User login
Combination beats misoprostol monotherapy on cost effectiveness
A combination of mifepristone followed by misoprostol was significantly more cost effective for the medical management of miscarriage than misoprostol alone, based on a decision-tree model and simulations using a range of patient income levels, cost variables, and practice patterns.
Although the American College of Obstetricians and Gynecologists recommends a combination of mifepristone and misoprostol for the medical management of miscarriage, some physicians may hesitate because of the high cost of mifepristone, wrote Holly H. Berkley, MD, of the Naval Medical Center, San Diego, and colleagues.
Previous research has supported the cost effectiveness of combination therapy, but the data came from a secondary analysis that limited the generalizability of the findings, they wrote. In a study published in Obstetrics & Gynecology, the researchers created a decision-tree model using two standard practice patterns.
In the first, patients received mifepristone and one dose of misoprostol (combination therapy) or one dose of misoprostol alone (monotherapy) at their initial visit with follow-up within 3 days. Combination therapy was defined as 200 mg of oral mifepristone followed by one or two doses of 800 micrograms of vaginal misoprostol; monotherapy was defined as one or two doses of 800 micrograms of vaginal misoprostol.
“If miscarriage is not completed, a second dose of misoprostol is given, and the patient will have a second follow-up visit 8 days after initiation of treatment. If miscarriage is not complete at the second follow-up visit, surgical management is prescribed,” Dr. Berkley and associates reported.
In the second pattern, patients receive two doses of misoprostol at the first visit and an initial follow-up visit 8 days later.
Patient hourly income was based on the wages of three employment levels of the military patient population, estimated at $7.25/hour, $15.90/hour, and $35.10 per hour. “For clinicians outside of the military health system, these wage categories may also serve as an estimate of earnings for low-income, low-middle income, and middle-income patients across the United States,” Dr. Berkley and colleagues noted.
The researchers also considered costs for time of work, transportation, and the costs of the medical visits. Costs also were computed for surgical management with in–operating room dilation and curettage or in-office manual vacuum aspiration, if needed.
The greatest difference in favor of combination therapy resulted in a savings of $190.20 per patient, compared with monotherapy, in the first practice pattern and the lowest wage group (19.5%).
“In every scenario, and for every wage level, the average cost of combination therapy is less than that of monotherapy,” Dr. Berkley and associates noted. In addition, the differences in cost between combination therapy and monotherapy increased with patients’ wages, “reflecting wage differences as well as the net savings owing to increased completion rates.”
Completion rates are key to cost effectiveness
“The higher completion rate of combination therapy leads to decreased time spent on treatment and therefore decreased time off work, decreased office visits, and a decreased need for surgical management for persistent pregnancy, which significantly reduces cost,” they noted.
The model shows that the cost of mifepristone, which some clinicians may see as a barrier, contributes little to the overall treatment costs, Dr. Berkley and colleagues emphasized.
The study findings were limited by several factors including the large ranges in costs for office visits and procedures and the inability to replicate all clinical settings and variables, the researchers noted. However, the results were strengthened by the use of current practice patterns and costs, and they support the mifepristone/misoprostol combination as being the most cost effective for medical management of miscarriage, they said.
The findings of the current study, combined with higher effectiveness reported in recent randomized controlled trials and the endorsement of the American College of Obstetricians and Gynecologists “make a strong case for mifepristone followed by misoprostol to become the standard, first-line treatment regimen for the medical management of miscarriage,” Dr. Berkley and associates concluded.
Research is clear, policy needs to catch up
“There is clear research showing that using mifepristone with misoprostol to medically manage early pregnancy loss is significantly more effective than misoprostol alone,” Sarah Prager, MD, of the University of Washington, Seattle, said in an interview. “The combination protocol does include an expensive medication, so it’s important to evaluate if the cost of this more effective method is ‘worth it,’ ” she said. “ including fewer projected days off work and fewer patients needing procedures to complete their miscarriage.”
Dr. Prager said she was not surprised by the study findings because similar results have been found in other studies evaluating treatment of abortion with misoprostol alone versus mifepristone and misoprostol. “When something is significantly more effective, it usually will also come with a cost benefit.”
Potential barriers to the use of combination therapy are related to policy rather than drug safety or effectiveness, according to Dr. Prager.
“The primary barrier is that mifepristone use is regulated by a REMS [Risk Evaluation and Mitigation Strategy] restriction which requires that providers dispense the medication directly to patients, rather than being able to prescribe it and have patients then pick it up at a pharmacy,” she said. “This restriction is typically used for medications that are dangerous and need to be closely controlled. In the case of mifepristone, the restriction does not serve a safety purpose; it simply limits access to the medication which is still primarily used to medically treat abortion.
“The secondary barrier is stigma against using a medication that is seen as an abortion medication. I fear many providers or practices may avoid putting it on formulary due to this stigma,” Dr. Prager noted.
“There is already sufficient evidence that the combination therapy is superior to monotherapy, and there is also evidence that mifepristone can be safely prescribed [not dispensed] and does not need the REMS requirement,” Dr. Prager said. “I don’t believe more research is needed; just policy change.”
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Prager had no financial conflicts to disclose.
SOURCE: Berkley HH et al. Obstet Gynecol. 2020 Oct. doi: 10.1097/AOG.0000000000004063.
A combination of mifepristone followed by misoprostol was significantly more cost effective for the medical management of miscarriage than misoprostol alone, based on a decision-tree model and simulations using a range of patient income levels, cost variables, and practice patterns.
Although the American College of Obstetricians and Gynecologists recommends a combination of mifepristone and misoprostol for the medical management of miscarriage, some physicians may hesitate because of the high cost of mifepristone, wrote Holly H. Berkley, MD, of the Naval Medical Center, San Diego, and colleagues.
Previous research has supported the cost effectiveness of combination therapy, but the data came from a secondary analysis that limited the generalizability of the findings, they wrote. In a study published in Obstetrics & Gynecology, the researchers created a decision-tree model using two standard practice patterns.
In the first, patients received mifepristone and one dose of misoprostol (combination therapy) or one dose of misoprostol alone (monotherapy) at their initial visit with follow-up within 3 days. Combination therapy was defined as 200 mg of oral mifepristone followed by one or two doses of 800 micrograms of vaginal misoprostol; monotherapy was defined as one or two doses of 800 micrograms of vaginal misoprostol.
“If miscarriage is not completed, a second dose of misoprostol is given, and the patient will have a second follow-up visit 8 days after initiation of treatment. If miscarriage is not complete at the second follow-up visit, surgical management is prescribed,” Dr. Berkley and associates reported.
In the second pattern, patients receive two doses of misoprostol at the first visit and an initial follow-up visit 8 days later.
Patient hourly income was based on the wages of three employment levels of the military patient population, estimated at $7.25/hour, $15.90/hour, and $35.10 per hour. “For clinicians outside of the military health system, these wage categories may also serve as an estimate of earnings for low-income, low-middle income, and middle-income patients across the United States,” Dr. Berkley and colleagues noted.
The researchers also considered costs for time of work, transportation, and the costs of the medical visits. Costs also were computed for surgical management with in–operating room dilation and curettage or in-office manual vacuum aspiration, if needed.
The greatest difference in favor of combination therapy resulted in a savings of $190.20 per patient, compared with monotherapy, in the first practice pattern and the lowest wage group (19.5%).
“In every scenario, and for every wage level, the average cost of combination therapy is less than that of monotherapy,” Dr. Berkley and associates noted. In addition, the differences in cost between combination therapy and monotherapy increased with patients’ wages, “reflecting wage differences as well as the net savings owing to increased completion rates.”
Completion rates are key to cost effectiveness
“The higher completion rate of combination therapy leads to decreased time spent on treatment and therefore decreased time off work, decreased office visits, and a decreased need for surgical management for persistent pregnancy, which significantly reduces cost,” they noted.
The model shows that the cost of mifepristone, which some clinicians may see as a barrier, contributes little to the overall treatment costs, Dr. Berkley and colleagues emphasized.
The study findings were limited by several factors including the large ranges in costs for office visits and procedures and the inability to replicate all clinical settings and variables, the researchers noted. However, the results were strengthened by the use of current practice patterns and costs, and they support the mifepristone/misoprostol combination as being the most cost effective for medical management of miscarriage, they said.
The findings of the current study, combined with higher effectiveness reported in recent randomized controlled trials and the endorsement of the American College of Obstetricians and Gynecologists “make a strong case for mifepristone followed by misoprostol to become the standard, first-line treatment regimen for the medical management of miscarriage,” Dr. Berkley and associates concluded.
Research is clear, policy needs to catch up
“There is clear research showing that using mifepristone with misoprostol to medically manage early pregnancy loss is significantly more effective than misoprostol alone,” Sarah Prager, MD, of the University of Washington, Seattle, said in an interview. “The combination protocol does include an expensive medication, so it’s important to evaluate if the cost of this more effective method is ‘worth it,’ ” she said. “ including fewer projected days off work and fewer patients needing procedures to complete their miscarriage.”
Dr. Prager said she was not surprised by the study findings because similar results have been found in other studies evaluating treatment of abortion with misoprostol alone versus mifepristone and misoprostol. “When something is significantly more effective, it usually will also come with a cost benefit.”
Potential barriers to the use of combination therapy are related to policy rather than drug safety or effectiveness, according to Dr. Prager.
“The primary barrier is that mifepristone use is regulated by a REMS [Risk Evaluation and Mitigation Strategy] restriction which requires that providers dispense the medication directly to patients, rather than being able to prescribe it and have patients then pick it up at a pharmacy,” she said. “This restriction is typically used for medications that are dangerous and need to be closely controlled. In the case of mifepristone, the restriction does not serve a safety purpose; it simply limits access to the medication which is still primarily used to medically treat abortion.
“The secondary barrier is stigma against using a medication that is seen as an abortion medication. I fear many providers or practices may avoid putting it on formulary due to this stigma,” Dr. Prager noted.
“There is already sufficient evidence that the combination therapy is superior to monotherapy, and there is also evidence that mifepristone can be safely prescribed [not dispensed] and does not need the REMS requirement,” Dr. Prager said. “I don’t believe more research is needed; just policy change.”
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Prager had no financial conflicts to disclose.
SOURCE: Berkley HH et al. Obstet Gynecol. 2020 Oct. doi: 10.1097/AOG.0000000000004063.
A combination of mifepristone followed by misoprostol was significantly more cost effective for the medical management of miscarriage than misoprostol alone, based on a decision-tree model and simulations using a range of patient income levels, cost variables, and practice patterns.
Although the American College of Obstetricians and Gynecologists recommends a combination of mifepristone and misoprostol for the medical management of miscarriage, some physicians may hesitate because of the high cost of mifepristone, wrote Holly H. Berkley, MD, of the Naval Medical Center, San Diego, and colleagues.
Previous research has supported the cost effectiveness of combination therapy, but the data came from a secondary analysis that limited the generalizability of the findings, they wrote. In a study published in Obstetrics & Gynecology, the researchers created a decision-tree model using two standard practice patterns.
In the first, patients received mifepristone and one dose of misoprostol (combination therapy) or one dose of misoprostol alone (monotherapy) at their initial visit with follow-up within 3 days. Combination therapy was defined as 200 mg of oral mifepristone followed by one or two doses of 800 micrograms of vaginal misoprostol; monotherapy was defined as one or two doses of 800 micrograms of vaginal misoprostol.
“If miscarriage is not completed, a second dose of misoprostol is given, and the patient will have a second follow-up visit 8 days after initiation of treatment. If miscarriage is not complete at the second follow-up visit, surgical management is prescribed,” Dr. Berkley and associates reported.
In the second pattern, patients receive two doses of misoprostol at the first visit and an initial follow-up visit 8 days later.
Patient hourly income was based on the wages of three employment levels of the military patient population, estimated at $7.25/hour, $15.90/hour, and $35.10 per hour. “For clinicians outside of the military health system, these wage categories may also serve as an estimate of earnings for low-income, low-middle income, and middle-income patients across the United States,” Dr. Berkley and colleagues noted.
The researchers also considered costs for time of work, transportation, and the costs of the medical visits. Costs also were computed for surgical management with in–operating room dilation and curettage or in-office manual vacuum aspiration, if needed.
The greatest difference in favor of combination therapy resulted in a savings of $190.20 per patient, compared with monotherapy, in the first practice pattern and the lowest wage group (19.5%).
“In every scenario, and for every wage level, the average cost of combination therapy is less than that of monotherapy,” Dr. Berkley and associates noted. In addition, the differences in cost between combination therapy and monotherapy increased with patients’ wages, “reflecting wage differences as well as the net savings owing to increased completion rates.”
Completion rates are key to cost effectiveness
“The higher completion rate of combination therapy leads to decreased time spent on treatment and therefore decreased time off work, decreased office visits, and a decreased need for surgical management for persistent pregnancy, which significantly reduces cost,” they noted.
The model shows that the cost of mifepristone, which some clinicians may see as a barrier, contributes little to the overall treatment costs, Dr. Berkley and colleagues emphasized.
The study findings were limited by several factors including the large ranges in costs for office visits and procedures and the inability to replicate all clinical settings and variables, the researchers noted. However, the results were strengthened by the use of current practice patterns and costs, and they support the mifepristone/misoprostol combination as being the most cost effective for medical management of miscarriage, they said.
The findings of the current study, combined with higher effectiveness reported in recent randomized controlled trials and the endorsement of the American College of Obstetricians and Gynecologists “make a strong case for mifepristone followed by misoprostol to become the standard, first-line treatment regimen for the medical management of miscarriage,” Dr. Berkley and associates concluded.
Research is clear, policy needs to catch up
“There is clear research showing that using mifepristone with misoprostol to medically manage early pregnancy loss is significantly more effective than misoprostol alone,” Sarah Prager, MD, of the University of Washington, Seattle, said in an interview. “The combination protocol does include an expensive medication, so it’s important to evaluate if the cost of this more effective method is ‘worth it,’ ” she said. “ including fewer projected days off work and fewer patients needing procedures to complete their miscarriage.”
Dr. Prager said she was not surprised by the study findings because similar results have been found in other studies evaluating treatment of abortion with misoprostol alone versus mifepristone and misoprostol. “When something is significantly more effective, it usually will also come with a cost benefit.”
Potential barriers to the use of combination therapy are related to policy rather than drug safety or effectiveness, according to Dr. Prager.
“The primary barrier is that mifepristone use is regulated by a REMS [Risk Evaluation and Mitigation Strategy] restriction which requires that providers dispense the medication directly to patients, rather than being able to prescribe it and have patients then pick it up at a pharmacy,” she said. “This restriction is typically used for medications that are dangerous and need to be closely controlled. In the case of mifepristone, the restriction does not serve a safety purpose; it simply limits access to the medication which is still primarily used to medically treat abortion.
“The secondary barrier is stigma against using a medication that is seen as an abortion medication. I fear many providers or practices may avoid putting it on formulary due to this stigma,” Dr. Prager noted.
“There is already sufficient evidence that the combination therapy is superior to monotherapy, and there is also evidence that mifepristone can be safely prescribed [not dispensed] and does not need the REMS requirement,” Dr. Prager said. “I don’t believe more research is needed; just policy change.”
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Prager had no financial conflicts to disclose.
SOURCE: Berkley HH et al. Obstet Gynecol. 2020 Oct. doi: 10.1097/AOG.0000000000004063.
FROM OBSTETRICS & GYNECOLOGY
Consider switching up treatment regimens for recurrent bacterial vaginosis
Limited data are available to guide treatment of recurrent bacterial vaginosis, but behavioral changes and switching between approved medication regimens may help, according to a presenter at the virtual conference on diseases of the vulva and vagina, hosted by the International Society for the Study of Vulvovaginal Disease.
Investigational treatments – such as a live biotherapeutic product delivered vaginally or vaginal microbiome transplantation – could someday be additional options if they prove safe and effective. said Debra L. Birenbaum, MD, assistant professor of obstetrics and gynecology at Dartmouth-Hitchcock Medical Center in Lebanon, N.H.
As for home remedies, Dr. Birenbaum and another presenter at the conference, Cynthia Rasmussen, MD, urged caution during a panel discussion.
“I think the vagina knows its business, and the more you mess with it, the more you invite trouble,” said Dr. Rasmussen, director emerita of vulvovaginal services at Atrius Health in Burlington, Mass. For instance, tea tree oil, often cited as a home remedy, can be an allergen and very irritating.
“I want to know what women are using, but I try and dissuade them,” said Dr. Birenbaum. “I have to be careful what I say, because you’ll antagonize patients” if you come out strongly against home treatments. “I try to encourage them not to go by things they read on the Internet, because I think that’s where many people are finding their home remedies.”
When counseling patients, an analogy shared during the meeting – the vagina is a self-cleaning oven – may help get the point across. “I love the comment,” Dr. Birenbaum said. “I’ve never used that before. I’m going to start saying that.”
Possible causes and risk factors
Bacterial vaginosis, also known as vaginal dysbiosis, is the most common cause of discharge in women of reproductive age worldwide. Growth of a biofilm may cause the condition, which is characterized by a shift in vaginal flora from a Lactobacilli-dominant environment to one of other bacterial types.
Risk factors include douching, smoking, sex with an uncircumcised partner, and having multiple sexual partners. Bacterial vaginosis may be associated with various complications and infections, including increased risk of preterm delivery, postpartum endometritis, postabortal infection, Trichomonas, chlamydia, and HIV.
Unlike recurrent yeast, which is characterized by four or more episodes per year, recurrent bacterial vaginosis has no official criteria, Dr. Birenbaum said. However, recurrence of bacterial vaginosis “is extremely common,” she said. “Up to 30% of women with [bacterial vaginosis] may recur within 3 months, and up to 50% after 12 months.”
Lifestyle changes and treatments
Recommendations to use condoms, stop smoking, and not douche are important.
In addition, 11 treatment regimens for four drugs – metronidazole, clindamycin, tinidazole, or secnidazole – are available for the treatment of bacterial vaginosis. For recurrent cases, adjusting and switching between the drugs and modes of delivery may help. If a patient started with vaginal gel, they can try an oral medication, or vice versa.
“There’s very little data to guide the optimal therapy for this,” Dr. Birenbaum said. “All of this is worth a try to see if you can beat this before this becomes an ongoing issue.”
As an example of one possible regimen for recurrent bacterial vaginosis, Dr. Birenbaum suggested that a patient could complete a 2- to 4-week course of oral metronidazole instead of the usual 1-week course. The regimen could incorporate boric acid vaginal suppositories 600 mg nightly for 21 days, followed by metronidazole gel 0.75% (one applicator twice per week for 6 months).
New therapies may be on the horizon
In a randomized, double-blind, phase 2b trial published in the New England Journal of Medicine that included more than 220 participants, patients who received an investigational product containing Lactobacillus crispatus CTV-05 (Lactin-V) were less likely to have recurrent bacterial vaginosis at 12 weeks, compared with those who received placebo (30% vs. 45%).
A product in development known as TOL-463, a boric acid–based vaginal anti-infective enhanced with ethylenediaminetetraacetic acid, may be safe and effective, a phase 2 study published in Clinical Infectious Diseases suggests.
Investigators in the United Kingdom designed a trial to compare lactic acid gel and metronidazole, and the findings published in the Trials journal may clarify inconsistent results from prior studies.
Furthermore, preclinical research in Pathogens and Disease has identified cationic amphiphiles that might help fight the biofilm that is formed with Gardnerella vaginalis in patients with bacterial vaginosis, Dr. Birenbaum said.
Finally, an exploratory study in Israel published in Nature Medicine evaluated vaginal microbiome transplants in five patients, three of whom required repeat transplantation. Four patients had long-term remission, and one had a reduction in symptoms
Dr. Birenbaum is a reviewer for UpToDate. Dr. Rasmussen had no relevant disclosures.
Limited data are available to guide treatment of recurrent bacterial vaginosis, but behavioral changes and switching between approved medication regimens may help, according to a presenter at the virtual conference on diseases of the vulva and vagina, hosted by the International Society for the Study of Vulvovaginal Disease.
Investigational treatments – such as a live biotherapeutic product delivered vaginally or vaginal microbiome transplantation – could someday be additional options if they prove safe and effective. said Debra L. Birenbaum, MD, assistant professor of obstetrics and gynecology at Dartmouth-Hitchcock Medical Center in Lebanon, N.H.
As for home remedies, Dr. Birenbaum and another presenter at the conference, Cynthia Rasmussen, MD, urged caution during a panel discussion.
“I think the vagina knows its business, and the more you mess with it, the more you invite trouble,” said Dr. Rasmussen, director emerita of vulvovaginal services at Atrius Health in Burlington, Mass. For instance, tea tree oil, often cited as a home remedy, can be an allergen and very irritating.
“I want to know what women are using, but I try and dissuade them,” said Dr. Birenbaum. “I have to be careful what I say, because you’ll antagonize patients” if you come out strongly against home treatments. “I try to encourage them not to go by things they read on the Internet, because I think that’s where many people are finding their home remedies.”
When counseling patients, an analogy shared during the meeting – the vagina is a self-cleaning oven – may help get the point across. “I love the comment,” Dr. Birenbaum said. “I’ve never used that before. I’m going to start saying that.”
Possible causes and risk factors
Bacterial vaginosis, also known as vaginal dysbiosis, is the most common cause of discharge in women of reproductive age worldwide. Growth of a biofilm may cause the condition, which is characterized by a shift in vaginal flora from a Lactobacilli-dominant environment to one of other bacterial types.
Risk factors include douching, smoking, sex with an uncircumcised partner, and having multiple sexual partners. Bacterial vaginosis may be associated with various complications and infections, including increased risk of preterm delivery, postpartum endometritis, postabortal infection, Trichomonas, chlamydia, and HIV.
Unlike recurrent yeast, which is characterized by four or more episodes per year, recurrent bacterial vaginosis has no official criteria, Dr. Birenbaum said. However, recurrence of bacterial vaginosis “is extremely common,” she said. “Up to 30% of women with [bacterial vaginosis] may recur within 3 months, and up to 50% after 12 months.”
Lifestyle changes and treatments
Recommendations to use condoms, stop smoking, and not douche are important.
In addition, 11 treatment regimens for four drugs – metronidazole, clindamycin, tinidazole, or secnidazole – are available for the treatment of bacterial vaginosis. For recurrent cases, adjusting and switching between the drugs and modes of delivery may help. If a patient started with vaginal gel, they can try an oral medication, or vice versa.
“There’s very little data to guide the optimal therapy for this,” Dr. Birenbaum said. “All of this is worth a try to see if you can beat this before this becomes an ongoing issue.”
As an example of one possible regimen for recurrent bacterial vaginosis, Dr. Birenbaum suggested that a patient could complete a 2- to 4-week course of oral metronidazole instead of the usual 1-week course. The regimen could incorporate boric acid vaginal suppositories 600 mg nightly for 21 days, followed by metronidazole gel 0.75% (one applicator twice per week for 6 months).
New therapies may be on the horizon
In a randomized, double-blind, phase 2b trial published in the New England Journal of Medicine that included more than 220 participants, patients who received an investigational product containing Lactobacillus crispatus CTV-05 (Lactin-V) were less likely to have recurrent bacterial vaginosis at 12 weeks, compared with those who received placebo (30% vs. 45%).
A product in development known as TOL-463, a boric acid–based vaginal anti-infective enhanced with ethylenediaminetetraacetic acid, may be safe and effective, a phase 2 study published in Clinical Infectious Diseases suggests.
Investigators in the United Kingdom designed a trial to compare lactic acid gel and metronidazole, and the findings published in the Trials journal may clarify inconsistent results from prior studies.
Furthermore, preclinical research in Pathogens and Disease has identified cationic amphiphiles that might help fight the biofilm that is formed with Gardnerella vaginalis in patients with bacterial vaginosis, Dr. Birenbaum said.
Finally, an exploratory study in Israel published in Nature Medicine evaluated vaginal microbiome transplants in five patients, three of whom required repeat transplantation. Four patients had long-term remission, and one had a reduction in symptoms
Dr. Birenbaum is a reviewer for UpToDate. Dr. Rasmussen had no relevant disclosures.
Limited data are available to guide treatment of recurrent bacterial vaginosis, but behavioral changes and switching between approved medication regimens may help, according to a presenter at the virtual conference on diseases of the vulva and vagina, hosted by the International Society for the Study of Vulvovaginal Disease.
Investigational treatments – such as a live biotherapeutic product delivered vaginally or vaginal microbiome transplantation – could someday be additional options if they prove safe and effective. said Debra L. Birenbaum, MD, assistant professor of obstetrics and gynecology at Dartmouth-Hitchcock Medical Center in Lebanon, N.H.
As for home remedies, Dr. Birenbaum and another presenter at the conference, Cynthia Rasmussen, MD, urged caution during a panel discussion.
“I think the vagina knows its business, and the more you mess with it, the more you invite trouble,” said Dr. Rasmussen, director emerita of vulvovaginal services at Atrius Health in Burlington, Mass. For instance, tea tree oil, often cited as a home remedy, can be an allergen and very irritating.
“I want to know what women are using, but I try and dissuade them,” said Dr. Birenbaum. “I have to be careful what I say, because you’ll antagonize patients” if you come out strongly against home treatments. “I try to encourage them not to go by things they read on the Internet, because I think that’s where many people are finding their home remedies.”
When counseling patients, an analogy shared during the meeting – the vagina is a self-cleaning oven – may help get the point across. “I love the comment,” Dr. Birenbaum said. “I’ve never used that before. I’m going to start saying that.”
Possible causes and risk factors
Bacterial vaginosis, also known as vaginal dysbiosis, is the most common cause of discharge in women of reproductive age worldwide. Growth of a biofilm may cause the condition, which is characterized by a shift in vaginal flora from a Lactobacilli-dominant environment to one of other bacterial types.
Risk factors include douching, smoking, sex with an uncircumcised partner, and having multiple sexual partners. Bacterial vaginosis may be associated with various complications and infections, including increased risk of preterm delivery, postpartum endometritis, postabortal infection, Trichomonas, chlamydia, and HIV.
Unlike recurrent yeast, which is characterized by four or more episodes per year, recurrent bacterial vaginosis has no official criteria, Dr. Birenbaum said. However, recurrence of bacterial vaginosis “is extremely common,” she said. “Up to 30% of women with [bacterial vaginosis] may recur within 3 months, and up to 50% after 12 months.”
Lifestyle changes and treatments
Recommendations to use condoms, stop smoking, and not douche are important.
In addition, 11 treatment regimens for four drugs – metronidazole, clindamycin, tinidazole, or secnidazole – are available for the treatment of bacterial vaginosis. For recurrent cases, adjusting and switching between the drugs and modes of delivery may help. If a patient started with vaginal gel, they can try an oral medication, or vice versa.
“There’s very little data to guide the optimal therapy for this,” Dr. Birenbaum said. “All of this is worth a try to see if you can beat this before this becomes an ongoing issue.”
As an example of one possible regimen for recurrent bacterial vaginosis, Dr. Birenbaum suggested that a patient could complete a 2- to 4-week course of oral metronidazole instead of the usual 1-week course. The regimen could incorporate boric acid vaginal suppositories 600 mg nightly for 21 days, followed by metronidazole gel 0.75% (one applicator twice per week for 6 months).
New therapies may be on the horizon
In a randomized, double-blind, phase 2b trial published in the New England Journal of Medicine that included more than 220 participants, patients who received an investigational product containing Lactobacillus crispatus CTV-05 (Lactin-V) were less likely to have recurrent bacterial vaginosis at 12 weeks, compared with those who received placebo (30% vs. 45%).
A product in development known as TOL-463, a boric acid–based vaginal anti-infective enhanced with ethylenediaminetetraacetic acid, may be safe and effective, a phase 2 study published in Clinical Infectious Diseases suggests.
Investigators in the United Kingdom designed a trial to compare lactic acid gel and metronidazole, and the findings published in the Trials journal may clarify inconsistent results from prior studies.
Furthermore, preclinical research in Pathogens and Disease has identified cationic amphiphiles that might help fight the biofilm that is formed with Gardnerella vaginalis in patients with bacterial vaginosis, Dr. Birenbaum said.
Finally, an exploratory study in Israel published in Nature Medicine evaluated vaginal microbiome transplants in five patients, three of whom required repeat transplantation. Four patients had long-term remission, and one had a reduction in symptoms
Dr. Birenbaum is a reviewer for UpToDate. Dr. Rasmussen had no relevant disclosures.
FROM THE ISSVD BIENNIAL CONFERENCE
PRGLAC recommendations
1. Include and integrate pregnant women and lactating women in the clinical research agenda.
2. Increase the quantity, quality, and timeliness of research on safety and efficacy of therapeutic products used by pregnant women and lactating women.
3. Expand the workforce of clinicians and research investigators with expertise in obstetric and lactation pharmacology and therapeutics.
4. Remove regulatory barriers to research in pregnant women.
5. Create a public awareness campaign to engage the public and health care providers in research on pregnant women and lactating women.
6. Develop and implement evidence-based communication strategies with health care providers on information relevant to research on pregnant women and lactating women.
7. Develop separate programs to study therapeutic products used off patent in pregnant women and lactating women using the National Institute of Health Best Pharmaceuticals for Children Act (BPCA) as a model.
8. Reduce liability to facilitate an evidence base for new therapeutic products that may be used by women who are or may become pregnant and by lactating women.
9. Implement a proactive approach to protocol development and study design to include pregnant women and lactating women in clinical research.
10. Develop programs to drive discovery and development of therapeutics and new therapeutic products for conditions specific to pregnant women and lactating women.
11. Utilize and improve existing resources for data to inform the evidence and provide a foundation for research on pregnant women and lactating women.
12. Leverage established and support new infrastructures/collaborations to perform research in pregnant women and lactating women.
13. Optimize registries for pregnancy and lactation.
14. The Department of Health & Human Services Secretary should consider exercising the authority provided in law to extend the PRGLAC Task Force when its charter expires in March 2019.
15. Establish an Advisory Committee to monitor and report on implementation of recommendations, updating regulations, and guidance, as applicable, regarding the inclusion of pregnant women and lactating women in clinical research.
Source: Task Force on Research Specific to Pregnant Women and Lactating Women; Report to Secretary, Health and Human Services, Congress, September 2018
1. Include and integrate pregnant women and lactating women in the clinical research agenda.
2. Increase the quantity, quality, and timeliness of research on safety and efficacy of therapeutic products used by pregnant women and lactating women.
3. Expand the workforce of clinicians and research investigators with expertise in obstetric and lactation pharmacology and therapeutics.
4. Remove regulatory barriers to research in pregnant women.
5. Create a public awareness campaign to engage the public and health care providers in research on pregnant women and lactating women.
6. Develop and implement evidence-based communication strategies with health care providers on information relevant to research on pregnant women and lactating women.
7. Develop separate programs to study therapeutic products used off patent in pregnant women and lactating women using the National Institute of Health Best Pharmaceuticals for Children Act (BPCA) as a model.
8. Reduce liability to facilitate an evidence base for new therapeutic products that may be used by women who are or may become pregnant and by lactating women.
9. Implement a proactive approach to protocol development and study design to include pregnant women and lactating women in clinical research.
10. Develop programs to drive discovery and development of therapeutics and new therapeutic products for conditions specific to pregnant women and lactating women.
11. Utilize and improve existing resources for data to inform the evidence and provide a foundation for research on pregnant women and lactating women.
12. Leverage established and support new infrastructures/collaborations to perform research in pregnant women and lactating women.
13. Optimize registries for pregnancy and lactation.
14. The Department of Health & Human Services Secretary should consider exercising the authority provided in law to extend the PRGLAC Task Force when its charter expires in March 2019.
15. Establish an Advisory Committee to monitor and report on implementation of recommendations, updating regulations, and guidance, as applicable, regarding the inclusion of pregnant women and lactating women in clinical research.
Source: Task Force on Research Specific to Pregnant Women and Lactating Women; Report to Secretary, Health and Human Services, Congress, September 2018
1. Include and integrate pregnant women and lactating women in the clinical research agenda.
2. Increase the quantity, quality, and timeliness of research on safety and efficacy of therapeutic products used by pregnant women and lactating women.
3. Expand the workforce of clinicians and research investigators with expertise in obstetric and lactation pharmacology and therapeutics.
4. Remove regulatory barriers to research in pregnant women.
5. Create a public awareness campaign to engage the public and health care providers in research on pregnant women and lactating women.
6. Develop and implement evidence-based communication strategies with health care providers on information relevant to research on pregnant women and lactating women.
7. Develop separate programs to study therapeutic products used off patent in pregnant women and lactating women using the National Institute of Health Best Pharmaceuticals for Children Act (BPCA) as a model.
8. Reduce liability to facilitate an evidence base for new therapeutic products that may be used by women who are or may become pregnant and by lactating women.
9. Implement a proactive approach to protocol development and study design to include pregnant women and lactating women in clinical research.
10. Develop programs to drive discovery and development of therapeutics and new therapeutic products for conditions specific to pregnant women and lactating women.
11. Utilize and improve existing resources for data to inform the evidence and provide a foundation for research on pregnant women and lactating women.
12. Leverage established and support new infrastructures/collaborations to perform research in pregnant women and lactating women.
13. Optimize registries for pregnancy and lactation.
14. The Department of Health & Human Services Secretary should consider exercising the authority provided in law to extend the PRGLAC Task Force when its charter expires in March 2019.
15. Establish an Advisory Committee to monitor and report on implementation of recommendations, updating regulations, and guidance, as applicable, regarding the inclusion of pregnant women and lactating women in clinical research.
Source: Task Force on Research Specific to Pregnant Women and Lactating Women; Report to Secretary, Health and Human Services, Congress, September 2018
Safe, effective therapies: Establishing a path forward
I have had friends and colleagues visibly shrink away when I say that my work involves the study of medication safety in pregnancy. “Yikes! I would never let my daughter participate in a clinical study if she was pregnant!” I hear. It’s an interesting response. Understandably protective of a loved one, except that the loved one is an adult woman who presumably can make her own choices. And the response reveals an assumption that medications are tested in all populations before approval for market. Sadly, the response is ill-informed given that pregnant women are still excluded from most if not all clinical research. My work, by the way, is focused on postapproval studies.
Translating the above response to a larger picture, health care providers and pharmaceutical manufacturers also have their concerns about pregnant women and lactating women participating in clinical research. Along with the patient and her loved ones, all parties’ concerns are valid. However, there is a harsh reality: According to a study in the American Journal of Obstetrics & Gynecology, an estimated 50% of U.S. women take one or more prescription medications during pregnancy. Once marketed, therapies are prescribed to pregnant women, knowingly and unknowingly, and without evidence-based knowledge of their safety. If postapproval safety studies are undertaken, decades may pass as data accrue and before results become available. In general, even less is known about the safety of medications in breastmilk.
Without a path forward that includes pregnant women and lactating women in clinical research, we will remain without timely knowledge of medication safety. Further, our understanding of efficacy will be based on clinical studies of nonpregnant women. Recognizing the need for this information, the Task Force on Research Specific to Pregnant Women and Lactating Women (PRGLAC) was convened in 2017 and tasked with determining this path forward.
The PRGLAC was established by the 21st Century Cures Act, a law designed to help speed up medical product development. Managed by the National Institutes of Health, the PRGLAC is made up of representatives of all federal agencies with responsibilities for women’s health and research, as well as clinicians, industry experts, and other experts. The PRGLAC’s work has been conducted in two phases.
In Phase I, PRGLAC was charged with identifying gaps in knowledge and research regarding safe and effective therapies for pregnant women and lactating women. The Task Force conducted four public meetings in 2017 and 2018, and submitted their conclusions to Congress and the Secretary of Health & Human Services in a publicly available report. The report provides 15 specific recommendations, several of which are directly relevant to obstetricians: No. 3 recommends expanding the workforce of clinicians and research investigators with expertise in obstetric and lactation pharmacology, No. 6 recommends the development and implementation of evidence-based communication strategies with health care providers, and No. 13 recommends optimization of registries for pregnancy and lactation. Obstetricians can make a positive contribution to accruing medication safety data by being aware of pregnancy registries and indicating their availability to eligible patients.
In the spring of 2019, the PRGLAC reconvened with a 2-year mandate and a new charge for Phase II: to develop plans for implementing the recommendations laid out in the Phase I report. Four working groups (WGs) were identified to address the recommendations of the report: WG1 Research and Training, WG2 Regulatory, WG3 Communication and Registries, and WG4 Discovery. The four groups have deliberated, and a new report is being finalized. The PRGLAC’s efforts provide a fresh conversation to address long-standing issues to provide evidence-based information for the treatment of pregnant and lactating women. Once available, the final report will be posted on the PRGLAC website.
The recommendations in this report, when implemented, are directly relevant to patient care and clinician training and will provide a path forward for the inclusion of pregnant and lactating women in clinical research or a firm justification for their exclusion.
Dr. Hardy is a consultant on global maternal-child health and pharmacoepidemiology. She also represents the Society for Birth Defects Research and Prevention and the Organization of Teratology Information Specialists at PRGLAC meetings. Dr. Hardy disclosed she has worked with multiple pharmaceutical manufacturers regarding medication safety studies in pregnancy, most recently Biohaven. Email her at [email protected].
I have had friends and colleagues visibly shrink away when I say that my work involves the study of medication safety in pregnancy. “Yikes! I would never let my daughter participate in a clinical study if she was pregnant!” I hear. It’s an interesting response. Understandably protective of a loved one, except that the loved one is an adult woman who presumably can make her own choices. And the response reveals an assumption that medications are tested in all populations before approval for market. Sadly, the response is ill-informed given that pregnant women are still excluded from most if not all clinical research. My work, by the way, is focused on postapproval studies.
Translating the above response to a larger picture, health care providers and pharmaceutical manufacturers also have their concerns about pregnant women and lactating women participating in clinical research. Along with the patient and her loved ones, all parties’ concerns are valid. However, there is a harsh reality: According to a study in the American Journal of Obstetrics & Gynecology, an estimated 50% of U.S. women take one or more prescription medications during pregnancy. Once marketed, therapies are prescribed to pregnant women, knowingly and unknowingly, and without evidence-based knowledge of their safety. If postapproval safety studies are undertaken, decades may pass as data accrue and before results become available. In general, even less is known about the safety of medications in breastmilk.
Without a path forward that includes pregnant women and lactating women in clinical research, we will remain without timely knowledge of medication safety. Further, our understanding of efficacy will be based on clinical studies of nonpregnant women. Recognizing the need for this information, the Task Force on Research Specific to Pregnant Women and Lactating Women (PRGLAC) was convened in 2017 and tasked with determining this path forward.
The PRGLAC was established by the 21st Century Cures Act, a law designed to help speed up medical product development. Managed by the National Institutes of Health, the PRGLAC is made up of representatives of all federal agencies with responsibilities for women’s health and research, as well as clinicians, industry experts, and other experts. The PRGLAC’s work has been conducted in two phases.
In Phase I, PRGLAC was charged with identifying gaps in knowledge and research regarding safe and effective therapies for pregnant women and lactating women. The Task Force conducted four public meetings in 2017 and 2018, and submitted their conclusions to Congress and the Secretary of Health & Human Services in a publicly available report. The report provides 15 specific recommendations, several of which are directly relevant to obstetricians: No. 3 recommends expanding the workforce of clinicians and research investigators with expertise in obstetric and lactation pharmacology, No. 6 recommends the development and implementation of evidence-based communication strategies with health care providers, and No. 13 recommends optimization of registries for pregnancy and lactation. Obstetricians can make a positive contribution to accruing medication safety data by being aware of pregnancy registries and indicating their availability to eligible patients.
In the spring of 2019, the PRGLAC reconvened with a 2-year mandate and a new charge for Phase II: to develop plans for implementing the recommendations laid out in the Phase I report. Four working groups (WGs) were identified to address the recommendations of the report: WG1 Research and Training, WG2 Regulatory, WG3 Communication and Registries, and WG4 Discovery. The four groups have deliberated, and a new report is being finalized. The PRGLAC’s efforts provide a fresh conversation to address long-standing issues to provide evidence-based information for the treatment of pregnant and lactating women. Once available, the final report will be posted on the PRGLAC website.
The recommendations in this report, when implemented, are directly relevant to patient care and clinician training and will provide a path forward for the inclusion of pregnant and lactating women in clinical research or a firm justification for their exclusion.
Dr. Hardy is a consultant on global maternal-child health and pharmacoepidemiology. She also represents the Society for Birth Defects Research and Prevention and the Organization of Teratology Information Specialists at PRGLAC meetings. Dr. Hardy disclosed she has worked with multiple pharmaceutical manufacturers regarding medication safety studies in pregnancy, most recently Biohaven. Email her at [email protected].
I have had friends and colleagues visibly shrink away when I say that my work involves the study of medication safety in pregnancy. “Yikes! I would never let my daughter participate in a clinical study if she was pregnant!” I hear. It’s an interesting response. Understandably protective of a loved one, except that the loved one is an adult woman who presumably can make her own choices. And the response reveals an assumption that medications are tested in all populations before approval for market. Sadly, the response is ill-informed given that pregnant women are still excluded from most if not all clinical research. My work, by the way, is focused on postapproval studies.
Translating the above response to a larger picture, health care providers and pharmaceutical manufacturers also have their concerns about pregnant women and lactating women participating in clinical research. Along with the patient and her loved ones, all parties’ concerns are valid. However, there is a harsh reality: According to a study in the American Journal of Obstetrics & Gynecology, an estimated 50% of U.S. women take one or more prescription medications during pregnancy. Once marketed, therapies are prescribed to pregnant women, knowingly and unknowingly, and without evidence-based knowledge of their safety. If postapproval safety studies are undertaken, decades may pass as data accrue and before results become available. In general, even less is known about the safety of medications in breastmilk.
Without a path forward that includes pregnant women and lactating women in clinical research, we will remain without timely knowledge of medication safety. Further, our understanding of efficacy will be based on clinical studies of nonpregnant women. Recognizing the need for this information, the Task Force on Research Specific to Pregnant Women and Lactating Women (PRGLAC) was convened in 2017 and tasked with determining this path forward.
The PRGLAC was established by the 21st Century Cures Act, a law designed to help speed up medical product development. Managed by the National Institutes of Health, the PRGLAC is made up of representatives of all federal agencies with responsibilities for women’s health and research, as well as clinicians, industry experts, and other experts. The PRGLAC’s work has been conducted in two phases.
In Phase I, PRGLAC was charged with identifying gaps in knowledge and research regarding safe and effective therapies for pregnant women and lactating women. The Task Force conducted four public meetings in 2017 and 2018, and submitted their conclusions to Congress and the Secretary of Health & Human Services in a publicly available report. The report provides 15 specific recommendations, several of which are directly relevant to obstetricians: No. 3 recommends expanding the workforce of clinicians and research investigators with expertise in obstetric and lactation pharmacology, No. 6 recommends the development and implementation of evidence-based communication strategies with health care providers, and No. 13 recommends optimization of registries for pregnancy and lactation. Obstetricians can make a positive contribution to accruing medication safety data by being aware of pregnancy registries and indicating their availability to eligible patients.
In the spring of 2019, the PRGLAC reconvened with a 2-year mandate and a new charge for Phase II: to develop plans for implementing the recommendations laid out in the Phase I report. Four working groups (WGs) were identified to address the recommendations of the report: WG1 Research and Training, WG2 Regulatory, WG3 Communication and Registries, and WG4 Discovery. The four groups have deliberated, and a new report is being finalized. The PRGLAC’s efforts provide a fresh conversation to address long-standing issues to provide evidence-based information for the treatment of pregnant and lactating women. Once available, the final report will be posted on the PRGLAC website.
The recommendations in this report, when implemented, are directly relevant to patient care and clinician training and will provide a path forward for the inclusion of pregnant and lactating women in clinical research or a firm justification for their exclusion.
Dr. Hardy is a consultant on global maternal-child health and pharmacoepidemiology. She also represents the Society for Birth Defects Research and Prevention and the Organization of Teratology Information Specialists at PRGLAC meetings. Dr. Hardy disclosed she has worked with multiple pharmaceutical manufacturers regarding medication safety studies in pregnancy, most recently Biohaven. Email her at [email protected].
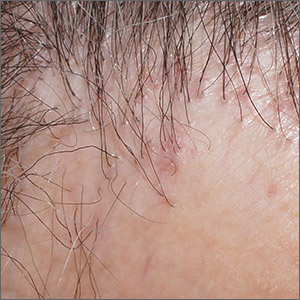
Hair loss and scalp papules
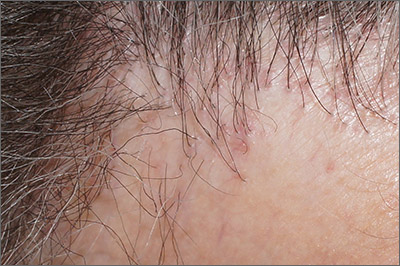
The punch biopsies were consistent with lichen planopilaris, an idiopathic, immune-mediated scarring alopecia that largely affects women between the ages of 40 and 70 years. In this variant of lichen planus, T cells target hair bulbs and cause destruction with scarring and permanent hair loss. Distribution may be patchy or may be more concentrated on the crown or involve the frontal scalp—a subtype called frontal fibrosing alopecia. Early recognition and intervention may save hair follicles and minimize disease severity.
The differential diagnosis includes traction alopecia, discoid lupus erythematosus, alopecia areata, centrifugal cicatricial alopecia, and folliculitis decalvans. The diagnosis may be confirmed with a scalp biopsy of actively inflamed follicles. Biopsy of scarred areas is likely to be nonspecific and unhelpful.
Treatment is targeted at slowing progression and symptom management. First-line therapy often includes potent corticosteroids (intralesional, topical, or systemic). Longer courses of steroid-sparing agents may be considered, including hydroxychloroquine, tacrolimus, ciclosporin, methotrexate, or acitretin. Hair styling and coloring, as well as hairpieces, often are used to conceal patches of hair loss. Hair transplantation is expensive but can be used to increase hair density in scarred areas once disease is controlled.
In this case, the patient was started on clobetasol solution 0.05% to be applied nightly to affected areas of the scalp. This treatment helped with the itching, but the inflammation and hair loss continued to worsen after 2 months. At that point, hydroxychloroquine 200 mg bid was added to the regimen, and hair loss and associated symptoms stopped. The patient remained on this therapy for 16 months. The hydroxychloroquine was then stopped, and the patient was advised to use the topical clobetasol, as needed.
Text courtesy of Tristan Reynolds, DO, Maine Dartmouth Family Medicine Residency, and Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
Errichetti E, Figini M, Croatto M, et al. Therapeutic management of classic lichen planopilaris: a systematic review. Clin Cosmet Investig Dermatol. 2018;11:91-102.

The punch biopsies were consistent with lichen planopilaris, an idiopathic, immune-mediated scarring alopecia that largely affects women between the ages of 40 and 70 years. In this variant of lichen planus, T cells target hair bulbs and cause destruction with scarring and permanent hair loss. Distribution may be patchy or may be more concentrated on the crown or involve the frontal scalp—a subtype called frontal fibrosing alopecia. Early recognition and intervention may save hair follicles and minimize disease severity.
The differential diagnosis includes traction alopecia, discoid lupus erythematosus, alopecia areata, centrifugal cicatricial alopecia, and folliculitis decalvans. The diagnosis may be confirmed with a scalp biopsy of actively inflamed follicles. Biopsy of scarred areas is likely to be nonspecific and unhelpful.
Treatment is targeted at slowing progression and symptom management. First-line therapy often includes potent corticosteroids (intralesional, topical, or systemic). Longer courses of steroid-sparing agents may be considered, including hydroxychloroquine, tacrolimus, ciclosporin, methotrexate, or acitretin. Hair styling and coloring, as well as hairpieces, often are used to conceal patches of hair loss. Hair transplantation is expensive but can be used to increase hair density in scarred areas once disease is controlled.
In this case, the patient was started on clobetasol solution 0.05% to be applied nightly to affected areas of the scalp. This treatment helped with the itching, but the inflammation and hair loss continued to worsen after 2 months. At that point, hydroxychloroquine 200 mg bid was added to the regimen, and hair loss and associated symptoms stopped. The patient remained on this therapy for 16 months. The hydroxychloroquine was then stopped, and the patient was advised to use the topical clobetasol, as needed.
Text courtesy of Tristan Reynolds, DO, Maine Dartmouth Family Medicine Residency, and Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

The punch biopsies were consistent with lichen planopilaris, an idiopathic, immune-mediated scarring alopecia that largely affects women between the ages of 40 and 70 years. In this variant of lichen planus, T cells target hair bulbs and cause destruction with scarring and permanent hair loss. Distribution may be patchy or may be more concentrated on the crown or involve the frontal scalp—a subtype called frontal fibrosing alopecia. Early recognition and intervention may save hair follicles and minimize disease severity.
The differential diagnosis includes traction alopecia, discoid lupus erythematosus, alopecia areata, centrifugal cicatricial alopecia, and folliculitis decalvans. The diagnosis may be confirmed with a scalp biopsy of actively inflamed follicles. Biopsy of scarred areas is likely to be nonspecific and unhelpful.
Treatment is targeted at slowing progression and symptom management. First-line therapy often includes potent corticosteroids (intralesional, topical, or systemic). Longer courses of steroid-sparing agents may be considered, including hydroxychloroquine, tacrolimus, ciclosporin, methotrexate, or acitretin. Hair styling and coloring, as well as hairpieces, often are used to conceal patches of hair loss. Hair transplantation is expensive but can be used to increase hair density in scarred areas once disease is controlled.
In this case, the patient was started on clobetasol solution 0.05% to be applied nightly to affected areas of the scalp. This treatment helped with the itching, but the inflammation and hair loss continued to worsen after 2 months. At that point, hydroxychloroquine 200 mg bid was added to the regimen, and hair loss and associated symptoms stopped. The patient remained on this therapy for 16 months. The hydroxychloroquine was then stopped, and the patient was advised to use the topical clobetasol, as needed.
Text courtesy of Tristan Reynolds, DO, Maine Dartmouth Family Medicine Residency, and Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
Errichetti E, Figini M, Croatto M, et al. Therapeutic management of classic lichen planopilaris: a systematic review. Clin Cosmet Investig Dermatol. 2018;11:91-102.
Errichetti E, Figini M, Croatto M, et al. Therapeutic management of classic lichen planopilaris: a systematic review. Clin Cosmet Investig Dermatol. 2018;11:91-102.
Study offers reassurance to postmenopausal women taking hormone therapy
References
- Chlebowski RT, Anderson GL, Aragaki AK, et al. Association of menopausal hormone therapy with breast cancer incidence and mortality during long-term follow-up of the Women’s Health Initiative randomized clinical trials. JAMA. 2020;32:369-380.
- The North American Menopause Society. Menopause Guidebook. 8th ed. www.menopause.org/publications/consumer-publications/-em-menopause-guidebook-em-8th-edition. Accessed September 25, 2020.
- US Preventive Services Task Force; Grossman DC, Curry SJ, Owens DK, et al. Hormone therapy for the primary prevention of chronic conditions in postmenopausal women: US Preventive Services Task Force Recommendation Statement. JAMA. 2017;318:2224-2233.
References
- Chlebowski RT, Anderson GL, Aragaki AK, et al. Association of menopausal hormone therapy with breast cancer incidence and mortality during long-term follow-up of the Women’s Health Initiative randomized clinical trials. JAMA. 2020;32:369-380.
- The North American Menopause Society. Menopause Guidebook. 8th ed. www.menopause.org/publications/consumer-publications/-em-menopause-guidebook-em-8th-edition. Accessed September 25, 2020.
- US Preventive Services Task Force; Grossman DC, Curry SJ, Owens DK, et al. Hormone therapy for the primary prevention of chronic conditions in postmenopausal women: US Preventive Services Task Force Recommendation Statement. JAMA. 2017;318:2224-2233.
References
- Chlebowski RT, Anderson GL, Aragaki AK, et al. Association of menopausal hormone therapy with breast cancer incidence and mortality during long-term follow-up of the Women’s Health Initiative randomized clinical trials. JAMA. 2020;32:369-380.
- The North American Menopause Society. Menopause Guidebook. 8th ed. www.menopause.org/publications/consumer-publications/-em-menopause-guidebook-em-8th-edition. Accessed September 25, 2020.
- US Preventive Services Task Force; Grossman DC, Curry SJ, Owens DK, et al. Hormone therapy for the primary prevention of chronic conditions in postmenopausal women: US Preventive Services Task Force Recommendation Statement. JAMA. 2017;318:2224-2233.
Three-step approach may help relieve one of the itchiest vulvar conditions
A three-step approach may help relieve itch in patients with lichen simplex chronicus, “one of the itchiest conditions that we ever see on the vulva,” an expert advised at the virtual conference on diseases of the vulva and vagina, hosted by the International Society for the Study of Vulvovaginal Disease.
For some patients, such as those with excessive sweating or underlying psoriasis, seeing a dermatologist may be beneficial, physicians at the meeting suggested.
Treatment should aim to optimize epithelial barrier function, reduce inflammation, and stop scratching, Lynette Margesson, MD, said in a lecture at the biennial meeting, which is held by the International Society for the Study of Vulvovaginal Disease (ISSVD). “With this condition, please look always for more than one problem.”
said Dr. Margesson, an obstetrician and gynecologist at Geisel School of Medicine at Dartmouth in Hanover, N.H. “It is because of chronic rubbing and scratching on top of something else.”
It may develop on top of atopic dermatitis, psoriasis, or contact dermatitis, as well as infection, lichen sclerosus, lichen planus, or neoplasia.
Lichen simplex chronicus is characterized by years of relentless itching, and patients may wake up at night scratching. The skin looks and feels leathery, and the condition can be localized or around the entire vulva. Heat, humidity, stress, and irritants may exacerbate the condition.
Patients often try to wash the rash away with scrubbers and cleansers, which only makes it worse, Dr. Margesson said.
To get patients better, improve barrier function, such as by controlling infections, reducing sweating, avoiding irritants, and stopping excessive hygiene. Immediate therapy may include soaks, cool compresses, and ointments.
A superpotent steroid taper (e.g., clobetasol 0.05% ointment), a prednisone taper, or intramuscular triamcinolone may reduce inflammation. Dr. Margesson usually uses clobetasol, although this treatment or halobetasol can burn if patients have open skin. In such cases, she uses prednisone or intramuscular triamcinolone.
Sedating medications may help patients stop scratching, especially at night. Hydroxyzine, doxepin, or amitriptyline 2-3 hours before bedtime can help. Scratching can be a form of obsessive-compulsive disorder, and a small dose of citalopram may help during the day. Patients with significant psychological factors can be difficult to manage and tend to relapse easily, Dr. Margesson said.
If lichen simplex chronicus recurs, test for infections and allergies. “Maybe they need a mild corticosteroid all the time, like 2.5% hydrocortisone to alternate with your superpotent steroid so you can use it longer without thinning the skin,” she suggested.
Although Dr. Margesson does not often treat hyperhidrosis, addressing excessive sweating can make a big difference for patients, she said.
If a gynecologist identifies a patient who may benefit from treatment of hyperhidrosis but has limited experience with medications for this condition, it might make sense to work with a dermatologist, Aruna Venkatesan, MD, chief of dermatology and director of the genital dermatology clinic at Santa Clara Valley Medical Center in San Jose, Calif., suggested during a panel discussion. Most dermatologists treat hyperhidrosis regularly, she said.
Dermatologists also may help treat patients with psoriasis who need systemic medication, Dr. Margesson said.
“In terms of ... doing the lab monitoring and knowing what side effects to look out for, your colleagues who use these medicines more are going to be more comfortable with that,” Dr. Venkatesan said. They also may have more experience navigating insurance denials to obtain a therapy. “Don’t think you are passing the buck to someone else. Sometimes that is the right thing to do, to get that help from someone else.”
Dr. Margesson is an author for UpToDate. Dr. Venkatesan had no conflicts of interest.
A three-step approach may help relieve itch in patients with lichen simplex chronicus, “one of the itchiest conditions that we ever see on the vulva,” an expert advised at the virtual conference on diseases of the vulva and vagina, hosted by the International Society for the Study of Vulvovaginal Disease.
For some patients, such as those with excessive sweating or underlying psoriasis, seeing a dermatologist may be beneficial, physicians at the meeting suggested.
Treatment should aim to optimize epithelial barrier function, reduce inflammation, and stop scratching, Lynette Margesson, MD, said in a lecture at the biennial meeting, which is held by the International Society for the Study of Vulvovaginal Disease (ISSVD). “With this condition, please look always for more than one problem.”
said Dr. Margesson, an obstetrician and gynecologist at Geisel School of Medicine at Dartmouth in Hanover, N.H. “It is because of chronic rubbing and scratching on top of something else.”
It may develop on top of atopic dermatitis, psoriasis, or contact dermatitis, as well as infection, lichen sclerosus, lichen planus, or neoplasia.
Lichen simplex chronicus is characterized by years of relentless itching, and patients may wake up at night scratching. The skin looks and feels leathery, and the condition can be localized or around the entire vulva. Heat, humidity, stress, and irritants may exacerbate the condition.
Patients often try to wash the rash away with scrubbers and cleansers, which only makes it worse, Dr. Margesson said.
To get patients better, improve barrier function, such as by controlling infections, reducing sweating, avoiding irritants, and stopping excessive hygiene. Immediate therapy may include soaks, cool compresses, and ointments.
A superpotent steroid taper (e.g., clobetasol 0.05% ointment), a prednisone taper, or intramuscular triamcinolone may reduce inflammation. Dr. Margesson usually uses clobetasol, although this treatment or halobetasol can burn if patients have open skin. In such cases, she uses prednisone or intramuscular triamcinolone.
Sedating medications may help patients stop scratching, especially at night. Hydroxyzine, doxepin, or amitriptyline 2-3 hours before bedtime can help. Scratching can be a form of obsessive-compulsive disorder, and a small dose of citalopram may help during the day. Patients with significant psychological factors can be difficult to manage and tend to relapse easily, Dr. Margesson said.
If lichen simplex chronicus recurs, test for infections and allergies. “Maybe they need a mild corticosteroid all the time, like 2.5% hydrocortisone to alternate with your superpotent steroid so you can use it longer without thinning the skin,” she suggested.
Although Dr. Margesson does not often treat hyperhidrosis, addressing excessive sweating can make a big difference for patients, she said.
If a gynecologist identifies a patient who may benefit from treatment of hyperhidrosis but has limited experience with medications for this condition, it might make sense to work with a dermatologist, Aruna Venkatesan, MD, chief of dermatology and director of the genital dermatology clinic at Santa Clara Valley Medical Center in San Jose, Calif., suggested during a panel discussion. Most dermatologists treat hyperhidrosis regularly, she said.
Dermatologists also may help treat patients with psoriasis who need systemic medication, Dr. Margesson said.
“In terms of ... doing the lab monitoring and knowing what side effects to look out for, your colleagues who use these medicines more are going to be more comfortable with that,” Dr. Venkatesan said. They also may have more experience navigating insurance denials to obtain a therapy. “Don’t think you are passing the buck to someone else. Sometimes that is the right thing to do, to get that help from someone else.”
Dr. Margesson is an author for UpToDate. Dr. Venkatesan had no conflicts of interest.
A three-step approach may help relieve itch in patients with lichen simplex chronicus, “one of the itchiest conditions that we ever see on the vulva,” an expert advised at the virtual conference on diseases of the vulva and vagina, hosted by the International Society for the Study of Vulvovaginal Disease.
For some patients, such as those with excessive sweating or underlying psoriasis, seeing a dermatologist may be beneficial, physicians at the meeting suggested.
Treatment should aim to optimize epithelial barrier function, reduce inflammation, and stop scratching, Lynette Margesson, MD, said in a lecture at the biennial meeting, which is held by the International Society for the Study of Vulvovaginal Disease (ISSVD). “With this condition, please look always for more than one problem.”
said Dr. Margesson, an obstetrician and gynecologist at Geisel School of Medicine at Dartmouth in Hanover, N.H. “It is because of chronic rubbing and scratching on top of something else.”
It may develop on top of atopic dermatitis, psoriasis, or contact dermatitis, as well as infection, lichen sclerosus, lichen planus, or neoplasia.
Lichen simplex chronicus is characterized by years of relentless itching, and patients may wake up at night scratching. The skin looks and feels leathery, and the condition can be localized or around the entire vulva. Heat, humidity, stress, and irritants may exacerbate the condition.
Patients often try to wash the rash away with scrubbers and cleansers, which only makes it worse, Dr. Margesson said.
To get patients better, improve barrier function, such as by controlling infections, reducing sweating, avoiding irritants, and stopping excessive hygiene. Immediate therapy may include soaks, cool compresses, and ointments.
A superpotent steroid taper (e.g., clobetasol 0.05% ointment), a prednisone taper, or intramuscular triamcinolone may reduce inflammation. Dr. Margesson usually uses clobetasol, although this treatment or halobetasol can burn if patients have open skin. In such cases, she uses prednisone or intramuscular triamcinolone.
Sedating medications may help patients stop scratching, especially at night. Hydroxyzine, doxepin, or amitriptyline 2-3 hours before bedtime can help. Scratching can be a form of obsessive-compulsive disorder, and a small dose of citalopram may help during the day. Patients with significant psychological factors can be difficult to manage and tend to relapse easily, Dr. Margesson said.
If lichen simplex chronicus recurs, test for infections and allergies. “Maybe they need a mild corticosteroid all the time, like 2.5% hydrocortisone to alternate with your superpotent steroid so you can use it longer without thinning the skin,” she suggested.
Although Dr. Margesson does not often treat hyperhidrosis, addressing excessive sweating can make a big difference for patients, she said.
If a gynecologist identifies a patient who may benefit from treatment of hyperhidrosis but has limited experience with medications for this condition, it might make sense to work with a dermatologist, Aruna Venkatesan, MD, chief of dermatology and director of the genital dermatology clinic at Santa Clara Valley Medical Center in San Jose, Calif., suggested during a panel discussion. Most dermatologists treat hyperhidrosis regularly, she said.
Dermatologists also may help treat patients with psoriasis who need systemic medication, Dr. Margesson said.
“In terms of ... doing the lab monitoring and knowing what side effects to look out for, your colleagues who use these medicines more are going to be more comfortable with that,” Dr. Venkatesan said. They also may have more experience navigating insurance denials to obtain a therapy. “Don’t think you are passing the buck to someone else. Sometimes that is the right thing to do, to get that help from someone else.”
Dr. Margesson is an author for UpToDate. Dr. Venkatesan had no conflicts of interest.
FROM ISSVD BIENNIAL CONFERENCE
Without Ginsburg, judicial threats to the ACA, reproductive rights heighten
On Feb. 27, 2018, I got an email from the Heritage Foundation that alerted me to a news conference that afternoon held by Republican attorneys general of Texas and other states. It was referred to only as a “discussion about the Affordable Care Act lawsuit.”
I sent the following note to my editor: “I’m off to the Hill anyway. I could stop by this. You never know what it might morph into.”
Few people took that case very seriously – barely a handful of reporters attended the news conference. But it has now “morphed into” the latest existential threat to the Affordable Care Act, scheduled for oral arguments at the Supreme Court a week after the general election in November. And with the death of Justice Ruth Bader Ginsburg on Friday, that case could well morph into the threat that brings down the law in its entirety.
Democrats are raising alarms about the future of the law without Ms. Ginsburg. House Speaker Nancy Pelosi, speaking on ABC’s “This Week” Sunday morning, said that part of the strategy by President Trump and Senate Republicans to quickly fill her seat was to help undermine the ACA.
“The president is rushing to make some kind of a decision because … Nov. 10 is when the arguments begin on the Affordable Care Act,” she said. “He doesn’t want to crush the virus. He wants to crush the Affordable Care Act.”
Ms. Ginsburg’s death could throw an already chaotic general election campaign during a pandemic into even more turmoil.
Let’s take them one at a time.
The ACA under fire – again
The GOP attorneys general argued in February 2018 that the Republican-sponsored tax cut bill Congress passed two months earlier had rendered the ACA unconstitutional by reducing to zero the ACA’s penalty for not having insurance. They based their argument on Chief Justice John Roberts’ 2012 conclusion that the ACA was valid, interpreting that penalty as a constitutionally appropriate tax.
Most legal scholars, including several who challenged the law before the Supreme Court in 2012 and again in 2015, find the argument that the entire law should fall to be unconvincing. “If courts invalidate an entire law merely because Congress eliminates or revises one part, as happened here, that may well inhibit necessary reform of federal legislation in the future by turning it into an ‘all or nothing’ proposition,” wrote a group of conservative and liberal law professors in a brief filed in the case.
Still, in December 2018, U.S. District Judge Reed O’Connor in Texas accepted the GOP argument and declared the law unconstitutional. In December 2019, a three-judge 5th Circuit appeals court panel in New Orleans agreed that without the penalty the requirement to buy insurance is unconstitutional. But it sent the case back to Mr. O’Connor to suggest that perhaps the entire law need not fall.
Not wanting to wait the months or years that reconsideration would take, Democratic attorneys general defending the ACA asked the Supreme Court to hear the case this year. (Democrats are defending the law in court because the Trump administration decided to support the GOP attorneys general’s case.) The court agreed to take the case but scheduled arguments for the week after the November election.
While the fate of the ACA was and is a live political issue, few legal observers were terribly worried about the legal outcome of the case, now known as Texas v. California, if only because the case seemed much weaker than the 2012 and 2015 cases in which Mr. Roberts joined the court’s four liberals. In the 2015 case, which challenged the validity of federal tax subsidies helping millions of Americans buy health insurance on the ACA’s marketplaces, both Mr. Roberts and now-retired Justice Anthony Kennedy voted to uphold the law.
But without Ms. Ginsburg, the case could wind up in a 4-4 tie, even if Mr. Roberts supports the law’s constitutionality. That could let the lower-court ruling stand, although it would not be binding on other courts outside of the 5th Circuit. The court could also put off the arguments or, if the Republican Senate replaces Ms. Ginsburg with another conservative justice before arguments are heard, Republicans could secure a 5-4 ruling against the law. Some court observers argue that Justice Brett Kavanaugh has not favored invalidating an entire statute if only part of it is flawed and might not approve overturning the ACA. Still, what started out as an effort to energize Republican voters for the 2018 midterms after Congress failed to “repeal and replace” the health law in 2017 could end up throwing the nation’s entire health system into chaos.
At least 20 million Americans – and likely many more who sought coverage since the start of the coronavirus pandemic — who buy insurance through the ACA marketplaces or have Medicaid through the law’s expansion could lose coverage right away. Many millions more would lose the law’s popular protections guaranteeing coverage for people with preexisting health conditions, including those who have had COVID-19.
Adult children under age 26 years would no longer be guaranteed the right to remain on their parents’ health plans, and Medicare patients would lose enhanced prescription drug coverage. Women would lose guaranteed access to birth control at no out-of-pocket cost.
But a sudden elimination would affect more than just health care consumers. Insurance companies, drug companies, hospitals, and doctors have all changed the way they do business because of incentives and penalties in the health law. If it’s struck down, many of the “rules of the road” would literally be wiped away, including billing and payment mechanisms.
A new Democratic president could not drop the lawsuit because the Trump administration is not the plaintiff (the GOP attorneys general are). But a Democratic Congress and president could in theory make the entire issue go away by reinstating the penalty for failure to have insurance, even at a minimal amount. However, as far as the health law goes, for now, nothing is a sure thing.
As Nicholas Bagley, a law professor at the University of Michigan, Ann Arbor, who specializes in health issues, tweeted: “Among other things, the Affordable Care Act now dangles from a thread.”
Reproductive rights
A woman’s right to abortion – and even to birth control – also has been hanging by a thread at the high court for more than a decade. This past term, Mr. Roberts joined the liberals to invalidate a Louisiana law that would have closed most of the state’s abortion clinics, but he made it clear it was not a vote for abortion rights. The Louisiana law was too similar to a Texas law the court (without his vote) struck down in 2016, Mr. Roberts argued.
Ms. Ginsburg had been a stalwart supporter of reproductive freedom for women. In her nearly 3 decades on the court, she always voted with backers of abortion rights and birth control and led the dissenters in 2007 when the court upheld a federal ban on a specific abortion procedure.
Adding a justice opposed to abortion to the bench – which is what Trump has promised his supporters – would almost certainly tilt the court in favor of far more dramatic restrictions on the procedure and possibly an overturn of the landmark 1973 ruling Roe v. Wade.
But not only is abortion on the line: The court in recent years has repeatedly ruled that employers with religious objections can refuse to provide contraception.
And waiting in the lower-court pipeline are cases involving federal funding of Planned Parenthood in both the Medicaid and federal family planning programs, and the ability of individual health workers to decline to participate in abortion and other procedures.
For Ms. Ginsburg, those issues came down to a clear question of a woman’s guarantee of equal status under the law.
“Women, it is now acknowledged, have the talent, capacity, and right ‘to participate equally in the economic and social life of the Nation,’ ” she wrote in her dissent in that 2007 abortion case. “Their ability to realize their full potential, the Court recognized, is intimately connected to ‘their ability to control their reproductive lives.’ ”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
On Feb. 27, 2018, I got an email from the Heritage Foundation that alerted me to a news conference that afternoon held by Republican attorneys general of Texas and other states. It was referred to only as a “discussion about the Affordable Care Act lawsuit.”
I sent the following note to my editor: “I’m off to the Hill anyway. I could stop by this. You never know what it might morph into.”
Few people took that case very seriously – barely a handful of reporters attended the news conference. But it has now “morphed into” the latest existential threat to the Affordable Care Act, scheduled for oral arguments at the Supreme Court a week after the general election in November. And with the death of Justice Ruth Bader Ginsburg on Friday, that case could well morph into the threat that brings down the law in its entirety.
Democrats are raising alarms about the future of the law without Ms. Ginsburg. House Speaker Nancy Pelosi, speaking on ABC’s “This Week” Sunday morning, said that part of the strategy by President Trump and Senate Republicans to quickly fill her seat was to help undermine the ACA.
“The president is rushing to make some kind of a decision because … Nov. 10 is when the arguments begin on the Affordable Care Act,” she said. “He doesn’t want to crush the virus. He wants to crush the Affordable Care Act.”
Ms. Ginsburg’s death could throw an already chaotic general election campaign during a pandemic into even more turmoil.
Let’s take them one at a time.
The ACA under fire – again
The GOP attorneys general argued in February 2018 that the Republican-sponsored tax cut bill Congress passed two months earlier had rendered the ACA unconstitutional by reducing to zero the ACA’s penalty for not having insurance. They based their argument on Chief Justice John Roberts’ 2012 conclusion that the ACA was valid, interpreting that penalty as a constitutionally appropriate tax.
Most legal scholars, including several who challenged the law before the Supreme Court in 2012 and again in 2015, find the argument that the entire law should fall to be unconvincing. “If courts invalidate an entire law merely because Congress eliminates or revises one part, as happened here, that may well inhibit necessary reform of federal legislation in the future by turning it into an ‘all or nothing’ proposition,” wrote a group of conservative and liberal law professors in a brief filed in the case.
Still, in December 2018, U.S. District Judge Reed O’Connor in Texas accepted the GOP argument and declared the law unconstitutional. In December 2019, a three-judge 5th Circuit appeals court panel in New Orleans agreed that without the penalty the requirement to buy insurance is unconstitutional. But it sent the case back to Mr. O’Connor to suggest that perhaps the entire law need not fall.
Not wanting to wait the months or years that reconsideration would take, Democratic attorneys general defending the ACA asked the Supreme Court to hear the case this year. (Democrats are defending the law in court because the Trump administration decided to support the GOP attorneys general’s case.) The court agreed to take the case but scheduled arguments for the week after the November election.
While the fate of the ACA was and is a live political issue, few legal observers were terribly worried about the legal outcome of the case, now known as Texas v. California, if only because the case seemed much weaker than the 2012 and 2015 cases in which Mr. Roberts joined the court’s four liberals. In the 2015 case, which challenged the validity of federal tax subsidies helping millions of Americans buy health insurance on the ACA’s marketplaces, both Mr. Roberts and now-retired Justice Anthony Kennedy voted to uphold the law.
But without Ms. Ginsburg, the case could wind up in a 4-4 tie, even if Mr. Roberts supports the law’s constitutionality. That could let the lower-court ruling stand, although it would not be binding on other courts outside of the 5th Circuit. The court could also put off the arguments or, if the Republican Senate replaces Ms. Ginsburg with another conservative justice before arguments are heard, Republicans could secure a 5-4 ruling against the law. Some court observers argue that Justice Brett Kavanaugh has not favored invalidating an entire statute if only part of it is flawed and might not approve overturning the ACA. Still, what started out as an effort to energize Republican voters for the 2018 midterms after Congress failed to “repeal and replace” the health law in 2017 could end up throwing the nation’s entire health system into chaos.
At least 20 million Americans – and likely many more who sought coverage since the start of the coronavirus pandemic — who buy insurance through the ACA marketplaces or have Medicaid through the law’s expansion could lose coverage right away. Many millions more would lose the law’s popular protections guaranteeing coverage for people with preexisting health conditions, including those who have had COVID-19.
Adult children under age 26 years would no longer be guaranteed the right to remain on their parents’ health plans, and Medicare patients would lose enhanced prescription drug coverage. Women would lose guaranteed access to birth control at no out-of-pocket cost.
But a sudden elimination would affect more than just health care consumers. Insurance companies, drug companies, hospitals, and doctors have all changed the way they do business because of incentives and penalties in the health law. If it’s struck down, many of the “rules of the road” would literally be wiped away, including billing and payment mechanisms.
A new Democratic president could not drop the lawsuit because the Trump administration is not the plaintiff (the GOP attorneys general are). But a Democratic Congress and president could in theory make the entire issue go away by reinstating the penalty for failure to have insurance, even at a minimal amount. However, as far as the health law goes, for now, nothing is a sure thing.
As Nicholas Bagley, a law professor at the University of Michigan, Ann Arbor, who specializes in health issues, tweeted: “Among other things, the Affordable Care Act now dangles from a thread.”
Reproductive rights
A woman’s right to abortion – and even to birth control – also has been hanging by a thread at the high court for more than a decade. This past term, Mr. Roberts joined the liberals to invalidate a Louisiana law that would have closed most of the state’s abortion clinics, but he made it clear it was not a vote for abortion rights. The Louisiana law was too similar to a Texas law the court (without his vote) struck down in 2016, Mr. Roberts argued.
Ms. Ginsburg had been a stalwart supporter of reproductive freedom for women. In her nearly 3 decades on the court, she always voted with backers of abortion rights and birth control and led the dissenters in 2007 when the court upheld a federal ban on a specific abortion procedure.
Adding a justice opposed to abortion to the bench – which is what Trump has promised his supporters – would almost certainly tilt the court in favor of far more dramatic restrictions on the procedure and possibly an overturn of the landmark 1973 ruling Roe v. Wade.
But not only is abortion on the line: The court in recent years has repeatedly ruled that employers with religious objections can refuse to provide contraception.
And waiting in the lower-court pipeline are cases involving federal funding of Planned Parenthood in both the Medicaid and federal family planning programs, and the ability of individual health workers to decline to participate in abortion and other procedures.
For Ms. Ginsburg, those issues came down to a clear question of a woman’s guarantee of equal status under the law.
“Women, it is now acknowledged, have the talent, capacity, and right ‘to participate equally in the economic and social life of the Nation,’ ” she wrote in her dissent in that 2007 abortion case. “Their ability to realize their full potential, the Court recognized, is intimately connected to ‘their ability to control their reproductive lives.’ ”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
On Feb. 27, 2018, I got an email from the Heritage Foundation that alerted me to a news conference that afternoon held by Republican attorneys general of Texas and other states. It was referred to only as a “discussion about the Affordable Care Act lawsuit.”
I sent the following note to my editor: “I’m off to the Hill anyway. I could stop by this. You never know what it might morph into.”
Few people took that case very seriously – barely a handful of reporters attended the news conference. But it has now “morphed into” the latest existential threat to the Affordable Care Act, scheduled for oral arguments at the Supreme Court a week after the general election in November. And with the death of Justice Ruth Bader Ginsburg on Friday, that case could well morph into the threat that brings down the law in its entirety.
Democrats are raising alarms about the future of the law without Ms. Ginsburg. House Speaker Nancy Pelosi, speaking on ABC’s “This Week” Sunday morning, said that part of the strategy by President Trump and Senate Republicans to quickly fill her seat was to help undermine the ACA.
“The president is rushing to make some kind of a decision because … Nov. 10 is when the arguments begin on the Affordable Care Act,” she said. “He doesn’t want to crush the virus. He wants to crush the Affordable Care Act.”
Ms. Ginsburg’s death could throw an already chaotic general election campaign during a pandemic into even more turmoil.
Let’s take them one at a time.
The ACA under fire – again
The GOP attorneys general argued in February 2018 that the Republican-sponsored tax cut bill Congress passed two months earlier had rendered the ACA unconstitutional by reducing to zero the ACA’s penalty for not having insurance. They based their argument on Chief Justice John Roberts’ 2012 conclusion that the ACA was valid, interpreting that penalty as a constitutionally appropriate tax.
Most legal scholars, including several who challenged the law before the Supreme Court in 2012 and again in 2015, find the argument that the entire law should fall to be unconvincing. “If courts invalidate an entire law merely because Congress eliminates or revises one part, as happened here, that may well inhibit necessary reform of federal legislation in the future by turning it into an ‘all or nothing’ proposition,” wrote a group of conservative and liberal law professors in a brief filed in the case.
Still, in December 2018, U.S. District Judge Reed O’Connor in Texas accepted the GOP argument and declared the law unconstitutional. In December 2019, a three-judge 5th Circuit appeals court panel in New Orleans agreed that without the penalty the requirement to buy insurance is unconstitutional. But it sent the case back to Mr. O’Connor to suggest that perhaps the entire law need not fall.
Not wanting to wait the months or years that reconsideration would take, Democratic attorneys general defending the ACA asked the Supreme Court to hear the case this year. (Democrats are defending the law in court because the Trump administration decided to support the GOP attorneys general’s case.) The court agreed to take the case but scheduled arguments for the week after the November election.
While the fate of the ACA was and is a live political issue, few legal observers were terribly worried about the legal outcome of the case, now known as Texas v. California, if only because the case seemed much weaker than the 2012 and 2015 cases in which Mr. Roberts joined the court’s four liberals. In the 2015 case, which challenged the validity of federal tax subsidies helping millions of Americans buy health insurance on the ACA’s marketplaces, both Mr. Roberts and now-retired Justice Anthony Kennedy voted to uphold the law.
But without Ms. Ginsburg, the case could wind up in a 4-4 tie, even if Mr. Roberts supports the law’s constitutionality. That could let the lower-court ruling stand, although it would not be binding on other courts outside of the 5th Circuit. The court could also put off the arguments or, if the Republican Senate replaces Ms. Ginsburg with another conservative justice before arguments are heard, Republicans could secure a 5-4 ruling against the law. Some court observers argue that Justice Brett Kavanaugh has not favored invalidating an entire statute if only part of it is flawed and might not approve overturning the ACA. Still, what started out as an effort to energize Republican voters for the 2018 midterms after Congress failed to “repeal and replace” the health law in 2017 could end up throwing the nation’s entire health system into chaos.
At least 20 million Americans – and likely many more who sought coverage since the start of the coronavirus pandemic — who buy insurance through the ACA marketplaces or have Medicaid through the law’s expansion could lose coverage right away. Many millions more would lose the law’s popular protections guaranteeing coverage for people with preexisting health conditions, including those who have had COVID-19.
Adult children under age 26 years would no longer be guaranteed the right to remain on their parents’ health plans, and Medicare patients would lose enhanced prescription drug coverage. Women would lose guaranteed access to birth control at no out-of-pocket cost.
But a sudden elimination would affect more than just health care consumers. Insurance companies, drug companies, hospitals, and doctors have all changed the way they do business because of incentives and penalties in the health law. If it’s struck down, many of the “rules of the road” would literally be wiped away, including billing and payment mechanisms.
A new Democratic president could not drop the lawsuit because the Trump administration is not the plaintiff (the GOP attorneys general are). But a Democratic Congress and president could in theory make the entire issue go away by reinstating the penalty for failure to have insurance, even at a minimal amount. However, as far as the health law goes, for now, nothing is a sure thing.
As Nicholas Bagley, a law professor at the University of Michigan, Ann Arbor, who specializes in health issues, tweeted: “Among other things, the Affordable Care Act now dangles from a thread.”
Reproductive rights
A woman’s right to abortion – and even to birth control – also has been hanging by a thread at the high court for more than a decade. This past term, Mr. Roberts joined the liberals to invalidate a Louisiana law that would have closed most of the state’s abortion clinics, but he made it clear it was not a vote for abortion rights. The Louisiana law was too similar to a Texas law the court (without his vote) struck down in 2016, Mr. Roberts argued.
Ms. Ginsburg had been a stalwart supporter of reproductive freedom for women. In her nearly 3 decades on the court, she always voted with backers of abortion rights and birth control and led the dissenters in 2007 when the court upheld a federal ban on a specific abortion procedure.
Adding a justice opposed to abortion to the bench – which is what Trump has promised his supporters – would almost certainly tilt the court in favor of far more dramatic restrictions on the procedure and possibly an overturn of the landmark 1973 ruling Roe v. Wade.
But not only is abortion on the line: The court in recent years has repeatedly ruled that employers with religious objections can refuse to provide contraception.
And waiting in the lower-court pipeline are cases involving federal funding of Planned Parenthood in both the Medicaid and federal family planning programs, and the ability of individual health workers to decline to participate in abortion and other procedures.
For Ms. Ginsburg, those issues came down to a clear question of a woman’s guarantee of equal status under the law.
“Women, it is now acknowledged, have the talent, capacity, and right ‘to participate equally in the economic and social life of the Nation,’ ” she wrote in her dissent in that 2007 abortion case. “Their ability to realize their full potential, the Court recognized, is intimately connected to ‘their ability to control their reproductive lives.’ ”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
Vacuum device quickly stops postpartum hemorrhage
A novel intrauterine device that uses suction to compress the uterus to control postpartum hemorrhage has received high marks for effectiveness and ease of use, researchers say.
Calling this approach “a stroke of brilliance,” and is less risky as well.
“This device can be placed in the uterus within a minute or so and does not need any initial anesthesia and would not be associated with the delay needed for a surgical approach,” Dr. Byrne explained. Dr. Byrne, who was not involved in the study, is chair of the department of obstetrics and gynecology at Santa Clara Valley Medical Center in San Jose, Calif.
To test the efficacy and safety of the device (Jada System, Alydia Health), Mary E. D’Alton, MD, and colleagues conducted a prospective, observational treatment study in 12 U.S. medical centers. They reported their findings in an article published online Sept. 9 in Obstetrics and Gynecology.
“The Jada System (novel intrauterine vacuum-induced hemorrhage-control device) was specifically designed to offer rapid treatment by applying low-level intrauterine vacuum to facilitate the physiologic forces of uterine contractions to constrict myometrial blood vessels and achieve hemostasis,” Dr. D’Alton, from New York–Presbyterian/Columbia University Irving Medical Center in New York, and colleagues wrote.
“The device had a low rate of adverse events during this study, all of which were expected risks and resolved with treatment without serious clinical sequelae. Investigators, all first-time users of the device, found the system easy to use, which suggests that, after device education and with availability of a quick reference guide outlining steps, there is a minimal learning curve for use,” they added.
Alydia Health, the company that developed the device, funded this study and supported the research staff, who recruited participants and gathered follow-up data on them. On Sept. 9, the U.S. Food and Drug Administration granted 510(k) clearance for the device, according to a company news release.
Effective, safe
The multicenter study included 107 patients (mean age, 29.7 years) with postpartum hemorrhage or abnormal postpartum uterine bleeding, 106 of whom received any study treatment with the device attached to vacuum. More than half (57%) of the participants were White, and just fewer than one-quarter (24%) were Black.
Treatment was successful in 94% (100/106) of participants, with “definitive control of abnormal bleeding” occurring in a median of 3 minutes after attachment to vacuum.
Eight adverse events were judged to have been possibly related to the device or procedure: four cases of endometritis, and one case each of presumed endometritis, bacterial vaginosis, vaginal candidiasis, and disruption of a vaginal laceration repair. The eight adverse events were identified as potential risks, and all resolved without serious clinical consequences.
Thirty-five patients required transfusions of 1-3 U of red blood cells, and five patients required at least 4 U of red blood cells.
Uterine atony most frequent culprit
As many as 80% of postpartum hemorrhages are caused by uterine atony, according to the authors.
Dr. Byrne explained that the uterus is a muscular organ that contains many “spiral arteries” that are “squeezed” by the uterus as it tightens down after childbirth, which prevents them from bleeding excessively.
“With uterine atony, the uterus muscle doesn’t squeeze effectively, and therefore it’s not one or two arteries, it’s hundreds and hundreds of small arteries and capillaries [and] arterioles all bleeding; it’s a wide area of uterus,” he continued.
When medications alone are ineffective at controlling bleeding, tamponade is often added to put outward pressure on the inner wall of the uterus for 12-24 hours. Although tamponade is effective in approximately 87% of atony-related cases of postpartum hemorrhage, the use of outward pressure on the uterine walls “is counterintuitive if the ultimate goal is uterine contraction,” the authors wrote.
Dr. Byrne said he and his colleagues saw this device several years ago, and they felt at the time that it appeared to be “more intuitive to use vacuum to compress the uterus inward compared to the nonetheless valuable and effective Bakri balloon and other techniques that expand the uterus outward.”
The fact that there is no need for prophylactic antibiotics also sets the vacuum device apart from the Bakri balloon, use of which routinely involves administration of prophylactic antibiotics, Dr. Byrne said.
In the current study, 64% of participants were obese, which makes management of postpartum hemorrhage “really challenging” because it’s difficult to effectively massage the uterus through adipose tissue, Dr. Byrne explained. Patients with obesity “also have different hemodynamics for how effectively [injected medications will] be delivered to the uterus,” he added.
“A device like this that could be placed and works so efficiently – even with an obese patient – that’s actually very powerful,” Dr. Byrne said.
Quick placement, almost immediate improvement
The discomfort experienced during placement of the device is similar to that experienced during sweeping of the uterus, Dr. Byrne explained. “You’d want a patient comfortable, ideally with an epidural already active, but if it’s an emergency, you wouldn’t have to wait for that; you could sweep the uterus quickly, place this, initiate suction, and it would all be so quick you could usually talk a patient through it and get it done,” Dr. Byrne continued.
Almost all of the investigators (98%) said the device was easy to use, and 97% said they would recommend it.
The vacuum device is made of medical-grade silicone and consists of an oval-shaped intrauterine loop at one end and a vacuum connector at the other end that can be attached to a standard suction cannister. On the inner side of the intrauterine loop are 20 vacuum pores covered by a shield that protects uterine tissue and prevents the vacuum pores from clogging with tissue or clotted blood.
Before insertion of the vacuum device, the clinician manually sweeps the uterus to identify retained placental fragments and to assess the uterine cavity. The distal end of the device is inserted into the uterus, and a cervical seal, positioned just outside the cervical os, is filled with 60 to 120 cc of sterile fluid. The proximal end is attached to low-level vacuum at a pressure of 80 ± 10 mm Hg. The device is left in place with continued suction for at least 1 hour after bleeding is controlled, at which time the suction is disconnected and the cervical seal is emptied. The device remains in place for at least 30 minutes, during which the patient is observed closely.
“It looks like 75%-80% of cases stop bleeding within 5 minutes. ... Then you stop the pressure after an hour [and] wait at least 30 minutes. You could actually have this out of the patient’s body within 2 hours,” Dr. Byrne said.
Dr. Byrne has disclosed no such financial relationships.
A version of this article originally appeared on Medscape.com.
A novel intrauterine device that uses suction to compress the uterus to control postpartum hemorrhage has received high marks for effectiveness and ease of use, researchers say.
Calling this approach “a stroke of brilliance,” and is less risky as well.
“This device can be placed in the uterus within a minute or so and does not need any initial anesthesia and would not be associated with the delay needed for a surgical approach,” Dr. Byrne explained. Dr. Byrne, who was not involved in the study, is chair of the department of obstetrics and gynecology at Santa Clara Valley Medical Center in San Jose, Calif.
To test the efficacy and safety of the device (Jada System, Alydia Health), Mary E. D’Alton, MD, and colleagues conducted a prospective, observational treatment study in 12 U.S. medical centers. They reported their findings in an article published online Sept. 9 in Obstetrics and Gynecology.
“The Jada System (novel intrauterine vacuum-induced hemorrhage-control device) was specifically designed to offer rapid treatment by applying low-level intrauterine vacuum to facilitate the physiologic forces of uterine contractions to constrict myometrial blood vessels and achieve hemostasis,” Dr. D’Alton, from New York–Presbyterian/Columbia University Irving Medical Center in New York, and colleagues wrote.
“The device had a low rate of adverse events during this study, all of which were expected risks and resolved with treatment without serious clinical sequelae. Investigators, all first-time users of the device, found the system easy to use, which suggests that, after device education and with availability of a quick reference guide outlining steps, there is a minimal learning curve for use,” they added.
Alydia Health, the company that developed the device, funded this study and supported the research staff, who recruited participants and gathered follow-up data on them. On Sept. 9, the U.S. Food and Drug Administration granted 510(k) clearance for the device, according to a company news release.
Effective, safe
The multicenter study included 107 patients (mean age, 29.7 years) with postpartum hemorrhage or abnormal postpartum uterine bleeding, 106 of whom received any study treatment with the device attached to vacuum. More than half (57%) of the participants were White, and just fewer than one-quarter (24%) were Black.
Treatment was successful in 94% (100/106) of participants, with “definitive control of abnormal bleeding” occurring in a median of 3 minutes after attachment to vacuum.
Eight adverse events were judged to have been possibly related to the device or procedure: four cases of endometritis, and one case each of presumed endometritis, bacterial vaginosis, vaginal candidiasis, and disruption of a vaginal laceration repair. The eight adverse events were identified as potential risks, and all resolved without serious clinical consequences.
Thirty-five patients required transfusions of 1-3 U of red blood cells, and five patients required at least 4 U of red blood cells.
Uterine atony most frequent culprit
As many as 80% of postpartum hemorrhages are caused by uterine atony, according to the authors.
Dr. Byrne explained that the uterus is a muscular organ that contains many “spiral arteries” that are “squeezed” by the uterus as it tightens down after childbirth, which prevents them from bleeding excessively.
“With uterine atony, the uterus muscle doesn’t squeeze effectively, and therefore it’s not one or two arteries, it’s hundreds and hundreds of small arteries and capillaries [and] arterioles all bleeding; it’s a wide area of uterus,” he continued.
When medications alone are ineffective at controlling bleeding, tamponade is often added to put outward pressure on the inner wall of the uterus for 12-24 hours. Although tamponade is effective in approximately 87% of atony-related cases of postpartum hemorrhage, the use of outward pressure on the uterine walls “is counterintuitive if the ultimate goal is uterine contraction,” the authors wrote.
Dr. Byrne said he and his colleagues saw this device several years ago, and they felt at the time that it appeared to be “more intuitive to use vacuum to compress the uterus inward compared to the nonetheless valuable and effective Bakri balloon and other techniques that expand the uterus outward.”
The fact that there is no need for prophylactic antibiotics also sets the vacuum device apart from the Bakri balloon, use of which routinely involves administration of prophylactic antibiotics, Dr. Byrne said.
In the current study, 64% of participants were obese, which makes management of postpartum hemorrhage “really challenging” because it’s difficult to effectively massage the uterus through adipose tissue, Dr. Byrne explained. Patients with obesity “also have different hemodynamics for how effectively [injected medications will] be delivered to the uterus,” he added.
“A device like this that could be placed and works so efficiently – even with an obese patient – that’s actually very powerful,” Dr. Byrne said.
Quick placement, almost immediate improvement
The discomfort experienced during placement of the device is similar to that experienced during sweeping of the uterus, Dr. Byrne explained. “You’d want a patient comfortable, ideally with an epidural already active, but if it’s an emergency, you wouldn’t have to wait for that; you could sweep the uterus quickly, place this, initiate suction, and it would all be so quick you could usually talk a patient through it and get it done,” Dr. Byrne continued.
Almost all of the investigators (98%) said the device was easy to use, and 97% said they would recommend it.
The vacuum device is made of medical-grade silicone and consists of an oval-shaped intrauterine loop at one end and a vacuum connector at the other end that can be attached to a standard suction cannister. On the inner side of the intrauterine loop are 20 vacuum pores covered by a shield that protects uterine tissue and prevents the vacuum pores from clogging with tissue or clotted blood.
Before insertion of the vacuum device, the clinician manually sweeps the uterus to identify retained placental fragments and to assess the uterine cavity. The distal end of the device is inserted into the uterus, and a cervical seal, positioned just outside the cervical os, is filled with 60 to 120 cc of sterile fluid. The proximal end is attached to low-level vacuum at a pressure of 80 ± 10 mm Hg. The device is left in place with continued suction for at least 1 hour after bleeding is controlled, at which time the suction is disconnected and the cervical seal is emptied. The device remains in place for at least 30 minutes, during which the patient is observed closely.
“It looks like 75%-80% of cases stop bleeding within 5 minutes. ... Then you stop the pressure after an hour [and] wait at least 30 minutes. You could actually have this out of the patient’s body within 2 hours,” Dr. Byrne said.
Dr. Byrne has disclosed no such financial relationships.
A version of this article originally appeared on Medscape.com.
A novel intrauterine device that uses suction to compress the uterus to control postpartum hemorrhage has received high marks for effectiveness and ease of use, researchers say.
Calling this approach “a stroke of brilliance,” and is less risky as well.
“This device can be placed in the uterus within a minute or so and does not need any initial anesthesia and would not be associated with the delay needed for a surgical approach,” Dr. Byrne explained. Dr. Byrne, who was not involved in the study, is chair of the department of obstetrics and gynecology at Santa Clara Valley Medical Center in San Jose, Calif.
To test the efficacy and safety of the device (Jada System, Alydia Health), Mary E. D’Alton, MD, and colleagues conducted a prospective, observational treatment study in 12 U.S. medical centers. They reported their findings in an article published online Sept. 9 in Obstetrics and Gynecology.
“The Jada System (novel intrauterine vacuum-induced hemorrhage-control device) was specifically designed to offer rapid treatment by applying low-level intrauterine vacuum to facilitate the physiologic forces of uterine contractions to constrict myometrial blood vessels and achieve hemostasis,” Dr. D’Alton, from New York–Presbyterian/Columbia University Irving Medical Center in New York, and colleagues wrote.
“The device had a low rate of adverse events during this study, all of which were expected risks and resolved with treatment without serious clinical sequelae. Investigators, all first-time users of the device, found the system easy to use, which suggests that, after device education and with availability of a quick reference guide outlining steps, there is a minimal learning curve for use,” they added.
Alydia Health, the company that developed the device, funded this study and supported the research staff, who recruited participants and gathered follow-up data on them. On Sept. 9, the U.S. Food and Drug Administration granted 510(k) clearance for the device, according to a company news release.
Effective, safe
The multicenter study included 107 patients (mean age, 29.7 years) with postpartum hemorrhage or abnormal postpartum uterine bleeding, 106 of whom received any study treatment with the device attached to vacuum. More than half (57%) of the participants were White, and just fewer than one-quarter (24%) were Black.
Treatment was successful in 94% (100/106) of participants, with “definitive control of abnormal bleeding” occurring in a median of 3 minutes after attachment to vacuum.
Eight adverse events were judged to have been possibly related to the device or procedure: four cases of endometritis, and one case each of presumed endometritis, bacterial vaginosis, vaginal candidiasis, and disruption of a vaginal laceration repair. The eight adverse events were identified as potential risks, and all resolved without serious clinical consequences.
Thirty-five patients required transfusions of 1-3 U of red blood cells, and five patients required at least 4 U of red blood cells.
Uterine atony most frequent culprit
As many as 80% of postpartum hemorrhages are caused by uterine atony, according to the authors.
Dr. Byrne explained that the uterus is a muscular organ that contains many “spiral arteries” that are “squeezed” by the uterus as it tightens down after childbirth, which prevents them from bleeding excessively.
“With uterine atony, the uterus muscle doesn’t squeeze effectively, and therefore it’s not one or two arteries, it’s hundreds and hundreds of small arteries and capillaries [and] arterioles all bleeding; it’s a wide area of uterus,” he continued.
When medications alone are ineffective at controlling bleeding, tamponade is often added to put outward pressure on the inner wall of the uterus for 12-24 hours. Although tamponade is effective in approximately 87% of atony-related cases of postpartum hemorrhage, the use of outward pressure on the uterine walls “is counterintuitive if the ultimate goal is uterine contraction,” the authors wrote.
Dr. Byrne said he and his colleagues saw this device several years ago, and they felt at the time that it appeared to be “more intuitive to use vacuum to compress the uterus inward compared to the nonetheless valuable and effective Bakri balloon and other techniques that expand the uterus outward.”
The fact that there is no need for prophylactic antibiotics also sets the vacuum device apart from the Bakri balloon, use of which routinely involves administration of prophylactic antibiotics, Dr. Byrne said.
In the current study, 64% of participants were obese, which makes management of postpartum hemorrhage “really challenging” because it’s difficult to effectively massage the uterus through adipose tissue, Dr. Byrne explained. Patients with obesity “also have different hemodynamics for how effectively [injected medications will] be delivered to the uterus,” he added.
“A device like this that could be placed and works so efficiently – even with an obese patient – that’s actually very powerful,” Dr. Byrne said.
Quick placement, almost immediate improvement
The discomfort experienced during placement of the device is similar to that experienced during sweeping of the uterus, Dr. Byrne explained. “You’d want a patient comfortable, ideally with an epidural already active, but if it’s an emergency, you wouldn’t have to wait for that; you could sweep the uterus quickly, place this, initiate suction, and it would all be so quick you could usually talk a patient through it and get it done,” Dr. Byrne continued.
Almost all of the investigators (98%) said the device was easy to use, and 97% said they would recommend it.
The vacuum device is made of medical-grade silicone and consists of an oval-shaped intrauterine loop at one end and a vacuum connector at the other end that can be attached to a standard suction cannister. On the inner side of the intrauterine loop are 20 vacuum pores covered by a shield that protects uterine tissue and prevents the vacuum pores from clogging with tissue or clotted blood.
Before insertion of the vacuum device, the clinician manually sweeps the uterus to identify retained placental fragments and to assess the uterine cavity. The distal end of the device is inserted into the uterus, and a cervical seal, positioned just outside the cervical os, is filled with 60 to 120 cc of sterile fluid. The proximal end is attached to low-level vacuum at a pressure of 80 ± 10 mm Hg. The device is left in place with continued suction for at least 1 hour after bleeding is controlled, at which time the suction is disconnected and the cervical seal is emptied. The device remains in place for at least 30 minutes, during which the patient is observed closely.
“It looks like 75%-80% of cases stop bleeding within 5 minutes. ... Then you stop the pressure after an hour [and] wait at least 30 minutes. You could actually have this out of the patient’s body within 2 hours,” Dr. Byrne said.
Dr. Byrne has disclosed no such financial relationships.
A version of this article originally appeared on Medscape.com.
Counterintuitive findings for domestic violence during COVID-19
Intimate partner violence (IPV) has not increased during the COVID-19 pandemic, at least during the early stages of the pandemic, new research suggests.
In April 2020, investigators surveyed over 1,750 individuals in intimate partner relationships. The survey was drawn from social media and email distribution lists. The researchers found that, of the roughly one-fifth who screened positive for IPV, half stated that the degree of victimization had remained the same since the COVID-19 outbreak; 17% reported that it had worsened; and one third reported that it had gotten better.
Those who reported worsening victimization said that sexual and physical violence, in particular, were exacerbated early in the pandemic’s course.
“I was surprised by this finding, and we certainly were not expecting it – in fact, I expected that the vast majority of victims would report that victimization got worse during stay-at-home policies, but that wasn’t the case,” lead author Katelyn Jetelina, PhD, MPH, assistant professor in the department of epidemiology, human genetics, and environmental sciences, University of Texas Health Science Center, Dallas, said in an interview.
“I think the biggest take-home message is that some victims got better, but the vast majority stayed the same. These victims, men and women, were isolated with their perpetrator during COVID-19, so she added.
The study was published online Sept. 1 in Injury Prevention.
‘Shadow pandemic?’
The World Health Organization called upon health care organizations to be prepared to curb a potential IPV “shadow pandemic” during the COVID-19 pandemic.
However, no study has specifically evaluated whether self-reported victimization, particularly with regard to the severity and type of abuse, changed during the early period after COVID-19 social distancing polices were mandated.
“We scrambled right away when the pandemic hit because it was a unique opportunity to examine how behaviors change due to early stay-at-home policies; and, as a violence and injury epidemiologist, I am always curious about IPV, and this was a small subanalysis of that larger question,” Dr. Jetelina said.
The researchers recruited participants through their university and private social media accounts as well as professional distribution lists. Of those who completed the survey, 1,759 (mean age, 42 years) reported that they currently had an intimate partner. These participants were included in the study.
IPV was determined using the five-item Extended Hurt, Insulted, Threatened, and Scream (E-HITS) construct. Respondents were asked how often their partner physically hurt them, insulted them, threatened them with harm, screamed or cursed at them, or forced them to engage in sexual activities.
Each item was answered using a 5-point Likert scale. Scores ranged from 1, indicating never, to 5, indicating frequently. Participants who scored ≥7 were considered IPV positive.
Participants were also asked whether IPV severity had gotten much/somewhat better, had remained the same, or had gotten somewhat/much worse.
First peek
Of the total sample, 18% screened positive for IPV. Of these, 54% reported that the victimization had remained the same, 17% reported that it had worsened, and 30% said it had improved.
The majority of IPV victims experienced being insulted (97%) or being screamed at (86%).
Among those who reported worsening of IPV, the risk for physical violence was 4.38 times higher than the risk for nonphysical victimization. The risk for sexual victimization was 2.31 times higher than the risk for nonsexual victimization.
Among those who reported that IPV had gotten better, the improvement was 3.47 times higher with regard to physical victimization, compared with nonphysical victimization. Dr. Jetelina acknowledged that the findings cannot be generalized to the broader population.
“This was a convenience sample, but it is the first peek into what is happening behind closed doors and a first step to hearing collecting data from the victims themselves to better understand this ‘shadow pandemic’ and inform creative efforts to create better services for them while they are in isolation,” she said.
Lethality indicators
Commenting on the study, Peter Cronholm, MD, MSCE, associate professor of family medicine and community health at the Hospital of the University of Pennsylvania, Philadelphia, questioned the use of a score of 7 on the E-HITS screen to determine the presence of IPV.
“I think there are other thresholds that might be important, and even low levels of sexual violence may be different than higher levels of emotional violence,” said Dr. Cronholm, who was not involved with the study.
“Someone may have been sexually assaulted frequently but not cross the threshold, so I think it would have been helpful for the researchers to look at different types of violence,” he said.
Also commenting on the study, Jessica Palardy, LSW, program supervisor at STOP Intimate Partner Violence, Philadelphia, said, the findings “solidify a trend we sensed was happening but couldn’t confirm.”
She said her agency’s clients “have had a wide variety of experiences, in terms of increases or decreases in victimization.”
Some clients were able to use the quarantine as an excuse to stay with family or friends and so could avoid seeing their partners. “Others indicated that because their partners were distracted by figuring out a new method of work, the tension shifted away from the victim,” said Ms. Palardy, who was not involved in the research.
“For those who saw an increase in victimization, we noticed that this increase also came with an increase in lethality indicators, such as strangulation, physical violence, use of weapons and substances, etc,” she said.
She emphasized that it is critical to screen people for IPV to ensure their safety.
“The goal is to connect people with resources before they are in a more lethal situation so that they can increase their safety and know their options,” Ms. Palardy said.
The National Domestic Violence Hotline and the Crisis Text Line are two sources of support for IPV victims.
Dr. Jetelina and coauthors, Dr. Cronholm, and Ms. Palardy reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Intimate partner violence (IPV) has not increased during the COVID-19 pandemic, at least during the early stages of the pandemic, new research suggests.
In April 2020, investigators surveyed over 1,750 individuals in intimate partner relationships. The survey was drawn from social media and email distribution lists. The researchers found that, of the roughly one-fifth who screened positive for IPV, half stated that the degree of victimization had remained the same since the COVID-19 outbreak; 17% reported that it had worsened; and one third reported that it had gotten better.
Those who reported worsening victimization said that sexual and physical violence, in particular, were exacerbated early in the pandemic’s course.
“I was surprised by this finding, and we certainly were not expecting it – in fact, I expected that the vast majority of victims would report that victimization got worse during stay-at-home policies, but that wasn’t the case,” lead author Katelyn Jetelina, PhD, MPH, assistant professor in the department of epidemiology, human genetics, and environmental sciences, University of Texas Health Science Center, Dallas, said in an interview.
“I think the biggest take-home message is that some victims got better, but the vast majority stayed the same. These victims, men and women, were isolated with their perpetrator during COVID-19, so she added.
The study was published online Sept. 1 in Injury Prevention.
‘Shadow pandemic?’
The World Health Organization called upon health care organizations to be prepared to curb a potential IPV “shadow pandemic” during the COVID-19 pandemic.
However, no study has specifically evaluated whether self-reported victimization, particularly with regard to the severity and type of abuse, changed during the early period after COVID-19 social distancing polices were mandated.
“We scrambled right away when the pandemic hit because it was a unique opportunity to examine how behaviors change due to early stay-at-home policies; and, as a violence and injury epidemiologist, I am always curious about IPV, and this was a small subanalysis of that larger question,” Dr. Jetelina said.
The researchers recruited participants through their university and private social media accounts as well as professional distribution lists. Of those who completed the survey, 1,759 (mean age, 42 years) reported that they currently had an intimate partner. These participants were included in the study.
IPV was determined using the five-item Extended Hurt, Insulted, Threatened, and Scream (E-HITS) construct. Respondents were asked how often their partner physically hurt them, insulted them, threatened them with harm, screamed or cursed at them, or forced them to engage in sexual activities.
Each item was answered using a 5-point Likert scale. Scores ranged from 1, indicating never, to 5, indicating frequently. Participants who scored ≥7 were considered IPV positive.
Participants were also asked whether IPV severity had gotten much/somewhat better, had remained the same, or had gotten somewhat/much worse.
First peek
Of the total sample, 18% screened positive for IPV. Of these, 54% reported that the victimization had remained the same, 17% reported that it had worsened, and 30% said it had improved.
The majority of IPV victims experienced being insulted (97%) or being screamed at (86%).
Among those who reported worsening of IPV, the risk for physical violence was 4.38 times higher than the risk for nonphysical victimization. The risk for sexual victimization was 2.31 times higher than the risk for nonsexual victimization.
Among those who reported that IPV had gotten better, the improvement was 3.47 times higher with regard to physical victimization, compared with nonphysical victimization. Dr. Jetelina acknowledged that the findings cannot be generalized to the broader population.
“This was a convenience sample, but it is the first peek into what is happening behind closed doors and a first step to hearing collecting data from the victims themselves to better understand this ‘shadow pandemic’ and inform creative efforts to create better services for them while they are in isolation,” she said.
Lethality indicators
Commenting on the study, Peter Cronholm, MD, MSCE, associate professor of family medicine and community health at the Hospital of the University of Pennsylvania, Philadelphia, questioned the use of a score of 7 on the E-HITS screen to determine the presence of IPV.
“I think there are other thresholds that might be important, and even low levels of sexual violence may be different than higher levels of emotional violence,” said Dr. Cronholm, who was not involved with the study.
“Someone may have been sexually assaulted frequently but not cross the threshold, so I think it would have been helpful for the researchers to look at different types of violence,” he said.
Also commenting on the study, Jessica Palardy, LSW, program supervisor at STOP Intimate Partner Violence, Philadelphia, said, the findings “solidify a trend we sensed was happening but couldn’t confirm.”
She said her agency’s clients “have had a wide variety of experiences, in terms of increases or decreases in victimization.”
Some clients were able to use the quarantine as an excuse to stay with family or friends and so could avoid seeing their partners. “Others indicated that because their partners were distracted by figuring out a new method of work, the tension shifted away from the victim,” said Ms. Palardy, who was not involved in the research.
“For those who saw an increase in victimization, we noticed that this increase also came with an increase in lethality indicators, such as strangulation, physical violence, use of weapons and substances, etc,” she said.
She emphasized that it is critical to screen people for IPV to ensure their safety.
“The goal is to connect people with resources before they are in a more lethal situation so that they can increase their safety and know their options,” Ms. Palardy said.
The National Domestic Violence Hotline and the Crisis Text Line are two sources of support for IPV victims.
Dr. Jetelina and coauthors, Dr. Cronholm, and Ms. Palardy reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Intimate partner violence (IPV) has not increased during the COVID-19 pandemic, at least during the early stages of the pandemic, new research suggests.
In April 2020, investigators surveyed over 1,750 individuals in intimate partner relationships. The survey was drawn from social media and email distribution lists. The researchers found that, of the roughly one-fifth who screened positive for IPV, half stated that the degree of victimization had remained the same since the COVID-19 outbreak; 17% reported that it had worsened; and one third reported that it had gotten better.
Those who reported worsening victimization said that sexual and physical violence, in particular, were exacerbated early in the pandemic’s course.
“I was surprised by this finding, and we certainly were not expecting it – in fact, I expected that the vast majority of victims would report that victimization got worse during stay-at-home policies, but that wasn’t the case,” lead author Katelyn Jetelina, PhD, MPH, assistant professor in the department of epidemiology, human genetics, and environmental sciences, University of Texas Health Science Center, Dallas, said in an interview.
“I think the biggest take-home message is that some victims got better, but the vast majority stayed the same. These victims, men and women, were isolated with their perpetrator during COVID-19, so she added.
The study was published online Sept. 1 in Injury Prevention.
‘Shadow pandemic?’
The World Health Organization called upon health care organizations to be prepared to curb a potential IPV “shadow pandemic” during the COVID-19 pandemic.
However, no study has specifically evaluated whether self-reported victimization, particularly with regard to the severity and type of abuse, changed during the early period after COVID-19 social distancing polices were mandated.
“We scrambled right away when the pandemic hit because it was a unique opportunity to examine how behaviors change due to early stay-at-home policies; and, as a violence and injury epidemiologist, I am always curious about IPV, and this was a small subanalysis of that larger question,” Dr. Jetelina said.
The researchers recruited participants through their university and private social media accounts as well as professional distribution lists. Of those who completed the survey, 1,759 (mean age, 42 years) reported that they currently had an intimate partner. These participants were included in the study.
IPV was determined using the five-item Extended Hurt, Insulted, Threatened, and Scream (E-HITS) construct. Respondents were asked how often their partner physically hurt them, insulted them, threatened them with harm, screamed or cursed at them, or forced them to engage in sexual activities.
Each item was answered using a 5-point Likert scale. Scores ranged from 1, indicating never, to 5, indicating frequently. Participants who scored ≥7 were considered IPV positive.
Participants were also asked whether IPV severity had gotten much/somewhat better, had remained the same, or had gotten somewhat/much worse.
First peek
Of the total sample, 18% screened positive for IPV. Of these, 54% reported that the victimization had remained the same, 17% reported that it had worsened, and 30% said it had improved.
The majority of IPV victims experienced being insulted (97%) or being screamed at (86%).
Among those who reported worsening of IPV, the risk for physical violence was 4.38 times higher than the risk for nonphysical victimization. The risk for sexual victimization was 2.31 times higher than the risk for nonsexual victimization.
Among those who reported that IPV had gotten better, the improvement was 3.47 times higher with regard to physical victimization, compared with nonphysical victimization. Dr. Jetelina acknowledged that the findings cannot be generalized to the broader population.
“This was a convenience sample, but it is the first peek into what is happening behind closed doors and a first step to hearing collecting data from the victims themselves to better understand this ‘shadow pandemic’ and inform creative efforts to create better services for them while they are in isolation,” she said.
Lethality indicators
Commenting on the study, Peter Cronholm, MD, MSCE, associate professor of family medicine and community health at the Hospital of the University of Pennsylvania, Philadelphia, questioned the use of a score of 7 on the E-HITS screen to determine the presence of IPV.
“I think there are other thresholds that might be important, and even low levels of sexual violence may be different than higher levels of emotional violence,” said Dr. Cronholm, who was not involved with the study.
“Someone may have been sexually assaulted frequently but not cross the threshold, so I think it would have been helpful for the researchers to look at different types of violence,” he said.
Also commenting on the study, Jessica Palardy, LSW, program supervisor at STOP Intimate Partner Violence, Philadelphia, said, the findings “solidify a trend we sensed was happening but couldn’t confirm.”
She said her agency’s clients “have had a wide variety of experiences, in terms of increases or decreases in victimization.”
Some clients were able to use the quarantine as an excuse to stay with family or friends and so could avoid seeing their partners. “Others indicated that because their partners were distracted by figuring out a new method of work, the tension shifted away from the victim,” said Ms. Palardy, who was not involved in the research.
“For those who saw an increase in victimization, we noticed that this increase also came with an increase in lethality indicators, such as strangulation, physical violence, use of weapons and substances, etc,” she said.
She emphasized that it is critical to screen people for IPV to ensure their safety.
“The goal is to connect people with resources before they are in a more lethal situation so that they can increase their safety and know their options,” Ms. Palardy said.
The National Domestic Violence Hotline and the Crisis Text Line are two sources of support for IPV victims.
Dr. Jetelina and coauthors, Dr. Cronholm, and Ms. Palardy reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.