leo, and Sanofi with respect to studies of atopic dermatitis and served on an advisory board for the National Eczema Association. Another author disclosed receiving grants from companies related to work with AD; other authors had no disclosures. Dr. Friedman reported having no relevant disclosures.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
One in five women report mistreatment during maternity care
“We have to do better at providing respectful and unbiased care to all mothers,” Debra E. Houry, MD, chief medical officer of the Centers for Disease Control and Prevention, said in a press briefing announcing the findings, which were published as a Vital Signs report in the CDC’s Morbidity and Mortality Weekly Report.
Previous research showed an increase in maternal deaths in the United States from 17.4 to 32.9 per 100,000 live births between 2018 and 2021, but approximately 80% of these deaths are preventable, wrote Yousra A. Mohamoud, PhD, of the CDC’s division of reproductive health, and colleagues.
“Maternal mortality review committees have identified discrimination as one factor contributing to pregnancy-related deaths,” the researchers wrote. Respectful care must be part of a larger strategy to prevent these deaths, they emphasized.
In the report, researchers reviewed data from 2,402 women who responded to an opt-in survey. The survey was conducted for the CDC through Porter Novelli, and no personally identifying information was included. Nearly 70% of the participants were White, 10.7% were Black, 10.2% were Hispanic, 4.8% were Asian, 1.5% were American Indian, Alaska Native, Pacific Islander, or Native Hawaiian, 2.8% were multiracial, and 0.5% were another race.
The survey included questions about maternity care experiences during pregnancy and delivery of the youngest child. For 65.5% of respondents, their youngest child was 5 years or older at the time of the survey.
Mistreatment during maternity care was defined using seven validated questions, including questions about violations of physical privacy, verbal abuse, and inattention to requests for help. Satisfaction with maternity care was defined as “very satisfied” or “somewhat satisfied.”
Participants also responded to questions about discrimination during maternity care based on factors such as race, ethnicity, skin color, age, and weight. Finally, participants were asked whether they refrained from asking questions about their health or raising concerns with health care providers.
Overall, 20.4% of respondents reported experiencing one of the defined forms of mistreatment during maternity care. The most common mistreatment reported by the women was being ignored by providers when they requested help (9.7%), followed by being shouted at or scolded (6.7%), having physical privacy violated (5.1%), and being forced to accept unwanted treatment or threatened with withholding of treatment (4.6%).
However, approximately 90% of women overall and 75% of those who reported any mistreatment were very or somewhat satisfied with their maternity care.
When stratified by race, mistreatment was reported most frequently by Black, Hispanic, and multiracial women (30%, 29%, and 27%, respectively).
Overall, 29% of women reported experiencing some type of discrimination; the most frequently reported reasons were age, weight, and income. Black women reported the highest rates of discrimination (40%) followed by multiracial women (39%) and Hispanic women (37%).
With regard to self-advocacy, 45% of women reported holding back from asking questions of health care providers; the most common reasons were thinking their health concerns were normal for pregnancy, being embarrassed, and being concerned that health care providers would consider them difficult.
In addition, more women with no insurance or public insurance at the time of delivery reported mistreatment during their maternity care than did women with private insurance (28%, 26%, and 16%, respectively).
The findings were limited by several factors, including the opt-in nature of the survey, which means that the data are likely not representative of the birthing population in the United States, the researchers noted. Other limitations included the reliance on self-reports, potential recall bias, use of English language only, and use of a combined category for respondents of American Indian, Alaska Native, Native Hawaiian, and Pacific Islander ethnicity.
However, the results highlight the need for improving respectful care as part of a larger strategy to reduce pregnancy-related deaths, the researchers said. At the system level, quality improvement programs are needed to standardize care and support providers in recognizing and reducing biases and increasing cultural awareness and communication. At the provider level, clinicians at all points in the maternity care process can improve patient experiences by providing equitable and respectful care, and by listening to and addressing patients’ concerns.
In addition, communication campaigns and community engagement can include perspectives of patients, families, and communities to support women and encourage them to ask questions and express concerns, the researchers said.
Improving respectful care can be part of actions to reduce mortality at all levels, the researchers noted. The Hear Her campaign, developed by the CDC Foundation with funding from Merck, provides resources for pregnant and postpartum women and their support networks to help reduce pregnancy-related deaths and complications by encouraging women to share concerns with providers and to recognize urgent maternal warning signs.
The study received no outside funding. The researchers had no financial conflicts to disclose.
“We have to do better at providing respectful and unbiased care to all mothers,” Debra E. Houry, MD, chief medical officer of the Centers for Disease Control and Prevention, said in a press briefing announcing the findings, which were published as a Vital Signs report in the CDC’s Morbidity and Mortality Weekly Report.
Previous research showed an increase in maternal deaths in the United States from 17.4 to 32.9 per 100,000 live births between 2018 and 2021, but approximately 80% of these deaths are preventable, wrote Yousra A. Mohamoud, PhD, of the CDC’s division of reproductive health, and colleagues.
“Maternal mortality review committees have identified discrimination as one factor contributing to pregnancy-related deaths,” the researchers wrote. Respectful care must be part of a larger strategy to prevent these deaths, they emphasized.
In the report, researchers reviewed data from 2,402 women who responded to an opt-in survey. The survey was conducted for the CDC through Porter Novelli, and no personally identifying information was included. Nearly 70% of the participants were White, 10.7% were Black, 10.2% were Hispanic, 4.8% were Asian, 1.5% were American Indian, Alaska Native, Pacific Islander, or Native Hawaiian, 2.8% were multiracial, and 0.5% were another race.
The survey included questions about maternity care experiences during pregnancy and delivery of the youngest child. For 65.5% of respondents, their youngest child was 5 years or older at the time of the survey.
Mistreatment during maternity care was defined using seven validated questions, including questions about violations of physical privacy, verbal abuse, and inattention to requests for help. Satisfaction with maternity care was defined as “very satisfied” or “somewhat satisfied.”
Participants also responded to questions about discrimination during maternity care based on factors such as race, ethnicity, skin color, age, and weight. Finally, participants were asked whether they refrained from asking questions about their health or raising concerns with health care providers.
Overall, 20.4% of respondents reported experiencing one of the defined forms of mistreatment during maternity care. The most common mistreatment reported by the women was being ignored by providers when they requested help (9.7%), followed by being shouted at or scolded (6.7%), having physical privacy violated (5.1%), and being forced to accept unwanted treatment or threatened with withholding of treatment (4.6%).
However, approximately 90% of women overall and 75% of those who reported any mistreatment were very or somewhat satisfied with their maternity care.
When stratified by race, mistreatment was reported most frequently by Black, Hispanic, and multiracial women (30%, 29%, and 27%, respectively).
Overall, 29% of women reported experiencing some type of discrimination; the most frequently reported reasons were age, weight, and income. Black women reported the highest rates of discrimination (40%) followed by multiracial women (39%) and Hispanic women (37%).
With regard to self-advocacy, 45% of women reported holding back from asking questions of health care providers; the most common reasons were thinking their health concerns were normal for pregnancy, being embarrassed, and being concerned that health care providers would consider them difficult.
In addition, more women with no insurance or public insurance at the time of delivery reported mistreatment during their maternity care than did women with private insurance (28%, 26%, and 16%, respectively).
The findings were limited by several factors, including the opt-in nature of the survey, which means that the data are likely not representative of the birthing population in the United States, the researchers noted. Other limitations included the reliance on self-reports, potential recall bias, use of English language only, and use of a combined category for respondents of American Indian, Alaska Native, Native Hawaiian, and Pacific Islander ethnicity.
However, the results highlight the need for improving respectful care as part of a larger strategy to reduce pregnancy-related deaths, the researchers said. At the system level, quality improvement programs are needed to standardize care and support providers in recognizing and reducing biases and increasing cultural awareness and communication. At the provider level, clinicians at all points in the maternity care process can improve patient experiences by providing equitable and respectful care, and by listening to and addressing patients’ concerns.
In addition, communication campaigns and community engagement can include perspectives of patients, families, and communities to support women and encourage them to ask questions and express concerns, the researchers said.
Improving respectful care can be part of actions to reduce mortality at all levels, the researchers noted. The Hear Her campaign, developed by the CDC Foundation with funding from Merck, provides resources for pregnant and postpartum women and their support networks to help reduce pregnancy-related deaths and complications by encouraging women to share concerns with providers and to recognize urgent maternal warning signs.
The study received no outside funding. The researchers had no financial conflicts to disclose.
“We have to do better at providing respectful and unbiased care to all mothers,” Debra E. Houry, MD, chief medical officer of the Centers for Disease Control and Prevention, said in a press briefing announcing the findings, which were published as a Vital Signs report in the CDC’s Morbidity and Mortality Weekly Report.
Previous research showed an increase in maternal deaths in the United States from 17.4 to 32.9 per 100,000 live births between 2018 and 2021, but approximately 80% of these deaths are preventable, wrote Yousra A. Mohamoud, PhD, of the CDC’s division of reproductive health, and colleagues.
“Maternal mortality review committees have identified discrimination as one factor contributing to pregnancy-related deaths,” the researchers wrote. Respectful care must be part of a larger strategy to prevent these deaths, they emphasized.
In the report, researchers reviewed data from 2,402 women who responded to an opt-in survey. The survey was conducted for the CDC through Porter Novelli, and no personally identifying information was included. Nearly 70% of the participants were White, 10.7% were Black, 10.2% were Hispanic, 4.8% were Asian, 1.5% were American Indian, Alaska Native, Pacific Islander, or Native Hawaiian, 2.8% were multiracial, and 0.5% were another race.
The survey included questions about maternity care experiences during pregnancy and delivery of the youngest child. For 65.5% of respondents, their youngest child was 5 years or older at the time of the survey.
Mistreatment during maternity care was defined using seven validated questions, including questions about violations of physical privacy, verbal abuse, and inattention to requests for help. Satisfaction with maternity care was defined as “very satisfied” or “somewhat satisfied.”
Participants also responded to questions about discrimination during maternity care based on factors such as race, ethnicity, skin color, age, and weight. Finally, participants were asked whether they refrained from asking questions about their health or raising concerns with health care providers.
Overall, 20.4% of respondents reported experiencing one of the defined forms of mistreatment during maternity care. The most common mistreatment reported by the women was being ignored by providers when they requested help (9.7%), followed by being shouted at or scolded (6.7%), having physical privacy violated (5.1%), and being forced to accept unwanted treatment or threatened with withholding of treatment (4.6%).
However, approximately 90% of women overall and 75% of those who reported any mistreatment were very or somewhat satisfied with their maternity care.
When stratified by race, mistreatment was reported most frequently by Black, Hispanic, and multiracial women (30%, 29%, and 27%, respectively).
Overall, 29% of women reported experiencing some type of discrimination; the most frequently reported reasons were age, weight, and income. Black women reported the highest rates of discrimination (40%) followed by multiracial women (39%) and Hispanic women (37%).
With regard to self-advocacy, 45% of women reported holding back from asking questions of health care providers; the most common reasons were thinking their health concerns were normal for pregnancy, being embarrassed, and being concerned that health care providers would consider them difficult.
In addition, more women with no insurance or public insurance at the time of delivery reported mistreatment during their maternity care than did women with private insurance (28%, 26%, and 16%, respectively).
The findings were limited by several factors, including the opt-in nature of the survey, which means that the data are likely not representative of the birthing population in the United States, the researchers noted. Other limitations included the reliance on self-reports, potential recall bias, use of English language only, and use of a combined category for respondents of American Indian, Alaska Native, Native Hawaiian, and Pacific Islander ethnicity.
However, the results highlight the need for improving respectful care as part of a larger strategy to reduce pregnancy-related deaths, the researchers said. At the system level, quality improvement programs are needed to standardize care and support providers in recognizing and reducing biases and increasing cultural awareness and communication. At the provider level, clinicians at all points in the maternity care process can improve patient experiences by providing equitable and respectful care, and by listening to and addressing patients’ concerns.
In addition, communication campaigns and community engagement can include perspectives of patients, families, and communities to support women and encourage them to ask questions and express concerns, the researchers said.
Improving respectful care can be part of actions to reduce mortality at all levels, the researchers noted. The Hear Her campaign, developed by the CDC Foundation with funding from Merck, provides resources for pregnant and postpartum women and their support networks to help reduce pregnancy-related deaths and complications by encouraging women to share concerns with providers and to recognize urgent maternal warning signs.
The study received no outside funding. The researchers had no financial conflicts to disclose.
FROM MMWR
Mothers in medicine: What can we learn when worlds collide?
Across all industries, studies by the U.S. Department of Labor have shown that women, on average, earn 83.7 percent of what their male peers earn. While a lot has been written about the struggles women face in medicine, there have been decidedly fewer analyses that focus on women who choose to become mothers while working in medicine.
I’ve been privileged to work with medical students and residents for the last 8 years as the director of graduate and medical student mental health at Rowan-Virtua School of Osteopathic Medicine in Mt. Laurel, N.J. Often, the women I see as patients speak about their struggles with the elusive goal of “having it all.” While both men and women in medicine have difficulty maintaining a work-life balance, I’ve learned, both personally and professionally, that many women face a unique set of challenges.
No matter what their professional status, our society often views a woman as the default parent. For example, the teacher often calls the mothers first. The camp nurse calls me first, not my husband, when our child scrapes a knee. After-school play dates are arranged by the mothers, not fathers.
But mothers also bring to medicine a wealth of unique experiences, ideas, and viewpoints. They learn firsthand how to foster affect regulation and frustration tolerance in their kids and become efficient at managing the constant, conflicting tug of war of demands.
Some may argue that, over time, women end up earning significantly less than their male counterparts because they leave the workforce while on maternity leave, ultimately delaying their upward career progression. It’s likely a much more complex problem. Many of my patients believe that, in our male-dominated society (and workforce), women are punished for being aggressive or stating bold opinions, while men are rewarded for the same actions. While a man may sound forceful and in charge, a women will likely be thought of as brusque and unappreciative.
Outside of work, many women may have more on their plate. A 2020 Gallup poll of more than 3,000 heterosexual couples found that women are responsible for the majority of household chores. Women continue to handle more of the emotional labor within their families, regardless of income, age, or professional status. This is sometimes called the “Mental Load’ or “Second Shift.” As our society continues to view women as the default parent for childcare, medical issues, and overarching social and emotional tasks vital to raising happy, healthy children, the struggle a female medical professional feels is palpable.
Raising kids requires a parent to consistently dole out control, predictability, and reassurance for a child to thrive. Good limit and boundary setting leads to healthy development from a young age.
Psychiatric patients (and perhaps all patients) also require control, predictability, and reassurance from their doctor. The lessons learned in being a good mother can be directly applied in patient care, and vice versa. The cross-pollination of this relationship continues to grow more powerful as a woman’s children grow and her career matures.
Pediatrician and psychoanalyst Donald Winnicott’s idea of a “good enough” mother cannot be a one-size-fits-all approach. Women who self-select into the world of medicine often hold themselves to a higher standard than “good enough.” Acknowledging that the demands from both home and work will fluctuate is key to achieving success both personally and professionally, and lessons from home can and should be utilized to become a more effective physician. The notion of having it all, and the definition of success, must evolve over time.
Dr. Maymind is director of medical and graduate student mental health at Rowan-Virtua School of Osteopathic Medicine in Mt. Laurel, N.J. She has no relevant disclosures.
Across all industries, studies by the U.S. Department of Labor have shown that women, on average, earn 83.7 percent of what their male peers earn. While a lot has been written about the struggles women face in medicine, there have been decidedly fewer analyses that focus on women who choose to become mothers while working in medicine.
I’ve been privileged to work with medical students and residents for the last 8 years as the director of graduate and medical student mental health at Rowan-Virtua School of Osteopathic Medicine in Mt. Laurel, N.J. Often, the women I see as patients speak about their struggles with the elusive goal of “having it all.” While both men and women in medicine have difficulty maintaining a work-life balance, I’ve learned, both personally and professionally, that many women face a unique set of challenges.
No matter what their professional status, our society often views a woman as the default parent. For example, the teacher often calls the mothers first. The camp nurse calls me first, not my husband, when our child scrapes a knee. After-school play dates are arranged by the mothers, not fathers.
But mothers also bring to medicine a wealth of unique experiences, ideas, and viewpoints. They learn firsthand how to foster affect regulation and frustration tolerance in their kids and become efficient at managing the constant, conflicting tug of war of demands.
Some may argue that, over time, women end up earning significantly less than their male counterparts because they leave the workforce while on maternity leave, ultimately delaying their upward career progression. It’s likely a much more complex problem. Many of my patients believe that, in our male-dominated society (and workforce), women are punished for being aggressive or stating bold opinions, while men are rewarded for the same actions. While a man may sound forceful and in charge, a women will likely be thought of as brusque and unappreciative.
Outside of work, many women may have more on their plate. A 2020 Gallup poll of more than 3,000 heterosexual couples found that women are responsible for the majority of household chores. Women continue to handle more of the emotional labor within their families, regardless of income, age, or professional status. This is sometimes called the “Mental Load’ or “Second Shift.” As our society continues to view women as the default parent for childcare, medical issues, and overarching social and emotional tasks vital to raising happy, healthy children, the struggle a female medical professional feels is palpable.
Raising kids requires a parent to consistently dole out control, predictability, and reassurance for a child to thrive. Good limit and boundary setting leads to healthy development from a young age.
Psychiatric patients (and perhaps all patients) also require control, predictability, and reassurance from their doctor. The lessons learned in being a good mother can be directly applied in patient care, and vice versa. The cross-pollination of this relationship continues to grow more powerful as a woman’s children grow and her career matures.
Pediatrician and psychoanalyst Donald Winnicott’s idea of a “good enough” mother cannot be a one-size-fits-all approach. Women who self-select into the world of medicine often hold themselves to a higher standard than “good enough.” Acknowledging that the demands from both home and work will fluctuate is key to achieving success both personally and professionally, and lessons from home can and should be utilized to become a more effective physician. The notion of having it all, and the definition of success, must evolve over time.
Dr. Maymind is director of medical and graduate student mental health at Rowan-Virtua School of Osteopathic Medicine in Mt. Laurel, N.J. She has no relevant disclosures.
Across all industries, studies by the U.S. Department of Labor have shown that women, on average, earn 83.7 percent of what their male peers earn. While a lot has been written about the struggles women face in medicine, there have been decidedly fewer analyses that focus on women who choose to become mothers while working in medicine.
I’ve been privileged to work with medical students and residents for the last 8 years as the director of graduate and medical student mental health at Rowan-Virtua School of Osteopathic Medicine in Mt. Laurel, N.J. Often, the women I see as patients speak about their struggles with the elusive goal of “having it all.” While both men and women in medicine have difficulty maintaining a work-life balance, I’ve learned, both personally and professionally, that many women face a unique set of challenges.
No matter what their professional status, our society often views a woman as the default parent. For example, the teacher often calls the mothers first. The camp nurse calls me first, not my husband, when our child scrapes a knee. After-school play dates are arranged by the mothers, not fathers.
But mothers also bring to medicine a wealth of unique experiences, ideas, and viewpoints. They learn firsthand how to foster affect regulation and frustration tolerance in their kids and become efficient at managing the constant, conflicting tug of war of demands.
Some may argue that, over time, women end up earning significantly less than their male counterparts because they leave the workforce while on maternity leave, ultimately delaying their upward career progression. It’s likely a much more complex problem. Many of my patients believe that, in our male-dominated society (and workforce), women are punished for being aggressive or stating bold opinions, while men are rewarded for the same actions. While a man may sound forceful and in charge, a women will likely be thought of as brusque and unappreciative.
Outside of work, many women may have more on their plate. A 2020 Gallup poll of more than 3,000 heterosexual couples found that women are responsible for the majority of household chores. Women continue to handle more of the emotional labor within their families, regardless of income, age, or professional status. This is sometimes called the “Mental Load’ or “Second Shift.” As our society continues to view women as the default parent for childcare, medical issues, and overarching social and emotional tasks vital to raising happy, healthy children, the struggle a female medical professional feels is palpable.
Raising kids requires a parent to consistently dole out control, predictability, and reassurance for a child to thrive. Good limit and boundary setting leads to healthy development from a young age.
Psychiatric patients (and perhaps all patients) also require control, predictability, and reassurance from their doctor. The lessons learned in being a good mother can be directly applied in patient care, and vice versa. The cross-pollination of this relationship continues to grow more powerful as a woman’s children grow and her career matures.
Pediatrician and psychoanalyst Donald Winnicott’s idea of a “good enough” mother cannot be a one-size-fits-all approach. Women who self-select into the world of medicine often hold themselves to a higher standard than “good enough.” Acknowledging that the demands from both home and work will fluctuate is key to achieving success both personally and professionally, and lessons from home can and should be utilized to become a more effective physician. The notion of having it all, and the definition of success, must evolve over time.
Dr. Maymind is director of medical and graduate student mental health at Rowan-Virtua School of Osteopathic Medicine in Mt. Laurel, N.J. She has no relevant disclosures.















