User login
Is your patient’s dizziness psychogenic?
Dizziness is common among patients age 65 and older, and more than one-third have a psychiatric disorder that is caused by or is causing their dizziness.1
When older patients present with dizziness, psychiatrists may be asked to alleviate the psychological symptoms and help identify the underlying disease state.2
More than 60 medical and psychiatric disorders and many medications can cause dizziness. To help you sort through the possibilities, we offer:
- six diagnostic questions to rule out underlying medical problems
- lists of commonly used psychotropics and other drugs that may cause dizziness
- advice on treating depression, anxiety, and panic disorder in an older patient with dizziness while avoiding side effects and drug interactions.
Table 1
Four types of dizziness and their usual causes
| Vertigo Benign positional vertigo CNS cause—tumor, demyelination, neurodegenerative disorders Labyrinthitis Meniere’s disease Peripheral vestibulopathy (in 50% of cases) Vestibular neuronitis |
| Presyncope Arrhythmias Carotid sinus disease Hypoglycemia Neurocardiogenic syncope Organic heart disease Orthostatic hypotension Seizures Situational Transient ischemic attacks |
| Disequilibrium Balance and gait disorder Mixed CNS diseases (ischemic, degenerative) Neurodegenerative CNS disorders Presbystasis Sensorimotor dysfunction |
| Psychogenic lightheadedness Agoraphobia Anxiety Depression Panic disorder Hyperventilation |
| Source: Adapted from reference 6 |
Many causes of dizziness
The term “dizziness” is hard to define because of its nonspecific and variable symptom description, multiple causes, and lack of clear diagnostic and management guidelines. In clinical use, dizziness encompasses abnormal sensations relating to perception of the body’s relationship to space.
Some researchers believe dizziness is a distinct geriatric syndrome because numerous factors related to aging cause dizziness,2 including physiologic changes (presbystasis), accumulated impairment, disease states, and interactions between multiple medications.
Anxiety, somatization, panic disorder, and depression cause dizziness in the elderly, as do:
- peripheral vestibular disorders
- brainstem cerebrovascular accident
- diabetes mellitus
- neurologic disorders such as Parkinson’s disease
- and cardiovascular disorders.
Selective serotonin reuptake inhibitors (SSRIs) and tricyclic antidepressants also have been shown to cause dizziness, as have numerous nonpsychotropic agents.
Recognizing patterns, testing hypotheses, and extending the diagnostic process over time can help you differentiate psychogenic from medicationinduced or neurologic dizziness.3 Because the presentation is so complex and the differential diagnosis so broad, algorithmic diagnosis is less effective than a flexible clinical approach that allows for uncertainty in evaluating initial symptoms.
Determining the cause
A thorough patient history and physical examination can uncover a cause of dizziness in 75% of cases.4 Look for duration of dizziness symptoms; history of heart disease, diabetes or other illnesses; family history of psychiatric disorders; and other illnesses among family members.
Ask the following six complaint-specific questions to help you narrow the differential diagnosis and rule out nonpsychiatric causes.5
1. WHAT TYPE OF DIZZINESS DOES THE PATIENT HAVE?
Four categories—vertigo, presyncope, disequilibrium, and lightheadedness—are used to classify dizziness (Table 1).6
Vertigo is a sense that the body or environment is Patients may feel as if the floor is tilting, sinking, rising or veering sideways, or they may feel pulled to one side.
Vertigo is commonly caused by peripheral vestibular disorders—including benign positional vertigo, Meniere’s disease, labyrinthitis, and vestibular neuronitis—and central vestibular disorders associated with cerebrovascular disease, tumors, demyelinating diseases, migraines, seizures, multiple sclerosis and other CNS diseases. Acute-onset vertigo and neurologic signs suggest brainstem infarction.
Nystagmus is usually present, horizontal, and may be rotational at times. A vertical-beating nystagmus points to a probable CNS cause and requires urgent neuroimaging and referral to a neurologist or otolaryngologist.
Presyncope describes near-fainting. A dimming of vision and roaring in the ears may precede presyncope.
Depending on its cause, presyncope may occur regardless of position or only when upright. Common causes include orthostatic hypotension, neurocardiogenic syncope, organic heart disease, arrhythmias, carotid sinus disease, seizures, hypoglycemia, and transient ischemic attacks.
Abrupt presyncopal attacks that occur regardless of position suggest a cardiovascular cause. If onset is gradual and not improved by lying down, suspect a cerebral metabolic cause such as hypoglycemia.
Syncope, like presyncope, often is traced to an underlying cardiovascular disease. Dizziness and syncope often coexist, and both can be multifactorial. Dizziness may precede or follow syncopal episodes.
Differentiating syncope and dizziness is important because many underlying causes of syncope can be fatal. By contrast, dizziness symptoms are usually benign and self-limiting.7
A thorough history is critical to distinguishing dizziness from presyncope. Assess medication effects—especially CNS-acting medications, cardiovascular drugs, antihypertensives, antibiotics, and over-the-counter medications such as dextromethorphan and acetaminophen compounds. Also check for dehydration.
Disequilibrium disorder signifies unsteadiness or a loss of balance primarily involving the lower extremities. Symptoms are evoked by walking or standing and relieved by sitting or lying down. Gait is abnormal and balance is compromised without abnormal head sensations.
Common causes include balance and gait disorders, sensorimotor dysfunction, presbystasis, neurodegenerative CNS disorders, and mixed ischemic and degenerative CNS diseases.
Vague lightheadedness is often associated with somatic symptoms such as headache. Some patients describe a floating sensation.
Lightheadedness is frequently associated with anxiety, panic disorder, depression, and somatization. Hyperventilation and agoraphobia are other common causes.
Multiple symptoms, multiple types. Classifying an older patient’s dizziness can be challenging because many patients report symptoms that suggest two or more subtypes.2 Also, patients often have trouble describing their dizziness symptoms, sometimes using terms such as “giddiness,” “wooziness,” or “confusion.”
To help patients explain dizziness symptoms more accurately, ask specific questions such as:
- Do you at times feel like you’re about to faint?
- Do you feel as if the room is moving?
- Do you sometimes feel as though you’re going to fall?
Table 2
Psychotropics that may cause dizziness
| Anti-Alzheimer’s medications Memantine, rivastigmine, tacrine |
| Anticonvulsants Phenytoin |
| Antidepressants Monoamine oxidase inhibitors (phenelzine, selegiline) Selective serotonin reuptake inhibitors (all) Tricyclics (amitriptyline, imipramine, nortriptyline, trazodone) Others (bupropion, buspirone, mirtazapine, nefazodone, venlafaxine) |
| Antipsychotics Typicals (chlorpromazine, fluphenazine, perphenazine, prochlorperazine, thioridazine, trifluoperazine) Atypicals (all except olanzapine) |
| Anxiolytics Alprazolam, chlordiazepoxide, clonazepam, diazepam, lorazepam, oxazepam |
| Hypnotics Estazolam, flurazepam, quazepam, temazepam, triazolam, zolpidem |
| Mood stabilizers Carbamazepine, divalproex/valproic acid, gabapentin, lamotrigine, oxcarbazepine |
| Source: Clinical Pharmacology version 2.11. Tampa, FL: Gold Standard MultiMedia, 2004. |
2. HOW DO DIZZINESS SYMPTOMS RELATE TO POSITION OR MOTION?
By reproducing dizziness symptoms, some quick-maneuver tests can help patients describe their symptoms and may reveal a medical cause.
Dix-Hallpike maneuver.3 Move the patient rapidly from a seated to prone position with the head below the horizontal plane and turned 45 degrees for 10 seconds; then have the patient sit up. Repeat with the head turned to the other side. If dizziness does not occur within a few seconds after each test, rule out benign positional vertigo.
Seated head turn, or head-thrust test, measures qualitative vestibular function.8 Move the head rapidly by 45 degrees in a brief, small-amplitude thrust to one side while the patient focuses on your nose; this gauges vestibularocular control. Repeat the test in the other direction. A refixation corrective saccade, occurring as the patient tries to fixate on the target, indicates a possible vestibular disorder.
‘Get-Up and Go’ test, which takes less than 10 seconds, measures balance in older patents.9 Have the patient stand up, walk 10 feet, turn around, walk back, and sit down. Watch for staggering, unsteadiness, and use of hands to balance. Onset of symptoms suggests dizziness brought on during activities of daily living and provides information on how dizziness is affecting the patient’s ability to function.
Romberg test. Have the patient stand with heels together, first with eyes open and then closed. Vision and proprioceptive signals are used to compensate for vestibular loss. Thus, a balance disturbance with eyes closed suggests vestibular or spinal proprioceptive problems and may predict risk of falls caused by inability to compensate.8
3. WHAT IS THE COURSE OF DIZZINESS?
Differentiating acute, sudden-onset dizziness from chronic, gradual-onset dizziness can help uncover the problem’s cause and seriousness. The latter often has a psychological cause or may point to vestibular or minor cardiovascular problems. Tinetti et al2 identified anxiety or depressive symptoms as risk factors among community-based older persons who reported dizziness episodes lasting 1 month.
Table 3
Recommended SSRI starting dosages for older patients
| SSRI | Starting dosage (mg/d) | Maximum dosage (mg/d) |
|---|---|---|
| Citalopram | 10 to 20 | 30 |
| Escitalopram | 10 | 10 |
| Fluoxetine* | 5 to 10 | 60 |
| Paroxetine | 5 | 40 |
| Sertraline | 25 to 50 | 200 |
| * Most patients will not need more than 20 mg/d. Dosages 40 mg/d should be divided into twice-daily doses. | ||
| Source: Adapted from Reuben DB, Herr K, Pacala JT, et al. Geriatrics at your fingertips (5th ed). Malden, MA: Blackwell Publishing, 2003:47. | ||
An acute presentation can suggest a panic disorder or acute anxiety state, but first rule out serious conditions such as acute myocardial infarction, arrhythmias, acute infections, GI bleeding, and carbon monoxide poisoning.
Also ask about:
- exacerbating and relieving factors. For example, positional changes, exercise or other physical activity, eating, or missing a meal can trigger presyncope. Also find out about situations that may bring on anxiety, panic, or phobia. Onset of dizziness following these situations may suggest psychogenesis.
- recent falls and injuries. Recurrent falls with presyncope suggest a probable orthostatic or cardiovascular diagnosis in older adults.
4. ANY PAST MEDICAL PROBLEMS?
Ask disease-specific questions. For example:
- Tinnitus or hearing loss could point to a vestibular disorder.
- Metabolic and cardiovascular disorders such as diabetes, ischemic heart disease, postural hypotension, and seizures can result in presyncope.
- Orthostasis, coronary ischemic events, hypoglycemia, and transient ischemic attacks may cause dizziness.
5. IS DIZZINESS RECURRENT?
Panic disorder, anxiety disorders, phobia, and psychogenic hyperventilation are commonly associated with chronic, recurrent dizziness episodes.
6. WHAT MEDICATIONS IS THE PATIENT TAKING?
All psychotropics are suspect when a patient presents with dizziness. When dizziness occurs after a dose or start of therapy, evaluate response to the medication and consider reducing the dosage or changing the medication. If symptoms persist, refer the patient back to the primary care physician to investigate for other causes of dizziness.
Psychotropics that may cause dizziness are listed in Table 2, For a list of other medications associated with dizziness, see this article at www.currentpsychiatry.com.
If the above strategies do not reveal a physical cause of dizziness despite multiple physical complaints, consider examining the patient for depression, anxiety, or panic disorder.
Treating a psychiatric cause
If dizziness is found to be psychogenic and the symptoms impede daily activities or contribute to functional decline, treat the psychiatric disorder but carefully weigh the risks and benefits of drug treatment.
Although SSRIs may cause dizziness, these agents are recommended first-line treatment for depression, anxiety, and/or phobia in older patients with dizziness because of their relative lack of anticholinergic action and side effects compared with other antidepressants or anxiolytics.
Coexisting medical symptoms may dictate choice of agent. For example, consider a sedating SSRI for a patient with sleep disturbances caused by dizziness or the psychiatric disorder; choose a nonsedating SSRI if the patient is sleeping normally.
Because SSRIs may cause weight loss, avoid giving them to patients with weight loss associated with dizziness or an underlying psychiatric illness. Mirtazapine, which is associated with weight gain, may offset weight loss. Start mirtazapine at 15 mg at bedtime for older patients.
Start low and go slow when prescribing an SSRI to an older patient. Dosing strategies applicable to younger patients should not be extrapolated to older patients, especially those with dizziness.
We have found that older patients respond well to minimum or below-normal SSRI dosages (Table 3). Titrate very slowly and instruct patients to report dizziness. Reduce the dosage if dizziness emerges.
If the patient does not respond to an SSRI or mirtazapine, consider a serotonin and norepinephrine reuptake inhibitor, which also has favorable anticholinergic and side-effect profiles.
Related resources
- WebMD Health—Dizziness: lightheadedness and vertigo. http://my.webMD.com/hw/health_guide_atoz/hw88500.asp.
- Sloane PD. Clinical research and geriatric dizziness: The blind men and the elephant. J Am Geriatr Soc 1999;47:113-14.
- Kroenke K, Hoffman RM, Einstadter D. How common are various forms of dizziness? A critical review. South Med J 2000;93:160-7.
Drug brand names
- Alprazolam • Xanax
- Amitriptyline • Elavil
- Bupropion • Wellbutrin
- Buspirone • BuSpar
- Carbamazepine • Tegretol
- Chlordiazepoxide • Librium
- Chlorpromazine • Thorazine
- Citalopram • Celexa
- Clonazepam • Klonopin
- Diazepam • Valium
- Divalproex/valproic acid • Depakote
- Escitalopram • Lexapro
- Estazolam • ProSom
- Fluoxetine • Prozac
- Fluphenazine • Prolixin
- Flurazepam • Dalmane
- Gabapentin • Neurontin
- Imipramine • Tofranil
- Lamotrigine • Lamictal
- Lorazepam • Ativan
- Memantine • Namenda
- Mirtazapine • Remeron
- Nefazodone • Serzone
- Nortriptyline • Pamelor
- Olanzapine • Zyprexa
- Oxazepam • Serax
- Oxcarbazepine • Trileptal
- Paroxetine • Paxil
- Perphenazine • Trilafon
- Phenelzine • Nardil
- Phenytoin • Dilantin
- Prochlorperazine • Compazine
- Quazepam • Doral
- Rivastigmine • Exelon
- Selegiline • Eldepryl
- Sertraline • Zoloft
- Tacrine • Cognex
- Temazepam • Restoril
- Thioridazine • Mellaril
- Trazodone • Desyrel
- Triazolam • Halcion
- Trifluoperazine • Vesprin
- Venlafaxine • Effexor
- Zolpidem • Ambien
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Acknowledgment
The authors thank Robert Cluxton, PharmD, University of Cincinnati College of Pharmacy, for helping to prepare this manuscript for publication.
1. Sloane PD, Hartman M, Mitchell CM. Psychological factors associated with chronic dizziness in patients aged 60 and older. J Am Geriatr Soc 1994;42:847-52.
2. Tinetti ME, Williams CS, Gill TM. Dizziness among older adults: a possible geriatric syndrome. Ann Intern Med 2000;132:337-44.
3. Sloane PD, Coeytaux RR, Beck RS, Dallara J. Dizziness: state of the science. Ann Intern Med 2001;134(9 pt 2):823-32.
4. Hoffman RM, Einstadter D, Kroenke K. Evaluating dizziness. Am J Med 1999;107:468-78.
5. Drachman DA. A 69-year-old man with chronic dizziness. JAMA 1998;280:2111-18.
6. Drachman DA, Hart CW. An approach to the dizzy patient. Neurology 1972;22:323-34.
7. Kapoor WN. Syncope. N Engl J Med 2000;343:1856-62.
8. Baloh RW. Hearing and equilibrium. In: Goldman L, Ansiello D (eds). Cecil textbook of medicine (22nd ed). Philadelphia: Saunders 2004;2436-42.
9. Mathias S, Nayak US, Isaacs B. Balance in elderly patients: the “get-up and go” test. Arch Phys Med Rehabil 1986;67:387-9.
Dizziness is common among patients age 65 and older, and more than one-third have a psychiatric disorder that is caused by or is causing their dizziness.1
When older patients present with dizziness, psychiatrists may be asked to alleviate the psychological symptoms and help identify the underlying disease state.2
More than 60 medical and psychiatric disorders and many medications can cause dizziness. To help you sort through the possibilities, we offer:
- six diagnostic questions to rule out underlying medical problems
- lists of commonly used psychotropics and other drugs that may cause dizziness
- advice on treating depression, anxiety, and panic disorder in an older patient with dizziness while avoiding side effects and drug interactions.
Table 1
Four types of dizziness and their usual causes
| Vertigo Benign positional vertigo CNS cause—tumor, demyelination, neurodegenerative disorders Labyrinthitis Meniere’s disease Peripheral vestibulopathy (in 50% of cases) Vestibular neuronitis |
| Presyncope Arrhythmias Carotid sinus disease Hypoglycemia Neurocardiogenic syncope Organic heart disease Orthostatic hypotension Seizures Situational Transient ischemic attacks |
| Disequilibrium Balance and gait disorder Mixed CNS diseases (ischemic, degenerative) Neurodegenerative CNS disorders Presbystasis Sensorimotor dysfunction |
| Psychogenic lightheadedness Agoraphobia Anxiety Depression Panic disorder Hyperventilation |
| Source: Adapted from reference 6 |
Many causes of dizziness
The term “dizziness” is hard to define because of its nonspecific and variable symptom description, multiple causes, and lack of clear diagnostic and management guidelines. In clinical use, dizziness encompasses abnormal sensations relating to perception of the body’s relationship to space.
Some researchers believe dizziness is a distinct geriatric syndrome because numerous factors related to aging cause dizziness,2 including physiologic changes (presbystasis), accumulated impairment, disease states, and interactions between multiple medications.
Anxiety, somatization, panic disorder, and depression cause dizziness in the elderly, as do:
- peripheral vestibular disorders
- brainstem cerebrovascular accident
- diabetes mellitus
- neurologic disorders such as Parkinson’s disease
- and cardiovascular disorders.
Selective serotonin reuptake inhibitors (SSRIs) and tricyclic antidepressants also have been shown to cause dizziness, as have numerous nonpsychotropic agents.
Recognizing patterns, testing hypotheses, and extending the diagnostic process over time can help you differentiate psychogenic from medicationinduced or neurologic dizziness.3 Because the presentation is so complex and the differential diagnosis so broad, algorithmic diagnosis is less effective than a flexible clinical approach that allows for uncertainty in evaluating initial symptoms.
Determining the cause
A thorough patient history and physical examination can uncover a cause of dizziness in 75% of cases.4 Look for duration of dizziness symptoms; history of heart disease, diabetes or other illnesses; family history of psychiatric disorders; and other illnesses among family members.
Ask the following six complaint-specific questions to help you narrow the differential diagnosis and rule out nonpsychiatric causes.5
1. WHAT TYPE OF DIZZINESS DOES THE PATIENT HAVE?
Four categories—vertigo, presyncope, disequilibrium, and lightheadedness—are used to classify dizziness (Table 1).6
Vertigo is a sense that the body or environment is Patients may feel as if the floor is tilting, sinking, rising or veering sideways, or they may feel pulled to one side.
Vertigo is commonly caused by peripheral vestibular disorders—including benign positional vertigo, Meniere’s disease, labyrinthitis, and vestibular neuronitis—and central vestibular disorders associated with cerebrovascular disease, tumors, demyelinating diseases, migraines, seizures, multiple sclerosis and other CNS diseases. Acute-onset vertigo and neurologic signs suggest brainstem infarction.
Nystagmus is usually present, horizontal, and may be rotational at times. A vertical-beating nystagmus points to a probable CNS cause and requires urgent neuroimaging and referral to a neurologist or otolaryngologist.
Presyncope describes near-fainting. A dimming of vision and roaring in the ears may precede presyncope.
Depending on its cause, presyncope may occur regardless of position or only when upright. Common causes include orthostatic hypotension, neurocardiogenic syncope, organic heart disease, arrhythmias, carotid sinus disease, seizures, hypoglycemia, and transient ischemic attacks.
Abrupt presyncopal attacks that occur regardless of position suggest a cardiovascular cause. If onset is gradual and not improved by lying down, suspect a cerebral metabolic cause such as hypoglycemia.
Syncope, like presyncope, often is traced to an underlying cardiovascular disease. Dizziness and syncope often coexist, and both can be multifactorial. Dizziness may precede or follow syncopal episodes.
Differentiating syncope and dizziness is important because many underlying causes of syncope can be fatal. By contrast, dizziness symptoms are usually benign and self-limiting.7
A thorough history is critical to distinguishing dizziness from presyncope. Assess medication effects—especially CNS-acting medications, cardiovascular drugs, antihypertensives, antibiotics, and over-the-counter medications such as dextromethorphan and acetaminophen compounds. Also check for dehydration.
Disequilibrium disorder signifies unsteadiness or a loss of balance primarily involving the lower extremities. Symptoms are evoked by walking or standing and relieved by sitting or lying down. Gait is abnormal and balance is compromised without abnormal head sensations.
Common causes include balance and gait disorders, sensorimotor dysfunction, presbystasis, neurodegenerative CNS disorders, and mixed ischemic and degenerative CNS diseases.
Vague lightheadedness is often associated with somatic symptoms such as headache. Some patients describe a floating sensation.
Lightheadedness is frequently associated with anxiety, panic disorder, depression, and somatization. Hyperventilation and agoraphobia are other common causes.
Multiple symptoms, multiple types. Classifying an older patient’s dizziness can be challenging because many patients report symptoms that suggest two or more subtypes.2 Also, patients often have trouble describing their dizziness symptoms, sometimes using terms such as “giddiness,” “wooziness,” or “confusion.”
To help patients explain dizziness symptoms more accurately, ask specific questions such as:
- Do you at times feel like you’re about to faint?
- Do you feel as if the room is moving?
- Do you sometimes feel as though you’re going to fall?
Table 2
Psychotropics that may cause dizziness
| Anti-Alzheimer’s medications Memantine, rivastigmine, tacrine |
| Anticonvulsants Phenytoin |
| Antidepressants Monoamine oxidase inhibitors (phenelzine, selegiline) Selective serotonin reuptake inhibitors (all) Tricyclics (amitriptyline, imipramine, nortriptyline, trazodone) Others (bupropion, buspirone, mirtazapine, nefazodone, venlafaxine) |
| Antipsychotics Typicals (chlorpromazine, fluphenazine, perphenazine, prochlorperazine, thioridazine, trifluoperazine) Atypicals (all except olanzapine) |
| Anxiolytics Alprazolam, chlordiazepoxide, clonazepam, diazepam, lorazepam, oxazepam |
| Hypnotics Estazolam, flurazepam, quazepam, temazepam, triazolam, zolpidem |
| Mood stabilizers Carbamazepine, divalproex/valproic acid, gabapentin, lamotrigine, oxcarbazepine |
| Source: Clinical Pharmacology version 2.11. Tampa, FL: Gold Standard MultiMedia, 2004. |
2. HOW DO DIZZINESS SYMPTOMS RELATE TO POSITION OR MOTION?
By reproducing dizziness symptoms, some quick-maneuver tests can help patients describe their symptoms and may reveal a medical cause.
Dix-Hallpike maneuver.3 Move the patient rapidly from a seated to prone position with the head below the horizontal plane and turned 45 degrees for 10 seconds; then have the patient sit up. Repeat with the head turned to the other side. If dizziness does not occur within a few seconds after each test, rule out benign positional vertigo.
Seated head turn, or head-thrust test, measures qualitative vestibular function.8 Move the head rapidly by 45 degrees in a brief, small-amplitude thrust to one side while the patient focuses on your nose; this gauges vestibularocular control. Repeat the test in the other direction. A refixation corrective saccade, occurring as the patient tries to fixate on the target, indicates a possible vestibular disorder.
‘Get-Up and Go’ test, which takes less than 10 seconds, measures balance in older patents.9 Have the patient stand up, walk 10 feet, turn around, walk back, and sit down. Watch for staggering, unsteadiness, and use of hands to balance. Onset of symptoms suggests dizziness brought on during activities of daily living and provides information on how dizziness is affecting the patient’s ability to function.
Romberg test. Have the patient stand with heels together, first with eyes open and then closed. Vision and proprioceptive signals are used to compensate for vestibular loss. Thus, a balance disturbance with eyes closed suggests vestibular or spinal proprioceptive problems and may predict risk of falls caused by inability to compensate.8
3. WHAT IS THE COURSE OF DIZZINESS?
Differentiating acute, sudden-onset dizziness from chronic, gradual-onset dizziness can help uncover the problem’s cause and seriousness. The latter often has a psychological cause or may point to vestibular or minor cardiovascular problems. Tinetti et al2 identified anxiety or depressive symptoms as risk factors among community-based older persons who reported dizziness episodes lasting 1 month.
Table 3
Recommended SSRI starting dosages for older patients
| SSRI | Starting dosage (mg/d) | Maximum dosage (mg/d) |
|---|---|---|
| Citalopram | 10 to 20 | 30 |
| Escitalopram | 10 | 10 |
| Fluoxetine* | 5 to 10 | 60 |
| Paroxetine | 5 | 40 |
| Sertraline | 25 to 50 | 200 |
| * Most patients will not need more than 20 mg/d. Dosages 40 mg/d should be divided into twice-daily doses. | ||
| Source: Adapted from Reuben DB, Herr K, Pacala JT, et al. Geriatrics at your fingertips (5th ed). Malden, MA: Blackwell Publishing, 2003:47. | ||
An acute presentation can suggest a panic disorder or acute anxiety state, but first rule out serious conditions such as acute myocardial infarction, arrhythmias, acute infections, GI bleeding, and carbon monoxide poisoning.
Also ask about:
- exacerbating and relieving factors. For example, positional changes, exercise or other physical activity, eating, or missing a meal can trigger presyncope. Also find out about situations that may bring on anxiety, panic, or phobia. Onset of dizziness following these situations may suggest psychogenesis.
- recent falls and injuries. Recurrent falls with presyncope suggest a probable orthostatic or cardiovascular diagnosis in older adults.
4. ANY PAST MEDICAL PROBLEMS?
Ask disease-specific questions. For example:
- Tinnitus or hearing loss could point to a vestibular disorder.
- Metabolic and cardiovascular disorders such as diabetes, ischemic heart disease, postural hypotension, and seizures can result in presyncope.
- Orthostasis, coronary ischemic events, hypoglycemia, and transient ischemic attacks may cause dizziness.
5. IS DIZZINESS RECURRENT?
Panic disorder, anxiety disorders, phobia, and psychogenic hyperventilation are commonly associated with chronic, recurrent dizziness episodes.
6. WHAT MEDICATIONS IS THE PATIENT TAKING?
All psychotropics are suspect when a patient presents with dizziness. When dizziness occurs after a dose or start of therapy, evaluate response to the medication and consider reducing the dosage or changing the medication. If symptoms persist, refer the patient back to the primary care physician to investigate for other causes of dizziness.
Psychotropics that may cause dizziness are listed in Table 2, For a list of other medications associated with dizziness, see this article at www.currentpsychiatry.com.
If the above strategies do not reveal a physical cause of dizziness despite multiple physical complaints, consider examining the patient for depression, anxiety, or panic disorder.
Treating a psychiatric cause
If dizziness is found to be psychogenic and the symptoms impede daily activities or contribute to functional decline, treat the psychiatric disorder but carefully weigh the risks and benefits of drug treatment.
Although SSRIs may cause dizziness, these agents are recommended first-line treatment for depression, anxiety, and/or phobia in older patients with dizziness because of their relative lack of anticholinergic action and side effects compared with other antidepressants or anxiolytics.
Coexisting medical symptoms may dictate choice of agent. For example, consider a sedating SSRI for a patient with sleep disturbances caused by dizziness or the psychiatric disorder; choose a nonsedating SSRI if the patient is sleeping normally.
Because SSRIs may cause weight loss, avoid giving them to patients with weight loss associated with dizziness or an underlying psychiatric illness. Mirtazapine, which is associated with weight gain, may offset weight loss. Start mirtazapine at 15 mg at bedtime for older patients.
Start low and go slow when prescribing an SSRI to an older patient. Dosing strategies applicable to younger patients should not be extrapolated to older patients, especially those with dizziness.
We have found that older patients respond well to minimum or below-normal SSRI dosages (Table 3). Titrate very slowly and instruct patients to report dizziness. Reduce the dosage if dizziness emerges.
If the patient does not respond to an SSRI or mirtazapine, consider a serotonin and norepinephrine reuptake inhibitor, which also has favorable anticholinergic and side-effect profiles.
Related resources
- WebMD Health—Dizziness: lightheadedness and vertigo. http://my.webMD.com/hw/health_guide_atoz/hw88500.asp.
- Sloane PD. Clinical research and geriatric dizziness: The blind men and the elephant. J Am Geriatr Soc 1999;47:113-14.
- Kroenke K, Hoffman RM, Einstadter D. How common are various forms of dizziness? A critical review. South Med J 2000;93:160-7.
Drug brand names
- Alprazolam • Xanax
- Amitriptyline • Elavil
- Bupropion • Wellbutrin
- Buspirone • BuSpar
- Carbamazepine • Tegretol
- Chlordiazepoxide • Librium
- Chlorpromazine • Thorazine
- Citalopram • Celexa
- Clonazepam • Klonopin
- Diazepam • Valium
- Divalproex/valproic acid • Depakote
- Escitalopram • Lexapro
- Estazolam • ProSom
- Fluoxetine • Prozac
- Fluphenazine • Prolixin
- Flurazepam • Dalmane
- Gabapentin • Neurontin
- Imipramine • Tofranil
- Lamotrigine • Lamictal
- Lorazepam • Ativan
- Memantine • Namenda
- Mirtazapine • Remeron
- Nefazodone • Serzone
- Nortriptyline • Pamelor
- Olanzapine • Zyprexa
- Oxazepam • Serax
- Oxcarbazepine • Trileptal
- Paroxetine • Paxil
- Perphenazine • Trilafon
- Phenelzine • Nardil
- Phenytoin • Dilantin
- Prochlorperazine • Compazine
- Quazepam • Doral
- Rivastigmine • Exelon
- Selegiline • Eldepryl
- Sertraline • Zoloft
- Tacrine • Cognex
- Temazepam • Restoril
- Thioridazine • Mellaril
- Trazodone • Desyrel
- Triazolam • Halcion
- Trifluoperazine • Vesprin
- Venlafaxine • Effexor
- Zolpidem • Ambien
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Acknowledgment
The authors thank Robert Cluxton, PharmD, University of Cincinnati College of Pharmacy, for helping to prepare this manuscript for publication.
Dizziness is common among patients age 65 and older, and more than one-third have a psychiatric disorder that is caused by or is causing their dizziness.1
When older patients present with dizziness, psychiatrists may be asked to alleviate the psychological symptoms and help identify the underlying disease state.2
More than 60 medical and psychiatric disorders and many medications can cause dizziness. To help you sort through the possibilities, we offer:
- six diagnostic questions to rule out underlying medical problems
- lists of commonly used psychotropics and other drugs that may cause dizziness
- advice on treating depression, anxiety, and panic disorder in an older patient with dizziness while avoiding side effects and drug interactions.
Table 1
Four types of dizziness and their usual causes
| Vertigo Benign positional vertigo CNS cause—tumor, demyelination, neurodegenerative disorders Labyrinthitis Meniere’s disease Peripheral vestibulopathy (in 50% of cases) Vestibular neuronitis |
| Presyncope Arrhythmias Carotid sinus disease Hypoglycemia Neurocardiogenic syncope Organic heart disease Orthostatic hypotension Seizures Situational Transient ischemic attacks |
| Disequilibrium Balance and gait disorder Mixed CNS diseases (ischemic, degenerative) Neurodegenerative CNS disorders Presbystasis Sensorimotor dysfunction |
| Psychogenic lightheadedness Agoraphobia Anxiety Depression Panic disorder Hyperventilation |
| Source: Adapted from reference 6 |
Many causes of dizziness
The term “dizziness” is hard to define because of its nonspecific and variable symptom description, multiple causes, and lack of clear diagnostic and management guidelines. In clinical use, dizziness encompasses abnormal sensations relating to perception of the body’s relationship to space.
Some researchers believe dizziness is a distinct geriatric syndrome because numerous factors related to aging cause dizziness,2 including physiologic changes (presbystasis), accumulated impairment, disease states, and interactions between multiple medications.
Anxiety, somatization, panic disorder, and depression cause dizziness in the elderly, as do:
- peripheral vestibular disorders
- brainstem cerebrovascular accident
- diabetes mellitus
- neurologic disorders such as Parkinson’s disease
- and cardiovascular disorders.
Selective serotonin reuptake inhibitors (SSRIs) and tricyclic antidepressants also have been shown to cause dizziness, as have numerous nonpsychotropic agents.
Recognizing patterns, testing hypotheses, and extending the diagnostic process over time can help you differentiate psychogenic from medicationinduced or neurologic dizziness.3 Because the presentation is so complex and the differential diagnosis so broad, algorithmic diagnosis is less effective than a flexible clinical approach that allows for uncertainty in evaluating initial symptoms.
Determining the cause
A thorough patient history and physical examination can uncover a cause of dizziness in 75% of cases.4 Look for duration of dizziness symptoms; history of heart disease, diabetes or other illnesses; family history of psychiatric disorders; and other illnesses among family members.
Ask the following six complaint-specific questions to help you narrow the differential diagnosis and rule out nonpsychiatric causes.5
1. WHAT TYPE OF DIZZINESS DOES THE PATIENT HAVE?
Four categories—vertigo, presyncope, disequilibrium, and lightheadedness—are used to classify dizziness (Table 1).6
Vertigo is a sense that the body or environment is Patients may feel as if the floor is tilting, sinking, rising or veering sideways, or they may feel pulled to one side.
Vertigo is commonly caused by peripheral vestibular disorders—including benign positional vertigo, Meniere’s disease, labyrinthitis, and vestibular neuronitis—and central vestibular disorders associated with cerebrovascular disease, tumors, demyelinating diseases, migraines, seizures, multiple sclerosis and other CNS diseases. Acute-onset vertigo and neurologic signs suggest brainstem infarction.
Nystagmus is usually present, horizontal, and may be rotational at times. A vertical-beating nystagmus points to a probable CNS cause and requires urgent neuroimaging and referral to a neurologist or otolaryngologist.
Presyncope describes near-fainting. A dimming of vision and roaring in the ears may precede presyncope.
Depending on its cause, presyncope may occur regardless of position or only when upright. Common causes include orthostatic hypotension, neurocardiogenic syncope, organic heart disease, arrhythmias, carotid sinus disease, seizures, hypoglycemia, and transient ischemic attacks.
Abrupt presyncopal attacks that occur regardless of position suggest a cardiovascular cause. If onset is gradual and not improved by lying down, suspect a cerebral metabolic cause such as hypoglycemia.
Syncope, like presyncope, often is traced to an underlying cardiovascular disease. Dizziness and syncope often coexist, and both can be multifactorial. Dizziness may precede or follow syncopal episodes.
Differentiating syncope and dizziness is important because many underlying causes of syncope can be fatal. By contrast, dizziness symptoms are usually benign and self-limiting.7
A thorough history is critical to distinguishing dizziness from presyncope. Assess medication effects—especially CNS-acting medications, cardiovascular drugs, antihypertensives, antibiotics, and over-the-counter medications such as dextromethorphan and acetaminophen compounds. Also check for dehydration.
Disequilibrium disorder signifies unsteadiness or a loss of balance primarily involving the lower extremities. Symptoms are evoked by walking or standing and relieved by sitting or lying down. Gait is abnormal and balance is compromised without abnormal head sensations.
Common causes include balance and gait disorders, sensorimotor dysfunction, presbystasis, neurodegenerative CNS disorders, and mixed ischemic and degenerative CNS diseases.
Vague lightheadedness is often associated with somatic symptoms such as headache. Some patients describe a floating sensation.
Lightheadedness is frequently associated with anxiety, panic disorder, depression, and somatization. Hyperventilation and agoraphobia are other common causes.
Multiple symptoms, multiple types. Classifying an older patient’s dizziness can be challenging because many patients report symptoms that suggest two or more subtypes.2 Also, patients often have trouble describing their dizziness symptoms, sometimes using terms such as “giddiness,” “wooziness,” or “confusion.”
To help patients explain dizziness symptoms more accurately, ask specific questions such as:
- Do you at times feel like you’re about to faint?
- Do you feel as if the room is moving?
- Do you sometimes feel as though you’re going to fall?
Table 2
Psychotropics that may cause dizziness
| Anti-Alzheimer’s medications Memantine, rivastigmine, tacrine |
| Anticonvulsants Phenytoin |
| Antidepressants Monoamine oxidase inhibitors (phenelzine, selegiline) Selective serotonin reuptake inhibitors (all) Tricyclics (amitriptyline, imipramine, nortriptyline, trazodone) Others (bupropion, buspirone, mirtazapine, nefazodone, venlafaxine) |
| Antipsychotics Typicals (chlorpromazine, fluphenazine, perphenazine, prochlorperazine, thioridazine, trifluoperazine) Atypicals (all except olanzapine) |
| Anxiolytics Alprazolam, chlordiazepoxide, clonazepam, diazepam, lorazepam, oxazepam |
| Hypnotics Estazolam, flurazepam, quazepam, temazepam, triazolam, zolpidem |
| Mood stabilizers Carbamazepine, divalproex/valproic acid, gabapentin, lamotrigine, oxcarbazepine |
| Source: Clinical Pharmacology version 2.11. Tampa, FL: Gold Standard MultiMedia, 2004. |
2. HOW DO DIZZINESS SYMPTOMS RELATE TO POSITION OR MOTION?
By reproducing dizziness symptoms, some quick-maneuver tests can help patients describe their symptoms and may reveal a medical cause.
Dix-Hallpike maneuver.3 Move the patient rapidly from a seated to prone position with the head below the horizontal plane and turned 45 degrees for 10 seconds; then have the patient sit up. Repeat with the head turned to the other side. If dizziness does not occur within a few seconds after each test, rule out benign positional vertigo.
Seated head turn, or head-thrust test, measures qualitative vestibular function.8 Move the head rapidly by 45 degrees in a brief, small-amplitude thrust to one side while the patient focuses on your nose; this gauges vestibularocular control. Repeat the test in the other direction. A refixation corrective saccade, occurring as the patient tries to fixate on the target, indicates a possible vestibular disorder.
‘Get-Up and Go’ test, which takes less than 10 seconds, measures balance in older patents.9 Have the patient stand up, walk 10 feet, turn around, walk back, and sit down. Watch for staggering, unsteadiness, and use of hands to balance. Onset of symptoms suggests dizziness brought on during activities of daily living and provides information on how dizziness is affecting the patient’s ability to function.
Romberg test. Have the patient stand with heels together, first with eyes open and then closed. Vision and proprioceptive signals are used to compensate for vestibular loss. Thus, a balance disturbance with eyes closed suggests vestibular or spinal proprioceptive problems and may predict risk of falls caused by inability to compensate.8
3. WHAT IS THE COURSE OF DIZZINESS?
Differentiating acute, sudden-onset dizziness from chronic, gradual-onset dizziness can help uncover the problem’s cause and seriousness. The latter often has a psychological cause or may point to vestibular or minor cardiovascular problems. Tinetti et al2 identified anxiety or depressive symptoms as risk factors among community-based older persons who reported dizziness episodes lasting 1 month.
Table 3
Recommended SSRI starting dosages for older patients
| SSRI | Starting dosage (mg/d) | Maximum dosage (mg/d) |
|---|---|---|
| Citalopram | 10 to 20 | 30 |
| Escitalopram | 10 | 10 |
| Fluoxetine* | 5 to 10 | 60 |
| Paroxetine | 5 | 40 |
| Sertraline | 25 to 50 | 200 |
| * Most patients will not need more than 20 mg/d. Dosages 40 mg/d should be divided into twice-daily doses. | ||
| Source: Adapted from Reuben DB, Herr K, Pacala JT, et al. Geriatrics at your fingertips (5th ed). Malden, MA: Blackwell Publishing, 2003:47. | ||
An acute presentation can suggest a panic disorder or acute anxiety state, but first rule out serious conditions such as acute myocardial infarction, arrhythmias, acute infections, GI bleeding, and carbon monoxide poisoning.
Also ask about:
- exacerbating and relieving factors. For example, positional changes, exercise or other physical activity, eating, or missing a meal can trigger presyncope. Also find out about situations that may bring on anxiety, panic, or phobia. Onset of dizziness following these situations may suggest psychogenesis.
- recent falls and injuries. Recurrent falls with presyncope suggest a probable orthostatic or cardiovascular diagnosis in older adults.
4. ANY PAST MEDICAL PROBLEMS?
Ask disease-specific questions. For example:
- Tinnitus or hearing loss could point to a vestibular disorder.
- Metabolic and cardiovascular disorders such as diabetes, ischemic heart disease, postural hypotension, and seizures can result in presyncope.
- Orthostasis, coronary ischemic events, hypoglycemia, and transient ischemic attacks may cause dizziness.
5. IS DIZZINESS RECURRENT?
Panic disorder, anxiety disorders, phobia, and psychogenic hyperventilation are commonly associated with chronic, recurrent dizziness episodes.
6. WHAT MEDICATIONS IS THE PATIENT TAKING?
All psychotropics are suspect when a patient presents with dizziness. When dizziness occurs after a dose or start of therapy, evaluate response to the medication and consider reducing the dosage or changing the medication. If symptoms persist, refer the patient back to the primary care physician to investigate for other causes of dizziness.
Psychotropics that may cause dizziness are listed in Table 2, For a list of other medications associated with dizziness, see this article at www.currentpsychiatry.com.
If the above strategies do not reveal a physical cause of dizziness despite multiple physical complaints, consider examining the patient for depression, anxiety, or panic disorder.
Treating a psychiatric cause
If dizziness is found to be psychogenic and the symptoms impede daily activities or contribute to functional decline, treat the psychiatric disorder but carefully weigh the risks and benefits of drug treatment.
Although SSRIs may cause dizziness, these agents are recommended first-line treatment for depression, anxiety, and/or phobia in older patients with dizziness because of their relative lack of anticholinergic action and side effects compared with other antidepressants or anxiolytics.
Coexisting medical symptoms may dictate choice of agent. For example, consider a sedating SSRI for a patient with sleep disturbances caused by dizziness or the psychiatric disorder; choose a nonsedating SSRI if the patient is sleeping normally.
Because SSRIs may cause weight loss, avoid giving them to patients with weight loss associated with dizziness or an underlying psychiatric illness. Mirtazapine, which is associated with weight gain, may offset weight loss. Start mirtazapine at 15 mg at bedtime for older patients.
Start low and go slow when prescribing an SSRI to an older patient. Dosing strategies applicable to younger patients should not be extrapolated to older patients, especially those with dizziness.
We have found that older patients respond well to minimum or below-normal SSRI dosages (Table 3). Titrate very slowly and instruct patients to report dizziness. Reduce the dosage if dizziness emerges.
If the patient does not respond to an SSRI or mirtazapine, consider a serotonin and norepinephrine reuptake inhibitor, which also has favorable anticholinergic and side-effect profiles.
Related resources
- WebMD Health—Dizziness: lightheadedness and vertigo. http://my.webMD.com/hw/health_guide_atoz/hw88500.asp.
- Sloane PD. Clinical research and geriatric dizziness: The blind men and the elephant. J Am Geriatr Soc 1999;47:113-14.
- Kroenke K, Hoffman RM, Einstadter D. How common are various forms of dizziness? A critical review. South Med J 2000;93:160-7.
Drug brand names
- Alprazolam • Xanax
- Amitriptyline • Elavil
- Bupropion • Wellbutrin
- Buspirone • BuSpar
- Carbamazepine • Tegretol
- Chlordiazepoxide • Librium
- Chlorpromazine • Thorazine
- Citalopram • Celexa
- Clonazepam • Klonopin
- Diazepam • Valium
- Divalproex/valproic acid • Depakote
- Escitalopram • Lexapro
- Estazolam • ProSom
- Fluoxetine • Prozac
- Fluphenazine • Prolixin
- Flurazepam • Dalmane
- Gabapentin • Neurontin
- Imipramine • Tofranil
- Lamotrigine • Lamictal
- Lorazepam • Ativan
- Memantine • Namenda
- Mirtazapine • Remeron
- Nefazodone • Serzone
- Nortriptyline • Pamelor
- Olanzapine • Zyprexa
- Oxazepam • Serax
- Oxcarbazepine • Trileptal
- Paroxetine • Paxil
- Perphenazine • Trilafon
- Phenelzine • Nardil
- Phenytoin • Dilantin
- Prochlorperazine • Compazine
- Quazepam • Doral
- Rivastigmine • Exelon
- Selegiline • Eldepryl
- Sertraline • Zoloft
- Tacrine • Cognex
- Temazepam • Restoril
- Thioridazine • Mellaril
- Trazodone • Desyrel
- Triazolam • Halcion
- Trifluoperazine • Vesprin
- Venlafaxine • Effexor
- Zolpidem • Ambien
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Acknowledgment
The authors thank Robert Cluxton, PharmD, University of Cincinnati College of Pharmacy, for helping to prepare this manuscript for publication.
1. Sloane PD, Hartman M, Mitchell CM. Psychological factors associated with chronic dizziness in patients aged 60 and older. J Am Geriatr Soc 1994;42:847-52.
2. Tinetti ME, Williams CS, Gill TM. Dizziness among older adults: a possible geriatric syndrome. Ann Intern Med 2000;132:337-44.
3. Sloane PD, Coeytaux RR, Beck RS, Dallara J. Dizziness: state of the science. Ann Intern Med 2001;134(9 pt 2):823-32.
4. Hoffman RM, Einstadter D, Kroenke K. Evaluating dizziness. Am J Med 1999;107:468-78.
5. Drachman DA. A 69-year-old man with chronic dizziness. JAMA 1998;280:2111-18.
6. Drachman DA, Hart CW. An approach to the dizzy patient. Neurology 1972;22:323-34.
7. Kapoor WN. Syncope. N Engl J Med 2000;343:1856-62.
8. Baloh RW. Hearing and equilibrium. In: Goldman L, Ansiello D (eds). Cecil textbook of medicine (22nd ed). Philadelphia: Saunders 2004;2436-42.
9. Mathias S, Nayak US, Isaacs B. Balance in elderly patients: the “get-up and go” test. Arch Phys Med Rehabil 1986;67:387-9.
1. Sloane PD, Hartman M, Mitchell CM. Psychological factors associated with chronic dizziness in patients aged 60 and older. J Am Geriatr Soc 1994;42:847-52.
2. Tinetti ME, Williams CS, Gill TM. Dizziness among older adults: a possible geriatric syndrome. Ann Intern Med 2000;132:337-44.
3. Sloane PD, Coeytaux RR, Beck RS, Dallara J. Dizziness: state of the science. Ann Intern Med 2001;134(9 pt 2):823-32.
4. Hoffman RM, Einstadter D, Kroenke K. Evaluating dizziness. Am J Med 1999;107:468-78.
5. Drachman DA. A 69-year-old man with chronic dizziness. JAMA 1998;280:2111-18.
6. Drachman DA, Hart CW. An approach to the dizzy patient. Neurology 1972;22:323-34.
7. Kapoor WN. Syncope. N Engl J Med 2000;343:1856-62.
8. Baloh RW. Hearing and equilibrium. In: Goldman L, Ansiello D (eds). Cecil textbook of medicine (22nd ed). Philadelphia: Saunders 2004;2436-42.
9. Mathias S, Nayak US, Isaacs B. Balance in elderly patients: the “get-up and go” test. Arch Phys Med Rehabil 1986;67:387-9.
8 steps to manage recurrent abdominal pain
Just three words—“My tummy hurts”—can mobilize a child’s parents into a high state of worry, especially on school days. They wonder: Is our child sick? Should he or she stay home? Why is this happening so often?
Although recurrent abdominal pain (RAP) is real, it usually is not caused by tissue damage or serious physical disease. When children with RAP are referred for psychiatric evaluation—often after extensive medical workups—we can help them and their parents manage the problem and function more normally. This article:
- describes physiologic mechanisms that may underlie recurrent GI distress
- discusses the high correlation of psychiatric comorbidities with RAP
- recommends judicious laboratory testing
- reviews evidence on medications and psychotherapies to improve RAP symptoms
- offers advice on building a therapeutic alliance with the patient and family.
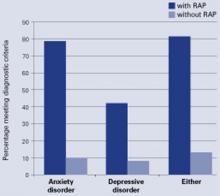
Figure Comorbid anxiety and depressive disorders in children with RAP
Children with functional RAP are much more likely to be anxious or depressed than similar pain-free children. A recent blinded study followed 80 children ages 8 to 15 (42 with RAP and 38 controls) identified through screening at primary care pediatric offices. Each was assessed using the Schedule for Affective Disorders and Schizophrenia for School Age Children, Present and Lifetime version (K-SADS-PL). Percentage meeting diagnostic criteria
Source: Reference 5.
RAP: A ‘Functional’ disorder
RAP is a somatoform (or “functional”) disorder, defined as physical symptoms not fully explained by a medical condition, effects of a substance, or another mental disorder. Symptoms cause distress and/or functional impairment and are not intentionally produced.1
A patient with RAP experiences at least three episodes of abdominal pain over 3 months that interfere with daily activities.2 RAP affects 7% to 25% of school-aged children and adolescents,3 most of whom have a functional disorder.4
RAP is equally common among prepubertal boys and girls but more common among girls during adolescence.3 RAP can impair school attendance and performance and stigmatize a child as “sickly.”
Common comorbid symptoms
Physical. Besides stomach pain, children with RAP often experience headaches (including migraines), other GI symptoms, general aches and pains, dizziness, and fatigue.
Patients with RAP who do not experience GI bleeding, anemia, fever, weight loss, growth failure, or persistent vomiting most likely do not have a serious underlying disease.
Psychiatric. Children with RAP have much higher rates of anxiety (80%) and depressive (40%) disorders than do their unaffected peers (Figure).5 We have also seen higher levels of suicidal thinking in children with RAP in primary care settings compared with pain-free controls (14% vs. 4%, P = 0.04; unpublished data).
In most cases, psychiatric comorbidities appear to precede or coincide with RAP onset. Separation fears, generalized anxiety, and social anxiety in particular are common in patients with RAP yet are seldom recognized in medical settings.
Having childhood RAP increases the risk of anxiety, depression, and hypochondriacal fears in adulthood.6 We do not know whether early intervention prevents later disability.
Use of medical services. Abdominal pain accounts for 2% to 4% of all pediatric office visits.7 In one study, 8% of middle school and high school students said they had visited a physician for evaluation of stomach pain during the previous year.8 Children with RAP make more ambulatory health and mental health visits than peers9 and are at risk for unnecessary and potentially dangerous medical tests, procedures, and treatments, including abdominal surgery.10
Four functional GI disorders
To better characterize youths with functional RAP, symptom-based criteria have been developed and applied for functional GI disorders, defined as chronic or recurrent GI symptoms without explanatory structural or biochemical abnormalities.11 Four such disorders are relevant in children with RAP.
Irritable bowel syndrome (IBS): RAP with at least two of the following symptoms: relief with defecation, change in stool frequency, and change in stool form or appearance (occurs in approximately 50% of RAP cases).
Functional dyspepsia: RAP centered in the upper abdomen that is not associated with changes in bowel habits.
Abdominal migraine: Paroxysmal midline abdominal pain lasting 2 hours to several days with symptom-free intervals of weeks to months and at least two of the following: headache during episodes, photophobia during episodes, unilateral headache, aura, and a family history of migraine.
Functional abdominal pain: Continuous or nearly continuous abdominal pain for 6 months or more.
The reliability, validity, and clinical relevance of these criteria have not been demonstrated. Some children with RAP do not meet any criteria for a specific functional GI disorder.
Gut-brain connections. RAP may be associated with a heightened sensitivity to visceral sensations (visceral hyperalgesia) and a low pressure-pain threshold, leading to speculation that these children are hypersensitive to pain.
High rates of anxiety disorders and temperamental harm avoidance also are seen in patients with RAP, along with a tendency to develop pain when faced with unexpected events. Whether these children are more likely than others to perceive novel internal or external perceptions as threatening is open to debate.
Table 1
Recurrent abdominal pain: 8 steps to assessment and diagnosis
|
Serotonin communicates nociceptive information between the gut and brain and may mediate visceral hyperalgesia. Gut enterochromaffin cells contain more than 90% of the body’s total serotonin. They act as sensory transducers, releasing serotonin in response to increased intraluminal pressure or inflammation.
The released serotonin can cause abdominal discomfort by stimulating 5-HT 3 receptors on vagal afferents and can influence gut peristaltic activity by stimulating enteric afferents. The same serotonin transporter responsible for CNS serotonin reuptake is expressed throughout the gut.
A constellation of clues
The ideal RAP evaluation includes information from the child, parents, educators, and other health care professionals (Table 1).
Begin by acknowledging the patient’s suffering and the parent’s concerns; do not challenge the pain’s subjective reality. Rather than prejudging its cause, document the pain’s timing, context, and characteristics, and review the patient’s history. A constellation of clues is most suggestive of RAP (Table 2); single clues are not definitive.12,13
Table 2
Clues that suggest functional pain*
|
| * No single clue is definitive. |
| Source: Adapted from reference 12. |
Diagnostic testing. Be judicious in selecting diagnostic tests and procedures. Continuing to order studies in a haphazard effort to rule out disease can generate concerns that “the doctor doesn’t know what’s wrong” and heighten the family’s fear that a disease has been missed.
The process of “ruling out” physical disease may have no apparent end. Unless you are reasonably comfortable that a serious physical disease has not been missed, it is difficult to explain RAP to the patient and family and lay the foundation for intervention.
On the other hand, you must balance the importance of minimizing your own and the family’s anxiety about unrecognized disease against the physical and psychological risks and costs associated with medical tests and procedures.
Social assessment. Assess social and familial reinforcement (secondary gain) of the pain. Parents sometimes inadvertently encourage their children’s sick-role behaviors by providing excessive attention, rewards, or opportunities to avoid uncomfortable situations. RAP can become an excuse for poor performance (self-handicapping), particularly in children with a learning disorder.
How to deliver the diagnosis
Functional abdominal pain is essentially a clinical diagnosis that relies on presentation, course, and findings. As mentioned, a constellation of “clues” is most supportive, as is having typical IBS symptoms.
Before declaring the diagnosis, discuss with the family the patient’s physical, emotional, and behavioral symptoms and the context in which RAP developed. Doing so can help maintain your credibility and establish a consensus.
Once you declare the diagnosis, discuss it clearly and frankly. Families are not likely to be reassured if you do not offer a plausible explanation for the lack of physical findings.
Precautions. When a definitive diagnosis is not possible, acknowledge that uncertainty. Although you must discuss any recognized psychiatric comorbidity, attempting to “explain” that the disorder is causing the pain is usually impractical and intellectually dishonest.
Also, given the pervasive nature of stigma, do not convey embarrassment or unease about diagnosing functional RAP or any comorbid psychiatric disorder.
Follow-up testing. Once you diagnose functional RAP, further testing is generally not necessary. Tests might be indicated if you:
- receive new information
- observe a change in clinical status
- or are convinced that treatment will not work unless the family is reassured by further investigation.
Collaborative treatment
Reassurance and education. Reassurance that the patient does not have a serious physical disease is necessary but rarely sufficient. Explain that the child’s pain does not appear to reflect tissue damage and is not threatening. On the other hand, avoid giving excessive reassurance, particularly when obsessional illness worry and hypochondriacal fears are prominent. Address illness worry as a problem to be solved together.
Discuss with the patient and family what is known and not known about functional RAP, and encourage them to ask questions. This is an opportunity for you to instill hope and cultivate positive expectations, but avoid promising cure. Discuss the gut-brain connection and relevance of visceral hyperalgesia, including serotonin’s potential roles in RAP pathogenesis.
Partners in care. Collaborative treatment increases the likelihood of success. Discuss the importance of a therapeutic partnership, and clarify any areas of disagreement with the diagnosis or treatment plan.
Clearly delineate your roles and responsibilities and those of the patient, family, and other health care team members. Poor communication is pediatricians’ most common complaint about psychiatrists.14 Good interdisciplinary communication decreases the chance that treatment strategies will be duplicated, diluted, or misinterpreted.
Consolidate medical care with a single clinician—often the primary care physician—based on discussions with the patient, family, and health care team. The coordinating clinician can mediate between the school and family when tensions develop over poor attendance or requests for special treatment. It is often useful for this clinician to spell out:
- what constitutes a legitimate medical excuse for school absence
- who will legitimize excuses.
All parties should understand that the school will view an unexcused absence as truancy and act appropriately.
Diet and lifestyle. Encourage the patient to maintain a regular schedule and a healthy diet. Specific dietary interventions have not been proven effective, despite speculation that lack of dietary fiber or lactose intolerance might cause RAP.15,16 Also encourage adequate sleep and regular exercise.
Medication and psychotherapy
Because no strong evidence-based guidelines address pediatric RAP intervention, family preferences usually guide initial treatment decisions. This highlights the importance of good communication and a therapeutic partnership among the clinician, family, and patient.
Antispasmodics, acid reducers, and antidepressants are commonly prescribed for RAP, though none are well-supported in the literature and no controlled studies have gauged medication’s impact on psychiatric comorbidity.16
Antidepressants. Selective serotonin reuptake inhibitors (SSRIs) might help relieve RAP symptoms, but the evidence is inconclusive. SSRIs are considered potentially beneficial in RAP because they may help communicate nociceptive information between the gut and brain and mediate visceral hyperalgesia.
SSRIs at first may increase serotonin at the synapse, which one might assume would to worsen abdominal symptoms. However, ongoing SSRI use could “down-regulate” postsynaptic 5-HT3 receptors and desensitize postsynaptic cells to the effects of local serotonin.
Our group recently conducted a 12-week open trial of citalopram for functional pediatric RAP.17 The 25 participants received 10 mg/d the first week, then 20 mg/d thereafter if tolerated. At week 4, nonresponders and partial responders who were tolerating the medication began receiving 40 mg/d.
Table 3
SSRI daily dosing for pediatric RAP
| Drug | Starting dosage* | Target dosage† | Maximum dosage‡ |
|---|---|---|---|
| Citalopram | 10 mg | 20 mg | 40 to 60 mg |
| Escitalopram | 5 mg | 10 mg | 20 to 30 mg |
| Fluoxetine | 10 mg | 20 mg | 40 mg |
| Fluvoxamine | 50 mg | 100 mg | 300 mg |
| Sertraline | 25 mg | 50 mg | 200 mg |
| * First 3 to 7 days. | |||
| † If patient tolerates starting dosage, increase to target dosage. | |||
| ‡ If patient does not respond to target dosage in 2 to 3 weeks, consider increasing the dosage. | |||
At trial’s end, more than two-thirds of participants were taking 40 mg/d. We rated 21 of 25 patients (84%) as “much improved” or “very much improved,” using the Clinical Global Impression-Improvement scale. Abdominal pain, anxiety, depression, other somatic symptoms, and functional impairment all improved significantly during treatment. Suicidal thoughts diminished progressively from baseline, and no patient reported suicidal thinking at study’s end. Citalopram was generally well tolerated.
With SSRI treatment, start at a low dosage for 3 to 7 days (Table 3). If tolerated, increase to a typical therapeutic dosage. If symptoms fail to respond after 2 or 3 weeks, consider a higher dosage. A short course of an oral benzodiazepine (such as clonazepam, 0.25 mg bid) during the first weeks of SSRI treatment sometimes helps particularly anxious patients or those whose pain appears closely associated with anxiety or “stress.”
Pediatric gastroenterologists often prescribe a low-dose tricyclic antidepressant as first-line therapy, but we discourage this. TCAs lack efficacy in pediatric depression and pose a greater risk of side effects and safety concerns than SSRIs.18
Other agents have been tried for RAP-associated conditions:
Famotidine, a histamine type 2 receptor blocker, may reduce pain in children with dyspepsia and RAP.19
Peppermint oil reduced abdominal pain in one study of children with IBS but had little effect on other symptoms.20
Medications such as alosetron and tegaserod that interrupt serotonergic neurotransmission in the gut have shown benefit in adults with IBS but have not been studied in children.
Psychotherapy. A few small studies suggest that cognitive-behavioral therapies (CBT) are helpful in RAP, but CBT may be difficult to deliver in medical settings.21,22 A simplified “rehabilitative” approach that incorporates CBT principles involves having the clinician and patient view RAP as a challenge to be overcome, rather than a burden to be endured. Such an approach emphasizes the child’s fundamental strength and adaptability rather than vulnerabilities.
The goal of therapy is redirected from finding a cure to coping with and overcoming the problem. This approach challenges the notion that the child cannot resume normal function until the pain is completely gone. It encourages active, problem-focused coping, and discourages passive acceptance—which is associated with greater symptom burden and functional impairment.
Work with parents to reinforce the child’s health-promoting behaviors and minimize negative and social reinforcements (secondary gain) associated with RAP. Advise parents to:
- encourage and reward full school attendance
- avoid home-bound instruction
- expect the child to function despite physical distress
- insist that the child perform age-appropriate household chores and other responsibilities.
Self-management skills—such as relaxation training, hypnosis, biofeedback, and guided imagery—may help reduce pain and manage physiologic arousal.16 Deception strategies such as placebo or sham interventions are unethical and impractical.
Related resources
- Campo JV. Functional recurrent abdominal pain in children and adolescents. Digestive Health Matters 2003;12(3):15-7.
- International Foundation for Functional Gastrointestinal Disorders. www.iffgd.org
- University of Pittsburgh. Advanced Center for Interventions and Services Research on Early Onset Mood and Anxiety Disorders. www.moodykids.org
Drug brand names
- Alosetron • Lotronex
- Citalopram • Celexa
- Clonazepam • Klonopin
- Escitalopram • Lexapro
- Famotidine • Pepcid
- Fluoxetine • Prozac
- Fluvoxamine • Luvox
- Sertraline • Zoloft
- Tegaserod • Zelnorm
Disclosure
Dr. Campo’s work has been supported by the National Institute of Mental Health (grant MH01780) and in part by the Advanced Center for Interventions and Services Research on Early Onset Mood and Anxiety Disorders (grant MH66371). He also receives grants from Forest Pharmaceuticals and is a consultant to Eli Lilly and Co.
1. American Psychiatric Association Diagnostic and statistical manual of mental disorders (4th ed, revised). Washington, DC: American Psychiatric Association, 2000.
2. Apley J, Naish N. Recurrent abdominal pains: a field survey of 1,000 school children. Arch Dis Child 1958;33(168):165-70.
3. Scharff L. Recurrent abdominal pain in children: a review of psychological factors and treatment. Clin Psychol Rev 1997;17(2):145-66.
4. Boyle JT. Recurrent abdominal pain: an update. Pediatr Rev 1997;18(9):310-20.
5. Campo JV, Bridge J, Ehmann M, et al. Recurrent abdominal pain, anxiety, and depression in primary care. Pediatrics 2004;113(4):817-24.
6. Campo JV, Di Lorenzo C, Chiappetta L, et al. Adult outcomes of pediatric recurrent abdominal pain: do they just grow out of it? Pediatrics 2001;108(1):E1.-
7. Starfield B, Gross E, Wood M, et al. Psychosocial and psychosomatic diagnoses in primary care of children. Pediatrics 1980;66(2):159-67.
8. Hyams JS, Burke G, Davis PM, et al. Abdominal pain and irritable bowel syndrome in adolescents: a community-based study. J Pediatr 1996;129(2):220-6.
9. Campo JV, Comer DM, Jansen-McWilliams L, et al. Recurrent pain, emotional distress, and health service use in childhood. J Pediatr 2002;141(1):76-83.
10. Stickler GB, Murphy DB. Recurrent abdominal pain. Am J Dis Child 1979;133(5):486-9.
11. Rasquin-Weber A, Hyman PE, Cucchiara S, et al. Childhood functional gastrointestinal disorders. Gut 1999;45(suppl 2):II60-8.
12. Campo JV, Fritz G. A management model for pediatric somatization. Psychosomatics 2001;42(6):467-76.
13. Campo JV, Garber J. Somatization. In: Ammerman RT, Campo JV (eds). Handbook of pediatric psychology and psychiatry. Vol 1 Boston: Allyn and Bacon, 1998;137-61.
14. Fritz GK, Bergman AS. Child psychiatrists seen through pediatricians’ eyes: results of a national survey. J Am Acad Child Psychiatry 1985;24(1):81-6.
15. Huertas-Ceballos A, Macarthur C, Logan S. Dietary interventions for recurrent abdominal pain (RAP) in childhood. Cochrane Database Syst Rev 2002;(2):CD003019.-
16. Weydert JA, Ball TM, Davis MF. Systematic review of treatments for recurrent abdominal pain. Pediatrics 2003;111(1):e1-11.
17. Campo JV, Perel J, Lucas A, et al. Citalopram treatment of pediatric recurrent abdominal pain and comorbid internalizing disorders: An exploratory study (poster). Miami Beach, FL: American Academy of Child and Adolescent Psychiatry annual meeting, October 2003.
18. Geller B, Reising D, Leonard HL, et al. Critical review of tricyclic antidepressant use in children and adolescents. J Am Acad Child Adolesc Psychiatry 1999;38(5):513-6.
19. See MC, Birnbaum AH, Schechter CB, et al. Double-blind, placebo-controlled trial of famotidine in children with abdominal pain and dyspepsia: global and quantitative assessment. Dig Dis Sci 2001;46(5):985-92.
20. Kline RM, Kline JJ, Di Palma J, Barbero GJ. Enteric-coated, pH-dependent peppermint oil capsules for the treatment of irritable bowel syndrome in children. J Pediatr 2001;138(1):125-8.
21. Sanders MR, Rebgetz M, Morrison M, et al. Cognitive-behavioral treatment of recurrent nonspecific abdominal pain in children: an analysis of generalization, maintenance, and side effects. J Consult Clin Psychol 1989;57(2):294-300.
22. Sanders MR, Shepherd RW, Cleghorn G, Woolford H. The treatment of recurrent abdominal pain in children: a controlled comparison of cognitive-behavioral family intervention and standard pediatric care. J Consult Clin Psychol 1994;62(2):306-14.
Just three words—“My tummy hurts”—can mobilize a child’s parents into a high state of worry, especially on school days. They wonder: Is our child sick? Should he or she stay home? Why is this happening so often?
Although recurrent abdominal pain (RAP) is real, it usually is not caused by tissue damage or serious physical disease. When children with RAP are referred for psychiatric evaluation—often after extensive medical workups—we can help them and their parents manage the problem and function more normally. This article:
- describes physiologic mechanisms that may underlie recurrent GI distress
- discusses the high correlation of psychiatric comorbidities with RAP
- recommends judicious laboratory testing
- reviews evidence on medications and psychotherapies to improve RAP symptoms
- offers advice on building a therapeutic alliance with the patient and family.
Figure Comorbid anxiety and depressive disorders in children with RAP
Children with functional RAP are much more likely to be anxious or depressed than similar pain-free children. A recent blinded study followed 80 children ages 8 to 15 (42 with RAP and 38 controls) identified through screening at primary care pediatric offices. Each was assessed using the Schedule for Affective Disorders and Schizophrenia for School Age Children, Present and Lifetime version (K-SADS-PL). Percentage meeting diagnostic criteria
Source: Reference 5.
RAP: A ‘Functional’ disorder
RAP is a somatoform (or “functional”) disorder, defined as physical symptoms not fully explained by a medical condition, effects of a substance, or another mental disorder. Symptoms cause distress and/or functional impairment and are not intentionally produced.1
A patient with RAP experiences at least three episodes of abdominal pain over 3 months that interfere with daily activities.2 RAP affects 7% to 25% of school-aged children and adolescents,3 most of whom have a functional disorder.4
RAP is equally common among prepubertal boys and girls but more common among girls during adolescence.3 RAP can impair school attendance and performance and stigmatize a child as “sickly.”
Common comorbid symptoms
Physical. Besides stomach pain, children with RAP often experience headaches (including migraines), other GI symptoms, general aches and pains, dizziness, and fatigue.
Patients with RAP who do not experience GI bleeding, anemia, fever, weight loss, growth failure, or persistent vomiting most likely do not have a serious underlying disease.
Psychiatric. Children with RAP have much higher rates of anxiety (80%) and depressive (40%) disorders than do their unaffected peers (Figure).5 We have also seen higher levels of suicidal thinking in children with RAP in primary care settings compared with pain-free controls (14% vs. 4%, P = 0.04; unpublished data).
In most cases, psychiatric comorbidities appear to precede or coincide with RAP onset. Separation fears, generalized anxiety, and social anxiety in particular are common in patients with RAP yet are seldom recognized in medical settings.
Having childhood RAP increases the risk of anxiety, depression, and hypochondriacal fears in adulthood.6 We do not know whether early intervention prevents later disability.
Use of medical services. Abdominal pain accounts for 2% to 4% of all pediatric office visits.7 In one study, 8% of middle school and high school students said they had visited a physician for evaluation of stomach pain during the previous year.8 Children with RAP make more ambulatory health and mental health visits than peers9 and are at risk for unnecessary and potentially dangerous medical tests, procedures, and treatments, including abdominal surgery.10
Four functional GI disorders
To better characterize youths with functional RAP, symptom-based criteria have been developed and applied for functional GI disorders, defined as chronic or recurrent GI symptoms without explanatory structural or biochemical abnormalities.11 Four such disorders are relevant in children with RAP.
Irritable bowel syndrome (IBS): RAP with at least two of the following symptoms: relief with defecation, change in stool frequency, and change in stool form or appearance (occurs in approximately 50% of RAP cases).
Functional dyspepsia: RAP centered in the upper abdomen that is not associated with changes in bowel habits.
Abdominal migraine: Paroxysmal midline abdominal pain lasting 2 hours to several days with symptom-free intervals of weeks to months and at least two of the following: headache during episodes, photophobia during episodes, unilateral headache, aura, and a family history of migraine.
Functional abdominal pain: Continuous or nearly continuous abdominal pain for 6 months or more.
The reliability, validity, and clinical relevance of these criteria have not been demonstrated. Some children with RAP do not meet any criteria for a specific functional GI disorder.
Gut-brain connections. RAP may be associated with a heightened sensitivity to visceral sensations (visceral hyperalgesia) and a low pressure-pain threshold, leading to speculation that these children are hypersensitive to pain.
High rates of anxiety disorders and temperamental harm avoidance also are seen in patients with RAP, along with a tendency to develop pain when faced with unexpected events. Whether these children are more likely than others to perceive novel internal or external perceptions as threatening is open to debate.
Table 1
Recurrent abdominal pain: 8 steps to assessment and diagnosis
|
Serotonin communicates nociceptive information between the gut and brain and may mediate visceral hyperalgesia. Gut enterochromaffin cells contain more than 90% of the body’s total serotonin. They act as sensory transducers, releasing serotonin in response to increased intraluminal pressure or inflammation.
The released serotonin can cause abdominal discomfort by stimulating 5-HT 3 receptors on vagal afferents and can influence gut peristaltic activity by stimulating enteric afferents. The same serotonin transporter responsible for CNS serotonin reuptake is expressed throughout the gut.
A constellation of clues
The ideal RAP evaluation includes information from the child, parents, educators, and other health care professionals (Table 1).
Begin by acknowledging the patient’s suffering and the parent’s concerns; do not challenge the pain’s subjective reality. Rather than prejudging its cause, document the pain’s timing, context, and characteristics, and review the patient’s history. A constellation of clues is most suggestive of RAP (Table 2); single clues are not definitive.12,13
Table 2
Clues that suggest functional pain*
|
| * No single clue is definitive. |
| Source: Adapted from reference 12. |
Diagnostic testing. Be judicious in selecting diagnostic tests and procedures. Continuing to order studies in a haphazard effort to rule out disease can generate concerns that “the doctor doesn’t know what’s wrong” and heighten the family’s fear that a disease has been missed.
The process of “ruling out” physical disease may have no apparent end. Unless you are reasonably comfortable that a serious physical disease has not been missed, it is difficult to explain RAP to the patient and family and lay the foundation for intervention.
On the other hand, you must balance the importance of minimizing your own and the family’s anxiety about unrecognized disease against the physical and psychological risks and costs associated with medical tests and procedures.
Social assessment. Assess social and familial reinforcement (secondary gain) of the pain. Parents sometimes inadvertently encourage their children’s sick-role behaviors by providing excessive attention, rewards, or opportunities to avoid uncomfortable situations. RAP can become an excuse for poor performance (self-handicapping), particularly in children with a learning disorder.
How to deliver the diagnosis
Functional abdominal pain is essentially a clinical diagnosis that relies on presentation, course, and findings. As mentioned, a constellation of “clues” is most supportive, as is having typical IBS symptoms.
Before declaring the diagnosis, discuss with the family the patient’s physical, emotional, and behavioral symptoms and the context in which RAP developed. Doing so can help maintain your credibility and establish a consensus.
Once you declare the diagnosis, discuss it clearly and frankly. Families are not likely to be reassured if you do not offer a plausible explanation for the lack of physical findings.
Precautions. When a definitive diagnosis is not possible, acknowledge that uncertainty. Although you must discuss any recognized psychiatric comorbidity, attempting to “explain” that the disorder is causing the pain is usually impractical and intellectually dishonest.
Also, given the pervasive nature of stigma, do not convey embarrassment or unease about diagnosing functional RAP or any comorbid psychiatric disorder.
Follow-up testing. Once you diagnose functional RAP, further testing is generally not necessary. Tests might be indicated if you:
- receive new information
- observe a change in clinical status
- or are convinced that treatment will not work unless the family is reassured by further investigation.
Collaborative treatment
Reassurance and education. Reassurance that the patient does not have a serious physical disease is necessary but rarely sufficient. Explain that the child’s pain does not appear to reflect tissue damage and is not threatening. On the other hand, avoid giving excessive reassurance, particularly when obsessional illness worry and hypochondriacal fears are prominent. Address illness worry as a problem to be solved together.
Discuss with the patient and family what is known and not known about functional RAP, and encourage them to ask questions. This is an opportunity for you to instill hope and cultivate positive expectations, but avoid promising cure. Discuss the gut-brain connection and relevance of visceral hyperalgesia, including serotonin’s potential roles in RAP pathogenesis.
Partners in care. Collaborative treatment increases the likelihood of success. Discuss the importance of a therapeutic partnership, and clarify any areas of disagreement with the diagnosis or treatment plan.
Clearly delineate your roles and responsibilities and those of the patient, family, and other health care team members. Poor communication is pediatricians’ most common complaint about psychiatrists.14 Good interdisciplinary communication decreases the chance that treatment strategies will be duplicated, diluted, or misinterpreted.
Consolidate medical care with a single clinician—often the primary care physician—based on discussions with the patient, family, and health care team. The coordinating clinician can mediate between the school and family when tensions develop over poor attendance or requests for special treatment. It is often useful for this clinician to spell out:
- what constitutes a legitimate medical excuse for school absence
- who will legitimize excuses.
All parties should understand that the school will view an unexcused absence as truancy and act appropriately.
Diet and lifestyle. Encourage the patient to maintain a regular schedule and a healthy diet. Specific dietary interventions have not been proven effective, despite speculation that lack of dietary fiber or lactose intolerance might cause RAP.15,16 Also encourage adequate sleep and regular exercise.
Medication and psychotherapy
Because no strong evidence-based guidelines address pediatric RAP intervention, family preferences usually guide initial treatment decisions. This highlights the importance of good communication and a therapeutic partnership among the clinician, family, and patient.
Antispasmodics, acid reducers, and antidepressants are commonly prescribed for RAP, though none are well-supported in the literature and no controlled studies have gauged medication’s impact on psychiatric comorbidity.16
Antidepressants. Selective serotonin reuptake inhibitors (SSRIs) might help relieve RAP symptoms, but the evidence is inconclusive. SSRIs are considered potentially beneficial in RAP because they may help communicate nociceptive information between the gut and brain and mediate visceral hyperalgesia.
SSRIs at first may increase serotonin at the synapse, which one might assume would to worsen abdominal symptoms. However, ongoing SSRI use could “down-regulate” postsynaptic 5-HT3 receptors and desensitize postsynaptic cells to the effects of local serotonin.
Our group recently conducted a 12-week open trial of citalopram for functional pediatric RAP.17 The 25 participants received 10 mg/d the first week, then 20 mg/d thereafter if tolerated. At week 4, nonresponders and partial responders who were tolerating the medication began receiving 40 mg/d.
Table 3
SSRI daily dosing for pediatric RAP
| Drug | Starting dosage* | Target dosage† | Maximum dosage‡ |
|---|---|---|---|
| Citalopram | 10 mg | 20 mg | 40 to 60 mg |
| Escitalopram | 5 mg | 10 mg | 20 to 30 mg |
| Fluoxetine | 10 mg | 20 mg | 40 mg |
| Fluvoxamine | 50 mg | 100 mg | 300 mg |
| Sertraline | 25 mg | 50 mg | 200 mg |
| * First 3 to 7 days. | |||
| † If patient tolerates starting dosage, increase to target dosage. | |||
| ‡ If patient does not respond to target dosage in 2 to 3 weeks, consider increasing the dosage. | |||
At trial’s end, more than two-thirds of participants were taking 40 mg/d. We rated 21 of 25 patients (84%) as “much improved” or “very much improved,” using the Clinical Global Impression-Improvement scale. Abdominal pain, anxiety, depression, other somatic symptoms, and functional impairment all improved significantly during treatment. Suicidal thoughts diminished progressively from baseline, and no patient reported suicidal thinking at study’s end. Citalopram was generally well tolerated.
With SSRI treatment, start at a low dosage for 3 to 7 days (Table 3). If tolerated, increase to a typical therapeutic dosage. If symptoms fail to respond after 2 or 3 weeks, consider a higher dosage. A short course of an oral benzodiazepine (such as clonazepam, 0.25 mg bid) during the first weeks of SSRI treatment sometimes helps particularly anxious patients or those whose pain appears closely associated with anxiety or “stress.”
Pediatric gastroenterologists often prescribe a low-dose tricyclic antidepressant as first-line therapy, but we discourage this. TCAs lack efficacy in pediatric depression and pose a greater risk of side effects and safety concerns than SSRIs.18
Other agents have been tried for RAP-associated conditions:
Famotidine, a histamine type 2 receptor blocker, may reduce pain in children with dyspepsia and RAP.19
Peppermint oil reduced abdominal pain in one study of children with IBS but had little effect on other symptoms.20
Medications such as alosetron and tegaserod that interrupt serotonergic neurotransmission in the gut have shown benefit in adults with IBS but have not been studied in children.
Psychotherapy. A few small studies suggest that cognitive-behavioral therapies (CBT) are helpful in RAP, but CBT may be difficult to deliver in medical settings.21,22 A simplified “rehabilitative” approach that incorporates CBT principles involves having the clinician and patient view RAP as a challenge to be overcome, rather than a burden to be endured. Such an approach emphasizes the child’s fundamental strength and adaptability rather than vulnerabilities.
The goal of therapy is redirected from finding a cure to coping with and overcoming the problem. This approach challenges the notion that the child cannot resume normal function until the pain is completely gone. It encourages active, problem-focused coping, and discourages passive acceptance—which is associated with greater symptom burden and functional impairment.
Work with parents to reinforce the child’s health-promoting behaviors and minimize negative and social reinforcements (secondary gain) associated with RAP. Advise parents to:
- encourage and reward full school attendance
- avoid home-bound instruction
- expect the child to function despite physical distress
- insist that the child perform age-appropriate household chores and other responsibilities.
Self-management skills—such as relaxation training, hypnosis, biofeedback, and guided imagery—may help reduce pain and manage physiologic arousal.16 Deception strategies such as placebo or sham interventions are unethical and impractical.
Related resources
- Campo JV. Functional recurrent abdominal pain in children and adolescents. Digestive Health Matters 2003;12(3):15-7.
- International Foundation for Functional Gastrointestinal Disorders. www.iffgd.org
- University of Pittsburgh. Advanced Center for Interventions and Services Research on Early Onset Mood and Anxiety Disorders. www.moodykids.org
Drug brand names
- Alosetron • Lotronex
- Citalopram • Celexa
- Clonazepam • Klonopin
- Escitalopram • Lexapro
- Famotidine • Pepcid
- Fluoxetine • Prozac
- Fluvoxamine • Luvox
- Sertraline • Zoloft
- Tegaserod • Zelnorm
Disclosure
Dr. Campo’s work has been supported by the National Institute of Mental Health (grant MH01780) and in part by the Advanced Center for Interventions and Services Research on Early Onset Mood and Anxiety Disorders (grant MH66371). He also receives grants from Forest Pharmaceuticals and is a consultant to Eli Lilly and Co.
Just three words—“My tummy hurts”—can mobilize a child’s parents into a high state of worry, especially on school days. They wonder: Is our child sick? Should he or she stay home? Why is this happening so often?
Although recurrent abdominal pain (RAP) is real, it usually is not caused by tissue damage or serious physical disease. When children with RAP are referred for psychiatric evaluation—often after extensive medical workups—we can help them and their parents manage the problem and function more normally. This article:
- describes physiologic mechanisms that may underlie recurrent GI distress
- discusses the high correlation of psychiatric comorbidities with RAP
- recommends judicious laboratory testing
- reviews evidence on medications and psychotherapies to improve RAP symptoms
- offers advice on building a therapeutic alliance with the patient and family.
Figure Comorbid anxiety and depressive disorders in children with RAP
Children with functional RAP are much more likely to be anxious or depressed than similar pain-free children. A recent blinded study followed 80 children ages 8 to 15 (42 with RAP and 38 controls) identified through screening at primary care pediatric offices. Each was assessed using the Schedule for Affective Disorders and Schizophrenia for School Age Children, Present and Lifetime version (K-SADS-PL). Percentage meeting diagnostic criteria
Source: Reference 5.
RAP: A ‘Functional’ disorder
RAP is a somatoform (or “functional”) disorder, defined as physical symptoms not fully explained by a medical condition, effects of a substance, or another mental disorder. Symptoms cause distress and/or functional impairment and are not intentionally produced.1
A patient with RAP experiences at least three episodes of abdominal pain over 3 months that interfere with daily activities.2 RAP affects 7% to 25% of school-aged children and adolescents,3 most of whom have a functional disorder.4
RAP is equally common among prepubertal boys and girls but more common among girls during adolescence.3 RAP can impair school attendance and performance and stigmatize a child as “sickly.”
Common comorbid symptoms
Physical. Besides stomach pain, children with RAP often experience headaches (including migraines), other GI symptoms, general aches and pains, dizziness, and fatigue.
Patients with RAP who do not experience GI bleeding, anemia, fever, weight loss, growth failure, or persistent vomiting most likely do not have a serious underlying disease.
Psychiatric. Children with RAP have much higher rates of anxiety (80%) and depressive (40%) disorders than do their unaffected peers (Figure).5 We have also seen higher levels of suicidal thinking in children with RAP in primary care settings compared with pain-free controls (14% vs. 4%, P = 0.04; unpublished data).
In most cases, psychiatric comorbidities appear to precede or coincide with RAP onset. Separation fears, generalized anxiety, and social anxiety in particular are common in patients with RAP yet are seldom recognized in medical settings.
Having childhood RAP increases the risk of anxiety, depression, and hypochondriacal fears in adulthood.6 We do not know whether early intervention prevents later disability.
Use of medical services. Abdominal pain accounts for 2% to 4% of all pediatric office visits.7 In one study, 8% of middle school and high school students said they had visited a physician for evaluation of stomach pain during the previous year.8 Children with RAP make more ambulatory health and mental health visits than peers9 and are at risk for unnecessary and potentially dangerous medical tests, procedures, and treatments, including abdominal surgery.10
Four functional GI disorders
To better characterize youths with functional RAP, symptom-based criteria have been developed and applied for functional GI disorders, defined as chronic or recurrent GI symptoms without explanatory structural or biochemical abnormalities.11 Four such disorders are relevant in children with RAP.
Irritable bowel syndrome (IBS): RAP with at least two of the following symptoms: relief with defecation, change in stool frequency, and change in stool form or appearance (occurs in approximately 50% of RAP cases).
Functional dyspepsia: RAP centered in the upper abdomen that is not associated with changes in bowel habits.
Abdominal migraine: Paroxysmal midline abdominal pain lasting 2 hours to several days with symptom-free intervals of weeks to months and at least two of the following: headache during episodes, photophobia during episodes, unilateral headache, aura, and a family history of migraine.
Functional abdominal pain: Continuous or nearly continuous abdominal pain for 6 months or more.
The reliability, validity, and clinical relevance of these criteria have not been demonstrated. Some children with RAP do not meet any criteria for a specific functional GI disorder.
Gut-brain connections. RAP may be associated with a heightened sensitivity to visceral sensations (visceral hyperalgesia) and a low pressure-pain threshold, leading to speculation that these children are hypersensitive to pain.
High rates of anxiety disorders and temperamental harm avoidance also are seen in patients with RAP, along with a tendency to develop pain when faced with unexpected events. Whether these children are more likely than others to perceive novel internal or external perceptions as threatening is open to debate.
Table 1
Recurrent abdominal pain: 8 steps to assessment and diagnosis
|
Serotonin communicates nociceptive information between the gut and brain and may mediate visceral hyperalgesia. Gut enterochromaffin cells contain more than 90% of the body’s total serotonin. They act as sensory transducers, releasing serotonin in response to increased intraluminal pressure or inflammation.
The released serotonin can cause abdominal discomfort by stimulating 5-HT 3 receptors on vagal afferents and can influence gut peristaltic activity by stimulating enteric afferents. The same serotonin transporter responsible for CNS serotonin reuptake is expressed throughout the gut.
A constellation of clues
The ideal RAP evaluation includes information from the child, parents, educators, and other health care professionals (Table 1).
Begin by acknowledging the patient’s suffering and the parent’s concerns; do not challenge the pain’s subjective reality. Rather than prejudging its cause, document the pain’s timing, context, and characteristics, and review the patient’s history. A constellation of clues is most suggestive of RAP (Table 2); single clues are not definitive.12,13
Table 2
Clues that suggest functional pain*
|
| * No single clue is definitive. |
| Source: Adapted from reference 12. |
Diagnostic testing. Be judicious in selecting diagnostic tests and procedures. Continuing to order studies in a haphazard effort to rule out disease can generate concerns that “the doctor doesn’t know what’s wrong” and heighten the family’s fear that a disease has been missed.
The process of “ruling out” physical disease may have no apparent end. Unless you are reasonably comfortable that a serious physical disease has not been missed, it is difficult to explain RAP to the patient and family and lay the foundation for intervention.
On the other hand, you must balance the importance of minimizing your own and the family’s anxiety about unrecognized disease against the physical and psychological risks and costs associated with medical tests and procedures.
Social assessment. Assess social and familial reinforcement (secondary gain) of the pain. Parents sometimes inadvertently encourage their children’s sick-role behaviors by providing excessive attention, rewards, or opportunities to avoid uncomfortable situations. RAP can become an excuse for poor performance (self-handicapping), particularly in children with a learning disorder.
How to deliver the diagnosis
Functional abdominal pain is essentially a clinical diagnosis that relies on presentation, course, and findings. As mentioned, a constellation of “clues” is most supportive, as is having typical IBS symptoms.
Before declaring the diagnosis, discuss with the family the patient’s physical, emotional, and behavioral symptoms and the context in which RAP developed. Doing so can help maintain your credibility and establish a consensus.
Once you declare the diagnosis, discuss it clearly and frankly. Families are not likely to be reassured if you do not offer a plausible explanation for the lack of physical findings.
Precautions. When a definitive diagnosis is not possible, acknowledge that uncertainty. Although you must discuss any recognized psychiatric comorbidity, attempting to “explain” that the disorder is causing the pain is usually impractical and intellectually dishonest.
Also, given the pervasive nature of stigma, do not convey embarrassment or unease about diagnosing functional RAP or any comorbid psychiatric disorder.
Follow-up testing. Once you diagnose functional RAP, further testing is generally not necessary. Tests might be indicated if you:
- receive new information
- observe a change in clinical status
- or are convinced that treatment will not work unless the family is reassured by further investigation.
Collaborative treatment
Reassurance and education. Reassurance that the patient does not have a serious physical disease is necessary but rarely sufficient. Explain that the child’s pain does not appear to reflect tissue damage and is not threatening. On the other hand, avoid giving excessive reassurance, particularly when obsessional illness worry and hypochondriacal fears are prominent. Address illness worry as a problem to be solved together.
Discuss with the patient and family what is known and not known about functional RAP, and encourage them to ask questions. This is an opportunity for you to instill hope and cultivate positive expectations, but avoid promising cure. Discuss the gut-brain connection and relevance of visceral hyperalgesia, including serotonin’s potential roles in RAP pathogenesis.
Partners in care. Collaborative treatment increases the likelihood of success. Discuss the importance of a therapeutic partnership, and clarify any areas of disagreement with the diagnosis or treatment plan.
Clearly delineate your roles and responsibilities and those of the patient, family, and other health care team members. Poor communication is pediatricians’ most common complaint about psychiatrists.14 Good interdisciplinary communication decreases the chance that treatment strategies will be duplicated, diluted, or misinterpreted.
Consolidate medical care with a single clinician—often the primary care physician—based on discussions with the patient, family, and health care team. The coordinating clinician can mediate between the school and family when tensions develop over poor attendance or requests for special treatment. It is often useful for this clinician to spell out:
- what constitutes a legitimate medical excuse for school absence
- who will legitimize excuses.
All parties should understand that the school will view an unexcused absence as truancy and act appropriately.
Diet and lifestyle. Encourage the patient to maintain a regular schedule and a healthy diet. Specific dietary interventions have not been proven effective, despite speculation that lack of dietary fiber or lactose intolerance might cause RAP.15,16 Also encourage adequate sleep and regular exercise.
Medication and psychotherapy
Because no strong evidence-based guidelines address pediatric RAP intervention, family preferences usually guide initial treatment decisions. This highlights the importance of good communication and a therapeutic partnership among the clinician, family, and patient.
Antispasmodics, acid reducers, and antidepressants are commonly prescribed for RAP, though none are well-supported in the literature and no controlled studies have gauged medication’s impact on psychiatric comorbidity.16
Antidepressants. Selective serotonin reuptake inhibitors (SSRIs) might help relieve RAP symptoms, but the evidence is inconclusive. SSRIs are considered potentially beneficial in RAP because they may help communicate nociceptive information between the gut and brain and mediate visceral hyperalgesia.
SSRIs at first may increase serotonin at the synapse, which one might assume would to worsen abdominal symptoms. However, ongoing SSRI use could “down-regulate” postsynaptic 5-HT3 receptors and desensitize postsynaptic cells to the effects of local serotonin.
Our group recently conducted a 12-week open trial of citalopram for functional pediatric RAP.17 The 25 participants received 10 mg/d the first week, then 20 mg/d thereafter if tolerated. At week 4, nonresponders and partial responders who were tolerating the medication began receiving 40 mg/d.
Table 3
SSRI daily dosing for pediatric RAP
| Drug | Starting dosage* | Target dosage† | Maximum dosage‡ |
|---|---|---|---|
| Citalopram | 10 mg | 20 mg | 40 to 60 mg |
| Escitalopram | 5 mg | 10 mg | 20 to 30 mg |
| Fluoxetine | 10 mg | 20 mg | 40 mg |
| Fluvoxamine | 50 mg | 100 mg | 300 mg |
| Sertraline | 25 mg | 50 mg | 200 mg |
| * First 3 to 7 days. | |||
| † If patient tolerates starting dosage, increase to target dosage. | |||
| ‡ If patient does not respond to target dosage in 2 to 3 weeks, consider increasing the dosage. | |||
At trial’s end, more than two-thirds of participants were taking 40 mg/d. We rated 21 of 25 patients (84%) as “much improved” or “very much improved,” using the Clinical Global Impression-Improvement scale. Abdominal pain, anxiety, depression, other somatic symptoms, and functional impairment all improved significantly during treatment. Suicidal thoughts diminished progressively from baseline, and no patient reported suicidal thinking at study’s end. Citalopram was generally well tolerated.
With SSRI treatment, start at a low dosage for 3 to 7 days (Table 3). If tolerated, increase to a typical therapeutic dosage. If symptoms fail to respond after 2 or 3 weeks, consider a higher dosage. A short course of an oral benzodiazepine (such as clonazepam, 0.25 mg bid) during the first weeks of SSRI treatment sometimes helps particularly anxious patients or those whose pain appears closely associated with anxiety or “stress.”
Pediatric gastroenterologists often prescribe a low-dose tricyclic antidepressant as first-line therapy, but we discourage this. TCAs lack efficacy in pediatric depression and pose a greater risk of side effects and safety concerns than SSRIs.18
Other agents have been tried for RAP-associated conditions:
Famotidine, a histamine type 2 receptor blocker, may reduce pain in children with dyspepsia and RAP.19
Peppermint oil reduced abdominal pain in one study of children with IBS but had little effect on other symptoms.20
Medications such as alosetron and tegaserod that interrupt serotonergic neurotransmission in the gut have shown benefit in adults with IBS but have not been studied in children.
Psychotherapy. A few small studies suggest that cognitive-behavioral therapies (CBT) are helpful in RAP, but CBT may be difficult to deliver in medical settings.21,22 A simplified “rehabilitative” approach that incorporates CBT principles involves having the clinician and patient view RAP as a challenge to be overcome, rather than a burden to be endured. Such an approach emphasizes the child’s fundamental strength and adaptability rather than vulnerabilities.
The goal of therapy is redirected from finding a cure to coping with and overcoming the problem. This approach challenges the notion that the child cannot resume normal function until the pain is completely gone. It encourages active, problem-focused coping, and discourages passive acceptance—which is associated with greater symptom burden and functional impairment.
Work with parents to reinforce the child’s health-promoting behaviors and minimize negative and social reinforcements (secondary gain) associated with RAP. Advise parents to:
- encourage and reward full school attendance
- avoid home-bound instruction
- expect the child to function despite physical distress
- insist that the child perform age-appropriate household chores and other responsibilities.
Self-management skills—such as relaxation training, hypnosis, biofeedback, and guided imagery—may help reduce pain and manage physiologic arousal.16 Deception strategies such as placebo or sham interventions are unethical and impractical.
Related resources
- Campo JV. Functional recurrent abdominal pain in children and adolescents. Digestive Health Matters 2003;12(3):15-7.
- International Foundation for Functional Gastrointestinal Disorders. www.iffgd.org
- University of Pittsburgh. Advanced Center for Interventions and Services Research on Early Onset Mood and Anxiety Disorders. www.moodykids.org
Drug brand names
- Alosetron • Lotronex
- Citalopram • Celexa
- Clonazepam • Klonopin
- Escitalopram • Lexapro
- Famotidine • Pepcid
- Fluoxetine • Prozac
- Fluvoxamine • Luvox
- Sertraline • Zoloft
- Tegaserod • Zelnorm
Disclosure
Dr. Campo’s work has been supported by the National Institute of Mental Health (grant MH01780) and in part by the Advanced Center for Interventions and Services Research on Early Onset Mood and Anxiety Disorders (grant MH66371). He also receives grants from Forest Pharmaceuticals and is a consultant to Eli Lilly and Co.
1. American Psychiatric Association Diagnostic and statistical manual of mental disorders (4th ed, revised). Washington, DC: American Psychiatric Association, 2000.
2. Apley J, Naish N. Recurrent abdominal pains: a field survey of 1,000 school children. Arch Dis Child 1958;33(168):165-70.
3. Scharff L. Recurrent abdominal pain in children: a review of psychological factors and treatment. Clin Psychol Rev 1997;17(2):145-66.
4. Boyle JT. Recurrent abdominal pain: an update. Pediatr Rev 1997;18(9):310-20.
5. Campo JV, Bridge J, Ehmann M, et al. Recurrent abdominal pain, anxiety, and depression in primary care. Pediatrics 2004;113(4):817-24.
6. Campo JV, Di Lorenzo C, Chiappetta L, et al. Adult outcomes of pediatric recurrent abdominal pain: do they just grow out of it? Pediatrics 2001;108(1):E1.-
7. Starfield B, Gross E, Wood M, et al. Psychosocial and psychosomatic diagnoses in primary care of children. Pediatrics 1980;66(2):159-67.
8. Hyams JS, Burke G, Davis PM, et al. Abdominal pain and irritable bowel syndrome in adolescents: a community-based study. J Pediatr 1996;129(2):220-6.
9. Campo JV, Comer DM, Jansen-McWilliams L, et al. Recurrent pain, emotional distress, and health service use in childhood. J Pediatr 2002;141(1):76-83.
10. Stickler GB, Murphy DB. Recurrent abdominal pain. Am J Dis Child 1979;133(5):486-9.
11. Rasquin-Weber A, Hyman PE, Cucchiara S, et al. Childhood functional gastrointestinal disorders. Gut 1999;45(suppl 2):II60-8.
12. Campo JV, Fritz G. A management model for pediatric somatization. Psychosomatics 2001;42(6):467-76.
13. Campo JV, Garber J. Somatization. In: Ammerman RT, Campo JV (eds). Handbook of pediatric psychology and psychiatry. Vol 1 Boston: Allyn and Bacon, 1998;137-61.
14. Fritz GK, Bergman AS. Child psychiatrists seen through pediatricians’ eyes: results of a national survey. J Am Acad Child Psychiatry 1985;24(1):81-6.
15. Huertas-Ceballos A, Macarthur C, Logan S. Dietary interventions for recurrent abdominal pain (RAP) in childhood. Cochrane Database Syst Rev 2002;(2):CD003019.-
16. Weydert JA, Ball TM, Davis MF. Systematic review of treatments for recurrent abdominal pain. Pediatrics 2003;111(1):e1-11.
17. Campo JV, Perel J, Lucas A, et al. Citalopram treatment of pediatric recurrent abdominal pain and comorbid internalizing disorders: An exploratory study (poster). Miami Beach, FL: American Academy of Child and Adolescent Psychiatry annual meeting, October 2003.
18. Geller B, Reising D, Leonard HL, et al. Critical review of tricyclic antidepressant use in children and adolescents. J Am Acad Child Adolesc Psychiatry 1999;38(5):513-6.
19. See MC, Birnbaum AH, Schechter CB, et al. Double-blind, placebo-controlled trial of famotidine in children with abdominal pain and dyspepsia: global and quantitative assessment. Dig Dis Sci 2001;46(5):985-92.
20. Kline RM, Kline JJ, Di Palma J, Barbero GJ. Enteric-coated, pH-dependent peppermint oil capsules for the treatment of irritable bowel syndrome in children. J Pediatr 2001;138(1):125-8.
21. Sanders MR, Rebgetz M, Morrison M, et al. Cognitive-behavioral treatment of recurrent nonspecific abdominal pain in children: an analysis of generalization, maintenance, and side effects. J Consult Clin Psychol 1989;57(2):294-300.
22. Sanders MR, Shepherd RW, Cleghorn G, Woolford H. The treatment of recurrent abdominal pain in children: a controlled comparison of cognitive-behavioral family intervention and standard pediatric care. J Consult Clin Psychol 1994;62(2):306-14.
1. American Psychiatric Association Diagnostic and statistical manual of mental disorders (4th ed, revised). Washington, DC: American Psychiatric Association, 2000.
2. Apley J, Naish N. Recurrent abdominal pains: a field survey of 1,000 school children. Arch Dis Child 1958;33(168):165-70.
3. Scharff L. Recurrent abdominal pain in children: a review of psychological factors and treatment. Clin Psychol Rev 1997;17(2):145-66.
4. Boyle JT. Recurrent abdominal pain: an update. Pediatr Rev 1997;18(9):310-20.
5. Campo JV, Bridge J, Ehmann M, et al. Recurrent abdominal pain, anxiety, and depression in primary care. Pediatrics 2004;113(4):817-24.
6. Campo JV, Di Lorenzo C, Chiappetta L, et al. Adult outcomes of pediatric recurrent abdominal pain: do they just grow out of it? Pediatrics 2001;108(1):E1.-
7. Starfield B, Gross E, Wood M, et al. Psychosocial and psychosomatic diagnoses in primary care of children. Pediatrics 1980;66(2):159-67.
8. Hyams JS, Burke G, Davis PM, et al. Abdominal pain and irritable bowel syndrome in adolescents: a community-based study. J Pediatr 1996;129(2):220-6.
9. Campo JV, Comer DM, Jansen-McWilliams L, et al. Recurrent pain, emotional distress, and health service use in childhood. J Pediatr 2002;141(1):76-83.
10. Stickler GB, Murphy DB. Recurrent abdominal pain. Am J Dis Child 1979;133(5):486-9.
11. Rasquin-Weber A, Hyman PE, Cucchiara S, et al. Childhood functional gastrointestinal disorders. Gut 1999;45(suppl 2):II60-8.
12. Campo JV, Fritz G. A management model for pediatric somatization. Psychosomatics 2001;42(6):467-76.
13. Campo JV, Garber J. Somatization. In: Ammerman RT, Campo JV (eds). Handbook of pediatric psychology and psychiatry. Vol 1 Boston: Allyn and Bacon, 1998;137-61.
14. Fritz GK, Bergman AS. Child psychiatrists seen through pediatricians’ eyes: results of a national survey. J Am Acad Child Psychiatry 1985;24(1):81-6.
15. Huertas-Ceballos A, Macarthur C, Logan S. Dietary interventions for recurrent abdominal pain (RAP) in childhood. Cochrane Database Syst Rev 2002;(2):CD003019.-
16. Weydert JA, Ball TM, Davis MF. Systematic review of treatments for recurrent abdominal pain. Pediatrics 2003;111(1):e1-11.
17. Campo JV, Perel J, Lucas A, et al. Citalopram treatment of pediatric recurrent abdominal pain and comorbid internalizing disorders: An exploratory study (poster). Miami Beach, FL: American Academy of Child and Adolescent Psychiatry annual meeting, October 2003.
18. Geller B, Reising D, Leonard HL, et al. Critical review of tricyclic antidepressant use in children and adolescents. J Am Acad Child Adolesc Psychiatry 1999;38(5):513-6.
19. See MC, Birnbaum AH, Schechter CB, et al. Double-blind, placebo-controlled trial of famotidine in children with abdominal pain and dyspepsia: global and quantitative assessment. Dig Dis Sci 2001;46(5):985-92.
20. Kline RM, Kline JJ, Di Palma J, Barbero GJ. Enteric-coated, pH-dependent peppermint oil capsules for the treatment of irritable bowel syndrome in children. J Pediatr 2001;138(1):125-8.
21. Sanders MR, Rebgetz M, Morrison M, et al. Cognitive-behavioral treatment of recurrent nonspecific abdominal pain in children: an analysis of generalization, maintenance, and side effects. J Consult Clin Psychol 1989;57(2):294-300.
22. Sanders MR, Shepherd RW, Cleghorn G, Woolford H. The treatment of recurrent abdominal pain in children: a controlled comparison of cognitive-behavioral family intervention and standard pediatric care. J Consult Clin Psychol 1994;62(2):306-14.
Treating affective illness in patients with chronic pain
Ms. A, age 44, fell from a 3-foot stool while reaching for a high kitchen shelf and suffered severe neck flexion. Her initial pain persisted for weeks and then months, resulting in chronic neck pain aggravated by movement.
Over the past year, her doctor has prescribed numerous analgesics and muscle relaxants, including tramadol, hydrocodone, oxycodone, tizanidine, and nonsteroidal anti-inflammatory drugs (NSAIDs). Treatments at a pain clinic have included triggerpoint injections, cervical epidural corticosteroid injection, left-sided cervical medial branch blocks, transcutaneous electrical nerve stimulation, and physical therapy. None provided sustained relief.
During a pain clinic visit, Ms. A wept and said she was tired of living with pain. She acknowledged depression and agreed to psychiatric consultation.
As in Ms. A’s case, physicians often refer patients with chronic pain and affective symptoms for psychiatric evaluation. These patients are often fearful, angry, and suspicious of any suggestion that their physical discomfort has a psychiatric component. They typically believe their pain had a clear onset and therefore should have an end point. Many have experienced unproductive specialty evaluations and failed treatments.
To help you overcome these obstacles when treating patients with chronic pain and depression, we discuss:
- strategies to gain patients’ trust and build a therapeutic alliance
- how to assess their pain, depression, and suicide risk
- the role of psychotherapy in treating chronic pain
- and evidence for choosing effective, nonaddicting medications.
Psychiatric evaluation
Depression and pain are linked psychologically and biochemically, sharing neurotransmitters involved in both nociceptive pathways and mood, especially serotonin and norepinephrine.1,2 One-third to one-half of patients with chronic pain report comorbid depression,3 and more than one-half of depressed patients presenting to primary care physicians report only somatic symptoms—various pain complaints among the most common.4,5
Primary care doctors tend to refer chronic pain and depression cases to psychiatrists when:
- patients are preoccupied with medication, have not followed treatment recommendations, or do not respond to treatment as expected
- extensive medical evaluations reveal few or equivocal findings
- somatic complaints are vague and diffuse, or there is marked disparity between pain complaints/disability and objective findings.6,7
Assessing pain. In the initial assessment, validate the patient’s pain experience by asking about the location, quality, and severity of pain. The visual analogue scale (VAS) is commonly used to measure pain severity. The patient marks a spot on a line from “no pain” to “worst possible pain,” or—on a numbered VAS—from 0 (no pain) to 10 (extreme pain). The least and most severe pain over the preceding month can be ranked as baseline values.8
Be sensitive to the patient’s fear that you will attribute the pain to psychosocial issues or imply that “the pain is in your head.” Emphasize that you intend to evaluate the “whole person,” not just the part that hurts. Focus on how the pain affects the patient’s lifestyle—rather than its cause—and explore medication use patterns.
Assessing depression. The word “depression” is emotionally charged for chronic pain patients, who view affective symptoms—if they acknowledge them at all—as secondary to pain. They may strongly resist treatment for anything but pain. One way to defuse this defensiveness is to avoid attributing the pain to stress or depression.
Begin by assessing vegetative symptoms, which overlap in chronic pain and depression. The Beck Depression Inventory-II (Beck-II) may be a useful screening tool in a busy practice; the short form (13 questions) takes about 5 minutes to complete.9
Explore cognitive and behavioral symptoms such as concentration, pleasure and interest level, activity, and self-esteem. Review the chronology of pain onset, mood changes, and stressors (proximate, remote, and cumulative).
Seek clues to endogenous factors by asking about past affective episodes, response to antidepressants, and family history of psychopathology. Substances that may induce depression include reserpine, interferon, and antiparkinsonian agents. Screen for potential organic mood disorders, such as depression secondary to hypothyroidism, corticosteroid use, Parkinson’s disease, lupus, HIV infection, or cerebrovascular disease. Where appropriate, obtain collateral information from family or friends.
Assessing suicide risk. Chronic pain patients may be at greater risk of suicide than the general population. Besides pain, other risk factors for suicide—such as major depression, anxiety disorders, alcohol/substance abuse, sleep disturbances, male gender, diminished social support, and recent loss—are common among these patients.10,11
Screen chronic pain patients with suicidal ideation for these risk factors. Interventions include:
- aggressively treat associated depression, anxiety, or insomnia
- elicit support from family or other caregivers
- pay close attention to talk about suicide
- hospitalize when necessary
- and, of course, treat pain.
Case continued: No stranger to depression
Ms. A’s psychiatric assessments revealed a pain severity ranking of 9 on a 1-to-10 scale, frequent crying, hopelessness, disrupted sleep, low energy, limited ability to concentrate, and fleeting suicidal thoughts. Her history included counseling during her first marriage and severe depression after separation from her second husband 3 years ago. An 8-week trial of fluoxetine, 20 mg/d, did not improve her depression then.
On examination, she displayed obvious pain behavior, constantly shifting her neck position and moving about the room. Her affect was tearful and her mood depressed. She was taking the NSAID celecoxib, 100 mg bid, and the skeletal muscle relaxant tizanidine, 4 mg tid. She was no longer using opioids and had no history of alcohol or illicit drug abuse.
Based on this assessment, the psychiatrist diagnosed Ms. A as having pain disorder with medical and psychological features, including symptom amplification and depression.
Table 1
4 treatment goals for patients with chronic pain and depression
|
Educating the patient
As part of your assessment, explain the reciprocal effects of depression and pain. Acknowledge that:
- chronic pain is different from acute pain, although the patient’s pain experience is the same
- treatment often becomes part of the problem in chronic pain.
Doctors tend to apply acute pain treatments chronically, risking long-term effects of polypharmacy to achieve short-term relief. Depressed patients may be more likely than nondepressed patients to receive opioids for chronic pain,12 and opioids and benzodiazepines may have depressive effects, as reflected by DSM-IV-TR’s inclusion of criteria for “opioid-induced mood disorder” and “sedative-, hypnotic-, or anxiolytic-induced mood disorder.”
To reduce patients’ resistance to antidepressants, reiterate any history of cumulative stressors and affective episodes unrelated to pain. Try using an analogy, such as “stress and pain are like waves on a rock” that eventually damage mood and coping mechanisms, or depression complicating pain is like having “too much on one’s plate.”
Finally, help patients understand that chronic pain is managed, not cured. Encourage them to set treatment goals beyond reducing pain (Table 1) and to make the transition from “patient with pain” to “client managing pain.”
Table 2
Dosing antidepressants and anticonvulsants
for chronic pain and depression
| Drug | Starting (mg/d) | Target (mg/d) | Administration tips |
|---|---|---|---|
| TCAs | Check serum levels for dosages ≥150 mg/d (nortriptyline 100 mg/d) to assess rapid metabolism, adherence, or toxic levels | ||
| Amitriptyline | 10 to 25 | 75 to 300 | |
| Clomipramine | 10 to 25 | 75 to 250 | |
| Desipramine | 10 to 25 | 75 to 200 | |
| Doxepin | 10 to 25 | 75 to 300 | |
| Imipramine | 10 to 25 | 75 to 300 | |
| Nortriptyline | 10 to 25 | 40 to 200 | |
| SNRI | |||
| Venlafaxine | 37.5 to 75 | 75 to 375 | Use XR form to minimize side effects and for once-daily dosing |
| SSRIs | |||
| Citalopram | 10 to 20 | 40 to 60 | |
| Fluoxetine | 10 to 20 | 20 to 80 | May increase carbamazepine, TCA blood levels and inhibit efficacy of codeine, dihydrocodeine, and hydrocodone |
| Paroxetine | 10 to 20 | 20 to 60 | Same as fluoxetine |
| Anticonvulsants | |||
| Carbamazepine | 200 | 800 to 1,200 | Check blood levels; may increase clomipramine levels, reduce acetaminophen, contraceptive levels |
| Clonazepam | 0.5 | 1 to 2 | Habituating potential with chronic use |
| Gabapentin | 300 to 900 | 3,600 to 4,800 | Blood monitoring not necessary |
| Valproate | 250 | 750 to 2,500 (maximum dosage 60 mg/kg/d) | Check blood levels (trough plasma level 50 to 100 μg/mL) |
| TCA: tricyclic antidepressant | |||
| SNRI: serotonin-norepinephrine reuptake inhibitor | |||
| SSRI: selective serotonin reuptake inhibitor | |||
Prescribing principles
Before adding any new pain medications, consider reducing dosages or discontinuing opioids or benzodiazepines and other substances the patient may be taking. Opioid use is associated with risks of dependence, addiction, and side effects including somnolence, cognitive impairment, and reduced activity that amplify depressive symptoms.
Benzodiazepines can generally be tapered by 10% per day, although you may need to extend the final taper over 3 to 4 days or longer, depending upon chronicity of use. Opioids may be tapered by 20% over 5 to 7 days. Breakthrough doses may be needed for marked withdrawal symptoms. Converting to longer half-life agents—such as clonazepam for benzodiazepines or methadone for opioids—often aids tapering, although other agents and strategies exist.13
To gauge patient attempts at self-medication, monitor use of alcohol or illicit drugs with urine screening. For patients with a substantial history of substance abuse or positive toxicology screens, monitor randomly every 2 to 4 weeks.
On the other hand, undertreated pain also may impair mood and function.1 If pain and mood improve and problematic drug-related behaviors resolve with increased opioid analgesia, consider maintaining opioids with regular re-evaluation of mood, coping, and medication adherence.11 Transfer from immediate-release to controlled-release opioids to reduce dosing frequency, clockwatching, and the likelihood of inter-dose pain escalation. In general, maintain and optimize the dosage of nonaddictive analgesics such as NSAIDs, anticonvulsants, or antidepressants.
Case continued: Switching medication
The psychiatrist started Ms. A on nortriptyline, 25 mg at bedtime, to be increased after 3 nights to 50 mg at bedtime. Tizanidine, which had been ineffective, was discontinued to reduce the risk of xerostomia and oversedation in combination with nortriptyline. If tolerated, nortriptyline was to be further increased by 25 mg every 3 days to an initial target dosage of 100 mg at bedtime. The psychiatrist explained to Ms. A that it might take 4 to 6 weeks to gauge the medication’s efficacy.
Psychoeducation addressed the importance of stress reduction, prioritizing commitments, and setting limits on other people’s expectations. The door was left open to future psychotherapeutic exploration of past cumulative stressors.
Because antidepressants may provide an analgesic effect,6,14 they are often used to treat affective symptoms in chronic pain. Headache and neuralgia tend to respond to antidepressants more robustly than do arthritis and low-back pain. Although some patients respond to low-dose antidepressants, a definitive trial requires full doses for 6 to 8 weeks (Table 2).
Matching a patient’s symptoms with medication side effects is useful when choosing antidepressants (Table 3). So-called “adverse” effects may have a corresponding benefit, depending on the clinical presentation. For example, a moreactivating antidepressant—such as the selective serotonin reuptake inhibitor (SSRI) fluoxetine—may help a patient with fatigue, whereas a moresedating agent—such as a tricyclic antidepressant (TCA) or mirtazapine—may improve sleep for a patient with insomnia.
Psychosocial therapies such as cognitive-behavioral therapy (CBT) or relaxation training (Table 4) may help patients with chronic pain to:
- process covert emotions such as fear and anger as well as guilt, loss, and disability
- reduce somatic preoccupation that is aggravating the pain
- adhere to treatment.
Evidence strongly supports using relaxation techniques to reduce chronic pain in many medical conditions and hypnosis to ameliorate cancer pain. CBT and biofeedback appear moderately effective in relieving chronic pain.15 CBT is significantly more effective than waiting list control conditions for relieving chronic nonheadache pain in measures of pain experience, mood/affect, cognitive coping and appraisal, pain behavior and activity level, and social role functioning.16
Pain and opioid medications can impair concentration and affective processing, so initial psychotherapy may need to be supportive while you provide other treatments and simplify medication regimens. Eventually the patient may be ready to address underlying issues that may be contributing to the pain syndrome, such as a history of abuse. However, it is important to address this potentially destabilizing subject only after carefully gauging a patient’s defenses and readiness.
Case continued: A bump in the road
The psychiatrist saw Ms. A 18 months later. Interim history revealed that her pain and mood improved on nortriptyline, 100 mg at bedtime. When she stopped taking nortriptyline 5 months earlier, her neck pain increased and she experienced a “deep blue mood.” Her physician restarted the nortriptyline.
At follow up, Ms. A reported no depressive symptoms and very little neck pain. The psychiatrist discussed with her depression’s relapse rate and the importance of continuing antidepressant therapy. As Ms. A was feeling much better and functioning normally, the psychiatrist decided additional psychotherapeutic intervention was not necessary.
Antidepressant options
TCAs provide analgesia via descending regulatory pathways by inhibiting serotonin and norepinephrine reuptake.17 When using TCAs for chronic pain, start with 10 to 25 mg at bedtime and increase by 10 to 25 mg every 3 to 7 days as tolerated. Increase incrementally until the pain responds or to the full antidepressant dosage (Table 2). Drug levels (when available) can help you provide an appropriate trial and monitor the patient’s adherence.
If the pain does not respond after 6 to 8 weeks, consider switching to another dual-action agent such as venlafaxine or to an SSRI.
SNRIs. Venlafaxine is a serotonin and norepineph-rine reuptake inhibitor (SNRI) with less-troublesome side effects than TCAs. It is structurally similar to tramadol18 and has combined serotonin and norepinephrine inhibition at dosages >75 mg/d. Although venlafaxine is not indicated for chronic pain, some studies have suggested possible benefits, including long-term analgesia, reduced polyneuropathic pain, and migraine prophylaxis.19-21 Venlafaxine may be a reasonable first or second choice for treating depression in patients with chronic pain, especially headache.14
Duloxetine—another SNRI—awaits FDA approval. Some studies have suggested that duloxetine improves painful physical symptoms as well as mood and functioning in major depression.22
SSRIs may be effective for certain types of pain, but the evidence is conflicting. Results of 41controlled trials support TCAs’ analgesic efficacy for neuropathic pain, headache, and central and post-stroke pain, whereas SSRIs’ analgesic efficacy varies from study to study. Comparisons of TCAs and SSRIs as analgesics uniformly show TCAs to be more effective, with the SSRIs often showing no analgesic effect.
Of three controlled trials of SSRIs for diabetic neuropathy, one showed fluoxetine similar to placebo, and two smaller studies showed paroxetine and citalopram more effective than placebo. Fluoxetine has shown analgesic effect for fibromyalgia in one study, but no effect in another. Citalopram showed no analgesic effect for fibromyalgia in another study.23
A prospective, double-blind study comparing fluoxetine, sertraline, paroxetine, and venlafaxine for migraines reported moderate to significant improvement in less than one-half of SSRI-treated patients vs two-thirds of venlafaxine-treated patients.21 SSRIs are no longer recognized by the International Headache Society as primary preventative medications for migraine.
Fluoxetine may help chronic daily headache, and paroxetine and citalopram may be useful for diabetic neuropathy. However, one cannot generalize that all SSRIs are similarly effective as analgesics.14
SSRIs have fewer side effects than TCAs and are less dangerous in overdose. In general, however, SSRIs are a second-line treatment for pain, to be used when dual-action agents pose disadvantageous side effects (Table 3) or have been poorly tolerated or ineffective.
Table 3
Antidepressant side effects:
Limitations and potential benefits in chronic pain
| Side effects/agents | Problems | Conditions potentially benefited | Possible alternatives |
|---|---|---|---|
| Anticholinergic TCAs | Xerostomia, constipation, urinary slowing (esp. when combined with opioids) | Diarrhea-predominant irritable bowel syndrome | SSRIs, nefazodone, venlafaxine |
| Sedation TCAs, mirtazapine, nefazodone, trazodone | Excessive sedation, cognitive impairment, driving risk (esp. when combined with opioids, benzodiazepines) | Pain with insomnia | SSRIs, venlafaxine, bupropion |
| Insomnia SSRIs, venlafaxine | Pain with pre-existing insomnia; equivocal analgesic effects | Excess sedation related to depression, polypharmacy for pain | TCAs, mirtazapine, trazodone, nefazodone |
| Orthostasis TCAs (esp. with methadone), nefazodone | Falls, especially in elderly patients | —— | Nortriptyline, SSRIs bupropion, venlafaxine |
| Weight gain TCAs, mirtazapine | Pain patients are often sedentary, get limited exercise | Pain and depression with weight loss | Bupropion, fluoxetine |
| Hypertension Bupropion, venlafaxine | Pre-existing hypertension | ? Hypotensive state | Citalopram (hypertensive side effects infrequent) |
| Cardiac TCAs | ECG abnormalities, conduction delays, arrhythmias aggravate pre-existing cardiac abnormalities; avoid if recent MI | ——— | SSRIs, bupropion |
| Overdose lethality TCAs | Prominent suicidal ideation | —— | SSRIs, venlafaxine |
| Seizures Esp. maprotiline, clomipramine, bupropion | Lower seizure threshold, aggravation of seizure disorders | ——- | SSRIs |
| Sexual dysfunction SSRIs | Pre-existing sexual dysfunction secondary to pain, medications, stress; equivocal analgesic effects | ——- | Bupropion, nefazodone, mirtazapine |
Table 4
How psychosocial therapies can help treat chronic pain and depression
| Therapies | Purpose/benefits |
|---|---|
| Behavioral therapy | Increase activity and learn to balance activity with limitations Reduce pain behaviors and analgesic use Decrease dependency and secondary gain |
| Cognitive-behavioral therapy | Identify automatic thoughts Challenge negative cognitions, catastrophizing Substitute and rehearse positive thoughts, capabilities Transition from patient role to self-care |
| Couples’ therapy | Assist adaptation to role changes Diminish spousal solicitousness or excessive caretaking Increase communication |
| Biofeedback, relaxation, imagery | Adjunctive role in pain management Reduce tension, comorbid anxiety |
| Hypnosis | Dissociate awareness of pain Substitute, displace, reinterpret pain sensations |
| Vocational rehabilitation | Increase activity, ability to distract Regain sense of control, identity, and productivity Increase socialization |
| Pain management program | Multiple treatment effects Useful for complex pain with affective states |
Monoamine oxidase inhibitors (MAOIs) may have some efficacy for neuropathy and headache, but the need for a tyramine-free diet and potential for drug-drug interactions limit their usefulness. Co-administering an MAOI and meperidine is always contraindicated, as this combination can produce fever, delirium, seizures, circulatory collapse, and death. Similarly, avoid using an MAOI with any other antidepressant.
Others. Evidence is very limited on using other antidepressants such as trazodone, nefazodone, bupropion, and mirtazapine in chronic pain:
- Trazodone may help pediatric migraine, but it is not a consistent analgesic and may not be well-tolerated.
- Case reports suggest bupropion may help with headaches and chronic low-back pain.14
- Mirtazapine and trazodone may be useful adjuncts for treating insomnia in depressed patients with chronic pain.
Other options
Anticonvulsants appear useful for neuropathic pain and are appropriate for chronic pain patients who cannot tolerate TCAs.24 Like TCAs, anticonvulsants are not addictive. Unlike TCAs, anticonvulsants may help stabilize other affective illnesses that may coexist with chronic pain, including bipolar disorder, schizoaffective disorder, and impulsivity/aggression related to dementia or personality disorder.6 If the starting dosage is not effective within 1 week, increase gradually every 2 to 3 days to target dosages comparable to those for anticonvulsant efficacy.
Carbamazepine and gabapentin are recommended first-line medications for neuropathy. Carbamazepine is indicated for treating trigeminal neuralgia, although its cytochrome P-450 3A3/4 isoenzyme induction may reduce serum levels of acetaminophen, opioids, and oral contraceptives. Gabapentin, although not clearly beneficial for bipolar disorder, has anxiolytic effects and a benign side-effect profile, which may help patients with chronic pain.
Valproate can help prevent migraines. Clonazepam can help reduce anxiety and restless legs syndrome but may be habituating. Anticonvulsants’ common adverse effects include sedation, GI upset, dizziness, and fatigue.
Lithium has known efficacy for mood stabilization in bipolar disorder and can ameliorate cluster headaches.
Antipsychotics. Evidence is sparse on whether antipsychotics have analgesic activity. Their side effects generally limit their usefulness to treating pain in patients with psychosis or delirium.6
Stimulants such as dextroamphetamine and methylphenidate can be helpful adjuncts for treating depression, especially for medical inpatients who require a rapid therapeutic response. Stimulants may reduce fatigue or excessive sedation and improve concentration in patients receiving opioids for chronic pain. They also may have analgesic effects when combined with opioids. Potential adverse effects include appetite suppression, anxiety or agitation, confusion, tics, and addiction.6
Precautions. The muscle relaxant carisoprodol is associated with potential dependence and withdrawal. Cyclobenzaprine, another muscle relaxant, has a TCA-like structure and can be lethal in overdose. Baclofen can be useful for chronic pain related to spasticity, although psychotic depression and mania have been reported with abrupt withdrawal.6
Related resources
- American Academy of Pain Medicine. www.painmed.org/
- American Academy of Pain Management. www.aapainmanage.org
- Pain.com: continuing medical education for anesthesiology professionals. www.pain.com/index.cfm
- Kerns RD, Turk DC, Rudy TE. The West Haven-Yale Multidimensional Pain Inventory (WHYMPI). Pain 1985;13:345-56
Drug brand names
- Amitriptyline • Elavil
- Baclofen • Lioresal
- Bupropion • Wellbutrin
- Carbamazepine • Tegretol
- Carisoprodol • Soma
- Celecoxib • Celebrex
- Citalopram • Celexa
- Clomipramine • Anafranil
- Clonazepam • Klonopin
- Cyclobenzaprine • Flexeril
- Desipramine • Norpramin
- Dihydrocodeine • Synalgos-DC
- Doxepin • Sinequan
- Duloxetine • Cymbalta
- Fluoxetine • Prozac
- Gabapentin • Neurontin
- Hydrocodone • Vicodin, Lortab
- Imipramine • Tofranil
- Lithium • Eskalith CR, Lithobid
- Maprotiline • Ludiomil
- Meperidine • Demerol
- Methadone • Dolophine
- Mirtazapine • Remeron
- Nefazodone • Serzone
- Nortriptyline • Pamelor
- Oxycodone • OxyContin
- Paroxetine • Paxil
- Sertraline • Zoloft
- Tizanidine • Zanaflex
- Tramadol • Ultram
- Trazodone • Desyrel
- Valproate • Depakote
- Venlafaxine • Effexor, Effexor XR
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Campbell LC, Clauw DJ, Keefe FJ. Persistent pain and depression: a biopsychosocial perspective. Biol Psychiatry 2003;54:399-409.
2. Fishbain DA, Cutler R, Rosomoff HL, et al. Chronic painassociated depression: antecedent or consequence of chronic pain? A review. Clin J Pain 1997;13(2):116-37.
3. Banks SM, Kerns RD. Explaining high rates of depression in chronic pain: a diathesis-stress framework. Psychol Bull 1996;119:95-110.
4. Simon GE, VonKorff M, Piccinelli M, et al. An international study of the relation between somatic symptoms and depression. N Engl J Med 1999;341(18):1329-35.
5. Kroenke K, Price RK. Symptoms in the community. Prevalence, classification, and psychiatric comorbidity. Arch Intern Med. 1993;153:2474-80.
6. Leo RJ. Concise guide to pain management for psychiatrists. Arlington, Va: American Psychiatric Publishing, Inc., 2003.
7. Sullivan MD, Turner JA, Romano J. Chronic pain in primary care. Identification and management of psychosocial factors. J Fam Pract 1991;32(2):193-9.
8. Holmgren A, Wise MG, Bouckoms AJ. Pain management. In: Wise MG, Rundell JR (eds). Psychiatry in the medically ill (2nd ed). Washington, DC: American Psychiatric Publishing Inc., 2002;989-1013.
9. Naifeh KH. Psychometric testing in functional GI disorders in: Olden K (ed). Handbook of functional GI disorders. New York: Marcel Dekker, 1996;79-126.
10. Fishbain DA. The association of chronic pain and suicide. Semin Clin Neuropsychiatry 1999;4(3):221-7.
11. Fishbain DA. Medico-legal rounds: medical-legal issues and breaches of “standards of medical care” in opioid tapering for alleged opioid addiction. Pain Med 2002;3(2):135-42.
12. Doan BD, Wadden NP. Relationships between depressive symptoms and descriptions of chronic pain. Pain 1989;36:75-84.
13. Franklin JE, Leamon MH, Frances RJ. Substance-related disorders. In: Wise MG, Rundell JR (eds). Psychiatry in the medically ill (2nd ed). Washington DC: American Psychiatric Publishing, 2002;417-53.
14. Ansari A. The efficacy of newer antidepressants in the treatment of chronic pain: a review of current literature. Harv Rev Psychiatry 2000;7(5):257-77.
15. NIH Technology Assessment Panel. Integration of behavioral relaxation approaches into the treatment of chronic pain and insomnia. JAMA 1996;276(4):313-18.
16. Morley S, Eccleston C, Williams A. Systematic review and metaanalysis of randomized controlled trials of cognitive behaviour therapy and behaviour therapy for chronic pain in adults, excluding headache. Pain 1999;80:1-13
17. Magni G. On the relationship between chronic pain and depression when there is no organic lesion. Pain 1987;31:1-21.
18. Markowitz JS, Patrick KS. Venlafaxine-tramadol similarities. Med Hypotheses 1998;5:167-8
19. Bradley RH, Barkin RL, Jerome J, et al. Efficacy of venlafaxine for the long-term treatment of chronic pain with associated major depressive disorder. Am J Ther 2003;10(5):318-23.
20. Sindrup SH, Bach FW, Madsen C, et al. Venlafaxine vs. imipramine in painful polyneuropathy—a randomized controlled trial. Neurology 2003;60:1284-9.
21. Kathpal GS. Role of SSRIs in the management of migraine. Headache Quarterly 1998;9:265-6.
22. Mallinckrodt CH, Goldstein DJ, Detke MJ, et al. Duloxetine: a new treatment for the emotional and physical symptoms of depression. Primary Care Companion J Clin Psychiatry 2003;5(1):19-28.
23. Lynch ME. Antidepressants as analgesics: a review of randomized control trials. J Psychiatry Neurosci 2001;26-36.
24. Swerdlow M. Anticonvulsant drugs and chronic pain. Clin Neuropharmacol 1984;7(1):51-82.
Ms. A, age 44, fell from a 3-foot stool while reaching for a high kitchen shelf and suffered severe neck flexion. Her initial pain persisted for weeks and then months, resulting in chronic neck pain aggravated by movement.
Over the past year, her doctor has prescribed numerous analgesics and muscle relaxants, including tramadol, hydrocodone, oxycodone, tizanidine, and nonsteroidal anti-inflammatory drugs (NSAIDs). Treatments at a pain clinic have included triggerpoint injections, cervical epidural corticosteroid injection, left-sided cervical medial branch blocks, transcutaneous electrical nerve stimulation, and physical therapy. None provided sustained relief.
During a pain clinic visit, Ms. A wept and said she was tired of living with pain. She acknowledged depression and agreed to psychiatric consultation.
As in Ms. A’s case, physicians often refer patients with chronic pain and affective symptoms for psychiatric evaluation. These patients are often fearful, angry, and suspicious of any suggestion that their physical discomfort has a psychiatric component. They typically believe their pain had a clear onset and therefore should have an end point. Many have experienced unproductive specialty evaluations and failed treatments.
To help you overcome these obstacles when treating patients with chronic pain and depression, we discuss:
- strategies to gain patients’ trust and build a therapeutic alliance
- how to assess their pain, depression, and suicide risk
- the role of psychotherapy in treating chronic pain
- and evidence for choosing effective, nonaddicting medications.
Psychiatric evaluation
Depression and pain are linked psychologically and biochemically, sharing neurotransmitters involved in both nociceptive pathways and mood, especially serotonin and norepinephrine.1,2 One-third to one-half of patients with chronic pain report comorbid depression,3 and more than one-half of depressed patients presenting to primary care physicians report only somatic symptoms—various pain complaints among the most common.4,5
Primary care doctors tend to refer chronic pain and depression cases to psychiatrists when:
- patients are preoccupied with medication, have not followed treatment recommendations, or do not respond to treatment as expected
- extensive medical evaluations reveal few or equivocal findings
- somatic complaints are vague and diffuse, or there is marked disparity between pain complaints/disability and objective findings.6,7
Assessing pain. In the initial assessment, validate the patient’s pain experience by asking about the location, quality, and severity of pain. The visual analogue scale (VAS) is commonly used to measure pain severity. The patient marks a spot on a line from “no pain” to “worst possible pain,” or—on a numbered VAS—from 0 (no pain) to 10 (extreme pain). The least and most severe pain over the preceding month can be ranked as baseline values.8
Be sensitive to the patient’s fear that you will attribute the pain to psychosocial issues or imply that “the pain is in your head.” Emphasize that you intend to evaluate the “whole person,” not just the part that hurts. Focus on how the pain affects the patient’s lifestyle—rather than its cause—and explore medication use patterns.
Assessing depression. The word “depression” is emotionally charged for chronic pain patients, who view affective symptoms—if they acknowledge them at all—as secondary to pain. They may strongly resist treatment for anything but pain. One way to defuse this defensiveness is to avoid attributing the pain to stress or depression.
Begin by assessing vegetative symptoms, which overlap in chronic pain and depression. The Beck Depression Inventory-II (Beck-II) may be a useful screening tool in a busy practice; the short form (13 questions) takes about 5 minutes to complete.9
Explore cognitive and behavioral symptoms such as concentration, pleasure and interest level, activity, and self-esteem. Review the chronology of pain onset, mood changes, and stressors (proximate, remote, and cumulative).
Seek clues to endogenous factors by asking about past affective episodes, response to antidepressants, and family history of psychopathology. Substances that may induce depression include reserpine, interferon, and antiparkinsonian agents. Screen for potential organic mood disorders, such as depression secondary to hypothyroidism, corticosteroid use, Parkinson’s disease, lupus, HIV infection, or cerebrovascular disease. Where appropriate, obtain collateral information from family or friends.
Assessing suicide risk. Chronic pain patients may be at greater risk of suicide than the general population. Besides pain, other risk factors for suicide—such as major depression, anxiety disorders, alcohol/substance abuse, sleep disturbances, male gender, diminished social support, and recent loss—are common among these patients.10,11
Screen chronic pain patients with suicidal ideation for these risk factors. Interventions include:
- aggressively treat associated depression, anxiety, or insomnia
- elicit support from family or other caregivers
- pay close attention to talk about suicide
- hospitalize when necessary
- and, of course, treat pain.
Case continued: No stranger to depression
Ms. A’s psychiatric assessments revealed a pain severity ranking of 9 on a 1-to-10 scale, frequent crying, hopelessness, disrupted sleep, low energy, limited ability to concentrate, and fleeting suicidal thoughts. Her history included counseling during her first marriage and severe depression after separation from her second husband 3 years ago. An 8-week trial of fluoxetine, 20 mg/d, did not improve her depression then.
On examination, she displayed obvious pain behavior, constantly shifting her neck position and moving about the room. Her affect was tearful and her mood depressed. She was taking the NSAID celecoxib, 100 mg bid, and the skeletal muscle relaxant tizanidine, 4 mg tid. She was no longer using opioids and had no history of alcohol or illicit drug abuse.
Based on this assessment, the psychiatrist diagnosed Ms. A as having pain disorder with medical and psychological features, including symptom amplification and depression.
Table 1
4 treatment goals for patients with chronic pain and depression
|
Educating the patient
As part of your assessment, explain the reciprocal effects of depression and pain. Acknowledge that:
- chronic pain is different from acute pain, although the patient’s pain experience is the same
- treatment often becomes part of the problem in chronic pain.
Doctors tend to apply acute pain treatments chronically, risking long-term effects of polypharmacy to achieve short-term relief. Depressed patients may be more likely than nondepressed patients to receive opioids for chronic pain,12 and opioids and benzodiazepines may have depressive effects, as reflected by DSM-IV-TR’s inclusion of criteria for “opioid-induced mood disorder” and “sedative-, hypnotic-, or anxiolytic-induced mood disorder.”
To reduce patients’ resistance to antidepressants, reiterate any history of cumulative stressors and affective episodes unrelated to pain. Try using an analogy, such as “stress and pain are like waves on a rock” that eventually damage mood and coping mechanisms, or depression complicating pain is like having “too much on one’s plate.”
Finally, help patients understand that chronic pain is managed, not cured. Encourage them to set treatment goals beyond reducing pain (Table 1) and to make the transition from “patient with pain” to “client managing pain.”
Table 2
Dosing antidepressants and anticonvulsants
for chronic pain and depression
| Drug | Starting (mg/d) | Target (mg/d) | Administration tips |
|---|---|---|---|
| TCAs | Check serum levels for dosages ≥150 mg/d (nortriptyline 100 mg/d) to assess rapid metabolism, adherence, or toxic levels | ||
| Amitriptyline | 10 to 25 | 75 to 300 | |
| Clomipramine | 10 to 25 | 75 to 250 | |
| Desipramine | 10 to 25 | 75 to 200 | |
| Doxepin | 10 to 25 | 75 to 300 | |
| Imipramine | 10 to 25 | 75 to 300 | |
| Nortriptyline | 10 to 25 | 40 to 200 | |
| SNRI | |||
| Venlafaxine | 37.5 to 75 | 75 to 375 | Use XR form to minimize side effects and for once-daily dosing |
| SSRIs | |||
| Citalopram | 10 to 20 | 40 to 60 | |
| Fluoxetine | 10 to 20 | 20 to 80 | May increase carbamazepine, TCA blood levels and inhibit efficacy of codeine, dihydrocodeine, and hydrocodone |
| Paroxetine | 10 to 20 | 20 to 60 | Same as fluoxetine |
| Anticonvulsants | |||
| Carbamazepine | 200 | 800 to 1,200 | Check blood levels; may increase clomipramine levels, reduce acetaminophen, contraceptive levels |
| Clonazepam | 0.5 | 1 to 2 | Habituating potential with chronic use |
| Gabapentin | 300 to 900 | 3,600 to 4,800 | Blood monitoring not necessary |
| Valproate | 250 | 750 to 2,500 (maximum dosage 60 mg/kg/d) | Check blood levels (trough plasma level 50 to 100 μg/mL) |
| TCA: tricyclic antidepressant | |||
| SNRI: serotonin-norepinephrine reuptake inhibitor | |||
| SSRI: selective serotonin reuptake inhibitor | |||
Prescribing principles
Before adding any new pain medications, consider reducing dosages or discontinuing opioids or benzodiazepines and other substances the patient may be taking. Opioid use is associated with risks of dependence, addiction, and side effects including somnolence, cognitive impairment, and reduced activity that amplify depressive symptoms.
Benzodiazepines can generally be tapered by 10% per day, although you may need to extend the final taper over 3 to 4 days or longer, depending upon chronicity of use. Opioids may be tapered by 20% over 5 to 7 days. Breakthrough doses may be needed for marked withdrawal symptoms. Converting to longer half-life agents—such as clonazepam for benzodiazepines or methadone for opioids—often aids tapering, although other agents and strategies exist.13
To gauge patient attempts at self-medication, monitor use of alcohol or illicit drugs with urine screening. For patients with a substantial history of substance abuse or positive toxicology screens, monitor randomly every 2 to 4 weeks.
On the other hand, undertreated pain also may impair mood and function.1 If pain and mood improve and problematic drug-related behaviors resolve with increased opioid analgesia, consider maintaining opioids with regular re-evaluation of mood, coping, and medication adherence.11 Transfer from immediate-release to controlled-release opioids to reduce dosing frequency, clockwatching, and the likelihood of inter-dose pain escalation. In general, maintain and optimize the dosage of nonaddictive analgesics such as NSAIDs, anticonvulsants, or antidepressants.
Case continued: Switching medication
The psychiatrist started Ms. A on nortriptyline, 25 mg at bedtime, to be increased after 3 nights to 50 mg at bedtime. Tizanidine, which had been ineffective, was discontinued to reduce the risk of xerostomia and oversedation in combination with nortriptyline. If tolerated, nortriptyline was to be further increased by 25 mg every 3 days to an initial target dosage of 100 mg at bedtime. The psychiatrist explained to Ms. A that it might take 4 to 6 weeks to gauge the medication’s efficacy.
Psychoeducation addressed the importance of stress reduction, prioritizing commitments, and setting limits on other people’s expectations. The door was left open to future psychotherapeutic exploration of past cumulative stressors.
Because antidepressants may provide an analgesic effect,6,14 they are often used to treat affective symptoms in chronic pain. Headache and neuralgia tend to respond to antidepressants more robustly than do arthritis and low-back pain. Although some patients respond to low-dose antidepressants, a definitive trial requires full doses for 6 to 8 weeks (Table 2).
Matching a patient’s symptoms with medication side effects is useful when choosing antidepressants (Table 3). So-called “adverse” effects may have a corresponding benefit, depending on the clinical presentation. For example, a moreactivating antidepressant—such as the selective serotonin reuptake inhibitor (SSRI) fluoxetine—may help a patient with fatigue, whereas a moresedating agent—such as a tricyclic antidepressant (TCA) or mirtazapine—may improve sleep for a patient with insomnia.
Psychosocial therapies such as cognitive-behavioral therapy (CBT) or relaxation training (Table 4) may help patients with chronic pain to:
- process covert emotions such as fear and anger as well as guilt, loss, and disability
- reduce somatic preoccupation that is aggravating the pain
- adhere to treatment.
Evidence strongly supports using relaxation techniques to reduce chronic pain in many medical conditions and hypnosis to ameliorate cancer pain. CBT and biofeedback appear moderately effective in relieving chronic pain.15 CBT is significantly more effective than waiting list control conditions for relieving chronic nonheadache pain in measures of pain experience, mood/affect, cognitive coping and appraisal, pain behavior and activity level, and social role functioning.16
Pain and opioid medications can impair concentration and affective processing, so initial psychotherapy may need to be supportive while you provide other treatments and simplify medication regimens. Eventually the patient may be ready to address underlying issues that may be contributing to the pain syndrome, such as a history of abuse. However, it is important to address this potentially destabilizing subject only after carefully gauging a patient’s defenses and readiness.
Case continued: A bump in the road
The psychiatrist saw Ms. A 18 months later. Interim history revealed that her pain and mood improved on nortriptyline, 100 mg at bedtime. When she stopped taking nortriptyline 5 months earlier, her neck pain increased and she experienced a “deep blue mood.” Her physician restarted the nortriptyline.
At follow up, Ms. A reported no depressive symptoms and very little neck pain. The psychiatrist discussed with her depression’s relapse rate and the importance of continuing antidepressant therapy. As Ms. A was feeling much better and functioning normally, the psychiatrist decided additional psychotherapeutic intervention was not necessary.
Antidepressant options
TCAs provide analgesia via descending regulatory pathways by inhibiting serotonin and norepinephrine reuptake.17 When using TCAs for chronic pain, start with 10 to 25 mg at bedtime and increase by 10 to 25 mg every 3 to 7 days as tolerated. Increase incrementally until the pain responds or to the full antidepressant dosage (Table 2). Drug levels (when available) can help you provide an appropriate trial and monitor the patient’s adherence.
If the pain does not respond after 6 to 8 weeks, consider switching to another dual-action agent such as venlafaxine or to an SSRI.
SNRIs. Venlafaxine is a serotonin and norepineph-rine reuptake inhibitor (SNRI) with less-troublesome side effects than TCAs. It is structurally similar to tramadol18 and has combined serotonin and norepinephrine inhibition at dosages >75 mg/d. Although venlafaxine is not indicated for chronic pain, some studies have suggested possible benefits, including long-term analgesia, reduced polyneuropathic pain, and migraine prophylaxis.19-21 Venlafaxine may be a reasonable first or second choice for treating depression in patients with chronic pain, especially headache.14
Duloxetine—another SNRI—awaits FDA approval. Some studies have suggested that duloxetine improves painful physical symptoms as well as mood and functioning in major depression.22
SSRIs may be effective for certain types of pain, but the evidence is conflicting. Results of 41controlled trials support TCAs’ analgesic efficacy for neuropathic pain, headache, and central and post-stroke pain, whereas SSRIs’ analgesic efficacy varies from study to study. Comparisons of TCAs and SSRIs as analgesics uniformly show TCAs to be more effective, with the SSRIs often showing no analgesic effect.
Of three controlled trials of SSRIs for diabetic neuropathy, one showed fluoxetine similar to placebo, and two smaller studies showed paroxetine and citalopram more effective than placebo. Fluoxetine has shown analgesic effect for fibromyalgia in one study, but no effect in another. Citalopram showed no analgesic effect for fibromyalgia in another study.23
A prospective, double-blind study comparing fluoxetine, sertraline, paroxetine, and venlafaxine for migraines reported moderate to significant improvement in less than one-half of SSRI-treated patients vs two-thirds of venlafaxine-treated patients.21 SSRIs are no longer recognized by the International Headache Society as primary preventative medications for migraine.
Fluoxetine may help chronic daily headache, and paroxetine and citalopram may be useful for diabetic neuropathy. However, one cannot generalize that all SSRIs are similarly effective as analgesics.14
SSRIs have fewer side effects than TCAs and are less dangerous in overdose. In general, however, SSRIs are a second-line treatment for pain, to be used when dual-action agents pose disadvantageous side effects (Table 3) or have been poorly tolerated or ineffective.
Table 3
Antidepressant side effects:
Limitations and potential benefits in chronic pain
| Side effects/agents | Problems | Conditions potentially benefited | Possible alternatives |
|---|---|---|---|
| Anticholinergic TCAs | Xerostomia, constipation, urinary slowing (esp. when combined with opioids) | Diarrhea-predominant irritable bowel syndrome | SSRIs, nefazodone, venlafaxine |
| Sedation TCAs, mirtazapine, nefazodone, trazodone | Excessive sedation, cognitive impairment, driving risk (esp. when combined with opioids, benzodiazepines) | Pain with insomnia | SSRIs, venlafaxine, bupropion |
| Insomnia SSRIs, venlafaxine | Pain with pre-existing insomnia; equivocal analgesic effects | Excess sedation related to depression, polypharmacy for pain | TCAs, mirtazapine, trazodone, nefazodone |
| Orthostasis TCAs (esp. with methadone), nefazodone | Falls, especially in elderly patients | —— | Nortriptyline, SSRIs bupropion, venlafaxine |
| Weight gain TCAs, mirtazapine | Pain patients are often sedentary, get limited exercise | Pain and depression with weight loss | Bupropion, fluoxetine |
| Hypertension Bupropion, venlafaxine | Pre-existing hypertension | ? Hypotensive state | Citalopram (hypertensive side effects infrequent) |
| Cardiac TCAs | ECG abnormalities, conduction delays, arrhythmias aggravate pre-existing cardiac abnormalities; avoid if recent MI | ——— | SSRIs, bupropion |
| Overdose lethality TCAs | Prominent suicidal ideation | —— | SSRIs, venlafaxine |
| Seizures Esp. maprotiline, clomipramine, bupropion | Lower seizure threshold, aggravation of seizure disorders | ——- | SSRIs |
| Sexual dysfunction SSRIs | Pre-existing sexual dysfunction secondary to pain, medications, stress; equivocal analgesic effects | ——- | Bupropion, nefazodone, mirtazapine |
Table 4
How psychosocial therapies can help treat chronic pain and depression
| Therapies | Purpose/benefits |
|---|---|
| Behavioral therapy | Increase activity and learn to balance activity with limitations Reduce pain behaviors and analgesic use Decrease dependency and secondary gain |
| Cognitive-behavioral therapy | Identify automatic thoughts Challenge negative cognitions, catastrophizing Substitute and rehearse positive thoughts, capabilities Transition from patient role to self-care |
| Couples’ therapy | Assist adaptation to role changes Diminish spousal solicitousness or excessive caretaking Increase communication |
| Biofeedback, relaxation, imagery | Adjunctive role in pain management Reduce tension, comorbid anxiety |
| Hypnosis | Dissociate awareness of pain Substitute, displace, reinterpret pain sensations |
| Vocational rehabilitation | Increase activity, ability to distract Regain sense of control, identity, and productivity Increase socialization |
| Pain management program | Multiple treatment effects Useful for complex pain with affective states |
Monoamine oxidase inhibitors (MAOIs) may have some efficacy for neuropathy and headache, but the need for a tyramine-free diet and potential for drug-drug interactions limit their usefulness. Co-administering an MAOI and meperidine is always contraindicated, as this combination can produce fever, delirium, seizures, circulatory collapse, and death. Similarly, avoid using an MAOI with any other antidepressant.
Others. Evidence is very limited on using other antidepressants such as trazodone, nefazodone, bupropion, and mirtazapine in chronic pain:
- Trazodone may help pediatric migraine, but it is not a consistent analgesic and may not be well-tolerated.
- Case reports suggest bupropion may help with headaches and chronic low-back pain.14
- Mirtazapine and trazodone may be useful adjuncts for treating insomnia in depressed patients with chronic pain.
Other options
Anticonvulsants appear useful for neuropathic pain and are appropriate for chronic pain patients who cannot tolerate TCAs.24 Like TCAs, anticonvulsants are not addictive. Unlike TCAs, anticonvulsants may help stabilize other affective illnesses that may coexist with chronic pain, including bipolar disorder, schizoaffective disorder, and impulsivity/aggression related to dementia or personality disorder.6 If the starting dosage is not effective within 1 week, increase gradually every 2 to 3 days to target dosages comparable to those for anticonvulsant efficacy.
Carbamazepine and gabapentin are recommended first-line medications for neuropathy. Carbamazepine is indicated for treating trigeminal neuralgia, although its cytochrome P-450 3A3/4 isoenzyme induction may reduce serum levels of acetaminophen, opioids, and oral contraceptives. Gabapentin, although not clearly beneficial for bipolar disorder, has anxiolytic effects and a benign side-effect profile, which may help patients with chronic pain.
Valproate can help prevent migraines. Clonazepam can help reduce anxiety and restless legs syndrome but may be habituating. Anticonvulsants’ common adverse effects include sedation, GI upset, dizziness, and fatigue.
Lithium has known efficacy for mood stabilization in bipolar disorder and can ameliorate cluster headaches.
Antipsychotics. Evidence is sparse on whether antipsychotics have analgesic activity. Their side effects generally limit their usefulness to treating pain in patients with psychosis or delirium.6
Stimulants such as dextroamphetamine and methylphenidate can be helpful adjuncts for treating depression, especially for medical inpatients who require a rapid therapeutic response. Stimulants may reduce fatigue or excessive sedation and improve concentration in patients receiving opioids for chronic pain. They also may have analgesic effects when combined with opioids. Potential adverse effects include appetite suppression, anxiety or agitation, confusion, tics, and addiction.6
Precautions. The muscle relaxant carisoprodol is associated with potential dependence and withdrawal. Cyclobenzaprine, another muscle relaxant, has a TCA-like structure and can be lethal in overdose. Baclofen can be useful for chronic pain related to spasticity, although psychotic depression and mania have been reported with abrupt withdrawal.6
Related resources
- American Academy of Pain Medicine. www.painmed.org/
- American Academy of Pain Management. www.aapainmanage.org
- Pain.com: continuing medical education for anesthesiology professionals. www.pain.com/index.cfm
- Kerns RD, Turk DC, Rudy TE. The West Haven-Yale Multidimensional Pain Inventory (WHYMPI). Pain 1985;13:345-56
Drug brand names
- Amitriptyline • Elavil
- Baclofen • Lioresal
- Bupropion • Wellbutrin
- Carbamazepine • Tegretol
- Carisoprodol • Soma
- Celecoxib • Celebrex
- Citalopram • Celexa
- Clomipramine • Anafranil
- Clonazepam • Klonopin
- Cyclobenzaprine • Flexeril
- Desipramine • Norpramin
- Dihydrocodeine • Synalgos-DC
- Doxepin • Sinequan
- Duloxetine • Cymbalta
- Fluoxetine • Prozac
- Gabapentin • Neurontin
- Hydrocodone • Vicodin, Lortab
- Imipramine • Tofranil
- Lithium • Eskalith CR, Lithobid
- Maprotiline • Ludiomil
- Meperidine • Demerol
- Methadone • Dolophine
- Mirtazapine • Remeron
- Nefazodone • Serzone
- Nortriptyline • Pamelor
- Oxycodone • OxyContin
- Paroxetine • Paxil
- Sertraline • Zoloft
- Tizanidine • Zanaflex
- Tramadol • Ultram
- Trazodone • Desyrel
- Valproate • Depakote
- Venlafaxine • Effexor, Effexor XR
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Ms. A, age 44, fell from a 3-foot stool while reaching for a high kitchen shelf and suffered severe neck flexion. Her initial pain persisted for weeks and then months, resulting in chronic neck pain aggravated by movement.
Over the past year, her doctor has prescribed numerous analgesics and muscle relaxants, including tramadol, hydrocodone, oxycodone, tizanidine, and nonsteroidal anti-inflammatory drugs (NSAIDs). Treatments at a pain clinic have included triggerpoint injections, cervical epidural corticosteroid injection, left-sided cervical medial branch blocks, transcutaneous electrical nerve stimulation, and physical therapy. None provided sustained relief.
During a pain clinic visit, Ms. A wept and said she was tired of living with pain. She acknowledged depression and agreed to psychiatric consultation.
As in Ms. A’s case, physicians often refer patients with chronic pain and affective symptoms for psychiatric evaluation. These patients are often fearful, angry, and suspicious of any suggestion that their physical discomfort has a psychiatric component. They typically believe their pain had a clear onset and therefore should have an end point. Many have experienced unproductive specialty evaluations and failed treatments.
To help you overcome these obstacles when treating patients with chronic pain and depression, we discuss:
- strategies to gain patients’ trust and build a therapeutic alliance
- how to assess their pain, depression, and suicide risk
- the role of psychotherapy in treating chronic pain
- and evidence for choosing effective, nonaddicting medications.
Psychiatric evaluation
Depression and pain are linked psychologically and biochemically, sharing neurotransmitters involved in both nociceptive pathways and mood, especially serotonin and norepinephrine.1,2 One-third to one-half of patients with chronic pain report comorbid depression,3 and more than one-half of depressed patients presenting to primary care physicians report only somatic symptoms—various pain complaints among the most common.4,5
Primary care doctors tend to refer chronic pain and depression cases to psychiatrists when:
- patients are preoccupied with medication, have not followed treatment recommendations, or do not respond to treatment as expected
- extensive medical evaluations reveal few or equivocal findings
- somatic complaints are vague and diffuse, or there is marked disparity between pain complaints/disability and objective findings.6,7
Assessing pain. In the initial assessment, validate the patient’s pain experience by asking about the location, quality, and severity of pain. The visual analogue scale (VAS) is commonly used to measure pain severity. The patient marks a spot on a line from “no pain” to “worst possible pain,” or—on a numbered VAS—from 0 (no pain) to 10 (extreme pain). The least and most severe pain over the preceding month can be ranked as baseline values.8
Be sensitive to the patient’s fear that you will attribute the pain to psychosocial issues or imply that “the pain is in your head.” Emphasize that you intend to evaluate the “whole person,” not just the part that hurts. Focus on how the pain affects the patient’s lifestyle—rather than its cause—and explore medication use patterns.
Assessing depression. The word “depression” is emotionally charged for chronic pain patients, who view affective symptoms—if they acknowledge them at all—as secondary to pain. They may strongly resist treatment for anything but pain. One way to defuse this defensiveness is to avoid attributing the pain to stress or depression.
Begin by assessing vegetative symptoms, which overlap in chronic pain and depression. The Beck Depression Inventory-II (Beck-II) may be a useful screening tool in a busy practice; the short form (13 questions) takes about 5 minutes to complete.9
Explore cognitive and behavioral symptoms such as concentration, pleasure and interest level, activity, and self-esteem. Review the chronology of pain onset, mood changes, and stressors (proximate, remote, and cumulative).
Seek clues to endogenous factors by asking about past affective episodes, response to antidepressants, and family history of psychopathology. Substances that may induce depression include reserpine, interferon, and antiparkinsonian agents. Screen for potential organic mood disorders, such as depression secondary to hypothyroidism, corticosteroid use, Parkinson’s disease, lupus, HIV infection, or cerebrovascular disease. Where appropriate, obtain collateral information from family or friends.
Assessing suicide risk. Chronic pain patients may be at greater risk of suicide than the general population. Besides pain, other risk factors for suicide—such as major depression, anxiety disorders, alcohol/substance abuse, sleep disturbances, male gender, diminished social support, and recent loss—are common among these patients.10,11
Screen chronic pain patients with suicidal ideation for these risk factors. Interventions include:
- aggressively treat associated depression, anxiety, or insomnia
- elicit support from family or other caregivers
- pay close attention to talk about suicide
- hospitalize when necessary
- and, of course, treat pain.
Case continued: No stranger to depression
Ms. A’s psychiatric assessments revealed a pain severity ranking of 9 on a 1-to-10 scale, frequent crying, hopelessness, disrupted sleep, low energy, limited ability to concentrate, and fleeting suicidal thoughts. Her history included counseling during her first marriage and severe depression after separation from her second husband 3 years ago. An 8-week trial of fluoxetine, 20 mg/d, did not improve her depression then.
On examination, she displayed obvious pain behavior, constantly shifting her neck position and moving about the room. Her affect was tearful and her mood depressed. She was taking the NSAID celecoxib, 100 mg bid, and the skeletal muscle relaxant tizanidine, 4 mg tid. She was no longer using opioids and had no history of alcohol or illicit drug abuse.
Based on this assessment, the psychiatrist diagnosed Ms. A as having pain disorder with medical and psychological features, including symptom amplification and depression.
Table 1
4 treatment goals for patients with chronic pain and depression
|
Educating the patient
As part of your assessment, explain the reciprocal effects of depression and pain. Acknowledge that:
- chronic pain is different from acute pain, although the patient’s pain experience is the same
- treatment often becomes part of the problem in chronic pain.
Doctors tend to apply acute pain treatments chronically, risking long-term effects of polypharmacy to achieve short-term relief. Depressed patients may be more likely than nondepressed patients to receive opioids for chronic pain,12 and opioids and benzodiazepines may have depressive effects, as reflected by DSM-IV-TR’s inclusion of criteria for “opioid-induced mood disorder” and “sedative-, hypnotic-, or anxiolytic-induced mood disorder.”
To reduce patients’ resistance to antidepressants, reiterate any history of cumulative stressors and affective episodes unrelated to pain. Try using an analogy, such as “stress and pain are like waves on a rock” that eventually damage mood and coping mechanisms, or depression complicating pain is like having “too much on one’s plate.”
Finally, help patients understand that chronic pain is managed, not cured. Encourage them to set treatment goals beyond reducing pain (Table 1) and to make the transition from “patient with pain” to “client managing pain.”
Table 2
Dosing antidepressants and anticonvulsants
for chronic pain and depression
| Drug | Starting (mg/d) | Target (mg/d) | Administration tips |
|---|---|---|---|
| TCAs | Check serum levels for dosages ≥150 mg/d (nortriptyline 100 mg/d) to assess rapid metabolism, adherence, or toxic levels | ||
| Amitriptyline | 10 to 25 | 75 to 300 | |
| Clomipramine | 10 to 25 | 75 to 250 | |
| Desipramine | 10 to 25 | 75 to 200 | |
| Doxepin | 10 to 25 | 75 to 300 | |
| Imipramine | 10 to 25 | 75 to 300 | |
| Nortriptyline | 10 to 25 | 40 to 200 | |
| SNRI | |||
| Venlafaxine | 37.5 to 75 | 75 to 375 | Use XR form to minimize side effects and for once-daily dosing |
| SSRIs | |||
| Citalopram | 10 to 20 | 40 to 60 | |
| Fluoxetine | 10 to 20 | 20 to 80 | May increase carbamazepine, TCA blood levels and inhibit efficacy of codeine, dihydrocodeine, and hydrocodone |
| Paroxetine | 10 to 20 | 20 to 60 | Same as fluoxetine |
| Anticonvulsants | |||
| Carbamazepine | 200 | 800 to 1,200 | Check blood levels; may increase clomipramine levels, reduce acetaminophen, contraceptive levels |
| Clonazepam | 0.5 | 1 to 2 | Habituating potential with chronic use |
| Gabapentin | 300 to 900 | 3,600 to 4,800 | Blood monitoring not necessary |
| Valproate | 250 | 750 to 2,500 (maximum dosage 60 mg/kg/d) | Check blood levels (trough plasma level 50 to 100 μg/mL) |
| TCA: tricyclic antidepressant | |||
| SNRI: serotonin-norepinephrine reuptake inhibitor | |||
| SSRI: selective serotonin reuptake inhibitor | |||
Prescribing principles
Before adding any new pain medications, consider reducing dosages or discontinuing opioids or benzodiazepines and other substances the patient may be taking. Opioid use is associated with risks of dependence, addiction, and side effects including somnolence, cognitive impairment, and reduced activity that amplify depressive symptoms.
Benzodiazepines can generally be tapered by 10% per day, although you may need to extend the final taper over 3 to 4 days or longer, depending upon chronicity of use. Opioids may be tapered by 20% over 5 to 7 days. Breakthrough doses may be needed for marked withdrawal symptoms. Converting to longer half-life agents—such as clonazepam for benzodiazepines or methadone for opioids—often aids tapering, although other agents and strategies exist.13
To gauge patient attempts at self-medication, monitor use of alcohol or illicit drugs with urine screening. For patients with a substantial history of substance abuse or positive toxicology screens, monitor randomly every 2 to 4 weeks.
On the other hand, undertreated pain also may impair mood and function.1 If pain and mood improve and problematic drug-related behaviors resolve with increased opioid analgesia, consider maintaining opioids with regular re-evaluation of mood, coping, and medication adherence.11 Transfer from immediate-release to controlled-release opioids to reduce dosing frequency, clockwatching, and the likelihood of inter-dose pain escalation. In general, maintain and optimize the dosage of nonaddictive analgesics such as NSAIDs, anticonvulsants, or antidepressants.
Case continued: Switching medication
The psychiatrist started Ms. A on nortriptyline, 25 mg at bedtime, to be increased after 3 nights to 50 mg at bedtime. Tizanidine, which had been ineffective, was discontinued to reduce the risk of xerostomia and oversedation in combination with nortriptyline. If tolerated, nortriptyline was to be further increased by 25 mg every 3 days to an initial target dosage of 100 mg at bedtime. The psychiatrist explained to Ms. A that it might take 4 to 6 weeks to gauge the medication’s efficacy.
Psychoeducation addressed the importance of stress reduction, prioritizing commitments, and setting limits on other people’s expectations. The door was left open to future psychotherapeutic exploration of past cumulative stressors.
Because antidepressants may provide an analgesic effect,6,14 they are often used to treat affective symptoms in chronic pain. Headache and neuralgia tend to respond to antidepressants more robustly than do arthritis and low-back pain. Although some patients respond to low-dose antidepressants, a definitive trial requires full doses for 6 to 8 weeks (Table 2).
Matching a patient’s symptoms with medication side effects is useful when choosing antidepressants (Table 3). So-called “adverse” effects may have a corresponding benefit, depending on the clinical presentation. For example, a moreactivating antidepressant—such as the selective serotonin reuptake inhibitor (SSRI) fluoxetine—may help a patient with fatigue, whereas a moresedating agent—such as a tricyclic antidepressant (TCA) or mirtazapine—may improve sleep for a patient with insomnia.
Psychosocial therapies such as cognitive-behavioral therapy (CBT) or relaxation training (Table 4) may help patients with chronic pain to:
- process covert emotions such as fear and anger as well as guilt, loss, and disability
- reduce somatic preoccupation that is aggravating the pain
- adhere to treatment.
Evidence strongly supports using relaxation techniques to reduce chronic pain in many medical conditions and hypnosis to ameliorate cancer pain. CBT and biofeedback appear moderately effective in relieving chronic pain.15 CBT is significantly more effective than waiting list control conditions for relieving chronic nonheadache pain in measures of pain experience, mood/affect, cognitive coping and appraisal, pain behavior and activity level, and social role functioning.16
Pain and opioid medications can impair concentration and affective processing, so initial psychotherapy may need to be supportive while you provide other treatments and simplify medication regimens. Eventually the patient may be ready to address underlying issues that may be contributing to the pain syndrome, such as a history of abuse. However, it is important to address this potentially destabilizing subject only after carefully gauging a patient’s defenses and readiness.
Case continued: A bump in the road
The psychiatrist saw Ms. A 18 months later. Interim history revealed that her pain and mood improved on nortriptyline, 100 mg at bedtime. When she stopped taking nortriptyline 5 months earlier, her neck pain increased and she experienced a “deep blue mood.” Her physician restarted the nortriptyline.
At follow up, Ms. A reported no depressive symptoms and very little neck pain. The psychiatrist discussed with her depression’s relapse rate and the importance of continuing antidepressant therapy. As Ms. A was feeling much better and functioning normally, the psychiatrist decided additional psychotherapeutic intervention was not necessary.
Antidepressant options
TCAs provide analgesia via descending regulatory pathways by inhibiting serotonin and norepinephrine reuptake.17 When using TCAs for chronic pain, start with 10 to 25 mg at bedtime and increase by 10 to 25 mg every 3 to 7 days as tolerated. Increase incrementally until the pain responds or to the full antidepressant dosage (Table 2). Drug levels (when available) can help you provide an appropriate trial and monitor the patient’s adherence.
If the pain does not respond after 6 to 8 weeks, consider switching to another dual-action agent such as venlafaxine or to an SSRI.
SNRIs. Venlafaxine is a serotonin and norepineph-rine reuptake inhibitor (SNRI) with less-troublesome side effects than TCAs. It is structurally similar to tramadol18 and has combined serotonin and norepinephrine inhibition at dosages >75 mg/d. Although venlafaxine is not indicated for chronic pain, some studies have suggested possible benefits, including long-term analgesia, reduced polyneuropathic pain, and migraine prophylaxis.19-21 Venlafaxine may be a reasonable first or second choice for treating depression in patients with chronic pain, especially headache.14
Duloxetine—another SNRI—awaits FDA approval. Some studies have suggested that duloxetine improves painful physical symptoms as well as mood and functioning in major depression.22
SSRIs may be effective for certain types of pain, but the evidence is conflicting. Results of 41controlled trials support TCAs’ analgesic efficacy for neuropathic pain, headache, and central and post-stroke pain, whereas SSRIs’ analgesic efficacy varies from study to study. Comparisons of TCAs and SSRIs as analgesics uniformly show TCAs to be more effective, with the SSRIs often showing no analgesic effect.
Of three controlled trials of SSRIs for diabetic neuropathy, one showed fluoxetine similar to placebo, and two smaller studies showed paroxetine and citalopram more effective than placebo. Fluoxetine has shown analgesic effect for fibromyalgia in one study, but no effect in another. Citalopram showed no analgesic effect for fibromyalgia in another study.23
A prospective, double-blind study comparing fluoxetine, sertraline, paroxetine, and venlafaxine for migraines reported moderate to significant improvement in less than one-half of SSRI-treated patients vs two-thirds of venlafaxine-treated patients.21 SSRIs are no longer recognized by the International Headache Society as primary preventative medications for migraine.
Fluoxetine may help chronic daily headache, and paroxetine and citalopram may be useful for diabetic neuropathy. However, one cannot generalize that all SSRIs are similarly effective as analgesics.14
SSRIs have fewer side effects than TCAs and are less dangerous in overdose. In general, however, SSRIs are a second-line treatment for pain, to be used when dual-action agents pose disadvantageous side effects (Table 3) or have been poorly tolerated or ineffective.
Table 3
Antidepressant side effects:
Limitations and potential benefits in chronic pain
| Side effects/agents | Problems | Conditions potentially benefited | Possible alternatives |
|---|---|---|---|
| Anticholinergic TCAs | Xerostomia, constipation, urinary slowing (esp. when combined with opioids) | Diarrhea-predominant irritable bowel syndrome | SSRIs, nefazodone, venlafaxine |
| Sedation TCAs, mirtazapine, nefazodone, trazodone | Excessive sedation, cognitive impairment, driving risk (esp. when combined with opioids, benzodiazepines) | Pain with insomnia | SSRIs, venlafaxine, bupropion |
| Insomnia SSRIs, venlafaxine | Pain with pre-existing insomnia; equivocal analgesic effects | Excess sedation related to depression, polypharmacy for pain | TCAs, mirtazapine, trazodone, nefazodone |
| Orthostasis TCAs (esp. with methadone), nefazodone | Falls, especially in elderly patients | —— | Nortriptyline, SSRIs bupropion, venlafaxine |
| Weight gain TCAs, mirtazapine | Pain patients are often sedentary, get limited exercise | Pain and depression with weight loss | Bupropion, fluoxetine |
| Hypertension Bupropion, venlafaxine | Pre-existing hypertension | ? Hypotensive state | Citalopram (hypertensive side effects infrequent) |
| Cardiac TCAs | ECG abnormalities, conduction delays, arrhythmias aggravate pre-existing cardiac abnormalities; avoid if recent MI | ——— | SSRIs, bupropion |
| Overdose lethality TCAs | Prominent suicidal ideation | —— | SSRIs, venlafaxine |
| Seizures Esp. maprotiline, clomipramine, bupropion | Lower seizure threshold, aggravation of seizure disorders | ——- | SSRIs |
| Sexual dysfunction SSRIs | Pre-existing sexual dysfunction secondary to pain, medications, stress; equivocal analgesic effects | ——- | Bupropion, nefazodone, mirtazapine |
Table 4
How psychosocial therapies can help treat chronic pain and depression
| Therapies | Purpose/benefits |
|---|---|
| Behavioral therapy | Increase activity and learn to balance activity with limitations Reduce pain behaviors and analgesic use Decrease dependency and secondary gain |
| Cognitive-behavioral therapy | Identify automatic thoughts Challenge negative cognitions, catastrophizing Substitute and rehearse positive thoughts, capabilities Transition from patient role to self-care |
| Couples’ therapy | Assist adaptation to role changes Diminish spousal solicitousness or excessive caretaking Increase communication |
| Biofeedback, relaxation, imagery | Adjunctive role in pain management Reduce tension, comorbid anxiety |
| Hypnosis | Dissociate awareness of pain Substitute, displace, reinterpret pain sensations |
| Vocational rehabilitation | Increase activity, ability to distract Regain sense of control, identity, and productivity Increase socialization |
| Pain management program | Multiple treatment effects Useful for complex pain with affective states |
Monoamine oxidase inhibitors (MAOIs) may have some efficacy for neuropathy and headache, but the need for a tyramine-free diet and potential for drug-drug interactions limit their usefulness. Co-administering an MAOI and meperidine is always contraindicated, as this combination can produce fever, delirium, seizures, circulatory collapse, and death. Similarly, avoid using an MAOI with any other antidepressant.
Others. Evidence is very limited on using other antidepressants such as trazodone, nefazodone, bupropion, and mirtazapine in chronic pain:
- Trazodone may help pediatric migraine, but it is not a consistent analgesic and may not be well-tolerated.
- Case reports suggest bupropion may help with headaches and chronic low-back pain.14
- Mirtazapine and trazodone may be useful adjuncts for treating insomnia in depressed patients with chronic pain.
Other options
Anticonvulsants appear useful for neuropathic pain and are appropriate for chronic pain patients who cannot tolerate TCAs.24 Like TCAs, anticonvulsants are not addictive. Unlike TCAs, anticonvulsants may help stabilize other affective illnesses that may coexist with chronic pain, including bipolar disorder, schizoaffective disorder, and impulsivity/aggression related to dementia or personality disorder.6 If the starting dosage is not effective within 1 week, increase gradually every 2 to 3 days to target dosages comparable to those for anticonvulsant efficacy.
Carbamazepine and gabapentin are recommended first-line medications for neuropathy. Carbamazepine is indicated for treating trigeminal neuralgia, although its cytochrome P-450 3A3/4 isoenzyme induction may reduce serum levels of acetaminophen, opioids, and oral contraceptives. Gabapentin, although not clearly beneficial for bipolar disorder, has anxiolytic effects and a benign side-effect profile, which may help patients with chronic pain.
Valproate can help prevent migraines. Clonazepam can help reduce anxiety and restless legs syndrome but may be habituating. Anticonvulsants’ common adverse effects include sedation, GI upset, dizziness, and fatigue.
Lithium has known efficacy for mood stabilization in bipolar disorder and can ameliorate cluster headaches.
Antipsychotics. Evidence is sparse on whether antipsychotics have analgesic activity. Their side effects generally limit their usefulness to treating pain in patients with psychosis or delirium.6
Stimulants such as dextroamphetamine and methylphenidate can be helpful adjuncts for treating depression, especially for medical inpatients who require a rapid therapeutic response. Stimulants may reduce fatigue or excessive sedation and improve concentration in patients receiving opioids for chronic pain. They also may have analgesic effects when combined with opioids. Potential adverse effects include appetite suppression, anxiety or agitation, confusion, tics, and addiction.6
Precautions. The muscle relaxant carisoprodol is associated with potential dependence and withdrawal. Cyclobenzaprine, another muscle relaxant, has a TCA-like structure and can be lethal in overdose. Baclofen can be useful for chronic pain related to spasticity, although psychotic depression and mania have been reported with abrupt withdrawal.6
Related resources
- American Academy of Pain Medicine. www.painmed.org/
- American Academy of Pain Management. www.aapainmanage.org
- Pain.com: continuing medical education for anesthesiology professionals. www.pain.com/index.cfm
- Kerns RD, Turk DC, Rudy TE. The West Haven-Yale Multidimensional Pain Inventory (WHYMPI). Pain 1985;13:345-56
Drug brand names
- Amitriptyline • Elavil
- Baclofen • Lioresal
- Bupropion • Wellbutrin
- Carbamazepine • Tegretol
- Carisoprodol • Soma
- Celecoxib • Celebrex
- Citalopram • Celexa
- Clomipramine • Anafranil
- Clonazepam • Klonopin
- Cyclobenzaprine • Flexeril
- Desipramine • Norpramin
- Dihydrocodeine • Synalgos-DC
- Doxepin • Sinequan
- Duloxetine • Cymbalta
- Fluoxetine • Prozac
- Gabapentin • Neurontin
- Hydrocodone • Vicodin, Lortab
- Imipramine • Tofranil
- Lithium • Eskalith CR, Lithobid
- Maprotiline • Ludiomil
- Meperidine • Demerol
- Methadone • Dolophine
- Mirtazapine • Remeron
- Nefazodone • Serzone
- Nortriptyline • Pamelor
- Oxycodone • OxyContin
- Paroxetine • Paxil
- Sertraline • Zoloft
- Tizanidine • Zanaflex
- Tramadol • Ultram
- Trazodone • Desyrel
- Valproate • Depakote
- Venlafaxine • Effexor, Effexor XR
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Campbell LC, Clauw DJ, Keefe FJ. Persistent pain and depression: a biopsychosocial perspective. Biol Psychiatry 2003;54:399-409.
2. Fishbain DA, Cutler R, Rosomoff HL, et al. Chronic painassociated depression: antecedent or consequence of chronic pain? A review. Clin J Pain 1997;13(2):116-37.
3. Banks SM, Kerns RD. Explaining high rates of depression in chronic pain: a diathesis-stress framework. Psychol Bull 1996;119:95-110.
4. Simon GE, VonKorff M, Piccinelli M, et al. An international study of the relation between somatic symptoms and depression. N Engl J Med 1999;341(18):1329-35.
5. Kroenke K, Price RK. Symptoms in the community. Prevalence, classification, and psychiatric comorbidity. Arch Intern Med. 1993;153:2474-80.
6. Leo RJ. Concise guide to pain management for psychiatrists. Arlington, Va: American Psychiatric Publishing, Inc., 2003.
7. Sullivan MD, Turner JA, Romano J. Chronic pain in primary care. Identification and management of psychosocial factors. J Fam Pract 1991;32(2):193-9.
8. Holmgren A, Wise MG, Bouckoms AJ. Pain management. In: Wise MG, Rundell JR (eds). Psychiatry in the medically ill (2nd ed). Washington, DC: American Psychiatric Publishing Inc., 2002;989-1013.
9. Naifeh KH. Psychometric testing in functional GI disorders in: Olden K (ed). Handbook of functional GI disorders. New York: Marcel Dekker, 1996;79-126.
10. Fishbain DA. The association of chronic pain and suicide. Semin Clin Neuropsychiatry 1999;4(3):221-7.
11. Fishbain DA. Medico-legal rounds: medical-legal issues and breaches of “standards of medical care” in opioid tapering for alleged opioid addiction. Pain Med 2002;3(2):135-42.
12. Doan BD, Wadden NP. Relationships between depressive symptoms and descriptions of chronic pain. Pain 1989;36:75-84.
13. Franklin JE, Leamon MH, Frances RJ. Substance-related disorders. In: Wise MG, Rundell JR (eds). Psychiatry in the medically ill (2nd ed). Washington DC: American Psychiatric Publishing, 2002;417-53.
14. Ansari A. The efficacy of newer antidepressants in the treatment of chronic pain: a review of current literature. Harv Rev Psychiatry 2000;7(5):257-77.
15. NIH Technology Assessment Panel. Integration of behavioral relaxation approaches into the treatment of chronic pain and insomnia. JAMA 1996;276(4):313-18.
16. Morley S, Eccleston C, Williams A. Systematic review and metaanalysis of randomized controlled trials of cognitive behaviour therapy and behaviour therapy for chronic pain in adults, excluding headache. Pain 1999;80:1-13
17. Magni G. On the relationship between chronic pain and depression when there is no organic lesion. Pain 1987;31:1-21.
18. Markowitz JS, Patrick KS. Venlafaxine-tramadol similarities. Med Hypotheses 1998;5:167-8
19. Bradley RH, Barkin RL, Jerome J, et al. Efficacy of venlafaxine for the long-term treatment of chronic pain with associated major depressive disorder. Am J Ther 2003;10(5):318-23.
20. Sindrup SH, Bach FW, Madsen C, et al. Venlafaxine vs. imipramine in painful polyneuropathy—a randomized controlled trial. Neurology 2003;60:1284-9.
21. Kathpal GS. Role of SSRIs in the management of migraine. Headache Quarterly 1998;9:265-6.
22. Mallinckrodt CH, Goldstein DJ, Detke MJ, et al. Duloxetine: a new treatment for the emotional and physical symptoms of depression. Primary Care Companion J Clin Psychiatry 2003;5(1):19-28.
23. Lynch ME. Antidepressants as analgesics: a review of randomized control trials. J Psychiatry Neurosci 2001;26-36.
24. Swerdlow M. Anticonvulsant drugs and chronic pain. Clin Neuropharmacol 1984;7(1):51-82.
1. Campbell LC, Clauw DJ, Keefe FJ. Persistent pain and depression: a biopsychosocial perspective. Biol Psychiatry 2003;54:399-409.
2. Fishbain DA, Cutler R, Rosomoff HL, et al. Chronic painassociated depression: antecedent or consequence of chronic pain? A review. Clin J Pain 1997;13(2):116-37.
3. Banks SM, Kerns RD. Explaining high rates of depression in chronic pain: a diathesis-stress framework. Psychol Bull 1996;119:95-110.
4. Simon GE, VonKorff M, Piccinelli M, et al. An international study of the relation between somatic symptoms and depression. N Engl J Med 1999;341(18):1329-35.
5. Kroenke K, Price RK. Symptoms in the community. Prevalence, classification, and psychiatric comorbidity. Arch Intern Med. 1993;153:2474-80.
6. Leo RJ. Concise guide to pain management for psychiatrists. Arlington, Va: American Psychiatric Publishing, Inc., 2003.
7. Sullivan MD, Turner JA, Romano J. Chronic pain in primary care. Identification and management of psychosocial factors. J Fam Pract 1991;32(2):193-9.
8. Holmgren A, Wise MG, Bouckoms AJ. Pain management. In: Wise MG, Rundell JR (eds). Psychiatry in the medically ill (2nd ed). Washington, DC: American Psychiatric Publishing Inc., 2002;989-1013.
9. Naifeh KH. Psychometric testing in functional GI disorders in: Olden K (ed). Handbook of functional GI disorders. New York: Marcel Dekker, 1996;79-126.
10. Fishbain DA. The association of chronic pain and suicide. Semin Clin Neuropsychiatry 1999;4(3):221-7.
11. Fishbain DA. Medico-legal rounds: medical-legal issues and breaches of “standards of medical care” in opioid tapering for alleged opioid addiction. Pain Med 2002;3(2):135-42.
12. Doan BD, Wadden NP. Relationships between depressive symptoms and descriptions of chronic pain. Pain 1989;36:75-84.
13. Franklin JE, Leamon MH, Frances RJ. Substance-related disorders. In: Wise MG, Rundell JR (eds). Psychiatry in the medically ill (2nd ed). Washington DC: American Psychiatric Publishing, 2002;417-53.
14. Ansari A. The efficacy of newer antidepressants in the treatment of chronic pain: a review of current literature. Harv Rev Psychiatry 2000;7(5):257-77.
15. NIH Technology Assessment Panel. Integration of behavioral relaxation approaches into the treatment of chronic pain and insomnia. JAMA 1996;276(4):313-18.
16. Morley S, Eccleston C, Williams A. Systematic review and metaanalysis of randomized controlled trials of cognitive behaviour therapy and behaviour therapy for chronic pain in adults, excluding headache. Pain 1999;80:1-13
17. Magni G. On the relationship between chronic pain and depression when there is no organic lesion. Pain 1987;31:1-21.
18. Markowitz JS, Patrick KS. Venlafaxine-tramadol similarities. Med Hypotheses 1998;5:167-8
19. Bradley RH, Barkin RL, Jerome J, et al. Efficacy of venlafaxine for the long-term treatment of chronic pain with associated major depressive disorder. Am J Ther 2003;10(5):318-23.
20. Sindrup SH, Bach FW, Madsen C, et al. Venlafaxine vs. imipramine in painful polyneuropathy—a randomized controlled trial. Neurology 2003;60:1284-9.
21. Kathpal GS. Role of SSRIs in the management of migraine. Headache Quarterly 1998;9:265-6.
22. Mallinckrodt CH, Goldstein DJ, Detke MJ, et al. Duloxetine: a new treatment for the emotional and physical symptoms of depression. Primary Care Companion J Clin Psychiatry 2003;5(1):19-28.
23. Lynch ME. Antidepressants as analgesics: a review of randomized control trials. J Psychiatry Neurosci 2001;26-36.
24. Swerdlow M. Anticonvulsant drugs and chronic pain. Clin Neuropharmacol 1984;7(1):51-82.
Hypnosis: Brief interventions offer key to managing pain and anxiety
Mr. M, a world-class athlete, collapsed suddenly in an alley. He was rushed to a hospital emergency room, where he nearly died of internal bleeding from a grapefruit-sized abdominal lymphoma. He was hospitalized and placed on chemotherapy.
Increasing doses of opiates hardly reduced his pain, and he became extremely anxious. Staff described him as “climbing the walls.” He lay in bed writhing, and his parents feared he was becoming a “drug addict.”
Anxiety about his life-threatening illness was clearly compounding his pain, so his attending physician ordered a psychiatric evaluation. When I interviewed the patient, I felt that hypnosis could help.
Hypnosis—as a state of highly focused attention—can help us treat patients’ anxiety, phobias, pain, posttraumatic stress disorder (PTSD), and dissociative disorders. With training, an experienced psychiatrist can quickly start using hypnosis as an adjunct to other therapies.
This article describes how hypnosis helped Mr. M and a young woman traumatized by a criminal assault. Based on my experience and the literature, I discuss what hypnosis is, what training is required, how to measure hypnotizability, and the value of hypnosis in helping patients control their anxiety, posttraumatic, and dissociative states.
Case continued: ‘Surfing’ in Hawaii
When I met Mr. M in the hospital, I acknowledged his distress and the reasons for it, saying “You don’t really want to be here, do you?”
“How many years of medical training did it take you to figure that out?” he replied.
“Well then,” I said, “let’s go somewhere else. Where would you like to be right now?”
He responded, “I’ve never surfed.”
“Good,” I replied, “let’s go to Hawaii.” In hypnosis, I had him picture himself surfing. He continued to groan, but the pattern changed. “What happened?” I asked. “I fell off the surfboard,” he replied. “OK, get back on, and do it right,” I told him.
He learned to practice self-hypnosis, which markedly reduced his anxiety and pain. Two days later he was off pain medications and joking with the nurses in the hall. The attending physician noted in the patient’s record: “Patient off pain meds. Tumor must be regressing.”
What is hypnosis?
Mr. M’s response, though unusually strong, underscores the fact that hypnosis can rapidly produce analgesia and anxiolysis in the medical setting. Hypnosis—often called “believed-in imagination”—is characterized by an ability to sustain a state of attentive, receptive, intense focal concentration with diminished peripheral awareness. The hypnotized person is awake and alert, not asleep. Hypnosis’ three main components are absorption, dissociation, and suggestibility.
Biological basis. The hypnotic state has no brain “signature” per se, but brain imaging portrays hypnosis as a state of alertness with altered anterior cingulate gyrus activation, which helps to focus attention.1-3 Hypnotized persons can demonstrably alter blood flow in brain regions involved in perceptual processing in response to suggestions of altered perception, whether somatosensory, visual, or olfactory.4,5 Thus, patients report not only reduced pain but changes in how they experience pain with hypnotic analgesia.
The brain’s dopamine neurotransmitter system—especially in the frontal lobes—also may be involved in hypnosis, as highly hypnotizable persons have elevated levels of dopamine metabolites in their cerebrospinal fluid.6
Hypnotic trance. The trance experience is often best explained to patients as similar to being absorbed in a good novel. One loses awareness of one’s surroundings and enters the imagined world. When the novel is finished, the reader requires a moment of reorientation to the surrounding world.
A trance is a state of sustained, attentive-receptive concentration in response to a signal from within or from someone else. The signal activates this shift of awareness and permits more-intensive concentration in a designated direction.
All hypnosis is self-hypnosis. Much of its clinical value is that it can be self-induced throughout the day and whenever symptoms emerge. During the first weeks, patients can be encouraged to practice every 1 or 2 hours.
Applying hypnosis to practice. A well-trained clinician can learn to use hypnosis in classes offered by the two professional hypnosis societies or the American Psychiatric Association (Box 1) Because hypnosis is not something “done to” a patient but rather a capacity to be measured, tapped, and utilized, psychiatrists can integrate hypnosis into clinical practice after some initial training, with ongoing learning and supervision.
Who can be hypnotized?
Not everyone is equally hypnotizable, and hypnotizability is a stable and measurable trait. Approximately one-quarter of adults cannot respond to hypnotic instructions, whereas 10% are extremely hypnotizable.7
Brief, clinically useful tests of hypnotic responsiveness have been developed, such as the Hypnotic Induction Profile (HIP).8 The clinician usually can induce the trance experience and systematically measure the patient’s response within 5 minutes. A HIP score of 5 indicates usable hypnotizability.
The HIP test includes instructions to produce a sense of lightness in the left arm and hand, with tests of response to this instruction. Response is characterized by dissociation, hand elevation after it is lowered, involuntariness, response to the cutoff signal, and altered sensation.
Turning hypnotic induction into a test of hypnotic capacity transforms the initial encounter by:
- removing pressure on the clinician to successfully hypnotize the subject
- reducing patients’ experiences of complying with the clinician’s wishes, rather than exploring and discovering their own hypnotic capacity.
Placing the hypnotic experience in the context of a test also makes it consonant with other medical examinations and procedures.8
Once a patient’s hypnotizability is determined, structured measurement is no longer necessary. The test-retest correlation for hypnotizability scores is 0.7 over 25 years, which is more consistent than IQ testing.7 Subsequent inductions usually can be generated by the patient or signaled by the clinician, and only seconds are required for the shift into trance.
Effective, safe work with hypnosis requires clinical expertise in diagnostic assessment and choosing treatment options. Psychiatrists can learn techniques for inducing, measuring, and using hypnotic responsiveness in introductory and advanced workshops, supplemented by local supervision.
Courses in hypnosis are offered by many medical schools. Postgraduate training is available at annual meetings of the American Psychiatric Association, Society for Clinical and Experimental Hypnosis, and American Society of Clinical Hypnosis. The two hypnosis societies offer intensive workshops for psychiatrists, psychologists, and other health care professionals.
Useful text books also are available:
- Spiegel H, Spiegel D. Trance and treatment: clinical uses of hypnosis. Washington, DC: American Psychiatric Publishing, 2004.
- Zarren JI, Eimer BN. Brief cognitive hypnosis: facilitating the change of dysfunctional behavior. New York: Springer Publishing, 2002.
- Lynn SJ, Kirsch I, Rhue JW. Casebook of clinical hypnosis. Washington, DC: American Psychological Association, 1996.
- Fromm E, Kahn SP. Self-hypnosis: the Chicago paradigm. New York: Guilford Press, 1990.
Reducing anxiety
Anxiety can be understood as a vaguely defined but immobilizing sense of distress. Lack of clarity about the discomfort’s source enhances the patient’s sense of helplessness and avoidance. One therapeutic challenge is to convert anxiety into fear—to give it a focus so that something can be done about it.
Imagine yourself floating in a bath, a lake, a hot tub, or just floating in space. With each breath out, let a little more tension out of your body. Just enjoy this pleasant sense of floating, and notice how you can use your store of memories and fantasies to help yourself and your body feel better.
“While you imagine yourself floating, in your mind’s eye visualize an imaginary screen: a movie, TV, or computer screen, or, if you wish, a piece of clear blue sky. On that screen project your thoughts, fears, worries, ideas, feelings, or memories, while you maintain the pleasant sense of floating in your body. You establish this clear sense of your body floating here, while you relate to your thoughts and ideas out there.
“Once you have established this screen, divide it in half. Use the left side as your ‘worry screen.’ Picture one thing that causes you anxiety on this screen and learn to manage the feelings of discomfort that accompany it. Now use the right side as your ‘problem-solving’ screen. Brainstorm something you can do about the problem on the left, all the while maintaining a sense of floating in your body.
“You may have to ‘freeze’ what is on the ‘worry screen’ and re-establish the floating several times. This allows you to develop new means of coping with the things that are making you anxious, one at a time.”
Anxiety sets up a negative feedback cycle between psychological preoccupation and somatic discomfort, a “snowball effect” in which subjective anxiety and somatic tension reinforce each other. Hypnosis can help reduce anxiety and induce relaxation,9 and its dissociative component can help separate anxiety’s psychological and somatic components.
Hypnosis is as effective at reducing anxiety as 1 mg of alprazolam, at least in a study of college students.10 Student volunteers with high and low hypnotizability were given alprazolam, 1 mg, and a hypnotic suggestion based on their reactions to the drug. Four days later, when students received hypnosis only and hypnosis plus alprazolam:
- combination therapy reduced anxiety more effectively than did hypnosis or alprazolam alone, as measured by the Profile of Mood States tension-anxiety scale
- improvement was comparable with hypnosis or alprazolam alone
- highly hypnotizable students showed significantly greater relaxation than did those with low hypnotizability in all three treatment groups
- EEG data showed similar frontal and occipital changes in the alprazolam and hypnotic suggestion groups.
In randomized trials, simple self-hypnosis training has reduced pain and anxiety during medical procedures, reducing procedure time by an average 17 minutes and resulting in fewer complications.11
A typical hypnotic instruction for managing anxiety is provided in Box 2. This approach teaches patients how to deal with stressors that complicate their anxiety and to control their somatic response. Hypnosis expands patients’ repertoire of responses and enables them to feel less helpless.
Confronting phobias
Phobic symptoms of fear and avoidance or exposure with distress respond especially well to brief hypnosis interventions. Although behavior modification and antidepressants also can treat phobias successfully, one or two hypnosis sessions often can reduce or cure phobic symptoms.
For example, one can help patients with airplane phobia prepare for flight by going into a hypnotic state and learning three concepts:
- Think of the airplane as an extension of the body, such as a bicycle.
- Float with the plane.
- Think about the difference between probability and possibility.
The hypnotic state—with its focused attention and physical relaxation—can amplify this cognitive restructuring technique. Phobic patients can feel more in control of their somatic reactions and, by extrapolation, the flying experience itself. In one study, 52% of patients taught this self-hypnosis exercise remained improved or cured at least 7 years later.12
Treating traumatic reactions
Evidence is growing that trauma elicits dissociation. Thus, hypnosis could help us understand and treat traumatic reactions, including patients with acute and posttraumatic stress disorder (PTSD) and dissociative disorders.
The hypnotic state’s controlled dissociation can be used to model the uncontrolled dissociation represented by posttraumatic phenomena such as flashbacks, numbing, and amnesia.13 This view is supported by evidence that PTSD is associated with high hypnotizability.14,15
Acute stress disorder—as introduced in DSM-IV16—is characterized by prominent dissociative symptoms, with intrusion, avoidance, and hyperarousal. These diagnostic criteria recognize that acute dissociation is a common and predictable reaction to trauma.
Hypnosis involving grief work, exploration of trauma-related transference issues, and emotional expression are effective psychotherapies for persons exposed to trauma. Becoming familiar with hypnotic states can teach patients to recognize, understand, and control their dissociative states.
Evidence suggests that hypnosis’ intense concentration may reverse the dissociative mind fragmentation caused by trauma.17 Traumatic memories may seem less overwhelming and intrusive once patients discover they can:
- exert greater control over memory access and retrieval
- work through and assimilate disturbing thoughts.
The controlled experience of hypnotic abreaction (reliving traumatic and other memories with strong emotion) provides boundaries for psychotherapeutic grief work.18,19 Instead of telling patients not to ruminate over a traumatic event, the clinician instructs the patient how to think about the experience.
The inferred message is that the patient can work on other things—such as relationships and daily living problems—after this therapeutic work is done.
Patients are slowly separated from the victim role. The goal is to help them restructure their memories, both cognitively and emotionally. They bear the memories’ impact, yet come to see the information differently.7 Traumatic input becomes more bearable when linked to a cognitively restructured recognition of an adaptive response.,20 For example, patients may acknowledge what they did during a traumatic event that was self-protective or helped others.
PTSD. Hypnosis shares common elements with other cognitive and behavioral treatments for PTSD, including exposure to traumatic memories for cognitive and emotional processing. Few studies have examined using hypnosis to treat PTSD, but evidence suggests it is at least as effective as other cognitive-behavioral treatments.20,21
Patients can be taught to view PTSD’s intrusive memories and bodily symptoms as re-experiencing painful memories. The memories often intrude less frequently after patients find a controlled method—such as self-hypnosis—to access and work them through.22
Ms. J hoped hypnosis could help her better visualize the face of an assailant who had attacked her as she returned at dusk from the grocery store. She had fought off his attempt to drag her into her apartment and rape her. The police showed little interest in pursuing him, however, because the sexual assault had not been completed. After the police left, she had a grand mal seizure. She had suffered a basalar skull fracture.
Ms. J was highly hypnotizable and learned the split-screen technique. While visualizing the assault on the left screen, she realized something that had not been clear to her before: “From the look on his face, I can see he wants to kill me. If he gets me into my apartment, he will kill me.”
She focused on this realization and the image of his hatred and threat to her. The therapist asked her to picture on the right screen something she had done to protect herself. She said: “He is surprised that I am fighting so hard. He doesn’t expect me to put up such a fight.”
She emerged from hypnosis understanding that she had been in more danger than she realized. Thus, despite the disappointment of having no clearer idea of what he looked like (it was quite dark when he attacked her), she had a restructured perspective about what had occurred.
Before this session, Ms. J had felt guilty that she had gotten herself so seriously injured. Afterward, she could better tolerate the memory of the attack because it was coupled with cognitive awareness that her actions may have saved her life.
Self-blame. Many trauma victims would rather feel guilty than helpless. They blame themselves inappropriately for events over which they had no control, rather than accept their helplessness. They misuse hindsight about the trauma to assume the events were predictable and therefore avoidable. They imagine they can replay the events and change the outcome.
Such an approach to trauma can be profoundly demoralizing, leaving victims burdened by needless guilt and shame. Helping them face and bear the feelings associated with traumatic events can free them from efforts to “undo” or take responsibility for the trauma and accept what happened.
Split-screen technique. Using hypnosis with a “split-screen” technique can help patients restructure the memory of trauma. The left screen symbolizes the trauma in condensed form. The right screen helps patients focus on how they tried to master the situation. This grief work allows patients to acknowledge, bear, and put into perspective the humiliation of the experience and their loss of invulnerability, health, or loved ones (Box 3).18
Dissociation. Dissociating during a threatening situation may enable a person to put aside some awareness of the danger and take self-protective action. Persistent dissociation, however, may make it too easy to avoid working through the traumatic experiences later on.22-24
Dissociation makes subsequent exposure to reminders of the trauma more similar to a reexperiencing rather than a controlled remembering of it. This can trigger physiologic stress reactions and lead to or worsen PTSD.25-27
Dissociative disorders can be understood as chronic and severe PTSDs.28 Many individuals with dissociative disorders have histories of sexual and physical abuse.29-31 Clearly, traumatic experiences sensitize survivors to subsequent trauma through conditioned activation of fear circuitry involving the amygdala, hippocampus, and frontal lobes.32
Hypnosis can be especially helpful—both for diagnosis and therapy.33 It can assist the controlled recovery of memories, while allowing some images to remain dissociated from cognition until the patient is ready to deal with them. The patient can turn memories on and off by entering and exiting the hypnotic state and thereby recover and reprocess memories at a tolerable pace.
Related resources
- Society for Clinical and Experimental Hypnosis. http://ijceh.educ.wsu.edu
- American Society of Clinical Hypnosis. www.asch.net
- American Psychological Association, Division 30. Society of Psychological Hypnosis. http://www.apa.org/about/division/div30.html
1. Spiegel D, Jasiukaitis P. Hypnosis: Brain basis. In: Smith BH (ed). Elsevier’s encyclopedia of neuroscience. The Netherlands: Elsevier Science, 1999.
2. Rainville P, Hofbauer RK, Bushnell MC, et al. Hypnosis modulates activity in brain structures involved in the regulation of consciousness. J Cogn Neurosci 2002;14:887-901.
3. Rainville P, Duncan GH, Price DD, et al. Pain affect encoded in human anterior cingulate but not somatosensory cortex. Science 1997;277:968-71.
4. Kosslyn SM, Thompson WL, Costantini-Ferrando MF, et al. Hypnotic visual illusion alters color processing in the brain. Am J Psychiatry 2000;157:1279-84.
5. Spiegel D. Negative and positive visual hypnotic hallucinations: attending inside and out. Int J Clin Exp Hypn 2003;51:130-46.
6. Spiegel D, King R. Hypnotizability and CSF HVA levels among psychiatric patients. Biol Psychiatry 1992;31:95-8.
7. Piccione C, Hilgard ER, Zimbardo PG. On the degree of stability of measured hypnotizability over a 25-year period. J Pers Soc Psychol 1989;56:289-95.
8. Spiegel H, Spiegel D. Trance and treatment: Clinical uses of hypnosis. Washington, DC: American Psychiatric Press, 2004.
9. Wertz JM, Sayette MA. Effects of smoking opportunity on attentional bias in smokers. Psychol Addict Behav 2001;15:268-71.
10. Nishith P, Barabasz A, Barabasz M, Warner D. Brief hypnosis substitutes for alprazolam use in college students: transient experiences and quantitative EEG responses. Am J Clin Hypn 1999;41:262-8.
11. Lang EV, Benotsch EG, Fick LJ, et al. Adjunctive nonpharmacological analgesia for invasive medical procedures: a randomised trial. Lancet 2000;355:1486-90.
12. Spiegel D, Frischholz EJ, Maruffi B, Spiegel H. Hypnotic responsitivity and the treatment of flying phobia. Am J Clin Hypn 1981;23:239-47.
13. Butler LD, Duran EFD, Jasiukaitis P, et al. Hypnotizability and traumatic experience: a diathesis-stress model of dissociative symptomatology. Am J Psychiatry 1996;153:42-63.
14. Spiegel D. Dissociation and hypnosis in post-traumatic stress disorder. J Trauma Stress 1988;1:17-33.
15. Stutman RK, Bliss EL. Posttraumatic stress disorder, hypnotizability, and imagery. Am J Psychiatry 1985;142:741-3.
16. Diagnostic and statistical manual of mental disorders (4th ed, text rev). Washington, DC: American Psychiatric Association, 2000.
17. Maldonado JR, Spiegel D. Trauma, dissociation and hypnotizability. In: Marmar R, Bremmer D (eds). Trauma, memory and dissociation. Washington, DC: American Psychiatric Press, 1998.
18. Lindemann E. Symptomatology and management of acute grief. Am J Psychiatry 1994;151:155-60.
19. Spiegel D. Vietnam grief work using hypnosis. Am J Clin Hypn 1981;24:33-40.
20. Foa EB, Davidson JRT, Frances A. Treatment of posttraumatic stress disorder. J Clin Psychiatry 1999;50:4-69.
21. Brom D, Kleber RJ, Defare PB. Brief psychotherapy for post-traumatic stress disorder. J Consult Clin Psychol 1989;57:607-12.
22. Spiegel D. Hypnosis and implicit memory: automatic processing of explicit content. Am J Clin Hypn 1998;40:231-40.
23. Spiegel D. Multiple personality as a post-traumatic stress disorder. Psychiatr Clin North Am 1984;7:101-10.
24. Kluft RP. Dissociation as a response to extreme trauma. In: Kluft RP (ed). Childhood antecedents of multiple personality. Washington, DC: American Psychiatric Press, 1985:66-97.
25. Marmar CR, Weiss DS, Metzler T. Peritraumatic dissociation and posttraumatic stress disorder. In: Bremner JD, Marmar C (eds). Trauma, memory, and dissociation. Washington, DC: American Psychiatric Press, 1998;229-52.
26. Birmes P. Peritraumatic dissociation, acute stress, and early posttraumatic stress disorder in victims of general crime. Can J Psychiatry 2001;46:649-51.
27. Spiegel D. Hypnosis, dissociation and trauma. In: Burrows GD, Stanley RO, Bloom PB (eds). Clinical hypnosis. New York: John Wiley & Sons, 2001;143-58.
28. Spiegel D, Cardena E. Disintegrated experience: the dissociative disorders revisited. J Abnorm Psychol 1991;100:366-78.
29. Chu JA, Dill DL. Dissociative symptoms in relation to childhood physical and sexual abuse. Am J Psychiatry 1990;147:887-92.
30. Kluft RP. Childhood antecedents of multiple personality. Washington, DC: American Psychiatric Press, 1985.
31. Spiegel D. Dissociating damage. Am J Clin Hypn 1986;29:123-31.
32. LeDoux J. Synaptic self: How our brains become who we are. New York: Viking Press, 2002.
33. Putnam FW. Using hypnosis for therapeutic abreactions. Psychiatr Med 1992;10:51-65.
Mr. M, a world-class athlete, collapsed suddenly in an alley. He was rushed to a hospital emergency room, where he nearly died of internal bleeding from a grapefruit-sized abdominal lymphoma. He was hospitalized and placed on chemotherapy.
Increasing doses of opiates hardly reduced his pain, and he became extremely anxious. Staff described him as “climbing the walls.” He lay in bed writhing, and his parents feared he was becoming a “drug addict.”
Anxiety about his life-threatening illness was clearly compounding his pain, so his attending physician ordered a psychiatric evaluation. When I interviewed the patient, I felt that hypnosis could help.
Hypnosis—as a state of highly focused attention—can help us treat patients’ anxiety, phobias, pain, posttraumatic stress disorder (PTSD), and dissociative disorders. With training, an experienced psychiatrist can quickly start using hypnosis as an adjunct to other therapies.
This article describes how hypnosis helped Mr. M and a young woman traumatized by a criminal assault. Based on my experience and the literature, I discuss what hypnosis is, what training is required, how to measure hypnotizability, and the value of hypnosis in helping patients control their anxiety, posttraumatic, and dissociative states.
Case continued: ‘Surfing’ in Hawaii
When I met Mr. M in the hospital, I acknowledged his distress and the reasons for it, saying “You don’t really want to be here, do you?”
“How many years of medical training did it take you to figure that out?” he replied.
“Well then,” I said, “let’s go somewhere else. Where would you like to be right now?”
He responded, “I’ve never surfed.”
“Good,” I replied, “let’s go to Hawaii.” In hypnosis, I had him picture himself surfing. He continued to groan, but the pattern changed. “What happened?” I asked. “I fell off the surfboard,” he replied. “OK, get back on, and do it right,” I told him.
He learned to practice self-hypnosis, which markedly reduced his anxiety and pain. Two days later he was off pain medications and joking with the nurses in the hall. The attending physician noted in the patient’s record: “Patient off pain meds. Tumor must be regressing.”
What is hypnosis?
Mr. M’s response, though unusually strong, underscores the fact that hypnosis can rapidly produce analgesia and anxiolysis in the medical setting. Hypnosis—often called “believed-in imagination”—is characterized by an ability to sustain a state of attentive, receptive, intense focal concentration with diminished peripheral awareness. The hypnotized person is awake and alert, not asleep. Hypnosis’ three main components are absorption, dissociation, and suggestibility.
Biological basis. The hypnotic state has no brain “signature” per se, but brain imaging portrays hypnosis as a state of alertness with altered anterior cingulate gyrus activation, which helps to focus attention.1-3 Hypnotized persons can demonstrably alter blood flow in brain regions involved in perceptual processing in response to suggestions of altered perception, whether somatosensory, visual, or olfactory.4,5 Thus, patients report not only reduced pain but changes in how they experience pain with hypnotic analgesia.
The brain’s dopamine neurotransmitter system—especially in the frontal lobes—also may be involved in hypnosis, as highly hypnotizable persons have elevated levels of dopamine metabolites in their cerebrospinal fluid.6
Hypnotic trance. The trance experience is often best explained to patients as similar to being absorbed in a good novel. One loses awareness of one’s surroundings and enters the imagined world. When the novel is finished, the reader requires a moment of reorientation to the surrounding world.
A trance is a state of sustained, attentive-receptive concentration in response to a signal from within or from someone else. The signal activates this shift of awareness and permits more-intensive concentration in a designated direction.
All hypnosis is self-hypnosis. Much of its clinical value is that it can be self-induced throughout the day and whenever symptoms emerge. During the first weeks, patients can be encouraged to practice every 1 or 2 hours.
Applying hypnosis to practice. A well-trained clinician can learn to use hypnosis in classes offered by the two professional hypnosis societies or the American Psychiatric Association (Box 1) Because hypnosis is not something “done to” a patient but rather a capacity to be measured, tapped, and utilized, psychiatrists can integrate hypnosis into clinical practice after some initial training, with ongoing learning and supervision.
Who can be hypnotized?
Not everyone is equally hypnotizable, and hypnotizability is a stable and measurable trait. Approximately one-quarter of adults cannot respond to hypnotic instructions, whereas 10% are extremely hypnotizable.7
Brief, clinically useful tests of hypnotic responsiveness have been developed, such as the Hypnotic Induction Profile (HIP).8 The clinician usually can induce the trance experience and systematically measure the patient’s response within 5 minutes. A HIP score of 5 indicates usable hypnotizability.
The HIP test includes instructions to produce a sense of lightness in the left arm and hand, with tests of response to this instruction. Response is characterized by dissociation, hand elevation after it is lowered, involuntariness, response to the cutoff signal, and altered sensation.
Turning hypnotic induction into a test of hypnotic capacity transforms the initial encounter by:
- removing pressure on the clinician to successfully hypnotize the subject
- reducing patients’ experiences of complying with the clinician’s wishes, rather than exploring and discovering their own hypnotic capacity.
Placing the hypnotic experience in the context of a test also makes it consonant with other medical examinations and procedures.8
Once a patient’s hypnotizability is determined, structured measurement is no longer necessary. The test-retest correlation for hypnotizability scores is 0.7 over 25 years, which is more consistent than IQ testing.7 Subsequent inductions usually can be generated by the patient or signaled by the clinician, and only seconds are required for the shift into trance.
Effective, safe work with hypnosis requires clinical expertise in diagnostic assessment and choosing treatment options. Psychiatrists can learn techniques for inducing, measuring, and using hypnotic responsiveness in introductory and advanced workshops, supplemented by local supervision.
Courses in hypnosis are offered by many medical schools. Postgraduate training is available at annual meetings of the American Psychiatric Association, Society for Clinical and Experimental Hypnosis, and American Society of Clinical Hypnosis. The two hypnosis societies offer intensive workshops for psychiatrists, psychologists, and other health care professionals.
Useful text books also are available:
- Spiegel H, Spiegel D. Trance and treatment: clinical uses of hypnosis. Washington, DC: American Psychiatric Publishing, 2004.
- Zarren JI, Eimer BN. Brief cognitive hypnosis: facilitating the change of dysfunctional behavior. New York: Springer Publishing, 2002.
- Lynn SJ, Kirsch I, Rhue JW. Casebook of clinical hypnosis. Washington, DC: American Psychological Association, 1996.
- Fromm E, Kahn SP. Self-hypnosis: the Chicago paradigm. New York: Guilford Press, 1990.
Reducing anxiety
Anxiety can be understood as a vaguely defined but immobilizing sense of distress. Lack of clarity about the discomfort’s source enhances the patient’s sense of helplessness and avoidance. One therapeutic challenge is to convert anxiety into fear—to give it a focus so that something can be done about it.
Imagine yourself floating in a bath, a lake, a hot tub, or just floating in space. With each breath out, let a little more tension out of your body. Just enjoy this pleasant sense of floating, and notice how you can use your store of memories and fantasies to help yourself and your body feel better.
“While you imagine yourself floating, in your mind’s eye visualize an imaginary screen: a movie, TV, or computer screen, or, if you wish, a piece of clear blue sky. On that screen project your thoughts, fears, worries, ideas, feelings, or memories, while you maintain the pleasant sense of floating in your body. You establish this clear sense of your body floating here, while you relate to your thoughts and ideas out there.
“Once you have established this screen, divide it in half. Use the left side as your ‘worry screen.’ Picture one thing that causes you anxiety on this screen and learn to manage the feelings of discomfort that accompany it. Now use the right side as your ‘problem-solving’ screen. Brainstorm something you can do about the problem on the left, all the while maintaining a sense of floating in your body.
“You may have to ‘freeze’ what is on the ‘worry screen’ and re-establish the floating several times. This allows you to develop new means of coping with the things that are making you anxious, one at a time.”
Anxiety sets up a negative feedback cycle between psychological preoccupation and somatic discomfort, a “snowball effect” in which subjective anxiety and somatic tension reinforce each other. Hypnosis can help reduce anxiety and induce relaxation,9 and its dissociative component can help separate anxiety’s psychological and somatic components.
Hypnosis is as effective at reducing anxiety as 1 mg of alprazolam, at least in a study of college students.10 Student volunteers with high and low hypnotizability were given alprazolam, 1 mg, and a hypnotic suggestion based on their reactions to the drug. Four days later, when students received hypnosis only and hypnosis plus alprazolam:
- combination therapy reduced anxiety more effectively than did hypnosis or alprazolam alone, as measured by the Profile of Mood States tension-anxiety scale
- improvement was comparable with hypnosis or alprazolam alone
- highly hypnotizable students showed significantly greater relaxation than did those with low hypnotizability in all three treatment groups
- EEG data showed similar frontal and occipital changes in the alprazolam and hypnotic suggestion groups.
In randomized trials, simple self-hypnosis training has reduced pain and anxiety during medical procedures, reducing procedure time by an average 17 minutes and resulting in fewer complications.11
A typical hypnotic instruction for managing anxiety is provided in Box 2. This approach teaches patients how to deal with stressors that complicate their anxiety and to control their somatic response. Hypnosis expands patients’ repertoire of responses and enables them to feel less helpless.
Confronting phobias
Phobic symptoms of fear and avoidance or exposure with distress respond especially well to brief hypnosis interventions. Although behavior modification and antidepressants also can treat phobias successfully, one or two hypnosis sessions often can reduce or cure phobic symptoms.
For example, one can help patients with airplane phobia prepare for flight by going into a hypnotic state and learning three concepts:
- Think of the airplane as an extension of the body, such as a bicycle.
- Float with the plane.
- Think about the difference between probability and possibility.
The hypnotic state—with its focused attention and physical relaxation—can amplify this cognitive restructuring technique. Phobic patients can feel more in control of their somatic reactions and, by extrapolation, the flying experience itself. In one study, 52% of patients taught this self-hypnosis exercise remained improved or cured at least 7 years later.12
Treating traumatic reactions
Evidence is growing that trauma elicits dissociation. Thus, hypnosis could help us understand and treat traumatic reactions, including patients with acute and posttraumatic stress disorder (PTSD) and dissociative disorders.
The hypnotic state’s controlled dissociation can be used to model the uncontrolled dissociation represented by posttraumatic phenomena such as flashbacks, numbing, and amnesia.13 This view is supported by evidence that PTSD is associated with high hypnotizability.14,15
Acute stress disorder—as introduced in DSM-IV16—is characterized by prominent dissociative symptoms, with intrusion, avoidance, and hyperarousal. These diagnostic criteria recognize that acute dissociation is a common and predictable reaction to trauma.
Hypnosis involving grief work, exploration of trauma-related transference issues, and emotional expression are effective psychotherapies for persons exposed to trauma. Becoming familiar with hypnotic states can teach patients to recognize, understand, and control their dissociative states.
Evidence suggests that hypnosis’ intense concentration may reverse the dissociative mind fragmentation caused by trauma.17 Traumatic memories may seem less overwhelming and intrusive once patients discover they can:
- exert greater control over memory access and retrieval
- work through and assimilate disturbing thoughts.
The controlled experience of hypnotic abreaction (reliving traumatic and other memories with strong emotion) provides boundaries for psychotherapeutic grief work.18,19 Instead of telling patients not to ruminate over a traumatic event, the clinician instructs the patient how to think about the experience.
The inferred message is that the patient can work on other things—such as relationships and daily living problems—after this therapeutic work is done.
Patients are slowly separated from the victim role. The goal is to help them restructure their memories, both cognitively and emotionally. They bear the memories’ impact, yet come to see the information differently.7 Traumatic input becomes more bearable when linked to a cognitively restructured recognition of an adaptive response.,20 For example, patients may acknowledge what they did during a traumatic event that was self-protective or helped others.
PTSD. Hypnosis shares common elements with other cognitive and behavioral treatments for PTSD, including exposure to traumatic memories for cognitive and emotional processing. Few studies have examined using hypnosis to treat PTSD, but evidence suggests it is at least as effective as other cognitive-behavioral treatments.20,21
Patients can be taught to view PTSD’s intrusive memories and bodily symptoms as re-experiencing painful memories. The memories often intrude less frequently after patients find a controlled method—such as self-hypnosis—to access and work them through.22
Ms. J hoped hypnosis could help her better visualize the face of an assailant who had attacked her as she returned at dusk from the grocery store. She had fought off his attempt to drag her into her apartment and rape her. The police showed little interest in pursuing him, however, because the sexual assault had not been completed. After the police left, she had a grand mal seizure. She had suffered a basalar skull fracture.
Ms. J was highly hypnotizable and learned the split-screen technique. While visualizing the assault on the left screen, she realized something that had not been clear to her before: “From the look on his face, I can see he wants to kill me. If he gets me into my apartment, he will kill me.”
She focused on this realization and the image of his hatred and threat to her. The therapist asked her to picture on the right screen something she had done to protect herself. She said: “He is surprised that I am fighting so hard. He doesn’t expect me to put up such a fight.”
She emerged from hypnosis understanding that she had been in more danger than she realized. Thus, despite the disappointment of having no clearer idea of what he looked like (it was quite dark when he attacked her), she had a restructured perspective about what had occurred.
Before this session, Ms. J had felt guilty that she had gotten herself so seriously injured. Afterward, she could better tolerate the memory of the attack because it was coupled with cognitive awareness that her actions may have saved her life.
Self-blame. Many trauma victims would rather feel guilty than helpless. They blame themselves inappropriately for events over which they had no control, rather than accept their helplessness. They misuse hindsight about the trauma to assume the events were predictable and therefore avoidable. They imagine they can replay the events and change the outcome.
Such an approach to trauma can be profoundly demoralizing, leaving victims burdened by needless guilt and shame. Helping them face and bear the feelings associated with traumatic events can free them from efforts to “undo” or take responsibility for the trauma and accept what happened.
Split-screen technique. Using hypnosis with a “split-screen” technique can help patients restructure the memory of trauma. The left screen symbolizes the trauma in condensed form. The right screen helps patients focus on how they tried to master the situation. This grief work allows patients to acknowledge, bear, and put into perspective the humiliation of the experience and their loss of invulnerability, health, or loved ones (Box 3).18
Dissociation. Dissociating during a threatening situation may enable a person to put aside some awareness of the danger and take self-protective action. Persistent dissociation, however, may make it too easy to avoid working through the traumatic experiences later on.22-24
Dissociation makes subsequent exposure to reminders of the trauma more similar to a reexperiencing rather than a controlled remembering of it. This can trigger physiologic stress reactions and lead to or worsen PTSD.25-27
Dissociative disorders can be understood as chronic and severe PTSDs.28 Many individuals with dissociative disorders have histories of sexual and physical abuse.29-31 Clearly, traumatic experiences sensitize survivors to subsequent trauma through conditioned activation of fear circuitry involving the amygdala, hippocampus, and frontal lobes.32
Hypnosis can be especially helpful—both for diagnosis and therapy.33 It can assist the controlled recovery of memories, while allowing some images to remain dissociated from cognition until the patient is ready to deal with them. The patient can turn memories on and off by entering and exiting the hypnotic state and thereby recover and reprocess memories at a tolerable pace.
Related resources
- Society for Clinical and Experimental Hypnosis. http://ijceh.educ.wsu.edu
- American Society of Clinical Hypnosis. www.asch.net
- American Psychological Association, Division 30. Society of Psychological Hypnosis. http://www.apa.org/about/division/div30.html
Mr. M, a world-class athlete, collapsed suddenly in an alley. He was rushed to a hospital emergency room, where he nearly died of internal bleeding from a grapefruit-sized abdominal lymphoma. He was hospitalized and placed on chemotherapy.
Increasing doses of opiates hardly reduced his pain, and he became extremely anxious. Staff described him as “climbing the walls.” He lay in bed writhing, and his parents feared he was becoming a “drug addict.”
Anxiety about his life-threatening illness was clearly compounding his pain, so his attending physician ordered a psychiatric evaluation. When I interviewed the patient, I felt that hypnosis could help.
Hypnosis—as a state of highly focused attention—can help us treat patients’ anxiety, phobias, pain, posttraumatic stress disorder (PTSD), and dissociative disorders. With training, an experienced psychiatrist can quickly start using hypnosis as an adjunct to other therapies.
This article describes how hypnosis helped Mr. M and a young woman traumatized by a criminal assault. Based on my experience and the literature, I discuss what hypnosis is, what training is required, how to measure hypnotizability, and the value of hypnosis in helping patients control their anxiety, posttraumatic, and dissociative states.
Case continued: ‘Surfing’ in Hawaii
When I met Mr. M in the hospital, I acknowledged his distress and the reasons for it, saying “You don’t really want to be here, do you?”
“How many years of medical training did it take you to figure that out?” he replied.
“Well then,” I said, “let’s go somewhere else. Where would you like to be right now?”
He responded, “I’ve never surfed.”
“Good,” I replied, “let’s go to Hawaii.” In hypnosis, I had him picture himself surfing. He continued to groan, but the pattern changed. “What happened?” I asked. “I fell off the surfboard,” he replied. “OK, get back on, and do it right,” I told him.
He learned to practice self-hypnosis, which markedly reduced his anxiety and pain. Two days later he was off pain medications and joking with the nurses in the hall. The attending physician noted in the patient’s record: “Patient off pain meds. Tumor must be regressing.”
What is hypnosis?
Mr. M’s response, though unusually strong, underscores the fact that hypnosis can rapidly produce analgesia and anxiolysis in the medical setting. Hypnosis—often called “believed-in imagination”—is characterized by an ability to sustain a state of attentive, receptive, intense focal concentration with diminished peripheral awareness. The hypnotized person is awake and alert, not asleep. Hypnosis’ three main components are absorption, dissociation, and suggestibility.
Biological basis. The hypnotic state has no brain “signature” per se, but brain imaging portrays hypnosis as a state of alertness with altered anterior cingulate gyrus activation, which helps to focus attention.1-3 Hypnotized persons can demonstrably alter blood flow in brain regions involved in perceptual processing in response to suggestions of altered perception, whether somatosensory, visual, or olfactory.4,5 Thus, patients report not only reduced pain but changes in how they experience pain with hypnotic analgesia.
The brain’s dopamine neurotransmitter system—especially in the frontal lobes—also may be involved in hypnosis, as highly hypnotizable persons have elevated levels of dopamine metabolites in their cerebrospinal fluid.6
Hypnotic trance. The trance experience is often best explained to patients as similar to being absorbed in a good novel. One loses awareness of one’s surroundings and enters the imagined world. When the novel is finished, the reader requires a moment of reorientation to the surrounding world.
A trance is a state of sustained, attentive-receptive concentration in response to a signal from within or from someone else. The signal activates this shift of awareness and permits more-intensive concentration in a designated direction.
All hypnosis is self-hypnosis. Much of its clinical value is that it can be self-induced throughout the day and whenever symptoms emerge. During the first weeks, patients can be encouraged to practice every 1 or 2 hours.
Applying hypnosis to practice. A well-trained clinician can learn to use hypnosis in classes offered by the two professional hypnosis societies or the American Psychiatric Association (Box 1) Because hypnosis is not something “done to” a patient but rather a capacity to be measured, tapped, and utilized, psychiatrists can integrate hypnosis into clinical practice after some initial training, with ongoing learning and supervision.
Who can be hypnotized?
Not everyone is equally hypnotizable, and hypnotizability is a stable and measurable trait. Approximately one-quarter of adults cannot respond to hypnotic instructions, whereas 10% are extremely hypnotizable.7
Brief, clinically useful tests of hypnotic responsiveness have been developed, such as the Hypnotic Induction Profile (HIP).8 The clinician usually can induce the trance experience and systematically measure the patient’s response within 5 minutes. A HIP score of 5 indicates usable hypnotizability.
The HIP test includes instructions to produce a sense of lightness in the left arm and hand, with tests of response to this instruction. Response is characterized by dissociation, hand elevation after it is lowered, involuntariness, response to the cutoff signal, and altered sensation.
Turning hypnotic induction into a test of hypnotic capacity transforms the initial encounter by:
- removing pressure on the clinician to successfully hypnotize the subject
- reducing patients’ experiences of complying with the clinician’s wishes, rather than exploring and discovering their own hypnotic capacity.
Placing the hypnotic experience in the context of a test also makes it consonant with other medical examinations and procedures.8
Once a patient’s hypnotizability is determined, structured measurement is no longer necessary. The test-retest correlation for hypnotizability scores is 0.7 over 25 years, which is more consistent than IQ testing.7 Subsequent inductions usually can be generated by the patient or signaled by the clinician, and only seconds are required for the shift into trance.
Effective, safe work with hypnosis requires clinical expertise in diagnostic assessment and choosing treatment options. Psychiatrists can learn techniques for inducing, measuring, and using hypnotic responsiveness in introductory and advanced workshops, supplemented by local supervision.
Courses in hypnosis are offered by many medical schools. Postgraduate training is available at annual meetings of the American Psychiatric Association, Society for Clinical and Experimental Hypnosis, and American Society of Clinical Hypnosis. The two hypnosis societies offer intensive workshops for psychiatrists, psychologists, and other health care professionals.
Useful text books also are available:
- Spiegel H, Spiegel D. Trance and treatment: clinical uses of hypnosis. Washington, DC: American Psychiatric Publishing, 2004.
- Zarren JI, Eimer BN. Brief cognitive hypnosis: facilitating the change of dysfunctional behavior. New York: Springer Publishing, 2002.
- Lynn SJ, Kirsch I, Rhue JW. Casebook of clinical hypnosis. Washington, DC: American Psychological Association, 1996.
- Fromm E, Kahn SP. Self-hypnosis: the Chicago paradigm. New York: Guilford Press, 1990.
Reducing anxiety
Anxiety can be understood as a vaguely defined but immobilizing sense of distress. Lack of clarity about the discomfort’s source enhances the patient’s sense of helplessness and avoidance. One therapeutic challenge is to convert anxiety into fear—to give it a focus so that something can be done about it.
Imagine yourself floating in a bath, a lake, a hot tub, or just floating in space. With each breath out, let a little more tension out of your body. Just enjoy this pleasant sense of floating, and notice how you can use your store of memories and fantasies to help yourself and your body feel better.
“While you imagine yourself floating, in your mind’s eye visualize an imaginary screen: a movie, TV, or computer screen, or, if you wish, a piece of clear blue sky. On that screen project your thoughts, fears, worries, ideas, feelings, or memories, while you maintain the pleasant sense of floating in your body. You establish this clear sense of your body floating here, while you relate to your thoughts and ideas out there.
“Once you have established this screen, divide it in half. Use the left side as your ‘worry screen.’ Picture one thing that causes you anxiety on this screen and learn to manage the feelings of discomfort that accompany it. Now use the right side as your ‘problem-solving’ screen. Brainstorm something you can do about the problem on the left, all the while maintaining a sense of floating in your body.
“You may have to ‘freeze’ what is on the ‘worry screen’ and re-establish the floating several times. This allows you to develop new means of coping with the things that are making you anxious, one at a time.”
Anxiety sets up a negative feedback cycle between psychological preoccupation and somatic discomfort, a “snowball effect” in which subjective anxiety and somatic tension reinforce each other. Hypnosis can help reduce anxiety and induce relaxation,9 and its dissociative component can help separate anxiety’s psychological and somatic components.
Hypnosis is as effective at reducing anxiety as 1 mg of alprazolam, at least in a study of college students.10 Student volunteers with high and low hypnotizability were given alprazolam, 1 mg, and a hypnotic suggestion based on their reactions to the drug. Four days later, when students received hypnosis only and hypnosis plus alprazolam:
- combination therapy reduced anxiety more effectively than did hypnosis or alprazolam alone, as measured by the Profile of Mood States tension-anxiety scale
- improvement was comparable with hypnosis or alprazolam alone
- highly hypnotizable students showed significantly greater relaxation than did those with low hypnotizability in all three treatment groups
- EEG data showed similar frontal and occipital changes in the alprazolam and hypnotic suggestion groups.
In randomized trials, simple self-hypnosis training has reduced pain and anxiety during medical procedures, reducing procedure time by an average 17 minutes and resulting in fewer complications.11
A typical hypnotic instruction for managing anxiety is provided in Box 2. This approach teaches patients how to deal with stressors that complicate their anxiety and to control their somatic response. Hypnosis expands patients’ repertoire of responses and enables them to feel less helpless.
Confronting phobias
Phobic symptoms of fear and avoidance or exposure with distress respond especially well to brief hypnosis interventions. Although behavior modification and antidepressants also can treat phobias successfully, one or two hypnosis sessions often can reduce or cure phobic symptoms.
For example, one can help patients with airplane phobia prepare for flight by going into a hypnotic state and learning three concepts:
- Think of the airplane as an extension of the body, such as a bicycle.
- Float with the plane.
- Think about the difference between probability and possibility.
The hypnotic state—with its focused attention and physical relaxation—can amplify this cognitive restructuring technique. Phobic patients can feel more in control of their somatic reactions and, by extrapolation, the flying experience itself. In one study, 52% of patients taught this self-hypnosis exercise remained improved or cured at least 7 years later.12
Treating traumatic reactions
Evidence is growing that trauma elicits dissociation. Thus, hypnosis could help us understand and treat traumatic reactions, including patients with acute and posttraumatic stress disorder (PTSD) and dissociative disorders.
The hypnotic state’s controlled dissociation can be used to model the uncontrolled dissociation represented by posttraumatic phenomena such as flashbacks, numbing, and amnesia.13 This view is supported by evidence that PTSD is associated with high hypnotizability.14,15
Acute stress disorder—as introduced in DSM-IV16—is characterized by prominent dissociative symptoms, with intrusion, avoidance, and hyperarousal. These diagnostic criteria recognize that acute dissociation is a common and predictable reaction to trauma.
Hypnosis involving grief work, exploration of trauma-related transference issues, and emotional expression are effective psychotherapies for persons exposed to trauma. Becoming familiar with hypnotic states can teach patients to recognize, understand, and control their dissociative states.
Evidence suggests that hypnosis’ intense concentration may reverse the dissociative mind fragmentation caused by trauma.17 Traumatic memories may seem less overwhelming and intrusive once patients discover they can:
- exert greater control over memory access and retrieval
- work through and assimilate disturbing thoughts.
The controlled experience of hypnotic abreaction (reliving traumatic and other memories with strong emotion) provides boundaries for psychotherapeutic grief work.18,19 Instead of telling patients not to ruminate over a traumatic event, the clinician instructs the patient how to think about the experience.
The inferred message is that the patient can work on other things—such as relationships and daily living problems—after this therapeutic work is done.
Patients are slowly separated from the victim role. The goal is to help them restructure their memories, both cognitively and emotionally. They bear the memories’ impact, yet come to see the information differently.7 Traumatic input becomes more bearable when linked to a cognitively restructured recognition of an adaptive response.,20 For example, patients may acknowledge what they did during a traumatic event that was self-protective or helped others.
PTSD. Hypnosis shares common elements with other cognitive and behavioral treatments for PTSD, including exposure to traumatic memories for cognitive and emotional processing. Few studies have examined using hypnosis to treat PTSD, but evidence suggests it is at least as effective as other cognitive-behavioral treatments.20,21
Patients can be taught to view PTSD’s intrusive memories and bodily symptoms as re-experiencing painful memories. The memories often intrude less frequently after patients find a controlled method—such as self-hypnosis—to access and work them through.22
Ms. J hoped hypnosis could help her better visualize the face of an assailant who had attacked her as she returned at dusk from the grocery store. She had fought off his attempt to drag her into her apartment and rape her. The police showed little interest in pursuing him, however, because the sexual assault had not been completed. After the police left, she had a grand mal seizure. She had suffered a basalar skull fracture.
Ms. J was highly hypnotizable and learned the split-screen technique. While visualizing the assault on the left screen, she realized something that had not been clear to her before: “From the look on his face, I can see he wants to kill me. If he gets me into my apartment, he will kill me.”
She focused on this realization and the image of his hatred and threat to her. The therapist asked her to picture on the right screen something she had done to protect herself. She said: “He is surprised that I am fighting so hard. He doesn’t expect me to put up such a fight.”
She emerged from hypnosis understanding that she had been in more danger than she realized. Thus, despite the disappointment of having no clearer idea of what he looked like (it was quite dark when he attacked her), she had a restructured perspective about what had occurred.
Before this session, Ms. J had felt guilty that she had gotten herself so seriously injured. Afterward, she could better tolerate the memory of the attack because it was coupled with cognitive awareness that her actions may have saved her life.
Self-blame. Many trauma victims would rather feel guilty than helpless. They blame themselves inappropriately for events over which they had no control, rather than accept their helplessness. They misuse hindsight about the trauma to assume the events were predictable and therefore avoidable. They imagine they can replay the events and change the outcome.
Such an approach to trauma can be profoundly demoralizing, leaving victims burdened by needless guilt and shame. Helping them face and bear the feelings associated with traumatic events can free them from efforts to “undo” or take responsibility for the trauma and accept what happened.
Split-screen technique. Using hypnosis with a “split-screen” technique can help patients restructure the memory of trauma. The left screen symbolizes the trauma in condensed form. The right screen helps patients focus on how they tried to master the situation. This grief work allows patients to acknowledge, bear, and put into perspective the humiliation of the experience and their loss of invulnerability, health, or loved ones (Box 3).18
Dissociation. Dissociating during a threatening situation may enable a person to put aside some awareness of the danger and take self-protective action. Persistent dissociation, however, may make it too easy to avoid working through the traumatic experiences later on.22-24
Dissociation makes subsequent exposure to reminders of the trauma more similar to a reexperiencing rather than a controlled remembering of it. This can trigger physiologic stress reactions and lead to or worsen PTSD.25-27
Dissociative disorders can be understood as chronic and severe PTSDs.28 Many individuals with dissociative disorders have histories of sexual and physical abuse.29-31 Clearly, traumatic experiences sensitize survivors to subsequent trauma through conditioned activation of fear circuitry involving the amygdala, hippocampus, and frontal lobes.32
Hypnosis can be especially helpful—both for diagnosis and therapy.33 It can assist the controlled recovery of memories, while allowing some images to remain dissociated from cognition until the patient is ready to deal with them. The patient can turn memories on and off by entering and exiting the hypnotic state and thereby recover and reprocess memories at a tolerable pace.
Related resources
- Society for Clinical and Experimental Hypnosis. http://ijceh.educ.wsu.edu
- American Society of Clinical Hypnosis. www.asch.net
- American Psychological Association, Division 30. Society of Psychological Hypnosis. http://www.apa.org/about/division/div30.html
1. Spiegel D, Jasiukaitis P. Hypnosis: Brain basis. In: Smith BH (ed). Elsevier’s encyclopedia of neuroscience. The Netherlands: Elsevier Science, 1999.
2. Rainville P, Hofbauer RK, Bushnell MC, et al. Hypnosis modulates activity in brain structures involved in the regulation of consciousness. J Cogn Neurosci 2002;14:887-901.
3. Rainville P, Duncan GH, Price DD, et al. Pain affect encoded in human anterior cingulate but not somatosensory cortex. Science 1997;277:968-71.
4. Kosslyn SM, Thompson WL, Costantini-Ferrando MF, et al. Hypnotic visual illusion alters color processing in the brain. Am J Psychiatry 2000;157:1279-84.
5. Spiegel D. Negative and positive visual hypnotic hallucinations: attending inside and out. Int J Clin Exp Hypn 2003;51:130-46.
6. Spiegel D, King R. Hypnotizability and CSF HVA levels among psychiatric patients. Biol Psychiatry 1992;31:95-8.
7. Piccione C, Hilgard ER, Zimbardo PG. On the degree of stability of measured hypnotizability over a 25-year period. J Pers Soc Psychol 1989;56:289-95.
8. Spiegel H, Spiegel D. Trance and treatment: Clinical uses of hypnosis. Washington, DC: American Psychiatric Press, 2004.
9. Wertz JM, Sayette MA. Effects of smoking opportunity on attentional bias in smokers. Psychol Addict Behav 2001;15:268-71.
10. Nishith P, Barabasz A, Barabasz M, Warner D. Brief hypnosis substitutes for alprazolam use in college students: transient experiences and quantitative EEG responses. Am J Clin Hypn 1999;41:262-8.
11. Lang EV, Benotsch EG, Fick LJ, et al. Adjunctive nonpharmacological analgesia for invasive medical procedures: a randomised trial. Lancet 2000;355:1486-90.
12. Spiegel D, Frischholz EJ, Maruffi B, Spiegel H. Hypnotic responsitivity and the treatment of flying phobia. Am J Clin Hypn 1981;23:239-47.
13. Butler LD, Duran EFD, Jasiukaitis P, et al. Hypnotizability and traumatic experience: a diathesis-stress model of dissociative symptomatology. Am J Psychiatry 1996;153:42-63.
14. Spiegel D. Dissociation and hypnosis in post-traumatic stress disorder. J Trauma Stress 1988;1:17-33.
15. Stutman RK, Bliss EL. Posttraumatic stress disorder, hypnotizability, and imagery. Am J Psychiatry 1985;142:741-3.
16. Diagnostic and statistical manual of mental disorders (4th ed, text rev). Washington, DC: American Psychiatric Association, 2000.
17. Maldonado JR, Spiegel D. Trauma, dissociation and hypnotizability. In: Marmar R, Bremmer D (eds). Trauma, memory and dissociation. Washington, DC: American Psychiatric Press, 1998.
18. Lindemann E. Symptomatology and management of acute grief. Am J Psychiatry 1994;151:155-60.
19. Spiegel D. Vietnam grief work using hypnosis. Am J Clin Hypn 1981;24:33-40.
20. Foa EB, Davidson JRT, Frances A. Treatment of posttraumatic stress disorder. J Clin Psychiatry 1999;50:4-69.
21. Brom D, Kleber RJ, Defare PB. Brief psychotherapy for post-traumatic stress disorder. J Consult Clin Psychol 1989;57:607-12.
22. Spiegel D. Hypnosis and implicit memory: automatic processing of explicit content. Am J Clin Hypn 1998;40:231-40.
23. Spiegel D. Multiple personality as a post-traumatic stress disorder. Psychiatr Clin North Am 1984;7:101-10.
24. Kluft RP. Dissociation as a response to extreme trauma. In: Kluft RP (ed). Childhood antecedents of multiple personality. Washington, DC: American Psychiatric Press, 1985:66-97.
25. Marmar CR, Weiss DS, Metzler T. Peritraumatic dissociation and posttraumatic stress disorder. In: Bremner JD, Marmar C (eds). Trauma, memory, and dissociation. Washington, DC: American Psychiatric Press, 1998;229-52.
26. Birmes P. Peritraumatic dissociation, acute stress, and early posttraumatic stress disorder in victims of general crime. Can J Psychiatry 2001;46:649-51.
27. Spiegel D. Hypnosis, dissociation and trauma. In: Burrows GD, Stanley RO, Bloom PB (eds). Clinical hypnosis. New York: John Wiley & Sons, 2001;143-58.
28. Spiegel D, Cardena E. Disintegrated experience: the dissociative disorders revisited. J Abnorm Psychol 1991;100:366-78.
29. Chu JA, Dill DL. Dissociative symptoms in relation to childhood physical and sexual abuse. Am J Psychiatry 1990;147:887-92.
30. Kluft RP. Childhood antecedents of multiple personality. Washington, DC: American Psychiatric Press, 1985.
31. Spiegel D. Dissociating damage. Am J Clin Hypn 1986;29:123-31.
32. LeDoux J. Synaptic self: How our brains become who we are. New York: Viking Press, 2002.
33. Putnam FW. Using hypnosis for therapeutic abreactions. Psychiatr Med 1992;10:51-65.
1. Spiegel D, Jasiukaitis P. Hypnosis: Brain basis. In: Smith BH (ed). Elsevier’s encyclopedia of neuroscience. The Netherlands: Elsevier Science, 1999.
2. Rainville P, Hofbauer RK, Bushnell MC, et al. Hypnosis modulates activity in brain structures involved in the regulation of consciousness. J Cogn Neurosci 2002;14:887-901.
3. Rainville P, Duncan GH, Price DD, et al. Pain affect encoded in human anterior cingulate but not somatosensory cortex. Science 1997;277:968-71.
4. Kosslyn SM, Thompson WL, Costantini-Ferrando MF, et al. Hypnotic visual illusion alters color processing in the brain. Am J Psychiatry 2000;157:1279-84.
5. Spiegel D. Negative and positive visual hypnotic hallucinations: attending inside and out. Int J Clin Exp Hypn 2003;51:130-46.
6. Spiegel D, King R. Hypnotizability and CSF HVA levels among psychiatric patients. Biol Psychiatry 1992;31:95-8.
7. Piccione C, Hilgard ER, Zimbardo PG. On the degree of stability of measured hypnotizability over a 25-year period. J Pers Soc Psychol 1989;56:289-95.
8. Spiegel H, Spiegel D. Trance and treatment: Clinical uses of hypnosis. Washington, DC: American Psychiatric Press, 2004.
9. Wertz JM, Sayette MA. Effects of smoking opportunity on attentional bias in smokers. Psychol Addict Behav 2001;15:268-71.
10. Nishith P, Barabasz A, Barabasz M, Warner D. Brief hypnosis substitutes for alprazolam use in college students: transient experiences and quantitative EEG responses. Am J Clin Hypn 1999;41:262-8.
11. Lang EV, Benotsch EG, Fick LJ, et al. Adjunctive nonpharmacological analgesia for invasive medical procedures: a randomised trial. Lancet 2000;355:1486-90.
12. Spiegel D, Frischholz EJ, Maruffi B, Spiegel H. Hypnotic responsitivity and the treatment of flying phobia. Am J Clin Hypn 1981;23:239-47.
13. Butler LD, Duran EFD, Jasiukaitis P, et al. Hypnotizability and traumatic experience: a diathesis-stress model of dissociative symptomatology. Am J Psychiatry 1996;153:42-63.
14. Spiegel D. Dissociation and hypnosis in post-traumatic stress disorder. J Trauma Stress 1988;1:17-33.
15. Stutman RK, Bliss EL. Posttraumatic stress disorder, hypnotizability, and imagery. Am J Psychiatry 1985;142:741-3.
16. Diagnostic and statistical manual of mental disorders (4th ed, text rev). Washington, DC: American Psychiatric Association, 2000.
17. Maldonado JR, Spiegel D. Trauma, dissociation and hypnotizability. In: Marmar R, Bremmer D (eds). Trauma, memory and dissociation. Washington, DC: American Psychiatric Press, 1998.
18. Lindemann E. Symptomatology and management of acute grief. Am J Psychiatry 1994;151:155-60.
19. Spiegel D. Vietnam grief work using hypnosis. Am J Clin Hypn 1981;24:33-40.
20. Foa EB, Davidson JRT, Frances A. Treatment of posttraumatic stress disorder. J Clin Psychiatry 1999;50:4-69.
21. Brom D, Kleber RJ, Defare PB. Brief psychotherapy for post-traumatic stress disorder. J Consult Clin Psychol 1989;57:607-12.
22. Spiegel D. Hypnosis and implicit memory: automatic processing of explicit content. Am J Clin Hypn 1998;40:231-40.
23. Spiegel D. Multiple personality as a post-traumatic stress disorder. Psychiatr Clin North Am 1984;7:101-10.
24. Kluft RP. Dissociation as a response to extreme trauma. In: Kluft RP (ed). Childhood antecedents of multiple personality. Washington, DC: American Psychiatric Press, 1985:66-97.
25. Marmar CR, Weiss DS, Metzler T. Peritraumatic dissociation and posttraumatic stress disorder. In: Bremner JD, Marmar C (eds). Trauma, memory, and dissociation. Washington, DC: American Psychiatric Press, 1998;229-52.
26. Birmes P. Peritraumatic dissociation, acute stress, and early posttraumatic stress disorder in victims of general crime. Can J Psychiatry 2001;46:649-51.
27. Spiegel D. Hypnosis, dissociation and trauma. In: Burrows GD, Stanley RO, Bloom PB (eds). Clinical hypnosis. New York: John Wiley & Sons, 2001;143-58.
28. Spiegel D, Cardena E. Disintegrated experience: the dissociative disorders revisited. J Abnorm Psychol 1991;100:366-78.
29. Chu JA, Dill DL. Dissociative symptoms in relation to childhood physical and sexual abuse. Am J Psychiatry 1990;147:887-92.
30. Kluft RP. Childhood antecedents of multiple personality. Washington, DC: American Psychiatric Press, 1985.
31. Spiegel D. Dissociating damage. Am J Clin Hypn 1986;29:123-31.
32. LeDoux J. Synaptic self: How our brains become who we are. New York: Viking Press, 2002.
33. Putnam FW. Using hypnosis for therapeutic abreactions. Psychiatr Med 1992;10:51-65.
Brain/body connection: Treating depression in patients with cardiovascular disease
Depression can exacerbate cardiovascular disease (CVD), and CVD can exacerbate depression (Figure). Thus, effectively treating depression enhances heart disease treatment, particularly if psychiatrists and medical physicians collaborate in providing patient care.
This article describes a patient with risk factors for heart disease, illustrates the physiologic pathways that link depression and CVD, and offers clinical tips to help you improve outcomes for patients with both disorders.
Case report: Trying to ‘get going’
Mr. D, age 51, presents with vegetative symptoms and a personal and family history of CVD, depression, and substance abuse disorders. He was born in a small town in Kentucky and raised in Louisville’s poorest neighborhood. After his mother died at age 42 of “hardening of the arteries,” his father started drinking more, working less, and “never really got going again.”
Figure Neuroendocrine pathways by which depression may cause or promote CVD
Among patients with a recent myocardial infarction (MI), as many as two-thirds report depressive symptoms.1 Major depression has been reported in:
- 16% to 22% of patients hospitalized post-MI,2,3 compared with 5% in the general population and 10% in the primary care population4
- 15% of patients with unstable angina5 and 20% of patients undergoing coronary artery bypass (CABG) surgery.6
Among the annual 1.5 million Americans who have an acute MI or unstable angina, 40% develop depression immediately thereafter. These 600,000 depressed patients are three times more likely to die within 6 months, compared with post-MI patients who are not depressed.7
Mr. D worked 20 years as a construction contractor, often running several work crews at once. At age 41, he slid into a depressive episode after his second divorce. He struggled with low energy, disturbed sleep, hopelessness, and increased smoking and drinking for 1 year, but he did not seek help.
Two years later, he suffered an inferior wall transmural myocardial infarction. His CVD risk factors included family history of early heart disease, smoking for 32 years, and elevated low-density lipoprotein (LDL) cholesterol. After subsequent episodes of unstable angina, stents were placed in two coronary arteries. Though his cardiologist cleared him to return to work, he felt able to work only part-time and erratically.
During a visit to their family doctor several years later, Mr. D’s wife suggested that her husband might be depressed. Reluctantly, Mr. D consulted a psychiatrist.
The psychiatrist diagnosed major depressive disorder and prescribed sertraline, 50 mg/d. Within 2 months, Mr. D’s symptoms had dropped by 50% on a symptom severity measure. He did not refill his prescription, however, because of concerns about sexual side effects. Two months later he was hospitalized for another episode of unstable angina. His depression had returned within 1 month of stopping sertraline.
The psychiatrist switched him to citalopram, 20 mg/d, and carefully monitored depressive symptoms, side effects, and medication adherence. Aside from talking with the psychiatrist for a half-hour in his family doctor’s office every few weeks, Mr. D refused to undergo psychotherapy. He eventually achieved depression remission with a combination of citalopram, 20 mg/d, and nefazodone, 200 mg/d.
Depression-CVD connection
As in Mr. D’s case, depression and CVD commonly occur together, often with serious consequences (Box). 1-7 The association between depression and CVD is not limited to depression’s effect on existing disease, however. Depression often precedes coronary disease by about 30 years—suggesting possible cause and effect. Two systematic reviews8,9 found that depression increased CVD risk by 64%.
Seven well-controlled studies5-7,10-13 compared the relative effect of depression on the cardiovascular system with that of established CVD predictors. All seven found depression’s independent effect to be significant and comparable to or greater than that of ejection fraction, previous MI history, or number of vessels with >50% narrowing.
Comorbid depression and CVD usually persists months or years,14 and most studies indicate a dose-response relationship; the more severe the depression, the greater the risk for CVD to develop or progress.8,15
The link between depression treatment and CVD risk has not been well-studied. The only randomized, controlled trial found that cognitive therapy for depression did not significantly reduce cardiac events among patients with known CVD.16
Possible mechanisms
Depression’s effect on CVD. How does depression affect CVD development and progression? Both behavioral and biological pathways may be involved.17 The behavioral pathway proposes that depression triggers behaviors—such as smoking, overeating, and sedentary lifestyles—that increase the risk of developing or worsening CVD. The biological pathway proposes that neuroendocrine changes during depression accelerate CVD development.
About one-half of persons with major depression exhibit hypothalamic-pituitary-adrenal (HPA) axis dysregulation, with excessive secretion of corticotropin releasing factor (CRF) and chronically elevated cortisol.18 This HPA dysregulation is related to defective negative feedback at the paraventricular nucleus of the hypothalamus. Chronic HPA axis dysregulation promotes vascular inflammation, and several studies have reported C-reactive protein elevation and cytokine changes in patients with major depression.19,20
Major depression is also associated with excessive sympathetic and diminished parasym-pathetic nervous system activity, potentially contributing to hypertension, increased resting heart rate, decreased heart rate variability, and altered endothelial function.2,21,22 Each of these factors facilitates arterial plaque formation.
Depression may also exacerbate chronic anxiety and other forms of distress. The combined effects of an overtaxed central nervous system, neuroendocrine dysregulation, and unhealthy behaviors may eventually overwhelm the cardiovascular system.
CVD’s effect on depression. How does CVD contribute to depression? The vascular depression hypothesis23 proposes that diffuse heart and brain atherosclerosis restricts perfusion of limbic and cortical structures that regulate mood. A first depressive episode after acute MI or CABG probably represents exacerbation of cerebrovascular insufficiency that preceded the coronary event.
Table
Four keys to effectively treat depression in patients with heart disease
|
In practical terms, this means that pathways linking depression and heart disease include not only biological factors but also:
- psychological factors such as depression, anxiety, and chronic stress
- behavioral factors such as smoking, physical inactivity, and high-fat diet.
How to improve outcomes
Patients with CVD commonly do not receive effective depression treatment:
- Internists and family physicians give preferential attention to physical illness.
- Patients may have insufficient access to mental health specialists.
- Physicians do not adequately monitor depression treatment.
- Patients are reluctant to accept the stigma of mental illness.
By collaborating with primary care physicians, you can improve the likelihood that depression treatment will achieve remission and prevent relapse (Table).
Risk factors for CVD. Depression contributes to heart disease by exacerbating four major CVD risk factors—smoking, diabetes, obesity, and physical inactivity. By effectively treating depression, you may help patients avoid common depressive symptoms—such as overeating and sedentary behaviors—that are related to low energy or fatigue.
Educate middle-aged patients with depression about CVD’s associated risk. Prochaska’s “stages of change” (see Related resources) can help them stop smoking, lose weight, and exercise.
Access to cardiac care. Depressed patients may be less motivated than nondepressed patients to pursue cardiac care.24 Therefore, you may need to:
- encourage your patients to take advantage of indicated state-of-the-art care, including stents, bypass surgery, and medications
- understand patients’ complex cardiac regimens and help them adhere when depression interferes with their motivation.
Effective depression treatment
Patient history. For depressed patients older than 40, take a careful inventory of CVD risk factors:
- family history of heart disease before age 60 for men and age 70 for women
- personal history of smoking, blood pressure >140/90 mm Hg, LDL cholesterol >100 mg/dL, type 2 diabetes, body mass index >30, or physical inactivity (<30 minutes of walking 3 days a week).
In general, the more risk factors, the greater the risk of CVD.
Antidepressant selection. Selective serotonin reuptake inhibitors (SSRIs) are safe and effective for treating major depression in CVD and congestive heart failure.25 Venlafaxine at doses >300 mg/d may increase blood pressure, so use this drug with caution in depressed patients with hypertension.
No controlled clinical trials have gauged the safety and efficacy of bupropion or mirtazapine in patients with CVD.
Tricyclic antidepressants are contraindicated for 6 months post-MI because they may contribute to arrhythmias. Avoid using them in depressed patients with CVD or conduction defects because of their quinidine-like effects on conduction.
Cardiac medications. Contrary to folk wisdom, beta blockers do not cause depression.26 Whether or not a patient is depressed, our primary care and cardiology colleagues can use beta blockers to help regulate the peripheral autonomic nervous system, reducing high blood pressure and the risk of arrhythmias.
SSRIs may increase blood levels of beta blockers, warfarin, and other cardiac medications via cytochrome P-450 isoenzyme inhibition. Make sure warfarin levels and other cardiac drug effects are well monitored when you adjust psychotropic dosages.
Divalproex and SSRIs also may reduce platelet aggregation. Patients who are receiving concomitant aspirin or warfarin may bruise or bleed easily and require dosage reductions or medication changes.
Psychotherapy. All patients with major or minor depression and CVD are considered high-risk and are candidates for a trial of brief psychotherapy. Therapeutic goals are to achieve full remission of depressive symptoms as rapidly as possible, prevent relapse, and maximize adherence to cardiac and depression drug regimens.
Collaborate closely with the cardiologist or primary care physician during the patient’s depressive episode and occasionally during maintenance treatment. Discuss or share notes on the patient’s depressive and cardiac disorders, medication management, symptom monitoring, and behavior changes needed to reduce cardiac risk.
With your added support, patients with depression and CVD are more likely to adhere to antidepressant medications and achieve symptom remission.
- National Institute of Mental Health. Depression and heart disease. www.nimh.nih.gov/publicat/depheart.cfm.
- Dewan NA, Suresh DP, Blomkalns A. Selecting safe psychotropics for post-MI patients. Current Psychiatry. 2003;2(3):15-21.
- Prochaska JO, Norcross JC, DiClemente CC. Changing for good. New York: Avon, 1994.
Drug brand names
- Bupropion • Wellbutrin
- Citalopram • Celexa
- Escitalopram • Lexapro
- Fluoxetine • Prozac
- Fluvoxamine • Luvox
- Paroxetine • Paxil
- Mirtazapine • Remeron
- Nefazodone • Serzone
- Sertraline • Zoloft
- Venlafaxine • Effexor
Disclosure
Dr. Wulsin is a consultant to Pfizer Inc. and Janssen Pharmaceutica.
Dr. Vieweg is a speaker for Janssen Pharmaceutica, Eli Lilly and Co., Pfizer Inc., Wyeth Pharmaceuticals, Forest Pharmaceuticals, and GlaxoSmithKline.
Dr. Fernandez reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Cassem N, Hackett T. Psychiatric condition in a coronary care unit. Ann Intern Med 1971;75:9-14.
2. Glassman A, Shapiro P. Depression and the course of coronary artery disease. Am J Psychiatry 1998;155:4-11.
3. Carney R, Freedland K, Sheline Y, Weiss E. Depression and coronary heart disease: a review for cardiologists. Clin Cardiol 1997;20:196-200.
4. Katon W, Schulbert H. Epidemiology of depression in primary care. Gen Hosp Psychiatry 1992;14:237-47.
5. Lesperance F, Frasure-Smith N, Theroux P. Depression and 1-year prognosis in unstable angina. Arch Intern Med 2000;160:1354-60.
6. Connerney I, Shapiro P, McLaughlin J, et al. Relation between depression after coronary artery bypass surgery and 12-month outcome: a prospective study. Lancet 2001;358:1766-71.
7. Frasure-Smith N, Lesperance F, Talajic M. Depression and 18-month prognosis after myocardial infarction. Circulation 1995;91:999-1005.
8. Rugulies R. Depression as a predictor for coronary heart disease. Am J Prev Med 2002;23:51-61.
9. Wulsin L, Singal B. Do depressive symptoms increase the risk for the onset of coronary disease? A systematic quantitative review. Psychosom Med 2003;65:201-10.
10. Carney R, Rich M, Freedland K, et al. Major depressive disorder predicts cardiac events in patients with coronary artery disease. Psychosom Med 1988;50:627-33.
11. Ladwig K, Roll G, Breithardt G, Borggrefe M. Extracardiac contributions to chest pain perception in patients 6 months after acute myocardial infarction. Am Heart J 1999;137:528-34.
12. Levine J, Covino N, Slack W, et al. Psychological predictors of subsequent medical care among patients hospitalized with cardiac disease. J Cardiopulm Rehabil 1996;16:109-16.
13. Lesperance F, Frasure-Smith N, Talajic M, Bourassa M. Five-year risk of cardiac mortality in relation to initial severity and one-year changes in depression symptoms after myocardial infarction. Circulation 2002;105:1049-53.
14. Dwight M, Stoudemire A. Effects of depressive disorders on coronary artery disease: a review. Harv Rev Psychiatry 1997;5:115-122.
15. Penninx B, Beekman A, Honig A, et al. Depression and cardiac mortality. Arch Gen Psychiatry 2001;58:221-7.
16. Writing committee of the ENRICHD investigators. Effects of treating depression and low perceived social support on clinical events after myocardial infarction. JAMA 2003;289:3106-16.
17. Carney RM, Freedland K, Miller G, Jaffe AS. Depression as a risk factor for cardiac mortality and morbidity: a review of potential mechanisms. J Psychosom Res 2002;53:897-902.
18. Musselman D, Evans D, Nemeroff C. The relationship of depression to cardiovascular disease. Arch Gen Psychiatry 1998;55:580-92.
19. Kop WJ. Chronic and acute psychological risk factors for clinical manifestations of coronary artery disease. Psychosom Med 1999;61:476-86.
20. Miller G, Cohen S, Herbert T. Pathways linking major depression and immunity in ambulatory female patients. Psychosom Med 1999;61:850-60.
21. Carney R, Freedland K, Stein P. Change in heart rate and heart rate variability during treatment for depression in patients with coronary heart disease. Psychosom Med 2000;62:639-47.
22. Carney R, Freedland K, Miller G, Jaffe A. Depression as a risk factor for cardiac mortality and morbidity: a review of potential mechanisms. J Psychosom Res 2002;53:897-902.
23. Alexopoulos G, Meyers B, Young R, et al. Vascular depression hypothesis. Psychosom Med 1997;58:113-121.
24. Ziegelstein R, Fauerbach J, Stevens S, et al. Patients with depression are less likely to follow recommendations to reduce cardiac risk during recovery from a myocardial infarction. Arch Intern Med 2000;160:1818-23.
25. Glassman AH, O’Connor C, Califf RM, et al. Sertraline treatment of major depression in patients with acute MI or unstable angina. JAMA 2002;288:701-9.
26. Ko D, Hebert P, Coffey C, et al. B-blocker therapy and symptoms of depression, fatigue, and sexual dysfunction. JAMA 2002;288:351-7.
Depression can exacerbate cardiovascular disease (CVD), and CVD can exacerbate depression (Figure). Thus, effectively treating depression enhances heart disease treatment, particularly if psychiatrists and medical physicians collaborate in providing patient care.
This article describes a patient with risk factors for heart disease, illustrates the physiologic pathways that link depression and CVD, and offers clinical tips to help you improve outcomes for patients with both disorders.
Case report: Trying to ‘get going’
Mr. D, age 51, presents with vegetative symptoms and a personal and family history of CVD, depression, and substance abuse disorders. He was born in a small town in Kentucky and raised in Louisville’s poorest neighborhood. After his mother died at age 42 of “hardening of the arteries,” his father started drinking more, working less, and “never really got going again.”
Figure Neuroendocrine pathways by which depression may cause or promote CVD
Among patients with a recent myocardial infarction (MI), as many as two-thirds report depressive symptoms.1 Major depression has been reported in:
- 16% to 22% of patients hospitalized post-MI,2,3 compared with 5% in the general population and 10% in the primary care population4
- 15% of patients with unstable angina5 and 20% of patients undergoing coronary artery bypass (CABG) surgery.6
Among the annual 1.5 million Americans who have an acute MI or unstable angina, 40% develop depression immediately thereafter. These 600,000 depressed patients are three times more likely to die within 6 months, compared with post-MI patients who are not depressed.7
Mr. D worked 20 years as a construction contractor, often running several work crews at once. At age 41, he slid into a depressive episode after his second divorce. He struggled with low energy, disturbed sleep, hopelessness, and increased smoking and drinking for 1 year, but he did not seek help.
Two years later, he suffered an inferior wall transmural myocardial infarction. His CVD risk factors included family history of early heart disease, smoking for 32 years, and elevated low-density lipoprotein (LDL) cholesterol. After subsequent episodes of unstable angina, stents were placed in two coronary arteries. Though his cardiologist cleared him to return to work, he felt able to work only part-time and erratically.
During a visit to their family doctor several years later, Mr. D’s wife suggested that her husband might be depressed. Reluctantly, Mr. D consulted a psychiatrist.
The psychiatrist diagnosed major depressive disorder and prescribed sertraline, 50 mg/d. Within 2 months, Mr. D’s symptoms had dropped by 50% on a symptom severity measure. He did not refill his prescription, however, because of concerns about sexual side effects. Two months later he was hospitalized for another episode of unstable angina. His depression had returned within 1 month of stopping sertraline.
The psychiatrist switched him to citalopram, 20 mg/d, and carefully monitored depressive symptoms, side effects, and medication adherence. Aside from talking with the psychiatrist for a half-hour in his family doctor’s office every few weeks, Mr. D refused to undergo psychotherapy. He eventually achieved depression remission with a combination of citalopram, 20 mg/d, and nefazodone, 200 mg/d.
Depression-CVD connection
As in Mr. D’s case, depression and CVD commonly occur together, often with serious consequences (Box). 1-7 The association between depression and CVD is not limited to depression’s effect on existing disease, however. Depression often precedes coronary disease by about 30 years—suggesting possible cause and effect. Two systematic reviews8,9 found that depression increased CVD risk by 64%.
Seven well-controlled studies5-7,10-13 compared the relative effect of depression on the cardiovascular system with that of established CVD predictors. All seven found depression’s independent effect to be significant and comparable to or greater than that of ejection fraction, previous MI history, or number of vessels with >50% narrowing.
Comorbid depression and CVD usually persists months or years,14 and most studies indicate a dose-response relationship; the more severe the depression, the greater the risk for CVD to develop or progress.8,15
The link between depression treatment and CVD risk has not been well-studied. The only randomized, controlled trial found that cognitive therapy for depression did not significantly reduce cardiac events among patients with known CVD.16
Possible mechanisms
Depression’s effect on CVD. How does depression affect CVD development and progression? Both behavioral and biological pathways may be involved.17 The behavioral pathway proposes that depression triggers behaviors—such as smoking, overeating, and sedentary lifestyles—that increase the risk of developing or worsening CVD. The biological pathway proposes that neuroendocrine changes during depression accelerate CVD development.
About one-half of persons with major depression exhibit hypothalamic-pituitary-adrenal (HPA) axis dysregulation, with excessive secretion of corticotropin releasing factor (CRF) and chronically elevated cortisol.18 This HPA dysregulation is related to defective negative feedback at the paraventricular nucleus of the hypothalamus. Chronic HPA axis dysregulation promotes vascular inflammation, and several studies have reported C-reactive protein elevation and cytokine changes in patients with major depression.19,20
Major depression is also associated with excessive sympathetic and diminished parasym-pathetic nervous system activity, potentially contributing to hypertension, increased resting heart rate, decreased heart rate variability, and altered endothelial function.2,21,22 Each of these factors facilitates arterial plaque formation.
Depression may also exacerbate chronic anxiety and other forms of distress. The combined effects of an overtaxed central nervous system, neuroendocrine dysregulation, and unhealthy behaviors may eventually overwhelm the cardiovascular system.
CVD’s effect on depression. How does CVD contribute to depression? The vascular depression hypothesis23 proposes that diffuse heart and brain atherosclerosis restricts perfusion of limbic and cortical structures that regulate mood. A first depressive episode after acute MI or CABG probably represents exacerbation of cerebrovascular insufficiency that preceded the coronary event.
Table
Four keys to effectively treat depression in patients with heart disease
|
In practical terms, this means that pathways linking depression and heart disease include not only biological factors but also:
- psychological factors such as depression, anxiety, and chronic stress
- behavioral factors such as smoking, physical inactivity, and high-fat diet.
How to improve outcomes
Patients with CVD commonly do not receive effective depression treatment:
- Internists and family physicians give preferential attention to physical illness.
- Patients may have insufficient access to mental health specialists.
- Physicians do not adequately monitor depression treatment.
- Patients are reluctant to accept the stigma of mental illness.
By collaborating with primary care physicians, you can improve the likelihood that depression treatment will achieve remission and prevent relapse (Table).
Risk factors for CVD. Depression contributes to heart disease by exacerbating four major CVD risk factors—smoking, diabetes, obesity, and physical inactivity. By effectively treating depression, you may help patients avoid common depressive symptoms—such as overeating and sedentary behaviors—that are related to low energy or fatigue.
Educate middle-aged patients with depression about CVD’s associated risk. Prochaska’s “stages of change” (see Related resources) can help them stop smoking, lose weight, and exercise.
Access to cardiac care. Depressed patients may be less motivated than nondepressed patients to pursue cardiac care.24 Therefore, you may need to:
- encourage your patients to take advantage of indicated state-of-the-art care, including stents, bypass surgery, and medications
- understand patients’ complex cardiac regimens and help them adhere when depression interferes with their motivation.
Effective depression treatment
Patient history. For depressed patients older than 40, take a careful inventory of CVD risk factors:
- family history of heart disease before age 60 for men and age 70 for women
- personal history of smoking, blood pressure >140/90 mm Hg, LDL cholesterol >100 mg/dL, type 2 diabetes, body mass index >30, or physical inactivity (<30 minutes of walking 3 days a week).
In general, the more risk factors, the greater the risk of CVD.
Antidepressant selection. Selective serotonin reuptake inhibitors (SSRIs) are safe and effective for treating major depression in CVD and congestive heart failure.25 Venlafaxine at doses >300 mg/d may increase blood pressure, so use this drug with caution in depressed patients with hypertension.
No controlled clinical trials have gauged the safety and efficacy of bupropion or mirtazapine in patients with CVD.
Tricyclic antidepressants are contraindicated for 6 months post-MI because they may contribute to arrhythmias. Avoid using them in depressed patients with CVD or conduction defects because of their quinidine-like effects on conduction.
Cardiac medications. Contrary to folk wisdom, beta blockers do not cause depression.26 Whether or not a patient is depressed, our primary care and cardiology colleagues can use beta blockers to help regulate the peripheral autonomic nervous system, reducing high blood pressure and the risk of arrhythmias.
SSRIs may increase blood levels of beta blockers, warfarin, and other cardiac medications via cytochrome P-450 isoenzyme inhibition. Make sure warfarin levels and other cardiac drug effects are well monitored when you adjust psychotropic dosages.
Divalproex and SSRIs also may reduce platelet aggregation. Patients who are receiving concomitant aspirin or warfarin may bruise or bleed easily and require dosage reductions or medication changes.
Psychotherapy. All patients with major or minor depression and CVD are considered high-risk and are candidates for a trial of brief psychotherapy. Therapeutic goals are to achieve full remission of depressive symptoms as rapidly as possible, prevent relapse, and maximize adherence to cardiac and depression drug regimens.
Collaborate closely with the cardiologist or primary care physician during the patient’s depressive episode and occasionally during maintenance treatment. Discuss or share notes on the patient’s depressive and cardiac disorders, medication management, symptom monitoring, and behavior changes needed to reduce cardiac risk.
With your added support, patients with depression and CVD are more likely to adhere to antidepressant medications and achieve symptom remission.
- National Institute of Mental Health. Depression and heart disease. www.nimh.nih.gov/publicat/depheart.cfm.
- Dewan NA, Suresh DP, Blomkalns A. Selecting safe psychotropics for post-MI patients. Current Psychiatry. 2003;2(3):15-21.
- Prochaska JO, Norcross JC, DiClemente CC. Changing for good. New York: Avon, 1994.
Drug brand names
- Bupropion • Wellbutrin
- Citalopram • Celexa
- Escitalopram • Lexapro
- Fluoxetine • Prozac
- Fluvoxamine • Luvox
- Paroxetine • Paxil
- Mirtazapine • Remeron
- Nefazodone • Serzone
- Sertraline • Zoloft
- Venlafaxine • Effexor
Disclosure
Dr. Wulsin is a consultant to Pfizer Inc. and Janssen Pharmaceutica.
Dr. Vieweg is a speaker for Janssen Pharmaceutica, Eli Lilly and Co., Pfizer Inc., Wyeth Pharmaceuticals, Forest Pharmaceuticals, and GlaxoSmithKline.
Dr. Fernandez reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Depression can exacerbate cardiovascular disease (CVD), and CVD can exacerbate depression (Figure). Thus, effectively treating depression enhances heart disease treatment, particularly if psychiatrists and medical physicians collaborate in providing patient care.
This article describes a patient with risk factors for heart disease, illustrates the physiologic pathways that link depression and CVD, and offers clinical tips to help you improve outcomes for patients with both disorders.
Case report: Trying to ‘get going’
Mr. D, age 51, presents with vegetative symptoms and a personal and family history of CVD, depression, and substance abuse disorders. He was born in a small town in Kentucky and raised in Louisville’s poorest neighborhood. After his mother died at age 42 of “hardening of the arteries,” his father started drinking more, working less, and “never really got going again.”
Figure Neuroendocrine pathways by which depression may cause or promote CVD
Among patients with a recent myocardial infarction (MI), as many as two-thirds report depressive symptoms.1 Major depression has been reported in:
- 16% to 22% of patients hospitalized post-MI,2,3 compared with 5% in the general population and 10% in the primary care population4
- 15% of patients with unstable angina5 and 20% of patients undergoing coronary artery bypass (CABG) surgery.6
Among the annual 1.5 million Americans who have an acute MI or unstable angina, 40% develop depression immediately thereafter. These 600,000 depressed patients are three times more likely to die within 6 months, compared with post-MI patients who are not depressed.7
Mr. D worked 20 years as a construction contractor, often running several work crews at once. At age 41, he slid into a depressive episode after his second divorce. He struggled with low energy, disturbed sleep, hopelessness, and increased smoking and drinking for 1 year, but he did not seek help.
Two years later, he suffered an inferior wall transmural myocardial infarction. His CVD risk factors included family history of early heart disease, smoking for 32 years, and elevated low-density lipoprotein (LDL) cholesterol. After subsequent episodes of unstable angina, stents were placed in two coronary arteries. Though his cardiologist cleared him to return to work, he felt able to work only part-time and erratically.
During a visit to their family doctor several years later, Mr. D’s wife suggested that her husband might be depressed. Reluctantly, Mr. D consulted a psychiatrist.
The psychiatrist diagnosed major depressive disorder and prescribed sertraline, 50 mg/d. Within 2 months, Mr. D’s symptoms had dropped by 50% on a symptom severity measure. He did not refill his prescription, however, because of concerns about sexual side effects. Two months later he was hospitalized for another episode of unstable angina. His depression had returned within 1 month of stopping sertraline.
The psychiatrist switched him to citalopram, 20 mg/d, and carefully monitored depressive symptoms, side effects, and medication adherence. Aside from talking with the psychiatrist for a half-hour in his family doctor’s office every few weeks, Mr. D refused to undergo psychotherapy. He eventually achieved depression remission with a combination of citalopram, 20 mg/d, and nefazodone, 200 mg/d.
Depression-CVD connection
As in Mr. D’s case, depression and CVD commonly occur together, often with serious consequences (Box). 1-7 The association between depression and CVD is not limited to depression’s effect on existing disease, however. Depression often precedes coronary disease by about 30 years—suggesting possible cause and effect. Two systematic reviews8,9 found that depression increased CVD risk by 64%.
Seven well-controlled studies5-7,10-13 compared the relative effect of depression on the cardiovascular system with that of established CVD predictors. All seven found depression’s independent effect to be significant and comparable to or greater than that of ejection fraction, previous MI history, or number of vessels with >50% narrowing.
Comorbid depression and CVD usually persists months or years,14 and most studies indicate a dose-response relationship; the more severe the depression, the greater the risk for CVD to develop or progress.8,15
The link between depression treatment and CVD risk has not been well-studied. The only randomized, controlled trial found that cognitive therapy for depression did not significantly reduce cardiac events among patients with known CVD.16
Possible mechanisms
Depression’s effect on CVD. How does depression affect CVD development and progression? Both behavioral and biological pathways may be involved.17 The behavioral pathway proposes that depression triggers behaviors—such as smoking, overeating, and sedentary lifestyles—that increase the risk of developing or worsening CVD. The biological pathway proposes that neuroendocrine changes during depression accelerate CVD development.
About one-half of persons with major depression exhibit hypothalamic-pituitary-adrenal (HPA) axis dysregulation, with excessive secretion of corticotropin releasing factor (CRF) and chronically elevated cortisol.18 This HPA dysregulation is related to defective negative feedback at the paraventricular nucleus of the hypothalamus. Chronic HPA axis dysregulation promotes vascular inflammation, and several studies have reported C-reactive protein elevation and cytokine changes in patients with major depression.19,20
Major depression is also associated with excessive sympathetic and diminished parasym-pathetic nervous system activity, potentially contributing to hypertension, increased resting heart rate, decreased heart rate variability, and altered endothelial function.2,21,22 Each of these factors facilitates arterial plaque formation.
Depression may also exacerbate chronic anxiety and other forms of distress. The combined effects of an overtaxed central nervous system, neuroendocrine dysregulation, and unhealthy behaviors may eventually overwhelm the cardiovascular system.
CVD’s effect on depression. How does CVD contribute to depression? The vascular depression hypothesis23 proposes that diffuse heart and brain atherosclerosis restricts perfusion of limbic and cortical structures that regulate mood. A first depressive episode after acute MI or CABG probably represents exacerbation of cerebrovascular insufficiency that preceded the coronary event.
Table
Four keys to effectively treat depression in patients with heart disease
|
In practical terms, this means that pathways linking depression and heart disease include not only biological factors but also:
- psychological factors such as depression, anxiety, and chronic stress
- behavioral factors such as smoking, physical inactivity, and high-fat diet.
How to improve outcomes
Patients with CVD commonly do not receive effective depression treatment:
- Internists and family physicians give preferential attention to physical illness.
- Patients may have insufficient access to mental health specialists.
- Physicians do not adequately monitor depression treatment.
- Patients are reluctant to accept the stigma of mental illness.
By collaborating with primary care physicians, you can improve the likelihood that depression treatment will achieve remission and prevent relapse (Table).
Risk factors for CVD. Depression contributes to heart disease by exacerbating four major CVD risk factors—smoking, diabetes, obesity, and physical inactivity. By effectively treating depression, you may help patients avoid common depressive symptoms—such as overeating and sedentary behaviors—that are related to low energy or fatigue.
Educate middle-aged patients with depression about CVD’s associated risk. Prochaska’s “stages of change” (see Related resources) can help them stop smoking, lose weight, and exercise.
Access to cardiac care. Depressed patients may be less motivated than nondepressed patients to pursue cardiac care.24 Therefore, you may need to:
- encourage your patients to take advantage of indicated state-of-the-art care, including stents, bypass surgery, and medications
- understand patients’ complex cardiac regimens and help them adhere when depression interferes with their motivation.
Effective depression treatment
Patient history. For depressed patients older than 40, take a careful inventory of CVD risk factors:
- family history of heart disease before age 60 for men and age 70 for women
- personal history of smoking, blood pressure >140/90 mm Hg, LDL cholesterol >100 mg/dL, type 2 diabetes, body mass index >30, or physical inactivity (<30 minutes of walking 3 days a week).
In general, the more risk factors, the greater the risk of CVD.
Antidepressant selection. Selective serotonin reuptake inhibitors (SSRIs) are safe and effective for treating major depression in CVD and congestive heart failure.25 Venlafaxine at doses >300 mg/d may increase blood pressure, so use this drug with caution in depressed patients with hypertension.
No controlled clinical trials have gauged the safety and efficacy of bupropion or mirtazapine in patients with CVD.
Tricyclic antidepressants are contraindicated for 6 months post-MI because they may contribute to arrhythmias. Avoid using them in depressed patients with CVD or conduction defects because of their quinidine-like effects on conduction.
Cardiac medications. Contrary to folk wisdom, beta blockers do not cause depression.26 Whether or not a patient is depressed, our primary care and cardiology colleagues can use beta blockers to help regulate the peripheral autonomic nervous system, reducing high blood pressure and the risk of arrhythmias.
SSRIs may increase blood levels of beta blockers, warfarin, and other cardiac medications via cytochrome P-450 isoenzyme inhibition. Make sure warfarin levels and other cardiac drug effects are well monitored when you adjust psychotropic dosages.
Divalproex and SSRIs also may reduce platelet aggregation. Patients who are receiving concomitant aspirin or warfarin may bruise or bleed easily and require dosage reductions or medication changes.
Psychotherapy. All patients with major or minor depression and CVD are considered high-risk and are candidates for a trial of brief psychotherapy. Therapeutic goals are to achieve full remission of depressive symptoms as rapidly as possible, prevent relapse, and maximize adherence to cardiac and depression drug regimens.
Collaborate closely with the cardiologist or primary care physician during the patient’s depressive episode and occasionally during maintenance treatment. Discuss or share notes on the patient’s depressive and cardiac disorders, medication management, symptom monitoring, and behavior changes needed to reduce cardiac risk.
With your added support, patients with depression and CVD are more likely to adhere to antidepressant medications and achieve symptom remission.
- National Institute of Mental Health. Depression and heart disease. www.nimh.nih.gov/publicat/depheart.cfm.
- Dewan NA, Suresh DP, Blomkalns A. Selecting safe psychotropics for post-MI patients. Current Psychiatry. 2003;2(3):15-21.
- Prochaska JO, Norcross JC, DiClemente CC. Changing for good. New York: Avon, 1994.
Drug brand names
- Bupropion • Wellbutrin
- Citalopram • Celexa
- Escitalopram • Lexapro
- Fluoxetine • Prozac
- Fluvoxamine • Luvox
- Paroxetine • Paxil
- Mirtazapine • Remeron
- Nefazodone • Serzone
- Sertraline • Zoloft
- Venlafaxine • Effexor
Disclosure
Dr. Wulsin is a consultant to Pfizer Inc. and Janssen Pharmaceutica.
Dr. Vieweg is a speaker for Janssen Pharmaceutica, Eli Lilly and Co., Pfizer Inc., Wyeth Pharmaceuticals, Forest Pharmaceuticals, and GlaxoSmithKline.
Dr. Fernandez reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Cassem N, Hackett T. Psychiatric condition in a coronary care unit. Ann Intern Med 1971;75:9-14.
2. Glassman A, Shapiro P. Depression and the course of coronary artery disease. Am J Psychiatry 1998;155:4-11.
3. Carney R, Freedland K, Sheline Y, Weiss E. Depression and coronary heart disease: a review for cardiologists. Clin Cardiol 1997;20:196-200.
4. Katon W, Schulbert H. Epidemiology of depression in primary care. Gen Hosp Psychiatry 1992;14:237-47.
5. Lesperance F, Frasure-Smith N, Theroux P. Depression and 1-year prognosis in unstable angina. Arch Intern Med 2000;160:1354-60.
6. Connerney I, Shapiro P, McLaughlin J, et al. Relation between depression after coronary artery bypass surgery and 12-month outcome: a prospective study. Lancet 2001;358:1766-71.
7. Frasure-Smith N, Lesperance F, Talajic M. Depression and 18-month prognosis after myocardial infarction. Circulation 1995;91:999-1005.
8. Rugulies R. Depression as a predictor for coronary heart disease. Am J Prev Med 2002;23:51-61.
9. Wulsin L, Singal B. Do depressive symptoms increase the risk for the onset of coronary disease? A systematic quantitative review. Psychosom Med 2003;65:201-10.
10. Carney R, Rich M, Freedland K, et al. Major depressive disorder predicts cardiac events in patients with coronary artery disease. Psychosom Med 1988;50:627-33.
11. Ladwig K, Roll G, Breithardt G, Borggrefe M. Extracardiac contributions to chest pain perception in patients 6 months after acute myocardial infarction. Am Heart J 1999;137:528-34.
12. Levine J, Covino N, Slack W, et al. Psychological predictors of subsequent medical care among patients hospitalized with cardiac disease. J Cardiopulm Rehabil 1996;16:109-16.
13. Lesperance F, Frasure-Smith N, Talajic M, Bourassa M. Five-year risk of cardiac mortality in relation to initial severity and one-year changes in depression symptoms after myocardial infarction. Circulation 2002;105:1049-53.
14. Dwight M, Stoudemire A. Effects of depressive disorders on coronary artery disease: a review. Harv Rev Psychiatry 1997;5:115-122.
15. Penninx B, Beekman A, Honig A, et al. Depression and cardiac mortality. Arch Gen Psychiatry 2001;58:221-7.
16. Writing committee of the ENRICHD investigators. Effects of treating depression and low perceived social support on clinical events after myocardial infarction. JAMA 2003;289:3106-16.
17. Carney RM, Freedland K, Miller G, Jaffe AS. Depression as a risk factor for cardiac mortality and morbidity: a review of potential mechanisms. J Psychosom Res 2002;53:897-902.
18. Musselman D, Evans D, Nemeroff C. The relationship of depression to cardiovascular disease. Arch Gen Psychiatry 1998;55:580-92.
19. Kop WJ. Chronic and acute psychological risk factors for clinical manifestations of coronary artery disease. Psychosom Med 1999;61:476-86.
20. Miller G, Cohen S, Herbert T. Pathways linking major depression and immunity in ambulatory female patients. Psychosom Med 1999;61:850-60.
21. Carney R, Freedland K, Stein P. Change in heart rate and heart rate variability during treatment for depression in patients with coronary heart disease. Psychosom Med 2000;62:639-47.
22. Carney R, Freedland K, Miller G, Jaffe A. Depression as a risk factor for cardiac mortality and morbidity: a review of potential mechanisms. J Psychosom Res 2002;53:897-902.
23. Alexopoulos G, Meyers B, Young R, et al. Vascular depression hypothesis. Psychosom Med 1997;58:113-121.
24. Ziegelstein R, Fauerbach J, Stevens S, et al. Patients with depression are less likely to follow recommendations to reduce cardiac risk during recovery from a myocardial infarction. Arch Intern Med 2000;160:1818-23.
25. Glassman AH, O’Connor C, Califf RM, et al. Sertraline treatment of major depression in patients with acute MI or unstable angina. JAMA 2002;288:701-9.
26. Ko D, Hebert P, Coffey C, et al. B-blocker therapy and symptoms of depression, fatigue, and sexual dysfunction. JAMA 2002;288:351-7.
1. Cassem N, Hackett T. Psychiatric condition in a coronary care unit. Ann Intern Med 1971;75:9-14.
2. Glassman A, Shapiro P. Depression and the course of coronary artery disease. Am J Psychiatry 1998;155:4-11.
3. Carney R, Freedland K, Sheline Y, Weiss E. Depression and coronary heart disease: a review for cardiologists. Clin Cardiol 1997;20:196-200.
4. Katon W, Schulbert H. Epidemiology of depression in primary care. Gen Hosp Psychiatry 1992;14:237-47.
5. Lesperance F, Frasure-Smith N, Theroux P. Depression and 1-year prognosis in unstable angina. Arch Intern Med 2000;160:1354-60.
6. Connerney I, Shapiro P, McLaughlin J, et al. Relation between depression after coronary artery bypass surgery and 12-month outcome: a prospective study. Lancet 2001;358:1766-71.
7. Frasure-Smith N, Lesperance F, Talajic M. Depression and 18-month prognosis after myocardial infarction. Circulation 1995;91:999-1005.
8. Rugulies R. Depression as a predictor for coronary heart disease. Am J Prev Med 2002;23:51-61.
9. Wulsin L, Singal B. Do depressive symptoms increase the risk for the onset of coronary disease? A systematic quantitative review. Psychosom Med 2003;65:201-10.
10. Carney R, Rich M, Freedland K, et al. Major depressive disorder predicts cardiac events in patients with coronary artery disease. Psychosom Med 1988;50:627-33.
11. Ladwig K, Roll G, Breithardt G, Borggrefe M. Extracardiac contributions to chest pain perception in patients 6 months after acute myocardial infarction. Am Heart J 1999;137:528-34.
12. Levine J, Covino N, Slack W, et al. Psychological predictors of subsequent medical care among patients hospitalized with cardiac disease. J Cardiopulm Rehabil 1996;16:109-16.
13. Lesperance F, Frasure-Smith N, Talajic M, Bourassa M. Five-year risk of cardiac mortality in relation to initial severity and one-year changes in depression symptoms after myocardial infarction. Circulation 2002;105:1049-53.
14. Dwight M, Stoudemire A. Effects of depressive disorders on coronary artery disease: a review. Harv Rev Psychiatry 1997;5:115-122.
15. Penninx B, Beekman A, Honig A, et al. Depression and cardiac mortality. Arch Gen Psychiatry 2001;58:221-7.
16. Writing committee of the ENRICHD investigators. Effects of treating depression and low perceived social support on clinical events after myocardial infarction. JAMA 2003;289:3106-16.
17. Carney RM, Freedland K, Miller G, Jaffe AS. Depression as a risk factor for cardiac mortality and morbidity: a review of potential mechanisms. J Psychosom Res 2002;53:897-902.
18. Musselman D, Evans D, Nemeroff C. The relationship of depression to cardiovascular disease. Arch Gen Psychiatry 1998;55:580-92.
19. Kop WJ. Chronic and acute psychological risk factors for clinical manifestations of coronary artery disease. Psychosom Med 1999;61:476-86.
20. Miller G, Cohen S, Herbert T. Pathways linking major depression and immunity in ambulatory female patients. Psychosom Med 1999;61:850-60.
21. Carney R, Freedland K, Stein P. Change in heart rate and heart rate variability during treatment for depression in patients with coronary heart disease. Psychosom Med 2000;62:639-47.
22. Carney R, Freedland K, Miller G, Jaffe A. Depression as a risk factor for cardiac mortality and morbidity: a review of potential mechanisms. J Psychosom Res 2002;53:897-902.
23. Alexopoulos G, Meyers B, Young R, et al. Vascular depression hypothesis. Psychosom Med 1997;58:113-121.
24. Ziegelstein R, Fauerbach J, Stevens S, et al. Patients with depression are less likely to follow recommendations to reduce cardiac risk during recovery from a myocardial infarction. Arch Intern Med 2000;160:1818-23.
25. Glassman AH, O’Connor C, Califf RM, et al. Sertraline treatment of major depression in patients with acute MI or unstable angina. JAMA 2002;288:701-9.
26. Ko D, Hebert P, Coffey C, et al. B-blocker therapy and symptoms of depression, fatigue, and sexual dysfunction. JAMA 2002;288:351-7.
Beware the men with toupees
HISTORY: Treatment-refractory depression
Mr. S, age 78, has a history of depression that has not responded to selective serotonin reuptake inhibitors and electroconvulsive therapy (ECT).
According to his niece, Mr. S had become withdrawn, suspicious, and forgetful. Several times over the past year, police found him wandering the streets and brought him to the community hospital’s emergency room.
During one emergency room visit, he complained of decreased appetite, poor sleep, and depressed mood. He was subsequently admitted to the psychiatric unit, where he was treated with ECT and discharged on citalopram, 20 mg/d. His symptoms did not improve and he became ataxic and incontinent of urine.
Mr. S’ family placed him in a nursing home, where he became increasingly paranoid. The attending physician prescribed risperidone, 3 mg/d, with no effect. He was then transferred to our psychiatric facility.
At admission, Mr. S told us that a group of men disguised in toupees and mustaches were out to kill him. He said these men had recently killed his niece—with whom he had just spoken on the phone and had seen at the hospital. He suspected that these men were after his money, hired a woman to impersonate his niece and spy on him, and planned to bury his body and his niece’s in a remote place.
On evaluation, Mr. S was suspicious, guarded, and uncooperative, and often ended conversations abruptly. He denied auditory and visual hallucinations, was not suicidal or homicidal, and denied abusing drugs or alcohol. He said constant fear of his imminent murder left him feeling depressed.
Physical and neurologic exams were unremarkable except for mild ataxia. Mr. S’ Folstein Mini-Mental State Examination score was 19/30, indicating moderate cognitive impairment.
Mr. S’ history and behavior suggest depression with psychotic features. Do we have enough information for a diagnosis?
Dr. Greenberg’s and Tampi’s observations
Mr. S is displaying mood symptoms consistent with his prior diagnosis of depression, but with new-onset psychosis as well.
Because of Mr. S’ neurobiologic symptoms, it is improper to diagnose depression with psychotic features without first performing a full medical and neurologic workup. The differential diagnosis needs to include medical and neurologic diagnoses, including:
- delirium secondary to urinary tract infection
- Alzheimer’s and/or vascular dementia
- normal-pressure hydrocephalus
- substance abuse.
A complete dementia and delirium workup and detailed medical history are imperative.
FURTHER HISTORY: Risky behavior
Further history reveals that Mr. S had been having sexual intercourse with prostitutes since his early teens and that this habit continued into his 70s. He had been diagnosed with syphilis in his teens and again in his 50s. Both times he refused to complete the recommended penicillin regimen because he was embarrassed by the diagnosis and had falsely believed that a single penicillin injection would cure him.
Lab tests showed a white blood cell count of 3.5 and a weakly reactive serum venereal disease research laboratory (VDRL) reading.
Reporting of syphilis cases in the United States began in 1941.1 At about that time, Yale University and the Mayo Clinic began conducting clinical trials of penicillin in syphilis treatment.2
Thanks to the advent of penicillin, syphilis incidence has declined dramatically since 1943, when 575,593 cases were reported.3 Only 5,979 cases were reported to the U.S. Centers for Disease Control and Prevention in 2000.4 A slight increase in cases, mainly among homosexual men, was reported in 2001.1,4
The AIDS epidemic and the emergence of crack/cocaine use5,6 were believed to have triggered a brief increase in cases that peaked in 1990. This was likely caused by the high-risk sexual behavior observed in individuals with sexually transmitted diseases and the practice of exchanging sex for drugs.6
Could Mr. S’ syphilis—inadequately treated in his youth—be causing his depression and paranoia decades later? If so, how would you confirm this finding?
Dr. Greenberg’s and Tampi’s observations
Mr. S has a longstanding history of syphilis secondary to high-risk sexual activity. This, combined with the lab findings and his worsening depression and paranoia, points to possible neurosyphilis.
Syphilis, caused by the spirochete Treponema pallidum., can traverse mucous membranes and abraded skin. Transmission is most common during sexual activity but also occurs through blood transfusions and nonsexual lesion contact and from mother to fetus.
Prevalence
- 6,103 cases reported in 2001
- More prevalent among men than women (2.1:1), probably because of elevated prevalence among homosexual men
- African-Americans accounted for 62% of cases in 2001. Prevalence in African–Americans that year was 16 times greater than in whites
Risk factors
- Presence of HIV infection or other sexually transmitted disease
- Unprotected sex
- Residence in urban areas
- Substance abuse
- Homosexuality
Source: References 5 and 6
Because syphilis and its psychiatric effects are relatively uncommon (Box 1), many psychiatrists do not consider neurosyphilis in high-risk patients who present with depression, dementia, or psychosis (Box 2).
HOW SYPHILIS BECOMES NEUROSYPHILIS
Primary syphilis incubates for 10 to 90 days following infection. After this period, an infectious chancre appears along with regional adenopathy. If untreated, the chancre will disappear but the infection will progress.
Secondary syphilis is characterized by skin manifestations and occasionally affects the joints, eyes, bones, kidneys, liver, and CNS. Common effects include condylomata—highly infectious warty lesions—and a diffuse maculopapular rash on the palms and soles. These lesions disappear if left untreated, but most patients then either enter syphilis’ latent stage or experience a potentially fatal relapse of secondary syphilis.5
Latent syphilis usually remains latent or resolves, but about one-third of patients with latent syphilis slowly progress to tertiary syphilis. Neurosyphilis, one of the main forms of tertiary syphilis, can surface 5 to 35 years after an untreated primary infection.7
There are four categories of neurosyphilis:
- General paresis results in dementia, changes in personality, transient hemiparesis, depression, and psychosis.
- Tabes dorsalis degenerates the posterior columns and dorsal root ganglia of the spinal cord. This results in ataxia, parasthesias, decreased proprioception and vibratory sense, Argyll Robertson pupil (an optical disorder in which the pupil does not react normally to light), neurogenic bladder, and sharp shooting pains throughout the body.
- Meningovascular neurosyphilis can result in cranial nerve abnormalities, symptoms of meningitis, and cerebral infarctions.
- Asymptomatic but with CSF positive for syphilis.
Neurosyphilis is fatal if untreated, and treatment usually does not eliminate symptoms but prevents further progression. Approximately 8% of patients with untreated primary syphilis develop neurosyphilis.5,7
Standard nontreponemal tests, such as the VDRL or rapid plasmin reagin, can be used to screen for syphilis. Because these tests often produce false positives, confirm positive results with a syphilis-specific test, such as the fluorescent treponemal antibody absorption (FTA-ABS) test, microhemagglutination assay for antibodies to T pallidum., and the T pallidum. hemagglutination assay.
If neurosyphilis is suspected, CSF testing is strongly recommended. Diagnostic findings include elevated white blood cell and protein counts and a positive VDRL. If the CSF is negative, refer the patient for treatment anyway because false negatives are common. Patients with consistent neurologic symptoms, positive VDRL and/or FTA-ABS, and negative CSF are diagnosed with neurosyphilis and warrant treatment.7
How would you manage Mr. S’ psychiatric symptoms concomitant with medical treatment of late-stage syphilis?
Dr. Greenberg’s and Tampi’s observations
Although no specific guidelines exist for treating psychosis secondary to neurosyphilis, atypical antipsychotics remain the first-line treatment. Atypicals do not interact significantly with penicillin and can be given safely with syphilis treatment. Atypicals also are better tolerated than typical antipsychotics and produce fewer extrapyramidal symptoms, which are common among older patients and those with neurologic diseases.
Screening for syphilis. Every patient with a history of high-risk sexual behavior who presents with new-onset dementia or psychosis should be screened for syphilis. Sexual history can be difficult to obtain from some patients and family members, so communication between providers becomes crucial. Obtain lab test results from other care team members to monitor compliance, and coordinate patient education with other doctors on safe sexual practices.
TREATMENT: Taking his medicine
Mr. S refused further testing and emergency conservatorship was sought. Citalopram was discontinued and risperidone was gradually increased to 6 mg at bedtime. He remained paranoid and delusional.
A brain MRI showed chronic ischemic small-vessel disease. HIV testing was negative, and serum FTA-ABS was reactive. CSF showed elevated protein and white blood cell count with a nonreactive VDRL and a reactive FTA-ABS. A diagnosis of neurosyphilis was made, and treatment was initiated with aqueous crystalline penicillin G, 4 million units every 4 hours for 2 weeks.
Mr. S was discharged back to the nursing home where his penicillin injections were continued. His paranoia diminished slightly but he remained ataxic, incontinent, and confused. He was discharged from the nursing home but needed confirmative HIV screening and repeated CSF testing to determine if syphilis treatment was effective.
Six months after treatment, Mr. S’ niece reports that his paranoia has decreased. He has not needed additional psychiatric hospitalizations.
Related resources
- Merck Manual. www.merck.com. Search: “syphilis”
- U.S. Centers for Disease Control and Prevention—Syphilis elimination: History in the making. www.cdc.gov. Click on “Health Topics A-Z,” then click on “S” and find “syphilis.”
- National Institute of Allergy and Infectious Disease. www.niaid.nih.gov. Search: “syphilis”
Drug brand names
- Citalopram • Celexa
- Risperidone • Risperdal
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. U.S. Centers for Disease Control and Prevention. Primary and secondary syphilis—United States, 2000-2001. MMWR Morb Mortal Wkly Rep. 2002;51:971-3.
2. Mandell GL, Petri WA. Antimicrobial agents: penicillins, cephalosporins, and other beta-lactam antibiotics. In:Hardman JG, Limbird LE, Molinoff PB, et al (eds) Goodman and Gilman’s the pharmacological basis of therapeutics. (9th ed). New York: McGraw-Hill, 1996;1073-4.
3. Lukehart SA, Holmes KK. Spirochetal diseases. In: Braunwald E, Fauci AS, Kasper DL, et al (eds). Harrison’s principles of internal medicine. (14th ed). New York: McGraw-Hill, 1998;1023.-
4. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2001 supplement, syphilis surveillance report. Available at: http://www.cdc.gov/std/stats/2001syphilis.htm. Accessed October 10, 2003.
5. Jacobs RA. Infectious diseases: spirochetal. In: Tierney LM, McPhee SJ, Papadakis MA (eds). Current medical diagnosis and treatment. (39th ed). New York: Lange Medical Books/McGraw-Hill, 2000;1376-86.
6. Hutto B. Syphilis in clinical psychiatry: a review. Psychosomatics. 2001;42:453-60.
7. Carpenter CJ, Lederman MM, Salata RA. Sexually transmitted diseases. In: Andreoli TE, Bennett JC, Carpenter CJ, Plum F (eds). Cecil essentials of medicine. (4th ed). Philadelphia: WB Saunders Co, 1997;742-5.
HISTORY: Treatment-refractory depression
Mr. S, age 78, has a history of depression that has not responded to selective serotonin reuptake inhibitors and electroconvulsive therapy (ECT).
According to his niece, Mr. S had become withdrawn, suspicious, and forgetful. Several times over the past year, police found him wandering the streets and brought him to the community hospital’s emergency room.
During one emergency room visit, he complained of decreased appetite, poor sleep, and depressed mood. He was subsequently admitted to the psychiatric unit, where he was treated with ECT and discharged on citalopram, 20 mg/d. His symptoms did not improve and he became ataxic and incontinent of urine.
Mr. S’ family placed him in a nursing home, where he became increasingly paranoid. The attending physician prescribed risperidone, 3 mg/d, with no effect. He was then transferred to our psychiatric facility.
At admission, Mr. S told us that a group of men disguised in toupees and mustaches were out to kill him. He said these men had recently killed his niece—with whom he had just spoken on the phone and had seen at the hospital. He suspected that these men were after his money, hired a woman to impersonate his niece and spy on him, and planned to bury his body and his niece’s in a remote place.
On evaluation, Mr. S was suspicious, guarded, and uncooperative, and often ended conversations abruptly. He denied auditory and visual hallucinations, was not suicidal or homicidal, and denied abusing drugs or alcohol. He said constant fear of his imminent murder left him feeling depressed.
Physical and neurologic exams were unremarkable except for mild ataxia. Mr. S’ Folstein Mini-Mental State Examination score was 19/30, indicating moderate cognitive impairment.
Mr. S’ history and behavior suggest depression with psychotic features. Do we have enough information for a diagnosis?
Dr. Greenberg’s and Tampi’s observations
Mr. S is displaying mood symptoms consistent with his prior diagnosis of depression, but with new-onset psychosis as well.
Because of Mr. S’ neurobiologic symptoms, it is improper to diagnose depression with psychotic features without first performing a full medical and neurologic workup. The differential diagnosis needs to include medical and neurologic diagnoses, including:
- delirium secondary to urinary tract infection
- Alzheimer’s and/or vascular dementia
- normal-pressure hydrocephalus
- substance abuse.
A complete dementia and delirium workup and detailed medical history are imperative.
FURTHER HISTORY: Risky behavior
Further history reveals that Mr. S had been having sexual intercourse with prostitutes since his early teens and that this habit continued into his 70s. He had been diagnosed with syphilis in his teens and again in his 50s. Both times he refused to complete the recommended penicillin regimen because he was embarrassed by the diagnosis and had falsely believed that a single penicillin injection would cure him.
Lab tests showed a white blood cell count of 3.5 and a weakly reactive serum venereal disease research laboratory (VDRL) reading.
Reporting of syphilis cases in the United States began in 1941.1 At about that time, Yale University and the Mayo Clinic began conducting clinical trials of penicillin in syphilis treatment.2
Thanks to the advent of penicillin, syphilis incidence has declined dramatically since 1943, when 575,593 cases were reported.3 Only 5,979 cases were reported to the U.S. Centers for Disease Control and Prevention in 2000.4 A slight increase in cases, mainly among homosexual men, was reported in 2001.1,4
The AIDS epidemic and the emergence of crack/cocaine use5,6 were believed to have triggered a brief increase in cases that peaked in 1990. This was likely caused by the high-risk sexual behavior observed in individuals with sexually transmitted diseases and the practice of exchanging sex for drugs.6
Could Mr. S’ syphilis—inadequately treated in his youth—be causing his depression and paranoia decades later? If so, how would you confirm this finding?
Dr. Greenberg’s and Tampi’s observations
Mr. S has a longstanding history of syphilis secondary to high-risk sexual activity. This, combined with the lab findings and his worsening depression and paranoia, points to possible neurosyphilis.
Syphilis, caused by the spirochete Treponema pallidum., can traverse mucous membranes and abraded skin. Transmission is most common during sexual activity but also occurs through blood transfusions and nonsexual lesion contact and from mother to fetus.
Prevalence
- 6,103 cases reported in 2001
- More prevalent among men than women (2.1:1), probably because of elevated prevalence among homosexual men
- African-Americans accounted for 62% of cases in 2001. Prevalence in African–Americans that year was 16 times greater than in whites
Risk factors
- Presence of HIV infection or other sexually transmitted disease
- Unprotected sex
- Residence in urban areas
- Substance abuse
- Homosexuality
Source: References 5 and 6
Because syphilis and its psychiatric effects are relatively uncommon (Box 1), many psychiatrists do not consider neurosyphilis in high-risk patients who present with depression, dementia, or psychosis (Box 2).
HOW SYPHILIS BECOMES NEUROSYPHILIS
Primary syphilis incubates for 10 to 90 days following infection. After this period, an infectious chancre appears along with regional adenopathy. If untreated, the chancre will disappear but the infection will progress.
Secondary syphilis is characterized by skin manifestations and occasionally affects the joints, eyes, bones, kidneys, liver, and CNS. Common effects include condylomata—highly infectious warty lesions—and a diffuse maculopapular rash on the palms and soles. These lesions disappear if left untreated, but most patients then either enter syphilis’ latent stage or experience a potentially fatal relapse of secondary syphilis.5
Latent syphilis usually remains latent or resolves, but about one-third of patients with latent syphilis slowly progress to tertiary syphilis. Neurosyphilis, one of the main forms of tertiary syphilis, can surface 5 to 35 years after an untreated primary infection.7
There are four categories of neurosyphilis:
- General paresis results in dementia, changes in personality, transient hemiparesis, depression, and psychosis.
- Tabes dorsalis degenerates the posterior columns and dorsal root ganglia of the spinal cord. This results in ataxia, parasthesias, decreased proprioception and vibratory sense, Argyll Robertson pupil (an optical disorder in which the pupil does not react normally to light), neurogenic bladder, and sharp shooting pains throughout the body.
- Meningovascular neurosyphilis can result in cranial nerve abnormalities, symptoms of meningitis, and cerebral infarctions.
- Asymptomatic but with CSF positive for syphilis.
Neurosyphilis is fatal if untreated, and treatment usually does not eliminate symptoms but prevents further progression. Approximately 8% of patients with untreated primary syphilis develop neurosyphilis.5,7
Standard nontreponemal tests, such as the VDRL or rapid plasmin reagin, can be used to screen for syphilis. Because these tests often produce false positives, confirm positive results with a syphilis-specific test, such as the fluorescent treponemal antibody absorption (FTA-ABS) test, microhemagglutination assay for antibodies to T pallidum., and the T pallidum. hemagglutination assay.
If neurosyphilis is suspected, CSF testing is strongly recommended. Diagnostic findings include elevated white blood cell and protein counts and a positive VDRL. If the CSF is negative, refer the patient for treatment anyway because false negatives are common. Patients with consistent neurologic symptoms, positive VDRL and/or FTA-ABS, and negative CSF are diagnosed with neurosyphilis and warrant treatment.7
How would you manage Mr. S’ psychiatric symptoms concomitant with medical treatment of late-stage syphilis?
Dr. Greenberg’s and Tampi’s observations
Although no specific guidelines exist for treating psychosis secondary to neurosyphilis, atypical antipsychotics remain the first-line treatment. Atypicals do not interact significantly with penicillin and can be given safely with syphilis treatment. Atypicals also are better tolerated than typical antipsychotics and produce fewer extrapyramidal symptoms, which are common among older patients and those with neurologic diseases.
Screening for syphilis. Every patient with a history of high-risk sexual behavior who presents with new-onset dementia or psychosis should be screened for syphilis. Sexual history can be difficult to obtain from some patients and family members, so communication between providers becomes crucial. Obtain lab test results from other care team members to monitor compliance, and coordinate patient education with other doctors on safe sexual practices.
TREATMENT: Taking his medicine
Mr. S refused further testing and emergency conservatorship was sought. Citalopram was discontinued and risperidone was gradually increased to 6 mg at bedtime. He remained paranoid and delusional.
A brain MRI showed chronic ischemic small-vessel disease. HIV testing was negative, and serum FTA-ABS was reactive. CSF showed elevated protein and white blood cell count with a nonreactive VDRL and a reactive FTA-ABS. A diagnosis of neurosyphilis was made, and treatment was initiated with aqueous crystalline penicillin G, 4 million units every 4 hours for 2 weeks.
Mr. S was discharged back to the nursing home where his penicillin injections were continued. His paranoia diminished slightly but he remained ataxic, incontinent, and confused. He was discharged from the nursing home but needed confirmative HIV screening and repeated CSF testing to determine if syphilis treatment was effective.
Six months after treatment, Mr. S’ niece reports that his paranoia has decreased. He has not needed additional psychiatric hospitalizations.
Related resources
- Merck Manual. www.merck.com. Search: “syphilis”
- U.S. Centers for Disease Control and Prevention—Syphilis elimination: History in the making. www.cdc.gov. Click on “Health Topics A-Z,” then click on “S” and find “syphilis.”
- National Institute of Allergy and Infectious Disease. www.niaid.nih.gov. Search: “syphilis”
Drug brand names
- Citalopram • Celexa
- Risperidone • Risperdal
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
HISTORY: Treatment-refractory depression
Mr. S, age 78, has a history of depression that has not responded to selective serotonin reuptake inhibitors and electroconvulsive therapy (ECT).
According to his niece, Mr. S had become withdrawn, suspicious, and forgetful. Several times over the past year, police found him wandering the streets and brought him to the community hospital’s emergency room.
During one emergency room visit, he complained of decreased appetite, poor sleep, and depressed mood. He was subsequently admitted to the psychiatric unit, where he was treated with ECT and discharged on citalopram, 20 mg/d. His symptoms did not improve and he became ataxic and incontinent of urine.
Mr. S’ family placed him in a nursing home, where he became increasingly paranoid. The attending physician prescribed risperidone, 3 mg/d, with no effect. He was then transferred to our psychiatric facility.
At admission, Mr. S told us that a group of men disguised in toupees and mustaches were out to kill him. He said these men had recently killed his niece—with whom he had just spoken on the phone and had seen at the hospital. He suspected that these men were after his money, hired a woman to impersonate his niece and spy on him, and planned to bury his body and his niece’s in a remote place.
On evaluation, Mr. S was suspicious, guarded, and uncooperative, and often ended conversations abruptly. He denied auditory and visual hallucinations, was not suicidal or homicidal, and denied abusing drugs or alcohol. He said constant fear of his imminent murder left him feeling depressed.
Physical and neurologic exams were unremarkable except for mild ataxia. Mr. S’ Folstein Mini-Mental State Examination score was 19/30, indicating moderate cognitive impairment.
Mr. S’ history and behavior suggest depression with psychotic features. Do we have enough information for a diagnosis?
Dr. Greenberg’s and Tampi’s observations
Mr. S is displaying mood symptoms consistent with his prior diagnosis of depression, but with new-onset psychosis as well.
Because of Mr. S’ neurobiologic symptoms, it is improper to diagnose depression with psychotic features without first performing a full medical and neurologic workup. The differential diagnosis needs to include medical and neurologic diagnoses, including:
- delirium secondary to urinary tract infection
- Alzheimer’s and/or vascular dementia
- normal-pressure hydrocephalus
- substance abuse.
A complete dementia and delirium workup and detailed medical history are imperative.
FURTHER HISTORY: Risky behavior
Further history reveals that Mr. S had been having sexual intercourse with prostitutes since his early teens and that this habit continued into his 70s. He had been diagnosed with syphilis in his teens and again in his 50s. Both times he refused to complete the recommended penicillin regimen because he was embarrassed by the diagnosis and had falsely believed that a single penicillin injection would cure him.
Lab tests showed a white blood cell count of 3.5 and a weakly reactive serum venereal disease research laboratory (VDRL) reading.
Reporting of syphilis cases in the United States began in 1941.1 At about that time, Yale University and the Mayo Clinic began conducting clinical trials of penicillin in syphilis treatment.2
Thanks to the advent of penicillin, syphilis incidence has declined dramatically since 1943, when 575,593 cases were reported.3 Only 5,979 cases were reported to the U.S. Centers for Disease Control and Prevention in 2000.4 A slight increase in cases, mainly among homosexual men, was reported in 2001.1,4
The AIDS epidemic and the emergence of crack/cocaine use5,6 were believed to have triggered a brief increase in cases that peaked in 1990. This was likely caused by the high-risk sexual behavior observed in individuals with sexually transmitted diseases and the practice of exchanging sex for drugs.6
Could Mr. S’ syphilis—inadequately treated in his youth—be causing his depression and paranoia decades later? If so, how would you confirm this finding?
Dr. Greenberg’s and Tampi’s observations
Mr. S has a longstanding history of syphilis secondary to high-risk sexual activity. This, combined with the lab findings and his worsening depression and paranoia, points to possible neurosyphilis.
Syphilis, caused by the spirochete Treponema pallidum., can traverse mucous membranes and abraded skin. Transmission is most common during sexual activity but also occurs through blood transfusions and nonsexual lesion contact and from mother to fetus.
Prevalence
- 6,103 cases reported in 2001
- More prevalent among men than women (2.1:1), probably because of elevated prevalence among homosexual men
- African-Americans accounted for 62% of cases in 2001. Prevalence in African–Americans that year was 16 times greater than in whites
Risk factors
- Presence of HIV infection or other sexually transmitted disease
- Unprotected sex
- Residence in urban areas
- Substance abuse
- Homosexuality
Source: References 5 and 6
Because syphilis and its psychiatric effects are relatively uncommon (Box 1), many psychiatrists do not consider neurosyphilis in high-risk patients who present with depression, dementia, or psychosis (Box 2).
HOW SYPHILIS BECOMES NEUROSYPHILIS
Primary syphilis incubates for 10 to 90 days following infection. After this period, an infectious chancre appears along with regional adenopathy. If untreated, the chancre will disappear but the infection will progress.
Secondary syphilis is characterized by skin manifestations and occasionally affects the joints, eyes, bones, kidneys, liver, and CNS. Common effects include condylomata—highly infectious warty lesions—and a diffuse maculopapular rash on the palms and soles. These lesions disappear if left untreated, but most patients then either enter syphilis’ latent stage or experience a potentially fatal relapse of secondary syphilis.5
Latent syphilis usually remains latent or resolves, but about one-third of patients with latent syphilis slowly progress to tertiary syphilis. Neurosyphilis, one of the main forms of tertiary syphilis, can surface 5 to 35 years after an untreated primary infection.7
There are four categories of neurosyphilis:
- General paresis results in dementia, changes in personality, transient hemiparesis, depression, and psychosis.
- Tabes dorsalis degenerates the posterior columns and dorsal root ganglia of the spinal cord. This results in ataxia, parasthesias, decreased proprioception and vibratory sense, Argyll Robertson pupil (an optical disorder in which the pupil does not react normally to light), neurogenic bladder, and sharp shooting pains throughout the body.
- Meningovascular neurosyphilis can result in cranial nerve abnormalities, symptoms of meningitis, and cerebral infarctions.
- Asymptomatic but with CSF positive for syphilis.
Neurosyphilis is fatal if untreated, and treatment usually does not eliminate symptoms but prevents further progression. Approximately 8% of patients with untreated primary syphilis develop neurosyphilis.5,7
Standard nontreponemal tests, such as the VDRL or rapid plasmin reagin, can be used to screen for syphilis. Because these tests often produce false positives, confirm positive results with a syphilis-specific test, such as the fluorescent treponemal antibody absorption (FTA-ABS) test, microhemagglutination assay for antibodies to T pallidum., and the T pallidum. hemagglutination assay.
If neurosyphilis is suspected, CSF testing is strongly recommended. Diagnostic findings include elevated white blood cell and protein counts and a positive VDRL. If the CSF is negative, refer the patient for treatment anyway because false negatives are common. Patients with consistent neurologic symptoms, positive VDRL and/or FTA-ABS, and negative CSF are diagnosed with neurosyphilis and warrant treatment.7
How would you manage Mr. S’ psychiatric symptoms concomitant with medical treatment of late-stage syphilis?
Dr. Greenberg’s and Tampi’s observations
Although no specific guidelines exist for treating psychosis secondary to neurosyphilis, atypical antipsychotics remain the first-line treatment. Atypicals do not interact significantly with penicillin and can be given safely with syphilis treatment. Atypicals also are better tolerated than typical antipsychotics and produce fewer extrapyramidal symptoms, which are common among older patients and those with neurologic diseases.
Screening for syphilis. Every patient with a history of high-risk sexual behavior who presents with new-onset dementia or psychosis should be screened for syphilis. Sexual history can be difficult to obtain from some patients and family members, so communication between providers becomes crucial. Obtain lab test results from other care team members to monitor compliance, and coordinate patient education with other doctors on safe sexual practices.
TREATMENT: Taking his medicine
Mr. S refused further testing and emergency conservatorship was sought. Citalopram was discontinued and risperidone was gradually increased to 6 mg at bedtime. He remained paranoid and delusional.
A brain MRI showed chronic ischemic small-vessel disease. HIV testing was negative, and serum FTA-ABS was reactive. CSF showed elevated protein and white blood cell count with a nonreactive VDRL and a reactive FTA-ABS. A diagnosis of neurosyphilis was made, and treatment was initiated with aqueous crystalline penicillin G, 4 million units every 4 hours for 2 weeks.
Mr. S was discharged back to the nursing home where his penicillin injections were continued. His paranoia diminished slightly but he remained ataxic, incontinent, and confused. He was discharged from the nursing home but needed confirmative HIV screening and repeated CSF testing to determine if syphilis treatment was effective.
Six months after treatment, Mr. S’ niece reports that his paranoia has decreased. He has not needed additional psychiatric hospitalizations.
Related resources
- Merck Manual. www.merck.com. Search: “syphilis”
- U.S. Centers for Disease Control and Prevention—Syphilis elimination: History in the making. www.cdc.gov. Click on “Health Topics A-Z,” then click on “S” and find “syphilis.”
- National Institute of Allergy and Infectious Disease. www.niaid.nih.gov. Search: “syphilis”
Drug brand names
- Citalopram • Celexa
- Risperidone • Risperdal
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. U.S. Centers for Disease Control and Prevention. Primary and secondary syphilis—United States, 2000-2001. MMWR Morb Mortal Wkly Rep. 2002;51:971-3.
2. Mandell GL, Petri WA. Antimicrobial agents: penicillins, cephalosporins, and other beta-lactam antibiotics. In:Hardman JG, Limbird LE, Molinoff PB, et al (eds) Goodman and Gilman’s the pharmacological basis of therapeutics. (9th ed). New York: McGraw-Hill, 1996;1073-4.
3. Lukehart SA, Holmes KK. Spirochetal diseases. In: Braunwald E, Fauci AS, Kasper DL, et al (eds). Harrison’s principles of internal medicine. (14th ed). New York: McGraw-Hill, 1998;1023.-
4. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2001 supplement, syphilis surveillance report. Available at: http://www.cdc.gov/std/stats/2001syphilis.htm. Accessed October 10, 2003.
5. Jacobs RA. Infectious diseases: spirochetal. In: Tierney LM, McPhee SJ, Papadakis MA (eds). Current medical diagnosis and treatment. (39th ed). New York: Lange Medical Books/McGraw-Hill, 2000;1376-86.
6. Hutto B. Syphilis in clinical psychiatry: a review. Psychosomatics. 2001;42:453-60.
7. Carpenter CJ, Lederman MM, Salata RA. Sexually transmitted diseases. In: Andreoli TE, Bennett JC, Carpenter CJ, Plum F (eds). Cecil essentials of medicine. (4th ed). Philadelphia: WB Saunders Co, 1997;742-5.
1. U.S. Centers for Disease Control and Prevention. Primary and secondary syphilis—United States, 2000-2001. MMWR Morb Mortal Wkly Rep. 2002;51:971-3.
2. Mandell GL, Petri WA. Antimicrobial agents: penicillins, cephalosporins, and other beta-lactam antibiotics. In:Hardman JG, Limbird LE, Molinoff PB, et al (eds) Goodman and Gilman’s the pharmacological basis of therapeutics. (9th ed). New York: McGraw-Hill, 1996;1073-4.
3. Lukehart SA, Holmes KK. Spirochetal diseases. In: Braunwald E, Fauci AS, Kasper DL, et al (eds). Harrison’s principles of internal medicine. (14th ed). New York: McGraw-Hill, 1998;1023.-
4. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2001 supplement, syphilis surveillance report. Available at: http://www.cdc.gov/std/stats/2001syphilis.htm. Accessed October 10, 2003.
5. Jacobs RA. Infectious diseases: spirochetal. In: Tierney LM, McPhee SJ, Papadakis MA (eds). Current medical diagnosis and treatment. (39th ed). New York: Lange Medical Books/McGraw-Hill, 2000;1376-86.
6. Hutto B. Syphilis in clinical psychiatry: a review. Psychosomatics. 2001;42:453-60.
7. Carpenter CJ, Lederman MM, Salata RA. Sexually transmitted diseases. In: Andreoli TE, Bennett JC, Carpenter CJ, Plum F (eds). Cecil essentials of medicine. (4th ed). Philadelphia: WB Saunders Co, 1997;742-5.
Tardive dyskinesia: How to prevent and treat a lingering nemesis
Atypical antipsychotics seldom cause tardive dyskinesia (TD), but we cannot let our guard down when prescribing them. Although they pose a much lower risk of TD than do conventional antipsychotics, atypicals can cause TD in vulnerable patients.
Less worrisome than in the past, TD’s associated problems linger, including insidious onset, tendency for persistence, and lack of reliably effective treatment. It is important, therefore, for psychiatrists to:
- identify patients at risk for developing TD
- recognize extrapyramidal symptoms (EPS) when they occur
- and manage these side effects appropriately.
A CHANGING CLINICAL PICTURE
The term “dyskinesias” covers a variety of abnormal involuntary movements (Box). The incidence and prevalence of TD have dropped markedly in the last 10 years, as:
- more and more older, chronically ill patients are switched from conventional to atypical agents
- younger psychotic patients are usually treated with atypicals as first-line therapy and are never exposed to conventional antipsychotics.
Tardive dyskinesia (TD) tends to develop in patients receiving long-term antipsychotic treatment. Its typical movements are choreiform (jerky) or athetoid (writhing), irregular, and purposeless.
TD onset is usually insidious and may occur during drug therapy or weeks after antipsychotics are discontined. Its signs are usually observed in the face or mouth, and typical orofacial dyskinetic movements are:
Lips: puckering, pouting, smacking
Jaw: chewing, biting, side-to-side movements, jaw openings
Tongue: twisting, rolling, undulations, protrusion, darting (“fly-catching”)
Face: blinking, frowning, grimacing.
The trunk and extremities are involved less often. Choreiform finger and wrist movements, flexion and rotation of the ankle, toe movements, foot tapping, and rocking or twisting of the neck, hip, and trunk may be seen. Patients are often oblivious to these movements, which may be only intermittently present and are absent during sleep. Anxiety and arousal states may aggravate TD.
TD prevalence of about 20%—as shown by earlier studies of long-term conventional agents1—is declining. Newer studies comparing atypicals with conventional antipsychotics demonstrate much lower prevalence rates.2,3
TD incidence—estimated by new cases of TD per year of drug treatment—may have declined 10-fold, from 5% with conventional antipsychotics to 0.5% with atypicals. Likewise, incidence in the elderly may have fallen from 25% to 2.5%.4
Risk factors. Despite these improvements, case reports5-7 demonstrate that TD is possible in patients treated with atypicals, even without previous exposure to a conventional antipsychotic. Besides antipsychotic use, risk factors for developing TD include:
- older age
- negative symptoms of schizophrenia
- affective disorders
- acute EPS
- and diabetes mellitus.8
RECOGNIZING TD SYMPTOMS
Recognizing TD may be complicated by the presence of other EPS, particularly drug-induced parkinsonism (DIP). DIP typically develops early and often when treating patients with conventional antipsychotics (Table 1). TD and DIP may occur simultaneously in the same patient, making accurate diagnosis even more difficult.
Other dyskinesias may complicate the diagnosis. Three common TD variants, which may be acute or tardive (occurring after long-term antipsychotic therapy), are:
- akathisia, a distressing and at times irresistible urge to move the legs or other parts of the body
- dystonia, abnormal muscle tone and posture and muscle spasms
- tics, brief muscle contractions, usually in the face, including vocal tics.
AIMS testing. Defining a “case of TD” by dyskinetic movement severity is somewhat arbitrary. A commonly accepted definition is two area scores of “mild” or one rating of “moderate” using the Abnormal Involuntary Movement Scale (AIMS).9 The AIMS has been widely used in epidemiologic and treatment studies of TD and is easy to administer in a clinical setting (see Related Resources).
A careful drug history is required before TD can be diagnosed definitively. Spontaneous dys kinesias—usually orofacial—are sometimes seen in older patients who are not taking neuroleptics.8 Antidepressants, mood stabilizers, or antihistamines may infrequently trigger neurologic side effects—including dyskinesias, akathisia, and tremor—which are almost invariably reversible after the causative agent is withdrawn.8,10
Table 1
Features that differentiate two common extrapyramidal symptoms
| Tardive dyskinesia (TD) | Drug-induced parkinsonism (DIP) | |
|---|---|---|
| Onset | Late | Early |
| Type of movement | Choreoathetoid | Tremor |
| Amount of movement | Increased | Decreased |
| Muscle tone | Decreased | Increased |
| Most common site | Orofacial | Extremities |
| Response to anticholinergics | Tends to worsen | Tends to improve |
MANAGING MILD TD
Atypical antipsychotics have radically altered the clinical outlook for patients with TD and improved our ability to manage their symptoms. The clinician treating a TD patient today rarely faces the dilemma that exists with conventional antipsychotics: discontinue treatment and risk psychotic relapse, or continue treatment and risk persistent TD.
Using atypicals. Today, patients who need antipsychotic therapy for TD are usually already taking atypicals, which may ameliorate TD and control psychotic symptoms. Case reports and some studies have shown therapeutic effects in patients with TD taking olanzapine,3 risperidone,2 quetiapine,11 ziprasidone,12 aripiprazole,13 or the substituted benzamides (such as sulpiride), which are not marketed in the United States.14
Interestingly, TD triggered by taking one atypical may respond to treatment with another. Suzuki et al15 reported that three patients who had developed early-onset TD while taking risperidone showed TD remission after risperidone was replaced by olanzapine in one patient and by quetiapine in the other two.
The atypicals are well tolerated but not without side effects. Weight gain is the most common problem and one with potentially serious health consequences.16
Using conventional agents. Even though atypicals are available, the clinician may consider continuing therapy with conventional antipsychotics in patients with TD when:
- the patient’s mental status has been satisfactory while taking conventional agents
- TD has been mild and stable over an extended time
- the patient has no side effects other than TD.
The literature supports the clinical experience that mild TD rarely worsens with continued antipsychotic therapy. Studies of 5 years or more tend to show TD stability with continued conventional antipsychotic therapy.17 It is prudent to maintain stable chronic psychotic patients with mild TD on the lowest effective dosages of conventional antipsychotics and to monitor them regularly for changes in dyskinesia and psychiatric status.
MANAGING COMPLICATED TD
Managing severe TD or patients showing dystonia, tics, marked akathisia, or DIP coexisting with TD usually calls for more-aggressive interventions (Algorithm).
Algorithm Clinical management of tardive dyskinesia (TD)
Clozapine remains the first-line treatment for difficult TD; it has a very low propensity for inducing DIP and very rarely causes TD.18 Controlled studies,18,19 case reports, and open trials demonstrate its efficacy for reducing TD of all types and severity at a usual dosage of 300 to 500 mg/d. Clozapine’s antidyskinetic effects may be attributed to the absence of rebound after withdrawal and its greater efficacy in more-severe cases.18
Long-term clozapine therapy is recommended for TD, as symptoms remit slowly. Because weight gain, sedation, and other side effects—as well as mandatory blood monitoring—make clozapine less-than-ideal in clinical practice, researchers are seeking other effective therapies for TD.
Other atypicals. The obvious place to look is the other atypicals, which are simpler than clozapine to administer long-term. To date, however, these drugs have not proven to be as reliably effective as clozapine for TD. A recent review concluded that among the atypicals only clozapine induces less EPS than low-potency conventional antipsychotics.20
Nonantipsychotic agents. Other antidyskinetic drugs have come and gone; none has stood the test of time or proven effective in controlled trials. These agents may benefit some TD patients, but improvement is usually not dramatic.
Vitamin E was found to be effective in some TD treatment studies14 but not more effective than placebo in the largest controlled trial.21 Long-term treatment with dopamine-blocking antipsychotics is thought to cause oxidative stressinduced neurotoxicity in the nigrostriatal system.22 Lipid-soluble antioxidants such as vitamin E decrease free-radical formation, and it is possible that vitamin E may yet emerge as a helpful agent in preventing TD.23
Melatonin, a stronger antioxidant than vitamin E, was found to reduce TD in a 6-week placebocontrolled study,22 but the degree of TD improvement was modest. Melatonin’s value as a therapeutic agent for TD remains dubious.23
Miscellaneous. Case reports and studies with small series of TD patients have advanced numerous compounds as possible therapeutic agents (Table 2). Other drugs that occasionally have shown benefit in TD include buspirone, propranolol, pyridoxine (vitamin B6), ondansetron, clonidine, and the neuropeptide ceruletide.
ECT and diet. Suggested nondrug treatments of TD include electroconvulsive therapy (ECT)14 and a diet of mixed branched-chain amino acids.24
Table 2
Compounds that occasionally show benefit in TD
| Class | Example |
|---|---|
| Cholinergics | Lecithin |
| Catecholamine depletors | Tetrabenazine (investigational orphan drug) |
| Calcium channel blockers | Verapamil |
| Gabaergic compounds | Baclofen |
| Benzodiazepines | Clonazepam |
MANAGING TD VARIANTS
TD variants are notoriously difficult to treat but tend to respond to clozapine.18 In addition:
- Tardive dystonia is often treated with reserpine, tetrabenazine, or high doses of anticholinergic drugs.25 Botulinum toxin A injections into affected muscles may be remarkably effective but must be repeated regularly.25
- Tardive akathisia may improve slowly with clozapine, propranolol, or benzodiazepines.25
Managing severe or atypical TD is usually beyond the expertise of the practicing psychiatrist. Obtaining consultation from a psychopharmacologist or a neurologist experienced in treating movement disorders is highly recommended.
PREVENTING TD
Conventional antipsychotics are still prescribed by psychiatrists, internists, and family physicians and are often given in emergency rooms. Avoiding these drugs whenever possible and using the lowest effective dosages will reduce the risk of TD.26
Patients at relatively high risk for TD—the elderly, those who are very sensitive to acute EPS, and those with affective disorders or diabetes mellitus—are rarely candidates for conventional neuroleptics if a suitable alternative exists. Genetic research may further identify individuals susceptible to TD.27
- Abnormal Involuntary Movement Scale (AIMS). www.dr-bob.org/tips/aims.html
- Bloom FE, Kupfer DJ (eds). Psychopharmacology: The fourth generation of progress. New York: Raven Press, 1995.
- Tandon R, Halbreich U (eds). Atypical antipsychotics: Efficacy and tolerability—achieving the optimal balance. Psychoneuroendocrinology 2003;28(suppl 1).
Drug brand names
- Aripiprazole • Abilify
- Baclofen • Lioresal
- Buspirone • BuSpar
- Clonazepam • Klonopin
- Clonidine • Catapres
- Clozapine • Clozaril
- Olanzapine • Zyprexa
- Ondansetron • Zofran
- Propranolol • Inderal
- Quetiapine • Seroquel
- Risperidone • Risperdal
- Verapamil • Calan, others
- Ziprasidone • Geodon
Disclosure
Dr. Gardos receives research grant support from Forest Laboratories.
1. Woerner M, Kane JM, Lieberman JA, et al. The prevalence of tardive dyskinesia, J Clin Psychopharmacology 1991;11:34-42.
2. Caroff SN, Mann SC, Campbell EC, et al. Movement disorders associated with atypical antipsychotic drugs. J Clin Psychiatry 2002;63(suppl 4):12-19.
3. Tollefson GD, Beasley CM, Tran PV, et al. Olanzapine versus haloperidol in the treatment of schizophrenia and schizophreniform disorders: results of an international collaborative trial. Am J Psychiatry 1997;154:457-65.
4. Jeste DV, Lacro JP, Bailey A, et al. Lower incidence of tardive dyskinesia with risperidone compared with haloperidol in older patients. J Am Geriatr Soc 1999;47:716-19.
5. Kumet R, Freeman MP. Clozapine and tardive dyskinesia. J Clin Psychiatry 2002;63:167-8.
6. Hong KS, Cheong SS, Woo J-M, Kim E. Risperidone-induced tardive dyskinesia. Am J Psychiatry 1999;156:1290.-
7. Ghaemi SN, Ko JY. Quetiapine-related tardive dyskinesia. Am J Psychiatry 2001;158:1737.-
8. Kane JM. Tardive dyskinesia: epidemiological and clinical presentation. In: Bloom FE, Kupfer DJ (eds.) Psychopharmacology: The fourth generation of progress. New York: Raven Press, Ltd, 1995;1485-95.
9. Guy W. ECDEU assessment manual for psychopharmacology (rev. ed). Washington, DC: Department of Health, Education and Welfare, 1976.
10. Madhusoodanan S, Brenner R. Reversible choreiform dyskinesia and extrapyramidal symptoms associated with sertraline therapy. J Clin Psychopharmacology 1997;17:138-9.
11. Glazer WM, Morgenstern H, Pultz JA, et al. Incidence of tardive dyskinesia is lower with quetiapine treatment than with typical antipsychotics in patients with schizophrenia and schizo-affective disorder. Schizophrenia Res 2000;41:206-7.
12. Hirsch SR, Kissling W, Bauml J, et al. A 28-week comparison of ziprasidone and haloperidol in outpatients with stable schizophrenia. J Clin Psychiatry 2002;63:516-23.
13. Kujawa M, Sala A, Ingenito GG, et al. Aripiprazole for long-term maintenance treatment of schizophrenia (poster presentation). Montreal, Canada: Collegium Internationale Neuropsychopharmacologicum 23rd congress, June 23-27, 2002.
14. Gupta S, Mosnik D, Black DW, et al. Tardive dyskinesia: review of treatments past, present and future. Ann Clin Psychiatry 1999;11:257-66.
15. Suzuki E, Obata M, Yoshida Y, Miyaoka H. Tardive dyskinesia with risperidone and anticholinergics. Am J Psychiatry 2002;159:1948.-
16. Nasrallah HA. A review of the effect of atypical antipsychotics on weight. Psychoneuroendocrinology 2003;28(suppl 1):83-96.
17. Gardos G, Casey DE, Cole JO, et al. Ten-year outcome of tardive dyskinesia. Am J Psychiatry 1994;151:836-41.
18. Lieberman JA, Saltz BL, Johns CA, et al. The effects of clozapine on tardive dyskinesia. Br J Psychiatry 1991;158:503-10.
19. Tamminga CA, Thaker GK, Moran M, et al. Clozapine in tardive dyskinesia: observations from human and animal model studies J Clin Psychiatry 1994;55(suppl B):102-6.
20. Leucht S, Wahlbeck C, Hermann J, Kissling W. New-generation antipsychotics versus low-potency conventional antipsychotics: a systematic review and meta-analysis. Lancet 2003;361:1581-9.
21. Adler LA, Rotrosen J, Edson R, et al. Vitamin E treatment of tardive dyskinesia. Arch Gen Psychiatry 1999;56:836-41.
22. Shamir E, Barak Y, Shalman I, et al. Melatonin treatment for tardive dyskinesia. Arch Gen Psychiatry 2001;58:1046-52.
23. Glazer WM, Woods SW. Should Sisyphus have taken melatonin? Arch Gen Psychiatry 2001;58:1054-5.
24. Richardson MA, Bevans M, Read LL, et al. Efficacy of the branched-chain amino acids in the treatment of tardive dyskinesia in men. Am J Psychiatry 2003;160:1117-24.
25. Gardos G, Cole JO. The evaluation and treatment of neurolepticinduced movement disorders. Harvard Rev Psychiatry 1995;3:130-9.
26. Lohr JB, Caligiuri MP, Edson R, et al. Treatment predictors of extrapyramidal side effects in patients with tardive dyskinesia: results from Veterans Affairs Cooperative Study 394. J Clin Psychopharmacol 2002;22:196-200.
27. Casey DE. Effect of clozapine therapy in schizophrenic individuals at risk for tardive dyskinesia. J Clin Psychiatry 1998;59(suppl 3):31-7.
Atypical antipsychotics seldom cause tardive dyskinesia (TD), but we cannot let our guard down when prescribing them. Although they pose a much lower risk of TD than do conventional antipsychotics, atypicals can cause TD in vulnerable patients.
Less worrisome than in the past, TD’s associated problems linger, including insidious onset, tendency for persistence, and lack of reliably effective treatment. It is important, therefore, for psychiatrists to:
- identify patients at risk for developing TD
- recognize extrapyramidal symptoms (EPS) when they occur
- and manage these side effects appropriately.
A CHANGING CLINICAL PICTURE
The term “dyskinesias” covers a variety of abnormal involuntary movements (Box). The incidence and prevalence of TD have dropped markedly in the last 10 years, as:
- more and more older, chronically ill patients are switched from conventional to atypical agents
- younger psychotic patients are usually treated with atypicals as first-line therapy and are never exposed to conventional antipsychotics.
Tardive dyskinesia (TD) tends to develop in patients receiving long-term antipsychotic treatment. Its typical movements are choreiform (jerky) or athetoid (writhing), irregular, and purposeless.
TD onset is usually insidious and may occur during drug therapy or weeks after antipsychotics are discontined. Its signs are usually observed in the face or mouth, and typical orofacial dyskinetic movements are:
Lips: puckering, pouting, smacking
Jaw: chewing, biting, side-to-side movements, jaw openings
Tongue: twisting, rolling, undulations, protrusion, darting (“fly-catching”)
Face: blinking, frowning, grimacing.
The trunk and extremities are involved less often. Choreiform finger and wrist movements, flexion and rotation of the ankle, toe movements, foot tapping, and rocking or twisting of the neck, hip, and trunk may be seen. Patients are often oblivious to these movements, which may be only intermittently present and are absent during sleep. Anxiety and arousal states may aggravate TD.
TD prevalence of about 20%—as shown by earlier studies of long-term conventional agents1—is declining. Newer studies comparing atypicals with conventional antipsychotics demonstrate much lower prevalence rates.2,3
TD incidence—estimated by new cases of TD per year of drug treatment—may have declined 10-fold, from 5% with conventional antipsychotics to 0.5% with atypicals. Likewise, incidence in the elderly may have fallen from 25% to 2.5%.4
Risk factors. Despite these improvements, case reports5-7 demonstrate that TD is possible in patients treated with atypicals, even without previous exposure to a conventional antipsychotic. Besides antipsychotic use, risk factors for developing TD include:
- older age
- negative symptoms of schizophrenia
- affective disorders
- acute EPS
- and diabetes mellitus.8
RECOGNIZING TD SYMPTOMS
Recognizing TD may be complicated by the presence of other EPS, particularly drug-induced parkinsonism (DIP). DIP typically develops early and often when treating patients with conventional antipsychotics (Table 1). TD and DIP may occur simultaneously in the same patient, making accurate diagnosis even more difficult.
Other dyskinesias may complicate the diagnosis. Three common TD variants, which may be acute or tardive (occurring after long-term antipsychotic therapy), are:
- akathisia, a distressing and at times irresistible urge to move the legs or other parts of the body
- dystonia, abnormal muscle tone and posture and muscle spasms
- tics, brief muscle contractions, usually in the face, including vocal tics.
AIMS testing. Defining a “case of TD” by dyskinetic movement severity is somewhat arbitrary. A commonly accepted definition is two area scores of “mild” or one rating of “moderate” using the Abnormal Involuntary Movement Scale (AIMS).9 The AIMS has been widely used in epidemiologic and treatment studies of TD and is easy to administer in a clinical setting (see Related Resources).
A careful drug history is required before TD can be diagnosed definitively. Spontaneous dys kinesias—usually orofacial—are sometimes seen in older patients who are not taking neuroleptics.8 Antidepressants, mood stabilizers, or antihistamines may infrequently trigger neurologic side effects—including dyskinesias, akathisia, and tremor—which are almost invariably reversible after the causative agent is withdrawn.8,10
Table 1
Features that differentiate two common extrapyramidal symptoms
| Tardive dyskinesia (TD) | Drug-induced parkinsonism (DIP) | |
|---|---|---|
| Onset | Late | Early |
| Type of movement | Choreoathetoid | Tremor |
| Amount of movement | Increased | Decreased |
| Muscle tone | Decreased | Increased |
| Most common site | Orofacial | Extremities |
| Response to anticholinergics | Tends to worsen | Tends to improve |
MANAGING MILD TD
Atypical antipsychotics have radically altered the clinical outlook for patients with TD and improved our ability to manage their symptoms. The clinician treating a TD patient today rarely faces the dilemma that exists with conventional antipsychotics: discontinue treatment and risk psychotic relapse, or continue treatment and risk persistent TD.
Using atypicals. Today, patients who need antipsychotic therapy for TD are usually already taking atypicals, which may ameliorate TD and control psychotic symptoms. Case reports and some studies have shown therapeutic effects in patients with TD taking olanzapine,3 risperidone,2 quetiapine,11 ziprasidone,12 aripiprazole,13 or the substituted benzamides (such as sulpiride), which are not marketed in the United States.14
Interestingly, TD triggered by taking one atypical may respond to treatment with another. Suzuki et al15 reported that three patients who had developed early-onset TD while taking risperidone showed TD remission after risperidone was replaced by olanzapine in one patient and by quetiapine in the other two.
The atypicals are well tolerated but not without side effects. Weight gain is the most common problem and one with potentially serious health consequences.16
Using conventional agents. Even though atypicals are available, the clinician may consider continuing therapy with conventional antipsychotics in patients with TD when:
- the patient’s mental status has been satisfactory while taking conventional agents
- TD has been mild and stable over an extended time
- the patient has no side effects other than TD.
The literature supports the clinical experience that mild TD rarely worsens with continued antipsychotic therapy. Studies of 5 years or more tend to show TD stability with continued conventional antipsychotic therapy.17 It is prudent to maintain stable chronic psychotic patients with mild TD on the lowest effective dosages of conventional antipsychotics and to monitor them regularly for changes in dyskinesia and psychiatric status.
MANAGING COMPLICATED TD
Managing severe TD or patients showing dystonia, tics, marked akathisia, or DIP coexisting with TD usually calls for more-aggressive interventions (Algorithm).
Algorithm Clinical management of tardive dyskinesia (TD)
Clozapine remains the first-line treatment for difficult TD; it has a very low propensity for inducing DIP and very rarely causes TD.18 Controlled studies,18,19 case reports, and open trials demonstrate its efficacy for reducing TD of all types and severity at a usual dosage of 300 to 500 mg/d. Clozapine’s antidyskinetic effects may be attributed to the absence of rebound after withdrawal and its greater efficacy in more-severe cases.18
Long-term clozapine therapy is recommended for TD, as symptoms remit slowly. Because weight gain, sedation, and other side effects—as well as mandatory blood monitoring—make clozapine less-than-ideal in clinical practice, researchers are seeking other effective therapies for TD.
Other atypicals. The obvious place to look is the other atypicals, which are simpler than clozapine to administer long-term. To date, however, these drugs have not proven to be as reliably effective as clozapine for TD. A recent review concluded that among the atypicals only clozapine induces less EPS than low-potency conventional antipsychotics.20
Nonantipsychotic agents. Other antidyskinetic drugs have come and gone; none has stood the test of time or proven effective in controlled trials. These agents may benefit some TD patients, but improvement is usually not dramatic.
Vitamin E was found to be effective in some TD treatment studies14 but not more effective than placebo in the largest controlled trial.21 Long-term treatment with dopamine-blocking antipsychotics is thought to cause oxidative stressinduced neurotoxicity in the nigrostriatal system.22 Lipid-soluble antioxidants such as vitamin E decrease free-radical formation, and it is possible that vitamin E may yet emerge as a helpful agent in preventing TD.23
Melatonin, a stronger antioxidant than vitamin E, was found to reduce TD in a 6-week placebocontrolled study,22 but the degree of TD improvement was modest. Melatonin’s value as a therapeutic agent for TD remains dubious.23
Miscellaneous. Case reports and studies with small series of TD patients have advanced numerous compounds as possible therapeutic agents (Table 2). Other drugs that occasionally have shown benefit in TD include buspirone, propranolol, pyridoxine (vitamin B6), ondansetron, clonidine, and the neuropeptide ceruletide.
ECT and diet. Suggested nondrug treatments of TD include electroconvulsive therapy (ECT)14 and a diet of mixed branched-chain amino acids.24
Table 2
Compounds that occasionally show benefit in TD
| Class | Example |
|---|---|
| Cholinergics | Lecithin |
| Catecholamine depletors | Tetrabenazine (investigational orphan drug) |
| Calcium channel blockers | Verapamil |
| Gabaergic compounds | Baclofen |
| Benzodiazepines | Clonazepam |
MANAGING TD VARIANTS
TD variants are notoriously difficult to treat but tend to respond to clozapine.18 In addition:
- Tardive dystonia is often treated with reserpine, tetrabenazine, or high doses of anticholinergic drugs.25 Botulinum toxin A injections into affected muscles may be remarkably effective but must be repeated regularly.25
- Tardive akathisia may improve slowly with clozapine, propranolol, or benzodiazepines.25
Managing severe or atypical TD is usually beyond the expertise of the practicing psychiatrist. Obtaining consultation from a psychopharmacologist or a neurologist experienced in treating movement disorders is highly recommended.
PREVENTING TD
Conventional antipsychotics are still prescribed by psychiatrists, internists, and family physicians and are often given in emergency rooms. Avoiding these drugs whenever possible and using the lowest effective dosages will reduce the risk of TD.26
Patients at relatively high risk for TD—the elderly, those who are very sensitive to acute EPS, and those with affective disorders or diabetes mellitus—are rarely candidates for conventional neuroleptics if a suitable alternative exists. Genetic research may further identify individuals susceptible to TD.27
- Abnormal Involuntary Movement Scale (AIMS). www.dr-bob.org/tips/aims.html
- Bloom FE, Kupfer DJ (eds). Psychopharmacology: The fourth generation of progress. New York: Raven Press, 1995.
- Tandon R, Halbreich U (eds). Atypical antipsychotics: Efficacy and tolerability—achieving the optimal balance. Psychoneuroendocrinology 2003;28(suppl 1).
Drug brand names
- Aripiprazole • Abilify
- Baclofen • Lioresal
- Buspirone • BuSpar
- Clonazepam • Klonopin
- Clonidine • Catapres
- Clozapine • Clozaril
- Olanzapine • Zyprexa
- Ondansetron • Zofran
- Propranolol • Inderal
- Quetiapine • Seroquel
- Risperidone • Risperdal
- Verapamil • Calan, others
- Ziprasidone • Geodon
Disclosure
Dr. Gardos receives research grant support from Forest Laboratories.
Atypical antipsychotics seldom cause tardive dyskinesia (TD), but we cannot let our guard down when prescribing them. Although they pose a much lower risk of TD than do conventional antipsychotics, atypicals can cause TD in vulnerable patients.
Less worrisome than in the past, TD’s associated problems linger, including insidious onset, tendency for persistence, and lack of reliably effective treatment. It is important, therefore, for psychiatrists to:
- identify patients at risk for developing TD
- recognize extrapyramidal symptoms (EPS) when they occur
- and manage these side effects appropriately.
A CHANGING CLINICAL PICTURE
The term “dyskinesias” covers a variety of abnormal involuntary movements (Box). The incidence and prevalence of TD have dropped markedly in the last 10 years, as:
- more and more older, chronically ill patients are switched from conventional to atypical agents
- younger psychotic patients are usually treated with atypicals as first-line therapy and are never exposed to conventional antipsychotics.
Tardive dyskinesia (TD) tends to develop in patients receiving long-term antipsychotic treatment. Its typical movements are choreiform (jerky) or athetoid (writhing), irregular, and purposeless.
TD onset is usually insidious and may occur during drug therapy or weeks after antipsychotics are discontined. Its signs are usually observed in the face or mouth, and typical orofacial dyskinetic movements are:
Lips: puckering, pouting, smacking
Jaw: chewing, biting, side-to-side movements, jaw openings
Tongue: twisting, rolling, undulations, protrusion, darting (“fly-catching”)
Face: blinking, frowning, grimacing.
The trunk and extremities are involved less often. Choreiform finger and wrist movements, flexion and rotation of the ankle, toe movements, foot tapping, and rocking or twisting of the neck, hip, and trunk may be seen. Patients are often oblivious to these movements, which may be only intermittently present and are absent during sleep. Anxiety and arousal states may aggravate TD.
TD prevalence of about 20%—as shown by earlier studies of long-term conventional agents1—is declining. Newer studies comparing atypicals with conventional antipsychotics demonstrate much lower prevalence rates.2,3
TD incidence—estimated by new cases of TD per year of drug treatment—may have declined 10-fold, from 5% with conventional antipsychotics to 0.5% with atypicals. Likewise, incidence in the elderly may have fallen from 25% to 2.5%.4
Risk factors. Despite these improvements, case reports5-7 demonstrate that TD is possible in patients treated with atypicals, even without previous exposure to a conventional antipsychotic. Besides antipsychotic use, risk factors for developing TD include:
- older age
- negative symptoms of schizophrenia
- affective disorders
- acute EPS
- and diabetes mellitus.8
RECOGNIZING TD SYMPTOMS
Recognizing TD may be complicated by the presence of other EPS, particularly drug-induced parkinsonism (DIP). DIP typically develops early and often when treating patients with conventional antipsychotics (Table 1). TD and DIP may occur simultaneously in the same patient, making accurate diagnosis even more difficult.
Other dyskinesias may complicate the diagnosis. Three common TD variants, which may be acute or tardive (occurring after long-term antipsychotic therapy), are:
- akathisia, a distressing and at times irresistible urge to move the legs or other parts of the body
- dystonia, abnormal muscle tone and posture and muscle spasms
- tics, brief muscle contractions, usually in the face, including vocal tics.
AIMS testing. Defining a “case of TD” by dyskinetic movement severity is somewhat arbitrary. A commonly accepted definition is two area scores of “mild” or one rating of “moderate” using the Abnormal Involuntary Movement Scale (AIMS).9 The AIMS has been widely used in epidemiologic and treatment studies of TD and is easy to administer in a clinical setting (see Related Resources).
A careful drug history is required before TD can be diagnosed definitively. Spontaneous dys kinesias—usually orofacial—are sometimes seen in older patients who are not taking neuroleptics.8 Antidepressants, mood stabilizers, or antihistamines may infrequently trigger neurologic side effects—including dyskinesias, akathisia, and tremor—which are almost invariably reversible after the causative agent is withdrawn.8,10
Table 1
Features that differentiate two common extrapyramidal symptoms
| Tardive dyskinesia (TD) | Drug-induced parkinsonism (DIP) | |
|---|---|---|
| Onset | Late | Early |
| Type of movement | Choreoathetoid | Tremor |
| Amount of movement | Increased | Decreased |
| Muscle tone | Decreased | Increased |
| Most common site | Orofacial | Extremities |
| Response to anticholinergics | Tends to worsen | Tends to improve |
MANAGING MILD TD
Atypical antipsychotics have radically altered the clinical outlook for patients with TD and improved our ability to manage their symptoms. The clinician treating a TD patient today rarely faces the dilemma that exists with conventional antipsychotics: discontinue treatment and risk psychotic relapse, or continue treatment and risk persistent TD.
Using atypicals. Today, patients who need antipsychotic therapy for TD are usually already taking atypicals, which may ameliorate TD and control psychotic symptoms. Case reports and some studies have shown therapeutic effects in patients with TD taking olanzapine,3 risperidone,2 quetiapine,11 ziprasidone,12 aripiprazole,13 or the substituted benzamides (such as sulpiride), which are not marketed in the United States.14
Interestingly, TD triggered by taking one atypical may respond to treatment with another. Suzuki et al15 reported that three patients who had developed early-onset TD while taking risperidone showed TD remission after risperidone was replaced by olanzapine in one patient and by quetiapine in the other two.
The atypicals are well tolerated but not without side effects. Weight gain is the most common problem and one with potentially serious health consequences.16
Using conventional agents. Even though atypicals are available, the clinician may consider continuing therapy with conventional antipsychotics in patients with TD when:
- the patient’s mental status has been satisfactory while taking conventional agents
- TD has been mild and stable over an extended time
- the patient has no side effects other than TD.
The literature supports the clinical experience that mild TD rarely worsens with continued antipsychotic therapy. Studies of 5 years or more tend to show TD stability with continued conventional antipsychotic therapy.17 It is prudent to maintain stable chronic psychotic patients with mild TD on the lowest effective dosages of conventional antipsychotics and to monitor them regularly for changes in dyskinesia and psychiatric status.
MANAGING COMPLICATED TD
Managing severe TD or patients showing dystonia, tics, marked akathisia, or DIP coexisting with TD usually calls for more-aggressive interventions (Algorithm).
Algorithm Clinical management of tardive dyskinesia (TD)
Clozapine remains the first-line treatment for difficult TD; it has a very low propensity for inducing DIP and very rarely causes TD.18 Controlled studies,18,19 case reports, and open trials demonstrate its efficacy for reducing TD of all types and severity at a usual dosage of 300 to 500 mg/d. Clozapine’s antidyskinetic effects may be attributed to the absence of rebound after withdrawal and its greater efficacy in more-severe cases.18
Long-term clozapine therapy is recommended for TD, as symptoms remit slowly. Because weight gain, sedation, and other side effects—as well as mandatory blood monitoring—make clozapine less-than-ideal in clinical practice, researchers are seeking other effective therapies for TD.
Other atypicals. The obvious place to look is the other atypicals, which are simpler than clozapine to administer long-term. To date, however, these drugs have not proven to be as reliably effective as clozapine for TD. A recent review concluded that among the atypicals only clozapine induces less EPS than low-potency conventional antipsychotics.20
Nonantipsychotic agents. Other antidyskinetic drugs have come and gone; none has stood the test of time or proven effective in controlled trials. These agents may benefit some TD patients, but improvement is usually not dramatic.
Vitamin E was found to be effective in some TD treatment studies14 but not more effective than placebo in the largest controlled trial.21 Long-term treatment with dopamine-blocking antipsychotics is thought to cause oxidative stressinduced neurotoxicity in the nigrostriatal system.22 Lipid-soluble antioxidants such as vitamin E decrease free-radical formation, and it is possible that vitamin E may yet emerge as a helpful agent in preventing TD.23
Melatonin, a stronger antioxidant than vitamin E, was found to reduce TD in a 6-week placebocontrolled study,22 but the degree of TD improvement was modest. Melatonin’s value as a therapeutic agent for TD remains dubious.23
Miscellaneous. Case reports and studies with small series of TD patients have advanced numerous compounds as possible therapeutic agents (Table 2). Other drugs that occasionally have shown benefit in TD include buspirone, propranolol, pyridoxine (vitamin B6), ondansetron, clonidine, and the neuropeptide ceruletide.
ECT and diet. Suggested nondrug treatments of TD include electroconvulsive therapy (ECT)14 and a diet of mixed branched-chain amino acids.24
Table 2
Compounds that occasionally show benefit in TD
| Class | Example |
|---|---|
| Cholinergics | Lecithin |
| Catecholamine depletors | Tetrabenazine (investigational orphan drug) |
| Calcium channel blockers | Verapamil |
| Gabaergic compounds | Baclofen |
| Benzodiazepines | Clonazepam |
MANAGING TD VARIANTS
TD variants are notoriously difficult to treat but tend to respond to clozapine.18 In addition:
- Tardive dystonia is often treated with reserpine, tetrabenazine, or high doses of anticholinergic drugs.25 Botulinum toxin A injections into affected muscles may be remarkably effective but must be repeated regularly.25
- Tardive akathisia may improve slowly with clozapine, propranolol, or benzodiazepines.25
Managing severe or atypical TD is usually beyond the expertise of the practicing psychiatrist. Obtaining consultation from a psychopharmacologist or a neurologist experienced in treating movement disorders is highly recommended.
PREVENTING TD
Conventional antipsychotics are still prescribed by psychiatrists, internists, and family physicians and are often given in emergency rooms. Avoiding these drugs whenever possible and using the lowest effective dosages will reduce the risk of TD.26
Patients at relatively high risk for TD—the elderly, those who are very sensitive to acute EPS, and those with affective disorders or diabetes mellitus—are rarely candidates for conventional neuroleptics if a suitable alternative exists. Genetic research may further identify individuals susceptible to TD.27
- Abnormal Involuntary Movement Scale (AIMS). www.dr-bob.org/tips/aims.html
- Bloom FE, Kupfer DJ (eds). Psychopharmacology: The fourth generation of progress. New York: Raven Press, 1995.
- Tandon R, Halbreich U (eds). Atypical antipsychotics: Efficacy and tolerability—achieving the optimal balance. Psychoneuroendocrinology 2003;28(suppl 1).
Drug brand names
- Aripiprazole • Abilify
- Baclofen • Lioresal
- Buspirone • BuSpar
- Clonazepam • Klonopin
- Clonidine • Catapres
- Clozapine • Clozaril
- Olanzapine • Zyprexa
- Ondansetron • Zofran
- Propranolol • Inderal
- Quetiapine • Seroquel
- Risperidone • Risperdal
- Verapamil • Calan, others
- Ziprasidone • Geodon
Disclosure
Dr. Gardos receives research grant support from Forest Laboratories.
1. Woerner M, Kane JM, Lieberman JA, et al. The prevalence of tardive dyskinesia, J Clin Psychopharmacology 1991;11:34-42.
2. Caroff SN, Mann SC, Campbell EC, et al. Movement disorders associated with atypical antipsychotic drugs. J Clin Psychiatry 2002;63(suppl 4):12-19.
3. Tollefson GD, Beasley CM, Tran PV, et al. Olanzapine versus haloperidol in the treatment of schizophrenia and schizophreniform disorders: results of an international collaborative trial. Am J Psychiatry 1997;154:457-65.
4. Jeste DV, Lacro JP, Bailey A, et al. Lower incidence of tardive dyskinesia with risperidone compared with haloperidol in older patients. J Am Geriatr Soc 1999;47:716-19.
5. Kumet R, Freeman MP. Clozapine and tardive dyskinesia. J Clin Psychiatry 2002;63:167-8.
6. Hong KS, Cheong SS, Woo J-M, Kim E. Risperidone-induced tardive dyskinesia. Am J Psychiatry 1999;156:1290.-
7. Ghaemi SN, Ko JY. Quetiapine-related tardive dyskinesia. Am J Psychiatry 2001;158:1737.-
8. Kane JM. Tardive dyskinesia: epidemiological and clinical presentation. In: Bloom FE, Kupfer DJ (eds.) Psychopharmacology: The fourth generation of progress. New York: Raven Press, Ltd, 1995;1485-95.
9. Guy W. ECDEU assessment manual for psychopharmacology (rev. ed). Washington, DC: Department of Health, Education and Welfare, 1976.
10. Madhusoodanan S, Brenner R. Reversible choreiform dyskinesia and extrapyramidal symptoms associated with sertraline therapy. J Clin Psychopharmacology 1997;17:138-9.
11. Glazer WM, Morgenstern H, Pultz JA, et al. Incidence of tardive dyskinesia is lower with quetiapine treatment than with typical antipsychotics in patients with schizophrenia and schizo-affective disorder. Schizophrenia Res 2000;41:206-7.
12. Hirsch SR, Kissling W, Bauml J, et al. A 28-week comparison of ziprasidone and haloperidol in outpatients with stable schizophrenia. J Clin Psychiatry 2002;63:516-23.
13. Kujawa M, Sala A, Ingenito GG, et al. Aripiprazole for long-term maintenance treatment of schizophrenia (poster presentation). Montreal, Canada: Collegium Internationale Neuropsychopharmacologicum 23rd congress, June 23-27, 2002.
14. Gupta S, Mosnik D, Black DW, et al. Tardive dyskinesia: review of treatments past, present and future. Ann Clin Psychiatry 1999;11:257-66.
15. Suzuki E, Obata M, Yoshida Y, Miyaoka H. Tardive dyskinesia with risperidone and anticholinergics. Am J Psychiatry 2002;159:1948.-
16. Nasrallah HA. A review of the effect of atypical antipsychotics on weight. Psychoneuroendocrinology 2003;28(suppl 1):83-96.
17. Gardos G, Casey DE, Cole JO, et al. Ten-year outcome of tardive dyskinesia. Am J Psychiatry 1994;151:836-41.
18. Lieberman JA, Saltz BL, Johns CA, et al. The effects of clozapine on tardive dyskinesia. Br J Psychiatry 1991;158:503-10.
19. Tamminga CA, Thaker GK, Moran M, et al. Clozapine in tardive dyskinesia: observations from human and animal model studies J Clin Psychiatry 1994;55(suppl B):102-6.
20. Leucht S, Wahlbeck C, Hermann J, Kissling W. New-generation antipsychotics versus low-potency conventional antipsychotics: a systematic review and meta-analysis. Lancet 2003;361:1581-9.
21. Adler LA, Rotrosen J, Edson R, et al. Vitamin E treatment of tardive dyskinesia. Arch Gen Psychiatry 1999;56:836-41.
22. Shamir E, Barak Y, Shalman I, et al. Melatonin treatment for tardive dyskinesia. Arch Gen Psychiatry 2001;58:1046-52.
23. Glazer WM, Woods SW. Should Sisyphus have taken melatonin? Arch Gen Psychiatry 2001;58:1054-5.
24. Richardson MA, Bevans M, Read LL, et al. Efficacy of the branched-chain amino acids in the treatment of tardive dyskinesia in men. Am J Psychiatry 2003;160:1117-24.
25. Gardos G, Cole JO. The evaluation and treatment of neurolepticinduced movement disorders. Harvard Rev Psychiatry 1995;3:130-9.
26. Lohr JB, Caligiuri MP, Edson R, et al. Treatment predictors of extrapyramidal side effects in patients with tardive dyskinesia: results from Veterans Affairs Cooperative Study 394. J Clin Psychopharmacol 2002;22:196-200.
27. Casey DE. Effect of clozapine therapy in schizophrenic individuals at risk for tardive dyskinesia. J Clin Psychiatry 1998;59(suppl 3):31-7.
1. Woerner M, Kane JM, Lieberman JA, et al. The prevalence of tardive dyskinesia, J Clin Psychopharmacology 1991;11:34-42.
2. Caroff SN, Mann SC, Campbell EC, et al. Movement disorders associated with atypical antipsychotic drugs. J Clin Psychiatry 2002;63(suppl 4):12-19.
3. Tollefson GD, Beasley CM, Tran PV, et al. Olanzapine versus haloperidol in the treatment of schizophrenia and schizophreniform disorders: results of an international collaborative trial. Am J Psychiatry 1997;154:457-65.
4. Jeste DV, Lacro JP, Bailey A, et al. Lower incidence of tardive dyskinesia with risperidone compared with haloperidol in older patients. J Am Geriatr Soc 1999;47:716-19.
5. Kumet R, Freeman MP. Clozapine and tardive dyskinesia. J Clin Psychiatry 2002;63:167-8.
6. Hong KS, Cheong SS, Woo J-M, Kim E. Risperidone-induced tardive dyskinesia. Am J Psychiatry 1999;156:1290.-
7. Ghaemi SN, Ko JY. Quetiapine-related tardive dyskinesia. Am J Psychiatry 2001;158:1737.-
8. Kane JM. Tardive dyskinesia: epidemiological and clinical presentation. In: Bloom FE, Kupfer DJ (eds.) Psychopharmacology: The fourth generation of progress. New York: Raven Press, Ltd, 1995;1485-95.
9. Guy W. ECDEU assessment manual for psychopharmacology (rev. ed). Washington, DC: Department of Health, Education and Welfare, 1976.
10. Madhusoodanan S, Brenner R. Reversible choreiform dyskinesia and extrapyramidal symptoms associated with sertraline therapy. J Clin Psychopharmacology 1997;17:138-9.
11. Glazer WM, Morgenstern H, Pultz JA, et al. Incidence of tardive dyskinesia is lower with quetiapine treatment than with typical antipsychotics in patients with schizophrenia and schizo-affective disorder. Schizophrenia Res 2000;41:206-7.
12. Hirsch SR, Kissling W, Bauml J, et al. A 28-week comparison of ziprasidone and haloperidol in outpatients with stable schizophrenia. J Clin Psychiatry 2002;63:516-23.
13. Kujawa M, Sala A, Ingenito GG, et al. Aripiprazole for long-term maintenance treatment of schizophrenia (poster presentation). Montreal, Canada: Collegium Internationale Neuropsychopharmacologicum 23rd congress, June 23-27, 2002.
14. Gupta S, Mosnik D, Black DW, et al. Tardive dyskinesia: review of treatments past, present and future. Ann Clin Psychiatry 1999;11:257-66.
15. Suzuki E, Obata M, Yoshida Y, Miyaoka H. Tardive dyskinesia with risperidone and anticholinergics. Am J Psychiatry 2002;159:1948.-
16. Nasrallah HA. A review of the effect of atypical antipsychotics on weight. Psychoneuroendocrinology 2003;28(suppl 1):83-96.
17. Gardos G, Casey DE, Cole JO, et al. Ten-year outcome of tardive dyskinesia. Am J Psychiatry 1994;151:836-41.
18. Lieberman JA, Saltz BL, Johns CA, et al. The effects of clozapine on tardive dyskinesia. Br J Psychiatry 1991;158:503-10.
19. Tamminga CA, Thaker GK, Moran M, et al. Clozapine in tardive dyskinesia: observations from human and animal model studies J Clin Psychiatry 1994;55(suppl B):102-6.
20. Leucht S, Wahlbeck C, Hermann J, Kissling W. New-generation antipsychotics versus low-potency conventional antipsychotics: a systematic review and meta-analysis. Lancet 2003;361:1581-9.
21. Adler LA, Rotrosen J, Edson R, et al. Vitamin E treatment of tardive dyskinesia. Arch Gen Psychiatry 1999;56:836-41.
22. Shamir E, Barak Y, Shalman I, et al. Melatonin treatment for tardive dyskinesia. Arch Gen Psychiatry 2001;58:1046-52.
23. Glazer WM, Woods SW. Should Sisyphus have taken melatonin? Arch Gen Psychiatry 2001;58:1054-5.
24. Richardson MA, Bevans M, Read LL, et al. Efficacy of the branched-chain amino acids in the treatment of tardive dyskinesia in men. Am J Psychiatry 2003;160:1117-24.
25. Gardos G, Cole JO. The evaluation and treatment of neurolepticinduced movement disorders. Harvard Rev Psychiatry 1995;3:130-9.
26. Lohr JB, Caligiuri MP, Edson R, et al. Treatment predictors of extrapyramidal side effects in patients with tardive dyskinesia: results from Veterans Affairs Cooperative Study 394. J Clin Psychopharmacol 2002;22:196-200.
27. Casey DE. Effect of clozapine therapy in schizophrenic individuals at risk for tardive dyskinesia. J Clin Psychiatry 1998;59(suppl 3):31-7.
A low-frustration strategy for treating somatization
Mrs. M, age 34, was referred for psychiatric evaluation by her primary care physician. She reluctantly agreed to the referral and tells the psychiatrist she “really should be seeing a cardiologist.” Numerous evaluations for chest pain and palpitations—including seven emergency room visits, ECGs and cardiac catheterization—have revealed no medical pathology.
A divorced mother of two children, she says she feels anxious about her “heart condition.” Her father died of a heart attack at age 51. She experiences chest pains at home and at work, particularly when under stress. Sometimes she feels her heart racing and numbness or tingling in her arms.
Although her primary care physician has seen her frequently during the past 6 months, she says the doctor is not taking her complaints seriously. “These chest pains are real,” she says, “so don’t try to tell me they’re all in my head.”
Psychiatrists may be the last doctors patients such as Mrs. M wish to see but the ones best equipped to relieve their suffering. Our experience in treating somatizing patients and the available evidence suggest that cognitive-behavioral therapy (CBT) combined with psychoeducation, reassurance, and sometimes drug therapy is the most effective approach.
Health-related fear—or “illness worry”—is common, occurring in nearly 10% of adults who responded to a recent community survey.2 When this fear drives individuals to their physicians for evaluation, frequently no organic cause is discovered. Full evaluations are expensive and lead to increased use of health care resources, including potentially dangerous invasive testing.3,4
Defining somatization has been a source of confusion.5,6 Some authors consider somatic complaints to be expressions of suppressed psychosocial stressors. Others label them as medically unexplained complaints, although this definition fails to exclude occult medical problems. Kleinman7 defines somatization as “a somatic idiom of psychosocial distress in a setting of medical health-care seeking.” This useful definition links psychosocial problems with somatic complaints and the behavioral drive to obtain a medical evaluation.
In DSM-IV,8 the defining characteristics of somatoform disorders are somatic complaints or disease fears that are out of proportion with any identifiable somatic cause. Entities include somatization disorder, undifferentiated somatization disorder, conversion disorder, pain disorder, hypochondriasis, body dysmorphic disorder, and somatoform disorder–not otherwise specified (NOS).
Subthreshold symptoms. Unfortunately, DSM-IV’s categorization of Axis I somatoform disorders does not capture subthreshold presentations, which are common. Patients with less than the required number of somatic complaints are labeled in a wastebasket fashion with “undifferentiated somatoform disorder.”9
Mrs. M’s persistent chest pain of noncardiac origin is a familiar health anxiety, along with functional GI complaints, headaches, chronic fatigue, and lower back pain. Frustrating to their doctors and frustrated themselves, patients with medically unexplained complaints consume an inordinate amount of physicians’ time.1
Without a clear definition of somatization (Box)2-9 or useful clinical guidelines, psychiatrists must rely on the literature for guidance in managing somatization disorders. This article summarizes the evidence and describes how we apply these findings to practice. And when all else fails, we offer last-ditch advice for managing patients who resist your treatment efforts.
IDENTIFYING COMORBIDITIES
Identifying psychiatric comorbidities is the first step in successfully treating patients with somatoform complaints. In an epidemiologic study, 60% of patients with somatoform complaints also had a mood disorder and 48% had an anxiety disorder.10 In a similar study of patients with hypochondriasis, 88% also had one or more Axis I diagnosis.11
If a patient meets criteria for a comorbid psychiatric disorder and is willing to be treated for it, the somatic complaints may resolve along with the underlying disorder. In fact, the presence of an identifiable Axis I disorder order may predict a more positive prognosis.12
Personality disorders. Somatization in patients with a personality disorder poses unique challenges.13 Granted, when making a diagnosis it is difficult to tease apart somatization from personality disorders because somatization itself may be considered a chronic, maladaptive coping style. However, symptoms such as deception, impulsivity, mood lability, and self-injurious behavior introduce treatment complications that exceed the scope of this article.
Posttraumatic stress disorder (PTSD)—particularly childhood sexual and physical abuse—also predisposes some patients to somatization disorders.14,15 Patients with comorbid PTSD and somatization disorder require highly specialized treatment that is beyond the scope of this review.
COGNITIVE-BEHAVIORAL TREATMENT
Cognitive-behavioral therapy (CBT) is the best-studied and most effective treatment for somatoform disorders.16 CBT for somatization relies on both physiologic and cognitive explanations to account for the patient’s experience, without committing to an “either/or” dichotomy. It offers patients an alternate explanation of what is wrong with them—illness anxiety instead of severe physical illness.
By making patients aware of their automatic thoughts, feelings, behaviors, and underlying beliefs, CBT helps them normalize and cope with their illness anxiety. CBT techniques can be applied in a predetermined course of therapy (such as 12 sessions with a mental health clinician), in a group setting, or piecemeal by any health care provider.
Effective strategies. In a review of 30 controlled trials of CBT for somatoform disorders, Looper17 showed overall effect ranging from 0.38 to 2.5, where 0.2 was defined as a small effect, 0.5 as medium, and 0.8 as large. Hypochondriasis, somatization disorder, body dysmorphic disorder, chronic pain, chronic fatigue, and noncardiac chest pain were included in this review. The most effective strategies:
- included 6 to 16 treatment sessions
- were symptom-focused as opposed to providing general relaxation training
- included maintenance sessions after the initial series.
Four factors of health anxiety. CBT primarily targets the patient’s false beliefs that he or she is physically ill. These beliefs are based on how the patient misinterprets innocuous physical symptoms and responds to them.18 The cognitive theory of health anxiety holds that health anxiety severity is affected by four factors:
- perceived likelihood of illness
- perceived burden of illness
- perceived ability to cope with illness
- perception of the extent to which external factors will help.19
Table 1
Common dysfunctional beliefs of somatizing patients
|
The first two factors worsen and the latter two mitigate health anxiety. An individual patient’s presenting fears often suggest which factors to address. For example, Mrs. M may describe the burden of illness as the focus of her fears (“If I have a heart attack, who will care for my children?”). This information cues you to shift the focus of therapy to helping her cope with child care needs despite her recurring symptoms.
If she focuses on her likelihood of illness, then uncoupling the symptoms from the diagnosis could be more productive. When she reports palpitations, diaphoresis, and dizziness, have her do breathing exercises that induce those symptoms without producing a heart attack.
Table 2
Journaling homework: 5 questions for patients to answer about one symptom each day
|
She might describe feeling unable to cope when she feels symptoms or when cardiologists tell her nothing is wrong with her heart. In that case, focus on relaxation techniques, global stress reduction, and reducing cardiac risk factors to bolster her ability to cope with her illness.
Journaling is a critical component of CBT in treating somatization disorders. Regular journaling by the patient can reveal dysfunctional beliefs that may be driving his or her health anxieties, such as those listed in Table 1. We find it useful to assign patients to answer five questions about one symptom experience each day (Table 2). This self-monitoring provides material to work on with the patient during each session.
Cognitive restructuring. During therapy sessions, we ask patients to suggest alternate explanations for the symptoms recorded in their journals. We then ask them to determine which explanations are more feasible.
For example, if Mrs. M develops palpitations during emotionally charged arguments, we would ask her to develop explanations other than, “I was having a heart attack.” Reality testing includes rhetorical questions such as, “Would you be alive today if you were having a heart attack every time you had palpitations?” Automatic thoughts are successively identified and then tested aloud with the patient:
- “Has every unexplained symptom led to the discovery of a serious illness?”
- “Does every instance of hurt equal harm?”
Eventually, patterns of automatic thoughts emerge, and these reveal the underlying dysfunctional beliefs.
Dysfunctional beliefs are maintained when patients selectively attend to and amplify somatic sensations. Behavioral experiments during sessions can demonstrate to the patient in vivo the process by which they misattribute illness to physical symptoms. For example, overbreathing with a patient during a session may elicit light-headedness, paresthesias, or tachycardia, which can then be linked to overbreathing rather than a chronic or catastrophic illness.
Furthermore, patients can be taught to control the experience. Some patients with headaches or GI pain may be made aware of symptoms by simply asking them to focus their attention on the respective organs. Simply explaining the cycle of misattribution, autonomic activation, and further symptom development with an in vivo demonstration can be illuminating.
Response prevention. Another behavioral technique is to cut back in small increments on actions the patient takes in response to physical symptoms and automatic thoughts. For example, a patient could take medicine and seek reassurance less frequently and avoid rubbing the affected area.
PSYCHOEDUCATION
Two psychoeducation programs for somatization behavior have been formally studied.
The Personal Health Improvement Program20—led by trained facilitators—includes classroom videos, cognitive-behavioral exercises, and home study assignments. After completing the 6-week course, 171 patients with somatization disorders reported reduced physical and psychological distress and increased function. They also visited their primary care physicians less often.
Table 3
How to effectively reassure somatizing patients
| Action | Benefit |
|---|---|
| Review records in front of patients | Demonstrates that you take complaints and histories seriously |
| Acknowledge the severity of patients’ distress | Validates subjective suffering |
| Schedule follow-up visits at regular intervals | Provides access to you and continuity of care; reduces extra phone calls and emergency visits |
| Use clear and simple language | Improves communication |
| Explain that they do not have life-threatening structural disease | Opens door to cognitive restructuring |
| Assign jobs, such as journaling 15 min/day and rounding up medical records | Builds therapeutic alliance, fosters patient responsibility, and restores patients’ sense of control |
| Identify and support the patient’s strengths | Builds self-esteem |
| Use specialty referrals sparingly | Reduces risk of further medical testing and patient anxiety while awaiting results |
Coping with Illness Anxiety21 relies on mini-lectures, demonstrations, videos, and focused group discussions. After six 2-hour sessions, 33 of 43 study patients (78%) used medical services less often and reported reduced disease conviction, consequences of bodily complaints, health anxiety, and checking and avoidance behaviors. Two psychology graduate students taught the course from a manual, with 6 to 9 patients per group.
Psychoeducation in this context relies on didactic presentations, readings, role playing, and videotaped material. The goal is to teach patients to recognize thoughts, emotions, and behaviors that lead to and result from somatic preoccupation. Patients can improve when they recognize dysfunctional behavioral patterns and learn alternate coping strategies.
Somatizing patients—with their aversion to the stigma of mental illness—may find psychoeducation particularly attractive. They can be treated as students who are being educated, rather than as patients who are being treated. Classrooms in both studies cited above were located in medical outpatient offices, not in mental health facilities.
REASSURANCE
Reassurance is a common therapeutic technique in medicine, although it is poorly understood, poorly taught, and not methodically applied. Reassurance alleviates anxiety, enables patients to endure dysphoria, encourages hope, gives insight, and enhances the doctor-patient relationship.22
Table 4
How to avoid becoming frustrated with persistent somatization
| Situation | Response |
|---|---|
| Despite patients’ urgency | Watch and wait, knowing that psychological distress has been chronic |
| Despite patients’ belief that a single pill or procedure will ‘cure’ them | Persist in ‘rehabilitative’ approach |
| Despite patients’ provocations to force you to take a dichotomous approach | Persist in using both physical and psychological explanations |
| Despite your knowledge that patients are actively maintaining their illness beliefs | Try to be patient as they attribute their misfortune to ‘fate,’ ‘bad luck,’ or ‘misfortune’ |
| Despite the fact that you have agreed to treat the patient | Realize that his or her family or culture may reinforce the ‘sick role’ as the only acceptable form of distress |
| Despite patients’ desire to discuss symptoms | Reorient them to sustaining daily function (such as parenting while tolerating fatigue) |
Whereas CBT seeks to challenge patients’ underlying beliefs and restructure their thought processes,23 reassurance can help them tolerate their dysfunctional beliefs and dissuade them from believing their health is dangerously impaired. Reassurance offers a substitute explanation of patients’ dysfunction, although this explanation is not as central or detailed as it is in CBT.
How to reassure. Patients may consider reassurance offered prematurely or by a stranger to be patronizing or dismissive. Reassurance is most effective when:
- given by a trusted person who is reliable, consistent, firm, and empathic
- the patient’s condition has been established as unresponsive to conventional diagnostics or biological therapies.
Patients are most receptive to reassurance when they express distress or frustration with their unexplained symptoms. Affirming that their suffering is legitimate opens the door to further treatment.
Reassurance is least effective when a patient is expressing anger or mistrust, although this is when the physician may feel most pressured to reassure. To successfully reassure a patient, the psychiatrist needs to:
- credibly identify with the patient’s distress
- and listen empathically (such as using body language and facial expressions that convey concern and consideration to the patient).24
Starcevic suggests useful techniques for providing reassurance (Table 3).22
DRUG THERAPIES
Psychotropics are considered a first-line treatment for patients with somatization disorders when:
- the patient spontaneously identifies any discrete, vegetative, or psychological complaints that may respond to drug therapy, such as insomnia, weight loss, sadness, or preoccupation
- the patient meets diagnostic criteria for comorbid anxiety or depressive disorders
- the therapeutic alliance is strong enough to weather the inevitable struggle with side effects and incomplete response to treatment. We do not recommend medication in the first encounter, when it may threaten a nascent alliance.
A common obstacle to prescribing psychotropics to somatizing patients is their sensitivity to suggestions that their complaints are “all in their heads.” To sidestep this resistance, describe the medication as treating the stress caused. by—not causing.—their chronic physical complaints. Proposing antidepressant therapy after—rather than instead of—physical exams and other diagnostics may elicit a more positive response.
Antidepressants. In clinical trials, somatoform complaints show moderate improvement after antidepressant treatment. In a meta-analysis of 6,595 patients with unexplained symptoms treated only with antidepressants, the number needed to treat was 3 to yield a positive response.25 This report of 94 medication trials included patients with headache, fibromyalgia, functional GI syndromes, idiopathic pain, tinnitus, or chronic fatigue.
In other trials:
- Amitriptyline has reduced somatic symptoms in patients labeled as having “masked depression.”26
- Sertraline has reduced disease fear, disease conviction, and bodily preoccupation in patients with hypochondriasis and panic disorder.27
Consider side effects when choosing medication for patients with somatoform disorders. Selective serotonin reuptake inhibitors (SSRIs) in general—and sertraline, citalopram, and escitalopram specifically—have fewer side effects than tricyclics. The adage of “start low, go slow” is appropriate for somatizing patients; we usually start with one-half the dosages recommended for treating depression.
Antipsychotics. In case reports, patients with “atypical psychosis,” “monosymptomatic hypochondriacal psychosis,” or “delusional disorder, somatic type” have responded to antipsychotics. These patients’ somatic beliefs are of delusional intensity, such as the rare fear of being eaten alive by an intestinal parasite (delusional parasitosis). Reported behaviors associated with the delusion include starvation, excessive laxative abuse, ingestion of sharp objects, and self-inflicted stab wounds. Treatments described in the literature include the typical agents pimozide and haloperidol and the atypicals olanzapine and risperidone.
TREATMENT-RESISTANT PATIENTS
Some patients with somatoform disorders will not accept CBT, psychotropics, reassurance, or referrals to group psychoeducation. Despite your best efforts, they may persist in focusing on somatic complaints. If you are willing to maintain a therapeutic relationship with them, be prepared to tolerate several ongoing paradoxes (Table 4).
Behaviorally, you must “listen more and do less.” Emotionally, you must be willing to enter into a long-term relationship with an inherently frustrating patient whose pathologies make you feel therapeutically hopeless and helpless. Understand that their physical symptoms function as a metaphor for psychological distress. You are not required to explore the source, content, or meaning of the metaphor in detail but simply listen to their somatic complaints through that psychological filter.
Related resources
- Starcevic V, Lipsitt D (eds). Hypochondriasis: modern perspectives on an ancient malady. New York: Oxford University Press, 2001.
- Information and support Web site for persons with health anxiety or hypochondria. www.healthanxiety.com
- Anxiety Disorders Association of America. www.adaa.org
Drug brand names
- Amitriptyline • Elavil
- Citalopram • Celexa
- Escitalopram • Lexapro
- Haloperidol • Haldol
- Olanzapine • Zyprexa
- Pimozide • Orap
- Risperidone • Risperdal
- Sertraline • Zoloft
Disclosure
Dr. Isaac reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Dr. Wise receives grant support from Eli Lilly & Co. and is a consultant or speaker for Eli Lilly & Co., Pfizer Inc., Bristol-Myers Squibb Co., and GlaxoSmithKline.
1. Katon W, Von Korff M, Lin E, et al. Distressed high utilizers of medical care. DSM-III-R diagnoses and treatment needs. Gen Hosp Psychiatry 1990;12:355-62.
2. Noyes R, Jr, Happel RL, Yagla SJ. Correlates of hypochondriasis in a nonclinical population. Psychosomatics 1999;40:461-9.
3. Mayou R, Sprigings D, Gilbert T. Patients with palpitations referred for 24-hour ECG recording. J Psychosom Res 1999;46:491-6.
4. Mayou RA, Bass C, Hart G, et al. Can clinical assessment of chest pain be made more therapeutic? Q J Med 2000;93:805-11.
5. Lipowski ZJ. Somatization: the experience and communication of psychological distress as somatic symptoms. Psychother Psychosom 1987;47:160-7.
6. Lipowski ZJ. Somatization: medicine’s unsolved problem. Psychosomatics 1987;28(6):294-297.
7. Ware NC, Kleinman A. Culture and somatic experience: the social course of illness in neurasthenia and chronic fatigue syndrome. Psychosom Med 1992;54:546-60.
8. Diagnostic and statistical manual of mental disorders (4th ed., text revision). Washington, DC: American Psychiatric Association, 2000.
9. Bass C, Peveler R, House A. Somatoform disorders: severe psychiatric illnesses neglected by psychiatrists. Br J Psychiatry 2001;179:11-14.
10. Smith GR. The epidemiology and treatment of depression when it coexists with somatoform disorders, somatization, or pain. Gen Hosp Psychiatry 1992;14:265-72.
11. Barsky AJ, Wyshak G, Klerman GL. Psychiatric comorbidity in DSM-III-R hypochondriasis. Arch Gen Psychiatry 1992;49:101-8.
12. Starcevic V. Role of reassurance and psychopathology in hypochondriasis. Psychiatry 1990;53(4):383-95.
13. Rost KM, Akins RN, Brown FW, Smith GR. The comorbidity of DSM-III-R personality disorders in somatization disorder. Gen Hosp Psychiatry 1992;14:322-6.
14. Morrison J. Childhood sexual histories of women with somatization disorder [comment]. Am J Psychiatry 1989;146:239-41.
15. Morse DS, Suchman AL, Frankel RM. The meaning of symptoms in 10 women with somatization disorder and a history of childhood abuse. Arch Fam Med 1997;6:468-76.
16. Kroenke K, Swindle R. Cognitive-behavioral therapy for somatization and symptom syndromes: a critical review of controlled clinical trials. Psychother Psychosom 2000;69:205-15.
17. Looper KJ, Kirmayer LJ. Behavioral medicine approaches to somatoform disorders. J Consult Clin Psychol 2002;70:810-27.
18. Warwick HM, Clark DM, Cobb AM, Salkovskis PM. A controlled trial of cognitive-behavioural treatment of hypochondriasis. Br J Psychiatry 1996;169:189-95.
19. Warwick HM, Salkovskis PM. Cognitive-behavioral treatment of hypochondriasis. In: Lipsitt DR, Starcevic V (eds). Hypochondriasis: Modern perspectives on an ancient malady. New York: Oxford Press, 2001;314-28.
20. McLeod CC, Budd MA. Treatment of somatization in primary care: evaluation of the Personal Health Improvement Program. HMO Pract 1997;11:88-94.
21. Bouman TK, Visser S. Cognitive and behavioural treatment of hypochondriasis. Psychother Psychosom 1998;67:214-21.
22. Starcevic V. Reassurance in the treatment of hypochondriasis. In: Lipsitt DR, Starcevic V (eds). Hypochondriasis: Modern perspectives on an ancient malady. New York: Oxford Press, 2001;291-313.
23. Clark DM, Salkovskis PM, Hackmann A, et al. Two psychological treatments for hypochondriasis. A randomised controlled trial. Br J Psychiatry 1998;173:218-25.
24. Schwartz L. Some notes on reassurance in medical practice. Psychosomatics 1966;7:290-4.
25. O’Malley PG, Jackson JL, Santoro J, et al. Antidepressant therapy for unexplained symptoms and symptom syndromes. J Fam Pract 1999;48:980-90.
26. Kellner R, Fava GA, Lisansky J, et al. Hypochondriacal fears and beliefs in DSM-III melancholia. Changes with amitriptyline. J Affect Disord 1986;10:21-6.
27. Noyes R, Reich J, Clancy J, O’Gorman TW. Reduction in hypochondriasis with treatment of panic disorder. Br J Psychiatry 1986;149:631-5.
Mrs. M, age 34, was referred for psychiatric evaluation by her primary care physician. She reluctantly agreed to the referral and tells the psychiatrist she “really should be seeing a cardiologist.” Numerous evaluations for chest pain and palpitations—including seven emergency room visits, ECGs and cardiac catheterization—have revealed no medical pathology.
A divorced mother of two children, she says she feels anxious about her “heart condition.” Her father died of a heart attack at age 51. She experiences chest pains at home and at work, particularly when under stress. Sometimes she feels her heart racing and numbness or tingling in her arms.
Although her primary care physician has seen her frequently during the past 6 months, she says the doctor is not taking her complaints seriously. “These chest pains are real,” she says, “so don’t try to tell me they’re all in my head.”
Psychiatrists may be the last doctors patients such as Mrs. M wish to see but the ones best equipped to relieve their suffering. Our experience in treating somatizing patients and the available evidence suggest that cognitive-behavioral therapy (CBT) combined with psychoeducation, reassurance, and sometimes drug therapy is the most effective approach.
Health-related fear—or “illness worry”—is common, occurring in nearly 10% of adults who responded to a recent community survey.2 When this fear drives individuals to their physicians for evaluation, frequently no organic cause is discovered. Full evaluations are expensive and lead to increased use of health care resources, including potentially dangerous invasive testing.3,4
Defining somatization has been a source of confusion.5,6 Some authors consider somatic complaints to be expressions of suppressed psychosocial stressors. Others label them as medically unexplained complaints, although this definition fails to exclude occult medical problems. Kleinman7 defines somatization as “a somatic idiom of psychosocial distress in a setting of medical health-care seeking.” This useful definition links psychosocial problems with somatic complaints and the behavioral drive to obtain a medical evaluation.
In DSM-IV,8 the defining characteristics of somatoform disorders are somatic complaints or disease fears that are out of proportion with any identifiable somatic cause. Entities include somatization disorder, undifferentiated somatization disorder, conversion disorder, pain disorder, hypochondriasis, body dysmorphic disorder, and somatoform disorder–not otherwise specified (NOS).
Subthreshold symptoms. Unfortunately, DSM-IV’s categorization of Axis I somatoform disorders does not capture subthreshold presentations, which are common. Patients with less than the required number of somatic complaints are labeled in a wastebasket fashion with “undifferentiated somatoform disorder.”9
Mrs. M’s persistent chest pain of noncardiac origin is a familiar health anxiety, along with functional GI complaints, headaches, chronic fatigue, and lower back pain. Frustrating to their doctors and frustrated themselves, patients with medically unexplained complaints consume an inordinate amount of physicians’ time.1
Without a clear definition of somatization (Box)2-9 or useful clinical guidelines, psychiatrists must rely on the literature for guidance in managing somatization disorders. This article summarizes the evidence and describes how we apply these findings to practice. And when all else fails, we offer last-ditch advice for managing patients who resist your treatment efforts.
IDENTIFYING COMORBIDITIES
Identifying psychiatric comorbidities is the first step in successfully treating patients with somatoform complaints. In an epidemiologic study, 60% of patients with somatoform complaints also had a mood disorder and 48% had an anxiety disorder.10 In a similar study of patients with hypochondriasis, 88% also had one or more Axis I diagnosis.11
If a patient meets criteria for a comorbid psychiatric disorder and is willing to be treated for it, the somatic complaints may resolve along with the underlying disorder. In fact, the presence of an identifiable Axis I disorder order may predict a more positive prognosis.12
Personality disorders. Somatization in patients with a personality disorder poses unique challenges.13 Granted, when making a diagnosis it is difficult to tease apart somatization from personality disorders because somatization itself may be considered a chronic, maladaptive coping style. However, symptoms such as deception, impulsivity, mood lability, and self-injurious behavior introduce treatment complications that exceed the scope of this article.
Posttraumatic stress disorder (PTSD)—particularly childhood sexual and physical abuse—also predisposes some patients to somatization disorders.14,15 Patients with comorbid PTSD and somatization disorder require highly specialized treatment that is beyond the scope of this review.
COGNITIVE-BEHAVIORAL TREATMENT
Cognitive-behavioral therapy (CBT) is the best-studied and most effective treatment for somatoform disorders.16 CBT for somatization relies on both physiologic and cognitive explanations to account for the patient’s experience, without committing to an “either/or” dichotomy. It offers patients an alternate explanation of what is wrong with them—illness anxiety instead of severe physical illness.
By making patients aware of their automatic thoughts, feelings, behaviors, and underlying beliefs, CBT helps them normalize and cope with their illness anxiety. CBT techniques can be applied in a predetermined course of therapy (such as 12 sessions with a mental health clinician), in a group setting, or piecemeal by any health care provider.
Effective strategies. In a review of 30 controlled trials of CBT for somatoform disorders, Looper17 showed overall effect ranging from 0.38 to 2.5, where 0.2 was defined as a small effect, 0.5 as medium, and 0.8 as large. Hypochondriasis, somatization disorder, body dysmorphic disorder, chronic pain, chronic fatigue, and noncardiac chest pain were included in this review. The most effective strategies:
- included 6 to 16 treatment sessions
- were symptom-focused as opposed to providing general relaxation training
- included maintenance sessions after the initial series.
Four factors of health anxiety. CBT primarily targets the patient’s false beliefs that he or she is physically ill. These beliefs are based on how the patient misinterprets innocuous physical symptoms and responds to them.18 The cognitive theory of health anxiety holds that health anxiety severity is affected by four factors:
- perceived likelihood of illness
- perceived burden of illness
- perceived ability to cope with illness
- perception of the extent to which external factors will help.19
Table 1
Common dysfunctional beliefs of somatizing patients
|
The first two factors worsen and the latter two mitigate health anxiety. An individual patient’s presenting fears often suggest which factors to address. For example, Mrs. M may describe the burden of illness as the focus of her fears (“If I have a heart attack, who will care for my children?”). This information cues you to shift the focus of therapy to helping her cope with child care needs despite her recurring symptoms.
If she focuses on her likelihood of illness, then uncoupling the symptoms from the diagnosis could be more productive. When she reports palpitations, diaphoresis, and dizziness, have her do breathing exercises that induce those symptoms without producing a heart attack.
Table 2
Journaling homework: 5 questions for patients to answer about one symptom each day
|
She might describe feeling unable to cope when she feels symptoms or when cardiologists tell her nothing is wrong with her heart. In that case, focus on relaxation techniques, global stress reduction, and reducing cardiac risk factors to bolster her ability to cope with her illness.
Journaling is a critical component of CBT in treating somatization disorders. Regular journaling by the patient can reveal dysfunctional beliefs that may be driving his or her health anxieties, such as those listed in Table 1. We find it useful to assign patients to answer five questions about one symptom experience each day (Table 2). This self-monitoring provides material to work on with the patient during each session.
Cognitive restructuring. During therapy sessions, we ask patients to suggest alternate explanations for the symptoms recorded in their journals. We then ask them to determine which explanations are more feasible.
For example, if Mrs. M develops palpitations during emotionally charged arguments, we would ask her to develop explanations other than, “I was having a heart attack.” Reality testing includes rhetorical questions such as, “Would you be alive today if you were having a heart attack every time you had palpitations?” Automatic thoughts are successively identified and then tested aloud with the patient:
- “Has every unexplained symptom led to the discovery of a serious illness?”
- “Does every instance of hurt equal harm?”
Eventually, patterns of automatic thoughts emerge, and these reveal the underlying dysfunctional beliefs.
Dysfunctional beliefs are maintained when patients selectively attend to and amplify somatic sensations. Behavioral experiments during sessions can demonstrate to the patient in vivo the process by which they misattribute illness to physical symptoms. For example, overbreathing with a patient during a session may elicit light-headedness, paresthesias, or tachycardia, which can then be linked to overbreathing rather than a chronic or catastrophic illness.
Furthermore, patients can be taught to control the experience. Some patients with headaches or GI pain may be made aware of symptoms by simply asking them to focus their attention on the respective organs. Simply explaining the cycle of misattribution, autonomic activation, and further symptom development with an in vivo demonstration can be illuminating.
Response prevention. Another behavioral technique is to cut back in small increments on actions the patient takes in response to physical symptoms and automatic thoughts. For example, a patient could take medicine and seek reassurance less frequently and avoid rubbing the affected area.
PSYCHOEDUCATION
Two psychoeducation programs for somatization behavior have been formally studied.
The Personal Health Improvement Program20—led by trained facilitators—includes classroom videos, cognitive-behavioral exercises, and home study assignments. After completing the 6-week course, 171 patients with somatization disorders reported reduced physical and psychological distress and increased function. They also visited their primary care physicians less often.
Table 3
How to effectively reassure somatizing patients
| Action | Benefit |
|---|---|
| Review records in front of patients | Demonstrates that you take complaints and histories seriously |
| Acknowledge the severity of patients’ distress | Validates subjective suffering |
| Schedule follow-up visits at regular intervals | Provides access to you and continuity of care; reduces extra phone calls and emergency visits |
| Use clear and simple language | Improves communication |
| Explain that they do not have life-threatening structural disease | Opens door to cognitive restructuring |
| Assign jobs, such as journaling 15 min/day and rounding up medical records | Builds therapeutic alliance, fosters patient responsibility, and restores patients’ sense of control |
| Identify and support the patient’s strengths | Builds self-esteem |
| Use specialty referrals sparingly | Reduces risk of further medical testing and patient anxiety while awaiting results |
Coping with Illness Anxiety21 relies on mini-lectures, demonstrations, videos, and focused group discussions. After six 2-hour sessions, 33 of 43 study patients (78%) used medical services less often and reported reduced disease conviction, consequences of bodily complaints, health anxiety, and checking and avoidance behaviors. Two psychology graduate students taught the course from a manual, with 6 to 9 patients per group.
Psychoeducation in this context relies on didactic presentations, readings, role playing, and videotaped material. The goal is to teach patients to recognize thoughts, emotions, and behaviors that lead to and result from somatic preoccupation. Patients can improve when they recognize dysfunctional behavioral patterns and learn alternate coping strategies.
Somatizing patients—with their aversion to the stigma of mental illness—may find psychoeducation particularly attractive. They can be treated as students who are being educated, rather than as patients who are being treated. Classrooms in both studies cited above were located in medical outpatient offices, not in mental health facilities.
REASSURANCE
Reassurance is a common therapeutic technique in medicine, although it is poorly understood, poorly taught, and not methodically applied. Reassurance alleviates anxiety, enables patients to endure dysphoria, encourages hope, gives insight, and enhances the doctor-patient relationship.22
Table 4
How to avoid becoming frustrated with persistent somatization
| Situation | Response |
|---|---|
| Despite patients’ urgency | Watch and wait, knowing that psychological distress has been chronic |
| Despite patients’ belief that a single pill or procedure will ‘cure’ them | Persist in ‘rehabilitative’ approach |
| Despite patients’ provocations to force you to take a dichotomous approach | Persist in using both physical and psychological explanations |
| Despite your knowledge that patients are actively maintaining their illness beliefs | Try to be patient as they attribute their misfortune to ‘fate,’ ‘bad luck,’ or ‘misfortune’ |
| Despite the fact that you have agreed to treat the patient | Realize that his or her family or culture may reinforce the ‘sick role’ as the only acceptable form of distress |
| Despite patients’ desire to discuss symptoms | Reorient them to sustaining daily function (such as parenting while tolerating fatigue) |
Whereas CBT seeks to challenge patients’ underlying beliefs and restructure their thought processes,23 reassurance can help them tolerate their dysfunctional beliefs and dissuade them from believing their health is dangerously impaired. Reassurance offers a substitute explanation of patients’ dysfunction, although this explanation is not as central or detailed as it is in CBT.
How to reassure. Patients may consider reassurance offered prematurely or by a stranger to be patronizing or dismissive. Reassurance is most effective when:
- given by a trusted person who is reliable, consistent, firm, and empathic
- the patient’s condition has been established as unresponsive to conventional diagnostics or biological therapies.
Patients are most receptive to reassurance when they express distress or frustration with their unexplained symptoms. Affirming that their suffering is legitimate opens the door to further treatment.
Reassurance is least effective when a patient is expressing anger or mistrust, although this is when the physician may feel most pressured to reassure. To successfully reassure a patient, the psychiatrist needs to:
- credibly identify with the patient’s distress
- and listen empathically (such as using body language and facial expressions that convey concern and consideration to the patient).24
Starcevic suggests useful techniques for providing reassurance (Table 3).22
DRUG THERAPIES
Psychotropics are considered a first-line treatment for patients with somatization disorders when:
- the patient spontaneously identifies any discrete, vegetative, or psychological complaints that may respond to drug therapy, such as insomnia, weight loss, sadness, or preoccupation
- the patient meets diagnostic criteria for comorbid anxiety or depressive disorders
- the therapeutic alliance is strong enough to weather the inevitable struggle with side effects and incomplete response to treatment. We do not recommend medication in the first encounter, when it may threaten a nascent alliance.
A common obstacle to prescribing psychotropics to somatizing patients is their sensitivity to suggestions that their complaints are “all in their heads.” To sidestep this resistance, describe the medication as treating the stress caused. by—not causing.—their chronic physical complaints. Proposing antidepressant therapy after—rather than instead of—physical exams and other diagnostics may elicit a more positive response.
Antidepressants. In clinical trials, somatoform complaints show moderate improvement after antidepressant treatment. In a meta-analysis of 6,595 patients with unexplained symptoms treated only with antidepressants, the number needed to treat was 3 to yield a positive response.25 This report of 94 medication trials included patients with headache, fibromyalgia, functional GI syndromes, idiopathic pain, tinnitus, or chronic fatigue.
In other trials:
- Amitriptyline has reduced somatic symptoms in patients labeled as having “masked depression.”26
- Sertraline has reduced disease fear, disease conviction, and bodily preoccupation in patients with hypochondriasis and panic disorder.27
Consider side effects when choosing medication for patients with somatoform disorders. Selective serotonin reuptake inhibitors (SSRIs) in general—and sertraline, citalopram, and escitalopram specifically—have fewer side effects than tricyclics. The adage of “start low, go slow” is appropriate for somatizing patients; we usually start with one-half the dosages recommended for treating depression.
Antipsychotics. In case reports, patients with “atypical psychosis,” “monosymptomatic hypochondriacal psychosis,” or “delusional disorder, somatic type” have responded to antipsychotics. These patients’ somatic beliefs are of delusional intensity, such as the rare fear of being eaten alive by an intestinal parasite (delusional parasitosis). Reported behaviors associated with the delusion include starvation, excessive laxative abuse, ingestion of sharp objects, and self-inflicted stab wounds. Treatments described in the literature include the typical agents pimozide and haloperidol and the atypicals olanzapine and risperidone.
TREATMENT-RESISTANT PATIENTS
Some patients with somatoform disorders will not accept CBT, psychotropics, reassurance, or referrals to group psychoeducation. Despite your best efforts, they may persist in focusing on somatic complaints. If you are willing to maintain a therapeutic relationship with them, be prepared to tolerate several ongoing paradoxes (Table 4).
Behaviorally, you must “listen more and do less.” Emotionally, you must be willing to enter into a long-term relationship with an inherently frustrating patient whose pathologies make you feel therapeutically hopeless and helpless. Understand that their physical symptoms function as a metaphor for psychological distress. You are not required to explore the source, content, or meaning of the metaphor in detail but simply listen to their somatic complaints through that psychological filter.
Related resources
- Starcevic V, Lipsitt D (eds). Hypochondriasis: modern perspectives on an ancient malady. New York: Oxford University Press, 2001.
- Information and support Web site for persons with health anxiety or hypochondria. www.healthanxiety.com
- Anxiety Disorders Association of America. www.adaa.org
Drug brand names
- Amitriptyline • Elavil
- Citalopram • Celexa
- Escitalopram • Lexapro
- Haloperidol • Haldol
- Olanzapine • Zyprexa
- Pimozide • Orap
- Risperidone • Risperdal
- Sertraline • Zoloft
Disclosure
Dr. Isaac reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Dr. Wise receives grant support from Eli Lilly & Co. and is a consultant or speaker for Eli Lilly & Co., Pfizer Inc., Bristol-Myers Squibb Co., and GlaxoSmithKline.
Mrs. M, age 34, was referred for psychiatric evaluation by her primary care physician. She reluctantly agreed to the referral and tells the psychiatrist she “really should be seeing a cardiologist.” Numerous evaluations for chest pain and palpitations—including seven emergency room visits, ECGs and cardiac catheterization—have revealed no medical pathology.
A divorced mother of two children, she says she feels anxious about her “heart condition.” Her father died of a heart attack at age 51. She experiences chest pains at home and at work, particularly when under stress. Sometimes she feels her heart racing and numbness or tingling in her arms.
Although her primary care physician has seen her frequently during the past 6 months, she says the doctor is not taking her complaints seriously. “These chest pains are real,” she says, “so don’t try to tell me they’re all in my head.”
Psychiatrists may be the last doctors patients such as Mrs. M wish to see but the ones best equipped to relieve their suffering. Our experience in treating somatizing patients and the available evidence suggest that cognitive-behavioral therapy (CBT) combined with psychoeducation, reassurance, and sometimes drug therapy is the most effective approach.
Health-related fear—or “illness worry”—is common, occurring in nearly 10% of adults who responded to a recent community survey.2 When this fear drives individuals to their physicians for evaluation, frequently no organic cause is discovered. Full evaluations are expensive and lead to increased use of health care resources, including potentially dangerous invasive testing.3,4
Defining somatization has been a source of confusion.5,6 Some authors consider somatic complaints to be expressions of suppressed psychosocial stressors. Others label them as medically unexplained complaints, although this definition fails to exclude occult medical problems. Kleinman7 defines somatization as “a somatic idiom of psychosocial distress in a setting of medical health-care seeking.” This useful definition links psychosocial problems with somatic complaints and the behavioral drive to obtain a medical evaluation.
In DSM-IV,8 the defining characteristics of somatoform disorders are somatic complaints or disease fears that are out of proportion with any identifiable somatic cause. Entities include somatization disorder, undifferentiated somatization disorder, conversion disorder, pain disorder, hypochondriasis, body dysmorphic disorder, and somatoform disorder–not otherwise specified (NOS).
Subthreshold symptoms. Unfortunately, DSM-IV’s categorization of Axis I somatoform disorders does not capture subthreshold presentations, which are common. Patients with less than the required number of somatic complaints are labeled in a wastebasket fashion with “undifferentiated somatoform disorder.”9
Mrs. M’s persistent chest pain of noncardiac origin is a familiar health anxiety, along with functional GI complaints, headaches, chronic fatigue, and lower back pain. Frustrating to their doctors and frustrated themselves, patients with medically unexplained complaints consume an inordinate amount of physicians’ time.1
Without a clear definition of somatization (Box)2-9 or useful clinical guidelines, psychiatrists must rely on the literature for guidance in managing somatization disorders. This article summarizes the evidence and describes how we apply these findings to practice. And when all else fails, we offer last-ditch advice for managing patients who resist your treatment efforts.
IDENTIFYING COMORBIDITIES
Identifying psychiatric comorbidities is the first step in successfully treating patients with somatoform complaints. In an epidemiologic study, 60% of patients with somatoform complaints also had a mood disorder and 48% had an anxiety disorder.10 In a similar study of patients with hypochondriasis, 88% also had one or more Axis I diagnosis.11
If a patient meets criteria for a comorbid psychiatric disorder and is willing to be treated for it, the somatic complaints may resolve along with the underlying disorder. In fact, the presence of an identifiable Axis I disorder order may predict a more positive prognosis.12
Personality disorders. Somatization in patients with a personality disorder poses unique challenges.13 Granted, when making a diagnosis it is difficult to tease apart somatization from personality disorders because somatization itself may be considered a chronic, maladaptive coping style. However, symptoms such as deception, impulsivity, mood lability, and self-injurious behavior introduce treatment complications that exceed the scope of this article.
Posttraumatic stress disorder (PTSD)—particularly childhood sexual and physical abuse—also predisposes some patients to somatization disorders.14,15 Patients with comorbid PTSD and somatization disorder require highly specialized treatment that is beyond the scope of this review.
COGNITIVE-BEHAVIORAL TREATMENT
Cognitive-behavioral therapy (CBT) is the best-studied and most effective treatment for somatoform disorders.16 CBT for somatization relies on both physiologic and cognitive explanations to account for the patient’s experience, without committing to an “either/or” dichotomy. It offers patients an alternate explanation of what is wrong with them—illness anxiety instead of severe physical illness.
By making patients aware of their automatic thoughts, feelings, behaviors, and underlying beliefs, CBT helps them normalize and cope with their illness anxiety. CBT techniques can be applied in a predetermined course of therapy (such as 12 sessions with a mental health clinician), in a group setting, or piecemeal by any health care provider.
Effective strategies. In a review of 30 controlled trials of CBT for somatoform disorders, Looper17 showed overall effect ranging from 0.38 to 2.5, where 0.2 was defined as a small effect, 0.5 as medium, and 0.8 as large. Hypochondriasis, somatization disorder, body dysmorphic disorder, chronic pain, chronic fatigue, and noncardiac chest pain were included in this review. The most effective strategies:
- included 6 to 16 treatment sessions
- were symptom-focused as opposed to providing general relaxation training
- included maintenance sessions after the initial series.
Four factors of health anxiety. CBT primarily targets the patient’s false beliefs that he or she is physically ill. These beliefs are based on how the patient misinterprets innocuous physical symptoms and responds to them.18 The cognitive theory of health anxiety holds that health anxiety severity is affected by four factors:
- perceived likelihood of illness
- perceived burden of illness
- perceived ability to cope with illness
- perception of the extent to which external factors will help.19
Table 1
Common dysfunctional beliefs of somatizing patients
|
The first two factors worsen and the latter two mitigate health anxiety. An individual patient’s presenting fears often suggest which factors to address. For example, Mrs. M may describe the burden of illness as the focus of her fears (“If I have a heart attack, who will care for my children?”). This information cues you to shift the focus of therapy to helping her cope with child care needs despite her recurring symptoms.
If she focuses on her likelihood of illness, then uncoupling the symptoms from the diagnosis could be more productive. When she reports palpitations, diaphoresis, and dizziness, have her do breathing exercises that induce those symptoms without producing a heart attack.
Table 2
Journaling homework: 5 questions for patients to answer about one symptom each day
|
She might describe feeling unable to cope when she feels symptoms or when cardiologists tell her nothing is wrong with her heart. In that case, focus on relaxation techniques, global stress reduction, and reducing cardiac risk factors to bolster her ability to cope with her illness.
Journaling is a critical component of CBT in treating somatization disorders. Regular journaling by the patient can reveal dysfunctional beliefs that may be driving his or her health anxieties, such as those listed in Table 1. We find it useful to assign patients to answer five questions about one symptom experience each day (Table 2). This self-monitoring provides material to work on with the patient during each session.
Cognitive restructuring. During therapy sessions, we ask patients to suggest alternate explanations for the symptoms recorded in their journals. We then ask them to determine which explanations are more feasible.
For example, if Mrs. M develops palpitations during emotionally charged arguments, we would ask her to develop explanations other than, “I was having a heart attack.” Reality testing includes rhetorical questions such as, “Would you be alive today if you were having a heart attack every time you had palpitations?” Automatic thoughts are successively identified and then tested aloud with the patient:
- “Has every unexplained symptom led to the discovery of a serious illness?”
- “Does every instance of hurt equal harm?”
Eventually, patterns of automatic thoughts emerge, and these reveal the underlying dysfunctional beliefs.
Dysfunctional beliefs are maintained when patients selectively attend to and amplify somatic sensations. Behavioral experiments during sessions can demonstrate to the patient in vivo the process by which they misattribute illness to physical symptoms. For example, overbreathing with a patient during a session may elicit light-headedness, paresthesias, or tachycardia, which can then be linked to overbreathing rather than a chronic or catastrophic illness.
Furthermore, patients can be taught to control the experience. Some patients with headaches or GI pain may be made aware of symptoms by simply asking them to focus their attention on the respective organs. Simply explaining the cycle of misattribution, autonomic activation, and further symptom development with an in vivo demonstration can be illuminating.
Response prevention. Another behavioral technique is to cut back in small increments on actions the patient takes in response to physical symptoms and automatic thoughts. For example, a patient could take medicine and seek reassurance less frequently and avoid rubbing the affected area.
PSYCHOEDUCATION
Two psychoeducation programs for somatization behavior have been formally studied.
The Personal Health Improvement Program20—led by trained facilitators—includes classroom videos, cognitive-behavioral exercises, and home study assignments. After completing the 6-week course, 171 patients with somatization disorders reported reduced physical and psychological distress and increased function. They also visited their primary care physicians less often.
Table 3
How to effectively reassure somatizing patients
| Action | Benefit |
|---|---|
| Review records in front of patients | Demonstrates that you take complaints and histories seriously |
| Acknowledge the severity of patients’ distress | Validates subjective suffering |
| Schedule follow-up visits at regular intervals | Provides access to you and continuity of care; reduces extra phone calls and emergency visits |
| Use clear and simple language | Improves communication |
| Explain that they do not have life-threatening structural disease | Opens door to cognitive restructuring |
| Assign jobs, such as journaling 15 min/day and rounding up medical records | Builds therapeutic alliance, fosters patient responsibility, and restores patients’ sense of control |
| Identify and support the patient’s strengths | Builds self-esteem |
| Use specialty referrals sparingly | Reduces risk of further medical testing and patient anxiety while awaiting results |
Coping with Illness Anxiety21 relies on mini-lectures, demonstrations, videos, and focused group discussions. After six 2-hour sessions, 33 of 43 study patients (78%) used medical services less often and reported reduced disease conviction, consequences of bodily complaints, health anxiety, and checking and avoidance behaviors. Two psychology graduate students taught the course from a manual, with 6 to 9 patients per group.
Psychoeducation in this context relies on didactic presentations, readings, role playing, and videotaped material. The goal is to teach patients to recognize thoughts, emotions, and behaviors that lead to and result from somatic preoccupation. Patients can improve when they recognize dysfunctional behavioral patterns and learn alternate coping strategies.
Somatizing patients—with their aversion to the stigma of mental illness—may find psychoeducation particularly attractive. They can be treated as students who are being educated, rather than as patients who are being treated. Classrooms in both studies cited above were located in medical outpatient offices, not in mental health facilities.
REASSURANCE
Reassurance is a common therapeutic technique in medicine, although it is poorly understood, poorly taught, and not methodically applied. Reassurance alleviates anxiety, enables patients to endure dysphoria, encourages hope, gives insight, and enhances the doctor-patient relationship.22
Table 4
How to avoid becoming frustrated with persistent somatization
| Situation | Response |
|---|---|
| Despite patients’ urgency | Watch and wait, knowing that psychological distress has been chronic |
| Despite patients’ belief that a single pill or procedure will ‘cure’ them | Persist in ‘rehabilitative’ approach |
| Despite patients’ provocations to force you to take a dichotomous approach | Persist in using both physical and psychological explanations |
| Despite your knowledge that patients are actively maintaining their illness beliefs | Try to be patient as they attribute their misfortune to ‘fate,’ ‘bad luck,’ or ‘misfortune’ |
| Despite the fact that you have agreed to treat the patient | Realize that his or her family or culture may reinforce the ‘sick role’ as the only acceptable form of distress |
| Despite patients’ desire to discuss symptoms | Reorient them to sustaining daily function (such as parenting while tolerating fatigue) |
Whereas CBT seeks to challenge patients’ underlying beliefs and restructure their thought processes,23 reassurance can help them tolerate their dysfunctional beliefs and dissuade them from believing their health is dangerously impaired. Reassurance offers a substitute explanation of patients’ dysfunction, although this explanation is not as central or detailed as it is in CBT.
How to reassure. Patients may consider reassurance offered prematurely or by a stranger to be patronizing or dismissive. Reassurance is most effective when:
- given by a trusted person who is reliable, consistent, firm, and empathic
- the patient’s condition has been established as unresponsive to conventional diagnostics or biological therapies.
Patients are most receptive to reassurance when they express distress or frustration with their unexplained symptoms. Affirming that their suffering is legitimate opens the door to further treatment.
Reassurance is least effective when a patient is expressing anger or mistrust, although this is when the physician may feel most pressured to reassure. To successfully reassure a patient, the psychiatrist needs to:
- credibly identify with the patient’s distress
- and listen empathically (such as using body language and facial expressions that convey concern and consideration to the patient).24
Starcevic suggests useful techniques for providing reassurance (Table 3).22
DRUG THERAPIES
Psychotropics are considered a first-line treatment for patients with somatization disorders when:
- the patient spontaneously identifies any discrete, vegetative, or psychological complaints that may respond to drug therapy, such as insomnia, weight loss, sadness, or preoccupation
- the patient meets diagnostic criteria for comorbid anxiety or depressive disorders
- the therapeutic alliance is strong enough to weather the inevitable struggle with side effects and incomplete response to treatment. We do not recommend medication in the first encounter, when it may threaten a nascent alliance.
A common obstacle to prescribing psychotropics to somatizing patients is their sensitivity to suggestions that their complaints are “all in their heads.” To sidestep this resistance, describe the medication as treating the stress caused. by—not causing.—their chronic physical complaints. Proposing antidepressant therapy after—rather than instead of—physical exams and other diagnostics may elicit a more positive response.
Antidepressants. In clinical trials, somatoform complaints show moderate improvement after antidepressant treatment. In a meta-analysis of 6,595 patients with unexplained symptoms treated only with antidepressants, the number needed to treat was 3 to yield a positive response.25 This report of 94 medication trials included patients with headache, fibromyalgia, functional GI syndromes, idiopathic pain, tinnitus, or chronic fatigue.
In other trials:
- Amitriptyline has reduced somatic symptoms in patients labeled as having “masked depression.”26
- Sertraline has reduced disease fear, disease conviction, and bodily preoccupation in patients with hypochondriasis and panic disorder.27
Consider side effects when choosing medication for patients with somatoform disorders. Selective serotonin reuptake inhibitors (SSRIs) in general—and sertraline, citalopram, and escitalopram specifically—have fewer side effects than tricyclics. The adage of “start low, go slow” is appropriate for somatizing patients; we usually start with one-half the dosages recommended for treating depression.
Antipsychotics. In case reports, patients with “atypical psychosis,” “monosymptomatic hypochondriacal psychosis,” or “delusional disorder, somatic type” have responded to antipsychotics. These patients’ somatic beliefs are of delusional intensity, such as the rare fear of being eaten alive by an intestinal parasite (delusional parasitosis). Reported behaviors associated with the delusion include starvation, excessive laxative abuse, ingestion of sharp objects, and self-inflicted stab wounds. Treatments described in the literature include the typical agents pimozide and haloperidol and the atypicals olanzapine and risperidone.
TREATMENT-RESISTANT PATIENTS
Some patients with somatoform disorders will not accept CBT, psychotropics, reassurance, or referrals to group psychoeducation. Despite your best efforts, they may persist in focusing on somatic complaints. If you are willing to maintain a therapeutic relationship with them, be prepared to tolerate several ongoing paradoxes (Table 4).
Behaviorally, you must “listen more and do less.” Emotionally, you must be willing to enter into a long-term relationship with an inherently frustrating patient whose pathologies make you feel therapeutically hopeless and helpless. Understand that their physical symptoms function as a metaphor for psychological distress. You are not required to explore the source, content, or meaning of the metaphor in detail but simply listen to their somatic complaints through that psychological filter.
Related resources
- Starcevic V, Lipsitt D (eds). Hypochondriasis: modern perspectives on an ancient malady. New York: Oxford University Press, 2001.
- Information and support Web site for persons with health anxiety or hypochondria. www.healthanxiety.com
- Anxiety Disorders Association of America. www.adaa.org
Drug brand names
- Amitriptyline • Elavil
- Citalopram • Celexa
- Escitalopram • Lexapro
- Haloperidol • Haldol
- Olanzapine • Zyprexa
- Pimozide • Orap
- Risperidone • Risperdal
- Sertraline • Zoloft
Disclosure
Dr. Isaac reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Dr. Wise receives grant support from Eli Lilly & Co. and is a consultant or speaker for Eli Lilly & Co., Pfizer Inc., Bristol-Myers Squibb Co., and GlaxoSmithKline.
1. Katon W, Von Korff M, Lin E, et al. Distressed high utilizers of medical care. DSM-III-R diagnoses and treatment needs. Gen Hosp Psychiatry 1990;12:355-62.
2. Noyes R, Jr, Happel RL, Yagla SJ. Correlates of hypochondriasis in a nonclinical population. Psychosomatics 1999;40:461-9.
3. Mayou R, Sprigings D, Gilbert T. Patients with palpitations referred for 24-hour ECG recording. J Psychosom Res 1999;46:491-6.
4. Mayou RA, Bass C, Hart G, et al. Can clinical assessment of chest pain be made more therapeutic? Q J Med 2000;93:805-11.
5. Lipowski ZJ. Somatization: the experience and communication of psychological distress as somatic symptoms. Psychother Psychosom 1987;47:160-7.
6. Lipowski ZJ. Somatization: medicine’s unsolved problem. Psychosomatics 1987;28(6):294-297.
7. Ware NC, Kleinman A. Culture and somatic experience: the social course of illness in neurasthenia and chronic fatigue syndrome. Psychosom Med 1992;54:546-60.
8. Diagnostic and statistical manual of mental disorders (4th ed., text revision). Washington, DC: American Psychiatric Association, 2000.
9. Bass C, Peveler R, House A. Somatoform disorders: severe psychiatric illnesses neglected by psychiatrists. Br J Psychiatry 2001;179:11-14.
10. Smith GR. The epidemiology and treatment of depression when it coexists with somatoform disorders, somatization, or pain. Gen Hosp Psychiatry 1992;14:265-72.
11. Barsky AJ, Wyshak G, Klerman GL. Psychiatric comorbidity in DSM-III-R hypochondriasis. Arch Gen Psychiatry 1992;49:101-8.
12. Starcevic V. Role of reassurance and psychopathology in hypochondriasis. Psychiatry 1990;53(4):383-95.
13. Rost KM, Akins RN, Brown FW, Smith GR. The comorbidity of DSM-III-R personality disorders in somatization disorder. Gen Hosp Psychiatry 1992;14:322-6.
14. Morrison J. Childhood sexual histories of women with somatization disorder [comment]. Am J Psychiatry 1989;146:239-41.
15. Morse DS, Suchman AL, Frankel RM. The meaning of symptoms in 10 women with somatization disorder and a history of childhood abuse. Arch Fam Med 1997;6:468-76.
16. Kroenke K, Swindle R. Cognitive-behavioral therapy for somatization and symptom syndromes: a critical review of controlled clinical trials. Psychother Psychosom 2000;69:205-15.
17. Looper KJ, Kirmayer LJ. Behavioral medicine approaches to somatoform disorders. J Consult Clin Psychol 2002;70:810-27.
18. Warwick HM, Clark DM, Cobb AM, Salkovskis PM. A controlled trial of cognitive-behavioural treatment of hypochondriasis. Br J Psychiatry 1996;169:189-95.
19. Warwick HM, Salkovskis PM. Cognitive-behavioral treatment of hypochondriasis. In: Lipsitt DR, Starcevic V (eds). Hypochondriasis: Modern perspectives on an ancient malady. New York: Oxford Press, 2001;314-28.
20. McLeod CC, Budd MA. Treatment of somatization in primary care: evaluation of the Personal Health Improvement Program. HMO Pract 1997;11:88-94.
21. Bouman TK, Visser S. Cognitive and behavioural treatment of hypochondriasis. Psychother Psychosom 1998;67:214-21.
22. Starcevic V. Reassurance in the treatment of hypochondriasis. In: Lipsitt DR, Starcevic V (eds). Hypochondriasis: Modern perspectives on an ancient malady. New York: Oxford Press, 2001;291-313.
23. Clark DM, Salkovskis PM, Hackmann A, et al. Two psychological treatments for hypochondriasis. A randomised controlled trial. Br J Psychiatry 1998;173:218-25.
24. Schwartz L. Some notes on reassurance in medical practice. Psychosomatics 1966;7:290-4.
25. O’Malley PG, Jackson JL, Santoro J, et al. Antidepressant therapy for unexplained symptoms and symptom syndromes. J Fam Pract 1999;48:980-90.
26. Kellner R, Fava GA, Lisansky J, et al. Hypochondriacal fears and beliefs in DSM-III melancholia. Changes with amitriptyline. J Affect Disord 1986;10:21-6.
27. Noyes R, Reich J, Clancy J, O’Gorman TW. Reduction in hypochondriasis with treatment of panic disorder. Br J Psychiatry 1986;149:631-5.
1. Katon W, Von Korff M, Lin E, et al. Distressed high utilizers of medical care. DSM-III-R diagnoses and treatment needs. Gen Hosp Psychiatry 1990;12:355-62.
2. Noyes R, Jr, Happel RL, Yagla SJ. Correlates of hypochondriasis in a nonclinical population. Psychosomatics 1999;40:461-9.
3. Mayou R, Sprigings D, Gilbert T. Patients with palpitations referred for 24-hour ECG recording. J Psychosom Res 1999;46:491-6.
4. Mayou RA, Bass C, Hart G, et al. Can clinical assessment of chest pain be made more therapeutic? Q J Med 2000;93:805-11.
5. Lipowski ZJ. Somatization: the experience and communication of psychological distress as somatic symptoms. Psychother Psychosom 1987;47:160-7.
6. Lipowski ZJ. Somatization: medicine’s unsolved problem. Psychosomatics 1987;28(6):294-297.
7. Ware NC, Kleinman A. Culture and somatic experience: the social course of illness in neurasthenia and chronic fatigue syndrome. Psychosom Med 1992;54:546-60.
8. Diagnostic and statistical manual of mental disorders (4th ed., text revision). Washington, DC: American Psychiatric Association, 2000.
9. Bass C, Peveler R, House A. Somatoform disorders: severe psychiatric illnesses neglected by psychiatrists. Br J Psychiatry 2001;179:11-14.
10. Smith GR. The epidemiology and treatment of depression when it coexists with somatoform disorders, somatization, or pain. Gen Hosp Psychiatry 1992;14:265-72.
11. Barsky AJ, Wyshak G, Klerman GL. Psychiatric comorbidity in DSM-III-R hypochondriasis. Arch Gen Psychiatry 1992;49:101-8.
12. Starcevic V. Role of reassurance and psychopathology in hypochondriasis. Psychiatry 1990;53(4):383-95.
13. Rost KM, Akins RN, Brown FW, Smith GR. The comorbidity of DSM-III-R personality disorders in somatization disorder. Gen Hosp Psychiatry 1992;14:322-6.
14. Morrison J. Childhood sexual histories of women with somatization disorder [comment]. Am J Psychiatry 1989;146:239-41.
15. Morse DS, Suchman AL, Frankel RM. The meaning of symptoms in 10 women with somatization disorder and a history of childhood abuse. Arch Fam Med 1997;6:468-76.
16. Kroenke K, Swindle R. Cognitive-behavioral therapy for somatization and symptom syndromes: a critical review of controlled clinical trials. Psychother Psychosom 2000;69:205-15.
17. Looper KJ, Kirmayer LJ. Behavioral medicine approaches to somatoform disorders. J Consult Clin Psychol 2002;70:810-27.
18. Warwick HM, Clark DM, Cobb AM, Salkovskis PM. A controlled trial of cognitive-behavioural treatment of hypochondriasis. Br J Psychiatry 1996;169:189-95.
19. Warwick HM, Salkovskis PM. Cognitive-behavioral treatment of hypochondriasis. In: Lipsitt DR, Starcevic V (eds). Hypochondriasis: Modern perspectives on an ancient malady. New York: Oxford Press, 2001;314-28.
20. McLeod CC, Budd MA. Treatment of somatization in primary care: evaluation of the Personal Health Improvement Program. HMO Pract 1997;11:88-94.
21. Bouman TK, Visser S. Cognitive and behavioural treatment of hypochondriasis. Psychother Psychosom 1998;67:214-21.
22. Starcevic V. Reassurance in the treatment of hypochondriasis. In: Lipsitt DR, Starcevic V (eds). Hypochondriasis: Modern perspectives on an ancient malady. New York: Oxford Press, 2001;291-313.
23. Clark DM, Salkovskis PM, Hackmann A, et al. Two psychological treatments for hypochondriasis. A randomised controlled trial. Br J Psychiatry 1998;173:218-25.
24. Schwartz L. Some notes on reassurance in medical practice. Psychosomatics 1966;7:290-4.
25. O’Malley PG, Jackson JL, Santoro J, et al. Antidepressant therapy for unexplained symptoms and symptom syndromes. J Fam Pract 1999;48:980-90.
26. Kellner R, Fava GA, Lisansky J, et al. Hypochondriacal fears and beliefs in DSM-III melancholia. Changes with amitriptyline. J Affect Disord 1986;10:21-6.
27. Noyes R, Reich J, Clancy J, O’Gorman TW. Reduction in hypochondriasis with treatment of panic disorder. Br J Psychiatry 1986;149:631-5.
Visual hallucinations and drug therapy
In your “Cases That Test Your Skills” article on Charles Bonnet syndrome (CBS) (Current Psychiatry, May 2003), the authors briefly mention substance-induced psychosis in the differential diagnosis of visual hallucinations.
Ms. K was taking two antimuscarinic drugs, tolterodine and oxybutynin; I would not have accepted CBS as a diagnosis until the role of these drugs was clarified.
One patient developed visual hallucinations when her urologist started her on imipramine for urinary incontinence. Interestingly, once she was reassured that imipramine was causing this effect, she chose to keep taking it. She preferred occasional hallucinations to incontinence.
William Braden MD
Providence, RI
The authors did not adequately consider anticholinergic toxicity from a longstanding medication regimen as a possible cause of Ms. K’s visual hallucinations.
It is true that Ms. K had been taking tolterodine and oxybutynin, both anticholinergic agents, for more than 2 years without apparent adverse effects. However, that does not rule out the possibility that her advancing age and other factors increased her vulnerability to such toxicity. While her normal Mini-Mental State Examination score and clear sensorium do rule out frank delirium, anticholinergic toxicity can occur without gross cognitive impairment.
Robert L. Marcus, MD
Dix Hills, NY
The author responds
Drs. Braden and Marcus raise valid concerns about the effects of tolterodine and oxybutynin on Ms. K’s visual hallucinations. Benzodiazepines, tricyclic antidepressants, analgesics, beta-blockers, and antimuscarinics have all been linked to substance-induced psychosis, particularly in older patients.
We felt comfortable excluding these medications as playing a role in Ms. K’s visual hallucinations. She had tolerated these agents well with no changes for more than 2 years, so visual hallucinations as a sudden adverse effect seemed unlikely. Looking back, maybe anticholinergic toxicity could have been considered as a possible cause.
Drs. Braden and Marcus remind us that when evaluating apparent psychosis in older psychiatric patients, we should rule out causes such as medications and illegal substances. When alternative explanations are exhausted, however, a diagnosis of CBS may be warranted in the visually impaired patient who retains cognitive function.
Lee I. Kubersky
Third-year medical student
University of Medicine and Dentistry of New Jersey
Cooper Hospital/University Medical Center
Camden, NJ
In your “Cases That Test Your Skills” article on Charles Bonnet syndrome (CBS) (Current Psychiatry, May 2003), the authors briefly mention substance-induced psychosis in the differential diagnosis of visual hallucinations.
Ms. K was taking two antimuscarinic drugs, tolterodine and oxybutynin; I would not have accepted CBS as a diagnosis until the role of these drugs was clarified.
One patient developed visual hallucinations when her urologist started her on imipramine for urinary incontinence. Interestingly, once she was reassured that imipramine was causing this effect, she chose to keep taking it. She preferred occasional hallucinations to incontinence.
William Braden MD
Providence, RI
The authors did not adequately consider anticholinergic toxicity from a longstanding medication regimen as a possible cause of Ms. K’s visual hallucinations.
It is true that Ms. K had been taking tolterodine and oxybutynin, both anticholinergic agents, for more than 2 years without apparent adverse effects. However, that does not rule out the possibility that her advancing age and other factors increased her vulnerability to such toxicity. While her normal Mini-Mental State Examination score and clear sensorium do rule out frank delirium, anticholinergic toxicity can occur without gross cognitive impairment.
Robert L. Marcus, MD
Dix Hills, NY
The author responds
Drs. Braden and Marcus raise valid concerns about the effects of tolterodine and oxybutynin on Ms. K’s visual hallucinations. Benzodiazepines, tricyclic antidepressants, analgesics, beta-blockers, and antimuscarinics have all been linked to substance-induced psychosis, particularly in older patients.
We felt comfortable excluding these medications as playing a role in Ms. K’s visual hallucinations. She had tolerated these agents well with no changes for more than 2 years, so visual hallucinations as a sudden adverse effect seemed unlikely. Looking back, maybe anticholinergic toxicity could have been considered as a possible cause.
Drs. Braden and Marcus remind us that when evaluating apparent psychosis in older psychiatric patients, we should rule out causes such as medications and illegal substances. When alternative explanations are exhausted, however, a diagnosis of CBS may be warranted in the visually impaired patient who retains cognitive function.
Lee I. Kubersky
Third-year medical student
University of Medicine and Dentistry of New Jersey
Cooper Hospital/University Medical Center
Camden, NJ
In your “Cases That Test Your Skills” article on Charles Bonnet syndrome (CBS) (Current Psychiatry, May 2003), the authors briefly mention substance-induced psychosis in the differential diagnosis of visual hallucinations.
Ms. K was taking two antimuscarinic drugs, tolterodine and oxybutynin; I would not have accepted CBS as a diagnosis until the role of these drugs was clarified.
One patient developed visual hallucinations when her urologist started her on imipramine for urinary incontinence. Interestingly, once she was reassured that imipramine was causing this effect, she chose to keep taking it. She preferred occasional hallucinations to incontinence.
William Braden MD
Providence, RI
The authors did not adequately consider anticholinergic toxicity from a longstanding medication regimen as a possible cause of Ms. K’s visual hallucinations.
It is true that Ms. K had been taking tolterodine and oxybutynin, both anticholinergic agents, for more than 2 years without apparent adverse effects. However, that does not rule out the possibility that her advancing age and other factors increased her vulnerability to such toxicity. While her normal Mini-Mental State Examination score and clear sensorium do rule out frank delirium, anticholinergic toxicity can occur without gross cognitive impairment.
Robert L. Marcus, MD
Dix Hills, NY
The author responds
Drs. Braden and Marcus raise valid concerns about the effects of tolterodine and oxybutynin on Ms. K’s visual hallucinations. Benzodiazepines, tricyclic antidepressants, analgesics, beta-blockers, and antimuscarinics have all been linked to substance-induced psychosis, particularly in older patients.
We felt comfortable excluding these medications as playing a role in Ms. K’s visual hallucinations. She had tolerated these agents well with no changes for more than 2 years, so visual hallucinations as a sudden adverse effect seemed unlikely. Looking back, maybe anticholinergic toxicity could have been considered as a possible cause.
Drs. Braden and Marcus remind us that when evaluating apparent psychosis in older psychiatric patients, we should rule out causes such as medications and illegal substances. When alternative explanations are exhausted, however, a diagnosis of CBS may be warranted in the visually impaired patient who retains cognitive function.
Lee I. Kubersky
Third-year medical student
University of Medicine and Dentistry of New Jersey
Cooper Hospital/University Medical Center
Camden, NJ
Irritable bowel syndrome and psychiatric illness: Three clinical challenges
Psychiatrists often treat patients with irritable bowel syndrome (IBS) and an accompanying mental illness. Knowledge of available treatments and communication with the referring doctor are crucial to treating both the IBS symptoms and the comorbidity.
This article presents three cases that illustrate the challenges of identifying target symptoms, avoiding drug-drug interactions, ruling out serious underlying medical problems, and formulating treatment.
WHO GETS IBS?
Approximately 12% of the United States population reports IBS symptoms (abdominal pain, bloating, altered bowel habits).1 These symptoms begin before age 35 in most patients and during childhood in some. Onset after age 65 is rare.
IBS is common among patients with alcohol abuse disorder (32%),2 chronic fatigue syndrome (92%), fibromyalgia (77%), or temporomandibular joint syndrome (64%).3 Seventy percent of patients with IBS are women.4 Chronic pelvic pain, dyspareunia, dysmenorrhea, or a history of abdominal surgeries are risk factors for IBS in women.
LINK BETWEEN IBS AND MENTAL ILLNESS
Although mental illness often coexists with IBS, no cause-effect relationship has been shown.5
IBS is often preceded by stressful life events, such as family death or divorce,3 and some believe IBS is a precursor to numerous psychiatric disorders. Generalized anxiety disorder, major depression, panic disorder, social phobia, somatization disorder, or dysthymia have been diagnosed in most IBS patients.2
CASE 1: IBS AND DEPRESSION
Ms. R, age 55, has had IBS for 10 years. She has occasional diarrhea and abdominal cramps relieved by bowel movements. She is taking a bulking agent but still sometimes suffers abdominal pain.
She is referred to a psychiatrist after complaining of fatigue, loss of interest in hobbies, and crying spells for 2 months. She denies suicidal ideations. Her referring physician reports that she is taking conjugated estrogens to manage menopause symptoms. She denies any recent stressful life events. Thyroid function, glucose, and CBC are normal.
The challenge: Deciding which to treat first—the IBS symptoms or the depression—and how.
Discussion: The predominant symptom (in Ms. R’s case, abdominal pain) can help determine choice of medication. Bulk-forming agents, antispasmodics, barbiturates, benzodiazepines, and serotonin reuptake inhibitors have historically been used to treat IBS,6 but scant evidence supports their use.
Obtaining a thorough prescription history from the primary care physician, OB/GYN, and other treatment team members is critical before formulating a treatment plan. Ms. R’s estrogen use will not affect the choice of psychotropic or IBS medication because there are no significant interactions between estrogen and these classes of drugs.
Ms. R’s abdominal pain and depression can be treated simultaneously. Randomized, controlled trials have demonstrated that tricyclic antidepressants reduce abdominal pain and that behavioral therapy (relaxation therapy, hypnotherapy, and cognitive-behavioral therapy) may relieve individual IBS symptoms.7
Case 1 concluded: After reviewing Ms. R’s medications, the psychiatrist starts:
- desipramine, 50 mg at bedtime, to minimize anticholinergic side effects
- and short-term psychotherapy, which helped her identify support mechanisms and ways to better balance her life stresses.
After 6 weeks, her Beck Depression Inventory score improved from 30 at baseline to 8. She reports her abdominal pain is “the best it has been in 10 years.” Six months after diagnosis, she continues to take desipramine and is doing well.
CASE 2: IBS, DEPRESSION, AND PSYCHOSIS
Ms. H, age 32, is referred to a psychiatrist for treatment of depression with paranoid features.
Four years ago, a gastroenterologist diagnosed her as having IBS. She experiences frequent diarrhea and lower abdominal cramping. For 2 years she has been taking the antimuscarinic dicyclomine, 10 mg tid, which has provided some relief from her cramps. An estimated 20 diarrhea attacks per day leaves her housebound much of the time, however.
She reports fatigue, loss of interest in hobbies across 2 months, and paranoid thinking. She denies hallucinations or delusions but believes that her teenage children are discussing her “sickness” and plotting to “drive her crazy.” She is not suicidal.
The challenge: Treating Ms. H’s depression and paranoia while avoiding drug-drug interactions.
Discussion: Adverse drug-drug interactions can occur when prescribing psychotropics to patients with IBS (Table 1). Additive constipation, diarrhea, abdominal pain, and sedation are common interactions between psychotropics and the 5-HT3 antagonists and 5HT4 agonists commonly prescribed for IBS.
Table 1
Interactions between psychotropics and agents prescribed for IBS
| Antispasmodics | Benzodiazepines | SSRIs | Tricyclics | |
|---|---|---|---|---|
| MAOIs | Additive sedation | Additive dizziness, sedation, dry mouth, | Contraindicated–hyperpyrexia and severe neurologic effects | Contraindicated–hyperpyrexia, seizures, and death |
| SSRIs | Additive sedation | Additive sedation | —- | Increased tricyclic levels with concurrent use |
| Tricyclics | Additive sedation, dry mouth | Additive sedation | Additive sedation, dry mouth, increased tricyclic levels | —- |
| Anticonvulsants | Additive sedation | Additive sedation | Increased levels of anticonvulsants | Additive sedation, dry mouth, constipation |
| Benzodiazepines | Additive sedation | —- | Additive sedation and dry mouth | Additive sedation |
| Buspirone | Additive sedation, dizziness | Additive sedation | Additive sedation, dizziness, nausea | Additive sedation, dry mouth, constipation, increased tricyclic level |
| Traditional antipsychotics | Additive sedation, CNS effects | Additive sedation, CNS effects | Additive sedation, dizziness | Additive sedation and anticholinergic effects; increased tricyclic level |
| Atypical antipsychotics | Additive sedation, CNS effects | Contraindicated–respiratory and cardiovascular collapse | Elevated antipsychotic levels | Levels of both drugs increased |
| Aripiprazole | Somnolence,constipation | Additive sedation | Increased blood levels of aripiprazole | Increased sedation and anticholinergic effects |
| Psychotropics and 5-HT3 antagonists taken concomitantly typically lead to additive constipation and abdominal pain. | ||||
| Psychotropics and 5-HT4 agonists taken concomitantly typically lead to additive diarrhea and/or abdominal pain. | ||||
| Source: Physician’s Desk Reference. Mobile PDR release version 32. Database version 437. Montvale, NJ: Thomson Healthcare 2003. | ||||
- Hematochezia
- Weight loss < 10 pounds
- Family history of colon cancer
- Recurrent fever
- Anemia
- Chronic severe diarrhea
Source: American College of Gastroenterology Functional Gastrointestinal Disorders Task Force. Am J Gastroenterol. 2002;97:S1-S5.
Other than fiber supplements, most traditional IBS medications are sedating and are associated with anticholinergic side effects. In Ms. H’s case, extreme caution is necessary before prescribing an antidepressant or antipsychotic because of dicyclomine’s additive sedating effects.
Case 2 concluded: After a thorough initial patient interview, the psychiatrist elects to treat Ms. H’s major depression with an antidepressant but delays the use of an antipsychotic to avoid additive sedation.
After talking with Ms. H’s family physician, the psychiatrist stops her dicyclomine and starts sertraline, 100 mg/d. She tolerates the sertraline well and the dosage is titrated across 1 month to 200 mg/d.
Four weeks later, Ms. H’s Beck Depression Inventory score has improved from 26 at baseline to 5, but her paranoid thoughts and frequent diarrhea persist. The psychiatrist adds low-dose olanzapine (5 mg at bedtime) to minimize extrapyramidal side effects. One month later, her depression and paranoia have resolved.
Ms. H’s gastroenterologist instructs her to begin taking alosetron, 1 mg bid, for her continued frequent diarrhea. Adding this agent to her sertraline/olanzapine regimen can lead to additive constipation and abdominal pain, so the psychiatrist monitors her psychiatric medications. One month later, she reports that her affect is much improved and her diarrhea is “gone.”
CASE 3: DEPRESSION AND ABDOMINAL PAIN
Mr. J, age 52, has had depression for 1 year. His depressive symptoms have improved significantly on fluoxetine, 20 mg/d; he once again enjoys life and has a more positive outlook.
The patient was in reasonably good health until about 1 month ago, when he began to experience abdominal pain. He has lost 14 lbs over the past month. He is not taking other medications.
The challenge: Find the cause of Mr. J’s persistent abdominal pain without undermining depression therapy.
Discussion: Although Mr. J’s symptoms might be side effects of fluoxetine, his abdominal pain and weight loss >10 lbs within 1 month are cause for concern. The American College of Gastroenterology has identified six alarm symptoms that could point to a serious medical problem in patients with severe abdominal pain (Box).7
Patients who exhibit any of these symptoms should be referred for endoscopic and stool studies. Colon cancer screening should be considered for all patients age 50 and older.
Patients with IBS usually present first to their primary care physicians with abdominal pain and altered bowel habits. These symptoms can occur in many gastrointestinal and systemic illnesses (Table 2).8
Table 2
Diagnosing irritable bowel syndrome: What to rule out
| Differential diagnosis | Examples |
|---|---|
| Inflammatory bowel disease | Crohn’s disease, ulcerative colitis |
| Medication effects | Laxatives, constipating agents |
| Infections | Parasitic, bacterial, viral, opportunistic |
| Malabsorption syndromes | Celiac disease, pancreatic insufficiency |
| Endocrine disorders | Hypothyroidism, hyperthyroidism, diabetes, Addison’s disease |
| Endocrine tumors (extremely uncommon) | Gastrinoma, carcinoid |
| Colorectal carcinoma | Adenocarcinoma, villous adenoma |
| Intestinal pseudo-obstruction | Diabetes, scleroderma |
| Lactose intolerance | —- |
| Psychiatric disorders | Depression, anxiety, somatization disorders |
| Source: Dalton CB, Drossman D. Am Fam Physician. 1997;55(3):875-80. | |
Case 3 concluded: The psychiatrist and primary care physician consult a gastroenterologist, who performs a colonoscopy and identifies a resectable Duke’s Class B adenocarcinoma in the transverse colon. A partial colectomy is performed.
Three years later, Mr. J is cancer-free and his depression is stable. The psychiatrist advises him to keep taking fluoxetine, 20 mg/d, because the stress of his cancer therapy increases the risk of depression recurrence.
Related resources
- National Institute of Diabetes and Digestive and Kidney Diseases —Irritable Bowel Syndrome www.niddk.nih.gov/health/digest/pubs/irrbowel/irrbowel.htm
Drug brand names
- Alosetron • Lotronex
- Aripiprazole • Abilify
- Buspirone • BuSpar
- Desipramine • Norpramin
- Dicyclomine • Bentyl
- Fluoxetine • Prozac
- Olanzapine • Zyprexa
- Sertraline • Zoloft
Disclosure
The author reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Locke GR, 3rd. The epidemiology of functional gastrointestinal disorders in North America. Gastroenterol Clin North Am. 1996;25:1-19.
2. Goldberg J, Davidson P. A biopsychosocial understanding of the irritable bowel syndrome: a review. Can J Psychiatry. 1997;42:835-40.
3. Aaron LA, Burke MM, Buchwald D. Overlapping conditions among chronic fatigue syndrome, fibromyalgia, and temporomandibular disorder. Arch Intern Med. 2000;160:221-7.
4. Smith RP. Lower gastrointestinal disease in women. Obstet Gynecol Clin North Am. 2001;28:351-62.
5. Olden KW, Drossman DA. Psychologic and psychiatric aspects of gastrointestinal disease. Med Clin North Am. 2000;84:1313-276.
6. Mobile PDR Release Version 32. Database Version 437. An abbreviated, up-to-date version of the PDR onto computing devices. Thomson Healthcare, Ortho-Biotech Oncology, 2003.
7. American College of Gastroenterology Functional Gastrointestinal Disorders Task Force. Evidence-based position statement on the management of irritable bowel syndrome in North America. Am J Gastroenterol. 2002;97:S1-S5.
8. Dalton CB, Drossman D. Diagnosis and treatment of irritable bowel syndrome. Am Fam Physician. 1997;55(3):875-80.
Psychiatrists often treat patients with irritable bowel syndrome (IBS) and an accompanying mental illness. Knowledge of available treatments and communication with the referring doctor are crucial to treating both the IBS symptoms and the comorbidity.
This article presents three cases that illustrate the challenges of identifying target symptoms, avoiding drug-drug interactions, ruling out serious underlying medical problems, and formulating treatment.
WHO GETS IBS?
Approximately 12% of the United States population reports IBS symptoms (abdominal pain, bloating, altered bowel habits).1 These symptoms begin before age 35 in most patients and during childhood in some. Onset after age 65 is rare.
IBS is common among patients with alcohol abuse disorder (32%),2 chronic fatigue syndrome (92%), fibromyalgia (77%), or temporomandibular joint syndrome (64%).3 Seventy percent of patients with IBS are women.4 Chronic pelvic pain, dyspareunia, dysmenorrhea, or a history of abdominal surgeries are risk factors for IBS in women.
LINK BETWEEN IBS AND MENTAL ILLNESS
Although mental illness often coexists with IBS, no cause-effect relationship has been shown.5
IBS is often preceded by stressful life events, such as family death or divorce,3 and some believe IBS is a precursor to numerous psychiatric disorders. Generalized anxiety disorder, major depression, panic disorder, social phobia, somatization disorder, or dysthymia have been diagnosed in most IBS patients.2
CASE 1: IBS AND DEPRESSION
Ms. R, age 55, has had IBS for 10 years. She has occasional diarrhea and abdominal cramps relieved by bowel movements. She is taking a bulking agent but still sometimes suffers abdominal pain.
She is referred to a psychiatrist after complaining of fatigue, loss of interest in hobbies, and crying spells for 2 months. She denies suicidal ideations. Her referring physician reports that she is taking conjugated estrogens to manage menopause symptoms. She denies any recent stressful life events. Thyroid function, glucose, and CBC are normal.
The challenge: Deciding which to treat first—the IBS symptoms or the depression—and how.
Discussion: The predominant symptom (in Ms. R’s case, abdominal pain) can help determine choice of medication. Bulk-forming agents, antispasmodics, barbiturates, benzodiazepines, and serotonin reuptake inhibitors have historically been used to treat IBS,6 but scant evidence supports their use.
Obtaining a thorough prescription history from the primary care physician, OB/GYN, and other treatment team members is critical before formulating a treatment plan. Ms. R’s estrogen use will not affect the choice of psychotropic or IBS medication because there are no significant interactions between estrogen and these classes of drugs.
Ms. R’s abdominal pain and depression can be treated simultaneously. Randomized, controlled trials have demonstrated that tricyclic antidepressants reduce abdominal pain and that behavioral therapy (relaxation therapy, hypnotherapy, and cognitive-behavioral therapy) may relieve individual IBS symptoms.7
Case 1 concluded: After reviewing Ms. R’s medications, the psychiatrist starts:
- desipramine, 50 mg at bedtime, to minimize anticholinergic side effects
- and short-term psychotherapy, which helped her identify support mechanisms and ways to better balance her life stresses.
After 6 weeks, her Beck Depression Inventory score improved from 30 at baseline to 8. She reports her abdominal pain is “the best it has been in 10 years.” Six months after diagnosis, she continues to take desipramine and is doing well.
CASE 2: IBS, DEPRESSION, AND PSYCHOSIS
Ms. H, age 32, is referred to a psychiatrist for treatment of depression with paranoid features.
Four years ago, a gastroenterologist diagnosed her as having IBS. She experiences frequent diarrhea and lower abdominal cramping. For 2 years she has been taking the antimuscarinic dicyclomine, 10 mg tid, which has provided some relief from her cramps. An estimated 20 diarrhea attacks per day leaves her housebound much of the time, however.
She reports fatigue, loss of interest in hobbies across 2 months, and paranoid thinking. She denies hallucinations or delusions but believes that her teenage children are discussing her “sickness” and plotting to “drive her crazy.” She is not suicidal.
The challenge: Treating Ms. H’s depression and paranoia while avoiding drug-drug interactions.
Discussion: Adverse drug-drug interactions can occur when prescribing psychotropics to patients with IBS (Table 1). Additive constipation, diarrhea, abdominal pain, and sedation are common interactions between psychotropics and the 5-HT3 antagonists and 5HT4 agonists commonly prescribed for IBS.
Table 1
Interactions between psychotropics and agents prescribed for IBS
| Antispasmodics | Benzodiazepines | SSRIs | Tricyclics | |
|---|---|---|---|---|
| MAOIs | Additive sedation | Additive dizziness, sedation, dry mouth, | Contraindicated–hyperpyrexia and severe neurologic effects | Contraindicated–hyperpyrexia, seizures, and death |
| SSRIs | Additive sedation | Additive sedation | —- | Increased tricyclic levels with concurrent use |
| Tricyclics | Additive sedation, dry mouth | Additive sedation | Additive sedation, dry mouth, increased tricyclic levels | —- |
| Anticonvulsants | Additive sedation | Additive sedation | Increased levels of anticonvulsants | Additive sedation, dry mouth, constipation |
| Benzodiazepines | Additive sedation | —- | Additive sedation and dry mouth | Additive sedation |
| Buspirone | Additive sedation, dizziness | Additive sedation | Additive sedation, dizziness, nausea | Additive sedation, dry mouth, constipation, increased tricyclic level |
| Traditional antipsychotics | Additive sedation, CNS effects | Additive sedation, CNS effects | Additive sedation, dizziness | Additive sedation and anticholinergic effects; increased tricyclic level |
| Atypical antipsychotics | Additive sedation, CNS effects | Contraindicated–respiratory and cardiovascular collapse | Elevated antipsychotic levels | Levels of both drugs increased |
| Aripiprazole | Somnolence,constipation | Additive sedation | Increased blood levels of aripiprazole | Increased sedation and anticholinergic effects |
| Psychotropics and 5-HT3 antagonists taken concomitantly typically lead to additive constipation and abdominal pain. | ||||
| Psychotropics and 5-HT4 agonists taken concomitantly typically lead to additive diarrhea and/or abdominal pain. | ||||
| Source: Physician’s Desk Reference. Mobile PDR release version 32. Database version 437. Montvale, NJ: Thomson Healthcare 2003. | ||||
- Hematochezia
- Weight loss < 10 pounds
- Family history of colon cancer
- Recurrent fever
- Anemia
- Chronic severe diarrhea
Source: American College of Gastroenterology Functional Gastrointestinal Disorders Task Force. Am J Gastroenterol. 2002;97:S1-S5.
Other than fiber supplements, most traditional IBS medications are sedating and are associated with anticholinergic side effects. In Ms. H’s case, extreme caution is necessary before prescribing an antidepressant or antipsychotic because of dicyclomine’s additive sedating effects.
Case 2 concluded: After a thorough initial patient interview, the psychiatrist elects to treat Ms. H’s major depression with an antidepressant but delays the use of an antipsychotic to avoid additive sedation.
After talking with Ms. H’s family physician, the psychiatrist stops her dicyclomine and starts sertraline, 100 mg/d. She tolerates the sertraline well and the dosage is titrated across 1 month to 200 mg/d.
Four weeks later, Ms. H’s Beck Depression Inventory score has improved from 26 at baseline to 5, but her paranoid thoughts and frequent diarrhea persist. The psychiatrist adds low-dose olanzapine (5 mg at bedtime) to minimize extrapyramidal side effects. One month later, her depression and paranoia have resolved.
Ms. H’s gastroenterologist instructs her to begin taking alosetron, 1 mg bid, for her continued frequent diarrhea. Adding this agent to her sertraline/olanzapine regimen can lead to additive constipation and abdominal pain, so the psychiatrist monitors her psychiatric medications. One month later, she reports that her affect is much improved and her diarrhea is “gone.”
CASE 3: DEPRESSION AND ABDOMINAL PAIN
Mr. J, age 52, has had depression for 1 year. His depressive symptoms have improved significantly on fluoxetine, 20 mg/d; he once again enjoys life and has a more positive outlook.
The patient was in reasonably good health until about 1 month ago, when he began to experience abdominal pain. He has lost 14 lbs over the past month. He is not taking other medications.
The challenge: Find the cause of Mr. J’s persistent abdominal pain without undermining depression therapy.
Discussion: Although Mr. J’s symptoms might be side effects of fluoxetine, his abdominal pain and weight loss >10 lbs within 1 month are cause for concern. The American College of Gastroenterology has identified six alarm symptoms that could point to a serious medical problem in patients with severe abdominal pain (Box).7
Patients who exhibit any of these symptoms should be referred for endoscopic and stool studies. Colon cancer screening should be considered for all patients age 50 and older.
Patients with IBS usually present first to their primary care physicians with abdominal pain and altered bowel habits. These symptoms can occur in many gastrointestinal and systemic illnesses (Table 2).8
Table 2
Diagnosing irritable bowel syndrome: What to rule out
| Differential diagnosis | Examples |
|---|---|
| Inflammatory bowel disease | Crohn’s disease, ulcerative colitis |
| Medication effects | Laxatives, constipating agents |
| Infections | Parasitic, bacterial, viral, opportunistic |
| Malabsorption syndromes | Celiac disease, pancreatic insufficiency |
| Endocrine disorders | Hypothyroidism, hyperthyroidism, diabetes, Addison’s disease |
| Endocrine tumors (extremely uncommon) | Gastrinoma, carcinoid |
| Colorectal carcinoma | Adenocarcinoma, villous adenoma |
| Intestinal pseudo-obstruction | Diabetes, scleroderma |
| Lactose intolerance | —- |
| Psychiatric disorders | Depression, anxiety, somatization disorders |
| Source: Dalton CB, Drossman D. Am Fam Physician. 1997;55(3):875-80. | |
Case 3 concluded: The psychiatrist and primary care physician consult a gastroenterologist, who performs a colonoscopy and identifies a resectable Duke’s Class B adenocarcinoma in the transverse colon. A partial colectomy is performed.
Three years later, Mr. J is cancer-free and his depression is stable. The psychiatrist advises him to keep taking fluoxetine, 20 mg/d, because the stress of his cancer therapy increases the risk of depression recurrence.
Related resources
- National Institute of Diabetes and Digestive and Kidney Diseases —Irritable Bowel Syndrome www.niddk.nih.gov/health/digest/pubs/irrbowel/irrbowel.htm
Drug brand names
- Alosetron • Lotronex
- Aripiprazole • Abilify
- Buspirone • BuSpar
- Desipramine • Norpramin
- Dicyclomine • Bentyl
- Fluoxetine • Prozac
- Olanzapine • Zyprexa
- Sertraline • Zoloft
Disclosure
The author reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Psychiatrists often treat patients with irritable bowel syndrome (IBS) and an accompanying mental illness. Knowledge of available treatments and communication with the referring doctor are crucial to treating both the IBS symptoms and the comorbidity.
This article presents three cases that illustrate the challenges of identifying target symptoms, avoiding drug-drug interactions, ruling out serious underlying medical problems, and formulating treatment.
WHO GETS IBS?
Approximately 12% of the United States population reports IBS symptoms (abdominal pain, bloating, altered bowel habits).1 These symptoms begin before age 35 in most patients and during childhood in some. Onset after age 65 is rare.
IBS is common among patients with alcohol abuse disorder (32%),2 chronic fatigue syndrome (92%), fibromyalgia (77%), or temporomandibular joint syndrome (64%).3 Seventy percent of patients with IBS are women.4 Chronic pelvic pain, dyspareunia, dysmenorrhea, or a history of abdominal surgeries are risk factors for IBS in women.
LINK BETWEEN IBS AND MENTAL ILLNESS
Although mental illness often coexists with IBS, no cause-effect relationship has been shown.5
IBS is often preceded by stressful life events, such as family death or divorce,3 and some believe IBS is a precursor to numerous psychiatric disorders. Generalized anxiety disorder, major depression, panic disorder, social phobia, somatization disorder, or dysthymia have been diagnosed in most IBS patients.2
CASE 1: IBS AND DEPRESSION
Ms. R, age 55, has had IBS for 10 years. She has occasional diarrhea and abdominal cramps relieved by bowel movements. She is taking a bulking agent but still sometimes suffers abdominal pain.
She is referred to a psychiatrist after complaining of fatigue, loss of interest in hobbies, and crying spells for 2 months. She denies suicidal ideations. Her referring physician reports that she is taking conjugated estrogens to manage menopause symptoms. She denies any recent stressful life events. Thyroid function, glucose, and CBC are normal.
The challenge: Deciding which to treat first—the IBS symptoms or the depression—and how.
Discussion: The predominant symptom (in Ms. R’s case, abdominal pain) can help determine choice of medication. Bulk-forming agents, antispasmodics, barbiturates, benzodiazepines, and serotonin reuptake inhibitors have historically been used to treat IBS,6 but scant evidence supports their use.
Obtaining a thorough prescription history from the primary care physician, OB/GYN, and other treatment team members is critical before formulating a treatment plan. Ms. R’s estrogen use will not affect the choice of psychotropic or IBS medication because there are no significant interactions between estrogen and these classes of drugs.
Ms. R’s abdominal pain and depression can be treated simultaneously. Randomized, controlled trials have demonstrated that tricyclic antidepressants reduce abdominal pain and that behavioral therapy (relaxation therapy, hypnotherapy, and cognitive-behavioral therapy) may relieve individual IBS symptoms.7
Case 1 concluded: After reviewing Ms. R’s medications, the psychiatrist starts:
- desipramine, 50 mg at bedtime, to minimize anticholinergic side effects
- and short-term psychotherapy, which helped her identify support mechanisms and ways to better balance her life stresses.
After 6 weeks, her Beck Depression Inventory score improved from 30 at baseline to 8. She reports her abdominal pain is “the best it has been in 10 years.” Six months after diagnosis, she continues to take desipramine and is doing well.
CASE 2: IBS, DEPRESSION, AND PSYCHOSIS
Ms. H, age 32, is referred to a psychiatrist for treatment of depression with paranoid features.
Four years ago, a gastroenterologist diagnosed her as having IBS. She experiences frequent diarrhea and lower abdominal cramping. For 2 years she has been taking the antimuscarinic dicyclomine, 10 mg tid, which has provided some relief from her cramps. An estimated 20 diarrhea attacks per day leaves her housebound much of the time, however.
She reports fatigue, loss of interest in hobbies across 2 months, and paranoid thinking. She denies hallucinations or delusions but believes that her teenage children are discussing her “sickness” and plotting to “drive her crazy.” She is not suicidal.
The challenge: Treating Ms. H’s depression and paranoia while avoiding drug-drug interactions.
Discussion: Adverse drug-drug interactions can occur when prescribing psychotropics to patients with IBS (Table 1). Additive constipation, diarrhea, abdominal pain, and sedation are common interactions between psychotropics and the 5-HT3 antagonists and 5HT4 agonists commonly prescribed for IBS.
Table 1
Interactions between psychotropics and agents prescribed for IBS
| Antispasmodics | Benzodiazepines | SSRIs | Tricyclics | |
|---|---|---|---|---|
| MAOIs | Additive sedation | Additive dizziness, sedation, dry mouth, | Contraindicated–hyperpyrexia and severe neurologic effects | Contraindicated–hyperpyrexia, seizures, and death |
| SSRIs | Additive sedation | Additive sedation | —- | Increased tricyclic levels with concurrent use |
| Tricyclics | Additive sedation, dry mouth | Additive sedation | Additive sedation, dry mouth, increased tricyclic levels | —- |
| Anticonvulsants | Additive sedation | Additive sedation | Increased levels of anticonvulsants | Additive sedation, dry mouth, constipation |
| Benzodiazepines | Additive sedation | —- | Additive sedation and dry mouth | Additive sedation |
| Buspirone | Additive sedation, dizziness | Additive sedation | Additive sedation, dizziness, nausea | Additive sedation, dry mouth, constipation, increased tricyclic level |
| Traditional antipsychotics | Additive sedation, CNS effects | Additive sedation, CNS effects | Additive sedation, dizziness | Additive sedation and anticholinergic effects; increased tricyclic level |
| Atypical antipsychotics | Additive sedation, CNS effects | Contraindicated–respiratory and cardiovascular collapse | Elevated antipsychotic levels | Levels of both drugs increased |
| Aripiprazole | Somnolence,constipation | Additive sedation | Increased blood levels of aripiprazole | Increased sedation and anticholinergic effects |
| Psychotropics and 5-HT3 antagonists taken concomitantly typically lead to additive constipation and abdominal pain. | ||||
| Psychotropics and 5-HT4 agonists taken concomitantly typically lead to additive diarrhea and/or abdominal pain. | ||||
| Source: Physician’s Desk Reference. Mobile PDR release version 32. Database version 437. Montvale, NJ: Thomson Healthcare 2003. | ||||
- Hematochezia
- Weight loss < 10 pounds
- Family history of colon cancer
- Recurrent fever
- Anemia
- Chronic severe diarrhea
Source: American College of Gastroenterology Functional Gastrointestinal Disorders Task Force. Am J Gastroenterol. 2002;97:S1-S5.
Other than fiber supplements, most traditional IBS medications are sedating and are associated with anticholinergic side effects. In Ms. H’s case, extreme caution is necessary before prescribing an antidepressant or antipsychotic because of dicyclomine’s additive sedating effects.
Case 2 concluded: After a thorough initial patient interview, the psychiatrist elects to treat Ms. H’s major depression with an antidepressant but delays the use of an antipsychotic to avoid additive sedation.
After talking with Ms. H’s family physician, the psychiatrist stops her dicyclomine and starts sertraline, 100 mg/d. She tolerates the sertraline well and the dosage is titrated across 1 month to 200 mg/d.
Four weeks later, Ms. H’s Beck Depression Inventory score has improved from 26 at baseline to 5, but her paranoid thoughts and frequent diarrhea persist. The psychiatrist adds low-dose olanzapine (5 mg at bedtime) to minimize extrapyramidal side effects. One month later, her depression and paranoia have resolved.
Ms. H’s gastroenterologist instructs her to begin taking alosetron, 1 mg bid, for her continued frequent diarrhea. Adding this agent to her sertraline/olanzapine regimen can lead to additive constipation and abdominal pain, so the psychiatrist monitors her psychiatric medications. One month later, she reports that her affect is much improved and her diarrhea is “gone.”
CASE 3: DEPRESSION AND ABDOMINAL PAIN
Mr. J, age 52, has had depression for 1 year. His depressive symptoms have improved significantly on fluoxetine, 20 mg/d; he once again enjoys life and has a more positive outlook.
The patient was in reasonably good health until about 1 month ago, when he began to experience abdominal pain. He has lost 14 lbs over the past month. He is not taking other medications.
The challenge: Find the cause of Mr. J’s persistent abdominal pain without undermining depression therapy.
Discussion: Although Mr. J’s symptoms might be side effects of fluoxetine, his abdominal pain and weight loss >10 lbs within 1 month are cause for concern. The American College of Gastroenterology has identified six alarm symptoms that could point to a serious medical problem in patients with severe abdominal pain (Box).7
Patients who exhibit any of these symptoms should be referred for endoscopic and stool studies. Colon cancer screening should be considered for all patients age 50 and older.
Patients with IBS usually present first to their primary care physicians with abdominal pain and altered bowel habits. These symptoms can occur in many gastrointestinal and systemic illnesses (Table 2).8
Table 2
Diagnosing irritable bowel syndrome: What to rule out
| Differential diagnosis | Examples |
|---|---|
| Inflammatory bowel disease | Crohn’s disease, ulcerative colitis |
| Medication effects | Laxatives, constipating agents |
| Infections | Parasitic, bacterial, viral, opportunistic |
| Malabsorption syndromes | Celiac disease, pancreatic insufficiency |
| Endocrine disorders | Hypothyroidism, hyperthyroidism, diabetes, Addison’s disease |
| Endocrine tumors (extremely uncommon) | Gastrinoma, carcinoid |
| Colorectal carcinoma | Adenocarcinoma, villous adenoma |
| Intestinal pseudo-obstruction | Diabetes, scleroderma |
| Lactose intolerance | —- |
| Psychiatric disorders | Depression, anxiety, somatization disorders |
| Source: Dalton CB, Drossman D. Am Fam Physician. 1997;55(3):875-80. | |
Case 3 concluded: The psychiatrist and primary care physician consult a gastroenterologist, who performs a colonoscopy and identifies a resectable Duke’s Class B adenocarcinoma in the transverse colon. A partial colectomy is performed.
Three years later, Mr. J is cancer-free and his depression is stable. The psychiatrist advises him to keep taking fluoxetine, 20 mg/d, because the stress of his cancer therapy increases the risk of depression recurrence.
Related resources
- National Institute of Diabetes and Digestive and Kidney Diseases —Irritable Bowel Syndrome www.niddk.nih.gov/health/digest/pubs/irrbowel/irrbowel.htm
Drug brand names
- Alosetron • Lotronex
- Aripiprazole • Abilify
- Buspirone • BuSpar
- Desipramine • Norpramin
- Dicyclomine • Bentyl
- Fluoxetine • Prozac
- Olanzapine • Zyprexa
- Sertraline • Zoloft
Disclosure
The author reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Locke GR, 3rd. The epidemiology of functional gastrointestinal disorders in North America. Gastroenterol Clin North Am. 1996;25:1-19.
2. Goldberg J, Davidson P. A biopsychosocial understanding of the irritable bowel syndrome: a review. Can J Psychiatry. 1997;42:835-40.
3. Aaron LA, Burke MM, Buchwald D. Overlapping conditions among chronic fatigue syndrome, fibromyalgia, and temporomandibular disorder. Arch Intern Med. 2000;160:221-7.
4. Smith RP. Lower gastrointestinal disease in women. Obstet Gynecol Clin North Am. 2001;28:351-62.
5. Olden KW, Drossman DA. Psychologic and psychiatric aspects of gastrointestinal disease. Med Clin North Am. 2000;84:1313-276.
6. Mobile PDR Release Version 32. Database Version 437. An abbreviated, up-to-date version of the PDR onto computing devices. Thomson Healthcare, Ortho-Biotech Oncology, 2003.
7. American College of Gastroenterology Functional Gastrointestinal Disorders Task Force. Evidence-based position statement on the management of irritable bowel syndrome in North America. Am J Gastroenterol. 2002;97:S1-S5.
8. Dalton CB, Drossman D. Diagnosis and treatment of irritable bowel syndrome. Am Fam Physician. 1997;55(3):875-80.
1. Locke GR, 3rd. The epidemiology of functional gastrointestinal disorders in North America. Gastroenterol Clin North Am. 1996;25:1-19.
2. Goldberg J, Davidson P. A biopsychosocial understanding of the irritable bowel syndrome: a review. Can J Psychiatry. 1997;42:835-40.
3. Aaron LA, Burke MM, Buchwald D. Overlapping conditions among chronic fatigue syndrome, fibromyalgia, and temporomandibular disorder. Arch Intern Med. 2000;160:221-7.
4. Smith RP. Lower gastrointestinal disease in women. Obstet Gynecol Clin North Am. 2001;28:351-62.
5. Olden KW, Drossman DA. Psychologic and psychiatric aspects of gastrointestinal disease. Med Clin North Am. 2000;84:1313-276.
6. Mobile PDR Release Version 32. Database Version 437. An abbreviated, up-to-date version of the PDR onto computing devices. Thomson Healthcare, Ortho-Biotech Oncology, 2003.
7. American College of Gastroenterology Functional Gastrointestinal Disorders Task Force. Evidence-based position statement on the management of irritable bowel syndrome in North America. Am J Gastroenterol. 2002;97:S1-S5.
8. Dalton CB, Drossman D. Diagnosis and treatment of irritable bowel syndrome. Am Fam Physician. 1997;55(3):875-80.