User login
Can viscous fiber lower glycemic markers in type 2 diabetes?
ILLUSTRATIVE CASE
A 57-year-old man who was given a diagnosis of T2D a year ago presents for an office visit. His hemoglobin A1C level at diagnosis was 8.3%. He is otherwise healthy and has been adhering well to a plan of metformin 1000 mg twice daily, regular exercise, and a low-carbohydrate diet you recommended. His most recent hemoglobin A1C is 7.3%. He is pleased with his progress, so he is discouraged when you tell him that he is not yet at goal. He asks if there are other things that he can do to further lower his hemoglobin A1C. What can you recommend for him?
According to the National Diabetes Statistics Report, 2020 from the Centers for Disease Control and Prevention, approximately 34.1 million US adults ≥ 18 years of age (13% of the adult population) have diabetes, 50% of whom have a hemoglobin A1C > 7%. The report also states that approximately 88 million US adults—more than one-third of the population—have prediabetes.2
The American Diabetes Association (ADA) estimated that diabetes-related health care costs in the United States for 2017 totaled $237 billion, an increase of 26% from 2012. More than $30 billion of this expense comes directly from diabetes medications; the remainder of these costs are related to lost wages, clinic visits, hospitalizations, and treatment for diabetic complications and comorbidities. After controlling for age and gender, medical expenditures for people with diabetes are 2.3 times higher than for those without diabetes.3
The 2019
STUDY SUMMARY
Effect on A1C exceeded the FDA threshold for new drugs
This systematic review and meta-analysis searched MEDLINE, Embase, and the Cochrane Central Register of Controlled Trials to identify randomized controlled trials that studied the effects of viscous fiber supplementation on glycemic control in patients with T2D. Eligible studies included those that: (1) had a duration ≥ 3 weeks; (2) allowed isolation of the viscous fiber effects; and (3) reported at least 1 of the following glycemic measures: hemoglobin A1C (n = 1148 patients), fasting glucose (n = 1394), fasting insulin (n = 228),
Data were pooled using the generic inverse variance method and expressed as mean difference (MD) with 95% confidence intervals (CIs). Heterogeneity was assessed and quantified (Cochran Q and I2 statistics, respectively). I2 ≥ 50% indicates substantial heterogeneity. The Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach was used to evaluate the overall strength of evidence.
Twenty-eight eligible studies were compared. The median age of included patients was 60 years. The median dose of viscous fiber was 13.1 g/d (range, 2.55-21). Viscous fiber type varied between the studies and included psyllium, guar gum, β-glucan, and konjac, and was consumed in powder, tablet, capsule, and limited food-based forms (in 1 of the included studies). The median trial duration was 8 weeks, with 11 trials lasting ≥ 12 weeks.
Continue to: The study found...
The study found moderate-grade evidence that, when added to standard of care, viscous fiber supplementation reduced hemoglobin A1C (MD = –0.58%; 95% CI, –0.88 to –0.28; P = .0002; I2 = 91%), fasting glucose (MD = –14.8 mg/dL; 95% CI, –23.8 to –5.58; P = .001; I2 = 92%), and HOMA-IR (MD = –1.89; 95% CI, –3.45 to –0.33; P = .02; I2 = 94%) compared with control. The effect on hemoglobin A1C exceeds the ≥ 0.3% threshold established by the US Food and Drug Administration for new antihyperglycemic drug development. There was no significant effect on fasting insulin or fructosamine, although the sample size for fructosamine was small. No significant evidence of a dose-dependent response effect was found. The studies had substantial heterogeneity. No evaluation of potential or real harm was noted in the analysis.
WHAT’S NEW
Potential glycemic benefit without large dietary increase
The glucose-lowering effects of increased fiber intake have often been hypothesized, but this meta-analysis is the first to focus specifically on the effect of viscous fiber supplements in patients with T2D. Prior meta-analyses, including those cited in the 2019 ADA recommendations mentioned above, included primarily whole-food dietary sources of fiber in the treatment arms and generally had more modest effects on outcomes.4,6,7
By focusing on viscous fiber supplements, this study isolated the effect of these supplements vs fiber-rich dietary changes. It illustrates a greater potential benefit with supplements than whole-food dietary sources of fiber, and at a lower dose of fiber than was seen in prior studies without requiring substantial increases in caloric intake. Viscous fiber supplementation is a potential adjunct to the usual evidence-based standards of care for glycemic control in patients with T2D.
CAVEATS
Limited study durations may raise uncertainty about long-term benefits
This meta-analysis does have its limitations. The heterogeneity among the studies analyzed makes it difficult to establish a single recommendation regarding dose, type, and brand of fiber to be used. Only 11 of the 28 studies lasted longer than 12 weeks, with a median duration of 8 weeks, making any long-term effects on hemoglobin A1C unknown. No adverse effects or reactions were described to evaluate safety and tolerability of the viscous fiber supplementation. No patient-oriented outcomes were reported.
CHALLENGES TO IMPLEMENTATION
Patients may not be eager to supplement with viscous fiber
The biggest challenge to implementation is patient compliance. Some forms of supplemental fiber are less palatable than others and may cause unpleasant gastrointestinal adverse effects, which may be an impediment for some patients. Cost may also be an issue for some patients. Diabetes medications can be expensive; however, they are often covered, at least partially, by medical insurance. Over-the-counter supplements are unlikely to be covered for most patients.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Jovanovski E, Khayyat R, Zurbau A, et al. Should viscous fiber supplements be considered in diabetes control? Results from a systematic review and meta-analysis of randomized controlled tria ls. Diabetes Care. 2019;42:755-766. Published correction appears in Diabetes Care. 2019;42:1604.
2. CDC. National Diabetes Statistics Report, 2020. Estimates of Diabetes and Its Burden in the United States. Atlanta, GA: Centers for Disease Control and Prevention, US Dept of Health and Human Services; 2020.
3. American Diabetes Association. Economic costs of diabetes in the U.S. in 2017. Diabetes Care. 2018;41:917-928.
4. Evert AB, Dennison M, Gardner CD, et al. Nutrition therapy for adults with diabetes or prediabetes: a consensus report. Diabetes Care. 2019;42:731-754.
5. American Diabetes Association. 5. Lifestyle management: Standards of Medical Care in Diabetes—2019. Diabetes Care. 2019;42(suppl 1):S46-S60.
6. Post RE, Mainous AG III, King DE, et al. Dietary fiber for the treatment of type 2 diabetes mellitus: a meta-analysis. J Am Board Fam Med. 2012;25:16-23.
7. Jenkins DJA, Kendall CWC, Augustin LSA, et al. Effect of legumes as part of a low glycemic index diet on glycemic control and cardiovascular risk factors in type 2 diabetes mellitus: a randomized controlled trial. Arch Intern Med. 2012;172:1653-1660.
ILLUSTRATIVE CASE
A 57-year-old man who was given a diagnosis of T2D a year ago presents for an office visit. His hemoglobin A1C level at diagnosis was 8.3%. He is otherwise healthy and has been adhering well to a plan of metformin 1000 mg twice daily, regular exercise, and a low-carbohydrate diet you recommended. His most recent hemoglobin A1C is 7.3%. He is pleased with his progress, so he is discouraged when you tell him that he is not yet at goal. He asks if there are other things that he can do to further lower his hemoglobin A1C. What can you recommend for him?
According to the National Diabetes Statistics Report, 2020 from the Centers for Disease Control and Prevention, approximately 34.1 million US adults ≥ 18 years of age (13% of the adult population) have diabetes, 50% of whom have a hemoglobin A1C > 7%. The report also states that approximately 88 million US adults—more than one-third of the population—have prediabetes.2
The American Diabetes Association (ADA) estimated that diabetes-related health care costs in the United States for 2017 totaled $237 billion, an increase of 26% from 2012. More than $30 billion of this expense comes directly from diabetes medications; the remainder of these costs are related to lost wages, clinic visits, hospitalizations, and treatment for diabetic complications and comorbidities. After controlling for age and gender, medical expenditures for people with diabetes are 2.3 times higher than for those without diabetes.3
The 2019
STUDY SUMMARY
Effect on A1C exceeded the FDA threshold for new drugs
This systematic review and meta-analysis searched MEDLINE, Embase, and the Cochrane Central Register of Controlled Trials to identify randomized controlled trials that studied the effects of viscous fiber supplementation on glycemic control in patients with T2D. Eligible studies included those that: (1) had a duration ≥ 3 weeks; (2) allowed isolation of the viscous fiber effects; and (3) reported at least 1 of the following glycemic measures: hemoglobin A1C (n = 1148 patients), fasting glucose (n = 1394), fasting insulin (n = 228),
Data were pooled using the generic inverse variance method and expressed as mean difference (MD) with 95% confidence intervals (CIs). Heterogeneity was assessed and quantified (Cochran Q and I2 statistics, respectively). I2 ≥ 50% indicates substantial heterogeneity. The Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach was used to evaluate the overall strength of evidence.
Twenty-eight eligible studies were compared. The median age of included patients was 60 years. The median dose of viscous fiber was 13.1 g/d (range, 2.55-21). Viscous fiber type varied between the studies and included psyllium, guar gum, β-glucan, and konjac, and was consumed in powder, tablet, capsule, and limited food-based forms (in 1 of the included studies). The median trial duration was 8 weeks, with 11 trials lasting ≥ 12 weeks.
Continue to: The study found...
The study found moderate-grade evidence that, when added to standard of care, viscous fiber supplementation reduced hemoglobin A1C (MD = –0.58%; 95% CI, –0.88 to –0.28; P = .0002; I2 = 91%), fasting glucose (MD = –14.8 mg/dL; 95% CI, –23.8 to –5.58; P = .001; I2 = 92%), and HOMA-IR (MD = –1.89; 95% CI, –3.45 to –0.33; P = .02; I2 = 94%) compared with control. The effect on hemoglobin A1C exceeds the ≥ 0.3% threshold established by the US Food and Drug Administration for new antihyperglycemic drug development. There was no significant effect on fasting insulin or fructosamine, although the sample size for fructosamine was small. No significant evidence of a dose-dependent response effect was found. The studies had substantial heterogeneity. No evaluation of potential or real harm was noted in the analysis.
WHAT’S NEW
Potential glycemic benefit without large dietary increase
The glucose-lowering effects of increased fiber intake have often been hypothesized, but this meta-analysis is the first to focus specifically on the effect of viscous fiber supplements in patients with T2D. Prior meta-analyses, including those cited in the 2019 ADA recommendations mentioned above, included primarily whole-food dietary sources of fiber in the treatment arms and generally had more modest effects on outcomes.4,6,7
By focusing on viscous fiber supplements, this study isolated the effect of these supplements vs fiber-rich dietary changes. It illustrates a greater potential benefit with supplements than whole-food dietary sources of fiber, and at a lower dose of fiber than was seen in prior studies without requiring substantial increases in caloric intake. Viscous fiber supplementation is a potential adjunct to the usual evidence-based standards of care for glycemic control in patients with T2D.
CAVEATS
Limited study durations may raise uncertainty about long-term benefits
This meta-analysis does have its limitations. The heterogeneity among the studies analyzed makes it difficult to establish a single recommendation regarding dose, type, and brand of fiber to be used. Only 11 of the 28 studies lasted longer than 12 weeks, with a median duration of 8 weeks, making any long-term effects on hemoglobin A1C unknown. No adverse effects or reactions were described to evaluate safety and tolerability of the viscous fiber supplementation. No patient-oriented outcomes were reported.
CHALLENGES TO IMPLEMENTATION
Patients may not be eager to supplement with viscous fiber
The biggest challenge to implementation is patient compliance. Some forms of supplemental fiber are less palatable than others and may cause unpleasant gastrointestinal adverse effects, which may be an impediment for some patients. Cost may also be an issue for some patients. Diabetes medications can be expensive; however, they are often covered, at least partially, by medical insurance. Over-the-counter supplements are unlikely to be covered for most patients.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
A 57-year-old man who was given a diagnosis of T2D a year ago presents for an office visit. His hemoglobin A1C level at diagnosis was 8.3%. He is otherwise healthy and has been adhering well to a plan of metformin 1000 mg twice daily, regular exercise, and a low-carbohydrate diet you recommended. His most recent hemoglobin A1C is 7.3%. He is pleased with his progress, so he is discouraged when you tell him that he is not yet at goal. He asks if there are other things that he can do to further lower his hemoglobin A1C. What can you recommend for him?
According to the National Diabetes Statistics Report, 2020 from the Centers for Disease Control and Prevention, approximately 34.1 million US adults ≥ 18 years of age (13% of the adult population) have diabetes, 50% of whom have a hemoglobin A1C > 7%. The report also states that approximately 88 million US adults—more than one-third of the population—have prediabetes.2
The American Diabetes Association (ADA) estimated that diabetes-related health care costs in the United States for 2017 totaled $237 billion, an increase of 26% from 2012. More than $30 billion of this expense comes directly from diabetes medications; the remainder of these costs are related to lost wages, clinic visits, hospitalizations, and treatment for diabetic complications and comorbidities. After controlling for age and gender, medical expenditures for people with diabetes are 2.3 times higher than for those without diabetes.3
The 2019
STUDY SUMMARY
Effect on A1C exceeded the FDA threshold for new drugs
This systematic review and meta-analysis searched MEDLINE, Embase, and the Cochrane Central Register of Controlled Trials to identify randomized controlled trials that studied the effects of viscous fiber supplementation on glycemic control in patients with T2D. Eligible studies included those that: (1) had a duration ≥ 3 weeks; (2) allowed isolation of the viscous fiber effects; and (3) reported at least 1 of the following glycemic measures: hemoglobin A1C (n = 1148 patients), fasting glucose (n = 1394), fasting insulin (n = 228),
Data were pooled using the generic inverse variance method and expressed as mean difference (MD) with 95% confidence intervals (CIs). Heterogeneity was assessed and quantified (Cochran Q and I2 statistics, respectively). I2 ≥ 50% indicates substantial heterogeneity. The Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach was used to evaluate the overall strength of evidence.
Twenty-eight eligible studies were compared. The median age of included patients was 60 years. The median dose of viscous fiber was 13.1 g/d (range, 2.55-21). Viscous fiber type varied between the studies and included psyllium, guar gum, β-glucan, and konjac, and was consumed in powder, tablet, capsule, and limited food-based forms (in 1 of the included studies). The median trial duration was 8 weeks, with 11 trials lasting ≥ 12 weeks.
Continue to: The study found...
The study found moderate-grade evidence that, when added to standard of care, viscous fiber supplementation reduced hemoglobin A1C (MD = –0.58%; 95% CI, –0.88 to –0.28; P = .0002; I2 = 91%), fasting glucose (MD = –14.8 mg/dL; 95% CI, –23.8 to –5.58; P = .001; I2 = 92%), and HOMA-IR (MD = –1.89; 95% CI, –3.45 to –0.33; P = .02; I2 = 94%) compared with control. The effect on hemoglobin A1C exceeds the ≥ 0.3% threshold established by the US Food and Drug Administration for new antihyperglycemic drug development. There was no significant effect on fasting insulin or fructosamine, although the sample size for fructosamine was small. No significant evidence of a dose-dependent response effect was found. The studies had substantial heterogeneity. No evaluation of potential or real harm was noted in the analysis.
WHAT’S NEW
Potential glycemic benefit without large dietary increase
The glucose-lowering effects of increased fiber intake have often been hypothesized, but this meta-analysis is the first to focus specifically on the effect of viscous fiber supplements in patients with T2D. Prior meta-analyses, including those cited in the 2019 ADA recommendations mentioned above, included primarily whole-food dietary sources of fiber in the treatment arms and generally had more modest effects on outcomes.4,6,7
By focusing on viscous fiber supplements, this study isolated the effect of these supplements vs fiber-rich dietary changes. It illustrates a greater potential benefit with supplements than whole-food dietary sources of fiber, and at a lower dose of fiber than was seen in prior studies without requiring substantial increases in caloric intake. Viscous fiber supplementation is a potential adjunct to the usual evidence-based standards of care for glycemic control in patients with T2D.
CAVEATS
Limited study durations may raise uncertainty about long-term benefits
This meta-analysis does have its limitations. The heterogeneity among the studies analyzed makes it difficult to establish a single recommendation regarding dose, type, and brand of fiber to be used. Only 11 of the 28 studies lasted longer than 12 weeks, with a median duration of 8 weeks, making any long-term effects on hemoglobin A1C unknown. No adverse effects or reactions were described to evaluate safety and tolerability of the viscous fiber supplementation. No patient-oriented outcomes were reported.
CHALLENGES TO IMPLEMENTATION
Patients may not be eager to supplement with viscous fiber
The biggest challenge to implementation is patient compliance. Some forms of supplemental fiber are less palatable than others and may cause unpleasant gastrointestinal adverse effects, which may be an impediment for some patients. Cost may also be an issue for some patients. Diabetes medications can be expensive; however, they are often covered, at least partially, by medical insurance. Over-the-counter supplements are unlikely to be covered for most patients.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Jovanovski E, Khayyat R, Zurbau A, et al. Should viscous fiber supplements be considered in diabetes control? Results from a systematic review and meta-analysis of randomized controlled tria ls. Diabetes Care. 2019;42:755-766. Published correction appears in Diabetes Care. 2019;42:1604.
2. CDC. National Diabetes Statistics Report, 2020. Estimates of Diabetes and Its Burden in the United States. Atlanta, GA: Centers for Disease Control and Prevention, US Dept of Health and Human Services; 2020.
3. American Diabetes Association. Economic costs of diabetes in the U.S. in 2017. Diabetes Care. 2018;41:917-928.
4. Evert AB, Dennison M, Gardner CD, et al. Nutrition therapy for adults with diabetes or prediabetes: a consensus report. Diabetes Care. 2019;42:731-754.
5. American Diabetes Association. 5. Lifestyle management: Standards of Medical Care in Diabetes—2019. Diabetes Care. 2019;42(suppl 1):S46-S60.
6. Post RE, Mainous AG III, King DE, et al. Dietary fiber for the treatment of type 2 diabetes mellitus: a meta-analysis. J Am Board Fam Med. 2012;25:16-23.
7. Jenkins DJA, Kendall CWC, Augustin LSA, et al. Effect of legumes as part of a low glycemic index diet on glycemic control and cardiovascular risk factors in type 2 diabetes mellitus: a randomized controlled trial. Arch Intern Med. 2012;172:1653-1660.
1. Jovanovski E, Khayyat R, Zurbau A, et al. Should viscous fiber supplements be considered in diabetes control? Results from a systematic review and meta-analysis of randomized controlled tria ls. Diabetes Care. 2019;42:755-766. Published correction appears in Diabetes Care. 2019;42:1604.
2. CDC. National Diabetes Statistics Report, 2020. Estimates of Diabetes and Its Burden in the United States. Atlanta, GA: Centers for Disease Control and Prevention, US Dept of Health and Human Services; 2020.
3. American Diabetes Association. Economic costs of diabetes in the U.S. in 2017. Diabetes Care. 2018;41:917-928.
4. Evert AB, Dennison M, Gardner CD, et al. Nutrition therapy for adults with diabetes or prediabetes: a consensus report. Diabetes Care. 2019;42:731-754.
5. American Diabetes Association. 5. Lifestyle management: Standards of Medical Care in Diabetes—2019. Diabetes Care. 2019;42(suppl 1):S46-S60.
6. Post RE, Mainous AG III, King DE, et al. Dietary fiber for the treatment of type 2 diabetes mellitus: a meta-analysis. J Am Board Fam Med. 2012;25:16-23.
7. Jenkins DJA, Kendall CWC, Augustin LSA, et al. Effect of legumes as part of a low glycemic index diet on glycemic control and cardiovascular risk factors in type 2 diabetes mellitus: a randomized controlled trial. Arch Intern Med. 2012;172:1653-1660.
PRACTICE CHANGER
Unless contraindicated, recommend viscous fiber supplementation to your patients with type 2 diabetes (T2D), in addition to the usual evidence-based standards of care, to improve markers of glycemic control.
STRENGTH OF RECOMMENDATION
C: Based on a meta-analysis and systematic review of 28 randomized controlled trials, without discussion of patient-oriented outcomes.1
Jovanovski E, Khayyat R, Zurbau A, et al. Should viscous fiber supplements be considered in diabetes control? Results from a systematic review and meta-analysis of randomized controlled trials. Diabetes Care. 2019;42:755-766. Published correction appears in Diabetes Care. 2019;42:1604.
Atopic dermatitis: More than just a rash
Atopic dermatitis (AD), also known as eczema, is a chronic inflammatory skin condition that is well known for its relapsing, pruritic rash in children and adults. Less recognized are its associated conditions—allergic rhinitis, asthma, food allergies, attention-deficit/hyperactivity disorder (ADHD), depression, and anxiety—and its burden on patients and their families. In fact, families that have children with AD report lower overall quality of life than those with otherwise healthy children.1 Given AD’s prevalence across age groups and its effect on the family, family physicians are uniquely positioned to diagnose, care for, and counsel patients with AD and its associated maladies.
The prevalence and pathogenesis of AD
AD affects up to 20% of children and 5% of adults in the United States.2 AD typically manifests before a child reaches age 5 (often in the first 6 months of life), and it is slightly more common in females (1.3:1). A family history of atopy (eczema, asthma, allergic rhinitis) is common. In fact, children with one atopic parent have a 2- to 3-fold increased risk of atopic dermatitis; those with 2 atopic parents have a 3- to 5-fold increased risk.3
The pathophysiology of AD is complex, culminating in impaired barrier function of the skin and transepidermal water loss resulting in dry and inflamed skin. Additionally, alterations in a cell-mediated immune response leading to an immunoglobulin (Ig) E-mediated hypersensitivity is also theorized to play a role in the development of AD.
Signs and symptoms
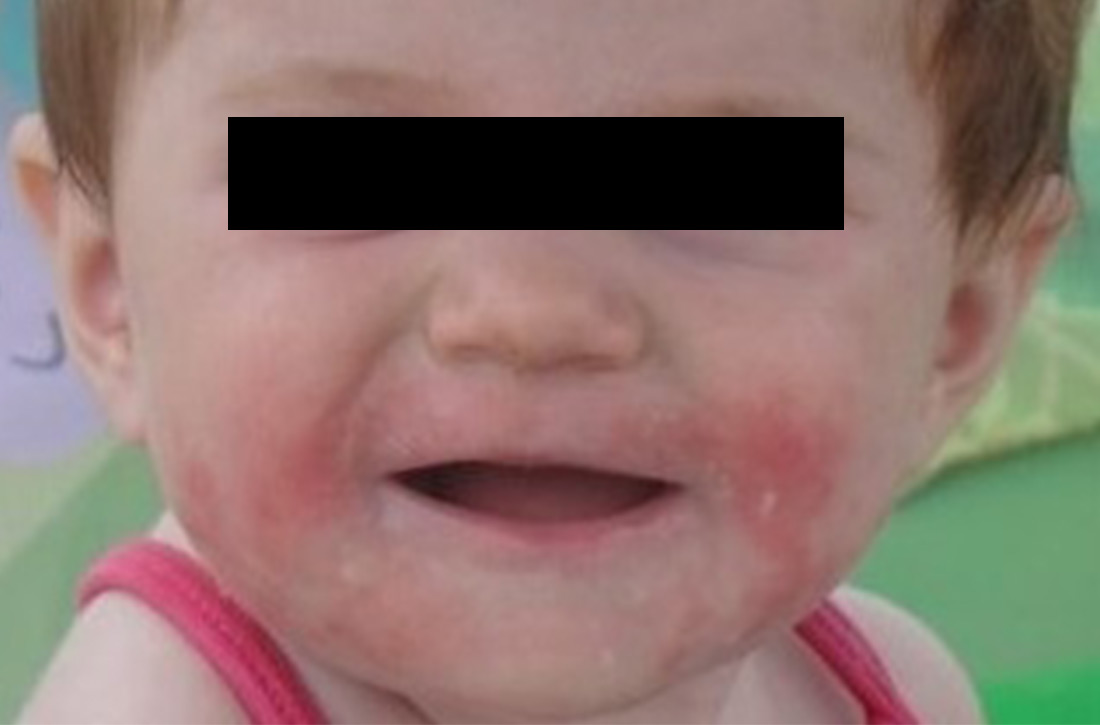
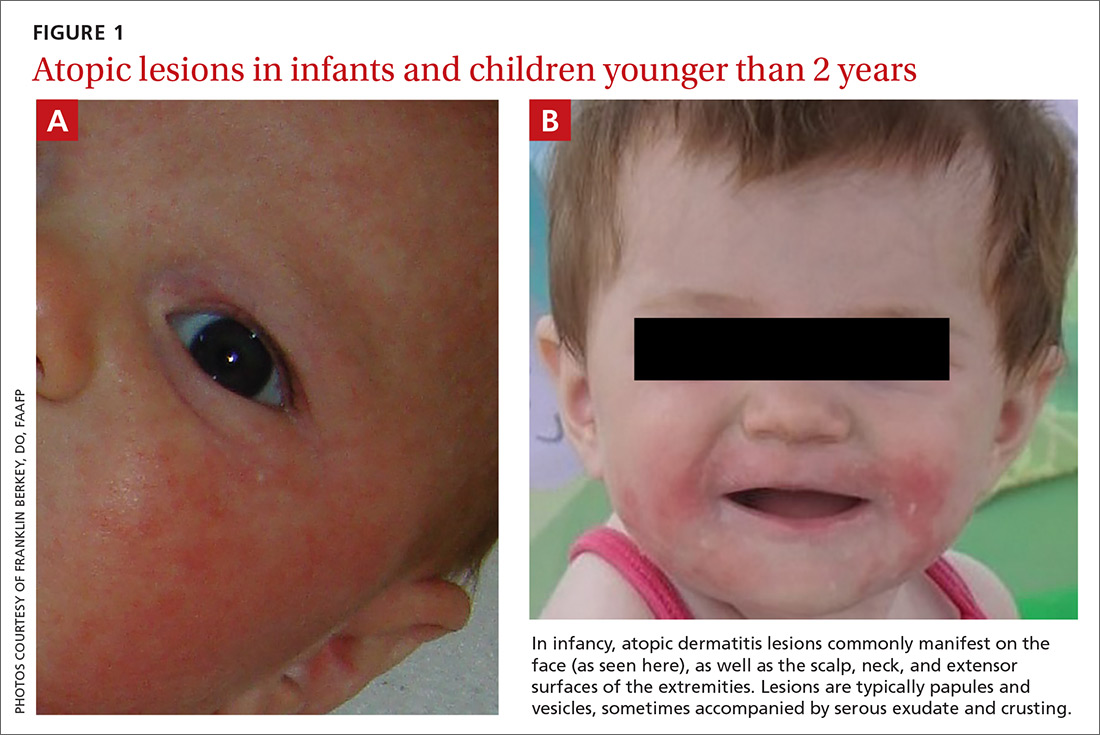
Signs at birth. Physical signs of atopic dermatitis typically appear between birth and 6 months. In infancy, lesions generally occur on the scalp, face (FIGURES 1A and 1B), neck, and extensor surfaces of the extremities. Lesions are typically papules and vesicles, sometimes accompanied by serous exudate and crusting. Eczematous lesions typically spare the groin and diaper area, and their presence in this area should raise suspicion for an alternative diagnosis.
Beginning at age 2 years, eczematous lesions are more commonly limited to the folds of the flexor surfaces. Instead of the weeping and crusting lesions seen in infancy, eczema in older children manifests as dry, lichenified papules and plaques in areas that are typically affected in adults: the wrist, hands, ankles, and popliteal and antecubital fossa.2
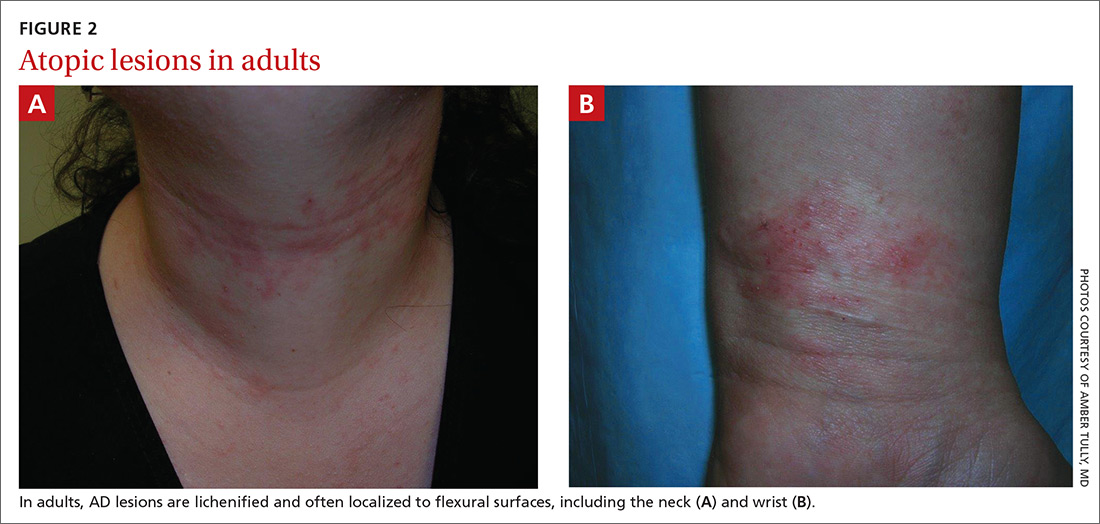
Although lesions in adults are similar to those of childhood, they may manifest in a more localized area (hand or eyelid, for example). As is the case in childhood, the lesions are dry, sometimes lichenified, and found on the flexural surfaces (FIGURES 2A and 2B).2
Symptom triggers are unproven
While anecdotal reports cite various triggers for AD flares, a systematic review found little scientific evidence to substantiate identifiable triggers.4 Triggers often cited and studied are foods, dust mite exposure, airborne allergens, detergents, sunlight, fabrics, bacterial infections, and stress. While as many as one-third of people with AD who also have confirmed dust mite allergy report worsening of symptoms when exposed to dust, a Cochrane review of 7 randomized controlled trials totaling 324 adults and children with eczema found that efforts at dust mite mitigation (laundering of bed covers, increased vacuuming, spraying for mites) were not effective in reducing symptoms.5
Continue to: How quality of life diminishes with AD
How quality of life diminishes with AD
AD substantially lessens quality of life. For children, the most distressing physical symptoms include itching that inhibits sleep and provokes scratching, pain, and bleeding. Emotional distress can cause irritability, crying, and uncooperativeness with treatments. Parents also report that they frequently restrict their children from activities, such as playing in the heat or swimming, that may lead to worsening of their eczema.6
The loss of sleep associated with AD is not completely understood but is likely multifactorial. Pruritus and scratching leading to sleeplessness is the most obvious culprit, but an altered circadian rhythm, immune system response, and changes in skin physiology are also likely factors.7 Whatever the cause, sleep disturbance is reported in as many as 60% of patients with AD, and the degree of sleep disturbance is proportional to increases in disease severity and worsening of quality-of-life scores.8 Lost sleep is not limited to patients; parents of children with AD also report significant loss of sleep and subsequent decreased work productivity and quality of life.9
Children with AD are often the target of bullying.10 A 2015 survey by the National Eczema Association indicates that 1 in 5 children reported being bullied due to their AD.11
Associated conditions and comorbidities
AD increases patients’ risks for other illnesses, due either to their underlying atopy or to the effects of
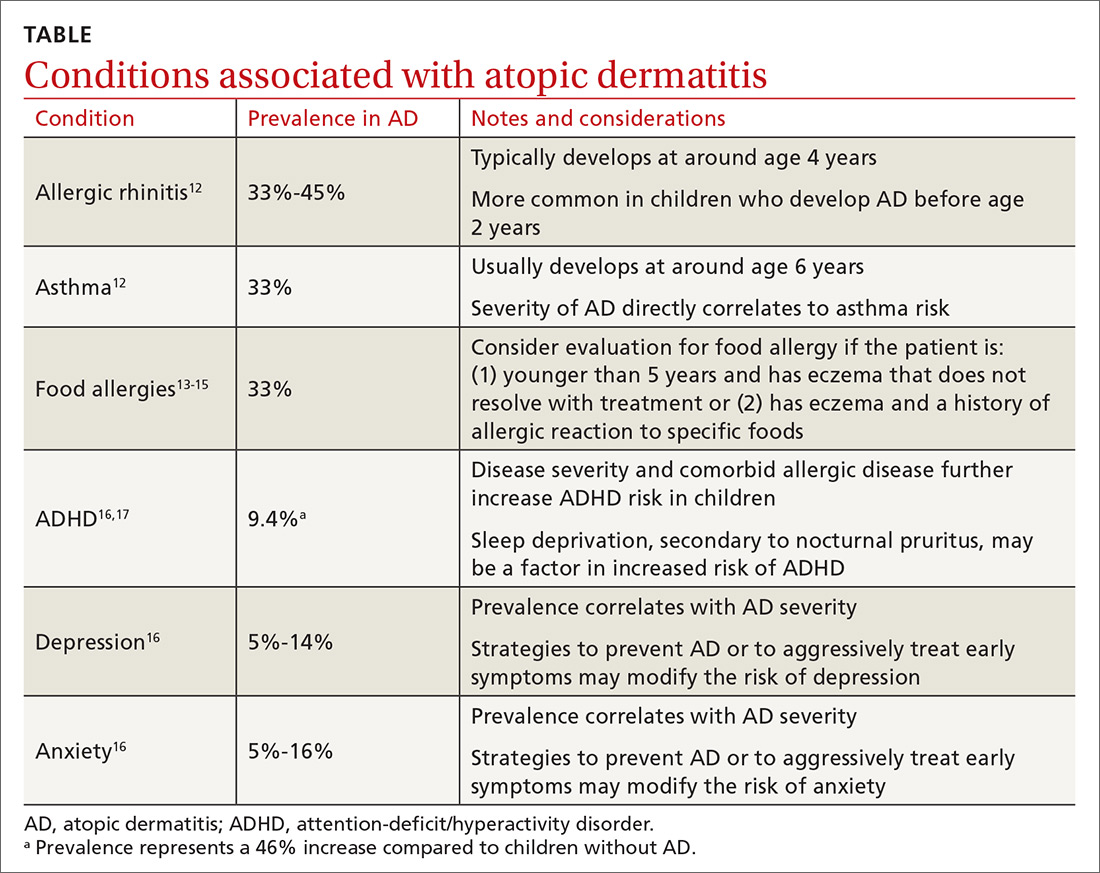
Atopic march
Atopic march—the clinical succession of AD, allergic rhinitis, and asthma—is a well-established clinical progression. The presence of all 3 conditions appears to be more common in children diagnosed with AD before 2 years of age.12 Typically, allergic rhinitis manifests at around age 4, and asthma develops between ages 6 and 8. The severity of AD predicts progression. Compared with an 8% chance of asthma developing among the general population, children with mild AD have a 20% to 30% chance of developing asthma, and those with severe AD have about a 70% chance.12
Continue to: Food allergies
Food allergies
Patients with AD are at higher risk for food-induced anaphylaxis, with up to one-third of AD patients having an IgE-mediated food allergy.13 While it is theorized that the impaired skin barrier of an atopic child may allow for early sensitization and allergy development, a landmark 2015 study demonstrated that early allergen introduction (specifically, peanuts) may serve as a preventive strategy in those at high risk of food allergies.14 Current guidelines recommend that physicians be aware of the increased possibility of food allergies in those with AD, and consider evaluating a child for milk, egg, peanut, wheat, and soy allergy if the child is younger than 5 years and has eczema that does not resolve with treatment, or has eczema and a history of an allergic reaction to a specific food.15
Interestingly, despite the strong association between AD and food allergies, it is not clear that food allergies trigger atopic flares; as such, elimination diets are not universally recommended in those without a proven food allergy.
Psychiatric diagnoses
Children with AD have an increased prevalence of several psychiatric conditions, including ADHD, depression, anxiety, conduct disorder, and autism when compared with peers who do not have AD, and the probability correlates with the severity of AD
What we do know is that one of the strongest associations between AD and a psychiatric condition is with ADHD, with a recent pooled meta-analysis showing a 46% increase in risk.17 The incidence of depression among children with AD appears to correlate with the severity of AD symptoms: estimated at 5% with mild AD, 7% with moderate disease, and 14% with severe disease (compared with 3% without AD). Similar incremental increases are seen when correlating AD and anxiety.16
Nonpharmacologic care
Bathing
Bathing habits are critical to controlling AD. While bathing serves to both hydrate the skin and remove allergens, the water’s evaporation off the skin surface can lead to increased transepidermal water loss. Combining bathing and immediate application of a moisturizer improves skin hydration in patients with AD vs bathing alone.18 Thus, consensus guidelines recommend once-daily bathing (bath or shower) to remove scale and crust, followed by immediate application of a moisturizing emollient.19
Continue to: Emollients
Emollients
Application of moisturizing emollients is the mainstay of nonpharmacologic care of AD, and there is strong evidence that their regimented use reduces disease burden and the need for prescription treatment.19 Emollient creams and ointments help retain moisture and improve the skin’s barrier. While ointments may provide a better barrier, patients tend to prefer creams as they are less greasy than ointments.
Emollient therapy may also help prevent development of AD, especially in those infants thought to be at high risk with a family history of atopy. In a multinational randomized controlled trial, infants who received daily full-body application of emollient beginning at 3 weeks of life were significantly less likely than controls to develop AD by 6 months.20 While the mechanism of action is not clearly understood, it is believed that early emollient use prevents skin dehydration and maintains the skin’s barrier integrity, thus decreasing allergen epidermal penetration and subsequent inflammation.
Bleach bath
A bleach bath, prepared by adding 1/2 cup of unconcentrated bleach (5.25% sodium hypochlorite) to a standard 40-gallon bathtub, produces a chlorine mixture equivalent to an average swimming pool. Soaking in a bleach bath for 10 minutes once or twice weekly is thought to reduce inflammation and bacteria on the skin, but studies of its efficacy in improving atopic symptoms are mixed.
In a pooled analysis of 5 studies evaluating bleach baths vs standard baths, there was no significant difference in disease severity at 4 weeks.21 Thus, while bleach baths were effective in decreasing disease severity, they appeared to be no more effective than a standard water bath.21 Bleach baths may be helpful, however, in cases of moderate-to-severe disease with frequent bacterial infections.19
Pharmacologic therapy
Steroids
For symptoms refractory to nonpharmacologic skin care, topical steroids are the initial pharmacologic treatment for AD.19 Choose steroid potency based on symptom severity and disease location. Low- to medium-potency is appropriate for mild disease, and medium- to high-potency is useful for moderate-to-severe symptoms. High-potency steroids are generally avoided on the face and skin folds; however, they can be used for short periods in these areas to induce remission. They must then be quickly tapered and discontinued.
Continue to: Frequency
Frequency. Topical corticosteroids are typically applied twice daily, although recent studies indicate that once-daily application is just as efficacious.22 In addition to treatment of an acute flare, topical steroids are useful as maintenance therapy for patients with recurrent outbreaks in the same anatomical site. Guidelines suggest once- or twice-weekly application of a medium-potency steroid to prolong time between flares.19
For children, a practical guide is for caregivers to apply the amount of steroid covering 1 adult fingertip to an area of the child’s skin equal to that of 2 adult palms.23 Topical steroids are generally well tolerated and have a good safety profile. Adverse effects are proportional to the amount and duration of use and include purpura, telangiectasias, striae, and skin atrophy. The risk of skin atrophy increases with higher potency steroids, occlusion (covering affected area after steroid application), use on thin-skinned areas, and older patient age.24
Reassure patients/parents about the safety of topical steroids, as fears regarding the potential adverse effects can limit compliance. In one study of 200 patients with AD, 72.5% of respondents expressed fear of using steroids on their own skin or that of their child, and 24% admitted being noncompliant with therapy based on these concerns.25
Treating flares. Oral steroids are sometimes needed to abort or control an AD flare in older children and adults. A tapering course of prednisone over 5 to 7 days, transitioning to medium- to high-dose topical steroids, may be needed to achieve symptom control.
Topical calcineurin inhibitors
Topical calcineurin inhibitors, including tacrolimus and pimecrolimus, are generally second-line therapy to topical corticosteroids. However, as nonsteroidal agents, topical calcineurin inhibitors do not cause skin atrophy and can be a first-line option in areas where atrophy is more common (face, eyelids, neck, and skin folds).26
Continue to: A Cochrane review found...
A Cochrane review found tacrolimus 0.1% to be better than low‐potency topical corticosteroids on the face and neck areas, while results were equivocal when compared with moderate‐potency topical corticosteroids on the trunk and extremities (no difference based on physician assessment, but marginal benefit favoring tacrolimus based on participant scoring).27 When compared head-to-head, tacrolimus was more effective than pimecrolimus, although tacrolimus has a higher rate of local irritation. The most common adverse effects are stinging and burning at the application site, although these adverse effects generally improve with repeated application.
There have been long-term safety concerns with topical calcineurin inhibitors—chiefly a 2006 Food and Drug Administration (FDA) black box warning regarding a possible link between topical calcineurin inhibitors and cancer. However, while there may be a slight increased risk of lymphoma in AD patients, a recent meta-analysis did not find an association between topical calcineurin inhibitors use and lymphoma.28 Given the initial concern—and pending additional data—the FDA currently recommends reserving topical calcineurin inhibitors for second-line therapy and only for the minimum amount of time to induce improvement. It also recommends avoiding their use in patients younger than 2 years and in those with compromised immune systems.
Cisaborole
Cisaborole, a topical phosphodiesterase 4 (PDE4) inhibitor, received FDA approval in 2016 for mild-to-moderate AD. By inhibiting PDE4, the drug limits inflammation. In a multicenter randomized trial, patients applying cisaborole 2% twice a day noted reductions in pruritus, inflammation, excoriation, and lichenification.29 Adverse effects are minimal and limited to application site irritation.
Systemic treatments
While beyond the care of a family physician, symptoms refractory to conservative, nonpharmacologic measures and combinations of topical pharmaceuticals can be treated with systemic immunomodulators such as cyclosporine, azathioprine, and methotrexate. Phototherapy is also effective in patients with more widespread skin involvement. Dupilumab, an injectable monoclonal antibody that binds to interleukin-4 receptor and inhibits inflammation, is approved to treat moderate-to-severe AD in adults.30
Ineffective therapies: Oral montelukast and probiotics
While oral antihistamines are frequently prescribed and used, there are no studies evaluating the use of antihistamines (H1) as monotherapy for AD.31 Nonetheless, while not altering the disease process, the sedative effect of antihistamines may palliate the nocturnal pruritus frequently associated with AD. Although nonsedating antihistamines may still have a role for atopic patients with concurrent seasonal and environmental allergies, there is no evidence to support their use in the treatment of AD.
Continue to: Data are limited...
Data are limited on the effectiveness of leukotriene receptor antagonists for AD, and all studies meeting inclusion for a Cochrane review assessed oral montelukast. The review found no benefit with the use of montelukast 10 mg in terms of severity of disease, pruritus, or need for topical steroids.32
A systematic review investigating the benefit of probiotics for the treatment of AD found no improvement in patient-rated eczema scores for quality of life.33 Additionally, a review of 11 randomized controlled trials including 596 participants found no evidence to suggest efficacy of fish oil, zinc, selenium, vitamin D, vitamin E, pyridoxine, sea buckthorn oil, hempseed oil, or sunflower oil in the treatment of AD.34
Education can reduce AD severity
Family physicians can be a source of education and support for patients and families of patients with AD. Support programs for adults with AD—including education, relaxation techniques, and cognitive behavioral therapy—have been shown to decrease disease severity.35 Comparable improvement in disease severity has been demonstrated in children with AD when similar education is provided to them and their families.
CORRESPONDENCE
Franklin Berkey, DO, Penn State Health, 1850 East Park Avenue, Suite 207, State College, PA 16803; fberkey@ pennstatehealth.psu.edu.
1. Carroll CL, Balkrishnan R, Feldman SR, et al. The burden of atopic dermatitis: impact on the patient, family, and society. Pediatr Dermatol. 2005;22:192-199.
2. Ahn C, Huang W. Clinical presentation of atopic dermatitis. In: Fortson E, Feldman SR, Stroud LC, eds. Management of Atopic Dermatitis: Methods and Challenges. Springer International Publishing; 2017:38-46.
3. Eichenfield LF, Tom WL, Chamblin SL, et al. Guidelines of care for the management of atopic dermatitis. Part 1: diagnosis and assessment of atopic dermatitis. J Am Acad Dermatol. 2014;70:338-351.
4. Langan SM, Williams HC. What causes worsening of eczema? A systematic review. Br J Dermatol. 2006;155:504-514.
5. Nankervis H, Pynn EV, Boyle RJ, et al. House dust mite reduction and avoidance measures for treating eczema. Cochrane Database Syst Rev. 2015:CD008426.
6. Chamlin SL, Frieden IJ, Williams ML, et al. Effects of atopic dermatitis on young American children and their families. Pediatrics. 2004;114:607-611.
7. Chang Y-S, Chiang B-L. Mechanism of sleep disturbance in children with atopic dermatitis and the role of the circadian rhythm and melatonin. Int J Mol Sci. 2016;17:462.
8. Camfferman D, Kennedy JD, Gold M, et al. Eczema and sleep and its relationship to daytime functioning in children. Sleep Med Rev. 2010;14:359-369.
9. Chamlin SL, Mattson CL, Frieden IJ, et al. The price of pruritus: sleep disturbance and cosleeping in atopic dermatitis. Arch Pediatr Adolesc Med. 2005;159:745-750.
10. Drucker AM, Wang AR, Li W-Q, et al. The burden of atopic dermatitis: summary of a report for the National Eczema Association. J Invest Dermatol. 2017;137:P26-P30.
11. National Eczema Association. Tools for school: addressing school bullying for kids with eczema. Accessed January 5, 2021. https://nationaleczema.org/children-with-eczema-experience-bullying/
12. Bantz SK, Zhu Z, Zhen T. The atopic march: progression from atopic dermatitis to allergic rhinitis and asthma. J Clin Cell Immunol. 2014;5:202
13. Laird M, Sicco KL. Defining and measuring the scope of atopic dermatitis. Adv Exp Med Biol. 2017;1027:93-104.
14. Du Toit G, Roberts G, Sayre PH, et al. Randomized trial of peanut consumption in infants at risk for peanut allergy. N Engl J Med. 2015;372:803-813.
15. Boyce JA, Assa’ad A, Burks AW, et al. Guidelines for the diagnosis and management of food allergy in the United States: report of the NIAID-sponsored expert panel. J Allergy Clin Immunol. 2010;126:S1–S58.
16. Yaghmaie P, Koudelka CW, Simpson EL. Mental health comorbidity in patients with atopic dermatitis. J Allergy Clin Immunol. 2013;131:428-433.
17. Strom MA, Fishbein AB, Paller AS, et al. Association between atopic dermatitis and attention deficit hyperactivity disorder in U.S. children and adults. Br J Dermatol. 2016;175:920-929.
18. Chiang C, Eichenfield LF. Quantitative assessment of combination bathing and moisturizing regimens on skin hydration in atopic dermatitis. Pediatr Dermatol. 2009;26:273-278.
19. Eichenfield LF, Tom WL, Berger TG, et al. Guidelines of care for the management of atopic dermatitis: section 2. Management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
20. Simpson EL, Chalmers JR, Hanifin JM, et al. Emollient enhancement of the skin barrier from birth offers effective atopic dermatitis prevention. J Allergy Clin Immunol. 2014;134:818-823.
21. Chopra R, Vakharia PP, Sacotte R, et al. Efficacy of bleach baths in reducing severity of atopic dermatitis: a systematic review and meta-analysis. Ann Allergy Asthma Immunol. 2017;119:435-440.
22. Williams HC. Established corticosteroid creams should be applied only once daily in patients with atopic eczema. BMJ. 2007;334:1272.
23. Long CC, Mills CM, Finlay AY. A practical guide to topical therapy in children. Br J Dermatol. 1998;138:293-296.
24. Callen J, Chamlin S, Eichenfield LF, et al. A systematic review of the safety of topical therapies for atopic dermatitis. Br J Dermatol. 2007;156:203-221.
25. Charman CR, Morris AD, Williams HC. Topical corticosteroid phobia in patients with atopic eczema. Br J Dermatol. 2000;142:931-936.
26. Ashcroft DM, Dimmock P, Garside R, et al. Efficacy and tolerability of topical pimecrolimus and tacrolimus in the treatment of atopic dermatitis: a meta-analysis of randomised controlled trials. BMJ. 2005;330:516.
27. Cury Martins J, Martins C, Aoki V, et al. Topical tacrolimus for atopic dermatitis. Cochrane Database Syst Rev. 2015:CD009864.
28. Legendre L, Barnetche T, Mazereeuw-Hautier J, et al. Risk of lymphoma in patients with atopic dermatitis and the role of topical treatment: a systematic review and meta-analysis. J Am Acad Dermatol. 2015;72:992-1002.
29. Paller AS, Tom WL, Lebwohl MG, et al. Efficacy and safety of crisaborole ointment, a novel, nonsteroidal phosphodiesterase 4 (PDE4) inhibitor for the topical treatment of atopic dermatitis (AD) in children and adults. J Am Acad Dermatol. 2016;75:494-503.
30. Dupilumab [package insert]. Tarrytown, NY: Regeneron Pharmaceuticals Inc; 2017.
31. van Zuuren EJ, Apfelbacher CJ, Fedorowicz Z, et al. No high level evidence to support the use of oral H1 antihistamines as monotherapy for eczema: a summary of a Cochrane systematic review. Syst Rev. 2014;3:25.
32. Ferguson L, Futamura M, Vakirlis E, et al. Leukotriene receptor antagonists for eczema. Cochrane Database Syst Rev. 2018:CD011224.
33. Makrgeorgou A, Leonardi-Bee J, Bath-Hextall FJ, et al. Probiotics for treating eczema. Cochrane Database Syst Rev. 2018:CD006135.
34. Bath-Hextall FJ, Jenkinson C, Humphreys R, et al. Dietary supplements for established atopic eczema. Cochrane Database Syst Rev. 2012:CD005205.
35. Sy W, Lamb AJ. Atopic dermatitis disease education. In: Fortson E, Feldman SR, Stroud LC, eds. Management of Atopic Dermatitis: Methods and Challenges. Springer International Publishing; 2017:179-184.
Atopic dermatitis (AD), also known as eczema, is a chronic inflammatory skin condition that is well known for its relapsing, pruritic rash in children and adults. Less recognized are its associated conditions—allergic rhinitis, asthma, food allergies, attention-deficit/hyperactivity disorder (ADHD), depression, and anxiety—and its burden on patients and their families. In fact, families that have children with AD report lower overall quality of life than those with otherwise healthy children.1 Given AD’s prevalence across age groups and its effect on the family, family physicians are uniquely positioned to diagnose, care for, and counsel patients with AD and its associated maladies.
The prevalence and pathogenesis of AD
AD affects up to 20% of children and 5% of adults in the United States.2 AD typically manifests before a child reaches age 5 (often in the first 6 months of life), and it is slightly more common in females (1.3:1). A family history of atopy (eczema, asthma, allergic rhinitis) is common. In fact, children with one atopic parent have a 2- to 3-fold increased risk of atopic dermatitis; those with 2 atopic parents have a 3- to 5-fold increased risk.3
The pathophysiology of AD is complex, culminating in impaired barrier function of the skin and transepidermal water loss resulting in dry and inflamed skin. Additionally, alterations in a cell-mediated immune response leading to an immunoglobulin (Ig) E-mediated hypersensitivity is also theorized to play a role in the development of AD.
Signs and symptoms
Signs at birth. Physical signs of atopic dermatitis typically appear between birth and 6 months. In infancy, lesions generally occur on the scalp, face (FIGURES 1A and 1B), neck, and extensor surfaces of the extremities. Lesions are typically papules and vesicles, sometimes accompanied by serous exudate and crusting. Eczematous lesions typically spare the groin and diaper area, and their presence in this area should raise suspicion for an alternative diagnosis.

Beginning at age 2 years, eczematous lesions are more commonly limited to the folds of the flexor surfaces. Instead of the weeping and crusting lesions seen in infancy, eczema in older children manifests as dry, lichenified papules and plaques in areas that are typically affected in adults: the wrist, hands, ankles, and popliteal and antecubital fossa.2
Although lesions in adults are similar to those of childhood, they may manifest in a more localized area (hand or eyelid, for example). As is the case in childhood, the lesions are dry, sometimes lichenified, and found on the flexural surfaces (FIGURES 2A and 2B).2

Symptom triggers are unproven
While anecdotal reports cite various triggers for AD flares, a systematic review found little scientific evidence to substantiate identifiable triggers.4 Triggers often cited and studied are foods, dust mite exposure, airborne allergens, detergents, sunlight, fabrics, bacterial infections, and stress. While as many as one-third of people with AD who also have confirmed dust mite allergy report worsening of symptoms when exposed to dust, a Cochrane review of 7 randomized controlled trials totaling 324 adults and children with eczema found that efforts at dust mite mitigation (laundering of bed covers, increased vacuuming, spraying for mites) were not effective in reducing symptoms.5
Continue to: How quality of life diminishes with AD
How quality of life diminishes with AD
AD substantially lessens quality of life. For children, the most distressing physical symptoms include itching that inhibits sleep and provokes scratching, pain, and bleeding. Emotional distress can cause irritability, crying, and uncooperativeness with treatments. Parents also report that they frequently restrict their children from activities, such as playing in the heat or swimming, that may lead to worsening of their eczema.6
The loss of sleep associated with AD is not completely understood but is likely multifactorial. Pruritus and scratching leading to sleeplessness is the most obvious culprit, but an altered circadian rhythm, immune system response, and changes in skin physiology are also likely factors.7 Whatever the cause, sleep disturbance is reported in as many as 60% of patients with AD, and the degree of sleep disturbance is proportional to increases in disease severity and worsening of quality-of-life scores.8 Lost sleep is not limited to patients; parents of children with AD also report significant loss of sleep and subsequent decreased work productivity and quality of life.9
Children with AD are often the target of bullying.10 A 2015 survey by the National Eczema Association indicates that 1 in 5 children reported being bullied due to their AD.11
Associated conditions and comorbidities
AD increases patients’ risks for other illnesses, due either to their underlying atopy or to the effects of

Atopic march
Atopic march—the clinical succession of AD, allergic rhinitis, and asthma—is a well-established clinical progression. The presence of all 3 conditions appears to be more common in children diagnosed with AD before 2 years of age.12 Typically, allergic rhinitis manifests at around age 4, and asthma develops between ages 6 and 8. The severity of AD predicts progression. Compared with an 8% chance of asthma developing among the general population, children with mild AD have a 20% to 30% chance of developing asthma, and those with severe AD have about a 70% chance.12
Continue to: Food allergies
Food allergies
Patients with AD are at higher risk for food-induced anaphylaxis, with up to one-third of AD patients having an IgE-mediated food allergy.13 While it is theorized that the impaired skin barrier of an atopic child may allow for early sensitization and allergy development, a landmark 2015 study demonstrated that early allergen introduction (specifically, peanuts) may serve as a preventive strategy in those at high risk of food allergies.14 Current guidelines recommend that physicians be aware of the increased possibility of food allergies in those with AD, and consider evaluating a child for milk, egg, peanut, wheat, and soy allergy if the child is younger than 5 years and has eczema that does not resolve with treatment, or has eczema and a history of an allergic reaction to a specific food.15
Interestingly, despite the strong association between AD and food allergies, it is not clear that food allergies trigger atopic flares; as such, elimination diets are not universally recommended in those without a proven food allergy.
Psychiatric diagnoses
Children with AD have an increased prevalence of several psychiatric conditions, including ADHD, depression, anxiety, conduct disorder, and autism when compared with peers who do not have AD, and the probability correlates with the severity of AD
What we do know is that one of the strongest associations between AD and a psychiatric condition is with ADHD, with a recent pooled meta-analysis showing a 46% increase in risk.17 The incidence of depression among children with AD appears to correlate with the severity of AD symptoms: estimated at 5% with mild AD, 7% with moderate disease, and 14% with severe disease (compared with 3% without AD). Similar incremental increases are seen when correlating AD and anxiety.16
Nonpharmacologic care
Bathing
Bathing habits are critical to controlling AD. While bathing serves to both hydrate the skin and remove allergens, the water’s evaporation off the skin surface can lead to increased transepidermal water loss. Combining bathing and immediate application of a moisturizer improves skin hydration in patients with AD vs bathing alone.18 Thus, consensus guidelines recommend once-daily bathing (bath or shower) to remove scale and crust, followed by immediate application of a moisturizing emollient.19
Continue to: Emollients
Emollients
Application of moisturizing emollients is the mainstay of nonpharmacologic care of AD, and there is strong evidence that their regimented use reduces disease burden and the need for prescription treatment.19 Emollient creams and ointments help retain moisture and improve the skin’s barrier. While ointments may provide a better barrier, patients tend to prefer creams as they are less greasy than ointments.
Emollient therapy may also help prevent development of AD, especially in those infants thought to be at high risk with a family history of atopy. In a multinational randomized controlled trial, infants who received daily full-body application of emollient beginning at 3 weeks of life were significantly less likely than controls to develop AD by 6 months.20 While the mechanism of action is not clearly understood, it is believed that early emollient use prevents skin dehydration and maintains the skin’s barrier integrity, thus decreasing allergen epidermal penetration and subsequent inflammation.
Bleach bath
A bleach bath, prepared by adding 1/2 cup of unconcentrated bleach (5.25% sodium hypochlorite) to a standard 40-gallon bathtub, produces a chlorine mixture equivalent to an average swimming pool. Soaking in a bleach bath for 10 minutes once or twice weekly is thought to reduce inflammation and bacteria on the skin, but studies of its efficacy in improving atopic symptoms are mixed.
In a pooled analysis of 5 studies evaluating bleach baths vs standard baths, there was no significant difference in disease severity at 4 weeks.21 Thus, while bleach baths were effective in decreasing disease severity, they appeared to be no more effective than a standard water bath.21 Bleach baths may be helpful, however, in cases of moderate-to-severe disease with frequent bacterial infections.19
Pharmacologic therapy
Steroids
For symptoms refractory to nonpharmacologic skin care, topical steroids are the initial pharmacologic treatment for AD.19 Choose steroid potency based on symptom severity and disease location. Low- to medium-potency is appropriate for mild disease, and medium- to high-potency is useful for moderate-to-severe symptoms. High-potency steroids are generally avoided on the face and skin folds; however, they can be used for short periods in these areas to induce remission. They must then be quickly tapered and discontinued.
Continue to: Frequency
Frequency. Topical corticosteroids are typically applied twice daily, although recent studies indicate that once-daily application is just as efficacious.22 In addition to treatment of an acute flare, topical steroids are useful as maintenance therapy for patients with recurrent outbreaks in the same anatomical site. Guidelines suggest once- or twice-weekly application of a medium-potency steroid to prolong time between flares.19
For children, a practical guide is for caregivers to apply the amount of steroid covering 1 adult fingertip to an area of the child’s skin equal to that of 2 adult palms.23 Topical steroids are generally well tolerated and have a good safety profile. Adverse effects are proportional to the amount and duration of use and include purpura, telangiectasias, striae, and skin atrophy. The risk of skin atrophy increases with higher potency steroids, occlusion (covering affected area after steroid application), use on thin-skinned areas, and older patient age.24
Reassure patients/parents about the safety of topical steroids, as fears regarding the potential adverse effects can limit compliance. In one study of 200 patients with AD, 72.5% of respondents expressed fear of using steroids on their own skin or that of their child, and 24% admitted being noncompliant with therapy based on these concerns.25
Treating flares. Oral steroids are sometimes needed to abort or control an AD flare in older children and adults. A tapering course of prednisone over 5 to 7 days, transitioning to medium- to high-dose topical steroids, may be needed to achieve symptom control.
Topical calcineurin inhibitors
Topical calcineurin inhibitors, including tacrolimus and pimecrolimus, are generally second-line therapy to topical corticosteroids. However, as nonsteroidal agents, topical calcineurin inhibitors do not cause skin atrophy and can be a first-line option in areas where atrophy is more common (face, eyelids, neck, and skin folds).26
Continue to: A Cochrane review found...
A Cochrane review found tacrolimus 0.1% to be better than low‐potency topical corticosteroids on the face and neck areas, while results were equivocal when compared with moderate‐potency topical corticosteroids on the trunk and extremities (no difference based on physician assessment, but marginal benefit favoring tacrolimus based on participant scoring).27 When compared head-to-head, tacrolimus was more effective than pimecrolimus, although tacrolimus has a higher rate of local irritation. The most common adverse effects are stinging and burning at the application site, although these adverse effects generally improve with repeated application.
There have been long-term safety concerns with topical calcineurin inhibitors—chiefly a 2006 Food and Drug Administration (FDA) black box warning regarding a possible link between topical calcineurin inhibitors and cancer. However, while there may be a slight increased risk of lymphoma in AD patients, a recent meta-analysis did not find an association between topical calcineurin inhibitors use and lymphoma.28 Given the initial concern—and pending additional data—the FDA currently recommends reserving topical calcineurin inhibitors for second-line therapy and only for the minimum amount of time to induce improvement. It also recommends avoiding their use in patients younger than 2 years and in those with compromised immune systems.
Cisaborole
Cisaborole, a topical phosphodiesterase 4 (PDE4) inhibitor, received FDA approval in 2016 for mild-to-moderate AD. By inhibiting PDE4, the drug limits inflammation. In a multicenter randomized trial, patients applying cisaborole 2% twice a day noted reductions in pruritus, inflammation, excoriation, and lichenification.29 Adverse effects are minimal and limited to application site irritation.
Systemic treatments
While beyond the care of a family physician, symptoms refractory to conservative, nonpharmacologic measures and combinations of topical pharmaceuticals can be treated with systemic immunomodulators such as cyclosporine, azathioprine, and methotrexate. Phototherapy is also effective in patients with more widespread skin involvement. Dupilumab, an injectable monoclonal antibody that binds to interleukin-4 receptor and inhibits inflammation, is approved to treat moderate-to-severe AD in adults.30
Ineffective therapies: Oral montelukast and probiotics
While oral antihistamines are frequently prescribed and used, there are no studies evaluating the use of antihistamines (H1) as monotherapy for AD.31 Nonetheless, while not altering the disease process, the sedative effect of antihistamines may palliate the nocturnal pruritus frequently associated with AD. Although nonsedating antihistamines may still have a role for atopic patients with concurrent seasonal and environmental allergies, there is no evidence to support their use in the treatment of AD.
Continue to: Data are limited...
Data are limited on the effectiveness of leukotriene receptor antagonists for AD, and all studies meeting inclusion for a Cochrane review assessed oral montelukast. The review found no benefit with the use of montelukast 10 mg in terms of severity of disease, pruritus, or need for topical steroids.32
A systematic review investigating the benefit of probiotics for the treatment of AD found no improvement in patient-rated eczema scores for quality of life.33 Additionally, a review of 11 randomized controlled trials including 596 participants found no evidence to suggest efficacy of fish oil, zinc, selenium, vitamin D, vitamin E, pyridoxine, sea buckthorn oil, hempseed oil, or sunflower oil in the treatment of AD.34
Education can reduce AD severity
Family physicians can be a source of education and support for patients and families of patients with AD. Support programs for adults with AD—including education, relaxation techniques, and cognitive behavioral therapy—have been shown to decrease disease severity.35 Comparable improvement in disease severity has been demonstrated in children with AD when similar education is provided to them and their families.
CORRESPONDENCE
Franklin Berkey, DO, Penn State Health, 1850 East Park Avenue, Suite 207, State College, PA 16803; fberkey@ pennstatehealth.psu.edu.
Atopic dermatitis (AD), also known as eczema, is a chronic inflammatory skin condition that is well known for its relapsing, pruritic rash in children and adults. Less recognized are its associated conditions—allergic rhinitis, asthma, food allergies, attention-deficit/hyperactivity disorder (ADHD), depression, and anxiety—and its burden on patients and their families. In fact, families that have children with AD report lower overall quality of life than those with otherwise healthy children.1 Given AD’s prevalence across age groups and its effect on the family, family physicians are uniquely positioned to diagnose, care for, and counsel patients with AD and its associated maladies.
The prevalence and pathogenesis of AD
AD affects up to 20% of children and 5% of adults in the United States.2 AD typically manifests before a child reaches age 5 (often in the first 6 months of life), and it is slightly more common in females (1.3:1). A family history of atopy (eczema, asthma, allergic rhinitis) is common. In fact, children with one atopic parent have a 2- to 3-fold increased risk of atopic dermatitis; those with 2 atopic parents have a 3- to 5-fold increased risk.3
The pathophysiology of AD is complex, culminating in impaired barrier function of the skin and transepidermal water loss resulting in dry and inflamed skin. Additionally, alterations in a cell-mediated immune response leading to an immunoglobulin (Ig) E-mediated hypersensitivity is also theorized to play a role in the development of AD.
Signs and symptoms
Signs at birth. Physical signs of atopic dermatitis typically appear between birth and 6 months. In infancy, lesions generally occur on the scalp, face (FIGURES 1A and 1B), neck, and extensor surfaces of the extremities. Lesions are typically papules and vesicles, sometimes accompanied by serous exudate and crusting. Eczematous lesions typically spare the groin and diaper area, and their presence in this area should raise suspicion for an alternative diagnosis.

Beginning at age 2 years, eczematous lesions are more commonly limited to the folds of the flexor surfaces. Instead of the weeping and crusting lesions seen in infancy, eczema in older children manifests as dry, lichenified papules and plaques in areas that are typically affected in adults: the wrist, hands, ankles, and popliteal and antecubital fossa.2
Although lesions in adults are similar to those of childhood, they may manifest in a more localized area (hand or eyelid, for example). As is the case in childhood, the lesions are dry, sometimes lichenified, and found on the flexural surfaces (FIGURES 2A and 2B).2

Symptom triggers are unproven
While anecdotal reports cite various triggers for AD flares, a systematic review found little scientific evidence to substantiate identifiable triggers.4 Triggers often cited and studied are foods, dust mite exposure, airborne allergens, detergents, sunlight, fabrics, bacterial infections, and stress. While as many as one-third of people with AD who also have confirmed dust mite allergy report worsening of symptoms when exposed to dust, a Cochrane review of 7 randomized controlled trials totaling 324 adults and children with eczema found that efforts at dust mite mitigation (laundering of bed covers, increased vacuuming, spraying for mites) were not effective in reducing symptoms.5
Continue to: How quality of life diminishes with AD
How quality of life diminishes with AD
AD substantially lessens quality of life. For children, the most distressing physical symptoms include itching that inhibits sleep and provokes scratching, pain, and bleeding. Emotional distress can cause irritability, crying, and uncooperativeness with treatments. Parents also report that they frequently restrict their children from activities, such as playing in the heat or swimming, that may lead to worsening of their eczema.6
The loss of sleep associated with AD is not completely understood but is likely multifactorial. Pruritus and scratching leading to sleeplessness is the most obvious culprit, but an altered circadian rhythm, immune system response, and changes in skin physiology are also likely factors.7 Whatever the cause, sleep disturbance is reported in as many as 60% of patients with AD, and the degree of sleep disturbance is proportional to increases in disease severity and worsening of quality-of-life scores.8 Lost sleep is not limited to patients; parents of children with AD also report significant loss of sleep and subsequent decreased work productivity and quality of life.9
Children with AD are often the target of bullying.10 A 2015 survey by the National Eczema Association indicates that 1 in 5 children reported being bullied due to their AD.11
Associated conditions and comorbidities
AD increases patients’ risks for other illnesses, due either to their underlying atopy or to the effects of

Atopic march
Atopic march—the clinical succession of AD, allergic rhinitis, and asthma—is a well-established clinical progression. The presence of all 3 conditions appears to be more common in children diagnosed with AD before 2 years of age.12 Typically, allergic rhinitis manifests at around age 4, and asthma develops between ages 6 and 8. The severity of AD predicts progression. Compared with an 8% chance of asthma developing among the general population, children with mild AD have a 20% to 30% chance of developing asthma, and those with severe AD have about a 70% chance.12
Continue to: Food allergies
Food allergies
Patients with AD are at higher risk for food-induced anaphylaxis, with up to one-third of AD patients having an IgE-mediated food allergy.13 While it is theorized that the impaired skin barrier of an atopic child may allow for early sensitization and allergy development, a landmark 2015 study demonstrated that early allergen introduction (specifically, peanuts) may serve as a preventive strategy in those at high risk of food allergies.14 Current guidelines recommend that physicians be aware of the increased possibility of food allergies in those with AD, and consider evaluating a child for milk, egg, peanut, wheat, and soy allergy if the child is younger than 5 years and has eczema that does not resolve with treatment, or has eczema and a history of an allergic reaction to a specific food.15
Interestingly, despite the strong association between AD and food allergies, it is not clear that food allergies trigger atopic flares; as such, elimination diets are not universally recommended in those without a proven food allergy.
Psychiatric diagnoses
Children with AD have an increased prevalence of several psychiatric conditions, including ADHD, depression, anxiety, conduct disorder, and autism when compared with peers who do not have AD, and the probability correlates with the severity of AD
What we do know is that one of the strongest associations between AD and a psychiatric condition is with ADHD, with a recent pooled meta-analysis showing a 46% increase in risk.17 The incidence of depression among children with AD appears to correlate with the severity of AD symptoms: estimated at 5% with mild AD, 7% with moderate disease, and 14% with severe disease (compared with 3% without AD). Similar incremental increases are seen when correlating AD and anxiety.16
Nonpharmacologic care
Bathing
Bathing habits are critical to controlling AD. While bathing serves to both hydrate the skin and remove allergens, the water’s evaporation off the skin surface can lead to increased transepidermal water loss. Combining bathing and immediate application of a moisturizer improves skin hydration in patients with AD vs bathing alone.18 Thus, consensus guidelines recommend once-daily bathing (bath or shower) to remove scale and crust, followed by immediate application of a moisturizing emollient.19
Continue to: Emollients
Emollients
Application of moisturizing emollients is the mainstay of nonpharmacologic care of AD, and there is strong evidence that their regimented use reduces disease burden and the need for prescription treatment.19 Emollient creams and ointments help retain moisture and improve the skin’s barrier. While ointments may provide a better barrier, patients tend to prefer creams as they are less greasy than ointments.
Emollient therapy may also help prevent development of AD, especially in those infants thought to be at high risk with a family history of atopy. In a multinational randomized controlled trial, infants who received daily full-body application of emollient beginning at 3 weeks of life were significantly less likely than controls to develop AD by 6 months.20 While the mechanism of action is not clearly understood, it is believed that early emollient use prevents skin dehydration and maintains the skin’s barrier integrity, thus decreasing allergen epidermal penetration and subsequent inflammation.
Bleach bath
A bleach bath, prepared by adding 1/2 cup of unconcentrated bleach (5.25% sodium hypochlorite) to a standard 40-gallon bathtub, produces a chlorine mixture equivalent to an average swimming pool. Soaking in a bleach bath for 10 minutes once or twice weekly is thought to reduce inflammation and bacteria on the skin, but studies of its efficacy in improving atopic symptoms are mixed.
In a pooled analysis of 5 studies evaluating bleach baths vs standard baths, there was no significant difference in disease severity at 4 weeks.21 Thus, while bleach baths were effective in decreasing disease severity, they appeared to be no more effective than a standard water bath.21 Bleach baths may be helpful, however, in cases of moderate-to-severe disease with frequent bacterial infections.19
Pharmacologic therapy
Steroids
For symptoms refractory to nonpharmacologic skin care, topical steroids are the initial pharmacologic treatment for AD.19 Choose steroid potency based on symptom severity and disease location. Low- to medium-potency is appropriate for mild disease, and medium- to high-potency is useful for moderate-to-severe symptoms. High-potency steroids are generally avoided on the face and skin folds; however, they can be used for short periods in these areas to induce remission. They must then be quickly tapered and discontinued.
Continue to: Frequency
Frequency. Topical corticosteroids are typically applied twice daily, although recent studies indicate that once-daily application is just as efficacious.22 In addition to treatment of an acute flare, topical steroids are useful as maintenance therapy for patients with recurrent outbreaks in the same anatomical site. Guidelines suggest once- or twice-weekly application of a medium-potency steroid to prolong time between flares.19
For children, a practical guide is for caregivers to apply the amount of steroid covering 1 adult fingertip to an area of the child’s skin equal to that of 2 adult palms.23 Topical steroids are generally well tolerated and have a good safety profile. Adverse effects are proportional to the amount and duration of use and include purpura, telangiectasias, striae, and skin atrophy. The risk of skin atrophy increases with higher potency steroids, occlusion (covering affected area after steroid application), use on thin-skinned areas, and older patient age.24
Reassure patients/parents about the safety of topical steroids, as fears regarding the potential adverse effects can limit compliance. In one study of 200 patients with AD, 72.5% of respondents expressed fear of using steroids on their own skin or that of their child, and 24% admitted being noncompliant with therapy based on these concerns.25
Treating flares. Oral steroids are sometimes needed to abort or control an AD flare in older children and adults. A tapering course of prednisone over 5 to 7 days, transitioning to medium- to high-dose topical steroids, may be needed to achieve symptom control.
Topical calcineurin inhibitors
Topical calcineurin inhibitors, including tacrolimus and pimecrolimus, are generally second-line therapy to topical corticosteroids. However, as nonsteroidal agents, topical calcineurin inhibitors do not cause skin atrophy and can be a first-line option in areas where atrophy is more common (face, eyelids, neck, and skin folds).26
Continue to: A Cochrane review found...
A Cochrane review found tacrolimus 0.1% to be better than low‐potency topical corticosteroids on the face and neck areas, while results were equivocal when compared with moderate‐potency topical corticosteroids on the trunk and extremities (no difference based on physician assessment, but marginal benefit favoring tacrolimus based on participant scoring).27 When compared head-to-head, tacrolimus was more effective than pimecrolimus, although tacrolimus has a higher rate of local irritation. The most common adverse effects are stinging and burning at the application site, although these adverse effects generally improve with repeated application.
There have been long-term safety concerns with topical calcineurin inhibitors—chiefly a 2006 Food and Drug Administration (FDA) black box warning regarding a possible link between topical calcineurin inhibitors and cancer. However, while there may be a slight increased risk of lymphoma in AD patients, a recent meta-analysis did not find an association between topical calcineurin inhibitors use and lymphoma.28 Given the initial concern—and pending additional data—the FDA currently recommends reserving topical calcineurin inhibitors for second-line therapy and only for the minimum amount of time to induce improvement. It also recommends avoiding their use in patients younger than 2 years and in those with compromised immune systems.
Cisaborole
Cisaborole, a topical phosphodiesterase 4 (PDE4) inhibitor, received FDA approval in 2016 for mild-to-moderate AD. By inhibiting PDE4, the drug limits inflammation. In a multicenter randomized trial, patients applying cisaborole 2% twice a day noted reductions in pruritus, inflammation, excoriation, and lichenification.29 Adverse effects are minimal and limited to application site irritation.
Systemic treatments
While beyond the care of a family physician, symptoms refractory to conservative, nonpharmacologic measures and combinations of topical pharmaceuticals can be treated with systemic immunomodulators such as cyclosporine, azathioprine, and methotrexate. Phototherapy is also effective in patients with more widespread skin involvement. Dupilumab, an injectable monoclonal antibody that binds to interleukin-4 receptor and inhibits inflammation, is approved to treat moderate-to-severe AD in adults.30
Ineffective therapies: Oral montelukast and probiotics
While oral antihistamines are frequently prescribed and used, there are no studies evaluating the use of antihistamines (H1) as monotherapy for AD.31 Nonetheless, while not altering the disease process, the sedative effect of antihistamines may palliate the nocturnal pruritus frequently associated with AD. Although nonsedating antihistamines may still have a role for atopic patients with concurrent seasonal and environmental allergies, there is no evidence to support their use in the treatment of AD.
Continue to: Data are limited...
Data are limited on the effectiveness of leukotriene receptor antagonists for AD, and all studies meeting inclusion for a Cochrane review assessed oral montelukast. The review found no benefit with the use of montelukast 10 mg in terms of severity of disease, pruritus, or need for topical steroids.32
A systematic review investigating the benefit of probiotics for the treatment of AD found no improvement in patient-rated eczema scores for quality of life.33 Additionally, a review of 11 randomized controlled trials including 596 participants found no evidence to suggest efficacy of fish oil, zinc, selenium, vitamin D, vitamin E, pyridoxine, sea buckthorn oil, hempseed oil, or sunflower oil in the treatment of AD.34
Education can reduce AD severity
Family physicians can be a source of education and support for patients and families of patients with AD. Support programs for adults with AD—including education, relaxation techniques, and cognitive behavioral therapy—have been shown to decrease disease severity.35 Comparable improvement in disease severity has been demonstrated in children with AD when similar education is provided to them and their families.
CORRESPONDENCE
Franklin Berkey, DO, Penn State Health, 1850 East Park Avenue, Suite 207, State College, PA 16803; fberkey@ pennstatehealth.psu.edu.
1. Carroll CL, Balkrishnan R, Feldman SR, et al. The burden of atopic dermatitis: impact on the patient, family, and society. Pediatr Dermatol. 2005;22:192-199.
2. Ahn C, Huang W. Clinical presentation of atopic dermatitis. In: Fortson E, Feldman SR, Stroud LC, eds. Management of Atopic Dermatitis: Methods and Challenges. Springer International Publishing; 2017:38-46.
3. Eichenfield LF, Tom WL, Chamblin SL, et al. Guidelines of care for the management of atopic dermatitis. Part 1: diagnosis and assessment of atopic dermatitis. J Am Acad Dermatol. 2014;70:338-351.
4. Langan SM, Williams HC. What causes worsening of eczema? A systematic review. Br J Dermatol. 2006;155:504-514.
5. Nankervis H, Pynn EV, Boyle RJ, et al. House dust mite reduction and avoidance measures for treating eczema. Cochrane Database Syst Rev. 2015:CD008426.
6. Chamlin SL, Frieden IJ, Williams ML, et al. Effects of atopic dermatitis on young American children and their families. Pediatrics. 2004;114:607-611.
7. Chang Y-S, Chiang B-L. Mechanism of sleep disturbance in children with atopic dermatitis and the role of the circadian rhythm and melatonin. Int J Mol Sci. 2016;17:462.
8. Camfferman D, Kennedy JD, Gold M, et al. Eczema and sleep and its relationship to daytime functioning in children. Sleep Med Rev. 2010;14:359-369.
9. Chamlin SL, Mattson CL, Frieden IJ, et al. The price of pruritus: sleep disturbance and cosleeping in atopic dermatitis. Arch Pediatr Adolesc Med. 2005;159:745-750.
10. Drucker AM, Wang AR, Li W-Q, et al. The burden of atopic dermatitis: summary of a report for the National Eczema Association. J Invest Dermatol. 2017;137:P26-P30.
11. National Eczema Association. Tools for school: addressing school bullying for kids with eczema. Accessed January 5, 2021. https://nationaleczema.org/children-with-eczema-experience-bullying/
12. Bantz SK, Zhu Z, Zhen T. The atopic march: progression from atopic dermatitis to allergic rhinitis and asthma. J Clin Cell Immunol. 2014;5:202
13. Laird M, Sicco KL. Defining and measuring the scope of atopic dermatitis. Adv Exp Med Biol. 2017;1027:93-104.
14. Du Toit G, Roberts G, Sayre PH, et al. Randomized trial of peanut consumption in infants at risk for peanut allergy. N Engl J Med. 2015;372:803-813.
15. Boyce JA, Assa’ad A, Burks AW, et al. Guidelines for the diagnosis and management of food allergy in the United States: report of the NIAID-sponsored expert panel. J Allergy Clin Immunol. 2010;126:S1–S58.
16. Yaghmaie P, Koudelka CW, Simpson EL. Mental health comorbidity in patients with atopic dermatitis. J Allergy Clin Immunol. 2013;131:428-433.
17. Strom MA, Fishbein AB, Paller AS, et al. Association between atopic dermatitis and attention deficit hyperactivity disorder in U.S. children and adults. Br J Dermatol. 2016;175:920-929.
18. Chiang C, Eichenfield LF. Quantitative assessment of combination bathing and moisturizing regimens on skin hydration in atopic dermatitis. Pediatr Dermatol. 2009;26:273-278.
19. Eichenfield LF, Tom WL, Berger TG, et al. Guidelines of care for the management of atopic dermatitis: section 2. Management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
20. Simpson EL, Chalmers JR, Hanifin JM, et al. Emollient enhancement of the skin barrier from birth offers effective atopic dermatitis prevention. J Allergy Clin Immunol. 2014;134:818-823.
21. Chopra R, Vakharia PP, Sacotte R, et al. Efficacy of bleach baths in reducing severity of atopic dermatitis: a systematic review and meta-analysis. Ann Allergy Asthma Immunol. 2017;119:435-440.
22. Williams HC. Established corticosteroid creams should be applied only once daily in patients with atopic eczema. BMJ. 2007;334:1272.
23. Long CC, Mills CM, Finlay AY. A practical guide to topical therapy in children. Br J Dermatol. 1998;138:293-296.
24. Callen J, Chamlin S, Eichenfield LF, et al. A systematic review of the safety of topical therapies for atopic dermatitis. Br J Dermatol. 2007;156:203-221.
25. Charman CR, Morris AD, Williams HC. Topical corticosteroid phobia in patients with atopic eczema. Br J Dermatol. 2000;142:931-936.
26. Ashcroft DM, Dimmock P, Garside R, et al. Efficacy and tolerability of topical pimecrolimus and tacrolimus in the treatment of atopic dermatitis: a meta-analysis of randomised controlled trials. BMJ. 2005;330:516.
27. Cury Martins J, Martins C, Aoki V, et al. Topical tacrolimus for atopic dermatitis. Cochrane Database Syst Rev. 2015:CD009864.
28. Legendre L, Barnetche T, Mazereeuw-Hautier J, et al. Risk of lymphoma in patients with atopic dermatitis and the role of topical treatment: a systematic review and meta-analysis. J Am Acad Dermatol. 2015;72:992-1002.
29. Paller AS, Tom WL, Lebwohl MG, et al. Efficacy and safety of crisaborole ointment, a novel, nonsteroidal phosphodiesterase 4 (PDE4) inhibitor for the topical treatment of atopic dermatitis (AD) in children and adults. J Am Acad Dermatol. 2016;75:494-503.
30. Dupilumab [package insert]. Tarrytown, NY: Regeneron Pharmaceuticals Inc; 2017.
31. van Zuuren EJ, Apfelbacher CJ, Fedorowicz Z, et al. No high level evidence to support the use of oral H1 antihistamines as monotherapy for eczema: a summary of a Cochrane systematic review. Syst Rev. 2014;3:25.
32. Ferguson L, Futamura M, Vakirlis E, et al. Leukotriene receptor antagonists for eczema. Cochrane Database Syst Rev. 2018:CD011224.
33. Makrgeorgou A, Leonardi-Bee J, Bath-Hextall FJ, et al. Probiotics for treating eczema. Cochrane Database Syst Rev. 2018:CD006135.
34. Bath-Hextall FJ, Jenkinson C, Humphreys R, et al. Dietary supplements for established atopic eczema. Cochrane Database Syst Rev. 2012:CD005205.
35. Sy W, Lamb AJ. Atopic dermatitis disease education. In: Fortson E, Feldman SR, Stroud LC, eds. Management of Atopic Dermatitis: Methods and Challenges. Springer International Publishing; 2017:179-184.
1. Carroll CL, Balkrishnan R, Feldman SR, et al. The burden of atopic dermatitis: impact on the patient, family, and society. Pediatr Dermatol. 2005;22:192-199.
2. Ahn C, Huang W. Clinical presentation of atopic dermatitis. In: Fortson E, Feldman SR, Stroud LC, eds. Management of Atopic Dermatitis: Methods and Challenges. Springer International Publishing; 2017:38-46.
3. Eichenfield LF, Tom WL, Chamblin SL, et al. Guidelines of care for the management of atopic dermatitis. Part 1: diagnosis and assessment of atopic dermatitis. J Am Acad Dermatol. 2014;70:338-351.
4. Langan SM, Williams HC. What causes worsening of eczema? A systematic review. Br J Dermatol. 2006;155:504-514.
5. Nankervis H, Pynn EV, Boyle RJ, et al. House dust mite reduction and avoidance measures for treating eczema. Cochrane Database Syst Rev. 2015:CD008426.
6. Chamlin SL, Frieden IJ, Williams ML, et al. Effects of atopic dermatitis on young American children and their families. Pediatrics. 2004;114:607-611.
7. Chang Y-S, Chiang B-L. Mechanism of sleep disturbance in children with atopic dermatitis and the role of the circadian rhythm and melatonin. Int J Mol Sci. 2016;17:462.
8. Camfferman D, Kennedy JD, Gold M, et al. Eczema and sleep and its relationship to daytime functioning in children. Sleep Med Rev. 2010;14:359-369.
9. Chamlin SL, Mattson CL, Frieden IJ, et al. The price of pruritus: sleep disturbance and cosleeping in atopic dermatitis. Arch Pediatr Adolesc Med. 2005;159:745-750.
10. Drucker AM, Wang AR, Li W-Q, et al. The burden of atopic dermatitis: summary of a report for the National Eczema Association. J Invest Dermatol. 2017;137:P26-P30.
11. National Eczema Association. Tools for school: addressing school bullying for kids with eczema. Accessed January 5, 2021. https://nationaleczema.org/children-with-eczema-experience-bullying/
12. Bantz SK, Zhu Z, Zhen T. The atopic march: progression from atopic dermatitis to allergic rhinitis and asthma. J Clin Cell Immunol. 2014;5:202
13. Laird M, Sicco KL. Defining and measuring the scope of atopic dermatitis. Adv Exp Med Biol. 2017;1027:93-104.
14. Du Toit G, Roberts G, Sayre PH, et al. Randomized trial of peanut consumption in infants at risk for peanut allergy. N Engl J Med. 2015;372:803-813.
15. Boyce JA, Assa’ad A, Burks AW, et al. Guidelines for the diagnosis and management of food allergy in the United States: report of the NIAID-sponsored expert panel. J Allergy Clin Immunol. 2010;126:S1–S58.
16. Yaghmaie P, Koudelka CW, Simpson EL. Mental health comorbidity in patients with atopic dermatitis. J Allergy Clin Immunol. 2013;131:428-433.
17. Strom MA, Fishbein AB, Paller AS, et al. Association between atopic dermatitis and attention deficit hyperactivity disorder in U.S. children and adults. Br J Dermatol. 2016;175:920-929.
18. Chiang C, Eichenfield LF. Quantitative assessment of combination bathing and moisturizing regimens on skin hydration in atopic dermatitis. Pediatr Dermatol. 2009;26:273-278.
19. Eichenfield LF, Tom WL, Berger TG, et al. Guidelines of care for the management of atopic dermatitis: section 2. Management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
20. Simpson EL, Chalmers JR, Hanifin JM, et al. Emollient enhancement of the skin barrier from birth offers effective atopic dermatitis prevention. J Allergy Clin Immunol. 2014;134:818-823.
21. Chopra R, Vakharia PP, Sacotte R, et al. Efficacy of bleach baths in reducing severity of atopic dermatitis: a systematic review and meta-analysis. Ann Allergy Asthma Immunol. 2017;119:435-440.
22. Williams HC. Established corticosteroid creams should be applied only once daily in patients with atopic eczema. BMJ. 2007;334:1272.
23. Long CC, Mills CM, Finlay AY. A practical guide to topical therapy in children. Br J Dermatol. 1998;138:293-296.
24. Callen J, Chamlin S, Eichenfield LF, et al. A systematic review of the safety of topical therapies for atopic dermatitis. Br J Dermatol. 2007;156:203-221.
25. Charman CR, Morris AD, Williams HC. Topical corticosteroid phobia in patients with atopic eczema. Br J Dermatol. 2000;142:931-936.
26. Ashcroft DM, Dimmock P, Garside R, et al. Efficacy and tolerability of topical pimecrolimus and tacrolimus in the treatment of atopic dermatitis: a meta-analysis of randomised controlled trials. BMJ. 2005;330:516.
27. Cury Martins J, Martins C, Aoki V, et al. Topical tacrolimus for atopic dermatitis. Cochrane Database Syst Rev. 2015:CD009864.
28. Legendre L, Barnetche T, Mazereeuw-Hautier J, et al. Risk of lymphoma in patients with atopic dermatitis and the role of topical treatment: a systematic review and meta-analysis. J Am Acad Dermatol. 2015;72:992-1002.
29. Paller AS, Tom WL, Lebwohl MG, et al. Efficacy and safety of crisaborole ointment, a novel, nonsteroidal phosphodiesterase 4 (PDE4) inhibitor for the topical treatment of atopic dermatitis (AD) in children and adults. J Am Acad Dermatol. 2016;75:494-503.
30. Dupilumab [package insert]. Tarrytown, NY: Regeneron Pharmaceuticals Inc; 2017.
31. van Zuuren EJ, Apfelbacher CJ, Fedorowicz Z, et al. No high level evidence to support the use of oral H1 antihistamines as monotherapy for eczema: a summary of a Cochrane systematic review. Syst Rev. 2014;3:25.
32. Ferguson L, Futamura M, Vakirlis E, et al. Leukotriene receptor antagonists for eczema. Cochrane Database Syst Rev. 2018:CD011224.
33. Makrgeorgou A, Leonardi-Bee J, Bath-Hextall FJ, et al. Probiotics for treating eczema. Cochrane Database Syst Rev. 2018:CD006135.
34. Bath-Hextall FJ, Jenkinson C, Humphreys R, et al. Dietary supplements for established atopic eczema. Cochrane Database Syst Rev. 2012:CD005205.
35. Sy W, Lamb AJ. Atopic dermatitis disease education. In: Fortson E, Feldman SR, Stroud LC, eds. Management of Atopic Dermatitis: Methods and Challenges. Springer International Publishing; 2017:179-184.
PRACTICE RECOMMENDATIONS
› Advise patients to regularly apply moisturizers, which reduces atopic dermatitis (AD) severity and may avert the need for pharmacologic intervention. A
› Assure patients that a topical corticosteroid is safe and effective as first-line treatment for AD symptoms refractory to nonpharmacologic recommendations. A
› Consider topical calcineurin inhibitors for both acute and chronic AD in adults and children, especially in areas more prone to topical corticosteroid adverse effects. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Tactics to prevent or slow progression of CKD in patients with diabetes
Chronic kidney disease (CKD) is a significant comorbidity of diabetes mellitus. The Kidney Disease Outcomes Quality Initiative (KDOQI) of the National Kidney Foundation defines CKD as the presence of kidney damage or decreased kidney function for ≥ 3 months. CKD caused by diabetes is called diabetic kidney disease (DKD), which is 1 of 3 principal microvascular complications of diabetes. DKD can progress to end-stage renal disease (ESRD), requiring kidney replacement therapy, and is the leading cause of CKD and ESRD in the United States.1-3 Studies have also shown that, particularly in patients with diabetes, CKD considerably increases the risk of cardiovascular events, which often occur prior to ESRD.1,4
This article provides the latest recommendations for evaluating and managing DKD to help you prevent or slow its progression.
Defining and categorizing diabetic kidney disease
CKD is defined as persistently elevated excretion of urinary albumin (albuminuria) and decreased estimated glomerular filtration rate (eGFR), or as the presence of signs of progressive kidney damage.5,6 DKD, also known as diabetic nephropathy, is CKD attributed to long-term diabetes. A patient’s eGFR is the established basis for assignment to a stage (1, 2, 3a, 3b, 4, or 5) of CKD (TABLE 17) and, along with the category of albuminuria (A1, A2, or A3), can indicate prognosis.
Taking its toll in diabetes
As many as 40% of patients with diabetes develop DKD.8-10 Most studies of DKD have been conducted in patients with type 1 diabetes (T1D), because the time of clinical onset is typically known.
Type 1 diabetes. DKD usually occurs 10 to 15 years, or later, after the onset of diabetes.6 As many as 30% of people with T1D have albuminuria approximately 15 years after onset of diabetes; almost one-half of those develop DKD.5,11 After approximately 22.5 years without albuminuria, patients with T1D have approximately a 1% annual risk of DKD.12
Type 2 diabetes (T2D). DKD is often present at diagnosis, likely due to a delay in diagnosis and briefer clinical exposure, compared to T1D. Albuminuria has been reported in as many as 40% of patients with T2D approximately 10 years after onset of diabetes.12,13
Multiple risk factors with no standout “predictor”
Genetic susceptibility, ethnicity, glycemic control, smoking, blood pressure (BP), and the eGFR have been identified as risk factors for renal involvement in diabetes; obesity, oral contraceptives, and age can also contribute. Although each risk factor increases the risk of DKD, no single factor is adequately predictive. Moderately increased albuminuria, the earliest sign of DKD, is associated with progressive nephropathy.12
Continue to: How great is the risk?
How great is the risk? From disease onset to proteinuria and from proteinuria to ESRD, the risk of DKD in T1D and T2D is similar. With appropriate treatment, albuminuria can regress, and the risk of ESRD can be < 20% at 10 years in T1D.12 As in T1D, good glycemic control might result in regression of albuminuria in T2D.14
For unknown reasons, the degree of albuminuria can exist independent of the progression of DKD. Factors responsible for a progressive decline in eGFR in DKD without albuminuria are unknown.12,15
Patient evaluation with an eye toward comorbidities
A comprehensive initial medical evaluation for DKD includes a review of microvascular complications; visits to specialists; lifestyle and behavior patterns (eg, diet, sleep, substance use, and social support); and medication adherence, adverse drug effects, and alternative medicines. Although DKD is often a clinical diagnosis, it can be ruled in by persistent albuminuria or decreased eGFR, or both, in established diabetes or diabetic retinopathy when other causes are unlikely (see “Recommended DKD screening protocol,” below).
Screening for mental health conditions and barriers to self-management is also key.6
Comorbidities, of course, can complicate disease management in patients with diabetes.16-20 Providers and patients therefore need to be aware of potential diabetic comorbidities. For example, DKD and even moderately increased albuminuria significantly increase the risk of cardiovascular disease (CVD).12 Other possible comorbidities include (but are not limited to) nonalcoholic steatohepatitis, fracture, hearing impairment, cancer (eg, liver, pancreas, endometrium, colon, rectum, breast, and bladder), pancreatitis, hypogonadism, obstructive sleep apnea, periodontal disease, anxiety, depression, and eating disorders.6
Continue to: Recommended DKD screening protocol
Recommended DKD screening protocol
In all cases of T2D, in cases of T1D of ≥ 5 years’ duration, and in patients with diabetes and comorbid hypertension, perform annual screening for albuminuria, an elevated creatinine level, and a decline in eGFR.
To confirm the diagnosis of DKD, at least 2 of 3 urine specimens must demonstrate an elevated urinary albumin:creatinine ratio (UACR) over a 3- to 6-month period.21 Apart from renal damage, exercise within 24 hours before specimen collection, infection, fever, congestive heart failure, hyperglycemia, menstruation, and hypertension can elevate the UACR.6
Levels of the UACR are established as follows22:
- Normal UACR is defined as < 30 milligrams of albumin per gram of creatinine (expressed as “mg/g”).
- Increased urinary albumin excretion is defined as ≥ 30 mg/g.
- Moderately increased albuminuria, a predictor of potential nephropathy, is the excretion of 30 to 300 mg/g.
- Severely increased albuminuria is excretion > 300 mg/g; it is often followed by a gradual decline in eGFR that, without treatment, eventually leads to ESRD.
The rate of decline in eGFR once albuminuria is severely increased is equivalent in T1D and T2D.12 Without intervention, the time from severely increased albuminuria to ESRD in T1D and T2D averages approximately 6 or 7 years.
Clinical features
DKD is typically a clinical diagnosis seen in patients with longstanding diabetes, albuminuria, retinopathy, or a reduced eGFR in the absence of another primary cause of kidney damage. In patients with T1D and DKD, signs of retinopathy and neuropathy are almost always present at diagnosis, unless a diagnosis is made early in the course of diabetes.12 Therefore, the presence of retinopathy suggests that diabetes is the likely cause of CKD.
Continue to: The presence of microvascular disease...
The presence of microvascular disease in patients with T2D and DKD is less predictable.12 In T2D patients who do not have retinopathy, consider causes of CKD other than DKD. Features suggesting that the cause of CKD is an underlying condition other than diabetes are rapidly increasing albuminuria or decreasing eGFR; urinary sediment comprising red blood cells or white blood cells; and nephrotic syndrome.6
As the prevalence of diabetes increases, it has become more common to diagnose DKD by eGFR without albuminuria—underscoring the importance of routine monitoring of eGFR in patients with diabetes.6
Sources of expert guidance. The Chronic Kidney Disease Epidemiology Collaboration equation23 is preferred for calculating eGFR from serum creatinine: An eGFR < 60 mL/min/1.73 m2 is considered abnormal.3,12 At these rates, the prevalence of complications related to CKD rises and screening for complications becomes necessary.
A more comprehensive classification of the stages of CKD, incorporating albuminuria and progression of CKD, has been recommended by Kidney Disease: Improving Global Outcomes (KDIGO).7 Because eGFR and excretion of albumin vary, abnormal test results need to be verified over time to stage the degree of CKD.3,12 Kidney damage often manifests as albuminuria, but also as hematuria, other types of abnormal urinary sediment, radiographic abnormalities, and other abnormal presentations.
Management
Nutritional factors
Excessive protein intake has been shown to increase albuminuria, worsen renal function, and increase CVD mortality in DKD.24-26 Therefore, daily dietary protein intake of 0.8 g/kg body weight is recommended for patients who are not on dialysis.3 Patients on dialysis might require higher protein intake to preserve muscle mass caused by protein-energy wasting, which is common in dialysis patients.6
Continue to: Low sodium intake
Low sodium intake in CKD patients has been shown to decrease BP and thus slow the progression of renal disease and lower the risk of CVD. The recommended dietary sodium intake in CKD patients is 1500-3000 mg/d.3
Low potassium intake. Hyperkalemia is a serious complication of CKD. A low-potassium diet is recommended in ESRD patients who have a potassium level > 5.5 mEq/L.6
Blood pressure
Preventing and treating hypertension is critical to slowing the progression of CKD and reducing cardiovascular risk. BP should be measured at every clinic visit. Aside from lifestyle changes, medication might be needed to reach target BP.
The American Diabetes Association recommends a BP goal of ≤ 140/90 mm Hg for hypertensive patients with diabetes, although they do state that a lower BP target (≤ 130/80 mm Hg) might be more appropriate for patients with DKD.27
The American College of Cardiology recommends that hypertensive patients with CKD have a BP target of ≤ 130/80 mm Hg.28
Continue to: ACE inhibitors and ARBs
Angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARBs) have renoprotective benefits. These agents are recommended as first-line medications for patients with diabetes, hypertension, and an eGFR < 60 mL/min/1.73 m2 and a UACR > 300 mg/g.29-31 Evidence also supports their use when the UACR is 30 to 299 mg/g.
Studies have shown that, in patients with DKD, ACE inhibitors and ARBs can slow the progression of renal disease.29,30,32 There is no difference between ACE inhibitors and ARBs in their effectiveness for preventing progression of DKD.6 There is no added benefit in combining an ACE inhibitor and an ARB33; notably, combination ACE inhibitor and ARB therapy can increase the risk of adverse events, such as hyperkalemia and acute kidney injury, especially in patients with DKD.33
There is no evidence for starting an ACE inhibitor or ARB to prevent CKD in patients with diabetes who are not hypertensive.5
ACE inhibitors and ARBs should be used with caution in women of childbearing age, who should use a reliable form of contraception if taking one of these drugs.
Diuretics. Thiazide-type and loop diuretics might potentiate the positive effects of ACE inhibitors and ARBs. KDOQI guidelines recommend that, in patients who require a second agent to control BP, a diuretic should be considered in combination with an ACE inhibitor or an ARB.20 A loop diuretic is preferred if the eGFR is < 30 mL/min/1.73 m2.
Continue to: Nondihydropyridine calcium-channel blockers
Nondihydropyridine calcium-channel blockers (CCBs), such as diltiazem and verapamil, have been shown to be more effective then dihydrophyridine CCBs, such as amlodipine and nifedipine, in slowing the progression of renal disease because of their antiproteinuric effects. However, the antiproteinuric effects of nondihydropyridine CCBs are not as strong as those of ACE inhibitors or ARBs, and these drugs do not appear to potentiate the effects of an ACE inhibitor or ARB when used in combination.20
Nondihydropyridine CCBs might be a reasonable alternative in patients who cannot tolerate an ACE inhibitor or an ARB.
Mineralocorticoid receptor antagonists in combination with an ACE inhibitor or ARB have been demonstrated to reduce albuminuria in short-term studies.34,35
Glycemic levels
Studies conducted in patients with T1D, and others in patients with T2D, have shown that tight glycemic control can delay the onset and slow the progression of albuminuria and a decline in the eGFR.10,36-39 The target glycated hemoglobin (A1C) should be < 7% to prevent or slow progression of DKD.40 However, patients with DKD have an increased risk of hypoglycemic events and increased mortality with more intensive glycemic control.40,41 Given those findings, some patients with DKD and significant comorbidities, ESRD, or limited life expectancy might need to have an A1C target set at 8%.6,42
Adjustments to antidiabetes medications in DKD
In patients with stages 3 to 5 DKD, several common antidiabetic medications might need to be adjusted or discontinued because they decrease creatinine clearance.
Continue to: First-generation sulfonylureas
First-generation sulfonylureas should be avoided in DKD. Glipizide and gliclazide are preferred among second-generation sulfonylureas because they do not increase the risk of hypoglycemia in DKD patients, although patients taking these medications still require close monitoring of their blood glucose level.20
Metformin. In 2016, recommendations changed for the use of metformin in patients with DKD: The eGFR, not the serum creatinine level, should guide treatment.43 Metformin can be used safely in patients with (1) an eGFR of < 60 mL/min/1.73 m2 and (2) an eGFR of 30 mL/min/1.73 m2 with close monitoring. Metformin should not be initiated if the eGFR is < 45 mL/min/1.73 m2.43
Antidiabetes medications with direct effect on the kidney
Several antidiabetes medications have a direct effect on the kidney apart from their effect on the blood glucose level.
Sodium-glucose co-transporter 2 (SGLT2) inhibitors have been shown to reduce albuminuria and slow the decrease of eGFR independent of glycemic control. In addition, SGLT2 inhibitors have also been shown to have cardiovascular benefits in patients with DKD.44,45
Glucagon-like peptide 1 (GLP-1) receptor agonists have been shown to delay and decrease the progression of DKD.46-48 Also, similar to what is seen with SGLT2 inhibitors, GLP-1 agonists have demonstrable cardiovascular benefit in patients with DKD.46,48
Continue to: Dyslipidemia and DKD
Dyslipidemia and DKD
Because the risk of CVD is increased in patients with DKD, addressing other modifiable risk factors, including dyslipidemia, is recommended in these patients. Patients with diabetes and stages 1 to 4 DKD should be treated with a high-intensity statin or a combination of a statin and ezetimibe.49,50
If a patient is taking a statin and starting dialysis, it’s important to discuss with him or her whether to continue the statin, based on perceived benefits and risks. It is not recommended that statins be initiated in patients on dialysis unless there is a specific cardiovascular indication for doing so. Risk reduction with a statin has been shown to be significantly less in dialysis patients than in patients who are not being treated with dialysis.49
Complications of CKD
Anemia is a common complication of CKD. KDIGO recommends measuring the hemoglobin concentration annually in DKD stage 3 patients without anemia; at least every 6 months in stage 4 patients; and at least every 3 months in stage 5. DKD patients with anemia should have additional laboratory testing: the absolute reticulocyte count, serum ferritin, serum transferrin saturation, vitamin B12, and folate.51
Mineral and bone disorder should be screened for in patients with DKD. TABLE 252 outlines when clinical laboratory tests should be ordered to assess for mineral bone disease.
When to refer to a nephrologist
Refer patients with stage 4 or 5 CKD (eGFR, ≤ 30 mL/min/1.73 m2) to a nephrologist for discussion of kidney replacement therapy.6 Patients with stage 3a CKD and severely increased albuminuria or with stage 3b CKD and moderately or severely increased albuminuria should also be referred to a nephrologist for intervention to delay disease progression.
Continue to: Identifying the need for early referral...
Identifying the need for early referral to a nephrologist has been shown to reduce the cost, and improve the quality, of care.53 Other indications for earlier referral include uncertainty about the etiology of renal disease, persistent or severe albuminuria, persistent hematuria, a rapid decline in eGFR, and acute kidney injury. Additionally, referral at an earlier stage of DKD might be needed to assist with complications associated with DKD, such as anemia, secondary hyperparathyroidism, mineral and bone disorder, resistant hypertension, fluid overload, and electrolyte disturbances.6
ACKNOWLEDGEMENT
The authors thank Colleen Colbert, PhD, and Iqbal Ahmad, PhD, for their review and critique of the manuscript of this article. They also thank Christopher Babiuch, MD, for his guidance in the preparation of the manuscript.
CORRESPONDENCE
Faraz Ahmad, MD, MPH, Care Point East Family Medicine, 543 Taylor Avenue, 2nd floor, Columbus, OH 43203; faraz. [email protected].
1. Radbill B, Murphy B, LeRoith D. Rationale and strategies for early detection and management of diabetic kidney disease. Mayo Clin Proc. 2008;83:1373-1381.
2. Saran R, Robinson B, Abbott KC, et al. US Renal Data System 2017 Annual Data Report: Epidemiology of kidney disease in the United States. Am J Kidney Dis. 2018;71(3 suppl 1):A7.
3. Tuttle KR, Bakris GL, Bilous RW, et al. Diabetic kidney disease: a report from an ADA Consensus Conference. Am J Kidney Dis. 2014;64:510-533.
4. Fox CS, Matsushita K, Woodward M, et al; . Associations of kidney disease measures with mortality and end-stage renal disease in individuals with and without diabetes: a meta-analysis. Lancet. 2012;380:1662-1673.
5. Orchard TJ, Dorman JS, Maser RE, et al. Prevalence of complications in IDDM by sex and duration. Pittsburgh Epidemiology of Diabetes Complications Study II. Diabetes. 1990;39:1116-1124.
6. American Diabetes Association. Standards of Medical Care in Diabetes—2018. Diabetes Care. 2018;41(suppl 1):S1-S159. Accessed January 5, 2021. https://care.diabetesjournals.org/content/41/Supplement_1
7. National Kidney Foundation. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int Suppl. 2013;3:1-150. Accessed January 5, 2021. https://kdigo.org/wp-content/uploads/2017/02/KDIGO_2012_CKD_GL.pdf
8. Afkarian M, Zelnick LR, Hall YN, et al. Clinical manifestations of kidney disease among US adults with diabetes, 1988-2014. JAMA. 2016;316:602-610.
9. de Boer IH, Rue TC, Hall YN, et al. Temporal trends in the prevalence of diabetic kidney disease in the United States. JAMA. 2011;305:2532-2539.
10. de Boer IH; DCCT/EDIC Research Group. Kidney disease and related findings in the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study. Diabetes Care. 2014;37:24-30.
11. Stanton RC. Clinical challenges in diagnosis and management of diabetic kidney disease. Am J Kidney Dis. 2014;63(2 suppl 2):S3-S21.
12. Mottl AK, Tuttle KR. Diabetic kidney disease: Pathogenesis and epidemiology. UpToDate. Updated August 19, 2019. Accessed January 5, 2021. www.uptodate.com/contents/diabetic-kidney-disease-pathogenesis-and-epidemiology
13. Bakris GL. Moderately increased albuminuria (microalbuminuria) in type 2 diabetes mellitus. UpToDate. Updated November 3, 2020. Accessed January 5, 2021. https://www.uptodate.com/contents/moderately-increased-albuminuria-microalbuminuria-in-type-2-diabetes-mellitus
14. Bandak G, Sang Y, Gasparini A, et al. Hyperkalemia after initiating renin-angiotensin system blockade: the Stockholm Creatinine Measurements (SCREAM) Project. J Am Heart Assoc. 2017;6:e005428.
15. Saran R, Robinson B, Abbott KC, et al. US Renal Data System 2016 Annual Data Report: Epidemiology of kidney disease in the United States. Am J Kidney Dis. 2017;69(3 suppl 1):A7-A8.
16. Nilsson E, Gasparini A, Ärnlöv J, et al. Incidence and determinants of hyperkalemia and hypokalemia in a large healthcare system. Int J Cardiol. 2017;245:277-284.
17. de Boer IH, Gao X, Cleary PA, et al; Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) Research Group. Albuminuria changes and cardiovascular and renal outcomes in type 1 diabetes: The DCCT/EDIC study. Clin J Am Soc Nephrol. 2016;11:1969-1977.
18. Sumida K, Molnar MZ, Potukuchi PK, et al. Changes in albuminuria and subsequent risk of incident kidney disease. Clin J Am Soc Nephrol. 2017;12:1941-1949.
19. Borch-Johnsen K, Wenzel H, Viberti GC, et al. Is screening and intervention for microalbuminuria worthwhile in patient with insulin dependent diabetes? BMJ. 1993;306:1722-1725.
20. KDOQI. KDOQI clinical practice guidelines and clinical practice recommendations for diabetes and chronic kidney disease. Am J Kidney Dis. 2007;49(2 suppl 2):S12-154.
21. Bakris GL. Moderately increased albuminuria (microalbuminuria) in type 1 diabetes mellitus. UpToDate. Updated December 3, 2019. Accessed January 5, 2021. https://www.uptodate.com/contents/moderately-increased-albuminuria-microalbuminuria-in-type-1-diabetes-mellitus
22. Delanaye P, Glassock RJ, Pottel H, et al. An age-calibrated definition of chronic kidney disease: rationale and benefits. Clin Biochem Rev. 2016;37:17-26.
23. Levey AS, Stevens LA, Schmid CH, et al; , A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604-612.
24. Wrone EM, Carnethon MR, Palaniappan L, et al; . Association of dietary protein intake and microalbuminuria in healthy adults: Third National Health and Nutrition Examination Survey. Am J Kidney Dis. 2003;41:580-587.
25. Knight EL, Stampfer MJ, Hankinson SE, et al. The impact of protein intake on renal function decline in women with normal renal function or mild renal insufficiency. Ann Intern Med. 2003;138:460-467.
26. Bernstein AM, Sun Q, Hu FB, et al. Major dietary protein sources and risk of coronary heart disease in women. Circulation. 2010;122:876-883.
27. de Boer, IH, Bangalore S, Benetos A, et al. Diabetes and hypertension: a position statement by the American Diabetes Association. Diabetes Care. 2017;40:1273-1284.
28. Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018;71:e127-e248.
29. Brenner BM, Cooper ME, de Zeeuw D, et al; Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med. 2001;345:861-869.
30. Lewis EJ, Hunsicker LG, Bain RP, et al. The effect of angiotensin-converting-enzyme inhibition on diabetic nephropathy. The Collaborative Study Group. N Engl J Med. 1993;329:1456-1462.
31. Heart Outcomes Prevention Evaluation (HOPE) Study Investigators. Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: results of the HOPE study and MICRO-HOPE substudy. Lancet. 2000;355;253-259.
32. Lewis EJ, Hunsicker LG, Clarke WR, et al; . Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med. 2001;345:851-860.
33. Fried LF, Emanuele N, Zhang JH, et al; . Combined angiotensin inhibition for the treatment of diabetic nephropathy. N Engl J Med. 2013;369:1892-1903.
34. Bakris GL, Agarwal R, Chan JC, et al; . Effect of finerenone on albuminuria in patients with diabetic nephropathy: a randomized clinical trial. JAMA. 2015;314:884-894.
35. Filippatos G, Anker SD, M, et al. Randomized controlled study of finerenone vs. eplerenone in patients with worsening chronic heart failure and diabetes mellitus and/or chronic kidney disease. Eur Heart J. 2016;37:2105-2114.
36. The ADVANCE Collaborative Group. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes.N Engl J Med. 2008;358:2560-2572.
37. Ismail-Beigi F, Craven T, Banerji MA, et al; . Effect of intensive treatment of hyperglycaemia on microvascular outcomes in type 2 diabetes: an analysis of the ACCORD randomised trial. Lancet. 2010;376:419-430.
38. Zoungas S, Chalmers J, Neal B, et al; . Follow-up of blood-pressure lowering and glucose control in type 2 diabetes. N Engl J Med. 2014;371:1392-1406.
39. Zoungas S, Arima H, Gerstein HC, et al; . Effects of intensive glucose control on microvascular outcomes in patients with type 2 diabetes: a meta-analysis of individual participant data from randomised controlled trials. Lancet Diabetes Endocrinol. 2017;5:431-437.
40. Miller ME, Bonds DE, Gerstein HC, et al; . The effects of baseline characteristics, glycaemia treatment approach, and glycated haemoglobin concentration on the risk of severe hypoglycaemia: post hoc epidemiological analysis of the ACCORD study. BMJ. 2010;340;b5444.
41. Papademetriou V, Lovato L, Doumas M, et al; . Chronic kidney disease and intensive glycemic control increase cardiovascular risk in patients with type 2 diabetes. Kidney Int. 2015;87:649-659.
42. National Kidney Foundation. KDOQI clinical practice guideline for diabetes and CKD: 2012 Update. Am J Kidney Dis. 2012;60:850-886.
43. Imam TH. Changes in metformin use in chronic kidney disease. Clin Kidney J. 2017;10:301-304.
44. Wanner C, Inzucchi SE, Lachin JM, et al; Empagliflozin and progression of kidney disease in type 2 diabetes. N Engl J Med. 2016;375:323-334.
45. Neal B, Perkovic V, Mahaffey KW, et al; . Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med. 2017;377:644-657.
46. Marso SP, Daniels GH, Brown-Frandsen K, et al; . Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375:311-322.
47. Mann JFE, DD, Brown-Frandsen K, et al; . Liraglutide and renal outcomes in type 2 diabetes. N Engl J Med. 2017;377:839-848.
48. Marso SP, Bain SC, Consoli A, et al; . Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375:1834-1844.
49. Wanner C, Tonelli M; Kidney Disease: Improving Global Outcomes Lipid Guideline Development Work Group Members. KDIGO clinical practice guideline for lipid management in CKD: summary of recommendation statements and clinical approach to the patient. Kidney Int. 2014;85:1303-1309.
50. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol. A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139:e1082-e1143.
51. National Kidney Foundation KDOQI. KDIGO clinical practice guideline for anemia in chronic kidney disease. Kidney Int Suppl. 2012;2:279-335. Accessed January 5, 2021. www.sciencedirect.com/journal/kidney-international-supplements/vol/2/issue/4
52. National Kidney Foundation KDOQI. Evaluation and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). 2010. Accessed January 5, 2021. www.kidney.org/sites/default/files/02-10-390B_LBA_KDOQI_BoneGuide.pdf
53. Smart MA, Dieberg G, Ladhani M, et al. Early referral to specialist nephrology services for preventing the progression to end-stage kidney disease. Cochrane Database Syst Rev. 2014;(6):CD007333.
Chronic kidney disease (CKD) is a significant comorbidity of diabetes mellitus. The Kidney Disease Outcomes Quality Initiative (KDOQI) of the National Kidney Foundation defines CKD as the presence of kidney damage or decreased kidney function for ≥ 3 months. CKD caused by diabetes is called diabetic kidney disease (DKD), which is 1 of 3 principal microvascular complications of diabetes. DKD can progress to end-stage renal disease (ESRD), requiring kidney replacement therapy, and is the leading cause of CKD and ESRD in the United States.1-3 Studies have also shown that, particularly in patients with diabetes, CKD considerably increases the risk of cardiovascular events, which often occur prior to ESRD.1,4
This article provides the latest recommendations for evaluating and managing DKD to help you prevent or slow its progression.
Defining and categorizing diabetic kidney disease
CKD is defined as persistently elevated excretion of urinary albumin (albuminuria) and decreased estimated glomerular filtration rate (eGFR), or as the presence of signs of progressive kidney damage.5,6 DKD, also known as diabetic nephropathy, is CKD attributed to long-term diabetes. A patient’s eGFR is the established basis for assignment to a stage (1, 2, 3a, 3b, 4, or 5) of CKD (TABLE 17) and, along with the category of albuminuria (A1, A2, or A3), can indicate prognosis.

Taking its toll in diabetes
As many as 40% of patients with diabetes develop DKD.8-10 Most studies of DKD have been conducted in patients with type 1 diabetes (T1D), because the time of clinical onset is typically known.
Type 1 diabetes. DKD usually occurs 10 to 15 years, or later, after the onset of diabetes.6 As many as 30% of people with T1D have albuminuria approximately 15 years after onset of diabetes; almost one-half of those develop DKD.5,11 After approximately 22.5 years without albuminuria, patients with T1D have approximately a 1% annual risk of DKD.12
Type 2 diabetes (T2D). DKD is often present at diagnosis, likely due to a delay in diagnosis and briefer clinical exposure, compared to T1D. Albuminuria has been reported in as many as 40% of patients with T2D approximately 10 years after onset of diabetes.12,13
Multiple risk factors with no standout “predictor”
Genetic susceptibility, ethnicity, glycemic control, smoking, blood pressure (BP), and the eGFR have been identified as risk factors for renal involvement in diabetes; obesity, oral contraceptives, and age can also contribute. Although each risk factor increases the risk of DKD, no single factor is adequately predictive. Moderately increased albuminuria, the earliest sign of DKD, is associated with progressive nephropathy.12
Continue to: How great is the risk?
How great is the risk? From disease onset to proteinuria and from proteinuria to ESRD, the risk of DKD in T1D and T2D is similar. With appropriate treatment, albuminuria can regress, and the risk of ESRD can be < 20% at 10 years in T1D.12 As in T1D, good glycemic control might result in regression of albuminuria in T2D.14
For unknown reasons, the degree of albuminuria can exist independent of the progression of DKD. Factors responsible for a progressive decline in eGFR in DKD without albuminuria are unknown.12,15
Patient evaluation with an eye toward comorbidities
A comprehensive initial medical evaluation for DKD includes a review of microvascular complications; visits to specialists; lifestyle and behavior patterns (eg, diet, sleep, substance use, and social support); and medication adherence, adverse drug effects, and alternative medicines. Although DKD is often a clinical diagnosis, it can be ruled in by persistent albuminuria or decreased eGFR, or both, in established diabetes or diabetic retinopathy when other causes are unlikely (see “Recommended DKD screening protocol,” below).
Screening for mental health conditions and barriers to self-management is also key.6
Comorbidities, of course, can complicate disease management in patients with diabetes.16-20 Providers and patients therefore need to be aware of potential diabetic comorbidities. For example, DKD and even moderately increased albuminuria significantly increase the risk of cardiovascular disease (CVD).12 Other possible comorbidities include (but are not limited to) nonalcoholic steatohepatitis, fracture, hearing impairment, cancer (eg, liver, pancreas, endometrium, colon, rectum, breast, and bladder), pancreatitis, hypogonadism, obstructive sleep apnea, periodontal disease, anxiety, depression, and eating disorders.6
Continue to: Recommended DKD screening protocol
Recommended DKD screening protocol
In all cases of T2D, in cases of T1D of ≥ 5 years’ duration, and in patients with diabetes and comorbid hypertension, perform annual screening for albuminuria, an elevated creatinine level, and a decline in eGFR.
To confirm the diagnosis of DKD, at least 2 of 3 urine specimens must demonstrate an elevated urinary albumin:creatinine ratio (UACR) over a 3- to 6-month period.21 Apart from renal damage, exercise within 24 hours before specimen collection, infection, fever, congestive heart failure, hyperglycemia, menstruation, and hypertension can elevate the UACR.6
Levels of the UACR are established as follows22:
- Normal UACR is defined as < 30 milligrams of albumin per gram of creatinine (expressed as “mg/g”).
- Increased urinary albumin excretion is defined as ≥ 30 mg/g.
- Moderately increased albuminuria, a predictor of potential nephropathy, is the excretion of 30 to 300 mg/g.
- Severely increased albuminuria is excretion > 300 mg/g; it is often followed by a gradual decline in eGFR that, without treatment, eventually leads to ESRD.
The rate of decline in eGFR once albuminuria is severely increased is equivalent in T1D and T2D.12 Without intervention, the time from severely increased albuminuria to ESRD in T1D and T2D averages approximately 6 or 7 years.
Clinical features
DKD is typically a clinical diagnosis seen in patients with longstanding diabetes, albuminuria, retinopathy, or a reduced eGFR in the absence of another primary cause of kidney damage. In patients with T1D and DKD, signs of retinopathy and neuropathy are almost always present at diagnosis, unless a diagnosis is made early in the course of diabetes.12 Therefore, the presence of retinopathy suggests that diabetes is the likely cause of CKD.
Continue to: The presence of microvascular disease...
The presence of microvascular disease in patients with T2D and DKD is less predictable.12 In T2D patients who do not have retinopathy, consider causes of CKD other than DKD. Features suggesting that the cause of CKD is an underlying condition other than diabetes are rapidly increasing albuminuria or decreasing eGFR; urinary sediment comprising red blood cells or white blood cells; and nephrotic syndrome.6
As the prevalence of diabetes increases, it has become more common to diagnose DKD by eGFR without albuminuria—underscoring the importance of routine monitoring of eGFR in patients with diabetes.6
Sources of expert guidance. The Chronic Kidney Disease Epidemiology Collaboration equation23 is preferred for calculating eGFR from serum creatinine: An eGFR < 60 mL/min/1.73 m2 is considered abnormal.3,12 At these rates, the prevalence of complications related to CKD rises and screening for complications becomes necessary.
A more comprehensive classification of the stages of CKD, incorporating albuminuria and progression of CKD, has been recommended by Kidney Disease: Improving Global Outcomes (KDIGO).7 Because eGFR and excretion of albumin vary, abnormal test results need to be verified over time to stage the degree of CKD.3,12 Kidney damage often manifests as albuminuria, but also as hematuria, other types of abnormal urinary sediment, radiographic abnormalities, and other abnormal presentations.
Management
Nutritional factors
Excessive protein intake has been shown to increase albuminuria, worsen renal function, and increase CVD mortality in DKD.24-26 Therefore, daily dietary protein intake of 0.8 g/kg body weight is recommended for patients who are not on dialysis.3 Patients on dialysis might require higher protein intake to preserve muscle mass caused by protein-energy wasting, which is common in dialysis patients.6
Continue to: Low sodium intake
Low sodium intake in CKD patients has been shown to decrease BP and thus slow the progression of renal disease and lower the risk of CVD. The recommended dietary sodium intake in CKD patients is 1500-3000 mg/d.3
Low potassium intake. Hyperkalemia is a serious complication of CKD. A low-potassium diet is recommended in ESRD patients who have a potassium level > 5.5 mEq/L.6
Blood pressure
Preventing and treating hypertension is critical to slowing the progression of CKD and reducing cardiovascular risk. BP should be measured at every clinic visit. Aside from lifestyle changes, medication might be needed to reach target BP.
The American Diabetes Association recommends a BP goal of ≤ 140/90 mm Hg for hypertensive patients with diabetes, although they do state that a lower BP target (≤ 130/80 mm Hg) might be more appropriate for patients with DKD.27
The American College of Cardiology recommends that hypertensive patients with CKD have a BP target of ≤ 130/80 mm Hg.28
Continue to: ACE inhibitors and ARBs
Angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARBs) have renoprotective benefits. These agents are recommended as first-line medications for patients with diabetes, hypertension, and an eGFR < 60 mL/min/1.73 m2 and a UACR > 300 mg/g.29-31 Evidence also supports their use when the UACR is 30 to 299 mg/g.
Studies have shown that, in patients with DKD, ACE inhibitors and ARBs can slow the progression of renal disease.29,30,32 There is no difference between ACE inhibitors and ARBs in their effectiveness for preventing progression of DKD.6 There is no added benefit in combining an ACE inhibitor and an ARB33; notably, combination ACE inhibitor and ARB therapy can increase the risk of adverse events, such as hyperkalemia and acute kidney injury, especially in patients with DKD.33
There is no evidence for starting an ACE inhibitor or ARB to prevent CKD in patients with diabetes who are not hypertensive.5
ACE inhibitors and ARBs should be used with caution in women of childbearing age, who should use a reliable form of contraception if taking one of these drugs.
Diuretics. Thiazide-type and loop diuretics might potentiate the positive effects of ACE inhibitors and ARBs. KDOQI guidelines recommend that, in patients who require a second agent to control BP, a diuretic should be considered in combination with an ACE inhibitor or an ARB.20 A loop diuretic is preferred if the eGFR is < 30 mL/min/1.73 m2.
Continue to: Nondihydropyridine calcium-channel blockers
Nondihydropyridine calcium-channel blockers (CCBs), such as diltiazem and verapamil, have been shown to be more effective then dihydrophyridine CCBs, such as amlodipine and nifedipine, in slowing the progression of renal disease because of their antiproteinuric effects. However, the antiproteinuric effects of nondihydropyridine CCBs are not as strong as those of ACE inhibitors or ARBs, and these drugs do not appear to potentiate the effects of an ACE inhibitor or ARB when used in combination.20
Nondihydropyridine CCBs might be a reasonable alternative in patients who cannot tolerate an ACE inhibitor or an ARB.
Mineralocorticoid receptor antagonists in combination with an ACE inhibitor or ARB have been demonstrated to reduce albuminuria in short-term studies.34,35
Glycemic levels
Studies conducted in patients with T1D, and others in patients with T2D, have shown that tight glycemic control can delay the onset and slow the progression of albuminuria and a decline in the eGFR.10,36-39 The target glycated hemoglobin (A1C) should be < 7% to prevent or slow progression of DKD.40 However, patients with DKD have an increased risk of hypoglycemic events and increased mortality with more intensive glycemic control.40,41 Given those findings, some patients with DKD and significant comorbidities, ESRD, or limited life expectancy might need to have an A1C target set at 8%.6,42
Adjustments to antidiabetes medications in DKD
In patients with stages 3 to 5 DKD, several common antidiabetic medications might need to be adjusted or discontinued because they decrease creatinine clearance.
Continue to: First-generation sulfonylureas
First-generation sulfonylureas should be avoided in DKD. Glipizide and gliclazide are preferred among second-generation sulfonylureas because they do not increase the risk of hypoglycemia in DKD patients, although patients taking these medications still require close monitoring of their blood glucose level.20
Metformin. In 2016, recommendations changed for the use of metformin in patients with DKD: The eGFR, not the serum creatinine level, should guide treatment.43 Metformin can be used safely in patients with (1) an eGFR of < 60 mL/min/1.73 m2 and (2) an eGFR of 30 mL/min/1.73 m2 with close monitoring. Metformin should not be initiated if the eGFR is < 45 mL/min/1.73 m2.43
Antidiabetes medications with direct effect on the kidney
Several antidiabetes medications have a direct effect on the kidney apart from their effect on the blood glucose level.
Sodium-glucose co-transporter 2 (SGLT2) inhibitors have been shown to reduce albuminuria and slow the decrease of eGFR independent of glycemic control. In addition, SGLT2 inhibitors have also been shown to have cardiovascular benefits in patients with DKD.44,45
Glucagon-like peptide 1 (GLP-1) receptor agonists have been shown to delay and decrease the progression of DKD.46-48 Also, similar to what is seen with SGLT2 inhibitors, GLP-1 agonists have demonstrable cardiovascular benefit in patients with DKD.46,48
Continue to: Dyslipidemia and DKD
Dyslipidemia and DKD
Because the risk of CVD is increased in patients with DKD, addressing other modifiable risk factors, including dyslipidemia, is recommended in these patients. Patients with diabetes and stages 1 to 4 DKD should be treated with a high-intensity statin or a combination of a statin and ezetimibe.49,50
If a patient is taking a statin and starting dialysis, it’s important to discuss with him or her whether to continue the statin, based on perceived benefits and risks. It is not recommended that statins be initiated in patients on dialysis unless there is a specific cardiovascular indication for doing so. Risk reduction with a statin has been shown to be significantly less in dialysis patients than in patients who are not being treated with dialysis.49
Complications of CKD
Anemia is a common complication of CKD. KDIGO recommends measuring the hemoglobin concentration annually in DKD stage 3 patients without anemia; at least every 6 months in stage 4 patients; and at least every 3 months in stage 5. DKD patients with anemia should have additional laboratory testing: the absolute reticulocyte count, serum ferritin, serum transferrin saturation, vitamin B12, and folate.51
Mineral and bone disorder should be screened for in patients with DKD. TABLE 252 outlines when clinical laboratory tests should be ordered to assess for mineral bone disease.

When to refer to a nephrologist
Refer patients with stage 4 or 5 CKD (eGFR, ≤ 30 mL/min/1.73 m2) to a nephrologist for discussion of kidney replacement therapy.6 Patients with stage 3a CKD and severely increased albuminuria or with stage 3b CKD and moderately or severely increased albuminuria should also be referred to a nephrologist for intervention to delay disease progression.
Continue to: Identifying the need for early referral...
Identifying the need for early referral to a nephrologist has been shown to reduce the cost, and improve the quality, of care.53 Other indications for earlier referral include uncertainty about the etiology of renal disease, persistent or severe albuminuria, persistent hematuria, a rapid decline in eGFR, and acute kidney injury. Additionally, referral at an earlier stage of DKD might be needed to assist with complications associated with DKD, such as anemia, secondary hyperparathyroidism, mineral and bone disorder, resistant hypertension, fluid overload, and electrolyte disturbances.6
ACKNOWLEDGEMENT
The authors thank Colleen Colbert, PhD, and Iqbal Ahmad, PhD, for their review and critique of the manuscript of this article. They also thank Christopher Babiuch, MD, for his guidance in the preparation of the manuscript.
CORRESPONDENCE
Faraz Ahmad, MD, MPH, Care Point East Family Medicine, 543 Taylor Avenue, 2nd floor, Columbus, OH 43203; faraz. [email protected].
Chronic kidney disease (CKD) is a significant comorbidity of diabetes mellitus. The Kidney Disease Outcomes Quality Initiative (KDOQI) of the National Kidney Foundation defines CKD as the presence of kidney damage or decreased kidney function for ≥ 3 months. CKD caused by diabetes is called diabetic kidney disease (DKD), which is 1 of 3 principal microvascular complications of diabetes. DKD can progress to end-stage renal disease (ESRD), requiring kidney replacement therapy, and is the leading cause of CKD and ESRD in the United States.1-3 Studies have also shown that, particularly in patients with diabetes, CKD considerably increases the risk of cardiovascular events, which often occur prior to ESRD.1,4
This article provides the latest recommendations for evaluating and managing DKD to help you prevent or slow its progression.
Defining and categorizing diabetic kidney disease
CKD is defined as persistently elevated excretion of urinary albumin (albuminuria) and decreased estimated glomerular filtration rate (eGFR), or as the presence of signs of progressive kidney damage.5,6 DKD, also known as diabetic nephropathy, is CKD attributed to long-term diabetes. A patient’s eGFR is the established basis for assignment to a stage (1, 2, 3a, 3b, 4, or 5) of CKD (TABLE 17) and, along with the category of albuminuria (A1, A2, or A3), can indicate prognosis.

Taking its toll in diabetes
As many as 40% of patients with diabetes develop DKD.8-10 Most studies of DKD have been conducted in patients with type 1 diabetes (T1D), because the time of clinical onset is typically known.
Type 1 diabetes. DKD usually occurs 10 to 15 years, or later, after the onset of diabetes.6 As many as 30% of people with T1D have albuminuria approximately 15 years after onset of diabetes; almost one-half of those develop DKD.5,11 After approximately 22.5 years without albuminuria, patients with T1D have approximately a 1% annual risk of DKD.12
Type 2 diabetes (T2D). DKD is often present at diagnosis, likely due to a delay in diagnosis and briefer clinical exposure, compared to T1D. Albuminuria has been reported in as many as 40% of patients with T2D approximately 10 years after onset of diabetes.12,13
Multiple risk factors with no standout “predictor”
Genetic susceptibility, ethnicity, glycemic control, smoking, blood pressure (BP), and the eGFR have been identified as risk factors for renal involvement in diabetes; obesity, oral contraceptives, and age can also contribute. Although each risk factor increases the risk of DKD, no single factor is adequately predictive. Moderately increased albuminuria, the earliest sign of DKD, is associated with progressive nephropathy.12
Continue to: How great is the risk?
How great is the risk? From disease onset to proteinuria and from proteinuria to ESRD, the risk of DKD in T1D and T2D is similar. With appropriate treatment, albuminuria can regress, and the risk of ESRD can be < 20% at 10 years in T1D.12 As in T1D, good glycemic control might result in regression of albuminuria in T2D.14
For unknown reasons, the degree of albuminuria can exist independent of the progression of DKD. Factors responsible for a progressive decline in eGFR in DKD without albuminuria are unknown.12,15
Patient evaluation with an eye toward comorbidities
A comprehensive initial medical evaluation for DKD includes a review of microvascular complications; visits to specialists; lifestyle and behavior patterns (eg, diet, sleep, substance use, and social support); and medication adherence, adverse drug effects, and alternative medicines. Although DKD is often a clinical diagnosis, it can be ruled in by persistent albuminuria or decreased eGFR, or both, in established diabetes or diabetic retinopathy when other causes are unlikely (see “Recommended DKD screening protocol,” below).
Screening for mental health conditions and barriers to self-management is also key.6
Comorbidities, of course, can complicate disease management in patients with diabetes.16-20 Providers and patients therefore need to be aware of potential diabetic comorbidities. For example, DKD and even moderately increased albuminuria significantly increase the risk of cardiovascular disease (CVD).12 Other possible comorbidities include (but are not limited to) nonalcoholic steatohepatitis, fracture, hearing impairment, cancer (eg, liver, pancreas, endometrium, colon, rectum, breast, and bladder), pancreatitis, hypogonadism, obstructive sleep apnea, periodontal disease, anxiety, depression, and eating disorders.6
Continue to: Recommended DKD screening protocol
Recommended DKD screening protocol
In all cases of T2D, in cases of T1D of ≥ 5 years’ duration, and in patients with diabetes and comorbid hypertension, perform annual screening for albuminuria, an elevated creatinine level, and a decline in eGFR.
To confirm the diagnosis of DKD, at least 2 of 3 urine specimens must demonstrate an elevated urinary albumin:creatinine ratio (UACR) over a 3- to 6-month period.21 Apart from renal damage, exercise within 24 hours before specimen collection, infection, fever, congestive heart failure, hyperglycemia, menstruation, and hypertension can elevate the UACR.6
Levels of the UACR are established as follows22:
- Normal UACR is defined as < 30 milligrams of albumin per gram of creatinine (expressed as “mg/g”).
- Increased urinary albumin excretion is defined as ≥ 30 mg/g.
- Moderately increased albuminuria, a predictor of potential nephropathy, is the excretion of 30 to 300 mg/g.
- Severely increased albuminuria is excretion > 300 mg/g; it is often followed by a gradual decline in eGFR that, without treatment, eventually leads to ESRD.
The rate of decline in eGFR once albuminuria is severely increased is equivalent in T1D and T2D.12 Without intervention, the time from severely increased albuminuria to ESRD in T1D and T2D averages approximately 6 or 7 years.
Clinical features
DKD is typically a clinical diagnosis seen in patients with longstanding diabetes, albuminuria, retinopathy, or a reduced eGFR in the absence of another primary cause of kidney damage. In patients with T1D and DKD, signs of retinopathy and neuropathy are almost always present at diagnosis, unless a diagnosis is made early in the course of diabetes.12 Therefore, the presence of retinopathy suggests that diabetes is the likely cause of CKD.
Continue to: The presence of microvascular disease...
The presence of microvascular disease in patients with T2D and DKD is less predictable.12 In T2D patients who do not have retinopathy, consider causes of CKD other than DKD. Features suggesting that the cause of CKD is an underlying condition other than diabetes are rapidly increasing albuminuria or decreasing eGFR; urinary sediment comprising red blood cells or white blood cells; and nephrotic syndrome.6
As the prevalence of diabetes increases, it has become more common to diagnose DKD by eGFR without albuminuria—underscoring the importance of routine monitoring of eGFR in patients with diabetes.6
Sources of expert guidance. The Chronic Kidney Disease Epidemiology Collaboration equation23 is preferred for calculating eGFR from serum creatinine: An eGFR < 60 mL/min/1.73 m2 is considered abnormal.3,12 At these rates, the prevalence of complications related to CKD rises and screening for complications becomes necessary.
A more comprehensive classification of the stages of CKD, incorporating albuminuria and progression of CKD, has been recommended by Kidney Disease: Improving Global Outcomes (KDIGO).7 Because eGFR and excretion of albumin vary, abnormal test results need to be verified over time to stage the degree of CKD.3,12 Kidney damage often manifests as albuminuria, but also as hematuria, other types of abnormal urinary sediment, radiographic abnormalities, and other abnormal presentations.
Management
Nutritional factors
Excessive protein intake has been shown to increase albuminuria, worsen renal function, and increase CVD mortality in DKD.24-26 Therefore, daily dietary protein intake of 0.8 g/kg body weight is recommended for patients who are not on dialysis.3 Patients on dialysis might require higher protein intake to preserve muscle mass caused by protein-energy wasting, which is common in dialysis patients.6
Continue to: Low sodium intake
Low sodium intake in CKD patients has been shown to decrease BP and thus slow the progression of renal disease and lower the risk of CVD. The recommended dietary sodium intake in CKD patients is 1500-3000 mg/d.3
Low potassium intake. Hyperkalemia is a serious complication of CKD. A low-potassium diet is recommended in ESRD patients who have a potassium level > 5.5 mEq/L.6
Blood pressure
Preventing and treating hypertension is critical to slowing the progression of CKD and reducing cardiovascular risk. BP should be measured at every clinic visit. Aside from lifestyle changes, medication might be needed to reach target BP.
The American Diabetes Association recommends a BP goal of ≤ 140/90 mm Hg for hypertensive patients with diabetes, although they do state that a lower BP target (≤ 130/80 mm Hg) might be more appropriate for patients with DKD.27
The American College of Cardiology recommends that hypertensive patients with CKD have a BP target of ≤ 130/80 mm Hg.28
Continue to: ACE inhibitors and ARBs
Angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARBs) have renoprotective benefits. These agents are recommended as first-line medications for patients with diabetes, hypertension, and an eGFR < 60 mL/min/1.73 m2 and a UACR > 300 mg/g.29-31 Evidence also supports their use when the UACR is 30 to 299 mg/g.
Studies have shown that, in patients with DKD, ACE inhibitors and ARBs can slow the progression of renal disease.29,30,32 There is no difference between ACE inhibitors and ARBs in their effectiveness for preventing progression of DKD.6 There is no added benefit in combining an ACE inhibitor and an ARB33; notably, combination ACE inhibitor and ARB therapy can increase the risk of adverse events, such as hyperkalemia and acute kidney injury, especially in patients with DKD.33
There is no evidence for starting an ACE inhibitor or ARB to prevent CKD in patients with diabetes who are not hypertensive.5
ACE inhibitors and ARBs should be used with caution in women of childbearing age, who should use a reliable form of contraception if taking one of these drugs.
Diuretics. Thiazide-type and loop diuretics might potentiate the positive effects of ACE inhibitors and ARBs. KDOQI guidelines recommend that, in patients who require a second agent to control BP, a diuretic should be considered in combination with an ACE inhibitor or an ARB.20 A loop diuretic is preferred if the eGFR is < 30 mL/min/1.73 m2.
Continue to: Nondihydropyridine calcium-channel blockers
Nondihydropyridine calcium-channel blockers (CCBs), such as diltiazem and verapamil, have been shown to be more effective then dihydrophyridine CCBs, such as amlodipine and nifedipine, in slowing the progression of renal disease because of their antiproteinuric effects. However, the antiproteinuric effects of nondihydropyridine CCBs are not as strong as those of ACE inhibitors or ARBs, and these drugs do not appear to potentiate the effects of an ACE inhibitor or ARB when used in combination.20
Nondihydropyridine CCBs might be a reasonable alternative in patients who cannot tolerate an ACE inhibitor or an ARB.
Mineralocorticoid receptor antagonists in combination with an ACE inhibitor or ARB have been demonstrated to reduce albuminuria in short-term studies.34,35
Glycemic levels
Studies conducted in patients with T1D, and others in patients with T2D, have shown that tight glycemic control can delay the onset and slow the progression of albuminuria and a decline in the eGFR.10,36-39 The target glycated hemoglobin (A1C) should be < 7% to prevent or slow progression of DKD.40 However, patients with DKD have an increased risk of hypoglycemic events and increased mortality with more intensive glycemic control.40,41 Given those findings, some patients with DKD and significant comorbidities, ESRD, or limited life expectancy might need to have an A1C target set at 8%.6,42
Adjustments to antidiabetes medications in DKD
In patients with stages 3 to 5 DKD, several common antidiabetic medications might need to be adjusted or discontinued because they decrease creatinine clearance.
Continue to: First-generation sulfonylureas
First-generation sulfonylureas should be avoided in DKD. Glipizide and gliclazide are preferred among second-generation sulfonylureas because they do not increase the risk of hypoglycemia in DKD patients, although patients taking these medications still require close monitoring of their blood glucose level.20
Metformin. In 2016, recommendations changed for the use of metformin in patients with DKD: The eGFR, not the serum creatinine level, should guide treatment.43 Metformin can be used safely in patients with (1) an eGFR of < 60 mL/min/1.73 m2 and (2) an eGFR of 30 mL/min/1.73 m2 with close monitoring. Metformin should not be initiated if the eGFR is < 45 mL/min/1.73 m2.43
Antidiabetes medications with direct effect on the kidney
Several antidiabetes medications have a direct effect on the kidney apart from their effect on the blood glucose level.
Sodium-glucose co-transporter 2 (SGLT2) inhibitors have been shown to reduce albuminuria and slow the decrease of eGFR independent of glycemic control. In addition, SGLT2 inhibitors have also been shown to have cardiovascular benefits in patients with DKD.44,45
Glucagon-like peptide 1 (GLP-1) receptor agonists have been shown to delay and decrease the progression of DKD.46-48 Also, similar to what is seen with SGLT2 inhibitors, GLP-1 agonists have demonstrable cardiovascular benefit in patients with DKD.46,48
Continue to: Dyslipidemia and DKD
Dyslipidemia and DKD
Because the risk of CVD is increased in patients with DKD, addressing other modifiable risk factors, including dyslipidemia, is recommended in these patients. Patients with diabetes and stages 1 to 4 DKD should be treated with a high-intensity statin or a combination of a statin and ezetimibe.49,50
If a patient is taking a statin and starting dialysis, it’s important to discuss with him or her whether to continue the statin, based on perceived benefits and risks. It is not recommended that statins be initiated in patients on dialysis unless there is a specific cardiovascular indication for doing so. Risk reduction with a statin has been shown to be significantly less in dialysis patients than in patients who are not being treated with dialysis.49
Complications of CKD
Anemia is a common complication of CKD. KDIGO recommends measuring the hemoglobin concentration annually in DKD stage 3 patients without anemia; at least every 6 months in stage 4 patients; and at least every 3 months in stage 5. DKD patients with anemia should have additional laboratory testing: the absolute reticulocyte count, serum ferritin, serum transferrin saturation, vitamin B12, and folate.51
Mineral and bone disorder should be screened for in patients with DKD. TABLE 252 outlines when clinical laboratory tests should be ordered to assess for mineral bone disease.

When to refer to a nephrologist
Refer patients with stage 4 or 5 CKD (eGFR, ≤ 30 mL/min/1.73 m2) to a nephrologist for discussion of kidney replacement therapy.6 Patients with stage 3a CKD and severely increased albuminuria or with stage 3b CKD and moderately or severely increased albuminuria should also be referred to a nephrologist for intervention to delay disease progression.
Continue to: Identifying the need for early referral...
Identifying the need for early referral to a nephrologist has been shown to reduce the cost, and improve the quality, of care.53 Other indications for earlier referral include uncertainty about the etiology of renal disease, persistent or severe albuminuria, persistent hematuria, a rapid decline in eGFR, and acute kidney injury. Additionally, referral at an earlier stage of DKD might be needed to assist with complications associated with DKD, such as anemia, secondary hyperparathyroidism, mineral and bone disorder, resistant hypertension, fluid overload, and electrolyte disturbances.6
ACKNOWLEDGEMENT
The authors thank Colleen Colbert, PhD, and Iqbal Ahmad, PhD, for their review and critique of the manuscript of this article. They also thank Christopher Babiuch, MD, for his guidance in the preparation of the manuscript.
CORRESPONDENCE
Faraz Ahmad, MD, MPH, Care Point East Family Medicine, 543 Taylor Avenue, 2nd floor, Columbus, OH 43203; faraz. [email protected].
1. Radbill B, Murphy B, LeRoith D. Rationale and strategies for early detection and management of diabetic kidney disease. Mayo Clin Proc. 2008;83:1373-1381.
2. Saran R, Robinson B, Abbott KC, et al. US Renal Data System 2017 Annual Data Report: Epidemiology of kidney disease in the United States. Am J Kidney Dis. 2018;71(3 suppl 1):A7.
3. Tuttle KR, Bakris GL, Bilous RW, et al. Diabetic kidney disease: a report from an ADA Consensus Conference. Am J Kidney Dis. 2014;64:510-533.
4. Fox CS, Matsushita K, Woodward M, et al; . Associations of kidney disease measures with mortality and end-stage renal disease in individuals with and without diabetes: a meta-analysis. Lancet. 2012;380:1662-1673.
5. Orchard TJ, Dorman JS, Maser RE, et al. Prevalence of complications in IDDM by sex and duration. Pittsburgh Epidemiology of Diabetes Complications Study II. Diabetes. 1990;39:1116-1124.
6. American Diabetes Association. Standards of Medical Care in Diabetes—2018. Diabetes Care. 2018;41(suppl 1):S1-S159. Accessed January 5, 2021. https://care.diabetesjournals.org/content/41/Supplement_1
7. National Kidney Foundation. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int Suppl. 2013;3:1-150. Accessed January 5, 2021. https://kdigo.org/wp-content/uploads/2017/02/KDIGO_2012_CKD_GL.pdf
8. Afkarian M, Zelnick LR, Hall YN, et al. Clinical manifestations of kidney disease among US adults with diabetes, 1988-2014. JAMA. 2016;316:602-610.
9. de Boer IH, Rue TC, Hall YN, et al. Temporal trends in the prevalence of diabetic kidney disease in the United States. JAMA. 2011;305:2532-2539.
10. de Boer IH; DCCT/EDIC Research Group. Kidney disease and related findings in the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study. Diabetes Care. 2014;37:24-30.
11. Stanton RC. Clinical challenges in diagnosis and management of diabetic kidney disease. Am J Kidney Dis. 2014;63(2 suppl 2):S3-S21.
12. Mottl AK, Tuttle KR. Diabetic kidney disease: Pathogenesis and epidemiology. UpToDate. Updated August 19, 2019. Accessed January 5, 2021. www.uptodate.com/contents/diabetic-kidney-disease-pathogenesis-and-epidemiology
13. Bakris GL. Moderately increased albuminuria (microalbuminuria) in type 2 diabetes mellitus. UpToDate. Updated November 3, 2020. Accessed January 5, 2021. https://www.uptodate.com/contents/moderately-increased-albuminuria-microalbuminuria-in-type-2-diabetes-mellitus
14. Bandak G, Sang Y, Gasparini A, et al. Hyperkalemia after initiating renin-angiotensin system blockade: the Stockholm Creatinine Measurements (SCREAM) Project. J Am Heart Assoc. 2017;6:e005428.
15. Saran R, Robinson B, Abbott KC, et al. US Renal Data System 2016 Annual Data Report: Epidemiology of kidney disease in the United States. Am J Kidney Dis. 2017;69(3 suppl 1):A7-A8.
16. Nilsson E, Gasparini A, Ärnlöv J, et al. Incidence and determinants of hyperkalemia and hypokalemia in a large healthcare system. Int J Cardiol. 2017;245:277-284.
17. de Boer IH, Gao X, Cleary PA, et al; Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) Research Group. Albuminuria changes and cardiovascular and renal outcomes in type 1 diabetes: The DCCT/EDIC study. Clin J Am Soc Nephrol. 2016;11:1969-1977.
18. Sumida K, Molnar MZ, Potukuchi PK, et al. Changes in albuminuria and subsequent risk of incident kidney disease. Clin J Am Soc Nephrol. 2017;12:1941-1949.
19. Borch-Johnsen K, Wenzel H, Viberti GC, et al. Is screening and intervention for microalbuminuria worthwhile in patient with insulin dependent diabetes? BMJ. 1993;306:1722-1725.
20. KDOQI. KDOQI clinical practice guidelines and clinical practice recommendations for diabetes and chronic kidney disease. Am J Kidney Dis. 2007;49(2 suppl 2):S12-154.
21. Bakris GL. Moderately increased albuminuria (microalbuminuria) in type 1 diabetes mellitus. UpToDate. Updated December 3, 2019. Accessed January 5, 2021. https://www.uptodate.com/contents/moderately-increased-albuminuria-microalbuminuria-in-type-1-diabetes-mellitus
22. Delanaye P, Glassock RJ, Pottel H, et al. An age-calibrated definition of chronic kidney disease: rationale and benefits. Clin Biochem Rev. 2016;37:17-26.
23. Levey AS, Stevens LA, Schmid CH, et al; , A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604-612.
24. Wrone EM, Carnethon MR, Palaniappan L, et al; . Association of dietary protein intake and microalbuminuria in healthy adults: Third National Health and Nutrition Examination Survey. Am J Kidney Dis. 2003;41:580-587.
25. Knight EL, Stampfer MJ, Hankinson SE, et al. The impact of protein intake on renal function decline in women with normal renal function or mild renal insufficiency. Ann Intern Med. 2003;138:460-467.
26. Bernstein AM, Sun Q, Hu FB, et al. Major dietary protein sources and risk of coronary heart disease in women. Circulation. 2010;122:876-883.
27. de Boer, IH, Bangalore S, Benetos A, et al. Diabetes and hypertension: a position statement by the American Diabetes Association. Diabetes Care. 2017;40:1273-1284.
28. Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018;71:e127-e248.
29. Brenner BM, Cooper ME, de Zeeuw D, et al; Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med. 2001;345:861-869.
30. Lewis EJ, Hunsicker LG, Bain RP, et al. The effect of angiotensin-converting-enzyme inhibition on diabetic nephropathy. The Collaborative Study Group. N Engl J Med. 1993;329:1456-1462.
31. Heart Outcomes Prevention Evaluation (HOPE) Study Investigators. Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: results of the HOPE study and MICRO-HOPE substudy. Lancet. 2000;355;253-259.
32. Lewis EJ, Hunsicker LG, Clarke WR, et al; . Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med. 2001;345:851-860.
33. Fried LF, Emanuele N, Zhang JH, et al; . Combined angiotensin inhibition for the treatment of diabetic nephropathy. N Engl J Med. 2013;369:1892-1903.
34. Bakris GL, Agarwal R, Chan JC, et al; . Effect of finerenone on albuminuria in patients with diabetic nephropathy: a randomized clinical trial. JAMA. 2015;314:884-894.
35. Filippatos G, Anker SD, M, et al. Randomized controlled study of finerenone vs. eplerenone in patients with worsening chronic heart failure and diabetes mellitus and/or chronic kidney disease. Eur Heart J. 2016;37:2105-2114.
36. The ADVANCE Collaborative Group. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes.N Engl J Med. 2008;358:2560-2572.
37. Ismail-Beigi F, Craven T, Banerji MA, et al; . Effect of intensive treatment of hyperglycaemia on microvascular outcomes in type 2 diabetes: an analysis of the ACCORD randomised trial. Lancet. 2010;376:419-430.
38. Zoungas S, Chalmers J, Neal B, et al; . Follow-up of blood-pressure lowering and glucose control in type 2 diabetes. N Engl J Med. 2014;371:1392-1406.
39. Zoungas S, Arima H, Gerstein HC, et al; . Effects of intensive glucose control on microvascular outcomes in patients with type 2 diabetes: a meta-analysis of individual participant data from randomised controlled trials. Lancet Diabetes Endocrinol. 2017;5:431-437.
40. Miller ME, Bonds DE, Gerstein HC, et al; . The effects of baseline characteristics, glycaemia treatment approach, and glycated haemoglobin concentration on the risk of severe hypoglycaemia: post hoc epidemiological analysis of the ACCORD study. BMJ. 2010;340;b5444.
41. Papademetriou V, Lovato L, Doumas M, et al; . Chronic kidney disease and intensive glycemic control increase cardiovascular risk in patients with type 2 diabetes. Kidney Int. 2015;87:649-659.
42. National Kidney Foundation. KDOQI clinical practice guideline for diabetes and CKD: 2012 Update. Am J Kidney Dis. 2012;60:850-886.
43. Imam TH. Changes in metformin use in chronic kidney disease. Clin Kidney J. 2017;10:301-304.
44. Wanner C, Inzucchi SE, Lachin JM, et al; Empagliflozin and progression of kidney disease in type 2 diabetes. N Engl J Med. 2016;375:323-334.
45. Neal B, Perkovic V, Mahaffey KW, et al; . Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med. 2017;377:644-657.
46. Marso SP, Daniels GH, Brown-Frandsen K, et al; . Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375:311-322.
47. Mann JFE, DD, Brown-Frandsen K, et al; . Liraglutide and renal outcomes in type 2 diabetes. N Engl J Med. 2017;377:839-848.
48. Marso SP, Bain SC, Consoli A, et al; . Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375:1834-1844.
49. Wanner C, Tonelli M; Kidney Disease: Improving Global Outcomes Lipid Guideline Development Work Group Members. KDIGO clinical practice guideline for lipid management in CKD: summary of recommendation statements and clinical approach to the patient. Kidney Int. 2014;85:1303-1309.
50. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol. A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139:e1082-e1143.
51. National Kidney Foundation KDOQI. KDIGO clinical practice guideline for anemia in chronic kidney disease. Kidney Int Suppl. 2012;2:279-335. Accessed January 5, 2021. www.sciencedirect.com/journal/kidney-international-supplements/vol/2/issue/4
52. National Kidney Foundation KDOQI. Evaluation and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). 2010. Accessed January 5, 2021. www.kidney.org/sites/default/files/02-10-390B_LBA_KDOQI_BoneGuide.pdf
53. Smart MA, Dieberg G, Ladhani M, et al. Early referral to specialist nephrology services for preventing the progression to end-stage kidney disease. Cochrane Database Syst Rev. 2014;(6):CD007333.
1. Radbill B, Murphy B, LeRoith D. Rationale and strategies for early detection and management of diabetic kidney disease. Mayo Clin Proc. 2008;83:1373-1381.
2. Saran R, Robinson B, Abbott KC, et al. US Renal Data System 2017 Annual Data Report: Epidemiology of kidney disease in the United States. Am J Kidney Dis. 2018;71(3 suppl 1):A7.
3. Tuttle KR, Bakris GL, Bilous RW, et al. Diabetic kidney disease: a report from an ADA Consensus Conference. Am J Kidney Dis. 2014;64:510-533.
4. Fox CS, Matsushita K, Woodward M, et al; . Associations of kidney disease measures with mortality and end-stage renal disease in individuals with and without diabetes: a meta-analysis. Lancet. 2012;380:1662-1673.
5. Orchard TJ, Dorman JS, Maser RE, et al. Prevalence of complications in IDDM by sex and duration. Pittsburgh Epidemiology of Diabetes Complications Study II. Diabetes. 1990;39:1116-1124.
6. American Diabetes Association. Standards of Medical Care in Diabetes—2018. Diabetes Care. 2018;41(suppl 1):S1-S159. Accessed January 5, 2021. https://care.diabetesjournals.org/content/41/Supplement_1
7. National Kidney Foundation. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int Suppl. 2013;3:1-150. Accessed January 5, 2021. https://kdigo.org/wp-content/uploads/2017/02/KDIGO_2012_CKD_GL.pdf
8. Afkarian M, Zelnick LR, Hall YN, et al. Clinical manifestations of kidney disease among US adults with diabetes, 1988-2014. JAMA. 2016;316:602-610.
9. de Boer IH, Rue TC, Hall YN, et al. Temporal trends in the prevalence of diabetic kidney disease in the United States. JAMA. 2011;305:2532-2539.
10. de Boer IH; DCCT/EDIC Research Group. Kidney disease and related findings in the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study. Diabetes Care. 2014;37:24-30.
11. Stanton RC. Clinical challenges in diagnosis and management of diabetic kidney disease. Am J Kidney Dis. 2014;63(2 suppl 2):S3-S21.
12. Mottl AK, Tuttle KR. Diabetic kidney disease: Pathogenesis and epidemiology. UpToDate. Updated August 19, 2019. Accessed January 5, 2021. www.uptodate.com/contents/diabetic-kidney-disease-pathogenesis-and-epidemiology
13. Bakris GL. Moderately increased albuminuria (microalbuminuria) in type 2 diabetes mellitus. UpToDate. Updated November 3, 2020. Accessed January 5, 2021. https://www.uptodate.com/contents/moderately-increased-albuminuria-microalbuminuria-in-type-2-diabetes-mellitus
14. Bandak G, Sang Y, Gasparini A, et al. Hyperkalemia after initiating renin-angiotensin system blockade: the Stockholm Creatinine Measurements (SCREAM) Project. J Am Heart Assoc. 2017;6:e005428.
15. Saran R, Robinson B, Abbott KC, et al. US Renal Data System 2016 Annual Data Report: Epidemiology of kidney disease in the United States. Am J Kidney Dis. 2017;69(3 suppl 1):A7-A8.
16. Nilsson E, Gasparini A, Ärnlöv J, et al. Incidence and determinants of hyperkalemia and hypokalemia in a large healthcare system. Int J Cardiol. 2017;245:277-284.
17. de Boer IH, Gao X, Cleary PA, et al; Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) Research Group. Albuminuria changes and cardiovascular and renal outcomes in type 1 diabetes: The DCCT/EDIC study. Clin J Am Soc Nephrol. 2016;11:1969-1977.
18. Sumida K, Molnar MZ, Potukuchi PK, et al. Changes in albuminuria and subsequent risk of incident kidney disease. Clin J Am Soc Nephrol. 2017;12:1941-1949.
19. Borch-Johnsen K, Wenzel H, Viberti GC, et al. Is screening and intervention for microalbuminuria worthwhile in patient with insulin dependent diabetes? BMJ. 1993;306:1722-1725.
20. KDOQI. KDOQI clinical practice guidelines and clinical practice recommendations for diabetes and chronic kidney disease. Am J Kidney Dis. 2007;49(2 suppl 2):S12-154.
21. Bakris GL. Moderately increased albuminuria (microalbuminuria) in type 1 diabetes mellitus. UpToDate. Updated December 3, 2019. Accessed January 5, 2021. https://www.uptodate.com/contents/moderately-increased-albuminuria-microalbuminuria-in-type-1-diabetes-mellitus
22. Delanaye P, Glassock RJ, Pottel H, et al. An age-calibrated definition of chronic kidney disease: rationale and benefits. Clin Biochem Rev. 2016;37:17-26.
23. Levey AS, Stevens LA, Schmid CH, et al; , A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604-612.
24. Wrone EM, Carnethon MR, Palaniappan L, et al; . Association of dietary protein intake and microalbuminuria in healthy adults: Third National Health and Nutrition Examination Survey. Am J Kidney Dis. 2003;41:580-587.
25. Knight EL, Stampfer MJ, Hankinson SE, et al. The impact of protein intake on renal function decline in women with normal renal function or mild renal insufficiency. Ann Intern Med. 2003;138:460-467.
26. Bernstein AM, Sun Q, Hu FB, et al. Major dietary protein sources and risk of coronary heart disease in women. Circulation. 2010;122:876-883.
27. de Boer, IH, Bangalore S, Benetos A, et al. Diabetes and hypertension: a position statement by the American Diabetes Association. Diabetes Care. 2017;40:1273-1284.
28. Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018;71:e127-e248.
29. Brenner BM, Cooper ME, de Zeeuw D, et al; Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med. 2001;345:861-869.
30. Lewis EJ, Hunsicker LG, Bain RP, et al. The effect of angiotensin-converting-enzyme inhibition on diabetic nephropathy. The Collaborative Study Group. N Engl J Med. 1993;329:1456-1462.
31. Heart Outcomes Prevention Evaluation (HOPE) Study Investigators. Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: results of the HOPE study and MICRO-HOPE substudy. Lancet. 2000;355;253-259.
32. Lewis EJ, Hunsicker LG, Clarke WR, et al; . Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med. 2001;345:851-860.
33. Fried LF, Emanuele N, Zhang JH, et al; . Combined angiotensin inhibition for the treatment of diabetic nephropathy. N Engl J Med. 2013;369:1892-1903.
34. Bakris GL, Agarwal R, Chan JC, et al; . Effect of finerenone on albuminuria in patients with diabetic nephropathy: a randomized clinical trial. JAMA. 2015;314:884-894.
35. Filippatos G, Anker SD, M, et al. Randomized controlled study of finerenone vs. eplerenone in patients with worsening chronic heart failure and diabetes mellitus and/or chronic kidney disease. Eur Heart J. 2016;37:2105-2114.
36. The ADVANCE Collaborative Group. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes.N Engl J Med. 2008;358:2560-2572.
37. Ismail-Beigi F, Craven T, Banerji MA, et al; . Effect of intensive treatment of hyperglycaemia on microvascular outcomes in type 2 diabetes: an analysis of the ACCORD randomised trial. Lancet. 2010;376:419-430.
38. Zoungas S, Chalmers J, Neal B, et al; . Follow-up of blood-pressure lowering and glucose control in type 2 diabetes. N Engl J Med. 2014;371:1392-1406.
39. Zoungas S, Arima H, Gerstein HC, et al; . Effects of intensive glucose control on microvascular outcomes in patients with type 2 diabetes: a meta-analysis of individual participant data from randomised controlled trials. Lancet Diabetes Endocrinol. 2017;5:431-437.
40. Miller ME, Bonds DE, Gerstein HC, et al; . The effects of baseline characteristics, glycaemia treatment approach, and glycated haemoglobin concentration on the risk of severe hypoglycaemia: post hoc epidemiological analysis of the ACCORD study. BMJ. 2010;340;b5444.
41. Papademetriou V, Lovato L, Doumas M, et al; . Chronic kidney disease and intensive glycemic control increase cardiovascular risk in patients with type 2 diabetes. Kidney Int. 2015;87:649-659.
42. National Kidney Foundation. KDOQI clinical practice guideline for diabetes and CKD: 2012 Update. Am J Kidney Dis. 2012;60:850-886.
43. Imam TH. Changes in metformin use in chronic kidney disease. Clin Kidney J. 2017;10:301-304.
44. Wanner C, Inzucchi SE, Lachin JM, et al; Empagliflozin and progression of kidney disease in type 2 diabetes. N Engl J Med. 2016;375:323-334.
45. Neal B, Perkovic V, Mahaffey KW, et al; . Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med. 2017;377:644-657.
46. Marso SP, Daniels GH, Brown-Frandsen K, et al; . Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375:311-322.
47. Mann JFE, DD, Brown-Frandsen K, et al; . Liraglutide and renal outcomes in type 2 diabetes. N Engl J Med. 2017;377:839-848.
48. Marso SP, Bain SC, Consoli A, et al; . Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375:1834-1844.
49. Wanner C, Tonelli M; Kidney Disease: Improving Global Outcomes Lipid Guideline Development Work Group Members. KDIGO clinical practice guideline for lipid management in CKD: summary of recommendation statements and clinical approach to the patient. Kidney Int. 2014;85:1303-1309.
50. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol. A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139:e1082-e1143.
51. National Kidney Foundation KDOQI. KDIGO clinical practice guideline for anemia in chronic kidney disease. Kidney Int Suppl. 2012;2:279-335. Accessed January 5, 2021. www.sciencedirect.com/journal/kidney-international-supplements/vol/2/issue/4
52. National Kidney Foundation KDOQI. Evaluation and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). 2010. Accessed January 5, 2021. www.kidney.org/sites/default/files/02-10-390B_LBA_KDOQI_BoneGuide.pdf
53. Smart MA, Dieberg G, Ladhani M, et al. Early referral to specialist nephrology services for preventing the progression to end-stage kidney disease. Cochrane Database Syst Rev. 2014;(6):CD007333.
PRACTICE RECOMMENDATIONS
› Screen patients with diabetes annually for diabetic kidney disease with measurement of urinary albumin and the estimated glomerular filtration rate. B
› Optimize blood glucose and blood pressure control in patients with diabetes to prevent or delay progression to diabetic kidney disease. A
› Treat hypertensive patients with diabetes and stages 1 to 4 chronic kidney disease with an angiotensin-converting enzyme inhibitor or angiotensin II-receptor blocker as a first-line antihypertensive, absent contraindications. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Breaking the cycle of medication overuse headache
Medication overuse headache (MOH), a secondary headache diagnosis, is a prevalent phenomenon that complicates headache diagnosis and treatment, increases the cost of care, and reduces quality of life. Effective abortive medication is essential for the headache sufferer; when an abortive is used too frequently, however, headache frequency increases—potentially beginning a cycle in which the patient then takes more medication to abort the headache. Over time, the patient suffers from an ever-increasing number of headaches, takes even more abortive medication, and so on. In the presence of MOH, there is a reduction in pain response to preventive and abortive treatments; when medication overuse is eliminated, pain response improves.1
Although MOH is well recognized among headache specialists, the condition is often overlooked in primary care. Since headache is a top complaint in primary care, however, and prevention is a major goal in family medicine, the opportunity for you to recognize, treat, and prevent MOH is significant. In fact, a randomized controlled trial showed that brief patient education about headache care and MOH provided by a primary care physician can lead to a significant reduction in headache frequency among patients with MOH.2
This article reviews the recognition and diagnosis of MOH, based on historical features and current criteria; addresses risk factors for abortive medication overuse and how to withdraw an offending agent; and explores the value of bridging and preventive therapies to reduce the overall frequency of headache.

What defines MOH?
Typically, MOH is a chronification of a primary headache disorder. However, in patients with a history of migraine who are undergoing treatment for another chronic pain condition with an opioid or other analgesic, MOH can be induced.3 An increase in the frequency of headache raises the specter of a concomitant increase in the level of disability4; psychiatric comorbidity5; and more headache days, with time lost from school and work.
The Migraine Disability Assessment (MIDAS) questionnaire, a validated instrument that helps the provider (1) measure the impact that headache has on a patient’s life and (2) follow treatment progress, also provides information to employers and insurance companies on treatment coverage and the need for work modification. The MIDAS score is 3 times higher in patients with MOH than in patients with episodic migraine.6,7
The annual associated cost per person of MOH has been estimated at $4000, resulting in billions of dollars in associated costs8; most of these costs are related to absenteeism and disability. After detoxification for MOH, annual outpatient medication costs are reduced by approximately 24%.9
Efforts to solve a common problem create another
Headache affects nearly 50% of the general population worldwide,10 accounting for about 4% of primary care visits11 and approximately 20% of outpatient neurology consultations.12 Although inpatient stays for headache are approximately half the duration of the overall average hospital stay, headache accounts for 3% of admissions.13 According to the Global Burden of Disease study, tension-type headache, migraine, and MOH are the 3 most common headache disorders.10 Headache is the second leading cause of disability among people 15 to 49 years of age.10
Continue to: The prevalence of MOH...
The prevalence of MOH in the general population is 2%.7,14,15 A population-based study showed that the rate of progression from episodic headache (< 15 d/mo) to chronic headache (≥ 15 d/mo) in the general population is 2.5% per year16; however, progression to chronic headache is 14% per year in patients with medication overuse. One-third of the general population with chronic migraine overuses symptomatic medication; in US headache clinics, roughly one-half of patients with chronic headache overuse acute medication.6
Definitions and diagnosis
MOH is a secondary headache diagnosis in the third edition of the International Classification of Headache Disorders (ICHD-3) (TABLE 1),17 which lists diagnostic criteria for recognized headache disorders.

Terminology. MOH has also been called rebound headache, drug-induced headache, and transformed migraine, but these terms are outdated and are not formal diagnoses. Patients sometimes refer to substance-withdrawal headaches (not discussed in this article) as rebound headaches, so clarity is important when discussing headache with patients: namely, that MOH is an exacerbation of an existing headache condition caused by overuse of abortive headache medications, including analgesics, combination analgesics, triptans, barbiturates, and opioids.
MOH was recognized in the early 1950s and fully differentiated as a diagnosis in 2005 in the second edition of the ICHD. The disorder is subcategorized by offending abortive agent (TABLE 217) because the frequency of analgesic use required to develop MOH differs by agent.

Risk factors for MOH and chronification of a primary headache disorder. There are several risk factors for developing MOH, and others that contribute to increasing headache frequency in general (TABLE 35,14,18-23). Some risk factors are common to each. All are important to address because some are modifiable.
Continue to: Pathophysiology
Pathophysiology. The pathophysiology and psychology behind MOH are largely unknown. Physiologic changes in pain processing and functional imaging changes have been demonstrated in patients with MOH, both of which are reversible upon withdrawal of medication.23 Genetic factors and changes in hormone and neurotransmitter levels are found in MOH patients; this is not the case in patients who have an episodic headache pattern only.24
Presentation. Diagnostic criteria for MOH do not include clinical characteristics. Typically, the phenotype of MOH in a given patient is similar to the underlying primary headache25—although this principle can be complicated to tease out because these medications can suppress some symptoms. Diagnosis of a primary headache disorder should be documented along with the diagnosis of MOH.
Medication overuse can exist without MOH: Not every patient who frequently uses an abortive medication develops MOH.
Treatment is multifaceted—and can become complex
Mainstays of treatment of MOH are education about the disorder and detoxification from the overused agent, although specific treatments can differ depending on the agent involved, the frequency and duration of its use, and a patient’s behavioral patterns and psychiatric comorbidities. Often, a daily medication to prevent headache is considered upon, or after, withdrawal of the offending agent. The timing of introducing a preventive might impact its effectiveness. Some refractory cases require more intensive therapy, including hospitalization at a specialized tertiary center.
But before we look at detoxification from an overused agent, it’s important to review one of the best strategies of all in combatting MOH.
Continue to: First and best strategy
First and best strategy: Avoid onset of MOH
Select an appropriate abortive to reduce the risk of MOH. With regard to specific acute headache medications, some nuances other than type of headache should be considered. Nonsteroidal anti-inflammatory drugs (NSAIDs) are recommended as abortive therapy by the American Headache Society for their efficacy, favorable adverse effect profile, and low cost. NSAIDs are protective against development of MOH if a patient’s baseline headache frequency is < 10/mo; at a frequency of 10 to 14 d/mo, however, the risk of MOH increases when using an NSAID.6 A similar effect has been seen with triptans.16 Longer-acting NSAIDs, such as nabumetone and naproxen, have been proposed as less likely to cause MOH, and are even used as bridging therapy sometimes (as long as neither of these was the overused medication).26
The time it takes to develop MOH is shortest with triptans, followed by ergots, then analgesics.27
Prospective cohort studies6,16 have shown that barbiturates and opioids are more likely to induce MOH; for that reason, agents in these analgesic classes are almost universally avoided unless no other medically acceptable options exist. Using barbiturate-containing compounds or opioids > 4 d/mo exponentially increases the likelihood of MOH.
Promising preclinical data demonstrate that the gepant, or small-molecule calcitonin gene-related peptide (CGRP) receptor antagonist, class of medications used as abortive therapy does not induce medication overuse cutaneous allodynia.28
Provide education. Primary prevention of MOH involves (1) increasing patients’ awareness of how to take medications appropriately and (2) restricting intake of over-the-counter abortive medications. Often, the expert recommendation is to limit abortives to approximately 2 d/wk because more frequent use places patients at risk of further increased use and subsequent MOH.
Continue to: A randomized controlled trial in Norway...
A randomized controlled trial in Norway compared outcomes in 2 groups of patients with MOH: One group was given advice on the disorder by a physician; the other group was not provided with advice. In the “business-as-usual” group, there was no significant improvement; however, when general practitioners provided simple advice (lasting roughly 9 minutes) about reducing abortive medication use to a safe level and cautioned patients that they would be “feeling worse before feeling better,” headache days were reduced by approximately 8 per month and medication days, by 16 per month.2
A subsequent, long-term follow-up study29 of patients from the Norway trial2 who had been given advice and education showed a relapse rate (ie, into overuse of headache medication) of only 8% and sustained reduction of headache days and medication use at 16 months.
Offer support and other nondrug interventions. A recent review of 3 studies23 recommended that extra support for patients from a headache nurse, close follow-up, keeping an electronic diary that provides feedback, and undertaking a short course of psychotherapy can reduce medication overuse and prevent relapse.
If MOH develops, initiate withdrawal, introduce a preventive
Withdraw overused medication. Most current evidence suggests that withdrawal of the offending agent is the most effective factor in reducing headache days and improving quality of life. A randomized controlled trial compared the effects of (1) complete and immediate withdrawal of an abortive medication with (2) reducing its use (ie, limiting intake to 2 d/wk), on headache frequency, disability, and quality of life.30 There was a reduction of headache days in both groups; however, reduction was much greater at 2 months in the complete withdrawal group than in the restricted intake group (respectively, a 41% and a 26% reduction in headache days per month). This effect was sustained at 6 and 12 months in both groups. The study confirmed the results of earlier research2,15: Abrupt withdrawal leads to reversion to an episodic pattern at 2 to 6 months in approximately 40% to 60% of patients.
More studies are needed to determine the most appropriate treatment course for MOH; however, complete withdrawal of the causative drug is the most important intervention.
Continue to: Consider withdrawal plus preventive treatment
Consider withdrawal plus preventive treatment. Use of sodium valproate, in addition to medication overuse detoxification, led to a significant reduction in headache days and improvement in quality of life at 12 weeks but no difference after 24 weeks, compared with detoxification alone in a randomized, double-blind, placebo-controlled study.31
A study of 61 patients showed a larger reduction (by 7.2 d/mo) in headache frequency with any preventive medication in addition to medication withdrawal, compared to withdrawal alone (by 4.1 d/mo) after 3 months; however, the relative benefit was gone at 6 months.32
A study of 98 patients compared immediate and delayed initiation of preventive medication upon withdrawal of overused abortive medication.33 Response was defined as a > 50% reduction in headache frequency and was similar in both groups; results showed a 28% response with immediate initiation of a preventive; a 23% response with delayed (ie, 2 months after withdrawal) initiation; and a 48% response in both groups at 12 months.
Collectively, these studies suggest that adding a preventive medication at the time of withdrawal has the potential to reduce headache frequency more quickly than withdrawal alone. However, after 3 to 6 months, the outcome of reduced headache frequency is the same whether or not a preventive medication is used—as long as the offending agent has been withdrawn.
Do preventives work without withdrawing overused medication? Patients with MOH often show little or no improvement with addition of a preventive medication only; their response to a preventive improves after withdrawal of the overused medication. Patients without previous headache improvement after addition of a preventive, who also did not improve 2 months after withdrawal, then demonstrated an overall reduction in headache by 26% when a preventive was reintroduced after withdrawal.2
Continue to: The research evidence for preventives
The research evidence for preventives. Medications for headache prevention have not been extensively evaluated specifically for treating MOH. Here is what’s known:
- Flunarizine, amitriptyline, and beta-blockers usually are ineffective for MOH.24
- Results for topiramate are mixed: A small, double-blind, placebo-controlled chronic migraine study in Europe showed that, in a subgroup of patients with MOH, topiramate led to a small but significant reduction (3.5 d/mo) in headache frequency, compared to placebo.27 A similar study done in the United States did not show a significant difference between the active-treatment and placebo groups.34
- Findings regarding onabotulinumtoxinA are intriguing: In a posthoc analysis of onabotulinumtoxinA to treat chronic migraine, patients with MOH who did not undergo detoxification had an 8 d/mo greater reduction in headache, compared to placebo.35 However, when compared to placebo in conjunction with detoxification, onabotulinumtoxinA demonstrated no benefit.36
- Newer CGRP antagonist and CGRP receptor antagonist monoclonal antibodies are successful preventive medications that have demonstrated a reduction in acute medication use days per month and headache days per month37; these compounds have not been compared to withdrawal alone.
Reducing the severity and duration of withdrawal symptoms
Withdrawal from overused abortive headache medications can lead to worsening headache, nausea, vomiting, hypotension, tachycardia, sleep disturbances, restlessness, anxiety, and nervousness. Symptoms usually last 2 to 10 days but can persist for as long as 4 weeks; duration of withdrawal symptoms varies with the medication that is being overused. In patients who have used a triptan, for example, mean duration of withdrawal is 4.1 days; ergotamine, 6.7 days; and NSAIDs, 9.5 days.23 Tapered withdrawal is sometimes recommended with opioids and barbiturates to reduce withdrawal symptoms. It is unclear whether starting a preventive medication during withdrawal assists in reducing withdrawal symptoms.38
Bridging therapy to reduce symptoms of withdrawal is often provided despite debatable utility. Available evidence does not favor one agent or method but suggests some strategies that could be helpful:
- A prednisone taper has a potential role during the first 6 days of withdrawal by reducing rebound headache and withdrawal symptoms39; however, oral prednisolone has been shown to have no benefit.40
- Alone, IV methylprednisolone seems not to be of benefit; however, in a retrospective study of 94 patients, IV methylprednisolone plus diazepam for 5 days led to a significant reduction in headache frequency and drug consumption that was sustained after 3 months.41
- Celecoxib was compared to prednisone over a 20-day course: a celecoxib dosage of 400 mg/d for the first 5 days, tapered by 100 mg every 5 days, and an oral prednisone dosage of 75 mg/d for the first 5 days, then tapered every 5 days. Patients taking celecoxib had lower headache intensity but there was no difference in headache frequency and acute medication intake between the groups.42
Other strategies. Using antiemetics and NSAIDs to reduce withdrawal symptoms is widely practiced, but no placebo-controlled trials have been conducted to support this strategy.
Patients in withdrawal might be more likely to benefit from inpatient care if they have a severe comorbidity, such as opioid or barbiturate use; failure to respond to, tolerate, or adhere to treatment; or relapse after withdrawal.38
Continue to: Cognitive behavioral therapy...
Cognitive behavioral therapy, exercise, a headache diary, and biofeedback should be considered in every patient’s treatment strategy because a multidisciplinary approach increases adherence and leads to improvement in headache frequency and a decrease in disability and medication use.43
Predictors of Tx success
A prospective cohort study determined that the rate of MOH relapse is 31% at 6 months, 41% at 1 year, and 45% at 4 years, with the highest risk of relapse during the first year.44 Looking at the correlation between type of medication overused and relapse rate, the research indicates that
- triptans have the lowest risk of relapse,44
- simple analgesics have a higher risk of relapse than triptans,22,44 and
- opioids have the highest risk of relapse.22
Where the data don’t agree. Data on combination analgesics and on ergots are conflicting.22 In addition, data on whether the primary type of headache predicts relapse rate conflict; however, migraine might predict a better outcome than tension-type headache.22
To recap and expand: Management pearls
The major goals of headache management generally are to rule out secondary headache, reach a correct diagnosis, reduce overall headache frequency, and provide effective abortive medication. A large component of reducing headache frequency is addressing and treating medication overuse.
Seek to understand the nature of the patient’s headache disorder. Components of the history are key in identifying the underlying headache diagnosis and ruling out other, more concerning secondary headache diagnoses. The ICHD-3 is an excellent resource for treating headache disorders because the classification lists specific diagnostic criteria for all recognized headache diagnoses.
Continue to: Medication withdrawal...
Medication withdrawal—with or without preventive medication—should reduce the frequency of MOH in 2 or 3 months. If headache does not become less frequent, however, the headache diagnosis might need to be reconsidered. Minimizing the use of abortive medication is generally recommended, but reduction or withdrawal of these medications does not guarantee that patients will revert to an episodic pattern of headache.
Treating withdrawal symptoms is a reasonable approach in some patients, but evidence does not support routinely providing bridging therapy.
Apply preventives carefully. Abortive medication withdrawal should generally be completed before initiating preventive medication; however, over the short term, starting preventive therapy while withdrawing the overused medication could assist in reducing headache frequency rapidly. This strategy can put patients at risk of medication adverse effects and using the medications longer than necessary, yet might be reasonable in certain patients, given their comorbidities, risk of relapse, and physician and patient preference. A preventive medication for an individual patient should generally be chosen in line with recommendations of the American Academy of Neurology45 and on the basis of the history and comorbidities.
Provide education, which is essential to lowering barriers to success. Patients with MOH must be counseled to understand that (1) a headache treatment that is supposed to be making them feel better is, in fact, making them feel worse and (2) they will get worse before they get better. Many patients are afraid to be without medication to use as needed. It is helpful to educate them on the different types of treatments (abortive, preventive); how MOH interferes with headache prophylaxis and medication efficacy; how MOH alters brain function (ie, aforementioned physiologic changes in pain processing and functional imaging changes23); and that such change is reversible when medication is withdrawn.
ACKNOWLEDGEMENT
The author thanks Jeffrey Curtis, MD, MPH, for his support and editing assistance with the manuscript.
CORRESPONDENCE
Allison Crain, MD, 2927 N 7th Avenue, Phoenix, AZ 85013; [email protected].
1. Zeeberg P, Olesen J, Jensen R. Discontinuation of medication overuse in headache patients: recovery of therapeutic responsiveness. Cephalalgia. 2006;26:1192-1198.
2. Kristoffersen ES, Straand J, Vetvik KG, et al. Brief intervention for medication-overuse headache in primary care. The BIMOH study: a double-blind pragmatic cluster randomised parallel controlled trial. J Neurol Neurosurg Psychiatry. 2015;86:505-512.
3. Bahra A, Walsh M, Menon S, et al. Does chronic daily headache arise de novo in association with regular use of analgesics? Headache. 2003;43:179-190.
4. Blumenfeld AM, Varon SF, Wilcox TK, et al. Disability, HRQoL and resource use among chronic and episodic migraineurs: results from the International Burden of Migraine Study (IBMS) Cephalalgia. 2011;31:301-315.
5. Chu H-T, Liang C-S, Lee J-T, et al. Associations between depression/anxiety and headache frequency in migraineurs: a cross-sectional study. Headache. 2018;58:407-415.
6. Bigal ME, Lipton RB. Excessive acute migraine medication use and migraine progression. Neurology. 2008;71:1821-1828.
7. Colás R, Muñoz P, Temprano R, et al. Chronic daily headache with analgesic overuse: epidemiology and impact on quality of life. Neurology. 2004;62:1338-1342.
8. Linde M, Gustavsson A, Stovner LJ, et al. The cost of headache disorders in Europe: the Eurolight project. Eur J Neurol. 2012;19:703-711.
9. Shah AM, Bendtsen L, Zeeberg P, et al. Reduction of medication costs after detoxification for medication-overuse headache. Headache. 2013;53:665-672.
10. . Global, regional, and national burden of migraine and tension-type headache, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018;17:954-976.
11. Kernick D, Stapley S, Goadsby PJ, et al. What happens to new-onset headache presenting to primary care? A case–cohort study using electronic primary care records. Cephalalgia. 2008;28:1188-1195.
12. Stone J, Carson A, Duncan R, et al. Who is referred to neurology clinics?—the diagnoses made in 3781 new patients. Clin Neurol Neurosurg. 2010;112:747-751.
13. Munoz-Ceron J, Marin-Careaga V, L, et al. Headache at the emergency room: etiologies, diagnostic usefulness of the ICHD 3 criteria, red and green flags. PloS One. 2019;14:e0208728.
14. Evers S, Marziniak M. Clinical features, pathophysiology, and treatment of medication-overuse headache. Lancet Neurol. 2010;9:391-401.
15. Tassorelli C, Jensen R, Allena M, et al; the . A consensus protocol for the management of medication-overuse headache: evaluation in a multicentric, multinational study. Cephalalgia. 2014;34:645-655.
16. Bigal ME, Serrano D, Buse D, et al. Acute migraine medications and evolution from episodic to chronic migraine: a longitudinal population-based study. Headache. 2008;48:1157-1168.
17. Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211.
18. Ferrari A, Leone S, Vergoni AV, et al. Similarities and differences between chronic migraine and episodic migraine. Headache. 2007;47:65-72.
19. Hagen K, Linde M, Steiner TJ, et al. Risk factors for medication-overuse headache: an 11-year follow-up study. The Nord-Trøndelag Health Studies. Pain. 2012;153:56-61.
20. Katsarava Z, Schneewiess S, Kurth T, et al. Incidence and predictors for chronicity of headache in patients with episodic migraine. Neurology. 2004;62:788-790.
21. Lipton RB, Fanning KM, Buse DC, et al. Migraine progression in subgroups of migraine based on comorbidities: results of the CaMEO study. Neurology. 2019;93:e2224-e2236.
22. Munksgaard SB, Madsen SK, Wienecke T. Treatment of medication overuse headache—a review. Acta Neurol Scand. 2019;139:405-414.
23. Ferraro S, Grazzi L, Mandelli M, et al. Pain processing in medication overuse headache: a functional magnetic resonance imaging (fMRI) study. Pain Med. 2012;13:255-262.
24. Diener H-C, Holle D, Solbach K, et al. Medication-overuse headache: risk factors, pathophysiology and management. Nat Rev Neurol. 2016;12:575-583.
25. Limmroth V, Katsarava Z, Fritsche G, et al. Features of medication overuse headache following overuse of different acute headache drugs. Neurology. 2002;59:1011-1014.
26. Mauskop A, ed. Migraine and Headache. 2nd ed. Oxford University Press; 2013.
27. Diener H-C, Bussone G, Van Oene JC, et al; . Topiramate reduces headache days in chronic migraine: a randomized, double-blind, placebo-controlled study. Cephalalgia. 2007;27:814-823.
28. Navratilova E, Behravesh S, Oyarzo J, et al. Ubrogepant does not induce latent sensitization in a preclinical model of medication overuse headache Cephalalgia. 2020;40:892-902.
29. Kristoffersen ES, Straand J, Russell MB, et al. Lasting improvement of medication-overuse headache after brief intervention—a long-term follow-up in primary care. Eur J Neurol. 2017;24:883-891.
30. Carlsen LN, Munksgaard SB, Jensen RH, et al. Complete detoxification is the most effective treatment of medication-overuse headache: a randomized controlled open-label trial. Cephalalgia. 2018;38:225-236.
31. Sarchielli P, Messina P, Cupini LM, et al; SAMOHA Study Group. Sodium valproate in migraine without aura and medication overuse headache: a randomized controlled trial. Eur Neuropsychopharmacol. 2014;24:1289-1297.
32. Hagen K, Stovner LJ. A randomized controlled trial on medication-overuse headache: outcome after 1 and 4 years. Acta Neurol Scand Suppl. 2011;124(suppl 191):38-43.
33. Munksgaard SB, Bendtsen L, Jensen RH. Detoxification of medication-overuse headache by a multidisciplinary treatment programme is highly effective: a comparison of two consecutive treatment methods in an open-label design. Cephalalgia. 2012;32:834-844.
34. Silberstein S, Lipton R, Dodick D, et al. Topiramate treatment of chronic migraine: a randomized, placebo-controlled trial of quality of life and other efficacy measures. Headache. 2009;49:1153-1162.
35. Silberstein SD, Blumenfeld AM, Cady RK, et al. OnabotulinumtoxinA for treatment of chronic migraine: PREEMPT 24-week pooled subgroup analysis of patients who had acute headache medication overuse at baseline. J Neurol Sci. 2013;331:48-56.
36. Sandrini G, Perrotta A, Tassorelli C, et al. Botulinum toxin type-A in the prophylactic treatment of medication-overuse headache: a multicenter, double-blind, randomized, placebo-controlled, parallel group study. J Headache Pain. 2011;12:427-433.
37. Tepper SJ. CGRP and headache: a brief review. Neurol Sci. 2019;40(suppl 1):99-105.
38. Diener H-C, Dodick D, Evers S, et al. Pathophysiology, prevention and treatment of medication overuse headache. Lancet Neurol. 2019;18:891-902.
39. Krymchantowski AV, Barbosa JS. Prednisone as initial treatment of analgesic-induced daily headache. Cephalalgia. 2000;20:107-113.
40. Bøe MG, Mygland A, Salvesen R. Prednisolone does not reduce withdrawal headache: a randomized, double-blind study. Neurology. 2007;69:26-31.
41. Paolucci M, Altamura C, Brunelli N, et al. Methylprednisolone plus diazepam i.v. as bridge therapy for medication overuse headache. Neurol Sci. 2017;38:2025-2029.
42. Taghdiri F, Togha M, Razeghi Jahromi S, et al. Celecoxib vs prednisone for the treatment of withdrawal headache in patients with medication overuse headache: a randomized, double-blind clinical trial. Headache. 2015;55:128-135.
43. Ramsey RR, Ryan JL, Hershey AD, et al. Treatment adherence in patients with headache: a systematic review. Headache. 2014;54:795-816.
44. Katsarava Z, Muessig M, Dzagnidze A, et al. Medication overuse headache: rates and predictors for relapse in a 4-year prospective study. Cephalalgia. 2005;25:12-15.
45. Silberstein SD, Holland S, Freitag F, et al; . Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012; 78:1137-1145.
Medication overuse headache (MOH), a secondary headache diagnosis, is a prevalent phenomenon that complicates headache diagnosis and treatment, increases the cost of care, and reduces quality of life. Effective abortive medication is essential for the headache sufferer; when an abortive is used too frequently, however, headache frequency increases—potentially beginning a cycle in which the patient then takes more medication to abort the headache. Over time, the patient suffers from an ever-increasing number of headaches, takes even more abortive medication, and so on. In the presence of MOH, there is a reduction in pain response to preventive and abortive treatments; when medication overuse is eliminated, pain response improves.1
Although MOH is well recognized among headache specialists, the condition is often overlooked in primary care. Since headache is a top complaint in primary care, however, and prevention is a major goal in family medicine, the opportunity for you to recognize, treat, and prevent MOH is significant. In fact, a randomized controlled trial showed that brief patient education about headache care and MOH provided by a primary care physician can lead to a significant reduction in headache frequency among patients with MOH.2
This article reviews the recognition and diagnosis of MOH, based on historical features and current criteria; addresses risk factors for abortive medication overuse and how to withdraw an offending agent; and explores the value of bridging and preventive therapies to reduce the overall frequency of headache.

What defines MOH?
Typically, MOH is a chronification of a primary headache disorder. However, in patients with a history of migraine who are undergoing treatment for another chronic pain condition with an opioid or other analgesic, MOH can be induced.3 An increase in the frequency of headache raises the specter of a concomitant increase in the level of disability4; psychiatric comorbidity5; and more headache days, with time lost from school and work.
The Migraine Disability Assessment (MIDAS) questionnaire, a validated instrument that helps the provider (1) measure the impact that headache has on a patient’s life and (2) follow treatment progress, also provides information to employers and insurance companies on treatment coverage and the need for work modification. The MIDAS score is 3 times higher in patients with MOH than in patients with episodic migraine.6,7
The annual associated cost per person of MOH has been estimated at $4000, resulting in billions of dollars in associated costs8; most of these costs are related to absenteeism and disability. After detoxification for MOH, annual outpatient medication costs are reduced by approximately 24%.9
Efforts to solve a common problem create another
Headache affects nearly 50% of the general population worldwide,10 accounting for about 4% of primary care visits11 and approximately 20% of outpatient neurology consultations.12 Although inpatient stays for headache are approximately half the duration of the overall average hospital stay, headache accounts for 3% of admissions.13 According to the Global Burden of Disease study, tension-type headache, migraine, and MOH are the 3 most common headache disorders.10 Headache is the second leading cause of disability among people 15 to 49 years of age.10
Continue to: The prevalence of MOH...
The prevalence of MOH in the general population is 2%.7,14,15 A population-based study showed that the rate of progression from episodic headache (< 15 d/mo) to chronic headache (≥ 15 d/mo) in the general population is 2.5% per year16; however, progression to chronic headache is 14% per year in patients with medication overuse. One-third of the general population with chronic migraine overuses symptomatic medication; in US headache clinics, roughly one-half of patients with chronic headache overuse acute medication.6
Definitions and diagnosis
MOH is a secondary headache diagnosis in the third edition of the International Classification of Headache Disorders (ICHD-3) (TABLE 1),17 which lists diagnostic criteria for recognized headache disorders.

Terminology. MOH has also been called rebound headache, drug-induced headache, and transformed migraine, but these terms are outdated and are not formal diagnoses. Patients sometimes refer to substance-withdrawal headaches (not discussed in this article) as rebound headaches, so clarity is important when discussing headache with patients: namely, that MOH is an exacerbation of an existing headache condition caused by overuse of abortive headache medications, including analgesics, combination analgesics, triptans, barbiturates, and opioids.
MOH was recognized in the early 1950s and fully differentiated as a diagnosis in 2005 in the second edition of the ICHD. The disorder is subcategorized by offending abortive agent (TABLE 217) because the frequency of analgesic use required to develop MOH differs by agent.

Risk factors for MOH and chronification of a primary headache disorder. There are several risk factors for developing MOH, and others that contribute to increasing headache frequency in general (TABLE 35,14,18-23). Some risk factors are common to each. All are important to address because some are modifiable.

Continue to: Pathophysiology
Pathophysiology. The pathophysiology and psychology behind MOH are largely unknown. Physiologic changes in pain processing and functional imaging changes have been demonstrated in patients with MOH, both of which are reversible upon withdrawal of medication.23 Genetic factors and changes in hormone and neurotransmitter levels are found in MOH patients; this is not the case in patients who have an episodic headache pattern only.24
Presentation. Diagnostic criteria for MOH do not include clinical characteristics. Typically, the phenotype of MOH in a given patient is similar to the underlying primary headache25—although this principle can be complicated to tease out because these medications can suppress some symptoms. Diagnosis of a primary headache disorder should be documented along with the diagnosis of MOH.
Medication overuse can exist without MOH: Not every patient who frequently uses an abortive medication develops MOH.
Treatment is multifaceted—and can become complex
Mainstays of treatment of MOH are education about the disorder and detoxification from the overused agent, although specific treatments can differ depending on the agent involved, the frequency and duration of its use, and a patient’s behavioral patterns and psychiatric comorbidities. Often, a daily medication to prevent headache is considered upon, or after, withdrawal of the offending agent. The timing of introducing a preventive might impact its effectiveness. Some refractory cases require more intensive therapy, including hospitalization at a specialized tertiary center.
But before we look at detoxification from an overused agent, it’s important to review one of the best strategies of all in combatting MOH.
Continue to: First and best strategy
First and best strategy: Avoid onset of MOH
Select an appropriate abortive to reduce the risk of MOH. With regard to specific acute headache medications, some nuances other than type of headache should be considered. Nonsteroidal anti-inflammatory drugs (NSAIDs) are recommended as abortive therapy by the American Headache Society for their efficacy, favorable adverse effect profile, and low cost. NSAIDs are protective against development of MOH if a patient’s baseline headache frequency is < 10/mo; at a frequency of 10 to 14 d/mo, however, the risk of MOH increases when using an NSAID.6 A similar effect has been seen with triptans.16 Longer-acting NSAIDs, such as nabumetone and naproxen, have been proposed as less likely to cause MOH, and are even used as bridging therapy sometimes (as long as neither of these was the overused medication).26
The time it takes to develop MOH is shortest with triptans, followed by ergots, then analgesics.27
Prospective cohort studies6,16 have shown that barbiturates and opioids are more likely to induce MOH; for that reason, agents in these analgesic classes are almost universally avoided unless no other medically acceptable options exist. Using barbiturate-containing compounds or opioids > 4 d/mo exponentially increases the likelihood of MOH.
Promising preclinical data demonstrate that the gepant, or small-molecule calcitonin gene-related peptide (CGRP) receptor antagonist, class of medications used as abortive therapy does not induce medication overuse cutaneous allodynia.28
Provide education. Primary prevention of MOH involves (1) increasing patients’ awareness of how to take medications appropriately and (2) restricting intake of over-the-counter abortive medications. Often, the expert recommendation is to limit abortives to approximately 2 d/wk because more frequent use places patients at risk of further increased use and subsequent MOH.
Continue to: A randomized controlled trial in Norway...
A randomized controlled trial in Norway compared outcomes in 2 groups of patients with MOH: One group was given advice on the disorder by a physician; the other group was not provided with advice. In the “business-as-usual” group, there was no significant improvement; however, when general practitioners provided simple advice (lasting roughly 9 minutes) about reducing abortive medication use to a safe level and cautioned patients that they would be “feeling worse before feeling better,” headache days were reduced by approximately 8 per month and medication days, by 16 per month.2
A subsequent, long-term follow-up study29 of patients from the Norway trial2 who had been given advice and education showed a relapse rate (ie, into overuse of headache medication) of only 8% and sustained reduction of headache days and medication use at 16 months.
Offer support and other nondrug interventions. A recent review of 3 studies23 recommended that extra support for patients from a headache nurse, close follow-up, keeping an electronic diary that provides feedback, and undertaking a short course of psychotherapy can reduce medication overuse and prevent relapse.
If MOH develops, initiate withdrawal, introduce a preventive
Withdraw overused medication. Most current evidence suggests that withdrawal of the offending agent is the most effective factor in reducing headache days and improving quality of life. A randomized controlled trial compared the effects of (1) complete and immediate withdrawal of an abortive medication with (2) reducing its use (ie, limiting intake to 2 d/wk), on headache frequency, disability, and quality of life.30 There was a reduction of headache days in both groups; however, reduction was much greater at 2 months in the complete withdrawal group than in the restricted intake group (respectively, a 41% and a 26% reduction in headache days per month). This effect was sustained at 6 and 12 months in both groups. The study confirmed the results of earlier research2,15: Abrupt withdrawal leads to reversion to an episodic pattern at 2 to 6 months in approximately 40% to 60% of patients.
More studies are needed to determine the most appropriate treatment course for MOH; however, complete withdrawal of the causative drug is the most important intervention.
Continue to: Consider withdrawal plus preventive treatment
Consider withdrawal plus preventive treatment. Use of sodium valproate, in addition to medication overuse detoxification, led to a significant reduction in headache days and improvement in quality of life at 12 weeks but no difference after 24 weeks, compared with detoxification alone in a randomized, double-blind, placebo-controlled study.31
A study of 61 patients showed a larger reduction (by 7.2 d/mo) in headache frequency with any preventive medication in addition to medication withdrawal, compared to withdrawal alone (by 4.1 d/mo) after 3 months; however, the relative benefit was gone at 6 months.32
A study of 98 patients compared immediate and delayed initiation of preventive medication upon withdrawal of overused abortive medication.33 Response was defined as a > 50% reduction in headache frequency and was similar in both groups; results showed a 28% response with immediate initiation of a preventive; a 23% response with delayed (ie, 2 months after withdrawal) initiation; and a 48% response in both groups at 12 months.
Collectively, these studies suggest that adding a preventive medication at the time of withdrawal has the potential to reduce headache frequency more quickly than withdrawal alone. However, after 3 to 6 months, the outcome of reduced headache frequency is the same whether or not a preventive medication is used—as long as the offending agent has been withdrawn.
Do preventives work without withdrawing overused medication? Patients with MOH often show little or no improvement with addition of a preventive medication only; their response to a preventive improves after withdrawal of the overused medication. Patients without previous headache improvement after addition of a preventive, who also did not improve 2 months after withdrawal, then demonstrated an overall reduction in headache by 26% when a preventive was reintroduced after withdrawal.2
Continue to: The research evidence for preventives
The research evidence for preventives. Medications for headache prevention have not been extensively evaluated specifically for treating MOH. Here is what’s known:
- Flunarizine, amitriptyline, and beta-blockers usually are ineffective for MOH.24
- Results for topiramate are mixed: A small, double-blind, placebo-controlled chronic migraine study in Europe showed that, in a subgroup of patients with MOH, topiramate led to a small but significant reduction (3.5 d/mo) in headache frequency, compared to placebo.27 A similar study done in the United States did not show a significant difference between the active-treatment and placebo groups.34
- Findings regarding onabotulinumtoxinA are intriguing: In a posthoc analysis of onabotulinumtoxinA to treat chronic migraine, patients with MOH who did not undergo detoxification had an 8 d/mo greater reduction in headache, compared to placebo.35 However, when compared to placebo in conjunction with detoxification, onabotulinumtoxinA demonstrated no benefit.36
- Newer CGRP antagonist and CGRP receptor antagonist monoclonal antibodies are successful preventive medications that have demonstrated a reduction in acute medication use days per month and headache days per month37; these compounds have not been compared to withdrawal alone.
Reducing the severity and duration of withdrawal symptoms
Withdrawal from overused abortive headache medications can lead to worsening headache, nausea, vomiting, hypotension, tachycardia, sleep disturbances, restlessness, anxiety, and nervousness. Symptoms usually last 2 to 10 days but can persist for as long as 4 weeks; duration of withdrawal symptoms varies with the medication that is being overused. In patients who have used a triptan, for example, mean duration of withdrawal is 4.1 days; ergotamine, 6.7 days; and NSAIDs, 9.5 days.23 Tapered withdrawal is sometimes recommended with opioids and barbiturates to reduce withdrawal symptoms. It is unclear whether starting a preventive medication during withdrawal assists in reducing withdrawal symptoms.38
Bridging therapy to reduce symptoms of withdrawal is often provided despite debatable utility. Available evidence does not favor one agent or method but suggests some strategies that could be helpful:
- A prednisone taper has a potential role during the first 6 days of withdrawal by reducing rebound headache and withdrawal symptoms39; however, oral prednisolone has been shown to have no benefit.40
- Alone, IV methylprednisolone seems not to be of benefit; however, in a retrospective study of 94 patients, IV methylprednisolone plus diazepam for 5 days led to a significant reduction in headache frequency and drug consumption that was sustained after 3 months.41
- Celecoxib was compared to prednisone over a 20-day course: a celecoxib dosage of 400 mg/d for the first 5 days, tapered by 100 mg every 5 days, and an oral prednisone dosage of 75 mg/d for the first 5 days, then tapered every 5 days. Patients taking celecoxib had lower headache intensity but there was no difference in headache frequency and acute medication intake between the groups.42
Other strategies. Using antiemetics and NSAIDs to reduce withdrawal symptoms is widely practiced, but no placebo-controlled trials have been conducted to support this strategy.
Patients in withdrawal might be more likely to benefit from inpatient care if they have a severe comorbidity, such as opioid or barbiturate use; failure to respond to, tolerate, or adhere to treatment; or relapse after withdrawal.38
Continue to: Cognitive behavioral therapy...
Cognitive behavioral therapy, exercise, a headache diary, and biofeedback should be considered in every patient’s treatment strategy because a multidisciplinary approach increases adherence and leads to improvement in headache frequency and a decrease in disability and medication use.43
Predictors of Tx success
A prospective cohort study determined that the rate of MOH relapse is 31% at 6 months, 41% at 1 year, and 45% at 4 years, with the highest risk of relapse during the first year.44 Looking at the correlation between type of medication overused and relapse rate, the research indicates that
- triptans have the lowest risk of relapse,44
- simple analgesics have a higher risk of relapse than triptans,22,44 and
- opioids have the highest risk of relapse.22
Where the data don’t agree. Data on combination analgesics and on ergots are conflicting.22 In addition, data on whether the primary type of headache predicts relapse rate conflict; however, migraine might predict a better outcome than tension-type headache.22
To recap and expand: Management pearls
The major goals of headache management generally are to rule out secondary headache, reach a correct diagnosis, reduce overall headache frequency, and provide effective abortive medication. A large component of reducing headache frequency is addressing and treating medication overuse.
Seek to understand the nature of the patient’s headache disorder. Components of the history are key in identifying the underlying headache diagnosis and ruling out other, more concerning secondary headache diagnoses. The ICHD-3 is an excellent resource for treating headache disorders because the classification lists specific diagnostic criteria for all recognized headache diagnoses.
Continue to: Medication withdrawal...
Medication withdrawal—with or without preventive medication—should reduce the frequency of MOH in 2 or 3 months. If headache does not become less frequent, however, the headache diagnosis might need to be reconsidered. Minimizing the use of abortive medication is generally recommended, but reduction or withdrawal of these medications does not guarantee that patients will revert to an episodic pattern of headache.
Treating withdrawal symptoms is a reasonable approach in some patients, but evidence does not support routinely providing bridging therapy.
Apply preventives carefully. Abortive medication withdrawal should generally be completed before initiating preventive medication; however, over the short term, starting preventive therapy while withdrawing the overused medication could assist in reducing headache frequency rapidly. This strategy can put patients at risk of medication adverse effects and using the medications longer than necessary, yet might be reasonable in certain patients, given their comorbidities, risk of relapse, and physician and patient preference. A preventive medication for an individual patient should generally be chosen in line with recommendations of the American Academy of Neurology45 and on the basis of the history and comorbidities.
Provide education, which is essential to lowering barriers to success. Patients with MOH must be counseled to understand that (1) a headache treatment that is supposed to be making them feel better is, in fact, making them feel worse and (2) they will get worse before they get better. Many patients are afraid to be without medication to use as needed. It is helpful to educate them on the different types of treatments (abortive, preventive); how MOH interferes with headache prophylaxis and medication efficacy; how MOH alters brain function (ie, aforementioned physiologic changes in pain processing and functional imaging changes23); and that such change is reversible when medication is withdrawn.
ACKNOWLEDGEMENT
The author thanks Jeffrey Curtis, MD, MPH, for his support and editing assistance with the manuscript.
CORRESPONDENCE
Allison Crain, MD, 2927 N 7th Avenue, Phoenix, AZ 85013; [email protected].
Medication overuse headache (MOH), a secondary headache diagnosis, is a prevalent phenomenon that complicates headache diagnosis and treatment, increases the cost of care, and reduces quality of life. Effective abortive medication is essential for the headache sufferer; when an abortive is used too frequently, however, headache frequency increases—potentially beginning a cycle in which the patient then takes more medication to abort the headache. Over time, the patient suffers from an ever-increasing number of headaches, takes even more abortive medication, and so on. In the presence of MOH, there is a reduction in pain response to preventive and abortive treatments; when medication overuse is eliminated, pain response improves.1
Although MOH is well recognized among headache specialists, the condition is often overlooked in primary care. Since headache is a top complaint in primary care, however, and prevention is a major goal in family medicine, the opportunity for you to recognize, treat, and prevent MOH is significant. In fact, a randomized controlled trial showed that brief patient education about headache care and MOH provided by a primary care physician can lead to a significant reduction in headache frequency among patients with MOH.2
This article reviews the recognition and diagnosis of MOH, based on historical features and current criteria; addresses risk factors for abortive medication overuse and how to withdraw an offending agent; and explores the value of bridging and preventive therapies to reduce the overall frequency of headache.

What defines MOH?
Typically, MOH is a chronification of a primary headache disorder. However, in patients with a history of migraine who are undergoing treatment for another chronic pain condition with an opioid or other analgesic, MOH can be induced.3 An increase in the frequency of headache raises the specter of a concomitant increase in the level of disability4; psychiatric comorbidity5; and more headache days, with time lost from school and work.
The Migraine Disability Assessment (MIDAS) questionnaire, a validated instrument that helps the provider (1) measure the impact that headache has on a patient’s life and (2) follow treatment progress, also provides information to employers and insurance companies on treatment coverage and the need for work modification. The MIDAS score is 3 times higher in patients with MOH than in patients with episodic migraine.6,7
The annual associated cost per person of MOH has been estimated at $4000, resulting in billions of dollars in associated costs8; most of these costs are related to absenteeism and disability. After detoxification for MOH, annual outpatient medication costs are reduced by approximately 24%.9
Efforts to solve a common problem create another
Headache affects nearly 50% of the general population worldwide,10 accounting for about 4% of primary care visits11 and approximately 20% of outpatient neurology consultations.12 Although inpatient stays for headache are approximately half the duration of the overall average hospital stay, headache accounts for 3% of admissions.13 According to the Global Burden of Disease study, tension-type headache, migraine, and MOH are the 3 most common headache disorders.10 Headache is the second leading cause of disability among people 15 to 49 years of age.10
Continue to: The prevalence of MOH...
The prevalence of MOH in the general population is 2%.7,14,15 A population-based study showed that the rate of progression from episodic headache (< 15 d/mo) to chronic headache (≥ 15 d/mo) in the general population is 2.5% per year16; however, progression to chronic headache is 14% per year in patients with medication overuse. One-third of the general population with chronic migraine overuses symptomatic medication; in US headache clinics, roughly one-half of patients with chronic headache overuse acute medication.6
Definitions and diagnosis
MOH is a secondary headache diagnosis in the third edition of the International Classification of Headache Disorders (ICHD-3) (TABLE 1),17 which lists diagnostic criteria for recognized headache disorders.

Terminology. MOH has also been called rebound headache, drug-induced headache, and transformed migraine, but these terms are outdated and are not formal diagnoses. Patients sometimes refer to substance-withdrawal headaches (not discussed in this article) as rebound headaches, so clarity is important when discussing headache with patients: namely, that MOH is an exacerbation of an existing headache condition caused by overuse of abortive headache medications, including analgesics, combination analgesics, triptans, barbiturates, and opioids.
MOH was recognized in the early 1950s and fully differentiated as a diagnosis in 2005 in the second edition of the ICHD. The disorder is subcategorized by offending abortive agent (TABLE 217) because the frequency of analgesic use required to develop MOH differs by agent.

Risk factors for MOH and chronification of a primary headache disorder. There are several risk factors for developing MOH, and others that contribute to increasing headache frequency in general (TABLE 35,14,18-23). Some risk factors are common to each. All are important to address because some are modifiable.

Continue to: Pathophysiology
Pathophysiology. The pathophysiology and psychology behind MOH are largely unknown. Physiologic changes in pain processing and functional imaging changes have been demonstrated in patients with MOH, both of which are reversible upon withdrawal of medication.23 Genetic factors and changes in hormone and neurotransmitter levels are found in MOH patients; this is not the case in patients who have an episodic headache pattern only.24
Presentation. Diagnostic criteria for MOH do not include clinical characteristics. Typically, the phenotype of MOH in a given patient is similar to the underlying primary headache25—although this principle can be complicated to tease out because these medications can suppress some symptoms. Diagnosis of a primary headache disorder should be documented along with the diagnosis of MOH.
Medication overuse can exist without MOH: Not every patient who frequently uses an abortive medication develops MOH.
Treatment is multifaceted—and can become complex
Mainstays of treatment of MOH are education about the disorder and detoxification from the overused agent, although specific treatments can differ depending on the agent involved, the frequency and duration of its use, and a patient’s behavioral patterns and psychiatric comorbidities. Often, a daily medication to prevent headache is considered upon, or after, withdrawal of the offending agent. The timing of introducing a preventive might impact its effectiveness. Some refractory cases require more intensive therapy, including hospitalization at a specialized tertiary center.
But before we look at detoxification from an overused agent, it’s important to review one of the best strategies of all in combatting MOH.
Continue to: First and best strategy
First and best strategy: Avoid onset of MOH
Select an appropriate abortive to reduce the risk of MOH. With regard to specific acute headache medications, some nuances other than type of headache should be considered. Nonsteroidal anti-inflammatory drugs (NSAIDs) are recommended as abortive therapy by the American Headache Society for their efficacy, favorable adverse effect profile, and low cost. NSAIDs are protective against development of MOH if a patient’s baseline headache frequency is < 10/mo; at a frequency of 10 to 14 d/mo, however, the risk of MOH increases when using an NSAID.6 A similar effect has been seen with triptans.16 Longer-acting NSAIDs, such as nabumetone and naproxen, have been proposed as less likely to cause MOH, and are even used as bridging therapy sometimes (as long as neither of these was the overused medication).26
The time it takes to develop MOH is shortest with triptans, followed by ergots, then analgesics.27
Prospective cohort studies6,16 have shown that barbiturates and opioids are more likely to induce MOH; for that reason, agents in these analgesic classes are almost universally avoided unless no other medically acceptable options exist. Using barbiturate-containing compounds or opioids > 4 d/mo exponentially increases the likelihood of MOH.
Promising preclinical data demonstrate that the gepant, or small-molecule calcitonin gene-related peptide (CGRP) receptor antagonist, class of medications used as abortive therapy does not induce medication overuse cutaneous allodynia.28
Provide education. Primary prevention of MOH involves (1) increasing patients’ awareness of how to take medications appropriately and (2) restricting intake of over-the-counter abortive medications. Often, the expert recommendation is to limit abortives to approximately 2 d/wk because more frequent use places patients at risk of further increased use and subsequent MOH.
Continue to: A randomized controlled trial in Norway...
A randomized controlled trial in Norway compared outcomes in 2 groups of patients with MOH: One group was given advice on the disorder by a physician; the other group was not provided with advice. In the “business-as-usual” group, there was no significant improvement; however, when general practitioners provided simple advice (lasting roughly 9 minutes) about reducing abortive medication use to a safe level and cautioned patients that they would be “feeling worse before feeling better,” headache days were reduced by approximately 8 per month and medication days, by 16 per month.2
A subsequent, long-term follow-up study29 of patients from the Norway trial2 who had been given advice and education showed a relapse rate (ie, into overuse of headache medication) of only 8% and sustained reduction of headache days and medication use at 16 months.
Offer support and other nondrug interventions. A recent review of 3 studies23 recommended that extra support for patients from a headache nurse, close follow-up, keeping an electronic diary that provides feedback, and undertaking a short course of psychotherapy can reduce medication overuse and prevent relapse.
If MOH develops, initiate withdrawal, introduce a preventive
Withdraw overused medication. Most current evidence suggests that withdrawal of the offending agent is the most effective factor in reducing headache days and improving quality of life. A randomized controlled trial compared the effects of (1) complete and immediate withdrawal of an abortive medication with (2) reducing its use (ie, limiting intake to 2 d/wk), on headache frequency, disability, and quality of life.30 There was a reduction of headache days in both groups; however, reduction was much greater at 2 months in the complete withdrawal group than in the restricted intake group (respectively, a 41% and a 26% reduction in headache days per month). This effect was sustained at 6 and 12 months in both groups. The study confirmed the results of earlier research2,15: Abrupt withdrawal leads to reversion to an episodic pattern at 2 to 6 months in approximately 40% to 60% of patients.
More studies are needed to determine the most appropriate treatment course for MOH; however, complete withdrawal of the causative drug is the most important intervention.
Continue to: Consider withdrawal plus preventive treatment
Consider withdrawal plus preventive treatment. Use of sodium valproate, in addition to medication overuse detoxification, led to a significant reduction in headache days and improvement in quality of life at 12 weeks but no difference after 24 weeks, compared with detoxification alone in a randomized, double-blind, placebo-controlled study.31
A study of 61 patients showed a larger reduction (by 7.2 d/mo) in headache frequency with any preventive medication in addition to medication withdrawal, compared to withdrawal alone (by 4.1 d/mo) after 3 months; however, the relative benefit was gone at 6 months.32
A study of 98 patients compared immediate and delayed initiation of preventive medication upon withdrawal of overused abortive medication.33 Response was defined as a > 50% reduction in headache frequency and was similar in both groups; results showed a 28% response with immediate initiation of a preventive; a 23% response with delayed (ie, 2 months after withdrawal) initiation; and a 48% response in both groups at 12 months.
Collectively, these studies suggest that adding a preventive medication at the time of withdrawal has the potential to reduce headache frequency more quickly than withdrawal alone. However, after 3 to 6 months, the outcome of reduced headache frequency is the same whether or not a preventive medication is used—as long as the offending agent has been withdrawn.
Do preventives work without withdrawing overused medication? Patients with MOH often show little or no improvement with addition of a preventive medication only; their response to a preventive improves after withdrawal of the overused medication. Patients without previous headache improvement after addition of a preventive, who also did not improve 2 months after withdrawal, then demonstrated an overall reduction in headache by 26% when a preventive was reintroduced after withdrawal.2
Continue to: The research evidence for preventives
The research evidence for preventives. Medications for headache prevention have not been extensively evaluated specifically for treating MOH. Here is what’s known:
- Flunarizine, amitriptyline, and beta-blockers usually are ineffective for MOH.24
- Results for topiramate are mixed: A small, double-blind, placebo-controlled chronic migraine study in Europe showed that, in a subgroup of patients with MOH, topiramate led to a small but significant reduction (3.5 d/mo) in headache frequency, compared to placebo.27 A similar study done in the United States did not show a significant difference between the active-treatment and placebo groups.34
- Findings regarding onabotulinumtoxinA are intriguing: In a posthoc analysis of onabotulinumtoxinA to treat chronic migraine, patients with MOH who did not undergo detoxification had an 8 d/mo greater reduction in headache, compared to placebo.35 However, when compared to placebo in conjunction with detoxification, onabotulinumtoxinA demonstrated no benefit.36
- Newer CGRP antagonist and CGRP receptor antagonist monoclonal antibodies are successful preventive medications that have demonstrated a reduction in acute medication use days per month and headache days per month37; these compounds have not been compared to withdrawal alone.
Reducing the severity and duration of withdrawal symptoms
Withdrawal from overused abortive headache medications can lead to worsening headache, nausea, vomiting, hypotension, tachycardia, sleep disturbances, restlessness, anxiety, and nervousness. Symptoms usually last 2 to 10 days but can persist for as long as 4 weeks; duration of withdrawal symptoms varies with the medication that is being overused. In patients who have used a triptan, for example, mean duration of withdrawal is 4.1 days; ergotamine, 6.7 days; and NSAIDs, 9.5 days.23 Tapered withdrawal is sometimes recommended with opioids and barbiturates to reduce withdrawal symptoms. It is unclear whether starting a preventive medication during withdrawal assists in reducing withdrawal symptoms.38
Bridging therapy to reduce symptoms of withdrawal is often provided despite debatable utility. Available evidence does not favor one agent or method but suggests some strategies that could be helpful:
- A prednisone taper has a potential role during the first 6 days of withdrawal by reducing rebound headache and withdrawal symptoms39; however, oral prednisolone has been shown to have no benefit.40
- Alone, IV methylprednisolone seems not to be of benefit; however, in a retrospective study of 94 patients, IV methylprednisolone plus diazepam for 5 days led to a significant reduction in headache frequency and drug consumption that was sustained after 3 months.41
- Celecoxib was compared to prednisone over a 20-day course: a celecoxib dosage of 400 mg/d for the first 5 days, tapered by 100 mg every 5 days, and an oral prednisone dosage of 75 mg/d for the first 5 days, then tapered every 5 days. Patients taking celecoxib had lower headache intensity but there was no difference in headache frequency and acute medication intake between the groups.42
Other strategies. Using antiemetics and NSAIDs to reduce withdrawal symptoms is widely practiced, but no placebo-controlled trials have been conducted to support this strategy.
Patients in withdrawal might be more likely to benefit from inpatient care if they have a severe comorbidity, such as opioid or barbiturate use; failure to respond to, tolerate, or adhere to treatment; or relapse after withdrawal.38
Continue to: Cognitive behavioral therapy...
Cognitive behavioral therapy, exercise, a headache diary, and biofeedback should be considered in every patient’s treatment strategy because a multidisciplinary approach increases adherence and leads to improvement in headache frequency and a decrease in disability and medication use.43
Predictors of Tx success
A prospective cohort study determined that the rate of MOH relapse is 31% at 6 months, 41% at 1 year, and 45% at 4 years, with the highest risk of relapse during the first year.44 Looking at the correlation between type of medication overused and relapse rate, the research indicates that
- triptans have the lowest risk of relapse,44
- simple analgesics have a higher risk of relapse than triptans,22,44 and
- opioids have the highest risk of relapse.22
Where the data don’t agree. Data on combination analgesics and on ergots are conflicting.22 In addition, data on whether the primary type of headache predicts relapse rate conflict; however, migraine might predict a better outcome than tension-type headache.22
To recap and expand: Management pearls
The major goals of headache management generally are to rule out secondary headache, reach a correct diagnosis, reduce overall headache frequency, and provide effective abortive medication. A large component of reducing headache frequency is addressing and treating medication overuse.
Seek to understand the nature of the patient’s headache disorder. Components of the history are key in identifying the underlying headache diagnosis and ruling out other, more concerning secondary headache diagnoses. The ICHD-3 is an excellent resource for treating headache disorders because the classification lists specific diagnostic criteria for all recognized headache diagnoses.
Continue to: Medication withdrawal...
Medication withdrawal—with or without preventive medication—should reduce the frequency of MOH in 2 or 3 months. If headache does not become less frequent, however, the headache diagnosis might need to be reconsidered. Minimizing the use of abortive medication is generally recommended, but reduction or withdrawal of these medications does not guarantee that patients will revert to an episodic pattern of headache.
Treating withdrawal symptoms is a reasonable approach in some patients, but evidence does not support routinely providing bridging therapy.
Apply preventives carefully. Abortive medication withdrawal should generally be completed before initiating preventive medication; however, over the short term, starting preventive therapy while withdrawing the overused medication could assist in reducing headache frequency rapidly. This strategy can put patients at risk of medication adverse effects and using the medications longer than necessary, yet might be reasonable in certain patients, given their comorbidities, risk of relapse, and physician and patient preference. A preventive medication for an individual patient should generally be chosen in line with recommendations of the American Academy of Neurology45 and on the basis of the history and comorbidities.
Provide education, which is essential to lowering barriers to success. Patients with MOH must be counseled to understand that (1) a headache treatment that is supposed to be making them feel better is, in fact, making them feel worse and (2) they will get worse before they get better. Many patients are afraid to be without medication to use as needed. It is helpful to educate them on the different types of treatments (abortive, preventive); how MOH interferes with headache prophylaxis and medication efficacy; how MOH alters brain function (ie, aforementioned physiologic changes in pain processing and functional imaging changes23); and that such change is reversible when medication is withdrawn.
ACKNOWLEDGEMENT
The author thanks Jeffrey Curtis, MD, MPH, for his support and editing assistance with the manuscript.
CORRESPONDENCE
Allison Crain, MD, 2927 N 7th Avenue, Phoenix, AZ 85013; [email protected].
1. Zeeberg P, Olesen J, Jensen R. Discontinuation of medication overuse in headache patients: recovery of therapeutic responsiveness. Cephalalgia. 2006;26:1192-1198.
2. Kristoffersen ES, Straand J, Vetvik KG, et al. Brief intervention for medication-overuse headache in primary care. The BIMOH study: a double-blind pragmatic cluster randomised parallel controlled trial. J Neurol Neurosurg Psychiatry. 2015;86:505-512.
3. Bahra A, Walsh M, Menon S, et al. Does chronic daily headache arise de novo in association with regular use of analgesics? Headache. 2003;43:179-190.
4. Blumenfeld AM, Varon SF, Wilcox TK, et al. Disability, HRQoL and resource use among chronic and episodic migraineurs: results from the International Burden of Migraine Study (IBMS) Cephalalgia. 2011;31:301-315.
5. Chu H-T, Liang C-S, Lee J-T, et al. Associations between depression/anxiety and headache frequency in migraineurs: a cross-sectional study. Headache. 2018;58:407-415.
6. Bigal ME, Lipton RB. Excessive acute migraine medication use and migraine progression. Neurology. 2008;71:1821-1828.
7. Colás R, Muñoz P, Temprano R, et al. Chronic daily headache with analgesic overuse: epidemiology and impact on quality of life. Neurology. 2004;62:1338-1342.
8. Linde M, Gustavsson A, Stovner LJ, et al. The cost of headache disorders in Europe: the Eurolight project. Eur J Neurol. 2012;19:703-711.
9. Shah AM, Bendtsen L, Zeeberg P, et al. Reduction of medication costs after detoxification for medication-overuse headache. Headache. 2013;53:665-672.
10. . Global, regional, and national burden of migraine and tension-type headache, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018;17:954-976.
11. Kernick D, Stapley S, Goadsby PJ, et al. What happens to new-onset headache presenting to primary care? A case–cohort study using electronic primary care records. Cephalalgia. 2008;28:1188-1195.
12. Stone J, Carson A, Duncan R, et al. Who is referred to neurology clinics?—the diagnoses made in 3781 new patients. Clin Neurol Neurosurg. 2010;112:747-751.
13. Munoz-Ceron J, Marin-Careaga V, L, et al. Headache at the emergency room: etiologies, diagnostic usefulness of the ICHD 3 criteria, red and green flags. PloS One. 2019;14:e0208728.
14. Evers S, Marziniak M. Clinical features, pathophysiology, and treatment of medication-overuse headache. Lancet Neurol. 2010;9:391-401.
15. Tassorelli C, Jensen R, Allena M, et al; the . A consensus protocol for the management of medication-overuse headache: evaluation in a multicentric, multinational study. Cephalalgia. 2014;34:645-655.
16. Bigal ME, Serrano D, Buse D, et al. Acute migraine medications and evolution from episodic to chronic migraine: a longitudinal population-based study. Headache. 2008;48:1157-1168.
17. Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211.
18. Ferrari A, Leone S, Vergoni AV, et al. Similarities and differences between chronic migraine and episodic migraine. Headache. 2007;47:65-72.
19. Hagen K, Linde M, Steiner TJ, et al. Risk factors for medication-overuse headache: an 11-year follow-up study. The Nord-Trøndelag Health Studies. Pain. 2012;153:56-61.
20. Katsarava Z, Schneewiess S, Kurth T, et al. Incidence and predictors for chronicity of headache in patients with episodic migraine. Neurology. 2004;62:788-790.
21. Lipton RB, Fanning KM, Buse DC, et al. Migraine progression in subgroups of migraine based on comorbidities: results of the CaMEO study. Neurology. 2019;93:e2224-e2236.
22. Munksgaard SB, Madsen SK, Wienecke T. Treatment of medication overuse headache—a review. Acta Neurol Scand. 2019;139:405-414.
23. Ferraro S, Grazzi L, Mandelli M, et al. Pain processing in medication overuse headache: a functional magnetic resonance imaging (fMRI) study. Pain Med. 2012;13:255-262.
24. Diener H-C, Holle D, Solbach K, et al. Medication-overuse headache: risk factors, pathophysiology and management. Nat Rev Neurol. 2016;12:575-583.
25. Limmroth V, Katsarava Z, Fritsche G, et al. Features of medication overuse headache following overuse of different acute headache drugs. Neurology. 2002;59:1011-1014.
26. Mauskop A, ed. Migraine and Headache. 2nd ed. Oxford University Press; 2013.
27. Diener H-C, Bussone G, Van Oene JC, et al; . Topiramate reduces headache days in chronic migraine: a randomized, double-blind, placebo-controlled study. Cephalalgia. 2007;27:814-823.
28. Navratilova E, Behravesh S, Oyarzo J, et al. Ubrogepant does not induce latent sensitization in a preclinical model of medication overuse headache Cephalalgia. 2020;40:892-902.
29. Kristoffersen ES, Straand J, Russell MB, et al. Lasting improvement of medication-overuse headache after brief intervention—a long-term follow-up in primary care. Eur J Neurol. 2017;24:883-891.
30. Carlsen LN, Munksgaard SB, Jensen RH, et al. Complete detoxification is the most effective treatment of medication-overuse headache: a randomized controlled open-label trial. Cephalalgia. 2018;38:225-236.
31. Sarchielli P, Messina P, Cupini LM, et al; SAMOHA Study Group. Sodium valproate in migraine without aura and medication overuse headache: a randomized controlled trial. Eur Neuropsychopharmacol. 2014;24:1289-1297.
32. Hagen K, Stovner LJ. A randomized controlled trial on medication-overuse headache: outcome after 1 and 4 years. Acta Neurol Scand Suppl. 2011;124(suppl 191):38-43.
33. Munksgaard SB, Bendtsen L, Jensen RH. Detoxification of medication-overuse headache by a multidisciplinary treatment programme is highly effective: a comparison of two consecutive treatment methods in an open-label design. Cephalalgia. 2012;32:834-844.
34. Silberstein S, Lipton R, Dodick D, et al. Topiramate treatment of chronic migraine: a randomized, placebo-controlled trial of quality of life and other efficacy measures. Headache. 2009;49:1153-1162.
35. Silberstein SD, Blumenfeld AM, Cady RK, et al. OnabotulinumtoxinA for treatment of chronic migraine: PREEMPT 24-week pooled subgroup analysis of patients who had acute headache medication overuse at baseline. J Neurol Sci. 2013;331:48-56.
36. Sandrini G, Perrotta A, Tassorelli C, et al. Botulinum toxin type-A in the prophylactic treatment of medication-overuse headache: a multicenter, double-blind, randomized, placebo-controlled, parallel group study. J Headache Pain. 2011;12:427-433.
37. Tepper SJ. CGRP and headache: a brief review. Neurol Sci. 2019;40(suppl 1):99-105.
38. Diener H-C, Dodick D, Evers S, et al. Pathophysiology, prevention and treatment of medication overuse headache. Lancet Neurol. 2019;18:891-902.
39. Krymchantowski AV, Barbosa JS. Prednisone as initial treatment of analgesic-induced daily headache. Cephalalgia. 2000;20:107-113.
40. Bøe MG, Mygland A, Salvesen R. Prednisolone does not reduce withdrawal headache: a randomized, double-blind study. Neurology. 2007;69:26-31.
41. Paolucci M, Altamura C, Brunelli N, et al. Methylprednisolone plus diazepam i.v. as bridge therapy for medication overuse headache. Neurol Sci. 2017;38:2025-2029.
42. Taghdiri F, Togha M, Razeghi Jahromi S, et al. Celecoxib vs prednisone for the treatment of withdrawal headache in patients with medication overuse headache: a randomized, double-blind clinical trial. Headache. 2015;55:128-135.
43. Ramsey RR, Ryan JL, Hershey AD, et al. Treatment adherence in patients with headache: a systematic review. Headache. 2014;54:795-816.
44. Katsarava Z, Muessig M, Dzagnidze A, et al. Medication overuse headache: rates and predictors for relapse in a 4-year prospective study. Cephalalgia. 2005;25:12-15.
45. Silberstein SD, Holland S, Freitag F, et al; . Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012; 78:1137-1145.
1. Zeeberg P, Olesen J, Jensen R. Discontinuation of medication overuse in headache patients: recovery of therapeutic responsiveness. Cephalalgia. 2006;26:1192-1198.
2. Kristoffersen ES, Straand J, Vetvik KG, et al. Brief intervention for medication-overuse headache in primary care. The BIMOH study: a double-blind pragmatic cluster randomised parallel controlled trial. J Neurol Neurosurg Psychiatry. 2015;86:505-512.
3. Bahra A, Walsh M, Menon S, et al. Does chronic daily headache arise de novo in association with regular use of analgesics? Headache. 2003;43:179-190.
4. Blumenfeld AM, Varon SF, Wilcox TK, et al. Disability, HRQoL and resource use among chronic and episodic migraineurs: results from the International Burden of Migraine Study (IBMS) Cephalalgia. 2011;31:301-315.
5. Chu H-T, Liang C-S, Lee J-T, et al. Associations between depression/anxiety and headache frequency in migraineurs: a cross-sectional study. Headache. 2018;58:407-415.
6. Bigal ME, Lipton RB. Excessive acute migraine medication use and migraine progression. Neurology. 2008;71:1821-1828.
7. Colás R, Muñoz P, Temprano R, et al. Chronic daily headache with analgesic overuse: epidemiology and impact on quality of life. Neurology. 2004;62:1338-1342.
8. Linde M, Gustavsson A, Stovner LJ, et al. The cost of headache disorders in Europe: the Eurolight project. Eur J Neurol. 2012;19:703-711.
9. Shah AM, Bendtsen L, Zeeberg P, et al. Reduction of medication costs after detoxification for medication-overuse headache. Headache. 2013;53:665-672.
10. . Global, regional, and national burden of migraine and tension-type headache, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018;17:954-976.
11. Kernick D, Stapley S, Goadsby PJ, et al. What happens to new-onset headache presenting to primary care? A case–cohort study using electronic primary care records. Cephalalgia. 2008;28:1188-1195.
12. Stone J, Carson A, Duncan R, et al. Who is referred to neurology clinics?—the diagnoses made in 3781 new patients. Clin Neurol Neurosurg. 2010;112:747-751.
13. Munoz-Ceron J, Marin-Careaga V, L, et al. Headache at the emergency room: etiologies, diagnostic usefulness of the ICHD 3 criteria, red and green flags. PloS One. 2019;14:e0208728.
14. Evers S, Marziniak M. Clinical features, pathophysiology, and treatment of medication-overuse headache. Lancet Neurol. 2010;9:391-401.
15. Tassorelli C, Jensen R, Allena M, et al; the . A consensus protocol for the management of medication-overuse headache: evaluation in a multicentric, multinational study. Cephalalgia. 2014;34:645-655.
16. Bigal ME, Serrano D, Buse D, et al. Acute migraine medications and evolution from episodic to chronic migraine: a longitudinal population-based study. Headache. 2008;48:1157-1168.
17. Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211.
18. Ferrari A, Leone S, Vergoni AV, et al. Similarities and differences between chronic migraine and episodic migraine. Headache. 2007;47:65-72.
19. Hagen K, Linde M, Steiner TJ, et al. Risk factors for medication-overuse headache: an 11-year follow-up study. The Nord-Trøndelag Health Studies. Pain. 2012;153:56-61.
20. Katsarava Z, Schneewiess S, Kurth T, et al. Incidence and predictors for chronicity of headache in patients with episodic migraine. Neurology. 2004;62:788-790.
21. Lipton RB, Fanning KM, Buse DC, et al. Migraine progression in subgroups of migraine based on comorbidities: results of the CaMEO study. Neurology. 2019;93:e2224-e2236.
22. Munksgaard SB, Madsen SK, Wienecke T. Treatment of medication overuse headache—a review. Acta Neurol Scand. 2019;139:405-414.
23. Ferraro S, Grazzi L, Mandelli M, et al. Pain processing in medication overuse headache: a functional magnetic resonance imaging (fMRI) study. Pain Med. 2012;13:255-262.
24. Diener H-C, Holle D, Solbach K, et al. Medication-overuse headache: risk factors, pathophysiology and management. Nat Rev Neurol. 2016;12:575-583.
25. Limmroth V, Katsarava Z, Fritsche G, et al. Features of medication overuse headache following overuse of different acute headache drugs. Neurology. 2002;59:1011-1014.
26. Mauskop A, ed. Migraine and Headache. 2nd ed. Oxford University Press; 2013.
27. Diener H-C, Bussone G, Van Oene JC, et al; . Topiramate reduces headache days in chronic migraine: a randomized, double-blind, placebo-controlled study. Cephalalgia. 2007;27:814-823.
28. Navratilova E, Behravesh S, Oyarzo J, et al. Ubrogepant does not induce latent sensitization in a preclinical model of medication overuse headache Cephalalgia. 2020;40:892-902.
29. Kristoffersen ES, Straand J, Russell MB, et al. Lasting improvement of medication-overuse headache after brief intervention—a long-term follow-up in primary care. Eur J Neurol. 2017;24:883-891.
30. Carlsen LN, Munksgaard SB, Jensen RH, et al. Complete detoxification is the most effective treatment of medication-overuse headache: a randomized controlled open-label trial. Cephalalgia. 2018;38:225-236.
31. Sarchielli P, Messina P, Cupini LM, et al; SAMOHA Study Group. Sodium valproate in migraine without aura and medication overuse headache: a randomized controlled trial. Eur Neuropsychopharmacol. 2014;24:1289-1297.
32. Hagen K, Stovner LJ. A randomized controlled trial on medication-overuse headache: outcome after 1 and 4 years. Acta Neurol Scand Suppl. 2011;124(suppl 191):38-43.
33. Munksgaard SB, Bendtsen L, Jensen RH. Detoxification of medication-overuse headache by a multidisciplinary treatment programme is highly effective: a comparison of two consecutive treatment methods in an open-label design. Cephalalgia. 2012;32:834-844.
34. Silberstein S, Lipton R, Dodick D, et al. Topiramate treatment of chronic migraine: a randomized, placebo-controlled trial of quality of life and other efficacy measures. Headache. 2009;49:1153-1162.
35. Silberstein SD, Blumenfeld AM, Cady RK, et al. OnabotulinumtoxinA for treatment of chronic migraine: PREEMPT 24-week pooled subgroup analysis of patients who had acute headache medication overuse at baseline. J Neurol Sci. 2013;331:48-56.
36. Sandrini G, Perrotta A, Tassorelli C, et al. Botulinum toxin type-A in the prophylactic treatment of medication-overuse headache: a multicenter, double-blind, randomized, placebo-controlled, parallel group study. J Headache Pain. 2011;12:427-433.
37. Tepper SJ. CGRP and headache: a brief review. Neurol Sci. 2019;40(suppl 1):99-105.
38. Diener H-C, Dodick D, Evers S, et al. Pathophysiology, prevention and treatment of medication overuse headache. Lancet Neurol. 2019;18:891-902.
39. Krymchantowski AV, Barbosa JS. Prednisone as initial treatment of analgesic-induced daily headache. Cephalalgia. 2000;20:107-113.
40. Bøe MG, Mygland A, Salvesen R. Prednisolone does not reduce withdrawal headache: a randomized, double-blind study. Neurology. 2007;69:26-31.
41. Paolucci M, Altamura C, Brunelli N, et al. Methylprednisolone plus diazepam i.v. as bridge therapy for medication overuse headache. Neurol Sci. 2017;38:2025-2029.
42. Taghdiri F, Togha M, Razeghi Jahromi S, et al. Celecoxib vs prednisone for the treatment of withdrawal headache in patients with medication overuse headache: a randomized, double-blind clinical trial. Headache. 2015;55:128-135.
43. Ramsey RR, Ryan JL, Hershey AD, et al. Treatment adherence in patients with headache: a systematic review. Headache. 2014;54:795-816.
44. Katsarava Z, Muessig M, Dzagnidze A, et al. Medication overuse headache: rates and predictors for relapse in a 4-year prospective study. Cephalalgia. 2005;25:12-15.
45. Silberstein SD, Holland S, Freitag F, et al; . Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012; 78:1137-1145.
PRACTICE RECOMMENDATIONS
› Avoid prescribing barbiturates or opioids for a headache disorder. A
› Limit use of a headache-abortive medication to twice a week when starting a patient on the drug. C
› Consider providing bridging therapy during detoxification of the overused medication. C
› Do not provide a preventive medication without withdrawing the overused agent. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Give women's mental health a seat at the health care table
Why it’s time for women’s mental health to be recognized as the subspecialty it already is
It wasn’t until I (Dr. Leistikow) finished my psychiatry residency that I realized the training I had received in women’s mental health was unusual. It was simply a required experience for PGY-3 residents at Johns Hopkins University, Baltimore.
All of us, regardless of interest, spent 1 afternoon a week over 6 months caring for patients in a specialty psychiatric clinic for women (run by Dr. Payne and Dr. Osborne). We discussed cases and received didactics on such topics as risk factors for postpartum depression; the risks of untreated mental illness in pregnancy, compared with the risks of various psychiatric medications; how to choose and dose medications for bipolar disorder as blood levels change across pregnancy; which resources to consult to determine the amounts and risks of various medications passed on in breast milk; and how to diagnose and treat premenstrual dysphoric disorder, to name a few lecture subjects.
By the time we were done, all residents had received more than 20 hours of teaching about how to treat mental illness in women across the reproductive life cycle. This was 20 hours more than is currently required by the American College of Graduate Medical Education, the accrediting body for all residencies, including psychiatry.1 It is time for that to change.
Women’s need for psychiatric treatment that addresses reproductive transitions is not new; it is as old as time. Not only do women who previously needed psychiatric treatment continue to need treatment when they get pregnant or are breastfeeding, but it is now well recognized that times of reproductive transition or flux – whether premenstrual, post partum, or perimenopausal – confer increased risk for both new-onset and exacerbations of prior mental illnesses.
What has changed is psychiatry’s ability to finally meet that need. Previously, despite the fact that women make up the majority of patients presenting for treatment, that nearly all women will menstruate and go through menopause, and that more than 80% of American women will have at least one pregnancy during their lifetime,psychiatrists practice as if these reproductive transitions were unfortunate blips getting in the doctor’s way.2 We mostly threw up our hands when our patients became pregnant, reflexively stopped all medications, and expected women to suffer for the sake of their babies.
with a large and growing research base, with both agreed-upon best practices and evolving standards of care informed by and responsive to the scientific literature. We now know that untreated maternal psychiatric illness carries its own risks for infants both before and after delivery; that many maternal pharmacologic treatments are lower risk for infants than previously thought; that protecting and treating women’s mental health in pregnancy has benefits for women, their babies, and the families that depend on them; and that there is now a growing evidence base informing both new and older treatments and enabling women and their doctors to make complex decisions balancing risk and benefit across the life cycle.
Many psychiatrists-in-training are hungry for this knowledge. At last count, in the United States alone, there were 16 women’s mental health fellowships available, up from just 3 in 2008.3 The problem is that none of them are accredited or funded by the ACGME, because reproductive psychiatry (here used interchangeably with the term women’s mental health) has not been officially recognized as a subspecialty. This means that current funding frequently rests on philanthropy, which often cannot be sustained, and clinical billing, which gives fellows in some programs such heavy clinical responsibilities that little time is left for scholarly work. Lack of subspecialty status also blocks numerous important downstream effects that would flow from this recognition.
Reproductive psychiatry clearly already meets criteria laid out by the American Board of Medical Specialties for defining a subspecialty field. As argued elsewhere, it has a distinct patient population with definable care needs and a standalone body of scientific medical knowledge as well as a national (and international) community of experts that has already done much to improve women’s access to care they desperately need.4 It also meets the ACGME’s criteria for a new subspecialty except for approval by the American Board of Psychiatry and Neurology.5 Finally, it also meets the requirements of the ABPN except for having 25 fellowship programs with 50 fellowship positions and 50 trainees per year completing fellowships, a challenging Catch-22 without the necessary funding that would accrue from accreditation.6
Despite growing awareness and demand, there remains a shortage of psychiatrists trained to treat women during times of reproductive transition and to pass their recommendations and knowledge on to their primary care and ob.gyn. colleagues. What official recognition would bring, in addition to funding for fellowships post residency, is a guaranteed seat at the table in psychiatry residencies, in terms of a required number of hours devoted to these topics for trainees, ensuring that all graduating psychiatrists have at least some exposure to the knowledge and practices so material to their patients.
It isn’t enough to wait for residencies to see the writing on the wall and voluntarily carve out a slice of pie devoted to women’s mental health from the limited time and resources available to train residents. A 2017 survey of psychiatry residency program training directors found that 23%, or almost a quarter of programs that responded, offered no reproductive psychiatry training at all, that 49% required 5 hours or less across all 4 years of training, and that 75% of programs had no required clinical exposure to reproductive psychiatry patients.7 Despite the fact that 87% of training directors surveyed agreed either that reproductive psychiatry was “an important area of education” or a subject general residents should be competent in, ACGME-recognized specialties take precedence.
A system so patchy and insufficient won’t do. It’s not good enough for the trainees who frequently have to look outside of their own institutions for the training they know they need. It’s not good enough for the pregnant or postpartum patient looking for evidence-based advice, who is currently left on her own to determine, prior to booking an appointment, whether a specific psychiatrist has received any training relevant to treating her. Adding reproductive psychiatry to the topics a graduating psychiatrist must have some proficiency in also signals to recent graduates and experienced attendings, as well as the relevant examining boards and producers of continuing medical education content, that women’s mental health is no longer a fringe topic but rather foundational to all practicing psychiatrists.
The oil needed to prime this pump is official recognition of the subspecialty that reproductive psychiatry already is. The women’s mental health community is ready. The research base is well established and growing exponentially. The number of women’s mental health fellowships is healthy and would increase significantly with ACGME funding. Psychiatry residency training programs can turn to recent graduates of these fellowships as well as their own faculty with reproductive psychiatry experience to teach trainees. In addition, the National Curriculum in Reproductive Psychiatry, over the last 4 years, has created a repository of free online modules dedicated to facilitating this type of training, with case discussions across numerous topics for use by both educators and trainees. The American Psychiatric Association recently formed the Committee on Women’s Mental Health in 2020 and will be publishing a textbook based on work done by the NCRP within the coming year.
Imagine the changed world that would open to all psychiatrists if reproductive psychiatry were given the credentials it deserves. When writing prescriptions, we would view pregnancy as the potential outcome it is in any woman of reproductive age, given that 50% of pregnancies are unplanned, and let women know ahead of time how to think about possible fetal effects rather than waiting for their panicked phone messages or hearing that they have stopped their medications abruptly. We would work to identify our patient’s individual risk factors for postpartum depression predelivery to reduce that risk and prevent or limit illness. We would plan ahead for close follow-up post partum during the window of greatest risk, rather than expecting women to drop out of care while taking care of their infants or languish on scheduling waiting lists. We would feel confident in giving evidence-based advice to our patients around times of reproductive transition across the life cycle, but especially in pregnancy and lactation, empowering women to make healthy decisions for themselves and their families, no longer abandoning them just when they need us most.
References
1. ACGME Program Requirements for Graduate Medical Education in Psychiatry. Accreditation Counsel for Graduate Medical Education. 2020 Jul 1.
2. Livingston G. “They’re waiting longer, but U.S. women today more likely to have children than a decade ago.” Pew Research Center’s Social & Demographic Trends Project. pewsocialtrends.org. 2018 Jan 18.
3. Nagle-Yang S et al. Acad Psychiatry. 2018 Apr;42(2):202-6.
4. Payne JL. Int Rev Psychiatry. 2019 May;31(3):207-9.
5. Accreditation Council for Graduate Medical Education Policies and Procedures. 2020 Sep 26.
6. American Board of Psychiatry and Neurology. Requirements for Subspecialty Recognition, Attachment A. 2008.
7. Osborne LM et al. Acad Psychiatry. 2018 Apr;42(2):197-201.
Dr. Leistikow is a reproductive psychiatrist and clinical assistant professor in the department of psychiatry at the University of Maryland, Baltimore, where she sees patients and helps train residents and fellows. She is on the education committee of the National Curriculum in Reproductive Psychiatry (NCRPtraining.org) and has written about women’s mental health for textbooks, scientific journals and on her private practice blog at www.womenspsychiatrybaltimore.com. Dr. Leistikow has no conflicts of interest.
Dr. Payne is associate professor of psychiatry and behavioral sciences and director of the Women’s Mood Disorders Center at Johns Hopkins University, Baltimore. In addition to providing outstanding clinical care for women with mood disorders, she conducts research into the genetic, biological, and environmental factors involved in postpartum depression. She and her colleagues have recently identified two epigenetic biomarkers of postpartum depression and are working hard to replicate this work with National Institutes of Health funding. Most recently, she was appointed to the American Psychiatric Association’s committee on women’s mental health and is serving as president-elect for both the Marcé of North America and the International Marcé Perinatal Mental Health Societies. She disclosed the following relevant financial relationships: serve(d) as a director, officer, partner, employee, adviser, consultant, or trustee for Sage Therapeutics and Janssen Pharmaceuticals.
Dr. Osborne is associate professor of psychiatry and behavioral sciences and of gynecology and obstetrics at Johns Hopkins University, where she directs a postdoctoral fellowship program in reproductive psychiatry. She is an expert on the diagnosis and treatment of mood and anxiety disorders during pregnancy, the post partum, the premenstrual period, and perimenopause. Her work is supported by the Brain and Behavior Foundation, the Doris Duke Foundation, the American Board of Psychiatry and Neurology, and the National Institute of Mental Health. She has no conflicts of interest.
Why it’s time for women’s mental health to be recognized as the subspecialty it already is
Why it’s time for women’s mental health to be recognized as the subspecialty it already is
It wasn’t until I (Dr. Leistikow) finished my psychiatry residency that I realized the training I had received in women’s mental health was unusual. It was simply a required experience for PGY-3 residents at Johns Hopkins University, Baltimore.
All of us, regardless of interest, spent 1 afternoon a week over 6 months caring for patients in a specialty psychiatric clinic for women (run by Dr. Payne and Dr. Osborne). We discussed cases and received didactics on such topics as risk factors for postpartum depression; the risks of untreated mental illness in pregnancy, compared with the risks of various psychiatric medications; how to choose and dose medications for bipolar disorder as blood levels change across pregnancy; which resources to consult to determine the amounts and risks of various medications passed on in breast milk; and how to diagnose and treat premenstrual dysphoric disorder, to name a few lecture subjects.
By the time we were done, all residents had received more than 20 hours of teaching about how to treat mental illness in women across the reproductive life cycle. This was 20 hours more than is currently required by the American College of Graduate Medical Education, the accrediting body for all residencies, including psychiatry.1 It is time for that to change.
Women’s need for psychiatric treatment that addresses reproductive transitions is not new; it is as old as time. Not only do women who previously needed psychiatric treatment continue to need treatment when they get pregnant or are breastfeeding, but it is now well recognized that times of reproductive transition or flux – whether premenstrual, post partum, or perimenopausal – confer increased risk for both new-onset and exacerbations of prior mental illnesses.
What has changed is psychiatry’s ability to finally meet that need. Previously, despite the fact that women make up the majority of patients presenting for treatment, that nearly all women will menstruate and go through menopause, and that more than 80% of American women will have at least one pregnancy during their lifetime,psychiatrists practice as if these reproductive transitions were unfortunate blips getting in the doctor’s way.2 We mostly threw up our hands when our patients became pregnant, reflexively stopped all medications, and expected women to suffer for the sake of their babies.
with a large and growing research base, with both agreed-upon best practices and evolving standards of care informed by and responsive to the scientific literature. We now know that untreated maternal psychiatric illness carries its own risks for infants both before and after delivery; that many maternal pharmacologic treatments are lower risk for infants than previously thought; that protecting and treating women’s mental health in pregnancy has benefits for women, their babies, and the families that depend on them; and that there is now a growing evidence base informing both new and older treatments and enabling women and their doctors to make complex decisions balancing risk and benefit across the life cycle.
Many psychiatrists-in-training are hungry for this knowledge. At last count, in the United States alone, there were 16 women’s mental health fellowships available, up from just 3 in 2008.3 The problem is that none of them are accredited or funded by the ACGME, because reproductive psychiatry (here used interchangeably with the term women’s mental health) has not been officially recognized as a subspecialty. This means that current funding frequently rests on philanthropy, which often cannot be sustained, and clinical billing, which gives fellows in some programs such heavy clinical responsibilities that little time is left for scholarly work. Lack of subspecialty status also blocks numerous important downstream effects that would flow from this recognition.
Reproductive psychiatry clearly already meets criteria laid out by the American Board of Medical Specialties for defining a subspecialty field. As argued elsewhere, it has a distinct patient population with definable care needs and a standalone body of scientific medical knowledge as well as a national (and international) community of experts that has already done much to improve women’s access to care they desperately need.4 It also meets the ACGME’s criteria for a new subspecialty except for approval by the American Board of Psychiatry and Neurology.5 Finally, it also meets the requirements of the ABPN except for having 25 fellowship programs with 50 fellowship positions and 50 trainees per year completing fellowships, a challenging Catch-22 without the necessary funding that would accrue from accreditation.6
Despite growing awareness and demand, there remains a shortage of psychiatrists trained to treat women during times of reproductive transition and to pass their recommendations and knowledge on to their primary care and ob.gyn. colleagues. What official recognition would bring, in addition to funding for fellowships post residency, is a guaranteed seat at the table in psychiatry residencies, in terms of a required number of hours devoted to these topics for trainees, ensuring that all graduating psychiatrists have at least some exposure to the knowledge and practices so material to their patients.
It isn’t enough to wait for residencies to see the writing on the wall and voluntarily carve out a slice of pie devoted to women’s mental health from the limited time and resources available to train residents. A 2017 survey of psychiatry residency program training directors found that 23%, or almost a quarter of programs that responded, offered no reproductive psychiatry training at all, that 49% required 5 hours or less across all 4 years of training, and that 75% of programs had no required clinical exposure to reproductive psychiatry patients.7 Despite the fact that 87% of training directors surveyed agreed either that reproductive psychiatry was “an important area of education” or a subject general residents should be competent in, ACGME-recognized specialties take precedence.
A system so patchy and insufficient won’t do. It’s not good enough for the trainees who frequently have to look outside of their own institutions for the training they know they need. It’s not good enough for the pregnant or postpartum patient looking for evidence-based advice, who is currently left on her own to determine, prior to booking an appointment, whether a specific psychiatrist has received any training relevant to treating her. Adding reproductive psychiatry to the topics a graduating psychiatrist must have some proficiency in also signals to recent graduates and experienced attendings, as well as the relevant examining boards and producers of continuing medical education content, that women’s mental health is no longer a fringe topic but rather foundational to all practicing psychiatrists.
The oil needed to prime this pump is official recognition of the subspecialty that reproductive psychiatry already is. The women’s mental health community is ready. The research base is well established and growing exponentially. The number of women’s mental health fellowships is healthy and would increase significantly with ACGME funding. Psychiatry residency training programs can turn to recent graduates of these fellowships as well as their own faculty with reproductive psychiatry experience to teach trainees. In addition, the National Curriculum in Reproductive Psychiatry, over the last 4 years, has created a repository of free online modules dedicated to facilitating this type of training, with case discussions across numerous topics for use by both educators and trainees. The American Psychiatric Association recently formed the Committee on Women’s Mental Health in 2020 and will be publishing a textbook based on work done by the NCRP within the coming year.
Imagine the changed world that would open to all psychiatrists if reproductive psychiatry were given the credentials it deserves. When writing prescriptions, we would view pregnancy as the potential outcome it is in any woman of reproductive age, given that 50% of pregnancies are unplanned, and let women know ahead of time how to think about possible fetal effects rather than waiting for their panicked phone messages or hearing that they have stopped their medications abruptly. We would work to identify our patient’s individual risk factors for postpartum depression predelivery to reduce that risk and prevent or limit illness. We would plan ahead for close follow-up post partum during the window of greatest risk, rather than expecting women to drop out of care while taking care of their infants or languish on scheduling waiting lists. We would feel confident in giving evidence-based advice to our patients around times of reproductive transition across the life cycle, but especially in pregnancy and lactation, empowering women to make healthy decisions for themselves and their families, no longer abandoning them just when they need us most.
References
1. ACGME Program Requirements for Graduate Medical Education in Psychiatry. Accreditation Counsel for Graduate Medical Education. 2020 Jul 1.
2. Livingston G. “They’re waiting longer, but U.S. women today more likely to have children than a decade ago.” Pew Research Center’s Social & Demographic Trends Project. pewsocialtrends.org. 2018 Jan 18.
3. Nagle-Yang S et al. Acad Psychiatry. 2018 Apr;42(2):202-6.
4. Payne JL. Int Rev Psychiatry. 2019 May;31(3):207-9.
5. Accreditation Council for Graduate Medical Education Policies and Procedures. 2020 Sep 26.
6. American Board of Psychiatry and Neurology. Requirements for Subspecialty Recognition, Attachment A. 2008.
7. Osborne LM et al. Acad Psychiatry. 2018 Apr;42(2):197-201.
Dr. Leistikow is a reproductive psychiatrist and clinical assistant professor in the department of psychiatry at the University of Maryland, Baltimore, where she sees patients and helps train residents and fellows. She is on the education committee of the National Curriculum in Reproductive Psychiatry (NCRPtraining.org) and has written about women’s mental health for textbooks, scientific journals and on her private practice blog at www.womenspsychiatrybaltimore.com. Dr. Leistikow has no conflicts of interest.
Dr. Payne is associate professor of psychiatry and behavioral sciences and director of the Women’s Mood Disorders Center at Johns Hopkins University, Baltimore. In addition to providing outstanding clinical care for women with mood disorders, she conducts research into the genetic, biological, and environmental factors involved in postpartum depression. She and her colleagues have recently identified two epigenetic biomarkers of postpartum depression and are working hard to replicate this work with National Institutes of Health funding. Most recently, she was appointed to the American Psychiatric Association’s committee on women’s mental health and is serving as president-elect for both the Marcé of North America and the International Marcé Perinatal Mental Health Societies. She disclosed the following relevant financial relationships: serve(d) as a director, officer, partner, employee, adviser, consultant, or trustee for Sage Therapeutics and Janssen Pharmaceuticals.
Dr. Osborne is associate professor of psychiatry and behavioral sciences and of gynecology and obstetrics at Johns Hopkins University, where she directs a postdoctoral fellowship program in reproductive psychiatry. She is an expert on the diagnosis and treatment of mood and anxiety disorders during pregnancy, the post partum, the premenstrual period, and perimenopause. Her work is supported by the Brain and Behavior Foundation, the Doris Duke Foundation, the American Board of Psychiatry and Neurology, and the National Institute of Mental Health. She has no conflicts of interest.
It wasn’t until I (Dr. Leistikow) finished my psychiatry residency that I realized the training I had received in women’s mental health was unusual. It was simply a required experience for PGY-3 residents at Johns Hopkins University, Baltimore.
All of us, regardless of interest, spent 1 afternoon a week over 6 months caring for patients in a specialty psychiatric clinic for women (run by Dr. Payne and Dr. Osborne). We discussed cases and received didactics on such topics as risk factors for postpartum depression; the risks of untreated mental illness in pregnancy, compared with the risks of various psychiatric medications; how to choose and dose medications for bipolar disorder as blood levels change across pregnancy; which resources to consult to determine the amounts and risks of various medications passed on in breast milk; and how to diagnose and treat premenstrual dysphoric disorder, to name a few lecture subjects.
By the time we were done, all residents had received more than 20 hours of teaching about how to treat mental illness in women across the reproductive life cycle. This was 20 hours more than is currently required by the American College of Graduate Medical Education, the accrediting body for all residencies, including psychiatry.1 It is time for that to change.
Women’s need for psychiatric treatment that addresses reproductive transitions is not new; it is as old as time. Not only do women who previously needed psychiatric treatment continue to need treatment when they get pregnant or are breastfeeding, but it is now well recognized that times of reproductive transition or flux – whether premenstrual, post partum, or perimenopausal – confer increased risk for both new-onset and exacerbations of prior mental illnesses.
What has changed is psychiatry’s ability to finally meet that need. Previously, despite the fact that women make up the majority of patients presenting for treatment, that nearly all women will menstruate and go through menopause, and that more than 80% of American women will have at least one pregnancy during their lifetime,psychiatrists practice as if these reproductive transitions were unfortunate blips getting in the doctor’s way.2 We mostly threw up our hands when our patients became pregnant, reflexively stopped all medications, and expected women to suffer for the sake of their babies.
with a large and growing research base, with both agreed-upon best practices and evolving standards of care informed by and responsive to the scientific literature. We now know that untreated maternal psychiatric illness carries its own risks for infants both before and after delivery; that many maternal pharmacologic treatments are lower risk for infants than previously thought; that protecting and treating women’s mental health in pregnancy has benefits for women, their babies, and the families that depend on them; and that there is now a growing evidence base informing both new and older treatments and enabling women and their doctors to make complex decisions balancing risk and benefit across the life cycle.
Many psychiatrists-in-training are hungry for this knowledge. At last count, in the United States alone, there were 16 women’s mental health fellowships available, up from just 3 in 2008.3 The problem is that none of them are accredited or funded by the ACGME, because reproductive psychiatry (here used interchangeably with the term women’s mental health) has not been officially recognized as a subspecialty. This means that current funding frequently rests on philanthropy, which often cannot be sustained, and clinical billing, which gives fellows in some programs such heavy clinical responsibilities that little time is left for scholarly work. Lack of subspecialty status also blocks numerous important downstream effects that would flow from this recognition.
Reproductive psychiatry clearly already meets criteria laid out by the American Board of Medical Specialties for defining a subspecialty field. As argued elsewhere, it has a distinct patient population with definable care needs and a standalone body of scientific medical knowledge as well as a national (and international) community of experts that has already done much to improve women’s access to care they desperately need.4 It also meets the ACGME’s criteria for a new subspecialty except for approval by the American Board of Psychiatry and Neurology.5 Finally, it also meets the requirements of the ABPN except for having 25 fellowship programs with 50 fellowship positions and 50 trainees per year completing fellowships, a challenging Catch-22 without the necessary funding that would accrue from accreditation.6
Despite growing awareness and demand, there remains a shortage of psychiatrists trained to treat women during times of reproductive transition and to pass their recommendations and knowledge on to their primary care and ob.gyn. colleagues. What official recognition would bring, in addition to funding for fellowships post residency, is a guaranteed seat at the table in psychiatry residencies, in terms of a required number of hours devoted to these topics for trainees, ensuring that all graduating psychiatrists have at least some exposure to the knowledge and practices so material to their patients.
It isn’t enough to wait for residencies to see the writing on the wall and voluntarily carve out a slice of pie devoted to women’s mental health from the limited time and resources available to train residents. A 2017 survey of psychiatry residency program training directors found that 23%, or almost a quarter of programs that responded, offered no reproductive psychiatry training at all, that 49% required 5 hours or less across all 4 years of training, and that 75% of programs had no required clinical exposure to reproductive psychiatry patients.7 Despite the fact that 87% of training directors surveyed agreed either that reproductive psychiatry was “an important area of education” or a subject general residents should be competent in, ACGME-recognized specialties take precedence.
A system so patchy and insufficient won’t do. It’s not good enough for the trainees who frequently have to look outside of their own institutions for the training they know they need. It’s not good enough for the pregnant or postpartum patient looking for evidence-based advice, who is currently left on her own to determine, prior to booking an appointment, whether a specific psychiatrist has received any training relevant to treating her. Adding reproductive psychiatry to the topics a graduating psychiatrist must have some proficiency in also signals to recent graduates and experienced attendings, as well as the relevant examining boards and producers of continuing medical education content, that women’s mental health is no longer a fringe topic but rather foundational to all practicing psychiatrists.
The oil needed to prime this pump is official recognition of the subspecialty that reproductive psychiatry already is. The women’s mental health community is ready. The research base is well established and growing exponentially. The number of women’s mental health fellowships is healthy and would increase significantly with ACGME funding. Psychiatry residency training programs can turn to recent graduates of these fellowships as well as their own faculty with reproductive psychiatry experience to teach trainees. In addition, the National Curriculum in Reproductive Psychiatry, over the last 4 years, has created a repository of free online modules dedicated to facilitating this type of training, with case discussions across numerous topics for use by both educators and trainees. The American Psychiatric Association recently formed the Committee on Women’s Mental Health in 2020 and will be publishing a textbook based on work done by the NCRP within the coming year.
Imagine the changed world that would open to all psychiatrists if reproductive psychiatry were given the credentials it deserves. When writing prescriptions, we would view pregnancy as the potential outcome it is in any woman of reproductive age, given that 50% of pregnancies are unplanned, and let women know ahead of time how to think about possible fetal effects rather than waiting for their panicked phone messages or hearing that they have stopped their medications abruptly. We would work to identify our patient’s individual risk factors for postpartum depression predelivery to reduce that risk and prevent or limit illness. We would plan ahead for close follow-up post partum during the window of greatest risk, rather than expecting women to drop out of care while taking care of their infants or languish on scheduling waiting lists. We would feel confident in giving evidence-based advice to our patients around times of reproductive transition across the life cycle, but especially in pregnancy and lactation, empowering women to make healthy decisions for themselves and their families, no longer abandoning them just when they need us most.
References
1. ACGME Program Requirements for Graduate Medical Education in Psychiatry. Accreditation Counsel for Graduate Medical Education. 2020 Jul 1.
2. Livingston G. “They’re waiting longer, but U.S. women today more likely to have children than a decade ago.” Pew Research Center’s Social & Demographic Trends Project. pewsocialtrends.org. 2018 Jan 18.
3. Nagle-Yang S et al. Acad Psychiatry. 2018 Apr;42(2):202-6.
4. Payne JL. Int Rev Psychiatry. 2019 May;31(3):207-9.
5. Accreditation Council for Graduate Medical Education Policies and Procedures. 2020 Sep 26.
6. American Board of Psychiatry and Neurology. Requirements for Subspecialty Recognition, Attachment A. 2008.
7. Osborne LM et al. Acad Psychiatry. 2018 Apr;42(2):197-201.
Dr. Leistikow is a reproductive psychiatrist and clinical assistant professor in the department of psychiatry at the University of Maryland, Baltimore, where she sees patients and helps train residents and fellows. She is on the education committee of the National Curriculum in Reproductive Psychiatry (NCRPtraining.org) and has written about women’s mental health for textbooks, scientific journals and on her private practice blog at www.womenspsychiatrybaltimore.com. Dr. Leistikow has no conflicts of interest.
Dr. Payne is associate professor of psychiatry and behavioral sciences and director of the Women’s Mood Disorders Center at Johns Hopkins University, Baltimore. In addition to providing outstanding clinical care for women with mood disorders, she conducts research into the genetic, biological, and environmental factors involved in postpartum depression. She and her colleagues have recently identified two epigenetic biomarkers of postpartum depression and are working hard to replicate this work with National Institutes of Health funding. Most recently, she was appointed to the American Psychiatric Association’s committee on women’s mental health and is serving as president-elect for both the Marcé of North America and the International Marcé Perinatal Mental Health Societies. She disclosed the following relevant financial relationships: serve(d) as a director, officer, partner, employee, adviser, consultant, or trustee for Sage Therapeutics and Janssen Pharmaceuticals.
Dr. Osborne is associate professor of psychiatry and behavioral sciences and of gynecology and obstetrics at Johns Hopkins University, where she directs a postdoctoral fellowship program in reproductive psychiatry. She is an expert on the diagnosis and treatment of mood and anxiety disorders during pregnancy, the post partum, the premenstrual period, and perimenopause. Her work is supported by the Brain and Behavior Foundation, the Doris Duke Foundation, the American Board of Psychiatry and Neurology, and the National Institute of Mental Health. She has no conflicts of interest.
Age at menarche signals potential cardiovascular health risk
“Increases in age at menarche are significantly associated with increases in cardiovascular health among women,” reported Yi Zheng, MPH, and colleagues at the University of Florida, Gainesville.
Mr. Zheng and colleagues conducted a cross-sectional analysis of 20,447 women aged 18 or older using data from a nationally representative sample of the 1999-2016 National Health and Nutrition Examinations Survey (NHANES). In all, 2,292 (11.2%) were determined to have ideal cardiovascular health (CVH).
Early menarche was confirmed to be related to increases in body mass index and greater incidence of type 2 diabetes, consistent with earlier studies, the authors confirmed. Those with nonideal CVH were more likely to have reported early menarche; those with ideal CVH were not only younger, but they also had college or graduate level education or above and higher poverty income ratio. Those with ideal CVH were also less likely to be to be of non-Hispanic Black heritage or to have been previously married.
BMI may be the missing link between early menarche and CVH
Unlike previous studies, the researchers found no significant link between early menarche and blood pressure, total cholesterol, smoking, physical activity, or diet using fully adjusted model data, leading them to conclude that “the associations between early menarche and CVH might be mainly driven by its associations with BMI.”
Mr. Zheng and colleagues suggested that future studies should evaluate the causal relationships between age at menarche and BMI and whether genetic factors and childhood lifestyle predispose women to early menarche and obesity.
“Our findings further highlighted that age at menarche may be used to identify high-risk population[s] and to guide targeted preventions to maintain and improve CVH,” the authors noted. Although they cited several strengths and limitations of the study, they emphasized that the wide use of Life’s Simple 7 factors (blood pressure, total cholesterol, glucose levels, smoking, BMI, physical activity, and diet) to measure CVH should “only be regarded as a surrogate construct, and future efforts are needed to better characterize CVH,” they cautioned.
The findings offer an opportunity to more closely track CVH in racial and ethnic groups
In a separate editorial, Ewa M. Gross-Sawicka, MD, PhD, and Eiran Z. Gorodeski, MD, MPH, both of the Harrington Heart and Vascular Institute, Cleveland, observed: “That the authors found African American women had the lowest overall CVH scores, even after adjusting for differences, highlights the importance of beginning cardiovascular health education earlier, especially for those in certain racial and ethnic groups.”
Dr. Gross-Sawicka and Dr. Gorodeski also raised several key questions that warrant further research: “1) Why do women who experience late menarche have improved cardiovascular health while those who experience early menarche have reduced cardiovascular health? 2) Why do the ‘beneficial’ effects of late menarche on CVH last 10 years longer than the ‘detrimental’ effects of early menarche? 3) Since both early and late menarche are associated with increased risk of cardiovascular disease, are women who experience menarche at an older age more cognizant of the cardiovascular risks compared with younger women and adjust their CVH accordingly?”
A key point also worth further consideration: “It is unclear whether age at menarche is directly associated with CVH, or if this relationship is mediated by the association of age at menarche and BMI and/or hyperglycemia,” said Dr. Gross-Sawicka and Dr. Gorodeski.
In an interview, Jan Shifren, MD, director, Midlife Women’s Health Center, Massachusetts General Hospital, Boston, noted, “The principal finding is that early menarche is associated with worse cardiovascular health, which may reflect the adverse impact of obesity and glucose intolerance on CVH, as obesity also is a risk factor for early menarche. The association between early menarche and worse CVH was significant only in women aged 25-34 years, but not in older women, possibly as other risk factors become more important as women age. One of the most concerning findings in this study ... is that only 11% had ideal CVH based on a combination of behavioral and health factors. As cardiovascular disease is the leading cause of death for women, we must do a better job of optimizing [their] cardiovascular health. Clinicians need to focus on optimizing cardiovascular health for all of their midlife patients, whether or not they experienced early menarche!”
Mr. Zheng and colleagues, as well as Dr. Shifren and Dr. Grodeski, had no conflicts of interest to report. Dr. Gross-Sawicka has received funding from Abbott and Novartis.
“Increases in age at menarche are significantly associated with increases in cardiovascular health among women,” reported Yi Zheng, MPH, and colleagues at the University of Florida, Gainesville.
Mr. Zheng and colleagues conducted a cross-sectional analysis of 20,447 women aged 18 or older using data from a nationally representative sample of the 1999-2016 National Health and Nutrition Examinations Survey (NHANES). In all, 2,292 (11.2%) were determined to have ideal cardiovascular health (CVH).
Early menarche was confirmed to be related to increases in body mass index and greater incidence of type 2 diabetes, consistent with earlier studies, the authors confirmed. Those with nonideal CVH were more likely to have reported early menarche; those with ideal CVH were not only younger, but they also had college or graduate level education or above and higher poverty income ratio. Those with ideal CVH were also less likely to be to be of non-Hispanic Black heritage or to have been previously married.
BMI may be the missing link between early menarche and CVH
Unlike previous studies, the researchers found no significant link between early menarche and blood pressure, total cholesterol, smoking, physical activity, or diet using fully adjusted model data, leading them to conclude that “the associations between early menarche and CVH might be mainly driven by its associations with BMI.”
Mr. Zheng and colleagues suggested that future studies should evaluate the causal relationships between age at menarche and BMI and whether genetic factors and childhood lifestyle predispose women to early menarche and obesity.
“Our findings further highlighted that age at menarche may be used to identify high-risk population[s] and to guide targeted preventions to maintain and improve CVH,” the authors noted. Although they cited several strengths and limitations of the study, they emphasized that the wide use of Life’s Simple 7 factors (blood pressure, total cholesterol, glucose levels, smoking, BMI, physical activity, and diet) to measure CVH should “only be regarded as a surrogate construct, and future efforts are needed to better characterize CVH,” they cautioned.
The findings offer an opportunity to more closely track CVH in racial and ethnic groups
In a separate editorial, Ewa M. Gross-Sawicka, MD, PhD, and Eiran Z. Gorodeski, MD, MPH, both of the Harrington Heart and Vascular Institute, Cleveland, observed: “That the authors found African American women had the lowest overall CVH scores, even after adjusting for differences, highlights the importance of beginning cardiovascular health education earlier, especially for those in certain racial and ethnic groups.”
Dr. Gross-Sawicka and Dr. Gorodeski also raised several key questions that warrant further research: “1) Why do women who experience late menarche have improved cardiovascular health while those who experience early menarche have reduced cardiovascular health? 2) Why do the ‘beneficial’ effects of late menarche on CVH last 10 years longer than the ‘detrimental’ effects of early menarche? 3) Since both early and late menarche are associated with increased risk of cardiovascular disease, are women who experience menarche at an older age more cognizant of the cardiovascular risks compared with younger women and adjust their CVH accordingly?”
A key point also worth further consideration: “It is unclear whether age at menarche is directly associated with CVH, or if this relationship is mediated by the association of age at menarche and BMI and/or hyperglycemia,” said Dr. Gross-Sawicka and Dr. Gorodeski.
In an interview, Jan Shifren, MD, director, Midlife Women’s Health Center, Massachusetts General Hospital, Boston, noted, “The principal finding is that early menarche is associated with worse cardiovascular health, which may reflect the adverse impact of obesity and glucose intolerance on CVH, as obesity also is a risk factor for early menarche. The association between early menarche and worse CVH was significant only in women aged 25-34 years, but not in older women, possibly as other risk factors become more important as women age. One of the most concerning findings in this study ... is that only 11% had ideal CVH based on a combination of behavioral and health factors. As cardiovascular disease is the leading cause of death for women, we must do a better job of optimizing [their] cardiovascular health. Clinicians need to focus on optimizing cardiovascular health for all of their midlife patients, whether or not they experienced early menarche!”
Mr. Zheng and colleagues, as well as Dr. Shifren and Dr. Grodeski, had no conflicts of interest to report. Dr. Gross-Sawicka has received funding from Abbott and Novartis.
“Increases in age at menarche are significantly associated with increases in cardiovascular health among women,” reported Yi Zheng, MPH, and colleagues at the University of Florida, Gainesville.
Mr. Zheng and colleagues conducted a cross-sectional analysis of 20,447 women aged 18 or older using data from a nationally representative sample of the 1999-2016 National Health and Nutrition Examinations Survey (NHANES). In all, 2,292 (11.2%) were determined to have ideal cardiovascular health (CVH).
Early menarche was confirmed to be related to increases in body mass index and greater incidence of type 2 diabetes, consistent with earlier studies, the authors confirmed. Those with nonideal CVH were more likely to have reported early menarche; those with ideal CVH were not only younger, but they also had college or graduate level education or above and higher poverty income ratio. Those with ideal CVH were also less likely to be to be of non-Hispanic Black heritage or to have been previously married.
BMI may be the missing link between early menarche and CVH
Unlike previous studies, the researchers found no significant link between early menarche and blood pressure, total cholesterol, smoking, physical activity, or diet using fully adjusted model data, leading them to conclude that “the associations between early menarche and CVH might be mainly driven by its associations with BMI.”
Mr. Zheng and colleagues suggested that future studies should evaluate the causal relationships between age at menarche and BMI and whether genetic factors and childhood lifestyle predispose women to early menarche and obesity.
“Our findings further highlighted that age at menarche may be used to identify high-risk population[s] and to guide targeted preventions to maintain and improve CVH,” the authors noted. Although they cited several strengths and limitations of the study, they emphasized that the wide use of Life’s Simple 7 factors (blood pressure, total cholesterol, glucose levels, smoking, BMI, physical activity, and diet) to measure CVH should “only be regarded as a surrogate construct, and future efforts are needed to better characterize CVH,” they cautioned.
The findings offer an opportunity to more closely track CVH in racial and ethnic groups
In a separate editorial, Ewa M. Gross-Sawicka, MD, PhD, and Eiran Z. Gorodeski, MD, MPH, both of the Harrington Heart and Vascular Institute, Cleveland, observed: “That the authors found African American women had the lowest overall CVH scores, even after adjusting for differences, highlights the importance of beginning cardiovascular health education earlier, especially for those in certain racial and ethnic groups.”
Dr. Gross-Sawicka and Dr. Gorodeski also raised several key questions that warrant further research: “1) Why do women who experience late menarche have improved cardiovascular health while those who experience early menarche have reduced cardiovascular health? 2) Why do the ‘beneficial’ effects of late menarche on CVH last 10 years longer than the ‘detrimental’ effects of early menarche? 3) Since both early and late menarche are associated with increased risk of cardiovascular disease, are women who experience menarche at an older age more cognizant of the cardiovascular risks compared with younger women and adjust their CVH accordingly?”
A key point also worth further consideration: “It is unclear whether age at menarche is directly associated with CVH, or if this relationship is mediated by the association of age at menarche and BMI and/or hyperglycemia,” said Dr. Gross-Sawicka and Dr. Gorodeski.
In an interview, Jan Shifren, MD, director, Midlife Women’s Health Center, Massachusetts General Hospital, Boston, noted, “The principal finding is that early menarche is associated with worse cardiovascular health, which may reflect the adverse impact of obesity and glucose intolerance on CVH, as obesity also is a risk factor for early menarche. The association between early menarche and worse CVH was significant only in women aged 25-34 years, but not in older women, possibly as other risk factors become more important as women age. One of the most concerning findings in this study ... is that only 11% had ideal CVH based on a combination of behavioral and health factors. As cardiovascular disease is the leading cause of death for women, we must do a better job of optimizing [their] cardiovascular health. Clinicians need to focus on optimizing cardiovascular health for all of their midlife patients, whether or not they experienced early menarche!”
Mr. Zheng and colleagues, as well as Dr. Shifren and Dr. Grodeski, had no conflicts of interest to report. Dr. Gross-Sawicka has received funding from Abbott and Novartis.
FROM THE JOURNAL OF THE NORTH AMERICAN MENOPAUSE SOCIETY
Case study: Maternal cervical cancer linked to neonate lung cancer
That’s the conclusion of two ground-breaking cases from Japan in which investigators describe lung cancer in two boys that “probably developed” from their respective mothers via vaginal transmission during birth.
“Transmission of maternal cancer to offspring is extremely rare and is estimated to occur in approximately 1 infant per every 500,000 mothers with cancer,” wrote Ayumu Arakawa, MD, of the National Cancer Center Hospital in Japan, and colleagues, in a paper published Jan. 7 in The New England Journal of Medicine.
Previous cases, of which only 18 have been recorded, have been presumed to occur via transplacental transmission, they said.
In the two new cases, genetic analyses and other evidence suggest that both boys’ lung cancers developed after aspirating uterine cervical cancer tumor cells into their lungs during passage through the birth canal.
Tragically, both mothers, each of whom was diagnosed with cervical cancer after the births, died while their boys were still infants.
“Most of the maternal-to-infant cases reported have been leukemia or melanoma,” said Mel Greaves, PhD, of the Institute of Cancer Research, London, who was asked for comment. In 2009, Dr. Greaves and colleagues published a case study of maternal-to-infant cancer transmission (presumably via the placenta). “It attracted an enormous amount of publicity and no doubt some alarm,” he said in an interview. He emphasized that the phenomenon is “incredibly rare.”
Dr. Greaves explains why such transmission is so rare. “We suspect that cancer cells do transit from mum to unborn child more often, but the foreign (aka paternal) antigens (HLA) on the tumor cells prompt immunological rejection. The extremely rare cases of successful transmission probably do depend on the fortuitous loss of paternal HLA.”
Advances in genetic technology may allow such cases, which have been recorded since 1950, to be rapidly identified now, he said.
“Where there is an adult-type cancer in an infant or child whose mother carried cancer when pregnant, then whole-genome sequencing should quickly tell if the infant’s tumor was of maternal origin,” Dr. Greaves explained.
“I think we will be seeing more reports like this in the future, now that this phenomenon has been described and next-generation sequencing is more readily available,” added Mae Zakhour, MD, of the University of California, Los Angeles, Jonsson Comprehensive Cancer Center, when asked for comment.
In the case of the Japanese boys, both cases were discovered incidentally during an analysis of the results of routine next-generation sequencing testing in a prospective gene-profiling trial in cancer patients, known as TOP-GEAR.
How do the investigators know that the spread happened vaginally and not via the placenta?
They explained that, in other cases of mother-to-fetus transmission, the offspring presented with multiple metastases in the brain, bones, liver, lungs, and soft tissues, which were “consistent with presumed hematogenous spread from the placenta.” However, in the two boys, tumors were observed only in the lungs and were localized along the bronchi.
That peribronchial pattern of tumor growth “suggested that the tumors arose from mother-to-infant vaginal transmission through aspiration of tumor-contaminated vaginal fluids during birth.”
In addition, the tumors in both boys lacked the Y chromosome and shared multiple somatic mutations, an HPV genome, and SNP alleles with tumors from the mothers.
“The identical molecular profiles of maternal and pediatric tumors demonstrated by next-generation sequencing, as well as the location of the tumors in the children, provides strong evidence for cancer transmission during delivery,” Dr. Zakhour summarized.
C-section question
The first of the cases reported by the Japanese team was a toddler (23 months) who presented to a local hospital with a 2-week history of a productive cough. Computed tomography revealed multiple masses scattered along the bronchi in both lungs, and a biopsy revealed neuroendocrine carcinoma of the lung.
Notably, the mother’s cervical cancer was not diagnosed during her pregnancy. A cervical cytologic test performed in the mother 7 months before the birth was negative. The infant was delivered transvaginally at 39 weeks of gestation.
It was only 3 months after the birth that the 35-year-old mother received a diagnosis of squamous cell carcinoma of the cervix. She then underwent radical hysterectomy with pelvic lymphadenectomy, followed by chemotherapy.
Had it been known that she had cervical cancer, she may have been advised to have a cesarean section.
The study authors propose, on the basis of their paper, that all women with cervical cancer should have a cesarean section.
But a U.S. expert questioned this, and said the situation is “a bit nuanced.”
William Grobman, MD, of Northwestern University in Chicago, said the current standard recommendation for many pregnant women known to have cervical cancer is to have a cesarean section and that “the strength of the recommendation is dependent on factors such as stage and size.”
However, in an interview, he added that “it may be premature to make a blanket recommendation for all people based on two reports without any idea of the frequency of this event, and with such uncertainty, it seems that disclosure of all information and shared decision-making would be a key approach.”
In this case report, the authors also noted that the cancer found in the toddler looked similar to the cancer in the mother.
“Histologic similarities between the tumor samples from the mother and child prompted us to compare the results of their next-generation sequencing tests,” they said.
The result? “The comparison of the gene profiles in the samples of tumor and normal tissue confirmed that transmission of maternal tumor to the child had occurred.”
The lung cancer in the toddler progressed despite two chemotherapy regimens, so he was enrolled in a clinical trial of nivolumab therapy. He had a response that continued for 7 months, with no appearance of new lesions. Lobectomy was performed to resect a single remaining nodule. The boy had no evidence of disease recurrence at 12 months after lobectomy.
His mother was also enrolled in a nivolumab trial, but her cervical cancer had spread, and she died 5 months after disease progression.
Second case
In the second reported case, a 6-year-old boy presented to a local hospital with chest pain on the left side. Computed tomography revealed a mass in the left lung, and mucinous adenocarcinoma was eventually diagnosed.
In this case, the mother had a cervical polypoid tumor detected during pregnancy. But, as in the other case, cervical cytologic analysis was negative. Because the tumor was stable without any intervention, the mother delivered the boy vaginally at 38 weeks of gestation.
However, after the delivery, biopsy of the cervical lesion revealed adenocarcinoma. The mother underwent radical hysterectomy and bilateral salpingo-oophorectomy 3 months after delivery. She died of the disease 2 years after the surgery.
The boy received chemotherapy and had a partial response, with a reduction in levels of the tumor marker CA19-9 to normal levels. But 3 months later, the disease recurred in the left lung. After more chemotherapy, he underwent total left pneumonectomy and was subsequently free of disease.
The study authors said that they did not suspect maternal transmission of the cancer when her child received a diagnosis at 6 years of age. They explained that metastatic cervical cancer is typically a fast-growing tumor and the slow growth in the child seemed inconsistent with the idea that the cancer had been transmitted to him.
However, the pathology exam showed that the boy had mucinous adenocarcinoma, “which is an unusual morphologic finding for a primary lung tumor, but it was similar to the uterine cervical tumor in the mother,” the authors reported.
Samples of the cervical tumor from the mother and from the lung tumor of the child were submitted for next-generation sequencing tests and, said the authors, indicated mother-to-infant transmission.
The study was supported by grants from the Japan Agency for Medical Research and Development; the National Cancer Center Research and Development Fund; and the Ministry of Education, Culture, Sports, Science and Technology; and funding from Ono Pharmaceutical.
A version of this article first appeared on Medscape.com.
That’s the conclusion of two ground-breaking cases from Japan in which investigators describe lung cancer in two boys that “probably developed” from their respective mothers via vaginal transmission during birth.
“Transmission of maternal cancer to offspring is extremely rare and is estimated to occur in approximately 1 infant per every 500,000 mothers with cancer,” wrote Ayumu Arakawa, MD, of the National Cancer Center Hospital in Japan, and colleagues, in a paper published Jan. 7 in The New England Journal of Medicine.
Previous cases, of which only 18 have been recorded, have been presumed to occur via transplacental transmission, they said.
In the two new cases, genetic analyses and other evidence suggest that both boys’ lung cancers developed after aspirating uterine cervical cancer tumor cells into their lungs during passage through the birth canal.
Tragically, both mothers, each of whom was diagnosed with cervical cancer after the births, died while their boys were still infants.
“Most of the maternal-to-infant cases reported have been leukemia or melanoma,” said Mel Greaves, PhD, of the Institute of Cancer Research, London, who was asked for comment. In 2009, Dr. Greaves and colleagues published a case study of maternal-to-infant cancer transmission (presumably via the placenta). “It attracted an enormous amount of publicity and no doubt some alarm,” he said in an interview. He emphasized that the phenomenon is “incredibly rare.”
Dr. Greaves explains why such transmission is so rare. “We suspect that cancer cells do transit from mum to unborn child more often, but the foreign (aka paternal) antigens (HLA) on the tumor cells prompt immunological rejection. The extremely rare cases of successful transmission probably do depend on the fortuitous loss of paternal HLA.”
Advances in genetic technology may allow such cases, which have been recorded since 1950, to be rapidly identified now, he said.
“Where there is an adult-type cancer in an infant or child whose mother carried cancer when pregnant, then whole-genome sequencing should quickly tell if the infant’s tumor was of maternal origin,” Dr. Greaves explained.
“I think we will be seeing more reports like this in the future, now that this phenomenon has been described and next-generation sequencing is more readily available,” added Mae Zakhour, MD, of the University of California, Los Angeles, Jonsson Comprehensive Cancer Center, when asked for comment.
In the case of the Japanese boys, both cases were discovered incidentally during an analysis of the results of routine next-generation sequencing testing in a prospective gene-profiling trial in cancer patients, known as TOP-GEAR.
How do the investigators know that the spread happened vaginally and not via the placenta?
They explained that, in other cases of mother-to-fetus transmission, the offspring presented with multiple metastases in the brain, bones, liver, lungs, and soft tissues, which were “consistent with presumed hematogenous spread from the placenta.” However, in the two boys, tumors were observed only in the lungs and were localized along the bronchi.
That peribronchial pattern of tumor growth “suggested that the tumors arose from mother-to-infant vaginal transmission through aspiration of tumor-contaminated vaginal fluids during birth.”
In addition, the tumors in both boys lacked the Y chromosome and shared multiple somatic mutations, an HPV genome, and SNP alleles with tumors from the mothers.
“The identical molecular profiles of maternal and pediatric tumors demonstrated by next-generation sequencing, as well as the location of the tumors in the children, provides strong evidence for cancer transmission during delivery,” Dr. Zakhour summarized.
C-section question
The first of the cases reported by the Japanese team was a toddler (23 months) who presented to a local hospital with a 2-week history of a productive cough. Computed tomography revealed multiple masses scattered along the bronchi in both lungs, and a biopsy revealed neuroendocrine carcinoma of the lung.
Notably, the mother’s cervical cancer was not diagnosed during her pregnancy. A cervical cytologic test performed in the mother 7 months before the birth was negative. The infant was delivered transvaginally at 39 weeks of gestation.
It was only 3 months after the birth that the 35-year-old mother received a diagnosis of squamous cell carcinoma of the cervix. She then underwent radical hysterectomy with pelvic lymphadenectomy, followed by chemotherapy.
Had it been known that she had cervical cancer, she may have been advised to have a cesarean section.
The study authors propose, on the basis of their paper, that all women with cervical cancer should have a cesarean section.
But a U.S. expert questioned this, and said the situation is “a bit nuanced.”
William Grobman, MD, of Northwestern University in Chicago, said the current standard recommendation for many pregnant women known to have cervical cancer is to have a cesarean section and that “the strength of the recommendation is dependent on factors such as stage and size.”
However, in an interview, he added that “it may be premature to make a blanket recommendation for all people based on two reports without any idea of the frequency of this event, and with such uncertainty, it seems that disclosure of all information and shared decision-making would be a key approach.”
In this case report, the authors also noted that the cancer found in the toddler looked similar to the cancer in the mother.
“Histologic similarities between the tumor samples from the mother and child prompted us to compare the results of their next-generation sequencing tests,” they said.
The result? “The comparison of the gene profiles in the samples of tumor and normal tissue confirmed that transmission of maternal tumor to the child had occurred.”
The lung cancer in the toddler progressed despite two chemotherapy regimens, so he was enrolled in a clinical trial of nivolumab therapy. He had a response that continued for 7 months, with no appearance of new lesions. Lobectomy was performed to resect a single remaining nodule. The boy had no evidence of disease recurrence at 12 months after lobectomy.
His mother was also enrolled in a nivolumab trial, but her cervical cancer had spread, and she died 5 months after disease progression.
Second case
In the second reported case, a 6-year-old boy presented to a local hospital with chest pain on the left side. Computed tomography revealed a mass in the left lung, and mucinous adenocarcinoma was eventually diagnosed.
In this case, the mother had a cervical polypoid tumor detected during pregnancy. But, as in the other case, cervical cytologic analysis was negative. Because the tumor was stable without any intervention, the mother delivered the boy vaginally at 38 weeks of gestation.
However, after the delivery, biopsy of the cervical lesion revealed adenocarcinoma. The mother underwent radical hysterectomy and bilateral salpingo-oophorectomy 3 months after delivery. She died of the disease 2 years after the surgery.
The boy received chemotherapy and had a partial response, with a reduction in levels of the tumor marker CA19-9 to normal levels. But 3 months later, the disease recurred in the left lung. After more chemotherapy, he underwent total left pneumonectomy and was subsequently free of disease.
The study authors said that they did not suspect maternal transmission of the cancer when her child received a diagnosis at 6 years of age. They explained that metastatic cervical cancer is typically a fast-growing tumor and the slow growth in the child seemed inconsistent with the idea that the cancer had been transmitted to him.
However, the pathology exam showed that the boy had mucinous adenocarcinoma, “which is an unusual morphologic finding for a primary lung tumor, but it was similar to the uterine cervical tumor in the mother,” the authors reported.
Samples of the cervical tumor from the mother and from the lung tumor of the child were submitted for next-generation sequencing tests and, said the authors, indicated mother-to-infant transmission.
The study was supported by grants from the Japan Agency for Medical Research and Development; the National Cancer Center Research and Development Fund; and the Ministry of Education, Culture, Sports, Science and Technology; and funding from Ono Pharmaceutical.
A version of this article first appeared on Medscape.com.
That’s the conclusion of two ground-breaking cases from Japan in which investigators describe lung cancer in two boys that “probably developed” from their respective mothers via vaginal transmission during birth.
“Transmission of maternal cancer to offspring is extremely rare and is estimated to occur in approximately 1 infant per every 500,000 mothers with cancer,” wrote Ayumu Arakawa, MD, of the National Cancer Center Hospital in Japan, and colleagues, in a paper published Jan. 7 in The New England Journal of Medicine.
Previous cases, of which only 18 have been recorded, have been presumed to occur via transplacental transmission, they said.
In the two new cases, genetic analyses and other evidence suggest that both boys’ lung cancers developed after aspirating uterine cervical cancer tumor cells into their lungs during passage through the birth canal.
Tragically, both mothers, each of whom was diagnosed with cervical cancer after the births, died while their boys were still infants.
“Most of the maternal-to-infant cases reported have been leukemia or melanoma,” said Mel Greaves, PhD, of the Institute of Cancer Research, London, who was asked for comment. In 2009, Dr. Greaves and colleagues published a case study of maternal-to-infant cancer transmission (presumably via the placenta). “It attracted an enormous amount of publicity and no doubt some alarm,” he said in an interview. He emphasized that the phenomenon is “incredibly rare.”
Dr. Greaves explains why such transmission is so rare. “We suspect that cancer cells do transit from mum to unborn child more often, but the foreign (aka paternal) antigens (HLA) on the tumor cells prompt immunological rejection. The extremely rare cases of successful transmission probably do depend on the fortuitous loss of paternal HLA.”
Advances in genetic technology may allow such cases, which have been recorded since 1950, to be rapidly identified now, he said.
“Where there is an adult-type cancer in an infant or child whose mother carried cancer when pregnant, then whole-genome sequencing should quickly tell if the infant’s tumor was of maternal origin,” Dr. Greaves explained.
“I think we will be seeing more reports like this in the future, now that this phenomenon has been described and next-generation sequencing is more readily available,” added Mae Zakhour, MD, of the University of California, Los Angeles, Jonsson Comprehensive Cancer Center, when asked for comment.
In the case of the Japanese boys, both cases were discovered incidentally during an analysis of the results of routine next-generation sequencing testing in a prospective gene-profiling trial in cancer patients, known as TOP-GEAR.
How do the investigators know that the spread happened vaginally and not via the placenta?
They explained that, in other cases of mother-to-fetus transmission, the offspring presented with multiple metastases in the brain, bones, liver, lungs, and soft tissues, which were “consistent with presumed hematogenous spread from the placenta.” However, in the two boys, tumors were observed only in the lungs and were localized along the bronchi.
That peribronchial pattern of tumor growth “suggested that the tumors arose from mother-to-infant vaginal transmission through aspiration of tumor-contaminated vaginal fluids during birth.”
In addition, the tumors in both boys lacked the Y chromosome and shared multiple somatic mutations, an HPV genome, and SNP alleles with tumors from the mothers.
“The identical molecular profiles of maternal and pediatric tumors demonstrated by next-generation sequencing, as well as the location of the tumors in the children, provides strong evidence for cancer transmission during delivery,” Dr. Zakhour summarized.
C-section question
The first of the cases reported by the Japanese team was a toddler (23 months) who presented to a local hospital with a 2-week history of a productive cough. Computed tomography revealed multiple masses scattered along the bronchi in both lungs, and a biopsy revealed neuroendocrine carcinoma of the lung.
Notably, the mother’s cervical cancer was not diagnosed during her pregnancy. A cervical cytologic test performed in the mother 7 months before the birth was negative. The infant was delivered transvaginally at 39 weeks of gestation.
It was only 3 months after the birth that the 35-year-old mother received a diagnosis of squamous cell carcinoma of the cervix. She then underwent radical hysterectomy with pelvic lymphadenectomy, followed by chemotherapy.
Had it been known that she had cervical cancer, she may have been advised to have a cesarean section.
The study authors propose, on the basis of their paper, that all women with cervical cancer should have a cesarean section.
But a U.S. expert questioned this, and said the situation is “a bit nuanced.”
William Grobman, MD, of Northwestern University in Chicago, said the current standard recommendation for many pregnant women known to have cervical cancer is to have a cesarean section and that “the strength of the recommendation is dependent on factors such as stage and size.”
However, in an interview, he added that “it may be premature to make a blanket recommendation for all people based on two reports without any idea of the frequency of this event, and with such uncertainty, it seems that disclosure of all information and shared decision-making would be a key approach.”
In this case report, the authors also noted that the cancer found in the toddler looked similar to the cancer in the mother.
“Histologic similarities between the tumor samples from the mother and child prompted us to compare the results of their next-generation sequencing tests,” they said.
The result? “The comparison of the gene profiles in the samples of tumor and normal tissue confirmed that transmission of maternal tumor to the child had occurred.”
The lung cancer in the toddler progressed despite two chemotherapy regimens, so he was enrolled in a clinical trial of nivolumab therapy. He had a response that continued for 7 months, with no appearance of new lesions. Lobectomy was performed to resect a single remaining nodule. The boy had no evidence of disease recurrence at 12 months after lobectomy.
His mother was also enrolled in a nivolumab trial, but her cervical cancer had spread, and she died 5 months after disease progression.
Second case
In the second reported case, a 6-year-old boy presented to a local hospital with chest pain on the left side. Computed tomography revealed a mass in the left lung, and mucinous adenocarcinoma was eventually diagnosed.
In this case, the mother had a cervical polypoid tumor detected during pregnancy. But, as in the other case, cervical cytologic analysis was negative. Because the tumor was stable without any intervention, the mother delivered the boy vaginally at 38 weeks of gestation.
However, after the delivery, biopsy of the cervical lesion revealed adenocarcinoma. The mother underwent radical hysterectomy and bilateral salpingo-oophorectomy 3 months after delivery. She died of the disease 2 years after the surgery.
The boy received chemotherapy and had a partial response, with a reduction in levels of the tumor marker CA19-9 to normal levels. But 3 months later, the disease recurred in the left lung. After more chemotherapy, he underwent total left pneumonectomy and was subsequently free of disease.
The study authors said that they did not suspect maternal transmission of the cancer when her child received a diagnosis at 6 years of age. They explained that metastatic cervical cancer is typically a fast-growing tumor and the slow growth in the child seemed inconsistent with the idea that the cancer had been transmitted to him.
However, the pathology exam showed that the boy had mucinous adenocarcinoma, “which is an unusual morphologic finding for a primary lung tumor, but it was similar to the uterine cervical tumor in the mother,” the authors reported.
Samples of the cervical tumor from the mother and from the lung tumor of the child were submitted for next-generation sequencing tests and, said the authors, indicated mother-to-infant transmission.
The study was supported by grants from the Japan Agency for Medical Research and Development; the National Cancer Center Research and Development Fund; and the Ministry of Education, Culture, Sports, Science and Technology; and funding from Ono Pharmaceutical.
A version of this article first appeared on Medscape.com.
Consensus bundle has potential to affect postpartum morbidity, mortality
Health care professionals miss an important opportunity if they do not use a holistic patient-centered approach in providing support to their postpartum patients, advised Alison M. Stuebe, MD, MSc, of the division of maternal-fetal medicine at the University of North Carolina at Chapel Hill, and colleagues.
Too often, the emphasis for health care professionals is on less common, highly morbid complications, while their patients’ needs are focused more on daily functioning.
More than half of maternal deaths occur not during delivery, but in the days, weeks, and months following birth. Many serious complications and management of less-emergent conditions such as flu-like symptoms go unnoticed or result in treatment delays. Routinely asking women who present for care whether they have been pregnant during the past year could go a long way in helping clinicians connect the dots for conditions they might not otherwise consider, suggested Dr. Stuebe and colleagues.
In response to what is becoming a growing burden of postpartum maternal morbidity and mortality in the United States, Dr. Stuebe and associates, in coordination with the Alliance of Innovation in Maternal Health, prepared a comprehensive consensus statement on postpartum care basics. The work came out of a day-long planning meeting of an interdisciplinary work group funded by AIMH to develop the maternal safety bundle, which established 28 goals across four phases – readiness, recognition and prevention, response, and reporting. Each phase is intended to occur as part of a coordinated effort between women experiencing pregnancy and their health care providers, with involvement from multiple clinical settings and health systems.
America needs to change the way it treats its new mothers
In a separate interview, Dr. Stuebe shared additional insights into the development of the consensus statement: “This article is part of a broader movement to change the way America treats new mothers. For too long, we’ve lavished attention on women in the weeks before birth and then left them alone to manage recovery, plunging hormones, sleepless nights, and new baby care. The goal of this article is to set out the ways that we can better support women in this critical transition period.
“We need to start during pregnancy, by working with women to plan for the fourth trimester and identify a care team that will support her and her baby. After birth, we need to adapt those plans and make sure she has the emotional and material support she needs to navigate the coming months. Many of these decisions are deeply personal, and we need to center the values and preferences of the woman and her family.”
Dr. Stuebe also spotlighted a new University of North Carolina–based program designed to support shared decision-making. Their Trimester Project team partnered with mothers to design NewMomHealth.com, the first national postpartum resource designed by and for new mothers, which is in the process of launching a culturally adapted Spanish-language site, saludmadre.com, in partnership with Urban Strategies.
Readiness prepares mothers, HCPs, and clinical settings for the fourth trimester
In the first phase, Readiness, there are three key parties involved: the woman experiencing pregnancy, the health care providers (HCPs) supporting her, and the clinical settings involved in her care. Each player has specific goals to achieve in this phase, women are responsible for four goals, health care providers have two goals, and clinical settings have five.
Prenatally, every woman should work with health care providers to develop a comprehensive, individualized postpartum care plan that designates a postpartum clinical “home” for addressing any care needed between birth and the “comprehensive postpartum visit.” The plan encourages the patient and her health care team give attention to social support, birth recovery, infant care and feeding plans, thoughts concerning future pregnancies, and specifically use of contraception, as well as any chronic health concerns and overall emotional well-being.
The plan should consist of a postpartum care team that includes friends and family and ensures that medical, material, and social support are available in the weeks following birth for both mother and baby.
The authors emphasized the importance for clinicians to develop a keen awareness of “reproductive justice,” which respects a woman’s right “to maintain personal bodily autonomy, have children, not have children, and parent the children we have in safe and sustainable communities.” They encouraged adopting a sensitivity to concerns deeply rooted in the history of family planning, which is perceived to have been “fraught by coercive tactics to incentivize sterilization and contraceptive implants among marginalized groups.” With this in mind, they urged clinicians to respectfully ask about the patient’s intentions before making any suggestions concerning future births and family planning preferences.
HCP readiness ensures that each woman “has a documented postpartum care plan and care team identified in the prenatal period.” HCPs are also responsible for developing and maintaining “a working knowledge of evidence-based evaluation and management of common issues facing the mother-infant dyad.”
Clinical setting readiness ensures that woman-centered postpartum care and education have been developed and optimized using adult learning principles whenever possible. Diversity of family structures, cultural traditions, and parenting practices are fully embraced. Dr. Stuebe and colleagues emphasized that traditional top-down teaching methods are ineffective and should be avoided.
Clinical setting readiness also ensures that systems are developed to pair families with community-based resources that provide medical follow-up as well as social and material support. Clinical protocols and reimbursement options should be available that give women easy access to preferred contraception. Systems should be in place that facilitate prompt, pertinent communication between in- and outpatient providers. Lastly, and perhaps most importantly, clinical setting readiness demands the development of protocols to screen and treat key postpartum concerns, such as depression, substance use disorders, family violence, and even incontinence in coordination with locally identified specialists.
Recognition and prevention promotes accountability and establishes key guidelines
Recognition and prevention require coordinated participation between women experiencing pregnancy and the clinical environments supporting their care. In this phase, women are responsible for three goals; clinical environments have four goals.
Women are to be identified and respected as the expert most knowledgeable of their own needs, cautioned the authors. They should also be empowered to trust their instincts, seeking care as early and as often as needed in the weeks after they give birth. Postpartum care needs to be viewed as an ongoing process, not just a singular encounter.
Women also need to take ownership for their postpartum care plan, reviewing and revising it as needed in coordination with their health care providers before maternity discharge is complete. Every plan should include a comprehensive list of warning signs and recommended action plans when faced with life-threatening complications, a list of resources for lactation support, and a “first-call” phone number that identifies the postpartum medical providers available to address breastfeeding issues and for postpartum care visits, including prescheduled dates and times.
Lastly, women are to take responsibility for attending their postpartum visit. Although new guidance suggests that the comprehensive visit take place no later than 12 weeks post partum, it also recognizes multiple visits may be required to address individual needs. All women should have contact with a maternal care provider within 3 weeks post partum, the American College of Obstetricians and Gynecologists recommends.
Responsibility for recognition and prevention in the clinical setting begins with determining guidelines for early postpartum visits, considering chronic conditions such as hypertension, diabetes, or substance abuse disorder and risk for postpartum conditions such as wound complications or postpartum depression, the authors wrote. Ongoing care between inpatient and outpatient settings as well as between maternal and infant care providers is coordinated to ensure health and well-being of both patients. Screening and treatment of common comorbidities such as mental health issues, smoking, and substance use as well as issues related to unstable housing and food insecurities are also addressed in the clinical environment. Lastly, clinicians are tasked with ensuring that every patient has selected a primary care provider for ongoing care.
Response ensures key parties and resources are connected at every step
In the response phase, clinical settings and health care providers are the key participants and they have two goals each.
Every clinical setting is tasked with implementing treatment protocols and providing needed care or facilitating referral in a timely fashion. The importance of a “warm hand-off” and “face-to-face introduction to the specialist to whom the patient is being referred” is encouraged. They are also responsible for keeping an updated inventory of community resources on hand for such unmet needs as 24-hour hotlines, food banks, diaper banks, lactation support groups, and home-visiting programs.
Every health care provider is tasked with developing strategies designed to reach women who do not attend their comprehensive postpartum visit. They are also responsible for assessing the severity of identified needs, and arranging immediate transportation to appropriate facilities in life-threatening circumstances. In nonacute cases, the woman and her care providers work with her to make appropriate decisions.
Reporting gives health systems the opportunity to assess and improve
Sole responsibility in the reporting phase falls to every health system. In this phase, they have a total of six goals.
Health systems are ideally organized to convene strategy-sharing sessions with inpatient as well as outpatient professionals to evaluate successes and opportunities for improvement. They are equally qualified to identify and monitor such quality measures as postpartum emergency department utilization and readmission rates. They are tasked with working toward quality metrics that compare postpartum outcomes and prenatal intentions, including patient receipt of preferred contraception or completion of planned breastfeeding duration.
Health systems are expected to conduct quality improvement measures designed to reduce postpartum morbidity when preventable. They are also the logical choice for maintaining a clearinghouse for resources, in collaboration with the community, that is designed to meet the postpartum needs of women.
Lastly, they play an important role in ensuring that reimbursement policies are structured such that they do not disincentivize postpartum visits.
The consensus bundle encourages change in the way America treats its new mothers
Angela Bianco, MD, maternal and fetal medicine specialist at Mount Sinai Hospital, New York, also interviewed separately, noted: “I think the fact that ACOG has created a postpartum bundle for providers to use for guidance is long overdue. The current prenatal care paradigm focuses on numerous/intensive prenatal visits but only a single postpartum visit. Many women report feeling ill-equipped to navigate the challenges that arise post delivery, including self-care, newborn care, and breastfeeding. In addition, most women experience the impact of dramatic hormonal fluctuations resulting in mood alterations. Add profound sleep deprivation to this and the result is often some degree of postpartum blues and/or depression. The importance of anticipatory guidance for our patients cannot be underestimated. Helping to facilitate potential social support structures be in place prior to the birth is optimal. Providers should reinforce the importance of access to a variety of tools, including digital apps, community support groups, and 24/7 web access services. Moving forward, it should be considered unconscionable to send a new mother home without ensuring the appropriate resources are in place. Postpartum care should be tailored to a woman’s needs and may require multiple visits and/or referrals.”
Dr. Stuebe and colleagues, as well as Dr. Bianco, disclosed receiving honoraria for various projects. Funding for the study was supported by the Alliance for Innovation on Maternal and Child Health, which is funded by a grant from the Health Resources and Services Administration.
Health care professionals miss an important opportunity if they do not use a holistic patient-centered approach in providing support to their postpartum patients, advised Alison M. Stuebe, MD, MSc, of the division of maternal-fetal medicine at the University of North Carolina at Chapel Hill, and colleagues.
Too often, the emphasis for health care professionals is on less common, highly morbid complications, while their patients’ needs are focused more on daily functioning.
More than half of maternal deaths occur not during delivery, but in the days, weeks, and months following birth. Many serious complications and management of less-emergent conditions such as flu-like symptoms go unnoticed or result in treatment delays. Routinely asking women who present for care whether they have been pregnant during the past year could go a long way in helping clinicians connect the dots for conditions they might not otherwise consider, suggested Dr. Stuebe and colleagues.
In response to what is becoming a growing burden of postpartum maternal morbidity and mortality in the United States, Dr. Stuebe and associates, in coordination with the Alliance of Innovation in Maternal Health, prepared a comprehensive consensus statement on postpartum care basics. The work came out of a day-long planning meeting of an interdisciplinary work group funded by AIMH to develop the maternal safety bundle, which established 28 goals across four phases – readiness, recognition and prevention, response, and reporting. Each phase is intended to occur as part of a coordinated effort between women experiencing pregnancy and their health care providers, with involvement from multiple clinical settings and health systems.
America needs to change the way it treats its new mothers
In a separate interview, Dr. Stuebe shared additional insights into the development of the consensus statement: “This article is part of a broader movement to change the way America treats new mothers. For too long, we’ve lavished attention on women in the weeks before birth and then left them alone to manage recovery, plunging hormones, sleepless nights, and new baby care. The goal of this article is to set out the ways that we can better support women in this critical transition period.
“We need to start during pregnancy, by working with women to plan for the fourth trimester and identify a care team that will support her and her baby. After birth, we need to adapt those plans and make sure she has the emotional and material support she needs to navigate the coming months. Many of these decisions are deeply personal, and we need to center the values and preferences of the woman and her family.”
Dr. Stuebe also spotlighted a new University of North Carolina–based program designed to support shared decision-making. Their Trimester Project team partnered with mothers to design NewMomHealth.com, the first national postpartum resource designed by and for new mothers, which is in the process of launching a culturally adapted Spanish-language site, saludmadre.com, in partnership with Urban Strategies.
Readiness prepares mothers, HCPs, and clinical settings for the fourth trimester
In the first phase, Readiness, there are three key parties involved: the woman experiencing pregnancy, the health care providers (HCPs) supporting her, and the clinical settings involved in her care. Each player has specific goals to achieve in this phase, women are responsible for four goals, health care providers have two goals, and clinical settings have five.
Prenatally, every woman should work with health care providers to develop a comprehensive, individualized postpartum care plan that designates a postpartum clinical “home” for addressing any care needed between birth and the “comprehensive postpartum visit.” The plan encourages the patient and her health care team give attention to social support, birth recovery, infant care and feeding plans, thoughts concerning future pregnancies, and specifically use of contraception, as well as any chronic health concerns and overall emotional well-being.
The plan should consist of a postpartum care team that includes friends and family and ensures that medical, material, and social support are available in the weeks following birth for both mother and baby.
The authors emphasized the importance for clinicians to develop a keen awareness of “reproductive justice,” which respects a woman’s right “to maintain personal bodily autonomy, have children, not have children, and parent the children we have in safe and sustainable communities.” They encouraged adopting a sensitivity to concerns deeply rooted in the history of family planning, which is perceived to have been “fraught by coercive tactics to incentivize sterilization and contraceptive implants among marginalized groups.” With this in mind, they urged clinicians to respectfully ask about the patient’s intentions before making any suggestions concerning future births and family planning preferences.
HCP readiness ensures that each woman “has a documented postpartum care plan and care team identified in the prenatal period.” HCPs are also responsible for developing and maintaining “a working knowledge of evidence-based evaluation and management of common issues facing the mother-infant dyad.”
Clinical setting readiness ensures that woman-centered postpartum care and education have been developed and optimized using adult learning principles whenever possible. Diversity of family structures, cultural traditions, and parenting practices are fully embraced. Dr. Stuebe and colleagues emphasized that traditional top-down teaching methods are ineffective and should be avoided.
Clinical setting readiness also ensures that systems are developed to pair families with community-based resources that provide medical follow-up as well as social and material support. Clinical protocols and reimbursement options should be available that give women easy access to preferred contraception. Systems should be in place that facilitate prompt, pertinent communication between in- and outpatient providers. Lastly, and perhaps most importantly, clinical setting readiness demands the development of protocols to screen and treat key postpartum concerns, such as depression, substance use disorders, family violence, and even incontinence in coordination with locally identified specialists.
Recognition and prevention promotes accountability and establishes key guidelines
Recognition and prevention require coordinated participation between women experiencing pregnancy and the clinical environments supporting their care. In this phase, women are responsible for three goals; clinical environments have four goals.
Women are to be identified and respected as the expert most knowledgeable of their own needs, cautioned the authors. They should also be empowered to trust their instincts, seeking care as early and as often as needed in the weeks after they give birth. Postpartum care needs to be viewed as an ongoing process, not just a singular encounter.
Women also need to take ownership for their postpartum care plan, reviewing and revising it as needed in coordination with their health care providers before maternity discharge is complete. Every plan should include a comprehensive list of warning signs and recommended action plans when faced with life-threatening complications, a list of resources for lactation support, and a “first-call” phone number that identifies the postpartum medical providers available to address breastfeeding issues and for postpartum care visits, including prescheduled dates and times.
Lastly, women are to take responsibility for attending their postpartum visit. Although new guidance suggests that the comprehensive visit take place no later than 12 weeks post partum, it also recognizes multiple visits may be required to address individual needs. All women should have contact with a maternal care provider within 3 weeks post partum, the American College of Obstetricians and Gynecologists recommends.
Responsibility for recognition and prevention in the clinical setting begins with determining guidelines for early postpartum visits, considering chronic conditions such as hypertension, diabetes, or substance abuse disorder and risk for postpartum conditions such as wound complications or postpartum depression, the authors wrote. Ongoing care between inpatient and outpatient settings as well as between maternal and infant care providers is coordinated to ensure health and well-being of both patients. Screening and treatment of common comorbidities such as mental health issues, smoking, and substance use as well as issues related to unstable housing and food insecurities are also addressed in the clinical environment. Lastly, clinicians are tasked with ensuring that every patient has selected a primary care provider for ongoing care.
Response ensures key parties and resources are connected at every step
In the response phase, clinical settings and health care providers are the key participants and they have two goals each.
Every clinical setting is tasked with implementing treatment protocols and providing needed care or facilitating referral in a timely fashion. The importance of a “warm hand-off” and “face-to-face introduction to the specialist to whom the patient is being referred” is encouraged. They are also responsible for keeping an updated inventory of community resources on hand for such unmet needs as 24-hour hotlines, food banks, diaper banks, lactation support groups, and home-visiting programs.
Every health care provider is tasked with developing strategies designed to reach women who do not attend their comprehensive postpartum visit. They are also responsible for assessing the severity of identified needs, and arranging immediate transportation to appropriate facilities in life-threatening circumstances. In nonacute cases, the woman and her care providers work with her to make appropriate decisions.
Reporting gives health systems the opportunity to assess and improve
Sole responsibility in the reporting phase falls to every health system. In this phase, they have a total of six goals.
Health systems are ideally organized to convene strategy-sharing sessions with inpatient as well as outpatient professionals to evaluate successes and opportunities for improvement. They are equally qualified to identify and monitor such quality measures as postpartum emergency department utilization and readmission rates. They are tasked with working toward quality metrics that compare postpartum outcomes and prenatal intentions, including patient receipt of preferred contraception or completion of planned breastfeeding duration.
Health systems are expected to conduct quality improvement measures designed to reduce postpartum morbidity when preventable. They are also the logical choice for maintaining a clearinghouse for resources, in collaboration with the community, that is designed to meet the postpartum needs of women.
Lastly, they play an important role in ensuring that reimbursement policies are structured such that they do not disincentivize postpartum visits.
The consensus bundle encourages change in the way America treats its new mothers
Angela Bianco, MD, maternal and fetal medicine specialist at Mount Sinai Hospital, New York, also interviewed separately, noted: “I think the fact that ACOG has created a postpartum bundle for providers to use for guidance is long overdue. The current prenatal care paradigm focuses on numerous/intensive prenatal visits but only a single postpartum visit. Many women report feeling ill-equipped to navigate the challenges that arise post delivery, including self-care, newborn care, and breastfeeding. In addition, most women experience the impact of dramatic hormonal fluctuations resulting in mood alterations. Add profound sleep deprivation to this and the result is often some degree of postpartum blues and/or depression. The importance of anticipatory guidance for our patients cannot be underestimated. Helping to facilitate potential social support structures be in place prior to the birth is optimal. Providers should reinforce the importance of access to a variety of tools, including digital apps, community support groups, and 24/7 web access services. Moving forward, it should be considered unconscionable to send a new mother home without ensuring the appropriate resources are in place. Postpartum care should be tailored to a woman’s needs and may require multiple visits and/or referrals.”
Dr. Stuebe and colleagues, as well as Dr. Bianco, disclosed receiving honoraria for various projects. Funding for the study was supported by the Alliance for Innovation on Maternal and Child Health, which is funded by a grant from the Health Resources and Services Administration.
Health care professionals miss an important opportunity if they do not use a holistic patient-centered approach in providing support to their postpartum patients, advised Alison M. Stuebe, MD, MSc, of the division of maternal-fetal medicine at the University of North Carolina at Chapel Hill, and colleagues.
Too often, the emphasis for health care professionals is on less common, highly morbid complications, while their patients’ needs are focused more on daily functioning.
More than half of maternal deaths occur not during delivery, but in the days, weeks, and months following birth. Many serious complications and management of less-emergent conditions such as flu-like symptoms go unnoticed or result in treatment delays. Routinely asking women who present for care whether they have been pregnant during the past year could go a long way in helping clinicians connect the dots for conditions they might not otherwise consider, suggested Dr. Stuebe and colleagues.
In response to what is becoming a growing burden of postpartum maternal morbidity and mortality in the United States, Dr. Stuebe and associates, in coordination with the Alliance of Innovation in Maternal Health, prepared a comprehensive consensus statement on postpartum care basics. The work came out of a day-long planning meeting of an interdisciplinary work group funded by AIMH to develop the maternal safety bundle, which established 28 goals across four phases – readiness, recognition and prevention, response, and reporting. Each phase is intended to occur as part of a coordinated effort between women experiencing pregnancy and their health care providers, with involvement from multiple clinical settings and health systems.
America needs to change the way it treats its new mothers
In a separate interview, Dr. Stuebe shared additional insights into the development of the consensus statement: “This article is part of a broader movement to change the way America treats new mothers. For too long, we’ve lavished attention on women in the weeks before birth and then left them alone to manage recovery, plunging hormones, sleepless nights, and new baby care. The goal of this article is to set out the ways that we can better support women in this critical transition period.
“We need to start during pregnancy, by working with women to plan for the fourth trimester and identify a care team that will support her and her baby. After birth, we need to adapt those plans and make sure she has the emotional and material support she needs to navigate the coming months. Many of these decisions are deeply personal, and we need to center the values and preferences of the woman and her family.”
Dr. Stuebe also spotlighted a new University of North Carolina–based program designed to support shared decision-making. Their Trimester Project team partnered with mothers to design NewMomHealth.com, the first national postpartum resource designed by and for new mothers, which is in the process of launching a culturally adapted Spanish-language site, saludmadre.com, in partnership with Urban Strategies.
Readiness prepares mothers, HCPs, and clinical settings for the fourth trimester
In the first phase, Readiness, there are three key parties involved: the woman experiencing pregnancy, the health care providers (HCPs) supporting her, and the clinical settings involved in her care. Each player has specific goals to achieve in this phase, women are responsible for four goals, health care providers have two goals, and clinical settings have five.
Prenatally, every woman should work with health care providers to develop a comprehensive, individualized postpartum care plan that designates a postpartum clinical “home” for addressing any care needed between birth and the “comprehensive postpartum visit.” The plan encourages the patient and her health care team give attention to social support, birth recovery, infant care and feeding plans, thoughts concerning future pregnancies, and specifically use of contraception, as well as any chronic health concerns and overall emotional well-being.
The plan should consist of a postpartum care team that includes friends and family and ensures that medical, material, and social support are available in the weeks following birth for both mother and baby.
The authors emphasized the importance for clinicians to develop a keen awareness of “reproductive justice,” which respects a woman’s right “to maintain personal bodily autonomy, have children, not have children, and parent the children we have in safe and sustainable communities.” They encouraged adopting a sensitivity to concerns deeply rooted in the history of family planning, which is perceived to have been “fraught by coercive tactics to incentivize sterilization and contraceptive implants among marginalized groups.” With this in mind, they urged clinicians to respectfully ask about the patient’s intentions before making any suggestions concerning future births and family planning preferences.
HCP readiness ensures that each woman “has a documented postpartum care plan and care team identified in the prenatal period.” HCPs are also responsible for developing and maintaining “a working knowledge of evidence-based evaluation and management of common issues facing the mother-infant dyad.”
Clinical setting readiness ensures that woman-centered postpartum care and education have been developed and optimized using adult learning principles whenever possible. Diversity of family structures, cultural traditions, and parenting practices are fully embraced. Dr. Stuebe and colleagues emphasized that traditional top-down teaching methods are ineffective and should be avoided.
Clinical setting readiness also ensures that systems are developed to pair families with community-based resources that provide medical follow-up as well as social and material support. Clinical protocols and reimbursement options should be available that give women easy access to preferred contraception. Systems should be in place that facilitate prompt, pertinent communication between in- and outpatient providers. Lastly, and perhaps most importantly, clinical setting readiness demands the development of protocols to screen and treat key postpartum concerns, such as depression, substance use disorders, family violence, and even incontinence in coordination with locally identified specialists.
Recognition and prevention promotes accountability and establishes key guidelines
Recognition and prevention require coordinated participation between women experiencing pregnancy and the clinical environments supporting their care. In this phase, women are responsible for three goals; clinical environments have four goals.
Women are to be identified and respected as the expert most knowledgeable of their own needs, cautioned the authors. They should also be empowered to trust their instincts, seeking care as early and as often as needed in the weeks after they give birth. Postpartum care needs to be viewed as an ongoing process, not just a singular encounter.
Women also need to take ownership for their postpartum care plan, reviewing and revising it as needed in coordination with their health care providers before maternity discharge is complete. Every plan should include a comprehensive list of warning signs and recommended action plans when faced with life-threatening complications, a list of resources for lactation support, and a “first-call” phone number that identifies the postpartum medical providers available to address breastfeeding issues and for postpartum care visits, including prescheduled dates and times.
Lastly, women are to take responsibility for attending their postpartum visit. Although new guidance suggests that the comprehensive visit take place no later than 12 weeks post partum, it also recognizes multiple visits may be required to address individual needs. All women should have contact with a maternal care provider within 3 weeks post partum, the American College of Obstetricians and Gynecologists recommends.
Responsibility for recognition and prevention in the clinical setting begins with determining guidelines for early postpartum visits, considering chronic conditions such as hypertension, diabetes, or substance abuse disorder and risk for postpartum conditions such as wound complications or postpartum depression, the authors wrote. Ongoing care between inpatient and outpatient settings as well as between maternal and infant care providers is coordinated to ensure health and well-being of both patients. Screening and treatment of common comorbidities such as mental health issues, smoking, and substance use as well as issues related to unstable housing and food insecurities are also addressed in the clinical environment. Lastly, clinicians are tasked with ensuring that every patient has selected a primary care provider for ongoing care.
Response ensures key parties and resources are connected at every step
In the response phase, clinical settings and health care providers are the key participants and they have two goals each.
Every clinical setting is tasked with implementing treatment protocols and providing needed care or facilitating referral in a timely fashion. The importance of a “warm hand-off” and “face-to-face introduction to the specialist to whom the patient is being referred” is encouraged. They are also responsible for keeping an updated inventory of community resources on hand for such unmet needs as 24-hour hotlines, food banks, diaper banks, lactation support groups, and home-visiting programs.
Every health care provider is tasked with developing strategies designed to reach women who do not attend their comprehensive postpartum visit. They are also responsible for assessing the severity of identified needs, and arranging immediate transportation to appropriate facilities in life-threatening circumstances. In nonacute cases, the woman and her care providers work with her to make appropriate decisions.
Reporting gives health systems the opportunity to assess and improve
Sole responsibility in the reporting phase falls to every health system. In this phase, they have a total of six goals.
Health systems are ideally organized to convene strategy-sharing sessions with inpatient as well as outpatient professionals to evaluate successes and opportunities for improvement. They are equally qualified to identify and monitor such quality measures as postpartum emergency department utilization and readmission rates. They are tasked with working toward quality metrics that compare postpartum outcomes and prenatal intentions, including patient receipt of preferred contraception or completion of planned breastfeeding duration.
Health systems are expected to conduct quality improvement measures designed to reduce postpartum morbidity when preventable. They are also the logical choice for maintaining a clearinghouse for resources, in collaboration with the community, that is designed to meet the postpartum needs of women.
Lastly, they play an important role in ensuring that reimbursement policies are structured such that they do not disincentivize postpartum visits.
The consensus bundle encourages change in the way America treats its new mothers
Angela Bianco, MD, maternal and fetal medicine specialist at Mount Sinai Hospital, New York, also interviewed separately, noted: “I think the fact that ACOG has created a postpartum bundle for providers to use for guidance is long overdue. The current prenatal care paradigm focuses on numerous/intensive prenatal visits but only a single postpartum visit. Many women report feeling ill-equipped to navigate the challenges that arise post delivery, including self-care, newborn care, and breastfeeding. In addition, most women experience the impact of dramatic hormonal fluctuations resulting in mood alterations. Add profound sleep deprivation to this and the result is often some degree of postpartum blues and/or depression. The importance of anticipatory guidance for our patients cannot be underestimated. Helping to facilitate potential social support structures be in place prior to the birth is optimal. Providers should reinforce the importance of access to a variety of tools, including digital apps, community support groups, and 24/7 web access services. Moving forward, it should be considered unconscionable to send a new mother home without ensuring the appropriate resources are in place. Postpartum care should be tailored to a woman’s needs and may require multiple visits and/or referrals.”
Dr. Stuebe and colleagues, as well as Dr. Bianco, disclosed receiving honoraria for various projects. Funding for the study was supported by the Alliance for Innovation on Maternal and Child Health, which is funded by a grant from the Health Resources and Services Administration.
FROM OBSTETRICS AND GYNECOLOGY
Black women show heightened risk for depression after early pregnancy loss
Black women are significantly more likely than non-Black women to develop major depression within a month of early pregnancy loss, based on data from a secondary analysis of 300 women.
Approximately 25% of women experience a pregnancy loss, and many of these women are at increased risk for psychological problems including major depression, wrote Jade M. Shorter, MD, of Stanford (Calif.) University, and colleagues.
Data from previous studies show that Black women experience higher rates of perinatal depression, compared with other racial groups, and that stress and adverse childhood experiences also are higher among Black individuals, they noted.
“Based on data showing higher rates of pregnancy loss, perinatal depression, and perceived stress in Black women, we hypothesized that the odds of having risk for major depression or high perceived stress 30 days after miscarriage treatment would be higher in Black participants when compared with non-Black participants,” they wrote.
In a study published in Obstetrics & Gynecology, the researchers conducted a secondary analysis of 300 women aged 18 years and older with nonviable intrauterine pregnancy between 5 and 12 weeks’ gestation who were part of a larger randomized trial conducted between May 2014 and April 2017. The women were randomized to medical treatment of either mifepristone 200 mg orally plus misoprostol 800 mcg vaginally after 24 hours or the usual treatment of misoprostol 800 mcg vaginally.
Depression was assessed using the Center for Epidemiological Studies–Depression scale, Perceived Stress Scale, and Adverse Childhood Experience scale. Adverse childhood experience data were collected at baseline; stress and depression data were collected at baseline and at 30 days after treatment.
A total of 120 participants self-identified as Black and 155 self-identified as non-Black.
Depression risk doubles in Black women
At 30 days after treatment for early pregnancy loss, 24% of women met criteria for major depression, including 57% of Black women and 43% of non-Black women. The odds of depression were twice as high among Black women, compared with non-Black women (odds ratio 2.02), and Black women were more likely to be younger, have lower levels of education, and have public insurance, compared with non-Black women.
The association between Black race and increased risk for depression at 30 days after treatment persisted after controlling for factors including parity, baseline depression, and adverse childhood experiences, the researchers noted.
The study findings were limited by several factors, including the potential for different depression risk in those from the original study who did and did not participate in the secondary analysis and by the use of the original Adverse Childhood Experience survey, which may not reflect the range of adversity faced by different demographic groups, the researchers noted. However, the results were strengthened by the collection of 30-day outcome data in the clinical setting and by the diverse study population.
“These findings should be not be used to stigmatize Black women; instead, it is important to consider the complex systemic factors, such as structural racism, that are the root causes of disparate health outcomes,” and to support appropriate mental health resources and interventions for all women who experience early pregnancy loss, the researchers emphasized.
Recognize risks, reduce barriers
“Early pregnancy loss is unfortunately a common event that affects 15%-20% of pregnancies,” Iris Krishna, MD, of Emory University, Atlanta, said in an interview.
However, “the mental health impact of early pregnancy loss is understudied, and as a result mental health disorders often go unnoticed and untreated,” she said.
Growing evidence shows that Black women in particular are at greater risk for chronic stressors that affect their overall health. “Black women are more likely to be exposed to trauma in their lifetime, such as physical and emotional abuse, neglect, and household instability, all of which predispose women to mental health disorders such as depression. Untreated maternal depression has an impact on future pregnancy outcomes such as increasing the risk of having a preterm delivery and/or delivering a low-birth-weight baby, outcomes where Black women are at disproportionately high risk in comparison to non-Black women,” Dr. Krishna said.
“This study found that the risk for depression after an early pregnancy loss is twice as high for Black women in comparison to non-Black women. The findings of this study further underscore the fact that Black women are at disproportionate high risk for poor maternal and pregnancy outcomes,” Dr. Krishna added.
“Structural racism is a major barrier to caring for the health of Black women. To care for the health of Black women we must overcome racial and ethnic disparities. Addressing disparities involves a multitiered approach, including identifying and addressing implicit bias in health care and improving access to health care for women of color,” she said.
“Additional research is needed in identifying at-risk women and mental health interventions that can improve the mental well-being of women after adverse pregnancy outcomes such as early pregnancy loss,” Dr. Krishna concluded.
The study was supported by the Society of Family Planning Research Fund. Lead author Dr. Shorter had no financial conflicts to disclose. Dr. Krishna had no financial conflicts to disclose.
SOURCE: Shorter JM et al. Obstet Gynecol. 2020 Dec 3. doi: 10.1097/AOG.0000000000004212.
Black women are significantly more likely than non-Black women to develop major depression within a month of early pregnancy loss, based on data from a secondary analysis of 300 women.
Approximately 25% of women experience a pregnancy loss, and many of these women are at increased risk for psychological problems including major depression, wrote Jade M. Shorter, MD, of Stanford (Calif.) University, and colleagues.
Data from previous studies show that Black women experience higher rates of perinatal depression, compared with other racial groups, and that stress and adverse childhood experiences also are higher among Black individuals, they noted.
“Based on data showing higher rates of pregnancy loss, perinatal depression, and perceived stress in Black women, we hypothesized that the odds of having risk for major depression or high perceived stress 30 days after miscarriage treatment would be higher in Black participants when compared with non-Black participants,” they wrote.
In a study published in Obstetrics & Gynecology, the researchers conducted a secondary analysis of 300 women aged 18 years and older with nonviable intrauterine pregnancy between 5 and 12 weeks’ gestation who were part of a larger randomized trial conducted between May 2014 and April 2017. The women were randomized to medical treatment of either mifepristone 200 mg orally plus misoprostol 800 mcg vaginally after 24 hours or the usual treatment of misoprostol 800 mcg vaginally.
Depression was assessed using the Center for Epidemiological Studies–Depression scale, Perceived Stress Scale, and Adverse Childhood Experience scale. Adverse childhood experience data were collected at baseline; stress and depression data were collected at baseline and at 30 days after treatment.
A total of 120 participants self-identified as Black and 155 self-identified as non-Black.
Depression risk doubles in Black women
At 30 days after treatment for early pregnancy loss, 24% of women met criteria for major depression, including 57% of Black women and 43% of non-Black women. The odds of depression were twice as high among Black women, compared with non-Black women (odds ratio 2.02), and Black women were more likely to be younger, have lower levels of education, and have public insurance, compared with non-Black women.
The association between Black race and increased risk for depression at 30 days after treatment persisted after controlling for factors including parity, baseline depression, and adverse childhood experiences, the researchers noted.
The study findings were limited by several factors, including the potential for different depression risk in those from the original study who did and did not participate in the secondary analysis and by the use of the original Adverse Childhood Experience survey, which may not reflect the range of adversity faced by different demographic groups, the researchers noted. However, the results were strengthened by the collection of 30-day outcome data in the clinical setting and by the diverse study population.
“These findings should be not be used to stigmatize Black women; instead, it is important to consider the complex systemic factors, such as structural racism, that are the root causes of disparate health outcomes,” and to support appropriate mental health resources and interventions for all women who experience early pregnancy loss, the researchers emphasized.
Recognize risks, reduce barriers
“Early pregnancy loss is unfortunately a common event that affects 15%-20% of pregnancies,” Iris Krishna, MD, of Emory University, Atlanta, said in an interview.
However, “the mental health impact of early pregnancy loss is understudied, and as a result mental health disorders often go unnoticed and untreated,” she said.
Growing evidence shows that Black women in particular are at greater risk for chronic stressors that affect their overall health. “Black women are more likely to be exposed to trauma in their lifetime, such as physical and emotional abuse, neglect, and household instability, all of which predispose women to mental health disorders such as depression. Untreated maternal depression has an impact on future pregnancy outcomes such as increasing the risk of having a preterm delivery and/or delivering a low-birth-weight baby, outcomes where Black women are at disproportionately high risk in comparison to non-Black women,” Dr. Krishna said.
“This study found that the risk for depression after an early pregnancy loss is twice as high for Black women in comparison to non-Black women. The findings of this study further underscore the fact that Black women are at disproportionate high risk for poor maternal and pregnancy outcomes,” Dr. Krishna added.
“Structural racism is a major barrier to caring for the health of Black women. To care for the health of Black women we must overcome racial and ethnic disparities. Addressing disparities involves a multitiered approach, including identifying and addressing implicit bias in health care and improving access to health care for women of color,” she said.
“Additional research is needed in identifying at-risk women and mental health interventions that can improve the mental well-being of women after adverse pregnancy outcomes such as early pregnancy loss,” Dr. Krishna concluded.
The study was supported by the Society of Family Planning Research Fund. Lead author Dr. Shorter had no financial conflicts to disclose. Dr. Krishna had no financial conflicts to disclose.
SOURCE: Shorter JM et al. Obstet Gynecol. 2020 Dec 3. doi: 10.1097/AOG.0000000000004212.
Black women are significantly more likely than non-Black women to develop major depression within a month of early pregnancy loss, based on data from a secondary analysis of 300 women.
Approximately 25% of women experience a pregnancy loss, and many of these women are at increased risk for psychological problems including major depression, wrote Jade M. Shorter, MD, of Stanford (Calif.) University, and colleagues.
Data from previous studies show that Black women experience higher rates of perinatal depression, compared with other racial groups, and that stress and adverse childhood experiences also are higher among Black individuals, they noted.
“Based on data showing higher rates of pregnancy loss, perinatal depression, and perceived stress in Black women, we hypothesized that the odds of having risk for major depression or high perceived stress 30 days after miscarriage treatment would be higher in Black participants when compared with non-Black participants,” they wrote.
In a study published in Obstetrics & Gynecology, the researchers conducted a secondary analysis of 300 women aged 18 years and older with nonviable intrauterine pregnancy between 5 and 12 weeks’ gestation who were part of a larger randomized trial conducted between May 2014 and April 2017. The women were randomized to medical treatment of either mifepristone 200 mg orally plus misoprostol 800 mcg vaginally after 24 hours or the usual treatment of misoprostol 800 mcg vaginally.
Depression was assessed using the Center for Epidemiological Studies–Depression scale, Perceived Stress Scale, and Adverse Childhood Experience scale. Adverse childhood experience data were collected at baseline; stress and depression data were collected at baseline and at 30 days after treatment.
A total of 120 participants self-identified as Black and 155 self-identified as non-Black.
Depression risk doubles in Black women
At 30 days after treatment for early pregnancy loss, 24% of women met criteria for major depression, including 57% of Black women and 43% of non-Black women. The odds of depression were twice as high among Black women, compared with non-Black women (odds ratio 2.02), and Black women were more likely to be younger, have lower levels of education, and have public insurance, compared with non-Black women.
The association between Black race and increased risk for depression at 30 days after treatment persisted after controlling for factors including parity, baseline depression, and adverse childhood experiences, the researchers noted.
The study findings were limited by several factors, including the potential for different depression risk in those from the original study who did and did not participate in the secondary analysis and by the use of the original Adverse Childhood Experience survey, which may not reflect the range of adversity faced by different demographic groups, the researchers noted. However, the results were strengthened by the collection of 30-day outcome data in the clinical setting and by the diverse study population.
“These findings should be not be used to stigmatize Black women; instead, it is important to consider the complex systemic factors, such as structural racism, that are the root causes of disparate health outcomes,” and to support appropriate mental health resources and interventions for all women who experience early pregnancy loss, the researchers emphasized.
Recognize risks, reduce barriers
“Early pregnancy loss is unfortunately a common event that affects 15%-20% of pregnancies,” Iris Krishna, MD, of Emory University, Atlanta, said in an interview.
However, “the mental health impact of early pregnancy loss is understudied, and as a result mental health disorders often go unnoticed and untreated,” she said.
Growing evidence shows that Black women in particular are at greater risk for chronic stressors that affect their overall health. “Black women are more likely to be exposed to trauma in their lifetime, such as physical and emotional abuse, neglect, and household instability, all of which predispose women to mental health disorders such as depression. Untreated maternal depression has an impact on future pregnancy outcomes such as increasing the risk of having a preterm delivery and/or delivering a low-birth-weight baby, outcomes where Black women are at disproportionately high risk in comparison to non-Black women,” Dr. Krishna said.
“This study found that the risk for depression after an early pregnancy loss is twice as high for Black women in comparison to non-Black women. The findings of this study further underscore the fact that Black women are at disproportionate high risk for poor maternal and pregnancy outcomes,” Dr. Krishna added.
“Structural racism is a major barrier to caring for the health of Black women. To care for the health of Black women we must overcome racial and ethnic disparities. Addressing disparities involves a multitiered approach, including identifying and addressing implicit bias in health care and improving access to health care for women of color,” she said.
“Additional research is needed in identifying at-risk women and mental health interventions that can improve the mental well-being of women after adverse pregnancy outcomes such as early pregnancy loss,” Dr. Krishna concluded.
The study was supported by the Society of Family Planning Research Fund. Lead author Dr. Shorter had no financial conflicts to disclose. Dr. Krishna had no financial conflicts to disclose.
SOURCE: Shorter JM et al. Obstet Gynecol. 2020 Dec 3. doi: 10.1097/AOG.0000000000004212.
FROM OBSTETRICS & GYNECOLOGY
Outpatient penicillin allergy testing found safe in pregnancy
Successful outpatient penicillin allergy testing with a low incidence of anaphylaxis during pregnancy demonstrates the feasibility of performing allergy testing in the outpatient setting, reported Nerlyne Desravines, MD, of the University of North Carolina, Chapel Hill, and colleagues.
In a prospective cohort study of 74 pregnant patients with previous self reports of penicillin allergy, Dr. Desravines and colleagues sought to determine the feasibility, acceptability, and safety of performing penicillin allergy testing in an outpatient setting. Patients included in the study were aged 18-55 years with gestational age between 14 and 36 weeks and planned delivery within the University of North Carolina heath care system receiving care between March 2019 and March 2020.
Of the 74 women enrolled to participate, 24 failed to present for testing, including some citing scheduling conflicts or fear of adverse reactions. Only 46 of the remaining 50 successfully completed testing; 4 patients were scheduled for testing but unable to participate because of COVID-19 restrictions.
Insurance status may affect participation in testing
Those who had public insurance were less likely to complete testing; those who completed testing were significantly more likely to be married and carry private insurance.
Fully 52% of the 46 women who completed testing were in the second trimester. The majority (85%) experienced their initial penicillin allergy reaction more than 10 years earlier.
Ultimately, 43 of the 46 women (93%) received a negative test result despite previous self reports of severe allergic reaction. Two of the three confirmed with penicillin allergy failed the 10% oral drug challenge; the other tested positive for penicillin G on intradermal testing. The two women who were found to have severe penicillin allergy experienced coughing, chest tightening, and skin and oropharynx pruritus within 30 minutes after their 10% amoxicillin drug challenge; they also experienced vomiting at 1 and 2 hours post ingestion. Following intramuscular injection of epinephrine, oral cetirizine with periodic vital sign measures, and albuterol updraft in one patient with a history of well controlled asthma, symptom resolution was achieved and both women were discharged without the need for further care.
The systemic reactions observed in just 4% of the study population is lower than normally reported in the general population, suggesting that the study sample size may underestimate the actual prevalence of systemic reactions, the authors noted. “The primary factor in safely conducting allergy testing in pregnancy is an outpatient facility that is appropriately outfitted with trained personnel and medications for possible serious reactions,” they added.
Noteworthy is the allergy testing protocol used by Dr. Desravines and colleagues in this study. Their graded oral drug challenge has not been used in previous studies of outpatient penicillin testing in pregnancy. Two of the three participants with positive test results had penicillin allergy confirmed following reaction to the first step (10% dose) of oral challenge to amoxicillin.
Prevalence of systemic reactions may be higher than expected
The authors cited ease of implementation in an obstetrics or allergy clinic as a strength of the study. One limitation is the observed rate of systemic reaction. The wide confidence interval observed indicates the rates of anaphylaxis may actually be as high as 15%, suggested the authors. The small sample size also limits the safety analysis for rare outcomes such as death.
Patient-reported barriers included time commitment for the testing visit. Rural women or those receiving prenatal care from health departments or community health centers were not able to be enrolled. Only one Spanish-speaking woman participated despite availability of bilingual staff and interpreters.
Such outpatient testing for those at greatest risk offers the opportunity to mitigate emerging drug resistance and should ideally take place preconception or at the time of initial allergic reaction, the authors advised. As emphasized in the latest Committee Opinion issued by the American College of Obstetricians and Gynecologists, obstetricians have a real opportunity to counsel patients preconception and postpartum regarding the benefits of penicillin allergy testing.
In a separate interview, Angela Martin, MD, assistant professor, maternal-fetal medicine, at University of Kansas, Kansas City, noted the large clinical implications of this study given that more than 90% of women undergoing allergy testing following self-reported penicillin allergy had a negative test result. “By performing allergy testing on appropriate candidates, as these authors have done, clinicians can treat infections and implement group B streptococcus prophylaxis with the narrowest spectrum antibiotic. This has potential to combat antibiotic resistance and may protect patients from harms caused by unnecessary broad-spectrum antibiotic use during pregnancy and beyond,” said Dr. Martin.
“It should be mentioned that 2 out of the 46 women tested (4%) had an anaphylactic reaction. This highlights the need to perform allergy testing in a qualified center capable of managing acute anaphylactic reactions should they occur,” she advised.
Dr. Desravines and colleagues, as well as Dr. Martin, had no conflicts of interest and no relevant financial disclosures.
SOURCE: Obstet Gynecol. 2021;137:56-61. doi: 10.1097/AOG.0000000000004213.
Successful outpatient penicillin allergy testing with a low incidence of anaphylaxis during pregnancy demonstrates the feasibility of performing allergy testing in the outpatient setting, reported Nerlyne Desravines, MD, of the University of North Carolina, Chapel Hill, and colleagues.
In a prospective cohort study of 74 pregnant patients with previous self reports of penicillin allergy, Dr. Desravines and colleagues sought to determine the feasibility, acceptability, and safety of performing penicillin allergy testing in an outpatient setting. Patients included in the study were aged 18-55 years with gestational age between 14 and 36 weeks and planned delivery within the University of North Carolina heath care system receiving care between March 2019 and March 2020.
Of the 74 women enrolled to participate, 24 failed to present for testing, including some citing scheduling conflicts or fear of adverse reactions. Only 46 of the remaining 50 successfully completed testing; 4 patients were scheduled for testing but unable to participate because of COVID-19 restrictions.
Insurance status may affect participation in testing
Those who had public insurance were less likely to complete testing; those who completed testing were significantly more likely to be married and carry private insurance.
Fully 52% of the 46 women who completed testing were in the second trimester. The majority (85%) experienced their initial penicillin allergy reaction more than 10 years earlier.
Ultimately, 43 of the 46 women (93%) received a negative test result despite previous self reports of severe allergic reaction. Two of the three confirmed with penicillin allergy failed the 10% oral drug challenge; the other tested positive for penicillin G on intradermal testing. The two women who were found to have severe penicillin allergy experienced coughing, chest tightening, and skin and oropharynx pruritus within 30 minutes after their 10% amoxicillin drug challenge; they also experienced vomiting at 1 and 2 hours post ingestion. Following intramuscular injection of epinephrine, oral cetirizine with periodic vital sign measures, and albuterol updraft in one patient with a history of well controlled asthma, symptom resolution was achieved and both women were discharged without the need for further care.
The systemic reactions observed in just 4% of the study population is lower than normally reported in the general population, suggesting that the study sample size may underestimate the actual prevalence of systemic reactions, the authors noted. “The primary factor in safely conducting allergy testing in pregnancy is an outpatient facility that is appropriately outfitted with trained personnel and medications for possible serious reactions,” they added.
Noteworthy is the allergy testing protocol used by Dr. Desravines and colleagues in this study. Their graded oral drug challenge has not been used in previous studies of outpatient penicillin testing in pregnancy. Two of the three participants with positive test results had penicillin allergy confirmed following reaction to the first step (10% dose) of oral challenge to amoxicillin.
Prevalence of systemic reactions may be higher than expected
The authors cited ease of implementation in an obstetrics or allergy clinic as a strength of the study. One limitation is the observed rate of systemic reaction. The wide confidence interval observed indicates the rates of anaphylaxis may actually be as high as 15%, suggested the authors. The small sample size also limits the safety analysis for rare outcomes such as death.
Patient-reported barriers included time commitment for the testing visit. Rural women or those receiving prenatal care from health departments or community health centers were not able to be enrolled. Only one Spanish-speaking woman participated despite availability of bilingual staff and interpreters.
Such outpatient testing for those at greatest risk offers the opportunity to mitigate emerging drug resistance and should ideally take place preconception or at the time of initial allergic reaction, the authors advised. As emphasized in the latest Committee Opinion issued by the American College of Obstetricians and Gynecologists, obstetricians have a real opportunity to counsel patients preconception and postpartum regarding the benefits of penicillin allergy testing.
In a separate interview, Angela Martin, MD, assistant professor, maternal-fetal medicine, at University of Kansas, Kansas City, noted the large clinical implications of this study given that more than 90% of women undergoing allergy testing following self-reported penicillin allergy had a negative test result. “By performing allergy testing on appropriate candidates, as these authors have done, clinicians can treat infections and implement group B streptococcus prophylaxis with the narrowest spectrum antibiotic. This has potential to combat antibiotic resistance and may protect patients from harms caused by unnecessary broad-spectrum antibiotic use during pregnancy and beyond,” said Dr. Martin.
“It should be mentioned that 2 out of the 46 women tested (4%) had an anaphylactic reaction. This highlights the need to perform allergy testing in a qualified center capable of managing acute anaphylactic reactions should they occur,” she advised.
Dr. Desravines and colleagues, as well as Dr. Martin, had no conflicts of interest and no relevant financial disclosures.
SOURCE: Obstet Gynecol. 2021;137:56-61. doi: 10.1097/AOG.0000000000004213.
Successful outpatient penicillin allergy testing with a low incidence of anaphylaxis during pregnancy demonstrates the feasibility of performing allergy testing in the outpatient setting, reported Nerlyne Desravines, MD, of the University of North Carolina, Chapel Hill, and colleagues.
In a prospective cohort study of 74 pregnant patients with previous self reports of penicillin allergy, Dr. Desravines and colleagues sought to determine the feasibility, acceptability, and safety of performing penicillin allergy testing in an outpatient setting. Patients included in the study were aged 18-55 years with gestational age between 14 and 36 weeks and planned delivery within the University of North Carolina heath care system receiving care between March 2019 and March 2020.
Of the 74 women enrolled to participate, 24 failed to present for testing, including some citing scheduling conflicts or fear of adverse reactions. Only 46 of the remaining 50 successfully completed testing; 4 patients were scheduled for testing but unable to participate because of COVID-19 restrictions.
Insurance status may affect participation in testing
Those who had public insurance were less likely to complete testing; those who completed testing were significantly more likely to be married and carry private insurance.
Fully 52% of the 46 women who completed testing were in the second trimester. The majority (85%) experienced their initial penicillin allergy reaction more than 10 years earlier.
Ultimately, 43 of the 46 women (93%) received a negative test result despite previous self reports of severe allergic reaction. Two of the three confirmed with penicillin allergy failed the 10% oral drug challenge; the other tested positive for penicillin G on intradermal testing. The two women who were found to have severe penicillin allergy experienced coughing, chest tightening, and skin and oropharynx pruritus within 30 minutes after their 10% amoxicillin drug challenge; they also experienced vomiting at 1 and 2 hours post ingestion. Following intramuscular injection of epinephrine, oral cetirizine with periodic vital sign measures, and albuterol updraft in one patient with a history of well controlled asthma, symptom resolution was achieved and both women were discharged without the need for further care.
The systemic reactions observed in just 4% of the study population is lower than normally reported in the general population, suggesting that the study sample size may underestimate the actual prevalence of systemic reactions, the authors noted. “The primary factor in safely conducting allergy testing in pregnancy is an outpatient facility that is appropriately outfitted with trained personnel and medications for possible serious reactions,” they added.
Noteworthy is the allergy testing protocol used by Dr. Desravines and colleagues in this study. Their graded oral drug challenge has not been used in previous studies of outpatient penicillin testing in pregnancy. Two of the three participants with positive test results had penicillin allergy confirmed following reaction to the first step (10% dose) of oral challenge to amoxicillin.
Prevalence of systemic reactions may be higher than expected
The authors cited ease of implementation in an obstetrics or allergy clinic as a strength of the study. One limitation is the observed rate of systemic reaction. The wide confidence interval observed indicates the rates of anaphylaxis may actually be as high as 15%, suggested the authors. The small sample size also limits the safety analysis for rare outcomes such as death.
Patient-reported barriers included time commitment for the testing visit. Rural women or those receiving prenatal care from health departments or community health centers were not able to be enrolled. Only one Spanish-speaking woman participated despite availability of bilingual staff and interpreters.
Such outpatient testing for those at greatest risk offers the opportunity to mitigate emerging drug resistance and should ideally take place preconception or at the time of initial allergic reaction, the authors advised. As emphasized in the latest Committee Opinion issued by the American College of Obstetricians and Gynecologists, obstetricians have a real opportunity to counsel patients preconception and postpartum regarding the benefits of penicillin allergy testing.
In a separate interview, Angela Martin, MD, assistant professor, maternal-fetal medicine, at University of Kansas, Kansas City, noted the large clinical implications of this study given that more than 90% of women undergoing allergy testing following self-reported penicillin allergy had a negative test result. “By performing allergy testing on appropriate candidates, as these authors have done, clinicians can treat infections and implement group B streptococcus prophylaxis with the narrowest spectrum antibiotic. This has potential to combat antibiotic resistance and may protect patients from harms caused by unnecessary broad-spectrum antibiotic use during pregnancy and beyond,” said Dr. Martin.
“It should be mentioned that 2 out of the 46 women tested (4%) had an anaphylactic reaction. This highlights the need to perform allergy testing in a qualified center capable of managing acute anaphylactic reactions should they occur,” she advised.
Dr. Desravines and colleagues, as well as Dr. Martin, had no conflicts of interest and no relevant financial disclosures.
SOURCE: Obstet Gynecol. 2021;137:56-61. doi: 10.1097/AOG.0000000000004213.
FROM OBSTETRICS & GYNECOLOGY